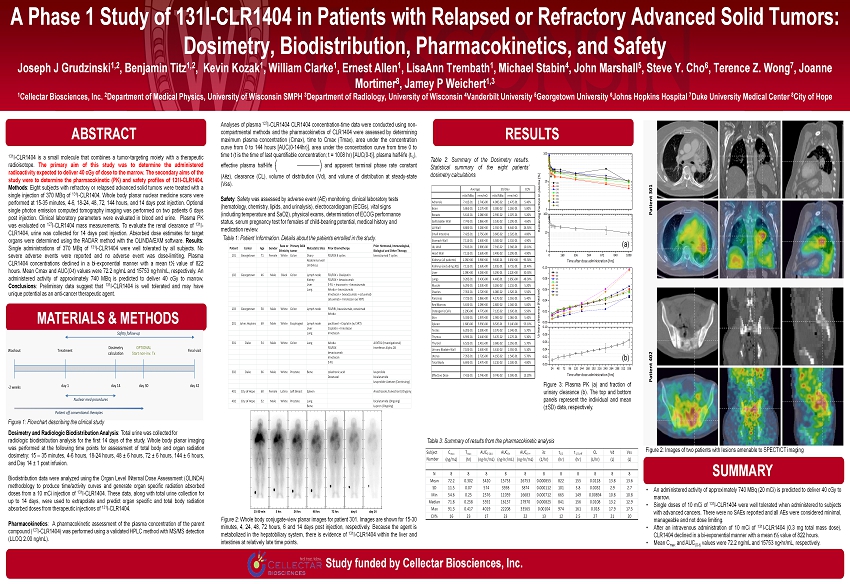
Analyses of plasma 127 I - CLR 1404 CLR 1404 concentration - time data were conducted using non - compartmental methods and the pharmacokinetics of CLR 1404 were assessed by determining maximum plasma concentration ( Cmax ), time to Cmax ( Tmax ), area under the concentration curve from 0 to 144 hours [AUC( 0 - 144 hr)], area under the concentration curve from time 0 to time t (t is the time of last quantifiable concentration ; t = 1008 hr ) [AUC( 0 - t)], plasma half - life (t ½ ), effective plasma half - life and apparent terminal phase rate constant ( λëz ), clearance (CL), volume of distribution ( Vd ), and volume of distribution at steady - state ( Vss ) . Safety : Safety was assessed by adverse event (AE) monitoring, clinical laboratory tests (hematology, chemistry, lipids, and urinalysis), electrocardiogram (ECGs), vital signs (including temperature and SaO 2 ), physical exams, determination of ECOG performance status, serum pregnancy test for females of child - bearing potential, medical history and medication review . A Phase 1 Study of 131I - CLR1404 in Patients with Relapsed or Refractory Advanced Solid Tumors: Dosimetry , Biodistribution , Pharmacokinetics, and Safety Joseph J Grudzinski 1,2 , Benjamin Titz 1,2 , Kevin Kozak 1 , William Clarke 1 , Ernest Allen 1 , LisaAnn Trembath 1 , Michael Stabin 4 , John Marshall 5 , Steve Y. Cho 6 , Terence Z. Wong 7 , Joanne Mortimer 8 , Jamey P Weichert 1,3 1 Cellectar Biosciences, Inc. 2 Department of Medical Physics, University of Wisconsin SMPH 3 Department of Radiology, University of Wisconsin 4 Vanderbilt University 5 Georgetown University 6 Johns Hopkins Hospital 7 Duke University Medical Center 8 City of Hope MATERIALS & METHODS RESULTS SUMMARY Study funded by Cellectar Biosciences, Inc. 131 I - CLR 1404 is a small molecule that combines a tumor - targeting moiety with a therapeutic radioisotope . The primary aim of this study was to determine the administered radioactivity expected to deliver 40 cGy of dose to the marrow . The secondary aims of the study were to determine the pharmacokinetic (PK) and safety profiles of 131 I - CLR 1404 . Methods : Eight subjects with refractory or relapsed advanced solid tumors were treated with a single injection of 370 MBq of 131 I - CLR 1404 . Whole body planar nuclear medicine scans were performed at 15 - 35 minutes, 4 - 6 , 18 - 24 , 48 , 72 , 144 hours, and 14 days post injection . Optional single photon emission computed tomography imaging was performed on two patients 6 days post injection . Clinical laboratory parameters were evaluated in blood and urine . Plasma PK was evaluated on 127 I - CLR 1404 mass measurements . To evaluate the renal clearance of 131 I - CLR 1404 , urine was collected for 14 days post injection . Absorbed dose estimates for target organs were determined using the RADAR method with the OLINDA/EXM software . Results : Single administrations of 370 MBq of 131 I - CLR 1404 were well tolerated by all subjects . No severe adverse events were reported and no adverse event was dose - limiting . Plasma CLR 1404 concentrations declined in a bi - exponential manner with a mean t½ value of 822 hours . Mean Cmax and AUC( 0 - t) values were 72 . 