 1 June 2009 (NASDAQ: AUXL) Exhibit 99.1 |
 2 Safe Harbor Statement We will make various remarks during this presentation that constitute “forward-looking statements” for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995, including statements regarding the pricing, time to market, size of market, growth potential and therapeutic benefits of the Company’s product candidates, including those for the treatment of Dupuytren’s contracture, Peyronie’s disease, and Frozen Shoulder syndrome; interpretation of market research data; competition within certain markets relevant to the Company’s product candidates; interpretation of clinical results, including the efficacy and tolerability of the Company’s product candidates; the timing of the commencement and completion of clinical trials and the timing of reporting of results therefrom; the timing of any action by the U.S. Food and Drug Administration on the Biologics License Application for XIAFLEX™ (clostridial collagenase for injection – formerly referred to as AA4500) for the treatment of Dupuytren’s contracture and the approval thereof; the timing of the availability of safety data from the clinical trials for XIAFLEX; the timing of the release of results from the phase IIb trial for XIAFLEX for the treatment of Peyronie’s disease; the timing of the launch of XIAFLEX for the treatment of Dupuytren’s contracture in the U.S.; the timing of the initiation of phase III for XIAFLEX for the treatment of Peyronie’s disease; the Company’s ability to manufacture XIAFLEX at the Company’s Horsham facility in sufficient quantities to meet several years of global launch expectations given annual capacity at current yields; the timing of the filing by Pfizer of the Marketing Authorization Application for XIAFLEX for the treatment of Dupuytren’s contracture in the European Union; competitive developments affecting the Company’s products and product candidates, including generic competition; the success of the Company’s development activities; future Testim market share, prescriptions and sales growth and factors that may drive such growth; size and growth potential of the testosterone replacement therapy market and the gel segment thereof and factors that may drive such growth; the protection for Testim afforded by U.S. Patent No. 7,320,968, and its listing in the Orange Book, the value of extending patent protection for Testim through January 2025, the value and likelihood that patents will be granted from the continuation and divisional applications filed by CPEX Pharmaceuticals, Inc.; the impact of the filing by Upsher-Smith Laboratories, Inc. of an ANDA for a testosterone gel; the Company’s development and operational goals and strategic priorities for fiscal 2009; the ability to fund future operations; and the Company’s expected financial performance during 2009 and financial milestones that it may achieve for 2009, including 2009 net revenues, research and development spending, selling, general and administrative expenses, stock-based compensation expenses, and net loss. All remarks other than statements of historical facts made during this presentation, including but not limited to, statements regarding future expectations, plans and prospects for the Company, statements regarding forward-looking financial information and other statements containing the words “believe,” “may,” “could,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” and similar expressions, as they relate to the Company, constitute forward-looking statements. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2008 under the heading “Risk Factors”, which is on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “Investor Relations - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 3 Focused on Sustainable Long-term Growth Testim XIAFLEX Dupuytren’s XIAFLEX Peyronie’s XIAFLEX New Indications XIAFLEX New Territories |
