 1 November 2011 (NASDAQ: AUXL) Exhibit 99.1 |
 2 Safe Harbor Statement We will make various remarks relating to our current plans, potential future events, and what we believe to be the prospects for the Company during this conference call that constitute forward-looking statements for purposes of the Safe Harbor provisions under the Private Securities Litigation Reform Act of 1995. Today our forward-looking statements will cover, among other things, the Company’s expected financial performance, results and strategic priorities during 2011; the impact of organizational changes; marketing strategies for XIAFLEX for Dupuytren’s contracture; the potential for XIAFLEX to change the treatment paradigm and become the standard of care for the treatment of Dupuytren’s; the size of the Dupuytren’s market; the durability of XIAFLEX treatment outcomes; future rates of recurrence; a clinical trial to assess multiple concurrent injections of XIAFLEX and the timing to report results of such trial; the value for XIAFLEX provided by the 2012 Medicare national average reimbursement rates for new procedure codes and the effect of new procedure codes on the reimbursement process and XIAFLEX growth; reductions in Medicare payment rates in accordance with the SGR formula for 2012; the effect of the identified leading indicators on the success of the XIAFLEX launch and future net revenues; additional Phase IV clinical trials for XIAFLEX; peer to peer dialogue programs for U.S. physicians and our U.S. patient education campaign; Pfizer’s ability to commercialize XIAPEX for Dupuytren’s contracture in EU and Eurasian markets; Asahi Kasei Pharma's ability to develop, register for approval and commercialize XIAFLEX for Dupuytren's contracture and Peyronie's disease in Japan; the potential for XIAFLEX to be used in multiple indications, including for the treatment of Peyronie’s, cellulite, Frozen Shoulder syndrome and canine and human lipomas, the effectiveness of XIAFLEX for any such indications, and the success of efforts to advance any such new indications; the design and methodology, and the timing to report results, of the phase III trials for XIAFLEX for the treatment of Peyronie’s; the Peyronie’s, Frozen Shoulder and cellulite markets; the timing, commencement, methodology, safety and efficacy of planned clinical trials for XIAFLEX for the treatments of cellulite, Frozen Shoulder syndrome, and human and canine lipomas; our future relationship with BioSpecifics Technologies Corp.; the growth of the testosterone replacement therapy market, and the growth opportunity for Testim; business development efforts and opportunities to build out the Company’s pipeline; our ability to maximize the value of XIAFLEX and Testim, deliver on our current pipeline, build out our pipeline and create shareholder value. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in Auxilium's Annual Report on Form 10-K for the year ended December 31, 2010 and Quarterly Report on Form 10-Q for the quarter ended September 30, 2011 under the heading “Risk Factors,” which are on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “For Investors - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward- looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward- looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation.. |
 3 Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Biopharmaceutical Company Maximize Value of Testim ® and XIAFLEX ® Deliver on Current Pipeline Build Out Pipeline in Specialty Therapeutic Areas |
 4 XIAFLEX and Testim Are Global Product Opportunities U.S. Canada EU Japan Open ROW Open Open Territory XIAFLEX Testim |
