 Making Hope A Reality Nasdaq : BLUE June 16, 2014 Exhibit 99.2 |
 Forward Looking Statement 2 These slides and the accompanying oral presentation contain forward-looking statements and information. The use of words such as “may,” “might,” “will,” “should,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “intend,” “future,” “potential,” or “continue,” and other similar expressions are intended to identify forward looking statements. For example, all statements we make regarding the initiation, timing, progress and results of our preclinical and clinical studies and our research and development programs, our ability to advance product candidates into, and successfully complete, clinical studies, and the timing or likelihood of regulatory filings and approvals are forward looking. The data for LentiGlobin are preliminary in nature and the HGB-205 trial is not completed. These data may not continue for these subjects or be repeated or observed in ongoing or future studies involving our LentiGlobin product candidate, including the HGB-205 Study, the Northstar Study or the HB-206 Study in sickle cell disease. All forward-looking statements are based on estimates and assumptions by our management that, although we believe to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that we expected. These statements are also subject to a number of material risks and uncertainties that are described in the our most recent quarterly report on Form 10-Q, as well as our subsequent fillings with the Securities and Exchange Commission. Any forward-looking statement speaks only as of the date on which it was made. We undertake no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law. |
 Summary - Key Messages Potential for one-time transformative treatments for severe genetic and orphan diseases Encouraging clinical data in beta-thalassemia major patients Promising proof of concept data in CCALD patients Industrialized platform across people, production, development and deployment Disruptive product platform with broad product and deal potential Industry leading team and culture funded for success 3 |
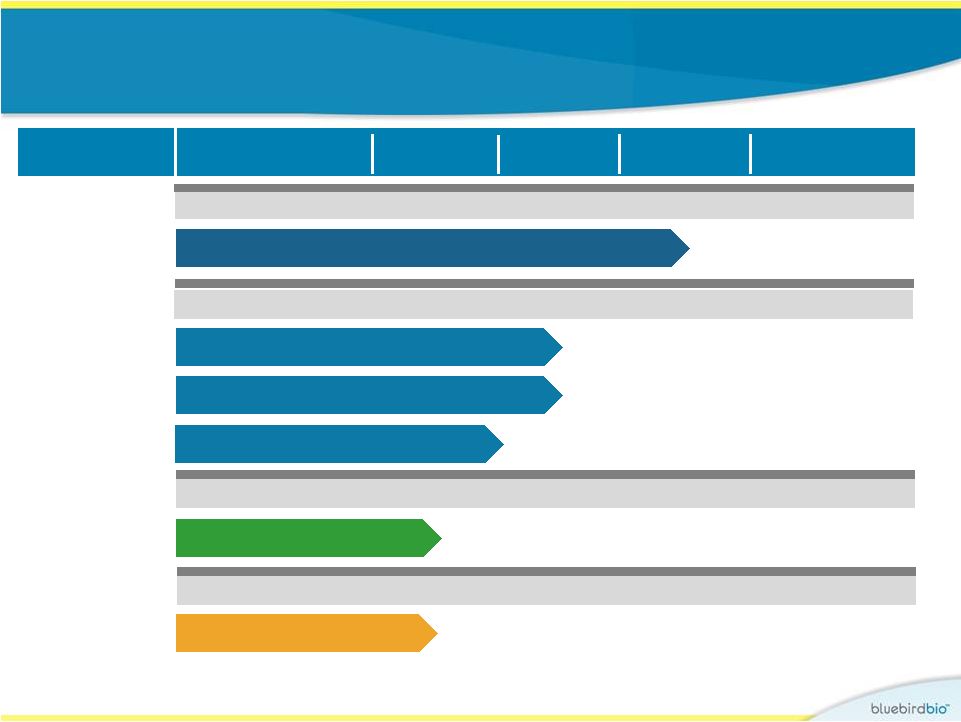 bluebird Pipeline CNS Diseases Hematologic/Solid Tumors Lenti-D LentiGlobin™ CAR-T Cells Undisclosed Worldwide Oncology Hematologic Diseases Products Early Pipeline Research Worldwide Worldwide Global Celgene Collaboration 4 Program Area Preclinical Phase I/II Phase II/III Rights b-thalassemia/SCD (France) – HGB-205 Study** b-thalassemia (U.S.) – Northstar Study** Sickle Cell Disease (U.S.) – HGB-206 Study * The Phase II/III Starbeam Study is our first clinical study of our current Lenti-D viral vector and product candidate. ** The Phase I/II HGB-205 and Northstar Studies are our first clinical studies of our current LentiGlobin viral vector and product candidate. Childhood Cerebral ALD – Starbeam Study* |
