Table of Contents
| Washington | 2834 | 91-1663741 | ||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
Craig E. Sherman, Esq. Mark J. Handfelt, Esq. Wilson Sonsini Goodrich & Rosati Professional Corporation 701 Fifth Avenue, Suite 5100 Seattle, Washington 98104 (206) 883-2500 | Marcia S. Kelbon, Esq. Alex F. Sutter, Esq. Omeros Corporation 1420 Fifth Avenue, Suite 2600 Seattle, Washington 98101 (206) 676-5000 | James R. Tanenbaum, Esq. Jonathan E. Kahn, Esq. Morrison & Foerster LLP 1290 Avenue of the Americas New York, New York 10104 (212) 468-8000 |
o Large accelerated filer | o Accelerated filer | þ Non-accelerated filer | o Smaller reporting company | |||
| (Do not check if a smaller reporting company) |
Table of Contents
| The information in this preliminary prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission is declared effective. This preliminary prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted. |

| Per Share | Total | |||
| Public offering price | $ | $ | ||
| Underwriting discounts and commissions | $ | $ | ||
| Proceeds, before expenses, to Omeros Corporation | $ | $ |
| Leerink Swann | Needham & Company, LLC |
| Page | ||||||||
| 1 | ||||||||
| 11 | ||||||||
| 32 | ||||||||
| 34 | ||||||||
| 35 | ||||||||
| 36 | ||||||||
| 38 | ||||||||
| 40 | ||||||||
| 42 | ||||||||
| 66 | ||||||||
| 105 | ||||||||
| 110 | ||||||||
| 126 | ||||||||
| 129 | ||||||||
| 131 | ||||||||
| 136 | ||||||||
| 139 | ||||||||
| 146 | ||||||||
| 146 | ||||||||
| 146 | ||||||||
| F-1 | ||||||||
| EX-5.1 | ||||||||
| EX-23.1 | ||||||||
i
Table of Contents
1
Table of Contents
2
Table of Contents
3
Table of Contents
4
Table of Contents
| • | obtain regulatory approval for our PharmacoSurgery product candidates OMS103HP, OMS302 and OMS201; | |
| • | maximize commercial opportunity for our PharmacoSurgery product candidates OMS103HP, OMS302 and OMS201; | |
| • | continue to leverage our business model to mitigate risk by combining our multiple late-stage PharmacoSurgery product candidates with our deep and diverse pipeline of preclinical development programs; | |
| • | further expand our broad patent portfolio; and | |
| • | manage our business with continued efficiency and discipline, while continuing to evaluate opportunities and acquire technologies that meet our business objectives. |
5
Table of Contents
| • | We are largely dependent on the success of our PharmacoSurgery product candidates, particularly our lead product candidate, OMS103HP, and our clinical trials may fail to adequately demonstrate the safety and efficacy of OMS103HP or our other PharmacoSurgery product candidates. If a clinical trial fails, if regulatory approval is delayed or if additional clinical trials are required, our development costs may increase and we will not have the anticipated revenue from that product candidate to fund our operations. | |
| • | We are a clinical-stage company with no product revenue and no products approved for marketing. The regulatory approval process is expensive, time-consuming and uncertain, and our product candidates have not been, and may not be, approved for sale by regulatory authorities. Even if approved for sale by the appropriate regulatory authorities, our products may not achieve market acceptance and we may never achieve profitability. | |
| • | Our preclinical development programs may not generate product candidates that are suitable for clinical testing or that can be successfully commercialized. | |
| • | Our patents may not adequately protect our present and future product candidates or permit us to gain or keep a competitive advantage. Our pending patents for our present and future product candidates may not be issued. |
6
Table of Contents
7
Table of Contents
| Shares of common stock offered by us | shares | |
| Shares of common stock to be outstanding after this offering | shares |
| Use of proceeds | We plan to use the net proceeds of this offering to fund (1) the completion of our Phase 3 clinical trials for OMS103HP and the submission of the related NDA(s) to the FDA, (2) the launch and commercialization of OMS103HP, (3) the clinical development of OMS302 and OMS201, (4) the development of our pipeline of preclinical programs and (5) working capital, capital expenditures, repayment of debt, potential acquisitions of products or technologies and general corporate purposes. See “Use of Proceeds.” |
| Proposed NASDAQ Global Market symbol | OMER |
| • | 5,441,744 shares of common stock issuable upon the exercise of options outstanding at March 31, 2009, at a weighted-average exercise price of $0.72 per share; |
| • | 205,000 shares of common stock issuable upon exercise of options granted from April 1, 2009 to June 15, 2009, at a weighted-average exercise price of $6.33 per share; |
| • | 22,613 shares of common stock issuable upon exercise of warrants outstanding at March 31, 2009, which will not automatically terminate upon the closing of this offering, at a weighted-average exercise price of $4.66 per share; and | |
| • | 2,121,855 shares of common stock available for future issuance under our 2008 Equity Incentive Plan. |
| • | the automatic conversion of all outstanding shares of our convertible preferred stock into 22,567,407 shares of common stock, effective upon the closing of this offering; | |
| • | the conversion of all outstanding warrants to purchase shares of our convertible preferred stock into warrants to purchase 409,578 shares of common stock, effective upon the closing of this offering, 387,030 of which must be exercised or will automatically terminate upon the closing of this offering; | |
| • | the issuance of shares of common stock assuming the cashless net exercise of warrants that will automatically terminate upon the closing of this offering based on the assumed initial public offering price of $ (the mid-point of the range set forth on the cover page of this prospectus); and | |
| • | no exercise by the underwriters of their right to purchase additional shares of common stock to cover over-allotments, if any. |
8
Table of Contents
| Period from | Period from | |||||||||||||||||||||||||||
| June 16, 1994 | June 16, 1994 | |||||||||||||||||||||||||||
| Three Months Ended | (Inception) to | (Inception) to | ||||||||||||||||||||||||||
| March 31, | March 31, | Year Ended December 31, | December 31, | |||||||||||||||||||||||||
| 2009 | 2008 | 2009 | 2008 | 2007 | 2006 | 2008 | ||||||||||||||||||||||
| (in thousands, except share and per share data) | ||||||||||||||||||||||||||||
Consolidated Statements of Operations Data: | ||||||||||||||||||||||||||||
| Grant revenue | $ | 197 | $ | 234 | $ | 3,590 | $ | 1,170 | $ | 1,923 | $ | 200 | $ | 3,393 | ||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||
| Research and development | 4,022 | 4,170 | 66,256 | 17,850 | 15,922 | 9,637 | 62,234 | |||||||||||||||||||||
| Acquired in-process research and development | — | — | 10,891 | — | — | 10,891 | 10,891 | |||||||||||||||||||||
| General and administrative | 1,410 | 1,596 | 33,893 | 7,845 | 10,398 | 3,625 | 32,483 | |||||||||||||||||||||
| Total operating expenses | 5,432 | 5,766 | 111,040 | 25,695 | 26,320 | 24,153 | 105,608 | |||||||||||||||||||||
| Loss from operations | (5,235 | ) | (5,532 | ) | (107,450 | ) | (24,525 | ) | (24,397 | ) | (23,953 | ) | (102,215 | ) | ||||||||||||||
| Investment income | 81 | 279 | 5,244 | 661 | 1,582 | 1,088 | 5,163 | |||||||||||||||||||||
| Interest expense | (590 | ) | (22 | ) | (1,219 | ) | (335 | ) | (151 | ) | (91 | ) | (629 | ) | ||||||||||||||
| Other income (expense) | 262 | 172 | 696 | 372 | (125 | ) | 179 | 434 | ||||||||||||||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (102,729 | ) | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | $ | (97,247 | ) | |||||||
| Basic and diluted net loss per common share | $ | (0.95 | ) | $ | (0.92 | ) | $ | (4.22 | ) | $ | (5.44 | ) | $ | (6.17 | ) | |||||||||||||
| Weighted-average shares used to compute basic and diluted net loss per common share | 5,740,914 | 5,522,711 | 5,651,583 | 4,248,212 | 3,694,388 | |||||||||||||||||||||||
| Pro forma basic and diluted net loss per common share (unaudited) | $ | (0.20 | ) | $ | (0.84 | ) | ||||||||||||||||||||||
| Weighted-average pro forma shares used to compute pro forma basic and diluted net loss per common share (unaudited) | 28,180,321 | 27,978,990 | ||||||||||||||||||||||||||
9
Table of Contents
| As of March 31, 2009 | ||||||||||||
| Pro Forma | ||||||||||||
| Pro | As | |||||||||||
| Actual | Forma | Adjusted (1) | ||||||||||
| (in thousands) | ||||||||||||
Consolidated Balance Sheet Data: | ||||||||||||
| Cash, cash equivalents and short-term investments | $ | 16,818 | $ | 16,818 | ||||||||
| Working capital (deficit) | (6,261 | ) | (6,261 | ) | ||||||||
| Total assets | 18,782 | 18,782 | ||||||||||
| Total notes payable | 16,344 | 16,344 | ||||||||||
| Preferred stock warrant liability | 1,720 | — | ||||||||||
| Convertible preferred stock | 91,019 | — | ||||||||||
| Deficit accumulated during the development stage | (102,729 | ) | (102,729 | ) | ||||||||
| Total shareholders’ equity (deficit) | (96,093 | ) | (3,354 | ) | ||||||||
| (1) | A $1.00 increase (decrease) in the assumed public offering price of $ would increase (decrease) each of cash, cash equivalents and short-term investments, working capital, total assets and total shareholders’ equity (deficit) by $ , assuming that the number of shares offered by us, as set forth on the cover page of this prospectus, remains the same, and after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us. |
10
Table of Contents
11
Table of Contents
12
Table of Contents
| • | discussions with the FDA or comparable foreign authorities regarding the scope or design of our clinical trials; | |
| • | delays or the inability to obtain required approvals from institutional review boards or other governing entities at clinical sites selected for participation in our clinical trials; | |
| • | delays in enrolling patients into clinical trials; | |
| • | lower than anticipated retention rates of patients in clinical trials; | |
| • | the need to repeat or conduct additional clinical trials as a result of problems such as inconclusive or negative results, poorly executed testing or unacceptable design; | |
| • | an insufficient supply of product candidate materials or other materials necessary to conduct our clinical trials; | |
| • | the need to qualify new suppliers of product candidate materials for FDA and foreign regulatory approval; |
13
Table of Contents
| • | an unfavorable FDA inspection or review of a clinical trial site or records of any clinical investigation; | |
| • | the occurrence of drug-related side effects or adverse events experienced by participants in our clinical trials; or | |
| • | the placement of a clinical hold on a trial. |
| • | failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols; | |
| • | inspection of the clinical trial operations or trial sites by the FDA or other regulatory authorities resulting in the imposition of a clinical hold; | |
| • | unforeseen safety issues or any determination that a trial presents unacceptable health risks; or | |
| • | lack of adequate funding to continue the clinical trial, including the incurrence of unforeseen costs due to enrollment delays, requirements to conduct additional trials and studies and increased expenses associated with the services of our contract research organizations, or CROs, and other third parties. |
| • | complete the Phase 3 clinical trials of OMS103HP for use in arthroscopic ACL reconstruction surgery; | |
| • | initiate, conduct and complete the Phase 3 clinical trials of OMS103HP for use in arthroscopic meniscectomy surgery; | |
| • | conduct and complete the clinical trials of OMS302 for use during lens replacement surgery; | |
| • | conduct and complete the clinical trials of OMS201 for use in endoscopic surgery of the urological tract; | |
| • | continue our research and development; | |
| • | make milestone payments to our collaborators; |
14
Table of Contents
| • | make principal and interest payments due under our debt facility with BlueCrest Venture Finance Master Fund Limited, or BlueCrest; | |
| • | initiate and conduct clinical trials for other product candidates; and | |
| • | launch and commercialize any product candidates for which we receive regulatory approval. |
15
Table of Contents
| • | our ability to provide acceptable evidence of safety and efficacy; | |
| • | availability, relative cost and relative efficacy of alternative and competing treatments; | |
| • | the effectiveness of our marketing and distribution strategy to, among others, hospitals, surgery centers, physiciansand/or pharmacists; | |
| • | prevalence of the surgical procedure or condition for which the product is approved; |
| • acceptance by physicians of each product as a safe and effective treatment; |
| • | perceived advantages over alternative treatments; | |
| • | relative convenience and ease of administration; | |
| • | the availability of adequate reimbursement by third parties; | |
| • | the prevalence and severity of adverse side effects; | |
| • | publicity concerning our products or competing products and treatments; and | |
| • | our ability to obtain sufficient third-party insurance coverage. |
16
Table of Contents
| • | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; | |
| • | the inability of sales personnel to obtain access to or persuade adequate numbers of hospitals, surgery centers, physiciansand/or pharmacists to purchase, use or prescribe our approved product candidates; | |
| • | the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines; and | |
| • | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
17
Table of Contents
18
Table of Contents
19
Table of Contents
20
Table of Contents
21
Table of Contents
| • | we might not have been the first to make the inventions covered by any of our patents, if issued, or our pending patent applications; | |
| • | we might not have been the first to file patent applications for these inventions; | |
| • | others may independently develop similar or alternative technologies or products or duplicate any of our technologies or products; | |
| • | it is possible that none of our pending patent applications will result in issued patents or, if issued, these patents may not be sufficient to protect our technology or provide us with a basis for commercially viable products and may not provide us with any competitive advantages; | |
| • | if our pending applications issue as patents, they may be challenged by third parties as not infringed, invalid or unenforceable under U.S. or foreign laws; | |
| • | if issued, the patents under which we hold rights may not be valid or enforceable; or | |
| • | we may develop additional proprietary technologies or products that are not patentable and which are unlikely to be adequately protected through trade secrets if, for example, a competitor were to independently develop duplicative, similar or alternative technologies or products. |
22
Table of Contents
23
Table of Contents
24
Table of Contents
25
Table of Contents
| • | develop and market products that are less expensive or more effective than any future products developed from our product candidates; | |
| • | commercialize competing products before we can launch any products developed from our product candidates; | |
| • | operate larger research and development programs, possess commercial-scale manufacturing operations or have substantially greater financial resources than we do; | |
| • | initiate or withstand substantial price competition more successfully than we can; | |
| • | have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; | |
| • | more effectively negotiate third-party licenses and strategic relationships; and | |
| • | take advantage of acquisition or other opportunities more readily than we can. |
26
Table of Contents
| • | restrictions on such product candidates or manufacturing processes; | |
| • | withdrawal of the product candidates from the market; | |
| • | voluntary or mandatory recalls; | |
| • | fines; | |
| • | suspension of regulatory approvals; | |
| • | product seizures; or | |
| • | injunctions or the imposition of civil or criminal penalties. |
27
Table of Contents
| • | a covered benefit under its health plan; | |
| • | safe, effective and medically necessary; | |
| • | appropriate for the specific patient; | |
| • | cost-effective; and | |
| • | neither experimental nor investigational. |
28
Table of Contents
| • | results from our clinical trial programs, including our ongoing Phase 3 clinical trials for OMS103HP for use in ACL reconstruction surgery, our Phase 2 clinical trial for OMS103HP for use in meniscectomy surgery, our ongoing Phase 2 clinical trial for OMS302, and our ongoing Phase 1/Phase 2 clinical trial for OMS201; | |
| • | FDA or international regulatory actions, including failure to receive regulatory approval for any of our product candidates; | |
| • | failure of any of our product candidates, if approved, to achieve commercial success; | |
| • | quarterly variations in our results of operations or those of our competitors; | |
| • | our ability to develop and market new and enhanced product candidates on a timely basis; | |
| • | announcements by us or our competitors of acquisitions, regulatory approvals, clinical milestones, new products, significant contracts, commercial relationships or capital commitments; | |
| • | third-party coverage and reimbursement policies; | |
| • | additions or departures of key personnel; | |
| • | commencement of, or our involvement in, litigation; | |
| • | our ability to meet our repayment and other obligations under our debt facility with BlueCrest, pursuant to which we have borrowed $17.0 million; | |
| • | changes in governmental regulations or in the status of our regulatory approvals; | |
| • | changes in earnings estimates or recommendations by securities analysts; | |
| • | any major change in our board or management; | |
| • | general economic conditions and slow or negative growth of our markets; and | |
| • | political instability, natural disasters, warand/or events of terrorism. |
29
Table of Contents
30
Table of Contents
31
Table of Contents
| • | our ability to complete the Phase 3 clinical trials of OMS103HP in patients undergoing ACL reconstruction surgery and to submit a related NDA to the FDA during the second half of 2010; | |
| • | our ability to review the data from our first Phase 2 trial of OMS103HP in patients undergoing arthroscopic meniscectomy surgery in the second half of 2009; | |
| • | our ability to market OMS103HP by 2011; |
| • | our ability to complete the Phase 2 clinical trial for OMS302 in patients undergoing cataract surgery in mid-2009; |
| • | our ability to complete the Phase 1/Phase 2 clinical trial of OMS201 in patients undergoing ureteroscopic removal or ureteral or renal stones in the first half of 2010; |
| • | our ability to achieve the expected near-term milestones in our pipeline of preclinical development programs, including the selection of a clinical product candidate for our MASP-2 program in the second half of 2009, submission of an IND to the FDA for our Addiction program in the second half of 2009 and the selection of one or more clinical product candidates for our PDE10 program in the second half of 2009, and the size of target markets; |
| • | our expectations regarding the growth in the number of arthroscopic, cataract and uroendoscopic operations, the rates at which each of our PharmacoSurgery product candidates will be reimbursed to the surgical facility for its utilization and to the surgeon for its use, the size of the markets for our PharmacoSurgery product candidates, in particular, the market opportunity for OMS103HP, and the rate and degree of adoption and market penetration of our PharmacoSurgery product candidates; | |
| • | our ability to obtain commercial supplies of our PharmacoSurgery product candidates, our competition and, if approved, our ability to successfully commercialize our PharmacoSurgery product candidates with a limited, hospital-based marketing and sales force; | |
| • | our expectations regarding the clinical benefits of our PharmacoSurgery product candidates; | |
| • | the extent of protection that our patents provide and our pending patent applications may provide, if patents issue from such applications, to our technologies and programs; | |
| • | our estimate regarding how long our existing cash, cash equivalents and short-term investments, along with the net proceeds from this offering, will be sufficient to fund our anticipated operating expenses and capital expenditures, the factors impacting our future |
32
Table of Contents
| capital expenditures and our expected number of full-time employees by the end of 2009; |
| • | our expectations regarding our ability to de-orphanize orphan GPCRs and the number of druggable targets among the orphan GPCRs; | |
| • | our ability to meet our repayment and other obligations under our debt facility with BlueCrest, pursuant to which we have borrowed $17.0 million; and | |
| • | our estimates regarding the use of the net proceeds from this offering and our future net losses, revenues, expenses and net operating loss carryforwards and research and development tax credit carryforwards. |
33
Table of Contents
| • | approximately $ to fund the completion of our clinical trials and our submission of the related NDA(s) to the FDA for our lead PharmacoSurgery product candidate, OMS103HP; | |
| • | approximately $ to fund the launch and commercialization of OMS103HP; | |
| • | approximately $ to fund the clinical development of our other PharmacoSurgery product candidates, OMS302 and OMS201, through Phase 2 clinical trials; and | |
| • | the remainder to continue to fund our pipeline of preclinical product development programs focused on inflammation and CNS disorders, and to fund working capital, capital expenditures, potential acquisitions of products or technologies and general corporate purposes. |
34
Table of Contents
35
Table of Contents
| • | on an actual basis; | |
| • | on a pro forma basis reflecting (a) the automatic conversion of all outstanding shares of our convertible preferred stock into 22,567,407 shares of our common stock upon the closing of this offering and (b) the automatic conversion of all outstanding warrants to purchase convertible preferred stock into warrants to purchase 409,578 shares of our common stock upon the closing of this offering, resulting in the reclassification of $1.7 million from preferred stock warrant liability to additional paid-in capital; | |
