Searchable text section of graphics shown above
| [LOGO] |
Certain matters discussed today or answers that may be given to questions asked could constitute forward-looking statements that are subject to risks and uncertainties relating to ev3’s future financial or business performance. ev3’s actual results could differ materially from those anticipated in these forward-looking statements. The factors that might affect our results are found in the “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections of our most recent periodic report filed with the Securities and Exchange Commission. You can access this report in the SEC’s EDGAR database found at www.sec.gov. Please note that ev3 is under no obligation to update any of the forward-looking statements discussed today.
|
|
Catheter-Based Devices for Treating Circulatory and Vascular Disorders
Peripheral | Coronary | Neuro |
|
|
|
[GRAPHIC] | [GRAPHIC] | [GRAPHIC] |
|
|
|
• Radiologists | • Cardiologists | • Neuro Radiologists |
• Vascular Surgeons |
| • Neuro Surgeons |
• Cardiologists |
|
|
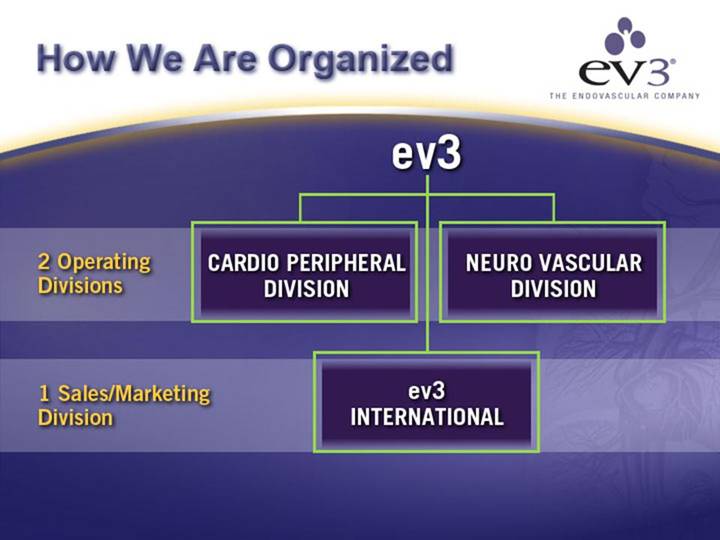
