Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
RCKT similar filings
- 10 Mar 23 Regulation FD Disclosure
- 27 Feb 23 Rocket Pharmaceuticals Reports Fourth Quarter and Full Year 2022 Financial and Operational Results
- 7 Feb 23 Rocket Pharmaceuticals Receives FDA Regenerative Medicine Advanced Therapy (RMAT) Designation for RP-A501 Gene Therapy for Danon Disease
- 9 Jan 23 Other Events
- 22 Dec 22 Rocket Pharmaceuticals Provides Update on Anticipated Registration Path for RP-A501 in Danon Disease
- 1 Dec 22 Rocket Pharmaceuticals Completes Acquisition of Renovacor
- 3 Nov 22 Results of Operations and Financial Condition
Filing view
External links
Exhibit 99.1

INVESTOR DECK INVESTOR DECK INVESTOR DECK 41st Annual J.P. Morgan Healthcare Conference Company Presentation Gaurav Shah, MD Chief Executive Officer January 9, 2023

DISCLAIMER Various statements in this presentation concerning Rocket's future expectations, plans and prospects, including without limitation, Rocket's expectations regarding its guidance for 2023 in light of COVID-19, the safety, effectiveness and timing of product candidates that Rocket may develop to treat Fanconi Anemia (FA), Leukocyte Adhesion Deficiency-I (LAD-I), Pyruvate Kinase Deficiency (PKD), Danon Disease (DD), and other diseases, and the safety, effectiveness and timing of related pre-clinical studies and clinical trials and related data readouts, may constitute forward-looking statements for the purposes of the safe harbor provisions under the Private Securities Litigation Reform Act of 1995 and other federal securities laws and are subject to substantial risks, uncertainties and assumptions. You should not place reliance on these forward-looking statements, which often include words such as "believe," "expect," "anticipate," "intend," "plan," "will give," "estimate," "seek," "will," "may," "suggest" or similar terms, variations of such terms or the negative of those terms. Although Rocket believes that the expectations reflected in the forward-looking statements are reasonable, Rocket cannot guarantee such outcomes. Actual results may differ materially from those indicated by these forward-looking statements as a result of various important factors, including, without limitation, Rocket's ability to monitor the impact of COVID-19 on its business operations and take steps to ensure the safety of patients, families and employees, the interest from patients and families for participation in each of Rocket’s ongoing trials, our expectations regarding when clinical trial sites will resume normal business operations, our expectations regarding the delays and impact of COVID-19 on clinical sites, patient enrollment, trial timelines and data readouts, our expectations regarding our drug supply for our ongoing and anticipated trials, actions of regulatory agencies, which may affect the initiation, timing and progress of pre-clinical studies and clinical trials of its product candidates, Rocket's dependence on third parties for development, manufacture, marketing, sales and distribution of product candidates, the outcome of litigation, and unexpected expenditures, as well as those risks more fully discussed in the section entitled "Risk Factors" in Rocket’s Annual Report on Form 10-K for the year ended December 31, 2021, filed February 28, 2022 with the SEC and subsequent filings with the SEC including our Quarterly Reports on Form 10-Q. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and Rocket undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.

v To develop first-in-class and best-in-class curative gene therapies for patients with devastating diseases Generosity Trust Elevate Values Mission Curiosity ABOUT ROCKET PHARMACEUTICALS Vision: Seeking Gene Therapy Cures
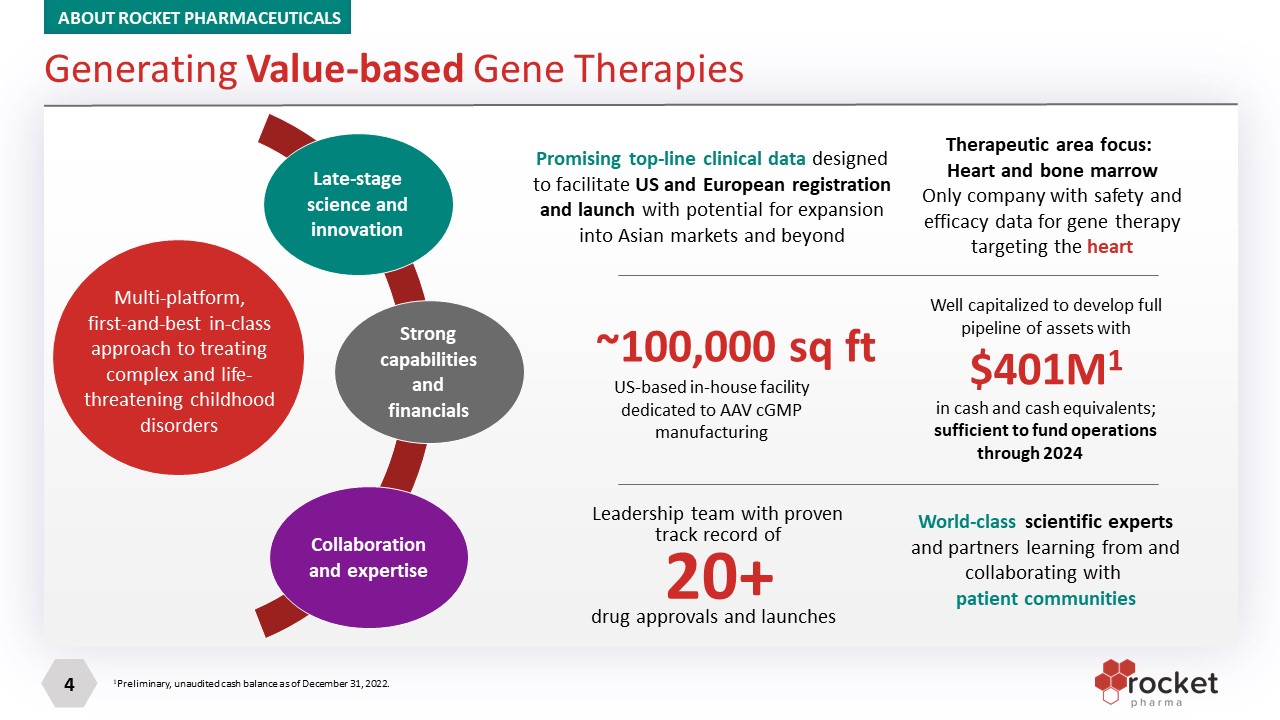
Multi-platform, first-and-best in-class approach to treating complex and life-threatening childhood disorders ???? Late-stage science and innovation Strong capabilities and financials Collaboration and expertise Promising top-line clinical data designed to facilitate US and European registration and launch with potential for expansion into Asian markets and beyond Therapeutic area focus: Heart and bone marrow Only company with safety and efficacy data for gene therapy targeting the heart US-based in-house facility dedicated to AAV cGMP manufacturing ~100,000 sq ft Well capitalized to develop full pipeline of assets with $401M1 in cash and cash equivalents; sufficient to fund operations through 2024 Leadership team with proven track record of drug approvals and launches 20+ World-class scientific experts and partners learning from and collaborating with patient communities Generating Value-based Gene Therapies ABOUT ROCKET PHARMACEUTICALS 1Preliminary, unaudited cash balance as of December 31, 2022.

