Exhibit 99.1

Advancing Medicines for Neglected and Rare Diseases Through Innovative Business Models OTC: KBIO www.kalobios.com | Corporate Overview - October 14, 2016 1

Forward-Looking Statements | Corporate Overview 2 This presentation contains forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements reflect management’s current knowledge, assumptions, judgment and expectations regarding future performance or events. Although management believes that the expectations reflected in such statements are reasonable, they give no assurance that such expectations will prove to be correct, and actual results could differ materially from the forward-looking statements.Words such as “will,” “expect,” “intend,” “plan,” “predict,” “potential,” “possible,” and similar expressions identify forward-looking statements, including, without limitation, statements related to the scope, progress, expansion, and costs of developing and commercializing our product candidates, anticipated regulatory incentives for product candidates, and anticipated expenses related to development activities, clinical trials and the development and potential commercialization of product candidates.Forward-looking statements are subject to risks and uncertainties including, but not limited to, the Company’s ability to execute its revised strategy and business plan; the ability of the Company to list its common stock on a national securities exchange; the Company’s access to limited cash reserves and its ability to obtain additional capital, including the additional capital which will be necessary to complete the clinical trials that the Company has initiated or plans to initiate; the potential timing and outcomes of clinical studies of benznidazole, lenzilumab, ifabotuzumab or any other product candidates; the commercial viability of the Company’s proposed drug pricing program; the ability of the Company to timely source adequate supply of its development products from third-party manufacturers on whom the Company depends; the potential, if any, for future development of any of its present or future products; the Company's ability to successfully progress, partner or complete further development of its programs; the ability of the Company to identify and develop additional products; the uncertainties inherent in clinical testing; the timing, cost and uncertainty of obtaining regulatory approvals and reaching other development milestones; the Company’s ability to apply or receive Priority Review Vouchers; the Company's ability to protect the Company's intellectual property; competition; and changes in the regulatory landscape or the imposition of regulations that affect the Company's products; and the various risks described in the "Risk Factors" and elsewhere in the Company's periodic and other filings with the Securities and Exchange Commission.You are cautioned not to place undue reliance on any forward-looking statements, which speak only as of the date of this release. The company has no obligation, and expressly disclaims any obligation to update, revise or correct any of the forward-looking statements, whether as a result of new information, future events or otherwise.

Turnaround focused on high-value neglected and rare diseases, including pediatricPipeline of products with multiple potential near- and longer-term catalystsBenznidazole: anticipated streamlined US approval process relying on existing data; potentially eligible for Priority Review Voucher (PRV) that may be able to be sold for significant returnLenzilumab (recombinant mAb) initially directed toward pediatric and orphan blood cancers; potential regulatory/development incentives, including possible eligibility for PRVIfabotuzumab (recombinant mAb) targeting pediatric brain cancers; ready for partneringExperienced, focused new management with track record of efficient execution, commitment to leadership in responsible, transparent pricing Compelling Investment Opportunity | Corporate Overview 3
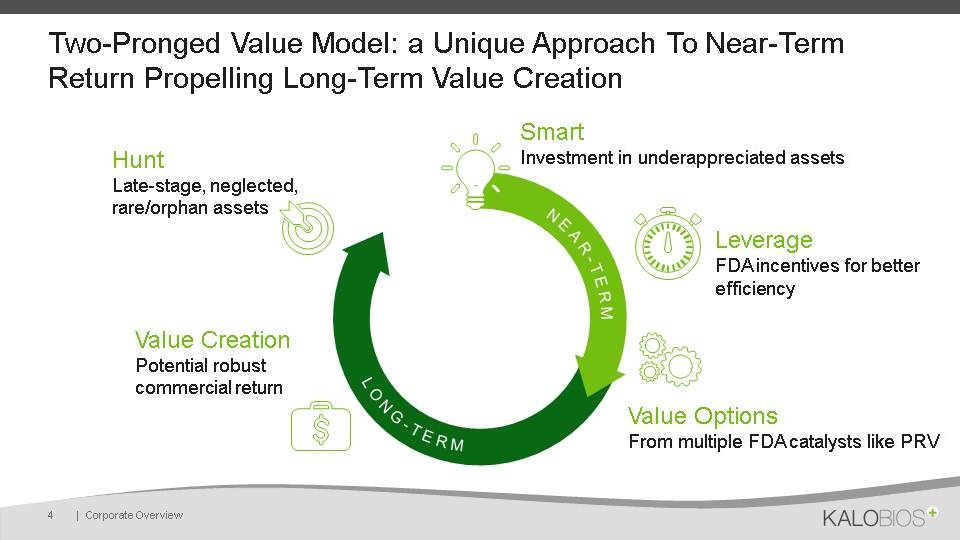
Two-Pronged Value Model: a Unique Approach To Near-Term Return Propelling Long-Term Value Creation | Corporate Overview 4 Smart Investment in underappreciated assets Leverage FDA incentives for better efficiency Value Options From multiple FDA catalysts like PRV Value Creation Potential robust commercial return HuntLate-stage, neglected, rare/orphan assets NEAR-TERM LONG-TERM

Leadership Cameron Durrant, MD, MBAChairman and CEOSenior pharmaceutical and biotech exec, turnaround specialistSenior exec roles at Pharmacia/Pfizer, J+J in US, Merck, GSK in Europe; experience as Exec Chairman, CEO and CFO; CEO roles at three specialty pharma groupsExpertise in anti-infectives, pediatrics, oncologyMorgan LamChief Scientific OfficerExtensive industry experience in clinical researchHead of Clinical Operations and Development KaloBiosExecutive Director, Medical Affairs, GeronDave Tousley, MBA, CPAInterim Chief Financial Officer More than 35 years experience in biotech, spec pharma, big pharmaSenior exec roles, President, COO, CFOPasteur, Merieux, Connaught, AVAX, airPharma, PediaMed, DARA Biosciences Tariq Arshad, MD, MBAConsultant focused on medical affairsExtensive industry experience in clinical developmentExperienced in orphan, pediatrics, oncology, pediatric oncology, immunologyPharmacia/Pfizer, Genentech, XomaNiv Caviar, MBAConsultant focused on corporate/business developmentSenior functional roles in marketing, business development, strategic planningSenior exec roles, CEO, EVP-CBO, CFO, VP Bus DevLa Jolla Pharma, Allergan, Suneva, SpineOvations, Affymetrix, AccentureChristopher BoweConsultant focused on corporate affairsDeep experience advising CEOs on articulating, executing strategy through corporate affairsFormer Strategic Affairs advisor at Schering-PloughIndustry thought leader, prior award-winning writer Financial Times Steve Pal, MBAConsultant focused on commercialGlobal pharma and consumer healthcare product commercialisationFormer Corporate VP Global Strategic Marketing, Health Outcomes, Strategy and Research, Global Medical Affairs, Allergan | Corporate Overview 5

