Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2005
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to .
Commission File Number: 000-50855
Auxilium Pharmaceuticals, Inc.
(Exact name of registrant as specified in its charter)
| DELAWARE | 23-3016883 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
40 Valley Stream Parkway Malvern, PA | 19355 | |
| (Address of principal executive offices) | (Zip Code) | |
(484) 321-5900
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Securities Exchange Act of 1934:
None
Securities registered pursuant to Section 12(g) of the Securities Exchange Act of 1934:
Common Stock, Par Value $0.01 Per Share
(Title of class)
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer and large accelerated filer” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ¨ Accelerated filer ¨ Non-accelerated filer x
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant, based on the last sale price for such stock on the NASDAQ National Market as of June 30, 2005, was approximately $58.2 million.
As of March 27, 2006, the number of shares outstanding of the issuer’s common stock, $0.01 par value per share, was 29,291,719.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the Registrant’s definitive Proxy Statement for its Annual Meeting of Stockholders to be held on June 6, 2006, to be filed with the Securities and Exchange Commission, are incorporated by reference into Part III of this Form 10-K.
Table of Contents
AUXILIUM PHARMACEUTICALS, INC.
FORM 10-K
December 31, 2005
TABLE OF CONTENTS
| Page | ||||
| PART I | ||||
| Item 1. | 3 | |||
| Item 1A. | 28 | |||
| Item 1B. | 54 | |||
| Item 2. | 54 | |||
| Item 3. | 54 | |||
| Item 4. | 54 | |||
| PART II | ||||
| Item 5. | 55 | |||
| Item 6. | 56 | |||
| Item 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 57 | ||
| Item 7A. | 73 | |||
| Item 8. | 74 | |||
| Item 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure | 112 | ||
| Item 9A. | 112 | |||
| Item 9B. | 115 | |||
| PART III | ||||
| Item 10. | 116 | |||
| Item 11. | 116 | |||
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 116 | ||
| Item 13. | 116 | |||
| Item 14. | 116 | |||
| PART IV | ||||
| Item 15. | 117 | |||
| 118 | ||||
| 119 | ||||
Exhibit 31.1 | 123 | |||
Exhibit 31.2 | 124 | |||
Exhibit 32 | 125 | |||
2
Table of Contents
This Report on Form 10-K contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, that involve risks and uncertainties. All statements other than statements of historical information provided herein are forward-looking statements and may contain projections relating to financial results, economic conditions, trends and known uncertainties. These statements are not guarantees of future performance or events. Our actual results may differ materially from those discussed in the Report. You should review the “Risk Factors” section, of this Report for a discussion of the important factors that could cause actual results to differ materially from those described in or implied by the forward-looking statements contained in this Report. Readers are cautioned not to place undue reliance on these forward-looking statements, which reflect management’s analysis, judgment, belief or expectation only as of the date hereof. We undertake no obligation to publicly reissue these forward-looking statements to reflect events or circumstances that arise after the date hereof.
PART I
| ITEM 1. | Business |
Company Overview
Company Description
We are a specialty pharmaceutical company with a focus on developing and marketing products for urology, sexual health and other indications within specialty markets. We currently have a sales and marketing organization of approximately 120 people. Our only marketed product, Testim®, is a proprietary, topical 1% testosterone once-a-day gel indicated for the treatment of hypogonadism. Hypogonadism is defined as reduced or absent secretion of testosterone which can lead to symptoms such as loss of libido, adverse changes in body composition, irritability and poor concentration. We reported revenues in 2005 of $42.8 million, an increase of 58.5% over the $27.0 million reported in 2004. According to National Prescription Audit data from IMS Health, Inc. or IMS, a pharmaceutical market research firm, Testim’s share of total prescriptions for the gel segment of the testosterone replacement therapy, or TRT, market was 16.3% for the month of December 2005, compared with 11.5% for the month of December 2004. For the full year 2005, Testim’s share of total prescriptions for the TRT market was 13.8%, versus 10.0% for the full year 2004.
We target diseases with unmet medical needs that we believe can be addressed by more innovative, effective or easy to use products. Our current product pipeline includes:
| • | Five projects in clinical development; |
| • | Two pain product candidates in pre-clinical development; and |
| • | Rights to six additional pain product candidates, options to additional indications for our injectable enzyme and other products for urology and sexual health. |
Our lead project is AA4500, an injectable enzyme. We have an option to license all non-topical uses of AA4500, and we currently are pursuing three indications: Dupuytren’s Disease, Peyronie’s Disease and Adhesive Capsulitis, or Frozen Shoulder Syndrome. We believe that AA4500 has completed Phase II of development for the treatment of Dupuytren’s Disease and is in Phase II of development for the treatment of Peyronie’s Disease and Frozen Shoulder Syndrome. Each of these disease states is characterized by patients suffering the debilitating effects and deformities of scar tissue. AA4500 was identified for in-licensing because of its application for the treatment of Peyronie’s Disease, which is a plaque or hard scar on the shaft of the penis that causes substantial distortion of the penis upon erection. Men suffering from Peyronie’s Disease are often unable to engage in sexual intercourse due to the deformity of their erection.
In the process of pursuing AA4500 for the treatment of Peyronie’s Disease, we saw completed clinical work and an opportunity to pursue AA4500 for the treatment of Dupuytren’s Disease. Dupuytren’s Disease results
3
Table of Contents
when a patient has excess scar tissue that collects at the tendons in the hand. Typically it results in forming a “cord” affecting the ring and small finger, causing the finger to contract, leaving the patient unable to fully extend the affected finger(s).
AA4500 was identified as a potential treatment for Frozen Shoulder Syndrome by the licensor of AA4500. We exercised our option to expand our license for AA4500 to include its application for the treatment of Frozen Shoulder Syndrome based on the results of a Phase II clinical trial conducted by the licensor of AA4500 which suggests that local injection of AA4500 is well-tolerated and may be effective in patients suffering from Frozen Shoulder Syndrome. Frozen Shoulder Syndrome is a disease of diminished shoulder motion, characterized by restriction in both active and passive range of motion of the shoulder joint.
We have in-licensed a platform technology for the delivery of hormones and pharmaceutical products through the oral mucosal membrane. AA2600, is a TRT for the treatment of hypogonadism that utilizes this transmucosal film technology. We believe AA2600 has completed Phase II of development and expect to commence Phase III trials early in the second quarter of 2006.
We also have a project, which we refer to as AA4010, and referred to erroneously as AA2010 in a press release issued and during our earnings conference call on March 7, 2006, in Phase I of development for the treatment for overactive bladder using the same transmucosal film delivery system. Overactive bladder is a medical condition affecting both men and women characterized by urinary urgency and increased frequency of urination.
We have two pain products using our transmucosal film delivery system in pre-clinical development. We also have rights to six additional pain products and products for hormone replacement and urologic disease using our transmucosal film delivery system.
Company Strategy
Our goal is to build a leading specialty pharmaceutical company by focusing on disease states that can be addressed through sales to predominantly specialist audiences, such as urologists, endocrinologists, certain targeted primary care physicians and subsets of orthopedic surgeons. We have an experienced development and regulatory team that has a proven track record. We intend to draw upon our track record and experience to successfully develop our product candidates. Our strategy will focus on optimizing Testim’s commercial success, developing and commercializing AA4500 and products utilizing our transmucosal film technology platform.
Optimizing Testim’s Commercial Success. Testim has demonstrated an ability to capture market share in the gel segment of the TRT market. We believe that we can continue to increase sales of Testim by capturing market share from competitive products, expanding the use of TRT through increased physician and patient awareness and benefiting from the on-going demographic changes that could create additional patient candidates for treatment with this product.
| • | Increase Testim market penetration. Through on-going evaluation of physician targeting and greater sales efficiencies, we intend to improve the effectiveness of our sales force. We will continue to work with accredited education groups, thought leaders and professional organizations to further educate physicians about diagnosis and the benefits of TRT, including Testim. In April 2005, we entered into a co-promotion agreement with Oscient Pharmaceuticals Corporation, or Oscient, for the promotion and sale of Testim in the U.S. Oscient commenced promotion of Testim in June 2005 and now details Testim with approximately 300 sales representatives to primary care physicians. Prior to our agreement with Oscient, many of these primary care physicians were not accessible to our specialty sales force. We also have entered into two collaborations with partners for the international commercialization of Testim. Bayer, Inc., or Bayer, plans to market Testim in Canada, and Ipsen Farmaceutica, B.V., or Ipsen, has begun to market Testim in Europe. |
4
Table of Contents
| • | Expand market opportunities for Testim. There are some conditions where low testosterone levels may have an impact on the underlying disease. In the fourth quarter of 2004, we began a clinical study of hypogonadal men with Type II diabetes to evaluate the impact of testosterone normalization on blood glucose control. If the results show that Testim contributes to improvement of glycemic control of hypogonadal men with Type II diabetes, we believe that more physicians and patients will understand that maintaining normalized levels of testosterone has positive health consequences beyond sexual health issues |
| • | Capture the benefits of an aging population.According to the U.S. Census Bureau, there were 30.4 million men aged 45 to 64 and 13.2 million men aged 65 to 84 in 2000, and these figures are expected to increase to 39.5 million and 15.1 million, respectively, by 2010. According to a 2001 article published inThe Journal of Clinical Endocrinology & Metabolism, hypogonadism affects approximately 20% of all U.S. men age 50 and older. As a result, we expect to see an increase in the number of potential patients over the coming years. We expect the percentage of men with hypogonadism seeking and receiving treatment will also increase because we believe men from the baby boom generation are more health conscious and expect to have a higher quality of life in their older years than their predecessors. |
Developing and Commercializing AA4500.We believe that AA4500 represents a significant opportunity for us, and we are devoting what we believe to be the appropriate resources to the development and pre-commercialization of this product. For Dupuytren’s and Peyronie’s Diseases, we believe there is a large unmet medical need for a non-surgical solution to these debilitating diseases and that the data that we have seen to date in our well-controlled Phase II studies is encouraging. The product has received orphan drug designation in the U.S. for both indications, and our preliminary market research leads us to believe that urologists and orthopedic surgeons would be very likely to use the product to delay or avoid surgery. As an alternative to surgery we expect that the product could offer a pharmaco-economic advantage for the payers and patients. We have exclusive worldwide rights to develop, market and sell products containing AA4500, other than dermal formulations labeled for topical administration, for the treatment of Dupytren’s and Peyronie’s Diseases and, as of December 2005, Frozen Shoulder Syndrome.
Developing and Commercializing Transmucosal Film Technology.We believe delivery of pharmaceuticals across the mucosal membrane of the mouth offers important advantages to patients, including the possibility to achieve therapeutic benefits with significantly lower doses of drug, and the possibility of a convenient, once or twice daily dosing regimen.
We believe that the dosage form that we are developing for AA2600 will address many of the issues that previous buccal tablets have encountered. Specifically, our product for delivery of testosterone is very thin and is applied to the gum above the back teeth, compared to the buccal tablet which has a much higher profile and is placed above the front teeth. Our product candidate has a coating on the outside that prevents the cheek from sticking to the film and also seals the drug substance. In our recently completed Phase II trial, 97% of patients stated that the dosage form was desirable (49%) or acceptable (48%). Our market research leads us to believe that there is a percentage of hypogonadal men who do not like to use a topical gel on a daily basis for their therapy. We believe that a transmucosal film would provide a viable treatment alternative for these patients, as well as those patients who require increased doses of a gel formulation to achieve therapeutic benefit.
5
Table of Contents
Testim Opportunity
Hypogonadism market
Hypogonadism , or low testosterone, is a disorder that affects millions of men in the U.S. According to a 2001 article published inThe Journal of Clinical Endocrinology & Metabolism, hypogonadism affects approximately 20% of the U.S. male population over age 50. We estimate there is a similar percentage of affected men in Europe. Hypogonadism also affects a high percentage of men with certain co-morbidities regardless of age. The documented associations include: Type II Diabetes (33%)1, HIV (30%)2, Erectile Dysfunction (18%)3 and up to 74% of men being treated with high doses of opioids for chronic pain4. The effects of low testosterone lead to symptoms such as low energy, diminished libido, irritability and poor concentration. According to a recent study the impact of these symptoms on an afflicted person can be high.

TRT is the standard treatment for hypogonadism. According to IMS the total prescriptions within the TRT market have grown from .97 million to 2.39 million prescriptions (146%) between 2000 and 2005. Before 2000, the TRT market consisted of oral, injectable and patch therapies. In 2000 the first topical gel was introduced, and this product segment has been the driver of much of the total growth in the TRT market. According to IMS, the gel segment accounted for approximately 69% of the total prescriptions in the TRT market in the fourth quarter of 2005. Despite this growth, estimates of the U.S. Food and Drug Administration, or FDA, indicate that only 5% of men with hypogonadism currently receive TRT. According to our physician research, this low diagnosis rate stems primarily from low patient and physician awareness of the symptoms, treatment options and monitoring requirements.
| 1 | Dhindsa S. et al.J. Clin Endocrinology Metab. 2004 |
| 2 | Dobs A.S.Clin Endocrinol Metab. 1998 |
| 3 | Bodie J. et al.J. Urol. 2003 |
| 4 | Daniell H.W.J. Pain 2002 |
6
Table of Contents
Testim has captured a significant portion of the gel prescription growth since its launch in early 2003, and Testim held a 16.3% prescription share of this market in December 2005. For the calendar year 2005, prescriptions of Testim grew by 70,100 compared to 2004. Despite its market leader status, the other gel in this market grew only by 17,000 prescriptions over the same period. Injectable formulations of TRT also grew (15,800 prescriptions), but the number of prescriptions for all other formulations (patches, orals, and buccals) dropped year over year.
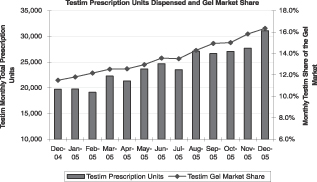 |  |
Source: IMS
We believe Testim’s growth has been due to Testim’s favorable clinical profile and increased sales and marketing efforts in 2005. Our sales organization of nearly 100 representatives promotes Testim to urologists, endocrinologists and key primary care physicians. In April 2005, we contracted with Oscient in order to utilize their then 250-person salesforce to expand our promotional reach to a much larger audience of primary care physicians. Oscient had increased its sales force to approximately 300 by the end of 2005. We believe this effort has helped to improve physician awareness of hypogonadism and to differentiate Testim.
Testim is a proprietary, topical 1% testosterone once-a-day gel that treats hypogonadism by restoring testosterone blood levels back to normal for a 24-hour period following application. Testim is packaged in convenient, easy-to-open, single-use tubes. Patients apply Testim once per day to the upper arms and shoulders, enabling Testim’s active ingredient, testosterone, to be absorbed through the skin and into the bloodstream.
Clinical data supporting the advantages of Testim. We have designed and conducted our clinical trials to provide prescribing physicians with comprehensive information regarding Testim’s benefits. Since 2001, we have completed 13 clinical studies involving 1,600 patients including the largest placebo controlled study (n=400) ever conducted to evaluate the benefits and risks of TRT. We conducted five of these studies using Testim and other TRT products, including a transdermal patch and the other commercial gel. The results from these five studies, taken together, demonstrate that in patients with hypogonadism, testosterone levels directly correlate with patient symptom improvement.
We continue to conduct clinical studies to enhance the Testim data and to address the full patient population that could benefit from its use. For example, we completed a study in over 600 patients in December 2003 that demonstrated that overall sexual function and mood improves significantly by the end of the first week of Testim treatment. We have also initiated a large, multi-centered, placebo controlled study to determine the impact of Testim therapy and testosterone normalization within hypogonadal men who have type II diabetes. A retrospective analysis of our initial studies revealed the fact that the subset of hypogonadal men who had type II diabetes not
7
Table of Contents
only received the typical benefits of testosterone therapy, but also a significant decrease in fasting glucose levels versus placebo. This promising data and the high prevalence of hypogonadism within men with type II diabetes led to the initiation of the more robust, prospective, placebo controlled study.
Cost effectiveness and convenience. We believe, based on clinical data regarding testosterone levels in patients, market feedback from physicians, and the results of our clinical trials, that a higher percentage of patients are treated with only one tube of Testim per day (one dose per day) compared to the other commercial gel. In a recent retrospective pharmacy analysis conducted by Verispan analyzing 12 month usage patterns in over 40,000 patients, we learned that 95% of the Testim treated patients utilized a daily 5 gram dose at month 3 and 97% utilized this dose at month 12. We believe that one tube of Testim per day is an advantage for patients because of less volume of drug to apply, and for payors, because of lower cost.
Broad prescription coverage. Currently, we have broad third-party payor reimbursement for Testim and are covered by the majority of reimbursement plans. Our efforts to achieve third-party payor coverage have expanded as we grow our national accounts group. Testim has advantaged or parity coverage status compared to the other commercial gel with managed care plans covering over 150 million lives. Advantaged coverage generally means lower co-pays for patients.
AA4500 Opportunity
AA4500 is an injectable enzyme we in-licensed from BioSpecifics Technologies Corp., or BioSpecifics. Under our agreement with BioSpecifics, we were granted exclusive worldwide rights to develop, market and sell certain products containing AA4500. Our licensed rights concern the development of products, other than dermal formulations labeled for topical administration. Currently, we are developing AA4500 for the treatment of Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome. We may expand our agreement with BioSpecifics, at our option, to cover other indications as they are developed by us or BioSpecifics.
Dupuytren’s Disease
Disease state and market: Dupuytren’s Disease is a condition that affects the connective tissue that lies beneath the skin in the palm. The disease is progressive in nature. Typically, painful nodules typically develop in the palm as collagen deposits accumulate. As the disease progresses, the collagen deposits form a cord that stretches from the palm of the hand to the base of the finger. Once this cord develops, the patient’s fingers contract and the function of the hand is impaired. Currently, surgery is the only effective treatment. According toDupuytren’s Disease by Leclercq published in 2000, Dupuytren’s Disease is common in Caucasians with diabetes, epilepsy, alcoholism or HIV. It is most common in men and the incidence increases with age. In most patients (65%) the disease affects both hands, and recurrence after surgical treatment is common (as high as 50% over a patient’s lifetime). Due to the recurrent nature of the disease, multiple corrective surgeries are often required, which become increasingly complex. AA4500 is injected directly into the cord and the procedure is performed on an out-patient basis. We believe AA4500 as a treatment for Dupuytren’s Disease represents an attractive and addressable market for us as a specialty pharmaceutical company.
R&D status: Based on Phase II data previously published inThe Journal of Hand Surgery in 2002 utilizing a mean of three injections of AA4500 resulted in a 90% success for the joint closest to the palm, known as the MP joint, and 70% for the middle joint in the fingers, known as the PIP joint. On average patients required 1.5 injections of AA4500. We believe that Phase III studies for AA4500 as a treatment for Dupuytren’s Disease will commence in the middle of 2006.
Peyronie’s Disease
Disease state and market: Peyronie’s Disease is characterized by a plaque or hard lump that occurs on the penis. It begins as a localized inflammation, which progresses to a hardened scar on the shaft of the penis that
8
Table of Contents
reduces flexibility and causes the penis to bend during erection. This may cause pain on erection and a deformity that may prevent sexual intercourse altogether. Our market research indicates that, aside from the physical consequences, depression and loss of self-esteem are common in men with Peyronie’s Disease. According to a 2004 article by Mulhall et. al. inThe Journal of Urology, it is currently believed that Peyronie’s Disease is caused by vascular trauma or injury to the penis. Generally, Peyronie’s Disease is treated by urologists. Some physicians have used injections of Verapamil or Interferon or oral administration of para-aminobenzoic acid with minimal success. Surgical treatment may be an option for some patients, although complications as well as loss of penile length can occur.
AA4500 is injected on an out-patient basis into the plaque causing the penile curvature. Early Phase II clinical work with AA4500 in patients with Peyronie’s Disease indicates that the product reduces scar tissue size, thereby improving the curvature of the penis, and in some cases, restoring the ability to have sexual intercourse. Additional dose optimization studies will be needed.
R&D status: Although previous Stage II studies by the licensor of the product indicate possible effectiveness of AA4500 in this disease state, we believe there is an opportunity to reduce the number of injections required. Therefore, we expect to commence a Phase IIb dose ranging study for AA4500 as a treatment for Peyronie’s Disease in the second half of 2006.
Frozen Shoulder Syndrome
Disease state and market: Frozen Shoulder Syndrome is a disorder of diminished shoulder motion, characterized by restriction in both active and passive range of motion of the shoulder joint. Frozen Shoulder Syndrome usually affects patients aged 40-70 years. According toHarrison’s Principles of Internal Medicine, it is estimated that 3% of people develop Frozen Shoulder Syndrome over their lifetime and that women tend to be affected more frequently than men. The condition may affect both shoulders, either simultaneously or in sequence, in up to 16% of patients. Recurrence of Frozen Shoulder Syndrome is common within five years of the onset of the disorder. According to Dahan et. al. in the 2005 eMedicine article “Adhesive Capsulitis,” a higher incidence of Frozen Shoulder Syndrome exists among patients with diabetes (10-20%) compared to the general population (2-5%) and incidence among patients with insulin-dependent diabetes is even higher (36%), with an increased frequency of bilateral shoulder involvement. The most common treatment for Frozen Shoulder Syndrome is extensive physical therapy, corticosteroids and/or arthroscopy. Drugs are used to control pain, but none have been demonstrated to have an impact on Frozen Shoulder Syndrome. Likewise, medications have not been shown to affect the duration of the disorder or the severity or duration of joint contracture.
R&D status: Our licensor has conducted a Phase II clinical trial using AA4500 for the treatment of Frozen Shoulder. Three different doses of the enzyme were compared to placebo in this prospective, randomized, 60-subject trial. The results from this trial suggest that local injection of the enzyme is well-tolerated and may be effective in patients suffering from Frozen Shoulder Syndrome. Additional studies are needed to assess the optimal dose and dosing regimen of AA4500 for this indication.
Transmucosal Film Technology
General Discussion of Technology
We are developing several product candidates using a new oral transmucosal drug delivery system based on patented technology licensed from Formulation Technologies, L.C.C., doing business as PharmaForm, or PharmaForm. The basis of this technology is a film that adheres to the upper gum. We have the exclusive worldwide right to use this technology platform in therapeutic products that contain hormones or that treat urologic disorders and in certain pain products. We currently have four product candidates using this technology:
| • | AA2600, which is our TRT for the treatment of hypogonadism for which we expect to commence Phase III trials early in the second quarter of 2006; |
9
Table of Contents
| • | AA4010, which is our therapy to treat overactive bladder in Phase I of development for which we expect to seek a partner to further develop the product; and |
| • | two pain product candidates in the pre-clinical phase of development for the management of pain. |
We believe the key benefits of these products candidates could include high absorption, potentially less frequent dosing for some therapeutic drugs and a relatively low cost of goods.
A2600 (Testosterone Replacement Film)
Disease state and market: We are developing AA2600 for the treatment of hypogonadism using the transmucosal film technology. This product candidate may provide an important line extension for the Testim brand. If approved, AA2600 will provide physicians with a novel alternative for the treatment of hypogonadism. We believe its benefits may include the fact that it is small, discrete and easy to use, without the need for drying time as with topical gels. It is our opinion that the most likely patient candidates for this treatment option are those that prefer a dosage form other than those currently available, i.e., gel, patch, injectable or buccal tablet.
R&D status: We have conducted four Phase I studies in Europe with AA2600 in 60 men with low testosterone. Our studies demonstrated that AA2600 was well tolerated and the drug was well absorbed with approximately 80% of the drug delivered and available to the body. AA2600 provided marked increased levels of the androgen within one hour of application. A Phase II proof-of-principle study was initiated in late 2004 in which hypogonadal men were treated for 14 days. In August 2005, we announced that data from the study showed that AA2600 increased serum testosterone levels in hypogonadal men. Furthermore, the data indicated that it was well tolerated and 97% of patients in the study rated the dosage form as desirable or acceptable.
AA4010 (Overactive Bladder Film)
Disease state and market: According to an article published inThe Journal of Urology in August 2002, overactive bladder affects an estimated 13 million people in the U.S. Based on data from the RxList, the internet drug index, we estimate sales of pharmaceutical products for overactive bladder exceeded $1.0 billion in 2004.
A leading oral treatment option for overactive bladder is oxybutynin in both immediate release, or IR, and extended release forms. The published side effects of these drugs are dry mouth, gastrointestinal disorders, difficulty in concentration and visual disturbances. We believe these side effects are impediments to their widespread adoption as a therapy. A recently introduced transdermal patch, which is applied once every three days, appears to have equivalent efficacy to oral medications such as oxybutynin IR but with better tolerability, except for skin reactions at patch application sites, according to an article published inThe Journal of Urology in August 2002.
Because of resources committed to AA4500 and AA2600, and the broad nature of the overactive bladder market, we may seek a partner to further develop this product candidate.
R&D status: We initiated the first Phase I study of AA4010 in December 2004 and commenced patient dosing in January 2005. The study was completed in June 2005 and recent pharmacokinetic results confirmed that oxybutinin is delivered through our licensed transmucosal film delivery system.
Pain Film
Disease state and market: According to the American Pain Foundation, an estimated 75 million Americans suffer from pain every year (50 million from chronic pain and 25 million from acute pain due to injuries or surgery). According to the American Pain Foundation, chronic pain, if left untreated, significantly impacts quality of life — affecting physical, psychological, social and spiritual sense of well-being. According to a 2003 article by Stewart et. al. in theJournal of the American Medical Associations, surveys from various pain
10
Table of Contents
organizations indicate that 1 in 3 American adults lose more than 20 hours of sleep each month due to pain and lost workdays due to pain add up to over 50 million per year. Estimates based on these surveys project that pain costs Americans $100 billion each year, of which $61 billion is attributed to lost productivity.
Pharmacological treatment of pain follows the principles of treatment of any condition. Guidelines from theNational Foundation For The Treatment Of Painindicate that the correct medicine must be selected; the proper doses prescribed; and the risks and side-effects must be carefully monitored and weighed against the benefits. Our efforts are focused around the specialty audience addressed by our sales force. The early products in the pain drug development program address “short-procedure” needs and associated pain management as well as break-through pain and possibly chronic pain in follow-on programs. For applicability of some or all of the eight pain products in areas beyond specialty markets, we are likely to take a partnering approach.
R&D status: In February 2005, we entered into an additional license agreement with PharmaForm, granting us the exclusive right to develop, manufacture and market eight compounds for the management of pain, including acute and chronic pain, using the transmucosal film delivery system. Laboratory scale formulations have been produced for the first two compounds and are currently being evaluated. We believe the next step will be to evaluate the readiness of the formulations for inclusion in Phase I.
Manufacturing
We currently use, and expect to continue to depend on, contract manufacturers to manufacture Testim. We may depend on contract manufacturers to manufacture any products for which we receive marketing approval, and to produce sufficient quantities of our product candidates for use in preclinical and clinical studies.
Testim is manufactured for us by DPT Laboratories, Ltd., or DPT, pursuant to a manufacturing and supply agreement that expires on December 31, 2010 , the terms of which are summarized below in“Material Agreements”.
Testosterone, a non-animal derived hormone, is currently available to us from only two sources. We purchase testosterone from both sources but do not have an agreement with either of these suppliers. We acquire CPD, a critical ingredient for the manufacture of Testim, from a single source. We endeavor to maintain what we believe is an adequate inventory of the key ingredients of Testim.
For AA4500, we are responsible, at our own cost and expense, for developing the formulation and finished dosage form of products and arranging for the clinical supply of products. As permitted under the terms of our agreement with BioSpecifics, we have qualified Cobra Biologics Ltd., or Cobra, as a supplier of AA4500 for use in our clinical trials. The terms of our agreement with Cobra are summarized below under“Material Agreements.”Pursuant to our agreement with Althea Technologies, Inc., or Althea, Althea will fill and lyophilize the AA4500 bulk substance produced by Cobra, manufacture placebo and produce sterile diluent. The terms of our agreement with Althea are summarized below under“Material Agreements.”
Under the terms of our agreement with BioSpecifics, BioSpecifics has the option, exercisable no later than six months after FDA approval of the first New Drug Application or Biologics License Application with respect to a product, to assume the right and obligation to supply, or arrange for the supply from a third party other than a back-up supplier qualified by us, of a specified portion of commercial product required by us. The terms of our agreement with BioSpecifics are summarized below under“Material Agreements.”
Our contract manufacturers are subject to an extensive governmental regulation process. Regulatory authorities in our markets require that drugs be manufactured, packaged and labeled in conformity with current good manufacturing practices, or cGMP. The cGMP requirements govern quality control of the manufacturing process and documentation policies and procedures. We have established an internal quality control and quality assurance program, including a set of standard operating procedures and specifications that we believe is cGMP-compliant.
11
Table of Contents
Material Agreements
We have entered into agreements for the in-licensing of technology and products and for the manufacture of our products. Additionally, we have secured collaboration partners for the sale of Testim in geographic locations where we do not have our own sales force and a co-promotion partner for the sale of Testim in the U.S. We intend to pursue other licensing agreements and collaborations in the future.
Bentley Pharmaceuticals
We entered into two separate license agreements with Bentley Pharmaceuticals, Inc., or Bentley, that give us access to Bentley’s patented transdermal gel technology. In May 2000, Bentley granted us an exclusive, worldwide, royalty-bearing license to make and sell products incorporating Bentley’s formulation technology that contains testosterone. The term of this agreement is determined on a country by country basis and extends until the later of patent right termination in a country or 10 years from the date of first commercial sale. We produce Testim under this license. Bentley has filed new patent applications in the U.S., Europe and Japan, which, if issued, could provide additional patent protection for Testim. In May 2001, Bentley granted us similar rights for a product containing another hormone, the term of which is perpetual. Under these agreements, we are required to make up-front and milestone payments to Bentley upon contract signing, the decision to develop the underlying product, and the receipt of FDA approval.
In addition to our royalty obligations, to date, we have paid Bentley approximately $0.6 million of up-front payments and milestones under the in-license agreements. If all events under our in-license agreements with Bentley occur, we would be obligated to pay a maximum of approximately $1.0 million in milestone payments. The timing of these payments, if any, is uncertain.
DPT
Testim is manufactured for us by DPT under the terms of a manufacturing and supply agreement that we entered into in April 2002 as amended most recently on September 14, 2005. This agreement expires on December 31, 2010. During the term of the agreement, subject to our right to qualify and order Testim from a back-up supplier, and subject to DPT’s ability to qualify its Lakewood, New Jersey facility as a secondary site for the production of Testim, DPT is required to manufacture all of our worldwide commercial requirements of Testim. Prior to DPT’s qualification of the Lakewood facility, we are permitted to qualify a back-up supplier and order a specified amount of Testim each year from a back-up supplier. Upon DPT’s qualification of the Lakewood facility, we will no longer be permitted to order commercial supply of Testim from the back-up supplier. However, in the event that DPT is unable to meet our Testim supply requirements, Testim may be manufactured for us by a back-up supplier. The manufacturing fees paid to DPT are based upon the specified annual volume of Testim ordered by us. If we order less than a specified amount of Testim in any calendar year, the parties are required to negotiate to establish new manufacturing fees, with the understanding that DPT will continue to produce Testim for a specified minimum period in order to enable us to identify and utilize another supplier. DPT may terminate the agreement only upon our insolvency or bankruptcy or upon our breach of the agreement. We are in the process of qualifying an alternate contract manufacturer until DPT’s Lakewood, New Jersey facility has been qualified as a secondary site for the production of Testim.
BioSpecifics
In June 2004, we entered into a development and license agreement with BioSpecifics and amended the agreement in May 2005 and December 2005 (the BioSpecifics Agreement). Under the BioSpecifics Agreement, we were granted exclusive worldwide rights to develop, market and sell certain products containing BioSpecifics’ enzyme, which we refer to as AA4500. Our licensed rights concern the development of products, other than dermal formulations labeled for topical administration, and currently, our licensed rights cover the indications of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome, for which we exercised our
12
Table of Contents
option in December 2005. We may further expand the BioSpecifics Agreement, at our option, to cover other indications as they are developed by us or BioSpecifics.
The BioSpecifics Agreement extends, on a country-by-country and product-by-product basis, for the longer of the patent life, the expiration of any regulatory exclusivity period or 12 years. We may terminate the BioSpecifics Agreement upon 90 days prior written notice.
We are responsible, at our own cost and expense, for developing the formulation and finished dosage form of products and arranging for the clinical supply of products. As permitted under the BioSpecifics Agreement, we have qualified Cobra as our primary supplier of clinical products under the agreement.
BioSpecifics has the option, exercisable no later than six months after FDA approval of the first New Drug Application or Biologics License Application with respect to a product, to assume the right and obligation to supply, or arrange for the supply from a third party other than a back-up supplier qualified by us, of a specified portion of our commercial product requirements. The BioSpecifics Agreement provides that we may withhold a specified amount of a milestone payment until (i) BioSpecifics executes an agreement, containing certain milestones, with a third party for the commercial manufacture of the product, (ii) BioSpecifics commences construction of a facility, compliant with cGMP, for the commercial supply of the product, or (iii) 30 days after BioSpecifics notifies us in writing that it will not exercise the supply option.
If BioSpecifics exercises the supply option, commencing on a specified date, BioSpecifics will be responsible for supplying either by itself or through a third party other than a back-up supplier qualified by us, a specified portion of the commercial supply of the product. If BioSpecifics does not exercise the supply option, we will be responsible for arranging for the entire commercial product supply. In the event BioSpecifics exercises the supply option, we and BioSpecifics are required to use commercially reasonable efforts to enter into a commercial supply agreement on customary and reasonable terms and conditions which are not worse than those with back-up suppliers qualified by us.
We must pay BioSpecifics on a country-by-country and product-by-product basis a specified percentage of net sales for products covered by the BioSpecifics Agreement. Such percentage may vary depending on whether BioSpecifics exercises the supply option. In addition, the percentage may be reduced if (i) BioSpecifics fails to supply commercial product supply in accordance with the terms of the BioSpecifics Agreement; (ii) market share of a competing product exceeds a specified threshold; or (iii) we are required to obtain a license from a third party in order to practice BioSpecifics’ patents without infringing such third party’s patent rights.
In addition to the payments set forth above, we must pay BioSpecifics an amount equal to a specified mark-up of the cost of goods sold for products sold by us that are not manufactured by or on behalf of BioSpecifics, provided that, in the event BioSpecifics exercises the supply option, no payment will be due for so long as BioSpecifics fails to supply the commercial supply of the product in accordance with the terms of the BioSpecifics Agreement.
Finally, we will be obligated to make contingent milestone payments upon the filing of regulatory applications and receipt of regulatory approval. Through December 31, 2005, we paid up-front and milestone payments under the BioSpecifics Agreement of $8.5 million. We could make up to an additional $5.5 million of contingent milestone payments under the BioSpecifics Agreement if all existing conditions are met. Additional milestone obligations will be due if we exercise an option to develop and license AA4500 for additional medical indications.
Cobra
In July 2005 we entered into an agreement with Cobra for the process development, scale up and manufacture of AA4500 for Phase II/III clinical trials (the Cobra Agreement). Accordingly, we will now rely on Cobra as our primary supplier of AA4500 for clinical trials.
13
Table of Contents
The Cobra Agreement is divided into five non-sequential stages with specific objectives, timelines, down payments and total payments that attach to each stage. We may terminate the Cobra Agreement at the completion of each stage if the objective or the timeline for such stage is not met. Through December 31, 2005, we incurred a total of $2.1 million under the Cobra Agreement. If all remaining stages are completed under the current Cobra Agreement, then we will pay Cobra an additional $2.4 million.
Althea
In order to prepare AA4500 for clinical trials, AA4500 bulk substance produced by Cobra must be filled and lyophilized, placebo must be manufactured and a sterile diluent must be produced (collectively, “pharmaceutical products”). On February 28, 2006, we entered into a Manufacturing and Clinical Supply Agreement with Althea to be effective as of January 17, 2006 that sets out the general terms and conditions under which Althea will produce these pharmaceutical products in bulk or finished dosage form for our development and/or clinical use only. At the time we executed the agreement, we and Althea entered into a project plan that provides the parameters for the production of the pharmaceutical products. Pursuant to the project plan we will supply bulk substance of AA4500 produced by Cobra to Althea which, in turn, will produce and deliver to us the pharmaceutical products. The agreement provides that we will pay Althea 50% of the total fees due to Althea upon signing of the project plan. Then, Althea will invoice us monthly for services completed during such month, and the balance of the fees will be invoiced upon delivery of the pharmaceutical products. Upon signing of the project plan in February 2006, we paid to Althea $174,000, and we expect to incur an additional $174,000 under this project plan in 2006.
PharmaForm
In June 2003, we expanded our drug delivery technology portfolio by entering into a license agreement with PharmaForm. Under this agreement, as amended, we were granted an exclusive, worldwide, royalty-bearing license to develop, make and sell products that contain hormones or that are used to treat urologic disorders incorporating PharmaForm’s oral transmucosal film technology for which there is an issued patent in the U.S. The term of this license agreement is for the life of the licensed patents.
In February 2005, we entered into an additional license agreement with PharmaForm. Under this agreement, we were granted exclusive, worldwide royalty-bearing rights to develop, manufacture and market eight compounds using PharmaForm’s proprietary transmucosal film technology for the management of pain, including acute and chronic pain. The compounds that may be developed include opioids as well other types of analgesics. In 2005, we paid an up-front payment of $0.5 million and could make up to an additional $21.2 million in contingent milestone payments if all underlying events for these eight products occur. In addition, we will have on-going royalty payment obligations to PharmaForm. The timing of the remaining payments, if any, is uncertain.
To speed the development of products using the licensed technology from PharmaForm, we entered into a research and development agreement with PharmaForm on a fee for service basis. We will be the sole owner of any intellectual property rights developed in connection with this agreement. We incurred $2.3 million under the research and development agreement in 2005 and expect to incur $3.1 million in 2006.
Bayer
We entered into an exclusive marketing and distribution agreement with Bayer in December 2003 to commercialize Testim in Canada. This agreement is effective for the life of the Bentley patent. The expiration of the Bentley patent in Canada is January 5, 2010. In July 2005, an additional patent covering Testim issued in Canada, and this patent is included in our license from Bentley. This patent expires in 2023. Bayer has a large commercial presence in Canada that includes approximately 450 sales representatives. We filed for regulatory approval of Testim in Canada in March 2004 and will not be able to sell Testim in Canada until regulatory
14
Table of Contents
approval is granted. We have responded to questions raised by the Canadian authorities, and they are reviewing our responses as part of the approval process. The terms of this agreement require Bayer to purchase all Testim supply for Canada from us and to make up-front, milestone and royalty payments. The milestone payments are due upon contract signing, filing for regulatory approval in Canada, the first sale of Testim in Canada and upon achieving specified sales levels. The royalty payments are based on net product revenue and are similar to the royalty payments due to Bentley.
Ipsen
We entered into a license and distribution agreement with Ipsen in March 2004. This agreement is exclusive and royalty-bearing for the longer of ten years from the date of first commercial sale by country or the life of the Bentley patent, including any newly issued patents. The European patent that we license from Bentley for Testim expires in November 2006. Bentley has filed a new patent application in the U.S., Europe and Japan, which, if issued, could provide additional patent protection for Testim. Ipsen is a European pharmaceutical company with a worldwide commercial presence and a strong urology focus. Ipsen currently has operations in 110 countries and has approximately 3,700 employees. Under this agreement, we granted Ipsen an exclusive license to use and sell Testim on a worldwide basis outside the U.S., Canada, Mexico and Japan. The U.K. was the first country outside of the U.S. where regulatory approval to market Testim was obtained. A mutual recognition application based on the U.K. approval was initiated on March 30, 2004 in order to obtain additional European approvals. As of December 31, 2005, Testim was approved for marketing in the U.S., the U.K., Belgium, Denmark, Finland, Germany, Greece, Iceland, Ireland, Italy, Luxembourg, the Netherlands, Norway, Portugal, Spain and Sweden. In 2005, Testim was launched in Belgium, Denmark, Finland, Germany, Ireland, Netherlands, Spain, Sweden, and the U.K. Under the terms of our agreement, Ipsen will seek regulatory approval in those non-E.U. countries in which it wishes to sell our product. The terms of this agreement require Ipsen to purchase all Testim supply from us or exercise their conditional option for a technology transfer and to make up-front, milestone and royalty payments. Future milestone payments will be due upon regulatory approval, product launch in various countries and supply price reductions. The royalty payments are based on net product revenue and are similar to royalty payments due to Bentley.
We do not expect sales of Testim outside of the U.S. pursuant to our collaborations with Bayer and Ipsen to have a material impact on our revenues or profitability. We believe that the primary benefit of these agreements is the demonstration of our global development and commercialization capabilities to future licensors, as well as providing us with up-front and milestone payments and opportunities for manufacturing efficiencies through increased sales of Testim.
Through December 31, 2005, we have collected $11.9 million in up-front and milestone fees from Bayer and Ipsen. We can potentially collect up to an additional $5.7 million in milestone fees under the Ipsen collaboration and up to $1.7 million under the Bayer collaboration, if all underlying events occur. The timing of the above payments, if any, is uncertain.
Oscient Pharmaceuticals
In April 2005, we entered into a co-promotion agreement (the Oscient Agreement) with Oscient. The agreement provides that Oscient will have the exclusive right to co-promote Testim to primary care physicians in the U.S. using its 300-person sales force, while we will continue to promote Testim using our specialty sales force that calls on urologists, endocrinologists and select primary care physicians. The initial term of the Oscient Agreement ends on April 30, 2007. Oscient may extend the Oscient Agreement for two consecutive two-year periods provided that Oscient has met certain milestones for each extension. If these milestones are met and Oscient does not elect to terminate the Oscient Agreement, the first extension period will commence on January 1, 2007 and end on December 31, 2008 and the second extension period will commence effective January 1, 2009 and end on April 30, 2011.
15
Table of Contents
We and Oscient utilize a joint marketing and promotion plan which sets forth the responsibilities of both parties with respect to the marketing and promotion of Testim in the U.S. primary care physician market, and we share equally these promotional expenses. Each party will be responsible for the costs associated with its own sales force. The 2005 combined spending for marketing and promotion of Testim in the primary care physician market in the U.S. was $7.1 million, and we estimate that the total costs for 2006 will be approximately $11.0 million to 13.0 million. These costs will be shared equally.
