UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
________________________________________
FORM 10-K
________________________________________
(Mark One)
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2018
or
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 001-34475
________________________________________
OMEROS CORPORATION
(Exact name of registrant as specified in its charter)
________________________________________
| Washington | 91-1663741 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification Number) | |
201 Elliott Avenue West Seattle, Washington | 98119 | |
| (Address of principal executive offices) | (Zip Code) | |
(206) 676-5000
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| Common Stock, $0.01 par value per share | The Nasdaq Stock Market LLC |
| (Title of each class) | (Name of each exchange on which registered) |
Securities registered pursuant to Section 12(g) of the Act:
None
___________________
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ¨ No x
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ¨ No x
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes x No ¨
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | x | Accelerated filer | ¨ | |||
| Non-accelerated filer | ¨ | Smaller reporting company | ¨ | |||
| Emerging growth company | ¨ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ¨ No x
The aggregate market value of the voting and non-voting common stock held by non-affiliates of the registrant as of the last business day of the registrant’s most recently completed second fiscal quarter was $827,171,756.
As of February 25, 2019, the number of outstanding shares of the registrant’s common stock, par value $0.01 per share, was 49,011,735.
DOCUMENTS INCORPORATED BY REFERENCE
Specified portions of the registrant’s proxy statement with respect to the 2019 Annual Meeting of Shareholders to be held June 7, 2019, which is to be filed pursuant to Regulation 14A within 120 days after the end of the registrant’s fiscal year ended December 31, 2018, are incorporated by reference into Part III of this Form 10-K.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, or the Exchange Act, which are subject to the “safe harbor” created by those sections for such statements. Forward-looking statements are based on our management’s beliefs and assumptions and on information currently available to our management. All statements other than statements of historical fact are “forward-looking statements.” Terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “goal,” “intend,” “likely,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would,” and similar expressions and variations thereof are intended to identify forward-looking statements, but these terms are not the exclusive means of identifying such statements. Examples of these statements include, but are not limited to, statements regarding:
| • | our expectations related to demand for OMIDRIA® (phenylephrine and ketorolac intraocular solution) 1%/0.3% from wholesalers, ambulatory surgery centers, or ASCs, and hospitals, and our expectations regarding OMIDRIA product sales; |
| • | our plans for marketing and distribution of OMIDRIA and our estimates of OMIDRIA chargebacks and rebates, distribution fees and product returns; |
| • | our estimates regarding how long our existing cash, cash equivalents, short-term investments and revenues will be sufficient to fund our anticipated operating expenses, capital expenditures and debt service obligations; |
| • | our expectations related to obtaining a permanent separate or similar reimbursement for OMIDRIA from the Centers for Medicare and Medicaid Services, or CMS, particularly for periods after September 30, 2020; |
| • | our expectations regarding the clinical, therapeutic and competitive benefits of OMIDRIA and our product candidates; |
| • | our ability to design, initiate and/or successfully complete clinical trials and other studies for our products and product candidates and our plans and expectations regarding our ongoing or planned clinical trials, including for our lead MASP-2 inhibitor, narsoplimab (also referred to as OMS721), and for OMS527 and OMS906; |
| • | in our narsoplimab clinical programs, our expectations regarding: whether enrollment in any or all ongoing and planned Phase 3 and Phase 2 clinical trials will proceed as expected; whether we can capitalize on the financial and regulatory incentives provided by orphan drug designations granted by the U.S. Food and Drug Administration, or FDA, the European Commission, or EC, or the European Medicines Agency, or EMA; and whether we can capitalize on the financial and regulatory incentives provided by fast-track and/or breakthrough therapy designations granted by FDA; |
| • | our expectations regarding clinical plans and anticipated or potential paths to regulatory approval of narsoplimab by FDA and/or EMA in hematopoietic stem cell transplant-associated thrombotic microangiopathy (HSCT-TMA), Immunoglobulin A (IgA) nephropathy, and/or atypical hemolytic uremic syndrome (aHUS); |
| • | whether and when a Biologics License Application, or BLA, may be filed with FDA for narsoplimab in any indication and whether FDA will grant accelerated or regular (full) approval for narsoplimab in any indication; |
| • | whether and when a marketing authorization application, or MAA, may be filed with EMA for narsoplimab in any indication, and whether EMA will grant approval for narsoplimab in any indication; |
| • | our anticipation that we will rely on contract manufacturers to manufacture OMIDRIA for commercial sale and to manufacture our product candidates for purposes of clinical supply and in anticipation of potential commercialization; |
| • | our ability to enter into acceptable arrangements with potential corporate partners or contract service providers, including with respect to OMIDRIA or our product candidates, and our ability and plans to effect any such arrangement with respect to OMIDRIA in the European Union, or EU, or in other foreign countries; |
| • | our ability to raise additional capital through the capital markets or through one or more corporate partnerships, equity offerings, debt financings, collaborations, licensing arrangements or asset sales; |
| • | our expectations about the commercial competition that OMIDRIA and our product candidates, if commercialized, face or may face; |
| • | the expected course and costs of existing claims, legal proceedings and administrative actions, our involvement in potential claims, legal proceedings and administrative actions, and the merits, potential outcomes and effects of both existing and potential claims, legal proceedings and administrative actions, as well as regulatory determinations, on our business, prospects, financial condition and results of operations; |
| • | the extent of protection that our patents provide and that our pending patent applications will provide, if patents are issued from such applications, for our technologies, programs, products and product candidates; |
| • | the factors on which we base our estimates for accounting purposes and our expectations regarding the effect of changes in accounting guidance or standards on our operating results; and |
i
| • | our expected financial position, performance, revenues, growth, costs and expenses, magnitude of net losses and the availability of resources. |
Our actual results could differ materially from those anticipated in these forward-looking statements for many reasons, including the risks, uncertainties and other factors described in Item 1A of Part I of this Annual Report on Form 10-K under the heading “Risk Factors” and in Item 7 of Part II under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and in our other filings with the Securities and Exchange Commission, or SEC. Given these risks, uncertainties and other factors, actual results or anticipated developments may not be realized or, even if substantially realized, they may not have the expected consequences to or effects on our company, business or operations. Accordingly, you should not place undue reliance on these forward-looking statements, which represent our estimates and assumptions only as of the date of the filing of this Annual Report on Form 10-K. You should read this Annual Report on Form 10-K completely and with the understanding that our actual results in subsequent periods may materially differ from current expectations. Except as required by applicable law, including the securities laws of the United States and the rules and regulations of the SEC, we assume no obligation to update or revise any forward-looking statements contained herein, whether as a result of any new information, future events or otherwise.
ii
OMEROS CORPORATION
ANNUAL REPORT ON FORM 10-K FOR THE YEAR ENDED DECEMBER 31, 2018
INDEX
| Page | |
iii
PART I
This Annual Report on Form 10-K contains forward-looking statements reflecting our current expectations that involve risks and uncertainties. Actual results may differ materially from those discussed in these forward-looking statements due to a number of factors, including those set forth in the section entitled “Risk Factors” and elsewhere in this Annual Report. Please refer to the special note regarding forward-looking statements at the beginning of this Annual Report on Form 10-K for further information.
| ITEM 1. | BUSINESS |
Overview
We are a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market as well as orphan indications targeting inflammation, complement-mediated diseases, disorders of the central nervous system and immune-related diseases, including cancers.
Our drug product OMIDRIA® is marketed in the United States, or U.S., for use during cataract surgery or intraocular lens, or IOL, replacement. In our pipeline we have clinical-stage development programs focused on: complement-associated thrombotic microangiopathies; complement-mediated glomerulonephropathies; and addictive and compulsive disorders. In addition, we have a diverse group of preclinical programs and two platforms: one capable of unlocking new G protein-coupled receptor, or GPCR, drug targets and the other used to generate antibodies. We have retained control of all commercial rights for OMIDRIA and each of our product candidates and programs.
Commercial Product -- OMIDRIA® (phenylephrine and ketorolac intraocular solution) 1%/0.3%
Overview. OMIDRIA is approved by the FDA for use during cataract surgery or IOL replacement to maintain pupil size by preventing intraoperative miosis (pupil constriction) and to reduce postoperative ocular pain. Outside of the U.S., we have received approval from the EC to market OMIDRIA in the European Economic Area, or EEA, for use during cataract surgery and other IOL replacement procedures for maintenance of intraoperative mydriasis (pupil dilation), prevention of intraoperative miosis and reduction of acute postoperative ocular pain. OMIDRIA is a proprietary drug product containing two active pharmaceutical ingredients, or APIs: ketorolac, an anti-inflammatory agent, and phenylephrine, a mydriatic, or pupil dilating, agent. Cataract and other lens replacement surgery involves replacement of the original lens of the eye with an artificial intraocular lens. These procedures are typically performed to replace a lens opacified by a cataract and/or to correct a refractive error. OMIDRIA is added to standard irrigation solution used during cataract and lens replacement surgery and is delivered intracamerally, or within the anterior chamber of the eye, to the site of the surgical trauma throughout the procedure. Preventing pupil constriction is essential for these procedures and, if miosis occurs, the risk of damaging structures within the eye and other complications increases, as does the operating time required to perform the procedure.
United States. We launched OMIDRIA in the U.S. in the second quarter of 2015 and sell OMIDRIA primarily through wholesalers which, in turn, sell to ASCs and hospitals. CMS, the federal agency responsible for administering the Medicare program, granted transitional pass-through reimbursement status for OMIDRIA in 2014, effective from January 1, 2015 through December 31, 2017. Pass-through status allows for separate payment (i.e., outside the packaged payment rate for the surgical procedure) under Medicare Part B. In March 2018, the Consolidated Appropriations Act of 2018, or the Appropriations Act, was signed into law. The Appropriations Act includes a provision by which Congress extended pass-through reimbursement status for a small number of drugs, including OMIDRIA, used during procedures performed on Medicare Part B fee-for-service patients for an additional two years, running from October 1, 2018 until October 1, 2020. We also continue to pursue permanent separate reimbursement for OMIDRIA. In the recently released 2019 final rule for CMS’ outpatient prospective payment system, or OPPS, CMS indicated that, in the ASC setting, it will separately pay for non-opioid drugs used during surgery that have an FDA-approved indication for postoperative pain relief payment and are currently packaged with the procedure in calendar year 2019. Although OMIDRIA is not specifically named because it currently is paid separately, we believe that OMIDRIA meets this definition and would qualify for separate payment under this provision if it is continued in subsequent years. The OPPS Final Rule also states that CMS will consider in future rule-making a policy that pays separately for drugs used during cataract surgery that have an FDA-approved indication to address postoperative issues. We believe that OMIDRIA also meets this definition. We are continuing to confirm these beliefs and to pursue other avenues of permanent separate payment or similar reimbursement for OMIDRIA beyond September 30, 2020, but can provide no assurance that these efforts will be successful. We also continue to pursue expansion of reimbursement for OMIDRIA by Medicare Advantage and other third-party payers.
OMIDRIA revenues in the first three quarters of 2018 were significantly reduced due to the expiration of pass-through reimbursement status on January 1, 2018. After pass-through reimbursement was reinstated effective October 1, 2018, weekly sales of OMIDRIA increased substantially from levels seen during the first nine months of the year, and fourth quarter 2018
1
OMIDRIA revenues were $22.0 million, our highest quarterly revenue to date. See Part II, Item 7, “Management’s Discussion and Analysis-Results of Operations” for further discussion of OMIDRIA reimbursement and pricing.
We have implemented a variety of programs and arrangements to facilitate the availability of OMIDRIA to cataract and IOL replacement patients in the U.S., including the following:
| • | various purchase volume-discount programs for OMIDRIA; |
| • | agreements to enable discounts on qualifying purchases of OMIDRIA by certain U.S. government purchasers and other eligible entities (e.g., 340B-eligible hospitals and clinics); and |
| • | the OMIDRIAssure® Reimbursement Services Program, or OMIDRIAssure. |
OMIDRIAssure provides coverage and reimbursement support services for surgeons and facilities to help remove uncertainties about coding, billing and coverage of OMIDRIA and to enable better access to the drug for patients facing financial barriers. Under our “Equal Access” patient assistance program, financially eligible uninsured and government-insured patients receive OMIDRIA free of charge for use during surgery. Through our “We Pay the Difference” program we pay the facility, on behalf of commercially insured patients, the difference between the facility’s acquisition cost for OMIDRIA, after accounting for any applicable volume discounts, and the amount covered by the patient’s insurance.
In December 2017, the FDA approved our supplemental new drug application, or sNDA, following review of efficacy and safety data from a pediatric clinical trial, expanding the indication for OMIDRIA to include use in pediatric patients (from birth through 17 years old). In addition to the label expansion now including both pediatric and adult patients, the FDA also granted OMIDRIA an additional six months of U.S. market exclusivity, subject to any licenses that we may have (including pursuant to the Settlement Agreements with Par and Lupin, which are described under “License and Development Agreements” below) or into which we may enter. Under section 505A of the Federal Food, Drug, and Cosmetic Act, or FDCA, this six-month extension of market exclusivity is attached to the term of the drug’s patents listed in FDA’s Orange Book.
European Union and other International Territories. In July 2018, we placed OMIDRIA on the market in the EU on a limited basis, which maintained the ongoing validity of the European marketing authorization for OMIDRIA. Decisions about price and reimbursement for OMIDRIA are made on a country-by-country basis and may be required before marketing may occur in a particular country. At this time we do not expect to see significant sales of OMIDRIA in any countries within the EEA or other international territories if we are unable to complete a broad sales launch in any such country either independently or through partnerships for the marketing and distribution of OMIDRIA. A marketing authorization will cease to be valid if a product previously placed on the market is no longer actually present on the market for three consecutive years. Timing of any partnerships or independent launch depends on numerous factors, including domestic sales of OMIDRIA, completion of mutual diligence exercises and/or entry into suitable agreements with contract service vendors.
We have an exclusive supply and distribution agreement with ITROM Trading Drug Store, or ITROM, for the sale of OMIDRIA in the Kingdom of Saudi Arabia, the United Arab Emirates and certain other countries in the Middle East. Under our agreement, ITROM is responsible for obtaining marketing authorizations for OMIDRIA on our behalf and for promoting, marketing, selling and distributing product supplied by us within the licensed territory. ITROM began selling OMIDRIA in December 2016 on a limited basis in the Kingdom of Saudi Arabia. Revenues to date under our agreement with ITROM have not been material.
Our Product Candidates and Development Programs
Our clinical product candidates consist of the following:
2
| Product Candidate/Program | Targeted Disease(s) | Development Status | Next Expected Milestone | Worldwide Rights |
| Clinical | ||||
| Narsoplimab (OMS721/MASP-2) - Lectin Pathway Disorders | Hematopoietic Stem-Cell Transplant-Associated Thrombotic Microangiopathy (HSCT-TMA) | Phase 3 | Preparation and Submission of Biologics License Application (BLA) and Marketing Authorization Application (MAA) | Omeros (In-licensed) |
| Narsoplimab (OMS721/MASP-2) - Lectin Pathway Disorders | Immunoglobulin A Nephropathy (IgAN) | Phase 3 | Complete Phase 3 Patient Enrollment or Perform 36-Week Assessment of Proteinuria | Omeros (In-licensed) |
| Narsoplimab (OMS721/MASP-2) - Lectin Pathway Disorders | Atypical Hemolytic Uremic Syndrome (aHUS) | Phase 3 | Complete Phase 3 Patient Enrollment | Omeros (In-licensed) |
| Narsoplimab (OMS721/MASP-2) - Lectin Pathway Disorders | Lupus Nephritis and Other Renal Diseases | Phase 2 | Review Data; Determine Whether to Initiate Phase 3 Program | Omeros (In-licensed) |
| PDE10 (OMS824) - Central Nervous System (CNS) Disorders | Huntington’s Disease; Schizophrenia | Phase 2 (1) | Internal Review of Compounds and Indications | Omeros |
| PPARγ (OMS405) - Addiction | Opioid and Nicotine Addiction | Phase 2 | Further Refine Development Path | Omeros |
| PDE7 (OMS527) | Addictions and Compulsive Disorders; Movement Disorders | Phase 1 | Complete Phase 1 Trial and Evaluate Data | Omeros (Compounds In-licensed) |
| (1) | No clinical trials for OMS824 are ongoing. Plans for continuation of the OMS824 program will be based on internal ongoing work and discussions with the FDA. Clinical trials in our Huntington’s program are authorized by the FDA to progress subject to dosing limitations. In light of these dosing limitations, we are currently focused on assessing the relative advantages of a number of our back-up compounds and potential indications. We are not authorized by the FDA to conduct further clinical trials in our schizophrenia program until we submit a study protocol to the FDA for review. For additional information, see “Other Clinical Programs-PDE10 Programs-OMS824 for Huntington’s Disease and Schizophrenia” below. |
3
Our pipeline of development programs consist of the following:
| Product Candidate/Program | Targeted Disease(s) | Development Status | Next Expected Milestone | Worldwide Rights |
| Preclinical / Platform | ||||
| MASP-3 (OMS906) - Alternative Pathway Disorders | Paroxysmal Nocturnal Hemoglobinuria (PNH) and Other Alternative Pathway Disorders | Preclinical | Complete Manufacturing Scale-up of the Clinical Candidate for IND-Enabling Toxicology Studies | Omeros |
| MASP-2 - Small-Molecule Inhibitors | aHUS, IgAN, HSCT-TMA and Age-Related Macular Degeneration (AMD) | Preclinical | Optimize Compounds | Omeros (In-Licensed) |
| MASP-3 - Small-Molecule Inhibitors | PNH and Other Alternative Pathway Disorders | Preclinical | Continue Medicinal Chemistry and Advance Co-Crystallization Efforts | Omeros |
| GPCR Platform, including GPR174, GPR151, GPR161, and other Class A Orphan GPCRs | Immunologic, Immuno-oncologic, Metabolic, CNS, Cardiovascular (CV), Musculoskeletal & Other Disorders | Preclinical | Continue Drug Discovery and Selected Medicinal Chemistry for Class A Orphan, Class B Orphan and Non-Orphan GPCRs | Omeros |
| Antibody Platform | Metabolic, CV, Oncologic, Musculoskeletal & Other Disorders | Preclinical | Continue Developing Antibodies Targeting Lectin and Alternative Pathway of Complement System and Expanding Antibody Library | Omeros (In-licensed) |
MASP Inhibitor Clinical Programs
MASP-2 Program - Narsoplimab (OMS721) - Lectin Pathway Disorders
Overview. Mannan-binding lectin-associated serine protease-2, or MASP-2, is a novel pro-inflammatory protein target involved in activation of the complement system, which is an important component of the immune system. The complement system plays a role in the body’s inflammatory response and becomes activated as a result of tissue damage or trauma or microbial pathogen invasion. Inappropriate or uncontrolled activation of the complement system can cause diseases characterized by serious tissue injury. Three main pathways can activate the complement system: classical, alternative and lectin. MASP-2 is recognized as the effector enzyme of the lectin pathway and is required for the function of this pathway. Importantly, inhibition of MASP-2 has been demonstrated not to interfere with the antibody-dependent classical complement activation pathway, a critical component of the acquired immune response to infection the abnormal function of which is associated with a wide range of autoimmune disorders.
Our proprietary, patented lead human monoclonal antibody targeting MASP-2, which we have referred to as OMS721, has been assigned the nonproprietary name narsoplimab by the United States Adopted Names Council, in consultation with the World Health Organization’s International Nonproprietary Names Expert Committee. The current development focus for narsoplimab is diseases in which the lectin pathway has been shown to contribute to significant tissue injury and pathology. When not treated, these diseases are typically characterized by significant end organ injuries, such as kidney or central nervous system injury. Phase 3 clinical programs are in process for narsoplimab in: hematopoietic stem cell transplant-associated thrombotic microangiopathy, or HSCT-TMA; Immunoglobulin A, or IgA, nephropathy; and atypical hemolytic uremic syndrome, or aHUS. In addition, we have two ongoing phase 2 clinical trials evaluating narsoplimab, one in patients with thrombotic microangiopathies, or TMAs, and the other in renal diseases, currently focused on patients with IgA nephropathy.
Thrombotic Microangiopathies
Phase 2 Clinical Trial - TMAs. We have an ongoing Phase 2 clinical trial in patients with TMAs, including initially aHUS, HSCT-TMA and thrombotic thrombocytopenia, or TTP. Currently, patients with TTP or aHUS are no longer being enrolled in this study. To be eligible for enrollment, HSCT-TMA patients are required to be adults with post-transplant TMA persisting at least two weeks following calcineurin inhibitor modification (conservative management). The primary efficacy
4
endpoint of the study is change in platelet count. Additional efficacy endpoints include changes in lactate dehydrogenase, or LDH, and haptoglobin levels.
In March 2017, we announced positive data in patients with HSCT-TMA from the ongoing Phase 2 clinical trial. Statistically significant and clinically meaningful improvements in TMA disease activity were observed over the course of treatment, specifically in mean platelet count, mean LDH and mean haptoglobin. In October 2017, we announced the presentation by a trial investigator of a case report of a patient in this Phase 2 clinical trial whose post-transplant course was complicated by multiple episodes of steroid-refractory grade IV (life-threatening) graft-versus-host disease, or GVHD. The patient then presented with TMA and a recurrence of GVHD, which both resolved following narsoplimab treatment.
In February 2018 and April 2018, we reported new results in patients with HSCT-TMA from this ongoing Phase 2 study. The data demonstrate an increase in estimated median survival and in 100-day mortality compared to a literature-based control. In addition to and consistent with the survival data reported, updated assessments of platelet count, LDH, and haptoglobin - all markers of TMA activity - continued to demonstrate clinically meaningful and statistically significant improvements in the HSCT-TMA patients treated with narsoplimab. Improvement in transfusion requirements and organ function were also observed. No safety concerns were identified. Adverse events and causes of death were consistent with the patients’ underlying diseases.
This study has continued to enroll since those reports. Improvements have continued to be observed in narsoplimab-treated patients with HSCT-TMA. We plan to submit for regulatory approval with data from the currently treated patients, and the Phase 2 study will continue to gather additional exposure and follow-up data.
Phase 3 Program - HSCT-TMA. We are in Phase 3 of development for narsoplimab in HSCT-TMA. We anticipate that data from the currently treated patients with HSCT-TMA will provide adequate clinical basis for marketing approval. To support the BLA and MAA, we intend to amend the ongoing Phase 2 protocol to collect additional data on currently treated patients based on discussions with the FDA and the EMA regarding expectations for our marketing applications (BLA and MAA).
We have met recently with the FDA and with multiple European national regulatory authorities to discuss potential approval pathways for narsoplimab for the treatment of HSCT-TMA. Feedback from the European national regulatory authorities has been positive, and includes uniform support to submit an MAA for full approval of narsoplimab in HSCT-TMA. A recent meeting with FDA: (1) provided additional clarity on the primary endpoint for approval; (2) included guidance that data from patients in our existing open-label Phase 2 trial may comprise the clinical data required to assess the efficacy of narsoplimab for approval (e.g., no additional clinical trials nor historical control will be required); (3) the data may warrant regular (full) approval or accelerated approval; and (4) narsoplimab for HSCT-TMA is entitled to a rolling BLA submission. We will continue to meet with FDA to finalize endpoint criteria and the analysis plan and to agree on a schedule for a rolling submission. We have already written the nonclinical sections of the BLA. We also plan to request a meeting with the rapporteur and co-rapporteur in Europe, who will work with us through the MAA submission and review process for narsoplimab in HSCT-TMA. We have already applied for, and EMA confirmed, eligibility to EMA’s centralized review procedure, which allows submission of a single MAA that, if approved, authorizes the product to be marketed in all EU member states and EEA countries rather than requiring separate national approvals. We have also provided EMA with our letter of intent to submit an MAA. Close interactions with the FDA and European regulatory agencies are ongoing and we continue preparations for U.S. BLA and European MAA submissions.
In the U.S., the FDA has granted narsoplimab (1) breakthrough therapy designation in patients who have persistent TMA despite modification of immunosuppressive therapy, (2) orphan drug designation for the prevention (inhibition) of complement-mediated TMAs, and (3) orphan drug designation for the treatment of HSCT-TMA. The EC also granted narsoplimab designation as an orphan medicinal product for treatment in hematopoietic stem cell transplantation.
Phase 3 Program - aHUS. We have an ongoing Phase 3 clinical program in patients with aHUS with active sites in both the U.S. and Europe. The single-arm (i.e., no control arm), open-label Phase 3 clinical trial in patients with newly diagnosed or ongoing aHUS is enrolling. This trial is targeting approximately 40 patients for EMA approval and U.S. accelerated approval with 80 patients required for full approval in the U.S. Dosing consists of an initial IV loading followed by daily subcutaneous dosing. Based on discussions with the FDA and the EMA, we expect that the clinical package for the BLA would be similar to that which formed the basis of approval for Soliris® (eculizumab), which is marketed by Alexion Pharmaceuticals, Inc.
The FDA has granted to narsoplimab orphan drug designation for the prevention (inhibition) of complement-mediated TMAs and fast-track designation for the treatment of patients with aHUS.
5
Renal Disease
Phase 2 Clinical Trial - Renal Diseases. We have been conducting a Phase 2 clinical trial in patients with complement-associated renal diseases, specifically designed to cover: (i) IgA nephropathy; (ii) membranous nephropathy; (iii) lupus nephritis; and (iv) complement component (C3) glomerulopathy. An initial open-label cohort of patients completed treatment in May 2017. No patients with C3 glomerulopathy were enrolled. Treatment is ongoing for a second cohort of patients with IgA nephropathy. This cohort consists of an initial 12-week double-blind, placebo-controlled period followed by a 20-month open-label period in which all patients receive narsoplimab.
All patients in the initial open-label cohort of the trial were required to have high levels of urinary protein, or proteinuria (a marker used by nephrologists to assess disease activity), despite ongoing treatment with corticosteroids. These inclusion criteria were intended to ensure that study patients are unlikely to improve spontaneously. Patients in this open-label cohort were treated with narsoplimab for a total of 12 weeks: four weeks maintaining their entry corticosteroid dose; four weeks of corticosteroid tapering, if tolerated; and four weeks of resultant corticosteroid dose maintenance or discontinuation. Patients were then followed post-treatment for six weeks.
All four patients with IgA nephropathy in the initial open-label cohort demonstrated clinically meaningful and statistically significant improvement in proteinuria during the 18-week trial period. Follow-up data collected after patients completed treatment in the trial period showed that three of the four IgA nephropathy patients in this cohort maintained the proteinuria reduction (as measured by urine albumin/creatinine ratio) shown in the clinical trial during the follow-up period (assessed at 12, 11 and three months, respectively, after cessation of dosing). Numerical improvement in estimated glomerular filtration rate, or eGFR, a measure of renal function, was also observed in three of the four patients after the trial. Narsoplimab was well-tolerated in the clinical trial with fatigue and anemia being the most commonly reported adverse events.
In the second cohort evaluating patients with IgA nephropathy in the U.S., patients must have elevated levels of urinary protein but are not treated with corticosteroids. Patients in this cohort were treated for 12 weeks with weekly dosing of either narsoplimab or placebo, then followed for six weeks with no treatment. After week 18, patients initially treated with placebo and patients initially treated with narsoplimab were able to receive additional 12-week courses of open-label dosing with narsoplimab at investigator discretion. Patients may receive more than one 12-week course of narsoplimab and will be followed for a total of two years. This cohort of the trial is ongoing.
In October 2018, we reported interim results from the second cohort of IgA nephropathy patients in the U.S. Unlike the first open-label cohort comprised of four IgA nephropathy patients who were taking corticosteroids at the time of study enrollment, patients in the second cohort are not taking steroids. The cohort included nine evaluable patients. At week 18, median reduction in proteinuria was 18.4% in the five narsoplimab-treated patients and 18.0% in the four placebo patients. This study did not include a run-in period during which time renin-angiotensin system, or RAS, blockade is optimized and patient compliance with RAS blockade is improved. The placebo response in this first 18-week period is consistent with improvement in placebo groups seen during the run-in periods of other studies in IgA nephropathy patients. After week 18, eight of the nine evaluable patients entered the extended dosing and observation period, all of whom received narsoplimab treatment during this period. At the last observation point for each the eight patients (between 31 and 54 weeks post-baseline) the median reduction from baseline proteinuria was 61%. Five of the eight patients have achieved greater than 50% proteinuria reductions (median reduction of 65%), with two of those five patients having received their last narsoplimab administration five months earlier. eGFR, measurements have also remained stable, consistent with preservation of renal function. Despite the small cohort of patients, and although median reductions in proteinuria in the placebo group were comparable to those in the narsoplimab group following the initial 12-week course of treatment, we and international experts in IgA nephropathy with whom we have consulted and who have reviewed the individual patient data believe that these data are positive and supportive of a substantial disease modifying effect. With proteinuria reduction in five of eight patients ranging from greater than 50% to approximately 70%, this magnitude is consistent with what we previously reported from the first cohort of this clinical trial. This trial also includes a third cohort of patients with all study sites for this cohort located in Hong Kong. Like the first and second IgA cohorts, this is a small study, slated to enroll approximately 10 patients. It has been redesigned to focus on subcutaneous dosing and associated biomarkers. Given what looks to be good activity with subcutaneous dosing, we will continue to determine the pharmacokinetics and pharmacodynamics of narsoplimab in IgA nephropathy patients when administered subcutaneously over a 12-week period. Data from this cohort will support the ongoing Phase 3 program in IgA nephropathy and planned lifecycle management for narsoplimab.
Also, in March 2017 we reported encouraging results in lupus nephritis patients from the first cohort in the Phase 2 renal disease trial. Four of five patients showed a substantial (mean of approximately 70%) reduction in 24-hour urine protein excretion over the treatment period. The fifth patient experienced a systemic disease flare and demonstrated a substantial increase in 24-hour urine protein excretion. The majority of lupus responders were able to taper their steroid doses. We are reviewing these data for possible further development in lupus nephritis.
6
Phase 3 Program - IgA Nephropathy. Patient enrollment is ongoing in our Phase 3 clinical trial evaluating narsoplimab, which is referred to as ARTEMIS-IGAN. The single Phase 3 trial design is a randomized, double-blind, placebo-controlled multicenter trial in patients at least 18 years of age with biopsy-confirmed IgA nephropathy and with 24-hour urine protein excretion greater than 1 g/day at baseline on optimized RAS blockade. This trial includes a run-in period. Initially, patients are expected to receive an IV dose of study drug each week for 12 weeks; additional weekly dosing can be administered to achieve optimal response. The primary endpoint, which could suffice for full or accelerated approval depending on the effect size, is reduction in proteinuria at 36 weeks after the start of dosing. The trial is designed to allow intra-trial adjustment in sample size. For the purposes of safety and efficacy assessments, the initial sample size for the proteinuria endpoint is estimated at 140 patients in each of the treatment and placebo groups. This will include a subset of patients with high levels of proteinuria (i.e., equal to or greater than 2 g/day) at baseline, and a substantial improvement at 36 weeks in this subset of patients alone could potentially form the basis for approval. We believe that the trial design will allow assessment for either full or accelerated approval at 36 weeks based on proteinuria results either (1) across the general population of study patients or (2) in the high-proteinuria subset of patients. In the event of full approval, eGFR becomes a safety endpoint only. In the event that the primary endpoint at 36 weeks results in accelerated approval from the FDA, change in eGFR is expected to be assessed at approximately three years after the start of dosing. These eGFR data, if satisfactory, would then likely form the basis for full approval. In response to investigators’ concerns about extended withholding of narsoplimab treatment from any high-proteinuria patient initially randomized to the placebo-treated group, the FDA will allow patients in that sub-population open-label treatment with narsoplimab after at least 1 year of blinded treatment.
In the U.S., narsoplimab has received from the FDA breakthrough therapy and orphan drug designations for the treatment of IgA nephropathy. In Europe narsoplimab has received orphan drug designation from the EC in patients with IgA nephropathy.
Expanded Access / Compassionate Use. We have received requests from investigators and other physicians for expanded access to narsoplimab. Expanded access, sometimes called “compassionate use,” is the use of an investigational medical product outside of a clinical trial. Expanded access is permitted by the FDA and other regulatory agencies under specific circumstances. Narsoplimab has been provided to several patients in compassionate use situations.
Licensing Arrangements. We hold worldwide exclusive licenses to rights related to MASP-2, the antibodies targeting MASP-2 and the therapeutic applications for those antibodies from the University of Leicester, from its collaborator, the Medical Research Council at Oxford University, or MRC, and from Helion Biotech ApS, or Helion. For a more detailed description of these licenses, see “License and Development Agreements” below.
MASP Inhibitor Preclinical Programs
MASP-3 Program - OMS906 - Alternative Pathway Disorders
Overview. As part of our MASP program, we have identified mannan-binding lectin-associated serine protease-3, or MASP-3, which has been shown to be the key activator of the complement system’s alternative pathway, or APC, and we believe that we are the first to make this and related discoveries associated with the APC. The complement system is part of the immune system’s innate response, and the APC is considered the amplification loop within the complement system. MASP-3 is responsible for the conversion of pro-factor D to factor D, and converted factor D is necessary for the activation of the APC. Based on our alternative pathway-related discoveries, we have expanded our intellectual property position to protect our inventions stemming from these discoveries beyond MASP-2-associated inhibition of the lectin pathway to include inhibition of the alternative pathway.
Our current primary focus in this program is developing MASP-3 inhibitors for the treatment of disorders related to the APC. We believe that MASP-3 inhibitors have the potential to treat patients suffering from a wide range of diseases and conditions, including: paroxysmal nocturnal hemoglobinuria, or PNH; C3 glomerulopathy; multiple sclerosis; arthritis; traumatic brain injury; neuromyelitis optica; pauci-immune necrotizing crescentic glomerulonephritis; disseminated intravascular coagulation; age-related macular degeneration; asthma; dense deposit disease; Bechet’s disease; aspiration pneumonia; TMA; ischemia-reperfusion injury; Guillain Barre syndrome; Alzheimer’s disease; amylotrophic lateral sclerosis; systemic lupus erythematosus; diabetic retinopathy; uveitis; chronic obstructive pulmonary disease; transplant rejection; acute respiratory distress syndrome; antineutrophil cytoplasmic antibody-associated vasculitis; anti-phospholipid syndrome; atherosclerosis; myasthenia gravis and others.