Board of Directors Cameron Durrant, MD, MBAChairman | Corporate Overview 6 Ronald Barliant, JDOf Counsel at Goldberg Kohn, extensive experience representing debtors and creditors in complex bankruptcy casesUnited States bankruptcy judge for the Northern District of Illinois 1988 to 2002Dale Chappell, MD, MBAManaging member of Black Horse Capital Management, LLCPreviously associate at Chilton Investment Company specializing in healthcareTimothy Morris, CPACFO AcelRx Pharmaceuticals; also Head of Business DevelopmentCFO, SVP Finance and Global Corporate Development VIVUS, Inc.Ezra FriedbergFounder and general partner of Multiplier Capital, LP>20 years investing experience across public and private companies
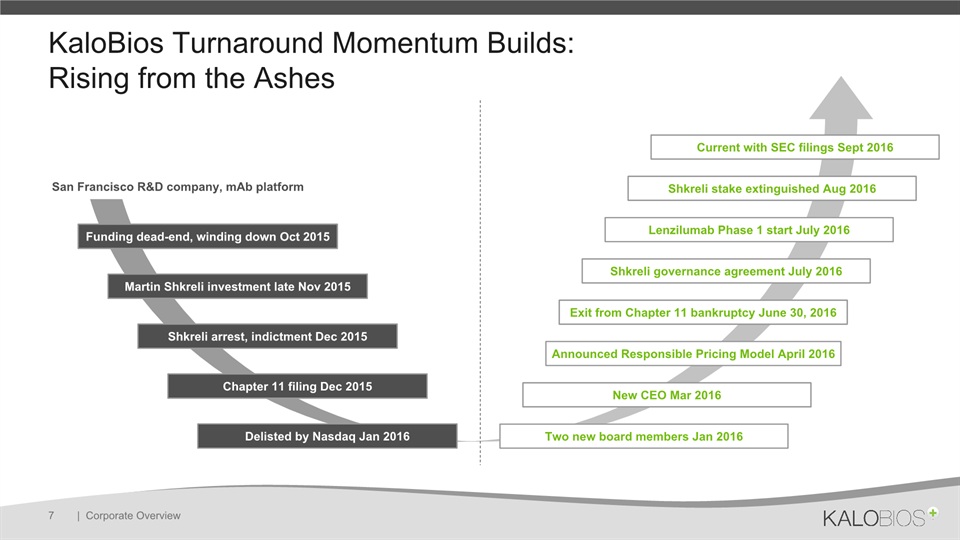
KaloBios Turnaround Momentum Builds: Rising from the Ashes | Corporate Overview 7 Funding dead-end, winding down Oct 2015 Martin Shkreli investment late Nov 2015 Shkreli arrest, indictment Dec 2015 Chapter 11 filing Dec 2015 Delisted by Nasdaq Jan 2016 Two new board members Jan 2016 New CEO Mar 2016 Announced Responsible Pricing Model April 2016 Exit from Chapter 11 bankruptcy June 30, 2016 Lenzilumab Phase 1 start July 2016 Shkreli governance agreement July 2016 Shkreli stake extinguished Aug 2016 Current with SEC filings Sept 2016 San Francisco R&D company, mAb platform
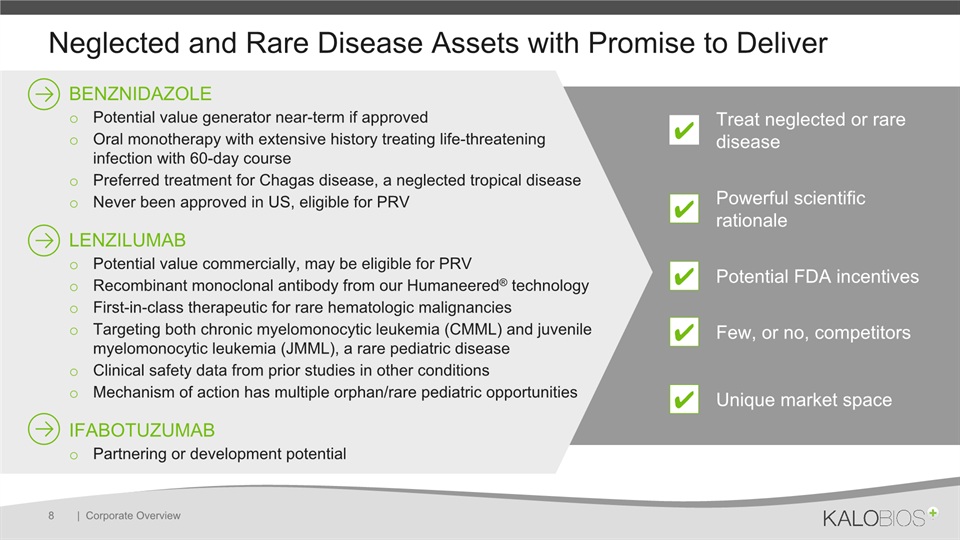
Neglected and Rare Disease Assets with Promise to Deliver | Corporate Overview 8 BENZNIDAZOLEPotential value generator near-term if approvedOral monotherapy with extensive history treating life-threatening infection with 60-day course Preferred treatment for Chagas disease, a neglected tropical diseaseNever been approved in US, eligible for PRVLENZILUMABPotential value commercially, may be eligible for PRVRecombinant monoclonal antibody from our Humaneered® technologyFirst-in-class therapeutic for rare hematologic malignanciesTargeting both chronic myelomonocytic leukemia (CMML) and juvenile myelomonocytic leukemia (JMML), a rare pediatric diseaseClinical safety data from prior studies in other conditionsMechanism of action has multiple orphan/rare pediatric opportunitiesIFABOTUZUMABPartnering or development potential ✔ Treat neglected or rare disease ✔ Powerful scientific rationale Potential FDA incentives ✔ ✔ Few, or no, competitors ✔ Unique market space
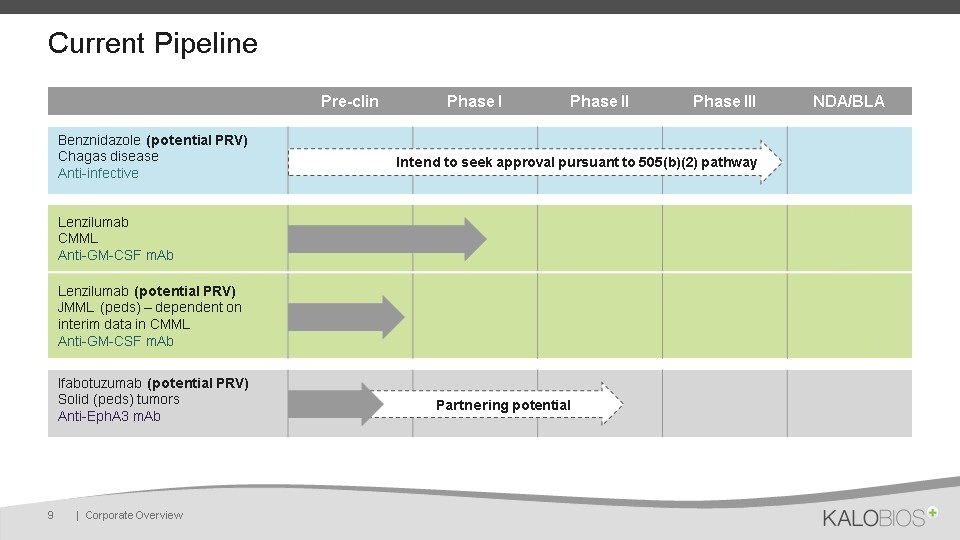
Current Pipeline | Corporate Overview 9 Pre-clin Phase I Phase II Phase III Target NDA/BLA Benznidazole (potential PRV)Chagas diseaseAnti-infective LenzilumabCMML Anti-GM-CSF mAb Lenzilumab (potential PRV)JMML (peds) – dependent on interim data in CMMLAnti-GM-CSF mAb Ifabotuzumab (potential PRV)Solid (peds) tumorsAnti-EphA3 mAb 2018 Partnering potential Intend to seek approval pursuant to 505(b)(2) pathway

