TABLE OF CONTENTS
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT
TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the month of: November, 2007
Commission File Number: 000-50393
NEUROCHEM INC.
275 Armand-Frappier Boulevard
Laval, Québec
H7V 4A7
Laval, Québec
H7V 4A7
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40 F.
Form 20-F¨ Form 40-Fþ
Form 20-F¨ Form 40-Fþ
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1):
Yes¨ Noþ
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7):
Yes¨ Noþ
Indicate by check mark whether the registrant by furnishing the information contained in this Form is also thereby furnishing the information to the Commission pursuant to Rule 12g-3 under the Securities Exchange Act of 1934.
Yes¨ Noþ
If “Yes” is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b):
SIGNATURES:
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| NEUROCHEM INC. | ||||
| November 8, 2007 | ||||
| By: | /s/ David Skinner | |||
| David Skinner, Vice-President | ||||
| General Counsel and Corporate Secretary | ||||
Table of Contents
 | Neurochem Inc. 275 Armand-Frappier Blvd. Laval, Quebec Canada H7V 4A7 |
For further information, please contact:
| Lise Hébert, Ph.D. Vice President, Corporate Communications | Tel.: 450-680-4572 lhebert@neurochem.com |
NEUROCHEM REPORTS RESULTS
FOR THIRD QUARTER OF FISCAL 2007
FOR THIRD QUARTER OF FISCAL 2007
Neurochem will host a live web conference Thursday November 8, 2007, at 8:30 AM ET.
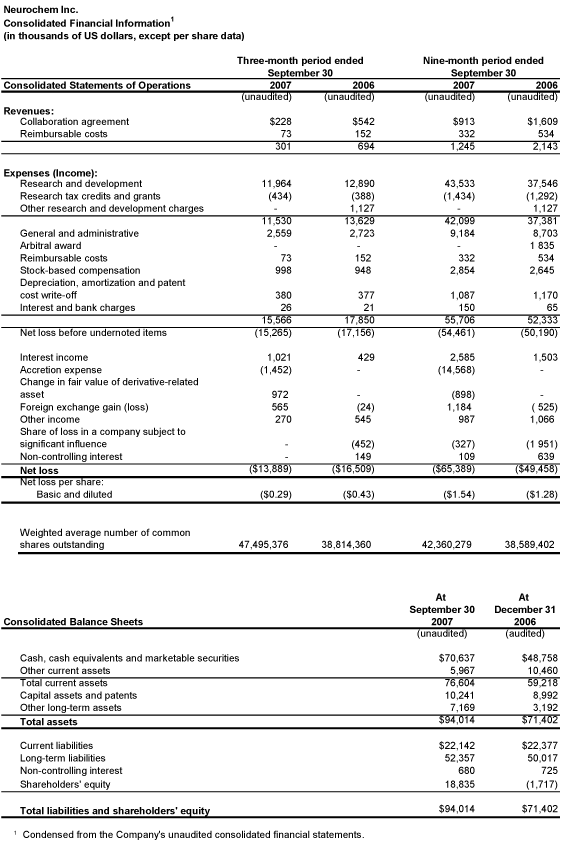
LAVAL, Quebec, November 7, 2007— Neurochem Inc. (NASDAQ: NRMX; TSX: NRM) reported results for the third quarter ended September 30, 2007. Effective July 1, 2007, the Company adopted the US dollar as its functional and reporting currency, thus all figures reported are reported in US dollars, unless otherwise specified. The Company reported a net loss of $13,889,000 ($0.29 per share), compared to $16,509,000 ($0.43 per share) for the corresponding period in the previous year. For the nine-month period ended September 30, 2007, the net loss amounted to $65,389,000 ($1.54 per share), compared to $49,458,000 ($1.28 per share) for the same period last year.
The net loss for the nine-month period ended September 30, 2007, includes a non-recurring charge in the second quarter of fiscal 2007 under Canadian GAAP of $10,431,000 relating to the $40 million 5% senior subordinated convertible notes, which were fully converted into common shares during the second quarter of 2007.
Research and development (R&D) expenses amounted to $11,964,000 this quarter compared to $12,890,000 for the same period last year. For the nine-month period, R&D expenses were $43,533,000 compared to $37,546,000 for the corresponding period of the previous year. The increase in the nine-month period compared to the same period the previous year is due to expenses incurred in relation to the development of tramiprosate (ALZHEMED™) primarily in respect of the ongoing Phase III clinical trial in Europe and the North American open-label extension of the Phase III study, as well as the conduct of a QT cardiac status Phase I study. Tramiprosate (ALZHEMED™) is the Company’s investigational product candidate for the treatment of Alzheimer’s disease (AD).