Our OMS906 program has generated positive data in a well-established animal model associated with PNH including in non-human primates. The program has also generated positive data in a well-established model of arthritis. In preparation for clinical trials, the manufacturing scale-up process is underway for a MASP-3 inhibitor antibody and we are currently targeting PNH as the first clinical indication for OMS906. Clinical trials are slated to begin in the first part of 2020.
7
Licensing Arrangements. We jointly own and hold worldwide exclusive license rights related to therapeutic applications for inhibiting MASP-3 from the University of Leicester. For a more detailed description of these licenses, see “License and Development Agreements” below.
Other MASP Inhibitor Preclinical Programs
We have generated positive preclinical data from MASP-2 inhibition in in vivo models of AMD, myocardial infarction, diabetic neuropathy, stroke, ischemia-reperfusion injury, and other diseases and disorders.
We are also developing small-molecule inhibitors of MASP-2 designed for oral administration that we are targeting for initiation of clinical trials in 2020, as well as additional antibodies targeting MASP-2. Development efforts are also directed to small-molecule inhibitors of MASP-3 and bispecific small- and large-molecule inhibitors of MASP-2/-3.
Other Clinical Programs
PDE7 Program - OMS527
Overview. Our phosphodiesterase 7, or PDE7, program is based on our discoveries of previously unknown links between PDE7 and any addiction or compulsive disorder, and between PDE7 and any movement disorders, such as Parkinson’s disease. PDE7 appears to modulate the dopaminergic system, which plays a significant role in regulating both addiction and movement. We believe that PDE7 inhibitors could be effective therapeutics for the treatment of addictions and compulsions as well as for movement disorders. Data generated in preclinical studies support the use of PDE7 inhibitors in both of these therapeutic areas. We have initiated a Phase 1 single-ascending- and multiple-ascending-dose clinical trial designed to assess the safety and pharmacokinetics of the compound in healthy subjects. We have completed dosing in all six cohorts in the single-ascending-dose portion of the trial, including a cohort to assess whether pharmacokinetics is affected by food. Dosing in the first two cohorts in the multiple-ascending-dose portion of the trial is also complete. The compound to date has been well tolerated and pharmacokinetic data support once-daily dosing, with or without food. Completion of the Phase 1 trial is expected in the second or third quarter of 2019. Following Phase 1 completion, if successful, we plan to conduct a Phase 2a study targeting nicotine addiction.
Exclusive License Agreement with Daiichi Sankyo Co., Ltd. We hold an exclusive license to certain PDE7 inhibitors claimed in patents and pending patent applications owned by Daiichi Sankyo Co., Ltd. (successor-in-interest to Asubio Pharma Co., Ltd.), or Daiichi Sankyo, for use in the treatment of movement, addiction and compulsive disorders as well as other specified indications. For a more detailed description of our agreement with Daiichi Sankyo, see “License and Development Agreements” below.
PDE10 Programs - OMS824 for Huntington’s Disease and Schizophrenia
Overview. Phosphodiesterase 10, or PDE10, is an enzyme that is expressed in areas of the brain strongly linked to diseases that affect cognition, including Huntington’s disease and schizophrenia. Cognitive dysfunction occurs early in these diseases and is responsible for substantial disability. PDE10 inhibitors have been shown to be effective in multiple animal models of behavior and cognition, and there remain substantial unmet clinical needs with current treatments. Our proprietary compound OMS824 inhibits PDE10 and is being developed in clinical programs for the treatment of cognitive disorders, including Huntington’s disease and schizophrenia. OMS824 has received orphan drug designation for the treatment of Huntington’s disease and fast-track designation for the treatment of cognitive impairment in patients with Huntington’s disease.
Clinical Trials. OMS824 is in a Phase 2 clinical program for the treatment of Huntington’s disease and a Phase 2 clinical program evaluating OMS824 for the treatment of schizophrenia. We are also evaluating other neurological indications for OMS824. The FDA has approved the advancement of clinical trials in Huntington’s disease with our lead candidate, subject to dosing limitations. Given the dosing limitations, we are currently focused on assessing the relative advantages of a number of our back-up compounds and potential alternative indications. Clinical trials evaluating OMS824 in schizophrenia remain suspended at the request of the FDA until we submit a protocol for a schizophrenia trial and receive the Agency’s clearance to proceed.
Funding Agreement with The Stanley Medical Research Institute. Our preclinical development of OMS824 was funded by The Stanley Medical Research Institute, or SMRI, a non-profit corporation that supports research on the causes and treatment of schizophrenia and bipolar disorder. For a more detailed description of our agreement with SMRI, see “License and Development Agreements” below.
8
PPARγ Program - OMS405
Overview. In our peroxisome proliferator-activated receptor gamma, or PPARγ, program, we are developing proprietary compositions that include PPARγ agonists for the treatment and prevention of addiction to substances of abuse, which may include opioids, nicotine and alcohol. We believe that Omeros is the first to demonstrate a link between PPARγ and addiction disorders. Data from clinical studies and from animal models of addiction suggest that PPARγ agonists could be efficacious in the treatment of a wide range of addictions.
Clinical trials. Our collaborators at The New York State Psychiatric Institute have completed two Phase 2 clinical trials related to our PPARγ program. These studies evaluated a PPARγ agonist, alone or in combination with other agents, for treatment of addiction to heroin and to nicotine. Recently published results of the heroin study demonstrated that, although not altering the reinforcing or positive subjective effects of heroin, the PPARγ agonist significantly reduced heroin craving and overall anxiety. The National Institute on Drug Abuse provided substantially all of the funding for these clinical trials and solely oversaw the conduct of these trials. We have the right or expect to be able to reference the data obtained from these studies for subsequent submissions to the FDA and continue to retain all other rights in connection with the PPARγ program. We have also reported positive results (i.e., decreased cravings and protection of brain white matter) from a Phase 2 clinical trial conducted by an independent investigator evaluating the effects of a PPARγ agonist in patients with cocaine use disorder.
Patent Assignment Agreement with Roberto Ciccocioppo, Ph.D. We acquired the patent applications and related intellectual property rights for our PPARγ program in February 2009 from Roberto Ciccocioppo, Ph.D., of the Università di Camerino, Italy, pursuant to a patent assignment agreement. For a more detailed description of our agreement with Dr. Ciccocioppo, see “License and Development Agreements” below.
Preclinical Programs and Platforms
GPCR Platform
Overview. GPCRs, which are cell surface membrane proteins involved in mediating both sensory and nonsensory functions, comprise one of the largest families of proteins in the genomes of multicellular organisms. Sensory GPCRs are involved in the perception of light, odors, taste and sexual attractants. Non-sensory GPCRs are involved in metabolism, behavior, reproduction, development, hormonal homeostasis and regulation of the central nervous system. The vast majority of GPCR drug targets are non-sensory. Although GPCRs form a super-family of receptors, individual GPCRs display a high degree of specificity and affinity for the functionally active molecules, or ligands, that bind to a given receptor. Ligands can either activate the receptor (agonists) or inhibit it (antagonists and inverse agonists). When activated by its ligand, the GPCR interacts with intracellular G proteins, resulting in a cascade of signaling events inside the cell that ultimately leads to the particular function linked to the receptor. Without a known ligand, there is no template from which medicinal chemistry efforts can be readily initiated, nor a means to identify the GPCR’s signaling pathway and, therefore, drugs are very difficult to develop against orphan GPCRs. “Unlocking” these orphan GPCRs by identifying one or more of their respective ligands could lead to the development of drugs that act at these new targets.
To our knowledge, despite efforts by others in the biopharmaceutical industry and academic community, Omeros’ technology is the first commercially viable technology capable of identifying ligands of orphan GPCRs in high throughput. We have developed a proprietary cellular redistribution assay, or CRA, which we use in a high-throughput manner to identify synthetic ligands, including antagonists, agonists and inverse agonists, that bind to and affect the function of orphan GPCRs. We have screened Class A orphan GPCRs against our small-molecule chemical libraries using the CRA. As of February 12, 2019, we had identified and confirmed compounds that interact with 54 of the 81 Class A orphan GPCRs linked to a wide range of indications including cancer as well as metabolic, cardiovascular, immunologic, inflammatory and central nervous system disorders. We are conducting in vitro and in vivo preclinical efficacy studies and optimizing compounds for a number of targets including: GPR151, linked to schizophrenia and cognition; GPR161, which is associated with triple negative breast cancer and various sarcomas; GPR174, which is involved in the modulation of the immune system and, in animal and ex vivo human studies, increases cytokine production and inhibits production of regulatory T cells, or “T-regs,” and checkpoint molecules, all of which are known to be important in cancer, organ transplantation and autoimmune disease, such as multiple sclerosis.
In addition to Class A orphan GPCRs, we have screened orphan and non-orphan Class B receptors. Class B GPCRs have large extracellular domains and their natural ligands are generally large peptides, making the development of orally active, small-molecule drugs against these receptors, such as glucagon and parathyroid hormone, a persistent challenge. Our CRA technology finds functionally active small molecules for GPCRs, which we believe could lead to the development of oral medications for many of the Class B GPCRs. While our focus to date has remained on Class A orphan GPCRs, we have identified and confirmed sets of compounds that interact selectively with, and modulate signaling of, a small subset of Class B GPCRs, namely glucagon-like peptide-1 receptor, or GLP-1R, and parathyroid hormone 1 receptor, or PTH-1R.
9
GPCR Platform Funding Agreements with Vulcan Inc. and the Life Sciences Discovery Fund. In October 2010, we entered into funding agreements for our GPCR program with Vulcan Inc. and its affiliate, which we refer to collectively as Vulcan, and with the Life Sciences Discovery Fund Authority, or LSDF, a granting agency of the State of Washington. For a more detailed description of these agreements, see “License and Development Agreements” below.
Antibody Platform
Overview. Our proprietary ex vivo platform for the discovery of novel, high-affinity monoclonal antibodies, which was in-licensed from the University of Washington and then further developed by our scientists, utilizes a chicken B-cell lymphoma cell line. It has successfully generated diverse antibodies that can be readily engineered. This platform offers several advantages over other antibody platforms. The ex vivo immunizations of our proprietary cell line are significantly more rapid than whole animal immunizations and conventional hybridoma technology. By avoiding immunization of mice or other animals, we believe the antibodies we generate from this platform are not limited by immunological tolerance. In addition, our platform is capable of producing novel antibodies against difficult targets, such as highly homologous proteins, enzymes, and receptors with short extracellular domains. Chicken antibodies also have unique features that enable binding capabilities distinct from mammalian antibodies.
Using our platform and other know-how and techniques, we have generated antibodies to several clinically significant targets, including highly potent antibodies against MASP-2, MASP-3 and MASP-1, and our platform continues to add to our pipeline antibodies against additional important targets.
Asset Purchase Agreement with Xori Corporation. In February 2012 we entered into an Asset Purchase Agreement, or the Xori APA, with Xori Corporation, or Xori, pursuant to which we acquired all of Xori’s rights and obligations in certain license and material transfer agreements, intellectual property, antibodies and other assets related to our antibody platform. We are obligated to make development and research-related milestone payments to Xori.
Exclusive License Agreement with the University of Washington. We hold a worldwide exclusive license to patent rights related to our antibody platform from the University of Washington. For a more detailed description of this agreement, see “License and Development Agreements” below.
Sales and Marketing
We have retained all worldwide marketing and distribution rights to OMIDRIA, our product candidates and our development programs. This allows us the opportunity to market and sell OMIDRIA and any product candidates that is approved in the future, either independently, through arrangements with third parties, or via some combination of these approaches.
With respect to OMIDRIA in the U.S., we have developed our own internal marketing and sales capabilities and, as of February 12, 2019, we employ 64 sales and reimbursement team members. In July 2018 we placed OMIDRIA on the market in the EU on a limited basis, which maintained the ongoing validity of the European marketing authorization for OMIDRIA. Our European marketing authorization will cease to be valid if a product previously placed on the market is no longer actually present on the market for three consecutive years. Outside of the U.S. and EU, we have the ITROM Agreement for certain countries in the Middle East. Other than the ITROM Agreement, we have not yet entered into any agreements with third parties to market OMIDRIA outside of the U.S.
Manufacturing, Supply and Commercial Operations
OMIDRIA. We use third parties to produce, store and distribute OMIDRIA and currently do not own or operate manufacturing facilities. Our agreements with these third parties include confidentiality and intellectual property provisions to protect our proprietary rights related to OMIDRIA. We require manufacturers that produce APIs and finished drug products to operate in accordance with current Good Manufacturing Practices, or cGMPs, and all other applicable laws and regulations.
We have an agreement with Hospira Worldwide, Inc., a wholly owned subsidiary of Pfizer, Inc., or Hospira, to provide commercial supply of OMIDRIA. The Hospira manufacturing site for OMIDRIA cleared the FDA in December 2017 and is expected to be cleared by the EMA during the first quarter of 2019. Under the agreement with Hospira, or the Hospira OMIDRIA Agreement, Hospira has agreed to manufacture and supply, and we have agreed to purchase, a minimum percentage of our requirements of OMIDRIA for commercial sales and clinical supplies for the development of additional therapeutic indications in the U.S. In addition, Hospira has agreed to manufacture and supply a portion of our requirements of OMIDRIA in the EU in an amount to be mutually agreed by the parties (not to exceed a maximum percentage of our EU requirements) by amendment, with there being no minimum purchase and supply requirement in the EU if the parties do not enter into such an
10
amendment to the agreement. The Hospira OMIDRIA Agreement expires in February 2022. The Hospira OMIDRIA Agreement may be terminated prior to the end of its term upon the occurrence of certain specified events, including without limitation an uncured breach of the agreement or bankruptcy or dissolution of a party. Upon termination of the Hospira OMIDRIA Agreement, except in the case of termination for an uncured breach by Hospira, we will be required to purchase all of Hospira’s inventory of OMIDRIA and, if applicable, all work-in-progress inventory and to reimburse Hospira for all supplies purchased or ordered based on firm purchase orders or our estimates of its requirements of OMIDRIA.
We have used multiple suppliers for the APIs for OMIDRIA in the past and we intend to leverage Hospira’s sourcing of APIs in the future under the Hospira OMIDRIA Agreement. Given the large amount of these APIs manufactured annually by these and other suppliers, and the quantities of these APIs we have on hand, we anticipate that we will be capable of addressing our commercial API supply needs for OMIDRIA in the near-term. We have not yet signed commercial agreements with suppliers for the supply of all of our anticipated commercial quantities of these APIs for OMIDRIA, although we may elect to do so in the future.
In the U.S., we sell OMIDRIA through a limited number of wholesalers that distribute the product to ASCs and hospitals. Title transfers upon delivery of OMIDRIA to the wholesaler. We use a single third-party logistics provider to handle warehousing and final packaging of our commercial supply of OMIDRIA in the U.S. and to ship OMIDRIA to our wholesalers. Our third-party logistics provider also performs certain support services on our behalf. Virtually all of our revenues for the last three fiscal years were generated from OMIDRIA product sales in the U.S. Our four major distributors--AmerisourceBergen Corporation, Cardinal Health, Inc., McKesson Corporation and FFF Enterprises, Inc.--together with entities under their common control each accounted for 10% or more, and nearly 100% in aggregate, of our total revenue in 2018.
Product Candidates. We have laboratories in-house for analytical method development, bioanalytical testing, formulation, stability testing and small-scale compounding of laboratory supplies of product candidates. We utilize contract manufacturers to produce sufficient quantities of product candidates for use in preclinical and clinical studies and to store and distribute our product candidates, and we currently do not own or operate manufacturing facilities for our product candidates. We require manufacturers that produce APIs and finished drug products for clinical use to operate in accordance with cGMPs and all other applicable laws and regulations. We anticipate that we will rely on contract manufacturers to develop and manufacture our product candidates for commercial sale. We maintain agreements with potential and existing manufacturers that include confidentiality and intellectual property provisions to protect our proprietary rights related to our product candidates. We have not yet entered into a commercial supply agreement for any of our product candidates, although we intend to do so prior to the applicable product candidate’s commercial launch. Given the nature of the manufacturing processes of our product candidates, we anticipate that we will be capable of identifying contract manufacturers to produce these product candidates and of entering into agreements for the commercial supply of these drugs.
License and Development Agreements
MASP Program. Under our exclusive license agreements with the University of Leicester and MRC, we have agreed to pay royalties to each of the University of Leicester and MRC that are a percentage of any proceeds we receive from the licensed MASP-2 technology during the terms of the agreements. Our exclusive license agreement with the University of Leicester, but not our agreement with the MRC, also applies to other MASPs. The continued maintenance of these agreements requires us to undertake development activities. We must pay low single-digit percentage royalties with respect to proceeds that we receive from products incorporating certain intellectual property within the licensed technology that are used, manufactured, directly sold or directly distributed by us, and we must pay royalties, in the range of a low single-digit percentage to a low double-digit percentage, with respect to proceeds we receive from sublicense royalties or fees that we receive from third parties to which we grant sublicenses to certain intellectual property within the licensed technology. We did not make any upfront payments for these exclusive licenses nor are there any milestone payments or reversion rights associated with these license agreements. We retain worldwide exclusive licenses from these institutions to develop and commercialize any intellectual property rights developed in the sponsored research. The term of each license agreement ends when there are no longer any pending patent applications, applications in preparation or unexpired issued patents related to any of the intellectual property rights we are licensing under the agreement. Both of these license agreements may be terminated prior to the end of their terms by us for convenience or by one party if the other party (1) breaches any material obligation under the agreement and does not cure such breach after notice and an opportunity to cure or (2) is declared or adjudged to be insolvent, bankrupt or in receivership and materially limited from performing its obligations under the agreement.
In April 2010, we entered into an exclusive license agreement with Helion Biotech ApS, or Helion, pursuant to which we received a royalty-bearing, worldwide exclusive license to all of Helion’s intellectual property rights related to MASP-2 antibodies, polypeptides and methods in the field of inhibition of mannan-binding lectin-mediated activation of the complement system for the prevention, treatment or diagnosis of any disease or condition. We are obligated to make remaining development and sales milestone payments to Helion of up to approximately $5.4 million upon the achievement of certain events, such as
11
receipt of marketing approval, and reaching specified sales milestones. We are obligated to pay Helion a low single-digit percentage royalty on net sales of a MASP-2 inhibitor product covered by the patents licensed under the agreement. The term of the agreement continues so long as there is a valid, subsisting and enforceable claim in any patents or patent applications covered by the agreement. The agreement may be terminated sooner by either party following a material breach of the agreement by the other party that has not been cured within 90 days.
OMS824. We have agreed to pay royalties to SMRI based on any net income we receive from sales of a PDE10 product until we have paid a maximum aggregate amount that is a low single-digit multiple of the amount of grant funding that we have received from SMRI. This multiple increases as time elapses from the date we received the grant funding. There are no minimum payment obligations under our agreement with SMRI. Based on the amount of grant funding that we received from SMRI, the maximum amount of royalties payable to SMRI is $12.8 million and payment is required only from any net income, after all related expenses, that we receive from sales of a PDE10 product. The funding agreement and our obligation to pay a royalty to SMRI terminate when we have repaid such amount in the form of royalties.
PPARγ. We acquired the patent applications and related intellectual property rights for our PPARγ program in February 2009 from Roberto Ciccocioppo, Ph.D. of the Università di Camerino, Italy, pursuant to a patent assignment agreement. In February 2011, we amended the agreement to include all intellectual property rights, including patent applications, related to nutraceuticals that increase PPARγ activity. Under the amended agreement, we have agreed to pay Dr. Ciccocioppo a low-single digit percentage royalty on net sales of any products that are covered by any patents that issue from the patent applications that we acquired from him. In addition, if we grant any third parties rights to manufacture, sell or distribute any such products, we must pay to Dr. Ciccocioppo a percentage of any associated fees we receive from such third parties in the range of low single-digits to low double-digits depending on the stage of development at which such rights are granted. We have also agreed to make total milestone payments of up to $3.8 million to Dr. Ciccocioppo upon the occurrence of certain development events, such as patient enrollment in a Phase 1 clinical trial and receipt of marketing approval of a product candidate covered by any patents that issue from the patent applications that we acquired from him. If we notify Dr. Ciccocioppo that we have abandoned all research and development and commercialization efforts related to the patent applications and intellectual property rights we acquired from him, Dr. Ciccocioppo has the right to repurchase those assets from us at a price equal to a double-digit percentage of our direct and indirect financial investments and expenditures in such assets. If he does not exercise his right to repurchase those assets within a limited period of time by paying the purchase price, we will have no further obligations to sell those assets to Dr. Ciccocioppo. The term of our agreement with Dr. Ciccocioppo ends when there are no longer any valid and enforceable patents related to the intellectual property rights we acquired from him, provided that either party may terminate the agreement earlier in case of an uncured breach by the other party. Under the terms of the agreement, we have agreed to pay a portion of the payments due to Dr. Ciccocioppo to the Università di Camerino without any increase to our payment obligations.
PDE7. Under an agreement with Daiichi Sankyo, we hold an exclusive worldwide license to PDE7 inhibitors claimed in certain patents and pending patent applications owned by Daiichi Sankyo for use in the treatment of (1) movement disorders and other specified indications, (2) addiction and compulsive disorders and (3) all other diseases except those related to dermatologic conditions. Under the agreement, we agreed to make milestone payments to Daiichi Sankyo of up to an aggregate total of $33.5 million upon the achievement of certain events in each of these three fields; however, if only one of the three indications is advanced through the milestones, the total milestone payments would be $23.5 million. The milestone payment events include successful completion of preclinical toxicology studies; dosing of human subjects in Phase 1, 2 and 3 clinical trials; receipt of marketing approval of a PDE7 inhibitor product candidate; and reaching specified sales milestones. In addition, Daiichi Sankyo is entitled to receive from us a low single-digit percentage royalty of any net sales of a PDE7 inhibitor licensed under the agreement by us and/or our sublicensee(s) provided that, if the sales are made by a sublicensee, then the amount payable by us to Daiichi Sankyo is capped at an amount equal to a low double-digit percentage of all royalty and specified milestone payments received by us from the sublicensee.
The term of the agreement with Daiichi Sankyo continues so long as there is a valid, subsisting and enforceable claim in any patents covered by the agreement. The agreement may be terminated sooner by us, with or without cause, upon 90 days advance written notice or by either party following a material breach of the agreement by the other party that has not been cured within 90 days or immediately if the other party is insolvent or bankrupt. Daiichi Sankyo also has the right to terminate the agreement if we and our sublicensee(s) cease to conduct all research, development and/or commercialization activities for a PDE7 inhibitor covered by the agreement for a period of six consecutive months, in which case all rights held by us under Daiichi Sankyo’s patents will revert to Daiichi Sankyo.
GPCR Platform Funding Agreements with Vulcan Inc. and the Life Sciences Discovery Fund. In October 2010, we entered into funding agreements for our GPCR program with Vulcan and LSDF. We received $20.0 million and $5.0 million, respectively, under the agreements with Vulcan and LSDF. Under these agreements, we have agreed to pay Vulcan and LSDF tiered percentages of the net proceeds, if any, that we derive from the GPCR program. The percentage rates of net proceeds
12
payable to Vulcan and LSDF decrease as the cumulative net proceeds reach specified thresholds, and the blended percentage rate payable to Vulcan and LSDF in the aggregate is in the mid-teens with respect to the first approximately $1.5 billion of cumulative net proceeds that we receive from our GPCR program. If we receive cumulative net proceeds in excess of approximately $1.5 billion, the percentage rate payable to Vulcan and LSDF in the aggregate decreases to one percent. An acquirer of the assets in our GPCR program may be required, and an acquirer of our company would be required, to assume all of our payment and other obligations under our agreements with Vulcan and LSDF.
Under our agreement with Vulcan, we granted Vulcan a security interest in our personal property related to the GPCR program, other than intellectual property, which security interest is junior to any existing or future security interests granted in connection with a financing transaction and which will be released automatically after Vulcan receives $25.0 million under the agreement. We also agreed not to grant any liens on intellectual property related to the GPCR program without Vulcan’s consent, subject to specified exceptions. These restrictions could limit our ability to pursue business opportunities involving the GPCR program or reduce the price that a potential buyer would pay for the GPCR assets. If we default under our agreement with Vulcan, in certain circumstances Vulcan may, subject to the rights of any holders of senior security interests, take control of such pledged assets. If we are liquidated, Vulcan’s right to receive any payments then due under our agreement would be senior to the rights of the holders of our common stock to receive any proceeds from the liquidation of our GPCR program assets.
The term of our agreement with Vulcan is 35 years, provided that the term will automatically extend until the cumulative net proceeds that we receive from the GPCR program are approximately $1.5 billion. The term of our agreement with LSDF expires on the six-month anniversary following the last date that we deliver a report related to our incurrence of grant-funded expenses described in the agreement, provided that certain obligations will survive the expiration of the term. The term of our payment obligations to LSDF is the same as that under our agreement with Vulcan.
Antibody Platform. We hold a worldwide exclusive license to patent rights related to our antibody platform from the University of Washington, or UW. Pursuant to the Xori APA, we acquired all of Xori’s exclusive rights under a license agreement with the UW to certain patents and patent applications related to our antibody platform owned by the UW in exchange for our agreement to make royalty and development milestone payments to UW.
OMIDRIA. We entered into settlement agreements and consent judgments with (i) Par Pharmaceutical, Inc. and its subsidiary, Par Sterile Products, LLC, or collectively Par, and (ii) Lupin Ltd. and Lupin Pharmaceuticals, Inc., or collectively Lupin, in October 2017 and May 2018, respectively. The settlement agreements resolved patent infringement lawsuits we filed against Par and Lupin in response to their respective filings with the FDA of Abbreviated New Drug Applications, or ANDAs, seeking approval to market a generic version of OMIDRIA prior to the expiration of our Orange Book-listed patents for OMIDRIA. Pursuant to the settlement agreements and consent judgments, Par and Lupin each acknowledged and confirmed the validity of the patents for OMIDRIA listed in the Orange Book at the time of the applicable settlement.
Under the terms of the settlement agreements and consent judgments, Par and Lupin are each prohibited from launching a generic version of OMIDRIA prior to a specified entry date. Par’s entry date is the earlier of (i) April 1, 2032 or (ii) the date on which we or a third-party, through licensing or any future final legal judgment, should one ever exist, with respect to our Orange Book listed patents, is able to launch a generic version of OMIDRIA. Lupin’s entry date is the earlier of (A) April 1, 2032 if Par has forfeited its six month first-ANDA filer exclusivity, (B) October 1, 2032 if Par has not forfeited its six month first-ANDA filer exclusivity, or (C) a date on which we or a third party (other than Par), through licensing of, any future final legal judgment regarding, or the delisting, abandonment or expiration of our U.S. OMIDRIA patents, is able to launch a generic version of OMIDRIA. Under the settlement agreements, we granted each of Par and Lupin a non-exclusive, non-sublicensable license to make, sell and distribute a generic version of OMIDRIA between their applicable entry dates and the latest expiration of our U.S. patents related to OMIDRIA (i.e., October 23, 2033). During this period, Par and Lupin, as applicable, are each required to pay us a royalty equal to 15% of net sales of its generic version of OMIDRIA.
Competition
Overview. The pharmaceutical industry is highly competitive and characterized by a number of established, large pharmaceutical companies as well as smaller companies like ours. We expect to compete with other pharmaceutical and biotechnology companies, and our competitors may:
| • | develop and market products that are less expensive, more effective or safer than our future products; |
| • | commercialize competing products before we can launch our products; |
| • | operate larger research and development programs, possess greater manufacturing capabilities or have substantially greater financial resources than we do; |
| • | initiate or withstand substantial price competition more successfully than we can; |
13
| • | have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; |
| • | more effectively negotiate third-party licenses and strategic relationships; and |
| • | take advantage of acquisition or other opportunities more readily than we can. |
We expect to compete for market share against large pharmaceutical and biotechnology companies, smaller companies that are collaborating with larger pharmaceutical companies, new companies, academic institutions, government agencies and other public and private research organizations. In addition, the pharmaceutical and biotechnology industry is characterized by rapid technological change. Because our research approach integrates many technologies, it may be difficult for us to remain current with the rapid changes in each technology. Further, our competitors may render our technologies obsolete by advancing their existing technological approaches or developing new or different approaches. If we fail to stay at the forefront of technological change, we may be unable to compete effectively.
OMIDRIA. We are not aware of any product that directly competes with OMIDRIA and is FDA-approved for intraoperative delivery in irrigation solutions during surgical procedures; however, OMIDRIA could face competition from products that are delivered intraoperatively, but that do not include a non-steroidal anti-inflammatory agent, as well as from preoperative and postoperative treatments for mydriasis, pain or inflammation. Our primary competition for OMIDRIA comes from surgeons’ current practices, which may include use of products obtained from distributors or compounding pharmacies at a relatively low cost. Title I (the Compounding Quality Act) of the Drug Quality and Security Act, which was enacted in November 2013, added Section 503B to the FDCA establishing a distinct category of drug compounders known as “outsourcing facilities.” Among other provisions, the Compounding Quality Act imposes restrictions on the materials that may be compounded at registered outsourcing facilities and traditional compounders and places conditions on the compounding of bulk substances. Surgeons may perceive that, since the enactment of the Compounding Quality Act, compounding pharmacies, particularly those that are registered as “outsourcing facilities,” are subject to rigorous regulatory oversight that assures the safety and manufacturing quality of compounded products, notwithstanding the relatively high frequency of recall events, warning letters and findings of insanitary conditions issued by FDA following inspection of registered outsourcing facilities. In addition, we anticipate that there are some surgeons who do not use intraoperative mydriatics and may not agree with the value proposition of maintaining pupil dilation and inhibiting miosis during the procedure, or with the use of a nonsteroidal anti-inflammatory drug intraoperatively to inhibit inflammation, prevent miosis and reduce postoperative pain. Although we are not aware of any companies developing similar approaches for maintenance of intraoperative pupil size and postoperative pain reduction as an FDA-approved product, such strategies may develop. In Europe, an inexpensive mydriatic and local anesthetic combination product is available but, unlike OMIDRIA, this product does not include an anti-inflammatory agent.
Product Candidates, Development Programs and Platforms. With respect to our complement system program, there are multiple companies developing potential therapies targeting the complement system, although none of these potential therapies, to our knowledge, inhibit the lectin pathway. Soliris® (eculizumab) and UltomirisTM (ravulizumab-cwvz) are monoclonal complement inhibitors administered intravenously and approved for commercial use with which our lead MASP-2 inhibitor, narsoplimab (OMS721), and/or our MASP-3 inhibitor OMS906 will compete, if either is approved for any indication(s) for which Soliris® and/or UltomirisTM are also approved. Alexion Pharmaceuticals, Inc., the manufacturer of Soliris® and UltomirisTM, has also announced development of follow-on antibodies that are directed to the same target, but which can be administered subcutaneously.
With respect to our PDE10 inhibitor program, we are developing potential product candidates for use in the treatment of Huntington’s disease, schizophrenia and other diseases that affect cognition. Other pharmaceutical companies, many with significantly greater resources than we have, are also developing, or may develop, PDE10 inhibitors for the treatment of these indications, and these companies may be further along in development. In 2017, Pfizer announced negative results in a clinical trial of a PDE10 inhibitor for the treatment of Huntington’s disease. Also, Pfizer previously announced negative results from schizophrenia trials with a PDE10 inhibitor and Takeda Pharmaceuticals previously announced that the primary endpoint was not met in a clinical trial of a different PDE10 inhibitor in a schizophrenia trial. In 2018, Roche announced the termination of its small-molecule inhibitor of PDE10 program for the treatment of schizophrenia.
We are aware of other companies attempting to de-orphanize orphan GPCRs. If any of these companies is able to de-orphanize an orphan GPCR before we unlock this receptor, we may be unable to establish an exclusive or commercially valuable intellectual property position around that orphan GPCR.
Intellectual Property
We have retained control of all worldwide manufacturing, marketing and distribution rights for OMIDRIA and each of our product candidates and programs. Some of our products and product candidates and programs are based on inventions and
14
other intellectual property rights that we acquired through assignments, exclusive licenses or acquisitions described in further detail under “License and Development Agreements” below.