Oscient will be compensated for its services based on a specified percentage of the gross profit from Testim sales attributable to primary care physicians in the U.S. that exceed a specified sales threshold (the Oscient co-promotion fees). All gross profit earned on sales to primary care physicians below the predetermined threshold and all gross profit earned on all sales to non primary care physicians are not subject to the terms of the Oscient Agreement. Once sales to primary care physicians have exceeded the predetermined sales threshold, the gross profit earned on such sales is available to be split between us and Oscient. Once all agreed upon promotional expenses are appropriately reimbursed to each party, the gross profit remaining will be divided according to agreed upon percentages. The specific percentages are based upon Testim sales levels above the threshold attributable to primary care physicians actually achieved. For the year ended December 31, 2005, we recorded $3.5 million of expense for the Oscient co-promotion fees.
The agreed upon promotional expenses for each year are reimbursable equally and simultaneously to each company through the profit split mechanism, except in 2005 when Oscient was disproportionately reimbursed its share of the promotional expenses through the profit split mechanism. As the agreed upon percentage of the gross profit available to be split in 2005 was insufficient to reimburse Oscient its share of the promotional expenses, that portion not reimbursed to Oscient will be carried over to 2006 and will be reimbursed through the 2006 profit split mechanism. Of the $3.5 million expense recorded by us for the year ended December 31, 2005, $0.6 million was due to this carry over reimbursement.
The Oscient Agreement may be terminated by either party upon the occurrence of certain termination events. We may be obligated to make termination payments in certain instances. Also, Oscient has been granted the exclusive option to co-promote any of our future product candidates that treat hypogonadism and contain testosterone as the active ingredient.
We will invoice and record in our financial statements all sales of Testim, including those sales to primary care physicians that exceed the specified threshold, as well as the gross profit earned on all sales. We will record our share of promotional expenses incurred and the Oscient co-promotion fees as “selling, general & administration” operating expenses in our consolidated statement of operations.
Competition
We face competition in North America, Europe and elsewhere from larger pharmaceutical companies, specialty pharmaceutical companies and biotechnology firms, universities and other research institutions and government agencies that are developing and commercializing pharmaceutical products. Many of our competitors have substantially greater financial, technical and human resources than we have and may, subsequently, develop products that are more effective, safer or less costly than any that have been or are being developed by us or that are generics. Our success will depend on our ability to acquire, develop and commercialize products and our ability to establish and maintain markets for Testim or any products for which we receive marketing approval.
AndroGel
The primary competition for Testim in the TRT market is AndroGel, marketed by Solvay Pharmaceuticals, Inc., or Solvay. Solvay has the advantage of having launched AndroGel three years prior to Testim as well as
16
Table of Contents
having a sales force of approximately 300 individuals. During 2005, Solvay and ICOS Corporation, or ICOS, co-promoted AndroGel. ICOS has reported that it will not continue to co-promote AndroGel in 2006. According to IMS, as of December 31, 2005, AndroGel accounted for 83.7% of the gel prescriptions, although this share declined each quarter in 2005, while Testim’s share of the gel market increased.
Other Delivery Options
Testim also competes with other TRTs such as injectables, patches, orals and a buccal tablet. The injectables have maintained presence in the market due to the low cost compared to the transdermal options. There are also a significant number of patients who prefer getting a shot every two to three weeks instead of utilizing a daily application of another product. Physicians perceive the disadvantage of the injectables as being the fact that many patients’ testosterone levels rise past the normal barrier of 1000 ng/dl which leads to a wide flux of levels in a patient and the potential for side effects such as gynecomastia. Androderm is a transdermal testosterone patch marketed by Watson Pharmaceuticals. Androderm is the leading patch product and accounted for approximately 10% of total TRT prescriptions in December of 2005. The number of Androderm prescriptions is decreasing. We believe this trend may be due to the high number of patients who experience skin rash. Oral therapy has decreased substantially in recent years. We believe this trend may be due to the concern of inconsistent testosterone levels and the chance for liver toxicity. The buccal therapy retains less than 1% share in the market.
Generic Competition
In July 2003, Watson Pharmaceuticals, or Watson, and Par Pharmaceuticals, or Par, filed abbreviated new drug applications, or ANDAs, with the FDA to be approved as generics for AndroGel. In response to these ANDAs, Solvay filed patent infringement lawsuits against these two companies to block the approval and marketing of the generic products. Any generic for AndroGel will not be a generic substitute for Testim. On November 1, 2004, Par Pharmaceuticals’ partner, Paddock Laboratories, or Paddock, received tentative approval of its ANDA from the FDA, and on January 12, 2006, Watson received approval of its ANDA from the FDA. In January 2006, the thirty-month stay in each patent action expired. We believe that Watson was the first to file an ANDA with FDA and, therefore, Watson, not Par/Paddock, is entitled to launch first with six-month exclusivity. As a result, Watson will have to decide whether to (1) launch before a final outcome in the patent litigation and risk treble damages if its product is found to infringe the Solvay patent or (2) wait for the final outcome of the litigation before launch. Any generic for AndroGel will not be a generic for Testim, since Testim is BX rated (or non-substitutable) with Androgel. However if one of the companies chooses to market its product, this would likely result in increased competition for Testim, most likely, at lower prices. Other pharmaceutical companies may develop generic versions of any products that we commercialize that are not subject to patent protection or other proprietary rights. Governmental and other cost containment pressures may result in physicians writing prescriptions for these generic products.
Future Delivery Options
In addition to potential generic competition, several other pharmaceutical companies have TRT products in development that may be approved for marketing in the U.S. Cellegy Pharmaceuticals Inc. submitted a new drug application, or NDA, to the FDA for the approval of a 2% testosterone gel that the FDA rejected in 2003. In addition, BioSante Pharmaceuticals Inc. licensed a testosterone gel in 2000 which is currently reported to be in late stage development. MacroChem Corp. has also developed a testosterone gel that is currently in Phase I studies. Ardana plc, a U.K. company, has announced plans to develop a testosterone cream for the U.S. market. Other new treatments are being sought for TRT, including a new isomer of an old product for centrally mediated treatment of primary hypogonadism as well as a new class of drugs called Selective Androgen Receptor Modulators, or SARMs. These products are in the early stages of development and their future impact on the treatment of testosterone deficiency is unknown.
17
Table of Contents
It has also been made public that Indevus Pharmaceuticals, Inc. has licensed a long acting testosterone injection that is currently marketed in Europe under the name of Nebido. We believe that this product could be launched in the U.S. in late 2007 or early 2008. This product may enhance compliance with some patients and should be considered as competition to Testim if it were to launch. Another potential introduction into the marketplace is an oral therapy, called Androxal, from Zonagen, Inc., or Zonagen. This molecule is designed to restore normal testosterone production in males rather than externally replacing testosterone like the current alternatives. According to Zonagen, an NDA may be filed for this product in 2008.
Finally, we face extensive competition in the acquisition or in-licensing of pharmaceutical products to enhance our portfolio of products. A number of more established companies, which have strategies to in-license or acquire products, may have competitive advantages as may other emerging companies taking similar or different approaches to product acquisitions. In addition, a number of established research-based pharmaceutical and biotechnology companies may acquire products in late stages of development to augment their internal product lines. These established companies may have a competitive advantage over us due to their size, resources and experience.
Government Regulation
Government authorities in the U.S., at the federal, state, and local level, and foreign countries extensively regulate, among other things, the following areas relating to our products and product candidates:
| • | research and development; |
| • | testing, manufacture, labeling and distribution; |
| • | advertising, promotion, sampling and marketing; and |
| • | import and export. |
All of our products require regulatory approval by government agencies prior to commercialization. In particular, human therapeutic products are subject to rigorous preclinical and clinical trials to demonstrate safety and efficacy and other approval procedures of the FDA and similar regulatory authorities in foreign countries. Various federal, state, local, and foreign statutes and regulations also govern testing, manufacturing, labeling, distribution, storage and record-keeping related to such products and their promotion and marketing. The process of obtaining these approvals and the compliance with federal, state, local, and foreign statutes and regulations require the expenditure of substantial time and financial resources. In addition, the current regulatory and political environment at FDA could lead to increased testing and data requirements which could impact regulatory timelines and costs.
We have received marketing approval for Testim for the indication of hypogonadism in the U.S., the U.K., Belgium, Denmark, Finland, Germany, Greece, Iceland, Ireland, Italy, Luxembourg, the Netherlands, Norway, Portugal, Spain and Sweden. Approval in the specified European countries was via the Mutual Recognition Procedure. We have applied for marketing approval for Testim in Canada. We have responded to questions raised by the Canadian authorities as part of the approval process. In Europe, Testim was launched in Belgium, Denmark, Finland, Germany, Ireland, Netherlands, Spain, Sweden, and the UK in 2005. None of our other product candidates has received marketing approval.
U.S. Government Regulation
In the U.S., the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act and implementing regulations. If we fail to comply with the applicable requirements at any time during the product development process, approval process, or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include:
| • | refusal to approve pending applications; |
18
Table of Contents
| • | withdrawals of approvals; |
| • | clinical holds; |
| • | warning letters; |
| • | product recalls and product seizures; and |
| • | total or partial suspension of our operations, injunctions, fines, civil penalties or criminal prosecution. |
Any agency enforcement action could have a material adverse effect on us.
Currently there is a substantial amount of congressional and administration review of the FDA and the regulatory approval process for drug candidates in the U.S. As a result, there may be significant changes made to the regulatory approval process in the U.S.
The steps required before a drug may be marketed in the U.S.include:
| • | preclinical laboratory tests, animal studies and formulation studies; |
| • | submission to the FDA of an investigational new drug exemption, or IND, which must become effective before human clinical trials may begin; |
| • | execution of adequate and well-controlled clinical trials to establish the safety and efficacy of the product for each indication for which approval is sought; |
| • | submission to the FDA of a new drug application (NDA), or biologics license application (BLA); |
| • | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP; and |
| • | FDA review and approval of the NDA or BLA, or any supplements thereto, including, if applicable, a determination of its controlled substance schedule. |
Preclinical studies generally are conducted in laboratory animals to evaluate the potential safety and activity of a product. Violation of the FDA’s good laboratory practices regulations can, in some cases, lead to invalidation of the studies, requiring these studies to be replicated. In the U.S., drug developers submit the results of preclinical trials, together with manufacturing information and analytical and stability data, to the FDA as part of the IND, which must become effective before clinical trials can begin in the U.S. An IND becomes effective 30 days after receipt by the FDA unless before that time the FDA raises concerns or questions about the proposed clinical trials outlined in the IND. In that case, the IND sponsor and the FDA must resolve any outstanding FDA concerns or questions before clinical trials can proceed. If these concerns or questions are unresolved, the FDA may not allow the clinical trials to commence.
19
Table of Contents
Clinical trials involve the administration of the investigational product candidate or approved products to human subjects under the supervision of qualified investigators. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in assessing the safety and the effectiveness of the drug. Typically, clinical evaluation involves a time-consuming and costly three-phase sequential process, but the phases may overlap. Each trial must be reviewed, approved and conducted under the auspices of an independent institutional review board, and each trial must include the patient’s informed consent. The following sets forth a brief description of the typical phases of clinical trials:
Phase I | Refers typically to closely monitored clinical trials and includes the initial introduction of an investigational new drug into human patients or healthy volunteer subjects. Phase I clinical trials are designed to determine the safety, metabolism and pharmacologic actions of a drug in humans, the potential side effects of the product candidates associated with increasing drug doses and, if possible, to gain early evidence of the product candidate’s effectiveness. Phase I trials also include the study of structure-activity relationships and mechanism of action in humans, as well as studies in which investigational drugs are used as research tools to explore biological phenomena or disease processes. During Phase I clinical trials, sufficient information about a drug’s pharmacokinetics and pharmacological effects should be obtained to permit the design of well-controlled, scientifically valid Phase II studies. The total number of subjects and patients included in Phase I clinical trials varies, but is generally in the range of 20 to 80 people. | |
Phase II | Refers to controlled clinical trials conducted to evaluate appropriate dosage and the effectiveness of a drug for a particular indication or indications in patients with a disease or condition under study and to determine the common short-term side effects and risks associated with the drug. These clinical trials are typically well-controlled, closely monitored and conducted in a relatively small number of patients, usually involving no more than several hundred subjects. Phase II studies can be sequenced as Phase IIa or Phase IIb. | |
Phase III | Refers to expanded controlled and uncontrolled clinical trials. These clinical trials are performed after preliminary evidence suggesting effectiveness of a drug has been obtained. Phase III clinical trials are intended to gather additional information about the effectiveness and safety that is needed to evaluate the overall benefit-risk relationship of the drug and to provide an adequate basis for physician labeling. Phase III trials usually include from several hundred to several thousand subjects. | |
Phase IV | Refers to trials conducted after approval of a new drug and which explore approved uses and approved doses of the product. These trials must also be approved and conducted under the auspices of an institutional review board. Phase IV studies may be required as a condition of approval. | |
Clinical testing may not be completed successfully within any specified time period, if at all. The FDA closely monitors the progress of each of the first three phases of clinical trials that are conducted in the U.S. and may, at its discretion, reevaluate, alter, suspend or terminate the testing based upon the data accumulated to that point and the FDA’s assessment of the risk/benefit ratio to the patient. The FDA can also provide specific guidance on the acceptability of protocol design for clinical trials. The FDA or we may suspend or terminate clinical trials at any time for various reasons, including a finding that the subjects or patients are being exposed to an unacceptable health risk. The FDA can also request additional clinical trials be conducted as a condition to product approval. During all clinical trials, physicians monitor the patients to determine effectiveness and to observe and report any reactions or other safety risks that may result from use of the drug candidate.
Assuming successful completion of the required clinical trial, drug developers submit the results of preclinical studies and clinical trials, together with other detailed information including information on the
20
Table of Contents
chemistry, manufacture and control of the product, to the FDA, in the form of an NDA or BLA, requesting approval to market the product for one or more indications. In most cases, the NDA/BLA must be accompanied by a substantial user fee. The FDA reviews an NDA/BLA to determine, among other things, whether a product is safe and effective for its intended use.
Before approving an application, the FDA will inspect the facility or facilities where the product is manufactured. The FDA will not approve the application unless cGMP compliance is satisfactory. The FDA will issue an approval letter if it determines that the application, manufacturing process and manufacturing facilities are acceptable. If the FDA determines that the application, manufacturing process or manufacturing facilities are not acceptable, it will outline the deficiencies in the submission and will often request additional testing or information. Notwithstanding the submission of any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval and refuse to approve the application by issuing a “not approvable” letter.
The testing and approval process requires substantial time, effort and financial resources, which may take several years to complete. The FDA may not grant approval on a timely basis, or at all. We may encounter difficulties or unanticipated costs in our efforts to secure necessary governmental approvals, which could delay or preclude us from marketing our products. Furthermore, the FDA may prevent a drug developer from marketing a product under a label for its desired indications or place other conditions, including restrictive labeling, on distribution as a condition of any approvals, which may impair commercialization of the product. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further FDA review and approval.
If the FDA approves the new drug application or biologics application, the drug can be marketed to physicians to prescribe in the U.S. After approval, the drug developer must comply with a number of post-approval requirements, including delivering periodic reports to the FDA (i.e., annual reports), submitting descriptions of any adverse reactions reported, biological product deviation reporting, and complying with drug sampling and distribution requirements. The holder of an approved NDA/BLA is required to provide updated safety and efficacy information and to comply with requirements concerning advertising and promotional labeling. Also, quality control and manufacturing procedures must continue to conform to cGMP after approval. Drug manufacturers and their subcontractors are required to register their facilities and are subject to periodic unannounced inspections by the FDA to assess compliance with cGMP which imposes procedural and documentation requirements relating to manufacturing, quality assurance and quality control. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain compliance with cGMP and other aspects of regulatory compliance. The FDA may require post-market testing and surveillance to monitor the product’s safety or efficacy, including additional studies to evaluate long-term effects.
In addition to studies requested by the FDA after approval, a drug developer may conduct other trials and studies to explore use of the approved drug for treatment of new indications, which require submission of a supplemental or new NDA and FDA approval of the new labeling claims. The purpose of these trials and studies is to broaden the application and use of the drug and its acceptance in the medical community.
We use, and will continue to use, third-party manufacturers to produce our products in clinical and commercial quantities. Future FDA inspections may identify compliance issues at our facilities or at the facilities of our contract manufacturers that may disrupt production or distribution, or require substantial resources to correct. In addition, discovery of problems with a product or the failure to comply with requirements may result in restrictions on a product, manufacturer or holder of an approved NDA/BLA, including withdrawal or recall of the product from the market or other voluntary or FDA-initiated action that could delay further marketing. Newly discovered or developed safety or effectiveness data may require changes to a product’s approved labeling, including the addition of new warnings and contraindications. Also, new government requirements may be established that could delay or prevent regulatory approval of our products under development.
21
Table of Contents
The Controlled Substances Act imposes various registration, record-keeping and reporting requirements, procurement and manufacturing quotas, labeling and packaging requirements, security controls, prescription and order form requirements and restrictions on prescription refills on some kinds of pharmaceutical products. A principal factor in determining the particular requirements of this act, if any, applicable to a product is its actual or potential abuse profile. A pharmaceutical product may be listed as a Schedule I, II, III, IV or V substance, with Schedule I substances considered to present the highest risk of substance abuse and Schedule V substances the lowest. Testosterone, the active drug substance in Testim, has been scheduled under the Controlled Substances Act as a Schedule III substance. All regular Schedule III drug prescriptions must be signed by a physician and may not be refilled. Furthermore, the amount of Schedule III substances we can obtain for clinical trials and commercial distribution is limited by the DEA and our quota may not be sufficient to complete clinical trials or meet commercial demand, if any. In addition to federal scheduling, Testim is subject to state-controlled substance regulation and may be placed in more restrictive schedules than those determined by the U.S. Drug Enforcement Agency and the FDA. However, to date, testosterone has not been placed in a more restrictive schedule by any state.
Foreign Regulation
Whether or not we obtain FDA approval for a product, we must obtain approval of a clinical trial or product by the comparable regulatory authorities of foreign countries before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time may be longer or shorter than that required for FDA approval. The requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement also vary greatly from country to country. Although governed by the applicable country’s regulations, clinical trials conducted outside of the U.S. typically are administered with the three-phase sequential process that is discussed above under “U.S. Government Regulation.” However, the foreign equivalent of an IND is not a prerequisite to performing pilot studies or Phase I clinical trials.
Under E.U. regulatory systems, we may submit marketing authorization applications either under centralized or decentralized procedures. The centralized procedure which is available for medicines produced by biotechnology or which are highly innovative, provides for the grant of a single marketing authorization that is valid for all E.U. member states. This authorization is a marketing authorization approval. Two decentralized procedures exist as alternatives for approval of products that are not obligated to be submitted by the centralized procedure. The first is the Mutual Recognition procedure, or MRP, which provides for mutual recognition of national approval decisions. Under this procedure, the holder of a national marketing authorization for a product may submit an application to the remaining member states. Within 90 days of receiving the applications and assessment report, each member state must decide whether to recognize approval. The second procedure, the Decentralized Procedure, was newly established in 2005. This procedure is available to products that have not been approved anywhere in the EU. The application is submitted simultaneously in the reference member state chosen by the applicant and in as many other member states as required. The reference member state carries out the main assessment and consults with the other member states during the process. Like the MRP, approval via this process results in national marketing approvals in the territories where scientific approval was received.
We received approval for Testim in the U.K. in June 2003. A mutual recognition application based on the U.K. approval was initiated on March 30, 2004. The procedure was completed on June 28, 2004 and resulted in scientific approval in the following 14 additional countries: Belgium, Denmark, Finland, Germany, Greece, Iceland, Ireland, Italy, Luxembourg, the Netherlands, Norway, Portugal, Spain and Sweden. Marketing approvals have been issued in each of these countries. Testim was launched in Belgium, Denmark, Finland, Germany, Ireland, Netherlands, Spain, Sweden, and the UK in 2005. In addition, in March 2004, we filed an application for Testim market authorization in Canada. We have responded to questions raised by the Canadian authorities as part of the approval process.
Furthermore, regulatory approval of prices is required in most countries other than the U.S. We face the risk that the resulting prices would be insufficient to generate an acceptable return to us or our collaborators.
22
Table of Contents
Fraud and Abuse Laws
We are subject to various federal and state laws pertaining to health care fraud and abuse, including anti-kickback laws, false claims laws and physician self-referral laws. Violations of these laws are punishable by criminal, civil and/or administrative sanctions, including, in some instances, imprisonment and exclusion from participation in federal and state health care programs, including Medicare, Medicaid and veterans’ health programs. Because of the far-reaching nature of these laws, there can be no assurance that the occurrence of one or more violations of these laws would not result in a material adverse effect on our business, financial condition and results of operations.
Anti-Kickback Laws. Our operations are subject to federal and state anti-kickback laws. Certain provisions of the Social Security Act prohibit entities such as us from knowingly and willingly offering, paying, soliciting or receiving any form of remuneration (including any kickbacks, bribe or rebate) in return for the referral of items or services for which payment may be made under a federal health care program, or in return for the recommendation, arrangement, purchase, lease or order of items or services for which payment may be made under a federal health care program. Violation of the federal anti-kickback law is a felony, punishable by criminal fines and imprisonment for up to five years or both. In addition, the Department of Health and Human Services may impose civil penalties and exclude violators from participation in federal health care programs such as Medicare and Medicaid. Many states have adopted similar prohibitions against payments intended to induce referrals of products or services paid by Medicaid or other third party payors.
Physician Self-Referral Laws. We also may be subject to federal and/or state physician self-referral laws. Federal physician self-referral legislation (known as the Stark law) prohibits, subject to certain exceptions, a physician from referring Medicare or Medicaid patients to an entity to provide designated health services, including, among other things, certain radiology and radiation therapy services and clinical laboratory services in which the physician or a member of his immediate family has an ownership or investment interest or has entered into a compensation arrangement. The Stark law also prohibits the entity receiving the improper referral from billing any good or service furnished pursuant to the referral. The penalties for violations include a prohibition on payment by these government programs and civil penalties of as much as $15,000 for each improper referral and $100,000 for participation in a circumvention scheme. Various state laws also contain similar provisions and penalties.
False Claims. The federal False Claims Act imposes civil and criminal liability on individuals or entities who submit (or cause the submission of) false or fraudulent claims for payment to the government. Violations of the federal False Claims Act may result in penalties equal to three times the damages which the government sustained, an assessment of between $5,000 and $10,000 per claim, civil monetary penalties and exclusion from participation in the Medicare and Medicaid programs.
The federal False Claims Act also allows a private individual to bring a qui tam suit on behalf of the government against an individual or entity for violations of the False Claims Act. In a qui tam suit, the private plaintiff is responsible for initiating a lawsuit that may eventually lead to the government recovering money of which it was defrauded. After the private plaintiff has initiated the lawsuit, the government must decide whether to intervene in the lawsuit and become the primary prosecutor. In the event the government declines to join the lawsuit, the private plaintiff may choose to pursue the case alone, in which case the private plaintiff’s counsel will have primary control over the prosecution (although the government must be kept apprised of the progress of the lawsuit). In return for bringing the suit on the government’s behalf, the statute provides that the private plaintiff is entitled to receive up to 30% of the recovered amount from the litigation proceeds if the litigation is successful plus reasonable expenses and attorneys fees. Recently, the number of qui tam suits brought against entities in the health care industry has increased dramatically. In addition, a number of states have enacted laws modeled after the False Claims Act that allow those states to recover money which was fraudulently obtained from the state.
Other Fraud and Abuse Laws. The Health Insurance Portability and Accountability Act of 1996 created, in part, two new federal crimes: Health Care Fraud and False Statements Relating to Health Care
23
Table of Contents
Matters. The Health Care Fraud statute prohibits the knowing and willful execution of a scheme or artifice to defraud any health care benefit program. A violation of the statute is a felony and may result in fines and/or imprisonment. The False Statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material fact by any trick, scheme or device or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services. A violation of this statute is a felony and may result in fines and/or imprisonment.
There are also several new state laws that require the reporting and disclosure of gifts or other value given to health care providers.
Compliance Program
The healthcare laws and fraud and abuse laws applicable to our business are complex and subject to variable interpretations. We maintain certain compliance, review, education and training and other programs to further our commitment to high standards of ethical and legal conduct and to minimize the likelihood that we would engage in conduct or enter into arrangements in violation of applicable authorities. We implemented a compliance program in June 2004 to help ensure that we remain in compliance with these laws. We have (i) established a compliance team consisting of representatives from our Legal, Human Resources and Regulatory Affairs/Quality Assurance departments that meets regularly; (ii) established a compliance hotline that permits our employees to report anonymously any compliance issues that may arise; and (iii) instituted other safeguards intended to help prevent any violations of the applicable fraud and abuse laws and healthcare laws, and to remediate any situations that could give rise to violations. We also review our transactions and agreements, both past and present, to help assure they are compliant.
Through our compliance efforts, we constantly strive to structure our business operations and relationships with our customers to comply with all applicable legal requirements. However, it is possible that governmental entities or other third parties could interpret these laws differently and assert non-compliance with respect to our business operations and relationships including these isolated arrangements. While there have been no claims asserted against us, if a claim were asserted and we were not to prevail, possible penalties and sanctions could have a material effect on our financial statements or our ability to conduct our operations.
Third-party Reimbursement and Pricing Controls
In the U.S. and elsewhere, sales of pharmaceutical products depend in significant part on the availability of reimbursement to the consumer from third-party payors, such as government and private insurance plans. To date, we have not experienced any problems with third-party payors; however, third-party payors increasingly are challenging the prices charged for medical products and services. It is, and will be, time consuming and expensive for us to go through the process of seeking reimbursement from Medicaid, Medicare and private payors. Our products may not be considered cost effective, and coverage and reimbursement may not be available or sufficient to allow us to sell our products on a competitive and profitable basis. The passage of the Medicare Prescription Drug and Modernization Act of 2003 imposes new requirements for the distribution and pricing of prescription drugs through Medicare which may affect the marketing of our products. We have elected to participate in the newly enacted Medicare Part D program. Although the impact this program will have on Testim has not been fully determined, we feel given the age of the population that suffers from hypogonadism, more men will have access to this therapy than previously. We have provided aggressive rebates to prescription drug plans which has put us in advantaged position for a majority of covered lives within the Medicare Part D population.
In many foreign markets, including the countries in the E.U., pricing of pharmaceutical products is subject to governmental control. In the U.S., there have been, and we expect that there will continue to be, a number of federal and state proposals to implement similar governmental pricing control. While we cannot predict whether such legislative or regulatory proposals will be adopted, the adoption of such proposals could have a material adverse effect on our business, financial condition and profitability.
24
Table of Contents
Patents and Proprietary Rights
Our success will depend in part on our ability to protect our existing products and the products we acquire or in-license by obtaining and maintaining a strong proprietary position both in the U.S. and in other countries. To develop and maintain such a position, we intend to continue relying upon patent protection, trade secrets, know-how, continuing technological innovations and licensing opportunities. In addition, we intend to seek patent protection whenever available for any products or product candidates and related technology we develop or acquire in the future.
We do not own any patents. We rely on patent protection of our licensors and on our trade secrets to maintain our competitive protection.
Bentley.We have licensed the underlying technology for Testim, our only approved product, from Bentley. The patent that we license from Bentley for Testim expires in June 2008 in the U.S., November 2006 in Europe and Japan and January 2010 in Canada. This patent covers methods and compositions for administering drugs, such as testosterone, across the skin and mucus membranes. Bentley has submitted a patent application for an additional formulation patent for Testim that, if issued, would provide us with further market protection around the world until approximately 2020. In July 2005, an additional patent covering Testim issued in Canada, and this patent is included in our license from Bentley. This patent expires in 2023. Testosterone, the active ingredient in Testim, does not have patent protection and is included in competing TRT products.
BioSpecifics.We licensed AA4500, our product candidate for the treatment of Dupuytren’s and Peyronie’s Diseases as well as Frozen Shoulder Syndrome, from BioSpecifics. In addition to the marketing exclusivity which comes with its orphan drug status as a treatment for Dupuytren’s and Peyronie’s Diseases, the enzyme underlying this product candidate is covered by two use patents in the U.S., one for the treatment of Dupuytren’s Disease and one for the treatment of Peyronie’s Disease. The Dupuytren’s patent expires in 2014, and the Peyronie’s patent expires in 2019. The patent relating to Dupuytren’s Disease is the subject of a reissue application in the U. S. Patent and Trademark Office, or USPTO, for, among other things, the purpose of submitting prior art that was not previously submitted during the prosecution of the Dupuytren’s patent. As a result, the patent may or may not be allowed by the USPTO, and therefore, may or may not be valid and enforceable. Both patents are limited to the use of the enzyme for the treatment of Dupuytren’s and Peyronie’s Diseases within certain dose ranges. While we believe that dose optimization trials will demonstrate that effective dosing will fall within the patented range, currently, there is not enough data to establish whether the ultimate approved product will be covered by the patents. Foreign patents also cover these products in certain countries. In January 2005, a patent application was filed with regard to AA4500 for the treatment of Frozen Shoulder Syndrome. If this patent is granted, it would expire in 2026. In January 2006, we filed a patent application with regard to the manufacturing process for AA4500; if this patent is granted, it would expire in January 2027.
PharmaForm.We have licensed the transmucosal film technology for our testosterone replacement, overactive bladder and pain product candidates from PharmaForm. The U.S. patent that we license from PharmaForm expires in 2020.
The scope of the intellectual property rights held by pharmaceutical firms involves complex legal, scientific and factual questions and consequently is generally uncertain. In addition, the coverage claimed in a patent application can be significantly reduced before the patent is issued. Consequently, we do not know whether any of our current patent applications, or the products or product candidates we develop, acquire or license will result in the issuance of patents or, if any patents are issued, whether they will provide significant proprietary protection or will be challenged, circumvented or invalidated. Because patent applications in the U.S. and some other jurisdictions are sometimes maintained in secrecy until patents issue, and since publication of discoveries in the scientific or patent literature often lags behind actual discoveries, we cannot be certain of the priority of inventions covered by pending patent applications. Moreover, we may have to participate in interference proceedings declared by the U.S. Patent and Trademark Office or a foreign patent office to determine priority of
25
Table of Contents
invention, or in opposition proceedings in a foreign patent office, either of which could result in substantial cost to us, even if the eventual outcome is favorable to us. There can be no assurance that the patents, if issued and challenged, in a court of competent jurisdiction would be found valid or enforceable. An adverse outcome could subject us to significant liabilities to third parties, require disputed rights to be licensed from third parties or require us to cease using such technology.
Although we believe these patent applications, if they issue as patents, will provide a competitive advantage, the patent positions of pharmaceutical and biotechnology companies are highly uncertain and involve complex legal and factual questions. We may not be able to develop patentable products or processes and may not be able to obtain patents from pending applications. Even if patent claims are allowed, the claims may not issue, or in the event of issuance, may not be sufficient to protect our technology. In addition, any patents or patent rights we obtain may be circumvented, challenged or invalidated by our competitors.
While we attempt to ensure that our product candidates and the methods we employ to manufacture them do not infringe other parties’ patents and proprietary rights, competitors or other parties may assert that we infringe on their proprietary rights. Additionally, because patent prosecution can proceed in secret prior to issuance of a patent, third parties may obtain other patents without our knowledge prior to the issuance of patents relating to our product candidates which they could attempt to assert against us.
Although we believe that our product candidates, production methods and other activities do not currently infringe the intellectual property rights of third parties, we cannot be certain that a third party will not challenge our position in the future. If a third party alleges that we are infringing its intellectual property rights, we may need to obtain a license from that third party, but there can be no assurance that any such license will be available on acceptable terms or at all. Any infringement claim that results in litigation could result in substantial cost to us and the diversion of management’s attention away from our core business and could also prevent us from marketing our products. To enforce patents issued to us or to determine the scope and validity of other parties’ proprietary rights, we may also become involved in litigation or in interference proceedings declared by the U.S. Patent and Trademark Office, which could result in substantial costs to us or an adverse decision as to the priority of our inventions. We may be involved in interference and/or opposition proceedings in the future. We believe there will continue to be litigation in our industry regarding patent and other intellectual property rights.
We also rely on trade secret protection for our confidential and proprietary information. No assurance can be given that others will not independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose such technology or that we can meaningfully protect our trade secrets.
It is our policy to require our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us. These agreements provide that all confidential information developed or made known to the individual during the course of the individual’s relationship with us is to be kept confidential and not disclosed to third parties except in specific circumstances. In the case of employees, the agreements provide that all inventions conceived by the individual shall be our exclusive property. There can be no assurance, however, that these agreements will provide meaningful protection or adequate remedies for our trade secrets in the event of unauthorized use or disclosure of such information.
Orphan Drug Status
The Orphan Drug provisions of the Federal Food, Drug, and Cosmetic Act provide incentives to drug and biologics suppliers to develop and supply drugs for the treatment of rare diseases, currently defined as diseases that affect fewer than 200,000 individuals in the U.S. or, for a disease that affects more than 200,000 individuals in the U.S. and for which there is no reasonable expectation that the cost of developing and making available in the U.S. a drug for such disease or condition will be recovered from its sales in the U.S. Under these provisions, a
26
Table of Contents
supplier of a designated orphan product can seek tax benefits, and the holder of the first FDA approval of a designated orphan product will be granted a seven-year period of marketing exclusivity for that product for the orphan indication. The marketing exclusivity of an orphan drug would prevent other sponsors from obtaining approval of the same drug for the same indication without demonstrating that it is clinically superior. It would not prevent other drugs from being approved for the same indication.
In the E.U., incentives for suppliers to develop medicinal products for the treatment of rare diseases are provided pursuant to the Orphan Medicinal Products Regulation. Orphan medicinal products are those products designed to diagnose, treat or prevent a condition which occurs so infrequently that the cost of developing and bringing the product to the market would not be recovered by the expected sale of the product. In the E.U., the criterion for designation is a prevalence of the relevant condition in no more than 5 per 10,000 of the population. The incentives include, among others, a reduction in the fees payable in respect of the marketing authorization application, protocol assistance for clinical trials in support of the application, and marketing exclusivity once the authorization is granted. In the E.U., marketing exclusivity is granted to products with an orphan drug designation for a period of 10 years during which the E.U. will not accept another application for a marketing authorization for the same therapeutic indication in respect of a similar medicinal product, unless the second applicant can show its product is safer, more effective or otherwise clinically superior. A similar medicinal product is defined as a medicinal product containing a similar active substance as contained in the authorized orphan medicinal product.
The FDA granted orphan drug status to AA4500 in the U.S. for each of the treatments of Dupuytren’s Disease and Peyronie’s Disease. The designations for the treatment of Dupuytren’s Disease and Peyronie’s Disease have been transferred to us. Orphan drug status means that, if AA4500 is the first product to receive FDA approval for the orphan indications, another application to market the same drug for the same indication may not be approved, except in limited circumstances, for a period of up to seven years in the U.S.
The Hatch-Waxman Act
Under the U.S. Drug Price Competition and Patent Term Restoration Act of 1984, known as the Hatch-Waxman Act, newly approved drugs and indications benefit from a statutory period of non-patent marketing exclusivity. The Hatch-Waxman Act provides five-year marketing exclusivity to the first applicant to gain approval of an NDA for a new chemical entity, meaning that the FDA has not previously approved any other new drug containing the same active ingredient. The Hatch-Waxman Act prohibits an abbreviated new drug application, or ANDA, where the applicant does not own or have a legal right of reference to all the data required for approval, to be submitted by another company for another version of such drug during the five-year exclusive period. Protection under the Hatch-Waxman Act will not prevent the filing or approval of another full NDA, however, the applicant would be required to conduct its own pre-clinical, adequate and well-controlled clinical trials to demonstrate safety and effectiveness. The Hatch-Waxman Act also provides three years of marketing exclusivity for the approval of new NDAs with new clinical trials for previously approved drugs and supplemental NDAs, for example, for new indications, dosages, or strengths of an existing drug, if new clinical investigations are essential to the approval. This three-year exclusivity covers only the new changes associated with the supplemental NDA and does not prohibit the FDA from approving ANDAs for drugs containing the original active ingredient or indications.
The Hatch-Waxman Act also permits a patent extension term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent extension cannot extend the remaining term of a patent beyond a total of 14 years. The patent term restoration period is generally one-half the time between the effective date of an IND and the submission date of an NDA, plus the time between the submission date of an NDA and the approval of that application. Only one patent applicable to an approved drug is eligible for the extension and it must be applied for prior to expiration of the patent. The U.S. Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for patent term extension.
27
Table of Contents
Employees
As of December 31, 2005, we had 174 full-time employees, of which 29 were in research and development, 121 were in sales and marketing and 24 were in general and administration, and five part-time employees. We believe that our relations with our employees are good and we have no history of work stoppages. Generally, our employees are at-will employees. However, we have entered into employment agreements with certain of our executive officers.
| ITEM 1A. | Risk Factors |
In addition to the other information included in this Report, the following factors should be considered in evaluating our business and future prospects. Any of the following risks, either alone or taken together, could materially and adversely affect our business, financial position or results of operations. If one or more of these or other risks or uncertainties materialize, or if our underlying assumptions prove to be incorrect, our actual results may vary materially from what we projected. There may be additional risks that we do not presently know or that we currently believe are immaterial which could also impair our business or financial position.
Risks Related to Our Financial Results and Need for Additional Financing
We have incurred significant losses since our inception and may not achieve profitability in the foreseeable future.
We have incurred significant losses since our inception, including net losses of $38.3 million for the twelve months ended December 31, 2005. As of December 31, 2005, we had an accumulated deficit of $137.5 million. Our only approved product is Testim. We expect to continue to make substantial expenditures related to the marketing and selling of Testim. As we launched Testim in the first quarter of 2003, we cannot be certain how effective our marketing and selling programs will be in the future. We continue to expand and enhance the marketing and selling of Testim. In April 2005, we entered into an agreement with Oscient for the co-promotion of Testim to primary care physicians in the U.S. As a result, marketing and selling expenses are likely to increase substantially in the future. In addition, we expect to continue to incur substantial expenses, including milestone payments, as we:
| • | develop AA4500, our product candidate for the treatment of Dupuytren’s Disease, Peyronie’s Disease and Adhesive Capsulitis, or Frozen Shoulder Syndrome; AA2600, our testosterone replacement transmucosal film product candidate; AA4010, our overactive bladder transmucosal film product candidate; and our pain transmucosal film product candidates and any other product candidates that we license or acquire; |
| • | develop our manufacturing processes at scale; |
| • | seek regulatory approval for our product candidates; |
| • | commercialize any of our product candidates that receive marketing approval or any approved products that we acquire or in-license; and |
| • | acquire or in-license new technologies or development stage or approved products. |
Accordingly, we expect to continue to incur substantial additional losses for the foreseeable future. In order to achieve and maintain profitability, we will need to generate significantly greater revenues from Testim and any other product candidate for which we receive marketing approval. As a result, we are unsure when we will become profitable, if at all. If we fail to achieve profitability within the time frame expected by investors, the value of our common stock may decline substantially.
28
Table of Contents
Because we have a limited operating history, our future results are unpredictable, and therefore, our common stock is a highly speculative investment.
We commenced operations in the fourth quarter of 1999 and have a very limited operating history for you to evaluate our business. Accordingly, you must consider our prospects in light of the risks and difficulties encountered by companies in their early stages. In order to address these risks, we must:
| • | increase the sales of Testim, our only product with marketing approval; |
| • | successfully develop, obtain marketing approval for and have manufactured our current product candidates, including AA4500, for the treatment of Dupuytren’s Disease, Peyronie’s Disease, and Frozen Shoulder, AA2600 for the treatment of hypogonandism, AA4010 for the treatment of overactive bladder and our pain transmucosal film product candidates; |
| • | successfully identify and develop new product candidates; |
| • | effectively commercialize any approved product candidates that we develop or any approved products that we acquire; |
| • | respond to competitive pressures from other businesses, including the launch of any products, including generics, that compete with Testim in the treatment of hypogonadism; |
| • | identify and negotiate favorable agreements with third parties for the manufacture, distribution and marketing and sales of our approved products; and |
| • | effectively manage our relationships with vendors and third parties. |
In addition, due to our limited operating history, we have minimal experience with product returns. If returns are higher than expected, our operating results could be materially harmed.
All of our revenues to date have been generated from the sale or out-licensing of Testim, and, if these revenues do not grow, and we cannot commercialize new products, we will not become profitable.
Our only product with marketing approval is Testim and our product revenues to date have been generated solely from the sale of Testim. Until such time as we develop, acquire or in-license additional products that are approved for marketing, we will continue to rely on Testim for all of our revenues. Accordingly, our success depends significantly on our ability to increase sales of Testim. Our sales and marketing efforts or those of our co-promotion partner may not be successful in increasing prescriptions for Testim. Sales of Testim are subject to the following risks, among others:
| • | growth of the overall androgen market which may be influenced by sales and marketing efforts of Solvay; |
| • | acceptance by the medical community or the general public of Testim as a safe or effective therapy for hypogonadism; |
| • | increasing awareness and continued acceptance of hypogonadism by the medical community, regulators or the general public as a medical disorder requiring treatment; |
| • | pressures from existing or new competing products, including generic products, that may provide therapeutic, convenience or pricing advantages over Testim; |
| • | failure of third-party payors to provide coverage and sufficient reimbursement for Testim; and |
| • | impact of coverage and reimbursement changes, included Medicare Part D coverage and reimbursement. |
For the foreseeable future, if we are unable to grow Testim sales out-licensing, we will be unable to increase our revenues or achieve profitability and we may be forced to delay or change our current plans to develop other product candidates.
29
Table of Contents
If we are unable to meet our needs for additional funding in the future, we may be required to limit, scale back or cease our operations.
