 September 2010 (NASDAQ: AUXL) Exhibit 99.1 1 |
 2 Safe Harbor Statement We will make various remarks during this presentation that constitute “forward-looking statements” for purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995, including statements regarding the pricing, time to market, size of market, growth potential and therapeutic benefits of the Company’s product candidates, including those for the treatment of Peyronie’s disease and Frozen Shoulder syndrome; the size of the Dupuytren’s market in the U.S. and EU; the potential for XIAFLEX to be a blockbuster opportunity; the potential for XIAFLEX to be used in multiple indications; interpretation of market research data; competition within certain markets relevant to the Company’s product candidates; interpretation of clinical results, including the efficacy and tolerability of the Company’s products and product candidates; the timing of the commencement and completion of clinical trials and the timing of reporting of results therefrom; the effect of the identified leading indicators on the success of the XIAFLEX launch and future net revenues; the ability to obtain reimbursement in the U.S. for XIAFLEX for the treatment of Dupuytren’s; the timing of new reimbursement codes for XIAFLEX; the average number of cords that Dupuytren’s contracture patients will have treated with XIAFLEX and the average number of vials used to treat each cord; the timing of the initiation of phase III for XIAFLEX for the treatment of Peyronie’s disease; physicians and sites that are moving from test drive to increasing usage; the approval of the Marketing Authorization Application for XIAFLEX for the treatment of Dupuytren’s contracture in the European Union; competitive developments affecting the Company’s products and product candidates, including generic competition; the success of the Company’s development activities; future Testim market share, prescriptions and sales growth and factors that may drive such growth; size and growth potential of the testosterone replacement therapy market and the gel segment thereof and factors that may drive such growth; the protection for Testim afforded by U.S. Patent No. 7,320,968, and those issued on October 27, 2009 and their listing in the Orange Book, the value of extending patent protection for Testim through January 2025, the value and likelihood that patents will be granted from the continuation applications filed by CPEX Pharmaceuticals, Inc.; the impact of the filing by Upsher-Smith Laboratories, Inc. of an ANDA for a testosterone