
AGTC Analyst and Investor Day June 22, 2015 Exhibit 99.1

Presentation Agenda Welcome Sue Washer Introduction Jeffery Chulay, MD Patient Panel Jeffery Chulay, MD Orphan Ophthalmic Opportunities William Hauswirth, PhD XLRS Clinical Overview Mark Pennesi, MD, PhD Achromatopsia Clinical Overview Joe Carroll, PhD XLRP Clinical Overview Steve Rose, PhD Vector Design & Manufacturing David Knop, PhD Q&A and Closing Remarks Sue Washer

Visionary Science for Life Changing Cures 2015 Jeffery Chulay, MD

Forward Looking Statements Today’s presentation includes forward-looking statements intended to qualify for the Safe Harbor from liability established by the Private Securities Litigation Reform Act of 1995. These forward-looking statements, including statements regarding our planned pre-clinical and clinical studies, regulatory approval process and demand for our product candidates, are subject to risks, uncertainties and other factors that could cause actual results to differ materially from those suggested by our forward-looking statements. These factors include, but are not limited to, the following: We have incurred significant losses since inception and anticipate that we will continue to incur significant losses for the foreseeable future. Our ability to generate revenue from product sales is highly uncertain. We may need to raise additional funding in the future, which may not be available on acceptable terms, or at all. No gene therapy products have been approved in the United States, and we may not be able to obtain regulatory approvals for our product candidates. We may encounter substantial delays in our clinical trials or fail to demonstrate safety and efficacy to the satisfaction of applicable regulatory authorities. We expect to rely on third parties to conduct, supervise and monitor our clinical trials and to conduct certain aspects of our product manufacturing and protocol development. The insurance coverage and reimbursement status of our product candidates is uncertain. Negative public opinion and increased regulatory scrutiny of gene therapy and genetic research may adversely affect public perception of our product candidates and prospects for our business. If we are unable to obtain and maintain adequate patent protection for our technology and products our competitors could develop and commercialize technology and products similar or identical to ours.
Our Vision Become the world leader in ophthalmology gene therapy

Our Mission Dramatically improve the lives of people across the globe by using science and innovation to develop gene based therapeutics. Specifically addressing the needs of the thousands of people globally whose sight is at risk from orphan ophthalmic disease

Why Ophthalmology No current treatments for diseases targeted by AGTC product candidates Renewed industry/regulatory interest in pediatric diseases of the eye People greatly fear losing sight Extensive preclinical data Highly predictive animal models Well characterized diseases Well-defined and recognized clinical endpoints simplify trial design Preliminary evidence for safe targeted delivery in human trials Restoring Sight Significant Unmet Medical Need Child with achromatopsia outdoors Same child indoors in dim light

Uniquely Positioned To Lead Multiple programs with potential to provide superior long-term value to patients, key inflection points in 2015 Broad Pipeline Robust understanding of vector selection, design, manufacturing and delivery Extensive Expertise Key IP, >100 patents, protects gene, vector capsid, regulatory elements, manufacturing and/or delivery for each candidate Intellectual Property Demonstrated success in balancing resources, risks and rewards; >100 patients treated in Phase 1/2 trials Track Record of Progress Optimized AAV vectors that deliver safe and sustained expression; manufacturing reproducibly at commercial scale Leading Gene Therapy Platform
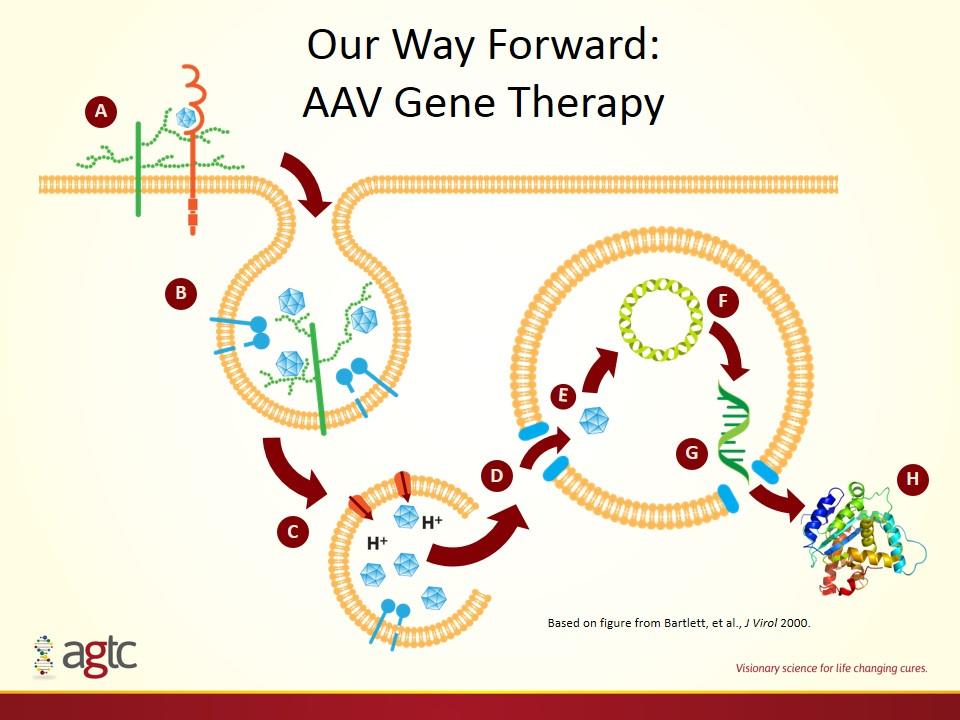
Our Way Forward: AAV Gene Therapy Based on figure from Bartlett, et al., J Virol 2000. A B E C D F G H

Attribute AAV Advantage Safe Not associated with human disease or clinical trial safety signals Simple Easily manipulated to create range of product candidates Stable Resistance to degradation simplifies purification and long term storage Capacity Vector capacity enables delivery of wide array of gene sequences Sustained Expression After single dose protein expression persists for long periods in non-dividing cells Promise of AAV ITR ITR rep cap AAV virus ITR ITR promoter cDNA AAV vector
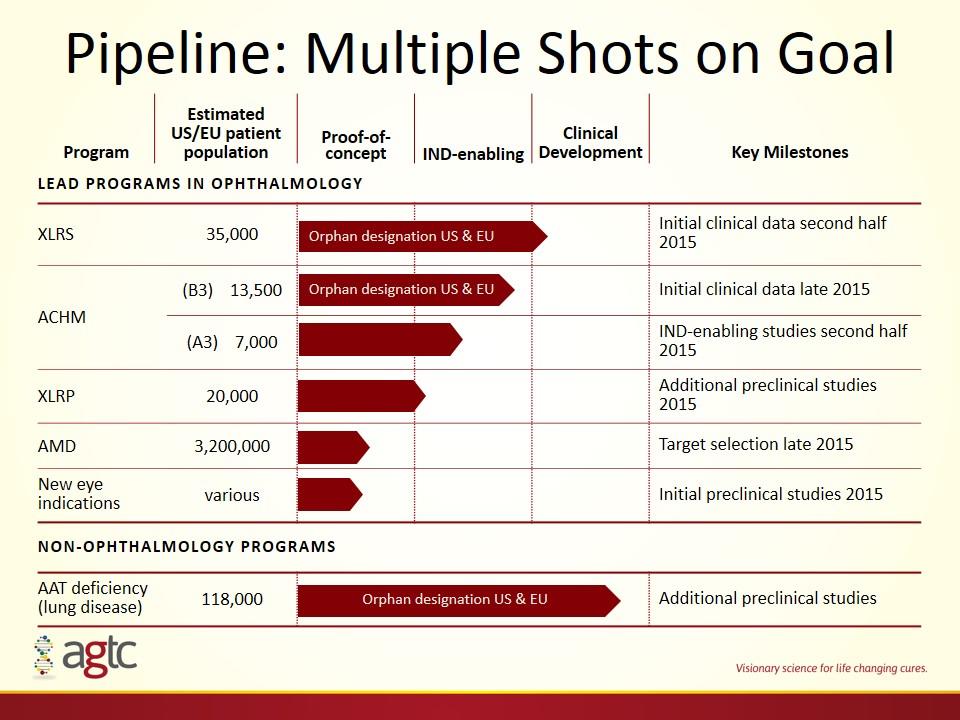
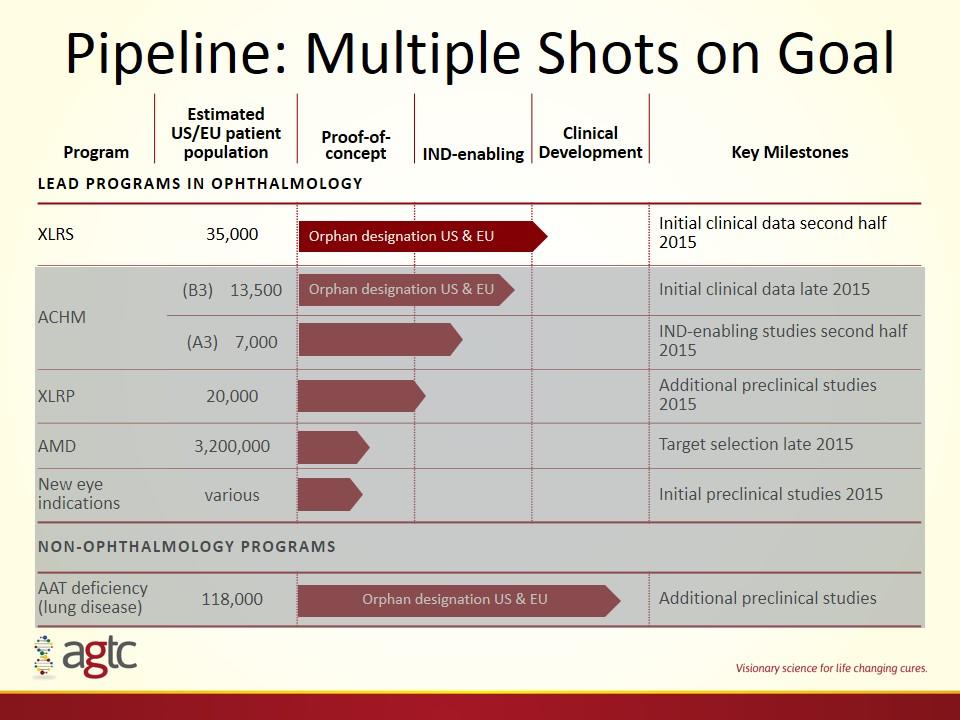
Pipeline: Multiple Shots on Goal Program Estimated US/EU patient population Proof-of-concept IND-enabling Clinical Development Key Milestones Lead programs in ophthalmology XLRS 35,000 Initial clinical data second half 2015 ACHM (B3) 13,500 Initial clinical data late 2015 (A3) 7,000 IND-enabling studies second half 2015 XLRP 20,000 Additional preclinical studies 2015 AMD 3,200,000 Target selection late 2015 New eye indications various Initial preclinical studies 2015 NON-ophthalmology PROGRAMS AAT deficiency (lung disease) 118,000 Additional preclinical studies Orphan designation US & EU Orphan designation US & EU Orphan designation US & EU
Patient Panel

Patient Panel William Warren Technology and business consultant XLRS Patient Diagnosed at age 15 Bridget Vissari Co-founder and President of Achroma Corp. Son diagnosed with Achromatopsia Mark Curley VP of Operations for family business XLRP Patient Diagnosed at age 13

Impact of XLRS, ACHM & RP on Patient Quality of Life 1Retinoschisis Parenting Forum. American Foundation® for the Blind Web site. http://www.afb.org/forum/parenting/retinoschisis/12. Accessed June 13, 2015. 2Faces of Achromatopsia. Achroma Corp Web site. http://www.achromacorp.org/Faces.html. Accessed June 17, 2015. 3Fighting Blindness Patient Stories. Fighting Blindness Web site. http://www.fightingblindness.ie/support/personal-stories/. Accessed June 13, 2015 Real patients or parents of children with XLRS1: I’m currently considering whether or not to go into Civil Engineering as not being able to drive would mean not being able to go to site. This would be incredibly frustrating after having studied at university for 4 years. I went to regular school just like everyone else. I was offered books on tape and large print books but never used them out of self pride I guess. To this day I am still self-conscious. School and Career Real patient with achromatopsia2: I experienced many of the same struggles as every person with achromatopsia – teasing as a child and ignorance from others as an adult, self-consciousness, education and job difficulties, and most of all, a lack of independence stemming from not being able to get a driver's license, which has had a pervasive effect on my life. Social Real patients with RP3: I kept crashing the car. I’d clip a wall on a narrow road, and it only seemed to be happening to me, not my friends. I knew something was wrong. It wasn’t long after the last crash that I was diagnosed with RP. I couldn’t drive anymore. When I’m walking by myself I’m constantly scanning up, down, left and right. If I’m going somewhere new and there are a lot of people around, I’ll scan to see where I can go or sit before I move. Safety

William Hauswirth, PhD Ophthalmology, University of Florida Orphan Ophthalmic Opportunities: An Introduction

