
bb2121 Investor & Analyst Event June 5, 2017 NASDAQ: BLUE Exhibit 99.1

These slides and the accompanying oral presentation contain forward-looking statements and information relating to bluebird bio, its product candidate bb2121 and oncology research and development plans. The use of words such as “may,” “might,” “will,” “should,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “intend,” “future,” “potential,” or “continue,” and other similar expressions are intended to identify forward-looking statements. For example, all statements we make regarding the initiation, timing, progress and results of our preclinical and clinical studies and our research and development programs, our ability to advance product candidates into, and successfully complete, clinical studies, and the timing or likelihood of regulatory filings and approvals are forward looking. All forward-looking statements are based on estimates and assumptions by our management that, although we believe to be reasonable, are inherently uncertain. All forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those that we expected. These statements are also subject to a number of material risks and uncertainties that are described in our most recent quarterly report on Form 10-Q, as well as our subsequent filings with the Securities and Exchange Commission. Any forward-looking statement speaks only as of the date on which it was made. We undertake no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law. Forward Looking Statements

Agenda Introduction bluebird Oncology Franchise CRB-401 Study Design and Clinical Overview CRB-401 Data Closing Q&A Manisha Pai, Nick Leschly Philip Gregory, D.Phil. M. Travis Quigley Jesus Berdeja, M.D. David Davidson, M.D. bluebird bio management Jesus Berdeja, M.D., principal investigator Michael Pehl, president, hematology & oncology, Celgene

Welcome Nick Leschly, chief bluebird
Our Vision: Make Hope a Reality TRUE BLUE BLUE MOJO OUR PATIENTS OUR PEOPLE

Advancing Multiple Programs in Parallel MM TDT SCD CALD
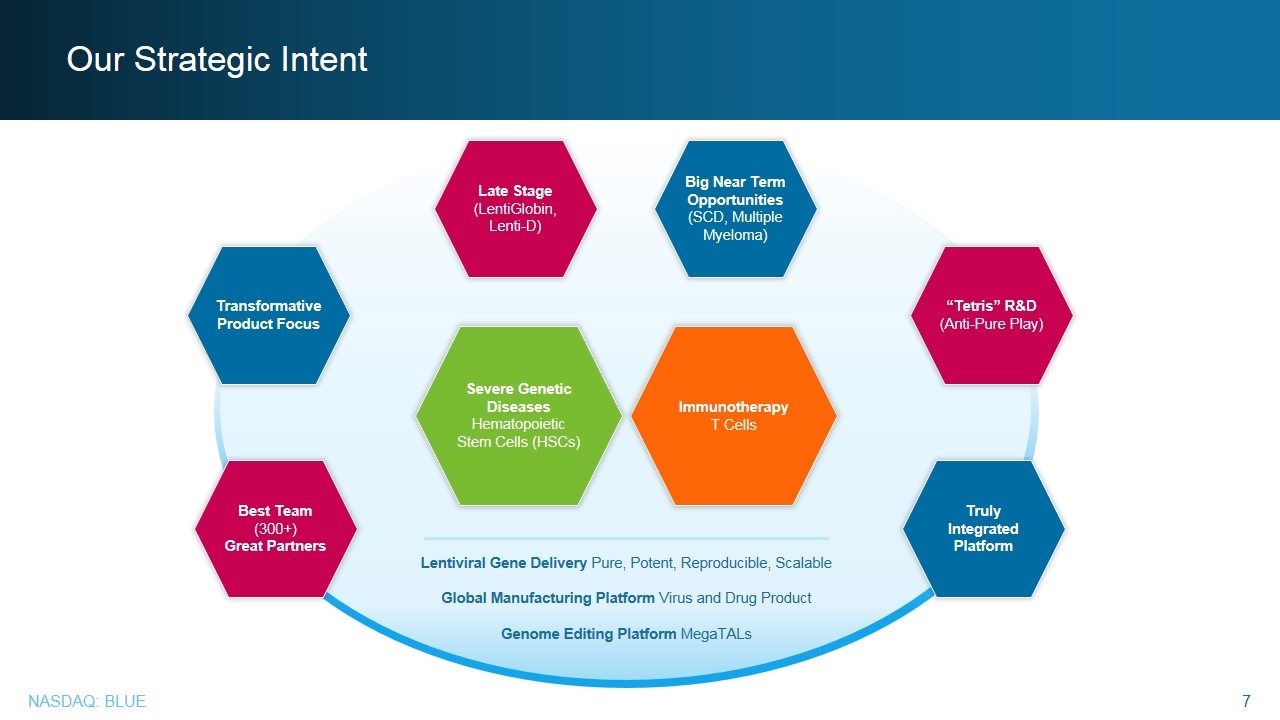
Our Strategic Intent Severe Genetic Diseases Hematopoietic Stem Cells (HSCs) Immunotherapy T Cells Late Stage (LentiGlobin, Lenti-D) Big Near Term Opportunities (SCD, Multiple Myeloma) Best Team (300+) Great Partners Truly Integrated Platform Transformative Product Focus “Tetris” R&D (Anti-Pure Play) Lentiviral Gene Delivery Pure, Potent, Reproducible, Scalable Global Manufacturing Platform Virus and Drug Product Genome Editing Platform MegaTALs

