 1 JP Morgan 2012 (NASDAQ: AUXL) Exhibit 99.1 |
 2 Safe Harbor Statement We will make various remarks relating to our current plans, potential future events, and what we believe to be the prospects for the Company in this presentation that constitute forward-looking statements for purposes of the Safe Harbor provisions under the Private Securities Litigation Reform Act of 1995. Today our forward-looking statements will cover, among other things, the Company’s expected financial performance, results and strategic priorities; marketing strategies for XIAFLEX for Dupuytren’s contracture; the potential for XIAFLEX to change the treatment paradigm and become the standard of care for the treatment of Dupuytren’s; the size of the Dupuytren’s market; the effect of new procedure codes on the reimbursement process and XIAFLEX growth; the effect of the identified leading indicators on the success of the XIAFLEX launch and future net revenues; additional Phase IV clinical trials for XIAFLEX; peer to peer dialogue programs for U.S. physicians and our U.S. patient education campaign; Pfizer’s ability to commercialize XIAPEX for Dupuytren’s contracture in EU and Eurasian markets; Asahi Kasei Pharma's ability to develop, register for approval and commercialize XIAFLEX for Dupuytren's contracture and Peyronie's disease in Japan; the potential for XIAFLEX to be used in multiple indications, including for the treatment of Peyronie’s, cellulite, frozen shoulder syndrome and canine and human lipomas, the effectiveness of XIAFLEX for any such indications, and the success of efforts to advance any such new indications; the design and methodology, and the timing to report results, of the phase III trials for XIAFLEX for the treatment of Peyronie’s; the growth of the testosterone replacement therapy market, and the growth opportunity for Testim; business development efforts and opportunities to build out the Company’s pipeline; our ability to maximize the value of XIAFLEX and Testim, deliver on our current pipeline, build out our pipeline and create shareholder value. Actual results may differ materially from those reflected in these forward-looking statements due to various factors, including general financial, economic, regulatory and political conditions affecting the biotechnology and pharmaceutical industries and those discussed in Auxilium's Annual Report on Form 10-K for the year ended December 31, 2010 and Quarterly Report on Form 10-Q for the quarter ended September 30, 2011 under the heading “Risk Factors,” which are on file with the Securities and Exchange Commission (the “SEC”) and may be accessed electronically by means of the SEC’s home page on the Internet at http://www.sec.gov or by means of the Company’s home page on the Internet at http://www.auxilium.com under the heading “For Investors - SEC Filings.” There may be additional risks that the Company does not presently know or that