 (NASDAQ: AUXL) December 2006 Exhibit 99.1 |
 2 (NASDAQ: AUXL) Safe Harbor Statement Safe Harbor Statement We will make various remarks during this presentation that constitute “forward-looking statements” for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995, including statements regarding future Testim market share, prescriptions and sales growth and factors that may drive such growth; size and growth potential of the testosterone replacement therapy market and the gel segment thereof and factors that may drive such growth; the effect of the Company’s expansion of its field force; the pricing, time to market, size of market, growth potential and therapeutic benefits of the Company’s product candidates, including those for the treatment of Dupuytren’s Contracture, Peyronie’s Disease, and Frozen Shoulder Syndrome and overactive bladder; interpretation of market research data; competition within certain markets relevant to the Company’s product candidates; interpretation of clinical results, including the efficacy and tolerability of the Company’s product candidates as well as the reduction of side effects; the timing of the commencement and completion of clinical trials; the timing of manufacturing scale up for the Company’s product candidates; the Company’s ability to manufacture AA4500 at the Company’s Horsham facility; the timing and outcome of our root cause analysis of the AA4500 manufacturing issue, the implementation of our corrective actions and their effect on AA4500; competitive developments affecting the Company’s products and product candidates; the success of the Company’s development activities; the Company’s development and operation goals for fiscal 2006; the ability to fund future operations; and the Company’s expected financial performance during 2006 and financial milestones that it may achieve for 2006, including 2006 net revenues, research and development spending, selling, general and administrative expenses, and net loss. All remarks other than statements of historical facts made during this presentation, including but not limited to, statements regarding future expectations, plans and prospects for the Company, statements regarding forward-looking financial information and other statements containing the words “believe,” “may,” “could,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” and similar expressions, as they relate to the Company, constitute forward- looking statements. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2005 and in the Company’s Quarterly Report on From 10-Q for the period ended September 30, 2006 under the heading “Risk Factors”, which is on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “Investor Relations - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 3 (NASDAQ: AUXL) Fastest Growing TRT product in the U.S. Company Transforming Pipeline Opportunities Poised for Growth + = |