gel; the Company’s development and operational goals and strategic priorities for fiscal 2010; the ability to fund future operations; and the Company’s expected financial performance during 2010 and financial milestones that it may achieve for 2010, including 2010 Testim net revenues, research and development spending, selling, general and administrative expenses, stock-based compensation expenses and the range of XIAFLEX net revenues for the third quarter 2010. All remarks other than statements of historical facts made during this presentation, including but not limited to, statements regarding future expectations, plans and prospects for the Company, statements regarding forward-looking financial information and other statements containing the words “believe,” “may,” “could,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “expect,” and similar expressions, as they relate to the Company, constitute forward-looking statements. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in Auxilium's Annual Report on Form 10-K for the year ended December 31, 2009 and in Auxilium’s Quarterly Report on Form 10-Q for the period ended June 30, 2010 under the heading "Risk Factors“, which are on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “Investor Relations - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 Opportunities to Build Shareholder Value Note: Seeking partners for Transmucosal film product candidates 3 |
 4 |
 5 XIAFLEX – Unique Blockbuster Opportunity • Potential to be the only effective non-surgical treatment for two high unmet needs: Approved on February 2, 2010 for Dupuytren’s Contracture in U.S. Phase III trial in Peyronie’s disease expected to start by end of 2010 • Well-characterized mode of action • Worldwide rights support growth Build company in North America Partnered with Pfizer in EU for Dupuytren’s and Peyronie’s Opportunity to add additional indications Rights for other territories or indications could generate additional cash • We believe worldwide peak revenues for XIAFLEX could be in excess of $1 Billion annually |
 6 • Excessive collagen deposition in fascia of hand • Nodules represent early, active form • Cords develop over time, are palpable, and result in contractures • Quality of life and daily activities can be significantly affected • Surgery has been the current standard of care and may be reserved for advanced disease due to unpredictable results, complications, long recovery and recurrence/additional surgeries Dupuytren’s Contracture is Debilitating for Patients |