the Company currently believes are immaterial which could also cause actual results to differ from those contained in the forward-looking statements. Given these risks and uncertainties, any or all of these forward-looking statements may prove to be incorrect. Therefore, you should not rely on any such factors or forward-looking statements. In addition, forward-looking statements provide the Company’s expectations, plans or forecasts of future events and views as of the date of this presentation. The Company anticipates that subsequent events and developments will cause the Company’s assessments to change. However, while the Company may elect to update these forward-looking statements at some point in the future, the Company specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing the Company’s assessments as of any date subsequent to the date of this presentation. |
 3 Auxilium – A Company Overview • Fully integrated specialty biopharmaceutical company, ~550 employees including a sales force of ~250 • 2010 global net revenues of $211.4 million; CAGR of 39.4% over past six years • 2011 revenue guidance of $254 – 271 million • Testim® • XIAFLEX® › Testosterone replacement therapy gel for treatment of hypogonadism, or low testosterone › Launched in U.S. in 2003 and EU in 2005 › Paradigm changing treatment for Dupuytren's contracture, launched in US/EU in 2010/11 › Top-line results of pivotal trials in Peyronie’s disease expected in Q2 2012 › Represents a pipeline within a product (Frozen Shoulder syndrome, cellulite, lipomas) |
 4 Auxilium – Vision and Growth Strategy Deliver on Current Pipeline Maximize Value of Testim and XIAFLEX Build Out Pipeline in Specialty Therapeutic Areas Shareholder Value Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Bio-Pharmaceutical Company |
 5 Auxilium – Vision and Growth Strategy Deliver on Current Pipeline Maximize Value of Testim and XIAFLEX Build Out Pipeline in Specialty Therapeutic Areas Shareholder Value Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Bio-Pharmaceutical Company |
 6 Taking Advantage of Global Opportunities Through Licensing Partnerships U.S. Canada EU Japan R.O.W. Exploring Opportunities |
 7 • 1% testosterone gel indicated for testosterone replacement therapy (TRT) in adult males for hypogonadism • Launched in U.S. in 2003 and EU in 2005 • TRT market value has grown at 25.4% CAGR since 2005 • > 8 years of use with established safety and efficacy › ~115M daily doses since launch › 16 clinical studies involving ~1,800 patients • Applied once daily at 5-10mg › 90% stay on starting dose of 5mg (one tube) › Simple application process and dosing • Patent protected through 2025 Testim – Product and Market Overview |
 8 TRT Landscape is Changing • New competition helping increase overall market in 2011 - 17% y/y prescription growth in 3Q11 • Aggressive strategy in select managed care plans has helped Testim minimize market share loss to new players • Despite loss of market share to new entrants, Testim revenues through September 2011 have increased 8% |