 4 •PDUFA Date August 28, 2009 •Tentative FDA Arthritis Advisory Committee Meeting (September 16, 2009) •12 month Dupuytren's Contracture safety presentation at medical conference • Publication of CORD I data in top-tier medical journal • U.S. Dupuytren’s Contracture Launch (~ 60 days post-approval) • Top-line results of Peyronie's Phase IIb Study (4Q09) • Pfizer files MAA for Dupuytren’s Contracture in EU (2010) • Initiation of Peyronie's Phase III Studies (2010) Timing Dupuytren’s Peyronie’s Anticipated XIAFLEX Events 2009 2010 |
 5 Pipeline Continues to Advance Note: Seeking partners for Transmucosal film product candidates PRODUCT LATE RESEARCH PRE-CLINICAL PHASE I PHASE II PHASE III MARKET TESTIM ® GEL XIAFLEX™ XIAFLEX™ XIAFLEX™ Transmucosal Film AA4010 Transmucosal Film Transmucosal Film Hypogonadism Dupuytren’s Contracture Peyronie’s Disease Frozen Shoulder Syndrome Overactive Bladder Pain Hormone & Urology |
 6 Strong Strategic Partner for XIAFLEX in EU Validates Market Potential • Compelling economics for Auxilium: • Up-front payment of $75 million • $150 million tied to regulatory milestones • $260 million based on sales milestones • Significant increasing tiered royalties based on sales of XIAFLEX in Pfizer’s territories • Primarily responsible for the global development of XIAFLEX, including all clinical & commercial manufacturing and supply. • Exclusive rights to commercialize XIAFLEX for Dupuytren’s contracture and Peyronie’s disease > 27 member countries of European Union (EU) and 19 other European and Eurasian countries • Primarily responsible for: • EU regulatory activities • All commercialization activities • Phase IV clinical development • First EU only partnership |
 7 XIAFLEX Driver of Long-Term Growth XIAFLEX product and packaging have not been approved by the FDA |
 8 XIAFLEX – Unique, Late-Stage, Blockbuster Opportunity • Potential to be the only effective non-surgical treatment for two high unmet needs: – Dupuytren’s contracture – Peyronie’s disease • Well-characterized mode of action • State of the art biological manufacturing facility • Worldwide rights generate and support growth – Build company in North America – Partnered with Pfizer in EU for Dupuytren’s and Peyronie’s – Opportunity to add additional indications – Rights for other territories or indications could generate additional cash • We believe there are at least 450,000 potential patients annually in U.S. and EU for Dupuytren’s & Peyronie’s indications or > $1 Billion opportunity, based on market research and analysis |
 9 Dupuytren’s Contracture is Debilitating for Patients • Excessive collagen deposition in fascia of hand • Nodules represent early, active form • Cords develop over time, are palpable, and result in contractures • Quality of life and daily activities can be significantly affected • Surgery may be reserved for advanced disease due to unpredictable results, complications, long recovery and recurrence/additional surgeries |
 10 Current Treatment Options Require Invasive Surgery with Significant Recuperation or Are Unapproved and Ineffective • Surgery • Non-surgical options > Splinting > Physical therapy > Corticosteroids • Needle fasciotomy/ aponeurotomy • Amputation |
