Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 20-F
(Mark One)
| ¨ | REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 |
OR
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended March 31, 2006
OR
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
| ¨ | SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Date of event requiring this shell company report
Commission file number: 000-51440
CHINA MEDICAL TECHNOLOGIES, INC.
(Exact name of Registrant as specified in its charter)
N/A
(Translation of Registrant’s name into English)
Cayman Islands
(Jurisdiction of incorporation or organization)
No. 24 Yong Chang North Road
Beijing Economic-Technological Development Area
Beijing 100176
People’s Republic of China
(Address of principal executive offices)
Securities registered or to be registered pursuant to Section 12(b) of the Act
American Depositary Shares, each representing ten ordinary shares, par value $0.10 per share,
Nasdaq National Market
Securities registered or to be registered pursuant to Section 12(g) of the Act
Name of each exchange and Title of each class on which registered:
None
(Title of Class)
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act.
None
(Title of Class)
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report. 273,600,001
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. ¨ Yes x No
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. ¨ Yes x No
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. x Yes ¨ No
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer and large accelerated filer” in Rule 12b-2 of the Exchange Act. (Check one):
¨ Large accelerated filer ¨ Accelerated filer x Non-accelerated filer
Indicate by check mark which financial statement item the registrant has elected to follow: ¨ Item 17 x Item 18
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act) ¨ Yes x No
Table of Contents
| INTRODUCTION | ||||
Item 1. | 3 | |||
Item 2. | 3 | |||
Item 3. | 3 | |||
Item 4. | 26 | |||
Item 5. | 45 | |||
Item 6. | 62 | |||
Item 7. | 69 | |||
Item 8. | 71 | |||
Item 9. | 72 | |||
Item 10. | 73 | |||
Item 11. | 80 | |||
Item 12. | 80 | |||
Item 13. | 81 | |||
Item 14. | Material Modifications to the Rights of Security Holders and Use of Proceeds | 81 | ||
Item 15. | 81 | |||
Item 16A. | 81 | |||
Item 16B. | 82 | |||
Item 16C. | 82 | |||
Item 16D. | 82 | |||
Item 16E. | Purchases of Equity Securities by the Issuer and Affiliated Purchasers | 82 | ||
Item 17. | 83 | |||
Item 18. | 83 | |||
Item 19. | 83 | |||
Table of Contents
Unless otherwise indicated, references in this annual report on Form 20-F to:
| • | “ADRs” are to the American depositary receipts, which, if issued, evidence our ADSs; |
| • | “ADSs” are to our American depositary shares, each of which represents ten ordinary shares; |
| • | “Articles of association” are to our memorandum and articles of association, as amended and restated from time to time; |
| • | “China” and the “PRC” are to the People’s Republic of China, excluding, for the purposes of this annual report on Form 20-F only, Taiwan and the special administrative regions of Hong Kong and Macau; |
| • | “RMB” and “Renminbi” are to the legal currency of China; |
| • | “shares” and “ordinary shares” are to our ordinary shares, par value US$0.10 per share; and |
| • | “US$” and “U.S. dollars” are to the legal currency of the United States. |
Unless the context indicates otherwise, “we,” “us,” “our company,” “our,” “China Medical” and “CMED” refer to China Medical Technologies, Inc. and its predecessor and wholly owned subsidiary, YDME Beijing.
In addition, unless otherwise indicated, references in this annual report on Form 20-F to:
| • | “Asset Managers” are to Asset Managers (China) Fund Co., Ltd.; |
| • | “Beijing Chengxuan” are to Beijing Chengxuan Economic and Trade Co., Ltd.; |
| • | “Beijing Weixiao” are to Beijing Weixiao Biological Technology Development Co., Ltd.; |
| • | “Chengxuan” are to Chengxuan International Ltd.; |
| • | “GE” are to General Electric Company or to one or more of its wholly owned subsidiaries, as the case may be and where applicable, to its relevant subsidiaries and its related companies; |
| • | “Golden Meditech” are to Golden Meditech (BVI) Company Ltd.; |
| • | “Green Wall” are to Green Wall Development Ltd.; |
| • | “Neon Liberty” are to certain investment funds managed by Neon Liberty Capital Management LLC; |
| • | “PUPH” are to Peking University People’s Hospital; and |
| • | “YDME Beijing” are to Beijing Yuande Bio-Medical Engineering Co., Ltd., our predecessor and wholly owned subsidiary in China. |
The following technical and industry specific terms used in this annual report on Form 20-F have the meanings set forth below:
| • | “Ablation” means elimination or removal; |
| • | “ECLIA analyzer” means an instrument used in in-vitro diagnostics; |
| • | “ECLIA technology” means a technology that improves chemical reactions and enables reagents to be more sensitive, accurate and stable; |
| • | “In-vitro diagnostics,” or “IVD,” means the detection and monitoring of diseases through laboratory evaluation and the analysis of bodily fluids and tissues from patients; |
| • | “IVD system” consists of the instrument (which we referred to as the “ECLIA analyzer”) and the reagent kits used in in-vitro diagnostics; |
1
Table of Contents
| • | “Luminescence immunoassay IVD system” means an advanced, light-based IVD system commonly used in the United States and Western Europe; |
| • | “Reagent” means a substance used in a chemical reaction to detect, measure and produce other substances; and |
| • | “Reagent kits” means commercially prepared reagent sets, with accessory devices, containing all major components necessary to perform IVD tests. |
This annual report on Form 20-F includes our audited consolidated financial statements for the years ended March 31, 2004, 2005 and 2006 and as of March 31, 2005 and 2006.
We and certain selling shareholders of our company completed the initial public offering of 7,360,000 ADSs, each representing ten ordinary shares in August 2005. On August 10, 2005, we listed our ADSs on the Nasdaq National Market under the symbol “CMED.” On March 27, 2006, we completed a follow-on offering of 5,750,000 ADSs, which were sold by certain of our existing shareholders.
2
Table of Contents
ITEM 1. IDENTITYOF DIRECTORS, SENIOR MANAGEMENTAND ADVISERS
Not Applicable.
ITEM 2. OFFER STATISTICSAND EXPECTED TIMETABLE
Not Applicable.
A. Selected Financial Data
The following selected consolidated statement of operations data for the years ended March 31, 2002, 2003, 2004, 2005 and 2006 and the consolidated balance sheet data as of March 31, 2002, 2003, 2004, 2005 and 2006 have been derived from our audited consolidated financial statements, which have been audited by KPMG, an independent registered public accounting firm. The report of KPMG on our consolidated financial statements as of March 31, 2005 and 2006 and for each of the three fiscal years ended March 31, 2004, 2005 and 2006 is included elsewhere in this annual report on Form 20-F. Our selected consolidated statement of operations data for the fiscal year ended March 31, 2002 and 2003 and our consolidated balance sheets as of March 31, 2002, 2003 and 2004 have been derived from our audited consolidated financial statements, which are not included in this annual report on Form 20-F. You should read the selected consolidated financial data in conjunction with those financial statements and the related notes and “Item 5. Operating and Financial Review and Prospects” included elsewhere in this annual report on Form 20-F. Our consolidated financial statements are prepared and presented in accordance with U.S. GAAP. Our historical results do not necessarily indicate our results expected for any future periods.
| Year ended March 31, | ||||||||||||||||||
| 2002 | 2003 | 2004 | 2005 | 2006 | 2006 | |||||||||||||
| RMB | RMB | RMB | RMB | RMB | US$ | |||||||||||||
| (in thousands, except for number of shares, per share and per ADS data) | ||||||||||||||||||
| Statement of operations data: | ||||||||||||||||||
Revenues, net(1) | 44,849 | 63,716 | 110,750 | 217,547 | 371,767 | 46,374 | ||||||||||||
Cost of revenues | (11,504 | ) | (20,627 | ) | (33,983 | ) | (64,579 | ) | (110,494 | ) | (13,783 | ) | ||||||
Gross profit | 33,345 | 43,089 | 76,767 | 152,968 | 261,273 | 32,591 | ||||||||||||
Operating expenses: | ||||||||||||||||||
Research and development | (1,192 | ) | (1,246 | ) | (2,036 | ) | (2,815 | ) | (14,374 | ) | (1,793 | ) | ||||||
Sales and marketing | (2,059 | ) | (1,963 | ) | (2,632 | ) | (4,960 | ) | (15,327 | ) | (1,912 | ) | ||||||
General and administrative | (8,805 | ) | (7,235 | ) | (5,619 | ) | (16,644 | ) | (38,309 | ) | (4,779 | ) | ||||||
Total operating expenses | (12,056 | ) | (10,444 | ) | (10,287 | ) | (24,419 | ) | (68,010 | ) | (8,484 | ) | ||||||
Operating income | 21,289 | 32,645 | 66,480 | 128,549 | 193,263 | 24,107 | ||||||||||||
Net interest (expense)/income and other income(2) | (1,884 | ) | (1,803 | ) | 14 | 1,748 | 23,838 | 2,973 | ||||||||||
Income before tax | 19,405 | 30,842 | 66,494 | 130,297 | 217,101 | 27,080 | ||||||||||||
Income tax benefit/(expense) | 433 | (3,286 | ) | (4,933 | ) | (11,854 | ) | (18,088 | ) | (2,256 | ) | |||||||
Net income(3) | 19,838 | 27,556 | 61,561 | 118,443 | 199,013 | 24,824 | ||||||||||||
Earnings per share (basic and diluted)(3) | 0.17 | 0.23 | 0.31 | 0.59 | 0.81 | 0.10 | ||||||||||||
Earnings per ADS (basic and diluted) (unaudited)(4) | 1.70 | 2.30 | 3.10 | 5.90 | 8.10 | 1.00 | ||||||||||||
Weighted average number of shares outstanding | ||||||||||||||||||
Basic | 120,000,000 | 122,411,000 | 200,000,001 | 200,000,001 | 246,878,001 | 246,878,001 | ||||||||||||
Diluted | 120,000,000 | 122,411,000 | 200,018,001 | 200,018,001 | 247,163,001 | 247,163,001 | ||||||||||||
3
Table of Contents
| (1) | Revenues, net, include revenue from sales of our products, net of value-added tax, or VAT, sales returns, trade discounts and allowances, but include VAT refunds on the sales of self-developed software embedded in our products. VAT refunds for the five fiscal years ended March 31, 2002, 2003, 2004, 2005, and 2006 were Nil, RMB3.6 million, RMB7.1 million, RMB14.7 million and RMB23.0 million (US $2.9 million), respectively. |
| (2) | Net interest (expense)/income and other income include “interest expense,” “interest income” and “other income” from our consolidated financial statements included elsewhere in this annual report on Form 20-F. |
| (3) | If YDME Beijing had not been exempted from paying the EIT for the calendar year ended December 31, 2001 and had not had a reduced EIT rate of 7.5% for the three calendar years ended December 31, 2002, 2003 and 2004 and 10% for the calendar year ended December 31, 2005, YDME Beijing would have had an EIT rate of 15%. With an EIT rate of 15%, our net income and basic and diluted earnings per share for the following periods would have been as follows: |
| Year ended March 31, | ||||||||||||
| 2002 | 2003 | 2004 | 2005 | 2006 | 2006 | |||||||
| RMB | RMB | RMB | RMB | RMB | US$ | |||||||
| (in thousands, except for per share and per ADS data) | ||||||||||||
| Statement of operations data: | ||||||||||||
Income tax expense | 3,455 | 6,166 | 9,471 | 17,603 | 31,088 | 3,878 | ||||||
Net income | 15,950 | 24,676 | 57,023 | 112,694 | 186,013 | 23,202 | ||||||
Earnings per share: | ||||||||||||
Basic | 0.13 | 0.20 | 0.29 | 0.56 | 0.75 | 0.09 | ||||||
Diluted | 0.13 | 0.20 | 0.29 | 0.56 | 0.75 | 0.09 | ||||||
| (4) | Each ADS represents 10 ordinary shares. |
| As of March 31, | |||||||||||||
| 2002 | 2003 | 2004 | 2005 | 2006 | 2006 | ||||||||
| RMB | RMB | RMB | RMB | RMB | US$ | ||||||||
| (in thousands) | |||||||||||||
| Balance sheet data: | |||||||||||||
Cash and cash equivalents | 1,268 | 27,371 | 24,114 | 14,646 | 843,791 | 105,254 | |||||||
Working capital(1) | 25,334 | 98,442 | 52,238 | (66,707 | ) | 906,875 | 113,124 | ||||||
Intangible assets | — | — | — | 239,662 | 224,744 | 28,034 | |||||||
Total assets | 86,544 | 179,810 | 192,161 | 457,768 | 1,375,841 | 171,621 | |||||||
Amounts due to related parties (current and non-current) | — | 520 | 4,807 | 102,714 | — | — | |||||||
Total shareholders’ equity | 49,724 | 141,312 | 135,510 | 253,953 | 1,246,534 | 155,492 | |||||||
| (1) | Working capital is current assets less current liabilities. |
Exchange Rate Information
This annual report on Form 20-F contains translations of certain RMB amounts into U.S. dollar amounts at specified rates. All translations from RMB to U.S. dollars were made at the noon buying rate in The City of New York for cable transfers of RMB as certified for customs purposes by the Federal Reserve Bank of New York, or the noon buying rate. Unless otherwise stated, the translations of RMB into U.S. dollars have been made at the noon buying rate in effect on March 31, 2006, which was RMB8.0167 to $1.00. We make no representation that the RMB or U.S. dollar amounts referred to in this annual report on Form 20-F could have been or could be converted into U.S. dollars or RMB, as the case may be, at any particular rate or at all. See “Item 3. Key Information—D. Risk Factors—Risks Related to Doing Business in China—Fluctuations in exchange rates may have a material adverse effect on your investment” and “—Restrictions on currency exchange may limit our ability to receive and use our revenues effectively” for discussions of the effects of fluctuating exchange rates and currency control on the value of our ADSs. On September 27, 2006, the noon buying rate was RMB7.9019 to US$1.00.
4
Table of Contents
The following table sets forth information concerning exchange rates between the RMB and the U.S. dollar for the periods indicated. These rates are provided solely for your convenience and are not necessarily the exchange rates that we used in this annual report or will use in the preparation of our periodic reports or any other information to be provided to you. The source of these rates is the Federal Reserve Bank of New York.
| Noon Buying Rate | ||||||||
Period | Period End | Average(1) | Low | High | ||||
| (RMB per $1.00) | ||||||||
2001 | 8.2766 | 8.2770 | 8.2786 | 8.2676 | ||||
2002 | 8.2800 | 8.2770 | 8.2800 | 8.2669 | ||||
2003 | 8.2767 | 8.2772 | 8.2800 | 8.2765 | ||||
2004 | 8.2765 | 8.2768 | 8.2771 | 8.2765 | ||||
2005 | 8.0702 | 8.1826 | 8.2765 | 8.0702 | ||||
2006 | ||||||||
March | 8.0167 | 8.0350 | 8.0505 | 8.0167 | ||||
April | 8.0165 | 8.0143 | 8.0248 | 8.0040 | ||||
May | 8.0215 | 8.0131 | 8.0300 | 8.0005 | ||||
June | 7.9943 | 8.0042 | 8.0225 | 7.9943 | ||||
July | 7.9690 | 7.9897 | 8.0018 | 7.9690 | ||||
August | 7.9538 | 7.9722 | 8.0000 | 7.9538 | ||||
September (through September 27) | 7.9019 | 7.9371 | 7.9019 | 7.9545 | ||||
| (1) | Annual averages are calculated from month-end rates. Monthly averages are calculated using the average of the daily rates during the relevant period. |
B. Capitalization and Indebtedness
Not Applicable.
C. Reasons for the Offer and Use of Proceeds
Not Applicable.
D. Risk Factors
Risks Related to Our Company
We are dependent on our core product, the HIFU therapy system. A reduction in revenues of our HIFU therapy system would cause our revenues to decline and could materially harm our business.
Prior to September 2004, we generated all of our revenues from sales of our HIFU therapy system. Although we began marketing and selling our ECLIA system in September 2004, we expect that sales of our HIFU therapy system will continue to comprise a substantial portion of our revenues in the future. Any reduction in revenues from sales of our HIFU therapy system will have a direct negative impact on our business, financial condition and results of operations. Due to this lack of product diversification, an investment in our company may entail more risk than investments in companies that offer a wider variety of products or services. In addition, sales of our ECLIA system may not increase to a level that would enable us to reduce our dependence on our HIFU therapy system, and we may be unable to develop or acquire new products or technologies that would enable us to diversify our business and reduce our dependence on these two products.
If we fail to increase awareness and acceptance of our HIFU therapy system in the medical community and among patients, we will not be able to grow or even sustain our business.
Our HIFU therapy system uses a relatively new, high intensity focused ultrasound technology that is very different from conventional treatments for tumors, such as surgery, radiotherapy and chemotherapy. As of
5
Table of Contents
March 31, 2006, we have sold over 200 units of our HIFU therapy system. To achieve greater penetration of the potential market in China, we must increase market awareness and use of our HIFU therapy system, which depends on, among other things, the following:
| • | the general levels of awareness and acceptance in the medical community and among patients of HIFU technology and our HIFU therapy system; |
| • | the effectiveness of our HIFU therapy system, which we intend to further investigate through long-term studies; |
| • | the relative costs and benefits of treatment using our HIFU therapy system as compared to other treatments; |
| • | the financial or other benefits gained by hospitals that use our HIFU therapy system; |
| • | the amount of resources we have available to increase product awareness and to educate potential purchasers and users of our HIFU therapy system; |
| • | our ability to continue to develop and enhance our HIFU therapy system; |
| • | our ability to provide good technical support and customer service; and |
| • | our ability to keep up with technological changes and remain competitive. |
We may not have the financial and operational resources required to promote awareness and acceptance of our HIFU therapy system as widely or rapidly as is necessary to grow or sustain our business. Even if we were to devote a substantial portion of our resources to promoting our product, we may not succeed in raising the levels of awareness and acceptance of our HIFU therapy system as quickly as is necessary to grow or sustain our business, if at all. If we fail to increase awareness and acceptance of our HIFU therapy system in the medical community and among patients, we will not be able to grow, or even sustain, our business as planned and our financial condition and results of operations will be harmed.
We may not succeed in developing a market for our ECLIA system.
We only began marketing and selling our ECLIA system in September 2004. The success of our ECLIA system depends on several factors, including our ability to:
| • | successfully promote product awareness of our ECLIA system in China; |
| • | competitively price our ECLIA system, including the ECLIA analyzer and reagents and accessory devices, or reagent kits; |
| • | develop new reagent kits to expand the applications of our ECLIA system and to respond to new forms of diseases and disorders; |
| • | select effective distributors; and |
| • | comply with new or changing regulatory requirements and obtain additional regulatory approvals or clearances for new reagent kits in a timely manner. |
Luminescence immunoassay technology is a well established method of clinical diagnosis. As a result, our ECLIA system is competing in a market in which there are already many established industry players. We cannot assure you that we will be able to successfully market or sell our ECLIA system in the future. We also cannot assure you that our ECLIA system, or any future enhancements to our ECLIA system, will generate adequate revenue to offset our investments and costs in acquiring, developing or marketing our ECLIA system. If there is insufficient demand for our ECLIA system, our business, financial condition and results of operations may be harmed. In addition, any announcement of new products, services or enhancements by us or our competitors may cause our customers to cancel or postpone purchasing decisions for our existing products in anticipation of these new products, services or enhancements.
6
Table of Contents
The long-term effectiveness of HIFU medical devices for tumor treatment has not been proven.
HIFU therapy for tumors is a relatively new type of tumor therapy. We sold our first HIFU therapy system in November 1999. Although current studies on the use of HIFU medical devices for tumor treatment, including our HIFU therapy system, have shown a reduction in short-term side effects compared to radiotherapy and chemotherapy and suggest an improvement in long-term results, there are presently very few long-term studies of over five years on the effectiveness of using HIFU technology for the treatment of tumors. We plan to participate in future long-term studies of the effectiveness of our HIFU therapy system. If these studies fail to confirm the effectiveness of our HIFU therapy system or other HIFU medical devices, our sales could decline. In addition, some of the data produced in currently available studies were not randomized and/or included small patient sample sizes. Consequently, the results of these studies may not be representative of the results that may be achieved in studies involving larger patient sample sizes. Moreover, there may be other clinical studies published on our HIFU therapy system of which we are not aware and which contain different conclusions with respect to the safety, effectiveness or other aspects of our technologies. Our customers and users of our products may conclude that our HIFU therapy system is not an acceptable treatment regimen, that the technologies underlying our system are ineffective or unsafe, or that our system is less effective or safe than other therapies. This could result in a decrease in our sales, which would have a material adverse effect on our business, results of operations and financial condition.
If we fail to protect our intellectual property rights, our competitors may take advantage of our proprietary technology and know-how and compete directly against us.
Our success depends, in part, on our ability to protect our proprietary technologies. In China, we own seven invention patents and six utility patents directed towards various aspects of our HIFU therapy system and one utility patent relating to our ECLIA system. We also have 12 invention patent applications and seven utility patent applications pending, which are directed towards our HIFU therapy system and ECLIA system. We have also made applications for four patents directed toward our HIFU technology in each of the following countries outside China: the United States, the United Kingdom, Germany, Japan, South Korea, Saudi Arabia and India. As of September 27, 2006, we have been granted three out of the four patents we have applied for in the United Kingdom. Due to the different regulatory bodies and varying requirements in these countries, we cannot assure you that we will be able to obtain patent protection for all or any aspects of our HIFU technology in all or any of these countries. We have not applied for any patents outside of China for our ECLIA system. The process of seeking patent protection can be lengthy and expensive, and we cannot assure you that our patent applications will result in patents being issued or that our existing or future issued patents will be sufficient to provide us with meaningful protection or commercial advantage. Our patents and patent applications may be challenged, invalidated or circumvented in the future. We cannot assure you that our current or potential competitors, many of which have substantial resources and have made substantial investments in competing technologies, do not have, and will not develop, products that compete directly with our products despite our intellectual property rights.
We also rely on trade secrets, proprietary know-how and other non-patentable technology, which we seek to protect through non-disclosure agreements with employees. We cannot assure you that these non-disclosure agreements will not be breached, that we will have adequate remedies for any breach, or that our trade secrets, proprietary know-how and other non-patentable technology will not otherwise become known to, or be independently developed by, our competitors.
Implementation and enforcement of PRC intellectual property-related laws has historically been deficient and ineffective, and is hampered by corruption and local protectionism. Accordingly, intellectual property rights and confidentiality protections in China may not be as effective as in the United States or other countries. Policing unauthorized use of proprietary technology is difficult and expensive, and we might need to resort to litigation to enforce or defend patents issued to us or to determine the enforceability, scope and validity of our proprietary rights or those of others. The experience and capabilities of PRC courts in handling intellectual property litigation varies, and outcomes are unpredictable. Further, such litigation may require significant
7
Table of Contents
expenditure of cash and management efforts and could harm our business, financial condition and results of operations. An adverse determination in any such litigation will impair our intellectual property rights and may harm our business, prospects and reputation.
We may be exposed to intellectual property infringement and other claims by third parties, which, if successful, could cause us to pay significant damage awards and incur other costs.
Our success also depends in large part on our ability to use and develop our technology and know-how without infringing the intellectual property rights of third parties. As litigation becomes more common in China in resolving commercial disputes, we face a higher risk of being the subject of intellectual property infringement claims. The validity and scope of claims relating to medical device technology patents involve complex scientific, legal and factual questions and analysis and, therefore, may be highly uncertain. The defense and prosecution of intellectual property suits, patent opposition proceedings and related legal and administrative proceedings can be costly and time consuming, with unpredictable outcomes, and may significantly divert the efforts and resources of our technical and management personnel. Courts in some jurisdictions in China are inexperienced in these types of cases, and may be influenced by local protectionism. An adverse determination in any such litigation or proceedings to which we may become a party could subject us to significant liability, including damage awards, to third parties, require us to seek licenses from third parties, to pay ongoing royalties, or to redesign our products or subject us to injunctions preventing the manufacture and sale of our products. Protracted litigation could also result in our customers or potential customers deferring or limiting their purchase or use of our products until resolution of such litigation.
We are dependent on a small number of distributors for a significant portion of our revenues, and this dependence is likely to continue in the future.
For the fiscal year ended March 31, 2006, sales to our top five customers accounted for 62% of our revenues. These customers are distributors of medical devices and supplies. We do not have long-term contracts with our distributors, but rather typically sell to them on a unit-by-unit basis. We expect that a substantial portion of our sales will continue to be generated by a relatively small group of distributors that may change from year to year. Because our main product, the HIFU therapy system, is sold at a relatively high unit price, the size and timing of individual orders can have a significant effect on our quarterly results. We cannot assure you that any distributor will continue to purchase our products at the same levels as in prior years or that such relationships will continue. We expect that in any given period a relatively small, and changing, number of distributors will continue to account for a significant portion of our revenues. If we are unable to manage our distributor network, our reputation among end users of our products and our revenues may be adversely affected.
Our business strategy to grow through acquisitions of new products or technologies may result in integration costs, failures and dilution to existing stockholders.
An important business strategy of ours is to acquire medical technologies with significant market potential and bring them to the market. We continue to seek suitable opportunities both inside and outside China to acquire new products or technologies, particularly those that could assist us in advancing our current products, technologies and market penetration, or in expanding our product offerings or technologies. If we decide to acquire another company or its assets in order to obtain its products or technologies, we would face a number of risks including consummating the acquisition on unfavorable terms and not obtaining adequate financing, which may adversely affect our ability to develop new products and services and to compete in our rapidly changing marketplace. Successful management and integration of acquisitions are subject to a number of risks, including difficulties in assimilating acquired operations and managing remote operations, potential loss of key employees, diversion of management’s attention from core business operations, assumption of contingent liabilities and incurrence of potentially significant write-offs, which may adversely affect our business or results of operations. In addition, if we consummate such an acquisition through an exchange of our securities, our existing stockholders could suffer dilution.
8
Table of Contents
Rapid growth and a rapidly changing operating environment may strain our limited resources.
Our growth strategy includes our efforts to build our brand, increase market penetration of our HIFU therapy system and grow our ECLIA system sales by targeting small- and medium-sized hospitals in China. This growth strategy requires significant capital resources, and we may not generate an adequate return on our investment. Our growth may involve the acquisition of new technologies, businesses, products or services or the creation of strategic alliances in areas in which we do not currently operate. This could require our management to develop expertise in new areas, manage new business relationships and attract new types of customers. We may also experience difficulties integrating these acquired businesses, products or services into our existing business and operations. The success of our growth strategy also depends in part on our ability to utilize our financial, operational and management resources and to attract, train, motivate and manage an increasing number of employees. The success of our growth strategy depends on a number of internal and external factors, such as:
| • | the growth of the market for medical devices and supplies in China; |
| • | competition from other manufacturers of HIFU tumor therapy devices and alternative methods of tumor therapy; and |
| • | competition from other companies that offer IVD systems in China, |
many of which are beyond our control. We may not be able to implement our growth strategy successfully or manage our expansion effectively.
Our limited operating history makes evaluating our business and prospects difficult.
We commenced operations in July 1999, and delivered the first commercial unit of our HIFU therapy system in November 1999. We only started to market and sell our ECLIA system in September 2004. As a result, we have a limited operating history which may not provide a meaningful basis for you to evaluate our business, financial performance and prospects. We may not have sufficient experience to address the risks frequently encountered by early-stage companies, and as a result we may not be able to:
| • | maintain profitability; |
| • | preserve our leading position in the market of HIFU tumor therapy devices; |
| • | diversify our revenue sources by successfully marketing and selling our ECLIA system; |
| • | acquire and retain customers; |
| • | attract, train, motivate and retain qualified personnel; |
| • | keep up with evolving industry standards and market developments; |
| • | increase the market awareness of our products; |
| • | respond to competitive market conditions; |
| • | maintain adequate control of our expenses; |
| • | manage our relationships with our suppliers and distributors; or |
| • | protect our proprietary technologies. |
If we are unsuccessful in addressing any of these risks, our business may be materially and adversely affected.
We are controlled by a small number of shareholders and their affiliated entities, and their interests may not be aligned with the interests of our other shareholders.
Currently, Chengxuan and Golden Meditech and their affiliated entities beneficially own over 37% of our outstanding ordinary shares. These shareholders, acting together, could exert substantial influence over matters such as electing directors and approving material mergers, acquisitions or other business combination transactions.
9
Table of Contents
The interests of these significant shareholders may differ from the interests of our other shareholders. We, Chengxuan, Golden Meditech and GE entered into a shareholders’ agreement in June 2005. The rights and obligations contained in the shareholders’ agreement may be terminated by written agreement among Chengxuan, Golden Meditech and GE. If any of Chengxuan’s, Golden Meditech’s or GE’s ownership interest in our company falls below 5% of our outstanding shares, calculated on a fully diluted basis, such shareholder will cease to be a party to the June 2005 shareholders’ agreement and will no longer have any rights or obligations under that agreement. On June 21, 2006, GE’s ownership interest in our company fell below 5% of our outstanding shares and ceased to be a party to the shareholders’ agreement.
As of September 27, 2006, the effective provisions of the June 2005 shareholders’ agreement include:
| • | Golden Meditech and its respective affiliates will have complete access to, and full use of, the clinical data of our subsidiary, YDME Beijing, relating to clinical cases from hospitals, other medical institutions and medical professionals using our HIFU therapy system in connection with their business, provided that we have first obtained the necessary consent. Such clinical data include hospital survey information, medical records, test reports and imaging records before and after treatment using our HIFU therapy system. |
| • | If any of Chengxuan or Golden Meditech is offered a business opportunity that is primarily based in China and that competes with our HIFU business, it will provide us with notice of its intention to pursue such business opportunity and use reasonable efforts to enable us to participate in such project if we express an interest in doing so. |
In addition, the contractual rights discussed above, together with the concentration of ownership of these shareholders, may discourage, delay or prevent a change in control of our company, which could deprive our shareholders of an opportunity to receive a premium for their shares as part of a sale of our company and might reduce the price of our ADSs. These actions may be taken even if they are opposed by our other shareholders. In cases where the interests of our significant shareholders are aligned and they vote together, these shareholders will also have the power to prevent or cause a change in control. In addition, these shareholders could divert business opportunities from us to themselves or others.
We are highly dependent on senior management and key research and development personnel.
We are highly dependent on our senior management to manage our business and operations and our key research and development personnel for the development of new technologies and applications and the enhancement of our existing products. In particular, we rely substantially on our chairman and chief executive officer, Mr. Xiaodong Wu, to manage our operations. We also depend on our key research personnel such as Mr. Xiaolin Yang and Mr. Houqing Zhu. In addition, we also rely on customer service personnel for the installation and support of our products and on marketing and sales personnel, engineers and other personnel with technical and industry knowledge to market, sell, install and service our products. We do not maintain key man life insurance on any of our senior management or key personnel. The loss of any one of them, in particular Mr. Wu, Mr. Yang or Mr. Zhu, would have a material adverse effect on our business and operations. Competition for senior management and research and development personnel is intense, and the pool of suitable candidates is limited. We may be unable to locate a suitable replacement for any senior management or key research and development personnel that we lose. In addition, if Mr. Wu, Mr. Yang, Mr. Zhu or any other member of our senior management or key research and development personnel joins a competitor or forms a competing company, they may compete with us for customers, business partners and other key professionals and staff members of our company. Although each of our senior management and key research and development personnel has signed a confidentiality and non-competition agreement in connection with his employment with us, we cannot assure you that we will be able to successfully enforce these provisions in the event of a dispute between us and any member of our senior management or key research and development personnel.
We compete for qualified personnel with other medical technology companies, medical device and supplies manufacturers, universities and research institutions. Intense competition for these personnel could cause our
10
Table of Contents
compensation costs to increase significantly, which could have a material adverse effect on our results of operations. Our future success and ability to grow our business will depend in part on the continued service of these individuals and our ability to identify, hire and retain additional qualified personnel. If we are unable to attract and retain qualified employees, we may be unable to meet our business and financial goals.
We rely on single-source suppliers to provide materials and major components. A significant interruption in supply could prevent or limit our ability to accept and fulfill orders for our products.
We purchase all our materials and major components from third-party suppliers and assemble our products in-house. We currently rely on single-source suppliers to provide a majority of our materials and major components. We do not currently have any long-term supply contracts. Our purchases are made on a purchase order basis. If the supply of certain materials, components or services were interrupted, our own manufacturing process would be delayed. We may be unable to secure alternative sources of supply in a timely and cost-effective manner. The ultrasound imaging system and the treatment bed, two of the major components of our HIFU therapy system, are manufactured by GE and Beijing Tianyuan Kehui Company Ltd., or Beijing Tianyuan Kehui, respectively, based on our specifications. If we are unable to obtain these components from GE and Beijing Tianyuan Kehui, it may be difficult to find alternate suppliers that can provide replacement parts of sufficient quality according to our specifications. We purchase the materials for our ECLIA system and reagent kits from various domestic and international suppliers, some of which are our sole supplier for certain materials. The failure of any of these suppliers to provide materials to us, particularly if we are unable to obtain these materials from alternative sources on a timely basis or on commercially reasonable terms, could impair our ability to manufacture our ECLIA system and reagent kits or increase our costs. We also plan to increase the production and sales of our products in the future. If we are unable to obtain required materials and components that meet our production standards in sufficient quantities or at acceptable cost, we will be unable to increase our production and sales as planned. Failure to obtain adequate supplies of materials, components or services in a timely and cost-effective manner could delay our delivery to our customers. This could harm our reputation, cause us to lose sales, and force us to use more expensive sources of supply, which could significantly increase our production costs and harm our operating results. Any of these occurrences could have a material adverse effect on our business, financial condition and results of operations.
We need to expand our existing sales force and distribution network to maintain and grow our business and revenues.
We currently market and sell our HIFU therapy system through several third-party distributors and our own direct sales force. We market and sell our ECLIA system through a number of third-party distributors. As part of our growth plan, we intend to increase the number of distributors we utilize to distribute our HIFU therapy system and ECLIA system. We have limited experience in managing a large distributor network. We cannot assure you that we will be able to maintain or develop our relationships with third-party distributors. If we fail to do so, our sales could fail to grow or could even decline, and our ability to grow our business could be adversely affected. The expansion of our distribution network is also likely to require a significant investment of financial resources and management efforts, and the benefits, if any, that we gain from such an expansion may not be sufficient to generate an adequate return on our investment.
Our costs could substantially increase if we experience a significant number of warranty claims.
We provide 12-month product warranties against technical defects of our HIFU therapy system and ECLIA analyzer. Our product warranty requires us to repair all mechanical malfunctions and, if necessary, replace defective components. Historically, we have received a limited number of warranty claims for our HIFU therapy system. As we only began selling our ECLIA analyzer in September 2004, we currently do not have sufficient historical data on the warranty claims for our ECLIA analyzer. Consequently, the costs associated with our warranty claims have historically been relatively low. We generally do not accrue any liability for potential warranty claims at the time of sales, but rather at the time claims are actually made. If we experience an increase
11
Table of Contents
in warranty claims or if our repair and replacement costs associated with warranty claims increase significantly, we will begin to incur liabilities for potential warranty claims at the time of sale of our products. An increase in the frequency of warranty claims or amount of warranty costs may harm our reputation and could have a material adverse effect on our financial condition and results of operations.
Our operations might be interrupted by the occurrence of a natural disaster or other catastrophic events.
All of our manufacturing and research and development facilities are located in a single location in Beijing, China. We do not maintain back-up facilities, so we depend on our manufacturing and other facilities for the continued operation of our business. Natural disasters or other catastrophic events, including power interruptions, water shortages, storms, fires, earthquakes, terrorist attacks and wars could disrupt our operations. We might suffer losses as a result of business interruptions and our operations and financial results might be materially and adversely affected. Moreover, any such event could delay our research and development programs.
If we are unable to successfully operate and manage our manufacturing operations, we may experience a decrease in revenues.
As we ramp up our manufacturing operations to accommodate our planned growth, we may encounter difficulties associated with increasing production scale, including shortages of qualified personnel to operate our equipment, assemble our products or manage manufacturing operations, as well as shortages of key raw materials or components for our products. In addition, we may also experience difficulties in producing sufficient quantities of products or in achieving desired product quality. If we are unable to successfully operate and manage our manufacturing operations to meet our needs, we may not be able to provide our customers with the quantity or quality of products they require in a timely manner. This could cause us to lose customers and result in reduced revenues.
Our business may suffer if we are unable to collect payments from our customers on a timely basis.
Our distributors and other customers must make a significant commitment of capital to purchase our products. Any downturn in the businesses of our distributors and other customers could reduce their willingness or ability to pay us. We have historically been able to collect substantially all of our receivable balances. Consequently, we have not found it necessary to provide an allowance for doubtful accounts historically. The failure of any of our distributors to make timely payments could require us to recognize an allowance for doubtful accounts, which could have a material adverse effect on our results of operations and financial condition.
Fluctuations in our quarterly operating results could cause our ADS price to decline.
Our revenues and operating results have fluctuated in the past and may continue to fluctuate significantly from quarter to quarter depending upon numerous factors. Our third and fourth fiscal quarters, ending December 31 and March 31, respectively, have historically been our strongest. We believe that the relatively stronger performance of our third and fourth fiscal quarters has been largely due to the budget cycles of hospitals in China. Hospitals in China typically make their capital expenditure decisions, such as decisions on whether to purchase our HIFU therapy system, between June and December. Based on orders during that period, our HIFU therapy systems are usually shipped to customers between September and March, which are our third and fourth fiscal quarters. Other factors that may affect the fluctuation of our quarterly operating results include changes in pricing policies by us or our competitors, the length of our sales cycle, the timing and market acceptance of new product introductions and product enhancements by us or our competitors, customer order deferrals in anticipation of new or enhanced products offered by us or our competitors, the loss of key sales personnel or distributors, changes in government policies or regulations and a downturn in general economic conditions in China. Furthermore, because we have historically sold a relatively small number of our main product, the HIFU therapy system, at a relatively high unit price, the size and timing of each individual order have a particularly
12
Table of Contents
significant effect on our results of operations in any given quarter. Many of these factors are beyond our control, and you should not rely on our results of operations for prior quarters as an indication of our results in any future period. As our revenues vary significantly from quarter to quarter, our business is difficult to predict and manage, and our quarterly results could fall below investor expectations, which could cause our ADS price to decline.
We are subject to product liability exposure and have limited insurance coverage.
As our main product, the HIFU therapy system, is a medical device for the treatment of patients, we are exposed to potential product liability claims in the event that the use of our products causes or is alleged to have caused personal injuries or other adverse effects. A successful product liability claim against us could require us to pay substantial damages. Product liability claims against us, whether or not successful, are costly and time-consuming to defend. Also, in the event that our products prove to be defective, we may be required to recall or redesign such products. We have a product liability insurance policy with an annual aggregate insured amount of RMB4.0 million (US$0.5 million) to cover potential product liability arising from the use of our HIFU therapy system. However, as the insurance industry in China is still in an early stage of development, product liability insurance available in China offers limited coverage compared to coverage offered in many other countries. As a result, future liability claims could be excluded from our policies or exceed the coverage limits of our policies. We also cannot assure you that product liability insurance will continue to be available on commercially reasonable terms, if at all. To date, we have not been subject to any product liability claim, but we cannot assure you that such claim will not be brought against us in the future. A product liability claim, with or without merit, could result in significant adverse publicity against us, and could have a material adverse effect on the marketability of our products and our reputation, which in turn, could have a material adverse effect on our business, financial condition and results of operations. In addition, we do not have any business interruption insurance coverage for our operations. Any business disruption or natural disaster could result in substantial costs and diversion of resources.
Our future capital needs are uncertain and we may need to raise additional funds in the future.
We may require additional cash resources in the future due to:
| • | changed business conditions or other future developments; |
| • | the receipt of, and the time and expenses required to obtain and maintain, regulatory clearances and approvals; |
| • | the resources we devote to developing, marketing and producing our products; |
| • | our ability to identify and our desire or need to pursue acquisitions or other investments; and |
| • | the extent to which our products generate market acceptance and demand. |
We cannot assure you that our revenues will be sufficient to meet our operational needs and capital requirements. In addition, we cannot assure you that financing will be available in amounts or on terms acceptable to us, if at all. Our future capital needs and other business reasons could require us to sell additional equity or debt securities or obtain a credit facility. The sale of additional equity or equity-linked securities could result in additional dilution to our shareholders. The incurrence of indebtedness would result in increased debt service obligations and could result in operating and financing covenants that would restrict our operations or our ability to pay dividends to our shareholders.
If a poll is not demanded at our shareholder meeting, voting will be by show of hands and shares will not be proportionately represented. Shareholder resolutions may be passed without the presence of the majority of our shareholders in person or by proxy.
Voting at any of our shareholder meetings is by show of hands unless a poll is demanded. A poll may be demanded by the chairman of our board of directors or by any shareholder present in person or by proxy. If a poll
13
Table of Contents
is demanded, each shareholder present in person or by proxy will have one vote for each ordinary share registered in his name. If a poll is not demanded, voting will be by show of hands and each shareholder present in person or by proxy will have one vote regardless of the number of shares registered in his name. In the absence of a poll, shares will therefore not be proportionately represented. In addition, the quorum required for our shareholder meetings consists of shareholders who hold at least one-third of our ordinary shares being present at a meeting in person or by proxy. Therefore, subject to the requisite majorities, shareholder resolutions may be passed at our shareholder meetings without the presence of the majority of our shareholders in person or by proxy.
If we fail to achieve and maintain effective internal control over financial reporting in accordance with the Sarbanes-Oxley Act, we may suffer a loss of investor confidence which may have a material adverse effect on the trading price of our ADSs.
We are subject to reporting obligations under the U.S. securities laws. SEC, as required by Section 404 of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, adopted rules requiring every public company to include a management report on such company’s internal control over financial reporting in its annual report, which must also contain management’s assessment of the effectiveness of our internal control over financial reporting. In addition, an independent registered public accounting firm must attest to and report on management’s assessment of the effectiveness of our internal control over financial reporting. These requirements will first apply to our annual report on Form 20-F for the fiscal year ending March 31, 2008. We cannot assure you that our management will be able to conclude that our internal control over financial reporting is effective when the requirements of Section 404 become applicable to us. Moreover, even if our management concludes that our internal control over financial reporting is effective, our independent registered public accounting firm may still conclude that our internal control is not effective. Furthermore, during the course of evaluation, documentation and attestation, we may identify weaknesses and deficiencies that we may not be able to remedy in time to meet the deadline imposed by the Sarbanes-Oxley Act for compliance with the requirements of Section 404. If we fail to achieve and maintain effective internal control over financial reporting in accordance with the Sarbanes-Oxley Act, we could suffer a loss of investor confidence in the reliability of our financial statements, which in turn could negatively impact the trading price of our ADSs, result in lawsuits being filed against us by our shareholders or otherwise harm our reputation. Furthermore, we have incurred and anticipate that we will continue to incur considerable costs and use significant management time and other resources in an effort to comply with Section 404 and other requirements of the Sarbanes-Oxley Act.
Our reported earnings could decline if we recognize impairment losses on intangible assets relating to our acquisition of the ECLIA technology.
