Exhibit 99.2
Lotus Pharmaceuticals, Inc. Reports Strong 2008 Financial Results
--Expands into Inner Mongolia to fuel future growth
- Revenues of $73.8 million
- Gross Profit of $33.4 million
- Net income of $12.8 million or $0.27 per diluted share
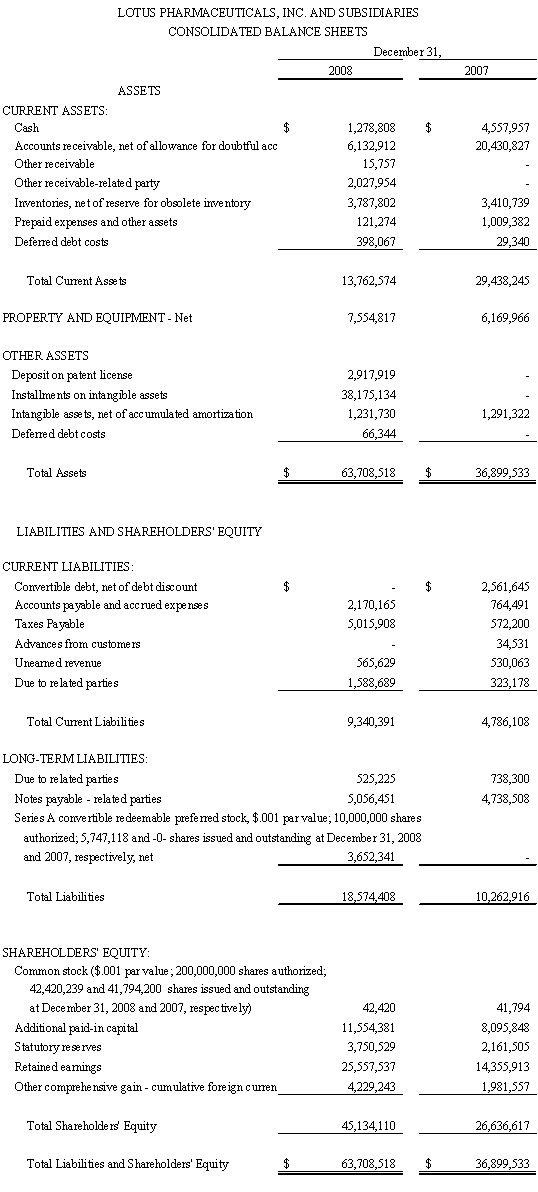
BEIJING, April 22, 2009 - Lotus Pharmaceuticals, Inc. (OTC Bulletin Board: LTUS.OB, “Lotus” or the “Company”) today reported its strong 2008 results with total net revenues increasing to $73.8 million, up 30% from 2007 total net revenues; gross profit increasing to $33.4 million, up 44% over 2007 gross profit of $23.2 million; net income $12.8 million or $0.27 diluted earnings per share, compared to net income of $11.2 million, or diluted earnings per share of $0.26, in 2007.
“We achieved substantially all of our goals for 2008. We exceeded the “make good” net income and remain on track to meet the 2009 full-year goal,” stated Dr. Zhongyi Liu, Chairman and CEO of Lotus. “We’ve also focused our expansion into Inner Mongolia with plans of new revenue lines, and as we establish these lines through 2009 till 2011 we are positioning ourselves for ambitious growth starting from 2011 and beyond, while maintaining steady growth from 2009 to 2011.”
“Gross profit growth has been strong and we believe has proven our business model’s resilience in the current global and local economic climate. We believe our business model can weather market and economic storms as it is supported by the diversification in revenues generated from prescription drugs ranging from self-produced and self-branded to third party manufactured, from retail sales of OTC drugs and from manufacturing and R&D for third party customers; Additionally, we believe we continuously make our operation more efficient and position Lotus to benefit from preferential tax policies offered in Inner Mongolia. Once our plant in Inner Mongolia begins to run in full production capacity, which we believe will be in 2013, we could save at least $15.5 million from significantly reduced taxes, utilities, and labor costs, in comparison to the cost of the same type of operations in Beijing.”
