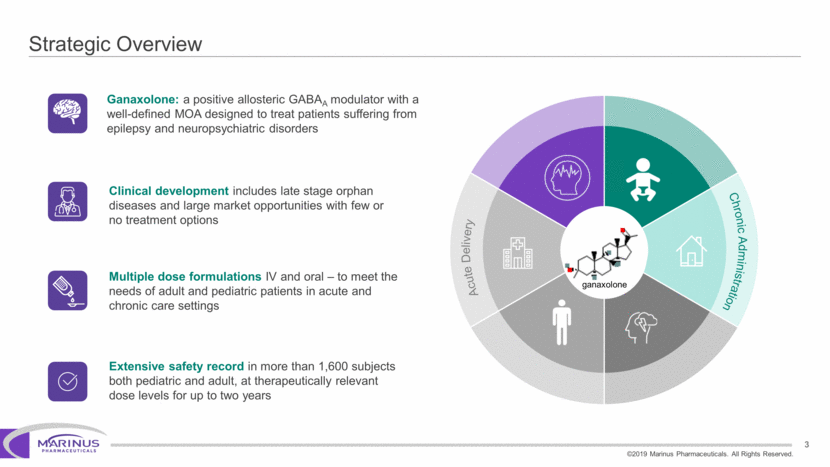
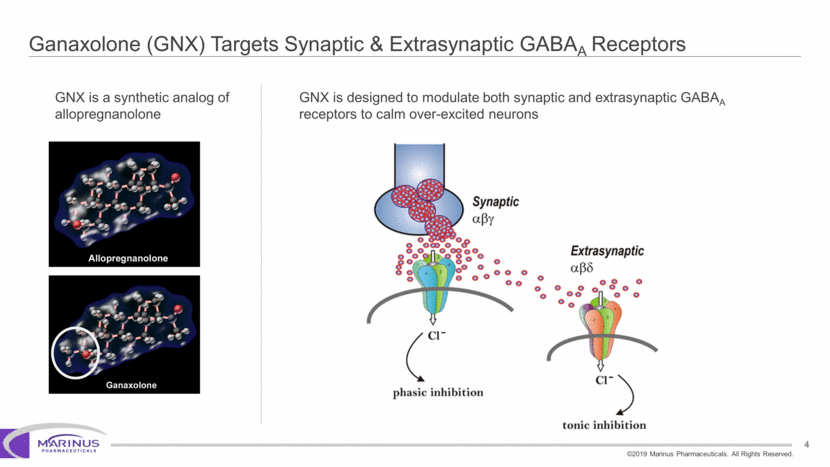
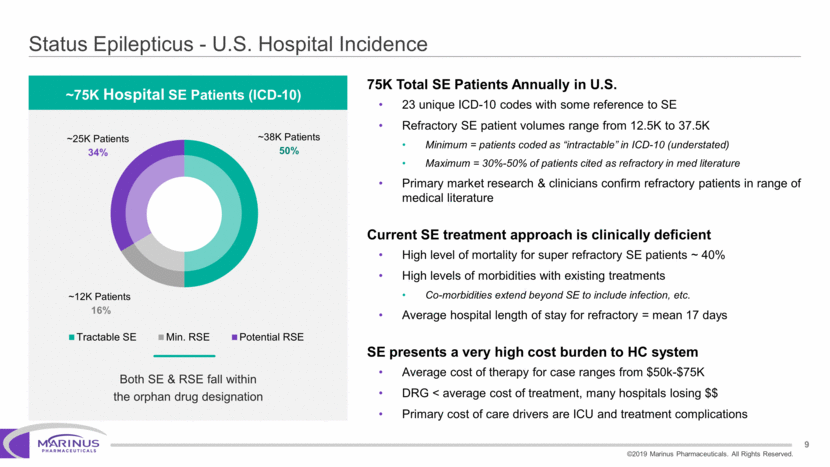
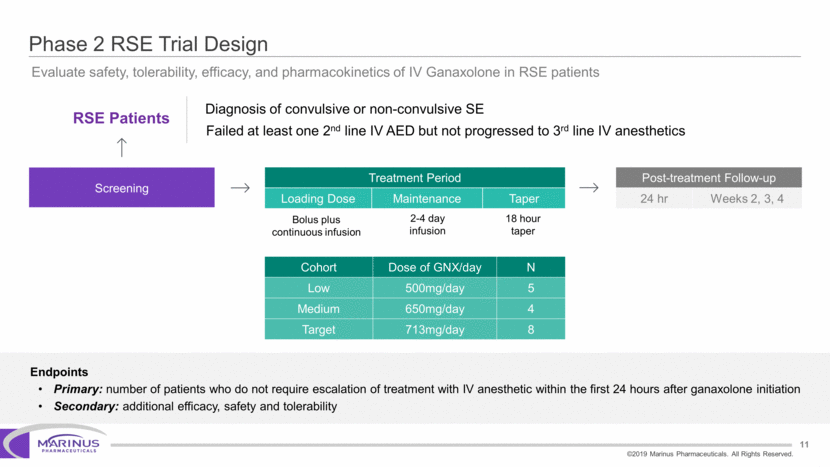
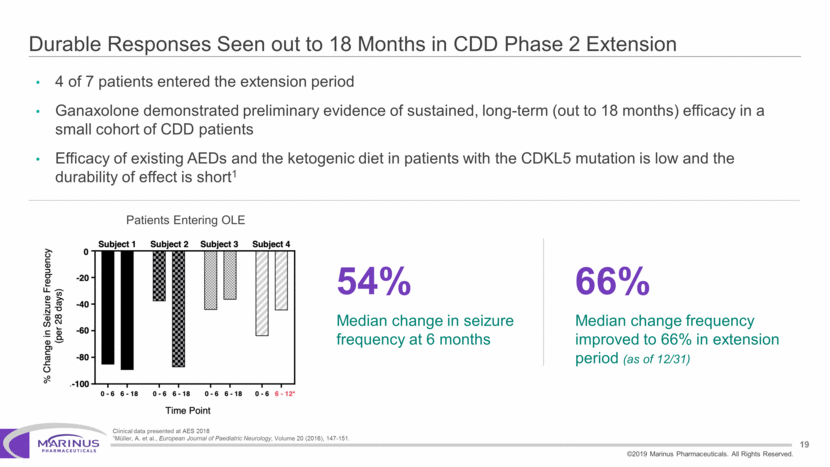
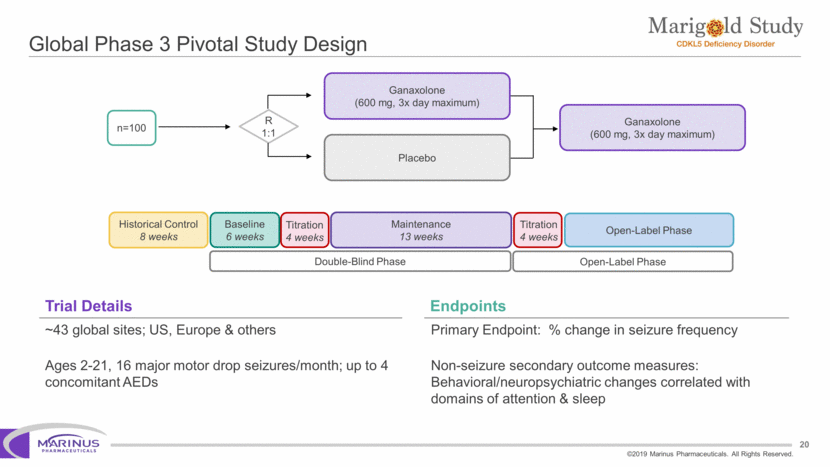
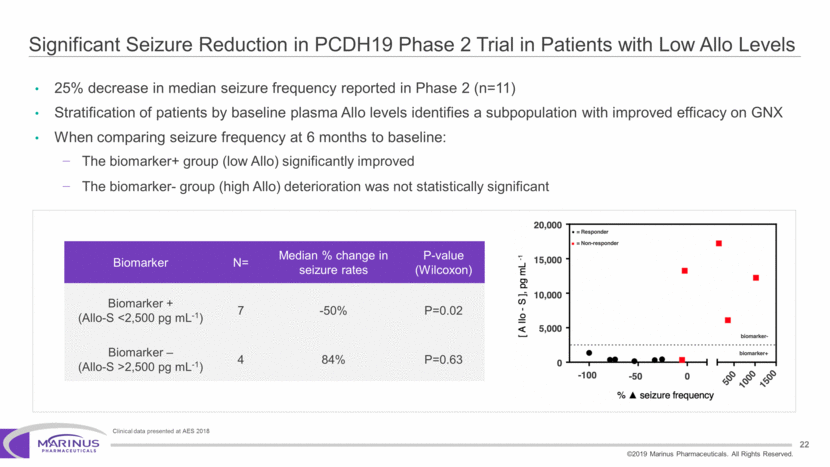
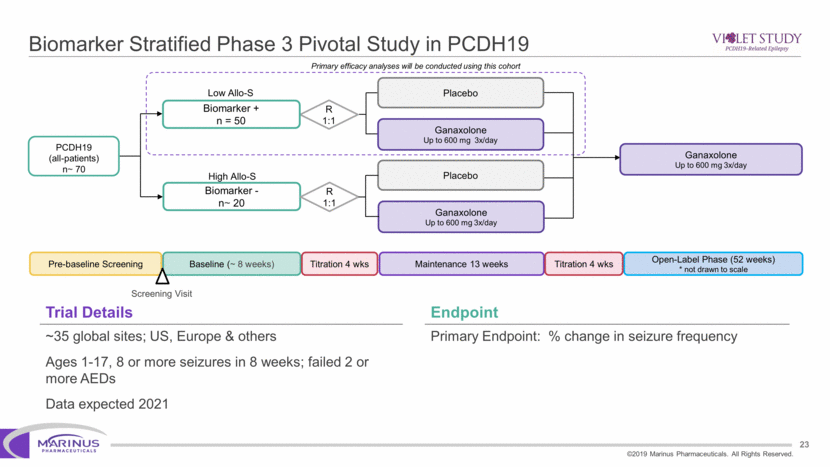
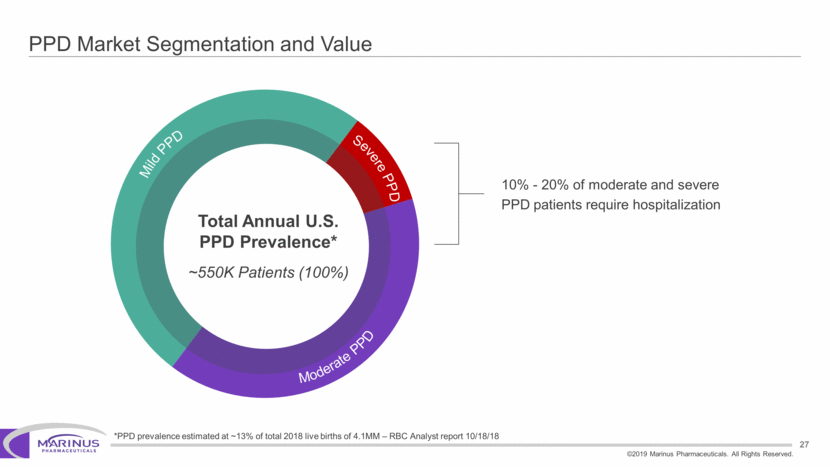
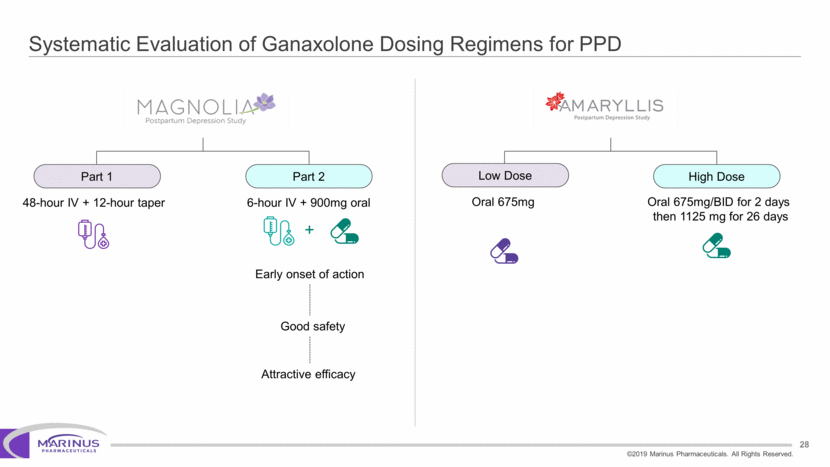
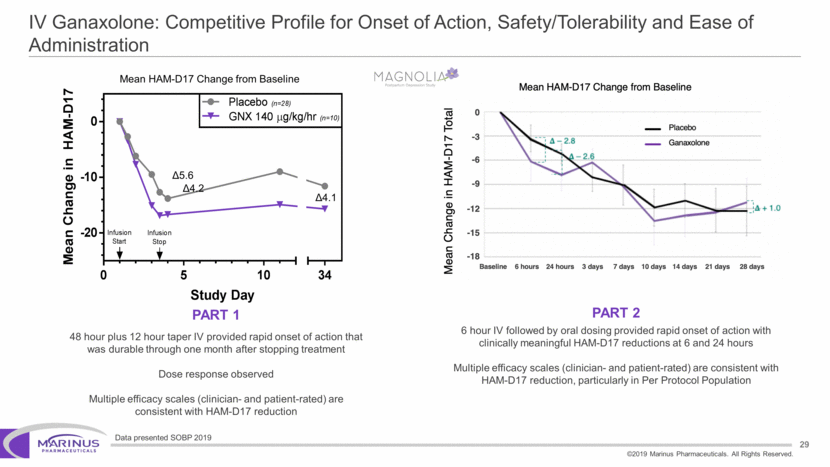
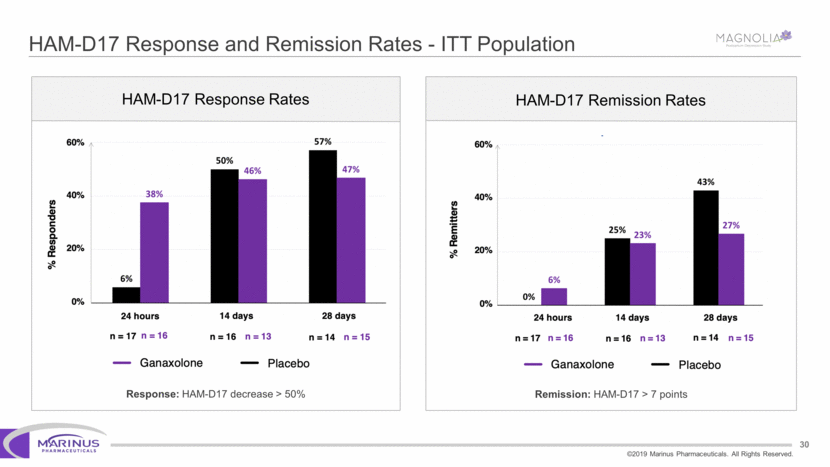
Safe Harbor Statement To the extent that statements contained in this presentation are not descriptions of historical facts regarding