Table of Contents
| (Mark One) | ||
| o | REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| or | ||
| þ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the fiscal year ended December 31, 2011 | ||
or | ||
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| For the transition period from to | ||
| or | ||
| o | SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |
| Date of event requiring this shell company report | ||
Title of each class | Name of each exchange on which registered | |
American Depositary Shares representing Preference Shares | New York Stock Exchange | |
Preference Shares, no par value | New York Stock Exchange(1) | |
American Depositary Shares representing Ordinary Shares | New York Stock Exchange | |
Ordinary Shares, no par value | New York Stock Exchange(1) |
| (1) | Not for trading, but only in connection with the registration of American Depositary Shares representing such shares. |
Large accelerated filer þ | Accelerated filer o | Non-accelerated filer o |
Table of Contents
| • | changes in governmental and commercial insurer reimbursement for our complete products and services portfolio, including the expanded Medicare reimbursement system for dialysis services; | |
| • | changes in utilization patterns for pharmaceuticals and in our costs of purchasing pharmaceuticals; | |
| • | the outcome of ongoing government investigations; | |
| • | the influence of private insurers and managed care organizations; | |
| • | the impact of recently enacted and possible future healthcare reforms; | |
| • | product liability risks; | |
| • | the outcome of ongoing potentially material litigation; | |
| • | risks relating to the integration of acquisitions and our dependence on additional acquisitions; | |
| • | the impact of currency fluctuations; | |
| • | introduction of generic or new pharmaceuticals that compete with our pharmaceutical products; |
1
Table of Contents
| • | changes in raw material and energy costs; and | |
| • | the financial stability and liquidity of our governmental and commercial payors. |
2
Table of Contents
| Item 1. | Identity of Directors, Senior Management and Advisors |
| Item 2. | Other Statistics and Expected Timetable |
| Item 3. | Key Information |
| A. | Selected Financial Data |
| 2011 | 2010 | 2009 | 2008 | 2007 | ||||||||||||||||
| (in millions except share and per share amounts) | ||||||||||||||||||||
Statement of Operations Data: | ||||||||||||||||||||
| Net revenues | $ | 12,795 | $ | 12,053 | $ | 11,247 | $ | 10,612 | $ | 9,720 | ||||||||||
| Cost of revenues | 8,274 | 7,908 | 7,415 | 6,983 | 6,364 | |||||||||||||||
| Gross profit | 4,521 | 4,145 | 3,832 | 3,629 | 3,356 | |||||||||||||||
| Selling, general and administrative | 2,366 | 2,133 | 1,987 | 1,877 | 1,709 | |||||||||||||||
| Research and development | 111 | 97 | 94 | 80 | 67 | |||||||||||||||
| Income from equity method investees | (31 | ) | (9 | ) | (5 | ) | — | — | ||||||||||||
| Operating income | 2,075 | 1,924 | 1,756 | 1,672 | 1,580 | |||||||||||||||
| Interest expense, net | 297 | 280 | 300 | 336 | 371 | |||||||||||||||
| Income before income taxes | 1,778 | 1,644 | 1,456 | 1,336 | 1,209 | |||||||||||||||
| Net income attributable to shareholders of FMC-AG & Co. KGaA | $ | 1,071 | $ | 979 | $ | 891 | $ | 818 | $ | 717 | ||||||||||
| Weighted average ordinary shares outstanding | 299,012,744 | 296,808,978 | 294,418,795 | 293,233,477 | 291,929,141 | |||||||||||||||
| Basic earnings per Ordinary share and Ordinary ADS | $ | 3.54 | $ | 3.25 | $ | 2.99 | $ | 2.75 | $ | 2.43 | ||||||||||
| Fully diluted earnings per Ordinary share and Ordinary ADS | 3.51 | 3.24 | 2.99 | 2.75 | 2.42 | |||||||||||||||
| Basic earnings per Preference share and Preference ADS | 3.56 | 3.28 | 3.02 | 2.78 | 2.45 | |||||||||||||||
| Fully diluted earnings per Preference share and Preference ADS | 3.54 | 3.27 | 3.02 | 2.78 | 2.44 | |||||||||||||||
Dividends declared and paid per Ordinary share (€)(a) | 0.65 | 0.61 | 0.58 | 0.54 | 0.47 | |||||||||||||||
Dividends declared and paid per Preference share (€)(a) | 0.67 | 0.63 | 0.60 | 0.56 | 0.49 | |||||||||||||||
Dividends declared and paid per Ordinary share ($)(a) | 0.93 | 0.77 | 0.78 | 0.85 | 0.64 | |||||||||||||||
Dividends declared and paid per Preference share ($)(a) | 0.96 | 0.79 | 0.81 | 0.88 | 0.67 | |||||||||||||||
Balance Sheet Data at December 31: | ||||||||||||||||||||
| Working capital | $ | 1,432 | $ | 1,363 | $ | 2,118 | $ | 1,068 | $ | 833 | ||||||||||
| Total assets | 19,533 | 17,095 | 15,821 | 14,920 | 14,170 | |||||||||||||||
| Total long-term debt (excluding current portion) | 5,495 | 4,310 | 5,084 | 4,598 | 4,668 | |||||||||||||||
| Shareholders’ equity | 8,061 | 7,524 | 6,798 | 5,961 | 5,567 | |||||||||||||||
| Capital Stock — Preference shares — Nominal Value | 4 | 4 | 4 | 4 | 4 | |||||||||||||||
| Capital Stock — Ordinary shares — Nominal Value | 372 | 369 | 366 | 363 | 361 | |||||||||||||||
| (a) | Amounts shown for each year from 2011 to 2007 represent dividends declared and paid in each such year with respect to our operations in the year preceding payment. Our general partner’s Management Board has proposed dividends with respect to our operations in 2011 of €0.69 per Ordinary share and €0.71 per Preference share. These dividends are subject to approval by our shareholders at our Annual General Meeting to be held on May 10, 2012. |
3
Table of Contents
| D. | Risk Factors |
4
Table of Contents
| • | the quality, safety and efficacy of medical and pharmaceutical products and supplies; | |
| • | the operation of manufacturing facilities, laboratories and dialysis clinics; | |
| • | product advertising and other promotion; | |
| • | accurate reporting and billing for government and third-party reimbursement; and | |
| • | compensation of medical directors and other financial arrangements with physicians and other referral sources. |
5
Table of Contents
| • | The Medicare Improvements for Patients and Providers Act of 2008, or “MIPPA;” | |
| • | The Patient Protection and Affordable Care Act, enacted in March 2010 and subsequently amended by the Health Care and Educational Affordability Reconciliation Act (as amended, “ACA”); | |
| • | the U.S. Budget Control Act of 2011 (“Budget Control Act”) enacted in August 2011; and | |
| • | the Temporary Payroll Tax Cut Continuation Act of 2011 enacted in December 2011. |
6
Table of Contents
| • | a portion of our business that is currently reimbursed by private insurers or hospitals may become reimbursed by managed care organizations, which generally have lower rates for our services; or | |
| • | a portion of our business that is currently reimbursed by private insurers at rates based on our billed charges may become reimbursed under contracts at lower rates. |
7
Table of Contents
| • | the economic situation in developing or other countries could deteriorate; | |
| • | fluctuations in exchange rates could adversely affect profitability; | |
| • | we could face difficulties in enforcing and collecting accounts receivable under some countries’ legal systems; | |
| • | local regulations could restrict our ability to obtain a direct ownership interest in dialysis clinics or other operations; | |
| • | political, social or economic instability, especially in developing and newly industrializing countries, could disrupt our operations; | |
| • | some customers and governments could increase their payment cycles, with resulting adverse effects on our cash flow; | |
| • | some countries could impose additional or higher taxes or restrict the import of our products; | |
| • | we could fail to receive or could lose required licenses, certifications or other regulatory approvals for the operation of subsidiaries or dialysis clinics, sale of equipment, products, services or acquisitions; |
8
Table of Contents
| • | civil unrest, turmoil, or outbreak of disease in one of more countries in which we have material operations or material product revenue; | |
| • | differing labor regulations and difficulty in staffing and managing geographically widespread operations; | |
| • | different or less robust regulatory regimes controlling the protection of our intellectual property; and | |
| • | transportation delays or interruptions. |
9
Table of Contents
10
Table of Contents
11
Table of Contents
| Item 4. | Information on the Company |
| A. | History and Development of the Company |
| • | all of the outstanding common stock of W.R. Grace & Co., whose sole business at the time of the transaction consisted of National Medical Care, Inc., its global dialysis business, in exchange for 94,080,000 Ordinary shares; and | |
| • | the publicly-held minority interest in Fresenius USA, Inc., in exchange for 10,290,000 Ordinary shares. |
12
Table of Contents
| Actual | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| (in millions) | ||||||||||||
| Capital expenditures for property, plant and equipment | ||||||||||||
| North America | $ | 238 | $ | 212 | $ | 212 | ||||||
| International | 201 | 188 | 202 | |||||||||
| Corporate | 159 | 123 | 160 | |||||||||
| Total Capital Expenditures | $ | 598 | $ | 523 | $ | 574 | ||||||
| Acquisitions and Investments | ||||||||||||
| North America | $ | 824 | $ | 359 | $ | 124 | ||||||
| International | 1,186 | 405 | 66 | |||||||||
| Corporate | 6 | 158 | 2 | |||||||||
| Total Acquisitions and Investments | $ | 2,016 | $ | 922 | $ | 192 | ||||||
13
Table of Contents
| B. | Business Overview |
| 2011 | 2010 | 2009 | ||||||||||
| (in millions) | ||||||||||||
| North America | ||||||||||||
| Dialysis Care | $ | 7,337 | $ | 7,303 | $ | 6,794 | ||||||
| Dialysis Products | 813 | 827 | 818 | |||||||||
| 8,150 | 8,130 | 7,612 | ||||||||||
| International | ||||||||||||
| Dialysis Care | 2,170 | 1,767 | 1,556 | |||||||||
| Dialysis Products | 2,458 | 2,156 | 2,079 | |||||||||
| 4,628 | 3,923 | 3,635 | ||||||||||
14
Table of Contents
| • | increased general life expectancy and the overall aging of the general population; | |
| • | shortage of donor organs for kidney transplants; | |
| • | improved dialysis technology that makes life-prolonging dialysis available to a larger patient population; |
15
Table of Contents
| • | greater access to treatment in developing countries; and | |
| • | better treatment and survival of patients with hypertension, diabetes and other illnesses that lead to ESRD. |
16
Table of Contents
| Actual 2011 | Annual objectives for the years 2011-2013 | |||
Annual revenue growth1) | 5% | 6-8% | ||
| Annual average interest rate | 5.3% | 6.0-6.5% | ||
| Operating income margin | 16.2% (16.0% in 2010) | Incremental increases of 10-20 basis points per annum | ||
| Effective tax rate | 34% | 35-36% | ||
| Net income attributable to shareholders of FMC AG & Co. KGaA (annual growth in %) | 9% | High single to low double digits | ||
| Earnings per share (annual growth in %) | 9% | High single to low double digits | ||
Cash flow from operations2) | 11% | > 10% | ||
Capital expenditures and acquisitions2) | 18% | > 7% |
| 1) | in constant currency (See “Item 5 — Operating and Financial Review and Prospects — B. Liquidity and Capital Resources —Non-U.S. GAAP measures — Constant Currency) |
| 2) | As a percent of revenue. |
17
Table of Contents
18
Table of Contents
| • | our reputation for quality patient care and treatment; | |
| • | our extensive network of dialysis clinics, which enables physicians to refer their patients to conveniently located clinics; and | |
| • | our reputation for technologically advanced products for dialysis treatment. |
19
Table of Contents
20
Table of Contents
21
Table of Contents
| Year Ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| Medicare ESRD program | 46.2 | % | 49.4 | % | 50.0 | % | ||||||
| Private / alternative payors | 42.8 | % | 42.3 | % | 41.1 | % | ||||||
| Medicaid and other government sources | 5.9 | % | 3.4 | % | 3.6 | % | ||||||
| Hospitals | 5.1 | % | 4.9 | % | 5.3 | % | ||||||
| Total | 100.0 | % | 100.0 | % | 100.0 | % | ||||||
22
Table of Contents
23
Table of Contents
| • | HD machines and PD cyclers | |
| • | Dialyzers, our largest product group | |
| • | PD solutions in flexible bags | |
| • | HD concentrates, solutions and granulates | |
| • | Bloodlines | |
| • | Systems for water treatment |
| Year Ended December 31, | ||||||||||||||||||||||||
| 2011 | 2010 | 2009 | ||||||||||||||||||||||
| Total | Total | Total | ||||||||||||||||||||||
| Product | % of | Product | % of | Product | % of | |||||||||||||||||||
| Revenues | Total | Revenues | Total | Revenues | Total | |||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||
| Hemodialysis Products | $ | 2,603 | 79 | $ | 2,348 | 79 | $ | 2,263 | 78 | |||||||||||||||
| Peritoneal Dialysis Products | 417 | 13 | 329 | 11 | 320 | 11 | ||||||||||||||||||
| Other | 268 | 8 | 306 | 10 | 314 | 11 | ||||||||||||||||||
| Total | $ | 3,288 | 100 | $ | 2,983 | 100 | $ | 2,897 | 100 | |||||||||||||||
24
Table of Contents
| • | Volumetric dialysate balancing and ultrafiltration control system. This system, which we introduced in 1977, provides for safe and more efficient use of highly permeable dialyzers, permitting efficient dialysis with controlled rates of fluid removal; | |
| • | Proven hydraulic systems, providing reliable operation and servicing flexibility; | |
| • | Compatibility with all manufacturers’ dialyzers and a variety of bloodlines and dialysis solutions, permitting maximum flexibility in both treatment and disposable products usage; | |
| • | Modular design, which permits us to offer dialysis clinics a broad range of options to meet specific patient or regional treatment requirements and specialized modules that provide monitoring and response capability for selected biophysical patient parameters, such as body temperature and relative blood volume. Modular design also allows upgrading through module substitution without replacing the entire machine; | |
| • | Sophisticated microprocessor controls, touchscreen interfaces, displaysand/or readout panels that are adaptable to local language requirements; | |
| • | Battery backup, which continues operation of the blood circuit and all protective systems up to 20 minutes following a power failure; | |
| • | Online clearance, measurement of dialyzer clearance for quality assurance with On-Line Clearance Monitoring, providing immediate effective clearance information, real time treatment outcome monitoring, and therapy adjustment during dialysis without requiring invasive procedures or blood samples; | |
| • | The series 2008k@home, a dialysis machine specifically developed for in home use with an intuitively designed user interface and the addition of the wetness detector for increased safety. The use of our most advanced technology and adaptability for in-home use makes this machine highly accessible for patients who would like more control throughout their dialysis process. | |
| • | In the series 5008, the most efficient therapy mode Online-Hemodiafilitration as standard; | |
| • | Online data collection capabilities and computer interfacing with our TDMSand/or FDS08 systems. Our systems enable us to: |
| — | monitor and assess prescribed therapy; | |
| — | connect a large number of hemodialysis machines and peripheral devices, such as patient scales, blood chemistry analyzers and blood pressure monitors, to a computer network; | |
| — | enter nursing records automatically at bedside; | |
| — | adapt to new data processing devices and trends; | |
| — | perform home hemodialysis with remote monitoring by a staff caregiver; and | |
| — | record and analyze trends in medical outcome factors in hemodialysis patients. |
| • | increased biological compatibility, resulting in reduced incidence of adverse reactions to the fibers; | |
| • | greater capacity to clear uremic toxins from patient blood during dialysis, permitting more thorough, more rapid dialysis, resulting in shorter treatment time; and |
25
Table of Contents
| • | a complete range of permeability or membrane pore size, which permits dialysis at prescribed rates — high flux and low flux, as well as ultra flux for acute dialysis and allows tailoring of dialysis therapy to individual patients. |
| • | Fewer possibilities for touch contamination. Our unique PIN and DISC technology was designed to reduce the number of steps in the fluid exchange process and by doing so has lessened the risk of infection, particularly in the disconnection step in which the patient connector is closed automatically without the need for manual intervention. | |
| • | Optimal biocompatibility. Our PD balance and bicaVera® solutions are pH neutral and have very low glucose degradation products providing greater protection for the peritoneal membrane and allowing for the protection of the residual renal function of the PD patients. | |
| • | Environmentally friendly material: Our stay•safe® system is made of Biofine®, a material, developed by Fresenius, which upon combustion is reduced to carbon dioxide and water and does not contain any plasticizers. |
| • | Improved quality of life. The patient is treated at night and can lead a more normal life during the day without fluid exchange every few hours. | |
| • | Improved adequacy of dialysis. By adjusting the parameters of treatment it is possible to provide more dialysis to the patient compared to conventional CAPD therapy. This therapy offers important options to physicians such as improving the delivered dose of dialysis for certain patients. |
