
INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Interim Consolidated Financial Statements June 30, 2014 Exhibit 99.2
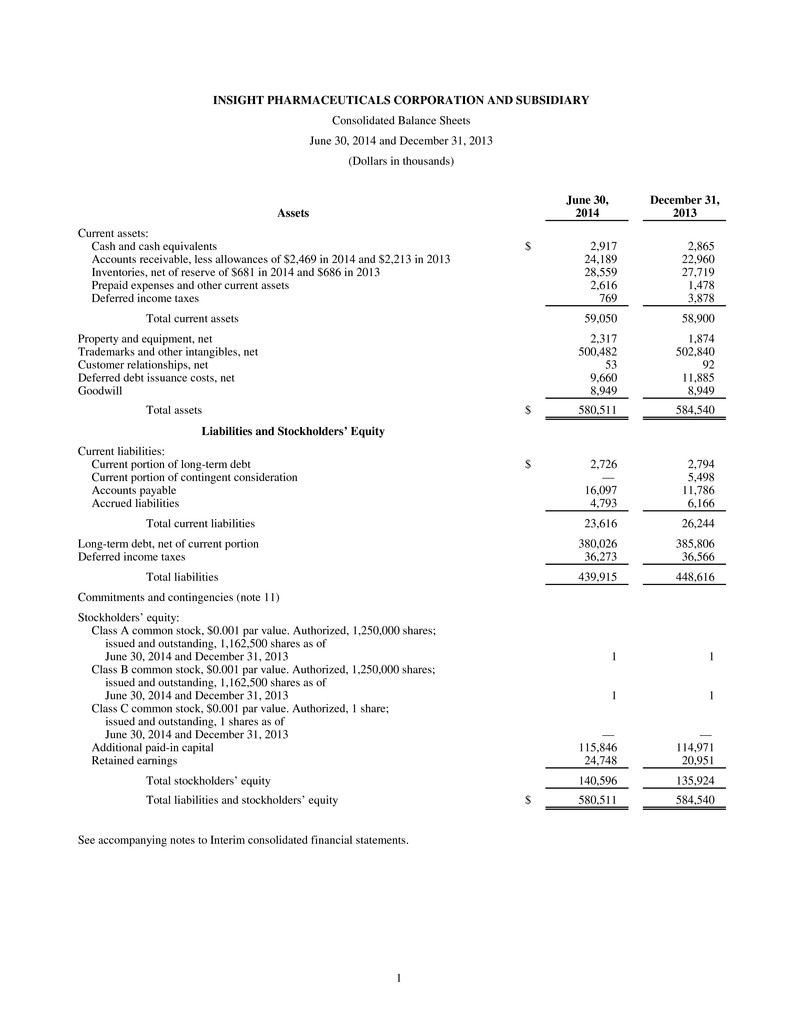
1 INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Consolidated Balance Sheets June 30, 2014 and December 31, 2013 (Dollars in thousands) June 30, December 31, Assets 2014 2013 Current assets: Cash and cash equivalents $ 2,917 2,865 Accounts receivable, less allowances of $2,469 in 2014 and $2,213 in 2013 24,189 22,960 Inventories, net of reserve of $681 in 2014 and $686 in 2013 28,559 27,719 Prepaid expenses and other current assets 2,616 1,478 Deferred income taxes 769 3,878 Total current assets 59,050 58,900 Property and equipment, net 2,317 1,874 Trademarks and other intangibles, net 500,482 502,840 Customer relationships, net 53 92 Deferred debt issuance costs, net 9,660 11,885 Goodwill 8,949 8,949 Total assets $ 580,511 584,540 Liabilities and Stockholders’ Equity Current liabilities: Current portion of long-term debt $ 2,726 2,794 Current portion of contingent consideration — 5,498 Accounts payable 16,097 11,786 Accrued liabilities 4,793 6,166 Total current liabilities 23,616 26,244 Long-term debt, net of current portion 380,026 385,806 Deferred income taxes 36,273 36,566 Total liabilities 439,915 448,616 Commitments and contingencies (note 11) Stockholders’ equity: Class A common stock, $0.001 par value. Authorized, 1,250,000 shares; issued and outstanding, 1,162,500 shares as of June 30, 2014 and December 31, 2013 1 1 Class B common stock, $0.001 par value. Authorized, 1,250,000 shares; issued and outstanding, 1,162,500 shares as of June 30, 2014 and December 31, 2013 1 1 Class C common stock, $0.001 par value. Authorized, 1 share; issued and outstanding, 1 shares as of June 30, 2014 and December 31, 2013 — — Additional paid-in capital 115,846 114,971 Retained earnings 24,748 20,951 Total stockholders’ equity 140,596 135,924 Total liabilities and stockholders’ equity $ 580,511 584,540 See accompanying notes to Interim consolidated financial statements.