 5 IMPROVING GENE THERAPY FOR -THALASSEMIA MAJOR: INITIAL RESULTS FROM STUDY HGB-205 M. Cavazzana, JA Ribeil*, E. Payen*, F. Suarez, O. Negre, Y. Beuzard, F. Touzot, R. Cavallesco, F. Lefrere, S. Chretien, P. Bourget, F. Monpoux, C. Pondarre, B. Neven, F. Bushman, M. Schmidt, C. von Kalle, L. Sandler, S. Soni, B. Ryu, R. Kutner, G. Veres, M. Finer, S. Blanche, O. Hermine, S. Hacein-Bey-Abina, P. Leboulch *these authors contributed equally |
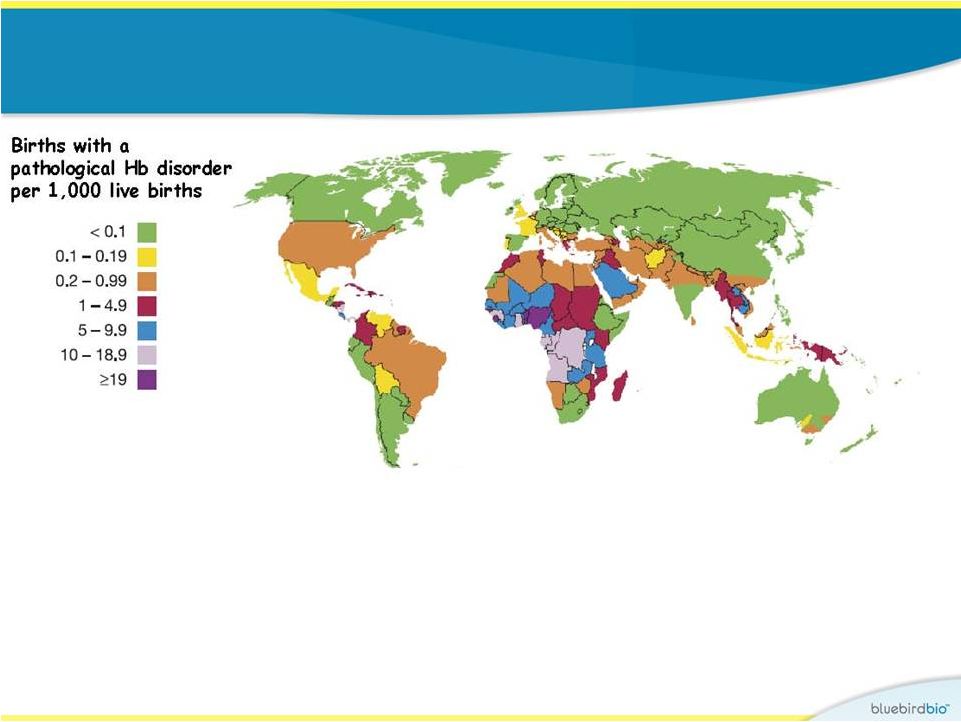 Epidemiology of hemoglobin disorders 6 The hemoglobinopathies are the most prevalent monogenetic disorders in the world – 7% of global population carry an abnormal hemoglobin gene Between 300,000 – 400,00 babies are born each year with a serious hemoglobin disorder • >40,000 with -thalassemia major/HbE -thalassemia • >200,000 with sickle cell disease Adapted from Williams and Weatherall 2012 and March of Dimes, 2006 b b |
 -thalassemia major is a serious disease potentially amenable to gene therapy 7 Only curative approach is allogeneic HSCT, but complicated by: difficulty in finding well-matched donors graft versus host disease prolonged immunosuppression At EHA we report data from two clinical trials of ex-vivo gene therapy in subjects with b-thalassemia major Study Lentiviral vector Current status 1 (LG001) HPV569 Study closed, update presented today 2 (HGB-205) BB305 Enrolling, initial results on first 2 subjects presented today |
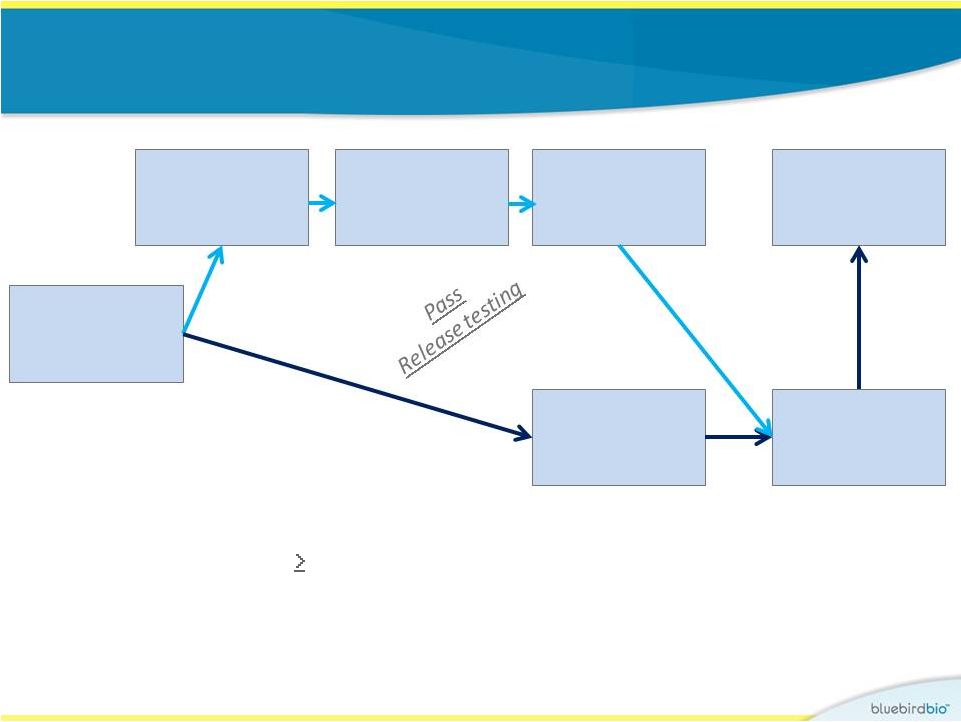 Identical study design for both gene therapy trials • Key eligibility: • b-Thalassemia major ( 100 mls pRBCs/kg/year) • Subjects with severe sickle cell disease are also eligible; none treated to date • No HLA-matched sibling donor HSC mobilization then apheresis Myeloablation with IV busulfan Infusion Follow-up CD34+ cells selection Cryopreserve Lentiviral vector transduction 8 |
