| Forward-Looking Statements This presentation includes, and our response to various questions may include, forward-looking statements. All statements contained in this presentation other than statements of historical facts, including statements regarding our future results of operations and financial position, our business strategy and plans and our objectives for future operations, are forward-looking statements. The words “anticipate,” “believe,” “continue,” “estimate,” “expect,” “intend,” “may,” “will” and similar expressions are intended to identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our financial condition, results of operations, business strategy, short-term and long-term business operations and objectives and financial needs. These forward-looking statements, including statements regarding clinical trials, clinical studies and other clinical work (including the funding therefor, anticipated patient enrollment, safety data, study data, trial outcomes, timing or associated costs), regulatory applications and related timelines, including the filing of an NDA for LIQ861, are subject to a number of risks, uncertainties and assumptions. Moreover, we operate in a very competitive and rapidly changing environment and our industry has inherent risks. New risks emerge from time to time. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the future events and trends discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance, achievements or events and circumstances reflected in the forward-looking statements will occur. We are under no duty to update any of these forward-looking statements after the date of this presentation to conform these statements to actual results or revised expectations, except as required by law. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. This presentation includes long-term goals that are forward-looking, are subject to significant business, economic, regulatory and competitive uncertainties and contingencies, many of which are beyond the control of us and our management, and are based upon assumptions with respect to future decisions, which are subject to change. Actual results will vary and those variations may be material. Nothing in this presentation should be regarded as a representation by any person that these goals will be achieved and we undertake no duty to update our goals. |