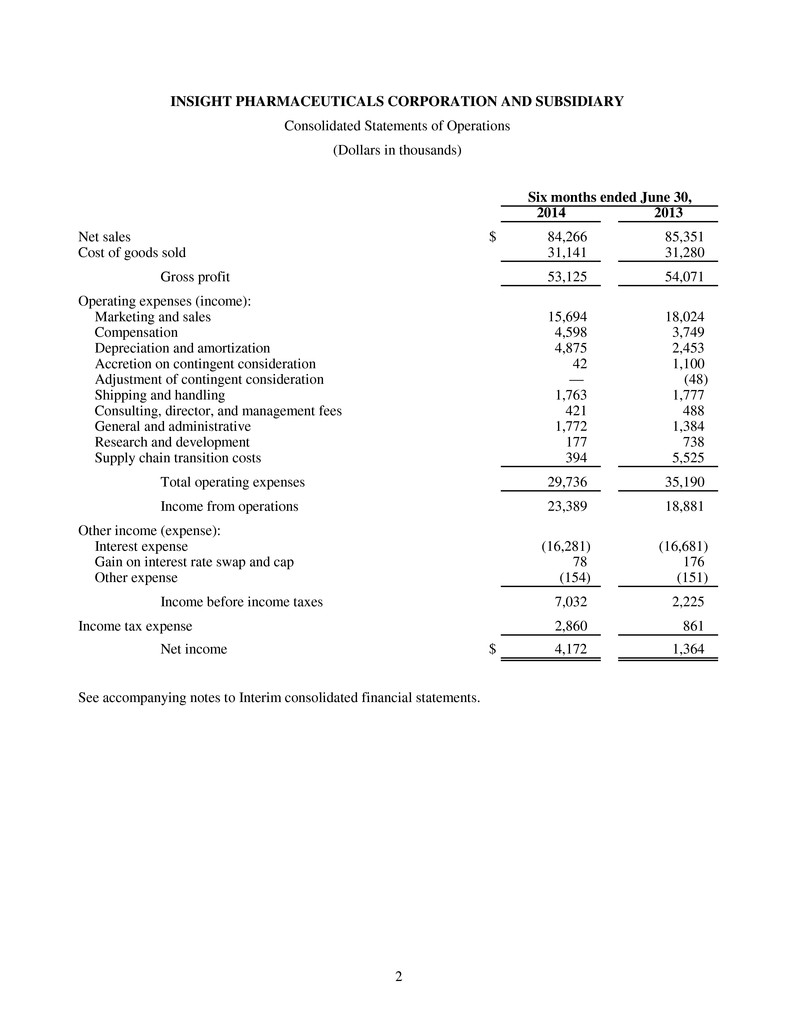
2 INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Consolidated Statements of Operations (Dollars in thousands) Six months ended June 30, 2014 2013 Net sales $ 84,266 85,351 Cost of goods sold 31,141 31,280 Gross profit 53,125 54,071 Operating expenses (income): Marketing and sales 15,694 18,024 Compensation 4,598 3,749 Depreciation and amortization 4,875 2,453 Accretion on contingent consideration 42 1,100 Adjustment of contingent consideration — (48) Shipping and handling 1,763 1,777 Consulting, director, and management fees 421 488 General and administrative 1,772 1,384 Research and development 177 738 Supply chain transition costs 394 5,525 Total operating expenses 29,736 35,190 Income from operations 23,389 18,881 Other income (expense): Interest expense (16,281) (16,681) Gain on interest rate swap and cap 78 176 Other expense (154) (151) Income before income taxes 7,032 2,225 Income tax expense 2,860 861 Net income $ 4,172 1,364 See accompanying notes to Interim consolidated financial statements.
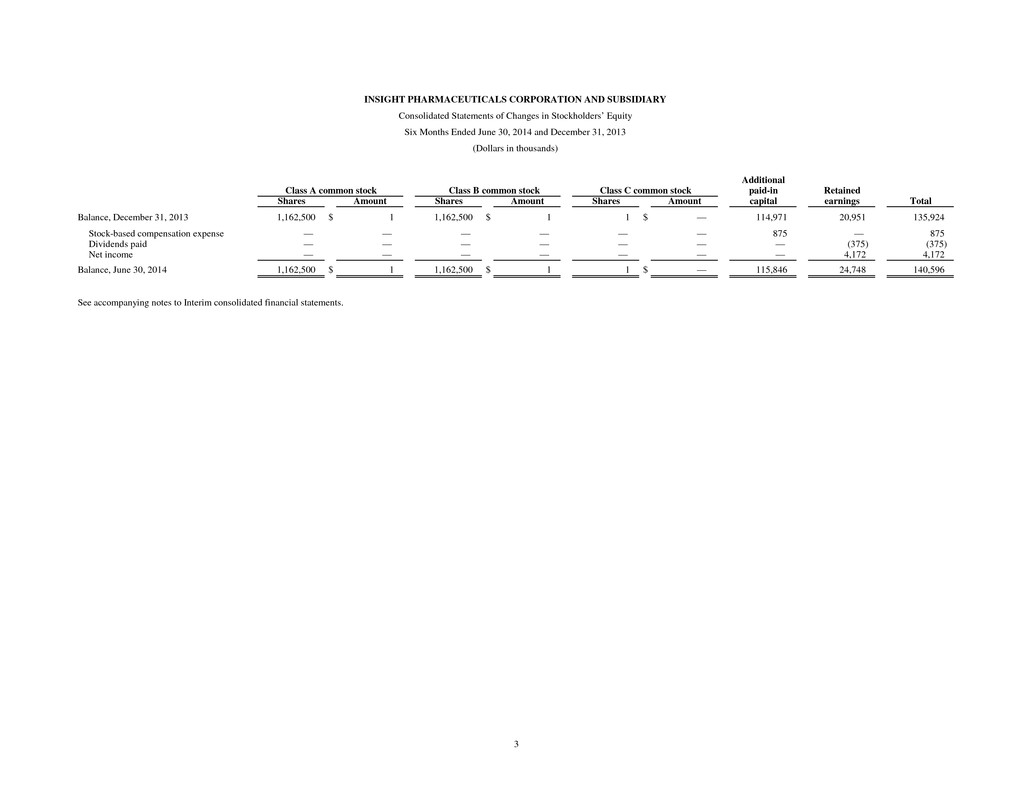
3 INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Consolidated Statements of Changes in Stockholders’ Equity Six Months Ended June 30, 2014 and December 31, 2013 (Dollars in thousands) Additional Class A common stock Class B common stock Class C common stock paid-in Retained Shares Amount Shares Amount Shares Amount capital earnings Total Balance, December 31, 2013 1,162,500 $ 1 1,162,500 $ 1 1 $ — 114,971 20,951 135,924 Stock-based compensation expense — — — — — — 875 — 875 Dividends paid — — — — — — — (375) (375) Net income — — — — — — — 4,172 4,172 Balance, June 30, 2014 1,162,500 $ 1 1,162,500 $ 1 1 $ — 115,846 24,748 140,596 See accompanying notes to Interim consolidated financial statements.
