 Welcome to Auxilium’s First Analyst Day – Focusing on Xiaflex! October 17, 2007 Exhibit 99.1 |
 Armando Anido Chief Executive Officer and President |
 Safe Harbor Statement We will make various remarks during this presentation that constitute “forward-looking statements” for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995, including statements regarding future Testim market share and sales growth and factors that may drive such growth; the issuance of a patent from the pending U.S. application for Testim; the pricing, time to market, size of market, growth potential and therapeutic benefits of the Company’s product candidates, including XIAFLEX™ (clostridial collagenase for injection), formerly referred to as AA4500, for the treatment of Dupuytren’s contracture, Peyronie’s disease, and Frozen Shoulder syndrome; interpretation of market research data; reimbursement for and pricing of XIAFLEX; competition within certain markets relevant to the Company’s product candidates; interpretation of clinical results, including the efficacy and tolerability of the Company’s product candidates; the timing of the enrollment in and completion of clinical trials and the timing of reporting of results therefrom; the timing of manufacturing scale up for the Company’s product candidates; the Company’s ability to manufacture XIAFLEX at the Company’s Horsham facility; the ability of third party manufactures to timely and effectively provide services; the timing of the enrollment of patients in phase III trials for XIAFLEX for the treatment of Dupuytren’s contracture; the timing of the filing of the Biologics License Application for approval of XIAFLEX for the treatment of Dupuytren’s contracture and the approval thereof; the timing of the phase IIb trials for XIAFLEX for the treatment of Peyronie’s disease; competitive developments affecting the Company’s products and product candidates; the success of the Company’s development activities; the Company’s development and operational goals for fiscal 2007. All remarks other than statements of historical facts made during this presentation, including but not limited to, statements regarding future expectations, plans and prospects for the Company, statements regarding forward-looking financial information and other statements containing the words “believe,” “may,” “could,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” and similar expressions, as they relate to the Company, constitute forward-looking statements. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2006 and the Company’s Quarterly Report on Form 10-Q for the period ended June 30, 2007 under the heading “Risk Factors”, which is on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “Investor Relations - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 Today’s Agenda Who is AUXL Overview of XIAFLEX™ Dupuytren’s Contracture ~ Disease overview & treatment options ~ Clinical Development Plan ~ Market Overview Peyronie’s Disease ~ Disease overview & treatment options ~ Clinical Development Plan ~ Market Overview Questions and Answers |
 Ground Rules For The Day • Please hold all questions until after we finish the formal presentations. • We will limit each person to 1 question, then allow you to get back in the queue • We may limit responses from our guest speakers to preserve competitive data or intellectual property rights •Speakers are independent and their views may not be the same as AUXL’s • Please silence your cell phones & step out of the room if you need to take a call • Coffee & soft drinks are available just outside of the rear doors |
 Medical Experts Participating Today • Larry Hurst, M.D., Professor and Chair, Chief of Hand Surgery, Department of Orthopaedics of the Health Science Center, State University of New York at Stony Brook • Gerald H. Jordan, M.D., F.A.C.S., F.A.A.P., Professor, Department of Urology, Eastern Virginia Medical School Director, Devine Center for Genitourinary Reconstructive Surgery, Sentara Norfolk General Hospital |
 Participating from Auxilium Jim Fickenscher, Chief Financial Officer Roger Graham, Executive Vice President, Sales & Marketing Dr. Jyrki Mattila, Executive Vice President, Business Development, R&D and Technical Operations |
 Fastest Growing TRT product in the U.S. Late Stage Company Transforming Product Poised for Growth + = |
 Auxilium Pharmaceuticals, Inc. (NASDAQ: AUXL) Auxilium Pharmaceuticals, Inc. (NASDAQ: AUXL) • Specialty biopharmaceutical company focused on Urologists, Endocrinologists, Orthopedists and certain PCPs • Founded in 1999 • Headquarters in Malvern, Pa. ~ Manufacturing in Horsham, Pa. ~ Approximately 290 employees (~190 commercial) I.P.O in July 2004 ~ Currently approximately 40.0 million shares issued & outstanding ~ Additional 7 million shares subject to issuance from stock options & warrants A component of the NASDAQ BIOTECH INDEX |
 Broad Pipeline Maturing in Coming Years Broad Pipeline Maturing in Coming Years Note: Worldwide rights for all products available, except Testim where ex -U.S. rights have been licensed. XIAFLEX™ (clostridial collagenase for injection) was formally referred to as AA4500 Auxilium has exclusive option for all non-topical uses of XIAFLEX. Product Testim® gel Approved in U.S. Europe & Canada XIAFLEX™ XIAFLEX™ XIAFLEX™ Trans. Film AA4010 Trans. Film Trans. Film Market Phase III Phase II Phase I Pre-clinical Late Research Hypogonadism Dupuytren’s Contracture Peyronie’s Disease Frozen Shoulder Syndrome Overactive Bladder Hormones and Urology Pain |
 XIAFLEX – XIAFLEX – Company Transforming Company Transforming Opportunity Opportunity •Potential to replace surgery •Great fit for Specialty Biopharmaceutical Company •Worldwide rights offer options to build company or generate cash •We believe there are approximately 450,000 potential patients annually in U.S. and EU for Dupuytren’s & Peyronie’s indications - > $1 Billion opportunity, based on market research and analysis •BLA for Dupuytren’s Contracture expected to file in early 2009 |
 Overview of XIAFLEX™ (Clostridial Collagenase for Injection) |
 XIAFLEX™ • Purified Clostridial Collagenase for injection • Specific combination of several subtypes of Collagenase • Produced by bacterial fermentation & purified at our Horsham facility • Filled and lyophilized by a third party manufacturing partner • Requires reconstitution with sterile diluent before injection |
