UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
☑ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2018
OR
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 000-55195
GI DYNAMICS, INC.
(Exact name of registrant as specified in its charter)
Delaware |
| 84-1621425 |
(State or other jurisdiction of incorporation or organization) |
| (I.R.S. Employer Identification Number) |
|
| |
PO Box 51915 |
|
|
Boston, Massachusetts |
| 02205 |
(Address of Principal Executive Offices) |
| (Zip Code) |
(781) 357-3300
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Exchange Act: None
Securities registered pursuant to Section 12(g) of the Exchange Act: Common Stock, $0.01 par value per share
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ☐ No ☒
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days: Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files): Yes ☒ No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ |
| Accelerated filer ☐ |
| Non-accelerated filer ☐ |
| Smaller reporting company ☒ |
|
|
|
|
|
| Emerging growth company ☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act): ☐ Yes ☒ No
The aggregate market value of the registrant’s common stock, in the form of CHESS Depositary Interests, or CDIs, held by non-affiliates of the registrant (without admitting that any person whose shares are not included in such calculation is an affiliate), computed by reference to the price at which the CDIs were last sold prior to December 31 2018, the last business day of the registrant’s most recently completed fiscal year, as reported on the Australian Securities Exchange, was $10,829,647 (A$15,343,790).
As of March 8, 2019, there were 19,277,545 shares of common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE:
Portions of the definitive proxy statement for our 2019 Annual Meeting of Stockholders are incorporated by reference into Part III of this report.
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements concerning our business, operations, financial performance and condition as well as our plans, objectives and expectations for our business, operations and financial performance and condition. Any statements contained in this Annual Report on Form 10-K that are not of historical facts may be deemed to be forward-looking statements. The forward-looking statements are contained principally in the section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Forward-looking statements include, but are not limited to, statements about:
| • | our ability to raise sufficient capital to continue operations beyond March 2019; |
| • | our expectations with respect to regulatory submissions and approvals; |
| • | our expectations with respect to our planned clinical trials; |
| • | our expectations with respect to our intellectual property position; |
| • | our ability to develop and commercialize EndoBarrier; |
| • | our ability to attract and retain talented professionals with the relevant experience; |
| • | our ability to develop and commercialize new products; |
| • | our expectation with regard to inventory; and |
| • | our estimates regarding our capital requirements and our need for additional financing. |
In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “could,” “would,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects,” “predicts,” “aims,” “assumes,” “goal,” “intends,” “objective,” “potential,” “positioned,” “target,” “continue,” “seek” and similar expressions intended to identify forward-looking statements.
These forward-looking statements are based on current expectations, estimates, forecasts and projections about our business and the industry in which we operate and our management’s beliefs and assumptions. These forward-looking statements are not guarantees of future performance or development and involve known and unknown risks, uncertainties and other factors that are in some cases beyond our control. As a result, any or all of our forward-looking statements in this Annual Report on Form 10-K may later become inaccurate. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements, and actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements we make. We have included important factors in the cautionary statements included in this Annual Report on Form 10-K, particularly in the “Risk Factors” section, that could cause actual results or events to differ materially from the forward-looking statements that we make.
You are urged to consider these factors carefully in evaluating the forward-looking statements and are cautioned not to place undue reliance on the forward-looking statements. You should read this Annual Report on Form 10-K and the documents that we have filed as exhibits to our Form 10-K completely and with the understanding that our actual future results may be materially different from what we expect. These forward-looking statements speak only as at the date of this Annual Report on Form 10-K. Unless required by law, we do not intend to publicly update or revise any forward-looking statements to reflect new information or future events or otherwise. You should, however, review the factors and risks we describe in the reports we will file from time to time with the SEC after the date of this Annual Report on Form 10-K.
ANNUAL REPORT ON FORM 10-K
FOR THE YEAR ENDED DECEMBER 31, 2018
|
|
|
| Page |
| |
|
| |||||
PART I |
|
|
|
| ||
|
|
| ||||
Item 1. |
|
|
| 5 |
| |
Item 1A. |
|
|
| 16 |
| |
Item 1B. |
|
|
| 31 |
| |
Item 2. |
|
|
| 31 |
| |
Item 3. |
|
|
| 31 |
| |
Item 4. |
|
|
| 31 |
| |
|
| |||||
|
|
|
| |||
|
|
| ||||
Item 5. |
|
|
| 32 |
| |
Item 6. |
|
|
| 32 |
| |
Item 7. |
| Management’s Discussion and Analysis of Financial Condition and Results of Operations |
|
| 33 |
|
Item 7A. |
|
|
| 45 |
| |
Item 8. |
|
|
| 45 |
| |
Item 9. |
| Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
|
| 45 |
|
Item 9A. |
|
|
| 46 |
| |
Item 9B. |
|
|
| 47 |
| |
|
| |||||
|
|
|
| |||
|
|
| ||||
Item 10. |
|
|
| 48 |
| |
Item 11. |
|
|
| 65 |
| |
Item 12. |
| Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
|
| 66 |
|
Item 13 |
| Certain Relationships and Related Transactions, and Director Independence |
|
| 66 |
|
Item 14 |
|
|
| 66 |
| |
|
| |||||
|
|
|
| |||
|
|
| ||||
Item 15. |
|
|
| 67 |
| |
|
|
|
| 70 |
| |
|
|
|
| F-1 |
| |
4
Overview
We are a clinical stage medical device company focused on the development and commercialization of EndoBarrier, a medical device system intended for treatment of patients with type 2 diabetes and obesity. EndoBarrier is a medical implant designated for the treatment of co-occurring type 2 diabetes and obesity, is approaching 4,000 implants since inception, and is the subject of an approved FDA pivotal trial in the United States which we plan to initiate in 2019. We believe that EndoBarrier represents a paradigm-breaking approach to traditional management of type 2 diabetes, which has focused historically on lifestyle intervention and diabetes medications. According to the article “Adherence to Therapies in Patients with Type 2 Diabetes,” by Luis-Emilio Garcia-Perez, these historical treatments for type 2 diabetes and obesity are limited in efficacy, with less than 50% of patients achieving glycemic control.
EndoBarrier represents the first material departure from this historical approach. We believe that EndoBarrier offers an adjunct to non-insulin diabetes pharmacotherapy, while providing patients a chance to significantly reduce or eliminate insulin and increase the likelihood of avoiding invasive and permanent bariatric or metabolic surgery. EndoBarrier is designed to mimic the mechanism of action of the duodenal-jejunal exclusion currently created by gastric bypass surgery (which is referred to as Roux-en-Y Gastric Bypass, or RYGB), as explained in the article “EndoBarrier: A Safe and Effective Novel Treatment for Obesity and Type 2 Diabetes?” by Nisha Patel. After implanting EndoBarrier, the patient’s food that exits the stomach will flow through the inside of the EndoBarrier Liner, and therefore not contact the wall of the upper intestine (duodenum and jejunum). The food will also not mix with fluids from the pancreas and bile duct until these fluids and the food reach the far end of the EndoBarrier Liner. It is believed that, in a similar fashion to RYGB, shunting the food to a more distant location in the upper intestines triggers hormonal responses that regulate key factors that help control hunger, satiety, and insulin sensitivity, which is also explained in the article by Nisha Patel.
EndoBarrier has been shown in multiple independent clinical studies to lower blood sugar levels (hemoglobin A1c or HbA1c), reduce excess body weight, increase insulin sensitivity, and positively affect other health metrics and comorbidities. Because it has been utilized in approximately 4,000 procedures, EndoBarrier has a thoroughly characterized benefit-risk profile. A recent independent meta-analysis published in American Diabetes Association (ADA) Diabetes Care, “Effects of the Duodenal-Jejunal Bypass Liner on Glycemic Control in Patients with Type 2 Diabetes with Obesity: A Meta-Analysis with Secondary Analysis on Weight Loss and Hormonal Changes,” by Pichamol Jirapinyo, MD provides the most comprehensive review and meta-analysis of EndoBarrier clinical data to date. In addition, two ongoing EndoBarrier registries in the UK and Germany have captured more than 800 patients between the two databases. These registries as well as other investigator-initiated trials continue to release clinical data on a regular basis, largely centered around the annual meetings hosted by Digestive Disease Week (DDW) in May, ADA in June, and EASD (European Association for the Study of Diabetes) in October.
Business Strategy
Our goal is to become the leading provider of alternative treatment options for type 2 diabetes and obesity. Our corporate priorities include:
1) | US Clinical Operations: Conduct Stage 1 of the EndoBarrier 18-1 clinical trial in the United States. |
The Company is focused on the successful execution of the 18-1 clinical trial in the United States. In August 2018, we received notification from the FDA that it had approved the Investigational Device Exemption (IDE) application for EndoBarrier, pending Institutional Review Board (IRB) approval, which was received in February 2019. The approved EndoBarrier IDE represents the pivotal clinical trial for EndoBarrier.
The 18-1 clinical trial is a randomized, controlled, double blinded trial with 3:1 randomization between patients who receive the EndoBarrier implant (3) and the control group made up of patients who will receive a sham procedure and not receive the EndoBarrier implant (1). Both arms will maintain pharmacotherapy in conjunction with ADA guidelines and lifestyle and nutritional counseling consistent with ADA guidelines. The primary endpoint of the trial is reduction of HbA1c from baseline to removal of the EndoBarrier at twelve months as compared to the control group. The study includes numerous secondary endpoints, including weight, insulin resistance, cardiovascular metrics, as well as NAFLD (non-alcoholic fatty liver disease) and NASH (non-alcoholic steatohepatitis).
5
The 18-1 clinical trial consists of two stages. The first stage consists of 50 EndoBarrier patients and approximately 17 control patients. At the end of stage 1, the Company will submit four DMC (Data Monitoring Committee) reports to the FDA for review. Upon a successful completion of the stage 1 review, we will submit a request to the FDA to complete the study by completing stage 2, which we anticipate will be comprised of 130 EndoBarrier patients and approximately 43 control patients for a study total of 240 patients (180 EndoBarrier and 60 control patients). There is no guarantee that this will be the final composition of the study, as the number of remaining stages or remaining patients may vary based on the stage 1 clinical data and the outcome of the stage 1 review with FDA. In addition to the multiple staged study, the FDA has mandated certain stopping rules which, if triggered, could result in the 18-1 study having enrollment or treatment delayed or stopped.
Assuming the successful completion of sufficient financing to ensure funds will be available at the start of the trial to ensure completion of the trial in a safe and patient-focused manner, we expect to begin enrolling patients in the 18-1 clinical trial and to complete the enrollment within 6 months of initiation of enrollment.
2) | India Partnership: Conduct the EndoBarrier clinical trial in India as part of the Apollo Sugar partnership. |
We are focused on the successful execution of the EndoBarrier trial in India as part of our partnership with Apollo Sugar, which was announced on November 27, 2018.
The EndoBarrier India study is also a randomized, controlled, double blinded trial with 3:1 randomization between patients who receive the EndoBarrier implant (3) and the control group made up of patients who will not receive the EndoBarrier implant (1). The primary endpoint of the trial is reduction of HbA1c from baseline to removal of the EndoBarrier at twelve months as compared to the control group. The study includes numerous secondary endpoints, including weight, insulin resistance, cardiovascular metrics, as well as NAFLD and NASH. The study includes 100 patients total, which consists of 75 patients who will receive the EndoBarrier implant and 25 control patients. Five clinical sites in India will participate in the study.
Apollo Sugar is a collaboration between Apollo Health & Lifestyle Limited and Sanofi. Apollo Sugar is a division of Apollo Hospitals Group (Apollo) focused on the treatment of metabolic disorders and operates an integrated network of centers of excellence for diabetes, obesity and endocrinology. Apollo is the largest private hospital system in India and has emerged as Asia’s foremost integrated healthcare services provider, maintaining a robust presence of hospitals, pharmacies, primary care and diagnostic clinics across the healthcare ecosystem. Since its inception, Apollo has treated over 65 million patients from 141 countries.
The Company and Apollo Sugar intend to conduct the clinical trial and seek regulatory approval in India and, in parallel, finalize the terms of a partnership that will focus on the marketing, distribution and clinical support of EndoBarrier to appropriate patients in India and Southeast Asia. Final terms of the proposed collaboration are subject to negotiation and will be disclosed upon completion.
3) | Gain CE Mark: Work with Intertek to gain EndoBarrier CE mark. |
CE Marking on a product is a manufacturer’s declaration that a product meets the applicable health, safety, and environmental requirements outlined in the appropriate European product legislation and has undergone the relevant conformity assessment procedure. A CE Mark is required before we can market EndoBarrier in the EU and certain Middle Eastern countries.
We have entered into an agreement with Intertek to serve as our notified body and to assist us with obtaining CE Marking for EndoBarrier as described in our announcement released on October 5, 2018. A notified body evaluates the conformity of products and the associated quality systems for manufacturers that seek to sell products in Europe.
4) | Ongoing clinical data: Support continued release of clinical data from registries and investigator-initiated clinical trials. |
We continue to support the ongoing efforts of multiple clinicians as they develop new EndoBarrier clinical data. Multiple investigator-initiated clinical trials released clinical data in 2018. It is expected that these clinical trials will continue to release data into 2019, 2020, and beyond.
6
We also support two primary registries that continue to release clinical data. The Association of British Clinical Diabetologists (ABCD) Worldwide EndoBarrier Registry contains over 500 patient data points with the goal of registering and gathering data on 1,000 patients. The German EndoBarrier Registry contains over 300 patient data points. The patients within these two registries are non-overlapping. Both registries are expected to continue releasing clinical data highlighting the EndoBarrier treatment effect on an annual basis.
Multiple studies continue to release data supporting the long-term treatment durability that now stands at two years after EndoBarrier implant and one year after EndoBarrier removal.
The clinical data releases have historically focused, and are expected to continue focusing, on the annual data releases associated with Digestive Disease Week (DDW) in May, The American Diabetes Association (ADA) in June, The European Association for the Study of Diabetes (EASD) in October, and Obesity Week in November.
5) | Development of Intellectual Property: Continue developing our intellectual property and protecting our patents. |
We will continue to invest in expanding our intellectual property portfolio. Our current patent portfolio is composed of 93 issued and pending U.S. and non-U.S. patents. We have been issued 60 U.S. patents and maintain 9 pending U.S. patent applications. We have also sought intellectual property protection outside the U.S. and have been issued 20 patents across Canada, China, the European Patent Convention region (including Germany and the United Kingdom), Hong Kong, Japan, Israel and India, and we have 7 pending PCT applications and 17 pending foreign patent applications.
As a result of a settled 2013 legal action, we granted W.L. Gore & Associates, Inc., or Gore, a nonexclusive, royalty-free license to use our patents within the vascular system. Gore is not licensed to use our patents for any applications in the gastrointestinal tract.
Our current issued patents expire between 2023 and 2031. We also actively monitor our intellectual property by regularly reviewing new developments to identify extensions to our patent portfolio. We employ external patent attorneys to assist us in managing our intellectual property portfolio.
Summary
In summary, we believe that EndoBarrier represents the most effective new treatment option in a market dominated by pharmaceutical companies generating annual revenue in excess $40 billion. We believe that EndoBarrier is poised to have a transformative and disruptive effect on the type 2 diabetes and obesity market. EndoBarrier is one of the few treatment options that treats type 2 diabetes concurrently with obesity and continues to show lasting treatment effects post treatment in many patients. As the duration of the EndoBarrier treatment effect continues to increase in published literature and because, to date, there has been minimal observed clinical risk after EndoBarrier removal, the EndoBarrier risk-benefit balance continues to evolve in an increasingly positive manner.
Background of the Disease
Diabetes mellitus type 2 (also known as type 2 diabetes) is a long-term progressive metabolic disorder characterized by high blood sugar, insulin resistance, and reduced insulin production. People with type 2 diabetes represent 90-95% of the worldwide diabetes population; only 5-10% of this population is diagnosed with type 1 diabetes (a form of diabetes mellitus wherein little to no insulin is produced).
Being overweight is a condition where the patient’s body mass index (BMI) is greater than 25 (kg/m2); obesity is a condition where the patient’s BMI is greater than 30. Obesity and its comorbidities contribute to the progression of type 2 diabetes. Many experts believe obesity contributes to higher levels of insulin resistance, which creates a feedback loop that increases the severity of type 2 diabetes.
When considering treatment for type 2 diabetes, it is optimal to address obesity concurrently with diabetes. According to the article “Mechanisms of Insulin Resistance in Obesity,” by Jianping Ye, absent the concurrent treatment of obesity with the treatment of type 2 diabetes, obesity will likely continue to contribute to the progressive nature of type 2 diabetes. It is our belief that the inability of the current pharmacologic options to fully treat type 2 diabetes is due to the fact that diabetes medications generally treat blood sugar levels only and do not contribute substantially to weight loss.
7
According to the American Diabetes Association 2018 Standards of Medical Care in Diabetes, the current treatment paradigm for type 2 diabetes is lifestyle therapy combined with pharmacological treatment, whereby treating clinicians prescribe a treatment regimen of one to four concurrent medications that could include insulin. Insulin usage carries a significant risk of increased mortality and may contribute to weight gain, which in turn may lead to higher levels of insulin resistance and increased levels of blood sugar, as explained in the article “Insulin-associated weight gain in diabetes--causes, effects and coping strategies,” by Russel-Jones D . Fewer than 50% of patients treated pharmacologically for type 2 diabetes are adequately managed, meaning that medication does not lower blood sugar adequately and does not halt the progressive nature of diabetes for these patients, as explained in the article “Adherence to Therapies in Patients with Type 2 Diabetes,” by Luis-Emilio Garcia-Perez.
The current pharmacological treatment algorithms for type 2 diabetes fall short of ideal, creating a large and unfilled treatment gap. We believe that EndoBarrier, which is designed to mimic the mechanism of action of duodenal-jejunal exclusion currently created by RYGB gastric bypass surgery and operates in a relatively similar manner by reducing both weight and long-term blood sugar levels, can fill this gap by treating type 2 diabetes and obesity in a unique minimally invasive and reversible manner.
Market Opportunity
Unmet Clinical Needs in the Treatment of Type 2 Diabetes and Obesity.
In 2017, the International Diabetes Federation estimated there were 425 million adults with diabetes worldwide, with ~90% diagnosed with type 2 diabetes. Diabetes is the leading cause of cardiovascular disease, kidney failure, blindness, and lower-limb amputation in almost all countries.
Three years after initial diagnosis, over half of patients with type 2 diabetes require multiple drug therapies. Studies have shown that less than 50% of the type 2 diabetes population is adequately managed pharmacologically, as explained in the article “Adherence to Therapies in Patients with Type 2 Diabetes,” by Luis-Emilio Garcia-Perez. . At ten years post diagnosis, most patients, despite insulin use in many, struggle to reach their hemoglobin A1c (HbA1c) treatment goals. HbA1c is a glycosylated hemoglobin molecule found in the bloodstream that is formed when red blood cells are exposed to blood glucose. HbA1c has become the generally accepted gold standard biomarker for measuring levels of diabetes control in clinical practice and in human trials.
Many patients and health care systems struggle to meet the financial burden imposed by the numerous concurrent medications required to attempt to control the progressive nature of type 2 diabetes.
According to the World Health Organization, in 2016 more than 1.9 billion adults 18 and older were overweight. Of these, 650 million people worldwide are diagnosed with obesity (BMI ³ > 30 kg/m 2), a condition often leading to serious health consequences such as cardiovascular disease, diabetes, musculoskeletal disorders, and some cancers.
Those suffering from both type 2 diabetes and obesity, also referred to as diabesity, total more than 169 million worldwide, represent as enormous public health problem not only in the United States (US) but also globally, as explained in the article “Epidemiology of Obesity and Diabetes and Their Cardiovascular Complications,” by Bhupathiraju, S. We believe that the unchecked worldwide rate of growth of the type 2 diabetes and obesity patient population represents one of the greatest unmanaged health risks in all of health care.
We believe EndoBarrier can treat patients with type 2 diabetes and obesity in a safe, effective, nonpharmacological, and nonpermanent manner.
The Treatment Gap
Our intent in developing and seeking regulatory approval for EndoBarrier is to help clinicians deliver a unique treatment option to those patients suffering from type 2 diabetes and obesity, a disease state that sorely lacks innovative new treatment options.
8
We and our scientific advisors feel there must be a change in how the medical establishment currently treats patients suffering from type 2 diabetes and obesity because current treatment options are not effective. According to the International Diabetes Federation, the number of patients progressing to later stages of type 2 diabetes and obesity continues to grow at an alarming rate. Yet less than half of all type 2 diabetes patients are adequately managed by pharmacotherapy, and insulin carries serious risks and may in many cases contribute to the further progression of obesity. At the extreme end of the treatment spectrum, the treatment options are limited to different types of bariatric or metabolic surgery, which are highly invasive and irreversible procedures. Less than 1% of patients who are eligible for bariatric or metabolic surgery opt to undergo the procedure, as explained in the Gut article “Recent advances in clinical practice challenges and opportunities in the management of obesity,” by Acosta A.
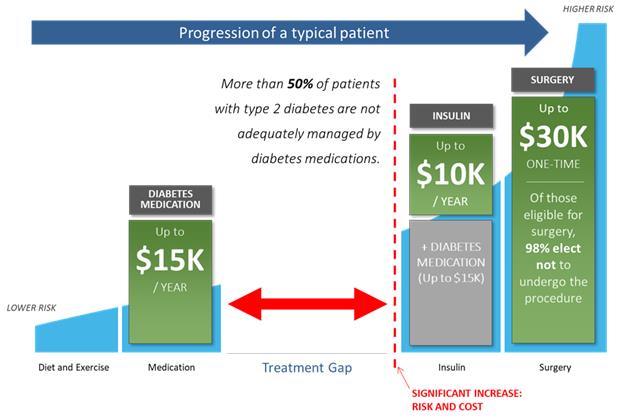
The graphic above illustrates the multiple treatment options during the course of progression of type 2 diabetes:
| • | Risk associated with treatment increases from left to right. |
| • | Progression of diabetes and obesity increases vertically. |
| • | Lifestyle therapy (lifestyle counseling including nutrition and exercise) is the first line of defense against the progression of type 2 diabetes and obesity. |
| • | Pharmacotherapy |
| o | Oral monotherapy follows lifestyle therapy, often with metformin as the first line of treatment. |
| o | Multiple combinations may then be administered, as recommended by the American Diabetes Association and other diabetes associations around the world. |
| o | Ultimately, as disease progression continues, injected insulin may be prescribed. |
| • | Bariatric or metabolic gastric bypass surgery may represent a final option. |
9
A significant and rapidly growing patient population falls into the treatment gap, wherein the patient is inadequately managed by medication yet unwilling to undergo gastric bypass surgery. For patients who elect to undergo bariatric or metabolic surgery, the clinical gains may not be permanent, while the risk associated with the procedure is permanent, according to the article “Gastric bypass surgery: Who is it for?” from the Mayo Clinic. These risks include, but are not limited to, excessive bleeding, blood clots, infection and in some cases death. We believe that because EndoBarrier is minimally invasive, can mimic the effects of gastric bypass, and has minimal side-effects, EndoBarrier can uniquely fill this treatment gap, as explained in the article “EndoBarrier: A Safe and Effective Novel Treatment for Obesity and Type 2 Diabetes?” by Nisha Patel.
The EndoBarrier Solution
EndoBarrier is intended for the treatment of type 2 diabetes and obesity in a minimally invasive and reversible manner. EndoBarrier is designed to mimic the mechanism of action of duodenal-jejunal exclusion created by gastric bypass surgery.
The EndoBarrier System consists of three primary components:

| • | EndoBarrier — The EndoBarrier or EndoBarrier Liner is a 60-cm-long implant consisting of a thin, flexible, impermeable fluoropolymer sleeve coupled to a proprietary nitinol anchor assembly. A gastroenterologist (GI clinician) implants the EndoBarrier Liner into the patient’s duodenum in a minimally invasive manner using the EndoBarrier Delivery System. The EndoBarrier Liner is placed via endoscopy (through the mouth and esophagus and into the stomach without cutting tissue) during a procedure that typically takes less than twenty minutes. Once the EndoBarrier Liner is properly positioned in the patient’s upper intestine just below the stomach, the EndoBarrier Delivery System is removed and EndoBarrier Liner remains, held in place by a proprietary anchoring mechanism. EndoBarrier remains in the body for a maximum intended duration of twelve months until removal, again via a minimally invasive endoscopic procedure using the EndoBarrier Retrieval System. The effect of EndoBarrier Liner begins promptly after device placement. |
| • | EndoBarrier Delivery System — EndoBarrier is delivered using our proprietary single-use delivery system. This includes a sterile custom-made delivery catheter 300 cm in length that is sufficiently flexible to be passed through the patient’s mouth, through the stomach, and into the intestine. The EndoBarrier Liner is provided pre-packed in the EndoBarrier Delivery System within the capsule at the distal end of the delivery catheter. The EndoBarrier Liner is deployed by the clinician using the delivery system controls at the proximal or near end of the system. The delivery procedure is brief, typically taking less than twenty minutes, during which the patient is either anaesthetized or semi-sedated. |
10
EndoBarrier has been shown in multiple company-sponsored and independent clinical studies to lower blood sugar levels (hemoglobin A1c or HbA1c), reduce excess body weight, and positively affect other health metrics and comorbidities.
EndoBarrier is not currently approved or commercially available in any jurisdiction. In August 2018, we received notification from the FDA that it had approved the IDE application, pending IRB approval which was received in February 2019. To gain regulatory approval to commercialize EndoBarrier in the United States, we must submit a pre-market authorization (PMA) application for review and approval by the FDA.
The EndoBarrier Effect
Obesity has been shown to exacerbate insulin resistance and contribute to the progression of type 2 diabetes. In situations where lifestyle modification and pharmacotherapy have failed and surgery is not an option or is considered a therapy of last resort, EndoBarrier is intended to reduce blood sugar and weight. In clinical trials in both the United States and outside of the United States, EndoBarrier has been shown to:
| • | significantly lower glucose levels; |
| • | significantly lower body weight; |
| • | lower cardiovascular-related risk factors. |
EndoBarrier utilization accomplishes this in many patients by affecting key hormones involved in insulin sensitivity, glucose metabolism, satiety, and food intake. (See the section titled “How EndoBarrier Works: EndoBarrier Mechanism of Action.”)
Our intent in positioning EndoBarrier to fill the type 2 diabetes and obesity treatment gap is to help patients and clinicians avoid the initiation of insulin therapy by helping many patients maintain lower HbA1c levels and slow or halt the progression of type 2 diabetes. Furthermore, for those patients whose type 2 diabetes has progressed to the point where insulin therapy is necessary, EndoBarrier has been shown in many cases to lower HbA1c levels to the point where insulin is no longer needed. Finally, if the progression of type 2 diabetes is severe enough to warrant gastric bypass surgery, we expect that EndoBarrier either may serve as an opportunity to control type 2 diabetes so that surgery may not be needed, or at least better prepare the patient for bariatric surgery by lowering weight and helping control other comorbidities prior to surgery.
How EndoBarrier Works: EndoBarrier Mechanism of Action
The EndoBarrier mechanism of action is thought to be based on its functional similarities in many ways to Roux-en-Y gastric bypass surgery (RYGB). Once the EndoBarrier is implanted into the duodenum and proximal jejunum, ingested food passing through the EndoBarrier during the normal digestive process is prevented from interacting with the epithelium, microbiota, mucosal layer, or biliopancreatic secretions within the duodenum and proximal jejunum. In addition, EndoBarrier acts as a physical barrier that prevents the interaction of food with pancreatic enzymes and bile until reaching the end of the EndoBarrier Liner. Pancreatic enzymes and bile pass outside EndoBarrier and mix with the food at the distal end of the liner, where absorption ultimately takes place in the intestine. Thus, EndoBarrier creates a functional but reversible bypass of the upper intestine. Unlike RYGB surgery, EndoBarrier does not require an invasive and permanent surgical procedure or permanent physical modification of the stomach and exclusion of the distal stomach from the alimentary flow.
11
Our scientific team and advisors have postulated the following EndoBarrier mechanisms of action, based on scientific evidence from such articles as the “Effects of the Duodenal-Jejunal Bypass Liner on Glycemic Control in Patients with Type 2 Diabetes with Obesity: A Meta-Analysis with Secondary Analysis on Weight Loss and Hormonal Changes,” by Pichamol Jirapinyo and “The EndoBarrier: Duodenal-Jejunal Bypass Liner for Diabetes and Weight Loss,” by Aruchuna Ruban.:
| • | Exclusion of the duodenum — This may offset an abnormality of gastrointestinal physiology responsible for insulin resistance and type 2 diabetes. |
| • | Increased nutrient delivery to the distal small bowel — Additional findings suggest that the exclusion of the proximal intestine (foregut theory) and increase in nutrient delivery to the distal small bowel (hindgut theory) created by EndoBarrier likely induce neuro-hormonal changes and nutrient sensing that affect energy balance and glucose homeostasis. |
| • | Secretion of GLP-1 — Partially digested nutrients reach the distal ileum, which stimulates the secretion of GLP-1 by L-cells located in this area. GLP-1 is known to regulate insulin secretion and action. |
| • | Increase in gut hormones — This contributes to the restoration of energy and glucose homeostasis. |
| • | Elevated GLP-1 and PYY levels — Both levels are elevated as quickly as one-week post-implantation. Both hormones may play a role in satiety and body weight control. |
| • | Increased levels of bile acids — This stimulates thermogenesis and gut hormone secretions. |
| • | Decreased caloric intake— Studies have demonstrated that patients with EndoBarrier, aided by increased satiety from GLP-1, eat less and feel full longer, leading to a decrease in caloric intake. |
There is no known evidence of occurrence of clinically significant caloric malabsorption with EndoBarrier. EndoBarrier covers only 60 cm of duodenal and proximal jejunal mucosa, which most likely represents less than 10% of the length of the small intestine and leaves almost the entire jejunum and ileum for digestion and absorption.
We began selling EndoBarrier in Europe and South America in 2010 and in Australia in 2011. Since inception, the Company has distributed almost 4,000 units of EndoBarrier and generated a total of $7.8 million in revenue. We have incurred net losses in each year since our inception.
We signed an agreement with our manufacturing partner, Proven Process Medical Devices (“PPMD”, “the Manufacturer”) on July 26, 2017.
We have five subsidiaries: GI Dynamics Securities Corporation, a Massachusetts-incorporated nontrading entity; GID Europe Holding B.V., a Netherlands-incorporated nontrading holding company; GID Europe B.V., a Netherlands-incorporated company that conducts certain of our European business operations; GID Germany GmbH, a German-incorporated company that conducts certain of our European business operations; and GI Dynamics Australia Pty Ltd, an Australia-incorporated company that conducts our Australian business operations.
We have raised net proceeds of approximately $264.0 million through sales of our equity of which $6.6 million was raised through our 2018 private placement offerings. We generated $75.7 million in proceeds, net of expenses, through the sale of convertible preferred stock to a number of US venture capital firms, two global medical device manufacturers, and individuals prior to going public. In June 2011, we issued convertible term promissory notes to several of our shareholders totaling $6.0 million, which were repaid concurrently with the closing of our IPO in September 2011 with the associated gross proceeds. In September 2011, we raised approximately $72.5 million, net of expenses, and repaid $6.0 million of convertible term promissory notes in our IPO in Australia and simultaneous private placement of Chess Depository Interests (CDIs) to accredited investors in the United States. In connection with the IPO, all our existing shares of preferred stock were converted into common stock.
In July and August 2013, we raised approximately $52.5 million, net of expenses, in an offering of our CDIs to sophisticated, professional, and accredited investors in Australia, the United States, and certain other jurisdictions. In May 2014, we raised approximately $30.8 million, net of expenses, in an offering of our CDIs to sophisticated, professional, and accredited investors in Australia, Hong Kong, the United Kingdom, and certain other jurisdictions.
12
In December 2016, we raised approximately $1.0 million, net of expenses, in an offering of our CDIs to sophisticated and professional investors in Australia and certain other jurisdictions.
In January 2017, we raised approximately $0.2 million, net of expenses, in an offering of our CDIs to eligible shareholders under a Security Purchase Plan (SPP) available to security holders with registered addresses in Australia or New Zealand.
In June 2017, we completed a Convertible Term Promissory Note (the “2017 Note”) secured financing for a gross amount of $5.0 million, which 2017 Note accrues interest at 5% per annum compounded annually. In December 2018, the maturity date of the 2017 Note was extended from December 31, 2018 to March 31, 2019 in exchange for payment of approximately $394 thousand, which was the total accrued interest on the 2017 Note at December 31, 2018.
In July 2017, we signed an agreement with our manufacturing partner Proven Process Medical Devices (“PPMD”, “the Manufacturer”).
In February and March 2018, we raised approximately $1.6 million in an offering of our CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom.
In May 2018, we completed a Convertible Term Promissory Note and Warrant (the “2018 Note and Warrant”) for a gross amount of $1.75 million. The $1.75 million 10% per annum convertible note can convert to CDI’s at A$ 0.018 per CDI and matures on May 30, 2023. The warrants consist of the right to purchase 97,222,200 CDIs for A$ 0.018 per CDI with anti-dilution protections.
In September and November 2018, we raised approximately $5.0 million in an offering of our CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom.
In October 2018, we announced that we signed an agreement with our new notified body Intertek to pursue CE marking of EndoBarrier in Europe.
In November 2018, we signed a clinical trial agreement and a memorandum of understanding with our clinical and potential commercial partner Apollo Sugar, a division of Apollo Hospitals Group located in India.
In December 2018, we announced the appointment of Charles Carter as the Company’s Chief Financial Officer and Secretary.
The rights of our shareholders are governed by Delaware general corporation law.
We are headquartered in Boston, Massachusetts, where the majority of the Company’s employees work. We have subsidiaries in the Netherlands, Germany, and Australia, with employees in Germany and Australia.
2018 in Review
For us, 2018 was a year primarily focused on working with the FDA to gain approval for a new EndoBarrier pivotal clinical trial in the United States and planning and preparing to initiate the clinical trial (18-1). We also focused on a critical partnership in India with Apollo Sugar with the intent of conducting a clinical trial in India leading to regulatory approval and EndoBarrier distribution through a partnership with Apollo Sugar. Additionally, we focused on stabilizing the Company and continuing to address operational issues, continuing to reduce cash burn, working with non-United States regulatory bodies and continuing to develop our intellectual property.
We applied for, and received, an Investigational Device Exemption (IDE) authorizing us to conduct a pivotal trial in the United States designed to support our Pre-Market Authorization application.
We executed a clinical trial agreement and memorandum of understanding for a partnership in India with Apollo Sugar, the joint venture formed by Sanofi and Apollo Hospital Systems.
13
We executed an agreement with Intertek, our new Notified Body in order to pursue a CE mark for EndoBarrier and work towards commercial sales in the European Union and select countries in the Middle East.
We announced the release of clinically significant data from independent investigator-initiated trials and two ongoing Registries throughout the year.
We continued to develop our intellectual property position.
We hired key staff and moved to a new office location in Boston, Massachusetts.
Strategic Focus in 2019: The Path Forward
Our goal is to become the leading provider of alternative treatment options in type 2 diabetes and obesity. We intend to do this by conducting the following activities:
1) U.S. clinical operations: | Conduct Stage 1 of the EndoBarrier 18-1 pivotal trial in the United States. |
2) India partnership: | Conduct the EndoBarrier pivotal trial in India as part of the Apollo Sugar partnership. |
3) Gain CE Mark: | Work with our notified body to gain the EndoBarrier CE mark. |
4) Ongoing clinical data: | Support continued release of clinical data from registries and investigator- initiated clinical trials. |
5) Development of Intellectual Property: | Continue developing our intellectual property and protecting our patents. |
We will need to raise additional capital in 2019.