“According to Chinese government’s recent universal healthcare plan, the government will spend RMB850 billion ($124.3 billion) in the next three years to provide accessible and affordable healthcare to the country’s 1.3 billion people. We see this as a wonderful opportunity because demands for drugs and active pharmaceutical ingredients (“APIs”) are expected to increase and we believe could continue to increase until 2020. We also anticipate the decrease of drugs’ pricings due to the open bidding system and government’s restrictions on hikes of drugs prices.”
“We entered into 2008 with a strong push to increase collections of our outstanding account receivables (“AR”) and reduced outstanding AR amount significantly in 2008. We intend to maintain the AR level of 2008 and to request customers to prepay orders prior to our shipment of drugs. We ended the year with committed footprint in Inner Mongolia, the acquisition of intellectual rights of two drugs, application and the submission of two drugs to the SFDA. We achieved our goals for the year and exceeded our financial objectives.”
Commentary
The 2008 revenue increase was attributed to wholesale revenues which increased by 57%. The increases were primarily due to strong sales in Brimonidine Tartrate Eye Drops product and third party manufactured drugs. 100% of the prescription drugs Lotus sold are covered under the National Health Insurance. The Company has promoted strongly the sale of Brimonidine Tartrate Eye Drops and gained national exclusive rights to five third- party manufactured prescription drugs. Lotus believes due to the strong market demand for these drugs, together with the Company’s sales and marketing efforts, steady growth is expected in 2009 regardless of the softening of economy. The decrease in retail sales from Lotus’ drug stores was primarily attributable to the slowdown in economic growth and to reduced local traffic resulting from restricted city traffic during the 2008 Olympics held in Beijing. Lotus believes retail revenues will increase in 2009 as it increases its efforts to expand its OTC drugs sales channels to include other drugs stores and increase drugs of high profit margin concerning gastrointestinal illness and influenza.
Gross profit increased 44% to $33.4 million in 2008, compared to $23.2 million in 2007. Gross margin was 45% in 2008 compared to 41% in 2007. The increase in gross margin was primarily due to increase in sales revenues and more efficient management of purchase prices of APIs and drugs manufactured by third parties, and production cost controls.
Operating expenses for 2008 increased 54% to approximately $18.3 million (25% of net revenues) compared to approximately $11.9 million (21% of net revenues) for 2007. This increase resulted from the increase in selling expenses from $6.5 million in 2007 to $14.9 in 2008 million, which was the result of increased product sales in 2008, and management’s high commission and bonuses policy to incentivize sales and collections in order to generate sufficient cash to meet near term capital commitments, such as the new manufacturing plant construction project and the acquisition of a Chinese Class I drug patent. However, management has modified its sales and collection incentive policy and reduced the sales commission percentage and collection bonuses for 2009. General and administrative expenses were $ 2.2 million in 2008 as compared to $3.0 million in 2007, a decrease of 27% from 2007, which was a result of our continuous efforts to control corporate spending despite increases in professional fees.
As of December 31, 2008, the Company had cash of $1.3 million. A decrease of working capital from $24.6 million at the end of 2007 to $4.4 million at the end of 2008 was primarily due to the Company’s cash outlays for deposits on land use rights for our planned new Inner Mongolia facility and the acquisition of intellectual properties. The significant decline of days sales outstanding in ARs in 2008 showed clients paid sooner, which translated into an overall decrease in cash conversion days. This was due to the Company’s determined efforts to make Company’s working capital less tied up in ongoing operations.
Cash flow provided by operating activities for 2008 was $37.4 million and was primarily used to fund the expansion into Inner Mongolia for the land use rights and related expenses of $32.6 million, and the purchases of new drugs’ patent rights. The Company believes capital for the expansion in Inner Mongolia in 2009 will come from cash flow provided by operating activities, debt and equity financings, and government grants.
Drugs Pipeline
The Company’s drug portfolio development strategy mainly targets the treatment of cardio-cerebro vascular diseases, diabetes and asthma. The two injection drugs that Lotus developed and submitted in 2006 and 2007 are likely to get SFDA’s approvals this year. They are Sodium Aescinate-Injection for surgery swollenness and Gatifloxacin Lactate-Injection for pneumonia. The below is the adjusted currently anticipated SFDA approval timetable for its five new drugs:
Drugs Name | Anticipated Production Year |
Sodium Aescinate-Injection | 2009 |
Gatifloxacin Lactate-Injection | 2009 |
Calcium Dibutyryl Adenosine Monophosphate-Injection | 2010 |
Isosorbide Mononitrate-Sustained Release Tablets | 2010 |
Laveo-Bambutero | 2012 |
2009 Outlook
Based on information available to management at this time, Lotus currently anticipates its growth in net income in 2009 to increase between 30% and 40% from 2008 to the range from $16.6 million to $17.9 million.