| • | on a pro forma as adjusted basis to give effect (a) to the issuance and sale by us of shares of common stock in this offering and the receipt of the net proceeds from our sale of these shares at an assumed initial public offering price of $ per share (the mid-point of the range set forth on the cover page of this prospectus), after deducting estimated underwriting discounts and commissions and estimated offering expenses payable by us and (b) to the issuance of shares of common stock assuming the cashless net exercise of warrants that will automatically terminate upon the closing of this offering based on the assumed initial public offering price. |
| As of March 31, 2009 | ||||||||||||
| Pro Forma | ||||||||||||
| Actual | Pro Forma | As Adjusted | ||||||||||
| (in thousands, except share | ||||||||||||
| and per share data) | ||||||||||||
| Cash, cash equivalents and short-term investments | $ | 16,818 | $ | 16,818 | $ | |||||||
| Total notes payable | $ | 16,344 | $ | 16,344 | ||||||||
| Preferred stock warrant liability | 1,720 | — | — | |||||||||
| Convertible preferred stock; Issued and outstanding shares—22,567,407 (0 pro forma and pro forma as adjusted) | 91,019 | — | — | |||||||||
| Shareholders’ equity (deficit): | ||||||||||||
| Preferred stock, par value $0.01 per share; Authorized shares—26,314,511 (20,000,000 pro forma and pro forma as adjusted; issued and outstanding shares—0 pro forma and pro forma as adjusted) | — | — | — | |||||||||
| Common stock, par value $0.01 per share; Authorized shares—40,000,000 (150,000 pro forma and pro forma as adjusted); issued and outstanding shares—5,787,899 (28,355,306 pro forma and pro forma as adjusted) | 57 | 284 | ||||||||||
| Additional paid-in capital | 6,586 | 99,098 | ||||||||||
| Accumulated other comprehensive loss | (7 | ) | (7 | ) | ||||||||
| Deficit accumulated during the development stage | (102,729 | ) | (102,729 | ) | ||||||||
| Total shareholders’ equity (deficit) | (96,093 | ) | (3,354 | ) | ||||||||
| Total capitalization | $ | 12,990 | $ | 12,990 | $ | |||||||
36
Table of Contents
| • | 5,441,744 shares of common stock issuable upon the exercise of options outstanding at March 31, 2009, at a weighted-average exercise price of $0.72 per share; |
| • | 205,000 shares of common stock issuable upon exercise of options granted from April 1, 2009 to June 15, 2009, at a weighted-average exercise price of $6.33 per share; |
| • | 22,613 shares of common stock issuable upon exercise of warrants outstanding at March 31, 2009, which will not automatically terminate upon the closing of this offering, at a weighted-average exercise price of $4.66 per share; and | |
| • | 2,121,855 shares of common stock available for future issuance under our 2008 Equity Incentive Plan. |
37
Table of Contents
| Assumed initial public offering price per share | $ | |||||||
| Historical net tangible book value per common share at March 31, 2009 | $ | (16.61 | ) | |||||
| Pro forma increase in net tangible book value per common share attributable to conversion of all outstanding convertible preferred stock into common stock and the reclassification of the preferred stock warrant liability to additional paid-in capital | (16.49 | ) | ||||||
| Pro forma net tangible book value per share as of March 31, 2009 | (0.12 | ) | ||||||
| Pro forma increase in net tangible book value per share attributable to investors participating in this offering | ||||||||
| Pro forma net tangible book value per share after this offering | ||||||||
| Dilution in pro forma net tangible book value per share to investors purchasing shares in this offering | $ | |||||||
38
Table of Contents
| Average | ||||||||||||||||||||
| Shares Purchased | Total Consideration | Price Per | ||||||||||||||||||
| Number | Percent | Amount | Percent | Share | ||||||||||||||||
| Existing shareholders | 28,355,306 | % | $ | 92,051,000 | % | $ | 3.25 | |||||||||||||
| Holders of warrants exercised at closing | ||||||||||||||||||||
| New investors | ||||||||||||||||||||
| Total | % | $ | % | $ | ||||||||||||||||
| • | 5,441,744 shares of common stock issuable upon the exercise of options outstanding at March 31, 2009, at a weighted-average exercise price of $0.72 per share; |
| • | 205,000 shares of common stock issuable upon the exercise of options granted from April 1, 2009 to June 15, 2009, at a weighted-average exercise price of $6.33 per share; |
| • | 22,613 shares of common stock issuable upon exercise of warrants outstanding at March 31, 2009, which will not automatically terminate upon the closing of this offering, at a weighted-average exercise price of $4.66 per share; and | |
| • | 2,121,855 shares of common stock available for future issuance under our 2008 Equity Incentive Plan. |
39
Table of Contents
| Period from | Period from | |||||||||||||||||||||||||||||||||||
| Three Months | June 16, 1994 | June 16, 1994 | ||||||||||||||||||||||||||||||||||
| Ended | (inception) to | (inception) to | ||||||||||||||||||||||||||||||||||
| March 31, | March 31, | Years Ended December 31, | December 31, | |||||||||||||||||||||||||||||||||
| 2009 | 2008 | 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | 2008 | ||||||||||||||||||||||||||||
| (in thousands, except share and per share data) | ||||||||||||||||||||||||||||||||||||
Consolidated Statements of Operations Data: | ||||||||||||||||||||||||||||||||||||
| Grant revenue | $ | 197 | $ | 234 | $ | 3,590 | $ | 1,170 | $ | 1,923 | $ | 200 | $ | — | $ | — | $ | 3,393 | ||||||||||||||||||
| Operating expenses: | ||||||||||||||||||||||||||||||||||||
| Research and development | 4,022 | 4,170 | 66,256 | 17,850 | 15,922 | 9,637 | 5,803 | 2,670 | 62,234 | |||||||||||||||||||||||||||
| Acquired in-process research and development | — | — | 10,891 | — | — | 10,891 | — | — | 10,891 | |||||||||||||||||||||||||||
| General and administrative | 1,410 | 1,596 | 33,893 | 7,845 | 10,398 | 3,625 | 1,904 | 2,079 | 32,483 | |||||||||||||||||||||||||||
| Total operating expenses | 5,432 | 5,766 | 111,040 | 25,695 | 26,320 | 24,153 | 7,707 | 4,749 | 105,608 | |||||||||||||||||||||||||||
| Loss from operations | (5,235 | ) | (5,532 | ) | (107,450 | ) | (24,525 | ) | (24,397 | ) | (23,953 | ) | (7,707 | ) | (4,749 | ) | (102,215 | ) | ||||||||||||||||||
| Investment income | 81 | 279 | 5,244 | 661 | 1,582 | 1,088 | 333 | 171 | 5,163 | |||||||||||||||||||||||||||
| Interest expense | (590 | ) | (22 | ) | (1,219 | ) | (335 | ) | (151 | ) | (91 | ) | — | — | (629 | ) | ||||||||||||||||||||
| Other income (expense) | 262 | 172 | 696 | 372 | (125 | ) | 179 | 8 | — | 434 | ||||||||||||||||||||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (102,729 | ) | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | $ | (7,366 | ) | $ | (4,578 | ) | $ | (97,247 | ) | |||||||||
| Basic and diluted net loss per common share | $ | (0.95 | ) | $ | (0.92 | ) | $ | (4.22 | ) | $ | (5.44 | ) | $ | (6.17 | ) | $ | (2.12 | ) | $ | (1.34 | ) | |||||||||||||||
| Weighted-average shares used to compute basic and diluted net loss per common share | 5,740,914 | 5,522,711 | 5,651,583 | 4,248,212 | 3,694,388 | 3,468,886 | 3,416,197 | |||||||||||||||||||||||||||||
| Pro forma basic and diluted net loss per common share (unaudited) | $ | (0.20 | ) | $ | (0.84 | ) | ||||||||||||||||||||||||||||||
| Weighted-average pro forma shares used to compute pro forma basic and diluted net loss per common share (unaudited) | 28,180,321 | 27,978,990 | ||||||||||||||||||||||||||||||||||
40
Table of Contents
| As of | ||||||||||||||||||||||||
| March 31, | As of December 31, | |||||||||||||||||||||||
| 2009 | 2008 | 2007 | 2006 | 2005 | 2004 | |||||||||||||||||||
| (in thousands) | ||||||||||||||||||||||||
Consolidated Balance Sheet Data: | ||||||||||||||||||||||||
| Cash, cash equivalents and short-term investments | $ | 16,818 | $ | 19,982 | $ | 24,082 | $ | 35,885 | $ | 12,372 | $ | 14,008 | ||||||||||||
| Working capital (deficit) | (6,261 | ) | (3,083 | ) | 16,526 | 32,277 | 10,672 | 13,664 | ||||||||||||||||
| Total assets | 18,782 | 21,681 | 27,162 | 38,432 | 13,109 | 14,600 | ||||||||||||||||||
| Total notes payable | 16,344 | 16,674 | 1,010 | 2,015 | — | — | ||||||||||||||||||
| Preferred stock warrant liability | 1,720 | 1,780 | 1,562 | 1,037 | 483 | — | ||||||||||||||||||
| Convertible preferred stock | 91,019 | 89,168 | 89,168 | 85,742 | 40,888 | 35,203 | ||||||||||||||||||
| Deficit accumulated in the development stage | (102,729 | ) | (97,247 | ) | (73,420 | ) | (50,329 | ) | (27,553 | ) | (20,187 | ) | ||||||||||||
| Total shareholders’ deficit | (96,093 | ) | (91,166 | ) | (69,941 | ) | (53,363 | ) | (29,743 | ) | (21,114 | ) | ||||||||||||
41
Table of Contents
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
42
Table of Contents
43
Table of Contents
| • | employee and consultant-related expenses, which include salaries and benefits; | |
| • | external research and development expenses incurred pursuant to agreements with third-party manufacturing organizations, contract research organizations and clinical trial sites; | |
| • | facilities, depreciation and other allocated expenses, which include direct and allocated expenses for rent and maintenance of facilities and depreciation of leasehold improvements and equipment; and | |
| • | third-party supplier expenses including laboratory and other supplies. |
| Development | Expected Near- | |||
| Project | Status | Term Milestone (1) | ||
| OMS103HP — Arthroscopic ACL reconstruction | Phase 3 | Complete Phase 3 trials and file NDA in second half of 2010 | ||
| OMS103HP — Arthroscopic meniscectomy | Phase 2 | Review data from Phase 2 trial in second half of 2009 | ||
| OMS302 — Cataract surgery | Phase 2 | Complete Phase 2 trial in mid-2009 | ||
| OMS201 — Ureteroscopy | Phase 1/ Phase 2 | Complete Phase 1/ Phase 2 trial in first half of 2010 | ||
| MASP-2 — Macular degeneration, ischemia-reperfusion injury, transplant surgery | Preclinical | Select clinical candidate in second half of 2009 | ||
| Addiction — Addiction and other compulsive behaviors | Preclinical | File IND in second half of 2009 | ||
| PDE10 — Schizophrenia | Preclinical | Select clinical candidate in second half of 2009 | ||
| PDE7 — Parkinson’s disease, Restless Legs Syndrome | Preclinical | Select clinical candidate | ||
| GPCR — Multiple CNS Disorders | Preclinical | Surrogate de-orphanization of orphan GPCR(s) |
| (1) | Following selection of a clinical candidate, we must conduct additional studies, including in vivo toxicity studies of the clinical candidate. We must submit the results of these studies, together with manufacturing information and analytical results related to the clinical candidate, to the FDA as part of an IND, which must become effective before we may commence clinical trials. Submission of an IND does not always result in the FDA allowing clinical trials to commence. Depending on the nature of information that we must obtain and include in an IND, it may take from 12 to 24 months from selection of the clinical candidate to IND submission, if it occurs at all. All of these expected near-term milestones are subject to a number of risks, uncertainties and assumptions, including those described in “Risk Factors,” and may not occur in the timelines set forth above or at all. |
44
Table of Contents
| Three Months Ended | ||||||||||||||||||||||||
| March 31, | Years Ended December 31, | |||||||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||||||
| (In thousands) | ||||||||||||||||||||||||
Clinical Research and Development | ||||||||||||||||||||||||
| Salaries, benefits, and related costs | $ | 922 | $ | 888 | $ | 3,521 | $ | 2,944 | $ | 1,849 | ||||||||||||||
| Clinical trials | 553 | 688 | 3,525 | 3,630 | 2,116 | |||||||||||||||||||
| Manufacturing services, consulting, laboratory supplies, and other costs | 332 | 516 | 2,080 | 1,943 | 825 | |||||||||||||||||||
| Other costs | 284 | 241 | 1,049 | 633 | 152 | |||||||||||||||||||
| Stock-based compensation | 130 | 186 | 590 | 280 | 181 | |||||||||||||||||||
| Total Clinical Research and Development Expenses | 2,221 | 2,519 | 10,765 | 9,430 | 5,123 | |||||||||||||||||||
Preclinical Research and Development | ||||||||||||||||||||||||
| Salaries, benefits, and related costs | 684 | 650 | 2,572 | 2,315 | 1,848 | |||||||||||||||||||
| Research and preclinical studies, consulting, laboratory supplies, and other costs | 658 | 504 | 2,774 | 2,566 | 1,604 | |||||||||||||||||||
| Other costs | 369 | 356 | 1,346 | 1,412 | 934 | |||||||||||||||||||
| Stock-based compensation | 90 | 141 | 393 | 199 | 128 | |||||||||||||||||||
| Total Preclinical Research and Development Expenses | 1,801 | 1,651 | 7,085 | 6,492 | 4,514 | |||||||||||||||||||
| Total Research and Development Expenses | $ | 4,022 | $ | 4,170 | $ | 17,850 | $ | 15,922 | $ | 9,637 | ||||||||||||||
Total AcquiredIn-process Research and Development Expense | $ | — | $ | — | $ | — | $ | — | $ | 10,891 | ||||||||||||||
45
Table of Contents
46
Table of Contents
| • | revenue recognition; | |
| • | research and development expenses, primarily clinical trial expenses; | |
| • | stock-based compensation; | |
| • | preferred stock warrant liability; and | |
| • | fair value measurement of financial instruments. |
47
Table of Contents
| Three Months | ||||||||||
| Ended March 31, | Years Ended December 31, | |||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||
| Expected volatility | 71% | 60% | 60% | 60% | 60% | |||||
| Expected term (in years) | 6.08 | 6.08 | 6.08 | 6.00-6.08 | 5.00-6.08 | |||||
| Risk-free interest rate | 2.13% | 2.80% - 3.40% | 2.80% - 3.40% | 3.78% - 4.78% | 4.57% - 5.04% | |||||
| Expected dividend yield | 0% | 0% | 0% | 0% | 0% | |||||
| • | the prices of our convertible preferred stock sold to outside investors in arms-length transactions, and the rights, preferences and privileges of our convertible preferred stock relative to those of our common stock including the liquidation preference of our preferred stock; | |
| • | our results of operations, financial position, and the status of our research and product development efforts, including continued enrollment in our Phase 3 clinical trials evaluating OMS103HP’s safety and ability to improve postoperative joint function and reduce pain following ACL reconstruction surgery, continued enrollment in our clinical trials for OMS302 and OMS201, and advancement of our preclinical development programs; | |
| • | our stage of development and business strategy; | |
| • | the composition of and changes to our management team; | |
| • | the market value of a comparison group of publicly traded pharmaceutical and biotechnology companies that are in a similar stage of development to us; | |
| • | the lack of liquidity of our common stock as a private company; |
48
Table of Contents
| • | contemporaneous valuations performed by an unrelated valuation specialist prepared in accordance with methodologies not outlined in theAICPA Practice Aid Valuation of Privately-Held-Company Equity Securities Issued as Compensation; and | |
| • | the likelihood of achieving a liquidity event for the shares of our common stock and underlying stock options, such as an initial public offering, or IPO, given prevailing market conditions. |
49
Table of Contents
50
Table of Contents
| Estimated | ||||||||||||||||
| Number of | Fair Value of | |||||||||||||||
| Shares | Common | |||||||||||||||
| Subject to | Exercise | Stock per | Intrinsic | |||||||||||||
| Options | Price per | Share at | Value per Share | |||||||||||||
Grant Date | Granted | Share | Date of Grant | at Date of Grant | ||||||||||||
| July 2006 | 23,000 | $ | 0.50 | $ | 0.89 | $ | 0.39 | |||||||||
| September 2006 | 28,000 | 0.50 | 0.89 | 0.39 | ||||||||||||
| December 2006 | 4,274,853 | 0.50 | 0.89 | 0.39 | ||||||||||||
| March 2007 | 308,500 | 1.00 | 1.05 | 0.05 | ||||||||||||
| May 2007 | 350,000 | 1.00 | 3.63 | 2.63 | ||||||||||||
| October 2007 | 275,733 | 1.25 | 6.23 | 4.98 | ||||||||||||
| December 2007 | 522,500 | 1.25 | 6.32 | 5.07 | ||||||||||||
| January 2008 | 45,000 | 1.25 | 6.32 | 5.07 | ||||||||||||
| March 2008 | 1,200 | 6.32 | 6.32 | — | ||||||||||||
| June 2008 | 27,000 | 6.32 | 6.88 | 0.56 | ||||||||||||
| September 2008 | 22,000 | 6.88 | 6.87 | — | ||||||||||||
| March 2009 | 15,500 | 6.36 | 6.33 | — | ||||||||||||
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study and advancement of additional product candidates through preclinical development; |
51
Table of Contents
| • | expanded activities in preparation for an IPO; and | |
| • | progress towards an IPO. |
| • | positive efficacy data in a preclinical study evaluating OMS302, our PharmacoSurgery product candidate for use during ophthalmological surgery, and its components in a primate model of lens replacement surgery; | |
| • | filing of an IND for OMS201, our PharmacoSurgery product candidate being developed for use during urological surgery; | |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study; and | |
| • | continued progress toward an IPO. |
| • | initiation of sites for the Phase 2 clinical trial of OMS103HP evaluating the safety and efficacy of the product candidate in patients undergoing meniscectomy surgery; | |
| • | initiation of sites for the OMS201 Phase 1 clinical trial; and | |
| • | continued progress toward an IPO together with an extension in the estimated completion date of the IPO compared to our estimate at September 30, 2007. |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study and Phase 1 study for OMS201; | |
| • | advancement of our preclinical development programs; |
52
Table of Contents
| • | filing of an IND for OMS302, our PharmacoSurgery product candidate being developed for use during cataract surgery; and | |
| • | continued progress toward an IPO together with an extension in the estimated completion date of the IPO compared to our estimate at December 31, 2007. |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study, Phase 1 study for OMS201, and Phase 1/Phase 2 Study for OMS302; | |
| • | advancement of our preclinical development programs; and | |
| • | continued progress toward an IPO together with an extension in the estimated completion date of the IPO compared to our estimate at March 31, 2008. |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study and Phase 1/Phase 2 Study for OMS302; | |
| • | completion of enrollment in our Phase 1 study for OMS201; | |
| • | advancement of our preclinical development programs; | |
| • | establishment of debt facility providing up to $20.0 million in borrowings; | |
| • | extension of an estimated date for an IPO; and | |
| • | weakness of the equity capital markets. |
| • | extension of an estimated date for an IPO; | |
| • | weakness of the equity capital markets; | |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study and Phase 1/Phase 2 study for OMS302; | |
| • | initiation of a Phase 1/Phase 2 study for OMS201; |
53
Table of Contents
| • | advancement of our preclinical development programs; and | |
| • | draw down of additional $12.0 million of debt under our debt facility. |
| • | extension of an estimated date for an IPO; | |
| • | weakness of the equity capital markets; | |
| • | continued advancement in our development programs, including additional patient enrollment in our Phase 3 ACL study, and completed enrollment in our Phase 1/Phase 2 study for OMS302; | |
| • | initiation of sites for a Phase 1/Phase 2 study for OMS201; and | |
| • | advancement of our preclinical development programs. |
| Three Months | ||||||||||||||||||||
| Ended March 31, | Years Ended December 31, | |||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Research and development | $ | 220 | $ | 327 | $ | 983 | $ | 482 | $ | 309 | ||||||||||
| General and administrative | 228 | 391 | 1,332 | 5,574 | 1,130 | |||||||||||||||
| Total | $ | 448 | $ | 718 | $ | 2,315 | $ | 6,056 | $ | 1,439 | ||||||||||
54
Table of Contents
55
Table of Contents
56
Table of Contents
| Period from | ||||
| January 1, | ||||
| 2006 | ||||
| to August 11, | ||||
| 2006 | ||||
| (in thousands) | ||||
| Grant revenue | $ | 200 | ||
| Research and development expenses | 2,394 | |||
| General and administrative expenses | 957 | |||
| Net loss | 3,219 | |||
57
Table of Contents
58
Table of Contents
59
Table of Contents
| • | in 1994, we issued and sold a total of 875,000 shares of Series A convertible preferred stock for aggregate net proceeds of $868,000; | |
| • | in 1998, we issued and sold a total of 2,663,244 shares of Series B convertible preferred stock for aggregate net proceeds of $4.4 million; | |
| • | in 2000, we issued and sold a total of 2,825,291 shares of Series C convertible preferred stock for aggregate net proceeds of $7.2 million; | |
| • | in 2002, we issued and sold a total of 972,580 shares of Series D convertible preferred stock for aggregate net proceeds of $3.7 million; and | |
| • | from 2004 through 2009, we issued and sold a total of 12,895,208 shares of Series E convertible preferred stock for aggregate net proceeds of $61.2 million. |
60
Table of Contents
61
Table of Contents
| • | the progress and results of our clinical trials for OMS103HP, OMS302 and OMS201; | |
| • | costs related to manufacturing services; | |
| • | whether the hiring of a number of new employees to support our continued growth during this period will occur at salary levels consistent with our estimates; | |
| • | the scope, rate of progress, results and costs of our preclinical testing, clinical trials and other research and development activities for additional product candidates; | |
| • | the terms and timing of payments of any collaborative or licensing agreements that we have or may establish, including pursuant to our agreements with Affitech AS and North Coast Biologics; | |
| • | market acceptance of our approved product candidates; | |
| • | the cost, timing and outcomes of the regulatory processes for our product candidates; | |
| • | the costs of commercialization activities, including product manufacturing, marketing, sales and distribution; | |
| • | the number and characteristics of product candidates that we pursue; | |
| • | the cost of establishing clinical and commercial supplies of our product candidates; | |
| • | the cost of preparing, filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; | |
| • | the extent to which we acquire or invest in businesses, products or technologies, although we currently have no commitments or agreements relating to any of these |
62
Table of Contents
| • | whether we receive grant funding for our programs; and | |
| • | our degree of success in commercializing OMS103HP and other product candidates. |
| Payments Due Within | ||||||||||||||||||||
| 1 Year | 2-3 Years | 4-5 Years | More Than 5 Years | Total | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Operating leases (1) | $ | 1,560 | $ | 2,697 | $ | 38 | $ | — | $ | 4,295 | ||||||||||
| License maintenance fees | 5 | 10 | 10 | 40 | 65 | |||||||||||||||
| Notes payable (principal and interest) | 3,704 | 11,759 | 1,730 | — | 17,193 | |||||||||||||||
| Total | $ | 5,269 | $ | 14,466 | $ | 1,778 | $ | 40 | $ | 21,553 | ||||||||||
| (1) | We are contracted to receive sublease income of $603,000 and $240,000 in 2009 and 2010, respectively, which is excluded from operating lease payment amounts. |