bluebird Oncology Franchise Philip Gregory, D.Phil., chief scientific officer
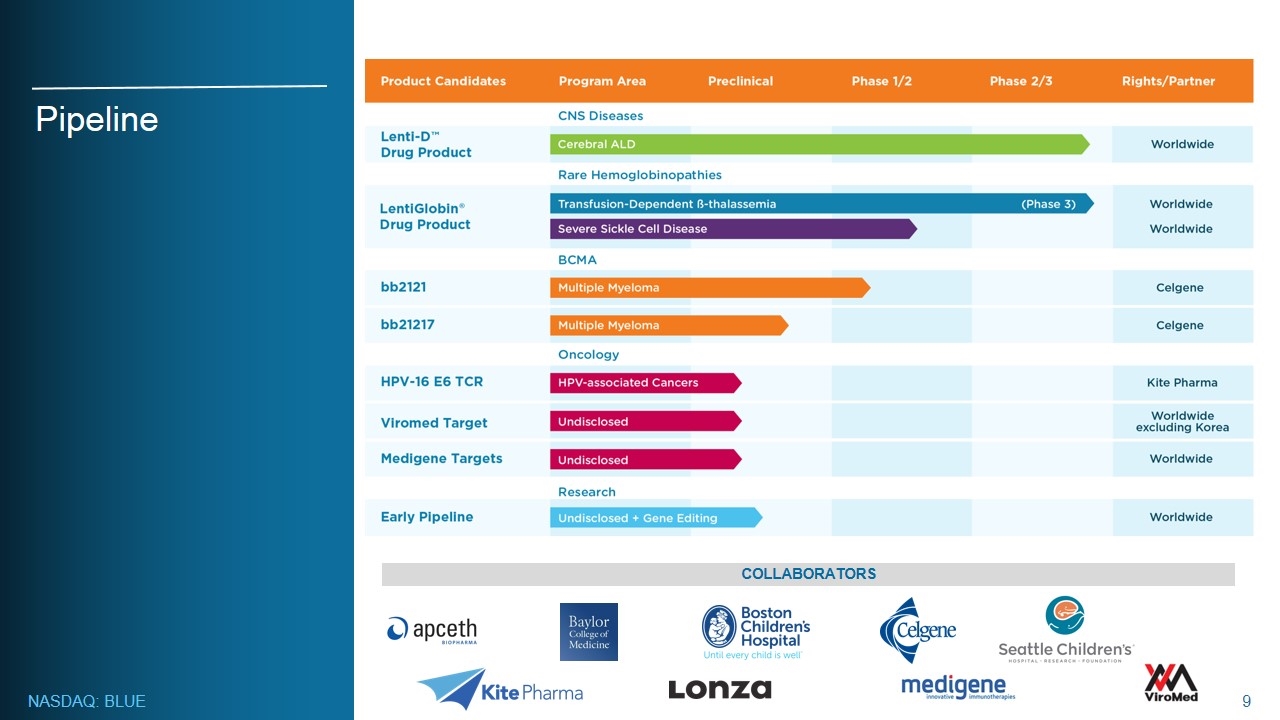
Pipeline COLLABORATORS
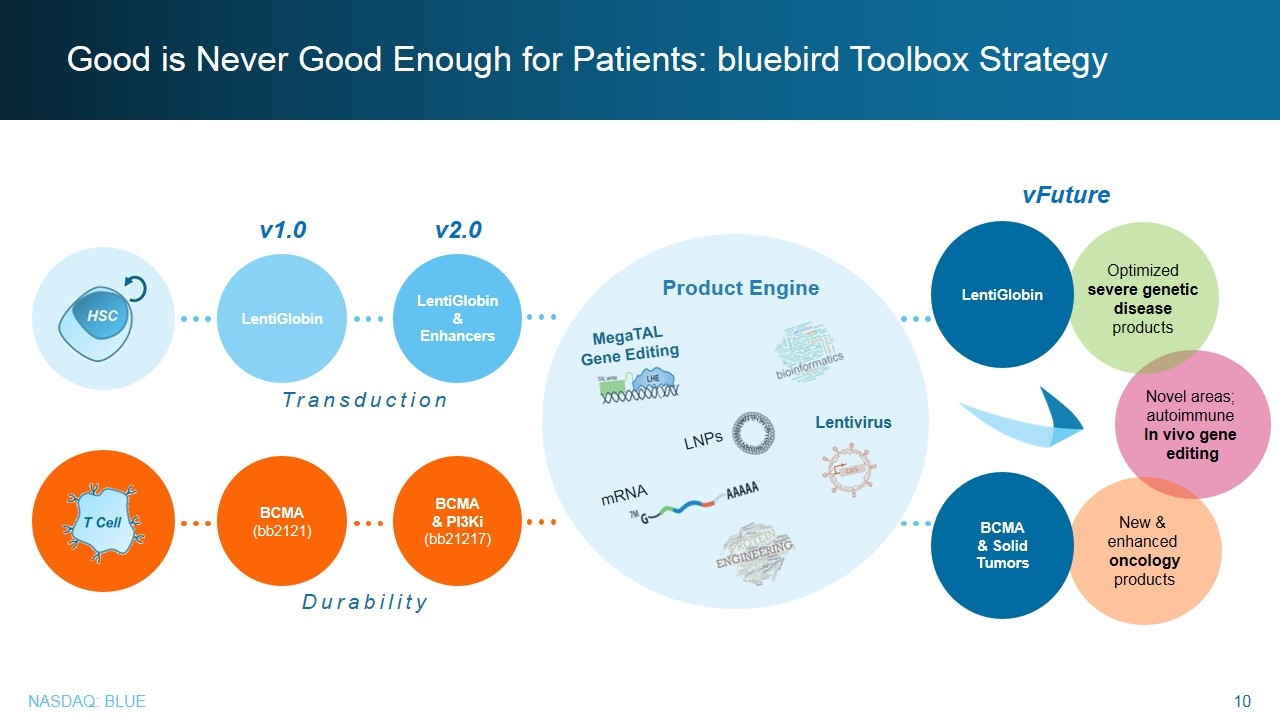
Good is Never Good Enough for Patients: bluebird Toolbox Strategy BCMA (bb2121) LentiGlobin v1.0 Transduction Durability BCMA & PI3Ki (bb21217) LentiGlobin & Enhancers v2.0 Novel areas; autoimmune In vivo gene editing Optimized severe genetic disease products New & enhanced oncology products vFuture BCMA & Solid Tumors LentiGlobin Product Engine MegaTAL Gene Editing LNPs Lentivirus mRNA
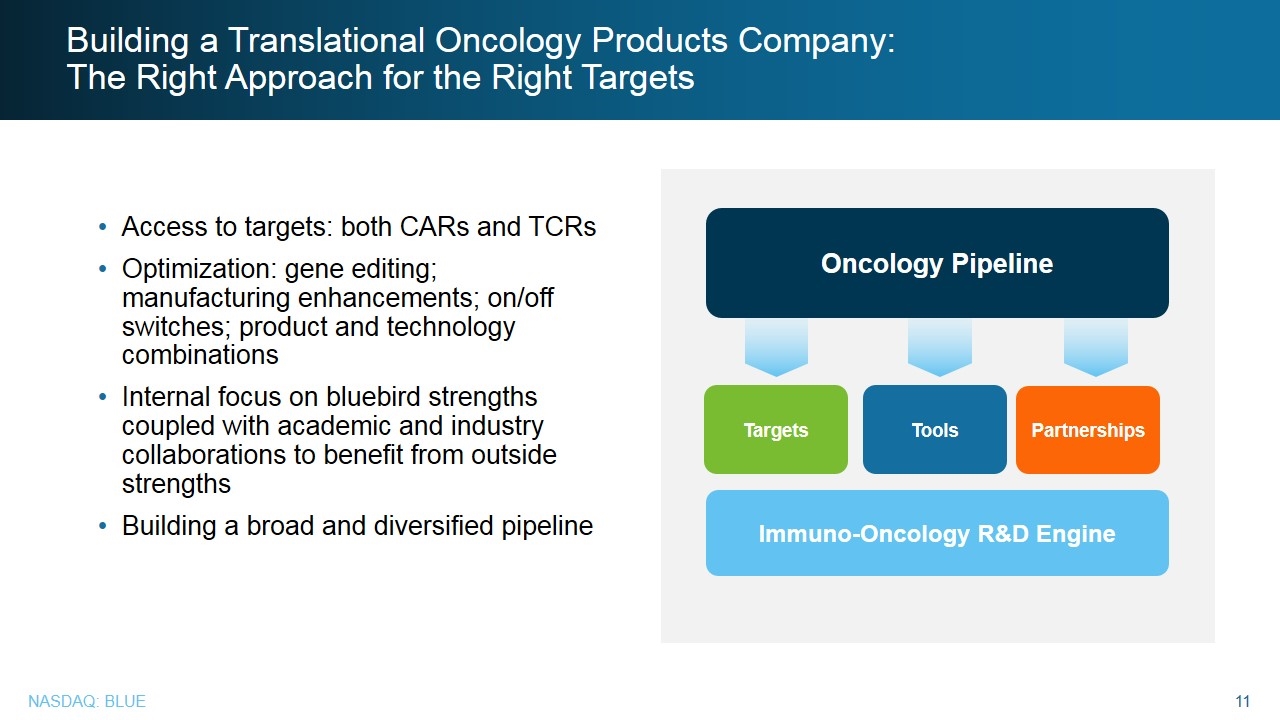
Building a Translational Oncology Products Company: The Right Approach for the Right Targets Targets Tools Partnerships Immuno-Oncology R&D Engine Oncology Pipeline Access to targets: both CARs and TCRs Optimization: gene editing; manufacturing enhancements; on/off switches; product and technology combinations Internal focus on bluebird strengths coupled with academic and industry collaborations to benefit from outside strengths Building a broad and diversified pipeline
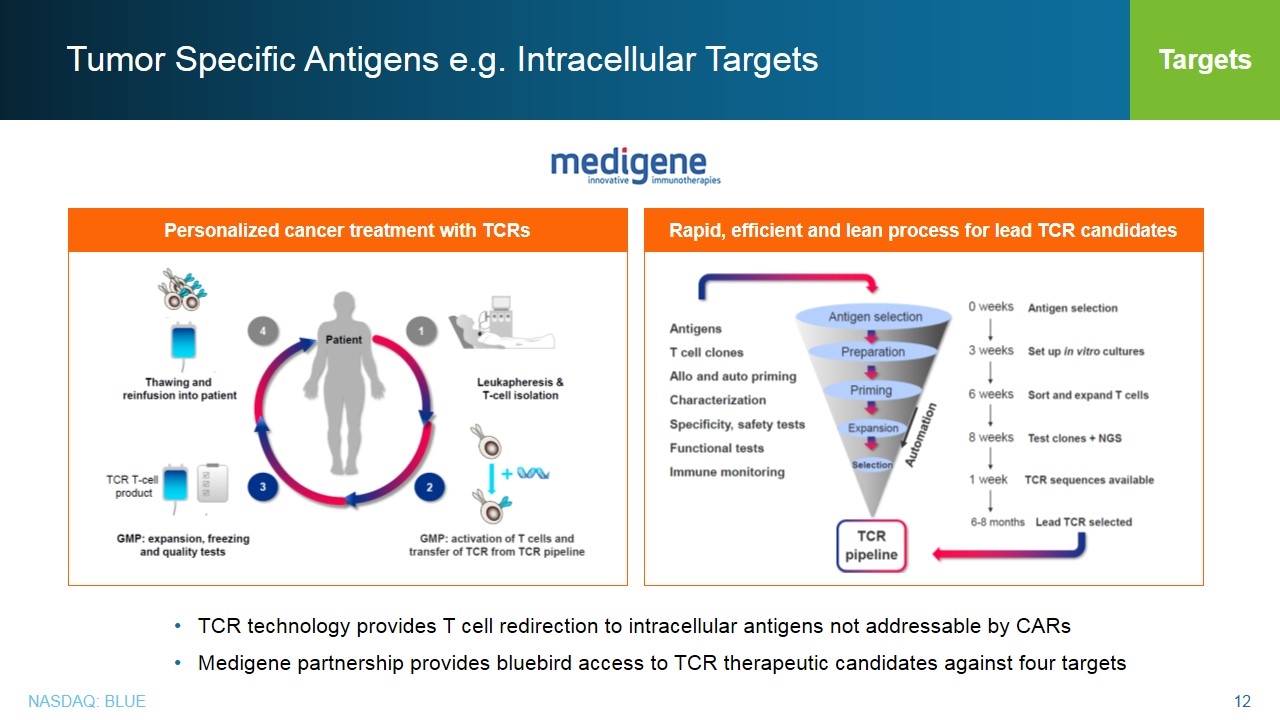
Tumor Specific Antigens e.g. Intracellular Targets TCR technology provides T cell redirection to intracellular antigens not addressable by CARs Medigene partnership provides bluebird access to TCR therapeutic candidates against four targets Targets Personalized cancer treatment with TCRs Rapid, efficient and lean process for lead TCR candidates
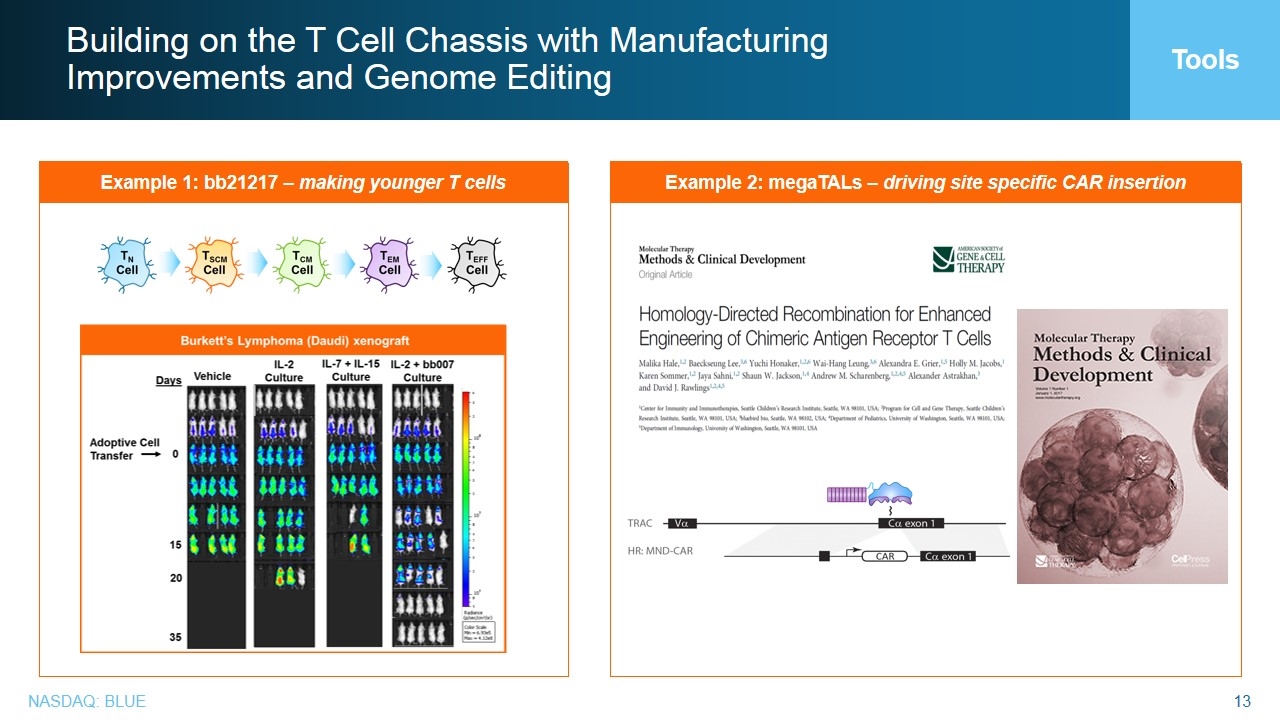
Example 1: bb21217 – making younger T cells Example 2: megaTALs – driving site specific CAR insertion Tools Building on the T Cell Chassis with Manufacturing Improvements and Genome Editing
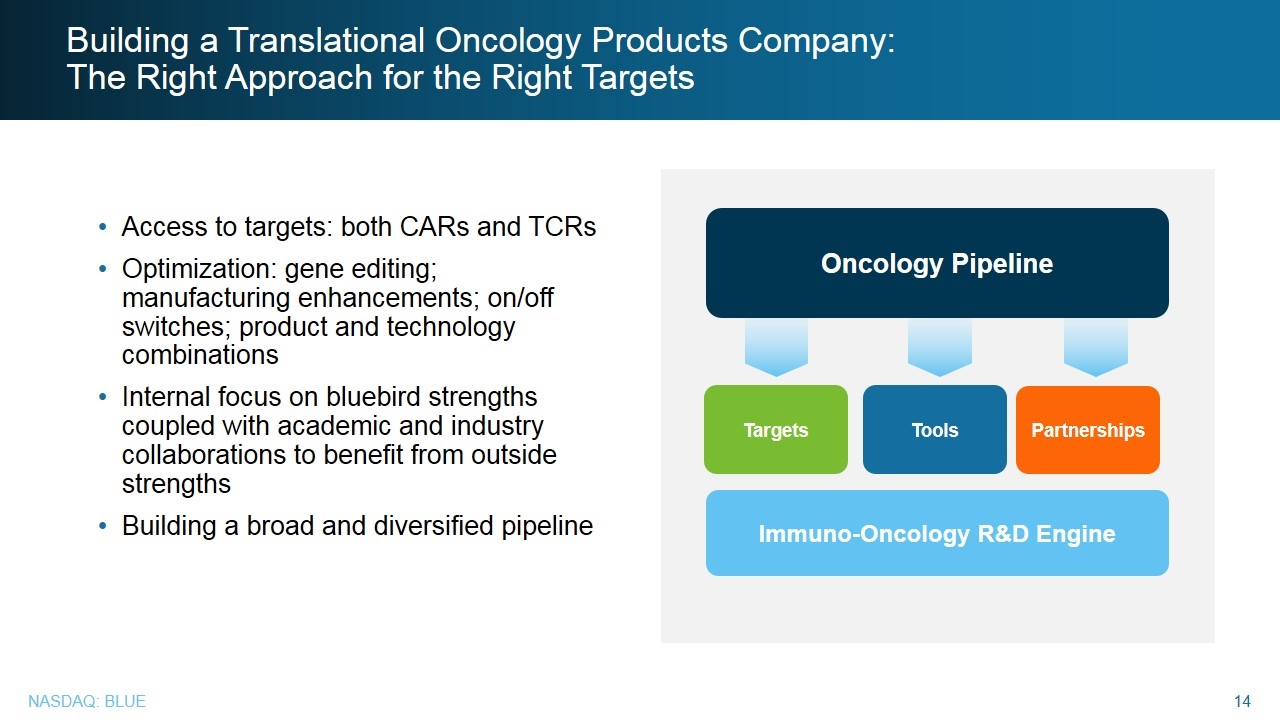
Building a Translational Oncology Products Company: The Right Approach for the Right Targets Access to targets: both CARs and TCRs Optimization: gene editing; manufacturing enhancements; on/off switches; product and technology combinations Internal focus on bluebird strengths coupled with academic and industry collaborations to benefit from outside strengths Building a broad and diversified pipeline Targets Tools Partnerships Immuno-Oncology R&D Engine Oncology Pipeline