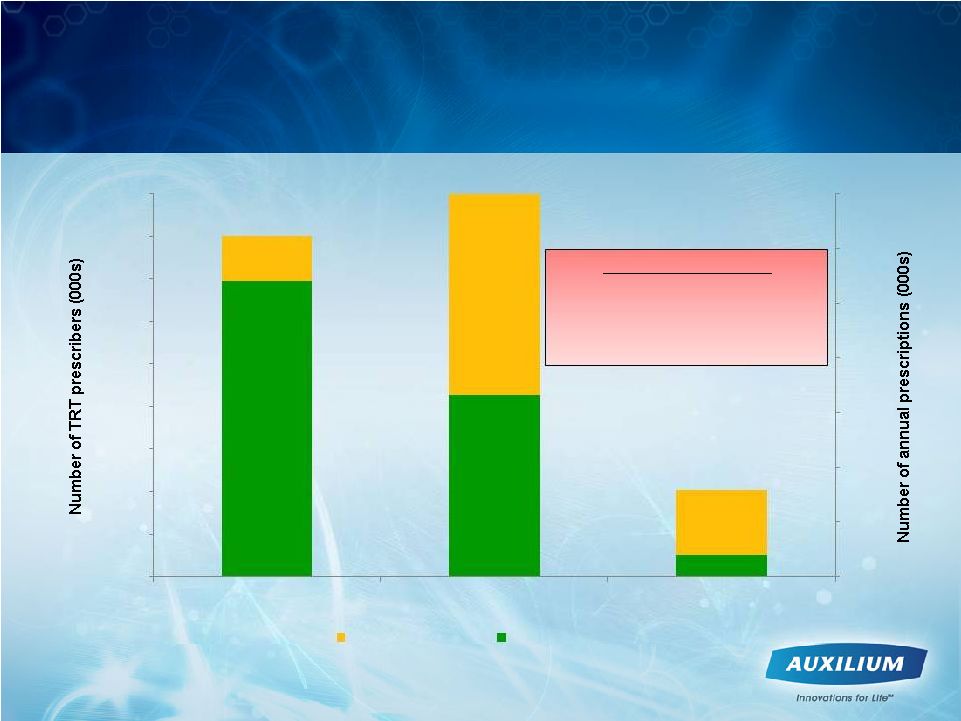 9 Testim Strategy Focuses on the Top TRT Prescribers 13% ~156K ~3,200K ~700K 50% Testim Market Share 72% Source: IMS Prescriber Plan Trak 12 month rolling estimates through November 2011 0 20 40 60 80 100 120 140 160 180 0 500 1000 1500 2000 2500 3000 3500 TRT Prescribers Gel TRx Testim TRx ~ 21% total gel TRT market share ~ 31% market share in targets ~ 12% market share in non-targets target prescribers non-target prescribers |
 10 XIAFLEX - XIAFLEX - A Paradigm-Changing, Minimally A Paradigm-Changing, Minimally Invasive Treatment Invasive Treatment • Approved to treat adults with Dupuytren’s contracture when a cord can be felt • A mixture of collagenases injected directly into Dupuytren's cord • Commercial launch in U.S. in March 2010 • Pfizer launch of XIAPEX® in Europe in April 2011 • Asahi pursuing development in Japan • Long protection period: Drug product composition patent (2028), Biologic Exclusivity (2022) & Orphan Drug protection (2017) |
 11 • Excessive collagen causing reduced elasticity in fascia of hand with nodules or pits as an early, active presentation • Rope-like cords develop in the finger and result in contractures affecting quality of life and daily activities • Fasciectomy is an invasive treatment with high potential for complications › Results can be unpredictable and may require long recovery time of 6-8 weeks, with intensive physical therapy › Recurrence of the disease can require additional surgeries › Surgical complication rate increases dramatically for retreated joints, primarily due to presence of scar tissue › Only ~25% of diagnosed patients estimated to receive surgery annually Dupuytren’s Contracture - A Debilitating Disease Pictures courtesy of Dr. Clayton Peimer 1. Dupuytren’s Disease – Tubiana, LeClerq, Hurst, Badalamente, Mackin 2. SDI Claims Data Based Projections 3. Medicare Data Based Projections (BESS database used, Medicare 5% database also used to validate numbers) 1 • Annually, estimates of 70K surgeries and 300K new diagnoses 1,2,3 |
 12 XIAFLEX - Provides Surgeons With A Minimally Invasive Tool Pre-XIAFLEX injection 30 days following XIAFLEX injection • Simple injection and manipulation procedure • Established mechanism of actions and selective for specific types of collagen • Efficient use of surgeon’s time • No active physical therapy requirement • Limited occurrence of scar tissue at XIAFLEX injection site* • First-line minimally invasive treatment maintains optionality if disease recurs *Blazar, ASSH 2011 poster |
 Adoption of XIAFLEX is Growing ~58% of enrolled sites have ordered XIAFLEX 13 2399 2511 2596 2668 2764 2819 2884 2973 3059 3163 3242 3312 1758 1844 1936 2001 2076 2133 2179 2249 2322 2395 2522 840 902 983 1047 1099 1155 1202 1243 1287 1351 1412 1458 PHYSICIAN ENROLLMENTS SITES ENROLLED UNIQUE SITES ORDERING 2462 |
