
2020 10-K Conference Call September 9, 2020 Exhibit 99.2

Forward-Looking Statements This presentation contains forward-looking statements that reflect AGTC's plans, estimates, assumptions and beliefs. Forward-looking statements include statements regarding the timing for and expected expansion of its XLRP clinical development program, the timing for reporting data in its XLRP and ACHM clinical programs, and its ability to enroll patients, effectively design and successfully complete its ongoing clinical trials, financial guidance, business strategies and operations, regulatory progress, potential growth and market opportunities, and the effects of competition. Forward-looking statements include all statements that are not historical facts and can be identified by terms such as "anticipates," "believes," "could," "seeks," "estimates," "expects," "intends," "may," "plans," "potential," "predicts," "projects," "should," "will," "would" or similar expressions and the negatives of those terms. Actual results could differ materially from those discussed in the forward-looking statements, due to a number of important factors. Risks and uncertainties that may cause actual results to differ materially include, among others: AGTC cannot predict when or if it will obtain regulatory approval to commercialize a product candidate or receive reasonable reimbursement; uncertainty inherent in clinical trials and the regulatory review process; risks and uncertainties associated with drug development and commercialization; risks related to the COVID-19 outbreak that may delay clinical trials; gene therapy is still novel with only a few approved treatments so far; factors that could cause actual results to differ materially from those described in the forward-looking statements are set forth under the heading "Risk Factors" in the Company's most recent Annual Report on Form 10-K and subsequent periodic reports filed with the SEC. Given these uncertainties, you should not place undue reliance on these forward-looking statements. Also, forward-looking statements represent management's plans, estimates, assumptions and beliefs only as of the date of this presentation. Except as required by law, we assume no obligation to update these forward-looking statements publicly or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future.

Call Agenda Recent accomplishments and latest developments – Sue Washer, CEO Clinical program update & preclinical pipeline review – Dr. Mark S. Shearman, CSO 4Q 2020 financial results review – Bill Sullivan, CFO Q&A Closing Remarks – Sue Washer, CEO