 5 Auxilium Has a 6 Year Net Revenue CAGR of 39.4% 3Q11 XIAFLEX Net Revenues $13.1M (including $10.3M in U.S. revenues) 124% y/y growth 3Q11 Testim U.S. Net Revenues $52.9M, 12% y/y growth |
 6 |
 7 • Excessive collagen in fascia of hand • Nodules or pits are an early, active presentation • Rope-like cords develop in the finger and result in contractures • Quality of life and daily activities affected • Surgery has been reserved for advanced disease due to the nature of the disease, unpredictable results, complications, long recovery time and recurrence/additional surgeries Dupuytren’s Contracture Is Debilitating for Patients and Surgery Has Been the Standard of Care Immediately post-operative Intra-operative open fasciectomy Pictures courtesy of Dr. Clayton Peimer pre-operative open fasciectomy |
 8 We Believe XIAFLEX Is a Paradigm Changing Treatment • Simple, non-invasive injection of XIAFLEX • Established mechanism of action and selective for specific types of collagen • XIAFLEX’s post-approval profile is consistent with clinical trial experience Pre XIAFLEX injection 30 days following XIAFLEX injection |
 9 Strategies for Establishing XIAFLEX as the Standard of Care • Leverage excellent clinical efficacy/safety data • Generate new data to expand treatable population • Facilitate peer to peer dialogue • Streamline the reimbursement process • Activate patients to seek a non-surgical option |
 10 Commercial Changes Designed to Speed Decision Making and Improve Xiaflex Performance • Eliminated EVP of Sales and Marketing position • SVP Sales, and SVP Marketing will report to CEO • Changed Professional Advertising Agency and Public Relations Firm • Changed Senior Director of Marketing |
 11 XIAFLEX Growth Strategy is Tailored to Customer Needs • ~ 6,000 target physicians – Hand surgeons, plastic surgeons, orthopedic surgeons, general surgeons, rheumatologists • ~ 100 field based personnel – Sales reps, sales managers, and reimbursement specialists • Medical science liaisons – Key opinion leader and regional opinion leader support |
 12 We Believe the U.S. Dupuytren’s Opportunity Is Significant 70,000 Surgical 2,3,4 Patients Annually 300,000 Diagnosed Patients Annually 2,3,4 >1 Million Diagnosed Patients 2,4 1. Dupuytren’s Disease – Tubiana, LeClerq, Hurst, Badalamente, Mackin 2. SDI Claims Data Based Projections 3. Medicare Data Based Projections (BESS database used, Medicare 5% database also used to validate numbers) 4. Auxilium Research (Patient Segmentation, Forecast Research, WK/AMA Databases) Sources: $350 Million Market Opportunity $1 Billion Annual Market Opportunity >$3 Billion Market Opportunity |
 13 Highlights of Data Released at ASSH Meeting |
 14 Building on Momentum from ASSH • Introduced Sample program for non-using physicians • Introduced Patient co-pay assistance program • Initiated Direct Response TV Campaign in 7 Ready Markets • Initiated Multi-Cord Phase IV clinical study • Obtained CPT Codes Specific for Xiaflex – Effective Jan.1, 2012 • Obtained Coverage at several BCBS Plans |
 15 Launch Update Through October 2011 |
 16 Leading Indicators Trending Positively - Cumulative Enrollment & sites that have ordered XIAFLEX |
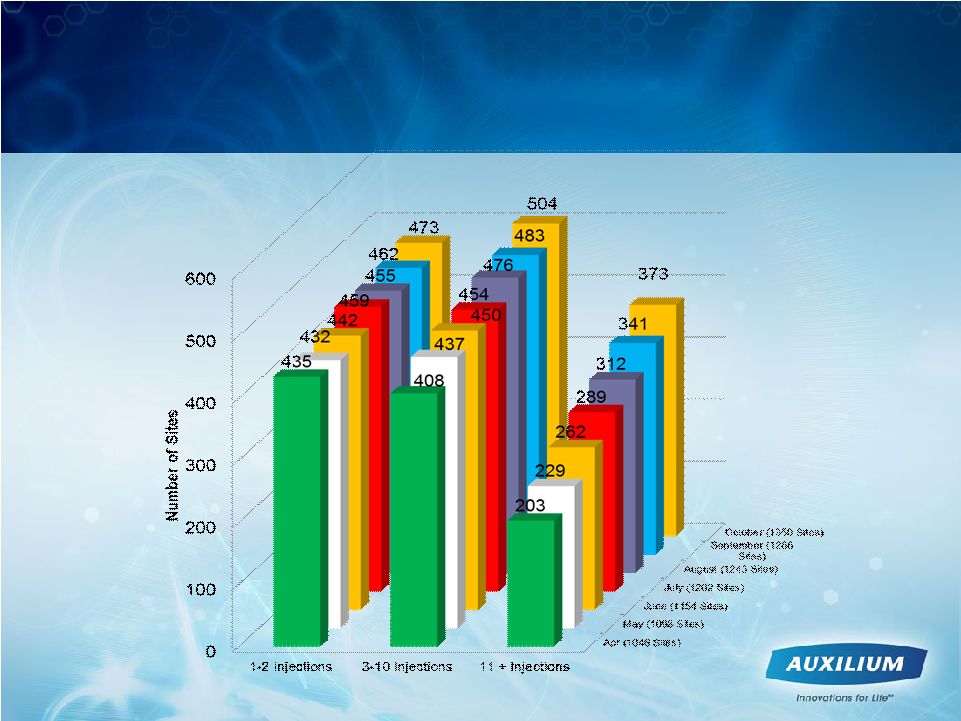 17 Paradigm Changing Progress Cumulative Injections per Site |