As a result of our acquisition of the ECLIA technology in August 2004, we have recorded certain intangible assets which are amortized over their respective estimated useful lives and are tested for impairment by us annually or more frequently if an event occurs or a circumstance develops that would likely result in impairment. Examples of such events or circumstances include, but are not limited to, a significant adverse change in legal or business climate, an adverse regulatory action or unanticipated competition. See “Item 5. Operating and Financial Review and Prospects—Critical Accounting Policies.” In the future, we could recognize impairment losses on intangible assets and that impairment could result in a charge to our reported results of operations, which would cause our reported earnings to decline.
Changes in accounting standards regarding stock option plans may have a material adverse effect on our reported earnings, may harm our ability to attract and retain employees, and may also negatively impact our results of operations.
Financial Accounting Standards Board Statement of Financial Accounting Standards No. 123 (Revised 2004), “Share-Based Payments,” or SFAS 123R, which we adopted for our fiscal period beginning April 1, 2006, requires all companies to recognize as an expense the fair value of stock options granted to employees. Prior to February 2005, we did not have any stock option plan, so we did not incur any share-based compensation
14
Table of Contents
charges. On February 2, 2005, we adopted our 2005 stock option plan and have, to date, granted options to acquire 800,000 of our ordinary shares. Prior to April 1, 2006, we accounted for these options in accordance with Accounting Principles Board Opinion No. 25, “Accounting for Stock Issued to Employees,” or APB 25, and its related interpretations, which require us to recognize compensation expense for stock options we grant where the exercise price is less than the deemed fair value of our ordinary shares on the date of the grant and were not required to record share-based compensation charges if the stock option exercise price equals or exceeds the deemed fair value of our ordinary shares at the date of grant. However, beginning on April 1, 2006, we started to account for the compensation costs for stock options using a fair-value based method and recognize expenses in our consolidated statement of income in accordance with the relevant U.S. GAAP rules, which may have a material and adverse effect on our reported earnings. Moreover, the additional expenses associated with the grant of options may reduce the attractiveness of granting stock options. However, if we reduce the scope of our employee stock option plan, we may not be able to attract and retain key personnel, as stock options play an important role in employee recruitment and retention. Accordingly, our results of operations will be negatively impacted if we grant options pursuant to our 2005 stock option plan as an employee recruitment and retention tool.
Risks Related to Our Industry
New product development in the medical device and supply industry is both costly and labor-intensive and has a very low rate of successful commercialization.
Our success will depend in part on our ability to enhance our existing products and technologies and to develop and acquire new products or technologies. The development process for medical technology is complex and uncertain, as well as time-consuming and costly. Product development requires the accurate assessment of technological and market trends as well as precise technological execution. We cannot assure you that:
| • | our product or technology development will be successfully completed; |
| • | necessary regulatory clearances or approvals will be granted by the SFDA or other regulatory bodies as required on a timely basis, or at all; or |
| • | any product or technology we develop can be commercialized or will achieve market acceptance. |
Also, we may be unable to locate suitable products or technologies to acquire or acquire such products or technologies on commercially reasonable terms. Failure to develop or acquire, obtain necessary regulatory clearances or approvals for, or successfully commercialize or market potential new products or technologies could have a material adverse effect on our financial condition and results of operations.
In order to manufacture and market our products, we are required to obtain various authorizations from governmental regulatory authorities in China. In some cases, we have been operating without these authorizations, which could subject us to significant penalties.
Under a regulation enacted by the SFDA in September 2002, reagents are required to be registered either as biological reagents, which are subject to regulatory controls similar to those governing pharmaceutical products, or chemical and bio-chemical reagents, which are subject to regulatory controls similar to those governing medical devices. We are required to obtain a registration certificate for each IVD reagent prior to selling that reagent. Of the 56 reagents that we market, 30 are covered by registration certificates. Of the 26 reagents we currently sell for which we do not have registration certificates, we have submitted registration applications for 22.
As a result, we are marketing 26 IVD reagents without the necessary registration certificates. We may not be able to obtain registration certificates for these IVD reagents or for any reagents that we develop in the future. Moreover, we may not be able to arrange for the transfer to us of the IVD registration certificates currently issued in the name of Beijing Weixiao.
15
Table of Contents
We are also required to obtain a registration certificate from the SFDA in order to sell our HIFU therapy system. We obtained the necessary registration certificate in 2003. As this certificate will expire in January 2007, we will need to renew the registration by re-submitting an application and obtaining SFDA approval. Renewing our registration certificate entails submission of various information and the review of that submission by the applicable regulatory authorities. We may not be able to obtain a renewal in a timely fashion or at all.
We believe that no additional approval from the SFDA will be required for our third generation HIFU product; however, the SFDA may interpret these regulations differently than we, and we may be required to obtain additional approval from the SFDA for this product. If we are unable to renew our registration certificate or obtain re-approval for our third generation HIFU therapy system in a timely manner, we may not be able to market this product.
The Chinese regulatory authorities may bring enforcement actions against us for past or on-going non-compliance with these regulations. These enforcement actions could result in fines, injunctions and civil penalties, recall or seizure of our products, the imposition of operating restrictions, partial suspension or complete shut down of our production facilities and criminal prosecution. Moreover, regulatory agencies in China are known to periodically, and sometimes abruptly, change their enforcement practices, and so prior enforcement activity, or lack of enforcement activity, is not necessarily predictive of future actions. Any enforcement actions against us could have a material and adverse effect on us and the market price of our ADSs.
Competition in the markets in which we operate is expected to increase in the future.
Our HIFU therapy system competes with both existing and emerging alternative treatment methods for tumors, including other tumor therapy devices that use similar technologies. Competition in the markets in which we operate is expected to increase in the future. For our HIFU therapy system, we believe that competition comes from new and existing manufacturers of HIFU tumor therapy devices, as well as traditional cancer treatment methods such as surgery, radiotherapy and chemotherapy. In addition, we also compete with other minimally invasive treatments, including radio frequency ablation, microwave thermo-coagulation and cryosurgery, and targeted therapeutics. We believe that other companies offer HIFU technology for the treatment of tumors including EDAP TMS S.A., or EDAP, Focus Surgery, Inc., or Focus Surgery, and InSightec. According to Focus Surgery’s website, its HIFU product is being used in China; and although no other overseas-based company currently offers or sells its products in China, they may choose to enter the China market in the future, either independently or in cooperation with our existing competitors in China. In China, competitors for our HIFU therapy system include Chongqing Haifu Technology Co., Ltd., or Chongqing Haifu, and Shanghai A&S Science Technology Development Co., Ltd., or Shanghai A&S. Our ECLIA system competes with IVD systems and reagents offered in the China market by many established international companies, including Abbott Diagnostics, Bayer AG, Beckman Coulter, Diagnostic Products Corporation, PerkinElmer, Inc., Johnson & Johnson and Roche Group. There are also a number of China-based manufacturers who also offer luminescence immunoassay systems but their market shares are relatively small. See “Item 4. Information on the Company—B. Business Overview—Competition.”
Certain of our existing and potential competitors have significantly greater financial, research and development, sales and marketing, personnel resources and other resources than we do. Competition will intensify as other companies enter our markets. Competing companies may succeed in developing products that are more effective or less costly than those that we may offer, and these companies may also be more successful in marketing their products. Competing companies may also introduce competitive pricing measures that adversely affect our sales levels and margins. If we do not adequately address our competitive challenges, we could lose sales and market share and fail to grow our business as planned, which would have a material adverse effect on our financial condition, results of operations and future growth.
In addition, we believe that corrupt practices in the healthcare industry in China still occur. In order to increase sales, certain manufacturers or distributors of medical devices may pay kickbacks to hospital personnel
16
Table of Contents
who make procurement decisions. We prohibit our employees from engaging in such practices and, to our knowledge, none of our distributors engages in such practices. However, as competition intensifies in the medical device and supplies industry in China, we may lose sales, customers or contracts to competitors to the extent we or our distributors refuse to engage in such practices.
The tumor therapy and IVD industries are characterized by constant technological change, and if we fail to respond effectively to technological changes, we could lose our competitive advantage.
The tumor therapy and IVD industries in which we currently compete are characterized by:
| • | changing technologies; |
| • | frequent new product introductions and enhancements; |
| • | changing customer needs; and |
| • | product obsolescence. |
To develop new products and designs, we must develop, gain access to and use leading technologies in a cost-effective and timely manner and continue to expand our technical and design expertise. Failure to do so could cause us to lose our competitive position and have a material adverse effect on our revenues in the future.
The price and sales of our HIFU therapy system may be adversely affected by reductions in treatment fees by the Chinese government.
Treatment fees for HIFU tumor therapy, like many other medical treatments, are subject to prices set by provincial governments in China, and these prices can be adjusted downward or upward from time to time. If the treatment fees for HIFU tumor therapy are reduced by the government, some hospitals and distributors may be discouraged from buying our HIFU therapy system, which would reduce our sales. We may need to decrease the price of our HIFU therapy system to provide hospitals acceptable returns on their purchases. We cannot assure you that our business or results of operations will not be adversely affected by a reduction in treatment fees for HIFU tumor therapy in the future.
Governmental and private health insurers may not provide sufficient, or any, coverage for the use of our HIFU therapy system.
Our ability to market and sell our HIFU therapy system to hospitals will depend in part on the availability of governmental and private health insurance in China for treatments using our HIFU therapy system. The insurance coverage for treatments using new medical devices, such as our HIFU therapy system, is subject to significant uncertainty and varies from region to region, as local government approvals for such coverage must be obtained in each geographic region in China. In addition, provincial governments may change, reduce or eliminate the government insurance coverage currently available for HIFU tumor therapy. We cannot assure you that adequate third-party insurance coverage will be generally available for patients who elect to use our HIFU therapy system for the treatment of tumors. In the absence of sufficient medical insurance coverage for the use of our HIFU therapy system, patients may choose alternative treatment methods that are covered by insurance, and hospitals may use their limited funds to buy products for these alternative treatments, which would reduce demand for our products and, consequently, our sales. If this occurs, our business, results of operations and financial condition could be materially and adversely affected.
Any product recall could have a material adverse effect on our business, results of operations and financial condition.
Complex medical devices, such as our HIFU therapy system, can experience performance problems that require review and possible corrective action by the manufacturer. From time to time, we receive reports from
17
Table of Contents
users of our products relating to performance problems they have encountered. We expect that we will continue to receive customer reports regarding performance problems they encounter through the use of our products. Furthermore, component failures, manufacturing errors or design defects that could result in an unsafe condition or injury to the patient might occur. Any serious failures or defects could cause us to withdraw or recall products, which could result in significant costs such as repair and product replacement costs. We cannot assure you that market withdrawals or product recalls will not occur in the future, which could have a material adverse effect on our business, financial condition and results of operations.
Risks Related to Doing Business in China
Adverse changes in political, economic and other policies of the Chinese government could have a material adverse effect on the overall economic growth of China, which could reduce the demand for our products and materially and adversely affect our competitive position.
All of our business operations are conducted in China, and all of our sales are made in China. Accordingly, our business, financial condition, results of operations and prospects are affected significantly by economic, political and legal developments in China. The Chinese economy differs from the economies of most developed countries in many respects, including:
| • | the extent of government involvement; |
| • | the level of development; |
| • | the growth rate; |
| • | the control of foreign exchange; |
| • | the allocation of resources; |
| • | an evolving regulatory system; and |
| • | lack of sufficient transparency in the regulatory process. |
While the Chinese economy has experienced significant growth in the past 20 years, growth has been uneven, both geographically and among various sectors of the economy. The Chinese government has implemented various measures to encourage economic growth and guide the allocation of resources. Some of these measures benefit the overall Chinese economy, but may also have a negative effect on us. For example, our financial condition and results of operations may be adversely affected by government control over capital investments or changes in tax regulations that are applicable to us.
The Chinese economy has been transitioning from a planned economy to a more market-oriented economy. Although in recent years the Chinese government has implemented measures emphasizing the utilization of market forces for economic reform, the reduction of state ownership of productive assets and the establishment of sound corporate governance in business enterprises, a substantial portion of the productive assets in China is still owned by the Chinese government. The continued control of these assets and other aspects of the national economy by the Chinese government could materially and adversely affect our business. The Chinese government also exercises significant control over Chinese economic growth through the allocation of resources, controlling payment of foreign currency-denominated obligations, setting monetary policy and providing preferential treatment to particular industries or companies. Efforts by the Chinese government to slow the pace of growth of the Chinese economy could result in decreased capital expenditure by hospitals, which in turn could reduce demand for our products.
Moreover, the political relationship between the United States, Europe, or other Asian nations and China is subject to sudden fluctuation and periodic tension. Changes in political conditions in China and changes in the state of foreign relations are difficult to predict and could adversely affect our operations or cause our services to become less attractive. This could lead to a decline in our profitability.
18
Table of Contents
Any adverse change in the economic conditions or government policies in China could have a material adverse effect on overall economic growth and the level of healthcare investments and expenditures in China, which in turn could lead to a reduction in demand for our products and consequently have a material adverse effect on our businesses.
Future changes in laws, regulations or enforcement policies in China could adversely affect our business.
Laws, regulations or enforcement policies in China, including those regulating medical devices and supplies, are evolving and subject to future change. For example, under a regulation enacted by the SFDA in September 2002, reagents are required to be registered with the SFDA and are subject to regulatory controls. To date, we have obtained SFDA registrations and/or approvals for 30 of the 56 reagents we sell and are in the process of obtaining the required registrations and/or approvals for our other reagents. Moreover, in October 2004, the SFDA imposed certain good manufacturing practice, or GMP, requirements for IVD reagents. As a result, all manufacturers of IVD reagents were obligated to meet GMP standards by January 1, 2006. We are currently in compliance with such GMP requirements and have obtained GMP certification for our ECLIA system in November 2005. Future changes in laws, regulations or administrative interpretations, or stricter enforcement policies by the Chinese government, could impose more stringent requirements on us, including fines or other penalties. Changes in applicable laws and regulations may also increase our operating costs. Compliance with such requirements could impose substantial additional costs or otherwise have a material adverse effect on our business, financial condition and results of operations. These changes may relax some requirements, which could be beneficial to our competitors or could lower market entry barriers and increase competition. Further, regulatory agencies in China may periodically, and sometimes abruptly, change their enforcement practice. Therefore, prior enforcement activity, or lack of enforcement activity, is not necessarily predictive of future actions. Any enforcement actions against us could have a material and adverse effect on us and the market price of our ADSs. In addition, any litigation or governmental investigation or enforcement proceedings in China may be protracted and may result in substantial cost and diversion of resources and management attention, negative publicity, damage to our reputation and decline in the price of our ADSs.
We rely on dividends paid by our subsidiary for our cash needs.
We are a holding company, and we rely on dividends paid by our wholly owned subsidiary, YDME Beijing, for our cash needs, including the funds necessary to pay dividends and other cash distributions to our shareholders, service any debt we may incur and pay our operating expenses. The payment of dividends in China is subject to limitations. Regulations in the PRC currently permit payment of dividends only out of accumulated profits as determined in accordance with accounting standards and regulations in China. YDME Beijing is also required to set aside at least 10% of its after-tax profit based on PRC accounting standards each year to its general reserves. These reserves are not distributable as cash dividends. In addition, if YDME Beijing incurs debt on its own behalf in the future, the instruments governing the debt may restrict YDME Beijing’s ability to pay dividends or make other distributions to us.
Restrictions on currency exchange may limit our ability to receive and use our revenues effectively.
We receive all of our revenues in Renminbi, which currently is not a freely convertible currency. A portion of our revenues may be converted into other currencies to meet our foreign currency obligations, including, among others, payment of dividends declared, if any, in respect of our ordinary shares. Under China’s existing foreign exchange regulations, YDME Beijing is able to pay dividends in foreign currencies without prior approval from the State Administration of Foreign Exchange, or the SAFE, by complying with certain procedural requirements. However, we cannot assure you that the PRC government will not take future measures to restrict access to foreign currencies for current account transactions.
YDME Beijing’s ability to obtain foreign exchange is subject to significant foreign exchange controls and, in the case of amounts under the capital account, requires the approval of and/or registration with PRC government
19
Table of Contents
authorities, including the SAFE. In particular, if YDME Beijing borrows foreign currency loans from us or other foreign lenders, it must do so within approved limits that satisfy its approval documentation and PRC debt to equity ratio requirements. Further, such loans must be registered with the SAFE. If we finance YDME Beijing by means of additional capital contributions, the amount of these capital contributions must first be approved by the relevant government approval authority. These limitations could affect the ability of YDME Beijing to obtain foreign exchange through debt or equity financing.
Fluctuation in the value of the Renminbi may have a material adverse effect on your investment.
The value of the Renminbi against the U.S. dollar and other currencies may fluctuate and is affected by, among other things, changes in China’s political and economic conditions. The conversion of Renminbi into foreign currencies, including U.S. dollars, has historically been set by the People’s Bank of China. On July 21, 2005, the PRC government changed its policy of pegging the value of the Renminbi to the U.S. dollar. Under the new policy, the Renminbi is permitted to fluctuate within a band against a basket of certain foreign currencies, determined by the Bank of China, against which it can rise or fall by as much as 0.3% each day. This change in policy resulted initially in an approximately 4.1% appreciation in the value of the Renminbi against the U.S. dollar. Since the adoption of this new policy, the value of Renminbi against the U.S. dollar has fluctuated on a daily basis within narrow ranges, but overall has further strengthened against the U.S. dollar. There remains significant international pressure on the PRC government to further liberalize its currency policy, which could result in a further and more significant appreciation in the value of the Renminbi against the U.S. dollar. As we import certain materials and supplies for reagent kits from the United States, Finland and Sweden, fluctuations in the value of the Renminbi against the currencies of those countries may increase the cost of our reagent kits. In addition, as we rely entirely on dividends paid to us by our wholly owned subsidiary YDME Beijing, any significant revaluation of the Renminbi may have a material adverse effect on our revenues and financial condition, and the value of, and any dividends payable on, our ADSs in foreign currency terms. For example, to the extent that we need to convert U.S. dollars we receive from our initial public offering into Renminbi for our operations, appreciation of the Renminbi against the U.S. dollar would have an adverse effect on the Renminbi amount we receive from the conversion. Conversely, if we decide to convert our Renminbi into U.S. dollars for the purpose of making payments for dividends on our ordinary shares or ADSs or for other business purposes, appreciation of the U.S. dollar against the Renminbi would have a negative effect on the U.S. dollar amount available to us. In addition, appreciation or depreciation in the value of the Renminbi relative to the U.S. dollar would affect our financial results reported in U.S. dollar terms without giving effect to any underlying change in our business or results of operations.
Our business benefits from certain government incentives. Expiration of, or changes to, these incentives could have a material adverse effect on our operating results.
The PRC government has provided various incentives to high technology companies, including YDME Beijing, in order to encourage development of the high technology industry. Such incentives include reduced tax rates and other measures. For example, as a high technology company operating in an approved economic-technological development area, YDME Beijing is entitled to an EIT rate of 15%, compared to an EIT rate of 33% applicable to most PRC companies. This classification also qualified YDME Beijing to a 50% reduction in EIT rate to 7.5% during the three calendar years ended December 31, 2002, 2003 and 2004. In addition, as a “foreign-invested advanced technology enterprise” certified by the Beijing Commerce Bureau, YDME Beijing is entitled to a reduced EIT rate of 10% for the three calendar years ending December 31, 2007. The reduced EIT rate will expire on December 31, 2007, after which the EIT rate of YDME Beijing will return to 15%. For the three fiscal years ended March 31, 2004, 2005 and 2006, YDME Beijing incurred income tax expense of RMB4.9 million, RMB11.9 million, and RMB18.1 million (US$2.3 million), respectively. If YDME Beijing had not had a reduced EIT rate of 7.5% for the two calendar years ended December 31, 2003 and 2004 and 10% for the calendar year ended December 31, 2005, it would have had an EIT rate of 15%, and our income tax expense for the three fiscal years ended March 31, 2004, 2005 and 2006 would have been RMB9.5 million, RMB17.6 million and RMB31.1 million (US$3.9 million), respectively.
20
Table of Contents
Additionally, any adverse changes to the PRC’s Corporate Income Tax Law may adversely affect the taxation of YDME Beijing and/or remittances by YDME Beijing to us. To date, the PRC government has not disclosed any details as to the changes in tax law slated for consideration. However, it is believed that among the possible changes are elimination of tax holidays and other incentives, increases in tax rates and imposition of a dividend withholding tax. Historically, we have benefited from tax holidays and incentives. The extent and timing of any such changes in the PRC’s tax laws is uncertain and it is not known whether any transitional or other relief will be granted to companies such as YDME Beijing that have already established operations in China. If adopted, these changes could significantly increase our tax expense.
YDME Beijing is also entitled to a refund of VAT on the sales of self-developed software embedded in our HIFU therapy system, which we recognize as part of our net revenues under U.S. GAAP. In the fiscal year ended March 31, 2006, VAT refunds accounted for 6.2% of our revenues.
Any increase of YDME Beijing’s EIT rate, changes in the PRC tax laws or regulations or any reduction or cancellation of the VAT refund in the future could have a material adverse effect on our financial condition and results of operations.
Recent PRC regulations relating to acquisitions of PRC companies by foreign entities may limit our ability to acquire PRC companies and adversely affect the implementation of our strategy as well as our business and prospects.
In 2005, the SAFE issued a number of rules regarding offshore investments by PRC residents. The currently effective rule, known as SAFE Notice 75, was issued in October 2005. It requires PRC residents to register with and receive approvals from the SAFE in connection with certain offshore investment activities. Since we are a Cayman Islands company that is controlled by Chengxuan, the controlling shareholder of which is Mr. Xiaodong Wu, our Chairman and a PRC resident, we are affected by the registration requirements imposed by SAFE Notice 75. YDME Beijing is also affected by SAFE Notice 75. Any failure by Mr. Wu or Chengxuan to comply with SAFE Notice 75, or change in SAFE policy and regulations in respect of Notice 75, could adversely affect us in a variety of ways.
SAFE Notice 75 provides, among other things, that prior to establishing or assuming control of an offshore company for the purpose of transferring to that offshore company assets of, or equity interests in, an enterprise in the PRC, each PRC resident (whether a natural or legal person) who is an ultimate controller of the offshore company must complete prescribed registration procedures with the relevant local branch of the SAFE. Such PRC resident must amend his or her SAFE registration under certain circumstances, including upon any further transfer of equity interests in, or assets of, an onshore enterprise to the offshore company as well as any material change in the capital of the offshore company, including by way of a transfer or swap of shares, a merger or division, a long-term equity or debt investment or the creation of any security interests in favor of third parties.
SAFE Notice 75 applies retroactively and to indirect shareholdings. PRC residents who have established or acquired direct or indirect control of offshore companies that have made onshore investments in the PRC in the past are required to complete the registration procedures by March 31, 2006.
The failure or inability of a PRC resident shareholder to receive any required approvals or make any required registrations could subject the PRC subsidiary to fines and legal sanctions, restrict the offshore company’s additional investments in the PRC subsidiary, or limit the PRC subsidiary’s ability to make distributions or pay dividends offshore.
Due in part to the lack of implementing rules and uncertainties relating to the interpretation and implementation of SAFE Notice 75, its effect on companies such as ours is difficult to predict.
Mr. Xiaodong Wu, who is subject to the above registration requirements, has made the required SAFE registration with respect to his investment in Chengxuan and our company. He has submited prior to the
21
Table of Contents
March 31, 2006 deadline the application for the required amendment to his SAFE registration in connection with our August 2005 initial public offering. Similarly, Mr. Wu has also submitted, within 30 days of the follow-on offering, an application for additional amendment to his SAFE registration in connection with the follow-on offering.
Further, we have established two entities in the British Virgin Islands, CMED Technologies Ltd., on January 9, 2006 and CMED Diagnostics Ltd. on January 11, 2006. The SAFE may interpret SAFE Notice 75 to require that Mr. Wu should submit a SAFE registration with respect to the establishment of these two entities within 30 days of the establishment of the respective entity. Mr. Wu has not submitted such registration, and he has advised us that he does not intend to submit such SAFE registration with respect to the two entities.
If Mr. Wu, whose actions we do not control, fails to comply with the registration procedures set forth in SAFE Notice 75, or if the SAFE determines that Mr. Wu should have previously made filings with respect to the offshore subsidiaries, YDME Beijing could be subject to fines and legal penalties, or the SAFE could impose restrictions on YDME Beijing’s foreign exchange activities, including the payment of dividends and other distributions to us or our affiliates and YDME Beijing’s ability to receive capital from us. Any of these actions could, among other things, materially and adversely affect our business operations, acquisition opportunities and financing alternatives.
We face risks related to health epidemics and outbreaks of contagious diseases, including avian influenza and Severe Acute Respiratory Syndrome, or SARS.
Our business could be adversely affected by the effects of avian influenza, SARS or other epidemics or outbreaks of contagious diseases. There have been recent reports of outbreaks of a highly pathogenic avian influenza, or avian flu, caused by the H5N1 virus in certain regions of Asia and Europe. In 2005 and 2006, there have been reports on the occurences of avian flu in various parts of China, including a few confirmed human cases. An outbreak of avian flu in the human population could result in a widespread health crisis that could adversely affect the economies and financial markets of many countries, particularly in Asia. Additionally, a recurrence of SARS, a highly contagious form of atypical pneumonia, similar to the occurrence in 2003 which affected China, Hong Kong, Taiwan, Singapore, Vietnam and certain other countries, would also have similar adverse effects. The 2003 SARS outbreak significantly and adversely affected our revenues and, consequently, our profitability. Since all of our operations and substantially all of our customers and suppliers are based in Asia, an outbreak of avian flu, SARS or other contagious diseases in China, other places in Asia or elsewhere, or the perception that such outbreak could occur, and the measures taken by the governments of countries affected, would adversely affect our business, financial condition or results of operations. We have not adopted any written preventive measures or contingency plans to combat any future outbreaks of avian flu, SARS or any other epidemics.
Risks Related to Our Ordinary Shares and ADSs
The market price for our ADSs may be volatile.
The market price for our ADSs has been and may continue to be subject to significant fluctuations. Since August 10, 2005, the intraday sales prices of our ADSs on the Nasdaq National Market have ranged from US$14.95 to US$44.93 per ADS, and the last reported sale price on September 27, 2006 was US$21.95 per ADS. The price of our ADSs may fluctuate in response to factors including the following:
| • | announcements of technological or competitive developments; |
| • | announcements regarding patent litigation or the issuance of patents to us or our competitors; |
| • | governmental developments in China, such as changes in fiscal policies or developments relating to the regulatory or health care reimbursement environment, affecting us or our competitors; |
| • | announcements of studies and reports relating to the effectiveness or safety of our products or those of our competitors; |
22
Table of Contents
| • | actual or anticipated fluctuations in our quarterly operating results and changes or revisions of our expected results; |
| • | changes in financial estimates by securities research analysts; |
| • | changes in the economic performance or market valuations of other medical technology companies; |
| • | addition or departure of our senior management and key research and development personnel; |
| • | any litigation, governmental investigation or enforcement proceedings brought against us by authorities and industry regulators in China or elsewhere; |
| • | release or expiry of lock-up or other transfer restrictions on our outstanding ordinary shares or ADSs; and |
| • | sales of additional ordinary shares or ADSs, or the perception that such sales might occur. |
In addition, the securities market has from time to time experienced significant price and volume fluctuations that are not related to the operating performance of particular companies. These market fluctuations may also have a material adverse effect on the market price of our ADSs.
Substantial future sales of our ADSs in the public market, or the perception that such sales might occur, could cause the price of our ADSs to decline.
Sales of our ADSs or ordinary shares in the public market, or the perception that these sales could occur, could cause the market price of our ADSs to decline. We currently have 273,600,001 ordinary shares outstanding, including 155,100,001 ordinary shares represented by 15,510,000 ADSs. All ADSs sold in our initial public offering in August 2005 and our follow on offering in March 2006 are freely transferable without restriction or additional registration under the U.S. Securities Act of 1933, as amended, or the Securities Act, except to the extent acquired by persons deemed to be our “affiliates.”
In addition, Golden Meditech, GE, Green Wall, Asset Managers and Neon Liberty have the right to cause us to register under the Securities Act the sale of an aggregate of 43,080,001 ordinary shares owned by them. Registration of these shares under the Securities Act would result in these shares becoming freely tradable without restriction under the Securities Act immediately upon the effectiveness of the registration. Sales of these registered shares in the public market could cause the price of our ADSs to decline.
Our articles of association contain anti-takeover provisions that could adversely affect the rights of holders of our ordinary shares and ADSs.
Our articles of association contain provisions that could limit the ability of others to acquire control of our company or cause us to engage in change-of-control transactions. These provisions could have the effect of depriving our shareholders of an opportunity to sell their shares at a premium over prevailing market prices by discouraging third parties from seeking to obtain control of our company in a tender offer or similar transaction. For example, our board of directors has the authority, without further action by our shareholders, to issue preferred shares in one or more series and to fix their designations, powers, preferences, privileges, and relative participating, optional or special rights and the qualifications, limitations or restrictions, including dividend rights, conversion rights, voting rights, terms of redemption and liquidation preferences, any or all of which may be greater than the rights associated with our ordinary shares, in the form of ADS or otherwise. Preferred shares could be issued quickly with terms calculated to delay or prevent a change in control of our company or make removal of management more difficult. If our board of directors issues preferred shares, the price of our ADSs may fall and the voting and other rights of the holders of our ordinary shares and ADSs may be adversely affected.
Holders of ADSs have fewer rights than shareholders and must act through the depositary to exercise those rights.
Holders of ADSs do not have the same rights as holders of our ordinary shares and may only exercise the voting rights with respect to the underlying ordinary shares in accordance with the provisions of the deposit
23
Table of Contents
agreement. Under the deposit agreement, if the vote is by show of hands, the depositary will vote the deposited securities in accordance with the voting instructions received from a majority of holders of ADSs that provided timely voting instructions. If the vote is by poll, the depositary will vote the deposited securities in accordance with the voting instructions it timely receives from ADS holders. In the event of poll voting, deposited securities for which no instructions are received will not be voted. Under our articles of association, the minimum notice period required to convene a general meeting is seven days. When a general meeting is convened, you may not receive sufficient notice of a shareholders’ meeting to permit you to withdraw your ordinary shares to allow you to cast your vote with respect to any specific matter. In addition, the depositary and its agents may not be able to send voting instructions to you or carry out your voting instructions in a timely manner. We will make all reasonable efforts to cause the depositary to extend voting rights to you in a timely manner, but we cannot assure you that you will receive the voting materials in time to ensure that you can instruct the depositary to vote your shares. Furthermore, the depositary and its agents will not be responsible for any failure to carry out any instructions to vote, for the manner in which any vote is cast or for the effect of any such vote. As a result, you may not be able to exercise your right to vote and you may lack recourse if your ordinary shares are not voted as you requested. In addition, in your capacity as an ADS holder, you will not be able to call a shareholder meeting.
You may be subject to limitations on transfers of your ADSs.
Your ADSs are transferable on the books of the depositary. However, the depositary may close its transfer books at any time or from time to time when it deems expedient in connection with the performance of its duties. In addition, the depositary may refuse to deliver, transfer or register transfers of ADSs generally when our books or the books of the depositary are closed, or at any time if we or the depositary deem it advisable to do so because of any requirement of law or of any government or governmental body, or under any provision of the deposit agreement, or for any other reason.
Your right to participate in any future rights offerings may be limited, which may cause dilution to your holdings, and you may not receive cash dividends if it is impractical to make them available to you.
We may from time to time distribute rights to our shareholders, including rights to acquire our securities. However, we cannot make rights available to you in the United States unless we register the rights and the securities to which the rights relate under the Securities Act or an exemption from the registration requirements is available. Also, under the deposit agreement, the depositary will not make rights available to you unless the distribution to ADS holders of both the rights and any related securities are either registered under the Securities Act, or exempted from registration under the Securities Act. We are under no obligation to file a registration statement with respect to any such rights or securities or to endeavor to cause such a registration statement to be declared effective. Moreover, we may not be able to establish an exemption from registration under the Securities Act. Accordingly, you may be unable to participate in our rights offerings and may experience dilution in your holdings.
In addition, the depositary of our ADSs has agreed to pay to you the cash dividends or other distributions it or the custodian receives on our ordinary shares or other deposited securities after deducting its fees and expenses. You will receive these distributions in proportion to the number of ordinary shares your ADSs represent. However, the depositary may, at its discretion, decide that it is inequitable or impractical to make a distribution available to any holders of ADSs. For example, the depositary may determine that it is not practicable to distribute certain property through the mail, or that the value of certain distributions may be less than the cost of mailing them. In these cases, the depositary may decide not to distribute such property, in which event you would not receive such distribution.
We are a Cayman Islands company, and because the rights of shareholders under Cayman Islands law differ from those under U.S. law, you may have difficulty protecting your shareholder rights.
We are an exempted company with limited liability incorporated under Cayman Islands law, and substantially all of our assets are located outside the United States. In addition, a majority of our directors and
24
Table of Contents
executive officers reside outside the United States, and a substantial portion of their assets is located outside of the United States. As a result, it may be difficult for you to effect service of process within the United States upon our directors or executive officers, or enforce judgments obtained in the United States courts against our directors or executive officers.
Our corporate affairs are governed by our articles of association, the Companies Law (2004 Revision) and the common law of the Cayman Islands. The rights of shareholders to take legal action against our directors and us, actions by minority shareholders and the fiduciary responsibilities of our directors to us under Cayman Islands law are to a large extent governed by the common law of the Cayman Islands. The common law of the Cayman Islands is derived in part from comparatively limited judicial precedent in the Cayman Islands as well as from English common law, which has persuasive, but not binding, authority on a court in the Cayman Islands. The rights of our shareholders and the fiduciary responsibilities of our directors under Cayman Islands law are not as clearly established as they would be under statutes or judicial precedents in the United States. In particular, the Cayman Islands has a less developed body of securities laws as compared to the United States and some states, such as Delaware, have more fully developed and judicially interpreted bodies of corporate law. In addition, shareholders in Cayman Islands companies may not have standing to initiate a shareholder derivative action before the federal courts of the United States and will have no general right under Cayman Islands law to inspect or obtain copies of our list of shareholders or our corporate records.
There is uncertainty regarding whether Cayman Islands courts would:
| • | recognize or enforce against us judgments of courts of the United States based on certain civil liability provisions of U.S. securities laws; and |
| • | impose liability against us, in original actions brought in the Cayman Islands, based on certain civil liability provisions of U.S. securities laws. |
There is no statutory recognition in the Cayman Islands of judgments obtained in the United States, although the courts of the Cayman Islands will generally recognize and enforce a non-penal judgment of a foreign court of competent jurisdiction of a fixed sum that is not contrary to natural justice or the public policy of the Cayman Islands without retrial on the merit.
As a result of all of the above, our public shareholders may have more difficulty in protecting their interests with respect to actions taken by our management, directors or major shareholders than they would as public shareholders of a U.S. company.
We may be considered a passive foreign investment company, which could result in adverse U.S. tax consequences for U.S. investors.
Based upon the composition of our income and valuation of our assets, including goodwill, we may be classified as a passive foreign investment company, or PFIC, for U.S. federal income tax purposes. Such characterization could result in adverse U.S. tax consequences to you if you are a U.S. investor. For example, if we are a PFIC, our U.S. investors may become subject to increased tax liabilities under U.S. tax laws and regulations and will become subject to burdensome reporting requirements. The determination of whether or not we are a PFIC is made on an annual basis and will depend on the composition of our income and assets from time to time. Specifically, for any taxable year we will be classified as a PFIC for U.S. tax purposes if either (i) 75% or more of our gross income in that taxable year is passive income or (ii) at least 50% of the value of our assets (based on an average of the quarterly values of our assets during a taxable year) is attributable to assets which produce or are held for the production of passive income. For this purpose, cash is a passive asset. The calculation of the value of our assets will be based, in part, on the quarterly market value of our ADSs and ordinary shares, which is subject to change. See “Taxation—United States Federal Income Taxation—Passive Foreign Investment Company.”
25
Table of Contents
ITEM 4. INFORMATIONONTHE COMPANY
A. History and Development of the Company
Our predecessor company, Beijing Yuande Bio-Medical Engineering Co. Ltd., or YDME Beijing, was incorporated in China and commenced business operations in July 1999. In anticipation of our initial public offering, we incorporated China Medical Technologies Inc., or China Medical, in the Cayman Islands as a listing vehicle in July 2004. China Medical is an exempted company with limited liability incorporated under Cayman Islands law. China Medical became our ultimate holding company when it issued shares to the existing shareholders of YDME Beijing in January 2005 in exchange for all of the shares that these shareholders held in YDME Beijing. We conduct substantially all of our operations through YDME Beijing.
Our principal executive offices are located at No. 24 Yong Chang North Road, Beijing Economic-Technological Development Area, Beijing 100176, People’s Republic of China. Our telephone number at this address is (8610) 6787-1166 and our fax number is (8610) 6788-9588.
Investor inquiries should be directed to us at the address and telephone number of our principal executive offices set forth above. Our website iswww.chinameditech.com.The information contained on our website is not part of this annual report on Form 20-F. Our agent for service of process in the United States is Corporation Service Company located at 1133 Avenue of the Americas, Suite 3100, New York, New York 10036.
B. Business Overview
Overview
We are a China-based medical device company that develops, manufactures and markets in China products using HIFU for the treatment of solid cancers and benign tumors. We believe that we are a leader in the design and development of HIFU tumor therapy devices in China. In addition, we believe that we are the first China-based company to offer an integrated luminescence immunoassay IVD system using ECLIA technology to detect and monitor various diseases and disorders through laboratory evaluation and analysis of blood, urine or other body fluids.
Our primary product, the HIFU therapy system, is an ultrasound-guided ablation system that is used for the non-invasive treatment of solid tumors. Our system is designed to destroy tumors by using HIFU waves to instantly raise the temperature of the targeted tumor tissue to between 60° C and 70° C (140° F and 158° F). Our proprietary designs enable our system to treat a wide range of tumors, many of which cannot be treated with other HIFU tumor therapy devices currently available in the market. Our HIFU therapy system is SFDA-approved to treat liver, breast and kidney tumors, solid tumors in the pelvic cavity or on bone and tumors in the four limbs or superficial tissues. Treatments using our system are performed without anesthesia and do not cause significant discomfort, skin-burn or hemorrhage. We acquired our HIFU technology in November 1999 and as of March 31, 2006, have sold over 200 units of our HIFU therapy system to date.
Our second product, the ECLIA system, is a luminescence immunoassay IVD system based on the ECLIA technology that we acquired in August 2004. Luminescence immunoassay IVD systems are advanced types of IVD systems commonly used in the United States and Western Europe, but are relatively new in China. We began marketing and selling our ECLIA system in September 2004 and had sold over 1,400 units of our ECLIA analyzer as of March 31, 2006. We currently offer 56 types of reagent kits for use with our ECLIA analyzer. These reagents are used to detect various thyroid disorders, diabetes, hepatitis, disorders related to reproduction and growth and various types of tumors, as well as to assess the effect of digoxin, a widely used drug for the treatment of heart failure. In addition, we were one of the first companies to obtain SFDA approval in China for an ECLIA reagent kit for the early detection of SARS.
Historically, we have benefited from a strong relationship with PUPH, a leading medical research institution and hospital in China affiliated with Peking University. PUPH was the initial developer of our HIFU and ECLIA technologies. We believe that our acquisition of the HIFU and ECLIA technologies and the development and marketing of products using such technologies demonstrate our ability to successfully identify and commercialize medical technologies with significant market potential.
26
Table of Contents
We sell our HIFU therapy systems primarily to distributors with a small percentage of additional sales made directly to hospitals. We sell our ECLIA systems solely to distributors.
Our Competitive Strengths
We believe that our principle competitive strengths include the following:
HIFU technology leader for tumor treatment
We believe that we are a leader in the design and development of HIFU tumor therapy devices in China. Our patented HIFU therapy system is SFDA-approved and has been used to treat more tumor patients in China than other available HIFU tumor therapy devices. As of March 31, 2006, we have sold over 200 units of our HIFU therapy system. Treatments using our system are performed without anesthesia and do not cause significant discomfort, skin-burn or hemorrhage. In addition, we are developing a non-invasive ultrasound technology for measuring the temperature of the tumor tissues being treated. We have incorporated this technology into a prototype for our third-generation HIFU therapy system, which is currently being tested in a leading hospital in China. We also intend to sell a non-invasive ultrasound-based heat measuring device separately as a product upgrade for existing users of our HIFU therapy system. We believe that we are the first company to develop this technology.
Innovative ECLIA system for in-vitro diagnostics
We believe that we were the first China-based company to offer an integrated luminescence immunoassay IVD system that does not require devices or reagents provided by outside sources. Our ECLIA system is a simple-to-use IVD platform and is sensitive, accurate and safe. Our ECLIA analyzer incorporates proprietary technologies that give our system several advantages over radioimmunoassay and enzymatic immunoassay IVD systems, the two most widely used IVD systems in China. Additionally, compared with the imported automatic luminescence immunoassay systems available in China, our ECLIA system, including our reagent kits, are priced significantly lower, but can achieve test results with comparable accuracy. We believe we have a significant market opportunity to market our ECLIA system to small- and medium-sized hospitals in China. Many of these hospitals currently use less advanced IVD systems because they lack sufficient financial means to purchase the more expensive automatic and imported luminescence immunoassay systems or do not have sufficient patient volume. We believe that we can introduce a fully automatic system for large hospitals at a lower price than our overseas-based competitors can achieve.
Strong research and development capability
We have strong research and development capabilities, and we plan to continue to focus our development efforts on new applications for or enhancements to our HIFU and ECLIA technologies. Many of the key research and development personnel who developed the HIFU and ECLIA technologies at the PUPH are currently employed by us. We believe that we have a strong patent portfolio compared to other medical technology companies based in China. In China, we currently have 14 patents directed towards our HIFU therapy system and ECLIA system, and have 19 patent applications pending in China directed towards our HIFU therapy and ECLIA systems. We have also made applications for four patents directed towards our HIFU technology in each of the following countries outside China: the United States, the United Kingdom, Germany, Japan, South Korea, Saudi Arabia and India. As of September 27, 2006, we have been granted three out of the four patents we have applied for in the United Kingdom. We have developed multiple generations of our HIFU therapy system. The two-transducer design of our second-generation HIFU therapy system enables the patient to be treated in the supine, side or seated positions so that more locations in the body can be targeted. In addition, our third-generation HIFU therapy system will be equipped with our recently developed non-invasive ultrasound temperature measuring technology. We have established relationships with a number of major hospitals and universities in China, as many of these hospitals and universities use our HIFU therapy system to treat patients and to conduct research. We believe that these relationships raise our profile, enhance awareness of our products in the medical community and among patients and provide us with valuable clinical data to improve our products and keep us abreast of industry trends and developments.
27
Table of Contents
Low manufacturing and operating costs
We have significant cost advantages that allow us to price our products competitively and to maintain favorable margins. We believe that our location in China allows us to maintain our cost of operations at a competitive level that could potentially deter other overseas-based competitors from entering the China market. This is due primarily to the lower wages and costs of components and raw materials in China relative to other locations. In addition, our sales and distribution costs are low because we market and sell our products mainly to distributors. Also, our research and development effort mainly focuses on developing new applications and improving the product designs of our HIFU therapy system and ECLIA system, which generally requires lower research and development expense than the development of new technologies and products.