How We Are Organized
| ev3 | |
|
|
|
2 Operating Divisions | CARDIO PERIPHERAL | NEURO VASCULAR |
|
|
|
1 Sales/Marketing Division | ev3 | |
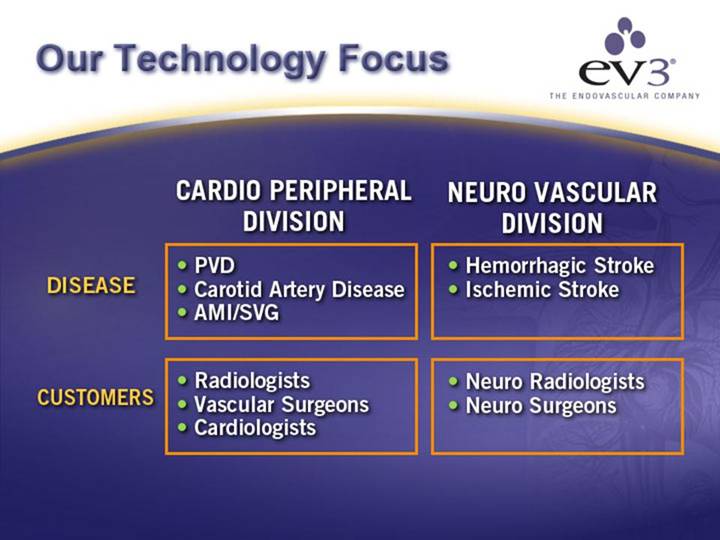
Our Technology Focus
| CARDIO PERIPHERAL | NEURO VASCULAR |
|
|
|
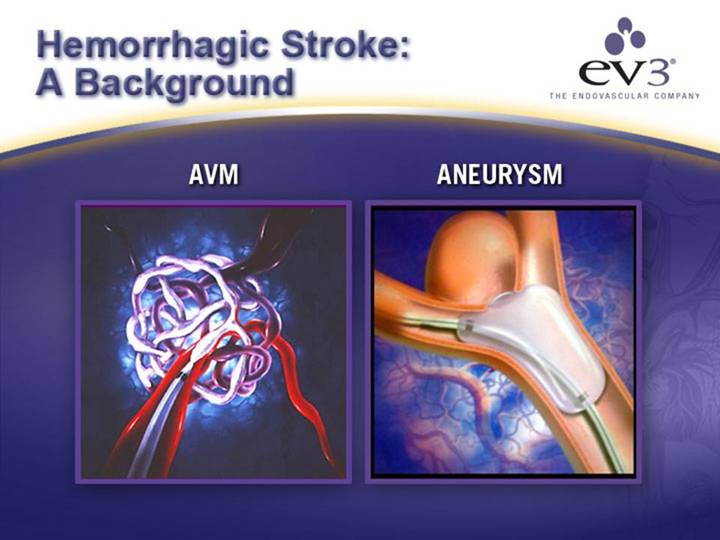
DISEASE | • PVD | • Hemorrhagic Stroke |
• Carotid Artery Disease | • Ischemic Stroke | |
• AMI/SVG |
| |
|
|
|
CUSTOMERS | • Radiologists | • Neuro Radiologists |
• Vascular Surgeons | • Neuro Surgeons | |
• Cardiologists |
|
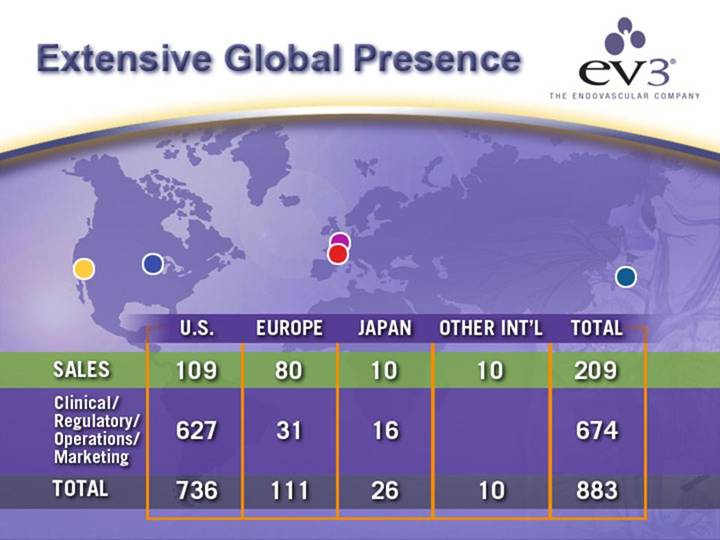
Extensive Global Presence
[GRAPHIC]
|
| U.S. |
| EUROPE |
| JAPAN |
| OTHER INT’L |
| TOTAL |
|
SALES |
| 109 |
| 80 |
| 10 |
| 10 |
| 209 |
|
Clinical/ Regulatory/ Operations/ Marketing |
| 627 |
| 31 |
| 16 |
|
|
| 674 |
|
TOTAL |
| 736 |
| 111 |
| 26 |
| 10 |
| 883 |
|
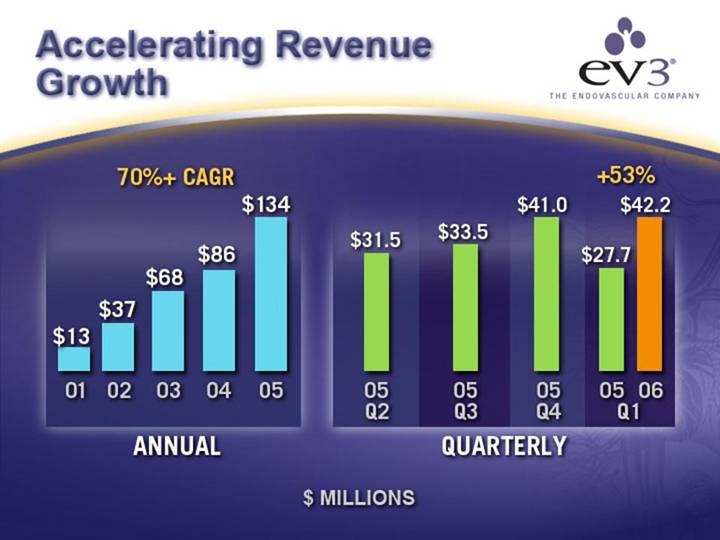
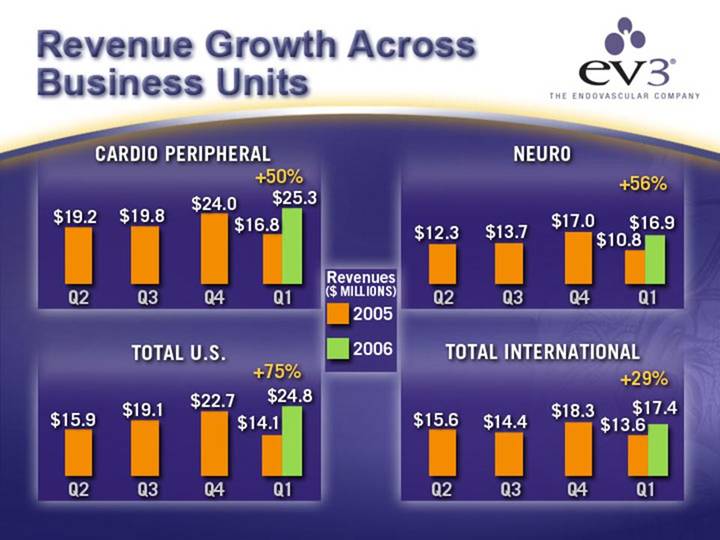
Revenue Growth Across Business Units
CARDIO PERIPHERAL | NEURO |
|
|
[CHART] | [CHART] |
|
|
TOTAL U.S. | TOTAL INTERNATIONAL |
|
|
[CHART] | [CHART] |
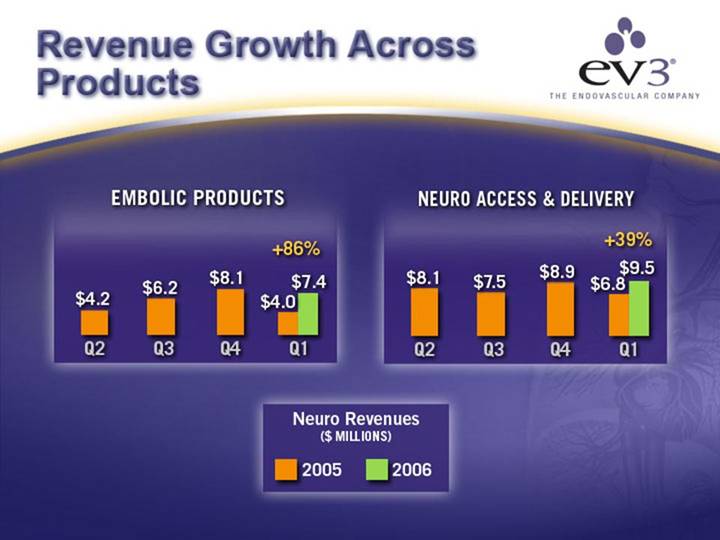