 11 XIAFLEX Offers an Improved Mechanism of Action • Clostridial collagenase is faster and more potent than human collagenase – Activity of XIAFLEX further enhanced by optimizing the ratio of collagenases in the product End products Fast cleavage Human collagenase Human collagenase (MMP) (MMP) Slow cleavage XIAFLEX XIAFLEX End products |
 12 CORD I - Largest Double-blind, Placebo- controlled Trial in Dupuytren’s Contracture • Primary endpoint is reduction in contracture to within 0 - 5 ° of normal > randomized in 2:1 ratio of active to placebo > enrolled a 2:1 ratio of MP:PIP > enrolled 1:1 ratio of less severe to more severe joints • Secondary endpoints to measure clinical improvement and time to success > 50% improvement in contracture > change in primary joint degree of contracture • 12 month safety profile |
  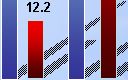         13 CORD I - 1 ° and 2 ° Endpoint Outcomes Should Compare Favorably To Surgery 60 50 40 30 20 10 0 79.3% reduction in contracture for XIAFLEX patients vs. 8.6% reduction for placebo patients (p<0.001) Baseline Last 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% * P < 0.001 Comparison of XIAFLEX vs. Placebo 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% % of Patients Achieving Less Than or Equal to 5 degrees Contractures Post Treatment XIAFLEX (N=203) Placebo (N=103) XIAFLEX (N=203) Placebo (N=103) XIAFLEX (N=203) Placebo (N=103) % of Patients Achieving At Least 50% reduction in Contracture Post-treatment |
 14 XIAFLEX Phase III AE and SAE Rates Should Compare Favorably To Surgical Complications • Common adverse events and rates were consistent with prior XIAFLEX trials Serious Adverse Events Number Rate/Inj. Tendon / Ligament: Confirmed Tendon Rupture 3 0.12% Ligament Injury 1 0.04% Total Tendon / Ligament 4 0.15% Deep Vein Thrombosis 1 0.04% Dupuytren’s Disease 1 0.04% Complex Regional Pain Syndrome 1 0.04% Total 7 0.27% Note: > 2,600 injections in approximately 1000 patients from CORD, JOINT and PK studies as of October 29, 2008. |
 15 XIAFLEX Offers a Non-Invasive Treatment for Dupuytren’s Contracture |
 16 XIAFLEX for Dupuytren’s Contracture Key Clinical Takeaways • Efficacy should compare favorably to surgery – Primary endpoint of 5° reached with statistical significance in multiple studies – Secondary endpoints support commercial uptake • Less severe contractures responded better than more severe contractures • MP joints responded better than PIP joints • 12 month percent recurrence comparable to earlier single center study (~6%) • Safety profile should compare favorably to surgery – No systemic exposure and no systemic hypersensitivity reactions |
 17 XIAFLEX I.P. Position for Dupuytren’s Contracture • U.S. Orphan Drug granted on May 23, 1996 provides exclusivity for 7 years post-approval • Market Exclusivity expected in EU for 10 years post-approval > Data protection granted for 8 years • Method of Use Patent in U.S. through 2014 • Use Patent in France, UK, Sweden through 2018 > Pending in Germany and Denmark • Highly Purified Collagenase product and manufacturing patent filed (if issued, expected expiry 2027) |
 18 XIAFLEX Supply Chain • API produced in Horsham, PA – 2 ° - 8 ° C storage expected at launch – Annual capacity at current yield should be sufficient for several years of global launch expectations – 1 manufacturing train in use and validated; capacity to expand for future demand • Fill and Lyophilization: Hollister-Stier; WA – FDA and EMEA-approved facility – Supplies U.S. and EU markets with injectable forms |