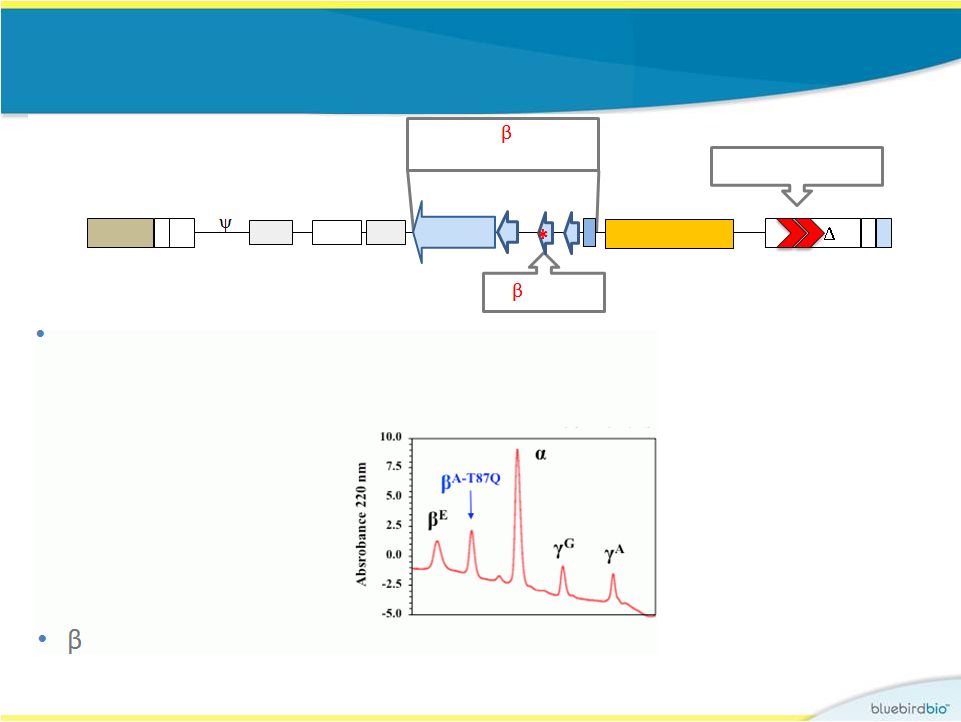 HPV569 lentiviral vector used in Study 1 9 Globin production is under transcriptional control of an erythroid-specific promoter and enhancer R U5 HIV U3 RRE cPPT pA gag 3’enh/pA Globin LCR U3 R A-T87Q -globin allows for monitoring of protein levels produced using HPLC Human -globin gene A-T87Q cHS4 insulators |
 Study 1 – characteristics of included subjects • No AEs related to drug product, including no RCL nor malignancy 10 Subject Outcome 1 Not treated 2 Low number of stem cells infused, no engraftment, received rescue cells 3 Engrafted, 6 years follow-up 4 Engrafted, 2 years follow-up Subject 3 Subject 4 Age 18 22 Genotype b 0 / b 0 / CD34 + VCN 0.6 0.3 CD34 + cell dose (x 10 6 /kg) 4.9* 4.3 *Subject 3 source of CD34+ cells was bone marrow E E b b |
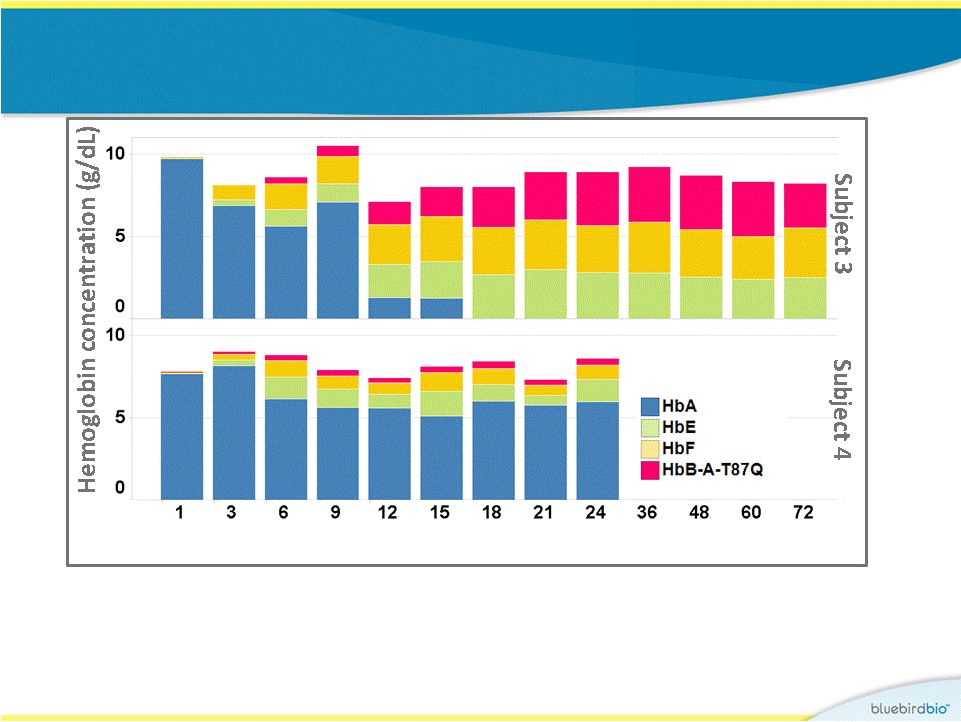 Study 1 : hemoglobin concentrations 11 • Subject 3: stable levels of b A-T87Q –globin beginning at Month 18, transfusion independent by Month 12. Producing 2.7 g/dL of b A-T87Q -globin at 6 years. • Subject 4: minimal levels of b A-T87Q -globin post-treatment, transfusion dependent. Producing 0.4 g/dL of b A-T87Q -globin at 2 years. Months post treatment 9.8 8.6 10.5 8.0 8.0 8.9 8.9 9.2 8.7 8.3 8.2 8.1 7.8 9.0 8.8 7.9 8.1 7.3 8.4 8.6 7.1 7.1 |