 18 2012 Average Medicare Reimbursement Rates for Unique XIAFLEX Procedure Codes* Expected to Provide Appropriate Value Description Code Amount Office or other outpatient visit, for the evaluation and management of a new patient 99203 $102.95 INJECTION, ENZYME (EG, COLLAGENASE), PALMAR FASCIAL CORD (IE, DUPUYTREN'S CONTRACTURE) 20527 $75.77 Finger Extension- MANIPULATION, PALMAR FASCIAL CORD (IE, DUPUYTREN'S CORD), POST ENZYME INJECTION (EG, COLLAGENASE), SINGLE CORD (10 day global) 26341 $98.53 Application of finger splint; static 29130 $39.07 *The above codes represent what the Company expects will be the typical treatment cycle (evaluation & management visit, injection procedure, manipulation procedure and splinting) of a new Dupuytren’s patient who eventually receives a single XIAFLEX injection in a Dupuytren’s cord. These 2012 rates reflect a national Medicare average of the non-facility allowable. As always, coding reflects clinical decisions, and physicians may opt to use other or additional codes based on their medical judgment. The above rates are calculated using the final 2012 relative value unit (RVU) for each of the above codes and multiplying it by the 2011 conversion factor (i.e. RVU x conversion factor = reimbursement rate). Without a change in the law from Congress, Medicare payment rates to providers that are paid under the Medicare Physician Fee Schedule will be reduced by 27.4 percent (via the proposed 2012 conversion factor) for services in 2012. This is the eleventh time the Sustainable Growth Rate (SGR) formula has proposed a payment cut although the reductions have been averted through legislation in ten of the past eleven years, with the exception of allowing the reduction to stand in 2002. |
 19 New CPT Codes for XIAFLEX Should Help Streamline Reimbursement in 2012 • We believe the codes should ease the reimbursement processing burden on office staff, speed up the time to payment and increase predictability of payment. • Based on experience with the J-code, we believe full utilization of the new CPT codes could take some time before affecting site reimbursement practices • 2012 XIAFLEX growth should be helped modestly by new CPT codes |
 20 Testim ® 1% Testosterone Gel • Indicated for testosterone replacement therapy in adult males for hypogonadism • Launched in U.S. in 2003 and EU in 2006 • >8 yrs of use with established safety and efficacy • Applied once daily at 5-10mg > 90% stay on starting dose of 5mg (one tube) > Simple application process and dosing > ~100M daily doses since launch > 16 clinical studies involving approx. 1,800 patients |
 21 Hypogonadism Is an Unmet Need and Under-penetrated Market • 39% of U.S. males over 45 yrs are hypogonadal 1 > Less than 10% of affected population receives treatment • Increasing physician and patient education and awareness should result in increased treatment 1 Mulligan T. et al. Int J. Clin Pract 2006 |
 22 Gels Continue to Drive Significant Growth in TRT Marketplace Source: IMS data $35 $117 $198 $281 $334 $371 $439 $545 $685 $892 $1,149 $49 $59 $77 $118 $210 $302 $390 $451 $483 $554 $663 $810 $1,041 0 200 400 600 800 1,000 1,200 1,400 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 Gel Patch Oral Injectables ($ in millions) $1,329 |
 23 Testosterone Replacement Therapy Landscape is Changing • New competition helping expand treated market in 2011 with 17% y/y growth from 3Q10 • Maintaining solid market share (21%) due to blunting of competitor launch tactics • Testim has expanded covered lives through some wins in managed care |
 24 XIAFLEX Pipeline |
 25 XIAFLEX Represents a Pipeline in a Product - U.S. patent protected through 2028 PRODUCT LATE RESEARCH PRE-CLINICAL PHASE I PHASE II PHASE III MARKET XIAFLEX XIAFLEX XIAFLEX XIAFLEX XIAFLEX XIAFLEX Dupuytren’s Contracture Peyronie’s Disease Frozen Shoulder Syndrome Canine Lipoma* Human Lipoma* Cellulite* *Right to exercise option for exclusive global license from BioSpecifics Technologies Corp. |