Functional Range of the Visual System Over 10 logarithmic units (in lux): Sun Daylight Roomlight Twilight Full Moon Starlight Photopic Vision Mesopic Vision Scotopic Vision Pure Cone Vision Pure Rod Vision >100,000 10,000 500 - 1000 100 - 30 0.3 0.0001
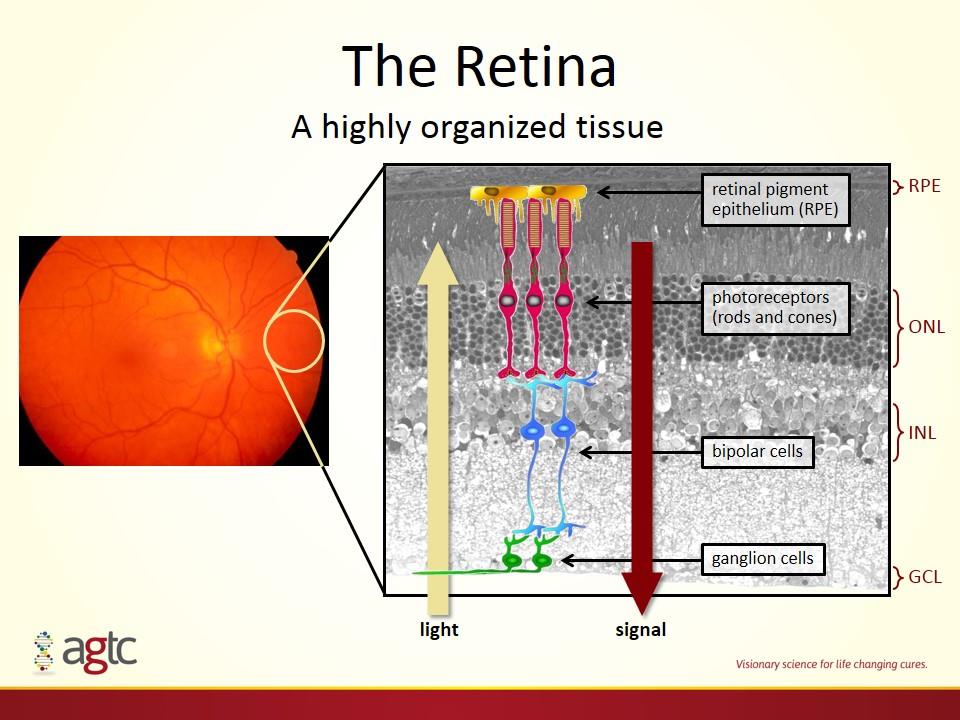
The Retina light bipolar cells photoreceptors (rods and cones) retinal pigment epithelium (RPE) ganglion cells signal ONL INL GCL RPE A highly organized tissue
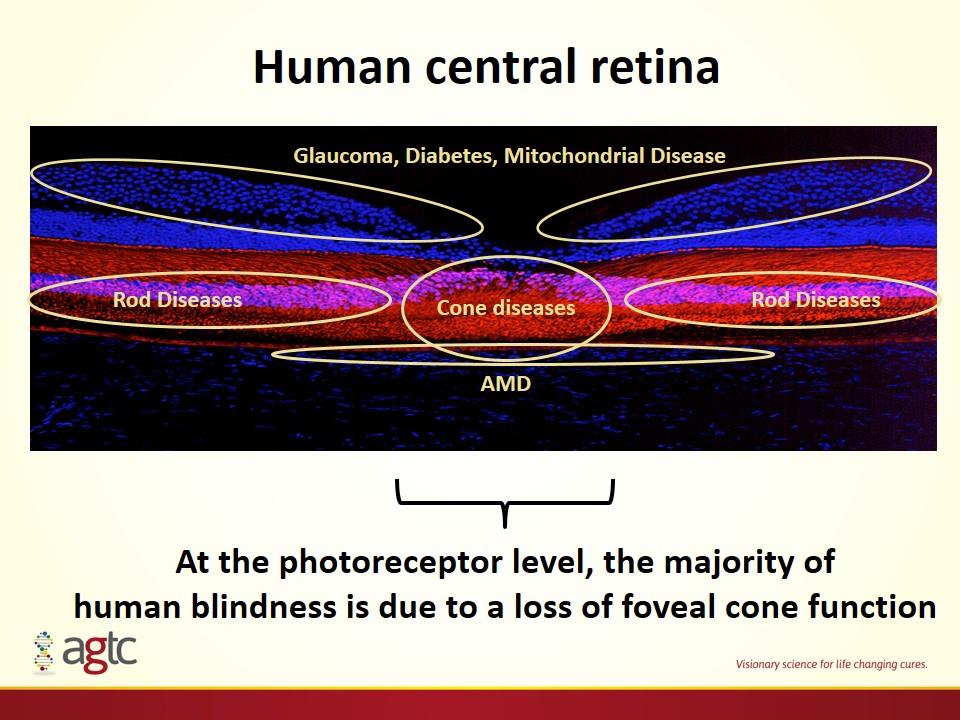
Human central retina At the photoreceptor level, the majority of human blindness is due to a loss of foveal cone function Cone diseases Rod Diseases Rod Diseases AMD Glaucoma, Diabetes, Mitochondrial Disease
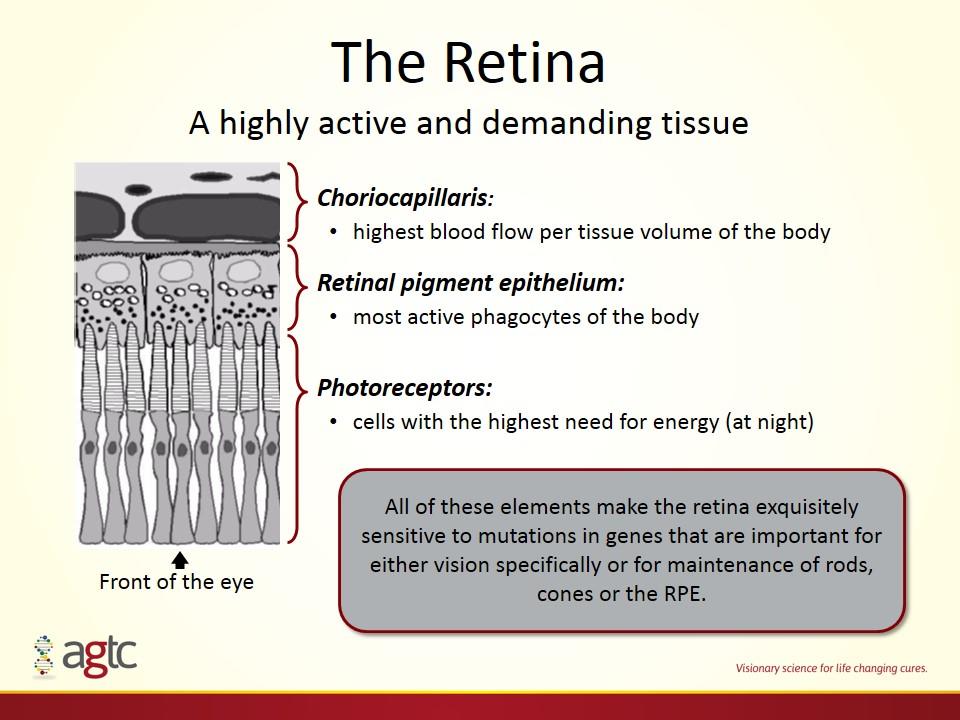
The Retina A highly active and demanding tissue All of these elements make the retina exquisitely sensitive to mutations in genes that are important for either vision specifically or for maintenance of rods, cones or the RPE. Choriocapillaris: highest blood flow per tissue volume of the body Retinal pigment epithelium: most active phagocytes of the body Photoreceptors: cells with the highest need for energy (at night) Front of the eye

AAV Vectors in the Retina Nonpathogenic Vector serotype and injection site control what sets of cells are the preferred target(s) Promoters control which retinal cells express the gene Up to 95% transduction of the targeted cell type Lifetime persistence in rodents, 10+yrs in dogs, 7+yrs and counting in humans.
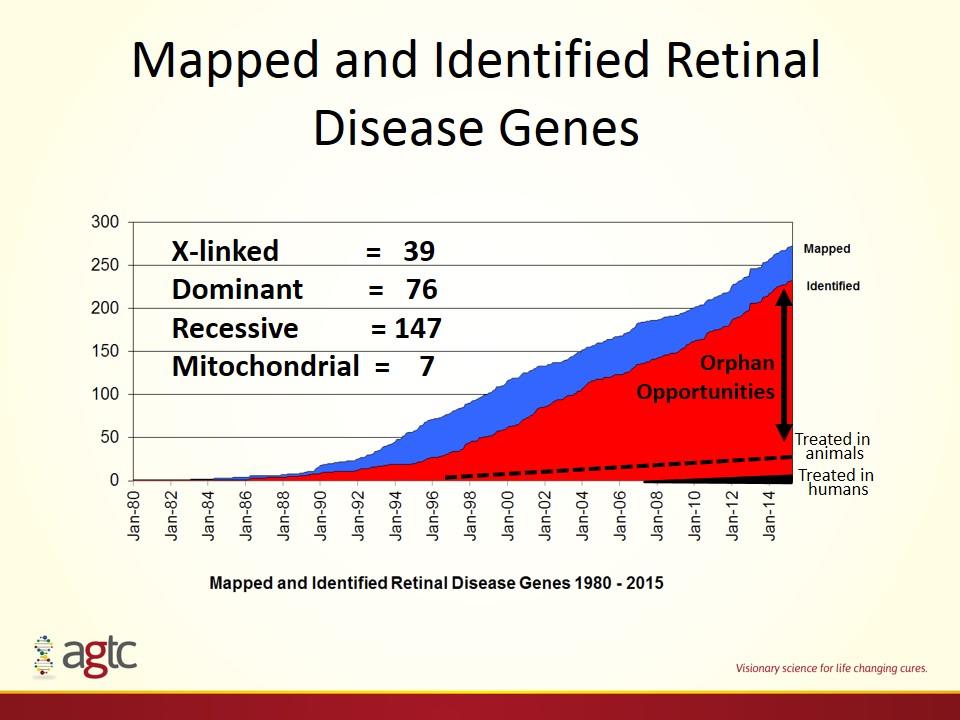
Treated in animals Treated in humans Orphan Opportunities Mapped and Identified Retinal Disease Genes X-linked = 39 Dominant = 76 Recessive = 147 Mitochondrial = 7
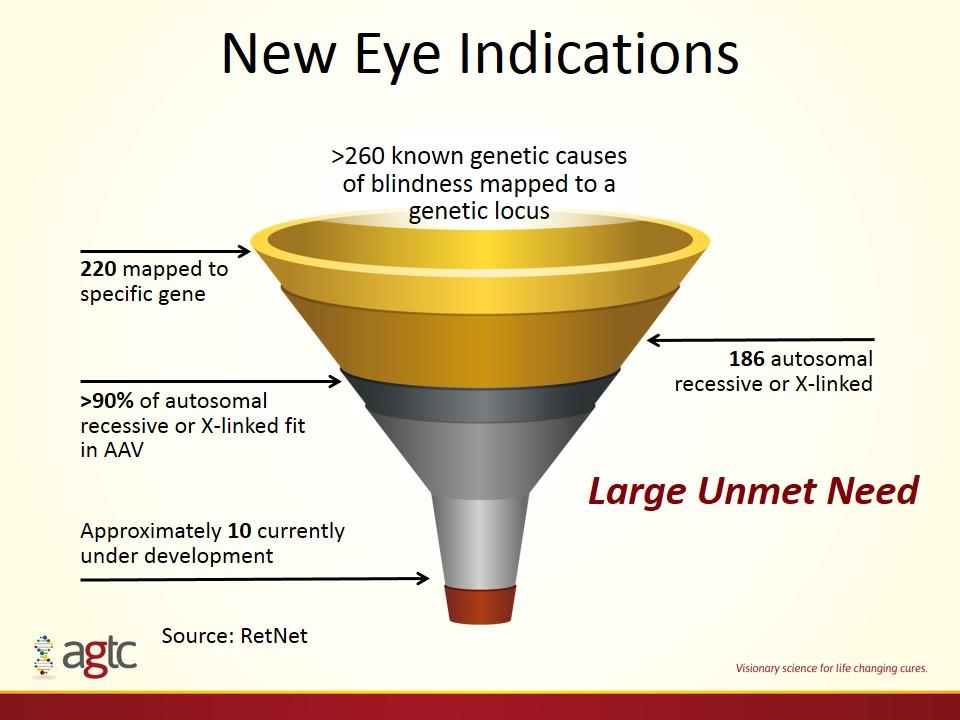
220 mapped to specific gene New Eye Indications 186 autosomal recessive or X-linked >90% of autosomal recessive or X-linked fit in AAV Approximately 10 currently under development >260 known genetic causes of blindness mapped to a genetic locus Source: RetNet Large Unmet Need

AAV Mediated Retinal Gene Therapy Projects -- Hauswirth Lab + Collaborators hum = current clinical trial Retinal Disease Species Publication Year(s) Reagent Type Species Publication Year(s) 24 7 64 18 5 33 none ms 97, 09, 11, 12, 13(2), 14(4) Neurotroph. RP (Rho) rt, ms, dg 98, 12 CNTF rt, ms, dg 03, 07, 13 RPE65 LCA dg, ms, hum 01, 05, 06, 07(2), 08(2), BDNF ms 03, 07 09, 10, 11, 13(2), 15(2) GDNF rt 01 GC1 LCA ms, 10, 13 FGF2 rt 03 MERTK rt, hum 08, 12, 13 Anti-apoptotic XLRS ms, hum 05, 08 XIAP rt, ms 03, 06, 07, 09, 11, 12 XLRP dog, ms, 12, 14, 15 Anti-oxidant Achromatopsia ms, dg, sh. 07, 10, 12, 14, 15 SOD2 ms 04, 07(2) RG Color blindness mke, gbl 08, 09 Catalase ms 98, 07 LHON (ND4) ms, hum 07, 09, 12, 13, 14(2) Chaperone LRAT LCA ms 04, 05, 11 BIP ms 10 RP (PDE6b) ms 08, 13 Stress response BBS4 ms 11 ERK1/2 ms 12 RP(GNAT2) ms 09 TrkB ms 02 Ush3 ms On going STAT3 ms 14 BBS (whirlin) ms 11 HSP70 ms 14(2) RP(LPCAT1) ms 14 End2 ms 13 Best's dg 13 Optogenetics MFRP ms 12 ChR2 ms 11 RP (RD3) ms 13 Anti-NV agent Usher 1B ms 13, 14 PEDF ms 02(2) RP (GCAP) ms 13 sFLT ms, mke, hum 09, 11, 13 BCM rt, ms On going siRNA Stgdt Dis ms On going p22phox ms 08 SPATA7 ms 15 GCAP1 ms 13 Retinal Gene Therapy Projects
Q&A
Pipeline: Multiple Shots on Goal Program Estimated US/EU patient population Proof-of-concept IND-enabling Clinical Development Key Milestones Lead programs in ophthalmology XLRS 35,000 Initial clinical data second half 2015 ACHM (B3) 13,500 Initial clinical data late 2015 (A3) 7,000 IND-enabling studies second half 2015 XLRP 20,000 Additional preclinical studies 2015 AMD 3,200,000 Target selection late 2015 New eye indications various Initial preclinical studies 2015 NON-ophthalmology PROGRAMS AAT deficiency (lung disease) 118,000 Additional preclinical studies Orphan designation US & EU Orphan designation US & EU Orphan designation US & EU