 Mechanism of Action • Collagen is a molecule made up of three peptide chains arranged as an alpha-triple helix ~ Renders collagen impervious to all proteolytic enzymes except collagenase Endogenous collagenases ~ May be deficient in Dupuytren’s & Peyronie’s patients ~ Degrade collagen at one site & are slow to act ~ Readily inactivated by locally secreted tissue inhibitors (TIMPs) We believe that XIAFLEX™ ~ Attacks several sites within collagen molecule (due in part to multiple subtypes) ~ Acts rapidly against collagen ~ Is resistant to Tissue Inhibitors of Matrix Metalloproteinase (“TIMP’s”) |
 XIAFLEX™ • Over 1,300 patients have been exposed to XIAFLEX™ in a range of indications • 237 Dupuytren’s patients have been treated • 280 Peyronie’s patients have been treated |
 Development History • First formulated in the 1980’s • Initial trials in Peyronie’s Disease – 1982 • Initial trials in Dupuytren’s Contracture – 1996 • Orphan Drug Designations Granted ~ Dupuytren’s Contracture – 1996 ~ Peyronie’s Disease – 1997 Auxilium Obtains License to AA4500 - 2004 • Initial Dupuytren’s Phase III Pivotal Trial Completed – 2005 |
 Development History • New manufacturing process developed – 2004 to 2006 • Option exercised for Frozen Shoulder – December, 2005 • Horsham manufacturing facility acquired – August, 2006 • Results announced from ~ Initial Phase 3 study in Dupuytren’s contracture – June, 2006 ~ Phase 2 studies in Peyronie’s disease – October 2006 Clinical hold due to meltback – December, 2006 • Results from suspended Ph 3 trials announced – July, 2007 • Journal of Hand Surgery publishes pivotal Ph 3 data – Aug, 2007 • Resumption of Ph 3 program for Dupuytren’s – Sept, 2007 |
 Lawrence C. Hurst, MD Current Professional Appointments: ~ Professor & Chairman; Chief, Division of Hand Surgery, Department of Orthopaedics, SUNY @ Stony Brook School Of Medicine ~ Program Director, Hand Fellowship Training ~ Adjunct Staff/Hand Surgery: St. Charles Hospital, Port Jefferson, NY Education: ~ MD Degree: University of Vermont College Of Medicine: 1973 ~ BA Degree: College of Arts & Science, University of Vermont: 1969 |
 |
 History of Dupuytren’s Disease (DD) • Orkney and Iceland in 12 th & 13 th writings mention DD • Felix Plater of Basel describes DD in 1614 but thought pathology secondary to tendon problem • John Hunter accurately describes DD in 1777 – the year of Dupuytren’s birth • Ashley Copper writes about DD in “A treatise on Dislocations & Fractures” in 1822 |
 History The eponym for Dupuytren Disease was coined because M. Dupuytren performed a fasciotomy using an open transverse incision in 1831 |
 History of DD Despite Dupuytren’s famous open surgical treatment, Ashley Cooper’s closed fasciotomy was the treatment of choice but neither procedure was used frequently until anesthesia was added in 1842 and Lister’s antiseptic techniques were added in 1865. |
 Basic Science |
 HISTOLOGY, CELL BIOLOGY & BIOCHEMISTRY The Myofibroblast and Attached collagen Electron Microscopic view of a Myofibroblast |
 HISTOLOGY, CELL BIOLOGY & BIOCHEMISTRY Increased collagen type III & GAG’s Collagen helix pattern with increased cross- links Fibrotactin - an extracellular matrix- cytoskeletal connector |
 Anatomy |
 |
 |
 PATHOANATOMY Common Pathologic Cords Central Cord Lateral Cord Natatory Cord Pretendinous Cord Spiral Cord Nodule |
 PATHOANATOMY Digital Nerve and Vessel Spiral Cord Note: Cord Actually Straight and the Neurovascular bundle spirals! |
 Incidence • Genetics Associated Diseases Ectopic Locations |
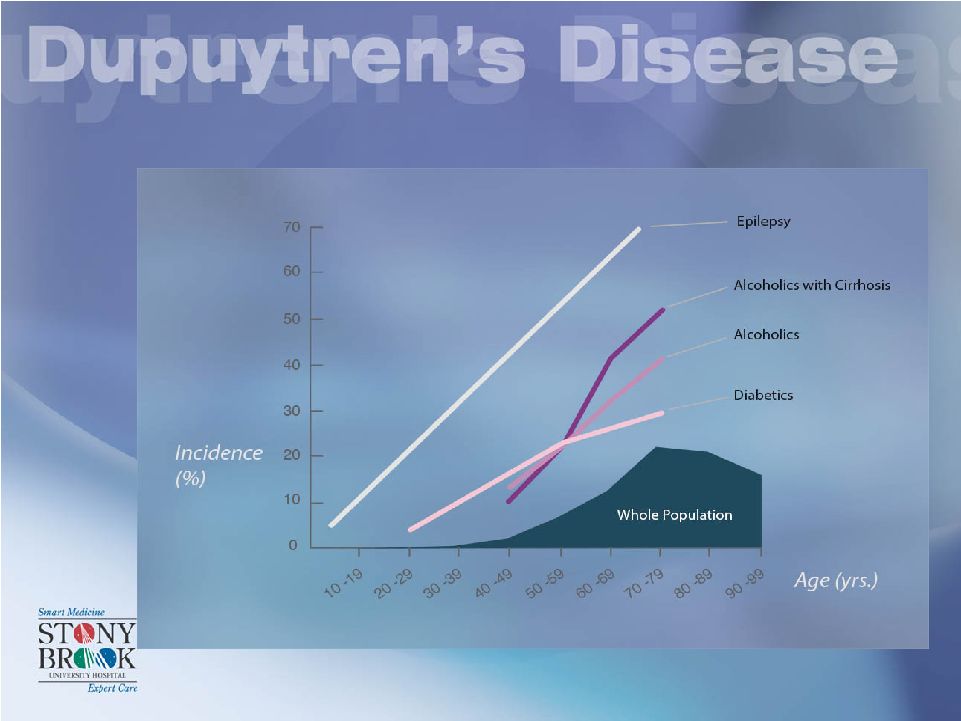
 INCIDENCE Mikkelsen in Norway found 9% incidence in males & 3% incidence in females Incidence: near 5% before age 20 above 20% at age 70 Brenner (2001) found 1.9 Million Germans affected |
 ASSOCIATED DISEASES |
 GENETICS: Autosomal dominant Mendelian Inheritance pattern with variable penetrance: |
 ECTOPIC FIBROMATOSES: - Foot – Plantar fibromatosis (Ledderhose) - Penile Fibromatosis (Peyronie) - Knuckle Pads |
 ECTOPIC FIBROMATOSES: - Foot – Plantar fibromatosis (Ledderhose) |
 Treatment Options Current Option - Surgery |
 Surgical Indications • 30 Degree MP Contracture • Any PIP Contracture • Nodule Over Trigger Finger Requiring Surgery • Anesthesia – Match type with Patient & Contracture-Local, Axillary Block or General |
 Surgical Indications • Positive Table Top Test The Dupuytren’s cord causes flexion contractures so that the palm and the fingers CAN NOT be placed on the same flat surface simultaneously !! |
 • These surgical indications suggest that surgeons recommend operative treatment for rather mild contractures. Why are these the standard textbook indications? |
 • Progressive - sometimes even aggressive. • Severe contractures are very difficult to correct surgically • Severe contractures can interfere significantly with activities of daily living (ADL) |
 Pre-op photo shows Dupuytren’s cord eroding through the skin pit base at first flexion crease of the finger Severe Contracture Can Cause Hygiene Problem |
 Imagine this hand easily getting into this pocket?? |
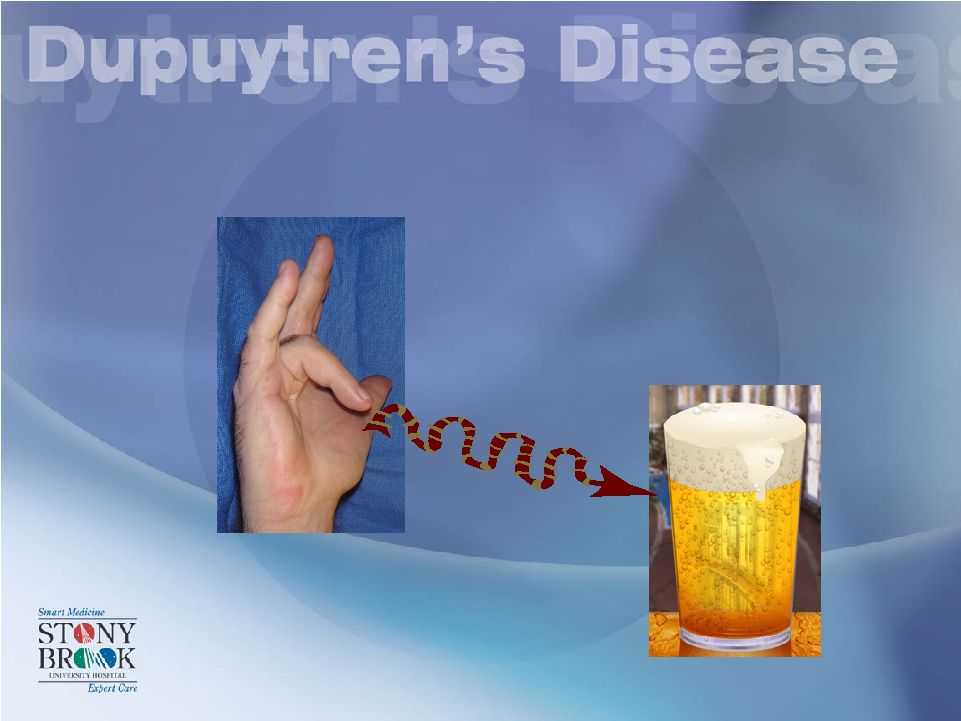 Imagine this hand holding this mug of beer? |
 Imagine this hand trying to wash your face? |
 Imagine this hand shaking your hand? |
 Many incisions have been tried over last century No incision has made surgery simple; always successful or guaranteed a long term correction! |
 COMMON USED INCISONS FOR OPERATIVE MANAGEMENT ARE: Brunner(zig-zag plasty), Standard Z-plasty, Or McCash (Transverse) |
 Operative Management in DD Distally Based Flap |
 Operative Management in DD |
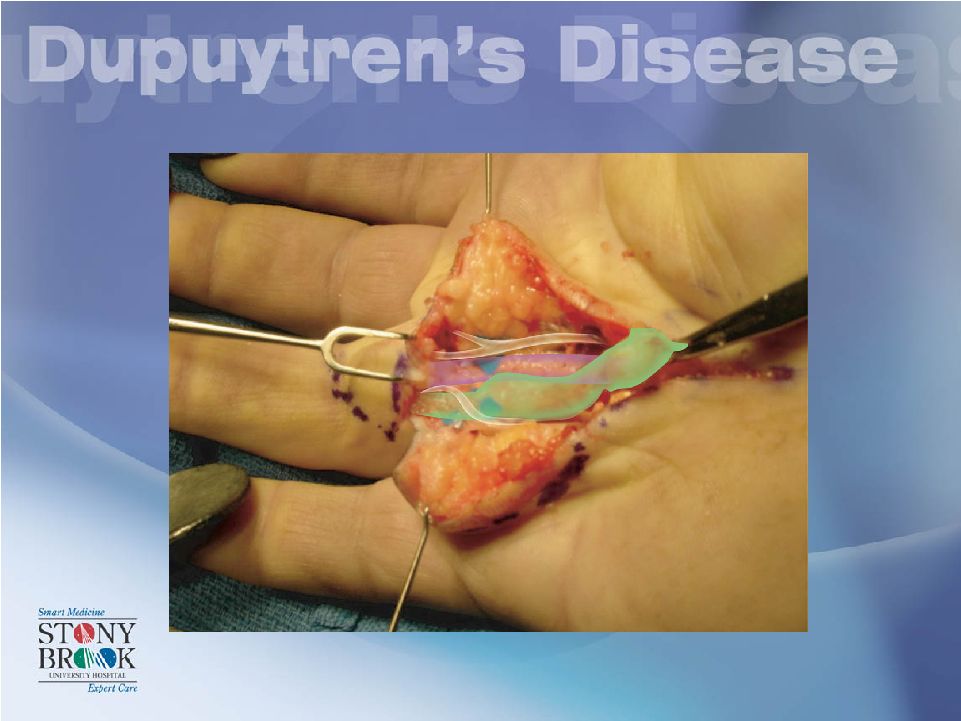
 Spiral Cords Operative Management in DD Digital nerve superficial to the spiral cord Spiral cord moves N-V Bundle Proximally, Centrally and Superficially |
 |
 Pre-tendonous Cord in: Cord out: Excised Dupuytren’s Cord |
 With tourniquet down check: Flap vascularity: |
 Surgical Treatment can be very extensive ! |
 Operative Management in DD: OPEN Palm (McCash) Technique At Surgery 2-3 Months Post-op |
 Operative Management of DD in the Radial Side of the Hand: Contracting Web Superficial and Deep Commissurial Cords |
