SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
TO
FORM S-1
REGISTRATION STATEMENT
The Securities Act of 1933
(Exact name of registrant as specified in its charter)
Delaware (State or other jurisdiction of incorporation or organization) | 8071 (Primary Standard Industrial Classification Code Number) | 06-1614015 (I.R.S. Employer Identification Number) |
Gaithersburg, MD 20878
(240) 813-1260
(Address, including zip code, and telephone number, including area code, of registrant's principal executive offices)
Mary J. Mullany, Esq. Ballard Spahr LLP 1735 Market Street 51st Floor Philadelphia, PA 19103 (215) 665-8500 |
Large Accelerated Filer ☐ | Accelerated Filer ☐ |
Non-Accelerated Filer ☐ | Smaller Reporting Company ☒ |
Emerging Growth Company ☒ |

| Page | |
| PROSPECTUS SUMMARY | 1 |
| THE OFFERING | 8 |
| SUMMARY FINANCIAL DATA | 11 |
| RISK FACTORS | 13 |
| INFORMATION REGARDING FORWARD-LOOKING STATEMENTS | 39 |
| USE OF PROCEEDS | 40 |
| CAPITALIZATION | 41 |
| DILUTION | 43 |
| PRICE RANGE FOR OUR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS | 44 |
| DESCRIPTION OF CAPITAL STOCK | 46 |
| DESCRIPTION OF SECURITIES WE ARE OFFERING | 50 |
| PLAN OF DISTRIBUTION | 54 |
| MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS | 56 |
| BUSINESS | 64 |
| MANAGEMENT | 88 |
| EXECUTIVE COMPENSATION | 91 |
| CERTAIN RELATIONSHIPS AND RELATED PERSON TRANSACTIONS | 99 |
| SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS | 101 |
| LEGAL MATTERS | 102 |
| EXPERTS | 102 |
| WHERE YOU CAN FIND ADDITIONAL INFORMATION | 102 |
| OPGEN, INC. INDEX TO AUDITED CONSOLIDATED FINANCIAL STATEMENTS | F-1 |
| · | Our Acuitas DNA tests provide rapid microbial identification and antibiotic resistance gene information. These products include our Acuitas antimicrobial resistance, or AMR, Gene Panel u5.47 for complicated urinary tract infections in development as a clinical diagnostic test and available for Research Use Only, the QuickFISH® family of FDA-cleared and CE-marked diagnostics used to rapidly detect pathogens in positive blood cultures, and our Acuitas Resistome Tests for genetic analysis of hospital surveillance isolates. |
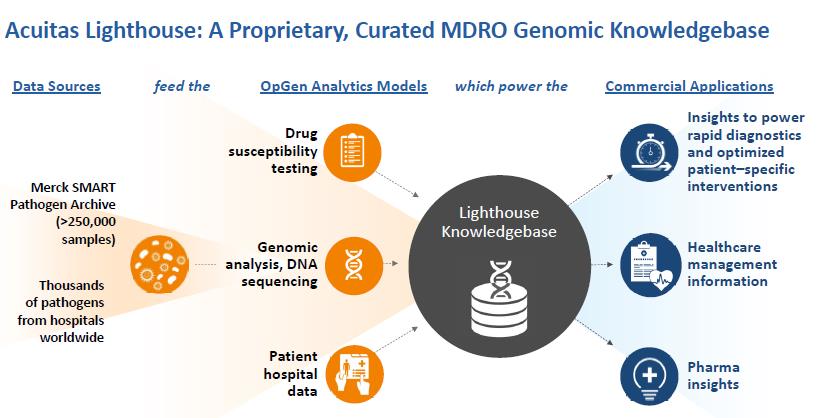
| · | Our Acuitas Lighthouse informatics systems are cloud-based HIPAA compliant informatics offerings that combine clinical lab test results with patient and hospital information to provide analytics and actionable insights to help manage MDROs in the hospital and patient care environment. Components of our informatics systems include the Acuitas Lighthouse Knowledgebase and the Acuitas Lighthouse Software. The Acuitas Lighthouse Knowledgebase is a relational database management system and a proprietary data warehouse of genomic data matched with antibiotic susceptibility information for bacterial pathogens. The Acuitas Lighthouse Software system includes the Acuitas Lighthouse Portal, a suite of web applications and dashboards, the Acuitas Lighthouse Prediction Engine, which is a data analysis software, and other supporting software components. The Acuitas Lighthouse Software can be customized and made specific to a healthcare facility or collaborator, such as a pharmaceutical company. |
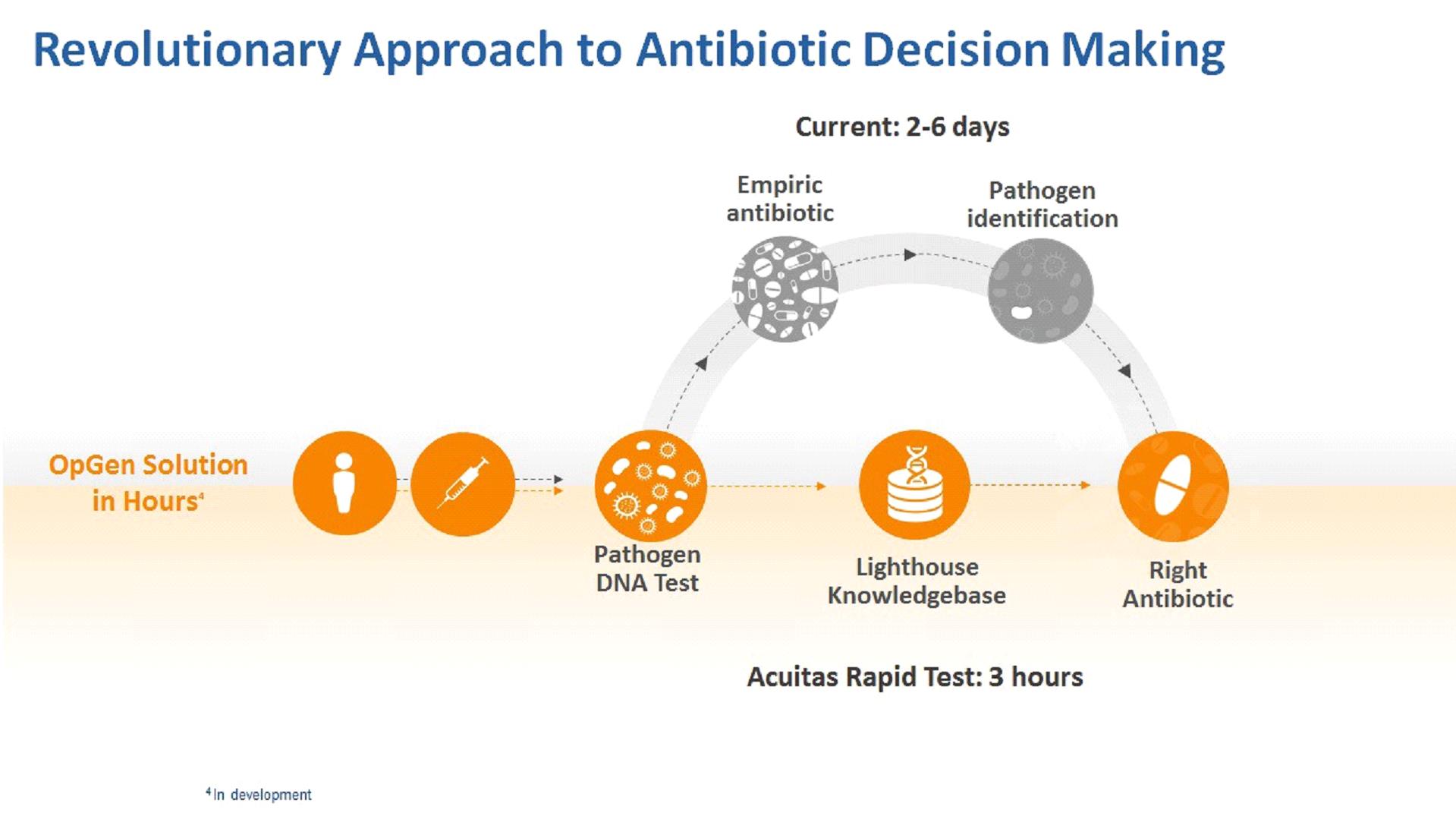
| · | Rapid diagnostics – We are developing OpGen-branded Acuitas AMR Gene Panel tests for use on the Thermo Fisher Scientific Applied Biosystems™ QuantStudio™ 5 Real-Time PCR System. The first of these new tests will be for management of patients with cUTI. We anticipate developing tests for additional clinical indications such as lower respiratory tract infections and for new antibiotic decision- making applications. The second rapid diagnostics growth driver will be through strategic partner relationships where we will work to expand channel access for our proprietary DNA tests through development and subsequent use of these tests, utilizing the Acuitas Lighthouse Software on established rapid in vitro diagnostic testing platforms. |
| · | Acuitas Lighthouse informatics and services – We are pursuing commercial opportunities to provide our Acuitas Lighthouse informatics and companion genomic testing to pharmaceutical companies and CROs, health systems, third party in vitro diagnostic companies, and government agencies. Through our Pharmaceutical/CRO services we are working to help accelerate clinical trials and new product launches and to establish early access for diagnostic tests to help guide decision-making for new antibiotics. Our focus in the health system segment is on helping guide antibiotic decision-making and supporting patient safety initiatives. We are actively pursuing government funding for development and deployment of our Acuitas Lighthouse informatics in the United States and internationally. |
| · | complete development, clinical evaluations, obtain necessary regulatory approvals, and successfully commercialize our AMR Gene Panel u5.47 for cUTI with a goal of achieving three-hour antibiotic resistance analysis from the time of specimen collection; |
| · | continue clinical evaluations for the AMR Gene Panel u5.47 in the first half of 2018; in January 2018 we introduced the AMR Gene Panel u5.47 as an RUO test and have a goal of making a FDA 510(k) submission in the fourth quarter of 2018 to support commercial launch; |
| · | obtain third party funding to expand our AMR Gene Panel development and access to additional third party rapid testing platforms; |
| · | expand our business collaborations with Merck and other pharmaceutical companies; |
| · | capitalize on opportunities to deploy our Acuitas Lighthouse informatics and genomic testing for pharmaceutical/CRO services; |
| · | grow our Acuitas Lighthouse data warehouse offerings for resistance and susceptibility data in hospital, hospital system, or broader community applications through continued development of the Acuitas Lighthouse Knowledgebase; |
| · | seek government funding to advance programs focused on identification and treatment of MDROs; and |
| · | continue development of our Acuitas Lighthouse Software and work to install Acuitas Lighthouse Software to customer sites in the United States and globally. |
| · | We are developing new diagnostic products for the more rapid identification of MDROs and antibiotic resistance genomic information. If we are unable to successfully develop, receive regulatory clearance or approval for or commercialize such new products and services, our business will be materially, adversely affected. |
| · | We expect to make significant additional investments in the future related to our diagnostic products and services, which investments will require additional financing transactions through the issuance of equity or debt. If we are unable to make such investments our business will suffer. |
| · | We have a history of losses, and we expect to incur losses for the next several years. Substantial doubt exists about our ability to continue as a going concern. |
| · | Our restructuring plans may not produce the cost savings we anticipate, and we may encounter difficulties associated with the related organizational change. |
| · | We are an early commercial stage company and may never be profitable. Our products and services may never achieve significant commercial market acceptance. |
| · | Our future success is dependent upon our ability to expand our customer base. |
| · | We have been awarded a contract by the CDC, and may enter into additional agreements with U.S. or other government agencies, which could be subject to uncertain future funding. |
| · | Our sales cycle for our marketed products and services is lengthy and variable, which makes it difficult for us to forecast revenue and other operating results. |
| · | We may enter into collaborations with third parties to develop product and services candidates. If these collaborations are not successful, our business could be adversely affected. |
| · | We may not be successful in finding strategic collaborators for continuing development of certain of our product or services candidates or successfully commercializing or competing in the market for certain indications. |
| · | The loss of key members of our senior management team or our inability to attract and retain highly skilled scientists and laboratory and field personnel could adversely affect our business. |
| · | We have limited experience in marketing and selling our products, and if we are unable to adequately address our customers' needs, it could negatively impact sales and market acceptance of our products and we may never generate sufficient revenue to achieve or sustain profitability. |
| · | If the utility of our current products and products in development is not supported by studies published in peer-reviewed medical publications, the rate of adoption of our current and future products and services by clinicians and healthcare facilities may be negatively affected. |
| · | The performance of clinical and economic utility studies is expensive and demands significant attention from our management team. |
| · | Our products and services are not covered by reimbursement by Medicare, Medicaid and other governmental and third-party payors. If we cannot convince our customers that the savings from use of our products and services will increase their overall reimbursement, our business could suffer. |
| · | If our sole laboratory facility or manufacturing facility becomes inoperable, we will be unable to perform Acuitas MDRO test services, or manufacture our QuickFISH and PNA Fish products, and our business will be harmed. |
| · | In order to meet the turn-around time required for our Acuitas MDRO test services, we rely on transport of specimens to our sole laboratory facility; any disruption in such transport could significantly adversely affect our business. |
| · | We rely on a limited number of suppliers or, in some cases, sole suppliers, for some of our laboratory instruments and materials and may not be able to find replacements or immediately transition to alternative suppliers. |
| · | If we cannot compete successfully with our competitors, we may be unable to increase or sustain our revenue or achieve and sustain profitability. |
| · | If we are unable to develop products to keep pace with rapid technological, medical and scientific change, our operating results and competitive position could be harmed. New test development involves a lengthy and complex process, and we may not be successful in our efforts to develop and commercialize our diagnostic and screening products and services. The further development and commercialization of additional diagnostic and screening product and service offering are key to our growth strategy. |
| · | Our insurance policies are expensive and protect us only from some business risks, which will leave us exposed to significant uninsured liabilities. |
| · | If we use hazardous materials in a manner that causes injury, we could be liable for damages. |
| · | If we are sued for product liability or errors and omissions liability, we could face substantial liabilities that exceed our resources. |
| · | Our ability to utilize our net operating loss carryforwards and certain other tax attributes may be limited. |
| · | We may be adversely affected by the current economic environment and future adverse economic environments. |
| Common Warrants | |
| Number of Common Warrants | 1,000,000 |