Our Products and Services
Our HIFU Solution
Treatment methods for cancer
The conventional treatment methods for cancer in China and elsewhere are generally surgery, radiotherapy and chemotherapy. High intensity focused ultrasound, radio frequency ablation, microwave thermo-coagulation and cryosurgery are some of the major new cancer treatment methods developed and commercialized in recent years. Biotherapy and gene therapy are currently still in experimental stages and are not currently available to patients generally. The selection of a particular treatment method for a patient depends on various factors, including the tumor’s receptivity to the treatment method, the location of the tumor, the stage of the tumor and the patient’s state of health. Cancer patients are usually treated by a combination of various treatment methods.
Surgery. Surgery is the most commonly used treatment method for most types of cancers and other tumors. Patients who undergo surgery face risks associated with anesthesia and complications, such as hemorrhages, infections or weakening of organ functions. In addition, the recurrence of cancer tumors is common and repeat surgery can involve high risks and significant complications.
Radiotherapy. Radiotherapy uses radiation or radioactive implants to destroy or inhibit the growth of tumor tissues. Radiotherapy is often used together as a complementary treatment method to surgery. Although radiotherapy is used to treat a wide range of cancer types, different types of tumor tissues have different sensitivities to radiation, and different patients have different levels of tolerance to radiation. Serious side effects, including skin ulceration or hemorrhage can result from excessive doses of radiation.
Chemotherapy. Chemotherapy uses drugs to destroy or inhibit the growth of tumor tissues and is commonly used as a supplemental treatment method together with surgery. Chemotherapy is relatively more effective than other cancer treatment methods for lymph tumors or metastatic tumors. However, chemotherapy destroys not only tumor tissues but also normal healthy tissues, resulting in serious side effects for the patient, including loss of appetite, nausea and vomiting, hair loss, as well as damage to the liver, bone marrow and immune functions.
Other minimally invasive treatments and targeted therapeutics. There are several relatively new treatment methods that have shown some clinical success in treating cancer. For example, radio frequency ablation, microwave thermo-coagulation and cryosurgery are minimally invasive treatments that attempt to destroy the tumor by either heating or freezing the targeted tissue with a needle, laser or cryogenic device that is inserted into the tumor. In addition, there is a new class of cancer therapeutics that have been developed by large pharmaceutical and biotechnology companies, such as Avastin®, Tarceva® and ERBITUX®, that are specifically designed to impede tumor development, while having little to no effect on healthy cells. The clinical applications of these treatments and targeted therapeutics are limited to certain types of cancers, and their long-term effectiveness is still being evaluated.
Therapeutic principles of HIFU
Heat therapy, also known as hyperthermia or thermotherapy, is a well-established method of medical treatment. It is generally accepted by the scientific community that the irreversible destruction of tissues,
28
Table of Contents
medically known as coagulation necrosis, occurs when tissues are exposed to temperatures above a certain level for a certain period of time. The maximum temperature under which tumor tissues can survive is 43º C (109.4º F), while normal tissues can tolerate a temperature of up to 45º C (113º F). The higher the temperature, the shorter the time needed to destroy tumor tissues. When the temperature reaches 60º C (140º F) or higher, coagulation necrosis will occur within one second.
In focused ultrasound ablation, a transducer directs ultrasound waves to converge on a small area of tissue inside the body, resulting in very high ultrasound intensity at this point. The effect of this process is similar to that of using a magnifying glass to focus the sun’s energy into a single spot. The ultrasound energy causes the temperature of the tumor tissues at the focal point to increase within a very short period of time to a level high enough to destroy the targeted tumor tissues. An important feature of this technology is that only a limited temperature increase occurs in the body tissue surrounding the focal point.
HIFU therapy can be used in conjunction with other treatment methods such as surgery, radiotherapy and chemotherapy, or as a stand-alone treatment. HIFU treatment increases the sensitivity and receptivity of body tissues to radiotherapy and chemotherapy. We believe that, because it is a relatively new procedure, HIFU therapy to date has typically been used for patients not eligible for one of the conventional therapies or when these other therapies have failed. When used in combination with conventional treatment methods, HIFU treatment often enhances their effectiveness and reduces the required chemical or radiation dosage. As a result, patients generally experience less side effects associated with radiotherapy and chemotherapy.
Our HIFU therapy system is SFDA-approved and has been used for the treatment of a wide range of tumors in China since November 1999. In addition, other companies have also developed HIFU technologies, and a few HIFU medical devices have been approved for the treatment of limited types of tumors, mostly prostate cancer and uterine fibroids, in the European Union, Canada, Russia, South Korea, Japan and the United States, and a larger number of HIFU tumor therapy devices have been approved for clinical studies and trials in these regions and elsewhere. See “—Competition.”
Benefits and limitations of our HIFU solution
Our HIFU therapy system can be used as a supplemental or stand-alone treatment method with surgery, radiotherapy and chemotherapy for the treatment of many types of solid tumors. We believe that the benefits of our HIFU solution include:
Non-invasive procedure. Our HIFU therapy system offers patients a non-invasive treatment alternative. Treatments performed with our HIFU therapy system do not require incisions and transfusions and as a result, minimize the risk and complications associated with invasive procedures. In addition, it is often difficult for patients who receive traditional treatments to later undergo the same treatment in the event of a relapse due to the invasive nature of surgery and the significant side effects associated with chemotherapy and radiotherapy. Patients can undergo repeated treatments for cancer using our system with minimal side effects.
Minimal side effects. Treatments performed with our HIFU therapy system have minimal side effects and a relatively low level of complications. Compared to other HIFU tumor therapy systems that are commercially available in China, our HIFU therapy system produces less intense ultrasound waves at the point of skin penetration, which significantly reduces the likelihood of skin burns and collateral damage to healthy body tissue.
High degree of safety. Treatments using our HIFU therapy system can be performed without anesthesia and do not cause significant discomfort, skin-burn or hemorrhage. However, improper operation and use of our HIFU therapy system could cause skin burn.
29
Table of Contents
Treatment for broad ranges of tumors. Our patented two-transducer system enables our HIFU therapy system to target tumors in more body locations than the other HIFU tumor therapy systems currently commercially available in China. Our HIFU therapy system is approved by the SFDA to treat liver, breast and kidney tumors, solid tumors in the pelvic cavity or on bone and tumors in the four limbs or superficial tissues. Based on published clinical studies conducted by researchers and practitioners of hospitals in China, we believe that hospitals have used our HIFU therapy system to treat many types of tumors, including the following:
• liver tumors • kidney tumors • breast tumors | • solid tumors in the pelvic cavity including: • bladder tumors • uterine tumors • uterine fibroids • prostate tumors • prostate hypertrophyrectal tumors | • tumors in the four limbs or superficial tissues • solid tumors on bone • pancreatic tumors • metastasis tumors of the abdominal lymph node • rear abdominal tumors |
We believe as our HIFU therapy system gains higher acceptance from the medical community and among patients, it could be used more frequently as a stand-alone treatment due to its low risk profile.
Cost-effective for hospitals. We believe our HIFU therapy system is a more cost-effective option for hospitals for the types of tumors it can treat compared to other currently commercially available cancer treatment methods in China. Chemotherapy requires expensive drugs while surgery requires the use of anesthesia and other medical supplies. Radiotherapy requires the use of significantly more expensive equipment than our HIFU therapy system. In addition, we believe our HIFU system can treat a broader range of tumors than other HIFU tumor therapy systems available in China.
However, the use of our HIFU solution is subject to a number of limitations. The clinical use of our HIFU therapy system has been limited mostly to patients with cancer at advanced stages or who are not eligible for conventional treatment. In addition, HIFU therapy is suitable for the treatment of a single tumor of an identifiable size or a group of tumors that are located in very close proximity. It is not suited for the treatment of a single tumor that is too small or tumors that are widespread. It is also not suitable to treat blood-borne cancers. Furthermore, as ultrasound cannot be transmitted in a straight line through solid bone or air, HIFU therapy is not suited for the treatment of tumors located in the head, neck, lung and tumors behind bones.
Clinical studies of our HIFU therapy system
To our knowledge, clinical studies that have used our HIFU therapy system have been reported in 15 published international and Chinese national level reports and peer-review journals. Some studies using our HIFU therapy system have been reported in multiple publications or presentations. These studies have reported approximately 1,520 cases for the treatment of rectal tumors, bladder tumors, colon tumors, gastric tumors, ampullary tumors, liver tumors, pancreatic tumors, metastasis tumors of the abdominal lymph nodes, prostate tumors, adrenal tumors, kidney tumors, smooth muscle tumors, endometrial tumors, ovarian carcinoma, rear abdominal tumors, lymphoma tumors, cervical tumors, breast tumors, hepatocellular tumors, non-Hodgkin’s lymphoma, neurofibroma, osteolytic sarcoma and skin squamous tissue cancers.
These clinical studies have been conducted by both physicians affiliated with us as consultants and other physicians in hospitals and universities in China, such as PUPH, Fudan University Zhongshan Hospital, Beijing University Medical School, the Second- Affiliated Hospitals of Sun Yat-Sen University, the First Hospital of Jilin University, Taian Central Hospitals, Qilu Hospital of Shandong University, the Central Railway Hospital of Liuzhou, and the First Hospital of Jiangxi Medical University. In addition, we plan to participate in future long-term studies of the effectiveness of our HIFU therapy system.
30
Table of Contents
The HIFU therapy system
We currently offer our second-generation two-transducer HIFU therapy system. We are currently testing a prototype of our third-generation product, a two-transducer HIFU therapy system equipped with our newly-developed non-invasive ultrasound temperature measuring technology.
Our HIFU therapy system consists of the following main components:
Integrated transducers. The integrated transducer consists of HIFU transmitting sources and drive circuits and can move in six directions with millimeter precision. Our HIFU transmitting sources are patented, and the patent for the HIFU transmitting sources expires in March 2019. The HIFU transmitting sources generate high-frequency ultrasound waves, which converge from a wide angle to focus on a designated focal point inside the body. We have obtained a patent for the wide convergence angle design in China, and the patent for this design expires in November 2021. Our second-generation HIFU therapy system is equipped with two transducers, which enable the patient to be treated in the supine, side or seated positions so that more locations in the body can be targeted and more tumors can be treated. This patented two-transducer design also increases the patient’s comfort level during treatment, and the patent for this design expires in November 2021.
Ultrasound imaging system. The ultrasound imaging system is an ultrasonic system that creates a computer image of the tumor that guides the physician in monitoring and targeting the ultrasound waves. The probe that captures the image of the tumor can monitor the patient from many angles and can be controlled automatically by a computer or manually by the physician.
Patient treatment table and seat. The table and seat frames are adjustable and can be rotated to facilitate treatment of tumors located in different parts of the body. The middle section of the patient treatment table can be removed to install a treatment seat so that the patient can sit while undergoing treatment for tumors in the pelvic cavity. The patient’s position depends on the location of the tumor. We have obtained a patent for the design of the table and seat frames in China, and this patent expires in November 2021. We have also applied for patents in other countries for the treatment seat design.
Control station. The control station consists of our proprietary software program, which controls the targeting and duration of the treatment, a computer, a monitor and peripheral equipment. The control station allows the operator to select automatic or manual treatment, as well as the duration of each ultrasound emission and other treatment parameters. If the operator inputs an unsafe ultrasound shot duration or treatment parameter, a built-in safety mechanism causes the HIFU therapy system to cease its operation immediately.
Conduct structure. Ultrasound energy does not transmit in a straight line through air. To introduce linear ultrasound waves into a patient’s body from the upper transducer, our HIFU therapy system contains a conduct structure, which consists of a semi-sphere container filled with degassed water. We have also developed a proprietary vacuum degassing device that converts large volumes of tap water into degassed water. We have obtained a patent for this device in China, and this patent expires in March 2009. We have also applied for patents for this semi-sphere container and the degassing device in other countries.
The treatment procedure
A physician and an ultrasound technician first locate the tumor tissues and define the area to be treated by using the ultrasound imaging system. The computer uses the physician’s input to plan the best way to target and ablate the tumor. Each individual high-energy focused ultrasonic shot ablates an oval-shaped targeted zone measuring up to ten millimeters in height by five millimeters in diameter. During the treatment, the transducer and the ultrasound imaging system automatically moves and ablates the targeted zone until the entire defined area has been treated. The shooting times can be selected between 0.1 and one second. The intervals between shots can also be selected from 0.1 to two seconds, depending on the depth of the tumor. Intervals between shots reduce the ultrasound volume passing through the skin over a period of time to avoid skin-burn. The individual targeted zone produces almost instantaneous coagulative necrosis of tissues due to an increase in temperature to between 60º C and 70º C (140º F and 158º F).
31
Table of Contents
The treatment duration and the number of treatment sessions vary according to the size of the tumor. For example, for a single four centimeter tumor, four to five sessions are usually performed on different days, with each treatment session lasting approximately half an hour.
Our ECLIA System
In-vitro diagnostics
IVD refers to laboratory tests that are performed on samples of blood, urine, saliva or other body fluids for the diagnosis and analysis of various diseases and disorders. Luminescence immunoassay IVD systems, including enhanced chemiluminescence immunoassay IVD systems such as our ECLIA system, are advanced IVD systems that are commonly used in the United States and Western Europe, but are relatively new in China. Luminescence immunoassay IVD systems are generally more sensitive, more accurate and safer to use. Compared to radioimmunoassay diagnostics, luminescence immunoassay diagnostics involve no radioactive contamination risk, and the reagent kits have a longer shelf life.
The ECLIA system
Our ECLIA system consists of an ECLIA analyzer, reagent kits and a data analysis system.
The ECLIA analyzer is a sensitive, accurate, low cost and simple-to-use IVD device capable of detecting minute levels of light triggered by combining reagents with body fluid samples to produce diagnostic results. The key component of our ECLIA analyzer is the photon counter. We have developed several technologies that make our photon counter more sensitive and accurate. Reagents are chemically active substances that are formulated to create different reactions with blood or other body fluid samples depending on the extent to which a disease or condition is present or absent. The data analysis system uses our proprietary integrated circuit card, or IC Card, input technology to analyze and organize the information produced. The IC Card contains encryption codes that prevent users from conducting tests with reagents produced by other manufacturers.
To use our ECLIA system, our reagents are mixed with a sample of body fluid from the patient in a microplate provided in our reagent kit. The mixture reacts and emits light, which is measured by the ECLIA analyzer. The light signal is analyzed by the data analysis system. The result is compared with the reference ranges for the relevant disease in order to reach a diagnosis.
Our reagent kits
We currently offer 56 types of reagent kits for detecting and monitoring various types of diseases and disorders, including thyroid disorders, diabetes, hepatitis, disorders related to reproduction and growth, SARS and various types of tumors. We are continuing to develop reagents for other applications.
32
Table of Contents
The following table sets out the major clinical applications of our reagent kits:
Types of test | Numbers of | Description | ||
| Thyroid disorders | 5 | The five assays comprise the basic tests hospitals perform to evaluate the functions of a patient’s thyroid gland. We believe that for many hospitals, thyroid testing is a mainstay of their routine endocrine tests. | ||
| Reproduction-related disorders | 6 | Five of our assays comprise the main tests performed by hospitals for evaluating the functions of the female reproductive organs, including the diagnosis of hormonal imbalance of postmenopausal women. One of our assays is a test to evaluate the male reproductive function. | ||
| Diabetes | 4 | Diabetes is also a common endocrine disorder in China. Two of our assays tests for insulin and C-Peptide level, two common indications of diabetes. Our other two assays test for and monitor diabetes where insulin and C-Peptide level may not be a reliable indicator. | ||
| Tumors | 12 | Our assays for “tumor markers” can detect the following types of tumors: | ||
• Prostate • Lung • Breast • Liver • Stomach • Intestine • Colon • Pancreas • Kidney • Ovary | ||||
| Growth | 1 | Our assay is a test for the early diagnosis, identification and prognosis evaluation of problems relating to growth and development. | ||
| SARS | 1 | SARS is an atypical pneumonia of unknown etiology, and was recognized in February 2003 in China, Taiwan, Hong Kong, Singapore and certain other regions in the world. | ||
| Hepatitis B | 10 | The ten assays comprise the basic tests hospitals perform to test for hepatitis B. | ||
| Digoxin monitoring | 1 | Our assay tests for the effectiveness of digoxin, a widely used drug for treatment of heart failure. | ||
| Sterility | 6 | Our assays test for the sterility caused by the mal-function of immune system. | ||
| TOCHR | 10 | TOCHR is the abbreviation of five viruses that could course the fetal malformation if infected during pregnancy. Our assays test for the infection. | ||
Manufacturing
Our manufacturing strategy combines our internal design expertise and proprietary technology with strategic outsourcing. We produce our reagent kits and conduct the final product assembly, testing and packaging of our HIFU therapy system and ECLIA analyzer at our facility in Beijing, China. We outsource the production of a majority of our components to, and purchase raw materials from, third-party suppliers.
We design or configure many key components of our products and outsource their production to qualified manufacturers. Our suppliers undergo quality certification in accordance with the standards developed by us, and we conduct frequent quality audits of each of our suppliers’ manufacturing facilities. We believe we maintain components and raw materials at an adequate level to ensure a stable supply.
33
Table of Contents
The major components of our HIFU therapy system are integrated ultrasound transducers, an ultrasound imaging system, a treatment table/seat, a control station and a conduct structure. The ultrasound imaging system and the treatment bed, two of the major components of our HIFU therapy system, are manufactured by GE and a local supplier, Beijing Tianyuan Kehui, respectively, and we purchase other components from other local suppliers. The major components of our ECLIA analyzer are the photon-multiplier, motor drive and various types of other electrical and plastic parts that are generally readily available in sufficient quantities from our local suppliers. The raw materials of our reagent kits are antibodies and antigens for the production of coated microplate and conjugator, chemicals for the production of reagents, and plastics and packaging such as vacant microplate and bottles. We purchase a portion of our supplies of antibodies and antigens overseas and source the other raw materials from local suppliers.
We generally produce our products based on firm orders from our customers and anticipated additional orders that we are reasonably confident will be obtained. Lead times for raw materials and components vary and depend on the specific supplier and the availability and demand for the raw materials or components. We currently use one or more suppliers for some of our key components for quality control purposes. We source the ultrasound imaging system, one of the major components of our HIFU therapy system, from GE, which produces the ultrasound imaging system based on our specifications. If we are unable to obtain this component from GE, it may be difficult to find an alternate supplier on a timely basis that can provide a replacement system with comparable quality as we require. We believe that we will be able to source most of our other key components from other suppliers if necessary. We do not currently have any long-term supply contracts, and our purchases are made on a purchase order basis.
We have recently streamlined our product assembly process to improve our responsiveness to changes in our business volume by training our employees to assemble multiple components. As our sales fluctuate from month to month, employees with multiple skill sets and multi-tasked teams will allow us to make quicker deliveries while enabling us to more effectively manage our costs.
On November 1, 2005, we obtained a Certificate of Good Manufacturing Practices for Pharmaceutical Products, or the GMP certificate, for the production of IVD reagents. The GMP certificate was issued by the State Drug Administration of China and certifies that our ECLIA manufacturing facilities comply with the requirements of good manufacturing practices for pharmaceutical products. The GMP certificate is valid for a term of five years.
In addition, in 2003 we obtained the registration certificate from the SFDA required for us to sell our HIFU therapy system. This certificate expires in January 2007. We obtained the requisite registration certificates to sell our ECLIA analyzer in March 2005. This certificate expires in 2009.
In addition to the product registration certificates described above, we are required to obtain manufacturing permits and to renew these permits six months prior to the expiration of the original permit. Our manufacturing permit for medical devices, which covers our HIFU system, our ECLIA system and certain of our IVD reagents, expired on December 31, 2005. We renewed this permit in July 2005, and it will expire in 2010. Our manufacturing permit for pharmaceuticals, which covers certain of our IVD reagents, also expired on December 31, 2005. We renewed this permit in January 2006, and it will expire in 2010.
Quality Control
We have our own independent quality control system and devote significant attention to quality control for the designing, manufacturing and testing of our products. We have established a quality control system in accordance with SFDA regulations. In 2001, we obtained ISO 9001 certification for the production of our HIFU therapy system, which indicates our compliance with recognized international standards for quality control. We inspect components prior to assembly, and inspect and test internally manufactured products both during and after the manufacturing process.
34
Table of Contents
Our quality control team is also responsible for ensuring that we are in compliance with all applicable regulations, standards and internal policies. Our senior management team is actively involved in setting quality policies and managing internal and external quality performance.
Markets and Customers
We believe that both our HIFU therapy system and our ECLIA system have significant market potential in China as there is a strong demand for tumor treatment devices and sensitive and reliable IVD systems. The target market for our HIFU therapy system is large- and medium-sized hospitals in China with oncology departments. According to the Ministry of Health of the central government in China, there were approximately 6,100 large- and medium-sized hospitals in China in 2004. As of March 31, 2006, we have sold over 200 units of our HIFU therapy system. The target market for our ECLIA system is small- and medium-sized hospitals in China. According to the Ministry of Health, there were approximately 17,500 small- and medium-sized hospitals in China in 2004. We began marketing and selling our ECLIA system in September 2004 and have sold over 1,400 ECLIA analyzers as of March 31, 2006.
In the three fiscal years ended March 31, 2004, 2005 and 2006, 95.2%, 97.0% and 98.9% respectively, of our HIFU therapy system were sold to distributors. Sales of our HIFU therapy system have been, and will continue to be, generated by sales to a relatively small group of distributors that change from year to year. We do not have long-term contracts with any of our distributors. Because our main product, the HIFU therapy system, is sold at a relatively high unit price, the size and timing of individual orders can have a significant effect on our quarterly results. See “Item 3. Key Information—D. Risk factors—Risks Related to Our Company—We are dependent on a small number of distributors for a significant portion of our revenues and this dependence is likely to continue in the future.”
Sales and Marketing
We market and sell our HIFU therapy systems primarily through distributors, with a small percentage sold to hospitals directly through our own direct sales force. We recognize revenues at the time our products are accepted by either our distributors or the hospitals designated by these distributors, which typically occurs within one to two weeks after shipment. We sign binding purchase agreements with our distributors or with our hospital customers each time we make a sale of our products. Under these purchase agreements, our distributors or hospital customers agree to pay a non-refundable, non-adjustable fixed amount of money for our products over a period of time. Our distributors take ownership of our products when either they or the hospitals they designate accept the products, depending on the terms of each contract. The acceptance typically occurs within one or two weeks of shipment, during the period when we help hospitals to install our products. Once the products are accepted, the one-year warranty period commences. For sales of our HIFU therapy system, we require an initial payment of 30% to 40% of the sale price when customers place purchase orders or after the shipment is accepted by either our distributors or the hospitals designated by these distributors. The balance is payable within one year of acceptance. Some of our distributors purchase our products to operate profit-sharing programs with hospitals. Under these profit-sharing programs, these distributors install our HIFU therapy system in hospitals without any up-front charge to hospitals and in return receive a portion of the net profits generated by the system.
We market and sell our ECLIA system solely to distributors. For sales of our ECLIA analyzer, we require a payment of 20% to 40% of the sale price before delivery or after acceptance and the balance within one year of acceptance. For sales of reagent kits, we do not require down payments, and we collect payments three months after acceptance.
We select our distributors based on their reputation, market coverage, sales experience and the size of their sales force. We conduct credit assessments of each of our distributors or hospital customers before we enter into a purchase agreement. Currently, all our products are being marketed and sold in China. However, we are also evaluating the opportunities to expand distribution of our products to other countries, including Japan and South Korea, in the future. We do not have exclusive distribution arrangement with any of our distributors.
35
Table of Contents
As of June 30, 2006, we had a team of 50 marketing and sales personnel who conduct direct sales of our HIFU therapy system, manage distributors for both our HIFU therapy system and our ECLIA system and perform other sales related tasks. Of these 50 marketing and sales personnel, 33 focus on sales of our HIFU therapy system and 17 focus on sales of our ECLIA system. We intend to increase the size of our sales force as our business grows.
The hospital’s chief administrator and the head of the department of oncology, in the case of our HIFU therapy system, or the head of the department of diagnostics, in the case of our ECLIA system, typically make the purchasing decision for our products. We believe that hospitals make purchase decisions on HIFU tumor therapy devices based on the safety and effectiveness of the device, the types of cancer treatment methods that they already offer, their available budget and the anticipated economic return. We believe that hospitals make purchase decisions on diagnostics equipment based on the performance and the range of reagent kits available, the prices of equipment and reagents and ease of use.
We have established relationships with many hospital administrators at prominent hospitals and other leading medical institutions, many of whom we believe are advocates for our HIFU therapy system. We believe our relationships with these major hospitals and medical institutions raise our profile, enhance awareness of our products in the medical community, medical equipment and supplies industry and among patients, provide us with valuable clinical data to improve our products and keep us abreast of industry trends and developments, all of which in turn helps us market and sell our products. We manage our relationships with ECLIA distributors by pricing our ECLIA system more attractively to distributors than imported alternatives.
Customer Support and Service
We maintain a dedicated team to provide customer support and service for our products. Our customer support and service team is based in Beijing, and it provides on-site and off-site technical and clinical support to hospitals across China upon customer request. As of June 30, 2006, this team consisted of 36 full-time employees, including physicians, biomedical engineers, acoustics engineers, computer science engineers, and service and repair technicians. We plan to build service centers and station service engineers in major cities in China to continue providing good customer support and service as we continue to grow. We believe that our ability to provide good customer support and service is an important part of our business.
We provide training to hospitals that purchase our HIFU therapy system and our ECLIA system. Training sessions are provided directly by us or through larger hospitals that have purchased our product. We offer a two-week training course to hospitals that purchase our HIFU therapy system. Hospital participants typically include a physician, an ultrasound technician, a nurse and a maintenance engineer. In addition, due to the technical nature of our HIFU therapy system, it is usually necessary for members of our customer support and service team to assist with the installation. Our ECLIA system training is generally only one day long as our ECLIA system is relatively easy and simple to operate.
Each of our HIFU therapy systems and ECLIA analyzers are typically sold with a 12-month unlimited warranty against technical defects. We have experienced a limited number of warranty claims in the past for our HIFU therapy system. As we only began selling our ECLIA analyzer in September 2004, we currently do not have sufficient historical data on the warranty claims for our ECLIA analyzer. Consequently, the costs associated with our warranty claims have historically been relatively low. We do not generally accrue any liability for potential warranty costs at the time of sale. See “Item 5. Operating and Financial Review and Prospects—Critical Accounting Policies—Warranty obligations.”
Intellectual Property
We are committed to developing and protecting our intellectual property portfolio. We own and have applied for patents to protect the technologies, inventions and improvements that we believe are significant to our business. As of September 27, 2006, we have obtained 14 patents in China, including six utility patents and seven
36
Table of Contents
patents for inventions, directed toward different aspects of our HIFU therapy system and one utility patent directed to our ECLIA system. The validity period for our seven utility patents and seven invention patents is 10 and 20 years, respectively, from the date the application was filed. See “—Our HIFU solution—the HIFU therapy system” for a detailed description of our most important patents. We also have 19 patent applications pending in China relating to our HIFU and ECLIA systems, including seven applications for utility patents and 12 applications for inventions. Generally, invention patents are viewed as offering more commercial protection than utility patents. As with patent rights in most other jurisdictions, a patent holder in the PRC enjoys the exclusive right to exclude others from using, licensing and otherwise exploiting the patent within the PRC. To protect some of the most important proprietary technologies internationally, we have applied for four HIFU technology patents in each of the United States, the United Kingdom, Germany, Japan, South Korea, Saudi Arabia and India. As of September 27, 2006, we have been granted three out of the four patents we applied for in the United Kingdom.
We have developed, among others, the following key technologies:
| • | a wide convergence angle range for ultrasound waves—Our HIFU therapy system is capable of producing ultrasound waves from a wide convergence angle range in order to reduce the intensity of ultrasound waves when they penetrate the surface of the skin. This technology significantly reduces the likelihood of skin burns and collateral damage to surrounding body tissues. We have obtained a patent for this technology in China and have applied for patents in other countries. |
| • | non-invasive ultrasound temperature detecting—This technology is designed to collect real time temperature data during treatment by generating “acoustic signals” while the focused ultrasound shots occur. The feedback data from these acoustic signals allow the physician to adjust the parameters of the HIFU therapy system so that the target temperature can be more precisely achieved at each focal point throughout the patient’s treatment, thus giving the physician more control over the treatment process and outcome. We have applied for a patent directed toward this technology in China and in other countries. |
| • | two-transducer design—This design enables the patient to be treated in the supine, side or seated positions so that more locations in the body can be targeted. This patented design also increases the patient’s comfort level during treatment. We have obtained a patent directed toward this technology in China and have applied for patents in other countries. |
| • | enhanced chemiluminescence substrate technology—This technology improves the chemical reaction of reagents with the patient’s body fluid sample to produce a long lasting and linear signal and enables our reagent kits to be more sensitive and accurate. We have a pending patent application directed toward this technology in China. |
| • | substance storage technology—This technology can improve the shelf life of our reagent kits from six months to two years, thus reducing the risk of inventory obsolescence. We have a pending patent application directed toward this technology in China. |
We require our employees, consultants and advisors to execute confidentiality agreements in connection with their employment, consulting and advisory relationships with us. We also require our employees, consultants and advisors who are privy to confidential information to agree to disclose and assign to us all inventions conceived during their terms of employment or contract. Despite any measures we take to protect our intellectual property, unauthorized parties may attempt to copy aspects of our products or our proprietary technology or to obtain and use information that we regard as proprietary.
We, like other medical device manufacturers in China and elsewhere, are subject to a number of uncertainties regarding the commercial value of our patent portfolio. See “Item 3. Key Information—D. Risk Factors—If we fail to protect our intellectual property rights, our competitors may take advantage of our proprietary technology and know-how and compete directly against us.”
37
Table of Contents
Competition
Rapid product development, technological advances, intense competition and a strong emphasis on proprietary products characterize the industry in which we compete. Our products could be rendered obsolete or uneconomical by the introduction and market acceptance of competing products or by technological advances of our current or potential competitors. We face direct competition from a number of publicly traded and privately held companies, including other manufacturers of tumor therapeutic and diagnostic products. Many of our existing and potential competitors have substantially greater financial, research and development, sales and marketing, personnel and other resources than we do and may have more experience in developing, manufacturing, marketing and supporting new products. Competition will intensify as more players enter this field. We believe that an important competitive advantage for a medical technology company is its ability to make continuous investments in research and development and to identify commercially viable technologies and successfully bring the technologies into the market.
Insurance
We maintain insurance coverage from PICC Property and Casualty Company Limited, with an annual aggregate insured amount of RMB4.0 million (US$0.5 million) to cover product liability claims arising from the use of our HIFU therapy system. We also have property insurance coverage from China Pacific Property Insurance Co., Ltd. to cover certain of our fixed assets. Our insurance coverage, however, may not be sufficient to cover any claim for product liability or damage to our fixed assets. See “Item 3. Key Information—D. Risk factors—Risks related to our company—We are subject to product liability exposure and have limited insurance coverage.”
Regulation
Our HIFU therapy system and ECLIA analyzer are medical devices and are subject to regulatory controls governing medical devices. Reagents used for IVD testings are divided into the categories of biological reagents and chemical and bio-chemical reagents. Biological reagents are subject to regulatory controls similar to those governing pharmaceutical products, while chemical and bio- chemical reagents are subject to regulatory controls similar to those governing medical devices. As a manufacturer of medical equipment and supplies we are subject to regulation and oversight by different levels of the food and drug administration in China, in particular, the State Food and Drug Administration, or the SFDA. We are also subject to other government laws and regulations which are applicable to manufacturers in general. SFDA requirements include obtaining production permits, compliance with clinical testing standards, manufacturing practices, quality standards, applicable industry standards and adverse event reporting, and advertising and packaging standards.
Classification of Medical Devices
In China, medical devices are classified into three different categories, Class I, Class II and Class III, depending on the degree of risk associated with each medical device and the extent of control needed to ensure safety and effectiveness. Classification of a medical device is important because the class to which a medical device is assigned determines, among other things, whether a manufacturer needs to obtain a production permit and the level of regulatory authority involved in obtaining such permit. Classification of a device also determines the types of registration required and the level of regulatory authority involved in effecting the registration.
Class I devices are those with low risk to the human body and are subject to “general controls.” Class I devices are regulated by the city level food and drug administration where the manufacturer is located. Class II devices are those with medium risk to the human body and are subject to “special controls.” Class II devices require product certification, usually through a quality system assessment, and are regulated by the provincial level food and drug administration where the manufacturer is located. Class III devices are those with high risk to the human body, such as life-sustaining, life-supporting or implantable devices, and are regulated by the SFDA under the strictest regulatory control.
38
Table of Contents
Our HIFU therapy system is classified as a Class III device, and it therefore is subject to all regulatory controls governing Class III medical devices, as well as special regulations governing HIFU medical devices. Our ECLIA analyzers are classified as Class II or Class III devices.
Production permit
A manufacturer must obtain a production permit from the provincial level food and drug administration before commencing the manufacture of Class II or Class III medical devices. A production permit, once obtained, is valid for five years and is renewable upon expiration. Our production permit for the manufacture of our HIFU therapy system and our ECLIA analyzer will expire on July 3, 2010. To renew a production permit, a manufacturer needs to submit to the provincial level food and drug administration an application to renew the permit, along with required information six months before the expiration date of the permit. We do not believe it will be difficult for us to renew our production permit.
Registration requirements
Before a medical device can be manufactured for commercial distribution, a manufacturer must effect medical device registration by proving the safety and effectiveness of the medical device to the satisfaction of respective levels of the food and drug administration. In order to conduct a clinical trial on a Class II or Class III medical device, the SFDA requires manufacturers to apply for and to obtain in advance a favorable inspection result for the device from a third party inspection center recognized by the SFDA. The application to the inspection center must be supported by appropriate data, such as animal and laboratory testing results. If the inspection center approves the application for clinical trial, and the respective levels of the food and drug administration approve the institutions which will conduct the clinical trials, the manufacturer may begin the clinical trial. A registration application for a Class II or Class III device must provide certain pre-clinical and clinical trial data and information about the device and its components regarding, among other things, device design, manufacturing and labeling. The provincial level food and drug administration, within 60 days of receiving an application for the registration of a Class II device, and the SFDA, within 90 days of receiving an application for the registration of a Class III device, will notify the applicant whether the application for registration is approved. If approved, a registration certificate will be issued within ten days of the notification. If the food and drug administration requires supplemental information, the approval process may take much longer. The registration is valid for four years.
Clinical trials for our HIFU therapy system are also subject to SFDA regulation governing HIFU medical devices. Under these regulations, the minimum clinical trial period for a HIFU medical device is one year, with a minimum of 100 subjects participating and utilizing a minimum of two units of the HIFU medical device. Compliance with SFDA regulations on clinical investigations also includes satisfactory clinical reports from a minimum of two institutions which participated in the clinical trial, proper labeling of the device, obtaining informed consent from each subject, and compliance with monitoring and record-keeping requirements.
The SFDA occasionally changes its policies, adopts additional regulations, revises existing regulations or tightens enforcement, each of which could block or delay the approval process for a medical device. For example, in November 2001, the SFDA adopted regulations requiring additional clinical trials to be conducted if a manufacturer of HIFU medical devices seeks to increase the types of tumors its device is approved to treat. A 2003 regulation further specifies that a 100-patient clinical trial is required before any medical device can be approved for commercial production.
We received SFDA approval for our first generation HIFU therapy system in September 1999 for the treatment of solid tumors in the pelvic cavity and the abdominal cavity. Solid tumors in the pelvic cavity include bladder, uterine, prostate and rectal tumors, uterine fibroids and prostate hypertrophy. Solid tumors in the abdominal cavity include pancreatic, rear abdominal, liver and kidney tumors and metastasis tumors of the abdominal lymph node. We discontinued the production of our first generation HIFU therapy system in
39
Table of Contents
December 2002. Although the September 1999 approval expired on May 24, 2004, it is still valid for all units of our first generation HIFU therapy system that were sold to hospitals before our approval expired. In January 2003, we received SFDA approval for our second generation HIFU Therapy system for the treatment of liver tumors, breast tumors, kidney tumors, solid tumors in the pelvic cavity, tumors in the four limbs or superficial tissues and solid tumors on bone. This approval is valid until January 24, 2007, and we do not believe it will be difficult for us to renew this approval.
Beijing Weixiao received SFDA approval for our ECLIA analyzer in February 2004. After our ECLIA acquisition, we applied for the transfer of the SFDA approval to us and completed the transfer in March 2005. The SFDA approval we have obtained is valid from March 21, 2005 to March 20, 2009 and we do not believe it will be difficult for us to renew this approval.
Regulation of IVD reagents
Under a regulation enacted by the SFDA in September 2002, IVD reagents are divided into the categories of biological reagents and chemical and bio-chemical reagents. Biological reagents are subject to regulatory controls similar to those governing pharmaceutical products, while chemical and bio-chemical reagents are subject to regulatory controls similar to those governing medical devices.
A manufacturer of pharmaceutical products must obtain a production permit from the food and drug administration at the provincial level. A production permit, once obtained, is valid for five years and is renewable upon expiration. Our production permit for the manufacture of reagents will expire on July 3, 2010. To renew the production permit, a manufacturer needs to submit to the provincial level food and drug administration an application to renew the permit, along with required information six months before the expiration date of the permit. We do not believe it will be difficult for us to renew our production permit. In addition, pharmaceutical manufacturing operations are also subject to periodic re-inspection and must comply with applicable quality regulations.
Prior to selling a pharmaceutical product, a manufacturer must obtain both SFDA registration of, and SFDA approval for, the product it plans to sell. The registration process starts with submitting a registration dossier to the SFDA. The registration dossier contains primarily detailed information concerning the effectiveness and quality of the reagent. It also contains details concerning the manufacturing process and the production facilities. The registration process can last a few months and the timing depends on the nature of the pharmaceutical product under review, the quality of the data and the general efficiency of the SFDA. The approval process involves the detailed examination of the testing, effectiveness, labeling, manufacturing and marketing of pharmaceutical products by the SFDA. We currently sell a total of 56 reagents in China. Of these 56 reagents, 30 are covered by registration certificates. Of the 26 reagents we currently sell for which we do not have registration certificates, we have submitted registration applications for 22.
Thus, although regulations require that both SFDA registration and approval be obtained before a biological reagent kit is sold and SFDA registration be obtained before a chemical or bio-chemical reagent kit is sold, we have sold many of our reagent kits without required registrations and/or approvals. In addition, we believe that many reagent kits sold in China, including some manufactured by our overseas-based competitors, are also sold without required registrations and/or approvals. Although no enforcement action by the SFDA has been brought against us to date, we cannot assure you that enforcement actions will not be brought against us or such enforcement actions, if brought, will not have a retroactive effect on us. These enforcement actions could result in fines, injunctions and civil penalties, recall or seizure of our products, the imposition of operating restrictions, partial suspension and complete shutdown of production and criminal prosecution. See “Item 3. Key Information—D. Risk factors—Risks related to our industry—In order to manufacture and market our products, we are required to obtain various authorizations from governmental regulatory authorities in China. In some cases, we have been operating without these authorizations, which could subject us to significant penalties.” and “Item 3. Key Information—D. Risk factors—Risks related to doing business in China—Future changes in laws, regulations or enforcement policies in China could adversely affect our business.”
40
Table of Contents
In October 2004, the SFDA imposed certain good manufacturing practice, or GMP, requirements for biological IVD reagents. As a result, all manufacturers of biological IVD reagents were obligated to meet GMP standards by January 1, 2006. We are currently in compliance with such GMP requirements and obtained GMP certification for our ECLIA system in November 2005. This GMP certification is valid until October 2010.
Continuing SFDA regulations
We are subject to continuing regulation by the SFDA. In the event of significant modification to an approved medical device, its labeling or its manufacturing process, a new pre-market approval or pre-market approval supplement may be required. Our HIFU therapy system and our ECLIA analyzer are subject to, among others, the following regulations:
| • | SFDA’s quality system regulations, which require manufacturers to create, implement and follow certain design, testing, control, documentation and other quality assurance procedures; |
| • | medical device reporting regulations, which require that manufacturers report to the SFDA certain types of adverse reactions and other events involving their products; and |
| • | SFDA’s general prohibition against promoting products for unapproved uses. |
Class II and III devices may also be subject to special controls applicable to them, such as supply purchase information, performance standards, quality inspection procedures and product testing, which may not be required for Class I devices. We believe we are in compliance with the applicable SFDA guidelines, but we could be required to change our compliance activities or be subject to other special controls if the SFDA changes or modifies its existing regulations or adopts new requirements.
We are also subject to inspection and market surveillance by the SFDA to determine compliance with regulatory requirements. If the SFDA decides to enforce its regulations and rules, the agency can institute a wide variety of enforcement actions, such as:
| • | fines, injunctions and civil penalties; |
| • | recall or seizure of our products; |
| • | the imposition of operating restrictions, partial suspension or complete shutdown of production; and |
| • | criminal prosecution. |
Other national and provincial level laws and regulations
We are subject to changing regulation under many other laws and regulations administered by governmental authorities at the national, provincial and city levels, some of which are, or may be, applicable to our business. Our hospital customers are also subject to a wide variety of laws and regulations that could affect the nature and scope of their relationships with us.
Laws regulating medical device manufacturers and hospitals cover a broad array of subjects. For example, regulations control the confidentiality of patient medical information and the circumstances under which patient medical information may be released for inclusion in our databases, or released by us to third parties. These laws and regulations governing both the disclosure and the use of confidential patient medical information may become more restrictive in the future.
We must also comply with numerous additional state and local laws relating to matters such as safe working conditions, manufacturing practices, environmental protection and fire hazard control. We believe we are currently in compliance with these laws and regulations; however, we may be required to incur significant costs to comply with these laws and regulations in the future. Unanticipated changes in existing regulatory requirements or adoption of new requirements could therefore have a material adverse effect on our business, results of operations and financial condition.
41
Table of Contents
Regulation of foreign currency exchange and dividend distribution
Foreign currency exchange regulation in China is primarily governed by the following rules:
| • | Foreign Currency Administration Rules (1996), as amended, or the Exchange Rules; and |
| • | Administration Rules of the Settlement, Sale and Payment of Foreign Exchange (1996), or the Administration Rules. |
Under the Exchange Rules, the Renminbi is convertible for current account items, including the distribution of dividends, interest payments, trade and service-related foreign exchange transactions. Conversion of Renminbi for capital account items, such as direct investment, loan, security investment and repatriation of investment, however, is still subject to the approval of the SAFE.
Under the Administration Rules, companies in China with foreign investments, such as YDME Beijing, may only buy, sell and/or remit foreign currencies at those banks authorized to conduct foreign exchange business after providing valid commercial documents and, in the case of capital account item transactions, obtaining approval from the SAFE. Capital investments by foreign-invested enterprises outside of China are also subject to limitations, which include approvals by the SAFE and other relevant government authorities.