 4 (NASDAQ: AUXL) Auxilium Pharmaceuticals, Inc. Auxilium Pharmaceuticals, Inc. Company Profile (NASDAQ: AUXL) Company Profile (NASDAQ: AUXL) • • Specialty biopharmaceutical company focused on Urologists, Specialty biopharmaceutical company focused on Urologists, Endocrinologists, Orthopedists and certain PCPs Endocrinologists, Orthopedists and certain PCPs • • Founded in 1999 Founded in 1999 • • Headquarters in Malvern, Pa. Headquarters in Malvern, Pa. – – Manufacturing in Horsham, Pa. Manufacturing in Horsham, Pa. – – approximately 290 employees (195 commercial, 25 mfg, 40 R&D) approximately 290 employees (195 commercial, 25 mfg, 40 R&D) • • I.P.O. in July 2004 – I.P.O. in July 2004 – raised $36.5 million raised $36.5 million – – Two offerings in 2005 and 2006 have raised a total of $83.5 million Two offerings in 2005 and 2006 have raised a total of $83.5 million • • Listed on NASDAQ BIOTECH INDEX – Listed on NASDAQ BIOTECH INDEX – November 2006 November 2006 |
 5 (NASDAQ: AUXL) • AA4500 – Lead Development Product – Reported Positive Results from Initial Pivotal Phase III Study in Dupuytren’s Contracture in June 2006 – Tech transfer to Horsham facility completed – Initial engineering batch manufactured in Horsham – Started ex-U.S. Dupuytren’s phase III trial in October – Final U.S. pivotal trial for first indication started November 20 – Suspended dosing in both phase III trials December 7 due to manufacturing issues • Testim® 1% Testosterone Gel – $49 million in revenues through Sept 30 – 64% growth over 2005 – Gel market share – 18.1% at October 2006 vs 14.9% October 2005 – Expansion of sales force to ~150 representatives completed Focused on Value Drivers Focused on Value Drivers |
 6 (NASDAQ: AUXL) Broad Pipeline Maturing in Coming Years Broad Pipeline Maturing in Coming Years Note: Worldwide rights for all products available, except Testim where ex US rights have been licensed. 5 6 Product Testim® gel Approved in U.S. Europe & Canada AA4500 AA4500 AA4500 Transmucosal Film AA4010 Transmucosal Film Transmucosal Film Phase III Phase II Phase I Pre-clinical Late Research Hypogonadism Dupuytren’s Contracture Peyronie’s Disease Frozen Shoulder Syndrome Overactive Bladder Hormones and Urology Pain Market |
 Testim ® 1% Testosterone Gel: Near-Term Growth Driver Executing on Opportunities |