| Terms of Common Warrants: | Each common warrant is exercisable to purchase one-twenty-fifth of a share of common stock at an exercise price of $10.625 per share and is exercisable at any time after the date of issuance for five years from its date of issuance. To better understand the terms of the common warrants, you should carefully read the "Description of the Offered Securities" section of this prospectus. You should also read the form of common warrant, which is filed as an exhibit to the registration statement of which this prospectus forms a part. |
| Placement Agent Warrants | |
Number of Placement Agent Warrants | 50,000 |
| Terms of Pre-Funded Warrants: | Each placement agent warrant is exercisable to purchase one-twenty-fifth of a share of common stock at an exercise price of $12.50 per share and is exercisable at any time after the date of issuance for five years from its date of issuance. |
| Common stock outstanding before this offering: | 5,289,919 shares of common stock |
| Common stock outstanding after this offering: | 6,339,919 shares of common stock |
| Use of Proceeds: | Assuming that the warrants are exercised for cash, we will use the net proceeds of this offering for general corporate purposes, including working capital and product development, particularly development of our one to three hour antibiotic resistance diagnostic product. See "Use of Proceeds" on page 40 of this prospectus. |
| Risk Factors: | Investing in our securities involves a high degree of risk. See "Risk Factors" beginning on page 13 of this prospectus and the other information included or incorporated by reference in this prospectus. |
| Nasdaq Capital Market symbol: | "OPGN." There is no established trading market for the common warrants or the placement agent warrants, and we do not expect a trading market to develop. We will not list the common warrants or the placement agent warrants on any securities exchange or other trading market. Without a trading market, the liquidity of the common warrants or placement agent warrants will be extremely limited. |
| · | 220,102 shares of common stock issuable upon the exercise of outstanding options granted as of April 2, 2018, under our equity incentive plans at a weighted average exercise price of $20.88 per share; |
| · | 3,148,873 shares of common stock issuable upon the exercise of outstanding warrants, other than the warrants being offered by this prospectus, issued as of April 2, 2018, at a weighted average exercise price of $13.05 per share; |
| · | 500 shares of common stock issuable upon vesting of outstanding restricted stock units granted as of April 2, 2018; |
| · | 42,289 shares of common stock available for future issuance under our equity incentive plans as of April 2, 2018; |
| · | 1,000,000 shares of common stock issuable upon the exercise of common warrants the subject of this offering; and |
| · | 50,000 shares of common stock issuable upon exercise of placement agent warrants the subject of this offering. |
| · | being permitted to present only two years of audited financial statements and only two years of related Management's Discussion and Analysis of Financial Condition and Results of Operations in this prospectus; |
| · | not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, as amended; |
| · | reduced disclosure obligations regarding executive compensation in our periodic reports, proxy statements and registration statements; and |
| · | exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. |
Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| (In thousands, except per share data) | ||||||||
| Statements of Operation Data: | ||||||||
| Revenue | $ | 3,211 | $ | 4,026 | ||||
| Operating expenses: | ||||||||
| Cost of products sold | 1,613 | 1,659 | ||||||
Cost of services(1) | 520 | 631 | ||||||
Research and development(1) | 6,883 | 8,613 | ||||||
General and administrative(1) | 6,693 | 6,603 | ||||||
Sales and marketing(1) | 2,768 | 5,529 | ||||||
| Transaction expenses | - | - | ||||||
Total operating expenses(1) | 18,477 | 23,035 | ||||||
| Operating loss | (15,266 | ) | (19,009 | ) | ||||
| Interest and other (expense) income | (87 | ) | (6 | ) | ||||
| Interest expense | (233 | ) | (144 | ) | ||||
| Foreign currency transaction gains (losses) | 23 | (8 | ) | |||||
| Change in fair value of derivative financial instruments | 144 | - | ||||||
| Provision for income taxes | - | - | ||||||
| Net loss | $ | (15,419 | ) | $ | (19,167 | ) | ||
| Net loss available to common stockholders | $ | (15,419 | ) | $ | (19,499 | ) | ||
| Net loss per common share, basic and diluted | (9.80 | ) | (27.59 | ) | ||||
| Weighted average shares outstanding—basic and diluted | 1,574 | 707 | ||||||
Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Cost of services | $ | 13,776 | $ | 6,003 | ||||
| Research and development | 237,103 | 236,341 | ||||||
| General and administrative | 603,787 | 599,550 | ||||||
| Sales and marketing | 56,732 | 103,567 | ||||||
| Total stock-based compensation | $ | 911,398 | $ | 945,461 | ||||
| As of December 31, 2017 | ||||||||
| Actual | As Adjusted | |||||||
| (In thousands) | ||||||||
| Balance Sheet Data: | ||||||||
| Cash and cash equivalents | $ | 1,847 | $ | 13,097 | ||||
| Working capital (deficit) | (1,288 | ) | 9,962 | |||||
| Total assets | 6,620 | 17,870 | ||||||
| Accumulated deficit | (148,710 | ) | (137,460 | ) | ||||
| Total stockholders' equity | 1,401 | 12,651 | ||||||
| · | on an actual basis; |
| · | on an as adjusted basis, to give further effect to the issuance and sale in this offering of 1,050,000 shares of common stock upon the exercise of the common warrants at an exercise price of $10.625 per share and of the placement agent warrants at an exercise price of $12.50 per share. |
| · | developing our Acuitas AMR Gene Panel products and services for antibiotic resistance testing, and our automated rapid molecular diagnostic products; |
| · | commercializing our rapid pathogen identification and Acuitas MDRO and Acuitas Lighthouse informatics services; |
| · | developing, presenting and publishing additional clinical and economic utility data intended to increase clinician adoption of our current and future products and services; |
| · | expansion of our operating capabilities; |
| · | development of collaborative arrangements during 2018; |
| · | maintenance, expansion and protection of our intellectual property portfolio and trade secrets; |
| · | future clinical trials as we seek regulatory approval for some of our product offerings; |
| · | expansion of the size and geographic reach of our sales force and our marketing capabilities to commercialize potential future products and services; and |
| · | continued focus on recruiting and retaining our quality assurance and compliance personnel and activities. |
| · | our ability to convince the medical community of the clinical utility of our products and services and their potential advantages over existing tests, including our surveillance services offering, despite the lack of reimbursement for such services; |
| · | our ability to successfully develop automated rapid pathogen identification and antibiotic resistance testing products and services, including bioinformatics, and convince hospitals and other healthcare providers of the patient safety, improved patient outcomes and potential cost savings that could result; |
| · | our ability to grow our microbial isolate and antibiotic resistance genes knowledgebase; |
| · | our ability to convince the medical community of the accuracy and speed of our products and services, as contrasted with the current methods available; and |
| · | the willingness of hospitals and physicians to use our products and services. |
| · | collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| · | collaborators may not perform their obligations as expected; |
| · | we may not achieve any milestones, or receive any milestone payments, under our collaborations, including milestones and/or payments that we expect to achieve or receive; |
| · | the clinical trials, if any, conducted as part of these collaborations may not be successful; |
| · | a collaborator might elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator's strategic focus or available funding or external factors, such as an acquisition, that diverts resources or creates competing priorities; |
| · | we may not have access to, or may be restricted from disclosing, certain information regarding product or services candidates being developed or commercialized under a collaboration and, consequently, may have limited ability to inform our stockholders about the status of such product or services candidates; |
| · | collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive than ours; |
| · | product or services candidates developed in collaboration with us may be viewed by our collaborators as competitive with their own product or services, which may cause collaborators to cease to devote resources to the commercialization of our product or services candidates; |
| · | a collaborator with marketing and distribution rights to one or more of our product or services candidates that achieve regulatory approval may not commit sufficient resources to the marketing and distribution of any such product candidate; |
| · | disagreements with collaborators, including disagreements over proprietary rights, contract interpretation or the preferred course of development of any product or services candidates, may cause delays or termination of the research, development or commercialization of such product or services candidates, may lead to additional responsibilities for us with respect to such product or services candidates or may result in litigation or arbitration, any of which would be time-consuming and expensive; |
| · | collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our intellectual property or proprietary information or expose us to potential litigation; |
| · | disputes may arise with respect to the ownership of intellectual property developed pursuant to a collaboration; |
| · | collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability; and |
| · | collaborations may be terminated for the convenience of the collaborator and, if terminated, we could be required to raise additional capital to pursue further development or commercialization of the applicable product or services candidates. |
| · | failure of the test at the research or development stage; |
| · | lack of clinical validation data to support the effectiveness of the test; |
| · | delays resulting from the failure of third-party suppliers or contractors to meet their obligations in a timely and cost-effective manner; |
| · | failure to obtain or maintain necessary certifications, licenses, clearances or approvals to market or perform the test; or |
| · | lack of commercial acceptance by in-patient healthcare facilities. |
| · | our ability to grow our revenue and customer base; |
| · | the announcement of new products or product enhancements by us or our competitors; |
| · | developments concerning regulatory oversight and approvals; |
| · | variations in our and our competitors' results of operations; |
| · | changes in earnings estimates or recommendations by securities analysts, if our common stock is covered by analysts; |
| · | successes or challenges in our collaborative arrangements or alternative funding sources; |
| · | developments in the health care and life science industries; |