Recent Accomplishments Received detailed EOP2 feedback from the FDA on the design of our Phase 2/3 protocol and future regulatory filings for our XLRP trial On track to initiate a Phase 2/3 trial in 1Q 2021 Expanding ongoing Phase 1/2 trial in order to dose approximately 12 patients in two masked dosing arms to collect additional data, including a mobility course added as a supplemental endpoint New preliminary data for Group 5 patients in the Phase 1/2 XLRP trial available Launched nationwide mobile vision testing to support our ongoing Phase 1/2 XLRP and ACHM trials Formalized our Patient Advisory Council to integrate patient and caregiver voices into our clinical and preclinical development strategies Initial focus on XLRP Insights from patients and caregivers with real-world understanding of XLRP and other inherited retinal disorders
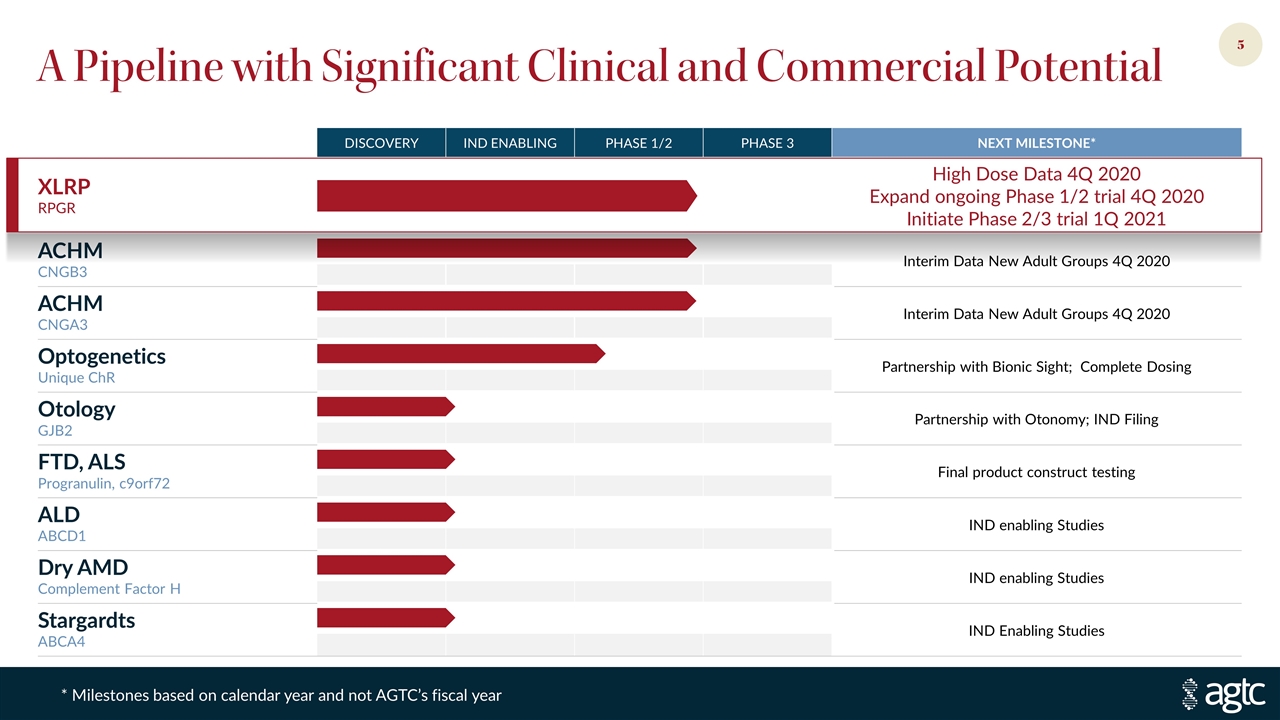
A Pipeline with Significant Clinical and Commercial Potential DISCOVERY IND ENABLING PHASE 1/2 PHASE 3 NEXT MILESTONE* XLRP RPGR High Dose Data 4Q 2020 Expand ongoing Phase 1/2 trial 4Q 2020 Initiate Phase 2/3 trial 1Q 2021 ACHM CNGB3 Interim Data New Adult Groups 4Q 2020 ACHM CNGA3 Interim Data New Adult Groups 4Q 2020 Optogenetics Unique ChR Partnership with Bionic Sight; Complete Dosing Otology GJB2 Partnership with Otonomy; IND Filing FTD, ALS Progranulin, c9orf72 Final product construct testing ALD ABCD1 IND enabling Studies Dry AMD Complement Factor H IND enabling Studies Stargardts ABCA4 IND Enabling Studies * Milestones based on calendar year and not AGTC’s fiscal year
Industry Leading Gene Therapy Company Poised for Success XLRP: Lead Product Candidate Advancing Toward Phase 2/3 Trial in 1Q 2021 Sustained Improvements in visual function for centrally dosed patients Expanding ongoing Phase 1/2 trial; expect dosing to begin in 4Q 2020 Expect enrollment of Phase 2/3 trial to begin in 1Q 2021 ACHM: Early Evidence of Biological Activity in Both ACHM Trials Dose escalation completed in adult patients Best-in-Class Technology Platform Process and scale expected to support late-phase development and commercialization Expertise in product design allows for best capsid, promoter and gene cassettes for each indication Robust use of disease specific animal modeling to guide clinical dosing Deep expertise and capabilities to support robust pipeline in ophthalmology, CNS, and otology

Positioned for Success in a Changing Landscape XLRP: Direct Path to Phase 2/3 in 1Q 2021 Expanding Phase 1/2 trial to increase total data set Received EOP2 FDA feedback Manufacturing of Phase 2/3 material on track Redundant manufacturing options contracted; CDMOs are considered essential businesses in their locations ACHM Adult enrollment complete in both trials 8 of 18 pediatric patients across both trials enrolled DSMC approval to highest pediatric dose