Anticipated KaloBios Milestones* Potential FDA Meeting Minutes IND SubmissionPotential Orphan Designation Bioavailability Results CMML P1 interim dataCMML Orphan designationJMML FDA meeting Other potential FDA designations JMML Orphan/RPD designationOther potential FDA designationsJMML P1 first patient dosedCMML Phase 1b/2 Start CMML P1 final data | Corporate Overview 10 * Does not include potential outlicensing, other deals, or development timelines for ifabotuzumab Pre-IND FDA Meeting Q42016 Q12017 Q22017 Q32017 Q42017 Benznidazole Lenzilumab

Priority Review Vouchers (PRVs) Create Options For Significant Potential Return existing PRV programs | Corporate Overview 11 Highest disclosed sale price so far for PRV is $350MM Rare Pediatric Diseases (RPD) Neglected Tropical Diseases (NTD) FDA can award a PRV for eligible products upon approvalMost recent PRV issued was for RPD on September 19, Sarepta’s eteplirsen for DMDIncentivizes companies to bring medicines through FDA approval to U.S. patients for overlooked diseasesHolder of PRV can receive a priority review for any drug/biologic application with FDAPRV can be sold to another company seeking a competitive jump 2

Two-Year Outlook We Expect PRV Policy to Remain Generally Stable Zika virus highlights importance of NTD incentives Post-election, congressional gridlock expected to continueFocus likely on big issues: Medicare Part D, Affordable Care Act May be less political will to change small issuesAdditional PRV programs for medical countermeasures and generics proposed | Corporate Overview 12 Immediate Outlook Rare Pediatric Diseases (RPD) Neglected Tropical Diseases (NTD) NTD PRVNo pending or expected change to policy RPD PRVCongress extended law through 2016Longer reauthorization anticipated

Benznidazole a potential treatment for Chagas disease | Corporate Overview 13
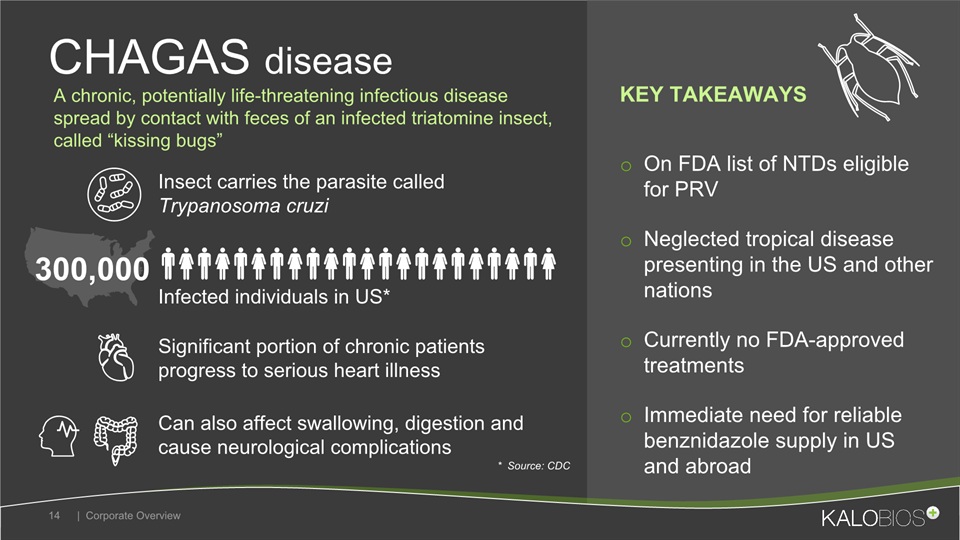
On FDA list of NTDs eligible for PRVNeglected tropical disease presenting in the US and other nations Currently no FDA-approved treatmentsImmediate need for reliable benznidazole supply in US and abroad CHAGAS disease | Corporate Overview 14 Insect carries the parasite called Trypanosoma cruzi Infected individuals in US* Can also affect swallowing, digestion and cause neurological complications 300,000 * Source: CDC A chronic, potentially life-threatening infectious disease spread by contact with feces of an infected triatomine insect, called “kissing bugs” KEY TAKEAWAYS Significant portion of chronic patients progress to serious heart illness
Benznidazole: the Preferred Treatment Globally for Chagas | Corporate Overview 15 40 years of clinical experience, primarily in Latin AmericaUsed in tens of thousands of patients Believed to work by inducing free radicals causing DNA damage within the T. cruzi parasite Efficacy ~60% or more in children and can be higher in young children, according to published studiesBetter tolerated in children; side effect profile extremely well knownPotential for streamlined development through FDA 505(b)(2) approval pathwayOnly available in US via special protocol with CDC Drug supply believed to be sporadic, product difficult to obtain N N HN N

Benznidazole Has Potential for 505(b)(2) New Drug Application | Corporate Overview 16 Less complexity in overall dossierOur manufacturing validation will be similar to that for a new drug – a competitive advantage 505(b)(2) Hybrid between an ANDA and full NDATo demonstrate efficacy & safety, some data will be drawn from studies done by othersNeed to file non-clinical & manufacturing packages 505(j) Abbreviated NDA (ANDA)Primarily for drugs that are the same as approved products (i.e. generics) Clinical studies to demonstrate equivalence onlyDo not need to repeat studies demonstrating efficacy & safety Usually do not require any non-clinical studies 505(b)(1) Full ApplicationFor drugs that have not been previously approved by the FDATo demonstrate efficacy & safety, data predominantly obtained from studies conducted by the sponsor to demonstrate efficacy & safetyNeed to file complete non-clinical and manufacturing packages
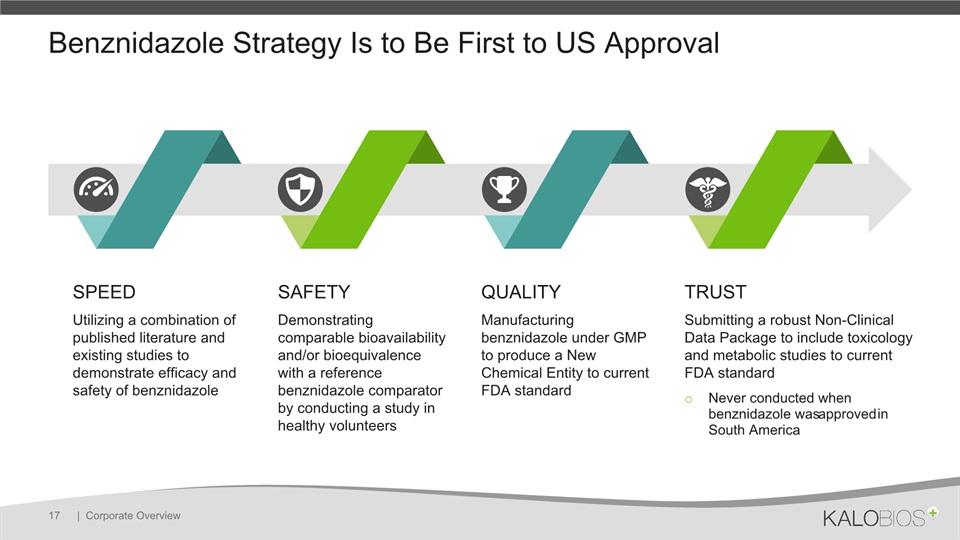
Benznidazole Strategy Is to Be First to US Approval | Corporate Overview 17 SPEEDUtilizing a combination of published literature and existing studies to demonstrate efficacy and safety of benznidazole SAFETYDemonstrating comparable bioavailability and/or bioequivalence with a reference benznidazole comparator by conducting a study in healthy volunteers QUALITYManufacturing benznidazole under GMP to produce a New Chemical Entity to current FDA standard TRUSTSubmitting a robust Non-Clinical Data Package to include toxicology and metabolic studies to current FDA standardNever conducted when benznidazole was approved in South America