| • | Pursuant to our agreement with SMRI, beginning the first calendar year after commencement of commercial sales of a product candidate from our PDE10 program, we will be obligated to pay royalties to SMRI based on net income, as defined in the agreement, not to exceed a set multiple of total grant funding received. Based on the amount of grant funding that we have received as of March 31, 2009, the maximum amount of royalties payable to SMRI is $12.8 million. |
| • | If we select a clinical product candidate for our PDE10 program that is a compound synthesized for us by ComGenex, Inc. (subsequently acquired by Albany Medical Research, Inc.), we may be required to pay ComGenex a low single-digit percentage royalty on sales of a PDE10 inhibitor product candidate that includes the compound and make milestones payments of up to $3.4 million upon the occurrence of certain development events, such as the filing of an IND, the initiation of clinical trials and the receipt of marketing approval. |
63
Table of Contents
| • | If we select a clinical product candidate for our PDE10 program that is a compound synthesized for us by Scottish Biomedical Research, Inc., we may be required to pay Scottish Biomedical a low single-digit percentage royalty on sales of a PDE10 inhibitor product candidate that includes the compound and make milestones payments of up to $178,000 per selected compound upon the occurrence of certain development events, such as the filing of an IND, the initiation of clinical trials and the receipt of marketing approval. |
| • | Pursuant to our MASP-2 antibody discovery and development agreement with Affitech AS, we may be required to pay a low single-digit percentage royalty on any net sales of a product containing a MASP-2 antibody developed by Affitech under the agreement. We also may be required to make additional milestone payments to Affitech of up to $10.1 million upon the achievement of certain development events related to an Affitech-generated MASP-2 antibody, such as the filing of an IND, initiation of clinical trials and the receipt of marketing approval. |
| • | Under our antibody discovery and development agreement with North Coast Biologics, LLC, we may be required to pay a low single-digit percentage royalty on any net sales of a product containing an antibody developed by North Coast under the agreement. Upon the achievement of certain development events, such as the filing of an IND, initiation of clinical trials and the receipt of marketing approval, we also may be required to make additional milestone payments to North Coast of up to $4.0 million for a MASP-2 antibody and $4.1 million per additional target antibody that we may select under the agreement. |
| • | Pursuant to our patent assignment agreement with Roberto Ciccocioppo, Ph.D. under which we acquired assets for our Addiction program, we may be required to pay a low single-digit percentage royalty on any net sales of a product from our Addiction program that is covered by any patents that issue from the patent application we acquired from Dr. Ciccocioppo. In addition, if we grant any third parties rights to manufacture, sell or distribute any such products, we must pay to Dr. Ciccocioppo a percentage of any associated fees we receive from such third parties in the range of low single-digits to low double-digits depending on stage of development at which such rights are granted. We also may be required to make milestone payments of up to $2.3 million upon the achievement of certain development events, such as the initiation of clinical trials and receipt of marketing approval. |
64
Table of Contents
65
Table of Contents
66
Table of Contents
67
Table of Contents
68
Table of Contents
| Product | Targeted | Development | Expected Near- | Worldwide | ||||
Candidate/Program | Procedure/Disease | Status | Term Milestone (1) | Rights | ||||
Inflammation | ||||||||
| OMS103HP — Arthroscopy | Arthroscopic ACL reconstruction | Phase 3 | Complete Phase 3 trials and file NDA in second half of 2010 | Omeros | ||||
| OMS103HP — Arthroscopy | Arthroscopic meniscectomy | Phase 2 | Review data from Phase 2 trial in second half of 2009 | Omeros | ||||
| OMS302 — Ophthalmology | Cataract surgery | Phase 2 | Complete Phase 2 trial in mid-2009 | Omeros | ||||
| OMS201 — Urology | Ureteroscopy | Phase 1/ Phase 2 | Complete Phase 1/ Phase 2 trial in first half of 2010 | Omeros | ||||
| MASP-2 | Macular degeneration, ischemia-reperfusion injury, transplant surgery | Preclinical | Select clinical candidate in second half of 2009 | In-licensed(2) | ||||
Central Nervous System | ||||||||
| Addiction | Addiction and other compulsive behaviors | Preclinical | File IND in second half of 2009 | Omeros | ||||
| PDE10 | Schizophrenia | Preclinical | Select clinical candidate in second half of 2009 | Omeros | ||||
| PDE7 | Parkinson’s disease, Restless Legs Syndrome | Preclinical | Select clinical candidate | Omeros | ||||
| GPCR | Multiple CNS Disorders | Preclinical | Surrogate de-orphanization of orphan GPCR(s) | Omeros |
| (1) | Following selection of a clinical candidate, we must conduct additional studies, including in vivo toxicity studies of the clinical candidate. We must submit the results of these studies, together with manufacturing information and analytical results related to the clinical candidate, to the FDA as part of an IND, which must become effective before we may commence clinical trials. Submission of an IND does not always result in the FDA allowing clinical trials to commence. Depending on the nature of information that we must obtain and include in an IND, it may take from 12 to 24 months from selection of the clinical candidate to IND submission, if it occurs at all. All of these expected near-term milestones are subject to a number of risks, uncertainties and assumptions, including those described in “Risk Factors,” and may not occur in the timelines set forth above or at all. | |
| (2) | We hold worldwide exclusive licenses to rights in connection with MASP-2, the antibodies targeting MASP-2 and the therapeutic applications for those antibodies from the University of Leicester and from its collaborator, Medical Research Council at Oxford University. |
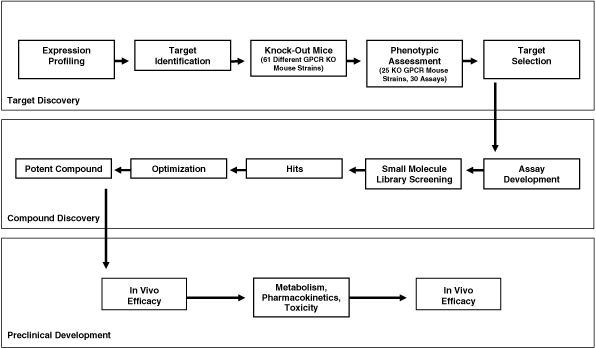
| • | Obtain regulatory approval for our PharmacoSurgery product candidates OMS103HP, OMS302 and OMS201.We are conducting Phase 3 and Phase 2 clinical trials for OMS103HP and we plan to submit an NDA for OMS103HP in the second half of 2010. In addition, we are conducting a Phase 2 clinical trial for OMS302 and a Phase 1/Phase 2 clinical trial for OMS201. Each of these PharmacoSurgery product candidates are specifically comprised of APIs contained in generic, FDA-approved drugs with established safety and pharmacological profiles, and are delivered to the surgical site in low concentrations with minimal systemic uptake and reduced risk of adverse side effects. All of these product candidates are eligible for submission under the potentially less-costly and time-consuming Section 505(b)(2) NDA process. |
69
Table of Contents
| • | Maximize commercial opportunity for our PharmacoSurgery product candidates OMS103HP, OMS302 and OMS201.Our PharmacoSurgery product candidates target large surgical markets with significant unmet medical needs. For each of our product candidates, we have retained all manufacturing, marketing and distribution rights and have not entered into any partnerships granting any of these rights to any third party. Our product candidates do not require a surgeon to change his or her operating procedure. In addition to ease of use, we believe that the clinical benefits of our product candidates could provide surgeons a competitive marketing advantage and may facilitate third-party payor acceptance, all of which we expect will drive adoption and market penetration. Because accessing the surgeons who perform the procedures targeted by our PharmacoSurgery product candidates requires a limited, hospital-based marketing and sales force, we believe that we are well positioned to successfully commercialize these product candidates independently or through third-party partnerships. | |
| • | Continue to leverage our business model to mitigate risk by combining our multiple late-stage PharmacoSurgery product candidates with our deep and diverse pipeline of preclinical development programs.Our lead PharmacoSurgery product is in clinical trials for two distinct therapeutic indications, providing two potential paths for commercialization. We are also advancing two additional PharmacoSurgery product candidates through clinical trials, and from our intellectual property estate we are able to develop a series of proprietary follow-on product candidates. Further, all of these current product candidates consist of generic APIs and are eligible for submission under the potentially less-costly and time-consuming Section 505(b)(2) NDA process. We believe that these attributes collectively mitigate the typical risks of late-stage clinical programs. Leveraging our clinical development experience and our expertise in inflammation and the CNS, we have built multiple development programs, including our PharmacoSurgery andMASP-2 programs targeting large markets focused on inflammation, and our Addiction, PDE10, PDE7 and GPCR programs targeting large markets in disorders of the CNS. By combining our late-stage PharmacoSurgery product candidates with this deep and diverse pipeline of preclinical development programs, we believe that our business model mitigates risk by creating multiple opportunities for commercial success. |
| • | Further expand our broad patent portfolio. We have made a significant investment in the development of our patent portfolio to protect our technologies and programs, and will continue to do so. We own a total of 21 issued or allowed patents and 52 pending patent applications in the United States, 84 issued or allowed patents and 96 pending patent applications in commercially significant foreign markets, and we also hold worldwide exclusive licenses to two pending United States patent applications, an issued foreign patent and two pending foreign patent applications. Our patent portfolio for our PharmacoSurgery platform is directed to locally delivered compositions and treatment methods using agents selected from broad therapeutic classes such as pain and inflammation inhibitory agents, spasm inhibitory agents, restenosis inhibitory agents, tumor cell adhesion inhibitory agents, mydriatic agents and agents that reduce intraocular pressure. We intend to continue to maintain an aggressive intellectual property strategy in the United States and other commercially significant markets and plan to seek additional patent protection for our existing programs as they advance, for our new inventions and for new products that we develop or acquire. |
| • | Manage our business with continued efficiency and discipline. We have efficiently utilized our capital and human resources to develop and acquire our product candidates and programs, build a modern research facility and vivarium and create a broad intellectual property portfolio. We operate cross-functionally and are led by an experienced management team with backgrounds in developing and commercializing product candidates. We use rigorous project management techniques to assist us in making disciplined strategic program decisions and to limit the risk profile of our product pipeline. |
70
Table of Contents
| In addition, we plan to continue to seek and access external sources of grant funding to support the development of our pipeline programs. We will continue to evaluate opportunities and, as appropriate, acquire technologies that meet our business objectives. We successfully implemented this strategy with our acquisition of nura in 2006, which expanded and diversified our CNS pipeline and strengthened our discovery research capabilities. In addition, we will also consider strategic partnerships to maximize commercial opportunities for our product candidates. |
| • | alterations in vascular caliber, or vasodilation, that lead to an increase in blood flow; | |
| • | structural changes in the microvasculature that permit plasma proteins to leave the circulation, or plasma extravasation; and | |
| • | white cell migration from the microcirculation to the site of tissue injury. |
| • | Ketoprofen, a non-steroidal anti-inflammatory drug, or NSAID, is a non-selective inhibitor of the pro-inflammatory mediators COX-1 and COX-2, with potent anti-inflammatory and analgesic actions that result from inhibiting the synthesis of the pro-inflammatory mediator PGE2, and antagonizing the effects of bradykinin, another inflammatory mediator; |
71
Table of Contents
| • | Amitriptylineis a compound with analgesic activity that inhibits the pro-inflammatory actions of histamine and serotonin released locally at the site of tissue trauma; and |
| • | Oxymetazolineis a vasoconstrictor and also activates serotonin receptors, located on a group of nerve fibers called primary afferents, that can inhibit the release of pro-inflammatory mediators such as substance P and calcitonin gene-related peptide, or CGRP. |
72
Table of Contents
| • | If approved, OMS103HP will be the first commercially available drug product for the improvement of function following arthroscopic surgery. | |
| • | OMS103HP will provide additional postoperative clinical benefits, including improved range of motion, reduced pain and earlier return to work. | |
| • | OMS103HP selectively targets multiple and discrete pro-inflammatory mediators and pathways within the inflammatory and pain cascade. | |
| • | By delivering OMS103HP to the joint at the initiation of surgical trauma, the inflammatory and pain cascade will be preemptively inhibited. | |
| • | Intra-operative delivery to the joint creates a constant concentration of OMS103HP, bathing and replenishing the joint with drug throughout the duration of the surgical procedure. | |
| • | Because OMS103HP is delivered locally to, and acts directly at, the site of tissue injury, it can be delivered in low concentration, and will not be subject to the substantial interpatient variability in metabolism that is associated with systemic delivery. | |
| • | By delivering low-concentration OMS103HP locally and only during the arthroscopic procedure, systemic absorption of the APIs will be minimized or avoided, thereby reducing the risk of adverse side effects. |
73
Table of Contents
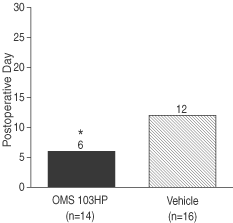
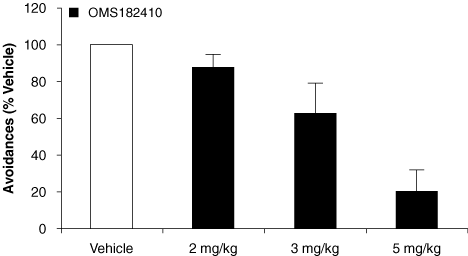
Figure 1: OMS103HP-Treated Patients Required Fewer Median Number of Days to Maximum Passive Flexion³ 90° without Pain | Figure 2: Median Last Day of Continuous Passive Motion Machine Use was Earlier for OMS103HP-Treated Patients | |
 |  | |
| *p = 0.016, log-rank | *p = 0.007, log rank | |
| Figure 1 depicts the median number of days to maximum passive flexion³ 90° without pain, which is a knee range of motion test, as measured in the clinic.† | Figure 2 depicts the number of days until the continuous passive motion, or CPM, machine was discontinued. CPM machines are often used postoperatively to move the knee through a range of motion. CPM usage, recorded in the patient diary, was discontinued at the direction of either the surgeon or rehabilitation therapist based on the patient’s progress, usually at the time the patient reproducibly attained at least 90° of flexion of the operated knee. CPM machine usage was significantly less for OMS103HP.† |
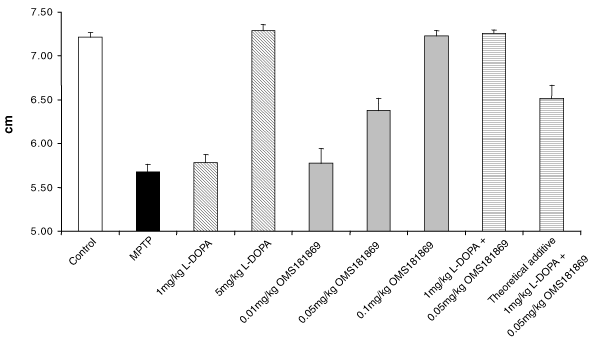
74
Table of Contents
Figure 3: OMS103HP-Treated Patients Demonstrated Better Quadriceps Strength Testing at Day 30 | Figure 4: OMS103HP-Treated Patients Demonstrated Better Hamstring Strength Testing at Day 30 | |
 |  | |
| *p = 0.040, FET | *p = 0.026, FET |
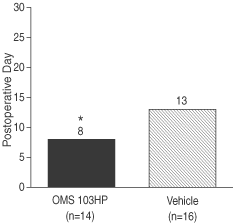
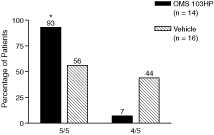
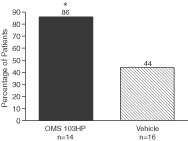
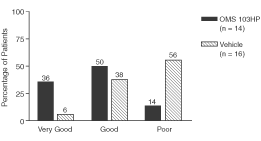
Figure 5: A Greater Percentage of OMS103HP-Treated Patients Demonstrated Successful Recovery of Knee Function as Defined by Knee Function Composite | Figure 6: A Greater Percentage of OMS103HP-Treated Patients Demonstrated Very Good and Good Ratings on the Knee Function Composite—Straight-Leg Raise | |
 |  | |
| *p = 0.026, FET | *p = 0.009, Wilcoxon rank sum test | |
| Figure 5 depicts the study’s primary endpoint, the Knee Function Composite, or KFC. The KFC is composed of the straight-leg raise, one-leg stance, shuttle press, and two-leg squat. Each test is a direct measure of knee function, and all four are routinely used by orthopedic surgeons and rehabilitation therapists to measure improvement in knee function during the early postoperative period following ACL reconstruction surgery. Success on the KFC requires success on all four of the component tests by the end of the30-day evaluation period.† | Very Good: Achievement of the KFC by the end of the 30-day evaluation period and achievement of the highest level of straight-leg raise, or SLR, by the 13th day after surgery Good: Achievement of the KFC by the end of the 30-day evaluation period without achievement of the highest level of SLR by the 13th day after surgery Poor: Failure to achieve the KFC by the end of the 30-day evaluation period | |
| Figure 6 depicts the Knee Function Composite — Straight-Leg Raise, or KFC-SLR, which combines the successful achievement of the KFC with a second key rehabilitation milestone, the ability to perform the highest level of the straight-leg raise by the 13th day after surgery following ACL reconstruction surgery. While the KFC accurately assesses knee function throughout the first 30-day period of postoperative rehabilitation therapy, an evaluation of postoperative function within the first two weeks also is important because early functional return is considered a key driver in successful post-arthroscopy outcomes. Of the four tests comprising the KFC, the straight-leg raise is the most important in the first two weeks following ACL reconstruction because it is used to determine the pace to progress exercises.† |
75
Table of Contents
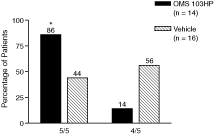
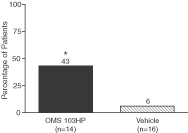
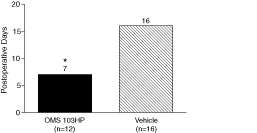
Figure 7: A Greater Percentage of OMS103HP-Treated Patients Achieved Successful Pain Management at Postoperative Week 1 | Figure 8: OMS103HP-Treated Patients Demonstrated a Lower Median Number of Days to Return to Work | |
 |  | |
| *p = 0.031, FET | *p = 0.048; log-rank test | |
| Figure 7 depicts the percentage of patients achieving Successful Pain Management, or SPM, which is a composite of pain assessment and narcotic usage based on data from clinic visits and the patient diary. The SPM composite sets two criteria that the patient must meet in order to be considered a responder. During the first postoperative week, at all clinic visits, the VAS pain score must be not greater than 20 mm with the operated knee at rest. A maximum of two narcotic tablets could be self-administered on each day during the first postoperative week. VAS pain scores of 20 mm or less are considered to be indicative of good to excellent pain control not requiring analgesic medication. The SPM allows pain assessments and narcotic use to be evaluated together, and provides a more complete evaluation of pain management than either VAS pain scores or narcotic usage considered individually because a low VAS pain score recorded by a patient taking high doses of opioid pain medications does not reflect the same level of pain management as that same low VAS pain score recorded in the absence of narcotic pain medications.† | Figure 8 depicts results related to patients’ ability to return to work following ACL reconstruction surgery. Patients were considered to have returned to work if they reported in the patient diary that they had gone to work outside of the home on two consecutive work days excluding weekends and holidays. Return to work was considered to have begun on the first of the two consecutive days. Patients who were unemployed or not working for pay were excluded from the analysis.† |
| † | As published inArthroscopy: The Journal of Arthroscopic and Related Surgery, Vol. 24, No. 6 (June), 2008: pp. 625-636. |
76
Table of Contents
77
Table of Contents
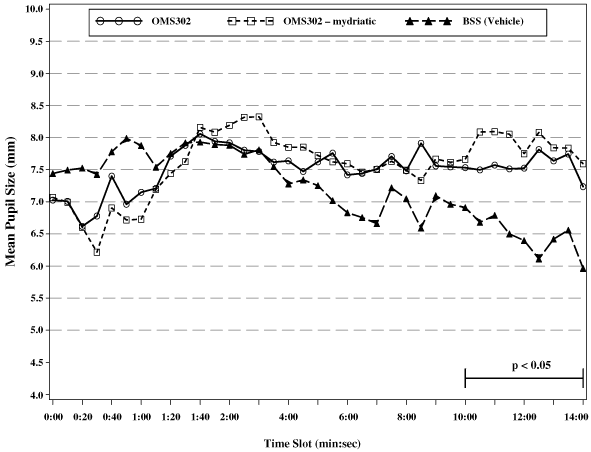
| • | The anti-inflammatory API in OMS302 inhibits miosis by blocking the synthesis of prostaglandins caused by surgical trauma. | |
| • | By delivering OMS302 intra-operatively, inflammation and discomfort will be reduced during the first 24 hours following surgery, the time during which anti-inflammatory topical drops are not commonly administered, as well as after this initial postoperative period. | |
| • | Intra-operative delivery of the mydriatic API in OMS302 will maintain pupil dilation throughout the surgical procedure, decreasing the risk of surgical damage to structures within the eye. | |
| • | Because the mydriatic API in OMS302 maintains pupil dilation, OMS302 will increase the ease of the surgical procedure, thereby increasing patient throughput for both the surgeon and the surgical facility. | |
| • | The mydriatic API in OMS302 prevents intra-operative floppy iris syndrome in many patients taking alpha adrenergic antagonists, such as FLOMAX®. | |
| • | Because OMS302 is delivered intracamerally in standard irrigation solution at a constant, defined concentration, maintaining a more consistent local tissue exposure during the surgical procedure, it will provide superior efficacy relative to topical drug products containing either API. | |
| • | OMS302 is delivered locally to, and acts directly at, the site of tissue injury and, therefore, can be delivered in low concentrations, and will not be subject to the substantial interpatient variability in pharmacokinetics that is associated with systemic delivery. |
78
Table of Contents
79
Table of Contents
80
Table of Contents
81
Table of Contents
| • | By delivering OMS201 intra-operatively, it will reduce inflammation, pain, smooth muscle spasm and lower urinary tract symptoms including frequency, urgency and painful urination, and improve patient outcomes. | |
| • | OMS201 will save health care costs and increase patient comfort by reducing the incidence of ureteral occlusion and the routine need for ureteral stents. | |
| • | By targeting inflammation and smooth muscle spasm, OMS201 will permit surgeons to more frequently place a standard larger-sized UAS, decreasing intra-operative trauma and shortening operative time, thereby saving costs. | |
| • | OMS201 is delivered locally to, and acts directly at, the site of tissue injury and, therefore, can be delivered in low concentrations, and will not be subject to the substantial interpatient variability in pharmacokinetics that is associated with systemic delivery. | |