| · | the results of product liability or intellectual property lawsuits; |
| · | future issuances of common stock or other securities; |
| · | the addition or departure of key personnel; |
| · | announcements by us or our competitors of acquisitions, investments or strategic alliances; and |
| · | general market conditions and other factors, including factors unrelated to our operating performance. |
| · | required compliance with existing and changing foreign health care and other regulatory requirements and laws, such as those relating to patient privacy; |
| · | required compliance with anti-bribery laws, such as the U.S. Foreign Corrupt Practices Act, or FCPA, and U.K. Bribery Act, data privacy requirements, labor laws and anti- competition regulations; |
| · | export or import restrictions; |
| · | various reimbursement and insurance regimes; |
| · | laws and business practices favoring local companies; |
| · | longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain foreign legal systems; |
| · | political and economic instability; |
| · | potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements and other trade barriers; |
| · | foreign exchange controls; |
| · | difficulties and costs of staffing and managing foreign operations; and |
| · | difficulties protecting or procuring intellectual property rights. |
| · | the federal Anti-Kickback Statute, which constrains certain marketing practices, educational programs, pricing policies and relationships with healthcare providers or other entities by prohibiting, among other things, soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce or in return for, the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
| · | federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third party payors that are false or fraudulent; |
| · | federal physician self-referral laws, such as the Stark Law, which prohibit a physician from making a referral to a provider of certain health services with which the physician or the physician's family member has a financial interest, and prohibit submission of a claim for reimbursement pursuant to a prohibited referral; and |
| · | state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to items or services reimbursed by any third party payor, including commercial insurers, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. |
| · | the completion of our development efforts for the AMR Gene Panel for cUTI and Acuitas Lighthouse Software, and the timing of commercialization; |
| · | our ability to sustain or grow our customer base for our current products; |
| · | our liquidity and working capital requirements, including our cash requirements over the next 12 months; |
| · | our ability to maintain compliance with the ongoing listing requirements for the Nasdaq Capital Market; |
| · | anticipated trends and challenges in our business and the competition that we face; |
| · | the execution of our business plan and our growth strategy; |
| · | our expectations regarding the size of and growth in potential markets; |
| · | our opportunity to successfully enter into new collaborative agreements; |
| · | regulations and changes in laws or regulations applicable to our business, including regulation by the FDA; |
| · | compliance with the U.S. and international regulations applicable to our business; and |
| · | our expectations regarding future revenue and expenses. |
| · | completion of development of AMR Gene Panel for cUTI and Acuitas Lighthouse Knowledgebase; |
| · | support of acquisitions of products and technologies; |
| · | initial commercialization efforts for the AMR Gene Panel for cUTI and Acuitas Lighthouse Knowledgebase; and |
| · | the balance for general corporate purposes, such as general and administrative expenses, capital expenditures and working capital needs. |
| · | on an actual basis; |
| · | on an as adjusted basis, to give further effect to the issuance and sale in this offering of 1,050,000 shares of common stock upon the exercise of the common warrants at an exercise price of $10.625 per share and of the placement agent warrants at an exercise price of $12.50 per share. |
| As of December 31, 2017 | ||||||||
| Actual | As Adjusted | |||||||
| (In thousands, except share and per share data) | ||||||||
| (Unaudited) | ||||||||
| Cash and cash equivalents | $ | 1,847 | $ | 13,097 | ||||
| Short-term debt, net of discount | $ | 1,011 | $ | 1,011 | ||||
| Stockholder's (deficit) equity: | ||||||||
| Common stock, par value $0.01 per share: 200,000,000 shares authorized, 2,265,320 shares issued and outstanding, actual; 200,000,000 shares authorized, 3,315,320 issued and outstanding, as adjusted | 23 | 33 | ||||||
| Additional paid-in capital | 150,115 | 161,354 | ||||||
| Accumulated other comprehensive loss | (26 | ) | (26 | ) | ||||
| Accumulated deficit | (148,710 | ) | (148,710 | ) | ||||
| Total stockholders' equity | 1,402 | 12,651 | ||||||
| Total capitalization | $ | 2,412 | $ | 13,662 | ||||
| · | 5,400 shares of common stock issued upon vesting of restricted stock units since December 31, 2017; |
| · | 178,087 shares of common stock issued pursuant to the exercise of pre-funded warrants since December 31, 2017; |
| · | 139,395 shares of common stock issuable upon the exercise of outstanding options granted as of December 31, 2017, under our equity incentive plans at a weighted average exercise price of $31.16 per share; |
| · | 2,841,152 shares of common stock issued to investors as part of our February 2018 Public Offering; |
| · | 445,028 shares of common stock issuable upon the exercise of outstanding warrants issued as of December 31, 2017, at a weighted average exercise price of $77.15 per share; |
| · | 500 shares of common stock issuable upon vesting of outstanding restricted stock units granted as of December 31, 2017; |
| · | 32,411 shares of common stock available for future issuance under our equity incentive plans as of December 31, 2017; |
| · | 1,846,153 shares of common stock issuable upon the exercise of common warrants issued to investors in the February 2018 Public Offering at an exercise price of $3.25 per share; |
| · | 673,077 shares of common stock issuable upon the exercise of pre-funded warrants issued to investors in the February 2018 Public Offering at an exercise price of $0.01 per share; |
| · | 184,615 shares of common stock issuable upon exercise of common warrants issued to investors in the February 2018 Public Offering at an exercise price of $4.0625 per share; and |
| · | 31 fractional shares cashed out as a result of the reverse stock split. |
| Weighted average exercise price of warrant shares | $ | 10.71 | ||||||
| Net tangible book value per share of as December 31, 2017 | $ | (0.24 | ) | |||||
| Increase in net tangible book value per share attributable to this offering | $ | 3.47 | ||||||
| As adjusted net tangible book value per share as of December 31, 2017, after giving effect to this offering | $ | 3.23 | ||||||
| Dilution per share to new investors purchasing our common stock in this offering | $ | 7.48 | ||||||
| · | 5,400 shares of common stock issued upon vesting of restricted stock units since December 31, 2017; |
| · | 178,087 shares of common stock issued pursuant to the exercise of pre-funded warrants since December 31, 2017; |
| · | 139,395 shares of common stock issuable upon the exercise of outstanding options granted as of December 31, 2017, under our equity incentive plans at a weighted average exercise price of $31.16 per share; |
| · | 2,841,152 shares of common stock issued to investors as part of our February 2018 Public Offering; |
| · | 445,028 shares of common stock issuable upon the exercise of outstanding warrants issued as of December 31, 2017, at a weighted average exercise price of $77.15 per share; |
| · | 500 shares of common stock issuable upon vesting of outstanding restricted stock units granted as of December 31, 2017; |
| · | 32,411 shares of common stock available for future issuance under our equity incentive plans as of December 31, 2017; |
| · | 1,846,153 shares of common stock issuable upon the exercise of common warrants issued to investors in the February 2018 Public Offering at an exercise price of $3.25 per share; |
| · | 673,077 shares of common stock issuable upon the exercise of pre-funded warrants issued to investors in the February 2018 Public Offering at an exercise price of $0.01 per share; |
| · | 184,615 shares of common stock issuable upon exercise of common warrants issued to investors in the February 2018 Public Offering at an exercise price of $4.0625 per share; and |
| · | 31 fractional shares cashed out as a result of the reverse stock split. |
| High | Low | |||||||
Common Stock: | ||||||||
| Year Ended December 31, 2016 | ||||||||
| First Quarter | $ | 49.20 | $ | 34.00 | ||||
| Second Quarter | $ | 54.50 | $ | 25.50 | ||||
| Third Quarter | $ | 116.25 | $ | 34.00 | ||||
| Fourth Quarter | $ | 77.50 | $ | 21.75 | ||||
IPO Warrants: | ||||||||
| Year Ended December 31, 2016 | ||||||||
| First Quarter | $ | 9.25 | $ | 4.61 | ||||
| Second Quarter | $ | 11.50 | $ | 2.80 | ||||
| Third Quarter | $ | 17.00 | $ | 3.13 | ||||
| Fourth Quarter | $ | 7.48 | $ | 2.01 | ||||
| High | Low | |||||||
Common Stock: | ||||||||
| Year Ended December 31, 2017 | ||||||||
| First Quarter | $ | 46.25 | $ | 24.50 | ||||
| Second Quarter | $ | 31.25 | $ | 13.00 | ||||
| Third Quarter | $ | 16.76 | $ | 5.30 | ||||
| Fourth Quarter | $ | 10.25 | $ | 3.78 | ||||
IPO Warrants: | ||||||||
| Year Ended December 31, 2017 | ||||||||
| First Quarter | $ | 4.50 | $ | 1.50 | ||||
| Second Quarter | $ | 10.00 | $ | 0.88 | ||||
| Third Quarter | $ | 4.88 | $ | 1.50 | ||||
| Fourth Quarter | $ | 2.10 | $ | 0.25 | ||||
| High | Low | |||||||
Common Stock: | ||||||||
| Year Ending December 31, 2018 | ||||||||
| First Quarter | $ | 7.60 | $ | 1.63 | ||||
Second Quarter (through April 2, 2018) | $ | 1.74 | $ | 1.63 | ||||
IPO Warrants: | ||||||||
| Year Ended December 31, 2018 | ||||||||
| First Quarter | $ | 1.37 | $ | 0.12 | ||||
Second Quarter (through April 2, 2018) | $ | 0.50 | $ | 0.50 | ||||
| · | before the stockholder became interested, our Board of Directors approved either the business combination or the transaction which resulted in the stockholder becoming an interested stockholder; |
| · | upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, shares owned by persons who are directors and also officers, and employee stock plans, in some instances, but not the outstanding voting stock owned by the interested stockholder; or |
| · | at or after the time the stockholder became interested, the business combination was approved by our Board of Directors and authorized at an annual or special meeting of the stockholders by the affirmative vote of at least two-thirds of the outstanding voting stock which is not owned by the interested stockholder. |
| · | any merger or consolidation involving the corporation and the interested stockholder; |
| · | any sale, transfer, lease, pledge or other disposition involving the interested stockholder of 10% or more of the assets of the corporation; |
| · | subject to exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder; |
| · | subject to exceptions, any transaction involving the corporation that has the effect of increasing the proportionate share of the stock of any class or series of the corporation beneficially owned by the interested stockholder; and |
