Exhibit 99.1

Investor Presentation NASDAQ/TSX - BLU March 21 st , 2023

Forward Looking Statements 2 Certain statements contained in this presentation, other than statements of fact that are independently verifiable at the dat e h ereof, may constitute "forward - looking statements" within the meaning of Canadian securities legislation and regulations, the U.S. Private Securities Litigation Reform Act of 1995, as ame nde d, and other applicable securities laws. Forward - looking statements are frequently, but not always, identified by words such as “expects,” “anticipates,” “believes,” “intends,” “esti mat es,” “potential,” “possible,” “projects,” “plans,” and similar expressions. Such statements, based as they are on the current expectations of management, inherently involve numerous import ant risks, uncertainties and assumptions, known and unknown, many of which are beyond BELLUS Health Inc.'s (“BELLUS Health”) control. Such statements include, but are not limite d t o, the potential of camlipixant (BLU - 5937) to successfully treat refractory chronic cough (“RCC”) and other hypersensitization - related disorders and benefit such patients, BE LLUS Health's expectations related to its preclinical studies and clinical trials, including the timing of initiation of and the design of the Phase 3 clinical trials of camlipixant in RCC, the timing and outcome of interactions with regulatory agencies, the potential activity and tolerability profile, selectivity, potency and other characteristics of camlipixant , including as compared to other competitor candidates, especially where head - to - head studies have not been conducted and cross - trial comparisons may not be directly comparable due to differences in st udy protocols, conditions and patient populations, the commercial potential of camlipixant , including with respect to patient population, pricing and labeling, BELLUS Health's financial position and sufficiency of c ash resources to bring BELLUS Health through topline results of CALM - 1 and CALM - 2 clinical trials, and the potential applicability of camlipixant and BELLUS Health's P2X3 platform to treat other disorders. Risk factors that may affect BELLUS Health's future results include but are not limited to: the benefits and impact on label of its enrichment strategy, estimates and projections regarding the size and opportunity of the addressable RCC market for camlipixant , the ability to expand and develop its project pipeline, the ability to obtain adequate financing, the ability of BELLUS Health to maintain its rights to intellectual property and obtain adequate protection of future products through such int ellectual property, the impact of general economic conditions, general conditions in the pharmaceutical industry, the impact of the ongoing COVID - 19 pandemic on BELLUS Health's op erations, plans and prospects, including to the initiation and completion of clinical trials in a timely manner or at all, changes in the regulatory environment in the juris dic tions in which BELLUS Health does business, supply chain impacts, stock market volatility, fluctuations in costs, changes to the competitive environment due to consolidation, achieve men t of forecasted burn rate, achievement of forecasted preclinical study and clinical trial milestones, reliance on third parties to conduct preclinical studies and clinical trials fo r camlipixant and that actual results may differ from topline results once the final and quality - controlled verification of data and analyses has been completed. In addition, the length of BELLUS He alth's product candidate's development process and its market size and commercial value are dependent upon a number of factors. Moreover, BELLUS Health's growth and future prospect s a re mainly dependent on the successful development, patient tolerability, regulatory approval, commercialization and market acceptance of its product candidate camlipixant and other products. Consequently, actual future results and events may differ materially from the anticipated results and events expressed in the forward - looking statements. BE LLUS Health believes that expectations represented by forward - looking statements are reasonable, yet there can be no assurance that such expectations will prove to be correct. The re ader should not place undue reliance, if any, on any forward - looking statements included in this presentation. These forward - looking statements speak only as of the date made, and B ELLUS Health is under no obligation and disavows any intention to update publicly or revise such statements as a result of any new information, future event, circumstances or oth erw ise, unless required by applicable legislation or regulation. Please see BELLUS Health's public filings with the Canadian securities regulatory authorities, including, but not limited to, it s Annual Information Form, and the United States Securities and Exchange Commission, including, but not limited to, its Annual Report on Form 40 - F, for further risk facto rs that might affect BELLUS Health and its business.

Company Overview

BELLUS Health – Working to Better the Lives of Patients Suffering from Persistent Cough Financials Key Upcoming Events Lead Indication - Refractory Chronic Cough (RCC) • Persistent cough >8 weeks that does not respond to treatment for underlying condition or is unexplained • Compelling results from the SOOTHE Phase 2b trial (Dec 2021) • First patients randomized in the Phase 3 CALM program (CALM - 1 and CALM - 2 trials) in 4Q 2022 • Population estimated at ~9M in the U.S., a large and growing market with limited competition • Topline results from CALM - 1 and CALM - 2 expected in 2H 2024 and 2025, respectively • Topline results from Phase 1 QD formulation expected in 2Q 2023 • Analyst Day planned for 2H 2023 Pipeline in a Product • Potential to study camlipixant in other cough indications Intellectual Property • Patents granted to 2034 (composition of matter) and 2038 (method of use) • 100% ownership of global rights Drug in Development: Camlipixant (BLU - 5937) • Oral P2X3 antagonist with potential best - in - class profile • US$337.1M cash position* • Cash runway extends to 2H 2025 * Cash position as of December 31, 2022

Strong Leadership and Advisory Group Management Board of Directors Clinical Advisory Board Dr. Surinder Birring, MB ChB (Hons), MD King’s College London Dr. Michael S. Blaiss, MD Medical College of Georgia Dr. Peter Dicpinigaitis, MD Albert Einstein Medical College Dr. Jacky Smith (Chair) , MB, ChB, FRCP, PhD Manchester University Dr. Denis Garceau, PhD Chief Scientific Officer Tony Matzouranis Senior Vice President, Business Development Roberto Bellini President & Chief Executive Officer Dr. Catherine Bonuccelli, MD Chief Medical Officer Ramzi Benamar, MBA Chief Financial Officer Franklin Berger Roberto Bellini Joseph Rus Pierre Larochelle Dr. Clarissa Desjardins, PhD Dr. Youssef Bennani, PhD Dr. Francesco Bellini, PhD Chair Dr. Bill Mezzanotte , MD, PhD CHRONIC COUGH 5 Dr. Andreas Orfanos, MBBCh, MBA, FFPM Chief Operations Officer