 Skin Grafts Cord tissue New Skin graft Healed graft from previous surgery Skin Grafts |
 Outcomes |
 PROGNOSIS – Mc Farlane 1202 Surgical patients 6-18 months Follow-up: -19% Perfect -56% Improved -25% same or worse |
 Another Surgical Option is the Closed fasciotomy which CAN BE DANGEROUS! Why? - Because This NERVE Might Be Right Here! |
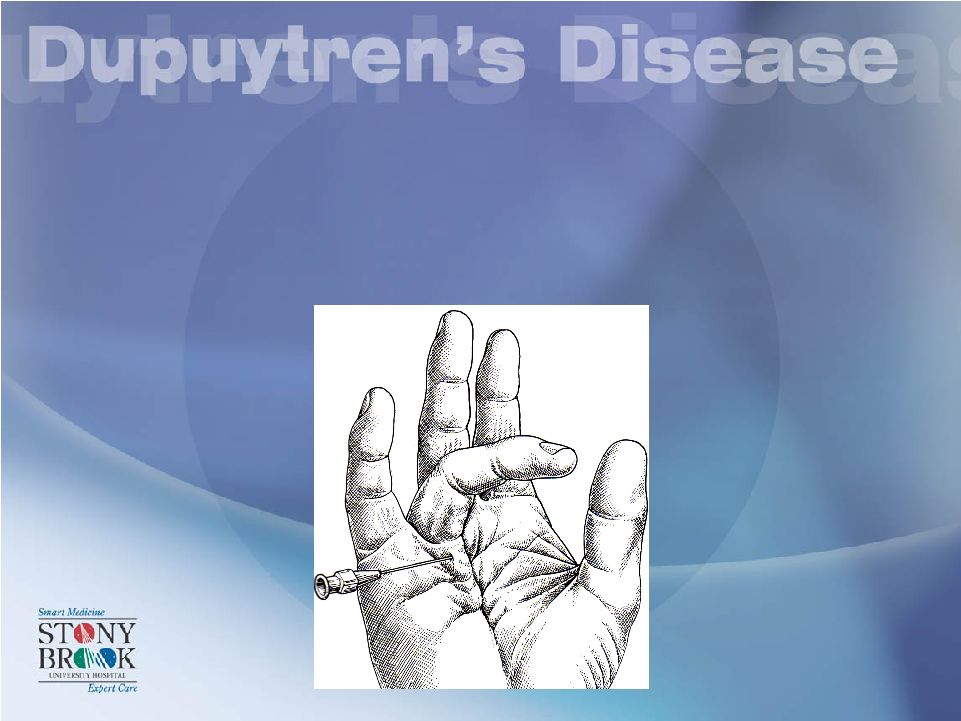 Another Surgical Option is the Needle fasciotomy. This too can be dangerous to digital nerves. |
 Operative Management in DD: Needle fasciotomy |
 Operative Management in DD: Needle fasciotomy Nerve, f/u |
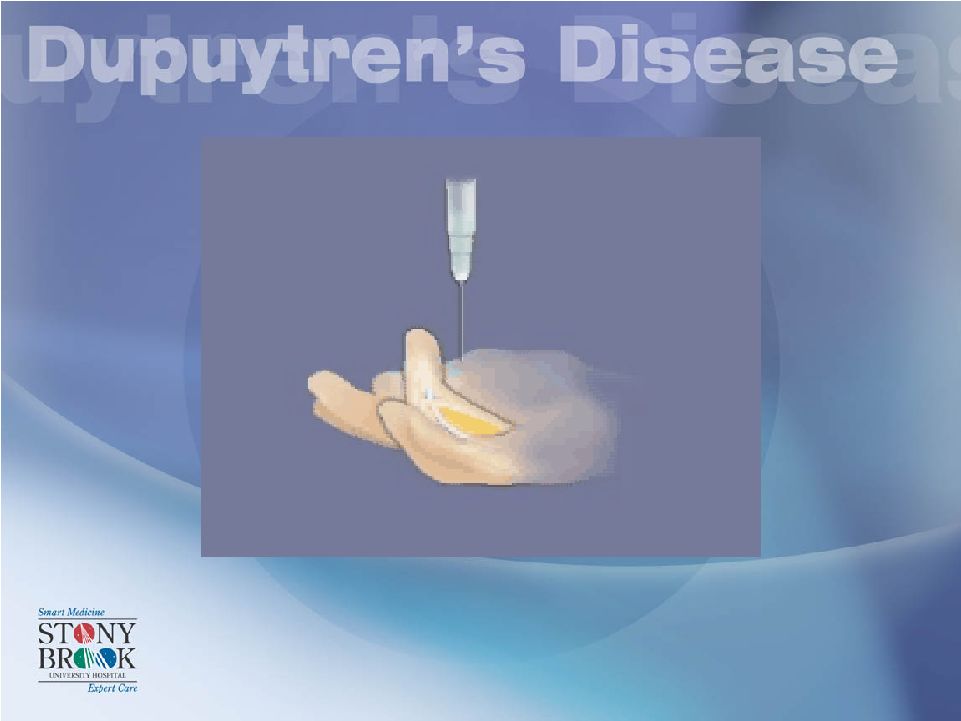
 Non-Operative Management: XIAFLEX • Indications = Surgical Indications ! |
 Non-Operative Management: XIAFLEX* *Currently not FDA approved .25 CC’s for MP and .20 CC’s for PIP Each with 0.58 mg of Xiaflex which equals 10,000 units |
 Non-Operative Management: XIAFLEX* *Currently not FDA approved Distal End Of Cord Proximal End Of Cord Area of Maximal “Bowstringing” (Separation of Cord and Flexor Tendons) Target areas for Xiaflex Injection |
 Non-Operative Management: XIAFLEX* *Currently not FDA approved 1/3 of dose in three separate places In the cord (not through the cord) |
 Non-Operative Management The needle should be placed in, not through the gritty gristly consistency of the cord (can be palpated with the end of the needle) |
 Non-Operative Management of “Y” Shaped Cord *Currently not FDA approved 1/3 of dose in three separate places In the cord (not through the cord) • Created by a combination of a central and natatory cord • The “point” of the Y should be injected |
 XIAFLEX Clinical Study Results |
 98 133 176 199 Total 19 16 19 19 § Open label study of the relative safety and efficacy of collagenase therapy in the treatment of residual type Dupuytren’s disease. DUPY- 404 (continuat ion from - 303) 14 16 23 35 (12 of 35 subjects had placebo only) Double-blind randomized, placebo controlled study of the relative safety and efficacy of collagenase therapy in the treatment of residual type Dupuytren’s disease. DUPY- 303 43 31 63 80 (17 of 80 subjects at lower dose) Double-blind randomized, parallel-group, placebo controlled, trial to determine the safety and efficacy of doses of 2500, 5000 or 10,000 units of collagenase compared to placebo. DUPY- 202 13 36 49 49 Double-blind, placebo controlled study to evaluate the safety and efficacy of collagenase in improving the degree of flexion deformity and range of finger motion in ’s with residual Dupuytren’s disease DUPY- 101 9 34 29 35 (6 of 35 subjects at lower dose) Open label evaluation of the efficacy of collagenase as a non-operative treatment for residual stage Dupuytren’s disease Open label study PIP MP # Subjects Number of Joints (at 10,000 U / Injection) No. of Subjects (10,000 U/Injection) No. Enrolled Subjects Protocol Title Study Number Summary of Clinical Studies |
 • 215 patients with Dupuytren’s contracture treated with Xiaflex – 0.58mg per injection appears to be the most effective dosage – One Phase 3 study with 35 patients completed Overview Of Clinical Data |
 Overview (cont.) • Two double-blind, placebo controlled trials – One Phase 2: 80 subjects – One Phase 3: 35 subjects • Clinical Success: return to within 0 –5 of normal O O |
 Efficacy Profile - DUPY 101 Double Blind Exploratory • One injection • 49 subjects • AA4500 Success (Primary Joint) – MP 14/18 = 78% – PIP 5/7 = 71% JHS 27A, 5, p.788-798 (2002) |
 Efficacy Profile - DUPY 202 Double Blind Phase 2 • One injection • 80 subjects • AA4500 Success (Primary Joint) – MP 13/16 = 81% – PIP 5/7 = 71% JHS 27A, 5, p.788-798 (2002) |
 Efficacy Profile - DUPY 303 Double Blind Phase 3 XIAFLEX Placebo P-value Primary End Point 21/23 (91%)* 0/12 < .001 - MP Joint 12/14 (86%) 0/7 < .001 - PIP Joint 9/9 (100%) 0/5 < .001 Secondary Joint 5/6 (83%) 0/3 .035 Median Time to Success 8 Days N/A Mean # Injections For Success 1.4 N/A * Success rates were 70% after a single injection JHS, 32A, 6, p.767-774 (2007) |
 Efficacy Profile – Summary from DB studies • MP joints: 81% (one injection) to 86% (multiple injections) • PIP joints: 71% (one injection) to 100% (multiple injections) |
 Safety Profile (202 & 303) • Generally well tolerated • Treatment-related AEs confined to the local / administration site – Related to the injection or manipulation procedure of rupturing the cord • Mild or moderate, non-serious, transient, and resolved within 2 - 4 weeks without sequelae. |
 Summary •Excellent efficacy profile, ranging from 71% - 100% after up to three injections •Most achieved clinical success within one month •General well tolerated after multiple injections •AEs confined locally and were mild, moderate, and resolved with 2- 4 weeks •In 202, no indication of antibody neutralization •In 202, no AEs deemed to be due to an immunological reaction |
 53 year old male complaining of inability to straighten his right little finger for the past 3 years. It “gets in my way!” Exam shows an MP contracture. post treatment Non-Operative Management: XIAFLEX* *Currently not FDA approved |
 51 y.o. male with severe recurrent DD Of the long finer. post treatment Non-Operative Management: XIAFLEX* *Currently not FDA approved |
 56 y.o. male complaining of a contracture of his left little finger present for five years. He can’t put his hand in his pocket and puts his finger in his eye while washing his face. Exam shows a PIP contracture post treatment Non-Operative Management: XIAFLEX* *Currently not FDA approved |
 |
 |
 Clinical Program for Dupuytren’s Contracture Jyrki Mattila Executive Vice President R&D |
 Suspended Phase 3 Studies • First clinical trials utilizing XIAFLEX ™ manufactured under process developed by Auxilium • Six sites in Australia and United States participated • Thirty patients were treated ~ 22 received XIAFLEX ™ ~ 8 received placebo injections • 14 of 22 patients injected with XIAFLEX ™ achieved clinical success • No placebo patients experienced clinical success • Adverse events were similar to those from earlier clinical studies |
 Xiaflex ™ Clinical Phase 3 Program CORD I Phase 3 double blind study with open label extension in the United States CORD II Phase 3 double blind study with open label extension in Australia JOINT I Open label phase 3 study in the United States JOINT II Open label phase 3 study in Australia and Europe |