As at September 30, 2007, the Company had available cash, cash equivalents and marketable securities of $70,637,000, compared to $48,758,000 at December 31, 2006.
1
Table of Contents
The increase is primarily due to proceeds received from the issue of convertible notes in May 2007 and is partially offset by funds used in operating activities.
Live Web Cast and Teleconference
Neurochem will host a teleconference and web cast at 8:30 A.M., E.T., Thursday, November 8, 2007. The live web cast (audio and visual) will be available on the Company’s web site atwww.neurochem.com. The telephone numbers to access the audio portion of the presentation only are (514) 868-1042 or 1 (866) 862-3907.
Three hours following the teleconference and the web cast, a replay of the presentation will be available until November 15, 2007. The telephone numbers to access the audio replay are (514) 861-2272 or 1 (800) 408-3053, passcode 3241051#. The replay of the web cast (audio and visual) will be available on the Company’s web site.
The dial-in number will allow participants to listen and ask questions, while the web cast will host a visual presentation. Please dial-in or access Neurochem’s web site approximately 15 minutes before the teleconference is scheduled to begin.
Consolidated Financial Results Highlights
The following discussion and analysis should be read in conjunction with the Company’s unaudited consolidated financial statements for the nine-month period ended September 30, 2007, as well as the Company’s audited consolidated financial statements for the year ended December 31, 2006, which have been prepared in accordance with Canadian generally accepted accounting principles (GAAP). For discussion regarding related-party transactions, contractual obligations, disclosure controls and procedures, internal control over financial reporting, critical accounting policies and estimates, recent accounting pronouncements, and risks and uncertainties, refer to the Annual Report and the Annual Information Form for the year ended December 31, 2006, as well as registration statements and other public filings, which are available on SEDAR at www.sedar.com or on EDGAR atwww.sec.gov.
Results of Operations
As previously reported, effective July 1, 2007, the Company adopted the US dollar as its functional and reporting currency, as a significant portion of its revenue, expenses, assets, liabilities and financing are denominated in US dollars. All currency figures reported in the third quarter financial statements and in this document, including comparative figures, are reported in US dollars, unless otherwise specified.
For the three-month period ended September 30, 2007, thenet lossamounted to $13,889,000 ($0.29 per share), compared to $16,509,000 ($0.43 per share) for the corresponding period in the previous year. For the nine-month period ended September 30, 2007, the net loss amounted to $65,389,000 ($1.54 per share), compared to $49,458,000 ($1.28 per share) for the same period last year.
The net loss for the nine-month period ended September 30, 2007, includes a non-recurring charge in the second quarter of fiscal 2007 under Canadian GAAP of $10,431,000 relating to the $40 million 5% senior subordinated convertible notes, which were fully converted into common shares during the second quarter of 2007. In total,
2
Table of Contents
accretion expense amounted to $14,568,000 for the nine-month period ended September 30, 2007.
Revenue from collaboration agreementamounted to $228,000 for the current quarter ($913,000 for the nine-month period), compared to $542,000 for the same period in the previous year ($1,609,000 for the nine-month period). This revenue is earned under the agreement with Centocor, Inc. (Centocor) in respect of eprodisate (KIACTA™), an oral investigational product candidate for the treatment of Amyloid A (AA) amyloidosis. Revenue recognized is in respect of the non-refundable upfront payment received from Centocor, which is being amortized over the estimated period through to the anticipated regulatory approval date of the investigational product candidate. The estimated period is subject to change based on additional information that the Company may receive periodically. The other portion of the upfront payment received from Centocor ($6,000,000) has been classified as deferred revenue and is not being amortized as earned revenue given that it is potentially refundable. In the event that the Company receives an approval letter issued by the US Food and Drug Administration (FDA), the amount would no longer be refundable and would be amortized as earned revenue. In July 2007, the Company received a second approvable letter from the FDA for eprodisate (KIACTA™) for the treatment of AA amyloidosis. In this action letter, the FDA indicated that an additional efficacy trial will be necessary before the FDA could approve the investigational product candidate. The approvable letter also states that additional submissions, filed by Neurochem as part of its response to this approvable letter, may address issues raised in this letter. The FDA has indicated that additional submissions could persuade the agency to eliminate the requirement for an additional trial. The FDA also asked for additional information, including further pharmacokinetic studies, and again acknowledged that a QT clinical study should be submitted as part of a Phase IV (post-approval) commitment. The Company filed a response to this second approvable letter in late September 2007. Neurochem has also submitted for marketing approval for eprodisate (KIACTA™) for the treatment of AA amyloidosis in the European Union and Switzerland. In September 2006, the European Medicines Agency (EMEA) confirmed that it had commenced a regulatory review of eprodisate (KIACTA™). An oral hearing is scheduled with the EMEA in November 2007 to discuss outstanding issues raised with respect to the eprodisate (KIACTATM) application and a decision from the EMEA is expected by year end, or shortly thereafter. The Marketing Authorization Application is being reviewed under the EMEA’s centralized procedure. An authorization from the EMEA would apply to all 27 European Union member states, as well as Norway and Iceland.