Refractory Chronic Cough Cough lasting ≥ 8 weeks that does not respond to treatment for underlying cause or is unexplained 1 No approved treatment , current options are inadequate and non - specific 3 Significant impact on patients ’ quality of life, including impact on social, physical and psychosocial well - being 2 Large patient population 4 - up to ~ 9 M refractory chronic cough patients in the U . S ., ~ 9 M in Europe Top - 5 and ~7M in China 1. Irwin RS et al, (2018) CHEST 153 (1): 196 - 209. 2. Kuzniar et al. (2007) Mayo Clin. Proc. 82(1) 56 - 60. 3. Ryan NM, (2018) Expert Opin Pharmacother 19(7): 687 - 711. 4. Company sponsored market research.
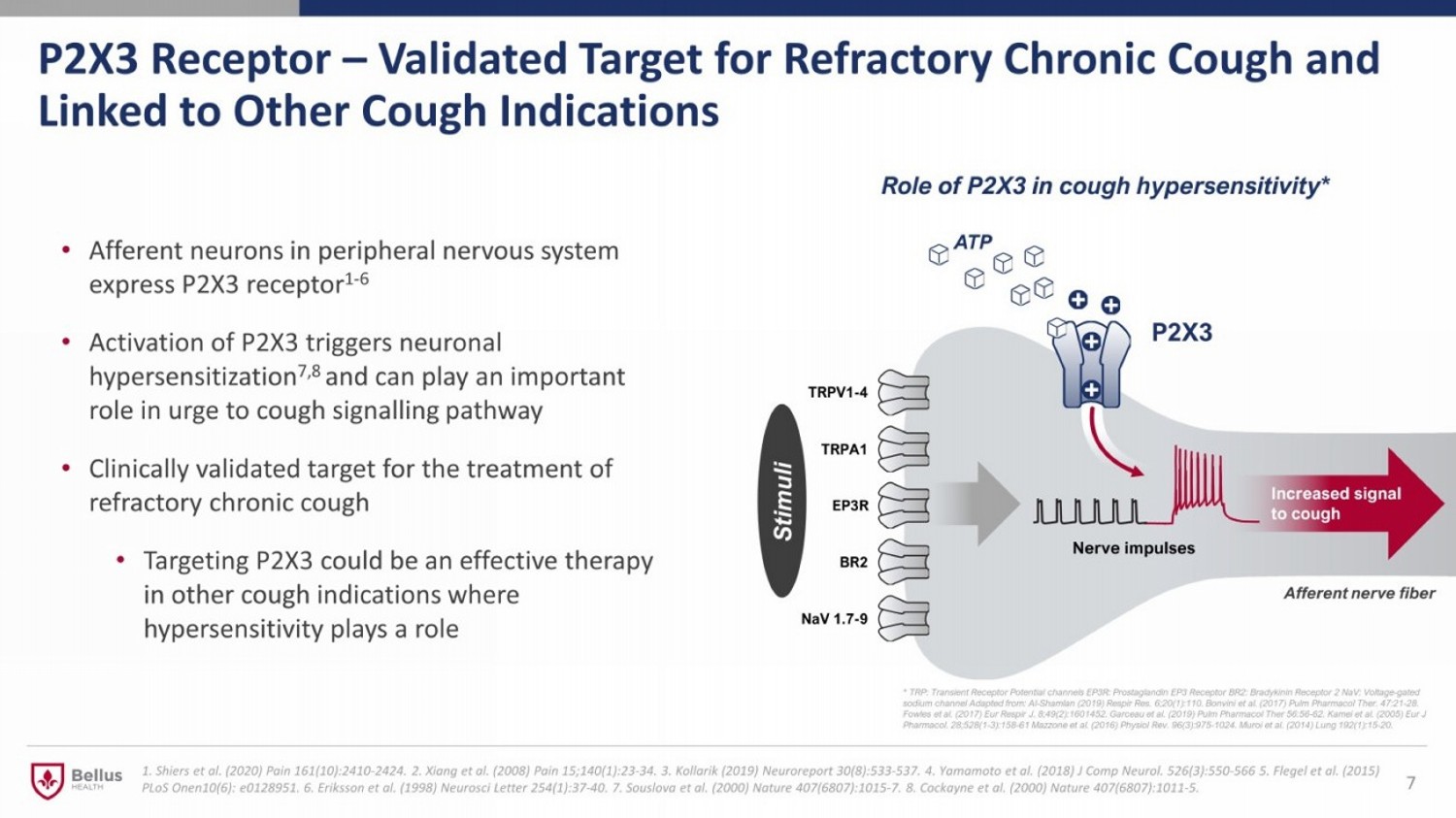
P2X3 Receptor – Validated Target for Refractory Chronic Cough and Linked to Other Cough Indications • Afferent neurons in peripheral nervous system express P2X3 receptor 1 - 6 • Activation of P2X3 triggers neuronal hypersensitization 7,8 and can play an important role in urge to cough signalling pathway • Clinically validated target for the treatment of refractory chronic cough • Targeting P2X3 could be an effective therapy in other cough indications where hypersensitivity plays a role 1. Shiers et al. (2020) Pain 161(10):2410 - 2424. 2. Xiang et al. (2008) Pain 15;140(1):23 - 34. 3. Kollarik (2019) Neuroreport 30(8):533 - 537. 4. Yamamoto et al. (2018) J Comp Neurol. 526(3):550 - 566 5. Flegel et al. (2015) PLoS Onen10(6): e0128951. 6. Eriksson et al. (1998) Neurosci Letter 254(1):37 - 40. 7. Souslova et al. (2000) Nature 407(6807):1015 - 7. 8. Cockayne et al. (2000) Nature 407(6807):1011 - 5. 7 ATP Stimuli Afferent nerve fiber Role of P2X3 in cough hypersensitivity* * TRP: Transient Receptor Potential channels EP3R: Prostaglandin EP3 Receptor BR2: Bradykinin Receptor 2 NaV : Voltage - gated sodium channel Adapted from: Al - Shamlan (2019) Respir Res . 6;20(1):110. Bonvini et al. (2017) Pulm Pharmacol Ther . 47:21 - 28. Fowles et al. (2017) Eur Respir J. 8;49(2):1601452. Garceau et al. (2019) Pulm Pharmacol Ther 56:56 - 62 . Kamei et al. (2005) Eur J Pharmacol . 28;528(1 - 3):158 - 61 Mazzone et al. (2016) Physiol Rev. 96(3):975 - 1024 . Muroi et al. (2014) Lung 192(1):15 - 20.

Camlipixant (BLU - 5937) - Best - In - Class Potential 8 Compelling efficacy from SOOTHE Phase 2b trial completed in December 2021 Well - tolerated with a low rate of class - related taste side effects Twice daily dosing with once - daily dosing in development camlipixant Highly potent oral P2X3 antagonist with best - in - class selectivity being developed to treat refractory chronic cough

Camlipixant (BLU - 5937) - Pipeline 9 PROGRAM DEVELOPMENT STATUS Indication / Project Preclinical Phase 1 Phase 2 Phase 3 Worldwide Rights Next Anticipated Step Camlipixant Refractory Chronic Cough (BID Formulation) 2H 2024: CALM - 1 Topline Results 2025: CALM - 2 Topline Results Refractory Chronic Cough (QD Formulation) 2Q 2023: Phase 1 Study Completion Potential for Additional Cough Indications POTENTIAL COUGH INDICATIONS UNDER EVALUATION • POST VIRAL COUGH • IPF COUGH • ASTHMA COUGH

