
ASX/MEDIA RELEASE | 21 February 2006 |
pSivida: Uveitis long term trial results positive for pSivida
Second set of follow-up trial results announced today
Global bio-nanotech company pSivida Limited (NASDAQ:PSDV, ASX:PSD, Xetra:PSI) today announced that preliminary three year follow-up data from Bausch and Lomb’s multi-center, randomized, dose-masked clinical trial of Retisert™ for the treatment of chronic non-infectious posterior segment uveitis has been published. Global eye health company, Bausch and Lomb (NYSE: BOL), exclusive licensee of Retisert™, conducted the study that showed the recurrence rate was significantly lower in eyes receiving Retisert™ than in non-implanted eyes. This study involved 278 patients from 27 hospitals in the United States and one in Singapore.
pSivida receives royalties from sales of Retisert™ which is presently priced at US$18,250. Covered in the United States by Medicare and Medicaid, Retisert™ is co-marketed in the United States by Bausch & Lomb and Novartis.
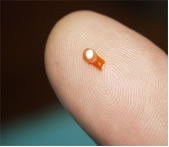 Surgically implantable RetisertTM | Posterior uveitis is the third leading cause of blindness in the United States where it afflicts an estimated 175,000 people. Approximately 800,000 people have the disease worldwide. Uveitis is a chronic auto-immune disease in which the bodies own defences attack the inner lining of the eye (the uvea). Retisert™, approved by the FDA in April 2005, is the only FDA approved drug for this disease. Retisert™ is surgically implanted into the eye and is approved to release a constant amount of the drug, fluocinolone acetonide, over a treatment period of 30 months. |
The study concluded that at three years control of uveitis in eyes implanted with Retisert™ was still significantly better than in non-implanted eyes, but was less effective than at two years and that some eyes may need to be re-implanted between 24 and 36 months.
“These results confirm the long term benefits of Retisert™ in the treatment of this devastating disease,” said Mr Gavin Rezos, CEO of pSivida Limited. “The possible need for an additional implant at between 24 and 36 months is consistent with the 30 month label from the FDA”.
In this study, patients were randomised to receive either a 0.59 mg or a 2.1 mg Retisert™ device. Data presented was the aggregate of the two doses. At three years, the recurrence rate of uveitis was 33% in the eye receiving Retisert™ compared to 57% of fellow eyes (p< 0.001). A greater number of eyes receiving Retisert™ experienced an improvement in vision of at least 15 letters (three lines on an eye chart) compared to fellow eyes (22% versus 6%, p<0.001). 45% of eyes receiving Retisert™ required an operation to relieve elevated intraocular pressure and 92% developed a cataract. Cataract surgery is a relatively uncomplicated and established procedure with a high success rate.
The abstract (#1523) detailing this data is available on the website of the Association for Research in Vision and Ophthalmology www.arvo.org. Fuller data will be presented at the ARVO conference in May 2006.
-ENDS-
pSivida Limited Brian Leedman Investor Relations pSivida Limited Tel: + 61 8 9226 5099 brianl@psivida.com | US Public Relations Beverly Jedynak President Martin E. Janis & Company, Inc Tel: +1 (312) 943 1100 ext. 12 bjedynak@janispr.com | UK & Europe Public Relations Mark Swallow / Helena Podd Citigate Dewe Rogerson Tel: +44 (0)20 7638 9571 mark.swallow@citigatedr.co.uk |
NOTES TO EDITORS:
What is Uveitis? An autoimmune condition, Uveitis manifests itself as an inflammation inside the eye, that can lead to sudden or gradual vision loss. It can be caused by diseases such as multiple sclerosis, rheumatoid arthritis pSivida is a global bio-nanotech company committed to the biomedical sector and the development of drug delivery products. Retisert™ is FDA approved for the treatment of uveitis. Vitrasert® is FDA approved for the treatment of AIDS-related CMV Retinitis. The technologies underlying both of these products are licensed to Bausch & Lomb. |  |
pSivida owns the rights to develop and commercialise a modified form of silicon (porosified or nano-structrured silicon) known as BioSilicon™, which has applications in drug delivery, wound healing, orthopaedics, and tissue engineering. pSivida’s subsidiary, AION Diagnostics Limited is developing diagnostic products and the subsidiary pSiNutria is developing food technology products both using BioSilicon™.
pSivida’s intellectual property portfolio consists of 70 patent families, 74 granted patents and over 290 patent applications.
pSivida conducts its operations from offices and facilities near Boston in Massachusetts, Malvern in the United Kingdom, Perth in Western Australia and Singapore.
pSivida is listed on NASDAQ (PSDV), the Australian Stock Exchange (PSD) and in Germany on the Frankfurt Stock Exchange on the XETRA system (German Symbol: PSI. Securities Code (WKN) 358705). pSivida is a founding member of the NASDAQ Health Care Index and the Merrill Lynch Nanotechnology Index.
The Company's largest shareholder and a strategic partner is QinetiQ, a leading international defence, security and technology company, formed in July 2001 from the UK Government's Defence Evaluation & Research Agency (DERA). QinetiQ was instrumental in discovering BioSilicon(TM) and pSivida enjoys a strong relationship with it having access to its cutting edge research and development facilities. For more information visit www.QinetiQ.com
For more information, visit www.psivida.com
This document contains forward-looking statements that involve risks and uncertainties. The statements are indicated by the use of words such as "believes", "expects", "anticipates" and similar words and phrases. Although we believe that the expectations reflected in such forward-looking statements are reasonable at this time, we can give no assurance that such expectations will prove to be correct. Given these uncertainties, readers are cautioned not to place undue reliance on such forward-looking statements. Actual results could differ materially from those anticipated in these forward-looking statements due to many important factors including: our failure to develop applications for BioSiliconTM due to regulatory, scientific or other issues, our inability to successfully integrate CDS’ operations and employees; the failure of the CDS’ products to achieve expected revenues and the combined entity’s inability to develop existing or proposed products; the failure of the Bausch & Lomb/Novartis co-promotion arrangement to provide faster royalty growth. Other reasons are contained in cautionary statements in the Registration Statement on Form 20-F filed with the U.S. Securities and Exchange Commission, including, without limitation, under Item 3.D, "Risk Factors" therein. We do not undertake to update any oral or written forward-looking statements that may be made by or on behalf of pSivida.