Reimbursable costs revenueamounted to $73,000 for the current quarter ($332,000 for the nine-month period), compared to $152,000 for the same period in the previous year ($534,000 for the nine-month period) and consists of costs reimbursable by Centocor in respect of eprodisate (KIACTA™)-related activities. The Company earns no margin on these reimbursable costs.
Research and development expenses, before research tax credits and grants, amounted to $11,964,000 for the current quarter ($43,533,000 for the nine-month period), compared to $12,890,000 for the same period in the previous year ($37,546,000 for the nine-month period). The increase in the nine-month period compared to the same period the previous
3
Table of Contents
year is due to expenses incurred in relation to the development of tramiprosate (ALZHEMED™), primarily in respect of the ongoing Phase III clinical trial in Europe and the North American open-label extension of the Phase III study, as well as the conduct of a QT cardiac status Phase I study. Tramiprosate (ALZHEMED™) is the Company’s investigational product candidate for the treatment of Alzheimer’s disease (AD), which completed its 18-month North American Phase III clinical trial during the first quarter of 2007. In August 2007, the Company announced top-line results from this trial, designed to assess the safety, efficacy and disease modification effect of tramiprosate (ALZHEMED™) for the treatment of AD. The North American Phase III clinical trial, despite the descriptive data showing numerical differences in favor of tramiprosate (ALZHEMED™), did not demonstrate a statistically significant difference in favor of the product candidate with respect to the primary endpoints over 18 months of treatment. However, a substantial difference observed in hippocampal volume did approach statistical significance. Due to significant interference from high between-site variations that complicated the statistical analyses beyond expectations, it was not possible to draw definitive conclusions with respect to the treatment effect of tramiprosate (ALZHEMED™). Neurochem has established a Special Advisory Board comprised of regulatory, medical and statistical experts from the fields of AD, therapeutics for the central nervous system, functional assessments, imaging, biomarkers, and clinical trial design. The mandate of the Special Advisory Board is to assist Neurochem in reviewing and analyzing the data from the North American Phase III clinical trial and to provide advice to Neurochem on the tramiprosate (ALZHEMED™) program. The North American Phase III clinical trial included 1,052 patients at 67 clinical centers across the US and Canada. All patients who completed the North American Phase III clinical trial were eligible to receive tramiprosate (ALZHEMED™) in an open-label extension of the Phase III study. Neurochem is also currently conducting a European Phase III clinical trial for tramiprosate (ALZHEMED™) for the treatment of AD, with 973 mild-to-moderate AD patients enrolled at 69 clinical centers in 10 European countries. In August 2007, Neurochem stopped patient screening activities as it had met its recruitment target. For the nine-month period ended September 30, 2007, research and development expenses also included costs incurred to support the North American Phase III clinical trial for tramiprosate (ALZHEMED™), the ongoing open-label extension of the eprodisate (KIACTA™) Phase II/III study, as well as ongoing drug discovery programs.
Research tax credits and grantsamounted to $434,000 this quarter ($1,434,000 for the nine-month period), compared to $388,000 for the corresponding period in the previous year ($1,292,000 for the nine-month period). Research tax credits represent refundable tax credits earned under the Quebec Scientific Research and Experimental Development Program for expenditures incurred in Quebec.
Other research and development chargesamounted to nil for the current quarter and nine-month period, compared to $1,127,000 for the quarter and nine-month period ended September 30, 2006. In 2006, the Quebec taxation authorities confirmed their position in the application of the tax credit program that denied tax credits on research and development taxable benefits relating to stock options for 2005 and prior years. Accordingly, management determined at that time that the criteria for recognition of these credits were no longer met and recorded a provision for these research tax credits.
4
Table of Contents
General and administrative expensestotaled $2,559,000 for the current quarter ($9,184,000 for the nine-month period), compared to $2,723,000 for the same quarter in the previous year ($8,703,000 for the nine-month period). These costs are incurred to support the overall activities of the Company.
Arbitral awardamounted to nil for the current quarter and nine-month period compared to nil for the quarter ended September 30, 2006 and $1,835,000 for the nine-month period ended September 30, 2006. This expense related to the dispute with Immtech Pharmaceuticals, Inc. (formerly known as Immtech International, Inc. (Immtech)), which came to a conclusion in January 2007 when Immtech, the University of North Carolina at Chapel Hill (UNC), and Georgia State University Research Foundation, Inc. filed with the Federal District Court for the Southern District of New York, U.S.A. a Notice of Voluntary Dismissal. The plaintiffs voluntarily dismissed their complaint against Neurochem in the Federal District Court without any payment, license, business agreement, concession or compromise by Neurochem.