Clinical Programs Update

Leading XLRP Program Moving Toward 1Q 2021 Phase 2/3 Sustained treatment effect, favorable safety profile Evidence of increase and expansion in central visual sensitivity Encouraging improvements in visual acuity Quality of life survey results support meaningful impact on patients’ lives Phase 1/2 trial initial targeted enrollment complete—28 patients in six dose groups All patients dosed continue to demonstrate a favorable safety profile
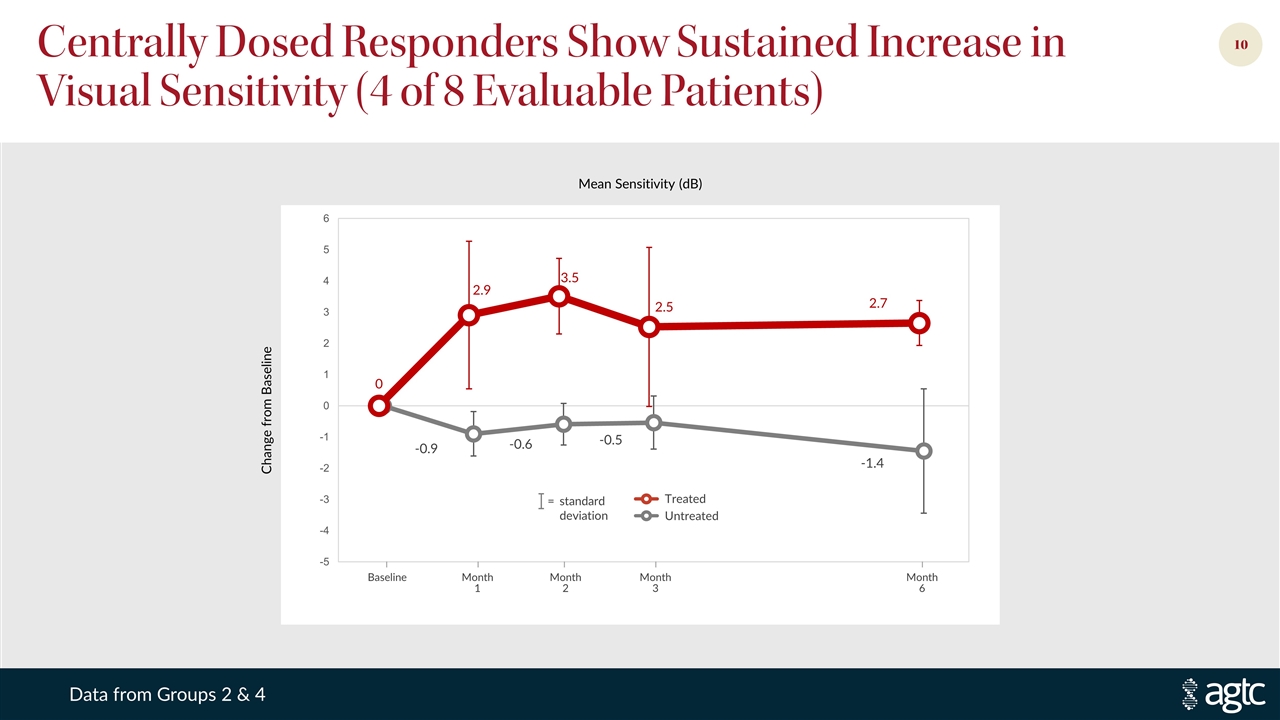
Centrally Dosed Responders Show Sustained Increase in Visual Sensitivity (4 of 8 Evaluable