SOOTHE Phase 2b Results

SOOTHE Trial Design 11 Randomized, double - blind, 4 - week placebo - controlled parallel arm study with 3 active doses PRIMARY ENDPOINT Placebo - adjusted change from baseline in 24H cough frequency (Day 28) POPULATION Refractory chronic cough for ≥1 year Awake cough frequency: ≥25 coughs/h 249 participants recruited from 64 North American sites ( 142 participants) 56 European sites (107 participants) Run - In Placebo (n= 63) Camlipixant 12.5 mg BID (n= 62) Camlipixant 50 mg BID (n= 62) Camlipixant 200 mg BID (n= 62) Follow - Up SOOTHE Refractory Chronic Cough Phase 2b Trial Design Screening 43 Days: - 16 0 28 Cough recordings: - 6 15 SECONDARY ENDPOINTS Leicester Cough Questionnaire (LCQ) Cough Severity Visual Analogue Scale (CS - VAS)
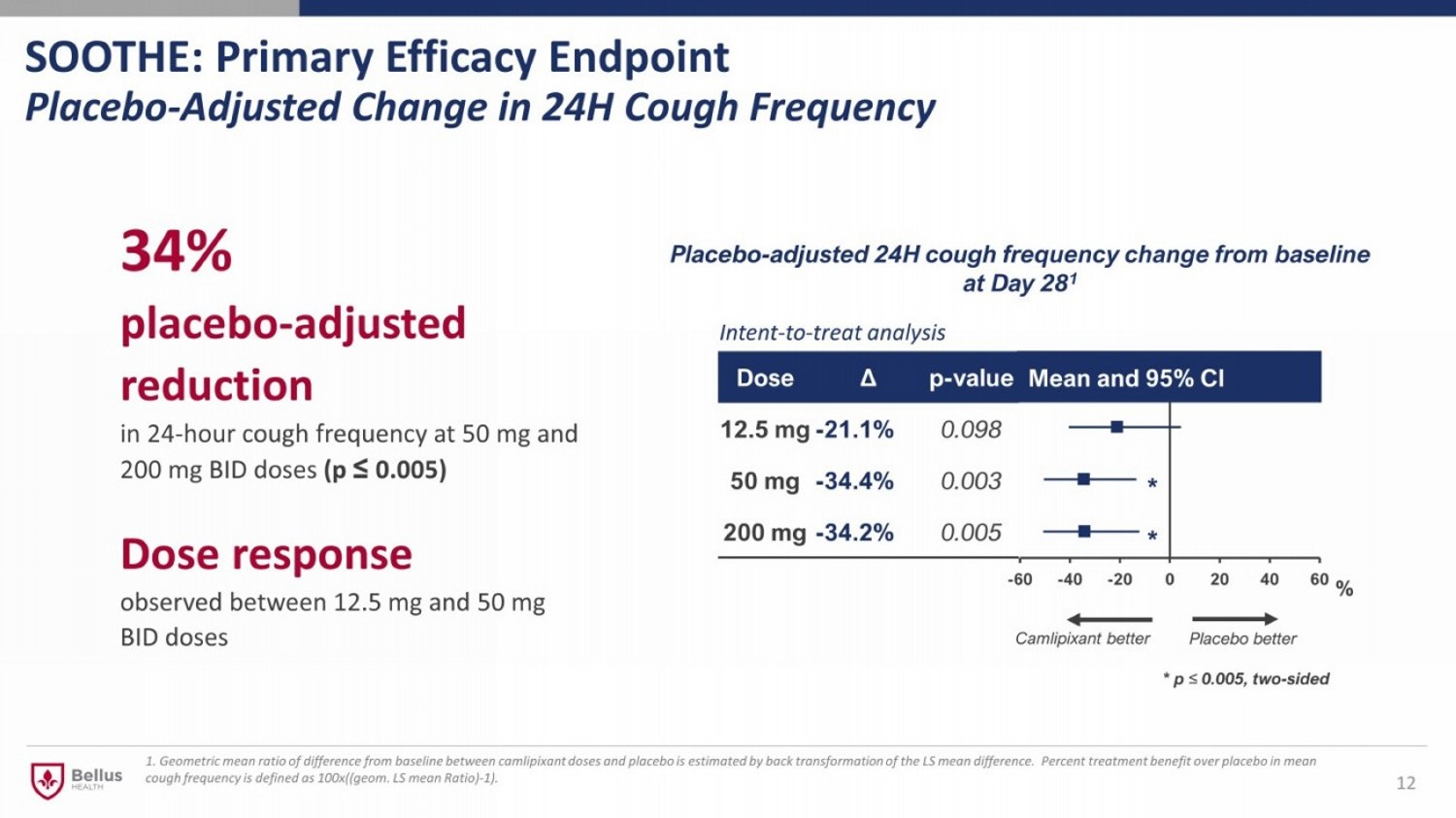
SOOTHE: Primary Efficacy Endpoint Placebo - Adjusted Change in 24H Cough Frequency 12 34% placebo - adjusted reduction in 24 - hour cough frequency at 50 mg and 200 mg BID doses (p ≤ 0.005) Dose response observed between 12.5 mg and 50 mg BID doses -60 -40 -20 0 20 40 60 Dose Δ p - value 12.5 mg - 21.1% 0.098 50 mg - 34.4% 0.003 200 mg - 34.2% 0.005 Mean and 95% CI Intent - to - treat analysis Placebo - adjusted 24H cough frequency change from baseline at Day 28 1 * * * p ≤ 0.005, two - sided Camlipixant better Placebo better % 1. Geometric mean ratio of difference from baseline between camlipixant doses and placebo is estimated by back transformation of th e LS mean difference. Percent treatment benefit over placebo in mean cough frequency is defined as 100x((geom. LS mean Ratio) - 1).

-60.0% -50.0% -40.0% -30.0% -20.0% -10.0% 0.0% Placebo 12.5 mg BID 50 mg BID 200 mg BID Relative change from baseline in 24H cough frequency (ITT) Baseline Day 15 Day 28 SOOTHE: Change from Baseline in 24H Cough Frequency 13 Percentage change in 24H cough frequency 53% reduction from baseline in 24 - hour cough frequency at day 28 with 50 mg and 200 mg BID doses * p ≤ 0.005, two - sided * * * *

36% 14% 7% 52% 35% 19% 61% 44% 25% 62% 48% 19% 0% 10% 20% 30% 40% 50% 60% 70% 80% ≥ 30% ≥ 50% ≥ 70% Placebo BLU-5937 12.5 mg BID BLU-5937 50 mg BID BLU-5937 200 mg BID Day 28 12.5 mg BID 50 mg BID 200 mg BID -1 1 3 5 7 9 11 13 15 p=0.0865 p=0.0077 p=0.0080 ≥ 30% -1 1 3 5 7 9 11 13 15 p=0.0147 p=0.0007 p=0.0003 ≥ 50% -1 1 3 5 7 9 11 13 15 p=0.0716 p=0.0157 p=0.0715 ≥ 70% Odds ratio (95% CI) 2.9 4.2 5.6 4.8 3.1 2.8 1.9 3.0 3.2 >60% of patients achieved ≥30% reduction in cough frequency at therapeutic doses Robust odds ratios favored treatment at every dose; almost all data points at therapeutic doses are statistically significant SOOTHE: Responder Rates in 24H Cough Frequency
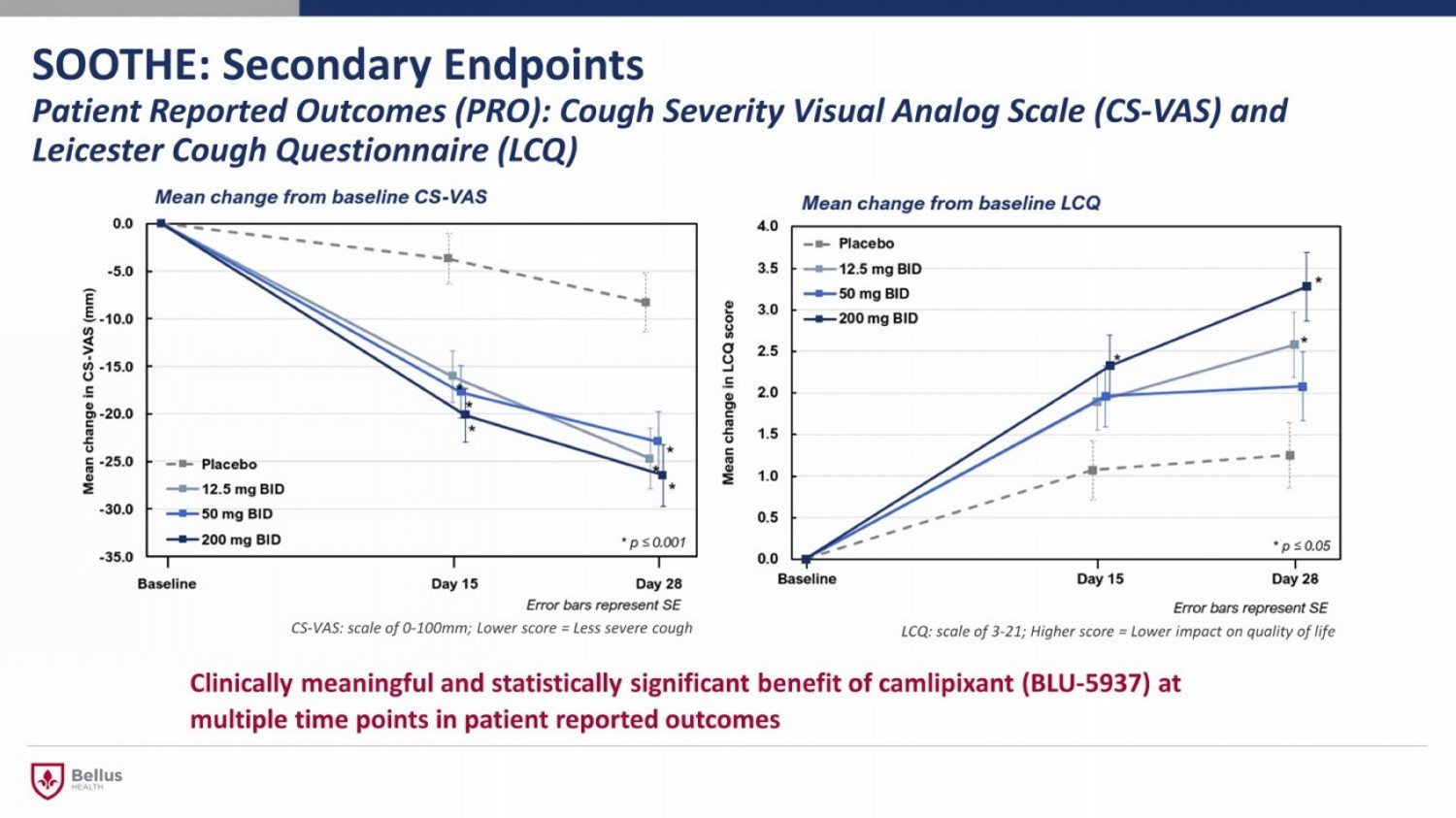
SOOTHE: Secondary Endpoints Patient Reported Outcomes (PRO): Cough Severity Visual Analog Scale (CS - VAS) and Leicester Cough Questionnaire (LCQ) Clinically meaningful and statistically significant benefit of camlipixant (BLU - 5937) at multiple time points in patient reported outcomes LCQ: scale of 3 - 21; Higher score = Lower impact on quality of life CS - VAS: scale of 0 - 100mm; Lower score = Less severe cough