Our operations to date have generated substantial and increasing needs for cash. Our negative cash flows from operations are expected to continue for at least the foreseeable future. Our strategy contains elements that we will not be able to execute without outside funding. Specifically, we may need to raise additional capital to:
| • | enhance or expand our sales and marketing efforts for Testim or other products we may co-promote, acquire or in-license; |
| • | fund our product development, including clinical trials; |
| • | commercialize any product candidates that receive regulatory approval; and |
| • | acquire or in-license approved products or product candidates or technologies for development. |
We believe that our existing cash resources and interest on these funds will be sufficient to meet our anticipated operating requirements until at least the end of 2007. Our future funding requirements will depend on many factors, including:
| • | Testim market acceptance and sales growth; |
| • | growth of the overall androgen market which may be influenced by Solvay’s sales and marketing efforts; |
| • | third-party payor reimbursement for Testim; |
| • | the cost of manufacturing, distributing, marketing and selling Testim; |
| • | the scope, rate of progress and cost of our product development activities; |
| • | future clinical trial results; |
| • | the degree to which our current co-promotion and licensing and distribution agreements generate revenue; |
| • | the terms and timing of any future collaborative, licensing, co-promotion and other arrangements that we may establish; |
| • | the cost and timing of regulatory approvals; |
| • | the costs of commercializing any of our other product candidates; |
| • | acquisition or in-licensing costs; |
| • | the effect of competing technological and market developments; |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; and |
| • | the extent to which we acquire or invest in businesses, products and technologies, although we currently have no commitments or agreements relating to any of these types of transactions. |
These factors could result in variations from our currently projected operating and liquidity requirements. If our existing resources are insufficient to satisfy our liquidity requirements, we may need to borrow money or sell additional equity or debt securities. We may not be able to borrow money on commercially reasonable terms. Moreover, the terms of the sale of any equity or debt securities may not be acceptable to us and could result in substantial dilution of your investment. If we are unable to obtain this additional financing, we may be required to:
| • | reduce the size or scope, or both, of our sales and marketing efforts for Testim or any of our future products; |
30
Table of Contents
| • | delay or reduce the scope of, or eliminate one or more of our planned development, commercialization or expansion activities; |
| • | seek collaborators for our product candidates at an earlier stage than otherwise would be desirable and on terms that are less favorable than might otherwise be available; or |
| • | relinquish, license or otherwise dispose of rights to technologies, product candidates or products that we would otherwise seek to develop or commercialize ourselves on terms that are less favorable than might otherwise be available. |
Our revenues, operating results and cash flows may fluctuate in future periods and we may fail to meet investor expectations, which may cause the price of our common stock to decline.
Variations in our quarterly operating results are difficult to predict and may fluctuate significantly from period to period. We are a relatively new company and our sales prospects are uncertain. We have been selling Testim, our only approved product, since the first quarter of 2003 and cannot predict with certainty the timing or level of sales of Testim in the future. If our quarterly sales or operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline substantially. In addition to the other factors discussed under these “Risk Factors,” specific factors that may cause fluctuations in our operating results include:
| • | demand and pricing for Testim, including any change in wholesaler purchasing patterns or cost structures for our products or their services; |
| • | growth of the overall androgen market which may be influenced by Solvay’s sales and marketing efforts; |
| • | prescription levels relating to physician and patient acceptance of, and prescription costs for, Testim or any of our future products; |
| • | government or private healthcare reimbursement policies; |
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | introduction of competing products, including generics; |
| • | any interruption in the manufacturing or distribution of Testim or any of our future products; |
| • | the degree to which our expenses generated by our co-promotion arrangements exceed revenue we derive from such arrangements; |
| • | our operating expenses, many of which are relatively fixed; |
| • | timing and costs associated with any new product or technology acquisitions we may complete; |
| • | variations in our rates of product returns, allowances and rebates and discounts; and |
| • | milestone payments. |
Forecasting our revenues is further complicated by difficulties in estimating inventory levels at our wholesaler and chain drug store customers, prescription levels, the timing of purchases by wholesalers and retailers to replenish inventory and the occurrence and amount of product returns.
As a result of these factors, we believe that period-to-period comparisons of our operating results are not a good indication of our future performance.
31
Table of Contents
Risks Related to Development of Our Product Candidates
We may not be able to develop product candidates into viable commercial products, which would impair our ability to grow and could cause a decline in the price of our stock.
The process of developing product candidates involves a high degree of risk and may take several years. Product candidates that appear promising in the early phases of development may fail to reach the market for several reasons, including:
| • | clinical trials may show our product candidates to be ineffective or not as effective as anticipated or to have harmful side effects or any unforeseen result; |
| • | product candidates may fail to receive regulatory approvals required to bring the products to market; |
| • | manufacturing costs, the inability to scale up to produce supplies for clinical trials or other factors may make our product candidates uneconomical; and |
| • | the proprietary rights of others and their competing products and technologies may prevent our product candidates from being effectively commercialized or to obtain exclusivity. |
Success in preclinical and early clinical trials does not ensure that large-scale clinical trials will be successful. Clinical results are frequently susceptible to varying interpretations that may delay, limit or prevent regulatory approvals. The length of time necessary to complete clinical trials and to submit an application for marketing approval for a final decision by a regulatory authority varies significantly and may be difficult to predict. Currently, there is substantial congressional and administration review of the regulatory approval process for drug candidates in the U.S. Any changes to the U.S. regulatory approval process could significantly increase the timing or cost of regulatory approval for our product candidates making further development uneconomical or impossible.
In addition, developing product candidates is very expensive and will have a significant impact on our ability to generate profits. Factors affecting our product development expenses include:
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | the cost and timing of manufacturing clinical supplies of product candidates; |
| • | our ability to raise any additional funds that we need to complete our trials; |
| • | the number and outcome of clinical trials conducted by us and/or our collaborators; |
| • | the number of products we may have in clinical development; |
| • | in-licensing or other partnership activities, including the timing and amount of related development funding, license fees or milestone payments; and |
| • | future levels of our revenue. |
Our product development efforts also could result in large and immediate write-offs, significant milestone payments, incurrence of debt and contingent liabilities or amortization of expenses related to intangible assets, any of which could negatively impact our financial results. Additionally, if we are unable to develop our product candidates into viable commercial products, we may remain reliant solely on Testim sales for our revenues, potentially limiting our growth opportunities.
32
Table of Contents
If clinical trials for our product candidates are delayed, we would be unable to commercialize our product candidates on a timely basis, which would materially harm our business.
Clinical trials that we may conduct may not begin on time or may need to be restructured after they have begun. Clinical trials can be delayed or may need to be restructured for a variety of reasons, including delays or restructuring related to:
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | obtaining an investigational new drug exemption, or IND, or other regulatory approval to commence a clinical trial; |
| • | negotiating acceptable clinical trial agreement terms with prospective investigators or trial sites; |
| • | obtaining institutional review board, or equivalent, approval to conduct a clinical trial at a prospective site; |
| • | recruiting subjects to participate in a clinical trial; |
| • | competition in recruiting clinical investigators; |
| • | shortage or lack of availability of clinical trial supplies; |
| • | the need to repeat clinical trials as a result of inconclusive results or poorly executed testing; |
| • | the placement of a clinical hold on a study; |
| • | the failure of third parties conducting and overseeing the operations of our clinical trials to perform their contractual or regulatory obligations in a timely fashion; and |
| • | exposure of clinical trial subjects to unexpected and unacceptable health risks or noncompliance with regulatory requirements, which may result in suspension of the trial. |
Prior to commencing clinical trials in the U.S., we must have an effective IND for each of our product candidates. An IND has been filed and is effective for AA4500 for the treatment of Dupuytren’s Disease, AA4500 for the treatment of Peyronie’s Disease, AA4500 for the treatment of Frozen Shoulder Syndrome and AA2600; however, INDs have not been filed for AA4010 or our pain transmucosal film product candidates. We will not be able to commence clinical trials in the U.S. for these product candidates until we file, and the FDA fails to object to, an IND for each candidate.
We have five projects in clinical development, specifically AA4500 for the treatment of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome, AA2600 and AA4010. Completion of clinical trials for each product candidate will be required before commercialization. If we experience delays in or termination of clinical trials or if the cost or timing of the regulatory approval process increases, our financial results and the commercial prospects for our product candidates will be adversely impacted. In addition, our product development costs would increase and our ability to generate additional revenue from new products could be impaired.
Adverse events or lack of efficacy in our clinical trials may force us to stop development of our product candidates or prevent regulatory approval of our product candidates, which could materially harm our business.
Patients participating in the clinical trials of our product candidates may experience serious adverse health events. A serious adverse health event includes death, a life-threatening condition, hospitalization, disability, congenital anomaly, or a condition requiring intervention to prevent permanent impairment or damage. The occurrence of any of these events could interrupt, delay or halt clinical trials of our product candidates and could result in the FDA, or other regulatory authorities, denying approval of our product candidates for any or all targeted indications. An institutional review board or independent data safety monitoring board, the FDA, other
33
Table of Contents
regulatory authorities or we may suspend or terminate clinical trials at any time. Our product candidates may prove not to be safe for human use. Any delay in the regulatory approval of our product candidates could increase our product development costs and allow our competitors additional time to develop or market competing products. If our product candidates do not receive the necessary regulatory approval, we may remain reliant on sales of Testim as our sole source of revenue.
Our failure to successfully in-license or acquire additional technologies, product candidates or approved products would impair our ability to grow.
We intend to in-license, acquire, develop and market additional products and product candidates so that we are not solely reliant on Testim sales for our revenues. Because we have limited internal research capabilities, we are dependent upon pharmaceutical and biotechnology companies and other researchers to sell or license products or technologies to us. The success of this strategy depends upon our ability to identify, select and acquire the right pharmaceutical product candidates, products and technologies. To date, we have in-licensed the formulation technology underlying Testim from Bentley, the transmucosal film technology underlying AA2600, AA4010 and pain transmucosal film product candidates from PharmaForm, and the enzyme underlying AA4500 from BioSpecifics.
We may not be able to successfully identify any other commercial products or product candidates to in-license, acquire or internally develop. Moreover, negotiating and implementing an economically viable in-licensing arrangement or acquisition is a lengthy and complex process. Other companies, including those with substantially greater financial, marketing and sales resources, may compete with us for the in-licensing or acquisition of product candidates and approved products. We may not be able to acquire or in-license the rights to additional product candidates and approved products on terms that we find acceptable, or at all. If we are unable to in-license or acquire additional commercial products or product candidates we may be reliant solely on Testim sales for revenues. As a result, our ability to grow our business or increase our profits could be severely limited.
If we engage in any acquisition, we will incur a variety of costs, and we may never realize the anticipated benefits of the acquisition.
Since our inception, we have acquired the underlying technology for Testim and all of our product candidates through in-licensing arrangements. The underlying technology for Testim was licensed from Bentley. The transmucosal film technology underlying AA2600, AA4010 and the pain product candidates was licensed from PharmaForm. AA4500, our product candidate for Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome, was licensed from BioSpecifics. One of our strategies for business expansion is the acquisition of additional products and product candidates. We may attempt to acquire these product candidates, or other potentially beneficial technologies, through in-licensing or the acquisition of businesses, services or products that we believe are a strategic fit with our business. If we undertake an acquisition, the process of integrating any newly acquired business, technology, service or product into our existing operations could be expensive and time consuming and may result in unforeseen operating difficulties and expenditures and may divert significant management attention from our ongoing business operations. Moreover, we may fail to realize the anticipated benefits of any acquisition for a variety of reasons, such as an acquired product candidate proving to not be safe or effective in later clinical trials or not reaching its forecasted commercial potential. We may fund any future acquisition by issuing equity or debt securities, which could dilute your ownership percentage or limit our financial or operating flexibility as a result of restrictive covenants related to new debt. Acquisition efforts can consume significant management attention and require substantial expenditures, which could detract from our other programs. In addition, we may devote resources to potential acquisitions that are never completed. If we are unable to acquire and successfully integrate product candidates through in-licensing or the acquisition of businesses, services or products, we may remain reliant solely on Testim sales for revenue. In pursuing our acquisition strategy, we have expended significant management time, consulting costs and legal expenses without consummating a transaction.
34
Table of Contents
Risks Related to Regulatory Approval of Our Product Candidates
We are subject to numerous complex regulatory requirements and failure to comply with these regulations, or the cost of compliance with these regulations, may harm our business.
The testing, development and manufacturing and distribution of our products are subject to regulation by numerous governmental authorities in the U.S., Europe and elsewhere. These regulations govern or affect the testing, manufacture, safety, labeling, storage, record-keeping, approval, distribution, advertising and promotion of Testim and our product candidates, as well as safe working conditions and the experimental use of animals. Noncompliance with any applicable regulatory requirements can result in refusal of the government to approve products for commercialization, criminal prosecution and fines, recall or seizure of products, total or partial suspension of production, prohibitions or limitations on the commercial sale of products or refusal to allow the entering into of federal and state supply contracts. FDA and comparable governmental authorities have the authority to withdraw product approvals that have been previously granted. Currently, there is a substantial amount of congressional and administrative review of the FDA and the regulatory approval process for drug candidates in the U.S. As a result, there may be significant changes made to the regulatory approval process in the U.S. In addition, the regulatory requirements relating to the manufacturing, testing, and promotion, marketing and distribution of our products may change in the U.S. or the other jurisdictions in which we may have obtained or be seeing regulatory approval for our products or product candidates. Such changes may increase our costs and adversely effect our operations.
Testosterone is listed by the U.S. Drug Enforcement Agency, or DEA, as a Schedule III substance under the Controlled Substances Act of 1970. The DEA classifies substances as Schedule I, II, III, IV or V substances, with Schedule I substances considered to present the highest risk of substance abuse and Schedule V substances the lowest risk. Our transmucosal film product candidates may also involve the use of scheduled substances. Scheduled substances are subject to DEA regulations relating to manufacturing, storage, distribution and physician prescription procedures. For example, all regular Schedule III drug prescriptions must be signed by a physician and may not be refilled. Furthermore, the amount of Schedule III substances we can obtain for clinical trials and commercial distribution is limited by the DEA and our quota may not be sufficient to complete clinical trials or meet commercial demand, if any.
Entities must be registered annually with the DEA to manufacture, distribute, dispense, import, export, and conduct research using controlled substances. State controlled substance laws also require registration for similar activities. In addition, the DEA requires entities handling controlled substances to maintain records and file reports, follow specific labeling and packaging requirements, and provide appropriate security measures to control against diversion of controlled substances. Failure to follow these requirements can lead to significant civil and/or criminal penalties and possibly even lead to a revocation of a DEA registration.
Products containing controlled substances may generate public controversy. As a result, these products may have their marketing rights or regulatory approvals withdrawn. Political pressures and adverse publicity could lead to delays in, and increased expenses for, and limit or restrict the introduction and marketing of our product candidates. For some scheduled substances, the FDA may require us to develop a comprehensive risk management program to reduce the inappropriate use of our products and product candidates, including the manner in which they are marketed and sold, so as to reduce the risk of improper patient selection and diversion or abuse of the product. Developing such a program in consultation with the FDA may be a time-consuming process and could delay approval of any of our product candidates. Such a program or delays of any approval from the FDA could increase our product development costs and may allow our competitors additional time to develop or market competing products.
Additionally, failure to comply with or changes to the regulatory requirements that are applicable to Testim or our other product candidates may result in a variety of consequences, including the following:
| • | restrictions on our products or manufacturing processes; |
35
Table of Contents
| • | warning letters; |
| • | withdrawal of Testim or a product candidate from the market; |
| • | voluntary or mandatory recall of Testim or a product candidate; |
| • | fines against us; |
| • | suspension or withdrawal of regulatory approvals for Testim or a product candidate; |
| • | suspension or termination of any of our ongoing clinical trials of a product candidate; |
| • | refusal to permit to import or export of our products; |
| • | refusal to approve pending applications or supplements to approved applications that we submit; |
| • | denial of permission to file an application or supplement in a jurisdiction; |
| • | product seizure; and |
| • | injunctions or the imposition of civil or criminal penalties against us. |
If our product candidates are not demonstrated to be sufficiently safe and effective, they will not receive regulatory approval and we will be unable to commercialize them.
Other than Testim, all of our other product candidates are in preclinical or clinical development and have not received regulatory approval from the FDA or any foreign regulatory authority. An IND has been filed and is effective for AA4500 for the treatment of Dupuytren’s Disease, AA4500 for the treatment of Peyronie’s Disease, AA4500 for the treatment of Frozen Shoulder Syndrome and AA2600; however, INDs have not been filed for AA4010 or our pain transmucosal film product candidates. The regulatory approval process typically is extremely expensive, takes many years and the timing or likelihood of any approval cannot be accurately predicted.
As part of the regulatory approval process, we must conduct preclinical studies and clinical trials for each product candidate to demonstrate safety and efficacy. The number of preclinical studies and clinical trials that will be required varies depending on the product candidate, the indication being evaluated, the trial results and regulations applicable to any particular product candidate.
The results of preclinical studies and initial clinical trials of our product candidates do not necessarily predict the results of later-stage clinical trials. Product candidates in later stages of clinical trials may fail to show the desired safety and efficacy despite having progressed through initial clinical trials. We cannot assure you that the data collected from the preclinical studies and clinical trials of our product candidates will be sufficient to support FDA or other regulatory approval. In addition, the continuation of a particular study after review by an institutional review board or independent data safety monitoring board does not necessarily indicate that our product candidate will achieve the clinical endpoint.
The FDA and other regulatory agencies can delay, limit or deny approval for many reasons, including:
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | a product candidate may not be deemed to be safe or effective; |
| • | the manufacturing processes or facilities we have selected may not meet the applicable requirements; and |
| • | changes in their approval policies or adoption of new regulations may require additional clinical trials or other data. |
Any delay in, or failure to receive, approval for any of our product candidates or the failure to maintain regulatory approval for Testim could prevent us from growing our revenues or achieving profitability.
36
Table of Contents
Our corporate compliance program cannot guarantee that we are in compliance with all potentially applicable regulations.
We are a relatively small company and we rely heavily on third parties to conduct many important functions. As a pharmaceutical company, we are subject to a large body of legal and regulatory requirements. In addition, as a publicly traded company we are subject to significant regulations, including the Sarbanes-Oxley Act of 2002, or SOA, some of which have either only recently been adopted or are currently proposals subject to change. We cannot assure you that we are or will be in compliance with all potentially applicable laws and regulations. Failure to comply with all potentially applicable laws and regulations could lead to the imposition of fines, cause the value of our common stock to decline, impede our ability to raise capital or lead to the de-listing of our stock.
We will incur increased costs as a result of recently enacted and proposed changes in laws and regulations relating to corporate governance and other matters.
Our compliance with recently enacted and proposed changes in the laws and regulations affecting public companies, including the provisions of the SOA, and rules adopted or proposed by the Securities and Exchange Commission (SEC) and by the Nasdaq Stock Market, will result in increased costs to us as we evaluate the implications of any new rules and respond to their requirements. Although we are not required to issue an evaluation of our internal control over financial reporting under Section 404 of SOA until March 2007, at the earliest, preparations for the issuance of this report have already resulted in increased costs to us, which will increase further. If we are not able to issue an evaluation of our internal control over financial reporting as required or we or our independent registered public accounting firm determine that our internal control over financial reporting is not effective, this shortcoming could have an adverse effect on our business and financial results and the price of our common stock could be negatively affected. The new rules could make it more difficult or more costly for us to obtain certain types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these events could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees and as executive officers. We cannot predict or estimate the amount of the additional costs we may incur or the timing of such costs to comply with these rules and regulations.
Risks Related to Commercialization
If medical doctors do not prescribe our products or the medical profession does not accept our products, our ability to grow our revenues will be limited.
Our business is dependent on market acceptance of our products by physicians, healthcare payors, patients and the medical community. Medical doctors’ willingness to prescribe our products depends on many factors, including:
| • | perceived safety and efficacy of our products; |
| • | convenience and ease of administration; |
| • | prevalence and severity of adverse side effects in both clinical trials and commercial use; |
| • | availability of alternative treatments; |
| • | cost effectiveness; |
| • | effectiveness of our marketing strategy and the pricing of our products; |
| • | publicity concerning our products or competing products; and |
| • | our ability to obtain third-party coverage or reimbursement. |
37
Table of Contents
Even though we have received regulatory approval for Testim, and even if we receive regulatory approval and satisfy the above criteria for any of our other product candidates, physicians may not prescribe our products if we do not promote our products effectively. Factors that could affect our success in marketing our products include:
| • | the effectiveness of our sales force and that of any co-promotion partners; |
| • | the adequacy and effectiveness of our production, distribution and marketing capabilities; |
| • | the success of competing products, including generics; and |
| • | the availability and extent of reimbursement from third-party payors. |
If any of our products or product candidates fails to achieve market acceptance, we may not be able to market and sell the products successfully, which would limit our ability to generate revenue and could harm our business.
If testosterone replacement therapies are perceived to create or create health risks, our sales of Testim may decrease and our operations may be harmed.
Recent studies of female hormone replacement therapy products have reported an increase in health risks. As a result of such studies, some companies that sell or develop female hormone replacement products have experienced decreased sales of these products, and in some cases, a decline in the value of their stock. Publications have, from time to time, suggested potential health risks associated with TRT. Potential health risks are described in various articles, including a 2002 article published inEndocrine Practice and a 1999 article published in theInternational Journal of Andrology. The potential health risks detailed are fluid retention, sleep apnea, breast tenderness or enlargement, increased red blood cells, development of clinical prostate disease, including prostate cancer, increased cardiovascular disease risk and the suppression of sperm production. It is possible that studies on the effects of TRT could demonstrate these or other health risks. This, as well as negative publicity about the risks of hormone replacement therapy, including TRT, could adversely affect patient or prescriber attitudes and impact Testim sales. In addition investors may become concerned about these issues and decide to sell our common stock. These factors could adversely affect our business and the price of our common stock.
Testim competes in a very competitive market, and if we are unable to compete effectively with the other companies that produce products for the treatment of urologic or sexual health disorders, our ability to generate revenues will be limited.
The primary competition for Testim is AndroGel®, marketed by Solvay. AndroGel® was launched approximately three years before Testim and, according to IMS, has a much larger share of the testosterone gel market than we do and also accounted for approximately 58% of total TRT prescriptions for the year ended December 31, 2005. Furthermore, Solvay is a much larger company than we are with greater resources and greater ability to promote its product through a larger sales force. Testim also competes with other forms of testosterone replacement therapies such as oral treatments, patches, injectables and a buccal tablet. Androderm® is a transdermal testosterone patch marketed by Watson. Androderm® is the leading patch product and accounted for 10% of total TRT prescriptions for the fourth quarter of 2005. Many of our competitors, such as Solvay and Watson, have substantially greater financial, technical and human resources than we have. These resources may be used to more effectively develop, market or acquire products and technologies. Additional mergers and acquisitions in the pharmaceutical industry may result in even more resources being concentrated in our competitors. Competition may increase further as a result of advances made in the commercial applicability of technologies and greater availability of capital for investment in these fields. In addition, our competitors are developing new testosterone treatment therapies.
The pharmaceutical industry is highly competitive. Our success will depend on our ability to acquire, develop and commercialize products and our ability to establish and maintain markets for Testim or any products
38
Table of Contents
for which we receive marketing approval. Potential competitors in North America, Europe and elsewhere include major pharmaceutical companies, specialty pharmaceutical companies and biotechnology firms, universities and other research institutions and government agencies. Many of our competitors have more substantial research and development capabilities and experience, and more substantial management, manufacturing, distribution, marketing and financial resources, than we do. Accordingly, our competitors may:
| • | develop or license products or other novel technologies that are more effective, safer or less costly than Testim or product candidates that are being developed by us; |
| • | obtain regulatory approval for products before we do; or |
| • | commit more resources than we can to developing, marketing and selling competing products. |
Other new treatments are being sought for TRT, including a new isomer of an old product for centrally mediated treatment of primary hypogonadism as well as a new class of drugs called Selective Androgen Receptor Modulators, or SARMs. These products are in the early stages of development and their future impact on the treatment of testosterone deficiency is unknown.
If other pharmaceutical companies develop generic versions of any products that compete with our commercialized products or any of our products, our business may be adversely affected.
Other pharmaceutical companies may develop generic versions of any products that compete with our commercialized products or any of our products that are not subject to patent protection or other proprietary rights. For example, because the ingredients of Testim are commercially available to third parties, it is possible that competitors may design formulations, propose dosages or develop methods of administration that would be outside the scope of the claims of one or more, or of all, of the patent rights that we in-license. This would enable their products to effectively compete with Testim. Governmental and other pressures to reduce pharmaceutical costs may result in physicians writing prescriptions for these generic products. Increased competition from the sale of competing generic pharmaceutical products could cause a material decrease in revenue from our products. Additionally, if a generic form of Testim is approved and sold in the U.S., our co-promotion partner for Testim may terminate our co-promotion agreement which could cause a material decrease in revenue from Testim.
The primary competition for Testim is AndroGel®, marketed by Solvay. In July 2003, Watson and Par filed abbreviated new drug applications, or ANDAs, with the FDA to be approved as generics for AndroGel. In response to these ANDAs, Solvay filed patent infringement lawsuits against these two companies to block the approval and marketing of the generic products. Any generic for AndroGel will not be a generic for Testim. On November 1, 2004, Par’s partner, Paddock, received tentative approval of its ANDA from the FDA, and on January 12, 2006, Watson received approval of its ANDA from the FDA. In January 2006, the thirty-month stay in each patent action expired. We believe that Watson was the first to file an ANDA with FDA and, therefore, Watson, not Par/Paddock, is entitled to launch first with six-month exclusivity. As a result, Watson will have to decide whether to (1) launch before a final outcome in the patent litigation and risk treble damages if its product is found to infringe the Solvay patent or (2) wait for the final outcome of the litigation before launch. Any generic for AndroGel will not be a generic for Testim, since Testim is BX rated (or non-substitutable) with AndroGel. However if one of the companies chooses to market their product this would likely result in increased competition for Testim, most likely, at lower prices. Other pharmaceutical companies may develop generic versions of any products that we commercialize that are not subject to patent protection or other proprietary rights. Governmental and other cost containment pressures may result in physicians writing prescriptions for these generic products.
If third-party payors do not reimburse customers for Testim or any of our product candidates that are approved for marketing, they might not be used or purchased, and our revenues and profits will not grow.
Our revenues and profits depend heavily upon the availability of coverage and reimbursement for the use of Testim, and any of our product candidates that are approved for marketing, from third-party healthcare and state
39
Table of Contents
and federal government payors, both in the U.S. and in foreign markets. Reimbursement by a third-party payor may depend upon a number of factors, including the third-party payor’s determination the product is:
| • | competitively priced; |
| • | safe, effective and medically necessary; |
| • | appropriate for the specific patient; |
| • | cost-effective; and |
| • | neither experimental nor investigational. |
Since reimbursement approval for a product is required from third-party and government payors, seeking this approval, particularly when seeking approval for a preferred form of reimbursement over other competitive products, is a time-consuming and costly process. Third-party payors may require cost-benefit analysis data from us in order to demonstrate the cost-effectiveness of any product we might bring to market. For any individual third-party payor, we may not be able to provide data sufficient to gain reimbursement on a similar or preferred basis to competitive products or at all. Once reimbursement at an agreed level is approved by a third-party payor, we may lose that reimbursement entirely or we may lose the similar or better reimbursement we receive compared to competitive products. As reimbursement is often approved for a period of time, this risk is greater at the end of the time period, if any, for which the reimbursement was approved. To date, we have not experienced any problems with third-party payors. This does not mean that we will not experience any problems with third-party payors in the future. We do not know what the impact of changes to reimbursement programs, including Medicare Part D, might be.
International commercialization of Testim and our product candidates faces significant obstacles.
As with Testim, we may plan to commercialize some of our product candidates internationally through collaborative relationships with foreign partners. We have limited foreign regulatory, clinical and commercial resources. We may not be able to enter into collaboration agreements with appropriate partners for important foreign markets on acceptable terms, or at all. Future collaborations with foreign partners may not be effective or profitable for us.
If incremental sales generated by our co-promotion partnership do not offset our obligations under the agreement, our profitability and results of operations may be negatively affected, which could cause the price of our common stock to decline.
In April 2005, we entered into a co-promotion agreement with Oscient. Under the terms of the agreement, Oscient promotes Testim to primary care physicians in the U.S. using its sales force, while we continue to promote Testim using our specialty sales force that calls on urologists, endocrinologists and select primary care physicians. With the exception of 2005, when there is disproportionate sharing of promotional expenses, we are obligated to share equally with Oscient an agreed upon amount of Testim promotional expenses relating to U.S. primary care physicians. In addition, we are obligated to pay Oscient a specified percentage of the gross profit from Testim sales attributable to primary care physicians in the U.S. that exceeds a specified sales threshold. The specific percentage is based upon Testim sales levels attributable to primary care physicians and the marketing expenses incurred by Oscient in connection with the promotion of Testim under the agreement. We anticipate that our selling and marketing expenses will increase significantly as a result of our obligations under the agreement. If we are unable to effectively utilize the combined sales forces or if the additional selling efforts do not sufficiently increase Testim sales, our profitability may be negatively affected, which may cause our stock price to decline.
In addition, we may be required to make termination payments under the agreement to Oscient upon the occurrence of certain termination events, such as in the event a generic form of Testim is approved and sold in the U.S. or in the event there is an interruption in supply of Testim. Such payments will adversely affect our results of operations.
40
Table of Contents
Healthcare reform measures could adversely affect our business.
The business and financial condition of pharmaceutical companies is affected by the efforts of governmental and third-party payors to contain or reduce the costs of healthcare. In the U.S. and in foreign jurisdictions there have been, and we expect that there will continue to be, a number of legislative and regulatory proposals aimed at changing the healthcare system. For example, in some countries other than the U.S., pricing of prescription drugs is subject to government control, and we expect proposals to implement similar controls in the U.S. to continue. In particular, in January 2006, new Medicare prescription drug coverage legislation (Medicare Part D) was implemented. The prescription drug program established by this legislation and future amendments or regulatory interpretations of the legislation could have the effect of reducing the prices that we are able to charge for any products we develop and sell through these plans. This prescription drug legislation and related amendments or regulations could also cause third-party payors other than the federal government, including the states under the Medicaid program, to discontinue coverage for Testim or any products we develop or to lower reimbursement amounts that they pay.
Further federal, state and foreign healthcare proposals and reforms are likely. While we cannot predict the legislative or regulatory proposals that will be adopted or what effect those proposals may have on our business, including the future reimbursement status of any of our product candidates, the pendency or approval of such proposals could result in a decrease in our stock price or limit our ability to raise capital or to obtain strategic partnerships or licenses.
We could be negatively impacted by future interpretation or implementation of federal and state fraud and abuse laws, including anti-kickback laws, false claims laws and federal and state anti-referral laws.
We are subject to various federal and state laws pertaining to health care fraud and abuse, including anti-kickback laws, false claims laws and physician self-referral laws. Violations of these laws are punishable by criminal and/or civil sanctions, including, in some instances, imprisonment and exclusion from participation in federal and state health care programs, including Medicare, Medicaid, and veterans’ health programs. We have not been challenged by a governmental authority under any of these laws and believe that our operations are in compliance with such laws.
However, because of the far-reaching nature of these laws, we may be required to alter one or more of our practices to be in compliance with these laws. Health care fraud and abuse regulations are complex, and even minor, inadvertent irregularities can potentially give rise to claims that the law has been violated. Any violations of these laws could result in a material adverse effect on our business, financial condition and results of operations. If there is a change in law, regulation or administrative or judicial interpretations, we may have to change our business practices or our existing business practices could be challenged as unlawful, which could have a material adverse effect on our business, financial condition and results of operations.
We could become subject to false claims litigation under federal or state statutes, which can lead to civil money penalties, criminal fines and imprisonment, and/or exclusion from participation in federal health care programs. These false claims statutes include the federal False Claims Act, which allows any person to bring suit alleging the false or fraudulent submission of claims for payment under federal programs or other violations of the statute and to share in any amounts paid by the entity to the government in fines or settlement. Such suits, known as qui tam actions, have increased significantly in recent years and have increased the risk that companies like us may have to defend a false claim action. We could also become subject to similar false claims litigation under state statutes. If we are unsuccessful in defending any such action, such action may have a material adverse effect on our business, financial condition and results of operations.
If product liability lawsuits are brought against us, we may incur substantial liabilities.
The commercialization of Testim and the clinical testing and, if approved, commercialization of our product candidates involves significant exposure to product liability claims. We have clinical trial and product liability
41
Table of Contents
insurance that covers Testim and the clinical trials of our other product candidates that we believe is adequate in both scope and amount and has been placed with what we believe are reputable insurers. We are self-insured for the first $1.0 million of liability under these policies. Our product liability policies have been written on a claims-made basis. If any of our product candidates are approved for marketing, we may seek additional coverage. We cannot predict all of the adverse health events that Testim or our product candidates may cause. As a result, our current and future coverages may not be adequate to protect us from all the liabilities that we may incur. If losses from product liability claims exceed our insurance coverage, we may incur substantial liabilities that exceed our financial resources. Whether or not we were ultimately successful in product liability litigation, such litigation could consume substantial amounts of our financial and managerial resources, and might result in adverse publicity, all of which would impair our business. We may not be able to maintain our clinical trial insurance or product liability insurance at an acceptable cost, if at all, and this insurance may not provide adequate coverage against potential claims or losses. If we are required to pay a product liability claim, we may not have sufficient financial resources and our business and results of operations may be harmed.
We may be exposed to liability claims associated with the use of hazardous materials and chemicals.
Our research and development activities and our commercial product, Testim, involve the use of testosterone and large amounts of alcohol which are classified as hazardous materials and chemicals. AA4500 is a biologic product. Biologic products may present a manufacturing health hazard due to risk of infection with the bacterial cell line used to produce the product or with potential viral contamination with the fermentation. Although we believe that our safety procedures for using, storing, handling and disposing of these materials comply with federal, state and local laws and regulations, we cannot completely eliminate the risk of accidental injury or contamination from these materials. In the event of such an accident, we could be held liable for any resulting damages and any liability could materially adversely affect our business, financial condition and results of operations. In addition, the federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous or radioactive materials and waste products may require us to incur substantial compliance costs that could materially adversely affect our business and financial condition. To our knowledge, we have not been the subject of any investigation by any agency or authority for failure to comply with any rules or regulations applicable to hazardous materials or chemicals. We do not maintain specific insurance for the handling of biological, hazardous and radioactive materials. We have contracts with third-party providers for the storage and disposal of hazardous waste and believe that any claims against us in these areas would be the responsibility of these third parties. However, we may be held responsible for these claims despite the fact that we have contracted with third parties for the storage and disposal of hazardous waste. If we are exposed to these types of claims, we could be held responsible for liabilities that exceed our financial resources, which could severely affect our operations.
Risks Related to Our Dependence on Third-Party Manufacturers and Service Providers
Since we rely on third-party manufacturers and suppliers, we may be unable to control the availability or cost of manufacturing our products, which could adversely affect our results of operations.
We do not manufacture Testim or any of our product candidates. Testim is manufactured for us by DPT under a contract that expires on December 31, 2010. We are in the process of qualifying a back-up supplier to manufacture Testim. We have contracted with Cobra for the production of our clinical supply of AA4500, our product candidate for the treatment of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome, and we have contracted with Althea to fill and lyophilize the AA4500 bulk substance produced by Cobra, manufacture placebo and produce sterile diluent. We plan to contract with one or more third-party manufacturers to manufacture our transmucosal film product candidates. The manufacture of pharmaceutical products requires significant expertise and capital investment. DPT, Cobra, Althea, any back-up supplier for Testim or AA4500, or any other third-party manufacturer may encounter difficulties in production. These problems may include:
| • | difficulties with production costs and yields; |
| • | availability of clinical supplies; |
42
Table of Contents
| • | quality control and assurance; |
| • | shortages of qualified personnel; |
| • | compliance with strictly enforced federal, state and foreign regulations; and |
| • | lack of capital funding. |
DPT, Cobra, Althea, any back-up supplier for Testim or AA4500, or any of our other third-party manufacturers may not perform as agreed or may terminate its agreement with us, which would adversely impact our ability to produce and sell Testim or our ability to produce AA4500 or other product candidates for clinical trials.
Under our contract with DPT, DPT is required to qualify its Lakewood, New Jersey facility as a secondary site for the production of Testim. Prior to DPT’s qualification of the Lakewood facility, we are permitted to qualify a back-up supplier. Although we are in the process of qualifying a back-up supplier to manufacture Testim, we do not have an alternate manufacturer of Testim at this time. We have capitalized as work-in-process inventory $1.1 million of Testim manufactured by a back-up supplier. We intend to sell this inventory after we receive approval of the alternate testing site and test method from the FDA. We capitalized this inventory based on favorable preliminary results from analytical testing, past product experience and our belief that it is probable that the inventory will be saleable. If FDA does not approve the alternate testing site and test method, we will have to write-off this inventory. If there is an interruption in the supply of Testim, our co-promotion partner for Testim may terminate our co-promotion agreement and in that event, we would be obligated to make a payment to Oscient the amount of which depends on when the supply interruption occurs. The number of third-party manufacturers with the expertise, required regulatory approvals and facilities to manufacture bulk drug substance on a commercial scale is extremely limited, and it would take a significant amount of time to arrange and receive regulatory approval for alternative arrangements. We may not be able to contract for the manufacturing of Testim on acceptable terms, if at all, which would materially impair our business.
Any of these factors could increase our costs and result in us being unable to effectively commercialize or develop our products. Furthermore, if DPT or any other third-party manufacturer fails to deliver the required commercial quantities of finished product on a timely basis and at commercially reasonable prices, we may be unable to meet the demand for our products and we may lose potential revenues.
We rely on a single source supplier and a limited number of suppliers for two of the primary ingredients for Testim and the loss of any of these suppliers could prevent us from selling Testim, which would materially harm our business.
We rely on third-party suppliers for our supply of testosterone and CPD two key ingredients of Testim. Testosterone is available to us from only two sources. We rely exclusively on one outside source for our supply of CPD. We do not have any agreements with these suppliers regarding these key ingredients. If either of the two sources that produce testosterone stops manufacturing it, or if we are unable to procure testosterone on commercially favorable terms, we may be unable to continue to produce or sell Testim on commercially viable terms, if at all. In addition, if our third-party source of CPD stops manufacturing pharmaceutical grade CPD, or does not make CPD available to us on commercially favorable terms, we may be unable to continue to produce or sell Testim on commercially viable terms, if at all. Furthermore, the limited number of suppliers of testosterone and CPD may provide such companies with greater opportunity to raise their prices. Any increase in price for testosterone or CPD may reduce our gross margins.
Due to our reliance on contract research organizations or other third parties to assist us in conducting clinical trials, we are unable to directly control all aspects of our clinical trials.
Currently, we rely in part on third parties to conduct our clinical trials for the expansion of Testim and the development of our product candidates. As a result, we have had and will continue to have less control over the
43
Table of Contents
conduct of the clinical trials, the timing and completion of the trials and the management of data developed through the trial than would be the case if we were relying entirely upon our own staff. Communicating with outside parties can also be challenging, potentially leading to mistakes as well as difficulties in coordinating activities. Outside parties may:
| • | have staffing difficulties; |
| • | experience regulatory compliance issues; or |
| • | undergo changes in priorities or may become financially distressed. |
These factors may adversely affect their willingness or ability to conduct our trials. We may experience unexpected cost increases that are beyond our control. Problems with the timeliness or quality of the work of a contract research organization may lead us to seek to terminate the relationship and use an alternative service provider. However, making this change may be costly and may delay our trials, and contractual restrictions may make such a change difficult or impossible. Our contracts with the contract research organizations on which we currently rely are each terminable upon 30-days prior written notice. If we must replace any of these contract research organizations or any other contract research organization we may use in the future to conduct our clinical trials, our trials may have to be suspended until we find another contract research organization that offers comparable services. The time that it takes us to find alternative organizations may cause a delay in the commercialization of our product candidates or may cause us to incur significant expenses to replicate data that may be lost. Although we do not believe that the contract research organizations on which we rely offer services that are not available elsewhere, it may be difficult to find a replacement organization that can conduct our trials in an acceptable manner and at an acceptable cost. Any delay in or inability to complete our clinical trials could significantly compromise our ability to secure regulatory approval of the relevant product candidate and preclude our ability to commercialize the product, thereby limiting our ability to generate revenue from the sales of products other than Testim, which may result in a decrease in our stock price.
Our third-party manufacturers are subject to regulatory oversight, which may delay or disrupt our development and commercialization efforts.
Third-party manufacturers of our products or product candidates must ensure that all of the processes, methods and equipment are compliant with the current Good Manufacturing Practices, or cGMP, and conduct extensive audits of vendors, contract laboratories and suppliers. The cGMP requirements govern quality control of the manufacturing process and documentation policies and procedures. Compliance by third-party manufacturers with cGMP requires record keeping and quality control to assure that the product meets applicable specifications and other requirements. Manufacturing facilities are subject to inspection by regulatory agencies at any time. If an inspection by regulatory authorities indicates that there are deficiencies, third-party manufacturers could be required to take remedial actions, stop production or close the facility, which would disrupt the manufacturing processes and limit the supplies of Testim or our product candidates. If they fail to comply with these requirements, we also may be required to curtail the clinical trials of our product candidates, which are also supplied by these manufacturers, and may not be permitted to sell our products or may be limited in the jurisdictions in which we are permitted to sell them.
Approximately 91% of our product shipments are to only four customers; if any of these customers refuse to distribute Testim on commercially favorable terms, or at all, our business will be adversely affected.
We sell Testim to wholesale drug distributors and chain drug stores who generally sell products to retail pharmacies, hospitals and other institutional customers. We do not promote Testim to these customers, and they do not determine Testim prescription demand. However, approximately 91% of our product shipments during the twelve-month period ended December 31, 2005 were to only four customers: Cardinal Health, Inc., McKesson Corporation, AmerisourceBergen Corporation and Walgreen Co. Our business would be harmed if any of these customers refused to distribute Testim or refuse to purchase Testim on commercially favorable terms to us.
44
Table of Contents
It is possible that wholesalers could decide to change their policies or fees, or both, at some time in the future. This could result in their refusal to distribute smaller volume products, or cause higher product distribution costs, lower margins or the need to find alternative methods of distributing products. Such alternative methods may not exist or may not be economically viable.
If we are unable to grow our sales, marketing or distribution capabilities or enter into agreements with third parties to perform some of these functions, or our current co-promotion partner for Testim does not perform as expected, we will not be able to grow our business.