 7 • Surgery • Needle fasciotomy/ aponeurotomy • Amputation Treatment Options Historically Limited to Invasive Surgery with Significant Recuperation or Are Unapproved and Ineffective • Non-surgical options > Splinting > Physical therapy > Corticosteroids |
 8 It Is About the Patient Pre-XIAFLEX treatment 30 days post-single injection of XIAFLEX Immediately post-operative Intra-operative open fasciectomy Pictures courtesy of Dr. Clayton Peimer |
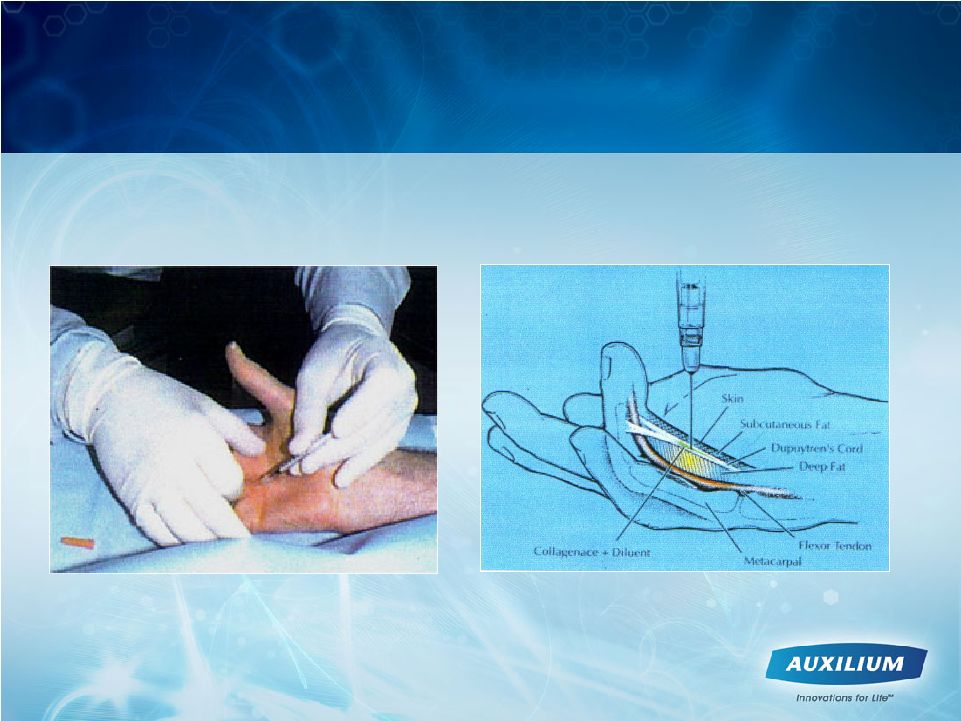 9 XIAFLEX Offers a Nonsurgical Treatment for Dupuytren’s Contracture |
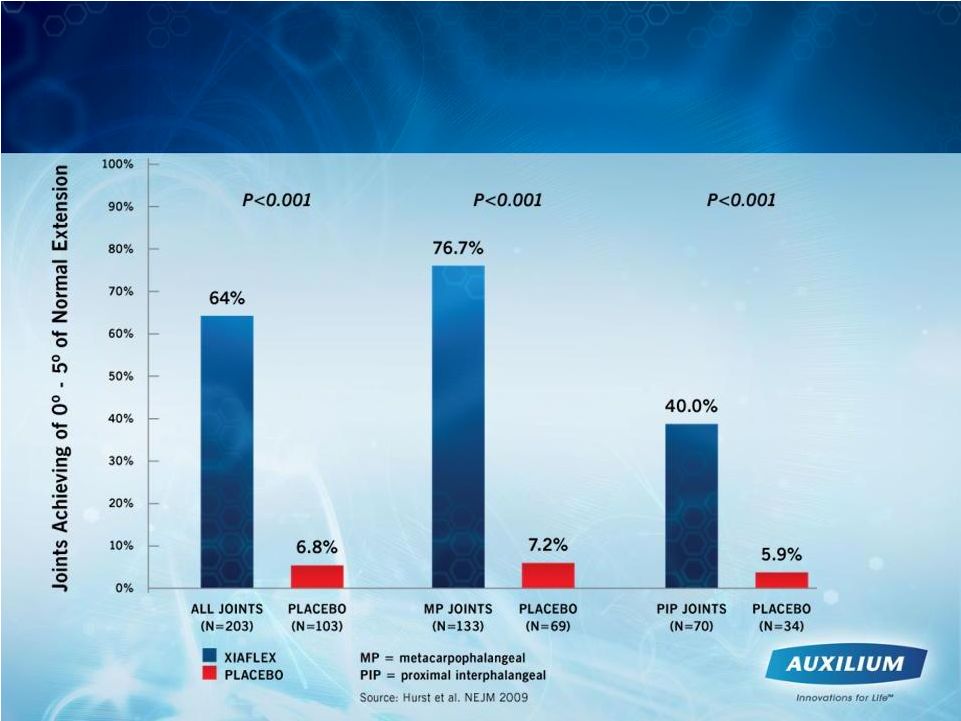 A Significant Number of Dupuytren’s Contracture Patients Benefited from Treatment with XIAFLEX 10 |
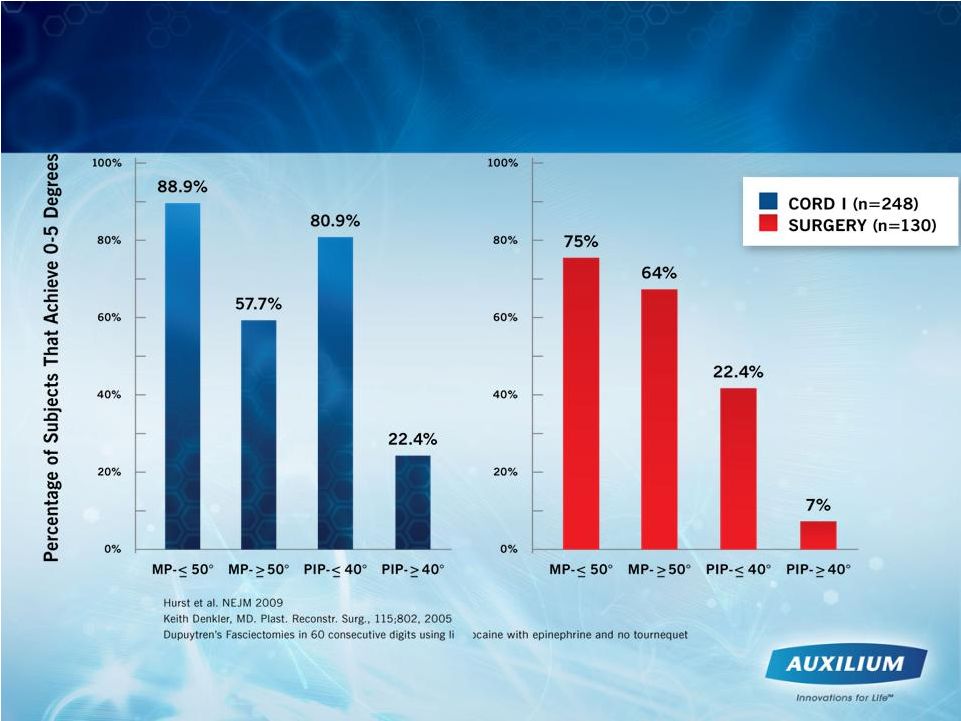 Early Intervention with XIAFLEX May Be the Most Effective Treatment Approach N=125 11 |
 12 • Most treatment related adverse events were mild or moderate in intensity and resolved without intervention within a median of 10 days across studies • Serious Adverse Events possibly related to treatment were limited in number (10 in total; 0.39% of >2600 injections) – 4 SAEs were tendon and ligament damage • No deaths, clinically meaningful changes in grip strength, arterial injuries or nerve injuries related to XIAFLEX reported • No clinically meaningful changes in laboratory values • No clinically meaningful systemic allergic reactions XIAFLEX Was Well-Tolerated in Phase III Studies |
 13 Contracture Recurrence Affects Patients • The earliest reports of recurrence for surgery were seen in 22% of female and 19% of male Dupuytren’s contracture patients at a mean of 12 months following fasciectomy . Additionally, a rate of up to 34% has been reported within the first 2 years following surgery . • Abstract for a prospective trial comparing needle fasciotomy to open fasciectomy reported a recurrence rate for needle fasciotomy of 85% at a mean of 2.3 years . • XIAFLEX Long term extension study of phase III trials will follow recurrence rates up to 5 years. Two year data now available. 1 2 3 (1) Anwar et al., The Journal of Hand Surgery, Vol. 32A No. 9 November 2007 ; 1423-1428 (2) Leclercq C. Epidemiology. In: Tubiana R, Leclercq C, Hurst LC, Badalamente MA, Mackin EJ, eds, Dupuytren’s disease. Martin Dunitz Ltd, London, 2000;239-249. (3) A.L. Van Rijssen, 2010 International Symposium on Dupuytren’s Disease |