2016 2017 H1 H2 H1 H2 Manufacturing (CMC) Analytical methods development Drug substance Drug product Stability studies Non-Clinical (Non-Human) Metabolism / PK Toxicity assessment T.cruzi biology Environmental assessment Clinical Summary of relevant clinical studies Right of Reference & data extraction for studies Human bioavailability (BA) Study Regulatory Pre-IND meeting & open IND Orphan drug status application Pediatric plan Pre-NDA meeting Assembly of NDA & NDA filing Benznidazole Target Approval Plan | Corporate Overview 18 Pre-IND meeting IND submission NDA filing 2018
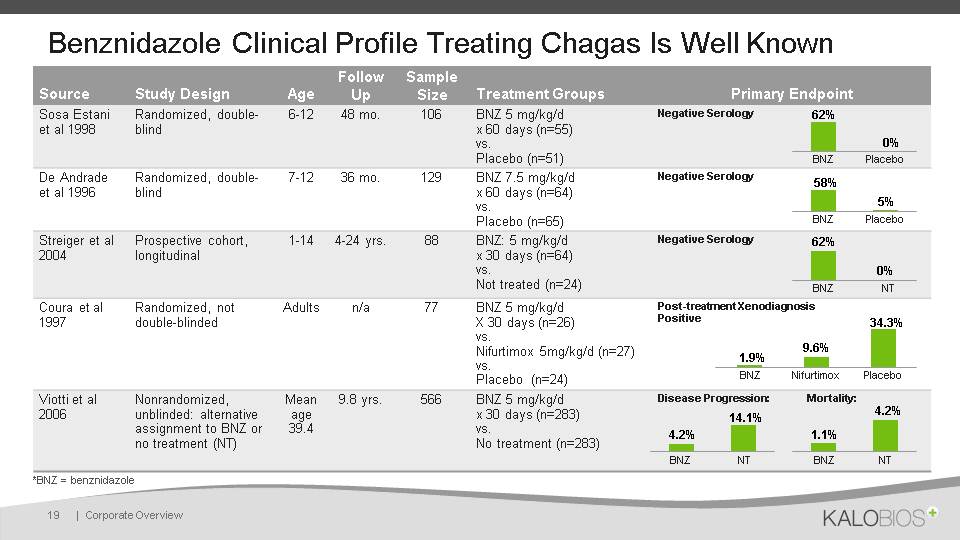
Benznidazole Clinical Profile Treating Chagas Is Well Known | Corporate Overview 19 Source Study Design Age Follow Up Sample Size Treatment Groups Primary Endpoint Sosa Estani et al 1998 Randomized, double-blind 6-12 48 mo. 106 BNZ 5 mg/kg/d x 60 days (n=55)vs.Placebo (n=51) Negative Serology De Andrade et al 1996 Randomized, double-blind 7-12 36 mo. 129 BNZ 7.5 mg/kg/d x 60 days (n=64)vs.Placebo (n=65) Negative Serology Streiger et al 2004 Prospective cohort, longitudinal 1-14 4-24 yrs. 88 BNZ: 5 mg/kg/d x 30 days (n=64)vs.Not treated (n=24) Negative Serology Coura et al 1997 Randomized, not double-blinded Adults n/a 77 BNZ 5 mg/kg/d X 30 days (n=26)vs.Nifurtimox 5mg/kg/d (n=27)vs.Placebo (n=24) Post-treatment Xenodiagnosis Positive Viotti et al2006 Nonrandomized, unblinded: alternative assignment to BNZ or no treatment (NT) Mean age 39.4 9.8 yrs. 566 BNZ 5 mg/kg/d x 30 days (n=283)vs.No treatment (n=283) Disease Progression: Mortality: BNZ Placebo BNZ Placebo BNZ Nifurtimox Placebo BNZ NT BNZ NT BNZ NT *BNZ = benznidazole

Lenzilumab a potential pipeline in a product | Corporate Overview 20

KaloBios Humaneered® Platform Murine segments replaced by human sequences from human donor libraryNew segments have amino acid identity very near to human germ-line segments | Corporate Overview 21 THERAPEUTIC GOALS Proprietary and patented technology platform to engineer antibodies, typically murine, into high-affinity human antibodies for therapies ENGINEERING PROCESS Designed to eliminate immunogenicity that may arise from chimeric or conventionally humanized antibodiesClinically tested in more than 200 subjects with no evidence of serious immunogenicityDesigned to bind better and to be prone to less rejection
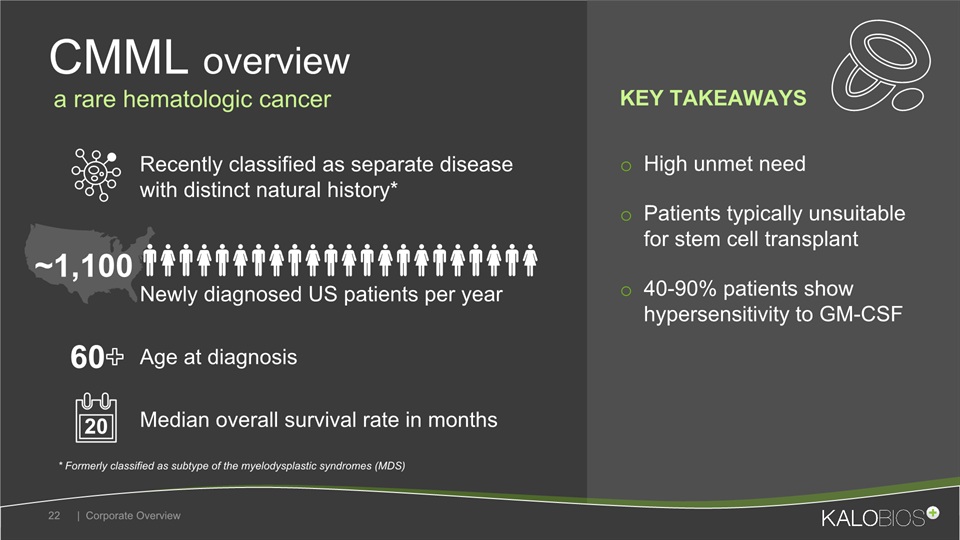
High unmet needPatients typically unsuitable for stem cell transplant40-90% patients show hypersensitivity to GM-CSF CMML overview | Corporate Overview 22 Recently classified as separate disease with distinct natural history* Median overall survival rate in months Newly diagnosed US patients per year Age at diagnosis ~1,100 * Formerly classified as subtype of the myelodysplastic syndromes (MDS) 60+ a rare hematologic cancer 20 KEY TAKEAWAYS
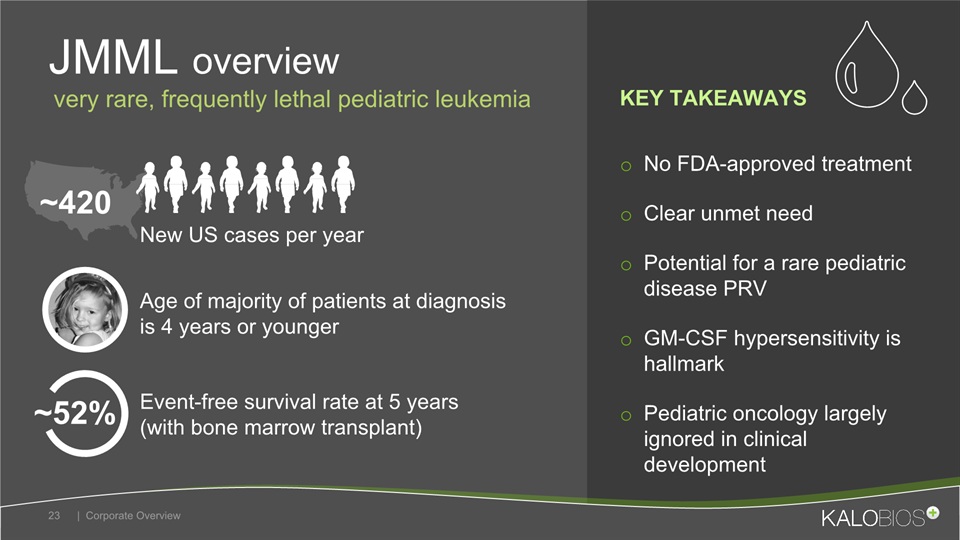
No FDA-approved treatmentClear unmet needPotential for a rare pediatric disease PRVGM-CSF hypersensitivity is hallmarkPediatric oncology largely ignored in clinical development JMML overview | Corporate Overview 23 Event-free survival rate at 5 years (with bone marrow transplant) New US cases per year Age of majority of patients at diagnosis is 4 years or younger ~420 very rare, frequently lethal pediatric leukemia ~52% KEY TAKEAWAYS