| Novel products via precise control of drug particles Late-stage clinical biopharmaceutical company focused on transforming the lives of patients Pulmonary Arterial Hypertension (PAH): New Drug Application (NDA); Decision Resources Group, Landscape & Forecast, PAH, Nov 2018 Preparing NDA LIQ861: inhaled dry powder targeting segment of PAH market ($3.7B U.S.) Completed clinical program and preparing NDA submission Pipeline Growth LIQ865: to manage local, post-operative pain for 3-5 days (Phase 1) Poised to expand PRINT Technology advantages into future products Proprietary Technology Broadly applicable across therapeutic areas, modalities and routes of delivery Fully scaled PRINT® platform offers multiple product advantages LIQ861 |
| LIQ861 for PAH PRINT® treprostinil, dry powder inhalation |
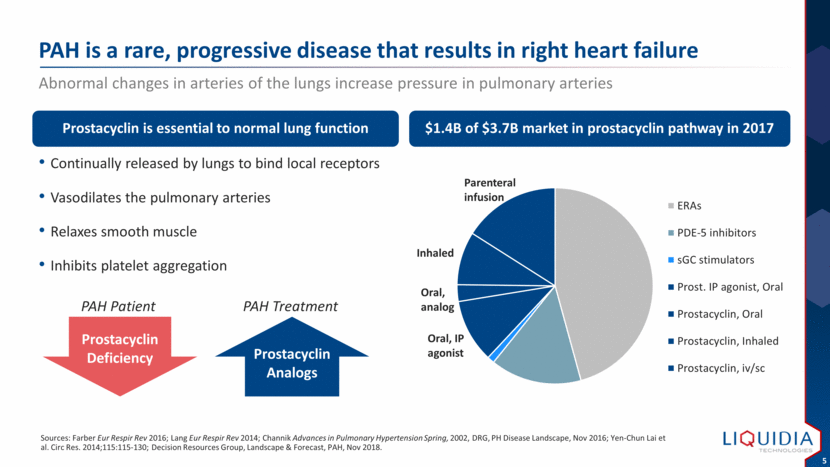
| Continually released by lungs to bind local receptors Vasodilates the pulmonary arteries Relaxes smooth muscle Inhibits platelet aggregation PAH is a rare, progressive disease that results in right heart failure Sources: Farber Eur Respir Rev 2016; Lang Eur Respir Rev 2014; Channik Advances in Pulmonary Hypertension Spring, 2002, DRG, PH Disease Landscape, Nov 2016; Yen-Chun Lai et al. Circ Res. 2014;115:115-130; Decision Resources Group, Landscape & Forecast, PAH, Nov 2018. Prostacyclin is essential to normal lung function $1.4B of $3.7B market in prostacyclin pathway in 2017 Abnormal changes in arteries of the lungs increase pressure in pulmonary arteries Prostacyclin Deficiency Prostacyclin Analogs PAH Patient PAH Treatment Parenteral infusion Inhaled Oral, analog Oral, IP agonist ERAs PDE-5 inhibitors sGC stimulators Prost. IP agonist, Oral Prostacyclin, Oral Prostacyclin, Inhaled Prostacyclin, iv/sc |
| Goal of prostacyclin therapy is to maximize exposure to highest tolerable level Local delivery generates fewer off-tissue effects Source: Decision Resources, Pulmonary Hypertension Disease landscape & Forecast, November 2018. Current prostacyclin products have clear tradeoffs Oral = Convenient, but with systemic toxicities and minimal symptom relief Increases side effects in GI, Nervous and Vascular systems Requires up-titration that can be challenging given side effects Nebulized = Targeted, but provides limited dose range Limits max dose due to throat irritation, adverse events Requires water, power, supplies, cleaning and time to dose Infusion = Effective, but systemic toxicities & site pain, limits on lifestyle Delivers continuously via i.v. or s.c. line, 24 hours a day Poses potential for infection risk |
| Treprostinil = Proven efficacy Proven compound with FDA approvals for i.v., s.c., inhaled and oral routes PRINT® = Deep-lung delivery Delivers higher dose levels than approved inhaled formulations Device = Simple, Disposable Compact, easy inhaler with established commercial track record LIQ861 combines Effective + Targeted + Convenient into one product RS00 Model 8 (DMF # 18418) Disposable & long track record Precise, Uniform, Trefoil Trusted prostacyclin-analog RS00 Model 8 (DMF # 18418) |