 14 Two Year Recurrence Rate of 19.3% for XIAFLEX in Dupuytren’s Contracture All Joints MP Joints PIP Joints Patients from All Phase III Studies (n=950) 1,568 920 648 Patients Enrolled in Extension Study (n=634) 1,065 641 424 Patients Successfully Treated and Enrolled in Extension Study (n=474) 619* 449* 170 Joints with Recurrence (n/%) (119/618) 19.3% (61/448) 13.6% (58/170) 34.1% Note: No patients at Year 2 had been retreated with commercial XIAFLEX * One patient had unrelated hand surgery and post-operative bandaging prevented an accurate assessment of recurrence at 2 years |
 15 Launch Update Through August 2010 |
 16 • Position XIAFLEX as a paradigm changing non-surgical treatment alternative for physicians, payers and patients • Achieve high awareness among targeted physicians and diagnosed patients • Get targeted physicians through the sales cycle – from interest to injection to reimbursement • Educate and assist physician offices with unfamiliar reimbursement processes at each step of the way Key Objectives for U.S. XIAFLEX Launch in 2010 |
 1. Dupuytren’s Disease – Tubiana, LeClerq, Hurst, Badalamente, Mackin 2. SDI Claims Data Based Projections 3. Medicare Data Based Projections (BESS database used, Medicare 5% database also used to validate numbers) 4. Auxilium Research (Patient Segmentation, Forecast Research, WK/AMA Databases) Sources: Targeting the Eligible Patient Population Should Drive Development of the U.S. Market to Its Full Potential 17 |
 18 XIAFLEX Launch Strategy is Tailored to Customer Needs • ~ 7,000 target physicians – Hand surgeons, plastic surgeons, orthopedic surgeons, general surgeons, rheumatologists • ~ 100 field based personnel – Sales reps, sales managers, and reimbursement specialists • Medical science liaisons – Key opinion leader and regional opinion leader support |
 19 Unique Sales Cycle for Targeted Physicians Physician Interest, Training & Enrollment Successful XIAFLEX XPERIENCE |
 20 Changing Treatment Paradigm and Reimbursement Cycle May Drive Physician Adoption Rate Prior to treatment decision Physician must go through: •Awareness •Interest •Training / Enrollment into XIAFLEX XPERIENCE program •Reimbursement education / comfort •Patient Identification / counseling 11 – 21 weeks from treatment decision through receipt of payment possible |
 21 • Accounts need customized support every step of the way • Miscellaneous J and CPT codes being used; component and bundled approaches are being accepted • C-code for institutional use effective as of July 1, 2010 • CMS has recommended a J-Code for XIAFLEX which will be effective January 1, 2011 • CPT code specific to XIAFLEX may come in 2012 Reimbursement Processes Are New to Target Physicians |