Neutralization of GM-CSF inhibits JAK-STAT and RAS signaling pathways in leukemic cells Mutations Affect GM-CSF Signaling, Confers Hypersensitivity in Leukemias | Corporate Overview 24 Inactivation of the NF1 tumor suppressor gene or oncogenic mutations in NRAS, KRAS2, CBL, or PTPN11. The values in parentheses are the percentages of individual genetic abnormalities among patients with JMML or CMML. Source: Adapted from Koike 2008, Loh 2009.
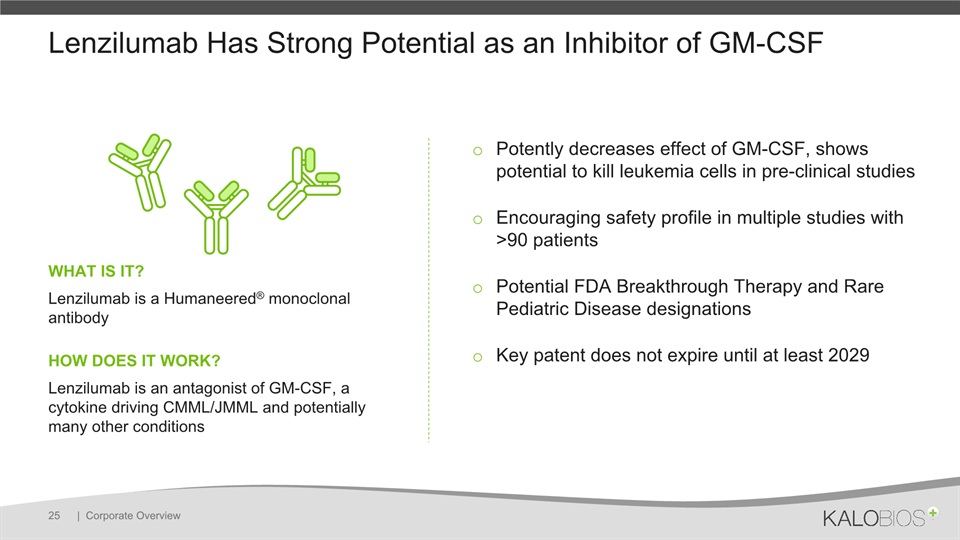
Lenzilumab Has Strong Potential as an Inhibitor of GM-CSF | Corporate Overview 25 Potently decreases effect of GM-CSF, shows potential to kill leukemia cells in pre-clinical studies Encouraging safety profile in multiple studies with >90 patientsPotential FDA Breakthrough Therapy and Rare Pediatric Disease designationsKey patent does not expire until at least 2029 WHAT IS IT? Lenzilumab is a Humaneered® monoclonal antibodyHOW DOES IT WORK?Lenzilumab is an antagonist of GM-CSF, a cytokine driving CMML/JMML and potentially many other conditions
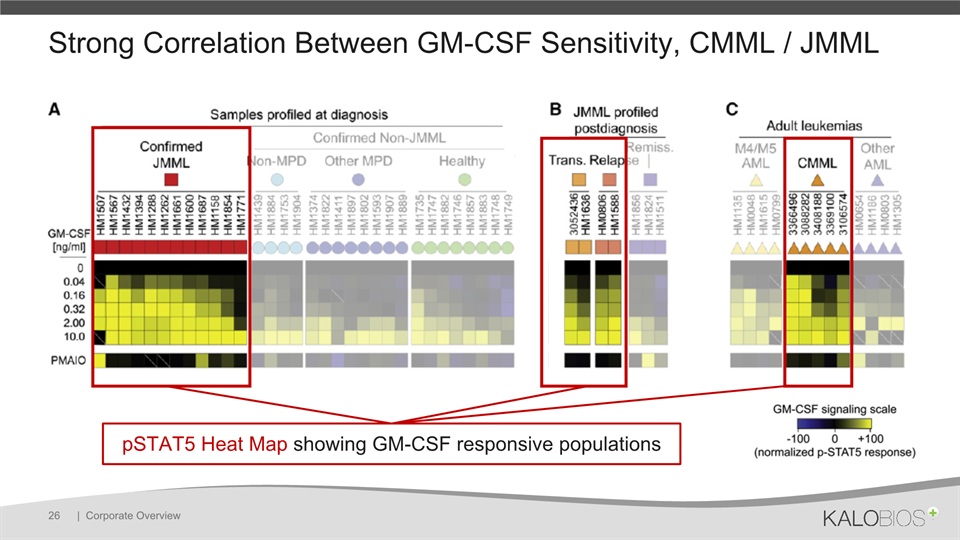
pSTAT5 Heat Map showing GM-CSF responsive populations Kotecha et al 2008 Strong Correlation Between GM-CSF Sensitivity, CMML / JMML | Corporate Overview 26
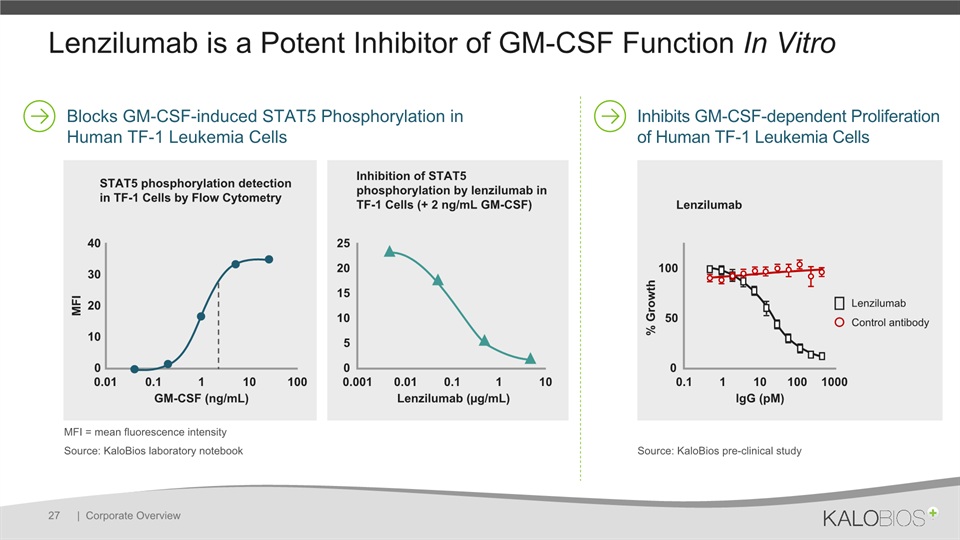
Lenzilumab is a Potent Inhibitor of GM-CSF Function In Vitro | Corporate Overview 27 Inhibits GM-CSF-dependent Proliferation of Human TF-1 Leukemia Cells Blocks GM-CSF-induced STAT5 Phosphorylation in Human TF-1 Leukemia Cells Source: KaloBios laboratory notebook STAT5 phosphorylation detection in TF-1 Cells by Flow Cytometry Inhibition of STAT5 phosphorylation by lenzilumab in TF-1 Cells (+ 2 ng/mL GM-CSF) 0.001 0.01 0.1 1 10 Lenzilumab (µg/mL) 25 20 15 10 0 5 40 30 20 10 0 MFI 0.01 0.1 1 10 100 GM-CSF (ng/mL) Lenzilumab 0.1 1 10 100 1000 lgG (pM) 100 50 0 Lenzilumab Control antibody % Growth Source: KaloBios pre-clinical study MFI = mean fluorescence intensity
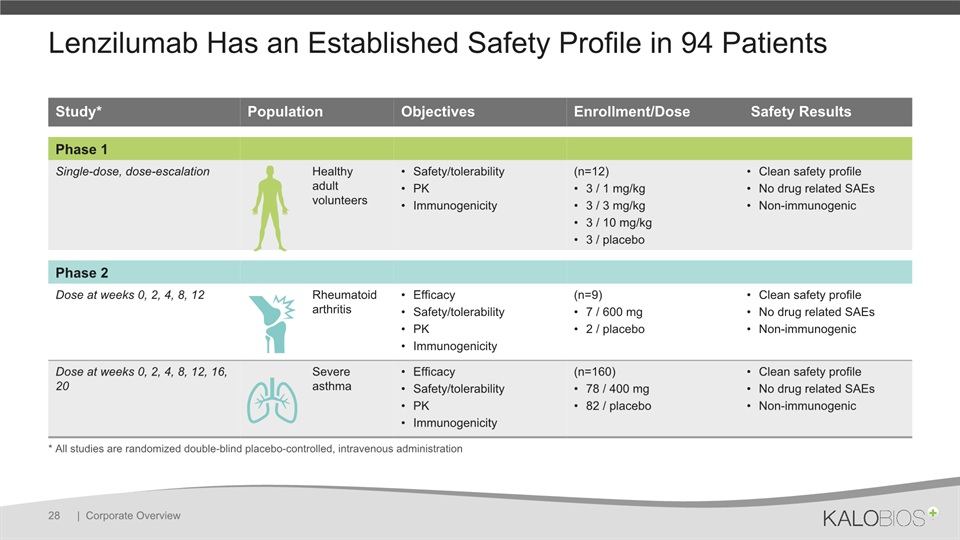