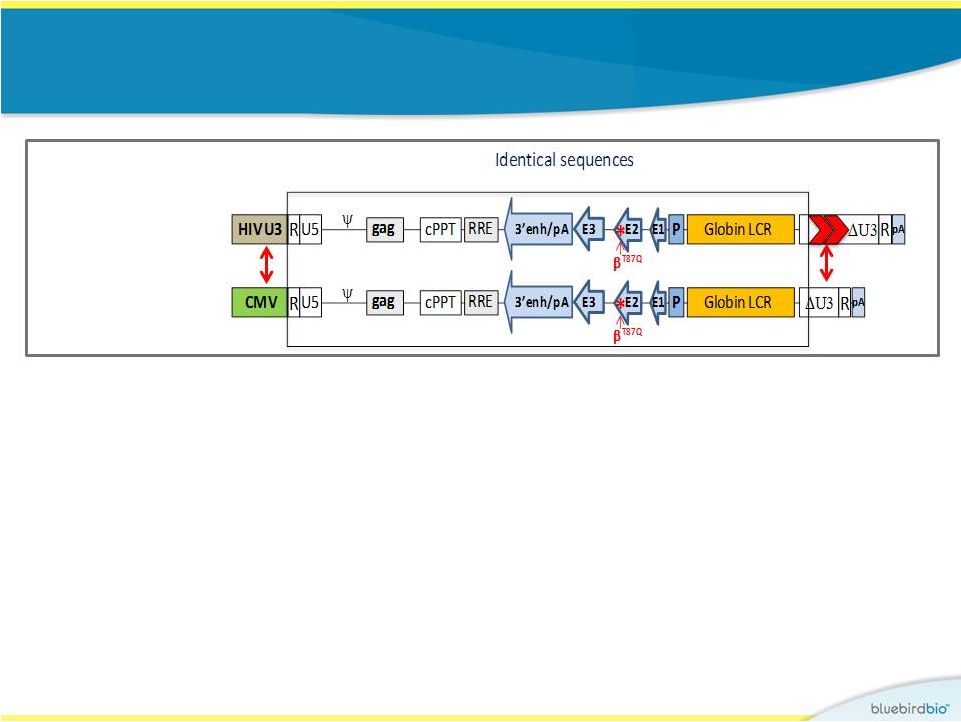 BB305 lentiviral vector used in Study 2 12 • Improvements made in vector design, vector manufacturing process, and drug product manufacturing, including: Study 2: Study 1: • CMV promoter to drive vector production (aim to increase vector titer) • cHS4 insulator elements were removed (aim to increase vector titer, potency, and stability) |
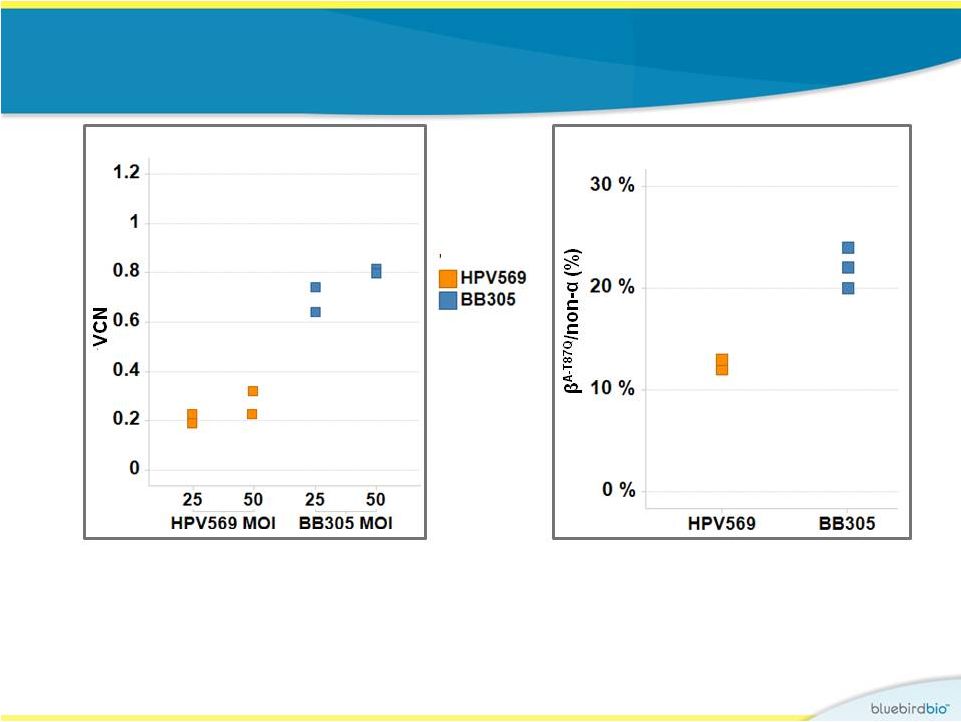 Improvements in BB305 vector design and manufacturing process increase transduction efficiency in vitro 13 • Mean VCN was 2- to 3-fold higher in transduced human CD34 + cells • b A-T87Q –globin was produced at a 2-fold higher level in differentiated erythroid lineage cells |
 Study 2 : characteristics of included subjects (to date) 14 Subject 1 Subject 2 Age at Enrollment 18 16 Genotype b 0 /b E b 0 /b E CD34 + VCN 1.5 2.1 CD34 + cell dose (x 10 6 /kg) 8.9 13.6 |
 Study 2 : Safety 15 Subject 1 Subject 2 Follow up period 4.5 months 2 months Day of neutrophil engraftment ANC > 500/µL Day 13 Day 15 Day of platelet engraftment Unsupported platelet count > 20,000/µL Day 17 Day 24 Non-laboratory Grade 3 AEs Mucositis 1 Mucositis SAEs occurring Day 0 None None Insertion site analysis At 3 Months: highly polyclonal (>1000), no clonal dominance Not yet available 1 Subject 1201 had an asymptomatic Grade 3 AST, ALT and GGT elevation from Days 23-90 • No AEs related to drug product, including no RCL nor malignancy |
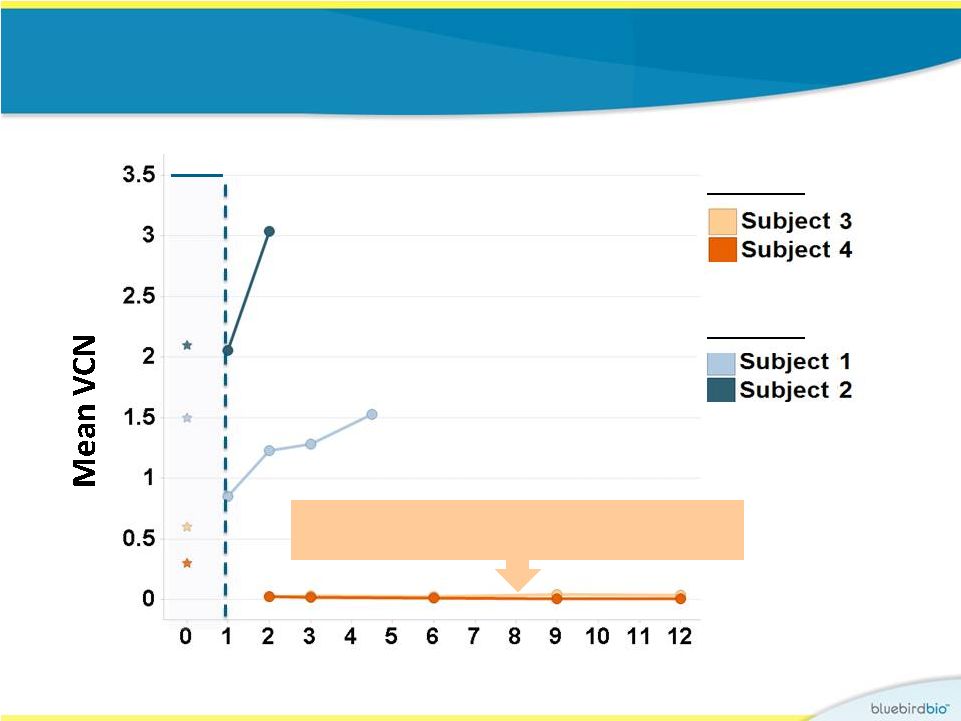    Study 2 vs. Study 1: VCN in peripheral blood leucocytes 16 Months post infusion CD34+ VCN Study 1 Study 2 In first 12 months, VCN in PBLs for Study 1 subjects were between 0.02 and 0.04 |