As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. We are currently focused primarily on our pivotal trial, which will support future regulatory submissions and potential commercialization activities. Until we are successful in gaining regulatory approvals, we are unable to sell our product in any market at this time. Without revenues, we are reliant on funding obtained from investment in the Company to maintain our business operations until we can generate positive cash flows from operations. We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.3 million. The Company expects to incur significant operating losses for the next several years. At December 31, 2018, the Company had approximately $3.8 million in cash and cash equivalents.
The Company will need to raise additional capital and restructure the terms of the 2017 Note before March 31, 2019 in order to continue to pursue its current business objectives as planned and to continue to fund its operations. The Company is looking to raise additional funds through any combination of additional equity and debt financings or from other sources. However, the Company has no guarantee that the 2017 Note will not mature on March 31, 2019 and has no guaranteed source of capital that will sustain operations into the second quarter of 2019. There can be no assurance that any such potential financing opportunities will be available on acceptable terms, if at all. If the Company is unable to raise sufficient capital on the Company’s required timelines and on acceptable terms to shareholders and the Board of Directors, it could be forced to cease operations, including activities essential to support regulatory applications to commercialize EndoBarrier. If access to capital is not achieved in the near term, it will materially harm the Company’s business, financial condition and results of operations to the extent that the Company may be required to cease operations altogether, file for bankruptcy, or undertake any combination of the foregoing. In such event, our shareholders may lose their entire investment in our company.
These factors raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
See “Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations — Liquidity and Capital Resources” for further information regarding our funding requirements.
14
Implications of Being an Emerging Growth Company and a Smaller Reporting Company
As a company with less than $1.07 billion in revenue during our most recently completed fiscal year, we qualify as an “emerging growth company” as defined in Section 2(a) of the Securities Act of 1933, as amended, which we refer to as the Securities Act, as modified by the Jumpstart Our Business Startups Act of 2012, or the JOBS Act. Additionally, Section Rule 12b-2 of the Securities Exchange Act of 1934, as amended, or the Exchange Act, establishes a class of company called a “smaller reporting company,” which effective September 10, 2018, was amended to include companies with a public float of less than $250 million as of the last business day of its most recently completed second fiscal quarter or, if such public float is less than $700 million, had annual revenues of less than $100 million during the most recently completed fiscal year for which audited financial statements are available. For the year ended December 31, 2018, we qualify as both an emerging growth company and as a smaller reporting company.
As an emerging growth company and/or smaller reporting company, we may take advantage of specified reduced disclosure and other requirements that are otherwise applicable, in general, to public companies that do not qualify for these classifications. These provisions include:
| • | Any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and financial statements, commonly known as an “auditor discussion and analysis”; |
| • | Reduced disclosure about our executive compensation arrangements; |
| • | No non-binding shareholder advisory votes on executive compensation or golden parachute arrangements; and |
| • | Exemption from the auditor attestation requirement in the assessment of our internal control over financial reporting. |
Emerging growth companies may take advantage of these exemptions for up to five years or such earlier time that we are no longer an emerging growth company. For as long as we continue to be an emerging growth company and/or a smaller reporting company, we expect that we will take advantage of the reduced disclosure obligations available to us as a result of those respective classifications. We would cease to be an emerging growth company if we have more than $1.07 billion in annual revenues as of the end of a fiscal year, if we are deemed to be a large-accelerated filer under the rules of the Securities and Exchange Commission, or the SEC, or if we issue more than $1.0 billion of non-convertible debt over a three-year-period. We will remain a smaller reporting company until we have a public float of $250.0 million or more as of the last business day of our most recently completed second fiscal quarter, and we could retain our smaller reporting company status indefinitely depending on the size of our public float.
The JOBS Act permits an emerging growth company to take advantage of an extended transition period to comply with new or revised accounting standards applicable to public companies. We are choosing to “opt out” of this provision.
Employees
As of March 1, 2019, we have 19 employees and contractors, 9 of which are full-time employees. None of our employees are represented by labor unions or covered by collective bargaining agreements.
Available Information
Financial and other information about us is available on our website. Our website address is www.gidynamics.com. We make available on our website, free of charge, copies of our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the U.S. Securities and Exchange Commission, or the SEC. The information contained in our website is not intended to be part of this filing.
15
Our business faces many risks. We believe the risks described below are the material risks that we face. However, the risks described below may not be the only risks that we face. Additional unknown risks or risks that we currently consider immaterial, may also impair our business operations. If any of the events or circumstances described below actually occur, our business, financial condition or results of operations could suffer, and the trading price of our CDIs could decline significantly. You should consider the specific risk factors discussed below together with the cautionary statements under the caption “Forward-Looking Statements” and the other information and documents that we file from time to time with the Securities and Exchange Commission, or SEC.
Risks Related to Our Business
We will need substantial additional funding and may be unable to raise capital when needed, which could force us to delay, reduce, or eliminate planned activities or result in our inability to operate as a going concern.
As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. We are currently focused primarily on our clinical trials, which will support future regulatory submissions and potential commercialization activities. Until we are successful in gaining regulatory approvals, we are unable to sell our product in any market at this time. Without revenues, we are reliant on funding obtained from investment in our Company to maintain our business operations until we can generate positive cash flows from operations. We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.3 million. The Company expects to incur significant operating losses for the next several years. At December 31, 2018 the Company had approximately $3.8 million in cash and cash equivalents.
The Company will need to raise additional capital and restructure the terms of the 2017 Note before March 31, 2019 in order to continue to pursue its current business objectives as planned and to continue to fund its operations. The Company is looking to raise additional funds through any combination of additional equity and debt financings or from other sources. However, the Company has no guarantee that the 2017 Note will not mature on March 31, 2019 and has no guaranteed source of capital that will sustain operations into the second quarter of 2019. There can be no assurance that any such potential financing opportunities will be available on acceptable terms, if at all. If the Company is unable to raise sufficient capital on the Company’s required timelines and on acceptable terms to shareholders and the Board of Directors, it could be forced to cease operations, including activities essential to support regulatory applications to commercialize EndoBarrier. If access to capital is not achieved in the near term, it will materially harm the Company’s business, financial condition and results of operations to the extent that the Company may be required to cease operations altogether, file for bankruptcy, or undertake any combination of the foregoing. In such event, our shareholders may lose their entire investment in our company.
These factors raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
In addition, if we do not meet our payment obligations to third parties as they become due, we may be subject to litigation claims and our credit worthiness would be adversely affected. Even if we are successful in defending against these claims, litigation could result in substantial costs and would be a distraction to management and may have other unfavorable results that could further adversely impact our financial condition.
As a result of the factors described above, our financial statements include a going-concern disclosure.
16
The terms of our indebtedness, in particular the terms of the 2017 Note, may result in the liquidation or winding down of our business, which would have a negative impact on holders of our CDIs and common stock.
In June 2017, we completed our 2017 Note secured financing for a gross amount of $5.0 million that accrues interest of 5% per annum compounded annually. In December 2018, the maturity date of the 2017 Note was extended from December 31, 2018 to March 31, 2019 in exchange for payment of approximately $394 thousand, which was the total accrued interest on the 2017 Note at December 31, 2018.
As of February 28, 2019, we currently have outstanding debt obligations of $7.3 million, including interest accrued through February 28, 2019. Approximately $5.5 million of this amount is due and payable on or before March 31, 2019 under the terms of the 2017 Note, unless (i) the 2017 Note is converted to equity prior to March 31, 2019 or (ii) we restructure the 2017 Note to extend the maturity date, as we did in December 2018. Our debt obligations are secured by all of our assets such that upon an event of default, we may be forced to sell some or all of our assets in order to make payment against the debt obligation when due. The proceeds from the sale of all of our assets may be insufficient to satisfy our debt obligations in full. As a result, it is unlikely that any proceeds would be available for distribution to holders of our CDIs or common stock on a sale of our assets or on a liquidation or winding down of our business.
Failure to achieve a positive outcome in the U.S. clinical trial of EndoBarrier could negatively impact our ability to raise additional capital and obtain regulatory approval in other countries.
We intend to conduct the U.S. pivotal trial of EndoBarrier under the FDA’s Investigational Device Exemption. We refer to this trial as 18-1 clinical trial in our statements and filings. Failure to achieve a positive outcome in this clinical trial could result in the failure of EndoBarrier to gain regulatory approval in the U. S. This outcome could negatively impact our ability to raise additional capital and obtain regulatory approval in other countries.
In order to complete the 18-1 clinical trial, we will need to enroll patients. If we are unable to enroll patients, or if the enrollment pace is slower than anticipated, it could have a negative effect on our ability to complete the clinical trial, which could adversely affect our business, operating results and prospects.
Conducting successful clinical studies will require the enrollment of patients, and suitable patients may be difficult to identify and recruit. Patient enrollment in clinical trials and completion of patient participation and follow-up depends on many factors, including the size of the patient population, the nature of the trial protocol, the desirability of, or the discomforts and risks associated with, the treatments received by enrolled subjects, the availability of appropriate clinical trial investigators and support staff, the proximity of patients to clinical sites, the ability of patients to comply with the eligibility and exclusion criteria for participation in the clinical trial and patient compliance. For example, patients may be discouraged from enrolling in our clinical trials if the trial protocol requires them to undergo extensive post-treatment procedures or follow-up to assess the safety and effectiveness of our product or if they determine that the treatments received under the trial protocols are not desirable or involve unacceptable risk or discomfort.
Development of sufficient and appropriate clinical protocols to demonstrate safety and efficacy are required and we may not adequately develop such protocols to support clearance or approval. Further, the FDA may require us to submit data on a greater number of patients than we originally anticipated and/or for a longer follow-up period or change the data collection requirements or data analysis applicable to our clinical trials. Delays in patient enrollment or failure of patients to continue to participate in a clinical trial may cause an increase in costs and delays in the approval or clearance and attempted commercialization of our product or result in the failure of the clinical trial. In addition, despite considerable time and expense invested in clinical trials, the FDA may not consider our data adequate to demonstrate safety and efficacy. Such increased costs and delays or failures could adversely affect our business, operating results and prospects.
The FDA has mandated certain stopping rules in the 18-1 clinical trial. If the stopping rules are triggered or other unanticipated adverse issues occur during the 18-1 clinical trial, the FDA may not permit the trial to continue. If that were to happen, our business, operating results and prospects would be materially and adversely affected.
Clinical trials involve the administration of the biological product candidate to patients under the supervision of qualified investigators, generally physicians not employed by or under the trial sponsor’s control. Clinical trials are conducted under protocols detailing, among other things, the objectives of the clinical trial, dosing procedures, subject selection and exclusion criteria, and the parameters to be used to monitor subject safety, including stopping rules that assure a clinical trial will be stopped if certain adverse events should occur. In the context of our 18-1 clinical trial, hepatic bleeding is an example of an adverse event that would be investigated and could trigger such stopping rules.
17
If, during the 18-1 clinical trial, the stopping rules are triggered or other unanticipated adverse issues occur, the FDA may prevent us from enrolling additional patients in the clinical trial or may not permit the clinical trial to be completed. If that were to happen, our business, operating results and prospects would be materially and adversely affected.
In order to commercialize our product in the U.S. and certain other countries, we will need to obtain regulatory and other approvals. Our inability to achieve, or a delay in achieving, such approvals could lead to the denial of marketing approval for EndoBarrier® or any of our other products.
At present, we have no regulatory approvals for marketing and sale of EndoBarrier. In November 2017, we received notification from SGS, the Company’s now former notified body in Europe, that they were withdrawing the Certificate of Conformity for EndoBarrier. The Certificate of Conformity is required for the sale of any product under CE marking. As a result, we are not permitted to supply the EndoBarrier, EndoBarrier Delivery System and EndoBarrier Retrieval System in Europe.
There is no guarantee that we will obtain additional approvals from regulatory bodies in the future, including the FDA in the U.S. In the U.S., we stopped our pivotal trial of EndoBarrier in 2015. We will not be able to obtain FDA approval to commercialize EndoBarrier in the U.S. until the 18-1 clinical trial is successfully completed and regulatory approval is obtained from the FDA through the PMA review process. The 18-1 trial will be conducted in two stages. At the end of the first stage, the FDA will review data collected to date from the 18-1 trial and must approve the continuation of the 18-1 trial to the second stage. There can be no assurance that the FDA will approve continuation of the trial at the end of the first stage.
Regulatory authorities in other countries may also require additional clinical trials. Any such clinical trials may be delayed, suspended or prematurely terminated because costs are greater than we anticipate or for a variety of other reasons, including: delay or failure in reaching agreement with the foreign regulatory authority(ies), on a trial design that we are able to execute; delay or failure in obtaining authorization to commence a trial or inability to comply with conditions imposed by a regulatory authority regarding the scope or design of a clinical trial; or regulators may require that we suspend or terminate our clinical trials for various reasons, including noncompliance with regulatory requirements, unforeseen safety issues or adverse side effects, or a finding that the participants are being exposed to unacceptable health risks. Any of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of marketing approval for EndoBarrier or any of our other products. Necessary regulatory approvals could also be delayed, which could significantly impact our ability to commercialize our technology in the U.S. and other countries.
We have a history of net losses and we may never achieve or maintain profitability.
We are a medical device company with a limited history of operations and have limited commercial experience with our product. As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. The Company continues to evaluate which markets are appropriate to continue pursuing reimbursement, market awareness and general market development and selling efforts, and continue to restructure its business and costs, establish new priorities, and evaluate strategic options. We expect to continue to incur significant operating losses for the foreseeable future as we incur costs, including those associated with commercializing our products, conducting clinical trials to test our products, attempting to secure regulatory approvals for our products (in the U.S. and other countries) and increased costs associated with being a public company in the U.S. As a result, if the Company remains in business, it expects to incur significant operating losses for the next several years.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.3 million. The Company expects to incur significant operating losses for the next several years. At December 31, 2018, the Company had approximately $3.8 million in cash and cash equivalents.
We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
18
We depend heavily on the success of our product, EndoBarrier. We cannot give any assurance that EndoBarrier will receive regulatory approval, which is necessary before it can be commercialized.
Our ability to generate product revenues will depend heavily on the successful commercialization of our product, EndoBarrier, if approved by regulatory agencies in the U.S. or abroad. We cannot be certain that EndoBarrier will be successful in clinical trials or receive regulatory approval. Further, EndoBarrier may not receive regulatory approval even if it is successful in clinical trials. If we do not receive regulatory approvals for EndoBarrier, we may not be able to continue our operations.
If we can obtain the required regulatory approvals in the U.S. and certain other countries, we expect to derive substantially all our revenue from sales of EndoBarrier. Accordingly, our ability to generate revenues in the future relies on our ability to market and sell this product.
The degree of market acceptance for EndoBarrier will depend on a number of factors, including:
| • | the efficacy, ease of use and perceived advantages and disadvantages of EndoBarrier over other available treatments and technologies for managing type 2 diabetes and obesity; |
| • | the prevalence and severity of any adverse events or side effects of EndoBarrier; |
| • | the extent to which physicians adopt EndoBarrier (which may be influenced by our ability to provide additional clinical data regarding the potential long-term benefits provided by EndoBarrier and the strength of our sales and marketing initiatives); |
| • | the price of EndoBarrier and the third-party coverage and reimbursement for procedures using EndoBarrier; |
| • | the extent to which reimbursement may be secured for each country in which EndoBarrier is commercialized; and |
| • | our ability to attract and retain professional sales personnel to drive EndoBarrier revenue. |
We cannot predict the outcome and timing of our current and future human clinical trials of EndoBarrier products. If the trials do not produce positive results, the commercial prospects for EndoBarrier will be impaired.
The results of current and future human clinical trials, whether investigator initiated, or Company sponsored, cannot be predicted. If EndoBarrier or new products that we develop and test in the future cause serious adverse events in future human clinical trials, these trials may need to be delayed or stopped. For example, we stopped our U.S. pivotal trial (ENDO) in 2015 because of adverse events associated with EndoBarrier in that trial. In addition, these clinical trials may not produce positive safety or efficacy results or may produce results that are not as favorable as those seen in previous clinical trials.
Negative safety or efficacy results of any future human clinical trials could require that we attempt to modify the EndoBarrier device or the treatment guidelines to address these issues and there is no guarantee that any potential modifications would be successfully developed.
If future human clinical trials of EndoBarrier products do not meet the required clinical specifications or cause serious adverse or unexpected events, such as those experienced in our U.S. pivotal trial, then these results could affect regulatory approvals and adoption and materially impact potential product sales and reimbursement. If we are not able to adequately address any adverse or unexpected events through training, education, changes in product design or product claims, this may significantly impair the commercial prospects for EndoBarrier.
19
Doctors may not accept EndoBarrier as a treatment option, which would harm our business and future revenues, if any.
The commercial success of EndoBarrier will require acceptance by physicians, who may be slow to adopt our product for the following reasons (among others):
| • | lack of long-term clinical data supporting patient benefits or cost savings over existing alternative treatments; |
| • | lack of experience with EndoBarrier and training time required before it can be used, which may drive preferences for other products or procedures; |
| • | lack of adequate payment to the physician for implanting the device or caring for the patient (driven by availability of adequate coverage and reimbursement for hospitals and implanting physicians); |
| • | perceived strength of products, procedures or pharmacotherapies as alternatives to EndoBarrier; and |
| • | perceived liability risks associated generally with the use of new products and procedures. |
Although we have developed relationships with physicians who are key opinion leaders in certain countries, we cannot assure that these existing relationships and arrangements can be maintained or that new relationships will be established in support of our products. If physicians do not consider our products to be adequate for the treatment of type 2 diabetes and obesity or if a sufficient number of physicians recommend and use competing products or pharmacotherapies, it could harm our business and future revenues, if any.
We have limited sales, marketing and distribution experience; therefore, we may be unable to successfully commercialize our products.
There can be no guarantee that we will be able to effectively commercialize our products. Developing direct sales, distribution and marketing capabilities will require the devotion of significant resources and require us to ensure compliance with all legal and regulatory requirements for sales, marketing and distribution. Failure to develop these capabilities and meet these requirements could jeopardize our ability to market our products or could subject us to substantial liability. In addition, for those countries where we commercialize our products through distributors or other third parties, we will rely heavily on the ability of our partners to effectively market and sell our products to physicians and other end users in those countries. We cannot guarantee that distributors or other third parties will be effective in commercializing our products.
We may compete against companies that have longer operating histories, more established or approved products, and greater resources than we do, which may prevent us from achieving market penetration with our products.
Competition in the medical device industry is intense and EndoBarrier will likely compete in part against more established procedures and products for the treatment of type 2 diabetes and obesity. Bariatric surgery, including gastric bypass surgery and the gastric band, have been used for many years with extensive publication histories on clinical effectiveness. Large multinational medical device companies sell supplies for these procedures and are formidable competitors to us. In addition, certain drugs have been approved, and are used, for the treatment of type 2 diabetes and obesity. Pharmaceutical companies with significantly greater resources than us market these drugs, and we may be unable to compete effectively against these companies.
Many of our competitors have significantly greater sales, marketing, financial and manufacturing capabilities than we have and have established reputations and/or significantly greater name recognition. Accordingly, there is no assurance that we will be able to win market share from these competitors or that these competitors will not succeed in developing products that are more effective or economic.
Additionally, we are likely to compete with companies offering new technologies in the future. We may also face competition from other medical therapies, which may focus on our target market as well as competition from manufacturers of pharmaceutical and other devices that have not yet been developed. Competition from these companies could adversely affect our business.
20
We do not have data regarding the long-term benefits of EndoBarrier, which may affect market acceptance. Furthermore, if the long-term data, once obtained, does not indicate that EndoBarrier is as safe or effective as other treatment options, our business would be harmed.
An important factor that may be relevant to market acceptance of EndoBarrier is whether it improves or maintains glycemic control and maintains weight loss over extended periods of time after removal of the device. While we have tested and evaluated our technology in several clinical trials with hundreds of patients which, in the aggregate, have shown that EndoBarrier is an effective treatment for type 2 diabetes and obesity, we do not yet have sufficient data to demonstrate any longer-term benefits of our product in the treatment of type 2 diabetes and obesity following removal of the device from the patient.
We are continuing to monitor some patients who participated in our clinical trials after device removal to determine the ongoing effects and longevity of results, however, we do not currently have long-term data that supports the safety and efficacy of EndoBarrier. Accordingly, we cannot provide assurance that the long-term data, once obtained, will prove lower HbA1c levels compared to alternative treatment options for type 2 diabetes. If the results obtained from our clinical trials indicate that EndoBarrier is not as safe or effective as other treatment options or as effective as our current short-term data would suggest, EndoBarrier may not be approved, or its adoption may suffer, and our business would be harmed.
If we fail to obtain and maintain adequate levels of reimbursement for our products by health insurers and other third-party payers, there may be no commercially viable markets for our products, or the markets may be much smaller than expected.
If our products are approved for sale, health care providers, including hospitals and physicians that may purchase our products, generally rely on third-party payers, particularly government-sponsored health care and private health insurance providers, to pay for all or a portion of the costs of the procedures, including the cost of the products used in such procedures. Reimbursement and health care payment systems vary significantly by country. Third-party payers may attempt to limit coverage and the level of reimbursement of new therapeutic products.
If we fail to obtain and maintain adequate levels of reimbursement for our products by health insurers and other third-party payers, there may be few commercially viable markets for our products, or the markets may be much smaller than expected. Third-party payers may demand additional clinical data requiring new clinical trials or economic models showing the cost savings of using our product, each of which would consume resources and may delay the decision on reimbursement. If the results of such studies are not satisfactory to third-party payers, then reimbursement may not be received in an acceptable amount or at all. In addition, the efficacy, safety, performance and cost-effectiveness of our products in comparison to any competing products or therapies may determine the availability and level of reimbursement.
We believe that future reimbursement may be subject to increased restrictions both in the U.S. and in international markets. Future legislation, regulation or reimbursement policies of health insurers or third-party payers may adversely affect the demand for our current and future products or limit our ability to sell these products on a profitable basis.
Our products may be subject to extensive, dynamic and ongoing regulation in countries where we plan to sell EndoBarrier, which may impede or hinder the approval or sale of our products and, in some cases, may ultimately result in our inability to obtain approval of certain products or may result in the recall or seizure of previously approved products.
The medical device industry is regulated extensively by governmental authorities, principally the FDA and corresponding state and foreign regulatory agencies and authorities, such as the E.U., legislative bodies and the European Economic Area, or EEA, Member State Competent Authorities. Additionally, the exit of Great Britain from the E.U. may create a separate and distinct regulatory agency with different requirements for regulatory approval in Great Britain.
Before we can market our products in the E.U., and in many other parts of the world, we must obtain and maintain CE Mark certification, which indicates that a product meets the essential requirements of applicable E.U. Directives and has been subject to the appropriate conformity assessment route. This conformity assessment procedure is often done through a self-certification, but depending on the type of product, may also require verification by an independent certification body, called a “notified body.” Notified bodies will also periodically conduct audits to ensure
21
continued compliance with the applicable requirements. The CE Mark allows free movement of products in the E.U., the EEA and Switzerland although any of the member countries may require medical devices to be registered and impose requirements relating to the language of the device information. Many non-European countries also recognize and accept the CE Mark. Failure to support product performance claims and demonstrate continued compliance with the applicable E.U. requirements, can result in the loss of the right to affix the CE Mark to the product, which prevents the product from being sold within the territory and in other countries that recognize the CE Mark.
Before we can market our products in other parts of the world, which are not regulated via CE mark or FDA approvals, we must obtain and maintain regulatory approvals in those countries. These approvals are dependent on clinical data and may require specific clinical trial data to be collected before applications for approval can be made. We do not currently have any assurance in any jurisdiction that such regulatory approvals will be granted or that more extensive clinical trials will not be required to secure such approvals. In India, we are partnering with Apollo Sugar to initiate and conduct clinical trials to generate data required by the Indian regulatory authorities, but as with any market, we do not have any assurance that these trials will generate the complete data set required to support a marketing approval.
In addition, even if we receive regulatory approval of our products in existing markets, we will be subject to ongoing regulatory requirements relating to our existing products in those markets. These include the requirement to timely file various reports with regulatory authorities in the countries in which we market our products, including reports of adverse events such as those experienced in our U.S. pivotal trial, events that may have caused or contributed to a death or serious injury and malfunctions that would likely cause or contribute to a death or serious injury if the malfunction were to recur. If these reports are not timely filed, regulators may impose sanctions, including temporarily suspending our market authorizations, and sales of EndoBarrier may suffer. In that case, we may be subject to product liability or regulatory enforcement actions, all of which could harm our business. Our failure to comply with EEA or other foreign regulations applicable in the countries where we operate could lead to the issuance of warning letters or untitled letters, the imposition of injunctions, suspensions or loss of regulatory clearance or approvals, product recalls, termination of distribution, product seizures or civil penalties. In the most extreme cases, criminal sanctions or closure of our manufacturing facilities are possible.
In addition, numerous new regulatory changes in the E.U. came into effect in 2016. We may not be able to comply with the new regulations and standards, despite compliance with prior regulations and standards.
In some countries, we may rely on our foreign distributors and agents to assist us in complying with foreign regulatory requirements, and we cannot be sure that they will always do so. If we or any of our suppliers, third-party manufacturers, distributors, agents or customers fail to comply with applicable requirements, we may face:
| • | adverse publicity; |
| • | investigations by governmental authorities; |
| • | fines and prosecutions; |
| • | inability to raise capital; |
| • | inability to attract and retain sales professionals; |
| • | inability to attract and agree to terms with business partners; |
| • | increased difficulty in obtaining required approvals; |
| • | loss of approvals already granted; |
| • | delays in purchasing decisions by customers or cancellation of existing orders; and |
| • | the inability to sell or import our products into such countries. |
Regulatory requirements affecting the development, manufacture and sale of medical devices are evolving and subject to future change. We cannot predict what impact, if any, those changes might have on our business. Failure to comply with regulatory requirements could have a material adverse effect on our business, financial condition and results of operations. Later discovery of previously unknown problems with a product or manufacturer could result in fines, delays or suspensions of regulatory approvals. The failure to receive product approval on a timely basis, or the withdrawal of product approval by regulatory agencies, could have a material adverse effect on our business, financial condition or results of operations.
22
We have had historical challenges with regulatory compliance, which led to a 2014 shipping hold in the E.U., multiple observations by the Therapeutic Goods Administration, which we refer to as the TGA, in Australia regarding our failure to comply with Essential Principals of the TGA, and compliance issues that led to the TGA’s cancellation of the EndoBarrier listing on the Australian Register of Therapeutic Goods in 2016. In addition, in May 2017, we received notification from our notified body, SGS, that the CE Mark for EndoBarrier had been suspended pending closure of non-conformances related to our quality management system required under International Organization for Standardization (“ISO”) regulations. On November 10, 2017, we received notification from SGS that SGS was withdrawing the Certificate of Conformance for EndoBarrier, ending the CE Marking of EndoBarrier in Europe and select Middle East countries.
Given the past challenges we have had in meeting all the requirements of a comprehensive global quality system, a risk exists that we will have compliance issues in the future.
Claims that our current or future products infringe or misappropriate the proprietary rights of others could adversely affect our ability to sell those products and cause us to incur additional costs.
If any third-party intellectual property claim against us is successful, we could be prevented from commercializing EndoBarrier or our other products.
There are numerous issued patents and pending patent applications in the U.S. and internationally that are owned by third parties and that contain patent claims in areas that are the focus of our product development efforts. We are aware of patents owned by third parties, to which we do not have licenses, that relate to, among other things, liner materials and anchoring. We have also employed individuals who were previously employed at other medical device companies, including competitors or potential competitors, which may result in claims from third parties that we have inadvertently or otherwise used or disclosed alleged trade secrets or other proprietary information of former employers of our employees.
In addition, because patent applications can take many years to issue, there may be currently-pending applications, unknown to us, which may later result in issued patents that pose a material risk to us.
We expect that we could be subject to third-party infringement claims if the number of competitors grows and the functionality of products and technology in different industry segments overlap. Third parties may currently have, or may eventually be issued, patents on which our current or future technologies may infringe.
If we are unable to obtain, maintain and enforce intellectual property protection covering our products, others may be able to make, use, or sell products substantially the same as ours, which could adversely affect our ability to compete in the market.
Our commercial success is dependent in part on obtaining, maintaining and enforcing intellectual property rights, including patents, covering EndoBarrier and our future products. If we are unable to obtain, maintain and enforce intellectual property protection covering our products, others may be able to make, use or sell products that are substantially the same as ours without incurring the sizeable development costs that we have incurred, which would adversely affect our ability to compete in the market. Certain patents covering EndoBarrier will begin to expire in 2023.
Even if our patents are determined by a court to be valid and enforceable, they may not be sufficiently broad or enforceable to prevent others from marketing products similar to ours or designing around our patents, despite our patent rights. Our products may also not have freedom to operate unimpeded by the patent rights of others.
In addition to patented technology, we rely on a combination of non-patented proprietary technology, trade secrets, processes, procedures, technical knowledge and know-how accumulated or acquired since inception. Any of our intellectual property rights could be challenged, invalidated, circumvented or misappropriated. In order to preserve and enforce our patent and other intellectual property rights, we may need to make claims or file lawsuits against third parties. This can entail significant costs and divert management’s attention from developing and commercializing EndoBarrier.
23
We rely on suppliers for certain key components in the manufacture of the EndoBarrier system. If these suppliers were to become unavailable to us, there could be delays in the commercialization of our products.
We rely on suppliers for several key components of EndoBarrier, in particular the material used to manufacture the sleeve used in EndoBarrier. Except as discussed below, we or our manufacturing partner, Proven Process Medical Devices, have supply agreements in place with suppliers of all components and services required to manufacture the EndoBarrier System. While we currently have adequate material required to make the sleeve to support us well into commercialization efforts, we do not presently have an active supply agreement with a manufacturer for additional material. Our reliance on third-party suppliers subjects us to risks that could harm our business, particularly with respect to the supply of key components or processes. Although we believe that alternative suppliers are available, the process of identifying and qualifying new suppliers who can produce the components to our specifications could cause delays in the commercialization of our products.
The use, misuse or off-label use of our products by physicians may harm our image in the marketplace or result in injuries that lead to product liability.
We cannot prevent a physician from using EndoBarrier for purposes outside of its eventual approved and intended use, which is known as off-label use. If physicians attempt to use our products off-label, there may be an increased risk of adverse events. Further, the use of our products for uses other than those uses for which our products have been approved may not effectively treat such conditions, which could harm our reputation in the marketplace among physicians and patients.
Physicians may also misuse our products or use improper techniques if they are not adequately trained, potentially leading to injury and an increased risk of product liability for us. Physicians may also treat patients from other countries where the product is not approved, and the physician is then unable to properly monitor the patient’s progress. If we are deemed to have engaged in the promotion of any of our products for off-label use, we could be subject to action by regulatory authorities and the imposition of sanctions, which could also affect our reputation and position within the industry.
Patients may not follow instructions from their physicians. Patients who ignore training and ignore their clinician’s request to comply with dietary instructions or pharmacotherapy label instructions, or to remove EndoBarrier at the end of the 12-month indication for treatment may be exposed to greater physical and clinical risk.
Product liability claims could damage our reputation or adversely affect our business or financial position.
We may be exposed to the risk of product liability claims, which are inherent in the design, manufacturing, marketing and use of medical devices and, in particular, implantable medical devices. We hold product liability insurance; however, adequate product liability insurance may not continue to be available on commercially-acceptable terms. Product liability claims may damage our reputation and, if our insurance coverage proves inadequate, may harm or destroy our business. Defending a suit, regardless of its merits, could be costly and could divert our management’s attention from our core business activities.
We will rely on a third-party manufacturer for our products and this manufacturer is required to comply with regulatory requirements. Failure of our manufacturer or suppliers to comply with regulatory requirements could disrupt the manufacturing process, which would, in turn, have a material adverse effect on our business and results of operations.
Future clinical trials and commercialization of our products require access to manufacturing facilities that meet applicable regulatory standards to manufacture a sufficient supply of our products. We will rely on a third-party manufacturer to meet the applicable regulatory requirements. Suppliers of components and products used to manufacture our products and our third-party manufacturer must also comply with applicable regulatory requirements. These often require significant time, resources, record-keeping and quality assurance efforts and subject us, our suppliers and third-party manufacturer to potential regulatory inspections and stoppages. Compliance with applicable regulatory requirements is subject to continual review and is rigorously monitored by regulatory authorities. Failure by us, our third-party manufacturer or suppliers to comply with regulatory requirements or failure to take satisfactory corrective action in response to an adverse inspection could result in enforcement actions that could have a material adverse effect on our business and results of operations.
24
In order to manufacture EndoBarrier in the quantities that we anticipate will be required to meet our clinical trial needs and potential future market demand, we will need to increase production capacity and efficiency over current levels, and our third-party manufacturer must therefore be able to provide us with sufficient quantities of our product in compliance with regulatory requirements, in accordance with agreed upon specifications, at acceptable cost and on a timely basis. Our potential future growth could strain the ability of our third-party manufacturer to deliver our product and obtain materials and components in sufficient quantities. Third-party manufacturers often experience difficulties in scaling up production, including problems with production yields and quality control and assurance. If we are unable to obtain a sufficient or consistent supply of EndoBarrier or any other product we are developing, or if we cannot do so efficiently, our revenue, business and financial prospects would be adversely affected.
We announced on July 26, 2017 that we executed an agreement with Proven Process Medical Devices to manufacture our product. We do not have extensive experience with this manufacturer, and we cannot guarantee that Proven Process Medical Devices will manufacture our product in a validated manner or to acceptable levels of quality. We are seeking other suppliers who will be able to manufacture our product in the event that our relationship with Proven Process Medical Devices is not successful.
We have been focused on efficiently running our operations in order to conserve resources which may impact our ability to retain our remaining employees and therefore we may not be able to sustain our business.
As of March 1, 2019, we have 19 employees and contractors (9 full time employees). Since our initial restructuring efforts in 2015, heavily dependent upon our ability to retain our remaining employees. The loss of service of any of our remaining employees, particularly members of our management team, may have an adverse effect on our business. Given the long period of reduced staff, the morale of our remaining employees may be lower, employees may be distracted and any one of our remaining employees could terminate his or her employment with us at any time. A departing employee’s expertise would be difficult to replace and the failure to do so on a timely basis could have a material adverse effect on our ability to achieve our business goals. There can be no assurance that we will have the financial resources to, or otherwise be successful in, retaining our remaining personnel and our failure to do so could have a material adverse effect on our business, financial condition and results of operations.
If we are unable to retain or hire key personnel, we may not be able to sustain or grow our business.
Our ability to operate successfully and manage our potential future growth depends significantly upon our ability to attract, retain and motivate highly-skilled and qualified research, technical, clinical, regulatory, sales, marketing, managerial and financial personnel. The competition for qualified employees in the medical device industry is intense and there are a limited number of persons with the necessary skills and experience.
Our performance is substantially dependent on our senior management and key technical staff to continue to develop and manage our operations. The loss or the inability to recruit and retain high-caliber staff could have a material adverse effect on us. We also rely on the technical and management abilities of certain key directors, key members of our executive team and employees, consultants and scientific advisers. The loss of any of these directors, members of the executive team, employees, consultants or scientific advisers could have an adverse effect on our business. On October 15, 2017, our former Vice President, International, resigned from the Company and on December 1, 2017, our former Chief Financial Officer departed the company for cost-management purposes. On April 13, 2018 the Company terminated the employment of its Chief Compliance Officer and Vice President, Finance and Accounting for cost-management purposes. We hired a Chief Financial Officer in December 2018, and a Vice President of Clinical and Regulatory Affairs in January, 2019.
We may be unable to effectively manage our anticipated growth.