On-target MOA; clear endpoints Sizeable market to maximize patient impact First-, best- and/or only-in-class Criteria used to select programs 6+ programs with 2 programs fast approaching regulatory filing and launch AAV, adeno-associated virus; ATMP, advanced therapy medicinal product; BLA, Biologics License Application; LV, lentiviral vector; MAA, Marketing Authorisation Application; MOA, mechanism of action; PRIME, PRIority MEdicines; RMAT, regenerative medicine advanced therapy. PKP2: plakophilin 2; ACM: Arrhythmogenic Cardiomyopathy; BAG3: BLC2-associated athanogene 3 DCM: Dilated Cardiomyopathy Data on file. Rocket Pharmaceuticals. 2023. Wave 2 programs Fast Track, Orphan Drug (US/EU) DISCOVERY PRECLINICAL PHASE 1 PHASE 2 (Pivotal) Submission and Approval DESIGNATIONS AAV RP-A501 Danon Disease LV RP-L102 Fanconi Anemia LV RP-L201 Leukocyte Adhesion Deficiency-I LV RP-L301 Pyruvate Kinase Deficiency Multiple Undisclosed Candidates AAV BAG3-DCM THERAPEUTIC AREA AAV RP-A601 PKP2-ACM CARDIOVASCULAR HEMATOLOGY RMAT, ATMP, Fast Track, Rare Pediatric, Orphan Drug (US/EU), PRIME Fast Track, Orphan Drug (US), Rare Pediatric Designation RMAT, ATMP, Fast Track, Rare Pediatric, Orphan Drug (US/EU), PRIME Strong Science, Carefully-selected Assets and Smart Execution: 6 Disclosed programs with compelling clinical and/or pre-clinical proof of concept ABOUT ROCKET PHARMACEUTICALS

IN VIVO platform EX VIVO platform RP-A501: Danon Disease RP-A601: PKP2-ACM AAV BAG3-DCM RP-L102: Fanconi Anemia RP-L201: Leukocyte Adhesion Deficiency-I RP-L301: Pyruvate Kinase Deficiency AAV, adeno-associated virus; HSC, hematopoietic stem cell; LV, lentiviral vector. Data on file. Rocket Pharmaceuticals. 2023 Laboratory- produced AAV Direct intravenous injection Therapeutic AAV Remove cells and isolate patient HSCs Infusion of modified HSCs Therapeutic LV Laboratory-produced LV Gene-modified HSCs All Rocket therapies transfer full (non-truncated) coding sequence to target tissue ABOUT ROCKET PHARMACEUTICALS Rocket Offers Multi-platform Gene Therapy Expertise

Looking Forward to a Catalyst-Rich 2023 Q1 Completed 2 In-house cGMP Danon Batches 2023 2024 cGMP, current Good Manufacturing Processes; FA, Fanconi anemia; H1, first half of the year; 2H, second half of the year; LAD-I, leukocyte adhesion deficiency-I; PKD, pyruvate kinase deficiency; Q1, first quarter of the yearQ2, second quarter of the year; Q3, third quarter of the year; Q4, fourth quarter of the year. Data on file. Rocket Pharmaceuticals. 2023. BAG3 IND Filing FA (C & G) IND Submission Non-Genotoxic Conditioning for LV Additional Wave 2 Assets Disclosed Q2 Q3 Q4 Planned Danon Phase 2 Study Initiation LAD-I Product Filing PKP2-ACM IND Filing Danon EU IMPD Filing FA Product Filing PKD Phase 2 Pivotal Study Initiation Danon Female Study Initiation LAD-I Moderate Study Initiation ABOUT ROCKET PHARMACEUTICALS Transition from Clinical to Commercial Stage

Standard of care: Heart transplant (HTx) Limitations: Considerable morbidity and mortality Only ~20% of patients receive HTx Not curative of extracardiac disease Therapeutic Challenges X-linked, dominant, monogenic disease Loss-of-function mutations in LAMP2 Disease Etiology Impaired autophagy Prominent autophagic vacuoles Myocardial disarray Clinical Manifestations Addressable Market – US and EU Prevalence of 15,000 to 30,000 individuals Annual incidence of 800 to 1,200 individuals Severe cardiomyopathy Mortality secondary to heart failure or arrhythmia Males: Aggressive disease course, median overall survival: 19 years Females: Delayed median presentation (~20 years later) due to additional X chromosome, highly morbid and fatal disorder Other clinical manifestations Skeletal myopathy CNS manifestations Ophthalmologic manifestations Danon Disease (DD): Serious Condition with Unmet Medical Need RP-A501: Danon Disease CNS, central nervous system; LAMP-2B, lysosome-associated membrane protein 2B. Boucek D, Jirikowic J, Taylor M. Natural history of Danon disease. Genet Med. 2011;13(6):563-568. Brambatti M, Caspi O, Maolo A, et al. Danon disease: Gender differences in presentation and outcomes. Int J Cardiol. 2019;286:92-98.

Non-randomized open label study in male DD patients PRIMARY OUTCOMES 6 to 36 months **Enrollment Complete** Data Reporting Details Pre-dose (baseline) value defined as the mean values from all visits prior to infusion Core lab data presented for echocardiographic parameters, cardiac serologies and cardiac histology Phase 1 Study: Treatment Completed RP-A501: Danon Disease Pediatric 8 to 14 years n=2 at CHOP Adults (and Adolescents) >15 years n=5 at UCSD Single intravenous dose of RP-A501 (AAV9.LAMP2B) delivering full coding sequence of the LAMP-2B gene CHOP, Children’s Hospital of Philadelphia; DD, Danon disease; LAMP-2B, lysosome-associated membrane protein 2B; UCSD, University of California San Diego. *No further enrollment at this dose. †Due to advanced heart failure at the time of dosing (LVEF <40%), patient 1007 received a heart transplant 5 months following infusion of RP-A501. Patient is currently stable Cohort Patient ID Age at infusion Time of follow-up (months) Low dose (6.7x1013 GC/kg) Pediatric 1008 12.3 12 1009 11.7 6 Low dose (6.7x1013 GC/kg) Adult and olderadolescent 1001 17.4 36 1002 20.3 36 1005 18.3 30 High dose* (1.1x1014 GC/kg) Adult and olderadolescent 1006 21.1 24 1007 20.7 N/A† Early and long-term safety Target tissue transduction and LAMP2B protein expression Improved myocardial histology Clinical improvement or stabilization