| • | sleep•safe: The sleep•safe machine has been used since 1999. It has automated connection technology thus further reducing the risk on touch contamination. Another key safety feature is the barcode recognition system for the types of solution bags used. This improves compliance and ensures that the prescribed dosage is administered to the patient. There is also a pediatric option for the treatment of infants. The sleep•safe machine allows for innovative and simple ways of individualizing APD prescriptions to achieve better treatment results. One of these is Adapted APD therapy in which, by using the same treatment volume and total treatment time but changing the profile of the cycles, better clearance and ultrafiltration are achieved. | |
| • | North American cycler portfolio: This includes: (a) the new Liberty® cycler introduced in 2008 incorporating many new operational and safety features with an innovative piston driven pumping cassette |
26
Table of Contents
| design, and user interface enhancements such as color touch screen which guides the patient through the setup and treatment (b) the Freedom® cyclers for Low Volume applications and acute markets, and (c) the Newton IQ® Cycler, which offers gentle gravity fills and drains as well as the option of pumping waste dialysate directly into the receptacle. The IQcardtm, in the form of a credit-card sized card or USB stick can provide actual treatment details and results for compliance monitoring to the physician and, when used with our North American PD cyclers, can upload the patient’s prescription into the machine. |
| • | Patient Management Software: We have developed specific patient management software tools to support both CAPD and APD therapies in the different regions of the world. These include: PatientOnLine, IQsystemtm, Pack-PD® and FITTessetm. These tools can be used by physicians and nurses to design and monitor treatment protocols thus ensuring that therapy is optimized and that patient care is maximized. |
27
Table of Contents
28
Table of Contents
29
Table of Contents
30
Table of Contents
31
Table of Contents
| • | failure to receive required licenses, certifications or other approvals for new facilities or products or significant delays in such receipt; | |
| • | complete or partial loss of various federal certifications, licenses, or other permits required under the laws of any state or other governmental authority by withdrawal, revocation, suspension, or termination or restrictions of such certificates and licenses by the imposition of additional requirements or conditions, or the initiation of proceedings possibly leading to such restrictions or the partial or complete loss of the required certificates, licenses or permits; | |
| • | a non-appealable finding of material violations of U.S. healthcare laws; and | |
| • | changes resulting from healthcare reform or other government actions that restrict our operations, reduce reimbursement or reduce or eliminate coverage for particular services we provide. |
32
Table of Contents
33
Table of Contents
34
Table of Contents
35
Table of Contents
36
Table of Contents
37
Table of Contents
38
Table of Contents
39
Table of Contents
| • | future changes in the ESA reimbursement methodologyand/or rate; | |
| • | a material reduction in the typical dosage per administration; | |
| • | increases in the cost of ESAs without offsetting increases in the ESRD PPS reimbursement rate; or | |
| • | reduction by the manufacturer of ESAs of the amount of overfill in the ESA vials. |
40
Table of Contents
41
Table of Contents
42
Table of Contents
43
Table of Contents
44
Table of Contents
45
Table of Contents
46
Table of Contents
47
Table of Contents
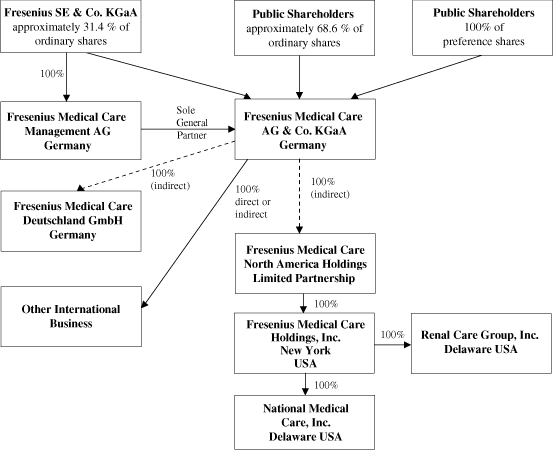
| C. | Organizational Structure |
| D. | Property, plant and equipment |
| Currently | ||||||||||
| Owned or | ||||||||||
| Floor Area | Leased by | |||||||||
| (Approximate | Fresenius | Lease | ||||||||
Location | Square Meters) | Medical Care | Expiration | Use | ||||||
| Bad Homburg, Germany | 18,700 | leased | December 2016 | Corporate headquarters and administration | ||||||
| Bad Homburg, Germany | 4,556 | leased | December 2012 | Administration building FMC GmbH Central Europe | ||||||
| St. Wendel, Germany | 73,136 | leased | December 2016 | Manufacture of polysulfone membranes, dialyzers and peritoneal dialysis solutions; research and development | ||||||
| Biebesheim, Germany | 33,500 | leased | December 2023 | Central distribution Europe, Asia Pacific and Latin America | ||||||
| Schweinfurt, Germany | 38,100 | leased | December 2016 | Manufacture of hemodialysis machines and peritoneal dialysis cyclers; research and development | ||||||
48
Table of Contents
| Currently | ||||||||||
| Owned or | ||||||||||
| Floor Area | Leased by | |||||||||
| (Approximate | Fresenius | Lease | ||||||||
Location | Square Meters) | Medical Care | Expiration | Use | ||||||
| Bad Homburg (OE), Germany | 10,304 | leased | December 2016 | Manufacture of hemodialysis concentrate solutions / technical services / logistics services | ||||||
| Stollberg, Germany | 3,600 | leased | July 2028 | Manufacture of sub-assemblies for hemodialysis machines | ||||||
| Palazzo Pignano, Italy | 19,990 | owned | Manufacture of bloodlines and tubing, office | |||||||
| L’Arbresle, France | 14,607 | owned | Manufacture of polysulfone dialyzers, special filters and dry hemodialysis concentrates | |||||||
| Nottinghamshire, UK | 5,110 | leased | June 2025 | Manufacture of hemodialysis concentrate solutions | ||||||
| Vrsac, Serbia | 3,331 | owned | Production area, laboratory, maintenance, administration, logistics | |||||||
| Barcelona, Spain | 2,000 | owned | Manufacture of hemodialysis concentrate solutions | |||||||
| Antalya, Turkey | 12,031 | leased | December 2037 | Manufacture of bloodlines | ||||||
| Casablanca, Morocco | 2,823 | owned | Manufacture of hemodialysis concentrate solutions | |||||||
| Guadalajara, México | 26,984 | owned | Manufacture of peritoneal dialysis bags | |||||||
| Buenos Aires, Argentina | 20,000 | owned | Manufacture of hemodialysis concentrate solutions, dry hemodialysis concentrates, bloodlines and disinfectants | |||||||
| São Paulo, Brazil | 8,615 | owned | Manufacture of hemodialysis concentrate solutions, dry hemodialysis concentrates, peritoneal dialysis bags, intravenous solutions bags, peritoneal dialysis and blood lines sets | |||||||
| São Paulo, Brazil | 5,430 | leased | March 2012 | Warehouse and technical service office | ||||||
| Bogotá, Colombia | 14,018 | owned | Manufacture of hemodialysis concentrate solutions, peritoneal dialysis bags, intravenous solutions, administration | |||||||
| Bogotá, Colombia | 1,600 | leased | July 2013 | Manufacture of peritoneal dialysis bags | ||||||
| Bogotá, Colombia | 2,619 | owned | Administration Building | |||||||
| Valencia, Venezuela | 3,648 | leased | June 2015 | Head office and warehouse | ||||||
| Hong Kong | 1,770 | leased | February 2014 | Warehouse | ||||||
| Suzhou, China (Changshu Plant) | 25,168 | owned | Manufacture of hemodialysis bloodline sets / AV fistula set | |||||||
| Smithfield NSW, Australia | 5,350 | owned | Manufacture of hemodialysis concentrate & warehouse | |||||||
| Scoresby, Australia | 6,263 | leased | December 2019 | VIC warehouse / seating & packs / production | ||||||
| Auckland, New Zealand | 2,170 | leased | May 2030 | Warehouse / office | ||||||
| Selangor, Malaysia | 3,149 | leased | May 2015 | Administration / warehouse | ||||||
| Yongin, South Korea | 1,650 | leased | June 2012 | Warehouse | ||||||
| Seaol, South Korea | 1,905 | leased | January 2013 | Administration | ||||||
| Sooncheon, South Korea | 5,112 | owned | Clinic | |||||||
49
Table of Contents
| Currently | ||||||||||
| Owned or | ||||||||||
| Floor Area | Leased by | |||||||||
| (Approximate | Fresenius | Lease | ||||||||
Location | Square Meters) | Medical Care | Expiration | Use | ||||||
| Taipei, Taiwan | 1,841 | leased | September 2015 | Sales, technical and administration office | ||||||
| Tai Chung, Taiwan | 3,053 | leased | January 2020 | Nephrocare clinic | ||||||
| Oita, Japan (Inukai Plant) | 3,065 | owned | Manufacture of polysulfone filters | |||||||
| Fukuoka, Japan (Buzen Plant) | 37,092 | owned | Manufacture of peritoneal dialysis bags and dialyzers | |||||||
| Fukuoka, Japan (Buzen Plant) - Site Area for future expansion | 27,943 | owned | Manufacture of peritoneal dialysis bags and dialyzers | |||||||
| Ibaragi, Japan | 7,111 | leased | August 2013 | Clinic | ||||||
| Waltham, Massachusetts | 25,588 | leased | April 2017 - July 2017 with a 10 year and a second 5 year renewal option | North American corporate headquarters | ||||||
| Lexington, Massachusetts | 6,425 | leased | April 2017 | IT headquarters and administration - North America | ||||||
| Nashville, Tennessee | 4,487 | leased | August 2013 | IT administration / payroll administration | ||||||
| Walnut Creek, California | 7,897 | leased | June 2013 | Manufacture of hemodialysis machines and peritoneal | ||||||
| Pittsburg, California | 7,135 | leased | June 2013 | Warehouse | ||||||
| Ogden, Utah | 74,322 | owned | Manufacture polysulfone membranes and dialyzers and peritoneal dialysis solutions; research and development | |||||||
| Ogden, Utah | 9,755 | leased | July 2033 | Plant expansion, manufacturing operations | ||||||
| Ogden, Utah | 24,452 | leased | December 2021 | Warehouse | ||||||
| Ogden, Utah | 8,933 | leased | December 2021 | Warehouse | ||||||
| Ogden, Utah | 2,072 | leased | year-to-year lease | Warehouse | ||||||
| Oregon, Ohio | 13,934 | leased | April 2019 | Manufacture of liquid hemodialysis concentrate solutions | ||||||
| Livingston, California | 7,885 | leased | December 2017 with two consequtive 5-year renewal options | Manufacture of liquid hemodialysis concentrates and resupply | ||||||
| Milpitas, California | 8,670 | leased | December 2015 with 5-year renewal option | Clinical laboratory testing | ||||||
| Rockleigh, New Jersey | 9,812 | leased | May 2012 | Clinical laboratory testing | ||||||
| Irving, Texas | 8,374 | leased | February 2014 | Manufacture of liquid hemodialysis solution | ||||||
| Reynosa, Mexico | 13,936 | leased | June 2013 | Manufacture of bloodlines | ||||||
| Reynosa, Mexico | 7,079 | leased | June 2013 | Warehouse | ||||||
| Reynosa, Mexico | 4,645 | owned | Warehouse | |||||||
| Lachine, Canada | 3,663 | leased | March 2014 | Warehouse | ||||||
| Montreal, Canada | 4,036 | leased | September 2020 | Warehouse | ||||||
| Richmond , Canada | 2,286 | leased | April 2014 | Warehouse | ||||||
| Richmond Hill, Canada | 5,948 | leased | November 2016 | Warehouse and administrative offices | ||||||
| Warrendale, Pennsylvania | 2,366 | leased | April 2013 | RSI administration and research facility | ||||||
| Oklahoma City, OK | 3,665 | leased | October 2015 | Manufacture of sorbent cartridges | ||||||
50
Table of Contents
| Item 4A. | Unresolved Staff Comments |
| Item 5. | Operating and Financial Review and Prospects |
51
Table of Contents
52
Table of Contents
| At December 31, 2011 | ||||||||||||||||||||||||||||
| overdue | overdue | |||||||||||||||||||||||||||
| overdue | more than | more than | overdue | % of | ||||||||||||||||||||||||
| by | 3 months | 6 months | by | net | ||||||||||||||||||||||||
| up to | up to | up to | more than | trade | ||||||||||||||||||||||||
| current | 3 months | 6 months | 1 year | 1 year | Total | A/R | ||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||
| U.S. Medicare and Medicaid Programs | $ | 379 | $ | 92 | $ | 51 | $ | 44 | $ | 29 | $ | 595 | 21 | |||||||||||||||
| U.S. Commercial Payors | 250 | 142 | 37 | 33 | 21 | 483 | 17 | |||||||||||||||||||||
| U.S. Hospitals | 101 | 25 | 5 | 2 | 1 | 133 | 5 | |||||||||||||||||||||
| Self-Pay of U.S. patients | 0 | 4 | 4 | 1 | 1 | 11 | 0 | |||||||||||||||||||||
| Other North America | 8 | 3 | 1 | 0 | 0 | 12 | 1 | |||||||||||||||||||||
| International product customers and dialysis payors | 772 | 289 | 144 | 140 | 219 | 1,564 | 56 | |||||||||||||||||||||
| Total | $ | 1,510 | $ | 555 | $ | 242 | $ | 220 | $ | 271 | $ | 2,798 | 100 | |||||||||||||||
| At December 31, 2010 | ||||||||||||||||||||||||||||
| overdue | overdue | |||||||||||||||||||||||||||
| overdue | more than | more than | overdue | % of | ||||||||||||||||||||||||
| by | 3 months | 6 months | by | net | ||||||||||||||||||||||||
| up to | up to | up to | more than | trade | ||||||||||||||||||||||||
| current | 3 months | 6 months | 1 year | 1 year | Total | A/R | ||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||
| U.S. Medicare and Medicaid Programs | $ | 372 | $ | 85 | $ | 41 | $ | 28 | $ | 20 | $ | 546 | 21 | |||||||||||||||
| U.S. Commercial Payors | 270 | 152 | 48 | 39 | 22 | 531 | 21 | |||||||||||||||||||||
| U.S. Hospitals | 88 | 28 | 3 | 2 | 3 | 124 | 5 | |||||||||||||||||||||
| Self-Pay of U.S. patients | 0 | 3 | 3 | 1 | — | 7 | 0 | |||||||||||||||||||||
| Other North America | 1 | 1 | 0 | 0 | — | 2 | 0 | |||||||||||||||||||||
| International product customers and dialysis payors | 777 | 227 | 116 | 112 | 131 | 1,363 | 53 | |||||||||||||||||||||
| Total | $ | 1,508 | $ | 496 | $ | 211 | $ | 182 | $ | 176 | $ | 2,573 | 100 | |||||||||||||||
53
Table of Contents
54
Table of Contents
55
Table of Contents
56
Table of Contents
| A. | Results of Operations |
| 2011 | 2010 | 2009 | ||||||||||||||
| (in millions) | ||||||||||||||||
| Total revenue | ||||||||||||||||
| North America | $ | 8,159 | $ | 8,135 | $ | 7,615 | ||||||||||
| International | 4,628 | 3,923 | 3,635 | |||||||||||||
| Corporate | 17 | — | — | |||||||||||||
| Totals | 12,804 | 12,058 | 11,250 | |||||||||||||
| Inter-segment revenue | ||||||||||||||||
| North America | 9 | 5 | 3 | |||||||||||||
| International | — | — | — | |||||||||||||
| Totals | 9 | 5 | 3 | |||||||||||||
| Total net revenue | ||||||||||||||||
| North America | 8,150 | 8,130 | 7,612 | |||||||||||||
| International | 4,628 | 3,923 | 3,635 | |||||||||||||
| Corporate | 17 | — | — | |||||||||||||
| Totals | 12,795 | 12,053 | 11,247 | |||||||||||||
| Amortization and depreciation | ||||||||||||||||
| North America | 269 | 254 | 233 | |||||||||||||
| International | 174 | 149 | 129 | |||||||||||||
| Corporate | 114 | 100 | 95 | |||||||||||||
| Totals | 557 | 503 | 457 | |||||||||||||
| Operating income | ||||||||||||||||
| North America | 1,435 | 1,386 | 1,250 | |||||||||||||
| International | 807 | 678 | 637 | |||||||||||||
| Corporate | (167 | ) | (140 | ) | (131 | ) | ||||||||||
| Totals | 2,075 | 1,924 | 1,756 | |||||||||||||
| Interest income | 60 | 25 | 21 | |||||||||||||
| Interest expense | (357 | ) | (305 | ) | (321 | ) | ||||||||||
| Income tax expense | (601 | ) | (578 | ) | (491 | ) | ||||||||||
| Net Income | 1,177 | 1,066 | 965 | |||||||||||||
| Less: Net Income attributable to noncontrolling interests | (106 | ) | (87 | ) | (74 | ) | ||||||||||
| Net Income attributable to shareholders of FMC-AG & Co. KGaA | $ | 1,071 | $ | 979 | $ | 891 | ||||||||||
57
Table of Contents
| Change in % | ||||||||||||||||
| at constant | ||||||||||||||||
| 2011 | 2010 | as reported | exchange rates(1) | |||||||||||||
| Number of treatments | 34,388,422 | 31,670,702 | 9 | % | ||||||||||||
| Same market treatment growth in % | 3.9 | % | 4.6 | % | ||||||||||||
| Revenue in $ million | 12,795 | 12,053 | 6 | % | 5 | % | ||||||||||
| Gross profit in % of revenue | 35.3 | % | 34.4 | % | ||||||||||||
| Selling, general and administrative costs in % of revenue | 18.5 | % | 17.7 | % | ||||||||||||
Net income attributable to shareholders ofFMC-AG & Co. KGaA in $ million | 1,071 | 979 | 9 | % | ||||||||||||
| (1) | For further information on “at constant exchange rates,” see“Non-U.S. GAAP Measures — Constant currency” below. |
58
Table of Contents
| 2011 | 2010 | Change in % | ||||||||||
| Number of treatments | 21,608,620 | 20,850,242 | 4 | % | ||||||||
| Same market treatment growth in % | 3.2 | % | 4.3 | % | ||||||||
| Revenue in $ million | 8,150 | 8,130 | 0 | % | ||||||||
| Depreciation and amortization in $ million | 269 | 254 | 6 | % | ||||||||
| Operating income in $ million | 1,435 | 1,386 | 4 | % | ||||||||
| Operating income margin in % | 17.6 | % | 17.0 | % | ||||||||
59
Table of Contents
| Change in % | ||||||||||||||||
| at constant | ||||||||||||||||
| 2011 | 2010 | as reported | exchange rates(1) | |||||||||||||
| Number of treatments | 12,779,802 | 10,820,460 | 18 | % | ||||||||||||
| Same market treatment growth in % | 5.4 | % | 5.1 | % | ||||||||||||
| Revenue in $ million | 4,628 | 3,923 | 18 | % | 14 | % | ||||||||||
| Depreciation and amortization in $ million | 174 | 149 | 17 | % | ||||||||||||
| Operating income in $ million | 807 | 678 | 19 | % | ||||||||||||
| Operating income margin in % | 17.4 | % | 17.3 | % | ||||||||||||
| (1) | For further information on “at constant exchange rates,” see“Non-U.S. GAAP Measures — Constant currency” below. |
60
Table of Contents
| Change in % | ||||||||||||||||
| at constant | ||||||||||||||||
| 2010 | 2009 | as reported | exchange rates(1) | |||||||||||||
| Number of treatments | 31,670,702 | 29,425,758 | 8 | % | ||||||||||||
| Same market treatment growth in % | 4.6 | % | 4.1 | % | ||||||||||||
| Revenue in $ million | 12,053 | 11,247 | 7 | % | 7 | % | ||||||||||
| Gross profit in % of revenue | 34.4 | % | 34.1 | % | ||||||||||||
| Selling, general and administrative costs in % of revenue | 17.7 | % | 17.6 | % | ||||||||||||
Net income attributable to shareholders ofFMC-AG & Co. KGaA in $ million | 979 | 891 | 10 | % | ||||||||||||
| (1) | For further information on “at constant exchange rates,” see“Non-U.S. GAAP Measures — Constant currency” below. |
61
Table of Contents
| 2010 | 2009 | Change in % | ||||||||||
| Number of treatments | 20,850,242 | 19,867,465 | 5 | % | ||||||||
| Same market treatment growth in % | 4.3 | % | 3.5 | % | ||||||||
| Revenue in $ million | 8,130 | 7,612 | 7 | % | ||||||||
| Depreciation and amortization in $ million | 254 | 233 | 9 | % | ||||||||
| Operating income in $ million | 1,386 | 1,250 | 11 | % | ||||||||
| Operating income margin in % | 17.0 | % | 16.4 | % | ||||||||