The principal regulations governing distribution of dividends paid by wholly foreign owned enterprises include:
| • | Wholly Foreign Owned Enterprise Law (1986), as amended; and |
| • | Wholly Foreign Owned Enterprise Law Implementation Rules (1990), as amended. |
Under these regulations, foreign-invested enterprises in China may pay dividends only out of their accumulated profits, if any, determined in accordance with PRC accounting standards and regulations. In addition, foreign-invested enterprises in China are required to set aside certain amounts out of their accumulated profits each year, if any, to fund certain reserve funds. These reserves are not distributable as cash dividends.
Regulation of foreign exchange in certain onshore and offshore transactions.
In January and April 2005, the SAFE issued two rules that require PRC residents to register with and receive approvals from the SAFE in connection with their offshore investment activities. The SAFE has announced that the purpose of these regulations is to achieve the proper balance of foreign exchange and the standardization of the cross-border flow of funds.
On October 21, 2005, the SAFE issued the Notice on Issues Relating to the Administration of Foreign Exchange in Fund-raising and Reverse Investment Activities of Domestic Residents Conducted via Offshore Special Purpose Companies, or Notice 75, which became effective as of November 1, 2005. Notice 75 replaced the two rules issued by the SAFE in January and April 2005 mentioned above.
According to Notice 75:
(i) prior to establishing or assuming control of an offshore company for the purpose of financing that offshore company with assets or equity interests in an onshore enterprise in the PRC, each PRC resident who is an ultimate controller, whether a natural or legal person, must complete the overseas investment foreign exchange registration procedures with the relevant local SAFE branch;
(ii) an amendment to the registration with the local SAFE branch is required to be filed by any PRC resident that directly or indirectly holds interests in that offshore company upon either (a) the injection of equity interests or assets of an onshore enterprise to the offshore company, or (b) the completion of any overseas fund raising by such offshore company; and
42
Table of Contents
(iii) an amendment to the registration with the local SAFE branch is also required to be filed by such PRC resident when there is any material change involving a change in the capital of the offshore company, such as (a) an increase or decrease in its capital, (b) a transfer or swap of shares, (c) a merger or division, (d) a long-term equity or debt investment, or (e) the provision of a guarantee to third parties.
Moreover, Notice 75 applies retroactively and to indirect shareholdings. As a result, PRC residents who have established or acquired direct or indirect control of offshore companies that have made onshore investments in the PRC in the past are required to complete the relevant overseas investment foreign exchange registration procedures by March 31, 2006. Under the relevant rules, failure to comply with the registration procedures set forth in Notice 75 may result in restrictions being imposed on the foreign exchange activities of the relevant onshore company, including the payment of dividends and other distributions to its offshore parent or affiliate and the capital inflow from the offshore entity, and may also subject relevant PRC residents to penalties under PRC foreign exchange administration regulations.
As of July 11, 2005, Mr. Xiaodong Wu, who is the shareholder of Chengxuan and a PRC resident and subject to the above registration requirements, had made the required SAFE registration in respect of his investment in Chengxuan and in our company. However, the change in Chengxuan’s shareholding in our company as a result of our initial public offering, our follow-on offering and equity transactions thereafter, as well as the establishment of CMED Technologies Ltd. and CMED Diagnostics Ltd., our two wholly owned subsidiaries incorporated in the British Virgin Islands in January 2006, are still subject to amendment registration with SAFE. Mr. Xiaodong Wu has advised us that he had submitted prior to the March 31, 2006 deadline the application for the required amendment to his SAFE registration in connection with our initial public offering, and had also submitted the required amendment in connection with our follow-offering within 30 days of the follow-on offering. However, Mr. Wu has advised us that he does not intend to submit a SAFE registration in connection with the establishment of the two entities in the British Virgin Islands.
As a result of the lack of implementing rules and other uncertainties relating to the interpretation and implementation of Notice 75, we cannot predict how these regulations will affect our business operations or strategies. For example, our present or future PRC subsidiaries’ ability to conduct foreign exchange activities, such as remittance of dividends and foreign-currency-denominated borrowings, may be subject to compliance with such SAFE registration requirements by relevant PRC residents, over whom we have no control. In addition, we cannot assure you that any such PRC residents will complete the necessary approval and registration procedures required by the SAFE regulations.
Company Law
On October 27, 2005, the Standing Committee of the National People’s Congress adopted amendments to the PRC Company Law, which substantially overhauled the PRC company law system and removed a number of legal restrictions and hurdles on the management and operations of limited liability companies and companies limited by shares. It is expected that the PRC Law of Wholly Foreign-owned Enterprises, or the WFOE Law, and its implementing regulations will be amended accordingly in order to align the WFOE Law with the amendments to the PRC Company Law. YDME Beijing is governed by both the PRC Company Law and the WFOE Law and their implementing regulations, and we believe that YDME Beijing will be able to benefit from a more flexible and business friendly company law regime under the new PRC Company Law. For example, the amended PRC Company Law eliminated a restriction which limited the amount of equity investments a company could make to a maximum of 50% of such company’s net assets. With the removal of this restriction, YDME Beijing may have increased flexibility in making equity investments or planning potential acquisitions. In addition, the amended PRC Company Law now permits the establishment of single-shareholder limited liability companies. As a result, YDME Beijing may acquire 100% of the equity interest in a PRC limited liability company and become the sole shareholder of such limited liability company.
43
Table of Contents
C. Organizational Structure
The following diagram illustrates our company’s organizational structure, and the place of formation, ownership interest and affiliation of each of our subsidiaries.
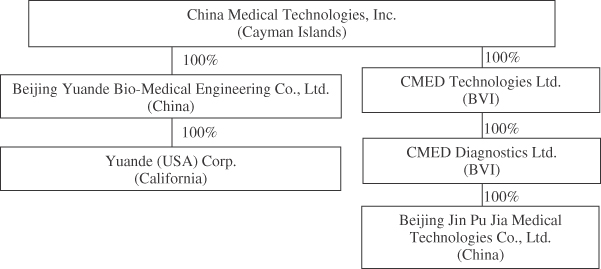
Our predecessor company, Beijing Yuande Bio-Medical Engineering Co. Ltd., or YDME Beijing, was incorporated in China and commenced business operations in July 1999. In anticipation of our initial public offering, we incorporated China Medical Technologies Inc., or China Medical, in the Cayman Islands as a listing vehicle in July 2004. China Medical became our ultimate holding company when it issued shares to the existing shareholders of YDME Beijing in January 2005 in exchange for all of the shares that these shareholders held in YDME Beijing. We conduct substantially all of our operations through YDME Beijing. In January 2005, YDME Beijing established a wholly owned subsidiary Yuande (USA) Corp., which is principally engaged in assisting research and development projects and clinical studies on using the HIFU therapy system in the United States.
| D. | Property, Plant and Equipment |
All of our manufacturing facilities are located in the Beijing Economic-Technological Development Area, where we own two buildings with an aggregate of approximately 10,000 square meters of office, research and development and manufacturing spaces. We own all of our manufacturing facilities. We believe that our existing facilities are adequate to meet our current and foreseeable future requirements.
In September 2005, we entered into a three-year lease for approximately 494 square meters of office space in a central business district of Beijing, China. We intend to use this office as our headquarters where our management and our sales and marketing team will be located.
44
Table of Contents
ITEM 5. OPERATINGAND FINANCIAL REVIEWAND PROSPECTS
You should read the following discussion and analysis of our financial condition and results of operations in conjunction with our consolidated financial statements and the related notes included elsewhere in this annual report on Form 20-F. This discussion may contain forward-looking statements based upon current expectations that involve risks and uncertainties. Our actual results may differ materially from those anticipated in these forward-looking statements as a result of various factors, including those set forth under “Item 3. Key Information—D. Risk Factors” or in other parts of this annual report on Form 20-F.
A. Operating Results
Overview
We are a China-based medical device company that develops, manufactures and markets in China products using HIFU for the treatment of solid cancers and benign tumors. We currently have two products and started reporting our financial results in two segments along product lines since our acquisition of the ECLIA technology in August 2004. Our main product, the HIFU therapy system, is an ultrasound-guided acoustic ablation system that is used for the non-invasive treatment of solid tumors. Our second product, the ECLIA system, is an enhanced chemiluminescence immunoassay IVD system that can be used to diagnose and analyze a variety of diseases and conditions, including various thyroid disorders, diabetes, hepatitis disorders related to reproduction and growth, various types of tumors and SARS, as well as to assess the effect of digoxin, a widely used drug for the treatment of heart failure.
The HIFU therapy system was our only product until September 2004. PUPH commenced research on HIFU medical devices for the treatment of tumors in the early 1990s with funding provided by Beijing Chengxuan Economic and Trade Co., Ltd, or Beijing Chengxuan, a company controlled by Mr. Xiaodong Wu. Mr. Wu is the chairman of our board of directors, our chief executive officer and a significant shareholder of our company. Clinical trials of an early version of the HIFU therapy system commenced in 1998. We acquired our HIFU technology at a price of RMB6.0 million from PUPH and Beijing Chengxuan and commenced the commercial production and distribution of our HIFU therapy system in November 1999. This was shortly after YDME Beijing was co-founded by PUPH and Beijing Chengxuan.
We began marketing and selling our ECLIA system in September 2004. PUPH commenced research on ECLIA technology in the early 1990s with funds provided by Beijing Weixiao, a company controlled by Mr. Wu. Beijing Weixiao began participating in ECLIA technology research in August 2001 and subsequently acquired the ECLIA technology from PUPH in July 2003. We acquired the ECLIA technology from Beijing Weixiao in August 2004. Pursuant to the acquisition agreement, we paid Beijing Weixiao a total purchase price of RMB250.0 million (US$31.0 million) for the ECLIA technology, related inventories and a noncompete commitment, which was paid in five installments from September 2004 to September 2005. This acquisition broadens our product offerings and presented us with a significant opportunity to market our ECLIA system to small- and medium-sized hospitals in China. Many of these hospitals currently use less advanced IVD systems because they lack sufficient financial means to purchase other luminescence immunoassay systems available in China, which are mostly automatic and imported. Many of the key research and development personnel who developed the HIFU and ECLIA technologies at PUPH are currently employed by our company.
We market and sell our HIFU therapy system to distributors and sell a small percentage of our HIFU therapy systems directly to hospitals. We market and sell our ECLIA system solely to distributors.
We have limited product offerings and depend entirely on two product lines. In addition, we have a limited operating history for you to use as a basis to evaluate our business. As a result, we are subject to the risks and difficulties frequently encountered by early-stage companies in new and rapidly evolving markets, such as the medical equipment and supplies market in China. Although we have been profitable since our fiscal year ended March 31, 2000, our business and operations have evolved rapidly and significantly since our inception in July 1999, and we expect such change and evolvement to continue in the future.
45
Table of Contents
Pricing
Distributors in China operate primarily on a for-profit basis, and hospitals in China are expected to use a substantial portion of their revenues to fund their operations. Treatment fees for HIFU tumor therapy are set by provincial governments in China, a factor we consider when pricing our HIFU therapy system. To gain market penetration, we price our HIFU therapy system at levels that we believe offer attractive economic returns to distributors and hospitals, taking into account the prices of competing products in the market. We market and sell our products to distributors at a price that is significantly lower than the price that hospitals pay for our products. We believe that our HIFU therapy system is competitively priced compared to other HIFU tumor therapy devices available in China.
We believe that our ECLIA system is competitively priced to offer attractive economic returns to our distributors. The prices of our reagent kits are significantly lower than those of foreign imports.
The provincial governments in China set the treatment fee rates for HIFU tumor therapy, and they may adjust the fee rates from time to time. If they reduce the fee rates, some hospitals and distributors may be discouraged from purchasing our HIFU therapy system, which would reduce our sales. In that event, we may need to decrease the price of our HIFU therapy system to provide our customers acceptable returns on their purchases. We cannot assure you that our business, financial condition and results of operations will not be adversely affected by any reduction in treatment fees for HIFU tumor therapy in the future.
Revenues
We currently derive revenues primarily from three sources:
| • | sales of our HIFU therapy system, which currently account for a substantial portion of our revenues; |
| • | sales of our ECLIA analyzer, which we expect to increase as a percentage of our revenues; and |
| • | sales of our ECLIA reagent kits, which currently generate limited revenue but we expect to grow significantly as more hospitals start using our ECLIA analyzer. |
Our net revenues are net of VAT, sales returns, trade discounts and allowances, but include VAT refunds on the sales of self- developed software embedded in our HIFU therapy system and our ECLIA system. See “—Taxation.”
Revenues from sales represent the invoiced value of goods, net of value added taxes, sales returns, trade discounts and allowances. We recognize revenues at the time our products are accepted by either our distributors or the hospitals designated by these distributors depending on contractual stipulation, which typically occurs within one to two weeks of shipment.
Our revenues, growth and results of operations depend on several factors, including the level of acceptance of our products among doctors, hospitals and patients and our ability to maintain prices for our products at levels that provide favorable margins. The level of acceptance among doctors, hospitals and patients is influenced by the performance and pricing of our products, our ability to educate distributors and the medical community about our products, our relationships with hospitals and major distributors, government reimbursement levels as well as other factors.
Our revenues have fluctuated significantly from quarter to quarter in the past due to the fluctuation in the sales of our HIFU therapy system. Sales of our HIFU therapy system are seasonal, and our third and fourth fiscal quarters, which are the quarters ending December 31 and March 31, respectively, have historically been our strongest. We believe that the relatively stronger performance in our third and fourth fiscal quarters is largely due to the budget cycles of hospitals in China. Hospitals in China typically make their capital expenditure decisions, such as decisions on whether to purchase our HIFU therapy system, between June and December. Our HIFU therapy system is usually shipped to customers between September to March. As our main product, the HIFU therapy system, is sold at a relatively high unit price, the size and timing of individual orders can have a
46
Table of Contents
significant effect on our results of operations in any given quarter. We only began to sell our ECLIA system in September 2004. We expect that sales of our ECLIA system will fluctuate to a lesser extent from quarter to quarter compared to sales of our HIFU therapy system.
Our sales have historically been made on the basis of unit-by-unit purchase orders rather than long-term commitments, and we do not have long-term contracts with any of our customers. We expect that in any given period a relatively small, and changing, number of distributors will continue to account for a significant portion of our revenues. For the fiscal year ended March 31, 2005 and 2006, sales to our top five customers accounted for 66.5% and 61.8%, respectively, of our revenues. In the earlier stage of our development, a significant percentage of the sales of our HIFU therapy systems was generated through our own direct sales. At present, we rely predominantly on sales to distributors rather than our own direct sales. In the three fiscal years ended March 31, 2004, 2005 and 2006, 95.2%, 97.0% and 98.9%, respectively, of our HIFU therapy systems were sold to distributors. As the target end markets for our HIFU therapy system and our ECLIA system are different, we expect our two products to be sold to different groups of distributors. We cannot assure you that any customer will continue to purchase our products at the same levels as in prior years or that our relationship with any of them will continue.
We expect that a substantial portion of our sales will continue to be generated by a relatively small group of distributors that may change from year to year. Such changes are due primarily to the fragmented nature of the medical equipment distribution industry in China. Distributors of our HIFU therapy systems are typically companies that only have the resources to target a limited number of hospitals at a given time. Once they have won orders from targeted hospitals, it takes some time to develop relationships with new hospitals. In addition, we require our distributors to pay us in full within a year of accepting our products, but our distributors may give their hospital customers longer payment terms under various types of sales arrangements, including profit-sharing programs, in order to facilitate their own sales. For example, under profit-sharing programs, distributors install our HIFU therapy system in hospitals for free and, in return, receive a portion of the net profits generated by the system. Such profit-sharing programs require upfront capital commitments from distributors upon their purchase of the HIFU therapy system from us. As a result, these distributors may need time to recover their financial resources to purchase additional products from us.
For sales of our HIFU therapy system, we generally collect 30.0% to 40.0% of the sale price when customers place purchase orders or after the shipment is accepted by either our distributors or the hospitals designated by these distributors, and the balance within one year of acceptance. For sales of ECLIA analyzer, we collect 20.0% to 40.0% of the sale price before delivery or after acceptance and the balance within one year of acceptance. For sales of reagent kits, we do not require down payments, and collect payments three months after acceptance.
Costs
Cost of revenues. Our cost of revenues primarily consists of material and component costs. It also includes amortization of intangible assets and direct costs incurred in the assembly, installation and service of our products, such as salaries and related personnel expenses and depreciation costs of plant and equipment used for production purposes. Depreciation of property, plant and equipment attributable to manufacturing activities is capitalized as part of inventory, and expensed as cost of revenues when product is sold. Depreciation for the fiscal years ended March 31, 2004, 2005 and 2006 in the amount of RMB0.8 million, RMB1.2 million and RMB1.6 million (US$0.2 million), respectively, were recorded as cost of revenue. We produce our reagent kits, and conduct the final product assembly, testing and packaging of our HIFU therapy systems and our ECLIA analyzers, in-house and outsource the production of a majority of our components to third-party suppliers.
As we source a significant portion of our components and raw materials in China, we currently have a relatively low cost base compared to medical technology companies in more developed countries. We expect the costs of components and raw materials in China will increase in the future as a result of further economic development in China. In addition, our focus on new generations and applications of our products may require
47
Table of Contents
higher cost components and raw materials. We plan to offset increases in our cost of raw materials and components through more efficient product designs and product assembly enhancements as well as through savings due to economies of scale.
Operating expenses. Our operating expenses primarily consist of research and development expenses, sales and marketing expenses and general and administrative expenses.
| • | Research and development. Research and development expenses primarily consist of costs associated with the design, development, testing and enhancement of our existing products. These costs consist of expenditures for purchases of supplies, clinical trials, salaries and related personnel expenses, and other relevant costs. Going forward, we expect to increase our research and development expenses, both on an absolute basis and as a percentage of revenue, to develop new applications and improve the product designs of our HIFU therapy system and ECLIA system. |
| • | Sales and marketing. Sales and marketing expenses consist primarily of salaries and related expenses for personnel engaged in sales, marketing and customer support functions and costs associated with advertising and other marketing activities. We also include warranty costs, if any, in sales and marketing expenses. Similar to most China-based manufacturers of medical equipment and supplies, our sales are made primarily to distributors. As a result, our sales and marketing expenses as a percentage of revenues are significantly lower than manufacturers of medical equipment and supplies that operate their own marketing and distribution networks and sell directly to hospitals. Going forward, we expect to increase our expenditures on sales and marketing, both on an absolute basis and as a percentage of revenue, to promote our products, especially our ECLIA system. |
| • | General and administrative. General and administrative expenses consist primarily of salaries and benefits and related costs for our administrative personnel and management, fees and expenses of our outside advisers, including legal, audit and valuation expenses, expenses associated with our administrative offices and the depreciation of equipment used for administrative purposes. We expect that our general and administrative expenses will increase, both on an absolute basis and as a percentage of revenue, as we hire additional personnel and incur costs related to the anticipated growth of our business. |
| • | Share-based Compensation Expenses.We adopted our 2005 stock option plan in February 2005. On March 14, 2005, we granted options to acquire 200,000 of our ordinary shares to each of two of our directors under this stock option plan and on August 8, 2005, we granted options to acquire 200,000 of our ordinary shares to each of two additional directors under this stock option plan. We currently account for these options in accordance with APB 25, and its related interpretations, which require us to record a compensation charge for the excess of the fair value for the stock at the grant date over the amount the grantee must pay to acquire the stock. We recognize the compensation expense over the applicable service period of the grantee, which is usually the vesting period. See “—Critical Accounting Policies—Share-based compensation.” |
Taxation
Under the current laws of the Cayman Islands, we are not subject to tax on our income or capital gains. In addition, no Cayman Islands withholding tax will be imposed on payments of dividends by us to our shareholders.
Under the current PRC laws, YDME Beijing is subject to EIT and VAT. YDME Beijing has been classified as a high technology company and currently operates in an approved economic-technological development area. As such, it is currently subject to an EIT rate of 15%, compared to a statutory rate of 33% for most companies in China. This classification also exempted YDME Beijing from paying the EIT for the three calendar years ended December 31, 1999, 2000 and 2001 and reduced YDME Beijing’s EIT rate to 7.5% for the three calendar years ended December 31, 2002, 2003 and 2004. In addition, as a “foreign-invested advanced technology enterprise” certified by the Beijing Commerce Bureau, YDME Beijing is entitled to a reduced EIT rate of 10% for the three
48
Table of Contents
calendar years ending December 31, 2007. YDME Beijing obtained approval from the tax authority in February 2006 regarding this reduction in EIT rate. YDME Beijing’s EIT rate will become 15% after December 31, 2007. For the three fiscal years ended March 31, 2004, 2005 and 2006, YDME Beijing incurred income tax expense of RMB4.9 million, RMB11.9 million and RMB18.1 million (US$2.3 million), respectively. If YDME Beijing had not had a reduced EIT rate of 7.5% for the two calendar years ended December 31, 2003 and 2004 and 10% for the calendar year ended December 31, 2005, it would have had an EIT rate of 15%, and our income tax expense for the three fiscal years ended March 31, 2004, 2005 and 2006 would have been RMB9.5 million, RMB17.6 million and RMB31.1 million (US$3.9 million) respectively.
VAT is charged based on the selling price of our products at a general rate of 17%. Our revenues are recorded net of this VAT. However, we are entitled to a 14% refund of VAT on the sales of self-developed software embedded in our HIFU therapy system and our ECLIA system. We recognize the VAT refund as part of our net revenues under U.S. GAAP. For the fiscal years ended March 31, 2006, 2005 and 2004, VAT refunds amounted to RMB23.0 million (US$2.9 million), RMB14.7 million and RMB7.1 million, respectively which accounted for 6.2%, 6.8% and 6.4% of our revenues for the respective year. See “Item 3. Key Information—D. Risk factors—Risks related to doing business in China—Our business benefits from certain government incentives. Expiration of, or changes to, these incentives could have a material adverse effect on our operating results.”
Critical Accounting Policies
We prepare financial statements in accordance with U.S. GAAP, which requires us to make judgments, estimates and assumptions that affect the reported amounts of our assets and liabilities and the disclosure of our contingent assets and liabilities at the end of each fiscal period and the reported amounts of revenues and expenses during each fiscal period. We continually evaluate these judgments and estimates based on our own historical experience, knowledge and assessment of current business and other conditions, our expectations regarding the future considering available information and assumptions that we believe to be reasonable, which together form our basis for making judgments about matters that are not readily apparent from other sources. Since the use of estimates is an integral component of the financial reporting process, our actual results could differ from those estimates. Some of our accounting policies involve a greater use of estimates or require a higher degree of judgment than others in their application.
The selection of critical accounting policies, the judgments and other uncertainties affecting application of those policies and the sensitivity of reported results to changes in conditions and assumptions are factors that should be considered when reviewing our consolidated financial statements. We believe the following accounting policies involve the most significant judgments and estimates used in the preparation of our consolidated financial statements.
Depreciation and amortization expenses
We depreciate property, plant and equipment and amortize our intangible assets on a straight-line basis over the respective estimated useful lives of these intangible assets, after taking into account their estimated residual values.
An existing patent is amortized over the remaining protective period of the patent as allowed by the relevant regulatory authorities. The patent we acquired as part of our ECLIA acquisition is amortized over its remaining protected period of 17 years.
In determining the estimated useful lives of our unpatented technology in our ECLIA acquisition, we consider the following pertinent factors:
| • | the expected protective period of similarly patented technology as allowed by the relevant regulatory authorities; |
| • | the planning horizon of our operations taking into account the medical equipment and supply market in China as a whole; and |
| • | the economic lives and history of other similar diagnostic technologies currently used by hospitals in the world. |
49
Table of Contents
As a result, we have estimated the useful life of the unpatented technology we acquired as part of our ECLIA acquisition to be 20 years.
The estimated useful life of the noncompete commitment we acquired in our ECLIA acquisition is based on the period of time during which the noncompete commitment is expected to be of value to us, which we consider to be the estimated time required for Beijing Weixiao to develop a similar technology and the length of the impact if Beijing Weixiao were to develop the technology and to compete with us. As a result, we have estimated the useful life of the noncompete commitment to be 10 years.
We review the estimated useful lives and estimated residual values of our assets regularly in order to determine the amount of depreciation and amortization expenses to be recorded during any reporting period. We determine the estimated useful lives for property, plant and equipment based on our historical usage experience with similar assets and taking into account anticipated technological changes.
We adjust the depreciation and amortization expenses for future periods if there are significant changes from previous estimates. For the periods presented, no significant changes in estimated useful lives or depreciation/amortization expenses occurred.
Recoverability of long-lived assets
We review periodically the carrying amounts of our long-lived assets to be held and used, including property, plant and equipment and intangible assets in order to assess whether the recoverable amounts, based on expected undiscounted net cash flows, have declined below the carrying amounts. We test these assets for impairment whenever events or changes in circumstances indicate that their recorded carrying amounts may not be recoverable. When such a decline has occurred, we reduce the carrying amount to the estimated fair value and record the difference as an impairment loss, which is charged against earnings. We determine estimated fair value using expected future cash flows generated by the assets and such future cash flows are discounted to their present value, which requires significant judgment in terms of projection of cash flows for future years and the assumption on the pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset. For the periods presented, we have not been required to recognize any impairment losses in our consolidated financial statements.
Collectibility of our accounts receivable
We evaluate the collectibility of our accounts receivable based on the aging of account balances, collection history, credit quality of the customer and current economic conditions that may affect a customer’s ability to pay.
We currently do not recognize an allowance for doubtful accounts in our consolidated financial statements because of our past collection experience. However, we have been dependent on a small number of distributors for a significant portion of our revenues. If a major customer’s financial condition deteriorates, or if our customer default rates become higher than historical levels, our estimates of the recoverability of amounts due to us could change, and an allowance could be required, which could have a material adverse effect on our results of operations.
Warranty obligations
Each of our HIFU therapy systems and ECLIA analyzers are typically sold with a 12-month unlimited warranty against technical defects, the term of which begins upon acceptance of the product by the customer. Costs for warranty claims are recorded as sales and marketing expenses when such claims are made. Based on the limited number of warranty claims in the past for our HIFU therapy system and the relatively low historical repair costs, we do not generally accrue any liability for potential warranty costs at the time of sale. However, if we begin to experience an increase in the frequency of claims or the amount of repair costs to cover our warranty obligations, we will begin to provide for such warranty costs at the time of product sale. If we had incurred a
50
Table of Contents
warranty claim rate of 1% of our net revenues, our estimated warranty costs for the three fiscal years ended March 31, 2004, 2005 and 2006 would have been approximately RMB1.1 million, RMB2.2 million and RMB3.7 million (US$0.5 million), respectively. Our customers do not have a right of return with respect to products purchased from us.
Share-based compensation
On February 2, 2005, we adopted the 2005 stock option plan pursuant to which our board of directors may grant stock options to directors and employees. Under the plan, we have reserved an aggregate of 30,000,000 of our ordinary shares for issuance under the Plan. The 2005 stock option plan will remain in effect for 5 years starting from the date of adoption.
On March 14, 2005, we granted two directors options to purchase an aggregate of 400,000 ordinary shares at an exercise price of US$1.36 per share. On August 8, 2005, we granted two other directors options to purchase an aggregate of 400,000 ordinary shares at an exercise price of US$1.5 per share. One-third of the options will become exercisable one year, 18 months and two years after the vesting commencement day, respectively. All outstanding options will expire on December 31, 2009.
The fair value of the options granted is estimated on the date of grant using the dividend adjusted Black-Scholes option-pricing model.
We determined the fair value of our ordinary shares issuable under the options granted on March 14, 2005 with reference to the estimated price range of US$1.40 to US$1.60 per ordinary share and with discussions with the underwriter of our initial public offering. Accordingly, we considered US$1.50 per ordinary share, the midpoint of the estimated range of our initial public offering price, to be an appropriate indicator of the fair value of its ordinary shares. Accordingly, share-based compensation of RMB463,000 (US$58,000) was recorded, which represents the difference between the exercise price of US$1.36 per ordinary share and the estimated fair value of US$1.50 per ordinary share in respect of 400,000 ordinary shares issuable under the options granted on March 14, 2005. Deferred share-based compensation expenses was amortized on a straight-line basis over the two-year vesting period. Amortization of this share-based compensation for the year ended March 31, 2006 was RMB238,000 (US$30,000).
We determined the fair value of our ordinary shares issuable under the options granted on August 8, 2005 with reference to the estimated price range of US$1.40 to US$1.60 per ordinary share and with discussions with the underwriter of our initial public offering. Accordingly, no share-based compensation was recorded as the exercise price of US$1.50 per ordinary share is the same as the estimated fair value of US$1.50 per ordinary share in respect of 400,000 ordinary shares issuable under the options granted on August 8, 2005.
Options to purchase 133,000 ordinary shares were exercisable as of March 31, 2006 and the weighted average remaining contractual life is 3.75 years. No options were forfeited during the year ended March 31, 2006.
51
Table of Contents
Results of Operations
The following table sets forth a summary, for the periods indicated, our consolidated results of operations and each item expressed as a percentage of our total net revenues. Our historical results presented below are not necessarily indicative of the results that may be expected for any future period.
| Year ended March 31, | ||||||||||||||||||
| 2004 | 2005 | 2006 | ||||||||||||||||
| RMB | % of net revenues | RMB | % of net revenues | RMB | % of net revenues | |||||||||||||
Revenues, net(1) | 110,750 | 100.0 | % | 217,547 | 100.0 | % | 371,767 | 100.0 | % | |||||||||
Cost of revenues | (33,983 | ) | (30.7 | ) | (64,579 | ) | (29.7 | ) | (110,494 | ) | (29.7 | ) | ||||||
Total gross profit | 76,767 | 69.3 | 152,968 | 70.3 | 261,273 | 70.3 | ||||||||||||
Operating expenses: | ||||||||||||||||||
Research and development | (2,036 | ) | (1.8 | ) | (2,815 | ) | (1.3 | ) | (14,374 | ) | (3.9 | ) | ||||||
Sales and marketing | (2,632 | ) | (2.4 | ) | (4,960 | ) | (2.3 | ) | (15,327 | ) | (4.1 | ) | ||||||
General and administrative | (5,619 | ) | (5.1 | ) | (16,644 | ) | (7.6 | ) | (38,309 | ) | (10.3 | ) | ||||||
Total operating expenses | (10,287 | ) | (9.3 | ) | (24,419 | ) | (11.2 | ) | (68,010 | ) | (18.3 | ) | ||||||
Operating income | 66,480 | 60.0 | 128,549 | 59.1 | 193,263 | 52.0 | ||||||||||||
Net interest income and other income(2) | 14 | 0.0 | 1,748 | 0.8 | 23,838 | 6.4 | ||||||||||||
Income before tax | 66,494 | 60.0 | 130,297 | 59.9 | 217,101 | 58.4 | ||||||||||||
Income tax expense | (4,933 | ) | (4.4 | ) | (11,854 | ) | (5.4 | ) | (18,088 | ) | (4.9 | ) | ||||||
Net income | 61,561 | 55.6 | % | 118,443 | 54.5 | % | 199,013 | 53.5 | % | |||||||||
| (1) | Revenues, net, include revenues from sales of our products, net of VAT, sales returns, trade discounts and allowances, but include VAT refunds on the sales of self-developed software embedded in our products. |
| (2) | Net interest income and other income include “interest expense,” “interest income” and “other income” from our consolidated financial statements included elsewhere in this annual report. |
Year Ended March 31, 2006 Compared to Year Ended March 31, 2005
Revenues, net. Our revenues were divided between two operating segments—HIFU therapy system sales and ECLIA system sales. HIFU therapy system sales and ECLIA system sales accounted for 66.4% and 33.6% of our sales, respectively, for the fiscal year ended March 31, 2006 and 81.5% and 18.5%, respectively, for the fiscal year ended March 31, 2005. We expect ECLIA sales, principally sales of ECLIA reagent kits, to grow at a higher rate than that of our HIFU therapy sales. See “—Revenues.”
Our revenues increased 70.9% to RMB371.8million (US$46.4million) for the fiscal year ended March 31, 2006, from RMB217.5 million for the fiscal year ended March 31, 2005. This increase was due primarily to the increase in sales of both our HIFU therapy system and our ECLIA system. Revenues from sales of our HIFU therapy system increased 39.3% from RMB177.3 million for the fiscal year ended March 31, 2005 to RMB246.9 million (US$30.8 million) for the fiscal year ended March 31, 2006. Sales of our HIFU therapy system increased to 89 units for the fiscal year ended March 31, 2006 from 65 units for the fiscal year ended March 31, 2005. This increase was attributable to the increased market penetration of our HIFU therapy system as it gains acceptance in the medical community and with patients. Sales to our largest customer for the fiscal year ended March 31, 2006, Beijing Wei Jian Da Medical Technology Co. Ltd., amounted to RMB91.8 million (US$11.5 million), or 24.7% of our net revenues during that year while sales to the same largest customer for the fiscal year ended March 31, 2005 amounted to RMB51.3 million, or 23.6% of our net revenues for that fiscal year. Revenues from sales of our ECLIA system increased significantly to RMB124.9 million (US$15.6 million) for the fiscal year ended March 31, 2006 from RMB40.3 million for the fiscal year ended March 31, 2005 as a result of the increasing acceptance of the ECLIA system in the medical community in China and higher recurring revenue
52
Table of Contents
from reagent kit sales due to the increased installed base of ECLIA analyzers. Our revenue increase was also due in part to a significant increase in VAT refunds, which we recognize as part of our net revenues. Our VAT refunds increased to RMB23.0 million (US$2.9 million) for the fiscal year ended March 31, 2006, from RMB14.7 million for the fiscal year ended March 31, 2005, as a result of the increase in the sales of our self-developed software embedded in our HIFU therapy system and ECLIA system.
Cost of revenues and gross margin. Our cost of revenues increased 71.1% to RMB110.5 million (US$13.8 million) for the fiscal year ended March 31, 2006, from RMB64.6 million for the fiscal year ended March 31, 2005. This increase resulted primarily from a 67.7% increase in our consumption of raw materials and components to RMB88.9 million (US$11.1 million) for the fiscal year ended March 31, 2006, from RMB53.0 million for the fiscal year ended March 31, 2005. The increase in the direct material cost was due primarily to the increase of our products sold.
Our overall gross margin remained stable at approximately 70.3% for the fiscal year ended March 31, 2005 and 2006. Gross margin for HIFU therapy system decreased to 70.5% for the fiscal year ended March 31, 2006 from 72.1% for the fiscal year ended March 31, 2005 due primarily to the higher purchase price of certain new components for the production of HIFU therapy system. Gross margin for ECLIA system increased to 69.9% for the fiscal year ended March 31, 2006 from 62.7% for the fiscal year ended March 31, 2005 due primarily to the increase in economies of scale in our ECLIA operation, which resulted in the decrease in per unit cost allocation of the depreciation and amortization and other related production overheads.
Operating expenses. Our operating expenses increased significantly to RMB68.0 million (US$8.5 million) for the fiscal year ended March 31, 2006, from RMB24.4 million for the fiscal year ended March 31, 2005. This increase was due primarily to significant increases in our research and development expenses, our sales and marketing expenses and our general and administrative expenses. Our operating expenses as a percentage of revenues increased to 18.3% for the fiscal year ended March 31, 2006 from 11.2% for the fiscal year ended March 31, 2005. Operating expenses for HIFU therapy system increased 83.5% to RMB32.3 million (US$4.0 million) for the fiscal year ended March 31, 2006 from RMB17.6 million for the fiscal year ended March 31, 2005. Operating expenses for ECLIA system increased significantly to RMB12.3 million (US$1.5 million) for the fiscal year ended March 31, 2006 from RMB3.9 million for the fiscal year ended March 31, 2005.
Our research and development expenses increased significantly to RMB14.4 million (US$1.8 million) for the fiscal year ended March 31, 2006, from RMB2.8 million for the fiscal year ended March 31, 2005. The principal reason for this increase was our increased headcount in our research and development department, especially in our ECLIA division, as well as our effort to develop new products. Our research and development expenses as a percentage of revenues increased to 3.9% for the fiscal year ended March 31, 2006 from 1.3% for the fiscal year ended March 31, 2005.
Our sales and marketing expenses increased significantly to RMB15.3 million (US$1.9 million) for the fiscal year ended March 31, 2006, from RMB5.0 million for the fiscal year ended March 31, 2005. This increase was due primarily to increased promotional activities, increased headcount and increased sales of our HIFU therapy system and ECLIA system, which increased expenditure on our customer support functions. In addition, we increased expenditure on our sales and marketing efforts during this period to further promote our ECLIA system. Our sales and marketing expenses as a percentage of revenues increased to 4.1% for the fiscal year ended March 31, 2006 from 2.3% for the fiscal year ended March 31, 2005.
Our general and administrative expenses increased significantly to RMB38.3 million (US$4.8 million) for the fiscal year ended March 31, 2006, from RMB16.6 million for the fiscal year ended March 31, 2005. This increase was due primarily to increased headcount, increased costs associated with being a public company and an increase in our contribution to the staff bonus and welfare fund, which was calculated based on a certain percentage of our net income, as a result of the increase in our net income for the fiscal year ended March 31, 2006. Our general and administrative expenses as a percentage of revenues increased to 10.3% for the fiscal year ended March 31, 2006 from 7.6% for the fiscal year ended March 31, 2005.
53
Table of Contents
Net interest income and other income. Our net interest income increased significantly to RMB14.0 million (US$1.7 million) for the fiscal year ended March 31, 2006, from RMB0.2 million for the fiscal year ended March 31, 2005. This increase was due primarily to an increase in interest income generated from the net proceeds received from our initial public offering in August 2005. Other income increased significantly to RMB9.8 million (US$1.2 million) for the fiscal year ended March 31, 2006 from RMB1.6 million for the fiscal year ended March 31, 2005, which was due primarily to increased non-refundable grants from the PRC government authority in recognition of development of our advanced HIFU therapy system and the arrangement fee received by the Company from certain shareholders for the Company’s arrangement of the follow-on offering in March 2006 these shareholders participated.
Income tax expense. Our effective tax rate for the fiscal year ended March 31, 2006 was 8.3%, compared to 9.1% for the fiscal year ended March 31, 2005.
Net income. Net income increased significantly to RMB199.0 million (US$24.8 million) for the fiscal year ended March 31, 2006, from RMB118.4 million for the fiscal year ended March 31, 2005, as a result of the cumulative effect of the above factors.
Year Ended March 31, 2005 Compared to Year Ended March 31, 2004
Revenues, net. Beginning in the fiscal year ended March 31, 2005, our revenues were divided between two operating segments—HIFU therapy system sales and ECLIA system sales. HIFU therapy system sales and ECLIA system sales accounted for 81.5% and 18.5% of our sales, respectively, for the year ended March 31, 2005 and 100.0% and 0%, respectively, for the year ended March 31, 2004. We expect ECLIA sales, including sales of ECLIA analyzer and reagent kits, to grow at a higher rate than that of our HIFU therapy sales. See “—Revenues.”
Our revenues increased 96.3% to RMB217.5 million for the fiscal year ended March 31, 2005, from RMB110.8 million for the fiscal year ended March 31, 2004. This increase was due primarily to a significant increase in revenues generated from the sale of our HIFU therapy system and, to a lesser extent, the introduction of our ECLIA system to the market during the fiscal year ended March 31, 2005. Revenues from sales of our HIFU therapy system increased by 60.0% from RMB110.8 million for the fiscal year ended March 31, 2004 to RMB177.3 million for the fiscal year ended March 31, 2005. Sales of our HIFU therapy system increased to 65 units for the fiscal year ended March 31, 2005 from 42 units for the fiscal year ended March 31, 2004. We believe that the increase in the HIFU therapy system sales volume resulted from the increased awareness and acceptance of our HIFU therapy system in the medical community and among patients. We sold HIFU therapy systems to a number of new customers for the fiscal year ended March 31, 2005. Sales to our largest customer for the fiscal year ended March 31, 2005, Beijing Wei Jian Da Medical Technology Co. Ltd., amounted to RMB51.3 million, or 23.6% of our net revenues during that period while sales to our largest customer for the fiscal year ended March 31, 2004, Beijing Jia Xing Fang Zhou Trade Co., Ltd., or Jia Xing Fang Zhou, amounted to RMB25.6 million, or 23.1% of our net revenue for that fiscal year. Jia Xing Fang Zhou and one of our other largest customers for the fiscal year ended March 31, 2004, Shanghai Yi Li System Technology Co., Ltd., did not purchase additional products from us in the fiscal year ended March 31, 2005; as they purchased our HIFU therapy systems to operate profit-sharing programs, we believe they would need time to recover sufficient financial resources to purchase additional products from us. See “—Revenues.” In addition, these two customers may not have enough additional experienced operating staff to manage additional profit-sharing programs. Our other top customers for the fiscal year ended March 31, 2004 remained as our top customers for the fiscal year ended March 31, 2005. These customers purchased our HIFU therapy systems either to resell or to operate profit-sharing programs. The increase was also attributable to the commencement of commercial sales of our ECLIA system in September 2004, which amounted to RMB40.3 million and accounted for 18.5% of our net revenues for the fiscal year ended March 31, 2005. Our revenue increase was also due in part to a significant increase in VAT refunds, which we recognize as part of our net revenues, to RMB14.7 million for the fiscal year ended March 31, 2005 from RMB7.1 million for the fiscal year ended March 31, 2004, as a result of the increase in the sales of our self-developed software embedded in our HIFU therapy system and ECLIA system.
54
Table of Contents
Cost of revenues and gross margin. Our cost of revenues increased 90.0% to RMB64.6 million for the fiscal year ended March 31, 2005, from RMB34.0 million for the fiscal year ended March 31, 2004. This increase resulted primarily from a 65.6% increase in our expenditures on raw materials and components to RMB53.0 million for the fiscal year ended March 31, 2005, from RMB32.0 million for the fiscal year ended March 31, 2004. The increase in the purchase of raw materials and components was due primarily to a significant increase in the number of HIFU therapy systems we sold as well as our sales of ECLIA systems for the fiscal year ended March 31, 2005.
Our gross margin increased slightly to 70.3% for the fiscal year ended March 31, 2005 from 69.3% for the fiscal year ended March 31, 2004. For our HIFU therapy system, our gross margin increased to 72.1% for the fiscal year ended March 31, 2005 from 69.3% for the fiscal year ended march 31, 2004. The increase was due primarily to the increase in economies of scale of our operations. As we increased the number of HIFU therapy systems we sold, the depreciation cost of plant and equipment and salaries expense for our production personnel on a per unit basis decreased. At the same time, we were also able to purchase raw materials and components at a lower unit cost. However, we cannot assure you that we will be able to maintain or improve our gross margin in any future period. Our gross margin for our ECLIA system was 62.7% for the fiscal year ended March 31, 2005. We did not sell any ECLIA system for the fiscal year ended March 31, 2004.