As of February 12, 2019, we owned or held worldwide exclusive licenses to a total of 77 issued patents and 61 pending patent applications in the U.S. and 809 issued patents and 485 pending patent applications in foreign markets directed to therapeutic compositions and methods related to our research and development programs. For each program, our decision to seek patent protection in specific foreign markets, in addition to the U.S., is based on many factors, including one or more of the following: our available resources, the size of the commercial market, the presence of a potential competitor or a contract manufacturer in the market and whether the legal authorities in the market effectively enforce patent rights.
| • | OMIDRIA-Ophthalmology. OMIDRIA is encompassed by our PharmacoSurgery patent portfolio. The relevant patents and patent applications in this portfolio are directed to combinations of agents, generic and/or proprietary to us or to others, drawn from therapeutic classes such as pain and inflammation inhibitory agents, mydriatic agents and agents that reduce intraocular pressure, delivered locally and intraoperatively to the site of ophthalmological procedures, including cataract and lens replacement surgery. As of February 12, 2019, we owned eight issued U.S. patents and three pending U.S. patent applications and 60 issued patents and 54 pending patent applications in foreign markets that are directed to OMIDRIA. Our OMIDRIA patents have terms that will expire as late as October 23, 2033 and, if currently pending patent applications are issued, as late as November 30, 2035. |
| • | MASP-2 Program - Narsoplimab (OMS721). We hold worldwide exclusive licenses to rights in connection with MASP-2, the antibodies targeting MASP-2 and the therapeutic applications for those antibodies from the University of Leicester, MRC and Helion. As of February 12, 2019, we exclusively controlled 20 issued patents and 33 pending patent applications in the U.S., and 314 issued patents and 246 pending patent applications in foreign markets, related to our MASP-2 program. Our MASP-2 and narsoplimab patents have terms that will expire as late as 2032 and, if currently pending patent applications are issued, as late as 2039. |
| • | MASP-3 Program - OMS906. We own and exclusively control under a license from the University of Leicester all rights to methods of treating various disorders and diseases by inhibiting MASP-3. As of February 12, 2019, we exclusively controlled three pending patent applications in the U.S. and 48 issued and 85 pending patent applications in foreign markets that are related to our MASP-3 program. |
| • | PDE10 Program - OMS824. As of February 12, 2019, we owned 14 issued patents and two pending patent applications in the U.S., and 44 issued patents and 40 pending patent applications in foreign markets, that are directed to proprietary PDE10 inhibitors. |
| • | PPARγ Program - OMS405. As of February 12, 2019, we owned two issued patents and one pending patent application in the U.S., and 29 issued patents and 10 pending patent applications in foreign markets, directed to our discoveries linking PPARγ and addictive disorders. |
| • | PDE7 Program - OMS527. As of February 12, 2019, we owned two issued patents and one pending patent application in the U.S., and 59 issued patents and six pending patent applications in foreign markets directed to our discoveries linking PDE7 to movement disorders, as well as one issued patent and two pending patent applications in the U.S., and 44 issued patents and 18 pending patent applications in foreign markets directed to the link between PDE7 and addiction and compulsive disorders. Additionally, under a license from Daiichi Sankyo, we exclusively control rights to three issued U.S. patents and 54 issued and four pending patent applications in foreign markets that are directed to proprietary PDE7 inhibitors. For a more detailed description of our agreement with Daiichi Sankyo, see “License and Development Agreements” below. |
| • | OMS103-Arthroscopy. As of February 12, 2019, we owned three issued U.S. patents and one pending U.S. patent application, together with 36 issued patents and eight pending patent applications in foreign markets that are directed to OMS103. Our OMS103 patents have terms that will expire as late as September 24, 2022 and, if currently pending patent applications are issued, as late as 2032. We are not actively pursuing development of OMS103 at this time. |
| • | GPCR Platform. As of February 12, 2019, we owned six issued patents and 13 pending patent applications in the U.S., and 55 issued patents and one pending patent application in foreign markets, which are directed to previously unknown links between specific molecular targets in the brain and a series of CNS disorders, to our cellular redistribution assay and to other research tools that are used in our GPCR program, and to orphan GPCRs and other GPCRs for which we have identified functionally interacting compounds using our cellular redistribution assay. |
| • | Antibody Platform. As of February 12, 2019, we owned and/or held worldwide exclusive license rights from the UW to eight issued patents and one pending patent application in the U.S., and 21 issued patents and eight pending patent applications in foreign markets, directed to our antibody platform and antibodies generated using our platform. |
15
All of our employees enter into our standard employee proprietary information and inventions agreement, which includes confidentiality provisions and provides us ownership of all inventions and other intellectual property made by our employees that pertain to our business or that relate to our employees’ work for us or that result from the use of our resources. Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection of the use, formulation and structure of our products and product candidates and the methods used to manufacture them, as well as on our ability to defend successfully these patents against third-party challenges. Our ability to protect our products and product candidates from unauthorized making, using, selling, offering to sell or importing by third parties is dependent on the extent to which we have rights under valid and enforceable patents that cover these activities.
The patent positions of pharmaceutical, biotechnology and other life sciences companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in biotechnology patents has emerged to date in the U.S., and tests used for determining the patentability of patent claims in all technologies are in flux. The pharmaceutical, biotechnology and other life sciences patent situation outside the U.S. is even more uncertain. Changes in either the patent laws or in interpretations of patent laws in the U.S. and other countries may diminish the value of our intellectual property. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in the patents that we own or have licensed or in third-party patents.
We sell OMIDRIA under trademarks that we consider in the aggregate to be important to our operations. We have registered, and intend to maintain, the trademarks “OMEROS”, “OMIDRIA”, “OMIDRIASSURE” and “PHARMACOSURGERY” with the U.S. Patent and Trademark Office, or USPTO, in connection with the products and services we offer. We are not aware of any material claims of infringement or other challenges to our right to use the “OMEROS”, “OMIDRIA”, “OMIDRIASSURE” or “PHARMACOSURGERY” trademarks in the U.S.
Government Regulation
Government authorities in the U.S., the EU and other countries extensively regulate the research, development, testing, manufacture, labeling, promotion, advertising, distribution, marketing, and export and import of drug and biologic products such as OMIDRIA and the product candidates that we are developing. Failure to comply with applicable requirements, both before and after receipt of regulatory approval, may subject us, our third-party manufacturers, and other partners to administrative and judicial sanctions, such as warning letters, product recalls, product seizures, a delay in approving or refusal to approve pending applications, civil and other monetary penalties, total or partial suspension of production or distribution, injunctions, and/or criminal prosecutions.
In the U.S., our products and product candidates are regulated by the FDA as drugs or biologics under the FDCA and implementing regulations and, in the case of biologics, also under the Public Health Service Act, or PHSA. In Europe, our products and product candidates are regulated by the EMA and national medicines regulators under the rules governing medicinal products in the EU as well as national regulations in individual countries. OMIDRIA has received marketing approval from the FDA and from the applicable regulatory authorities in the EU. Our product candidates are in various stages of testing and none of our product candidates has received marketing approval from the FDA or the applicable regulatory authorities in the EU.
The steps required before a product may be approved for marketing by the FDA, or the applicable regulatory authorities outside of the U.S., typically include the following:
| • | formulation development and manufacturing process development; |
| • | preclinical laboratory and animal testing; |
| • | submission to the FDA of an Investigational New Drug application, or IND, for human clinical testing, which must become effective before human clinical trials may begin; and in countries outside the U.S., a Clinical Trial Application, or CTA, is filed according to the country’s local regulations; |
| • | adequate and well-controlled human clinical trials to establish the efficacy and safety of the product for each indication for which approval is sought; |
| • | adequate assessment of drug product stability to determine shelf life/expiry dating; |
| • | in Europe, submission to the EMA or national regulatory authority of a marketing authorization application, or MAA, and in the U.S., submission to the FDA of a New Drug Application, or NDA, in the case of a drug product, or a BLA in the case of a biologic product; |
| • | satisfactory completion of inspections of one or more clinical sites at which clinical trials with the product were carried out and of the manufacturing facility or facilities at which the product is produced to assess compliance with Good Clinical Practices, or GCP, and current good manufacturing practices, or cGMP; and |
16
| • | FDA review and approval of an NDA or BLA, or review and approval of an MAA by the applicable regulatory authorities in the EU. |
Manufacturing. Manufacturing of drug products for use in clinical trials must be conducted according to relevant national and international guidelines, for example, cGMP. Process and formulation development are undertaken to design suitable routes to manufacture the drug substance and the drug product for administration to animals or humans. Analytical development is undertaken to obtain methods to quantify the potency, purity and stability of the drug substance and drug product as well as to measure the amount of the drug substance and its metabolites in biological fluids, such as blood.
Preclinical Tests. Preclinical tests include laboratory evaluations and animal studies to assess efficacy, toxicity and pharmacokinetics. The results of the preclinical tests, together with manufacturing information, analytical data, clinical development plan, and other available information are submitted as part of an IND or CTA.
The IND/CTA Process. An IND or CTA must become effective before human clinical trials may begin. An IND will automatically become effective 30 days after receipt by the FDA unless, before that time, the FDA raises concerns or questions and imposes a clinical hold. In that event, the IND sponsor and the FDA must resolve any outstanding FDA concerns or questions before the clinical hold is lifted and clinical trials can proceed. Similarly, a CTA must be cleared by the local independent ethics committee and competent authority prior to conducting a clinical trial in the country in which it was submitted. This process can take from two weeks to several months. There can be no assurance that submission of an IND or CTA will result in authorization to commence clinical trials. Once an IND or CTA is in effect, there are certain reporting requirements.
Clinical Trials. Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified personnel and must be conducted in accordance with local regulations and GCP. Clinical trials are conducted under protocols detailing, for example, the parameters to be used in monitoring patient safety and the efficacy criteria, or endpoints, to be evaluated. Each trial must be reviewed and approved by an independent institutional review board or ethics committee for each clinical site at which the trial will be conducted before it can begin. Clinical trials are typically conducted in three defined phases, but the phases may overlap or be combined:
| • | Phase 1 usually involves the initial administration of the investigational product to human subjects, who may or may not have the disease or condition for which the product is being developed, to evaluate the safety, dosage tolerance, pharmacodynamics and, if possible, to gain an early indication of the effectiveness of the product. |
| • | Phase 2 usually involves trials in a limited patient population with the disease or condition for which the product is being developed to evaluate appropriate dosage, to identify possible adverse side effects and safety risks, and to evaluate preliminarily the effectiveness of the product for specific indications. |
| • | Phase 3 clinical trials usually further evaluate and confirm effectiveness and test further for safety by administering the product in its final form in an expanded patient population. |
We, our product development partners, institutional review boards or ethics committees, the FDA or other regulatory authorities may suspend clinical trials at any time on various grounds, including a belief that the subjects are being exposed to an unacceptable health risk.
Disclosure of Clinical Trial Information. Sponsors of clinical trials of certain FDA-regulated products, including prescription drugs, are required to register and disclose certain clinical trial information on a public website maintained by the U.S. National Institutes of Health. Information related to the product, patient population, phase of investigation, study sites and investigator, and other aspects of the clinical trial is made public as part of the registration. Sponsors are also obligated to disclose the results of these trials after completion. Disclosure of the results of these trials can be delayed for up to two years if the sponsor certifies that it is seeking approval of an unapproved product or that it will file an application for approval of a new indication for an approved product within one year. Clinical trials conducted in European countries are required to be registered at a similar public database maintained and overseen by European health authorities. Competitors may use this publicly available information to gain knowledge regarding the design and progress of our development programs.
The Application Process. If the necessary clinical trials are successfully completed, the results of the preclinical trials and the clinical trials, together with other detailed information, including information on the manufacture and composition of the product, are submitted to the FDA in the form of an NDA or a BLA, as applicable, and to the EMA or national regulators in the form of an MAA, requesting approval to market the product for a specified indication. In the EU, an MAA may be submitted to the EMA for review and, if the EMA gives a positive opinion, the EC may grant a marketing authorization that is valid across the EU (centralized procedure). Alternatively, an MAA may be submitted to one or more national regulators in the EU according to one of several national or decentralized procedures. The type of submission in Europe depends on various factors
17
and must be cleared by the appropriate authority prior to submission. For most of our product candidates, the centralized procedure will be either mandatory or available as an option.
If the regulatory authority determines that the application is not acceptable, it may refuse to accept the application for filing and review, outlining the deficiencies in the application and specifying additional information needed to file the application. Notwithstanding the submission of any requested additional testing or information, the regulatory authority ultimately may decide that the application does not satisfy the criteria for approval. Before approving an NDA or BLA, or an MAA, the FDA or the EMA, respectively, may inspect one or more of the clinical sites at which the clinical study(ies) were conducted to ensure that GCPs were followed and may inspect facility(ies) at which the product is manufactured to ensure satisfactory compliance with cGMP. After approval, changes to the approved product such as adding new indications, manufacturing changes, or additional labeling claims will require submission of a supplemental application, referred to as a variation in the EU, or, in some instances, a new application, for further review and approval. The testing and approval process requires substantial time, effort, and financial resources, and we cannot be sure that any future approval will be granted on a timely basis, if at all.
Some of our drug products may be eligible for NDA submissions to the FDA for approval under the Section 505(b)(2) process. Section 505(b)(2) applications may be submitted for drug products that represent a modification, such as a new indication or new dosage form, of a previously approved drug. Section 505(b)(2) applications may rely on the FDA’s previous findings for the safety and effectiveness of the previously approved drug in addition to information obtained by the 505(b)(2) applicant to support the modification of the previously approved drug. Preparing Section 505(b)(2) applications may be less costly and time-consuming than preparing an NDA that is based entirely on new data and information.
Some of our product candidates, such as those from our MASP-2 and MASP-3 programs, are considered biologics because they are derived from natural sources as opposed to being chemically synthesized. The added complexity associated with manufacturing biologics may result in additional monitoring of the manufacturing process and product changes.
In addition, we, our suppliers and our contract manufacturers are required to comply with extensive regulatory requirements both before and after approval. For example, we must establish a pharmacovigilance system and are required to report adverse reactions and production problems, if any, to the regulatory authorities. We must also comply with certain requirements concerning advertising and promotion for our products. The regulatory authorities may impose specific obligations as a condition of the marketing authorization, such as additional safety monitoring, or the conduct of additional clinical trials or post-marketing safety studies. Also, quality control and manufacturing procedures must continue to conform to cGMPs after approval. Accordingly, manufacturers must continue to expend time, money, and effort in all areas of regulatory compliance, including production and quality control to comply with cGMPs. In addition, discovery of problems such as safety issues may result in changes in labeling or restrictions on a product manufacturer or marketing authorization holder, including removal of the product from the market.
Fast-Track and Priority Review Designations. Section 506(b) of the FDCA provides for the designation of a drug as a fast-track product if it is intended, whether alone or in combination with one or more other drugs, for the treatment of a serious or life-threatening disease or condition, and it demonstrates the potential to address unmet medical needs for such a disease or condition. A program with fast-track status is afforded greater access to the FDA for the purpose of expediting the product’s development, review and potential approval. Many products that receive fast-track designation are also considered appropriate to receive priority review, and their respective applications may be accepted by the FDA as a rolling submission in which portions of an NDA or BLA are reviewed before the complete application is submitted. Together, these may reduce time of development and FDA review time. In Europe, products that are considered to be of major public health interest are eligible for accelerated assessment, which shortens the review period. The grant of fast-track status, priority review or accelerated assessment does not alter the standard regulatory requirements for obtaining marketing approval.
Breakthrough Therapy Designation. In 2012, Congress enacted the Food and Drug Administration Safety and Innovation Act. This law established a regulatory process allowing for increased interactions with the FDA with the goal of expediting development and review of products designated as “breakthrough therapies.” A product may be designated as a breakthrough therapy if it is intended, either alone or in combination with one or more other products, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The FDA may take certain actions with respect to breakthrough therapies, including holding meetings with the sponsor throughout the development process; providing timely advice to the product sponsor regarding development and approval; involving more senior staff in the review process; assigning a cross-disciplinary project lead for the review team; and taking other steps to design the clinical trials in an efficient manner.
18
Accelerated Approval. The FDA may grant accelerated approval to a product for a serious or life-threatening condition that provides meaningful therapeutic advantage to patients over existing treatments based upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The FDA may also grant accelerated approval for such a condition when the product has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality and that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit. In both cases, FDA must take into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. Studies that are conducted to demonstrate a drug’s effect on a surrogate or intermediate clinical endpoint for accelerated approval must be adequate and well-controlled as required by the FDCA.
Following accelerated approval, the FDA requires that the company provide confirmatory evidence, which may include certain adequate and well-controlled post-marketing clinical studies to verify and describe the clinical benefit of the product, and the FDA may impose restrictions on distribution to assure safe use. Confirmatory studies are typically required to be underway at the time of the accelerated approval. If the required confirmatory studies fail to verify the clinical benefit of the drug, or if the applicant fails to perform the required confirmatory studies with due diligence, the FDA may withdraw approval of the drug under streamlined procedures in accordance with the Agency’s regulations. The Agency may also withdraw approval of a drug if, among other things, other evidence demonstrates that the drug product is not shown to be safe or effective under its conditions of use.
The EU also has accelerated approval programs. In the EU, a marketing authorization may be granted on the basis of less complete data than are normally required in certain “exceptional circumstances,” such as when the product’s indication is encountered so rarely that the applicant cannot reasonably be expected to provide comprehensive data. Alternatively, a conditional marketing authorization may be granted prior to obtaining the comprehensive clinical data required for a full MAA if a product fulfills an unmet medical need and the benefit to public health of the product’s immediate availability outweighs the risk inherent in the incomplete data.
Orphan Drug Designation. Under the Orphan Drug Act, or ODA, the FDA may grant orphan drug designation to drugs or biologics intended to treat a rare disease or condition that affects fewer than 200,000 individuals in the U.S. or more than 200,000 individuals in the U.S. for which the cost of developing and making the product available in the U.S. for this type of disease or condition is not likely to be recovered from U.S. sales for that product. The granting of orphan designation does not alter the standard regulatory requirements (other than payment of certain fees), nor does it alter the standards or process for obtaining marketing approval. If a product that has an orphan drug designation subsequently receives the first FDA approval for the indication for which it has been designated as an orphan drug, the sponsor of the product qualifies for various development incentives specified in the ODA, including tax credits for qualified clinical testing (although Congress recently reduced the tax credit from 50% to 25% in 2017 tax legislation). Furthermore, the product is entitled to an orphan drug exclusivity period, which means that the FDA may not grant approval to any other application to market the same drug for the same indication for a period of seven years except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity for the protected indication. If the FDA designates an orphan drug based on a finding of clinical superiority, the FDA must provide a written notification to the sponsor that states the basis for orphan designation, including “any plausible hypothesis” relied on by the FDA. The FDA must also publish a summary of its clinical superiority findings upon granting orphan drug exclusivity based on clinical superiority. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition. The EU has a similar Orphan Drug program to that of the U.S., and it is administered through the EMA’s Committee for Orphan Medicinal Products, or COMP.
Pediatric Testing and Exclusivity. In the U.S., NDAs and BLAs are subject to both mandatory pediatric testing requirements and voluntary pediatric testing incentives in the form of exclusivity. An additional six months of exclusivity in the U.S. may be granted to a sponsor of an NDA or BLA if the sponsor conducts certain pediatric studies. This process is initiated when the FDA issues a Written Request for pediatric studies to determine if the drug or biologic could have meaningful pediatric health benefits. If the FDA determines that the sponsor has conducted the requested pediatric studies in accordance with the written request, then an additional six months of exclusivity may attach in the case of a drug to any other regulatory exclusivity or patent protection applicable to the drug and, in the case of a biologic, to any other regulatory exclusivity applicable to the biologic. The EU has a similar requirement and incentive for the conduct of pediatric studies according to the pediatric investigation plan, which must be adopted by the EMA before an MAA may be submitted.
Expanded Access and Right to Try. “Expanded access” refers to the use of an investigational drug where the primary purpose is to diagnose, monitor, or treat a patient’s disease or condition rather than to collect information about the safety or effectiveness of a drug. There are three FDA-recognized categories of expanded access trials: expanded access for individual patients, including for emergency use; expanded access for intermediate-size patient populations; and expanded access for large patient populations under a treatment IND or treatment protocol. For all types of expanded access, the FDA must determine
19
prior to authorizing expanded access that: (1) the patient or patients to be treated have a serious or life-threatening disease or condition and there is no comparable or satisfactory alternative therapy; (2) the potential patient benefit justifies the potential risks of use and that the potential risks are not unreasonable in the context of the disease or condition to be treated; and (3) granting the expanded access will not interfere with the initiation, conduct, or completion of clinical studies in support of the drug’s approval. Only a licensed physician or the drug’s manufacturer may apply for expanded access. Manufacturers are not required to supply the investigational product. The FDA has established streamlined processes for physicians to request individual patient expanded access whereby physicians can submit an abbreviated application. In cases of individual patient emergency expanded access, physicians can receive FDA approval for access by phone and follow up with the abbreviated form. In addition, the sponsor of an expanded access IND must submit IND safety reports and, in the cases of protocols continuing for one year or longer, annual reports to the FDA. Expanded access programs are not intended to yield information relevant to evaluating a drug’s effectiveness for regulatory purposes. INDs for expanded access trials may be sponsored by physicians or by manufacturers.
A manufacturer or distributor of an investigational drug for the diagnosis, monitoring or treatment of a serious disease or condition must make available its policy for evaluating and responding to requests for individual patient access to the investigational drug. A manufacturer or distributor must make its expanded access policy publicly available on: (1) the date of initiation of a Phase 2 or 3 study with respect to the investigational drug, or (2) if such date is applicable and earlier, 15 days after the drug receives a designation as a breakthrough therapy, fast-track product or regenerative advanced therapy. The policy must be made public and readily available, such as by posting on the Internet, and may be generally applicable to all of the manufacturer’s or distributor’s investigational drugs. Posting a policy does not guarantee access to an investigational drug by any individual patient, and the manufacturer or distributor may revise the policy at any time.
On May 30, 2018 the Trickett Wendler, Frank Mongiello, Jordan McLinn, and Matthew Bellina Right to Try Act of 2017, or the Right to Try Act, was signed into U.S. law. “Right to Try” is another way for patients to access an investigational drug outside the context of a clinical trial. Under Right To Try, only eligible patients may access eligible investigational drugs. An eligible patient is one who: (1) has been diagnosed with a life-threatening disease or condition, (2) has exhausted approved treatment options and is unable to participate in a clinical trial involving the eligible investigational drug (as certified by a physician), and (3) has provided (or their legally authorized representative has provided) written informed consent regarding the eligible investigational drug to the treating physician. An eligible investigational drug is an investigational drug: (1) for which a Phase 1 clinical trial has been completed, (2) that has not been approved or licensed by FDA for any use, (3)(a) for which a marketing application has been filed with the FDA, or (b) an IND has been submitted to FDA and the drug is under investigation in a clinical trial that is intended to form the primary basis of a claim of effectiveness in support of FDA approval, and (4) whose active development or production is ongoing and has not been discontinued by the manufacturer or placed on clinical hold by FDA. As under the expanded access programs, manufacturers are not required to supply the investigational drug under Right to Try. Additionally, FDA generally may not use a clinical outcome associated with use of an eligible investigational drug to delay or adversely affect the review or approval of such drug, unless the FDA makes a determination that use of such clinical outcome is critical to determining the safety of the eligible investigational drug, or the sponsor requests use of such outcomes. Unlike the expanded access programs, however, FDA and institutional review boards are not involved in the determination regarding whether a patient is eligible to receive the investigational drug under Right to Try.
U.S. Labeling, Marketing and Promotion. The FDA closely regulates the labeling, marketing and promotion of drugs. Failure to comply with these requirements can result in adverse publicity, warning letters, corrective advertising, injunctions and potential civil and criminal penalties.
In addition to regulation by the FDA, the research, manufacturing, distribution, sale and promotion of drug products in the U.S. are potentially subject to regulation by various federal, state and local authorities, including CMS, other divisions of the U.S. Department of Health and Human Services (e.g., the Office of Inspector General), the U.S. Department of Justice, state Attorneys General, and other state and local government agencies. All of these activities are also potentially subject to federal and state consumer protection and unfair competition laws. Violations of these laws are punishable by prison sentences, criminal fines, administrative civil money penalties, and exclusion from participation in federal healthcare programs.
There are also an increasing number of state laws that require manufacturers to make reports to states on pricing and marketing information or impose other special requirements for the sale and marketing of drug products. Many of these laws contain ambiguities as to what is required to comply with the laws. In addition, federal and state “transparency laws” require manufacturers to track and report certain payments made to healthcare providers and, under some state laws, other information concerning our products. These laws may affect our sales, marketing and other promotional activities by imposing administrative and compliance burdens on us. In addition, our reporting actions could be subject to the penalty provisions of the pertinent state and federal authorities.
20
Drug Supply Chain Security Act. Title II (the Drug Supply Chain Security Act, or DSCSA), of the Drug Quality Security Act imposes on manufacturers of certain pharmaceutical products new obligations related to product tracking and tracing, among others, which began a several-year phase-in process in 2015. Among the requirements of this legislation, manufacturers subject to the DSCSA will be required to provide certain information regarding the drug product to individuals and entities to which product ownership is transferred, label drug product with a product identifier (i.e., serialize) and keep certain records regarding the drug product. The transfer of information to subsequent product owners by manufacturers must be done electronically. Covered manufacturers will also be required to verify that purchasers of the manufacturers’ products are appropriately licensed. Further, under the DSCSA, covered manufacturers have drug product investigation, quarantine, disposition, and notification responsibilities related to product suspected or reasonably believed to be counterfeit, diverted, stolen, intentionally adulterated the subject of fraudulent transactions or otherwise unfit for distribution such that they would be reasonably likely to result in serious health consequences or death. Anti-counterfeiting and serialization requirements similar to those under the DSCSA have also been adopted in the EU and became effective in February 2019.
Foreign Regulatory Requirements. Outside of the U.S., our ability to conduct clinical trials or market our products will also depend on receiving the requisite authorizations from the appropriate regulatory authorities. The foreign regulatory approval processes include similar requirements and many of the risks associated with the FDA and/or the EMA approval process described above, although the precise requirements may vary from country to country. In the EU, once an MAA is granted, the product must be “placed on the market” in at least one EEA country within three years of the date of authorization. “Placed on the market” is defined as when the medicinal product is “released into the distribution chain,” i.e., out of the direct control of the marketing authorization holder. In July 2018, we placed OMIDRIA on the market in the EU, on a limited basis, which maintained the ongoing validity of the European marketing authorization for OMIDRIA. A marketing authorization will cease to be valid if a product previously placed on the market is no longer actually present on the market for three consecutive years.
Hatch-Waxman Act. In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent with claims that cover the applicant’s drug or an approved method of use of the drug. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential competitors in support of approval of an ANDA or a 505(b)(2) application. An ANDA provides for marketing of a drug that has the same active ingredients and, in some cases (e.g., ophthalmology), also the same inactive ingredients, in the same strengths and dosage form as the listed drug and has been shown through testing to be bioequivalent to the listed drug or receives a waiver from bioequivalence testing. ANDA applicants are generally not required to conduct or submit results of preclinical or clinical tests to prove the safety or effectiveness of their drug, other than the requirement for bioequivalence testing. Drugs approved in this way are considered therapeutically equivalent, and are commonly referred to as “generic equivalents” to the listed drug. These drugs then generally can be substituted by pharmacists under prescriptions written for the original listed drug.
The ANDA or 505(b)(2) applicant is required to certify to the FDA concerning any patents listed for the referenced approved drug in the FDA’s Orange Book. Specifically, the applicant must certify that: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid, unenforceable or will not be infringed by the new drug. A certification that the new drug will not infringe the already approved drug’s listed patents or that such patents are invalid or unenforceable is called a Paragraph IV certification. If the ANDA or 505(b)(2) applicant does not include a Paragraph IV certification, the ANDA or 505(b)(2) application will not be approved until all of the listed patents claiming the referenced drug have expired, except for any listed patents that only apply to uses of the drug not being sought by the ANDA or 505(b)(2) applicant.
If the ANDA or 505(b)(2) applicant has made a Paragraph IV certification, the applicant must also send notice of a Paragraph IV Notice Letter to the NDA and patent holders once the ANDA or 505(b)(2) application has been accepted for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV Notice Letter. The filing of a patent infringement lawsuit within 45 days of the receipt of notice of a Paragraph IV Notice Letter automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, modification by a court or a decision in the infringement case that is favorable to the ANDA or 505(b)(2) applicant.
The ANDA or 505(b)(2) application also will not be approved until any applicable non-patent exclusivity, such as exclusivity for obtaining approval of a new chemical entity, listed in the Orange Book for the referenced drug has expired. The U.S. Drug Price Competition and Patent Term Restoration Act of 1984, more commonly known as the “Hatch-Waxman Act,” provides a period of five years following approval of a drug containing no previously approved active moiety, during which ANDAs for generic versions of those drugs and 505(b)(2) applications referencing those drugs cannot be submitted unless the
21
submission contains a Paragraph IV challenge to a listed patent, in which case the submission may be made four years following the original drug approval. Federal law provides for a period of three years of exclusivity following approval of a listed drug that contains previously approved active ingredients but is approved in a new dosage form, route of administration or combination, or for a new use, the approval of which was supported by new clinical trials other than bioavailability studies that were essential to the approval and conducted by or for the sponsor. During those three years of exclusivity, FDA cannot grant approval of an ANDA or 505(b)(2) application for the protected dosage form, route of administration or combination, or use of that listed drug.
Biosimilars. In the U.S., the FDA regulates biologics under the FDCA, the PHSA, and implementing regulations. The enactment of federal health care reform legislation in March 2010 provided a new pathway for approval of follow-on biologics (i.e., biosimilars) under the PHSA. Licensure by the FDA is dependent upon many factors, including a showing that the proposed biosimilar is “highly similar” to the reference product, notwithstanding minor differences in clinically inactive components, and has no clinically meaningful differences from the reference product in terms of safety, purity, and potency. The types of data ordinarily required in a biosimilar application to show high similarity include analytical data, animal studies (including toxicity studies), and clinical studies (including immunogenicity and pharmacokinetic/pharmacodynamic studies). A biosimilar must seek licensure for a condition of use for which the reference product is licensed.
Furthermore, the PHSA provides that for a biosimilar to be considered “interchangeable” (i.e., the biological product may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product), the applicant must make an additional showing that the biosimilar can be expected to produce the same clinical result as the reference product in any given patient, and if the product is administered more than once to a patient, that risks in terms of safety or diminished efficacy of alternating or switching between the biological product and the reference product is no greater than the risk of using the reference product without switching. Although the FDA has provided guidance on what information and data an applicant should submit to enable an interchangeability determination, thus far the FDA has not licensed any biologic as being interchangeable with its reference product.
In the EU, a pathway for the approval of biosimilars has existed since 2005.
Healthcare compliance laws. In the U.S., commercialization of OMIDRIA and our product candidates, if approved, is subject to regulation and enforcement under a number of federal and state healthcare compliance laws administered and enforced by various agencies. These include, but are not limited to, the following:
| • | the federal Anti-Kickback Statute, which prohibits offering or paying anything of value to a person or entity to induce or reward referrals for goods or services reimbursed by a federal health care program such as Medicare or Medicaid; |
| • | the federal False Claims Act, which prohibits presenting or causing to be presented a false claim for payment by a federal health care program, and which has been interpreted to also include claims caused by improper drug-manufacturer product promotion or the payment of kickbacks; |
| • | a variety of governmental pricing, price reporting, and rebate requirements, including those under Medicaid and the Veterans Health Care Act; and |
| • | the so-called Sunshine Act and certain provisions of the Affordable Care Act, which require that we report to the federal government information on certain financial payments and other transfers of value made to certain healthcare providers and institutions, as well as certain information regarding our distribution of drug samples. |
In addition to these federal law requirements, several U.S. states have enacted similar laws requiring periodic reporting and/or disclosure related to our marketing, sales and other activities, or regulating certain sales and marketing activities, such as provision of meals, gifts or entertainment to certain healthcare providers. We may also be subject to federal or state privacy laws if we receive protected patient health information.
Similar requirements apply to our operations outside of the U.S. Laws in the U.S. such as the Foreign Corrupt Practices Act prohibit the offering or payment of bribes or inducements to foreign public officials for business, including physicians or other medical professionals who are employees of public health care entities. In addition, many non-U.S. jurisdictions in which we operate, or may operate in the future, have their own laws similar to the healthcare compliance laws that exist in the U.S.
Pharmaceutical Pricing and Reimbursement
Overview. In both U.S. and foreign markets, our ability to commercialize our products and product candidates successfully, and to attract commercialization partners for our products and product candidates, depends in significant part on the availability of adequate financial coverage and reimbursement from third-party payers including, in the U.S., managed care organizations and other private health insurers as well as governmental payers such as the Medicare and Medicaid programs.
22
Reimbursement by a third-party payer may depend on a number of factors, including the payer’s determination that use of a product is:
| • | a covered benefit under its health plan; |
| • | safe, effective and medically necessary; |
| • | appropriate for the specific patient; |
| • | cost-effective; and |
| • | neither experimental nor investigational. |
Reimbursement by government payers is based on statutory authorizations and complex regulations that may change with annual or more frequent rulemaking, as well as legislative reform measures.
Third-party private and governmental payers are increasingly challenging the prices charged for medicines and examining their cost-effectiveness in addition to their safety and efficacy. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the cost effectiveness of our products or product candidates. Even with the availability of such studies, third-party private and/or governmental payers may not provide coverage and reimbursement for our products or product candidates, in whole or in part.
United States. Political, economic and regulatory influences are subjecting the healthcare industry in the U.S. to fundamental changes. There have been, and we expect there will continue to be, legislative and regulatory proposals to change the healthcare system in ways that could significantly affect our business. For example, the 2010 Affordable Care Act, or the ACA, is intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. Other legislative changes included a two percent across-the-board reduction to Medicare payments to providers, effective April 1, 2013, which, due to subsequent legislative amendments, will stay in effect through fiscal year 2025 unless additional congressional action is taken. The American Taxpayer Relief Act of 2012, among other things, reduced Medicare payments to several providers, and increased the period for the government to recover overpayments to providers from three to five years. President Trump and various members of Congress have expressed a desire to repeal all or portions of the ACA and, in December 2017, portions of the ACA dealing with the individual mandate insurance requirement were effectively repealed by the Tax Cuts and Jobs Act of 2017. In December 2018, a federal district court judge in Texas found the ACA to be unconstitutional, although the ruling was stayed while the case is appealed. President Trump, the Secretary of Health and Human Services, various members of Congress and CMS have made statements and/or issued proposals regarding containment of drug prices through various means, including enabling CMS to negotiate U.S. drug pricing to align with foreign drug pricing, pricing transparency measures, reform of drug rebate programs, and conditioning coverage and reimbursement of certain drugs upon the prior failure or inadequacy of less expensive therapies (sometimes referred to as “step therapy”). We cannot predict the ultimate content, timing or effect of any healthcare reform legislation or executive order or the impact that the resulting changes may have on us.
We are unable to predict what additional legislation, regulations, policies or court orders, if any, relating to the healthcare industry or coverage and reimbursement may be enacted or imposed in the future or what effect such legislation, regulations, policies or court orders would have on our business. Any cost-containment measures, including those listed above, or other healthcare system reforms that are adopted could have a material adverse effect on our business prospects and financial operations.
Europe. Governments in the various member states of the EU influence or control the price of medicinal products in their countries through their pricing and reimbursement rules and control of national healthcare systems that fund a large part of the cost of those products to consumers. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials or pharmacoeconomic studies that assess the cost-effectiveness of a product or product candidate relative to currently available therapies or relative to a specified standard. The downward pressure on healthcare costs in general, and prescription medicines in particular, has become very intense and is creating increasingly high barriers to the entry of new products in these markets.
Research and Development
We have built a research and development organization that includes expertise in discovery research, preclinical development, product formulation, analytical and medicinal chemistry, manufacturing, clinical development and regulatory and quality assurance. We operate cross-functionally and are led by an experienced research and development management team. We use rigorous project management techniques to make disciplined strategic decisions regarding our research and development programs and to limit the risk profile of our product pipeline. We also access relevant market information and key
23
opinion leaders in creating target product profiles and, when appropriate, as we advance our programs to commercialization. We engage third parties on a limited basis to conduct portions of our preclinical research; however, we are not substantially dependent on any third parties for our preclinical research nor do any of these third parties conduct a major portion of our preclinical research. We also engage multiple clinical sites to conduct our clinical trials. None of these sites conduct the major portion of our clinical trials and we are not substantially dependent on any one of them.