SOOTHE: Safety and Tolerability 16 Generally well - tolerated S imilar rate of treatment emergent adverse events (TEAEs) reported for placebo and camlipixant (BLU - 5937) n (%) Placebo (n= 63) Camlipixant 12.5 mg BID (n= 62) Camlipixant 50 mg BID (n= 62) Camlipixant 200 mg BID (n= 62) Subjects with ≥1 TEAE 22 (34.9%) 23 (37.1%) 13 (21.0%) 19 (30.6%) Subjects with ≥1 TE SAE 0 0 0 0 Subjects with TEAE leading to discontinuation, n (%) * 1 (1.6%) 0 0 2 (3.2%) Most Common TEAEs (≥5% at any dose) † Nausea 0 0 5 (8.1%) 2 (3.2%) Dysgeusia 0 3 (4.8%) 4 (6.5%) 3 (4.8%) UTI 0 3 (4.8%) 0 0 † No TEAE reported with an incidence ≥5% in the exploratory population * Placebo: worsening of cough; Camlipixant 200 mg BID: worsening of cough, dry mouth
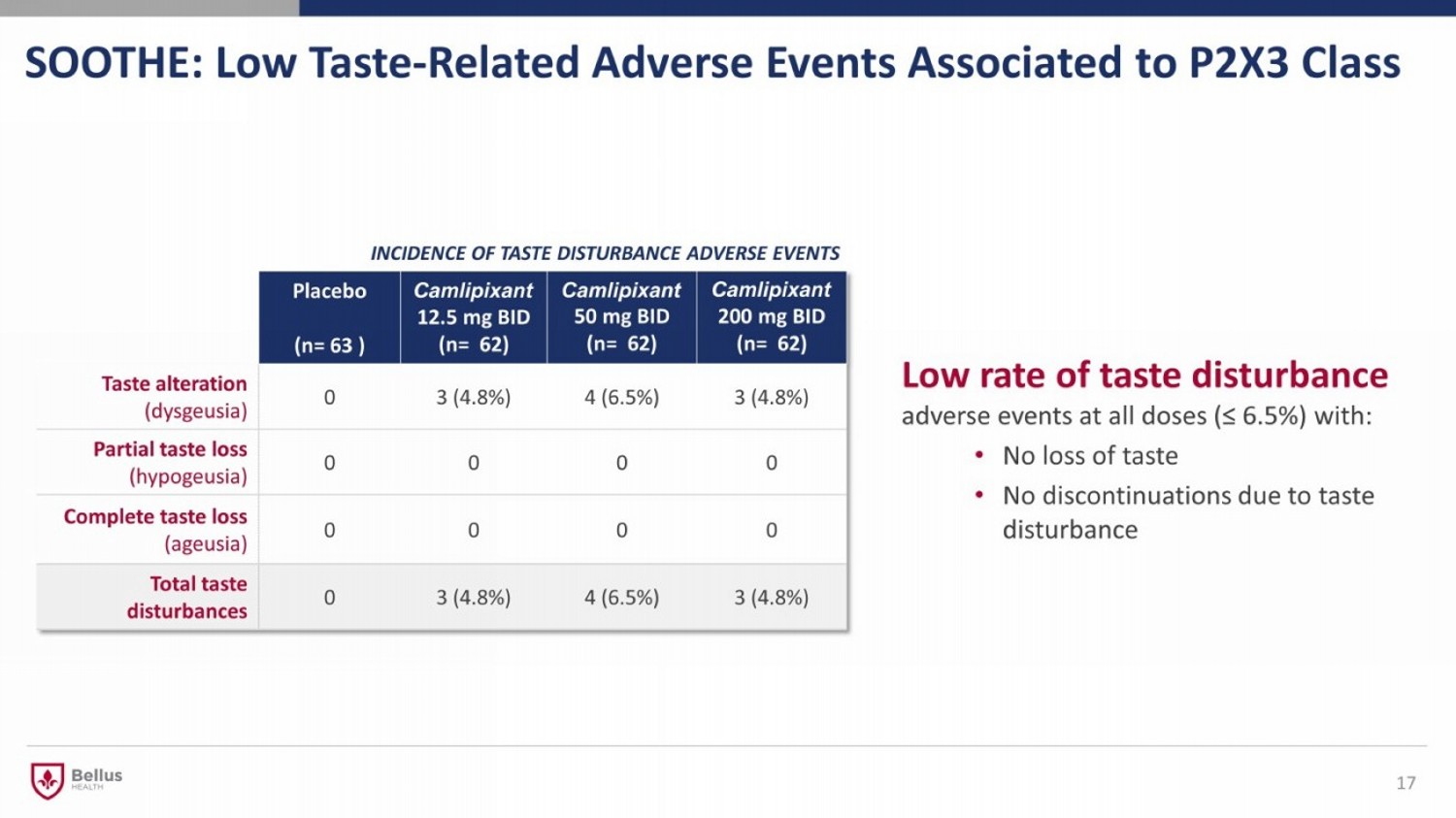
SOOTHE: Low Taste - Related Adverse Events Associated to P2X3 Class 17 Low rate of taste disturbance adverse events at all doses (≤ 6.5%) with: • No loss of taste • No discontinuations due to taste disturbance INCIDENCE OF TASTE DISTURBANCE ADVERSE EVENTS Placebo (n= 63 ) Camlipixant 12.5 mg BID (n= 62) Camlipixant 50 mg BID (n= 62) Camlipixant 200 mg BID (n= 62) Taste alteration (dysgeusia) 0 3 (4.8%) 4 (6.5%) 3 (4.8%) Partial taste loss (hypogeusia) 0 0 0 0 Complete taste loss (ageusia) 0 0 0 0 Total taste disturbances 0 3 (4.8%) 4 (6.5%) 3 (4.8%)