| • | By delivering OMS201 locally and only during the uroendoscopic procedure, systemic absorption of the APIs will be minimized or avoided, thereby reducing the risk of adverse side effects. |
82
Table of Contents
83
Table of Contents
84
Table of Contents
85
Table of Contents
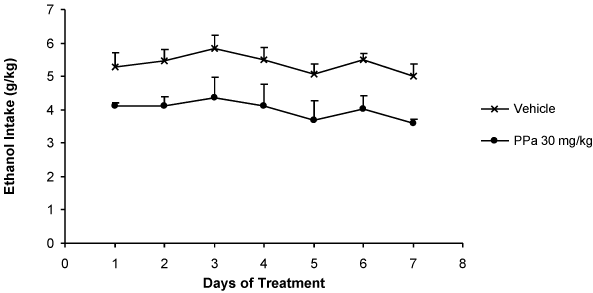
86
Table of Contents
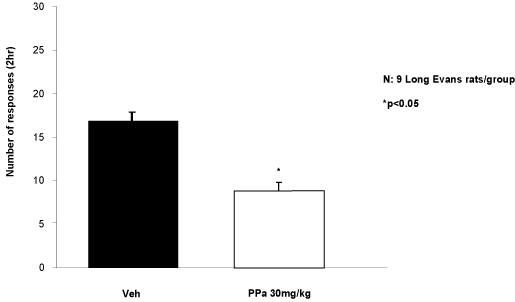
87
Table of Contents
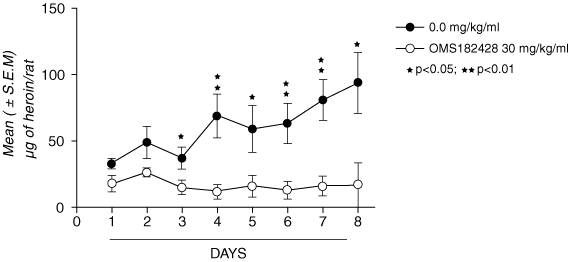
88
Table of Contents
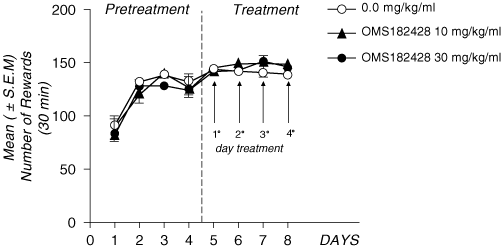
89
Table of Contents
90
Table of Contents
91
Table of Contents
92
Table of Contents
93
Table of Contents
94
Table of Contents
95
Table of Contents
96
Table of Contents
97
Table of Contents
| • | develop and market products that are less expensive, more effective or safer than our future products; | |
| • | commercialize competing products before we can launch any products developed from our product candidates; | |
| • | operate larger research and development programs, possess greater manufacturing capabilities or have substantially greater financial resources than we do; | |
| • | initiate or withstand substantial price competition more successfully than we can; | |
| • | have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; | |
| • | more effectively negotiate third-party licenses and strategic relationships; and | |
| • | take advantage of acquisition or other opportunities more readily than we can. |
98
Table of Contents
| • | OMS103HP — Arthroscopy. OMS103HP is protected by our PharmacoSurgery patent portfolio. The relevant patents and patent applications in this portfolio cover combinations of agents, genericand/or proprietary to us or others, drawn from therapeutic classes such as pain and inflammation inhibitory agents and vasoconstrictive agents, delivered locally and intra-operatively to the site of medical or surgical procedures, including arthroscopy. We currently own four issued U.S. Patents, two pending U.S. Patent Applications, and 12 issued patents and 9 pending patent applications in foreign markets (Australia, Brazil, Canada, China, Europe, Hong Kong, Japan, Mexico, Norway, Russia, Singapore and South Korea) that cover OMS103HP. |
| • | OMS302 — Ophthalmology. OMS302 is protected by our PharmacoSurgery patent portfolio. The relevant patents and patent applications in this portfolio cover combinations of agents, genericand/or proprietary to us or others, drawn from therapeutic classes such as pain and inflammation inhibitory agents, mydriatic agents and agents that reduce intraocular pressure, delivered locally and intra-operatively to the site of ophthalmological procedures, including cataract and lens replacement surgery. We currently own two pending U.S. Patent Applications and eight pending patent applications in foreign markets (Australia, Canada, China, Europe, Hong Kong and Japan) that cover OMS302. |
| • | OMS201 — Urology. OMS201 is protected by our PharmacoSurgery patent portfolio. The relevant patents and patent applications in this portfolio cover combinations of agents, genericand/or proprietary to us or others, drawn from therapeutic classes such as pain and inflammation inhibitory agents and spasm inhibitory agents, delivered locally and intra-operatively to the site of medical or surgical procedures, including uroendoscopy. We currently own three issued U.S. Patents, two pending U.S. Patent Applications, and an additional 10 issued patents and 16 pending patent applications in foreign markets (Australia, Brazil, Canada, China, Europe, Hong Kong, India, Japan, Mexico, Norway, Russia, Singapore and South Korea) that cover OMS201. | |
| • | MASP-2 Program. We hold worldwide exclusive licenses to rights in connection with MASP-2, the antibodies targeting MASP-2 and the therapeutic applications for those antibodies from the University of Leicester and from its collaborator, Medical Research Council at Oxford University. These licenses include what we believe to be each institution’s joint ownership rights in patent applications and patents related to MASP-2 antibodies initially filed by researchers at Aarhus Universitet, Denmark. We currently |
99
Table of Contents
| exclusively control four pending U.S. Patent Applications and 21 pending patent applications in foreign markets (Australia, Brazil, Canada, China, Hong Kong, Europe, India, Indonesia, Japan, Mexico, New Zealand, Russia and South Korea) related to our MASP-2 program. |
| • | Addiction Program. We own three pending U.S. Patent Applications and a pending International Patent Cooperation Treaty, or PCT, Patent Application directed to the previously unknown link between PPARγ and addictive disorders. |
| • | PDE10 Program. Medicinal chemistry developments in our PDE10 program have resulted in four pending U.S. and two pending PCT Patent Applications that claim what we believe to be novel chemical structures, as well as claiming the use of a broader set, or genus, of chemical structures as inhibitors of PDE10 for the treatment of schizophrenia and other psychotic disorders. |
| • | PDE7 Program. We own two pending U.S. Patent Applications and a pending international PCT Patent Application directed to the previously unknown link between PDE7 and movement disorders. | |
| • | GPCR Program. We own one issued U.S. Patent, three pending U.S. Patent Applications, one international PCT Patent Application and an additional two issued patents and four pending patent applications in foreign markets (Australia, Canada, Europe and Japan), which are directed to previously unknown links between specific molecular targets in the brain and a series of CNS disorders, and to research tools that are used in our GPCR program. |
| • | PharmacoSurgery Platform. Our scientific co-founders, Gregory A. Demopulos, M.D. and Pamela Pierce Palmer, M.D., Ph.D., conceived the initial invention underlying our PharmacoSurgery platform and transferred all of their related intellectual property rights to us in 1994. Other than their rights as shareholders, our co-founders have not retained any rights to our PharmacoSurgery platform, except that if we file for liquidation under |
100
Table of Contents
| Chapter 7 of the U.S. Bankruptcy Act or voluntarily liquidate or dissolve, other than in connection with a merger, reorganization, consolidation or sale of assets, ourco-founders have the right to repurchase the initial PharmacoSurgery intellectual property at the then-current fair market value. Subsequent developments of the PharmacoSurgery intellectual property were assigned to us by Dr. Demopulos, Dr. Palmer and other of our employees and consultants, without restriction. |
| • | MASP-2 Program. We hold worldwide exclusive licenses to rights related to MASP-2, the antibodies targeting MASP-2 and the therapeutic applications for the antibodies from the University of Leicester and from its collaborator, Medical Research Council at Oxford University, or MRC. Concurrent with execution of the license agreement with the University of Leicester, two provisional US Patent Applications directed to methods of treating conditions associated with complement activation by inhibiting MASP-2 or a related protein, and a British application directed to MASP-2 knock-out mice, were filed. Exclusive licenses to these three initial patent applications were conveyed to us by the University of Leicester license agreement. Under the terms of the University of Leicester and MRC license agreements, we have agreed to pay royalties to each of the University of Leicester and MRC based on a percentage of any proceeds we receive from the licensed technology during the terms of the agreements. We must pay low single-digit royalties with respect to proceeds that we receive from products incorporating the licensed technology that are used, manufactured, directly sold or directly distributed by us, and we must pay royalties, initially in the range of low single-digit to low double-digit and decreasing over time to low single-digit, with respect to proceeds we receive from sublicense royalties or fees that we receive from third parties to which we grant sublicenses to the licensed technology. We may also sponsor research of MASP-2 by these institutions and retain worldwide exclusive licenses from these institutions to develop and commercialize any intellectual property rights developed in the sponsored research. The term of each license agreement ends when there are no longer any pending patent applications, applications in preparation or unexpired issued patents related to any of the intellectual property rights we are licensing under the agreement. Both of these license agreements may be terminated prior to the end of their terms by us for convenience or by a party if the other party (1) breaches any material obligation under the agreement and does not cure such breach after notice and an opportunity to cure or (2) is declared or adjudged to be insolvent, bankrupt or in receivership and materially limited from performing its obligations under the agreement. |
| • | Addiction Program. We acquired the patent applications and related intellectual property rights for our Addiction program in 2009 from Roberto Ciccocioppo, Ph.D. of the Università di Camerino, Italy, pursuant to a Patent Assignment Agreement. We have agreed to pay Dr. Ciccocioppo royalties and milestone payments related to any products that are covered by the patents we acquired from him. For a more detailed description of this agreement, see “Business — Our Product Candidates and Development Programs — Addiction Program.” |
| • | PDE10, PDE7 and GPCR Programs. We acquired our PDE10, PDE7 and GPCR programs and some of our related patents and other intellectual property rights as a result of our acquisition of nura, inc. in August 2006 for an aggregate purchase price of $14.4 million. We hold an exclusive option to purchase the CRA for our GPCR program from Patobios Limited for approximately $10.8 million CAD. Our exclusive option with Patobios ends on December 4, 2009, provided that we have the right to extend our option for one additional six-month period ending June 4, 2010 by paying Patobios $650,000 CAD. |
101
Table of Contents
| • | preclinical laboratory and animal tests, and formulation studies; | |
| • | submission to the FDA of an Investigational New Drug Application, or IND, for human clinical testing, which must become effective before human clinical trials may begin in the United States; | |
| • | adequate and well-controlled human clinical trials to establish the efficacy and safety of the product candidate for each indication for which approval is sought; | |
| • | submission to the FDA of a New Drug Application, or NDA; | |
| • | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the drug is produced to assess compliance with cGMP; and | |
| • | FDA review and approval of an NDA. |
102
Table of Contents
| • | Phase 1 usually involves the initial administration of the investigational drug product to human subjects to evaluate its safety, dosage tolerance, pharmacodynamics and, if possible, to gain an early indication of its effectiveness. | |
| • | Phase 2 usually involves trials in a limited patient population, with the disease or condition for which the product candidate is being developed, to evaluate dosage tolerance and appropriate dosage, identify possible adverse side effects and safety risks, and preliminarily evaluate the effectiveness of the drug for specific indications. | |
| • | Phase 3 trials usually further evaluate effectiveness and test further for safety by administering the drug in its final form in an expanded patient population. |
103
Table of Contents
104
Table of Contents
| Name | Age | Position(s) | ||||
Executive Officers: | ||||||
| Gregory A. Demopulos, M.D. | 50 | President, Chief Executive Officer, Chief Medical Officer and Chairman of the Board of Directors and, in an interim capacity, Chief Financial Officer and Treasurer | ||||
| Marcia S. Kelbon, Esq. | 49 | Vice President, Patent and General Counsel and Secretary | ||||
Key Employees: | ||||||
| George A. Gaitanaris, M.D., Ph.D. | 52 | Vice President, Science | ||||
| Wayne R. Gombotz, Ph.D. | 50 | Vice President, Pharmaceutical Operations | ||||
| Stephen R. Murray, M.D., Ph.D. | 46 | Vice President, Clinical Development | ||||
| J. Greg Perkins, Ph.D. | 64 | Vice President, Regulatory Affairs and Quality Systems | ||||
| Clark E. Tedford, Ph.D. | 50 | Vice President, Research | ||||
Directors: | ||||||
| Ray Aspiri (2) | 73 | Director | ||||
| Thomas J. Cable (1)(2) | 69 | Director | ||||
| Peter A. Demopulos, M.D., FACC | 55 | Director | ||||
| Leroy E. Hood, M.D, Ph.D. | 70 | Director | ||||
| Jean-Philippe Tripet (1) | 46 | Director | ||||
| (1) | Member of our audit committee. | |
| (2) | Member of our compensation committee. | |
| (3) | Member of our nominating and corporate governance committee. |
105
Table of Contents
106
Table of Contents
| • | Class I, which will consist of Ray Aspiri and Jean-Philippe Tripet, and whose term will expire at our first annual meeting of shareholders to be held following the completion of this offering; | |
| • | Class II, which will consist of Thomas J. Cable and Peter A. Demopulos, M.D., and whose term will expire at our second annual meeting of shareholders to be held following the completion of this offering; and | |
| • | Class III, which will consist of Gregory A. Demopulos, M.D. and Leroy E. Hood, M.D., Ph.D., and whose term will expire at our third annual meeting of shareholders to be held following the completion of this offering. |
107
Table of Contents
| • | selecting and hiring our independent auditors, and approving the audit and non-audit services to be performed by our independent registered public accounting firm; | |
| • | evaluating the qualifications, performance and independence of our independent registered public accounting firm; | |
| • | monitoring the integrity of our financial statements and our compliance with legal and regulatory requirements as they relate to financial statements or accounting matters; | |
| • | reviewing with our independent registered public accounting firm and management significant issues that arise regarding accounting principles and financial statement presentation, and matters concerning the scope, adequacy and effectiveness of our financial controls; | |
| • | reviewing the adequacy and effectiveness of our internal control policies and procedures; | |
| • | establishing procedures for the receipt, retention and treatment of complaints received by us regarding accounting, internal accounting controls or auditing matters; | |
| • | reviewing and approving in advance any proposed related-party transactions and monitoring compliance with our code of business conduct and ethics; and | |
| • | preparing the audit committee report that the SEC requires in our annual proxy statement. |
108
Table of Contents
| • | evaluating and recommending to our board of directors the compensation and other terms of employment of our executive officers and reviewing and approving corporate performance goals and objectives relevant to such compensation; | |
| • | evaluating and recommending to our board of directors the type and amount of compensation to be paid or awarded to board members; | |
| • | evaluating and recommending to our board of directors the equity incentive plans, compensation plans and similar programs advisable for us; | |
| • | administering our equity incentive plans; | |
| • | reviewing and approving the terms of any employment agreements, severance arrangements, change in control protections and any other compensatory arrangements for our executive officers; and | |
| • | preparing the compensation committee report that the SEC requires in our annual proxy statement. |
| • | assisting the board in identifying prospective director nominees and recommending director nominees to our board for each annual meeting of shareholders; | |
| • | evaluating nominations by shareholders of candidates for election to our board; | |
| • | recommending governance principles to our board; | |
| • | overseeing the evaluation of our board of directors and management; | |
| • | reviewing shareholder proposals for our annual meetings; | |
| • | evaluating proposed changes to our charter documents and board committee charters; | |
| • | reviewing and assessing our senior management succession plan; and | |
| • | recommending to our board the members for each board committee. |
109
Table of Contents
| Option Awards | Total | |||||||
Name | ($)(1) (2)(3) | ($) | ||||||
| Ray Aspiri | — | — | ||||||
| Thomas J. Cable | — | — | ||||||
| Peter A. Demopulos, M.D. | — | — | ||||||
| Leroy E. Hood, M.D, Ph.D. | — | — | ||||||
| David A. Mann | 45,599 | 45,599 | ||||||
| Jean-Philippe Tripet | — | — | ||||||
| (1) | Our directors did not receive any cash compensation during 2008. Amounts shown in this column represent the compensation cost for the year ended December 31, 2008 of option awards granted to each of our non-employee directors as determined in accordance with Statement of Financial Accounting Standards No. 123(revised), or SFAS 123R, using the Black-Scholes option valuation model. The assumptions used to calculate the value of option awards are set forth in Note 11 to our consolidated financial statements included elsewhere in this prospectus. Pursuant to SEC rules, the amounts shown exclude the impact of estimated forfeiture related to service-based vesting conditions. | |
| (2) | As of December 31, 2008, Mr. Mann held an option award to purchase 25,000 shares of our common stock with an exercise price of $1.25 per share that vested over a three-year period in equal annual installments. Mr. Mann exercised this option award for 8,333 shares of our common stock in January 2009. Mr. Mann resigned from our board of directors in March 2009. | |
| (3) | As of December 31, 2008, Mr. Aspiri, Mr. Cable and Dr. Hood held option awards to purchase 30,000, 45,000 and 50,000 shares of our common stock, respectively. All of these option awards were fully vested and exercisable as of December 31, 2008. |
110
Table of Contents
111
Table of Contents
112
Table of Contents
| Option | All Other | |||||||||||||||||||
| Salary | Awards | Compensation | Total | |||||||||||||||||
Name and Principal Position | Year | ($) | ($) (1) | ($) | ($) | |||||||||||||||
| Gregory A. Demopulos, M.D. | 2008 | 475,000 | 594,203 | 25,225 | (2) | 1,094,428 | ||||||||||||||
| President, Chief Executive Officer, Chief Medical Officer and Chairman of the Board of Directors | ||||||||||||||||||||
| Marcia S. Kelbon, Esq. | 2008 | 285,000 | 67,706 | 3,049 | 355,755 | |||||||||||||||
| Vice President, Patent and General Counsel and Secretary | ||||||||||||||||||||
| Richard J. Klein (3) | 2008 | 250,000 | 202,577 | 4,092 | 456,669 | |||||||||||||||
| Chief Financial Officer and Treasurer | ||||||||||||||||||||
| (1) | Amounts shown do not reflect compensation actually received by the named executive officers. Instead, the dollar amounts shown in this column represent the compensation cost for the year ended December 31, 2008 of option awards granted to each of our named executive officers as determined pursuant to SFAS 123R using the Black-Scholes option valuation model. The assumptions used to calculate the value of option awards are set forth in Note 11 to our consolidated financial statements included elsewhere in this prospectus. Pursuant to SEC rules, the amounts shown exclude the impact of estimated forfeiture related to service-based vesting conditions. | |
| (2) | Represents $25,088 in perquisites and other personal benefits, which included payments for medical malpractice insurance, parking expenses, legal fees, medical practice fees and travel expenses, and $137 in life insurance premiums. | |
| (3) | Mr. Klein’s employment with us ended in January 2009. |
113
Table of Contents
114
Table of Contents
| • | the annual base salary he was receiving as of his termination, provided that if he terminates his employment for good reason because of a reduction in his annual base salary, then the annual base salary we will be obligated to pay him will be his annual base salary in effect prior to such reduction; plus | |
| • | the greater of (1) the average annual bonus he received in the preceding two calendar years and (2) any bonus he would have been entitled to in the year of his termination as determined by our board of directors in good faith. |
115
Table of Contents
| • | his willful misconduct or gross negligence in performance of his duties, including his refusal to comply in any material respect with the legal directives of our board of directors so long as such directives are not inconsistent with his position and duties, and such refusal to comply is not remedied within ten working days after written notice from the board of directors; | |
| • | dishonest or fraudulent conduct that materially discredits us, a deliberate attempt to do an injury to us, or conduct that materially discredits us or is materially detrimental to the reputation of us, including conviction of a felony; or | |
| • | his material breach, if incurable, of any element of his confidential information and invention assignment agreement with us, including without limitation, his theft or other misappropriation of our proprietary information. |
| • | any material diminution in his authority, duties or responsibilities; | |
| • | any material diminution in his base salary; | |
| • | we relocate his principal work location to a place that is more than 50 miles from our current location; or | |
| • | we materially breach his employment agreement, which may include, for example, our failure to enter into a new employment agreement by May 1, 2009 because of our actions or omissions. |
116
Table of Contents
| • | a “change in control” means proposed sale of all or substantially all of the assets of us, or the merger of us with or into another corporation, or other change in control; | |
| • | a termination for “cause” means a termination of an employee for any of the following reasons: (1) his or her willful failure to substantially perform his or her duties and responsibilities to us or a deliberate violation of a company policy; (2) his or her commission of any act of fraud, embezzlement, dishonesty or any other willful misconduct that has caused or is reasonably expected to result in material injury to us; (3) unauthorized use or disclosure by him or her of any proprietary information or trade secrets of ours or any other party to whom he or she owes an obligation of nondisclosure as a result of his or her relationship with us; or (4) his or her willful breach of any of his or her obligations under any written agreement or covenant with us; and | |