RP-A501: Danon Disease ADULT COHORT PEDIATRIC COHORT (Low dose) Revised Immunomodulatory Protocol: More rigorous daily monitoring of labs in initial days following infusion with independent clinical review team Reduced steroid dose with earlier taper Administration of sirolimus and rituximab Low Dose No SAEs related to drug product: 2 steroid related SAEs (myopathy) High Dose One instance of reversible TMA; led to enhanced RMP One instance of steroid myopathy Platelets remained within normal range No reported skeletal myopathy or late transaminitis with initial steroid dose reduction and more rapid taper, and introduction of sirolimus Minimal complement activation No complement-related clinical or laboratory AEs All AEs were transient and reversible No treatment-related SAEs AE, adverse event; SAE, serious AE; TMA, thrombotic microangiopathy. Data cut-off September 27, 2022, with source data verification through July 11, 2022. RP-A501 Demonstrates Favorable Safety Profile With Enhanced Immunomodulation Protocol All SAEs observed within initial 2-4 months following dosing; reversible with supportive care Both high and low-doses continue to be well tolerated at 2-3 years post treatment No additional SAEs observed following initial 2-4 months No RP-A501–related SAEsAll AEs were transient and reversible, with 8 and 13 months of follow up in 1008 and 1009, respectively
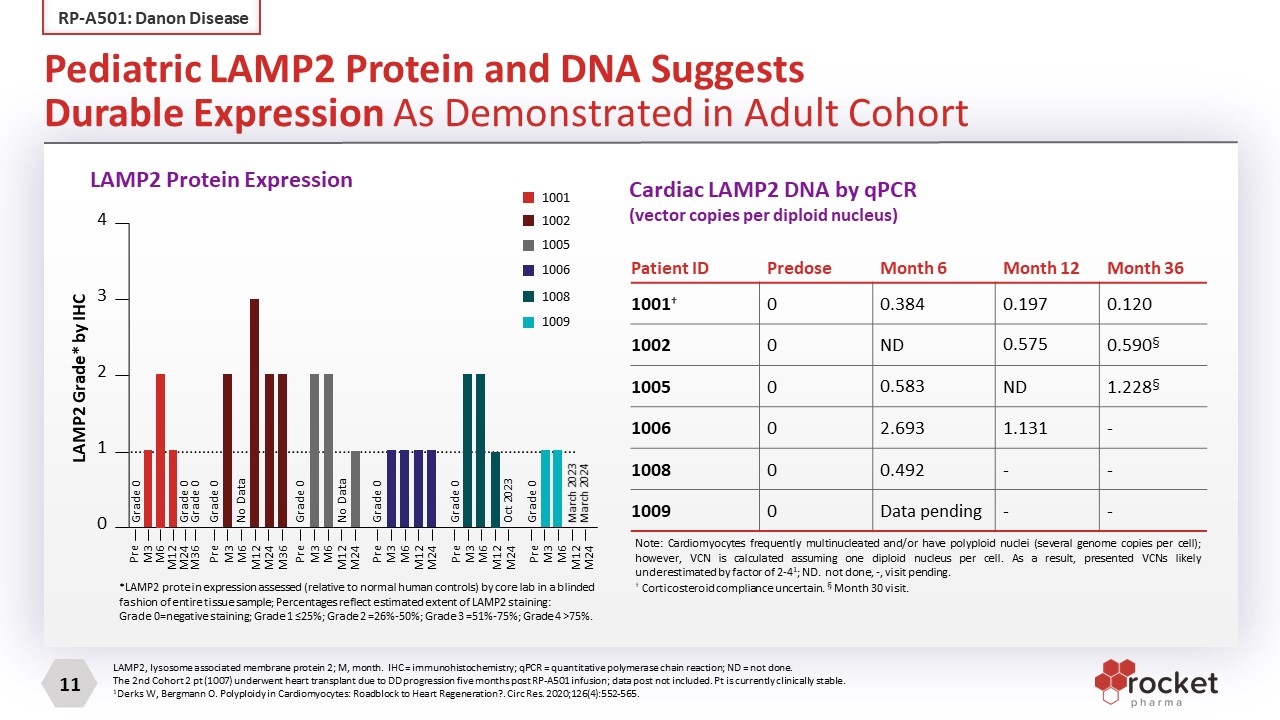
Patient ID Predose Month 6 Month 12 Month 36 1001† 0 0.384 0.197 0.120 1002 0 ND 0.575 0.590§ 1005 0 0.583 ND 1.228§ 1006 0 2.693 1.131 - 1008 0 0.492 - - 1009 0 Data pending - - LAMP2, lysosome associated membrane protein 2; M, month. IHC = immunohistochemistry; qPCR = quantitative polymerase chain reaction; ND = not done. The 2nd Cohort 2 pt (1007) underwent heart transplant due to DD progression five months post RP-A501 infusion; data post not included. Pt is currently clinically stable. 1Derks W, Bergmann O. Polyploidy in Cardiomyocytes: Roadblock to Heart Regeneration?. Circ Res. 2020;126(4):552-565. Note: Cardiomyocytes frequently multinucleated and/or have polyploid nuclei (several genome copies per cell); however, VCN is calculated assuming one diploid nucleus per cell. As a result, presented VCNs likely underestimated by factor of 2-41; ND. not done, -, visit pending. † Corticosteroid compliance uncertain. § Month 30 visit. Pediatric LAMP2 Protein and DNA Suggests Durable Expression As Demonstrated in Adult Cohort RP-A501: Danon Disease LAMP2 Protein Expression (VENDOR) Cardiac LAMP2 DNA by qPCR (vector copies per diploid nucleus) *LAMP2 protein expression assessed (relative to normal human controls) by core lab in a blinded fashion of entire tissue sample; Percentages reflect estimated extent of LAMP2 staining: Grade 0=negative staining; Grade 1 ≤25%; Grade 2 =26%-50%; Grade 3 =51%-75%; Grade 4 >75%. 1001 1002 1005 1006 1008 1009 4 3 2 1 0 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 Pre M3 M6 M12 M24 M36 Grade 0 Grade 0 Grade 0 Grade 0 No Data Grade 0 No Data Grade 0 Grade 0 Oct 2023 Grade 0 March 2023 March 2024 LAMP2 Grade* by IHC LAMP2 Protein Expression M36

Cohort Patient ID Most recent visit (months) Δ hsTnI Δ BNP Δ LV mass Δ LV max wall thickness Δ NYHA class Δ KCCQ score Low dose pediatric 1008 12 ↓86% ↓83% ↓29%1 ↓15%1 II -> I +32.3 1009 6 ↓90% ↓62% ↓21% ↑3% II -> I +26 Low dose adult/ adolescent 1001 36 ↓98% ↑8% ↓32% ↓9% II -> II +5.3 1002 36 ↓96% ↓94% ↓48% ↓40% II -> I2 +17.8 1005 30 ↓46% ↑6% ↓14% ↓27% II -> I +8.33 High dose adult/ adolescent 1006 24 ↓63% ↓69% ↓27% ↓15% II -> I +3.1 BNP, brain natriuretic peptide; DD, Danon disease; hsTnI, high-sensitivity troponin I; KCCQ, Kansas City Cardiomyopathy Questionnaire; LAMP2, lysosome-associated membrane protein 2; LV, left ventricle; NYHA, New York Heart Association. Does not include pt 1007 in Ph1 trial who had advanced HF with EF<40% at enrollment and received HTx 5M following tx due to pre-existing advanced HF. Patient is currently stable. 11008 echocardiographic parameters are M9 visit (M12 pending). 21002 NYHA class depicted for M30 visit (M36 pending). 31005 KCCQ score depicted for M24 visit (M30 pending). Darker Green = improved; Lighter Green = minimal change (stabilization) Improvement or Stabilization Observed Across Key Biomarker, Echo Findings and Functional Measures in Phase 1 RP-A501 Study RP-A501: Danon Disease