CALM Phase 3 Program
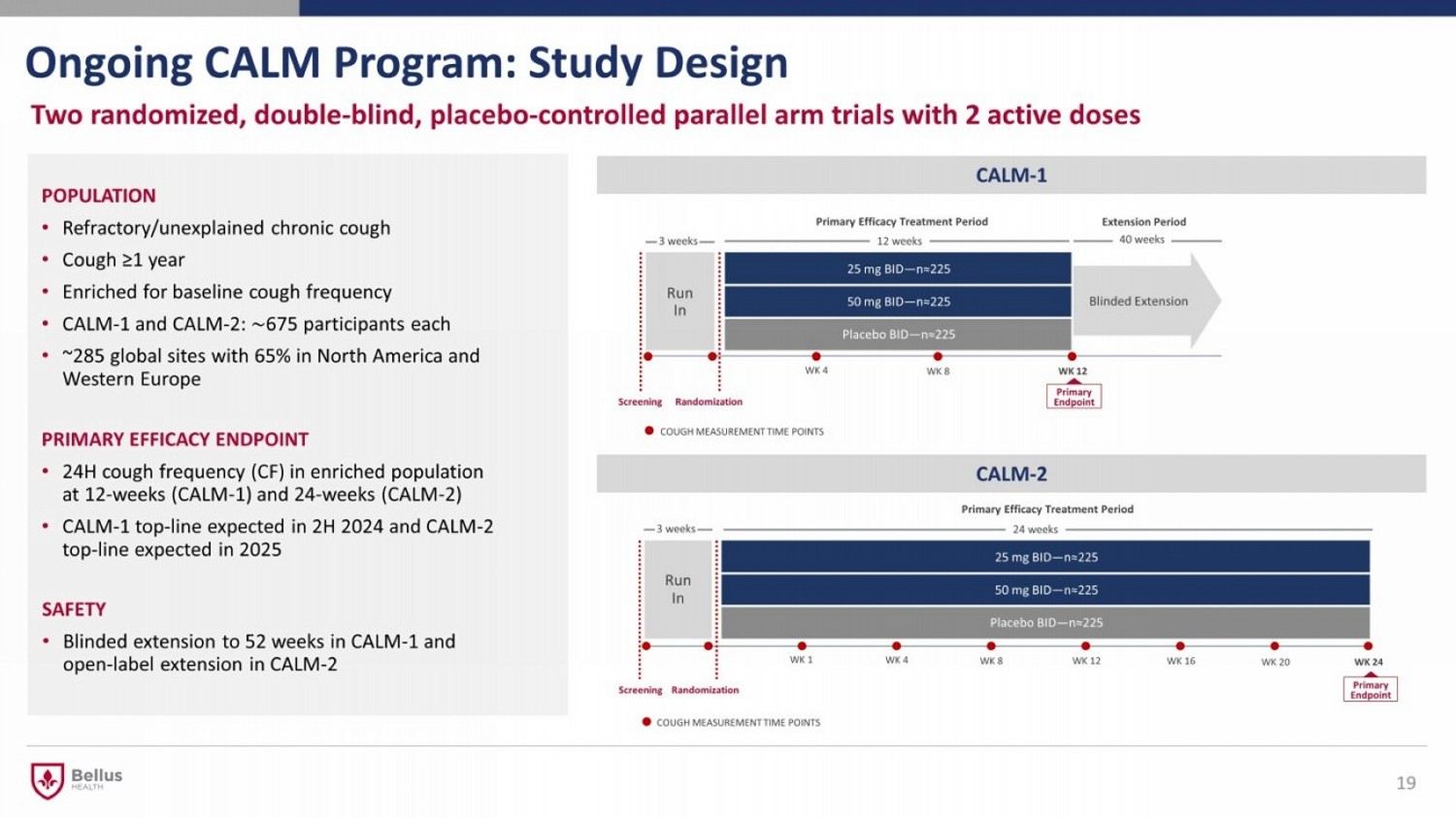
Run In 25 mg BID — n≈225 50 mg BID — n≈225 Placebo BID — n≈225 Run In 25 mg BID — n≈225 50 mg BID — n≈225 Placebo BID — n≈225 Ongoing CALM Program: Study Design Two randomized, double - blind, placebo - controlled parallel arm trials with 2 active doses 19 POPULATION • Refractory/unexplained chronic cough • Cough ≥1 year • Enriched for baseline cough frequency • CALM - 1 and CALM - 2: ∼ 675 participants each • ~285 global sites with 65% in North America and Western Europe PRIMARY EFFICACY ENDPOINT • 24H cough frequency (CF) in enriched population at 12 - weeks (CALM - 1) and 24 - weeks (CALM - 2) • CALM - 1 top - line expected in 2H 2024 and CALM - 2 top - line expected in 2025 SAFETY • Blinded extension to 52 weeks in CALM - 1 and open - label extension in CALM - 2 CALM - 1 Primary Endpoint Primary Efficacy Treatment Period 12 weeks 40 weeks CALM - 2 Screening Randomization WK 24 WK 16 WK 20 WK 8 WK 12 WK 4 WK 1 Primary Endpoint Screening Randomization C OUGH M EASUREMENT T IME P OINTS 24 weeks Primary Efficacy Treatment Period Extension Period Blinded Extension 3 weeks WK 8 WK 12 WK 4 C OUGH M EASUREMENT T IME P OINTS 3 weeks

Ongoing CALM Program: Enrichment Strategy 20 CALM - 1 and CALM - 2 populations to be enriched for baseline cough frequency PRIMARY EFFICACY ENDPOINT: COUGH FREQUENCY IN PRIMARY POPULATION (90% POWER) • 24H cough frequency vs placebo in Primary Population SECONDARY EFFICACY ENDPOINTS (80% POWER ) • Leicester Cough Questionnaire (LCQ), Cough Severity VAS (CS - VAS) • 24H cough frequency vs placebo in Overall Population PRIMARY POPULATION • ≥ 20 coughs/h (24H CF) • Equivalent to ≥25 cough/h (awake CF) population in successful SOOTHE Phase 2b trial EXTENDED POPULATION • <20 coughs/h (24H CF) • Expected 1:3 ratio of Extended Population to Primary Population PBO BLU

VitaloJAK is the cough recording and counting system used to capture the 24H cough frequency data in most cough trials • Used in camlipixant (BLU - 5937) and gefapixant RCC trials The Company conducted validation work on VitaloJAK • Validation work consisted of comparing compressed vs non - compressed recordings in SOOTHE Phase 2b trial participants • 45 SOOTHE Phase 2b trial participants showed a sensitivity of 98.7% with no systematic errors identified • Validation protocol and statistical plan submitted to FDA in Q4 2022 • Validation work has no impact on start of Phase 3 VITALOJAK COUGH MONITORING DEVICE Ongoing CALM Phase 3: VitaloJAK Cough Monitoring System 21