Patients) = standard deviation Treated Untreated Baseline Month 1 Month 2 Month 3 Month 6 Change from Baseline Mean Sensitivity (dB) Data from Groups 2 & 4
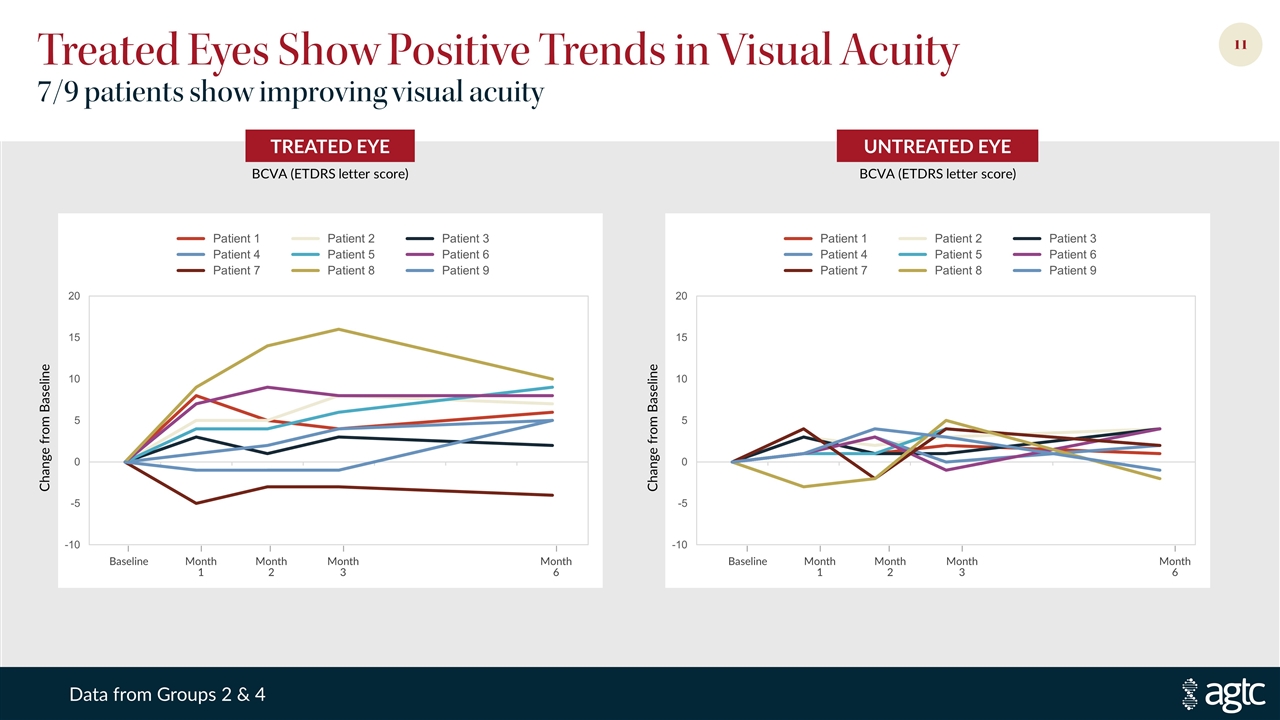
Treated Eyes Show Positive Trends in Visual Acuity 7/9 patients show improving visual acuity UNTREATED EYE TREATED EYE BCVA (ETDRS letter score) BCVA (ETDRS letter score) Change from Baseline Change from Baseline Baseline Month 1 Month 2 Month 3 Month 6 Baseline Month 1 Month 2 Month 3 Month 6 Data from Groups 2 & 4
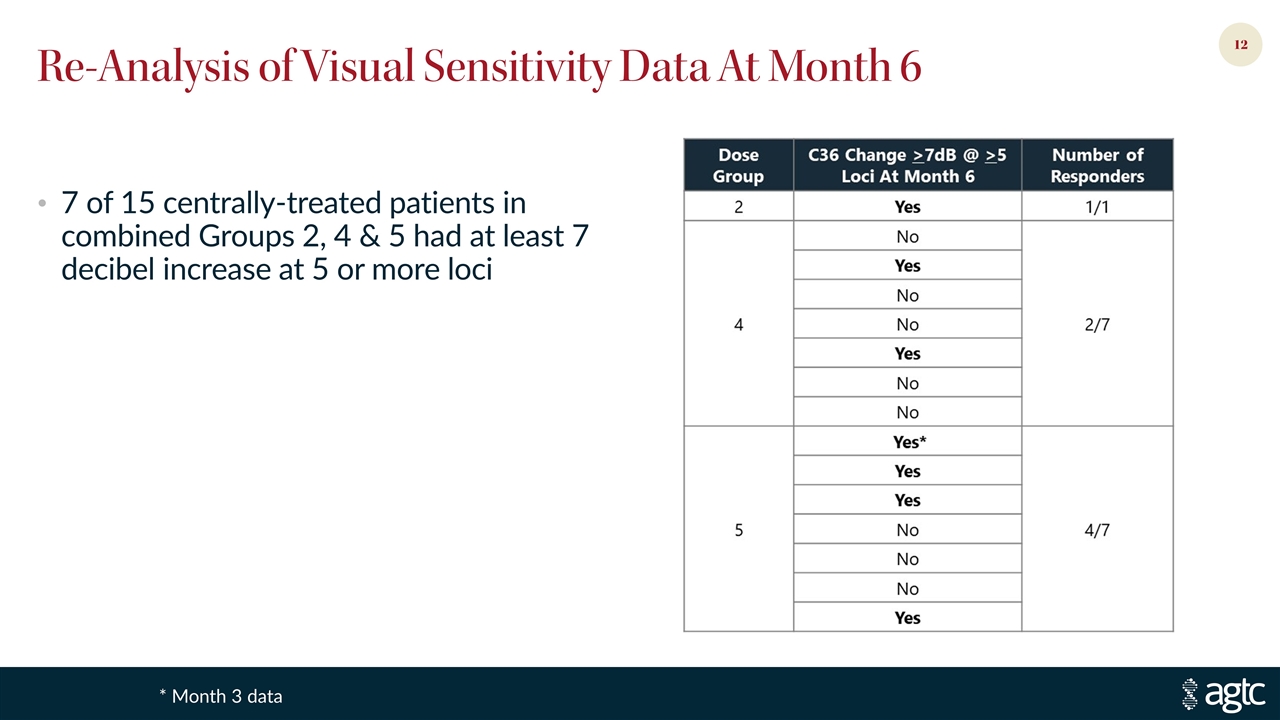