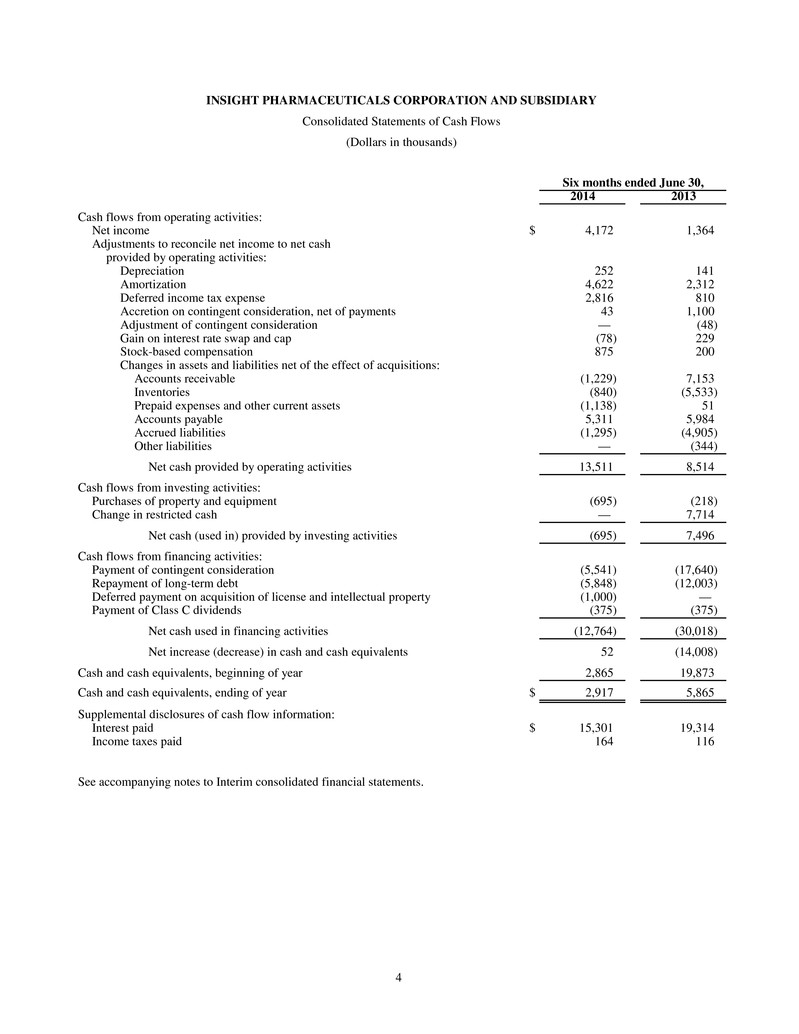
4 INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Consolidated Statements of Cash Flows (Dollars in thousands) Six months ended June 30, 2014 2013 Cash flows from operating activities: Net income $ 4,172 1,364 Adjustments to reconcile net income to net cash provided by operating activities: Depreciation 252 141 Amortization 4,622 2,312 Deferred income tax expense 2,816 810 Accretion on contingent consideration, net of payments 43 1,100 Adjustment of contingent consideration — (48) Gain on interest rate swap and cap (78) 229 Stock-based compensation 875 200 Changes in assets and liabilities net of the effect of acquisitions: Accounts receivable (1,229) 7,153 Inventories (840) (5,533) Prepaid expenses and other current assets (1,138) 51 Accounts payable 5,311 5,984 Accrued liabilities (1,295) (4,905) Other liabilities — (344) Net cash provided by operating activities 13,511 8,514 Cash flows from investing activities: Purchases of property and equipment (695) (218) Change in restricted cash — 7,714 Net cash (used in) provided by investing activities (695) 7,496 Cash flows from financing activities: Payment of contingent consideration (5,541) (17,640) Repayment of long-term debt (5,848) (12,003) Deferred payment on acquisition of license and intellectual property (1,000) — Payment of Class C dividends (375) (375) Net cash used in financing activities (12,764) (30,018) Net increase (decrease) in cash and cash equivalents 52 (14,008) Cash and cash equivalents, beginning of year 2,865 19,873 Cash and cash equivalents, ending of year $ 2,917 5,865 Supplemental disclosures of cash flow information: Interest paid $ 15,301 19,314 Income taxes paid 164 116 See accompanying notes to Interim consolidated financial statements.