| · | the receipt by the interested stockholder of the benefit of any loans, advances, guarantees, pledges or other financial benefits provided by or through the corporation. |
| · | ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| · | block trades in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
| · | purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| · | an exchange distribution in accordance with the rules of the applicable exchange; |
| · | privately negotiated transactions; |
| · | settlement of short sales entered into after the effective date of the registration statement of which this prospectus is a part; |
| · | broker-dealers may agree with the holders to sell a specified number of such shares at a stipulated price per share; |
| · | through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| · | a combination of any such methods of sale; or |
| · | any other method permitted pursuant to applicable law (including underwritten transactions). |
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
| · | The Company's Acuitas DNA tests provide rapid microbial identification and antibiotic resistance gene information. These products include the Acuitas AMR Gene Panel u5.47 for complicated urinary tract infections in development as a clinical diagnostic test and available for Research Use Only, the QuickFISH® family of FDA-cleared and CE-marked diagnostics used to rapidly detect pathogens in positive blood cultures, and the Acuitas Resistome Tests for genetic analysis of hospital surveillance isolates. |
| · | The Company's Acuitas Lighthouse informatics systems are cloud-based HIPAA compliant informatics offerings that combine clinical lab test results with patient and hospital information to provide analytics and actionable insights to help manage MDROs in the hospital and patient care environment. |
| · | On July 18, 2017, the Company closed its July 2017 Public Offering of 18,164,195 units at $0.40 per unit, and 6,835,805 pre-funded units at $0.39 per pre-funded unit, raising gross proceeds of approximately $10 million and net proceeds of approximately $8.8 million. jVen Capital, an affiliate of Evan Jones, the Company's Chairman of the Board and Chief Executive Officer, and three employees of the Company participated in the July 2017 Public Offering. Each unit included one twenty-fifth of a share of common stock and one common warrant to purchase one twenty-fifth of a share of common stock at an exercise price of $10.625 per share. Each pre-funded unit included one pre-funded warrant to purchase one twenty-fifth of a share of common stock for an exercise price of $0.25 per share, and one common warrant to purchase one twenty-fifth of a share of common stock at an exercise price of $10.625 per share. The common warrants are exercisable immediately and have a five-year term from the date of issuance. Approximately $1 million of the gross proceeds was used to repay the outstanding Bridge Financing Notes to jVen Capital in July 2017. |
| · | On May 31, 2017, the Company entered into a Note Purchase Agreement with jVen Capital, under which jVen Capital agreed to provide bridge financing in an aggregate principal amount of up to $1,500,000 to the Company in up to three separate tranches of bridge financing notes, each a Bridge Financing Note. The interest rate on each Bridge Financing Note was ten percent (10%) per annum (subject to increase upon an event of default). In connection with the Bridge Financing Notes, the Company issued jVen Capital stock purchase warrants to acquire 5,633 shares with an exercise price of $19.50 per share, and stock purchase warrants to acquire 6,349 shares at an exercise price of $17.25 per share. On June 14, 2017, the Company drew down on the first of three Bridge Financing Notes, with $1 million remaining capacity available. The Company drew down on the second Bridge Financing Note on July 5, 2017 and the third Bridge Financing Note was never issued. The outstanding Bridge Financing Notes were repaid in full upon the closing of the July 2017 Public Offering. |
| · | On June 6, 2017, as amended on June 28, 2017, the Company issued the amended and restated MGHIF Note to MGHIF, which extended the maturity date of the MGHIF Note from July 14, 2017 to July 14, 2018. In return for MGHIF's consent to such extension, the Company increased the interest rate of the MGHIF Note to 10% per annum and issued warrants to purchase shares of common stock to MGHIF equal to 20% of the principal balance of the MGHIF Note, plus interest accrued thereon, as of June 28, 2017. |
| · | During the year ended December 31, 2017, the Company sold approximately 227 thousand shares of its common stock under its at the market offering resulting in aggregate net proceeds to the Company of approximately $3.8 million, and gross proceeds of $4.0 million. |
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Product sales | $ | 2,771,869 | $ | 3,524,178 | ||||
| Laboratory services | 41,960 | 228,904 | ||||||
| Collaboration revenue | 397,178 | 272,603 | ||||||
| Total revenue | $ | 3,211,007 | $ | 4,025,685 | ||||
| · | Product Sales: the decrease in revenue of 21% in 2017 as compared to 2016 is primarily attributable to a reduction in the sale of our legacy Argus Whole Genome Mapping System, or Argus, products, as we transitioned from our legacy mapping products to the introduction of Acuitas MDRO products sales, and a reduction in the sale of our rapid pathogen ID testing products; |
| · | Laboratory Services: the decrease in revenue of 82% in 2017 as compared to 2016 is a result of decreases in sales of our Acuitas MDRO test products; and |
| · | Collaboration Revenue: the increase in collaboration revenue of 46% in 2017 as compared to 2016 is primarily the result of increased revenue associated with our CDC contract. |
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Cost of products sold | $ | 1,612,838 | $ | 1,658,571 | ||||
| Cost of services | 520,338 | 631,333 | ||||||
| Research and development | 6,883,293 | 8,613,236 | ||||||
| General and administrative | 6,692,659 | 6,602,608 | ||||||
| Sales and marketing | 2,767,670 | 5,529,274 | ||||||
| Total operating expenses | $ | 18,476,798 | $ | 23,035,022 | ||||
| · | Costs of products sold: expenses for the year ended December 31, 2017 decreased approximately 3% when compared to the same period in 2016. The change in costs of products sold is primarily attributable to the decrease in legacy Argus sales offset by increased payroll and facility costs; |
| · | Costs of services: expenses for the year ended December 31, 2017 decreased approximately 18% when compared to the same period in 2016. The change in costs of services is primarily attributable to a decrease in sales of Acuitas Lighthouse services; |
| · | Research and development: expenses for the year ended December 31, 2017 decreased approximately 20% when compared to the same period in 2016, primarily due to a decrease in costs related to the automated rapid pathogen identification project; |
| · | General and administrative: expenses for the year ended December 31, 2017 increased approximately 1% when compared to the same period in 2016, primarily due to legal costs; and |
| · | Sales and marketing: expenses for the year ended December 31, 2017 decreased approximately 50% when compared to the same period in 2016, primarily due to costs associated with marketing studies conducted in the first half of 2016 and the reductions in the size of our commercial organization that occurred in June through December 2017. |
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Interest expense | $ | (233,505 | ) | $ | (143,347 | ) | ||
| Foreign currency transaction gains/(losses) | 23,179 | (8,102 | ) | |||||
| Change in fair value of derivative financial instruments | 144,064 | - | ||||||
| Interest and other expense | (87,255 | ) | (5,967 | ) | ||||
| Total other expense | $ | (153,517 | ) | $ | (157,416 | ) | ||
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Net cash used in operating activities | $ | (14,303,880 | ) | $ | (17,250,637 | ) | ||
| Net cash used in investing activities | (276,950 | ) | (123,514 | ) | ||||
| Net cash provided by financing activities | 12,348,194 | 13,664,690 | ||||||
| · | Our Acuitas DNA tests provide rapid microbial identification and antibiotic resistance gene information. These products include our Acuitas antimicrobial resistance, or AMR, Gene Panel u5.47 for complicated urinary tract infections in development as a clinical diagnostic test and available for Research Use Only, the QuickFISH® family of FDA-cleared and CE-marked diagnostics used to rapidly detect pathogens in positive blood cultures, and our Acuitas Resistome Tests for genetic analysis of hospital surveillance isolates. |
| · | Our Acuitas Lighthouse informatics systems are cloud-based HIPAA compliant informatics offerings that combine clinical lab test results with patient and hospital information to provide analytics and actionable insights to help manage MDROs in the hospital and patient care environment. Components of our informatics systems include the Acuitas Lighthouse Knowledgebase and the Acuitas Lighthouse Software. The Acuitas Lighthouse Knowledgebase is a relational database management system and a proprietary data warehouse of genomic data matched with antibiotic susceptibility information for bacterial pathogens. The Acuitas Lighthouse Software system includes the Acuitas Lighthouse Portal, a suite of web applications and dashboards, the Acuitas Lighthouse Prediction Engine, which is a data analysis software, and other supporting software components. The Acuitas Lighthouse Software can be customized and made specific to a healthcare facility or collaborator, such as a pharmaceutical company. |

| · | Rapid diagnostics – We are developing OpGen-branded Acuitas AMR Gene Panel tests for use on the Thermo Fisher Scientific Applied Biosystems™ QuantStudio™ 5 Real-Time PCR System. The first of these new tests will be for management of patients with cUTI. We anticipate developing tests for additional clinical indications such as lower respiratory tract infections and for new antibiotic decision- making applications. The second rapid diagnostics growth driver will be through strategic partner relationships where we will work to expand channel access for our proprietary DNA tests through development and subsequent use of these tests, utilizing the Acuitas Lighthouse Software on established rapid in vitro diagnostic testing platforms. |
| · | Acuitas Lighthouse informatics and services – We are pursuing commercial opportunities to provide our Acuitas Lighthouse informatics and companion genomic testing to pharmaceutical companies and CROs, health systems, third party in vitro diagnostic companies, and government agencies. Through our Pharmaceutical/CRO services we are working to help accelerate clinical trials and new product launches and to establish early access for diagnostic tests to help guide decision-making for new antibiotics. Our focus in the health system segment is on helping guide antibiotic decision-making and supporting patient safety initiatives. We are actively pursuing government funding for development and deployment of our Acuitas Lighthouse informatics in the United States and internationally. |
| · | complete development, clinical evaluations, obtain necessary regulatory approvals, and successfully commercialize our AMR Gene Panel u5.47 for cUTI with a goal of achieving three-hour antibiotic resistance analysis from the time of specimen collection; |