 14 Steady Growth in Experienced Sites Cumulative Injections per Site |
 15 Strategies for Establishing XIAFLEX as the Standard of Care Motivate Physicians • Compelling clinical efficacy and safety data • Facilitate peer-to-peer dialogue • Activate a critical mass of experienced prescribers • Sample program for XIAFLEX-naïve physicians • Multi-cord phase IV clinical study initiated Activate Patients • Patient copay assistance program d • Direct Response TV campaign in 7 ready markets Streamline Reimbursement • Additional wins with managed care – 96% of covered lives have access to XIAFLEX • 2012 XIAFLEX growth should be helped modestly by new CPT codes |
 16 Auxilium – Vision and Growth Strategy Deliver on Current Pipeline Maximize Value of Testim and XIAFLEX Build Out Pipeline in Specialty Therapeutic Areas Shareholder Value Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Bio-Pharmaceutical Company |
 17 XIAFLEX - Pipeline Opportunities LATE RESEARCH PRE-CLINICAL PHASE I PHASE II PHASE III * Right to exercise option for exclusive global license from BioSpecifics Technologies Corp. PEYRONIE’S DISEASE FROZEN SHOULDER SYNDROME CANINE LIPOMA* HUMAN LIPOMA* CELLULITE* Top-line phase III data in 2Q12 Top-line phase IIa data in 1H13 Clinical trials to start soon Clinical trials to start soon Begin phase Ib in 1Q12, data in 2H12 EXPECTED TIMELINE |
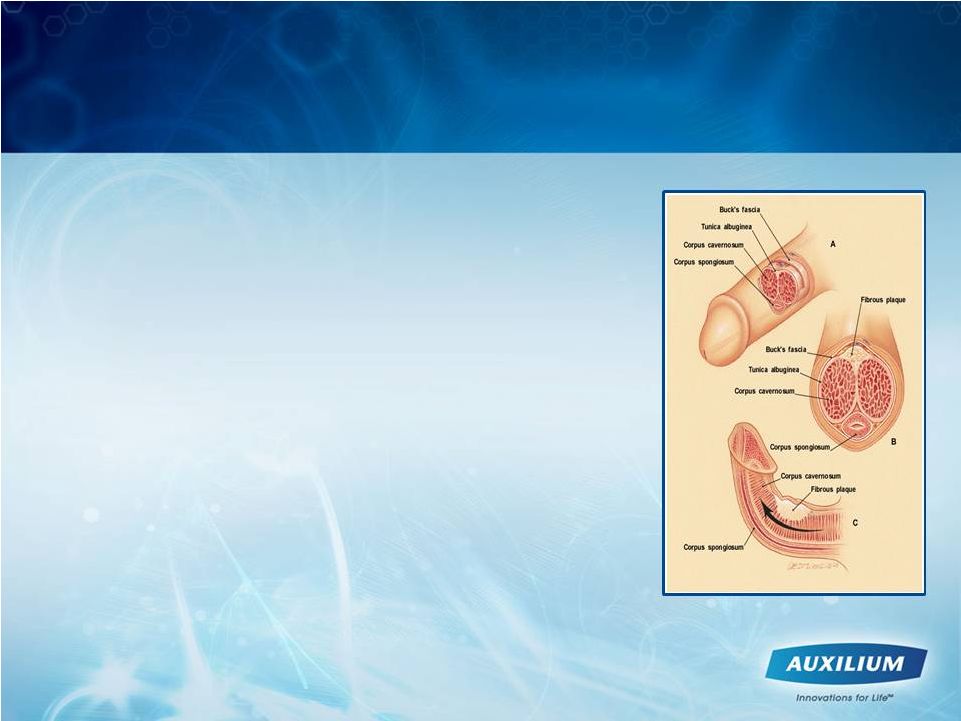 18 • Scarring phenomenon affecting the tunica albuginea 1 • show excessive collagen deposition 2 • Potential symptoms: › Pain with erection, penile curvature/deviation, penile shortening, indentations, and/or erectile dysfunction › May have difficulty with sexual intercourse, loss of self-esteem and depression • Prevalence of Peyronie’s disease is estimated to be approximately 5% in adult men 3,4,5 › The average age of disease onset is 53 years 6 • High association with other diseases such as: › Diabetes, erectile dysfunction, Dupuytren’s contracture, plantar fascial contracture, tympanosclerosis, gout, and Paget’s disease • Surgery is a treatment of last resort and most patients are treated first-line with unapproved medical therapy Peyronie’s Disease: A Devastating Disorder 1 Smith BH. Am J Clin Pathol. 1966;45:670-678. 2 Somers KD, Dawson DM. J Urol. 1997;157:311-315. 3 Bella A. Peyronie’s Disease J Sex Med 2007;4:1527–1538 4 Lue TF, et al. Summary of the recommendations on sexual dysfunctions in men. J Sex Med 2004;1:6–23. 5 Mulhall JP, et al. Subjective and objective analysis of the prevalence of Peyronie’s disease in a population of men presenting for prostate cancer screening. J Urol 2004;171:2350–3. 6 Lindsay MB, J Urol. 1991;146:1007-1009. 7 Nyberg L, J Urol.128: 48, 1982 7 |