Market and Competitive Landscape
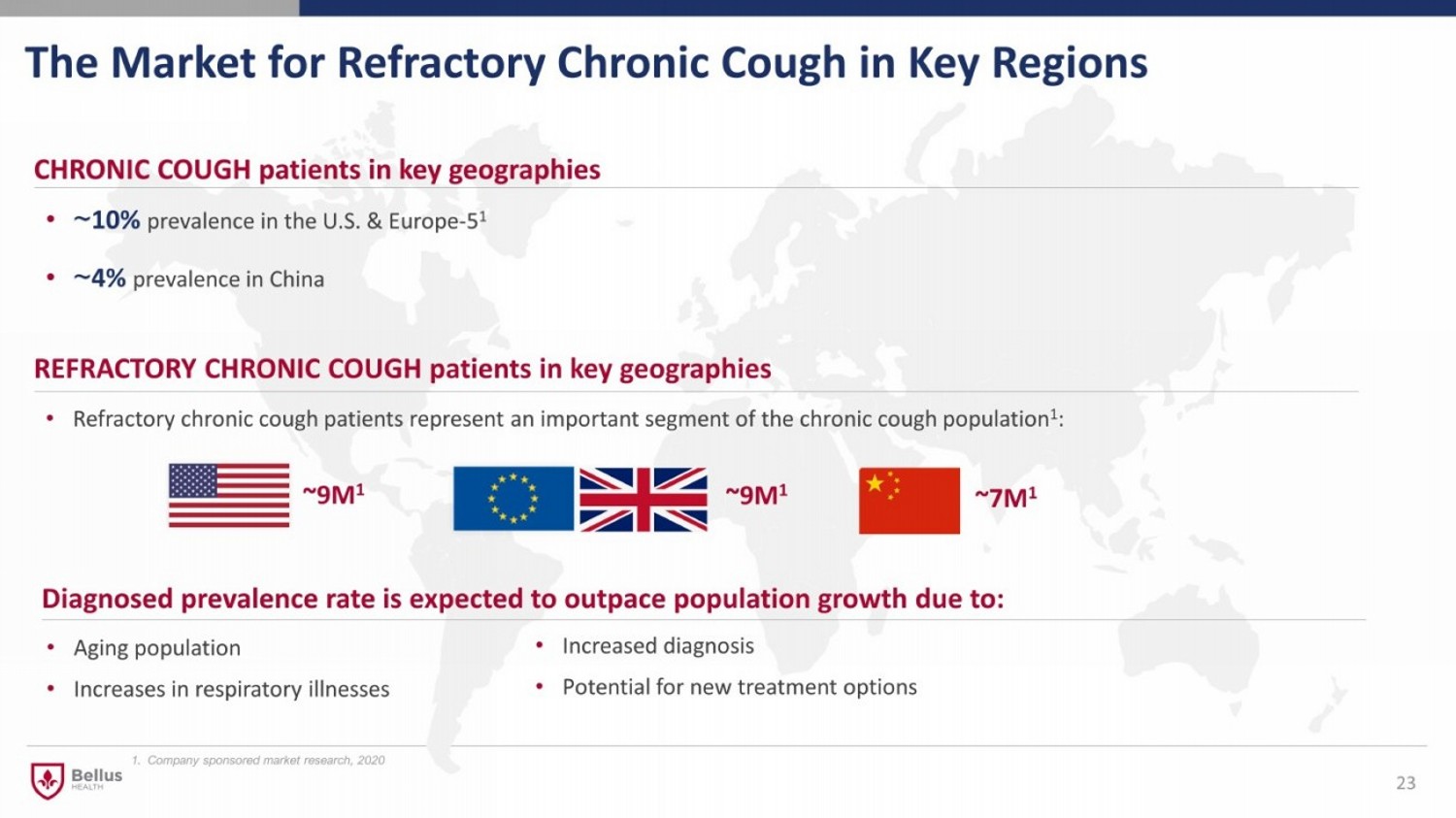
The Market for Refractory Chronic Cough in Key Regions 23 CHRONIC COUGH patients in key geographies • ~ 10% prevalence in the U.S. & Europe - 5 1 • ~ 4% prevalence in China • Refractory chronic cough patients represent an important segment of the chronic cough population 1 : REFRACTORY CHRONIC COUGH patients in key geographies ~9M 1 ~9M 1 • Aging population • Increases in respiratory illnesses Diagnosed prevalence rate is expected to outpace population growth due to: • Increased diagnosis • Potential for new treatment options 1. Company sponsored market research, 2020 ~7M 1

24 P2X3 Competitive Landscape 1 Company Candidate Gefapixant Sivopixant Camlipixant (BLU - 5937) Stage of Development Approved in Japan, Switzerland EU/US Under Review Phase 2b Phase 3 Expected Next Steps Submit additional information in U.S./EU in 1H 2023 * Evaluating Next Steps ** CALM - 1 topline results expected in 2H 2024 Dosing BID QD BID / QD in development P2X3 vs. P2X2/3 Selectivity 3 - 7x 2 ~ 250x 3 ~ 1500x 1 ST IN CLASS P2X3 ANTAGONIST 2 ND GENERATION P2X3 ANTAGONISTS Best - in - class P2X3 selectivity may support favorable clinical and commercial profile if approved 1. Active programs; Limited head to head studies have been conducted; data presented is derived from company specific disclosures . 2. Ford et al. (2013) FASEB J. 27: 887.5 - 887.5 3. Kai et al. 2020 Abstract presented at: ACS Fall 2020 Virtual & Meeting Exposition; August 17 - 20, 2020 *Merck 10K, Feb 23, 2023. Merck’s NDA for gefapixant received a CRL by U.S. FDA in February 2022; **Shionogi R&D Day, October 2022 RB0
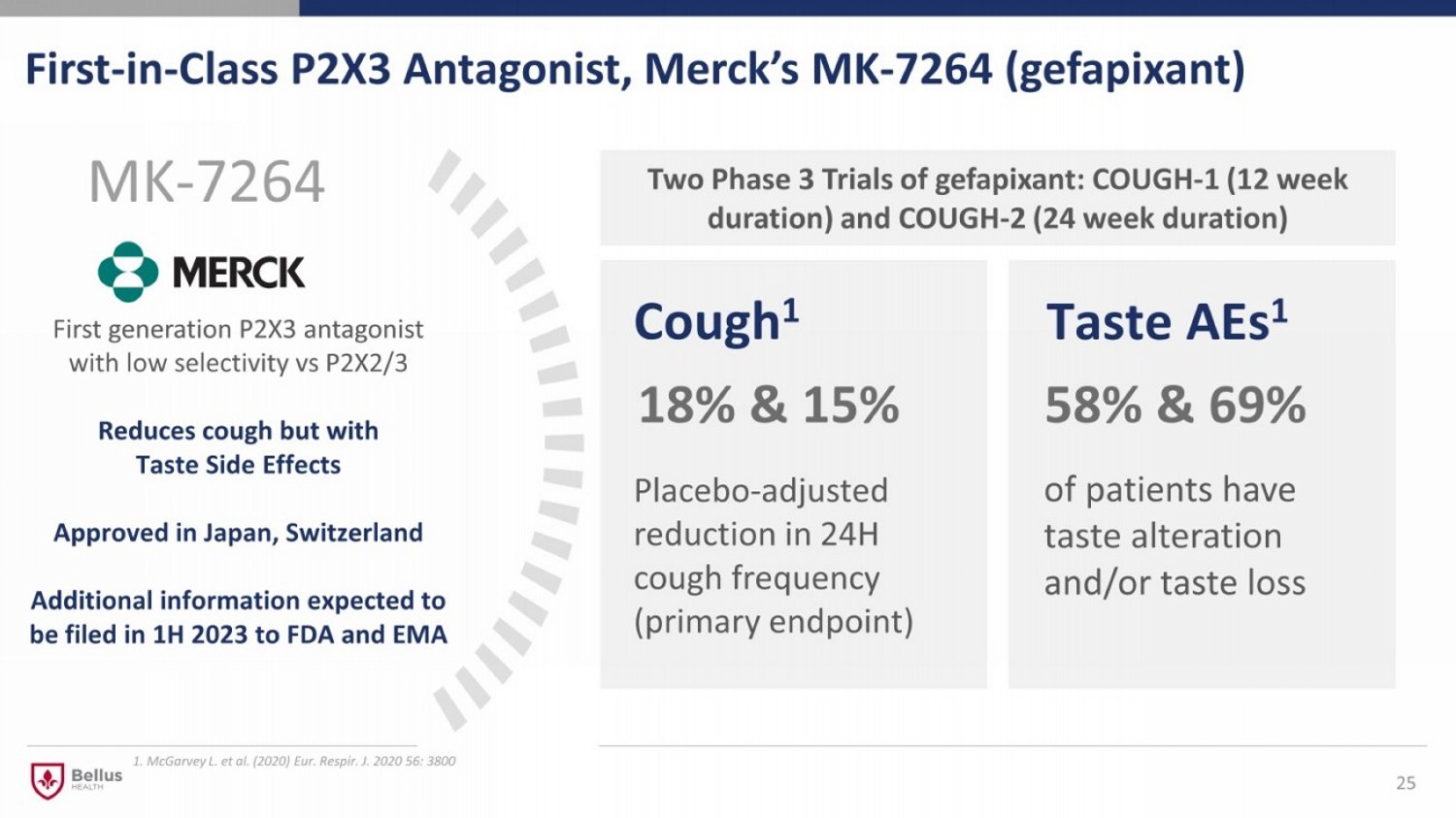
First - in - Class P2X3 Antagonist, Merck’s MK - 7264 ( gefapixant ) 25 18% & 15 % Cough 1 MK - 7264 58% & 69% Taste AEs 1 Placebo - adjusted reduction in 24H cough frequency (primary endpoint) of patients have taste alteration and/or taste loss Two Phase 3 Trials of gefapixant: COUGH - 1 (12 week duration) and COUGH - 2 (24 week duration) First generation P2X3 antagonist with low selectivity vs P2X2/3 Reduces cough but with Taste Side Effects Approved in Japan, Switzerland Additional information expected to be filed in 1H 2023 to FDA and EMA 1. McGarvey L. et al. (2020) Eur. Respir. J. 2020 56: 3800