As an organization, we have a limited history in sales, marketing and distribution. To directly market and distribute Testim or any of our product candidates which receive regulatory approval, we must continue to enhance our sales and marketing efforts. For our direct sales efforts, we will need to hire additional salespeople in order to grow and to manage any turnover that occurs in our sales force. For some market opportunities, we may need to enter into co-promotion agreements such as our agreement with Oscient or other licensing arrangements with pharmaceutical or biotechnology firms in order to increase the commercial success of our products. The success of our co-promotion agreement with Oscient is dependent upon Oscient’s performance and the amount of resources Oscient devotes to Testim promotion. The failure of Oscient to devote sufficient resources or any disagreement with Oscient as to whether Oscient is performing under the agreement could preclude us from gaining any benefit from the Oscient agreement and significantly distract our management from their operational responsibilities, which would negatively impact our ability to grow our business.
We may not be able to grow our direct sales, marketing and distribution capabilities or enter into future co-promotion or similar licensing arrangements with third parties in a timely manner, or on acceptable terms. To the extent that we enter into future co-promotion or other licensing arrangements, our product revenues may be lower than if we directly marketed and sold our products, and some or all of the revenues we receive will depend upon the efforts of third parties, and these efforts may not be successful or our future partners may not devote sufficient resources to our products. Additionally, building marketing and distribution capabilities may be more expensive than we anticipate, requiring us to divert capital from other intended purposes or preventing us from building our marketing and distribution capabilities to the desired levels.
Risks Related to Intellectual Property
If we breach any of the agreements under which we license commercialization rights to products or technology from others, we could lose license rights that are critical to our business.
We are a party to a number of license agreements by which we have rights to use the intellectual property of third parties that are necessary for us to operate our business. In particular, we have obtained the exclusive right to develop and commercialize Testim pursuant to a license agreement with Bentley. Bentley may unilaterally terminate the agreement if we fail to make payments under this agreement and this failure continues for a period of 30 days following written notice to us by Bentley. If the agreement is properly terminated by Bentley, we may not be able to manufacture or sell Testim.
Additionally, we have obtained exclusive rights to make and sell products that are used for hormone replacement, to treat any type of urologic disorder or as a pain therapy incorporating PharmaForm’s transmucosal film technology. This agreement continues for the life of the licensed patent rights. Either party may terminate this agreement under certain events of bankruptcy or insolvency by the other party. PharmaForm may unilaterally terminate this agreement if:
| • | we fail to make payments under this agreement and this failure continues for a period of 30 days following written notice to us by PharmaForm; or |
| • | we fail to initiate clinical trials within two years of availability of final formulation in quantities adequate for clinical testing and associated documentation for clinical trials. |
45
Table of Contents
If our agreement with PharmaForm is properly terminated by PharmaForm, we may not be able to execute our strategy to develop, manufacture or sell transmucosal film products. To the extent that unpatented PharmaForm trade secrets are necessary for the manufacture of the transmucosal product candidates, our license to such trade secrets would expire with the expiration of the licensed patents in 2020 or later and potentially impair our continued commercialization of our transmucosal product candidates thereafter.
We have also obtained exclusive worldwide rights from BioSpecifics to develop, market and sell products, other than dermal formulations labeled for topical administration, that contain BioSpecifics’ enzyme, which we refer to as AA4500, for the treatment of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome. We may expand the agreement, at our option, to license products which contain BioSpecifics’ enzyme for other indications, excluding dermal formulations labeled for topical administration, as they are developed by BioSpecifics or by us. This agreement continues on a product by product and country by country basis until the later of:
| • | the last to expire valid claim of a patent covering such product; |
| • | the expiration of the regulatory period conveyed by orphan drug designation with respect to such product; and |
| • | 12 years. |
Upon expiration of the agreement pursuant to the preceding sentence, we will have a fully paid-up license to the products developed under the agreement. Either party may terminate this agreement in the event of bankruptcy or insolvency by the other party. Additionally, either party may terminate this agreement if the other party is in material breach of its obligations under the agreement which continues for a period of 90 days following receipt of written notice of such material breach. We may terminate this agreement in its entirety, or on a country by country basis, on an indication by indication basis, or on a product by product basis, at any time upon 90 days prior written notice to BioSpecifics. If this agreement is properly terminated by BioSpecifics, we may not be able to execute our strategy to develop and commercialize AA4500 for the treatment of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome or to develop and commercialize future product candidates utilizing BioSpecifics’ enzyme.
We expect to enter into additional licenses in the future. These licenses may impose various development, commercialization, funding, royalty, diligence or other obligations on us. If we breach any of these obligations, the licensor may have the right to terminate the license or render the license non-exclusive, which could make it impossible for us to develop, manufacture or sell the products covered by the license.
Disputes may arise with respect to our licensing agreements regarding manufacturing, development and commercialization of any products relating to our in-licensed intellectual property, including Testim. These disputes could lead to delays in or termination of the development, manufacture and commercialization of Testim or our product candidates or to litigation.
We have only limited patent protection for Testim and our product candidates, and we may not be able to obtain, maintain and protect proprietary rights necessary for the development and commercialization of Testim or our product candidates.
Our business and competitive positions are dependent upon our ability to obtain and protect our proprietary position for Testim and our product candidates in the U.S., Europe and elsewhere. Because of the substantial length of time and expense associated with development of new products, we, along with the rest of the biopharmaceutical industry, place considerable importance on obtaining and maintaining patent protection for new technologies, products and processes. The patent positions of pharmaceutical, biopharmaceutical and biotechnology companies, including ours, are generally uncertain and involve complex legal and factual questions. Our and our licensors’ patents and patent applications may not protect our technologies and products because, among other things:
| • | there is no guarantee that any of our or our licensors’ pending patent applications will result in issued patents; |
46
Table of Contents
| • | we may develop additional proprietary technologies that are not patentable; |
| • | there is no guarantee that any patents issued to us, our collaborators or our licensors will provide us with any competitive advantage or cover our product candidates; |
| • | there is no guarantee that any patents issued to us or our collaborators or our licensors will not be challenged, circumvented or invalidated by third parties; and |
| • | there is no guarantee that any patents previously issued to others or issued in the future will not have an adverse effect on our ability to do business. |
We attempt to protect our intellectual property position by filing or obtaining licenses to patent applications and, where appropriate, patent applications in other countries related to our proprietary technology, inventions and improvements that are important to the development of our business. Our PharmaForm transmucosal film product candidates are covered by a formulation patent in the U.S. only. This patent expires in 2020. Testosterone, the active ingredient in Testim, is off-patent and is included in competing TRT products. The U.S. patent that we license from Bentley covers the composition of matter and formulation of Testim and expires in June 2008. The European and Japanese patents that we license from Bentley for Testim expire in November 2006. The Canadian patent that we license from Bentley expires in January 2010. In July 2005, an additional patent covering Testim issued in Canada, and this patent is included in our license from Bentley. This patent expires in 2023. Bentley has filed a new patent application in the U.S., Europe and Japan which, if issued, could provide additional patent protection for Testim.
AA4500 licensed from BioSpecifics is covered by two method of use patents in the U.S., one for the treatment of Dupuytren’s Disease and one for the treatment of Peyronie’s Disease. The Dupuytren’s patent expires in 2014, and the Peyronie’s patent expires in 2019. The patents are limited to the use of AA4500 for the treatment of Dupuytren’s and Peyronie’s Diseases, respectively, with certain dose ranges and/or concentration ranges. In January 2005, a patent application was filed with regard to AA4500 for the treatment of Frozen Shoulder Syndrome. If this patent is granted, it would expire in 2026. Currently there is not enough data to establish whether any approved product for Dupuytren’s Disease or Peyronie’s Disease or Frozen Shoulder Syndrome will be covered by these patents. In January 2006, we filed a patent application with regard to the manufacturing process for AA4500; if this patent is granted, it would expire in January 2027.
Foreign patents and pending applications may also cover these products in certain countries depending upon the concentration and dosage ranges of such products. Some countries will not grant patents on patent applications that are filed after the public sale or disclosure of the material claimed in the patent application. The U.S., by contrast, allows a one year grace period after public disclosure in which to file a patent application. Because the European patent application was filed after Testim went on the market in the U.S., our ability to obtain additional patent protection outside of the U.S. may be limited.
The standards that the USPTO and its foreign counterparts use to grant patents are not always applied predictably or uniformly and can change. Limitations on patent protection in some countries outside the U.S., and the differences in what constitutes patentable subject matter in these countries, may limit the protection we seek outside of the U.S. For example, methods of treating humans are not patentable subject matter in many countries outside of the U.S. In addition, laws of foreign countries may not protect our intellectual property to the same extent as would laws of the U.S. In determining whether or not to seek a patent or to license any patent in a particular foreign country, we weigh the relevant costs and benefits, and consider, among other things, the market potential of our product candidates in the jurisdiction, and the scope and enforceability of patent protection afforded by the law of the jurisdiction. Failure to obtain adequate patent protection for our proprietary product candidates and technology would impair our ability to be commercially competitive in these markets. Accordingly, we do not know the degree of future protection for our proprietary rights or the breadth of claims allowed in any patents issued to us or others.
47
Table of Contents
We intend to seek patent protection whenever it is available for any products or product candidates we acquire in the future. However, any patent applications for future products may not issue as patents, and any patent issued on such products may be challenged, invalidated, held unenforceable or circumvented. Furthermore, the claims in patents which have been issued on products we may acquire in the future may not be sufficiently broad to prevent third parties from commercializing competing products. If we fail to obtain adequate patent protection for our products, our ability to compete could be impaired.
Technology in-licensed by us is important to our business. We may not control the patent prosecution, maintenance or enforcement of our in-licensed technology. Accordingly, we may be unable to exercise the same degree of control over this intellectual property as we would over our internally developed intellectual property. For example, in-licensed patents for which our rights are limited to a particular field of use could be or become licensed in other fields to other licensees and, therefore, become subject to enforcement by such other licensees without our participation. Consequently, such licensed patents could be held invalid or unenforceable or could have claims construed in a manner adverse to our interests in litigation, which we would not control or to which we would not be a party. If any of the intellectual property rights of our licensors is found to be invalid, this could have a material adverse impact on our operations.
We control the filing, prosecution and maintenance of BioSpecifics’ patents relating to products licensed under our development and license agreement with Biospecifics. Pursuant to the terms of the agreement, we must pay a specified percentage of the fees and expenses incurred by us in connection with filing, prosecution and maintenance of BioSpecifics’ patents and shall receive a credit against future royalties payable to BioSpecifics under our agreement with BioSpecific equal to a specified percentage of such fees and expenses. Additionally, BioSpecifics shall reimburse us for 100% of fees and expenses in connection with filings, prosecution and/or maintenance of patents pertaining to pharmaceutical applications containing BioSpecifics’ enzyme for which we decline to exercise our option under the development and license agreement to pursue and which BioSpecifics pursues.
The patent relating to Dupuytren’s Disease is the subject of a a reissue application in the USPTO for, among other things, the purpose of submitting prior art that was not previously submitted during the prosecution of the Dupuytren’s patent. As a result, the patent may not be allowed by the USPTO, and therefore, may not be valid and enforceable.
If we are not able to protect and control unpatented trade secrets, know-how and other technological innovation, we may suffer competitive harm. We also rely on trade secrets, know-how and technology, which are not protected by patents, to maintain our competitive position. However, trade secrets are difficult to protect. To maintain the confidentiality of trade secrets and proprietary information, we generally seek to enter into confidentiality agreements with our employees, consultants and collaborators upon the commencement of a relationship with us. However, we may not obtain these agreements in all circumstances. Nor can we guarantee that these agreements will provide meaningful protection, that these agreements will not be breached, or that we will have an adequate remedy for any such breach. In addition, adequate remedies may not exist in the event of unauthorized use or disclosure of this information. Others may have developed, or may develop in the future, substantially similar or superior know-how and technology. The loss or exposure of our trade secrets, know-how and other proprietary information, as well as independent development of similar or superior know-how, could harm our operating results, financial condition and future growth prospects. Many of our employees and consultants were, and many of our consultants may currently be, parties to confidentiality agreements with other companies. Although our confidentiality agreements with these employees and consultants require that they do not bring to us, or use without proper authorization, any third party’s proprietary technology, if they violate their agreements, we could suffer claims or liabilities.
48
Table of Contents
We may have to engage in costly litigation to enforce or protect our proprietary technology or to defend challenges to our proprietary technology by our competitors, which may harm our business, results of operations, financial condition and cash flow.
The pharmaceutical field is characterized by a large number of patent filings involving complex legal and factual questions, and, therefore, we cannot predict with certainty whether our licensed patents will be enforceable. Competitors may have filed applications for or have been issued patents and may obtain additional patents and proprietary rights related to products or processes that compete with or are similar to ours. We may not be aware of all of the patents potentially adverse to our interests that may have been issued to others. Litigation may be necessary to protect our proprietary rights, and we cannot be certain that we will have the required resources to pursue litigation or otherwise to protect our proprietary rights.
Competitors may infringe our patents or successfully avoid them through design innovation. To prevent infringement or unauthorized use, we may need to file infringement lawsuits, which are expensive and time-consuming. In particular, if a competitor were to file a paragraph IV certification under the United States Drug Price Competition and Patent Term Restoration Act of 1984, known as the Hatch-Waxman Act, in connection with that competitor’s submission to the FDA of an abbreviated new drug application, or ANDA, for approval of a generic version of any of our products for which we believed we held a valid patent, then we would have 45 days in which to initiate a patent infringement lawsuit against such competitor. In any infringement proceeding, a court may decide that a patent of ours is not valid or is unenforceable, may narrow our patent claims or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover its technology. If a court so found that one of our patents was invalid or not infringed in an infringement suit under paragraph IV of the Hatch-Waxman Act, then the FDA would be permitted to approve the competitor’s ANDA resulting in a competitive generic product.
Our ability to market our products may be impaired by the intellectual property rights of third parties.
Our commercial success will depend in part on not infringing the patents or proprietary rights of third parties. We are aware of competing intellectual property relating to the TRT gel market. While we currently believe we have freedom to operate in the TRT gel market, others may challenge our position in the future. We are also aware of at least one third-party patent relating to BioSpecifics’ enzyme that may impair BioSpecifics’ freedom to operate in the manufacture of AA4500. We are also aware of third-party patents relating to PharmaForm’s transmucosal film delivery system that may impair PharmaForm’s freedom to operate in the manufacture of product candidates using a transmucosal film delivery system. There has been, and we believe that there will continue to be, significant litigation in the pharmaceutical industry regarding patent and other intellectual property rights.
Third parties could bring legal actions against us claiming damages and seeking to enjoin clinical testing, manufacturing and marketing of the affected product or products. A third party might request a court to rule that the patents we in-license are invalid or unenforceable. In such a case, even if the validity or enforceability of those patents were upheld, a court might hold that the third party’s actions do not infringe the patent we in-license thereby, in effect, limiting the scope of our patent rights. If we become involved in any litigation, it could consume a substantial portion of our resources, regardless of the outcome of the litigation. If any of these actions are successful, in addition to any potential liability for damages, we could be required to obtain a license to continue to manufacture or market the affected product, in which case we may be required to pay substantial royalties or grant cross-licenses to our patents. However, there can be no assurance that any such license will be available on acceptable terms or at all. Ultimately, we could be prevented from commercializing a product, or forced to cease some aspect of our business operations, as a result of patent infringement claims, which could harm our business.
We may not be able to obtain or maintain orphan drug exclusivity for our product candidates, and our competitors may obtain orphan drug exclusivity prior to us, which could significantly harm our business.
Some jurisdictions, including Europe and the U.S., may designate drugs for relatively small patient populations as orphan drugs. The FDA granted orphan drug status to AA4500 in the U.S. for the treatment of
49
Table of Contents
Dupuytren’s and Peyronie’s Diseases. Orphan drug designation must be requested before submitting an application for marketing authorization. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process, but does make the product eligible for orphan drug exclusivity and specific tax credits in the U.S. Generally, if a product with an orphan drug designation subsequently receives the first marketing approval for the indication for which it has such designation, the product is entitled to orphan drug exclusivity. Orphan drug exclusivity means that another application to market the same drug for the same indication may not be approved, except in limited circumstances, for a period of up to 10 years in Europe and for a period of seven years in the U.S. Obtaining orphan drug designations and orphan drug exclusivity for our product candidates, including AA4500 for the treatment of Dupuytren’s and Peyronie’s Diseases, may be critical to the success of these product candidates. Our competitors may obtain orphan drug exclusivity for products competitive with our product candidates before we do, in which case we would be excluded from that market. Even if we obtain orphan drug exclusivity for any of our product candidates, we may not be able to maintain it. For example, if a competitive product is shown to be clinically superior to our product, any orphan drug exclusivity we have obtained will not block the approval of such competitive product. In addition, even if we obtain orphan drug exclusivity for any of our product candidates, a viable commercial market may never develop and we may never derive any meaningful revenues from the sales of these products.
If Testim or our future products or product candidates infringe the intellectual property of our competitors or other third parties, we may be required to pay license fees or cease these activities and pay damages, which could significantly harm our business.
Even if we have our own patents which protect our products, Testim and our product candidates may nonetheless infringe the patents or violate the proprietary rights of third parties. In these cases, we may be required to obtain-licenses to patents or proprietary rights of others in order to continue to sell and use Testim and develop and commercialize our product candidates. We may not, however, be able to obtain any licenses required under any patents or proprietary rights of third parties on acceptable terms, or at all. Even if we were able to obtain rights to a third party’s intellectual property, these rights may be non-exclusive, thereby giving our competitors potential access to the same intellectual property.
Third parties may assert patent or other intellectual property infringement claims against us, or our licensors or collaborators, with respect to technologies used in potential product candidates. Any claims that might be brought against us relating to infringement of patents may cause us to incur significant expenses and, if successfully asserted against us, may cause us to pay substantial damages. We may not have sufficient resources to effectively litigate these claims. Even if we were to prevail, any litigation could be costly and time-consuming and could divert the attention of our management and key personnel from business operations. In addition, any patent claims brought against our licensors or collaborators could affect their ability to carry out their obligations to us.
Furthermore, if a patent infringement suit were brought against us, or our licensors or collaborators, the development, manufacture or potential sale of product candidates claimed to infringe a third party’s intellectual property may have to cease or be delayed. Ultimately, we may be unable to commercialize one or more of our product candidates, our patent claims may be substantially limited or may have to cease some portion of our operations as a result of patent infringement claims, which could severely harm our business.
50
Table of Contents
Risks Related to Employees and Growth
If we are not able to retain our current management team or attract and retain qualified scientific, technical and business personnel, our business will suffer.
We are dependent on the members of our management team, in particular, our Chief Executive Officer, Gerri Henwood, for our business success. In addition, an important element of our strategy is to leverage the development, regulatory and commercialization expertise of our current management, including Ms. Henwood, in our development activities. We currently carry a $1.0 million key-man life insurance policy on the life of Ms. Henwood. Our employment agreements with our executive officers are terminable on short notice or no notice. The loss of any one of our executive officers may result in a significant loss in the knowledge and experience that we, as an organization, possess and could cause significant delays, or outright failure, in the development and further commercialization of Testim or our product candidates. If we are unable to attract and retain qualified and talented senior management personnel, our business may suffer.
To grow we will need to hire a significant number of qualified commercial, scientific and administrative personnel. However, there is intense competition for human resources, including management in the technical fields in which we operate, and we may not be able to attract and retain qualified personnel necessary for the successful commercialization of Testim and the development and commercialization of our product candidates. As we grow, the inability to attract new employees when needed or to retain existing employees could severely limit our growth and harm our business.
Changes in the expensing of stock-based compensation will result in unfavorable accounting charges and may require us to change our compensation practices. Any change in our compensation practices may adversely affect our ability to attract and retain qualified scientific, technical and business personnel.
We rely heavily on stock options to compensate existing employees and attract new employees. We currently are not required to record stock-based compensation charges if the employee’s stock option exercise price equals or exceeds the fair value of our common stock at the date of grant. We also have an employee stock purchase plan under which employees can purchase our common stock at a discount. The Financial Accounting Standards Board has recently announced new rules for recording expense for the fair value of stock options and purchase rights under employee stock purchase plans. As a result of these new rules, commencing on January 1, 2006, we will expense the fair value of stock options and purchase rights under our employee stock purchase plan, thereby increasing our operating expenses and reported losses. Although we intend to continue to include various forms of equity in our compensation plans, if the extent to which we use forms of equity in our plans is reduced due to the negative effects on earnings, it may be difficult for us to attract and retain qualified scientific, technical and business personnel.
Our operations may be impaired unless we can successfully manage our growth.
As of December 31, 2005 we had 179 full and part-time employees. In order to continue to expand Testim’s sales and commercialize our other product candidates, we currently anticipate that we will need to add some managerial, selling, operational, financial and other employees to our existing departments over each of the next two years. Expansion may place, a significant strain on our management, operational and financial resources. Moreover, higher than expected market growth of Testim, the acquisition or in-licensing of additional products, as well as the development and commercialization of our other product candidates or marketing arrangements with third parties, could accelerate our hiring needs beyond our current expectations. To manage further growth, we will be required to continue to improve existing, and implement additional, operational and financial systems, procedures and controls, and hire, train and manage additional employees. Our current and planned personnel, systems, procedures and controls may not be adequate to support our anticipated growth and we may not be able to hire, train, retain, motivate and manage required personnel. Our failure to manage growth effectively could limit our ability to achieve our business goals. In addition, our direct sales force consists of relatively newly hired employees. Turnover in our direct sales force or marketing team could adversely affect Testim and future product sales growth.
51
Table of Contents
Risks Related to Stock Market Price
Our stock price is likely to be volatile, and the market price of our common stock may drop below the current price.
Prior to our initial public offering in July 2004, there was no public market for our common stock. Since our initial public offering, our stock price has, at times, been volatile.
Market prices for securities of pharmaceutical, biotechnology and specialty pharmaceutical companies have been particularly volatile. Some of the factors that may cause the market price of our common stock to fluctuate include:
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | Testim market acceptance and sales growth; |
| • | growth of the overall androgen market which may be influenced by Solvay’s sales and marketing efforts; |
| • | developments concerning therapies that compete with Testim in the treatment of hypogonadism; |
| • | our ability to manufacture any products to commercial standards; |
| • | results of our clinical trials; |
| • | the regulatory status of our product candidates; |
| • | failure of any of our product candidates, if approved, to achieve commercial success; |
| • | regulatory developments in the U.S. and foreign countries; |
| • | developments or disputes concerning our patents or other proprietary rights; |
| • | public concern over our drugs; |
| • | litigation involving us, our general industry or both; |
| • | future sales of our common stock; |
| • | changes in the structure of healthcare payment systems, including developments in price control legislation; |
| • | departure of key personnel; |
| • | the degree to which our co-promotion arrangements generate revenue; |
| • | period-to-period fluctuations in our financial results or those of companies that are perceived to be similar to us; |
| • | announcements of material events by those companies that are our competitors or perceived to be similar to us; |
| • | changes in estimates of our financial results or recommendations by securities analysts; |
| • | investors’ general perception of us; and |
| • | general economic, industry and market conditions. |
If any of these risks occurs, it could cause our stock price to fall and may expose us to class action lawsuits that, even if unsuccessful, could be costly to defend and a distraction to management.
52
Table of Contents
If we do not comply with registration rights, we may be required to pay substantial damages to certain holders of our restricted securities.
On June 28, 2005, we entered into securities purchase agreements with certain qualified purchasers to sell an aggregate of 8,242,796 shares of common stock and warrants to purchase an aggregate of 2,060,687 shares of common stock with an exercise price of $5.84 per share in a private placement transaction. We completed the sale on June 30, 2005. As required by the terms of the securities purchase agreements, we filed a registration statement with the SEC on August 12, 2005 to register for resale of the shares of common stock and the shares of common stock issuable upon the exercise of the warrants sold in the private placement. Such registration statement was declared effective by the SEC. Under the terms of the securities purchase agreements, as amended, we are obligated to use our reasonable best efforts to maintain the effectiveness of the registration statement. If we fail to meet this obligation certain holders of our securities may bring breach of contract claims against us, and even if such suit is without merit, we may incur substantial costs in defending these claims and substantial damages if we are found to be in breach.
A significant portion of our total outstanding common stock is restricted from resale but may be sold into the market upon removal of the restrictions or registration of the shares. Sales of a substantial number of shares of our common stock in the public market, or the perception in the market that the holders of a large number of shares intend to sell shares, could depress the market price of our common stock and impair our ability to raise capital through the sale of additional equity securities, even if our business is doing well.
As of March 27, 2006, we have 29,291,719 shares of common stock outstanding, of which approximately 14.7 million shares, or 50%, are restricted securities under Rule 144 of the General Rules and Regulations under the Securities Act of 1933. Of these 14.7 million shares, approximately 1.7 million shares of common stock were freely tradable under Rule 144(k) and approximately 13.0 million shares of common stock were eligible for resale under Rule 144, subject to volume limitations. Alternatively, the vast majority of the 14.7 million shares have rights, subject to some conditions, to require us to file registration statements covering their shares or to include their shares in registration statements that we may file for ourselves or other stockholders.
Possible dilutive effect of outstanding options and warrants.
As of December 31, 2005, stock options to purchase 2,101,498 shares of common stock and warrants to purchase 3,793,363 shares of common stock were outstanding. In addition, as of December 31, 2005 a total of 313,614 stock options are available for grant under our 2004 Equity Compensation Plan. A total of 562,870 of the outstanding options and warrants are “in the money” and exercisable as of December 31, 2005. “In the money” means that the current market price of the common stock is above the exercise price of the shares subject to the warrant or option. The issuance of common stock upon the exercise of these options and warrants could adversely affect the market price of the common stock or result in substantial dilution to our existing stockholders.
Our executive officers, directors and major stockholders have the ability to control all matters submitted to stockholders for approval.
At December 31, 2005, our executive officers, directors and certain of our stockholders who beneficially own more than 5% of our outstanding common stock, and their affiliates, in the aggregate, beneficially own shares representing approximately 62% of our common stock. Beneficial ownership includes shares over which an individual or entity has investment or voting power and includes shares that could be issued upon the exercise of options and warrants within 60 days. As a result, if these stockholders were to choose to act together, they would be able to control all matters submitted to our stockholders for approval, as well as our management and
53
Table of Contents
affairs. For example, these individuals, if they chose to act together, will control the election of directors and approval of any merger, consolidation or sale of all or substantially all of our assets. This concentration of voting power could delay or prevent an acquisition of our company on terms that other stockholders may desire.
Provisions in our certificate of incorporation and bylaws and under Delaware law may prevent or frustrate a change in control in management that stockholders believe is desirable.
Provisions of our certificate of incorporation and bylaws may discourage, delay or prevent a merger, acquisition or other change in control that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares. These provisions may also prevent or frustrate attempts by our stockholders to replace or remove our management. These provisions include:
| • | limitations on the removal of directors; |
| • | advance notice requirements for stockholder proposals and nominations; |
| • | the inability of stockholders to act by written consent or to call special meetings; and |
| • | the ability of our board of directors to designate the terms of and issue new series of preferred stock without stockholder approval, which could be used to institute a rights plan that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by our board of directors. |
The affirmative vote of the holders of at least two-thirds of our shares of capital stock entitled to vote is necessary to amend or repeal the above provisions of our certificate of incorporation. In addition, absent approval of our board of directors, our bylaws may only be amended or repealed by the affirmative vote of the holders of at least two-thirds of our shares of capital stock entitled to vote.
In addition, Section 203 of the General Corporation Law of the State of Delaware prohibits a publicly held Delaware corporation from engaging in a business combination with an interested stockholder, generally a person which together with its affiliates owns or within the last three years has owned 15% of our voting stock, for a period of three years after the date of the transaction in which the person became an interested stockholder, unless the business combination is approved in a prescribed manner. Accordingly, Section 203 may discourage, delay or prevent a change in control of our company.
| ITEM 1B. | Unresolved Staff Comments |
None.
| ITEM 2. | Properties |
We currently lease approximately 31,092 square feet of space for our headquarters in Malvern, Pennsylvania, of which approximately 26,130 square feet is dedicated to office space and approximately 4,962 square feet is dedicated to warehousing and packaging operations. The initial term of the lease is 90 months and commenced on June 25, 2005.
The lease for approximately 16,274 square feet of space for our former headquarters in Norristown, Pennsylvania expired on January 31, 2006.
| ITEM 3. | Legal Proceedings |
We are not engaged in any material legal proceedings.
| ITEM 4. | Submission of Matters to a Vote of Security Holders |
No matters were submitted to the vote of stockholders through the solicitation of proxies or otherwise during the quarter ended December 31, 2005.
54
Table of Contents
| ITEM 5. | Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Market Information
Our common stock began trading on the NASDAQ National Market on July 23, 2004 under the symbol “AUXL”. Prior to that time there had been no market for our common stock. The following table sets forth the high and low closing sales prices per share for our common stock for the periods indicated, as reported by the NASDAQ National Market:
Year Ended December 31, 2004: | High | Low | ||||
Third Quarter (beginning July 23) | $ | 9.53 | $ | 6.74 | ||
Fourth Quarter | $ | 9.96 | $ | 7.55 | ||
Year Ended December 31, 2005: | High | Low | ||||
First Quarter | $ | 9.00 | $ | 5.26 | ||
Second Quarter | $ | 5.85 | $ | 4.15 | ||
Third Quarter | $ | 7.20 | $ | 4.79 | ||
Fourth Quarter | $ | 6.50 | $ | 3.90 | ||
Holders of record
As of March 27, 2006, there were approximately 79 holders of record of our common stock. Because many of such shares are held by brokers and other institutions on behalf of stockholders, the Company is unable to estimate the total number of stockholders represented by these record holders.
Dividends
We have never paid or declared any cash dividends on our common stock. We currently intend to retain any earnings for future growth and, therefore, do not expect to pay cash dividends in the foreseeable future.
Securities Authorized for Issuance Under Equity Compensation Plans
The information under the heading “Equity Compensation Plan Information” in our definitive Proxy Statement for our Annual Meeting of Stockholders to be held on June 6, 2006, to be filed with the SEC, is incorporated herein by reference.
Recent Sales of Unregistered Securities
During the quarter ended December 31, 2005, we did not issue any unregistered shares of our common stock.
Use of Proceeds From Registered Securities
On July 28, 2004, we sold 5,500,000 shares of our common stock in our initial public offering at a price of $7.50 per share pursuant to a Registration Statement on Form S-1 (the “Registration Statement”) (Registration No. 333-114685), which was declared effective by the Securities and Exchange Commission on July 23, 2004. After deducting expenses of the offering, we received net offering proceeds of approximately $36.5 million. During the period from the offering through December 31, 2005, we have used the net proceeds from our initial public offering as follows:
| • | approximately $18.3 million to fund further commercialization of Testim, including Phase IV clinical studies; |
55
Table of Contents
| • | approximately $12.7 million for milestone obligations and other costs associated with the development of AA4500 for the treatment of Dupuytren’s and Peyronie’s Diseases and Frozen Shoulder Syndrome; and |
| • | approximately $5.5 million to fund development of AA2600 and AA4010. |
The foregoing amounts represent management’s reasonable estimate of the amount of net offering proceeds applied to such activity instead of the actual amount of net offering proceeds used. The remainder of the proceeds have been invested in short-term investment-grade securities and money market accounts. None of the net proceeds were directly or indirectly paid to: (i) any of our directors, officers or their associates; (ii) any person(s) owning 10% or more of any class of our equity securities; or (iii) any of our affiliates.
| ITEM 6. | Selected Financial Data |
SELECTED CONSOLIDATED FINANCIAL DATA
(In thousands, except share and per share data)
The following selected consolidated financial data should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and the related notes appearing elsewhere in this Report. The consolidated statements of operations data for the years ended December 31, 2005, 2004 and 2003 and the consolidated balance sheet data as of December 31, 2005 and 2004 have been derived from our audited consolidated financial statements and related notes, which are included elsewhere in this Report. The consolidated statement of operations data for the years ended December 31, 2002 and 2001 and the consolidated balance sheet data as of December 31, 2003, 2002 and 2001 have been derived from audited financial statements which do not appear in this Report. The historical results presented are not necessarily indicative of results to be expected in any future period.
| Years Ended December 31, | ||||||||||||||||||||
| 2005 | 2004 | 2003 | 2002 | 2001 | ||||||||||||||||
Consolidated Statement of Operations Data: | ||||||||||||||||||||
Net revenues | $ | 42,804 | $ | 27,025 | $ | 8,822 | $ | — | $ | — | ||||||||||
Operating expenses: | ||||||||||||||||||||
Cost of goods sold | 13,119 | 8,148 | 3,762 | 122 | — | |||||||||||||||
Research and development | 24,301 | 15,993 | 7,175 | 14,130 | 16,294 | |||||||||||||||
Selling, general, and administrative | 43,935 | 31,210 | 27,475 | 5,626 | 2,346 | |||||||||||||||
Total operating expenses | 81,355 | 55,351 | 38,412 | 19,878 | 18,640 | |||||||||||||||
Loss from operations | (38,551 | ) | (28,326 | ) | (29,590 | ) | (19,878 | ) | (18,640 | ) | ||||||||||
Interest income (expense), net | 1,496 | (192 | ) | (1,007 | ) | 334 | 188 | |||||||||||||
Other income (expense), net | (1,203 | ) | — | 1,722 | — | — | ||||||||||||||
Net loss | (38,258 | ) | (28,518 | ) | (28,875 | ) | (19,544 | ) | (18,452 | ) | ||||||||||
Accretion of redeemable convertible preferred stock | — | (395 | ) | (490 | ) | (384 | ) | (154 | ) | |||||||||||
Deemed dividend to warrant holders | — | (173 | ) | — | — | — | ||||||||||||||
Dividends accrued on redeemable convertible preferred stock | — | — | (3,909 | ) | (4,554 | ) | (1,230 | ) | ||||||||||||
Net loss applicable to common stockholders | $ | (38,258 | ) | $ | (29,086 | ) | $ | (33,274 | ) | $ | (24,482 | ) | $ | (19,836 | ) | |||||
Basic and diluted net loss per common share (1) | $ | (1.54 | ) | $ | (3.11 | ) | $ | (59.08 | ) | $ | (51.49 | ) | $ | (42.78 | ) | |||||
Weighted average common shares outstanding (1) | 24,913,087 | 9,361,153 | 563,194 | 475,458 | 463,626 | |||||||||||||||
| (1) | The increase in weighted average common shares outstanding and the corresponding decrease in basic and diluted net loss per common share in 2005 and 2004 is primarily the result of the conversion of all the Company’s redeemable convertible preferred stock and the issuance of common shares both related to the Company’s initial public offering in July of 2004. |
56
Table of Contents
| As of December 31, | ||||||||||||||||||
| 2005 | 2004 | 2003 | 2002 | 2001 | ||||||||||||||
Consolidated Balance Sheet Data: | ||||||||||||||||||
Cash and cash equivalents | $ | 31,380 | $ | 32,707 | $ | 28,446 | $ | 12,221 | $ | 19,898 | ||||||||
Restricted cash | — | — | 9,500 | — | — | |||||||||||||
Short-term investments | 25,350 | 14,100 | — | — | — | |||||||||||||
Working capital | 47,052 | 42,028 | 25,364 | 9,658 | 15,723 | |||||||||||||
Total assets | 72,695 | 61,040 | 49,759 | 16,633 | 20,518 | |||||||||||||
Other long-term liabilities | 11,578 | 8,823 | 577 | — | — | |||||||||||||
Redeemable convertible preferred stock | — | — | 93,287 | 57,839 | 38,082 | |||||||||||||
Total stockholders’ equity (deficit) | 39,873 | 36,244 | (65,804 | ) | (45,581 | ) | (21,901 | ) | ||||||||||
| ITEM 7. | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes appearing at the end of this Report. Some of the information contained in this discussion and analysis or set forth elsewhere in this Report, including information with respect to our plans and strategy for our business and related financing, includes forward-looking statements that involve risks and uncertainties. You should review the “Risk Factors” section of this Report for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are a specialty pharmaceutical company with a focus on developing and marketing products for urology, sexual health and other indications within specialty markets. We currently have a sales and marketing organization of approximately 120 people. Our only marketed product, Testim®, is a proprietary, topical 1% testosterone once-a-day gel indicated for the treatment of hypogonadism. Hypogonadism is defined as reduced or absent secretion of testosterone which can lead to symptoms such as loss of libido, adverse changes in body composition, irritability and poor concentration. We reported revenues in 2005 of $42.8 million, an increase of 58.5% over the $27.0 million reported in 2004. According to National Prescription Audit data from IMS Health, Inc. or IMS, a pharmaceutical market research firm, Testim’s share of total prescriptions for the gel segment of the testosterone replacement therapy, or TRT, market was 16.3% for the month of December 2005, compared with 11.5% for the month of December 2004. For the full year 2005, Testim’s share of total prescriptions for the TRT market was 13.8%, versus 10.0% for the full year 2004.
We target diseases with unmet medical needs that we believe can be addressed by more innovative, effective or easy to use products. Our current product pipeline includes:
| • | Five projects in clinical development; |
| • | Two pain product candidates in pre-clinical development; and |
| • | Rights to six additional pain product candidates, options to additional indications for our injectable enzyme and other products for urology and sexual health. |
Our lead project is AA4500, an injectable enzyme. We have an option to license all non-topical uses of AA4500, and we currently are pursuing three indications: Dupuytren’s Disease, Peyronie’s Disease and Adhesive Capsulitis, or Frozen Shoulder Syndrome. We believe that AA4500 has completed Phase II of development for the treatment of Dupuytren’s Disease and is in Phase II of development for the treatment of Peyronie’s Disease and Frozen Shoulder Syndrome. Each of these disease states is characterized by patients suffering the debilitating effects and deformities of scar tissue. AA4500 was identified for in-licensing because of its application for the treatment of Peyronie’s Disease, which is a plaque or hard scar on the shaft of the penis that causes substantial distortion of the penis upon erection. Men suffering from Peyronie’s Disease are often unable to engage in sexual intercourse due to the deformity of their erection.
57
Table of Contents
In the process of pursuing AA4500 for the treatment of Peyronie’s Disease, we saw completed clinical work and an opportunity to pursue AA4500 for the treatment of Dupuytren’s Disease. Dupuytren’s Disease results when a patient has excess scar tissue that collects at the tendons in the hand. Typically it results in forming a “cord” affecting the ring and small finger, causing the finger to contract, leaving the patient unable to fully extend the affected finger(s).
AA4500 was identified as a potential treatment for Frozen Shoulder Syndrome by the licensor of AA4500. We exercised our option to expand our license for AA4500 to include its application for the treatment of Frozen Shoulder Syndrome based on the results of a Phase II clinical trial conducted by the licensor of AA4500 which suggests that local injection of AA4500 is well-tolerated and may be effective in patients suffering from Frozen Shoulder Syndrome. Frozen Shoulder Syndrome is a disease of diminished shoulder motion, characterized by restriction in both active and passive range of motion of the shoulder joint.
We have in-licensed a platform technology for the delivery of hormones and pharmaceutical products through the oral mucosal membrane. AA2600, is a TRT for the treatment of hypogonadism that utilizes this transmucosal film technology. We believe AA2600 has completed Phase II of development and expect to commence Phase III trials early in the second quarter of 2006.
We also have a project, which we refer to as AA4010, and referred to as AA2010 erroneously in a press release and during our earnings conference call on March 7, 2006, in Phase I of development for the treatment for overactive bladder using the same transmucosal film delivery system. Overactive bladder is a medical condition affecting both men and women characterized by urinary urgency and increased frequency of urination.
We have two pain products using our transmucosal film delivery system in pre-clinical development. We also have rights to six additional pain products and products for hormone replacement and urologic disease using our transmucosal film delivery system.
We have never been profitable and have incurred an accumulated deficit of $137.5 million as of December 31, 2005. We expect to incur significant operating losses for the foreseeable future. Commercialization expenses, development costs, and in-licensing milestone payments related to existing and new product candidates and to enhance our core technologies will continue to increase in the near term. In particular, we expect to incur increased costs for selling and marketing as a result of our co-promotion agreement with Oscient Pharmaceuticals Corp., or Oscient, and as we continue to market Testim, additional development and pre-commercialization costs for existing and new product opportunities, acquisition costs for new product opportunities and increased general and administration expense to support the infrastructure required to grow and operate as a public company. We will need to generate significant revenues to achieve profitability.
We expect that quarterly and annual results of operations will fluctuate for the foreseeable future due to several factors including the overall growth of the androgen market, the timing and extent of research and development efforts, milestone payments liability and the outcome and extent of clinical trial activities. Our limited operating history makes accurate prediction of future operating results difficult.
Net Revenues. To date, all of our net revenues have been generated by the sales and out-licensing of Testim. We expect Testim to continue to be the primary source of our net revenues. We sell Testim to pharmaceutical wholesalers and some chain drug stores. These companies have the right to return Testim. As a result, we recognize revenue based on prescription units dispensed to patients, since they are not subject to return. We will continue to recognize revenue upon prescription units dispensed until we have sufficient history to estimate product returns. When we are able to reasonably estimate our product returns, we will recognize a one-time increase in net revenue related to the recognition of revenue previously deferred, net of appropriate allowances for cash discounts, rebates, patient coupons and product returns. We continue to gather experience with our returns and believe that we will be in a position to reasonably estimate returns during 2006. We do not anticipate
58
Table of Contents
sales of Testim outside of the U.S., pursuant to our agreements with Bayer, Inc., or Bayer, and Ipsen Farmaceutica, B.V., or Ipsen, to have a material impact on our revenues or profitability. In addition to the up-front and milestone payments these agreements afford us, we believe they also benefit us by providing opportunities for manufacturing efficiencies through increased sales of Testim, as well as, making us an attractive partner to future licensors by providing us with global development and commercialization capabilities. We may seek to generate additional revenues from the expansion of Testim into new markets and the commercialization of new products.