CRB-401 Study Design and Clinical Overview M. Travis Quigley BCMA program leader & senior director, clinical development
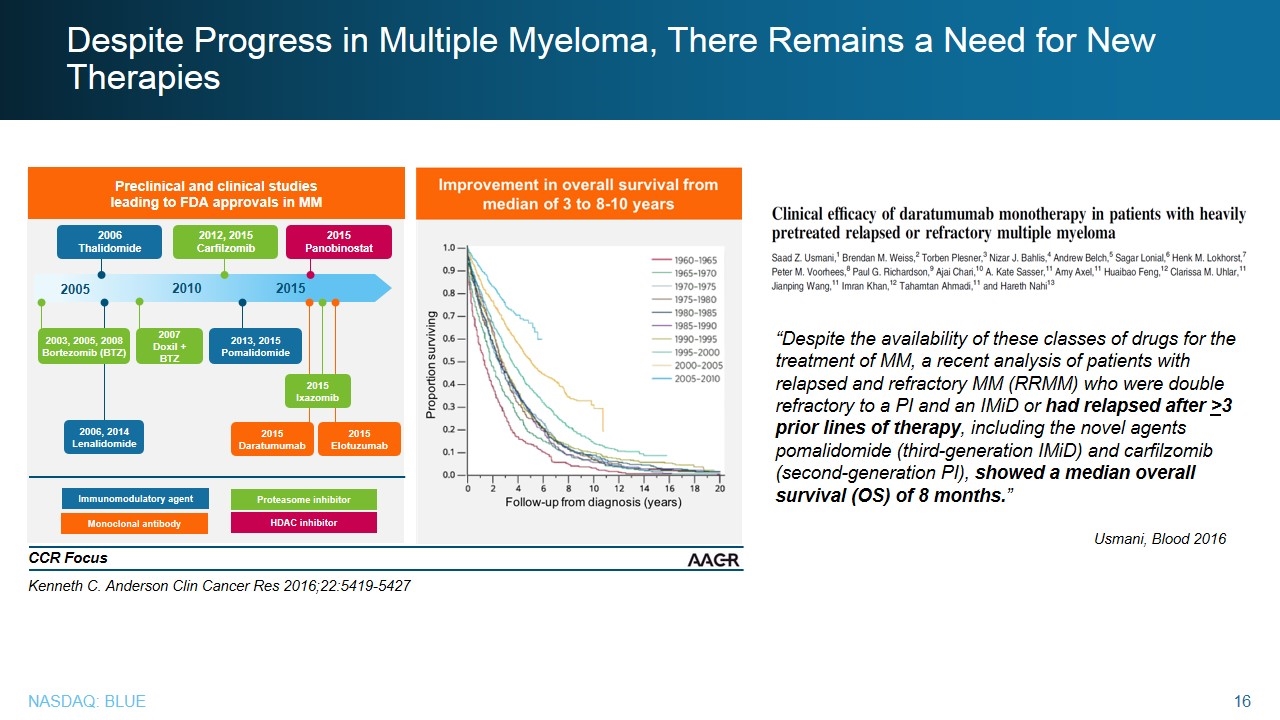
Despite Progress in Multiple Myeloma, There Remains a Need for New Therapies Kenneth C. Anderson Clin Cancer Res 2016;22:5419-5427 “Despite the availability of these classes of drugs for the treatment of MM, a recent analysis of patients with relapsed and refractory MM (RRMM) who were double refractory to a PI and an IMiD or had relapsed after >3 prior lines of therapy, including the novel agents pomalidomide (third-generation IMiD) and carfilzomib (second-generation PI), showed a median overall survival (OS) of 8 months.” Usmani, Blood 2016 Improvement in overall survival from median of 3 to 8-10 years Proportion surviving Follow-up from diagnosis (years) Preclinical and clinical studies leading to FDA approvals in MM 2015 Daratumumab 2015 Elotuzumab 2006 Thalidomide 2005 2010 2015 2007 Doxil + BTZ 2003, 2005, 2008 Bortezomib (BTZ) 2012, 2015 Carfilzomib 2015 Panobinostat 2013, 2015 Pomalidomide 2015 Ixazomib Immunomodulatory agent Monoclonal antibody Proteasome inhibitor HDAC inhibitor 2006, 2014 Lenalidomide CCR Focus © 2016 American Association for Cancer Research
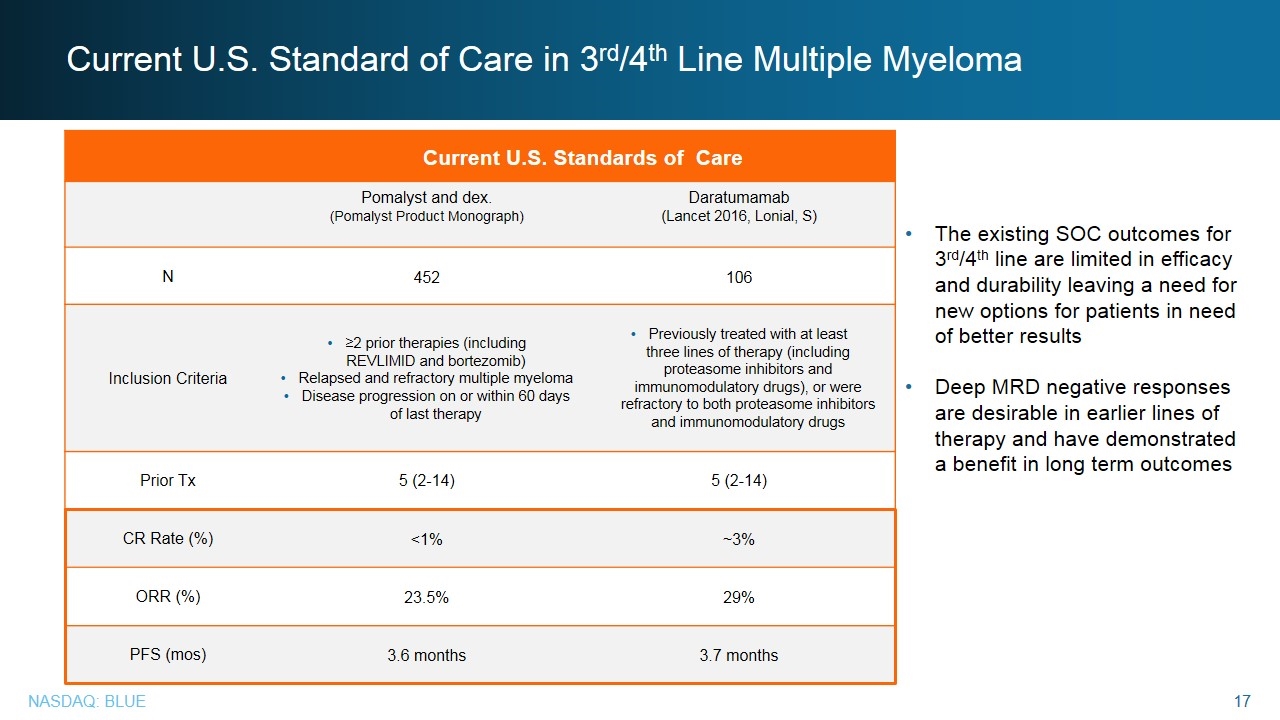
Current U.S. Standard of Care in 3rd/4th Line Multiple Myeloma Current U.S. Standards of Care Pomalyst and dex. (Pomalyst Product Monograph) Daratumamab (Lancet 2016, Lonial, S) N 452 106 Inclusion Criteria ≥2 prior therapies (including REVLIMID and bortezomib) Relapsed and refractory multiple myeloma Disease progression on or within 60 days of last therapy Previously treated with at least three lines of therapy (including proteasome inhibitors and immunomodulatory drugs), or were refractory to both proteasome inhibitors and immunomodulatory drugs Prior Tx 5 (2-14) 5 (2-14) CR Rate (%) <1% ~3% ORR (%) 23.5% 29% PFS (mos) 3.6 months 3.7 months The existing SOC outcomes for 3rd/4th line are limited in efficacy and durability leaving a need for new options for patients in need of better results Deep MRD negative responses are desirable in earlier lines of therapy and have demonstrated a benefit in long term outcomes

