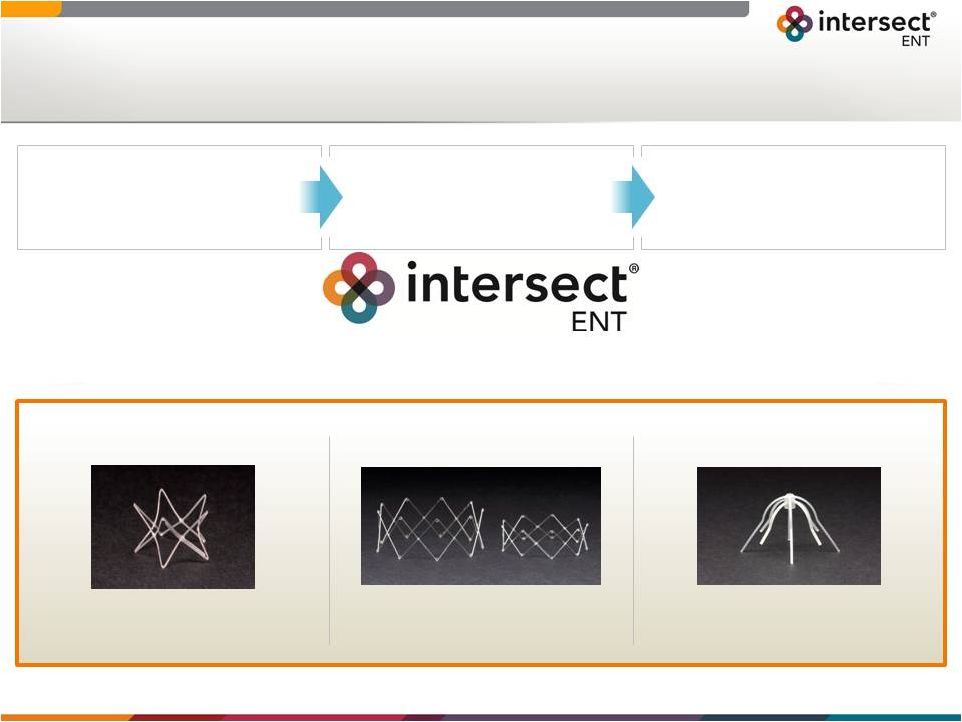
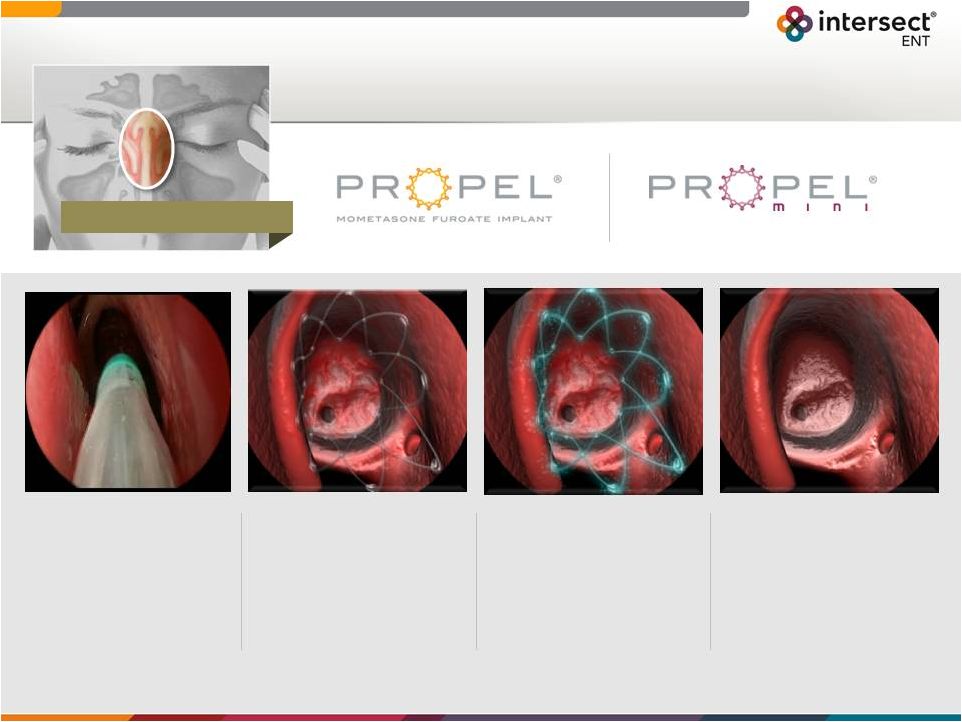
Forward-Looking Statements 1 Certain statements in this presentation constitute “forward-looking statements” within the meaning of the Securities Act of 1933, as amended (the “Securities Act”), and Securities Exchange Act of 1934, as amended (“Exchange Act”), including, without limitation, statements regarding our outlook for financial performance, sales force growth, clinical studies, approval of new products and indications and the receipt of reimbursement. We intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in the Securities Act and the Exchange Act and are making this statement for purposes of complying with those safe harbor provisions. These forward-looking statements reflect our current views about our plans, intentions, expectations, strategies and prospects, which are based on the information currently available to us and on assumptions we have made. Although we believe that our plans, intentions, expectations, strategies and prospects as reflected in or suggested by those forward-looking statements are reasonable, we can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a variety of risks and factors that are beyond our control, including those risks and uncertainties discussed under “Risk Factors” in our Form 10-K filing dated March 11, 2015 and subsequent quarterly filings with the SEC. All information in this presentation is as of the date of this presentation, and we undertake no duty to update this information unless required by law. |