 CORD I Collagenase Option for Reduction of Dupuytren’s ------------------------- A Phase 3, Double-Blind, Randomized, Placebo-Controlled Study of Xiaflex ™ in the Treatment of Dupuytren’s Contracture |
 Objectives – CORD I Primary efficacy objective • Reducing the degree of contracture of the primary MP or PIP joints Secondary efficacy objective • Reducing the degree of contracture in multiple MP and PIP joints Safety Objective • Provide data on the tolerability of Xiaflex ™ up to 12 months post first injection |
 Study Design – CORD I Total of 216 subjects will be enrolled in approximately 15 study sites in the United States • Subjects stratified by ~ the primary joint type (144 MP joints and 72 PIP joints) ~ severity of primary joint contracture (< > 50°/40° for MP/PIP joints, respectively) • Subjects randomized in a 2:1 ratio to either Xiaflex ™ (144 subjects) or placebo (72 subjects) |
 Inclusion Criteria - CORD I Dupuytren’s Contracture of at least one finger, • At least 20° but no greater than 100° for MP joint • At least 20 but no greater than 80° for PIP joint • Caused by a palpable cord that has never been treated with Xiaflex ™ . Subject has a positive “table top test” |
 Treatment and Follow-up – CORD I Double blind placebo controlled phase (3 month): • Maximum of 3 injections of study drug (Day 0, 30, and 60). • Follow-up visits will be 1, 7 and 30 days after each injection. Open label extension (9 months): Nine month follow-up after completion of the double blind phase • Subjects may receive up to 5 additional injections of Xiaflex ™ |
 Efficacy Definitions – CORD I PRIMARY EFFICACY ENDPOINT: • The proportion of subjects who achieve clinical success of the primary joint after up to three injections Clinical success: Reduction in contracture to < 5° of normal 30 days after last injection. |
 Secondary efficacy endpoints – CORD I Recorded for the primary joint • Clinical success after the first injection • Clinical improvement • Percent reduction from baseline in contracture • Percent change from baseline in range of motion • Time to reach clinical success Clinical improvement: A > 50% reduction from baseline in contracture |
 Expected Timeline – CORD I • Enrollment completed by end of 2007 • Top line results of the double blind portion available 2Q 2008 • 12 month follow-up completed by end of 2008 • Study ready for BLA submission early 2009 |
 CORD II 60 patients, following protocol similar to CORD I • 3 month double blind • 9 month open label Five study sites in Australia Top line data from the double blind portion expected to be available 2Q 2008 |
 JOINT I & II Joint Open-Label Injection Non-Surgical Treatment ------------------------- Phase 3, Open-Label Studies of the Safety and Efficacy of Xiaflex ™ in the Treatment of Dupuytren’s Contracture |
 Design - JOINT I & II Objective: To evaluate the safety and efficacy of up to 5 injections of Xiaflex ™ in multiple joints of subjects with Dupuytren’s contracture • No placebo control, all subjects receive Xiaflex ™ • JOINT I includes approximately 10 sites in the United States and enrolls 100 subjects • JOINT II includes approximately 15 sites in Europe and Australia and enrolls 340 subjects |
 Design - JOINT I & II • Inclusion and exclusion criteria same as in CORD I • Efficacy definitions same as in CORD I • Following safety endpoints will be monitored: ~ Adverse events ~ Clinical laboratory tests and vital signs ~ Grip strength of the treated hand ~ Immunogenicity |
 Clinical Phase 3 Program Summary: • All studies up and running at this point •Enrollment to CORD I and CORD II expected to complete by end of 2007 •Approximately 725 subjects are planned to be enrolled in total •Phase 3 program to complete to allow BLA submission early 2009 |
 Market Overview for XIAFLEX: Dupuytren’s Disease Roger D. Graham, Jr. Executive Vice President Sales & Marketing |
 Market Research Methodology • Approximately 600 Orthopedic and Hand Surgeons ~ 415 U.S. Physicians ~ 193 E.U. Physicians Qualitative and Quantitative Research ~ 1 on 1 in depth interviews with KOLs (High Treaters) ~ Web based Surveys ~ Second quantitative study designed to provide 95% confidence level and 7.5% margin of error |
 Surgeon’s Motivation To Use XIAFLEX • Dissatisfied With Surgery ~ Flexion contractures often not fully corrected (especially PIP joints) ~ High rate of complications ~ Difficult to meet patients’ expectations Believe XIAFLEX Fills An Unmet Need ~ Earlier use than surgery ~ Appropriate for poor surgical candidates ~ As efficacious as surgery, perhaps more efficacious (especially for PIP joints) ~ Significantly fewer complications ~ Requires skill of a surgeon ~ Efficient to administer (time economies vs surgery) Source: Market Research, Auxilium Pharmaceuticals |
 Reimbursement and Surgical Costs • Reimbursement ~ All payers anticipate coverage for XIAFLEX, including EU ~ New CPT codes may be needed for injection & manipulation ~ Miscellaneous J-code at launch Surgical Costs - Ambulatory (99%) ~ $ 4,800* • In-patient Surgery ~ $12,000** * Pharmetrics ** Verispan Total Cost Total Cost |
 XIAFLEX Potential: Dupuytren’s Contracture Patients Seeking Treatment ~1.03M No Prior Surgery ~540,000 Had Surgery Previously ~490,000 Watchful Waiting ~270,000 Medical/ Surgical Treatment ~270,000 Medical/ Surgical Treatment ~125,000 Watchful Wait/ In Follow Up ~365,000 XIAFLEX Potential Candidates ~240,000 ~150,000 in U.S. ~ 90,000 in Europe Prevalence 3 – 6% of Caucasian Population ~13.5M to 27.0M Source: Auxilium research and analysis |
 Xiaflex Commercial Opportunity We believe: There is a pent-up demand for non-surgical therapy Surgeons and patients prefer non-invasive treatment Each patient on average has 2.2 joints which will require treatment; 49% have contractures in both hands Early data indicates on average 1.4 vials of XIAFLEX required per joint ~ 2.2 joints per patient x 1.4 vials per joint = 3.1 vials per patient on average XIAFLEX treatment can be priced competitively to cost of surgery (~$5,000 in the U.S.) |
 Gerald H. Jordan, MD, FACS, FAAP Current Professional Appointments: ~ Professor Urology Department, Eastern Virginia Medical School ~ Director, The Devine Center For Genitourinary Reconstruction, Sentara Norfolk General Hospital Education: ~ MD Degree: University of Texas Health Sciences Center Medical School: 1977 ~ Pre-Med: United States International University: 1973 ~ BS Degree: United States Naval Academy: 1966 |
