Exhibit 99.1
| Bringing Innovative Therapeutics for People with Orphan Liver Disease MARCH 2015 NASDAQ: OCRX |
| Forward-Looking Statements Certain statements in this presentation constitute “forward-looking statements” within the meaning of the Securities Act of 1933, as amended (the “Securities Act”), and Securities Exchange Act of 1934, as amended (“Exchange Act”), including, without limitation, all statements related to the OCR-002 clinical development program, including patient enrollment estimates, expected timing for the receipt of clinical data, and the potential success of OCR-002 in clinical trials, and we intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in the Securities Act and the Exchange Act and are making this statement for purposes of complying with those safe harbor provisions. These forward-looking statements reflect our current views about our plans, intentions, expectations, strategies and prospects, which are based on the information currently available to us and on assumptions we have made. Although we believe that our plans, intentions, expectations, strategies and prospects as reflected in or suggested by those forward-looking statements are reasonable, we can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a variety of risks and factors that are beyond our control, including those risks and uncertainties discussed under “Risk Factors” in our Annual Report on form 10-K for the year ended December 31, 2013, as well as other risks detailed in our subsequent filings with the SEC. All information in this presentation is as of the date of this presentation, and we undertake no duty to update this information unless required by law. |
| “When the liver fails, the brain fails.” Professor Rajiv Jalan; University College London |
| Building a Liver Disease Medicines Company Clinical-stage development for treatment of Hepatic Encephalopathy IV and oral formulations Strong patent coverage Differentiated Product Candidate: OCR-002 STOP-HE Phase 2b interim analysis Completion of Phase 2b enrollment Results of two investigator-sponsored Phase 2a studies Multiple Catalysts in 2015 Funds to support Ocera through upcoming milestones Strong Balance Sheet |
| Liver Disease: Caused by Many Conditions Chronic Liver Disease in U.S. 8 Million Alcohol Use Hepatitis Obesity Drug Exposure NASH / Fatty Liver Diabetes Autoimmune Diseases |