BCMA – A Promising Target in Multiple Myeloma Multiple myeloma cells expressing BCMA (brown color = BCMA protein) BCMA is member of the TNF receptor superfamily expressed nearly universally on multiple myeloma cells with expression largely restricted to plasma cells and some mature B cells Initial proof of anti-BCMA activity has been demonstrated using T cells transduced with a gamma-retroviral vector encoding an anti-BCMA CAR with a CD28 costimulatory domain, but significant cytokine release syndrome occurred in patients with high disease burden (Ali et al., Blood 2016) B-Cell Maturation Antigen (BCMA) Despite the availability of various approved therapies, including proteasome inhibitors, IMiDs and, more recently, anti-CD38 antibodies, multiple myeloma remains an incurable disease.
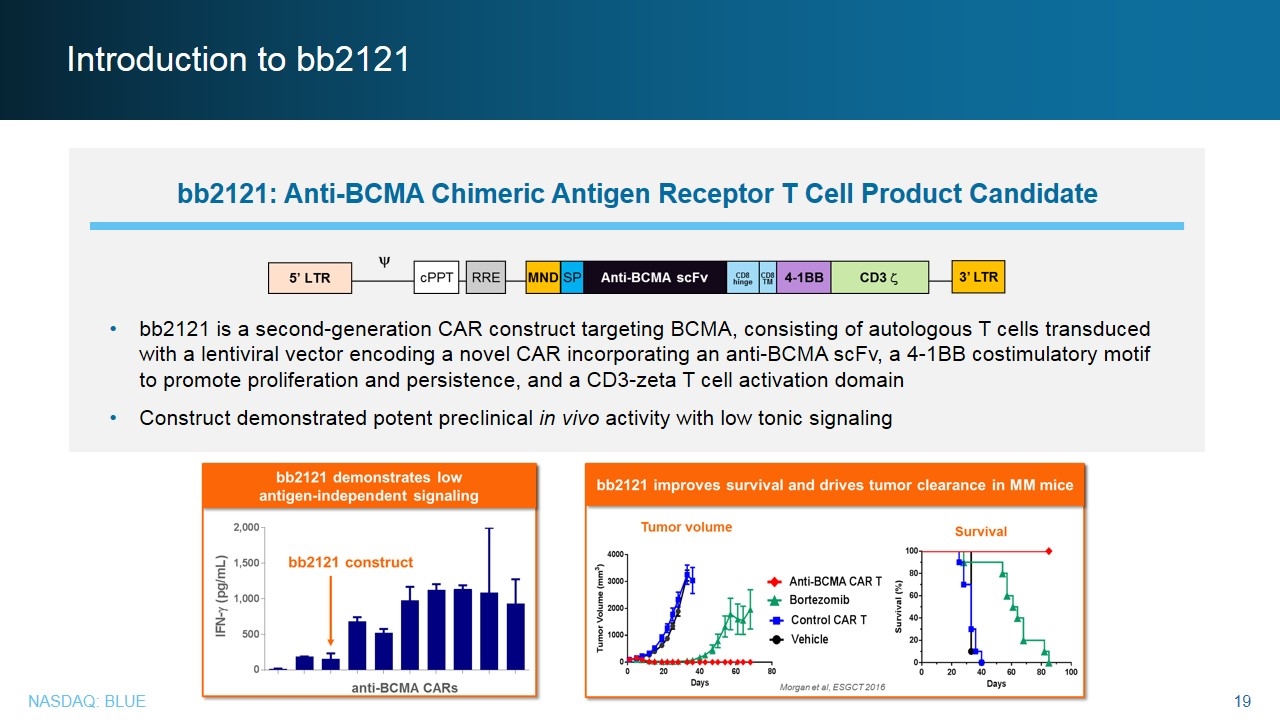
Introduction to bb2121 bb2121 is a second-generation CAR construct targeting BCMA, consisting of autologous T cells transduced with a lentiviral vector encoding a novel CAR incorporating an anti-BCMA scFv, a 4-1BB costimulatory motif to promote proliferation and persistence, and a CD3-zeta T cell activation domain Construct demonstrated potent preclinical in vivo activity with low tonic signaling bb2121: Anti-BCMA Chimeric Antigen Receptor T Cell Product Candidate