Reimbursable costsamounted to $73,000 for the current quarter ($332,000 for the nine-month period), compared to $152,000 for the same period in the previous year ($534,000 for the nine-month period), and consist of costs incurred on behalf of Centocor in respect of eprodisate (KIACTA™)-related activities and reimbursable by Centocor.
Stock-based compensationamounted to $998,000 for the current quarter ($2,854,000 for the nine-month period), compared to $948,000 for the corresponding quarter in the previous year ($2,645,000 for the nine-month period). This expense relates to stock options and stock-based incentives, whereby compensation cost in relation to stock options is measured at fair value at the date of grant and is expensed over the award’s vesting period.
Interest incomeamounted to $1,021,000 for the current quarter ($2,585,000 for the nine-month period), compared to $429,000 for the same quarter in the previous year ($1,503,000 for the nine-month period). The increase is mainly attributable to higher average cash balances during the current periods, compared to the same periods in the previous year.
Accretion expenseamounted to $1,452,000 for the current quarter ($14,568,000 for the nine-month period), and mainly represents the imputed interest under GAAP on the $42,085,000 aggregate principal amount of 6% convertible senior notes issued in November 2006, as well as on the $40,000,000 6% senior convertible notes (Senior Notes) and $40,000,000 5% senior subordinated convertible notes (Junior Notes) issued in May 2007. The Company accretes the carrying values of the convertible notes to their face value through a charge to earnings over their expected lives of 60 months, 54 months and 1 month, respectively. Of the total accretion expense recorded in the nine-month period ended September 30, 2007, $10,431,000 relates to accretion expense on the Junior Notes, which were fully converted during the second quarter of 2007.
Change in fair value of derivative-related assetamounted to a gain of $972,000 for the current quarter (loss of $898,000 for the nine-month period) and represents the variation
5
Table of Contents
in the fair value of the embedded derivatives included in the aggregate $80,000,000 Senior and Junior Notes issued in May 2007.
Foreign exchange gainamounted to $565,000 for the current quarter (gain of $1,184,000 for the nine-month period), compared to a loss of $24,000 for the same quarter in the previous year (loss of $525,000 for the nine-month period). Foreign exchange gains or losses arise on the movement in foreign exchange rates in relation to the Company’s net monetary assets held in currencies other than US dollars, which is its functional and reporting currency, and consists primarily of assets held in Canadian dollars.
Other incomeamounted to $270,000 for the current quarter ($987,000 for the nine-month period), compared to $545,000 for the same quarter in the previous year ($1,066,000 for the nine-month period). Other income consists of non-operating revenue, primarily sub-lease revenue. The previous year’s quarter includes an amount of $293,000 in respect of the recovery of prior years’ property taxes.
Share of loss in a company subject to significant influenceamounted to nil for the current quarter ($327,000 for the nine-month period), compared to $452,000 for the corresponding quarter in the previous year ($1,951,000 for the nine-month period).Non-controlling interestamounted to nil for the current quarter ($109,000 for the nine-month period), compared to $149,000 for the corresponding quarter in the previous year ($639,000 for the nine-month period). These items result from the consolidation of the Company’s interest in a holding company (Innodia Holding) that owns shares of Innodia Inc., for which Neurochem is the primary beneficiary. The share of loss recorded in the current year has reduced the Company’s long-term investment in Innodia Holding to a nominal value. Innodia Inc. is a private, development-stage company engaged in developing novel drugs for the treatment of type 2 diabetes and underlying diseases.
Liquidity and Capital Resources
As at September 30, 2007, the Company had available cash, cash equivalents and marketable securities of $70,637,000, compared to $48,758,000 at December 31, 2006. The increase is primarily due to proceeds received from the issue of convertible notes in May 2007 and is partially offset by funds used in operating activities.