Note: Graphs do not depict serologic values during initial months after therapy; BL, baseline; M, month. Does not include pt 1007 in Ph1 trial who had advanced HF with EF<40% at enrollment and received HTx 5M following tx due to pre-existing advanced HF. Patient is currently stable. Indicates normal range (>100pg/mL BNP; >0.04ng/mL Troponin-I) Improvement or Stabilization Observed Across Key Cardiac Biomarkers RP-A501: Danon Disease
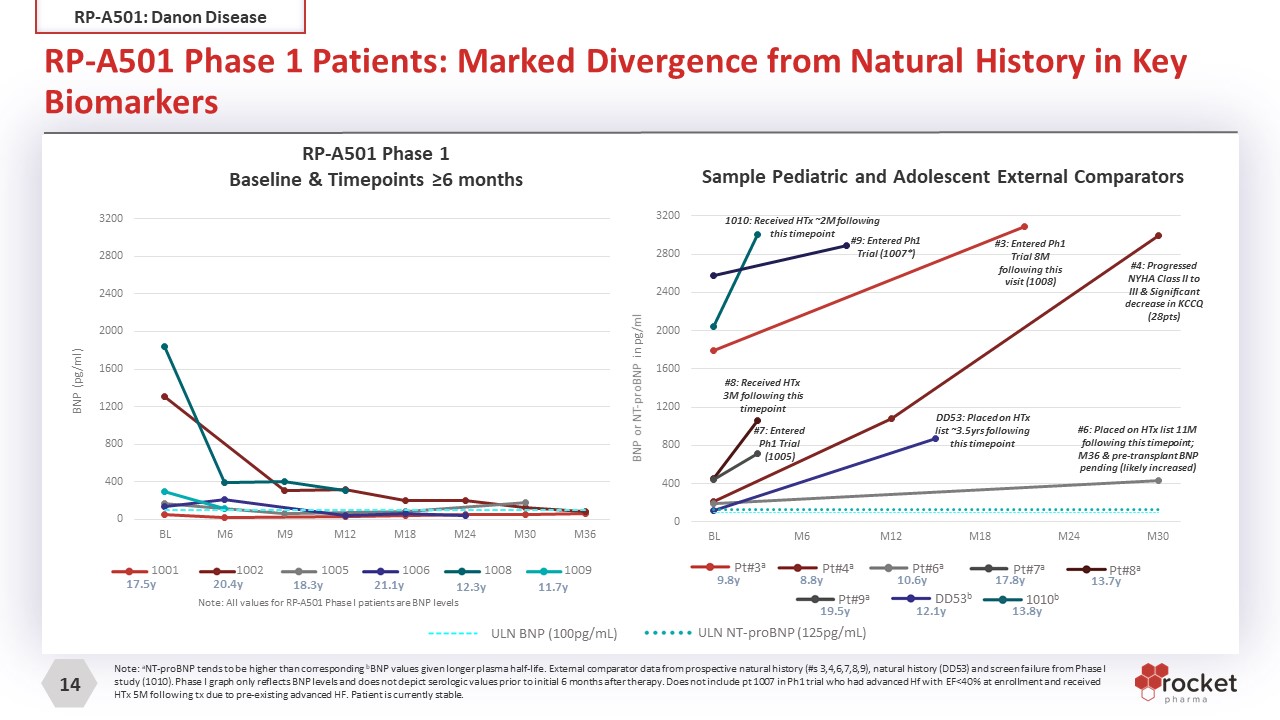
RP-A501 Phase 1 Patients: Marked Divergence from Natural History in Key Biomarkers RP-A501: Danon Disease Note: aNT-proBNP tends to be higher than corresponding bBNP values given longer plasma half-life. External comparator data from prospective natural history (#s 3,4,6,7,8,9), natural history (DD53) and screen failure from Phase I study (1010). Phase I graph only reflects BNP levels and does not depict serologic values prior to initial 6 months after therapy. Does not include pt 1007 in Ph1 trial who had advanced Hf with EF<40% at enrollment and received HTx 5M following tx due to pre-existing advanced HF. Patient is currently stable. RP-A501 Phase 1Baseline & Timepoints ≥6 months ULN NT-proBNP (125pg/mL) ULN BNP (100pg/mL) 17.5y 20.4y 18.3y 21.1y 12.3y 11.7y 1001 1002 1005 1006 1008 1009 Note: All values for RP-A501 Phase I patients are BNP levels Sample Pediatric and Adolescent External Comparators Pt#4a 8.8y Pt#6a 10.6y Pt#8a 13.7y DD53b 12.1y 1010b 13.8y Pt#7a 17.8y Pt#9a 19.5y Pt#3a 9.8y #8: Received HTx 3M following this timepoint #6: Placed on HTx list 11M following this timepoint; M36 & pre-transplant BNP pending (likely increased) DD53: Placed on HTx list ~3.5yrs following this timepoint #7: Entered Ph1 Trial (1005) #3: Entered Ph1 Trial 8M following this visit (1008) 1010: Received HTx ~2M following this timepoint #4: Progressed NYHA Class II to III & Significant decrease in KCCQ (28pts) #9: Entered Ph1 Trial (1007*)
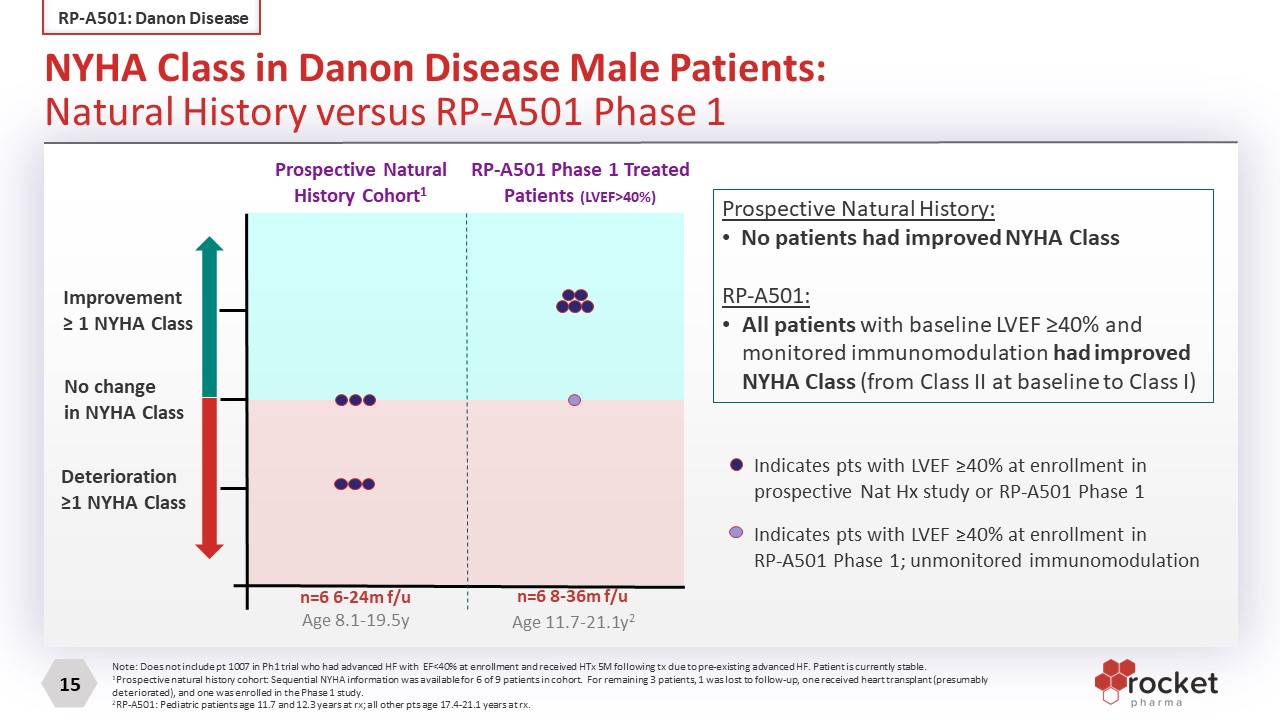
Improvement≥ 1 NYHA Class No changein NYHA Class Deterioration≥1 NYHA Class Prospective Natural History Cohort1 RP-A501 Phase 1 Treated Patients (LVEF>40%) n=6 8-36m f/u n=6 6-24m f/u Age 11.7-21.1y2 Age 8.1-19.5y Indicates pts with LVEF ≥40% at enrollment inprospective Nat Hx study or RP-A501 Phase 1 Indicates pts with LVEF ≥40% at enrollment inRP-A501 Phase 1; unmonitored immunomodulation Prospective Natural History: No patients had improved NYHA Class RP-A501: All patients with baseline LVEF ≥40% and monitored immunomodulation had improved NYHA Class (from Class II at baseline to Class I) Note: Does not include pt 1007 in Ph1 trial who had advanced HF with EF<40% at enrollment and received HTx 5M following tx due to pre-existing advanced HF. Patient is currently stable. 1Prospective natural history cohort: Sequential NYHA information was available for 6 of 9 patients in cohort. For remaining 3 patients, 1 was lost to follow-up, one received heart transplant (presumably deteriorated), and one was enrolled in the Phase 1 study. 2RP-A501: Pediatric patients age 11.7 and 12.3 years at rx; all other pts age 17.4-21.1 years at rx. NYHA Class in Danon Disease Male Patients: Natural History versus RP-A501 Phase 1 RP-A501: Danon Disease

Insights from Danon Disease Patients Treated on the Phase 1 Trial RP-A501: Danon Disease He went to overnight summer camp on his own for the first time and is no longer out of breath walking up stairs. -Pt 1008 Prior to therapy, he was afraid of dying and wanted a chance at life......After gene therapy, we see him smile more now, he bought his own place and working a couple of days a week, he has started to open up for meeting more friends in real life and has gotten a whole new peace of mind now ...he feels better, and he didn’t think that would ever happen –Pt 1006 He walked a 10K with his father following treatment. He is exercise training twice a week for an hour. -Pt 1009 He can walk upstairs without being short of breath or having to stop half-way. He doesn’t have chest pain or fast heart rates like he used to. Another amazing thing we have seen is about 4 months after his therapy trial he started working and stopped using his motorized scooter altogether. -Pt 1005

Based on End of Phase I Regulatory Discussion and ongoing dialogue with FDA. Elements in Discussion Key Agreements Reached with FDA 6.7x1013 GC/kg dose Single-arm, open-label study (randomization not appropriate) Support for use of natural history as external comparator information Potential for accelerated approval based on a composite biomarker-driven endpoint 6MWT, CPET are not appropriate endpoints in DD Specific components of composite endpoint including LAMP2 expression Trial duration and time to endpoint 2 patient run-in for pediatric enrollment (age 8-14 years) Confirmation pending submission of Phase II protocol and FDA review Will utilize revised Phase I eligibility criteria (i.e. LVEF >50%) Age 8 years and older Optimized immunomodulatory regimen used in Phase I pediatric cohort All drug product will be produced in-house at Cranbury, NJ facility Additional Study Elements Projected Pivotal Study Design RP-A501: Danon Disease