We are a relatively new company and our sales prospects are uncertain. We expect our revenues to fluctuate due to:
| • | market acceptance and pricing for Testim, including any change in wholesaler purchasing patterns; |
| • | commercialization of and market acceptance of our future products, if any; |
| • | regulatory approvals and market acceptance of new and competing products, including generics; |
| • | government or private healthcare reimbursement policies, particularly with respect to Testim and our other product candidates; |
| • | promotional efforts of Solvay and its impact on sales; and |
| • | a switch to recognizing revenues at the point when sales are made to wholesalers. |
We use a third-party distributor, ICS, a division of Amerisource Bergen, for commercial distribution activities. The majority of our sales in the U.S. are to pharmaceutical wholesalers that, in turn, distribute product to chain and other retail pharmacies, hospitals, mail-order providers and other institutional customers. Approximately 91% of Testim purchases are from four customers: Cardinal Health, McKesson, Amerisource Bergen and Walgreens. Outside of the U.S., we currently rely on third parties to market, sell and distribute Testim and any of our product candidates for which we receive marketing approval from any foreign jurisdictions. For Canada, we have an agreement with Bayer to market and distribute Testim. We have entered into an agreement with Ipsen for the rest of the world, excluding the U.S., Canada, Japan and Mexico.
Cost of Goods Sold. All of our cost of goods sold currently relate to the sale of Testim. Cost of goods sold consists of:
| • | raw materials; |
| • | fees paid to our contract manufacturers and related costs; |
| • | royalty payments, which currently consist solely of payments due to Bentley; |
| • | personnel costs associated with quality assurance and manufacturing oversight; and |
| • | distribution costs, including warehousing, freight and product liability insurance. |
We do not anticipate any material changes in our gross margin rate in the U.S. We anticipate our gross margins for sales outside the U.S. to be significantly lower than those seen in the U.S. This is due to a combination of factors which include the terms of our distribution agreements with Bayer and Ipsen and the royalty payments due to our licensor.
Research and Development. Our research and development expenses consist of:
| • | salaries and expenses for our development personnel; |
| • | payments to consultants, investigators, contract research organizations and manufacturers in connection with our preclinical and clinical trials; |
59
Table of Contents
| • | costs of developing and obtaining regulatory approvals; and |
| • | product license and milestone fees paid prior to regulatory approval. |
Since our inception through December 31, 2005, we have spent $28.3 million on Testim, $15.6 million on the development of AA4500 for the treatment of Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome and $7.0 million on AA2600, our testosterone replacement film product candidate, and AA4010, our overactive bladder transmucosal film product candidate. These amounts represent our external research and development costs, including up-front and milestone payments, for these projects. We do not allocate salaries or administrative expenses to development projects. Due to the significant risks and uncertainties inherent in the clinical development and regulatory approval processes, the cost to complete projects in development cannot be reasonably estimated. Currently, Testim is our only marketed product. We believe AA4500 has completed Phase II of development for the treatment of Dupuytren’s Disease. AA4500 is in phase II of development for the treatment of Peyronie’s Disease and Frozen Shoulder Syndrome. We believe AA2600 has completed Phase II of development and AA4010 is in Phase I of development. Results from clinical trials may not be favorable. Further, data from clinical trials is subject to varying interpretation, and may be deemed insufficient by the regulatory bodies reviewing applications for marketing approvals. As such, clinical development and regulatory programs are subject to risks and changes that may significantly impact cost projections and timelines. We expect our research and development expenses for our current products to increase as a result of further development of AA4500 for Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome, and our testosterone replacement transmucosal film. In addition, the current regulatory and political environment at the U.S. Food and Drug Administration, or FDA, could lead to increased data requirements which could impact regulatory timelines and costs. We expect further significant increases in our expenditures to develop any other potential new product candidates that we would in-license or acquire. Expenditures will increase to develop any other product candidates that we in-license or acquire.
Selling, General and Administrative. Selling, general and administrative expenses consist primarily of salaries and other related costs for personnel, marketing and promotion costs, professional fees and facilities costs. We anticipate increases in selling, general and administrative expenses due to sales and marketing costs associated with Testim, hiring of additional personnel, our co-promotion agreement with Oscient, investor relations and other activities associated with operating as a publicly traded company. See a description of our co-promote agreement with Oscient set forth below under the heading“Contractual Commitments.”
Results of Operations
Years Ended December 31, 2005 and 2004
Net revenues. Net revenues were $42.8 million and $27.0 million for the years ended December 31, 2005 and 2004, respectively. Prescription units dispensed increased from approximately 203,900 in 2004 to approximately 303,500 in 2005. We believe that Testim prescription growth in 2005 over 2004 was attributed to increasing physician and patient awareness that Testim provides better patient outcomes, the shift in prescriptions away from the testosterone patch product to Testim, the continued focus of our sales force on the promotion of Testim to urologists, endocrinologists and select primary care physicians, and our co-promotion agreement with Oscient, which focuses on broadening our exposure to primary care physicians. The increase in net revenues in 2005 over 2004 also are a result of price increases having a cumulative impact of 11% offset by increases in wholesaler discounts due to increased distribution costs, product rebates as Testim became more widely prescribed by managed care providers and had experienced an increased utilization of patient coupons. Net revenues in 2005 also include $1.0 million of product shipped to Ipsen and $0.5 million of revenues related to up-front and milestone payments for which there are no comparable amounts in 2004. Net revenues for Testim product sales are reflected net of cash discounts, rebates and patient coupons. Shipments in the U.S. not recognized as revenue, net of anticipated cash discounts, were $2.2 million and $3.7 million at December 31, 2005 and 2004, respectively and are reflected in deferred revenue, current portion.
60
Table of Contents
Cost of goods sold.Cost of goods sold were $13.1 million and $8.1 million for the years ended December 31, 2005 and 2004, respectively. The increase in cost of goods sold in 2005 over 2004 was directly attributable to the increase in Testim prescription revenue in the U.S. and $0.9 million of costs associated with product shipped to Ipsen for which there is no comparable amount in 2004. Cost of goods sold in the 2004 period would have been approximately $0.4 million higher had purchases of a key raw material not been charged-off in 2002. Gross margin on our net revenues was 69.4% in 2005 compared to 69.9% in 2004. Gross margin for U.S. revenues was 70.4% in 2005 and would have been 68.4% in 2004 had purchases of a key raw material not been charged-off in 2002. The improvement in gross margin in 2005 over 2004 resulted primarily from the net effect of price increases offset by increases in wholesaler discounts, product rebates and increased utilization of patient coupons. In 2002, we purchased approximately $0.9 million of this raw material prior to FDA approval of Testim and, based on our policy, charged the purchase price to research and development expense because we would have had no alternative use for the raw material if Testim were not approved. As of December 31, 2004, we did not have any of the raw material with zero book value remaining in inventory.
Research and development expenses. Research and development expenses were $24.3 million and $16.0 million for the years ended December 31, 2005 and 2004, respectively. The increase in 2005 over 2004 was primarily due to $5.9 million increase in development work for AA4500, a $1.7 million increase in costs associated with Phase IV Clinical Trials for Testim, a $1.0 million increase in development work for AA2600, and the $0.5 million up-front payment for the in-license of eight separate pain products using the transmucosal film technology in February 2005. These increases were offset by a $1.5 million decrease in milestone payments for AA4500, our Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome product candidate.
Selling, general and administrative expenses. Selling, general and administrative expenses were $43.9 million and $31.2 million for the years ended December 31, 2005 and 2004, respectively. The increase in 2005 over 2004 was due primarily to selling and marketing expenses increasing by $9.8 million resulting from increases in our sales force made in late 2004, higher marketing costs associated with our co-promote agreement with Oscient and the Oscient co-promotion fees. We anticipate significant increases in selling and marketing expenses due to, among other things, our share of increased marketing costs associated with our co-promotion agreement with Oscient and increases in the Oscient co-promotion fees. General and administrative expenses increased by $2.9 million as a result of the investigation initiated by the Audit Committee of our Board of Directors into certain transactions and the effectiveness of our internal disclosure controls and procedures described in more detail in Item 9A of this Report, severance accruals related to the termination of our former President and Chief Operating Officer, an increase in recruiting costs, costs associated with moving to our new headquarters and incremental costs associated with operating as a publicly traded company.
Interest income (expense), net.Interest income (expense), net was $1.5 and $(0.2) million for the years ended 2005 and 2004, respectively. Net interest income in 2005 relates primarily to interest earned on the invested proceeds from our initial public offering in July of 2004 and our private placement in June of 2005. The expense in 2004 relates primarily to non-cash amortization of deferred debt issuance costs related to a line of credit and to interest paid on the line of credit, which was repaid and terminated in March 2004.
Other Income (expense), net. Other income (expense), net was $(1.2) million and $0 for the years ended December 31, 2005 and 2004, respectively. Other income (expense), net in 2005 relates to $0.2 million of placement agent fees and transaction costs associated with the June 30, 2005 private placement which were allocated to the financing-related liability and the non-cash change in the fair value of the financing-related liability of $1.0 million during 2005. On December 30, 2005, we reclassified the financing related liability of $7.2 million to permanent equity as a result of amendments to the Securities Purchase Agreements. We do not expect any future expense related to the financing-related liability.
Dividends and accretion of redeemable convertible preferred stock.Accretion of redeemable convertible preferred stock was $0 and $0.6 million for the years ended December 31, 2005 and 2004, respectively. In 2004,
61
Table of Contents
we were accreting the carrying value of redeemable convertible preferred stock to its redemption value. There is no corresponding accretion in 2005 as all previously outstanding shares of this stock were converted into our common stock in connection with our initial public offering.
At December 31, 2005, we had federal net operating loss carryforwards of approximately $116.6 million, which will expire in 2019 through 2025, if not utilized. In addition, we had state net operating loss carryforwards of approximately $96.0 million, of which $56.2 million relate to Pennsylvania. The Pennsylvania state net operating losses are subject to a $2.0 million annual limitation for 20 years and expire in 2009 through 2025. At December 31, 2005, we had federal research and development credits of approximately $1.7 million that will expire in 2020 through 2025, if not utilized.
The Internal Revenue Code provides for a limitation on the annual use of net operating loss and research and development tax credit carryforwards following certain ownership changes that could limit our ability to utilize these carryforwards. We may have experienced various ownership changes, as a result of past financings. Accordingly, our ability to utilize the aforementioned carryforwards may be limited. Additionally, U.S. tax laws limit the time during which these carryforwards may be applied against future taxes; therefore we may not be able to take full advantage of these carryforwards for federal income tax purposes
Years Ended December 31, 2004 and 2003
Net revenues.Net revenues were $27.0 million and $8.8 million for 2004 and 2003 , respectively. Net revenues consist solely of revenues resulting from approximately 203,900 and 70,300 prescription units dispensed in 2004 and 2003, respectively. We believe that the increase in net revenues and underlying prescription units is the result of many factors, including: 2004 was the first complete year of sales as we launched Testim in the first quarter of 2003; growth in the TRT gel market; growth in our market share and new prescriptions; increased effectiveness of our sales and marketing organization; positioning of Testim’s attributes versus the category leader; price increases and contribution of better formulary positions with managed care providers. While Testim continued to gain market share throughout 2004, the annual rate of growth in the overall gel segment slowed. The annual growth rate in total gel prescriptions was 11.8% in 2004 versus 34.4% in 2003. Net revenues for Testim product sales are reflected net of cash discounts, rebates and patient coupons. Shipments in the U.S. not recognized as revenue, net of anticipated cash discounts, were $3.7 million and $4.3 million at December 31, 2004 and 2003, respectively and are reflected in deferred revenue, current portion.
Cost of goods sold.Cost of goods sold were $8.1 million and $3.8 million for 2004 and 2003, respectively. The increase in cost of goods sold for 2004 over 2003 was directly attributable to the increase in Testim prescription revenue. Cost of goods sold in 2004 and 2003 would have been approximately $0.4 million and $0.3 million higher, respectively, had purchases of a key raw material not been charged-off in 2002. Gross margin on our net product sales was 69.9% in 2004 compared to 57.4% in 2003. The improvement in gross margin in 2004 over 2003 resulted primarily from fixed manufacturing costs spread across an increased number of units sold. Gross margin would have been 68.4% for 2004 and 54.5% for 2003 had purchases of a key raw material not been written-off in 2002. In 2002, we purchased approximately $0.9 million of this raw material prior to FDA approval of Testim and, based on our policy, charged the purchase price to research and development expense because we would have had no alternative use for the raw material if Testim were not approved. As of December 31, 2004, we did not have any of the raw material with zero book value remaining in inventory.
Research and development expense.Research and development expense was $16.0 million and $7.2 million for 2004 and 2003, respectively. The increase in 2004 over 2003 was due primarily to $5.0 million in up-front and milestone payments made to BioSpecifics Technologies Corp., or BioSpecifics, for the in-license of AA4500, our Dupuytren’s and Peyronie’s Diseases product candidate, and $0.5 million of additional development expenses for AA4500. In addition, the increase in 2004 over 2003 is a result of the $2.0 million increase in development expenses on our testosterone replacement and overactive bladder transmucosal film product candidates and $0.5 million increase in expenses associated with the Testim European regulatory filing and Testim clinical trials.
62
Table of Contents
Selling, general and administrative expenses.Selling, general and administrative expenses were $31.2 million and $27.5 million for 2004 and 2003, respectively. The increase in 2004 over 2003 was due primarily to a $2.4 million increase in general and administrative expenses and a $1.3 million increase in selling and marketing expenses. The increase in general and administrative expenses results from additional hiring of key personnel, relocation and recruitment expenses and incremental costs associated with operating as a publicly traded company. The increase in selling and marketing expenses results from compensation and related expenses for our professional sales organization being greater in 2004 versus 2003 due to higher headcount and incentive compensation related to sales volume achievements.
Interest income (expense), net.Interest income (expense), net was $(0.2) million and $(1.0) million for 2004 and 2003, respectively. The net expense in 2004 and 2003 relates primarily to non-cash amortization of deferred debt issuance costs related to a line of credit and to interest paid on the line of credit. The line of credit was entered into in May 2003 and repaid and terminated in March 2004. Although we arranged for a replacement line of credit with Comerica Bank, the weighted average borrowings on the replacement line in 2004 were lower than under the previously existing line in 2003 in an attempt to control interest costs. The debt issuance costs were the result of warrants issued to investors to guarantee the line of credit.
Other income.Other income for 2003 was $1.7 million due to a one-time gain from the settlement of a lawsuit with a vendor.
Dividends and accretion on redeemable convertible preferred stock.Dividends accrued and accretion on redeemable convertible preferred stock and dividends paid to warrant holders were $0.6 million and $4.4 million for 2004 and 2003, respectively. The decrease in 2004 compared to 2003 was due to the termination and cancellation of the accumulated dividend provisions of our preferred stock in connection with the private placement of shares of our series D preferred stock in October 2003. In April 2004, we removed a variable feature of our outstanding warrants to purchase series D preferred stock and fixed both the amount of warrants and the exercise price of the warrants. As a result, we recorded a $0.2 million deemed dividend in the second quarter of 2004 for the increase in the fair value of the warrants attributable to the amendments of the warrant terms. We anticipate no future dividends and accretion on redeemable convertible preferred stock as all previously outstanding shares of this stock were converted into our common stock in connection with our initial public offering.
Liquidity and Capital Resources
Since inception through December 31, 2005, we have financed our product development, operations and capital expenditures primarily from private and public sales of equity securities. Since inception through December 31, 2005, we received net proceeds of approximately $174.1 million from our initial public offering, private sales of securities and the exercise of stock options. We had approximately $31.4 million and $32.7 million in cash and cash equivalents as of December 31, 2005 and December 31, 2004, respectively. As of December 31, 2005 and December 31, 2004, we also had $25.4 million and $14.1 million, respectively, in short-term investments.
We believe that our current financial resources and sources of liquidity will be adequate to fund our anticipated operations until at least the end of 2007. We may, however, need to raise additional funds prior to this time in order to support our obligations under our existing or any new co-promotion agreement or to enhance our sales and marketing efforts for additional products we may acquire, commercialize any product candidates that receive regulatory approval, and acquire or in-license approved products or product candidates or technologies for development. Insufficient funds may cause us to delay, reduce the scope of, or eliminate one or more of our development, commercialization or expansion activities. Our future capital needs and the adequacy of our available funds will depend on many factors, including the effectiveness of our sales and marketing activities, the cost of clinical studies and other actions needed to obtain regulatory approval of our products in development, and the timing and cost of any acquisitions. If additional funds are required, we may raise such funds from time
63
Table of Contents
to time through public or private sales of equity or debt securities or from bank or other loans. Financing may not be available on acceptable terms, or at all, and our failure to raise capital when needed could materially adversely impact our growth plans and our financial condition or results of operations. Additional equity financing, if available, may be dilutive to the holders of our common stock and may involve significant cash payment obligations and covenants that restrict our ability to operate our business.
We have two equipment loans with General Electric Capital Corporation totaling $0.3 million outstanding at December 31, 2005. The equipment facilities bear interest at a weighted-average rate of 9.3% per annum.
Sources and Uses of Cash
Cash used in operations was $28.8 million, $16.8 million and $26.5 million for 2005, 2004 and 2003, respectively. Negative operating cash flows during 2005 were caused primarily by operating losses. Negative operating cash flows during 2004 result from operating losses offset primarily by $8.1 million of cash collections of up-front and milestone payments. Negative operating cash flows during 2003 were caused primarily by operating losses.
Cash used in investing activities was $12.5 million, $15.3 million and $1.1 million for 2005, 2004 and 2003, respectively. Cash used in investing activities for 2005 relates primarily to the purchase of short-term investments with part of the proceeds from our June 30, 2005 private placement. Cash used in investing activities for 2004 relates primarily to the purchase of short-term investments with part of the proceeds from our initial public offering. Cash used in investing activities for 2003 relates primarily to the purchase of manufacturing equipment, office furniture and computer equipment, and the build-out of a manufacturing line for Testim.
Cash provided by financing activities was $40.0 million, $36.4 million and $43.8 million in 2005, 2004 and 2003, respectively. Cash provided by financing activity in 2005 results primarily from the $39.0 million received from the June 30, 2005 private placement and the $0.7 million in cash receipts from stock option exercises. Cash provided by financing activities in 2004 was related primarily to the initial public offering of common stock. During 2003, we received $43.0 million from the private placement of shares of our series D preferred stock and an additional $10.4 million primarily from a line of credit offset by a $9.5 million certificate of deposit reflected as restricted cash to secure borrowing under a line of credit agreement.
Our actual cash needs might vary materially from those now planned because of a number of factors, including:
| • | Testim market acceptance and sales growth; |
| • | third-party payor coverage and reimbursement for Testim; |
| • | the cost of manufacturing, distributing, marketing and selling Testim; |
| • | the scope, rate of progress and cost of our product development activities; |
| • | future clinical trial results; |
| • | the terms and timing of any collaborative, licensing and other arrangements that we have or may establish; |
| • | the cost and timing of regulatory approvals; |
| • | the costs of commercializing any of our current product candidates or any other product candidates that we may acquire or in-license; |
| • | acquisition or in-licensing costs; |
| • | the effect of competing technological and market developments; |
64
Table of Contents
| • | changes to the regulatory approval process for product candidates in those jurisdictions, including the U.S., in which we may be seeking approval for our product candidates; |
| • | the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; and |
| • | the extent to which we acquire or invest in businesses and technologies, although we currently have no commitments or agreements relating to any of these types of transactions. |
We might seek additional debt or equity financing or both to fund our operations or product acquisitions. If we increase our debt levels, we might be restricted in our ability to raise additional capital and might be subject to financial and restrictive covenants. Our stockholders may experience dilution as a result of the issuance of additional equity securities. This dilution may be significant depending upon the amount of equity securities that we issue and the prices at which we issue any securities.
In 2005 and 2004, we received $3.8 million and $8.1 million, respectively, in payments from existing agreements for Testim in other territories. These receipts are the result of up-front and milestone payments primarily related to contract signings and regulatory approvals. In addition, we could receive as much as an additional $0.4 million in 2006 if certain regulatory approvals are received outside the U.S. We might also enter into additional arrangements with corporate partners that could provide us with additional funding in the form of equity, debt, licensing, milestone or royalty payments. We might not be able to enter into such arrangements or raise any additional funds on terms favorable to us or at all.
Contractual Commitments
We are involved with in-licensing of products which are generally associated with payments to the partner from whom we have licensed the product. Such payments frequently take the form of:
| • | an up-front payment, the size of which varies depending on the phase of the product and how many other companies would like to obtain the product, which is paid very soon after signing a license agreement; |
| • | milestone payments which are paid when certain parts of the overall development program are accomplished, or in some cases, when a patent issues; |
| • | payments upon certain regulatory events, such as the filing of an IND, and NDA, approval of an NDA, or the equivalents in other countries; and |
| • | payments for achievement of certain sales thresholds, such as a payment due the first year a product achieves $100 million in sales. |
We may also out-license products, for which we hold the rights, to other companies for commercialization in other territories, or at times, for other uses. When this happens, typically there is:
| • | an up-front payment made at or shortly after signing a partnering agreement; |
| • | milestone payments that may be made on completion of a phase of a clinical program, or regulatory approval in a given territory; and |
| • | a payment or payments made upon achievement of a certain level of sales in a given year. |
Bentley
In May 2000, we entered into a license agreement with Bentley Pharmaceuticals, Inc., or Bentley, for its patented transdermal gel technology. Under the agreement, as amended, we were granted an exclusive, worldwide, royalty-bearing license to make and sell products incorporating Bentley’s formulation technology that contain testosterone. We produce Testim under this agreement. The term of this agreement is determined on
65
Table of Contents
a country-by-country basis and extends until the later of patent right termination in a country or 10 years after the first sale of product in that country. In May 2001, we entered into a license with Bentley granting similar rights for a product containing another hormone, the term of which is perpetual. In May 2001, we also entered into a separate license granting similar rights for a product containing a therapeutic drug.
In addition to its royalty obligations, we are obligated to pay certain upfront payments and milestones under the license agreements with Bentley. Through December 31, 2005, we have made aggregate upfront and milestone payments under the three license agreements with Bentley of approximately $0.6 million. If all events under the Bentley license agreements occur, we would be obligated to pay a maximum of approximately $1.0 million in milestone payments. The timing of the above payments, if any, is uncertain.
BioSpecifics
In June 2004, we entered into a development and license agreement with BioSpecifics and amended such agreement in May 2005 and in December 2005 (the BioSpecifics Agreement). Under the BioSpecifics Agreement, as amended, we were granted exclusive worldwide rights to develop, market and sell certain products containing BioSpecifics’ enzyme, which we refer to as AA4500. Our licensed rights concern the development of products, other than dermal formulations labeled for topical administration. Currently, we have rights to develop, and are developing, AA4500 for the treatment of Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome. We may expand the BioSpecifics Agreement, at our option, to cover other indications as they are developed by us or BioSpecifics.
The BioSpecifics Agreement extends, on a country-by-country and product-by-product basis, for the longer of the patent life, the expiration of any regulatory exclusivity period or 12 years. We may terminate the BioSpecifics Agreement with 90 days written notice.
We are responsible, at our own cost and expense, for developing the formulation and finished dosage form of products containing AA4500 and arranging for the clinical supply of such products. As permitted under the BioSpecifics Agreement, we have qualified Cobra Biologics Ltd., or Cobra, as our primary supplier of clinical supply under the agreement. Accordingly, we will now rely on Cobra as our primary supplier of AA4500 for clinical trials.
BioSpecifics has the option, exercisable no later than six months after FDA approval of the first New Drug Application or Biologics License Application with respect to a product, to assume the right and obligation to supply, or arrange for the supply from a third party other than a back-up supplier qualified by us, of a specified portion of commercial product required by us. The agreement provides that we may withhold a specified amount of a milestone payment until (i) BioSpecifics executes an agreement, containing certain milestones, with a third party for the commercial manufacture of the product, (ii) BioSpecifics commences construction of a facility, compliant with current Good Manufacturing Practices, for the commercial supply of the product, or (iii) 30 days after BioSpecifics notifies us in writing that it will not exercise the supply option.
If BioSpecifics exercises the supply option, commencing on a specified date, BioSpecifics will be responsible for supplying either by itself or through a third party other than a back-up supplier qualified by us, a specified portion of the commercial supply of the product. If BioSpecifics does not exercise the supply option, we will be responsible for arranging for the entire commercial product supply. In the event BioSpecifics exercises the supply option, we and BioSpecifics are required to use commercially reasonable efforts to enter into a commercial supply agreement on customary and reasonable terms and conditions which are not worse than those with back-up suppliers qualified by us.
We must pay BioSpecifics on a country-by-country and product-by-product basis a specified percentage of net sales for products covered by the BioSpecifics Agreement. Such percentage may vary depending on whether BioSpecifics exercises the supply option. In addition, the percentage may be reduced if (i) BioSpecifics fails to
66
Table of Contents
supply commercial product supply in accordance with the terms of the BioSpecifics Agreement; (ii) market share of a competing product exceeds a specified threshold; or (iii) we are required to obtain a license from a third party in order to practice BioSpecifics’ patents without infringing such third party’s patent rights.
In addition to the payments set forth above, we must pay BioSpecifics an amount equal to a specified mark-up of the cost of goods sold for products sold by us that are not manufactured by or on behalf of BioSpecifics, provided that, in the event BioSpecifics exercises the supply option, no payment will be due for so long as BioSpecifics fails to supply the commercial supply of the product in accordance with the terms of the BioSpecifics Agreement.
Finally, we are obligated to make contingent milestone payments upon contract initiation, receipt of technical data, manufacturing process development, the one-year anniversary of the BioSpecifics Agreement, filing of regulatory applications and receipt of regulatory approval. Through December 31, 2004, we paid up-front and milestone payments under the BioSpecifics Agreement of $5.0 million. In June 2005, we paid the one-year contract anniversary payment of $3.0 million. In December 2005, we paid the option exercise fee of $0.5 million for the Frozen Shoulder Syndrome indication. We could make up to an additional $5.5 million of contingent milestone payments under the BioSpecifics Agreement if all existing conditions are met. Additional milestone obligations may be due if we exercise an option to develop and license AA4500 for additional medical indications.
Cobra
In July 2005 we entered into an agreement with Cobra for the process development, scale up and manufacture of AA4500 for Phase II/III clinical trials (the Cobra Agreement). Accordingly, we will now rely on Cobra as our primary supplier of AA4500 for clinical trials. The Cobra Agreement is divided into five non-sequential stages with specific objectives, timelines, down payments and total payments that attach to each stage. We may terminate the Cobra Agreement at the completion of each stage if the objective or the timeline for such stage is not met. Through December 31, 2005, we incurred a total of $2.1 million under the Cobra Agreement. If all remaining stages are completed under the current Cobra Agreement, then we will pay Cobra an additional $2.4 million.
Althea
In order to prepare AA4500 for clinical trials, AA4500 bulk substance produced by Cobra must be filled and lyophilized, placebo must be manufactured and a sterile diluent must be produced (collectively, “pharmaceutical products”). On February 28, 2006, we entered into a Manufacturing and Clinical Supply Agreement with Althea Technologies, Inc., or Althea, to be effective as of January 17, 2006 that sets out the general terms and conditions under which Althea will produce these pharmaceutical products in bulk or finished dosage form for our development and/or clinical use only. At the time we executed the agreement, we and Althea entered into a project plan that provides the parameters for the production of pharmaceutical products. Pursuant to the project plan we will supply bulk substance of AA4500 produced by Cobra to Althea which, in turn, will produce and deliver to us the pharmaceutical products. The agreement provides that we will pay Althea 50% of the total fees due to Althea upon signing of the project plan. Then, Althea will invoice us monthly for services completed during such month, and the balance of the fees will be invoiced upon delivery of the pharmaceutical products. Upon signing of the project plan in February 2006, we paid to Althea $174,000, and we expect to incur an additional $174,000 under this project plan in 2006.
PharmaForm
In June 2003, we expanded our drug delivery technology portfolio by entering into a license agreement with Formulation Technologies, L.L.C., d/b/a/ PharmaForm, or PharmaForm. Under this agreement, as amended, we were granted an exclusive, worldwide, royalty-bearing license to develop, make and sell products that contain
67
Table of Contents
hormones or that are used to treat urologic disorders incorporating PharmaForm’s oral transmucosal film technology for which there is an issued patent in the U.S. The term of this license agreement is for the life of the licensed patents.
In February 2005, we entered into an additional license agreement with PharmaForm. Under this agreement, we were granted exclusive, worldwide royalty-bearing rights to develop, manufacture and market eight compounds using PharmaForm’s proprietary transmucosal film technology for the management of pain, including acute and chronic pain. The compounds that may be developed include opioids as well other types of analgesics. In 2005, we paid an up-front payment of $0.5 million and could make up to an additional $21.2 million in contingent milestone payments if all underlying events for these eight products occur. In addition, we will have on-going royalty payment obligations to PharmaForm. The timing of the remaining payments, if any, is uncertain.
To speed the development of products using the licensed technology from PharmaForm, we entered into a research and development agreement with PharmaForm on a fee for service basis. We will be the sole owner of any intellectual property rights developed in connection with this agreement. We incurred $2.3 million under the research and development agreement in 2005 and expect to incur $3.1 million in 2006.
Oscient
In April 2005, we entered into a co-promotion agreement (the Oscient Agreement) with Oscient whereby we and Oscient will co-promote Testim in the U.S. Oscient was granted the exclusive right to promote Testim jointly with us to primary care physicians by means of its sales force. Oscient commenced co-promotion of Testim in May 2005. The initial term of the Oscient Agreement ends on April 30, 2007. Oscient may extend the Oscient Agreement for two consecutive two-year periods provided that Oscient has met certain milestones for each extension. If these milestones are met and Oscient does not elect to terminate the Oscient Agreement, the first extension period will commence on January 1, 2007 and end on December 31, 2008 and the second extension period will commence effective January 1, 2009 and end on April 30, 2011.
We and Oscient utilize a joint marketing and promotion plan which sets forth the responsibilities of both parties with respect to the marketing and promotion of Testim in the U.S. primary care physician market. We and Oscient share equally these promotional expenses. Each party will be responsible for the costs associated with its own sales force. The 2005 combined spending for marketing and promotion of Testim in the primary care physician market in the U.S. was $7.1 million, and we estimate that the total costs for 2006 will be approximately $11.0 million to $13.0 million. These costs will be shared equally.
Oscient will be compensated for its services based on a specified percentage of the gross profit from Testim sales attributable to primary care physicians in the U.S. that exceed a specified sales threshold (the Oscient co-promotion fees). All gross profit earned on sales to primary care physicians below the predetermined threshold and all gross profit earned on all sales to non primary care physicians are not subject to the terms of the Oscient Agreement. Once sales to primary care physicians have exceeded the predetermined sales threshold, the gross profit earned on such sales is available to be split between us and Oscient. Once all agreed upon promotional expenses are appropriately reimbursed to each party, the gross profit remaining will be divided according to agreed upon percentages. The specific percentages are based upon Testim sales levels above the threshold attributable to primary care physicians actually achieved. For the year ended December 31, 2005, we recorded $3.5 million of expense for the Oscient co-promotion fees.
The agreed upon promotional expenses for each year are reimbursable equally and simultaneously to each company through the profit split mechanism, except in 2005 when Oscient will be disproportionately reimbursed its share of the promotional expenses through the profit split mechanism. As the agreed upon percentage of the gross profit available to be split in 2005 was insufficient to reimburse Oscient its share of the promotional expenses, that portion not reimbursed to Oscient will be carried over to 2006 and will be reimbursed through the 2006 profit split mechanism. Of the $3.5 million expense recorded by us for the year ended December 31, 2005, $0.6 million was due to this carry over reimbursement.
68
Table of Contents
The Oscient Agreement may be terminated by either party upon the occurrence of certain termination events. We may be obligated to make termination payments in certain instances. Also, Oscient has been granted the exclusive option to co-promote any future product candidate of ours that treats hypogonadism and contains testosterone as the active ingredient.
We will invoice and record in its financial statements all sales of Testim, including those sales to primary care physicians that exceed the specified threshold, as well as the gross profit earned on all sales. We will record our share of promotional expenses incurred and the Oscient co-promotion fees as “selling, general & administration” operating expenses in our consolidated statement of operations.
The following summarizes our contractual commitments as of December 31, 2005 (in thousands):
| Total | Less than 1 year | 2-3 years | 4-5 years | 5+ years | |||||||||||
Long-term debt | $ | 327 | $ | 260 | $ | 62 | $ | 5 | $ | — | |||||
Operating leases | 3,232 | 485 | 938 | 909 | 900 | ||||||||||
Purchase commitment (1) | 950 | 190 | 380 | 380 | — | ||||||||||
| $ | 4,509 | $ | 935 | $ | 1,380 | $ | 1,294 | $ | 900 | ||||||
| (1) | Amount represents a minimum annual manufacturing fee of $0.2 million. |
The contractual commitments reflected in this table exclude $27.7 million of contingent milestone payments that we may be obligated to pay in the future. We do not expect to pay any of these contingent milestone payments through December 31, 2006. These remaining milestones relate primarily to manufacturing process development, filing of regulatory applications and receipt of regulatory approval. The above table also excludes future royalty payments and wholesaler distribution fees because they are contingent on product sales. In addition, the above table excludes any co-promotional expenses and fees that may be due Oscient because they are contingent upon combined promotional expenditures and future product sales to primary care physicians, respectively.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements as defined in Item 303(a)(4) of Regulation S-K.
Critical Accounting Policies and Significant Judgments and Estimates
Our management’s discussion and analysis of our financial condition and results of operations, which are based on our consolidated financial statements, have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements as well as the reported revenues and expenses during the reporting periods. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. We are subject to uncertainties that may cause actual results to differ from these estimates, such as changes in the healthcare environment, competition, legislation and regulation. We believe the following accounting policies, which have been discussed with our audit committee, are the most critical because they involve the most significant judgments and estimates used in the preparation of our consolidated financial statements:
| • | revenue recognition; |
| • | inventory valuation; and |
| • | estimating the value of our equity instruments for use in stock-based compensation calculations. |
69
Table of Contents
Revenue Recognition. We sell Testim to pharmaceutical wholesalers and chain drug stores. These companies have the right to return Testim for any reason up to one-year after product expiration. As a result of Testim’s launch in the first quarter of 2003, we do not have sufficient sales history to estimate product returns. In accordance with Statement of Financial Accounting Standards (SFAS) No. 48, we defer recognition of revenue on product shipments of Testim to our U.S. customers until such time as Testim units are dispensed through patient prescriptions, since they are not subject to return. Under SFAS No. 48, we cannot recognize revenue on product shipments until we can reasonably estimate returns relating to these shipments. We estimate the volume of prescription units dispensed at pharmacies based on data provided by external, independent sources. These sources poll pharmacies, hospitals, mail order and other retail outlets for Testim prescriptions and project this sample on a national level. We will continue to recognize revenue based on prescription units until we have sufficient history to estimate product returns. When we are able to reasonably estimate our product returns, we will recognize a one-time increase in net revenue related to the recognition of revenue previously deferred, net of appropriate allowances for cash discounts, rebates, patient coupons and product returns. We continue to gather experience with our returns and believe that we will be in a position to reasonably estimate returns during 2006. In addition, we must make estimates for rebates provided to third-party payers and coupons provided to patients. Testim is covered by Medicaid and numerous independent insurance and pharmacy benefit managers, or PBMs. Some of these programs have negotiated rebates. We obtain estimates of prescription units dispensed under the various rebate programs from the same external sources discussed above. We make estimates of the rebates based on the external-source reports of volumes and actual contract terms. In addition, we provide coupons to physicians for use with prescriptions as promotional incentives. We utilize a contract service provider to process and pay claims to patients for actual coupon usage. As Testim becomes more widely used and as we continue to add managed care and PBM contracts, our estimates may result in differences to actual results. To date, our estimates have not resulted in any material adjustments to our operating results.
For revenues associated with licensing agreements, we use revenue recognition criteria outlined in Staff Accounting Bulletin (SAB) No. 104 and Emerging Issues Task Force (EITF) Issue No. 00-21,Revenue Arrangements with Multiple Deliverables. Accordingly, revenues from licensing agreements are recognized based on the performance requirements of the agreement. We entered into two license and distribution agreements to market and distribute Testim worldwide, excluding the U.S., Mexico and Japan. Under these agreements, the distributor and the licensee are each required to purchase Testim from us and make up-front, milestone and royalty payments. Under these agreements, the distributor and the licensee may each cancel the agreements if there is a breach of contract or if any party files for bankruptcy. Only milestone payments for product supply price reductions are refundable if we subsequently increase the supply price; otherwise, no up-front or milestone payments are refundable in any instance. Product shipments are subject to return and refund only if the product does not comply with technical specifications or if any of the agreements are terminated due to our nonperformance. We are obligated under the agreements to provide multiple deliverables; the license/product distribution rights, regulatory filing services and commercial product supply. Under EITF No. 00-21 the deliverables under each of these agreements are treated as single units of accounting as we do not have evidence to support that the consideration for the undelivered item, Testim units to be shipped, is at fair value. Revenue for Testim units shipped will be recognized upon product shipment, net of estimated product returns. We have deferred revenue recognition for non-refundable up-front and milestone payments until at least the first product shipment under each agreement.
We previously stated that non-refundable up-front and milestone revenue will be recognized as revenue as a percentage of Testim units shipped in the period over total Testim units expected to be shipped over the distribution term and that no such revenue will be recognized if a reasonable estimate of total future Testim unit shipments cannot be made. However, we came to the conclusion in the third quarter of 2005 that this accounting method was impractical to implement due to the uncertainty that would be created in estimating potential future sales in a large number of countries outside of the U.S. over an extended period of time.
Accordingly, commencing in the third quarter of 2005, non-refundable up-front and milestone revenue will be recognized as revenue on a straight line basis. The amortization period for the agreement with Ipsen is based
70
Table of Contents
upon contractual terms and currently estimated to be 17 years. We have started to amortize milestones received over this period since the first product has been shipped. When earned by us in future periods, additional milestone payments achieved will be amortized over the remaining period. Refundable milestone payments will be deferred and only included in the revenue recognition equation with non-refundable milestone consideration when the refund right effectively lapses. No product shipments have been made to Bayer and, therefore, no revenue relating to upfront or milestone payments have been recognized.
Through December 31, 2005 we collected $11.9 million of license payments under these agreements. Of this amount $10.3 million relates to non-refundable milestones from Ipsen. The remaining $1.6 million consists of a $1.0 million refundable milestone from Ipsen and $0.6 million of milestones from Bayer that cannot be recognized at this time. During the year ended December 31, 2005, we recorded $0.5 million of revenue for up-front and milestone amounts received from Ipsen. At December 31, 2005, $0.7 million was classified as current deferred revenue and $10.7 million was classified as long-term deferred revenue.
Inventory Valuation. We rely on a single source supplier for one of the key ingredients in Testim. As a result, we previously purchased a substantial safety stock of this ingredient on which we have limited shelf-life data. We continue to perform stability testing on the ingredient to determine its maximum shelf-life. In addition, our finished goods inventory has a definitive shelf-life. We estimate that the demand for Testim will be such that the raw materials and finished goods on hand will ultimately be used in future production and sold to our customers. As a result, we have not recorded a material valuation allowance for potential excess inventory as of December 31, 2005. We will continually evaluate the demand for Testim and the data from the stability testing to determine whether an allowance for excess inventory is warranted. In addition, at December 31, 2005 we have capitalized as work-in-process inventory $1.1 million of Testim manufactured by a back-up supplier. We intend to sell this inventory after we receive approval of the alternate testing site and the test method from the FDA. We capitalized this inventory based on favorable preliminary results from analytical testing, past product experience and our belief that it is probable that the inventory will be saleable. If the FDA does not approve the alternate testing site and test method, we will have to write-off this inventory.
Valuation of Equity Instruments used in Stock-Based Compensation. We account for employee stock options using the intrinsic-value method in accordance with Accounting Principles Board (APB) Opinion No. 25,Accounting for Stock Issued to Employees, and related interpretations and have adopted the disclosure-only provisions of SFAS No. 148,Accounting for Stock-Based Compensation-Transition and Disclosure, an amendment of SFAS No. 123,Accounting of Stock-Based Compensation. The disclosure-only provisions of SFAS No. 148 require us to disclose pro forma net loss applicable to common stockholders and net loss per common share as if we accounted for employee stock options at fair value. SFAS No. 123 also requires us to estimate the value of purchase rights granted to employees under our 2004 Employee Stock Purchase Plan for inclusion in the SFAS No. 148 pro forma disclosure. In addition, we account for stock-based compensation to non-employees using the fair-value method in accordance with SFAS No. 123 and EITF Issue No. 96-18,Accounting for Equity Instruments That Are Issued to Other Than Employees for Acquiring, or in conjunction with Selling, Goods or Services. Our disclosure under SFAS No. 148 and our accounting under EITF Issue No. 96-18 require us to estimate the value of equity instruments issued. We utilize the Black-Scholes option-pricing model to value stock options issued to employees and non-employees and purchase rights granted to employees under our 2004 Employee Stock Purchase Plan. The Black-Scholes option-pricing model requires us to make assumptions of the estimated life of the underlying options, the anticipated volatility of the price of our common stock, the risk-free interest rate and the expected dividend yield of our common stock.
71
Table of Contents
Recent Accounting Pronouncements
In December 2004, the Financial Accounting Standards Board, or FASB, issued SFAS No. 123R,Share-Based Payment. SFAS No. 123R revises SFAS No. 123 and supersedes APB Opinion No. 25. Generally the approach in SFAS No. 123R is similar to the approach described in SFAS No. 123. SFAS No. 123R requires that all share-based payments to employees, including grants of employee stock options, be recognized in the statement of operations based on their grant-date fair values over the period during which employees are required to provide services in exchange for the award. In addition, under SFAS No. 123R our 2004 Employee Stock Purchase Plan will be considered compensatory. Pro forma disclosure of stock-based compensation under fair value methods is no longer an alternative. SFAS No. 123R provides guidance on how to determine the grant-date fair value for awards of equity instruments as well as alternative methods of adopting its requirements. On April 14, 2005, the Securities and Exchange Commission delayed the effective date of SFAS No. 123R to the beginning of the first fiscal year after June 15, 2005. As a result, we anticipate adopting SFAS No. 123R on January 1, 2006. As we currently account for employee stock-based compensation under the intrinsic value method permitted under APB Opinion No. 25, the adoption of SFAS No. 123R will result in significant additional compensation charges in our statements of operations, although it will have no impact on our overall financial position. On September 20, 2005, our Compensation Committee of the Board of Directors approved the acceleration of the vesting of the unvested portion of “out-of-the-money” stock options held by current employees. The decision to accelerate the vesting of these “out-of-the-money” options was made to avoid recognizing compensation expense in future periods upon the adoption of SFAS No. 123R. (See Note 2(o) of the Consolidated Financial Statements)
In May 2005, the FASB issued SFAS No. 154, “Accounting Changes and Error Corrections-a replacement of APB Opinion No. 20 and FASB Statement No. 3” (SFAS No. 154). This Statement replaces APB Opinion No. 20, “Accounting Changes” and FASB Statement No. 3, “Reporting Accounting Changes in Interim Financial Statements,” and changes the requirements for the accounting for and the reporting of a change in accounting principle. SFAS No. 154 applies to all voluntary changes in an accounting principle. It establishes, unless impractical, retrospective application as the required method for reporting a voluntary change in accounting principle. It also applies to changes required by an accounting pronouncement in the unusual instance that the pronouncement does not include specific transition provisions. When a pronouncement includes specific transition provisions, those provisions should be followed. SFAS No. 154 is effective for accounting changes and error corrections occurring in fiscal years beginning after December 15, 2005.