Revenue Growth Across Products
STENTS | |
| |
[CHART] | |
|
|
THROMBECTOMY & EMBOLIC PROTECTION | PROCEDURAL SUPPORT & OTHER CP |
|
|
[CHART] | [CHART] |
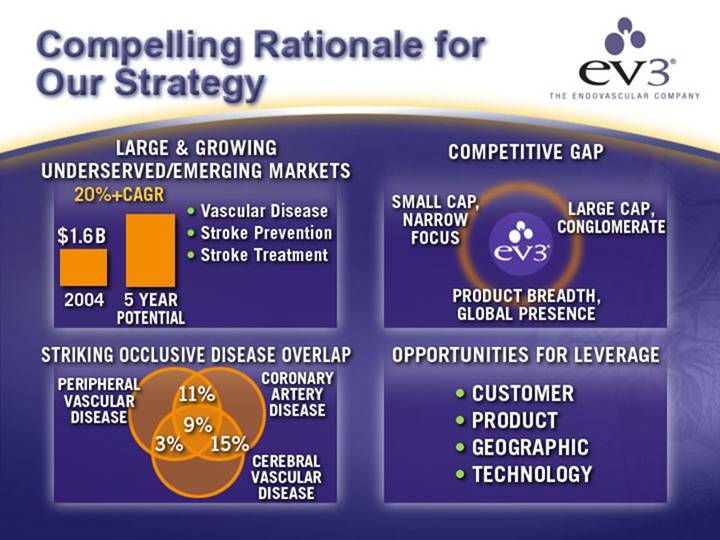
Compelling Rationale for Our Strategy
LARGE & GROWING | COMPETITIVE GAP | |||
|
| |||
[CHART] | • Vascular Disease | SMALL CAP, | [GRAPHIC] | LARGE CAP, |
• Stroke Prevention | ||||
• Stroke Treatment | ||||
| PRODUCT BREADTH, | |||
|
| |||
STRIKING OCCLUSIVE DISEASE OVERLAP | OPPORTUNITIES FOR LEVERAGE | |||
|
| |||
[CHART] | • CUSTOMER | |||
| • PRODUCT | |||
| • GEOGRAPHIC | |||
| • TECHNOLOGY | |||
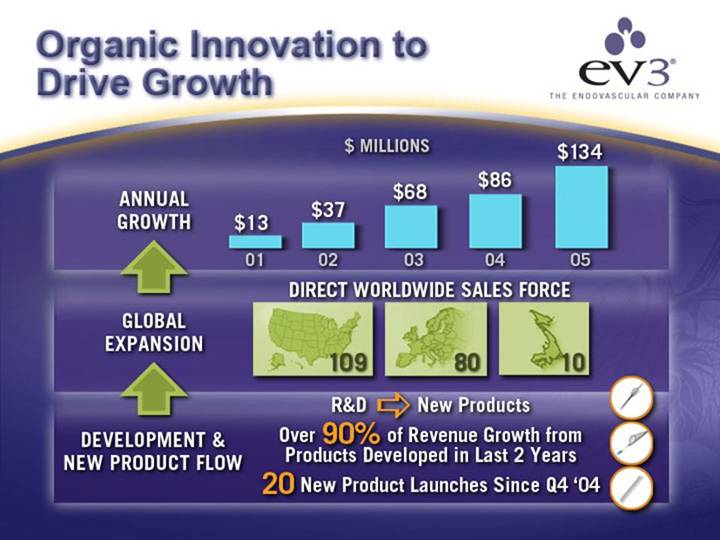
Organic Innovation to Drive Growth
ANNUAL GROWTH | [CHART] |
|
|
|
|
GLOBAL EXPANSION | DIRECT WORLDWIDE SALES FORCE |
|
|
| |
[GRAPHIC] |
| |
|
|
|
DEVELOPMENT & | R&D | [GRAPHIC] |
Over 90% of Revenue Growth from Products Developed in Last 2 Years | [GRAPHIC] | |
20 New Product Launches Since Q4 ‘04 | [GRAPHIC] |
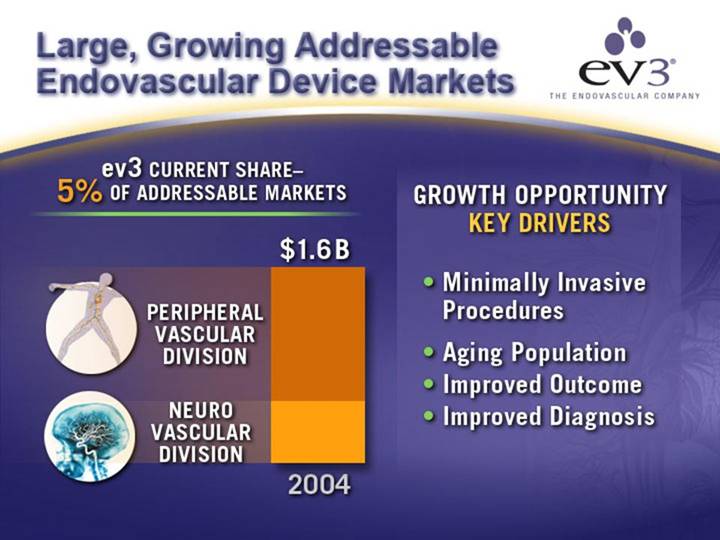
Large, Growing Addressable Endovascular Device Markets
ev3 CURRENT SHARE– 5% OF ADDRESSABLE MARKETS
[CHART]