Employees
As of February 12, 2019, we had 236 full-time employees, 60 of whom are in research and development, 132 of whom are in sales and marketing and 44 of whom are in finance, legal, business development and administration. Our full-time employees include six with M.D.s and 37 with Ph.Ds., of whom five and 36, respectively, are in research and development. None of our employees is represented by a labor union, and we consider our employee relations to be good.
Executive Officers and Significant Employees
The following table provides information regarding our executive officers and significant employees as of March 1, 2019:
Name | Age | Position(s) | ||
| Executive Officers: | ||||
| Gregory A. Demopulos, M.D. | 60 | President, Chief Executive Officer and Chairman of the Board of Directors | ||
| Michael A. Jacobsen | 60 | Vice President, Finance, Chief Accounting Officer and Treasurer | ||
| Marcia S. Kelbon, J.D., M.S. | 59 | Vice President, Patent, General Counsel and Secretary | ||
| Significant Employees: | ||||
| Christopher S. Bral, Ph.D. | 53 | Vice President, Nonclinical Development | ||
| Timothy M. Duffy | 58 | Vice President, Business Development | ||
| Timi Edeki, M.D., Ph.D. | 58 | Vice President, Clinical Development | ||
| George A. Gaitanaris, M.D., Ph.D. | 62 | Vice President, Science and Chief Scientific Officer | ||
| Daniel G. Kirby | 47 | Vice President, Head of Commercial | ||
| Eckhard S.D. Leifke, M.D. | 55 | Vice President, Clinical Development and Chief Medical Officer | ||
| Justin McCue, Ph.D. | 44 | Vice President, Chemistry, Manufacturing and Controls | ||
| Catherine A. Melfi, Ph.D. | 59 | Vice President, Regulatory Affairs & Quality Systems and Chief Regulatory Officer | ||
| J. Steven Whitaker, M.D., J.D. | 63 | Vice President, Clinical Development | ||
Gregory A. Demopulos, M.D. founded our company and has served as our president, chief executive officer and chairman of the board of directors since June 1994. He also served as our chief financial officer and treasurer from January 2009 to October 2013 in an interim capacity and as our chief medical officer from June 1994 to March 2010. Prior to founding Omeros, Dr. Demopulos completed his residency in orthopedic surgery at Stanford University and his fellowship training in hand and microvascular surgery at Duke University. Dr. Demopulos currently serves on the board of trustees of the Smead Funds Trust, an open-end mutual fund company registered under the Investment Company Act of 1940. Dr. Demopulos received his M.D. from the Stanford University School of Medicine and his B.S. from Stanford University. Dr. Demopulos is the brother of Peter A. Demopulos, M.D., a member of our board of directors.
Michael A. Jacobsen has served as our vice president, finance, chief accounting officer and treasurer since October 2013. Prior to joining Omeros, Mr. Jacobsen served as vice president of finance of Sarepta Therapeutics, Inc. from September 2011 to
24
May 2013 and as its chief accounting officer from September 2011 to December 2012. From April 2007 to August 2011, Mr. Jacobsen was vice president and chief accounting officer at ZymoGenetics, Inc. Prior to his service with ZymoGenetics, Mr. Jacobsen held various roles at ICOS Corporation, including senior director of finance and corporate controller. From April 1995 to October 2001, Mr. Jacobsen held vice president of finance or chief financial officer roles at three companies in the software, computer hardware and internet retailing industries, two of which were publicly traded. Mr. Jacobsen is a certified public accountant and received his bachelor’s degree in accounting from Idaho State University.
Marcia S. Kelbon, J.D., M.S. has served as our vice president, patent and general counsel since October 2001 and as our secretary since September 2007. Prior to joining Omeros, Ms. Kelbon was a partner with the firm of Christensen O’Connor Johnson & Kindness, PLLC, where she specialized in U.S. and international intellectual property procurement, management, licensing and enforcement issues. Ms. Kelbon received her J.D. and her M.S. in chemical engineering from the University of Washington and her B.S. from The Pennsylvania State University.
Christopher S. Bral, Ph.D. has served as our vice president, nonclinical development since October 2015. From April 2014 to October 2015, Dr. Bral was the executive director, toxicology at Arrowhead Research Corporation, a biopharmaceutical company. From June 2008 to April 2014, Dr. Bral served as director, drug safety evaluation at Vertex Pharmaceuticals, a biotechnology company. Prior to Vertex, Dr. Bral held various pre-clinical drug safety positions of increasing responsibility at Schering-Plough Research Institute including associate director, drug safety evaluation. Dr. Bral received his Ph.D. in biochemistry and biophysics from Texas A&M University and his B.S. in chemistry from John Carroll University. He has been board-certified in toxicology through the American Board of Toxicology since 2000.
Timothy M. Duffy has served as our vice president, business development since March 2010. From November 2008 to March 2010, Mr. Duffy served as the managing director of Pacific Crest Ventures, a life science consulting firm that he founded. From June 2004 through September 2008, Mr. Duffy served at MDRNA, Inc. (formerly Nastech Pharmaceutical Company, Inc.), a biotechnology company. At MDRNA, he held roles of increasing responsibility in marketing and business development, most recently as the chief business officer. Prior to MDRNA, Mr. Duffy served as vice president, business development at Prometheus Laboratories, Inc., a specialty pharmaceutical company, and as a customer marketing manager at The Procter & Gamble Company. Mr. Duffy received his B.S. from Loras College.
Timi Edeki, M.D., Ph.D. has served as our vice president, clinical development since May 2017. From 2006 through 2016, Dr. Edeki served as principal physician for AstraZeneca PLC, a biopharmaceutical company, and during most of that period also served as senior director, research and development. Prior to his service with AstraZeneca, Dr. Edeki was associate director at Abbott Laboratories from 2003 to 2006. Dr. Edeki is a fellow of the American College of Clinical Pharmacology and currently serves on the editorial board of Clinical Pharmacology and Therapeutics. Dr. Edeki currently holds academic appointments as adjunct Professor of Pharmacology, Physiology, and Internal Medicine at Drexel University and previously as Clinical Professor of Medicine at the Chicago Medical School. Dr. Edeki received his medical and Ph.D. degrees from the University of Lagos and London, respectively, and received fellowship training at Vanderbilt University. He is a Diplomate of both the American Board of Internal Medicine and Clinical Pharmacology.
George A. Gaitanaris, M.D., Ph.D. has served as our vice president, science since August 2006 and as our chief scientific officer since January 2012. From August 2003 until our acquisition of nura, inc., in August 2006, Dr. Gaitanaris served as the chief scientific officer of nura, a company that he co-founded, and that developed treatments for central nervous system disorders. From 2000 to 2003, Dr. Gaitanaris served as president and chief scientific officer of Primal, Inc., a biotechnology company that was acquired by nura in 2003. Prior to co-founding Primal, Dr. Gaitanaris served as staff scientist at the National Cancer Institute. Dr. Gaitanaris received his Ph.D. in cellular, molecular and biophysical studies and his M.Ph. and M.A. from Columbia University and his M.D. from the Aristotelian University of Greece.
Daniel G. Kirby has served as our vice president, head of commercial since November 2018. From March 2018 until September 2018 Mr. Kirby was the U.S. CAR T-cell commercial lead for Celgene Corporation, having joined Celgene following its acquisition of Juno Therapeutics, Inc. Mr. Kirby served as Juno’s Vice President of Market and Market Access from March 2016 until its acquisition by Celgene in March 2018. Prior to his work at Celgene and Juno, Mr. Kirby was the head of marketing at Medivation, Inc. from 2015 to 2016, and spent nearly 15 years in commercial leadership roles at Amgen Inc. In his final role at Amgen, Mr. Kirby oversaw the U.S. customer-facing marketing efforts for the G-CSF franchise, NEULASTA® and NEUPOGEN®. Mr. Kirby’s experience also includes commercial roles at GlaxoSmithKline. Mr. Kirby holds an undergraduate degree from the University of Maryland at College Park.
Eckhard S.D. Leifke, M.D. joined Omeros as our vice president, clinical development in June 2018 and assumed the role as our chief medical officer in August 2018. He brings to Omeros more than 20 years of drug development experience, having built and headed global teams at leading pharmaceutical companies including Bayer, Takeda and, most recently, Sanofi where he was global project head/vice president of early project and external opportunities - cardiovascular and metabolism and
25
global head/vice president of late-stage development diabetes from November 2012 to May 2018. Dr. Leifke has led the global development of multiple early- and late-stage small-molecule and biologic drug candidates to successful market authorizations in the U.S., Europe, Japan and other countries. Dr. Leifke holds a Medical Doctorate from the University of Freiburg, Germany, and is Board-certified in Internal Medicine and Endocrinology.
Justin McCue, Ph.D. has served as our vice president, chemistry, manufacturing and controls (CMC) since September 2018. From March 2018 to September 2018 Dr. McCue served as senior director, process development at Celgene Corporation, having joined Celgene following its acquisition of Juno Therapeutics, Inc. Dr. McCue served as Juno’s senior director, process development from September 2017 until its acquisition by Celgene in March 2018. Prior to Juno, Dr. McCue served at Biogen Inc. between 2004 to 2017, where he held positions of increasing responsibility within CMC, including roles as director of manufacturing science from January 2014 to March 2015 and director of product quality management from March 2015 to September 2016. He began his career in the biopharmaceutical division at Millipore. Dr. McCue earned his Ph.D. in chemical engineering at Massachusetts Institute of Technology after receiving a degree in chemical engineering from the University of California at Berkeley.
Catherine A. Melfi, Ph.D. has served as our vice president, regulatory affairs and quality systems since October 2012 and has served as our chief regulatory officer since April 2016. Dr. Melfi previously served from January 1996 to September 2012 at Eli Lilly and Company, where she held technical and leadership roles of increasing scope and responsibility, including as senior director and scientific director in global health outcomes and regulatory affairs, respectively. Prior to joining Eli Lilly, Dr. Melfi held various faculty and research positions at Indiana University, including appointments in its Economics Department, in the School of Public and Environmental Affairs, and in the Indiana University School of Medicine. Dr. Melfi received her Ph.D. in Economics from the University of North Carolina - Chapel Hill and B.S. in Economics from John Carroll University.
J. Steven Whitaker, M.D., J.D. has served as our vice president, clinical development since joining Omeros in 2010, and served as our chief medical officer from March 2010 to August 2018. From May 2008 to March 2010, Dr. Whitaker served as the chief medical officer, vice president of clinical development at Allon Therapeutics, Inc., a biotechnology company focused on developing drugs for neurodegenerative diseases. From August 2007 to May 2008, he served as a medical consultant to Accelerator Corporation, a biotechnology-company investor and incubator. From May 1994 to May 2007, Dr. Whitaker served at ICOS Corporation, which was acquired by Eli Lilly and Company in 2007. At ICOS, he held roles of increasing responsibility in clinical research and medical affairs, most recently as divisional vice president, clinical research as well as medical director of the Cialis® global product team. Dr. Whitaker received his M.D. from the Indiana University School of Medicine, his J.D. from the University of Washington and his B.S. from Butler University.
Corporate Information
We were incorporated in 1994 as a Washington corporation. Our principal executive offices are located at 201 Elliott Avenue West, Seattle, Washington, 98119, and our telephone number is (206) 676-5000. Our website address is www.omeros.com. We make available, free of charge through our investor relations website at investor.omeros.com, our annual report on Form 10-K, our quarterly reports on Form 10-Q, our current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act, including exhibits to those reports, as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Our websites and the information contained therein or incorporated therein are not intended to be incorporated into this Annual Report on Form 10-K. The SEC maintains a website that contains reports, proxy and information statements, and other information regarding reports that we file or furnish electronically with them at www.sec.gov.
ITEM 1A. RISK FACTORS
The risks and uncertainties described below may have a material adverse effect on our business, prospects, financial condition or operating results. In addition, we may be adversely affected by risks that we currently deem immaterial or by other risks that are not currently known to us. You should carefully consider these risks before making an investment decision. The trading price of our common stock could decline due to any of these risks and you may lose all or part of your investment. In assessing the risks described below, you should also refer to the other information contained in this Annual Report on Form 10-K.
Risks Related to Our Products, Programs and Operations
Our ability to achieve profitability is highly dependent on the commercial success of OMIDRIA. To the extent OMIDRIA is not successful, our business, financial condition and results of operations may be materially adversely affected and the price of our common stock may decline.
26
OMIDRIA is our only product that has been approved by the FDA for commercial sale in the U.S. For the three and 12 months ended December 31, 2018, we recorded net sales of OMIDRIA of $22.0 million and $29.9 million, respectively. Revenues from sales of OMIDRIA have not been sufficient to fund our operations fully in prior periods and we cannot provide assurance that revenues from OMIDRIA sales will be sufficient to fund our operations fully in the future. We will need to generate substantially more product revenue from OMIDRIA to achieve and sustain profitability. We may be unable to sustain or increase revenues generated from OMIDRIA product sales for a number of reasons, including:
| • | pricing, coverage and reimbursement policies of government and private payers such as Medicare, Medicaid, the U.S. Department of Veterans Affairs, group purchasing organizations, insurance companies, health maintenance organizations and other plan administrators; |
| • | a lack of acceptance by physicians, patients and other members of the healthcare community; |
| • | the availability, relative price and efficacy of the product as compared to alternative treatment options or branded, compounded or generic competing products; |
| • | an unknown safety risk; |
| • | the failure to enter into and maintain acceptable partnering arrangements for marketing and distribution of OMIDRIA outside of the U.S.; |
| • | changed or increased regulatory restrictions in the U.S., EU and/or other foreign territories; and |
| • | a lack of adequate financial or other resources. |
We cannot definitively project OMIDRIA sales in future periods, or if OMIDRIA will continue to be reimbursed separately after September 30, 2020.
From January 1, 2018 through September 30, 2018, OMIDRIA was not eligible for separate reimbursement under Medicare Part B and, as a result, ASCs and hospitals received from CMS the same procedural payment whether or not OMIDRIA was used for the procedure. Due to the absence of separate reimbursement, we saw a significant reduction in ASC and hospital demand for OMIDRIA beginning in December 2017 and a corresponding decrease in sales to our wholesalers that continued through the third quarter of 2018. Pass-through reimbursement status for OMIDRIA resumed on October 1, 2018 for a two-year period, consistent with the statutory changes included in the Appropriations Act. Although OMIDRIA sales quickly returned to late 2017 levels after restoration of pass-through reimbursement, we cannot predict OMIDRIA sales in future periods with any certainty.
We continue to confirm and seek permanent separate or similar reimbursement for OMIDRIA. However, this requires action from legislative and/or administrative authorities and we cannot guarantee that any such action will be taken or, if taken, when such action will be effective. Accordingly, we can provide no assurances that separate reimbursement for OMIDRIA will continue to be available on or after September 30, 2020, or if available, that the reimbursement rate will be adequate.
If we are unable to obtain permanent separate or similar reimbursement for OMIDRIA, or if such reimbursement is delayed or the reimbursement rate is inadequate, our ability to generate revenue and attain profitability would be adversely affected and there could be a material adverse effect on our financial condition, results of operations and growth prospects and the trading price of our stock could decline.
If OMIDRIA or any other product that we develop and commercialize does not receive adequate coverage or reimbursement from governments and/or private payers, or if we do not establish and maintain market-acceptable pricing for OMIDRIA or those potential other commercialized products, our prospects for revenue and profitability would suffer.
Our revenues depend heavily on the pricing, availability and duration of adequate coverage or reimbursement for the use of products that we or our third-party business partners commercialize, including OMIDRIA, from government, private and other third-party payers, both in the U.S. and in other countries. Any product that we bring to market may not be considered cost-effective and/or the amount reimbursed for any product may be insufficient to allow us to sell the product profitably. Obtaining coverage and reimbursement for any product from each government or third-party payer can be a time-consuming and costly process that may require expansion of staff and/or increased use of third parties and could require us to provide additional supporting scientific, clinical and cost-effectiveness data for the use of our approved products to each payer. We can provide no assurances at this time regarding the cost-effectiveness of OMIDRIA or any of our product candidates. Further, we can provide no assurance that the amounts, if any, reimbursed to surgical facilities for utilization of any of our surgery-related products, including OMIDRIA or any of our product candidates, or to surgeons for the administration and delivery of these products or product candidates will be considered adequate to justify the use of these products or product candidates. The Trump administration has proposed increasing CMS’ power to negotiate with drug makers and possibly set reimbursement based on an index of international prices, and a proposed rule by CMS would, if implemented, empower Part D sponsors and Medicare Advantage plan to use prior authorization and step therapy more aggressively, each of which could adversely impact pricing of our products, including OMIDRIA. We also can provide no assurance that Merit-Based Incentive Payment System,
27
or MIPS, payment adjustments for eligible professionals under the Medicare Access and CHIP Reauthorization Act of 2015 will not adversely impact OMIDRIA sales revenue.
There may be significant delays in obtaining coverage or reimbursement for newly approved products, and we may not be able to provide data sufficient to be granted adequate coverage or reimbursement. Even when a payer determines that a product is eligible for reimbursement, coverage may be limited to the uses of a product that are either approved by the FDA (or, in other countries, the relevant country’s regulatory agency) and/or appear in a recognized drug compendium, or other conditions may apply. Moreover, eligibility for coverage does not mean that any product will be reimbursed at a rate that allows us to make a profit in all cases or at a rate that covers our costs, including research, development, manufacturing, sales and distribution. Increasingly, government and private third-party payers that reimburse for healthcare services and products are requiring that companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products, which could adversely impact the pricing of our products. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payers. Pricing may also be adversely affected by changes in the terms, scope and/or complexity of government pricing requirements. Even if we achieve coverage or reimbursement for a product, the initial rate or method at which the product will be reimbursed could become unfavorable to us at the time reimbursement is initiated or in the future or may be of a limited duration. In addition, obtaining acceptable coverage and reimbursement from one payer does not guarantee that we will obtain similar acceptable coverage or reimbursement from another payer.
In non-U.S. jurisdictions, we must obtain separate reimbursement approvals and comply with related foreign legal and regulatory requirements. In some countries, including those in the EU, our products may be subject to government price controls. Pricing negotiations with governmental authorities can take a considerable amount of time and expenditure of resources after the receipt of marketing approval for a product. We provide no assurances that the price of any product in one or more of these countries or regions will allow us to make a profit or cover our costs, including research, development, manufacturing, sales and distribution, and as a result we may decide to delay, potentially indefinitely, initiating sales in the particular country or region.
If the reimbursement or pricing that we are able to obtain and maintain for any product that we develop and commercialize, including OMIDRIA, is inadequate, is significantly delayed or is subject to overly restrictive conditions, our ability to generate revenue, attain profitability and/or commercialize our product candidates may be impaired and there could be a material adverse effect on our business, financial condition, results of operations and growth prospects and the trading price of our stock could decline.
Our operating results are unpredictable and may fluctuate.
Our operating results are difficult to predict and will likely fluctuate from quarter to quarter and year to year. We believe that our quarterly and annual results of operations may be affected by a variety of factors, including:
| • | the level and timing of commercial sales of OMIDRIA, as well as our product candidates if and when approved or commercialized; |
| • | the extent of coverage and reimbursement for OMIDRIA; |
| • | the amount of OMIDRIA chargebacks, rebates and product returns; |
| • | the extent of any payments received from collaboration arrangements and development funding as well as the achievement of development and clinical milestones under collaboration and license agreements that we may enter into from time to time and that may vary significantly from quarter to quarter; and |
| • | the timing, cost and level of investment in our research and development activities as well as expenditures we will or may incur to acquire or develop additional technologies, products and product candidates, or in preparation for potential commercialization of our product candidates. |
In addition, the number of procedures in which OMIDRIA or any of our product candidates, if commercialized, would be used may be significantly less than the total number of such procedures performed or total possible market size. These and other factors, including our limited history of product sales since the reinstatement of pass-through reimbursement for OMIDRIA, make it difficult for us to forecast and provide accurate guidance (including updates to prior guidance) related to our expected financial performance. If our operating results are below the expectations of securities analysts or investors, the trading price of our stock could decline.
We have incurred cumulative operating losses since inception. If we are unable to raise additional capital when needed, our commercial operations may be limited and we may be unable to complete the development and commercialization of our product candidates or to continue our other preclinical development programs.
28
Our operations have consumed substantial amounts of cash since our incorporation and, as of December 31, 2018, we had an accumulated deficit of approximately $650.1 million. We expect to continue to spend substantial amounts to:
| • | initiate and conduct clinical trials and manufacture clinical and registration batches for our programs and product candidates; |
| • | continue OMIDRIA sales and marketing; |
| • | continue research and development in our programs; |
| • | make principal, interest and fee payments as required under our 6.25% Convertible Senior Notes due 2023, or the Convertible Notes; and |
| • | commercialize and launch product candidates for which we may receive regulatory approval. |
We expect to continue to incur additional losses until such time as we generate significant revenue from the sale of OMIDRIA, other commercial products and/or partnering. We are unable to predict the extent of any future losses and cannot provide assurance that we will generate sufficient revenue from OMIDRIA or other commercial products in the future to fund our operations fully. If we are unable to generate sufficient revenue from the sale of OMIDRIA, other commercialized products and/or partnering arrangements, we may never become and remain profitable and will be required to raise additional capital to continue to fund our operations. We cannot be certain that additional capital will be available to us on acceptable terms, if at all, when required. Adverse developments to our financial condition or business, as well as disruptions in the global equity and credit markets, may limit our ability to access capital. If we do not raise additional capital when needed through one or more funding avenues, such as debt or equity financings or corporate partnering, we may have to significantly delay, scale back or discontinue the development or commercialization of one or more of our product candidates or one or more of our preclinical programs or other research and development initiatives. In addition, we may be required to seek collaborators for one or more of our current or future products at an earlier stage than otherwise would be desirable or on terms that are less favorable than otherwise might be available or to relinquish or license on unfavorable terms our rights to technologies or products that we otherwise would seek to develop or commercialize ourselves. We also may have insufficient funds or otherwise be unable to advance our preclinical programs, such as potential new drug targets developed from our GPCR program, to a point where they can generate revenue through partnerships, collaborations or other arrangements. Any of these actions could limit the amount of revenue we are able to generate and harm our business and prospects.
Management, as well as our independent registered public accounting firm, have concluded that a substantial doubt is deemed to exist concerning our ability to continue as a going concern.
Accounting Standards Update, or ASU, 2014-15, requires management to assess our ability to continue as a going concern for one year after the date the financial statements are issued. As further discussed in Part II, Item 8, “Note 1--Organization and Basis of Presentation” to our Consolidated Financial Statements in this Annual Report on Form 10-K, substantial doubt is deemed to exist about the company’s ability to continue as a going concern through March 1, 2020. Our financial statements do not include any adjustment relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern. Our ability to continue as a going concern will require us to generate positive cash flow from operations, obtain additional financing, enter into strategic alliances and/or sell assets. The reaction of investors to the inclusion of a going concern statement in this Annual Report on Form 10-K, our current lack of cash resources and our potential inability to continue as a going concern may materially adversely affect our share price and our ability to raise new capital, enter into strategic alliances and/or make our scheduled debt payments on a timely basis or at all. If we become unable to continue as a going concern, we may have to liquidate our assets and the values we receive for our assets in liquidation or dissolution could be significantly lower than the values reflected in our financial statements.
We are subject to extensive government regulation and the failure to comply with these regulations may have a material adverse effect on our operations and business.
Both before and after approval of any product, we and our suppliers, contract manufacturers and clinical investigators are subject to extensive regulation by governmental authorities in the U.S. and other countries, covering, among other things, testing, manufacturing, quality control, clinical trials, post-marketing studies, risk management plans, labeling, advertising, promotion, distribution, import and export, governmental pricing, price reporting and rebate requirements. Failure to comply with applicable requirements could result in one or more of the following actions: warning letters; unanticipated expenditures; delays in approval or refusal to approve a product candidate; product recall or seizure; interruption of manufacturing or clinical trials; operating or marketing restrictions; injunctions; criminal prosecution and civil or criminal penalties including fines and other monetary penalties; adverse publicity; and disruptions to our business. Further, government investigations into potential violations of these laws would require us to expend considerable resources and face adverse publicity and the potential disruption of our business even if we are ultimately found not to have committed a violation.
29
Obtaining FDA approval of our product candidates requires substantial time, effort and financial resources and may be subject to both expected and unforeseen delays, and there can be no assurance that any approval will be granted on any of our product candidates on a timely basis, if at all. Even if we discuss with, and obtain feedback from, the FDA regarding our proposed clinical trials, clinical data collection protocols and nonclinical studies before initiating those trials or studies, the FDA may decide that the design of our clinical trials or clinical data collection protocols as actually run, or our resulting data, are insufficient for approval of our product candidates and may require us to run additional preclinical, clinical or other studies or perform additional work related to chemistry, manufacturing and controls. In addition, we, the FDA or an independent institutional review board or ethics committee may suspend or terminate human clinical trials at any time on various grounds, including a finding that the patients are or would be exposed to an unacceptable health risk or because of the way in which the investigators on whom we rely carry out the trials. If we are required to conduct additional trials or to conduct other testing of our product candidates beyond that which we currently contemplate for regulatory approval, if we are unable to complete our clinical trials or other testing successfully, or if the results of these and other trials or tests fail to demonstrate efficacy or raise safety concerns, we may face substantial additional expenses, be delayed in obtaining marketing approval for our product candidates or may never obtain marketing approval.
We are also required to comply with extensive governmental regulatory requirements after a product has received marketing authorization. Governing regulatory authorities may require post-marketing studies that may negatively impact the commercial viability of a product. Once on the market, a product may become associated with previously undetected adverse effects and/or may develop manufacturing difficulties. We are required to comply with other post-marketing requirements including cGMPs, advertising and promotion restrictions, pharmacovigilance requirements including risk management activities, reporting and recordkeeping obligations, and other requirements. As a result of any of these or other problems, a product’s regulatory approval could be withdrawn, which could harm our business and operating results. In addition, we must establish and maintain an effective healthcare compliance program in order to comply with U.S. and other laws applicable to marketed drug products and, in particular, laws (such as the Anti-Kickback Statute, the False Claims Act and the Sunshine Act) applicable when drug products are purchased or reimbursed by a federal or state healthcare program. U.S. laws such as the Foreign Corrupt Practices Act prohibit the offering or payment of bribes or inducements to foreign public officials, including potentially physicians or other medical professionals who are employees of public health care entities in jurisdictions outside the U.S. In addition, many countries have their own laws similar to the healthcare compliance laws that exist in the U.S. Implementing and maintaining an effective compliance program requires the expenditure of significant time and resources. If we are found to be in violation of any of these laws, we may be subject to significant penalties, including but not limited to civil or criminal penalties, damages and fines as well as exclusion from government healthcare programs.
We may face difficulties from changes to current regulations as well as future legislation.
Existing regulatory policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the U.S. or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability.
There is uncertainty with respect to the impact that health care reform legislation may have on coverage and reimbursement for healthcare items and services covered by plans that are authorized by the ACA. President Trump and various members of Congress have expressed a desire to repeal all or portions of the ACA and, in December 2017, portions of the ACA dealing with the individual mandate insurance requirement were effectively repealed by the Tax Cuts and Jobs Act of 2017. Congress and/or President Trump may seek to repeal other aspects of the ACA. In December 2018, a federal district court judge in Texas found the ACA to be unconstitutional, although the ruling was stayed while the case is appealed. President Trump, the Secretary of Health and Human Services, various members of Congress and CMS have made statements and/or issued proposals regarding containment of drug prices through various means, including measures designed to enable CMS greater power to negotiate U.S. drug pricing and/or set reimbursement based on an index of international drug prices, pricing transparency measures, and reforms that would empower Part D sponsors and Medicare Advantage plans to more aggressively use prior authorization and step therapy as a means to control prescription drug costs. We cannot predict the ultimate content, timing or effect of any healthcare reform legislation, rulemaking or executive order, or the impact that the resulting changes may have on us.
We expect that the ACA, if it remains in effect, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and apply downward pressure on the price that we receive for any approved product. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payers. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate sufficient revenue, attain and/or maintain profitability or commercialize our product
30
candidates. We cannot be sure whether additional legislative changes will be enacted, or whether FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on OMIDRIA or the marketing approvals of our product candidates, if any, may be.
Failure to obtain and maintain regulatory approval in foreign jurisdictions would prevent us from marketing our products internationally.
We intend to have OMIDRIA and our product candidates, if approved, marketed outside the U.S. In order to market our products in non-U.S. jurisdictions, we or our partners must obtain separate regulatory approvals and comply with numerous and varying regulatory requirements. The regulatory approval procedure varies among countries and can involve additional testing and data review. The requirements governing marketing authorization, the conduct of clinical trials, pricing and reimbursement vary from country to country. Approval by the FDA or the EMA does not ensure approval by regulatory agencies in other jurisdictions, and approval by one foreign regulatory authority does not ensure approval by regulatory agencies in other foreign countries or by the FDA. The time required to obtain regulatory approval outside the U.S. and EU may differ from that required to obtain FDA or EMA approval. The foreign regulatory approval process may include all of the risks associated with obtaining FDA approval discussed in these “Risk Factors” and we may not obtain foreign regulatory approvals on a timely basis, or at all. In addition, even if we were able to obtain regulatory approval for a product in one or more foreign jurisdictions, we may need to complete additional requirements to maintain that approval and our ability to market the product in the applicable jurisdiction.
OMIDRIA, as well as any of our product candidates, if approved, that are marketed outside of the United States, may face a variety of risks associated with international operations that, if realized, could materially adversely affect our business.
We may be subject to additional risks for OMIDRIA or any of our product candidates that are marketed outside the U.S., including:
| • | reduced protection for intellectual property rights; |
| • | unexpected changes in tariffs, trade barriers and regulatory requirements, including those associated with the withdrawal of the United Kingdom from the EU; |
| • | economic weakness, including inflation, or political instability in particular foreign economies and markets; |
| • | foreign currency fluctuations and other obligations incident to doing business in another country; and |
| • | business interruptions resulting from geopolitical actions, including war and terrorism or natural disasters including earthquakes, typhoons, floods and fires. |
Any of these risks, if realized, could increase our operating expenses and reduce our revenues.
We have no internal capacity to manufacture commercial or clinical supplies of OMIDRIA or our product candidates and intend to rely solely on third-party manufacturers. If the contract manufacturers that we rely on experience difficulties manufacturing and supplying OMIDRIA or our product candidates, or fail FDA or other regulatory inspections, our clinical trials, regulatory submissions and ability to sell OMIDRIA or any other commercialized product and generate revenue may be significantly limited or delayed.
We rely and intend to continue to rely on third-party manufacturers to produce commercial quantities of OMIDRIA and clinical drug supplies of our product candidates that are needed for clinical trials and to support NDAs, BLAs, or similar applications to regulatory authorities seeking marketing approval for our product candidates, as well as to produce inventory of our product candidates in anticipation of potential commercialization. We cannot provide any assurance that we will be able to enter into or maintain these types of arrangements on commercially reasonable terms, or at all. If we or the manufacturer were to terminate one of these arrangements early, or the manufacturer was unable to supply product quantities sufficient to meet our requirements, we would be required to transfer manufacturing to an approved alternative facility and/or establish additional manufacturing and supply arrangements. We may also need to establish additional or replacement manufacturers, potentially with little or no notice, in the event that one of our manufacturers fails to comply with FDA and/or other pharmaceutical manufacturing regulatory requirements. Even if we are able to establish additional or replacement manufacturers, identifying these sources and entering into definitive supply agreements and obtaining regulatory approvals may require a substantial amount of time and cost and may create a shortage of the product. It can take several years to qualify and validate a new contract manufacturer, and we cannot guarantee that we would be able to complete in a successful and timely manner the appropriate validation processes or obtain the necessary regulatory approvals for one or more additional or replacement manufacturers. Such alternate supply arrangements may not be available on commercially reasonable terms, or at all.
31
Additionally, if we are unable to engage multiple suppliers to manufacture our products, we may have inadequate supply to meet demand for our product.
In addition, narsoplimab is a biologic drug product and any other product candidate from certain of our programs, including but not limited to MASP-2 and MASP-3, could be a biologic drug product. We do not have the internal capability to produce biologics for use in clinical trials or on a commercial scale. There are only a limited number of manufacturers of biologic drug products and we may be unable to enter into agreements on commercially reasonable terms with a sufficient number of them to meet clinical and/or commercial demand, if at all. The regulatory requirements for commercial supply are more stringent than for clinical supply and we cannot guarantee that a contract manufacturer producing drug product for clinical trials will be able to complete in a successful and timely manner the appropriate validation processes or obtain the necessary regulatory approvals for marketing approval and commercial supply in a timely manner or at all.
Our contract manufacturers may encounter difficulties with formulation, manufacturing, supply chain and/or release processes that could result in delays in clinical trials and/or regulatory submissions or that could impact adversely the commercialization of our products or product candidates, as well as in the initiation of enforcement actions by the FDA and other regulatory authorities. These difficulties also could result in the recall or withdrawal of a product from the market or a failure to have adequate supplies to meet market demand. If the safety or manufacturing quality of OMIDRIA or any product candidate supplied by contract manufacturers is compromised due to one or more of those contract manufacturers’ failure to adhere to applicable laws or for other reasons, we may not be able to maintain regulatory approval of OMIDRIA, to continue sales and marketing of OMIDRIA, to run clinical trials or to obtain and maintain regulatory approval for one or more of our product candidates, which would harm our business and prospects significantly.
Any significant delays in the manufacture and/or supply of clinical or commercial supplies could materially harm our business, financial condition, results of operations and prospects.
Ingredients, excipients, test kits and other materials necessary to manufacture OMIDRIA or our product candidates may not be available on commercially reasonable terms, or at all, which may adversely affect the development and commercialization of OMIDRIA or those product candidates.