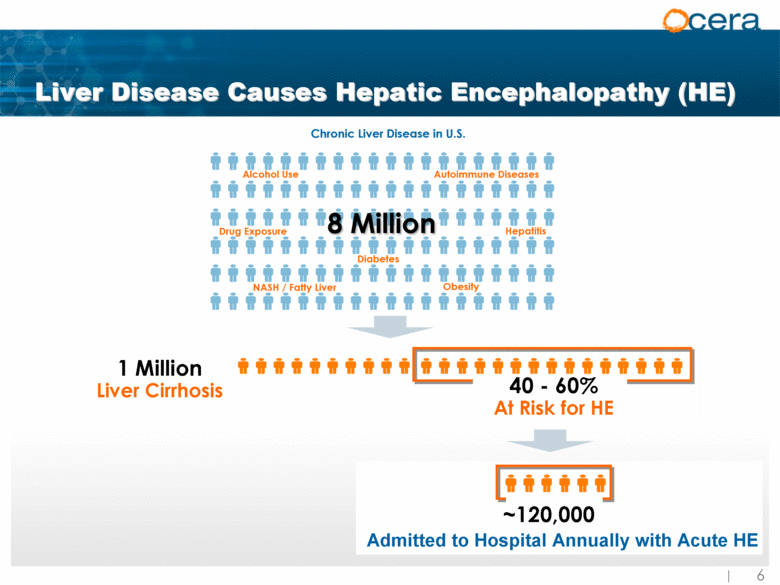
| Liver Disease Causes Hepatic Encephalopathy (HE) 1 Million Liver Cirrhosis 40 - 60% At Risk for HE ~120,000 Admitted to Hospital Annually with Acute HE |
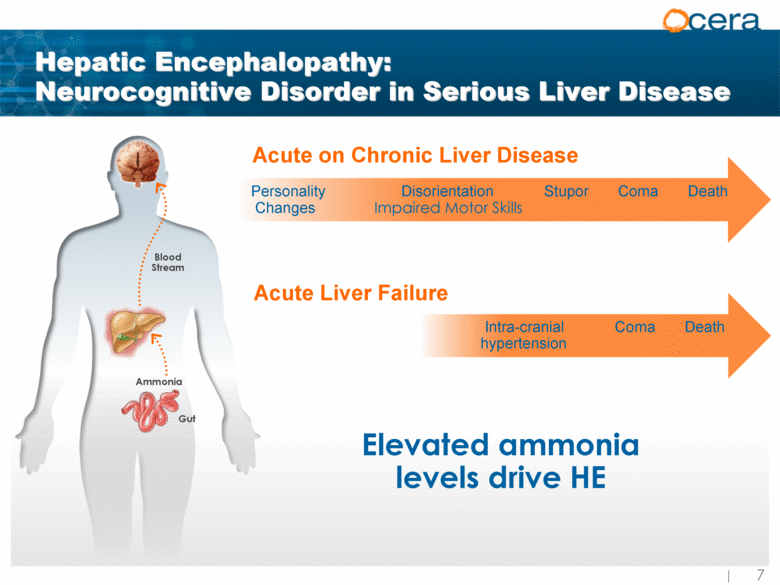
| Hepatic Encephalopathy: Neurocognitive Disorder in Serious Liver Disease Blood Stream Ammonia Gut Acute on Chronic Liver Disease Acute Liver Failure Elevated ammonia levels drive HE Personality Disorientation Stupor Coma Death Changes Impaired Motor Skills Intra-cranial Coma Death hypertension |
| Growing Body of Evidence Supports Correlation Between Blood Ammonia Levels and HE Severity of HE Symptoms Ammonia Intracranial Pressure Ammonia Ammonia measurements shown to correlate (p < 0.001) with severity of HE Independent, prospective study of 121 hospitalized patients with chronic cirrhosis1 Higher ammonia levels shown to correlate with greater frequency of HE and elevated intracranial pressure2 Independent study in acute liver failure 1 “Correlation between Ammonia Levels and the Severity of Hepatic Encephalopathy” Ong, et al. Am J Med. 2003; 114:188-193 2 “Arterial Ammonia and Clinical Risk Factors for Encephalopathy and Intracranial Hypertension in Acute Liver Failure” Bernal, et al. Hepatology, Vol. 46, No.6, 2007 |
| Treatment of Acute HE: Need for New Therapy Standard of care in acute and chronic HE Laxative, indirectly reduces ammonia by flushing gut Difficult administration and diarrhea Poor patient compliance Lactulose Non-absorbed antibiotic approved for prevention of HE Limited evidence of efficacy in acute care of hospitalized cirrhotic patients with HE1 Does not directly clear circulating ammonia Rifaximin 1 “Rifaximin vs Conventional Oral Therapy for Hepatic Encephalopathy: a Meta-analysis” Eltawil, et al. World J of Gastroenterology, 2012 |
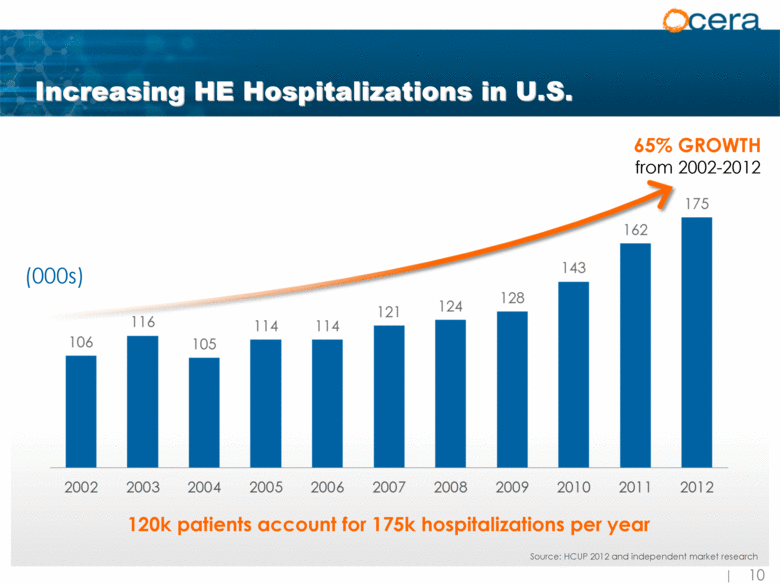
| Increasing HE Hospitalizations in U.S. (000s) Source: HCUP 2012 and independent market research 65% GROWTH from 2002-2012 120k patients account for 175k hospitalizations per year Source: HCUP 2012 and independent market research |