62
Table of Contents
| Change in % | ||||||||||||||||
| at constant | ||||||||||||||||
| 2010 | 2009 | as reported | exchange rates(1) | |||||||||||||
| Number of treatments | 10,820,460 | 9,558,293 | 13 | % | ||||||||||||
| Same market treatment growth in % | 5.1 | % | 5.3 | % | ||||||||||||
| Revenue in $ million | 3,923 | 3,635 | 8 | % | 8 | % | ||||||||||
| Depreciation and amortization in $ million | 149 | 129 | 16 | % | ||||||||||||
| Operating income in $ million | 678 | 637 | 6 | % | ||||||||||||
| Operating income margin in % | 17.3 | % | 17.5 | % | ||||||||||||
| (1) | For further information on “at constant exchange rates,” see“Non-U.S. GAAP Measures — Constant currency” below. |
63
Table of Contents
| B. | Liquidity and Capital Resources |
64
Table of Contents
| December 31, | December 31, | |||||||
| 2011 | 2010 | |||||||
| North America days sales outstanding | 55 | 54 | ||||||
| International days sales outstanding | 121 | 116 | ||||||
| FMC-AG & Co. KGaA average days sales outstanding | 80 | 76 | ||||||
65
Table of Contents
66
Table of Contents
67
Table of Contents
| Available Sources of Liquidity | Expiration per period of | |||||||||||
in millions | Total | 1 Year | 2-5 Years | |||||||||
Accounts receivable facility(a) | $ | 266 | $ | — | $ | 266 | ||||||
Revolving Credit Facility of the Amended 2006 Senior Credit Agreement(b) | 960 | — | 960 | |||||||||
| Other Unused Lines of Credit | 234 | 234 | — | |||||||||
| $ | 1,460 | $ | 234 | $ | 1,226 | |||||||
| (a) | Subject to availability of sufficient accounts receivable meeting funding criteria. |
| (b) | At December 31, 2011, the Company had letter of credit outstanding in the amount of $181 which reduces the availability under the Revolving Credit Facility to the amount shown in this table. |
| Contractual Obligations and Commitments | Payments due by period of | |||||||||||||||
in millions | Total | 1 Year | 2-5 Years | Over 5 Years | ||||||||||||
Long Term Debt(a)(b) | $ | 7,854 | $ | 1,716 | $ | 3,243 | $ | 2,895 | ||||||||
| Capital Lease Obligations | 19 | 5 | 7 | 7 | ||||||||||||
| Operating Leases | 2,707 | 511 | 1,457 | 739 | ||||||||||||
| Unconditional Purchase Obligations | 2,598 | 533 | 1,338 | 727 | ||||||||||||
| Other Long-term Obligations | 116 | 99 | 17 | — | ||||||||||||
| Letters of Credit | 181 | — | 181 | — | ||||||||||||
| $ | 13,474 | $ | 2,864 | $ | 6,243 | $ | 4,368 | |||||||||
| (a) | Includes expected interest payments which are based upon the principal repayment schedules and fixed interest rates or estimated variable interest rates considering the applicable interest rates (e.g. Libor, Prime), the applicable margins, and the effects of related interest rate swaps. |
| (b) | Excludes our 55/8% Senior Notes and 5.25% Euro-denominated Senior Notes due 2019 and our 57/8% Senior Notes due 2022 issued on January 26, 2012. |
68
Table of Contents
| 2012 | 2013 | |||
Revenue1) | ~$14 billion | |||
Revenue growth2) | ~14% | 6-8% | ||
| Operating Income Margin | ~16.9% | |||
| Net Income | ~$1.3 billion | |||
| Net Income attributable to shareholders of FMC-AG & Co. KGaA | ~$1.14 billion | ³ revenue growth | ||
| Dividends | based on development of earnings | based on development of earnings | ||
| Capital Expenditures | ~$0.7 billion | |||
| Acquisitions | ~$1.8 billion | |||
| Capital Expenditures and Acquisitions in % of revenue | ~7-9% | |||
| Debt/EBITDA Ratio | < 3.0 | < 2.7 |
| 1) | Revenue outlook has been made in accordance with U.S. GAAP upon initial application of Accounting Standards Update2011-07, Health Care Entities (Topic 954): Presentation and Disclosure of Patient Service Revenues, Provision for Bad Debts and the Allowance for Doubtful Accounts for Certain Health Care Entities (see Note 1s), “The Company and Basis ofPresentation-Recent Pronouncements.” The comparable revenue for 2011 was $12.571 billion. |
| 2) | At constant currency (See“Non-U.S. GAAP Constant Currency” below,) |
69
Table of Contents
| For the years ended December 31, | ||||||||||||
| 2011 | 2010 | 2009 | ||||||||||
| (in millions) | ||||||||||||
| Total EBITDA | $ | 2,632 | $ | 2,427 | $ | 2,213 | ||||||
| Interest expense (net of interest income) | (297 | ) | (280 | ) | (300 | ) | ||||||
| Income tax expense, net | (601 | ) | (578 | ) | (490 | ) | ||||||
| Change in deferred taxes, net | 147 | 15 | 22 | |||||||||
| Changes in operating assets and liabilities | (397 | ) | (237 | ) | (140 | ) | ||||||
| Stock Compensation expense | 29 | 28 | 34 | |||||||||
| Other items, net | (67 | ) | (7 | ) | — | |||||||
| Net cash provided by operating activities | $ | 1,446 | $ | 1,368 | $ | 1,339 | ||||||
70
Table of Contents
| C. | Research and Development |
71
Table of Contents
| D. | Trend information |
| F. | Tabular Disclosure of contractual obligations |
| Item 6. | Directors, Senior Management and Employees |
| A. | Directors and senior management |
72
Table of Contents
| Age as of | ||||
| December 31, | ||||
Name | 2011 | |||
Dr. Ulf M. Schneider, Chairman(1) | 46 | |||
Dr. Dieter Schenk, Vice Chairman(4) | 59 | |||
Dr. Gerd Krick(1)(2) | 73 | |||
Mr. Rolf A. Classon(3)(4) | 66 | |||
Dr. Walter L. Weisman(1)(2)(3) | 76 | |||
Mr. William P. Johnston(1)(2)(3)(4) | 67 | |||
| (1) | Members of the Human Resources Committee of the Supervisory Board of Management AG |
| (2) | Members of the Audit and Corporate Governance Committee of FMC-AG & Co. KGaA |
| (3) | Independent director for purposes of our pooling agreement |
| (4) | Member of the Regulatory and Reimbursement Assessment Committee of the Supervisory Board of Management AG |
73
Table of Contents
| Age as of | ||||||||||
| December 31, | Year term | |||||||||
Name | 2011 | Position | expires | |||||||
| Dr. Ben J. Lipps | 71 | Chief Executive Officer and Chairman of the Management Board | 2012 | |||||||
| Rice Powell | 56 | Vice Chairman of the Management Board and Chief Executive Officer, Fresenius Medical Care North America | 2014 | |||||||
| Michael Brosnan | 56 | Chief Financial Offier | 2012 | |||||||
| Roberto Fusté | 59 | Chief Executive Officer for Asia Pacific | 2016 | |||||||
| Dr. Emanuele Gatti | 56 | Chief Executive Officer for Europe, Middle East, Africa and Latin America and Chief Strategist for FMC-AG & Co. KGaA | 2012 | |||||||
| Dr. Rainer Runte | 52 | Chief Administrative Officer for Global Law, Compliance, Intellectual Property and Corporate Business Development and Labor Relations Director for Germany | 2014 | |||||||
| Kent Wanzek | 52 | Head of Global Manufacturing Operations | 2012 | |||||||
74
Table of Contents
75
Table of Contents
| B. | Compensation |
76
Table of Contents
| • | performance-unrelated compensation (basic salary) | |
| • | performance-related compensation (variable bonus) | |
| • | components with long-term incentive effects (stock options, share-based compensation with cash settlement). |
77
Table of Contents
| Amount of Cash Payments | ||||||||||||||||||||||||||||||||
| Non-Performance Related | Cash Compensation | |||||||||||||||||||||||||||||||
| Compensation | Performance Related Compensation | (without long-term | ||||||||||||||||||||||||||||||
| Salary | Other1) | Bonus | Incentive Components) | |||||||||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||||||||
| in thousands | in thousands | in thousands | in thousands | |||||||||||||||||||||||||||||
| Dr. Ben Lipps | $ | 1,200 | $ | 1,200 | $ | 254 | $ | 469 | $ | 1,500 | $ | 1,554 | $ | 2,954 | $ | 3,223 | ||||||||||||||||
| Michael Brosnan | 650 | 650 | 255 | 183 | 813 | 821 | 1,718 | 1,654 | ||||||||||||||||||||||||
| Roberto Fusté | 696 | 597 | 262 | 245 | 768 | 740 | 1,726 | 1,582 | ||||||||||||||||||||||||
| Dr. Emanuele Gatti | 940 | 862 | 169 | 139 | 1,022 | 1,086 | 2,131 | 2,087 | ||||||||||||||||||||||||
| Rice Powell | 950 | 950 | 37 | 36 | 1,361 | 1,319 | 2,348 | 2,305 | ||||||||||||||||||||||||
| Dr. Rainer Runte | 592 | 564 | 59 | 47 | 740 | 729 | 1,391 | 1,340 | ||||||||||||||||||||||||
| Kent Wanzek | 500 | 500 | 24 | 25 | 716 | 727 | 1,240 | 1,252 | ||||||||||||||||||||||||
| Total | $ | 5,528 | $ | 5,323 | $ | 1,060 | $ | 1,144 | $ | 6,920 | $ | 6,976 | $ | 13,508 | $ | 13,443 | ||||||||||||||||
| 1) | Includes insurance premiums, private use of company cars, rent supplements, contributions to pension and health insurance and other benefits. |
78
Table of Contents
| Components with Long-term Incentive Effect | ||||||||||||||||||||||||||||||||
| Share-based Compensation with Cash | ||||||||||||||||||||||||||||||||
| Stock Options | Settlement1) | Total | ||||||||||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||||||||
| Number | in thousands | in thousands | in thousands | |||||||||||||||||||||||||||||
| Dr. Ben Lipps | 74,700 | 99,600 | $ | 1,444 | $ | 1,040 | $ | 967 | $ | 518 | $ | 2,411 | $ | 1,558 | ||||||||||||||||||
| Michael Brosnan | 37,350 | 49,800 | 722 | 520 | 504 | 301 | 1,226 | 821 | ||||||||||||||||||||||||
| Roberto Fusté | 37,350 | 49,800 | 722 | 520 | 489 | 207 | 1,211 | 727 | ||||||||||||||||||||||||
| Dr. Emanuele Gatti | 29,880 | 49,800 | 578 | 520 | 715 | 553 | 1,293 | 1,073 | ||||||||||||||||||||||||
| Rice Powell | 56,025 | 74,700 | 1,083 | 780 | 804 | 539 | 1,887 | 1,319 | �� | |||||||||||||||||||||||
| Dr. Rainer Runte | 34,860 | 49,800 | 674 | 520 | 527 | 243 | 1,201 | 763 | ||||||||||||||||||||||||
| Kent Wanzek | 37,350 | 49,800 | 722 | 520 | 472 | 242 | 1,194 | 762 | ||||||||||||||||||||||||
| Total | 307,515 | 423,300 | $ | 5,945 | $ | 4,420 | $ | 4,478 | $ | 2,603 | $ | 10,423 | $ | 7,023 | ||||||||||||||||||
| 1) | This includes Phantom Stocks granted to Board Members during the fiscal year. The share-based compensation amounts are based on the grant date fair value. |
79
Table of Contents
| Development and status of the stock options | ||||||||||||||||||||||||||||||||
| Dr. Ben | Michael | Roberto | Dr. Emanuele | Rice | Dr. Rainer | Kent | ||||||||||||||||||||||||||
| Lipps | Brosnan | Fusté | Gatti | Powell | Runte | Wanzek | Total | |||||||||||||||||||||||||
| Options outstanding at January 1, 2011 | ||||||||||||||||||||||||||||||||
| Number | 598,870 | 269,598 | 339,986 | 375,876 | 224,100 | 284,469 | 85,800 | 2,178,699 | ||||||||||||||||||||||||
| Weighted average exercise price in $ | 41.60 | 40.03 | 38.65 | 36.67 | 47.55 | 42.49 | 50.36 | 41.17 | ||||||||||||||||||||||||
| Options granted during the fiscal year | ||||||||||||||||||||||||||||||||
| Number | 74,700 | 37,350 | 37,350 | 29,880 | 56,025 | 34,860 | 37,350 | 307,515 | ||||||||||||||||||||||||
| Weighted average exercise price in $ | 75.47 | 75.47 | 75.47 | 75.47 | 75.47 | 75.47 | 75.47 | 75.47 | ||||||||||||||||||||||||
| Options exercised during the fiscal year | ||||||||||||||||||||||||||||||||
| Number | 100,870 | — | — | 30,469 | — | — | — | 131,339 | ||||||||||||||||||||||||
| Weighted average exercise price in $ | 23.99 | — | — | 18.59 | — | — | — | 22.74 | ||||||||||||||||||||||||
| Weighted average share price in $ | 63.68 | — | — | 64.69 | — | — | — | 63.92 | ||||||||||||||||||||||||
| Options outstanding at December 31, 2011 | ||||||||||||||||||||||||||||||||
| Number | 572,700 | 306,948 | 377,336 | 375,287 | 280,125 | 319,329 | 123,150 | 2,354,875 | ||||||||||||||||||||||||
| Weighted average exercise price in $ | 48.13 | 43.43 | 41.54 | 40.62 | 51.62 | 45.27 | 55.68 | 45.69 | ||||||||||||||||||||||||
| Weighted average remaining contractual life in years | 4.1 | 4.0 | 3.5 | 3.4 | 4.9 | 3.9 | 5.7 | 4.0 | ||||||||||||||||||||||||
| Range of exercise price in $ | 39.45 - 75.47 | 14.78 - 75.47 | 14.78 - 75.47 | 14.78 -75.47 | 41.37 - 75.47 | 18.72 - 75.47 | 41.37 - 75.47 | 14.78 - 75.47 | ||||||||||||||||||||||||
| Options exercisable at December 31, 2011 | ||||||||||||||||||||||||||||||||
| Number | 298,800 | 186,798 | 240,386 | 245,807 | 99,600 | 184,869 | 18,000 | 1,274,260 | ||||||||||||||||||||||||
| Weighted average exercise price in $ | 43.08 | 35.75 | 34.65 | 34.20 | 44.90 | 39.37 | 45.92 | 38.35 | ||||||||||||||||||||||||
| Total Compensation | ||||||||||||||||||||||||
| Components with | ||||||||||||||||||||||||
| long-term | ||||||||||||||||||||||||
| Cash Compensation (without long-term Incentive components) | Incentive Effect | Total Compensation (including long-term Incentive Components) | ||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||
| in thousands | in thousands | in thousands | ||||||||||||||||||||||
| Dr. Ben Lipps | $ | 2,954 | $ | 3,223 | $ | 2,411 | $ | 1,558 | $ | 5,365 | $ | 4,781 | ||||||||||||
| Michael Brosnan | 1,718 | 1,654 | 1,226 | 821 | 2,944 | 2,475 | ||||||||||||||||||
| Roberto Fusté | 1,726 | 1,582 | 1,211 | 727 | 2,937 | 2,309 | ||||||||||||||||||
| Dr. Emanuele Gatti | 2,131 | 2,087 | 1,293 | 1,073 | 3,424 | 3,160 | ||||||||||||||||||
| Rice Powell | 2,348 | 2,305 | 1,887 | 1,319 | 4,235 | 3,624 | ||||||||||||||||||
| Dr. Rainer Runte | 1,391 | 1,340 | 1,201 | 763 | 2,592 | 2,103 | ||||||||||||||||||
| Kent Wanzek | 1,240 | 1,252 | 1,194 | 762 | 2,434 | 2,014 | ||||||||||||||||||
| Total | $ | 13,508 | $ | 13,443 | $ | 10,423 | $ | 7,023 | $ | 23,931 | $ | 20,466 | ||||||||||||
80
Table of Contents
| Expenses for Long-term Incentive Components | ||||||||||||||||||||||||
| Stock Options | Share-based Compensation with Cash Settlement | Share-based Compensation | ||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||
| in thousands | in thousands | in thousands | ||||||||||||||||||||||
| Dr. Ben Lipps | $ | 1,529 | $ | 1,165 | $ | 1,085 | $ | 1,140 | $ | 2,614 | $ | 2,305 | ||||||||||||
| Michael Brosnan | 259 | 74 | 133 | — | 392 | 74 | ||||||||||||||||||
| Roberto Fusté | 568 | 583 | 175 | 61 | 743 | 644 | ||||||||||||||||||
| Dr. Emanuele Gatti | 553 | 583 | 564 | 426 | 1,117 | 1,009 | ||||||||||||||||||
| Rice Powell | 698 | 620 | 611 | 713 | 1,309 | 1,333 | ||||||||||||||||||
| Dr. Rainer Runte | 563 | 583 | 416 | 502 | 979 | 1,085 | ||||||||||||||||||
| Kent Wanzek | 259 | 74 | 111 | — | 370 | 74 | ||||||||||||||||||
| Total | $ | 4,429 | $ | 3,682 | $ | 3,095 | $ | 2,842 | $ | 7,524 | $ | 6,524 | ||||||||||||
81
Table of Contents
| Development and status of pension commitments | ||||||||||||
| As of January 1, | As of December 31, | |||||||||||
| 2011 | increase | 2011 | ||||||||||
| in thousands | ||||||||||||
| Dr. Ben Lipps | $ | 536 | $ | 303 | $ | 839 | ||||||
| Michael Brosnan | 68 | 21 | 89 | |||||||||
| Roberto Fusté | 2,398 | 361 | 2,759 | |||||||||
| Dr. Emanuele Gatti | 4,619 | 259 | 4,878 | |||||||||
| Rice Powell | 131 | 39 | 170 | |||||||||
| Dr. Rainer Runte | 1,081 | 50 | 1,131 | |||||||||
| Total | $ | 8,833 | $ | 1,033 | $ | 9,866 | ||||||
82
Table of Contents
83
Table of Contents
| Fixed | Fixed | |||||||||||||||||||||||||||||||||||||||
| compensation for | compensation for | Compensation for | ||||||||||||||||||||||||||||||||||||||
| Supervisory Board | Supervisory Board | committee services | ||||||||||||||||||||||||||||||||||||||
| at FMC | at FMC-AG | Compensation for committee services | at FMC-AG | Non-Performance Related | ||||||||||||||||||||||||||||||||||||
| Management AG | & Co. KGaA | at FMC Management AG | & Co. KGaA | Compensation | ||||||||||||||||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||||||||||||||
| in thousand | in thousand | in thousand | in thousand | in thousand | ||||||||||||||||||||||||||||||||||||
| Dr. Gerd Krick | $ | 40 | $ | 40 | $ | 120 | $ | 120 | $ | 60 | $ | 60 | $ | 40 | $ | 30 | $ | 260 | $ | 250 | ||||||||||||||||||||
| Dr. Dieter Schenk | 60 | 60 | 60 | 60 | 50 | 50 | — | — | 170 | 170 | ||||||||||||||||||||||||||||||
Dr. Ulf M. Schneider2) | 160 | 160 | — | — | 70 | 70 | — | — | 230 | 230 | ||||||||||||||||||||||||||||||
| Dr. Walter L. Weisman | 40 | 40 | 40 | 40 | 50 | 50 | 60 | 50 | 190 | 180 | ||||||||||||||||||||||||||||||
John Gerhard Kringel3) | 20 | 40 | 15 | 40 | 30 | 60 | — | — | 65 | 140 | ||||||||||||||||||||||||||||||
| William P. Johnston | 40 | 40 | 40 | 40 | 120 | 120 | 40 | 30 | 240 | 230 | ||||||||||||||||||||||||||||||
Prof. Dr. Bernd Fahrholz4) | — | — | 80 | 80 | — | — | 45 | 30 | 125 | 110 | ||||||||||||||||||||||||||||||
Rolf A. Classon5) | 20 | — | 31 | — | 30 | — | — | — | 81 | — | ||||||||||||||||||||||||||||||
| Total | $ | 380 | $ | 380 | $ | 386 | $ | 380 | $ | 410 | $ | 410 | $ | 185 | $ | 140 | $ | 1,361 | $ | 1,310 | ||||||||||||||||||||
| 1) | Shown without VAT and withholding tax | |
| 2) | Chairman of the supervisory board of FMC Management AG, but not member of the supervisory board of FMC-AG & Co. KGaA; compensation paid by FMC Management AG | |
| 3) | Member of the supervisory board of FMC-AG & Co. KGaA until May 12, 2011, Member of the supervisory board and Member of committee of FMC Management AG until July 7, 2011 | |
| 4) | Member of the supervisory board of FMC-AG & Co. KGaA, but not member of the supervisory board of FMC Management AG; compensation paid by FMC-AG & Co. KGaA | |
| 5) | Member of the supervisory board of FMC-AG & Co. KGaA as of May 12, 2011, Member of the supervisory board of FMC Management AG as of July 7, 2011 |
| Performance Related | ||||||||||||||||||||||||||||||||
| Compensation | Performance Related | |||||||||||||||||||||||||||||||
| in FMC | Compensation in | Performance Related | Total | |||||||||||||||||||||||||||||
| Management AG | FMC-AG & KGaA | Compensation | compensation | |||||||||||||||||||||||||||||
| 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | 2011 | 2010 | |||||||||||||||||||||||||
| in thousand | in thousand | in thousand | in thousands | |||||||||||||||||||||||||||||
| Dr. Gerd Krick | $ | 30 | $ | — | $ | 30 | $ | — | $ | 60 | $ | — | $ | 320 | $ | 250 | ||||||||||||||||
| Dr. Dieter Schenk | 30 | — | 30 | — | 60 | — | 230 | 170 | ||||||||||||||||||||||||
Dr. Ulf M. Schneider1) | 60 | — | — | — | 60 | — | 290 | 230 | ||||||||||||||||||||||||