Marinus, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “may”, “will”, “expect”, “anticipate”, “estimate”, “intend”, “believe”, and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward-looking statements contained in this press release include, among others, statements regarding our interpretation of preclinical studies, development plans for our product candidate, including the development of dose forms, the clinical trial testing schedule and milestones, the ability to complete enrollment in our clinical trials, interpretation of scientific basis for ganaxolone use, timing for availability and release of data, the safety, potential efficacy and therapeutic potential of our product candidate and our expectation regarding the sufficiency of our working capital. Forward-looking statements in this release involve substantial risks and uncertainties that could cause our clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, the uncertainties inherent in the conduct of future clinical trials, the timing of the clinical trials, enrollment in clinical trials, availability of data from ongoing clinical trials, expectations for regulatory approvals, the attainment of clinical trial results that will be supportive of regulatory approvals, and other matters, including the development of formulations of ganaxolone, and the availability or potential availability of alternative products or treatments for conditions targeted by the company that could affect the availability or commercial potential of our drug candidates. Marinus undertakes no obligation to update or revise any forward-looking statements. For a further description of the risks and uncertainties that could cause actual results to differ from those expressed in these forward-looking statements, as well as risks relating to the business of the Company in general, see Marinus' 10-Q dated August 8, 2019 and other filings by the company with the U.S. Securities and Exchange Commission. You may access these documents for free by visiting EDGAR on the SEC web site at www.sec.gov. 2