2 ng /mL and 15753 ng∙hr /mL, respectively . An administered activity of approximately 740 MBq is predicted to deliver 40 cGy to marrow . Conclusions : Preliminary data suggest that 131 I - CLR 1404 is well tolerated and may have unique potential as an anti - cancer therapeutic agent . ABSTRACT • An administered activity of approximately 740 MBq ( 20 mCi ) is predicted to deliver 40 cGy to marrow . • Single doses of 10 mCi of 131 I - CLR 1404 were well tolerated when administered to subjects with advanced cancers . There were no SAEs reported and all AEs were considered minimal, manageable and not dose limiting . • After an intravenous administration of 10 mCi of 131 I - CLR 1404 ( 0 . 3 mg total mass dose), CLR 1404 declined in a bi - exponential manner with a mean t½ value of 822 hours . • Mean C max and AUC ( 0 - t) values were 72 . 2 ng /mL and 15753 ng • hr /mL, respectively . Siegel R, Naishadham D, Jemal A. Cancer Statistics , 2013. CA Cancer J Clin. 2013;63(1):11 – 30. doi:10.3322/caac.21166. 2. LAG R, Melbert D, Krapcho M, et al. SEER Cancer Statistics Review, 1975 - 2005. 2008. Available at: http://seer.cancer.gov/csr /1975_2005/. 3. Pinchuk AN, Rampy MA, Longino MA, et al. Synthesis and structure - activity relationship effects on the tumor avidity of radio iodinated phospholipid ether analogues. J Med Chem. 2006;49(7):2155 – 2165. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16570911. 4. Deming D a, Leystra A a, Farhoud M, et al. mTOR inhibition elicits a dramatic response in PI3K - dependent colon cancers. PLoS One. 2013;8(4):e60709. doi:10.1371/journal.pone.0060709. 5. Zasadny KR, Longino MA, Fisher SJ, Counsell RE, Wahl RL. A Phospholipid Ether Agent for Tumor Imaging and Possible Therapy. J Nucl Med. 1999;40:39. 6. Weichert JP. Alkylphosphocholine Analogs for broad Spectrum Cancer Imaging and Therapy. Sci Transl Med. 2013. 7. Stabin MG, Siegel JA. Physical Models and Dose Factors for Use in Internal Dose Assessment. Heal Phys. 2003;85(3):294 – 310. A vailable at: http://journals.lww.com/health - physics/Fulltext/2003/09000/Physical_Models_and_Dose_Factors_for_Use_in.6.aspx. 8. Siegel JA, Thomas SR, Stubbs JB, et al. MIRD pamphlet no. 16: Techniques for quantitative radiopharmaceutical biodistributio n data acquisition and analysis for use in human radiation dose estimates. J Nucl Med. 1999;40(2):37S – 61S. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=1 0025848. 9. Barrett PH, Bell BM, Cobelli C, et al. SAAM II: Simulation, Analysis, and Modeling Software for tracer and pharmacokinetic s tudies. Metabolism. 1998;47(4):484 – 92. Available at: http://www.ncbi.nlm.nih.gov/pubmed/9550550. 10. Stabin MG, da Luz LC. Decay data for internal and external dose assessment. Heal Phys. 2002;83(4):471 – 475. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12240721. 11. Stabin MG, Sparks RB, Crowe E. OLINDA/EXM: the second - generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med. 2005;46(6):1023 – 1027. Available at: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15937315. 