GROWTH OPPORTUNITY
KEY DRIVERS
• Minimally Invasive Procedures
• Aging Population
• Improved Outcome
• Improved Diagnosis
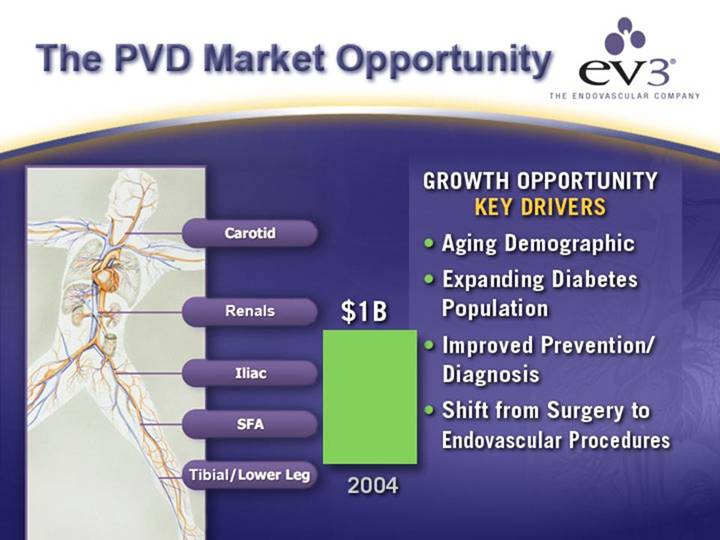
The PVD Market Opportunity
[GRAPHIC]
Carotid
Renals
Iliac
SFA
Tibial/Lower Leg
[CHART]
GROWTH OPPORTUNITY
KEY DRIVERS
• Aging Demographic
• Expanding Diabetes Population
• Improved Prevention/Diagnosis
• Shift from Surgery to Endovascular Procedures
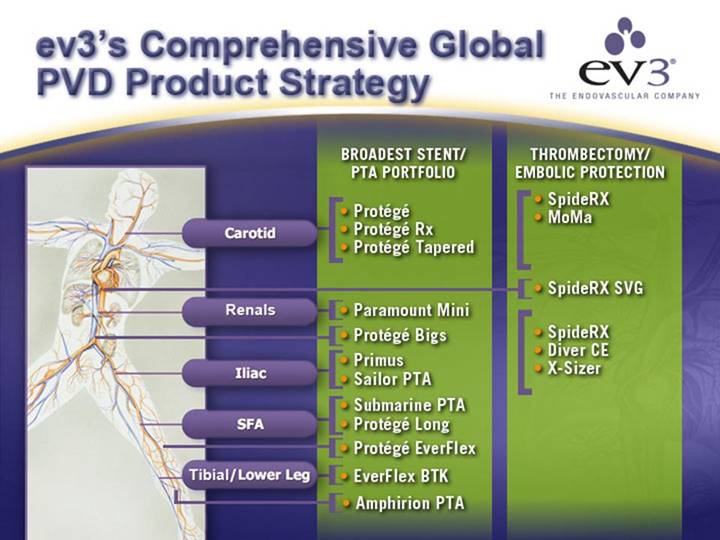
ev3’s Comprehensive Global PVD Product Strategy
[GRAPHIC]
|
| BROADEST STENT/ |
| THROMBECTOMY/ |
|
|
|
|
|
Carotid |
| • Protégé |
| • SpideRX |
| • Protégé Rx |
| • MoMa | |
| • Protégé Tapered |
|
| |
|
|
|
| • SpideRX SVG |
Renals |
| • Paramount Mini |
|
|
| • Protégé Bigs |
| • SpideRX | |
Iliac |
| • Primus |
| • Diver CE |
|
| • Sailor PTA |
| • X-Sizer |
SFA |
| • Submarine PTA |
|
|
| • Protégé Long |
|
| |
|
| • Protégé EverFlex |
|
|
Tibial/Lower Leg |
| • EverFlex BTK |
|
|
|
| • Amphirion PTA |
|
|
PROTÉGÉ EverFlex Durability
Up to 5-10 Times More Durable Than Competitive Stents
[CHART]
* Bench testing conducted at ev3. Data on file.