| Dr. Walter L. Weisman | 30 | — | 30 | — | 60 | — | 250 | 180 | ||||||||||||||||||||||||
John Gerhard Kringel2) | 16 | — | 11 | — | 27 | — | 92 | 140 | ||||||||||||||||||||||||
| William P. Johnston | 30 | — | 30 | — | 60 | — | 300 | 230 | ||||||||||||||||||||||||
Prof. Dr. Bernd Fahrholz3) | — | — | 60 | — | 60 | — | 185 | 110 | ||||||||||||||||||||||||
Rolf A. Classon4) | 14 | — | 19 | — | 33 | — | 114 | — | ||||||||||||||||||||||||
| Total | $ | 210 | $ | — | $ | 210 | $ | — | $ | 420 | $ | — | $ | 1,781 | $ | 1,310 | ||||||||||||||||
| 1) | Chairman of the supervisory board of FMC Management AG, but not member of the supervisory board of FMC-AG & Co. KGaA; | |
| 2) | Member of the supervisory board of FMC-AG & Co. KGaA until May 12, 2011 and of FMC Management AG until July 7, 2011 | |
| 3) | Member of the supervisory board of FMC-AG & Co. KGaA, but not member of the supervisory board of FMC Management AG | |
| 4) | Member of the supervisory board of FMC-AG & Co. KGaA effective from May 12, 2011 and of FMC Management AG effective from July 7, 2011 |
| C. | Board Practices |
84
Table of Contents
| • | overseeing management’s conduct of our financial reporting process and the internal accounting and financial control systems and auditing of our financial statements; | |
| • | monitoring our internal controls risk program; | |
| • | monitoring our corporate governance performance according to the German corporate governance codex; | |
| • | monitoring the independence and performance of our outside auditors; | |
| • | providing an avenue of communication among the outside auditors, management and the Supervisory Board; | |
| • | reviewing the report of our general partner on relations with related parties and for reporting to the overall supervisory board thereon; | |
| • | recommending the appointment of our independent auditors to audit our German statutory financial statements (subject to the approval by our shareholders at our Annual General Meeting) and approval of their fees; | |
| • | retaining the services of our independent auditors to audit our U.S. GAAP financial statements and approval of their fees; and | |
| • | pre-approval of all audit and non-audit services performed by KPMG, our independent auditors. |
| • | transactions between us and Fresenius SE with a value in excess of 0.25% of our consolidated revenue, and | |
| • | acquisitions and sales of significant participations and parts of our business, the spin-off of significant parts of our business, initial public offerings of significant subsidiaries and similar matters. A matter is “significant” for purposes of this approval requirement if 40% of our consolidated revenues, our consolidated balance sheet total assets or consolidated profits, determined by reference to the arithmetic average of the said amounts shown in our audited consolidated accounts for the previous three fiscal years, are affected by the matter. |
85
Table of Contents
| D. | Employees |
| 2011 | 2010 | 2009 | ||||||||||
| North America | ||||||||||||
| Dialysis Care | 37,584 | 36,488 | 35,188 | |||||||||
| Dialysis Products | 7,904 | 7,557 | 6,916 | |||||||||
| 45,488 | 44,045 | 42,104 | ||||||||||
| International | ||||||||||||
| Dialysis Care | 22,787 | 19,647 | 16,413 | |||||||||
| Dialysis Products | 10,697 | 9,584 | 9,312 | |||||||||
| 33,484 | 29,231 | 25,725 | ||||||||||
| Corporate | 187 | 176 | 159 | |||||||||
| Total Company | 79,159 | 73,452 | 67,988 | |||||||||
| E. | Share ownership |
86
Table of Contents
87
Table of Contents
88
Table of Contents
| Item 7. | Major Shareholders and Related Party Transactions |
| A. | Major Shareholders |
89
Table of Contents
| B. | Related party transactions |
| • | the term “we (or us) and our affiliates” refers only to Fresenius Medical Care AG & Co. KGaA and its subsidiaries; and | |
| • | the term “Fresenius SE and its affiliates” refers only to Fresenius SE and affiliates of Fresenius SE other than Fresenius Medical Care AG & Co. KGaA and its subsidiaries. |
90
Table of Contents
| • | to use the “Fresenius Medical Care” mark in the then current National Medical Care non-renal business if it is used as part of “Fresenius Medical Care” together with one or more descriptive words, such as “Fresenius Medical Care Home Care” or “Fresenius Medical Care Diagnostics”; | |
| • | to use the “F” logo mark in the National Medical Care non-renal business, with the consent of Fresenius SE. That consent will not be unreasonably withheld if the mark using the logo includes one or more additional descriptive words or symbols; and | |
| • | to use “Fresenius Medical Care” as a trade name in the renal business |
91
Table of Contents
92
Table of Contents
| Item 8. | Financial information |
93
Table of Contents
Per Share Amount | 2011 | 2010 | 2009 | |||||||||
| Preference share | € | 0.67 | € | 0.63 | € | 0.60 | ||||||
| Ordinary share | € | 0.65 | € | 0.61 | € | 0.58 | ||||||
| Item 9. | The Offer and Listing Details |
94
Table of Contents
| Price per | Price per | |||||||||||||||||||
| ordinary share (€) | preference share (€) | |||||||||||||||||||
| High | Low | High | Low | |||||||||||||||||
| 2012 | January | 55.05 | 53.22 | 44.90 | 43.00 | |||||||||||||||
| 2011 | December | 52.50 | 49.63 | 43.00 | 40.55 | |||||||||||||||
| November | 51.52 | 48.50 | 45.00 | 41.83 | ||||||||||||||||
| October | 53.54 | 48.66 | 45.00 | 39.13 | ||||||||||||||||
| September | 52.10 | 46.60 | 42.72 | 40.27 | ||||||||||||||||
| August | 52.72 | 45.41 | 45.90 | 37.99 | ||||||||||||||||
| 2011 | Fourth Quarter | 53.54 | 48.50 | 45.00 | 39.13 | |||||||||||||||
| Third Quarter | 55.13 | 45.41 | 45.90 | 37.99 | ||||||||||||||||
| Second Quarter | 53.06 | 48.23 | 45.50 | 40.67 | ||||||||||||||||
| First Quarter | 49.46 | 41.11 | 41.35 | 34.48 | ||||||||||||||||
| 2010 | Fourth Quarter | 45.79 | 43.01 | 38.00 | 33.50 | |||||||||||||||
| Third Quarter | 45.51 | 41.13 | 38.00 | 35.00 | ||||||||||||||||
| Second Quarter | 44.71 | 38.21 | 38.50 | 32.35 | ||||||||||||||||
| First Quarter | 41.78 | 36.10 | 35.87 | 28.20 | ||||||||||||||||
| 2011 | Annual | 55.13 | 41.11 | 45.90 | 34.48 | |||||||||||||||
| 2010 | Annual | 45.79 | 36.10 | 38.50 | 28.20 | |||||||||||||||
| 2009 | Annual | 37.71 | 26.07 | 35.30 | 25.24 | |||||||||||||||
| 2008 | Annual | 39.10 | 29.73 | 37.60 | 28.31 | |||||||||||||||
| 2007 | Annual | 38.67 | 33.05 | 36.78 | 31.32 | |||||||||||||||
95
Table of Contents
| Price per ordinary ADS ($) | Price per preference ADS ($) | |||||||||||||||||||
| High | Low | High | Low | |||||||||||||||||
| 2012 | January | 71.50 | 67.86 | 58.16 | 55.00 | |||||||||||||||
| 2011 | December | 69.75 | 65.51 | 59.01 | 55.00 | |||||||||||||||
| November | 70.73 | 64.96 | 61.49 | 57.24 | ||||||||||||||||
| October | 76.03 | 64.97 | 61.49 | 55.27 | ||||||||||||||||
| September | 71.04 | 65.93 | 58.72 | 55.27 | ||||||||||||||||
| August | 74.65 | 64.20 | 60.47 | 52.51 | ||||||||||||||||
| 2011 | Fourth Quarter | 76.03 | 64.96 | 61.49 | 55.00 | |||||||||||||||
| Third Quarter | 79.92 | 64.20 | 62.10 | 52.51 | ||||||||||||||||
| Second Quarter | 78.57 | 68.58 | 62.80 | 54.22 | ||||||||||||||||
| First Quarter | 69.74 | 55.75 | 55.17 | 46.00 | ||||||||||||||||
| 2010 | Fourth Quarter | 64.01 | 56.53 | 53.11 | 48.00 | |||||||||||||||
| Third Quarter | 61.80 | 52.17 | 49.00 | 45.00 | ||||||||||||||||
| Second Quarter | 56.70 | 47.57 | 49.70 | 38.88 | ||||||||||||||||
| First Quarter | 56.20 | 49.62 | 50.72 | 42.70 | ||||||||||||||||
| 2011 | Annual | 79.92 | 55.75 | 62.80 | 46.00 | |||||||||||||||
| 2010 | Annual | 64.01 | 47.57 | 53.11 | 38.88 | |||||||||||||||
| 2009 | Annual | 54.96 | 35.66 | 50.00 | 32.00 | |||||||||||||||
| 2008 | Annual | 59.01 | 39.84 | 55.00 | 28.87 | |||||||||||||||
| 2007 | Annual | 56.70 | 43.69 | 53.50 | 40.00 | |||||||||||||||
| Item 10. | Additional information |
| B. | Articles of Association |
| • | the development, production and distribution of as well as the trading in healthcare products, systems and procedures, including dialysis; | |
| • | the projecting, planning, establishment, acquisition and operation of healthcare businesses, including dialysis centers, also in separate enterprises or through third parties as well as the participation in such dialysis centers; |
96
Table of Contents
| • | the development, production and distribution of other pharmaceutical products and the provision of services in this field; | |
| • | the provision of advice in the medical and pharmaceutical areas as well as scientific information and documentation; | |
| • | the provision of laboratory services for dialysis and non-dialysis patients and homecare medical services. |
97
Table of Contents
98
Table of Contents
| • | we increase our share capital against contributions in cash; | |
| • | the amount of the capital increase does not exceed 10% of our existing share capital; and | |
| • | the issue price of the new shares is not significantly lower than the price for the shares quoted on a stock exchange. |
99
Table of Contents
100
Table of Contents
| • | Cash. The depositary shall convert cash distributions from foreign currency to U.S. dollars if this is permissible and can be done on a reasonable basis. The depositary will endeavor to distribute cash in a practicable manner, and may deduct any taxes or other governmental charges required to be withheld, any expenses of converting foreign currency and transferring funds to the United States, and certain other fees and expenses. In addition, before making a distribution the depositary will deduct any taxes withheld. If exchange rates fluctuate during a time when the depositary cannot convert a foreign currency, investors may lose some or all of the value of the distribution. | |
| • | Shares. If we make a distribution in shares, the depositary may deliver additional ADSs to represent the distributed shares, unless the number of ordinary shares or preference shares represented by our ADSs is adjusted in connection with the distribution. Only whole ADSs will be issued. Any shares which would result in fractional ADSs will be sold and the net proceeds will be distributed to the ADS holders otherwise entitled to receive fractional ADSs. | |
| • | Rights to receive additional shares. In the case of a distribution of pre-emption rights to subscribe for ordinary shares or preference shares, or other subscription rights, if we provide satisfactory evidence that the depositary may lawfully distribute the rights, the depositary may arrange for ADS holders to instruct the depositary as to the exercise of the rights. However, if we do not furnish the required evidence or if the depositary determines it is not practical to distribute the rights, the depositary may: |
| • | allow the rights to lapse, in which case ADS holders will receive nothing, or | |
| • | sell the rights if practicable and distribute the net proceeds as cash. |
| • | Other Distributions. If we make a distribution of securities or property other than those described above, the depositary may either: |
| • | distribute the securities or property in any manner it deems fair and equitable; | |
| • | sell the securities or property and distribute any net proceeds in the same way it distributes cash; or | |
| • | hold the distributed property in which case the ADSs will also represent the distributed property. |
101
Table of Contents
| • | temporary delays caused by closing our transfer books or those of the depositary, or the deposit of shares in connection with voting at a shareholders’ meeting, or the payment of dividends, | |
| • | the payment of fees, taxes and similar charges, or | |
| • | compliance with any U.S. or foreign laws or governmental regulations relating to the ADRs. |
102
Table of Contents
| • | we do not wish a discretionary proxy to be given; | |
| • | we think there is substantial shareholder opposition to the particular question; or | |
| • | we think the particular question would have an adverse impact on our shareholders. |
| • | are only obligated to take the actions specifically set forth in the applicable deposit agreement without negligence or bad faith; | |
| • | are not liable if we are or it is prevented or delayed by law or circumstances beyond our control from performing our or its obligations under the applicable deposit agreement; | |
| • | are not liable if we or it exercises discretion permitted under the applicable deposit agreement; | |
| • | have no obligation to become involved in a lawsuit or other proceeding related to the ADSs or the applicable deposit agreement on your behalf or on behalf of any other person; and | |
| • | may rely upon any documents we believe or it believes in good faith to be genuine and to have been signed or presented by the proper person. |
103
Table of Contents
| • | payment of stock transfer or other taxes or other governmental charges and transfer or registration fees charged by third parties for the transfer of any shares or other deposited securities; | |
| • | satisfactory proof of the identity and genuineness of any signature or other information it deems necessary; and | |
| • | compliance with regulations it may establish, from time to time, consistent with the deposit agreement, including presentation of transfer documents. |
104
Table of Contents
| • | maintain the effectiveness of (i) the deposit agreement for the ordinary shares, or a similar agreement, and to assure that the ADSs evidencing the ordinary shares are listed on either the New York Stock Exchange or the Nasdaq Stock Market and (ii), while the preference ADSs are eligible for listing on the New York Exchange or the Nasdaq Stock Market, the deposit agreement for the preference shares, or a similar agreement, and to assure that, if eligible for such listing, the ADSs evidencing the preference shares are listed on either the New York Stock Exchange or the Nasdaq Stock Market; | |
| • | file all reports, required by the New York Stock Exchange or the Nasdaq Stock Market, as applicable, the Securities Act, the Securities Exchange Act of 1934, as amended, and all other applicable laws; | |
| • | prepare all financial statements required for any filing in accordance with generally accepted accounting principles of the U.S. (“U.S. GAAP”); | |
| • | on an annual basis, prepare audited consolidated financial statements in accordance with U.S. GAAP, and, on a quarterly basis, prepare and furnish to the SEC consolidated financial statements prepared in accordance with U.S. GAAP under cover ofform 6-K or a comparable successor form; | |
| • | furnish materials with the SEC with respect to annual and special shareholder meetings under cover ofForm 6-K and make the materials available to the depositary for distribution to holders of ordinary share ADSs and, if we maintain a preference share ADS facility, to holders of preference share ADSs at any time that holders of preference shares are entitled to voting rights; and | |
| • | make available to the depositary for distribution to holders of ADSs representing our ordinary shares and, if we maintain a preference share ADS facility, ADSs representing our preference shares on an annual basis, a copy of any report prepared by the supervisory board or the supervisory board of the general partner and provided to our shareholders generally pursuant to Section 314(2) of the German Stock Corporation Act, or any successor provision. These reports concern the results of the supervisory board’s examination of the managing board’s report on our relation with affiliated enterprises. |
| • | Fresenius SE or its affiliates acquire all our voting shares; | |
| • | Fresenius SE’s beneficial ownership of our outstanding share capital is reduced to less than 25%; | |
| • | Fresenius SE or an affiliate of Fresenius SE ceases to own the general partner interest in FMC-AG & Co. KGaA; or |
105
Table of Contents
| • | we no longer meet the minimum threshold for obligatory registration of the ordinary shares or ADSs representing our ordinary shares and the preference shares or ADSs representing our preference shares, as applicable, under Section 12(g)(1) of the Securities Exchange Act of 1934, as amended, andRule 12g-1 thereunder. |
| C. | Material contracts |
| D. | Exchange controls |
106
Table of Contents
| E. | Taxation |
107
Table of Contents
108
Table of Contents
| H. | Documents on display |
109
Table of Contents
| Item 11. | Quantitative and Qualitative Disclosures About Market Risk |
| • | changes in reimbursement rates; | |
| • | intense competition; | |
| • | foreign exchange rate and interest rate fluctuations; | |
| • | varying degrees of acceptance of new product introductions; | |
| • | technological developments in our industry; | |
| • | uncertainties in litigation or investigative proceedings and regulatory developments in the healthcare sector; and | |
| • | the availability of financing. |
110
Table of Contents
| Credit | ||||||||||||||||||||||||||||||||
| 2012 | 2013 | 2014 | 2015 | 2016 | Total | Fair value | risk | |||||||||||||||||||||||||
| Purchase of EUR against US$ | $ | 837 | 31 | — | — | — | $ | 868 | $ | (18 | ) | $ | 1 | |||||||||||||||||||
| Sale of EUR against US$ | 978 | — | — | — | — | 978 | 50 | 50 | ||||||||||||||||||||||||
| Purchase of EUR against others | 922 | 117 | 30 | 28 | — | 1,097 | (36 | ) | 9 | |||||||||||||||||||||||
| Sale of EUR against others | 337 | 58 | 30 | 28 | — | 453 | 3 | 3 | ||||||||||||||||||||||||
| Others | 31 | 1 | — | — | — | 32 | (3 | ) | — | |||||||||||||||||||||||
| Total | $ | 3,105 | 207 | 60 | 56 | — | $ | 3,428 | $ | (4 | ) | $ | 63 | |||||||||||||||||||
111
Table of Contents
| Year’s | Year’s | Year’s | Year’s | |||||||||||||
Year ending December 31, | High | Low | Average | Close | ||||||||||||
| 2007 US$ per EUR | 1.4874 | 1.2893 | 1.3705 | 1.4721 | ||||||||||||