| · | continue clinical evaluations for the AMR Gene Panel u5.47 in the first half of 2018; in January 2018 we introduced the AMR Gene Panel u5.47 as an RUO test and have a goal of making a FDA 510(k) submission in the fourth quarter of 2018 to support commercial launch; |
| · | obtain third party funding to expand our AMR Gene Panel development and access to additional third party rapid testing platforms; |
| · | expand our business collaborations with Merck and other pharmaceutical companies; |
| · | capitalize on opportunities to deploy our Acuitas Lighthouse informatics and genomic testing for pharmaceutical/CRO services; |
| · | grow our Acuitas Lighthouse data warehouse offerings for resistance and susceptibility data in hospital, hospital system, or broader community applications through continued development of the Acuitas Lighthouse Knowledgebase; |
| · | seek government funding to advance programs focused on identification and treatment of MDROs; and |
| · | continue development of our Acuitas Lighthouse Software and work to install Acuitas Lighthouse Software to customer sites in the United States and globally. |


| QuickFISH | PNA FISH |
| Staphylococcus | Staphylococcus |
| Enterococcus | Enterococcus |
| Gram-negative bacteria | Gram-negative bacteria |
| Candida | Candida |
| · | Our Acuitas MDRO Gene Test, launched in 2014, is, to our knowledge, the first CLIA lab-based test able to provide information regarding the presence of ten MDRO resistance genes from one patient specimen. The ten drug-resistant genes identified by our Acuitas MDRO Gene Test are associated with CRE, ESBL and VRE organisms, and are gastrointestinal organisms frequently associated with antibiotic-resistant infections. The test results can be used by healthcare providers to identify patients colonized with organisms expressing the drug-resistant genes or who are actively infected. |
| · | Our Acuitas CR Elite Test, launched in 2014, adds the ability for the healthcare provider to order a microbiology culture screen to be performed from the same specimen sent for our Acuitas MDRO Gene Test, thereby providing additional information about the organism(s) associated with an active infection, as well as an antibiotic susceptibility profile for such organism(s). |
| · | Our Acuitas Resistome Test, launched in the second quarter of 2015, is a more comprehensive MDRO molecular test which detects 49 genes covering over 900 subtypes associated with antibiotic resistance. The test includes additional resistance genes for carbapenemases, ESBLs and AmpC genes, in replacement of the Vancomycin resistant genes found in the Acuitas MDRO Gene Test. We use Acuitas Resistome Test results for Acuitas Lighthouse profiling of specimens collected in hospitals and clinical isolates from infected patients. Information from our Acuitas Resistome Test provides additional gene detection information to supplement our Acuitas MDRO Gene Test. |
| · | development of the Acuitas AMR Gene Panel tests for additional indications, and clinical trial work to support a 510(k) application for commercial launch of the Acuitas AMR Gene Panel 5u.47; |
| · | continued investments in our Acuitas Lighthouse informatics platform, focused on (i) data warehouse and portal for MDRO data and (ii) antibiotic analysis; |
| · | expanding our clinical decision support capabilities by completing the work under the CDC contract to develop smartphone-based clinical decision support solutions for antimicrobial stewardship and infection control in low- and middle-income countries; |
| · | working with pharmaceutical companies to add new or recently FDA approved antibiotics to the Acuitas Lighthouse Software |
| · | further development of our Acuitas MDRO Gene Test, Acuitas Resistome Test and Acuitas Whole Genome Sequence Analysis; and |
| · | converting our CLIA lab-based products to IVD kits that can be sold, upon receipt of FDA clearance and other approvals, directly to our customers and to other clinical reference laboratories. |
| · | product listing and establishment registration, which helps facilitate FDA inspections and other regulatory action; |
| · | QSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the development and manufacturing process; |
| · | labeling regulations and FDA prohibitions against the promotion of products for uncleared, unapproved or off-label use or indication; |
| · | clearance of product modifications that could significantly affect safety or efficacy or that would constitute a major change in intended use of one of our cleared devices; |
| · | approval of product modifications that affect the safety or effectiveness of one of our cleared devices; |
| · | medical device reporting regulations, which require that manufacturers comply with FDA requirements to report if their device may have caused or contributed to a death or serious injury, or has malfunctioned in a way that would likely cause or contribute to a death or serious injury if the malfunction of the device or a similar device were to recur; |
| · | post-approval restrictions or conditions, including post-approval study commitments; |
| · | post-market surveillance regulations, which apply when necessary to protect the public health or to provide additional safety and effectiveness data for the device; |
| · | the FDA's recall authority, whereby it can ask, or under certain conditions order, device manufacturers to recall from the market a product that is in violation of governing laws and regulations; |
| · | regulations pertaining to voluntary recalls; and |
| · | notices of corrections or removals. |
| · | denial of payment for the services provided in violation of the prohibition; |
| · | refunds of amounts collected by an entity in violation of the Stark Law; |
| · | a civil penalty of up to $15,000 for each service arising out of the prohibited referral; |
| · | possible exclusion from Federal healthcare programs, including Medicare and Medicaid; and |
| · | a civil penalty of up to $100,000 against parties that enter into a scheme to circumvent the Stark Law's prohibition. |
| Name | Age | Position |
Directors | ||
| Evan Jones | 60 | Chief Executive Officer, Director and Chairman of the Board |
| Harry J. D'Andrea | 61 | Director |
| Timothy J.R. Harris, Ph.D., D.Sc | 67 | Director |
| Tina S. Nova, Ph.D. | 64 | Director |
| David M. Rubin, Ph.D. | 52 | Director |
| Misti Ushio, Ph.D. | 46 | Director |
Other Executive Officers | ||
| Timothy C. Dec | 59 | Chief Financial Officer and Corporate Secretary |
| Vadim Sapiro | 47 | Chief Information Officer |
| Named Executive Officer and Principal Position | Year | Salary ($) | Bonus ($) | Stock Awards (1)($) | Option Awards (1)($) | Non-Equity Incentive Plan Compensation (2)($) | All Other Compensation ($) | Total ($) | |||||||||||||||||||||
| Evan Jones | 2017 | $ | 375,962 | $ | - | $ | - | $ | 43,210 | $ | - | $ | - | $ | 419,172 | ||||||||||||||
| Chief Executive Officer | 2016 | $ | 316,538 | $ | - | $ | - | $ | 499,352 | $ | - | $ | - | $ | 815,890 | ||||||||||||||
| Timothy Dec | 2017 | $ | 291,102 | $ | - | $ | 10,325 | $ | 45,580 | $ | - | $ | - | $ | 347,007 | ||||||||||||||
| Chief Financial Officer | 2016 | $ | 273,462 | $ | - | $ | - | $ | 44,996 | $ | - | $ | - | $ | 318,458 | ||||||||||||||
| Vadim Sapiro | 2017 | $ | 291,102 | $ | - | $ | 10,325 | $ | 38,559 | $ | 50,000 | $ | - | $ | 389,986 | ||||||||||||||
| Chief Information Officer | 2016 | $ | 280,385 | $ | - | $ | - | $ | 29,997 | $ | - | $ | - | $ | 310,882 | ||||||||||||||
| (1) | The "Stock Awards" column reflects the grant date fair value for all restricted stock units awarded under the 2015 Plan during 2017 and 2016. The "Option Awards" column reflects the grant date fair value for all stock option awards granted under the 2015 Plan or the 2008 Plan during 2017 and 2016, respectively, except for Mr. Jones – the 2016 stock option grant was made outside of the 2015 Plan, subject to stockholder approval that was obtained on June 22, 2016. These amounts are determined in accordance with FASB Accounting Standards Codification 718 (ASC 718), without regard to any estimate of forfeiture for service vesting. Assumptions used in the calculation of the amounts in these columns for 2017 and 2016 will be included in a footnote to the Company's consolidated audited financial statements for the year ended December 31, 2017. |
| (2) | On March 7, 2018, the Board upon the recommendation of the Compensation Committee approved the 2017 bonuses for the executive officers of the Company, including Vadim Sapiro, Chief Information Officer of the Company. During 2017, the Compensation Committee recommended, and the Board approved, a short-term annual cash bonus plan for employees, including the named executive officers (the "2017 Bonus Plan"). The 2017 Bonus Plan had six general corporate goals and a bonus pool funding mechanism. The general corporate goals related to: (1) successful completion of development goals for the Acuitas AMR Gene Panel and Acuitas Lighthouse products and services in development; (2) receipt of third party collaboration or partnering arrangement funding from third parties; (3) commercial traction for the Company's Acuitas AMR Gene Panel and Acuitas Lighthouse products and services in development; (4) consummation of capital raising transaction or transactions; (5) pursuit of strategic technology acquisitions and strengthening the management team; and (6) implementation of a restructuring plan to consolidate the Company's operations in its Gaithersburg MD facility. The bonus pool funding mechanism was designed to fund the pool only if annual revenues for 2017 exceeded a $3.0 million threshold. The Committee determined that the bonus pool funding was achieved at a level above threshold and that the corporate goals were achieved at 73% of target. Management recommended, and the Committee approved, payment of bonuses under the 2017 Bonus Plan at 55% of target. After discussion with the Committee and the Board, each of Mr. Jones and Mr. Dec agreed to forego receipt of a bonus under the 2017 Bonus Plan given the loss of shareholder value during 2017 despite the operational progress, to allow the Company to retain the cash to advance its operations. A bonus of $50,000, representing 55% of target, was earned by Mr. Sapiro under the 2017 Bonus Plan. |
| OPTION AWARDS | STOCK AWARDS | ||||||||
| Name | (1) Number of Securities Underlying Unexercised Options Exercisable | (1) Number of Securities Underlying Unexercised Options Unexercisable | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options | Option Exercise Price ($) | Option Expiration Date | Number of Shares of Stock that have not Vested | Market Value of Shares of Stock that have not Vested ($) | Equity Incentive Plan Awards: Number of Unearned Shares, Units or Other Rights that have not Vested | Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or other Rights that have not Vested ($) (2) |
| Evan Jones(3) | 3 | - | - | 1,976.25 | 7/23/2018 | - | - | - | |
| 73 | - | - | 2,767.00 | 9/21/2020 | - | - | - | - | |
| 6,097 | 872 | - | 1.25 | 4/24/2024 | - | - | - | - | |
| 6,500 | 1,500 | - | 15.25 | 10/23/2024 | - | - | - | - | |
| 15,330 | 15,330 | - | 33.75 | 4/28/2026 | - | - | - | - | |
| 1,000 | 2,200 | - | 25.75 | 2/23/2027 | - | - | - | - | |