 Peyronie’s Disease Gerald H. Jordan, MD Gerald H. Jordan, MD Department of Urology Department of Urology Eastern Virginia Medical School Eastern Virginia Medical School Norfolk, VA Norfolk, VA |
 Peyronie’s disease is a scarring phenomenon affecting the tunica albuginea of the penis¹ Scar tissue forms “plaques” that can result in pain with erection, penile curvature/deviation, penile shortening, indentations, and/or erectile dysfunction Histological staining shows fibrin accumulation and excessive depolarized collagen deposition² It is associated with difficulty with sexual intercourse, loss of self-esteem, and depression There are no approved therapies for the treatment of Peyronie’s disease 1 Smith, BH. Am J Clin Pathol. 1966;45:670-678. 2 Somers, KD and Dawson, DM. J Urol. 1997;157:311–315. Background |
 FALLOPIUS -- FALLOPIUS -- 1561 1561 de la PEYRONIE -- de la PEYRONIE -- 1743 1743 Peyronie’s Disease in History INDURATIO PLASTICA PENE |
 Peyronie’s Disease In History |
 Incurable Incurable Reassurance Reassurance Medical Therapy Medical Therapy Surgery Surgery * Peyronie’s Disease |
 Fibrous Lesions - Fibrous Lesions - Impede Expansion Impede Expansion Curvature And / Or Curvature And / Or Indentation And / Or Indentation And / Or Foreshortening Foreshortening Peyronie’s Disease |
 Peyronie’s Disease Update |
 Beta Blockers Beta Blockers Phenytoin Phenytoin Paget’s Disease Paget’s Disease Diabetes Mellitus Diabetes Mellitus Incidence of Peyronie’s Disease |
 Familial Familial Dupuytren’s 15 - Dupuytren’s 15 - 30 % 30 % 10 - 10 - 40 % 40 % Autosomal Autosomal Dominant Dominant Nyberg. JU, 1982; Ralph. JU, 1997 Nyberg. JU, 1982; Ralph. JU, 1997 Incidence of Peyronie’s Disease |
 1 - 100 Age of Onset - 53yrs (MEAN) Prevalence - 0.4 % Rheu Arth, Hypertension Asymptomatic Prevalence: 22 / 100 Jordan. Cont Urol, 1999, Lindsay. JU, 1991; Smith. Am J Clin Path, 1969 Incidence of Peyronie’s Disease |
 *** 45 - 45 - 65 Years 65 Years PEAK MID 50’s PEAK MID 50’s Loss Of Tissue Elasticity Loss Of Tissue Elasticity Erectile Dysfunction Erectile Dysfunction Incidence of Peyronie’s Disease |
 Causes of Peyronie’s Disease Trauma - Trauma - Insertion Septum Insertion Septum Localized Aberration Wound Localized Aberration Wound Healing Healing Associated With ED Associated With ED Associated Fibrin Deposition Associated Fibrin Deposition Devine. JU, 1997, Jarow. JU, 1997, Somers. JU, 1997 Devine. JU, 1997, Jarow. JU, 1997, Somers. JU, 1997 |
 Etiology - Trauma Midline Septal Insertion - Midline Septal Insertion - Vulnerable with Buckling Vulnerable with Buckling Devine. JU, 1997, Jarow. JU, 1997, Gelbard, 1993 Devine. JU, 1997, Jarow. JU, 1997, Gelbard, 1993 |
 Etiology - Genetics Paget’s Disease Of The Bone Dupuytren's Contracture Certain HLA Subtypes Autoimmune Disease Hyperactive Wound Healing Lyles. J Bone/Mineral Res, 1997, Nyberg. JU, 1982, Nyberg. JU, 1997, Ralph. JU, 1997, Leffell. JU, 1997 Schiavino. Uro, 1997, Stewart. JU, 1994, Devine. Clin Plas Surg, 1988, Diegelman. JU, 1997 |
 Pathology Round Cell Infiltration Round Cell Infiltration Obliterated Space of Smith Obliterated Space of Smith Fibrin Deposits Fibrin Deposits Smith. Am J Clin Path,1969, Devine. JU,1997, Somers. JU,1997 Smith. Am J Clin Path,1969, Devine. JU,1997, Somers. JU,1997 |
 Pathology – Round Cell Infiltration; Obliterated Space of Smith Smith. Am J Clin Path,1969, Devine. JU,1997 Smith. Am J Clin Path,1969, Devine. JU,1997 |
 Pathology – Fibrin Deposits Somers. JU,1997 Somers. JU,1997 |
 Pathology Dense Collagen Dense Collagen Decreased Elastin Decreased Elastin Dystrophic Calcification Dystrophic Calcification Cartilaginous Metaplasia Cartilaginous Metaplasia Gelbard. JU,1988, Ehrlich. JU,1997 Gelbard. JU,1988, Ehrlich. JU,1997 |
 Anatomy - Tunica Albuginea Multilayered Multilayered 1.5 - 1.5 - 3.0 mm thick 3.0 mm thick Incompetent Septum Incompetent Septum Longitudinal Layer Longitudinal Layer Thinnest Thinnest 3 3 - - 9 9 O’clock O’clock Hsu et al. Int J Imp,1992, Brock et al. JU,1997 Hsu et al. Int J Imp,1992, Brock et al. JU,1997 |
 Dorsal Opposite Ventral - Upward Bending Septum Joins Circ Layer - Delamination Ralph. JU,1997, Devine. Clin Plast Surg,1988, Jarow. JU,1997, Devine. JU,1997 Anatomy - Tunica Albuginea |
 Anatomy - Tunica Albuginea Plaque Usually Dorsal Devine. JU, 1997, Gelbard. JU, 1993, El-Saka et al. Bju,1997 |
 Anatomy - Tunica Albuginea Compliance of the Tunica Compliance of the Tunica Collagen is Brittle Collagen is Brittle Elastin Stretches Elastin Stretches Helical Helical Arrangement Arrangement Straightens Straightens Elastin Elastin Stretches Stretches Ground Ground Substance Substance Displaces Displaces |
 Peyronie’s Disease Update |
 Molecular Basis of Injury Molecular Basis of Injury Repeated Mechanical Stress - Microvascular Trauma Delamination Bleeding Fibrinogen Devine. JU,1997, Jarow. JU,1997, Devine. Clin Plas Surg,1988 |
 Molecular Basis of Injury Molecular Basis of Injury Pain And Inflammation Vasoactive Factors Is The Inflammatory Response “Trapped?” PDGF-A and PDGF-B, TGF-b1 Diegelman. JU,1997, Van de Water. JU,1997, Somers. JU,1997 |
 Molecular Basis of Injury Molecular Basis of Injury Causes Increased Causes Increased Connective Tissue Connective Tissue Inhibits Inhibits Collagenases Collagenases Can Induce It’s Own Can Induce It’s Own Production Production Border. NEJM,1994, Border. J Clin Inves,1992, Moreland. Int J Impot,1998 Implicated in Soft Implicated in Soft Tissue Fibrosis Tissue Fibrosis Implicated in ED Implicated in ED Increases Synthesis of Increases Synthesis of Fibroblasts – Fibroblasts – Fibrosis Fibrosis Wound Healing Wound Healing TGF-b1 TGF-b1 |
 Active Phase Active Phase Painful Painful Erections Erections – – Sometimes Sometimes Changing Changing Deformity Deformity Secondary Phase Secondary Phase Pain Resolves Pain Resolves Stable Deformity Stable Deformity Gelbard. JU, 1990, Williams. JU, 1970 Gelbard. JU, 1990, Williams. JU, 1970 * Disease Progression |