Operating expenses. Our operating expenses increased significantly to RMB24.4 million for the fiscal year ended March 31, 2005, from RMB10.3 million for the fiscal year ended March 31, 2004. This increase was due primarily to significant increases in our general and administrative and sales and marketing expenses, and to a lesser degree, an increase in our research and development expenses. Our operating expenses as a percentage of revenues increased to 11.2% for the fiscal year ended March 31, 2005 from 9.3% for the fiscal year ended March 31, 2004. Operating expenses for our HIFU therapy system increased 74.3% to RMB17.6 million for the fiscal year ended March 31, 2005 from RMB10.1 million for the fiscal year ended March 31, 2004. We incurred operating expenses of RMB3.8 million for our ECLIA system for the fiscal year ended March 31, 2005.
Our research and development expenses increased by 38.3% to RMB2.8 million for the fiscal year ended March 31, 2005, from RMB2.0 million for the fiscal year ended March 31, 2004. The principal reason for this increase was our increased expenditure related to research and development of our ECLIA system. However, our research and development expenses as a percentage of revenues decreased to 1.3% for the fiscal year ended March 31, 2005 from 1.8% for the fiscal year ended March 31, 2004. The percentage decrease was primarily due to the 96.3% increase in our revenues for the fiscal year ended March 31, 2005 compared to the fiscal year ended March 31, 2004.
Our sales and marketing expenses increased significantly to RMB5.0 million for the fiscal year ended March 31, 2005, from RMB2.6 million for the fiscal year ended March 31, 2004. This increase was due primarily to increased traveling costs associated with product promotion activities. Our sales and marketing expenses as a percentage of revenues decreased slightly to 2.3% for the fiscal year ended March 31, 2005 from 2.4% for the fiscal year ended March 31, 2004.
Our general and administrative expenses increased significantly to RMB16.6 million for the fiscal year ended March 31, 2005, from RMB5.6 million for the fiscal year ended March 31, 2004. This increase was due primarily to a financial advisory fee in the amount of RMB6.2 million incurred in connection with the reorganization of YDME Beijing in preparation for our initial public offering in August 2005. The increase was also attributable to the increase in our staff bonus and welfare fund and the increase in the depreciation expense arising from the purchase of a building in March 2004. Our general and administrative expenses as a percentage of revenues increased to 7.6% for the fiscal year ended March 31, 2005 from 5.1% for the fiscal year ended March 31, 2004.
Net interest income and other income. Our net interest income increased significantly to RMB0.2 million for the fiscal year ended March 31, 2005, from RMB14,000 for the fiscal year ended March 31, 2004, which was due
55
Table of Contents
primarily to the repayment of a bank loan in the fiscal year ended March 31, 2004. We generated other income amounting to RMB1.6 million for the fiscal year ended March 31, 2005 because YDME Beijing, as a new high technology company, received a non-refundable grant from the PRC government authority.
Income tax expense. Our effective tax rate for the fiscal year ended March 31, 2005 was 9.1%, compared to 7.4% for the fiscal year ended March 31, 2004. The increase was due primarily to the expiry of the income tax concession on December 31, 2004, which led to an increase in our PRC income tax rate to 15% on January 1, 2005 from 7.5% in 2004.
Net income. Net income increased significantly to RMB118.4 million for the fiscal year ended March 31, 2005, from RMB61.6 million for the fiscal year ended March 31, 2004, as a result of the cumulative effect of the above factors.
| B. | Liquidity and Capital Resources |
Liquidity and Capital Resources
The following table sets forth a summary of our net cash flows for the periods indicated:
| Year ended March 31, | ||||||||||||
| 2004 | 2005 | 2006 | 2006 | |||||||||
| RMB | RMB | RMB | US$ | |||||||||
| (in thousands) | ||||||||||||
Net cash provided by operating activities | 67,594 | 144,771 | 166,912 | 20,820 | ||||||||
Net cash used in investing activities | (4,001 | ) | (127,667 | ) | (136,778 | ) | (17,062 | ) | ||||
Net cash (used in) provided by financing activities | (66,850 | ) | (26,572 | ) | 804,438 | 100,345 | ||||||
Net (decrease) increase in cash and cash equivalents | (3,257 | ) | (9,468 | ) | 829,145 | 103,427 | ||||||
Cash and cash equivalents at beginning of period | 27,371 | 24,114 | 14,646 | 1,827 | ||||||||
Cash and cash equivalents at end of period | 24,114 | 14,646 | 843,791 | 105,254 | ||||||||
As of March 31, 2006, we had working capital of RMB906.9 million (US$113.1 million). We believe that our current cash and cash equivalents and cash flow from operations will be sufficient to meet our anticipated cash needs, including our cash needs for working capital, research and development expenditures and capital expenditures for the foreseeable future. We may, however, require additional cash resources due to changing business conditions or other future developments, including any investments or acquisitions we may decide to pursue. If our existing cash resources are insufficient to meet our requirements, we may seek to sell additional equity securities, debt securities or borrow from banks. We cannot assure you that financing will be available in the amounts we need or on terms acceptable to us, if at all. The sale of additional equity securities, including convertible debt securities, would result in additional dilution to our shareholders. The incurrence of indebtedness would result in debt service obligations and could result in operating and financial covenants that restrict our operations and our ability to pay dividends to our shareholders. If we are unable to obtain additional equity or debt financing as required, our business, operations and prospects may suffer.
In the fiscal years ended March 31, 2004, 2005 and 2006, we have financed our operations primarily through cash generated by our operating activities and our initial public offering in August 2005. Our principal uses of cash during the fiscal year ended March 31, 2006 were for our working capital requirements, payment of RMB100 million to Beijing Weixiao in relation to our acquisition of the ECLIA technology, payment of RMB10 million in relation to the expansion of our ECLIA reagent production facility and payment of RMB17.6 million in relation to the purchase of research equipment. We received net proceeds (after deducting underwriting discounts and commissions and other expenses) of approximately US$99.7 million from our initial public offering in August 2005. As of March 31, 2006, RMB 137.6 million (US$17.2 million) of the net offering proceeds from our initial public offering had been applied towards the payment for the acquisition of the ECLIA technology, the expansion of our ECLIA production facility, the support of our sales and marketing activities, the purchase of research equipment and the support of product development activities. Our principal uses of cash for
56
Table of Contents
the fiscal year ended March 31, 2005 were for our working capital requirements, payments in connection with our ECLIA acquisition and dividend payments to our shareholders. Our principal uses of cash in the fiscal year ended March 31, 2004, were for our working capital requirements, the repayments for the principal amount of a short-term bank loan and dividend payments to our shareholders. Our external financing in the fiscal years ended March 31, 2004, 2005 and 2006 consisted primarily of the proceeds from the capital contribution of Golden Meditech and GE and our initial public offering in August 2005. As of March 31, 2006, we had RMB843.8 million (US$105.3 million) in cash and cash equivalents. Our cash and cash equivalents primarily consist of cash on hand and bank balances which are mainly held in RMB denominated accounts with banks in China U.S. dollar denominated accounts with banks in Hong Kong, Singapore and the United States.
Operating Activities
Net cash provided by operating activities increased to RMB166.9 million (US$20.8 million) for the fiscal year ended March 31, 2006, from RMB144.8 million for the fiscal year ended March 31, 2005. This increase was due primarily to a significant increase in the amount of cash provided by sales of our HIFU therapy system and ECLIA system.
Net cash provided by operating activities increased to RMB144.8 million for the fiscal year ended March 31, 2005, from RMB67.6 million for the fiscal year ended March 31, 2004. This increase was due primarily to a significant increase in the amount of cash provided by sales of our HIFU therapy system and the commencement of sales of our ECLIA system since September 2004.
Investing Activities
Net cash used in investing activities increased to RMB136.8 million (US$17.1 million) for the fiscal year ended March 31, 2006, from RMB127.7 million for the fiscal year ended March 31, 2005. Net cash used in investing activities for the fiscal year ended March 31, 2006 consisted primarily of our repayment of RMB100 million (US$12.5 million) to a related party for the remaining balance of the acquisition of ECLIA technology.
Net cash used in investing activities was RMB127.7 million for the fiscal year ended March 31, 2005, compared to net cash used in investing activities of RMB4.0 million for the fiscal year ended March 31, 2004. Net cash used in investing activities for the fiscal year ended March 31, 2005 consisted primarily of our payment of RMB148.4 million in connection with our ECLIA acquisition, partially offset by proceeds from an early withdrawal of time deposits. As a result of this acquisition, our net intangible assets amounted to RMB239.7 million and amounts due to related parties amounted to RMB102.7 million as of March 31, 2005.
Financing Activities
Net cash provided by financing activities was RMB804.4 million (US$100.3 million) for the fiscal year ended March 31, 2006, compared to net cash used in financing activities of RMB26.6 million for the fiscal year ended March 31, 2005. Net cash provided by financing activities for the fiscal year ended March 31, 2006 consisted primarily of proceeds of RMB799.1 million (US$99.7 million) raised from our initial public offering in August 2005 and advances of RMB9.6 million (US$1.2 million) from selling shareholders in our secondary offering in March 2006 for the payment of the offering expenses, partially offset by a dividend payment of RMB4.2 million (US$0.5 million).
Net cash used in financing activities decreased to RMB26.6 million for the fiscal year ended March 31, 2005 from RMB66.9 million for the fiscal year ended March 31, 2004. This decrease was due primarily to our repayments of RMB30.0 million short-term loans in May 2003.
We are a holding company, and we rely on dividends paid by our wholly owned subsidiary, YDME Beijing, for our cash needs, including the funds necessary to pay dividends and other cash distributions to our shareholders, service any debt we may incur and pay our operating expenses. The payment of dividends in China
57
Table of Contents
is subject to limitations. Regulations in the PRC currently permit payment of dividends only out of accumulated profits as determined in accordance with accounting standards and regulations in China. YDME Beijing is also required to set aside at least 10% of its after-tax profit based on PRC accounting standards each year to fund certain reserve funds. These reserves can be used to recoup previous years’ losses, if any, and, subject to the approval of the relevant government authority, may be converted into share capital in proportion to their existing shareholdings, or by increasing the par value of the shares currently held by them. Such reserves, however, are not distributable as cash dividends. In addition, if YDME Beijing incurs debt on its own behalf in the future, the instruments governing the debt may restrict YDME Beijing’s ability to pay dividends or make other distributions to us.
The ability of our subsidiary YDME Beijing to convert Renminbi into U.S. dollars and make payments to us is subject to PRC foreign exchange regulations. Under these regulations, the Renminbi is convertible for current account items, including the distribution of dividends, interest payments, trade and service-related foreign exchange transactions. Conversion of Renminbi for capital account items, such as direct investment, loan, security investment and repatriation of investment, however, is still subject to the approval of the SAFE.
Our working capital requirements vary from period to period depending on manufacturing volumes, the timing of deliveries, our payment cycles to our suppliers and the payment cycles of our customers.
Capital expenditures
We made capital expenditures of RMB0.5 million, RMB157.0 million and RMB134.1 million (US$16.7 million) in the three fiscal years ended March 31, 2004, 2005 and 2006 respectively. We estimate that our capital expenditures for the fiscal year ending March 31, 2007 will be approximately RMB46.0 million (US$5.7 million), which includes RMB38.0 million (US$4.7 million) for the expansion of our ECLIA reagent production facility. Our past capital expenditures principally consisted of purchases of a building in February 2004, production equipment for our HIFU therapy system and office equipment.
Recent Accounting Pronouncements
In July 2006, the FASB issued FASB Interpretation No. 48, “Accounting for Uncertainty in Income Taxes—an Interpretation of FASB Statement No. 109” (“FIN 48”), which clarifies the accounting for uncertainty in tax positions. This Interpretation requires that us recognize in our financial statements the impact of a tax position if that position is more likely than not of being sustained on audit, based on the technical merits of the position. The provisions of FIN 48 are effective for us on January 1, 2007, with the cumulative effect of the change in accounting principle, if any, recorded as an adjustment to opening retained earnings. We are currently evaluating the impact of adopting FIN 48 on our consolidated financial statements.
In September 2006, the SEC issued Staff Accounting Bulletin (“SAB”) No. 108, which provides guidance on the process of quantifying financial statement misstatements. In SAB No. 108, the SEC staff establishes an approach that requires quantification of financial statement errors, under both the iron-curtain and the roll-over methods, based on the effects of the error on each of our financial statements and the related financial statement disclosures. SAB No. 108 is generally effective for annual financial statements in the first fiscal year ending after November 15, 2006. The transition provisions of SAB No. 108 permits existing public companies to record the cumulative effect in the first year ending after November 15, 2006 by recording correcting adjustments to the carrying values of assets and liabilities as of the beginning of that year with the offsetting adjustment recorded to the opening balance of retained earnings. We are currently evaluating the impact of adopting SAB No. 108 on our consolidated financial statements.
In September 2006, the FASB issued SFAS No.157 Fair Value Measurements (“SFAS 157”). SFAS 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles, and expands disclosures about fair value measurements. SFAS 157 applies under other accounting
58
Table of Contents
pronouncements that require or permit fair value measurements, where fair value is the relevant measurement attribute. The standard does not require any new fair value measurements. SFAS 157 is effective for financial statements issued for fiscal years beginning after November 15, 2007, and interim periods within those fiscal years. We are currently evaluating the impact of adopting SFAS 157 on our consolidated financial statements.
| C. | Research and Development |
Research and Development
As of June 30, 2006, our research and development team consisted of 48 research personnel and medical professionals, which accounted for 17.0% of our employees. Many of the key research and development personnel who developed the HIFU and ECLIA technologies at the PUPH are currently employed by us. Our research and development staff consists of medical professionals, biomedical engineers, acoustics engineers, electrical engineers and computer science engineers, many of whom have experience in the healthcare and medical device research fields, including experience working with hospitals who are customers of our products. One of our senior researchers, Mr. Xiaolin Yang, is a council member of the scientific advisory board of International Society for Bioluminescence & Chemiluminescence and the vice president of Photobiology Committee of China Biophysics Association and is one of the original inventors of our ECLIA technology. Another of our senior researchers, Mr. Zuwen Qian, is a council member of Acoustical Society of China and a member of American Association for the Advancement of Science and is one of the original inventors of key patents and technologies for our HIFU therapy system.
Our research and development focuses primarily on developing new applications and improving the product designs of our HIFU therapy system and ECLIA system.
| • | HIFU therapy system research and development. We have expertise in the core research and development of all of the components of our HIFU therapy system. We have recently developed an ultrasound-based temperature measuring technology that can collect real time temperature data through acoustic signals that are generated by the ultrasound waves used for treatment. As the amount of ultrasound energy required to achieve the destruction of tumor tissues can vary with the location of the tumor and the specific characteristics of the tumor tissues to be treated, real time temperature data allows the physician to adjust and more precisely achieve target temperature. This device is non-invasive and provides the physician with more control over the treatment process and the outcome. We plan to incorporate this new technology in our third-generation HIFU therapy system and to sell the non-invasive ultrasound temperature detecting device separately as a product upgrade to existing users of our system. Our HIFU therapy system has been recognized as one of the Chinese National Promotion Projects of High Technology Industrialization. Our research projects include increasing the types of tumors our HIFU therapy system can treat and increasing patients’ comfort level during treatment. |
| • | ECLIA system research and development. Our ECLIA system research and development focuses on expanding the range of reagent kits for applications that are commonly used in China. For example, we have recently developed reagents to monitor treatments using digoxin, the widely used drug for the treatment of heart failure, and reagents to test for hepatitis. In addition to developing reagents to test for other contagious diseases such as HIV, we are also developing a fully automatic system for large hospitals and a smaller automatic system for private clinics, smaller hospitals and specialized departments in large hospitals. We are also working on improving the response time and efficiency of our ECLIA analyzer and developing an on-site test system for the detection of food pesticide residue, microbial contamination and drug abuse. |
We actively involve scientists and physicians during our research and development process to produce products that provide clinical and economic benefits to hospitals and their patients. In recent years, we have increased our investment in research and development to accelerate the commercialization of new technologies. We expect to continue devoting significant resources to research and development.
59
Table of Contents
| D. | Trend Information |
Other than as disclosed elsewhere in this annual report, we are not aware of any trends, uncertainties, demands, commitments or events for the period from April 1, 2004 to March 31, 2006 that are reasonably likely to have a material adverse effect on our net revenues, income, profitability, liquidity or capital resources, or that caused the disclosed financial information to be not necessarily indicative of future operating results or financial conditions.
| E. | Off-Balance Sheet Arrangements |
We do not, and did not, have any interest in variable interest entities or any other off-balance sheet arrangements that require disclosure.
| F. | Tabular Disclosure of Contractual Obligations |
The following table sets forth our contractual obligations as of March 31, 2006:
| Payment due by period | ||||||||||
Contractual obligations | Less than 1 year | 1-2 years | 2-5 years | More than 5 years | Total | |||||
| (RMB in thousands) | ||||||||||
Operating lease obligations | 1,433 | 1,028 | 553 | — | 3,014 | |||||
Purchase obligations(1) | 38,000 | — | — | — | 38,000 | |||||
Total | 39,433 | 1,028 | 553 | — | 41,014 | |||||
| (1) | Relates to the remaining balance on a contractual obligation to carry out certain construction work for RMB48.0 million (US$6.0 million). |
Other than the contractual obligations and commercial commitments set forth above, we do not have any other long-term debt obligations, operating lease obligations, purchase obligations or other long-term liabilities.
Inflation
Inflation in China has not had a material impact on our results of operations in recent years. According to the National Bureau of Statistics of China, the change in the Consumer Price Index in China was 1.2%, 3.9% and 1.8% in 2003, 2004 and 2005, respectively.
| G. | Safe Harbor |
This annual report on Form 20-F contains forward-looking statements that relate to future events, including our future operating results and conditions, our prospects and our future financial performance and condition. The forward-looking statements are contained principally in the sections entitled “Item 3. Key Information—D. Risk Factors,” “Item 4. Information on the Company” and “Item 5. Operating and Financial Review and Prospects.” These statements involve known and unknown risks, uncertainties and other factors, which may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements.
Forward-looking statements typically are identified by words or phrases such as “may,” “will,” “expect,” “anticipate,” “aim,” “estimate,” “intend,” “plan,” “believe,” “potential,” “continue,” “is/are likely to” or other similar expressions or the negative of these words or expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. We cannot guarantee the accuracy of the forward-looking statements, and you should be aware that our actual results could differ materially from those contained in the forward-looking statements due to a number of factors, including:
| • | our anticipated growth strategies; |
60
Table of Contents
| • | our future business development, results of operations and financial condition; |
| • | our ability to develop and market future generations of our HIFU therapy system and future applications of our ECLIA system; |
| • | the expected market growth for medical devices and supplies in China; |
| • | market acceptance of our technology and products; |
| • | our expectations regarding hospital or patient demand for tumor treatment; |
| • | our ability to expand our production, sales and distribution network and other aspects of our operations; |
| • | our ability to diversify our product range and stay abreast of technological changes; |
| • | competition from other HIFU tumor therapy device manufacturers and alternative methods of tumor therapy; |
| • | competition from other companies that offer IVD systems in China; |
| • | our ability to effectively protect our intellectual property and not infringe on the intellectual property of others; |
| • | our ability to expand our sales and distribution of our HIFU system outside of China; |
| • | our ability to identify and acquire new medical technologies and products; |
| • | changes in the healthcare industry in China, including changes in the healthcare policies and regulations of the PRC government and changes in the healthcare insurance sector in the PRC; and |
| • | fluctuations in general economic and business conditions in China. |
This annual report contains market information related to the medical devices and supplies industry, healthcare insurance, hospitals, the cancer treatment and IVD markets in China. Unlike in the United States, there is limited authoritative data on the healthcare industry in China, particularly on a nationwide basis. For example, there is limited data concerning health and patient trends or the medical devices and supplies industry. In addition, any data that is available may not be current. This annual report contains projections that are based on a number of assumptions. For example, we have made assumptions regarding the growth in the number of tumor patients and the demand for tumor treatment. Our business and marketing plan for the ECLIA system also assumes that small- and medium-sized hospitals in China will be interested in buying an IVD system such as ours. Any or all of our assumptions may turn out to be incorrect. The tumor therapy market may not expand at the rates projected by the market information, or at all. If demand for our HIFU therapy system and ECLIA system does not grow at the projected rates, our business and the market price of our ADSs would suffer. In addition, the complex and changing nature of broad macroeconomic factors subject any projections or estimates relating to the growth prospects or future conditions of our market to significant uncertainties.
The forward-looking statements made in this annual report relate only to events or information as of the date on which the statements are made in this annual report. Except as required by law, we undertake no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this annual report and the documents that we reference in this annual report and have filed as exhibits to the registration statement, of which this annual report is a part, completely and with the understanding that our actual future results may be materially different from what we expect.
61
Table of Contents
ITEM 6. DIRECTORS, SENIOR MANAGEMENTAND EMPLOYEES
| A. | Directors and Senior Management |
Directors and Executive Officers
The following table sets forth information regarding our directors and executive officers as of June 30, 2006.
Name | Age | Position/title | ||
Xiaodong Wu | 50 | Chairman of the board of directors, chief executive officer | ||
Iain Ferguson Bruce(1)(2)(3) | 66 | Director | ||
Cole R. Capener(1) | 51 | Director | ||
Lawrence A. Crum(1)(2)(3) | 65 | Director | ||
Ruyu Du(3) | 71 | Director | ||
Takyung (Sam) Tsang | 35 | Chief financial officer | ||
Minshi (Mike) Shen | 41 | Chief operating officer | ||
Feng (Charles) Zhu | 37 | Vice president |
| (1) | Member of the audit committee. |
| (2) | Member of the compensation committee. |
| (3) | Member of the nomination committee. |
Mr. Xiaodong Wu, our founder, currently serves as our chairman of the board of directors and chief executive officer. Mr. Wu has served on our board of directors since July 2004 and as the chairman of our board of directors since February 2005. Mr. Wu is also a director and the chief executive officer of YDME Beijing, positions he has served since the inception of YDME Beijing in July 1999. Mr. Wu serves on the board of directors of Chengxuan, Beijing Chengxuan, an investment holding company, Beijing Weixiao, a medical technology company, Beijing Jia Chen Hong Biological Technologies Co., Ltd., a medical technology company, and Beijing Wan De Jin Da Industry and Trade Co., Ltd., a property management company. Mr. Wu worked in the foreign investment department of the Jiangxi provincial government from 1990 to 1996. From 1980 to 1989, Mr. Wu worked in Jiangxi Municipal Science and Technology Commission and accumulated extensive experience in assessing and managing hi-tech projects. Mr. Wu is an advisor to Beijing Medical University for technology development projects. Mr. Wu received his bachelor’s degree in Physics from Jiangxi University.
Mr. Iain Ferguson Bruce has served as a member of our board of directors since February 2005. Mr. Bruce is a member of the audit, compensation and nomination committees. Mr. Bruce joined KPMG in Hong Kong in 1964 and was elected to its partnership in 1971. He was the Senior Partner of KPMG from 1991 until his retirement in 1996 and also concurrently served as Chairman of KPMG Asia Pacific from 1993 to 1997. Since 1964, Mr. Bruce has been a member of the Chartered Accountants of Scotland and is a fellow of the Hong Kong Institute of Certified Public Accountants with over 40 years’ experience in the accounting profession. Mr. Bruce is currently an independent non-executive director of Paul Y Engineering Group Limited, a construction and engineering company, Vitasoy International Holdings Ltd., a beverage manufacturing company, Wing On Company International Ltd., a department store operating and real property investment company, and Tencent Holdings Limited, a provider of Internet services and mobile value-added services. All of these companies are listed companies on the Hong Kong Stock Exchange. Mr. Bruce is also a non-executive director of Noble Group Limited, a commodity trading company that is listed on the Singapore Stock Exchange.
Mr. Cole R. Capener has served as a member of our board of directors since June 2005 and is a member of the audit committee. He obtained his law degree, with honors, from The George Washington University National Law Centre in 1981. Thereafter, he practiced law in the New York City office of Shearman & Sterling and later joined the international law firm of Baker & McKenzie in its Beijing office in 1986. Mr. Capener became a partner in Baker & McKenzie in 1989, and his practice consisted of advising multinational corporate clients
62
Table of Contents
doing business in China. In 2001, he left the firm to establish and manage a non-profit, 501(c)(3) corporation, Saving African Families Enterprise, which raises funds to procure antiretroviral medicines for families living with HIV/AIDS in Sub-Saharan African. He currently serves as an independent non-executive director on the board of directors of BMW’s manufacturing joint venture in Shenyang, China, BMW Brilliance Automotive Co. Ltd. He has published a large number of articles on Chinese legal topics. He is admitted to practice law in the states of New York, California and Utah.
Dr. Lawrence A. Crum has served as a member of our Board of Directors since February 2005 and serves as a member of the audit, compensation and nomination committees. Dr. Crum is the director of the Center for Industrial and Medical Ultrasound and a research professor in electrical engineering and bioengineering at the University of Washington in Seattle in the United States. He holds a Ph.D. degree in physics from the University of Ohio, has served as the president of the Acoustical Society of America and the International Commission for Acoustics and holds an honorary doctorate degree from the University of Brussels. He has published over 250 articles in professional journals and holds 15 patents.
Mr. Ruyu Duhas served as a member of our board of directors since June 2005 and is a member of the nomination committee. Mr. Du joined Peking University People’s Hospital in 1959 as a surgeon and served as the president of the hospital from 1983 until 1998, when he retired. Mr. Du currently serves as a professor of surgery at Peking University, and he is also honorary president of Peking University People’s Hospital. Mr. Du is a member of the Standing Committee of the Chinese Medical Association, a director of the Surgery Society of China Medical Association, and the vice chairman of the Chinese Medical Doctor Association. Mr. Du has been the chief editor for the Chinese Journal of Surgery since 1992 and served as deputy chief editor of the Clinical Journal of General Surgery from 1985 to 1992 and of the Chinese Journal of Experimental Surgery from 1996 to 2000. Mr. Du is the author of over 150 articles published in Chinese and international professional journals. Mr. Du has been a member of the Chinese delegation to the International Society of Surgery since 1998. Mr. Du holds his bachelor’s and master’s degrees in medical surgery from Peking University.
Mr. Minshi (Mike) Shen has served as our chief operating officer since February 2005. Prior to joining us, Mr. Shen worked as the director of marketing and business development in the Greater China area at GE Healthcare, and was responsible for marketing, key accounts management, sales training, telemarketing, communication and business development. In addition, Mr. Shen worked at various other positions at GE Healthcare’s China operation from 1999 to 2005. From 1996 to 1999, Mr. Shen worked as a sales and channel manager at Shell (China) Co., Ltd. Mr. Shen received his bachelor’s degree in English from Tsinghua University and a diploma in management from China-Europe International Business School in Shanghai.
Mr. Takyung (Sam) Tsang joined our company as our chief financial officer in January 2005. Mr. Tsang was an advisor to the company for the areas of accounting and finance from February 2004 to December 2004. Prior to joining us, Mr. Tsang worked as a manager of KPMG Hong Kong from March 2001 to January 2004, and also worked as a manager of Ernst & Young China from January 1999 to February 2001. Mr. Tsang is a member of the American Institute of Certified Public Accountants and the Hong Kong Institute of Certified Public Accountants. Mr. Tsang received his bachelor’s degree in business administration from the Chinese University of Hong Kong.
Mr. Feng (Charles) Zhu joined our company as our vice president for business development and investor relationships in January 2005. Prior to joining us, Mr. Zhu worked as a business development manager for GE Healthcare in China from February 2002 to December 2004, responsible for merger and acquisition projects. From May 2000 to February 2002, Mr. Zhu also worked as a business analyst for a Siemens joint venture in China, advising clients on fund raising projects. Mr. Zhu received his bachelor’s degree in English from Foreign Affairs College in China and his master’s degree in business administration from Texas Christian University.
The business address of our directors and executive officers is China Medical Technologies Inc. at No. 24 Yong Chang North Road, Beijing Economic-Technological Development Area, Beijing 100176, People’s Republic of China.
63
Table of Contents
| B. | Compensation |
Compensation of Directors and Executive Officers
For the fiscal year ended March 31, 2006, the aggregate cash compensation to our directors and executive officers was approximately RMB8.8 million (US$1.1 million). The aggregate amount set aside or accrued to provide retirement, pension or other similar benefits for our executive officers and key employees was approximately RMB160,000 (US$20,000) for the fiscal year ended March 31, 2006. If we terminate an executive officer without cause, we are required to pay compensation equal to nine months of the executive officer’s salary. Except for directors who are also executive officers, none of our directors receive benefits upon termination.
Stock Options
Our board of directors has adopted a stock option plan on February 1, 2005. The purpose of the plan is to attract and retain the best available personnel for positions of substantial responsibility, provide additional incentive to employees, directors and consultants and promote the success of our business. Our board of directors believes that our company’s long-term success is dependent upon our ability to attract and retain superior individuals who, by virtue of their ability, experience and qualifications, make important contributions to our business.
We have reserved an aggregate of 30,000,000 of our ordinary shares for issuance under our 2005 stock option plan. As of June 30, 2006, options to purchase an aggregate of 800,000 of our ordinary shares were issued and outstanding.
Termination of Options. Where the option agreement permits the exercise of the options granted for a certain period of time following the recipient’s termination of services with us, or the recipient’s disability or death, the options will terminate to the extent any is not exercised or purchased on the last day of the specified period or the last day of the original term of the options, whichever occurs first.
Administration.Our stock option plan is administered by an administrative committee designated by our board of directors. The administrative committee will determine the provisions, terms and conditions of each option grant, including, but not limited to, the option vesting schedule, repurchase provisions, rights of first refusal, forfeiture provisions, form of payment upon settlement of the award, payment contingencies and satisfaction of any performance criteria.
Vesting Schedule.One-third of the options to be granted under our stock option plan vest one year after a specified vesting commencement date. Another one-third of the options vest 18 months after the specified vesting commencement date. The remaining one-third vest two years after the specified vesting commencement date. The vesting schedule is subject to the optionee continuing to be a service provider of our company on each of such dates.
Option Agreement.Options granted under our stock option plan are evidenced by an option agreement that contains, among other things, provisions concerning exercisability and forfeiture upon termination of employment or consulting arrangements by reason of death, disability or otherwise, as determined by our board. In addition, the option agreement also provides that options granted under each plan are subject to a 180-day lock-up period following the effective date of a registration statement filed by us under the Securities Act, if so requested by us or any representative of the underwriters in connection with any registration of the offering of any of our securities.
Option Exercise. The term of options granted under the 2005 stock option plan may not exceed five years from the date of grant. The consideration to be paid for our ordinary shares upon exercise of an option or purchase of shares underlying the option will be determined by the stock option plan administrator and may include cash, check, ordinary shares, a promissory note, consideration received by us under a cashless exercise program implemented by us in connection with our stock option plan, or any combination of the foregoing methods of payment.
64
Table of Contents
Third-party Acquisition. If a third-party acquires us through the purchase of all or substantially all of our assets, a merger or other business combination, all outstanding stock options or share purchase rights will be assumed or equivalent stock options or share purchase rights will be substituted by the successor corporation or parent or subsidiary of successor corporation. In the event that the successor corporation refuses to assume or substitute for the stock options or share purchase rights, all stock options or share purchase rights will become fully vested and exercisable immediately prior to such transaction and all unexercised options will terminate unless, in either case, the options are assumed by the successor corporation or its parent.
Termination of Plan. Unless terminated earlier, the 2005 stock option plan will expire in 2010. Our board of directors has the authority to amend or terminate our stock option plan subject to shareholder approval to the extent necessary to comply with applicable law and regulations. However, no such action may (i) impair the rights of any optionee unless agreed by the optionee and the stock option plan administrative committee, or (ii) affect the stock option plan administrative committee’s ability to exercise the powers granted to it under our stock option plan.
The following table summarizes the outstanding options that we granted to four of our directors under our 2005 stock option plan as of September 27, 2006:
Name | Number of shares to be issued upon exercise of options | Per share exercise price (in US$) | Date of grant | Date of expiration | |||||
Iain Ferguson Bruce | * | US$ | 1.36 | March 14, 2005 | December 31, 2009 | ||||
Lawrence A. Crum | * | US$ | 1.36 | March 14, 2005 | December 31, 2009 | ||||
Cole R. Capener | * | US$ | 1.50 | August 8, 2005 | December 31, 2009 | ||||
Ruyu Du | * | US$ | 1.50 | August 8, 2005 | December 31, 2009 | ||||
Total | 800,000 | ||||||||
| * | Upon exercise of all options granted, would beneficially own less than 1% of our outstanding ordinary shares. |
These options vest in thirds at each of 12, 18 and 24 months after the grant date.
| C. | Board Practices |
Committees of the Board of Directors
Audit Committee
Our audit committee consists of Mr. Iain Ferguson Bruce, Mr. Cole R. Capener and Dr. Lawrence A. Crum, and is chaired by Mr. Bruce. All of the three directors satisfy the “independence” requirements of the Nasdaq corporate governance rules. The audit committee oversees our accounting and financial reporting processes and the audits of the financial statements of our company. The audit committee is responsible for, among other things:
| • | selecting our independent auditors and pre-approving all auditing and non-auditing services permitted to be performed by our independent auditors; |
| • | reviewing with our independent auditors any audit problems or difficulties and management’s response; |
| • | reviewing and approving all proposed related-party transactions, as defined in Item 404 of Regulation S-K under the Securities Act; |
| • | discussing our annual audited consolidated financial statements with management and our independent auditors; |
| • | reviewing major issues as to the adequacy of our internal controls and any special audit steps adopted in light of material control deficiencies; |
| • | annually reviewing and reassessing the adequacy of our audit committee charter; |
| • | such other matters that are specifically delegated to our audit committee by our board of directors from time to time; |
65
Table of Contents
| • | meeting separately and periodically with management and our internal and independent auditors; and |
| • | reporting regularly to the full board of directors. |
Compensation Committee
Our compensation committee consists of Mr. Iain Ferguson Bruce and Dr. Lawrence A. Crum, both of whom satisfy the “independent” requirements of the Nasdaq corporate governance rules. The committee is chaired by Mr. Bruce. Our compensation committee assists the board in reviewing and approving the compensation structure of our directors and executive officers, including all forms of compensation to be provided to our directors and executive officers. Members of the compensation committee are not prohibited from direct involvement in determining their own compensation. Our chief executive officer may not be present at any committee meeting during which his compensation is deliberated. The compensation committee is responsible for, among other things:
| • | approving and overseeing the compensation package for our executive officers; |
| • | reviewing and making recommendations to the board with respect to the compensation of our directors; |
| • | reviewing and approving corporate goals and objectives relevant to the compensation of our chief executive officer, evaluating the performance of our chief executive officer in light of those goals and objectives, and setting the compensation level of our chief executive officer based on this evaluation; and |
| • | reviewing periodically and making recommendations to the board regarding any long-term incentive compensation or equity plans, programs or similar arrangements, annual bonuses, employee pension and welfare benefit plans. |
Nomination Committee
We established a nomination committee in November 2005. Our nomination committee consists of Mr. Iain Ferguson Bruce, Dr. Lawrence A. Crum and Mr. Ruyu Du, and is chaired by Mr. Bruce. All of the three directors satisfy the “independence” requirements of the Nasdaq corporate governance rules. The nomination committee assists the board of directors in identifying individuals qualified to become our directors and in determining the composition of the board and its committees. The nomination committee is responsible for, among other things:
| • | identifying and recommending to the board nominees for election or re-election to the board, or for appointment to fill any vacancy; and |
| • | reviewing annually with the board the current composition of the board in light of the characteristics of independence, qualification, experience and availability of service to us. |
Duties of Directors
Under Cayman Islands law, our directors have a duty of loyalty to act honestly, in good faith and with a view to our best interests. Our directors also have a duty to exercise the skill they actually possess and such care and diligence that a reasonably prudent person would exercise in comparable circumstances. In fulfilling their duty of care to us, our directors must ensure compliance with our memorandum and articles of association, as amended and re-stated from time to time. A shareholder has the right to seek damages if a duty owed by our directors is breached.
The functions and powers of our board of directors include, among others:
| • | convening shareholders’ annual general meetings and reporting its work to shareholders at such meetings; |
| • | declaring dividends and distributions; |
66
Table of Contents
| • | appointing officers and determining the term of office of officers; |
| • | exercising the borrowing powers of our company and mortgaging the property of our company; and |
| • | approving the transfer of shares of our company, including the registering of such shares in our share register. |
Terms of Directors and Executive Officers
All directors hold office until their successors have been duly elected and qualified. Officers are elected by and serve at the discretion of the board of directors. Currently, directors are not subject to a term of office and hold office until such time as they are removed from office by special resolution or the unanimous written resolution of all shareholders. If a director has been appointed by Chengxuan or Golden Meditech, upon written notice to us, such director may be removed and replaced by its respective nominator, at any time for any reason.
With the expiration of the directors’ terms under our articles of association, elections for directors will be held each year on the date of annual general meeting of shareholders. A director will be removed from office automatically if, among other things, the director (i) becomes bankrupt or makes any arrangement or composition with his creditors; or (ii) dies or is found by our company to be or becomes of unsound mind.
| D. | Employees |
Employees
We had 78, 87, 137 and 265 employees as of March 31, 2003, 2004, 2005 and 2006 respectively. As of June 30, 2006, we had 283 employees. The following table sets forth the number of our employees categorized by function as of June 30, 2006.
As of June 30, 2006 | Percentage of Total (%) | |||
Manufacturing and services | 101 | 35.6 | ||
Research and development | 48 | 17.0 | ||
General and administration | 48 | 17.0 | ||
Marketing and sales(1) | 50 | 17.7 | ||
Customer support and service | 36 | 12.7 | ||
Total | 283 | 100.0 | ||
| (1) | Includes 33 employees focusing on the sale of our HIFU therapy systems and 17 employees focusing on the sales of our ECLIA systems. |
From time to time, we also employ independent contractors to support our marketing and sales and clinical support and research. We plan to hire additional employees for marketing and sales, customer service and manufacturing and assembly as we grow our business. None of our employees is represented by a labor union. We consider our relationship with our employees to be good.
In accordance with applicable regulations in the PRC, we participate in a pension contribution plan, a medical insurance plan, an unemployment insurance plan and a personal injury insurance plan for our employees. We have made adequate provisions in accordance with applicable regulations, which require us to contribute amounts equal to 20%, 10%, 1.5% and 0.8%, respectively, of our employees’ aggregate base salaries to these statutory plans.
Also, in accordance with PRC regulations, we contribute amounts equal to 8%, 4% and 0.8% of our employees’ aggregate base salaries towards a housing fund, a supplemental medical insurance fund and a maternity fund, respectively.
67
Table of Contents
| E. | Share Ownership |
The following table sets forth information with respect to the beneficial ownership of our ordinary shares as of September 27, 2006, the latest practicable date, by:
| • | each of our directors and executive officers; and |
| • | each person known to us to own beneficially more than 5.0% of our ordinary shares. |
| Ordinary shares beneficially owned(1) | ||||
| Number | % | |||
Directors and executive officers: | ||||
Xiaodong Wu(2)(4) | 71,420,001 | 26.1 | ||
Iain Ferguson Bruce | * | * | ||
Lawrence A. Crum | * | * | ||
Cole R. Capener | * | * | ||
Ruyu Du | * | * | ||
All directors and executive officers as a group(3) | 71,819,999 | 26.2 | ||
Principal shareholders: | ||||
Chengxuan International Ltd.(4) | 71,420,001 | 26.1 | ||
Golden Meditech (BVI) Company Ltd.(5) | 27,100,000 | 9.9 | ||
| * | Indicates less than 1%. |
| (1) | Beneficial ownership is determined in accordance with Rule 13d-3 of the General Rules and Regulations under the Securities Exchange Act of 1934, as amended, and includes shares as to which the indicated person holds voting or investment power with respect to the securities. |
| (2) | Mr. Wu holds his shares through Chengxuan International Ltd. |
| (3) | Includes ordinary shares issuable upon exercise of options beneficially held by all of our directors and executive officers as a group. |
| (4) | Chengxuan International Ltd., a British Virgin Islands company, is owned by Mr. Xiaodong Wu. The address for Chengxuan is The Mill Mall, PO Box 92, Road Town, Tortola, British Virgin Islands. |
| (5) | Golden Meditech (BVI) Company Ltd., a British Virgin Islands company, is a wholly owned subsidiary of Golden Meditech Company Limited, a public company listed on the Hong Kong Stock Exchange. The address for Golden Meditech is PO Box 957, Offshore Incorporations Centre, Road Town, Tortola, British Virgin Islands. |
As of September 27, 2006, other than the 64.0% of our outstanding ordinary shares underlying the outstanding ADSs which were held by our custodian, Citibank Hong Kong Branch, on behalf of Citibank, N.A., the depositary, none of our ordinary shares were held in the United States. Our ordinary shares underlying the ADSs quoted on the Nasdaq National Market are held in Hong Kong by the custodian, Citibank, N.A., Hong Kong Branch, on behalf of Citibank, N.A., the depositary.
None of our shareholders has different voting rights from other shareholders. We are not aware of any arrangement that may, at a subsequent date, result in a change of control of our company.
ITEM 7. MAJOR SHAREHOLDERSAND RELATED PARTY TRANSACTIONS
A. Major Shareholders
Please refer to “Item 6. Directors, Senior Management and Employees—Share Ownership.”
68
Table of Contents
B. Related Party Transactions
After the completion of our initial public offering on August 15, 2005, we adopted an audit committee charter, which requires that the audit committee review all related party transactions on an ongoing basis and all such transactions be approved by the committee. Set forth below is a description of all of our related party transactions for the fiscal years ended March 31, 2004, 2005 and 2006.
Transactions with GE
GE became a shareholder of YDME Beijing and a distributor of our HIFU therapy system in April 2003. In January 2005, GE became our shareholder. For the fiscal years ended March 31, 2004, 2005 and 2006, sales of our HIFU therapy system to GE amounted to RMB12.0 million, RMB4.1 million and RMB 3.9 million (US $0.5 million), respectively, which accounted for 12.0%, 1.9% and 1.1% of our net revenues, respectively. Sales of our HIFU therapy system to GE were generally at prices lower than those charged to third parties.
In addition, we purchased the ultrasound imaging system, one of the major components of our HIFU therapy system, from GE. Under the exclusive supply agreement between GE and us, as long as GE holds 5% or more of our ordinary shares, GE is entitled to be the sole supplier of the ultrasound imaging system. On June 21, 2006, GE’s ownership interest in our company fell below 5% of our outstanding shares and ceased to be a party to the shareholders’ agreement. For the fiscal years ended March 31, 2004, our purchases of the ultrasound imaging system from GE amounted to RMB6.8 million. We purchased GE’s ultrasound imaging system through an import company for the fiscal years ended March 31, 2005 and 2006.
In April 2004, we entered into a service agreement with GE under which we agreed to provide repair services for medical equipment GE sold to its customers in China. Under the service agreement, GE agreed to reimburse all expenses we incur in connection with our repair services. In addition, we also charge GE a service fee equal to 10% of expenses we incurred in connection with our repair services. We generated repair service income in the amount of RMB 3,000 and RMB 145,000 (US $18,000) under the service agreement for the fiscal years ended March 31, 2005 and 2006 respectively. The service agreement expired, and we discontinued providing these services on December 31, 2005.