 19 Peyronie’s Phase IIb Efficacy Results (2009) • Penile curvature and domains of the patient reported outcomes (PRO) survey were measured › Three dosing cycles – up to six injections › Penile modeling was compared to no modeling • In all subjects, penile curvature and Peyronie’s disease bother, a PRO domain, were statistically significantly improved vs. placebo › Other domains of the PRO trended toward significance › Curvature improvement was greatest during the dosing phase › Improvement in curvature continued through 36 weeks • In the modeling arm, curvature and Peyronie’s disease bother were statistically significantly improved vs. placebo › Mean 35% improvement in curvature vs. placebo › Mean 41% improvement in Peyronie’s disease bother vs. placebo • In the non-modeling arm, curvature and Peyronie’s disease bother were not statistically significantly improved vs. placebo › Greater than expected placebo effect occurred in the no modeling arm › 5 of 16 placebo patients with <12 months since diagnosis had an apparent spontaneous resolution of disease |
 20 Changes to the Phase IIb Trial Design Should Increase the Chance of Success in Phase III • 4 cycles of therapy in the Phase III trial • All patients receive modeling in the Phase III trial • All patients must have greater than 12 months of disease since diagnosis to enter the Phase III trial • Active to placebo ratio of 2:1 Mean net improvements in phase IIb penile curvature and bother domain for modeling arm would have met statistical significance using phase III co-primary endpoint requirements |
 21 Peyronie’s Disease: Pivotal Phase III Update • Active dosing phase complete • Double-blind patients complete 52 week follow-up phase in March 2012 • Top-line results should be available in 2Q12 • sBLA filing expected by end of 2012 • Potential commercial launch planned in late 2013 |
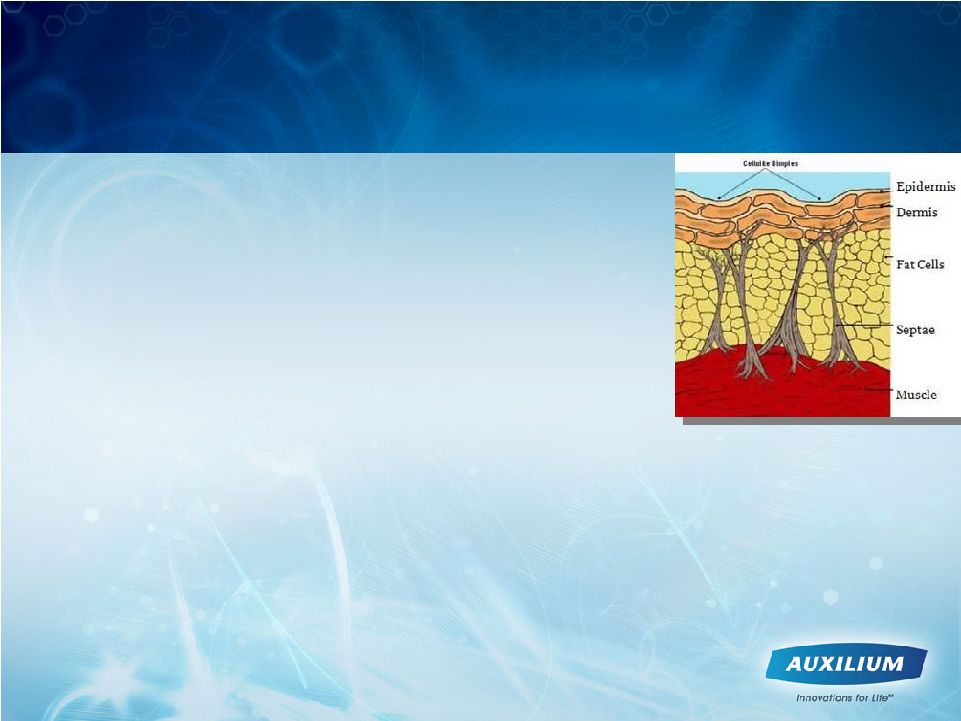 22 Edematous Fibrosclerotic Panniculopathy (EFP) Represents a Compelling Opportunity • There are currently no FDA-approved medical therapies for EFP, commonly known as cellulite • Current non-surgical options haven’t shown benefit › OTC pills, creams and lotions › Unapproved mixtures of enzymes and fat dissolvers › Various devices, including lasers • Surgical approaches have shown some benefit, but are invasive › Liposuction removes fat cells › Subscision surgically disrupts collagen septae and underlying dermal tissues • We believe XIAFLEX may enable physicians to enzymatically and specifically lyse the connective septae, in a more targeted fashion than subscision, possibly resulting in a smoother appearance to the skin surface |
 23 Frozen Shoulder Syndrome: An Unmet Medical Need • Current treatment options have limited results › Non-steroidal pain relievers › Oral steroids › Corticosteroid injections › Physical therapy • Manipulation of the shoulder under anesthesia, long-term intensive physical therapy or arthroscopic surgery can disrupt the frozen capsule • Currently no FDA approved non-surgical therapies for Frozen Shoulder syndrome • We believe that XIAFLEX may act by thinning the thickened collagen capsule allowing improved range of motion of the affected shoulder Normal Shoulder Frozen Shoulder |