XLRS Clinical Overview Genetic Background Mark Pennesi, MD, PhD

XLRS Genetics and Mechanisms Rare, X-Linked, Recessive Incidence: 1:5,000 to 1:30,000 Highest in Scandinavia Prevalence Overall ~35,000 patients in US and EU Mutation in single gene (RS1) in all cases 24 kDa protein secreted by photoreceptor and bipolar cells Forms homo-octameric complexes that help maintain retinal cellular organization Adapted from Molday et al. JBC 2007;282:32792-32801.
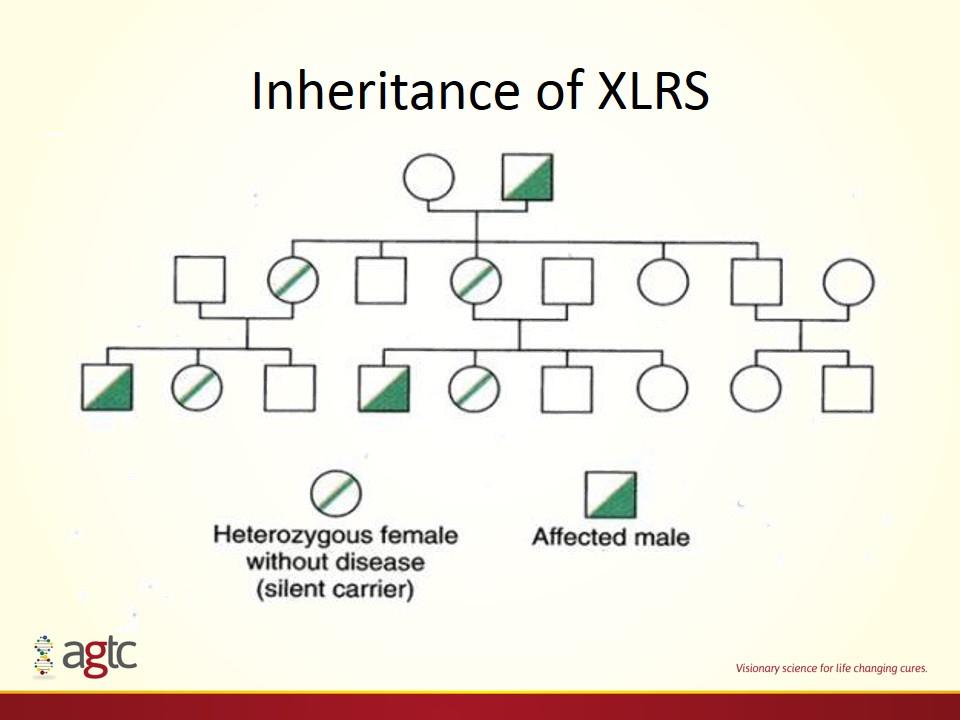
Inheritance of XLRS
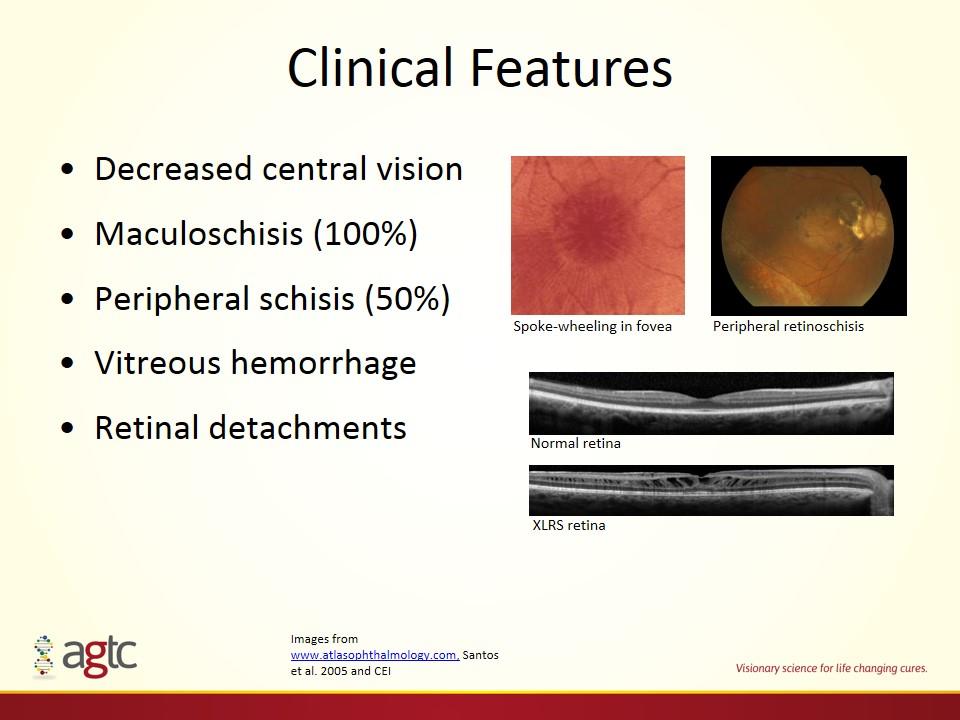
Clinical Features Decreased central vision Maculoschisis (100%) Peripheral schisis (50%) Vitreous hemorrhage Retinal detachments Spoke-wheeling in fovea Peripheral retinoschisis XLRS retina Normal retina Images from www.atlasophthalmology.com, Santos et al. 2005 and CEI
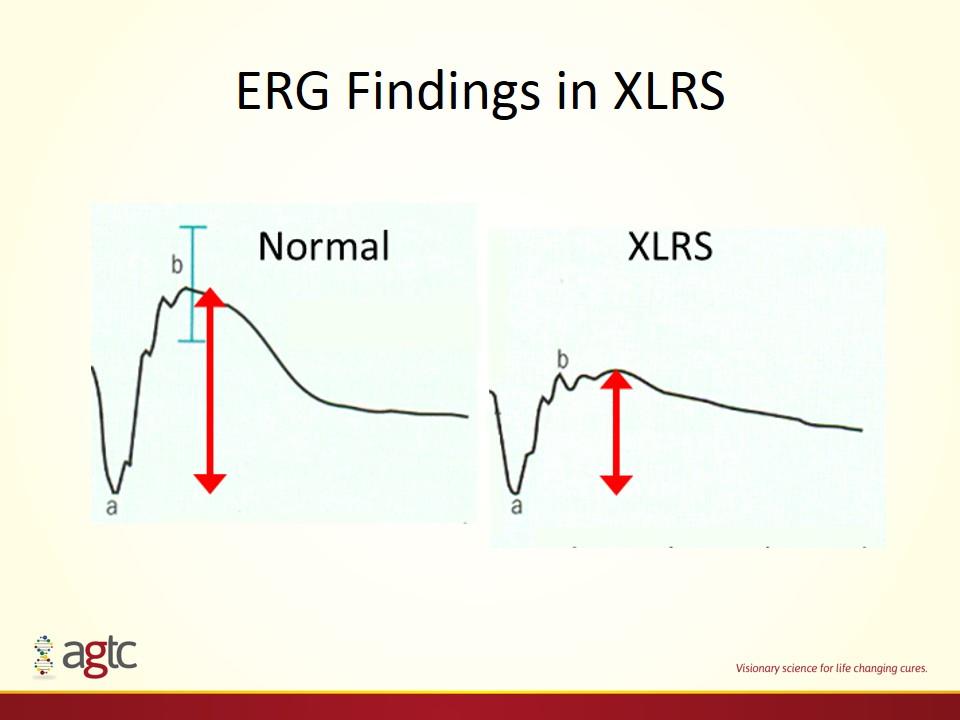
ERG Findings in XLRS