Shionogi’s S - 600918 (sivopixant) 26 12 % Cough 1 Sivopixant 33% Taste AEs 1 Placebo - adjusted reduction in 24H cough frequency (primary endpoint) of patients with taste alteration and/or taste loss Phase 2b Trial (4 week duration) 300mg QD Selective P2X3 antagonist Three doses tested with none achieving statistical significance 1 Trial completed in December 2020 Program under evaluation as of October 2022 2 1. Sivopixant dose - finding study in RCC/UCC, Shionogi, Twelfth London International Cough Symposium, July 14 th 2022 2. Shionogi R&D Day Presentation, October 11 th 2022

Camlipixant (BLU - 5937) Well - Positioned for Potential Class Differentiation Source: Bellus Health, BLU - 5937 Commercial Opportunity Assessment Criteria • Treatment effect vs. placebo • Taste effects • Positive Phase 2b results with potential best - in - class profile • Best - in - class selectivity • Well - designed clinical trials Camlipixant Considerations CAMLIPIXANT WELL - POSITIONED TO BE A POTENTIAL LEADER IN P2X3 CLASS Efficacy and Tolerability Patient Persistence and Compliance Payer Preference Timing of Market Entry • Launch timing • HCP readiness • Referral and treatment patterns • Focused on efficient development program • Ease of use, dosing regimen • Duration of treatment • Twice - daily formulation with once - daily formulation development started • Price • Potential for modest premium to first in class P2X3 antagonist 27

U.S. Commercialization Strategy

91 % 29 25 M 8.6 M Chronic Cough Refractory Chronic Cough Established Unmet Need and Willingness to Rx in RCC with Specialists 2 Source: ZS Associates/Bellus Health Market Research (2022) 1. All - comer survey (n=1483) of US Pulmonologists (n= 289) , Allergists (n= 207) , Otolaryngologists (n= 217) , Gastroenterologists (n= 197) , and Primary Care Physicians (n= 573) • “In a typical calendar year, of all the adult patients you see, how many Chronic Cough / Refractory Chronic Cough patients are you the physician primarily responsible for continuing to try to treat or monitor their persistent cough?” 2. Survey (n=179; >30 RCC patients per year) of US Pulmonologists, Allergists, Otolaryngologists, Gastroenterologists 3. “How much time would it take for you to prescribe Product Y (product with profile like camlipixant based on Phase 2b SOOTHE data) broadly to your patients with Refractory Chronic Cough (RCC)?” Number of CC/RCC Patients at Physician Offices Annually 1 EAGER TO PRESCRIBE 3 of specialists would prescribe camlipixant (BLU - 5937) within 12 months of approval say RCC is an extremely burdensome condition for patients 3 % Satisfaction rate with current RCC treatment options 1.8M at specialists 92 % UNMET NEED Updated Quantitative U.S. Physician Surveys Demonstrate Large RCC Patient Pool and Eagerness to Prescribe New Therapy
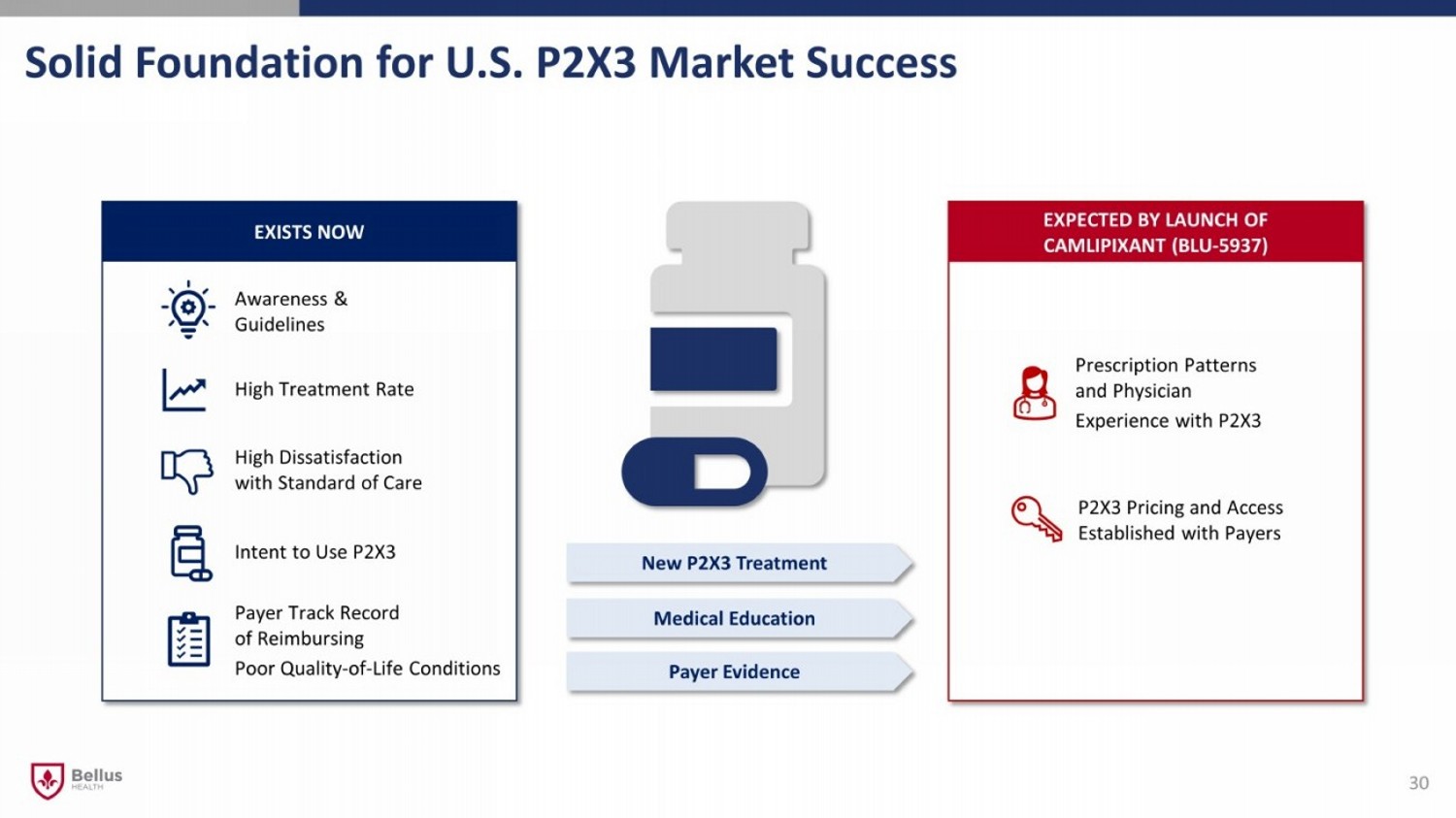
30 EXPECTED BY LAUNCH OF CAMLIPIXANT (BLU - 5937) EXISTS NOW High Treatment Rate Medical Education Awareness & Guidelines Payer Track Record of Reimbursing Poor Quality - of - Life Conditions Prescription Patterns and Physician Experience with P2X3 Intent to Use P2X3 P2X3 Pricing and Access Established with Payers Payer Evidence New P2X3 Treatment High Dissatisfaction with Standard of Care Solid Foundation for U.S. P2X3 Market Success