To manage our anticipated growth and to commercialize our products, we will need to expand our operations (research and development, product development, quality, regulatory, manufacturing, sales, marketing and administrative). This expansion will place a significant strain on our management, infrastructure and operational and financial resources, particularly in light of the restructuring we have undertaken in the last few years. Specifically, we will need to manage relationships with various persons and entities participating in our clinical trials, quality systems, manufacturers, suppliers and other organizations, including various regulatory bodies in the U.S. and other jurisdictions. We may not be able to implement the required improvements in an efficient and timely manner and may discover deficiencies in existing systems and controls. The failure to accomplish any of these tasks could materially harm our business and our ability to commercialize EndoBarrier. As a result, our revenue, business and financial prospects would be adversely affected.
25
As long as we are listed on the Australian Securities Exchange (ASX), we will incur increased costs because we are a public company in the U.S. whose equity securities are listed on the Australian Securities Exchange.
As of December 31, 2013, we became subject to the periodic reporting requirements of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Although the existing listing of our Chess Depositary Interests, or CDIs, on the ASX requires us to file financial information and make certain other filings with the ASX, our status as a reporting company under the Exchange Act causes us to incur additional legal, accounting and other expenses, including costs related to compliance with the requirements of the Sarbanes-Oxley Act of 2002, insurance costs related to being an Exchange Act reporting company and other administrative costs. We also expect all these rules and regulations may make it more difficult and more expensive for us to obtain director and officer liability insurance and we may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and retain qualified individuals to serve on our Board of Directors or as executive officers. We cannot predict or estimate the amount of additional costs we may incur or the timing of such costs.
Our shares of our common stock are publicly traded on the ASX in the form of CDIs. As a result, we must comply with the ASX Listing Rules. We have policies and procedures that we believe are designed to provide reasonable assurance of our compliance with the ASX Listing Rules. If, however, we do not follow those procedures and policies, or they are not sufficient to prevent non-compliance, we could be subject to liability, fines and lawsuits. These laws, regulations and standards are subject to varying interpretations and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. We intend to invest resources to comply with evolving laws, regulations and standards, and this investment may result in increased general and administrative expenses and a diversion of management’s time and attention from revenue-generating activities to compliance activities. If, notwithstanding our efforts to comply with new laws, regulations and standards, we fail to comply, regulatory authorities may initiate legal proceedings against us and our business may be harmed.
We currently have material weaknesses in our internal controls over financial reporting. If we fail to maintain proper and effective internal controls, our ability to produce accurate and timely financial statements could be impaired, which could harm our operating results, and investors could lose confidence in our financial reports, which could adversely affect the value of our CDIs and common stock.
As a public company in the U.S., we are required, pursuant to Section 404 of the Sarbanes-Oxley Act of 2002, or Section 404, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting as of the end of the fiscal year. The controls and other procedures are designed to ensure that information required to be disclosed by us in the reports that we file with the SEC, is disclosed accurately and is recorded, processed, summarized and reported within the time periods specified in SEC rules and forms. Our first report on compliance with Section 404 was furnished in connection with our financial statements for the year ended December 31, 2014. As an Emerging Growth Company under the 2012 Jobs act and currently under the Smaller Reporting Company designation of Regulation S-X, the company has been and currently is exempted from the requirement to include an external auditor opinion on our internal controls in our filings. We continue to update, document and evaluate our internal control over financial reporting to the requirements of Section 404.
We continue to evaluate our existing internal controls over financial reporting using the standards adopted by the Public Company Accounting Oversight Board. During the course of our ongoing evaluation of the internal controls, we may identify areas requiring improvement, and may have to design enhanced processes and controls to address issues identified through this review. Specifically, in the three months ended September 30, 2018, we identified the following material weakness in our internal controls over financial reporting: our internal controls failed to prevent or timely detect unauthorized cash disbursements made from August 2016 through May 2018. Due to (i) a lack of controls within our bank portal to prevent manual edits to wire payment templates and (ii) ineffective reporting of payables to detect duplicate payments, these controls were not adequate to safeguard our cash assets from duplicate payments being diverted by a former employee to such employee’s personal bank account. The material weakness in our controls resulted in the inability to prevent and timely detect the fraud loss.
Remediating any deficiencies, significant deficiencies or material weaknesses that we or our independent registered public accounting firm may identify, including the material weakness described above, may require us to incur significant costs and expend significant time and management resources. We cannot assure you that any of the measures we implement to remedy any such deficiencies will effectively mitigate or remedy such deficiencies. The existence of one or more material weaknesses could affect the accuracy and timing of our financial reporting. Investors could lose confidence in our financial reports, and the value of our CDIs and common stock may be adversely affected, if our internal controls over financial reporting are found not to be effective by management or by an independent registered public accounting firm or if we make disclosure of existing or potential significant deficiencies or material weaknesses in those controls.
26
Fluctuations in foreign currency exchange rates could adversely affect our financial results.
In the past, when we sold EndoBarrier outside of the U.S., our activities produced revenues and we incurred expenses in a variety of different currencies, which exposed us to exchange rate risk. If we are successful in once again obtaining CE Mark or other foreign regulatory approvals that will allow us to sell EndoBarrier outside of the U.S., we will again be exposed to exchange rate risk. Exchange rate risk may affect our financial performance and position. Furthermore, some of our funds may be held in euros, Australian dollars or other currencies, while our functional currency is U.S. dollars. We are not currently hedging against exchange rate fluctuations, and consequently we will be at the risk of any adverse movement in the U.S. dollar exchange rate if we exchange funds held in one currency into another currency.
Our shares of common stock, in the form of CDIs, are listed on the ASX and priced in Australian dollars. However, our reporting currency is U.S. dollars. As a result, movements in foreign exchange rates may cause the price of our securities to fluctuate for reasons unrelated to our financial condition or performance and may result in a discrepancy between our actual results of operations and investors’ expectations of returns on our securities expressed in Australian dollars.
Risks Related to Our Industry
The medical device industry is subject to rapid technology change, which may result in obsolescence of our products.
The medical device industry is subject to rapid technology change. In order for us to remain competitive and to retain and build market share, we must continually develop new products as well as improve our existing ones. Accordingly, we must devote substantial resources to research, development and commercialization activities.
There can be no assurance that we will be successful in developing competitive new products and/or improving existing products so that such products remain competitive and avoid obsolescence. There can also be no assurance that any or all our research and development projects for new products will demonstrate safety and efficacy and result in commercial products, or that if such products are successfully designed and launched, they will be profitable.
Health care reform legislation could adversely affect our future revenue and financial condition.
In recent years, there have been numerous initiatives by governments throughout the world for comprehensive reforms affecting the delivery of and payment for health care. We cannot predict the changes that will be made and the effect such changes will have on the use of EndoBarrier. Decisions to increase or decrease reimbursement or coverage for treatments for type 2 diabetes and/or obesity could have a material impact on our business and results of operations.
CE Mark designation requires ongoing compliance, reporting and testing requirements established by regulatory agencies. While we have adopted comprehensive compliance programs to attempt to comply with these regulations, it is possible that some of our business activities could be subject to challenge. If our past or present operations, or those of our former independent sales agents and distributors, are found to be in violation of any of such applicable governmental regulations, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from health care reimbursement, product recall, or retraction of approval to market our product.
New legislation in the U.S. has also been enacted that imposes additional reporting requirements and penalties on the medical device industry. While we have adopted comprehensive compliance programs to attempt to comply with these regulations, it is possible that some of our business activities could be subject to challenge under one or more of such laws. If our past or present operations, or those of our former independent sales agents and distributors, are found to be in violation of any of such laws or any other applicable governmental regulations, we may be subject to penalties, including civil and criminal penalties, damages, fines, exclusion from health care reimbursement, product recall, or retraction of approval to market our product.
Health care related taxation could adversely affect our future financial condition.
In the U.S. the Affordable Care Act (ACA) of 2010 included a revenue-based tax imposed on the medical device industry. While the tax may be repealed prior to our earning significant U.S. revenues, this or other forms of medical device specific revenue-based taxation may have a significant impact on our net income after we commercialize EndoBarrier in the U.S.
27
Subsequent to future regulatory approvals for EndoBarrier, pricing pressures in the health care industry could lead to demands for price concessions, which could have an adverse effect on our business, financial condition or results of operations.
Due to the significant rise in health care costs over the past decade, numerous initiatives and reforms initiated by governments and third-party payers to curb these costs have resulted in difficulties in maintaining or raising the number and price of procedures. The increase in pricing pressure is driven by the competitive environment in the medical device industry as many larger companies cut prices as they struggle to retain market share.
The type 2 diabetes and obesity market has seen increasing resistance from payers regarding local and national reimbursement coverage. We expect that market demand, government regulation, third-party reimbursement policies and societal pressures could exert downward pressure on the prices of our products in the future after regulatory approval is granted and may adversely impact our business, financial condition or results of operations on a country by country basis.
Manufacturing facilities for medical devices must comply with applicable regulatory requirements.
Completion of our current and future clinical trials and commercialization of our products requires access to manufacturing facilities that meet applicable regulatory standards to manufacture a sufficient supply of our products. The third-party manufacturer and suppliers of components and parts that we intend to use to manufacture our products must also comply with applicable regulatory requirements, which often require significant time, money, resources and record-keeping and quality assurance efforts and subject us, our third-party manufacturer and our suppliers to potential regulatory inspections and stoppages.
Compliance with applicable regulatory requirements is subject to continual review and is rigorously monitored through periodic inspections by regulatory authorities. Failure by us, our third-party manufacturer or our suppliers to comply with regulatory requirements or failure to take satisfactory corrective action in response to an adverse inspection could result in enforcement actions, including a public warning letter, a shutdown of, or restrictions on, our ability to obtain sufficient quantities of our products, delays in approving or clearing a product, refusal to permit the import or export of our products or other enforcement action.
Inadequate funding for the FDA, the SEC and other government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent EndoBarrier from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal business functions on which the operation of our business rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years, including beginning on December 22, 2018, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA and the SEC, have had to furlough critical FDA, SEC and other government employees and stop critical activities. If a prolonged government shutdown occurs, it could significantly affect the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, in our operations as a public company, future government shutdowns could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Risks Related to Our CDIs and Our Common Stock
There is no current trading market for our common stock in the U.S. and no such market may develop.
Although our CDIs are currently listed on the ASX in Australia, there is not any current trading market for our CDIs or the underlying shares of common stock in the U.S. Should at some stage in the future, we determine to submit a listing application with Nasdaq (or any other exchange) to list our common stock on the Nasdaq Capital Market (or any
28
other exchange), Nasdaq may not approve our listing application. Even if Nasdaq (or such other exchange) approves our listing application, an active trading market for our common stock may never develop in the U.S. (or other relevant country) and shareholders may not be able to transfer or resell their common stock at its fair value, or at all.
We are eligible to be treated as an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012, and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies will make our common stock and CDIs less attractive to investors.
We are an “emerging growth company” as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including (1) not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, (2) reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements and (3) exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We could be an emerging growth company until the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common equity securities pursuant to an effective Securities Act registration statement, although circumstances could cause us to lose that status earlier, including if the market value of our common stock and CDIs held by non-affiliates exceeds $700.0 million as of any June 30 before that time or if we have total annual gross revenue of $1.0 billion or more during any fiscal year before that time, in which cases we would no longer be an emerging growth company as of the following December 31 or, if we issue more than $1.0 billion in non-convertible debt during any three-year period before that time, we would cease to be an emerging growth company immediately. We cannot predict if investors will find our common stock or CDIs less attractive because we may rely on these exemptions. If some investors find our common stock or CDIs less attractive as a result, there may be a less active trading market for our common stock or CDIs and our stock price may be more volatile.
Under the JOBS Act, emerging growth companies can also delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
We are eligible to be treated as an “Smaller Reporting Company” as defined in the amended regulation S-K, section 210, and we cannot be certain if the reduced disclosure requirements applicable to Smaller Reporting Companies will make our common stock and CDIs less attractive to investors.
The Securities and Exchange Commission amended regulation S-K, section 210 in September 2018 to reduce the scale of reporting for Smaller Reporting Companies, keeping many of the scaled reporting requirements from the Emerging Growth Company requirements under the JOBS act. Smaller Reporting Companies are defined based on public capital and revenue limits and we qualify as a Smaller Reporting Company for the period ended December 31, 2018. As a result, our reporting is compliant with the Smaller Reporting Company requirements. We cannot predict if investors will find our common stock or CDIs less attractive because we may rely on these reporting exemptions. If some investors find our common stock or CDIs less attractive as a result, there may be a less active trading market for our common stock or CDIs and our stock price may be more volatile.
Changes in economic conditions may adversely affect our business.
Changes in the general economic climate in which we operate may adversely affect our financial performance and the value of our assets. Factors that contribute to that general economic climate include:
| • | contractions in the world economy or increases in the rate of inflation; |
| • | international currency fluctuations; |
| • | changes in interest rates; |
| • | new or increased government taxes or duties or changes in taxation laws; or |
| • | changes in government regulatory policy. |
29
Stock market fluctuations may adversely affect the price of our CDIs and common stock.
There are a number of risks associated with any stock market investment. Our CDIs have been traded on the ASX since September 7, 2011. The price of our CDIs has been, and is likely to continue to be, volatile, which means that the value could decline substantially within a short period of time. The value of the CDIs will be determined by the stock market and will be subject to a range of factors beyond our control. These factors include movements in local and international stock exchanges, local interest rates and exchange rates, domestic and international economic and political conditions, government taxation, market supply, competition and demand and other legal, regulatory or policy changes.
The trading volume of our CDIs is low, which may result in reduced liquidity for our shareholders.
Our CDIs are only listed on the ASX and are not listed for trading on any other securities exchanges in Australia, the U.S. or elsewhere. As such, there can be no guarantee that an active market in the CDIs will develop or continue, or that the market price of the CDIs will increase. If a market does not develop or is not sustained, it may be difficult for investors to sell their CDIs. Furthermore, the market price for the CDIs may fall or be made more volatile because of the relatively low volume of trading in our securities. When trading volume is low, significant price movement can be caused by trading in a relatively small number of shares.
Sales of a substantial number of CDIs, or the perception that these sales may occur, could cause the market price of our CDIs to decline. Sales by our current shareholders of a substantial number of CDIs, or the expectation that such sales may occur, could significantly reduce the market price of our CDIs. We may also offer additional CDIs in subsequent offerings, which may adversely affect the market price for the CDIs.
Some of our current shareholders can exert control over us and may not make decisions that are in the best interests of all shareholders.
As of March 15, 2019, 4 shareholders and their affiliated entities owned approximately 80% of our outstanding shares of common stock, or CDIs representing common stock, in the aggregate. As a result, these shareholders, if they act together, would be able to exert a significant degree of influence over our management and affairs and over matters requiring shareholder approval, including the election of directors and approval of significant corporate transactions. This concentration of ownership may harm the market price of the CDIs and our common stock by delaying or preventing a change in control, even if a change is in the best interests of our other shareholders. This concentration of ownership may also delay, deter or prevent acts that may be favored by our other stockholders, as the interests of these stockholders may not always coincide with the interests of our other stockholders. In addition, this concentration of share ownership may adversely affect the trading price of our shares because it may limit the trading volume and purchase demand for outstanding shares, could adversely affect our stock price should any of these stockholders elect to sell some or all of their shares, and investors may perceive disadvantages in owning shares in a company with significant stockholders.
Provisions of our certificate of incorporation, our bylaws and Delaware law could make an acquisition of us more difficult.
Certain provisions of our certificate of incorporation and bylaws could discourage, delay or prevent a merger, acquisition or other change of control that our shareholders may consider favorable, including transactions in which our shareholders might otherwise receive a premium for their CDIs or common stock. These provisions could also limit the price that investors might be willing to pay in the future for the CDIs or common stock, thereby depressing the market price of these securities. Shareholders who wish to participate in these transactions may not have the opportunity to do so. In addition, we are incorporated in the State of Delaware and, as such, are governed by the provisions of Section 203 of the Delaware General Corporation Law, which may, unless certain criteria are met, prohibit large shareholders, in particular those owning 15% or more of the voting rights on our common stock, from merging or combining with us for a prescribed period of time.
We do not intend to pay cash dividends on our common stock in the foreseeable future.
We have never declared or paid any cash dividends on our shares of common stock, and we currently do not anticipate paying any cash dividends in the foreseeable future. We intend to retain any earnings to finance the development and expansion of our products and business. Accordingly, our shareholders will not realize a return on their investment unless the trading price of our CDIs or common stock appreciates.
30
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Our ability to utilize our federal net operating losses and federal tax credits may be limited under Sections 382 and 383 of the Internal Revenue Code. The limitations apply if an ownership change, as defined by Section 382, occurs. Generally, an ownership change occurs when certain shareholders increase their aggregated ownership by more than 50 percentage points over their lowest ownership percentage in a testing period (typically three years). We may already be subject to Section 382 limitations due to previous ownership changes. In addition, future changes in stock ownership may also trigger an ownership change and, consequently, a Section 382 limitation. Due to the significant complexity and cost associated with a change in control study, and the expectation of continuing to incur losses whereby the net operating losses and federal tax credits are not anticipated to be used in the foreseeable future, we have not assessed whether there have been changes in control since our formation. If we have experienced changes in control at any time since our formation, utilization of our net operating losses or research and development credit carryforwards would be subject to annual limitations under Section 382. Any limitation may result in expiration of a portion of the net operating loss or research and development credit carryforwards before utilization, which would reduce our gross deferred tax assets and corresponding valuation allowance. As a result, if we earn net taxable income, our ability to use our pre-change net operating loss carryforwards to offset U.S. federal taxable income may be subject to limitations, which could potentially result in increased future tax liability to us.
Item 1B. UNRESOLVED STAFF COMMENTS
None.
In June 2016, we entered into a non-cancelable agreement to lease approximately 4,200 square feet of office and laboratory space in Boston, Massachusetts. The lease commenced in June 2016 and expired in April 2018.
In December 2018, we entered into a membership agreement with WeWork for 985 square feet of office space located in Boston, Massachusetts. The committed lease term expires in May 2019 and contains a two-month cancellation provision. The company is exploring term extensions or other suitable options for office space subsequent to the current term expiry with WeWork.
None.
ITEM 4. MINE SAFETY DISCLOSURES
Not Applicable.
31
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Market Information
Our CDIs, each representing one-fiftieth of one share of our Common Stock after giving effect to our April 9, 2015 1:10 reverse stock split, have been listed on the ASX under the trading symbol “GID” since September 7, 2011. Prior to such time there was no public market for our securities
On December 31, 2018, the last reported sale price of our CDIs was A$0.016 per CDI, or $0.80 per share of Common Stock.
As of December 31, 2018, 1,413,412 of our shares were subject to outstanding options, restricted stock units and warrants to purchase shares of Common Stock.
Holders
As of January 30, 2019, we had 19,277,546 shares of Common Stock issued and outstanding with approximately 16 holders of record. The holders included CHESS Depositary Nominees Pty Limited, or CDN, which held 19,179,737 shares of our Common Stock in the form of CDIs on behalf of the CDI holders; there were approximately 874 registered owners of our CDIs on January 30, 2019.
Equity Compensation Plan Information
The information required to be disclosed by Item 201(d) of Regulation S-K, “Securities Authorized for Issuance Under Equity Compensation Plans” is referenced under Item 12 of Part III of this Annual Report on Form 10-K.
ITEM 6. SELECTED CONSOLIDATED FINANCIAL DATA
Not applicable.
32
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and related notes to those financial statements appearing elsewhere in this Annual Report on Form 10-K. This discussion and analysis contains forward-looking statements that involve risks, uncertainties and assumptions. You should review the “Risk Factors” section of this Annual Report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements described in the following discussion and analysis.
Overview
We are a clinical stage medical device company located in Boston, Massachusetts. We have developed EndoBarrier, a medical device intended for treatment of patients with type 2 diabetes and obesity, and we are taking the steps necessary to obtain the regulatory approvals required to market this product. In order to market EndoBarrier in the U.S., we must obtain approval from the FDA. In order to market EndoBarrier outside of the U.S., we are required to comply with various regulations imposed by the countries in which we seek to sell the product.
In 2010, EndoBarrier received CE Marking for sale in the European Union and in 2011, EndoBarrier was listed on the Australian Register of Therapeutic Goods. As a result, during 2013 and 2014, we received approximately $2.8 million and $2.25 million in revenue from the sale of EndoBarrier in Europe, South America and the Asia Pacific region. In the U.S. in 2013, we began enrollment of patients in the initial pivotal trial of EndoBarrier, which we refer to as the ENDO Trial.
In the third quarter of 2015, we announced our decision to discontinue the ENDO Trial because patients were experiencing a higher than previously observed level of hepatic (liver) abscesses. In the fourth quarter of 2016, we received formal notification from the Therapeutic Goods Administration of the Australian government of the cancellation of EndoBarrier’s inclusion on the Australian Register of Therapeutic Goods. In the fourth quarter of 2017 we received formal notification of CE mark withdrawal from our notified body in Europe, preventing the sale of EndoBarrier in Europe and select Middle Eastern countries. We undertook comprehensive cost-cutting measures throughout 2015 and 2016, including significantly reducing the number of our employees.
Following our decision to discontinue the ENDO Trial, we undertook significant investigational and scientific analysis with the goal of reducing the incidence rate and severity of hepatic abscess that present concurrent with the EndoBarrier treatment. This investigational work focused on understanding the root cause of hepatic abscess and how to reduce the rate of occurrence. This included: DNA analysis of normal EndoBarrier removals as well as hepatic abscess EndoBarrier removals, numerous meta-analyses and responder cohort analyses, investigation into the contributing factors represented by proton pump inhibitors (PPIs), leaky gut syndrome and microbiome analyses, among other research. This allowed the company to modify the medications utilized with EndoBarrier, most notably discontinuation of chronic double-dose PPI usage during EndoBarrier implant.
As a result of the efforts described above, in August 2018, we received an investigational device exemption from the FDA to begin enrollment in a pivotal trial evaluating the safety and efficacy of EndoBarrier in the United States pending Institutional Review Board approval. In this report, we refer to this pivotal trial as the GID 18-1 clinical trial.
For financial reporting purposes, we have one reportable segment, which designs, manufactures and plans to market EndoBarrier.
To date, we have devoted substantially all of our efforts to research and development, business planning, clinical research, clinical study management, reimbursement development, product commercialization, acquiring operating assets and raising capital. We have incurred significant operating losses since our inception in 2003. As of December 31, 2018, we had an accumulated deficit of approximately $267 million. We expect to incur net losses for the next several years while we continue to evaluate which markets are appropriate to continue pursuing reimbursement, market awareness and general market development efforts, and continue to restructure our business and costs, establish new priorities, continue limited research, and evaluate strategic options.
To date, we have raised net proceeds of approximately $269 million through the issuance of convertible debt and sales of our equity securities.
33
In October 2016, we received final cancellation notification from the Therapeutic Goods Administration (“TGA”) for the listing of EndoBarrier on the Australian Register of Therapeutic Goods (“ARTG”).
In May 2017, we received notification from our notified body, SGS United Kingdom Limited (“SGS”), that the CE Mark for EndoBarrier had been suspended pending closure of non-conformances related to its quality management system required under International Organization for Standardization (“ISO”) regulations.
On November 10, 2017, we received notification that SGS was withdrawing the Certificate of Conformance for EndoBarrier, ending the CE Marking of EndoBarrier in Europe and select Middle East countries.
In December 2017, we received notification from the Medicines and Healthcare Products Regulatory Agency (“MHRA”) that all EndoBarrier delivery systems (liners) in customers’ inventory need to be returned to the Company.
In August 2018, we received notification from the FDA that it had approved the IDE application for EndoBarrier, pending Institutional Review Board (IRB) approval, which was received in February 2019.
From its inception in 2003 to its initial public offering (“IPO”) in 2011, we have been financed by a series of preferred stock financings. In September 2011, the Company completed its initial public offering (“IPO”) of common stock in the form of CHESS Depositary Interests (“CDIs”) in Australia. As a result of the IPO and simultaneous private placement in the U.S., the Company raised a total of approximately $72.5 million in proceeds, net of expenses and repayment of $6.0 million of the Company’s Convertible Term Promissory Notes. Additionally, in July and August 2013, the Company issued CDIs on the Australian Securities Exchange (“ASX”) through a private placement and Share Purchase Plan (“SPP”), which raised a total of approximately $52.5 million, net of expenses. In May 2014, the Company raised an additional total of approximately $30.8 million, net of expenses, when it issued CDIs on the ASX through a private placement.
On December 20, 2016, the Company completed a private placement issue of 69,865,000 CDIs (1,397,300 shares) at an issue price of $0.022 per CDI raising approximately $1.0 million, net of issuance costs. In January 2017, the Company completed the issue of 12,481,600 CDI’s (249,632 shares) to eligible investors under a Security Purchase Plan for approximately $0.83 per share (A$0.022 per CDI) resulting in net proceeds after issuance costs of approximately $0.2 million.
In June 2017, the Company completed a Convertible Term Promissory Note (the “2017 Note”) secured financing with its largest shareholder Crystal Amber for a gross amount of $5.0 million that accrues interest at 5% per annum compounded annually. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. The Note was due by December 31, 2018 and contains provisions for conversion during its term and is also subject to security arrangements in favor of Crystal Amber(See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
In January and March 2018, the Company raised approximately $1.6 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom.
In May 2018, the Company completed a Convertible Term Promissory Note (the “2018 Note”) and Warrant (the “2018 Warrant”) financing with its largest shareholder Crystal Amber for a gross amount of $1.75 million that accrues interest at 10% per annum compounded annually. The Note and Warrant financing was approved in a vote of the shareholders of GI Dynamics during the Annual Meeting of Stockholders held on May 24, 2018. The 2018 Note matures and the 2018 Warrant expires on May 30, 2023. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. (See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
In September 2018, the Company received commitments for a private placement of approximately $5 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom. The first tranche of $2.2 million closed and cash was received in September 2018. The second and final tranche of $2.8 million was contingent upon shareholder approval which was received in October 2018. Cash proceeds were received in November 2018.
34
In December 2018, the maturity of the 2017 note was extended to March 31, 2019 in exchange for payment of $394 thousand which was the total accrued interest on the 2017 Note at December 31, 2018.
Our costs include employee salaries and benefits, compensation paid to consultants, materials and supplies for research, costs associated with development activities including materials, sub-contractors, travel and administration, legal expenses, sales and marketing costs, general and administrative expenses, and other costs associated with an early stage, publicly-traded medical technology company. We currently have 9 full-time employees. The number of employees required to support our activities as we move the EndoBarrier through the GID 18-1 clinical trial, as well as in the areas of research and development, sales and marketing, and general and administrative functions, may increase. We expect to continue to incur consulting expenses related to technology development that will increase as we enter into the recruitment phase of the 18-1 and India clinical trials, and we expect to continue to incur expenses to protect our intellectual property.
The amount that we spend for any specific purpose may vary significantly from quarter to quarter or year to year, and could depend on a number of factors including, but not limited to, the pace of progress of the GID-18 and India clinical trials, our commercialization and development efforts and actual needs with respect to development and research.
Research, development, and commercial acceptance of new medical technologies are, by their nature, unpredictable. Although we will undertake development and commercialization efforts with reasonable diligence, there can be no assurance that the net proceeds from our securities offerings will be sufficient to enable us to develop our technology to the extent needed to create future sales to sustain operations. If the net proceeds from these offerings are insufficient for this purpose, we will consider other options to continue our path to commercialization, including, but not limited to, additional financing through follow-on equity offerings, debt financing, co-development agreements, sale or licensing of developed intellectual or other property, or other alternatives.
We cannot assure that our technology will be accepted, that we will ever earn revenues sufficient to support our operations, or that we will ever be profitable. Furthermore, we have no committed source of financing and we cannot assure that we will be able to raise money as and when we need it to continue our operations. If we cannot raise funds as and when we need them, we may be required to scale back our development plans by reducing expenditures for employees, consultants, business development and marketing efforts or to otherwise severely curtail, or even to cease, our operations.
Our corporate headquarters are located in Boston, Massachusetts. We lease approximately 985 square feet of office space for approximately $18,000 per month pursuant to a short-term space and services lease which expires in May 2019. This space is adequate for our current operations.
Critical Accounting Policies and Estimates
Our discussion and analysis of our financial condition and results of operations is based upon our consolidated financial statements prepared in accordance with generally accepted accounting principles in the U.S. The preparation of these financial statements requires us to make certain estimates and assumptions that may affect the reported amounts of assets and liabilities, and the reported amounts of revenues and expenses during the reported periods and related disclosures. These estimates and assumptions, including those related to revenue recognition, inventory valuation including reserves for excess and obsolete inventory, impairment of long-lived assets, valuation of warrant liabilities income taxes including the valuation allowance for deferred tax assets, research and development expenses, estimates used in assessing our ability to continue as a going concern, contingencies and stock-based compensation are monitored and analyzed by us for changes in facts and circumstances, and material changes in these estimates could occur in the future. We base our estimates on our historical experience and various other assumptions that are believed to be reasonable under the circumstances. Actual results may differ from our estimates under different assumptions or conditions.
We believe that our application of the following accounting policies, each of which require significant judgments and estimates on the part of management, are the most critical to aid in fully understanding and evaluating our reported financial results. Our significant accounting policies are more fully described in Note 2, “Summary of Significant Accounting Policies and Basis of Presentation”, to our consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K.
35
The preparation of consolidated financial statements in accordance with generally accepted accounting principles in the U.S. requires the Company’s management to make estimates and judgments that may affect the reported amounts of assets, liabilities, revenues and expenses, and the related disclosure of contingent assets and liabilities. On an ongoing basis, the Company’s management evaluates its estimates, including those related to revenue recognition, inventory valuation including reserves for excess and obsolete inventory, impairment of long-lived assets, income taxes including the valuation allowance for deferred tax assets, research and development, contingencies, valuation of warrant liabilities, estimates used to assess its ability to continue as a going concern and stock-based compensation. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable, the results of which form the basis for making judgments about the carrying values of assets and liabilities. Actual results may differ from these estimates under different assumptions or conditions. Changes in estimates are reflected in reported results in the period in which they become known.
Revenue Recognition
We had no revenues for the year ended December 31, 2018. For the year ended December 31, 2017, we had revenues of $53 thousand of sales of EndoBarrier directly to health care providers or to third-party distributors who resell to health care providers. The adoption of ASC 606 requires us to follow a multi-step process to determine the elements of the implied contract with the customer when we sell and support our products. The steps we follow are to:
| o | Identify the implied contract with the customer: We recognize a contract exists with a customer when an authorized purchase order is submitted, placing a binding order with the Company such that transfer of the product and/or services, including any implied performance obligations on the part of the Company, goods sold (“G/S”) and the expectation to receive consideration for the G/S. |
| o | Identify the explicit and implicit performance obligations within the contract: Our G/S offering includes an underlying product and closely-related ongoing technical support for the product and its use. Assurance warranties offering a right of return for defective products are excluded from the contractual performance obligations under ASC 606. Technical support, as the remaining performance obligations does not have standalone value as it is only available for our product and only to those who have purchased and are using our product. |
| o | Determine the transaction price: The new guidance defines the transaction price as the expected amount to be received for G/S. This includes probability and magnitude of customer discounts, any expected collection risk, and any additional factors which may reduce the ultimate amount expected to be received. Estimates of these expected amounts will be based on established pricing and historical patterns once captured and management judgement until sufficient historical data is available. |
| o | The transaction price is allocated to the product and performance obligations of the contract: The allocation price will be allocated to the various G/S components, especially to any performance obligations that remain after product delivery or product implantation. In our current model, the transfer of title of the product occurs on customer receipt of shipment; product invoicing will occur after shipment and collectability expectations will be confirmed or adjusted after a sufficient period to gauge likelihood of eventual collection; and the technical support is present from shipment and is discharged after successful EndoBarrier sleeve retrieval (approximately one year after successful implantation). |
| o | As the performance obligations are discharged, the associated allocated portion of the revenue will be recognized. In our current model, we expect to book a) revenue associated with the product allocation on shipment of the product, b) revenue associated with any true up/adjustment to collectability as payment is received or when non-payment is probable, and c) revenue associated with technical support is discharged when the EndoBarrier is successfully retrieved from the patient. |
36
We state inventory at the lower of first-in, first-out cost or net realizable value. We record a provision for excess, expired, and obsolete inventory based primarily on estimates of forecasted revenues. A significant change in the timing or level of demand for products as compared to forecasted amounts may result in recording additional provisions for excess, expired, and obsolete inventory in the future. When capitalizing inventory, we consider factors such as status of regulatory approval, alternative use of inventory, and anticipated commercial use of the product.
The valuation of inventory also required us to estimate obsolete or excess inventory. We maintained reserves for excess and obsolete inventory based on forecasted product sales, new product introductions by us or our competitors, product expirations and historical experience. The inventory reserves we recognized were based on estimates of how these factors were expected to impact the amount and value of inventory we expected to sell. Forecasting demand for EndoBarrier in a market in which there are few, if any, comparable approved devices and for which reimbursement from third-party payers is limited. We fully reserved our inventory as of December 31, 2017 and subsequently wrote off all inventory and reserves in 2018 as the materials on hand were not expected to be usable for future sales. There is no inventory or reserves against inventory on the balance sheet at December 31, 2018.
Research and Development Costs
Research and development costs are expensed when incurred. Research and development costs include costs of all basic research activities as well as other research, engineering, and technical effort required to develop a new product or service or make significant improvement to an existing product or manufacturing process. Research and development costs also include preapproval regulatory and clinical trial expenses.
Stock-Based Compensation
We account for stock-based compensation in accordance with the Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, 718, Stock Compensation, or ASC 718, which requires that stock-based compensation be measured and recognized as an expense in the financial statements and that such expense be measured at the grant date fair value.
For awards that vest based on service conditions, we use the straight-line method to allocate compensation expense to reporting periods. The grant date fair value of options granted is calculated using the Black-Scholes option pricing model, which requires the use of subjective assumptions including volatility, expected term and the fair value of the underlying Common Stock, among others.
The assumptions used in determining the fair value of stock-based awards represent management’s best estimates, but these estimates involve inherent uncertainties and the application of management judgment. As a result, if factors change, and we use different assumptions, our stock-based compensation could be materially different in the future. The risk-free interest rate used for each grant is based on a zero-coupon U.S. Treasury instrument with a remaining term similar to the expected term of the stock-based award. Because we do not have a sufficient history to estimate the expected term, we use the simplified method for estimating the expected term. The simplified method is based on the average of the vesting tranches and the contractual life of each grant. We use historical price volatility data to estimate our expected future stock volatility. We have not paid and do not anticipate paying cash dividends on our shares of Common Stock; therefore, the expected dividend yield is assumed to be zero.
The Company elected to adopt ASU 2016-09 in the first quarter of 2017 retrospectively to January 1, 2017. As a result of adopting ASU No. 2016-09 during the year ended December 31, 2017, the Company adjusted its accumulated deficit related to the accounting policy election to recognize the impact of share-based award forfeitures only as they occur rather than by applying an estimated forfeiture rate as previously required. ASU No. 2016-09 requires that this change be applied using a modified-retrospective transition method by means of a cumulative-effect adjustment to accumulated deficit as of the beginning of the fiscal year in which the guidance is adopted. As a result of this adoption, the Company recorded a decrease to accumulated deficit of approximately $28 thousand with an offset to Additional Paid in Capital as of January 1, 2017.
We periodically issue performance-based awards. For these awards, vesting will occur upon the achievement of certain milestones. When achievement of the milestone is deemed probable, we expense the compensation of the respective stock award over the implicit service period.