On May 2, 2007, the Company issued $80,000,000 aggregate principal amount of convertible notes, consisting of $40,000,000 6% senior convertible notes due in 2027 and $40,000,000 5% senior subordinated convertible notes due in 2012. The 6% senior convertible notes have an initial conversion price equal to the lesser of $12.68 or the 5-day weighted average trading price of the common shares preceding any conversion, subject to adjustments in certain circumstances. The Company will pay interest on the 6% senior convertible notes until maturity on May 2, 2027, subject to earlier repurchase, redemption or conversion. The 5% senior subordinated convertible notes were subject to mandatory conversion into common shares under certain circumstances. In connection with this transaction, the Company issued warrants to purchase an aggregate of 2,250,645 common shares until May 2, 2012, at an initial purchase price of $12.68 per share, subject to adjustments in certain circumstances. During the quarter ended June 30, 2007, $10,500,000 of the 6% senior convertible notes were converted into 1,653,859 common shares and the totality of the 5% senior subordinated convertible notes were converted
6
Table of Contents
into 4,444,449 common shares. During the quarter ended September 30, 2007, an additional $25,000,000 6% senior convertible notes were converted into 3,965,462 common shares. Net proceeds from the offering were $74,279,000 and, as of September 30, 2007, $46,239,000 has yet to be spent. The use of proceeds continues to conform in all material respects with the expectations set forth in the documents filed publicly.
In August 2006, the Company entered into a securities purchase agreement in respect of an equity line of credit facility (ELOC) with Cityplatz Limited (Cityplatz), that provides the Company up to $60,000,000 of funds in return for the issuance of common shares at a discount of 3.0% to market price at the time of draw downs over term, less a placement fee equal to 2.4% of gross proceeds payable to the placement agent, Rodman & Renshaw, LLC. The ELOC established by the securities purchase agreement will terminate on February 9, 2009. The ELOC shall also terminate if (i) the Company’s common shares are de-listed from NASDAQ unless the common shares are listed at such time on another trading market specified in the agreement and such de-listing is in connection with a subsequent listing on another trading market specified in the agreement, (ii) the Company is subject to a change of control transaction or (iii) the Company suffers a material adverse effect which cannot be cured prior to the next drawdown notice. The Company may terminate the securities purchase agreement (i) if Cityplatz fails to fund a properly notified drawdown within five trading days of the end of the applicable settlement period or (ii) after it has drawn down at least $25,000,000 under the ELOC. Either party may also terminate the securities purchase agreement if the volume-weighted average price of the Company’s common shares is below $5 per share for more than 30 consecutive trading days. Given that the current price per share has been below the minimum price as per the agreement, the agreement may be terminated at any time. The parties are currently in discussions with respect to the future prospects of this agreement and no assurance can be given that any agreement may be reached. As at September 30, 2007, the Company had not drawn any funds under the ELOC.
As previously reported, “Restricted Cash” presented on the Consolidated Balance Sheet represents investments pledged to the bank to secure letters of credit. As at September 30, 2007, these investments are composed of Asset-Backed Commercial Paper (ABCP). During the third quarter of 2007, a disruption in the credit markets, particularly in the ABCP market, resulted in these investments having matured but not having been paid, and they currently remain outstanding. At the time these investments were acquired, the ABCP was rated R1-high by Dominion Bond Rating Service, which is the highest credit rating for this type of investment. At the present time, the credit rating is under review by the rating agency. On September 6, 2007, a Pan Canadian Committee was formed to oversee the proposed restructuring process of the ABCP. Also during the third quarter of 2007, the $6,000,000 letter of credit was renewed upon annual expiry and was extended to September 30, 2008, with the ABCP as collateral. The Company is monitoring the developments and restructuring process, and potential losses, if any, are presently indeterminable.
As at September 30, 2007, the Company’s workforce comprised 172 employees.
As at October 31, 2007, the Company had 48,846,595 common shares outstanding, 220,000 common shares issuable to the Chief Executive Officer upon the achievement of
7
Table of Contents
specified performance targets, 2,738,934 options granted under the stock option plan, 2,884,471 shares currently issuable under the convertible notes, and 2,250,645 warrants outstanding, for a total of 56,940,645 common shares, on a fully diluted basis.
The Company believes that its available cash and short-term investments, expected interest income, potential funding from partnerships, research collaborations and licensing agreements, potential proceeds from the ELOC, research tax credits, grants, and access to capital markets should be sufficient to finance the Company’s operations and capital needs during the ensuing year. However, in light of the uncertainties associated with the regulatory approval process, clinical trial results, and the Company’s ability to secure additional licensing, partnership and/or other agreements, further financing may be required to support the Company’s operations in the future.