 24 Auxilium- Vision and Growth Strategy Deliver on Current Pipeline Maximize Value of Testim and XIAFLEX Build Out Pipeline in Specialty Therapeutic Areas Shareholder Value Our Vision Is to Become a Rapidly Growing, Profitable and Sustainable Bio-Pharmaceutical Company |
 25 Business Development Efforts Focused on Building Out Pipeline • Leverage current infrastructure of Testim and XIAFLEX field forces with in-licensed marketed products • Seeking phase II and later assets for specialty markets (Urology, Orthopedics) and Orphan diseases |
 26 Financials Financials |
 27 3Q11 Net Revenues Up 24% over 3Q10 $53.6 $66.7 +24% |
 28 ** Weighted average common shares were 47,592,975 in Q3 2011 and 47,933,447 in Q3 2011. Total revenues $53.6 $66.7 +24% Cost of goods sold* 11.6 13.5 +17% Gross Profit $42.1 $53.2 +26% % of revenues 78% 80% Operating expenses: Research and development* 14.4 14.2 -2% Sales, general and administrative* 40.3 43.3 +7% Net loss (12.8) (4.1) +68% Net loss per common share** (0.27) (0.08) +68% *Stock based compensation expense 3.5 3.8 Cash and Cash Equivalents $149.6 (amounts in millions, except per share amounts) % change Q3 2011 Q3 2010 Auxilium - Q3 2011 Financial Results |
 29 Auxilium - 2011 Preliminary Net Revenue • Global net revenues should be above midpoint of previously issued guidance of $254 to 271 million and slightly above current 2011 sell side consensus of $263.9 million. • Global Testim net revenues should be above midpoint of previously issued guidance of $200 to 210 million and slightly above current 2011 sell side consensus of $206.5 million. • U.S. XIAFLEX net revenues will be below previously issued guidance of $45 to 50 million and slightly below current 2011 consensus of $44.2 million. |
 30 Auxilium - Strategic Priorities to Drive Long - Term Growth • Maximize Testim revenue growth in a changing competitive environment • Establish XIAFLEX as the standard of care for Dupuytren’s contracture in the U.S. • Support Pfizer’s launch of XIAPEX for the treatment of Dupuytren’s contracture • Complete Peyronie’s Disease double-blind studies and announce top- line data in 2Q12 • Announce top-line phase Ib data for cellulite in 2H12 and phase IIa data for Frozen Shoulder syndrome in 1H13 • Work collaboratively with BTC as they initiate studies using XIAFLEX |
 31 Auxilium - Investment Rationale • Track record of developing and commercializing novel treatments for unmet medical needs • Pipeline with five potential new XIAFLEX indications in development • Global commercialization through licensing partnerships • Opportunity to build urology, orthopedics and other specialty franchises • Strong intellectual property protection for assets • Solid financial position with no debt |