| PAYMENT AMOUNT ON HE INPATIENT CLAIMS (2011)1 1 2011 CMS Medicare data and other commercial payors; N = 73,467 2 Ocera commissioned market research showing that 80% of the 106 hospital surveyed would support OCR-002 at $1,300 per 24 hours if it reduces ICU stay Median Mean CMS $10,111 $15,056 Other Payors $18,095 $38,723 Hospital Charges $31,693 $63,564 Hospitals Financially Rewarded for Reducing Length of Stay Hospitals Lose Money on Patients with HE Medicare payments for HE relatively flat despite LOS Medicare pays $8-13K per HE episode for 3-14 day LOS Commercial payors better but still less than hospital charges Hospitals are highly motivated to reduce LOS Third-party price sensitivity analysis supports $1,300 for OCR-002 per 24 hours if it reduces ICU stay2 |
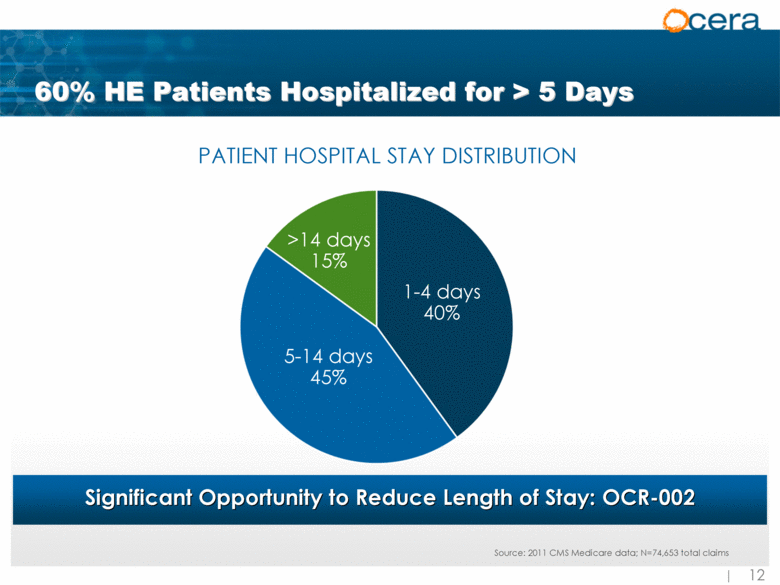
| 60% HE Patients Hospitalized for > 5 Days Significant Opportunity to Reduce Length of Stay: OCR-002 >14 days 15% 5-14 days 45% 1-4 days 40% PATIENT HOSPITAL STAY DISTRIBUTION Source: 2011 CMS Medicare data; N=74,653 total claims |
| The Demons of the Disease Progressive, episodic disorder with high mortality rate Majority of affected individuals eventually lose independence; significant impact on quality of life Advanced HE marked by delirium, overactive reflexes and coma1 Hospitalized patients can be very sick, stuporous, intubated and unable to eat 1 Al Sibae MR, McGuire BM. Current trends in the treatment of hepatic encephalopathy. Ther Clin Risk Manag. 2009;5(3):617-626 |
| Hospitalized HE Patients Need Rapid Treatment OCR-002 IV Ammonia scavenger Ammonia lowering observed in cirrhotic patients IV formulation for immediate, systemic action Well-tolerated in clinical trials to date |
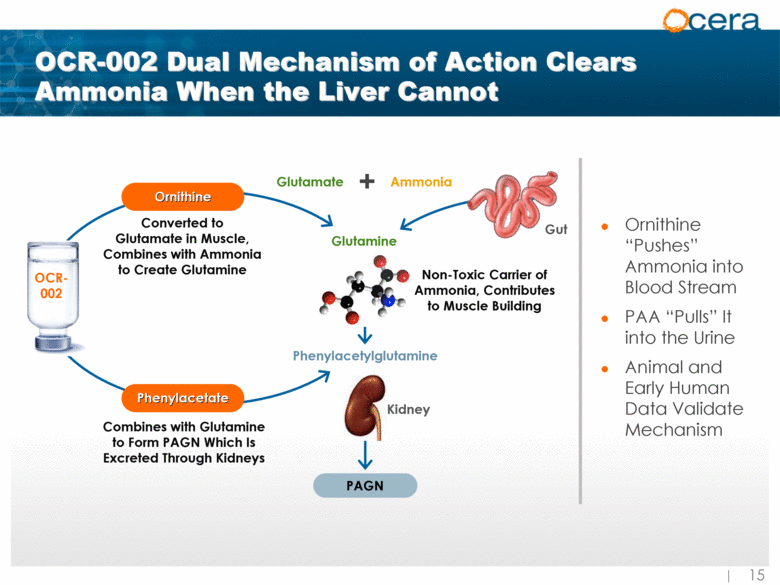
| Phenylacetate Ornithine Non-Toxic Carrier of Ammonia, Contributes to Muscle Building OCR-002 Dual Mechanism of Action Clears Ammonia When the Liver Cannot Ornithine “Pushes” Ammonia into Blood Stream PAA “Pulls” It into the Urine Animal and Early Human Data Validate Mechanism Converted to Glutamate in Muscle, Combines with Ammonia to Create Glutamine Gut Glutamine Glutamate Ammonia + OCR-002 Kidney PAGN Combines with Glutamine to Form PAGN Which Is Excreted Through Kidneys Phenylacetylglutamine Gut |
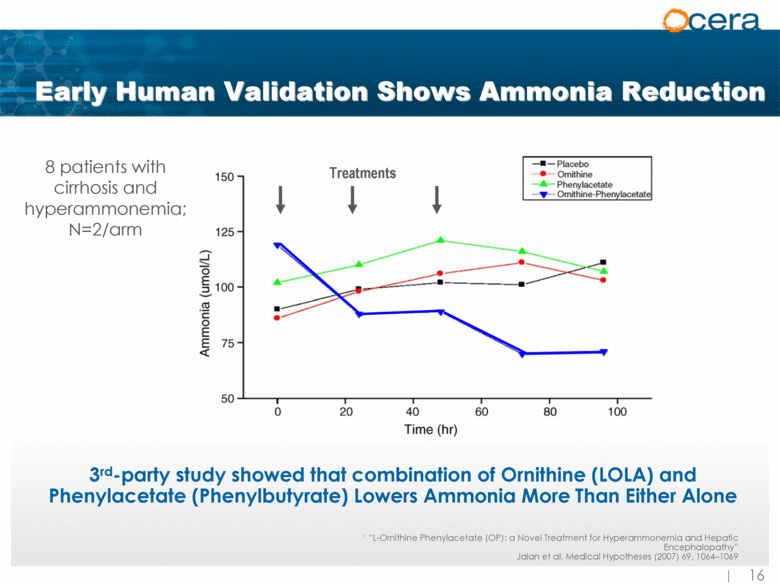