** Tests stopped with no stent fractures.
Test was not intended to be indicative of clinical performance.
• Spiral cell interconnections are constructed to withstand extreme movement
• New wave peak design produces expansion force that resists compression while providing excellent wall apposition
• Multi-step finishing process reduces surface imperfections
• Offers the longest, bare metal stents in the marketplace (up to 150 mm)
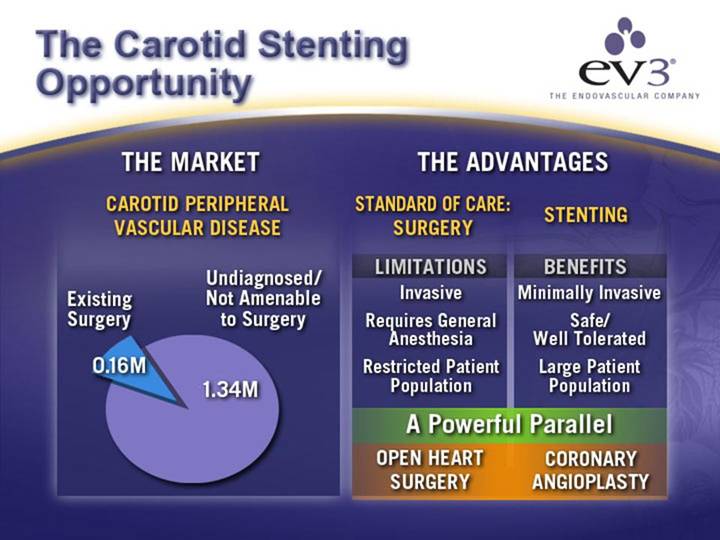
The Carotid Stenting Opportunity
THE MARKET
CAROTID PERIPHERAL VASCULAR DISEASE
[CHART]
THE ADVANTAGES
STANDARD OF CARE: SURGERY |
| STENTING |
|
|
|
LIMITATIONS |
| BENEFITS |
|
|
|
Invasive |
| Minimally Invasive |
|
|
|
Requires General Anesthesia |
| Safe/Well Tolerated |
|
|
|
Restricted Patient Population |
| Large Patient Population |
|
|
|
A Powerful Parallel | ||
|
|
|
OPEN HEART SURGERY |
| CORONARY ANGIOPLASTY |
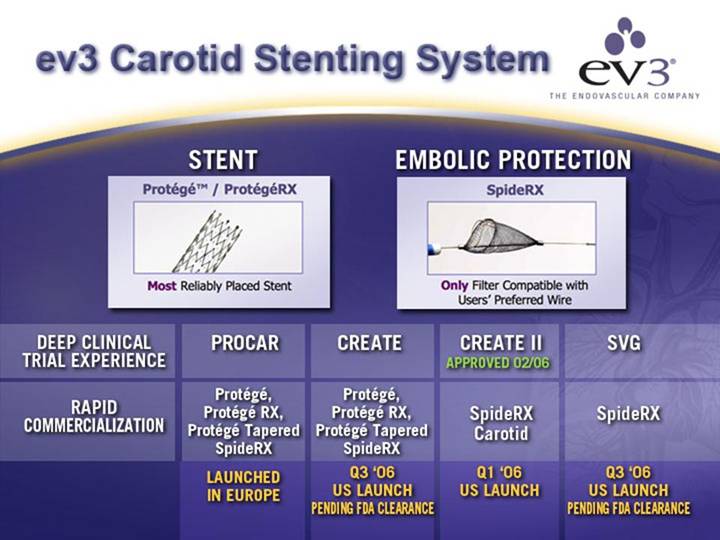
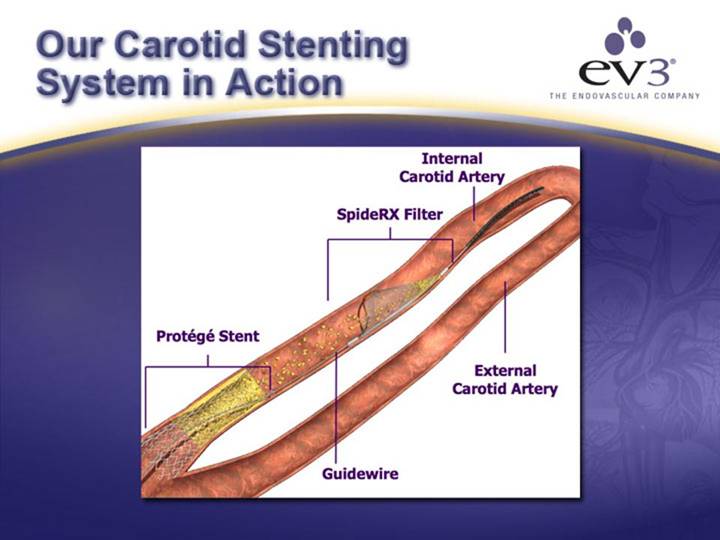
ev3 Carotid Stenting System
STENT
[GRAPHIC]
EMBOLIC PROTECTION
[GRAPHIC]
DEEP CLINICAL |
|
|
|
|
| CREATE II |
|
|
|
TRIAL EXPERIENCE |
| PROCAR |
| CREATE |
| APPROVED 02/06 |
| SVG |
|
|
|
|
|
|
|
|
|
|
|
RAPID COMMERCIALIZATION |
| Protégé, |
| Protégé, |
| SpideRX |
| SpideRX |
|
|
|
|
|
|
|
|
|
|
|
|
| LAUNCHED |
| Q3 ‘06 |
| Q1 ‘06 |
| Q3 ‘06 |
|
Clinical Trial Momentum