37
Stock awards to non-employees are accounted for in accordance with ASC 505-50, Equity-Based Payments to Non-Employees (“ASC 505-50”). The measurement date for non-employee awards is generally the date performance of services required from the non-employee is complete. For non-employee awards that vest based on service conditions, the Company expenses the value of the awards over the related service period, provided they expect the service condition to be met. The Company records the expense of services rendered by non- employees based on the estimated fair value of the stock option using the Black-Scholes option pricing model over the contractual term of the non-employee. The fair value of unvested non-employee awards is remeasured at each reporting period and expensed over the vesting term of the underlying stock options on a straight-line basis.
Impairment of Long-Lived Assets
We regularly review the carrying amount of our long-lived assets to determine whether indicators of impairment may exist that warrant adjustments to carrying values or estimated useful lives. If indications of impairment exist, projected future undiscounted cash flows associated with the asset are compared to the carrying amount to determine whether the asset’s value is recoverable. If the carrying value of the asset exceeds such projected undiscounted cash flows, the asset will be written down to its estimated fair value.
Foreign Currency Remeasurement
The functional currency of GID Europe Holding B.V., GID Europe B.V., GID Germany GmbH and GI Dynamics Australia Pty Ltd is the U.S. dollar. Foreign denominated monetary assets are remeasured into U.S. dollars using the exchange rate in effect at the consolidated balance sheet date while non-monetary consolidated balance sheet items are remeasured at historical rates. Expenses are remeasured using the average exchange rate in effect during the reporting period. Gains and losses arising from remeasurement of our wholly owned subsidiaries’ financial statements are included in the determination of net loss.
Emerging Growth Company Status
The JOBS Act permits an “emerging growth company” such as us to take advantage of an extended transition period to comply with new or revised accounting standards applicable to public companies. We have chosen to “opt out” of this provision and, as a result, we will comply with new or revised accounting standards as required when they are adopted. This decision to opt out of the extended transition period under the JOBS Act is irrevocable.
Smaller Reporting Company Status
The Securities and Exchange Commission amended regulation S-K, section 210 in September 2018 to reduce the scale of Smaller Reporting Companies. Smaller Reporting Companies are defined based on public capital and revenue limits and we qualify as a Smaller Reporting Company for the period ended December 31, 2018. As a result, our reporting is compliant with the Smaller Reporting Company requirements.
Results of Operations
The following is a description of significant components of our operations, including significant trends and uncertainties that we believe are important to an understanding of our business and results of operations.
Year Ended December 31, 2018 Compared to Year Ended December 31, 2017
|
| Years Ended |
|
|
|
| ||||||||||
| December 31, |
|
| Change |
| |||||||||||
|
| 2018 |
|
| 2017 |
|
| $ |
|
| % |
| ||||
|
| (dollars in thousands) |
|
|
|
|
| |||||||||
Revenue |
| $ | — |
|
| $ | 53 |
|
| $ | (53 | ) |
|
| — |
|
Cost of revenue |
|
| — |
|
|
| 265 |
|
|
| (265 | ) |
|
| — |
|
Gross loss |
| $ | — |
|
| $ | (212 | ) |
| $ | 212 |
|
|
| — |
|
Revenue. The decrease in revenue to $0 for the year ended December 31, 2018 compared to $53 thousand for the year ended December 31, 2017 was primarily due to the suspension of the CE Mark in May 2017 and subsequent
38
notification from SGS on November 10, 2017 that SGS was withdrawing the Certificate of Conformance for EndoBarrier, ending the CE Marking of EndoBarrier in Europe and select Middle East countries.
In the near-term, we intend to focus our efforts on requalifying for the CE mark to allow commercialization in the European Union and to conduct clinical trials to support regulatory authorization to commercialize EndoBarrier in India and the United States.
Cost of Revenue. Cost of revenue decreased to $0 for the year ended December 31, 2018 compared to $265 thousand for the year ended December 31, 2017. The decrease in cost of revenue is related to the CE Mark withdrawal.
Our gross loss decreased to $0 for the year ended December 31, 2018 compared to $212 thousand for the year ended December 31, 2017 for the reasons described above. Until we regain commercial approvals and gain reimbursement, our gross margin or gross loss will vary, based on reimbursement levels and ongoing efforts to develop a secure and efficient contract manufacturing process.
Operating Expenses
|
| Years Ended |
|
|
|
|
|
|
|
|
| |||||
| December 31, |
|
| Change |
| |||||||||||
|
| 2018 |
|
| 2017 |
|
| $ |
|
| % |
| ||||
|
| (dollars in thousands) |
|
|
|
|
| |||||||||
Research and development expense |
| $ | 1,947 |
|
| $ | 3,560 |
|
| $ | (1,613 | ) |
|
| (45.3 | )% |
Sales and marketing expense |
|
| 866 |
|
|
| 1,811 |
|
|
| (945 | ) |
|
| (52.2 | )% |
General and administrative expense |
|
| 4,809 |
|
|
| 4,951 |
|
|
| (142 | ) |
|
| (2.9 | )% |
Total operating expenses |
| $ | 7,622 |
|
| $ | 10,322 |
|
| $ | (2,700 | ) |
|
| (26.2 | )% |
Research and Development Expense. The decrease in research and development expense of approximately $1.6 million for the year ended December 31, 2018 compared to the year ended December 31, 2017 was primarily due to a decrease of approximately $1.3 million in quality assurance and medical monitoring consulting fees due to the Company’s CE Mark withdrawal. Other decreases were approximately $148 thousand in third-party manufacturing costs, also due to CE Mark withdrawal, and approximately $147 thousand in employee and related expenses mostly due to the departure of the Company’s Chief Compliance Officer.
Though we expect to implement a more efficient cost structure in order to extend our cash runway, it may be partially offset by the costs associated with rebuilding the research and development management team, improving our quality and regulatory systems and relationships and finalizing the protocol for our U.S. investigational device exemption trial in anticipation of initiating and beginning enrollment of the trial in 2019 in the event such trial receives regulatory approval.
Sales and Marketing Expense. The decrease in sales and marketing expense of approximately $1.0 million for the year ended December 31, 2018 compared to the year ended December 31, 2017 was primarily the result of a decrease of approximately $468 thousand in compensation and employee related expenses related to our decision to reduce international headcount during the year ended December 31, 2017, as well as a decrease of approximately $360 thousand in consulting expenses and $133 thousand in miscellaneous sales and marketing related activities.
General and Administrative Expense. The decrease in general and administrative expense of approximately $142 thousand for the year ended December 31, 2018 compared to the year ended December 31, 2017 was primarily the result of a decrease of approximately $214 thousand in financial consulting and $171 thousand in patent-related attorney costs, offset by an increase in compensation and employee-related expenses of $256 thousand.
We continue to look for ways to streamline our general and administrative expenses. In so doing, we recognize that some costs, particularly those associated with being a public company with multiple reporting jurisdictions are unavoidable. We will continue to provide general and administrative support for scope and size of the entire company.
39
|
| Years Ended |
|
|
|
|
|
|
|
|
| |||||
| December 31, |
|
| Change |
| |||||||||||
|
| 2018 |
|
| 2017 |
|
| $ |
|
| % |
| ||||
|
| (dollars in thousands) |
|
|
|
|
| |||||||||
Other income (expense): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
| $ | 29 |
|
| $ | 33 |
|
| $ | (4 | ) |
|
| (12.1 | )% |
Interest expense |
|
| (1,151 | ) |
|
| (321 | ) |
|
| (830 | ) |
|
| 258.6 | % |
Foreign exchange gain (loss) |
|
| 13 |
|
|
| (10 | ) |
|
| 23 |
|
|
| (230.0 | )% |
Income (expense) from diverted funds and insurance claim |
|
| 224 |
|
|
| (170 | ) |
|
| 394 |
|
|
| (231.8 | )% |
Re-measurement of derivative liabilities |
|
| 482 |
|
|
| 126 |
|
|
| 356 |
|
|
| 282.5 | % |
Total other income (expense), net |
| $ | (403 | ) |
| $ | (342 | ) |
| $ | (61 | ) |
|
| 17.8 | % |
Interest income. The decrease in interest income of approximately $4 thousand for the year ended December 31, 2018 compared to the year ended December 31, 2017 was due to lower average cash and cash equivalents balances in 2017.
Interest expense. Interest expense increased by approximately $830 thousand for the year ended December 31, 2018 compared to the year ended December 31, 2017 primarily due to non-cash interest expenses related to the Company’s 2017 Convertible Note Financing and the 2018 Convertible Note and Warrant Financing.
Income (expense) from diverted funds and insurance claim. In July 2018, after a third-party investigation isolated the activity, an insurance claim was filed for $271 thousand for fraudulent diversion of cash from the Company’s account into a personal account that occurred during the years 2016 through 2018. In 2017, an entry aggregated and reclassified the fraudulent expenses totaling $170 thousand from operating expenses to the other income account entitled income (expense) from diverted funds and insurance claim. Expense in the year ended December 31, 2017 represents fraudulent activity comprised of $37 thousand of research and development, $10 thousand of sales and marketing and $123 thousand of general and administrative expense and income in the year ended December 31, 2018 represents net insurance proceeds on the claim less $22 thousand of research and development expense. The Company has since implemented internal controls to correct the identified control deficiencies associated with the fraud.
Foreign exchange gain (loss). The change to a foreign exchange gain of approximately $13 thousand for the year ended December 31, 2018 from a loss of approximately $10 thousand for the year ended December 31, 2017 is primarily due to lower average foreign currency balances in the year ended December 31, 2018 compared to the year ended December 31, 2017 and changes in valuation of foreign exchange rates.
Re-measurement of derivative liabilities. The change in the re-measurement of derivative liabilities of approximately $356 thousand for the year ended December 31, 2018 compared to the year ended December 31, 2017 was the result of a decrease in the fair value of the conversion rights in the 2017 Note and the revaluation of the consulting warrants issued in 2016.
Liquidity and Capital Resources
As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. We are currently focused primarily on our pivotal trial, which will support future regulatory submissions and potential commercialization activities. Until we are successful in gaining regulatory approvals, we are unable to sell our product in any market at this time. Without revenues, we are reliant on funding obtained from investment in our Company to maintain our business operations until we can generate positive cash flows from operations. We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.3 million. GI Dynamics expects to incur significant operating losses for the next several years. At December 31, 2018 the Company had approximately $3.8 million in cash and cash equivalents.
40
The Company will need to raise additional capital and restructure the terms of the 2017 Note before March 31, 2019 in order to continue to pursue its current business objectives as planned and to continue to fund its operations. The Company is looking to raise additional funds through any combination of additional equity and debt financings or from other sources. However, the Company has no guarantee that the 2017 Note will not mature on March 31, 2019 and has no guaranteed source of capital that will sustain operations into the second quarter of 2019. There can be no assurance that any such potential financing opportunities will be available on acceptable terms, if at all. If the Company is unable to raise sufficient capital on the Company’s required timelines and on acceptable terms to shareholders and the Board of Directors, it could be forced to cease operations, including activities essential to support regulatory applications to commercialize EndoBarrier. If access to capital is not achieved in the near term, it will materially harm the Company’s business, financial condition and results of operations to the extent that the Company may be required to cease operations altogether, file for bankruptcy, or undertake any combination of the foregoing. In such event, our shareholders may lose their entire investment in our company.
These factors raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
In January 2017, the Company completed the issue of 12,481,600 CDI’s (249,632 shares) to eligible investors under a Security Purchase Plan for approximately $0.83 per share (A$0.022 per CDI) resulting in net proceeds after issuance costs of approximately $0.2 million.
In June 2017, the Company completed a Convertible Term Promissory Note (the “2017 Note”) secured financing with its largest shareholder Crystal Amber for a gross amount of $5.0 million that accrues interest at 5% per annum compounded annually. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. The Note was due by December 31, 2018 and contains provisions for conversion during its term and is also subject to security arrangements in favor of Crystal Amber (See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
In January and March 2018, the Company raised approximately $1.6 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom.
In May 2018, the Company completed the 2018 Note and 2018 Warrant financing with its largest shareholder Crystal Amber for a gross amount of $1.75 million that accrues interest at 10% per annum compounded annually. The Note and Warrant financing was approved in a vote of the shareholders of the Company during the Annual Meeting of Stockholders held on May 24, 2018. The 2018 Note matures and the 2018 Warrant expires on May 30, 2023. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. (See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
In September 2018, the Company received commitments for a private placement of approximately $5 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom. The first tranche of $2.2 million closed and cash was received in September 2018. The second and final tranche of $2.8 million was contingent upon shareholder approval which was received in October 2018. Cash proceeds were received in November 2018.
In December 2018, the maturity of the 2017 Note was extended to March 31, 2019 in exchange for payment of $394 thousand which was the total accrued interest on the 2017 Note at December 31, 2018.
During the year ended December 31, 2018, our cash, cash equivalents and restricted cash balance increased by approximately $0.8 million primarily due to various financings that exceeded our cash payments related to, among other things, research and development and general and administrative expenses as we continued to focus on clinical and regulatory strategies.
41
The following table sets forth the major sources and uses of cash for each of the periods set forth below:
| Years Ended December 31, |
| ||||||
|
| 2018 |
|
| 2017 |
| ||
|
| (in thousands) |
| |||||
Net cash (used in) provided by: |
|
|
|
|
|
|
|
|
Operating activities |
| $ | (7,324 | ) |
| $ | (10,123 | ) |
Investing activities |
|
| (3 | ) |
|
| (5 | ) |
Financing activities |
|
| 8,099 |
|
|
| 4,869 |
|
Net increase (decrease) in cash, cash equivalents and restricted cash |
| $ | 772 |
|
| $ | (5,259 | ) |
Cash Flows used in Operating Activities
Net cash used in operating activities totaled approximately $7.3 million for the year ended December 31, 2018. The primary uses of cash were:
| • | to fund our net loss of approximately $8.0 million; |
| • | a net negative adjustment to cash flow from changes in working capital of approximately $0.1 million resulting primarily from increases in accrued expenses and accounts receivable, partially offset by a decrease in accounts payable and an increase in prepaid expenses; and |
| • | a net positive adjustment to cash flow from non-cash items of approximately $0.8 million, primarily from non-cash interest expense, change in derivative valuation and debt accretion of $0.7 million and $0.1 million of stock-based compensation. |
Net cash used in operating activities totaled approximately $10.1 million for the year ended December 31, 2017. The primary uses of cash were:
| • | to fund our net loss of approximately $10.9 million; |
| • | a net positive adjustment to cash flow from changes in working capital of approximately $0.4 million resulting primarily from decreases in inventory and prepaid expenses and an increase in accounts payable, partially offset by a decrease in accrued expenses and other current liabilities; and |
| • | a net positive adjustment to cash flow from non-cash items of approximately $0.4 million, primarily from elimination of inventory reserves, stock-based compensation expense and depreciation and amortization of property and equipment and debt issuance costs. |
Cash Flows used in Investing Activities
Cash used in investing activities for the year ended December 31, 2018 totaled approximately $3 thousand from the purchase of office equipment.
Cash used in investing activities for the year ended December 31, 2017 totaled approximately $5 thousand from the purchase of office equipment.
Cash Flows provided by Financing Activities
Cash provided by financing activities for the year ended December 31, 2018 totaled approximately $8.0 million and primarily resulted from the net proceeds from our private placement of CDIs.
Cash provided by financing activities for the year ended December 31, 2017 totaled approximately $4.9 million and primarily resulted from the net proceeds from our private placement of CDIs and proceeds from the 2017 Note.
42
As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. We are currently focused primarily on our pivotal trial, which will support future regulatory submissions and potential commercialization activities. Until we are successful in gaining regulatory approvals, we are unable to sell our product in any market at this time. Without revenues, we are reliant on funding obtained from investment in the Company to maintain our business operations until we can generate positive cash flows from operations. We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.3 million. GI Dynamics expects to incur significant operating losses for the next several years. At December 31, 2018 the Company had approximately $3.8 million in cash and cash equivalents.
The Company will need to raise additional capital and restructure the terms of the 2017 Note before March 31, 2019 in order to continue to pursue its current business objectives as planned and to continue to fund its operations. The Company is looking to raise additional funds through any combination of additional equity and debt financings or from other sources. However, the Company has no guarantee that the 2017 Note will not mature on March 31, 2019 and has no guaranteed source of capital that will sustain operations into the second quarter of 2019. There can be no assurance that any such potential financing opportunities will be available on acceptable terms, if at all. If the Company is unable to raise sufficient capital on the Company’s required timelines and on acceptable terms to shareholders and the Board of Directors, it could be forced to cease operations, including activities essential to support regulatory applications to commercialize EndoBarrier. If access to capital is not achieved in the near term, it will materially harm the Company’s business, financial condition and results of operations to the extent that the Company may be required to cease operations altogether, file for bankruptcy, or undertake any combination of the foregoing. In such event, our shareholders may lose their entire investment in our company.
These factors raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
Our forecast of the period of time through which our financial resources will be adequate to support our operations are forward-looking statements and involve risks and uncertainties, and actual results could vary materially and negatively as a result of a number of factors, including the factors discussed in the “Risk Factors” section of this Annual Report on Form 10-K. We have based these estimates on assumptions that may prove to be wrong, and we could utilize our available capital resources sooner than we currently expect.
Due to the numerous risks and uncertainties associated with securing regulatory approval for EndoBarrier, at this time we are unable to estimate precisely the amounts of capital outlays and operating expenditures necessary to complete the task of obtaining regulatory approval for EndoBarrier. Our funding requirements will depend on many factors, including, but not limited to, the following:
| • | the rate of progress and cost of our commercialization activities after regulatory approval; |
| • | the expenses we may incur in marketing and selling EndoBarrier subject to future regulatory approvals; |
| • | the timing and decisions of payer organizations related to reimbursement; |
| • | the revenue generated by sales of EndoBarrier; |
| • | the safety and efficacy of our product in addressing diabetes and obesity; |
| • | the success of our investment in our manufacturing and supply chain infrastructure; |
| • | the time and costs involved in obtaining regulatory approvals for EndoBarrier in new markets; |
| • | the success of our research and development efforts; |
| • | the costs associated with any additional clinical trial(s) required in the U.S. and other countries on a case by case basis; |
43
| • | the emergence of competing or complementary developments; and |
| • | the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights. |
We will continue to manage our capital structure and to consider all financing opportunities, whenever they may occur, that could strengthen our long-term liquidity profile. Any such capital transactions may or may not be similar to transactions in which we have engaged in the past and the ownership interests of our existing stockholders may be materially diluted.
Off–Balance Sheet Arrangements
We do not have any relationships with unconsolidated entities or financial partnerships, such as entities often referred to as structured finance or special purpose entities, that would have been established for the purpose of facilitating off-balance sheet arrangements (as that term is defined in Item 303(a)(4)(ii) of Regulation S-K) or other contractually narrow or limited purposes. As such, we are not exposed to any financing, liquidity, market or credit risk that could arise if we had engaged in those types of relationships. We enter into guarantees in the ordinary course of business related to the guarantee of our own performance and the performance of our subsidiaries.
Contractual Obligations and Commitments
Our commitments for operating leases relate to our lease of office space in Boston, Massachusetts which expire in April 2019.
Recent Accounting Pronouncements
For a discussion of recent accounting pronouncements please refer to Note 2, “Summary of Significant Accounting Policies and Basis of Presentation”, to our consolidated financial statements included in this Annual Report on Form 10-K.
44
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are focused on developing clinical data and regulatory approvals for EndoBarrier and our earnings and cash flows are exposed to market risk from changes in currency exchange rates and interest rates.
Interest Rate Sensitivity
Our cash and cash equivalents of $3.8 million at December 31, 2018 consisted of cash and money market funds, all of which will be used for working capital purposes. We do not enter into investments for trading or speculative purposes. The goals of our investment policy are preservation of capital, fulfillment of liquidity needs and fiduciary control of cash and investments. We also seek to maximize income from our investments without assuming significant risk. Our primary exposure to market risk is interest income sensitivity, which is affected by changes in the general level of interest rates in the U.S. and Australia. Because of the short-term nature of our cash and cash equivalents, we do not believe that we have any material exposure to changes in their fair values as a result of changes in interest rates. The continuation of historically low interest rates in the U.S. will limit our earnings on investments held in U.S. dollars.
Foreign Currency Risk
We conduct business in foreign countries. For U.S. reporting purposes, we remeasure all assets and liabilities of our non-U.S. entities at the period-end exchange rate and revenue and expenses at the average exchange rates in effect during the periods. The net effect of these remeasurement adjustments is shown in the accompanying consolidated financial statements as a component of net loss.
We have generated a significant portion of our revenue and collected receivables in foreign currencies. Fluctuations in the exchange rate of the U.S. dollar against major foreign currencies, including the euro, British Pound and Australian dollar, can result in foreign currency exchange gains and losses that may significantly impact our financial results. These foreign currency transactions and remeasurement gains and losses are presented as a separate line item on our consolidated statements of operations. Continued fluctuation of these exchange rates could result in financial results that are not comparable from quarter to quarter. We do not currently utilize foreign currency contracts to mitigate the gains and losses generated by the remeasurement of non-functional currency assets and liabilities but do hold cash reserves in currencies in which those reserves are anticipated to be expended.
All proceeds from our equity offerings were denominated in Australian dollars and as of December 31, 2018 we held the total equivalent of approximately U.S. $12 thousand denominated in Australian dollars and euros. Accordingly, we have had and will continue to have exposure to foreign currency exchange rate fluctuations. A change of 10% or more in foreign currency exchange rates of the Australian dollar or the euro would not have a material impact on our financial position and results of operations.
Effects of Inflation
We do not believe that inflation and changing prices over the years ended December 31, 2018 and 2017 had a significant impact on our results of operations.
ITEM 8. CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
Our consolidated financial statements, together with the independent registered public accounting firm report thereon, appear on pages F-1 through F-32 of this Annual Report on Form 10-K.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
45
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
As required by Rule 13a-15(b) of the Securities Exchange Act of 1934, or the Exchange Act, our management, including our principal executive officer and our principal financial and accounting officer, conducted an evaluation as of the end of the period covered by this Annual Report on Form 10-K of the effectiveness of the design and operation of our disclosure controls and procedures. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms, and such information required to be disclosed by us in the reports we file under the Exchange Act is accumulated and communicated to our management, including our principal executive officer and principal financial and accounting officer, as appropriate to allow timely decisions regarding required disclosure. After conducting the evaluation, our principal officers have concluded that our disclosure controls and procedures were not effective at the reasonable assurance level due to the material weaknesses in our internal control over financial reporting further described below. We believe that the control issues did not have a material impact on the financial statements.
Material Weakness in Internal Control Over Financial Reporting
A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim consolidated financial statements will not be prevented or detected and corrected on a timely basis. In connection with the preparation of our annual financial statements for the year ended December 31, 2018, we identified the following material weaknesses in our internal control over financial reporting:
| • | Failure to adequately review transactions before posting to general ledger; and |
| • | Lack of effective controls to adequately restrict access and segregate duties. Specifically, due to the limited number of staff in our accounting function; |
| o | Reconciliations, trial balances, and financial statements were prepared and reviewed by the principal financial and accounting officer. |
| o | The principal financial and accounting officer had administrative rights to the financial reporting system. |
| o | The principal financial and accounting officer was able to generate, print and sign checks up to a certain threshold, record the payments, and reconcile the main bank account. |
| o | Lack of requisite accounting knowledge and inadequate written policies and procedures surrounding complex debt and equity arrangements with respect to both GAAP and the guidelines of the SEC. |
| o | Lack of authorization restrictions within our bank portal to prevent manual edits to wire payment templates. |
| o | Ineffective reporting of payables to detect duplicate payment entries. |
Upon identifying these material weaknesses, we performed additional procedures to evaluate the impact on the financial statements.
Based on these procedures, we believe the material weaknesses did not result in any material misstatements to our financial statements and we believe the consolidated financial statements included in this Annual Report present, in all material respects, our financial position, results of operations and cash flows for the periods presented in conformity with U.S. generally accepted accounting principles. However, these material weaknesses could result in a misstatement of our accounts or disclosures that would result in a material misstatement of our financial statements that would not be prevented or detected.
46
We intend to remedy our material weakness with regard to insufficient segregation of duties by hiring additional employees in order to segregate duties in a manner that establishes effective internal controls once resources become available. Until we have processes that have been tested and shown to remediate the material weaknesses across the entire reporting period, we will continue to state a condition of material weakness.
We have partially addressed the identified material weaknesses through the implementation of the following:
| • | In September 2018, the Company hired a part-time consultant who is a seasoned medical technology Chief Financial Officer to advise the Chief Executive Officer and manage the finance team. In December 2018, the consultant was appointed Chief Financial Officer and Corporate Secretary; |
| • | The availability of another senior finance member has allowed segregation of duties so that a single individual is no longer preparing, approving, disbursing, and/or reconciling any transaction; |
| • | Elimination of non-template electronic payment initiation, editing by initiator, and initiation of two-party review of new recipient entries, including bank information; |
| • | Management reporting reformatting to highlight invoice and payment duplications and unusual payables, including recent activity reporting by vendor; and |
| • | Increase internal communications to improve security awareness and to emphasize the importance of exercising professional skepticism. |
Changes in Internal Control
As required by Rule 13a-15(d) of the Exchange Act, our management, including our principal executive officer and our principal financial and accounting officer, conducted an evaluation of the internal control over financial reporting to determine whether any changes occurred during the year ended December 31, 2018 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting. Based on that evaluation, our principal executive officer and principal financial and accounting officer concluded that changes in internal control outlined above could materially affect our internal control over financial reporting.
Inherent Limitations on Controls and Procedures
Our management, including our principal executive officer and principal financial officer, does not expect that our disclosure controls and procedures or our internal control over financial reporting will prevent all error and all fraud. A control system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, have been detected. Thus, misstatements due to error or fraud may occur and not be detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of controls.
None.
47
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
The other information required by this Item 10 related to our directors is incorporated by reference to the applicable information in our proxy statement for our 2018 Annual Meeting of Stockholders to be filed with the SEC.
Information regarding our executive officers required by this Item 10 is incorporated by reference to the applicable information in our proxy statement for our 2018 Annual Meeting of Stockholders to be filed with the SEC.
Code of Business Conduct and Ethics
We have adopted a code of business conduct and ethics applicable to our directors, executive officers and all other employees. A copy of that code is available on our corporate website at http://www.gidynamics.com. Any amendments to the code of business conduct and ethics, and any waivers thereto involving our executive officers, also will be available on our corporate website. A printed copy of these documents will be made available upon request. The content on our website is not incorporated by reference into this Annual Report on Form 10-K.
Directors of the Registrant
The following table sets forth the name, age and position of each of our directors as of February 15, 2019:
| Age |
| Position | |
Daniel J. Moore (3) |
| 57 |
| Non-executive Chairman of the Board |
Timothy J. Barberich (1)(2) |
| 71 |
| Non-executive Director |
Juliet Thompson (1) |
| 52 |
| Non-executive Director |
Oern R. Stuge, M.D. (2)(3) |
| 64 |
| Non-executive Director |
1. | Member of the audit committee. |
2. | Member of compensation committee. |
3. | Member of nominating and corporate governance committee. |
Daniel J. Moore has served as a director of the Company since 2014, as our vice-chairman from March 2015 to April 2016 and our chairman since May 2016. Mr. Moore’s extensive experience in domestic and international sales, operations and executive management in global medical device manufacturers and years of service on other boards makes him qualified to serve on our board of directors.
Mr. Moore has served as president, chief executive officer and director of Cyberonics, Inc., a medical technology company with core expertise in neuromodulation, from 2007 to October 2015. From 1989 to 2007, Mr. Moore held positions in sales, marketing, and senior management in the U.S. and in Europe at Boston Scientific Corporation, a diverse maker of minimally invasive medical products. His last position at Boston Scientific was President, International Distributor Management. Prior to that role, he held the position of President, Inter-Continental, the fourth largest business unit of Boston Scientific, with more than 1,000 global employees and revenues exceeding $700 million. Mr. Moore previously held senior management positions at several Boston Scientific U.S. and international divisions.
Mr. Moore currently serves as the chairman of LivaNova PLC (the company resulting from the merger of Sorin S.p.A. and Cyberonics, Inc.), chairman of ViewRay, a member of the board of directors for the Epilepsy Foundation of America, and as a member of the boards or advisory boards for BioHouston, Inc. and the Weldon School of Biomedical Engineering at Purdue University. He currently serves on the board of privately-held BrainScope Company, Inc., a medical technology company focused on traumatic brain injury, where he serves as Chairman. Past board positions include Smiling Kids, Inc., the Epilepsy Foundation of Texas (past-Chair), the Epilepsy Foundation of Texas — Houston (past-President), the Medical Device Manufacturers Association (past-Chair), Cyberonics, Inc., Topera, Inc. (acquired by Abbott) and TriVascular Technologies, Inc. (acquired by Endologix).
48
Mr. Moore holds a B.A. from Harvard University and earned an MBA from Boston University.
Timothy J. Barberich has been a director of the Company since June 2011. Mr. Barberich has nearly 40 years’ experience in pharmaceutical and medical device companies, in technical, sales, marketing and management positions, including as chief executive officer and chairman of the board. Mr. Barberich is the founder and former president, chief executive officer and chairman of Sepracor, Inc., a NASDAQ-listed-pharmaceutical company based in Massachusetts, which was acquired by Dainippon Sumitomo Pharma Co., Ltd. in 2009. Mr. Barberich founded Sepracor in 1984 and served as its chief executive officer from 1984 to 2007 and chairman of the board from 1990 to 2007. From 2007 to 2008, Mr. Barberich served as executive chairman of Sepracor and then chairman of the board from 2008 to 2009. Mr. Barberich led Sepracor through its early-stage research and development, product approvals, commercialization, private financings and initial public offering, partnerships with major companies, several successful spin-outs and achievement of revenues in excess of $1 billion. Through his work at Sepracor, Mr. Barberich brings to our board invaluable knowledge and experience of leading a company in the health care industry through every stage of its life cycle. Prior to founding Sepracor, Mr. Barberich spent 10 years as a senior executive at Millipore Corporation, a company that provides separations products to the life science research, pharmaceutical, biotechnology and electronic markets. Mr. Barberich brings to our board the knowledge and experience of leading a company in the health care industry through every stage of its life cycle. We believe this experience and familiarity with the types of risks we may face, together with his broad medical device and pharmaceutical industry experience, makes Mr. Barberich uniquely suited to serve on our board.
Mr. Barberich is currently chairman and CEO of BioNevia Pharmaceuticals, Inc., a director of Verastem Ocology, a NASDAQ-listed biotechnology company, TScan and Frequency Theraputics, both privately funded biotechnology companies. Mr. Barberich was formerly a director of Inotek Pharmaceuticals, a NASDAQ-listed biotechnology company, Neurovance, Inc., a privately held biotechnology company, HeartWare International, Inc., a NASDAQ-listed medical device company, Tokai Pharmaceuticals, Inc., a NASDAQ-listed biopharmaceutical company, MirImmune Inc., which was acquired in 2016, BioSphere Medical, Inc., a NASDAQ-listed biotechnology company, Gemin X Biotechnologies, Inc. and Resolvyx Pharmaceuticals, which were acquired in 2011 and 2010, respectively, and Virucon, a publicly traded biotechnology company.
Mr. Barberich holds a Bachelor of Science degree in Chemistry from Kings College in Pennsylvania and has taken graduate courses from the School of Chemistry at Rutgers University.
Juliet Thompson has been a director of the Company since August 22, 2017. Ms. Thompson also assumed the role of chair of the Company’s audit committee. Ms. Thompson has spent approximately 20 years working as an investment banker and strategic advisor to healthcare companies in Europe. She has a strong track record of advising companies on corporate strategy across numerous transactions.
Since March 2015, Ms. Thompson has served on the board of Nexstim Limited, a medical technology company listed on Nasdaq First North Finland and Sweden. Prior to that, Ms. Thompson led the European healthcare practice at Stifel Financial Corp., a diversified financial services holding company, serving as a partner from October 2013 to April 2015. In 2003, Ms. Thompson co-founded Code Securities, a healthcare investment banking firm that was purchased by Nomura and renamed Nomura Code Securities Limited (“Nomura Code”) in 2005, and served as Head of Corporate Finance and as a member of the board of Nomura Code until 2013. She is also currently a non-executive director of Vectura PLC, a company listed on the London Stock Exchange plc, and Novacyt S.A., a French-based company whose shares are admitted to trade on AIM, Ms. Thompson is a member of the Institute of Chartered Accountants in England and Wales (ACA) and holds a BSc degree in Economics from the University of Bristol. Her experience also includes roles at WestLB Panmure, ICI PLC, Deloitte and Touche and HM Treasury.
Oern R. Stuge, M.D. has served as a director of the Company since his appointment in January 2017. Dr. Stuge’s extensive experience in domestic and international sales, management and operations in a global medical device manufacturer makes him qualified to serve on our board of directors.
Dr. Stuge has served as an executive in various medical device, health care and life sciences companies over the last thirty years. Since January 2011, Dr. Stuge has been Chairman of Orsco Lifesciences AG, a management firm that specializes in medical technology through which he supports several companies. Prior to that, Dr. Stuge served in various positions, including as Senior Vice-President, at Medtronic Inc., from May 1998 to December 2009. Dr. Stuge is currently Chairman of Mainstay Medical Limited, a Euronext Paris-listed and Irish Stock Exchange-listed medical devices company and Luminas Limited, formerly a NASDAQ-listed medical company. Dr. Stuge is chairman of the board of OrthoD Ltd.
49
and serves on the board of several private companies including Balt Extrusion SAS, Pulmonx International SA, and Phagenesis Limited. Furthermore, until December 2016, Dr. Stuge served on the board of Bonesupport AB, a private medical technology company.
Dr. Stuge received an M.D. from the University of Oslo, Norway, an M.B.A. from IMD and an INSEAD Certification in Corporate Governance.
Australian Disclosure Requirements
Because we are listed on the Australian Securities Exchange, or ASX, we are required to comply with various disclosure requirements as set out in the ASX Listing Rules. The following information is provided to comply with the ASX Listing Rules and is not intended to fulfill SEC information required by Part III of this Annual Report on Form 10-K.
Overview
Our securities are listed for quotation in the form of CHESS Depositary Interests, or CDIs, on the ASX and trade under the symbol “GID.” Each share of our Common Stock is equivalent to fifty CDIs. The shareholder information below was applicable as at January 11, 2019.