| Timothy Dec(4) | 3,143 | 1,429 | - | 150.00 | 5/4/2025 | - | - | 1,900 | 8,892 |
| 1,125 | 875 | - | 42.50 | 11/10/2025 | - | - | - | - | |
| 1,050 | 1,350 | - | 38.75 | 6/13/2026 | - | - | - | - | |
| - | 2,720 | - | 25.75 | 2/23/2027 | - | - | - | - | |
| - | 2,400 | - | 7.375 | 8/9/2027 | - | - | - | - | |
| Vadim Sapiro(5) | 2 | - | - | 197.75 | 3/23/2022 | - | - | 1,400 | 6,552 |
| 36 | - | - | 197.75 | 3/23/2022 | - | - | - | - | |
| 10 | - | - | 197.75 | 2/12/2023 | - | - | - | - | |
| 5 | - | - | 197.75 | 2/12/2023 | - | - | - | - | |
| 25 | - | - | 197.75 | 7/25/2023 | - | - | - | - | |
| 143 | - | - | 1.25 | 4/24/2024 | - | - | - | - | |
| 1,500 | 500 | - | 15.25 | 10/23/2024 | - | - | - | - | |
| 1,000 | - | - | 150.00 | 5/4/2025 | - | - | - | - | |
| 700 | 900 | - | 38.750 | 6/13/2026 | - | - | - | - | |
| - | 2,200 | - | 25.75 | 2/23/2027 | - | - | - | - | |
| - | 2,400 | - | 7.375 | 8/9/2027 | - | - | - | - | |
| (1) | The standard vesting schedule for all stock option grants is vesting over four years with twenty-five percent (25%) vesting on the first anniversary of the date of grant and six and one-quarter percent (6.25%) vesting on the last day of the next fiscal quarter over three years. |
| (2) | Calculated based on the closing price of the common stock the Nasdaq Capital Market on December 29, 2017 of $4.68 per share. |
| (3) | The stock option awards made to Mr. Jones were awarded on July 23, 2008 (3 shares), February 15, 2011 (73 shares), April 24, 2014 (6,969 shares), October 23, 2014 (8,000 shares) and April 28, 2016 (30,660 shares) and have the vesting schedule set forth in footnote (1). Mr. Jones was granted a stock option award on February 23, 2017 (3,200), which vests over four years with twenty-five percent (25%) vesting on February 23, 2018 and six and one-quarter percent (6.25%) vesting on the first business day of each quarter thereafter over the next three years. |
| (4) | Mr. Dec was granted stock option awards on May 4, 2015 (4,572 shares), November 10, 2015 (2,000 shares), June 13, 2016 (2,400 shares), February 23, 2017 (2,720), and August 9, 2017 (2,400). One-forty-eighth of Mr. Dec's stock option award granted on May 4, 2015 vested on the one month anniversary of the date of grant and thereafter vest over four years with twenty-five percent (25%) vesting on the first yearly anniversary of the date of grant and six and one-quarter percent (6.25%) vesting on the last day of the next fiscal quarter over three years. Mr. Dec's stock option awards granted on November 10, 2015 and June 13, 2016 have the vesting schedule set forth in footnote (1). Mr. Dec's stock option award granted on February 23, 2017 vests over four years with twenty-five percent (25%) vesting on February 23, 2018 and six and one-quarter percent (6.25%) vesting on the first business day of each quarter thereafter over the next three years. Mr. Dec's stock option award granted on August 9, 2017 vests on August 9, 2018. Mr. Dec was granted restricted stock units on November 10, 2015. Twenty-five percent (25%) of the entire restricted stock units award vests on the first four anniversaries of the date of grant. Mr. Dec was granted restricted stock units on August 9, 2017. The restricted stock units vest upon the successful launch of the Company's AMR Gene Panel for cUTI in the RUO market. |
| (5) | The stock option awards granted to Mr. Sapiro on March 23, 2012 (2 shares and 36 shares), February 12, 2013 (10 shares), July 25, 2013 (25 shares), October 23, 2014 (2,000 shares) and June 13, 2016 (1,600 shares) have the vesting schedule set forth in footnote (1). The stock option award granted to Mr. Sapiro on February 12, 2013 for 5 shares vested in full on the first anniversary of the date of grant, February 12, 2014. The stock option award granted to Mr. Sapiro on April 24, 2014 for 143 shares is vesting over four years with twenty-five percent (25%) vesting on December 31, 2014 and six and one-fourth percent (6.25%) vesting quarterly thereafter in equal proportions over the remaining three years. The stock option granted to Mr. Sapiro on May 4, 2015 vested quarterly over the first year following the date of grant. The stock option award granted to Mr. Sapiro on February 23, 2017 for 2,200 shares vest over four years with twenty-five percent (25%) vesting on February 23, 2018 and six and one-quarter percent (6.25%) vesting on the first business day of each quarter over the next three years. The stock option award granted to Mr. Sapiro on August 9, 2017 for 2,400 shares vests on August 9, 2018. Mr. Sapiro was granted restricted stock units on August 9, 2017. The restricted stock units vest upon the successful launch of the Company's AMR Gene Panel for cUTI in the RUO market. |
| Name | Fees Earned or Paid in Cash ($) | Option Awards ($)(1) | All Other Compensation ($) | Total ($) | ||||||||||||
| Brian G. Atwood (2) (4) | $ | 16,000 | $ | - | $ | - | $ | 16,000 | ||||||||
| Harry J. D'Andrea (4) | $ | 40,000 | $ | 13,632 | $ | - | $ | 53,632 | ||||||||
| Timothy J.R. Harris (4) | $ | 33,500 | $ | 13,632 | $ | - | $ | 47,132 | ||||||||
| Laurence R. McCarthy (2) (4) | $ | 18,750 | $ | - | $ | - | $ | 18,750 | ||||||||
| Tina S. Nova (4) | $ | 20,417 | $ | 16,173 | $ | - | $ | 36,590 | ||||||||
| David M. Rubin (3) | $ | - | $ | - | $ | - | $ | - | ||||||||
| Misti Ushio (4) | $ | 40,250 | $ | 13,632 | $ | - | $ | 53,882 | ||||||||
| (1) | The "Option Awards" column reflects the grant date fair value for all stock option awards granted under the 2015 Plan during 2016. These amounts are determined in accordance with FASB Accounting Standards Codification 718 (ASC 718), without regard to any estimate of forfeiture for service vesting. Assumptions used in the calculation of the amounts will be included in a footnote to the Company's consolidated audited financial statements for the year ended December 31, 2017. |
| (2) | Mr. Atwood and Dr. McCarthy did not stand for re-election at the 2017 Annual Meeting of the Stockholders. |
| (3) | As managing director of MGHIF, Dr. Rubin is precluded from receiving compensation for serving as a director of OpGen, Inc. |
| (4) | As of December 31, 2017, the non-employee directors held the following vested stock options: Atwood (500); D'Andrea (1,150); Harris (1,031); McCarthy (2,182); Nova (250); and Ushio (750). |
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights(1) | Weighted average exercise price of outstanding options, warrants and rights(2) | Number of securities remaining available for future issuance | |||||||||
| Equity compensation plans approved by security holders | 145,295 | $ | 31.16 | 32,411 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 145,295 | $ | 31.16 | 32,411 | ||||||||
| · | the amounts involved exceeded or will exceed the lesser of $120,000 or one percent of the average of the Company's total assets at year end for the past two completed fiscal years; and |
| · | any of our directors, executive officers or holders of more than 5% of our capital stock, or any member of the immediate family of the foregoing persons, had or will have a direct or indirect material interest. |
| Name and Address of Beneficial Owner | Number of Shares of Common Stock | Percentage of Outstanding Common Shares | ||||||
| Directors and Named Executive Officers | ||||||||
| Evan Jones (1) | 397,410 | 4.4 | % | |||||
| Harry D'Andrea (2) | 3,371 | * | ||||||
| Timothy J.R. Harris, Ph.D., D.Sc. (3) | 7,730 | * | ||||||
| Tina S. Nova, Ph.D.(4) | 900 | * | ||||||
| David M. Rubin, Ph.D. (5) | - | - | ||||||
| Misti Ushio, Ph.D. (6) | 2,571 | * | ||||||
| Timothy C. Dec (7) | 17,208 | * | ||||||
| Vadim Sapiro (8) | 8,630 | * | ||||||
| All current Directors and Executive Officers as a group (8 individuals) (9) | 437,941 | 8.0 | % | |||||
| (1) | Consists of (i) 227,138 shares of common stock and currently exercisable warrants to acquire an additional 134,317 shares of common stock beneficially owned by jVen Capital, LLC, (ii) 5,246 shares of common stock and currently exercisable warrants to acquire an additional 834 shares of common stock owned by Mr. Jones' spouse, and (iii) stock options to purchase 29,875 shares of common stock that are currently vested or that will become vested within 60 days. Mr. Jones is a managing member of jVen Capital, LLC and has voting and investment authority over the shares owned by that entity. |
| (2) | Consists of (i) 1,571 shares of common stock and (ii) stock options to purchase 1,800 shares of common stock that are currently vested or that will become vested within 60 days. |
| (3) | Consists of (i) 2,587 shares of common stock, (ii) currently exercisable warrants to acquire an additional 1,567 shares of common stock, and (iii) stock options to purchase 2,606 shares of common stock that are currently vested or that will become vested within 60 days. |
| (4) | Consists of stock options to purchase 900 shares of common stock that are currently vested or that will become vested within 60 days. |
| (5) | Dr. Rubin is the managing director of MGHIF, but does not have nor share voting power over the shares of our common stock owned by MGHIF. |
| (6) | Consists of (i) 1,571 shares of common stock and (ii) stock options to purchase 1,000 shares of common stock that are currently vested or that will become vested within 60 days. |
| (7) | Consists of (i) 6,408 shares of common stock, (ii) currently exercisable warrants to acquire an additional 4,071 shares of common stock, and (iii) stock options to purchase 6,729 shares of common stock that are currently vested or that will become vested within 60 days. |
| (8) | Consists of (i) 3,004 shares of common stock, (ii) currently exercisable warrants to acquire an additional 1,393 shares of common stock, and (iii) stock options to purchase 4,233 shares of common stock that are currently vested or that will become vested within 60 days. |
| (9) | See the beneficial ownership described in footnotes (1) through (8). |
Report of Independent Registered Public Accounting Firm | F-2 |
Consolidated Balance Sheets | F-3 |
Consolidated Statements of Operations and Comprehensive Loss | F-4 |
Consolidated Statements of Stockholders' Equity | F-5 |
Consolidated Statements of Cash Flows | F-6 |
Notes to Consolidated Financial Statements | F-7 |
| 2017 | 2016 | |||||||
Assets | ||||||||
| Current assets | ||||||||
| Cash and cash equivalents | $ | 1,847,171 | $ | 4,117,324 | ||||
| Accounts receivable, net | 809,540 | 542,420 | ||||||
| Inventory, net | 533,425 | 692,368 | ||||||
| Prepaid expenses and other current assets | 311,644 | 329,646 | ||||||
| Total current assets | 3,501,780 | 5,681,758 | ||||||
| Property and equipment, net | 835,537 | 800,723 | ||||||
| Goodwill | 600,814 | 600,814 | ||||||
| Intangible assets, net | 1,353,182 | 1,620,998 | ||||||
| Other noncurrent assets | 328,601 | 279,752 | ||||||
| Total assets | $ | 6,619,914 | $ | 8,984,045 | ||||
Liabilities and Stockholders' Equity | ||||||||
| Current liabilities | ||||||||
| Accounts payable | $ | 1,691,712 | $ | 2,232,563 | ||||
| Accrued compensation and benefits | 746,924 | 578,480 | ||||||
| Accrued liabilities | 1,160,714 | 1,215,283 | ||||||
| Deferred revenue | 24,442 | 37,397 | ||||||
| Short-term notes payable | 1,010,961 | 1,023,815 | ||||||
| Current maturities of long-term capital lease obligation | 154,839 | 184,399 | ||||||
| Total current liabilities | 4,789,592 | 5,271,937 | ||||||
| Deferred rent | 290,719 | 398,084 | ||||||