 22 193 598 1132 1420 1628 1746 1,853 98 394 755 957 1117 1216 1,307 0 200 400 600 800 1000 1200 1400 1600 1800 2000 Feb Mar Apr May Jun Jul Aug PHYSICIANS SITES Over 1,800 physicians and 1,300 sites have enrolled |
 Leading Indicators Trending Positively - Cumulative Call Center Volume 23 |
 24 Leading Indicators Trending Positively - Cumulative unique sites that have ordered XIAFLEX 15 60 137 243 348 473 0 50 100 150 200 250 300 350 400 450 500 Mar Apr May Jun Jul Aug number of sites |
 Some Sites Moving from Test Drive to Increasing Usage Injections per Site 25 |
 ~91% of insured lives have access 26 |
 Out of Pocket Costs In Line With Expectations % Of Treatment Cost Paid by Patient 55% 5% 7% 8% 23% 2% 0% 10% 20% 30% 40% 50% 60% 0% Up to 2% 2.1-9.9% 10-19.9% 20% >20% A significant portion of Medicare patients with a 20% liability have secondary insurance, which may cover a portion of this cost 27 |
 28 Distribution Network is Operating Smoothly |
 29 Direct to Patient Program Launching |
 30 Other Patient Programs Launching • XIAFLEX facebook.com webpage • XIAFLEX.com re-design • XIAFLEX and Dupuytren’s contracture WebMD pages • Handraisers.com |
 31 We Believe Launch Strategy Is Sound: • Product efficacy and safety are consistent with clinical data • Access to targeted physicians is good • Usage is characterized by “test driving”, which is typical for paradigm changing treatment • Payers willing to provide access to drug and procedures • All distribution channels active and operating as expected • Leading indicators trending positively |
 32 Sustainable Blockbuster Market for Dupuytren’s Contracture Anticipated • High disease prevalence Based on literature, ~ 6M to 11M patients in U.S. have a Dupuytren’s pit, nodule or cord, but only ~300,000 new patients annually diagnosed in U.S. • Sustainable patient pool New patients within an ageing population Average of 1.5 cords expected to be treated and average 1.1 injections per cord ~ 50% of patients have bilateral disease Disease progression to additional joints High recurrence rate for surgery 1,2 • No nonsurgical competition • Market development represents upside 1 Mikkelsen 1976 2 Tubiana 2006 |
 33 XIAFLEX I.P. Position for Dupuytren’s Contracture • U.S. Orphan Drug designation granted on May 23, 1996 provides exclusivity for 7 years post-approval in February 2010 • Market Exclusivity expected in U.S. for 12 years post-approval, per 2010 Patient Protection and Affordable Care Act • Market Exclusivity expected in EU for 10 years post-approval Data protection granted for 8 years • Method of Use Patent in U.S. through 2014 • Use Patent in France, UK, Sweden through 2018 Pending in Germany and Denmark • Highly Purified Collagenase product and manufacturing patent filed (application # 11/699,302; if issued, expected expiry 2027) |