Current Therapy for XLRS There is currently no approved treatment for XLRS Focus is management Low vision aids (high-powered magnifiers) for reading Preferential seating in front of the class for school children Anecdotal reports suggest topical carbonic anhydrase inhibitors may reduce schisis and improve VA Effect observed in some but not all patients No reported controlled clinical trials
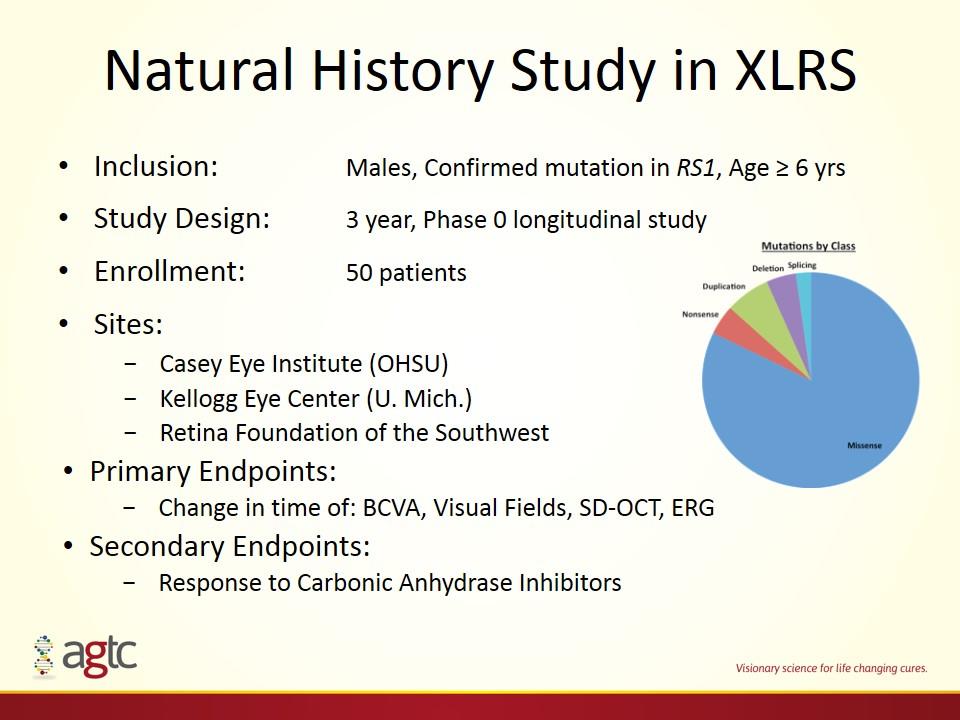
Natural History Study in XLRS Inclusion: Males, Confirmed mutation in RS1, Age ≥ 6 yrs Study Design: 3 year, Phase 0 longitudinal study Enrollment: 50 patients Sites: Casey Eye Institute (OHSU) Kellogg Eye Center (U. Mich.) Retina Foundation of the Southwest Primary Endpoints: Change in time of: BCVA, Visual Fields, SD-OCT, ERG Secondary Endpoints: Response to Carbonic Anhydrase Inhibitors
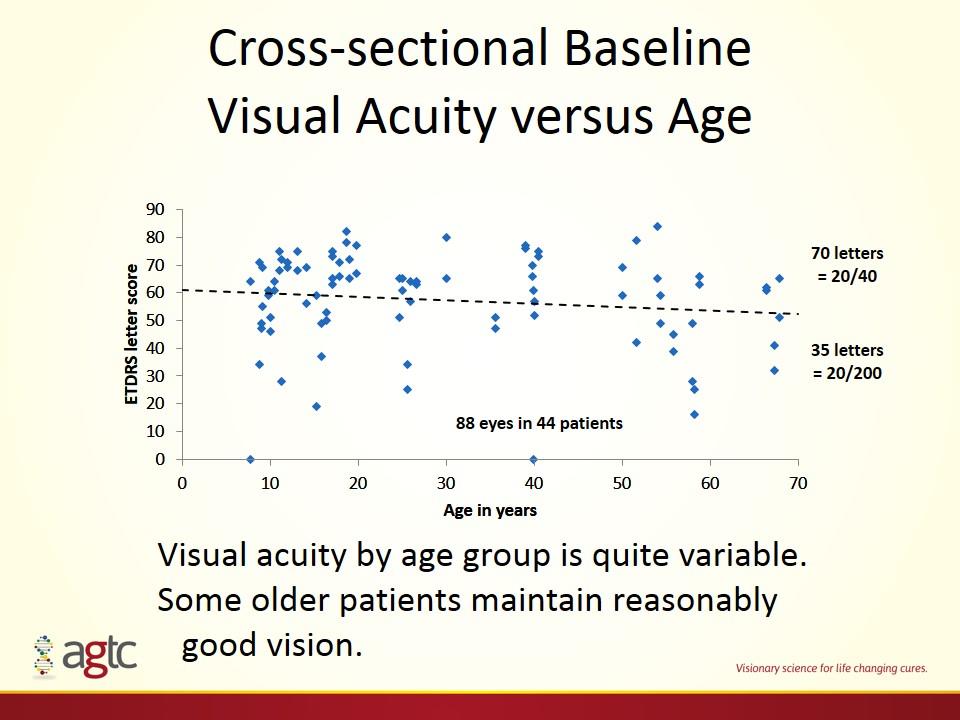
Cross-sectional Baseline Visual Acuity versus Age Visual acuity by age group is quite variable. Some older patients maintain reasonably good vision. 88 eyes in 44 patients 70 letters = 20/40 35 letters = 20/200
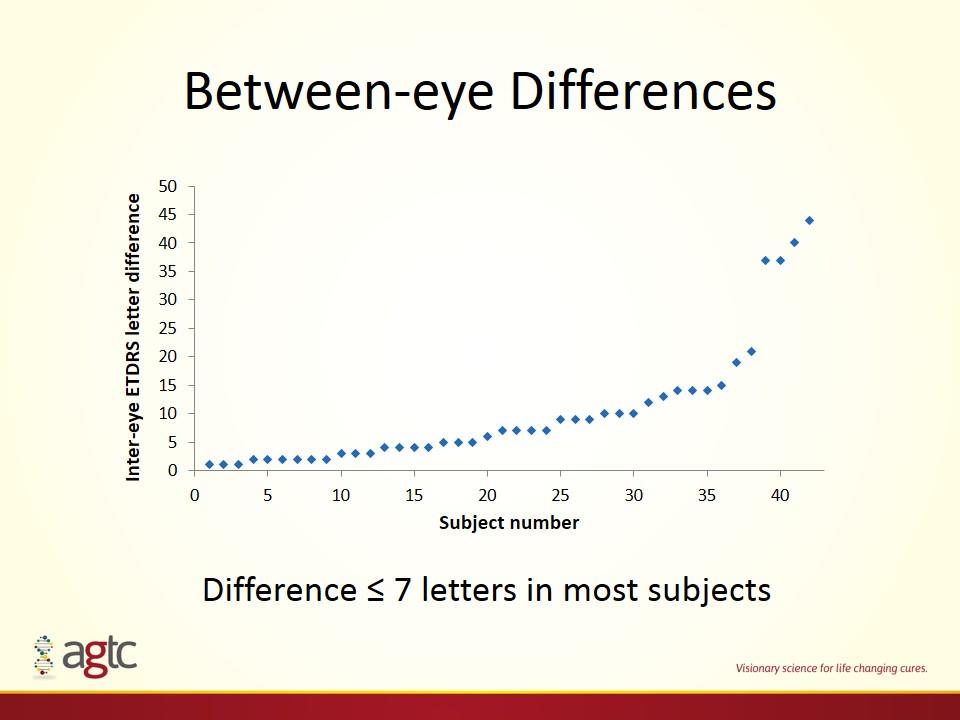
Between-eye Differences Difference ≤ 7 letters in most subjects
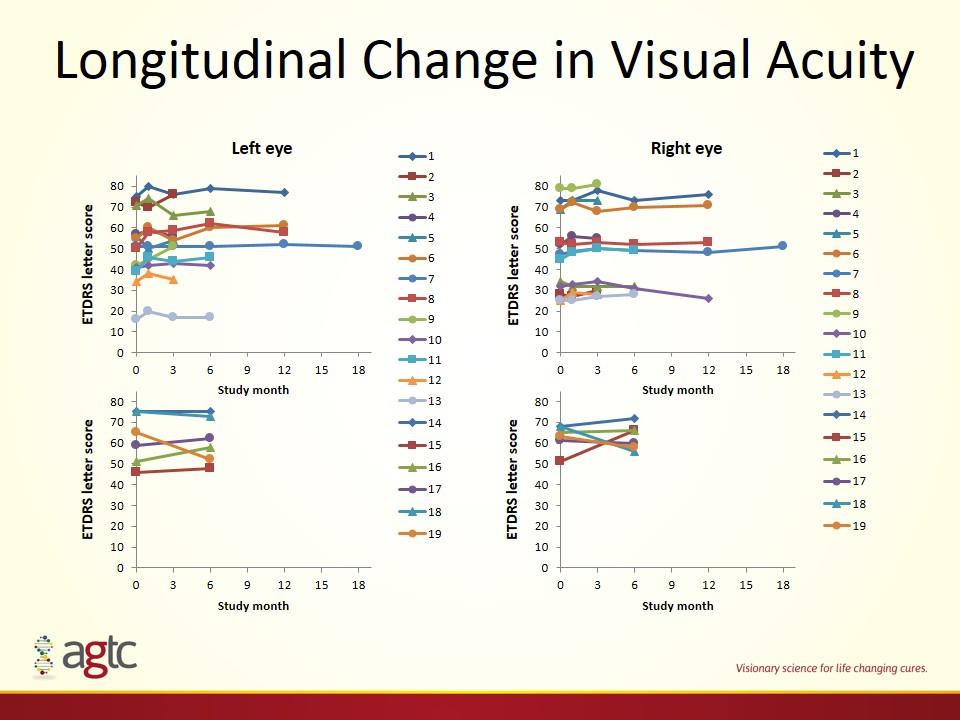
Longitudinal Change in Visual Acuity
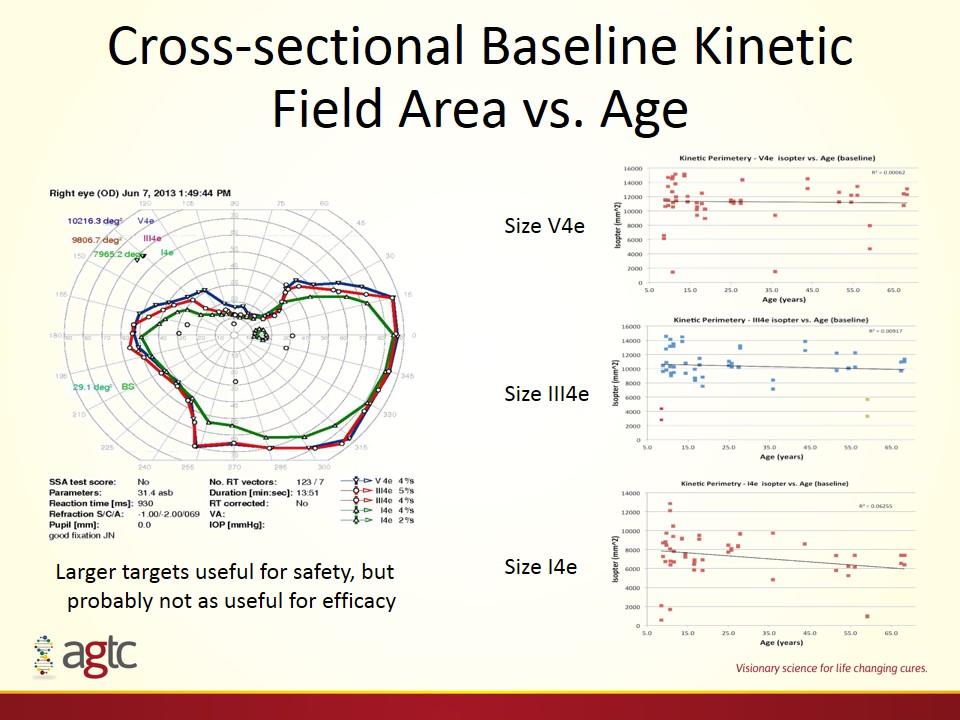
Cross-sectional Baseline Kinetic Field Area vs. Age Size V4e Size III4e Size I4e Larger targets useful for safety, but probably not as useful for efficacy
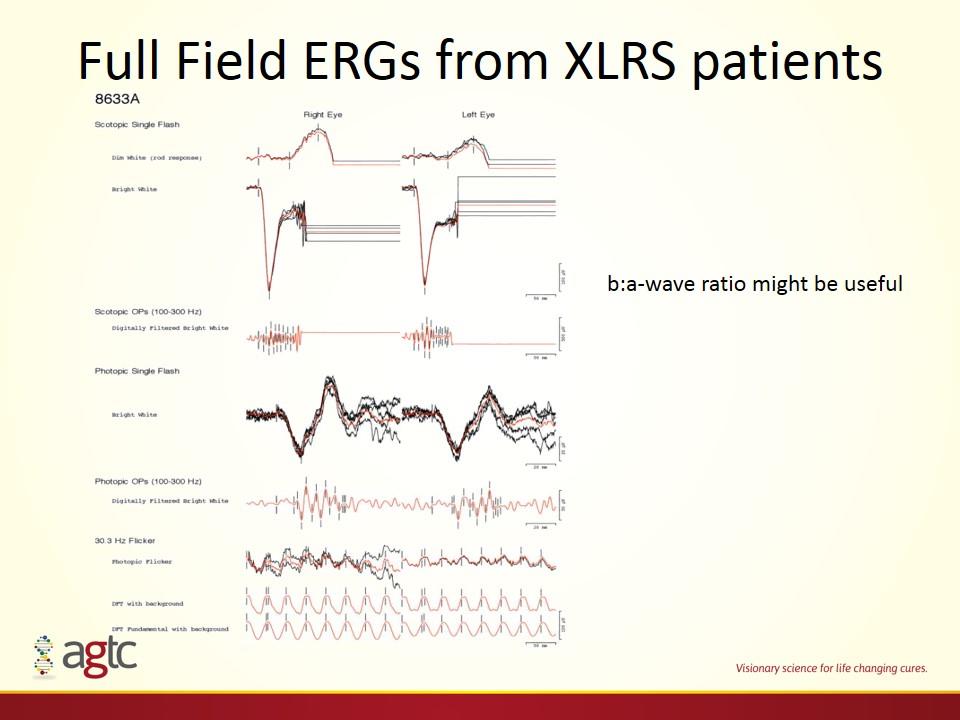
Full Field ERGs from XLRS patients b:a-wave ratio might be useful
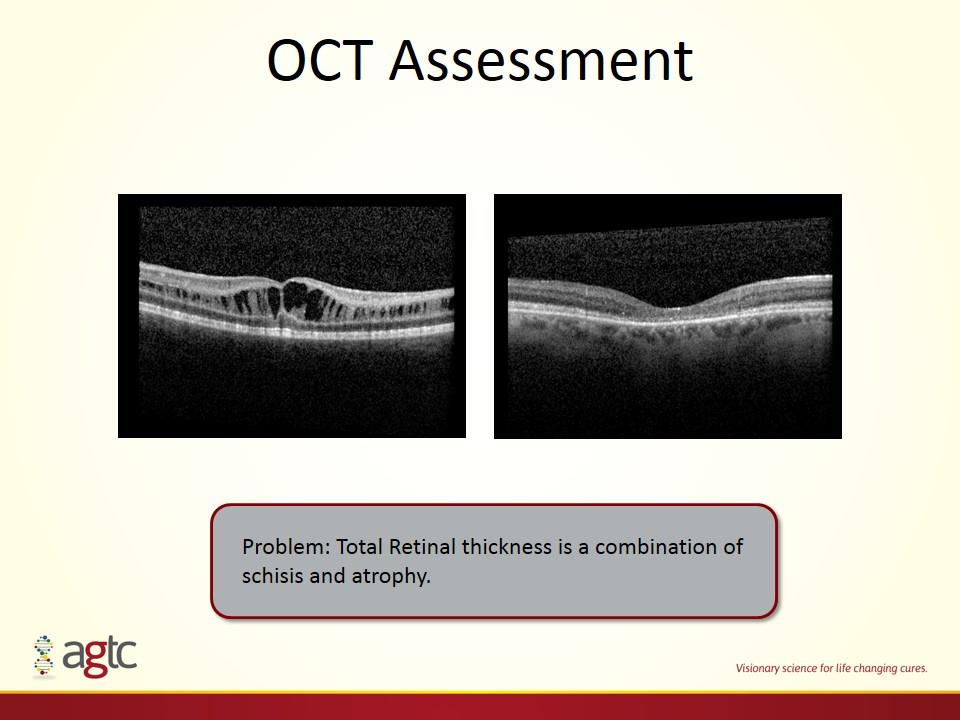
OCT Assessment Problem: Total Retinal thickness is a combination of schisis and atrophy.
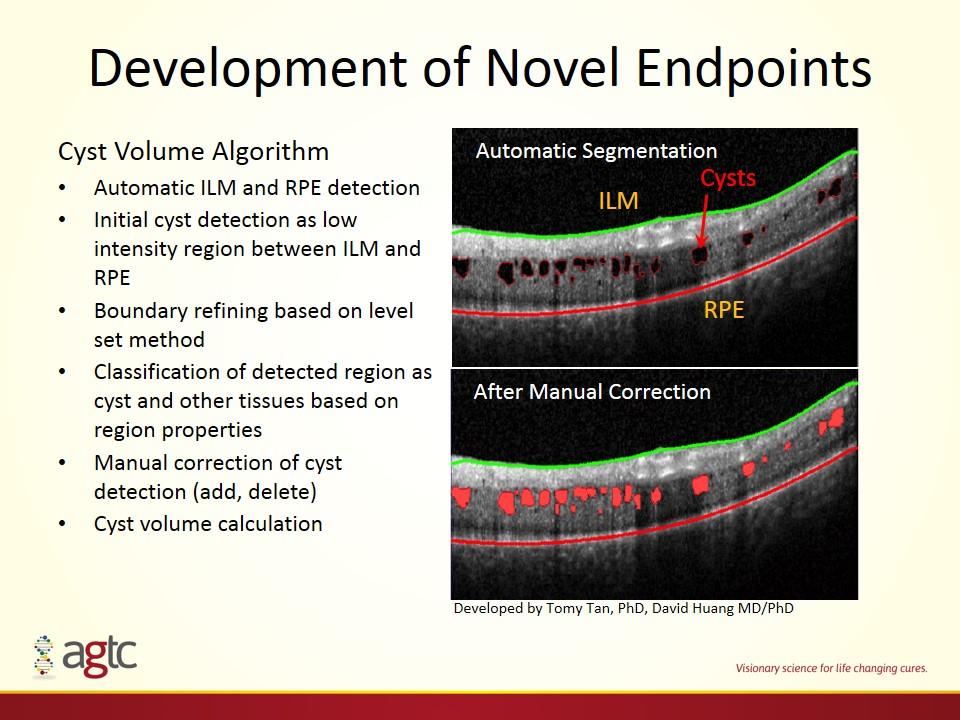
Development of Novel Endpoints Cyst Volume Algorithm Automatic ILM and RPE detection Initial cyst detection as low intensity region between ILM and RPE Boundary refining based on level set method Classification of detected region as cyst and other tissues based on region properties Manual correction of cyst detection (add, delete) Cyst volume calculation Automatic Segmentation ILM RPE Cysts After Manual Correction Developed by Tomy Tan, PhD, David Huang MD/PhD
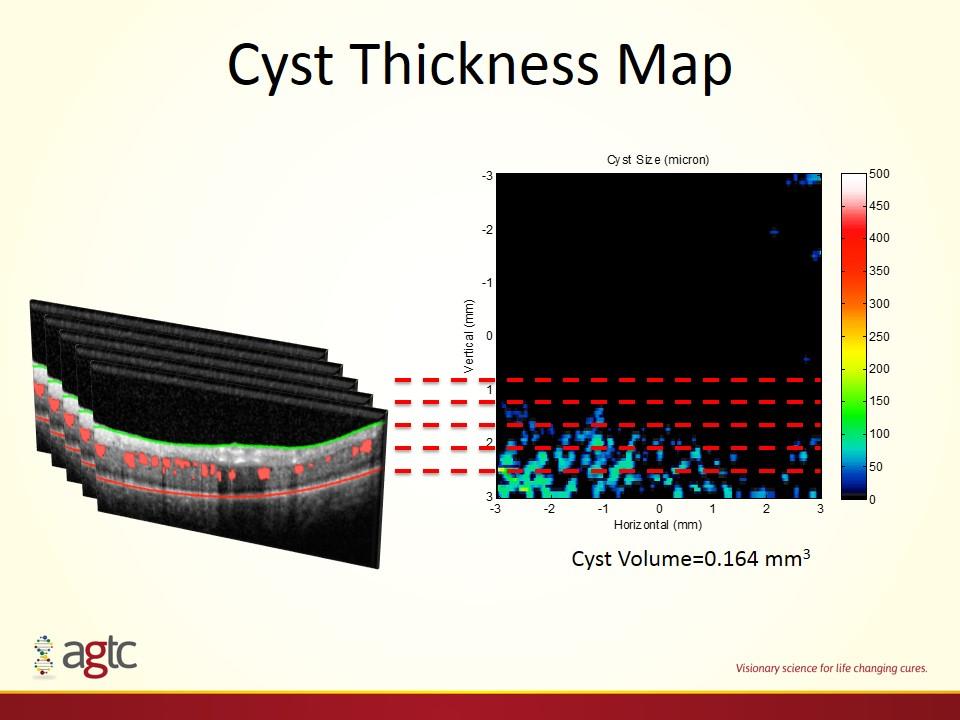
Cyst Thickness Map Cyst Volume=0.164 mm3
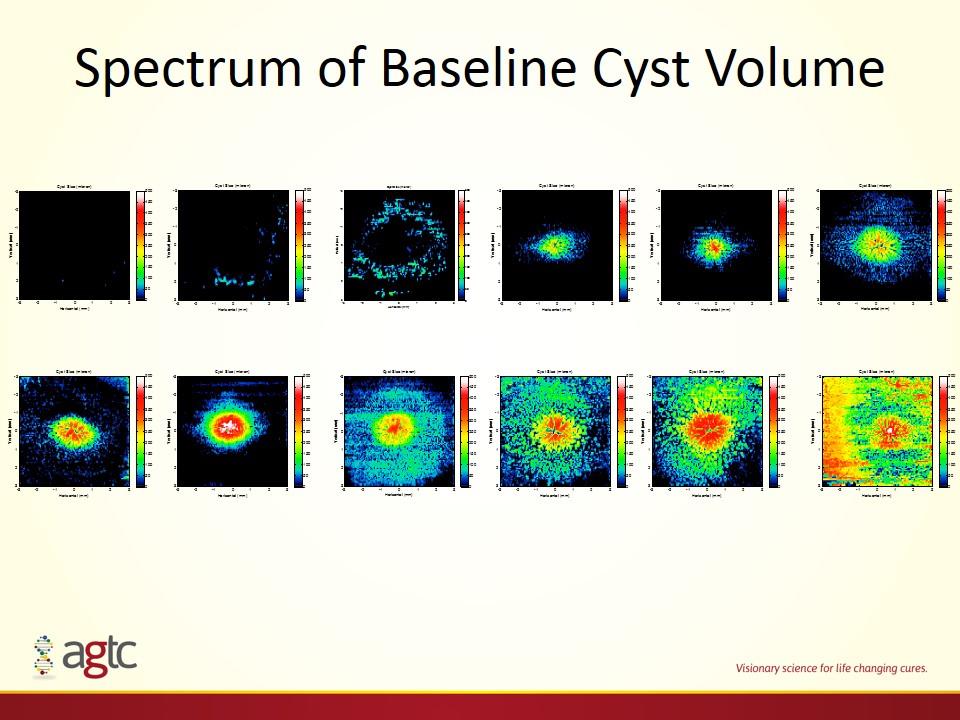
Spectrum of Baseline Cyst Volume
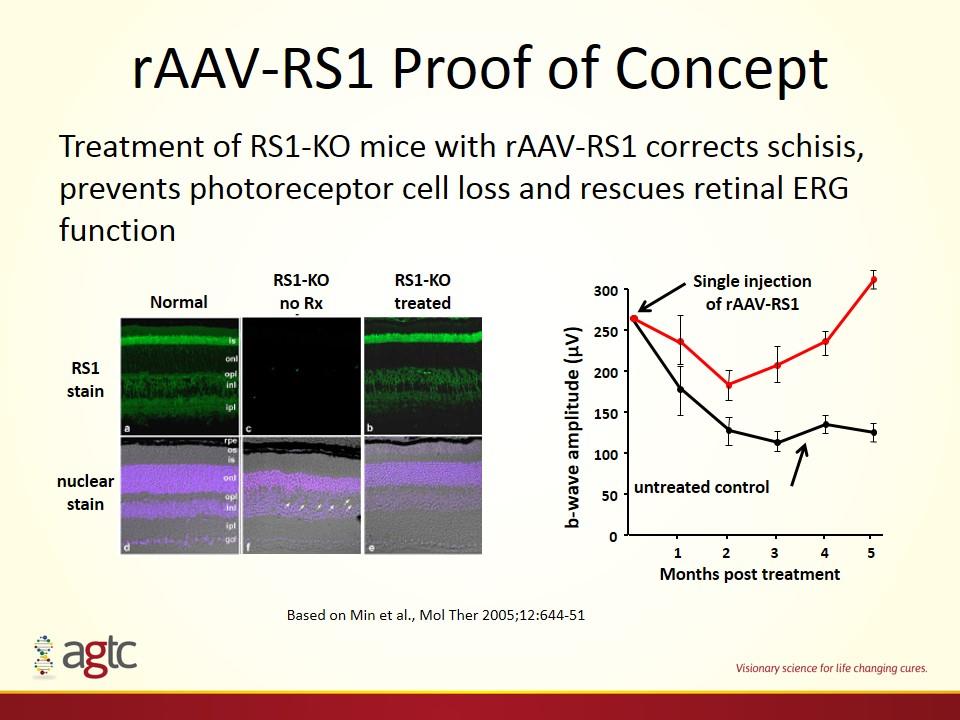
rAAV-RS1 Proof of Concept Treatment of RS1-KO mice with rAAV-RS1 corrects schisis, prevents photoreceptor cell loss and rescues retinal ERG function RS1 stain nuclear stain Normal RS1-KO no Rx RS1-KO treated Months post treatment 0 50 100 150 200 250 300 b-wave amplitude (µV) 1 2 3 4 5 Single injection of rAAV-RS1 untreated control Based on Min et al., Mol Ther 2005;12:644-51