Highlights of Recent Developments
• | Management has decided to start the construction of Inner Mongolia plant buildings on May 9, 2009 and to attempt to complete it by the end of 2009. |
2
• | In addition to the cost of land use rights and related expenses (approximately $32.6 million) which Liang Fang has paid, other components of the project are yet to be funded, including construction costs of approximately $17.5 million, costs associated with the various production lines estimated at approximately $33.6 million and working capital of approximately $7.3 million. |
• | The Company entered into an intellectual rights transfer contract with Beijing Yipuan Bio-Medical Technology Co., Ltd. in December 2008 to acquire the drug Yipubishan, a highly effective and stable octreotide acetate injection solution, In the first quarter of 2009, the Company has paid up $7.9 million as required in the transfer. The Company believes this could contribute $11.0 million to 2009’s sale revenues with net margin of 75%. |
• | The Company announced two drugs entering into clinical testing of SFDA in 2008, Isosorbide Mononitrate sustained release tablet, a highly effective drug for the prevention and treatment of angina; and Laevo-Bambutero, a Class 1 drug to treat asthma. |
• | Lotus obtained the patent, along with exclusive production rights for Laevo-Bambutero in China, through the technology transfer agreement with Dongguan Kaifa for a cash payment of $7.0 million and a 3% royalty on products sales. The Company has already paid $2.9 million to Dongguan Kaifa. The Company intends to market Laevo-Bambutero as a better alternative to Bambutero for the treatment of asthma, since it uses an integrated method of composition and has fewer side effects. Lotus plans to launch the drug by 2012, pending on the approval from SFDA. Class I drug in China means that it is a breakthrough invention in China and there is no similar drug elsewhere outside of China. Currently the Company believes 30-50 million patients in China suffer from asthma. |
About Lotus Pharmaceuticals, Inc. (www.lotuspharma.com)
Lotus Pharmaceuticals, Inc. controls and operates Liang Fang Pharmaceutical, Ltd. (“Liang Fang”) and Enze Jiashi Pharmaceutical, Ltd. (“Enze”), two pharmaceutical companies located in Beijing, China. Together, Liang Fang and Enze (“Lotus East”) undertake the development, production, marketing and distribution of pharmaceutical products. The Company believes Lotus East has some of the most advanced pharmaceutical production equipment in China and its manufacturing facilities meet national Good Manufacturing Practices. The Company distributes its own portfolio of drugs and pharmaceutical products produced by other manufacturers in the PRC through an extensive national distribution network. Lotus owns ten pharmacies in Beijing through which it directly sells over 5,000 western drugs, Traditional Chinese Medicines and medical equipment items.
Safe Harbor Statement
This press release contains “forward-looking statements” within the meaning of the “safe-harbor” provisions of the Private Securities Litigation Reform Act of 1995. Such statements involve known and unknown risks, uncertainties and other factors that could cause the actual results of the Company to differ materially from the results expressed or implied by such statements, including, but not limited to, changes from anticipated levels of sales, future national or regional economic and competitive and regulatory conditions, changes in relationships with customers, access to capital, increased costs, difficulties in developing and marketing new products, marketing existing products, customer acceptance of existing and new products, the time to get new drugs approved by the SFDA and other factors. Additional information regarding risks can be found in the Company’s Annual Report on Form 10K filed with the SEC. Accordingly, although the Company believes that the expectations reflected in such forward-looking statements are reasonable, there can be no assurance that such expectations will prove to be correct. The Company has no obligation to update the forward-looking information contained in this press release.
Lotus Pharmaceuticals, Inc.
Mr. Adam Wasserman, CFO
Phone: | +1-877-801-0344 |
info@LotusEast.com
http://www.lotuspharma.com
3
4

5

6