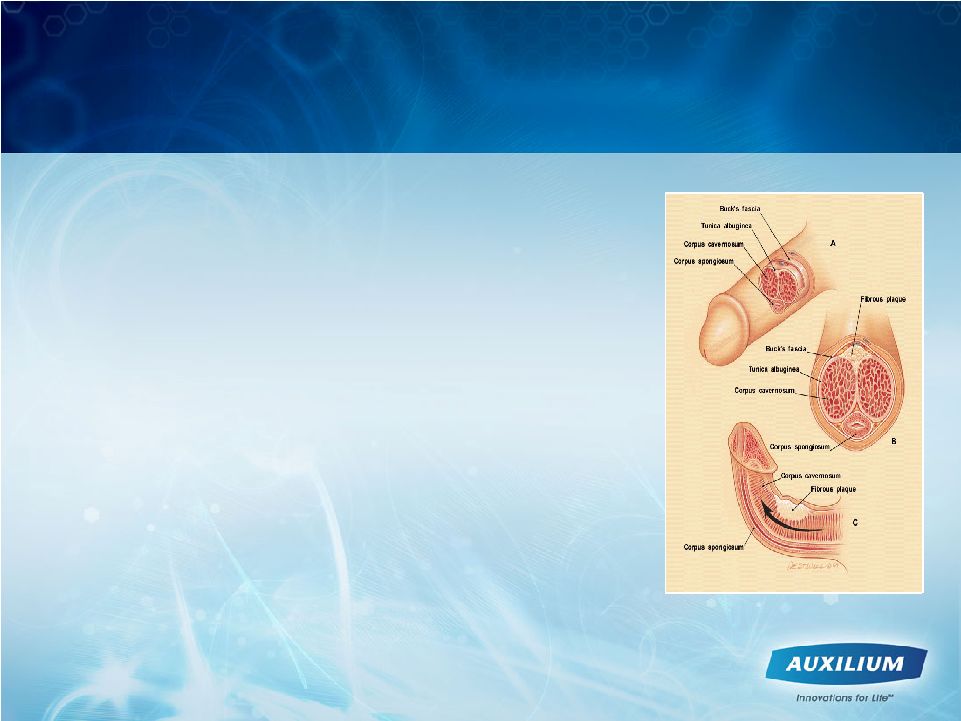 34 1 Smith BH. Am J Clin Pathol. 1966;45:670-678. 2 Somers KD, Dawson DM. J Urol. 1997;157:311-315. • Scarring phenomenon affecting the tunica albuginea 1 • Plaques show excessive collagen deposition 2 • Potential Symptoms > Pain with erection, penile curvature/ deviation, penile shortening, indentations, and/or erectile dysfunction > May experience difficulty with sexual intercourse, loss of self-esteem, and depression • There are no approved therapies for the treatment of Peyronie’s disease Peyronie’s Disease is a Devastating Disorder |
 35 1 Schwarzer U, The prevalence of Peyronie’s disease: Results of a large survey. BJU Int 2001;88:727-30; Mulhall et al: J Urology 2004:171: 2350 - 2353;Rhoden et al: Int J. Impot Res 2001 :13 : 291 - 293; La Pera et al : EUR Urology 2001: 40 :525 - 530. 2 Smith BH. Am J Clin Pathol. 1966;45:670-678. 3 Lindsay MB, J Urol. 1991;146:1007-1009. 4 Nyberg L, J Urol.128: 48, 1982 • Prevalence of Peyronie’s disease is estimated at 1 in 20 adult men 1 > Actual prevalence may be higher, based on autopsies 2 • Prevalence rate increases with age > The average age of disease onset is 53 years 3 • High association with other diseases such as: > Diabetes, erectile dysfunction (ED), Dupuytren’s contracture, plantar fascial contracture, tympanosclerosis, gout, and Paget’s disease 4 • We believe Peyronie’s disease is under-diagnosed and under-treated Peyronie’s Disease - an Unmet Medical Need |
 36 • The goal of surgery is simply to make the two sides of the penis equal in size through reduction of the longer side • Post-surgically, graft or prosthetic may be required • Cost of surgery in U.S. is ~$10-12K • Patients are highly motivated to attempt other treatments first Current Surgical Options Are the Treatment of Last Resort |
 37 • Verapamil • Vitamin E • Colchicine • Potassium aminobenzoate (Potaba) • Tamoxifen • Interferon alpha-2a • Corticosteroids • Energy transfer treatment including extracorporeal shock wave therapy (ESWT), laser and ultrasound therapy, and orthovoltage radiation Unapproved Treatments for Peyronie’s Disease Have Been Used With Little Reported Success |