Lenzilumab Has an Established Safety Profile in 94 Patients | Corporate Overview 28 Study* Population Objectives Enrollment/Dose Safety Results Phase 1 Single-dose, dose-escalation Healthy adult volunteers Safety/tolerabilityPKImmunogenicity (n=12)3 / 1 mg/kg3 / 3 mg/kg3 / 10 mg/kg3 / placebo Clean safety profileNo drug related SAEsNon-immunogenic Phase 2 Dose at weeks 0, 2, 4, 8, 12 Rheumatoid arthritis EfficacySafety/tolerabilityPKImmunogenicity (n=9)7 / 600 mg2 / placebo Clean safety profileNo drug related SAEsNon-immunogenic Dose at weeks 0, 2, 4, 8, 12, 16, 20 Severe asthma EfficacySafety/tolerabilityPKImmunogenicity (n=160)78 / 400 mg82 / placebo Clean safety profileNo drug related SAEsNon-immunogenic * All studies are randomized double-blind placebo-controlled, intravenous administration
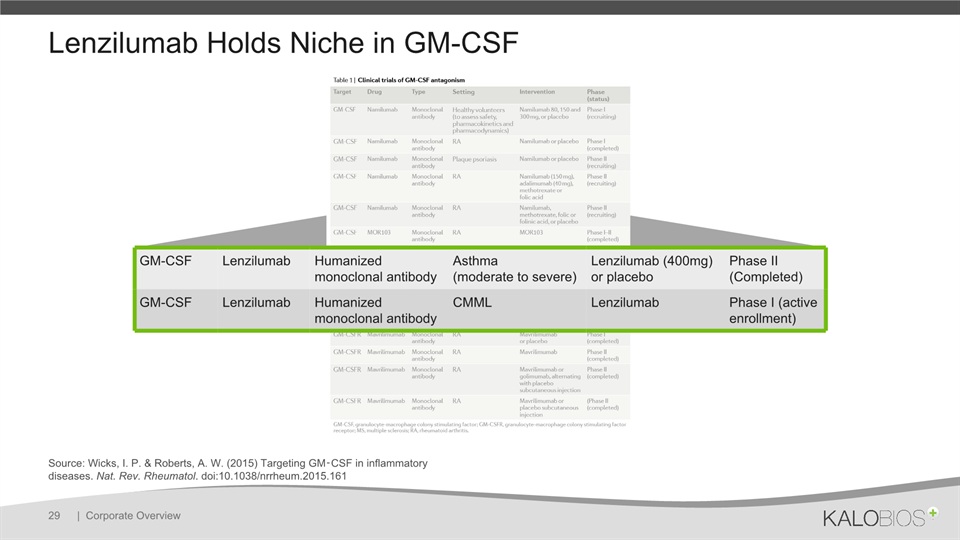
Lenzilumab Holds Niche in GM-CSF | Corporate Overview 29 Source: Wicks, I. P. & Roberts, A. W. (2015) Targeting GM‑CSF in inflammatory diseases. Nat. Rev. Rheumatol. doi:10.1038/nrrheum.2015.161 GM-CSF Lenzilumab Humanized monoclonal antibody Asthma (moderate to severe) Lenzilumab (400mg) or placebo Phase II (Completed) GM-CSF Lenzilumab Humanized monoclonal antibody CMML Lenzilumab Phase I (active enrollment)
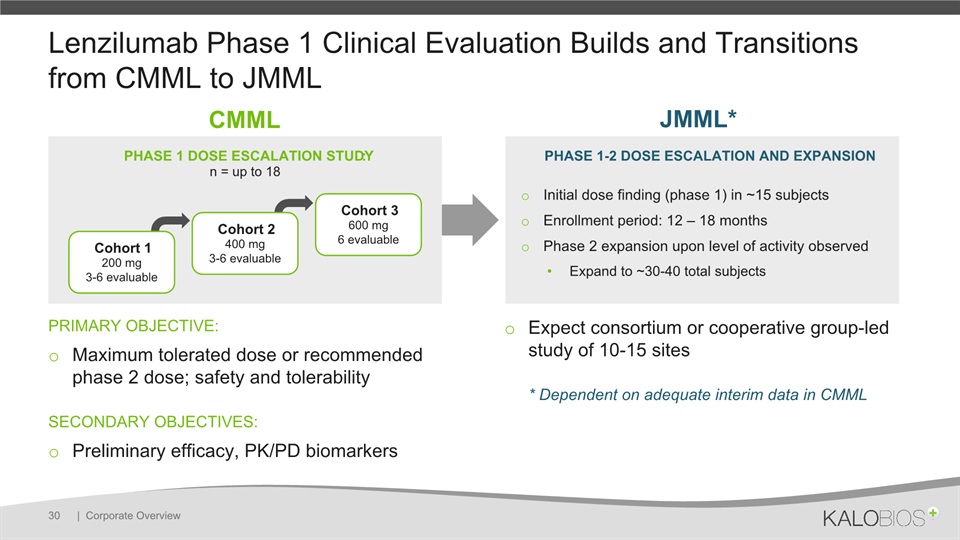
Lenzilumab Phase 1 Clinical Evaluation Builds and Transitions from CMML to JMML PRIMARY OBJECTIVE: Maximum tolerated dose or recommended phase 2 dose; safety and tolerabilitySECONDARY OBJECTIVES: Preliminary efficacy, PK/PD biomarkers Expect consortium or cooperative group-led study of 10-15 sites* Dependent on adequate interim data in CMML | Corporate Overview 30 PHASE 1-2 DOSE ESCALATION AND EXPANSIONInitial dose finding (phase 1) in ~15 subjectsEnrollment period: 12 – 18 monthsPhase 2 expansion upon level of activity observedExpand to ~30-40 total subjects CMML JMML* PHASE 1 DOSE ESCALATION STUDY:n = up to 18 Cohort 1200 mg3-6 evaluable Cohort 2400 mg3-6 evaluable Cohort 3600 mg6 evaluable