| Warrant liability | 8,453 | — | ||||||
| Long-term capital lease obligation and other noncurrent liabilities | 130,153 | 146,543 | ||||||
| Total liabilities | 5,218,917 | 5,816,564 | ||||||
| Commitments (Note 8) | ||||||||
| Stockholders' equity | ||||||||
Common stock, $0.01 par value; 200,000,000 shares authorized; 2,265,320 and 1,012,171 shares issued and outstanding at December 31, 2017 and December 31, 2016, respectively | 22,653 | 10,122 | ||||||
Preferred stock, $0.01 par value; 10,000,000 shares authorized; none issued and outstanding at December 31, 2017 and December 31, 2016, respectively | — | — | ||||||
| Additional paid-in capital | 150,114,671 | 136,442,302 | ||||||
| Accumulated other comprehensive (loss)/income | (25,900 | ) | 6,176 | |||||
| Accumulated deficit | (148,710,427 | ) | (133,291,119 | ) | ||||
| Total stockholders' equity | 1,400,997 | 3,167,481 | ||||||
| Total liabilities and stockholders' equity | $ | 6,619,914 | $ | 8,984,045 | ||||
| 2017 | 2016 | |||||||
| Revenue | ||||||||
| Product sales | $ | 2,771,869 | $ | 3,524,178 | ||||
| Laboratory services | 41,960 | 228,904 | ||||||
| Collaboration revenue | 397,178 | 272,603 | ||||||
| Total revenue | 3,211,007 | 4,025,685 | ||||||
| Operating expenses | ||||||||
| Cost of products sold | 1,612,838 | 1,658,571 | ||||||
| Cost of services | 520,338 | 631,333 | ||||||
| Research and development | 6,883,293 | 8,613,236 | ||||||
| General and administrative | 6,692,659 | 6,602,608 | ||||||
| Sales and marketing | 2,767,670 | 5,529,274 | ||||||
| Total operating expenses | 18,476,798 | 23,035,022 | ||||||
| Operating loss | (15,265,791 | ) | (19,009,337 | ) | ||||
| Other income/(expense) | ||||||||
| Interest and other expense | (87,255 | ) | (5,967 | ) | ||||
| Interest expense | (233,505 | ) | (143,347 | ) | ||||
| Foreign currency transaction gains/(losses) | 23,179 | (8,102 | ) | |||||
| Change in fair value of derivative financial instruments | 144,064 | — | ||||||
| Total other expense | (153,517 | ) | (157,416 | ) | ||||
| Loss before income taxes | (15,419,308 | ) | (19,166,753 | ) | ||||
| Provision for income taxes | — | — | ||||||
| Net loss | (15,419,308 | ) | (19,166,753 | ) | ||||
| Preferred stock dividends and beneficial conversion | — | (332,550 | ) | |||||
| Net loss available to common stockholders | $ | (15,419,308 | ) | $ | (19,499,303 | ) | ||
| Net loss per common share - basic and diluted | $ | (9.80 | ) | $ | (27.59 | ) | ||
| Weighted average shares outstanding - basic and diluted | 1,573,769 | 706,702 | ||||||
| Net loss | $ | (15,419,308 | ) | $ | (19,166,753 | ) | ||
| Other comprehensive (loss)/income - foreign currency translation | (32,076 | ) | 7,235 | |||||
| Comprehensive loss | $ | (15,451,384 | ) | $ | (19,159,518 | ) | ||
| Common Stock | Preferred Stock | Additional | Accumulated Other | |||||||||||||||||||||||||||||
Number of Shares | Amount | Number of Shares | Amount | Paid- in Capital | Comprehensive (Loss) / Income | Accumulated Deficit | Total | |||||||||||||||||||||||||
| Balances at December 31, 2015 | 501,907 | $ | 5,019 | — | — | $ | 121,611,452 | $ | (1,059 | ) | $ | (114,124,366 | ) | $ | 7,491,046 | |||||||||||||||||
| Stock option exercises | 2,660 | 27 | — | — | 23,744 | — | — | 23,771 | ||||||||||||||||||||||||
| Private offering of common stock, preferred stock and warrants, net of issuance costs | 269,765 | 2,697 | 2,309,428 | 23,094 | 9,434,958 | — | — | 9,460,749 | ||||||||||||||||||||||||
| Preferred stock conversion | 92,377 | 924 | (2,309,428 | ) | (23,094 | ) | 22,170 | — | — | — | ||||||||||||||||||||||
| At the market offering, net of offering costs | 144,795 | 1,448 | — | — | 4,404,525 | — | — | 4,405,973 | ||||||||||||||||||||||||
| Issuance of RSUs | 667 | 7 | — | — | (8 | ) | — | — | (1 | ) | ||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | 945,461 | — | — | 945,461 | ||||||||||||||||||||||||
| Foreign currency translation | — | — | — | — | — | 7,235 | — | 7,235 | ||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | (19,166,753 | ) | (19,166,753 | ) | ||||||||||||||||||||||
| Balances at December 31, 2016 | 1,012,171 | 10,122 | — | — | 136,442,302 | 6,176 | (133,291,119 | ) | 3,167,481 | |||||||||||||||||||||||
| Stock option exercises | 1,167 | 12 | — | — | 8,168 | — | — | 8,180 | ||||||||||||||||||||||||
| Public offering of common stock and warrants, net of issuance costs | 1,000,000 | 10,000 | — | — | 8,813,242 | — | — | 8,823,242 | ||||||||||||||||||||||||
| At the market offering, net of offering costs | 227,216 | 2,272 | — | — | 3,806,564 | — | — | 3,808,836 | ||||||||||||||||||||||||
| Issuance of RSUs | 6,025 | 60 | — | — | (60 | ) | — | — | — | |||||||||||||||||||||||
| Stock compensation expense | — | — | — | — | 911,398 | — | — | 911,398 | ||||||||||||||||||||||||
| Legal settlement in common stock | 15,843 | 158 | — | — | 109,841 | — | — | 109,999 | ||||||||||||||||||||||||
| Vendor payment in common stock | 2,898 | 29 | — | — | 23,216 | — | — | 23,245 | ||||||||||||||||||||||||
| Foreign currency translation | — | — | — | — | — | (32,076 | ) | — | (32,076 | ) | ||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | (15,419,308 | ) | (15,419,308 | ) | ||||||||||||||||||||||
| Balances at December 31, 2017 | 2,265,320 | $ | 22,653 | — | $ | — | $ | 150,114,671 | $ | (25,900 | ) | $ | (148,710,427 | ) | $ | 1,400,997 | ||||||||||||||||
| 2017 | 2016 | |||||||
| Cash flows from operating activities | ||||||||
| Net loss | $ | (15,419,308 | ) | $ | (19,166,753 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities | ||||||||
| Depreciation and amortization | 669,088 | 656,047 | ||||||
| Loss on disposal of property and equipment | — | 6,309 | ||||||
| Noncash interest expense | 185,294 | 4,527 | ||||||
| Share-based compensation | 911,398 | 945,461 | ||||||
| Inventory obsolescence | — | 113,465 | ||||||
| Change in fair value of warrant liabilities | (144,064 | ) | — | |||||
| Unamortized discount on bridge loan at repayment | 85,932 | — | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (260,471 | ) | 136,226 | |||||
| Inventory | 161,027 | 20,179 | ||||||
| Other assets | (315,688 | ) | 263,882 | |||||
| Accounts payable | (563,357 | ) | (53,229 | ) | ||||
| Accrued compensation and other liabilities | 399,224 | (163,223 | ) | |||||
| Deferred revenue | (12,955 | ) | (13,528 | ) | ||||
| Net cash used in operating activities | (14,303,880 | ) | (17,250,637 | ) | ||||
| Cash flows from investing activities | ||||||||
| Purchases of property and equipment (net of proceeds on disposals) | (276,950 | ) | (123,514 | ) | ||||
| Net cash used in investing activities | (276,950 | ) | (123,514 | ) | ||||
| Cash flows from financing activities | ||||||||
| Proceeds from issuance of common stock, net of issuance costs | 3,808,836 | 4,405,973 | ||||||
| Proceeds from issuance of promissory notes, net of issuance costs | — | 204,895 | ||||||
| Proceeds from private offering of common stock, preferred stock and warrants, net of issuance costs | — | 9,460,749 | ||||||
| Proceeds from issuance of units, net of selling costs | 8,754,882 | — | ||||||
| Proceeds from exercise of stock options and warrants | 76,537 | 23,771 | ||||||
| Proceeds from debt, net of issuance costs | 1,168,222 | — | ||||||
| Payments on debt | (1,255,198 | ) | (178,997 | ) | ||||
| Payments on capital lease obligations | (205,085 | ) | (251,701 | ) | ||||
| Net cash provided by financing activities | 12,348,194 | 13,664,690 | ||||||
| Effects of exchange rates on cash | (37,517 | ) | 12,565 | |||||
| Net decrease in cash and cash equivalents | (2,270,153 | ) | (3,696,896 | ) | ||||
| Cash and cash equivalents at beginning of year | 4,117,324 | 7,814,220 | ||||||
| Cash and cash equivalents at end of year | $ | 1,847,171 | $ | 4,117,324 | ||||
| Supplemental disclosure of cash flow information | ||||||||
| Cash paid for interest | $ | 48,211 | $ | 58,564 | ||||
| Supplemental disclosures of noncash investing and financing activities: | ||||||||
| Unpaid deferred offering costs | $ | 48,398 | $ | — | ||||
| Shares issued to settle obligations | $ | 133,245 | $ | — | ||||
| Issuance of placement agent warrant | $ | 93,677 | $ | — | ||||
| · | Its Acuitas DNA tests provide rapid microbial identification and antibiotic resistance gene information. These products include our Acuitas Rapid Test for complicated urinary tract infection in development, the QuickFISH family of FDA-cleared and CE-marked diagnostics used to rapidly detect pathogens in positive blood cultures, and its Acuitas Resistome Tests for genetic analysis of hospital surveillance isolates. |
| · | Its Acuitas Lighthouse informatics systems are cloud-based HIPAA compliant informatics offerings that combine clinical lab test results with patient and hospital information to provide analytics and actionable insights to help manage MDROs in the hospital and patient care environment. Components of its informatics systems are the Acuitas Lighthouse Knowledgebase, a proprietary data warehouse of genomic data matched with antibiotic susceptibility information for bacterial pathogens and its Acuitas Lighthouse informatics, which can be specific to a healthcare facility or collaborator, such as a pharmaceutical company. |
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Raw materials and supplies | $ | 360,134 | $ | 479,479 | ||||
| Work-in process | 51,233 | 27,422 | ||||||
| Finished goods | 122,058 | 185,467 | ||||||
| Total | $ | 533,425 | $ | 692,368 | ||||
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Laboratory and manufacturing equipment | $ | 4,109,367 | $ | 3,785,133 | ||||
| Office furniture and equipment | 700,299 | 688,952 | ||||||
| Computers and network equipment | 1,505,651 | 1,472,144 | ||||||
| Leasehold improvements | 729,504 | 662,506 | ||||||
| 7,044,821 | 6,608,735 | |||||||
| Less accumulated depreciation | (6,209,284 | ) | (5,808,012 | ) | ||||
| Property and equipment, net | $ | 835,537 | $ | 800,723 | ||||
| December 31, 2017 | December 31, 2016 | |||||||||||||||||||
| Cost | Accumulated Amortization | Net Balance | Accumulated Amortization | Net Balance | ||||||||||||||||
| Trademarks and tradenames | $ | 461,000 | $ | (113,679 | ) | $ | 347,321 | $ | (67,575 | ) | $ | 393,425 | ||||||||
| Developed technology | 458,000 | (161,322 | ) | 296,678 | (95,898 | ) | 362,102 | |||||||||||||
| Customer relationships | 1,094,000 | (384,817 | ) | 709,183 | (228,529 | ) | 865,471 | |||||||||||||
| $ | 2,013,000 | $ | (659,818 | ) | $ | 1,353,182 | $ | (392,002 | ) | $ | 1,620,998 | |||||||||
| Year Ending December 31, | ||||
| 2018 | $ | 267,816 | ||
| 2019 | 267,816 | |||
| 2020 | 267,816 | |||
| 2021 | 267,816 | |||
| 2022 | 165,117 | |||
| Thereafter | 116,801 | |||
| Total | $ | 1,353,182 | ||
Finite-lived intangible assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of the asset may not be recoverable. If any indicators were present, the Company would test for recoverability by comparing the carrying amount of the asset to the net undiscounted cash flows expected to be generated from the asset. If those net undiscounted cash flows do not exceed the carrying amount (i.e., the asset is not recoverable), the Company would perform the next step, which is to determine the fair value of the asset and record an impairment loss, if any. During the years ended December 31, 2017 and 2016, the Company determined that its finite-lived intangible assets were not impaired.