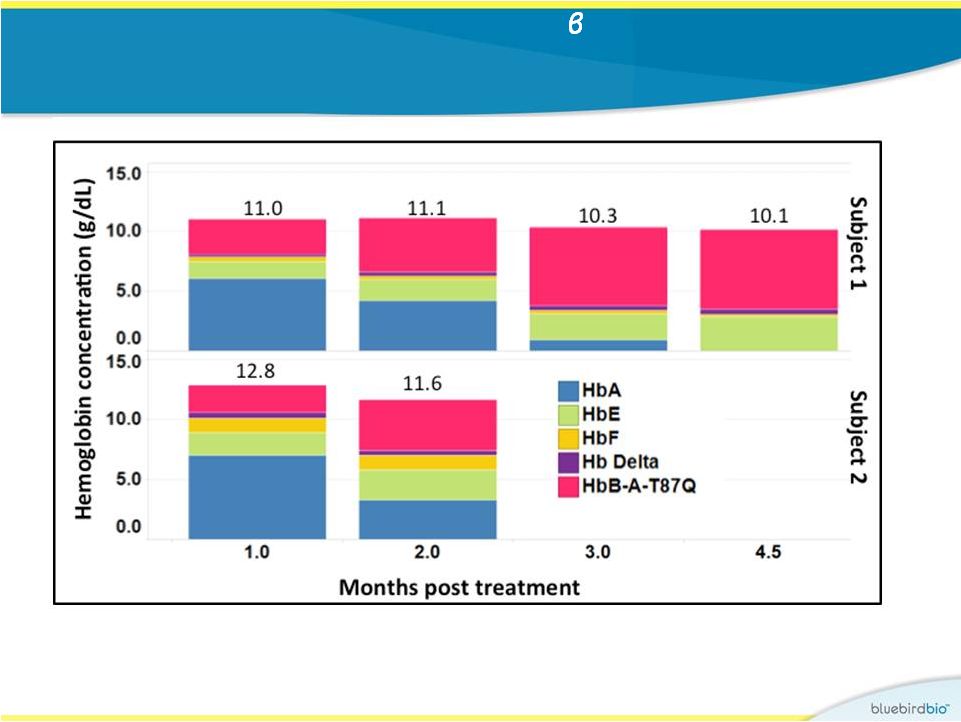 In study 2, early and high production of A-T87Q -globin resulting in rapid transfusion-independence at near normal Hb levels in both patients 17 • Subject 1: producing 6.6 g/dL of b A-T87Q -globin at 4.5 months • Subject 2: producing 4.2 g/dL of b A-T87Q -globin at 2 months |
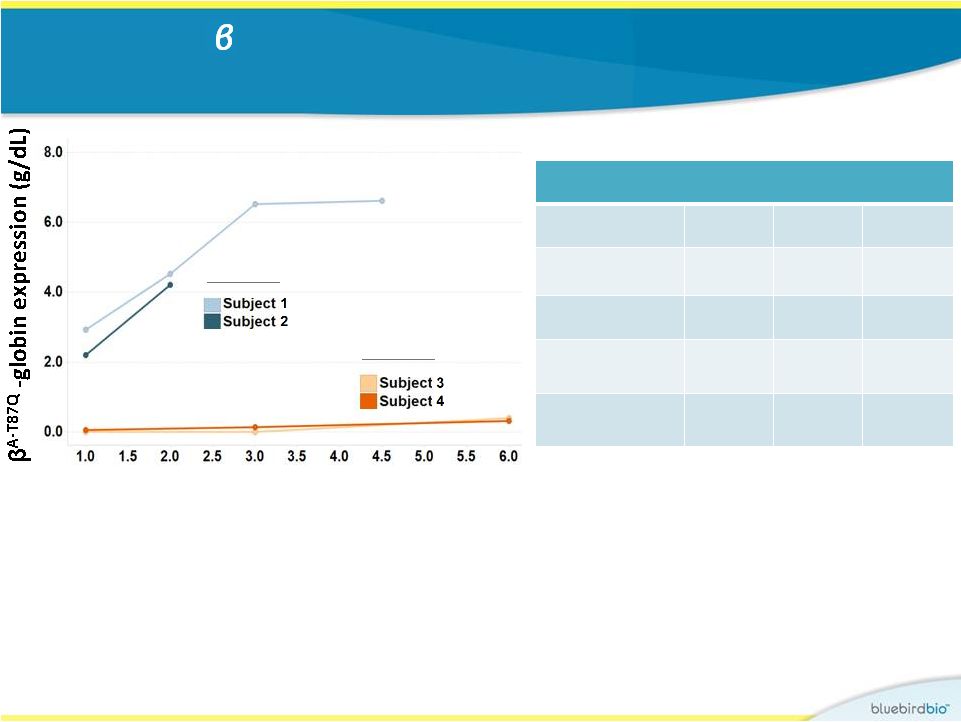 Kinetics of A-T87Q expression and transfusion independence 18 Study 2 Study 1 • In Study 2, rapid production of therapeutic globin (weeks as opposed to one year) • Both subjects in Study 2 have near-normal hemoglobin levels without transfusion support (neither subject has required a transfusion post- engraftment) RBC transfusion Independence Subject 3 Subject 1 Subject 2 Study 1 LG001 2 HGB-205 2 HGB-205 Vector HPV569 BB305 BB305 Day of last transfusion Month 12 Day 10 Day 12 Duration since last transfusion >5 years >125 days >48 days Months post treatment |
 Conclusions 19 • BB305 lentiviral vector and improved manufacturing process produce superior transduction efficiency as compared with HPV569 • With BB305 lentiviral vector in Study 2, neither subject has received a transfusion since the second week post transplantation • Production of b A-T87Q -globin has been rapid and clinically significant resulting in near- normal hemoglobin levels • Initial safety profile is consistent with autologous transplantation, without gene-therapy related adverse events, and with polyclonal reconstitution in the first subject • These data demonstrate