| After consulting FDA, initiated Phase 3 INSPIRE pursuant to 505(b)(2) Investigation of the Safety and Pharmacology of Dry Powder Inhalation of Treprostinil Sources: https://clinicaltrials.gov/ct2/show/NCT03399604; PGI – prostacyclin; TEAEs – treatment-emergent adverse events; SAEs – serious adverse events; Quote from Nicholas Hill, MD, Chief Pulmonary, Critical Care & Sleep Division and Professor of Medicine at Tufts University School of Medicine and INSPIRE Principal Investigator. 1. Adjusting dose levels to comparable Tyvaso® emitted dose Design Open-label, U.S. multicenter Population At least 100 WHO Group I (PAH) patients; NYHA Class II, III and IV Criteria On stable dose of Tyvaso® for >3 months (or) taking <2 approved non-PGI oral PAH therapies Primary endpoint Incidence of TEAEs and SAEs at 2 months Exploratory endpoints 6 minute walk distance (6MWD) Sustained treatment transition (Tyvaso® transitions) NYHA functional class improvement Quality of life using Minnesota Living with Heart Failure Questionnaire (MLHFQ) PK Sub-Study1 Transitions from Tyvaso® in a one-directional crossover to compare bioavailability and PK Data collection Baseline, Week 2, Month 1, Month 2 Visits, with bimonthly follow up for up to 30 months We will continue to treat patients and collect data in a roll-over safety study |
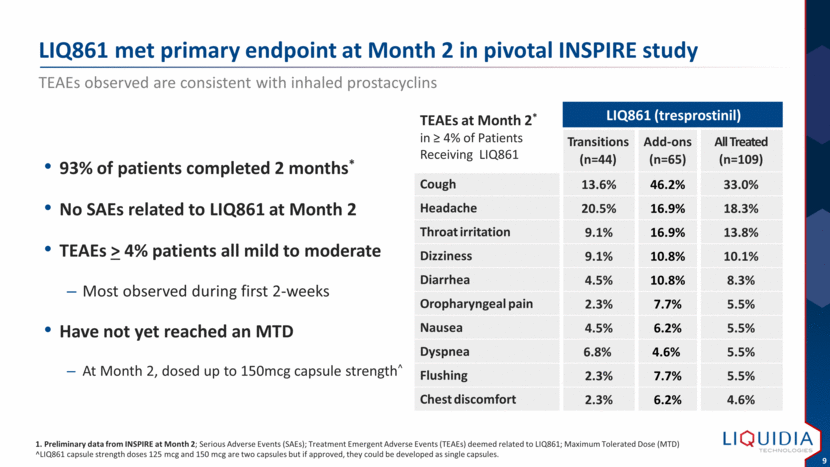
| LIQ861 met primary endpoint at Month 2 in pivotal INSPIRE study TEAEs observed are consistent with inhaled prostacyclins 1. Preliminary data from INSPIRE at Month 2; Serious Adverse Events (SAEs); Treatment Emergent Adverse Events (TEAEs) deemed related to LIQ861; Maximum Tolerated Dose (MTD) ^LIQ861 capsule strength doses 125 mcg and 150 mcg are two capsules but if approved, they could be developed as single capsules. 93% of patients completed 2 months* No SAEs related to LIQ861 at Month 2 TEAEs > 4% patients all mild to moderate Most observed during first 2-weeks Have not yet reached an MTD At Month 2, dosed up to 150mcg capsule strength^ TEAEs at Month 2* in > 4% of Patients Receiving LIQ861 LIQ861 (tresprostinil) Transitions (n=44) Add-ons (n=65) All Treated (n=109) Cough 13.6% 46.2% 33.0% Headache 20.5% 16.9% 18.3% Throat irritation 9.1% 16.9% 13.8% Dizziness 9.1% 10.8% 10.1% Diarrhea 4.5% 10.8% 8.3% Oropharyngeal pain 2.3% 7.7% 5.5% Nausea 4.5% 6.2% 5.5% Dyspnea 6.8% 4.6% 5.5% Flushing 2.3% 7.7% 5.5% Chest discomfort 2.3% 6.2% 4.6% |
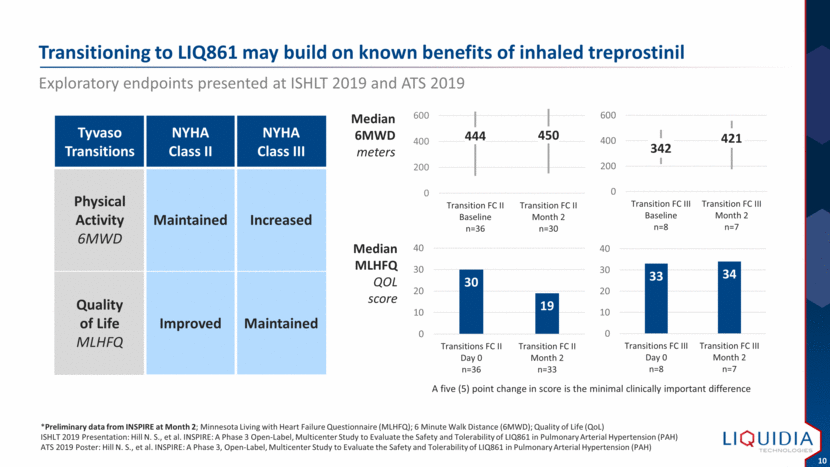