Our share capital was as follows:
| Number of Securities |
|
| Equivalent in CDIs |
| |||
Common Stock |
|
| 97,809 |
|
|
| 4,890,450 |
|
CDIs |
|
| 958,986,849 |
|
|
| 958,986,849 |
|
Total |
|
|
|
|
|
| 963,877,299 |
|
|
|
|
|
|
|
|
|
|
Options1 |
|
| 1,127,883 |
|
|
| 56,394,150 |
|
Restricted stock units1 |
|
| 250,000 |
|
|
| 12,500,000 |
|
Warrants |
|
| 1,972,976 |
|
|
| 98,648,800 |
|
Total |
|
|
|
|
|
| 1,131,420,249 |
|
1. | As of December 31, 2018, an additional 1,408,812 shares of Common Stock were available for grant under our 2011 Employee, Director and Consultant Equity Incentive Plan. |
Substantial Holders
The number of CDIs held by our substantial shareholders (being shareholders who, together with their associates, have a relevant interest in at least 5% of our voting shares) assuming the conversion of Common Stock held by those shareholders into CDIs and based on the information in the substantial holder notices we received as of January 11, 2019, was as follows:
| Number of CDIs Held |
|
| % of Total CDIs |
| |||
Crystal Amber Fund Limited |
|
| 466,935,612 |
|
|
| 48.44 | % |
Richard Cashin |
|
| 199,364,713 |
|
|
| 20.68 | % |
50
Distribution of Equity Security Holders
There was a total of 19,277,546 shares of Common Stock on issue, 19,179,737 of which were held as CDIs (being 958,986,849 CDIs in total). The table below presents the number of shares of Common Stock and the number of CDIs on issue, as well as the number of options, restricted stock units and warrants on issue by size of holding as of January 11, 2019:
| Common Stock (unlisted) |
|
| CDIs |
|
| Options (unlisted) |
|
| Restricted Stock Units (unlisted) |
|
| Warrants (unlisted) |
| ||||||||||||||||||||||||||
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
|
| Number |
| ||||||||||
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
|
| of |
| ||||||||||
|
| Holders |
|
| Shares |
|
| Holders |
|
| Shares |
|
| Holders |
|
| Shares |
|
| Holders |
|
| Shares |
|
| Holders |
|
| Shares |
| ||||||||||
1 – 1,000 |
| — |
|
| — |
|
|
| 127 |
|
|
| 43,982 |
|
|
| 1 |
|
|
| 196 |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||||
1,001 – 5,000 |
|
| 1 |
|
|
| 28 |
|
|
| 222 |
|
|
| 620,435 |
|
|
| 2 |
|
|
| 6,920 |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||
5,001 – 10,000 |
|
| 1 |
|
|
| 105 |
|
|
| 128 |
|
|
| 1,083,068 |
|
|
| 1 |
|
|
| 5,500 |
|
| — |
|
| — |
|
| — |
|
| — |
| ||||
10,001 – 100,000 |
|
| 9 |
|
|
| 8,526 |
|
|
| 260 |
|
|
| 9,710,138 |
|
|
| 10 |
|
|
| 394,667 |
|
| — |
|
| — |
|
|
| 1 |
|
|
| 28,532 |
| ||
100,001 – and over |
|
| 8 |
|
|
| 89,150 |
|
|
| 126 |
|
|
| 947,529,226 |
|
|
| 1 |
|
|
| 720,600 |
|
|
| 1 |
|
|
| 250,000 |
|
|
| 1 |
|
|
| 1,944,444 |
|
Total |
|
| 19 |
|
|
| 97,809 |
|
|
| 863 |
|
|
| 958,986,849 |
|
|
| 15 |
|
|
| 1,127,883 |
|
|
| 1 |
|
|
| 250,000 |
|
|
| 2 |
|
|
| 1,972,976 |
|
Unmarketable Parcels
As of January 11, 2019, the number of shareholders holding less than a marketable parcel (for the purposes of the ASX Listing Rules) was 615, based on the closing market price as of January 11, 2019.
Top 20 Holders
Holders of CDIs Only
The table below provides a list of the top 20 holders of our CDIs. Related but separate legal entities are not aggregated.
| Name of Holder |
| Number of CDIs Held |
|
| % of Total CDIs |
| |||
1 |
| Crystal Amber Advisers |
|
| 466,935,612 |
|
|
| 48.44 | % |
2 |
| Mr Richard Cashin |
|
| 199,364,713 |
|
|
| 20.68 | % |
3 |
| Medtronic |
|
| 39,115,442 |
|
|
| 4.06 | % |
4 |
| Mr Paul Cozzi |
|
| 35,493,578 |
|
|
| 3.68 | % |
5 |
| Advanced Technology Ventures |
|
| 33,172,769 |
|
|
| 3.44 | % |
6 |
| Johnson & Johnson Development Corp |
|
| 28,278,460 |
|
|
| 2.93 | % |
7 |
| Polaris Venture Partners |
|
| 18,516,181 |
|
|
| 1.92 | % |
8 |
| Mr David Brock |
|
| 10,000,000 |
|
|
| 1.04 | % |
9 |
| Mr & Mrs Ian Moore |
|
| 7,210,420 |
|
|
| 0.75 | % |
10 |
| Fidelity Mgt & Research |
|
| 5,561,290 |
|
|
| 0.58 | % |
11 |
| Invus |
|
| 5,185,659 |
|
|
| 0.54 | % |
12 |
| Ms Rena C Merchant |
|
| 4,685,000 |
|
|
| 0.49 | % |
13 |
| UBS |
|
| 3,794,165 |
|
|
| 0.39 | % |
14 |
| Integrated Financial Arrangements |
|
| 3,790,771 |
|
|
| 0.39 | % |
15 |
| Mr Mark B Konda |
|
| 3,437,363 |
|
|
| 0.36 | % |
16 |
| Mr Timothy J Barberich |
|
| 3,103,705 |
|
|
| 0.32 | % |
17 |
| E Trade |
|
| 2,889,188 |
|
|
| 0.30 | % |
18 |
| Private Clients of UBS AG Hong Kong |
|
| 2,718,601 |
|
|
| 0.28 | % |
19 |
| Mr Charles H Warman |
|
| 2,698,300 |
|
|
| 0.28 | % |
20 |
| Mr & Mrs James Hochroth |
|
| 2,681,800 |
|
|
| 0.28 | % |
Total CDIs held by top 20 CDI holders |
|
| 878,633,017 |
|
|
| 91.16 | % | ||
Total CDIs held by all other CDI holders |
|
| 80,353,832 |
|
|
| 8.84 | % | ||
|
| TOTAL |
|
| 958,986,849 |
|
|
|
|
|
51
Holders of CDIs and Common Stock Combined
The table below provides a list of the top 20 holders of our securities taking into account securities held in the form of both Common Stock and CDIs. Information presented below is prepared on the assumption that all shares of Common Stock on issue are held as CDIs. Related but separate legal entities are not aggregated.
Details of Shareholders if all shares of Common Stock on issue are held as CDIs
| Name of Holder |
| Number of CDIs Held |
|
| % of Total CDIs |
| |||
1 |
| Crystal Amber Advisers |
|
| 466,935,612 |
|
|
| 48.44 | % |
2 |
| Mr Richard Cashin |
|
| 199,364,713 |
|
|
| 20.68 | % |
3 |
| Medtronic |
|
| 39,115,442 |
|
|
| 4.06 | % |
4 |
| Mr Paul Cozzi |
|
| 35,493,578 |
|
|
| 3.68 | % |
5 |
| Advanced Technology Ventures |
|
| 33,172,769 |
|
|
| 3.44 | % |
6 |
| Johnson & Johnson Development Corp |
|
| 28,278,460 |
|
|
| 2.93 | % |
7 |
| Polaris Venture Partners |
|
| 18,516,181 |
|
|
| 1.92 | % |
8 |
| Mr David Brock |
|
| 10,000,000 |
|
|
| 1.04 | % |
9 |
| Mr & Mrs Ian Moore |
|
| 7,210,420 |
|
|
| 0.75 | % |
10 |
| Fidelity Mgt & Research |
|
| 5,561,290 |
|
|
| 0.58 | % |
11 |
| Invus |
|
| 5,185,659 |
|
|
| 0.54 | % |
12 |
| Ms Rena C Merchant |
|
| 4,685,000 |
|
|
| 0.49 | % |
13 |
| UBS |
|
| 3,794,165 |
|
|
| 0.39 | % |
14 |
| Integrated Financial Arrangements |
|
| 3,790,771 |
|
|
| 0.39 | % |
15 |
| Mr Mark B Konda |
|
| 3,437,363 |
|
|
| 0.36 | % |
16 |
| Mr Timothy J Barberich |
|
| 3,103,705 |
|
|
| 0.32 | % |
17 |
| E Trade |
|
| 2,889,188 |
|
|
| 0.30 | % |
18 |
| Private Clients of UBS AG Hong Kong |
|
| 2,718,601 |
|
|
| 0.28 | % |
19 |
| Mr Charles H Warman |
|
| 2,698,300 |
|
|
| 0.28 | % |
20 |
| Mr & Mrs James Hochroth |
|
| 2,681,800 |
|
|
| 0.28 | % |
Total securities held by top 20 securities holders |
|
| 878,633,017 |
|
|
| 91.16 | % | ||
Total securities held by all other securities holders |
|
| 85,244,282 |
|
|
| 8.84 | % | ||
|
|
|
|
| 963,877,299 |
|
|
|
|
|
Options (not listed on ASX)
There were 1,127,883 options on issue to purchase shares of Common Stock under the 2011 Employee, Director and Consultant Equity Incentive Plan and the 2003 Omnibus Stock Plan with varying exercise prices. These options are held by 15 individuals.
Restricted Stock Units (not listed on ASX)
There were 142,659 restricted stock units on issue for 142,659 shares of Common Stock under the 2011 Employee, Director and Consultant Equity Incentive Plan. These restricted stock units are held by 1 individual.
Warrants (not listed on ASX)
There was one warrant on issue to subscribe for in aggregate 28,532 shares of Common Stock at an exercise price of $0.64 per share.
Restricted Securities
There were no restricted securities on issue.
52
Our bylaws provide that each shareholder has one vote for every share of Common Stock entitled to vote held of record by such shareholder and a proportionate vote for each fractional share of Common Stock entitled to vote so held, unless otherwise provided by Delaware General Corporation Law or in the certificate of incorporation.
Holders of CDIs have one vote for every fifty CDIs held of record by such shareholder. If holders of CDIs wish to attend our general meetings, they will be able to do so. Under the ASX Listing Rules, the Company, as an issuer of CDIs, must allow CDI holders to attend any meeting of the holders of the underlying securities unless relevant U.S. law at the time of the meeting prevents CDI holders from attending those meetings.
In order to vote at such meetings, CDI holders have the following options:
| a) | Instructing CDN, as the legal owner, to vote the shares of Common Stock underlying their CDIs in a particular manner. The instruction form must be completed and returned to our share registry prior to the meeting; |
| b) | Informing us that they wish to nominate themselves or another person to be appointed as CDN’s proxy for the purposes of attending and voting at the general meeting; and |
| c) | Converting their CDIs into a holding of shares of Common Stock and voting these at the meeting (however, if thereafter the former CDI holder wishes to sell their investment on the ASX, it would be necessary to convert the shares of Common Stock back to CDIs). This must be done prior to the record date for the meeting. |
As holders of CDIs will not appear on our share register as the legal holders of shares of Common Stock, they will not be entitled to vote at our shareholder meetings unless one of the above steps is undertaken.
Proxy forms and details of these alternatives will be included in each notice of meeting we send to CDI holders.
Holders of restricted stock units, issued but unexercised options and warrants are not entitled to vote.
Required Statements
GI Dynamics, Inc. makes the following disclosures:
| a) | There is no current on-market buy-back of our securities. |
| b) | GI Dynamics, Inc. is incorporated in the state of Delaware in the United States of America. |
| c) | GI Dynamics, Inc. is not subject to Chapters 6, 6A, 6B or 6C of the Corporations Act 2001 (Cth), or Corporations Act, dealing with the acquisitions of shares (including substantial shareholdings and takeovers). |
| d) | Under the Delaware General Corporation Law, shares are generally freely transferable subject to restrictions imposed by U.S. federal or state securities laws, by our certificate of incorporation or bylaws, or by an agreement signed with the holders of the shares at issue. Our amended and restated certificate of incorporation and bylaws do not impose any specific restrictions on transfer. Section 203 of the Delaware General Corporation Law prohibits a publicly-held Delaware corporation from engaging in a “business combination” with an “interested shareholder” for a period of three years following the time the person became an interested shareholder, unless the business combination or acquisition of shares that resulted in a shareholder becoming an interested shareholder is approved in a prescribed manner. A “business combination” can include a merger, asset or share sale, or other transaction resulting in a financial benefit to an interested shareholder. Generally, an interested shareholder is a person who, together with its affiliates and associates, owns (or within three years prior to the determination of interested shareholder status did own) 15% or more of a corporation’s voting shares. The existence of this provision would be expected to have an anti-takeover effect with respect to transactions not approved in advance by the board of directors, including discouraging attempts that might result in a premium over the market price for the shares of Common Stock held by shareholders. As a general matter, Section 203 applies solely to corporations with a class of voting stock listed on a national securities exchange in the U.S. or held of record by 2,000 or more stockholders, neither of which currently apply to us, but may at any time in the future. |
53
| f) | The securities of GI Dynamics, Inc. are not quoted on any exchange other than the ASX. |
| g) | The name of our Corporate Secretary is Charles R. Carter. |
| h) | The address and telephone number of our principal registered office in Australia is: |
KPMG
Tower Three
International Towers Sydney
300 Barangaroo Avenue
Sydney NSW 2000 Australia
Telephone: +61 2 9335 8054
| i) | Registers of securities are held as follows: |
| 1. | for CDIs in Australia at: |
Link Market Services Limited
Level 12, 680 George Street
Sydney NSW 2000
Telephone: +61 1300 554 474
| 2. | for Common Stock in the United States at: |
American Stock Transfer & Trust Company, LLC
6201 15th Avenue
Brooklyn New York 11219
Telephone: +1 718 921 8124
Australian Corporate Governance Statement
The Company’s board of directors, or Board, and employees are committed to developing, promoting and maintaining a strong culture of good corporate governance and ethical conduct.
The Board confirms that the Company’s corporate governance framework is generally consistent with the ASX Corporate Governance Council’s “Corporate Governance Principles and Recommendations 3rd Edition” (“ASX Governance Recommendations”), other than as set out below. To this end, the Company provides below a review of its corporate governance framework using the same numbering as adopted for the principles as set out in the ASX Governance Recommendations.
This corporate governance statement relates to the financial year ended December 31, 2018 and has been approved by the Board.
Copies of the Company’s codes and policies may be downloaded from the corporate governance section of the Company’s website at www.gidynamics.com.
Principle 1 – Lay solid foundations for management and oversight
Recommendation 1.1 – A listed entity should disclose:
| a) | the respective roles and responsibilities of its board and management; and |
| b) | those matters expressly reserved to the board and those delegated to management. |
The Board’s responsibilities are recognized and documented by the charter of the Board (“Board Charter”), a copy of which is available on the Company’s website at www.gidynamics.com, and there is a clear delineation between the Board’s responsibility for the Company’s strategy and activities, and the day-to-day management of operations conferred upon the Company’s officers.
54
The Board Charter provides that the role of the Board, as the body ultimately responsible for the corporate governance of the Company, includes the following major functions:
| • | providing input into and final approval of management’s development of corporate strategy and performance objectives; |
| • | reviewing, ratifying and monitoring systems of risk management and internal control, codes of conduct, and legal compliance; |
| • | ensuring appropriate resources are available to senior executives; |
| • | approving and monitoring the progress of major capital expenditure, capital management and acquisitions and divestments; |
| • | approving and monitoring financial and other reporting; |
| • | evaluating the overall effectiveness of the Board and its committees; and |
| • | evaluating, selecting and recommending an appropriate slate of candidates for election as directors. |
Management is responsible for implementing the strategic objectives set by the Board, carrying out the day-to-day operations of the Company, and making accurate, timely, and clear reports to the Board.
Recommendation 1.2 – A listed entity should:
| a) | undertake the appropriate checks before appointing a person, or putting forward to security holders a candidate for election, as a director; and |
| b) | provide security holders with all material information in its possession relevant to a decision on whether or not to elect or re-elect a director. |
The nominating and corporate governance committee of the Company is responsible for reviewing, with the Board from time to time, the appropriate skills and characteristics required of Board members in the context of the current make-up of the Board and the Company’s business needs. When considering Board appointments, the committee ensures that appropriate checks are undertaken on the candidate’s character, education, qualifications, criminal record and bankruptcy history and that sufficient information is provided to security holders when a candidate is standing for election or re-election as a director to enable them to make an informed decision on whether or not to elect or re-elect the candidate. Information regarding the directors who were re-appointed at the Company’s 2018 annual general meeting was provided in the notice of meeting disclosed to the ASX and shareholders on May 24, 2018.
Recommendation 1.3 – A listed entity should have a written agreement with each director and senior executive setting out the terms of their appointment.
The terms of Board membership are set forth in the Company’s Board Charter and the remuneration paid to Board members is provided in accordance with shareholder approval (where required) following the compensation committee’s recommendation. While the Company does not have a separate written agreement with each of its Board members, it believes these guidelines are adequate to provide a clear understanding of the roles and responsibilities of Board members. In the case of senior executives, the Company has provided a letter or contract of employment to each executive detailing the terms of employment and has developed job descriptions setting forth the position, duties, and reporting structure. Where there are any agreed entitlements upon termination, such agreed items are set forth in the employment letters or contracts.
55
Recommendation 1.4 - The company secretary of a listed entity should be accountable directly to the board, through the chair, on all matters to do with the proper functioning of the board:
The role and responsibilities of the Company’s secretary are set forth in the Company’s bylaws. The Board is responsible for electing or appointing the secretary and for prescribing the duties and powers of the secretary. Each director is able to communicate freely and directly with the secretary and vice versa. The secretary is accountable to the Board, through the chairman of the Board, for all matters to do with the proper functioning of the Board, including:
| • | monitoring the Company’s compliance in respect of all corporate governance matters; |
| • | drafting and circulating the minutes of meetings of the Board and all committees for approval at the next meeting; and |
| • | monitoring the Company’s compliance with all disclosure obligations and regularly reviewing Company policies and procedures relating to compliance with such disclosure obligations. |
Recommendation 1.5 – A listed entity should:
| a) | have a diversity policy which includes requirements for the board or a relevant committee of the board to set measurable objectives for achieving gender diversity and to assess annually both the objectives and the entity’s progress in achieving them; |
| b) | disclose that policy or a summary of it; and |
| c) | disclose as at the end of each reporting period the measurable objectives for achieving gender diversity set by the board or a relevant committee of the board in accordance with the entity’s diversity policy and its progress towards achieving them, and either: |
| i. | the respective proportions of men and women on the board, in senior executive positions and across the whole organization (including how the entity has defined ‘senior executive’ for these purposes); or |
| ii. | if the entity is a “relevant employer” under the Workplace Gender Equality Act, the entity’s most recent “Gender Equality Indicators”, as defined in and published under that Act. |
The Company has adopted a Diversity Policy, a copy of which is available on the corporate governance section of the Company’s website. The Company’s Diversity Policy includes requirements for the Board to establish measurable objectives to assist the Company in achieving diversity.
The Board continued to evaluate the gender diversity of the Company’s employees, its senior executives, and the Board during 2018. During 2018, the Board, in considering measurable objectives to set for achieving gender diversity, concluded that, because of the current stage of the Company’s development and the ongoing restructuring efforts, the Company should continue to recruit employees from a diverse pool of talented candidates without regard to gender while continuing to focus on the necessary skills and experience required to achieve the Company’s performance objectives. As a result, the Company did not set measurable objectives for achieving gender diversity in 2018 but used 2017 data as a baseline to measure gender diversity among its employees, senior executives and Board for 2018.
At December 31, 2018, the proportion of women in the Company as a percentage of its total employees decreased from 54% (7 out of 13 in 2017) to 33% (3 out of 9) based on data maintained by the Company’s human resources organization. In senior executive positions (vice president and above), the proportion of women remained the same at 0% (none out of 2 in 2017 and 2018). The proportion of women on the Board remained the same at 25% (1 out of 4 in 2017 and 2018).
Recommendation 1.6 – A listed entity should:
| a) | have and disclose a process for periodically evaluating the performance of the board, its committees and individual directors; and |
| b) | disclose, in relation to each reporting period, whether a performance evaluation was undertaken in the reporting period in accordance with that process. |
56
In accordance with the Company’s agreed evaluation process, during the reporting period ended December 31, 2018, the Board and each committee performed a self-evaluation. Each director provided their assessments of the effectiveness of the Board and the committees on which they serve to the nominating and corporate governance committee. The individual assessments were summarized by the nominating and corporate governance committee and reported for discussion to the full Board and the committees. The nominating and corporate governance committee completed its assessment of the Board’s compliance with the principles set forth in the Board Charter and did not identify any areas in which the Board or committees needed to improve performance and has reviewed and approved disclosures relating to any departures from the ASX Governance Recommendations. During the reporting period ended December 31, 2018, the nominating and corporate governance committee also evaluated individual directors in accordance with the criteria set by the nominating and corporate governance committee and the Board from time to time. Based on such assessments, the nominating and corporate governance committee has determined that the Board, its committees and each director were effective.
Recommendation 1.7 – A listed entity should:
| a) | have and disclose a process for periodically evaluating the performance of its senior executives; and |
| b) | disclose, in relation to each reporting period, whether a performance evaluation was undertaken in the reporting period in accordance with that process. |
Under the Board Charter, the directors of the Company are ultimately responsible for monitoring the performance of the senior management team and the compensation committee, in accordance with its charter, reviews and approves corporate and personal performance goals and objectives relevant to the compensation of all executive officers. At the end of each calendar year, the chief executive officer presents to the compensation committee his assessment of the performance during the year of each executive officer (other than himself) against pre-established performance objectives. The compensation committee considers this assessment and determines each executive officer’s (including the chief executive officer’s) compensation, including but not limited to salary, bonus, incentive compensation and equity awards based on such an evaluation. In addition, the compensation committee is responsible for regularly reviewing the Company’s compensation, recruitment, retention and termination policies for senior executives.
In 2019, a performance evaluation of the Company’s executive officers for the year ended December 31, 2018 will be undertaken. Following each performance evaluation, the Company’s compensation committee will review and approve changes to the compensation of the Company’s executive officers based on the individual levels of achievement against pre-established performance objectives.
Further information regarding executive compensation for the year ended December 31, 2018, as required by Item 11 of this Annual Report on Form 10-K, is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders, to be filed with the SEC and the ASX within 120 days of December 31, 2018. Such information is incorporated herein by reference.
Principle 2 – Structure the board to add value
Recommendation 2.1 – The board of a listed entity should:
| a) | have a nomination committee which: |
| i. | has at least three members, a majority of whom are independent directors; and |
| ii. | is chaired by an independent director; |
and disclose:
| iii. | the charter of the committee; |
| iv. | the members of the committee; and |
| v. | as at the end of each reporting period the number of times the committee met throughout the period and the individual attendances of the members at those meetings. |
As of December 31, 2018, the members of the nominating and corporate governance committee were Daniel Moore and Dr. Oern Stuge (Chair). Mr. Moore and Dr. Stuge are considered independent directors for ASX, NASDAQ and SEC purposes and a copy of the Nominating and Corporate Governance Committee Charter is available on the corporate governance section of the Company’s website. The nominating and corporate governance committee met once during 2018 and both members were present at the meeting.
57
At December 31, 2018, there were only two directors appointed as members of the nominating and corporate governance committee (instead of the recommended three members), which was a decision made by the Board initially at the end of 2017 (and reported in the corporate governance statement for 31 December 2017) in order to properly utilize the resources of the four members of the Board across all of the various committees. As a result of only having two members, the Company was not fully compliant with recommendation 2.1 for the 2018 reporting period. The Board of Directors will continue to periodically assess the effectiveness of this committee, including the size and the experience of the members appointed, with a view to ensuring that the committee’s performance accords with the best possible practice in the context of the overall Board size and structure.
Recommendation 2.2 – A listed entity should have and disclose a board skills matrix setting out the mix of skills and diversity that the board currently has or is looking to achieve in its membership.
The nominating and corporate governance committee is responsible for reviewing with the Board from time to time the appropriate skills and characteristics required of Board members in the context of the current make-up of the Board and the Company’s business needs. This assessment includes, among other things, an individual’s business experience and skills (including skills in core areas such as operations, management, technology, medical device industry knowledge, accounting and finance, marketing, leadership, strategic planning and international markets), independence, judgment, integrity and ability to commit sufficient time and attention to the activities of the Board, as well as the absence of any potential conflicts with the Company’s interests. The nominating and corporate governance committee considers these criteria in the context of an assessment of the perceived needs of the Board as a whole and seeks to achieve diversity of occupational and personal backgrounds on the Board.
Information regarding the skills, experience and expertise relevant to each director is set out in the section titled “Directors of the Registrant” in this Item 10.
While the Board did not disclose the Board skills matrix for the reporting period the Board believes that its members possess the mix of skills and diversity that the Company needs at this stage of its development. The Board continues to evolve a board skills matrix setting out the mix of skills and diversity that the Board currently has or is looking to achieve in its membership.
Recommendation 2.3 – A listed entity should disclose:
| a) | the names of the directors considered by the board to be independent directors; |
| b) | if a director has an interest, position, association or relationship of the type described in this recommendation but the board is of the opinion that it does not compromise the independence of the director, the nature of the interest, position, association or relationship in question and an explanation of why the board is of that opinion; and |
| c) | the length of service of each director. |
The Company considers that a director is an independent director where that director is free from any interest and any business or other relationship which could, or could reasonably be perceived to, materially interfere with the director’s decisions relating to the Company or with the director’s ability to act in the best interests of the Company. The Company also assesses the independence of its directors with regard to requirements for independence set out under ASX Governance Recommendation 2.3.
58
The composition and tenure of the Board as of December 31, 2018, as well as each member’s independence status during 2018, was as follows:
|
|
|
|
|
|
| Committees | |||||
|
|
|
|
|
|
|
|
|
|
|
| Nominating |
|
|
|
|
|
|
|
|
|
|
|
| and |
|
|
|
|
|
|
|
|
|
|
|
| Corporate |
Director |
| Director Position |
| Tenure1 |
| Independent |
| Audit |
| Compensation |
| Governance |
Daniel J. Moore |
| Non-executive Chairman |
| 4.3 years |
| Yes |
|
|
|
|
| X |
Timothy J. Barberich |
| Non-executive Director |
| 7.6 years |
| Yes |
| X |
| Chair |
|
|
Dr. Oern Stuge |
| Non-executive Director |
| 2.0 years |
| Yes |
|
|
| X |
| Chair |
Juliet Thompson |
| Non-executive Director |
| 1.3 years |
| Yes |
| Chair |
|
|
|
|
1. | Calculated as of December 31, 2018. |
The number of directors’ meetings (including meetings of committees) and number of meetings attended by each of the directors during the reporting period are as follows
|
|
|
|
|
| Committee Meetings | ||||||||||
|
|
|
|
|
|
|
|
| Nominating and |
| Compensation | |||||
|
| Directors’ Meetings |
| Audit Committee |
| Corporate Governance |
| Committee | ||||||||
Director |
| A |
| B |
| A |
| B |
| A |
| B |
| A |
| B |
Daniel J. Moore |
| 7 |
| 7 |
| — |
| — |
| 1 |
| 1 |
| — |
| — |
Timothy J. Barberich |
| 7 |
| 7 |
| 4 |
| 4 |
| — |
| — |
| 2 |
| 2 |
Dr. Oern Stuge |
| 7 |
| 7 |
| — |
| — |
| 1 |
| 1 |
| 2 |
| 2 |
Juliet Thompson |
| 7 |
| 7 |
| 4 |
| 4 |
| — |
| — |
| — |
| — |
1 – Calculated as of December 31, 2018
A – Number of meetings attended.
B – Number of meetings held during the time the director held office during the reporting period.
Independent advice
At the Company’s expense, each member of the Board and each member of a committee of the Board is entitled to seek advice from independent external advisers in relation to any matter that is considered necessary to fulfil their relevant duties and responsibilities.
Recommendation 2.4 – A majority of the board of a listed entity should be independent directors.
The Board for the reporting period comprised a majority of independent directors.
Recommendation 2.5 – The chair of the board of a listed entity should be an independent director and, in particular, should not be the same person as the CEO of the entity.
In compliance with the ASX Governance Recommendations, the chairman of the Board is an independent director and the roles of the chairman and the chief executive officer of the Company are not currently exercised by the same individual. However, the Company’s Board Charter does not specifically address whether or not the offices of chairman and chief executive officer should be vested in the same person or two different people, or whether the chairman should be an employee of the Company or should be elected from among the non-executive directors. The needs of the Company and the individuals available to serve in these roles may dictate different outcomes at different times, and the Board believes that retaining flexibility in these decisions is in the best interest of the Company and its shareholders.
59
Recommendation 2.6 – A listed entity should have a program for inducting new directors and provide appropriate professional development opportunities for directors to develop and maintain the skills and knowledge needed to perform their roles as directors effectively.
The nominating and corporate governance committee of the Board continually assesses the needs of the Company and the skills and knowledge required of its Board members. On appointment, new directors are provided with induction information that generally includes historical information about the Company and its operations, details of the Company’s directors’ and officers’ insurance, the Company’s corporate governance guidelines, and other Company governance policies. The induction process also involves one-on-one discussions with the Chairman and other directors and briefings from senior management to help new directors participate actively in Board decision making at the earliest opportunity. When it is necessary, resources are provided for the Board as a whole, and for individual Board members as needed, to supplement their skills and knowledge and fill any identified gaps.
Principle 3 – Act ethically and responsibly
Recommendation 3.1 – A listed entity should:
| a) | have a code of conduct for its directors, senior executives and employees; and |
| b) | disclose that code or a summary of it. |
The Company has adopted a Code of Business Conduct and Ethics and an Insider Trading Policy, copies of which are available on the corporate governance section of the Company’s website.
Principle 4 – Safeguard integrity in corporate reporting
Recommendation 4.1 – The board of a listed entity should:
| a) | have an audit committee which: |
| i. | has at least three members, all of whom are non-executive directors and a majority of whom are independent directors; and |
| ii. | is chaired by an independent director, who is not the chair of the board; |
and disclose:
| iii. | the charter of the committee; |
| iv. | the relevant qualifications and experience of the members of the committee; and |
| v. | in relation to each reporting period, the number of times the committee met throughout the period and the individual attendances of the members at those meetings. |
The Board has established an audit committee to oversee the management of the Company’s financial and internal risks and reporting. As of December 31, 2018, the members of the audit committee were Juliet Thompson and Timothy Barberich, both independent, non-executive directors. The audit committee is chaired by Juliet Thompson who is an independent director and not chair of the Board. At December 31, 2017, in order to fully utilize resources of the four members of the Board of Directors, each Director was assigned to one or two committees.
The audit committee met four times during 2018 with Ms. Thompson and Mr. Barberich attending on all four occasions.
At December 31, 2018, there were only two directors appointed as members of the audit committee (instead of the recommended three members), which was a decision made by the Board initially at the end of 2017 (and reported in the corporate governance statement for 31 December 2017) in order to properly utilize the resources of the four members of the Board across all of the various committees. As a result of only having two members, the Company was not fully compliant with recommendation 4.1 for the 2018 reporting period. The Board of Directors will continue to periodically assess the effectiveness of this committee, including the size and the experience of the members appointed, with a view to ensuring that the committee’s performance accords with the best possible practice in the context of the overall Board size and structure.
60
The members of the audit committee must be financially literate and have familiarity with financial and accounting matters, with at least one member a qualified accountant or other financial professional with appropriate expertise in financial and accounting matters. The qualifications of those appointed to the audit committee are set out in the section titled “Directors of the Registrant” in this Item 10.
The audit committee is governed by the Audit Committee Charter, a copy of which is available on the corporate governance section of the Company’s website.
In its Audit Committee Charter, the Company has disclosed its policy for the selection and appointment of the Company’s independent auditor and for the rotation of the lead audit partner having primary responsibility for the audit and the audit partner responsible for reviewing the audit every five years. The audit committee will regularly report to the Board about committee activities, issues and related recommendations.
Recommendation 4.2 – The board of a listed entity should, before it approves the entity’s financial statements for a financial period, receive from its CEO and CFO a declaration that, in their opinion, the financial records of the entity have been properly maintained and that the financial statements comply with the appropriate accounting standards and give a true and fair view of the financial position and performance of the entity and that the opinion has been formed on the basis of a sound system of risk management and internal controls which is operating effectively.
As the Company prepares and files its financial statements under United States accounting practices and laws, management is required to provide representations to the Board on a wide range of issues, including the effectiveness of the Company’s disclosure controls and procedures as well as the design and operation of internal control over financial reporting. However, as the Company is incorporated in the State of Delaware, United States, it is not required to provide a declaration under section 295A of the Corporations Act. To this end, shareholders’ attention is drawn to Item 9A of this Annual Report on Form 10-K and the certifications provided by the principal executive officer and the principal financial officer at the end of the Annual Report on Form 10-K. As stated above, Item 9A discloses information regarding the Company’s controls and procedures and management’s evaluation of the effectiveness of its internal control over financial reporting. As required by Rule 13a-15(d) of the Exchange Act, our management, including our principal executive officer and our principal financial and accounting officer, conducted an evaluation of the internal control over financial reporting to determine whether any changes occurred during the year ended December 31, 2018 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting. Based on that evaluation, our principal executive officer and principal financial and accounting officer have concluded that, based on and as of the time of such evaluation, our disclosure controls and procedures were not effective at the reasonable assurance level. After further investigation, we believe the material weaknesses did not result in any material misstatements to our financial statements and we believe the consolidated financial statements included in this Annual Report present, in all material respects, our financial position, results of operations and cash flows for the periods presented in conformity with U.S. generally accepted accounting principles. Additionally, management is taking steps to remediate the weaknesses, as described in Item 9A and the Board has been frequently updated as to progress of remediation efforts.
Recommendation 4.3 – A listed entity that has an annual general meeting should ensure that its external auditor attends its AGM and is available to answer questions from security holders relevant to the audit.
The Company’s policy is to ensure its external auditor attends the annual general meeting of shareholders, in person, to have an opportunity to make a statement, if desired, and to respond to appropriate questions from security holders regarding the audit. The Company’s auditor for the years ended December 31, 2018 and December 31, 2017 was Moody, Famiglietti & Andronico, LLP (“MFA”). MFA attended the 2018 annual general meeting in respect of the financial year ended December 31, 2017.
Principle 5 – Make timely and balanced disclosure
Recommendation 5.1 – A listed entity should:
| a) | have a written policy for complying with its continuous disclosure obligations under the Listing Rules; and |
| b) | disclose that policy or a summary of it. |
The Company is committed to providing timely and balanced disclosure to the market in accordance with its continuous disclosure obligations. In accordance with its commitment to fully comply with its continuous disclosure
61
obligations and to ensure accountability at a senior management level for that compliance, the Company has adopted a Continuous Disclosure Policy, together with other internal mechanisms and reporting requirements. A copy of the Company’s Continuous Disclosure Policy is available on the corporate governance section of the Company’s website. In addition, a copy of all of the Company’s ASX announcements, financial reports and related public information is also available on the Company’s website.
Principle 6 – Respect the rights of security holders
Recommendation 6.1 – A listed entity should provide information about itself and its governance to investors via its website.
The Company aims to provide shareholders with comprehensive and timely access to Company documents and releases through its website, including:
| • | details of the Company’s certificate of incorporation and bylaws, Board and committee charters and key corporate governance policies; |
| • | copies of all material information lodged with ASX and any other applicable securities regulators and securities exchanges; |
| • | copies of material announcements, financial reports, briefings and speeches made to the market or media; |
| • | a means for the shareholders to submit enquiries directly to the Company; |
| • | the full text of notices of shareholder meetings and explanatory material; and |
| • | advance notice of all open briefings to institutional investors and analysts, including copies of presentation materials. |
Other information may be provided to shareholders via periodic mail-outs. In addition, the Company allows shareholders to elect to receive email communications where appropriate.
Recommendation 6.2 – A listed entity should design and implement an investor relations program to facilitate effective two-way communications with investors.
The Company has adopted a Shareholder Communications Policy which supports effective two-way communication with its shareholders, a copy of which is available on the corporate governance section of the Company’s website. The Company seeks to utilize numerous modes of communication, including electronic communication, to ensure that its communication with shareholders is frequent, clear, and accessible. Shareholders are entitled to and encouraged to participate in briefing calls and/or contact the Company directly with questions or concerns. Contact information is provided in each communication with shareholders, as well as on the Company’s website.
Recommendation 6.3 – A listed entity should disclose the policies and processes it has in place to facilitate and encourage participation at meetings of security holders.