* Pending FDA Clearance
|
| CREATE PIVOTAL * |
| CREATE RX |
| SPIDER SVG* |
|
PRINCIPAL |
| Dr. Rob Safian |
| Dr. Rob Safian |
| Dr. William O’Neil |
|
|
|
|
|
|
|
|
|
TOTAL PATIENTS |
|
|
|
|
| 747 |
|
|
|
|
|
|
|
|
|
DEVICE EVALUATED |
| Spider RX + ProtégéRX |
| SpiderRX + ProtégéRX |
| Non Inferiority Compared to Guardwire and Filterwire |
|
|
|
|
|
|
|
|
|
30 DAY ENDPOINT |
| 6.2% |
| 5.6% |
| 9.1% |
|
|
|
|
|
|
|
|
|
COMPETITIVE TRIAL ENDPOINTS |
| AT 1 Year • SAPPHIRE (Cordis) 11.9% |
|
|
| AT 30 Days • SAFER (Medtronic Guardwire) 9.6% |
|
|
|
|
|
|
|
|
|
TIMING |
| Submitted January 2006 |
| • Submitted 510K |
|
|
|
Critical Limb Ischemia – Global Product Strategy
[GRAPHIC]
|
| BROADEST STENT/ |
| THROMBECTOMY/ |
|
|
|
|
|
Iliac |
| • Sailor PTA |
|
|
SFA |
| • Protégé EverFlex |
|
|
| • Submarine PTA |
|
| |
|
|
|
| • SpideRX |
|
|
|
| • Diver CE |
|
|
|
| • X-Sizer |
Tibial/Lower Leg |
| • EverFlex BTK |
|
|
|
|
|
|
|
|
| • Amphirion PTA |
|
|
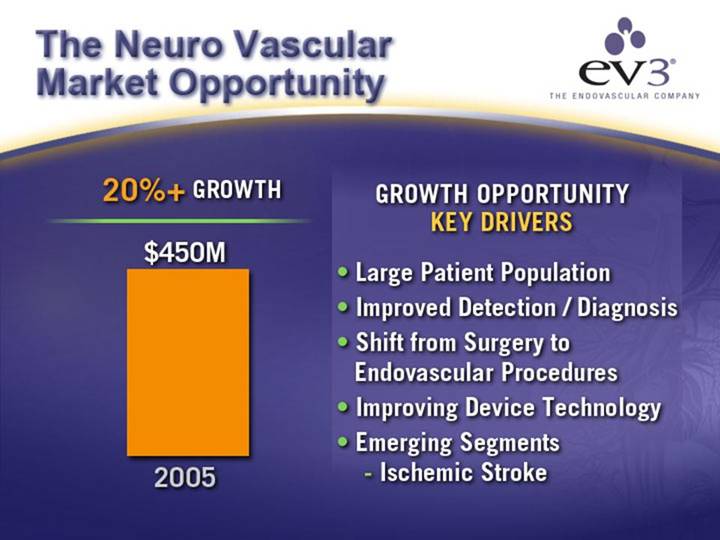
The Neuro Vascular Market Opportunity
20%+ GROWTH
[CHART]
GROWTH OPPORTUNITY
KEY DRIVERS
• Large Patient Population
• Improved Detection / Diagnosis
• Shift from Surgery to Endovascular Procedures
• Improving Device Technology
• Emerging Segments
• Ischemic Stroke
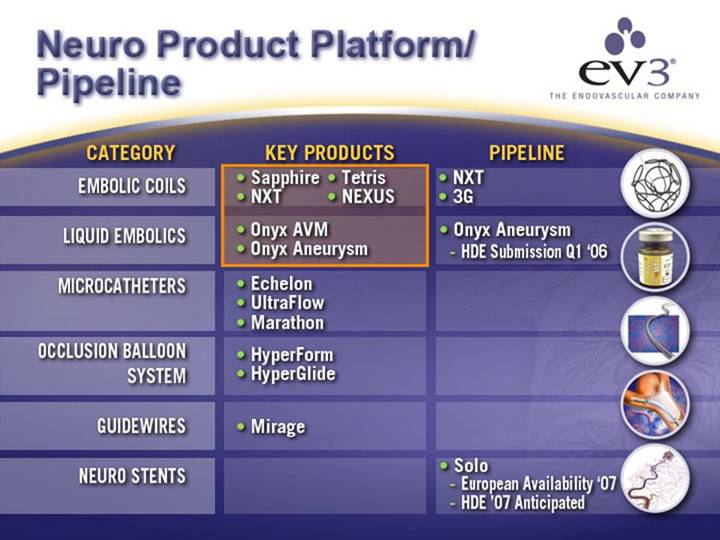
Neuro Product Platform/Pipeline
CATEGORY |