| Transitioning to LIQ861 may build on known benefits of inhaled treprostinil Exploratory endpoints presented at ISHLT 2019 and ATS 2019 *Preliminary data from INSPIRE at Month 2; Minnesota Living with Heart Failure Questionnaire (MLHFQ); 6 Minute Walk Distance (6MWD); Quality of Life (QoL) ISHLT 2019 Presentation: Hill N. S., et al. INSPIRE: A Phase 3 Open-Label, Multicenter Study to Evaluate the Safety and Tolerability of LIQ861 in Pulmonary Arterial Hypertension (PAH) ATS 2019 Poster: Hill N. S., et al. INSPIRE: A Phase 3, Open-Label, Multicenter Study to Evaluate the Safety and Tolerability of LIQ861 in Pulmonary Arterial Hypertension (PAH) Median 6MWD meters Median MLHFQ QOL score Tyvaso Transitions NYHA Class II NYHA Class III Physical Activity 6MWD Maintained Increased Quality of Life MLHFQ Improved Maintained A five (5) point change in score is the minimal clinically important difference 444 450 0 200 400 600 Transition FC II Baseline n=36 Transition FC II Month 2 n=30 342 421 0 200 400 600 Transition FC III Baseline n=8 Transition FC III Month 2 n=7 30 19 0 10 20 30 40 Transitions FC II Day 0 n=36 Transition FC II Month 2 n=33 33 34 0 10 20 30 40 Transitions FC III Day 0 n=8 Transition FC III Month 2 n=7 |
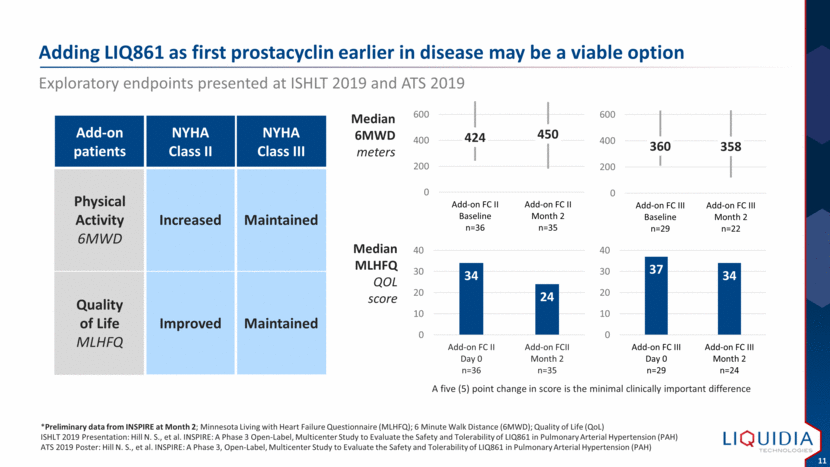
| Adding LIQ861 as first prostacyclin earlier in disease may be a viable option Exploratory endpoints presented at ISHLT 2019 and ATS 2019 *Preliminary data from INSPIRE at Month 2; Minnesota Living with Heart Failure Questionnaire (MLHFQ); 6 Minute Walk Distance (6MWD); Quality of Life (QoL) ISHLT 2019 Presentation: Hill N. S., et al. INSPIRE: A Phase 3 Open-Label, Multicenter Study to Evaluate the Safety and Tolerability of LIQ861 in Pulmonary Arterial Hypertension (PAH) ATS 2019 Poster: Hill N. S., et al. INSPIRE: A Phase 3, Open-Label, Multicenter Study to Evaluate the Safety and Tolerability of LIQ861 in Pulmonary Arterial Hypertension (PAH) Median MLHFQ QOL score Add-on patients NYHA Class II NYHA Class III Physical Activity 6MWD Increased Maintained Quality of Life MLHFQ Improved Maintained A five (5) point change in score is the minimal clinically important difference Median 6MWD meters 34 24 0 10 20 30 40 Add-on FC II Day 0 n=36 Add-on FCII Month 2 n=35 37 34 0 10 20 30 40 Add-on FC III Day 0 n=29 Add-on FC III Month 2 n=24 424 450 0 200 400 600 Add-on FC II Baseline n=36 Add-on FC II Month 2 n=35 360 358 0 200 400 600 Add-on FC III Baseline n=29 Add-on FC III Month 2 n=22 |
| LIQ865 for Local Post-Operative Pain PRINT® bupivacaine, sustained-release injectable |
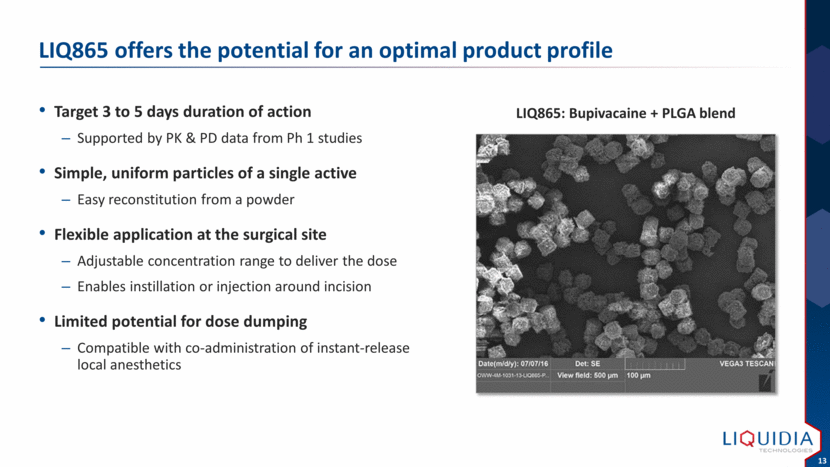