Transactions with Beijing Weixiao
In February 2004, we signed a purchase agreement with Beijing Weixiao, our affiliate company that is controlled by Mr. Xiaodong Wu, the chairman of our board of directors and our chief executive officer, for the purchase of a building to be used as our office, manufacturing facilities and research and development center for RMB64.8 million. A prepayment of RMB60.0 million was made to Beijing Weixiao in March 2003 and the remaining RMB4.8 million was paid in April 2004.
In August 2004, we acquired the ECLIA technology from Beijing Weixiao. Under the acquisition agreement, we agreed to pay Beijing Weixiao a total purchase price of RMB250.0 million before September 30, 2005 for the ECLIA technology, related inventories and a noncompete commitment. We made payments of RMB80.0 million, RMB12.0 million, RMB8.0 million, RMB50.0 million and RMB100.0 million in September 2004, December 2004, February 2005, March 2005 and September 2005, respectively.
Transactions with Chengxuan
Chengxuan International Ltd., or Chengxuan, is one of our shareholders and controlled by Mr. Xiaodong Wu. Chengxuan paid RMB2.3 million in cash on our behalf during the fiscal year ended March 31, 2005 to settle expenses we incurred in connection with the preparation of our initial public offering and our operating activities. These amounts were unsecured, non-interest bearing and have no fixed terms of repayment and was repaid during the fiscal year ended March 31, 2006.
69
Table of Contents
Secondary Offering
On March 22, 2006, we completed a secondary public offering of 57,500,000 shares held by certain existing shareholders. In this offering, the selling shareholders received all net proceeds from the sale of shares and all of the expenses, including underwriting discounts and commissions, related to this offering were shared by the selling shareholders. During the year ended March 31, 2006, we received RMB10.4 million (US$1.3 million) from the selling shareholders to pay the expenses on behalf of them and the amount was recorded in other payables. In March 2006, RMB802,000 (US$100,000) was paid and the outstanding balance as of March 31, 2006 was RMB9.6 million (US$1.2 million). Upon the completion of the secondary offering, we received RMB5.9 million (US$0.7 million) from certain selling shareholders for our arrangement of the secondary offering.
Shareholder’s Agreement
In connection with the March 2003 private placement, YDME Beijing entered into a shareholders’ agreement with all of its nine shareholders. This agreement provided for certain matters relating to the management of YDME Beijing. Under this agreement, the board of directors of YDME Beijing would consist of nine directors. Each of Beijing Chengxuan, PUPH and Golden Meditech was entitled to designate two directors, and Beijing Boda Technology Investment and Development Co., Ltd. was entitled to designate one director. In addition, Beijing Chengxuan was entitled to nominate two independent directors, and a representative of GE was entitled to attend all board meetings in a non-voting capacity.
In May 2004, in connection with Beijing Chengxuan’s acquisition of ownership interests in YDME Beijing held by certain minority shareholders, the remaining shareholders of YDME Beijing, Beijing Chengxuan, Golden Meditech and GE, adopted a revised memorandum and articles of association, which superseded the March 2003 shareholders’ agreement. Under the revised memorandum and articles of association, which became effective as of June 2004, the board of directors of YDME Beijing would consist of three directors. Each of Beijing Chengxuan, Golden Meditech and GE was entitled to designate one director. As a result, Golden Meditech and GE, which held equity interests of 25% and 15%, respectively, in YDME Beijing, effectively had equal voting power to make significant decisions expected to be made in the ordinary course of YDME Beijing’s business operations. The shareholders believed that the delegation of the decision-making authority to the board was necessary to protect the interests of minority shareholders and to enhance corporate governance.
In January 2005, we and Chengxuan, Golden Meditech and GE entered into an equity transfer agreement by which YDME Beijing was restructured as our wholly owned subsidiary. In January 2005, we and Chengxuan, Golden Meditech and GE, entered into a shareholders’ agreement. In June 2005, we and these shareholders entered into a new shareholders’ agreement, which replaced the January 2005 shareholders’ agreement upon completion of our A. Under the June 2005 shareholders’ agreement:
| • | A representative of GE may attend all of our board meetings in a non-voting capacity. |
| • | GE and Golden Meditech and their respective affiliates will have complete access to, and full use of, the clinical data of our subsidiary, YDME Beijing, relating to clinical cases from hospitals, other medical institutions and medical professionals using our HIFU therapy system in connection with their business, provided that we have first obtained the necessary consent. Such clinical data include hospital survey information, medical records, test reports and imaging records before and after treatment using our HIFU therapy system. |
| • | If any of Chengxuan, Golden Meditech and GE is offered a business opportunity that is primarily based in China and that competes with our HIFU business, it will provide us with notice of its intention to pursue such business opportunity and use reasonable efforts to enable us to participate in such project if we express an interest in doing so. |
70
Table of Contents
| • | We will provide GE with notice of our intention to undertake any HIFU-related project, including, among other things: |
| • | any project relating to any joint venture between us and InSightec; |
| • | any expansion of our existing HIFU production facilities; or |
| • | any development of new HIFU facilities anywhere in the world. |
In addition, we will use reasonable efforts to enable GE to participate in such project if GE expresses an interest in doing so.
| • | We have also granted GE a right of first refusal to act as the sole and exclusive distributor of our HIFU therapy system outside China. In the event that we sell or otherwise transfer all or substantially all of our assets or HIFU business, or as a result of a merger, we are no longer a surviving company, we will cause the transferee or the surviving company of the merger, as the case may be, to honor GE’s right of first refusal. We will also cause the transferee or the surviving company to continue purchasing the ultrasound imaging system from GE. |
These rights and obligations may be terminated by written agreement among Chengxuan, Golden Meditech and GE. If any of Chengxuan’s, Golden Meditech’s or GE’s ownership interest in our company falls below 5% of our outstanding shares, calculated on a fully diluted basis, such shareholder will cease to be a party to the June 2005 shareholders’ agreement and will no longer have any rights or obligations under that agreement.
On June 21, 2006, GE’s ownership interest in our company fell below 5% of our outstanding shares and ceased to be a party to the shareholders’ agreement.
Equity Incentive Plan
See Item 6.B., “—Stock Options.”
C. Interests of Experts and Counsel
Not applicable.
A. Consolidated Statements and Other Financial Information
We have appended consolidated financial statements filed as part of this annual report.
Legal and Administrative Proceedings
Neither we nor any of our directors and executive officers are a party to any material legal or administrative proceedings, and we are not aware of threatened material legal or administrative proceedings against us. We may from time to time become a party to various legal or administrative proceedings arising in the ordinary course of our business.
Dividend Policy
Dividends of RMB39.2 million in respect of the calendar years 2001 and 2002 and of RMB28.2 million in respect of the calendar year 2003 were approved by our shareholders in February and March 2004, respectively, and were subsequently paid. We have not declared any cash dividends since March 2004 and do not have any present plan to pay any cash dividends on our ordinary shares in the foreseeable future. We currently intend to retain most, if not all, of our available funds and any future earnings to operate and expand our business.
71
Table of Contents
We rely principally on dividends paid to us by YDME Beijing, our wholly owned subsidiary in China, for our cash requirements, including the funds necessary to pay dividends and other cash distributions to our shareholders, service any debt we may incur and pay our operating expenses. In China, the payment of dividends is subject to limitations. PRC regulations currently permit payment of dividends only out of accumulated profits as determined in accordance with PRC accounting standards and regulations. Under current PRC laws, regulations and accounting standards, YDME Beijing is required to allocate at least 10% of its after-tax profits to its general reserves. Allocation to these reserves is not required after these reserves have reached 50% of the registered capital of YDME Beijing. In addition, at the discretion of its board of directors, YDME Beijing may allocate a portion of its after-tax profits to its enterprise expansion funds and staff welfare and bonus funds. Staff welfare and bonus funds may not be distributed to equity owners.
Our board of directors has complete discretion as to whether we will distribute dividends in the future. Even if our board of directors decides to distribute dividends, the form, frequency and amount of our dividends will depend upon our future operations and earnings, capital requirements and surplus, financial condition, contractual restrictions and other factors that our board of directors may deem relevant. Any dividend we declare will be paid to the holders of ADSs, subject to the terms of the deposit agreement and applicable laws, to the same extent as holders of our ordinary shares, less the fees and expenses payable under the deposit agreement. Any dividend we declare will be distributed by the depositary bank to the holders of our ADSs. Cash dividends on our ordinary shares, if any, will be paid in U.S. dollars. See “Description of American Depositary Shares.”
B. Significant Changes
We have not experienced any significant changes since the date of our audited consolidated financial statements included in this annual report.
A. Offering and Listing Details
Our ADSs, each representing ten of our ordinary shares, have been listed on the Nasdaq National Market since August 10, 2005 under the symbol “CMED.” On March 22, 2006, we completed a follow-on offering of 5,750,000 ADSs, which were sold by certain of our selling shareholders.
For the period from August 10, 2005 to March 31, 2006, the trading price of our ADSs on Nasdaq National Market ranged from US$14.95 to US$44.93 per ADS. For the period from April 1, 2006 to September 27, 2006, the trading price of our ADSs on Nasdaq National Market ranged from US$18.30 to US$30.92 per ADS.
72
Table of Contents
The following table provides the high and low trading prices for our ADSs on the Nasdaq National Market for (1) the third and fourth quarters in 2005, and the first and second quarters in 2006, and (2) each of the months since February 2006.
Sales Price | ||||
High | Low | |||
| Quarterly High and Low | ||||
Third Quarter 2005 (from August 10) | US$23.99 | US$14.95 | ||
Fourth Quarter 2005 | US$42.60 | US$18.69 | ||
First Quarter 2006 | US$44.93 | US$25.56 | ||
Second Quarter 2006 | US$30.92 | US$18.30 | ||
| Monthly Highs and Lows | ||||
March 2006 | US$35.07 | US$25.56 | ||
April 2006 | US$30.92 | US$22.35 | ||
May 2006 | US$28.24 | US$20.50 | ||
June 2006 | US$25.84 | US$18.30 | ||
July 2006 | US$22.72 | US$18.50 | ||
August 2006 | US$24.73 | US$21.15 | ||
September 2006 (through September 27) | US$23.20 | US$19.94 | ||
B. Plan of Distribution
Not applicable.
C. Markets
Our ADSs, each representing ten of our ordinary shares, have been listed on the Nasdaq National Market since August 10, 2005 under the symbol “CMED.”
D. Selling Shareholders
Not applicable.
E. Dilution
Not applicable.
F. Expenses of the Issue
Not applicable.
ITEM 10. ADDITIONAL INFORMATION
A. Share Capital
As of the date of this annual report on Form 20-F, our authorized capital consists of 500,000,000 ordinary shares, par value of US$0.10 each and 200,000,001 ordinary shares issued and outstanding.
Our predecessor and directly wholly owned subsidiary YDME Beijing was co-founded in July 1999 by Beijing Chengxuan (with 70% equity interest) and PUPH (with 30% equity interest). In November 2000, Beijing Chengxuan purchased 20% of PUPH’s ownership interest in YDME Beijing for RMB10 million and
73
Table of Contents
subsequently sold 32% of its ownership interests in YDME Beijing to five additional investors for RMB24.7 million. In March 2003, through a private placement, YDME Beijing issued 16,775,000 and 10,065,000 ordinary shares to Golden Meditech and GE, for 25% and 15% ownership interests, respectively, in YDME Beijing. In June 2004, Beijing Chengxuan acquired 25.2% ownership interests of YDME Beijing from PUPH and the group of five investors for RMB42.1 million (US$5.2 million), after which the ownership interests of Beijing Chengxuan, Golden Meditech and GE in YDME Beijing became 60%, 25% and 15%, respectively. In June 2004, Chengxuan acquired all of Beijing Chengxuan’s ownership interests in YDME Beijing for US$8.5 million. In November 2004, GE purchased 4.99% ownership interests of YDME Beijing from Chengxuan for US$1.1 million, after which the ownership interests of Chengxuan, Golden Meditech and GE in YDME Beijing became 55.01%, 25% and 19.99%, respectively. We created a holding company structure by incorporating China Medical Technologies, Inc. in the Cayman Islands in July 2004. In January 2005, through a corporate reorganization, YDME Beijing became our wholly owned subsidiary. As part of this corporate reorganization, Chengxuan, GE and Golden Meditech subscribed for 110,020,000, 39,980,000 and 50,000,000 of our ordinary shares, respectively, in exchange for their respective outstanding ownership interests in YDME Beijing.
In connection with its purchase of Beijing Chengxuan’s ownership interests in YDME Beijing, in July 2004, Chengxuan issued to Green Wall a US$8.5 million exchangeable note with an annual interest rate of 10%. The term of the note was one year, and the holder of the note had the right, at any time, to exchange the note for the ordinary shares in our company representing 10% ownership interests of YDME Beijing. In February 2005, Green Wall resold 30% of the exchangeable note to Asset Managers for US$5.0 million and 50% of the exchangeable note to Neon Liberty for US$8.5 million. The note was automatically exchanged into 20,000,000 of our ordinary shares held by Chengxuan upon completion of our initial public offering. As a result, upon completion of our initial public offering, Green Wall, Asset Managers and Neon Liberty held 4,000,001, 6,000,000 and 9,999,999 shares of our ordinary shares, respectively, some of which they are selling in this offering.
In February 2005, we adopted our 2005 stock option plan and reserved 30,000,000 ordinary shares for issuance under our 2005 stock option plan. We have granted options to four of our directors. As of December 31, 2005, options to purchase an aggregate of 800,000 ordinary shares of our company were outstanding. See “Management—Stock options.”
On August 15, 2005, we completed our initial public offering, which involved the sale by us of 6,400,000 of our ADSs, representing 64,000,000 of our ordinary shares at an initial public offering price of US$15.00 per ADS. On August 29, 2005, we sold an additional 960,000 ADSs representing 9,600,000 ordinary shares at the initial public offering price pursuant to the underwriter’s exercise of its over-allotment option.
B. Memorandum and Articles of Association
We incorporate by reference into this annual report the description of our amended and restated memorandum of association contained in our F-1 registration statement (File No. 333-132214), as amended, initially filed with the Commission on July 15, 2005. Our shareholders adopted our amended and restated memorandum and articles of association by unanimous resolutions in June 2004.
C. Material Contracts
We have not entered into any material contracts other than in the ordinary course of business and other than those described in “Item 4. Information on the Company” or elsewhere in this annual report on Form 20-F.
D. Exchange Controls
Foreign Currency Exchange
Foreign currency exchange regulation in China is primarily governed by the following rules:
| • | Foreign Currency Administration Rules (1996), as amended, or the Exchange Rules; and |
| • | Administration Rules of the Settlement, Sale and Payment of Foreign Exchange (1996), or the Administration Rules; |
74
Table of Contents
Under the Exchange Rules, the Renminbi is convertible for current account items, including the distribution of dividends, interest payments, trade and service-related foreign exchange transactions. Conversion of Renminbi for capital account items, such as direct investment, loan, security investment and repatriation of investment, however, is still subject to the approval of the PRC State Administration of Foreign Exchange, or SAFE.
Under the Administration Rules, foreign-invested enterprises may only buy, sell and/or remit foreign currencies at those banks authorized to conduct foreign exchange business after providing valid commercial documents and, in the case of capital account item transactions, obtaining approval from the SAFE. Capital investments by foreign-invested enterprises outside of China are also subject to limitations, which include approvals by the Ministry of Commerce, the SAFE and the State Reform and Development Commission.
E. Taxation
Cayman Islands Taxation
According to Walkers, our Cayman Islands counsel, the Cayman Islands currently levies no taxes on individuals or corporations based upon profits, income, gains or appreciation and there is no taxation in the nature of inheritance tax or estate duty. There are no other taxes likely to be material to us levied by the Government of the Cayman Islands except for stamp duties which may be applicable on instruments executed in, or brought within, the jurisdiction of the Cayman Islands. The Cayman Islands is not party to any double tax treaties. There are no exchange control regulations or currency restrictions in the Cayman Islands.
United States Federal Income Taxation
The following discussion describes certain U.S. federal income tax consequences to U.S. Holders (defined below) under present law of an investment in the ADSs or ordinary shares. This summary applies only to U.S. Holders that hold the ADSs or ordinary shares as capital assets and that have the U.S. dollar as their functional currency. This discussion is based on the Internal Revenue Code of 1986, as amended (the “Code”) as in effect on the date of this annual report on Form 20-F and on U.S. Treasury regulations in effect or, in some cases, proposed, as of the date of this annual report on Form 20-F, as well as judicial and administrative interpretations thereof available on or before such date. All of the foregoing authorities are subject to change, which change could apply retroactively and could affect the tax consequences described below.
The following discussion does not address the tax consequences to any particular investor or to persons in special tax situations such as:
| • | banks; |
| • | certain financial institutions; |
| • | insurance companies; |
| • | broker dealers; |
| • | U.S. expatriates; |
| • | traders that elect to mark to market; |
| • | tax-exempt entities; |
| • | persons liable for alternative minimum tax; |
| • | persons holding an ADS or ordinary share as part of a straddle, hedging, conversion or integrated transaction; |
| • | persons that actually or constructively own 10.0% or more of our voting stock; or |
| • | persons holding ADSs or ordinary shares through partnerships or other pass-through entities. |
75
Table of Contents
The discussion below of the U.S. federal income tax consequences to “U.S. Holders” will apply if you are a beneficial owner of ADSs or ordinary shares and you are, for U.S. federal income tax purposes,
| • | an individual citizen or resident of the United States; |
| • | a corporation (or other entity taxable as a corporation for U.S. federal income tax purposes) organized under the laws of the United States, any State thereof or the District of Columbia; |
| • | an estate whose income is subject to U.S. federal income taxation regardless of its source; or |
| • | a trust that (1) is subject to the primary supervision of a court within the United States and the control of one or more U.S. persons or (2) has a valid election in effect under applicable U.S. Treasury regulations to be treated as a U.S. person. |
If you are a partner in a partnership or other entity taxable as a partnership that holds ADSs or ordinary shares, your tax treatment generally will depend on your status and the activities of the partnership, and you should consult your tax advisors.
The discussion below assumes that the representations contained in the deposit agreement are true and that the obligations in the deposit agreement and any related agreement will be complied with in accordance with their terms. If you hold ADSs, you should be treated as the holder of the underlying ordinary shares represented by those ADSs for U.S. federal income tax purposes. Accordingly, deposits or withdrawals of ordinary shares for ADSs should not be subject to U.S. federal income tax.
The U.S. Treasury has expressed concerns that intermediaries in the chain of ownership between the holder of an ADS and the issuer of the security underlying the ADS may be taking actions that are inconsistent with the claiming, by U.S. Holders of ADSs, of foreign tax credits for U.S. federal income tax purposes. Such actions would also be inconsistent with the claiming of the reduced rate of tax applicable to dividends received by certain non-corporate U.S. Holders, as described below. Accordingly, the availability of the reduced tax rate for dividends received by certain non-corporate U.S. Holders could be affected by future actions that may be taken by the U.S. Treasury or intermediaries in the chain of ownership between the holder of an ADS and our company.
The discussion below does not contain a detailed description of all the U.S. federal income tax consequences to you in light of your particular circumstances and does not address the effects of any state, local or non-United States tax laws.If you are considering the purchase, ownership or disposition of our ADSs or ordinary shares, you should consult your own tax advisors concerning the U.S. federal income tax consequences to you in light of your particular situation as well as any consequences arising under the laws of any other taxing jurisdiction.
Taxation of Dividends and Other Distributions on the ADSs or Ordinary Shares
Subject to the passive foreign investment company rules discussed below, the gross amount of all our distributions to you with respect to the ADSs or ordinary shares generally will be included in your gross income as foreign source dividend income on the date of receipt by the depositary, in the case of ADSs, or by you, in the case of ordinary shares, but only to the extent that the distribution is paid out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). The dividends will not be eligible for the dividends-received deduction allowed to corporations in respect of dividends received from other U.S. corporations.
With respect to non-corporate U.S. Holders including individual U.S. Holders, for taxable years beginning before January 1, 2011, dividends may constitute “qualified dividend income” and thus be taxed at the lower applicable capital gains rate, provided that (1) the ADSs or ordinary shares are readily tradable on an established securities market in the United States, (2) we are not a passive foreign investment company (as discussed below) for either our taxable year in which the dividend was paid or the preceding taxable year, and (3) certain holding
76
Table of Contents
period requirements are met. Under Internal Revenue Service authority, common or ordinary shares, or ADSs representing such shares, are considered for the purpose of clause (1) above to be readily tradable on an established securities market in the United States if they are listed on the Nasdaq National Market, as our ADSs are. We do not believe, however, that dividends that we pay on ordinary shares that are not backed by ADSs currently meet the conditions to be taxed as qualified dividend income. There can be no assurance that our ADSs will be considered readily tradable on an established securities market in the United States in later years. Non-corporate U.S. Holders that elect to treat the dividend income as “investment income” pursuant to Section 163(d)(4) of the Code will not be eligible for the reduced rates of taxation. In addition, the rate reduction will not apply to dividends if the recipient of a dividend is obligated to make related payments with respect to positions in substantially similar or related property. This disallowance applies even if the minimum holding period has been met. You should consult your tax advisors regarding the availability of the lower rate for dividends paid with respect to our ADSs or ordinary shares.
To the extent that the amount of any distribution exceeds our current and accumulated earnings and profits, it will be treated first as a tax-free return of your tax basis in your ADSs or ordinary shares, and to the extent the amount of the distribution exceeds your tax basis, the excess will be taxed as capital gain. We do not intend to calculate our earnings and profits under U.S. federal income tax principles. Therefore, a U.S. Holder should expect that a distribution will generally be treated as a dividend.
Taxation of Disposition of Shares
Subject to the passive foreign investment company rules discussed below, you will recognize taxable gain or loss on any sale, exchange or other taxable disposition of an ADS or ordinary share equal to the difference between the amount realized for the ADS or ordinary share and your tax basis in the ADS or ordinary share. The gain or loss generally will be capital gain or loss. If you are a non-corporate U.S. Holder, including an individual U.S. Holder, who has held the ADS or ordinary share for more than one year, you will be eligible for reduced tax rates. The deductibility of capital losses is subject to limitations. Any such gain or loss that you recognize will generally be treated as U.S. source income or loss for foreign tax credit limitation purposes.
Passive Foreign Investment Company
A non-U.S. corporation is considered a passive foreign investment company (“PFIC”) for any taxable year if either
| • | at least 75% of its gross income is passive income, or |
| • | at least 50% of the value of its assets (based on an average of the quarterly values of the assets during a taxable year) is attributable to assets that produce or are held for the production of passive income. |
For this purpose, cash is a passive asset and passive income generally includes dividends, interest, royalties and rents (other than royalties and rents derived in the active conduct of a trade or business and not derived from a related person). We will be treated as owning our proportionate share of the assets and earning our proportionate share of the income of any other corporation in which we own, directly or indirectly, 25% or more (by value) of the stock.
Based on the composition of our income and valuation of our assets, including goodwill, we believe that we were not a PFIC for our taxable year ended March 31, 2006, and we do not expect to be a PFIC for our current taxable year for U.S. federal income tax purposes. We must make a separate determination each year as to whether we are a PFIC. As a result, our PFIC status may change. In particular, our PFIC status may be determined in large part based on the market price of our ADSs and ordinary shares, which is likely to fluctuate (and may fluctuate considerably given that market prices of technology companies have been especially volatile). Accordingly, fluctuations in the market price of the ADSs and ordinary shares may result in our being a PFIC for any year. Thus, there can be no assurance that we will not be considered a PFIC for our current taxable year or any future taxable year. If we are a PFIC for any year during which you hold ADSs or ordinary shares, we generally will continue to be treated as a PFIC for all succeeding years during which you hold ADSs or ordinary shares.
77
Table of Contents
If we are a PFIC for any taxable year during which you hold ADSs or ordinary shares, you will be subject to special tax rules with respect to any “excess distribution” that you receive and any gain you realize from a sale or other disposition (including a pledge) of the ADSs or ordinary shares, unless you make a “mark-to-market” election as discussed below. Distributions you receive in a taxable year that are greater than 125% of the average annual distributions you received during the shorter of the three preceding taxable years or your holding period for the ADSs or ordinary shares will be treated as excess distributions. Under these special tax rules: the excess distribution or gain will be allocated ratably over your holding period for the ADSs or ordinary shares, the amount allocated to the current taxable year, and any taxable year prior to the first taxable year in which we became a PFIC, will be treated as ordinary income, and the amount allocated to each other year will be subject to the highest tax rate in effect for that year and the interest charge generally applicable to underpayments of tax will be imposed on the resulting tax attributable to each such year.
The tax liability for amounts allocated to years prior to the year of disposition or “excess distribution” cannot be offset by any net operating losses for such years, and gains (but not losses) realized on the sale of the ADSs or ordinary shares cannot be treated as capital, even if you hold the ADSs or ordinary shares as capital assets.
Alternatively, a U.S. Holder of “marketable stock” (as defined below) in a PFIC may make a mark-to-market election for such stock of a PFIC to elect out of the tax treatment discussed in the two preceding paragraphs. If you make a mark-to-market election for the ADSs or ordinary shares, you will include in income each year an amount equal to the excess, if any, of the fair market value of the ADSs or ordinary shares as of the close of your taxable year over your adjusted basis in such ADSs or ordinary shares. You are allowed a deduction for the excess, if any, of the adjusted basis of the ADSs or ordinary shares over their fair market value as of the close of the taxable year. However, deductions are allowable only to the extent of any net mark-to-market gains on the ADSs or ordinary shares included in your income for prior taxable years. Amounts included in your income under a mark-to-market election, as well as gain on the actual sale or other disposition of the ADSs or ordinary shares, are treated as ordinary income. Ordinary loss treatment also applies to the deductible portion of any mark-to-market loss on the ADSs or ordinary shares, as well as to any loss realized on the actual sale or disposition of the ADSs or ordinary shares, to the extent that the amount of such loss does not exceed the net mark-to-market gains previously included for such ADSs or ordinary shares. Your basis in the ADSs or ordinary shares will be adjusted to reflect any such income or loss amounts. The tax rules that apply to distributions by corporations that are not PFICs would apply to distributions by us, except that the preferential dividend rates with respect to “qualified dividend income” would not apply.
The mark-to-market election is available only for “marketable stock,” which is stock that is traded in other than de minimus quantities on at least 15 days during each calendar quarter on a qualified exchange, including the Nasdaq National Market, or other market, as defined in applicable U.S. Treasury regulations. The ADSs are listed on the Nasdaq National Market and, consequently, if you are a holder of ADSs the mark-to-market election would be available to you were we to be or become a PFIC, provided the ADSs were traded in sufficient quantities. If you make a mark-to-market election it will be effective for the taxable year for which the election is made and all subsequent taxable years unless the ADSs no longer qualify as “marketable stock” or the Internal Revenue Service consents to the revocation of the election.
Alternatively, you can sometimes avoid the rules described above by electing to treat us as a “qualified electing fund” under Section 1295 of the Code. This option is not available to you because we do not intend to comply with the requirements necessary to permit you to make this election.
If you hold ADSs or ordinary shares in any year in which we are a PFIC, you will be required to file Internal Revenue Service Form 8621 regarding distributions received on the ADSs or ordinary shares and any gain realized on the disposition of the ADSs or ordinary shares. You are urged to consult your tax advisor regarding the application of the PFIC rules to your investment in ADSs or ordinary shares.
78
Table of Contents
Information Reporting and Backup Withholding
Dividend payments with respect to ADSs or ordinary shares and proceeds from the sale, exchange or redemption of ADSs or ordinary shares may be subject to information reporting to the Internal Revenue Service and possible U.S. backup withholding at a current rate of 28%. Backup withholding will not apply, however, to a U.S. Holder who furnishes a correct taxpayer identification number and makes any other required certification or who is otherwise exempt from backup withholding. U.S. Holders who are required to establish their exempt status generally must provide such certification on Internal Revenue Service Form W-9. U.S. Holders should consult their tax advisors regarding the application of the U.S. information reporting and backup withholding rules.
Backup withholding is not an additional tax. Amounts withheld as backup withholding may be credited against your U.S. federal income tax liability, and you may obtain a refund of any excess amounts withheld under the backup withholding rules by filing the appropriate claim for refund with the Internal Revenue Service and furnishing any required information.
| F. | Dividends and Paying Agents |
Not applicable.
| G. | Statement by Experts |
Not applicable.
| H. | Documents on Display |
We have previously filed with the Commission our registration statement on Form F-1, as amended.
We are subject to the periodic reporting and other informational requirements of the Securities Exchange Act of 1934, as amended, or the Exchange Act. Under the Exchange Act, we are required to file reports and other information with the Securities and Exchange Commission. Specifically, we are required to file annually a Form 20-F no later than six months after the close of each fiscal year, which is September 30. Copies of reports and other information, when so filed, may be inspected without charge and may be obtained at prescribed rates at the public reference facilities maintained by the Securities and Exchange Commission at 100 F. Street, N.E., Washington, D.C. 20549, and at the regional office of the Securities and Exchange Commission located at Citicorp Center, 500 West Madison Street, Suite 1400, Chicago, Illinois 60661. The public may obtain information regarding the Washington, D.C. Public Reference Room by calling the Commission at 1-800-SEC-0330. The SEC also maintains a Web site at www.sec.gov that contains reports, proxy and information statements, and other information regarding registrants that make electronic filings with the SEC using its EDGAR system. As a foreign private issuer, we are exempt from the rules under the Exchange Act prescribing the furnishing and content of quarterly reports and proxy statements, and officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act.
Our financial statements have been prepared in accordance with U.S. GAAP.
We will furnish our shareholders with annual reports, which will include a review of operations and annual audited consolidated financial statements prepared in conformity with U.S. GAAP.
| I. | Subsidiary Information |
For a listing of our subsidiaries, see Item 4. C. of this annual report, “Information on the Company—Organizational Structure”.
79
Table of Contents
ITEM 11. QUANTITATIVEAND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Foreign Exchange Risk
Our revenues, costs and expenses are currently denominated entirely in Renminbi, but the Renminbi prices of some of the materials and supplies for reagent kits that are imported from companies in the United States, Finland and Sweden may be affected by fluctuations in the value of Renminbi against the currencies of those countries. We do not believe that we currently have any significant direct foreign currency exchange rate risk and have not hedged exposures denominated in foreign currencies or any other derivative financial instruments. Although our revenues and cost of revenues are denominated in Renminbi, fluctuations in the value of the Renminbi may affect the price competitiveness of our products once we commence international sales of our HIFU therapy system. On July 21, 2005, the PRC government changed its policy of pegging the value of the Renminbi to the U.S. dollar. Under the new policy, the Renminbi will be permitted to fluctuate within a band against a basket of certain foreign currencies. This change in policy resulted initially in an approximately 2.0% appreciation in the value of the Renminbi against the U.S. dollar. Since the adoption of this new policy, the value of Renminbi against the U.S. dollar has fluctuated on a daily basis within narrow ranges but overall has further strengthened against the U.S. dollar. There remains significant international pressure on the PRC government to further liberalize its currency policy, which could result in a further and more significant appreciation in the value of the Renminbi against the U.S. dollar. Furthermore, a decline in the value of Renminbi against the U.S. dollar could reduce the U.S. dollar equivalent amounts of our financial results, the value of your investment in our company and the dividends we may pay in the future, if any, all of which may have a material adverse effect on the prices of our ADSs.
Interest Rate Risk
Our risk exposure from changes in interest rates relates primarily to the interest expenses associated with our historical bank borrowings as well as the interest income generated by excess cash invested in demand and time deposits. We have not historically used, and do not expect to use in the future, any derivative financial instruments to manage our interest risk exposure. Interest-earning instruments and borrowings carry a degree of interest rate risk. We have not been exposed nor do we anticipate being exposed to material risks due to changes in interest rates.
ITEM 12. DESCRIPTIONOF SECURITIES OTHERTHAN EQUITY SECURITIES
Not Applicable.
80
Table of Contents
ITEM 13. DEFAULTS, DIVIDEND ARREARAGESAND DELINQUENCIES
None of these events occurred in any of the years ended March 31, 2004, 2005 and 2006.
ITEM 14. MATERIAL MODIFICATIONSTOTHE RIGHTSOF SECURITY HOLDERSAND USEOF PROCEEDS
See “Item 10. Additional Information” for a description of the rights of securities holders, which remain unchanged.
We completed our initial public offering of 64,000,000 ordinary shares, in the form of ADSs, at US$15.00 per ADS on August 10, 2005, after our ordinary shares and American Depositary Receipts were registered under the Securities Act. The aggregate price of the offering amount sold was US$110.4 million, of which we received net proceeds of US$99.5 million. The effective date of our registration statement on Form F-1 (File number: 333-132214) was August 9, 2005. UBS Investment Bank was the sole underwriter/book runner for the global offering of our ADSs.
We have used the net proceeds received from our initial public offering as follows:
| • | approximately US$12.0 million has been used for the remaining payments relating to our acquisition of the ECLIA technology to Beijing Weixiao Biological Technology Development Co., Ltd., or Beijing Weixiao, a company controlled by Mr. Xiaodong Wu, our chairman of board of directors, our chief executive officer and a significant shareholder of our company; |
| • | approximately US$1.1 million has been used to fund our research and development of new applications and product design improvements for our HIFU therapy system and our ECLIA system; |
| • | approximately US$1.2 million has been used to purchase additional production equipment and renovate and expand our ECLIA reagent production facility; and |
| • | approximately US$250,000 has been used to increase our sales and marketing efforts. |
As of March 31, 2006, our cash resources amounted to RMB843.8 million (US$105.3 million), comprising of cash on hand and demand deposits.
ITEM 15. CONTROLSAND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Our Chief Executive Officer and our Chief Financial Officer have performed an evaluation of our disclosure controls and procedures, as that term is defined in Rules 13a-14(c) and 15d-14(c) of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), within 90 days prior to the date of this report, and each has concluded that such disclosure controls and procedures are effective to ensure that information required to be disclosed in our periodic reports filed under the Exchange Act is recorded, processed, summarized and reported, within the time period specified by the Securities and Exchange Commission’s rules and regulations.
Changes in Internal Controls
No significant changes in our internal controls or in other factors that could significantly affect these controls subsequent to the date of the evaluation, including any corrective actions with regard to significant deficiencies and material weaknesses, were made as a result of the evaluation.
ITEM 16A. AUDIT COMMITTEE FINANCIAL EXPERT
Our Board of Directors has determined that Mr. Iain Ferguson Bruce qualify as “audit committee financial expert” as defined in Item 16A of Form 20-F. Each of the members of the Audit Committee is an “independent director” as defined in the Nasdaq Marketplace Rules.
81
Table of Contents
Our board of directors has adopted a code of ethics that applies to our directors, officers, employees and agents, including certain provisions that specifically apply to our chief executive officer, chief financial officer, chief operating officer, chief technology officer, vice presidents and any other persons who perform similar functions for us. We have filed our code of business conduct and ethics as an exhibit to this annual report on Form 20-F. We hereby undertake to provide to any person without charge, a copy of our code of business conduct and ethics within ten working days after we receive such person’s written request.
ITEM 16C. PRINCIPAL ACCOUNTANT FEESAND SERVICES
The following table sets forth the aggregate fees by categories specified below in connection with certain professional services rendered by KPMG, our principal external auditors, for the periods indicated. We did not pay any other fees to our auditors during the periods indicated below.
| For the Year Ended March 31, | ||||||
| 2004 | 2005 | 2006 | ||||
| US$ | US$ | US$ | ||||
| (In thousands) | ||||||
Audit fees(1) | 154 | 423 | 167 | |||
Audit-related fees(2)…. | — | — | 200 | |||
Tax Fees(3)…. | — | — | — | |||
| (1) | “Audit fees” means the aggregate fees billed for professional services rendered by our principal auditors for the audit of our annual financial statements for each of the three years ended March 31, 2005, in connection with our initial public offering and in August 2005 and audit of our historical financial statements for each of the three years ended March 31, 2006. |
| (2) | “Audit-related fees” means the aggregate fees billed by our principal auditors for services rendered that are reasonably related to the performance of the audit or review of our financial statements and are not reported under “Audit Fees”. These fees relate primarily to providing review of the comparative interim financial statements for the nine months ended December 31, 2005 in connection with our follow on offering in March 2006. |
| (3) | “Tax fees” include fees billed for tax consultations. |
The policy of our audit committee is to pre-approve all audit and non-audit services provided by KPMG, including audit services, audit-related services, tax services and other services as described above, other than those for de minimus services which are approved by the Audit Committee prior to the completion of the audit.
ITEM 16D. EXEMPTIONSFROMTHE LISTING STANDARDSFOR AUDIT COMMITTEES
Not applicable.
ITEM 16E. PURCHASESOF EQUITY SECURITIESBYTHE ISSUERAND AFFILIATED PURCHASERS.
None.
82
Table of Contents
We have elected to provide financial statements pursuant to Item 18.
The following financial statements are filed as part of this Annual Report on Form 20-F, together with the report of the independent auditors:
INDEX TO THE FINANCIAL STATEMENTS
| Exhibit Number | Description of Document | |
| 1.1* | Amended and Restated Memorandum and Articles of Association of China Medical Technologies Inc., as currently in effect | |
| 1.2† | Form of Second Amended and Restated Memorandum and Articles of Association of China Medical Technologies Inc. | |
| 2.1* | Specimen Certificate for Ordinary Shares of China Medical Technologies Inc. | |
| 2.2* | Specimen American Depositary Receipt of China Medical Technologies Inc. | |
| 2.3† | Form of Deposit Agreement among China Medical Technologies Inc., the depositary and holders and beneficial owners of the American Depositary Shares issued thereunder | |
| 4.1† | Stock Subscription and Purchase Agreement, dated as of September 23, 2002, among GE (China) Co., Ltd., Golden Meditech (BVI) Company Limited, Beijing Chengxuan Economic and Trade Co., Ltd., Beijing Boda Technology Investment and Development Co., Ltd., Beijing University People’s Hospital, Bi Xiaoqiong, He Shenxu, Bengbu Wanrong Equity Investment Advisory Co., Ltd., Beijing Dongfang Chuanzhi Science and Technology Development Co., Ltd. and Beijing Yuande Bio-Medical Engineering Co., Ltd. | |
| 4.2† | Share Subscription Agreement, dated as of January 19, 2005, among China Medical Technologies, Inc., Chengxuan International Ltd., General Electric International Operations Company, Inc. and Golden Meditech (BVI) Company Limited | |
| 4.3† | Registration Rights Agreement, dated as of January 19, 2005, among the Registrant and other parties therein | |
| 4.4† | Shareholders’ Agreement, dated as of March 19, 2003, among GE (China) Co., Ltd., Golden Meditech (BVI) Company Limited, Beijing Chengxuan Economic and Trade Co., Ltd., Beijing Boda Technology Investment and Development co., Ltd., Beijing University People’s Hospital, Bi Xiaoqiong, He Shenxu, Bengbu Wanrong Equity Investment Advisory Co., Ltd., Beijing Dongfang Chuanzhi Science and Technology Development Co., Ltd. and Beijing Yuande Biological and Engineering Co., Ltd. | |
83
Table of Contents
| Exhibit Number | Description of Document | |
| 4.5† | Acquisition Agreement regarding the ECLIA technology dated as of January 18, 2005 between Beijing Weixiao Biological Technology Development Co., Ltd. and Beijing Yuande Bio-Medical Engineering Co., Ltd. | |
| 4.6† | 2005 stock option plan adopted on February 2, 2005 | |
| 4.7† | Form of Indemnification Agreement with the Registrant’s directors | |
| 4.8† | Form of Employment Agreement between the Registrant and a Senior Executive Officer of the Registrant | |
| 8.1* | List of Subsidiaries | |
| 11.1†† | Code of Business Conduct and Ethics | |
| 12.1†† | CEO Certification Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
| 12.2†† | CFO Certification Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |
| 13.1†† | CEO Certification Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
| 13.2†† | CFO Certification Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |
| 15.1†† | Consent of Walkers | |
| * | Previously filed with the Registrant’s registration statement on Form F-1 (File No. 333-132214). |
| † | Previously filed with the Registrant’s registration statement on Form F-1 (File No. 333-126630). |
| †† | Filed with this Annual Report on Form 20-F. |
84
Table of Contents
SIGNATURES
The registrant hereby certifies that it meets all of the requirements for filing its annual report on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its behalf.
| CHINA MEDICAL TECHNOLOGIES, INC. | ||
By | /S/ XIAODONG WU | |
| Name: | Mr. Xiaodong Wu | |
| Title: | Chief Executive Officer | |
Date: September 29, 2006
Table of Contents
Report of Independent Registered Public Accounting Firm
The Board of Directors and Shareholders of
China Medical Technologies, Inc.:
We have audited the accompanying consolidated balance sheets of China Medical Technologies, Inc. and its subsidiaries as of March 31, 2005 and 2006, and the related consolidated statements of income and comprehensive income, shareholders’ equity and cash flows for each of the years in the three-year period ended March 31, 2006. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of China Medical Technologies, Inc. and its subsidiaries as of March 31, 2005 and 2006, and the results of their operations and their cash flows for each of the years in the three-year period ended March 31, 2006, in conformity with U.S. generally accepted accounting principles.
The accompanying consolidated financial statements as of and for the year ended March 31, 2006 have been translated into United States dollars solely for the convenience of the reader. We have audited the translation and, in our opinion, such financial statements expressed in Renminbi have been translated into United States dollars on the basis set forth in Note 2 to the consolidated financial statements.
/s/ KPMG
Hong Kong, China
September 22, 2006
F-1
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Consolidated Balance Sheets
As of March 31, 2005 and 2006
(RMB and US$ expressed in thousands, except per share data)
| Note | 2005 | 2006 | 2006 | |||||||
| RMB | RMB | US$ | ||||||||
Assets | ||||||||||
Current assets | ||||||||||
Cash and cash equivalents | 14,646 | 843,791 | 105,254 | |||||||
Trade accounts receivable | 4 | 85,766 | 155,751 | 19,428 | ||||||
Prepayments and other receivables | 5 | 20,573 | 26,260 | 3,276 | ||||||
Inventories | 6 | 13,830 | 10,380 | 1,295 | ||||||
Total current assets | 134,815 | 1,036,182 | 129,253 | |||||||
Property, plant and equipment, net | 7 | 75,288 | 105,570 | 13,169 | ||||||
Intangible assets, net | 8 | 239,662 | 224,744 | 28,034 | ||||||
Lease prepayments, net | 9 | 8,003 | 7,811 | 974 | ||||||
Deferred income taxes | 20 | — | 1,534 | 191 | ||||||
Total assets | 457,768 | 1,375,841 | 171,621 | |||||||
Liabilities | ||||||||||
Current liabilities | ||||||||||
Trade accounts payable | 22,595 | 33,833 | 4,220 | |||||||
Amounts due to related parties | 21 | 100,421 | — | — | ||||||
Accrued liabilities and other payables | 10 | 64,862 | 73,872 | 9,215 | ||||||
Income tax payable | 9,395 | 21,602 | 2,694 | |||||||
Dividend payable | 4,225 | — | — | |||||||
Current installments of long-term loan | 11 | 24 | — | — | ||||||
Total current liabilities | 201,522 | 129,307 | 16,129 | |||||||
Amount due to a related party | 21 | 2,293 | — | — | ||||||
Total liabilities | 203,815 | 129,307 | 16,129 | |||||||
Shareholders’ equity | ||||||||||
Share capital | 12(a) | 165,530 | 225,125 | 28,082 | ||||||
Ordinary shares US$0.10 par value: 500,000,000 authorized; 200,000,001 and 273,600,001 issued and outstanding as of March 31, 2005 and 2006, respectively | ||||||||||
Additional paid-in capital | — | 739,936 | 92,299 | |||||||
General reserve fund | 12(b) | 14,852 | 22,275 | 2,779 | ||||||
Unearned compensation | — | (226 | ) | (28 | ) | |||||
Accumulated other comprehensive loss | — | (5,737 | ) | (716 | ) | |||||
Retained earnings | 12(d) | 73,571 | 265,161 | 33,076 | ||||||
Total shareholders’ equity | 253,953 | 1,246,534 | 155,492 | |||||||
Total liabilities and shareholders’ equity | 457,768 | 1,375,841 | 171,621 | |||||||
See accompanying notes to the consolidated financial statements.