HTx, heart transplant; LVEF, LVEF, left ventricle ejection fraction. *Brambatti M, Caspi O, Maolo A, et al. Danon disease: Gender differences in presentation and outcomes. Int J Cardiol. 2019;286:92-98. RP-A501: Danon Disease US Retrospective Natural History 9 Males 1 Female 51 Males Males (goal 60) Females (goal 60) Initiated in 2019, expansion planned Initiated in 2006 Initiated in 2022 5 in follow-up 4 referred to RP-A501-0219 2 underwent HTx 3 enrolled 1 not enrolled (Phase 1 enrollment closed) 13 HTx Literature review (Brambatti 2019 publication*) 1 with low LVEF at enrollment progressed rapidly to end stage 7 died 9 HTx 1 died 67 Females Robust Ongoing Natural History Efforts to Support External Comparator Sample Prospective Natural History Study EU Retrospective Natural History Study

2 Successful Danon AAV cGMP batches produced in Q4 2022 Superior specifications to Phase I material; allow for full dosing with lower total viral particles, potentially further improving safety profile Productivity: ~3X increase in number of patient treatments per batch Product Quality: Significant increase in full versus empty viral particles Product Comparability: All attributes tested to date are comparable or improved Regulatory progress and production capacity can support pivotal study and commercialization FDA clearance on continued utilization of HEK-293 cell-based process through commercial FDA alignment on comparability approach Potency assay developed in-house Overall, in-house cGMP manufacturing delivers commercial-ready product with higher yield, improved quality, and likely enhances safety profile In-House Manufacturing to Support Danon Pivotal Study and Commercial Production RP-A501: Danon Disease

Total Lab Space: ~30,000 sq. ft. for process development, analytical development, MS&T and QC Manufacturing capability from small-scale to toxicology-scale material Streamlined tech transfer timeline for pipeline assets from plasmid selection to IND in <15 months Manufacturing expansion to add media and buffer production capability Incorporating fully automated in-house vial filler suite Anticipated 2X capacity increase in 2023 Enables rapid, robust and cost-efficient internal development capability for new and existing programs in addition to full-scale commercial manufacturing facility in Cranbury, NJ ~100,000 ft2 MS&T, manufacturing, science and technology. QC, quality control. R&D, research and development. IND, Investigational new drug. Cranbury R&D and Manufacturing Facility Overview RP-A501: Danon Disease

Moving toward pivotal global Phase 2 study FDA, Food and Drug Administration; PRV, priority review voucher. Data on file. Rocket Pharmaceuticals. 2023. RP-A501: Danon Disease Development Plan GLOBAL REGISTRATIONAL PHASE 2 STUDY Final Phase 2 Study Design and Endpoints Initiate Phase 2 Global Pivotal Study Activities Expanded natural history study Ongoing Activities Phase 1 treatment completed in males Orphan Drug, Rare Pediatric and Fast Track designations in the US (eligible for PRV) Completed 2 in-house cGMP batches End of Phase 1 Regulatory Meeting held with FDA Study Milestones

FA-A is an autosomal recessive disease caused by FANCA gene mutations FA proteins enable DNA repair FA-A accounts for 60% to 70% of FA cases Disease etiology BMF, bone marrow failure; FA, Fanconi Anemia; FA-A, FA, group A; FANC, FA complementation group; GvHD, graft-versus-host disease; HLA, human leukocyte antigen; HSCT, hematopoietic stem cell transplantation. Alter BP, Giri N, Savage SA, et al. Malignancies and survival patterns in the National Cancer Institute inherited bone marrow failure syndromes cohort study. Br J Haematol. 2010;150(2):179-188. Disorder of DNA repair characterized by: Progressive BMF; 80% of patients experience BMF within first decade of life Predisposition to hematologic malignancies and solid tumors Clinical manifestations Standard of care: Allogeneic HSCT Limitations: Significant toxicities, especially for patients who do not have an HLA-identical sibling donor (~80%) 100-day mortality GvHD Increased long-term cancer risk Therapeutic challenges Market Opportunity – US and EU Prevalence of 5,500 to 7,000 individuals Annual incidence of 200 to 275 individuals Fanconi Anemia (A, C, and G) RP-L102: Fanconi Anemia RP-L102 for Fanconi Anemia Complementation Group A (FA-A) Gene therapy approach: Selective advantage of corrected cells allows for ex-vivo LV therapy without conditioning; highly favorable benefit risk profile

Progressive increases in gene markings in PB and BM in 7 patients BM, bone marrow; CD34, cluster of differentiation 34; PB, peripheral blood; PBMC, PB mononuclear cells; VCN, vector copy number. Not shown: PB and BM VCN in Patient 2 (1002), who was withdrawn from the study at 18 months post–RP-L102 infusion. BM VCN not available at some stipulated time points due to insufficient sample to run assay. PBMC VCN BM VCN VCN in bone marrow mononuclear cells VCN in bone marrow CD34+ cells VCN in peripheral blood mononuclear cells RP-L102: Fanconi Anemia Progressively Increasing and Sustained Genetic Correction in 7 of 10 Patients ≥1 Year Post–RP-L102

For 5 patients, increased BM CFC MMC resistance ranging from 51% to 94% was observed at 18 to 24 months post–RP-L102 administration MMC resistance of >20% achieved at 2 consecutive timepoints ≥12 months for n=5 12m post gene therapy 20% resistance to MMC BM, bone marrow; CFC, colony-forming cells; MMC, mitomycin-C. Not shown: MMC resistance in Patient 2 (1002), who was withdrawn from the study at 18 months post–RP-L102 infusion. *One additional patient (Patient 1: 1001) was noted to have BM MMC resistance of 49% at ~40 months post–RP-L102 infusion. Sustained BM CFC MMC resistance observed in at least 6 patients* RP-L102: Fanconi Anemia Increasing Phenotypic Correction (MMC-resistance) over 1 to 3 Years Post–RP-L102

Additional life-cycle management activities: Expansion to FANC C and G Exploration of non-genotoxic conditioning and HSC expansion Rejection of null hypothesis with minimum of 5 patients with increased MMC resistance >10% at 2 timepoints between 12 and 36 months Initial comprehensive efficacy in 6/10 evaluable patients (≥12-month follow-up) No cytotoxic conditioning, only 1 transient RP-L102 related SAE (Grade 2) INITIAL EFFICACY AND HIGHLY FAVORABLE SAFETY PROFILE Update: CMC and clinical FDA discussions support BLA activities 2 patients to be treated with product from commercial cell processing site in preparation for US launch NEXT STEPS TOP-LINE DATA READOUT ACHIEVED Anticipated simultaneous BLA/MAA filings ATMP, advanced therapy medicinal product; BLA, Biologics License Application; CMC, chemistry, manufacturing, and controls; FANC, Fanconi Anemia complementation group; FDA, Food and Drug Administration; HSC, hematopoietic stem cell; MAA, Market Authorization Application; MMC, mitomycin-C; PRIME, PRIority MEdicines; PRV, priority review voucher; RMAT, Regenerative Medicine Advanced Therapy; SAE, severe adverse event. Data on file. Rocket Pharmaceuticals. 2023. REGULATORY DESIGNATIONS: RMAT, PRIME Orphan Drug designation in the US/EU Rare Pediatric Disease designation (eligible for PRV) Fast Track (US), ATMP Moving toward BLA/MAA filing Development Plan RP-L102: Fanconi Anemia

Patients suffer from recurrent infections; fatal in majority Severe LAD-I: Death prior to age 2 in 60% to 75% of patients, infrequent survival >5 years in absence of allogeneic HSCT Moderate LAD-I: Death prior to age 40 in >50% of patients, extensive morbidity with recurrent infections and inflammatory lesions Clinical manifestations ITGB2 gene mutations (21q22.3), encoding the beta-2-integrin, CD18; essential for leukocyte adhesion to endothelium CD18 absent or reduced on neutrophils Disease etiology Standard of care: Allogeneic HSCT Limitations: Donor availability Infections Frequent GvHD Graft failure Therapeutic challenges CD18, cluster of differentiation 18; GvHD, graft-versus-host disease; HSCT, hematopoietic stem cell transplant; ITGB2, integrin subunit beta 2; LAD-I, leukocyte adhesion deficiency-I. Almarza NE, Kasbekar S, Thrasher AJ, et al. Leukocyte adhesion deficiency-I: A comprehensive review of all published cases. J Allergy Clin Immunol Pract. 2018;6(4):1418-1420.e10. Market Opportunity – US and EU Prevalence of 800 to 1,000 individuals Annual incidence of 50 to 75 individuals RP-L201 for LAD-I: ITGB2 Gene Mutation RP-L201: LAD-I