Proof-of-Concept Studies AAV serotype cDNA Promoter Route of administration Dose level µL, vg/mL Laboratory 2 Mouse RS1 CMV Subretinal & Intravitreal 1.5-2 2.3 × 1013 Sieving 5 Human RS1 mOp500 Subretinal 1 4 × 1013 Hauswirth 8 Human RS1 hRS1 Intravitreal 1.5 5 × 1013 Sieving 5 Human RS1 CBA Subretinal 1 6.5 × 1012 Hauswirth 7m8 Human RS1 CBA or rho Intravitreal 1 5 × 1013 Flannery Efficacy demonstrated by ERG b-wave improvement in all studies. Schisis and visual function by optokinetic response tested in some studies and showed improvement.
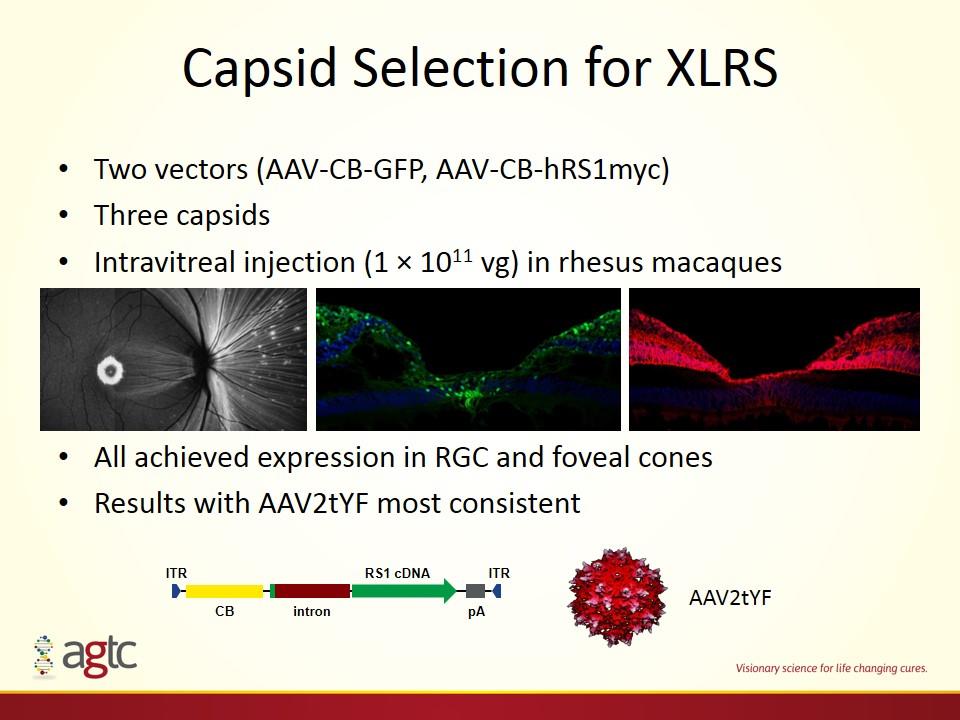
Capsid Selection for XLRS Two vectors (AAV-CB-GFP, AAV-CB-hRS1myc) Three capsids Intravitreal injection (1 × 1011 vg) in rhesus macaques All achieved expression in RGC and foveal cones Results with AAV2tYF most consistent ITR ITR RS1 cDNA pA intron CB AAV2tYF
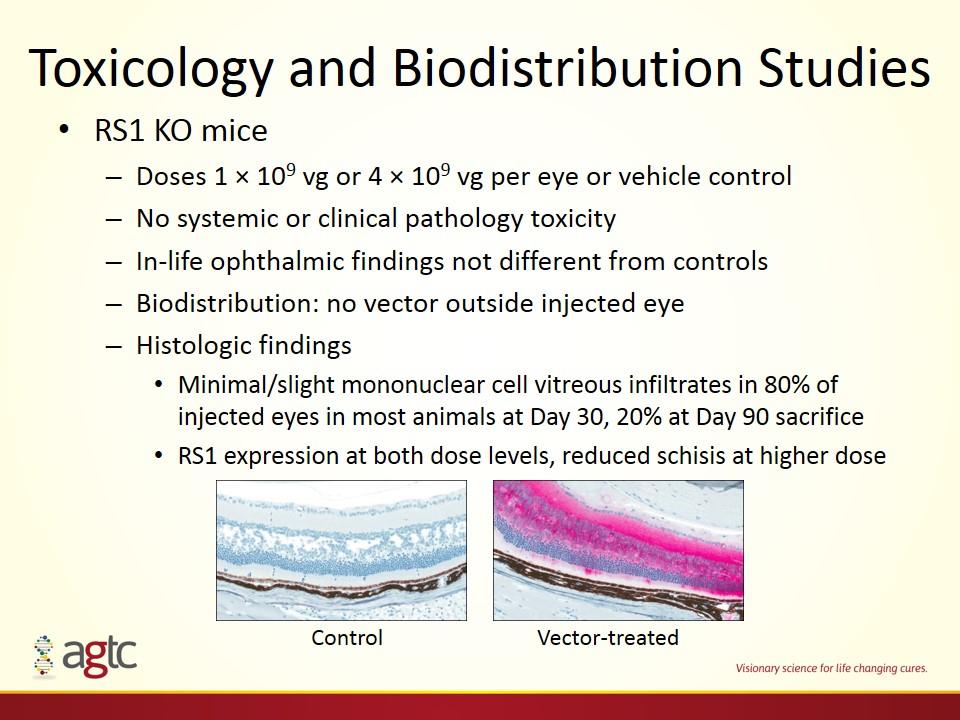
Toxicology and Biodistribution Studies RS1 KO mice Doses 1 × 109 vg or 4 × 109 vg per eye or vehicle control No systemic or clinical pathology toxicity In-life ophthalmic findings not different from controls Biodistribution: no vector outside injected eye Histologic findings Minimal/slight mononuclear cell vitreous infiltrates in 80% of injected eyes in most animals at Day 30, 20% at Day 90 sacrifice RS1 expression at both dose levels, reduced schisis at higher dose Control Vector-treated

Toxicology and Biodistribution Studies Normal cynomolgus macaques Doses 4 × 1010 and 4 × 1011 vg per eye vs. control No systemic or clinical pathology toxicity No effect on IOP, ERG or VEP Dose-related ophthalmic inflammatory response that improved over time Histologic findings Minimal/moderate mononuclear cell vitreous infiltrates in injected eyes in 2 of 6 low dose and 4 of 6 high dose animals RS1 expression at both dose levels Biodistribution: little vector outside injected eye
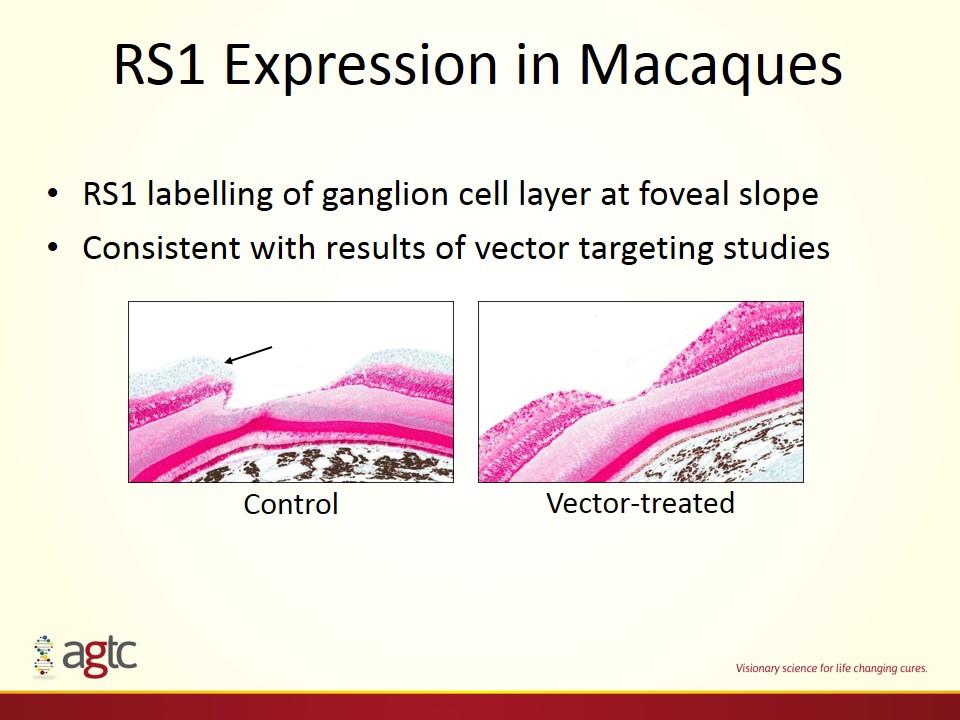
RS1 Expression in Macaques RS1 labelling of ganglion cell layer at foveal slope Consistent with results of vector targeting studies Control Vector-treated
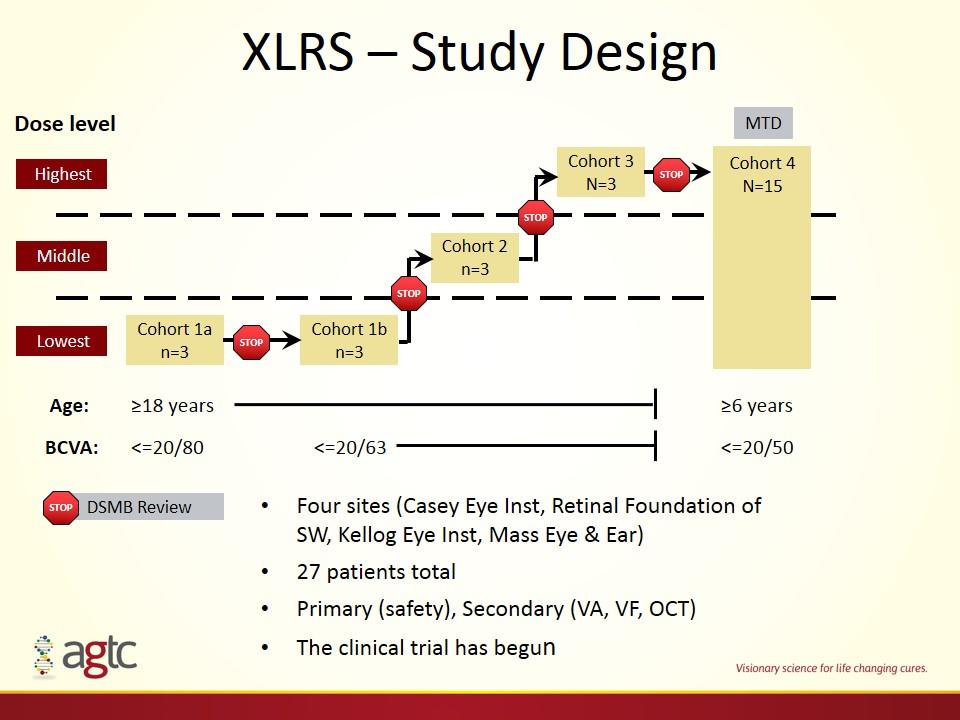
XLRS – Study Design Cohort 2 n=3 Cohort 1a n=3 Cohort 1b n=3 Cohort 3 N=3 Lowest Middle Highest DSMB Review Dose level STOP Four sites (Casey Eye Inst, Retinal Foundation of SW, Kellog Eye Inst, Mass Eye & Ear) 27 patients total Primary (safety), Secondary (VA, VF, OCT) The clinical trial has begun Age: ≥18 years ≥6 years BCVA: <=20/80 Cohort 4 N=15 MTD <=20/63 <=20/50 STOP STOP STOP STOP
Q&A