| • | a “constructive termination” means the occurrence of any of the following events: (1) there is a material adverse change in an employee’s position causing such position to be of materially reduced stature or responsibility; (2) a reduction of more than 30% of an employee’s base compensation unless in connection with similar decreases of other similarly situated employees; or (3) an employee’s refusal to comply with our request to relocate to a facility or location more than 50 miles from our current location; provided that in order for an employee to be constructively terminated, he or she must voluntarily terminate his or her employment within 30 days of the applicable material change or reduction. |
117
Table of Contents
| Successor in | Successor in | Employee is Terminated | ||||||||||
| Change in Control | Change in Control | Without Cause or | ||||||||||
| Assumes or | does not Assume | Constructively Terminated | ||||||||||
| Replaces Option | or Replace Option | within Twelve Months of | ||||||||||
Name | Awards ($) | Awards ($) | Change in Control ($) | |||||||||
| Gregory A. Demopulos, M.D. | ||||||||||||
| Marcia S. Kelbon, Esq. | ||||||||||||
| Richard J. Klein | ||||||||||||
118
Table of Contents
119
Table of Contents
120
Table of Contents
| • | five percent of the outstanding shares of our common stock on the last day of the immediately preceding fiscal year; | |
| • | 3,500,000 shares; and | |
| • | such other amount as our board of directors may determine. |
121
Table of Contents
122
Table of Contents
| Option Awards | Stock Awards | |||||||||||||||||||||||
| Number of | ||||||||||||||||||||||||
| Number of | Securities | |||||||||||||||||||||||
| Securities | Underlying | Number of | Market Value of | |||||||||||||||||||||
| Underlying | Unexercised | Option | Shares or Units | Shares or Units | ||||||||||||||||||||
| Unexercised | Options | Exercise | Option | of Stock That | of Stock That | |||||||||||||||||||
| Options | (#) | Price | Expiration | Have Not | Have Not | |||||||||||||||||||
Name | (#) Exercisable | Unexercisable(1) | ($) | Date | Vested (#) | Vested ($)(2) | ||||||||||||||||||
| Gregory A. Demopulos, M.D. | 3,025 | — | 0.265 | 12/10/11 | — | — | ||||||||||||||||||
| 766,666 | 33,334 | (3) | 0.50 | 12/11/16 | — | — | ||||||||||||||||||
| 1,150,000 | 50,000 | (3) | 0.50 | 12/11/16 | — | — | ||||||||||||||||||
| 50,000 | 150,000 | (4) | 1.25 | 12/29/17 | — | — | ||||||||||||||||||
| Marcia S. Kelbon, Esq. | 300,833 | 79,167 | (5) | 0.50 | 12/11/16 | — | — | |||||||||||||||||
| 2,500 | 7,500 | (4) | 1.25 | 12/29/17 | — | — | ||||||||||||||||||
| Richard J. Klein | 100,000 | (6) | — | 1.00 | 05/13/17 | 51,042 | (6) | |||||||||||||||||
| 2,500 | 7,500 | (4) | 1.25 | 12/29/17 | — | — | ||||||||||||||||||
| (1) | These option awards were granted pursuant to the 1998 Stock Plan, which provides for the automatic vesting of at least a portion of any unvested options upon a change of control transaction as described under the section of this prospectus entitled “Management — Employee Benefit Plans — Second Amended and Restated 1998 Stock Option Plan.” | |
| (2) | The market value of shares of stock that have not vested has been calculated using the assumed initial public offering price of $ per share (the mid-point of the range set forth on the cover page of this prospectus). | |
| (3) | The shares subject to the option award vest on a monthly basis in equal amounts over a four-year period that began on February 28, 2005. | |
| (4) | 1/4th of the shares subject to the option award vest on December 30, 2008 and 1/48th of the shares subject to the option award vest each month thereafter. |
123
Table of Contents
| (5) | The shares subject to the option award vest on a monthly basis in equal amounts over a four-year period that began on October 1, 2005. | |
| (6) | A total of 250,000 shares were subject to this option award. 1/4th of the shares subject to the option vested on May 14, 2008 and 1/48th of the shares vested each month thereafter. Pursuant to the terms of the option award, Mr. Klein had the right to purchase unvested shares, provided that if his employment terminated for any reason prior to him vesting into any shares that he exercised, we have the right, but not the obligation, to repurchase at the original purchase price any shares that he exercised and is not vested in as of the date of his termination. As of December 31, 2008, Mr. Klein had purchased 150,000 of these shares, 98,958 of which were vested. Mr. Klein’s employment with us ended in January 2009, at which time 104,166 of these shares were vested. Our right to repurchase the unvested shares ends in July 2009. |
| Stock Vested | ||||||||
| Number of | ||||||||
| Shares Acquired | Value Realized | |||||||
| on Vesting | on Vesting | |||||||
Name | (#) | (#)(1) | ||||||
| Gregory A. Demopulos, M.D. | — | — | ||||||
| Marcia S. Kelbon, Esq. | — | — | ||||||
| Richard J. Klein | 98,958 | |||||||
| (1) | The value realized on vesting has been calculated using the assumed initial public offering price of $ per share (the mid-point of the range set forth on the cover page of this prospectus). |
| • | acts or omissions that involve intentional misconduct or a knowing violation of law; | |
| • | unlawful distributions; or | |
| • | any transaction from which the director will personally receive a benefit in money, property or services to which the director is not legally entitled. |
124
Table of Contents
125
Table of Contents
| Series E Convertible | ||||||||
| Preferred Stock | Common Stock | |||||||
Name | (#) | (#) | ||||||
| Aravis Venture I, L.P.(1) | 559,551 | 6,925 | ||||||
| Entities affiliated with ARCH Venture Partners (2) | 839,326 | 7,741 | ||||||
| (1) | Jean-Philippe Tripet, a member of our board of directors, is managing partner of Aravis Venture I, L.P. Mr. Tripet holds the title of Director of Aravis General Partner Ltd., which serves as general partner of Aravis Venture I, L.P. |
126
Table of Contents
| Mr. Tripet disclaims beneficial ownership of the shares held by Aravis Venture I, L.P., except to the extent of his proportionate pecuniary interest therein. |
| (2) | Represents (a) 833,787 and 7,690 shares of Series E convertible preferred stock and common stock, respectively, held by ARCH Venture Fund V, L.P. and (b) 5,539 and 51 shares of Series E convertible preferred stock and common stock, respectively, held by ARCH V Entrepreneurs Fund, L.P. These two associated partnerships together hold more than five percent of our capital stock. |
| Series E Convertible | Aggregate Purchase | |||||||
| Preferred Stock | Price | |||||||
Name | (#) | ($) | ||||||
| Aravis Venture I, L.P. | 400,000 | 2,000,000 | ||||||
| Entities affiliated with ARCH Venture Partners (1) | 600,000 | 3,000,000 | ||||||
| (1) | Represents 595,984 and 4,016 shares of Series E convertible preferred stock that we issued and sold to ARCH Venture Fund V, L.P. and ARCH V Entrepreneurs Fund, L.P., respectively. |
127
Table of Contents
128
Table of Contents
| • | each person who we know beneficially owns more than five percent of our common stock; | |
| • | each of our directors; | |
| • | each of our named executive officers; and | |
| • | all of our directors and executive officers as a group. |
| Number of | Percentage of Shares | |||||||||||
| Shares | Beneficially Owned | |||||||||||
| Beneficially | Before | |||||||||||
Name of Beneficial Owner | Owned | Offering | After Offering | |||||||||
5% Shareholders: | ||||||||||||
| Entities affiliated with ARCH Venture Partners (1) | 1,447,067 | 5.1 | % | |||||||||
Directors and Executive Officers: | ||||||||||||
| Gregory A. Demopulos, M.D. (2) | 4,965,396 | 16.3 | % | |||||||||
| Marcia S. Kelbon, Esq. (3) | 553,957 | 1.9 | % | |||||||||
| Richard J. Klein (4) | 152,916 | * | ||||||||||
| Ray Aspiri (5) | 317,857 | 1.1 | % | |||||||||
| Thomas J. Cable (6) | 194,163 | * | ||||||||||
| Peter A. Demopulos, M.D., FACC (7) | 517,045 | 1.8 | % | |||||||||
| Leroy E. Hood, M.D., Ph.D. (8) | 106,603 | * | ||||||||||
| Jean-Philippe Tripet (9) | 966,476 | 3.4 | % | |||||||||
| All executive officers and directors as a group (8 persons) (10) | 7,774,413 | 25.2 | % | |||||||||
| * | Less than one percent |
| (1) | Represents (a) 1,437,461 shares of common stock held by ARCH Venture Fund V, L.P., or ARCH V, and (b) 9,606 shares of common stock held by ARCH V Entrepreneurs Fund, L.P., or the Entrepreneurs Fund. ARCH Venture Partners V, L.P., or the GPLP, as the sole general partner of ARCH V and the Entrepreneurs Fund, has |
129
Table of Contents
| the power to vote and dispose of the shares held of record by ARCH V and the Entrepreneurs Fund and may be deemed to beneficially own certain of the shares held of record by ARCH V and the Entrepreneurs Fund. The GPLP disclaims beneficial ownership of all shares held of record by ARCH V and the Entrepreneurs Fund in which the GPLP does not have an actual pecuniary interest. ARCH Venture Partners V, LLC, or the GPLLC, as the sole general partner of the GPLP, has the power to vote and dispose of the shares held of record by ARCH V and the Entrepreneurs Fund and may be deemed to beneficially own certain of the shares held of record by ARCH V and the Entrepreneurs Fund. The GPLLC disclaims beneficial ownership of all shares held of record by ARCH V and the Entrepreneurs Fund in which it does not have an actual pecuniary interest. Keith Crandell, Steven Lazarus, Clinton Bybee and Robert Nelsen are the managing directors of the GPLLC, share the power to vote and dispose of the shares held of record by ARCH V and the Entrepreneurs Fund and may be deemed to beneficially own certain of the shares held of record by ARCH V and the Entrepreneurs Fund. The managing directors disclaim beneficial ownership of all shares held of record by ARCH V and the Entrepreneurs Fund in which they do not have an actual pecuniary interest. The address of all filing persons is 8725 W. Higgins Road, Suite 290, Chicago, IL 60631. |
| (2) | Includes 2,073,858 shares of common stock that Dr. Demopulos has the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. | |
| (3) | Includes 343,957 shares of common stock that Ms. Kelbon has the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. | |
| (4) | Represents (a) 150,000 shares of common stock that Mr. Klein acquired from us pursuant to the exercise of an option award and (b) 2,916 shares of common stock that Mr. Klein had the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. Pursuant to the terms of one of his option awards, Mr. Klein had the right to exercise that option award for shares that he was not vested in. When Mr. Klein’s employment with us ended in January 2009, he held 150,000 shares of common stock that he purchased by exercising that option award, 104,166 shares of which were vested. Pursuant to the term of the option award, we have the right, but not the obligation, to repurchase the 45,834 shares that he had exercised but not vested in as of the date of his termination at a cost of $1.00 per share. Our right to repurchase the unvested shares ends in July 2009. See “Management — Executive Compensation — Executive Employment Agreements — Richard J. Klein” for a description of the vesting terms of Mr. Klein’s option awards. | |
| (5) | Represents (a) 30,000 shares of common stock that Mr. Aspiri has the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards and (b) 287,857 shares of common stock held by Aspiri Enterprises LLC. Mr. Aspiri is the managing partner and a member of Aspiri Enterprises LLC. | |
| (6) | Includes 45,000 shares of common stock that Mr. Cable has the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. | |
| (7) | Includes 322,188 shares of common stock held by the Demopulos Family Trust, of which Dr. Peter A. Demopulos is the trustee and a beneficiary along with his mother and sister. Dr. Peter A. Demopulos disclaims beneficial ownership of the shares held by the Demopulos Family Trust except to the extent of his pecuniary interest therein. | |
| (8) | Includes 50,000 shares of common stock that Dr. Hood has the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. | |
| (9) | Represents 966,476 shares of common stock held by Aravis Venture I, L.P. Mr. Tripet holds the title of director of Aravis General Partner Ltd., which serves as general partner of Aravis Venture I, L.P. Mr. Tripet disclaims beneficial ownership of the shares held by Aravis Venture I, L.P., except to the extent of his proportionate pecuniary interest therein. | |
| (10) | Includes 2,545,731 shares of common stock that our executive officers and directors have the right to acquire from us within 60 days of March 31, 2009 pursuant to the exercise of option awards. |
130
Table of Contents
| • | 150,000,000 shares will be designated as common stock; and | |
| • | 20,000,000 shares will be designated as preferred stock. |
131
Table of Contents
| • | A warrant that we assumed in connection with our acquisition of nura on August 11, 2006 to purchase 22,613 shares of our common stock with an exercise price of $4.66 per share. This warrant will terminate upon the earlier of (a) April 26, 2015 and (b) certain acquisitions of us as described in the warrant. | |
| • | Warrants issued on March 29, 2007 to purchase an aggregate of 387,030 shares of our common stock with an exercise price of $6.25 per share. If not exercised, these warrants will terminate on the earlier of (a) completion of this offering, (b) a change of control as defined in the warrants and (c) March 28, 2012. | |
| • | Warrants that we issued on September 12, 2008 to purchase up to an aggregate of 58,136 shares of our common stock with an exercise price of $6.88 per share in connection with loans we received from BlueCrest Venture Finance Master Fund Limited. As of March 31, 2009, 49,416 shares of common stock subject to these warrants were vested and the remaining 8,720 shares were not vested. The 8,720 shares of common stock would have vested only if we borrowed additional amounts from Blue Crest on or before March 31, 2009. Because we did not borrow those additional amounts on or before March 31, 2009, these 8,720 shares will not vest. If not exercised, the warrants will terminate on the earlier of (a) completion of this offering, (b) a change of control as defined in the warrants and (c) September 12, 2018. |
132
Table of Contents
133
Table of Contents
134
Table of Contents
| • | a merger or share exchange with, disposition of assets to, or issuance or redemption of stock to or from, the acquiring person; | |
| • | a termination of five percent or more of the employees of the target corporation as a result of the acquiring person’s acquisition of 10% or more of the shares; or | |
| • | a transaction in which the acquiring person is allowed to receive a disproportionate benefit as a shareholder. |
135
Table of Contents
Date | Number of Shares | |||
| On the date of this prospectus | ||||
| Between 90 and 180 days after the date of this prospectus | ||||
| At various times beginning more than 180 days after the date of this prospectus | ||||
| • | one percent of the number of shares of common stock then outstanding, which will equal approximately shares immediately after the offering; and | |
| • | the average weekly trading volume of the common stock during the four calendar weeks preceding the filing of a notice on Form 144 with respect to such sale. |
136
Table of Contents
| • | during the last 17 days of the 180-day restricted period we issue an earnings release or material news, or a material event relating to us occurs; or | |
| • | prior to the expiration of the 180-day restricted period we announce that we will release earnings results during the 16-day period following the last day of the 180-day period, |
137
Table of Contents
138
Table of Contents
Underwriter | Number of Shares | |||
| Deutsche Bank Securities Inc. | ||||
| Wedbush Morgan Securities, Inc. | ||||
| Leerink Swann LLC | ||||
| Needham & Company, LLC | ||||
| Total | ||||
| Total Fees | ||||||||||||
| Without Exercise of | With Full Exercise | |||||||||||
| Fee per | Over-Allotment | of Over-Allotment | ||||||||||
| share | Option | Option | ||||||||||
| Discounts and commissions paid by us | $ | $ | $ | |||||||||
139
Table of Contents
| • | during the last 17 days of the 180-day restricted period we issue an earnings release or material news, or a material event relating to us occurs; or | |
| • | prior to the expiration of the 180-day restricted period we announce that we will release earnings results during the 16-day period following the last day of the 180-day period, |
140
Table of Contents
141
Table of Contents
| • | prevailing market conditions; | |
| • | our results of operations in recent periods; | |
| • | the present stage of our development; | |
| • | the market capitalizations and stages of development of other companies that we and the representative of the underwriters believe to be comparable to our business; and | |
| • | estimates of our business potential. |
142
Table of Contents
FORNON-UNITED STATES HOLDERS OF COMMON STOCK
143
Table of Contents
| • | the gain is effectively connected with the conduct by thenon-United States holder of a U.S. trade or business (in which case the special rules described below apply); | |
| • | thenon-U.S. holder is an individual who is present in the United States for 183 days or more in the taxable year of the sale, exchange or other disposition of our common stock, and certain other requirements are met; | |
| • | thenon-United States holder was a citizen or resident of the United States and thus is subject to special rules that apply to expatriates; or | |
| • | the rules of the Foreign Investment in Real Property Tax Act, or FIRPTA, treat the gain as effectively connected with a U.S. trade or business. |
144
Table of Contents
145
Table of Contents
146
Table of Contents
| Page | ||||
OMEROS CORPORATION | ||||
| Report of Independent Registered Public Accounting Firm | F-2 | |||
| Consolidated Balance Sheets | F-3 | |||
| Consolidated Statements of Operations | F-5 | |||
| Consolidated Statements of Convertible Preferred Stock and Shareholders’ Equity (Deficit) | F-7 | |||
| Consolidated Statements of Cash Flows | F-14 | |||
| Notes to Consolidated Financial Statements | F-16 | |||
F-1
Table of Contents
F-2
Table of Contents
(A Development Stage Company)
| March 31, | December 31, | |||||||||||
| 2009 | 2008 | 2007 | ||||||||||
| Assets | (unaudited) | |||||||||||
| Current assets: | ||||||||||||
| Cash and cash equivalents | $ | 9,899 | $ | 12,726 | $ | 5,925 | ||||||
| Short-term investments | 6,919 | 7,256 | 18,157 | |||||||||
| Grant and other receivables | 235 | 207 | 190 | |||||||||
| Prepaid expenses and other current assets | 436 | 289 | 189 | |||||||||
| Total current assets | 17,489 | 20,478 | 24,461 | |||||||||
| Deferred offering costs | 204 | — | 1,462 | |||||||||
| Property and equipment, net | 830 | 918 | 839 | |||||||||
| Intangible assets, net | 34 | 60 | 164 | |||||||||
| Restricted cash | 193 | 193 | 209 | |||||||||
| Other assets | 32 | 32 | 27 | |||||||||
| Total assets | $ | 18,782 | $ | 21,681 | $ | 27,162 | ||||||
F-3
Table of Contents
(A Development Stage Company)
CONSOLIDATED BALANCE SHEETS—(Continued)
(In thousands, except share and per share data)
| Pro Forma | ||||||||||||||||
| Shareholders’ | ||||||||||||||||
| Equity at | ||||||||||||||||
| March 31, | December 31, | March 31, | ||||||||||||||
| 2009 | 2008 | 2007 | 2009 | |||||||||||||
| (unaudited) | (Unaudited) | |||||||||||||||
| (Note 1) | ||||||||||||||||
Liabilities, convertible preferred stock and shareholders’ equity (deficit) | ||||||||||||||||
| Current liabilities: | ||||||||||||||||
| Accounts payable | $ | 1,153 | $ | 1,229 | $ | 2,567 | ||||||||||
| Accrued expenses | 3,322 | 3,764 | 2,296 | |||||||||||||
| Preferred stock warrant liability | 1,720 | 1,780 | 1,562 | — | ||||||||||||
| Deferred revenue | 1,317 | 232 | 500 | |||||||||||||
| Current portion of notes payable | 16,238 | 16,556 | 1,010 | |||||||||||||
| Total current liabilities | 23,750 | 23,561 | 7,935 | |||||||||||||
| Notes payable, less current portion | 106 | 118 | — | |||||||||||||
| Commitments and contingencies | ||||||||||||||||
| Convertible preferred stock: | ||||||||||||||||
| Issued and outstanding shares—22,567,407 at March 31, 2009 (unaudited) and 22,327,407 at December 31, 2008 and 2007 (0 pro forma) | ||||||||||||||||
| Liquidation preference of $93,279 at March 31, 2009 (unaudited) and $92,079 at December 31, 2008 and 2007 | 91,019 | 89,168 | 89,168 | — | ||||||||||||
| Shareholders’ equity (deficit): | ||||||||||||||||
| Preferred stock, par value $0.01 per share: | ||||||||||||||||
| Authorized shares—26,314,511 at March 31, 2009 (unaudited) and December 31, 2008 and 2007 (20,000,000 pro forma) | ||||||||||||||||
| Designated convertible—26,314,511 at March 31, 2009 (unaudited) and December 31, 2008 and 2007 (0 pro forma) | — | — | — | — | ||||||||||||
| Common stock, par value $0.01: | ||||||||||||||||
| Authorized shares—40,000,000 at March 31, 2009 (unaudited) and December 31, 2008 and 2007 (150,000,000 pro forma); issued and outstanding shares—5,787,899, 5,784,631 and 5,648,319 at March 31, 2009 (unaudited) and December 31, 2008 and 2007, respectively (28,355,306 pro forma) | 57 | 57 | 56 | $ | 284 | |||||||||||
| Additional paid-in capital | 6,586 | 6,123 | 3,439 | 99,098 | ||||||||||||
| Accumulated other comprehensive loss | (7 | ) | (99 | ) | (4 | ) | (7 | ) | ||||||||
| Deferred stock-based compensation | — | — | (12 | ) | — | |||||||||||
| Deficit accumulated during the development stage | (102,729 | ) | (97,247 | ) | (73,420 | ) | (102,729 | ) | ||||||||
| Total shareholders’ deficit | (96,093 | ) | (91,166 | ) | (69,941 | ) | $ | (3,354 | ) | |||||||
| Total liabilities, convertible preferred stock, and shareholders’ equity (deficit) | $ | 18,782 | $ | 21,681 | $ | 27,162 | ||||||||||
F-4
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF OPERATIONS
(In thousands, except share and per share data)
| Period from | ||||||||||||||||
| June 16, | ||||||||||||||||
| 1994 | ||||||||||||||||
| (Inception) | ||||||||||||||||
| through | ||||||||||||||||
| Year Ended December 31, | December 31, | |||||||||||||||
| 2008 | 2007 | 2006 | 2008 | |||||||||||||
| Grant revenue | $ | 1,170 | $ | 1,923 | $ | 200 | $ | 3,393 | ||||||||
| Operating expenses: | ||||||||||||||||
| Research and development | 17,850 | 15,922 | 9,637 | 62,234 | ||||||||||||
| Acquired in-process research and development | — | — | 10,891 | 10,891 | ||||||||||||
| General and administrative | 7,845 | 10,398 | 3,625 | 32,483 | ||||||||||||
| Total operating expenses | 25,695 | 26,320 | 24,153 | 105,608 | ||||||||||||
| Loss from operations | (24,525 | ) | (24,397 | ) | (23,953 | ) | (102,215 | ) | ||||||||
| Investment income | 661 | 1,582 | 1,088 | 5,163 | ||||||||||||
| Interest expense | (335 | ) | (151 | ) | (91 | ) | (629 | ) | ||||||||
| Other income (expense) | 372 | (125 | ) | 179 | 434 | |||||||||||
| Net loss | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | $ | (97,247 | ) | ||||
| Basic and diluted net loss per common share | $ | (4.22 | ) | $ | (5.44 | ) | $ | (6.17 | ) | |||||||