CRB-401 Phase 1 Study in Relapsed / Refractory Multiple Myeloma Objectives: Determine preliminary safety and efficacy and recommended phase 2 dose N = 50 patients, standard 3 + 3 dose escalation + expansion cohort Eligibility: Relapsed / refractory MM with ≥ 3 prior lines of therapy (including PI and IMiD), or double refractory Measurable disease ≥ 50% BCMA expression 9 U.S. Clinical Sites, 1 Centralized Manufacturing Site CRB-401 Open-label Phase 1 Clinical Study of bb2121

CRB-401 Data Jesus Berdeja, M.D. Sarah Cannon Research Institute & Tennessee Oncology

First-in-Human Multicenter Study of bb2121 anti-BCMA CAR T Cell Therapy for Relapsed/Refractory Multiple Myeloma: Updated Results Jesus G. Berdeja, MD1, Yi Lin, MD, PhD2, Noopur Raje, MD3, Nikhil Munshi, MD4, David Siegel, MD, PhD5, Michaela Liedtke, MD6, Sundar Jagannath, MD7, Marcela Maus, MD, PhD3, Ashley Turka8, Lyh Ping Lam8, Kristen Hege, MD9, Richard Morgan, PhD8, M. Travis Quigley8, and James N. Kochenderfer, MD10 1-Sarah Cannon Research Institute and Tennessee Oncology, Nashville, TN; 2-Mayo Clinic, Rochester, MN; 3-Massachusetts General Hospital Cancer Center, Boston, MA; 4-Dana Farber Cancer Institute, Boston, MA; 5-Hackensack University Medical Center, Hackensack, NJ; 6-Stanford University Medical Center, Palo Alto, CA; 7-Mt. Sinai Medical Center, New York, NY; 8-bluebird bio, Inc., Cambridge, MA; 9-Celgene, San Francisco, CA; 10-Experimental Transplantation and Immunology Branch, National Cancer Institute/National Institutes of Health, Bethesda, MD
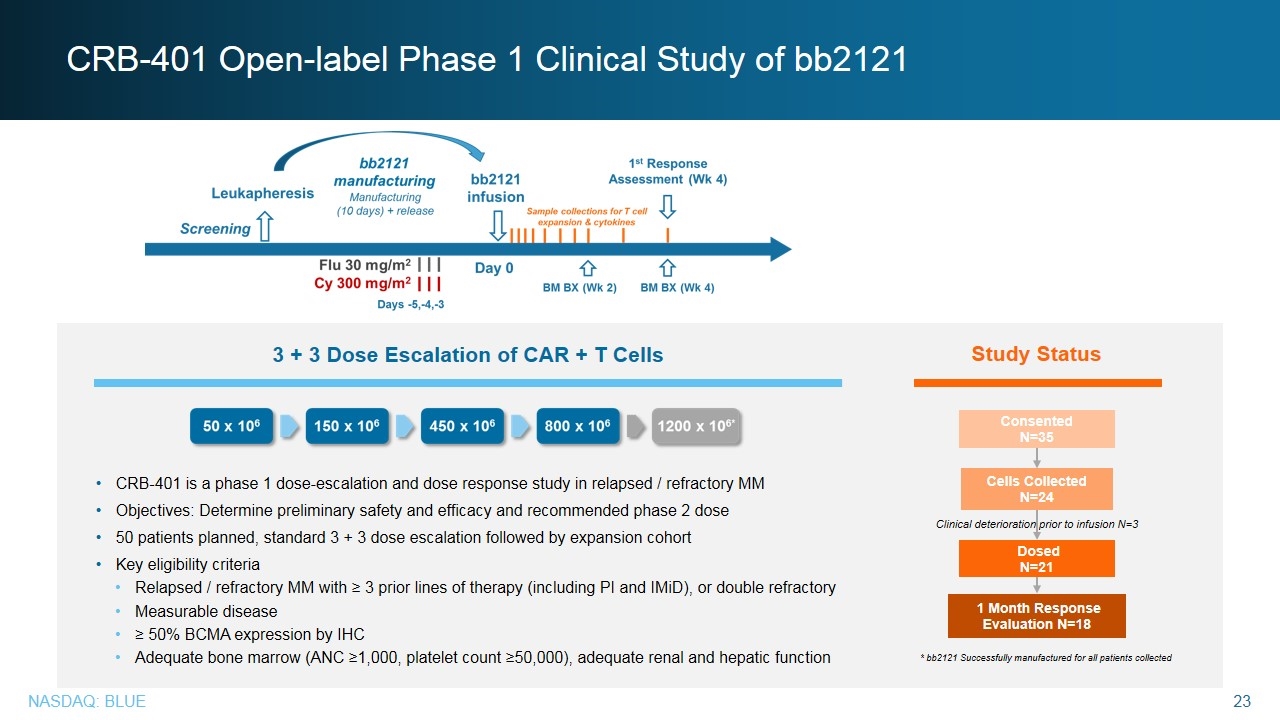
CRB-401 Open-label Phase 1 Clinical Study of bb2121 Study Status CRB-401 is a phase 1 dose-escalation and dose response study in relapsed / refractory MM Objectives: Determine preliminary safety and efficacy and recommended phase 2 dose 50 patients planned, standard 3 + 3 dose escalation followed by expansion cohort Key eligibility criteria Relapsed / refractory MM with ≥ 3 prior lines of therapy (including PI and IMiD), or double refractory Measurable disease ≥ 50% BCMA expression by IHC Adequate bone marrow (ANC ≥1,000, platelet count ≥50,000), adequate renal and hepatic function 3 + 3 Dose Escalation of CAR + T Cells Consented N=35 Cells Collected N=24 Dosed N=21 Clinical deterioration prior to infusion N=3 1 Month Response Evaluation N=18 * bb2121 Successfully manufactured for all patients collected