All shareholders are invited to attend the Company’s annual general meeting either in person or by proxy. The Board regards the annual general meeting as an excellent forum in which to discuss issues relevant to the Company and accordingly encourages full participation by shareholders. To facilitate attendance, the Company arranges the annual general meeting to be held in an easily accessed location and announces the date and location of the meeting in advance of the meeting. Shareholders have an opportunity to submit questions to the Board and the Company’s auditor. The meeting may also be audio cast and/or webcast to provide access to those shareholders who are unable to attend the annual general meeting in person.
Recommendation 6.4 – A listed entity should give security holders the option to receive communications from, and send communications to, the entity and its security registry electronically.
The Company provides its shareholders with the option to receive communications from, and send communications to, the Company and the Company’s share registry electronically.
62
Principle 7 – Recognize and manage risk
Recommendation 7.1 – The board of a listed entity should:
| a) | have a committee or committees to oversee risk, each of which: |
| i. | has at least three members, a majority of whom are independent directors; and |
| ii. | is chaired by an independent director; |
and disclose:
| iii. | the charter of the committee; |
| iv. | the members of the committee; and |
| v. | as at the end of each reporting period, the number of times the committee met throughout the period and the individual attendances of the members at those meetings. |
The risks that the Company faces are continually changing in line with the development of the Company. In simple terms, risk is inherent in all activities undertaken by the Company. Many of these risks are beyond the control of the Company and, as such, it is important that risk be mitigated on a continuous basis, particularly if the Company is to preserve shareholder value.
To ensure appropriate oversight and management of material business risks, the Company has adopted a Risk Management Policy that sets forth the process to identify, assess, and manage risk in the Company’s business operations. A copy of the policy is available on the corporate governance section of the Company’s website.
The day-to-day oversight and management of the Company’s risk management program has been conferred upon the audit committee. The audit committee is responsible for ensuring that the Company maintains effective risk management and internal control systems and processes and provides regular reports to the Board on the effectiveness of the risk management program in identifying and addressing material business risks. Details of the audit committee are set out above in response to ASX Governance Recommendation 4.1.
In addition, the Board is responsible for reviewing and ratifying the risk management structure, processes and guidelines which are developed and maintained by senior management. The audit committee or management may also refer particular risk management issues to the Board for final consideration and direction.
Recommendation 7.2 – The board or a committee of the board should:
| a) | review the entity’s risk management framework at least annually to satisfy itself that it continues to be sound; and |
| b) | disclose, in relation to each reporting period, whether such a review has taken place. |
While the Board does not currently conduct a formal annual review of the material risks to the Company and the methods used to identify and communicate those risks, the Board continually assesses these matters. The Board holds regular meetings by teleconference as well as at the Company’s facility in Boston, Massachusetts, for the purposes of discussing and reviewing operational developments and reviewing the effectiveness of the implementation of the Company’s risk management systems.
The Company’s Risk Management Policy (a copy of which is available on the corporate governance section of the Company’s website) also requires that management report on an on-going basis to the Board, primarily through the audit committee which has the responsibility for day-to-day oversight of the Company’s risk management program, on the status and effectiveness of the risk management program.
Recommendation 7.3 – A listed entity should disclose:
| a) | if it has an internal audit function, how the function is structured and what role it performs; or |
| b) | if it does not have an internal audit function, that fact and the processes it employs for evaluating and continually improving the effectiveness of its risk management and internal control processes. |
63
The Company does not currently have an internal audit function. Rather, the Company has implemented the following processes to evaluate and continually improve the effectiveness of its risk management and internal control processes:
| • | the Board has conferred responsibility on senior management to develop and maintain a risk management program in light of the day-to-day needs of the Company; |
| • | the Board has established three standing committees to provide focused support in key areas; namely the nominating and corporate governance committee, audit committee and compensation committee; |
| • | management provides the Board with frequent updates on the state of the Company’s business, including the risks that the Company faces from time-to-time allowing the Board to assess the Company’s management of its material business risks. These updates include up-to-date financial information, operational activity, clinical status and competitor updates. These updates are founded on internal communications that are fostered internally through weekly management meetings and other internal communications; and |
| • | these processes operate in addition to the Company’s system of internal controls over financial reporting, its quality system, complaint handling processes, employee policies and standard operating procedures, which are all designed to address various forms of risk. |
| • | Management is taking steps to remediate the weaknesses, as described in Item 9A and the Board has been frequently updated as to progress of remediation efforts. |
Recommendation 7.4 – A listed entity should disclose whether it has any material exposure to economic, environmental and social sustainability risks and, if it does, how it manages or intends to manage those risks.
The economic risks that the Company is subject to and must manage are set out in the “Risk Factors” section of this Annual Report on Form 10-K. In general, the Board considers that the Company is not susceptible to material environmental or social sustainability risks in operating its business.
Principle 8 – Remunerate fairly and responsibly
Recommendation 8.1 – The board of a listed entity should:
| a) | have a remuneration committee which: |
| i. | has at least three members, a majority of whom are independent directors; and |
| ii. | is chaired by an independent director; |
and disclose:
| iii. | that charter of the committee; |
| iv. | the members of the committee; and |
| v. | as at the end of each reporting period, the number of times the committee met throughout the period and the individual attendances of the members at those meetings. |
The Board has established a compensation committee to review and assess executive and director compensation. The compensation committee is governed by the Compensation Committee Charter, a copy of which is available on the corporate governance section of the Company’s website.
As of December 31, 2018, the members of the compensation committee were Timothy J. Barberich (Chair) and Dr. Oern Stuge both independent non-executive directors. The committee has two directors appointed (instead of the recommended three members), which was a decision made by the Board initially during 2017 (and reported in the corporate governance statement for 31 December 2017) in order to properly utilize the resources of the four members of the Board across all of the various committees. As a result of only having two members, the Company was not fully compliant with recommendation 8.1 for the 2018 reporting period. The Board of Directors will periodically assess the effectiveness of this committee, including the size and the experience of the members appointed, with a view to ensuring that the committee’s performance accords with the best possible practice in the context of the overall Board size and structure.
64
While the compensation committee reviews and reports compensation items to the Board for both non-executive directors and executive management, including each individual’s skills, knowledge, and contributions to the Company, the compensation committee does not provide a separate report of compensation by gender.
Recommendation 8.2 – A listed entity should separately disclose its policies and practices regarding the remuneration of non-executive directors and the remuneration of executive directors and other senior executives.
In accordance with the Compensation Committee Charter, the compensation committee is responsible for ensuring that the structure of non-executive and executive directors’ and other senior executives’ compensation is clearly distinguished.
The Company has adopted a non-executive director compensation policy pursuant to which non-executive directors are compensated for their services to the Board which includes annual cash fees for serving as a member or the chair of the Board and for serving as a member or the chair of the Board committees. In addition, the policy provides that our non-executive directors may receive grants of a fixed number of options upon their joining the Board and annual grants (which commenced in 2014) of a fixed number of options and restricted stock units, in each case subject to the terms of the non-executive director compensation policy as well as the approval of shareholders. Juliet Thompson was granted 30,000 non-qualified stock options on August 22, 2017, and approval was obtained at the Company’s May 2018 annual general meeting.
The Company has adopted a separate executive compensation program that consists of base salary, equity-based incentives, performance-based cash bonuses, severance benefits, and other customary benefits such as health insurance on the same basis as provided to all other employees. None of the Company’s non-executive directors are entitled to any retirement benefits.
Further information regarding the compensation committee, as required by Item 10 of this Annual Report on Form 10-K, is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders to be filed with the SEC and ASX.
Recommendation 8.3 – A listed entity which has an equity-based remuneration scheme should:
| a) | have a policy on whether participants are permitted to enter into transactions (whether through the use of derivatives or otherwise) which limit the economic risk of participating in the scheme; and |
| b) | disclose that policy or a summary of it. |
The Company provides compensation in the form of equity-based awards to non-executive directors (upon approval by shareholders), senior executives, and employees of the Company. Awards are made under the Company’s 2011 Employee, Director and Consultant Equity Incentive Plan, as amended, which has been approved by shareholders. The Company’s Insider Trading Policy, a copy of which is available on the corporate governance section of the Company’s website, provides a summary of the Company’s policy on prohibiting entering into transactions in associated products which limit the economic risk of participating in unvested entitlements under any equity-based remuneration schemes. This policy operates to help limit the economic risk to the Company’s securities.
This report is made in accordance with a resolution of the Board.
ITEM 11. EXECUTIVE COMPENSATION
The information required by this Item 11 is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders to be filed with the SEC and ASX.
65
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The information relating to security ownership of certain beneficial owners of our Common Stock and information relating to the security ownership of our management required by this Item 12 is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders to be filed with the SEC and ASX.
The table below sets forth information with regard to shares authorized for issuance under our equity compensation plans as of December 31, 2018. As of December 31, 2018, we had two active equity compensation plans, each of which was approved by our stockholders:
| • | Our 2003 Omnibus Stock Plan; and |
| • | Our 2011 Employee, Director and Consultant Equity Incentive Plan. |
| Number of shares to be issued |
|
| Weighted-average |
| Number of shares remaining |
| |||
| upon exercise of outstanding |
|
| exercise price of |
| available for future issuance |
| |||
|
| options or vesting of |
|
| outstanding |
| under equity compensation |
| ||
Plan Category |
| restricted stock units |
|
| options |
| plans1 |
| ||
Equity compensation plans approved by security holders |
|
| 1,377,883 |
|
| 1.77 |
|
| 1,052,917 |
|
Equity compensation plans not approved by security holders |
| — |
|
|
|
| — |
| ||
Total |
|
| 1,377,883 |
|
| 1.77 |
|
| 1,052,917 |
|
1) | Our 2011 Employee, Director and Consultant Equity Incentive Plan allows for an annual increase in the number of shares available for issue commencing on the first day of each fiscal year during the period beginning in fiscal year 2012 and ending in fiscal year 2020. The annual increase in the number of shares shall be equal to the lowest of: (i) 500,000 shares; (ii) 4% of the number of common shares outstanding as of such date; and (iii) an amount determined by our board of directors or our compensation committee. |
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
The information required by this Item 13 is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders to be filed with the SEC and ASX.
ITEM 14. PRINCIPAL ACCOUNTANT FEES AND SERVICES
The information required by this Item 14 is incorporated by reference to the applicable information in our proxy statement for our 2019 Annual Meeting of Stockholders to be filed with the SEC and ASX.
66
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
| (a) | List of documents filed as part of this Annual Report on Form 10-K |
| (1) | Consolidated Financial Statements listed under Part II, Item 8 and included herein by reference. |
| (2) | Consolidated Financial Statement Schedules |
No schedules are submitted because they are not applicable, not required or because the information is included in the Consolidated Financial Statements or Notes to Consolidated Financial Statements.
| (3) | Exhibits |
The exhibits listed in the accompanying Exhibit Index are filed or incorporated by reference as part of this Annual Report on Form 10-K.
Not applicable.
Exhibit No: |
| Description |
|
|
|
3.1.1 |
| |
|
|
|
3.1.2 |
| |
|
|
|
3.1.3 |
| |
|
|
|
3.2 |
| |
|
|
|
4.1 |
| |
|
|
|
4.2 |
| |
|
|
|
4.3 |
| |
|
|
|
10.1† |
| |
|
|
|
10.2† |
| |
|
|
|
10.3 |
| |
|
|
|
67
10.4 |
| Sublease Agreement, dated May 23, 2013, between GI Dynamics, Inc. and Cambridge Technology, Inc. incorporated by reference to Exhibit 10.8 of GI Dynamics, Inc.’s registration statement on Form 10, filed with the SEC on April 30, 2014 |
|
|
|
10.5 |
| |
|
|
|
10.6† |
| |
|
|
|
10.7† |
| |
|
|
|
10.8† |
| |
|
|
|
10.9† |
| |
|
|
|
10.11 |
| |
|
|
|
10.12 |
| |
|
|
|
10.13* |
| |
|
|
|
10.14 |
| |
|
|
|
10.15* |
| |
|
|
|
10.16 |
| |
|
|
|
10.17 |
| |
|
|
|
10.18 |
| |
|
|
|
21.1 |
| |
|
|
|
23.1* |
| |
|
|
|
31.1* |
| Certification of principal executive officer pursuant to Rules 13a-14 or 15d-14 of the Exchange Act |
|
|
|
31.2* |
| Certification of principal financial officer pursuant to Rules 13a-14 or 15d-14 of the Exchange Act |
|
|
|
32.1‡ |
| |
|
|
|
68
| ||
|
|
|
101.INS |
| XBRL Instance Document |
|
|
|
101.SCH |
| XBRL Taxonomy Extension Schema Document |
|
|
|
101.CAL |
| XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
101.LAB |
| XBRL Taxonomy Extension Label Linkbase Database |
|
|
|
101.PRE |
| XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
101.DEF |
| XBRL Taxonomy Extension Definition Linkbase Document |
* | Filed herewith. |
‡ | Furnished herewith. |
† | Management contract or compensatory plan or arrangement. |
69
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, hereunto duly authorized.
|
| GI Dynamics, Inc. | ||
Date: March 12, 2019 |
| By: |
| /s/ SCOTT W. SCHORER |
|
| Name: |
| Scott W. Schorer |
|
| Title: |
| President, Chief Executive Officer |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons in the capacities and on the dates indicated.
Signature |
| Title |
| Date |
|
|
|
|
|
/s/ SCOTT W. SCHORER
Scott W. Schorer |
| President, Chief Executive Officer (Principal Executive Officer) |
| March 12, 2019
|
|
|
|
|
|
/s/ CHARLES R. CARTER
Charles R. Carter |
| Chief Financial Officer, Secretary (Principal Financial and Accounting Officer) |
| March 12, 2019
|
|
|
|
|
|
/s/ DANIEL J. MOORE
Daniel J. Moore |
| Chairman and Director |
| March 12, 2019 |
|
|
|
|
|
/s/ TIMOTHY J. BARBERICH
Timothy J. Barberich |
| Director
|
| March 12, 2019
|
|
|
|
|
|
/s/ OERN STUGE, MD
Oern Stuge, MD |
| Director
|
| March 12, 2019
|
|
|
|
|
|
/s/ JULIET THOMPSON
Juliet Thompson |
| Director
|
| March 12, 2019
|
70
GI Dynamics, Inc. and Subsidiaries
Index to Consolidated Financial Statements
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of
GI Dynamics, Inc.
Boston, Massachusetts
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of GI Dynamics, Inc. and Subsidiaries (the Company) as of December 31, 2018 and 2017, and the related consolidated statements of operations, stockholders’ equity (deficit), and cash flows for each of the years in the two-year period ended December 31, 2018, and the related notes (collectively referred to as the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2018 and 2017, and the results of their operations and their cash flows for each of the years in the two-year period then ended, in conformity with accounting principles generally accepted in the United States of America.
Going Concern Uncertainty
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has incurred operating losses since inception and at December 31, 2018, has an accumulated deficit and working capital deficiency. These matters raise substantial doubt about the Company’s ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Moody, Famiglietti & Andronico, LLP
We have served as the Company’s auditor since 2016. | |
|
|
Tewksbury, Massachusetts | |
|
|
March 12, 2019 |
|
F-2
GI Dynamics, Inc. and Subsidiaries
(In thousands, except share and per share amounts)
|
| December 31, |
| |||||
|
| 2018 |
|
| 2017 |
| ||
Assets |
|
|
|
|
|
|
|
|
Current assets: |
|
|
|
|
|
|
|
|
Cash and cash equivalents |
| $ | 3,806 |
|
| $ | 3,034 |
|
Restricted cash |
|
| 30 |
|
|
| 30 |
|
Accounts receivable, net |
|
| — |
|
|
| 40 |
|
Inventory, net |
|
| — |
|
|
| — |
|
Prepaid expenses and other current assets |
|
| 530 |
|
|
| 265 |
|
Total current assets |
|
| 4,366 |
|
|
| 3,369 |
|
Property and equipment, net |
|
| 63 |
|
|
| 97 |
|
Total assets |
| $ | 4,429 |
|
| $ | 3,466 |
|
Liabilities and stockholders’ deficit |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
| $ | 1,050 |
|
| $ | 1,191 |
|
Accrued expenses |
|
| 1,645 |
|
|
| 1,021 |
|
Deferred revenue |
|
| — |
|
|
| 11 |
|
Short term debt to related party, net of debt discount |
|
| 4,960 |
|
|
| 4,465 |
|
Derivative liabilities |
|
| 51 |
|
|
| 493 |
|
Total current liabilities |
|
| 7,706 |
|
|
| 7,181 |
|
Long term debt to related party, net of discount |
|
| 168 |
|
|
| — |
|
Total liabilities |
|
| 7,874 |
|
|
| 7,181 |
|
Commitments (Note 11) |
|
|
|
|
|
|
|
|
Stockholders’ deficit: |
|
|
|
|
|
|
|
|
Preferred stock, $0.01 par value – 500,000 shares authorized; no shares issued and outstanding at December 31, 2018 and 2017 |
|
| — |
|
|
| — |
|
Common stock, $0.01 par value – 50,000,000 shares authorized at December 31, 2018 and 2017, respectively; 19,277,545 and 11,157,489 shares issued and outstanding at December 31, 2018 and 2017, respectively |
|
| 193 |
|
|
| 112 |
|
Class B common stock, $0.01 par value – zero and 1,000,000 shares authorized at December 31, 2018 and 2017; no shares issued and outstanding at December 31, 2018 and 2017 |
|
| — |
|
|
| — |
|
Additional paid-in capital |
|
| 263,521 |
|
|
| 255,294 |
|
Accumulated deficit |
|
| (267,159 | ) |
|
| (259,121 | ) |
Total stockholders’ deficit |
|
| (3,445 | ) |
|
| (3,715 | ) |
Total liabilities and stockholders’ deficit |
| $ | 4,429 |
|
| $ | 3,466 |
|
The accompanying notes are an integral part of these consolidated financial statements.
F-3
GI Dynamics, Inc. and Subsidiaries
Consolidated Statements of Operations
(In thousands, except share and per share amounts)
|
| Years Ended December 31, |
| |||||
|
| 2018 |
|
| 2017 |
| ||
Revenue |
| $ | — |
|
| $ | 53 |
|
Cost of revenue |
|
| — |
|
|
| 265 |
|
Gross loss |
|
| — |
|
|
| (212 | ) |
|
|
|
|
|
|
|
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
Research and development |
|
| 1,947 |
|
|
| 3,560 |
|
Sales and marketing |
|
| 866 |
|
|
| 1,811 |
|
General and administrative |
|
| 4,809 |
|
|
| 4,951 |
|
Total operating expenses |
|
| 7,622 |
|
|
| 10,322 |
|
Loss from operations |
|
| (7,622 | ) |
|
| (10,534 | ) |
|
|
|
|
|
|
|
|
|
Other income (expense): |
|
|
|
|
|
|
|
|
Interest income |
|
| 29 |
|
|
| 33 |
|
Interest expense |
|
| (1,151 | ) |
|
| (321 | ) |
Foreign exchange gain (loss) |
|
| 13 |
|
|
| (10 | ) |
Income (expense) from diverted funds and insurance claim |
|
| 224 |
|
|
| (170 | ) |
Re-measurement of derivative liabilities |
|
| 482 |
|
|
| 126 |
|
Other income (expense), net |
|
| (403 | ) |
|
| (342 | ) |
|
|
|
|
|
|
|
|
|
Loss before income tax expense |
|
| (8,025 | ) |
|
| (10,876 | ) |
Income tax expense |
|
| 13 |
|
|
| 14 |
|
Net loss |
| $ | (8,038 | ) |
| $ | (10,890 | ) |
Basic and diluted net loss per common share |
| $ | (0.59 | ) |
| $ | (0.98 | ) |
Weighted-average number of common shares used in basic and diluted net loss per common share |
|
| 13,699,585 |
|
|
| 11,157,489 |
|
The accompanying notes are an integral part of these consolidated financial statements.
F-4
GI Dynamics, Inc. and Subsidiaries
Consolidated Statements of Stockholders’ Equity (Deficit)
(In thousands, except share amounts)
|
| Common Stock |
|
| Additional |
|
|
|
|
|
| Total |
| |||||||
|
| Shares |
|
| Par Value |
|
| Paid-in Capital |
|
| Accumulated Deficit |
|
| Stockholders' Equity (deficit) |
| |||||
Balance at December 31, 2016 |
|
| 10,907,857 |
|
| $ | 109 |
|
| $ | 254,884 |
|
| $ | (248,203 | ) |
| $ | 6,790 |
|
Issuance of shares upon private placement, net of issuance costs |
|
| 249,632 |
|
|
| 3 |
|
|
| 195 |
|
|
| — |
|
|
| 198 |
|
Stock-based compensation expense related to accounting change with respect to forfeiture rate |
|
| — |
|
|
| — |
|
|
| 28 |
|
|
| (28 | ) |
|
| — |
|
Stock-based compensation expense |
|
| — |
|
|
| — |
|
|
| 187 |
|
|
| — |
|
|
| 187 |
|
Net loss |
|
| — |
|
|
| — |
|
|
| — |
|
|
| (10,890 | ) |
|
| (10,890 | ) |
Balance at December 31, 2017 |
|
| 11,157,489 |
|
|
| 112 |
|
|
| 255,294 |
|
|
| (259,121 | ) |
|
| (3,715 | ) |
Issuance of shares upon private placement, net of issuance costs |
|
| 8,120,056 |
|
|
| 81 |
|
|
| 6,353 |
|
|
| — |
|
|
| 6,434 |
|
Beneficial conversion feature discount associated with 2018 Note |
|
|
|
|
|
|
|
|
|
| 1,007 |
|
|
|
|
|
|
| 1,007 |
|
Relative fair value of warrant issued with 2018 Note |
|
| — |
|
|
| — |
|
|
| 743 |
|
|
| — |
|
|
| 743 |
|
Stock-based compensation expense |
|
| — |
|
|
| — |
|
|
| 124 |
|
|
| — |
|
|
| 124 |
|
Net loss |
|
| — |
|
|
| — |
|
|
| — |
|
|
| (8,038 | ) |
|
| (8,038 | ) |
Balance at December 31, 2018 |
|
| 19,277,545 |
|
| $ | 193 |
|
| $ | 263,521 |
|
| $ | (267,159 | ) |
| $ | (3,445 | ) |
The accompanying notes are an integral part of these consolidated financial statements.
F-5
GI Dynamics, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
(In thousands)
|
| Years Ended December 31, |
| |||||
|
| 2018 |
|
| 2017 |
| ||
Operating activities: |
|
|
|
|
|
|
|
|
Net loss |
| $ | (8,038 | ) |
| $ | (10,890 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
Depreciation and amortization |
|
| 35 |
|
|
| 57 |
|
Loss on disposal of leasehold improvements |
|
| 2 |
|
|
| — |
|
Stock-based compensation expense |
|
| 124 |
|
|
| 187 |
|
Amortization of debt issuance costs to non-cash interest expense |
|
| 156 |
|
|
| 44 |
|
Non-cash interest expense |
|
| 359 |
|
|
| 136 |
|
Accretion of debt discount |
|
| 168 |
|
|
| — |
|
Non-cash interest expense from derivative liabilities |
|
| 464 |
|
|
| 138 |
|
Non-cash gain from re-measurement of derivative liabilities |
|
| (482 | ) |
|
| (126 | ) |
Change in inventory reserve |
|
| — |
|
|
| (76 | ) |
Changes in operating assets and liabilities: |
|
|
|
|
|
|
|
|
Accounts receivable |
|
| 40 |
|
|
| (10 | ) |
Prepaid expenses and other current assets |
|
| (265 | ) |
|
| 218 |
|
Inventory |
|
| — |
|
|
| 289 |
|
Accounts payable |
|
| (141 | ) |
|
| 185 |
|
Accrued expenses |
|
| 265 |
|
|
| (275 | ) |
Deferred revenue |
|
| (11 | ) |
|
| — |
|
Net cash used in operating activities |
|
| (7,324 | ) |
|
| (10,123 | ) |
Investing activities |
|
|
|
|
|
|
|
|
Purchases of property and equipment |
|
| (3 | ) |
|
| (5 | ) |
Net cash used in investing activities |
|
| (3 | ) |
|
| (5 | ) |
Financing activities |
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock, net of issuance costs |
|
| 6,434 |
|
|
| 198 |
|
Debt issuance costs |
|
| (85 | ) |
|
| (115 | ) |
Proceeds from long term debt-related party |
|
| 1,750 |
|
|
| 5,000 |
|
Payments on short term note payable |
|
| — |
|
|
| (214 | ) |
Net cash provided by financing activities |
|
| 8,099 |
|
|
| 4,869 |
|
Net increase (decrease) in cash, cash equivalents and restricted cash |
|
| 772 |
|
|
| (5,259 | ) |
Cash, cash equivalents and restricted cash at beginning of year |
|
| 3,064 |
|
|
| 8,323 |
|
Cash, cash equivalents and restricted cash at end of year |
| $ | 3,836 |
|
| $ | 3,064 |
|
Supplemental cash flow disclosures |
|
|
|
|
|
|
|
|
Income taxes paid |
| $ | 12 |
|
| $ | 31 |
|
Effect on retained earnings of adopting ASU No. 2016-09 in 2017 |
|
| — |
|
|
| 28 |
|
Beneficial conversion feature discount associated with 2018 Note |
|
| 1,007 |
|
|
| — |
|
Relative fair value of warrant issued with 2018 Note |
|
| 743 |
|
|
| — |
|
Beneficial conversion feature discount associated with 2017 Note modification |
| $ | 40 |
|
| $ | — |
|
The accompanying notes are an integral part of these consolidated financial statements.
F-6
GI Dynamics, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
1. Nature of Business
GI Dynamics® is a clinical stage medical device company focused on the development and commercialization of EndoBarrier, a medical device intended for treatment of patients with type 2 diabetes and obesity.
Diabetes mellitus type 2 (also known as type 2 diabetes) is a long-term progressive metabolic disorder characterized by high blood sugar, insulin resistance, and reduced insulin production. According to the Centers for Disease Control and Prevention (CDC), people with type 2 diabetes represent 90% of the worldwide diabetes population, whereas 10% of this population is diagnosed with type 1 diabetes (a form of diabetes mellitus in which not enough insulin is produced).
Being overweight is a condition where the patient’s body mass index (BMI) is greater than 25 (kg/m2); obesity is a condition where the patient’s BMI is greater than 30, according to the CDC. Obesity and its comorbidities contribute to the progression of type 2 diabetes. Many experts believe obesity contributes to higher levels of insulin resistance, which creates a feedback loop that increases the severity of type 2 diabetes.
When considering treatment for type 2 diabetes, it is optimal to address obesity concurrently with diabetes.
EndoBarrier® is intended for the treatment of type 2 diabetes and obesity in a minimally invasive and reversible manner.
The current treatment paradigm for type 2 diabetes is lifestyle therapy combined with pharmacological treatment, whereby treating clinicians prescribe a treatment regimen of one to four concurrent medications that could include insulin for patients with higher levels of blood sugar. Insulin carries a significant risk of increased mortality and may contribute to weight gain, which in turn may lead to higher levels of insulin resistance and increased levels of blood sugar. Fewer than 50% of patients treated pharmacologically for type 2 diabetes are adequately managed, meaning that medication does not lower blood sugar adequately and does not halt the progressive nature of diabetes of these patients.
The current pharmacological treatment algorithms for type 2 diabetes fall short of ideal, creating a large and unfilled treatment gap.
Our vision is to make EndoBarrier the essential nonpharmacological and non-anatomy-altering treatment for patients with type 2 diabetes and obesity. We intend to achieve this vision by providing a safe and effective device, focusing on optimal patient care, supporting treating clinicians, adding to the extensive body of clinical evidence around EndoBarrier, gaining appropriate regulatory approvals, continuing to improve our products and systems, operating the company in a lean fashion, and maximizing shareholder value.
EndoBarrier® is intended for the treatment of type 2 diabetes and obesity in a minimally invasive and reversible manner and is designed to mimic the mechanism of action of duodenal-jejunal exclusion created by gastric bypass surgery.
Since incorporation, the Company has devoted substantially all of its efforts to product commercialization, research and development, business planning, recruiting management and technical staff, acquiring operating assets, and raising capital. The Company currently operates in one reportable business segment.
In 2011, the Company began commercial sales of its product, the EndoBarrier, which was approved and commercially available in multiple countries outside the U.S. at the time.
In the U.S., the Company received approval from the Food and Drug Administration (“FDA”), to commence its pivotal trial of EndoBarrier (the “ENDO Trial”), which the Company began in 2013 and announced the decision to stop the ENDO Trial in the second half of fiscal year 2015. The Company announced on August 21, 2015 that as a result of stopping the trial, it was reducing headcount by approximately 46% as part of its efforts to restructure its business and expenses and to ensure sufficient cash remained available for it to establish new priorities, continue limited market development and research, and to evaluate strategic options.
F-7
In the second and third quarters of fiscal year 2016, the Company took additional actions that it thought necessary to allow the opportunity to evolve its strategic options. These actions resulted in non-recurring charges totaling approximately $1.1 million, including $0.4 million related to restructuring charges in our second quarter, $0.6 million related to employee departures in both our second and third quarters and $0.1 million related to abandonment of our former headquarters in Lexington, MA.
In October 2016, the Company received final cancellation notification from the Therapeutic Goods Administration (“TGA”) for the listing of EndoBarrier on the Australian Register of Therapeutic Goods (“ARTG”).
In May 2017, the Company received notification from its notified body, SGS United Kingdom Limited (“SGS”), that the CE Mark for EndoBarrier had been suspended pending closure of non-conformances related to its quality management system required under International Organization for Standardization (“ISO”) regulations.
On November 10, 2017, the Company received notification from SGS that SGS was withdrawing the Certificate of Conformance for EndoBarrier, ending the CE Marking of EndoBarrier in Europe and select Middle East countries.
In December 2017, the Company received notification from the Medicines and Healthcare Products Regulatory Agency (“MHRA”) that all EndoBarrier delivery systems (liners) in inventory needed to be returned to the Company.
In August 2018, the Company received approval of an IDE from the FDA to begin enrollment in a pivotal trial evaluating the safety and efficacy of EndoBarrier in the United States pending IRB approval, which was received in February 2019.
From its inception in 2003 to its initial public offering (“IPO”) in 2011, the Company was financed by a series of preferred stock financings. In September 2011, the Company completed its IPO of common stock in the form of CHESS Depositary Interests (“CDIs”) in Australia. As a result of the IPO and simultaneous private placement in the U.S., the Company raised a total of approximately $72.5 million in proceeds, net of expenses and repayment of $6.0 million of the Company’s Convertible Term Promissory Notes. Additionally, in July and August 2013, the Company issued CDIs on the Australian Securities Exchange (“ASX”) through a private placement and Share Purchase Plan (“SPP”), which raised a total of approximately $52.5 million, net of expenses. In May 2014, the Company raised an additional total of approximately $30.8 million, net of expenses, when it issued CDIs on the ASX through a private placement.
On December 20, 2016, the Company completed a private placement issue of 69,865,000 CDIs (1,397,300 shares) at an issue price of $0.022 per CDI raising approximately $1.0 million, net of issuance costs. In January 2017, the Company completed the issue of 12,481,600 CDI’s (249,632 shares) to eligible investors under a Security Purchase Plan for approximately $0.83 per share (A$0.022 per CDI) resulting in net proceeds after issuance costs of approximately $0.2 million.
In June 2017, the Company completed a Convertible Term Promissory Note (the “2017 Note”) secured financing with its largest shareholder Crystal Amber for a gross amount of $5.0 million that accrues interest at 5% per annum compounded annually. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. The 2017 Note was due by December 31, 2018 and contains provisions for conversion during its term and is also subject to security arrangements in favor Crystal Amber (See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
In January and March 2018, the Company raised approximately $1.6 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom.
In May 2018, the Company completed a Convertible Term Promissory Note (the “2018 Note”) and Warrant (the “2018 Warrant”) financing with its largest shareholder Crystal Amber for a gross amount of $1.75 million that accrues interest at 10% per annum compounded annually. The Note and Warrant financing was approved in a vote of the shareholders of GI Dynamics during the Annual Meeting of Stockholders held on May 24, 2018. The 2018 Note matures and the 2018 Warrant expires on May 30, 2023. Crystal Amber is deemed a Related Party of the Company for ASX purposes due to the size of its ownership position. (See Note 10 of the Consolidated Financial Statements for a more complete description of the terms and conditions).
F-8
In September 2018, the Company received commitments for a private placement of approximately $5 million in an offering of its CDIs to sophisticated and professional investors, including certain existing investors, in Australia, the United States and the United Kingdom. The first tranche of $2.2 million closed and cash was received in September 2018. The second and final tranche of $2.8 million was contingent upon shareholder approval which was received in October 2018. Cash proceeds were received in November 2018.
In December 2018, the maturity of the 2017 note was extended to March 31, 2019 in exchange for payment of $394 thousand which was the total accrued interest on the 2017 Note at December 31, 2018.
Going Concern Evaluation
As of December 31, 2018, the Company’s primary source of liquidity is its cash and cash equivalents balances. We are currently focused primarily on our pivotal trial, which will support future regulatory submissions and potential commercialization activities. Until we are successful in gaining regulatory approvals, we are unable to sell our product in any market at this time. Without revenues, we are reliant on funding obtained from investment in the Company to maintain our business operations until we can generate positive cash flows from operations. We cannot predict the extent of our future operating losses and accumulated deficit, and we may never generate sufficient revenues to achieve or sustain profitability.
The Company has incurred operating losses since inception and at December 31, 2018, had an accumulated deficit of approximately $267 million and a working capital deficit of $3.4 million. GI Dynamics expects to incur significant operating losses for the next several years. At December 31, 2018 the Company had approximately $3.8 million in cash and cash equivalents.
The Company will need to raise additional capital and restructure the terms of the 2017 Note before March 31, 2019 in order to continue to pursue its current business objectives as planned and to continue to fund its operations. The Company is looking to raise additional funds through any combination of additional equity and debt financings or from other sources. However, the Company has no guarantee that the 2017 Note will not mature on March 31, 2019 and has no guaranteed source of capital that will sustain operations into the second quarter of 2019. There can be no assurance that any such potential financing opportunities will be available on acceptable terms, if at all. If the Company is unable to raise sufficient capital on the Company’s required timelines and on acceptable terms to shareholders and the Board of Directors, it could be forced to cease operations, including activities essential to support regulatory applications to commercialize EndoBarrier. If access to capital is not achieved in the near term, it will materially harm the Company’s business, financial condition and results of operations to the extent that the Company may be required to cease operations altogether, file for bankruptcy, or undertake any combination of the foregoing. In such event, our shareholders may lose their entire investment in our company.
These factors raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the realization of assets and liabilities and commitments in the normal course of business. The consolidated financial statements for the year ended December 31, 2018 do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from uncertainty related to the Company’s ability to continue as a going concern within one year after the date that the financial statements are issued.
2. Summary of Significant Accounting Policies and Basis of Presentation
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of GI Dynamics, Inc. and its wholly owned subsidiaries. All intercompany transactions and balances are eliminated in consolidation.
F-9
The functional currency of GID Europe Holding B.V., GID Europe B.V., GID Germany GmbH and GI Dynamics Australia Pty Ltd is the U.S. dollar. Consolidated balance sheet accounts of the Company’s subsidiaries are remeasured into U.S. dollars using the exchange rate in effect at the consolidated balance sheet date while expenses are remeasured using the average exchange rate in effect during the period. Gains and losses arising from remeasurement of the wholly owned subsidiaries’ financial statements are included in the determination of net loss.