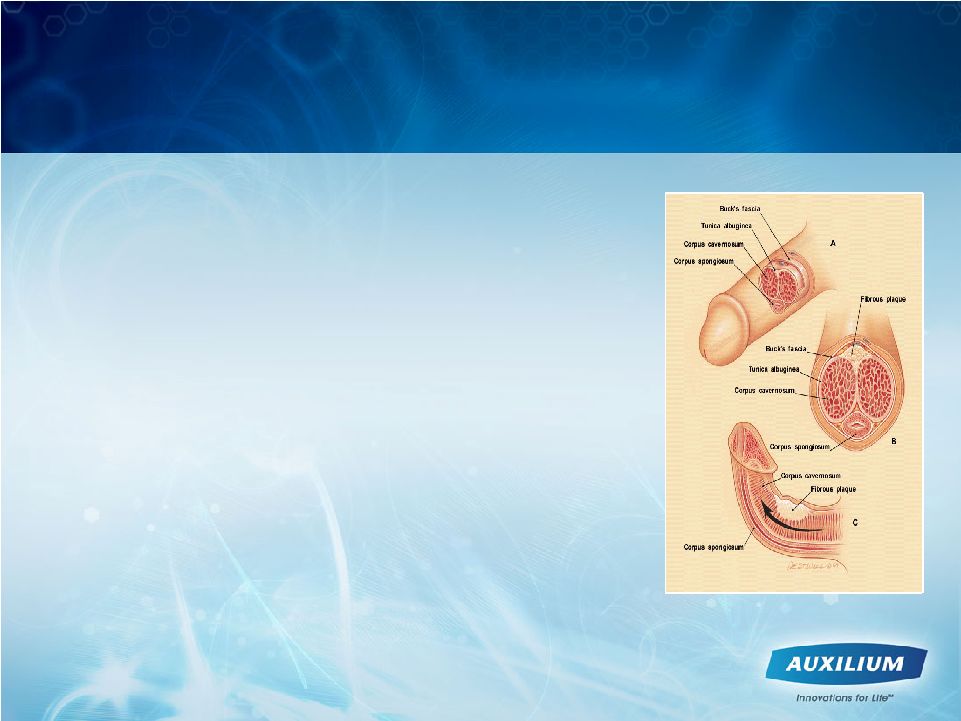 26 1 Smith BH. Am J Clin Pathol. 1966;45:670-678. 2 Somers KD, Dawson DM. J Urol. 1997;157:311-315. • Scarring phenomenon affecting the tunica albuginea 1 • Plaques show excessive collagen deposition 2 • Potential Symptoms > Pain with erection, penile curvature/ deviation, penile shortening, indentations, and/or erectile dysfunction > May experience difficulty with sexual intercourse, loss of self-esteem, and depression Peyronie’s disease Is a Devastating Disorder with No Approved Therapies |
 27 1 Bella A. Peyronie’s Disease J Sex Med 2007;4:1527–1538 2 Lue TF, et al. Summary of the recommendations on sexual dysfunctions in men. J Sex Med 2004;1:6–23. 3 Mulhall JP, et al. Subjective and objective analysis of the prevalence of Peyronie’s disease in a population of men presenting for prostate cancer screening. J Urol 2004;171:2350–3. 4 Smith BH. Am J Clin Pathol. 1966;45:670-678. 5 Lindsay MB, J Urol. 1991;146:1007-1009. 6 Nyberg L, J Urol.128: 48, 1982 • Prevalence of Peyronie’s disease is estimated to be approximately 5% in adult men 1,2,3 > Actual prevalence may be higher, based on autopsies 4 • Prevalence rate increases with age > The average age of disease onset is 53 years 5 • High association with other diseases such as: > Diabetes, erectile dysfunction (ED), Dupuytren’s contracture, plantar fascial contracture, tympanosclerosis, gout, and Paget’s disease 6 We believe Peyronie’s Disease Is Under- diagnosed and Under-treated |
 28 Peyronie’s Disease Phase III Development Program Study Type ~ # Subjects Sites XIAFLEX: Placebo Duration AUX-CC-803 Double-blind, placebo controlled 400 ~ 30 US 5 AUS 2:1 52 Wks AUX-CC-804 Double-blind, placebo controlled 400 ~ 30 US 5 AUS 2:1 52 Wks AUX-CC-802 Open Label 300 12 US 6 NZ ~ 21 EU N/A 36 Wks AUX-CC-805 Pharmacokinetics 20 1 US N/A 4 Wks XIAFLEX 0.58 mg Two injections per treatment cycle 24 to 72 hours between injections Penile plaque modeling following each cycle Up to 4 cycles at 6 week intervals Co-primary endpoints of disease bother and penile curvature |
 29 We Believe Changes to the Phase IIb Trial Design Should Increase the Chance of Success in Phase III • 4 cycles of therapy in the phase III trial • All patients receive modeling • All patients must have at least 12 months of disease since diagnosis Mean net improvements in phase IIb penile curvature and bother domain would have met statistical significance using phase III co-primary endpoint requirements |
 30 Baseline Characteristics in the Phase III Double- blind Trials are Similar to the Phase IIb Clinical Trial • Phase III baseline curvature and Peyronie’s disease bother are comparable to the phase IIb baseline. • Degree of penile curvature deformity did not correlate with PD bother at baseline (e.g. low curvature may result in high PD bother and vice-versa) • The majority of patients with penile curvature =60° or >60° were: – very bothered or extremely bothered the last time they looked at their erect penis (58% and 73%, respectively) – reported difficulty in having vaginal intercourse (70% and 76%, respectively) – reported having sexual intercourse less frequently (81%) • A significant correlation exists between PD bother and patient reported distress. |
 31 Peyronie’s Pivotal Phase III Update • Active dosing phase complete • Double-blind patients are now in the 34 week follow-up phase of studies • Top line results in Peyronie’s should be available in 2Q12 |