| Early Human Validation Shows Ammonia Reduction 3rd-party study showed that combination of Ornithine (LOLA) and Phenylacetate (Phenylbutyrate) Lowers Ammonia More Than Either Alone 8 patients with cirrhosis and hyperammonemia; N=2/arm 1 “L-Ornithine Phenylacetate (OP): a Novel Treatment for Hyperammonemia and Hepatic Encephalopathy” Jalan et al. Medical Hypotheses (2007) 69, 1064–1069 Treatments |
| OCR-002 IV Well-Tolerated in Phase 1 Trial No Serious Adverse Events; No Deaths or Discontinuations PK Profile Established Safety Margins Identified Stable Cirrhotic Patients, Ammonia Levels not Measured Maximum Dose Tested 40g per 24 hours Most frequent dose related toxicities at 40g included dizziness, headache, nausea, blurred vision Supports Phase 2b dose of 20g per 24 hours OCR-002 PHASE 1 PK/SAFETY PARALLEL ASCENDING DOSE STUDY Healthy Volunteers (N=48) Stable Cirrhotic Patients (N=43) |
| 2015 Phase 2 Trials of OCR-002 Phase 2b Trial in Acute HE Phase 2a Trial for Acute Liver Failure/Injury Phase 2a Trial in Cirrhosis and Upper GI Bleed IN PROGRESS COMPLETE |
| STOP-HE Phase 2b Ocera Study: OCR-002 IV for Acute HE Efficacy RANDOMIZED DOUBLE-BLIND TRIAL OCR-002 20g** per 24 hours + Standard of Care Placebo + Standard of Care HOSPITALIZED CIRRHOTICS Key Inclusion West Haven* Score > 2 Ammonia >ULN Key Exclusion Kidney failure/dialysis Life expectancy < 2 weeks ~140 patients ~100 U.S. and Europe sites Interim analysis for futility & sample size SECONDARY ENDPOINTS: Time to complete resolution of HE symptoms, ammonia reduction, length of hospital stay PRIMARY ENDPOINT: Time to meaningful clinical improvement in encephalopathy symptoms Powered 80% to detect 23% absolute treatment benefit using time to response analysis *As scored by a modified version of the West Haven Scale **May be adjusted to 15 or 10g based on liver impairment to achieve comparable exposure levels |
| Meaningful Clinical Improvement as Scored by Modified Version of West Haven Scale 4 3 2 1 0 --- Hospitalization --- Comatose Stuporous Disoriented Difficult to Arouse Asterixis Mild Confusion Normal Grade / Symptoms Meaningful Clinical Improvement 4 to 2 or lower 3 to 2 or lower 2 to 1 or lower Moderate Mild Severe |