F-2
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Consolidated Statements of Income and Comprehensive Income
For the years ended March 31, 2004, 2005 and 2006
(RMB and US$ expressed in thousands, except per share data)
| Note | 2004 | 2005 | 2006 | 2006 | ||||||||||||||
| RMB | RMB | RMB | US$ | |||||||||||||||
Revenues, net(a) | 3(i), 21 | 110,750 | 217,547 | 371,767 | 46,374 | |||||||||||||
Cost of revenues(a) | 15, 21 | (33,983 | ) | (64,579 | ) | (110,494 | ) | (13,783 | ) | |||||||||
Gross profit | 76,767 | 152,968 | 261,273 | 32,591 | ||||||||||||||
Operating expenses: | ||||||||||||||||||
Research and development | 15 | (2,036 | ) | (2,815 | ) | (14,374 | ) | (1,793 | ) | |||||||||
Sales and marketing | 16 | (2,632 | ) | (4,960 | ) | (15,327 | ) | (1,912 | ) | |||||||||
General and administrative | 15, 17 | (5,619 | ) | (16,644 | ) | (38,309 | ) | (4,779 | ) | |||||||||
Total operating expenses | (10,287 | ) | (24,419 | ) | (68,010 | ) | (8,484 | ) | ||||||||||
Operating income | 66,480 | 128,549 | 193,263 | 24,107 | ||||||||||||||
Other income(a) | 18 | — | 1,559 | 9,790 | 1,221 | |||||||||||||
Interest income | 126 | 194 | 14,048 | 1,752 | ||||||||||||||
Interest expense | (112 | ) | (5 | ) | — | — | ||||||||||||
Income before tax | 66,494 | 130,297 | 217,101 | 27,080 | ||||||||||||||
Income tax expense | 20 | (4,933 | ) | (11,854 | ) | (18,088 | ) | (2,256 | ) | |||||||||
Net income | 61,561 | 118,443 | 199,013 | 24,824 | ||||||||||||||
Other comprehensive loss—foreign currency translation adjustment, net of nil tax | — | — | (5,737 | ) | (716 | ) | ||||||||||||
Total comprehensive income | 61,561 | 118,443 | 193,276 | 24,108 | ||||||||||||||
Basic and diluted earnings per share | 14, 20 | RMB | 0.31 | RMB | 0.59 | RMB | 0.81 | US$ | 0.10 | |||||||||
| (a) | Includes the following income/(expenses) resulting from transactions with related parties (see Note 21): |
| 2004 | 2005 | 2006 | 2006 | ||||||
| RMB | RMB | RMB | US$ | ||||||
Revenues, net | 11,966 | 4,145 | 3,932 | 490 | |||||
Cost of revenues—purchase of raw materials in connection with sales to: | |||||||||
—related party | (2,728 | ) | — | — | — | ||||
—external customers | (4,092 | ) | — | — | — | ||||
Other income | — | 3 | 145 | 18 | |||||
Arrangement fee income received from selling shareholders | — | — | 5,905 | 733 | |||||
See accompanying notes to the consolidated financial statements.
F-3
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Consolidated Statements of Shareholders’ Equity
For the years ended March 31, 2004, 2005 and 2006
(RMB and US$ expressed in thousands, except per share data)
| Note | Share capital (Note 12(a)) | Registered capital | Additional paid-in capital | Contribution surplus | General fund | Unearned compensation | Accumulated other comprehensive loss | Retained earnings | Total shareholders’ equity | |||||||||||||||||||
| Number | RMB | RMB | RMB | RMB | RMB | RMB | RMB | RMB | RMB | |||||||||||||||||||
Balance as of April 1, 2003 | — | — | 67,100 | — | 37,192 | 4,899 | — | — | 32,121 | 141,312 | ||||||||||||||||||
Net income | — | — | — | — | — | — | — | — | 61,561 | 61,561 | ||||||||||||||||||
Transfer to reserve | 12(b) | — | — | — | — | — | 3,314 | — | — | (3,314 | ) | — | ||||||||||||||||
Dividends declared | 19 | — | ||||||||||||||||||||||||||
—RMB0.9735 per share | — | — | — | — | — | — | — | — | (39,192 | ) | (39,192 | ) | ||||||||||||||||
—RMB0.4198 per share | — | — | — | — | — | — | — | — | (28,171 | ) | (28,171 | ) | ||||||||||||||||
Balance as of March 31, 2004 | — | — | 67,100 | — | 37,192 | 8,213 | — | — | 23,005 | 135,510 | ||||||||||||||||||
Capital on incorporation | 12(a) | 1 | — | — | — | — | — | — | — | — | — | |||||||||||||||||
Effect of reorganization | 12(a) | 200,000,000 | 165,530 | (67,100 | ) | — | (37,192 | ) | — | — | — | (61,238 | ) | — | ||||||||||||||
Net income | — | — | — | — | — | — | — | — | 118,443 | 118,443 | ||||||||||||||||||
Transfer to reserve | 12(b) | — | — | — | — | — | 6,639 | — | — | (6,639 | ) | — | ||||||||||||||||
Balance as of March 31, 2005 | 200,000,001 | 165,530 | — | — | — | 14,852 | — | — | 73,571 | 253,953 | ||||||||||||||||||
Issue of new shares, net of expenses | 12(a) | 73,600,000 | 59,595 | — | 739,472 | — | — | — | — | — | 799,067 | |||||||||||||||||
Unearned share-based compensation | 27 | — | — | — | 464 | — | — | (464 | ) | — | — | — | ||||||||||||||||
Amortization of share-based compensation expense | 27 | — | — | — | — | — | — | 238 | — | — | 238 | |||||||||||||||||
Net effect of foreign currency translation adjustment, net of nil tax | — | — | — | — | — | — | — | (5,737 | ) | — | (5,737 | ) | ||||||||||||||||
Net income | — | — | — | — | — | — | — | — | 199,013 | 199,013 | ||||||||||||||||||
Transfer to reserve | 12(b) | — | — | — | — | — | 7,423 | — | — | (7,423 | ) | — | ||||||||||||||||
Balance as of March 31, 2006 | 273,600,001 | 225,125 | — | 739,936 | — | 22,275 | (226 | ) | (5,737 | ) | 265,161 | 1,246,534 | ||||||||||||||||
Balance as of March 31, 2006 (US$) | 273,600,001 | 28,082 | — | 92,299 | — | 2,779 | (28 | ) | (716 | ) | 33,076 | 155,492 | ||||||||||||||||
See accompanying notes to the consolidated financial statements.
F-4
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Consolidated Statements of Cash Flows
For the years ended March 31, 2004, 2005 and 2006
(RMB and US$ expressed in thousands)
| Note | 2004 | 2005 | 2006 | 2006 | ||||||||||
| RMB | RMB | RMB | US$ | |||||||||||
Cash flow from operating activities | ||||||||||||||
Net income | 61,561 | 118,443 | 199,013 | 24,824 | ||||||||||
Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||||||||
Depreciation of property, plant and equipment | 1,370 | 3,379 | 3,782 | 472 | ||||||||||
Loss on disposal of property, plant and equipment | 17 | — | 146 | — | — | |||||||||
Amortization of lease prepayments | 86 | 190 | 190 | 24 | ||||||||||
Amortization of intangible assets | — | 8,702 | 14,918 | 1,861 | ||||||||||
Amortization of share-based compensation | — | — | 238 | 30 | ||||||||||
Deferred income taxes | 243 | — | (1,534 | ) | (191 | ) | ||||||||
Changes in assets and liabilities: | ||||||||||||||
Trade accounts receivable | 1,737 | (43,927 | ) | (69,985 | ) | (8,730 | ) | |||||||
Prepayments and other receivables | (3,117 | ) | (14,537 | ) | (5,687 | ) | (709 | ) | ||||||
Inventories | (4,869 | ) | (5,704 | ) | 3,450 | 430 | ||||||||
Trade accounts payable | 12,178 | 9,971 | 11,238 | 1,402 | ||||||||||
Accrued liabilities and other payables | (1,599 | ) | 59,237 | (918 | ) | (115 | ) | |||||||
Income tax payable | 2,774 | 6,621 | 12,207 | 1,522 | ||||||||||
Amount with related party | 21 | (2,770 | ) | 2,250 | — | — | ||||||||
Net cash provided by operating activities | 67,594 | 144,771 | 166,912 | 20,820 | ||||||||||
Cash flow from investing activities | ||||||||||||||
Capital expenditures | (513 | ) | (3,809 | ) | (34,064 | ) | (4,249 | ) | ||||||
Purchase of intangible assets from a related party | (a), 21 | — | (148,364 | ) | — | — | ||||||||
Purchase of time deposits with original maturity over three months | (26,500 | ) | — | — | — | |||||||||
Proceeds of time deposits with original maturity over three months | — | 26,500 | — | — | ||||||||||
Repayment to a related party for the remaining balance on the purchase for a building and intangible assets | (a), 21 | — | (4,807 | ) | (100,000 | ) | (12,474 | ) | ||||||
Net repayments and advances from related parties | 21 | 23,012 | 2,813 | (2,714 | ) | (339 | ) | |||||||
Net cash used in investing activities | (4,001 | ) | (127,667 | ) | (136,778 | ) | (17,062 | ) | ||||||
Cash flow from financing activities | ||||||||||||||
Principal payments on short-term bank loan | (30,000 | ) | — | — | — | |||||||||
Principal payments on long-term loan | (142 | ) | (142 | ) | (24 | ) | (3 | ) | ||||||
Dividends paid | (36,708 | ) | (26,430 | ) | (4,225 | ) | (527 | ) | ||||||
Advances from selling shareholders in secondary offering | 21(g) | — | — | 10,422 | 1,300 | |||||||||
Payment for secondary offering expenses of selling shareholders | 21(g) | — | — | (802 | ) | (100 | ) | |||||||
Net proceeds from issue of new shares | — | — | 799,067 | 99,675 | ||||||||||
Net cash (used in) provided by financing activities | (66,850 | ) | (26,572 | ) | 804,438 | 100,345 | ||||||||
Effect of foreign currency exchange rate change on cash | — | — | (5,427 | ) | (676 | ) | ||||||||
Net (decrease) increase in cash and cash equivalents | (3,257 | ) | (9,468 | ) | 829,145 | 103,427 | ||||||||
Cash and cash equivalents: | ||||||||||||||
At beginning of year | 27,371 | 24,114 | 14,646 | 1,827 | ||||||||||
At end of year | 24,114 | 14,646 | 843,791 | 105,254 | ||||||||||
Supplemental disclosures of cash flow information: | ||||||||||||||
Cash paid during the year for: | ||||||||||||||
Income taxes | 1,916 | 5,233 | 7,415 | 925 | ||||||||||
Interest expense | 112 | 5 | — | — | ||||||||||
(a) Non-cash Transactions | ||||||||||||||
| Note | 2004 | 2005 | 2006 | 2006 | ||||||||||
| RMB | RMB | RMB | US$ | |||||||||||
Partial consideration for acquisition of a building in the form of amount due to a related party | 31 | 4,807 | — | — | — | |||||||||
Partial consideration for acquisition of certain intangible assets and related inventory in the form of amount due to a related party | 31 | — | 100,000 | — | — | |||||||||
See accompanying notes to the consolidated financial statements.
F-5
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006
(expressed in RMB and US$)
| (1) | Reorganization and Principal Activities |
YDME Medical Equipment Inc. was incorporated in the Cayman Islands in July, 2004 as a limited liability company, and changed its name to China Medical Technologies, Inc. (the “Company”) subsequently. The Company was established in connection with the reorganization and initial public offering (“IPO”) of Beijing Yuande Bio-Medical Engineering Co., Ltd. (“Beijing Yuande”). On January 19, 2005, as part of a corporate reorganization, the Company issued 200,000,000 ordinary shares, par value of US$0.1, to all three shareholders of Beijing Yuande in exchange for their beneficial interests in Beijing Yuande. As a result of this reorganization, all three shareholders of Beijing Yuande, Chengxuan International Ltd. (“Chengxuan International”), Golden Meditech (BVI) Company Ltd. (“Golden Meditech”) and GE China Co., Ltd. (“GE”), subscribed for and were issued 110,020,000, 50,000,000 and 39,980,000 ordinary shares of the Company, respectively. Unless otherwise indicated, references to “GE” are General Electric Company or to one or more of its wholly owned subsidiaries, as the case may be and where applicable, to its relevant subsidiaries and its related companies.
Beijing Yuande was established under the laws of the PRC in July 1999 and is principally engaged in the commercialization, manufacture and sale of medical devices and supplies to customers primarily in the People’s Republic of China (the “PRC”). It became a Sino-Foreign Investment Enterprise in March 2003 and became a Wholly Foreign Owned Enterprises (“WFOE”) in June 2004.The Company and its subsidiary, Beijing Yuande, are collectively referred to as the “Group”.
Yuande (USA) Corp, a subsidiary of Beijing Yuande, was incorporated in January 2005 and is principally engaged in carrying out research and development projects and clinical studies on using the HIFU therapy system.
In January 2006, the Group established CMED Technologies Ltd. and CMED Diagnostics Ltd., as wholly-owned subsidiaries in the British Virgin Islands. These subsidiaries are acting as investment holding companies.
IPO
On August 9, 2005, the Company was successfully listed on the Nasdaq National Market and offered 6,400,000 ADSs, representing 64,000,000 ordinary shares, at an initial public offering price of US$15.00 per ADS. On August 29, 2005, an additional 960,000 ADSs, representing 9,600,000 ordinary shares, were sold at the initial public offering price of US$15.00 per ADS pursuant to the underwriter’s exercise of its over-allotment option. In connection with the initial public offering of the Company, net proceeds, after deduction of the related expenses, with aggregate amount of RMB799,067,000 (US$99.7 million) were received.
Secondary Offering
On March 22, 2006, the Company completed a secondary public offering of 57,500,000 ordinary shares held by certain existing shareholders. The selling shareholders received the entire net proceeds from the sale of shares sold in this offering.
| (2) | Basis of Presentation |
The Company’s consolidated financial statements are presented in accordance with generally accepted accounting principles in the United States of America (U.S. GAAP).
Because the Company and Beijing Yuande were under common control, the Company’s acquisition of Beijing Yuande in 2005 has been accounted for in a manner similar to a pooling-of-interests. Consequently, the
F-6
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
consolidated financial statements of the Company include the accounts of Beijing Yuande at their historical amounts. Furthermore, the consolidated financial statements recognize the recapitalization and acquisition retroactively, as if the Company was created and the acquisition occurred as of the beginning of the earliest period presented.
The Group’s functional and reporting currency is RMB. For the United State dollar convenience translation amounts included in the accompanying financial statements, the Renminbi equivalents were translated into United State dollar at the rate of US$1.00=RMB8.0167, on March 31, 2006, representing the noon buying rate in the City of New York for cable transfers of Renminbi, as certified for customs purposes by the Federal Reserve Bank of New York. No representation is made that the Renminbi amounts could have been, or could be, converted into United State dollars at that rate or at any other certain rate on March 31, 2006 or at any other date.
| (3) | Summary of Significant Accounting Policies and Practices |
| (a) | Principles of Consolidation |
The consolidated financial statements include the financial statements of the Company and its majority owned subsidiaries. All significant inter-company balances and transactions have been eliminated in consolidation.
| (b) | Cash and Cash Equivalents |
Cash and cash equivalents consist of cash on hand and in bank. For purposes of the consolidated statement of cash flows, the Company considers all highly liquid debt instruments with original maturities of three months or less to be cash equivalents. None of the Group’s cash is restricted as to withdrawal.
| (c) | Trade Accounts Receivable |
Trade accounts receivable are recorded at the invoiced amount after deduction of trade discounts and allowances, if any, and do not bear interest. The allowance for doubtful accounts is the Group’s best estimate of the amount of probable credit losses in the Group’s existing accounts receivable. The Group determines the allowance based on historical write-off experience, customer specific facts and economic condition.
The Group reviews its allowance for doubtful accounts monthly. Past due balances over 90 days and over a specified amount are reviewed individually for collectibility. All other balances are reviewed on a pooled basis by aging of such balances. Account balances are charged off against the allowance after all means of collection have been exhausted and the potential for recovery is considered remote. The Group does not have any off-balance-sheet credit exposure related to its customers.
| (d) | Inventories |
Inventories are stated at the lower of cost and market. Cost is determined using first-in, first-out method. Cost of work in progress and finished goods comprises direct material, direct production cost and an allocation proportion of production overheads.
| (e) | Property, Plant and Equipment |
Property, plant, and equipment are stated at cost less accumulated depreciation.
F-7
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
Depreciation on property, plant and equipment is calculated on the straight-line method (after taking into account their respective estimated residual values) over the estimated useful lives of the assets as follows:
| Years | ||
Buildings | 30 | |
Production equipment | 5 | |
Research equipment | 5 | |
Furniture, fixtures and office equipment | 5 | |
Computer equipment and system software | 5 | |
Motor vehicles | 5 |
Leasehold improvements are amortized straight line over the shorter of the lease term or estimated useful life of the asset. Depreciation of property, plant and equipment attributable to manufacturing activities is capitalized as part of inventory, and expensed to cost of revenues as inventory is sold. Depreciation related to abnormal amounts from idle capacity is charged to cost of revenues for the period incurred. Total depreciation for the years ended March 31, 2004, 2005, 2006 were RMB1,370,000, RMB3,379,000 and RMB3,782,000, respectively, of which 62%, 35% and 41% were recorded in cost of revenues, Nil, Nil and 2% were recorded in research and development expenses and 38%, 65% and 57% were recorded in general and administrative expenses, respectively.
No depreciation is provided in respect of construction in progress.
| (f) | Intangible Assets |
The Group acquired certain intangible assets for use in an integrated luminescence immunoassay in-vitro diagnostics system using enhanced chemiluminescence (“ECLIA”) technology. These intangible assets include completed technology and know-how in the production of ECLIA analyzers and reagent kits (together known as “ECLIA System”), production permits, a patent and a non-compete commitment. These intangible assets are stated at their relative estimated fair values in relation to the total acquisition cost, less accumulated amortization. Amortization expense is recognized on a straight-line basis over the estimated respective useful lives of these intangible assets, which are as follows:
| Years | ||
Patented technology | 17 | |
Unpatented technology | 20 | |
Non-compete commitment | 10 |
The Group reviews the carrying values of its purchased intangible assets if the facts and circumstances suggest these assets may be impaired. To the extent that the review indicates that the carrying values of these assets may not be recoverable, as determined based on their estimated future undiscounted cash flows over the remaining amortization period, the carrying values of these assets will be reduced to their estimated fair values.
Given the environment in which the Group currently operates, it is reasonably possible that the estimated economic useful lives of these assets or the Group’s estimate that it will recover their carrying amounts from future operations could change in the future.
F-8
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (g) | Lease Prepayments |
Lease prepayments represent land use rights paid to the PRC’s land bureau. Land use rights are carried at cost and amortized on a straight-line basis over the respective periods of rights which range from 44 to 47 years. The current portion of lease prepayments has been included in prepayments and other receivables in the balance sheet.
| (h) | Impairment of Long-Lived Assets |
Long-lived assets, such as property, plant, and equipment and intangible assets with estimable useful lives, are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable.
Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to the estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated future cash flows, an impairment charge is recognized by the amount by which the carrying amount of the asset exceeds the fair value of the asset. Assets to be disposed of are separately presented in the balance sheet and reported at the lower of the carrying amount or fair value less costs to sell, and are no longer depreciated. The assets and liabilities of a disposed group classified as held for sale are presented separately in the appropriate asset and liability sections of the balance sheet.
| (i) | Revenue Recognition |
Sales of medical equipment and supplies represent the invoiced value of goods, net of value added taxes, sales returns, trade discounts and allowances. The Group recognizes revenue when products are delivered and the customer takes ownership and assumes risk of loss, collection of the relevant receivable is probable, persuasive evidence of an arrangement exists and the sales price is fixed or determinable. The sales price of the medical equipment includes training and installation services. These services are ancillary to the purchase of medical equipment by customers and are normally considered by the customers to be an integral part of the acquired equipment. As the delivered items (training and installation services) do not have determinable fair values, the Group recognizes revenue for the entire arrangement upon customer acceptance, which occurs after delivery, installation and training.
In the PRC, value added tax (“VAT”) of 17% on invoice amount is collected in respect of the sales of goods on behalf of tax authorities. The VAT collected is not a revenue of the Group; instead, the amount is recorded as a liability on the balance sheet until such VAT is paid to the authorities.
Pursuant to the laws and regulations of the PRC, Beijing Yuande is entitled to a refund of VAT on the sales of self-developed software embedded in the medical equipment. The VAT refund represents the amount of VAT collected from customers and paid to the authorities in excess of 3% of relevant sales. The amount of VAT refund is calculated on a monthly basis. As the refund relates directly to the sale of self-developed software that is embedded in the product, the Group recognizes the VAT refund at the time the product is sold. The amount is included in the line item “Revenues, net” in the statement of income and is recorded on an accrual basis. VAT refunds included in revenue for the years ended March 31, 2004, 2005 and 2006 were RMB7,102,000, RMB14,725,000 and RMB23,009,000 (US$2,870,000), respectively.
The medical equipment sold by the Group has embedded self-developed software. In all cases, the medical equipment is marketed and sold based on its performance and functionality as a whole. The self- developed software is not sold on a standalone basis. As the self-developed software embedded in the
F-9
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
medical equipment is incidental to the product as a whole, the Group does not account for the software revenue in accordance with Statement of Position 97-2 “Software Revenue Recognition” issued by American Institute of Certified Public Accountants.
| (j) | Government grant |
Non-refundable grant is made by the PRC government authorities to Beijing Yuande as it qualified under one of the government’s technology grant programmes in Beijing. The grant can be used for enterprise development and technology innovation purposes. This government grant is recognized in the statement of income as other income when the grant is received.
| (k) | Income Taxes |
Income taxes are accounted for under the asset and liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases and operating loss and tax credit carry forwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date.
| (l) | Stock-based compensation |
The Company grants stock options to its employees and directors. The Company accounts for employee stock-based compensation in accordance with Accounting Principles Board Opinion No. 25, “Accounting for Stock Issued to Employees” (“APB No. 25”), and its related interpretations which require the Company to record a compensation charge for the excess of the fair value for the stock at the grant date over the amount an employee must pay to acquire the stock. The compensation expense is recognized over the applicable service period, which is usually the vesting period.
Had compensation cost for the options granted to employees and directors in the fiscal year ended March 31, 2006 under the Company’s 2005 stock option plan (the “Plan”) (see note 27) been determined based on the fair value at the grant date, as prescribed in Statement of Financial Accounting Standard No. 123,“Accounting for Stock-Based Compensation”, the Company’s pro forma net income would have been as follows:
| Year ended March 31 | |||||||||||
| 2004 | 2005 | 2006 | |||||||||
| RMB’000 | RMB’000 | RMB’000 | |||||||||
Net income as reported | 61,561 | 118,443 | 199,013 | ||||||||
Add: Stock compensation as reported, net of tax | — | — | 238 | ||||||||
Less: Stock compensation determined using the fair value method, net of tax | — | (30 | ) | (1,092 | ) | ||||||
Pro forma net income | 61,561 | 118,413 | 198,159 | ||||||||
Shares used in computing basic earnings per share (‘000) | 200,000 | 200,000 | 246,878 | ||||||||
Shares used in computing diluted earnings per share (‘000) | 200,018 | 200,018 | 247,163 | ||||||||
Net income per share: | |||||||||||
Basic and diluted, as reported | RMB | 0.31 | RMB | 0.59 | RMB | 0.81 | |||||
Basic and diluted—pro forma | RMB | 0.31 | RMB | 0.59 | RMB | 0.81 | |||||
F-10
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
The fair value each option granted is estimated on the date of grant using the Black-Scholes option-pricing model, with the following assumptions used for grants during the applicable period.
Option grants | 2005 | 2006 | ||
Average risk-free interest rate of return | 3.911% | 3.911% | ||
Weighted average expected option life | 2.75 years | 2.75 years | ||
Volatility rate | 30.00% | 30.00% | ||
Dividend yield | 0.00% | 0.00% |
| (m) | Research and Development Costs |
Research and development costs are expensed as incurred. Research and development costs amounted to RMB2,036,000, RMB2,815,000 and RMB14,374,000 (US$1,793,000) for the years ended March 31, 2004, 2005 and 2006, respectively.
| (n) | Advertising Costs |
Advertising costs are expensed as incurred. Advertising costs amounted to RMB84,000, RMB562,000 and RMB1,212,000 (US$151,000) for the years ended March 31, 2004, 2005 and 2006, respectively.
| (o) | Retirement and Other Postretirement Benefits |
Contributions to retirement schemes (which are defined contribution plans) are charged to statements of income as and when the related employee service is provided.
| (p) | Foreign Currency Transactions and Translation |
Transactions denominated in currencies other than RMB are translated into RMB at the exchange rates quoted by the People’s Bank of China (the “PBOC”) prevailing at the dates of transactions. Monetary assets and liabilities denominated in foreign currencies are translated into RMB using the applicable exchange rates quoted by the PBOC at the balance sheet dates. The resulting exchange differences are recorded in the statements of income as part of the related transaction amounts.
RMB is not fully convertible into foreign currencies. All foreign exchange transactions involving RMB must take place either through the PBOC or other institutions authorized to buy and sell foreign exchange. The exchange rate adopted for the foreign exchange transactions are the rates of exchange quoted by the PBOC which are determined largely by supply and demand.
Asset and liabilities of foreign group companies whose functional currency is not the RMB are translated into RMB using the exchange rate on the balance sheet date. Revenues and expenses are translated at average rates prevailing during the year. The gains and losses resulting from translation of financial statements of foreign group companies are recorded as a separate component of accumulated other comprehensive income/(loss) within shareholders’ equity. Cumulative translation adjustments are recognized as income or expense upon disposal or liquidation of a foreign group company.
| (q) | Warranty Costs |
The Group records a liability required for specific product repairs covered under warranty when sales are made. The term of the product warranty is generally twelve months. Based on the limited number of actual warranty claims and the historically low cost of such repairs, the Group has not recognized a liability for warranty claims.
F-11
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (r) | Earnings per Share |
Basic earnings per share (“EPS”) excludes dilutive potential ordinary shares and is computed by dividing net income attributable to ordinary shareholders by the weighted average number of ordinary shares outstanding during the period. Diluted earnings per share is calculated by dividing net income by the weighted average number of ordinary and dilutive potential ordinary shares outstanding during the period.
| (s) | Use of Estimates |
The preparation of the financial statements in accordance with U.S. GAAP requires management of the Company to make a number of estimates and assumptions relating to the reported amount of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the period. Significant items subject to such estimates and assumptions include the recoverability of the carrying amount and the estimated useful lives of long-lived assets, valuation allowances for receivables, realizable values for inventories and estimated warranty obligations. Actual results could differ from those estimates.
| (t) | Segment Reporting |
The Group uses the “management approach” in determining reportable operating segments. The management approach considers the internal organization and reporting used by the Group’s chief operations decision maker for making operating decisions and assessing performance as the source for determining the Group’s reportable segments. The Group’s chief operating decision maker is the Chief Executive Officer of the Group. Based on this assessment, the Group has determined it has two reportable operating segments, namely High Intensity Focused Ultrasound (“HIFU”) System and ECLIA System.
In view of the fact that the Group operates and manages its business substantially in the PRC, no geographical segment information is presented.
| (u) | Contingencies |
In the normal course of business, the Group is subject to contingencies, including legal proceedings and claims arising out of the businesses that relate to a wide range of matters, including among others, product liability. The Group records accruals for such contingency based upon the assessment of the probability of occurrence and, where determinable, an estimate of the liability. The Group may consider many factors in making these assessments including past history, scientific evidence and the specifics of each matter. As the Group has not become aware of any product liability claim arising from any medical incident over the last three years, the Group has not recognized a liability for product liability claims.
| (v) | Recently Issued Accounting Pronouncements |
In November 2004, the FASB issued SFAS No.151, “Inventory Costs—an amendment of ARB No. 43”. SFAS 151 requires amounts of idle facility expense, freight, handling costs and wasted material (spoilage) be recognized as current-period charges. In addition, it requires that the allocation of fixed production overhead to the costs of conversion be based on the normal capacity of the production facilities. SFAS 151 is effective for inventory costs incurred during fiscal years beginning after June 15, 2005. The Group does not believe the adoption of SFAS 151 will have a significant impact on its consolidated financial statements.
F-12
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
In December 2004, the FASB issued SFAS No.153, “Exchanges of Nonmonetary Assets, and amendment of APB Opinion No. 29”. The guidance in APB Opinion No. 29 “Accounting for Nonmonetary Transactions”, is based on the principle that exchanges of nonmonetary assets should be measured based on the fair value of the assets exchanged. The guidance in that opinion, however, included certain exceptions to that principle. SFAS 153 amends APB Opinion 29 to eliminate the exception for nonmonetary exchanges of similar productive assets and replaces it with a general exception for exchanges of nonmonetary assets that do not have commercial substance. A nonmonetary exchange has commercial substance if the future cash flows of the entity are expected to change significantly as result of the exchange. SFAS 153 is effective for nonmonetary asset exchanges occurring in fiscal periods beginning after June 15, 2005. The Group currently does not contemplate entering into any transactions within the scope of SFAS 153. Consequently, the Group does not believe the adoption of SFAS 153 will have any impact on its consolidated financial statements.
In December 2004, the FASB issued SFAS No.123R “Share-Based Payment” (“SFAS 123R”), which requires companies to measure and recognize compensation expense for all stock-based payments at fair value. SFAS 123R replaces SFAS No.123, “Accounting for Stock-Based Compensation”and supersedes APB Opinion No.25, “Accounting for Stock Issued to Employees”. SFAS 123R is effective for all interim and annual periods beginning after June 15, 2005. Under a new US Securities and Exchange Commission rule, a company may elect to adopt the provisions of SFAS 123R at the beginning of their first annual period beginning after June 15, 2005. Consequently, the Company has elected to defer adoption of SFAS 123R until April 1, 2006. The Company expects to recognize amortization of this compensation expense of RMB1,256,000 (US$157,000) and RMB193,000 (US$24,000) for the years ending March 31, 2007 and 2008, respectively, upon the adoption of SFAS 123R. The pro forma results disclosed in Note 3(l) are not necessarily indicative of what the impact of SFAS 123R will be upon adoption. In March 2005, the SEC issued Staff Accounting Bulletin No. 107 with respect to the SEC’s interpretation of SFAS 123(R) and the valuation of share-based payments for public companies.
In May 2005, the FASB issued SFAS No.154 “Accounting Changes and Error Corrections” (“SFAS 154). SFAS 154 requires restatement of prior periods’ financial statements for changes in accounting principles, unless it is impracticable to determine either the period-specific effects or the cumulative effect of the change. Also, SFAS 154 requires that retrospective application of a change in accounting principle be limited to the direct effects of the change. SFAS 154 is effective for accounting changes and corrections of errors made in fiscal years beginning after December 15, 2005. Management does not believe that the adoption of SFAS 154 will have a material impact on the Company’s consolidated financial statements.
In September 2005, the FASB Emerging Issues Task Force (EITF) issued EITF Issue No. 04-13,Accounting for Purchases and Sales of Inventory with the Same Counterparty. EITF 04-13 provides guidance as to when purchases and sales of inventory with the same counterparty should be accounted for as a single exchange transaction. EITF 04-13 also provides guidance as to when a nonmonetary exchange of inventory should be accounted for at fair value. EITF 04-13 will be applied to new arrangements entered into, and modifications or renewals of existing arrangements occurring after January 1, 2007. Management does not believe that the application of EITF 04-13 will have a material impact on the Company’s consolidated financial statements.
In July 2006, the FASB issued FASB Interpretation No. 48, “Accounting for Uncertainty in Income Taxes—an Interpretation of FASB Statement No. 109” (“FIN 48”), which clarifies the accounting for uncertainty in tax positions. This Interpretation requires that the Company recognize in its financial statements the impact of a tax position if that position is more likely than not of being sustained on audit, based on the technical merits of the position. The provisions of FIN 48 are effective for the Company on
F-13
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
January 1, 2007, with the cumulative effect of the change in accounting principle, if any, recorded as an adjustment to opening retained earnings. The Company is currently evaluating the impact of adopting FIN 48 on the Company’s consolidated financial statements.
In September 2006, the SEC issued Staff Accounting Bulletin (“SAB”) No. 108, which provides guidance on the process of quantifying financial statement misstatements. In SAB No. 108, the SEC staff establishes an approach that requires quantification of financial statement errors, under both the iron-curtain and the roll-over methods, based on the effects of the error on each of our financial statements and the related financial statement disclosures. SAB No. 108 is generally effective for annual financial statements in the first fiscal year ending after November 15, 2006. The transition provisions of SAB No. 108 permits existing public companies to record the cumulative effect in the first year ending after November 15, 2006 by recording correcting adjustments to the carrying values of assets and liabilities as of the beginning of that year with the offsetting adjustment recorded to the opening balance of retained earnings. The Company is currently evaluating the impact of adopting SAB No. 108 on the consolidated financial statements.
In September 2006, the FASB issued SFAS No. 157 “Fair Value Measurements” (“SFAS 157”). SFAS 157 defines fair value, establishes a framework for measuring fair value in generally accepted accounting principles, and expands disclosures about fair value measurements. SFAS 157 applies under other accounting pronouncements that require or permit fair value measurements, where fair value is the relevant measurement attribute. The standard does not require any new fair value measurements. SFAS 157 is effective for financial statements issued for fiscal years beginning after November 15, 2007, and interim periods within those fiscal years. The Company is currently evaluating the impact of adopting SFAS 157 on the consolidated financial statements.
| (4) | Trade Accounts Receivable |
The Group performs ongoing credit evaluations of its customers’ financial condition. The Group requires certain customers including hospitals to provide advanced cash deposits for their purchases. The Group has been able to collect on all of its receivable balances, and accordingly, the Group did not provide an allowance for doubtful accounts at March 31, 2005 or 2006.
| (5) | Prepayments and Other Receivables |
| 2005 | 2006 | 2006 | ||||
| RMB’000 | RMB’000 | US$’000 | ||||
VAT refund receivables | 9,554 | 20,303 | 2,533 | |||
Prepayments (notes (a) and (b)) | 10,722 | 4,284 | 534 | |||
Other receivables | 297 | 1,673 | 209 | |||
| 20,573 | 26,260 | 3,276 | ||||
| (a) | Prepayments at March 31, 2005 and 2006 include an amount of RMB190,000 and RMB192,000 (US$24,000), respectively, representing the current portion of lease prepayments of the Group (see Note 9). |
| (b) | Prepayments at March 31, 2005 and March 31, 2006 also include an amount of RMB7,901,000 and RMBNil (US$Nil), respectively, representing the deferred expenses in connection with the IPO of the Company. |
F-14
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (6) | Inventories |
Inventories at March 31, 2005 and 2006 consist of the following:
| 2005 | 2006 | 2006 | ||||
| RMB’000 | RMB’000 | US$’000 | ||||
Raw materials | 3,826 | 2,686 | 335 | |||
Work-in-progress | 4,691 | 4,619 | 576 | |||
Finished goods | 5,313 | 3,075 | 384 | |||
| 13,830 | 10,380 | 1,295 | ||||
| (7) | Property, Plant and Equipment, net |
Property, plant and equipment consist of the following:
| 2005 | 2006 | 2006 | |||||||
| RMB’000 | RMB’000 | US$’000 | |||||||
Buildings | 75,286 | 75,286 | 9,391 | ||||||
Leasehold improvements | 630 | 1,405 | 175 | ||||||
Production equipment | 3,182 | 4,904 | 612 | ||||||
Research equipment | — | 17,741 | 2,213 | ||||||
Furniture, fixtures and office equipment | 599 | 1,272 | 159 | ||||||
Computer equipment and system software | 468 | 1,507 | 188 | ||||||
Motor vehicles | 1,278 | 3,525 | 440 | ||||||
| 81,443 | 105,640 | 13,178 | |||||||
Accumulated depreciation | (6,296 | ) | (10,078 | ) | (1,257 | ) | |||
| 75,147 | 95,562 | 11,921 | |||||||
Construction-in-progress (note (a)) | 141 | 10,008 | 1,248 | ||||||
Net carrying amount | 75,288 | 105,570 | 13,169 | ||||||
| (a) | Construction-in-progress |
Construction-in-progress at March 31, 2005 and 2006 mainly represent expenditures in respect of the leasehold improvements and the expansion of Beijing Yuande’s facilities for the production of ECLIA reagent kits, respectively.
All of the Group’s buildings are located in the PRC.
F-15
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (8) | Intangible Assets, net |
The following table summarizes the Group’s net amortized cost of its intangible assets as of March 31, 2005 and 2006:
| 2006 | |||||||||
| Gross carrying amount | Accumulated amortization | Net | Net | ||||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | ||||||
Patented technology | 10,530 | (980 | ) | 9,550 | 1,191 | ||||
Unpatented technology | 189,699 | (15,018 | ) | 174,681 | 21,790 | ||||
Non-compete commitment | 48,135 | (7,622 | ) | 40,513 | 5,053 | ||||
| 248,364 | (23,620 | ) | 224,744 | 28,034 | |||||
| 2005 | |||||||||
| Gross carrying amount | Accumulated amortization | Net | |||||||
| RMB’000 | RMB’000 | RMB’000 | |||||||
Patented technology | 10,530 | (361 | ) | 10,169 | |||||
Unpatented technology | 189,699 | (5,533 | ) | 184,166 | |||||
Non-compete commitment | 48,135 | (2,808 | ) | 45,327 | |||||
| 248,364 | (8,702 | ) | 239,662 | ||||||
As described in Note 21, the Group purchased certain assets, including intangible assets from Beijing Weixiao Biological Technology Development Co., Ltd. (“Beijing Weixiao”), a related party controlled by Mr. Wu Xiaodong, in August 2004. These intangible assets include completed technology and know-how in the production of the ECLIA System, a patent and a non-compete commitment.
Patented technology represents a patent for the production of a component of the ECLIA analyzer. The patent was applied by Beijing Weixiao prior to the acquisition.
Unpatented technology represents specific application and processes related to the production of the ECLIA analyzer and the ECLIA reagent kits, and the Group has begun the process of securing patents with respect to the unpatented technology in applicable jurisdictions. A significant portion of the acquisition cost was allocated to the unpatented technology due to the significant contribution of the unpatented technology to the future cash flows of the Group as compared to the patented technology and the non-compete commitment. The unpatented technology arises from legal rights that existed at the date of the ECLIA acquisition. The technology-based intangible asset is capable of being separated or divided and can be sold, transferred, licensed, rented or exchanged with a related contract.
In connection with the acquisition of the ECLIA System, a commitment not to compete against Beijing Yuande was entered into with Beijing Weixiao, a related party controlled by Mr. Wu Xiaodong, from which the Group acquired the intangible assets.
For the period from June 2004 through January 2005, the voting interests and, therefore, the authority to make significant operating and policy decisions, were delegated to the three members of the board of directors of Beijing Yuande. As each shareholder of Beijing Yuande had one member as a director on its board, the voting interests in Beijing Yuande were effectively allocated equally among Chengxuan International, a company controlled by Mr. Wu Xiaodong, Golden Meditech and GE. Consequently, Chengxuan International,
F-16
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
notwithstanding its greater than 50% of the equity interests in Beijing Yuande, did not have a majority voting interest nor a controlling financial interest over Beijing Yuande. Accordingly, the acquisition of ECLIA assets in August 2004 from this related party was recorded at their relative fair values in relation to the total consideration paid.
Amortization expenses for these intangible assets were RMBNil, RMB8,702,000 and RMB14,918,000 (US$1,861,000) for the years ended March 31, 2004, 2005 and 2006, respectively. Estimated amortization expense for the next five years is RMB14,918,000 (US$1,861,000) each year.
| (9) | Lease Prepayments, net |
The balance represents the land use rights of the Group as follows:
| 2005 | 2006 | 2006 | |||||||
| RMB’000 | RMB’000 | US$’000 | |||||||
Land use rights prepaid | 8,641 | 8,641 | 1,078 | ||||||
Accumulated amortization | (448 | ) | (638 | ) | (80 | ) | |||
| 8,193 | 8,003 | 998 | |||||||
The balance is analyzed as follows: | |||||||||
Lease prepayments—non current portion | 8,003 | 7,811 | 974 | ||||||
Amount to be amortized next year (Note 5) | 190 | 192 | 24 | ||||||
| 8,193 | 8,003 | 998 | |||||||
| (10) | Accrued Liabilities and Other Payables |
Accrued liabilities and other payables at March 31, 2005 and 2006 consist of the following:
| 2005 | 2006 | 2006 | ||||
| RMB’000 | RMB’000 | US$’000 | ||||
Other payables (note (a)) | 12,702 | 22,508 | 2,808 | |||
VAT payables | 13,885 | 35,009 | 4,367 | |||
Accruals for salaries and welfare | 5,183 | 13,557 | 1,691 | |||
Receipts in advance (note (b)) | 33,092 | 2,798 | 349 | |||
| 64,862 | 73,872 | 9,215 | ||||
| (a) | Other payables at March 31, 2005 and 2006 include an amount of RMB3,889,000 and RMBNil (US$Nil), respectively, representing the accrued expenses in connection with the proposed IPO of the Company. |
| Other payables at March 31, 2005 and 2006 also include an amount of RMB6,207,000 and RMBNil (US$Nil), respectively, representing the financial advisory expenses payable for the reorganization of the Group. |
| Other payables at March 31, 2006 include amounts of RMB9,620,000 (US$1,200,000) and RMB7,806,000 (US$974,000), representing the cost incurred for the secondary offering of the Company on March 22, 2006 (Note 21(g)) and provision for professional service fees respectively. |
| (b) | Receipts in advance represent cash deposits received from customers in connection with the sales of products. |
F-17
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (11) | Long-term loan |
| 2005 | 2006 | 2006 | |||||
| RMB’000 | RMB’000 | US$’000 | |||||
RMB denominated loan: | |||||||
—Fixed interest rate of 5.49% per annum as of March 31, 2005, with maturity in May 2005 | 24 | — | — | ||||
Less: Amount due within 1 year included under current liabilities | (24 | ) | — | — | |||
Amount due after 1 year | — | — | — | ||||
As of March 31, 2005, a loan of RMB24,000 was used for the purchase of a motor vehicle.
| (12) | Shareholders’ Equity |
(a) The Company has a simple capital structure with one class of ordinary shares. On July 6, 2004, the Company issued 1 share for par value of US$0.0001 on incorporation with an authorized share capital of 500,000,000 ordinary shares at par value of US$0.0001 each.