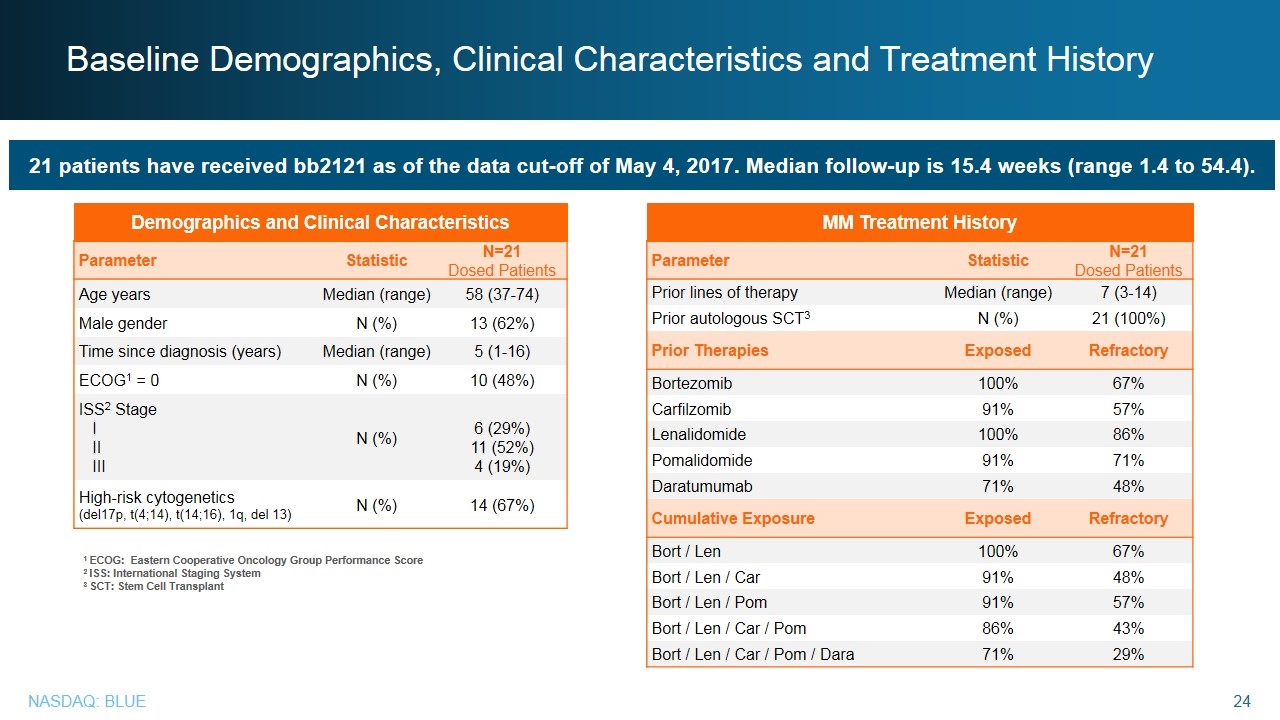
Baseline Demographics, Clinical Characteristics and Treatment History 21 patients have received bb2121 as of the data cut-off of May 4, 2017. Median follow-up is 15.4 weeks (range 1.4 to 54.4). 1 ECOG: Eastern Cooperative Oncology Group Performance Score 2 ISS: International Staging System 3 SCT: Stem Cell Transplant Parameter Statistic N=21 Dosed Patients Age years Median (range) 58 (37-74) Male gender N (%) 13 (62%) Time since diagnosis (years) Median (range) 5 (1-16) ECOG1 = 0 N (%) 10 (48%) ISS2 Stage I II III N (%) 6 (29%) 11 (52%) 4 (19%) High-risk cytogenetics (del17p, t(4;14), t(14;16), 1q, del 13) N (%) 14 (67%) Demographics and Clinical Characteristics Parameter Statistic N=21 Dosed Patients Prior lines of therapy Median (range) 7 (3-14) Prior autologous SCT3 N (%) 21 (100%) Prior Therapies Exposed Refractory Bortezomib 100% 67% Carfilzomib 91% 57% Lenalidomide 100% 86% Pomalidomide 91% 71% Daratumumab 71% 48% Cumulative Exposure Exposed Refractory Bort / Len 100% 67% Bort / Len / Car 91% 48% Bort / Len / Pom 91% 57% Bort / Len / Car / Pom 86% 43% Bort / Len / Car / Pom / Dara 71% 29% MM Treatment History
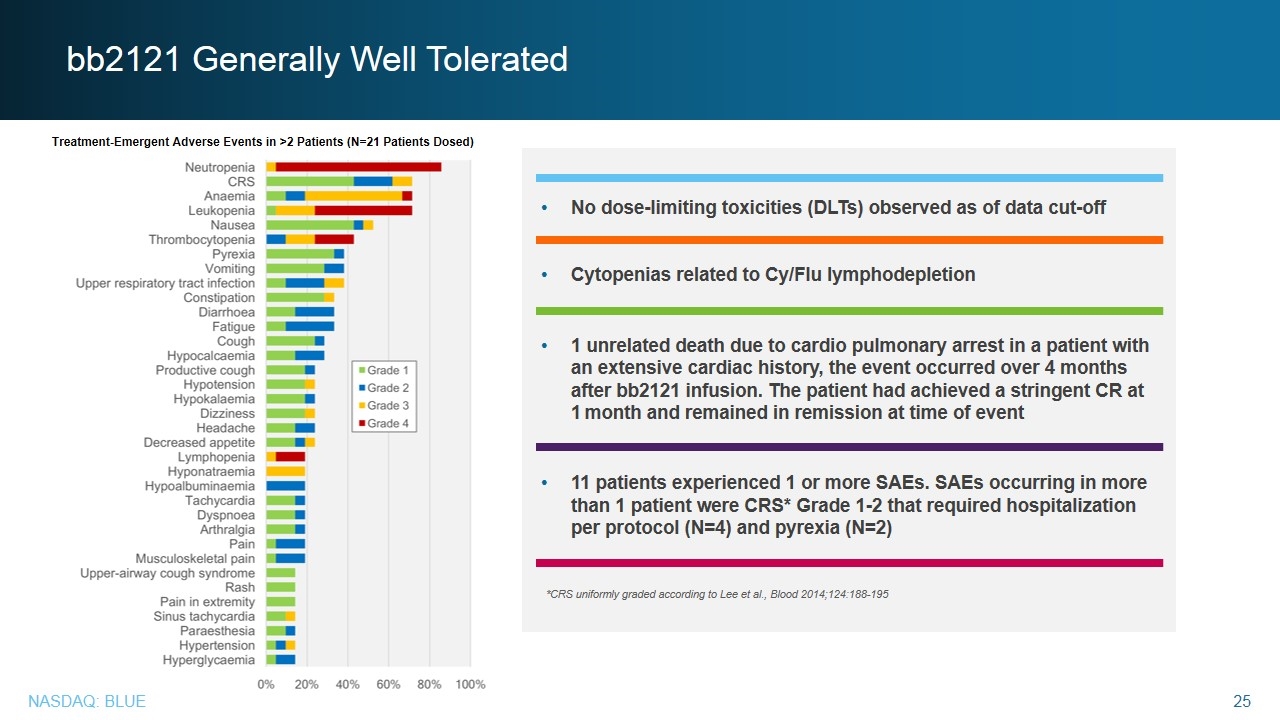
No dose-limiting toxicities (DLTs) observed as of data cut-off Cytopenias related to Cy/Flu lymphodepletion 1 unrelated death due to cardio pulmonary arrest in a patient with an extensive cardiac history, the event occurred over 4 months after bb2121 infusion. The patient had achieved a stringent CR at 1 month and remained in remission at time of event 11 patients experienced 1 or more SAEs. SAEs occurring in more than 1 patient were CRS* Grade 1-2 that required hospitalization per protocol (N=4) and pyrexia (N=2) bb2121 Generally Well Tolerated *CRS uniformly graded according to Lee et al., Blood 2014;124:188-195 Treatment-Emergent Adverse Events in >2 Patients (N=21 Patients Dosed)
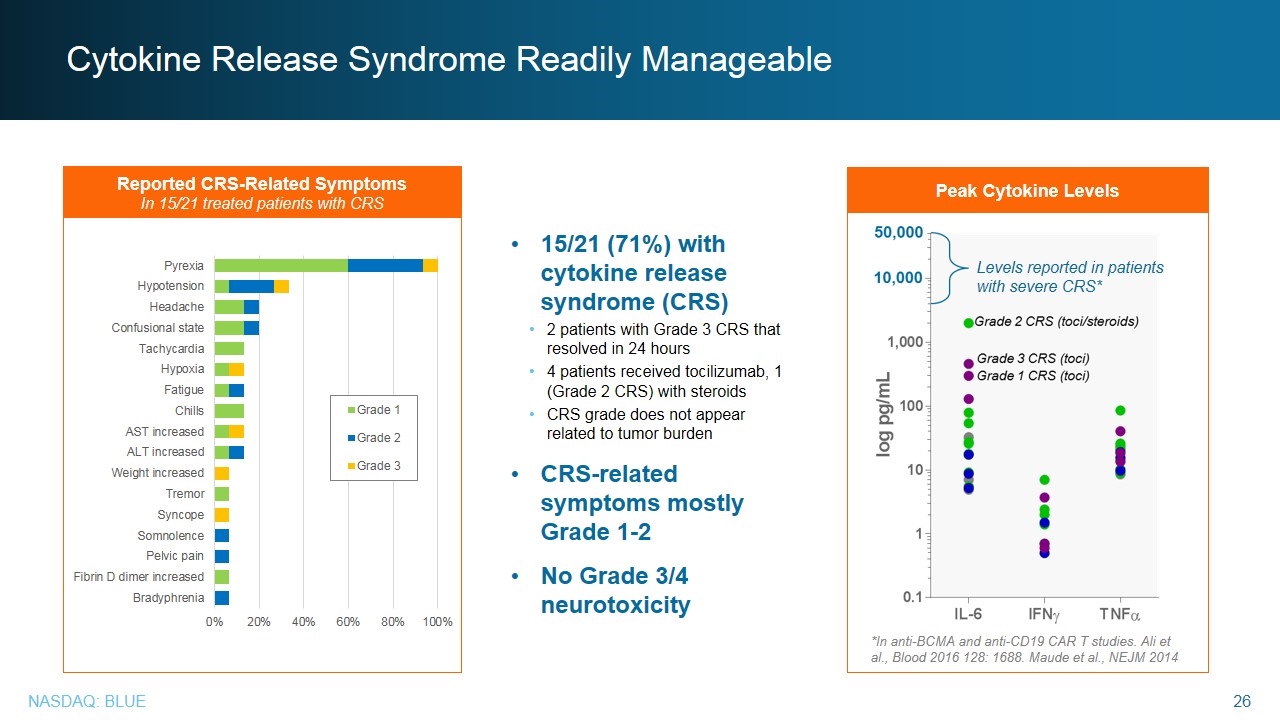