 8 (NASDAQ: AUXL) Testim Testim ® ® is Fastest Growing TRT Gel is Fastest Growing TRT Gel • Proprietary, topical 1% testosterone gel – Once-a-day application – Favorable clinical and commercial profile • Comparative studies support advantages. • BX rated to Androgel • Cost-effective and convenient • Broad prescription coverage • Growing prescriber loyalty |
 9 (NASDAQ: AUXL) Male Hypogonadism (Testosterone Deficiency) Overview Male Hypogonadism (Testosterone Deficiency) Overview • Affects >20% of U.S. males over 50 yrs and causes serious medical conditions – Estimate that about 5% of affected population receives treatment • Diagnosis increasing through education and awareness Signs/symptoms – Brain Function Libido and erections Energy and vigor Cognitive function Sleep quality Irritability and depressed mood Signs/symptoms – Body Composition Muscle mass and strength Bone mineral density Male hair density and skin thickness Fat mass – abdominal obesity |
 10 (NASDAQ: AUXL) Clinical Results Show Testim Clinical Results Show Testim ® ® Changes Body Composition Improves Sexual Activity • Sexual Activity 59% • Statistically significant increases in sexual desire • Lean Body Mass 4.8 lbs • Fat Mass 4.0 lbs • Bone Mineral Density 2.6 % 12-month study Placebo-controlled study, 90-day results Source: Auxilium studies published in Journal of Clinical Metabolism & Review of Urology |
       11 (NASDAQ: AUXL) Patient Benefits are Clinically Proven Patient Benefits are Clinically Proven Note: Adjusted geometric means (CV b %) of a single-dose (50 mg testosterone), randomized, complete crossover study of 29 hypogonadal men. Total Testosterone 2,000 3,000 4,000 5,000 6,000 Mean AUC 0-24 (ng*h/dL) Testim AndroGel Free Testosterone 0 50 100 150 200 250 Mean AUC 0-24 (ng*h/dL) Testim AndroGel • 14 clinical studies involving approx. 1,800 patients – largest placebo-controlled study ever conducted • Clinical trial of Testim ® vs. AndroGel ® – Testim provides 30% higher testosterone absorption (p<0.001) |
 12 (NASDAQ: AUXL) Gels Fastest Growing TRT Segment Gels Fastest Growing TRT Segment Source: IMS data ($ in millions) 0 100 200 300 400 500 Gel Patch Oral Injectables $399 $118 $77 $59 $49 $210 $302 1997 1998 1999 2000 2001 2002 2003 $459 2004 $497 $198 $288 $340 $117 $35 2005 $381 |
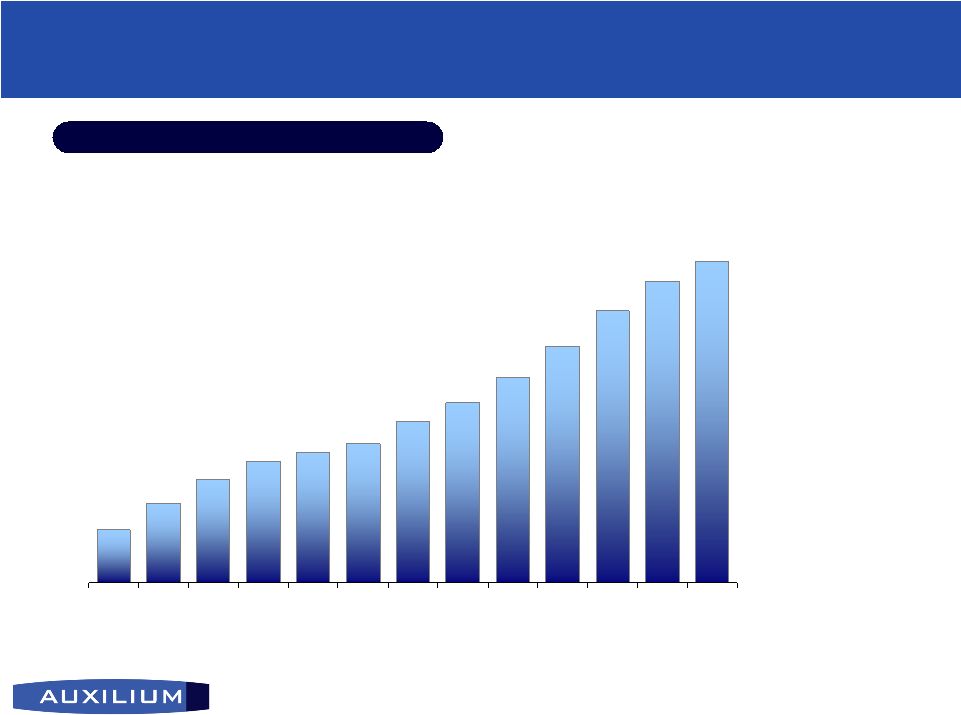 13 (NASDAQ: AUXL) Testim ® ® quarterly net revenue ($ in thousands) Established Track Record of Consistent Growth Established Track Record of Consistent Growth Q3 2006 Y/Y Growth: +57% Q3 2006 Y/Y Growth: +57% $2,893 $4,307 $5,634 $6,619 $7,157 $7,615 $8,797 $9,839 $11,230 $12,938 $14,893 $16,471 $17,640 Q303 Q403 Q104 Q204 Q304 Q404 Q105 Q205 Q305 Q405 Q106 Q206 Q306 |