 Poorly Defined in the Literature Poorly Defined in the Literature Mentioned Only In Passing Mentioned Only In Passing Lowsley. Lowsley. JU, JU, 1950, 1950, Gelbard. Gelbard. JU,1990, JU,1990, Williams. Williams. JU, JU, 1970, 1970, Devine. Devine. JU, JU, 1974, 1974, Mellman, Mellman, Ju Ju 1978, 1978, Pryor. Pryor. JU, JU, 1979, Ralph. Bju ,1992, Jones. JU, 1984 1979, Ralph. Bju ,1992, Jones. JU, 1984 Psychological Aspects |
 Disturbed Body Image Disturbed Body Image Fearful Of Treatment Fearful Of Treatment Many Functional Aspects of ED Many Functional Aspects of ED Like “Death” Like “Death” Denial Denial Ambivalence Ambivalence Anxiety Anxiety Depression Depression Shame Shame Embarrassment Embarrassment Self Disgust Self Disgust Jones. AUA Update, 1998, Rosen. Data In Process, 2005 Jones. AUA Update, 1998, Rosen. Data In Process, 2005 Psychological Aspects |
 Youthful Libido Youthful Libido Aging Tissues Aging Tissues Relate With Intercourse Relate With Intercourse Do Not “ Do Not “ Like ” Like ” To Talk About the To Talk About the Problem Problem Jones. AUA Update, 1998, Rosen. Data In Process, 2005 Jones. AUA Update, 1998, Rosen. Data In Process, 2005 Psychological Aspects |
 Most Patients: Most Patients: Told “End Of Sex Life” Told “End Of Sex Life” Admit … Admit … Are Coping Poorly Are Coping Poorly Believe “Sex Is Intercourse” Believe “Sex Is Intercourse” Must Be Told To Keep “Sexual Expression Must Be Told To Keep “Sexual Expression Alive” Alive” Jones. AUA Update, 1998 Jones. AUA Update, 1998 Psychological Aspects |
 Plaque or Induration: Plaque or Induration: Usually Located Dorsal Septal Insertion Usually Located Dorsal Septal Insertion Can Be Multiple Can Be Multiple Can Involve Septal Fibers Can Involve Septal Fibers Patients May Not Be Aware Patients May Not Be Aware Burford. JU, 1943, Bystrom.Scan J Urol Neph,1976, Williams. BJU, Burford. JU, 1943, Bystrom.Scan J Urol Neph,1976, Williams. BJU, 1980, Furlow. JU, 1975 1980, Furlow. JU, 1975 Clinical Features |
 Penile Pain: Usually Only With Erection Usually Not Severe But Can Interfere With Intercourse Spontaneous Improvement The Rule As Inflammation Resolves Burford. JU, 1943, Gelbard. JU, 1990, Williams. BJU, 1980, Furlow. JU, 1975 Clinical Features |
 Curvature and/or Deformity: Usually Dorsal Curve Curvature Can Be Complex Lateral or Ventral – Disabling Can Cause Indentation Burford. JU, 1943, Gelbard. JU, 1990, Williams. BJU, 1980, Furlow. JU, 1975 Clinical Features |
 Clinical Features |
 Erectile Dysfunction: Reported Incidence - Variable Bystrom. Scan J Urol Neph,1976, Levine. JU,1996, Stecker. JU,1984, Jordan. JU,1993, Furlow. JU,1975, Gasior. WJU,1990, Amin. Bj Rad,1993, Montorsi. JU,1994, Pryor. ACTA UROL BELG,1998, Lopez et al. JU,1993, Chilton. BJU,1982, Ralph. BJU,1992, Metz. JU,1983 Clinical Features |
 Reduced Elastin - Increased Type III Collagen Associated with Veno-occlusive Problems Cavernosal Fibrosis Can Interfere with Arterial Flow Chisang et al. Euro Urol,1992, Akkus et al. BJU,1997, Ralph. BJU,1992 Clinical Features |
 Vitamin E (tocopherol) – Vitamin E (tocopherol) – antioxidant antioxidant Potaba Potaba (Para Aminobenzoate) (Para Aminobenzoate) Allegra Allegra (Seldane) (Seldane) Colchicine Colchicine Tamoxifen Tamoxifen Carnitine Carnitine (Acetyl-L-Carnitine/Propionyl- (Acetyl-L-Carnitine/Propionyl- L-Carnitine) L-Carnitine) Trental Trental (Pentoxifylline) (Pentoxifylline) Natulan Natulan (Procarbazine) (Procarbazine) Steroids or NSAIDs Steroids or NSAIDs PDE-5 Inhibitors PDE-5 Inhibitors Medical Management - Oral |
 Rigorous Well Designed / Controlled Studies Are Needed As It Stands Now, The Role Of Oral Therapy To Alter The Progress Of Peyronie's Disease Is Probably Limited To The Acute Phase Of Disease Mynderse And Monga, I J Imp Res, 2002 Medical Management - Oral |
 Steroids Steroids Parathyroid Hormone Parathyroid Hormone Orgotein Orgotein Verapamil Verapamil Interferon Interferon -2b -2b Medical Management – Intralesional Injection |
 Medical Management - Topicals Steroids, Verapamil, Beta – Steroids, Verapamil, Beta – Aminopropionitrile, Orgotein Aminopropionitrile, Orgotein Transcutaneous Transcutaneous Absorption of Drug Absorption of Drug No Blinded / Controlled Studies No Blinded / Controlled Studies No Proven Efficacy - No Proven Efficacy - Single Reports Only Single Reports Only |
 Medical Management – Iontophoresis / Electromotive Therapy Steroids, Verapamil, Orgotein, Histamine Steroids, Verapamil, Orgotein, Histamine Enhanced Transcutaneous Enhanced Transcutaneous Absorption Of Absorption Of Drug Using Surface Delivered Heat Or Drug Using Surface Delivered Heat Or Current Current No Blinded / Controlled Studies No Blinded / Controlled Studies No Proven Efficacy - No Proven Efficacy - Single Reports Only Single Reports Only |
 Non-Surgical Management Lithotripsy Questionable Rationale - Ostensibly Mechanical Disruption of the Plaque Enhancing Compliance No Blinded / Controlled Studies No Proven Efficacy To Date, ? Ill Effects on the Erectile Tissue, ? Adjuvant to Intralesional Injection Bellorofonte. Arch Ital Urol Neph Androl,1988 Hauck JU, 2004, Hauck, Eur Urol 2000, Lebret Bju Int, 2000, Hamm, Eur Urol 2001, Abdel Salaam, 1999 |
 Medical Management - Combined Therapy Intralesional Injection And Oral Interferon ~-2b + Vit E Verapamil + Carnitine Verapamil + Tamoxifen Oral Vit E + Colchicine Lithotripsy + Intralesional Verapamil Birmngham Study Underway Ved + Intralesional Verapamil Study Protocol Under Evaluation Hellstrom, Bju Int 2003, Prieto Castro, Bju Int 2003, CAVALLINI Et Al, BJU INT2002 |
 Medical Management - Future Directions Investigations Suggest That Part Of The Peyronie’s Process Involves Supression Of “Antifibrotics” Matrixmetalloproteinases 1 Antitrypsin Cyclic GMP Lue . J Sex Med 2005, Hauck. J Sex Med 2005 ~ |
 Surgical Management Indications When The Deformity Precludes Intercourse Erectile Dysfunction Precludes Intercourse |
 Surgical Management Pre-operative Assessment Stable / Quiescent Disease Stable / Quiescent Disease Usually > 1yr from onset of symptoms Usually > 1yr from onset of symptoms Stable deformity for 3 – Stable deformity for 3 – 5 Mos 5 Mos Pain Free Pain Free Detailed Assessment of Erectile Detailed Assessment of Erectile Function Function CDU CDU DICC DICC True Informed Consent True Informed Consent |