Cytokine Release Syndrome Readily Manageable 15/21 (71%) with cytokine release syndrome (CRS) 2 patients with Grade 3 CRS that resolved in 24 hours 4 patients received tocilizumab, 1 (Grade 2 CRS) with steroids CRS grade does not appear related to tumor burden CRS-related symptoms mostly Grade 1-2 No Grade 3/4 neurotoxicity Reported CRS-Related Symptoms In 15/21 treated patients with CRS *In anti-BCMA and anti-CD19 CAR T studies. Ali et al., Blood 2016 128: 1688. Maude et al., NEJM 2014 Peak Cytokine Levels Levels reported in patients with severe CRS*
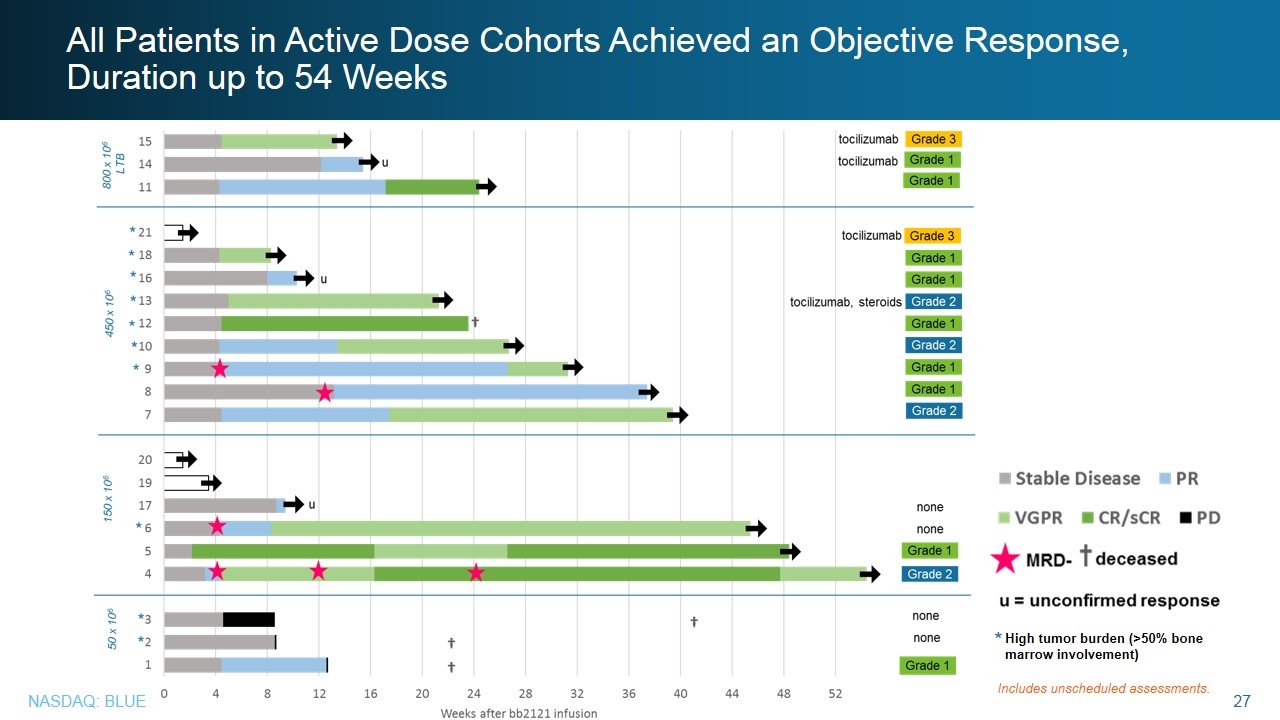
All Patients in Active Dose Cohorts Achieved an Objective Response, Duration up to 54 Weeks * High tumor burden (>50% bone marrow involvement) Includes unscheduled assessments.
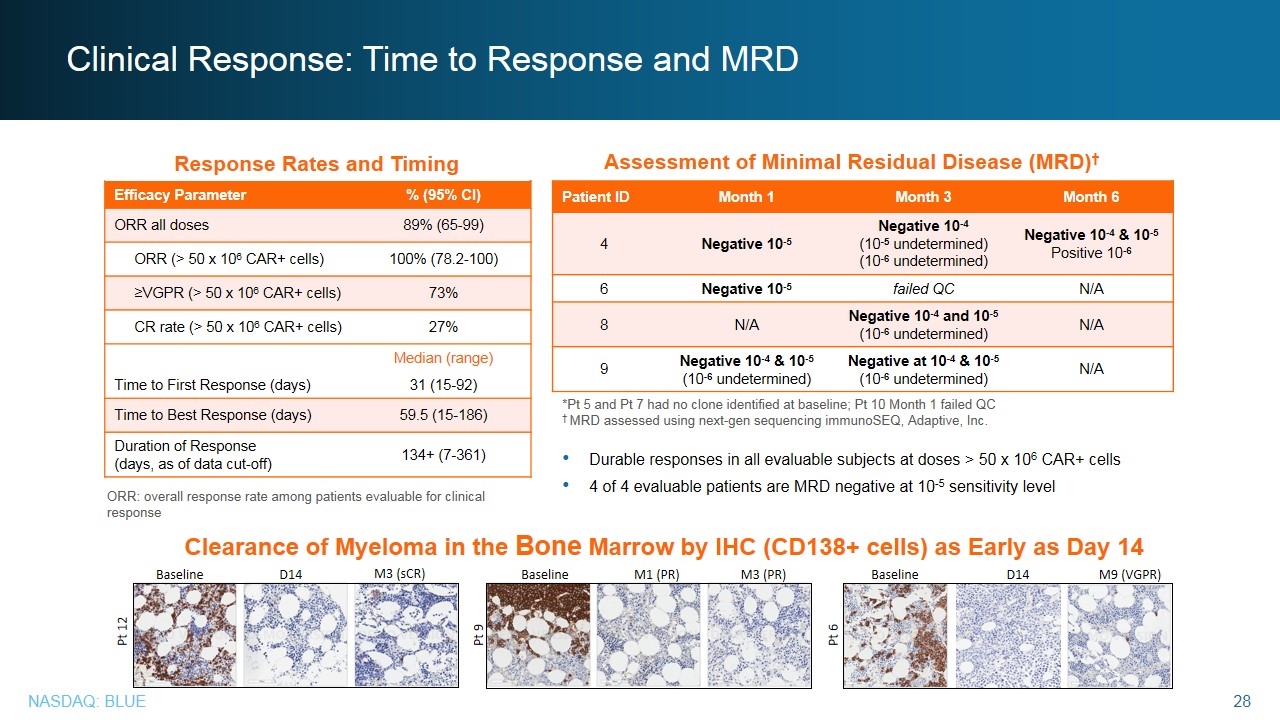
Clinical Response: Time to Response and MRD Efficacy Parameter % (95% CI) ORR all doses 89% (65-99) ORR (> 50 x 106 CAR+ cells) 100% (78.2-100) ≥VGPR (> 50 x 106 CAR+ cells) 73% CR rate (> 50 x 106 CAR+ cells) 27% Median (range) Time to First Response (days) 31 (15-92) Time to Best Response (days) 59.5 (15-186) Duration of Response (days, as of data cut-off) 134+ (7-361) Patient ID Month 1 Month 3 Month 6 4 Negative 10-5 Negative 10-4 (10-5 undetermined) (10-6 undetermined) Negative 10-4 & 10-5 Positive 10-6 6 Negative 10-5 failed QC N/A 8 N/A Negative 10-4 and 10-5 (10-6 undetermined) N/A 9 Negative 10-4 & 10-5 (10-6 undetermined) Negative at 10-4 & 10-5 (10-6 undetermined) N/A *Pt 5 and Pt 7 had no clone identified at baseline; Pt 10 Month 1 failed QC † MRD assessed using next-gen sequencing immunoSEQ, Adaptive, Inc. Durable responses in all evaluable subjects at doses > 50 x 106 CAR+ cells 4 of 4 evaluable patients are MRD negative at 10-5 sensitivity level Assessment of Minimal Residual Disease (MRD)† ORR: overall response rate among patients evaluable for clinical response Response Rates and Timing Pt 9 M1 (PR) M3 (PR) Baseline Pt 12 Baseline D14 M3 (sCR) Pt 6 D14 Baseline M9 (VGPR) Clearance of Myeloma in the Bone Marrow by IHC (CD138+ cells) as Early as Day 14
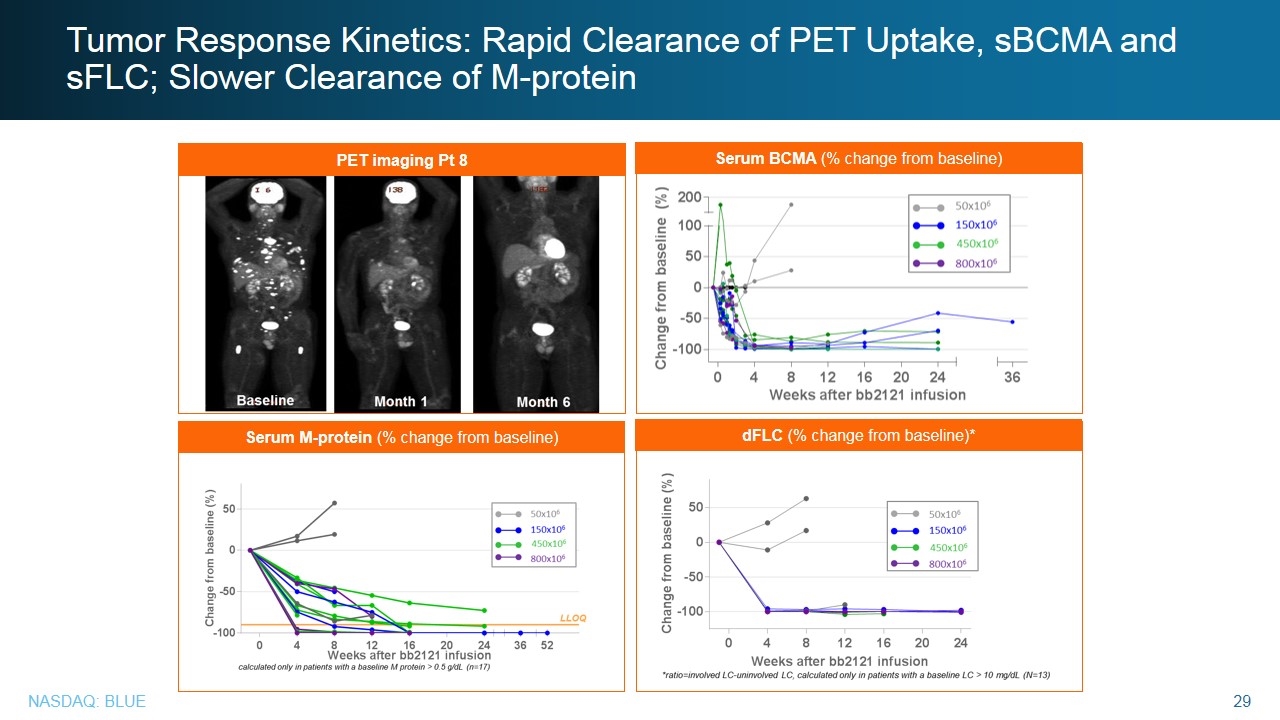
Tumor Response Kinetics: Rapid Clearance of PET Uptake, sBCMA and sFLC; Slower Clearance of M-protein PET imaging Pt 8 dFLC (% change from baseline)* Serum M-protein (% change from baseline) Serum BCMA (% change from baseline)
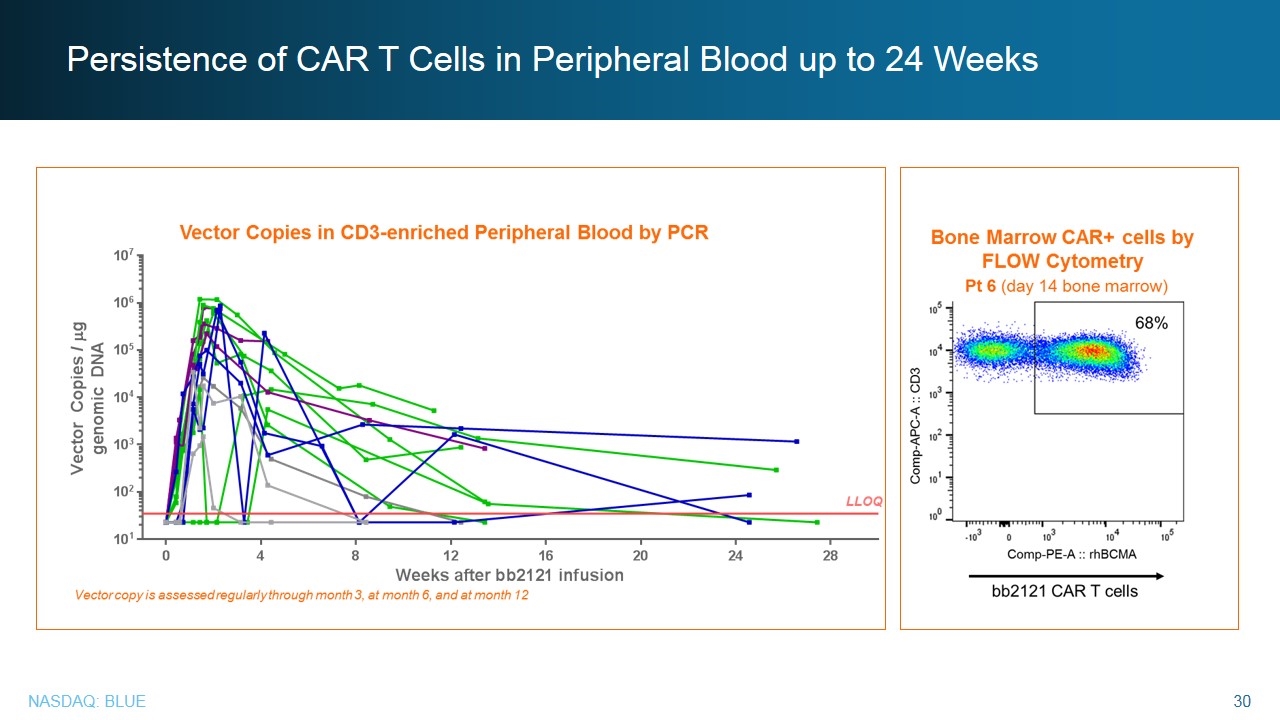
Persistence of CAR T Cells in Peripheral Blood up to 24 Weeks