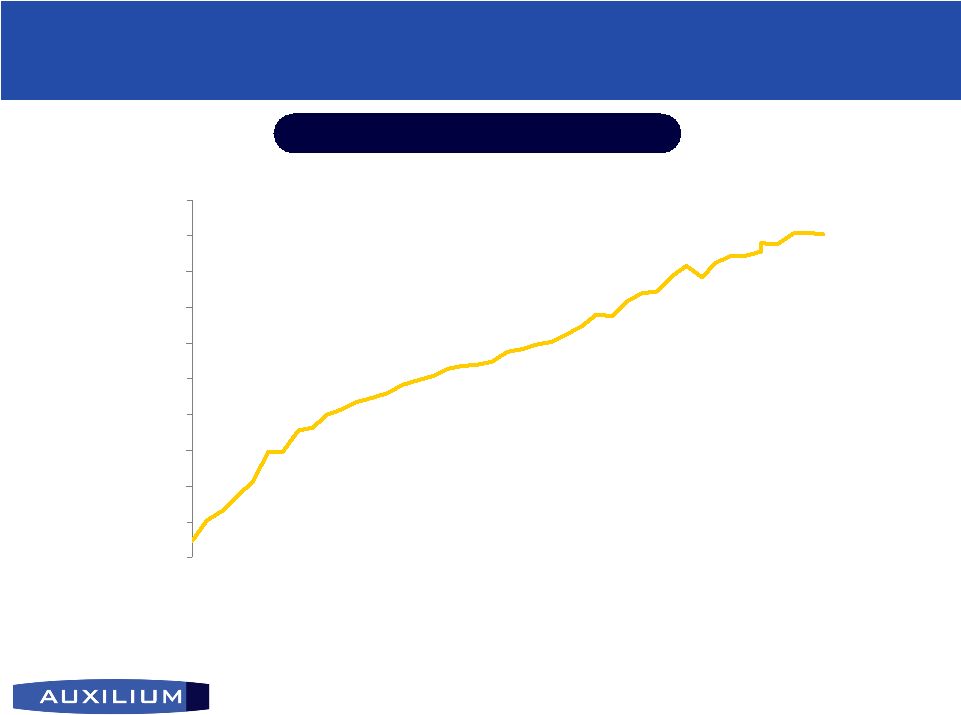 14 (NASDAQ: AUXL) Testim Testim ® ® Driving Growth in Market Driving Growth in Market Source: IMS data Testim ® Share of TRx of Gel Market Strong market share with high-prescribing urologists; 31.6% gel market share in October 2006 1.0% 18.1% 0% 2% 4% 6% 8% 10% 12% 14% 16% 18% 20% Mar-03 Oct-06 |
 15 (NASDAQ: AUXL) • Paragraph IV challenges to AndroGel Settled • Testim ® patent coverage ��� Current Testim ® patent expires in 2008 – New patent filed in April 2003 • USPTO expected to act in first half 2007 • Will expire in 2023 if granted – New patent issued in Canada; expires 2023 Generic Uncertainty Mitigated Generic Uncertainty Mitigated |
 16 (NASDAQ: AUXL) Positioned for Continued Growth Positioned for Continued Growth Market Drivers Product Drivers • Male sexual health awareness • ED promotional impact • Increased screening and testing – Normal “T” enhances outcome in ED patients – Other health parameters improved with normal “T” • Diabetic / Metabolic Syndrome awareness • Increase awareness through KOLs • Leverage clinical data • Expanded sales force • Leverage third-party payer coverage |
 Pipeline: “First Mover” Products Advancing a Broad Portfolio of Opportunities |
 18 (NASDAQ: AUXL) AA4500 – AA4500 – Company Transforming Opportunity Company Transforming Opportunity • We believe that AA4500 has significant commercial potential – First-mover value – No approved non-surgical treatments – Worldwide rights offer options to build company or generate cash • 3 indications currently licensed from BioSpecifics Technologies: – Dupuytren’s Contracture, Peyronie’s Disease and Frozen Shoulder Syndrome – Options for in-licensing additional indications |
 19 (NASDAQ: AUXL) AA4500 for Dupuytren’s AA4500 for Dupuytren’s Contracture Contracture • Patented through 2014; Orphan Drug Exclusivity for 7 years post approval I.P. Position • Surgery is only approved treatment - expensive and characterized by numerous complications Current treatment • Phase III clinical studies temporarily suspended due to manufacturing issues Clinical status • Caucasian men and women; prevalence highest among people of northern European descent Target population • Lyophilized Collagenase injectable; up to 0.25 ml/dose • Affected fingers become permanently flexed Product Description Disease description Source: Badalamente, M. et al. J. Hand Surgery, September 2002 |
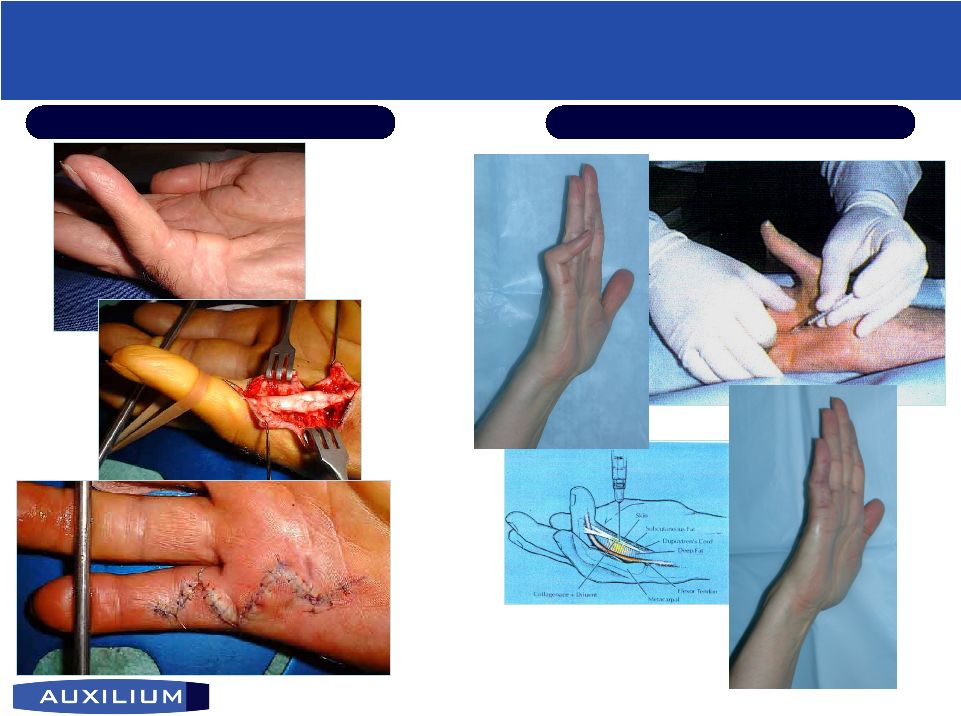 20 (NASDAQ: AUXL) Revolutionizing Treatment for Dupuytren’s Contracture Revolutionizing Treatment for Dupuytren’s Contracture Current Treatment AA4500 Treatment |