CD18 Expression in PB Polymorphonuclear Cells (PMNs) At 3 to 24 months after infusion, 9/9 patients sustained stable CD18 expression (median: 56%) with no therapy-related serious adverse events *Dim/weak CD18 expression reported at baseline for Subject L201-003-1004 in ~63% of cells in conjunction with <2% CD11a/CD11b expression, likely indicating abnormal/unstable protein. LAD-I, Leukocyte Adhesion Deficiency-I; PB, peripheral blood; PMN, polymorphonuclear neutrophil. Data on file. Rocket Pharmaceuticals. 2022. Data Cut-Off: April 6, 2022; Preliminary interim results are presented from the ongoing clinical study. RP-L201: LAD-I *

Survival without allogeneic HSCT Primary outcomes ≥2 years of age AND ≥1-year post–RP-L201 infusion 100% overall survival Kaplan–Meier estimate Significant reduction in incidence of hospitalizations Post engraftment EOS, end of study; HSCT, hematopoietic stem cell transplantation; LAD-I, leukocyte adhesion deficiency-I. Data cut-off: March 9, 2022. Data on file. Rocket Pharmaceuticals. 2022. Significant Reduction in Hospitalizations and 100% Overall Survival RP-L201: LAD-I All patients have been able to stop prophylactic antibiotics

Life-cycle management Potential label expansion to include moderate LAD-I population Potential study initiation in 2023 ATMP, advanced therapy medicinal product; BLA, Biologics License Application; GvHD, graft-versus-host disease; LAD-I, leukocyte adhesion deficiency-I; MAA, Market Authorization Application; PRIME, PRIority MEdicines; PRV, priority review voucher; RMAT, Regenerative Medicine Advanced Therapy; SAE, severe adverse event. Data cut-off: March 9, 2022. Data on file. Rocket Pharmaceuticals. 2022. Survival for 9/9 patients, ≥2 years age and ≥1 year post-treatment No graft failure, GvHD No RP-L201 related SAEs Enrollment completed; 9/9 patients treated Efficacy observed in 9/9 patients with 3 to 24 months follow-up Efficacy is comprehensive, across all efficacy parameters including CD18 expression and survival ENROLLMENT AND INITIAL EFFICACY Progression to regulatory filing activities NEXT STEPS TOP-LINE DATA READOUT Q2 2022 Guiding Q2 2023 regulatory filing REGULATORY DESIGNATIONS: RMAT, PRIME Orphan Drug designation in the US/EU Rare Pediatric Disease designation (eligible for PRV) Fast Track (US), ATMP Moving toward product filing Development Plan RP-L201: LAD-I
Lifelong chronic hemolysis Other clinical manifestations: Anemia Jaundice Iron overload Clinical manifestations Standard of care: Chronic blood transfusions and splenectomy Limitations: Iron overload Extensive end-organ damage Splenectomy confers lifelong infection and thrombotic risk Therapeutic challenges Autosomal recessive inheritance Pyruvate kinase deficient RBCs cannot synthesize ATP, resulting in hemolytic anemia Disease etiology ATP, adenosine triphosphate; PKD, pyruvate kinase deficiency; PKLR, pyruvate kinase L/R; RBC, red blood cell. Zanella A, Fermo E, Bianchi P, Valentini G. Red cell pyruvate kinase deficiency: molecular and clinical aspects. Br J Haematol. 2005;130(1):11-25. Market Opportunity – US and EU Prevalence of 4,000 to 8,000 individuals Annual incidence of 75 to 125 individuals RP-L301 for PKD: PKLR Gene Mutation RP-L301: PKD
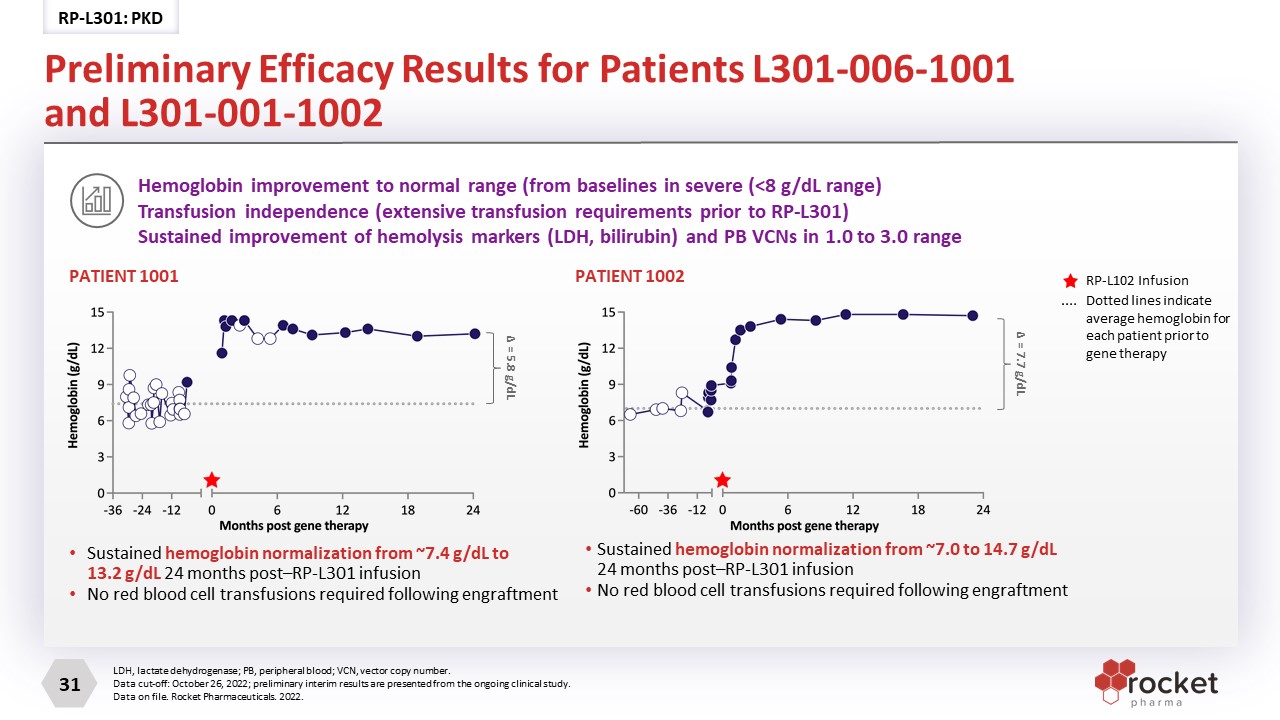
Sustained hemoglobin normalization from ~7.4 g/dL to 13.2 g/dL 24 months post–RP-L301 infusion No red blood cell transfusions required following engraftment PATIENT 1001 PATIENT 1002 Sustained hemoglobin normalization from ~7.0 to 14.7 g/dL 24 months post–RP-L301 infusion No red blood cell transfusions required following engraftment Hemoglobin improvement to normal range (from baselines in severe (<8 g/dL range)Transfusion independence (extensive transfusion requirements prior to RP-L301) Sustained improvement of hemolysis markers (LDH, bilirubin) and PB VCNs in 1.0 to 3.0 range LDH, lactate dehydrogenase; PB, peripheral blood; VCN, vector copy number. Data cut-off: October 26, 2022; preliminary interim results are presented from the ongoing clinical study. Data on file. Rocket Pharmaceuticals. 2022. Δ = 5.8 g/dL Δ = 7.7 g/dL RP-L102 Infusion Dotted lines indicate average hemoglobin for each patient prior to gene therapy Preliminary Efficacy Results for Patients L301-006-1001 and L301-001-1002 RP-L301: PKD