Pipeline: Multiple Shots on Goal Program Estimated US/EU patient population Proof-of-concept IND-enabling Clinical Development Key Milestones Lead programs in ophthalmology XLRS 35,000 Initial clinical data second half 2015 ACHM (B3) 13,500 Initial clinical data late 2015 (A3) 7,000 IND-enabling studies second half 2015 XLRP 20,000 Additional preclinical studies 2015 AMD 3,200,000 Target selection late 2015 New eye indications various Initial preclinical studies 2015 NON-ophthalmology PROGRAMS AAT deficiency (lung disease) 118,000 Additional preclinical studies Orphan designation US & EU Orphan designation US & EU Orphan designation US & EU

Achromatopsia Clinical Overview Joseph Carroll, PhD Medical College of Wisconsin

ACHM first described in late 19th century Autosomal recessive; ~1:30,000 incidence Caused by defects in phototransduction proteins – CNGA3, CNGB3, GNAT2, PDE6C, PDE6H Most cases caused by CNGA3 or CNGB3 defects Achromatopsia
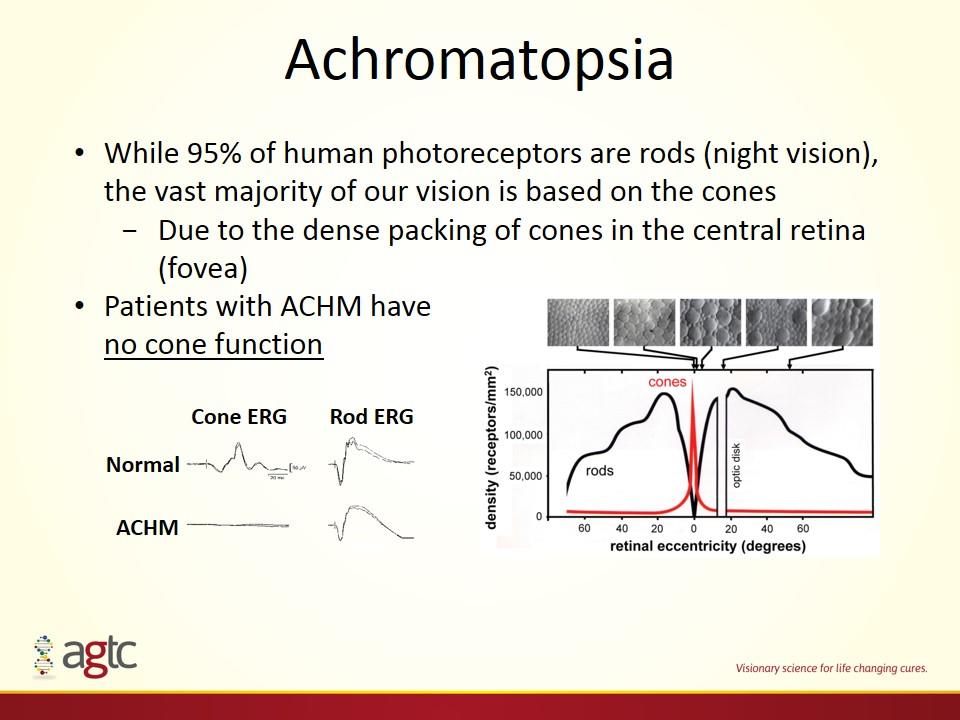
While 95% of human photoreceptors are rods (night vision), the vast majority of our vision is based on the cones Due to the dense packing of cones in the central retina (fovea) Patients with ACHM have no cone function ACHM Normal Cone ERG Rod ERG Achromatopsia
Often recognized at an early age Characterized by light aversion (photophobia), unsteady fixation, and nystagmus Proper diagnosis requires color vision testing and electroretinography (ERG) Clinically, visual acuity is substantially reduced – around 20/80-20/200 Generally, the fundus appears normal Diagnosis

Current Therapy for ACHM No specific treatment, focus is management Dark lenses reduce light-induced discomfort Low vision aids (high-powered magnifiers) for reading Preferential seating in the front of the class for school children

Restoration of cone function and/or cone-guided visual behavior has been demonstrated in multiple animal models: Dog Komaromy et al., 2010 Mouse Michalakis et al., 2012; Pang et al., 2012; Carvalho et al., 2011; Michalakis et al., 2010; Alexander et al., 2007 Sheep Banin et al., 2014 Animal Models for ACHM

Dog Model of Achromatopsia CNGB3 mutation causes absent ERG cone response, inability to navigate maze in bright light Subretinal delivery of AAV vector expressing CNGB3 rescues ERG responses and maze navigation in light-adapted conditions CNGB3-/- untreated CNGB3-/- treated Normal ERG Cone Response ACHM dog video FPO
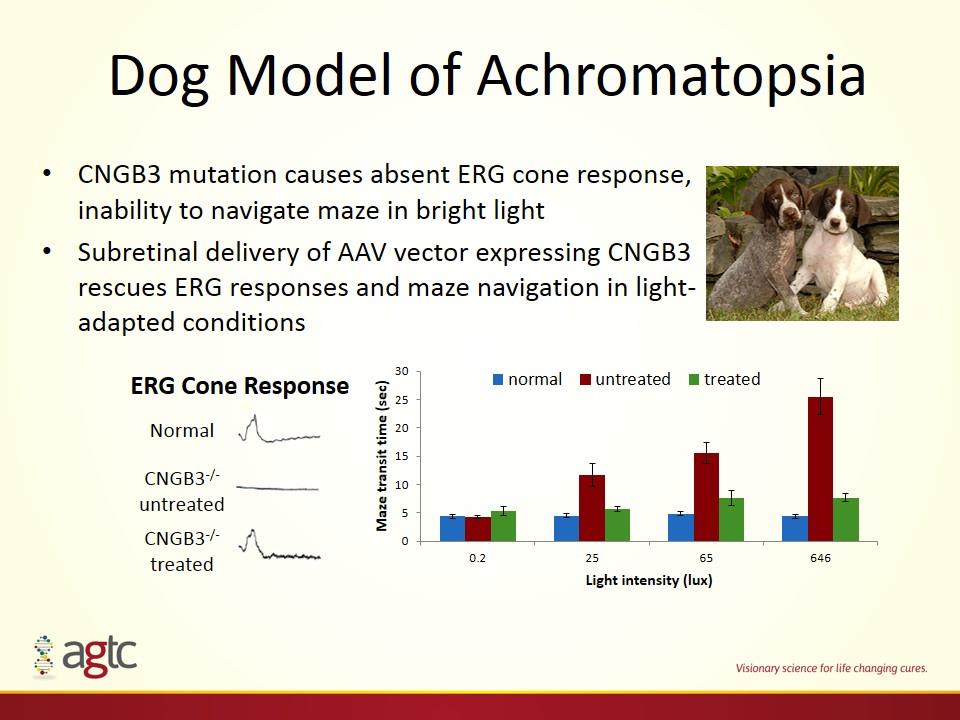
Dog Model of Achromatopsia CNGB3 mutation causes absent ERG cone response, inability to navigate maze in bright light Subretinal delivery of AAV vector expressing CNGB3 rescues ERG responses and maze navigation in light-adapted conditions CNGB3-/- untreated CNGB3-/- treated Normal ERG Cone Response

Photoreceptor Structure in ACHM Highly variable findings from histology Normal cones in the periphery, reduced numbers throughout, normal numbers in the fovea, or abnormal cones throughout… “…identifying and then targeting retinal locations with retained photoreceptors will be a prerequisite for successful gene therapy in humans….” Jacobson et al. (2005) Need for noninvasive tools for assessing photoreceptor structure in patients with ACHM Clinical and Genetic Characterization of Individuals With Achromatopsia. Clinicaltrials.gov Web site. https://clinicaltrials.gov/ct2/show/NCT01846052?term=NCT01846052&rank=1. Accessed June 10, 2015.

Natural History Study Five sites Casey Eye Institute, Bascom Palmer, Chicago Lighthouse, Vitreo Retinal Associates, Medical College of Wisconsin 50 patients with CNGB3-related ACHM Testing at 0, 6 and 12 months BCVA, light discomfort, color vision, perimetry, reading speed, OCT, AOSLO, ERG
Imaging the Human Retina
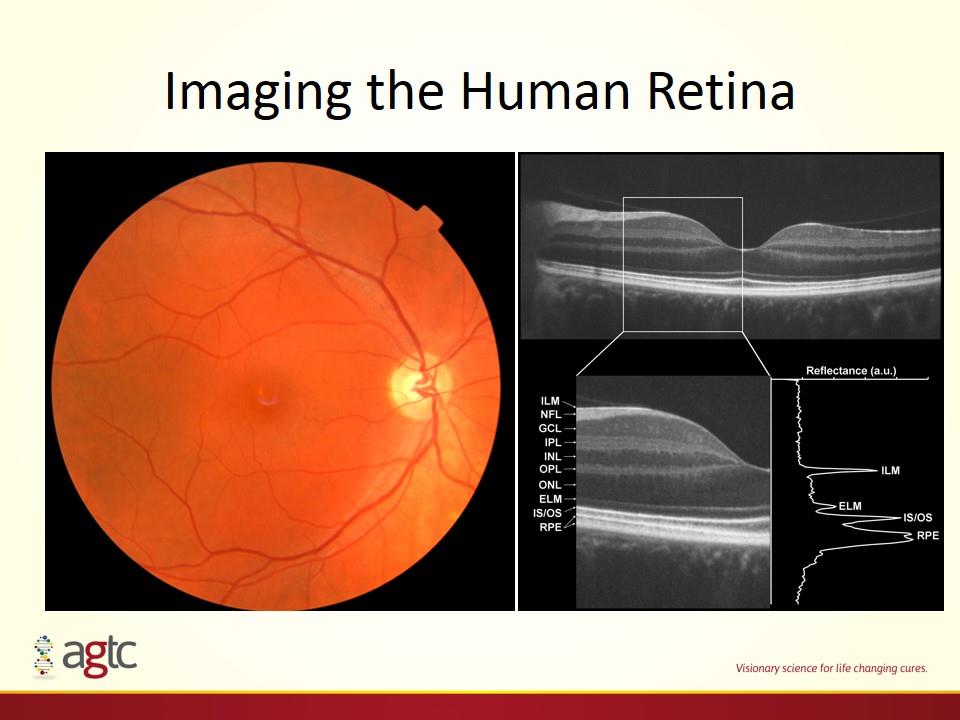
Imaging the Human Retina
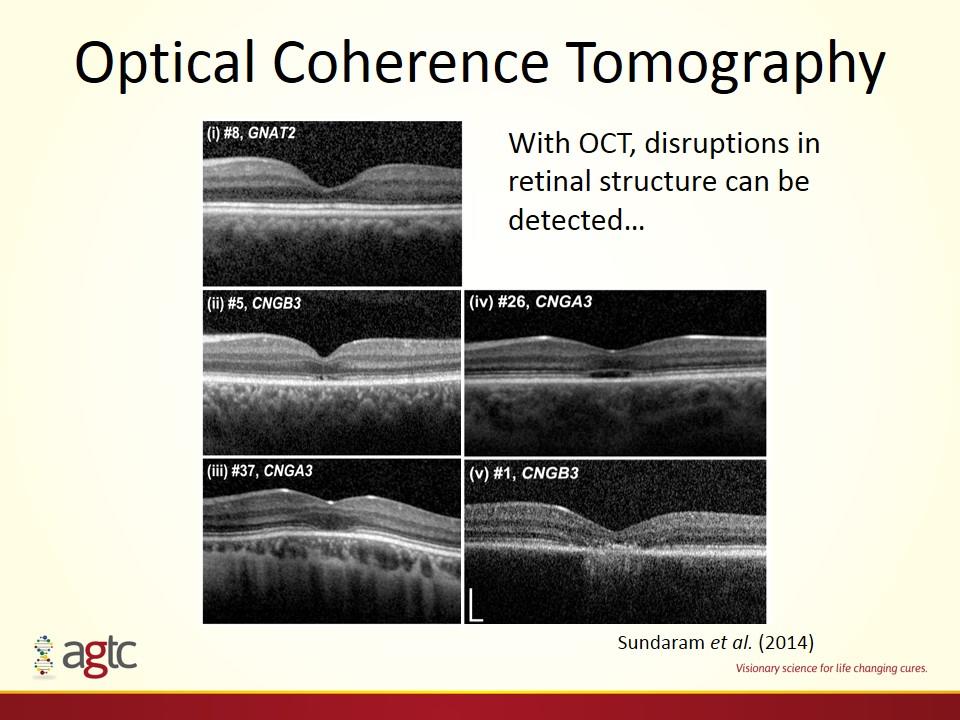
With OCT, disruptions in retinal structure can be detected… Sundaram et al. (2014) Optical Coherence Tomography
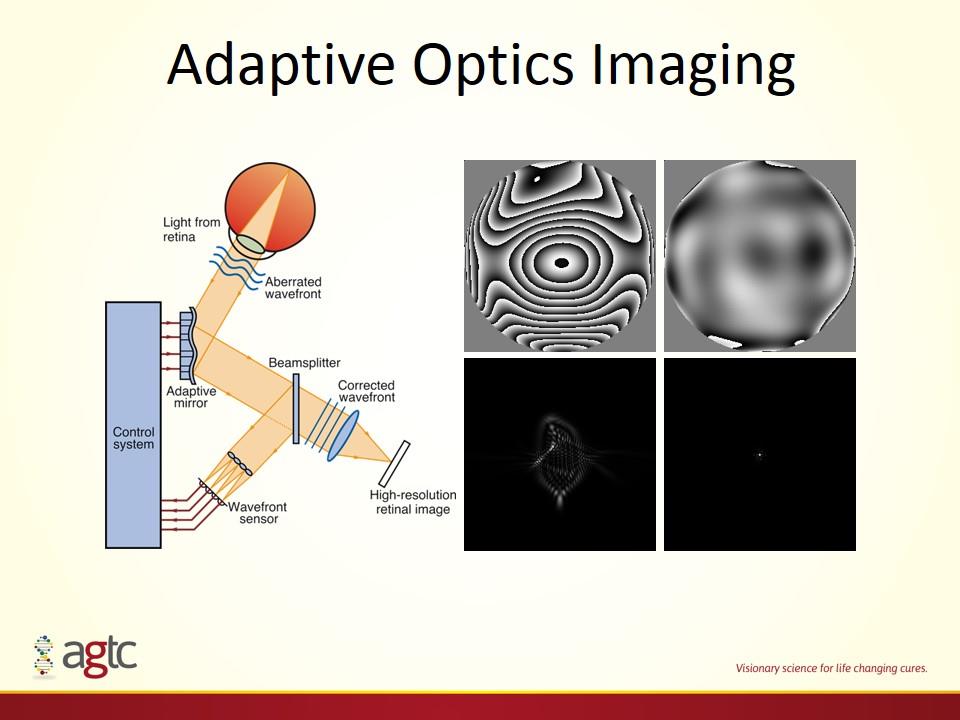
Adaptive Optics Imaging