 38 Promising Phase IIb Data in Peyronie’s Disease • Overall – Significant XIAFLEX effect – Reduction of penile curvature – Improvement in Peyronie’s Disease Bother (PRO Domain) • XIAFLEX With Modeling – Significant effect in both penile curvature and Peyronie’s disease bother endpoints • XIAFLEX Without Modeling – Not significant - Due to strong placebo response • Safety profile consistent with previous XIAFLEX studies – Well tolerated – Immunogenicity profile expected – Injection site bruising, edema, pain – most common – No drug related SAEs – No systemic immunologic events |
 39 Strategic Partnership with Pfizer for XIAFLEX in EU • Strong working relationship with goal to obtain EMA approval • Auxilium is primarily responsible for the global development of XIAFLEX, including all clinical & commercial manufacturing and supply. • Pfizer is primarily responsible for: European territory regulatory activities for Dupuytren’s and Peyronie’s All European territory commercialization activities for Dupuytren’s and Peyronie’s All European territory phase IV clinical development for Dupuytren’s and Peyronie’s • Compelling economics for Auxilium: Up-front payment of $75 million $150 million tied to regulatory milestones ($15M received for MAA acceptance in 1Q10) $260 million based on sales milestones Significant increasing double-digit tiered royalties based on sales of XIAFLEX in Pfizer’s territories • First EU only partnership for Pfizer |
 40 Testim ® 1% Testosterone Gel |
 41 *Mulligan T. et al. Int J. Clin Pract 2006 • Proprietary, topical 1% testosterone gel > Once-a-day application > Favorable clinical and commercial profile • Recent study indicates 39% of U.S. males over 45 yrs are hypogonadal* > We estimate that <10% of affected population receives treatment • We believe diagnosis is increasing through education and awareness Testim ® Maintains Double-Digit Growth in Testosterone Replacement Therapy Market |
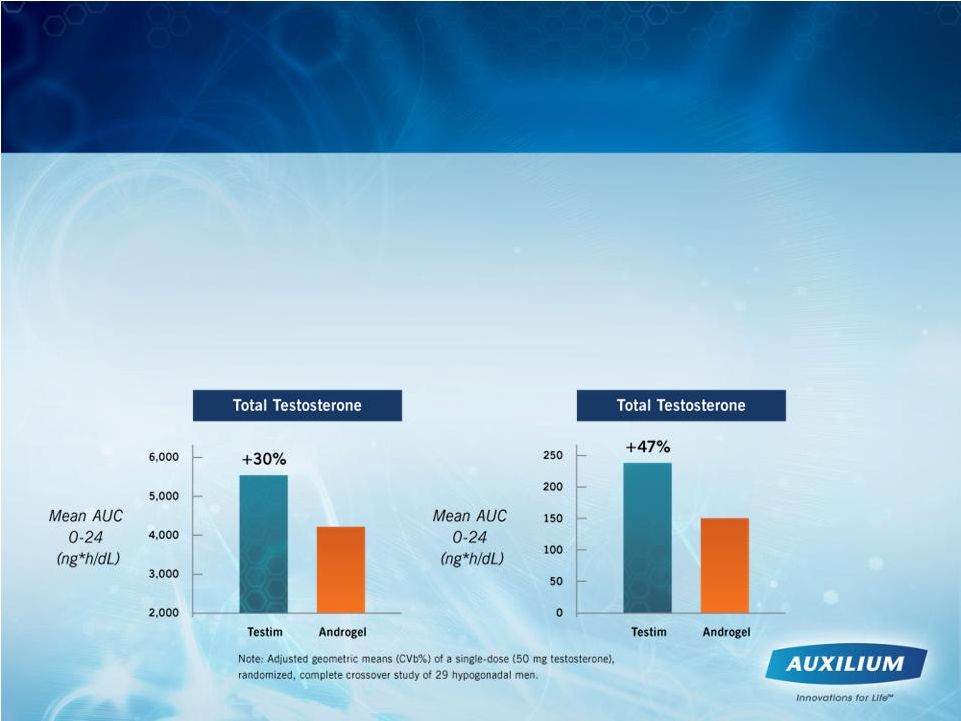 • 16 clinical studies involving approx. 1,800 patients > largest placebo-controlled study ever conducted • Clinical trial of Testim vs. AndroGel ® > Testim provides 30% higher testosterone absorption (p<0.001) Patient Results Were Proven in Clinical Studies 42 |
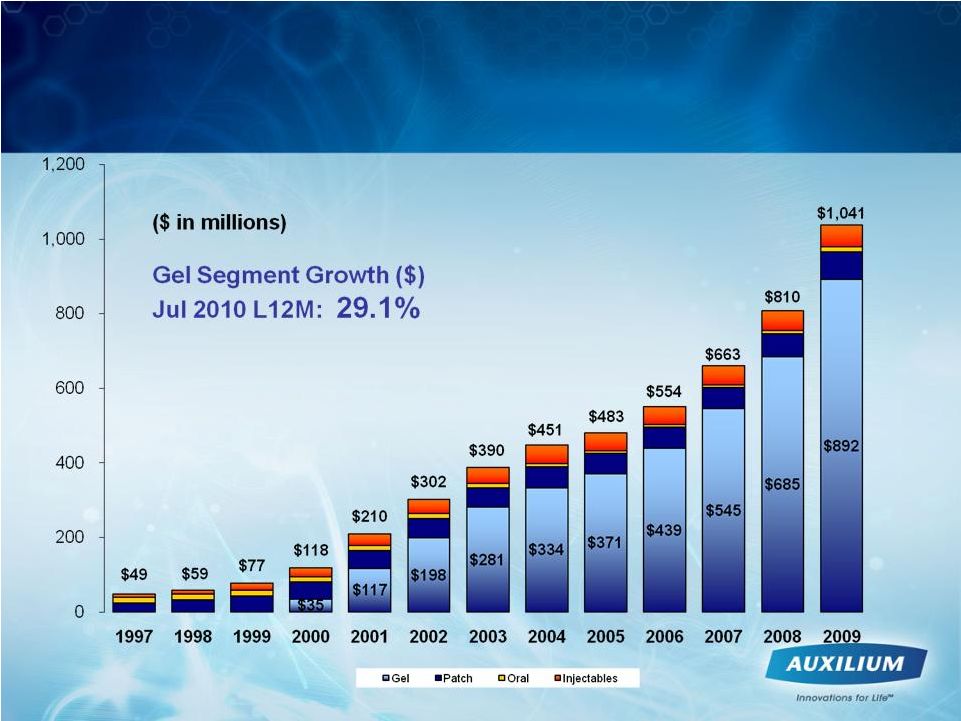 43 Gels Continue to Drive Significant Growth in TRT Marketplace Source: IMS data |
 Continuing Track Record of Consistent Revenue Growth 44 |