Development Plan Moving toward pivotal Phase 2 study Key endpoints selected Hemoglobin increase ↓ 50% in transfusions or transfusion independence Well-delineated natural history in recent PKD NHS publications Complete Phase 1 pediatric cohort dosing (N=2 to 3) End of Phase 1 regulatory meeting with FDA in 2023 Approve and launch RP-L301; seek regulatory approval in the US and EU PLAN FOR PHASE 2 AND LAUNCH Anticipated expansion study to pre-splenectomy patients Exploration of non-genotoxic conditioning LIFE-CYCLE MANAGEMENT FDA, Food and Drug Administration; NHS, National Health Service; PKD, pyruvate kinase deficiency; PRV, priority review voucher. Data on file. Rocket Pharmaceuticals. 2022. REGULATORY DESIGNATIONS Fast Track, Orphan Drug (US/EU), Rare Pediatric Disease (eligible for PRV) RP-L301: PKD

Future Therapies: Wave 2 (AAV) We continue to build our pipeline based on our core R&D strategy, identifying the “most impactful” indications for the most efficient development path. AAV, adeno-associated virus; MOA, mechanism of action; R&D, research and development. Data on file. Rocket Pharmaceuticals. 2022. On-target MOA; clear endpoints Sizeable market to maximize patient impact First-, best- and/or only-in-class Current Clinical Pipeline Focused R&D Strategy for Sustainable Innovation 3 therapeutic areas (CV, heme and undisclosed) FUTURE DIRECTIONS

We believe that a gene therapy approach is best positioned to restore the broad biological functions of BAG3 in the heart BAG3, BLC2-associated athanogene 3; BCL2, B-cell lymphoma 2. Knezevic T, Myers VD, Su F, et al. Adeno-associated Virus Serotype 9 - Driven Expression of BAG3 Improves Left Ventricular Function in Murine Hearts with Left Ventricular Dysfunction Secondary to a Myocardial Infarction. JACC Basic Transl Sci. 2016;1(7):647-656. Myers VD, Gerhard GS, McNamara DM, et al. Association of Variants in BAG3 With Cardiomyopathy Outcomes in African American Individuals JAMA Cardiol. 2018;3(10):929-938. 4 Cardiac contractility Enhances contractility by linking the β-adrenergic receptor and L-type Ca2+ channel Provides support for the sarcomere by linking actin myofibrils with the Z-disc Structural support Facilitates autophagy as a co- chaperone with heat shock proteins, recycling misfolded proteins Protein quality control Inhibits apoptosis (programmed cell death) through binding of BCL2 Anti-apoptosis BAG3 Dilated Cardiomyopathy BAG3 Regulates Critical Functions in Cardiomyocytes
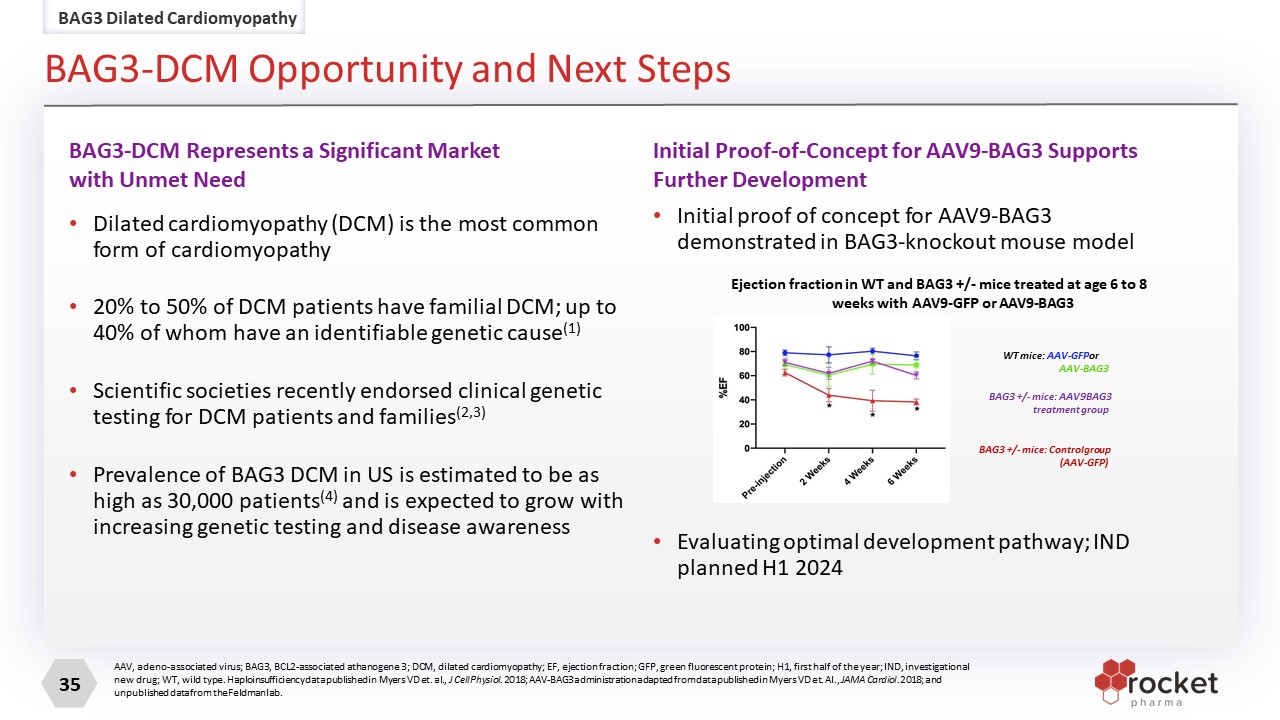
AAV, adeno-associated virus; BAG3, BCL2-associated athanogene 3; DCM, dilated cardiomyopathy; EF, ejection fraction; GFP, green fluorescent protein; H1, first half of the year; IND, investigational new drug; WT, wild type. Haploinsufficiency data published in Myers VD et. al., J Cell Physiol. 2018; AAV-BAG3 administration adapted from data published in Myers VD et. Al., JAMA Cardiol. 2018; and unpublished data from the Feldman lab. Dilated cardiomyopathy (DCM) is the most common form of cardiomyopathy 20% to 50% of DCM patients have familial DCM; up to 40% of whom have an identifiable genetic cause(1) Scientific societies recently endorsed clinical genetic testing for DCM patients and families(2,3) Prevalence of BAG3 DCM in US is estimated to be as high as 30,000 patients(4) and is expected to grow with increasing genetic testing and disease awareness Initial proof of concept for AAV9-BAG3 demonstrated in BAG3-knockout mouse model Evaluating optimal development pathway; IND planned H1 2024 BAG3-DCM Represents a Significant Market with Unmet Need Initial Proof-of-Concept for AAV9-BAG3 Supports Further Development WT mice: AAV-GFP or AAV-BAG3 BAG3 +/- mice: AAV9 BAG3 treatment group BAG3 +/- mice: Control group (AAV-GFP) Ejection fraction in WT and BAG3 +/- mice treated at age 6 to 8 weeks with AAV9-GFP or AAV9-BAG3 BAG3 Dilated Cardiomyopathy BAG3-DCM Opportunity and Next Steps

Project Pegasus (PKP2-ACM)