 21 (NASDAQ: AUXL) MP joint PIP joint Clinical Results from AA4500 Pivotal Study Clinical Results from AA4500 Pivotal Study • 91% success rate for primary endpoint of less than five degrees of contracture after up to three injections; no placebo response (p<0.001) • After single injection, 70% of subjects achieved therapeutic success; no placebo response (p<0.001) Source: Auxilium data • No serious adverse events reported. Most commonly reported adverse events were pain and swelling (edema) of the hand at injection site, and post-injection transient, self-limiting swelling in lymphnode area of armpit • Results consistent with those from Phase II study published in The Journal of Hand Surgery (2002:27A:788-798) |
 22 (NASDAQ: AUXL) Dupuytren’s Clinical plan Dupuytren’s Clinical plan • US Pivotal Phase III trial: – Over 200 patients in up to 15 sites – Double blind placebo controlled, randomized in 2:1 ratio of active to placebo – Primary endpoint is reduction in contracture to within 0 - 5 o of normal • Ex-US placebo controlled study: – Approximately 50 patients in several countries, following protocol similar to US study • Open label studies to generate additional safety data |
 23 (NASDAQ: AUXL) Market Research Performed – Market Research Performed – Dupuytren’s Dupuytren’s • Multiple prevalence data sets proved to be highly variable in numbers of patients • Two rounds of quantitative primary research have been commissioned by Auxilium on AA4500 for Dupuytren’s Contracture • A combined 571 Orthopedic Surgeons have been interviewed to estimate market size (401 in US; 170 in Europe) and 391 have given feedback on potential usage of AA4500 (221 US; 170 Europe) • Second study (n=444 total & 340 in depth) was designed to provide 95% confidence level and 7.5% margin of error |
 24 (NASDAQ: AUXL) Patients Seeking Treatment ~1,030,000 No Prior Surgery ~540,000 Had Surgery Previously ~490,000 Watchful Waiting ~270,000 Medical/ Surgical Treatment ~270,000 Medical/ Surgical Treatment ~125,000 Watchful Wait/ In Follow Up ~365,000 AA4500 Potential Candidates ~240,000 240,000 Dupuytren’s Candidates In US & Europe 240,000 Dupuytren’s Candidates In US & Europe Annually Annually Source: Auxilium research and analysis ~150,000 in US ~ 90,000 in Europe |
 25 (NASDAQ: AUXL) We believe: • There is a pent-up demand for non-surgical therapy • Surgeons and patients prefer non-invasive treatment • Each patient on average has 2.2 joints which will require treatment; 49% have contractures in both hands • Early data indicates on average 1.4 vials of AA4500 required per joint • AA4500 should command a premium price Dupuytren’s Commercial Opportunity Dupuytren’s Commercial Opportunity Source: Dhindsa, S. et al J Clin Endocrinol Metab 2004; Dobs AS Bailleres Clin Endocrinol Metal 1998; Bodie, J. J. Urol. et al 2003 |