 Surgical Management Options Plication Or Tunical Resection Tunical Resection With Plicating Closure Corporoplasty Nesbitt. Ju 1965, Ebbehoj. Urol 1985, Essed. Urol 1985, Lue. Ju 1998, Geertsen. Bju 1996, Emberger. Bju 1984, Sassine. Urol 1994, Kelami. Bju 1987, Licht. Ju 1997, Yachia. Ju 1990, Rehman. Ju 1997;Pryor And Fitzpatrick. Ju 1979, Ralph. Ju 1995, Porst. Springer – Verlag 1997 |
 Surgical Management Options Plication Or Tunical Resection |
 Surgical Management Options Excision / Incision With Grafting Excision / Incision With Grafting |
 Surgical Management Options |
 Prosthetic Implantation With Straightening Pryor. Churchill-Livingston, 1987, Wilson. JU, 1994, Montague. JU, 1006, Knoll. Urol, 1990, Eigner. JU, 1991, Carson. Ijir, 1998, Carwson. Ijir, 2002 Surgical Management Options |
 Medical Management – XIAFLEX Clostridial Clostridial Collagenase (XIAFLEX) Collagenase (XIAFLEX) Creation of “Chemical Incisions” Creation of “Chemical Incisions” with with Initiation of Remodeling Initiation of Remodeling Blinded / Controlled Studies Underway Blinded / Controlled Studies Underway Available Only on Protocol Available Only on Protocol Gelbard. Gelbard. Urol Urol Res, Res, 1982, 1982, Gelbard. Gelbard. JU,1985, JU,1985, Gelbard. Gelbard. JU,1993, JU,1993, National National Collagenase Collagenase Study Study Group, Group, Jordan. Jordan. J J Sexual Sexual Medicine, Medicine, 2007 2007 |
 Safety and Efficacy of Injectable Safety and Efficacy of Injectable Mixed Mixed Collagenase Subtypes (XIAFLEX) in the Collagenase Subtypes (XIAFLEX) in the Treatment of Peyronie’s Disease Treatment of Peyronie’s Disease |
 To assess the impact of injectable mixed collagenase subtypes (XIAFLEX, Auxilium) on the angle of penile deviation and plaque size To assess the tolerability and safety of XAIFLEX for the treatment of Peyronie’s disease Objectives |
 Methods: Study Design Study A (N=25) Baseline Week 12 Month 6 Follow-up Month 9 Follow-up 3 injections (7 10 d) 3 injections (7 10 d) X X X X X X Study B (N=10) Baseline Week 12 Month 6 Follow-up Month 9 Follow-up 3 injections (5 10 d) 3 injections (5 10 d) Week 6 3 injections (5 10 d) X X X X X X X X X |
 Methods: Patients Key inclusion criteria: Key inclusion criteria: Men aged 18 Men aged 18 75 years 75 years Diagnosis of Diagnosis of Peyronie’s disease Peyronie’s disease Key exclusion criteria: Key exclusion criteria: Prior surgery for Prior surgery for Peyronie’s disease Peyronie’s disease Extensively calcified Extensively calcified plaque plaque Substantial erectile Substantial erectile deficits due to deficits due to Peyronie’s disease Peyronie’s disease Penile bend <20 Penile bend <20 degrees degrees Sexual dysfunction due Sexual dysfunction due to other causes to other causes |
 Assessment of Efficacy and Tolerability Study A: Study A: Primary: changes from Primary: changes from baseline in the angle of baseline in the angle of deviation and plaque size deviation and plaque size Secondary: patient Secondary: patient responses on VAS for responses on VAS for pain and sexual function pain and sexual function Secondary: investigators’ Secondary: investigators’ global evaluation of global evaluation of change change Study B: Study B: Primary: patients’ Primary: patients’ responses on the responses on the Peyronie’s Patient Peyronie’s Patient Questionnaire at month 9 Questionnaire at month 9 Secondary: changes from Secondary: changes from baseline in angle of baseline in angle of deviation and plaque size deviation and plaque size Secondary: investigators’ Secondary: investigators’ global evaluation of global evaluation of change change • Tolerability: adverse events monitored in both studies |
 Results: Demographics and Disposition Study A: Study A: 25 men enrolled and 18 25 men enrolled and 18 completed the study completed the study Mean age was 55 years; Mean age was 55 years; 92% were white 92% were white Mean duration of illness Mean duration of illness was 39 months was 39 months Study B: Study B: 10 men enrolled; 9 10 men enrolled; 9 completed the study completed the study Mean age was 58 years; Mean age was 58 years; 90% were white 90% were white Mean duration of illness Mean duration of illness was 32 months was 32 months |
 Results: Clinical Success,* Deviation Angle Study A 58 53 35 0 20 40 60 80 100 M3 M6 M9 Study B 50 100 100 89 0 20 40 60 80 100 W6 M3 M6 M9 *Clinical success: 25% reduction in the angle of deviation. |
 Results: Clinical Success,* Plaque Size Study A 95 65 67 0 20 40 60 80 100 M3 M6 M9 *Clinical success: 25% reduction in plaque size. |
 Results: Mean Change From Baseline in Deviation Angle and Plaque Size Deviation Angle Deviation Angle ( ( ) ) Plaque Length Plaque Length (cm) (cm) Plaque Width Plaque Width (cm) (cm) Study A Study A Study B Study B Study A Study A Study B Study B Study A Study A Study B Study B Baseline Baseline 52.8 52.8 50.2 50.2 5.8 5.8 7.85 7.85 0.80 0.80 0.48 0.48 W6 W6 NA NA 15.2* 15.2* NA NA 0.05 0.05 NA NA 0.16 0.16 M3 M3 12.7* 12.7* 20.6 20.6 † † 0.94 0.94 † † 1.59 1.59 0.13* 0.13* 0.11 0.11 M6 M6 11.1 11.1 † † 28.4 28.4 † † 0.48 0.48 ‡ ‡ 0.11 0.11 0.15 0.15 ‡ ‡ 0.21 0.21 ‡ ‡ M9 M9 6.6 6.6 26.2 26.2 † † 0.68 0.68 0.00 0.00 0.12 0.12 ‡ ‡ 0.18 0.18 *P 0.007 versus baseline. † P 0.001 versus baseline. ‡ P 0.05 versus baseline. |
 Results: Peyronie’s Patient Questionnaire (Study B) Patient-reported improvements in sexual function were observed for: Enjoyment of intercourse (P=0.023) Satisfaction with sexual relationships (P=0.043) Freedom from pain during erection (P=0.009) At baseline, 4 patients reported no sexual activity, and at month 9, 100% (9/9) were sexually active All 9 patients reported pain-free erections |
 Results: Physical Symptoms (Study B) Pain with erection: resolved in 6 patients Pain with erection: resolved in 6 patients Plaque palpability: disappeared in 4 patients Plaque palpability: disappeared in 4 patients with moderate-to-severe palpation with moderate-to-severe palpation Penile indentation: disappeared in 3 patients Penile indentation: disappeared in 3 patients and improved from severe to moderate in 1 and improved from severe to moderate in 1 patient patient Penile shortening: improved from severe to Penile shortening: improved from severe to moderate in 1 patient moderate in 1 patient Penile numbness: moderate-to-severe Penile numbness: moderate-to-severe completely resolved in 1 patient completely resolved in 1 patient |