| 2008 US$ per EUR | 1.5990 | 1.2460 | 1.4713 | 1.3917 | ||||||||||||
| 2009 US$ per EUR | 1.5120 | 1.2555 | 1.3948 | 1.4406 | ||||||||||||
| 2010 US$ per EUR | 1.4563 | 1.1942 | 1.3259 | 1.3362 | ||||||||||||
| 2011 US$ per EUR | 1.4882 | 1.2889 | 1.3920 | 1.2939 | ||||||||||||
| The Reference Rate on February 17, 2012 was $1.3159 per €1.00. | ||||||||||||||||
112
Table of Contents
| Fair Value | ||||||||||||||||||||||||||||||||
| Dec. 31, | ||||||||||||||||||||||||||||||||
| 2012 | 2013 | 2014 | 2015 | 2016 | Thereafter | Totals | 2011 | |||||||||||||||||||||||||
| FLOATING RATE US$ DEBT | ||||||||||||||||||||||||||||||||
| Principal payments on Senior Credit Agreement | $ | 1,262 | 1,534 | $ | 2,796 | $ | 2,775 | |||||||||||||||||||||||||
| Variable interest rate = 2.05% | ||||||||||||||||||||||||||||||||
| Accounts receivable securitization programs | $ | 535 | $ | 535 | $ | 535 | ||||||||||||||||||||||||||
| Variable interest rate = 0.42% | ||||||||||||||||||||||||||||||||
| EIB loans | $ | 165 | $ | 165 | $ | 165 | ||||||||||||||||||||||||||
| Variable interest rate = 0.68% | ||||||||||||||||||||||||||||||||
| FLOATING RATE € DEBT | ||||||||||||||||||||||||||||||||
| Euro Notes 2009/2012 | $ | 155 | $ | 155 | $ | 157 | ||||||||||||||||||||||||||
| Variable interest rate = 6.79% | ||||||||||||||||||||||||||||||||
| Euro Notes 2009/2014 | $ | 5 | 5 | 29 | $ | 39 | $ | 40 | ||||||||||||||||||||||||
| Variable interest rate = 7.29% | ||||||||||||||||||||||||||||||||
| EIB loan | $ | 181 | $ | 181 | $ | 181 | ||||||||||||||||||||||||||
| Variable interest rate = 2.32% | ||||||||||||||||||||||||||||||||
| Senior Notes 2011/2016 | ||||||||||||||||||||||||||||||||
| Variable interest rate = 5.072% | $ | 129 | $ | 129 | $ | 131 | ||||||||||||||||||||||||||
| FIXED RATE US$ DEBT | ||||||||||||||||||||||||||||||||
| Senior Notes 2007/2017; fixed interest rate = 6.875% | $ | 495 | $ | 495 | $ | 513 | ||||||||||||||||||||||||||
| Senior Notes 2011/2018; fixed interest rate = 6.50% | $ | 395 | $ | 395 | $ | 428 | ||||||||||||||||||||||||||
| Senior Notes 2011/2021; fixed interest rate = 5.75% | $ | 644 | $ | 644 | $ | 637 | ||||||||||||||||||||||||||
| FIXED RATE € DEBT | ||||||||||||||||||||||||||||||||
| Euro Notes 2009/2012 | $ | 46 | $ | 46 | $ | 48 | ||||||||||||||||||||||||||
| Fixed interest rate = 7.4065% | ||||||||||||||||||||||||||||||||
| Euro Notes 2009/2014 | $ | 2 | 2 | 15 | $ | 19 | $ | 21 | ||||||||||||||||||||||||
| Fixed interest rate = 8.3835% | ||||||||||||||||||||||||||||||||
| Senior Notes 2010/2016 | $ | 321 | $ | 321 | $ | 340 | ||||||||||||||||||||||||||
| Fixed interest rate = 5.50% | ||||||||||||||||||||||||||||||||
| Senior Notes 2011/2018 | $ | 511 | $ | 511 | $ | 556 | ||||||||||||||||||||||||||
| Fixed interest rate = 6.50% | ||||||||||||||||||||||||||||||||
| Senior Notes 2011/2021 | $ | 388 | $ | 388 | $ | 384 | ||||||||||||||||||||||||||
| Fixed interest rate = 5.25% | ||||||||||||||||||||||||||||||||
| INTEREST RATE DERIVATIVES | ||||||||||||||||||||||||||||||||
| US$ Payer Swaps Notional amount | $ | 2,650 | $ | 2,650 | $ | (124 | ) | |||||||||||||||||||||||||
| Average fixed pay rate = 3.55% | 3.55 | % | ||||||||||||||||||||||||||||||
Receive rate =3-month $LIBOR | ||||||||||||||||||||||||||||||||
| € Payer Swaps Notional Amount | $ | 129 | 129 | $ | 258 | (7 | ) | |||||||||||||||||||||||||
| Average fixed pay rate = 2.27% | 2.80 | % | 1.73 | % | ||||||||||||||||||||||||||||
Receive rate =3-month EURIBOR | ||||||||||||||||||||||||||||||||
113
Table of Contents
| Item 12. | Description of Securities other than Equity Securities |
| D. | American Depositary Shares |
| D.3. | Fees and expenses |
| • | a fee of $0.02 or less per ADS (or portion thereof) for any cash distribution made pursuant to the deposit agreement; | |
| • | a fee of $0.02 per ADS (or portion thereof) per year for services performed by the depositary in administering our ADS program (which fee shall be assessed against holders of ADSs as of the record date set by the depositary not more than once each calendar year and shall be payable in the manner described in the next succeeding provision); | |
| • | any other charge payable by any of the depositary, any of the depositary’s agents, including, without limitation, the custodian, or the agents of the depositary’s agents in connection with the servicing of our shares or other deposited securities (which charge shall be assessed against registered holders of our ADSs as of the record date or dates set by the depositary and shall be payable at the sole discretion of the depositary by billing such registered holders or by deducting such charge from one or more cash dividends or other cash distributions); | |
| • | a fee for the distribution of securities (or the sale of securities in connection with a distribution), such fee being in an amount equal to the fee for the execution and delivery of ADSs which would have been charged as a result of the deposit of such securities (treating all such securities as if they were shares) but which securities or the net cash proceeds from the sale thereof are instead distributed by the depositary to those holders entitled thereto; | |
| • | stock transfer or other taxes and other governmental charges; | |
| • | cable, telex and facsimile transmission and delivery charges incurred at the request of holders of our shares; | |
| • | transfer or registration fees for the registration of transfer of deposited securities on any applicable register in connection with the deposit or withdrawal of deposited securities; and | |
| • | expenses of the depositary in connection with the conversion of foreign currency into U.S. dollars. |
| D.4. | Amounts payable by the depositary to the Company |
114
Table of Contents
| Item 13. | Defaults, Dividend Arrearages and Delinquencies |
| Item 14. | Material Modifications to the Rights of Security Holders and Use of Proceeds |
| Item 15A. | Disclosure Controls and Procedures |
| Item 15B. | Management’s annual report on internal control over financial reporting |
115
Table of Contents
| Item 15C. | Attestation report of the registered public accounting firm |
| Item 15D. | Changes in Internal Control over Financial Reporting |
| Item 16A. | Audit Committee Financial Expert |
| Item 16B. | Code of Ethics |
| Item 16C. | Principal Accountant Fees and Services. |
| 2011 | 2010 | |||||||
| (in thousands) | ||||||||
| Audit fees | $ | 10,236 | $ | 10,433 | ||||
| Audit related fees | 885 | 856 | ||||||
| Tax fees | 707 | 932 | ||||||
| Total | $ | 11,828 | $ | 12,221 | ||||
116
Table of Contents
| Item 16D. | Exemptions from the Listing Standards for Audit Committees |
| Item 16E. | Purchase of Equity Securities by the Issuer and Affiliated Purchasers |
| Item 16F. | Change in Registrant’s Certifying Accountant |
| Item 16G. | Corporate Governance |
117
Table of Contents
118
Table of Contents
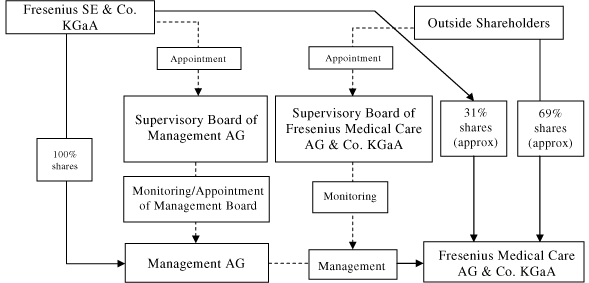
119
Table of Contents
| • | The acquisition, disposal and encumbrance of real property if the value or the amount to be secured exceeds a specified threshold (€10 million); | |
| • | The acquisition, formation, disposal or encumbrance of an equity participation in other enterprises if the value of the transaction exceeds a specified threshold (€10 million); | |
| • | The adoption of new or the abandonment of existing lines of business or establishments; | |
| • | Conclusion, amendment and termination of affiliation agreements; and | |
| • | Certain inter-company transactions. |
120
Table of Contents
121
Table of Contents
122
Table of Contents
123
Table of Contents
124
Table of Contents
| Item 17. | Financial Statements |
| Item 18. | Financial Statements |
| Item 19. | Exhibits |
125
Table of Contents
126
Table of Contents
127
Table of Contents
128
Table of Contents
129
Table of Contents
| (1) | Confidential treatment has been granted as to certain portions of this document in accordance with the applicable rules of the Securities and Exchange Commission. |
| (2) | Portions of this exhibit have been omitted pursuant to a request for confidential treatment and file separately with the Securities and Exchange Commission. |
130
Table of Contents
a partnership limited by shares, represented by:
its general partner
| Title: | Chief Executive Officer and |
| Title: | Chief Financial Officer and member |
131
Audited Consolidated Financial Statements | ||||
| F-2 | ||||
| F-3 | ||||
| F-4 | ||||
| F-5 | ||||
| F-6 | ||||
| F-7 | ||||
| F-8 | ||||
| F-9 | ||||
| F-10 | ||||
| S-II |
F-1
Table of Contents
OVER FINANCIAL REPORTING
a partnership limited by shares, represented by:
| Title: | Chief Executive Officer and Chairman |
| Title: | Chief Financial Officer and member |
F-2
Table of Contents
F-3
Table of Contents
ON INTERNAL CONTROL OVER FINANCIAL REPORTING
F-4
Table of Contents
| 2011 | 2010 | 2009 | ||||||||||
| Net revenue: | ||||||||||||
| Dialysis Care | $ | 9,507,173 | $ | 9,070,546 | $ | 8,350,233 | ||||||
| Dialysis Products | 3,287,887 | 2,982,944 | 2,897,244 | |||||||||
| 12,795,060 | 12,053,490 | 11,247,477 | ||||||||||
| Costs of revenue: | ||||||||||||
| Dialysis Care | 6,677,215 | 6,345,135 | 5,945,724 | |||||||||
| Dialysis Products | 1,597,144 | 1,563,634 | 1,470,241 | |||||||||
| 8,274,359 | 7,908,769 | 7,415,965 | ||||||||||
| Gross profit | 4,520,701 | 4,144,721 | 3,831,512 | |||||||||
| Operating (income) expenses: | ||||||||||||
| Selling, general and administrative | 2,365,934 | 2,133,333 | 1,986,640 | |||||||||
| Research and development | 110,834 | 96,532 | 93,810 | |||||||||
| Income from equity method investees | (30,959 | ) | (8,949 | ) | (4,534 | ) | ||||||
| Operating income | 2,074,892 | 1,923,805 | 1,755,596 | |||||||||
| Other (income) expense: | ||||||||||||
| Interest income | (59,825 | ) | (25,409 | ) | (21,397 | ) | ||||||
| Interest expense | 356,358 | 305,473 | 321,360 | |||||||||
| Income before income taxes | 1,778,359 | 1,643,741 | 1,455,633 | |||||||||
| Income tax expense | 601,097 | 578,345 | 490,413 | |||||||||
| Net income | 1,177,262 | 1,065,396 | 965,220 | |||||||||
| Less: Net income attributable to noncontrolling interests | 106,108 | 86,879 | 74,082 | |||||||||
| Net income attributable to shareholders of FMC-AG & Co. KGaA | $ | 1,071,154 | $ | 978,517 | $ | 891,138 | ||||||
| Basic income per ordinary share | $ | 3.54 | $ | 3.25 | $ | 2.99 | ||||||
| Fully diluted income per ordinary share | $ | 3.51 | $ | 3.24 | $ | 2.99 | ||||||
F-5
Table of Contents
| 2011 | 2010 | 2009 | ||||||||||
| Net Income | $ | 1,177,262 | $ | 1,065,396 | $ | 965,220 | ||||||
| Gain (loss) related to cash flow hedges | (102,446 | ) | (8,109 | ) | 30,082 | |||||||
| Actuarial gains (losses) on defined benefit pension plans | (81,906 | ) | (35,654 | ) | 9,708 | |||||||
| Gain (loss) related to foreign currency translation | (181,234 | ) | (110,888 | ) | 82,545 | |||||||
| Income tax benefit (expense) related to components of other comprehensive income | 72,617 | 12,821 | (18,971 | ) | ||||||||
| Other comprehensive income (loss), net of tax | (292,969 | ) | (141,830 | ) | 103,364 | |||||||
| Total comprehensive income | $ | 884,293 | $ | 923,566 | $ | 1,068,584 | ||||||
| Comprehensive income attributable to noncontrolling interests | 104,861 | 89,370 | 75,886 | |||||||||
| Comprehensive income attributable to shareholders of FMC-AG & Co. KGaA | $ | 779,432 | $ | 834,196 | $ | 992,698 | ||||||
F-6
Table of Contents
| December 31, | December 31, | |||||||
| 2011 | 2010 | |||||||
| Assets | �� | |||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 457,292 | $ | 522,870 | ||||
| Trade accounts receivable less allowance for doubtful accounts of $299,751 in 2011 and $277,139 in 2010 | 2,798,318 | 2,573,258 | ||||||
| Accounts receivable from related parties | 111,008 | 113,976 | ||||||
| Inventories | 967,496 | 809,097 | ||||||
| Prepaid expenses and other current assets | 1,035,366 | 783,231 | ||||||
| Deferred taxes | 325,539 | 350,162 | ||||||
| Total current assets | 5,695,019 | 5,152,594 | ||||||
| Property, plant and equipment, net | 2,629,701 | 2,527,292 | ||||||
| Intangible assets | 686,652 | 692,544 | ||||||
| Goodwill | 9,186,650 | 8,140,468 | ||||||
| Deferred taxes | 88,159 | 93,168 | ||||||
| Investment in equity method investees | 692,025 | 250,373 | ||||||
| Other assets and notes receivable | 554,644 | 238,222 | ||||||
| Total assets | $ | 19,532,850 | $ | 17,094,661 | ||||
| Liabilities and shareholders’ equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 541,423 | $ | 420,637 | ||||
| Accounts payable to related parties | 111,226 | 121,887 | ||||||
| Accrued expenses and other current liabilities | 1,704,273 | 1,537,423 | ||||||
| Short-term borrowings and other financial liabilities | 98,801 | 670,671 | ||||||
| Short-term borrowings from related parties | 28,013 | 9,683 | ||||||
| Current portion of long-term debt and capital lease obligations | 1,589,776 | 263,982 | ||||||
| Company-obligated mandatorily redeemable preferred securities of subsidiary Fresenius Medical Care Capital Trusts holding solely Company-guaranteed debentures of subsidiaries — current portion | — | 625,549 | ||||||
| Income tax payable | 162,354 | 117,542 | ||||||
| Deferred taxes | 26,745 | 22,349 | ||||||
| Total current liabilities | 4,262,611 | 3,789,723 | ||||||
| Long-term debt and capital lease obligations, less current portion | 5,494,810 | 4,309,676 | ||||||
| Other liabilities | 236,628 | 294,015 | ||||||
| Pension liabilities | 290,493 | 190,150 | ||||||
| Income tax payable | 189,000 | 200,581 | ||||||
| Deferred taxes | 587,800 | 506,896 | ||||||
| Total liabilities | 11,061,342 | 9,291,041 | ||||||
| Noncontrolling interests subject to put provisions | 410,491 | 279,709 | ||||||
| Shareholders’ equity: | ||||||||
| Preference shares, no par value, €1.00 nominal value, 7,066,522 shares authorized, 3,965,691 issued and outstanding | 4,452 | 4,440 | ||||||
| Ordinary shares, no par value, €1.00 nominal value, 385,396,450 shares authorized, 300,164,922 issued and outstanding | 371,649 | 369,002 | ||||||
| Additional paid-in capital | 3,362,633 | 3,339,781 | ||||||
| Retained earnings | 4,648,585 | 3,858,080 | ||||||
| Accumulated other comprehensive (loss) income | (485,767 | ) | (194,045 | ) | ||||
| Total FMC-AG & Co. KGaA shareholders’ equity | 7,901,552 | 7,377,258 | ||||||
| Noncontrolling interests not subject to put provisions | 159,465 | 146,653 | ||||||
| Total equity | 8,061,017 | 7,523,911 | ||||||
| Total liabilities and equity | $ | 19,532,850 | $ | 17,094,661 | ||||
F-7
Table of Contents
| 2011 | 2010 | 2009 | ||||||||||
| Operating Activities: | ||||||||||||
| Net income | $ | 1,177,262 | $ | 1,065,396 | 965,220 | |||||||
| Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||||||
| Depreciation and amortization | 557,283 | 503,224 | 457,085 | |||||||||
| Change in deferred taxes, net | 147,454 | 14,687 | 22,002 | |||||||||
| (Gain) loss on sale of investments | (7,679 | ) | (5,888 | ) | (1,250 | ) | ||||||
| (Gain) loss on sale of fixed assets | (1,306 | ) | (628 | ) | 1,308 | |||||||
| Compensation expense related to stock options | 29,071 | 27,981 | 33,746 | |||||||||
| Cash outflow from hedging | (58,113 | ) | — | — | ||||||||
| Changes in assets and liabilities, net of amounts from businesses acquired: | ||||||||||||
| Trade accounts receivable, net | (252,794 | ) | (300,274 | ) | (41,994 | ) | ||||||
| Inventories | (151,890 | ) | 18,326 | (88,933 | ) | |||||||
| Prepaid expenses, other current and non-current assets | (150,090 | ) | (60,305 | ) | (147,105 | ) | ||||||
| Accounts receivable from related parties | (11,669 | ) | 125,962 | (144,224 | ) | |||||||
| Accounts payable to related parties | (4,495 | ) | (135,001 | ) | 138,506 | |||||||
| Accounts payable, accrued expenses and other current and non-current liabilities | 132,406 | 124,279 | 71,092 | |||||||||
| Income tax payable | 41,042 | (9,634 | ) | 73,164 | ||||||||
| Net cash provided by (used in) operating activities | 1,446,482 | 1,368,125 | 1,338,617 | |||||||||
| Investing Activities: | ||||||||||||
| Purchases of property, plant and equipment | (597,855 | ) | (523,629 | ) | (573,606 | ) | ||||||
| Proceeds from sale of property, plant and equipment | 27,325 | 16,108 | 11,730 | |||||||||
| Acquisitions and investments, net of cash acquired, and purchases of intangible assets | (1,785,329 | ) | (764,338 | ) | (188,113 | ) | ||||||
| Proceeds from divestitures | 9,990 | 146,835 | 51,965 | |||||||||
| Net cash provided by (used in) investing activities | (2,345,869 | ) | (1,125,024 | ) | (698,024 | ) | ||||||
| Financing Activities: | ||||||||||||
| Proceeds from short-term borrowings and other financial liabilities | 189,987 | 281,022 | 107,192 | |||||||||
| Repayments of short-term borrowings and other financial liabilities | (248,821 | ) | (258,561 | ) | (169,175 | ) | ||||||
| Proceeds from short-term borrowings from related parties | 146,872 | — | 18,830 | |||||||||
| Repayments of short-term borrowings from related parties | (127,015 | ) | — | (118,422 | ) | |||||||
| Proceeds from long-term debt and capital lease obligations (net of debt issuance costs and other hedging costs of $127,854 in 2011 and $31,458 in 2010) | 2,706,105 | 947,346 | 709,540 | |||||||||
| Repayments of long-term debt and capital lease obligations | (957,235 | ) | (1,072,941 | ) | (566,241 | ) | ||||||
| Redemption of trust preferred securities | (653,760 | ) | — | — | ||||||||
| Increase (decrease) of accounts receivable securitization program | 24,500 | 296,000 | (325,000 | ) | ||||||||
| Proceeds from exercise of stock options | 94,893 | 109,518 | 72,394 | |||||||||
| Dividends paid | (280,649 | ) | (231,967 | ) | (231,940 | ) | ||||||
| Distributions to noncontrolling interests | (129,542 | ) | (111,550 | ) | (68,004 | ) | ||||||