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Cost of services | $ | 13,776 | $ | 6,003 | ||||
| Research and development | 237,103 | 236,341 | ||||||
| General and administrative | 603,787 | 599,550 | ||||||
| Sales and marketing | 56,732 | 103,567 | ||||||
| $ | 911,398 | $ | 945,461 | |||||
Number of Options | Weighted- Average Exercise Price | Weighted- Average Remaining Contractual Life (in years) | Aggregate Intrinsic Value | |||||||||||||
| Outstanding at January 1, 2016 | 86,431 | $ | 65.00 | 9.1 | $ | 1,575,646 | ||||||||||
| Granted | 58,546 | $ | 35.25 | |||||||||||||
| Exercised | (2,660 | ) | $ | 9.00 | $ | 79,406 | ||||||||||
| Forfeited | (22,868 | ) | $ | 99.75 | ||||||||||||
| Expired | (343 | ) | $ | 212.25 | ||||||||||||
| Outstanding at December 31, 2016 | 119,106 | $ | 44.00 | 8.6 | $ | 663,298 | ||||||||||
| Granted | 58,324 | $ | 17.58 | |||||||||||||
| Exercised | (1,167 | ) | $ | 7.01 | $ | 11,256 | ||||||||||
| Forfeited | (24,538 | ) | $ | 36.31 | ||||||||||||
| Expired | (12,330 | ) | $ | 83.49 | ||||||||||||
| Outstanding at December 31, 2017 | 139,395 | $ | 31.16 | 8.3 | $ | 37,339 | ||||||||||
| Vested and expected to vest | 139,395 | $ | 31.16 | 8.3 | $ | 37,339 | ||||||||||
| Exercisable at December 31, 2017 | 63,365 | $ | 4.73 | 7.7 | $ | 33,575 | ||||||||||
| Year Ended December 31, | ||||||||
| 2017 | 2016 | |||||||
| Annual dividend | — | — | ||||||
| Expected life (in years) | 5.25 - 6.25 | 5.25 - 6.25 | ||||||
| Risk free interest rate | 1.8 - 2.3% | 1.2 - 2.2% | ||||||
| Expected volatility | 44.2 - 53.0% | 42.0 - 49.8% | ||||||
| Outstanding at December 31, | ||||||||||||||
| Issuance | Exercise Price | Expiration | 2017 | 2016 | ||||||||||
| August 2007 | $ | 197.75 | August 2017 | - | 357 | |||||||||
| March 2008 | $ | 19,763.50 | March 2018 | 2 | 2 | |||||||||
| November 2009 | $ | 197.75 | November 2019 | 267 | 267 | |||||||||
| January 2010 | $ | 197.75 | January 2020 | 267 | 267 | |||||||||
| March 2010 | $ | 197.75 | March 2020 | 51 | 51 | |||||||||
| November 2011 | $ | 197.75 | November 2021 | 209 | 209 | |||||||||
| December 2011 | $ | 197.75 | December 2021 | 27 | 27 | |||||||||
| March 2012 | $ | 2,747.50 | March 2019 | 165 | 165 | |||||||||
| February 2015 | $ | 165.00 | February 2025 | 9,000 | 9,000 | |||||||||
| May 2015 | $ | 165.00 | May 2020 | 138,310 | 138,310 | |||||||||
| May 2016 | $ | 32.81 | May 2021 | 189,574 | 189,574 | |||||||||
| June 2016 | $ | 32.81 | May 2021 | 82,033 | 82,033 | |||||||||
| June 2017 | $ | 19.50 | June 2022 | 18,754 | - | |||||||||
| July 2017 | $ | 17.25 | July 2022 | 6,349 | - | |||||||||
| July 2017 | $ | 12.50 | July 2022 | 50,000 | - | |||||||||
| July 2017 | $ | 10.63 | July 2022 | 1,000,000 | - | |||||||||
| 1,495,008 | 420,262 | |||||||||||||
| December 31, | ||||||||
| 2017 | 2016 | |||||||
| Deferred tax assets: | ||||||||
| NOL carryforward | $ | 46,326,407 | $ | 60,357,220 | ||||
| R&E credit carryforward | 2,559,479 | 2,559,479 | ||||||
| Share-based compensation | 345,088 | 448,534 | ||||||
| Inventory reserve | 45,338 | 269,708 | ||||||
| Depreciation | 71,756 | 117,629 | ||||||
| Accruals and other | 247,093 | 333,126 | ||||||
| Total deferred tax assets | 49,595,161 | 64,085,696 | ||||||
| Valuation allowance | (49,251,408 | ) | (63,520,548 | ) | ||||
| Deferred tax liabilities: | ||||||||
| Intangible assets | (343,753 | ) | (565,148 | ) | ||||
| Net deferred tax liability | $ | — | $ | — | ||||
| 2017 | 2016 | |||||||
| Federal income tax benefit at statutory rates | 34.0 | % | 34.0 | % | ||||
| State income tax benefit, net of Federal benefit | 6.8 | % | 6.5 | % | ||||
| Tax reform impact | (134.5 | )% | 0.0 | % | ||||
| Change in valuation allowance | 93.0 | % | (37.3 | )% | ||||
| Change in state tax rates and other | 0.7 | % | (3.2 | )% | ||||
| 0.0 | % | 0.0 | % | |||||
| Year ending December 31, | Capital Leases | Operating Leases | Total | |||||||||
| 2018 | $ | 177,464 | $ | 1,041,323 | $ | 1,218,787 | ||||||
| 2019 | 85,393 | 1,054,374 | 1,139,767 | |||||||||
| 2020 | 56,981 | 1,072,748 | 1,129,729 | |||||||||
| 2021 | 1,773 | 482,058 | 483,831 | |||||||||
| 2022 and thereafter | - | 35,647 | 35,647 | |||||||||
| Total | 321,611 | $ | 3,686,150 | $ | 4,007,761 | |||||||
| Less: amount representing interest | (36,619 | ) | ||||||||||
| Net present value of future minimum lease payments | 284,992 | |||||||||||
| Current maturities | (154,839 | ) | ||||||||||
| Long-term maturities | $ | 130,153 | ||||||||||
| 2017 | 2016 | |||||||
| Laboratory and manufacturing equipment | $ | 850,792 | $ | 560,829 | ||||
| Office furniture and equipment | 64,790 | 64,790 | ||||||
| Computers and network equipment | 24,350 | 24,350 | ||||||
| Less accumulated amortization | (454,471 | ) | (270,808 | ) | ||||
| Capital lease assets, net | $ | 485,461 | $ | 379,161 | ||||
| · | Level 1 - defined as observable inputs such as quoted prices in active markets; |
| · | Level 2 - defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and |
| · | Level 3 - defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions such as expected revenue growth and discount factors applied to cash flow projections. |
| Description | Balance at December 31, 2016 | Established in 2017 | Change in Fair Value | Expensed | Balance at December 31, 2017 | |||||||||||||||
| Embedded conversion option liability | $ | - | $ | 4,500 | $ | - | $ | (4,500 | ) | $ | - | |||||||||
| Warrant liability | $ | - | $ | 152,517 | $ | (144,064 | ) | $ | - | $ | 8,453 | |||||||||

Exhibit Number |
| Description |
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| 5.1 * | Opinion of Ballard Spahr LLP | |
| ||
|
|
|
| ||
|
|
|
|
Exhibit Number |
| Description |
|
| |
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
|
Exhibit Number |
| Description |
|
| |
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
| ||
|
|
|
|
|
|
| 21.1 * | Power of Attorney (including on signature page to Form S-1 Registration Statement, File No. 333-218392 filed on June 1, 2017) | |
|
|
|
101 * |
| Interactive data files pursuant to Rule 405 of Regulation S-T; (i) the Consolidated Balance Sheets, (ii) the Consolidated Statements of Operations and Comprehensive Loss, (iii) the Consolidated Statements of Stockholders’ Equity, (iv) the Consolidated Statements of Cash Flows and (v) the Notes to the Consolidated Financial Statements |
| * | Filed herewith |
| ! | Denotes management compensation plan or contract |
| ± | Confidential treatment has been requested for certain portions of this agreement pursuant to an application for confidential treatment filed with the Securities and Exchange Commission on June 19, 2017. Such provisions have been filed separately with the Commission. |
| OPGEN, INC. | |||
| By: | /s/ Evan Jones | ||
| Evan Jones | |||
| Chief Executive Officer | |||
| Signature | Title | Date |
| /s/ Evan Jones | Chief Executive Officer and Director (principal executive officer) | April 4, 2018 |
| Evan Jones | ||
| /s/ Timothy C. Dec | Chief Financial Officer (principal financial officer and | April 4, 2018 |
| Timothy C. Dec | principal accounting officer) | |
| * | Director | April 4, 2018 |
| Harry J. D'Andrea | ||
| * | Director | April 4, 2018 |
| Timothy J.R. Harris | ||
| * | Director | April 4, 2018 |
| Tina S. Nova | ||
| Director | April 4, 2018 | |
| David M. Rubin | ||
| * | Director | April 4, 2018 |
| Misti Ushio | ||
| * /s/ Timothy C. Dec | April 4, 2018 | |
| Timothy C. Dec, as attorney in fact |