72
Table of Contents
| ITEM 7A. | Quantitative and Qualitative Disclosures About Market Risk |
We do not use derivative financial instruments or derivative commodity instruments for trading purposes. Our financial instruments consist of cash, cash equivalents, short-term investments, trade accounts receivable, accounts payable and long-term obligations. We consider investments that, when purchased, have a remaining maturity of 90 days or less to be cash equivalents.
We invest in marketable securities in accordance with our investment policy. The primary objectives of our investment policy are to preserve principal, maintain proper liquidity to meet operating needs and maximize yields. Our investment policy specifies credit quality standards for our investments. The maximum allowable duration of a single issue is three months. Our investment policy permits us to invest in Auction Rate Securities (ARS) with maturities of greater than three months, provided that the interest reset date, the date in which the ARS may be readily liquidated to a market maker, for the ARS is less than three months. As the underlying maturity date of our ARS investments is greater than three months, and as we classify these investments as available for sale, we classify our ARS investments as short-term investments on our consolidated balance sheet.
Our investment portfolio is subject to interest rate risk and will fall in value in the event market interest rates increase. All our cash, cash equivalents and short-term investments at December 31, 2005, amounting to approximately $56.7 million, were maintained in bank demand accounts, money market accounts, U.S. government backed securities and ARS. We do not hedge our interest rate risks as we believe reasonably possible near-term changes in interest rates would not materially affect our results of operations, financial position or cash flows.
Transactions relating to Auxilium UK, Limited are recorded in pounds sterling. Upon consolidation of this subsidiary into our consolidated financial statements, we translate the balance sheet asset and liability accounts to the U.S. dollar based on exchange rates as of the balance sheet date; balance sheet equity accounts are translated into the U.S. dollar at historical exchange rates; and all statements of operations and cash flows amounts are translated into the U.S. dollar at the average exchange rates for the period. Exchange gains or losses resulting from the translation are included as a separate component of stockholders’ equity. In addition, we conduct clinical trials in the Netherlands, exposing us to cost increases if the U.S. dollar declines in value compared to the euro. We do not hedge our foreign exchange risks as we believe reasonably possible near-term fluctuations of exchange rates would not materially affect our results of operations, financial position or cash flows.
73
Table of Contents
| ITEM 8. | Financial Statements and Supplementary Data |
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
74
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders
Auxilium Pharmaceuticals, Inc.:
In our opinion, the accompanying consolidated balance sheet, and the related consolidated statements of operations, stockholders’ equity and comprehensive loss, and cash flows present fairly, in all material respects, the financial position of Auxilium Pharmaceuticals, Inc. and its subsidiaries at December 31, 2005, and the results of their operations and their cash flows for the year ended December 31, 2005, in conformity with U.S. generally accepted accounting principles. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audit. We conducted our audit of these statements in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, and evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
/s/ PricewaterhouseCoopers LLP
Philadelphia, Pennsylvania
March 14, 2006
75
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Stockholders
Auxilium Pharmaceuticals, Inc.:
We have audited the accompanying consolidated balance sheet of Auxilium Pharmaceuticals, Inc. and subsidiaries as of December 31, 2004, and the related consolidated statements of operations, redeemable convertible preferred stock, stockholders’ equity (deficit) and comprehensive loss, and cash flows for each of the years in the two-year period ended December 31, 2004. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Auxilium Pharmaceuticals, Inc. and subsidiaries as of December 31, 2004, and the results of their operations and their cash flows for each of the years in the two-year period ended December 31, 2004, in conformity with U.S. generally accepted accounting principles.
/s/ KPMG LLP
Philadelphia, Pennsylvania
April 15, 2005
76
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Balance Sheets
(In thousands, except share and per share amounts)
| December 31, | ||||||||
| 2005 | 2004 | |||||||
| Assets | ||||||||
Current assets: | ||||||||
Cash and cash equivalents | $ | 31,380 | $ | 32,707 | ||||
Short-term investments | 25,350 | 14,100 | ||||||
Accounts receivable, trade | 4,614 | 4,586 | ||||||
Accounts receivable, other | 200 | 400 | ||||||
Inventories | 4,387 | 4,014 | ||||||
Prepaid expenses and other current assets | 2,365 | 2,194 | ||||||
Total current assets | 68,296 | 58,001 | ||||||
Property and equipment, net | 3,690 | 2,554 | ||||||
Other assets | 709 | 485 | ||||||
Total assets | $ | 72,695 | $ | 61,040 | ||||
| Liabilities and Stockholders’ Equity | ||||||||
Current liabilities: | ||||||||
Notes payable, current portion | $ | 260 | $ | 427 | ||||
Accounts payable | 5,109 | 2,868 | ||||||
Accrued expenses | 12,841 | 8,644 | ||||||
Deferred revenue, current portion | 2,911 | 4,034 | ||||||
Deferred rent, current portion | 123 | — | ||||||
Total current liabilities | 21,244 | 15,973 | ||||||
Notes payable, long-term portion | 65 | 323 | ||||||
Deferred revenue, long-term portion | 10,671 | 8,500 | ||||||
Deferred rent, long-term portion | 842 | — | ||||||
Commitments and contingencies (Note 9) | ||||||||
Stockholders’ equity: | ||||||||
Preferred stock, $0.01 par value per share, 5,000,000 shares authorized, no shares issued or outstanding | — | — | ||||||
Common stock, $0.01 par value per share; authorized 120,000,000 at December 31, 2005 and 2004; issued and outstanding 29,241,482 and 20,648,988 shares at December 31, 2005 and 2004, respectively | 292 | 206 | ||||||
Additional paid-in capital | 177,429 | 135,856 | ||||||
Accumulated deficit | (137,515 | ) | (99,257 | ) | ||||
Deferred compensation | (437 | ) | (565 | ) | ||||
Notes receivable from stockholders | — | (17 | ) | |||||
Accumulated other comprehensive income | 104 | 21 | ||||||
Total stockholders’ equity | 39,873 | 36,244 | ||||||
Total liabilities and stockholders’ equity | $ | 72,695 | $ | 61,040 | ||||
See accompanying notes to consolidated financial statements.
77
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Statements of Operations
(In thousands, except share and per share amounts)
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Net revenues | $ | 42,804 | $ | 27,025 | $ | 8,822 | ||||||
Operating expenses: | ||||||||||||
Cost of goods sold | 13,119 | 8,148 | 3,762 | |||||||||
Research and development | 24,301 | 15,993 | 7,175 | |||||||||
Selling, general, and administrative | 43,935 | 31,210 | 27,475 | |||||||||
Total operating expenses | 81,355 | 55,351 | 38,412 | |||||||||
Loss from operations | (38,551 | ) | (28,326 | ) | (29,590 | ) | ||||||
Interest income | 1,553 | 424 | 73 | |||||||||
Interest expense | (57 | ) | (616 | ) | (1,080 | ) | ||||||
Other income (expense), net | (1,203 | ) | — | 1,722 | ||||||||
Net loss | (38,258 | ) | (28,518 | ) | (28,875 | ) | ||||||
Accretion of redeemable convertible preferred stock | — | (395 | ) | (490 | ) | |||||||
Deemed dividend to warrant holders | — | (173 | ) | — | ||||||||
Dividends accrued on redeemable convertible preferred stock | — | — | (3,909 | ) | ||||||||
Net loss applicable to common stockholders | $ | (38,258 | ) | $ | (29,086 | ) | $ | (33,274 | ) | |||
Basic and diluted net loss per common share | $ | (1.54 | ) | $ | (3.11 | ) | $ | (59.08 | ) | |||
Weighted average common shares outstanding | 24,913,087 | 9,361,153 | 563,194 | |||||||||
See accompanying notes to consolidated financial statements.
78
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Statements of Stockholders’ Equity and Comprehensive Loss
Year ended December 31, 2005
(In thousands, except share amounts)
| Common stock | Additional capital | Accumulated deficit | Deferred compensation | Notes stockholders | Accumulated income | Total Equity | ||||||||||||||||||||||||
| Shares | Amount | |||||||||||||||||||||||||||||
Balance, January 1, 2005 | 20,648,988 | $ | 206 | $ | 135,856 | $ | (99,257 | ) | $ | (565 | ) | $ | (17 | ) | $ | 21 | $ | 36,244 | ||||||||||||
Net loss | — | — | — | (38,258 | ) | — | — | — | (38,258 | ) | ||||||||||||||||||||
Foreign currency translation adjustment | — | — | — | — | — | — | — | — | ||||||||||||||||||||||
Unrealized gain on available for sale securities | — | — | — | — | — | — | 83 | 83 | ||||||||||||||||||||||
Total comprehensive loss | (38,258 | ) | 83 | (38,175 | ) | |||||||||||||||||||||||||
Private placement, net of transaction costs | 8,242,796 | 83 | 32,955 | — | — | — | — | 33,038 | ||||||||||||||||||||||
Issuance of private placement warrants | — | — | 7,151 | — | — | — | — | 7,151 | ||||||||||||||||||||||
Exercise of common stock options | 210,687 | 2 | 738 | — | — | — | — | 740 | ||||||||||||||||||||||
Employee Stock Purchase Plan purchases | 125,127 | 1 | 501 | — | — | — | — | 502 | ||||||||||||||||||||||
Issuance of restricted stock | 25,000 | — | 120 | — | (120 | ) | — | — | — | |||||||||||||||||||||
Cancellation of restricted stock | (11,116 | ) | — | (83 | ) | — | 83 | — | — | — | ||||||||||||||||||||
Amortization of deferred compensation | — | — | — | — | 165 | — | — | 165 | ||||||||||||||||||||||
Compensation on stock option grants to nonemployees for services provided | — | — | 84 | — | — | — | — | 84 | ||||||||||||||||||||||
Compensation on stock option grants | — | — | 107 | — | — | — | — | 107 | ||||||||||||||||||||||
Interest income accrued on notes receivable | — | — | — | — | — | (1 | ) | — | (1 | ) | ||||||||||||||||||||
Repayment of notes receivable | — | — | — | — | — | 18 | — | 18 | ||||||||||||||||||||||
Balance, December 31, 2005 | $ | 29,241,482 | $ | 292 | $ | 177,429 | $ | (137,515 | ) | $ | (437 | ) | $ | — | $ | 104 | $ | 39,873 | ||||||||||||
See accompanying notes to consolidated financial statements.
79
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Statements of Redeemable Convertible Preferred Stock, Stockholders’ Equity (Deficit) and Comprehensive Loss
Year ended December 31, 2004
(In thousands, except share amounts)
| Stockholders’ equity (deficit) | |||||||||||||||||||||||||||||||||||||
| Redeemable convertible preferred stock | Common stock | Additional capital | Accumulated deficit | Deferred compensation | Notes stockholders | Accumulated income (loss) | Total | ||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||||
Balance, January 1, 2004 | 70,212,517 | $ | 93,287 | 935,071 | $ | 9 | $ | 5,121 | $ | (70,739 | ) | $ | (57 | ) | $ | (107 | ) | $ | (31 | ) | $ | (65,804 | ) | ||||||||||||||
Comprehensive loss: | |||||||||||||||||||||||||||||||||||||
Net loss | — | — | — | — | — | (28,518 | ) | — | — | — | (28,518 | ) | |||||||||||||||||||||||||
Foreign currency translation adjustment | — | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
Unrealized gain on available for sale cash equivalents | — | — | — | — | — | — | — | — | 52 | 52 | |||||||||||||||||||||||||||
Total comprehensive loss | (28,518 | ) | 52 | (28,466 | ) | ||||||||||||||||||||||||||||||||
Accretion of preferred stock | — | 395 | — | — | (395 | ) | — | — | — | — | (395 | ) | |||||||||||||||||||||||||
Exercise of preferred warrants | 80,000 | 100 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||
Conversion of preferred to common | (70,292,517 | ) | (93,782 | ) | 14,058,482 | 141 | 93,641 | — | — | — | — | 93,782 | |||||||||||||||||||||||||
Initial public offering, net of transaction costs | — | — | 5,500,000 | 55 | 36,456 | — | — | — | — | 36,511 | |||||||||||||||||||||||||||
Restricted stock issued | — | — | 93,437 | 1 | 700 | — | (701 | ) | — | — | — | ||||||||||||||||||||||||||
Cancellation of restricted stock | — | — | (5,382 | ) | — | (41 | ) | — | 41 | — | — | — | |||||||||||||||||||||||||
Amortization of deferred compensation | — | — | — | — | — | — | 152 | — | — | 152 | |||||||||||||||||||||||||||
Exercise of common stock options | — | — | 53,112 | — | 232 | — | — | — | — | 232 | |||||||||||||||||||||||||||
Employee Stock Purchase Plan purchases | — | — | 14,268 | — | 91 | — | — | — | — | 91 | |||||||||||||||||||||||||||
Compensation on stock option grants to nonemployees for services provided | — | — | — | — | 51 | — | — | — | — | 51 | |||||||||||||||||||||||||||
Receipt of payment on notes receivable | — | — | — | — | — | — | — | 93 | — | 93 | |||||||||||||||||||||||||||
Interest income accrued on notes receivable | — | — | — | — | — | — | — | (3 | ) | — | (3 | ) | |||||||||||||||||||||||||
Balance, December 31, 2004 | — | $ | — | 20,648,988 | $ | 206 | $ | 135,856 | $ | (99,257 | ) | $ | (565 | ) | $ | (17 | ) | $ | 21 | $ | 36,244 | ||||||||||||||||
See accompanying notes to consolidated financial statements.
80
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Statements of Redeemable Convertible Preferred Stock, Stockholders’ Deficit and Comprehensive Loss
Year ended December 31, 2003
(In thousands, except share amounts)
| Stockholders’ deficit | |||||||||||||||||||||||||||||||||||
| Redeemable convertible preferred stock | Common stock | Additional capital | Accumulated deficit | Deferred compensation | Notes stockholders | Accumulated loss | Total | ||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | ||||||||||||||||||||||||||||||||
Balance, January 1, 2003 | 41,460,152 | $ | 57,839 | 497,925 | $ | 5 | $ | — | $ | (45,383 | ) | $ | (73 | ) | $ | (101 | ) | $ | (29 | ) | $ | (45,581 | ) | ||||||||||||
Comprehensive loss: | |||||||||||||||||||||||||||||||||||
Net loss | — | — | — | — | — | (28,875 | ) | — | — | — | (28,875 | ) | |||||||||||||||||||||||
Foreign currency translation adjustment | — | — | — | — | — | — | — | — | (2 | ) | (2 | ) | |||||||||||||||||||||||
Total comprehensive loss | (28,875 | ) | (2 | ) | (28,877 | ) | |||||||||||||||||||||||||||||
Issuance of Series D redeemable convertible preferred stock and warrants, net of expenses | 28,752,365 | 40,969 | — | — | 2,032 | — | — | — | — | 2,032 | |||||||||||||||||||||||||
Accretion of preferred stock | — | 490 | — | — | (490 | ) | — | — | — | — | (490 | ) | |||||||||||||||||||||||
Accrual of preferred dividends | — | 3,909 | — | — | (3,909 | ) | — | — | — | — | (3,909 | ) | |||||||||||||||||||||||
Recapitalization in connection with Series D transaction | — | (9,920 | ) | 423,796 | 4 | 6,397 | 3,519 | — | — | — | 9,920 | ||||||||||||||||||||||||
Issuance of warrants for debt guarantee | — | — | — | — | 1,008 | — | — | — | — | 1,008 | |||||||||||||||||||||||||
Exercise of common stock options | — | — | 12,175 | — | 54 | — | — | — | — | 54 | |||||||||||||||||||||||||
Compensation on stock and stock option grants to nonemployees for services provided | — | — | 1,175 | — | 11 | — | — | — | — | 11 | |||||||||||||||||||||||||
Compensation on stock option vesting acceleration to directors | — | — | — | — | 18 | — | — | — | — | 18 | |||||||||||||||||||||||||
Amortization of deferred compensation | — | — | — | — | — | — | 16 | — | — | 16 | |||||||||||||||||||||||||
Interest income accrued on notes receivable | — | — | — | — | — | — | — | (6 | ) | — | (6 | ) | |||||||||||||||||||||||
Balance, December 31, 2003 | 70,212,517 | $ | 93,287 | 935,071 | $ | 9 | $ | 5,121 | $ | (70,739 | ) | $ | (57 | ) | $ | (107 | ) | $ | (31 | ) | $ | (65,804 | ) | ||||||||||||
See accompanying notes to consolidated financial statements.
81
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
Consolidated Statements of Cash Flows
(In thousands)
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Cash flows from operating activities: | ||||||||||||
Net loss | $ | (38,258 | ) | $ | (28,518 | ) | $ | (28,875 | ) | |||
Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
Depreciation and amortization | 1,087 | 1,283 | 1,275 | |||||||||
Stock-based compensation | 356 | 203 | 45 | |||||||||
Interest income accrued on notes receivable from stockholders | — | (3 | ) | (6 | ) | |||||||
Change in fair value of financing related liability | 990 | — | — | |||||||||
Unrealized gain on available for sale cash equivalents | 83 | 52 | — | |||||||||
Changes in operating assets and liabilities: | ||||||||||||
Accounts receivable | 172 | (3,211 | ) | (1,775 | ) | |||||||
Inventories | (373 | ) | 1,363 | (4,098 | ) | |||||||
Prepaid expenses and other current assets | (602 | ) | (211 | ) | (1,015 | ) | ||||||
Accounts payable and accrued expenses | 6,474 | 3,952 | 3,645 | |||||||||
Deferred revenue | 1,048 | 8,248 | 4,286 | |||||||||
Deferred rent | 192 | — | — | |||||||||
Net cash used in operating activities | (28,831 | ) | (16,842 | ) | (26,518 | ) | ||||||
Cash flows from investing activities: | ||||||||||||
Purchases of property and equipment | (1,246 | ) | (1,193 | ) | (582 | ) | ||||||
Purchases of short-term investments | (47,275 | ) | (32,100 | ) | — | |||||||
Redemptions of short-term investments | 36,025 | 18,000 | — | |||||||||
Purchases of other assets | — | (4 | ) | (508 | ) | |||||||
Net cash used in investing activities | (12,496 | ) | (15,297 | ) | (1,090 | ) | ||||||
Cash flows from financing activities: | ||||||||||||
Proceeds from common shares and warrants issued in private placement, net of transaction costs | 39,198 | — | — | |||||||||
Proceeds from Initial Public Offering, net of transaction costs | — | 36,511 | — | |||||||||
Proceeds from issuance of Series D preferred stock and warrants, net of expenses | — | — | 43,001 | |||||||||
Net borrowings (repayments) under lines of credit, net of transaction costs | — | (9,548 | ) | 9,342 | ||||||||
Movement in restricted cash to secure borrowings | — | 9,500 | (9,500 | ) | ||||||||
Proceeds from Series B warrants exercised | — | 100 | — | |||||||||
Proceeds from debt financings, net of transaction costs | — | 171 | 1,091 | |||||||||
Payments on debt financings | (427 | ) | (768 | ) | (166 | ) | ||||||
Employee Stock Purchase Plan purchases | 502 | 91 | — | |||||||||
Proceeds from exercise of common stock options | 740 | 232 | 54 | |||||||||
Collection of notes receivable from stockholders | 18 | 93 | — | |||||||||
Net cash provided by financing activities | 40,031 | 36,382 | 43,822 | |||||||||
Effect of exchange rate changes on cash | (31 | ) | 18 | 11 | ||||||||
Increase (decrease) in cash and cash equivalents | (1,327 | ) | 4,261 | 16,225 | ||||||||
Cash and cash equivalents, beginning of period | 32,707 | 28,446 | 12,221 | |||||||||
Cash and cash equivalents, end of period | $ | 31,380 | $ | 32,707 | $ | 28,446 | ||||||
Supplemental cash flow information: | ||||||||||||
Cash paid for interest | $ | 51 | $ | 254 | $ | 259 | ||||||
See accompanying notes to consolidated financial statements.
82
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(1) Organization and Description of Business
(a) The Company
Auxilium Pharmaceuticals, Inc. along with its subsidiaries, or the Company, is a specialty pharmaceutical company with a focus on developing and marketing products for urology, sexual health and other indications within specialty markets. The Company has one marketed product, Testim®, a proprietary, topical 1% testosterone gel indicated for the treatment of hypogonadism. The Company launched Testim in the first quarter of 2003. The Company markets Testim in the U.S. through its own sales force and through a co-promotion agreement with Oscient Pharmaceuticals Corp., or Oscient, signed in April 2005 (see Note 9d). Testim was launched by Auxilium’s licensee, Ipsen Farmaceutica, B.V., or Ipsen, in Belgium, Denmark, Finland, Germany, Ireland, Netherlands, Spain, Sweden, and the United Kingdom in 2005.
In the fourth quarter of 2002, the Company received approval from the U.S. Food and Drug Administration, or FDA, to market Testim. Testim is approved for marketing in Belgium, Denmark, Finland, Germany, Greece, Iceland, Ireland, Italy, Luxembourg, The Netherlands, Norway, Portugal, Spain, Sweden and the United Kingdom. In 2004, the Company filed for regulatory approval to market Testim in Canada. The Company has responded to questions raised by the Canadian authorities as part of the approval process. Upon receipt of regulatory approvals, the Company will rely on Ipsen and other third parties to market, sell and distribute Testim outside of the U.S.
(b) Liquidity
On June 30, 2005, the Company completed a private placement in which the Company issued 8,242,796 shares of common stock and warrants to purchase an aggregate of 2,060,687 shares of common stock with an exercise price of $5.84 per share resulting in gross proceeds to the Company of approximately $40,400,000. In connection with the private placement, the Company paid $1,242,000 in placement agent fees and incurred approximately $175,000 in other transaction costs. The net proceeds to the Company were approximately $38,983,000.
The Company commenced operations in the fourth quarter of 1999. Since inception, it has incurred losses and negative cash flows from operations. The Company has been dependent upon external financing, including primarily private and public sales of securities, to fund operations. As of December 31, 2005, the Company had an accumulated deficit of approximately $137,515,000, and expects to incur additional operating losses.
The Company may require additional financing in the future to execute its intended business strategy. There can be no assurances that the Company will be able to obtain additional debt or equity financing on terms acceptable to the Company, when and if needed. Failure to raise needed funds on satisfactory terms could have a material impact on the Company’s business, operating results or financial condition.
(2) Summary of Significant Accounting Policies
(a)Principles of Consolidation
The accompanying consolidated financial statements include the accounts of Auxilium Pharmaceuticals, Inc. and its wholly owned subsidiaries. All intercompany balances and transactions have been eliminated in consolidation.
(b)Use of Estimates
The preparation of financial statements in conformity with U.S. generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, and disclosure of contingencies at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from these estimates.
83
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(c) Translation of Foreign Financial Statements
The Company established a foreign subsidiary in the United Kingdom in 2000, which uses the pound sterling as its functional currency. Assets and liabilities of the Company’s foreign subsidiary are translated at the year-end rate of exchange. The statements of operations for this subsidiary are translated at the average rate of exchange for the year. Gains or losses from translating foreign currency financial statements are accumulated in other comprehensive loss in stockholders’ equity (deficit).
(d) Fair Value of Financial Instruments
Management believes that the carrying amounts of the Company’s financial instruments, including cash, cash equivalents, restricted cash, short-term investments, accounts receivable, accounts payable, accrued expenses and notes payable approximate fair value due to the short-term nature of those instruments.
(e)Revenue Recognition
The Company sells Testim to wholesalers and chain drug stores, who have the right to return purchased product. In accordance with Statement of Financial Accounting Standards (SFAS) No. 48,Revenue Recognition When Right of Return Exists, the Company defers recognition of revenue on product shipments of Testim to its customers until Testim units are dispensed through patient prescriptions. Under SFAS No. 48, the Company cannot recognize revenue on product shipments until it can reasonably estimate returns relating to these shipments. The Company estimates prescription units dispensed based on distribution channel data provided by external, independent sources. Testim dispensed to patients through prescription is not subject to return. The gross sales price of product shipments in the U.S. was $49,009,000, $30,863,000 and $14,121,000 for the years ended December 31, 2005, 2004 and 2003, respectively. Gross revenue for prescription units dispensed was $50,398,000, $30,279,000 and $9,740,000 for the years ended December 31, 2005, 2004 and 2003, respectively, while product revenue net of cash discounts, rebates and patient coupons was $41,306,000, $27,025,000 and $8,822,000 for the years ended December 31, 2005, 2004 and 2003, respectively. Product shipments in the U.S. not recognized as revenue, net of anticipated cash discounts, were $2,217,000 and $3,723,000 at December 31, 2005 and 2004, respectively and are reflected in deferred revenue, current portion. The cost of Testim units shipped to customers that has not been recognized as revenue in accordance with the Company’s policy is reflected as inventory subject to return (see Note 4). The Company will continue to recognize revenue upon prescription units dispensed until it can reasonably estimate product returns based on its product returns experience. At this time, the Company will record a one-time increase in net revenue related to the recognition of revenue previously deferred.
The following individual customers each accounted for at least 10% of total product shipments for any of the respective periods:
| Years Ended December 31, | |||||||||
| 2005 | 2004 | 2003 | |||||||
Customer A | 31.3 | % | 25.7 | % | 21.5 | % | |||
Customer B | 31.2 | % | 28.6 | % | 32.8 | % | |||
Customer C | 18.7 | % | 21.0 | % | 17.1 | % | |||
Customer D | 9.9 | % | 12.0 | % | 9.6 | % | |||
| 91.1 | % | 87.3 | % | 81.0 | % | ||||
84
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
For revenues associated with licensing agreements, the Company uses revenue recognition criteria outlined in Staff Accounting Bulletin (SAB) No. 104 and Emerging Issues Task Force (EITF) Issue No. 00-21,Revenue Arrangements with Multiple Deliverables. Accordingly, revenues from licensing agreements are recognized based on the performance requirements of the agreement. The Company entered into two license and distribution agreements to market and distribute Testim worldwide, excluding the U.S., Mexico and Japan (see Note 14). Under these agreements, the distributor and the licensee are each required to purchase Testim from the Company and make up-front, milestone and royalty payments. Under these agreements, the distributor and the licensee may each cancel the agreements if there is a breach of contract or if any party files for bankruptcy. Only milestone payments for product supply price reductions are refundable if the Company subsequently increases the supply price; otherwise, no up-front or milestone payments are refundable in any instance. Product shipments are subject to return and refund only if the product does not comply with technical specifications or if any of the agreements are terminated due to the Company’s nonperformance. The Company is obligated under the agreements to provide multiple deliverables; the license/product distribution rights, regulatory filing services and commercial product supply. Under EITF No. 00-21 the deliverables under each of these agreements are treated as single units of accounting as the Company does not have evidence to support that the consideration for the undelivered item, Testim units to be shipped, is at fair value. Revenue for Testim units shipped will be recognized upon product shipment, net of estimated product returns. The Company has deferred revenue recognition for non-refundable up-front and milestone payments until at least the first product shipment under each agreement.
The Company previously stated that non-refundable up-front and milestone revenue will be recognized as revenue as a percentage of Testim units shipped in the period over total Testim units expected to be shipped over the distribution term and that no such revenue will be recognized if a reasonable estimate of total future Testim unit shipments cannot be made. However, the Company came to the conclusion in the third quarter of 2005 that this accounting method was impractical to implement due to the uncertainty that would be created in estimating potential future sales in a large number of countries outside of the U.S. over an extended period of time.
Accordingly, commencing in the third quarter of 2005, non-refundable up-front and milestone revenue will be recognized as revenue on a straight line basis. The amortization period for the agreement with Ipsen is based upon contractual terms and currently estimated to be 17 years. The Company has started to amortize milestones received over this period since the first product has been shipped. When earned by the Company in future periods, additional milestone payments achieved will be amortized over the remaining period. Refundable milestone payments will be deferred and only included in the revenue recognition equation with non-refundable milestone consideration when the refund right effectively lapses. No product shipments have been made to Bayer, Inc. (Bayer), and, therefore, no revenue relating to upfront or milestone payments has been recognized.
In December 2004, the Company shipped its first product totaling $311,000 to Ipsen for ultimate sale in the United Kingdom and Germany. The Company deferred recognition of this revenue at December 31, 2004 as the right of return had not lapsed until 2005. The first license payments under these arrangements totaling $8,100,000 were received in 2004. As of December 31, 2004, deferred revenue from licensing agreements of $8,500,000 was classified as long-term as the Company was not in a position to reasonably estimate total product demand under these agreements. The remaining milestone payments due at December 31, 2004 under these agreements of $400,000 is included in accounts receivable, other on the accompanying consolidated balance sheet.
Through December 31, 2005 the Company collected $11,900,000 of license payments under these agreements. Of this amount $10,300,000 relates to non-refundable milestones from Ipsen. The remaining $1,600,000 consists of a $1,000,000 refundable milestone from Ipsen and $600,000 of milestones from Bayer that cannot be recognized at this time. During the year ended December 31, 2005, the Company recorded $535,000 of revenue for up-front and milestone amounts received from Ipsen. At December 31, 2005, $694,000 was classified as current deferred revenue and $10,671,000 was classified as long-term deferred revenue.
85
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(f) Cash, Cash Equivalents and Short-term Investments
Cash, cash equivalents and short-term investments are stated at market value. Cash equivalents include only securities having a maturity of three months or less at the time of purchase. The Company limits its credit risk associated with cash, cash equivalents and short-term investments by placing its investments with banks it believes are highly creditworthy and with highly rated money market funds, U.S. government securities, or short-term commercial paper. The Company considers its investments, including securities with maturities in excess of one year, as “available for sale” under SFAS No. 115,Accounting for Certain Investments in Debt and Equity Securities, and accordingly classifies them as current, as management can sell these investments at any time at their option. Related unrealized gains and losses are recorded as a component of accumulated other comprehensive income (loss) in the equity section on the accompanying consolidated balance sheet.
(g)Supplemental Non-cash Disclosure
The following table represents the non-cash financing activity for the periods presented (in thousands):
| Years Ended December 31, | ||||||||
| 2005 | 2004 | 2003 | ||||||
Conversion of redeemable convertible preferred stock into common stock | — | $ | 93,782 | $ | — | |||
Grant of restricted stock to employees, net of cancellations | 37 | 660 | — | |||||
Grant of stock options | 107 | — | — | |||||
Grant of stock options to non employees | 84 | — | — | |||||
Accretion of preferred stock redemption value | — | 395 | 490 | |||||
Accrual of preferred stock dividends | — | — | 3,909 | |||||
Deemed dividend to warrant holders | — | 173 | — | |||||
Removal of preferred stock dividend feature | — | — | 9,920 | |||||
Issuance of warrants in connection with debt | — | — | 1,008 | |||||
Issuance of note payable for payment of insurance premiums | — | 421 | — | |||||
See Notes 8 and 11c for additional information on non-cash financing activity.
(h) Accounts Receivable
Accounts receivable, trade consist of amounts due from wholesalers and chain drug stores for the purchase of Testim. Ongoing credit evaluations of customers are performed and collateral is generally not required.
Accounts receivable, trade are net of allowances for cash discounts, actual returns and bad debts of $452,000 and $578,000 at December 31, 2005 and 2004, respectively.
The following individual customers each accounted for at least 10% of accounts receivable, trade on either of the respective dates:
| December 31, | ||||||
| 2005 | 2004 | |||||
Customer A | 44.3 | % | 43.2 | % | ||
Customer B | 30.9 | % | 26.4 | % | ||
Customer C | 17.7 | % | 12.0 | % | ||
Customer D | 1.1 | % | 10.6 | % | ||
| 94.0 | % | 92.2 | % | |||
See Note 2e regarding accounts receivable, other.
86
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(i) Inventories
Inventories are stated at the lower of cost or market, as determined using the first-in, first-out method.
(j) Concentration of Supply
The Company has a single source of supply for pentadecalactone and only two sources of supply for testosterone. In addition, a single source contract manufacturer produces Testim. The Company attempts to mitigate the risk of supply interruption by maintaining adequate safety stock of raw materials and by scheduling production runs to create safety stock of finished goods. The Company evaluates secondary sources of supply for all its raw materials and finished goods. The Company does not have any long-term minimum commitments for finished goods production or raw materials (see Note 6).
(k)Property and Equipment
Property and equipment are recorded at cost. Maintenance and repairs are charged to expense as incurred, and costs of improvements are capitalized. Depreciation is recognized using the straight-line method based on the estimated useful life of the related assets. Amortization of leasehold improvements is recognized using the straight-line method based on the shorter of the estimated useful life of the related assets or the lease term.
(l) Long-Lived Assets
The Company reviews long-lived assets, such as property and equipment, and purchased intangibles subject to amortization, for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable, in accordance with SFAS No. 144,Accounting for the Impairment or Disposal of Long-Lived Assets. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the asset. As of December 31, 2005, management believes that there is no impairment of long-lived assets. Also as of December 31, 2005 the Company has not revised the remaining useful lives of any significant assets.
Included in other assets is the unamortized balance of a license agreement milestone payment. This payment is being amortized over the 67-month estimated useful life of the related product which is consistent with the remaining lives of the underlying patents.
(m)Research and Development Costs
Research and development costs include salaries and related expenses for development personnel as well as fees and costs paid to external service providers. Costs of external service providers include both clinical trial costs and the costs associated with non-clinical support activities such as toxicology testing, manufacturing process development and regulatory affairs. External service providers include contract research organizations, contract manufacturers, toxicology laboratories, physician investigators and academic collaborators. Research and development costs, including the cost of product licenses prior to regulatory approval, are charged to expense as incurred.
(n) Income Taxes
Income taxes are accounted for under the asset-and-liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying
87
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
amounts of existing assets and liabilities and their respective tax bases and operating loss and tax credit carryforwards. Valuation allowances are established when necessary to reduce deferred tax assets to the amount expected to be realized. Deferred tax assets and liabilities are measured at the balance sheet date using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in the period such tax rate changes are enacted.
(o) Stock-Based Compensation
In December 2002, the Financial Accounting Standards Board (FASB) issued SFAS No. 148,Accounting for Stock-Based Compensation-Transition and Disclosure. This standard amends the transition and disclosure requirements of SFAS No. 123,Accounting for Stock-Based Compensation. As permitted by SFAS No. 148, the Company follows Accounting Principles Board (APB) Opinion No. 25,Accounting for Stock Issued to Employees, and related interpretations in accounting for equity-based awards issued to employees and directors for director services. Under APB Opinion No. 25, if the exercise price of the award equals or exceeds the fair value of the underlying stock on the measurement date, no compensation expense is recognized. The measurement date is the date on which the final number of shares and exercise price are known and is generally the grant date for awards to employees and directors. If the exercise price of the award is below the fair value of the underlying stock on the measurement date, then compensation cost is recorded, using the intrinsic-value method, and is recognized in the consolidated statements of operations over the vesting period of the award. In addition, the Company’s 2004 Employee Stock Purchase Plan is considered noncompensatory under APB Opinion No. 25 as it is a qualified plan under the Internal Revenue Code.
Had compensation cost for employee and director service option grants and purchase rights under the Company’s 2004 Employee Stock Purchase Plan been determined based on the fair value at the grant dates for those options and rights or the minimum-value method as discussed below, consistent with SFAS No. 123, net loss and net loss per common share would have increased to the pro forma amounts indicated below (in thousands, except per share amounts):
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Net loss applicable to common stockholders, as reported | $ | (38,258 | ) | $ | (29,086 | ) | $ | (33,274 | ) | |||
Add: total stock based employee compensation expense recognized under the intrinsic value based method | 272 | 152 | 34 | |||||||||
Deduct: total stock based employee compensation expense determined under fair value based method | (3,685 | ) | (1,191 | ) | (143 | ) | ||||||
Pro forma net loss applicable to common stockhoders | $ | (41,671 | ) | $ | (30,125 | ) | $ | (33,383 | ) | |||
Net loss per common share: | ||||||||||||
Basic and diluted, as reported | $ | (1.54 | ) | $ | (3.11 | ) | $ | (59.08 | ) | |||
Basic and diluted, pro forma | $ | (1.67 | ) | $ | (3.22 | ) | $ | (59.27 | ) | |||
On September 20, 2005, the Compensation Committee of the Board of Directors of the Company approved the acceleration of the vesting of the unvested portion of “out-of-the-money” stock options held by current employees, including executive officers. A stock option was considered “out-of-the-money” if the option exercise price was greater than $6.00 per share. The closing price of the Company’s common stock on September 20, 2005 was $5.69. All other terms and conditions of these “out-of-the-money” options remained unchanged.
88
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
As a result of the acceleration, options to purchase 523,795 shares of the Company’s common stock became exercisable immediately. The accelerated options range in exercise price from $7.50 to $12.50 and the weighted average exercise price of the accelerated options was $9.64 per share.
The decision to accelerate the vesting of these “out-of-the-money” options was made to avoid recognizing compensation expense in future financial statements upon the adoption of Statement of Financial Accounting Standards No. 123R (revised 2004), “Share-Based Payment” (“SFAS No. 123R), which, when effective, will require all share-based payments to employees, including unvested portions of previous grants of employee stock options, to be recognized in the Company’s financial statements based on their fair values. SFAS No. 123R will become effective as to the Company beginning January 1, 2006. The accelerated vesting of these “out-of-the-money” options does not result in a charge in the Company’s statement of operations in the current period based on U.S. generally accepted accounting principles. The Company recorded approximately $1.9 million of compensation cost applicable to these “out-of-the-money” options in the pro forma SFAS No. 123 table, shown above, for the year ended December 31, 2005.
Prior to the initial public offering (“IPO”), the value of option grants under SFAS No. 123 was determined using the minimum-value method. The minimum-value method considers the expected life of an option, the risk-free interest rate prevalent at the date of grant and the expected dividend yield. Upon completion of the IPO, the Company is required to include a volatility factor in its estimation of fair value. The Company has elected to utilize the Black-Scholes option-pricing model to estimate fair value of option grants under SFAS No. 123. Information with respect to the value of option grants under the minimum-value method and the Black-Scholes option pricing model for the periods presented is summarized as follows:
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Weighted average grant date value: | ||||||||||||
Grants below fair value | $ | — | $ | — | $ | — | ||||||
Grants at fair value | 5.01 | 5.02 | 0.95 | |||||||||
Grants above fair value | — | — | — | |||||||||
Weighted average assumptions: | ||||||||||||
Expected life of options (in years) | 5.5 | 6.8 | 7.0 | |||||||||
Risk-free interest rate | 4.02 | % | 3.91 | % | 3.57 | % | ||||||
Expected volatility | 80.00 | % | 59.49 | % | 0.00 | % | ||||||
Expected dividend yield | 0.00 | % | 0.00 | % | 0.00 | % | ||||||
Because the determination of the value of all options granted before the Company became a publicly traded entity do not include an expected volatility factor in addition to the factors described above and because additional option grants are expected in the future, the pro forma disclosures above are not representative of the pro forma effects of stock-based compensation on reported net operating results in future years (also see Note 2t). The Company utilized an expected volatility rate of 80% to value all option grants in 2004 after the IPO. The weighted average disclosed in the table above assumes a volatility of zero for all option grants prior to the IPO.
Included in the pro forma net loss applicable to common stockholders in 2005 and 2004 above is $335,000 and $277,000, respectively, of compensation under SFAS No. 123 related to the 2004 Employee Stock Purchase Plan. The fair value of employee purchase rights in 2005 was estimated utilizing the Black-Scholes option pricing model with the following weighted average assumptions: expected life of 0.7 years, risk-free interest rate of 3.63%, expected volatility of 87.56% and no expected dividend yield. The weighed average fair value of purchase rights granted in 2005 was $1.33 per share. The fair value of employee purchase rights in 2004 was
89
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
estimated utilizing the Black-Scholes option pricing model with the following weighted average assumptions: expected life of 1.3 years, risk-free interest rate of 2.25%, expected volatility of 80.63% and no expected dividend yield. The weighed average fair value of purchase rights granted in 2004 was $3.41 per share.
The Company accounts for stock-based compensation to non-employees using the fair-value method in accordance with SFAS No. 123 and EITF Issue No. 96-18,Accounting for Equity Instruments That Are Issued to Other Than Employees for Acquiring, or in Conjunction with Selling, Goods or Services. For the years ended December 31, 2005, 2004 and 2003, the Company granted a total of 51,500, 1,500 and 2,000 stock options, respectively, to consultants. In addition, in 2003 the Company issued a stock grant of 1,175 shares of common stock to a consultant. The options and stock granted generally vested immediately. For the years ended December 31, 2005, 2004, and 2003, the Company recorded stock-based compensation expense of $84,000, $51,000 and $11,000, respectively, related to stock and stock option grants to non-employees. The Company valued the stock option grants using the Black-Scholes option-pricing model utilizing the same assumptions above except that the expected life for each grant was the contractual term and the expected volatility for each grant was 80% in 2005, 70% in 2004 and 60% in 2003.