| Weighted-average shares used to compute basic and diluted net loss per common share | 5,651,583 | 4,248,212 | 3,694,388 | |||||||||||||
| Pro forma basic and diluted net loss per common share (unaudited) | $ | (0.84 | ) | |||||||||||||
| Weighted-average pro forma shares used to compute pro forma basic and diluted net loss per share (unaudited) | 27,978,990 | |||||||||||||||
F-5
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF OPERATIONS—(Continued)
(In thousands, except share and per share data)
(unaudited)
| Period from | ||||||||||||
| June 16, | ||||||||||||
| 1994 | ||||||||||||
| (Inception) | ||||||||||||
| through | ||||||||||||
| Three Months Ended March 31, | March 31, | |||||||||||
| 2009 | 2008 | 2009 | ||||||||||
| Grant revenue | $ | 197 | $ | 234 | $ | 3,590 | ||||||
| Operating expenses: | ||||||||||||
| Research and development | 4,022 | 4,170 | 66,256 | |||||||||
| Acquired in-process research and development | — | — | 10,891 | |||||||||
| General and administrative | 1,410 | 1,596 | 33,893 | |||||||||
| Total operating expenses | 5,432 | 5,766 | 111,040 | |||||||||
| Loss from operations | (5,235 | ) | (5,532 | ) | (107,450 | ) | ||||||
| Investment income | 81 | 279 | 5,244 | |||||||||
| Interest expense | (590 | ) | (22 | ) | (1,219 | ) | ||||||
| Other income | 262 | 172 | 696 | |||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (102,729 | ) | |||
| Basic and diluted net loss per common share | $ | (0.95 | ) | $ | (0.92 | ) | ||||||
| Weighted-average shares used to compute basic and diluted net loss per common share | 5,740,914 | 5,522,711 | ||||||||||
| Pro forma basic and diluted net loss per common share | $ | (0.20 | ) | |||||||||
| Weighted-average pro forma shares used to compute pro forma basic and diluted net loss per share | 28,180,321 | |||||||||||
F-6
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND SHAREHOLDERS’ EQUITY (DEFICIT)
(In thousands, except share and per share data)
| Deficit | |||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | |||||||||||||||||||||||||||||||||||||||
| Convertible | Additional | Other | Deferred | Receivable | During the | Total | |||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | ||||||||||||||||||||||||||||||||
| Balance at June 16, 1994 | — | $ | — | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | $ | — | |||||||||||||||||||||||
| Issuance of common stock to founders for $0.01 per share | — | — | 3,500,000 | 35 | — | — | — | — | — | 35 | |||||||||||||||||||||||||||||||
| Issuance of Series A convertible preferred stock for $1.00 per share and $7 in financing costs | 875,000 | 875 | — | — | (7 | ) | — | — | — | — | (7 | ) | |||||||||||||||||||||||||||||
| Net loss from inception to December 31, 1994 | — | — | — | — | — | — | — | — | (140 | ) | (140 | ) | |||||||||||||||||||||||||||||
| Balance at December 31, 1994 | 875,000 | 875 | 3,500,000 | 35 | (7 | ) | — | — | — | (140 | ) | (112 | ) | ||||||||||||||||||||||||||||
| Net loss and comprehensive loss | — | — | — | — | — | — | — | — | (327 | ) | (327 | ) | |||||||||||||||||||||||||||||
| Balance at December 31, 1995 | 875,000 | 875 | 3,500,000 | 35 | (7 | ) | — | — | — | (467 | ) | (439 | ) | ||||||||||||||||||||||||||||
| Net loss and comprehensive loss | — | — | — | — | — | — | — | — | (495 | ) | (495 | ) | |||||||||||||||||||||||||||||
| Balance at December 31, 1996 | 875,000 | 875 | 3,500,000 | 35 | (7 | ) | — | — | — | (962 | ) | (934 | ) | ||||||||||||||||||||||||||||
| Net loss and comprehensive loss | — | — | — | — | — | — | — | — | (787 | ) | (787 | ) | |||||||||||||||||||||||||||||
| Balance at December 31, 1997 | 875,000 | 875 | 3,500,000 | 35 | (7 | ) | — | — | — | (1,749 | ) | (1,721 | ) | ||||||||||||||||||||||||||||
| Issuance of Series B convertible preferred stock for $1.75 per share and $302 in financing costs | 2,663,244 | 4,661 | — | — | (302 | ) | — | — | — | — | (302 | ) | |||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 6 | — | — | — | — | 6 | |||||||||||||||||||||||||||||||
| Unrealized holding loss on available-for-sale securities for the year ended December 31, 1998 | — | — | — | — | — | (22 | ) | — | — | — | (22 | ) | |||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (930 | ) | (930 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss | (952 | ) | |||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 1998 | 3,538,244 | $ | 5,536 | 3,500,000 | $ | 35 | $ | (303 | ) | $ | (22 | ) | $ | — | $ | — | $ | (2,679 | ) | $ | (2,969 | ) | |||||||||||||||||||
| Repurchase of common stock issued to founders | — | — | (371,875 | ) | (4 | ) | (61 | ) | — | — | — | — | (65 | ) | |||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 per share | — | — | 1,200 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||
| Issuance of common stock for services at $0.18 per share | — | — | 17,537 | — | 3 | — | — | — | — | 3 | |||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 4 | — | — | — | — | 4 | |||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 1999 | — | — | — | — | — | 3 | — | — | — | 3 | |||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (1,801 | ) | (1,801 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss | (1,798 | ) | |||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 1999 (carried forward) | 3,538,244 | 5,536 | 3,146,862 | 31 | (357 | ) | (19 | ) | — | — | (4,480 | ) | (4,825 | ) | |||||||||||||||||||||||||||
F-7
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDERS’ EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | |||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | |||||||||||||||||||||||||||||||||||||||
| Convertible | Additional | Other | Deferred | Receivable | During the | Total | |||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | ||||||||||||||||||||||||||||||||
| Balance at December 31, 1999 (brought forward) | 3,538,244 | 5,536 | 3,146,862 | 31 | (357 | ) | (19 | ) | — | — | (4,480 | ) | (4,825 | ) | |||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock for $2.65 per share and $262 in financing costs | 2,825,291 | 7,487 | — | — | (262 | ) | — | — | — | — | (262 | ) | |||||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock warrants for services | — | 12 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock upon exercise of warrants for $2.65 purchase | 9,433 | 25 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $0.27 per share | — | — | 50,614 | 1 | 9 | — | — | — | — | 10 | |||||||||||||||||||||||||||||||
| Issuance of common stock for services at $0.18 per share | — | — | 9,264 | — | 2 | — | — | — | — | 2 | |||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 8 | — | — | — | — | 8 | |||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 2000 | — | — | — | — | — | 18 | — | — | — | 18 | |||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (1,363 | ) | (1,363 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss | (1,345 | ) | |||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2000 | 6,372,968 | 13,060 | 3,206,740 | 32 | (600 | ) | (1 | ) | — | — | (5,843 | ) | (6,412 | ) | |||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $0.27 per share | — | — | 48,125 | — | 9 | — | — | — | — | 9 | |||||||||||||||||||||||||||||||
| Issuance of common stock for services at $0.27 per share | — | — | 12,268 | — | 3 | — | — | — | — | 3 | |||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 20 | — | — | — | — | 20 | |||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 2001 | — | — | — | — | — | 33 | — | — | — | 33 | |||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (2,554 | ) | (2,554 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss | (2,521 | ) | |||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2001 (carried forward) | 6,372,968 | $ | 13,060 | 3,267,133 | $ | 32 | $ | (568 | ) | $ | 32 | $ | — | $ | — | $ | (8,397 | ) | $ | (8,901 | ) | ||||||||||||||||||||
F-8
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDERS’ EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | ||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | ||||||||||||||||||||||||||||||||||||||
| Convertible | Additional | Other | Deferred | Receivable | During the | Total | ||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | |||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | |||||||||||||||||||||||||||||||
| Balance at December 31, 2001 (brought forward) | 6,372,968 | $ | 13,060 | 3,267,133 | $ | 32 | $ | (568 | ) | $ | 32 | $ | — | $ | — | $ | (8,397 | ) | $ | (8,901 | ) | |||||||||||||||||||
| Issuance of Series D convertible preferred stock for $3.97 per share and $124 in financing costs | 972,580 | 3,861 | — | — | (124 | ) | — | — | — | — | (124 | ) | ||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.19 to $0.27 per share | — | — | 423,660 | 4 | 84 | — | — | — | — | 88 | ||||||||||||||||||||||||||||||
| Deferred stock-based compensation | — | — | — | — | �� | 9 | — | (9 | ) | — | — | — | ||||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation | — | — | — | — | — | — | 2 | — | — | 2 | ||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 121 | — | — | (65 | ) | — | 56 | |||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 2002 | — | — | — | — | — | 16 | — | — | — | 16 | ||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (3,152 | ) | (3,152 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (3,136 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2002 | 7,345,548 | 16,921 | 3,690,793 | 36 | (478 | ) | 48 | (7 | ) | (65 | ) | (11,549 | ) | (12,015 | ) | |||||||||||||||||||||||||
| Issuance of Series B convertible preferred stock upon exercise of warrants for $1.75 per share | 11,829 | 21 | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Repurchase of Series A convertible preferred stock | (100,000 | ) | (100 | ) | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $0.40 per share | — | — | 349,058 | 4 | 91 | — | — | — | — | 95 | ||||||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation | — | — | — | — | — | — | 4 | — | — | 4 | ||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 406 | — | (9 | ) | (86 | ) | — | 311 | ||||||||||||||||||||||||||||
| Unrealized holding loss on available-for-sale securities for the year ended December 31, 2003 | — | — | — | — | — | (37 | ) | — | — | — | (37 | ) | ||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (4,060 | ) | (4,060 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (4,097 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2003 | 7,257,377 | 16,842 | 4,039,851 | 40 | 19 | 11 | (12 | ) | (151 | ) | (15,609 | ) | (15,702 | ) | ||||||||||||||||||||||||||
| Issuance of Series E convertible preferred stock for $5.00 per share and $1,119 in financing costs | 3,672,293 | 18,361 | — | — | (1,119 | ) | — | — | — | — | (1,119 | ) | ||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $0.40 per share | — | — | 55,687 | 1 | 10 | — | — | — | — | 11 | ||||||||||||||||||||||||||||||
| Deferred stock-based compensation | — | — | — | — | 77 | — | (77 | ) | — | — | — | |||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 263 | — | 10 | — | — | 273 | ||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 2004 | — | — | — | — | — | 1 | — | — | — | 1 | ||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (4,578 | ) | (4,578 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (4,577 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2004 (carried forward) | 10,929,670 | $ | 35,203 | 4,095,538 | $ | 41 | $ | (750 | ) | $ | 12 | $ | (79 | ) | $ | (151 | ) | $ | (20,187 | ) | $ | (21,114 | ) | |||||||||||||||||
F-9
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDERS’ EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | ||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | ||||||||||||||||||||||||||||||||||||||
| Convertible | Additional | Other | Deferred | Receivable | During the | Total | ||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | |||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | |||||||||||||||||||||||||||||||
| Balance at December 31, 2004 (brought forward) | 10,929,670 | $ | 35,203 | 4,095,538 | $ | 41 | $ | (750 | ) | $ | 12 | $ | (79 | ) | $ | (151 | ) | $ | (20,187 | ) | $ | (21,114 | ) | |||||||||||||||||
| Issuance of Series E convertible preferred stock for $5 per share and $278 in financing costs | 1,120,215 | 5,601 | — | — | (278 | ) | — | — | — | — | (278 | ) | ||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $0.29 per share | — | — | 387,100 | 4 | 102 | — | — | — | — | 106 | ||||||||||||||||||||||||||||||
| Issuance of Series C convertible preferred stock upon exercise of warrants for $2.65 per share | 31,995 | 84 | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation | — | — | — | — | — | — | 23 | — | — | 23 | ||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | (530 | ) | — | — | (88 | ) | — | (618 | ) | |||||||||||||||||||||||||||
| Reclassification of preferred stock warrants to liabilities | — | — | — | — | (490 | ) | — | — | — | — | (490 | ) | ||||||||||||||||||||||||||||
| Unrealized holding loss on available-for-sale securities for the year ended December 31, 2005 | — | — | — | — | — | (6 | ) | — | — | — | (6 | ) | ||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (7,366 | ) | (7,366 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (7,372 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2005 | 12,081,880 | 40,888 | 4,482,638 | 45 | (1,946 | ) | 6 | (56 | ) | (239 | ) | (27,553 | ) | (29,743 | ) | |||||||||||||||||||||||||
| Issuance of Series E convertible preferred stock for $5.00 per share and $1,821 in financing costs | 6,156,700 | 30,784 | — | — | (1,821 | ) | — | — | — | — | (1,821 | ) | ||||||||||||||||||||||||||||
| Issuance of Series E preferred stock warrants to placement agents | — | — | — | — | (607 | ) | — | — | — | — | (607 | ) | ||||||||||||||||||||||||||||
| Issuance of Series E convertible preferred stock and common stock for the acquisition of nura | 3,398,445 | 14,070 | 36,246 | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash at $0.18 to $5.42 per share | — | — | 453,716 | 5 | 121 | — | — | — | — | 126 | ||||||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation | — | — | — | — | — | — | 23 | — | — | 23 | ||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 1,416 | — | — | — | — | 1,416 | ||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities for the year ended December 31, 2006 | — | — | — | — | — | 20 | — | — | — | 20 | ||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (22,777 | ) | (22,777 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (22,757 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2006 (carried forward) | 21,637,025 | $ | 85,742 | 4,972,600 | $ | 50 | $ | (2,838 | ) | $ | 26 | $ | (33 | ) | $ | (239 | ) | $ | (50,329 | ) | $ | (53,363 | ) | |||||||||||||||||
F-10
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDERS’ EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | ||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | ||||||||||||||||||||||||||||||||||||||
| Convertible | Additional | Other | Deferred | Receivable | During the | Total | ||||||||||||||||||||||||||||||||||
| Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | |||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | |||||||||||||||||||||||||||||||
| Balance at December 31, 2006 (brought forward) | 21,637,025 | $ | 85,742 | 4,972,600 | $ | 50 | $ | (2,838 | ) | $ | 26 | $ | (33 | ) | $ | (239 | ) | $ | (50,329 | ) | $ | (53,363 | ) | |||||||||||||||||
| Issuance of Series D convertible preferred stock upon exercise of warrants for $3.97 per share | 24,382 | 96 | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Issuance of Series E convertible preferred stock for $5.00 per share and $90 in financing costs | 666,000 | 3,330 | — | — | (90 | ) | — | — | — | — | (90 | ) | ||||||||||||||||||||||||||||
| Issuance of Series E Preferred stock Warrants to placement agents | — | — | — | — | (22 | ) | — | — | — | — | (22 | ) | ||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of common stock warrants | — | — | 107,142 | 1 | 186 | — | — | — | — | 187 | ||||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash of $0.18 to $1.00 per share | — | — | 408,857 | 5 | 168 | — | — | — | — | 173 | ||||||||||||||||||||||||||||||
| Issuance of common stock in connection with early-exercise of stock options for cash of $0.50 to $1.00 per share | — | — | 159,063 | 2 | 153 | — | — | — | — | 155 | ||||||||||||||||||||||||||||||
| Early exercise of common stock subject to repurchase | — | — | — | (2 | ) | (153 | ) | — | — | — | — | (155 | ) | |||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation, net of cancellations | — | — | — | — | (4 | ) | — | 21 | — | — | 17 | |||||||||||||||||||||||||||||
| Stock-based compensation | — | — | 657 | — | 6,039 | — | — | — | — | 6,039 | ||||||||||||||||||||||||||||||
| Repayment of note receivable from related party | — | — | — | — | — | — | — | 239 | — | 239 | ||||||||||||||||||||||||||||||
| Unrealized holding loss on available-for-sale securities for the year ended December 31, 2007 | — | — | — | — | — | (30 | ) | — | — | — | (30 | ) | ||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (23,091 | ) | (23,091 | ) | ||||||||||||||||||||||||||||
| Comprehensive loss | (23,121 | ) | ||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2007 (Carried forward) | 22,327,407 | $ | 89,168 | 5,648,319 | $ | 56 | $ | 3,439 | $ | (4 | ) | $ | (12 | ) | $ | — | $ | (73,420 | ) | $ | (69,941 | ) | ||||||||||||||||||
F-11
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDER’S EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | |||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | |||||||||||||||||||||||||||||||||||||||
| Additional | Other | Deferred | Receivable | During the | Total | ||||||||||||||||||||||||||||||||||||
| Convertible Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | ||||||||||||||||||||||||||||||||
| Balance at December 31, 2007 (brought forward) | 22,327,407 | $ | 89,168 | 5,648,319 | $ | 56 | $ | 3,439 | $ | (4 | ) | $ | (12 | ) | $ | — | $ | (73,420 | ) | $ | (69,941 | ) | |||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash of $0.18 to $1.25 per share | — | — | 136,312 | 1 | 39 | — | — | — | — | 40 | |||||||||||||||||||||||||||||||
| Issuance of common stock warrants in connection with notes payable | — | — | — | — | 241 | — | — | — | — | 241 | |||||||||||||||||||||||||||||||
| Vesting of early-exercised stock options | — | — | — | — | 101 | — | — | — | — | 101 | |||||||||||||||||||||||||||||||
| Stock-based compensation | — | — | — | — | 2,303 | — | — | — | — | 2,303 | |||||||||||||||||||||||||||||||
| Amortization of deferred stock-based compensation | — | — | — | — | — | — | 12 | — | — | 12 | |||||||||||||||||||||||||||||||
| Unrealized holding loss on available-for-sale securities for the year ended December 31, 2008 | — | — | — | — | — | (95 | ) | — | — | — | (95 | ) | |||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | — | (23,827 | ) | (23,827 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss | — | — | — | — | — | — | — | — | — | (23,922 | ) | ||||||||||||||||||||||||||||||
| Balance at December 31, 2008 | 22,327,407 | $ | 89,168 | 5,784,631 | $ | 57 | $ | 6,123 | $ | (99 | ) | $ | — | $ | — | $ | (97,247 | ) | $ | (91,166 | ) | ||||||||||||||||||||
F-12
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CONVERTIBLE PREFERRED STOCK AND
SHAREHOLDER’S EQUITY (DEFICIT)—(Continued)
(In thousands, except share and per share data)
| Deficit | |||||||||||||||||||||||||||||||||||||||||
| Accumulated | Notes | Accumulated | |||||||||||||||||||||||||||||||||||||||
| Additional | Other | Deferred | Receivable | During the | Total | ||||||||||||||||||||||||||||||||||||
| Convertible Preferred Stock | Common Stock | Paid-in | Comprehensive | Stock-Based | from Related | Development | Shareholders’ | ||||||||||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Income (Loss) | Compensation | Party | Stage | Deficit | ||||||||||||||||||||||||||||||||
| Balance at December 31, 2008 (brought forward) | 22,327,407 | $ | 89,168 | 5,784,631 | $ | 57 | $ | 6,123 | $ | (99 | ) | $ | — | $ | — | $ | (97,247 | ) | ($ | 91,166 | ) | ||||||||||||||||||||
| Issuance of Series E convertible preferred stock for cash of $7.71 per share in connection with research and development funding agreement (unaudited) | 240,000 | 1,851 | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||
| Issuance of common stock upon exercise of stock options for cash of $1.25 per share (unaudited) | — | — | 8,333 | — | 10 | — | — | — | — | 10 | |||||||||||||||||||||||||||||||
| Vesting of early-exercised stock options (unaudited) | — | — | — | — | 5 | — | — | — | — | 5 | |||||||||||||||||||||||||||||||
| Repurchase of early-exercised stock options (unaudited) | — | — | (5,065 | ) | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||
| Stock-based compensation (unaudited) | — | — | — | — | 448 | — | — | — | — | 448 | |||||||||||||||||||||||||||||||
| Unrealized holding gain on available-for-sale securities (unaudited) | — | — | — | — | — | 92 | — | — | — | 92 | |||||||||||||||||||||||||||||||
| Net loss (unaudited) | — | — | — | — | — | — | — | — | (5,482 | ) | (5,482 | ) | |||||||||||||||||||||||||||||
| Comprehensive loss (unaudited) | — | — | — | — | — | — | — | — | — | (5,390 | ) | ||||||||||||||||||||||||||||||
| Balance at March 31, 2009 (unaudited) | 22,567,407 | $ | 91,019 | 5,787,899 | $ | 57 | $ | 6,586 | $ | (7 | ) | $ | — | $ | — | $ | (102,729 | ) | $ | (96,093 | ) | ||||||||||||||||||||
F-13
Table of Contents
| Period from | ||||||||||||||||
| June 16, 1994 | ||||||||||||||||
| (Inception) | ||||||||||||||||
| Year Ended | through | |||||||||||||||
| December 31, | December 31, | |||||||||||||||
| 2008 | 2007 | 2006 | 2008 | |||||||||||||
Operating activities | ||||||||||||||||
| Net loss | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | $ | (97,247 | ) | ||||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||||||
| Depreciation and amortization | 434 | 375 | 232 | 1,551 | ||||||||||||
| Stock-based compensation expense | 2,315 | 6,056 | 1,439 | 10,158 | ||||||||||||
| (Gain) loss on remeasurement of preferred stock warrant values | 218 | 503 | (117 | ) | 595 | |||||||||||
| Non-cash interest expense | 55 | — | — | 55 | ||||||||||||
| (Gain) loss on sale of investment securities | 76 | (145 | ) | (145 | ) | 45 | ||||||||||
| Write-off of deferred public offering costs | 1,948 | — | — | 1,948 | ||||||||||||
| Acquired in-process research and development | — | — | 10,891 | 10,891 | ||||||||||||
| Other than temporary impairment loss on investments | — | — | — | 163 | ||||||||||||
| Changes in operating assets and liabilities, net of effect from nura acquisition in 2006: | ||||||||||||||||
| Grant and other receivables | (17 | ) | 1,110 | — | 1,093 | |||||||||||
| Prepaid expenses and other current and noncurrent assets | 19 | (22 | ) | 150 | (172 | ) | ||||||||||
| Deferred public offering costs | (486 | ) | (1,462 | ) | — | (1,948 | ) | |||||||||