31 • Leverage potential best - in - class profile to target P2X3 naïve and first - generation switchers / discontinuations • 150 - 175 sales representatives targeting top 30% respiratory specialists • Multichannel consumer outreach, referral, and telemedicine platforms • Achieve optimal payer access • Continued disease awareness and medical education • Build patient advocacy • Prepare access landscape Pre - Launch Establishing r eadiness • Expand sales reach targeting broader pool of RCC patients through: • Further breadth and depth with respiratory specialists • Expand sales force reach to high volume (top 10%) primary care physicians Launch Potential rapid penetration of c amlipixant as first choice in RCC Market Expansion Opportunity Camlipixant (BLU - 5937): Early U.S. Commercialization Strategy Initial Targeted Approach with Potential to Expand Alongside Market

Camlipixant (BLU - 5937) Potential Additional Indications
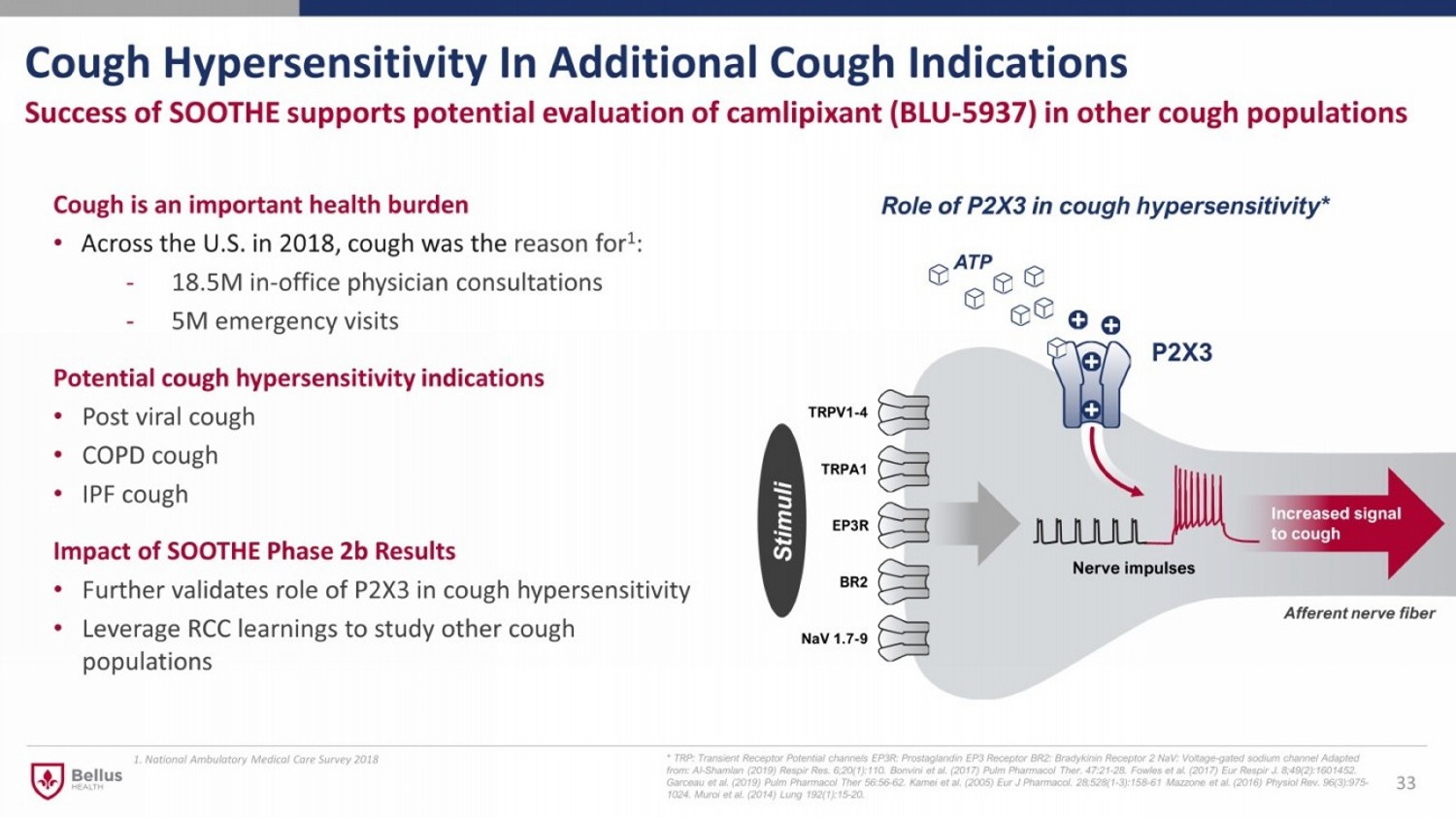
Cough Hypersensitivity In Additional Cough Indications 33 Success of SOOTHE supports potential evaluation of camlipixant (BLU - 5937) in other cough populations Cough is an important health burden • Across the U.S. in 2018, cough was the reason for 1 : - 18.5M in - office physician consultations - 5M emergency visits Potential cough hypersensitivity indications • Post viral cough • COPD cough • IPF cough Impact of SOOTHE Phase 2b Results • Further validates role of P2X3 in cough hypersensitivity • Leverage RCC learnings to study other cough populations * TRP: Transient Receptor Potential channels EP3R: Prostaglandin EP3 Receptor BR2: Bradykinin Receptor 2 NaV : Voltage - gated sodium channel Adapted from: Al - Shamlan (2019) Respir Res . 6;20(1):110. Bonvini et al. (2017) Pulm Pharmacol Ther . 47:21 - 28. Fowles et al. (2017) Eur Respir J. 8;49(2):1601452. Garceau et al. (2019) Pulm Pharmacol Ther 56:56 - 62 . Kamei et al. (2005) Eur J Pharmacol . 28;528(1 - 3):158 - 61 Mazzone et al. (2016) Physiol Rev. 96(3):975 - 1024 . Muroi et al. (2014) Lung 192(1):15 - 20. ATP Stimuli Afferent nerve fiber Role of P2X3 in cough hypersensitivity* 1. National Ambulatory Medical Care Survey 2018

IP and Corporate Summary

100% Owned Intellectual Property Portfolio 35 N N X N R 1 R 3 O N O R 9 R 2 R 4 R 6 R 5 R 8 R 7 • All intellectual property 100% owned by BELLUS with no future obligations owed • U.S. and international patent estate covering camlipixant and related compounds • Composition of matter patent for camlipixant and related P2X3 antagonists granted in the U.S., Europe, Japan, and China (expires in 2034 not including potential patent term extension) • Method of use patent for the treatment of cough granted in the U.S. (expires 2038) Camlipixant (BLU - 5937) composition of matter patent expires in 2034

Stock and Financial Information 36 126.6M basic shares 138.9M fully diluted shares CAPITAL STRUCTURE Cash, cash equivalent and short - term investments position of US$337.1M* CASH POSITION * Cash position as of December 31, 2022

EXPECTED MILESTONES Potential Catalysts & Upcoming Events 37 Camlipixant (BLU - 5937) in Refractory Chronic Cough Camlipixant Platform CALM - 1 & CALM - 2 trial initiations (Q4 2022) Topline results from CALM - 1 (2H 2024) Topline results from CALM - 2 (2025) Once - daily extended release formulation Phase 1 trial initiation (Q4 2022) Topline results from Phase 1 QD formulation (Q2 2023) x Third Party P2X3 Programs Merck’s gefapixant FDA resubmission (1H 2023) Conferences AAAAI (Feb. 24 - 27, 2023) ASCPT (Mar. 22 - 24, 2023) ATS (May 19 - 24, 2023) EXPECTED EVENTS x Corporate Events Analyst Day (2H 2023) ACC (June 9 - 10, 2023) ERS (Sept. 9 - 13, 2023) x

Investor Contact: Ramzi Benamar CFO investors@bellushealth.com





