Change in functional and reporting currency
Effective July 1, 2007, the Company adopted the US dollar as its functional and reporting currency, as a significant portion of its revenues, expenses, assets, liabilities and financing are denominated in US dollars. Prior to that date, the Company’s operations were measured in Canadian dollars and the consolidated financial statements were expressed in Canadian dollars. The Company followed the recommendations of the Emerging Issues Committee (EIC) of the Canadian Institute of Chartered Accountants (CICA), set out in EIC-130, “Translation method when the reporting currency differs from the measurement currency or there is a change in the reporting currency”. In accordance with EIC-130, assets and liabilities as of June 30, 2007 were translated in US dollars using the exchange rate in effect on that date; revenues, expenses and cash flows were translated at the average rate in effect during the six-month period ended June 30, 2007 and equity transactions were translated at historical rates. For comparative purposes, historical financial statements have been restated into US dollars using the current rate method. Under this method, assets and liabilities are translated at the closing rate in effect at the end of these periods, revenues, expenses and cash flows are translated at the average rates in effect during these periods and equity transactions are translated at historical rates. Any exchange differences resulting from the translation are included in accumulated other comprehensive income presented in shareholders’ equity.
8
Table of Contents
9
Table of Contents
About Neurochem
Neurochem Inc. is focused on the development and commercialization of innovative therapeutics to address critical unmet medical needs. Eprodisate (KIACTA™) is currently being developed for the treatment of Amyloid A amyloidosis, and is under regulatory review for marketing approval by the U.S. Food and Drug Administration, the European Medicines Agency and Swissmedic. Tramiprosate (ALZHEMED™), for the treatment of Alzheimer’s disease, has been the subject of a completed Phase III clinical trial in North America and is currently being studied in a Phase III clinical trial in Europe, while tramiprosate (CEREBRIL™), for the prevention of hemorrhagic stroke caused by cerebral amyloid angiopathy, has been studied in a completed Phase IIa clinical trial.
To Contact Neurochem
For additional information on Neurochem and its drug development programs, please call the North American toll-free number 1 (877) 680-4500 or visit the Web site atwww.neurochem.com.
Certain statements contained in this news release, other than statements of fact that are independently verifiable at the date hereof, may constitute forward-looking statements. Such statements, based as they are on the current expectations of management, inherently involve numerous risks and uncertainties, known and unknown, many of which are beyond Neurochem’s control. Such risks include but are not limited to: the impact of general economic conditions, general conditions in the pharmaceutical industry, changes in the regulatory environment in the jurisdictions in which Neurochem does business, stock market volatility, fluctuations in costs, and changes to the competitive environment due to consolidation, that actual results may vary once the final and quality-controlled verification of data and analyses has been completed, as well as other risks disclosed in public filings of Neurochem. Consequently, actual future results may differ materially from the anticipated results expressed in the forward-looking statements. The reader should not place undue reliance, if any, on the forward-looking statements included in this news release. These statements speak only as of the date made and Neurochem is under no obligation and disavows any intention to update or revise such statements as a result of any event, circumstances or otherwise. Please see the Annual Information Form for further risk factors that might affect the Company and its business.
10
Table of Contents
 | Neurochem Inc. 275 Armand-Frappier Blvd. Laval, Quebec Canada H7V 4A7 |
For further information, please contact:
| Lise Hébert, PhD Vice President, Corporate Communications | Tel.: 450-680-4572 lhebert@neurochem.com |
NEUROCHEM ANNOUNCES IMPORTANT INITIATIVES
TO PROVIDE MEDICAL AND HEALTH BENEFITS TO PATIENTS
Neurochem will host a live web conference today November 8, 2007, at 8:30 AM ET
TO PROVIDE MEDICAL AND HEALTH BENEFITS TO PATIENTS
Neurochem will host a live web conference today November 8, 2007, at 8:30 AM ET
LAVAL, CANADA, November 8, 2007 —Neurochem Inc. (NASDAQ: NRMX, TSX: NRM) announces today important initiatives including the expansion of its product candidate portfolio, the refocusing of its proprietary R&D assets and the expected reduction of its burn rate compared to fiscal 2007. As part of these initiatives, Neurochem is:
| • | Moving its next generation lead candidate NRM-8499 into preclinical development for the treatment of Alzheimer’s disease (AD) and is also announcing the early termination of its European Phase III clinical trial for tramiprosate (ALZHEMED™). |
| • | Taking steps, in light of promising results from preliminary post-hoc analysis of the data from the North American Phase III trial for tramiprosate, to commercialize this product as a branded nutraceutical, starting as early as 2008. |
| • | Continuing to advance its programs for Amyloid A (AA) amyloidosis as well as for Diabetes Type II/features of metabolic syndrome. |
“Our goal is to provide innovative health solutions to patients suffering from serious diseases as soon as possible,” said Dr. Francesco Bellini, Chairman, President and CEO of Neurochem. “With the strategy unveiled today, the Company plans to make tramiprosate available to consumers. As well, Neurochem is confirming its commitment to developing new drugs for AD by advancing into preclinical development a prodrug which improves tramiprosate concentration in the brain several fold, thus leveraging the wealth of knowledge the Company has gained from the work accomplished to date in this field. Neurochem will be dedicating its resources to building a strong and diversified pipeline of product candidates, which could potentially provide cash flow in the near term. As a result, our business model is expanding beyond the traditional biopharmaceutical strategy to one that embraces shorter term projects. These projects should fund research and development activities, while complementing our longer term prescription drug programs geared at sustaining the Company’s future growth,” he concluded.