| UGIB Phase 2a Investigator Trial - Completed OCR-002 IV in Cirrhosis & Upper GI Bleed (UGIB) PART B: 10g PER 24 HOURS PLACEBO-CONTROLLED(N=38) OCR-002 treatment arm vs. placebo: Greater absolute and relative reductions in median plasma ammonia at 12 hours after baseline More rapid hourly decline in plasma ammonia over the first 24 hours of infusion Statistically significant difference in urinary excretion of ammonia PART A: OPEN-LABEL OCR-002 (N=10) Rapid ammonia lowering in 24 hours in active cohort vs. no ammonia lowering within 24 hours in historical controls “The Current Study Demonstrates the Safety of OCR-002 when Administered to Patients with Decompensated Cirrhosis” * P < 0.05; Source: Ventura-Cots, et al., Safety of Ornithine Phenylacetate in Cirrhotic Decompensated Patients, J. of Clinical Gastroenterology, Volume 47, Number 10, Nov/Dec 2013 Ammonia Levels (mean) Baseline 24h After Baseline Historical Control (N=10) 83 µmol/L 84.5 µmol/L Active Cohort Dose-escalating (N=10) 80 µmol/L 52.9 µmol/L* Ammonia Levels (median) Baseline 12h After Baseline Hourly Decline First 24h Absolute Difference Rate Placebo (N=18) 95.83 µmol/L (N=16) -2.0 µmol/L (N=13) -0.24 µmol/L (N=18) OCR-002 10g/24hrs (N=20) 78.70 µmol/L (N=20) -19.35 µmol/L (N=17) -0.85 µmol/L (N=19) |
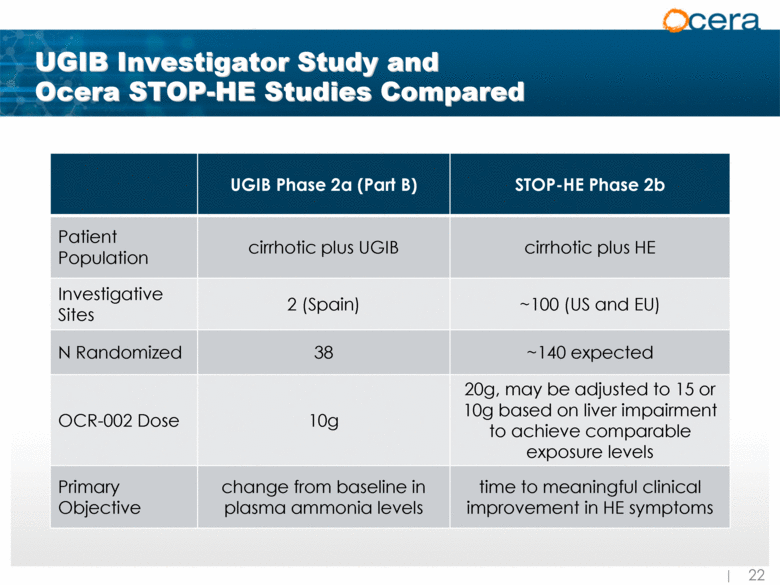
| UGIB Investigator Study and Ocera STOP-HE Studies Compared UGIB Phase 2a (Part B) STOP-HE Phase 2b Patient Population cirrhotic plus UGIB cirrhotic plus HE Investigative Sites 2 (Spain) ~100 (US and EU) N Randomized 38 ~140 expected OCR-002 Dose 10g 20g, may be adjusted to 15 or 10g based on liver impairment to achieve comparable exposure levels Primary Objective change from baseline in plasma ammonia levels time to meaningful clinical improvement in HE symptoms |
| STOP-ALF Phase 2a NIH-Sponsored Study: OCR-002 IV in Acute Liver Failure ACUTE LIVER FAILURE PATIENTS: ULTRA ORPHAN POPULATION (2,000/YR) Often young, otherwise healthy Acetaminophen overdose and other acute liver shocks ~40-65% spontaneous recovery1,2 ~30% mortality1,2 ~8-30% require transplantation1,2 Goal: protect brain while liver recovers or is transplanted NIH SUPPORTED TRIAL Open label safety study All patients receive active drug up to 10g per 24 hours Two cohorts (24 patients) One with normal renal function One with compromised renal function 1 Hepatology Vol 42, Issue 6, pp 1364-1372, December 2005 2 Annals Internal Medicine 2002; 137(12):947-954 |