We and our third-party manufacturers must obtain from third-party suppliers the active pharmaceutical ingredients, excipients, and/or other raw materials plus primary and secondary packaging materials necessary for our contract manufacturers to produce OMIDRIA and our product candidates for our clinical trials and, to the extent approved or commercialized, for commercial distribution. Although we have entered or intend to enter into agreements with third-party suppliers that will guarantee the availability and timely delivery of active pharmaceutical ingredients, excipients, test kits and materials for OMIDRIA and our product candidates, we have not yet entered into agreements for the supply of all such ingredients, excipients, test kits or materials, and we may be unable to secure all such supply agreements or guarantees on commercially reasonable terms, if at all. Even if we were able to secure such agreements or guarantees, our suppliers may be unable or choose not to provide us the ingredients, excipients, test kits or materials in a timely manner or in the quantities required. If we or our third-party manufacturers are unable to obtain the quantities of these ingredients, excipients or materials that are necessary for the manufacture of commercial supplies of OMIDRIA, our ability to generate revenue from the sale of OMIDRIA would be materially and adversely affected. Further, if we or our third-party manufacturers are unable to obtain active pharmaceutical ingredients, excipients, test kits and materials as necessary for our clinical trials or for the manufacture of commercial supplies of our product candidates, if approved, potential regulatory approval or commercialization would be delayed, which would materially and adversely affect our ability to generate revenue from the sale of our product candidates.
If our clinical trials or clinical protocols are delayed, suspended or terminated, we may be unable to develop our product candidates on a timely basis, which would adversely affect our ability to obtain regulatory approvals, increase our development costs and delay or prevent commercialization of approved products.
We cannot predict whether we will encounter problems with any of our completed, ongoing or planned clinical trials or clinical data collection protocols that will cause regulatory agencies, institutional review boards or ethics committees, or us to delay our clinical trials or suspend or delay the analysis of the data from those trials. Clinical trials and clinical data protocols can be delayed for a variety of reasons, including:
| • | discussions with the FDA, the EMA or other foreign authorities regarding the scope or design of our clinical trials or clinical data collection protocols; |
| • | delays or the inability to obtain required approvals from institutional review boards, ethics committees or other responsible entities at clinical sites selected for participation in our clinical trials; |
| • | delays in enrolling patients into clinical trials or collecting historical control data for any reason including disease severity, trial or data collection protocol design, study eligibility criteria, patient population size (e.g., for orphan diseases or for some pediatric indications), proximity and/or availability of clinical trial sites for prospective patients, |
32
availability of competing therapies and clinical trials, regional differences in diagnosis and treatment, perceived risks and benefits of the product or product candidate, physician patient referral practices or the ability to monitor patients adequately before and after treatment;
| • | lower than anticipated retention rates of patients in clinical trials; |
| • | the need to repeat or conduct additional clinical trials as a result of inconclusive or negative results, failure to replicate positive early clinical data in subsequent clinical trials, failure to deliver an efficacious dose of a product candidate, poorly executed testing, a failure of a clinical site to adhere to the clinical protocol, an unacceptable study design or other problems; |
| • | adverse findings in clinical or nonclinical studies related to the safety of our product candidates in humans; |
| • | an insufficient supply of product candidate materials or other materials necessary to conduct our clinical trials; |
| • | the need to qualify new suppliers of product candidate materials for FDA and foreign regulatory approval; |
| • | an unfavorable inspection or review by the FDA or other regulatory authority of a clinical trial site or records of any clinical investigation; |
| • | the occurrence of unacceptable drug-related side effects or adverse events experienced by participants in our clinical trials; |
| • | the suspension by a regulatory agency of a trial by imposing a clinical hold; or |
| • | the amendment of clinical trial or data collection protocols to reflect changes in regulatory requirements and guidance or other reasons as well as subsequent re-examination of amendments to clinical trial or data collection protocols by institutional review boards or ethics committees. |
In addition, a clinical trial or development program may be suspended or terminated by us, the FDA or other regulatory authorities, or institutional review boards or ethics committees due to a number of factors, including:
| • | failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols; |
| • | inspection of the clinical trial operations or trial sites by the FDA or other regulatory authorities resulting in the imposition of a clinical hold; |
| • | the failure to remove a clinical hold in a timely manner, if at all; |
| • | unforeseen safety issues or any determination that a trial presents unacceptable health risks; |
| • | inability to deliver an efficacious dose of a product candidate; or |
| • | lack of adequate funding to continue the clinical trial or development program, including as a result of unforeseen costs due to enrollment delays, requirements to conduct additional trials and studies and/or increased expenses associated with the services of our contract research organizations, or CROs, or other third-parties. |
If the results of our clinical trials are not available when we expect or if we encounter any delay in the analysis of data from our clinical trials, we may be unable to file for regulatory approval or conduct additional clinical trials on the schedule we currently anticipate. Many of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of a product candidate. Any delays in completing our clinical trials could increase our development costs, could slow down our product development and regulatory submission process, could delay our receipt of product revenue and could make it difficult to raise additional capital. In addition, significant clinical trial delays also could allow our competitors to bring products to market before we do and impair our ability to commercialize our future products, potentially harming our business.
Because we have a number of product candidates and development programs, we may expend our limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications for which there is a greater likelihood of obtaining regulatory approval and that may be more profitable, if approved.
We have limited resources and must focus on the product candidates and clinical and preclinical development programs that we believe are the most promising. As a result, we may forgo or delay the pursuit of opportunities with other product candidates or other indications that later prove to have greater commercial potential and may not be able to progress development programs as rapidly as otherwise possible. Further, if we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product through collaboration, license or other royalty arrangements in cases in which it would have been advantageous for us to retain sole development and commercialization rights.
Our preclinical programs may not produce product candidates that are suitable for clinical trials, our product candidates may not successfully complete clinical development and/or our product candidates may not be suitable for successful commercialization or generation of revenue through partnerships.
33
We must successfully complete preclinical testing, which may include demonstrating efficacy and the lack of toxicity in established animal models, before commencing clinical trials for any product candidate. Many pharmaceutical and biological product candidates do not successfully complete preclinical testing. There can be no assurance that positive results from preclinical studies will be predictive of results obtained from subsequent preclinical studies or clinical trials. Even if preclinical testing is successfully completed, we cannot be certain that any product candidates that do advance into clinical trials will successfully demonstrate safety and efficacy in clinical trials. Even if we achieve positive results in early clinical trials, they may not be predictive of the results in later trials, and safety and/or efficacy outcomes of early clinical trials may not be consistent with outcomes of subsequent clinical trials.
We may incur substantial costs as a result of commercial disputes, claims, litigation or other legal proceedings relating to our business operations, especially with regard to patent and other intellectual property rights, and such costs or an adverse outcome in such a proceeding may adversely affect our financial condition, results of operations and/or stock price.
Our business involves numerous commercial contractual arrangements, important intellectual property rights, potential product liability, uncertainties with respect to clinical development, manufacture and regulatory approvals and other aspects that create heightened risks of disputes, claims and legal proceedings. These include claims that may be faced in one or more jurisdictions related to the safety of our product candidates and products, the development of our product candidates, our ability to obtain regulatory approval for our product candidates, our expectations regarding product development and regulatory approval, sales and marketing practices, commercial disputes including with contract manufacturers, competition, environmental matters, employment matters and other matters. These matters could consume significant time and resources, even if we are successful. Some of our competitors may be able to sustain the costs of complex litigation more effectively than we can because they have substantially greater resources. In addition, we may pay damage awards or settlements or become subject to equitable remedies that could, individually or in the aggregate, have a material negative effect on our financial condition, results of operations or stock price. Any uncertainties resulting from the initiation and continuation of any litigation also could have a material adverse effect on our ability to raise the capital necessary to continue our operations.
We may initiate or become subject to litigation regarding patents and other intellectual property rights. Patent infringement litigation involves many complex technical and legal issues and its outcome is often difficult to predict and the risk involved in doing so can be substantial. Generic drug manufacturers could seek approval to market a generic version of our products or challenge our intellectual property rights with respect to our product candidates.
If we choose to go to court or take other action to enforce our intellectual property rights, we may become subject to claims that our underlying patents are invalid or should not be enforced against that third-party. These lawsuits are expensive and would consume time and other resources even if we were successful in stopping the infringement of our patents. In addition, a lawsuit could result in a finding that some or all of the claims of one or more of our relevant patents are invalid, unenforceable and/or not infringed, and could result in a generic version of any of our products being launched prior to the expiration of our related patents. There is also the risk that, even if the validity of these patents is upheld, a court will refuse to stop the other party on the ground that such other party’s activities do not infringe our patents.
It may not be feasible to detect and undertake patent enforcement action to stop infringing activity by a number of individual entities, each on a small scale, such as compounding pharmacies. Further, our industry has produced a large number of patents and it is not always clear which patents cover various types of products or methods of use. A third party may claim that we or our contract manufacturers are using inventions covered by the third party’s patent rights and may go to court to stop us from engaging in the alleged infringing activity, including making, using or selling our products and product candidates. These lawsuits are costly and could affect our results of operations and divert the attention of managerial and technical personnel. There is a risk that a court would decide that we, or our contract manufacturers, are infringing the third party’s patents and would order us or our contractors to stop the activities covered by the patents. In addition, if we or our contract manufacturers are found to have violated a third party’s patent, we or our contract manufacturers could be ordered to pay damages to the other party. We have agreed to or may agree to indemnify our contract manufacturers against certain patent infringement claims and thus may be responsible for any of their costs associated with such claims and actions. If we were sued for patent infringement, we would need to demonstrate that our products and product candidates or methods of use either do not infringe the patent claims of the relevant patent or that the patent claims are invalid, and we might be unable to do this. Proving invalidity, in particular, is difficult since it requires clear and convincing evidence to overcome the presumption of validity enjoyed by issued patents.
34
It is difficult and costly to protect our intellectual property and our proprietary technologies, and we may not be able to ensure their protection.
Our commercial success will depend in part on obtaining and maintaining patent protection and trade secret protection for the use, formulation and structure of our products and product candidates, the methods used to manufacture them, the related therapeutic targets and associated methods of treatment as well as on successfully defending these patents against potential third-party challenges. Our ability to protect our products and product candidates from unauthorized making, using, selling, offering to sell or importing by third parties is dependent on the extent to which we have rights under valid and enforceable patents that cover these activities.
The patent positions of pharmaceutical, biotechnology and other life sciences companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. Changes in either the patent laws or in interpretations of patent laws in the U.S. and other countries may diminish the value of our intellectual property. Further, the determination that a patent application or patent claim meets all of the requirements for patentability is a subjective determination based on the application of law and jurisprudence. The ultimate determination by the USPTO or by a court or other trier of fact in the U.S., or corresponding foreign national patent offices or courts, on whether a claim meets all requirements of patentability cannot be assured. Although we have conducted searches for third-party publications, patents and other information that may affect the patentability of claims in our various patent applications and patents, we cannot be certain that all relevant information has been identified. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced in our patents or patent applications, in our licensed patents or patent applications or in third-party patents.
We cannot provide assurances that any of our patent applications will be found to be patentable, including over our own prior art patents, or will issue as patents. Neither can we make assurances as to the scope of any claims that may issue from our pending and future patent applications nor to the outcome of any proceedings by any potential third parties that could challenge the patentability, validity or enforceability of our patents and patent applications in the U.S. or foreign jurisdictions. Any such challenge, if successful, could limit patent protection for our products and product candidates and/or materially harm our business.
The degree of future protection for our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
| • | we may not be able to generate sufficient data to support full patent applications that protect the entire breadth of developments in one or more of our programs or platforms; |
| • | it is possible that one or more of our pending patent applications will not become an issued patent or, if issued, that the patent(s) will be sufficient to protect our technology, provide us with a basis for commercially viable products or provide us with any competitive advantages; |
| • | if our pending applications issue as patents, they may be challenged by third parties as not infringed, invalid or unenforceable under U.S. or foreign laws; or |
| • | if issued, the patents under which we hold rights may not be valid or enforceable. |
In addition, to the extent that we are unable to obtain and maintain patent protection for one of our products or product candidates or in the event that such patent protection expires or is limited to method of use patent protection, it may no longer be cost-effective to extend our portfolio by pursuing additional development of a product or product candidate for follow-on indications.
We also may rely on trade secrets to protect our technologies or products, especially where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, outside scientific collaborators and other advisers may unintentionally or willfully disclose our information to competitors. Enforcing a claim that a third-party entity illegally obtained and is using any of our trade secrets is expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the U.S. are sometimes less willing to protect trade secrets. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how.
Our indebtedness and liabilities could limit the cash flow available for our operations and expose us to risks that could adversely affect our business, financial condition and results of operations.
In November 2018 we issued $210.0 million aggregate principal amount of our Convertible Notes, and as of December 31, 2018 we had approximately $2.4 million of outstanding capital lease obligations. We may incur additional indebtedness to meet future financing needs. Our indebtedness could have significant negative consequences for our security holders and our business, results of operations and financial condition by, among other things:
35
| • | increasing our vulnerability to adverse economic and industry conditions; |
| • | limiting our ability to obtain additional financing; |
| • | requiring a substantial portion of our cash flow from operations to service our indebtedness, which will reduce the amount of cash available for other purposes; |
| • | limiting our flexibility to plan for, or react to, changes in our business; |
| • | diluting the interests of our existing stockholders as a result of issuing shares of our common stock upon conversion of the Convertible Notes; and |
| • | placing us at a possible competitive disadvantage with competitors that are less leveraged than we are or have better access to capital. |
Our ability to make scheduled payments of the principal of, to pay interest on, or to refinance our indebtedness, including the Convertible Notes, depends on our future performance, which is subject to many factors, including, economic, financial, competitive and other circumstances beyond our control. Our business may not generate sufficient funds, and we may otherwise be unable to maintain sufficient cash reserves, to pay amounts due under our indebtedness, including the Convertible Notes, and our cash needs may increase in the future. In addition, future indebtedness that we may incur may contain, financial and other restrictive covenants that limit our ability to operate our business, raise capital or make payments under our other indebtedness. If we fail to comply with these covenants or to make payments under our indebtedness when due, then we would be in default under that indebtedness, which could, in turn, result in that and our other indebtedness becoming immediately payable in full.
Our competitors may develop products that are less expensive, safer or more effective, or which may otherwise diminish or eliminate the success of any products that we may commercialize.
We may not achieve commercial success if our competitors, many of whom have significantly more resources and experience than we, market products that are safer, more effective, less expensive or faster to reach the market than any products that we may develop and commercialize. Our competitors also may market a product that proves to be unsafe or ineffective, which may affect the market for our competing product, or future product, regardless of the safety or efficacy of our product. The failure of OMIDRIA or any future product that we may market to compete effectively with products marketed by our competitors would impair our ability to generate revenue, which would have a material adverse effect on our future business, our financial condition and our results of operations.
The loss of members of our management team could substantially disrupt our business operations.
Our success depends to a significant degree on the continued individual and collective contributions of our management team. The members of our management team are at-will employees, and we do not maintain any key-person life insurance policies other than on the life of Gregory A. Demopulos, M.D., our president, chief executive officer and chairman of the board of directors. Losing the services of any key member of our management team, whether from death or disability, retirement, competing offers or other causes, without having a readily available and appropriate replacement could delay the execution of our business strategy, cause us to lose a strategic partner, or otherwise materially affect our operations.
We rely on highly skilled personnel and, if we are unable to retain or motivate key personnel or hire qualified personnel, we may not be able to maintain our operations or grow effectively.
Our performance is largely dependent on the talents and efforts of highly skilled individuals. Our future success depends on our continuing ability to identify, hire, develop, motivate and retain highly skilled personnel for all areas of our organization. If we are unable to hire and train a sufficient number of qualified employees for any reason, we may not be able to implement our current initiatives or grow effectively. We maintain a rigorous, highly selective and time-consuming hiring process. We believe that our approach to hiring has significantly contributed to our success to date. If we do not succeed in attracting qualified personnel and retaining and motivating existing personnel, our existing operations may suffer and we may be unable to grow effectively.
We may encounter difficulties managing our growth, which could delay our business plans or adversely affect our results of operations.
To manage our future growth, we must continue to implement and improve our managerial, operational and financial systems and continue to recruit and train additional qualified personnel. Due to our limited financial resources, we may not be able to manage effectively the expansion of our operations or recruit and train additional qualified personnel. The physical expansion of our operations may lead to significant costs and may divert our management and business development resources. Any inability to manage growth could delay the execution of our business plans or disrupt our operations. Additionally, our inability to manage growth effectively could cause our operating costs to grow even faster than we currently are anticipating.
36
Product liability claims may damage our reputation and, if insurance proves inadequate, these claims may harm our business.
We may be exposed to the risk of product liability claims that is inherent in the biopharmaceutical industry. A product liability claim may damage our reputation by raising questions about our product’s safety and efficacy and could limit our ability to sell one or more products by preventing or interfering with commercialization of our products and product candidates. In addition, product liability insurance for the biopharmaceutical industry is generally expensive to the extent it is available at all. There can be no assurance that we will be able to obtain or maintain such insurance on acceptable terms or that we will be able to secure and maintain increased coverage for OMIDRIA or for our product candidates, if commercialization progresses, or that future claims against us will be covered by our product liability insurance. Further, our product liability insurance coverage may not reimburse us or may be insufficient to reimburse us for any or all expenses or losses we may suffer. A successful claim against us with respect to uninsured liabilities or in excess of insurance coverage could have a material adverse effect on our business, financial condition and results of operations.
We rely on third parties to conduct portions of our preclinical research and clinical trials. If these third parties do not perform as contractually required or otherwise expected, or if we fail to adequately supervise or monitor these parties, we may not be able to obtain regulatory approval for or commercialize our product candidates.
We rely on third parties, such as CROs, medical and research institutions and clinical investigators, to conduct a portion of our preclinical research and assist us in conducting our clinical trials. Nonetheless, we are responsible for confirming that our preclinical research and clinical trials are conducted in accordance with applicable regulations, the relevant trial protocol and within the context of approvals by an institutional review board or ethics committee, and we may not always be successful in ensuring such compliance. Our reliance on these third parties does not relieve us of responsibility for ensuring compliance with FDA and other regulations and standards for conducting, monitoring, recording and reporting the results of preclinical research and clinical trials to assure that data and reported results are credible and accurate and that the trial participants are adequately protected. If these third parties do not successfully carry out their contractual duties or regulatory obligations or meet expected deadlines, if the third parties need to be replaced or if the quality or accuracy of the data they obtain is compromised due to their failure to adhere to our clinical protocols or regulatory requirements or for other reasons, our preclinical and clinical development processes may be extended, delayed, suspended or terminated, and we may not be able to commercialize or obtain regulatory approval for our product candidates.
We may need to maintain licenses for active ingredients from third parties to develop and commercialize some of our product candidates, which could increase our development costs and delay our ability to commercialize those product candidates.
Should we decide to use active pharmaceutical ingredients in any of our product candidates that are proprietary to one or more third parties, such as our PDE7 program (OMS527), we would need to maintain licenses to those active ingredients from those third parties. If we are unable to continue to access rights to these active ingredients prior to conducting preclinical toxicology studies intended to support clinical trials, we may need to develop alternate product candidates from these programs by either accessing or developing alternate active ingredients, resulting in increased development costs and delays in commercialization of these product candidates. If we are unable to maintain continued access rights to the desired active ingredients on commercially reasonable terms or develop suitable alternate active ingredients, or if we do not meet diligence or other obligations under the corresponding licenses, we may not be able to commercialize product candidates from these programs.
We use hazardous materials in our business and must comply with environmental laws and regulations, which can be expensive.
Our research operations produce hazardous waste products, which include chemicals and radioactive and biological materials. We are subject to a variety of federal, state and local regulations relating to the use, handling, storage and disposal of these materials. Although we believe that our safety procedures for handling and disposing of these materials comply with applicable legal regulations, the risk of accidental contamination or injury from these materials cannot be eliminated. We generally contract with third parties for the disposal of such substances and store our low-level radioactive waste at our facility until the materials are no longer considered radioactive. We may be required to incur further costs to comply with current or future environmental and safety regulations. In addition, although we carry insurance, in the event of accidental contamination or injury from these materials, we could be held liable for any damages that result and any such liability could exceed our insurance coverage and other resources.
Cyber-attacks or other failures in telecommunications or information technology systems could result in information theft, data corruption and significant disruption of our business operations.
37
We utilize information technology systems and networks to process, transmit and store electronic information in connection with our business activities. As use of digital technologies has increased, cyber incidents, including deliberate attacks and attempts to gain unauthorized access to computer systems and networks, have increased in frequency and sophistication. These threats pose a risk to the security of our systems and networks, the confidentiality and the availability and integrity of our data. There can be no assurance that we will be successful in preventing cyber-attacks or mitigating their effects. Similarly, there can be no assurance that our collaborators, CROs, third-party logistics providers, distributors and other contractors and consultants will be successful in protecting our clinical and other data that is stored on their systems. Any cyber-attack or destruction or loss of data could have a material adverse effect on our business and prospects. In addition, we may suffer reputational harm or face litigation or adverse regulatory action as a result of cyber-attacks or other data security breaches and may incur significant additional expense to implement further data protection measures.
Risks Related to Our Common Stock
Our stock price has been and may continue to be volatile, and the value of an investment in our common stock may decline.
During the 12-month period ended December 31, 2018, our stock traded as high as $27.00 per share and as low as $8.36 per share. The trading price of our common stock is likely to continue to be highly volatile and could be subject to wide fluctuations in response to numerous factors, many of which are beyond our control. In addition, the stock market has experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of publicly traded companies. Broad market and industry factors may seriously affect the market price of companies’ stock, including ours, regardless of actual operating performance. These fluctuations may be even more pronounced in the trading market for our stock. In addition, in the past, following periods of volatility in the overall market and the market price of a particular company’s securities, securities class action litigation has often been instituted against these companies. This litigation, if instituted against us, could result in substantial costs and a diversion of our management’s attention and resources.
If we issue additional shares of our common stock or other securities that may be convertible into, or exercisable or exchangeable for, our common stock, our existing shareholders would experience further dilution.
To the extent that we raise additional funds in the future by issuing equity securities, our shareholders would experience dilution, which may be significant and could cause the market price of our common stock to decline significantly. In addition, 10,556,253 shares of common stock were subject to outstanding options and warrants as of December 31, 2018 and may become eligible for sale in the public market to the extent permitted by the provisions of various vesting agreements. As of December 31, 2018, we also had 2,092,210 shares of common stock reserved for future issuance under our employee benefit plans that are not subject to outstanding options. Further, to the extent we issue common stock upon conversion of the Convertible Notes, such conversion would dilute the ownership interests of existing stockholders despite the expected reduction of such dilution as a result of the Capped Call Transaction (as defined below) that we entered into in connection with issuance of the Convertible Notes. If the holders of outstanding options or warrants elect to exercise some or all of them, or if the shares subject to our employee benefit plans are issued and become eligible for sale in the public market, or we issue common stock upon conversion of the Convertible Notes, our shareholders would experience dilution and the market price of our common stock could decline.
Anti-takeover provisions in our charter documents and under Washington law could make an acquisition of us, which may be beneficial to our shareholders, difficult and prevent attempts by our shareholders to replace or remove our current management.
Provisions in our articles of incorporation and bylaws and under Washington law may delay or prevent an acquisition of us or a change in our management. These provisions include a classified board of directors, a prohibition on shareholder actions by less than unanimous written consent, restrictions on the ability of shareholders to fill board vacancies and the ability of our board of directors to issue preferred stock without shareholder approval. In addition, because we are incorporated in Washington, we are governed by the provisions of Chapter 23B.19 of the Washington Business Corporation Act, which, among other things, restricts the ability of shareholders owning 10% or more of our outstanding voting stock from merging or combining with us. Although we believe these provisions collectively provide for an opportunity to receive higher bids by requiring potential acquirers to negotiate with our board of directors, they would apply even if an offer may be considered beneficial by some shareholders. In addition, these provisions may frustrate or prevent any attempts by our shareholders to replace or remove our current management by making it difficult for shareholders to replace members of our board of directors, which is responsible for appointing the members of our management.
38
We have never declared or paid dividends on our capital stock, and we do not anticipate paying dividends in the foreseeable future.
Our business requires significant funding. We currently plan to invest all available funds and future earnings, if any, in the development and growth of our business. Therefore, we currently do not anticipate paying any cash dividends on our common stock in the foreseeable future. As a result, a rise in the market price of our common stock, which is uncertain and unpredictable, will be the sole source of potential gain for shareholders in the foreseeable future, and an investment in our common stock for dividend income should not be relied upon.
39
| ITEM 1B. | UNRESOLVED STAFF COMMENTS |
None.
| ITEM 2. | PROPERTIES |
We lease approximately 108,000 square feet for our principal office and laboratory space in the building located at 201 Elliott Avenue West, Seattle, Washington, or The Omeros Building, which includes approximately 6,077 square feet of laboratory space that we are subleasing to third parties. The lease term for our space is through November 2027. We also have two options to extend the lease term, each by five years. The annual base rent due under the lease for our principal office and laboratory space is $5.4 million for 2019, $5.9 million for 2020 and $6.1 million for 2021 and will increase by approximately 2.3% each year thereafter. In addition, we are responsible for paying our proportionate share of the building’s utilities, taxes, insurance and maintenance as well as a property management fee.
We believe that our facilities are sufficient for our anticipated near-term needs.
ITEM 3. LEGAL PROCEEDINGS
From time to time, in the ordinary course of business, we may be involved in various claims, lawsuits and other proceedings. As of the date of filing of this Annual Report on Form 10-K, we were not involved in any material legal proceedings.
| ITEM 4. | MINE SAFETY DISCLOSURES |
Not applicable.
40
PART II
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED SHAREHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock is traded on The Nasdaq Global Market under the symbol “OMER.”
Holders
As of February 16, 2018, there were approximately 107 holders of record of our common stock.
Dividends
We have never declared or paid any cash dividends on our capital stock. We expect to retain all available funds and future earnings to fund the development and growth of our business and we do not anticipate paying any cash dividends in the foreseeable future.
Recent Sales of Unregistered Securities
We did not sell any equity securities during the fiscal year ended December 31, 2018 in transactions that were not registered under the Securities Act, other than as previously disclosed in our Current Reports on Form 8-K filed with the SEC on April 13, 2018 and November 15, 2018.
Stock Performance Graph
The following graph compares the cumulative total shareholder return for our common stock (OMER), the Nasdaq Biotechnology Index (NBI) and the Nasdaq U.S. Benchmark TR Index (NQUSBT) for the period beginning December 31, 2013 and ending December 31, 2018. This graph assumes that $100 was invested on December 31, 2013 in our common stock, the Nasdaq Biotechnology Index and the Nasdaq U.S. Benchmark TR Index. It also assumes that any dividends were reinvested. The data shown in the following graph are not necessarily indicative of future stock price performance.
41
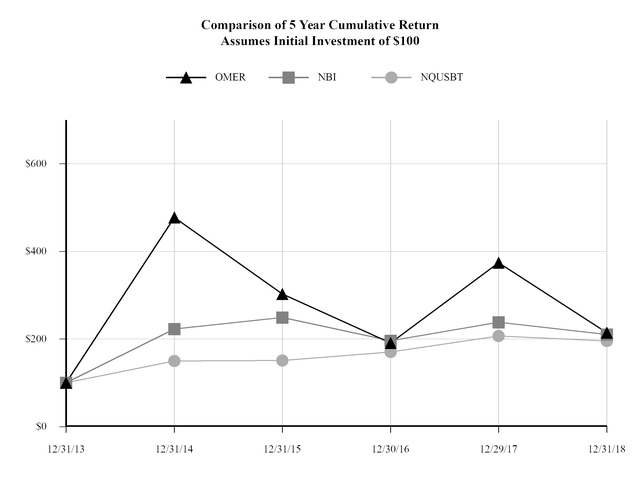
The foregoing information shall not be deemed to be “soliciting material” or to be “filed” for purposes of Section 18 of the Exchange Act or otherwise subject to liability under that Section. In addition, the foregoing information shall not be deemed to be incorporated by reference into any of our filings under the Exchange Act or the Securities Act of 1933, except to the extent that we specifically incorporate this information by reference.
42
| ITEM 6. | SELECTED CONSOLIDATED FINANCIAL DATA |
The following selected consolidated financial data should be read in conjunction with “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated financial statements and the accompanying notes included elsewhere in this Annual Report on Form 10-K. Our historical results are not necessarily indicative of the results to be expected in any future period due in part to the expiration of pass-through reimbursement for OMIDRIA effective January 1, 2018.
Year Ended December 31, | |||||||||||||||||||
| 2018 | 2017 | 2016 | 2015 | 2014 | |||||||||||||||
| (In thousands, except per share and share data) | |||||||||||||||||||
| Consolidated Statements of Operations and Comprehensive Loss Data: | |||||||||||||||||||
| Revenues: | |||||||||||||||||||
| Product sales, net | $ | 29,868 | $ | 64,826 | $ | 41,617 | $ | 13,509 | $ | 539 | |||||||||
| Costs and expenses: | |||||||||||||||||||
| Cost of product sales | 512 | 1,078 | 1,412 | 1,041 | — | ||||||||||||||
| Research and development | 89,860 | 55,599 | 50,699 | 48,379 | 47,946 | ||||||||||||||
| Selling, general and administrative | 51,718 | 52,044 | 43,782 | 35,327 | 22,601 | ||||||||||||||
| Total costs and expenses | 142,090 | 108,721 | 95,893 | 84,747 | 70,547 | ||||||||||||||
| Loss from operations | (112,222 | ) | (43,895 | ) | (54,276 | ) | (71,238 | ) | (70,008 | ) | |||||||||
| Loss on early extinguishment of debt | (12,993 | ) | — | (5,595 | ) | (1,315 | ) | — | |||||||||||
| Interest expense | (16,252 | ) | (11,030 | ) | (7,819 | ) | (3,573 | ) | (3,470 | ) | |||||||||
| Other income (expense) | 1,781 | 1,444 | 945 | 1,030 | (195 | ) | |||||||||||||
| Loss before income taxes | (139,686 | ) | (53,481 | ) | (66,745 | ) | (75,096 | ) | (73,673 | ) | |||||||||
| Income tax benefit | 12,929 | — | — | — | — | ||||||||||||||
| Net loss | $ | (126,757 | ) | $ | (53,481 | ) | $ | (66,745 | ) | $ | (75,096 | ) | $ | (73,673 | ) | ||||
| Comprehensive loss | $ | (126,757 | ) | $ | (53,481 | ) | $ | (66,745 | ) | $ | (75,096 | ) | $ | (73,673 | ) | ||||
| Basic and diluted net loss per share | $ | (2.61 | ) | $ | (1.17 | ) | $ | (1.65 | ) | $ | (2.00 | ) | $ | (2.22 | ) | ||||
| Weighted-average shares used to compute basic and diluted net loss per share | 48,582,636 | 45,539,362 | 40,446,410 | 37,560,257 | 33,234,294 | ||||||||||||||
As of December 31, | |||||||||||||||||||
| 2018 | 2017 | 2016 | 2015 | 2014 | |||||||||||||||
| (In thousands) | |||||||||||||||||||
| Consolidated Balance Sheet Data: | |||||||||||||||||||
| Cash, cash equivalents and short-term investments | $ | 60,498 | $ | 83,749 | $ | 45,331 | $ | 28,263 | $ | 6,886 | |||||||||
| Working capital (deficit) | 52,511 | 82,065 | 44,191 | 20,893 | (9,274 | ) | |||||||||||||
| Restricted cash and investments | 1,154 | 5,835 | 5,835 | 10,679 | 679 | ||||||||||||||
| Total assets | 95,936 | 116,328 | 67,278 | 48,995 | 10,834 | ||||||||||||||
| Notes payable and lease financing obligations, net | 2,467 | 84,117 | 79,512 | 49,842 | 32,453 | ||||||||||||||
| Unsecured convertible senior notes | 148,981 | — | — | — | — | ||||||||||||||
| Accumulated deficit | (650,125 | ) | (523,368 | ) | (469,887 | ) | (403,142 | ) | (328,046 | ) | |||||||||
| Total shareholders’ deficit | (100,156 | ) | (2,814 | ) | (37,447 | ) | (26,234 | ) | (42,654 | ) | |||||||||
43
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following discussion and analysis should be read in conjunction with the audited annual consolidated financial statements and the related notes thereto included elsewhere in this Annual Report on Form 10-K. This discussion contains forward-looking statements reflecting our current expectations that involve risks and uncertainties. Actual results may differ materially from those discussed in these forward-looking statements due to a number of factors, including those set forth in the section entitled “Risk Factors” and elsewhere in this Annual Report on Form 10-K. For further information regarding forward-looking statements, please refer to the special note regarding forward-looking statements at the beginning of this Annual Report on Form 10-K. Throughout this discussion, unless the context specifies or implies otherwise, the terms “Company,” “we,” “us” and “our” refer to Omeros Corporation and our wholly owned subsidiaries.
Overview
We are a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market as well as orphan indications targeting inflammation, complement-mediated diseases, disorders of the central nervous system, and immune-related diseases, including cancers.
Our drug product OMIDRIA® is marketed in the United States, or U.S., for use during cataract surgery or intraocular lens, or IOL, replacement procedures to maintain pupil size by preventing intraoperative miosis (pupil constriction) and to reduce postoperative ocular pain. In our pipeline we have clinical-stage development programs focused on: complement-associated thrombotic microangiopathies; complement-mediated glomerulonephropathies; and addictive and compulsive disorders. In addition, we have a diverse group of preclinical programs and two platforms: one capable of unlocking new G protein-coupled receptor, or GPCR, drug targets and the other used to generate antibodies. For OMIDRIA and each of our product candidates and our programs, we have retained control of all commercial rights.
Financial Summary
We recognized net losses of $126.8 million, $53.5 million, and $66.7 million for the years ended December 31, 2018, 2017 and 2016, respectively.
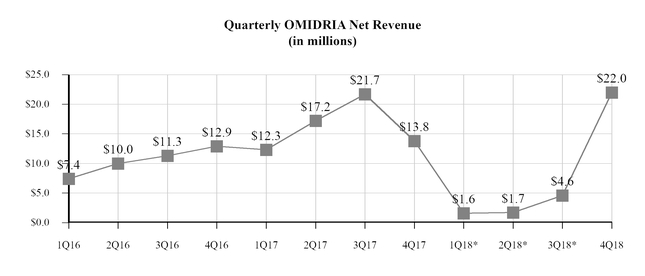
* Fiscal quarters without pass-through reimbursement.