| LIQ865 offers the potential for an optimal product profile Target 3 to 5 days duration of action Supported by PK & PD data from Ph 1 studies Simple, uniform particles of a single active Easy reconstitution from a powder Flexible application at the surgical site Adjustable concentration range to deliver the dose Enables instillation or injection around incision Limited potential for dose dumping Compatible with co-administration of instant-release local anesthetics LIQ865: Bupivacaine + PLGA blend |
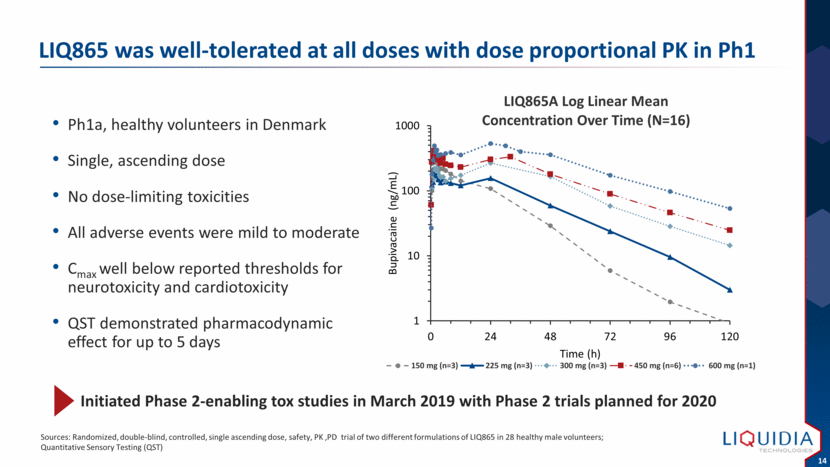
| Ph1a, healthy volunteers in Denmark Single, ascending dose No dose-limiting toxicities All adverse events were mild to moderate Cmax well below reported thresholds for neurotoxicity and cardiotoxicity QST demonstrated pharmacodynamic effect for up to 5 days LIQ865 was well-tolerated at all doses with dose proportional PK in Ph1 Sources: Randomized, double-blind, controlled, single ascending dose, safety, PK ,PD trial of two different formulations of LIQ865 in 28 healthy male volunteers; Quantitative Sensory Testing (QST) Initiated Phase 2-enabling tox studies in March 2019 with Phase 2 trials planned for 2020 1 10 100 1000 0 24 48 72 96 120 Bupivacaine (ng/mL) Time (h) LIQ865A Log Linear Mean Concentration Over Time (N=16) 150 mg (n=3) 225 mg (n=3) 300 mg (n=3) 450 mg (n=6) 600 mg (n=1) |
| PRINT® Technology |
| Compatible with nearly any material, payload and route of delivery Examples, not exhaustive PLGA Porous Hydrogel Nucleic Acid PLGA/DC-Chol Crystalline API Formulated Protein PEG Hydrogel PLGA/RNA Albumin/Lactose PLGA-Steroid Monoclonal Antibody 100 % Cyclosporine |
| PRINT® production has been scaled for clinical and commercial demands Lab Line 2 (2008) Highly agile platform enabling process experimentation Ideal for early stage process development Lab Line 3 (non-cGMP 2015; cGMP 2017) Capable of larger batches with increased process control We believe Lab Line 3 is fully cGMP compliant to support product launch Commercial Line 1 (expected 2019) Optimized drug substance production process Designed for continued market supply and scale Preclinical and R&D Highly versatile, flexible cGMP Process Development Optimization, scale-up cGMP Production Repeatable and deployable |
| Conclusion |
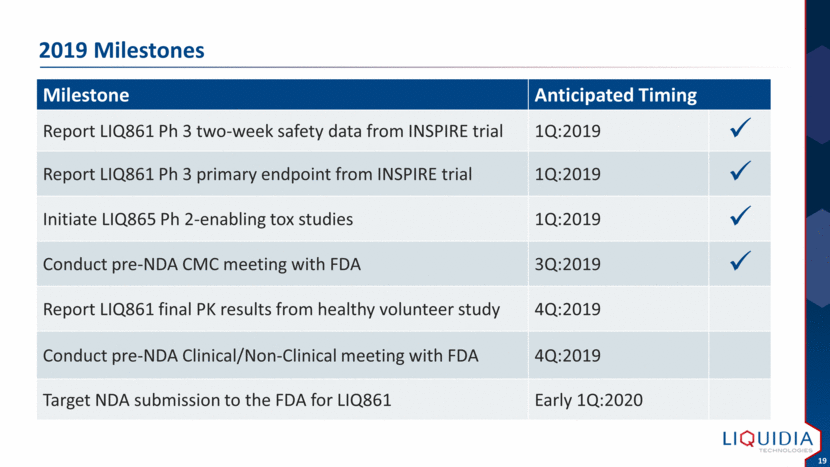
| 2019 Milestones Milestone Anticipated Timing Report LIQ861 Ph 3 two-week safety data from INSPIRE trial 1Q:2019 Report LIQ861 Ph 3 primary endpoint from INSPIRE trial 1Q:2019 Initiate LIQ865 Ph 2-enabling tox studies 1Q:2019 Conduct pre-NDA CMC meeting with FDA 3Q:2019 Report LIQ861 final PK results from healthy volunteer study 4Q:2019 Conduct pre-NDA Clinical/Non-Clinical meeting with FDA 4Q:2019 Target NDA submission to the FDA for LIQ861 Early 1Q:2020 |
| Thank You |