 26 (NASDAQ: AUXL) AA4500 for Peyronie’s Disease AA4500 for Peyronie’s Disease • Patented through 2019 • Orphan Drug exclusivity for 7 years post approval I.P. Position • Treated by urologists • Lacks good treatment options – PABA, Vitamin E, Verapamil ineffective – Surgery may shorten penis; cause scarring • Unmet need for non-surgical treatment Current treatment • Phase II data showed efficacy and tolerability • Local effects / tolerability pre-clinical study outside of plaque requested by FDA; expected to start by year end. • Phase IIb expected to start in 2007 Clinical status • Affects men predominantly over 50 Target population • Plaque or hard scar area on the penis • Deformity of the penis during erection precludes intercourse Disease description Source: Mulhall, J. et al, June 2004 |
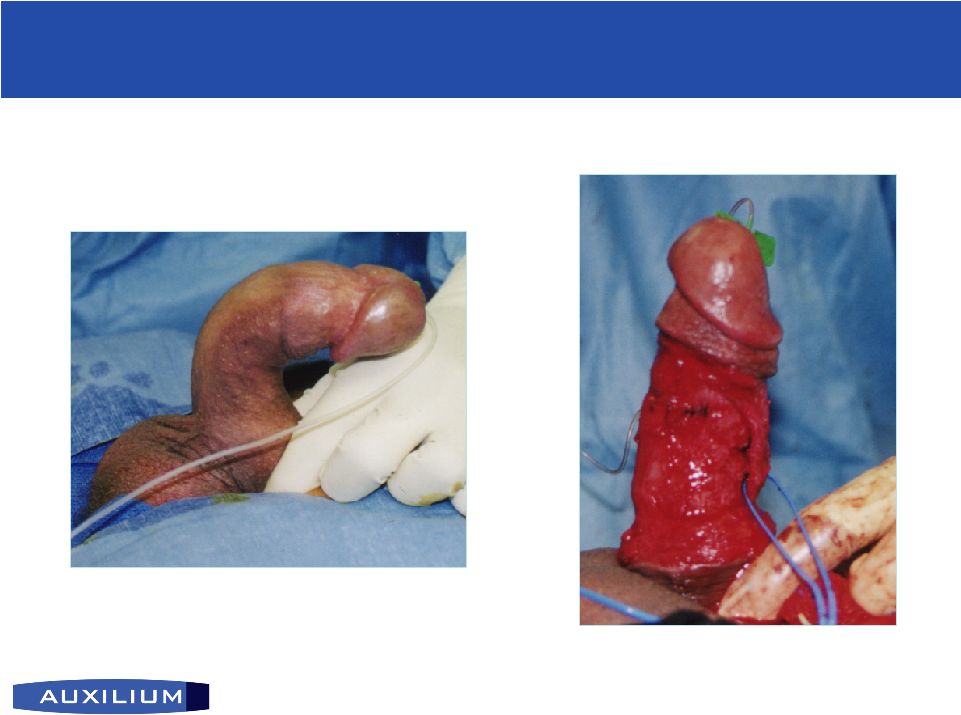 27 (NASDAQ: AUXL) Erect penis with Peyronie’s Peyronie’s Disease and Current Treatment Peyronie’s Disease and Current Treatment Surgical Procedure |
 28 (NASDAQ: AUXL) Market Research Performed – Market Research Performed – Peyronie’s Peyronie’s • Two rounds of quantitative primary research have been commissioned by Auxilium on AA4500 for Peyronie’s Contracture • A combined 575 Urologists have been interviewed to estimate market size (415 in US; 160 in Europe) and 383 have given feedback on potential usage of AA4500 (223 US; 160 Europe) • Second study (n=472 total & 333 in depth) was designed to provide 95% confidence level and 7.5% margin of error |
 29 (NASDAQ: AUXL) Patients Seeking Treatment ~475,000 Surgery Not Recommended ~365,000 Watchful Waiting ~160,000 Medical or Intralesional Therapy ~205,000 Surgery Recommended ~110,000 AA4500 Potential Candidates ~210,000 210,000 Peyronie’s Candidates In US and Europe 210,000 Peyronie’s Candidates In US and Europe Annually Annually Source: Auxilium research ~135,000 in US ~ 75,000 in Europe |
 30 (NASDAQ: AUXL) • Highly motivated patients • Phase IIa data used up to 9 injections of AA4500; Phase IIb will determine the dosing regimen to be used in Phase III • We expect AA4500 should command a premium price Commercial Potential Also High for Peyronie’s Commercial Potential Also High for Peyronie’s |
 31 (NASDAQ: AUXL) AA4500 for Frozen Shoulder Syndrome AA4500 for Frozen Shoulder Syndrome • Arthroscopic procedure or surgery Current treatment • Phase IIa complete • Data showed efficacy and tolerability • Additional studies needed to assess optimal dose and dosing regimen Clinical status • Estimated 3% of people develop frozen shoulder syndrome over their lifetime – Higher incidence among patients with diabetes (10- 20%) vs general population (2-5%) – Higher incidence among patients with insulin- dependent diabetes (36%), with increased frequency of bilateral shoulder involvement • Most common in patients between 45 – 70 yrs Target population • Diminished shoulder motion, characterized by restriction in active and passive range of motion Disease description Source: Pearsall, A. Adhesive capsulitis. eMedicine. 2005 http://www.emedicine.com/pmr/topics372.htm |