| KEY PRODUCTS |
| PIPELINE |
|
|
|
|
|
|
|
|
|
EMBOLIC COILS |
| • Sapphire • NXT • Tetris • NEXUS |
| • NXT • 3G |
| [GRAPHIC] |
|
|
|
|
|
|
|
LIQUID EMBOLICS |
| • Onyx AVM • Onyx Aneurysm |
| • Onyx Aneurysm • HDE Submission Q1 ‘06 |
| [GRAPHIC] |
|
|
|
|
|
|
|
MICROCATHETERS |
| • Echelon • UltraFlow • Marathon |
|
|
| [GRAPHIC] |
|
|
|
|
|
|
|
OCCLUSION BALLOON SYSTEM |
| • HyperForm • HyperGlide |
|
|
| [GRAPHIC] |
|
|
|
|
|
|
|
GUIDEWIRES |
| • Mirage |
|
|
| [GRAPHIC] |
|
|
|
|
|
|
|
NEURO STENTS |
|
|
| • Solo • European Availability ‘07 • HDE ‘07 Anticipated |
| [GRAPHIC] |
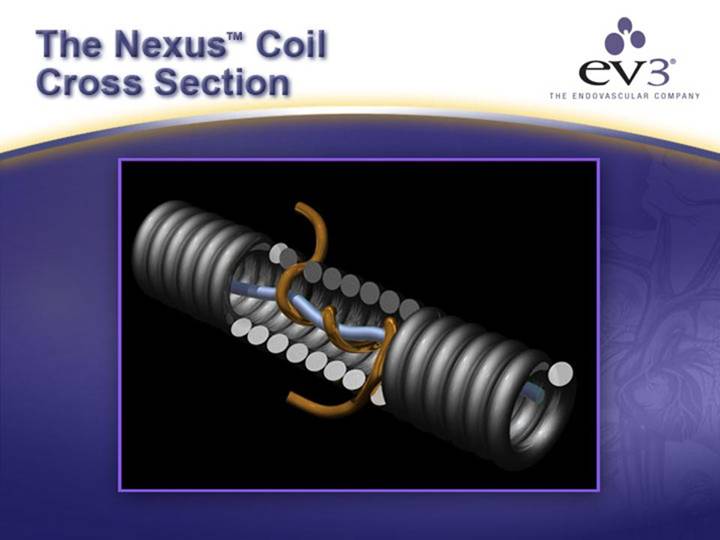
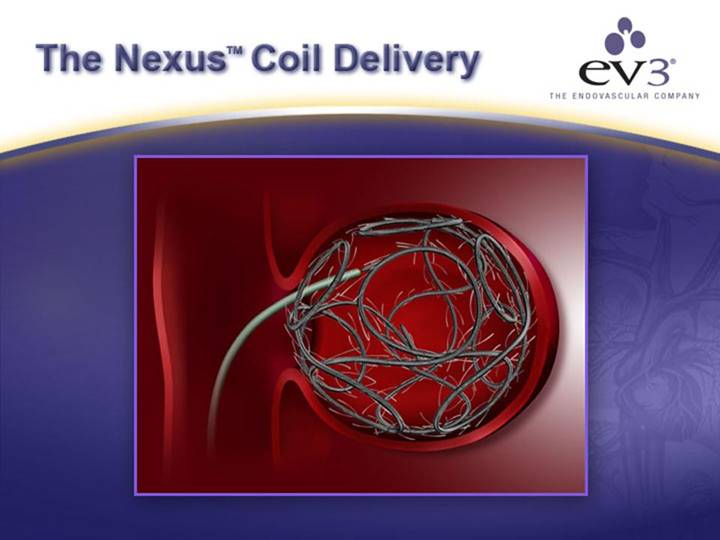
ev3’s NexusTM Coil
THE PROBLEM WITH TRADITIONAL COILS
Aneurysm Recanalization
• INCOMPLETE FILL | • COIL COMPACTION | • RE-GROWTH OF ANEURYSM |
OUR NEXUSTM SOLUTION
[GRAPHIC] |
| SUPERIOR DESIGN • Easier Deployment • Increased Mass (99%+) • Better Wall Contact |
THE RESULT
SUCCESSFUL, LONG LASTING SOLUTION
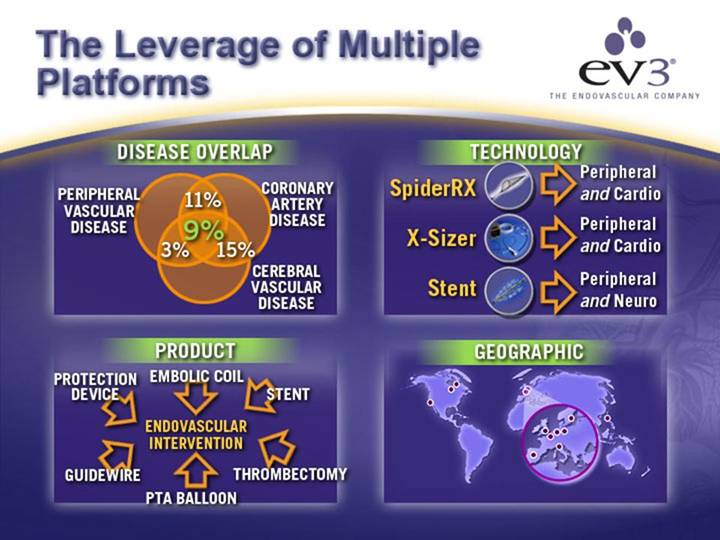
The Leverage of Multiple Platforms
DISEASE OVERLAP |
| TECHNOLOGY |
|
|
|
[GRAPHIC] |
| [GRAPHIC] |
|
|
|
PRODUCT |
| GEOGRAPHIC |
|
|
|
[GRAPHIC] |
| [GRAPHIC] |
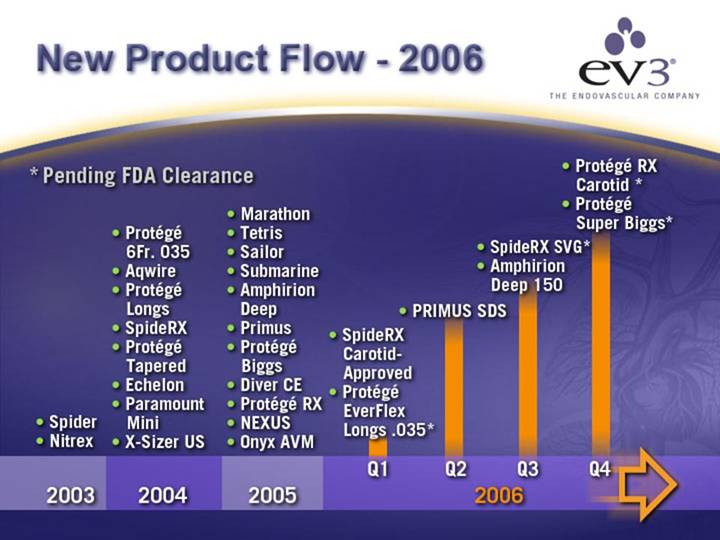
Project Launch Momentum
• Accelerating New Product Launch Rate
• 90%+ of Today’s Revenues from Products Developed in Last 2 Years