12. Jiang H, Cannon MJ, Banach M, et al. Quantification of CLR1401, a novel alkylphosphocholine anticancer agent, in rat plasma by hydrophilic interaction liquid chromatography - tandem mass spectrometric detection. J Chromatogr B Analyt Technol Biomed Life Sci. 2010;878(19):1513 – 8. doi:10.1016/j.jchromb.2010.04.002. Subject Number Cmax (ng/mL) Tmax (hr) AUC0 - 144 (ng hr/mL) AUC0 - t (ng hr/mL) AUC0 - ∞ (ng hr/mL) λ z (1/hr) t1/2 (hr) t1/2,eff (hr) CL (L/hr) Vd (L) Vss (L) 101 66.9 0.417 3848 22208 33565 0.000829 836 156 0.00894 10.8 11.0 102 54.6 0.383 2576 11059 16683 0.001 691 150 0.018 17.9 17.5 103 91.3 0.250 4019 16185 26452 0.00104 665 149 0.0113 10.9 10.8 201 73.5 0.250 3219 14255 25803 0.000791 877 158 0.0116 14.7 14.5 301 63.5 0.350 2599 11560 19851 0.000848 818 156 0.0151 17.8 17.5 302 70.1 0.250 3471 16129 29489 0.000793 874 158 0.0102 12.8 12.7 401 84 0.267 3916 18285 31216 0.00082 846 157 0.00961 11.7 11.3 402 73.8 0.250 3713 16347 30962 0.000712 974 161 0.00969 13.6 13.2 N 8 8 8 8 8 8 8 8 8 8 8 Mean 72.2 0.302 3420 15753 26753 0.000855 822 155 0.0118 13.8 13.6 SD 11.5 0.07 574 3598 5874 0.000112 101 3.8 0.0032 2.9 2.7 Min 54.6 0.25 2576 11059 16683 0.000712 665 149 0.00894 10.8 10.8 Median 71.8 0.258 3592 16157 27970 0.000825 841 156 0.0108 13.2 12.9 Max 91.3 0.417 4019 22208 33565 0.00104 974 161 0.018 17.9 17.5 CV% 16 23 17 23 22 13 12 2.5 27 21 20 T max AUC 0-∞ λz t 1/2 t 1/2,eff CL Vd Vss (hr) (ng hr/mL) (1/hr) (hr) (hr) (L/hr) (L) (L) N 8 8 8 8 8 8 8 8 8 8 8 Mean 72.2 0.302 3420 15753 26753 0.000855 822 155 0.0118 13.8 13.6 SD 11.5 0.07 574 3598 5874 0.000112 101 3.8 0.0032 2.9 2.7 Min 54.6 0.25 2576 11059 16683 0.000712 665 149 0.00894 10.8 10.8 Median 71.8 0.258 3592 16157 27970 0.000825 841 156 0.0108 13.2 12.9 Max 91.3 0.417 4019 22208 33565 0.00104 974 161 0.018 17.9 17.5 CV% 16 23 17 23 22 13 12 2.5 27 21 20 Subject Number C max (ng/mL) AUC 0-144 (ng hr/mL) AUC 0-t (ng hr/mL) Table 3 : Summary of results from the pharmacokinetic analysis Figure 2: Images of two patients with lesions amenable to SPECT/CT imaging Figure 1: Flowchart describing the clinical study Table 2 : Summary of the Dosimetry results . Statistical summary of the eight patients’ dosimetry calculations COV mSv/MBq rem/mCi mSv/MBq rem/mCi Adrenals 7.41E-01 2.74E+00 4.00E-02 1.47E-01 5.40% Brain 5.86E-01 2.17E+00 3.38E-02 1.26E-01 5.80% Breasts 5.61E-01 2.08E+00 2.93E-02 1.07E-01 5.20% Gallbladder Wall 7.74E-01 2.86E+00 3.53E-02 1.29E-01 4.60% LLI Wall 8.88E-01 3.28E+00 2.35E-01 8.64E-01 26.50% Small Intestine 7.42E-01 2.75E+00 3.60E-02 1.32E-01 4.80% Stomach Wall 7.11E-01 2.63E+00 3.50E-02 1.31E-01 4.90% ULI Wall 7.81E-01 2.89E+00 7.91E-02 2.94E-01 10.10% Heart Wall 7.11E-01 2.63E+00 3.49E-02 1.29E-01 4.90% Kidneys (all patients) 1.05E+00 3.90E+00 9.83E-01 3.65E+00 93.30% Kidneys (excluding 301) 7.11E-01 2.63E+00 1.81E-01 6.71E-01 25.40% Liver 1.09E+00 4.03E+00 3.29E-01 1.22E+00 30.30% Lungs 9.28E-01 3.43E+00 4.48E-01 1.65E+00 48.30% Muscle 6.29E-01 2.33E+00 3.26E-02 1.21E-01 5.20% Ovaries 7.35E-01 2.72E+00 4.08E-02 1.52E-01 5.50% Pancreas 7.72E-01 2.86E+00 4.17E-02 1.55E-01 5.40% Red Marrow 5.63E-01 2.09E+00 2.80E-02 1.04E-01 5.00% Osteogenic Cells 1.29E+00 4.77E+00 7.12E-02 2.63E-01 5.50% Skin 5.33E-01 1.97E+00 2.90E-02 1.06E-01 