 19 Dupuytren’s Contracture –an Unmet Medical Need • Disease prevalence is estimated at 3%-6% of adult Caucasian population, or 13.5 to 27 Million, in U.S. and EU, but occurs in all populations • Higher prevalence in patients of northern-European descent • Hereditary component in approximately 40% of patients • On average, patients have 2.2 affected joints at the time of diagnosis, and about half of patients have bilateral disease • Recurrence rate in surgical patients > 30% during 1st and 2nd postoperative years > 55% in 10 years • We believe Dupuytren’s contracture is under-diagnosed and under-treated 1,4 3 2 1 1 1 Tubiana 2006. 2 Hueston 1963 3 Skoog 1948 4 Mikkelsen 1976 |
 20 Extensive Market Research Performed with Dupuytren’s Surgeons • Multiple prevalence data sets proved to be highly variable in numbers of patients • Two rounds of quantitative primary research have been commissioned by Auxilium on XIAFLEX for Dupuytren’s contracture • A combined 571 Orthopedic Surgeons have been interviewed to estimate market size (401 in U.S.; 170 in Europe) and 391 have given feedback on potential usage of XIAFLEX (221 U.S.; 170 Europe) • Second study (n=444 total & 340 in depth) was designed to provide 95% confidence level and 7.5% margin of error |
 21 Source: 2006 Auxilium research and analysis Annually, >240,000 Dupuytren’s Candidates Could Exist between U.S. & EU |
 22 Sustainable Blockbuster Market for Dupuytren’s Contracture Anticipated • High disease prevalence – Based on literature, ~ 13.5M to 27M patients in U.S. and EU, however only ~ 1M patients annually seeking treatment in U.S. and EU • Sustainable patient pool – New patients within an ageing population • Average of 2.2 joints affected at time of diagnosis • ~ 50% of patients have bilateral disease – Disease progression to additional joints – High recurrence rate for surgery • No non-invasive competition - pricing expected to be comparable to surgery • Market development represents upside |
 23 1 Smith BH. Am J Clin Pathol. 1966;45:670-678. 2 Somers KD, Dawson DM. J Urol. 1997;157:311-315. Peyronie’s Disease is a Devastating Disorder • Scarring phenomenon affecting the tunica albuginea • Plaques show excessive collagen deposition • Potential Symptoms > Pain with erection, penile curvature/ deviation, penile shortening, indentations, and/or erectile dysfunction > May experience difficulty with sexual intercourse, loss of self-esteem, and depression • There are no approved therapies for the treatment of Peyronie’s disease 1 2 |
 24 Peyronie’s Disease - an Unmet Medical Need • Prevalence of Peyronie’s disease is estimated at 1 in 20 adult men > Actual prevalence may be higher, based on autopsies • Prevalence rate increases with age > The average age of disease onset is 53 years3 • High association with other diseases such as: > Diabetes, erectile dysfunction (ED), Dupuytren’s contracture, plantar fascial contracture, tympanosclerosis, gout, and Paget’s disease • We believe Peyronie’s disease is under-diagnosed and under-treated 1 2 4 1 Schwarzer U, The prevalence of Peyronie’s disease: Results of a large survey. BJU Int 2001;88:727-30; Mulhall et al: J Urology 2004:171: 2350 - 2353;Rhoden et al: Int J. Impot Res 2001 :13 : 291 - 293; La Pera et al : EUR Urology 2001: 40 :525 - 530. 2 Smith BH. Am J Clin Pathol. 1966;45:670-678. 3 Lindsay MB, J Urol. 1991;146:1007-1009. 4 Nyberg L, J Urol.128: 48, 1982 |