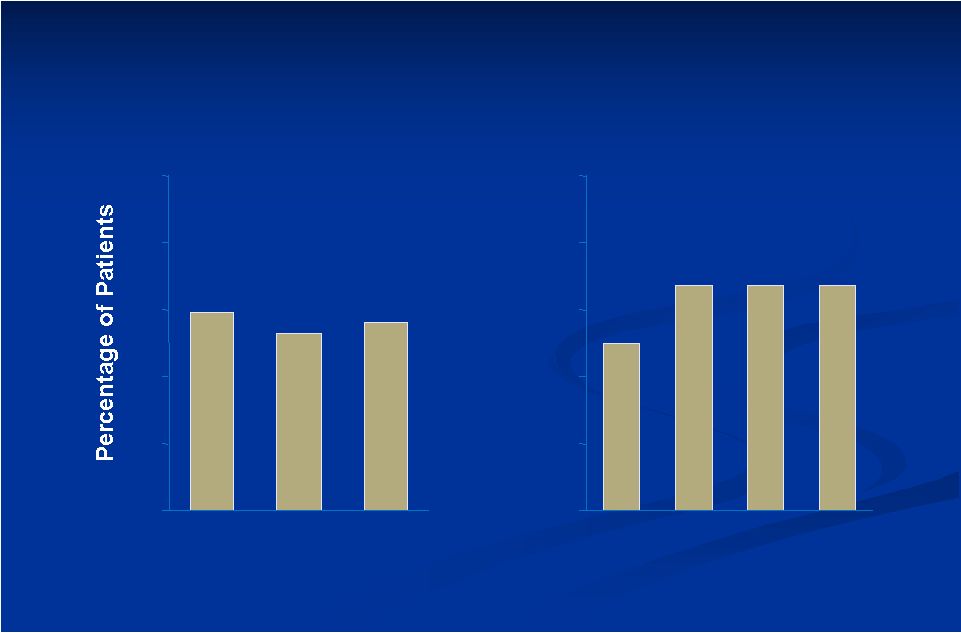 Results: Investigators’ Global Assessment of Patients Much Improved/Very Much Improved Study A 59 53 56 0 20 40 60 80 100 M3 M6 M9 Study B 50 67 67 67 0 20 40 60 80 100 W6 M3 M6 M9 |
 Adverse Events: Study A Relationship to Study Drug, n (%) Relationship to Study Drug, n (%) Adverse Event Adverse Event Possibly Possibly Probably Probably Definitely Definitely Reproductive system Reproductive system 1 (4) 1 (4) 1 (4) 1 (4) 14 (56) 14 (56) Erectile disturbance Erectile disturbance 0 (0) 0 (0) 1 (4) 1 (4) 0 (0) 0 (0) Penile disorder NOS Penile disorder NOS 0 (0) 0 (0) 0 (0) 0 (0) 5 (20) 5 (20) Penile pain Penile pain 1 (4) 1 (4) 0 (0) 0 (0) 12 (48) 12 (48) Penile swelling Penile swelling 0 (0) 0 (0) 0 (0) 0 (0) 6 (24) 6 (24) Skin and subcutaneous tissue Skin and subcutaneous tissue disorders disorders 0 (0) 0 (0) 2 (8) 2 (8) 19 (76) 19 (76) Contusions Contusions 0 (0) 0 (0) 2 (8) 2 (8) 14 (56) 14 (56) Ecchymosis Ecchymosis 0 (0) 0 (0) 0 (0) 0 (0) 11 (44) 11 (44) NOS=not otherwise specified. |
 Adverse Events: Study B Treatment-Related Adverse Event, n (%) Treatment-Related Adverse Event, n (%) Total (N=10) Total (N=10) General disorders and administration site conditions General disorders and administration site conditions 5 (50) 5 (50) Local swelling Local swelling 2 (20) 2 (20) Suprapubic Suprapubic pain pain 4 (40) 4 (40) Injury, poisoning, and procedural pain Injury, poisoning, and procedural pain 10 (100) 10 (100) Contusion Contusion 10 (100) 10 (100) Reproductive system and breast disorders Reproductive system and breast disorders 9 (90) 9 (90) Erectile dysfunction Erectile dysfunction 1 (10) 1 (10) Painful erection Painful erection 1 (10) 1 (10) Penile pain Penile pain 7 (70) 7 (70) Penile swelling Penile swelling 7 (70) 7 (70) Penis disorder Penis disorder 1 (10) 1 (10) Vascular disorders Vascular disorders 1 (10) 1 (10) Hematoma Hematoma 1 (10) 1 (10) |
 Adverse Events The most common treatment-related The most common treatment-related adverse events were localized adverse events were localized administration site reactions of mild administration site reactions of mild intensity and of limited duration intensity and of limited duration (typically within 3 weeks) (typically within 3 weeks) The most common adverse events were The most common adverse events were contusion, penile pain, and ecchymosis contusion, penile pain, and ecchymosis All adverse events resolved without All adverse events resolved without medical intervention medical intervention |
 Conclusions XIAFLEX, administered in up to 3 series XIAFLEX, administered in up to 3 series of multiple injections, appears to be of multiple injections, appears to be effective for the treatment of Peyronie’s effective for the treatment of Peyronie’s disease disease Treatment with XIAFLEX was generally Treatment with XIAFLEX was generally well tolerated well tolerated Larger controlled clinical trials are Larger controlled clinical trials are needed to confirm these promising needed to confirm these promising results and are in the process of results and are in the process of development development |
 Peyronie’s Disease Gerald H. Jordan, MD Gerald H. Jordan, MD Department of Urology Department of Urology Eastern Virginia Medical School Eastern Virginia Medical School Norfolk, VA Norfolk, VA |
 Development Plans for Peyronie’s Disease Jyrki Mattila Executive Vice President R&D |
 Preclinical local tolerability study • Four anatomic structures exposed to Xiaflex ™ ~ Tunica albuginea, Corpus cavernosum, NAV-complex and Urethra • Single dose and repeated dose groups • Dosing mimics expected clinical use • Histopathology, toxicokinetics and immunogenecity to be recorded • Study completed and results expected to be submitted to the FDA first half 2008 |
 Phase 2 b study • Injections of 0.58 mg in 0.25 mL volume given into Peyronie’s plaque ~ Two regimens of either six or nine total doses to be compared Patient Reported Outcome Survey likely to be used as a primary endpoint ~ AUXL developed its own proprietary questionnaire for Peyronie’s disease ~ Final validation in Phase 2b study Other endpoints include: ~ Reduction of penile curvature ~ Pain relief Phase 2b dose ranging study expected to begin first half 2008 |
 Market Overview for XIAFLEX: Peyronie’s Disease Roger D. Graham, Jr. Executive Vice President Sales & Marketing |
 Market Research Methodology • Approximately 600 Urologists/Surgeons ~ 415 U.S. Physicians ~ 193 E.U. Physicians Qualitative and Quantitative Research ~ 1 on 1 in depth interviews with KOLs (High Treaters) ~ Web based Surveys ~ Second quantitative study designed to provide 95% confidence level and 7.5% margin of error |
 Surgeon Motivation to Use Xiaflex • Seeking effective treatment options ~ No effective medical therapy ~ Surgery is last resort; many patients choose not to have it ~ Penile shortening ~ Erectile dysfunction/impotence ~ Many surgical patients dissatisfied with outcomes High Interest In XIAFLEX ~ Higher success rate than with off-label injections of verapamil or interferon; equal to surgery ~ Less trauma to penis/shorter recovery time than surgery ~ In-office procedure performed by urologists Source: Market Research, Auxilium Pharmaceuticals, Inc. |
 Reimbursement and Surgical Costs • All payers (US & EU) anticipate coverage for XIAFLEX • New CPT codes may be needed • Ambulatory (70%) & In-Patient (30%) Surgery ~ Approximate Cost (Blended) $ 13,000* • In-patient Surgery ~ $ 23,000** * Pharmetrics ** Verispan Approximate Cost |
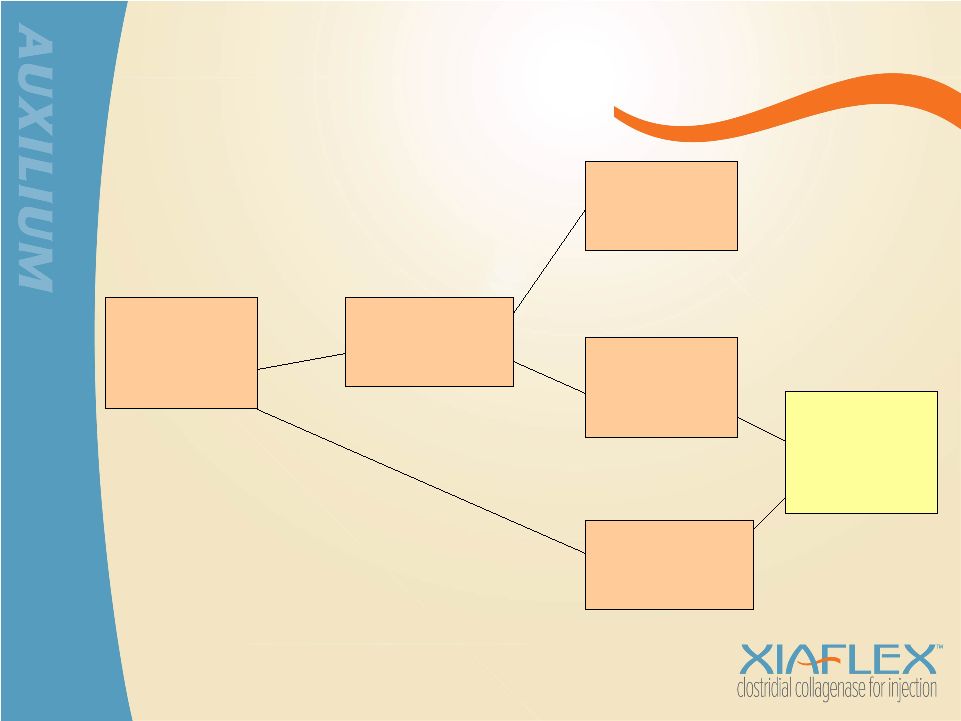 XIAFLEX Potential: Peyronie’s Disease Patients Seeking Treatment ~475,000 Surgery Not Recommended ~365,000 Watchful Waiting ~160,000 Medical or Intralesional Therapy ~205,000 Surgery Recommended ~110,000 XIAFLEX Potential Candidates ~210,000 ~ 135,000 in U.S. ~ 75,000 in Europe Source: Auxilium research and analysis |
 Xiaflex Commercial Opportunity We believe: • High need opportunity (MD and Patient) • Surgeons and patients prefer non-invasive treatment • Early indication that each patient will require 6 to 9 vials of Xiaflex • Reimbursement environment is positive • Peyronie’s represents a second significant commercial opportunity |
 Question & Answer Session |