Re-Analysis of Visual Sensitivity Data At Month 6 * Month 3 data 7 of 15 centrally-treated patients in combined Groups 2, 4 & 5 had at least 7 decibel increase at 5 or more loci
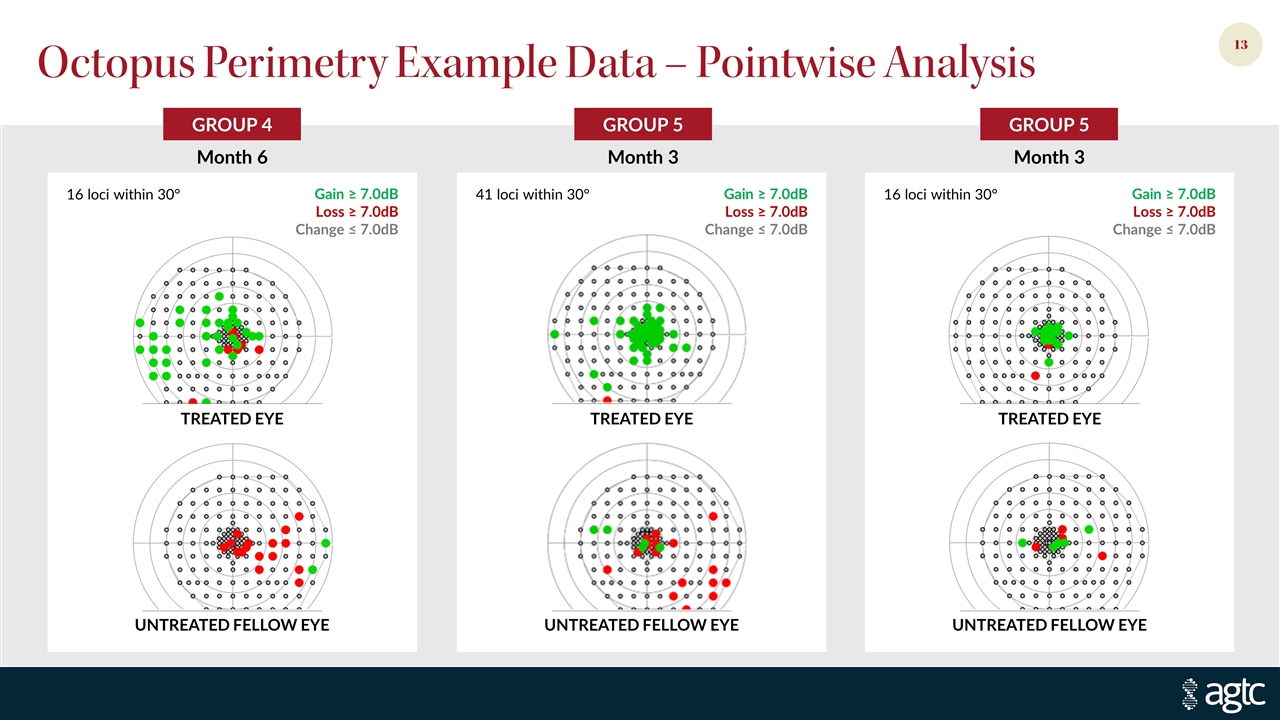
GROUP 5 GROUP 5 GROUP 4 Octopus Perimetry Example Data – Pointwise Analysis Month 6 TREATED EYE UNTREATED FELLOW EYE Loss ≥ 7.0dB Gain ≥ 7.0dB Change ≤ 7.0dB 16 loci within 30° 41 loci within 30° 16 loci within 30° UNTREATED FELLOW EYE TREATED EYE UNTREATED FELLOW EYE TREATED EYE Loss ≥ 7.0dB Gain ≥ 7.0dB Change ≤ 7.0dB Loss ≥ 7.0dB Gain ≥ 7.0dB Change ≤ 7.0dB Month 3 Month 3
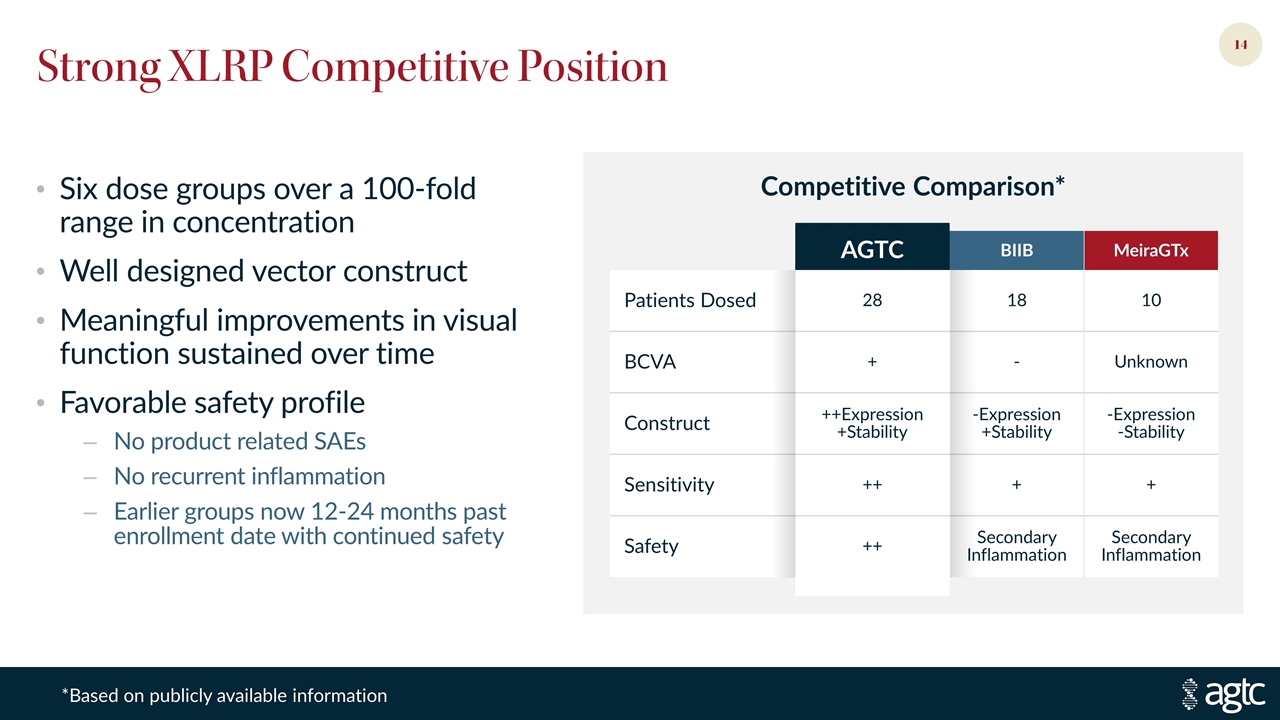
Strong XLRP Competitive Position Six dose groups over a 100-fold range in concentration Well designed vector construct Meaningful improvements in visual function sustained over time Favorable safety profile No product related SAEs No recurrent inflammation Earlier groups now 12-24 months past enrollment date with continued safety AGTC BIIB MeiraGTx Patients Dosed 28 18 10 BCVA + - Unknown Construct ++Expression +Stability -Expression +Stability -Expression -Stability Sensitivity ++ + + Safety ++ Secondary Inflammation Secondary Inflammation Competitive Comparison* *Based on publicly available information