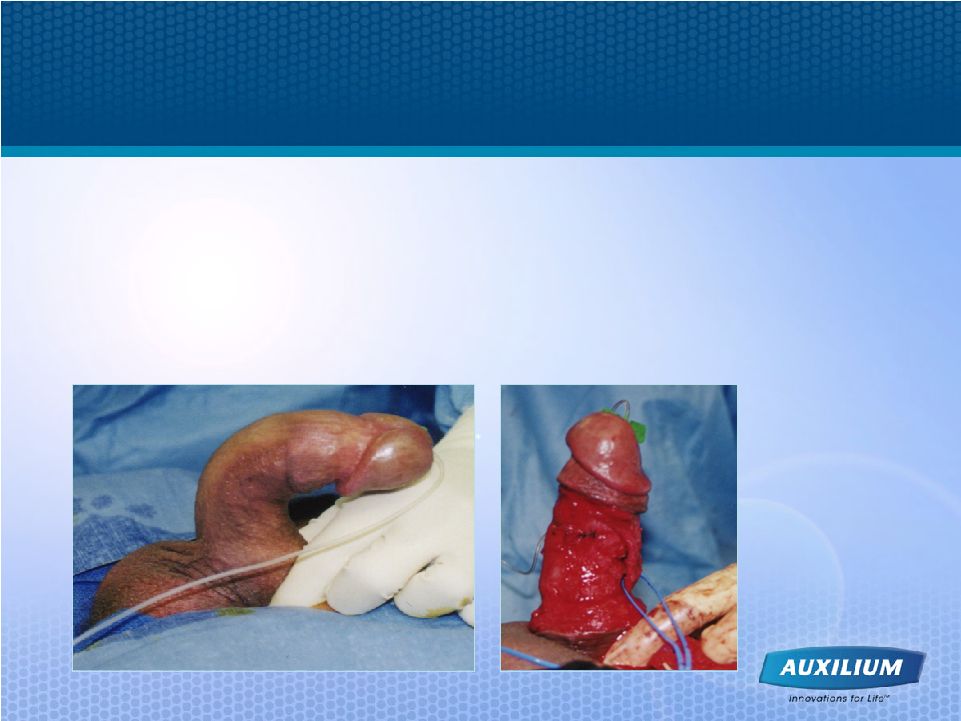 25 Current Surgical Options - Treatment of Last Resort • The goal of surgery is simply to make the two sides of the penis equal in size through reduction of the longer side • Post-surgically, graft or prosthetic may be required • Cost of surgery in U.S. is ~$10-12K • Patients are highly motivated to attempt other treatments first |
 26 Unapproved Treatments for Peyronie’s Disease Have Been Used With Little Reported Success • Verapamil • Vitamin E • Colchicine • Potassium aminobenzoate (Potaba) • Tamoxifen • Interferon alpha-2a • Corticosteroids • Energy transfer treatment including extracorporeal shock wave therapy (ESWT), laser and ultrasound therapy, and orthovoltage radiation |
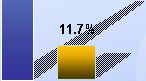
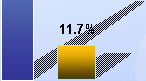
             27 Promising Phase II Data with 25% Reduction in the Angle of Deviation for Most Patients 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% 100% 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% |
 28 Peyronie’s Phase IIb Study Top-line Data Expected in 4Q09 • Study designed to assess the safety and efficacy with XIAFLEX. – XIAFLEX will be administered two times a week every six weeks for up to three treatment cycles (2 x 3) – ~120 patients – 12 U.S. sites • Patients must be able to maintain a rigid erection and have a penile contracture between 30 and 90 degrees. – Stratification occurs by the degree of penile curvature (i.e. 30 ° to 60 ° versus > 60 ° ). • Active dosing period has been completed. – Patients will be monitored for 36 weeks following first injection. |
 29 Peyronie’s Phase IIb Study Design Arm Study Drug Plaque Modeling Targeted Number of Patients A XIAFLEX yes 45 B Placebo yes 15 C XIAFLEX no 45 D Placebo no 15 120 • Patients will be randomized in a 3:1 ratio of XIAFLEX to placebo and a 1:1 ratio to receive penile plaque modeling or no modeling. |
 30 Peyronie’s Phase IIb Trial Utilizes a Novel Patient Reported Outcome (PRO) • Study is designed to validate Auxilium’s proprietary Peyronie’s Patient Reported Outcome (PRO) questionnaire. • PRO will measure four domains of patients’ sexual quality of life, over a 36 week period: – penile pain – Peyronie’s disease bother – intercourse discomfort – intercourse constraint • PRO likely to be used as a primary efficacy endpoint in Phase III clinical trials. |
 31 Extensive Market Research Performed with Peyronie’s Surgeons • Two rounds of quantitative primary research have been commissioned by Auxilium on XIAFLEX for Peyronie’s disease • A combined 575 Urologists have been interviewed to estimate market size (415 in U.S.; 160 in Europe) and 383 have given feedback on potential usage of XIAFLEX (223 U.S.; 160 Europe) • Second study (n=472 total & 333 in depth) was designed to provide 95% confidence level and 7.5% margin of error |
 32 Source: 2006 Auxilium research and analysis Annually, >210,000 Peyronie’s Candidates Could Exist between U.S. and Europe |
 33 XIAFLEX I.P. Position for Peyronie’s Disease • U.S. Orphan Drug granted on March 12, 1996 provides exclusivity for 7 years post-approval • Market Exclusivity in EU could be extended to 11 years on approval of additional indication • Method of Use Patent in U.S. through 2019 • Use Patent Granted in France, UK, and Ireland (expiry 2020) > Pending in Germany, Denmark, and Norway • Highly Purified Collagenase product and manufacturing patent filed (if issued, expected expiry 2027) |
 34 Testim ® 1% Testosterone Gel: Near-Term Growth Driver |