 32 Collagen Septae Enable Edematous Fibrosclerotic Panniculopathy (EFP) Cellulite Collagen Network |
 33 EFP Represents an Unmet Medical Need • Current non-surgical treatment options have not shown benefit in EFP – OTC pills, creams and lotions. – Unapproved mixtures of enzymes and fat dissolvers – Various devices, including lasers • There are currently no FDA approved medical therapies for EFP • Surgical approaches have shown some benefit, but are invasive – Liposuction – Subscision • We believe XIAFLEX may enable physicians to enzymatically and specifically lyse the septae, in a less invasive fashion than subscision, possibly resulting in a smoother appearance to the skin surface. |
 34 Early Pilot Phase I Trial was Encouraging • Drs. Dagum and Badalamente at SUNY Stony Brook • 10 female subjects were treated with a single 0.58 mg dose of collagenase • 76% reduction in the physician reported appearance of cellulite and a patient satisfaction score of 1.75 at 6 months – Scale for the patient satisfaction scoring ranged from 1 being completely satisfied and 4 being not satisfied |
 35 Considerable Work Done in Cellulite • Pre-IND meeting with FDA to determine a path forward • Pre-clinical work done with subcutaneous injections of XIAFLEX at variable doses in a mini-pig study – Lysis of collagen in dermal septae was observed at all doses • Completing stability work on new diluent and dosage formulations • Preparation for the filing of an IND in the 4Q11 |
 36 Cellulite – Phase Ib Dose Escalation Study • Expected to begin in 1Q12 • Assess the safety and efficacy of XIAFLEX in the treatment of cellulite – Multiple doses in various volumes and concentrations – 10 injection sites over an 8 x 10cm surface area – Seven cohorts followed for 90 days • Physician and patient reported improvement of cellulite endpoints • Topline results expected in 4Q12 |
 37 Collagen Accumulation Can Result in Adhesive Capsulitis Normal Shoulder Frozen Shoulder |
 38 Adhesive Capsulitis Represents an Unmet Medical Need • Current treatment options have limited results – non-steroidal pain relievers – oral steroids – corticosteroid injections – physical therapy all with limited results. • Manipulation of the shoulder under anesthesia or arthroscopic surgery can disrupt the frozen capsule • There are currently no FDA approved non-surgical therapies for Frozen Shoulder • We believe that XIAFLEX may act by thinning the thickened collagen capsule allowing improved range of motion of the affected shoulder. |
 39 Early Pilot Phase II Trial was Encouraging • Drs. Wang and Badalamente at SUNY Stony Brook – 60 subjects treated with varying doses of collagenase – Following a single injection of collagenase, 8 subjects in the collagenase groups or approximately 16% achieved successful treatment outcomes – In the subsequent open label portion, 46 subjects received additional injections of 0.58 mg of collagenase up to a total of 4 injections – By the third injection, 43 of the 46 subjects in the open label extension or 93%, had achieved a response with increased shoulder motion • Saline injection study – Injected sterile saline under ultrasound guidance – All subjects confirmed appropriate location of injection as extraarticular |
 40 Frozen Shoulder – Phase IIa Dose Escalation Study • To assess the safety and efficacy of XIAFLEX in the treatment of Frozen Shoulder – Two doses (0.29 and 0.58 mg) given under ultrasound guidance – Approximately 50 subjects across 5 cohorts – Up to three injections 21 days apart – Subjects followed for 90 days • Endpoint will be range of motion parameters for the affected shoulder • Topline results expected in the 1H13 |