Proposed Phase 2/3 Trial Design = Sub-retinal Treatment with AGTC-501 Group 2 (N~20) Group 5 (N~20) Untreated (N~20) M3b M6b M1b M9b M12b All eligible untreated control eyes will receive treatment after their M12 visit Screening & Randomization Interim Analysis at M6 * * * * 2 3 1 M12 M3 M6 M1 M9 M18 Y2 Y3 Y4 Y5 6M I/A 12M Endpoint Revised protocol includes two masked active arms, untreated control
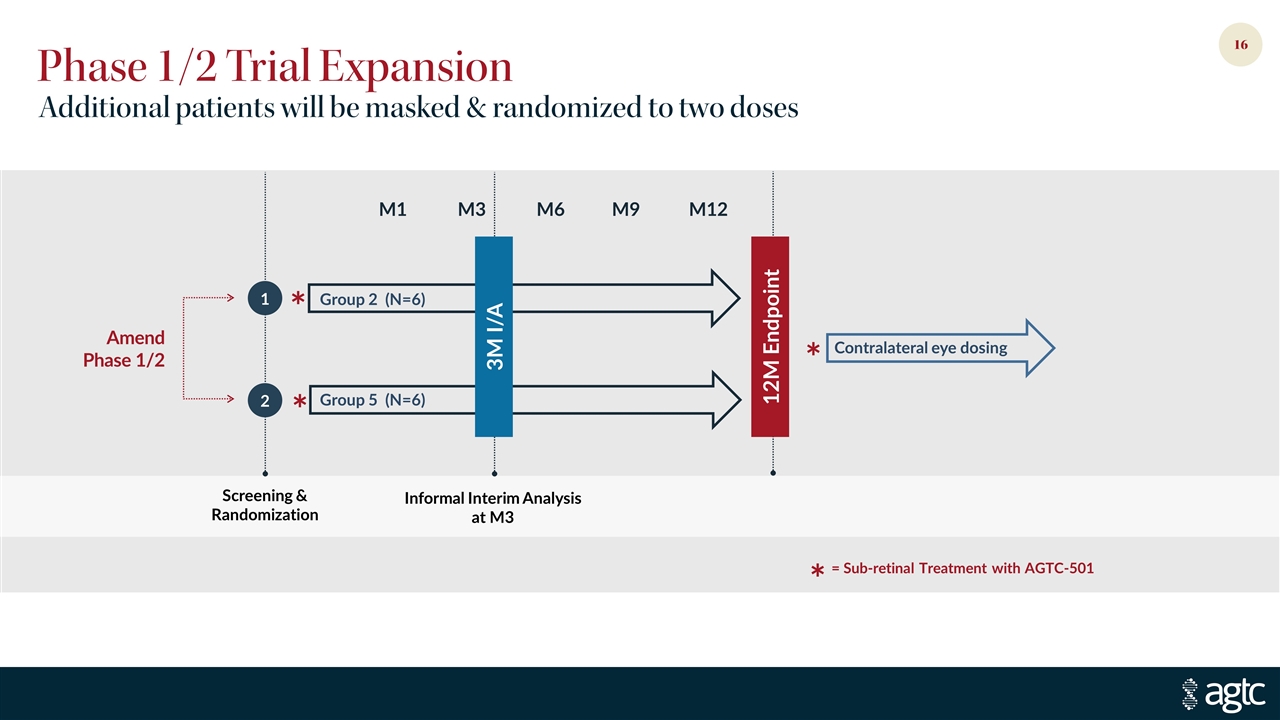
Phase 1/2 Trial Expansion Group 2 (N=6) Group 5 (N=6) Contralateral eye dosing Amend Phase 1/2 = Sub-retinal Treatment with AGTC-501 * * * * 3M I/A Informal Interim Analysis at M3 M12 M3 M6 M1 M9 Screening & Randomization 2 1 12M Endpoint Additional patients will be masked & randomized to two doses