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) (1) Nature of Business Insight Pharmaceuticals Corporation and Subsidiary (hereinafter referred to as the Company or Insight) markets, distributes, and sells quality healthcare products to consumers, principally through the drug, mass merchandising, and food channels of distribution. The Company outsources its warehousing and distribution operations and employs contract manufacturers to make products that are sold primarily in North America. (2) Summary of Significant Accounting Policies (a) Principles of Consolidation The consolidated financial statements of the Company include the accounts of Insight Pharmaceuticals Corporation and its wholly owned subsidiary, Insight Pharmaceuticals, LLC. All significant intercompany accounts and transactions have been eliminated in consolidation. (b) Revenue Recognition Revenue is recognized when the customer takes ownership and assumes risk of loss, collection of the relevant receivable is probable, persuasive evidence of an arrangement exists, and the sales price is fixed and determinable. Net sales include the sales price, net of promotional allowances specifically identified for the related products sold, less sales returns, pricing credits, unsalables, and miscellaneous sales adjustments. Sales returns and pricing credits are directly attributable to the products sold. The Company estimates its sales returns allowance based on past experience. (c) Cash and Cash Equivalents and Restricted Cash For purposes of reporting cash flows, the Company considers all highly liquid debt instruments purchased with a maturity of three months or less to be cash equivalents. The Company maintains its cash and cash equivalents in bank deposit accounts, which, at times, may exceed federally insured limits. The Company does not believe it is exposed to any significant credit risk on cash and cash equivalents. (d) Accounts Receivable Trade receivables are carried at original invoice amount less an estimate made for doubtful receivables and other allowances. Management determines the allowance for doubtful accounts by regularly evaluating individual customer receivables and considering a customer’s financial condition, credit history, and current economic conditions. Trade receivables are written off when deemed uncollectible. Recoveries of trade receivables previously written off are recorded when cash is received. (e) Inventories Inventories are stated at the lower of cost, determined on the first-in, first-out method, or market. The Company records as a charge to cost of goods sold any amount required to reduce the carrying value of inventories to the net realizable sales value. 5 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) (f) Property and Equipment Property and equipment are stated at cost. Depreciation is computed on a straight-line basis over the following estimated useful lives: Furniture and fixtures 7 years Machinery and equipment 3 years Computer equipment and software 3 years (g) Impairment of Long-Lived Assets The Company reviews long-lived assets, including property and equipment and intangible assets subject to amortization, for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable. Recoverability of assets is measured by a comparison of the carrying amount of an asset to estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated future cash flows, then an impairment charge is recognized for the amount by which the carrying value of the asset exceeds the fair value of the asset. (h) Intangible Assets Intangible assets consist of goodwill, trademarks, and customer relationships. Goodwill reflects the costs in excess of the fair value of the net assets acquired in purchase transactions. Trademarks are related to the purchase of over-the-counter pharmaceutical brands. The Company accounts for goodwill and trademarks in accordance with Accounting Standards Codification (ASC) 350, Intangibles – Goodwill and Other. The Company performs its annual impairment test date as of September 30 each year. The Company tests goodwill for impairment using the two-step process prescribed by ASC 350. The first step tests for potential impairment and the second step measures the amount of impairment, if any. There was no impairment of goodwill during 2014 or 2013. The Company annually evaluates current events and circumstances (including period to period sales and profitability by brand, industry trends, projected sales and gross margins, and advertising effectiveness) to determine whether the infinite life classification for a trademark continues to be valid. If the Company determines the indefinite life classification is no longer valid, the Company will begin to amortize the trademark over its estimated useful life. The Company tests its indefinite lived trademarks for impairment annually, or if events or circumstances indicate the trademarks might be impaired. During 2013, based on the results of the annual impairment test, the Company recognized an impairment of $1,400. No impairments have seen recognized during the six months ended June 30, 2014, See note 5. Customer relationships are being amortized on an accelerated basis over an estimated five-year life. 6 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) (i) Deferred Debt Issuance Costs Costs incurred in connection with procuring debt, consisting primarily of debt origination fees, are deferred and amortized as a charge to income, over the term of the related debt, using the effective-interest method. (j) Income Taxes Income taxes are accounted for under the asset and liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases and operating loss and tax credit carryforwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. The Company recognizes the effect of income tax positions only if those positions are more likely than not of being sustained. Recognized income tax positions are measured at the largest amount that is greater than 50% likely of being realized. Changes in recognition or measurement are reflected in the period in which the change in judgment occurs. (k) Derivatives Certain of the Company’s derivative instruments do not qualify for hedge accounting treatment under the Financial Accounting Standards Board guidance for derivatives and hedging; for others, the Company chooses not to maintain the required documentation to apply hedge accounting treatment. These undesignated instruments are used to economically hedge the Company’s exposure to fluctuations in the interest rates on certain portions of the Company’s floating rate debt. The Company’s derivative policy permits the use of undesignated derivatives and the change in fair value is reported currently through earnings in gain or loss on interest rate swap and cap on the