* Pending FDA Clearance
[CHART]
Clinical Trial and Submission Progress
Driving Clinical Indications for Commercial Approval and Clinical Data
[CHART]
Senior Management Team
NAME |
| ev3 POSITION |
| PAST EXPERIENCE |
|
|
|
|
|
JIM CORBETT |
| President & CEO |
| Boston Scientific, SciMED, Baxter |
|
|
|
|
|
PAT SPANGLER |
| CFO |
| Empi, Medtronic |
|
|
|
|
|
MATTHEW JENUSAITIS |
| President, Neuro Division |
| Boston Scientific, SciMED, Baxter |
|
|
|
|
|
STACY ENXING SENG |
| President, Cardio Peripheral Division |
| Boston Scientific, SciMED, Baxter |
|
|
|
|
|
PASCAL GIRIN |
| President, International |
| Baxter |
|
|
|
|
|
JEFF PETERS |
| VP, R&D Cardio Peripheral Division & CTO |
| SciMED |
|
|
|
|
|
CECILY HINES |
| VP, Secretary & Chief Legal Officer |
| Boston Scientific, SciMED |
|
|
|
|
|
GREG MORRISON |
| VP, Human Resources |
| West Publishing, Pfizer |
Target Operating Model
|
| 2004 |
| TARGET |
|
|
|
|
|
|
|
REVENUES |
| 100 | % | 100 | % |
|
|
|
|
|
|
GROSS MARGIN |
| 54 | % | 65-70 | % |
|
|
|
|
|
|
OPERATING EXPENSES |
|
|
|
|
|
SG&A |
| 119 | % | 30 | % |
R&D |
| 45 | % | 15 | % |
AMORTIZATION |
| 11 | % | 5 | % |
GAIN ON SALES OF ASSETS |
| (17 | )% |
| % |
|
|
|
|
|
|
OPERATING MARGIN |
| (105 | )% | 15-20 | % |
Strategies for Sustainable Growth
|
| BROADEN |
|
|
|
| GLOBAL EXPANSION |
|
|
|
|
CONTINUED PRODUCT |
|
|