During the years ended December 31, 2018, 2017 and 2016, OMIDRIA revenues of $29.9 million, $64.8 million, and $41.6 million respectively, helped offset a portion of our 2018, 2017 and 2016 operating expenses. During 2018 we experienced an unusual decline in revenue as compared to prior years. OMIDRIA net revenue for the three months ended December 31, 2018 was $22.0 million, compared to $7.9 million for the nine months ended September 30, 2018, during which period reimbursement for OMIDRIA was packaged with the payment rate for surgical procedures involving patients covered by Medicare Part B rather than being separately reimbursed. Separate reimbursement for OMIDRIA was restored effective October 1, 2018 as a result of securing a two-year extension of pass-through reimbursement status for use of OMIDRIA during procedures performed on Medicare Part B fee-for-service patients. See Part I, Item 1, “Business-Commercial Product- OMIDRIA” for details regarding pass-through reimbursement status for OMIDRIA.
44
We expect our net losses will continue until such time as we derive sufficient revenues from sales of OMIDRIA and/or other sources, such as licensing, product sales and other revenues from our product candidates, that are sufficient to cover our operating expenses, capital expenditures and debt service obligations.
As of December 31, 2018, we had $60.5 million in cash and cash equivalents and short-term investments available for general corporate use and $22.8 million in accounts receivable that we anticipate collecting in the first quarter of 2019.
Results of Operations
Revenue
On January 1, 2018, we adopted Accounting Standards Update (ASU) 2014-09-Revenue from Contracts with Customers (Topic 606) using the modified retrospective transition method. The adoption of ASU 2014-09 did not change the timing or the amounts of our previously recognized revenue. For more information regarding revenue recognition, see Part II, Item 8, “Note 2--Significant Accounting Policies” to our Consolidated Financial Statements in this Annual Report on Form 10-K.
Our revenue consists of OMIDRIA product sales to ambulatory surgery centers, or ASCs, and hospitals in the U.S. Our product sales, net are as follows:
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Product sales, net | $ | 29,868 | $ | 64,826 | $ | 41,617 | |||||
Pass-through status for OMIDRIA allows for separate reimbursement payment (i.e., outside the packaged procedural payment) to ASCs and hospitals using OMIDRIA in procedures involving patients covered by Medicare Part B. CMS originally granted transitional pass-through reimbursement status for OMIDRIA through December 31, 2017. During the period January 1, 2018 to September 30, 2018, OMIDRIA was not reimbursed separately when used for procedures involving patients covered by Medicare Part B.
In March 2018, the Appropriations Act was signed into law, which among other things extended pass-through reimbursement status for certain drugs, including OMIDRIA, for a two-year period beginning October 1, 2018. CMS has since implemented the Appropriations Act for dates of service beginning October 1, 2018. In the 2019 final rule for CMS’ OPPS, CMS indicated that it will separately pay in the ASC setting for non-opioid drugs used during surgery that have an FDA-approved indication for postoperative pain relief and are currently packaged in calendar year 2019. Although OMIDRIA is not specifically named because it is not currently packaged, we believe that OMIDRIA meets this definition and would qualify for separate payment under this provision if it is continued in subsequent years. The OPPS Final Rule also indicates that CMS will consider in future rule-making a policy that pays separately for drugs used during cataract surgery that have an FDA-approved indication to address postoperative issues. We believe that OMIDRIA also meets this definition. We are continuing to confirm these beliefs and to pursue other avenues of permanent separate payment or similar reimbursement for OMIDRIA beyond September 30, 2020, but can provide no assurance that these efforts will be successful.
In 2018, OMIDRIA revenue decreased $35.0 million or 53.9% as compared to the year ended December 31, 2017. The decrease in OMIDRIA revenue in 2018 was primarily attributable to the lack of separate payment for OMIDRIA under Medicare Part B from January 1, 2018 through September 30, 2018. With the reinstatement of pass-through reimbursement effective October 1, 2018, fourth quarter revenues recovered and reached a historical high of $22.0 million. Additionally, in the fourth quarter of 2018, OMIDRIA sales from our wholesalers to our ASC and hospital customers (sell-through) increased significantly as compared to the first three quarters of 2018, also reaching an all-time high.
In 2017, OMIDRIA revenue increased $23.2 million or 55.8% as compared to the year ended December 31, 2016. The increase in OMIDRIA revenue in 2017 was primarily due to the continued acceptance of OMIDRIA in the ophthalmic surgery community, as evidenced by an increase in the number of ASCs and hospitals purchasing OMIDRIA and increased penetration into existing customer accounts.
We anticipate that OMIDRIA product sales, net, will continue to increase during 2019 but we are unable to predict with precision the magnitude of those increases at the current time due to the recent relaunch of OMIDRIA.
Gross-to-Net Deductions
45
We record OMIDRIA product sales net of estimated chargebacks, rebates, distribution fees and product returns. These deductions are generally referred to as gross-to-net deductions. Our total gross-to-net provisions for the years ended December 31, 2018, 2017 and 2016 were 28.1%, 27.8% and 12.0%, respectively, of gross OMIDRIA product sales. The primary reason for the increases in gross-to-net deductions from 2016 through 2018 is due to increased rebates under our OMIDRIAssure Reimbursement Services Program and our volume-purchase discount program. We expect that 2019 gross-to-net provisions will increase moderately compared to the prior year.
Our gross-to-net provision and payments for the years ended December 31, 2018, 2017 and 2016 are summarized below:
| Chargebacks and Rebates | Distribution Fees and Product Return Allowances | Total | ||||||||||
| (In thousands) | ||||||||||||
| Balance as of December 31, 2015 | $ | 180 | $ | 277 | $ | 457 | ||||||
| Provisions | 4,203 | 1,434 | 5,637 | |||||||||
| Payments | (2,754 | ) | (1,230 | ) | (3,984 | ) | ||||||
| Balance as of December 31, 2016 | 1,629 | 481 | 2,110 | |||||||||
| Provisions | 19,188 | 5,741 | 24,929 | |||||||||
| Payments | (15,093 | ) | (2,849 | ) | (17,942 | ) | ||||||
| Balance as of December 31, 2017 | 5,724 | 3,373 | 9,097 | |||||||||
| Provisions | 10,341 | 1,309 | 11,650 | |||||||||
| Payments | (9,050 | ) | (3,197 | ) | (12,247 | ) | ||||||
| Balance as of December 31, 2018 | $ | 7,015 | $ | 1,485 | $ | 8,500 | ||||||
Chargebacks and Rebates
We record a provision for estimated chargebacks and rebates at the time we recognize OMIDRIA product sales revenue and reduce the accrual when payments are made or credits are granted. Our chargebacks are related to a Pharmaceutical Pricing Agreement, a Federal Supply Schedule agreement, a 340B prime vendor agreement and a Medicaid Drug Rebate Agreement. We also record a provision for estimated rebates for our OMIDRIAssure Reimbursement Services Program and our rebates under our purchase volume-discount programs.
Distribution Fees and Product Return Allowances
We pay our wholesalers a distribution fee for services they perform for us based on the dollar value of their purchases of OMIDRIA. We record a provision for these charges as a reduction to revenue at the time of sale to the wholesaler and make payments to our wholesalers based on contractual terms.
We allow for the return of product up to 12 months past its expiration date, or for product that is damaged or not used by our customers. We record a provision for returns upon sale of OMIDRIA to our wholesaler. When a return or claim is received, we issue a credit memo to the wholesaler against its outstanding receivable to us or we reimburse the customer.
Research and Development Expenses
Our research and development expenses can be divided into three categories: direct external expenses, which include clinical research and development and preclinical research and development activities; internal, overhead and other expenses; and stock-based compensation expense. Direct external expenses consist primarily of expenses incurred pursuant to agreements with third-party manufacturing organizations, contract research organizations, or CROs, clinical trial sites, collaborators, consultants, and lab supplies. Costs are reported in preclinical research and development until the program enters the clinic. Internal, overhead and other expenses consist of personnel costs, overhead costs such as rent, utilities and depreciation and other miscellaneous costs. We do not generally allocate our internal resources, employees and infrastructure to any individual research project because we deploy them across multiple clinical and preclinical projects that we are advancing in parallel.
46
The following table illustrates our expenses associated with these activities:
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Direct external expenses: | |||||||||||
| Clinical research and development: | |||||||||||
| MASP-2 Program - OMS721 (narsoplimab) | $ | 46,383 | $ | 19,557 | $ | 17,241 | |||||
| OMIDRIA - Ophthalmology | 2,388 | 3,458 | 3,864 | ||||||||
| PDE7 - OMS527 | 3,586 | — | — | ||||||||
| Other clinical programs | 838 | 1,714 | 500 | ||||||||
| Total clinical research and development | 53,195 | 24,729 | 21,605 | ||||||||
| Preclinical research and development | 5,627 | 4,269 | 1,731 | ||||||||
| Total direct external expenses | 58,822 | 28,998 | 23,336 | ||||||||
| Internal, overhead and other expenses | 26,077 | 21,361 | 21,059 | ||||||||
| Stock-based compensation expense | 4,961 | 5,240 | 6,304 | ||||||||
| Total research and development expenses | $ | 89,860 | $ | 55,599 | $ | 50,699 | |||||
The $29.8 million, or 102.8%, increase in direct external expenses for the year ended December 31, 2018 as compared to the year ended December 31, 2017 was due primarily to higher third-party manufacturing scale-up costs for our narsoplimab program as we continue to increase our production capacity to meet anticipated clinical and commercial requirements, higher clinical costs associated with initiating our narsoplimab IgA nephropathy Phase 3 clinical trial and the initiation, in July 2018, of our Phase 1 clinical trial for OMS527, our PDE7 program for addiction and compulsive disorders. In addition, we also incurred higher third-party preclinical development expenses as we advanced OMS527 into the clinic and continued preclinical development of our small-molecule MASP-2 inhibitors, OMS906 and our GPCR programs. These increases were partially offset by decreased costs due to completing the transfer of OMIDRIA manufacturing to a new facility in December 2017.
The $5.7 million, or 24.3%, increase in direct external expenses for the year ended December 31, 2017 as compared to the year ended December 31, 2016 was due primarily to higher third-party manufacturing scale-up costs for our narsoplimab program and to incremental clinical costs as our narsoplimab clinical trials advanced. In addition, higher third-party development expenses were incurred as we continued to advance our preclinical product candidates including OMS527, OMS906 and small-molecule MASP-2 inhibitors toward the clinic. These increases were partially offset by the completion of a post-marketing OMIDRIA pediatric trial in 2016 and decreased manufacturing costs associated with the product validation of OMIDRIA at a new commercial manufacturing facility.
The increases in internal, overhead and other expenses in all years presented are primarily due to increased employee-related costs to support our increased research and development activities.
The changes in stock-based compensation in all years presented are primarily due to timing of stock grants.
During 2019, the majority of our research and development expenses will be related to narsoplimab. We expect research and development costs to increase in 2019 as we continue our ongoing Phase 3 clinical programs and incur manufacturing scale-up costs and other expenses as we continue preparations for our anticipated submission of marketing applications and potential commercialization of narsoplimab in HSCT-TMA in the U.S. and Europe.
At this time, we are unable to estimate with any certainty the longer-term costs we will incur in the continued development of our product candidates due to the inherently unpredictable nature of our preclinical and clinical development activities. Clinical development timelines, the probability of success and development costs can differ materially as new data become available and as expectations change. Our future research and development expenses will depend, in part, on the preclinical or clinical success of each product candidate as well as ongoing assessments of each program’s commercial potential. In addition, we cannot forecast with precision which product candidates, if any, may be subject to future collaborations, when such arrangements will be secured, if at all, and to what degree such arrangements would affect our development plans and capital requirements.
We are required to expend substantial resources in the development of our product candidates due to the lengthy process of completing clinical trials and seeking regulatory approval. Any failure or delay in completing clinical trials, or in obtaining regulatory approvals, could delay our generation of product revenue and increase our research and development expenses,
47
which, in turn, could have a material adverse effect on our results of operations, financial condition and liquidity. Because of the factors above, we are not able to estimate with any certainty when or if we would recognize any net cash inflows from our research and development projects.
Selling, General and Administrative Expenses
Our selling, general and administrative, or SG&A, expenses are comprised primarily of salaries, benefits and stock-compensation costs for sales, marketing and other personnel not directly engaged in research and development, marketing and selling expenses, professional and legal services, general corporate costs and an allocation of our occupancy costs.
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Selling, general and administrative expenses, excluding stock-based compensation expense | $ | 44,966 | $ | 44,596 | $ | 36,504 | |||||
| Stock-based compensation expense | 6,752 | 7,448 | 7,278 | ||||||||
| Total selling, general and administrative expenses | $ | 51,718 | $ | 52,044 | $ | 43,782 | |||||
The increase in selling, general and administrative expenses, excluding stock-based compensation during the year ended December 31, 2018 as compared to the prior year was primarily due to increased fees related to patent applications, consulting and professional service fees, and headcount-related costs, partially offset by reduced legal costs associated with pursuing patent infringement claims against generic drug manufacturers that sought to obtain FDA approval for a generic version of OMIDRIA, which were favorably resolved. See Part I, Item 1, “Business--License and Development Agreements” for more information regarding the successful conclusion of our patent infringement lawsuits. The change in stock-based compensation is primarily due to timing of stock grants and related changes in value of our grants.
The increase in selling, general and administrative expenses, excluding stock-based compensation during the year ended December 31, 2017 compared to 2016 was primarily due to increased legal costs associated with pursuing patent infringement claims against generic drug manufacturers that sought to obtain FDA approval for a generic version of OMIDRIA and, to a lesser degree, increased employee costs. See Part I, Item 1, “Business--License and Development Agreements” for more information regarding the successful conclusion of our patent infringement lawsuits.
We expect that our selling, general and administrative expenses for 2019 will increase from 2018, primarily due to preparations for potential commercialization of narsoplimab in HSCT-TMA as well as incremental headcount additions supporting OMIDRIA sales and pre-commercial activities for narsoplimab.
Interest Expense
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Interest Expense | $ | 16,252 | $ | 11,030 | $ | 7,819 | |||||
The increase in interest expense over the years 2018, 2017 and 2016 is primarily due to the increase in our outstanding notes payable balance under our former term loan agreement with CRG Servicing LLC, which we refer to as the CRG Loan, and with other lenders during the comparative periods. The November 2018 issuance of $210.0 million aggregate principal amount of our 6.25% Convertible Senior Notes due 2023, or the Convertible Notes, also contributed to the 2018 increase. For more information regarding our notes payable and our Convertible Notes, see Part II, Item 8, “Note 7--Notes Payable” and “Note 8--Convertible Senior Notes” to our Consolidated Financial Statements in this Annual Report on Form 10-K.
We expect interest expense will increase in 2019. Interest expense for the first quarter of 2019 is expected to be approximately $5.5 million, of which $2.2 million is anticipated to be non-cash interest related to the amortization of the conversion feature and debt issuance costs associated with our Convertible Notes.
48
Loss on Early Extinguishment of Debt
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Loss on early extinguishment of debt | $ | 12,993 | $ | — | $ | 5,595 | |||||
In November 2018, we issued Convertible Notes and repaid all amounts outstanding under the CRG Loan.
We incurred a loss on early extinguishment of debt of $13.0 million associated with the unamortized lender facility fee, debt issuance costs, debt discount and prepayment fees in connection with the repayment of the CRG Loan.
In November 2016, we entered into the CRG Loan and repaid our then-outstanding loan under our loan and security agreement with other lenders. We incurred a loss of $5.6 million associated with the unamortized loan maturity fee and the prepayment fee related to the then-outstanding loans.
Other Income
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Other Income | $ | 1,781 | $ | 1,444 | $ | 945 | |||||
Other income principally includes sublease rental income and interest earned. The increase during the year ended December 31, 2018 is primarily due to increased interest income offset by non-cash fair value adjustment to our Capped Call Transaction (as defined below). Sublease rental remained consistent during the year ended December 31, 2018 compared to 2017.
The increase during the year ended December 31, 2017 was due to incremental subleased space as compared to 2016.
Financial Condition - Liquidity and Capital Resources
As of December 31, 2018, we had $60.5 million in cash, cash equivalents and short-term investments available for general corporate use held primarily in money-market accounts as compared to $83.7 million at December 31, 2017. In addition, as of December 31, 2018 we had $22.8 million in accounts receivable that we anticipate collecting in the first quarter of 2019.
In November 2018, we issued $210.0 million aggregate principal amount of Convertible Notes. We used approximately$146.0 million of the proceeds to repay the CRG Loan, including associated prepayment fees and accrued interest, and $33.2 million to enter into the Capped Call Transaction described below, resulting in net proceeds available for corporate use, after deducting the initial purchasers’ discounts and commissions and our offering expenses, of $24.0 million. For more information regarding the Convertible Notes, see Part II, Item 8, “Note 8--Convertible Senior Notes” to our Consolidated Financial Statements in this Annual Report on Form 10-K.
49
We have historically generated net losses and incurred negative cash flows. For the years ended December 31, 2018, 2017, and 2016 we incurred net losses of $126.8 million, $53.5 million and $66.7 million, respectively, and also incurred negative cash flows from operations of $103.7 million, $36.2 million and $51.5 million in 2018, 2017 and 2016, respectively.
As described earlier in this section under “Results of Operations - Revenue,” pass-through status for OMIDRIA allows for separate reimbursement payment (i.e., outside the packaged procedural payment) to ASCs and hospitals using OMIDRIA in procedures involving patients covered by Medicare Part B. CMS originally granted transitional pass-through reimbursement for OMIDRIA through December 31, 2017. OMIDRIA was not subject to pass-through reimbursement during the first nine months of the year 2018 and, therefore, was not reimbursed separately from the packaged payment rate for procedures involving Medicare Part B patients. In March 2018, the Appropriations Act was signed into law, which extended pass-through reimbursement for certain drugs, including OMIDRIA, for a two-year period beginning October 1, 2018.
We continue to advance a series of clinical and preclinical programs (including three programs currently in Phase 3). As described above, on October 1, 2018, OMIDRIA pass-through reimbursement was reinstated for a two-year period and OMIDRIA net sales increased significantly to $22.0 million in the fourth quarter of 2018. Prior to the lapse in pass-through reimbursement, OMIDRIA revenue was substantially offsetting our operating expenses and debt service costs. We believe OMIDRIA sales will continue to grow in 2019; however, due to the recent re-introduction of OMIDRIA, we cannot predict with precision the extent of growth in OMIDRIA revenues in 2019. As a result, despite the significant quarterly revenue growth in OMIDRIA sales prior to the loss of OMIDRIA pass-through reimbursement, meaningful growth in OMIDRIA sales in 2019 and beyond, are not included in the going concern evaluation. In addition, we are not able to include an accounts receivable-based line of credit even though we received initial approval from a lending institution supporting such an instrument. Similarly, we are unable to include in the evaluation any cash inflows from potential partnerships, debt or other financing instruments, despite our successful track record in accessing capital through each of these avenues. The conditions described above when evaluated within the constraints of the accounting literature raise substantial doubt with respect to our ability to meet our obligations through March 1, 2020 and, therefore, to continue as a going concern.
We plan to continue to fund our operations through proceeds from sales of OMIDRIA and, in addition, we have received initial approval for an accounts receivable-based line of credit, which would allow us to borrow up to $50 million based on our available accounts-receivable borrowing base. Should it be necessary or determined to be strategically advantageous, we also could pursue debt financings, public and private offerings of our equity securities similar to those we have completed previously, and/or other strategic transactions, which may include licensing a portion of our existing technology. If these capital sources, for any reason, are needed but inaccessible, it would have a significantly negative effect on our financial condition. Should it be necessary to manage our operating expenses, we would reduce our projected cash requirements through reduction of our expenses by delaying clinical trials, reducing selected research and development efforts, and/or implementing other restructuring activities.
The accompanying consolidated financial statements have been prepared on a going-concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The accompanying consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from uncertainty related to our ability to continue as a going concern.
Cash Flow Data
| Year ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Cash provided by (used in): | |||||||||||
| Operating activities | $ | (103,737 | ) | $ | (36,227 | ) | $ | (51,504 | ) | ||
| Investing activities | 25,151 | (37,598 | ) | (16,335 | ) | ||||||
| Financing activities | 81,053 | 74,995 | 68,698 | ||||||||
Operating Activities
Net cash used in operating activities increased for the year ended December 31, 2018 by $67.5 million as compared to the same period in 2017. The increase largely resulted from the $73.3 million increase in our net loss from 2017 due to a decrease in OMIDRIA product sales of $35.0 million and an increase in research and development expense of $34.3 million. In addition, increases in changes in operating assets and liabilities of $4.1 million and non-cash charges of $1.3 million in 2018 when compared to 2017 positively impacted the change in our cash used in operating activities.
50
Net cash used in operating activities decreased for the year ended December 31, 2017 by $15.3 million as compared to the same period in 2016. The reduction in cash used in operating activities in 2017 largely resulted from the $13.3 million decline in our net loss from 2016 due primarily to an increase in OMIDRIA product sales of $23.2 million, which was partially offset by a $12.8 million increase in total costs and expenses. In addition, in 2017, non-cash charges decreased by $4.0 million when compared to 2016, which negatively impacted the change in our cash used in operating activities. A $6.0 million net change in operating assets and liabilities between 2017 and 2016 also positively affected the change in our cash used in operations.
Investing Activities. Cash flows from investing activities primarily reflect cash used to purchase short-term investments and proceeds from the sale and maturity of short-term investments, thus causing a shift between our cash and cash equivalents and short-term investment balances. Because we manage our cash usage with respect to our cash, cash equivalents and short-term investments on an aggregated basis, we do not consider the fluctuations between cash, cash equivalents and our short-term investment balances to be important to the understanding of our liquidity and capital resources. The remaining component of cash flows from investing activities is the purchase of property and equipment.
Net cash provided by investing activities in the year ended December 31, 2018 was $25.2 million, an increase of $62.7 million from the $37.6 million used in investing activities in 2017. The cash provided by investing activities in 2018 was primarily due to proceeds from the sale of short-term investments for $94.5 million, which were partially offset by the sale and maturity of $68.8 million of short-term investments.
Net cash used in investing activities in the year ended December 31, 2017 was $37.6 million, an increase of $21.3 million from 2016, primarily due to the purchase of short-term investments for $65.3 million with the $63.7 million of net proceeds received from the sale of common stock in our August 2017 public offering. These purchases were partially offset by the sale and maturity of $28.1 million of short-term investments to provide cash for operating activities.
Financing Activities. Net cash provided by financing activities in the year ended December 31, 2018 was $81.1 million, a net increase of $6.1 million over the same period in 2017, as follows:
| • | $24.0 million of net proceeds available for corporate use from the issuance of Convertible Notes, net of the $146.0 million used to repay our then outstanding CRG Loan, $33.2 million used to enter into the Capped Call Transaction and $6.8 million of debt issuance costs; |
| • | $44.6 million in net proceeds from borrowings under our now extinguished CRG Loan without a similar borrowing in 2017; |
•$6.7 million in proceeds from exercises of stock options compared to $11.8 million in 2017;
•$63.7 million in proceeds from issuance of common stock in 2017 without a similar issuance in 2018; and
•$4.7 million in funds made available in 2018 due to a decrease in restricted investments.
Net cash provided by financing activities in the year ended December 31, 2017 was $75.0 million, an increase of $6.3 million over the prior year. In 2017, we received $63.7 million of net proceeds from the sale of common stock in our public offering in August compared to $38.0 million of net proceeds from the sale of common stock and pre-funded warrants in our August 2016 public offering. In 2016, we also received $22.8 million in net additional borrowings under our then-existing loan agreements and did not have any similar borrowings in 2017. During 2017, we also received $11.8 million from the exercise of employee stock options and warrants as compared to $3.2 million in 2016.
Convertible Notes
On November 15, 2018, we issued at face value $210.0 million aggregate principal amount of 6.25% Convertible Senior Notes due 2023, or the Convertible Notes. The Convertible Notes are due in November 2023 unless earlier repurchased, redeemed or converted in accordance with their terms. We received net proceeds available for corporate use of $24.0 million, as summarized below:
| (In thousands) | |||
| Convertible Notes | $ | 210,000 | |
| Repayment of the CRG Loan | (146,046 | ) | |
| Purchase of Capped Call | (33,180 | ) | |
| Issuance costs | (6,800 | ) | |
| Net proceeds available for corporate use | $ | 23,974 | |
51
The Convertible Notes accrue interest at an annual rate of 6.25% per annum, payable semi-annually in arrears on May 15 and November 15 of each year, beginning on May 15, 2019. The Convertible Notes are unsecured obligations.
The Convertible Notes will be convertible into cash, shares of our common stock or a combination thereof, as we elect at our sole discretion. The initial conversion rate is 52.0183 shares of our common stock per $1,000 principal amount of Convertible Notes (equivalent to an initial conversion price of approximately $19.22 per share of common stock), subject to adjustment in certain circumstances.
The Convertible Notes will be convertible, at the option of the holders on or after May 15, 2023 at any time prior to the close of business on the second scheduled trading day immediately preceding the stated maturity date of November 15, 2023. Additionally, holders may convert their Convertible Notes at their option at specified times prior to the maturity date of November 15, 2023, only if:
(1) during any calendar quarter beginning after March 31, 2019, the last reported sale price per share of our common stock exceeds 130% of the conversion price of the Convertible Notes for each of at least 20 trading days in the period of 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter;
(2) during the five consecutive business days immediately after any five-consecutive-trading-day period (such five- consecutive-trading-day period, the “measurement period”) in which the trading price per $1,000 principal amount of Convertible Notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day;
(3) upon the occurrence of certain corporate events or distributions of our common stock; or
(4) we call the Convertible Notes for redemption.
Subject to the satisfaction of certain conditions, we may redeem all, but not less than all, of the Convertible Notes at our option beginning November 15, 2019 through the maturity date at a cash redemption price equal to the principal amount of the Convertible Notes to be redeemed, plus any accrued and unpaid interest. The Convertible Notes are subject to redemption only if certain requirements are satisfied, including that the last reported sale price per share of our common stock exceeds 150% of the conversion price on (i) each of at least 20 trading days, whether or not consecutive, during the 30 consecutive trading days ending on, and including, the trading day immediately before the date we send the related redemption notice and (ii) the trading day immediately before the date we send such notice.
In order to reduce the dilutive impact or potential cash expenditure associated with conversion of the Convertible Notes, we used $33.2 million of the proceeds from sale of the Convertible Notes to enter into a capped call transaction, or the Capped Call Transaction. The Capped Call Transaction will cover, subject to anti-dilution adjustments substantially similar to those applicable to the Convertible Notes, the number of shares of our common stock underlying the Convertible Notes when our common stock is trading between the initial conversion price of approximately $19.22 and $28.8360. However, if the market price of our common stock exceeds the $28.8360 cap price, then the conversion of the Convertible Notes would have a dilutive impact and/or require a cash expenditure to the extent the market price exceeds the cap price. For more information regarding the Notes, see Part II, Item 8, “Note 8--Convertible Senior Notes” to our Consolidated Financial Statements in this Annual Report on Form 10-K.
Contractual Obligations and Commitments
The following table presents a summary of our contractual obligations and commitments as of December 31, 2018.
Payments Due Within | |||||||||||||||||||
1 Year | 2-3 Years | 4-5 Years | More than 5 Years | Total | |||||||||||||||
| (In thousands) | |||||||||||||||||||
| Operating leases | $ | 5,361 | $ | 11,987 | $ | 12,551 | $ | 22,798 | $ | 52,697 | |||||||||
| Capital leases (principal and interest) | 1,174 | 1,536 | 347 | — | 3,057 | ||||||||||||||
| Unsecured convertible senior notes | 13,198 | 26,396 | 236,396 | — | 275,990 | ||||||||||||||
| Goods & services | $ | 11,283 | $ | 339 | $ | — | $ | — | $ | 11,622 | |||||||||
| Total | $ | 31,016 | $ | 40,258 | $ | 249,294 | $ | 22,798 | $ | 343,366 | |||||||||
52
Operating Leases
We lease our office and laboratory space in The Omeros Building under a lease agreement with BMR - 201 Elliott Avenue LLC. The initial term of the lease ends in November 2027 and we have two options to extend the lease term, each by five years. We lease office and laboratory equipment under various operating and capital lease agreements with initial terms of five years or less.
As of December 31, 2018, the remaining aggregate non-cancelable rent payable under the initial term of the lease, excluding common area maintenance and related operating expenses, is $52.7 million.
Convertible Notes
Refer to “Financial Condition - Liquidity and Capital Resources - Convertible Notes” above.
Goods & Services
We have certain non-cancelable obligations under other agreements for the acquisitions of goods and services associated with the manufacturing of our product candidates, which contain firm commitments. As of December 31, 2018, our aggregate firm commitments are $11.6 million.
We may be required, in connection with in-licensing or asset acquisition agreements, to make certain royalty and milestone payments and we cannot, at this time, determine when or if the related milestones will be achieved or whether the events triggering the commencement of payment obligations will occur. Therefore, such payments are not included in the table above. For information regarding agreements that include these royalty and milestone payment obligations, see Part II, Item 8, “Note 10--Commitments and Contingencies” to our Consolidated Financial Statements in this Annual Report on Form 10-K.
Critical Accounting Policies and Significant Judgments and Estimates
The preparation of our consolidated financial statements, in conformity with U.S. generally accepted accounting principles, or GAAP, requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances; however, actual results could differ from those estimates. An accounting policy is considered critical if it is important to a company’s financial condition and results of operations and if it requires the exercise of significant judgment and the use of estimates on the part of management in its application. Although we believe that our judgments and estimates are appropriate, actual results may differ materially from our estimates.
We believe the following to be our critical accounting policies because they are both important to the portrayal of our financial condition and results of operations and they require critical judgment by management and estimates about matters that are uncertain:
| • | revenue recognition; |
| • | research and development expenses, primarily clinical trial expenses and manufacturing of drug product and clinical drug supply; and |
| • | stock-based compensation. |
If actual results or events differ materially from those contemplated by us in making these estimates, our reported financial condition and results of operations for future periods could be materially affected.
Revenue Recognition
On January 1, 2018, we adopted Accounting Standards Update (ASU) 2014-09-Revenue from Contracts with Customers (Topic 606) using the modified retrospective transition method. Upon adoption, we evaluated our existing contracts with customers and determined the adoption of the standard did not change the timing or the amounts of our previously recognized revenues.
When we enter into a customer contract, we perform the following five steps: (i) identify the contract with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy a performance obligation.
Chargebacks and Rebates: Provisions for chargebacks are determined utilizing historical and projected payer mix and information regarding sell-through and inventory on-hand received directly from wholesalers. Chargebacks are generally settled within four weeks of recording product sales revenue.
53
We provide reimbursement support services and financial assistance in the form of a rebate to patients whose commercial insurance is inadequate to cover the full cost of OMIDRIA. We apply an experience ratio to product sales to determine the rebate accrual. This experience ratio is reviewed and updated periodically to reflect actual results.
We provide rebate payments for which ASCs qualify by meeting or exceeding purchase volumes of OMIDRIA under our purchase volume-discount program. We calculate rebate payment amounts due under this program based on actual qualifying purchase volumes and apply a contractual discount rate. For purchases of OMIDRIA not yet reported as sold-through to the ASC by our wholesalers, we apply an experience ratio to product sales to determine the rebate accrual. This experience ratio is being reviewed and updated periodically to reflect actual results.
Distribution Fees and Product Return Allowances: We pay our wholesalers a distribution fee for services that they perform for us based on the wholesaler average cost value of their purchases of OMIDRIA. We record a provision against product sales for these charges at the time of sale to the wholesaler.
We allow for the return of product up to 12 months past its expiration date or for product that is damaged. In estimating product returns, we take into consideration our return experience to date, the remaining shelf-life of product we have previously sold, inventory in the wholesale channel and our expectation that product is typically not held by the healthcare providers based on the frequency of their reorders.
Research and Development Expenses
Research and development costs are comprised primarily of costs for personnel, including salaries, benefits and stock compensation; an allocation of our occupancy costs; clinical study costs; contracted research; manufacturing; consulting arrangements; depreciation; materials and supplies; milestones; and other expenses incurred to sustain our overall research and development programs. Clinical trial expenses for investigational sites require certain estimates. We estimate these costs based on a cost per patient that varies depending on the clinical trial site. As actual costs become known to us, we adjust our estimates; these changes in estimates may result in understated or overstated expenses at any given point in time. Research and development costs are expensed as incurred.
Advanced payments for goods or services that will be used or rendered for future research and development activities are deferred and then recognized as an expense as the related goods are delivered or as the services are performed, or when the goods or services are no longer expected to be provided.
Stock-Based Compensation
Stock-based compensation expense is recognized for all share-based payments made to employees, directors and non-employees based on estimated fair values. The fair value of our stock options is calculated using the Black-Scholes option valuation model, which requires assumptions, including volatility, forfeiture rates and expected option life. If any of the assumptions used in the Black-Scholes model change significantly, stock-based compensation expense for new awards may differ materially from that recorded for existing awards and stock-based compensation for non-employees will vary as the awards are re-measured over the vesting term.
As stock-based compensation expense is based on options ultimately expected to vest, the expense has been reduced for estimated forfeitures. We estimate forfeitures for expense recognition based on our historical experience. Groups of employees that have similar historical forfeiture behavior are considered separately. We use the straight-line method to allocate compensation cost to reporting periods over each optionee’s respective requisite service period for employees and directors, which is generally the vesting period.
Stock options granted to non-employees are accounted for using the fair-value approach using the Black-Scholes option-pricing model and are re-measured over the vesting term as earned. The estimated fair value is charged to expense over the applicable service period.
Recent Accounting Pronouncements
Please refer to Note 2 in our Consolidated Financial Statements for information regarding recent accounting pronouncements.
Off-Balance Sheet Arrangements
We have not engaged in any off-balance sheet arrangements.