Table of Contents
Seeking Medical and Health Benefits in Significant Therapeutic Areas
Alzheimer’s disease — Next Generation — Tramiprosate (ALZHEMED™)
As part of the initiatives announced today, Neurochem will leverage the numerous years of accumulated knowledge and the experience it has gained in developing tramiprosate (ALZHEMED™) for AD, and will prioritize and accelerate the development of its next generation lead compound NRM-8499, a new chemical entity (NCE) and a prodrug1 of tramiprosate for the treatment of AD. Pharmacokinetic studies performed in mice with NRM-8499 have already demonstrated greater than five-fold increases in brain concentration as compared to the original tramiprosate molecule.
As part of the initiatives announced today, Neurochem will leverage the numerous years of accumulated knowledge and the experience it has gained in developing tramiprosate (ALZHEMED™) for AD, and will prioritize and accelerate the development of its next generation lead compound NRM-8499, a new chemical entity (NCE) and a prodrug1 of tramiprosate for the treatment of AD. Pharmacokinetic studies performed in mice with NRM-8499 have already demonstrated greater than five-fold increases in brain concentration as compared to the original tramiprosate molecule.
As announced previously, descriptive data from the North American Phase III clinical trial shows numerical differences in favor of tramiprosate on the primary clinical endpoints. The descriptive data also shows by magnetic resonance imaging differences between groups on the primary disease modification endpoint of change in the volume of the hippocampus, a structure of the brain that is considered to be important in memory function.
Additional findings obtained from a preliminary post-hoc analysis performed by the Company’s external team of statisticians that allowed adjustment for potential confounding factors showed a dose-dependent reduction in hippocampal atrophy in patients treated with tramiprosate. When compared to placebo, patients treated with 200 mg of tramiprosate per day experienced significantly less atrophy (relative difference of 65%; P = 0.036) and patients treated with tramiprosate with 300 mg of tramiprosate per day presented no atrophy (P = 0.003).
The Company is committed to analyzing and understanding the wealth of data generated by the Phase III clinical trials. It is pursuing several post-hoc analyses which are revealing promising preliminary results. Neurochem is currently working to submit results for publication in a peer-reviewed journal.
Nutraceutical Initiative
In view of these results, coupled to the large number of physicians and families requesting access to the compound, and given that tramiprosate occurs naturally in certain foods, Neurochem is planning to provide commercial access to tramiprosate as a branded nutraceutical product, potentially as early as 2008, via the creation of a new self-sustaining company. This decision was taken in view of the fact that AD is a devastating disease and remains an unmet medical need.
In view of these results, coupled to the large number of physicians and families requesting access to the compound, and given that tramiprosate occurs naturally in certain foods, Neurochem is planning to provide commercial access to tramiprosate as a branded nutraceutical product, potentially as early as 2008, via the creation of a new self-sustaining company. This decision was taken in view of the fact that AD is a devastating disease and remains an unmet medical need.
Early discontinuation of Phase III Tramiprosate Clinical Trial in Europe
In light of the information gathered to date, Neurochem was faced with the decision of completing the European Phase III clinical trial and/or initiating another Phase III study to support the approval of tramiprosate by regulatory agencies and/or investing in the development of a next generation compound related to the original product candidate. Considering the current scientific and medical knowledge about AD, and the changing environment with widespread use of currently available medications, Management strongly believes that the expected lengthy and costly clinical and regulatory paths for tramiprosate prohibit its further development as a pharmaceutical entity.
In light of the information gathered to date, Neurochem was faced with the decision of completing the European Phase III clinical trial and/or initiating another Phase III study to support the approval of tramiprosate by regulatory agencies and/or investing in the development of a next generation compound related to the original product candidate. Considering the current scientific and medical knowledge about AD, and the changing environment with widespread use of currently available medications, Management strongly believes that the expected lengthy and costly clinical and regulatory paths for tramiprosate prohibit its further development as a pharmaceutical entity.
Table of Contents
The Special Advisory Board highlighted the inherent challenges associated with advancing Phase III clinical trials for AD with large cohorts of patients over very long periods of time. The challenges include enrolling the patients that have the right disease profile while dealing with a high degree of patient heterogeneity.
In summary, Neurochem strongly believes that the best course of action is to terminate early the drug development activities surrounding the European Phase III clinical trial for the study of tramiprosate, but to complete the ongoing North American Phase III open-label extension study. The Company will redirect its in-house AD efforts to the development of NRM-8499 and plans to make tramiprosate available as a branded nutraceutical as soon as possible.