Summary bb2121 has induced durable and deepening responses in a heavily pre-treated population with relapsed/refractory multiple myeloma, including: 100% ORR, 73% VGPR or better, 27% CR (at doses > 50 x 106) MRD negative results in all evaluable patients (N=4) No disease progression in patients treated with doses > 50 x 106, with 1 patient past 1 year and 8 patients past 6 months To date, the safety profile of bb2121 has been manageable through doses as high as 800 x 106 The 2 reported events of grade 3 CRS resolved within 24 hours No grade 3/4 neurotoxicity reported These results will inform identification of the dose(s) to bring forward into the expansion phase of the study in future development

Closing David Davidson, M.D., chief medical officer

Key Takeaways As of May 4 Data Cut-off No DLTs; toxicity readily manageable, CRS largely grade 1/2, two Grade 3 CRS resolved within 24 hours with tocilizumab Safety Profile Responses Durability 100% response rate in active dose cohorts (above 50x106) 27% complete response rate 73% VGPR or better All patients tested for MRD status (N=4) were found to be MRD-negative No patients with clinical progression in active dose cohorts (above 50x106) Heavily pre-treated; all had prior auto transplant, median of 7 prior lines of therapy Patient Population

Q&A

Q&A Nick Leschly, chief bluebird David Davidson, M.D., chief medical officer Philip Gregory, D.Phil., chief scientific officer M. Travis Quigley, BCMA program leader & senior director, clinical development Jesus Berdeja, M.D., Sarah Cannon Research Institute & Tennessee Oncology Michael Pehl, president, hematology & oncology, Celgene