XLRP Moving Forward to Phase 2/3 Trial in 1Q 2021 On track to finalize Phase 2/3 protocol for initiation in 1Q 2021 Clinical trial material in-process Overlapping CDMOs and GLP testing labs—on track to support trial initiation in 1Q 2021 Next data report 4Q 2020 Twelve-month analysis for Groups 1-4—evaluate durability of effect and continued safety Interim analysis for Groups 5 and 6—evaluate safety and efficacy at higher doses 2 1 3

ACHM Clinical Trials Update Early signs of biologic activity supported by patient-reported outcomes No dose-limiting toxicity observed DSMC-approved dosing of pediatric patients to highest dose Adult enrollment complete 22 adult patients enrolled in ACHM B3 15 adult patients enrolled in ACHM A3 Pediatric enrollment ongoing 4 of 9 patients enrolled in ACHM B3 4 of 9 patients enrolled in ACHM A3 Data on File

DISCOVERY IND ENABLING PHASE 1/2 PHASE 3 Milestone/Product Differentiation Dry AMD Complement Factor H IND Enabling Tox Studies; engineered gene cassette Stargardts ABCA4 IND Enabling Tox Studies Otology GJB2 IND Filing; novel capsid tropism profile; partnered with Otonomy ALD ABCD1 IND Enabling Tox Studies Frontotemporal Dementia, ALS Progranulin, c9orf72 Final product construct testing; Novel trivalent cassette components Exciting Preclinical Pipeline Advancing multiple important pipeline candidates in ophthalmology, otology and central nervous system disorders Manageable short-term capital investment for potential long-term significant value creation
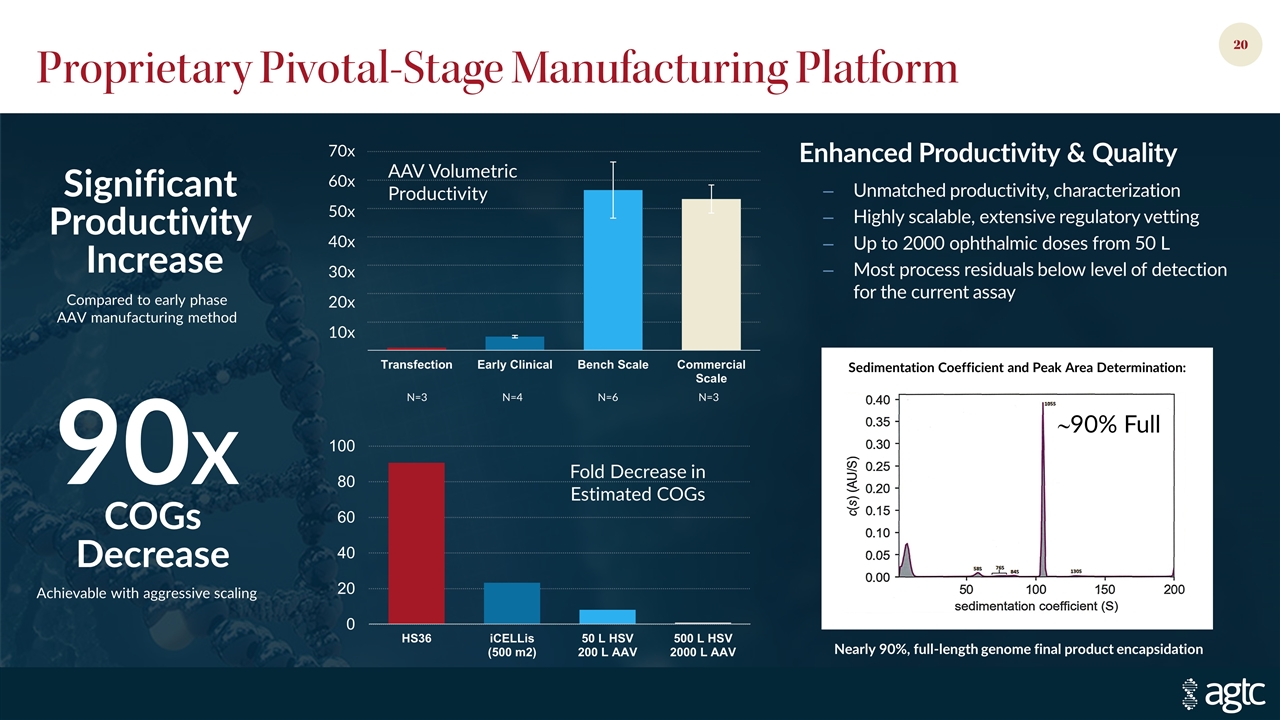
Proprietary Pivotal-Stage Manufacturing Platform Enhanced Productivity & Quality Unmatched productivity, characterization Highly scalable, extensive regulatory vetting Up to 2000 ophthalmic doses from 50 L Most process residuals below level of detection for the current assay Compared to early phase AAV manufacturing method Significant Productivity Increase COGs Decrease 90X Achievable with aggressive scaling Fold Decrease in Estimated COGs Sedimentation Coefficient and Peak Area Determination: Nearly 90%, full-length genome final product encapsidation ~90% Full AAV Volumetric Productivity 70x 60x 50x 40x 30x 20x 10x N=3 N=4 N=6 N=3

Q4 Financial Results

Financial Results 4Q 2020 net loss of $14.5 million compared to net loss of $10.5 million in 4Q 2019 Strong balance sheet $80.5 million in cash, cash equivalents, and investments as of June 30, 2020 Runway into 4Q 2021 The company believes these funds will be sufficient to allow AGTC to: Generate data from ongoing clinical programs Initiate a Phase 2/3 registration trial in XLRP Fund currently planned prioritized preclinical programs

Q&A

Upcoming Virtual Investor Conferences 2020 Wells Fargo Virtual Healthcare Conference Ms. Washer will participate in a fireside chat at 9:20 a.m. ET on Thursday, September 10 H.C. Wainwright 22nd Annual Global Investment Virtual Conference AGTC management will participate in investor 1x1 meetings on Monday, September 14 and Tuesday, September 15 Cantor Fitzgerald Virtual Global Healthcare Conference 2020 Ms. Washer will participate in a fireside chat at 2:40 p.m. ET on Wednesday, September 16

2020 10-K Conference Call September 9, 2020