Adaptive Optics Imaging
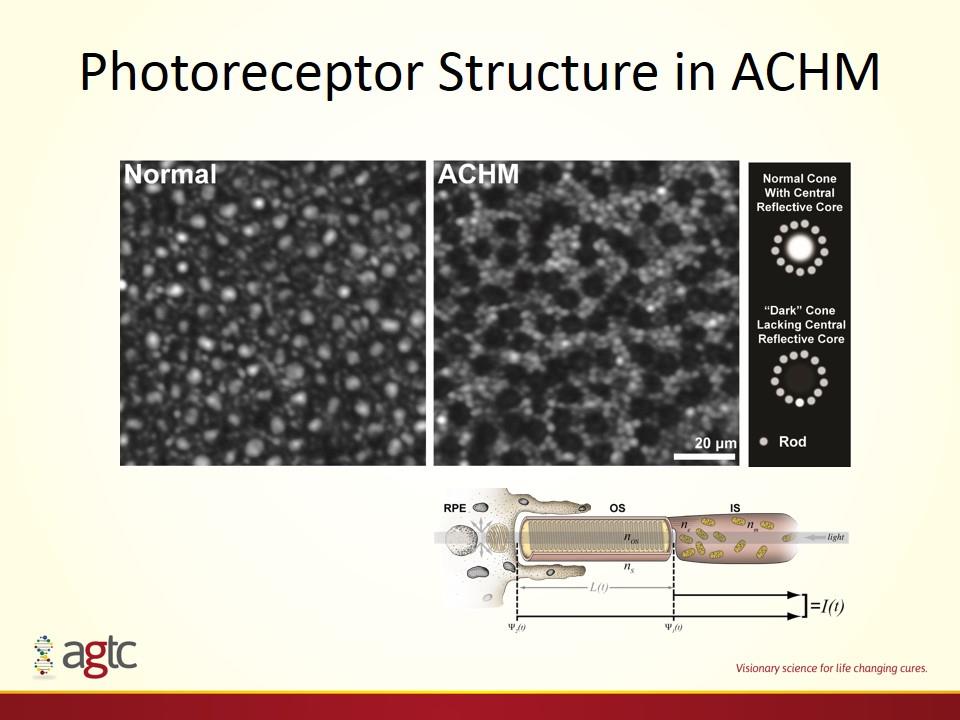
Photoreceptor Structure in ACHM

Photoreceptor Structure in ACHM

Remnant Cone Structure in ACHM

Remnant Cone Structure in ACHM

Remnant Cone Structure in ACHM

Remnant Cone Structure in ACHM High variability in the number of remaining cones, nearly 10-fold No evidence of any progression over an 18-month follow-up period Remaining cones represent the potential target for gene therapy efforts in ACHM patients, thus may define the therapeutic potential on an individualized basis Imaging may also provide a means to assess therapeutic efficacy
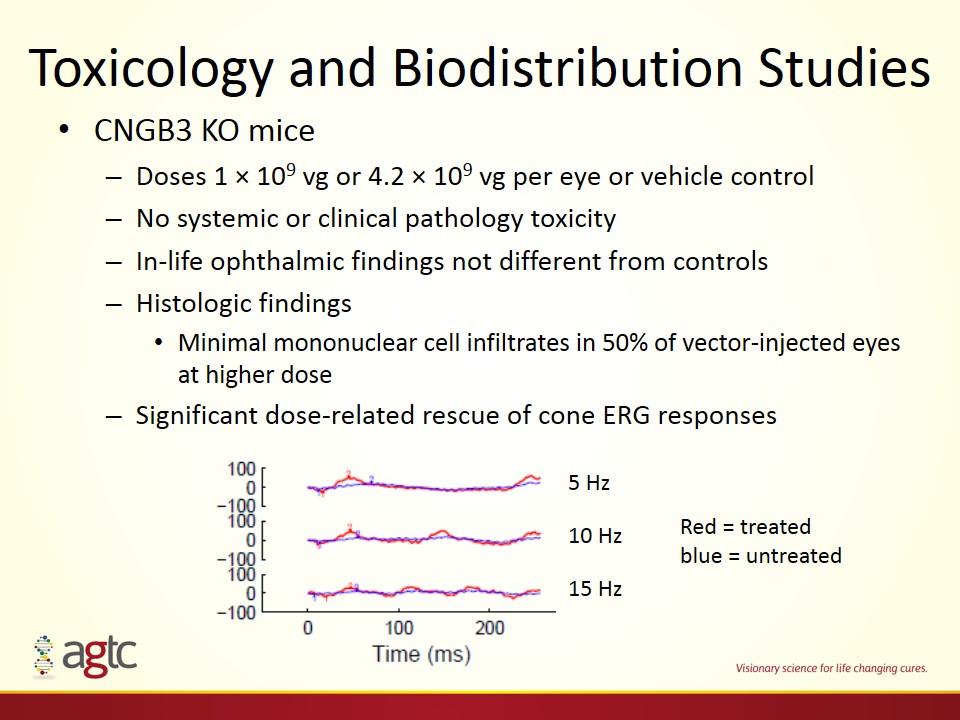
Toxicology and Biodistribution Studies CNGB3 KO mice Doses 1 × 109 vg or 4.2 × 109 vg per eye or vehicle control No systemic or clinical pathology toxicity In-life ophthalmic findings not different from controls Histologic findings Minimal mononuclear cell infiltrates in 50% of vector-injected eyes at higher dose Significant dose-related rescue of cone ERG responses 5 Hz 10 Hz 15 Hz Red = treated blue = untreated

Toxicology and Biodistribution Studies Normal Cynomolgus Macaques Doses 1.2 × 1011 and 1.2 × 1012 vg per eye vs. control No systemic or clinical pathology toxicity No effect on IOP, ERG or VEP Dose-related ophthalmic inflammatory response that improved over time Histologic findings Minimal mononuclear cell infiltrates in vitreous/optic disc in vector-injected eyes at both dose levels Biodistribution: little vector outside injected eye
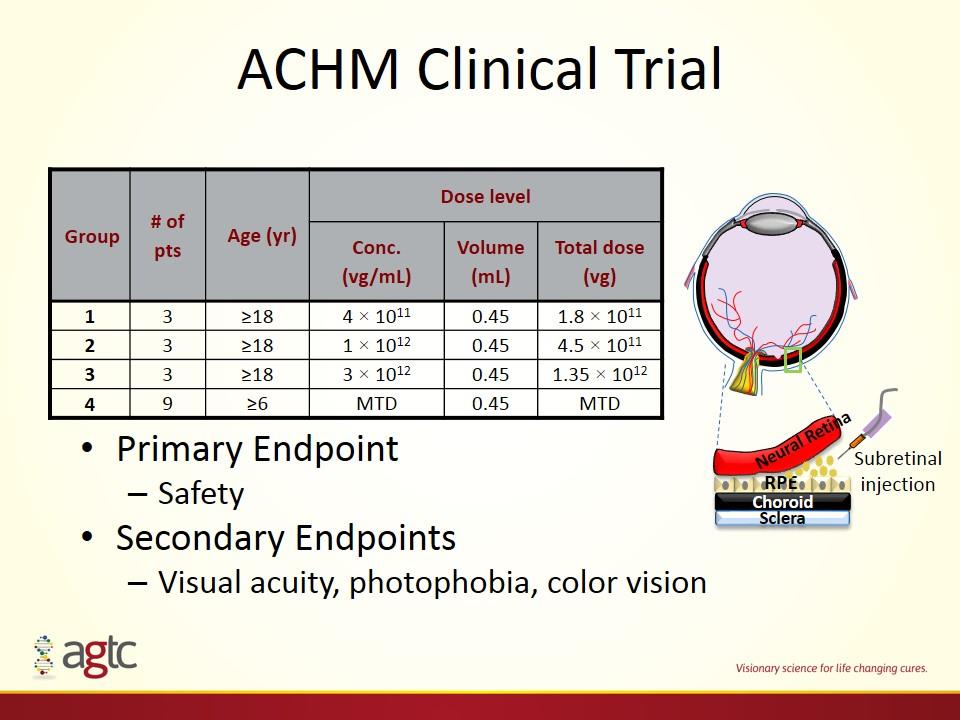
ACHM Clinical Trial Primary Endpoint Safety Secondary Endpoints Visual acuity, photophobia, color vision Group # of pts Age (yr) Dose level Conc. (vg/mL) Volume (mL) Total dose (vg) 1 3 ≥18 4 × 1011 0.45 1.8 × 1011 2 3 ≥18 1 × 1012 0.45 4.5 × 1011 3 3 ≥18 3 × 1012 0.45 1.35 × 1012 4 9 ≥6 MTD 0.45 MTD Neural Retina RPE Choroid Sclera Subretinal injection
Q&A
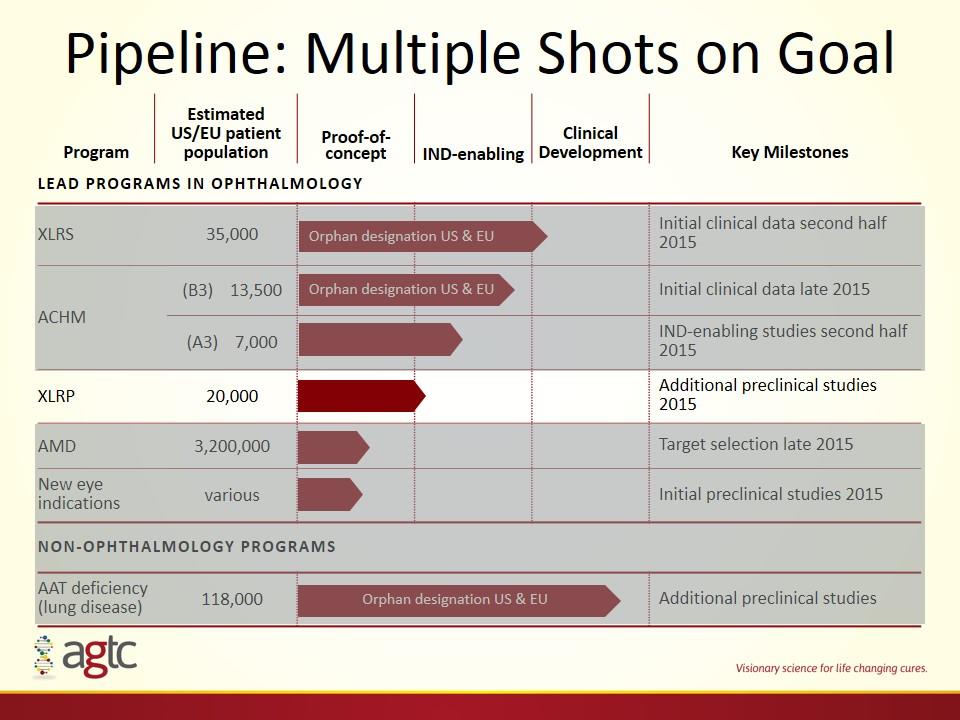
Pipeline: Multiple Shots on Goal Program Estimated US/EU patient population Proof-of-concept IND-enabling Clinical Development Key Milestones Lead programs in ophthalmology XLRS 35,000 Initial clinical data second half 2015 ACHM (B3) 13,500 Initial clinical data late 2015 (A3) 7,000 IND-enabling studies second half 2015 XLRP 20,000 Additional preclinical studies 2015 AMD 3,200,000 Target selection late 2015 New eye indications various Initial preclinical studies 2015 NON-ophthalmology PROGRAMS AAT deficiency (lung disease) 118,000 Additional preclinical studies Orphan designation US & EU Orphan designation US & EU Orphan designation US & EU

X-linked Retinitis Pigmentosa Stephen M. Rose, Ph.D. Chief Research Officer Foundation Fighting Blindness

Unknown Other ARRP ADRP XLRP Usher BBS LCA Genetics of Retinitis Pigmentosa RP is an inherited degenerative eye disease Severe vision impairment and often blindness Prevalence ≈1:4000, up to 100,000 patients in US Estimated that ≈15% of RP cases caused by mutations in Retinitis Pigmentosa GTPase Regulator (RPGR) gene One of most common causes of RP RPGR mutations account for 70% of X-linked retinitis pigmentosa (XLRP) cases, 8% of adRP, 15% of isolated male case About 15,000 patients in US

Inheritance of XLRP Similar to XLRS, but some females are affected Span the range from very mild to almost as severe as males Due to mosaicism of X chromosome inactivation

RPGR Retinitis pigmentosa GTPase regulator Cilium connects inner and outer segments of photoreceptors RPGR helps to transport proteins from inner to outer segments Mutations in RPGR result in abnormal transport and leads to photoreceptor degeneration 19 ORFs, multiple mRNA isoforms ORF 1-19 isoform expressed in various cells ORF 1-15 (“ORF15”) expressed in photoreceptors 3459 bp, 1152 amino acids 1.2 kb highly repetitive at exon 15 (mutation “hot spot”) RPE cell rod RPGR Outer segment Inner segment

XLRP Due to RPGR Mutations Rapidly progressive form of RP Night blindness in first decade Progressive visual field constriction 4% annual loss of BCVA Legally blind by median age of 40-45 years

XLRP Diagnosis Suspect in male with onset of RP in teens Confirm by genetic testing Highly repetitive ORF15 complicates testing

Current Therapy for XLRP There is currently no effective treatment for XLRP Vitamin A may slow rate of progression Artificial retina for patients who progress to complete loss of vision

Animal Models Mouse models rd9 mice have ORF15 frameshift → truncated protein RPGR KO mice have disruption of exons 4 to 6 Both have relatively slow rate of retinal degeneration Dog models XLPRA1, 4 bp RPGR deletion, truncated protein, juvenile onset, progresses over several years XLPRA2, 2 bp RPGR deletion, truncated protein, early onset, rapid progression

Proof-of-concept in XLRP Dog Model From Beltran et al., PNAS, 2012 Red = treated Black untreated Cone responses Rod responses a-wave b-wave ERG Preservation Photoreceptor Preservation Treated area Untreated area Photoreceptor nuclei Photoreceptor nuclei