Use of Estimates
The preparation of consolidated financial statements in accordance with generally accepted accounting principles in the U.S. requires the Company’s management to make estimates and judgments that may affect the reported amounts of assets, liabilities, revenues and expenses, and the related disclosure of contingent assets and liabilities. On an ongoing basis, the Company’s management evaluates its estimates, including those related to revenue recognition, inventory valuation including reserves for excess and obsolete inventory, impairment of long-lived assets, income taxes including the valuation allowance for deferred tax assets, research and development, contingencies, valuation of warrant liabilities, estimates used to assess its ability to continue as a going concern and stock-based compensation. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable, the results of which form the basis for making judgments about the carrying values of assets and liabilities. Actual results may differ from these estimates under different assumptions or conditions. Changes in estimates are reflected in reported results in the period in which they become known.
Cash, Cash Equivalents and Restricted Cash
The Company considers all highly liquid investment instruments with an original maturity when purchased of three months or less to be cash equivalents. Investments qualifying as cash equivalents primarily consist of money market funds and have a carrying amount that approximates fair value. The amount of cash equivalents included in cash and cash equivalents was approximately $1.1 million and $2.6 million at December 31, 2018 and 2017, respectively.
At December 31, 2018 and 2017, the Company had approximately $0.01 million and $0.3 million, respectively, of cash and cash equivalents denominated in Australian dollars or Euros that is subject to foreign currency gain and loss.
The Company has $30 thousand in restricted cash used to secure a corporate credit card account.
Inventory
We state inventory at the lower of first-in, first-out cost or net realizable value. We record a provision for excess, expired, and obsolete inventory based primarily on estimates of forecasted revenues. A significant change in the timing or level of demand for products as compared to forecasted amounts may result in recording additional provisions for excess, expired, and obsolete inventory in the future. When capitalizing inventory, we consider factors such as status of regulatory approval, alternative use of inventory, and anticipated commercial use of the product.
The valuation of inventory also required us to estimate obsolete or excess inventory. We maintained reserves for excess and obsolete inventory based on forecasted product sales, new product introductions by us or our competitors, product expirations and historical experience. The inventory reserves we recognized were based on estimates of how these factors were expected to impact the amount and value of inventory we expected to sell. Forecasting demand for EndoBarrier in a market in which there are few, if any, comparable approved devices and for which reimbursement from third-party payers is limited. We fully reserved our inventory as of December 31, 2017 and subsequently wrote off all inventory and reserves in 2018. There is no inventory or reserves against inventory on the balance sheet at December 31, 2018.
F-10
Property and equipment, including leasehold improvements, are recorded at cost and are depreciated when placed in service using the straight-line method based on their estimated useful lives as follows:
| Estimated Useful Life (In Years) |
| ||
Laboratory and manufacturing equipment |
|
| 5 |
|
Computer equipment and software |
|
| 3 |
|
Office furniture and equipment |
|
| 5 |
|
Included in property and equipment are certain costs of software obtained for internal use. Costs incurred during the preliminary project stage are expensed as incurred, while costs incurred during the application development stage are capitalized and amortized over the estimated useful life of the software. The Company also capitalizes costs related to specific upgrades and enhancements when it is probable the expenditures will result in additional functionality. Maintenance and training costs related to software obtained for internal use are expensed as incurred.
Leasehold improvements are amortized over the shorter of the estimated useful life of the asset or the remaining lease term. Costs for capital assets not yet placed into service have been capitalized as construction in progress and will be depreciated in accordance with the above guidelines once placed into service. Maintenance and repair costs are expensed as incurred.
Revenue Recognition
We had no revenues for the year ended December 31, 2018. For the year ended December 31, 2017, we had revenues of $53 thousand of sales of EndoBarrier directly to health care providers or to third-party distributors who resell to health care providers. The adoption of ASC 606 requires us to follow a multi-step process to determine the elements of the implied contract with the customer when we sell and support our products. The steps we follow are to:
| o | Identify the implied contract with the customer: We recognize a contract exists with a customer when an authorized purchase order is submitted, placing a binding order with the Company such that transfer of the product and/or services, including any implied performance obligations on the part of the Company, goods sold (“G/S”) and the expectation to receive consideration for the G/S. |
| o | Identify the explicit and implicit performance obligations within the contract: Our G/S offering includes an underlying product and closely-related ongoing technical support for the product and its use. Assurance warranties offering a right of return for defective products are excluded from the contractual performance obligations under ASC 606. Technical support, as the remaining performance obligations does not have standalone value as it is only available for our product and only to those who have purchased and are using our product. |
| o | Determine the transaction price: The new guidance defines the transaction price as the expected amount to be received for G/S. This includes probability and magnitude of customer discounts, any expected collection risk, and any additional factors which may reduce the ultimate amount expected to be received. Estimates of these expected amounts will be based on established pricing and historical patterns once captured and management judgement until sufficient historical data is available. |
| o | The transaction price is allocated to the product and performance obligations of the contract: The allocation price will be allocated to the various G/S components, especially to any performance obligations that remain after product delivery or product implantation. In our current model, the transfer of title of the product occurs on customer receipt of shipment; product invoicing will occur after shipment and collectability expectations will be confirmed or adjusted after a sufficient period to gauge likelihood of eventual collection; and the technical support is present from shipment and is discharged after successful EndoBarrier sleeve retrieval (approximately one year after successful implantation). |
| o | As the performance obligations are discharged, the associated allocated portion of the revenue will be recognized. In our current model, we expect to book a) revenue associated with the product allocation on shipment of the product, b) revenue associated with any true up/adjustment to collectability as payment is received or when non-payment is probable, and c) revenue associated with technical support is discharged when the EndoBarrier is successfully retrieved from the patient. |
F-11
Shipping and handling costs are included in costs of revenue as they are not a performance obligation of the contract with the customer as shipping is our expense and occurs prior to title transfer of the product.
Research and Development Costs
Research and development costs are expensed when incurred. Research and development costs include costs of all basic research activities as well as other research, engineering, and technical effort required to develop a new product or service or make significant improvement to an existing product or manufacturing process. Research and development costs also include preapproval regulatory and clinical trial expenses.
Patent Costs
The Company expenses as incurred all costs, including legal expenses, associated with obtaining patents until the patented technology becomes feasible. All costs incurred after the patented technology is feasible will be capitalized as an intangible asset. As of December 31, 2018, no such costs had been capitalized since inception of the Company. The Company has expensed approximately $0.2 million and $0.4 million of patent costs within general and administrative expenses in the consolidated statements of operations for each year ended December 31, 2018 and 2017.
Stock-Based Compensation
We account for stock-based compensation in accordance with the Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, 718, Stock Compensation, or ASC 718, which requires that stock-based compensation be measured and recognized as an expense in the financial statements and that such expense be measured at the grant date fair value.
For awards that vest based on service conditions, we use the straight-line method to allocate compensation expense to reporting periods. The grant date fair value of options granted is calculated using the Black-Scholes option pricing model, which requires the use of subjective assumptions including volatility, expected term and the fair value of the underlying Common Stock, among others.
The assumptions used in determining the fair value of stock-based awards represent management’s best estimates, but these estimates involve inherent uncertainties and the application of management judgment. As a result, if factors change, and we use different assumptions, our stock-based compensation could be materially different in the future. The risk-free interest rate used for each grant is based on a zero-coupon U.S. Treasury instrument with a remaining term similar to the expected term of the stock-based award. Because we do not have a sufficient history to estimate the expected term, we use the simplified method for estimating the expected term. The simplified method is based on the average of the vesting tranches and the contractual life of each grant. Because there was no public market for our Common Stock prior to our IPO, we lacked company-specific historical and implied volatility information. We do have approximately 6 years of historical price volatility since our IPO in September 2011. Therefore, we estimate our expected stock volatility based on a combination of our to-date historical price volatility and that of publicly-traded peer companies. For purposes of identifying publicly-traded peer companies, we selected publicly-traded companies that develop, manufacture, and market medical devices, have operating businesses in the design and development of products that focus in the treatment of diabetes, and have sufficient trading history to derive a historic volatility rate. We expect to rely more heavily on our own price volatility in the future. We have not paid and do not anticipate paying cash dividends on our shares of Common Stock; therefore, the expected dividend yield is assumed to be zero.
The Company elected to adopt ASU 2016-09 in the first quarter of 2017 retrospectively to January 1, 2017. As a result of adopting ASU No. 2016-09 during the year ended December 31, 2017, the Company adjusted its accumulated deficit related to the accounting policy election to recognize the impact of share-based award forfeitures only as they occur rather than by applying an estimated forfeiture rate as previously required. ASU No. 2016-09 requires that this change be applied using a modified-retrospective transition method by means of a cumulative-effect adjustment to accumulated deficit as of the beginning of the fiscal year in which the guidance is adopted. As a result of this adoption, the Company recorded a decrease to accumulated deficit of approximately $28 thousand with an offset to Additional Paid in Capital as of January 1, 2017.
F-12
We periodically issue performance-based awards. For these awards, vesting will occur upon the achievement of certain milestones. When achievement of the milestone is deemed probable, we expense the compensation of the respective stock award over the implicit service period.
Stock awards to non-employees are accounted for in accordance with ASC 505-50, Equity-Based Payments to Non-Employees (“ASC 505-50”). The measurement date for non-employee awards is generally the date performance of services required from the non-employee is complete. For non-employee awards that vest based on service conditions, the Company expenses the value of the awards over the related service period, provided they expect the service condition to be met. The Company records the expense of services rendered by non- employees based on the estimated fair value of the stock option using the Black-Scholes option pricing model over the contractual term of the non-employee. The fair value of unvested non-employee awards is remeasured at each reporting period and expensed over the vesting term of the underlying stock options on a straight-line basis.
Impairment of Long-Lived Assets
The Company regularly reviews the carrying amount of its long-lived assets to determine whether indicators of impairment may exist that warrant adjustments to carrying values or estimated useful lives. If indications of impairment exist, projected future undiscounted cash flows associated with the asset are compared to the carrying amount to determine whether the asset’s value is recoverable. If the carrying value of the asset exceeds such projected undiscounted cash flows, the asset will be written down to its estimated fair value.
Loss Contingencies
In accordance with ASC 450, Contingencies, the Company accrues anticipated costs of settlement, damages, and losses for loss contingencies based on historical experience or to the extent specific losses are probable and estimable. Otherwise, the Company expenses these costs as incurred. If the estimate of a probable loss is a range, and no amount within the range is more likely, the Company accrues the minimum amount of the range.
Fraudulent Diversion of Funds and Related Insurance Proceeds
In July 2018, after a third-party investigation isolated the activity, an insurance claim was filed for $271 thousand for fraudulent diversion of cash from the Company’s account into a personal account that occurred during the years 2016 through 2018. In 2017, an entry aggregated and reclassified the fraudulent expenses totaling $170 thousand from operating expenses to the other income account entitled income (expense) from insurance proceeds. Expense in the year ended December 31, 2017 represents fraudulent activity comprised of $37 thousand of research and development, $10 thousand of sales and marketing and $123 thousand of general and administrative expense and income in the year ended December 31, 2018 represents net insurance proceeds on the claim less $22 thousand of research and development expense. The Company has since implemented internal controls to correct the identified control deficiencies associated with the fraud.
Income Taxes
The Company provides for income taxes under the liability method. The Company records deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the Company’s financial reporting and the tax bases of assets and liabilities measured using the enacted tax rates expected to be in effect in the years in which the differences are expected to reverse. Deferred tax assets are reduced by a valuation allowance to reflect the uncertainty associated with their ultimate realization.
The Company accounts for uncertain tax positions recognized in the consolidated financial statements by applying a more-likely-than-not threshold for financial statement recognition and measurement of a tax position taken or expected to be taken in a tax return.
Guarantees
The Company has identified the guarantees described below as disclosable, in accordance with ASC 460, Guarantees.
F-13
As permitted under Delaware law, the Company indemnifies its officers and directors for certain events or occurrences while the officer or director is, or was, serving at the Company’s request in such capacity. The maximum potential amount of future payments the Company could be required to make is unlimited; however, the Company has directors’ and officers’ insurance coverage that should limit its exposure and enable it to recover a portion of any future amounts paid.
The Company is a party to a number of agreements entered into in the ordinary course of business that contain typical provisions that obligate the Company to indemnify the other parties to such agreements upon the occurrence of certain events. Such indemnification obligations are usually in effect from the date of execution of the applicable agreement for a period equal to the applicable statute of limitations. The aggregate maximum potential future liability of the Company under such indemnification provisions is uncertain.
As of December 31, 2018, and 2017, the Company had not experienced any material losses related to these indemnification obligations, and no material claims with respect thereto were outstanding. The Company does not expect significant claims related to these indemnification obligations and, consequently, concluded that the fair value of these obligations is negligible. As a result, no related reserves have been established.
Subsequent Events
The Company evaluates events occurring after the date of its consolidated balance sheet for potential recognition or disclosure in its consolidated financial statements. There have been no subsequent events that occurred through the date the Company issued its consolidated financial statements that require disclosure in or adjustment to its consolidated financial statements.
New Accounting Pronouncements
In May 2014, the FASB issued Accounting Standards Update (“ASU”) No. 2014-09 (“ASU 2014-09”), Revenue from Contracts with Customers, which supersedes the revenue recognition requirements in ASC Topic 605, Revenue Recognition, and most industry-specific guidance. The new standard requires that an entity recognize revenue to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. ASU 2014-09 also requires additional disclosure about the nature, amount, timing and uncertainty of revenue and cash flows arising from customer contracts, including significant judgments and changes in judgments and assets recognized from costs incurred to obtain or fulfill a contract. In July 2015, ASU 2015-14 announced a one-year deferral of the effective date of this standard to annual reporting periods, and interim reporting periods within those years, beginning after December 15, 2017. Early adoption is permitted to the original effective date of December 15, 2016, including interim reporting periods within those years. Subsequently, the FASB issued further implementation refinements, clarification, and guidance in the forms of ASU 2016-08, Revenue from Contracts with Customers-Principal versus Agent Considerations (Reporting Revenue Gross versus Net); ASU 2016-10, Revenue from Contracts with Customers-Identifying Performance Obligations and Licensing; ASU 2016-12, Revenue from Contracts with Customers-Narrow-Scope Improvements and Practical Expedients; ASU 2016-20, Technical Corrections and Improvements to Topic 606, Revenue from Contracts with Customers; and ASU 2017-05 Other Income—Gains and Losses from the Derecognition of Nonfinancial Assets-Clarifying the Scope of Asset Derecognition Guidance and Accounting for Partial Sales of Nonfinancial Assets. The refined adoption period for this series of updates is for fiscal years beginning after December 15, 2017. The Company adopted this standard effective January 1, 2018 using the modified retrospective method. The adoption of this guidance did not have a significant impact on the Company’s consolidated financial statements.
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842), or ASU 2016-02. ASU 2016-02 requires that lessees recognize in the statement of financial position for all leases (with the exception of short-term leases) a lease liability, which is a lessee’s obligation to make lease payments arising from a lease, measured on a discounted basis, and a right-of-use asset, which is an asset representing the lessee’s right to use the underlying asset for the lease term. ASU 2016-02 is effective for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years, with early adoption permitted. Lessees must apply a modified retrospective transition approach for leases existing at, or entered into after, the beginning of the earliest comparative period presented in the financial statements. The modified retrospective approach would not require any transition accounting for leases that expired before the earliest comparative period presented. Lessees may not apply a full retrospective transition approach. We have elected not to apply the guidance to short-term leases and the adoption of these Accounting Standards has no impact on our consolidated financial statements for the periods ended December 31, 2017 and 2018.
F-14
In August 2016, the FASB issued ASU No. 2016-15, Classification of Certain Cash Receipts and Cash Payments (a consensus of the Emerging Issues Task Force), or ASU 2016-15. The amendments in ASU 2016-15 address eight specific cash flow issues and apply to all entities that are required to present a statement of cash flows under FASB Accounting Standards Codification 230, Statement of Cash Flows. The amendments in ASU 2016-15 are effective for public business entities for fiscal years beginning after December 15, 2017, and interim periods within those fiscal years. Early adoption is permitted, including adoption during an interim period. The Company adopted this standard effective January 1, 2018. The adoption of ASU No. 2016-15 has no impact on our consolidated financial statements for period ending December 31, 2018.
In November 2016, the FASB issued ASU No. 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash (a consensus of the FASB Emerging Issue Task Force), or ASU 2016-18. This new standard addresses the diversity that exists in the classification and presentation of changes in restricted cash on the statement of cash flows. The amendments in ASU 2016-18 require that a statement of cash flows explain the change during the period in the total of cash, cash equivalents, and amounts generally described as restricted cash or restricted cash equivalents. Therefore, amounts generally described as restricted cash and restricted cash equivalents should be included with cash and cash equivalents when reconciling the beginning-of-period and end-of-period total amounts shown on the statement of cash flows. This guidance is effective for fiscal years beginning after December 15, 2017, including interim periods within the year of adoption, with early adoption permitted. The Company adopted this standard effective January 1, 2018 and retrospectively adjusted the Statement of Cash Flows for the period ended December 31, 2017. Adoption of ASU 2016-18 does not have a material impact on our consolidated financial statements for the period ended December 31, 2018.
In May 2017, the FASB issued ASU No. 2017-09, Compensation—Stock Compensation (Topic 718): Scope of Modification Accounting. This new standard addresses the diversity that exists in practice and reduces cost and complexity of applying the guidance in Topic 718, Compensation – Stock Compensation in situations when a hare-based payment award terms are modified. This guidance is effective for fiscal years beginning after December 15, 2017, including interim periods within the year of adoption, with early adoption permitted. The Company adopted this standard effective January 1, 2018. Adoption of ASU 2017-09 does not have a material impact on our consolidated financial statements for the period ended December 31, 2018.
In July 2017, the FASB issued ASU No. 2017-11, Earnings Per Share (ASC 260) Distinguishing Liabilities from Equity (ASC 480); Derivatives and Hedging (ASC 815, which addresses the complexity of accounting for certain financial instruments with down round features. The Company adopted ASU 2017-11 during the three months ended June 30, 2018. The impact is reflected in the financial statements.
In September 2017, the FASB issued ASU No. 2017-13, Revenue Recognition (Topic 605), Revenue from Contracts with Customers (Topic 606), Leases (Topic 840), and Leases (Topic 842) - Amendments to SEC Paragraphs Pursuant to the Staff Announcement at the July 20, 2017 EITF Meeting and Rescission of Prior SEC Staff Announcements and Observer Comments. This new standard allows for an extension to certain public business entities to adopt the series of ASU’s defining revenue recognition and ASU 2016-02 regarding Leases. We qualify as a Public Business Entity and do not qualify for the extensions.
In June 2018, the FASB issued ASU No. 2018-07, Compensation – Stock Compensation (Topic 718), or ASU 2018-07, which provides measurement provisions and clarifications for the accounting for Non-employee Share-Based Payments (“NESBP”). Changes within the amendments include grant-date fair value measurement of awards, probability adjustment of satisfying performance requirements for payment, and the elimination of the need to reclass awards on vesting. Additionally, ASU 2018-07 amends guidance specific to non-public entities, which do not apply given our status as a public entity. ASU 2018-07 is effective for public business entities for fiscal years beginning after December 15, 2018, including interim periods within those fiscal years, with early adoption permitted but not in advance of adoption of Topic 606. Remeasurement should only be performed on liability classified awards that have not been settled as of the date of adoption or which have no established measurement date. Remeasurement will result in an adjustment to retained earnings as of the beginning of the fiscal year of adoption. Upon transition, those open awards are remeasured as of the date of adoption. The Company is currently evaluating the potential impact of ASU 2018-07 and expect adoption to have no material impact on its consolidated financial statements.
In July 2018, the FASB issued ASU No. 2018-09, Codification Improvements, or ASU 2018-09, which affects a wide variety of topics, including the following. Amendments to Subtopic 220-10, Income Statement— Reporting Comprehensive Income—Overall relates to income taxes not payable in cash. Amendments to Subtopic 470-50, Debt—Modifications and Extinguishments relates to debt extinguishment and requires that the net carrying amount of
F-15
extinguished fair value elected debt equals its fair value at reacquisition and related gains or losses in other comprehensive income must be included in net income upon extinguishment of the debt. Amendments to Subtopic 480-10, Distinguishing Liabilities from Equity—Overall relates to combinations of freestanding financial instruments with non-controlling interests. Amendments to Subtopic 718-740, Compensation—Stock Compensation—Income Taxes relate to recognition timing clarification for excess tax benefits or deficiencies for compensation expense. Amendments to Subtopic 805-740, Business Combinations— Income Taxes relate to allocating tax provisions to an acquired entity. Amendments to Subtopic 815-10, Derivatives and Hedging— Overall relate to accounting for offsetting derivatives. Amendments to Subtopic 820-10, Fair Value Measurement— Overall relate to the wording with respect to how transfer restrictions effect the fair value of an asset and adds explicit wording to allow entities to measure fair value on a net basis for those portfolios in which financial assets and financial liabilities and nonfinancial instruments are managed and valued together. Amendments to Subtopic 940-405, Financial Services—Brokers and Dealers—Liabilities relate to guidance about offsetting on the balance sheet. Amendments to Subtopic 962-325, Plan Accounting—Defined Contribution Pension Plans—Investments—Other relate to plan evaluation of whether a readily determinable fair value exists to determine whether those investments may qualify for the practical expedient to measure at net asset value in accordance with Topic 820. The transition and selection of an effective date is based on the facts and circumstances of each amendment, but many of the amendments have transition guidance with effective dates for annual periods beginning after December 15, 2018, for public business entities. The Company is currently evaluating the relevance of each component and potential impact of ASU 2018-09 components on its consolidated financial statements.
In July 2018, the FASB issued ASU No. 2018-11, Leases (Topic 842), Targeted Improvements, or ASU 2018-11, which primarily provides the ability to elect not to implement a modified retrospective presentation in the financial statements during transition periods and provides guidance to lessors on contract component separation. The Company declines the election and doing so has no impact on its consolidated financial statements.
In August 2018, the FASB issued ASU No. 2018-13, Fair Value Measurement (Topic 820), Disclosure Framework—Changes to the Disclosure Requirements for Fair Value Measurement, or ASU 2018-13, which provides guidance focused on the disclosure requirements for disclosing fair value estimates, assumptions, and methodology. Removed requirements to disclose details around amount and reasoning for level 1 to level 2 transfers, timing policies for transfer between levels, and the valuation processes for level 3 fair value measurements. Modified requirements include details regarding net asset redemption restrictions and timing related to uncertainty disclosures. Added requirements to include disclosures of changes in unrealized gains and losses for recurring level 3 measurements held as of the reporting date and disclosures around the range and weighted average of significant inputs used to develop level 3 fair value measurements. These amendments are effective for all entities for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2019. The amendments on changes in unrealized gains and losses, the range and weighted average of significant unobservable inputs used to develop Level 3 fair value measurements, and the narrative description of measurement uncertainty should be applied prospectively for only the most recent interim or annual period presented in the initial fiscal year of adoption. All other amendments should be applied retrospectively to all periods presented upon their effective date. Early adoption is permitted upon issuance of this Update. An entity is permitted to early adopt any removed or modified disclosures upon issuance of this Update and delay adoption of the additional disclosures until their effective date. The Company is currently evaluating the individual components and as these are disclosure refinements, expects no impact to its consolidated financial statements on adoption.
3. Net Loss per Common Share
Basic net loss per common share is computed by dividing net loss by the weighted-average number of common shares outstanding during the period. Potential common stock equivalents are determined using the treasury stock method. For diluted net loss per share purposes, the Company excludes stock options and other stock-based awards, including shares issued as a result of option exercises but which are subject to repurchase by the Company, whose effect would be anti-dilutive from the calculation. During each of the years ended December 31, 2018 and 2017, common stock equivalents were excluded from the calculation of diluted net loss per common share, as their effect was anti-dilutive due to the net loss incurred. Therefore, basic and diluted net loss per share was the same in all periods presented.
F-16
The following potentially dilutive securities have been excluded from the computation of diluted weighted- average shares outstanding as of December 31, 2018 and 2017, as they would be anti-dilutive:
| Years Ended December 31, |
| ||||||
|
| 2018 |
|
| 2017 |
| ||
Warrants to purchase common stock |
|
| 1,972,976 |
|
|
| 28,532 |
|
Options to purchase common stock and other stock-based awards |
|
| 1,545,719 |
|
|
| 1,384,880 |
|
Total |
|
| 3,518,695 |
|
|
| 1,413,412 |
|
4. Common Stock Warrants
On May 4, 2016, the Company entered into a consulting agreement pursuant to which the consulting firm will provide strategic advisory, finance, accounting, human resources and administrative functions, including chief financial officer services, to the Company. In connection with the consulting agreement, the Company granted the consulting firm a warrant (“Consultant Warrant,” together with the IPO Warrants, the “Warrants”) to purchase up to 28,532 shares of the Company’s common stock at an exercise price per share equal to $0.64. The Consultant Warrant vests on a monthly basis over two years and has a term of five years. The Company has reserved 28,532 shares of common stock related to the Consultant Warrant. As of December 31, 2018, no Consultant Warrants had been exercised.
On May 30, 2018, the Company entered into a Note and Warrant Purchase agreement that included a warrant to purchase 97,222,200 CDIs (representing 1,944,444 shares of common stock). The exercise price is US$0.018 per CDI and the warrants can be exercised with cash or as a net exercise. The warrants are immediately exercisable on issuance and expire on May 30, 2023.
The Company accounts for the warrants under ASC 815, Derivatives and Hedging (“ASC 815”). In accordance with the guidance included in ASC 815, because the Company’s functional currency is the U.S. dollar and the exercise price of the warrants is in Australian dollars, the Company is exposed to currency exchange risk related to the warrants. As a result, the warrants are not considered indexed to the Company’s own stock, and therefore, the warrants are classified as a liability and the fair value of the warrants must be remeasured at each reporting period. At the time the warrants were issued, the Company estimated the fair value of the warrants using the Black-Scholes option pricing model. The Company remeasured the fair value of the warrants at each reporting period using current assumptions and current foreign exchange rates, with changes in value recorded as other income or expense (Note 5).
5. Fair Value Measurements
The tables below present information about the Company’s assets and liabilities that are measured at fair value on a recurring basis as of December 31, 2018 and 2017 and indicates the fair value hierarchy of the valuation techniques the Company used to determine such fair value. In general, fair values determined by Level 1 inputs utilize observable inputs such as quoted prices in active markets for identical assets or liabilities. Fair values determined by Level 2 inputs utilize data points that are either directly or indirectly observable, such as quoted prices, interest rates and yield curves. Fair values determined by Level 3 inputs utilize unobservable data points in which there is little or no market data, requiring the Company to develop its own assumptions for the asset or liability.
F-17
The following tables present the assets and liabilities the Company has measured at fair value on a recurring basis (in thousands):
|
|
|
|
|
| Fair Value Measurements at |
| |||||||||
|
|
|
|
| Reporting Date Using |
| ||||||||||
|
|
|
|
|
| Quoted Prices in |
|
| Significant Other |
|
| Significant |
| |||
|
|
|
|
|
| Active Markets for |
|
| Observable |
|
| Unobservable |
| |||
|
|
|
|
|
| Identical Assets |
|
| Inputs |
|
| Inputs |
| |||
Description |
| December 31, 2018 |
|
| (Level 1) |
|
| (Level 2) |
|
| (Level 3) |
| ||||
Assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds (included in cash and cash equivalents) |
| $ | 1,097 |
|
| $ | 1,097 |
|
| $ | — |
|
| $ | — |
|
Total assets |
| $ | 1,097 |
|
| $ | 1,097 |
|
| $ | — |
|
| $ | — |
|
Liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative liabilities |
| $ | 51 |
|
| $ | — |
|
| $ | — |
|
| $ | 51 |
|
Total liabilities |
| $ | 51 |
|
| $ | — |
|
| $ | — |
|
| $ | 51 |
|
|
|
|
|
|
| Fair Value Measurements at |
| |||||||||
|
|
|
|
| Reporting Date Using |
| ||||||||||
|
|
|
|
|
| Quoted Prices in |
|
| Significant Other |
|
| Significant |
| |||
|
|
|
|
|
| Active Markets for |
|
| Observable |
|
| Unobservable |
| |||
|
|
|
|
|
| Identical Assets |
|
| Inputs |
|
| Inputs |
| |||
Description |
| December 31, 2017 |
|
| (Level 1) |
|
| (Level 2) |
|
| (Level 3) |
| ||||
Assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Money market funds (included in cash and cash equivalents) |
| $ | 2,584 |
|
| $ | 2,584 |
|
| $ | — |
|
| $ | — |
|
Total assets |
| $ | 2,584 |
|
| $ | 2,584 |
|
| $ | — |
|
| $ | — |
|
Liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative liabilities |
| $ | 493 |
|
| $ | — |
|
| $ | — |
|
| $ | 493 |
|
Total liabilities |
| $ | 493 |
|
| $ | — |
|
| $ | — |
|
| $ | 493 |
|
The assumptions used in the Black-Scholes option pricing model to determine the fair value of the common stock warrants at December 31, 2018 and 2017 were as follows:
|
| December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Exercise price (A$55.00 at the then current exchange rate) |
| $ | 0.64 |
|
| $ | 0.64 |
|
Fair value of common stock |
| $ | 0.57 |
|
| $ | 0.60 |
|
Expected volatility |
|
| 134.0 | % |
|
| 142.6 | % |
Expected term (in years) |
| 2.35 |
|
| 3.34 |
| ||
Risk-free interest rate |
|
| 2.48 | % |
|
| 2.02 | % |
Expected dividend yield |
| — | % |
| — | % | ||
F-18
The conversion feature associated with the long-term debt to related party was valued using a financial model with unobservable inputs relating to volatility, variable outcomes and projected stock values. This model incorporated assumptions similar to those used to value the common stock warrants with the primary difference being a change in the expected term to reflect the contractual terms of the debt instrument.
The following table rolls forward the fair value of the derivatives, where fair value is determined by Level 3 inputs (in thousands):
Balance at December 31, 2016 |
| $ | 17 |
|
Initial valuation of conversion feature associated with 2017 Note |
|
| 602 |
|
Decrease in fair value of 2017 Note-related conversion feature upon re-measurement |
|
| (138 | ) |
Increase in fair value of warrants upon re-measurement |
|
| 12 |
|
Balance at December 31, 2017 |
|
| 493 |
|
Decrease in fair value of 2017 Note-related conversion feature upon re-measurement |
|
| (464 | ) |
Decrease in fair value of warrants upon re-measurement |
|
| (18 | ) |
Fair value of beneficial conversion feature discount associated with 2017 Note modification |
|
| 40 |
|
Balance at December 31, 2018 |
| $ | 51 |
|
Cash, cash equivalents, restricted cash, accounts receivable, prepaid expenses and other current assets, accounts payable, accrued expenses, short-term debt to related party and other current liabilities at December 31, 2018 and 2017 are carried at amounts that approximate fair value due to their short-term maturities and highly liquid nature of these instruments.
6. Concentrations of Credit Risk, Accounts Receivable and Related Valuation Account
Financial instruments that subject the Company to credit risk primarily consist of cash and cash equivalents, restricted cash and accounts receivable. The Company maintains its cash and cash equivalent balances with high quality financial institutions, and consequently, the Company believes that such funds are subject to minimal credit risk. The Company’s short-term investments potentially subject the Company to concentrations of credit risk. The Company has adopted an investment policy that limits the amounts the Company may invest in any one type of investment and requires all investments held by the Company to hold at least an A rating from a recognized credit rating agency, thereby reducing credit risk concentration.
Accounts receivable primarily consist of amounts due from customers, including distributors and health care providers in different countries. In light of the current economic state of many foreign countries, the Company continues to monitor the creditworthiness of its customers.
In May 2017, the Company entered into a sales arrangement with certain distributors totaling $517 thousand of EndoBarrier inventory. Due to certain extended right of return periods and payment terms, the Company determined that the revenue and related product costs associated with this transaction should be deferred for accounting purposes. As a result, the Company recorded an adjustment to accounts receivable of $559 thousand for the unpaid portion of deferred revenue which includes an adjustment of approximately $40 thousand for revaluation of receivables denominated in foreign currency at December 31, 2017. For the year ended December 31, 2017, the Company recognized revenue of $18 thousand and related costs of $27 thousand for this transaction.
The Company had no accounts receivable at December 31, 2018. At December 31, 2017, four health care providers accounted for approximately 100% of the Company’s accounts receivable balance, of approximately equal sums.
The Company grants credit to customers in the normal course of business but generally does not require collateral or any other security to support its receivables. The Company makes judgments as to its ability to collect outstanding receivables and provides an allowance for receivables when collection becomes doubtful. Provisions are made based upon a specific review of all significant outstanding invoices and the overall quality and age of those invoices not individually reviewed. In certain circumstances, the Company allows customers to return defective or nonconforming products for credit or replacement products. Defective or nonconforming products typically include those products that resulted in an unsuccessful implant procedure. The Company records an estimate for product returns based upon historical trends. The
F-19
associated reserve for product returns is recorded as a reduction of the Company’s accounts receivable. Amounts determined to be uncollectible are written off against the total reserve. The Company recorded write-offs of uncollectible accounts receivable of approximately $42 thousand in the year ended December 31, 2018 and $22 thousand in the year ended December 31, 2017. As of December 31, 2018, the Company had no accounts receivable and no reserves for uncollectible accounts receivable.
The following table shows the components of the Company’s accounts receivable at December 31, 2018 and 2017 (in thousands):
|
| December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Accounts receivable |
| $ | — |
|
| $ | 85 |
|
Less: allowance for doubtful accounts |
|
| — |
|
|
| (42 | ) |
Less: allowance for sales returns |
|
| — |
|
|
| (3 | ) |
Total |
| $ | — |
|
| $ | 40 |
|
The following is a roll forward of the Company’s allowance for doubtful accounts (in thousands):
|
| Year Ended December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Beginning balance |
| $ | 42 |
|
| $ | 16 |
|
Net charges to expenses |
|
| — |
|
|
| 48 |
|
Utilization of allowances |
|
| (42 | ) |
|
| (22 | ) |
Ending balance |
| $ | — |
|
| $ | 42 |
|
7. Inventory
The Company states inventory at the lower of first-in, first-out cost or net realizable value. The Company records a provision for excess, expired, and obsolete inventory based primarily on estimates of forecasted revenues. When capitalizing inventory, the Company considers factors such as status of regulatory approval, alternative use of inventory, and anticipated commercial use of the product.
A significant change in the timing or level of demand for products as compared to forecasted amounts may result in recording additional provisions for excess, expired, and obsolete inventory in the future. Currently, the determination of obsolete or excess inventory requires the Company to estimate regulatory approval probability and timing and subsequent demand for its products within approved markets. The estimated future demand is compared to inventory levels to determine the amount, if any, of obsolete and excess inventory. Given the probability and timing of regulatory approval and appropriate inventory life span, we fully reserved our inventory as of December 31, 2017 and subsequently wrote off all inventory and reserves in 2018 as the materials on hand were not expected to be usable for future sales. There is no inventory or reserves against inventory on the balance sheet at December 31, 2018.
F-20
Property and equipment consisted of the following (in thousands):
| December 31, |
| ||||||
|
| 2018 |
|
| 2017 |
| ||
Laboratory and manufacturing equipment |
| $ | 591 |
|
| $ | 591 |
|
Computer equipment and software |
|
| 1,182 |
|
|
| 1,179 |
|
Office furniture and equipment |
|
| 183 |
|
|
| 183 |
|
Leasehold improvements |
|
| — |
|
|
| 21 |
|
|
|
| 1,956 |
|
|
| 1,974 |
|
Less accumulated depreciation and amortization |
|
| (1,893 | ) |
|
| (1,877 | ) |
Total |
| $ | 63 |
|
| $ | 97 |
|
The Company recorded a loss on disposal of $2 thousand in connection with the expiration of its office lease and the related leasehold improvements on April 13, 2018.