| Contributions from noncontrolling interests | 27,824 | 26,416 | 12,699 | |||||||||
| Net cash provided by (used in) financing activities | 793,159 | (14,717 | ) | (558,127 | ) | |||||||
| Effect of exchange rate changes on cash and cash equivalents | 40,650 | (6,739 | ) | (2,825 | ) | |||||||
| Cash and Cash Equivalents: | ||||||||||||
| Net increase (decrease) in cash and cash equivalents | (65,578 | ) | 221,645 | 79,641 | ||||||||
| Cash and cash equivalents at beginning of period | 522,870 | 301,225 | 221,584 | |||||||||
| Cash and cash equivalents at end of period | $ | 457,292 | $ | 522,870 | 301,225 | |||||||
F-8
Table of Contents
| Total | ||||||||||||||||||||||||||||||||||||||||
| Accumulated | FMC-AG & | Noncontrolling | ||||||||||||||||||||||||||||||||||||||
| Preference Shares | Ordinary Shares | Additional | Other | Co. KGaA | interests not | |||||||||||||||||||||||||||||||||||
| Number of | No par | Number of | No par | paid in | Retained | comprehensive | shareholders’ | subject to put | Total | |||||||||||||||||||||||||||||||
| shares | value | shares | value | capital | earnings | income (loss) | equity | provisions | Equity | |||||||||||||||||||||||||||||||
| Balance at December 31, 2008 | 3,810,540 | $ | 4,240 | 293,932,036 | $ | 363,076 | $ | 3,188,089 | $ | 2,452,332 | $ | (151,284 | ) | $ | 5,856,453 | $ | 104,167 | $ | 5,960,620 | |||||||||||||||||||||
| Proceeds from exercise of options and related tax effects | 73,788 | 103 | 1,814,599 | 2,596 | 64,585 | — | — | 67,284 | — | 67,284 | ||||||||||||||||||||||||||||||
| Compensation expense related to stock options | — | — | — | — | 33,746 | — | — | 33,746 | — | 33,746 | ||||||||||||||||||||||||||||||
| Dividends paid | — | — | — | — | — | (231,940 | ) | — | (231,940 | ) | — | (231,940 | ) | |||||||||||||||||||||||||||
| Purchase/ sale of noncontrolling interests | — | — | — | — | (3,138 | ) | — | — | (3,138 | ) | 12,929 | 9,791 | ||||||||||||||||||||||||||||
| Contributions from/ to noncontrolling interests | — | — | — | — | — | — | — | — | (41,284 | ) | (41,284 | ) | ||||||||||||||||||||||||||||
| Changes in fair value of noncontrolling interests subject to put provisions | — | — | — | — | (39,816 | ) | — | — | (39,816 | ) | — | (39,816 | ) | |||||||||||||||||||||||||||
| Net income | — | — | — | — | — | 891,138 | — | 891,138 | 45,487 | 936,625 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | — | — | — | — | — | — | 101,560 | 101,560 | 1,804 | 103,364 | ||||||||||||||||||||||||||||||
| Comprehensive income | — | — | — | — | — | — | — | 992,698 | 47,291 | 1,039,989 | ||||||||||||||||||||||||||||||
| Balance at December 31, 2009 | 3,884,328 | $ | 4,343 | 295,746,635 | $ | 365,672 | $ | 3,243,466 | $ | 3,111,530 | $ | (49,724 | ) | $ | 6,675,287 | $ | 123,103 | $ | 6,798,390 | |||||||||||||||||||||
| Proceeds from exercise of options and related tax effects | 72,840 | 97 | 2,532,366 | 3,330 | 98,819 | — | — | 102,246 | — | 102,246 | ||||||||||||||||||||||||||||||
| Compensation expense related to stock options | — | — | — | — | 27,981 | — | — | 27,981 | — | 27,981 | ||||||||||||||||||||||||||||||
| Dividends paid | — | — | — | — | — | (231,967 | ) | — | (231,967 | ) | — | (231,967 | ) | |||||||||||||||||||||||||||
| Purchase/ sale of noncontrolling interests | — | — | — | — | (6,263 | ) | — | — | (6,263 | ) | 17,295 | 11,032 | ||||||||||||||||||||||||||||
| Contributions from/ to noncontrolling interests | — | — | — | — | — | — | — | — | (54,225 | ) | (54,225 | ) | ||||||||||||||||||||||||||||
| Changes in fair value of noncontrolling interests subject to put provisions | — | — | — | — | (24,222 | ) | — | — | (24,222 | ) | — | (24,222 | ) | |||||||||||||||||||||||||||
| Net income | — | — | — | — | — | 978,517 | — | 978,517 | 58,040 | 1,036,557 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | — | — | — | — | — | — | (144,321 | ) | (144,321 | ) | 2,440 | (141,881 | ) | |||||||||||||||||||||||||||
| Comprehensive income | — | — | — | — | — | — | — | 834,196 | 60,480 | 894,676 | ||||||||||||||||||||||||||||||
| Balance at December 31, 2010 | 3,957,168 | $ | 4,440 | 298,279,001 | $ | 369,002 | $ | 3,339,781 | $ | 3,858,080 | $ | (194,045 | ) | $ | 7,377,258 | $ | 146,653 | $ | 7,523,911 | |||||||||||||||||||||
| Proceeds from exercise of options and related tax effects | 8,523 | 12 | 1,885,921 | 2,647 | 85,887 | — | — | 88,546 | — | 88,546 | ||||||||||||||||||||||||||||||
| Compensation expense related to stock options | — | — | — | — | 29,071 | — | — | 29,071 | — | 29,071 | ||||||||||||||||||||||||||||||
| Dividends paid | — | — | — | — | — | (280,649 | ) | — | (280,649 | ) | — | (280,649 | ) | |||||||||||||||||||||||||||
| Purchase/ sale of noncontrolling interests | — | — | — | — | (5,873 | ) | — | — | (5,873 | ) | 9,662 | 3,789 | ||||||||||||||||||||||||||||
| Contributions from/ to noncontrolling interests | — | — | — | — | — | — | — | — | (59,066 | ) | (59,066 | ) | ||||||||||||||||||||||||||||
| Changes in fair value of noncontrolling interests subject to put provisions | — | — | — | — | (86,233 | ) | — | — | (86,233 | ) | — | (86,233 | ) | |||||||||||||||||||||||||||
| Net income | — | — | — | — | — | 1,071,154 | — | 1,071,154 | 63,251 | 1,134,405 | ||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | — | — | — | — | — | — | (291,722 | ) | (291,722 | ) | (1,035 | ) | (292,757 | ) | ||||||||||||||||||||||||||
| Comprehensive income | — | — | — | — | — | — | — | 779,432 | 62,216 | 841,648 | ||||||||||||||||||||||||||||||
| Balance at December 31, 2011 | 3,965,691 | $ | 4,452 | 300,164,922 | $ | 371,649 | $ | 3,362,633 | $ | 4,648,585 | $ | (485,767 | ) | $ | 7,901,552 | $ | 159,465 | $ | 8,061,017 | |||||||||||||||||||||
F-9
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(in thousands, except share data)
| 1. | The Company and Basis of Presentation |
F-10
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | |||||||
| Trade accounts receivable, net | $ | 73,172 | $ | 60,070 | ||||
| Other current assets | 65,576 | 26,981 | ||||||
| Property, plant and equipment, intangible assets & other non-current assets | 25,978 | 29,597 | ||||||
| Goodwill | 52,251 | 56,883 | ||||||
| Accounts payable, accrued expenses and other liabilities | 148,924 | 105,662 | ||||||
| Non-current loans to related parties | 13,000 | 12,998 | ||||||
| Equity | 55,053 | 54,870 | ||||||
F-11
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-12
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-13
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-14
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2. | Subsequent Events |
F-15
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 3. | Investments |
| 4. | Related Party Transactions |
F-16
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-17
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 5. | Inventories |
| 2011 | 2010 | |||||||||||
| Raw materials and purchased components | $ | 163,030 | $ | 158,163 | ||||||||
| Work in process | 60,128 | 56,345 | ||||||||||
| Finished goods | 610,569 | 475,641 | ||||||||||
| Healthcare supplies | 133,769 | 118,948 | ||||||||||
| Inventories | $ | 967,496 | $ | 809,097 | ||||||||
F-18
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 6. | Prepaid Expenses and Other Current Assets |
| 2011 | 2010 | |||||||
| Rebates | $ | 185,152 | $ | 165,218 | ||||
| Taxes Refundable | 180,721 | 124,536 | ||||||
| Derivatives | 60,877 | 7,220 | ||||||
| Payments on account | 40,476 | 38,654 | ||||||
| Prepaid rent | 39,468 | 40,321 | ||||||
| Leases receivable | 38,175 | 38,838 | ||||||
| Other | 490,497 | 368,444 | ||||||
| Total prepaid expenses and other current assets | $ | 1,035,366 | $ | 783,231 | ||||
| 7. | Property, Plant and Equipment |
| 2011 | 2010 | |||||||
| Land and improvements | $ | 53,147 | $ | 50,505 | ||||
| Buildings and improvements | 1,975,839 | 1,856,968 | ||||||
| Machinery and equipment | 3,060,132 | 2,893,643 | ||||||
| Machinery, equipment and rental equipment under capitalized leases | 36,450 | 28,406 | ||||||
| Construction in progress | 275,006 | 238,812 | ||||||
| 5,400,574 | 5,068,334 | |||||||
| Accumulated depreciation | (2,770,873 | ) | (2,541,042 | ) | ||||
| Property, plant and equipment, net | $ | 2,629,701 | $ | 2,527,292 | ||||
F-19
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 8. | Intangible Assets and Goodwill |
| 2011 | 2010 | |||||||||||||||
| Gross | Gross | |||||||||||||||
| Carrying | Accumulated | Carrying | Accumulated | |||||||||||||
| Amount | Amortization | Amount | Amortization | |||||||||||||
Amortizable Intangible Assets | ||||||||||||||||
| Non-compete Agreements | $ | 257,466 | $ | (186,659 | ) | $ | 243,575 | $ | (167,801 | ) | ||||||
| Technology | 110,866 | (32,582 | ) | 110,850 | (25,346 | ) | ||||||||||
| License and distribution agreements | 223,828 | (80,622 | ) | 233,460 | (70,189 | ) | ||||||||||
| Self-developed Software | 55,600 | (28,193 | ) | 46,955 | (21,861 | ) | ||||||||||
| Other | 317,579 | (227,274 | ) | 286,021 | (214,382 | ) | ||||||||||
| Construction in progress | 58,661 | — | 55,781 | — | ||||||||||||
| $ | 1,024,000 | $ | (555,330 | ) | $ | 976,642 | $ | (499,579 | ) | |||||||
| 2011 | 2010 | |||||||
| Carrying Amount | Carrying Amount | |||||||
Non-amortizable Intangible Assets | ||||||||
| Tradename | $ | 209,640 | $ | 210,424 | ||||
| Management contracts | 8,342 | 5,057 | ||||||
| $ | 217,982 | $ | 215,481 | |||||
Total Intangible Assets | $ | 686,652 | $ | 692,544 | ||||
| 2009 | $ | 60,225 | ||
| 2010 | $ | 70,294 | ||
| 2011 | $ | 77,845 |
| 2012 | $ | 70,716 | ||
| 2013 | $ | 66,543 | ||
| 2014 | $ | 63,162 | ||
| 2015 | $ | 61,096 | ||
| 2016 | $ | 59,968 |
F-20
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| North | ||||||||||||||||
| America | International | Corporate | Total | |||||||||||||
| Balance as of January 1, 2010 | $ | 6,694,711 | $ | 656,906 | $ | 159,817 | $ | 7,511,434 | ||||||||
| Goodwill acquired | 115,040 | 314,338 | 132 | 429,510 | ||||||||||||
| Reclassifications | 214,706 | — | — | 214,706 | ||||||||||||
| Foreign Currency Translation Adjustment | 288 | (15,470 | ) | — | (15,182 | ) | ||||||||||
| Balance as of December 31, 2010 | $ | 7,024,745 | $ | 955,774 | $ | 159,949 | $ | 8,140,468 | ||||||||
| Goodwill acquired | 517,213 | 626,863 | — | 1,144,076 | ||||||||||||
| Reclassifications | (226,900 | ) | (20,449 | ) | 247,480 | 131 | ||||||||||
| Foreign Currency Translation Adjustment | (436 | ) | (98,099 | ) | 511 | (98,024 | ) | |||||||||
| Balance as of December 31, 2011 | $ | 7,314,622 | $ | 1,464,089 | $ | 407,940 | $ | 9,186,650 | ||||||||
| 9. | Accrued Expenses and Other Current Liabilities |
| 2011 | 2010 | |||||||
| Accrued salaries, wages and incentive plan compensations | $ | 420,613 | $ | 389,434 | ||||
| Derivative financial instruments | 192,729 | 124,171 | ||||||
| Accrued insurance | 162,149 | 163,240 | ||||||
| Unapplied cash and receivable credits | 158,006 | 169,657 | ||||||
| Special charge for legal matters | 115,000 | 115,000 | ||||||
| Other | 655,776 | 575,921 | ||||||
| Total accrued expenses and other current liabilities | $ | 1,704,273 | $ | 1,537,423 | ||||
F-21
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 10. | Short-Term Borrowings, Other Financial Liabilities and Short-Term Borrowings from Related Parties |
| 2011 | 2010 | |||||||
| Borrowings under lines of credit | $ | 91,899 | $ | 131,791 | ||||
| Accounts receivable facility | — | 510,000 | ||||||
| Other financial liabilities | 6,902 | 28,880 | ||||||
| Short-term borrowings and other financial liabilities | 98,801 | 670,671 | ||||||
| Short-term borrowings from related parties (see Note 4.c.) | 28,013 | 9,683 | ||||||
| Short-term borrowings, Other financial liabilities and Short-term borrowings from related parties | $ | 126,814 | $ | 680,354 | ||||
F-22
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 11. | Long-term Debt and Capital Lease Obligations |
| 2011 | 2010 | |||||||
| Amended 2006 Senior Credit Agreement | $ | 2,795,589 | $ | 2,953,890 | ||||
| Senior Notes | 2,883,009 | 824,446 | ||||||
| Euro Notes | 258,780 | 267,240 | ||||||
| European Investment Bank Agreements | 345,764 | 351,686 | ||||||
| Accounts receivable facility | 534,500 | — | ||||||
| Capital lease obligations | 17,993 | 15,439 | ||||||
| Other | 248,952 | 160,957 | ||||||
| 7,084,587 | 4,573,658 | |||||||
| Less current maturities | (1,589,776 | ) | (263,982 | ) | ||||
| $ | 5,494,810 | $ | 4,309,676 | |||||
| • | a $1,200,000 revolving credit facility (with specifiedsub-facilities for letters of credit, borrowings in certainnon-U.S. currencies, and swing line loans in U.S. dollars and certainnon-U.S. currencies, with the total outstanding under thosesub-facilities not exceeding $1,200,000) which will be due and payable on March 31, 2013. | |
| • | a term loan facility (“Term Loan A”) of $1,215,000 also scheduled to mature on March 31, 2013. Quarterly repayments of $30,000 are required at the end of each quarter with the remaining balance outstanding due on March 31, 2013. |
F-23
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| • | a term loan facility (“Term Loan B”) of $1,521,619 scheduled to mature on March 31, 2013 with 1 quarterly repayment of $4,036 followed by 4 quarterly repayments of $379,396 each due at the end of its respective quarter. |
| Maximum Amount Available | Balance Outstanding | |||||||||||||||
| December 31, | December 31, | |||||||||||||||
| 2011 | 2010 | 2011 | 2010 | |||||||||||||
| Revolving Credit | $ | 1,200,000 | $ | 1,200,000 | $ | 58,970 | $ | 81,126 | ||||||||
| Term Loan A | 1,215,000 | 1,335,000 | 1,215,000 | 1,335,000 | ||||||||||||
| Term Loan B | 1,521,619 | 1,537,764 | 1,521,619 | 1,537,764 | ||||||||||||
| $ | 3,936,619 | $ | 4,072,764 | $ | 2,795,589 | $ | 2,953,890 | |||||||||
F-24
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| Notional | ||||||||||||||
Issuer/Transaction | Amount | Maturity | Coupon | Book value | ||||||||||
| FMC Finance VI S.A. 2010/2016 | € | 250,000 | July 15, 2016 | 5.50 | % | $ | 320,427 | |||||||
FMC Finance VIII S.A. 2011/2016(1) | € | 100,000 | October 15, 2016 | 5.072 | % | $ | 129,390 | |||||||
| FMC US Finance, Inc. 2007/2017 | $ | 500,000 | July 15, 2017 | 67/8 | % | $ | 495,118 | |||||||
| FMC Finance VIII S.A. 2011/2018 | € | 400,000 | September 15, 2018 | 6.50 | % | $ | 510,730 | |||||||
| FMC US Finance II, Inc. 2011/2018 | $ | 400,000 | September 15, 2018 | 6.50 | % | $ | 394,724 | |||||||
| FMC US Finance, Inc. 2011/2021 | $ | 650,000 | February 15, 2021 | 5.75 | % | $ | 644,450 | |||||||
| FMC Finance VII S.A. 2011/2021 | € | 300,000 | February 15, 2021 | 5.25 | % | $ | 388,170 | |||||||
| $ | 2,883,009 | |||||||||||||
| (1) | This note carries a variable interest rate which was 5.072% at December 31, 2011. |
F-25
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| Maximum amount available | ||||||||||||||||||||
| December 31, | Balance outstanding December 31, | |||||||||||||||||||
| Maturity | 2011 | 2010 | 2011 | 2010 | ||||||||||||||||
| Revolving Credit | 2013 | € | 90,000 | € | 90,000 | $ | 115,812 | $ | 115,812 | |||||||||||
| Loan 2005 | 2013 | 41,000 | 41,000 | 48,806 | 48,806 | |||||||||||||||
| Loan 2006 | 2014 | 90,000 | 90,000 | 116,451 | 120,258 | |||||||||||||||
| Loan 2009 | 2014 | 50,000 | 50,000 | 64,695 | 66,810 | |||||||||||||||
| € | 271,000 | € | 271,000 | $ | 345,764 | $ | 351,686 | |||||||||||||
F-26
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2012 | $ | 1,589,776 | ||
| 2013 | 1,776,771 | |||
| 2014 | 794,842 | |||
| 2015 | 28,049 | |||
| 2016 | 455,527 | |||
| Thereafter | 2,465,205 | |||
| $ | 7,110,172 | |||
| 12. | Employee Benefit Plans |
F-27
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | |||||||
| Change in benefit obligation: | ||||||||
| Benefit obligation at beginning of year | $ | 425,472 | $ | 386,852 | ||||
| Foreign currency translation | (6,207 | ) | (8,898 | ) | ||||
| Service cost | 10,625 | 7,982 | ||||||
| Interest cost | 24,822 | 22,615 | ||||||
| Transfer of plan participants | 61 | 181 | ||||||
| Actuarial (gain) loss | 69,769 | 26,655 | ||||||
| Benefits paid | (11,797 | ) | (9,915 | ) | ||||
| Benefit obligation at end of year | $ | 512,745 | $ | 425,472 | ||||
| Change in plan assets: | ||||||||
| Fair value of plan assets at beginning of year | $ | 232,325 | $ | 236,633 | ||||
| Actual return on plan assets | (4,174 | ) | 3,191 | |||||
| Employer contributions | 556 | 600 | ||||||
| Benefits paid | (9,717 | ) | (8,099 | ) | ||||
| Fair value of plan assets at end of year | $ | 218,990 | $ | 232,325 | ||||
| Funded status at end of year | $ | 293,755 | $ | 193,147 | ||||
F-28
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| Actuarial | ||||
| losses (gains) | ||||
| Adjustments related to pensions at January 1, 2010 | $ | 67,218 | ||
| Additions | 40,917 | |||
| Releases | (5,313 | ) | ||
| Foreign currency translation adjustment | 50 | |||
| Adjustments related to pensions at December 31, 2010 | $ | 102,872 | ||
| Additions | 91,693 | |||
| Releases | (8,737 | ) | ||
| Foreign currency translation adjustment | (1,050 | ) | ||
| Adjustments related to pensions at December 31, 2011 | $ | 184,778 | ||
in % | 2011 | 2010 | ||||||
| Discount rate | 5.10 | 5.70 | ||||||
| Rate of compensation increase | 3.69 | 4.00 | ||||||
Components of net periodic benefit cost: | 2011 | 2010 | 2009 | |||||||||
| Service cost | $ | 10,625 | $ | 7,982 | $ | 7,500 | ||||||
| Interest cost | 24,822 | 22,615 | 21,397 | |||||||||
| Expected return on plan assets | (17,750 | ) | (17,453 | ) | (15,767 | ) | ||||||
| Amortization of unrealized losses | 8,737 | 5,313 | 4,592 | |||||||||
| Settlement loss | — | — | 812 | |||||||||
| Net periodic benefit costs | $ | 26,434 | $ | 18,457 | $ | 18,534 | ||||||
in % | 2011 | 2010 | 2009 | |||||||||