54
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Our exposure to market risk is primarily confined to our investment securities and notes payable. The primary objective of our investment activities is to preserve our capital to fund operations, and we do not enter into financial instruments for trading or speculative purposes. We also seek to maximize income from our investments without assuming significant risk. To achieve our objectives, we maintain a portfolio of investments in high-credit-quality securities. As of December 31, 2018, we had cash, cash equivalents and short-term investments of $60.5 million. In accordance with our investment policy, we invest funds in highly liquid, investment-grade securities. The securities in our investment portfolio are not leveraged and are classified as available-for-sale. We currently do not hedge interest rate exposure. Because of the short-term maturities of our investments, we do not believe that an increase in market rates would have a material negative effect on the realized value of our investment portfolio. We actively monitor changes in interest rates and, with our current portfolio of short-term investments, we are not exposed to potential loss due to changes in interest rates.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
Index to Consolidated Financial Statements
55
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors
Omeros Corporation
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Omeros Corporation (the Company) as of December 31, 2018 and 2017, the related consolidated statements of operations and comprehensive loss, shareholders' deficit and cash flows for each of the three years in the period ended December 31, 2018, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the consolidated financial position of the Company at December 31, 2018 and 2017, and the consolidated results of its operations and its cash flows for each of the three years in the period ended December 31, 2018, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company's internal control over financial reporting as of December 31, 2018, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated March 1, 2019 expressed an unqualified opinion thereon.
The Company's Ability to Continue as a Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has suffered losses from operations and has stated that substantial doubt exists about the Company’s ability to continue as a going concern. Management's evaluation of the events and conditions and management’s plans regarding these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 1998.
Seattle, Washington
March 1, 2019
56
OMEROS CORPORATION
CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share data)
| December 31, | |||||||
| 2018 | 2017 | ||||||
| Assets | |||||||
| Current assets: | |||||||
| Cash and cash equivalents | $ | 5,861 | $ | 3,394 | |||
| Short-term investments | 54,637 | 80,355 | |||||
| Receivables, net | 22,818 | 17,144 | |||||
| Inventory | 88 | 443 | |||||
| Prepaid expense and other assets | 6,463 | 7,036 | |||||
| Total current assets | 89,867 | 108,372 | |||||
| Property and equipment, net | 3,845 | 2,121 | |||||
| Restricted investments | 1,154 | 5,835 | |||||
| Advanced payments, non-current | 1,070 | — | |||||
| Total assets | $ | 95,936 | $ | 116,328 | |||
| Liabilities and shareholders’ deficit | |||||||
| Current liabilities: | |||||||
| Accounts payable | $ | 6,281 | $ | 6,691 | |||
| Accrued expenses | 30,186 | 19,126 | |||||
| Current portion of lease financing obligations | 889 | 490 | |||||
| Total current liabilities | 37,356 | 26,307 | |||||
| Lease obligation, net of current portion | 1,578 | 810 | |||||
| Notes payable | — | 83,307 | |||||
| Unsecured convertible senior notes | 148,981 | — | |||||
| Deferred rent | 8,177 | 8,718 | |||||
| Commitments and contingencies (Note 10) | |||||||
| Shareholders’ equity (deficit) | |||||||
| Preferred stock, par value $0.01 per share, 20,000,000 shares authorized and none issued at December 31, 2018 and 2017. | — | — | |||||
| Common stock, par value $0.01 per share, 150,000,000 shares authorized at December 31, 2018 and 2017; 49,011,684 and 48,211,226 issued and outstanding at December 31, 2018 and 2017, respectively. | 490 | 482 | |||||
| Additional paid-in capital | 549,479 | 520,072 | |||||
| Accumulated deficit | (650,125 | ) | (523,368 | ) | |||
| Total shareholders’ deficit | (100,156 | ) | (2,814 | ) | |||
| Total liabilities and shareholders’ deficit | $ | 95,936 | $ | 116,328 | |||
See accompanying Notes to Consolidated Financial Statements
57
OMEROS CORPORATION
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(In thousands, except share and per share data)
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| Revenues: | |||||||||||
| Product sales, net | $ | 29,868 | $ | 64,826 | $ | 41,617 | |||||
| Costs and expenses: | |||||||||||
| Cost of product sales | 512 | 1,078 | 1,412 | ||||||||
| Research and development | 89,860 | 55,599 | 50,699 | ||||||||
| Selling, general and administrative | 51,718 | 52,044 | 43,782 | ||||||||
| Total costs and expenses | 142,090 | 108,721 | 95,893 | ||||||||
| Loss from operations | (112,222 | ) | (43,895 | ) | (54,276 | ) | |||||
| Loss on early extinguishment of debt | (12,993 | ) | — | (5,595 | ) | ||||||
| Interest expense | (16,252 | ) | (11,030 | ) | (7,819 | ) | |||||
| Other income | 1,781 | 1,444 | 945 | ||||||||
| Loss before income taxes | (139,686 | ) | (53,481 | ) | (66,745 | ) | |||||
| Income tax benefit | 12,929 | — | — | ||||||||
| Net loss | $ | (126,757 | ) | $ | (53,481 | ) | $ | (66,745 | ) | ||
| Comprehensive loss | $ | (126,757 | ) | $ | (53,481 | ) | $ | (66,745 | ) | ||
| Basic and diluted net loss per share | $ | (2.61 | ) | $ | (1.17 | ) | $ | (1.65 | ) | ||
| Weighted-average shares used to compute basic and diluted net loss per share | 48,582,636 | 45,539,362 | 40,446,410 | ||||||||
See accompanying Notes to Consolidated Financial Statements
58
OMEROS CORPORATION
CONSOLIDATED STATEMENTS OF SHAREHOLDERS’ DEFICIT
(In thousands, except share data)
| Common Stock | Additional Paid-in Capital | Accumulated Deficit | Total Shareholders’ Deficit | |||||||||||||||
| Shares | Amount | |||||||||||||||||
| Balance at December 31, 2015 | 38,040,891 | 380 | 376,528 | (403,142 | ) | (26,234 | ) | |||||||||||
| Issuance of common stock in direct offering, net of offering costs | 3,478,260 | 35 | 37,279 | — | 37,314 | |||||||||||||
| Issuance of common stock upon exercise of stock options | 1,486,167 | 15 | 3,131 | — | 3,146 | |||||||||||||
| Warrant exercises | 749,250 | 7 | — | — | 7 | |||||||||||||
| Issuance of common stock in connection with the ATM, net of offering costs | 64,565 | 1 | 724 | — | 725 | |||||||||||||
| Stock-based compensation | — | — | 13,582 | — | 13,582 | |||||||||||||
| Fair value of warrants in connection with amendment to notes payable | — | — | 758 | — | 758 | |||||||||||||
| Net loss | — | — | — | (66,745 | ) | (66,745 | ) | |||||||||||
| Balance at December 31, 2016 | 43,819,133 | 438 | 432,002 | (469,887 | ) | (37,447 | ) | |||||||||||
| Issuance of common stock in direct offering, net of offering costs | 3,000,000 | 30 | 63,627 | — | 63,657 | |||||||||||||
| Issuance of common stock upon exercise of stock options | 1,392,093 | 14 | 11,755 | — | 11,769 | |||||||||||||
| Stock-based compensation | — | — | 12,688 | — | 12,688 | |||||||||||||
| Net loss | — | — | — | (53,481 | ) | (53,481 | ) | |||||||||||
| Balance at December 31, 2017 | 48,211,226 | 482 | 520,072 | (523,368 | ) | (2,814 | ) | |||||||||||
| Issuance of common stock upon exercise of stock options | 800,458 | 8 | 6,724 | — | 6,732 | |||||||||||||
| Issuance of warrants in connection with amendment of notes payable | — | — | 1,424 | — | 1,424 | |||||||||||||
| Stock-based compensation | — | — | 11,713 | — | 11,713 | |||||||||||||
| Premium paid for Capped Call in connection with Convertible Notes | — | — | (33,180 | ) | — | (33,180 | ) | |||||||||||
| Equity component of issuance of Convertible Notes | — | — | 55,655 | — | 55,655 | |||||||||||||
| Tax benefit related to equity component of Convertible Notes | — | — | (12,929 | ) | — | (12,929 | ) | |||||||||||
| Net loss | — | — | — | (126,757 | ) | (126,757 | ) | |||||||||||
| Balance at December 31, 2018 | 49,011,684 | $ | 490 | $ | 549,479 | $ | (650,125 | ) | $ | (100,156 | ) | |||||||
See accompanying Notes to Consolidated Financial Statements
59
OMEROS CORPORATION
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| Operating activities: | |||||||||||
| Net loss | $ | (126,757 | ) | $ | (53,481 | ) | $ | (66,745 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | |||||||||||
| Stock-based compensation expense | 11,713 | 12,688 | 13,582 | ||||||||
| Non-cash interest expense | 5,635 | 4,187 | 1,977 | ||||||||
| Depreciation and amortization | 962 | 551 | 300 | ||||||||
| Loss on early extinguishment of debt | 12,993 | — | 5,595 | ||||||||
| Deferred income tax | (12,929 | ) | — | — | |||||||
| Fair value adjustment to convertible notes in equity | 354 | — | — | ||||||||
| Changes in operating assets and liabilities: | |||||||||||
| Receivables | (5,674 | ) | (5,107 | ) | (5,520 | ) | |||||
| Inventory | 355 | 685 | (656 | ) | |||||||
| Prepaid expenses and other assets | (498 | ) | (5,270 | ) | 347 | ||||||
| Accounts payable and accrued expenses | 10,544 | 9,616 | (421 | ) | |||||||
| Deferred rent | (435 | ) | (96 | ) | 37 | ||||||
| Net cash used in operating activities | (103,737 | ) | (36,227 | ) | (51,504 | ) | |||||
| Investing activities: | |||||||||||
| Purchases of property and equipment | (567 | ) | (350 | ) | (126 | ) | |||||
| Purchases of investments | (68,782 | ) | (65,326 | ) | (73,966 | ) | |||||
| Proceeds from the sale and maturities of investments | 94,500 | 28,078 | 57,757 | ||||||||
| Net cash provided by (used in) investing activities | 25,151 | (37,598 | ) | (16,335 | ) | ||||||
| Financing activities: | |||||||||||
| Proceeds from issuance of convertible senior notes | 210,000 | — | — | ||||||||
| Proceeds from borrowings under notes payable | 44,550 | — | 100,000 | ||||||||
| Proceeds upon exercise of stock options and warrants | 6,732 | 11,769 | 3,153 | ||||||||
| Proceeds from issuance of common stock, net | — | 63,657 | 38,039 | ||||||||
| Release in restricted investments | 4,681 | — | 4,844 | ||||||||
| Repayment of notes payable | (132,077 | ) | — | (70,137 | ) | ||||||
| Premium paid for capped call option | (33,180 | ) | — | — | |||||||
| Payments on debt prepayment and extinguishment | (11,902 | ) | — | (5,700) | |||||||
| Payments for debt issuance costs | (6,800 | ) | — | (1,501 | ) | ||||||
| Payments on lease financing obligations | (951 | ) | (431 | ) | |||||||
| Net cash provided by financing activities | 81,053 | 74,995 | 68,698 | ||||||||
| Net increase in cash and cash equivalents | 2,467 | 1,170 | 859 | ||||||||
| Cash and cash equivalents at beginning of period | 3,394 | 2,224 | 1,365 | ||||||||
| Cash and cash equivalents at end of period | $ | 5,861 | $ | 3,394 | $ | 2,224 | |||||
| Supplemental cash flow information | |||||||||||
| Cash paid for interest | $ | 8,896 | $ | 6,895 | $ | 5,293 | |||||
| Conversion of accrued interest to notes payable | $ | 3,408 | $ | 3,315 | $ | 516 | |||||
| Issuance of warrants in connection with amendments to notes payable | $ | 1,424 | $ | — | $ | 758 | |||||
| Property acquired under capital lease | $ | 2,118 | $ | 1,141 | $ | 404 | |||||
60
See accompanying Notes to Consolidated Financial Statements
61
OMEROS CORPORATION
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Note 1—Organization and Basis of Presentation
Organization
We are a commercial-stage biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market as well as orphan indications targeting inflammation, complement-mediated diseases, disorders of the central nervous system, and immune-related diseases, including cancers. Our first drug product, OMIDRIA, is marketed in the United States (U.S.) for use during cataract surgery or intraocular lens replacement.
Basis of Presentation
Our consolidated financial statements include the financial position and results of operations of Omeros Corporation (Omeros) and our wholly owned subsidiaries. All inter-company transactions have been eliminated. The accompanying consolidated financial statements have been prepared in accordance with U.S. generally accepted accounting principles (GAAP). Certain prior year amounts in the balance sheet, statement of cash flows and the footnotes have been reclassified in the consolidated financial statements to conform to the current year presentation.
As of December 31, 2018, we had cash, cash equivalents and investments of $60.5 million and net working capital of $52.5 million. We have incurred losses from operations of $112.2 million and $43.9 million in 2018 and 2017, respectively, used cash in operating activities of $103.7 million and $36.2 million in 2018 and 2017, respectively and anticipate that we will continue to incur losses until such time as revenues exceed operating costs.
We continue to advance a series of clinical and preclinical programs (including three programs currently in Phase 3). CMS granted transitional pass-through reimbursement status for OMIDRIA from January 1, 2015 through December 31, 2017 for patients covered by Medicare Part B. On October 1, 2018, OMIDRIA pass-through reimbursement, as a result of Congressional legislation, was reinstated for a two-year period and quarterly OMIDRIA net sales returned to exceed historical levels. See “Note 15--Quarterly Information (Unaudited)” for additional details. Prior to the lapse in pass-through reimbursement, OMIDRIA revenue was substantially offsetting our operating expenses and debt service costs. We believe OMIDRIA sales will continue to grow in 2019; however, due to the recent re-introduction of OMIDRIA, we cannot predict with precision the extent of growth in OMIDRIA revenues in 2019. As a result, despite our significant historical growth in OMIDRIA sales, meaningful growth in OMIDRIA sales in 2019 and beyond are not included in the determination regarding our prospects as a going concern, nor are we able to include an accounts receivable-based line of credit even though we received initial credit approval from a lending institution supporting such an instrument. Similarly, we are unable to include in the determination any revenues from potential partnerships, debt or other financing instruments, despite our successful track record in accessing capital through each of these avenues. The conditions described above, when evaluated within the constraints of the accounting literature, raise substantial doubt with respect to our ability to meet our obligations through March 1, 2020 and, therefore, to continue as a going concern.
We plan to continue to fund our operations through proceeds from sales of OMIDRIA and, if necessary, through other revenue sources and financial instruments as noted above. If these capital sources, for any reason, are needed but inaccessible, it would have a significantly negative effect our financial condition. Should it be necessary to manage our operating expenses, we would reduce our projected cash requirements through reduction of our expenses by delaying clinical trials, reducing selected research and development efforts, and/or implementing other restructuring activities.
The accompanying consolidated financial statements have been prepared on a going-concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The accompanying consolidated financial statements do not include any adjustments to reflect the possible future effects on the recoverability and classification of assets or the amounts and classification of liabilities that may result from uncertainty related to our ability to continue as a going concern.
Segments
We operate in one segment. Management uses cash flow as the primary measure to manage our business and does not segment our business for internal reporting or decision-making.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Significant items subject to such estimates include revenue recognition, stock-based compensation expense and accruals for clinical trials, manufacturing of drug product and clinical drug supply and contingencies. We base our estimates on historical experience and on various other factors that we believe are reasonable under the circumstances; however, actual results could differ from these estimates.
62
Note 2—Significant Accounting Policies
Cash and Cash Equivalents, Short-Term Investments and Restricted Investments
Cash and cash equivalents include highly liquid investments with a maturity of three months or less on the date of purchase. Short-term investment securities are classified as available-for-sale and are carried at fair value. Unrealized gains and losses, if any, are reported as a separate component of shareholders’ deficit. Amortization, accretion, interest and dividends, realized gains and losses and declines in value judged to be other-than-temporary are included in other income. The cost of securities sold is based on the specific-identification method. Investments in securities with maturities of less than one year, or those for which management intends to use the investments to fund current operations, are included in current assets. We evaluate whether an investment is other-than-temporarily impaired based on the specific facts and circumstances. Factors that are considered in determining whether an other-than-temporary decline in value has occurred include: the market value of the security in relation to its cost basis; the financial condition of the investee; and the intent and ability to retain the investment for a sufficient period of time to allow for recovery in the market value of the investment. Restricted cash and investments are held in certificates of deposit and money-market funds.
As of December 31, 2018 and 2017, all investments are classified as short-term and available-for-sale on the accompanying Consolidated Balance Sheets. Investment income, which is included as a component of other income, consists primarily of interest earned.
Inventory
Inventory is stated at the lower of cost or market determined on a specific identification basis in a manner that approximates the first-in, first-out (FIFO) method. Costs include amounts related to third-party manufacturing, transportation and internal labor and overhead. Capitalization of costs as inventory begins when the product candidate receives regulatory approval in the U.S. or the European Union (EU). We expense inventory costs related to product candidates as research and development expenses prior to receiving regulatory approval in the respective territory. Inventory is reduced to net realizable value for excess and obsolete inventories based on forecasted demand.
Receivables, Net
Receivables relate primarily to sales of OMIDRIA to wholesalers and include reductions for estimated chargebacks and product returns that are expected to be settled through reductions in receivables. Remaining receivables consist of amounts from subleases for space in The Omeros Building. Considering the nature and historic collectability of our receivables, we concluded an allowance for doubtful accounts is not necessary as of December 31, 2018 and 2017.
Property and Equipment, Net
Property and equipment are stated at cost, and depreciation is calculated using the straight-line method over the estimated useful life of the assets, which is generally three to 10 years. Equipment financed under capital leases is recorded as property and equipment and is amortized over the shorter of the useful lives of the related assets or the lease term. Expenditures for repairs and maintenance are expensed as incurred.
Impairment of Long-Lived Assets
We assess the impairment of long-lived assets, primarily property and equipment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. Recoverability of these assets is measured by comparing the carrying value to future undiscounted cash flows that the asset is expected to generate. If the asset is considered to be impaired, the amount of any impairment will be reflected in the results of operations in the period of impairment. We have not recognized any impairment losses for the years ending December 31, 2018, 2017 and 2016.
Deferred Rent
We recognize rent expense on a straight-line basis over the noncancelable term of The Omeros Building operating lease and, accordingly, record the difference between cash rent payments and the recognition of rent expense as an increase or decrease in deferred rent liability. We also record landlord-funded lease incentives, such as reimbursable leasehold improvements, as an increase in deferred rent liability, which is amortized as a reduction of rent expense over the noncancelable terms of The Omeros Building operating lease.
On January 1, 2019, we adopted ASU 2016-02 related to lease accounting, which requires lessees to record assets and liabilities on the balance sheet for all leases with terms greater than 12 months. Upon adoption, the net lease asset was adjusted by the reversal of the outstanding balance of our deferred rent. (see Recent Accounting Pronouncements below for further discussion of ASU 2016-02 adoption).
63
Revenue Recognition
On January 1, 2018, we adopted Accounting Standards Update (ASU) 2014-09-Revenue from Contracts with Customers (Topic 606) using the modified retrospective transition method. Upon adoption, we evaluated our existing contracts with customers and determined that the adoption of the standard did not change the timing or the amounts of our previously recognized revenues.
When we enter into a customer contract, we perform the following five steps: (i) identify the contract with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) we satisfy a performance obligation.
Product Sales, Net
We generally record revenue from product sales when the product is delivered to our wholesalers. Product sales are recorded net of wholesaler distribution fees and estimated chargebacks, rebates, returns and purchase-volume discounts. Accruals or allowances are established for these deductions in the same period when revenue is recognized, and actual amounts incurred are offset against the applicable accruals or allowances. We reflect each of these accruals or allowances as either a reduction in the related accounts receivable or as an accrued liability depending on how the amount is expected to be settled.
Research and Development
Research and development costs are comprised primarily of costs for personnel, including salaries, benefits and stock compensation; clinical study costs; contracted research; manufacturing; consulting arrangements; depreciation; materials and supplies; milestones; an allocation of our occupancy costs; and other expenses incurred to sustain our overall research and development programs. Research and development costs are expensed as incurred.
Advance payments for goods or services that will be used or rendered for future research and development activities are deferred and then recognized as an expense as the related goods are delivered or the services are performed, or when the goods or services are no longer expected to be provided.
Patents
We generally apply for patent protection on processes and product candidates we or our licensors conceive or develop. Patent costs are comprised primarily of external legal fees, filing fees incurred to file patent applications, and periodic renewal fees to keep the patent in force and are expensed as incurred as a component of general and administrative expense.
Selling, General and Administrative
Selling, general and administrative (SG&A) expenses are comprised primarily of salaries, benefits, and stock-compensation costs for sales, marketing, and other personnel not directly engaged in research and development. Additionally, SG&A includes marketing and selling expenses, professional and legal services; patent costs; depreciation, an allocation of our occupancy costs; and other general corporate expenses. Advertising costs, which we consider to be media and marketing materials, are expensed as incurred and were $2.5 million, $328,000 and $672,000 during the years ended December 31, 2018, 2017 and 2016, respectively.
Income Taxes
Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their tax bases. Deferred tax assets and liabilities are measured using enacted tax rates applied to taxable income in the years in which those temporary differences are expected to be recovered or settled. We recognize the effect of income tax positions only if those positions are more likely than not of being sustained upon an examination. A valuation allowance is established when it is more likely than not that the deferred tax assets will not be realized.
Stock-Based Compensation
Stock-based compensation expense is recognized for all share-based payments made to employees, directors and non-employees based on estimated fair values. The fair value of our stock options is calculated using the Black-Scholes option-pricing model which requires judgmental assumptions including volatility, forfeiture rates and expected option life. For employees and directors, we use the straight-line method to allocate compensation cost to reporting periods over each optionee’s requisite service period, which is generally the vesting period. Stock options granted to non-employees are accounted for using the fair-value approach and are subject to periodic revaluation over their vesting terms as earned. The stock compensation cost related to non-employee stock options is based on changes in estimated fair value and is charged to expense over the applicable service period.
64
Accumulated Other Comprehensive Loss
Accumulated other comprehensive loss is comprised of net loss and certain changes in equity that are excluded from net loss. There was no difference between comprehensive loss and net loss for the years ended December 31, 2018, 2017 or 2016.
Financial Instruments and Concentrations of Credit Risk
Cash and cash equivalents, receivables, accounts payable and accrued liabilities, which are recorded at invoiced amount or cost, approximate fair value based on the short-term nature of these financial instruments. The fair value of short-term investments is based on quoted market prices. Financial instruments that potentially subject us to concentrations of credit risk consist primarily of cash and cash equivalents, short-term investments and receivables. Cash and cash equivalents are held by financial institutions and are federally insured up to certain limits. At times, our cash and cash equivalents balance held at a financial institution may exceeds the federally insured limits. To limit the credit risk, we invest our excess cash in high-quality securities such as money market mutual funds, certificates of deposit and commercial paper.
Major Customers
We sell OMIDRIA through a limited number of wholesalers. Each of these wholesalers, together with entities under their common control, accounted for greater than 10% of total revenues for the years ended December 31, 2018, 2017 and 2016 and greater than 10% of accounts receivable as of December 31, 2018 and 2017 as noted below.
| 2018 | 2017 | 2016 | |||||||||||||
| Percentage of Total Revenue | Percentage of Accounts Receivable | Percentage of Total Revenue | Percentage of Accounts Receivable | Percentage of Total Revenue | |||||||||||
| Distributor A | 31 | % | 27 | % | 29 | % | 31 | % | 32 | % | |||||
| Distributor B | 27 | % | 25 | % | 26 | % | 23 | % | 31 | % | |||||
| Distributor C | 22 | % | 25 | % | 22 | % | 26 | % | 28 | % | |||||
| Distributor D | 20 | % | 23 | % | 23 | % | 20 | % | * | ||||||
* Distributor did not account for greater than 10% of total revenues for the year ended December 31, 2016.
Major Suppliers
We use a single contract manufacturer to supply the OMIDRIA drug substance and a separate company to perform final packaging of OMIDRIA for commercial sale. We generally use one to two contract manufacturers to produce clinical trial material for each of our clinical trials, which creates a concentration of risk for us.
We endeavor to maintain reasonable levels of drug supply for our commercial and clinical trial use and other manufacturers are available should we need to change suppliers. A change in suppliers, however, could cause a delay in delivery of OMIDRIA or our clinical trial material that would adversely affect our business.
Recently Adopted Accounting Pronouncements
On January 1, 2018, we adopted Accounting Standards Update (ASU) 2014-09-Revenue from Contracts with Customers (Topic 606) using the modified retrospective transition method. Upon adoption, we evaluated our existing contracts with customers and determined the adoption of the standard did not change the timing or the amounts of our previously recognized revenues.
On January 1, 2018, we adopted ASU 2017-09 related to stock-based compensation, which provides guidance on the accounting for changes to the terms and conditions of stock-based payment arrangements. The adoption of the standard did not have a material impact on our stock-based compensation expense.
Recent Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02 amending the accounting for leases. This new standard requires lessees to recognize an asset and a lease liability for most leases. Presentation of leases within the consolidated statements of operations and consolidated statements of cash flows will be generally consistent with the current lease accounting guidance. The ASU is effective for reporting periods beginning after December 15, 2018. We adopted this ASU on January 1, 2019 using a modified retrospective transition method rather than retrospectively adjusting prior periods. This adoption approach will result in a balance sheet presentation that will not be comparable to the prior period. As described further in Note 9 - Lease Obligations, the adoption will lead to an increase in the assets and liabilities recorded on our Condensed Consolidated Balance Sheets due to the lease agreements for our office building.
65
In June 2018, the FASB issued ASU 2018-07 that simplifies the accounting for share-based payments granted to non-employees for services by generally aligning it with the accounting for share-based payments to employees. We adopted the standard on January 1, 2019 and do not expect the adoption of this standard to have a material impact on our condensed consolidated financial statements.
In August 2018, the FASB issued ASU 2018-15 related to the accounting for cloud computing arrangements to follow the internal-use software guidance in determining which implementation costs to defer and recognize as an asset. The guidance is applied either retrospectively or prospectively to all implementation costs incurred after the dated of adoption. We expect to adopt the standard January 1, 2020 on a prospective basis.
Note 3—Net Loss Per Share
Basic net loss per share is calculated by dividing the net loss by the weighted-average number of common shares outstanding for the period. Diluted net loss per share is computed by dividing the net loss by the weighted-average number of common shares and dilutive common share equivalents outstanding for the period, determined using the treasury-stock method. Common share equivalents are excluded from the diluted net loss per share computation if their effect is anti-dilutive.
The basic and diluted net loss per share amounts for the years ended December 31, 2018, 2017 and 2016 were computed based on the shares of common stock outstanding during the respective periods. Potentially dilutive securities excluded from the diluted loss per share calculation are as follows:
| Year Ended December 31, | ||||||||
| 2018 | 2017 | 2016 | ||||||
| Outstanding options to purchase common stock | 10,313,138 | 9,657,259 | 9,809,374 | |||||
| Warrants to purchase common stock | 243,115 | 100,602 | 100,602 | |||||
| Total potentially dilutive securities | 10,556,253 | 9,757,861 | 9,909,976 | |||||
Note 4—Accounts Receivable, Net
Accounts receivable, net consists of the following:
| December 31, | |||||||
| 2018 | 2017 | ||||||
| (In thousands) | |||||||
| Trade receivables, net | $ | 22,654 | $ | 17,079 | |||
| Subleases and other receivables | 164 | 65 | |||||
| Accounts receivables, net | $ | 22,818 | $ | 17,144 | |||
Trade receivables are shown net of $439,000 and $198,000 of chargeback and product return allowances as of December 31, 2018 and 2017, respectively.
66
Note 5—Fair-Value Measurements
As of December 31, 2018 and 2017, all investments were classified as short-term and available-for-sale on the accompanying Consolidated Balance Sheets. Investment income, which was included as a component of other income, consists of interest earned.
On a recurring basis, we measure certain financial assets at fair value. Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability, an exit price, in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The accounting standard establishes a fair value hierarchy that requires an entity to maximize the use of observable inputs, where available. The following summarizes the three levels of inputs required:
Level 1—Observable inputs for identical assets or liabilities, such as quoted prices in active markets;
Level 2—Inputs other than quoted prices in active markets that are either directly or indirectly observable; and
Level 3—Unobservable inputs in which little or no market data exists, therefore they are developed using estimates and assumptions developed by us, which reflect those that a market participant would use.
Our fair-value hierarchy for our financial assets measured at fair value on a recurring basis are as follows:
| December 31, 2018 | |||||||||||||||
| Level 1 | Level 2 | Level 3 | Total | ||||||||||||
| (In thousands) | |||||||||||||||
| Assets: | |||||||||||||||
| Money-market funds classified as non-current restricted cash and investments | $ | 1,154 | $ | — | $ | — | $ | 1,154 | |||||||
| Money-market funds classified as short-term investments | 54,637 | — | — | 54,637 | |||||||||||
| Total | $ | 55,791 | $ | — | $ | — | $ | 55,791 | |||||||
| December 31, 2017 | |||||||||||||||
| Level 1 | Level 2 | Level 3 | Total | ||||||||||||
| (In thousands) | |||||||||||||||
| Assets: | |||||||||||||||
| Money-market funds classified as non-current restricted cash and investments | $ | 5,835 | $ | — | $ | — | $ | 5,835 | |||||||
| Money-market funds classified as short-term investments | 80,355 | — | — | 80,355 | |||||||||||
| Total | $ | 86,190 | $ | — | $ | — | $ | 86,190 | |||||||
Cash held in demand deposit accounts of $5.9 million and $3.4 million is excluded from our fair-value hierarchy disclosure as of December 31, 2018 and 2017, respectively. There were no unrealized gains or losses associated with our short-term investments as of December 31, 2018 or 2017. The carrying amounts reported in the accompanying Consolidated Balance Sheets for receivables, accounts payable, other current monetary assets and liabilities, and notes payable and lease financing obligations approximate fair value.
See “Note 8--Convertible Senior Notes” for the carrying amount and estimated fair value of our 6.25% Convertible Senior Notes due 2023.
Note 6—Certain Balance Sheet Accounts
Property and Equipment, Net
Property and equipment, net consists of the following:
67
| December 31, | |||||||
| 2018 | 2017 | ||||||
| (In thousands) | |||||||
| Capital lease equipment | $ | 4,034 | $ | 1,915 | |||
| Laboratory equipment | 2,569 | 2,180 | |||||
| Computer equipment | 862 | 684 | |||||
| Office equipment and furniture | 625 | 625 | |||||
| Total cost | 8,090 | 5,404 | |||||
| Less accumulated depreciation and amortization | (4,245 | ) | (3,283 | ) | |||
| Total property and equipment, net | $ | 3,845 | $ | 2,121 | |||
For the years ended December 31, 2018, 2017 and 2016, depreciation and amortization expenses were $962,000, $551,000 and $300,000, respectively.
Accrued Expenses
Accrued expenses consist of the following:
| December 31, | |||||||
| 2018 | 2017 | ||||||
| (In thousands) | |||||||
| Contract research and development | $ | 12,012 | $ | 4,251 | |||
| Sales rebates, fees and discounts | 8,075 | 6,561 | |||||
| Employee compensation | 2,714 | 2,309 | |||||
| Consulting and professional fees | 3,669 | 1,627 | |||||
| Interest payable | 1,677 | — | |||||
| Clinical accrual | 820 | 1,026 | |||||
| ASC/Hospital return liability | — | 2,350 | |||||
| Other accruals | 1,219 | 1,002 | |||||
| Total accrued liabilities | $ | 30,186 | $ | 19,126 | |||
Note 7—Notes Payable
2015 Oxford/EWB Loan
In December 2015, we entered into a Loan and Security Agreement (the Oxford/EWB Loan) with Oxford Finance, LLC (Oxford) and East West Bank (EWB) pursuant to which we borrowed $50.0 million. In May 2016, we amended the Oxford/EWB Loan (the Amendment) and borrowed an additional $20.0 million and received $19.9 million in net proceeds. In connection with the Amendment, we issued warrants to purchase an aggregate of 100,602 shares of Omeros common stock (the Warrants) to Oxford and EWB at the then current market price of $9.94 per share. We accounted for the Amendment as debt modification and for the Warrants as a discount to our notes payable. See “Note 11--Shareholders’ Equity” for further discussion of the Warrants.
2016 CRG Loan Agreement
In October 2016, we entered into the CRG Loan with CRG Servicing LLC (CRG), as administrative and collateral agent, and the lenders identified therein and, in November 2016, borrowed $80.0 million. We used $75.7 million of the loan proceeds to repay all amounts we owed under our Oxford/EWB Loan. After deducting the loan initiation costs and related fees on the CRG Loan, we received $3.0 million in net proceeds. We accounted for the termination of the Oxford/EWB Loan as a debt extinguishment and, accordingly, incurred a $5.6 million loss associated with the unamortized loan maturity fee, loan initiation costs and prepayment fees. In May 2018, we borrowed the remaining $45.0 million available under the CRG Loan and issued warrants to purchase up to 200,000 shares of our common stock with an exercise price of $23.00 per share and total fair value of $1.4 million. The warrants have a five-year term and remained outstanding as of December 31, 2018.
68
The CRG Loan was secured by substantially all our assets including our intellectual property and required certain minimum net revenue amounts or a minimum market capitalization threshold as well as certain restricted cash balances to be maintained.
Interest accrued on the CRG Loan at an annual rate of 12.25% (4.00% of which we could defer by adding such amount to the aggregate principal amount outstanding). During the year ended December 31, 2018, 2017 and 2016, we deferred $3.2 million, $3.3 million and $0.5 million of interest.
The CRG Loan required us to pay a back-end lender facility fee equal to 5.00% of the aggregate principal amount borrowed and, upon early repayment, we were subject to a 4.0% prepayment fee.
In November 2018, we issued unsecured convertible senior notes (see “Note 8--Convertible Senior Notes”) and repaid the CRG Loan. We incurred a loss on early extinguishment of debt of $13.0 million associated with the unamortized lender facility fee, debt issuance costs, debt discount and prepayment fees upon repayment of the CRG Loan.
Note 8— Convertible Senior Notes
On November 15, 2018, we issued at face value $210.0 million aggregate principal amount 6.25% Convertible Senior Notes due 2023 (the Convertible Notes) and received net proceeds of $24.0 million, as summarized below:
| (In thousands) | |||
| Convertible Notes initially issued | $ | 210,000 | |
| Repayment of the CRG Loan | (146,046 | ) | |
| Purchase of Capped Call | (33,180 | ) | |
| Issuance costs | (6,800 | ) | |
| Net proceeds available for corporate use | $ | 23,974 | |
The Convertible Notes accrue interest at an annual rate of 6.25% per annum, payable semi-annually in arrears on May 15 and November 15 of each year, beginning on May 15, 2019. The Convertible Notes mature on November 15, 2023, unless earlier repurchased, redeemed or converted in accordance with their terms. The Convertible Notes are unsecured obligations.