Company’s consolidated statements of operations. At December 31, 2013, the notional principal amount of the swap was $27,550, which reduces as debt principal payments are made, and which matured on April 1, 2014. For the six month ended June 30, 2014 and June 30, 2013, the Company recorded a gain of $78 and $176 on this instrument, respectively. A swap liability of $78 is included in the accompanying consolidated balance sheets as of December 31, 2013. The fair value of the Company’s interest rate swap was the estimated amount the Company would receive or pay to terminate these agreements at the reporting date, taking into account current interest rates and the creditworthiness of the Company. See note 13 for additional disclosure on the fair value of this interest rate swap. At December 31, 2013, the notional principal amount of the cap was $167,350 which reduces as debt principal payments are made, and which matured on January 1, 2014. For the six months ended June 30, 2014 and 2013, the Company recorded no gain or loss on this instrument. The fair value of the Company’s cap is the estimated amount the Company would receive or pay to terminate these agreements at the reporting date, taking into account current interest rates and the creditworthiness of 7 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) the counterparty for assets and creditworthiness of the Company for liabilities. See note 13 for additional disclosure on the fair value of this cap. (l) Marketing and Sales Expenses Marketing and sales costs are expensed as incurred. Marketing and sales expenses represent various costs associated with selling the Company’s products including broker commissions, travel expenses, advertising, and promotion costs. Advertising costs represent various media expenses to promote brand awareness and were $10,631 and $12,253 for the six months ended June 30, 2014 and June 30, 2013, respectively. Promotion costs are composed of marketing costs including displays, market research expenditures, and package design costs. (m) Research and Development Cost Research and development costs are charged to operations in the period incurred and were $177 and $738 for the six months ended June 30, 2014 and June 30, 2013, respectively. (n) Supply Chain Transition Costs The Company incurred costs of $394 and $5,525 for the six months ended June 30, 2014 and June 30, 2013, respectively related to switching suppliers for one of its main products. The costs primarily relate to termination penalties from the old supplier, but also include costs for packaging changes, consulting fees, and salary expense for employees working on the transition. (o) Stock-Based Compensation The Company has a stock option plan, which is accounted for under ASC 718, Compensation – Stock Compensation, which addresses accounting for equity-based compensation arrangements, including employee stock options, and requires entities to measure the cost of employee services received in exchange for awards of equity instruments at the grant date of the award using a fair-value-based method. The Company measures employee stock-based awards at grant-date fair value and records employee compensation expense on a straight-line basis over the vesting period of the award. Determining the appropriate fair value of stock-based awards requires the input of subjective assumptions, including the fair value of the Company’s common stock and for stock options, the expected life of the option, and expected stock price volatility. The Company uses the Black-Scholes option pricing model to value its stock option awards. The assumptions used in calculating the fair value of stock-based awards represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and management uses different assumptions, stock-based compensation expense could be materially different for future awards. The expected life of stock options was estimated using the “simplified method,” as the Company has no historical information to develop reasonable expectations about future exercise patterns and post vesting employment termination behavior for its stock options grants. The simplified method is 8 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) based on the average of the vesting tranches and the contractual life of each grant. For stock price volatility, the Company uses comparable public companies as a basis for its expected volatility to calculate the fair value of options grants. The risk-free interest rate is based on U.S. Treasury notes with a term approximating the expected life of the option. He dividend yield of zero is based on the fact that the Company has never paid cash dividends on its common stock, and has no present intention to pay cash dividends. (p) Use of Estimates The preparation of consolidated financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates. (3) Acquisitions On August 26, 2011, the Company acquired substantially all of the assets, consisting primarily of intellectual property and inventory used in connection with the Monistat brand from McNEIL-PPC, Inc. (the Seller) for $353,453, including contingent consideration with an estimated fair value of $68,399. The Company financed this transaction and refinanced its then existing debt, with an additional capital contribution of $65,000, and a new credit facility totaling $420,000 ($255,000 First Lien, $145,000 Second Lien, and $20,000 Revolving Loan Commitment) as further described in note 6. The contingent consideration arrangement required the Company to pay quarterly amounts to the Seller based on gross sales of Monistat in 2013 and 2014, up to a maximum amount of $90,000. During 2014 and 2013, the Company paid the Seller $5,541 and $28,825, respectively, under the arrangement. The remaining estimated undiscounted amount of payments to be paid under the contingent consideration arrangement at December 31, 2013 was $5,541. For the six months ended June 30, 2014 and 2013, the Company accreted $42 and $1,100, respectively, of interest expense related to this liability which is recorded as accretion on contingent consideration on the consolidated statements of operations. For the six month ended June 30, 2013 the Company recognized $48 of income as a result of a decrease in the estimated amounts due for contingent consideration due to a decrease in Monistat gross revenues, net of other miscellaneous adjustments. The Company recorded $5,498, as the estimated fair value of the payments in the consolidated balance sheets as of December 31, 2013. As the final payment was made in January 2014, the liability was zero as of June 30, 2014. The acquisition was recorded in accordance with ASC 805, Business Combinations. As a result of the acquisition, the Company increased its market share in quality healthcare