| Discount rate | 5.70 | 6.00 | 6.15 | |||||||||
| Expected return of plan assets | 7.50 | 7.50 | 7.50 | |||||||||
| Rate of compensation increase | 4.00 | 4.01 | 4.19 | |||||||||
F-29
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2012 | $ | 14,233 | ||
| 2013 | 15,390 | |||
| 2014 | 16,786 | |||
| 2015 | 18,257 | |||
| 2016 | 19,934 | |||
| 2017-2021 | 126,553 |
| Fair Value Measurements at December 31, 2011 | Fair Value Measurements at December 31, 2010 | |||||||||||||||||||||||
| Quoted Prices in | Significant | Quoted Prices in | Significant | |||||||||||||||||||||
| Active Markets for | Observable | Active Markets | Observable | |||||||||||||||||||||
| Identical Assets | Inputs | for Identical Assets | Inputs | |||||||||||||||||||||
Asset Category | Total | (Level 1) | (Level 2) | Total | (Level 1) | (Level 2) | ||||||||||||||||||
Equity Investments | ||||||||||||||||||||||||
| Common Stocks | $ | — | $ | — | $ | — | $ | 2,565 | $ | 2,565 | $ | — | ||||||||||||
Index Funds(1) | 55,538 | — | 55,538 | 65,621 | — | 65,621 | ||||||||||||||||||
Fixed Income Investments | ||||||||||||||||||||||||
Government Securities(2) | 6,612 | 5,025 | 1,587 | 4,479 | 1,967 | 2,512 | ||||||||||||||||||
Corporate Bonds(3) | 143,782 | — | 143,782 | 152,564 | — | 152,564 | ||||||||||||||||||
Other Bonds(4) | 483 | — | 483 | 2,442 | — | 2,442 | ||||||||||||||||||
U.S. Treasury Money Market Funds(5) | 6,600 | 6,600 | — | 4,232 | 4,232 | — | ||||||||||||||||||
Other types of investments | ||||||||||||||||||||||||
Cash, Money Market and Mutual Funds(6) | 5,975 | 5,975 | — | 422 | 422 | — | ||||||||||||||||||
Total | $ | 218,990 | $ | 17,600 | $ | 201,390 | $ | 232,325 | $ | 9,186 | $ | 223,139 | ||||||||||||
| (1) | This category comprises low-cost equity index funds not actively managed that track the S&P 500, S&P 400, Russell 2000, MSCI Emerging Markets Index and the Morgan Stanley International EAFE Index | |
| (2) | This Category comprises fixed income investments by the U.S. government and government sponsored entities | |
| (3) | This Category primarily represents investment grade bonds of U.S. issuers from diverse industries | |
| (4) | This Category comprises privat placement bonds as well as collateralized mortgage obligations | |
| (5) | This Category represents funds that invest in treasury obligations directly or in treasury backed obligations | |
| (6) | This Category represents cash, money market funds as well as mutual funds comprised of high grade corporate bonds |
| • | Common stocks are valued at their market prices as of the balance sheet date. | |
| • | Index funds are valued based on market quotes. | |
| • | Government bonds are valued based on both market prices and market quotes. | |
| • | Corporate bonds and other bonds are valued based on market quotes as of the balance sheet date. | |
| • | Cash is stated at nominal value which equals the fair value. | |
| • | U.S. Treasury money market funds as well as other money market and mutual funds are valued at their market price. |
F-30
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 13. | Mandatorily Redeemable Trust Preferred Securities |
| Mandatory | ||||||||||||||||||||||||
| Year | Stated | Interest | Redemption | |||||||||||||||||||||
| Issued | Amount | Rate | Date | 2011 | 2010 | |||||||||||||||||||
| Fresenius Medical Care Capital Trust IV | 2001 | $225,000 | 77/8 | % | June 15, 2011 | $ | — | $ | 224,835 | |||||||||||||||
| Fresenius Medical Care Capital Trust V | 2001 | €300,000 | 73/8 | % | June 15, 2011 | — | 400,714 | |||||||||||||||||
| $ | — | $ | 625,549 | |||||||||||||||||||||
| 14. | Noncontrolling Interests Subject to Put Provisions |
F-31
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | 2009 | ||||||||||
| Beginning balance as of January 1, 2011, 2010 and 2009 | $ | 279,709 | $ | 231,303 | $ | 162,166 | ||||||
| Contributions to noncontrolling interests | (43,104 | ) | (38,964 | ) | (16,930 | ) | ||||||
| Purchase/ sale of noncontrolling interests | 37,786 | 28,969 | 12,548 | |||||||||
| Contributions from noncontrolling interests | 7,222 | 5,289 | 5,108 | |||||||||
| Changes in fair value of noncontrolling interests | 86,233 | 24,222 | 39,816 | |||||||||
| Net income | 42,857 | 28,839 | 28,595 | |||||||||
| Other comprehensive income (loss) | (212 | ) | 51 | — | ||||||||
| Ending balance as of December 31, 2011, 2010 and 2009 | $ | 410,491 | $ | 279,709 | $ | 231,303 | ||||||
| 15. | Shareholders’ Equity |
F-32
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-33
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 16. | Earnings Per Share |
| 2011 | 2010 | 2009 | ||||||||||
Numerators: | ||||||||||||
| Net income attributable to shareholders of FMC-AG & Co. KGaA | $ | 1,071,154 | $ | 978,517 | $ | 891,138 | ||||||
| less: | ||||||||||||
| Dividend preference on Preference shares | 110 | 104 | 107 | |||||||||
| Income available to all classes of shares | $ | 1,071,044 | $ | 978,413 | $ | 891,031 | ||||||
Denominators: | ||||||||||||
| Weighted average number of: | ||||||||||||
| Ordinary shares outstanding | 299,012,744 | 296,808,978 | 294,418,795 | |||||||||
| Preference shares outstanding | 3,961,617 | 3,912,348 | 3,842,586 | |||||||||
| Total weighted average shares outstanding | 302,974,361 | 300,721,326 | 298,261,381 | |||||||||
| Potentially dilutive Ordinary shares | 1,795,743 | 1,311,042 | — | |||||||||
| Potentially dilutive Preference shares | 20,184 | 35,481 | 66,314 | |||||||||
| Total weighted average Ordinary shares outstanding assuming dilution | 300,808,487 | 298,120,020 | 294,418,795 | |||||||||
| Total weighted average Preference shares outstanding assuming dilution | 3,981,801 | 3,947,829 | 3,908,900 | |||||||||
| Basic income per Ordinary share | $ | 3.54 | $ | 3.25 | $ | 2.99 | ||||||
| Plus preference per Preference shares | 0.02 | 0.03 | 0.03 | |||||||||
| Basic income per Preference share | $ | 3.56 | $ | 3.28 | $ | 3.02 | ||||||
| Fully diluted income per Ordinary share | $ | 3.51 | $ | 3.24 | $ | 2.99 | ||||||
| Plus preference per Preference shares | 0.03 | 0.03 | 0.03 | |||||||||
| Fully diluted income per Preference share | $ | 3.54 | $ | 3.27 | $ | 3.02 | ||||||
| 17. | Stock Options |
F-34
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-35
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-36
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-37
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| Weighted | Weighted | |||||||||||
| average | average | |||||||||||
| Options | exercise | exercise | ||||||||||
| (in thousands) | price | price | ||||||||||
| € | $ | |||||||||||
Stock options for ordinary shares | ||||||||||||
| Balance at December 31, 2010 | 12,152 | 33.78 | 43.71 | |||||||||
| Granted | 1,947 | 52.45 | 67.87 | |||||||||
| Exercised | 1,886 | 30.87 | 39.94 | |||||||||
| Forfeited | 188 | 34.93 | 45.20 | |||||||||
| Balance at December 31, 2011 | 12,025 | 37.24 | 48.18 | |||||||||
Stock options for preference shares | ||||||||||||
| Balance at December 31, 2010 | 59 | 19.19 | 24.83 | |||||||||
| Exercised | 9 | 22.52 | 29.14 | |||||||||
| Forfeited | 1 | 18.21 | 23.56 | |||||||||
| Balance at December 31, 2011 | 49 | 18.64 | 24.12 | |||||||||
| Fully Vested Outstanding and Exercisable Options | ||||||||||||||||||||||||
| Weighted | ||||||||||||||||||||||||
| average | Weighted | Weighted | ||||||||||||||||||||||
| Number | remaining | average | average | Aggregate | Aggregate | |||||||||||||||||||
| of | contractual | exercise | exercise | intrinsic | intrinsic | |||||||||||||||||||
| Options | life in years | price | price | value | value | |||||||||||||||||||
| (in thousands) | € | US$ | € | US$ | ||||||||||||||||||||
| Options for preference shares | 49 | 2.80 | 18.64 | 24.11 | 1,189 | 1,538 | ||||||||||||||||||
| Options for ordinary shares | 4,767 | 2.79 | 30.57 | 39.56 | 104,520 | 135,238 | ||||||||||||||||||
F-38
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | |||||||
| Expected dividend yield | 1.62% | 1.98% | ||||||
| Risk-free interest rate | 2.55% | 2.28% | ||||||
| Expected volatility | 22.22% | 22.92% | ||||||
| Expected life of options | 8 years | 7 years | ||||||
| Weighted average exercise price (in €) | 52.45 | 42.71 | ||||||
| Weighted average exercise price (in US-$) | 67.87 | 57.07 | ||||||
| 18. | Income Taxes |
| 2011 | 2010 | 2009 | ||||||||||
| Germany | $ | 344,267 | $ | 303,954 | $ | 296,326 | ||||||
| United States | 1,122,800 | 1,084,756 | 904,083 | |||||||||
| Other | 311,292 | 255,031 | 255,224 | |||||||||
| $ | 1,778,359 | $ | 1,643,741 | $ | 1,455,633 | |||||||
| 2011 | 2010 | 2009 | ||||||||||
| Current: | ||||||||||||
| Germany | $ | 67,484 | $ | 100,635 | $ | 68,442 | ||||||
| United States | 278,634 | 355,739 | 318,589 | |||||||||
| Other | 106,087 | 101,206 | 81,236 | |||||||||
| 452,205 | 557,580 | 468,267 | ||||||||||
| Deferred: | ||||||||||||
| Germany | 14,565 | (16,479 | ) | 5,041 | ||||||||
| United States | 139,282 | 52,648 | 22,498 | |||||||||
| Other | (4,955 | ) | (15,404 | ) | (5,393 | ) | ||||||
| 148,892 | 20,765 | 22,146 | ||||||||||
| $ | 601,097 | $ | 578,345 | $ | 490,413 | |||||||
F-39
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | 2009 | ||||||||||
| Expected corporate income tax expense | $ | 506,121 | $ | 471,836 | $ | 423,953 | ||||||
| Tax free income | (38,926 | ) | (24,088 | ) | (33,284 | ) | ||||||
| Income from at equity investments | (6,883 | ) | (550 | ) | — | |||||||
| Tax rate differentials | 140,079 | 118,495 | 96,237 | |||||||||
| Non-deductible expenses | 4,536 | 6,934 | 3,947 | |||||||||
| Taxes for prior years | 144 | 11,994 | 6,663 | |||||||||
| Change in valuation allowance | 5,544 | (2,259 | ) | 8,950 | ||||||||
| Noncontrolling partnership interests | (31,300 | ) | (26,870 | ) | (26,876 | ) | ||||||
| Other | 21,782 | 22,853 | 10,823 | |||||||||
| Actual income tax expense | $ | 601,097 | $ | 578,345 | $ | 490,413 | ||||||
| Effective tax rate | 33.8 | % | 35.2 | % | 33.7 | % | ||||||
| 2011 | 2010 | |||||||
| Deferred tax assets: | ||||||||
| Accounts receivable | $ | 5,943 | $ | 28,538 | ||||
| Inventory | 42,824 | 35,172 | ||||||
| Property, plant and equipment, intangible and other non current assets | 70,652 | 79,244 | ||||||
| Accrued expenses and other liabilities | 265,624 | 257,957 | ||||||
| Pensions | 87,248 | 52,773 | ||||||
| Net operating loss carryforwards, tax credit carryforwards and interest carryforwards | 91,402 | 93,165 | ||||||
| Derivatives | 60,056 | 60,199 | ||||||
| Stock-based compensation | 24,191 | 24,112 | ||||||
| Other | 12,586 | 12,626 | ||||||
| Total deferred tax assets | $ | 660,526 | $ | 643,786 | ||||
| Less: valuation allowance | (80,418 | ) | (71,799 | ) | ||||
| Net deferred tax assets | $ | 580,108 | $ | 571,987 | ||||
| Deferred tax liabilities: | ||||||||
| Accounts receivable | $ | 25,937 | $ | 12,549 | ||||
| Inventory | 10,899 | 7,730 | ||||||
| Property, plant and equipment, intangible and other non current assets | 616,430 | 522,907 | ||||||
| Accrued expenses and other liabilities | 24,582 | 32,747 | ||||||
| Other | 103,107 | 81,969 | ||||||
| Total deferred tax liabilities | 780,955 | 657,902 | ||||||
| Net deferred tax assets (liabilities) | $ | (200,847 | ) | $ | (85,915 | ) | ||
F-40
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2012 | $ | 24,916 | ||
| 2013 | 14,363 | |||
| 2014 | 22,917 | |||
| 2015 | 18,527 | |||
| 2016 | 41,705 | |||
| 2017 | 19,262 | |||
| 2018 | 17,872 | |||
| 2019 | 13,167 | |||
| 2020 | 5,049 | |||
| 2021 and thereafter | 4,746 | |||
| Without expiration date | 111,705 | |||
| Total | $ | 294,229 | ||
F-41
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | 2009 | ||||||||||
Unrecognized tax benefits (net of interest) | ||||||||||||
| Balance at January 1, 2011 | $ | 375,900 | $ | 410,016 | $ | 379,327 | ||||||
| Increases in unrecognized tax benefits prior periods | 24,046 | 12,782 | 59,833 | |||||||||
| Decreases in unrecognized tax benefits prior periods | (24,897 | ) | (11,429 | ) | (13,911 | ) | ||||||
| Increases in unrecognized tax benefits current period | 16,157 | 13,588 | 7,587 | |||||||||
| Changes related to settlements with tax authorities | (217,484 | ) | (34,410 | ) | (8,599 | ) | ||||||
| Reductions as a result of a lapse of the statute of limitations | (3,100 | ) | (129 | ) | — | |||||||
| Foreign currency translation | 14,207 | (14,518 | ) | (14,221 | ) | |||||||
| Balance at December 31, 2011 | $ | 184,829 | $ | 375,900 | $ | 410,016 | ||||||
| 19. | Operating Leases |
F-42
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2012 | $ | 510,891 | ||
| 2013 | 453,324 | |||
| 2014 | 389,469 | |||
| 2015 | 335,328 | |||
| 2016 | 278,781 | |||
| Thereafter | 739,234 | |||
| 2,707,027 | ||||
| 20. | Commitments and Contingencies |
F-43
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-44
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-45
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-46
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 21. | Financial Instruments |
F-47
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 2011 | 2010 | |||||||||||||||
| Carrying | Fair | Carrying | Fair | |||||||||||||
| Amount | Value | Amount | Value | |||||||||||||
| Non-derivatives | ||||||||||||||||
| Assets | ||||||||||||||||
| Cash and cash equivalents | $ | 457,292 | $ | 457,292 | $ | 522,870 | $ | 522,870 | ||||||||
| Accounts Receivable | 2,909,326 | 2,909,326 | 2,687,234 | 2,687,234 | ||||||||||||
| Long-term Notes Receivable | 234,490 | 233,514 | — | — | ||||||||||||
| Liabilities | ||||||||||||||||
| Accounts payable | 652,649 | 652,649 | 542,524 | 542,524 | ||||||||||||
Short-term borrowings(1) | 98,801 | 98,801 | 670,671 | 670,671 | ||||||||||||
| Short-term borrowings from related parties | 28,013 | 28,013 | 9,683 | 9,683 | ||||||||||||
Long term debt, excluding Amended 2006 Senior Credit Agreement, Euro Notes and Senior Notes(1) | 1,147,209 | 1,147,209 | 528,082 | 528,082 | ||||||||||||
| Amended 2006 Senior Credit Agreement | 2,795,589 | 2,774,951 | 2,953,890 | 2,937,504 | ||||||||||||
| Senior Notes | 2,883,009 | 2,989,307 | 824,446 | 880,366 | ||||||||||||
| Euro Notes | 258,780 | 265,655 | 267,240 | 276,756 | ||||||||||||
| Trust Preferred Securities | — | — | 625,549 | 643,828 | ||||||||||||
| Noncontrolling interests subject to put provisions | 410,491 | 410,491 | 279,709 | 279,709 | ||||||||||||
| (1) | At December 31, 2010 the A/R Facility was classified as a short-term borrowing. The A/R Facility was renewed during the third quarter of 2011 for a period of three years. As a result, the A/R Facility has been classified as long-term debt as of December 31, 2011. At December 31, 2011, there were borrowings of $534,500 under the A/R Facility. |
F-48
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
F-49
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| December 31, 2011 | December 31, 2010 | |||||||||||||||
| Assets(2) | Liabilities(2) | Assets(2) | Liabilities(2) | |||||||||||||
Derivatives in cash flow hedging relationships(1) | ||||||||||||||||
| Current | ||||||||||||||||
| Foreign exchange contracts | 4,117 | (24,908 | ) | 3,703 | (51,816 | ) | ||||||||||
| Interest rate contracts | — | (130,579 | ) | — | (51,604 | ) | ||||||||||
| Non-current | ||||||||||||||||
| Foreign exchange contracts | 742 | (3,706 | ) | 810 | (486 | ) | ||||||||||
| Interest rate contracts | — | (1,076 | ) | — | (73,221 | ) | ||||||||||
| Total | $ | 4,859 | $ | (160,269 | ) | $ | 4,513 | $ | (177,127 | ) | ||||||
Derivatives not designated as hedging instruments(1) | ||||||||||||||||
| Current | ||||||||||||||||
| Foreign exchange contracts | 56,760 | (37,242 | ) | 3,517 | (20,751 | ) | ||||||||||
| Non-current | ||||||||||||||||
| Foreign exchange contracts | 1,382 | (1,459 | ) | 509 | (213 | ) | ||||||||||
| Total | $ | 58,142 | $ | (38,701 | ) | $ | 4,026 | $ | (20,964 | ) | ||||||
| (1) | As of December 31, 2011 and December 31, 2010, the valuation of the Company’s derivatives was determined using Significant Other Observable Inputs (Level 2) in accordance with the fair value hierarchy levels established in U.S. GAAP. | |
| (2) | Derivative instruments are marked to market each reporting period resulting in carrying amounts being equal to fair values at the reporting date. |
F-50
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| Amount of (Gain) | ||||||||||||||||||
| Amount of Gain or | or Loss Reclassified | |||||||||||||||||
| (Loss) Recognized in | from AOCI in | |||||||||||||||||
| OCI on Derivatives | Income (Effective | |||||||||||||||||
| (Effective Portion) | Location of (Gain) | Portion) for the | ||||||||||||||||
| for the year ended | or Loss Reclassified | year ended | ||||||||||||||||
| Derivatives in Cash Flow | December 31, | from AOCI in Income | December 31, | |||||||||||||||
Hedging Relationships | 2011 | 2010 | (Effective Portion) | 2011 | 2010 | |||||||||||||
| Interest rate contracts | $ | (80,678 | ) | $ | (18,708 | ) | Interest income/expense | 5,946 | — | |||||||||
| Foreign exchange contracts | (23,452 | ) | 3,046 | Costs of Revenue | $ | (4,262 | ) | $ | 7,553 | |||||||||
| $ | (104,130 | ) | $ | (15,662 | ) | $ | 1,684 | $ | 7,553 | |||||||||
| Amount of (Gain) or Loss Recognized in | ||||||||||||
| Location of (Gain) or | Income on Derivatives | |||||||||||
| Derivatives not Designated as | Loss Recognized in | for the year ended December 31, | ||||||||||
Hedging Instruments | Income on Derivative | 2011 | 2010 | |||||||||
| Foreign exchange contracts | Selling, general and administrative expense | $ | (76,496 | ) | $ | 72,454 | ||||||
| Interest income/expense | 6,598 | (8,622 | ) | |||||||||
| $ | (69,898 | ) | $ | 63,832 | ||||||||
| 22. | Other Comprehensive Income (Loss) |
| Year ended December 31, 2011 | Year ended December 31, 2010 | Year ended December 31, 2009 | ||||||||||||||||||||||||||||||||||