The Convertible Notes will be convertible into cash, shares of our common stock or a combination thereof, as we elect at our sole discretion. The initial conversion rate is 52.0183 shares of our common stock per $1,000 of note principal (equivalent to an initial conversion price of approximately $19.22 per share of common stock), subject to adjustment in certain circumstances.
The Convertible Notes will be convertible at the option of the holders on or after May 15, 2023 at any time prior to the close of business on the second scheduled trading day immediately preceding the stated maturity date of November 15, 2023. Additionally, holders may convert their Convertible Notes at their option at specified times prior to the maturity date of November 15, 2023, only if:
(1) during any calendar quarter beginning after March 31, 2019, the last reported sale price per share of our common stock exceeds 130% of the conversion price of the Convertible Notes for each of at least 20 trading days in the period of 30 consecutive trading days ending on, and including, the last trading day of the immediately preceding calendar quarter;
(2) during the five consecutive business days immediately after any five-consecutive-trading-day period (such five-consecutive-trading-day period, the “measurement period”) in which the trading price per $1,000 principal amount of Convertible Notes for each trading day of the measurement period was less than 98% of the product of the last reported sale price per share of our common stock on such trading day and the conversion rate on such trading day;
(3) upon the occurrence of certain corporate events or distributions of our common stock; or
(4) we call the Convertible Notes for redemption.
Subject to the satisfaction of certain conditions, we may redeem all, but not less than all, of the Convertible Notes at our option beginning November 15, 2019 through the maturity date at a cash redemption price equal to the principal amount of the Convertible Notes to be redeemed, plus any accrued and unpaid interest. The Convertible Notes are subject to redemption only if certain requirements are satisfied, including that the last reported sale price per share of our common stock exceeds 150% of the conversion price on (i) each of at least 20 trading days, whether or not consecutive, during the 30 consecutive trading days ending on, and including, the trading day immediately before the date we send the related redemption notice and (ii) the trading day immediately before the date we send such notice.
69
In order to reduce the dilutive impact or potential cash expenditure associated with conversion of the Convertible Notes, we used $33.2 million of the proceeds from sale of the Convertible Notes to enter into a capped call transaction (the Capped Call Transaction). The Capped Call Transaction will cover, subject to anti-dilution adjustments substantially similar to those applicable to the Convertible Notes, the number of shares of our common stock underlying the Convertible Notes when our common stock is trading between the initial conversion price of approximately $19.22 and $28.8360. However, if the market price of our common stock exceeds the $28.8360 cap price, then the conversion of the Convertible Notes would have a dilutive impact and/or require a cash expenditure to the extent the market price exceeds the cap price.
We evaluated the accounting for the issuance of the Convertible Notes and concluded that the embedded conversion features meet the requirements for a derivative scope exception for instruments that are both indexed to an entity’s own stock and classified in stockholders’ equity in its balance sheet, and that the cash conversion guidance applies. Therefore, the Convertible Notes issuance proceeds of $210.0 million are allocated first to the liability component based on the fair value of non-convertible debt with otherwise identical residual terms with the residual proceeds allocated to equity for the conversion features. The debt issuance costs associated with the Convertible Notes of $6.8 million are allocated to the liability and equity component in the same proportion as the issuance proceeds. We also evaluated the interest feature on default and redemption features embedded in the Convertible Notes and concluded that they are clearly and closely related to the Convertible Notes, and therefore they would not be separately accounted for as a derivative. For the contingent interest features unrelated to our creditworthiness, we concluded that they should be separately accounted for as a compound derivative instrument with de-minimis value. Further, we concluded the Capped Call Transaction qualifies for a derivative scope exception for instruments that are both indexed to an entity’s own stock and classified in stockholders’ equity in its balance sheet. Consequently, the fair value of the Capped Call of $33.2 million is classified as equity and will not be subsequently remeasured.
The balance of our Convertible Notes at December 31, 2018, is as follows:
| (In thousands) | ||||
| Principal amount | $ | 210,000 | ||
| Unamortized debt discount | (56,156 | ) | ||
| Unamortized debt issuance costs attributable to principal amount | (4,863 | ) | ||
| Net carrying amount of Convertible Notes | $ | 148,981 | ||
| Fair value of outstanding Convertible Notes | $ | 186,900 | ||
| Amount by which the Convertible Notes if-converted value exceeds their principal amount | $ | — | ||
The unamortized debt discount and debt issuance costs related to the Convertible Notes are being amortized to interest expense using the effective interest method through the scheduled maturity of November 15, 2023.
The estimated fair value of the Convertible Notes at December 31, 2018, as determined through consideration of quoted market prices, was $186.9 million. The fair value is classified as Level 3 due to the limited trading activity for the Convertible Notes.
Note 9—Lease Obligations
Operating Lease Obligations
We lease office and laboratory space in The Omeros Building. The initial term of the lease is through November 2027 and we have two options to extend the lease term, each by five years. As of December 31, 2018, the remaining aggregate non-cancelable rent under the initial terms of the lease, excluding common area maintenance and related operating expenses, was $52.7 million. The deferred rent balance of $8.7 million relates to rent deferrals and landlord funded lease incentives and is being amortized to research and development and selling, general and administrative expense on a straight-line basis through the initial term of the lease.
Rent expense, including the amortization of lease incentives and rent deferrals, totaled $4.1 million for the year ended December 31, 2018, and $4.4 million for both years ended December 31, 2017 and 2016, respectively. We sublease unused space in The Omeros Building to third-party tenants. Rental income received under these subleases was $848,000, $886,000 and $737,000 for the years ended December 31, 2018, 2017 and 2016, respectively. Rental income is recorded as other income in the accompanying Consolidated Statements of Operations and Comprehensive Loss.
As of December 31, 2018, our other remaining non-cancelable rental payments are immaterial.
70
Capital Lease Financing Obligations
We have capital leases for certain laboratory and office equipment that have lease terms expiring through December 2021. Equipment costs related to these capital leases of $4.0 million and $1.9 million are included in our property and equipment as of December 31, 2018 and December 31, 2017, respectively and the accumulated depreciation on this equipment was $1.2 million and $530,000, respectively. The remaining principal payments under these capital leases totaled $2.4 million as of December 31, 2018.
Future Minimum Lease Payments
Future minimum lease payments as of December 31, 2018 under our operating leases and capital equipment financing leases, based on stated contractual maturities, are as follows:
| Year Ending December 31, | The Omeros Building Lease | Building Sublease Income | Operating Lease, Net | Capital Leases | Total | |||||||||||||||
| (in thousands) | ||||||||||||||||||||
| 2019 | $ | 5,361 | $ | (558 | ) | $ | 4,803 | $ | 1,174 | $ | 5,977 | |||||||||
| 2020 | 5,926 | — | 5,926 | 955 | 6,881 | |||||||||||||||
| 2021 | 6,061 | — | 6,061 | 581 | 6,642 | |||||||||||||||
| 2022 | 6,203 | — | 6,203 | 252 | 6,455 | |||||||||||||||
| 2023 | 6,348 | — | 6,348 | 95 | 6,443 | |||||||||||||||
| Thereafter | 22,798 | — | 22,798 | — | 22,798 | |||||||||||||||
| Total | $ | 52,697 | $ | (558 | ) | $ | 52,139 | $ | 3,057 | $ | 55,196 | |||||||||
New Lease Accounting Standard
In February 2016, the FASB issued ASU 2016-02 related to lease accounting which requires lessees to record assets and liabilities on the balance sheet for all leases with terms greater than 12 months. Leases will be classified as either finance or operating, with classification affecting the pattern of expense recognition in the income statement.
We adopted the new standard as of January 1, 2019 and elected the package of practical expedients permitted under the transition guidance, which allows us to carryforward the historical lease classification. We also elected the hindsight practical expedient to determine the reasonably certain lease term for existing leases. Our election of the hindsight practical expedient did not alter the lease terms for any of our existing leases. We also made an accounting policy election to not record leases with an initial term of 12 months or less on our balance sheet and will continue to recognize those lease payments in the Consolidated Statements of Operations on a straight-line basis over the lease term.
We estimate adoption of the standard will result in recognition of additional net lease assets and lease liabilities of approximately $15.0 million to $21.0 million and $23.0 million to $29.0 million, respectively, in our Consolidated Balance Sheets as of January 1, 2019. The balance of the net lease asset will also be adjusted by the reversal of the outstanding balance of deferred rent. We do not believe the standard will materially affect our consolidated net earnings or have a notable impact on our liquidity.
Note 10—Commitments and Contingencies
Contracts
We have various agreements with third parties that collectively require payment of termination fees totaling $11.6 million as of December 31, 2018 if we cancel the work within specific time frames, either prior to commencing or during performance of the contracted services.
Development Milestones and Product Royalties
We have licensed a variety of intellectual property from third parties that we are currently developing or may develop in the future. These licenses may require milestone payments during the clinical development processes as well as low single to low double-digit royalties on the net income or net sales of the product. For the years ended December 31, 2018, 2017 and 2016, development milestones incurred were immaterial and we did not owe any royalties.
71
Note 11—Shareholders’ Equity
Common Stock
As of December 31, 2018, we had reserved shares of common stock for the following purposes:
| Options granted and outstanding | 10,313,138 | |
| Options available for future grant | 2,092,210 | |
| Common stock warrants | 243,115 | |
| Total shares reserved | 12,648,463 | |
Securities Offerings - In August 2017, we sold 3.0 million shares of our common stock at a public offering price of $22.75 per share. After deducting underwriter discounts and offering expenses, we received net proceeds from the transaction of $63.6 million.
In August 2016, we sold 3.5 million shares of our common stock at a public offering price of $11.50 per share. After deducting underwriter discounts and offering expenses, we received net proceeds from the offering of $37.3 million.
At Market Issuance Sales Agreement - In 2016, under an at market issuance sales agreement, we sold 64,565 shares of our common stock at an average price of $11.41 and received net proceeds of $724,000.
Warrants
The following table summarizes our outstanding warrants at December 31, 2018:
| Outstanding At December 31, 2018 | Expiration Date | Exercise Price | ||
| 43,115 | May 18, 2023 | $9.94 | ||
| 200,000 | April 12, 2023 | $23.00 | ||
In connection with the May 2016 Amendment to the Oxford/EWB Loan, we issued warrants to purchase up to 100,602 shares of our common stock at an exercise price per share of $9.94 per share and total fair value of $758,000. In September 2018, warrant holders with a right to purchase 57,487 shares of our common stock initiated a cash-less exercise of their warrants which resulted in the issuance of 34,509 shares or our common stock. As of December 31, 2018, 43,115 of these warrants remain outstanding. The warrants are exercisable through May 18, 2023.
In connection with the April 2018 amendment to the CRG Loan, we issued warrants to purchase up to 200,000 shares of our common stock with an exercise price of $23.00 per share and total fair value of $1.4 million. The warrants have a five-year term.
See “Note 8 - Convertible Senior Notes” for discussion of our outstanding Convertible Notes.
Note 12—Stock-Based Compensation
On June 16, 2017, our shareholders approved the Omeros Corporation 2017 Omnibus Incentive Compensation Plan (the 2017 Plan), which provides for the grant of incentive and non-statutory stock options, stock appreciation rights, restricted stock, restricted stock units, performance units, performance shares and other stock and cash awards to employees, directors and consultants and subsidiary corporations’ employees and consultants. The 2017 Plan replaces the Omeros Corporation 2008 Equity Incentive Plan (the 2008 Plan). Any stock option awards granted under the 2008 Plan that were outstanding as of the effective date of the 2017 Plan remain in effect pursuant to their terms and, if the award is canceled or is repurchased, the shares underlying such award become available for grant under the 2017 Plan. Under the 2017 Plan, stock options must be granted with exercise prices not less than the fair market value of the common stock subject to the stock option on the date of the grant and the options may not exceed 10 years.
72
Under the 2008 Plan, we granted incentive and non-statutory stock options to employees, directors and non-employees. Options were granted with exercise prices equal to the closing fair market value of the common stock on the date of the grant. The options granted were generally for 10-year terms and vested over a four-year period.
As of December 31, 2018, a total of 12,648,463 shares were reserved for issuance under our stock plans and outstanding warrants, of which 2,092,210 were available for future grants. In February 2019, annual stock option grants totaling approximately 1.5 million shares with an exercise price of $13.45 per share were granted to all eligible employees. The options vest monthly on a straight-line basis over four years.
Stock-based compensation expense includes amortization of stock options granted to employees, directors and non-employees and has been reported in our Consolidated Statements of Operations and Comprehensive Loss as follows:
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Research and development | $ | 4,961 | $ | 5,240 | $ | 6,304 | |||||
| Selling, general and administrative | 6,752 | 7,448 | 7,278 | ||||||||
| Total stock-based compensation expense | $ | 11,713 | $ | 12,688 | $ | 13,582 | |||||
The fair value of each option grant to employees and directors is estimated on the date of grant using the Black-Scholes option-pricing model. The following assumptions were applied to employee and director stock option grants during the periods ended:
| Year Ended December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| Estimated weighted-average fair value | $ | 10.32 | $ | 8.66 | $ | 6.89 | |||||
| Weighted-average assumptions: | |||||||||||
| Expected volatility | 78 | % | 74 | % | 74 | % | |||||
| Expected term, in years | 6.0 | 6.0 | 5.7 | ||||||||
| Risk-free interest rate | 2.68 | % | 2.05 | % | 1.63 | % | |||||
| Expected dividend yield | — | % | — | % | — | % | |||||
During the years ended December 31, 2018 and 2017, we granted to non-employees options to purchase 20,000 shares and 25,000 shares of common stock, respectively. In connection with the non-employee options, we recognized expense of $187,000, $549,000, and $313,000 during the years ended December 31, 2018, 2017 and 2016, respectively.
Stock option activity for all stock plans is as follows:
Options Outstanding | Weighted- Average Exercise Price per Share | Remaining Contractual Life (in years) | Aggregate Intrinsic Value (In thousands) | |||||||||
| Balance at December 31, 2017 | 9,657,259 | $ | 10.39 | |||||||||
| Granted | 1,891,757 | 15.01 | ||||||||||
| Exercised | (765,949 | ) | 8.79 | |||||||||
| Forfeited/expired | (469,929 | ) | 13.42 | |||||||||
| Balance at December 31, 2018 | 10,313,138 | $ | 11.22 | 6.41 | $ | 11,319 | ||||||
| Vested and expected to vest at December 31, 2018 | 10,005,615 | $ | 11.13 | 6.34 | $ | 11,283 | ||||||
| Exercisable at December 31, 2018 | 7,468,806 | $ | 10.21 | 5.51 | $ | 10,984 | ||||||
The total intrinsic value of options exercised during the years ended December 31, 2018, 2017 and 2016 was $11.4 million, $16.4 million and $13.6 million, respectively.
At December 31, 2018, there were 2,844,332 unvested options outstanding that will vest over a weighted-average period of 8.78 years. Excluding non-employee stock options, the remaining estimated compensation expense to be recognized in connection with these unvested options is $334.3 million.
73
Note 13—Income Taxes
The components of income tax benefit are as follows:
December 31, | |||||||||||
| 2018 | 2017 | 2016 | |||||||||
| (In thousands) | |||||||||||
| Current income tax benefit: | |||||||||||
| Federal | $ | — | $ | — | $ | — | |||||
| State | — | — | — | ||||||||
| Total current income tax benefit | — | — | — | ||||||||
| Deferred income tax benefit: | |||||||||||
| Federal | 11,261 | — | — | ||||||||
| State | 1,668 | — | — | ||||||||
| Total deferred income tax benefit | 12,929 | — | — | ||||||||
| Income tax benefit | $ | 12,929 | $ | — | $ | — | |||||
We have a history of losses and therefore have historically not made a provision for income taxes. However, in 2018 we recorded an income tax benefit of $12.9 million related to the issuance of our Convertible Notes. In accordance with intra-period tax allocation rules, the deferred tax liability related to the equity component of the convertible debt is a source of income that can be used to recognize the tax benefit of the current year loss through continuing operations. The tax benefit related to the issuance of our Convertible Notes will not recur in future years. Deferred income taxes reflect the tax effect of net operating loss and tax credit carryforwards and the net temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes.
Significant components of deferred income taxes are as follows:
December 31, | |||||||
| 2018 | 2017 | ||||||
| (In thousands) | |||||||
| Deferred tax assets: | |||||||
| Net operating loss carryforwards | $ | 113,928 | $ | 90,498 | |||
| Research and development tax credits | 35,739 | 26,748 | |||||
| Stock-based compensation | 8,969 | 7,829 | |||||
| Deferred rent | 2,037 | 2,123 | |||||
| Other | 10,564 | 4,759 | |||||
| Total deferred tax assets | 171,237 | 131,957 | |||||
| Deferred tax liabilities: | |||||||
| Property and equipment | (151 | ) | (10 | ) | |||
| Equity component of Convertible Notes | (12,706 | ) | — | ||||
| Total deferred tax liabilities | (12,857 | ) | (10 | ) | |||
| Net deferred tax assets before valuation allowance | 158,380 | 131,947 | |||||
| Less valuation allowance | (158,380 | ) | (131,947 | ) | |||
| Net deferred tax assets | $ | — | $ | — | |||
As of December 31, 2018 and 2017, we had federal net operating loss carryforwards of approximately $513.2 million and $414.5 million, respectively, and state net operating losses of approximately $125.0 million and $68.9 million, respectively.
As of December 31, 2017, we remeasured certain deferred tax assets and liabilities based on the rates at which they are expected to reverse in the future, which is generally 21.0% pursuant to the passage of the Tax Cuts and Jobs Act of 2017 (the Act). As permitted by Staff Accounting Bulletin No. 118, we recorded a $61.3 million provisional reduction in our net deferred tax assets related to the remeasurement in December 2017. During 2018 we completed our analysis relating to the Act which resulted in no material adjustment to our original remeasurement.
74
In certain circumstances, due to ownership changes, our net operating loss and tax credit carryforwards may be subject to limitations under Section 382 of the Internal Revenue Code. To date, we have not completed a Section 382 study. Unless previously utilized or otherwise limited, our net operating loss and research and development tax credit carryforwards originating in 2017 and prior years expire between 2019 and 2038. Net operating loss originating in 2018 have an indefinite life.
We have established a 100% valuation allowance due to the uncertainty of our ability to generate sufficient taxable income to realize the deferred tax assets. Our valuation allowance increased $26.4 million in 2018, and $24.9 million in 2016 due to net operating losses incurred during these periods. During 2017, our valuation allowance decreased $32.0 million due to the passage of the Act partially offset by additional net operating losses incurred during the year.
Reconciliation of income tax computed at federal statutory rates to the reported provisions for income taxes is as follows:
| Year ended December 31, | ||||||||
| 2018 | 2017 | 2016 | ||||||
| U.S. Federal statutory rate on net loss | (21 | )% | (34 | )% | (34 | )% | ||
| State tax, net of federal tax benefit | (2 | )% | (2 | )% | (2 | )% | ||
| Effects of statutory rate change | — | % | 115 | % | — | % | ||
| Change in valuation allowance | 19 | % | (60 | )% | 37 | % | ||
| Tax credits | (5 | )% | (11 | )% | (4 | )% | ||
| Other | — | % | (8 | )% | 3 | % | ||
| Effective tax rate | (9 | )% | — | % | — | % | ||
We file federal and certain state income tax returns, which provides varying statutes of limitations on assessments. However, because of net operating loss carryforwards, substantially all of our tax years remain open to federal and state tax examination.
We recognize interest and penalties related to the underpayment of income taxes as a component of income tax expense. To date, there have been no interest or penalties charged to us in relation to the underpayment of income taxes.
Note 14—401(k) Retirement Plan
We have adopted a 401(k) plan. Beginning in 2017, our 401(k) retirement plan provides for an annual company discretionary match on employee contributions, initially set at a maximum of 4.0% of each participating employee’s eligible earnings, with a maximum company match of $4,000 per employee per year. All employees are eligible to participate, provided they meet the requirements of the plan.
75
Note 15—Quarterly Information (Unaudited)
The following table summarizes the unaudited statements of operations and comprehensive loss for each quarter of 2018 and 2017 (in thousands, except per share amounts):
| 2018 | For the Quarter Ended | |||||||||||||||
| March 31, | June 30, | September 30, | December 31, | |||||||||||||
| Revenue (1) | $ | 1,588 | $ | 1,655 | $ | 4,608 | $ | 22,017 | ||||||||
| Total costs and expenses | 29,277 | 32,272 | 40,050 | 40,491 | ||||||||||||
| Loss from operations | (27,689 | ) | (30,617 | ) | (35,442 | ) | (18,474 | ) | ||||||||
| Net loss | (30,054 | ) | (33,696 | ) | (39,472 | ) | (23,534 | ) | ||||||||
| Basic and diluted net loss per share | $ | (0.62 | ) | $ | (0.70 | ) | $ | (0.81 | ) | $ | (0.48 | ) | ||||
| 2017 | For the Quarter Ended | |||||||||||||||
| March 31, | June 30, | September 30, | December 31, | |||||||||||||
| Revenue | $ | 12,257 | $ | 17,151 | $ | 21,658 | $ | 13,760 | ||||||||
| Total costs and expenses | 24,982 | 29,090 | 26,768 | 27,881 | ||||||||||||
| Loss from operations | (12,725 | ) | (11,939 | ) | (5,110 | ) | (14,121 | ) | ||||||||
| Net loss | (15,089 | ) | (14,359 | ) | (7,482 | ) | (16,551 | ) | ||||||||
| Basic and diluted net loss per share | $ | (0.34 | ) | $ | (0.33 | ) | $ | (0.16 | ) | $ | (0.34 | ) | ||||
(1) As further described in Note 1, OMIDRIA was reimbursed under Medicare Part B from January 1, 2015 through December 31, 2017. For the period January 1, 2018 through September 30, 2018, OMIDRIA was not reimbursed separately for procedures involving patients covered under Medicare Part B. Beginning October 1, 2018, OMIDRIA was again reimbursed separately under Medicare Part B.
76
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
ITEM 9A. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
Our management, with the participation of our principal executive officer and principal financial officer, evaluated the effectiveness of our disclosure controls and procedures, as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act, as of December 31, 2018. Management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving their objectives, and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on the evaluation of our disclosure controls and procedures as of December 31, 2018, our principal executive and principal financial officers concluded that, as of such date, our disclosure controls and procedures were effective at the reasonable assurance level.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles.
Internal control over financial reporting cannot provide absolute assurance of achieving financial reporting objectives because of its inherent limitations. Internal control over financial reporting is a process that involves human diligence and compliance and is subject to lapses in judgment and breakdowns resulting from human failures. Internal control over financial reporting also can be circumvented by collusion or improper management override. Because of such limitations, there is a risk that material misstatements may not be prevented or detected on a timely basis by internal control over financial reporting. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design into the process safeguards to reduce, though not eliminate, this risk.
Our management, with the participation of our principal executive and principal financial officers, conducted an assessment of the effectiveness of our internal control over financial reporting as of December 31, 2018. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework (2013 framework). Based on the results of this assessment and on those criteria, our management concluded that our internal control over financial reporting was effective as of December 31, 2018.
Ernst & Young LLP has independently assessed the effectiveness of our internal control over financial reporting as of December 31, 2018 and its report is included below.
There was no change in our internal control over financial reporting identified in connection with the evaluation required by Rules 13a-15(d) and 15d-15(d) of the Exchange Act that occurred during our fourth fiscal quarter of 2018 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
77
Report of Independent Registered Public Accounting Firm
To the Shareholders and the Board of Directors
Omeros Corporation
Opinion on Internal Control over Financial Reporting
We have audited Omeros Corporation’s internal control over financial reporting as of December 31, 2018, based on criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), (the COSO criteria). In our opinion, Omeros Corporation (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2018, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of Omeros Corporation (the Company) as of December 31, 2018 and 2017, the related consolidated statements of operations and comprehensive loss, shareholders' deficit and cash flows for each of the three years in the period ended December 31, 2018, and the related notes of the Company and our report dated March 1, 2019 expressed an unqualified opinion thereon that included an explanatory paragraph regarding the Company’s ability to continue as a going concern.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management’s Annual Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
Seattle, Washington
March 1, 2019
78
| ITEM 9B. | OTHER INFORMATION |
None.
79
PART III
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
The information required by this item will be contained in our definitive proxy statement issued in connection with the 2019 Annual Meeting of Shareholders and is incorporated herein by reference. Certain information required by this item concerning executive officers is set forth in Part I of this Annual Report on Form 10-K under the heading “Business-Executive Officers and Significant Employees.”
| ITEM 11. | EXECUTIVE COMPENSATION |
The information required by this item will be contained in our definitive proxy statement issued in connection with the 2019 Annual Meeting of Shareholders and is incorporated herein by reference.
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED SHAREHOLDER MATTERS |
Except for the information set forth below, the information required by this item will be contained in our definitive proxy statement issued in connection with the 2019 Annual Meeting of Shareholders and is incorporated herein by reference.
Securities Authorized for Issuance Under Equity Compensation Plans
The following table provides certain information regarding our equity compensation plans in effect as of December 31, 2018:
Number of Securities to be Issued Upon Exercise of Outstanding Options, Warrants and Rights | Weighted-Average Exercise Price of Outstanding Options, Warrants and Rights | Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans | |||||||
| Equity compensation plans approved by security holders: | |||||||||
| 2017 Omnibus Incentive Compensation Plan (1) | 1,914,730 | $ | 15.60 | 2,092,210 | |||||
| 2008 Equity Incentive Plan (2) | 8,398,408 | $ | 10.22 | — | |||||
| Total | 10,313,138 | $ | 11.22 | 2,092,210 | |||||
(1) Our 2017 Omnibus Incentive Compensation Plan, or the 2017 Plan, provides for the grant of incentive and nonstatutory stock options, restricted stock, restricted stock units, stock appreciation rights, performance units and performance shares to employees, directors and consultants and subsidiary corporations’ employees and consultants. The 2017 Plan replaced the Omeros Corporation 2008 Equity Incentive Plan, or the 2008 Plan, and as a result we will not grant any new awards under the 2008 Plan. Any stock option awards granted under the 2008 Plan that were outstanding as of the effective date of the 2017 Plan remain in effect pursuant to their terms and, if the award is canceled or is repurchased, the shares underlying such award become available for grant under the 2017 Plan.
(2) The 2008 Plan provided for the grant of incentive and nonstatutory stock options, restricted stock, stock appreciation rights, performance units and performance shares to employees, directors and consultants and subsidiary corporations’ employees and consultants.
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
The information required by this item will be contained in our definitive proxy statement issued in connection with the 2019 Annual Meeting of Shareholders and is incorporated herein by reference.
80
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The information required by this item will be contained in our definitive proxy statement issued in connection with the 2019 Annual Meeting of Shareholders and is incorporated herein by reference.
81
PART IV
| ITEM 15. | EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
1. Financial Statements
See the Index to Consolidated Financial Statements in Part II, Item 8 of this Form 10-K.
2. Financial Statement Schedules
All schedules have been omitted as the required information is either not required, not applicable or otherwise included in the Financial Statements and notes thereto.
3. Exhibits
The following list of exhibits includes exhibits submitted with this Form 10-K as filed with the SEC and those incorporated by reference to other filings
EXHIBIT INDEX
| Exhibit Description | Incorporated by Reference | |||||
Exhibit No. | Form | File No. | Exhibit No. | Filing Date | Filed Herewith | |
| 3.1 | 10-K | 001-34475 | 3.1 | 03/31/2010 | ||
| 3.2 | 10-K | 001-34475 | 3.2 | 03/31/2010 | ||
| 4.1 | S-1/A | 333-148572 | 4.1 | 10/02/2009 | ||
| 4.2 | 8-K | 001-34475 | 10.3 | 05/19/2016 | ||
| 4.3 | 8-K | 001-34475 | 10.2 | 4/13/2018 | ||
| 4.4 | 8-K | 001-34475 | 4.1 | 11/15/2018 | ||
| 10.1* | S-1 | 333-148572 | 10.1 | 01/09/2008 | ||
| 10.2* | 10-K | 001-34475 | 10.6 | 03/16/2017 | ||
| 10.3* | 10-Q | 001-34475 | 10.2 | 11/07/2013 | ||
| 10.4* | S-8 | 333-218882 | 4.3 | 06/21/2017 | ||
| 10.5* | S-8 | 333-218882 | 4.4 | 06/21/2017 | ||
| 10.6* | 8-K | 001-34475 | 10.1 | 04/12/2010 | ||
| 10.7* | S-1 | 333-148572 | 10.12 | 01/09/2008 | ||
82
| 10.8* | S-1 | 333-148572 | 10.14 | 01/09/2008 | ||
| 10.9 | S-1 | 333-148572 | 10.15 | 01/09/2008 | ||
| 10.10* | S-1 | 333-148572 | 10.16 | 01/09/2008 | ||
| 10.11 | S-1 | 333-148572 | 10.17 | 01/09/2008 | ||
| 10.12* | 10-Q | 001-34475 | 10.3 | 08/08/2017 | ||
| 10.13 | 8-K | 001-34475 | 10.1 | 02/01/2012 | ||
| 10.14 | 10-Q | 001-34475 | 10.2 | 11/09/2012 | ||
| 10.15 | 10-K | 001-34475 | 10.18 | 03/18/2013 | ||
| 10.16 | 10-K | 001-34475 | 10.18 | 03/13/2014 | ||
| 10.17 | 10-Q | 001-34475 | 10.3 | 11/09/2015 | ||
| 10.18 | 10-Q | 001-34475 | 10.1 | 05/10/2017 | ||
| 10.19 | X | |||||
| 10.20† | S-1/A | 333-148572 | 10.29 | 09/16/2009 | ||
| 10.21† | S-1 | 333-148572 | 10.30 | 01/09/2008 | ||
| 10.22† | 10-K | 001-34475 | 10.24 | 03/16/2015 | ||
| 10.23† | S-1/A | 333-148572 | 10.31 | 09/16/2009 | ||
| 10.24† | S-1 | 333-148572 | 10.32 | 01/09/2008 | ||
83
| 10.25† | S-1/A | 333-148572 | 10.33 | 05/15/2009 | ||
| 10.26† | S-1/A | 333-148572 | 10.47 | 09/16/2009 | ||
| 10.27† | 10-K | 001-34475 | 10.28 | 03/18/2013 | ||
| 10.28† | 10-Q | 001-34475 | 10.1 | 05/12/2010 | ||
| 10.29† | 10-Q | 001-34475 | 10.1 | 05/10/2011 | ||
| 10.30† | 10-Q | 001-34475 | 10.1 | 05/09/2013 | ||
| 10.31† | 10-Q | 001-34475 | 10.2 | 08/10/2010 | ||
| 10.32† | 10-K | 001-34475 | 10.44 | 03/15/2011 | ||
| 10.33† | 10-K | 001-34475 | 10.45 | 03/15/2011 | ||
| 10.34† | 10-K | 001-34475 | 10.46 | 03/16/2015 | ||
| 10.35† | 10-Q | 001-34475 | 10.1 | 11/09/2015 | ||
| 10.36 | 8-K | 001-34475 | 10.2 | 11/15/2018 | ||
| 10.37 | 8-K | 001-34475 | 10.1 | 10/05/2017 | ||
| 10.38 | 8-K | 001-34475 | 10.1 | 05/24/2018 | ||
| 10.39 | 10-Q | 001-34475 | 10.2 | 11/09/2016 | ||
| 10.40 | 8-K | 001-34475 | 10.1 | 10/17/2017 | ||
84
| 10.41 | 10-K | 001-34475 | 10.40 | 03/01/2018 | ||
| 10.42 | 8-K | 001-34475 | 10.1 | 04/13/2018 | ||
| 23.1 | X | |||||
| 31.1 | X | |||||
| 31.2 | X | |||||
| 32.1 | X | |||||
| 32.2 | X | |||||
| 101.INS | XBRL Instance Document | X | ||||
| 101.SCH | XBRL Taxonomy Extension Schema Document | X | ||||
| 101.CAL | XBRL Taxonomy Extension Calculation Linkbase Document | X | ||||
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase Document | X | ||||
| 101.LAB | XBRL Taxonomy Extension Labels Linkbase Document | X | ||||
| 101.PRE | XBRL Taxonomy Extension Presentation Linkbase Document | X | ||||
| * | Indicates management contract or compensatory plan or arrangement. |
| † | Portions of this exhibit are redacted in accordance with a grant of confidential treatment. |
85
| ITEM 16. | FORM 10-K SUMMARY |
Not included.
86
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| OMEROS CORPORATION |
| /s/ GREGORY A. DEMOPULOS, M.D. |
| Gregory A. Demopulos, M.D. |
| President, Chief Executive Officer and Chairman of the Board of Directors |
Dated: March 1, 2019
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature | Title | Date |
| /s/ GREGORY A. DEMOPULOS, M.D. | President, Chief Executive Officer and Chairman of the Board of Directors (Principal Executive Officer) | March 1, 2019 |
| Gregory A. Demopulos, M.D. | ||
| /s/ MICHAEL A. JACOBSEN | Vice President, Finance, Chief Accounting Officer and Treasurer (Principal Financial Officer and Principal Accounting Officer) | March 1, 2019 |
| Michael A. Jacobsen | ||
| /s/ RAY ASPIRI | Director | March 1, 2019 |
| Ray Aspiri | ||
| /s/ THOMAS F. BUMOL, PH.D. | Director | March 1, 2019 |
| Thomas F. Bumol, Ph.D. | ||
| /s/ THOMAS J. CABLE | Director | March 1, 2019 |
| Thomas J. Cable | ||
| /s/ PETER A. DEMOPULOS, M.D. | Director | March 1, 2019 |
| Peter A. Demopulos, M.D. | ||
| /s/ ARNOLD C. HANISH | Director | March 1, 2019 |
| Arnold C. Hanish | ||
| /s/ LEROY E. HOOD, M.D., PH.D. | Director | March 1, 2019 |
| Leroy E. Hood, M.D., Ph.D. | ||
| /s/ RAJIV SHAH, M.D. | Director | March 1, 2019 |
| Rajiv Shah, M.D. | ||
87