(p)Comprehensive Income (Loss)
The Company follows SFAS No. 130,Reporting Comprehensive Income. SFAS No. 130 established standards for the reporting and presentation of comprehensive income and its components in financial statements. The Company’s comprehensive loss consists of net loss, foreign currency translation adjustments and unrealized gains on available for sale securities. The Company’s comprehensive loss is presented within the accompanying consolidated statements of stockholders’ equity (deficit). For the years ended December 31, 2005, 2004 and 2003, the Company had foreign currency translation adjustments of $0, $0 and $2,000, respectively, relating to its subsidiary in the United Kingdom. Unrealized gains on available for sale securities were $83,000 and $52,000 for the years ended December 31, 2005 and 2004, respectively.
(q) Net Loss Per Common Share
Net loss per common share is calculated in accordance with SFAS No. 128,Earnings Per Share and SAB No. 98. Under the provisions of SFAS No. 128 and SAB No. 98, basic net loss per common share is computed by dividing the net loss applicable to common stockholders for the period by the weighted average number of common shares outstanding during the period reduced, where applicable, for unvested outstanding restricted shares.
90
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
The following table sets forth the computation of basic and diluted net loss per common share (in thousands, except share and per share amounts):
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Numerator: | ||||||||||||
Net loss | $ | (38,258 | ) | $ | (28,518 | ) | $ | (28,875 | ) | |||
Accretion of redeemable convertible preferred stock | — | (395 | ) | (490 | ) | |||||||
Deemed dividend to warrant holders | — | (173 | ) | — | ||||||||
Dividends accrued on redeemable convertible preferred stock | — | — | (3,909 | ) | ||||||||
Net loss applicable to common stockholders | (38,258 | ) | (29,086 | ) | (33,274 | ) | ||||||
Denominator: | ||||||||||||
Weighted-average common shares outstanding | 24,997,747 | 9,402,776 | 571,715 | |||||||||
Weighted-average unvested restricted common shares subject to forfeiture | (84,660 | ) | (41,623 | ) | (8,521 | ) | ||||||
Shares used in calculating net loss applicable to common stockholders per share | 24,913,087 | 9,361,153 | 563,194 | |||||||||
Basic and diluted net loss per common share | $ | (1.54 | ) | $ | (3.11 | ) | $ | (59.08 | ) | |||
Diluted net loss per common share is computed giving effect to all dilutive potential common stock, including options, warrants and redeemable convertible preferred stock. Diluted net loss per common share for all periods presented is the same as basic net loss per common share because the potential common stock is anti-dilutive. Anti-dilutive common shares not included in diluted net loss per common share are summarized as follows:
| December 31, | ||||||
| 2005 | 2004 | 2003 | ||||
Common stock options | 2,101,498 | 1,858,700 | 977,420 | |||
Warrants | 3,793,363 | 1,732,676 | 1,748,676 | |||
Restricted common stock | 81,451 | 88,055 | 7,250 | |||
Redeemable convertible preferred stock | — | — | 14,042,482 | |||
| 5,976,312 | 3,679,431 | 16,775,828 | ||||
(r) Segment Information
The Company is managed and operated as one business. The entire business is managed by a single management team that reports to the chief executive officer. The Company does not operate separate lines of business or separate business entities with respect to any of its product candidates. Accordingly, the Company does not prepare discrete financial information with respect to separate product areas and does not have separately reportable segments as defined by SFAS No. 131,Disclosures about Segments of an Enterprise and Related Information.
(s) Advertising Costs
Advertising costs, included in selling, general and administrative expenses, are charged to expense as incurred. Advertising expenses for the years ended December 31, 2005, 2004 and 2003 were $461,000, $368,000 and $908,000, respectively.
91
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(t) New Accounting Pronouncements
In December 2004, the Financial Accounting Standards Board, or FASB, issued SFAS No. 123R, Share-Based Payment. SFAS No. 123R revises SFAS No. 123 and supersedes APB Opinion No. 25. Generally the approach in SFAS No. 123R is similar to the approach described in SFAS No. 123. SFAS No. 123R requires that all share-based payments to employees, including grants of employee stock options, be recognized in the statement of operations based on their grant-date fair values over the period during which employees are required to provide services in exchange for the award. In addition, under SFAS No. 123R, the Company’s 2004 Employee Stock Purchase Plan will be considered compensatory. Pro forma disclosure of stock-based compensation under fair value methods is no longer an alternative. SFAS No. 123R provides guidance on how to determine the grant-date fair value for awards of equity instruments as well as alternative methods of adopting its requirements. On April 14, 2005, the SEC delayed the effective date of SFAS No. 123R to the beginning of the first fiscal year after June 15, 2005. As a result, the Company anticipates adopting SFAS No. 123R on January 1, 2006. As the Company currently accounts for employee stock-based compensation under the intrinsic value method permitted under APB Opinion No. 25, the adoption of SFAS No. 123R will result in significant additional compensation charges in the Company’s statements of operations, although it will have no impact on its overall financial position.
In May 2005, the FASB issued SFAS No. 154, “Accounting Changes and Error Corrections-a replacement of APB Opinion No. 20 and FASB Statement No. 3” (SFAS No. 154). This Statement replaces APB Opinion No. 20, “Accounting Changes” and FASB Statement No. 3, “Reporting Accounting Changes in Interim Financial Statements,” and changes the requirements for the accounting for and the reporting of a change in accounting principle. SFAS No. 154 applies to all voluntary changes in an accounting principle. It establishes, unless impractical, retrospective application as the required method for reporting a voluntary change in accounting principle. It also applies to changes required by an accounting pronouncement in the unusual instance that the pronouncement does not include specific transition provisions. When a pronouncement includes specific transition provisions, those provisions should be followed. SFAS No. 154 is effective for accounting changes and error corrections occurring in fiscal years beginning after December 15, 2005.
(3) Cash, Cash Equivalents and Short-term Investments
Cash and cash equivalents include only securities having a maturity of three months or less at the time of purchase. At December 31, 2005 and 2004 demand accounts, money market accounts and U.S. Treasury and agency obligations comprised all of the Company’s cash and cash equivalents.
Short-term investments consist of corporate auction-rate securities with original maturities of greater than three months. As of December 31, 2005 and 2004, all the auction-rate securities held have maturities in excess of 25 years. The Company’s investment policy permits investments in auction-rate securities that have interest reset dates of three months or less at the time of purchase. The reset date is the date in which the underlying interest rate is revised based on a Dutch auction and the underlying security may be readily sold. Although the securities held have extended maturities, the Company classifies these securities as current as they are available for sale under SFAS No. 115.
92
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
The following summarizes cash, cash equivalents and short-term investments (in thousands):
| Cost | Gross unrealized gains | Gross unrealized losses | Fair Value | |||||||||
Cash and Cash Equivalents | ||||||||||||
Demand deposits | $ | 2,151 | $ | — | $ | — | $ | 2,151 | ||||
Money market accounts | 3,234 | — | — | 3,234 | ||||||||
Obligations of U.S. Government | 25,860 | 135 | — | 25,995 | ||||||||
December 31, 2005 | $ | 31,245 | $ | 135 | $ | — | $ | 31,380 | ||||
Short-Term Investments | ||||||||||||
Auction-rate securities | $ | 25,350 | $ | — | $ | — | $ | 25,350 | ||||
December 31, 2005 | $ | 25,350 | $ | — | $ | — | $ | 25,350 | ||||
Cash and Cash Equivalents | ||||||||||||
Demand deposits | $ | 1,299 | $ | — | $ | — | $ | 1,299 | ||||
Money market accounts | 11,055 | — | — | $ | 11,055 | |||||||
Obligations of U.S. Government | 20,301 | 52 | — | $ | 20,353 | |||||||
December 31, 2004 | $ | 32,655 | $ | 52 | $ | — | $ | 32,707 | ||||
Short-Term Investments | ||||||||||||
Auction-rate securities | $ | 14,100 | $ | — | $ | — | $ | 14,100 | ||||
December 31, 2004 | $ | 14,100 | $ | — | $ | — | $ | 14,100 | ||||
There were no realized gains and losses recorded in interest income for the years ended December 31, 2005, 2004 and 2003.
(4) Inventories
Inventories consist of the following (in thousands):
| December 31, | ||||||
| 2005 | 2004 | |||||
Raw materials | $ | 1,258 | $ | 2,737 | ||
Work-in-process | 1,422 | 481 | ||||
Finished goods | 1,491 | 318 | ||||
Inventory subject to return | 216 | 478 | ||||
| $ | 4,387 | $ | 4,014 | |||
Testim is manufactured for the Company by DPT Laboratories, Ltd., or DPT, under a contract that was to expire on December 31, 2005. On September 14, 2005, the Company amended the contract with DPT to, among other things, extend the contract for an additional five years through December 31, 2010.
Inventory subject to return represents the amount of Testim shipped to wholesalers and chain drug stores that had not been recognized as revenue (see Note 2e).
93
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Prior to the FDA approval of Testim in the fourth quarter of 2002, the Company purchased approximately $900,000 of a key raw material for which it would have had no future alternative future use if Testim were not approved. The Company charged these raw materials directly to research and development expenses in 2002. By December 31, 2004, all this raw material had been utilized.
Included in work-in-process inventory at December 31, 2005 is $1,091,000 of Testim validation batches manufactured by a back-up supplier. The Company intends to sell this inventory after it receives approval of the manufacturing facility and process from the relevant regulatory authority. Although additional required testing of this inventory remains to be completed, the Company capitalized this inventory based on favorable preliminary results from analytical testing, past product experience and Management’s belief that it is probable that the inventory will be saleable.
(5) Property and Equipment
Property and equipment consists of the following:
Estimated useful life | December 31, | |||||||||
| 2005 | 2004 | |||||||||
Office furniture, computer equipment and software | 3 to 5 years | $ | 1,850 | $ | 1,753 | |||||
Manufacturing equipment | 3 to 10 years | 1,943 | 1,139 | |||||||
Laboratory equipment | 7 years | 260 | 260 | |||||||
Leasehold improvements | lease term | 1,001 | 68 | |||||||
| 5,054 | 3,220 | |||||||||
Less accumulated depreciation and amortization | (1,572 | ) | (1,131 | ) | ||||||
| 3,482 | 2,089 | |||||||||
Construction-in-progress—laboratory and manufacturing equipment | 208 | 465 | ||||||||
| $ | 3,690 | $ | 2,554 | |||||||
Depreciation expense was $883,000, $655,000 and $395,000 for the years ended December 31, 2005, 2004 and 2003, respectively.
(6) Supply Agreement
During 2002, the Company entered into a supply agreement, which was amended most recently in September 2005, with a contract manufacturer for the production of Testim over the first three market years. In connection with the agreement, the Company agreed to make non-refundable payments totaling approximately $2.1 million in order to modify the manufacturer’s facility specifically for Testim manufacturing. Approximately $1.9 million of the modification was incurred prior to Testim FDA approval and was charged to research and development expense in 2002, as there was no alternative future use for the supply agreement prior to FDA approval. The remaining amount of approximately $220,000 was recorded as a supply agreement deferred cost (included in other assets) and is being amortized over the remaining life of the supply agreement. The Company purchased approximately $1.1 million of packaging equipment that is used by the contract manufacturer in Testim production. The Company owns the equipment. The packaging equipment was placed in service at the end of 2003 and is being amortized over the estimated life of the equipment. Although the Company is not required to purchase any minimum amount of Testim from its manufacturer, it is required to pay a minimum annual manufacturing fee of $190,000.
94
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(7) Accrued Expenses
Accrued expenses consist of the following (in thousands):
| December 31, | December 31, | |||||
| 2005 | 2004 | |||||
Payroll and related expenses | $ | 3,672 | $ | 3,030 | ||
Milestone and royalty expenses | 2,846 | 1,882 | ||||
Research and development expenses | 2,018 | 1,285 | ||||
Sales and marketing expenses | 777 | 530 | ||||
Testim coupon and rebate accrual | 2,214 | 929 | ||||
Other expenses | 1,314 | 988 | ||||
| $ | 12,841 | $ | 8,644 | |||
(8) Notes Payable
Notes payable consists of the following (in thousands):
| December 31, | ||||||||
| 2005 | 2004 | |||||||
Bank lines of credit | $ | — | — | |||||
Equipment promissory notes | 327 | 754 | ||||||
| 327 | 754 | |||||||
Less unamortized discount | (2 | ) | (4 | ) | ||||
Less current portion of notes payable | (260 | ) | (427 | ) | ||||
Notes payable, long-term portion | $ | 65 | $ | 323 | ||||
In May 2003, the Company entered into a line of credit with a commercial bank pursuant to which the Company had the ability to borrow up to $9,750,000 under a revolving promissory note maturing on May 1, 2004. This note bore interest at the greater of the bank’s prime rate or 4.25% and was due monthly, with principal due only at maturity. At December 31, 2003, principal outstanding under the line was $9,500,000 and the interest rate was 4.25%. Interest incurred under the line during 2004 and 2003 was $117,000 and $224,000, respectively. The line was secured by all the Company’s assets. In order to obtain the line, the Company issued warrants to purchase 399,994 shares of the Company’s common stock to certain of its preferred stockholders in consideration for their guarantee of the borrowings under the line. The Company estimated the fair value of the warrants based on the Black-Scholes option-pricing model to be $1,000,000. The value of the warrants, plus transaction costs of $187,000, was capitalized as debt issuance costs and was amortized into interest expense over the life of the line on a straight-line basis. At December 31, 2003, accumulated amortization on these debt issuance costs was $790,000. The remaining debt issuance costs were amortized in 2004, see below. The debt issuance costs were included in other current assets on the consolidated balance sheet as of December 31, 2003.
In connection with the Company’s Series D preferred stock transaction in October 2003 (see Note 11c), the guarantee of the bank line was terminated, the warrants issued for the guarantee were cancelled and the bank accepted a $9,500,000 certificate of deposit from the Company to secure the borrowings under the line of credit (see Note 3) until new terms could be negotiated. In March 2004, the Company fully repaid the $9,500,000 existing bank line of credit by using the proceeds from the certificate of deposit. In the first quarter of 2004, the unamortized debt issuance costs of approximately $140,000 were charged to interest expense.
95
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
In March 2004, the Company entered into a new agreement with another commercial bank, Comerica Bank. The new line of credit was for $5,000,000 and expired in March 2005. The line bears interest at the bank’s prime rate plus 0.5% and was secured by all the Company’s tangible assets. If the amount of the Company’s cash balance at the bank fell below $10.0 million and the Company had borrowings outstanding on the line, the bank would have restricted cash on deposit based on a formula of accounts receivable. The maximum restricted cash amount would have been the full amount of the line. In addition, if the Company’s cash balance fell below $10.0 million, the Company would have been subject to financial covenants. The Company drew under this line in early 2004 and paid $23,000 in interest during 2004. There were no amounts outstanding under this facility as of December 31, 2004.
During 2002, the Company entered into an equipment debt agreement with a financing institution. In June 2003, the Company borrowed $1,091,000 under this facility to fund the acquisition of manufacturing equipment and other fixed assets. The borrowings are secured by the underlying financed assets. The facility bears interest at 9.5% and is repayable in 36 equal monthly payments. Interest incurred under the facility during 2004 and 2003 was $73,000 and $54,000, respectively. In addition, in connection with entering into the facility, the lender received a warrant to purchase 5,817 shares of the Company’s common stock at $7.50 per share exercisable for 5 years. The Company estimated the fair value of the warrants based on the Black-Scholes option-pricing model to be $8,000. The Company recorded the fair value of these warrants as a debt discount in 2003 and is amortizing it as additional interest expense over the term of the agreement.
In December 2004, the Company entered into a second equipment debt agreement with the same financing institution. The Company borrowed $171,000 under this facility to fund the acquisition of computer equipment and other fixed assets. The borrowings are secured by the underlying financed assets. The facility bears interest at 8.8% and is repayable in 36 equal monthly payments. Interest incurred under the facility during 2004 was $1,000.
In February 2004, the Company entered into a debt agreement with a financing institution to finance $421,000 of insurance premiums. The facility bore interest at 5.45% and was repaid entirely in 2004. Interest paid under this facility in 2004 was $11,000.
Maturities of notes payable at December 31, 2005 are as follows (in thousands):
2006 | $ | 260 | |
2007 | 62 | ||
2008 | 5 | ||
| $ | 327 | ||
(9) Commitments and Contingencies
(a) Leases
The Company leases its main office space under a noncancelable operating lease that expires in 2012. The Company also leases satellite office space, office equipment and automobiles. Rent expense was $782,000, $457,000 and $450,000 for the years ended December 31, 2005, 2004, and 2003, respectively.
On December 31, 2004, the Company entered into a 90-month lease agreement to move its headquarters facility to Malvern, Pennsylvania. The Company moved into the new office space in June of 2005. The Company paid a $400,000 security deposit under this new lease in January 2005, which is included in other assets at
96
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
December 31, 2005. The Company received an allowance to improve the new facility of up to $781,000. The Company recorded the cost of the improvements as a fixed asset and the allowance as a deferred rent credit. The Company amortizes the leasehold improvement asset over the shorter of the life of the improvements or the life of the lease. The Company amortizes the deferred rent credit as a reduction of rent expense on a straight-line basis over the life of the lease. The new lease includes a period of free rent and escalating minimum rent payments. The Company records rent expense for the minimum lease payments on a straight-line basis. In addition, the Company recorded a charge to general and administrative expenses in June 2005 of $211,000 when it ceased use of its former office facility. This charge represents the rent to be paid for unutilized office space under the existing noncancelable lease.
Future minimum lease payments under noncancelable operating leases for office space, equipment and automobiles as of December 31, 2005 are as follows (in thousands):
2006 | $ | 485 | |
2007 | 478 | ||
2008 | 460 | ||
2009 | 453 | ||
2010 | 456 | ||
2011 and thereafter | 900 | ||
| $ | 3,232 | ||
(b)Research, Development and License Agreements
The Company has entered into various licensing agreements. Through December 31, 2005, the Company has made aggregate milestone payments under all agreements of $9,625,000. The Company could make an additional $27,725,000 of contingent milestone payments under all agreements if all underlying events occur over the life of the agreements.
Bentley Pharmaceuticals, Inc.
In May 2000, the Company entered into a license agreement with Bentley Pharmaceuticals, Inc., or Bentley, for its patented transdermal gel technology. Under the agreement, as amended, the Company was granted an exclusive, worldwide, royalty-bearing license to make and sell products incorporating Bentley’s formulation technology that contain testosterone. The Company produces Testim under this agreement. The term of this agreement is determined on a country-by-country basis and extends until the later of patent right termination in a country or 10 years after the first sale of product in that country. In May 2001, the Company entered into a license with Bentley granting similar rights for a product containing another hormone, the term of which is perpetual. In May 2001, the Company also entered into a separate license granting similar rights for a product containing a therapeutic drug.
In addition to its royalty obligations, the Company is obligated to pay certain upfront payments and milestones under the license agreements with Bentley. Through December 31, 2005, the Company has made aggregate upfront and milestone payments under the three license agreements with Bentley of $625,000. If all events under the Bentley license agreements occur, the Company would be obligated to pay a maximum of an additional $1,025,000 in milestone payments.
97
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
BioSpecifics Technologies Corp.
In June 2004, the Company entered into a development and license agreement with BioSpecifics Technologies Corp., or BioSpecifics, and amended such agreement in May 2005 (the BioSpecifics Agreement). Under the BioSpecifics Agreement, as amended, the Company was granted exclusive worldwide rights to develop, market and sell certain products containing BioSpecifics’ enzyme AA4500. The Company’s licensed rights concern the development of products, other than dermal formulations labeled for topical administration. Currently, the Company is developing AA4500 for the treatment of Dupuytren’s Disease, Peyronie’s Disease and Frozen Shoulder Syndrome. The Company may expand the BioSpecifics Agreement, at its option, to cover other indications as they are developed by the Company or BioSpecifics.
The BioSpecifics Agreement extends, on a country-by-country and product-by-product basis, for the longer of the patent life, the expiration of any regulatory exclusivity period or 12 years. The Company may terminate the BioSpecifics Agreement with 90 days written notice.
The Company is responsible, at its own cost and expense, for developing the formulation and finished dosage form of products and arranging for the clinical supply of products. As permitted under the BioSpecifics Agreement, the Company has qualified Cobra Biologics Ltd., or Cobra, as its primary supplier of clinical supply under the agreement.
BioSpecifics has the option, exercisable no later than six months after FDA approval of the first New Drug Application or Biologics License Application with respect to a product, to assume the right and obligation to supply, or arrange for the supply from a third party other than a back-up supplier qualified by the Company, of a specified portion of commercial product required by the Company. The agreement provides that the Company may withhold a specified amount of a milestone payment until (i) BioSpecifics executes an agreement, containing certain milestones, with a third party for the commercial manufacture of the product, (ii) BioSpecifics commences construction of a facility, compliant with current Good Manufacturing Practices, for the commercial supply of the product, or (iii) 30 days after BioSpecifics notifies the Company in writing that it will not exercise the supply option.
If BioSpecifics exercises the supply option, commencing on a specified date, BioSpecifics will be responsible for supplying either by itself or through a third party other than a back-up supplier qualified by the Company, a specified portion of the commercial supply of the product. If BioSpecifics does not exercise the supply option, the Company will be responsible for arranging for the entire commercial product supply. In the event BioSpecifics exercises the supply option, BioSpecifics and the Company are required to use commercially reasonable efforts to enter into a commercial supply agreement on customary and reasonable terms and conditions which are not worse than those with back-up suppliers qualified by the Company.
The Company must pay BioSpecifics on a country-by-country and product-by-product basis a specified percentage of net sales for products covered by the BioSpecifics Agreement. Such percentage may vary depending on whether BioSpecifics exercises the supply option. In addition, the percentage may be reduced if (i) BioSpecifics fails to supply commercial product supply in accordance with the terms of the BioSpecifics Agreement; (ii) market share of a competing product exceeds a specified threshold; or (iii) the Company is required to obtain a license from a third party in order to practice BioSpecifics’ patents without infringing such third party’s patent rights.
In addition to the payments set forth above, the Company must pay BioSpecifics an amount equal to a specified mark-up of the cost of goods sold for products sold by the Company that are not manufactured by or on behalf of BioSpecifics, provided that, in the event BioSpecifics exercises the supply option, no payment will be due for so long as BioSpecifics fails to supply the commercial supply of the product in accordance with the terms of the BioSpecifics Agreement.
98
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Finally, the Company is obligated to make contingent milestone payments upon contract initiation, receipt of technical data, the one-year anniversary of the BioSpecifics Agreement, filing of regulatory applications and receipt of regulatory approval. Through December 31, 2004, the Company paid up-front and milestone payments under the BioSpecifics Agreement of $5,000,000. The Company recorded two payments of $2,500,000 each as an in-process research and development expense in each of the second and third quarters of 2004 because AA4500 is in the early stages of Phase II clinical trials and there is no alternative future use for AA4500 prior to FDA approval. In March 2005, the Company accrued the one-year contract anniversary payment of $3,000,000 which was paid in June 2005. The Company recorded this accrual as an additional in-process research and development expense in the first quarter of 2005 because AA4500 is in the early stages of Phase II clinical trials and there is no alternative future use for AA4500 prior to FDA approval. In December 2005, the Company paid and expensed the option exercise fee of $500,000 for the Frozen Shoulder Syndrome indication. The Company could make up to an additional $5,500,000 of contingent milestone payments under the BioSpecifics Agreement if all existing conditions are met. Additional milestone obligations may be due if the Company exercises an option to develop and license AA4500 for additional medical indications.
Formulation Technologies, L.L.C., d/b/a PharmaForm
In June 2003, the Company expanded its drug delivery technology portfolio by entering into a license agreement with Formulation Technologies, L.L.C., d/b/a PharmaForm, or PharmaForm. Under this agreement, as amended, the Company was granted an exclusive, worldwide, royalty-bearing license to develop, make and sell products that contain hormones or that are used to treat urologic disorders incorporating PharmaForm’s oral transmucosal film technology for which there is an issued patent in the U.S. The term of this license agreement is for the life of the licensed patents.
In February 2005, the Company entered into an additional license agreement with PharmaForm. Under this agreement, the Company was granted exclusive, worldwide royalty-bearing rights to develop, manufacture and market eight compounds using PharmaForm’s proprietary transmucosal film technology for the management of pain, including acute and chronic pain. The compounds that may be developed include opioids as well other types of analgesics. In 2005, the Company paid an up-front payment of $500,000 and could make up to an additional $21,200,000 in contingent milestone payments if all underlying events for these eight products occur. In addition, the Company will have on-going royalty payment obligations to PharmaForm. The timing of the remaining payments, if any, is uncertain.
To speed the development of products using the licensed technology from PharmaForm, the Company entered into a research and development agreement with PharmaForm on a fee for service basis. The Company will be the sole owner of any intellectual property rights developed in connection with this agreement. The Company incurred $2.3 million under the research and development agreement in 2005.
(c) Manufacturing Agreement
The Company has qualified Cobra as a back-up supplier of AA4500 under the BioSpecifics Agreement. On July 27, 2005 the Company entered into an agreement with Cobra for the process development, scale up and manufacture of AA4500 for Phase II/III clinical trials (the Cobra Agreement). Accordingly, the Company will now rely on Cobra as its primary supplier of AA4500 for clinical trials.
The Cobra Agreement is divided into five stages with specific objectives, timelines, down payments and total payments that attach to each stage. The Company may terminate the Cobra Agreement at the completion of each stage if the objective or the timeline for such stage is not met. Through December 31, 2005, the Company incurred a total of $2.1 million under the Cobra Agreement.
99
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(d) Co-promotion Agreement
In April 2005, the Company entered into a co-promotion agreement (the Oscient Agreement) with Oscient whereby the Company and Oscient will co-promote Testim in the U.S. Oscient was granted the exclusive right to promote Testim jointly with the Company to primary care physicians by means of its sales force. Oscient commenced co-promotion of Testim in May 2005. The initial term of the Oscient Agreement ends on April 30, 2007. Oscient may extend the Oscient Agreement for two consecutive two-year periods provided that Oscient has met certain milestones for each extension. If these milestones are met and Oscient does not elect to terminate the Oscient Agreement, the first extension period will commence on January 1, 2007 and end on December 31, 2008 and the second extension period will commence effective January 1, 2009 and end on April 30, 2011.
The Company and Oscient utilize a joint marketing and promotion plan which sets forth the responsibilities of both parties with respect to the marketing and promotion of Testim in the U.S. primary care physician market. The Company and Oscient share equally these promotional expenses. Each party will be responsible for the costs associated with its own sales force. The combined spending for marketing and promotion of Testim in the primary care physician market in the U.S. for the year ended December 31, 2005 was $7,072,000.
Oscient will be compensated for its services based on a specified percentage of the gross profit from Testim sales attributable to primary care physicians in the U.S. that exceed a specified sales threshold (the “Oscient co-promotion fees”). All gross profit earned on sales to primary care physicians below the predetermined threshold and all gross profit earned on all sales to non primary care physicians are not subject to the terms of the Oscient Agreement. Once sales to primary care physicians have exceeded the predetermined sales threshold, the gross profit earned on such sales is available to be split between the Company and Oscient. Once all agreed upon promotional expenses are appropriately reimbursed to each party, the gross profit remaining will be divided according to agreed upon percentages. The specific percentages are based upon Testim sales levels above the threshold attributable to primary care physicians actually achieved. For the year ended December 31, 2005, the Company has recorded $3,536,000 of expense for the Oscient co-promotion fees.
The agreed upon promotional expenses for each year are reimbursable equally and simultaneously to each company through the profit split mechanism, except in 2005 when Oscient will be disproportionately reimbursed its share of the promotional expenses through the profit split mechanism. As the agreed upon percentage of the gross profit available to be split in 2005 was insufficient to reimburse Oscient its share of the promotional expenses, that portion not reimbursed to Oscient will be carried over to 2006 and will be reimbursed through the 2006 profit split mechanism. Of the $3,536,000 of expense recorded by the Company for the year ended December 31, 2005, $582,000 was due to this carry over reimbursement.
The Oscient Agreement can be terminated by either party upon the occurrence of certain termination events. The Company may be obligated to make termination payments in certain instances. Also, Oscient has been granted the exclusive option to co-promote any future product candidate of the Company that treats hypogonadism and contains testosterone as the active ingredient.
The Company will invoice and record in its financial statements all sales of Testim, including those sales to primary care physicians that exceed the specified threshold, as well as the gross profit earned on all sales. The Company will record its share of promotional expenses incurred and the Oscient co-promotion fees as “selling, general and administrative” operating expenses on the consolidated statement of operations.
100
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
(e) Employment Agreements
As of December 31, 2005, the Company has outstanding employment agreements with five of its executive officers. These agreements provide for annual compensation reviews to be performed by either the board of directors or the compensation committee. These agreements also provide that the executives may participate in all incentive compensation and benefit plans provided to all employees. If the Company terminates the employment of any of these executives without cause, as defined in the agreements, or due to a disability, the Company is obligated to pay severance equal to nine or 12 months of the executive’s base salary and continue to pay medical, dental and prescription drug benefits for the same period. In the event of a change in control and the termination of the executive without cause or if the executive resigns for good reason, as defined in the agreements, the Company is obligated to pay lump sum payments ranging from 0.75 to 1.25 times the executive’s annual base salary, plus 0.75 or 1.25 times the executive’s average annual bonus, plus continued medical, dental and prescription drug benefits for a 9-month or 15-month period as well as the immediate vesting of all outstanding options and stock awards. If payments become due as a result of a change of control and the excise tax imposed by Internal Revenue Code Section 4999 applies, the terms of the employment agreements require the Company to pay the executives the amount of this excise tax plus the amount of income and other taxes due as a result of the payment of the amount of the excise tax.
(f) Litigation
In March 2004, the Company received a judgment against it in a lawsuit tried in October 2003 relating to a breach-of-contract claim. At December 31, 2003, the Company accrued the $722,000 judgment and included it in selling, general and administrative expenses in the accompanying consolidated statement of operations. The amount accrued was paid in April 2004.
(10)Income Taxes
At December 31, 2005, the Company had Federal net operating loss carryforwards of approximately $116.6 million, which will expire in 2019 through 2025, if not utilized. In addition, the Company had state net operating loss carryforwards of approximately $96.0 million, of which $56.2 million relate to Pennsylvania. The Pennsylvania state net operating losses are subject to a $2.0 million annual limitation for 20 years and expire in 2009 through 2025. At December 31, 2005, the Company had Federal research and development credits of approximately $1.7 million that will expire in 2020 through 2025, if not utilized.
The Tax Reform Act of 1986 (the Act) provides for a limitation on the annual use of net operating loss and research and development tax credit carryforwards following certain ownership changes (as defined by the Act) that could limit the Company’s ability to utilize these carryforwards. The Company may have experienced various ownership changes, as defined by the Act, as a result of past financings. Accordingly, the Company’s ability to utilize the aforementioned carryforwards may be limited. Additionally, U.S. tax laws limit the time during which these carryforwards may be applied against future taxes; therefore the Company may not be able to take full advantage of these carryforwards for Federal income tax purposes.
101
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
The components of the net deferred tax assets are as follows (in thousands):
| December 31, | ||||||||
| 2005 | 2004 | |||||||
Net operating losses | $ | 44,041 | $ | 31,187 | ||||
Research and development credit | 1,668 | 1,172 | ||||||
Depreciation and amortization | 809 | 1,767 | ||||||
Accruals and reserves | 822 | 411 | ||||||
Deferred profit | 4,964 | 4,240 | ||||||
Other temporary differences | 417 | 140 | ||||||
Gross deferred tax assets | 52,721 | 38,917 | ||||||
Deferred tax assets valuation allowance | (52,721 | ) | (38,917 | ) | ||||
| $ | — | $ | — | �� | ||||
In assessing the realizability of deferred tax assets, the Company considers whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which the temporary differences representing net future deductible amounts become deductible. Due to the Company’s history of losses, the deferred tax assets are fully offset by a valuation allowance at December 31, 2005 and 2004. The valuation allowance in 2005 increased by $13,804,000 over 2004 related primarily to additional net operating losses incurred by the Company.
(11) Common Stock and Redeemable Convertible Preferred Stock
(a)Common Stock
The Company is authorized to issue 120,000,000 shares of common stock, with a par value of $0.01 per share.
On June 21, 2004, the Company effected a one-for-five reverse split of its common stock. All share and per share amounts included in these consolidated financial statements and notes thereto have been adjusted retroactively for all periods presented to give effect to the reverse stock split. The Company’s actual preferred shares, preferred shares prices and per preferred share amounts have not been adjusted for this stock split. However, as a result of the stock split, actual conversion ratios of all preferred stock have been adjusted retroactively from one-to-one to five-to-one.
To date, the Company has funded its operations entirely through equity and bank financings. The fundings include $235,000 raised through December 31, 1999 and an additional $222,000 raised in early 2000 through sales of common stock and stock option exercises of officers and non-employee directors.
On March 2, 2000, the Company borrowed $90,000 in the form of a convertible loan from an individual investor. In accordance with its terms, upon completion of the Series A preferred stock financing described below, the loan principal along with accrued interest of $3,000 was converted into 16,255 shares of common stock of the Company.
In 2003, 423,796 shares of common stock were issued in conjunction with the Series D preferred stock transaction (see Note 11c). Also, 12,175 shares of common stock were issued upon the exercise of common stock options and an additional 1,175 shares were granted to a consultant (see Note 2o).
102
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
On July 28, 2004, the Company completed an IPO in which the Company issued 5,500,000 shares of common stock at $7.50 per share resulting in gross proceeds of $41,250,000. In connection with the offering, the Company paid $2,887,000 in underwriting discounts and commissions and incurred $1,852,000 in other transaction costs. The net proceeds were $36,511,000. Also in connection with the IPO, all of the outstanding shares of the Company’s redeemable convertible preferred stock were converted into shares of common stock.
On June 30, 2005, the Company completed a private placement financing in which the Company issued 8,242,796 shares of common stock and warrants to purchase an aggregate of 2,060,687 shares of common stock with an exercise price of $5.84 per share resulting in gross proceeds to the Company of approximately $40,400,000. In connection with the private placement financing, the Company paid $1,242,000 in placement agent fees and incurred approximately $175,000 in other transaction costs. Approximately $216,000 of the placement agent fees and transaction costs which were allocated to the warrants were expensed by the Company during the quarter ended June 30, 2005. The net proceeds to the Company were approximately $38,983,000.
The warrants have an exercise price per share of $5.84, with a five-year life and are fully vested and exercisable. In accordance with EITF 00-19, “Accounting for Derivative Financial Instruments Indexed to, and Potentially Settled in, a Company’s own stock,” (“EITF 00-19”) the warrants were recorded as a long-term liability and valued at fair value on the date of issuance. The fair value of the warrants on the date of issuance was $6,161,000 and was determined using the Black-Scholes model with the following assumptions: dividend yield of 0%; estimated volatility of 80%; risk free interest rate of 3.76% and a contractual life of five years.
As required by the terms of the securities purchase agreements pursuant to which the private placement was consummated, the Company filed a registration statement with the SEC on August 12, 2005 to register for resale the shares of common stock and the shares of common stock issuable upon the exercise of the warrants sold in the private placement. Such registration statement was declared effective by the SEC on September 9, 2005.
Under the terms of the securities purchase agreements, the Company agreed to use its reasonable best efforts to keep the registration statement effective until the earlier of two years after the effective date of the registration statement, the date on which the shares of common stock and the shares of common stock issuable upon exercise of the warrants become eligible for resale pursuant to Rule 144(k) under the Securities Act of 1933, as amended, or any other rule of similar effect, or such time as all such shares have been sold by the purchasers. If, after the registration statement is declared effective, the Company suspends the use of the registration statement by the purchasers for the resale of the shares, the Company has agreed to pay each purchaser as liquidated damages an amount equal to 0.0333% of the purchase price paid by each such purchaser for the securities in the private placement for each day that the use of the registration statement is suspended in excess of 45 consecutive days or 60 days in the aggregate in any 12-month period.
EITF 00-19 addresses accounting for equity derivative contracts indexed to, and potentially settled in, a company’s own stock, or equity derivatives, by providing guidance for distinguishing between permanent equity, temporary equity and assets and liabilities. Under EITF 00-19 to qualify as permanent equity, the equity derivative must permit the issuer to settle in unregistered shares. The terms of the Company’s securities purchase agreements by which the private placement was consummated permit settlement of the warrant in unregistered shares; however, the damages payable in cash arising from failure to register or failure to keep the registration statement effective for up to two years are so significant that they precluded equity treatment. Thus, this equity derivative could not be classified as permanent equity. Equity derivative contracts not qualifying for permanent equity accounting are recorded at fair value as a liability with subsequent changes in fair value recognized through the statement of operations.
103
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
While the Company viewed this liquidated damages contingency as neither probable nor reasonably estimable, the Company recorded the estimated fair value of the warrants as of June 30, 2005 of $6,161,000 as a long-term financing–related liability in its consolidated balance sheet at June 30, 2005, in accordance with EITF 00-19.
On December 30, 2005, the Company and the purchasers amended the securities purchase agreements (the “Amendments”). The Amendments delete in its entirety the liquidated damages provision discussed above. The estimated fair value of the warrants on December 30, 2005 of $7,151,000 was reclassified to additional paid-in capital since the equity derivative qualifies as permanent equity under EITF 00-19. The non-cash change in the fair value of the warrants of $990,000 was recorded as other expense for the year ended December 31, 2005.
(b) Preferred Stock
The Company is authorized to issue 5,000,000 shares of preferred stock, with a par value of $0.01 per share. No preferred stock is issued or outstanding.
(c) Redeemable Convertible Preferred Stock
In connection with the IPO, all shares of redeemable convertible preferred stock were converted into common stock on a five-to-one basis resulting in the issuance of 14,058,482 shares of common stock. After the IPO, there are no longer any shares of redeemable convertible preferred stock authorized. In conjunction with the Series D redeemable convertible preferred stock transaction in October 2003, 8,634,339 warrants were issued that were originally exercisable into shares of Series D preferred stock for seven years at $1.50 per share. Initially, the exercise price and the amount of warrants were subject to change based on the achievement of certain business milestones. In April 2004, the warrants were amended to remove the variable features by fixing the amount of warrants and the warrant exercise price at $1.125 per share. The Company recorded, in the second quarter of 2004, a non-cash deemed dividend to the warrant holders utilizing the Black-Scholes option pricing model to estimate the fair value of the modification to the warrants. The amount of the deemed dividend represents the increase in the fair value of the warrants attributable to the amendment to the warrant terms. The fair value estimates included an assessment of the probability of the variable factors existing immediately before the amendment. The deemed dividend of $173,000 increased the net loss applicable to common stockholders in the second quarter of 2004. In connection with the IPO, the warrants to purchase Series D preferred stock became warrants to purchase 1,726,859 shares of common stock at $5.625 per share.
As of December 31, 2003, the authorized and outstanding redeemable convertible preferred stock and their principal terms were as follows (in thousands, except share amounts):
Series | Shares authorized | Shares outstanding | Carrying amount | Liquidation value | ||||||
A | 8,771,928 | 8,771,928 | $ | 9,763 | $ | 10,000 | ||||
B | 22,484,888 | 22,404,888 | 27,447 | 28,006 | ||||||
C | 10,283,336 | 10,283,336 | 15,036 | 15,425 | ||||||
D | 37,386,704 | 28,752,365 | 41,041 | 43,129 | ||||||
| 78,926,856 | 70,212,517 | $ | 93,287 | $ | 96,560 | |||||
104
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
The following table summarizes the activity in the carrying amount of redeemable convertible preferred stock since January 1, 2003 (in thousands):
| Series A | Series B | Series C | Series D | Total | ||||||||||||||||
Balance, January 1, 2003 | 11,949 | 30,363 | 15,527 | — | 57,839 | |||||||||||||||
Issuance of preferred stock | — | — | — | 40,969 | 40,969 | |||||||||||||||
Accretion of preferred stock | 84 | 197 | 137 | 72 | 490 | |||||||||||||||
Accrual of preferred dividends | 820 | 2,113 | 976 | — | 3,909 | |||||||||||||||
Recapitalization | (3,090 | ) | (5,226 | ) | (1,604 | ) | — | (9,920 | ) | |||||||||||
Balance, December 31, 2003 | 9,763 | 27,447 | 15,036 | 41,041 | 93,287 | |||||||||||||||
Accretion of preferred stock | 28 | 68 | 47 | 252 | 395 | |||||||||||||||
Exercise of preferred stock warrant | — | 100 | — | — | 100 | |||||||||||||||
Conversion of preferred stock in initial public offering | (9,791 | ) | (27,615 | ) | (15,083 | ) | (41,293 | ) | (93,782 | ) | ||||||||||
Balance, December 31, 2004 | $ | — | $ | — | $ | — | $ | — | $ | — | ||||||||||
The following table summarizes the issuances of redeemable convertible preferred stock and warrants through December 31, 2004 (in thousands, except share and per share amounts):
| Allocated Proceeds | ||||||||||||||||||
Month | Series | Price | Shares issued | Warrants issued | Preferred stock | Warrants | Total | |||||||||||
July 2000 | A | $ | 1.14 | 4,385,964 | 877,192 | $ | 4,426 | $ | 574 | $ | 5,000 | |||||||
May 2001 | A | 1.14 | 4,385,964 | — | 5,000 | — | 5,000 | |||||||||||
Oct. 2001 | B | 1.25 | 22,404,888 | 1,344,288 | 26,992 | 1,014 | 28,006 | |||||||||||
June 2002 | C | 1.50 | 10,283,336 | 616,997 | 14,819 | 606 | 15,425 | |||||||||||
Oct. 2003 | D | 1.50 | 28,752,365 | 8,634,339 | 40,969 | 2,160 | 43,129 | |||||||||||
July 2004 | B | 1.25 | 80,000 | — | 100 | — | 100 | |||||||||||
In each of the above transactions, the Company allocated the proceeds to the preferred stock and warrants based on the estimated relative fair values of each instrument. The Company used the Black-Scholes option-pricing model to determine the fair value of the warrants issued in each of these transactions. Also, the warrants issued with the Series A, Series B and Series C transactions were each issued at or above the preferred stock issuance price and were all cancelled in conjunction with the Series D transaction. The Series D warrants were converted into common stock warrants in conjunction with the IPO. The Company evaluated the Series A, B, C and D preferred stock transactions together with the bridge financings discussed below and determined that none of these transactions had a beneficial conversion feature as identified in EITF Issue No. 00-27,Application of Issue No. 98-5 to Certain Convertible Instruments.