Lenzilumab CMML/JMML Planned Clinical Trial Execution Matrix | Corporate Overview 31 2016 2017 2018 2019 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 2019 CMML Phase 1 Dose Escalation Study Phase 1b/2 Expand Single Arm + Combination Study First patient dosed Preliminary safety/activity data Interim analysis ASH abstract JMML Phase 1 Dose Escalation Study Phase 2 Expansion JMML scientific advisory meeting Protocol design FDA meeting on JMML Phase 1 First patent dosed
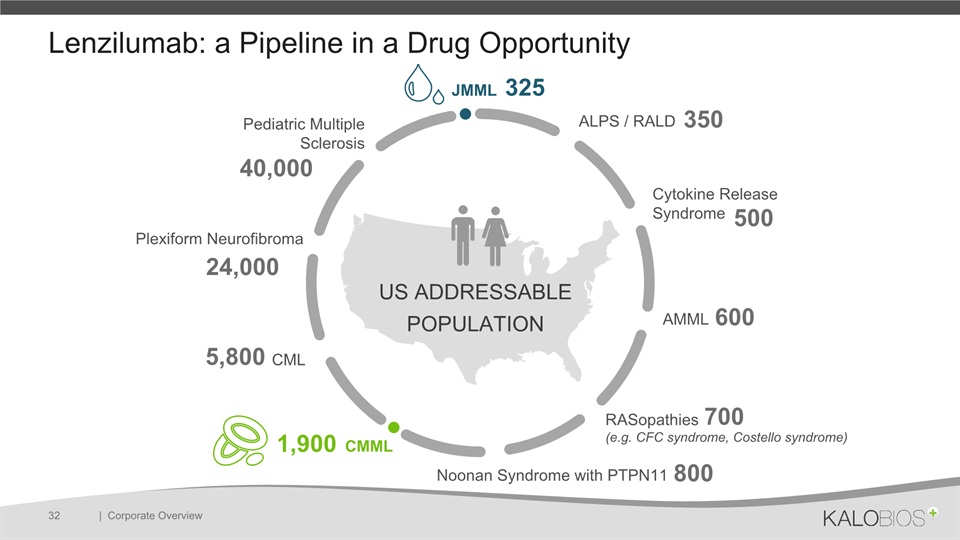
Lenzilumab: a Pipeline in a Drug Opportunity | Corporate Overview 32 JMML ALPS / RALD Cytokine Release Syndrome AMML RASopathies (e.g. CFC syndrome, Costello syndrome) Noonan Syndrome with PTPN11 CMML CML Plexiform Neurofibroma Pediatric Multiple Sclerosis US ADDRESSABLE POPULATION 325 350 500 600 700 800 1,900 5,800 24,000 40,000