 45 *Note: The referenced Testim patents are owned by CPEX Pharmaceuticals, Inc. U.S. • U.S. Patent # 7,320,968* covering method of use claims for Testim issued January 22, 2008; expires 2025 • 6 additional U.S. Patents (# 7,608,605 through 610*) covering method of use claims for Testim issued October 27, 2009; expire 2023 • Upsher-Smith Laboratories, Inc. filed an ANDA with paragraph IV certification referring to the ’968 patent; AUXL filed lawsuit under Hatch-Waxman on Dec. 4, 2008; 30 month stay intact ROW • Patent issued in Canada; expires 2023 • Patent issued in Europe; expires 2023 • Patents granted and applications pending in numerous countries worldwide Testim Patent Coverage |
 46 • Auxilium’s Citizen’s Petition was filed in February 2009 and FDA response was received in August 2009. • FDA has agreed with some of the statements we made in our Citizen's Petition regarding the testing required for generic versions of Testim and disagreed with other statements. • Although not commenting upon any filing in particular, the FDA did state that "The practical effect of this determination is that any application for a testosterone gel product that has different penetration enhancers than the reference listed drug cannot be submitted as an ANDA and, instead, will have to be submitted as an NDA under section 505(b) of the Act.“ FDA Has Granted Our Citizen's Petition In Part And Denied It In Part |
 47 2010 1H ’10 1H ’09 Guidance Testim Revenues $91.6 $72.1 $185-195 XIAFLEX Revenues $4.3 $1.8 N/A R&D Expense* $19.6 $27.3 $50-60 SG&A Expense* $77.3 $55.8 $160-170 Net Loss ($22.1) ($26.0) N/A Stock – Based Comp Expense (* included) $8.4 $9.4 $22-25 Cash & Cash Equivalents $165.0 2010 Financial Results and 2010 Guidance ($ Millions) Currently, approximately 47.7 million shares of common stock outstanding, plus 6.3 million outstanding options to purchase shares of our common stock |
 48 Auxilium’s Plan to Provide Visibility on XIAFLEX Launch • For 3Q 2010, we anticipate that XIAFLEX net revenues will be in the range of $5.5 million to $6.5 million, including approximately $1.1 million in revenue recognized from milestones previously received under the Pfizer contract. • We will continue to provide quarterly revenue guidance through 2010. |
 49 Strategic Priorities in 2010 • Execute the launch in the U.S. for XIAFLEX in Dupuytren’s contracture; • Support Pfizer in the ongoing regulatory review of the EU Dupuytren’s submission and prepare for their eventual launch; • Commence phase III for XIAFLEX in Peyronie’s disease by year end of 2010; • Advance XIAFLEX new indication(s); and, • Continue to maximize Testim revenues while vigorously defending our intellectual property. |
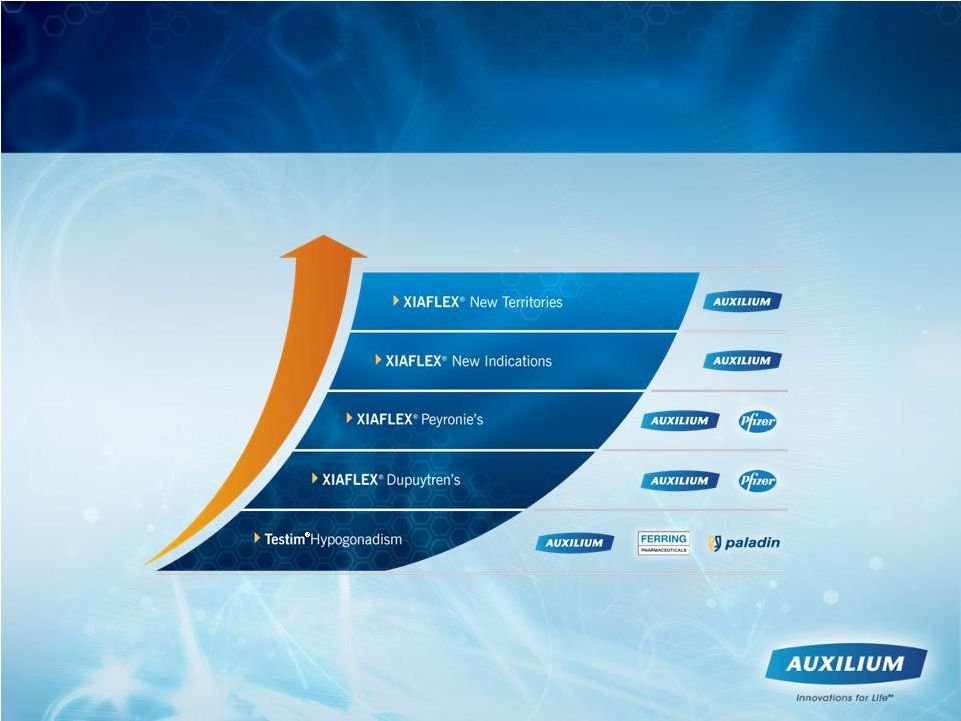 50 Opportunities to Build Shareholder Value Note: Seeking partners for Transmucosal film product candidates |