products sold to consumers using preexisting infrastructure and distribution channels established by the Company. On September 4, 2013, the Company entered into an exclusive licensing agreement with Common Sense Limited. This agreement provides the Company exclusive access to patented technology in connection with women’s health and home diagnostics, and to otherwise use, make, distribute, offer for sale, promote, advertise, or import the products in the United States. Upon execution of the license agreement, the Company paid Common Sense Limited $1,500. An additional $1,000 was paid to Common Sense Limited 9 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) in March 2014. The Company will pay up to an additional $3,500 in milestone payments upon the achievement of certain regulatory and commercialization events for products to be sold over-the-counter. There are also royalty payments to be paid at varying percentages of net sales for these products, which range from 5% to 7% depending on the year. (4) Property and Equipment Property and equipment consisted of the following as of June 30, 2014 and December 31, 2013: June 30, December 31, 2014 2013 Furniture and fixtures $ 332 332 Machinery and equipment 1,456 1,296 Computer equipment and software 1,575 1,040 3,363 2,668 Less accumulated depreciation 1,046 794 $ 2,317 1,874 Depreciation expense for the six months ended June 30, 2014 and 2013 was $252 and $141, respectively. (5) Intangible Assets Intangible assets subject to amortization as of June 30, 2014 consist of the following: Weighted average Gross Net amortization carrying Accumulated carrying period amount amortization amount Customer relationships 5 $ 865 (812) 53 Trademarks subject to amortization 13 57,662 (3,537) 54,125 Intangible assets subject to amortization as of December 31, 2013 consist of the following: Weighted average Gross Net amortization carrying Accumulated carrying period amount amortization amount Customer relationships 5 $ 865 (773) 92 Trademarks subject to amortization 13 57,662 (1,179) 56,483 10 (Continued)
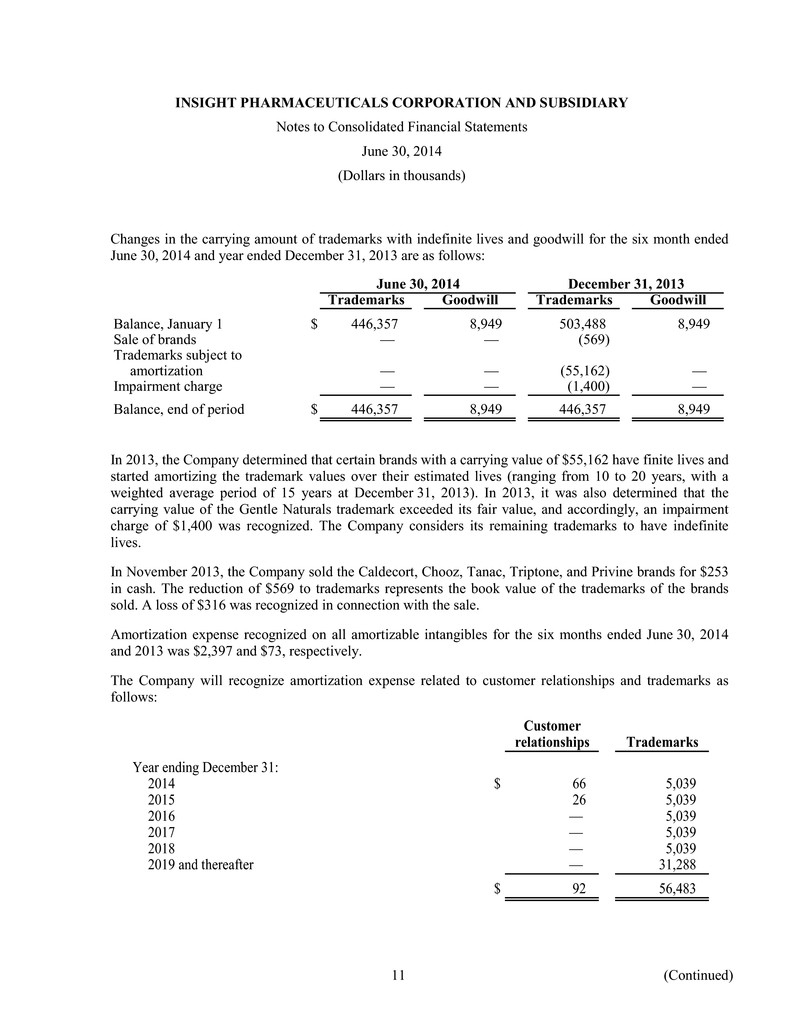
INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) Changes in the carrying amount of trademarks with indefinite lives and goodwill for the six month ended June 30, 2014 and year ended December 31, 2013 are as follows: June 30, 2014 December 31, 2013 Trademarks Goodwill Trademarks Goodwill Balance, January 1 $ 446,357 8,949 503,488 8,949 Sale of brands — — (569) Trademarks subject to amortization — — (55,162) — Impairment charge — — (1,400) — Balance, end of period $ 446,357 8,949 446,357 8,949 In 2013, the Company determined that certain brands with a carrying value of $55,162 have finite lives and started amortizing the trademark values over their estimated lives (ranging from 10 to 20 years, with a weighted average period of 15 years at December 31, 2013). In 2013, it was also determined that the carrying value of the Gentle Naturals trademark exceeded its fair value, and accordingly, an impairment charge of $1,400 was recognized. The Company considers its remaining trademarks to have indefinite lives. In November 2013, the Company sold the Caldecort, Chooz, Tanac, Triptone, and Privine brands for $253 in cash. The reduction of $569 to trademarks represents the book value of the trademarks of the brands sold. A loss of $316 was recognized in connection with the sale. Amortization expense recognized on all amortizable intangibles for the six months ended June 30, 2014 and 2013 was $2,397 and $73, respectively. The Company will recognize amortization expense related to customer relationships and trademarks as follows: Customer relationships Trademarks Year ending December 31: 2014 $ 66 5,039 2015 26 5,039 2016 — 5,039 2017 — 5,039 2018 — 5,039 2019 and thereafter — 31,288 $ 92 56,483 11 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) (6) Debt Long-term debt as of June 30, 2014 and December 31, 2013 is as follows: June 30, December 31, 2014 2013 First Lien $ 268,502 276,600 Revolver 2,250 — Second Lien 112,000 112,000 382,752 388,600 Less current portion 2,726 2,794 Long-term debt, net of current portion $ 380,026 385,806 The Company has a credit facility totaling $422,000 ($290,000 First Lien, $112,000 Second Lien, $20,000 Revolving Loan Commitment). The interest on the First Lien is LIBOR plus 6.00% with a 1.50% floor to LIBOR plus 5.00% with a 1.25% floor. The First Lien requires quarterly principal payments of $698 on January 1 and April 1, 2014, and $682 commencing July 1, 2014 with the balance payable in August of 2016. Principal payments were reduced due to the Company making a $6,701 excess cash flow payment as required based on the results of the Leverage Ratio per the Company’s debt covenants. The First Lien bears interest at LIBOR plus an applicable margin (5% at June 30, 2014) and is subject to certain affirmative and negative covenants, which, among other matters, require the Company to maintain certain financial ratios, limit capital expenditures, limit distributions to the Company’s stockholders, limit fees paid to the Company’s directors, and limit management fees paid to the Company’s stockholders. The Second Lien bears interest at LIBOR plus an applicable margin (11.75% at June 30, 2014) and matures in August 2017. In addition, if any amount of the loan is paid prior to the due date, there will be a prepayment fee calculated as a percentage of the loan ranging from 3.00% to 1.00%. The loan is subject to certain affirmative and negative covenants, which, among other matters, require the Company to maintain certain financial ratios, limit capital expenditures, limit distributions to the Company’s stockholders, limit fees paid to the Company’s directors, and limit management fees paid to the Company’s stockholders. The revolving loan commitment of $20,000 bears interest at LIBOR plus 5.00%, has a due date of August 2016 and can be used to fund the contingent consideration payments (note 3) or to fund operations. As of June 30, 2014 and December 31, 2013, $2,250 and $0, respectively were outstanding under this revolving loan. Under the credit agreement terms, the Company is required to pay a quarterly commitment fee equal to 0.5% of the unused commitment. Under these credit facilities, the Company’s obligations are secured by a lien on substantially all of the Company’s assets. The Second Lien is subordinated to the Company’s First Lien. 12 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) The aggregate maturities of long-term debt are as follows as of June 30, 2014: 2014 $ 1,363 2015 2,726 2016 266,663 2017 112,000 Total $ 382,752 Amortization of deferred debt issuance costs for the six months ended June 30, 2014 and 2013, was $2,225 and $2,239, respectively. (7) Stockholders’ Equity (a) Common Stock The Company is authorized to issue 2,500,001 shares of common stock. This includes 1,250,000 shares of Class A common stock, par value $0.001 per share; 1,250,000 shares of Class B common stock, par value $0.001 per share; and 1 share of Class C common stock, par value $0.001 per share. In 2011, the Company issued 1,162,500 shares of the Class A common stock; 1,162,500 shares of the Class B common stock; and 1 share of the Class C common stock. Class A common stock shares have the right to vote on all matters with the exception of election of directors of the Company; the right to participate in dividends when declared by the board of directors of the Company except for special dividends paid to the owner of the Class C shares; and the right to participate in distributions upon the liquidation, dissolution, or winding-up of the affairs of the Company. Class B common stock shares have the right to vote for the election of directors of the Company and the right to receive $0.002 per share upon the liquidation, dissolution, or winding-up of the affairs of the Company. Class C common stock shares have the right to receive a special dividend on terms and conditions set forth in an agreement between the Company and shareholder and the right to vote on any matter that would adversely affect the rights and preferences of the Class C common stock with respect to the special dividends. (b) Stock Option Plan On June 17, 2009, the Company adopted a stock option plan (the Plan).The Plan is a compensation benefit plan designed to provide incentives to present and future employees, directors, officers, consultants, or advisors of the Company and falls within the meaning of Rule 701 of the Securities Act of 1933, as amended. A committee of the board of directors has the sole authority to grant options. 13 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) The Plan, as amended, authorizes a maximum of 100,000 shares to be granted and if any options previously granted are forfeited, they will become available for future grants. The Plan vests in increments of 20% per year on each anniversary date and ending on the fifth anniversary. No incentive stock options will be granted after June 17, 2019. Consistent with Section 422(b) of the Internal Revenue Code, the options (i) shall have an exercisable price per share of common stock of not less than 100% of the fair market value at the date of the grant, (ii) shall not be exercisable 10 years after date of grant, (iii) shall generally not be transferable other than by will or under the laws of descent and distribution and, during the lifetime of the participant, to whom such incentive stock options were granted, may be exercised only by such participant, and (iv) shall generally be exercisable only during the participant’s employment with certain exceptions. The Company used the Black-Scholes option pricing model to estimate the grant˗date fair value of the stock-based awards. As of June 30, 2014, there was $575 of unrecognized compensation costs related to share-based payments, which is expected to be recognized over a weighted average period of 3.5 years. Compensation expense was $890 and $200, respectively, for the six months ended June 30, 2014 and June 30, 2013. The weighted average grant-date fair value for stock options granted in 2013 was $78.15. There were no stock options graded in the six months ended June 30, 2014. The following table represents stock option activity for the six month period ended June 30, 2014: Weighted Weighted average average remaining Number of exercise contractual shares price life Balance as of December 31, 2013 81,306 95.49 Granted — — Exercised — — Forfeited — — Balance as of June 30, 2014 81,306 95.49 6.4 years Exercisable at June 30, 2014 53,233 83.16 6.0 years Shares available for future stock option grants to employees under the existing Plan were 18,694 as of June 30, 2014. 14 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) (8) Income Taxes The provision for income taxes is comprised of the following for the six months ended June 30, 2014 and, 2013: June 30, June 30, 2014 2013 Current $ 44 51 Deferred 2,816 810 $ 2,860 861 Current taxes arise principally from state income taxes. The provision for income taxes differs from the amount determined by applying the U.S. federal statutory rate to pretax income due primarily to state income taxes and non-deductible expenses. Deferred taxes primarily represent temporary differences related to net operating loss carryforwards, intangible assets, transaction related expenses, contingent consideration at December 31, 2013 and accrued expenses. At June 30, 2014, the Company has net operating loss carryforwards for federal income tax purposes of $53.8 million, which are available to offset future federal taxable income, if any, through 2033. The Company has net operating loss carryforwards for state income tax purposes of $39.1 million which are available to offset future state taxable income through 2033. Under the Tax Reform Act of 1986 and certain state statutes, the utilization of a corporation’s net operating loss carryforward is limited following a greater-than-50% change in ownership during a three-year period. Any unused annual limitation may be carried forward to future years for the balance of the net operating loss period. Under these rules, prior ownership changes may create a limitation in the Company’s ability to use certain tax carryforwards for federal and state income tax purposes. Certain state net operating losses may also be limited, including those related to Pennsylvania, which has a dollar limitation on the amount of net operating losses utilized in any one year. The Company recognized the tax benefit of uncertain tax positions only if it is more likely than not that the tax position will be sustained on examination by taxing authorities, based on the technical merits of the position, the tax benefits recognized in the consolidated financial statements from such a position are measured based on the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement. The guidance on accounting for uncertainty in income taxes also addresses derecognition, classification, interest and penalties on income taxes, and accounting in interim periods. Management has evaluated the Company’s historical tax positions and has concluded that the Company has taken no uncertain tax positions that require a reserve to be provided in these consolidated financial statements. 