that early transfusion independence (within weeks of transplantation) with near-normal levels of hemoglobin can be achieved with ex-vivo gene therapy using BB305 lentiviral vector in subjects with b 0 /b E -thalassemia major |
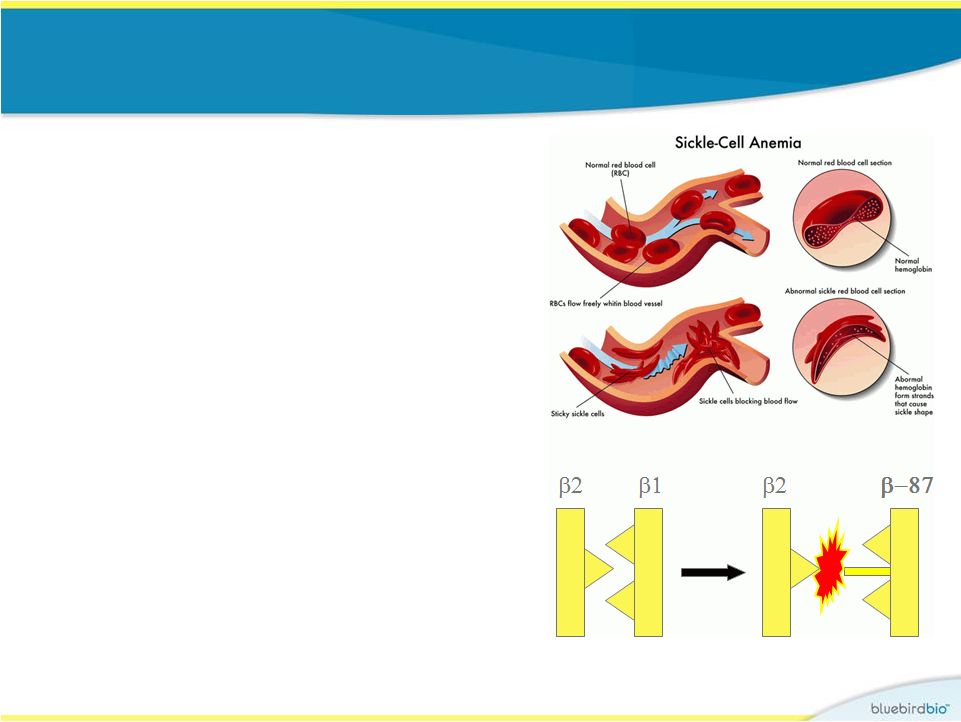 Evidence for why BB305 globin may work for sickle cell disease • BB305 globin incorporates an anti- sickling amino acid that is found in fetal hemoglobin (glutamine at position 87) • Anti-sickling activity of b A-T87Q -globin has been demonstrated in a mouse model of SCD (Science 2001) • Elevated fetal hemoglobin from hereditary persistence of fetal hemoglobin (HPFH) or treatment with hydroxurea has shown clinical benefit F L T V F L Q polymerization destabilization 20 V |
 Ongoing Studies using BB305 lentiviral vector 21 Study Centers Indication Planned subjects Current Status HGB-205 (trial reported today) 1 in France 7 4 subjects enrolled 2 subjects treated to date Northstar Study (HGB-204) 4 in US 1 in Australia 1 in Thailand 15 6 subjects enrolled 1 subject treated to date HGB-206 3-6 planned, all in US Severe sickle cell disease 8 Open IND, pending initiation b- thalassemia major and severe sickle cell disease b- thalassemia major |
 Recent and Upcoming News Flow Enroll first SCD patient in HGB-205 or HGB-206 (2014) Preliminary Thal Northstar & HGB-205 data (late 2014) Various clinical publications Signed global Oncology collaboration with Celgene Completed IPO Initiated phase II/III Starbeam Study Initiated two phase I/II Thal studies (Northstar & HGB- 205) First patient transplanted in Starbeam Study First patient transplanted in Thal HGB-205 study 2013 2013 22 Complete enrollment of Starbeam Study Complete enrollment Thal Northstar & HGB- 205 Preliminary SCD data 2015 2015 2014 2014 First patient transplanted in Northstar Study File IND for sickle cell disease (SCD) study Preliminary Thal HGB-205 data at EHA |
 Q&A |
 Backups |
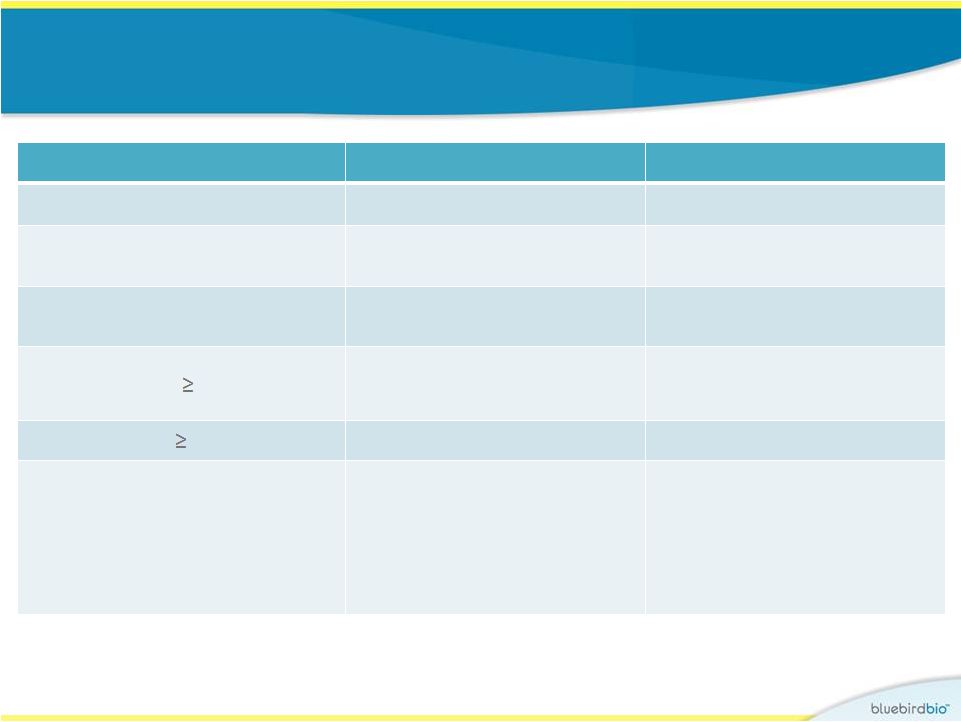 Study 1 (LG001) : Safety • No AEs related to drug product, including no RCL nor malignancy Subject 3 Subject 4 Follow-up period 6 years 2 years Day of neutrophil engraftment ANC > 500/µL Day 27 Day 19 Day of platelet engraftment Unsupported platelet count > 20,000/µL Day 40 Day 130 Non-laboratory Grade 3 AEs None Mucositis, metrorrhagia, epistaxis, mouth bleeding SAEs occurring Day 0 None Thrombocytopenia Insertion site analysis Multiple clones (25-50 detected at each timepoint), including HMGA2, many observed repeatedly over the following 5 years Polyclonal (90-200 clones detected at each timepoint) reconstitution without clonal dominance at Year 1 |
 Subject 3 (LG001) : prominence of HMGA2 clone decreasing over time • By Year 5, SPATS2 and ZZEF1 have replaced HMGA2 as the most common clones identified by LAM-PCR • In spite of decrease in HMGA2 clone, therapeutic effect has been maintained • No hematological or clinical effects of the HMGA2 clone have been noted in over 6 years of follow-up Months 0 5 10 15 20 0 10 20 30 40 50 60 70 vector-modified cells HMGA2- modified cells |