Ifabotuzumab significant potential for partnering and development | Corporate Overview 33
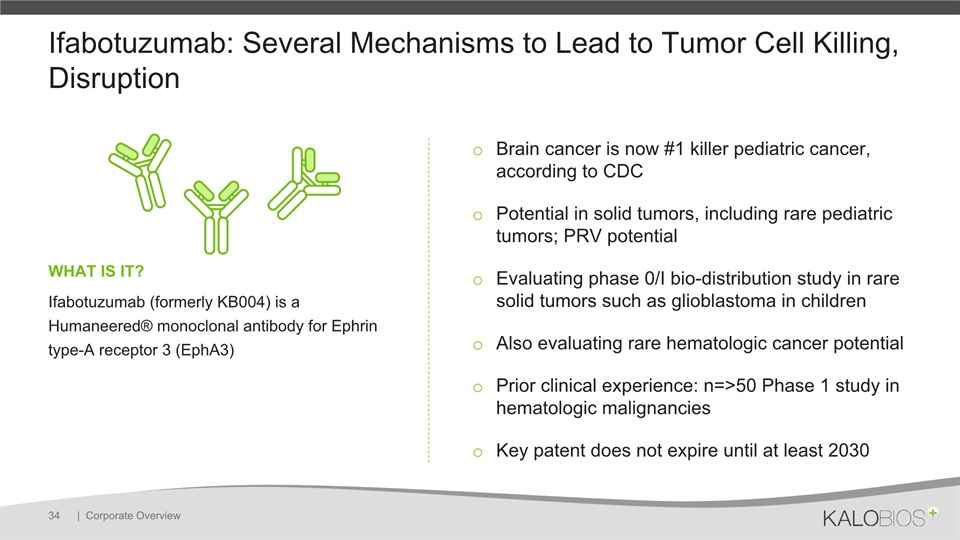
Ifabotuzumab: Several Mechanisms to Lead to Tumor Cell Killing, Disruption | Corporate Overview 34 Brain cancer is now #1 killer pediatric cancer, according to CDCPotential in solid tumors, including rare pediatric tumors; PRV potentialEvaluating phase 0/I bio-distribution study in rare solid tumors such as glioblastoma in children Also evaluating rare hematologic cancer potentialPrior clinical experience: n=>50 Phase 1 study in hematologic malignanciesKey patent does not expire until at least 2030 WHAT IS IT? Ifabotuzumab (formerly KB004) is a Humaneered® monoclonal antibody for Ephrin type-A receptor 3 (EphA3)
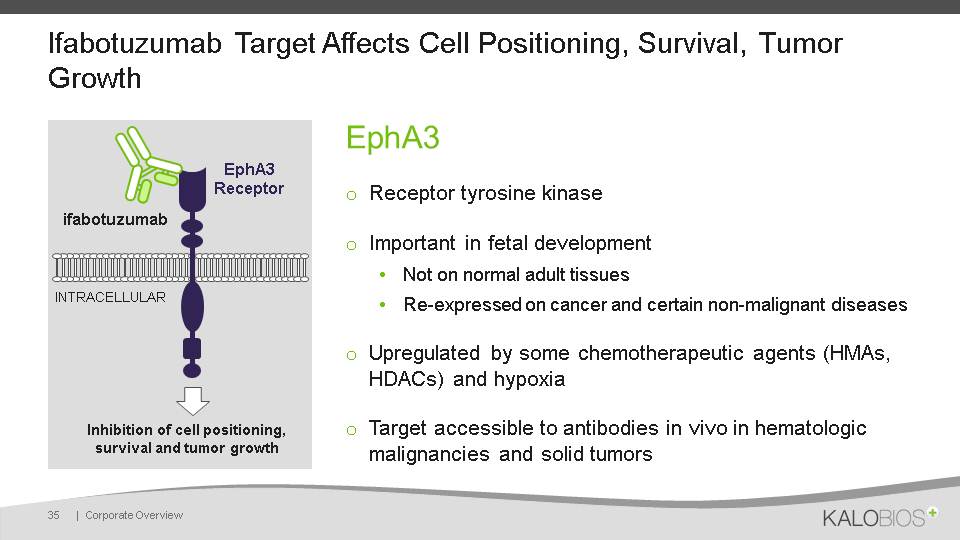
Ifabotuzumab Target Affects Cell Positioning, Survival, Tumor Growth EphA3Receptor tyrosine kinaseImportant in fetal developmentNot on normal adult tissuesRe-expressed on cancer and certain non-malignant diseasesUpregulated by some chemotherapeutic agents (HMAs, HDACs) and hypoxiaTarget accessible to antibodies in vivo in hematologic malignancies and solid tumors 35 | Corporate Overview EphA3 Receptor ifabotuzumab INTRACELLULAR Inhibition of cell positioning, survival and tumor growth
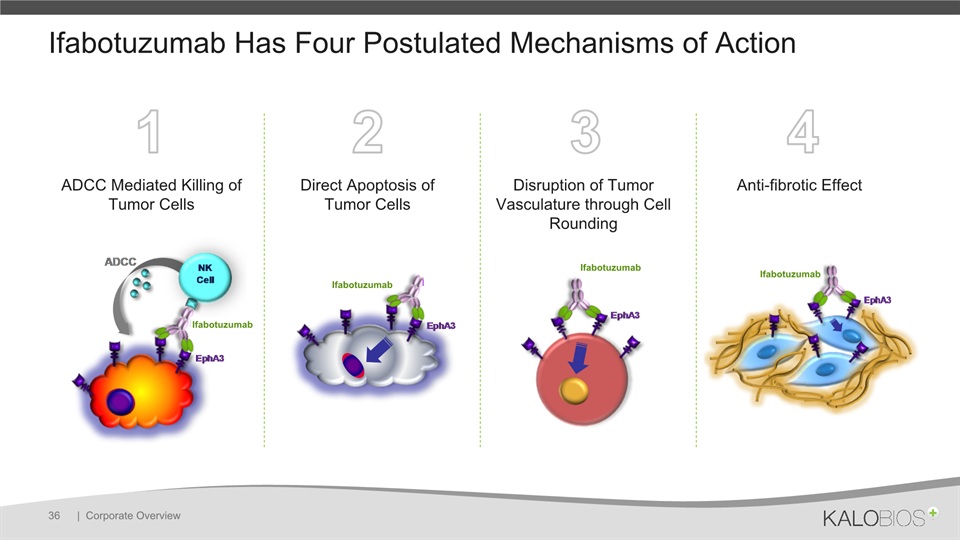
1 2 3 4 Ifabotuzumab Has Four Postulated Mechanisms of Action | Corporate Overview 36 ADCC Mediated Killing of Tumor Cells Direct Apoptosis of Tumor Cells Disruption of Tumor Vasculature through Cell Rounding Anti-fibrotic Effect Ifabotuzumab Ifabotuzumab Ifabotuzumab Ifabotuzumab
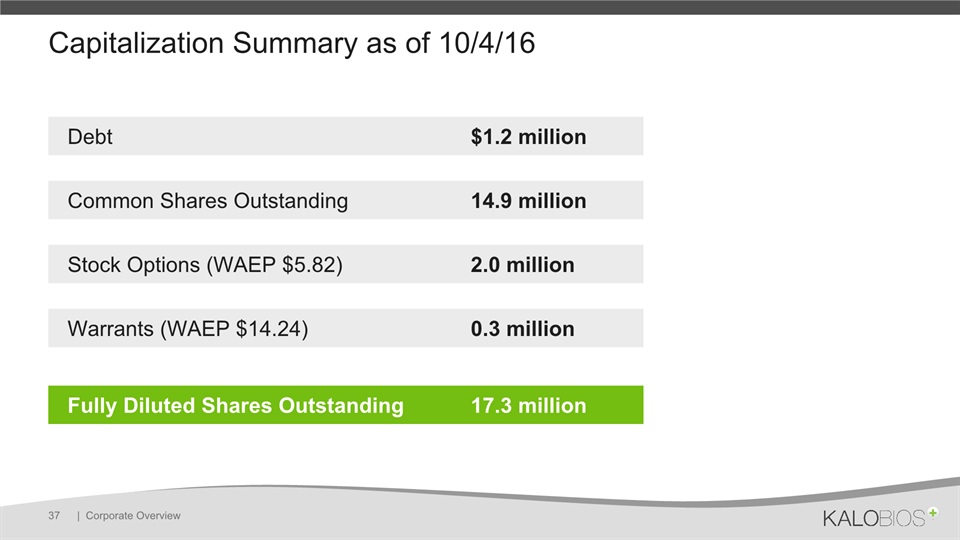
Capitalization Summary as of 10/4/16 | Corporate Overview 37 Debt $1.2 million Common Shares Outstanding 14.9 million Stock Options (WAEP $5.82) 2.0 million Warrants (WAEP $14.24) 0.3 million Fully Diluted Shares Outstanding 17.3 million

Demonstrated History of Swift Execution in Challenging Circumstances | Corporate Overview 38 JUN JUL AUG SEP OCT Emerge in six months from complex Chapter 11 bankruptcy Lenzilumab Phase 1 trial started Extinguished Martin Shkreli’s stake in the company Current with Securities and Exchange Commission periodic reporting requirementsAssembled and filed three 10-Qs and a 10-K within three months Market cap ~$48MM versus <$4MM preceding bankruptcy 30 24 29 23 4 2016

Summary Unique near-term/long-term potential value creation opportunity Thoughtful business strategy that leverages existing U.S. regulatory and development incentivesAttractive asset portfolio with multiple potential milestones and partnering prospectsDeeply committed and focused new management team with an unwavering commitment to success | Corporate Overview 39