In connection with the Series A transaction, the Company issued as bridge financing $500,000 of two-year convertible promissory notes in June 2000, along with warrants to purchase 21,930 shares of common stock for $5.70 per share. The notes were converted into Series A preferred stock in July 2000. After allocating $15,000 of the bridge note proceeds to the warrants, based on the estimated relative fair values of the notes and the warrants using the Black-Scholes option pricing model, the debt was recorded at $485,000. The debt discount was amortized to interest expense over the period the notes were outstanding. These warrants were cancelled in conjunction with the Series D preferred stock transaction.
105
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
In conjunction with the Series B transaction, the Company issued as bridge financing a $1,000,000 one-year convertible promissory note in September 2001, along with warrants to purchase 80,000 shares of Series B preferred stock for $1.25 per share. The note, along with $6,000 of accrued interest, was converted into Series B preferred stock. After allocating $55,000 of the bridge note proceeds to the warrants, based on the estimated relative fair values of the notes and the warrants using the Black-Scholes option pricing model, the debt was recorded at $945,000. The debt discount was amortized to interest over the period the notes were outstanding. These warrants were exercised in July 2004 and the acquired shares of Series B preferred stock converted into 16,000 shares of common stock in connection with the IPO.
The Series A preferred stock was issued with a 10% cumulative dividend provision and was redeemable at the option of the holders on or after July 18, 2005. In conjunction with the Series B preferred stock transaction, the Series A preferred stockholders agreed, on a prospective basis, to reduce their cumulative dividend right to the 8% rate payable to the Series B preferred stockholders. The Series A preferred stockholders also agreed to delay their redemption right to be concurrent with the Series B redemption right on or after July 18, 2006. The Series C preferred stock was issued with a cumulative dividend at 8% and was redeemable at the option of the holder on or after July 18, 2006. In conjunction with the Series D preferred stock transaction, the Series A, Series B and Series C cumulative dividend rights and outstanding common stock warrants were cancelled and the redemption right was made concurrent with the Series D redemption right at the option of the holder on or after October 30, 2008. In addition, the Series A, Series B and Series C preferred stockholders received 423,796 shares of common stock and their guarantee of the Company’s bank line of credit (see Note 8) was terminated. These events were accounted for as a recapitalization equity transaction of the Company with no gain or loss recorded.
Prior to the Series D transaction, all preferred stock dividends were payable in the respective shares of preferred stock and were accrued each period with charges to additional paid-in capital, to the extent available, and otherwise to accumulated deficit. These accumulated dividends were reversed when forgone in 2003. All preferred stock were entitled to dividends only when and if declared by the Board of Directors. Additionally, all preferred stock were entitled to participating dividends if declared to common stockholders by the Board of Directors. In such a case, preferred stockholders would have received the same dividend per share as if their preferred stock were converted into common stock. Series D preferred stock was senior to all other series of preferred stock.
The Series A, Series B, Series C and Series D preferred stock was redeemable at $1.14, $1.25, $1.50 and $1.50 per share, respectively, plus any declared but unpaid dividends. Each of the Series A, Series B, Series C and Series D preferred stock carrying values were being accreted to the redemption amount on a straight-line basis, which was not materially different from the effective-interest method.
Each of the Series A, Series B, Series C and Series D preferred stock was convertible into the Company’s common stock on a five-to-one basis. The conversion rate was subject to adjustment for dilution. Automatic conversion was triggered concurrent with a firm underwritten public offering. Each series of preferred stock would also automatically convert upon the vote of the holders of at least 66 2/3% of the then outstanding shares for Series B, Series C and Series D or the majority of the then outstanding shares of Series A.
In the event of any liquidation, dissolution, or winding up of the Company, holders of preferred stock were entitled to receive preference to any distributions to holders of common stock at an amount equal to the original purchase price per share of the applicable series of preferred stock plus any declared but unpaid dividends. Series D preferred stock is senior to all other preferred stock.
106
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Preferred stockholders were entitled to the number of votes equal to the number of shares of common stock into which each share of preferred stock was convertible. The Series A, Series B and Series D preferred stockholders each had the right to appoint one, one and two members, respectively, to the Company’s board of directors. All preferred stockholders as a class had the right to appoint three members of the Company’s board of directors. The common and preferred stockholders as a class had the right to appoint one member to the Company’s board of directors.
(12)Stock Options and Warrants
In June 2004, the board of directors adopted and the stockholders approved the 2004 Equity Compensation Plan (the “2004 Plan”) authorizing the granting of incentive and nonqualified stock options and stock awards to employees, non-employee directors and service providers. In June 2004, the stockholders also approved the merger of the 2000 Equity Compensation Plan (the “2000 Plan”) into the 2004 Plan and authorized the increase of shares authorized under the 2004 Plan to 2,845,450 shares upon the effectiveness of the registration statement for the IPO. Upon effectiveness of the registration statement for the IPO, 604,780 stock options and 93,437 restricted stock awards were granted to employees and directors. The stock options granted in connection with the IPO vest over four years with an initial vesting date of June 5, 2005 and are exercisable at $7.50 per share. The restricted stock awards vest over four years with an initial vesting date of June 5, 2005. On May 18, 2005, a new officer of the Company was granted 25,000 shares of restricted stock. The restrictions on these shares will lapse as follows: (i) 5,000 shares on May 18, 2007, (ii) 10,000 shares on May 18, 2008 and (iii) 10,000 shares on May 18, 2009.
The Company has recorded $120,000 and $701,000 of deferred compensation for the years ended December 31, 2005 and 2004, respectively, for the fair value of restricted stock awards. The deferred compensation is amortized on a straight-line basis over the vesting period. The Company recorded $165,000 and $95,000 of amortization during the years ended December 31, 2005 and 2004, respectively.
The compensation committee of the board of directors administers the 2004 Plan. The committee determines who will receive stock options or awards. To date, the Company has granted only nonqualified stock options and restricted stock awards under the 2004 Plan. Generally, options granted vest over four years, with immediate vesting upon a change of control of the Company, and have a term of ten years.
During 2000, the Company granted a stock award of 3,000 shares of common stock to a consultant for services provided. These shares were valued at $2,000. Also during 2000, the Company granted options to purchase 67,000 shares of Company common stock to certain directors and officers of the Company that vested in 30 days. Upon exercise, the shares acquired were restricted as to further disposition. The restrictions on the shares release evenly over a four-year period. The Company had the option to repurchase any restricted shares at the original exercise price if the stockholder ceased to provide services to the Company. All of these options were exercised. In order to purchase the restricted shares, the Company made full-recourse stockholder loans for the exercise price of $116,000. These loans are interest-bearing and have a term of five years. Upon the resignation of one of these individuals in 2001, the Company reacquired 18,750 shares of common stock in exchange for canceling $33,000 of the full-recourse loan. In 2004, three of these loans totaling $93,000, including interest, were collected.
Included in the stock option tables below, the Company granted 390,000 nonqualified stock options to three senior executives in 2002. These options vested in five years, but contain vesting acceleration provisions. These options immediately vest upon an initial public offering of the Company’s stock or upon a change of control. Also, these options contained performance-vesting criteria whereby one-third of the options vested immediately upon attaining each of three individual milestones. These option grants were approved with an exercise price of
107
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
$3.125 per share when the fair value of common stock was $3.75 per share. Accordingly, the $244,000 intrinsic value of these options was recorded as deferred compensation. During 2002, two of the three performance criteria were met resulting in a compensation charge of $163,000. The remaining one-third was being amortized over the five-year cliff-vesting period, but was accelerated in the third quarter of 2004 upon the completion of the IPO. Total amortization of this deferred compensation was $57,000 and $16,000 for the years ended December 31, 2004 and 2003, respectively.
Information with respect to the nonqualified stock options granted under the 2004 Plan for the periods identified is summarized as follows:
| Years Ended December 31, | ||||||||||||
| 2005 | 2004 | 2003 | ||||||||||
Options outstanding: | ||||||||||||
Outstanding at beginning of period | 1,858,700 | 977,420 | 887,457 | |||||||||
Granted | 1,070,592 | 1,085,010 | 145,540 | |||||||||
Exercised | (210,687 | ) | (53,112 | ) | (12,175 | ) | ||||||
Canceled | (617,107 | ) | (150,618 | ) | (43,402 | ) | ||||||
Outstanding at end of period | 2,101,498 | 1,858,700 | 977,420 | |||||||||
Exercisable at end of period | 1,186,909 | 728,206 | 512,325 | |||||||||
Weighted average exercise prices: | ||||||||||||
Outstanding at beginning of period | $ | 6.55 | $ | 3.77 | $ | 3.74 | ||||||
Granted | 5.01 | 9.04 | 4.19 | |||||||||
Exercised | 3.52 | 4.39 | 4.37 | |||||||||
Canceled | 6.93 | 6.78 | 4.37 | |||||||||
Outstanding at end of period | 5.92 | 6.55 | 3.77 | |||||||||
Exercisable at end of period | 6.56 | 3.64 | 3.67 | |||||||||
The following table summarizes information about options outstanding at December 31, 2005:
Range of exercise price per share | Shares | Weighted average exercise price per share | Weighted average remaining contractual life in years | ||||
Options outstanding: | |||||||
$1.75 | 400 | $ | 1.75 | 4.1 | |||
$3.125 – $4.25 | 666,759 | 3.64 | 7.1 | ||||
$4.50 – $6.06 | 854,294 | 5.29 | 8.6 | ||||
$7.50 – $8.90 | 406,615 | 8.18 | 8.8 | ||||
$12.50 | 173,430 | 12.50 | 8.2 | ||||
| 2,101,498 | 5.92 | 8.1 | |||||
108
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
The following table summarizes information about options exercisable at December 31, 2005:
Range of exercise price per share | Shares | Weighted average exercise price per share | |||
Options exercisable: | |||||
$1.75 | 400 | $ | 1.75 | ||
$3.125 – $4.25 | 482,670 | 3.46 | |||
$4.50 – $6.06 | 123,794 | 5.06 | |||
$7.50 – $8.90 | 406,615 | 8.18 | |||
$12.50 | 173,430 | 12.50 | |||
| 1,186,909 | 6.56 | ||||
As of December 31, 2005, 313,614 options were available for future grant under the 2004 Plan.
As of December 31, 2005, the Company had the following common stock warrants outstanding:
Expiration date | Number of warrants | Exercise price | |||||
Reference | |||||||
Note 8 | May 2007 | 5,817 | $ | 7.50 | |||
Note 11c | Oct. 2010 | 1,726,859 | 5.625 | ||||
Note 11a | June 2010 | 2,060,687 | 5.840 | ||||
| 3,793,363 | |||||||
(13) Stock Purchase Plan
In June 2004, the Board of Directors adopted and the stockholders approved the 2004 Employee Stock Purchase Plan. The 2004 Employee Stock Purchase Plan permits employees to purchase shares of the Company’s common stock at a 15% discount through payroll deductions. Employees are enrolled in a two-year option period. Employees purchase stock in half-year intervals at 85% of the lower of the fair market value of common stock on the date of purchase or the beginning of the two year option period. There is a maximum of 200,000 shares issuable under the 2004 Employee Stock Purchase Plan. In November 2004, employees purchased an aggregate of 14,268 shares of common stock at a price of $6.375 per share. In May 2005, employees purchased an aggregate amount of 66,530 shares of common stock at a price of $4.063 per share. In November 2005, employees purchased an aggregate of 58,597 shares of common stock at a price of $3.9525 per share.
(14) Testim Marketing and Distribution Agreements
In December 2003, the Company entered into an exclusive marketing and distribution agreement with Bayer, Inc. (“Bayer”) in Canada. The Company filed for regulatory approval of Testim in Canada in March 2004. Under the agreement, Bayer will purchase product and pay certain royalty and milestone payments. The milestones payments are due upon contract signing, filing for regulatory approval in Canada, the first sale of Testim in Canada and upon achieving specified sales levels. The term of the agreement is for the life of the patent covering Testim in Canada.
In March 2004, the Company signed a sublicense agreement with Ipsen Farmaceutica B.V. (“Ipsen”) to use and sell Testim on a worldwide basis outside of the US, Canada, Mexico and Japan. Through February 2005, Testim is approved for marketing in the U.S., the United Kingdom, Belgium, Denmark, Finland, Germany,
109
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
Iceland, the Netherlands, Norway, Portugal, Spain and Sweden. In June 2004, the Company received scientific approval for Testim in an additional four European countries. The Company anticipates that marketing approvals in the remaining countries will be received once local administrative procedures to finalize the packaging text have been completed. Ipsen is responsible for obtaining regulatory approval in non-EU countries. In addition, Ipsen is obligated to purchase product from the Company and to pay royalty and certain milestone payments. The milestone payments are due upon contract signing, regulatory approval, product launch in various countries and supply price reductions. The license is exclusive and royalty-bearing for the longer of ten years from market launch or the expiration of patent protection in any particular country.
Through December 31, 2005, the Company collected $11,900,000 of license payments under these agreements. Of this amount $10,300,000 relates to non-refundable milestones from Ipsen. The remaining $1,600,000 consists of a $1,000,000 refundable milestone from Ipsen and $600,000 of milestones from Bayer that cannot be recognized at this time. During the year ended December 31, 2005, the Company recorded $535,000 of revenue for up-front and milestone amounts received from Ipsen. At December 31, 2005, $694,000 was classified as current deferred revenue and $10,671,000 was classified as long-term deferred revenue. (See Note 2e).
(15)Employee Benefit Plan
The Company sponsors a 401(k) defined contribution plan covering substantially all U.S. employees. The board of directors determines contributions made by the Company annually. The Company made matching contributions of $136,000, $93,000, and $68,000 in 2005, 2004, and 2003, respectively.
In 2001, the Company adopted a defined contribution plan covering substantially all United Kingdom employees. The Company made matching contributions of $70,000, $65,000, and $56,000 in 2005, 2004, and 2003, respectively.
(16) Related-Party Transactions
In 2005, the Company paid $90,000 to Lathian Systems Inc., or Lathian, for marketing services. New Leaf Ventures, L.L.C., or New Leaf, has entered into sub-advisory agreements with two investment funds which have investments in Lathian. One of the Company’s Directors is a Managing Director of New Leaf.
The Company used Prosit, LLC, or Prosit, for printing and copying services. The brother of the Company’s chief executive officer holds a majority ownership interest in Prosit. Prosit received payments from the Company for printing and reproduction services provided of $415,000, $1,099,000 and $846,000 in 2005, 2004 and 2003 respectively. HH Capital Partners, an investment partnership controlled by the Company’s chief executive officer and its former general counsel, converted its majority equity interest in Prosit into a $240,000 loan in 2001. This loan bears interest at 4% and is repayable in four annual payments of $60,000 beginning on June 30, 2003. As of December 31, 2004, Prosit owed HH Capital Partners $180,000 under this loan. In addition, Prosit leases printing and copying equipment from HH Capital Partners. The 48-month lease commenced on January 1, 2001 and requires monthly payments of approximately $1,100. On April 14, 2005, HH Capital Partners forgave Prosit the payment of any outstanding loan, lease and interest payments.
The Company used ePocrates, Inc., or ePocrates, in 2004 for Testim promotion services and new product surveys. A member of our board of directors was also a member of ePocrates’ board. The venture capital firm with which our board member was affiliated beneficially owned approximately 14% of the Company’s common stock and also maintained an investment of greater than 10% in ePocrates. The Company paid ePocrates $109,000 in 2004.
110
Table of Contents
AUXILIUM PHARMACEUTICALS, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS—(Continued)
In 2003, the Company issued 399,994 warrants to purchase the Company’s common stock to certain of its preferred stockholders in connection with the guarantee of borrowings under its bank line of credit. In addition, the Company reimbursed $87,000 of legal fees incurred by one preferred stockholder in connection with their guarantee of the borrowings under the Company’s line of credit. The guarantee and the warrants were cancelled in connection with the Series D preferred stock transaction (see Notes 8 and 11c).
Two siblings and a sister-in-law of the Company’s chief executive officer are employed by the Company. As of December 31, 2005, these individuals held the positions of Vice President of Regulatory and Quality Assurance, Director of Production Operations and Director of Human Resources. The Vice President of Regulatory and Quality Assurance earned, including bonuses, $170,000, $151,000 and $111,000 in 2005, 2004, and 2003, respectively. The Director of Production Operations earned, including bonuses, $113,000, $107,000 and $85,000 in 2005, 2004 and 2003, respectively. The Director of Human Resources earned, including bonuses, $111,000, $97,000 and $70,000 in 2005, 2004 and 2003, respectively.
In 2003, the Company paid $103,000 to the husband of an officer of the Company for marketing consulting services.
(17)Other Income
In the first quarter of 2003, the Company received a payment in connection with the settlement of a lawsuit with a vendor. The settlement, net of expenses of $278,000, is reflected as other income in the accompanying consolidated statement of operations.
(18)Unaudited Quarterly Financial Information (in thousands, except share and per share amounts):
| 2005 | ||||||||||||||||
| 1st Qtr | 2nd Qtr | 3rd Qtr | 4th Qtr | |||||||||||||
Net revenues | $ | 8,797 | $ | 9,839 | $ | 11,230 | $ | 12,938 | ||||||||
Cost of goods sold | 2,828 | 3,149 | 3,279 | 3,863 | ||||||||||||
Research and development expenses | 9,077 | 4,250 | 4,524 | 6,450 | ||||||||||||
Selling, general and administrative expenses | 8,664 | 11,049 | 10,714 | 13,508 | ||||||||||||
Loss from operations | (11,772 | ) | (8,609 | ) | (7,287 | ) | (10,883 | ) | ||||||||
Net loss | (11,552 | ) | (8,589 | ) | (7,068 | ) | (11,049 | ) | ||||||||
Basic and diluted net loss per common share (1) | (0.56 | ) | (0.41 | ) | (0.24 | ) | (0.38 | ) | ||||||||
Weighted average basic and diluted common shares outstanding | 20,565,499 | 20,751,489 | 29,066,266 | 29,129,000 | ||||||||||||
| 2004 | ||||||||||||||||
| 1st Qtr | 2nd Qtr | 3rd Qtr | 4th Qtr | |||||||||||||
Net revenues | $ | 5,634 | $ | 6,619 | $ | 7,157 | $ | 7,615 | ||||||||
Cost of goods sold | 1,789 | 1,996 | 1,979 | 2,384 | ||||||||||||
Research and development expenses | 2,370 | 4,814 | 4,899 | 3,910 | ||||||||||||
Selling, general and administrative expenses | 7,422 | 7,530 | 7,831 | 8,427 | ||||||||||||
Loss from operations | (5,947 | ) | (7,721 | ) | (7,552 | ) | (7,106 | ) | ||||||||
Net loss | (6,410 | ) | (7,720 | ) | (7,471 | ) | (6,917 | ) | ||||||||
Dividends and accretion of redeemable convertible preferred stock | (169 | ) | (342 | ) | (57 | ) | — | |||||||||
Net loss applicable to common stockholders | (6,579 | ) | (8,062 | ) | (7,528 | ) | (6,917 | ) | ||||||||
Basic and diluted net loss per common share (1) | (7.08 | ) | (8.56 | ) | (0.51 | ) | (0.34 | ) | ||||||||
Weighted average basic and diluted common shares outstanding | 929,906 | 942,131 | 14,856,088 | 20,533,331 | ||||||||||||
| (1) | The sum of the quarterly loss per share amounts may differ from the full year amount due to changes in the number of shares outstanding during the year. |
111
Table of Contents
| ITEM 9. | Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
None.
| ITEM 9A. | Controls and Procedures |
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures during the period covered by this Report and as of the end of the period covered by this Report. Disclosure controls and procedures are controls and procedures designed to ensure that the information required to be disclosed by us in reports filed under the Securities Exchange Act of 1934 is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in reports filed under the Securities Exchange Act of 1934 is accumulated and communicated to our management, including the Chief Executive Officer and Chief Financial Officer, as appropriate to allow timely decisions regarding disclosure. Based on their evaluation, and in particular the report of our General Counsel of the results of the report of the internal investigation conducted by the Audit and Compliance Committee of our Board of Directors, or the Audit Committee, described below, the Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures during the period covered by this Report had, at times, been ineffective and there had been, at times, material weaknesses in internal control over financial reporting as a result of non-compliance by certain employees, as described below, but that such disclosure controls and procedures were effective as of December 31, 2005. In addition, we believe that by taking the actions described below, we had, as of June 30, 2005 and for the remainder of the period covered by this Report, remediated those material weaknesses in our internal control over financial reporting that were the result of the non-compliance by certain employees that we had identified.
Based upon those events described below that occurred prior to and during the period covered by this Report, we took the following actions during the period covered by this Report to improve our disclosure controls and procedures and internal controls over financial reporting:
| • | On April 14, 2005, Gerri Henwood, our Chairman and Chief Executive Officer and Interim President, resigned her position as Chairman. Ms. Henwood continues to serve as our Chief Executive Officer and Interim President and as a member of our Board of Directors. |
| • | On April 14, 2005, one of our independent directors and our Vice Chairman and member of our Audit Committee, Rolf A. Classon, was appointed to the position of Chairman of our Board of Directors. As such, Mr. Classon no longer serves as our Vice Chairman. In his position as Chairman, Mr. Classon provides enhanced oversight of the: |
| • | implementation and operation of our disclosure controls and procedures; and |
| • | management of our operational affairs including, but not limited to, sales and marketing, research and development, government regulation, manufacturing and employee matters, and monitor and review the implementation of policies and procedures established by the Board within such areas. |
| • | As Chairman, Mr. Classon: |
| • | presides over any meeting of our Board of Directors that Mr. Classon attends, and when so acting has all the powers of and be subject to all the restrictions upon our Chairman; |
| • | provides ongoing advice and counsel to our Chief Executive Officer and otherwise assist our Chief Executive Officer in the conduct of the responsibilities of our Chief Executive Officer; |
| • | provides active oversight of our management and operational affairs, including, but not limited to, sales and marketing, research and development, government regulation, manufacturing and employee matters, and monitor and review the implementation of policies and procedures established by the Board within such areas; |
112
Table of Contents
| • | provides our Board of Directors with periodic updates of oversight activities; |
| • | sets the agenda for each meeting of our Board of Directors and organize preparation of each such meeting; |
| • | presides over any meeting of outside directors that may be held after meetings of our Board of Directors that is attended by Mr. Classon, the purpose of which may be to review the effectiveness of management, collect feedback to management and relay any such feedback to management; and |
| • | such other duties, responsibilities and authority as may be prescribed, from time to time, by resolution of the Board of Directors. |
| • | We appointed our General Counsel as the chairman of our Disclosure Committee so that our internal disclosure procedures are overseen by management personnel that are familiar with the disclosure standards that are applicable to us. |
| • | We adopted a plan to substantially reduce our business relationship with Prosit LLC. |
| • | We instituted a specific related-party transaction approval process that requires our General Counsel to approve and countersign any purchase order with a related party. We have designated an employee to monitor all known related-parties on a quarterly basis to determine if new related-party transactions have been initiated. In addition, we have assigned a member of our accounting department having knowledge of the relevant disclosure standards the responsibility for canvassing our directors, significant stockholders and relevant members of management on a regular basis in order to ensure that any new related-party transactions are identified, reviewed, and reported where appropriate and monitoring transactions on an ongoing basis to identify any related-party transactions. This individual will provide a quarterly report of the related-party review and any related-party activity to the Audit Committee. |
| • | We have instituted a management training program focused on educating our management personnel on the types of issues that require disclosure under the rules and regulations of the SEC. The program is designed to promote effective communication with our General Counsel when our officers consider amending or modifying any of our material agreements and regarding disclosure issues generally. |
We believe that the efforts described above addressed the deficiencies described below that affected our internal disclosure controls at times during the period covered by this Report and during prior periods. Accordingly, our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures as of the end of the period covered by this Report, and the Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures as of the end of the period covered by this Report were effective. In addition, we believe that the efforts described above remediated those material weaknesses in our internal control over financial reporting that were the result of the non-compliance by certain employees described below that we had identified during the period covered by this Report. A controls system cannot provide absolute assurance that the objectives of the controls system are met, and no evaluation of controls can provide absolute assurance that all controls issues and instances of fraud, if any, within a company have been detected.
On March 16, 2005, members of our management alerted our Board of Directors to the fact that:
| • | there was an amendment to the severance provisions of our former President and Chief Operating Officer’s employment agreement that had not been previously disclosed (the Employment Agreement Amendment); and |
| • | That we had engaged in various related-party transactions involving Prosit LLC, a printing company owned by the brother of our Chief Executive Officer and Interim President (the Related Party Transactions), that had not been pre-approved by the Audit Committee. |
113
Table of Contents
As a result of disclosures by members of our management, on March 21, 2005, the Audit Committee commenced an internal investigation regarding the Employment Agreement Amendment, the Related Party Transactions, and our internal disclosure controls and procedures to determine if there were any other transactions that we had not reported. The Audit Committee retained our outside counsel to assist it with the internal investigation. The Audit Committee’s report of the internal investigation concluded that:
| • | In August 2004, our Chief Executive Officer and Interim President and then President and Chief Operating Officer entered into the Employment Agreement Amendment. They failed to (a) advise our then General Counsel or any other member of management that the Employment Agreement Amendment had been executed or (b) receive the formal approval of our Board of Directors for the Employment Agreement Amendment. The Employment Agreement Amendment provides that the President and Chief Operating Officer would receive 12 months of salary, rather than nine months, as severance in certain situations. As a result, we did not provide correct disclosure regarding our severance obligations to our then President and Chief Operating Officer. |
| • | From August 2004 though April 2005 we engaged in the Related Party Transactions without obtaining the prior approval of the Audit Committee. This violated our Code of Conduct, which, among other things, requires pre-clearance of related party transactions. |
| • | We previously disclosed that an investment partnership, HH Capital Partners, controlled by our Chief Executive Officer and Interim President and previous General Counsel held an equity investment in Prosit LLC that was terminated in 2001. We were not advised that at the time this equity interest was terminated, it was converted into a promissory note in principal amount of $240,000, of which $180,000 was outstanding as of December 31, 2004, bearing an annual interest rate of 4% (the Prosit Note). Interest earned on the Prosit Note was to be paid in monthly installments and principal was to be repaid in six annual installments of $40,000. Because our disclosure policies were not followed, our ability to report the Prosit Loan on a timely basis was jeopardized. |
| • | During February of 2005, our Chief Executive Officer and Interim President executed an amended and restated employment agreement that would have amended the terms of employment of one of our executive officers without receiving the formal approval of our Board of Directors. The amended and restated employment agreement was not signed by the executive officer, and the executive officer has acknowledged that the amended and restated employment agreement is not enforceable against the Company. |
The Audit Committee discussed the results of the internal investigation with our former independent registered public accounting firm. Because of certain failures, including those enumerated above, this firm determined, in connection with its audit of the Company’s consolidated financial statements as of December 31, 2004 and for the year then ended, that material weaknesses in internal control over financial reporting existed.
Changes in Internal Controls
No change in our internal control over financial reporting occurred during our fiscal quarter ended December 31, 2005 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
To remediate the material weaknesses in our internal control over financial reporting described in “Item 9A.Controls and Procedures” above, subsequent to December 31, 2004 we implemented the changes described therein.
114
Table of Contents
| ITEM 9B. | Other Information |
On March 31, 2006, the Company entered into an agreement with KPMG LLP, or KPMG, its former independent registered public accounting firm, pursuant to which the Company agreed to indemnify and hold KPMG harmless against and from any and all legal costs and expenses incurred by KPMG in its successful defense of any legal action or proceeding that may arise as a result of its consent to the incorporation by reference of its report on the Company’s consolidated financial statements incorporated by reference in the Registration Statements No. 333-127289 on Form S-3 and No. 333-117595 on Form S-8 of the Company, which report appears in the December 31, 2005 Annual Report on Form 10-K of the Company. The agreement also provides that KPMG shall not be indemnified, and shall refund to the Company, any amounts paid to KPMG pursuant to the indemnification in the event there is court adjudication that KPMG is guilty of professional malpractice, or in the event that KPMG becomes liable for any part of the plaintiff’s damages by virtue of settlement.
115
Table of Contents
| ITEM 10. | Directors and Executive Officers of the Registrant |
The information required by this item is incorporated herein by reference to the sections captioned “Election of Directors,” “Executive Officers,” and “Section 16(a) Beneficial Ownership Reporting Compliance” in our definitive Proxy Statement relating to our 2006 Annual Meeting of Stockholders.
We have adopted a Code of Conduct that applies to all of our directors, officers and employees. Our Code of Conduct contains provisions that satisfy the standards for a “code of ethics” set forth in Item 406 of Regulation S-K of the rules and regulations of the Securities and Exchange Commission. Our Code of Conduct also contains a special Code of Ethics that is applicable to our Chief Executive Officer and our senior financial officers. We have posted our Code of Conduct on our Internet website, the address of which is www.auxilium.com. Our Code of Conduct is available under the heading “Investor Relations — Corporate Governance” on our Internet website.
To the extent that we amend any provision of our Code of Conduct or grant a waiver from any provision of our Code of Conduct that is applicable to any of our directors or our principal executive officer, principal financial officer, principal accounting officer or controller or persons performing similar functions, we intend to satisfy our disclosure obligations under Item 5.05 of Form 8-K by posting such information on our Internet website under the heading “Investor Relations — Corporate Governance”.
| ITEM 11. | Executive Compensation |
The information required by this item is incorporated herein by reference to the section captioned “Executive Compensation” in our definitive Proxy Statement relating to our 2006 Annual Meeting of Stockholders.
| ITEM 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
The information required by this item is incorporated herein by reference to the sections captioned “Security Ownership of Certain Beneficial Owners and Management” and “Executive Compensation — Equity Compensation Plan Information” in our definitive Proxy Statement relating to our 2006 Annual Meeting of Stockholders.
| ITEM 13. | Certain Relationships and Related Transactions |
The information required by this item is incorporated herein by reference to the section captioned “Certain Relationships and Related Transactions” in our definitive Proxy Statement relating to our 2006 Annual Meeting of Stockholders.
| ITEM 14. | Principal Accountant Fees and Services |
The information required by this item is incorporated herein by reference to the section captioned “Ratification of Selection of Independent Registered Public Accounting Firm” in our definitive Proxy Statement relating to our 2006 Annual Meeting of Stockholders.
116
Table of Contents
| ITEM 15. | Exhibits, Financial Statement Schedules |
(a) The following documents are filed as part of this Report:
(1)Consolidated Financial Statements
See Index to Consolidated Financial Statements on page 74.
(2)Financial Statement Schedules
All schedules to the consolidated financial statements are omitted as the required information is either inapplicable or presented in the consolidated financial statements.
(3)Exhibits
The information required by this Item is set forth in the Exhibit Index hereto which is incorporated herein by reference.
(b) Exhibits
The information required by this Item is set forth in the Exhibit Index hereto which is incorporated herein by reference.
117
Table of Contents
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| AUXILIUM PHARMACEUTICALS, INC. | ||
| By: | /s/ GERRI A. HENWOOD | |
| Gerri A. Henwood | ||
Chief Executive Officer and Interim President | ||
Date: March 31, 2006
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Gerri A. Henwood and James E. Fickenscher as true and lawful attorneys-in-fact and agents with full power of substitution and resubstitution, for him or her and in his or her name, place and stead, in any and all capacities to sign this Report filed herewith and any or all amendments to said Report, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission granting unto said attorneys-in-fact and agents the full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about the foregoing, as to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them, or his or her substitute, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this Report has been signed by the following persons on behalf of the Registrant in the capacities and on the dates indicated.
Signature | Title | Date | ||
/s/ GERRI A. HENWOOD Gerri A. Henwood | Chief Executive Officer, Interim President (Principal Executive Officer) and Director | March 31, 2006 | ||
/s/ JAMES E. FICKENSCHER James E. Fickenscher | Chief Financial Officer (Principal Financial and Accounting Officer) | March 31, 2006 | ||
/s/ ROLF A. CLASSON Rolf A. Classon | Chairman | March 31, 2006 | ||
/s/ AL ALTOMARI Al Altomari | Director | March 31, 2006 | ||
/s/ EDWIN A. BESCHERER, JR. Edwin A. Bescherer, Jr. | Director | March 31, 2006 | ||
/s/ PHILIPPE O. CHAMBON Philippe O. Chambon | Director | March 31, 2006 | ||
/s/ WINSTON J. CHURCHILL Winston J. Churchill | Director | March 31, 2006 | ||
/s/ OLIVER S. FETZER Oliver S. Fetzer | Director | March 31, 2006 | ||
/s/ DENNIS J. PURCELL Dennis J. Purcell | Director | March 31, 2006 | ||
118
Table of Contents
Exhibit No. | Description | |
| 3.1 | Fifth Restated Certificate of Incorporation of the Registrant (filed as Exhibit 3.1 to the Registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2004, File No. 000-50855, and incorporated by reference herein). | |
| 3.2 | Amended and Restated Bylaws of the Registrant (filed as Exhibit 3.2 to the Registrant’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2004, File No. 000-50855, and incorporated by reference herein). | |
| 4 | Third Amended and Restated Investors Rights Agreement, dated October 31, 2003, by and among the Registrant and parties listed therein (filed as Exhibit 4.2 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.1* | License Agreement, dated May 31, 2000, as amended, between Bentley Pharmaceuticals, Inc. and the Registrant (filed as Exhibit 10.1 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.2* | License Agreement, dated May 31, 2001, as amended, between Bentley Pharmaceuticals, Inc. and the Registrant (filed as Exhibit 10.2 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.3* | License Agreement, dated June 20, 2003, between Formulation Technologies L.L.C. d/b/a PharmaForm and the Registrant (filed as Exhibit 10.3 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.4 | Amendment, dated April 19, 2004, to License Agreement between Formulation Technologies, L.L.C. d/b/a PharmaForm and the Registrant, dated June 20, 2003 (filed as Exhibit 10.20 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.5 | Amendment No. 2, dated June 17, 2004, to License Agreement between Formulation Technologies L.L.C. d/b/a PharmaForm and the Registrant, dated June 20, 2003 (filed as Exhibit 10.25 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.6* | Research and Development Agreement, dated June 20, 2003, between Formulation Technologies L.L.C. d/b/a PharmaForm and the Registrant (filed as Exhibit 10.8 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.7* | Amendment, dated February 8, 2005, to Research and Development Agreement between Formulation Technologies L.L.C. d/b/a PharmaForm and the Registrant, dated June 20, 2003 (filed as Exhibit 10.7 to the Registrant’s Annual Report on Form 10-K for the years ended December 31, 2004 and incorporated herein by reference). | |
| 10.8 | Research and Development Agreement, dated February 24, 2005, by and between Cobra Biologics Ltd and the Registrant (filed as Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q filed on August 12, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.9* | License Agreement, dated February 8, 2005, between Formulation Technologies, L.L.C. d/b/a PharmaForm and the Registrant (filed as Exhibit 10.8 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2004, File No. 000-50855, and incorporated by reference herein). | |
| 10.10* | Development and License Agreement, dated June 3, 2004, by and between BioSpecifics Technologies Corp. and the Registrant (filed as Exhibit 10.19 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
119
Table of Contents
Exhibit No. | Description | |
| 10.11* | Amendment No. 1, dated May 10, 2005, to the Development and License Agreement by and between BioSpecifics Technologies Corp. and the Registrant, dated June 3, 2004 (filed as Exhibit 10.4 to the Registrant’s Quarterly Report on Form 10-Q filed on August 12, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.12* | Distribution Agreement, dated December 29, 2003, between Bayer Inc. and the Registrant (filed as Exhibit 10.4 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.13* | License and Distribution Agreement, dated March 26, 2004, between Ipsen Farmaceutica B.V. and the Registrant (filed as Exhibit 10.7 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.14* | Manufacturing Agreement, dated April 23, 2002, between DPT Laboratories, Ltd. and Registrant (filed as Exhibit 10.5 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.15* | First Amendment, dated May 28, 2002 to Manufacturing Agreement between DPT Laboratories, Ltd. and the Registrant, dated April 23, 2002 (filed as Exhibit 10.6 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.16* | Third Amendment, dated September 14, 2005, to Manufacturing Agreement between DPT Laboratories, Ltd. and the Registrant, dated April 23, 2002 (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on September 20, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.17* | Process Development and cGMP Manufacture of Phase III CTM Agreement, dated June 28, 2005, by and between Cobra Biomanufacturing Plc and the Registrant (filed as Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q filed on August 12, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.18 | Gross Lease, dated February 28, 2000, as amended, between Brandywine Norriton, L.P. and the Registrant (filed as Exhibit 10.13 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.19 | Agreement of Lease, dated December 31, 2004, between Liberty Property Limited Partnership, as landlord, and the Registrant, as tenant (filed as Exhibit 99.1 to the Registrant’s Current Report on Form 8-K filed on January 6, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.20 | Promissory Note, dated June 13, 2003, made by the Registrant in favor of General Electric Capital Corporation (filed as Exhibit 10.11 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.21 | Master Security Agreement, dated June 16, 2002, between General Electric Capital Corporation and the Registrant (filed as Exhibit 10.10 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.22* | Loan and Security Agreement, dated March 11, 2004, between Comerica Bank and the Registrant (filed as Exhibit 10.9 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.23* | Commercial Outsourcing Services Agreement, dated November 13, 2002, between Integrated Commercialization Solutions Inc. and the Registrant (filed as Exhibit 10.12 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
120
Table of Contents
Exhibit No. | Description | |
| 10.24* | Co-Promotion Agreement, dated April 11, 2005, between Oscient Pharmaceuticals Corp. and the Registrant (filed as Exhibit 10 to the Registrant’s Quarterly Report on Form 10-Q filed on May 6, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.25 | Form of Securities Purchase Agreement, dated June 28, 2005 (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on July 1, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.26 | Form of Amendment to Securities Purchase Agreement, dated December 30, 2005 (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on January 6, 2006, File No. 000-50855, and incorporated by reference herein). | |
| 10.27# | Registrant’s 2004 Equity Compensation Plan (filed as Exhibit 10.23 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.28# | Form of Non-qualified Stock Option Agreement for awards under the Registrant’s 2004 Equity Compensation Plan, including provision for immediate vesting upon a change of control of the Registrant (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K filed on March 22, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.29# | Form of Non-qualified Stock Option Agreement for awards under the Registrant’s 2004 Equity Compensation Plan (filed as Exhibit 10.2 to the Registrant’s Current Report on Form 8-K filed on March 22, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.30# | Form of Incentive Stock Option Agreement for awards under the Registrant’s 2004 Equity Compensation Plan, including provision for immediate vesting upon a change of control of the Registrant (filed as Exhibit 10.3 to the Registrant’s Current Report on Form 8-K filed on March 22, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.31# | Form of Incentive Stock Option Agreement for awards under the Registrant’s 2004 Equity Compensation Plan (filed as Exhibit 10.4 to the Registrant’s Current Report on Form 8-K filed on March 22, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.32# | Form of Amendment to Stock Option Grant Agreement for the acceleration of vesting of “out-of-the-money” stock options (filed as filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on September 26, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.33# | Registrant’s 2004 Employee Stock Purchase Plan (filed as Exhibit 10.24 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.34# | Employment Agreement, dated May 18, 2005, between James E. Fickenscher and the Registrant (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on May 20, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.35# | Employment Agreement, dated June 5, 2004, between Geraldine A. Henwood and the Registrant (filed as Exhibit 10.14 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.36# | Employment Agreement, dated June 1, 2004, between Robert S. Whitehead and the Registrant (filed as Exhibit 10.21 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.37# | Employment Agreement Addendum #1 between Robert S. Whitehead and the Registrant (August 2004) (filed as Exhibit 10.28 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2004, File No. 000-50855, and incorporate by reference herein). | |
121
Table of Contents
Exhibit No. | Description | |
| 10.38# | Separation Agreement and General Release, dated June 2, 2005, between Robert S. Whitehead and the Registrant (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on June 3, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.39# | Employment Agreement, dated April 19, 2004, between Cornelius H. Lansing II and the Registrant (filed as Exhibit 10.16 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.40# | Amended and Restated Employment Agreement, dated May 18, 2005, between Cornelius H. Lansing II and the Registrant (filed as Exhibit 10 to the Registrant’s Current Report on Form 8-K filed on May 26, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.41# | Separation of Employment Agreement and General Release, dated April 13, 2005, between Terri B. Sebree and the Registrant (filed as Exhibit 10.30 to the Registrant’s Annual Report on Form 10-K for the year ended December 31, 2004, File No. 000-50855, and incorporate by reference herein). | |
| 10.42# | Employment Agreement, dated April 19, 2004, between Dr. Jyrki Mattila and the Registrant (filed as Exhibit 10.18 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 10.43# | Employment Agreement, dated March 22, 2005, between Jennifer Evans Stacey and the Registrant (filed as Exhibit 10.5 to the Registrant’s Current Report on Form 8-K filed on March 22, 2005, File No. 000-50855, and incorporated by reference herein). | |
| 10.44 | Indemnification Agreement, dated March 31, 2006, between KPMG LLP and the Registrant. | |
| 21 | Subsidiaries of the Registrant (filed as Exhibit 21.1 to the Registrant’s Registration Statement on Form S-1, Registration No. 333-114685, and incorporated by reference herein). | |
| 23.1 | Consent of PricewaterhouseCoopers LLP. | |
| 23.2 | Consent of KPMG LLP. | |
| 24 | Powers of Attorney (included on signature page). | |
| 31.1 | Certification of Gerri A. Henwood, the Registrant’s Principal Executive Officer, required by Rule 13a-14(a) or Rule 15d-14(a). | |
| 31.2 | Certification of James E. Fickenscher, the Registrant’s Principal Financial Officer, required by Rule 13a-14(a) or Rule 15d-14(a). | |
| 32 | Certification of Gerri A. Henwood, the Registrant’s Principal Executive Officer, and James E. Fickenscher, the Registrant’s Principal Financial Officer, required by Rule 13a-14(b) or Rule 15d-14(b) and Section 1350 of Chapter 63 of Title 18 of the U.S. Code (18 U.S.C. 1350). | |
| * | Certain information in this exhibit has been omitted pursuant to an Order Granting Confidential Treatment issued by the SEC. |
| # | Indicates management contract or compensatory plan or arrangement. |
122