Natural History Study Single site (Scheie Eye Institute) 30 patients with RPGR-related XLRP Testing at 0 and 18-24 months BCVA, ophthalmic exam, OCT with quantification of retinal layers, 2-color rod and cone sensitivity, autofluorescence imaging
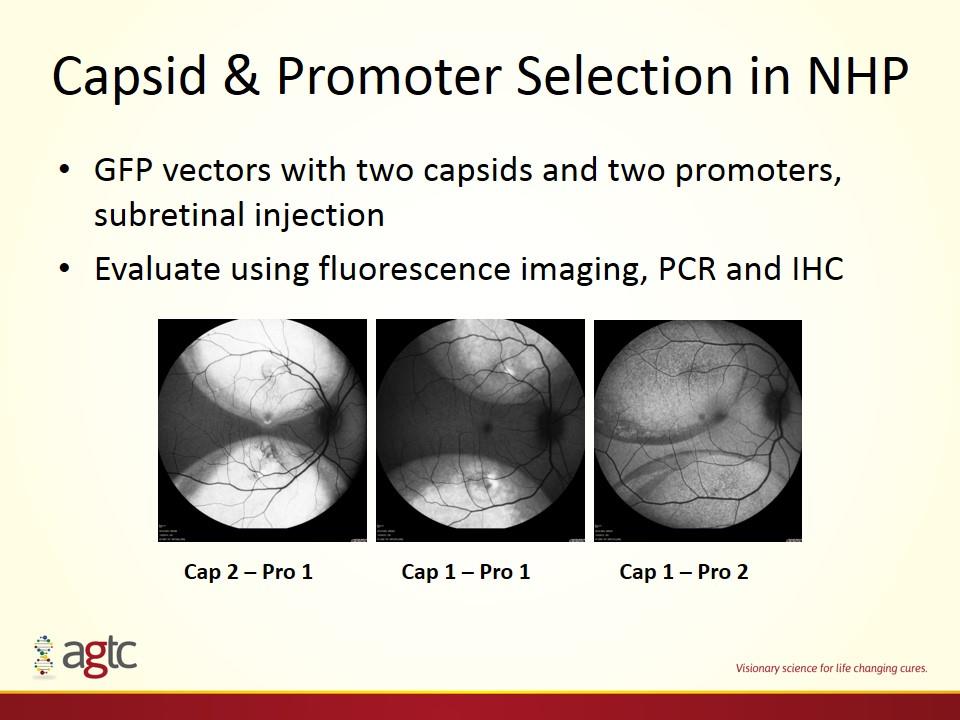
Capsid & Promoter Selection in NHP GFP vectors with two capsids and two promoters, subretinal injection Evaluate using fluorescence imaging, PCR and IHC Cap 2 – Pro 1 Cap 1 – Pro 1 Cap 1 – Pro 2

Additional Pre-IND Activities Re-confirm efficacy in RPGR-deficient dogs Toxicology and biodistribution Design based on studies with other products Production of clinical trial study drug Using AGTC’s HSV-based method
Q&A

Vector Design & Manufacturing David R. Knop Sr. Director, Process Development

Overview Manufacturing by H.A.V.E. Herpes-assisted vector expansion Safety & Characterization Summary
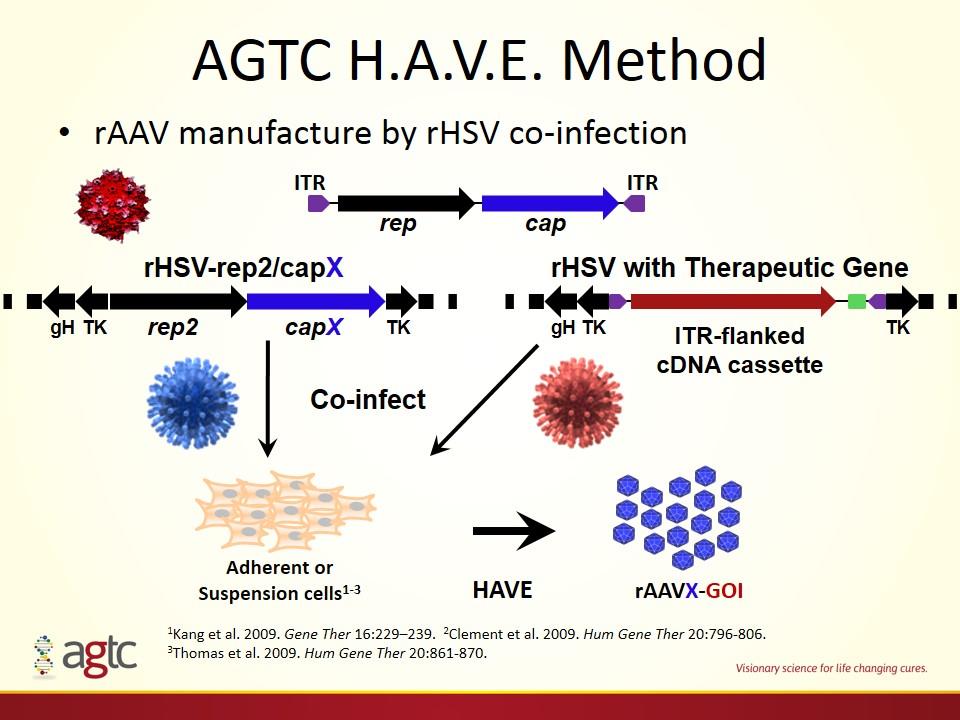
AGTC H.A.V.E. Method rAAV manufacture by rHSV co-infection HAVE rAAVX-GOI 1Kang et al. 2009. Gene Ther 16:229–239. 2Clement et al. 2009. Hum Gene Ther 20:796-806. 3Thomas et al. 2009. Hum Gene Ther 20:861-870. rHSV-rep2/capX rep2 capX TK ITR-flanked cDNA cassette rHSV with Therapeutic Gene Co-infect TK gH TK gH TK rep cap ITR ITR Adherent or Suspension cells1-3

Competitive Manufacturing Edge Attribute Advantage Yield Cost effective – 25-30 fold increase in potency & purity achieved Development Minimize time to the clinic - reproducible genetic manipulation, stable constructs Characterization AAV product candidates evaluated by qualified assays Scale Commercial scale for eye programs - all unit operations scalable Reproducible Successfully transferred to partners & CMOs Flexibility Multiple serotypes & transgenes – rAAV1 & rAAV2 in the clinic Regulatory FDA & EMA approved for use in human trials
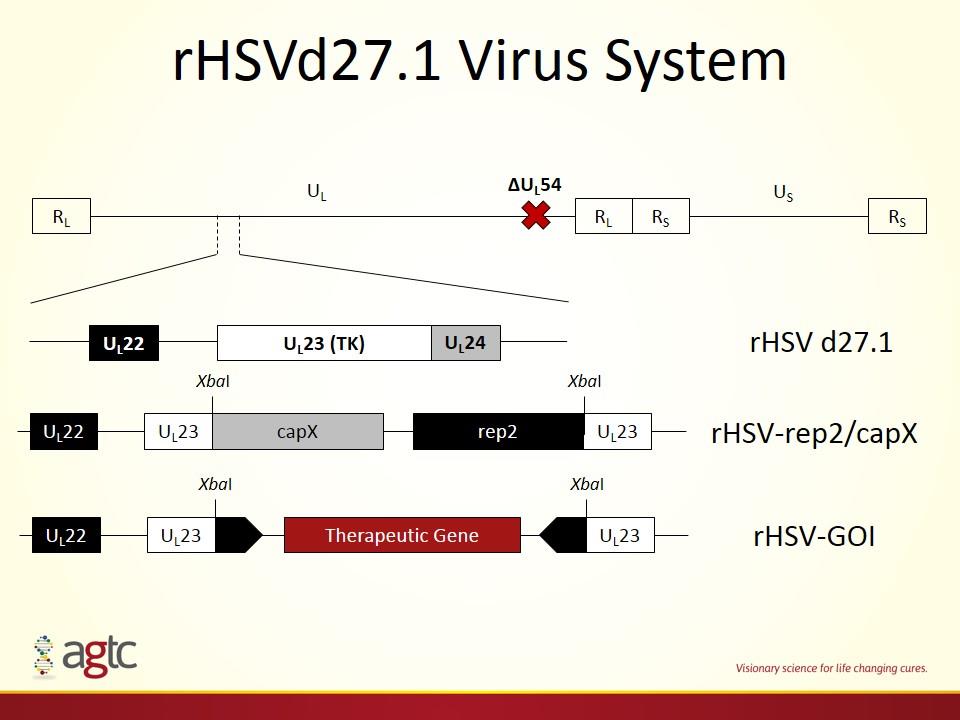
rHSVd27.1 Virus System UL23 (TK) UL22 UL24 rHSV-rep2/capX UL22 UL23 UL23 capX rep2 XbaI XbaI rHSV d27.1 RL RL RS RS UL US ∆UL54 UL22 UL23 UL23 Therapeutic Gene XbaI XbaI rHSV-GOI
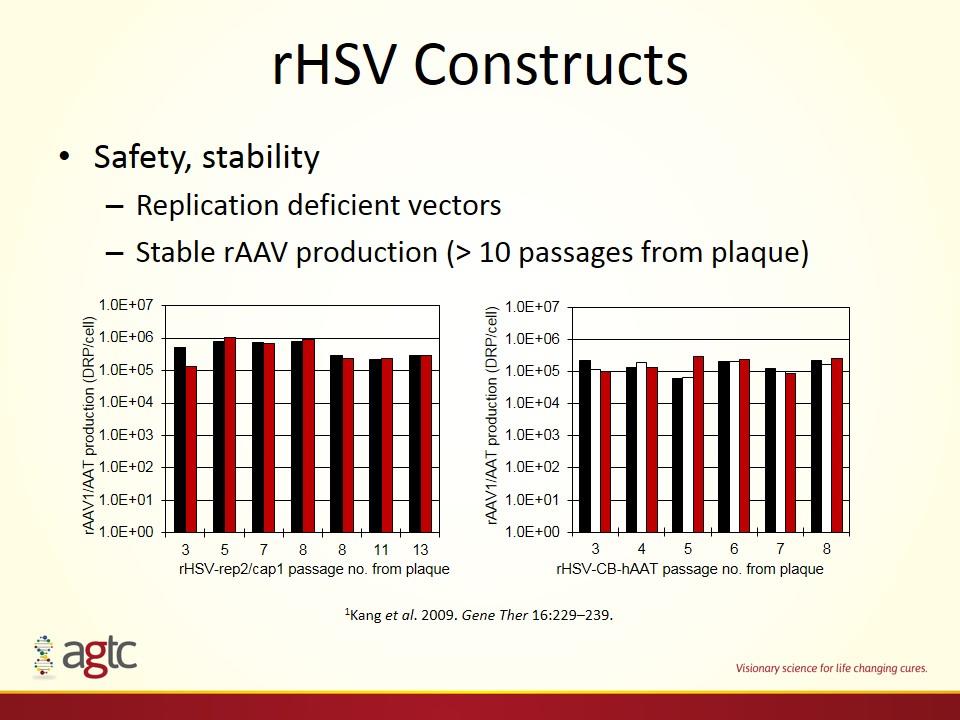
rHSV Constructs Safety, stability Replication deficient vectors Stable rAAV production (> 10 passages from plaque) 1Kang et al. 2009. Gene Ther 16:229–239.

rAAV Production & Processing Ophthalmology Wave bioreactor, suspension Primary clarification (filtration) Filter Drug Substance: TFF/DF (concentrate, formulate) Cation exchange chromatography Affinity capture chromatography Drug Product: Aliquot & store
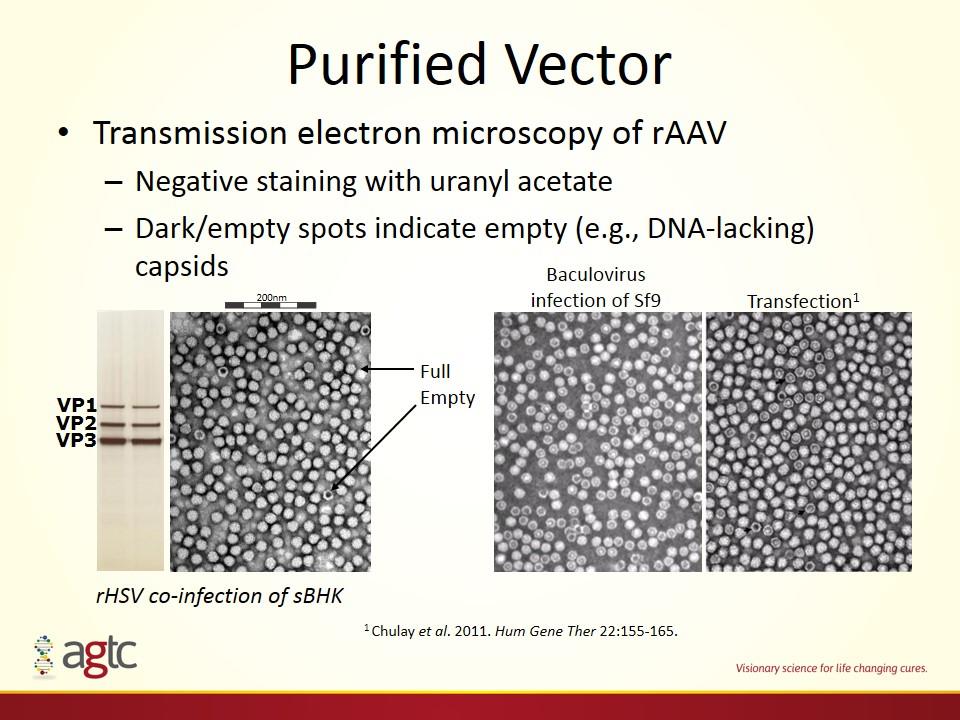
Purified Vector Transmission electron microscopy of rAAV Negative staining with uranyl acetate Dark/empty spots indicate empty (e.g., DNA-lacking) capsids 200nm Full Empty VP1 VP2 VP3 rHSV co-infection of sBHK Baculovirus infection of Sf9 Transfection1 1 Chulay et al. 2011. Hum Gene Ther 22:155-165.

Viral Clearance Viral clearance studies for multiple serotypes Downstream challenged with wild-type HSV High safety margin – XLRS example A single dose would deliver < 2.64 × 10-7 pfu of HSV Parameter Clearance (log10) Overall clearance > 13.6 Detergent > 7.34 Column 1 4.54 Column 2 1.75

H.A.V.E. method addresses off-target DNA packaging concerns Helper virus DNA packaging Chadeuf et al. 20051 (0.5%-6.3% ampR packaging) 2012 EMA assessment report of Glybera2 EMA requested risk assessment of protein expression from packaged helper vector DNA Previously evaluated by AGTC for HSV-made AAV3 Gene size - UL26.5 (1.0 kb), UL33 (1.8 kb), UL35 (0.38 kb) Function/kinetic class - a0, UL26.5, UL29 (IE, E, L) Helper virus & cellular DNA levels: 0.007-0.012% Genes Assessment Method Result UL26.5, UL29, UL33, UL35, UL36 Transcription RT-qPCR < LLOD a0, UL26.5, UL29 Translation Immunofluorescence < LLOD

Characterization Over 30 assays developed & transferred Biologic Assay description Assays developed rHSV banks Identity, genetic 8 NAb (myco, in vitro, porcine, bovine) 3 Concentration 1 Replication competent HSV 1 AAV CTM Expression, protein 3 Identity, genetic 5 Purity 1 Infectivity 1 Concentration 1 Replication competent AAV, HSV 2 Host cell contaminants 2 Helper virus contaminants 2

Summary Suspension processes for helper and product All unit operations scalable (reduce COGS) Robust process Multiple preclinical and clinical lots manufactured Transferred to CMOs/partner Multiple serotypes Process reviewed by regulatory agencies FDA in approved INDs EMA in approved CTA

Closing Remarks Sue Washer President and CEO

/AppliedGeneticTechnologiesCorporation @AGTC_ www.agtc.com

AGTC Analyst and Investor Day June 22, 2015