 32 (NASDAQ: AUXL) • Taking Control of AA4500 API Manufacturing – 1 st engineering batch produced end of October • Technology transfer from Cobra complete • Capacity to produce all active ingredient required for several years • Renovations should be complete for inclusion in BLA – lease includes allowance for renovations • Laboratories to support production and future product development Horsham Facility a Strategic Asset Horsham Facility a Strategic Asset |
 33 (NASDAQ: AUXL) Transmucosal Film Technology Transmucosal Film Technology • Rights to utilize for products that contain hormones, treat urologic disorders and eight selected analgesic compounds for pain management, including acute and chronic pain • Developing product for treatment of overactive bladder – Looking to partner overactive bladder program |
 34 (NASDAQ: AUXL) • ~450,000 patients in US & Europe are potential candidates for AA4500 in Dupuytren’s & Peyronie’s indications – Sales value likely to be high – Dupuytren’s is a great product for a specialist approach – Peyronie’s is a natural fit for Urology field force • Ability to generate cash from ex-U.S. rights to pipeline is possible Pipeline Brings Value in Short to Mid Term Pipeline Brings Value in Short to Mid Term |
 Financials |
 36 (NASDAQ: AUXL) 2006 Financial Results / Guidance 2006 Financial Results / Guidance Y.T.D Full Yr. Q3 ’06 9/30/06 Guidance Revenues $17.6 $49.0 $64-68 R&D Expense $8.5 $25.7 $38-42 SG&A Expense $17.1 $45.5 $59-61 Net Loss $11.8 $33.2 $48-51 Net Loss per share (0.39) ($1.13) N/A Cash Balance $67.3 ($ millions except per share) |
 37 (NASDAQ: AUXL) Strategic Priorities Strategic Priorities • Continue Driving Testim Growth • Continue Development of AA4500: – Complete root cause investigation of AA4500 manufacturing issues & implement corrective actions – Resume and complete Phase III Dupuytren’s trials as soon as possible – Complete BLA batches for AA4500 in Horsham facility – Continue market research activities; prepare commercial team – Continue development of AA4500 in Peyronie’s Disease and Frozen Shoulder Syndrome • Implement infrastructure and processes to allow for rapid growth |
 38 (NASDAQ: AUXL) Fastest Growing TRT product in the U.S. Company Transforming Pipeline Opportunities Poised for Growth + = |