| Accounts payable and accrued expenses | (140 | ) | 3,162 | 155 | 4,658 | |||||||||||
| Deferred revenue | (268 | ) | (800 | ) | — | (1,068 | ) | |||||||||
| Net cash used in operating activities | (19,673 | ) | (14,314 | ) | (10,172 | ) | (69,278 | ) | ||||||||
Investing activities | ||||||||||||||||
| Purchases of property and equipment | (164 | ) | (534 | ) | (166 | ) | (1,793 | ) | ||||||||
| Purchases of investments | — | (30,562 | ) | (9,541 | ) | (83,897 | ) | |||||||||
| Proceeds from the sale of investments | 5,572 | 11,450 | 2,007 | 32,671 | ||||||||||||
| Proceeds from the maturities of investments | 5,158 | 13,555 | 7,333 | 43,664 | ||||||||||||
| Cash paid for acquisition of nura, net of cash acquired of $87 | — | — | (212 | ) | (212 | ) | ||||||||||
| Net cash provided by (used in) investing activities | 10,566 | (6,091 | ) | (579 | ) | (9,567 | ) | |||||||||
Financing activities | ||||||||||||||||
| Proceeds from borrowings under note payable, net of loan origination costs | 16,878 | — | — | 16,928 | ||||||||||||
| Payments on notes payable | (1,010 | ) | (1,005 | ) | (391 | ) | (2,456 | ) | ||||||||
| Proceeds from issuance of common stock and exercise of stock options | 40 | 360 | 126 | 642 | ||||||||||||
| Proceeds from the repayment of related party notes receivable | — | 239 | — | 239 | ||||||||||||
| Proceeds from issuance of convertible preferred stock, net of issuance costs | — | 3,336 | 28,963 | 71,183 | ||||||||||||
| Issuance of Series E convertible preferred stock for $5.00 per share concurrent with acquisition of nura | — | — | 5,200 | 5,200 | ||||||||||||
| Repurchase of Series A convertible preferred stock and common stock | — | — | — | (165 | ) | |||||||||||
| Net cash provided by financing activities | 15,908 | 2,930 | 33,898 | 91,571 | ||||||||||||
| Net increase (decrease) in cash and cash equivalents | 6,801 | (17,475 | ) | 23,147 | 12,726 | |||||||||||
| Cash and cash equivalents at beginning of period | 5,925 | 23,400 | 253 | — | ||||||||||||
| Cash and cash equivalents at end of period | $ | 12,726 | $ | 5,925 | $ | 23,400 | $ | 12,726 | ||||||||
Supplemental cash flow information | ||||||||||||||||
| Cash paid for interest | $ | 222 | $ | 151 | $ | 91 | $ | 516 | ||||||||
| Purchase of equipment included in accounts payable and accrued expenses | $ | 52 | $ | — | $ | — | $ | 52 | ||||||||
| Purchase of software financed with note payable | $ | 193 | $ | — | $ | — | $ | 193 | ||||||||
| Vesting of early-exercised stock options | $ | 101 | $ | — | $ | — | $ | 101 | ||||||||
| Issuance of common stock warrants in connection with notes payable | $ | 241 | $ | — | $ | — | $ | 241 | ||||||||
| Issuance of common stock in exchange for note receivable from related party | $ | — | $ | — | $ | — | $ | 239 | ||||||||
| Preferred stock and common stock issued in connection with nura acquisition | $ | — | $ | — | $ | 14,070 | $ | 14,070 | ||||||||
F-14
Table of Contents
(A Development Stage Company)
CONSOLIDATED STATEMENTS OF CASH FLOWS—(Continued)
(In thousands)
(unaudited)
| Period from | ||||||||||||
| June 16,1994 | ||||||||||||
| (Inception) | ||||||||||||
| Three Months Ended | through | |||||||||||
| March 31, | March 31, | |||||||||||
| 2009 | 2008 | 2009 | ||||||||||
Operating activities | ||||||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (102,729 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||||||
| Depreciation and amortization | 122 | 98 | 1,673 | |||||||||
| Stock-based compensation expense | 448 | 718 | 10,606 | |||||||||
| (Gain) loss on remeasurement of preferred stock warrant values | (60 | ) | (56 | ) | 535 | |||||||
| Non-cash interest expense | 59 | — | 114 | |||||||||
| (Gain) loss on sale of investment securities | 6 | 13 | 51 | |||||||||
| Write-off of deferred public offering costs | — | — | 1,948 | |||||||||
| Acquired in-process research and development | — | — | 10,891 | |||||||||
| Other than temporary impairment loss on investments | — | — | 163 | |||||||||
| Changes in operating assets and liabilities, net of effect from nura acquisition in 2006: | ||||||||||||
| Grant and other receivables | (28 | ) | 94 | 1,065 | ||||||||
| Prepaid expenses and other current and noncurrent assets | (160 | ) | 7 | (332 | ) | |||||||
| Deferred public offering costs | (204 | ) | (378 | ) | (2,152 | ) | ||||||
| Accounts payable and accrued expenses | (510 | ) | (686 | ) | 4,148 | |||||||
| Deferred revenue | 1,085 | (195 | ) | 17 | ||||||||
| Net cash used in operating activities | (4,724 | ) | (5,488 | ) | (74,002 | ) | ||||||
Investing activities | ||||||||||||
| Purchases of property and equipment | (8 | ) | (30 | ) | (1,801 | ) | ||||||
| Purchases of investments | — | — | (83,897 | ) | ||||||||
| Proceeds from the sale of investments | — | — | 32,671 | |||||||||
| Proceeds from the maturities of investments | 423 | 2,083 | 44,087 | |||||||||
| Cash paid for acquisition of nura, net of cash acquired of $87 | — | — | (212 | ) | ||||||||
| Net cash provided by (used in) investing activities | 415 | 2,053 | (9,152 | ) | ||||||||
Financing activities | ||||||||||||
| Proceeds from issuance of convertible preferred stock, net of issuance costs | 1,851 | — | 73,034 | |||||||||
| Proceeds from borrowings under note payable, net of debt issuance costs | — | — | 16,928 | |||||||||
| Payments on notes payable | (376 | ) | (266 | ) | (2,832 | ) | ||||||
| Proceeds from issuance of common stock and exercise of stock options | 10 | 37 | 652 | |||||||||
| Proceeds from the repayment of related party notes receivable | — | — | 239 | |||||||||
| Issuance of Series E convertible preferred stock for $5.00 per share concurrent with acquisition of nura | — | — | 5,200 | |||||||||
| Repurchase of Series A convertible preferred stock and unvested common stock | (3 | ) | — | (168 | ) | |||||||
| Net cash provided by (used in) financing activities | 1,482 | (229 | ) | 93,053 | ||||||||
| Net (decrease) increase in cash and cash equivalents | (2,827 | ) | (3,664 | ) | 9,899 | |||||||
| Cash and cash equivalents at beginning of period | 12,726 | 5,925 | — | |||||||||
| Cash and cash equivalents at end of period | $ | 9,899 | $ | 2,261 | $ | 9,899 | ||||||
Supplemental cash flow information | ||||||||||||
| Cash paid for interest | $ | 531 | $ | 22 | $ | 1,047 | ||||||
| Purchase of equipment included in accounts payable and accrued expenses | $ | — | $ | — | $ | 22 | ||||||
| Purchase of software financed with note payable | $ | — | $ | — | $ | 166 | ||||||
| Vesting of early-exercised stock options | $ | 5 | $ | — | $ | 106 | ||||||
| Issuance of warrants in connection with notes payable | $ | — | $ | — | $ | 253 | ||||||
| Issuance of common stock in exchange for note receivable from related party | $ | — | $ | — | $ | 239 | ||||||
| Preferred stock and common stock issued in connection with nura acquisition | $ | — | $ | — | $ | 14,070 | ||||||
F-15
Table of Contents
(A Development Stage Company)
F-16
Table of Contents
(A Development Stage Company)
F-17
Table of Contents
(A Development Stage Company)
| March 31, | December 31, | |||||||||||
| 2009 | 2008 | 2007 | ||||||||||
| (in thousands) | ||||||||||||
| Grant revenue receivable | $ | 200 | $ | 180 | $ | 143 | ||||||
| Sublease income receivable | 24 | 27 | 9 | |||||||||
| Other receivables | 11 | — | 38 | |||||||||
| Grant and other receivables | $ | 235 | $ | 207 | $ | 190 | ||||||
F-18
Table of Contents
(A Development Stage Company)
| March 31, | December 31, | |||||||||||||||
| 2009 | 2008 | 2007 | ||||||||||||||
| (in thousands) | ||||||||||||||||
| Clinical trials | $ | 1,559 | $ | 1,644 | $ | 906 | ||||||||||
| Contract preclinical research | 190 | 423 | 11 | |||||||||||||
| Employee compensation | 339 | 319 | 463 | |||||||||||||
| Success fee liability related to notes payable | 310 | 310 | — | |||||||||||||
| Other accruals | 924 | 1,068 | 916 | |||||||||||||
| Accrued expenses | $ | 3,322 | $ | 3,764 | $ | 2,296 | ||||||||||
F-19
Table of Contents
(A Development Stage Company)
F-20
Table of Contents
(A Development Stage Company)
F-21
Table of Contents
(A Development Stage Company)
| Three Months Ended March 31, | Year Ended December 31, | |||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | |||||
| Unrealized gain (loss) on available-for-sale securities | 92 | 19 | (95 | ) | (30 | ) | 20 | |||||||||||||
| Other comprehensive loss | $ | (5,390 | ) | $ | (5,084 | ) | $ | (23,922 | ) | $ | (23,121 | ) | $ | (22,757 | ) | |||||
F-22
Table of Contents
(A Development Stage Company)
| Three Months Ended March 31, | Year Ended December 31, | |||||||||||||||||||
| �� | 2009 | 2008 | 2008 | 2007 | 2006 | |||||||||||||||
Historical Numerator: | ||||||||||||||||||||
| Net loss | $ | (5,482 | ) | $ | (5,103 | ) | $ | (23,827 | ) | $ | (23,091 | ) | $ | (22,777 | ) | |||||
Denominator: | ||||||||||||||||||||
| Weighted-average common shares outstanding | 5,789,555 | 5,680,708 | 5,757,946 | 5,260,867 | 4,622,315 | |||||||||||||||
| Less: Weighted-average unvested common shares subject to repurchase | (48,641 | ) | (157,997 | ) | (106,363 | ) | (84,728 | ) | — | |||||||||||
| Less: Common shares subject to shareholder note receivable | — | — | — | (927,927 | ) | (927,927 | ) | |||||||||||||
| Denominator for basic and diluted net loss per common share | 5,790,914 | 5,522,711 | 5,651,583 | 4,248,212 | 3,694,388 | |||||||||||||||
| Basic and diluted net loss per common share | $ | (0.95 | ) | $ | (0.92 | ) | $ | (4.22 | ) | $ | (5.44 | ) | $ | (6.17 | ) | |||||
F-23
Table of Contents
(A Development Stage Company)
| Three Months Ended March 31, | December 31, | |||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||
| Convertible preferred stock | 22,567,407 | 22,327,407 | 22,327,407 | 22,327,407 | 21,637,025 | |||||||||||||||
| Outstanding options to purchase common stock | 5,441,744 | 5,824,382 | 5,566,243 | 5,908,182 | 5,073,594 | |||||||||||||||
| Common stock subject to shareholder note receivable | — | — | — | 927,927 | 927,927 | |||||||||||||||
| Warrants to purchase common stock and convertible preferred stock | 459,059 | 409,643 | 459,059 | 409,643 | 550,981 | |||||||||||||||
| Common stock subject to repurchase | 45,833 | 157,730 | 56,373 | 158,530 | — | |||||||||||||||
| Total | 28,514,043 | 28,719,162 | 28,409,082 | 29,731,689 | 28,189,527 | |||||||||||||||
| Three Months | ||||||||
| Ended | Year Ended | |||||||
| March 31, | December 31, | |||||||
| 2009 | 2008 | |||||||
Pro Forma (unaudited) | ||||||||
Numerator: | ||||||||
| Net loss | $ | (5,482 | ) | $ | (23,827 | ) | ||
| Plus: other expense (income) attributable to the convertible preferred stock warrants assumed to have been converted to common stock warrants | (60 | ) | 218 | |||||
| Pro forma net loss | $ | (5,542 | ) | $ | (23,609 | ) | ||
Denominator: | ||||||||
| Denominator for basic and diluted net loss per common share | 5,740,914 | 5,651,583 | ||||||
| Plus: weighted-average pro forma adjustments to reflect assumed conversion of convertible preferred stock | 22,439,407 | 22,327,407 | ||||||
| Denominator for pro forma basic and diluted net loss per common share | 28,180,321 | 27,978,990 | ||||||
| Pro forma basic and diluted net loss per common share | $ | (0.20 | ) | $ | (0.84 | ) | ||
F-24
Table of Contents
(A Development Stage Company)
F-25
Table of Contents
(A Development Stage Company)
| March 31, 2009 | ||||||||||||||||
| Gross | Gross | |||||||||||||||
| Amortized | Unrealized | Unrealized | ||||||||||||||
| Cost | Gains | Losses | Fair Value | |||||||||||||
| (in thousands) | ||||||||||||||||
| Cash and cash equivalents | $ | 10,092 | $ | — | $ | — | $ | 10,092 | ||||||||
| Mortgage-backed securities | 6,926 | 19 | (26 | ) | 6,919 | |||||||||||
| Total | $ | 17,018 | $ | 19 | $ | (26 | ) | $ | 17,011 | |||||||
| Amounts classified as cash and cash equivalents | $ | 9,899 | ||||||||||||||
| Amounts classified as restricted cash | 193 | |||||||||||||||
| Amounts classified as short-term investments | 6,919 | |||||||||||||||
| Total | $ | 17,011 | ||||||||||||||
| December 31, 2008 | ||||||||||||||||
| Gross | Gross | |||||||||||||||
| Amortized | Unrealized | Unrealized | ||||||||||||||
| Cost | Gains | Losses | Fair Value | |||||||||||||
| (in thousands) | ||||||||||||||||
| Cash and cash equivalents | $ | 12,919 | $ | — | $ | — | $ | 12,919 | ||||||||
| Mortgage-backed securities | 7,355 | 3 | (102 | ) | 7,256 | |||||||||||
| Total | $ | 20,274 | $ | 3 | $ | (102 | ) | $ | 20,175 | |||||||
| Amounts classified as cash and cash equivalents | $ | 12,726 | ||||||||||||||
F-26
Table of Contents
(A Development Stage Company)
| December 31, 2008 | ||||||||||||||||
| Gross | Gross | |||||||||||||||
| Amortized | Unrealized | Unrealized | ||||||||||||||
| Cost | Gains | Losses | Fair Value | |||||||||||||
| (in thousands) | ||||||||||||||||
| Amounts classified as restricted cash | 193 | |||||||||||||||
| Amounts classified as short-term investments | 7,256 | |||||||||||||||
| Total | $ | 20,175 | ||||||||||||||
| December 31, 2007 | ||||||||||||||||
| Gross | Gross | |||||||||||||||
| Amortized | Unrealized | Unrealized | ||||||||||||||
| Cost | Gains | Losses | Fair Value | |||||||||||||
| (in thousands) | ||||||||||||||||
| Cash and cash equivalents | $ | 1,135 | $ | — | $ | — | $ | 1,135 | ||||||||
| Commercial paper | 4,995 | 4 | — | 4,999 | ||||||||||||
| Mortgage-backed securities | 18,165 | 32 | (40 | ) | 18,157 | |||||||||||
| Total | $ | 24,295 | $ | 36 | $ | (40 | ) | $ | 24,291 | |||||||
| Amounts classified as cash and cash equivalents | $ | 5,925 | ||||||||||||||
| Amounts classified as restricted cash | 209 | |||||||||||||||
| Amounts classified as short-term investments | 18,157 | |||||||||||||||
| Total | $ | 24,291 | ||||||||||||||
F-27
Table of Contents
(A Development Stage Company)
| Three Month Ended March 31, | Year Ended December 31, | |||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Gross interest income | $ | 87 | $ | 302 | $ | 737 | $ | 1,437 | $ | 943 | ||||||||||
| Gross realized gains on investments | — | — | 16 | 310 | 270 | |||||||||||||||
| Gross realized losses on investments | (6 | ) | (23 | ) | (92 | ) | (165 | ) | (125 | ) | ||||||||||
| Total investment income | $ | 81 | $ | 279 | $ | 661 | $ | 1,582 | $ | 1,088 | ||||||||||
F-28
Table of Contents
(A Development Stage Company)
| March 31, 2009 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| (in thousands) | ||||||||||||||||
| Short-term investments: | ||||||||||||||||
| Money market funds | $ | 7,125 | $ | — | $ | — | $ | 7,125 | ||||||||
| Mortgage-backed securities | — | 6,919 | — | 6,919 | ||||||||||||
| Total | $ | 7,125 | $ | 6,919 | $ | — | $ | 14,044 | ||||||||
| Liabilities: | ||||||||||||||||
| Preferred stock warrant liability | $ | — | $ | — | $ | 1,720 | $ | 1,720 | ||||||||
| Notes payable success fee liability | — | — | 310 | 310 | ||||||||||||
| Total | $ | — | $ | — | $ | 2,030 | $ | 2,030 | ||||||||
| December 31, 2008 | ||||||||||||||||
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| (in thousands) | ||||||||||||||||
| Short-term investments: | ||||||||||||||||
| Money market funds | $ | 12,783 | $ | — | $ | — | $ | 12,783 | ||||||||
| Mortgage-backed securities | — | 7,256 | — | 7,256 | ||||||||||||
| Total | $ | 12,783 | $ | 7,256 | $ | — | $ | 20,039 | ||||||||
| Liabilities: | ||||||||||||||||
| Preferred stock warrant liability | $ | — | $ | — | $ | 1,780 | $ | 1,780 | ||||||||
| Notes payable success fee liability | — | — | 310 | 310 | ||||||||||||
| Total | $ | — | $ | — | $ | 2,090 | $ | 2,090 | ||||||||
F-29
Table of Contents
(A Development Stage Company)
| Notes Payable | ||||||||
| Preferred Stock | Success Fee | |||||||
| Warrant Liability | Liability | |||||||
| (in thousands) | ||||||||
| Fair value at December 31, 2007 | $ | 1,562 | $ | — | ||||
| Additions measured at fair value | — | 319 | ||||||
| Change in fair value | 218 | (9 | ) | |||||
| Fair value at December 31, 2008 | $ | 1,780 | $ | 310 | ||||
| Change in fair value | (60 | ) | — | |||||
| Fair value at March 31, 2009 | $ | 1,720 | $ | 310 | ||||
| March 31 | December 31, | |||||||||||
| 2009 | 2008 | 2007 | ||||||||||
| (in thousands) | ||||||||||||
| Computer equipment | $ | 274 | $ | 266 | $ | 267 | ||||||
| Computer software | 319 | 319 | 46 | |||||||||
| Office equipment and furniture | 284 | 284 | 268 | |||||||||
| Leasehold improvements | 278 | 278 | 276 | |||||||||
| Laboratory equipment | 1,016 | 1,016 | 953 | |||||||||
| Total | 2,171 | 2,163 | 1,810 | |||||||||
| Less accumulated depreciation and amortization | (1,341 | ) | (1,245 | ) | (971 | ) | ||||||
| Property and equipment, net | $ | 830 | $ | 918 | $ | 839 | ||||||
F-30
Table of Contents
(A Development Stage Company)
F-31
Table of Contents
(A Development Stage Company)
F-32
Table of Contents
(A Development Stage Company)
| Loan and Security | Software Financing | |||||||||||
Year Ending December 31, | Agreement | Arrangement | Total | |||||||||
| 2009 | $ | 3,629 | $ | 75 | $ | 3,704 | ||||||
| 2010 | 5,459 | 64 | 5,523 | |||||||||
| 2011 | 6,182 | 54 | 6,236 | |||||||||
| 2012 | 1,730 | — | 1,730 | |||||||||
| Total principal payments | 17,000 | 193 | 17,193 | |||||||||
| Less current portion | (17,000 | ) | (75 | ) | (17,075 | ) | ||||||
| Total notes payable, net of current portion | — | $ | 118 | $ | 118 | |||||||
F-33
Table of Contents
(A Development Stage Company)
F-34
Table of Contents
(A Development Stage Company)
| Cash | $ | 87 | ||
| Prepaid assets and other current assets | 233 | |||
| Cash investment from existing nura institutional investors | 5,200 | |||
| Equipment | 182 | |||
| Assumed liabilities | (2,535 | ) | ||
| Net tangible assets | 3,167 | |||
| Assembled workforce | 310 | |||
| Acquired in-process research and development | 10,891 | |||
| Total fair value of assets acquired, net of liabilities assumed | $ | 14,368 | ||
F-35
Table of Contents
(A Development Stage Company)
| Lease | Sublease | Net Lease | ||||||||||
Year Ending December 31, | Payments | Income | Payments | |||||||||
| (in thousands) | ||||||||||||
| 2009 | $ | 1,560 | $ | 603 | $ | 957 | ||||||
| 2010 | 1,563 | 240 | 1,323 | |||||||||
| 2011 | 1,134 | — | 1,134 | |||||||||
| 2012 | 23 | — | 23 | |||||||||
| 2013 | 15 | — | 15 | |||||||||
| Total | $ | 4,295 | $ | 843 | $ | 3,452 | ||||||
F-36
Table of Contents
(A Development Stage Company)
F-37
Table of Contents
(A Development Stage Company)
F-38
Table of Contents
(A Development Stage Company)
| March 31, 2009 | December 31, 2008 | |||||||||||||||||||||||
| Weighted- | Weighted- | |||||||||||||||||||||||
| Warrants | Fair | Average | Warrants | Fair | Average | |||||||||||||||||||
| Outstanding | Value | Exercise Price | Outstanding | Value | Exercise Price | |||||||||||||||||||
| Common stock | 49,481 | $ | — | $ | 6.88 | 49,481 | $ | — | $ | 6.88 | ||||||||||||||
| Series E preferred stock | 409,578 | 1,720 | 6.16 | 409,578 | 1,780 | 6.16 | ||||||||||||||||||
| Total | 459,059 | $ | 1,720 | $ | 6.24 | 459,059 | $ | 1,780 | $ | 6.24 | ||||||||||||||
| March 31, | December 31, | |||||||
| 2009 | 2008 | 2007 | 2006 | |||||
| Risk-free interest rate | 2.13% | 2.3% | 3.78% | 4.57% | ||||
| Weighted-average expected life (in years) | 3.00-5.00 | 3.25-5.00 | 4.25-5.00 | 5.00-6.08 | ||||
| Expected dividend yield | — | — | — | — | ||||
| Expected volatility rate | 71% | 71% | 60% | 60% | ||||
F-39
Table of Contents
(A Development Stage Company)
| March 31, 2009 | ||||||||||||||||||||
| Shares | ||||||||||||||||||||
| Issued | Authorized | Issued and | Aggregate | |||||||||||||||||
| Price per | and | Outstanding | Liquidation | Carrying | ||||||||||||||||
| Share | Designated | Shares | Preference | Value | ||||||||||||||||
| Series A | $ | 1.00 | 775,000 | 775,000 | $ | 775 | $ | 775 | ||||||||||||
| Series B | $ | 1.75 | 2,675,073 | 2,675,073 | 4,681 | 4,682 | ||||||||||||||
| Series C | $ | 2.65 | 2,866,719 | 2,866,719 | 7,597 | 7,608 | ||||||||||||||
| Series D | $ | 3.97 | 997,719 | 996,962 | 3,958 | 3,957 | ||||||||||||||
| Series E* | $ | 5.00 | 19,000,000 | 15,253,653 | 76,268 | 73,997 | ||||||||||||||
| Total | 26,314,511 | 22,567,407 | $ | 93,279 | $ | 91,019 | ||||||||||||||
| December 31, 2008 and 2007 | ||||||||||||||||||||
| Shares | ||||||||||||||||||||
| Issued | Authorized | Issued and | Aggregate | |||||||||||||||||
| Price per | and | Outstanding | Liquidation | Carrying | ||||||||||||||||
| Share | Designated | Shares | Preference | Value | ||||||||||||||||
| Series A | $ | 1.00 | 775,000 | 775,000 | $ 775 | $ 775 | ||||||||||||||
| Series B | $ | 1.75 | 2,675,073 | 2,675,073 | 4,681 | 4,682 | ||||||||||||||
| Series C | $ | 2.65 | 2,866,719 | 2,866,719 | 7,597 | 7,608 | ||||||||||||||
| Series D | $ | 3.97 | 997,719 | 996,962 | 3,958 | 3,957 | ||||||||||||||
| Series E* | $ | 5.00 | 19,000,000 | 15,013,653 | 75,068 | 72,146 | ||||||||||||||
| Total | 26,314,511 | 22,327,407 | $ | 92,079 | $ | 89,168 | ||||||||||||||
| (*) | Shares issued in conjunction with the nura acquisition totaled 3,398,445 at a price of $4.14 per share. |
F-40
Table of Contents
(A Development Stage Company)
F-41
Table of Contents
(A Development Stage Company)
| March 31, | December 31, | |||||||||||
| 2009 | 2008 | |||||||||||
| Options granted and outstanding under the 2008 stock option plan | 65,700 | 50,200 | ||||||||||
| Options available for future grant under the 2008 stock option plan | 2,121,855 | 2,000,627 | ||||||||||
| Options granted and outstanding under the 1998 stock option plan | 5,311,171 | 5,451,167 | ||||||||||
| Options granted and outstanding outside of the stock option plans | 58,806 | 58,806 | ||||||||||
| Options granted and outstanding under the nura 2003 stock option plan | 6,067 | 6,070 | ||||||||||
| Conversion of convertible preferred stock | 22,567,407 | 22,327,407 | ||||||||||
| Convertible preferred stock warrants | 409,578 | 409,578 | ||||||||||
| Common stock warrants | 49,481 | 49,481 | ||||||||||
| Total shares reserved | 30,590,065 | 30,353,336 | ||||||||||
F-42
Table of Contents
(A Development Stage Company)
| • | five percent of the outstanding shares of the Company’s common stock on the last day of the immediately preceding fiscal year; | |
| • | 3,500,000 shares; or | |
| • | such other amount as the Company’s board of directors may determine. |
F-43
Table of Contents
(A Development Stage Company)
| Weighted- | ||||||||||||
| Average | ||||||||||||
| Shares | Exercise | |||||||||||
| Available for | Options | Price per | ||||||||||
| Grant | Outstanding | Share | ||||||||||
| Balance at January 1, 2006 | 198,883 | 1,246,095 | 0.35 | |||||||||
| Authorized increase in Plan shares | 5,700,000 | — | — | |||||||||
| Assumption of outstanding nura stock options | — | 15,192 | 5.42 | |||||||||
| Granted | (4,325,853 | ) | 4,325,853 | 0.50 | ||||||||
| Exercised | — | (453,716 | ) | 0.28 | ||||||||
| Cancelled nura stock options | — | (8,184 | ) | 5.42 | ||||||||
| Cancelled | 51,646 | (51,646 | ) | 0.37 | ||||||||
| Balance at December 31, 2006 | 1,624,676 | 5,073,594 | 0.49 | |||||||||