 35 Testim ® Maintains Double-Digit Growth in Testosterone Replacement Therapy Market *Mulligan T. et al. Int J. Clin Pract 2006 • Proprietary, topical 1% testosterone gel > Once-a-day application > Favorable clinical and commercial profile • Recent study indicates 39% of U.S. males over 45 yrs are hypogonadal* > We estimate that <10% of affected population receives treatment • We believe diagnosis is increasing through education and awareness |
         36 Patient Results Were Proven in Clinical Studies Total Testosterone 2,000 3,000 4,000 5,000 6,000 Mean AUC 0-24 (ng*h/dL) Testim AndroGel Free Testosterone 0 50 100 150 200 250 Mean AUC 0-24 (ng*h/dL) Testim AndroGel • 16 clinical studies involving approx. 1,800 patients > largest placebo-controlled study ever conducted • Clinical trial of Testim ® vs. AndroGel ® > Testim provides 30% higher testosterone absorption (p<0.001) Note: Adjusted geometric means (CV b %) of a single-dose (50 mg testosterone), randomized, complete crossover study of 29 hypogonadal men. |
 37 Gels Continue to Drive Significant Growth in TRT Marketplace Source: IMS data $35 $117 $200 $287 $340 $383 $449 $690 $563 $49 $59 $77 $118 $210 $302 $399 $459 $499 $568 $685 $819 0 100 200 300 400 500 600 700 800 900 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 Gel Patch Oral Injectables ($ in millions) Gel Segment Growth ($) Mar 2009 L12M: 26.7% |
 38 Testim® Quarterly Net Revenues and Scripts ($ in millions) Continuing Track Record of Consistent Revenue Growth 1Q09 Net Testim Revenues Y/Y Growth: +24.7% 1Q09 TRX Y/Y Growth: +14.1% Source: Auxilium and IMS data $42.8M $68.9M $95.7M $125.1M $33.8M |
 39 Testim Patent Coverage *Note: The referenced Testim patents are owned by CPEX Pharmaceuticals, Inc. U.S. • U.S. Patent # 7,320,968* covering method of use claims for Testim issued January 22, 2008; expires 2025 • Divisional application and continuations filed with U.S. PTO in late 2007 with a plan to protect additional inventions and seek additional claims • Upsher-Smith Laboratories, Inc. filed an ANDA with paragraph IV certification referring to the ’968 patent; AUXL filed lawsuit under Hatch-Waxman on Dec. 4, 2008; 30 month stay expires June 2011 ROW • Patent issued in Canada; expires 2023 • Patent issued in Europe; expires 2023 • Patents granted and applications pending in numerous countries worldwide |
 40 2009 Q1 ’09 Q1 ‘08 Guidance Revenues $34.7 $27.1 $150-155 R&D Expense $13.5 $13.2 $45-50 SG&A Expense $26.4 $21.0 $120-130 Net Loss ($13.2) ($12.3) ($55-60) Stock – Based Comp Expense $4.5 $2.1 $16-19 Cash & Cash Equivalents $90.2 Q1 ‘09 Financial Results and 2009 Guidance ($ Millions) Currently approximately 42.7 million shares issued & outstanding. Additional 6.2 million shares subject to issuance from stock options & warrants |
 41 Strategic Priorities in 2009 • Continue our pre-launch commercialization efforts and be prepared to launch in ~60 days after approval; • Release top-line data from the XIAFLEX Peyronie’s disease Phase IIb trial, which we anticipate in 4Q09; • Work diligently with Pfizer to prepare for a 2010 EU filing for XIAFLEX in Dupuytren's contracture; • Vigorously defend Testim IP and continue to grow scripts. |
 42 Focused on Sustainable Long-term Growth Testim XIAFLEX Dupuytren’s XIAFLEX Peyronie’s XIAFLEX New Indications XIAFLEX New Territories |
 43 Management Contact James E. Fickenscher/CFO Auxilium Pharmaceuticals, Inc. (484) 321-5900 jfickenscher@auxilium.com William Q. Sargent Jr./ VP IR Auxilium Pharmaceuticals, Inc. (484) 321-5900 wsargent@auxilium.com |
 44 June 2009 (NASDAQ: AUXL) |