At December 31, 2018 and 2017, the Company had no assets under capital lease.
Depreciation and amortization expense of property and equipment totaled approximately $35 thousand and $57 thousand for the years ended December 31, 2018 and 2017.
9. Accrued Expenses
Accrued expenses consisted of the following (in thousands):
|
| December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Payroll and related liabilities |
| $ | 386 |
|
| $ | 181 |
|
Professional fees |
|
| 573 |
|
|
| 421 |
|
Credit refunds |
|
| 186 |
|
|
| 279 |
|
Interest payable |
|
| 494 |
|
|
| 136 |
|
Other |
|
| 6 |
|
|
| 4 |
|
Total |
| $ | 1,645 |
|
| $ | 1,021 |
|
In 2017, following the notification by MHRA, the Company notified its customers to return their inventory on hand. The Company calculated an estimate for returns, reversed its revenue and recorded an accrued expense estimate of $202 thousand of product return related costs in addition to $77 thousand of credit memos granted to customers. No related activity in 2018 resulted in the amounts being unchanged and the Company still anticipates claims against these accruals in the future.
In 2017, the Company recorded $136 thousand of interest payable on the 2017 Note. In 2018, the Company recorded $359 thousand of interest payable on the 2017 and 2018 Notes. In December 2018, the maturity of the 2017 Note was extended to March 31, 2019 in exchange for payment of $394 thousand which was the total accrued interest on the 2017 Note at December 31, 2018.
10. Notes Payable
2017 Convertible Note Financing
On June 15, 2017, the Company entered into a Note Purchase Agreement by and between the Company, as borrower, and Crystal Amber Fund Limited, as purchaser (the “Purchaser”). Pursuant to the Note Purchase Agreement, the Company issued and sold to the Purchaser a Senior Secured Convertible Promissory Note in an aggregate original principal amount of $5.0 million (the “2017 Note”). The Purchaser is a related party for ASX purposes and is the Company’s largest shareholder.
F-21
The 2017 Note accrues interest at a rate equal to 5% per annum, compounded annually, other than during the continuance of an event of default, when the 2017 Note accrues interest at a rate of 8% per annum. The entire outstanding principal balance and all unpaid accrued interest thereon was initially due on the original maturity date, December 31, 2018 but was extended to March 31, 2019 in December 2018.
The 2017 Note is secured by a first priority security interest in substantially all tangible and intangible assets of the Company, including intellectual property (the “Collateral”). In the event of an uncured default, the Purchaser is authorized to sell, transfer, assign or otherwise deal in or with the collateral or the proceeds thereof or any related goods securing the Collateral, as fully and effectually as if the Purchaser were the absolute owner thereof.
The ASX provided the Company with a waiver to allow all asset liens (the “Security”) to be granted to the Purchaser without the normal requirement of having to obtain shareholder approval for the grant of a security to a related party of the Company (which the Purchaser is for ASX purposes). As a result of the waiver, the Security contains a provision that provides that if an event of default occurs and the Purchaser exercises its rights under the Security, neither the Purchaser nor any of its associates can acquire any legal or beneficial interest in an asset of the Company or its subsidiaries in full or part satisfaction of the Company’s obligations under the Security, or otherwise deal with the assets of the Company or its subsidiaries, without the Company first having complied with any applicable ASX Listing Rules, including ASX Listing Rule 10.1, other than as required by law or through a receiver, or receiver or manager (or analogous person) appointed by the Purchaser exercising its power of sale under the Security and selling the assets to an unrelated third party on arm’s length commercial terms and conditions and distributing the cash proceeds to the Purchaser or any of its associates in accordance with their legal entitlements.
The entire outstanding principal balance under the 2017 Note and all unpaid accrued interest thereon is convertible into CHESS Depositary Interests (“CDIs”), each representing 1/50th of a share of the Company’s common stock, (i) prior to the maturity date, at the option of the Purchaser at a conversion price calculated based on the five-day volume weighted average closing price of the Company’s CDIs on the ASX (“Optional Conversion Price”), or (ii) automatically upon the occurrence of an equity financing in which the Company raises at least $10 million (a “Qualified Financing”) at the price per CDI of the CDIs issued and sold in such financing.
In the event that the Borrower issues additional CDIs in a subsequent equity financing at a price per CDI that is less than the then-effective optional conversion price (based on the five-day volume weighted average price on the ASX), the Purchaser has a 30-day option to convert at an adjusted conversion price reflecting, on a weighted average basis, the lower price per CDI. The number of CDIs that the Purchaser may acquire upon conversion of the 2017 Note at this adjusted conversion price is limited to the number that maintains the Purchaser’s fully-diluted ownership percentage of the Company at the same level as existed immediately preceding the applicable subsequent equity financing.
In addition, upon a change of control of the Company (other than a change of control resulting from a Qualified Financing) in which the Company’s stockholders receive cash consideration, the Company is obligated to prepay all accrued and unpaid interest plus 110% of the remaining outstanding unconverted principal balance. If the consideration received for such change of control is a non-cash consideration, the Purchaser may convert the entire outstanding principal balance under the 2017 Note and all unpaid accrued interest thereon into CDIs at the abovementioned Optional Conversion Price. Other than as described above, the Company may not prepay the 2017 Note without the consent of the Purchaser.
The 2017 Note Purchase Agreement contains customary events of default including a failure to perform obligations under the 2017 Note Purchase Agreement, bankruptcy, a decision by the board of directors of the Company to wind up the Company, or if the Company otherwise ceases to carry on its ongoing business operations. If a default occurs and is not cured within the applicable cure period or is not waived, any outstanding obligations under the 2017 Note may be accelerated. The 2017 Note Purchase Agreement and related 2017 Note documents also contain additional representations and warranties, covenants and conditions, in each case customary for transactions of this type.
The Company recorded the $5 million 2017 Note, net of debt issuance costs of $115 thousand and amortized the debt issuance costs over the life of the 2017 Note. For the year ended December 31, 2017, the Company accrued $136 thousand of interest expense and $44 thousand in amortization of debt issuance costs related to the 2017 Note. For comparative purposes, an adjustment was made to separate the short-term debt balance and the 2017 Note derivative liability related to the value of the beneficial conversion rights. This included an adjustment of $602 thousand to record the value of the beneficial conversion rights in derivative liability and the short-term debt balance on the date of issuance and an adjustment of $138 thousand recorded as a subsequent gain in the change in derivative liability and interest expense. Additionally, for comparative purposes, the 2017 interest accrual was shown separately from the change in accrued expenses in the Statement of Cash Flows in order to conform to 2018 presentation.
F-22
For the year ended December 31, 2018, the Company accrued $257 thousand of interest expense and recorded $71 thousand in amortization of debt issuance costs and interest of $464 thousand related to the conversion rights.
Due to the timing of the finalization of the 2017 Note financing in 2017, the 2017 Note was issued without stockholder approval. As a consequence, while the 2017 Note contains conversion provisions, the Purchaser had, for a period of time, no right to exercise those rights until such rights of exercise were approved by the stockholders of the Company. Stockholder approval of the Purchaser’s right to convert the 2017 Note was obtained at the Company’s Annual Meeting on May 24, 2018.
In December 2018, the maturity of the 2017 Note was extended to March 31, 2019 in exchange for payment of $394 thousand which was the total accrued interest on the 2017 Note at December 31, 2018. Payment of this amount was made in January 2019. The modification extended the beneficial conversion rights and resulted in an additional $40 thousand of derivative liability being recorded.
2018 Convertible Note and Warrant Financing
On May 30, 2018, the Company entered into a Note Purchase Agreement by and between the Company, as borrower, and Crystal Amber Fund Limited, as purchaser (the “Purchaser”). Pursuant to the Note Purchase Agreement, the Company issued and sold to the Purchaser a Senior Unsecured Convertible Promissory Note in an aggregate original principal amount of $1.75 million (the “2018 Note”). The Purchaser is a related party and is the Company’s largest shareholder.
The 2018 Note accrues interest at a rate equal to 10% per annum, compounded annually, other than during the continuance of an event of default, when the 2018 Note accrues interest at a rate of 16% per annum. The entire outstanding principal balance and all unpaid accrued interest thereon is due on the maturity date, May 30, 2023.
The entire outstanding principal balance under the 2018 Note and all unpaid accrued interest thereon is convertible into CHESS Depositary Interests (“CDIs”), each representing 1/50th of a share of the Company’s common stock, at the option of the Purchaser at a conversion price of US$0.018 per CDI. In the event that the Borrower issues additional CDIs in a subsequent equity financing at a price per CDI that is less than US$0.018, the conversion price of the 2018 Note will adjust to the lower CDI conversion price. In addition, upon a change of control of the Company, the Purchaser may demand prepayment of accrued and unpaid interest plus 110% of the remaining outstanding unconverted principal balance of the 2018 Note.
The 2018 Note contains customary events of default including a failure to perform obligations under the 2018 Note Purchase Agreement, bankruptcy, a decision by the board of directors of the Company to wind up the Company, or if the Company otherwise ceases to carry on its ongoing business operations. If a default occurs and is not cured within the applicable cure period or is not waived, any outstanding obligations under the 2018 Note may be accelerated. The 2018 Note Purchase Agreement and related 2018 Note documents also contain additional representations and warranties, covenants and conditions, in each case customary for transactions of this type.
In connection with the issuance of the 2018 Note, the Company also issued to the Purchaser a warrant to purchase 97,222,200 CDIs at an initial exercise price of US$0.018 per CDI, subject to adjustment as described in the warrant, which warrant expires on May 30, 2023 (the “2018 Warrant”). The 2018 Warrant may be exercised at any time on a cash or cashless basis. The 2018 Warrant includes a price protection clause. If the Company issues securities in a subsequent financing at a per CDI price of less than US$0.018, the exercise price of the 2018 Warrant will be reduced to the lowest such price per CDI (or the equivalent for shares of common stock) at which the newly issued securities were sold.
The Company has evaluated the guidance ASC 480-10 Distinguishing Liabilities from Equity, ASC 815-40 Contracts in an Entity's Own Equity and ASC 470-20 Debt with Conversion and Other Options to determine the appropriate classification of the 2018 Note and 2018 Warrant. The 2018 Warrant was determined to be a freestanding instrument meeting the requirements for equity classification. Accordingly, the relative fair value estimated for the 2018 Warrant, totaling approximately $743 thousand, has been recorded as a discount to the debt with the offset to additional paid-in capital. The 2018 Note was also evaluated for beneficial conversion feature ("BCF") subsequent to the allocation of proceeds among the 2018 Note and 2018 Warrant. Based upon the effective conversion price of the 2018 Note after considering the stock price at the date of issuance and the allocation of estimated fair value to the 2018 Warrant, it was determined that the 2018 Note contained a BCF. The value of the BCF was computed to be approximately $1.2 million but has been capped at approximately $1.0 million so as to not exceed the total proceeds from the 2018 Note after deducting the value allocated to the 2018 Note and 2018 Warrant. The effective interest rate on the note after the discounts is 26.4%.
F-23
The Company recorded the 2018 Note, net of the total debt discount of $1.75 million and will amortize the debt discount over the life of the 2018 Note. For the year ended December 31, 2018, the Company recognized interest expense of $102 thousand, debt discount amortization of $168 thousand, and interest expense derived from issuance costs of $85 thousand.
11. Commitments and Contingencies
Lease Commitments
In June 2016, the Company entered into a non-cancelable agreement to lease approximately 4,200 square feet of office space in Boston, Massachusetts. The lease commenced in June 2016 and expired in April 2018. Rent during the term was $11.9 thousand per month.
In December 2018, the Company entered into a membership agreement with WeWork for 985 square feet of office space located in Boston, Massachusetts. The committed lease term expires in May 2019 and contains a two-month cancellation provision. The Company is exploring term extensions or other suitable options for office space subsequent to the current term expiry with WeWork.
Future minimum lease payments under all non-cancelable lease arrangements at December 31, 2018 are as follows (in thousands):
|
|
|
| |
2019 |
| $ | 91 |
|
2020 |
|
| — |
|
2021 |
|
| — |
|
2022 |
|
| — |
|
2023 |
|
| — |
|
Total future minimum lease payments |
| $ | 91 |
|
Rent expense on non-cancelable operating leases was approximately $0.1 million for each of the years ended December 31, 2018 and 2017, respectively.
License Agreement
In 2003, the Company entered into a license agreement (“License Agreement”) for certain intellectual property. The License Agreement required the Company to pay defined annual amounts until the date of first commercial sale of the product, which occurred in 2011. Payments were next due in the form of royalty payments when U.S. commercial sales of product commenced. U.S. commercial sales did not commence prior to the expiration of the patent in 2017, and the patent expiry eliminated future royalty obligations.
12. Stockholders’ Equity (Deficit)
On April 9, 2015, the Company amended its certificate of incorporation to reflect the one-for-ten reverse stock split approved by its shareholders.
On May 22, 2017, the Stockholders of the Company approved an increase of its authorized shares of Common Stock from 13,000,000 to 50,000,000 and to eliminate Class B shares of Common Stock of the Company. As of December 31, 2018, the authorized capital stock of the Company consists of 50,500,000 shares, of which 50,000,000 shares are designated as Common Stock and 500,000 shares are designated as Preferred Stock.
In January 2017, the Company completed the issue of 12,481,600 CDI’s (249,632 shares) to eligible investors under a Security Purchase Plan for A$0.022 per CDI (approximately US$0.83 per share) resulting in net proceeds after issuance costs of approximately $0.2 million.
F-24
In 2018, the Company received commitments for two private placements to sophisticated and professional investors in Australia, the United States and the United Kingdom, consisting of U.S. and non-U.S. persons (as defined in Regulation S (“Regulation S”) of the Securities Act of 1933 (the “Securities Act”)) to raise up to approximately $6.61 million (the “2018 Placements”). The first placement (“First Quarter 2018 Placement”) consisted of a total of 58,780,619 fully paid CDIs of the Company (representing 1,175,612 shares of common stock) at an issue price of A$0.035 per CDI. The issue of CDIs under the First Quarter 2018 Placement occurred in two tranches. The first tranche closed on January 22, 2018 (US EST), pursuant to which the Company issued 28,467,063 CDIs (representing 569,341 shares of common stock) resulting in gross proceeds of approximately $781 thousand and related issuance costs of $63 thousand. The closing of the second tranche of the First Quarter 2018 Placement resulted in the raising of $824 thousand and related issuance costs of $40 thousand by the issue of 30,313,556 CDIs (606,271 shares) following stockholder approval granted on February 27, 2018. There were two participants in the First Quarter 2018 Placement second tranche; Crystal Amber Fund, a related party for ASX purposes, purchased 27,391,756 CDIs. A Board member of the Company purchased 2,921,800 CDIs.
The second placement (“Autumn 2018 Placement”) consisted of a total of 347,222,250 fully paid CDIs of the Company (representing 6,944,445 shares of common stock) at an issue price of A$0.020 per CDI. The investors in the Autumn 2018 Placement included certain existing investors. The issue of these CDIs occurred in two tranches. The first tranche closed on September 20, 2018 (US EST), pursuant to which the Company issued 150,000,000 CDIs (representing 3,000,000 shares of common stock) resulting in gross proceeds of approximately $2.2 million and related issuance costs of $56 thousand. The closing of the second tranche resulted in the raising of $2.8 million and related issuance costs of $12 thousand by the issue of 197,222,250 CDIs (representing 3,944,445 shares of common stock) following stockholder approval at the adjourned Special Meeting of stockholders on October 29, 2018. There were three participants in the second tranche; Crystal Amber Fund, a related party for ASX purposes, purchased 168,194,450 CDIs. Existing investors in the United States and Australia also purchased 23,819,450 and 5,208,350 CDIs, respectively. All second tranche CDIs were allotted to investors in November 2018.
Common Stock
The Company had authorized Class B common stock in order to meet the Listing Rules of the ASX so far as they apply to escrowed securities. In the event that holders of common stock, who were subject to ASX-imposed escrow, breached the terms of their escrow agreement or the Listing Rules as they apply to escrowed securities, their common stock would have been automatically converted into Class B common stock until the earlier to occur of the expiration of the escrow period or the breach being rectified. The Class B common stock was identical to and ranked equally with the common stock except that Class B common stock had no voting rights and was not entitled to any dividends. Class B Common Stock of the Company was eliminated on May 22, 2017. No shares of common stock were subject to such an escrow.
13. Share-Based Compensation
The Company has two stock-based compensation plans. The Board of Directors adopted the 2003 Omnibus Stock Plan (the “2003 Plan”), which provides for the grant of qualified incentive stock options and nonqualified stock options or other awards to the Company’s employees, officers, directors, advisors, and outside consultants to purchase up to an aggregate of 922,086 shares of the Company’s common stock.
In August 2011, the Board of Directors adopted the 2011 Employee, Director and Consultant Equity Incentive Plan (the “2011 Plan”, together with the 2003 Plan, the “Plans”) as the successor to the 2003 Plan. Under the 2011 Plan, the Company may grant incentive stock options, nonqualified stock options, restricted and unrestricted stock awards and other stock-based awards. The Company had initially reserved 450,000 shares of its common stock for issue under the 2011 Plan. Awards that are returned to the Company’s 2003 Plan as a result of their forfeiture, expiration or cancellation without delivery of common stock shares or that result in the forfeiture of shares back to the Company on or after August 1, 2011, the date the 2011 Plan became effective, are automatically made available for issuance under the 2011 Plan. At August 1, 2011, 80,235 shares available for grant under the 2003 Plan were transferred to the 2011 Plan. As of December 31, 2018, an additional 1,173,917 shares of Common Stock were available for grant under our 2011 Employee, Director and Consultant Equity Incentive Plan.
In addition, the 2011 Plan allows for an annual increase in the number of shares available for issue under the 2011 Plan commencing on the first day of each fiscal year during the period beginning in fiscal year 2012 and ending in fiscal year 2020. The annual increase in the number of shares shall be equal to the lowest of:
F-25
| • | 4% of the number of common shares outstanding as of such date; and |
| • | an amount determined by the Board of Directors or the Company’s compensation committee. Accordingly, during year ended December 31, 2018 and December 31, 2017, 0 and 436,314 shares were added to the 2011 Plan, respectively. |
Stock-Based Compensation
Stock-based compensation is reflected in the consolidated statements of operations as follows for the years ended December 31, 2018 and 2017 (in thousands):
| Years Ended December 31, |
| ||||||
|
| 2018 |
|
| 2017 |
| ||
Research and development |
| $ | 6 |
|
| $ | 19 |
|
Sales and marketing |
|
| 23 |
|
|
| 75 |
|
General and administrative |
|
| 95 |
|
|
| 93 |
|
|
| $ | 124 |
|
| $ | 187 |
|
The stock options granted under the Plans generally vest over a four-year period and expire ten years from the date of grant. From time to time, the Company grants stock options to purchase common stock subject to performance-based milestones. The vesting of these stock options will occur upon the achievement of certain milestones. When achievement of the milestone is deemed probable, the Company expenses the compensation of the respective stock option over the implicit service period.
In calculating stock-based compensation costs, the Company estimates the fair value of stock options using the Black-Scholes option-pricing model. The Black-Scholes option-pricing model was developed for use in estimating the fair value of short-lived, exchange-traded options that have no vesting restrictions and are fully transferable. The Company estimates the number of awards that will be forfeited in calculating compensation costs. Such costs are then recognized over the requisite service period of the awards on a straight-line basis.
Determining the fair value of stock-based awards using the Black-Scholes option-pricing model requires the use of highly subjective assumptions, including the expected term of the award and expected stock price volatility. The weighted-average assumptions used to estimate the fair value of employee stock options using the Black-Scholes option-pricing model were as follows for the years ended December 31, 2018 and 2017:
| Years Ended December 31, |
| ||||||
|
| 2018 |
|
| 2017 |
| ||
Expected volatility |
|
| 120.7 | % |
|
| 93.3 | % |
Expected term (in years) |
|
| 6.05 |
|
|
| 6.05 |
|
Risk-free interest rate |
|
| 2.6 | % |
|
| 2.3 | % |
Expected dividend yield |
|
| 0 | % |
|
| 0 | % |
Expected Volatility
Volatility measures the amount that a stock price has fluctuated or is expected to fluctuate during a period. As the Company was not publicly traded prior to September 2011 and therefore had no trading history, stock price volatility was estimated based on an analysis of historical and implied volatility of comparable public companies.
Expected Term
The Company has limited historical information to develop reasonable expectations about future exercise patterns and post-vesting employment termination behavior for its stock option grants. As a result, for stock option grants made during the years ended December 31, 2018 and 2017, the expected term was estimated using the “simplified method.” The simplified method is based on the average of the contractual term of the option and the weighted-average vesting period of the option. For options granted to non-employees, the Company used the remaining contractual life to estimate the expected term of non-employee awards for the years ended December 31, 2018 and 2017.
F-26
The risk-free interest rate used for each grant is based on a zero-coupon U.S. Treasury instrument with a remaining term similar to the expected term of the stock-based award.
Expected Dividend Yield
The Company has not paid and does not anticipate paying cash dividends on its shares of common stock in the foreseeable future; therefore, the expected dividend yield is assumed to be zero.
Stock Options
The following table summarizes share-based activity under the Company’s stock option plans:
| Shares of Common Stock Attributable to Options |
|
| Weighted- Average Exercise Price |
|
| Weighted- Average Contractual Life |
| Aggregate Intrinsic Value |
| ||||
|
|
|
|
|
|
|
|
|
| (in years) |
| (in thousands) |
| |
Outstanding at December 31, 2017 |
|
| 992,221 |
|
| $ | 4.88 |
|
| 7.57 |
| $ | — |
|
Granted |
|
| 472,500 |
|
| $ | 0.78 |
|
| 9.83 |
|
|
|
|
Exercised |
| — |
|
|
|
|
|
|
|
|
|
|
| |
Cancelled |
|
| (479,497 | ) |
| $ | 5.84 |
|
| 7.34 |
| $ | 25 |
|
Outstanding at December 31, 2018 |
|
| 985,224 |
|
| $ | 2.24 |
|
| 8.48 |
| $ | — |
|
Exercisable at December 31, 2018 |
|
| 344,603 |
|
| $ | 4.87 |
|
| 7.11 |
| $ | — |
|
Restricted Stock Units & Performance Stock Units
Each restricted stock unit and performance stock unit (“RSU & PSU”) represents a contingent right to receive one share of the Company’s common stock. There is no consideration payable on the vesting of RSUs & PSUs issued under the Plans. Upon vesting, the RSUs & PSUs are exercised automatically and settled in shares of the Company’s common stock. During the years ended December 31, 2018 and 2017, the Company awarded no RSUs & PSUs, to employees and directors of the Company. Due to the resignation of an employee, one grant of 142,659 RSU & PSU was cancelled in 2018.
The following table summarizes information related to RSU & PSU activity during the year ended December 31, 2018:
| Number of Units |
|
| Weighted- Average Contractual Life |
|
| Aggregate Intrinsic Value |
| ||||
|
|
|
|
|
| (in years) |
|
| (in thousands) |
| ||
Outstanding at December 31, 2017 |
|
| 392,659 |
|
|
| 8.27 |
|
| $ | 429 |
|
Granted |
|
|
|
|
|
|
|
|
|
|
|
|
Exercised |
|
|
|
|
|
|
|
|
|
|
|
|
Cancelled |
|
| (142,659 | ) |
|
| 7.99 |
|
| $ | 187 |
|
Outstanding at December 31, 2018 |
|
| 250,000 |
|
|
| 7.23 |
|
| $ | 141 |
|
The aggregate intrinsic value at December 31, 2018 and 2017 noted in the table above represents the closing price of the Company’s common stock multiplied by the number of RSUs & PSUs outstanding. The fair value of each RSU & PSU award equals the closing price of the Company’s common stock on the date of grant.
At December 31, 2018, 250,000 of the RSUs & PSUs outstanding are subject to performance-based vesting criteria. For these awards, the vesting will occur upon the achievement of certain milestones. When achievement of the milestone
F-27
is deemed probable, the Company will expense the compensation of the respective stock award over the implicit service period.
During the years ended December 31, 2018 and 2017, the Company recognized no stock-based compensation related to RSUs & PSUs performance-based vesting.
As of December 31, 2018, there was approximately $144 thousand of unrecognized stock-based compensation expense related to non-vested RSU & PSU awards that have service-based vesting.
Non-employee awards
The Company accounts for non-employee awards in accordance with ASC 505-50. Stock-based compensation expense related to stock options granted to non-employees is recognized as services are rendered, generally on a straight-line basis. The Company believes that the fair value of the stock options is more reliably measurable than the fair value of the services rendered. The fair value of the stock options granted is remeasured at each reporting date using the Black-Scholes option pricing model as prescribed by ASC 718. During the year ended December 31, 2018, the Company did not grant options to purchase shares of common stock to non-employees. During the year ended December 31, 2017, the Company granted options to purchase 115,500 shares of common stock to non-employees with an aggregate fair value of approximately $171 thousand of which 46,500 shares with an aggregate fair value of approximately $78 thousand were cancelled during year ending December 31, 2017.
The Company has recorded non-employee stock-based compensation expense in accordance with ASC 505-50 of approximately $20 thousand and $34 thousand during the years ended December 31, 2018 and 2017, respectively, which is included in the total stock-based compensation expense.
14. Segment Reporting
The Company has one reportable segment which designs, develops, manufactures and markets medical devices for non-surgical approaches to treating type 2 diabetes and obesity.
Geographic Reporting
All the Company’s revenue was attributable to customers outside the U.S. The Company is dependent on favorable economic and regulatory environments for its products. Products were sold to customers located in Europe, the Middle East, and the Asia Pacific region and sales were attributed to a country or region based on the location of the customer to whom the products were sold. There were no product sales in 2018.
At December 31, 2018 and 2017, all long-lived assets comprised of property and equipment are held in the U.S.
Product sales by geographic location for the years ended December 31, 2018 and 2017 are listed in the table below (in thousands).
15. Retirement Plans
The Company has a 401(k) retirement and savings plan (“401(k) Plan”) covering all qualified U.S. employees. The 401(k) Plan is a defined contribution plan and allows each participant to contribute up to 100% of the participant’s base wages up to an amount not to exceed an annual statutory maximum. The Company has made discretionary contributions to the 401(k) Plan and recorded expenses of approximately $24 thousand and $37 thousand for the years ended December 31, 2018 and 2017, respectively.
F-28
The Company maintains a defined contribution plan for certain international employees. The Company contributes 100% of the cost of the defined contribution. The Company recorded expenses of approximately $24 thousand and $38 thousand for the years ended December 31, 2018 and 2017, respectively, under this plan.
16. Income Taxes
On December 22, 2017, the United States enacted new tax reform (“Tax Act”). The Act contains provisions with separate effective dates but is generally effective for taxable years beginning after December 31, 2017. In December 2017, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act (SAB 118) which allows the Company to record provisional amounts during a measurement period not to extend beyond one year of the enactment date, which does not extend beyond the year ended December 31, 2018. Since the Tax Act was passed late in the fourth quarter of 2017, and ongoing guidance and accounting interpretation were expected over the next 12 months, the Company considered the accounting of the transition tax and deferred tax re-measurements to be incomplete at December 31, 2017 due to the forthcoming guidance and ongoing analysis of final year-end data and tax positions.
Included in the Tax Act are provisions which repatriate the aggregate of post-1986 earnings and profits of foreign corporations. At December 31, 2017, the Company calculated the impact of repatriation on a provisional basis under SAB 118. Repatriation reduced Federal U.S. tax attributes by $23 thousand for the year ended December 31, 2017. For the year ended December 31, 2018, immaterial adjustments were made to the utilization of attributions for repatriation. Beginning with the year ending December 31, 2018, the corporate statutory rates on U.S. earnings were reduced from 34% to 21%. The impact of the rate reduction resulted in a decrease to the deferred tax assets and an offset to the valuation allowance at December 31, 2017 by $28.9 million relating to the revaluation of the net deferred tax asset.
In accordance with SAB 118, the Company completed the accounting for the tax effects of the enactment of the Tax Act during the year ended December 31, 2018 with no adjustment to the provisional amounts recorded at December 31, 2017.
The Tax Act also includes provisions for tax on global intangible low-taxed income (“GILTI”). The GILTI provisions imposes a tax on foreign income in excess of a deemed return on tangible assets of foreign corporations. During 2018, the Company elected to treat future taxes related to GILTI as a period expense in the reporting period in which the tax is incurred.
Loss before provision for income taxes consisted of the following (in thousands):
|
| Years Ended December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Domestic |
| $ | (8,054 | ) |
| $ | (10,923 | ) |
Foreign |
|
| 29 |
|
|
| 47 |
|
Total |
| $ | (8,025 | ) |
| $ | (10,876 | ) |
The provision for income taxes in the accompanying consolidated statements of operations consisted of the following (in thousands):
|
| Years Ended December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Current Provision: |
|
|
|
|
|
|
|
|
Federal |
| $ | — |
|
| $ | — |
|
State |
|
| 1 |
|
|
| 1 |
|
Foreign |
|
| 13 |
|
|
| 13 |
|
Total |
|
| 14 |
|
|
| 14 |
|
|
|
|
|
|
|
|
|
|
Deferred (Benefit) Provision: |
|
|
|
|
|
|
|
|
Federal |
|
| — |
|
|
| — |
|
State |
|
| — |
|
|
| — |
|
Foreign |
|
| (1 | ) |
|
| — |
|
Total |
|
| (1 | ) |
|
| — |
|
Total provision |
| $ | 13 |
|
| $ | 14 |
|
F-29
A reconciliation of income taxes from operations computed using the U.S. federal statutory rates of 21% and 34% for the years ended December 31, 2018 and 2017, respectively, to that reflected in operations follows (in thousands):
|
| Years Ended December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Income tax benefit using U.S. federal statutory rate |
| $ | (1,675 | ) |
| $ | (3,698 | ) |
Rate Changes |
|
| — |
|
|
| 28,942 |
|
Permanent differences |
|
| 40 |
|
|
| 35 |
|
State income taxes, net of federal benefit |
|
| (438 | ) |
|
| (141 | ) |
Stock compensation |
|
| 24 |
|
|
| 53 |
|
Tax credits |
|
| (45 | ) |
|
| (127 | ) |
Foreign tax rate differential |
|
| 3 |
|
|
| (2 | ) |
Change in the valuation allowance |
|
| 2,041 |
|
|
| (25,161 | ) |
Unrealized gain |
|
| — |
|
|
| — |
|
Other |
|
| 63 |
|
|
| 113 |
|
Total |
| $ | 13 |
|
| $ | 14 |
|
Components of the Company’s deferred tax assets and liabilities are as follows (in thousands):
|
| December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Deferred tax assets: |
|
|
|
|
|
|
|
|
Net operating loss carryforwards |
| $ | 63,881 |
|
| $ | 59,823 |
|
Research and development credit carryforwards |
|
| 4,026 |
|
|
| 4,005 |
|
Capitalized research and development costs |
|
| 123 |
|
|
| 464 |
|
Capitalized start-up expenses |
|
| 2,490 |
|
|
| 2,846 |
|
Depreciation and other |
|
| 305 |
|
|
| 1,920 |
|
Total deferred tax assets |
|
| 70,825 |
|
|
| 69,058 |
|
Valuation allowance |
|
| (70,815 | ) |
|
| (69,058 | ) |
Net deferred tax asset |
| $ | 10 |
|
| $ | — |
|
Management of the Company has evaluated the positive and negative evidence bearing upon the realizability of the Company’s deferred tax assets and determined that it is more likely than not that the Company will not recognize the benefits of the deferred tax assets related to the U.S. and the Netherlands. As a result, a valuation allowance of approximately $70.8 million and $69.1 million was established at December 31, 2018 and 2017, respectively. The valuation allowance increased by approximately $1.7 million during the year ended December 31, 2018, primarily due to the valuation of the net deferred tax asset at the reduced 21% U.S. corporate income tax future rate.
At December 31, 2018, the Company had U.S. federal and state net operating loss carryforwards of approximately $237.8 million and $220.5 million, respectively. These operating loss carryforwards will expire at various times beginning in 2024 through 2037 for federal purposes and begin to expire in 2030 and will continue to expire through 2037 for state purposes. The 2018 net operating loss carryforward does not expire.
In addition, at December 31, 2018, the Company also has U.S. federal and state research and development tax credit carryforwards (excluding ASC 740, Income Taxes (“ASC 740”), valuation allowance of approximately $2.8 million and $1.6 million, respectively, to offset future income taxes. These tax credit carryforwards will expire at various times beginning in 2023 through 2037 for federal purposes and will expire at various times beginning in 2018 through 2032 for state purposes.
Utilization of net operating loss carryforwards and research and development credit carryforwards may be subject to a substantial annual limitation due to ownership change limitations that have occurred previously or that could occur in the future in accordance with Section 382 of the Internal Revenue Code of 1986 (“IRC Section 382”) and with Section 383 of the Internal Revenue Code of 1986, as well as similar state provisions. These ownership changes may limit the amount of net operating loss carryforwards and research and development credit carryforwards that can be utilized annually to offset future taxable income and taxes, respectively. In general, an ownership change, as defined by IRC Section 382, results
F-30
from transactions increasing the ownership of certain stockholders or public groups in the stock of a corporation by more than 50 percentage points over a three-year period. The Company has completed several financings since its inception, which may have resulted in a change in control as defined by IRC Section 382 or could result in a change in control in the future. As of December 31, 2018, the Company has not, as yet, conducted an IRC Section 382 study, which could impact its ability to utilize net operating loss and tax credit carryforwards annually in the future to offset the Company’s taxable income, if any.
The Company applies ASC 740-10, which provides guidance on the accounting for uncertainty in income taxes recognized in financial statements and requires the impact of a tax position to be recognized in the financial statements if that position is more likely than not of being sustained by the taxing authority. When uncertain tax positions exist, the Company recognizes the tax benefit of tax positions to the extent that the benefit will more likely than not be realized. The determination as to whether the tax benefit will more likely than not be realized is based upon the technical merits of the tax position as well as consideration of the available facts and circumstances. At December 31, 2018 and 2017, the Company had unrecognized tax liabilities of approximately $1.5 million for each year.
The following is a roll forward of the Company’s unrecognized tax benefits (in thousands):
|
| December 31, |
| |||||
| 2018 |
|
| 2017 |
| |||
Unrecognized tax benefit – as of the beginning of the year |
| $ | 1,481 |
|
| $ | 1,449 |
|
Gross increases – tax positions of the prior periods |
|
| (35 | ) |
|
| — |
|
Gross increases – current period tax positions |
|
| 16 |
|
|
| 32 |
|
Unrecognized tax benefits – as of the end of the year |
| $ | 1,462 |
|
| $ | 1,481 |
|
The Company will recognize interest and penalties related to uncertain tax positions, should they be assessed, in income tax expense. As of December 31, 2018, and 2017, the Company had no accrued interest or penalties related to uncertain tax positions, and no amounts have been recognized in the Company’s consolidated statements of operations.
The statute of limitations for assessment by the Internal Revenue Service (“IRS”) and state tax authorities is open for tax years ended December 31, 2014 through December 31, 2018, although carryforward attributes that were generated prior to tax year 2014 may still be adjusted upon examination by the IRS or state tax authorities if they either have been or will be used in a future period. The statute of limitations for assessment by foreign tax authorities is open for tax years ended December 31, 2014 through December 31, 2018. There are currently no federal or state audits in progress.
The Company has not yet completed a study of its research and development credit carryforwards. Once completed, this study may result in an adjustment to the Company’s research and development credit carryforwards. A full valuation allowance has been provided against the Company’s research and development credits, and if an adjustment is required at the time the study is completed, this adjustment would be offset by an adjustment to the deferred tax asset established for the research and development credit carryforward and the valuation allowance.
17. Subsequent Events
None.
F-31