5.40% Spleen 1.60E+00 5.93E+00 8.52E-01 3.14E+00 53.10% Testes 6.20E-01 2.30E+00 3.57E-02 1.34E-01 5.70% Thymus 6.59E-01 2.44E+00 3.47E-02 1.27E-01 5.30% Thyroid 6.52E-01 2.41E+00 3.68E-02 1.35E-01 5.70% Urinary Bladder Wall 7.10E-01 2.63E+00 3.61E-02 1.35E-01 5.10% Uterus 7.35E-01 2.72E+00 4.15E-02 1.54E-01 5.70% Total Body 6.69E-01 2.47E+00 3.21E-02 1.18E-01 4.80% Effective Dose 7.41E-01 2.74E+00 9.74E-02 3.59E-01 13.20% Average Std Dev Figure 3 : Plasma PK (a) and fraction of urinary clearance (b) . The top and bottom panels represent the individual and mean ( ± SD) data, respectively . Dosimetry and Radiologic Biodistribution Analysis : Total urine was collected for radiologic biodistribution analysis for the first 14 days of the study . Whole body planar imaging was performed at the following time points for assessment of total body and organ radiation dosimetry : 15 – 35 minutes, 4 - 6 hours, 18 - 24 hours, 48 ± 6 hours, 72 ± 6 hours, 144 ± 6 hours, and Day 14 ± 1 post infusion . Biodistribution data were analyzed using the Organ Level INternal Dose Assessment (OLINDA) methodology to produce time/activity curves and generate organ specific radiation absorbed doses from a 10 mCi injection of 131 I - CLR 1404 . These data, along with total urine collection for up to 14 days, were used to extrapolate and predict organ specific and total body radiation absorbed doses from therapeutic injections of 131 I - CLR 1404 . Pharmacokinetics : A pharmacokinetic assessment of the plasma concentration of the parent compound ( 127 I - CLR 1404 ) was performed using a validated HPLC method with MS/MS detection (LLOQ 2 . 00 ng /mL) . Table 1 : Patient Information . Details about the patients enrolled in the study . Figure 2 : Whole body conjugate - view planar images for patient 301 . Images are shown for 15 - 30 minutes, 4 , 24 , 48 , 72 hours, 6 and 14 days post injection, respectively . Because the agent is metabolized in the hepatobiliary system, there is evidence of 131 I - CLR 1404 within the liver and intestines at relatively late time points . (a) (b) Patient Center Age Gender Race or Ethnicity Primary Solid tumor Metastatic Sites Prior Chemotherpy Prior Hormonal, Immunological, Biological and Other Therapy 101 Georgetown 71 Female White Colon Ovary FOLFOX 8 cycles bevacizumab 7 cycles Abdominal wall Umbilicus 102 Georgetown 46 Male Black Colon Lymph node FOLFOX + Oxalipatin Kidney FOLFOX + bevacizumab Liver 5-FU + leucovorin + bevacizumab Lung Xeloda + bevacizumab Irinotecan + bevacizumab + cetuximab cetuximab + Irinotecan (w/ XRT) 103 Georgetown 58 Male White Colon Lymph node FOLFIRI, bevacizumab, cetuximab Xeloda 201 Johns Hopkins 69 Male White Esophageal Lymph node paclitaxel + Cisplatin (w/ XRT) Liver Cisplatin + Irinotecan Lung Irinotecan 301 Duke 54 Male White Colon Lung Xeloda AVX701 (Investigational) FOLFOX Interferon Alpha 2B bevacizumab Irinotecan 5-FU 302 Duke 66 Male White Prostate Bone zoledronic acid leuprolide Docetaxel bicalutamide Leuprolide Acetate (Continuing) 401 City of Hope 60 Female Latino Left Breast Spleen Anastrozole, fulvestrant (Ongoing) 402 City of Hope 52 Male White Prostate Lung bicalutamide (Ongoing) Bone Lupron (Ongoing)