Current standard of care includes beta-blockers, anti-arrhythmic agents, and ablation Available treatments do not modify disease progression; no curative therapeutic options Therapeutic Challenges Autosomal dominant mutations in PKP2 gene, which encodes for Plakophilin-2, a component of the desmosome localized to cardiac intercalated discs Disease Etiology Clinical Manifestations Estimated Prevalence (US+EU): ~50,000 Advanced ACM Heart with fibrofatty replacement in right ventricle ACM: Diminished Myocardial PKP2 Normal patient PKP2-ACM patient N-Cadherin PKP2 PKP2-Arrhythmogenic Cardiomyopathy (ACM)*: A high-risk disease with no curative options RP-A601: PKP2-ACM Kaplan-Meier Incidence of ICD Firing Event free survival in ACM patients with ICDs based on VT inducibility on pre-ICD EPS study >50% of patients who were inducible on EP study had an ICD firing over 2 year follow-up Mean age at presentation: 35y (±18) 1 5-10% annual risk of sustained ventricular arrhythmias (VA), with higher risk in patients who present with disease symptoms (index patients)2-3 Lifetime VA risk approximately 100% in index patients 4 ICD placement in >80% of index pts 5 For pts with ICDs: 45-75% will have ICD firing (shock) over 3-5 years ≥50% 2 year incidence of firing in subgroups:• male; • EPS-induced VT; • history of VT; • ≥3 ECG leads with TWI; • >1000 PVC/24h 5-6 Biopsy figure adapted from: Asimaki et al. NEJM, 2009; Table adapted from Dalal et al. Circulation 2006. SOC: standard of care; CM: cardiomyopathy; HF: heart failure: HTx: heart transplantation; RV: Right ventricular; SD: Standard Deviation; VT: ventricular tachycardia; LBBB: left bundle branch block; ICD: implantable cardioverter defibrillator; RVEF: right ventricular ejection fraction; LV: left ventricle; SVA: sustained ventricular arrhythmia. * This cardiomyopathy initially manifests in the right ventricular free wall, so the disease was termed arrhythmogenic right ventricular dysplasia/cardiomyopathy (ARVD/ARVC). However, since left dominant and biventricular forms have also been observed, this has led more recently to the use of the term “ACM”. 1. Bhonsale. EHJ 2015; 36: 847-55. 2. Towbin JA. Heart Rhythm 2019;16(11). 3. Cadrin-Tourigny J. Eur Heart J 2022;43. 4. Groenweg. Circ Cardiovasc Genet 2015; 8: 437-46. 5. Calkins. Circ 2017; 136: 2068-82. 6. Orgeron. J Am Heart Assoc 2017: e006242. Electrical manifestations can precede structural abnormalities

Peters S, Trümmel M, Meyners W. Prevalence of right ventricular dysplasia-cardiomyopathy in a non-referral hospital. Int J Cardiol. 2004;97(3):499-501. McKenna WJ, Judge DP. Epidemiology of the inherited cardiomyopathies. Nat Rev Cardiol. 2021;18(1):22-36. 1 Data on file 2,572 ACM patients assessed from 13 publications an aggregated mean of 32.9% had PKP2 mutations1 Utilizing the conservative ACM prevalence (1:5000) and the 32.9% PKP2 mutation frequency in ACM PKP2-ACM Prevalence in the US and EU RP-A601: PKP2-ACM 1:1000 to 1:5000 ACM prevalence 32.9% PKP2 variants ~50,000 ACM-PKP2 US & EU Prevalence Peters 2004, McKenna 2021

Proof of Concept in Translationally Relevant Animal Model PROGRAM 4: RP-L301 for PKD RP-A601: PKP2-ACM Initial POC evaluated 4 AAV Vectors: Cardiac Functional & Structural Analyses Dose-related effects evaluated with 2 AAV vectors: Cardiac Functional & Structural Analyses Evaluated Survival, Functional, and Anatomic Benefit in ‘Arrest Progression’ Models Including delivery of AAV +7 or +14 Days after induction of PKP2 knockout and subsequent disease onset Completed RCKT Studies with Cardiomyocyte-specific PKP2 Knockout Mouse Model of ACM Analyses Include: Survival Echocardiography and ECG PKP2 expression (IF and WB) Cardiac pathology & fibrosis Vector DNA, transgene mRNA General safety including pathology Academic Partner: Mario Delmar, MD, PhD Patricia and Robert Martinsen Professor of Cardiology, Department of Medicine; Division of Cardiology, NYU Grossman School of Medicine Marina Cerrone, MD Research Associate Professor, Co-Director, Inherited Arrhythmia Clinic, Department of Medicine; Division of Cardiology, NYU Grossman School of Medicine Ongoing sponsored research. No future royalty obligations NYU Grossman School of Medicine

Tamoxifen-induced ACM in the PKP2-cKO Mouse Model The PKP2-cKO mouse model recapitulates ACM following induction of PKP2 KO by tamoxifen (TAM) injection Progression of cardiomyopathy evidenced by Masson’s trichrome staining of heart sections in PKP2-cKO mice from 14 to 42 days post-TAM (dpi) 100% mortality by day ~50 following TAM injection Left ventricular ejection fraction (LVEF) diminishes significantly across time Right ventricular (RV) enlargement occurs across time Premature Ventricular Contractions (PVCs) are a clinical hallmark of ACM and emerge in the animal model because of Pkp2 loss Cerrone M, Montnach J, Lin X, et al. Plakophilin-2 is required for transcription of genes that control calcium cycling and cardiac rhythm. Nat Commun. 2017;8(1):106. Published 2017 Jul 24. ISO, isoproterenol; cKO: conditional knockout RP-A601: PKP2-ACM

1Data not illustrated in Figures Increased Survival & Preserved Cardiac Function in the PKP2-cKO Model RP-A601: PKP2-ACM AAVrh.74-PKP2 delivered 7 days post-TAM: 100% survival to 5 months, compared to 100% mortality by day ~50 in PKP2-cKO control animals Preserved Ejection Fraction and Right Ventricular Area at 28 Days, sustained to 5 months AAVrh.74-PKP2 delivered 14 days post-TAM: Mitigated isoproterenol-induced PVCs and arrhythmia, disease-related characteristics of ACM Robust benefit on survival, cardiac function & structure to 5 months1 *p <0.0001 vs PKP2-cKO FB *p <0.0001 vs PKP2-cKO FB *p <0.05 vs PKP2-cKO FBISO = isoproterenol; TAM = tamoxifen; ECG = Electrocardiography ISO-Induced Arrhythmia AAV+14D Post-TAM; ECG at 21D post-TAM AAV +7D post-TAM AAVrh.74-PKP2a Dose 1 (n = 10) Control-FB (n = 9) PKP2-cKO-FB (n = 10) -

cDNA/isoform: PKP2a: full wild type coding sequence of therapeutic gene, protein loss drives ACM AAV Serotype: AAV.rh74 serotype associated with favorable safety profile in DMD/LGMD2E1-2; potential for safe administration at optimal doses for adult ACM patients Cardiac-Specific Promoter: Effectively drives expression of therapeutic transgene in cardiomyocytes; minimizes off-target effects Route of Administration: Intravenous (IV) Pharmacology studies demonstrate efficient cardiac transduction with IV administration Robust Proof of Concept in Disease Relevant Animal Model: NYU Cardiac-specific cKO-PKP2 mouse (biologically relevant translational model) Optimal Gene Therapy for PKP2-ACM, Expected to be First-and Best-In-Class RP-A601: PKP2-ACM 1Rodino-Klapac et. al. Safety, β-Sarcoglycan Expression, and Functional Outcomes From Systemic Gene Transfer of rAAVrh74.MHCK7.hSGCB in LGMD2E/R4. Presented at the Muscular Dystrophy Association (MDA) Conference. Nashville, TN, March 13−16, 2022. 2Mendell et. al. A Phase 2 clinical trial evaluating the safety and efficacy of delandistrogene moxeparvovec (SRP-9001) in patients with Duchenne muscular dystrophy. Presented at the 2022 Muscular Dystrophy Association (MDA) Conference Nashville, TN, March 13–16, 2022.

Clinical Development Plan Moving Toward Phase 1 Dose Escalation Study FDA, Food and Drug Administration; 1H, first half of the year. FIH, first in human. a potential for up to 3 additional patients per cohort in setting of DLTs in 1st 3 pts Study design: FIH, multi-center, 3+3 dose escalationa study Two dose levels planned Target population: High risk adult PKP2-ACM patients Primary endpoint: Safety events related to RP-A601 Secondary and exploratory endpoints: TBD High Level Phase 1 Proposed Trial Design GMP drug product manufacturing completed Pharmacology and GLP toxicology studies Potency assay Upcoming Scientific Advisory Board Clinical trial planning activities, including site selection, underway Submit Orphan Disease Designation FDA IND submission anticipated Q2 2023 Completed or Ongoing Activities To provide context for the Phase 1 study results, we will leverage data from existing ACM registries as well as longitudinal and population-level data from published case series Natural History RP-A601: PKP2-ACM

Pre-eminent maturing gene therapy pipeline in which each program is First- and Best-in-Class Experienced management team with a history of delivering transformative and curative therapies to patients with devastating diseases Well-capitalized and poised to elevate from a clinical-stage to a commercial-stage company Rocket – The Leader in Rare Disease Gene Therapy

THANK YOU!