| Granted | (1,456,733 | ) | 1,456,733 | 1.21 | ||||||||
| Exercised | — | (567,920 | ) | 0.58 | ||||||||
| Cancelled nura stock options | — | (639 | ) | 5.42 | ||||||||
| Cancelled | 53,586 | (53,586 | ) | 0.54 | ||||||||
| Balance at December 31, 2007 | 221,529 | 5,908,182 | $ | 0.66 | ||||||||
| Authorized increase in Plan shares | 2,050,827 | — | — | |||||||||
| Expired | (477,356 | ) | — | — | ||||||||
| Granted | (95,200 | ) | 95,200 | 4.05 | ||||||||
| Exercised | — | (136,312 | ) | 0.30 | ||||||||
| Cancelled | 300,827 | (300,827 | ) | 0.89 | ||||||||
| Balance at December 31, 2008 | 2,000,627 | 5,566,243 | $ | 0.71 | ||||||||
| Authorized increase in Plan shares | 136,728 | — | — | |||||||||
| Expired | (131,666 | ) | — | — | ||||||||
| Granted | (15,500 | ) | 15,500 | 6.36 | ||||||||
| Exercised | — | (8,333 | ) | 1.25 | ||||||||
| Cancelled | 131,666 | (131,666 | ) | 1.05 | ||||||||
| Balance at March 31, 2009 | 2,121,855 | 5,441,744 | $ | 0.72 | ||||||||
F-44
Table of Contents
(A Development Stage Company)
| March 31, 2009 | ||||||||||||||||||||
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Weighted- | ||||||||||||||||||||
| Average | ||||||||||||||||||||
| Remaining | Weighted- | Weighted- | ||||||||||||||||||
| Range of | Number of | Contractual | Average | Number of | Average | |||||||||||||||
Exercise Price | Options | Life (Years) | Exercise Price | Options | Exercise Price | |||||||||||||||
| $0.18-0.40 | 168,306 | 1.88 | $ | 0.28 | 168,306 | $ | 0.28 | |||||||||||||
| $0.50 | 4,163,590 | 7.53 | $ | 0.50 | 3,775,894 | $ | 0.50 | |||||||||||||
| $1.00-1.25 | 1,013,081 | 8.42 | $ | 1.18 | 418,063 | $ | 1.17 | |||||||||||||
| $5.00-6.88 | 96,767 | 8.79 | $ | 6.06 | 7,012 | $ | 5.60 | |||||||||||||
| $0.18-6.88 | 5,441,744 | 7.54 | $ | 0.72 | 4,369,275 | $ | 0.56 | |||||||||||||
| December 31, 2008 | ||||||||||||||||||||
| Options Outstanding | Options Exercisable | |||||||||||||||||||
| Weighted- | ||||||||||||||||||||
| Average | ||||||||||||||||||||
| Remaining | Weighted- | Weighted- | ||||||||||||||||||
| Range of | Number of | Contractual | Average | Number of | Average | |||||||||||||||
Exercise Price | Options | Life (Years) | Exercise Price | Options | Exercise Price | |||||||||||||||
| $0.18-0.40 | 168,306 | 2.66 | $ | 0.28 | 168,306 | $ | 0.28 | |||||||||||||
| $0.50 | 4,164,184 | 7.77 | $ | 0.50 | 3,584,304 | $ | 0.50 | |||||||||||||
| $1.00-1.25 | 1,152,483 | 8.71 | $ | 1.17 | 351,132 | $ | 1.16 | |||||||||||||
| $5.00-6.88 | 81,270 | 8.83 | $ | 6.00 | 6,288 | $ | 5.49 | |||||||||||||
| $0.18-6.88 | 5,566,243 | 7.82 | $ | 0.71 | 4,110,030 | $ | 0.56 | |||||||||||||
F-45
Table of Contents
(A Development Stage Company)
| Three Months Ended | ||||||||||
| March 31, | Years Ended December 31, | |||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||
| Expected volatility | 71% | 60% | 60% | 60% | 60% | |||||
| Expected term (in years) | 6.08 | 6.08 | 6.08 | 6.00-6.08 | 5.00-6.08 | |||||
| Risk-free interest rate | 2.13% | 2.80%-3.40% | 2.80%-3.40% | 3.78%-4.78% | 4.57% -5.04% | |||||
| Expected dividend yield | 0% | 0% | 0% | 0% | 0% | |||||
F-46
Table of Contents
(A Development Stage Company)
| Estimated | ||||||||||||||||
| Number of | Fair Value of | |||||||||||||||
| Shares | Common | |||||||||||||||
| Subject to | Exercise | Stock per | Intrinsic | |||||||||||||
| Options | Price per | Share at | Value per Share | |||||||||||||
Grant Date | Granted | Share | Date of Grant | at Date of Grant | ||||||||||||
| July 2006 | 23,000 | $ | 0.50 | $ | 0.89 | $ | 0.39 | |||||||||
| September 2006 | 28,000 | 0.50 | 0.89 | 0.39 | ||||||||||||
| December 2006 | 4,274,853 | 0.50 | 0.89 | 0.39 | ||||||||||||
| March 2007 | 308,500 | 1.00 | 1.05 | 0.05 | ||||||||||||
| May 2007 | 350,000 | 1.00 | 3.63 | 2.63 | ||||||||||||
| October 2007 | 275,733 | 1.25 | 6.23 | 4.98 | ||||||||||||
| December 2007 | 522,500 | 1.25 | 6.32 | 5.07 | ||||||||||||
| January 2008 | 45,000 | 1.25 | 6.32 | 5.07 | ||||||||||||
| March 2008 | 1,200 | 6.32 | 6.32 | — | ||||||||||||
| June 2008 | 27,000 | 6.32 | 6.88 | 0.56 | ||||||||||||
| September 2008 | 22,000 | 6.88 | 6.87 | — | ||||||||||||
| March 2009 | 15,500 | 6.36 | 6.33 | — | ||||||||||||
F-47
Table of Contents
(A Development Stage Company)
| Three Months | ||||||||||||||||||||
| Ended March 31, | Years Ended December 31, | |||||||||||||||||||
| 2009 | 2008 | 2008 | 2007 | 2006 | ||||||||||||||||
| (in thousands) | ||||||||||||||||||||
| Research and development | $ | 220 | $ | 327 | $ | 983 | $ | 482 | $ | 309 | ||||||||||
| General and administrative | 228 | 391 | 1,332 | 5,574 | 1,130 | |||||||||||||||
| Total | $ | 448 | $ | 718 | $ | 2,315 | $ | 6,056 | $ | 1,439 | ||||||||||
F-48
Table of Contents
(A Development Stage Company)
| December 31, | ||||||||
| 2008 | 2007 | |||||||
| (in thousands) | ||||||||
| Deferred tax assets: | ||||||||
| Net operating loss carryforwards | $ | 24,658 | $ | 18,105 | ||||
| Deferred revenue | 79 | 170 | ||||||
| Stock-based compensation | 120 | 41 | ||||||
| Research and development tax credits | 2,281 | 1,580 | ||||||
| Other | 133 | 138 | ||||||
| 27,271 | 20,034 | |||||||
| Less valuation allowance | (27,271 | ) | (20,034 | ) | ||||
| Net deferred tax assets | $ | — | $ | — | ||||
| December 31, | ||||||||||||
| 2008 | 2007 | 2006 | ||||||||||
| (in thousands) | ||||||||||||
| Statutory tax rate | (34 | %) | (34 | %) | (34 | )% | ||||||
| Permanent differences | 6 | 9 | 19 | |||||||||
| Change in valuation allowance | 21 | 20 | 14 | |||||||||
| Other | 7 | 5 | 1 | |||||||||
| Effective tax rate | — | — | — | |||||||||
F-49
Table of Contents
(A Development Stage Company)
F-50
Table of Contents
(A Development Stage Company)
F-51
Table of Contents
| Page | ||||
| 1 | ||||
| 11 | ||||
| 32 | ||||
| 34 | ||||
| 35 | ||||
| 36 | ||||
| 38 | ||||
| 40 | ||||
| 42 | ||||
| 66 | ||||
| 105 | ||||
| 110 | ||||
| 126 | ||||
| 129 | ||||
| 131 | ||||
| 136 | ||||
| 139 | ||||
| 146 | ||||
| 146 | ||||
| 146 | ||||
| F-1 | ||||

Table of Contents
| ITEM 13. | OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION. |
| SEC registration fee | $ | 4,520 | ||
| NASDAQ Global Market listing fee | 125,000 | |||
| FINRA filing fee | 12,000 | |||
| Printing and engraving | * | |||
| Legal fees and expenses | * | |||
| Accounting fees and expenses | * | |||
| Transfer agent and registrar fees | * | |||
| Director and officer insurance | * | |||
| Miscellaneous | * | |||
| Total | * |
| * | To be completed by amendment. |
| ITEM 14. | INDEMNIFICATION OF DIRECTORS AND OFFICERS. |
II-1
Table of Contents
| ITEM 15. | RECENT SALES OF UNREGISTERED SECURITIES. |
II-2
Table of Contents
| ITEM 16. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES. |
| Exhibit | ||
Number | Description | |
| 1.1* | Form of Underwriting Agreement. | |
| 2.1* | Agreement and Plan of Reorganization among the registrant, Epsilon Acquisition Corporation, nura, inc. and ARCH Venture Corporation dated August 4, 2006 | |
| 3.1* | Form of Amended and Restated Articles of Incorporation of the registrant, to be in effect upon the completion of this offering. | |
| 3.2* | Form of Amended and Restated Bylaws of the registrant, to be in effect upon the completion of this offering. | |
| 4.1** | Form of registrant’s common stock certificate. | |
| 4.2* | Stock Purchase Warrant issued by nura, inc. to Oxford Finance Corporation dated April 26, 2005 (assumed by the registrant on August 11, 2006). | |
| 4.3* | Amended and Restated Investors’ Rights Agreement among the registrant and holders of capital stock dated October 15, 2004. | |
| 5.1 | Opinion of Wilson Sonsini Goodrich & Rosati, Professional Corporation. | |
| 10.1* | Form of Indemnification Agreement to be entered into between the registrant and its directors and officers. | |
| 10.2* | Second Amended and Restated 1998 Stock Option Plan. | |
| 10.3* | Form of Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (that does not permit early exercise). | |
| 10.4* | Form of Amendment to Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (to permit early exercise). | |
| 10.5* | Form of Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (that permits early exercise). | |
| 10.6* | nura, inc. 2003 Stock Plan. | |
| 10.7* | Form of Stock Option Agreement under the nura, inc. 2003 Stock Plan. | |
| 10.8* | 2008 Equity Incentive Plan. | |
| 10.9* | Form of Stock Option Award Agreement under the 2008 Equity Incentive Plan (to be used following the completion of this offering). | |
| 10.10* | Second Amended and Restated Employment Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 30, 2007. | |
| 10.11* | Non-Plan Stock Option Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 11, 2001. | |
| 10.12* | Offer Letter between the registrant and Marcia S. Kelbon, Esq. dated August 16, 2001. | |
| 10.13* | Offer Letter between the registrant and Richard J. Klein dated May 11, 2007. | |
| 10.14* | Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated June 16, 1994. | |
| 10.15* | Technology Transfer Agreement between the registrant and Pamela A. Pierce, M.D., Ph.D. dated June 16, 1994. |
II-3
Table of Contents
| Exhibit | ||
Number | Description | |
| 10.16* | Second Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 11, 2001. | |
| 10.17* | Second Technology Transfer Agreement between the registrant and Pamela Pierce, M.D., Ph.D. dated March 22, 2002. | |
| 10.18* | Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated June 16, 1994 (related to tendon splice technology). | |
| 10.19* | Master Security Agreement between the nura, inc. and Oxford Finance Corporation dated April 26, 2005. | |
| 10.20* | Guaranty from the registrant to Oxford Finance Corporation dated August 11, 2006. | |
| 10.21* | U.S. Bank Centre Office Lease Agreement between Bentall City Centre LLC and Scope International, Inc. dated September 28, 1998. | |
| 10.22* | Assignment and Amendment of Lease among the registrant, City Centre Associates and Navigant Consulting, Inc. dated August 1, 2002. | |
| 10.23* | Second Amendment to Office Lease Agreement between the registrant and City Centre Associates dated January 4, 2006. | |
| 10.24* | Lease Agreement between Alexandria Real Estate Equities, Inc. and Primal, Inc. dated April 6, 2000. | |
| 10.25* | Lease Agreement between Alexandria Real Estate Equities, Inc. and Primal, Inc. dated September 28, 2001. | |
| 10.26* | Assignment and Assumption and Modification of Lease Documents among Alexandria Real Estate Equities, Inc., Primal, Inc., and nura, inc. dated October 23, 2003. | |
| 10.27* | Assignment and Assumption and Modification of Lease Documents among Alexandria Real Estate Equities, Inc., nura, inc., and the registrant dated September 26, 2007. | |
| 10.28†* | Commercial Supply Agreement between the registrant and Hospira Worldwide, Inc. dated October 9, 2007. | |
| 10.29†* | Exclusive License and Sponsored Research Agreement between the registrant and the University of Leicester dated June 10, 2004. | |
| 10.30†* | Research and Development Agreement First Amendment between the registrant and the University of Leicester dated October 1, 2005. | |
| 10.31†* | Exclusive License and Sponsored Research Agreement between the registrant and the Medical Research Council dated October 31, 2005. | |
| 10.32†* | Amendment dated May 8, 2007 to Exclusive License and Sponsored Research Agreement between the registrant and the Medical Research Council dated October 31, 2005. | |
| 10.33†* | Funding Agreement between the registrant and The Stanley Medical Research Institute dated December 18, 2006. | |
| 10.34†* | Services and Materials Agreement between the registrant and Scottish Biomedical Limited dated April 20, 2007. | |
| 10.35†* | Amendment dated April 30, 2007 of the Services and Materials Agreement between the registrant and Scottish Biomedical Limited dated April 20, 2007. | |
| 10.36†* | Drug Product Development and Clinical Supply Agreement between the registrant and Althea Technologies, Inc. dated January 20, 2006. | |
| 10.37†* | Project Plan for Non-GMP and cGPM Fill and Finish of OMS302 between the registrant and Althea Technologies, Inc. dated May 31, 2007. | |
| 10.38†* | Master Services Agreement between nura, inc. and ComGenex, Inc. dated January 27, 2005. | |
| 10.39* | Landlord Consent to Sublease among Christensen O’Connor Johnson Kindness PLLC, City Centre Associates and the registrant dated January 29, 2008. |
II-4
Table of Contents
| Exhibit | ||
Number | Description | |
| 10.40* | Form of Stock Option Award Agreement under the 2008 Equity Incentive Plan (used prior to the completion of this offering). | |
| 10.41†* | Agreement for Antibody Discovery and Development between the registrant and Affitech AS dated July 25, 2008. | |
| 10.42†* | Exclusive Technology Option Agreement between the registrant, Patobios Limited, Susan R. George, M.D., Brian F. O’Dowd, Ph.D. and U.S. Bank National Association as escrow agent dated September 4, 2008. | |
| 10.43* | Loan and Security Agreement between the registrant and BlueCrest Capital Finance, L.P. dated September 12, 2008. | |
| 10.44* | Promissory Note issued by the registrant to BlueCrest Capital Finance, L.P. dated September 12, 2008. | |
| 10.45* | Promissory Note issued by the registrant to BlueCrest Capital Finance, L.P. dated December 23, 2008. | |
| 10.46†* | Agreement for Antibody Development between the registrant and North Coast Biologics LLC dated October 31, 2008. | |
| 10.47†* | Patent Assignment Agreement between the registrant and Roberto Ciccocioppo, Ph.D. dated February 23, 2009. | |
| 10.48* | Amendment to Exercise Notice and Restricted Stock Purchase Agreements between the registrant and Richard J. Klein dated April 29, 2009. | |
| 21.1* | List of significant subsidiaries of the registrant. | |
| 23.1 | Consent of Independent Registered Public Accounting Firm. | |
| 23.4 | Consent of Wilson Sonsini Goodrich & Rosati, Professional Corporation (included in Exhibit 5.1). | |
| 24.1* | Power of Attorney. | |
| 99.1* | Consent of The Reimbursement Group. |
| * | Previously Filed. | |
| ** | To be filed by amendment. | |
| † | Confidential treatment will be requested for portions of this exhibit. These portions will be omitted from this Registration Statement and will be filed separately with the Securities and Exchange Commission. |
| ITEM 17. | UNDERTAKINGS. |
II-5
Table of Contents
II-6
Table of Contents
| By: | /s/ Gregory A. Demopulos, M.D. |
Signature | Title | Date | ||||
/s/ Gregory A. Demopulos, M.D. Gregory A. Demopulos, M.D. | President, Chief Executive Officer, Chief Medical Officer and Chairman of the Board of Directors (Principal Executive, Financial and Accounting Officer) | June 23, 2009 | ||||
* Ray Aspiri | Director | June 23, 2009 | ||||
* Thomas J. Cable | Director | June 23, 2009 | ||||
* Peter A. Demopulos, M.D. | Director | June 23, 2009 | ||||
* Leroy E. Hood, M.D., Ph.D. | Director | June 23, 2009 | ||||
* Jean-Philippe Tripet | Director | June 23, 2009 | ||||
*By: /s/ Gregory A. Demopulos, M.D. Gregory A. Demopulos, M.D. Attorney-in-Fact | ||||||
II-7
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 1 | .1* | Form of Underwriting Agreement. | ||
| 2 | .1* | Agreement and Plan of Reorganization among the registrant, Epsilon Acquisition Corporation, nura, inc. and ARCH Venture Corporation dated August 4, 2006 | ||
| 3 | .1* | Form of Amended and Restated Articles of Incorporation of the registrant, to be in effect upon the completion of this offering. | ||
| 3 | .2* | Form of Amended and Restated Bylaws of the registrant, to be in effect upon the completion of this offering. | ||
| 4 | .1** | Form of registrant’s common stock certificate. | ||
| 4 | .2* | Stock Purchase Warrant issued by nura, inc. to Oxford Finance Corporation dated April 26, 2005 (assumed by the registrant on August 11, 2006). | ||
| 4 | .3* | Amended and Restated Investors’ Rights Agreement among the registrant and holders of capital stock dated October 15, 2004. | ||
| 5 | .1 | Opinion of Wilson Sonsini Goodrich & Rosati, Professional Corporation. | ||
| 10 | .1* | Form of Indemnification Agreement to be entered into between the registrant and its directors and officers. | ||
| 10 | .2* | Second Amended and Restated 1998 Stock Option Plan. | ||
| 10 | .3* | Form of Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (that does not permit early exercise). | ||
| 10 | .4* | Form of Amendment to Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (to permit early exercise). | ||
| 10 | .5* | Form of Stock Option Agreement under the Second Amended and Restated 1998 Stock Option Plan (that permits early exercise). | ||
| 10 | .6* | nura, inc. 2003 Stock Plan. | ||
| 10 | .7* | Form of Stock Option Agreement under the nura, inc. 2003 Stock Plan. | ||
| 10 | .8* | 2008 Equity Incentive Plan. | ||
| 10 | .9* | Form of Stock Option Award Agreement under the 2008 Equity Incentive Plan (to be used following the completion of this offering). | ||
| 10 | .10* | Second Amended and Restated Employment Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 30, 2007. | ||
| 10 | .11* | Non-Plan Stock Option Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 11, 2001. | ||
| 10 | .12* | Offer Letter between the registrant and Marcia S. Kelbon, Esq. dated August 16, 2001. | ||
| 10 | .13* | Offer Letter between the registrant and Richard J. Klein dated May 11, 2007. | ||
| 10 | .14* | Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated June 16, 1994. | ||
| 10 | .15* | Technology Transfer Agreement between the registrant and Pamela Pierce, M.D., Ph.D. dated June 16, 1994. | ||
| 10 | .16* | Second Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated December 11, 2001. | ||
| 10 | .17* | Second Technology Transfer Agreement between the registrant and Pamela Pierce, M.D., Ph.D. dated March 22, 2002. | ||
| 10 | .18* | Technology Transfer Agreement between the registrant and Gregory A. Demopulos, M.D. dated June 16, 1994 (related to tendon splice technology). | ||
| 10 | .19* | Master Security Agreement between the nura, inc. and Oxford Finance Corporation dated April 26, 2005. | ||
| 10 | .20* | Guaranty from the registrant to Oxford Finance Corporation dated August 11, 2006. | ||
| 10 | .21* | U.S. Bank Centre Office Lease Agreement between Bentall City Centre LLC and Scope International, Inc. dated September 28, 1998. | ||
| 10 | .22* | Assignment and Amendment of Lease among the registrant, City Centre Associates and Navigant Consulting, Inc. dated August 1, 2002. | ||
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 10 | .23* | Second Amendment to Office Lease Agreement between the registrant and City Centre Associates dated January 4, 2006. | ||
| 10 | .24* | Lease Agreement between Alexandria Real Estate Equities, Inc. and Primal, Inc. dated April 6, 2000. | ||
| 10 | .25* | Lease Agreement between Alexandria Real Estate Equities, Inc. and Primal, Inc. dated September 28, 2001. | ||
| 10 | .26* | Assignment and Assumption and Modification of Lease Documents among Alexandria Real Estate Equities, Inc., Primal, Inc., and nura, inc. dated October 23, 2003. | ||
| 10 | .27* | Assignment and Assumption and Modification of Lease Documents among Alexandria Real Estate Equities, Inc., nura, inc., and the registrant dated September 26, 2007. | ||
| 10 | .28†* | Commercial Supply Agreement between the registrant and Hospira Worldwide, Inc. dated October 9, 2007. | ||
| 10 | .29†* | Exclusive License and Sponsored Research Agreement between the registrant and the University of Leicester dated June 10, 2004. | ||
| 10 | .30†* | Research and Development Agreement First Amendment between the registrant and the University of Leicester dated October 1, 2005. | ||
| 10 | .31†* | Exclusive License and Sponsored Research Agreement between the registrant and the Medical Research Council dated October 31, 2005. | ||
| 10 | .32†* | Amendment dated May 8, 2007 to Exclusive License and Sponsored Research Agreement between the registrant and the Medical Research Council dated October 31, 2005. | ||
| 10 | .33†* | Funding Agreement between the registrant and The Stanley Medical Research Institute dated December 18, 2006. | ||
| 10 | .34†* | Services and Materials Agreement between the registrant and Scottish Biomedical Limited dated April 20, 2007. | ||
| 10 | .35†* | Amendment dated April 30, 2007 of the Services and Materials Agreement between the registrant and Scottish Biomedical Limited dated April 20, 2007. | ||
| 10 | .36†* | Drug Product Development and Clinical Supply Agreement between the registrant and Althea Technologies, Inc. dated January 20, 2006. | ||
| 10 | .37†* | Project Plan for Non-GMP and cGMP Fill and Finish of OMS302 between the registrant and Althea Technologies, Inc. dated May 31, 2007. | ||
| 10 | .38†* | Master Services Agreement between nura, inc. and ComGenex, Inc. dated January 27, 2005 | ||
| 10 | .39* | Landlord Consent to Sublease among Christensen O’Connor Johnson Kindness PLLC, City Centre Associates and the registrant dated January 29, 2008. | ||
| 10 | .40* | Form of Stock Option Award Agreement under the 2008 Equity Incentive Plan (used prior to the completion of this offering). | ||
| 10 | .41†* | Agreement for Antibody Discovery and Development between the registrant and Affitech AS dated July 25, 2008. | ||
| 10 | .42†* | Exclusive Technology Option Agreement between the registrant, Patobios Limited, Susan R. George, M.D., Brian F. O’Dowd, Ph.D. and U.S. Bank National Association as escrow agent dated September 4, 2008. | ||
| 10 | .43* | Loan and Security Agreement between the registrant and BlueCrest Capital Finance, L.P. dated September 12, 2008. | ||
| 10 | .44* | Promissory Note issued by the registrant to BlueCrest Capital Finance, L.P. dated September 12, 2008. | ||
| 10 | .45* | Promissory Note issued by the registrant to BlueCrest Capital Finance, L.P. dated December 23, 2008. | ||
| 10 | .46†* | Agreement for Antibody Development between the registrant and North Coast Biologics LLC dated October 31, 2008. | ||
Table of Contents
| Exhibit | ||||
Number | Description | |||
| 10 | .47†* | Patent Assignment Agreement between the registrant and Roberto Ciccocioppo, Ph.D. dated February 23, 2009. | ||
| 10 | .48* | Amendment to Exercise Notice and Restricted Stock Purchase Agreements between the registrant and Richard J. Klein dated April 29, 2009. | ||
| 21 | .1* | List of significant subsidiaries of the registrant. | ||
| 23 | .1 | Consent of Independent Registered Public Accounting Firm. | ||
| 23 | .4 | Consent of Wilson Sonsini Goodrich & Rosati, Professional Corporation (included in Exhibit 5.1). | ||
| 24 | .1* | Power of Attorney. | ||
| 99 | .1* | Consent of The Reimbursement Group. | ||
| * | Previously Filed. | |
| ** | To be filed by amendment. | |
| † | Confidential treatment will be requested for portions of this exhibit. These portions will be omitted from this Registration Statement and will be filed separately with the Securities and Exchange Commission. |