On January 17, 2005, the Company:
(i) raised capital through the issuance of 999 ordinary shares in a private placement at a price of US$0.0001 per share for a consideration of US$0.0999;
(ii) consolidated 1,000 ordinary shares in issue with par value of US$0.0001 each to 1 ordinary share with par value of US$0.1 each;
(iii) consolidated 499,999,000 un-issued ordinary shares with par value of US$0.0001 each to 499,999 ordinary shares with par value of US$0.1 each; and
(iv) increased the authorized share capital from 500,000 ordinary shares with par value of US$0.1 each to 500,000,000 ordinary shares of US$0.1 each by the creation of an additional 499,500,000 ordinary shares of US$0.1 each.
On January 19, 2005, in connection with the reorganization, the Company issued 200,000,000 ordinary shares on a pro-rata basis to the shareholders of Beijing Yuande. As part of this corporate reorganization, all three shareholders of Beijing Yuande, Chengxuan International, Golden Meditech and GE, subscribed for and were issued 110,020,000, 50,000,000 and 39,980,000 ordinary shares of the Company, respectively, in exchange for their respective outstanding ownership interests in Beijing Yuande. GE’s ordinary shares in the Company are held in trust by GEIO. Because the three shareholders’ relative ownership interests in the Company are identical to their ownership interests in Beijing Yuande, the reorganization is deemed to lack substance for financial reporting purposes, and amounts were recorded at their historical cost basis. The excess of the Company’s ordinary shares over the registered capital of Beijing Yuande was first applied to reduce contribution surplus to zero with the remaining amount applied to reduce retained earnings.
During the year ended March 31, 2006, the Company issued 73,600,000 new shares through an IPO. See Note 1 to the Consolidated Financial Statements for details of the IPO.
(b) In accordance with the relevant laws and regulations for sino-foreign investment enterprises incorporated under the Law of the PRC on Joint Venture Using Chinese and Foreign Investment, the appropriation to Beijing Yuande’s general reserve fund is at the discretion of the board of directors of the respective joint venturers. In
F-18
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
January 2005, Beijing Yuande became a WFOE. Under the Law of the PRC on Enterprises with Sole Foreign Investment, Beijing Yuande is required to set aside at least 10% of its respective after-tax profit determined under the relevant PRC accounting regulations to general reserve fund. The appropriations are required until the balance reaches 50% of Beijing Yuande’s registered capital. Statutory general reserve can be used to recoup previous years’ losses, if any, and may be converted into share capital by the issue of new shares to shareholders in proportion to their existing shareholdings, or by increasing the par value of the shares currently held by them, provided that the reserve balance after such issue is not less than 25% of the registered share capital. The transfer to this reserve must be made before distribution of any dividend to shareholders. For the years ended March 31, 2004, 2005 and 2006, Beijing Yuande transferred RMB3,314,000, RMB6,639,000 and RMB7,423,000 (US$926,000), respectively, being 10% of the respective year’s net income determined in accordance with PRC accounting rules and regulations, to this reserve.
(c) In accordance with the relevant laws and regulations in the PRC, Beijing Yuande is required to make appropriation to a staff welfare and bonus reserve. The amount allocated to this fund is at the discretion of the board of directors. The appropriation to staff welfare and bonus reserve has been reclassified as expense as it is a liability to the employees. This fund is recorded as a current liability and can only be utilized for making special bonuses or collective welfare to the employees of Beijing Yuande, such as construction of dormitories, canteen and other welfare facilities. For the years ended March 31, 2004, 2005 and 2006, the directors authorized and approved, the transfer of RMB2,407,000 RMB3,750,000 and RMB2,227,000 (US$278,000), respectively, being 5%, 3% and 3% of the respective year’s net income determined in accordance with the PRC accounting rules and regulations, to this fund.
(d) According to the Articles of Association of Beijing Yuande, the amount of retained earnings available for distribution to shareholders is the amount determined in accordance with the PRC accounting rules and regulations. As of December 31, 2003, 2004 and 2005, the statutory year end for Beijing Yuande, the amount of retained earnings available for distribution was RMB67,363,000, RMB66,387,000 and RMB122,334,000 (US$15,260,000) respectively, being the amount determined in accordance with the PRC accounting rules and regulations. Dividends of RMB39,192,000 in respect of the statutory years 2001 and 2002 and RMB28,171,000 in respect of the statutory year 2003 were declared after December 31, 2003 and had been recognized as a liability at the balance sheet date March 31, 2004. No dividend in respect of the statutory year 2004 and 2005 was declared as of the balance sheet date of March 31, 2005 and 2006 (see Note 19).
| (13) | Shareholders’ Agreement |
In May 2004, a revised Memorandum and Articles of Association (the “Revised M&A”) was adopted by the shareholders of Beijing Yuande and duly executed. All matters relating to the management of Beijing Yuande as stipulated in the Beijing Yuande Shareholders’ Agreement were superseded by the Revised M&A. As such, Beijing Yuande Shareholders’ Agreement was null and void.
Under the Revised M&A, Golden Meditech and GE, which hold an equity interest of 25% and 15% respectively in Beijing Yuande each held one of the three seats on the Board of Directors, and as a result, effectively had an equal voting interest to make significant decisions that are expected to be made in the ordinary course of Beijing Yuande’s business operations. Parties to the Revised M&A believed the delegation of the decision-making authority to the board members was necessary to protect the interests of the minority shareholders of Beijing Yuande and to encourage the participation of GE and Golden Meditech to enhance the corporate governance of Beijing Yuande.
On January 19, 2005, Chengxuan International (formerly “Beijing Chengxuan”), Golden Meditech and GE entered into a new Shareholders’ Agreement (the “January 2005 Shareholders’ Agreement”), which provides for
F-19
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
certain matters relating to the management of the Company. The January 2005 Shareholders’ Agreement provided that the Board of Directors of the Company will consist of five directors. Chengxuan International and Golden Meditech were entitled to designate two and one directors, respectively. In addition, the January 2005 Shareholders’ Agreement also provided that Chengxuan International will be entitled to designate two independent directors to the Board of Directors of the Company. If any vacancy occurs on the Board, the shareholders entitled to designate such outgoing directors shall have the right to designate a replacement.
The January 2005 Shareholders’ Agreement provided an observer right to the representative of GE, who may attend all meetings of the Board in a nonvoting capacity but shall not disclose any business of the Board to any person, other than an affiliate of such representative.
| (14) | Earnings per share |
Basic earnings per share for the years ended March 31, 2004 and 2005 has been calculated by dividing net income by the number of shares issued and outstanding of 200,000,001 immediate prior to the Company’s IPO, as if such shares had been outstanding for the periods presented. Basic earnings per share for the year ended March 31, 2006 has been calculated by dividing net income by the weighted average number of ordinary shares issued and outstanding during the period.
The following table sets forth the computation of basic and diluted earnings per share for the years indicated:
| 2004 | 2005 | 2006 | 2006 | |||||||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||||||
Numerator: | ||||||||||||
Net income | 61,561 | 118,443 | 199,013 | 24,824 | ||||||||
Denominator: | ||||||||||||
Weighted average number of ordinary shares outstanding and shares used in computing basic earnings per share (‘000) | 200,000 | 200,000 | 246,878 | 246,878 | ||||||||
Weighted average number of potential ordinary shares (‘000) | 18 | 18 | 285 | 285 | ||||||||
Shares used in computing diluted earnings per share (‘000) | 200,018 | 200,018 | 247,163 | 247,163 | ||||||||
Basic and diluted earnings per share | RMB | 0.31 | RMB | 0.59 | RMB | 0.81 | US$ | 0.10 | ||||
The weighted average number of ordinary shares outstanding used to calculate earnings per share for periods prior to the corporate reorganization in January 2005 has been retroactively determined based upon the exchange ratio used to issue shares as part of the reorganization. For the years ended March 31, 2004 and 2005, diluted earnings per share have been computed to give effect to 400,000 ordinary shares of the Company issuable upon exercise of the options granted on March 14, 2005. Diluted earnings per share for the year ended March 31, 2006 has been computed to give effect to aggregate 800,000 ordinary shares of the Company issuable upon exercise of the options granted on March 14, 2005 and August 8, 2005.
F-20
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (15) | Depreciation and Amortization |
Depreciation of property, plant and equipment and amortization of intangible assets and lease prepayments are included in the following captions:
| 2004 | 2005 | 2006 | 2006 | |||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||
Cost of revenues | 903 | 9,950 | 16,558 | 2,065 | ||||
Research and development | — | — | 80 | 10 | ||||
General and administrative | 553 | 2,321 | 2,253 | 281 | ||||
| 1,456 | 12,271 | 18,891 | 2,356 | |||||
| (16) | Sales and Marketing Expenses |
Sales and marketing expenses for the years ended March 31, 2004, 2005 and 2006 include repairing and warranty expenses of RMB8,000, RMB28,000 and RMB12,000 (US$2,000), respectively.
| (17) | General and Administrative Expenses |
General and administrative expenses for the years ended March 31, 2004, 2005 and 2006 include the following:
| 2004 | 2005 | 2006 | 2006 | |||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||
Personnel expenses | 3,445 | 5,409 | 17,112 | 2,135 | ||||
Financial advisory expenses | — | 6,207 | — | — | ||||
Loss on disposal of property, plant and equipment | — | 146 | — | — | ||||
Financial advisory expenses represent amounts incurred for the reorganization of the Group.
| (18) | Other income |
Included in other income for the years ended March 31, 2005 and 2006 was a non-refundable grant of RMB1,556,000 and RMB3,740,000 (US$467,000), respectively, granted by the PRC government authorities as Beijing Yuande operates in the high and new technology business sector. The income can be used for enterprise development and technology innovation purposes.
In addition, other income for 2006 included Rmb5,905,000 (US$733,000) received by the Company from certain selling shareholders for the Company’s arrangement of the secondary offering in March 2006 (Note 21(g)). This other income was to compensate the time, resources and effort of the Company’s management in facilitating the secondary offering.
| (19) | Dividends |
Pursuant to a resolution passed at two Directors’ meetings of Beijing Yuande in January and February 2004, final dividends of RMB0.9735 and RMB0.4198 per share totalling RMB39,192,000 in respect of the statutory years 2001 and 2002, and RMB28,171,000 in respect of the statutory year 2003, were declared during the year
F-21
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
ended March 31, 2004, respectively. Shareholders approved the resolutions at two meetings in February and March 2004, respectively. No dividend in respect of the statutory year 2004 and 2005 was declared by the board of Beijing Yuande as of the balance sheet date of March 31, 2005 and 2006.
| (20) | Income Taxes |
Cayman Islands Tax
Under the current laws of the Cayman Islands, the Company is not subject to tax on income or capital gain. In addition, upon payments of dividends by the Company to its shareholders, no Cayman Islands withholding tax is imposed.
PRC Tax
The normal income tax rate for foreign-invested manufacturing enterprises located in an approved economic-technological development area, such as Beijing Yuande’s location, is 15%. As Beijing Yuande is a foreign owned enterprise and high technology enterprise with operations in an economic-technological development area in the PRC, it is eligible for an exemption from income tax for three years starting from the first profitable year of operations and thereafter a 50 percent relief from income tax for the following three years under the Income Tax Law of the PRC (the “tax holiday”). Beijing Yuande generated taxable income during its statutory year ended December 31, 1999 and was entitled to full income tax exemption until December 31, 2001. The statutory income tax rate was 15% under the relevant PRC income tax laws and Beijing Yuande was entitled to 50% exemption from income tax for statutory years ended December 31, 2002, 2003 and 2004.
On February 8, 2006, Beijing Yuande, being a “foreign-invested advanced technology enterprise” accredited by Beijing Commerce Bureau, was granted by the tax authority a reduction from 15% to 10% in its PRC income tax rate for the period from January 1, 2005 to December 31, 2007. The effect of the tax rate change was recognized in the consolidated financial statements during the year ended March 31, 2006.
U.S. Tax
Yuande (USA) Corp, a subsidiary of Beijing Yuande, was incorporated in January 2005 and is subject to income taxes in the U.S., which has a corporate tax rate of 35% and in the state of California, which has a corporate tax rate of 8.84%.
The components of income (loss) before tax are as follows:
| 2004 | 2005 | 2006 | 2006 | ||||||||
| RMB’000 | RMB’000 | RMB’000 | RMB’000 | ||||||||
PRC | 66,494 | 130,915 | 220,757 | 27,536 | |||||||
Other countries | — | (618 | ) | (3,656 | ) | (456 | ) | ||||
| 66,494 | 130,297 | 217,101 | 27,080 | ||||||||
F-22
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
Income tax expense (benefit) consists of:
| Current | Deferred | Total | |||||
| RMB’000 | RMB’000 | RMB’000 | |||||
Year ended March 31, 2006 | |||||||
PRC Income tax | 19,622 | (1,534 | ) | 18,088 | |||
U.S Tax. | — | — | — | ||||
| 19,622 | (1,534 | ) | 18,088 | ||||
Year ended March 31, 2005 | |||||||
PRC Income tax | 11,854 | — | 11,854 | ||||
U.S Tax. | — | — | — | ||||
| 11,854 | 11,854 | ||||||
Year ended March 31, 2004 | |||||||
PRC Income tax | 4,690 | 243 | 4,933 | ||||
U.S Tax | — | — | — | ||||
| 4,690 | 243 | 4,933 | |||||
Income tax expense reported in the statements of income differs from the amount computed by applying the PRC income tax rate of 15% for the three years ended March 31, 2006 for the following reasons:
| 2004 | 2005 | 2006 | 2006 | |||||||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||||||
Income before tax | 66,494 | 130,297 | 217,101 | 27,080 | ||||||||
Computed “expected” tax expense (note (a)) | 9,974 | 19,545 | 32,565 | 4,062 | ||||||||
Non-deductible expenses | 398 | 619 | 1,875 | 233 | ||||||||
Non-taxable income | (1,062 | ) | (2,443 | ) | (4,012 | ) | (500 | ) | ||||
Tax holiday | (4,538 | ) | (5,749 | ) | (10,353 | ) | (1,291 | ) | ||||
Retroactive change in enacted tax rate | — | — | (2,567 | ) | (320 | ) | ||||||
Valuation allowance | — | — | 898 | 112 | ||||||||
Foreign tax rate differential | — | (25 | ) | (248 | ) | (31 | ) | |||||
Other | 161 | (92 | ) | (70 | ) | (9 | ) | |||||
Actual income tax expense | 4,933 | 11,854 | 18,088 | 2,256 | ||||||||
| (a) | The PRC income tax rate has been used since substantially all of the Group’s operations and taxable income are generated in the PRC. |
The significant components of deferred income tax benefit attributable to income from continuing operations for the years ended March 31, 2004, 2005 and 2006 are as follows:
| 2004 | 2005 | 2006 | 2006 | |||||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||||
Deferred tax benefit (exclusive of the effect of the component below) | — | — | (1,534 | ) | (191 | ) | ||||
The benefit of operating loss carryforwards | 243 | — | — | — | ||||||
| 243 | — | (1,534 | ) | (191 | ) | |||||
F-23
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
The tax effect of temporary differences that gives rise to significant portions of the deferred tax assets and deferred tax liabilities are presented below:
| 2005 | 2006 | 2006 | ||||||
| RMB’000 | RMB’000 | USD’000 | ||||||
Deferred tax assets: | ||||||||
Operating loss carryforwards | — | 898 | 112 | |||||
Intangible assets, net | — | 1,534 | 191 | |||||
Total gross deferred tax assets | — | 2,432 | 303 | |||||
Less: valuation allowance | — | (898 | ) | (112 | ) | |||
Net deferred tax assets | — | 1,534 | 191 | |||||
In assessing the realizability of deferred tax assets, management considers whether it is more likely than not that some portion or all of the deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers the scheduled reversal of deferred tax liabilities, projected future taxable income, and tax planning strategies in making this assessment. Based upon the level of historical taxable income and projections for future taxable income over the periods in which the deferred tax assets are deductible or can be utilized, management has provided a valuation allowance of RMB898,000 (US$112,000) as of March 31, 2006. After reducing the deferred tax assets by the valuation allowance, the remaining net amount of RMB1,534,000 (US$191,000) represents the tax benefits of the Company that have been profitable and of those that are expected to be realized in the foreseeable future. The management believes it is more likely than not that the Company will realize the benefits of these net deductible differences at March 31, 2006. The amount of the deferred tax asset considered realizable, however, could be reduced in the near term if estimates of future taxable income during the carryforward period are reduced.
At March 31, 2006, the Company has net operating loss carryforwards for U.S. Federal and state income tax purposes of RMB2,049,000 (US$256,000) which are available to offset future U.S. taxable federal and state income, if any, through 2025 for the U.S. Federal net operating loss carryforwards and 2010 for state net operating loss carryforwards.
If Beijing Yuande was not in a tax holiday period for the years ended March 31, 2004, 2005 and 2006, income tax expense and earnings per share amounts would be as follows:
| 2004 | 2005 | 2006 | 2006 | |||||||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||||||
Income tax expense | 9,471 | 17,603 | 31,088 | 3,878 | ||||||||
Basic and diluted earnings per share | RMB | 0.29 | RMB | 0.56 | RMB | 0.75 | US$ | 0.09 | ||||
F-24
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (21) | Related Party Transactions |
The Group has undertaken significant business transactions in the ordinary course of business with related parties in the PRC during the three years ended March 31, 2004, 2005 and 2006.
| 2004 | 2005 | 2006 | 2006 | |||||
| RMB’000 | RMB’000 | RMB’000 | US$’000 | |||||
Sales of medical equipment(a) | 11,966 | 4,145 | 3,932 | 490 | ||||
Purchase of raw materials(b) | 6,820 | — | — | — | ||||
Provision of repairing services(c) | — | 3 | 145 | 18 | ||||
Purchase of building and land use rights(d) | 4,807 | — | — | — | ||||
Purchase of ECLIA System(e) | — | 250,000 | — | �� | — | |||
Arrangement fee income received from selling shareholders(g) | — | — | 5,905 | 733 | ||||
| (a) | The Group sold six, two and two units of products to PUPH and GEMS Trade and Deve (Shanghai) Co., Ltd., a related company of GE, in 2004, 2005 and 2006 respectively. |
The Group has significant transactions and relationships with GEMS Trade and Deve (Shanghai) Co., Ltd., a related company of GE which is a shareholder of the Company and Beijing Yuande, during the respective years. Because of this relationship, it is possible that the terms of these transactions are not the same as those that would result from transactions with third parties. Sales of medical equipment to GE Shanghai are generally at prices which are less than prices charged to third parties
| (b) | The Group purchases ultrasound imaging system used in the assembly of products from a related company of GE. As of March 31, 2005 and 2006, no amounts were due to the related company respectively. |
| (c) | The Group signed a service agreement with GE for the provision of repairing service to the customers of this related company. Expenses incurred during the provision of services are reimbursed from GE. In addition, a repairing service income, representing 10% mark-up on the expenses incurred, is chargeable and receivable from GE. The service agreement expired on December 31, 2005. |
| (d) | On February 27, 2004, the Group signed a sale and purchase agreement with Beijing Weixiao, a related party controlled by Mr. Wu Xiaodong, for the purchase of a building for a purchase consideration of RMB64,807,000. A prepayment of RMB60,000,000 was made in March 2003. The remaining balance on the purchase of the building has been reflected as amounts due to a related party in the balance sheet as of March 31, 2004 and subsequently paid in April 2004. |
| (e) | In August 2004, the Group purchased from Beijing Weixiao, a related party controlled by Mr. Wu Xiaodong, the ECLIA System. |
The ECLIA System is designed to detect and monitor various disorders through laboratory evaluation and analysis of blood, urine or other body fluids. The Group currently offers different types of reagent kits for use with the ECLIA System analyzer for various thyroid disorders, diabetes, disorders related to reproduction and growth, various types of tumors and various infectious diseases.
The ECLIA System was launched to the market in September 2004.
Under the acquisition agreement, the Group agreed to pay Beijing Weixiao an aggregate purchase price of RMB250 million for the ECLIA System technology and related inventory, of which RMB150 million was paid as of March 31, 2005 and the remaining RMB100 million to be paid by September 30,
F-25
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
2005. The aggregate purchase price of RMB250 million was allocated to inventories (RMB1.6 million) and intangible assets (RMB248.4 million) (see Note 8). In September 2005, the remaining RMB100 million was paid to Beijing Weixiao.
| (f) | Mr. Wu Xiaodong is the Chairman of the Board of Directors, the Chief Executive Officer and a significant shareholder of the Company as of March 31, 2005 and 2006. |
| (g) | On March 22, 2006, the Company completed a secondary public offering of 57,500,000 shares held by certain existing shareholders. In this offering, the selling shareholders received all net proceeds from the sale of shares sold and all of the expenses, including underwriting discounts and commissions, related to this offering were shared by the selling shareholders. During the year ended March 31, 2006, the Company received RMB10,422,000 (US$1,300,000) from the selling shareholders to pay the expenses on behalf of them and the amount was recorded in other payables. During March 2006, RMB802,000 (US$100,000) was paid and the outstanding balance as of March 31, 2006 was RMB9,620,000 (US$1,200,000) (Note 10(a)). |
Upon the completion of the secondary offering, the Company received Rmb5,905,000 (US$733,000) from certain selling shareholders for the Company’s arrangement of the secondary offering and this amount was recorded in other income (Note 18) in the consolidated financial statements. This other income was to compensate the time, resources and effort of the Company’s management in facilitating the secondary offering.
Amounts due to related parties
| Note | 2005 | 2006 | 2006 | |||||
| RMB’000 | RMB’000 | US$’000 | ||||||
Current liabilities | ||||||||
Amounts due to: | ||||||||
Beijing Weixiao—non-trade | (i) | 100,000 | — | — | ||||
GE—non-trade | (ii) | 421 | — | — | ||||
| 100,421 | — | — | ||||||
Non-current liability | ||||||||
Amount due to: | ||||||||
Shareholder—non-trade | (iii) | 2,293 | — | — | ||||
Amounts due to related parties comprise mainly:
| (i) | remaining balance payable to Beijing Weixiao for the purchase of the ECLIA System in August 2004. The amount due was fully paid during the year ended March 31, 2006. |
| (ii) | advance payment from GE in respect of the provision of repairing service to the customers of this related company. The amount was paid upon the termination of the repairing service agreement on December 31, 2005. The balances due were unsecured, non-interest bearing and repayable within one year. |
| (iii) | payables to Chengxuan International for the payment of expenses in connection with the preparation of the IPO and operating expenses incurred on behalf of the Company. Chengxuan International is a company controlled by Mr. Wu Xiaodong. The amount was repaid during the year ended March 31, 2006. The balances due were unsecured, non-interest bearing and have no fixed terms of repayment. |
F-26
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (22) | Segment Information |
The Group’s principal operating segments coincide with the types of products to be sold. The products from which revenues are derived are consistent with the reporting structure of the Group’s internal organization. The Group has two reportable segments: HIFU System and ECLIA System.
The Group’s segment information for the three years ended March 31, 2004, 2005 and 2006 is prepared using the following methodology:
| • | Revenues and expenses directly associated with each segment are included in determining segment income. |
| • | Expenses not directly associated with specific segments are allocated based on the most relevant measures applicable, including headcount, square footage and other factors. |
| • | Segment assets are based on those directly associated with each segment, and include an allocation of certain assets relating to segment and general use, based on the most relevant measures applicable, including headcount, square footage and other factors. |
F-27
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
The accounting policies of the segments are the same as those used by the Group.
| HIFU System | ECLIA System | Total | |||||||
| RMB’000 | RMB’000 | RMB’000 | |||||||
Year ended March 31,2006 | |||||||||
Net sales to external customers | 246,878 | 124,889 | 371,767 | ||||||
Cost of revenues | (72,849 | ) | (37,645 | ) | (110,494 | ) | |||
Gross profit | 174,029 | 87,244 | 261,273 | ||||||
Operating expenses | (32,298 | ) | (12,335 | ) | (44,633 | ) | |||
Segment income | 141,731 | 74,909 | 216,640 | ||||||
Reconciling items (note (a)) | (23,377 | ) | |||||||
Operating income | 193,263 | ||||||||
Other income | 9,790 | ||||||||
Interest income | 14,048 | ||||||||
Income before tax | 217,101 | ||||||||
Segment assets (noted (b)) | 142,601 | 352,435 | 495,036 | ||||||
Expenditures for long-lived assets | (22,608 | ) | (10,500 | ) | (33,108 | ) | |||
Depreciation and amortization | (786 | ) | (15,772 | ) | (16,558 | ) | |||
Year ended March 31,2005 | |||||||||
Net sales to external customers | 177,275 | 40,272 | 217,547 | ||||||
Cost of revenues | (49,539 | ) | (15,040 | ) | (64,579 | ) | |||
Gross profit | 127,736 | 25,232 | 152,968 | ||||||
Operating expenses | (17,609 | ) | (3,873 | ) | (21,482 | ) | |||
Segment income | 110,127 | 21,359 | 131,486 | ||||||
Reconciling items (note (a)) | (2,937 | ) | |||||||
Operating income | 128,549 | ||||||||
Other income | 1,559 | ||||||||
Interest income | 194 | ||||||||
Interest expense | (5 | ) | |||||||
Income before tax | 130,297 | ||||||||
Segment assets (note (b)) | 96,805 | 275,293 | 372,098 | ||||||
Expenditures for long-lived assets | (3,357 | ) | (248,724 | ) | (252,081 | ) | |||
Depreciation and amortization | (817 | ) | (9,133 | ) | (9,950 | ) | |||
F-28
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| HIFU System | ECLIA System | Total | ||||||
| RMB’000 | RMB’000 | RMB’000 | ||||||
Year ended March 31,2004 | ||||||||
Net sales to external customers | 110,750 | — | 110,750 | |||||
Cost of revenues | (33,983 | ) | — | (33,983 | ) | |||
Gross profit | 76,767 | — | 76,767 | |||||
Operating expenses | (10,122 | ) | — | (10,122 | ) | |||
Segment income | 66,645 | — | 66,645 | |||||
Reconciling items (note (a)) | (165 | ) | ||||||
Operating income | 66,480 | |||||||
Interest income | 126 | |||||||
Interest expense | (112 | ) | ||||||
Income before tax | 66,494 | |||||||
Segment assets (note (b)) | 98,097 | n/a | 98,097 | |||||
Expenditures for long-lived assets | (513 | ) | — | (513 | ) | |||
Depreciation and amortization | (1,291 | ) | — | (1,291 | ) | |||
| (a) | Reconciliation of total segment income to consolidated operating income |
| March 31, | |||||||||
| 2004 | 2005 | 2006 | |||||||
| RMB’000 | RMB’000 | RMB’000 | |||||||
Total segment income | 66,645 | 131,486 | 216,640 | ||||||
Depreciation on property, plant and equipment | (165 | ) | (2,131 | ) | (2,142 | ) | |||
Others | — | (806 | ) | (21,235 | ) | ||||
Consolidated total operating income | 66,480 | 128,549 | 193,263 | ||||||
| (b) | Reconciliation of total segment assets to consolidated total assets |
| March 31, | ||||||
| 2004 | 2005 | 2006 | ||||
| RMB’000 | RMB’000 | RMB’000 | ||||
Total assets for reportable segments | 98,097 | 372,098 | 495,036 | |||
Others:- | ||||||
Cash and cash equivalents | 24,114 | 14,646 | 843,791 | |||
Property, plant and equipment (note (i)) | 64,232 | 54,690 | 28,289 | |||
Other assets (note (ii)) | 5,718 | 16,334 | 8,725 | |||
Consolidated total assets | 192,161 | 457,768 | 1,375,841 | |||
| (i) | Property, plant and equipment comprise of certain fixed assets, or allocated portions thereof that are not uniquely identifiable with the operations of particular segment. |
| (ii) | Other assets primarily include prepayments and other receivables and lease prepayments. |
F-29
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
In view of the fact that the Group operates and manages its business substantially in the PRC, no geographical segment information is presented.
| (23) | Pension and Other Postretirement Benefits |
Pursuant to the relevant PRC regulations, Beijing Yuande is required to make contributions at approximately 19% of the employees’ salaries and wages to a defined contribution retirement scheme organized by the Beijing Social Security Bureau in respect of the retirement benefits for Beijing Yuande’s employees in the PRC. The contributions for the years ended March 31, 2004, 2005 and 2006 amounted to RMB186,000, RMB271,000 and RMB909,000 (US$113,000) respectively.
The Group has no other obligation to make payments in respect of retirement benefits of its employees.
| (24) | Derivative Instruments and Hedging Activities |
The Group did not enter into any derivative financial instruments for any purpose during the years presented. The Group does not hedge risk exposures or speculate using derivative instruments.
| (25) | Fair Value of Financial Instruments |
The fair value of a financial instrument is the amount at which the instrument could be exchanged in a current transaction between willing parties. The carrying amount of financial assets, such as cash and cash equivalents, trade accounts receivable, other receivables, trade accounts payable and other payables, approximates their fair values because of the short term maturity of these instruments. The Group does not hold nor issue financial instruments for trading purposes.
| (26) | Business and Credit Concentrations |
(a) All of the Group’s customers are located in the PRC. The following are the customers that individually comprised 10% or more of revenue in any year during the relevant periods:
| 2004 | 2005 | 2006 | 2006 | |||||||||||
| RMB’000 | % | RMB’000 | % | RMB’000 | % | US$’000 | ||||||||
Beijing Jia Xing Fang Zhou Trade Co., Ltd. | 25,641 | 23 | — | — | — | |||||||||
Kunshan Creation Industry Development Co., Ltd. | 15,385 | 14 | 30,769 | 14 | 25,641 | 7 | 3,198 | |||||||
Ningbo Ruichang Industry Co., Ltd. | 15,385 | 14 | 12,821 | 6 | 12,821 | 3 | 1,599 | |||||||
Shanghai Silai Trade Co., Ltd. | 9,479 | 8 | 25,641 | 12 | 30,769 | 8 | 3,838 | |||||||
Shanghai Yi Li System Technology Co., Ltd. | 12,820 | 12 | — | — | — | |||||||||
GEMS Trade and Deve (Shanghai) Co., Ltd. | 11,966 | 11 | 4,145 | 2 | 3,932 | 1 | 490 | |||||||
Beijing Wei Jian Da Medical Technology Co., Ltd. | — | 51,282 | 24 | 91,776 | 25 | 11,448 | ||||||||
Beijing Yi Min Wei Kang Technology Co., Ltd. | — | 16,427 | 8 | 42,023 | 11 | 5,242 | ||||||||
The Group had two customers which accounted for thirty-six percent of revenue for the year ended March 31, 2006, three customers which accounted for fifty percent of revenue for the year ended March 31, 2005 and five customers which accounted for seventy-four percent of revenue for the year ended March 31,
F-30
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
2004, respectively. The Group performs ongoing credit evaluations of its customers’ financial condition and, generally, requires advanced deposits as collateral from its customers.
(b) The Group is dependent on single-source suppliers for much of the materials and major components used to manufacture its products. If the supply of certain materials or components were interrupted, the Group’s own manufacturing process could be delayed and could cause a possible loss of sales, which would adversely affect operating results. The ultrasound imaging system, one of the major components of products sold, is manufactured by a related company of GE and purchased from a local supplier while the treatment bed, another major component of products sold, is manufactured by a local supplier. Purchases (net of VAT) made from a related company of GE for imaging system for the years ended March 31, 2004, 2005 and 2006 were RMB5,829,000, RMBNil and RMBNil (US$Nil), respectively. Purchases (net of VAT) made from the two local suppliers for the ultrasound imaging system and treatment bed for the years ended March 31, 2004, 2005 and 2006 were RMB22,248,000, RMB45,509,000 and RMB53,288,000 (US$6,647,000), respectively.
(c) Accounts receivable from the three and five customers, who individually comprise 10% or more of the outstanding balance, represented 62% and 68%, respectively, of the balance of the account at March 31, 2005 and 2006, respectively.
| (27) | Stock option plan |
On February 2, 2005, the Company adopted the 2005 stock option plan (the “Plan”) pursuant to which the Company’s Board of Directors may grant stock options to directors and employees. Under the Plan, the Company has reserved an aggregate of 30,000,000 of its ordinary shares for issuance under the Plan. The Plan will remain in effect for 5 years starting from the date of adoption.
On March 14, 2005, the Company granted two directors options to purchase an aggregate of 400,000 ordinary shares at an exercise price of US$1.36 per share. On August 8, 2005, the Company granted two other directors options to purchase an aggregate of 400,000 ordinary shares at an exercise price of US$1.5 per share. One-third of the options will become exercisable one year after the Vesting Commencement Date (the “Date”), another one-third will become exercisable 18 months after the Date and the remaining one-third will become exercisable two years after the Date. All outstanding options will expire on December 31, 2009.
Stock option activity during the years indicated is as follows:
| Number of shares | Weighted average exercise price | Weighted average contractual life | ||||
| ‘000 | US$ | Years | ||||
Balance at April 1, 2004 | — | — | ||||
Granted | 400 | 1.36 | ||||
Balance at March 31, 2005 | 400 | 1.36 | 4.75 | |||
Granted | 400 | 1.50 | ||||
Balance of March 31, 2006 | 800 | 1.43 | 3.75 | |||
F-31
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
The fair value of the options granted is estimated on the date of grant using the dividend adjusted Black-Scholes option-pricing model based on the following assumptions:
Option grants | ||
Average risk-free interest rate of return | 3.911% | |
Weighted average expected option life | 2.75 years | |
Volatility rate | 30.00% | |
Dividend yield | 0.00% |
The fair value of the Company’s ordinary shares issuable under the options granted on March 14, 2005 was determined by the Company with reference to the estimated price range of US$1.40 to US$1.60 per ordinary share and with discussions with the underwriter of the Company’s IPO. Accordingly, the Company considered US$1.50 per ordinary share, the midpoint of the estimated range of the IPO price, to be an appropriate indicator of the fair value of its ordinary shares. Accordingly, share-based compensation of RMB464,000 should be recorded, which represents the difference between the exercise price of US$1.36 per ordinary share and the estimated fair value of US$1.50 per ordinary share in respect of 400,000 ordinary shares issuable under the options granted on March 14, 2005. Deferred share-based compensation expenses should be amortized on a straight-line basis over the two-year vesting period. Amortization of this share-based compensation for the year ended March 31, 2006 was RMB238,000 (US$30,000).
The fair value of the Company’s ordinary shares issuable under the options granted on August 8, 2005 was determined by the Company with reference to the estimated price range of US$1.40 to US$1.60 per ordinary share and with discussions with the underwriter of the Company’s IPO. Accordingly, no share-based compensation was recorded as the exercise price of US$1.50 per ordinary share is the same as the estimated fair value of US$1.50 per ordinary share in respect of 400,000 ordinary shares issuable under the options granted on August 8, 2005
Options to purchase 133,000 ordinary shares were exercisable as of March 31, 2006 and the weighted average remaining contractual life is 3.75 years. No options were forfeited during the year ended March 31, 2006.
| (28) | Purchase obligations |
The Group has entered into an agreement with a contractor for carrying out certain construction works for a contract sum of RMB48,000,000 (US$5,988,000). As of March 31, 2006, a payment of RMB10,000,000 (US$1,247,000) was made to the contractor.
| (29) | Operating lease obligations |
Rental expense for obligations under operating leases was RMB Nil, RMB219,000 and RMB1,268,000 (US$158,000), respectively, for the years ended March 31, 2004, 2005 and 2006. As of March 31, 2006, the total future minimum lease payments under non-cancellable operating leases are payable as follows:
| RMB’000 | ||
Fiscal year ending March 31, | ||
2007 | 1,433 | |
2008 | 1,028 | |
2009 | 553 | |
| 3,014 | ||
F-32
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
| (30) | Other risks |
Beijing Yuande’s sales, purchase and expense transactions are denominated in RMB and its assets and liabilities are denominated in RMB. The RMB is not freely convertible into foreign currencies. In PRC, foreign exchange transactions are required by law to be transacted only by authorised financial institutions at exchange rates set by the People’s Bank of China (“PBOC”). Remittances in currencies other than RMB by Beijing Yuande must be processed through the PBOC or other PRC foreign exchange regulatory bodies and require certain supporting documentation in order to effect the remittance.
| (31) | Additional financial information |
Non-cash transactions
On February 27, 2004, Beijing Yuande signed a sale and purchase agreement for the purchase of a building from Beijing Weixiao, a related party controlled by Mr. Wu Xiaodong, for a purchase consideration of RMB64,807,000. Part of the consideration was settled by the repayment of RMB60,000,000 made in March 2003. The remaining balance on the purchase of the building has been reflected as amounts due to Beijing Weixiao in the balance sheet as of March 31, 2004.
In August 2004, Beijing Yuande acquired certain intangible assets and related inventory for RMB250 million from Beijing Weixiao, of which RMB150 million has been paid as of March 31, 2005. The remaining RMB100 million payable is reflected as an amount due to a related party with an offsetting increase to intangible assets in the balance sheet as of March 31, 2005 (see Note 21).
| (32) | China Medical Technologies, Inc. (the Company only) |
Under PRC regulations, foreign-invested enterprises in China may pay dividends only out of their accumulated profits, if any, determined in accordance with PRC accounting standards and regulations. In addition, the Company’s wholly owned subsidiary, Beijing Yuande is required to set aside at least 10% of its respective accumulated profits each year, if any, to fund certain reserve funds unless such reserve funds have reached 50% of its respective registered capital. These reserves are not distributable in the form of cash dividends to the Company.
As of March 31, 2006, Beijing Yuande had no third-party loans or borrowing arrangements or any amounts outstanding. In addition, the Company has no borrowings from Beijing Yuande.
As of March 31, 2006, the amount of restricted net assets of Beijing Yuande, which may not be transferred to the Company in the forms of loans, advances or cash dividends without the consent of PRC government authorities, was more than 25% of the Company’s consolidated net assets as discussed above.
F-33
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
The following presents condensed financial information of the Company only.
Condensed Balance Sheets
| As of March 31, | ||||||||
| 2005 | 2006 | 2006 | ||||||
| RMB’000 | RMB’000 | US$’000 | ||||||
Current assets | ||||||||
Cash and cash equivalents | — | 520,969 | 64,986 | |||||
Prepayments and other receivables | 7,902 | 1,732 | 216 | |||||
Amount due from a subsidiary | — | 51,367 | 6,407 | |||||
Total current assets | 7,902 | 574,068 | 71,609 | |||||
Investments in subsidiaries | 269,781 | 710,958 | 88,685 | |||||
Total assets | 277,683 | 1,285,026 | 160,294 | |||||
Liabilities | ||||||||
Current liabilities | ||||||||
Accrued liabilities and other payables | 4,120 | 22,635 | 2,824 | |||||
Amount due to a related party | 4,130 | — | — | |||||
Amount due to a subsidiary | — | 348 | 43 | |||||
Total liabilities | 8,250 | 22,983 | 2,867 | |||||
Shareholders’ equity | ||||||||
Share capital | 165,530 | 225,125 | 28,082 | |||||
Ordinary shares US$0.10 par value: 500,000,000 authorized; 200,000,001 and 273,600,001 issued and outstanding as of March 31, 2005 and 2006, respectively | ||||||||
Additional paid-in capital | — | 739,936 | 92,299 | |||||
Unearned compensation | — | (226 | ) | (28 | ) | |||
Accumulated other comprehensive loss | — | (5,710 | ) | (712 | ) | |||
Retained earnings | 103,903 | 302,918 | 37,786 | |||||
Total shareholders’ equity | 269,433 | 1,262,043 | 157,427 | |||||
Total liabilities and shareholders’ equity | 277,683 | 1,285,026 | 160,294 | |||||
No financial statements of the Company for the year ended March 31, 2004 were presented as the Company was incorporated in July, 2004. The Company had no contingencies, long-term obligations and guarantees as of March 31, 2005 and 2006.
F-34
Table of Contents
CHINA MEDICAL TECHNOLOGIES, INC.
Notes to the Consolidated Financial Statements
March 31, 2004, 2005 and 2006, continued
(expressed in RMB and US$)
Condensed Statements of Income
| For the period from July 9, 2004 (date of incorporation) to March 31, 2005 | For the year ended March 31, 2006 | ||||||||
| RMB’000 | RMB’000 | US$’000 | |||||||
Interest income | — | 12,261 | 1,529 | ||||||
Other non-operating income | — | 5,905 | 737 | ||||||
Share of net income of subsidiaries | 104,251 | 200,854 | 25,054 | ||||||
General and administrative expenses | (348 | ) | (20,007 | ) | (2,496 | ) | |||
Income before tax | 103,903 | 199,013 | 24,824 | ||||||
Income tax expense | — | — | — | ||||||
Net income | 103,903 | 199,013 | 24,824 | ||||||
Condensed Statements of Cash Flows
| For the period from July 9, 2004 (date of incorporation) to March 31, 2005 | For the year ended March 31, 2006 | ||||||||
| RMB’000 | RMB’000 | US$’000 | |||||||
Cash flow from operating activities | |||||||||
Net income | 103,903 | 199,013 | 24,824 | ||||||
Adjustments to reconcile net income to net cash used in operating activities: | |||||||||
Amortization of share-based compensation | — | 238 | 30 | ||||||
Share of net income of subsidiaries | (104,251 | ) | (200,854 | ) | (25,054 | ) | |||
Changes in assets and liabilities: | |||||||||
Prepayments and other receivables | (7,902 | ) | 6,170 | 770 | |||||
Amount due from a subsidiary | — | (51,367 | ) | (6,407 | ) | ||||
Accrued liabilities and other payables | 4,120 | 8,597 | 1,072 | ||||||
Amount due to a related party | 4,130 | (4,130 | ) | (515 | ) | ||||
Amount due to a subsidiary | — | 348 | 43 | ||||||
Net cash used in operating activities | — | (41,985 | ) | (5,237 | ) | ||||
Cash flow from investing activities | |||||||||
Investments in subsidiaries | — | (240,323 | ) | (29,977 | ) | ||||
Net cash used in investing activities | — | (240,323 | ) | (29,977 | ) | ||||
Cash flow from financing activities | |||||||||
Advances from selling shareholders in secondary offering | — | 10,422 | 1,300 | ||||||
Payment for secondary offering expenses of selling shareholders | — | (802 | ) | (100 | ) | ||||
Net proceeds from issue of new shares | — | 799,067 | 99,675 | ||||||
Net cash provided by financing activities | — | 808,687 | 100,875 | ||||||
Effect of foreign currency exchange rate change on cash | — | (5,410 | ) | (675 | ) | ||||
Net increase in cash and cash equivalents | — | 520,969 | 64,986 | ||||||
Cash and cash equivalents: | |||||||||
At beginning of period/year | — | — | — | ||||||
At end of period/year | — | 520,969 | 64,986 | ||||||
F-35