| OCR-002: Continuity of Care for HE Patients OCR-002 CANDIDATES Unmet Medical Need Optimal Formulation for Acute Patient Population Treatment of Hospitalized HE All IV ORAL Maintenance of Chronic HE Remission All Continuity of Care post IV Non-Antibiotic Alternative to rifaximin, or combined with rifaximin Standard of Care Objective Indication Commercial Rights Value |
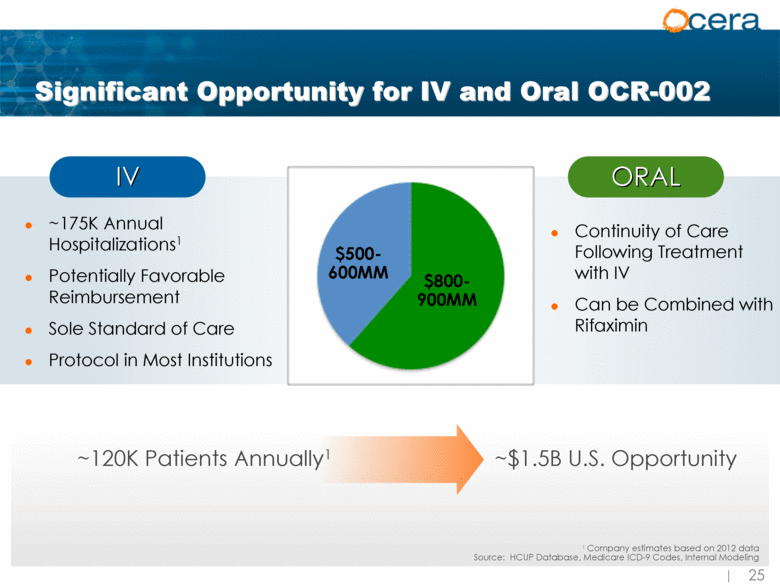
| Significant Opportunity for IV and Oral OCR-002 ~175K Annual Hospitalizations1 Potentially Favorable Reimbursement Sole Standard of Care Protocol in Most Institutions ORAL IV Continuity of Care Following Treatment with IV Can be Combined with Rifaximin 1 Company estimates based on 2012 data Source: HCUP Database, Medicare ICD-9 Codes, Internal Modeling ~120K Patients Annually1 $500-600MM $800-900MM ~$1.5B U.S. Opportunity |
| Strong Patent Coverage of OCR-002 2030 2025 2031 2025 Broad Composition Claims Granted/Allowed in Europe, Japan, China and Several Other Countries Specific Drug Substance Granted/Allowed in U.S., China, and Several Other Countries; Pending Worldwide Treatment of Hepatic Encephalopathy Granted in U.S., Europe, Japan, China and Several Other Countries Pending Worldwide PATENT EXPIRATION COMPOSITION OF MATTER USE CLAIMS METHODS OF MANUFACTURE 2030 Treatment of Portal Hypertension Granted/Allowed in Europe, China and Other Countries; Pending Worldwide |
| OCR-002 (IV) – STOP-HE Acute HE Initiated Phase 2b trial DMC recommendation from interim analysis (Q1-2015) Enrollment completion (YE-2015) OCR-002 (IV) – Externally Sponsored Phase 2a Readouts Cirrhosis and GI bleed (Spain) STOP-ALF Acute Liver Failure (NIH) OCR-002 (Oral) Commenced formulation development Initiate Phase 1 trial (H2-2015) Executing Against our Clinical Objectives |
| Team with Deep Experience Linda S. Grais, MD, JD President & Chief Executive Officer Gaurav Aggarwal, MD Chief Business Officer Michael Byrnes, MBA Chief Financial Officer Rajiv Patni, MD Chief Development Officer |
| Building a Liver Disease Medicines Company Clinical-stage development for treatment of Hepatic Encephalopathy IV and oral formulations Strong patent coverage Differentiated Product Candidate: OCR-002 STOP-HE Phase 2b interim analysis Completion of Phase 2b enrollment Results of two investigator-sponsored Phase 2a studies Multiple Catalysts in 2015 Funds to support Ocera through upcoming milestones Strong Balance Sheet Ver 02.28.15 |