AA amyloidosis
Following a second approvable letter received by the Company in July 2007, the United States Food and Drug Administration (FDA) is completing its review of Neurochem’s New Drug Application for eprodisate (KIACTA™) for the treatment of AA amyloidosis. Neurochem filed a response to the approvable letter in September 2007.
Following a second approvable letter received by the Company in July 2007, the United States Food and Drug Administration (FDA) is completing its review of Neurochem’s New Drug Application for eprodisate (KIACTA™) for the treatment of AA amyloidosis. Neurochem filed a response to the approvable letter in September 2007.
In September 2006, the European Medicines Agency (EMEA) confirmed that it had commenced a regulatory review of eprodisate (KIACTA™). The Marketing Authorization Application is being reviewed under the Agency’s centralized procedure. This means that an authorization from this Agency would apply to all 27 European Union member states, as well as Norway and Iceland. An oral hearing is scheduled with the EMEA later this month to discuss outstanding issues raised with respect to the eprodisate (KIACTA™) application and a decision from the EMEA is expected by year-end, or shortly thereafter.
The Company also filed for marketing approval for eprodisate (KIACTA™) for the treatment of AA amyloidosis in Switzerland. A decision is expected in 2008.
DiabetesType II/Features of Metabolic Syndrome
As announced in May 2007, eprodisate has shown beneficial effects in preclinicalin vivomodels of Diabetes Type II/features of metabolic syndrome. Preliminary results have shown that eprodisate protects the kidney function in obese diabetic rats. As well, eprodisate has shown an impact on metabolic changes associated with Diabetes and obesity, including a significant decrease of triglyceride levels and cholesterol, a significant decrease of glycemia and an increase in insulin plasma levels.
As announced in May 2007, eprodisate has shown beneficial effects in preclinicalin vivomodels of Diabetes Type II/features of metabolic syndrome. Preliminary results have shown that eprodisate protects the kidney function in obese diabetic rats. As well, eprodisate has shown an impact on metabolic changes associated with Diabetes and obesity, including a significant decrease of triglyceride levels and cholesterol, a significant decrease of glycemia and an increase in insulin plasma levels.
The Company is on track to initiate a Phase II clinical trial in diabetic patients in Canada in early 2008.
Live Web Conference
Neurochem will host a teleconference and web cast at 8:30 A.M., E.T., today, November 8, 2007. The live web cast (audio and visual) will be available on the Company’s web site at www.neurochem.com. The telephone numbers to access the audio portion of the presentation only are (514) 868-1042 or 1 (866) 862-3907.
Three hours following the teleconference and the web cast, a replay of the presentation will be available until November 15, 2007. The telephone numbers to access the audio
Table of Contents
replay are (514) 861-2272 or 1 (800) 408-3053, passcode 3241051#. The replay of the web cast (audio and visual) will be available on the Company’s web site.
The dial-in number will allow participants to listen and ask questions, while the web cast will host a visual presentation. Please dial-in or access Neurochem’s web site approximately 15 minutes before the teleconference is scheduled to begin.
About Neurochem
Neurochem Inc. is a global health company focused in research, development and commercialization of products to provide innovative health solutions to patients suffering from serious diseases.
To Contact Neurochem
For additional information on Neurochem and its drug development programs, please call the North American toll-free number 1 877 680-4500 or visit our Web Site at:www.neurochem.com.
| 1 | — | A prodrug is a pharmaceutical substance which is administered in an inactive form and once absorbed is metabolized in vivo into its active form (namely tramiprosate). |
Certain statements contained in this news release, other than statements of fact that are independently verifiable at the date hereof, may constitute forward-looking statements. Such statements, based as they are on the current expectations of management, inherently involve numerous risks and uncertainties, known and unknown, many of which are beyond Neurochem’s control. Such risks include but are not limited to: the impact of general economic conditions, general conditions in the pharmaceutical and/or nutraceutical industry, changes in the regulatory environment in the jurisdictions in which Neurochem does business, stock market volatility, fluctuations in costs, and changes to the competitive environment due to consolidation, that actual results may vary once the final and quality-controlled verification of data and analyses has been completed, as well as other risks disclosed in public filings of Neurochem. Consequently, actual future results may differ materially from the anticipated results expressed in the forward-looking statements. The reader should not place undue reliance, if any, on the forward-looking statements included in this news release. These statements speak only as of the date made and Neurochem is under no obligation and disavows any intention to update or revise such statements as a result of any event, circumstances or otherwise. Please see the Annual Information Form for further risk factors that might affect the Company and its business.