 41 BTC Will Advance Clinical Studies in Canine and Human Lipomas • Lipomas consist of normal fat encapsulated within a collagen capsule or shell • Current treatment options for lipoma include liposuction or surgical excision • There are no approved non-surgical therapies • We believe XIAFLEX may be beneficial in lysing that collagen capsule allowing redistribution of the normal fat and reduction or disappearance of the palpable lipoma |
 42 XIAFLEX – Additional Data and Development Work Ongoing • Some small pilot studies were conducted examining the effects of collagenase on lipomas in humans and animals. • Investigators noted improvement in lipomas, specifically reduction in their size, following injections of collagenase. • BTC is planning a pilot dose escalation study with treatment of benign lipomas with XIAFLEX in 14 human subjects. • BTC is planning a study in 32 canines in which they will examine several doses for the treatment of benign lipomas with XIAFLEX. |
 43 Business Development Efforts Expected to Focus on Building Out Pipeline • Leverage current infrastructure of Testim and XIAFLEX field forces with additional marketed products • Seeking assets in Urology, Orthopedics and Orphan Diseases |
 44 Net Revenues Up 24% over 3Q10 Quarter End Quarter End Increase 9/30/2011 9/30/2010 (Decrease) Testim U.S. Revenue $52.9 $47.4 $5.5 Testim Ex – U.S. & Contract Revenue 0.7 0.5 0.2 Total Testim Revenue $53.6 $47.9 $5.7 XIAFLEX U.S. Revenue $10.3 $4.6 $5.7 XIAFLEX Ex – U.S. and Contract Revenue $2.8 1.1 1.7 Total XIAFLEX Revenue $13.1 $5.7 $7.4 Total Worldwide Revenue $66.7 $53.6 $13.1 |
 45 3Q11 Financial Results ($ Millions) (Decr.) 3Q11 3Q10 $ % Total Revenues $ 66.7 $ 53.6 $ 13.1 24% Cost of Goods Sold * $ 13.5 $ 11.6 $ 2.0 17% Gross Profit $ 53.2 $ 42.1 $ 11.1 26% % of Revenues 80% 78% R & D Expense * $ 14.2 $ 14.4 $ (0.3) (2%) SG&A Expense * $ 43.3 $ 40.3 $ 2.9 7% Net Loss $ (4.1) $ (12.8) $ 8.7 68% Earnings per share $ (0.08) $ (0.27) $ 0.18 68% * Stock Based Comp Expense $ 3.8 $ 3.5 $ 0.3 10% Cash & Cash Equivalents $ 149.6 Incr. / |
 46 2011 Guidance Global Testim Revenues $200 - 210 million Xiaflex: US Revenues 45 - 50 million Ex-U.S. / Def Rev* 9 - 11 million Total Xiaflex $54 - 61 million Total Revenues $254 - 271 million R & D Expense $60 - 70 million S G & A Expense $170 - 180 million Net Income (Loss) $(40) - (45) million Stock Base Comp Expense $15 - 20 million *Following Pfizer’s first sale in Spain, the third major market, we expect to exceed the upper end of this guidance by $1 to 2 million. |
 47 We Believe That Auxilium is Well-Positioned for Long-Term Growth • Establish XIAFLEX as the standard of care for Dupuytren’s contracture in the U.S. • Maximize Testim in a changing competitive environment • Support Pfizer’s launch of XIAPEX for the treatment of Dupuytren’s contracture • Complete the Peyronie’s Disease double blind studies and announce top- line data in 2Q12 • Initiate phase IIa clinical trial for Frozen Shoulder in 4Q11 and Phase Ib trial for cellulite in early 2012 • Work collaboratively with BTC to assist them as they initiate studies using XIAFLEX |
 48 Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Biopharmaceutical Company Maximize Value of Testim and XIAFLEX Deliver on Current Pipeline Build Out Pipeline in Specialty Therapeutic Areas |
 49 Management Contact James E. Fickenscher/CFO Auxilium Pharmaceuticals, Inc. (484) 321-5900 jfickenscher@auxilium.com William Q. Sargent Jr./ VP IR Auxilium Pharmaceuticals, Inc. (484) 321-5900 wsargent@auxilium.com |