15 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) The Company files domestic federal and state income tax returns. For years prior to 2010, the Company is no longer subject to income tax examinations by the U.S. federal, state, or local taxing authorities. (9) Significant Customers Three customers represented approximately 60% of net sales for the six months ended June 30, 2014, with three customers representing 26%, 19%, and 15%. These three customers represented approximately 62% of accounts receivable as of June 30, 2014. Three customers represented approximately 60% of net sales for the six month ended June 30, 2013, with three customers representing 29%, 18%, and 13%. These three customers represented approximately 61% of accounts receivable as of December 31, 2013. (10) Related-Party Transactions The Company was party to a management agreement with the Company’s Former stockholders, pursuant to which the Company paid an annual base fee of $750 plus dividends of $750 on Class C shares. The fees and dividends are payable quarterly and the Company is billed for expenses and additional special projects as incurred, subject to limitations under the terms of the Company’s credit facilities. Total amounts paid with respect to the management agreement were management fees and expenses of $421 and $488 for the six months ended June 30, 2014 and 2013, respectively, and Class C dividends of $375 and $375 for the six months ended June 30, 2014 and 2013, respectively. Effective January 1, 2011, the Company engaged The Emerson Group and Emerson Healthcare (Emerson) to provide selling and logistics related services to the Company. A principal of Emerson subsequently became a stockholder and was a member of the Company’s Board of Directors. Fees paid for the six months ended June 30, 2014 and 2013 to Emerson in connection to services provided to the Company were $3,617 and $3,474, respectively. (11) Commitments and Contingencies The Company has a lease for office space. Future minimum lease payments under the Company’s operating lease agreements for its office space is as follows: Year ending December 31: 2014 $ 189 2015 387 2016 394 2017 402 2018 409 Total $ 1,781 Rent expense for the six months ended June 30, 2014 and 2013 was $175 and $156, respectively. The Company is party to various claims arising in the ordinary course of business. Although the ultimate outcome of the matters is presently not determinable, management, after consultation with legal counsel, 16 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) does not believe that the outcome of these matters will have a material adverse effect on the Company’s financial position or results of operations. (12) Benefits The Company has a 401(k) “safe harbor” plan that covers all employees and allows for a maximum match of 6% of compensation or a maximum of $17 per individual. For the six months ended June 30, 2014 and 2013, the Company contributed $143 and $123, respectively, to the plan. (13) Fair Value of Financial Instruments Accounts receivable, accounts payable, and accrued expenses are reflected in the consolidated financial statements at carrying value, which approximates fair value because of the short-term nature of these instruments. The Company believes that the carrying value of the Company’s long-term debt approximates fair value based on the variable rates and the current rates available to the Company for similar instruments. The estimated carrying and fair values of the Company’s financial instruments are as follows at December 31, 2013. December 31, 2013 Carrying Estimated value fair value Financial liabilities: Interest rate swap $ (78) (78) The Company had no other outstanding financial instruments at June 30, 2014. The fair value of the interest rate cap and swap are the estimated amount the Company would receive (pay) to terminate the agreements at the reporting date, taking into account current interest rates. The Company also considers the creditworthiness of the cap and swap counterparties for assets and creditworthiness of the Company for liabilities. The Company follows ASC 820, Fair Value Measurements and Disclosures, and applies it to all assets and liabilities that are being measured and reported on a fair value basis. The statement requires that assets and liabilities carried at fair value will be classified and disclosed in one of the following three categories: Level 1: Quoted market prices in active markets for identical assets or liabilities Level 2: Observable market based inputs or unobservable inputs that are corroborated by market data Level 3: Unobservable inputs that are not corroborated by market data 17 (Continued)

INSIGHT PHARMACEUTICALS CORPORATION AND SUBSIDIARY Notes to Consolidated Financial Statements June 30, 2014 (Dollars in thousands) The level in the fair value within which a fair value measurement falls is based on the lowest level input that is significant to the fair value measurement in its entirety. The Company’s interest rate cap and swap are pay-fixed, receive-variable interest rate instruments based on LIBOR cap and swap rates. The LIBOR cap and swap rates are observable at commonly quoted intervals for the full term of the swaps, and, therefore, are considered Level 2 measurements. See note 2 for additional disclosure on derivatives. Certain assets and liabilities are measured at fair value on a nonrecurring basis; that is, the instruments are not measured at fair value on an ongoing basis but are subject to fair value adjustments in certain circumstances (for example, when there is evidence of impairment). The fair value of the trademarks and other intangibles and goodwill are considered Level 3 measurements. Trademarks measured at fair value on a nonrecurring basis relate to trademarks that were acquired in connection with an acquisition. The fair value of these intangible assets was calculated based upon discounted cash flow projections. These projections incorporate management’s assumptions about future cash flows based upon past experience and future expectations. The expected cash flows are then discounted using a discount rate that the Company believes is commensurate with the risks involved. Goodwill is measured at fair value on a nonrecurring basis. There were no impairment losses recorded in the six months ended June 30, 2014 and 2013. The fair value of goodwill was calculated using an income approach and a market approach. These approaches incorporated management’s assumptions about future cash flows based on past experience and future expectations and financial metrics from comparable public companies. (14) Subsequent Events On September 3, 2014, the Prestige Holdings, Inc. acquired all of the outstanding stock of the Company pursuant to the Stock Purchase Agreement, dated as of April 25, 2014 for $750,000 in cash, subject to adjustments. The Company considered all subsequent events through November 18, 2014, the date the consolidated financial statements were available to be issued, on no additional subsequent events were noted. 18