| Tax | Tax | Tax | ||||||||||||||||||||||||||||||||||
| Pretax | Effect | Net | Pretax | Effect | Net | Pretax | Effect | Net | ||||||||||||||||||||||||||||
| Other comprehensive income (loss) relating to cash flow hedges: | ||||||||||||||||||||||||||||||||||||
| Changes in fair value of cash flow hedges during the period | (104,130 | ) | 41,825 | (62,305 | ) | (15,662 | ) | 2,241 | (13,421 | ) | 36,053 | (16,419 | ) | 19,634 | ||||||||||||||||||||||
| Reclassification adjustments | 1,684 | (796 | ) | 888 | 7,553 | (1,928 | ) | 5,625 | (5,971 | ) | 1,375 | (4,596 | ) | |||||||||||||||||||||||
| Total other comprehensive income (loss) relating to cash flow hedges: | (102,446 | ) | 41,029 | (61,417 | ) | (8,109 | ) | 313 | (7,796 | ) | 30,082 | (15,044 | ) | 15,038 | ||||||||||||||||||||||
| Foreign-currency translation adjustment | (181,234 | ) | — | (181,234 | ) | (110,888 | ) | — | (110,888 | ) | 82,545 | — | 82,545 | |||||||||||||||||||||||
| Adjustments related to pension obligations | (81,906 | ) | 31,588 | (50,318 | ) | (35,654 | ) | 12,508 | (23,146 | ) | 9,708 | (3,927 | ) | 5,781 | ||||||||||||||||||||||
| Other comprehensive income (loss) | $ | (365,586 | ) | $ | 72,617 | $ | (292,969 | ) | $ | (154,651 | ) | $ | 12,821 | $ | (141,830 | ) | $ | 122,335 | $ | (18,971 | ) | $ | 103,364 | |||||||||||||
F-51
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| 23. | Business Segment Information |
F-52
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| North | Segment | |||||||||||||||||||
| America | International | Total | Corporate | Total | ||||||||||||||||
2011 | ||||||||||||||||||||
| Net revenue external customers | $ | 8,150,017 | $ | 4,627,950 | $ | 12,777,967 | $ | 17,093 | $ | 12,795,060 | ||||||||||
| Inter-segment revenue | 9,196 | — | 9,196 | (9,196 | ) | — | ||||||||||||||
| Revenue | 8,159,213 | 4,627,950 | 12,787,163 | 7,897 | 12,795,060 | |||||||||||||||
| Depreciation and amortization | (269,055 | ) | (173,600 | ) | (442,655 | ) | (114,628 | ) | (557,283 | ) | ||||||||||
| Operating Income | 1,435,450 | 807,437 | 2,242,887 | (167,995 | ) | 2,074,892 | ||||||||||||||
| Income (loss) from equity method investees | 32,387 | 69 | 32,456 | (1,497 | ) | 30,959 | ||||||||||||||
Segment assets(1) | 11,761,777 | 5,589,421 | 17,351,198 | 2,181,652 | 19,532,850 | |||||||||||||||
| thereof investments in equity method investees | 322,990 | 370,447 | 693,437 | (1,412 | ) | 692,025 | ||||||||||||||
Capital expenditures, acquisitions and investments(2) | 1,055,183 | 1,161,825 | 2,217,008 | 166,176 | 2,383,184 | |||||||||||||||
2010 | ||||||||||||||||||||
| Net revenue external customers | $ | 8,129,737 | $ | 3,923,301 | $ | 12,053,038 | $ | 452 | $ | 12,053,490 | ||||||||||
| Inter-segment revenue | 5,419 | — | 5,419 | (5,419 | ) | — | ||||||||||||||
| Revenue | 8,135,156 | 3,923,301 | 12,058,457 | (4,967 | ) | 12,053,490 | ||||||||||||||
| Depreciation and amortization | (254,205 | ) | (148,852 | ) | (403,057 | ) | (100,167 | ) | (503,224 | ) | ||||||||||
| Operating Income | 1,385,651 | 677,630 | 2,063,281 | (139,476 | ) | 1,923,805 | ||||||||||||||
| Income (loss) from equity method investees | 8,753 | 196 | 8,949 | — | 8,949 | |||||||||||||||
| Segment assets | 11,720,495 | 4,787,479 | 16,507,974 | 586,687 | 17,094,661 | |||||||||||||||
| thereof investments in equity method investees | 243,452 | 6,921 | 250,373 | — | 250,373 | |||||||||||||||
Capital expenditures, acquisitions and investments(3) | 448,327 | 559,774 | 1,008,101 | 279,866 | 1,287,967 | |||||||||||||||
2009 | ||||||||||||||||||||
| Net revenue external customers | $ | 7,611,500 | $ | 3,635,373 | $ | 11,246,873 | $ | 604 | $ | 11,247,477 | ||||||||||
| Inter-segment revenue | 2,752 | — | 2,752 | (2,752 | ) | — | ||||||||||||||
| Revenue | 7,614,252 | 3,635,373 | 11,249,625 | (2,148 | ) | 11,247,477 | ||||||||||||||
| Depreciation and amortization | (233,094 | ) | (129,461 | ) | (362,555 | ) | (94,530 | ) | (457,085 | ) | ||||||||||
| Operating Income | 1,249,769 | 636,665 | 1,886,434 | (130,838 | ) | 1,755,596 | ||||||||||||||
| Income (loss) from equity method investees | 4,383 | 151 | 4,534 | — | 4,534 | |||||||||||||||
| Segment assets | 11,202,999 | 4,253,058 | 15,456,057 | 365,258 | 15,821,315 | |||||||||||||||
| thereof investments in equity method investees | 25,578 | 5,795 | 31,373 | — | 31,373 | |||||||||||||||
Capital expenditures, acquisitions and investments(4) | 335,857 | 264,746 | 600,603 | 161,116 | 761,719 | |||||||||||||||
| (1) | If production were still managed within the segments, as it was in 2010, segment assets would have been $12,805,094 in North America, $6,212,698 in International and $515,058 in Corporate in 2011. | |
| (2) | North America and International acquisitions exclude $6,000 and $225,034, respectively, of non-cash acquisitions and investments for 2011. | |
| (3) | North America, International and Corporate acquisitions exclude $122,847, $32,935 and $2,125, respectively, of non-cash acquisitions and investments for 2010. | |
| (4) | International acquisitions exclude $4,151 of non-cash acquisitions for 2009. |
F-53
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| North | Rest of | |||||||||||||||
| Germany | America | the World | Total | |||||||||||||
2011 | ||||||||||||||||
| Net revenue | $ | 425,507 | $ | 8,150,017 | $ | 4,219,536 | $ | 12,795,060 | ||||||||
| Long-lived assets | 417,805 | 10,318,964 | 3,010,780 | 13,747,549 | ||||||||||||
2010 | ||||||||||||||||
| Net revenue | $ | 374,883 | $ | 8,129,737 | $ | 3,548,870 | $ | 12,053,490 | ||||||||
| Long-lived assets | 471,537 | 9,236,166 | 2,139,877 | 11,847,580 | ||||||||||||
2009 | ||||||||||||||||
| Net revenue | $ | 358,060 | $ | 7,611,500 | $ | 3,277,917 | $ | 11,247,477 | ||||||||
| Long-lived assets | 350,194 | 8,864,165 | 1,809,114 | 11,023,473 | ||||||||||||
| 24. | Supplementary Cash Flow Information |
| 2011 | 2010 | 2009 | ||||||||||
| Supplementary cash flow information: | ||||||||||||
| Cash paid for interest | $ | 259,835 | $ | 264,525 | $ | 332,731 | ||||||
Cash paid for income taxes(1) | $ | 455,805 | $ | 520,766 | $ | 425,945 | ||||||
| Cash inflow for income taxes from stock option exercises | $ | 13,010 | $ | 13,313 | $ | 8,123 | ||||||
| Supplemental disclosures of cash flow information: | ||||||||||||
| Details for acquisitions: | ||||||||||||
| Assets acquired | $ | (1,684,630 | ) | $ | (668,198 | ) | $ | (241,745 | ) | |||
| Liabilities assumed | 215,253 | 102,698 | 20,574 | |||||||||
| Noncontrolling interest subject to put provisions | 26,684 | — | — | |||||||||
| Noncontrolling interest | 20,983 | 36,141 | 35,448 | |||||||||
| Notes assumed in connection with acquisition | 20,016 | 31,666 | 4,151 | |||||||||
| Cash paid | (1,401,694 | ) | (497,693 | ) | (181,572 | ) | ||||||
| Less cash acquired | 47,461 | 16,318 | 7,059 | |||||||||
| Net cash paid for acquisitions | $ | (1,354,233 | ) | $ | (481,375 | ) | $ | (174,513 | ) | |||
| (1) | Net of tax refund |
| 25. | Supplemental Condensed Combining Information |
F-54
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| For the year ended December 31, 2011 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| US Finance | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Net revenue | $ | — | $ | — | $ | 1,931,016 | $ | — | $ | 13,723,111 | $ | (2,859,067 | ) | $ | 12,795,060 | |||||||||||||
| Cost of revenue | — | — | 1,210,733 | — | 9,876,457 | (2,812,831 | ) | 8,274,359 | ||||||||||||||||||||
| Gross profit | — | — | 720,283 | — | 3,846,654 | (46,236 | ) | 4,520,701 | ||||||||||||||||||||
| Operating expenses (income): | ||||||||||||||||||||||||||||
| Selling, general and administrative | 1 | 158,222 | 208,022 | 67,587 | 1,979,854 | (78,711 | ) | 2,334,975 | ||||||||||||||||||||
| Research and development | — | — | 68,876 | — | 41,958 | — | 110,834 | |||||||||||||||||||||
| Operating (loss) income | (1 | ) | (158,222 | ) | 443,385 | (67,587 | ) | 1,824,842 | 32,475 | 2,074,892 | ||||||||||||||||||
| Other (income) expense: | ||||||||||||||||||||||||||||
| Interest, net | (5,351 | ) | 90,148 | 6,867 | 82,205 | 140,567 | (17,903 | ) | 296,533 | |||||||||||||||||||
| Other, net | — | (1,379,577 | ) | 297,281 | (724,492 | ) | — | 1,806,788 | — | |||||||||||||||||||
| Income (loss) before income taxes | 5,350 | 1,131,207 | 139,237 | 574,700 | 1,684,275 | (1,756,410 | ) | 1,778,359 | ||||||||||||||||||||
| Income tax expense (benefit) | 2,016 | 60,053 | 124,322 | (59,093 | ) | 685,166 | (211,367 | ) | 601,097 | |||||||||||||||||||
| Net Income (loss) | 3,334 | 1,071,154 | 14,915 | 633,793 | 999,109 | (1,545,043 | ) | 1,177,262 | ||||||||||||||||||||
| Net Income attributable to noncontrolling interests | — | — | — | — | — | 106,108 | 106,108 | |||||||||||||||||||||
| Net income (loss) attributable to shareholders of FMC-AG & Co. KGaA | $ | 3,334 | $ | 1,071,154 | $ | 14,915 | $ | 633,793 | $ | 999,109 | $ | (1,651,151 | ) | $ | 1,071,154 | |||||||||||||
| For the year ended December 31, 2010 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| Finance III | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Net revenue | $ | — | $ | — | $ | 1,587,720 | $ | — | $ | 12,744,881 | $ | (2,279,111 | ) | $ | 12,053,490 | |||||||||||||
| Cost of revenue | — | — | 1,022,617 | — | 9,148,969 | (2,262,817 | ) | 7,908,769 | ||||||||||||||||||||
| Gross profit | — | — | 565,103 | — | 3,595,912 | (16,294 | ) | 4,144,721 | ||||||||||||||||||||
| Operating expenses (income): | ||||||||||||||||||||||||||||
| Selling, general and administrative | 31 | 113,176 | 158,538 | 20,158 | 1,843,241 | (10,760 | ) | 2,124,384 | ||||||||||||||||||||
| Research and development | — | — | 62,435 | — | 34,097 | — | 96,532 | |||||||||||||||||||||
| Operating (loss) income | (31 | ) | (113,176 | ) | 344,130 | (20,158 | ) | 1,718,574 | (5,534 | ) | 1,923,805 | |||||||||||||||||
| Other (income) expense: | ||||||||||||||||||||||||||||
| Interest, net | (719 | ) | 39,113 | 2,388 | 56,047 | 191,638 | (8,403 | ) | 280,064 | |||||||||||||||||||
| Other, net | — | (1,200,299 | ) | 210,649 | (664,020 | ) | — | 1,653,670 | — | |||||||||||||||||||
| Income (loss) before income taxes | 688 | 1,048,010 | 131,093 | 587,815 | 1,526,936 | (1,650,801 | ) | 1,643,741 | ||||||||||||||||||||
| Income tax expense (benefit) | 196 | 69,493 | 99,957 | (30,025 | ) | 635,054 | (196,330 | ) | 578,345 | |||||||||||||||||||
| Net Income (loss) | 492 | 978,517 | 31,136 | 617,840 | 891,882 | (1,454,471 | ) | 1,065,396 | ||||||||||||||||||||
| Net Income attributable to noncontrolling interests | — | — | — | — | — | 86,879 | 86,879 | |||||||||||||||||||||
| Net income (loss) attributable to shareholders of FMC-AG & Co. KGaA | $ | 492 | $ | 978,517 | $ | 31,136 | $ | 617,840 | $ | 891,882 | $ | (1,541,350 | ) | $ | 978,517 | |||||||||||||
F-55
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| For the year ended December 31, 2009 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| Finance III | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Net revenue | $ | — | $ | — | $ | 1,521,831 | $ | — | $ | 12,041,002 | $ | (2,315,356 | ) | $ | 11,247,477 | |||||||||||||
| Cost of revenue | — | — | 997,257 | — | 8,734,160 | (2,315,452 | ) | 7,415,965 | ||||||||||||||||||||
| Gross profit | — | — | 524,574 | — | 3,306,842 | 96 | 3,831,512 | |||||||||||||||||||||
| Operating expenses (income): | ||||||||||||||||||||||||||||
| Selling, general and administrative | 28 | 87,774 | 173,215 | (19,877 | ) | 1,753,586 | (12,620 | ) | 1,982,106 | |||||||||||||||||||
| Research and development | — | — | 64,911 | — | 28,899 | — | 93,810 | |||||||||||||||||||||
| Operating (loss) income | (28 | ) | (87,774 | ) | 286,448 | 19,877 | 1,524,357 | 12,716 | 1,755,596 | |||||||||||||||||||
| Other (income) expense: | ||||||||||||||||||||||||||||
| Interest, net | (720 | ) | 35,184 | 6,070 | 56,269 | 231,559 | (28,399 | ) | 299,963 | |||||||||||||||||||
| Other, net | — | (1,032,515 | ) | 190,345 | (560,286 | ) | — | 1,402,456 | — | |||||||||||||||||||
| Income (loss) before income taxes | 692 | 909,557 | 90,033 | 523,894 | 1,292,798 | (1,361,341 | ) | 1,455,633 | ||||||||||||||||||||
| Income tax expense (benefit) | 197 | 18,419 | 86,728 | (14,338 | ) | 518,329 | (118,922 | ) | 490,413 | |||||||||||||||||||
| Net Income (loss) | 495 | 891,138 | 3,305 | 538,232 | 774,469 | (1,242,419 | ) | 965,220 | ||||||||||||||||||||
| Net Income attributable to noncontrolling interests | — | — | — | — | — | 74,082 | 74,082 | |||||||||||||||||||||
| Net income (loss) attributable to shareholders of FMC-AG & Co. KGaA | $ | 495 | $ | 891,138 | $ | 3,305 | $ | 538,232 | $ | 774,469 | $ | (1,316,501 | ) | $ | 891,138 | |||||||||||||
F-56
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| At December 31, 2011 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| US Finance | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Current assets: | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 1 | $ | 2 | $ | 144 | $ | — | $ | 457,145 | $ | — | $ | 457,292 | ||||||||||||||
| Trade accounts receivable, less allowance for doubtful accounts | — | — | 143,313 | — | 2,655,005 | — | 2,798,318 | |||||||||||||||||||||
| Accounts receivable from related parties | 1,273,649 | 3,507,671 | 1,058,327 | 700,929 | 4,214,468 | (10,644,036 | ) | 111,008 | ||||||||||||||||||||
| Inventories | — | — | 224,601 | — | 857,521 | (114,626 | ) | 967,496 | ||||||||||||||||||||
| Prepaid expenses and other current assets | — | 195,428 | 16,973 | 50 | 834,932 | (12,017 | ) | 1,035,366 | ||||||||||||||||||||
| Deferred taxes | — | 32,466 | — | — | 266,164 | 26,909 | 325,539 | |||||||||||||||||||||
| Total current assets | 1,273,650 | 3,735,567 | 1,443,358 | 700,979 | 9,285,235 | (10,743,770 | ) | 5,695,019 | ||||||||||||||||||||
| Property, plant and equipment, net | — | 356 | 175,798 | — | 2,560,913 | (107,366 | ) | 2,629,701 | ||||||||||||||||||||
| Intangible assets | — | 266 | 54,811 | — | 631,575 | — | 686,652 | |||||||||||||||||||||
| Goodwill | — | — | 53,788 | — | 9,132,862 | — | 9,186,650 | |||||||||||||||||||||
| Deferred taxes | — | 15,923 | 2,457 | — | 125,462 | (55,683 | ) | 88,159 | ||||||||||||||||||||
| Other assets | — | 8,142,771 | 653,871 | 10,995,245 | (6,082,225 | ) | (12,462,993 | ) | 1,246,669 | |||||||||||||||||||
| Total assets | $ | 1,273,650 | $ | 11,894,883 | $ | 2,384,083 | $ | 11,696,224 | $ | 15,653,822 | $ | (23,369,812 | ) | $ | 19,532,850 | |||||||||||||
| Current liabilities: | ||||||||||||||||||||||||||||
| Accounts payable | $ | — | $ | 668 | $ | 26,463 | $ | — | $ | 514,292 | $ | — | $ | 541,423 | ||||||||||||||
| Accounts payable to related parties | 3,700 | 1,547,946 | 1,057,625 | 1,557,976 | 6,697,551 | (10,753,572 | ) | 111,226 | ||||||||||||||||||||
| Accrued expenses and other current liabilities | 29,771 | 156,119 | 102,410 | 2,132 | 1,406,886 | 6,955 | 1,704,273 | |||||||||||||||||||||
| Short-term borrowings | — | 94 | — | — | 98,707 | — | 98,801 | |||||||||||||||||||||
| Short-term borrowings from related parties | — | — | — | — | (25,820 | ) | 53,833 | 28,013 | ||||||||||||||||||||
| Current portion of long-term debt and capital lease obligations | — | 295,825 | — | 1,142,224 | 151,727 | — | 1,589,776 | |||||||||||||||||||||
| Company obligated mandatorily redeemable preferred securities of subsidiary Fresenius Medical Care Capital Trusts holding solely Company-guaranteed debentures of subsidiaries-current portion | — | — | — | — | — | — | — | |||||||||||||||||||||
| Income tax payable | 2,016 | 128,218 | — | — | 32,120 | — | 162,354 | |||||||||||||||||||||
| Deferred taxes | — | — | 7,292 | — | 28,799 | (9,346 | ) | 26,745 | ||||||||||||||||||||
| Total current liabilities | 35,487 | 2,128,870 | 1,193,790 | 2,702,332 | 8,904,262 | (10,702,130 | ) | 4,262,611 | ||||||||||||||||||||
| Long term debt and capital lease obligations, less current portion | 1,177,329 | 507,898 | — | 438,366 | 7,372,794 | (4,001,577 | ) | 5,494,810 | ||||||||||||||||||||
| Long term borrowings from related parties | — | 1,348,717 | 203,156 | 408,942 | (399,065 | ) | (1,561,750 | ) | — | |||||||||||||||||||
| Other liabilities | — | 2,424 | 12,977 | 183,839 | 11,553 | 25,835 | 236,628 | |||||||||||||||||||||
| Pension liabilities | — | 5,163 | 146,555 | — | 138,775 | — | 290,493 | |||||||||||||||||||||
| Income tax payable | — | 259 | — | — | 50,309 | 138,432 | 189,000 | |||||||||||||||||||||
| Deferred taxes | — | — | — | — | 608,444 | (20,644 | ) | 587,800 | ||||||||||||||||||||
| Total liabilities | 1,212,816 | 3,993,331 | 1,556,478 | 3,733,479 | 16,687,072 | (16,121,834 | ) | 11,061,342 | ||||||||||||||||||||
| Noncontrolling interests subject to put provisions | — | — | — | — | 410,491 | — | 410,491 | |||||||||||||||||||||
| Total FMC-AG & Co. KGaA shareholders’ equity | 60,834 | 7,901,552 | 827,605 | 7,962,745 | (1,603,206 | ) | (7,247,978 | ) | 7,901,552 | |||||||||||||||||||
| Noncontrolling interests not subject to put provisions | — | — | �� | — | — | 159,465 | — | 159,465 | ||||||||||||||||||||
| Total equity | 60,834 | 7,901,552 | 827,605 | 7,962,745 | (1,443,741 | ) | (7,247,978 | ) | 8,061,017 | |||||||||||||||||||
| Total liabilities and equity | $ | 1,273,650 | $ | 11,894,883 | $ | 2,384,083 | $ | 11,696,224 | $ | 15,653,822 | $ | (23,369,812 | ) | $ | 19,532,850 | |||||||||||||
F-57
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| At December 31, 2010 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| Finance III | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Current assets: | ||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | 123 | $ | 147,177 | $ | 225 | $ | — | $ | 342,401 | $ | 32,944 | $ | 522,870 | ||||||||||||||
| Trade accounts receivable, less allowance for doubtful accounts | — | — | 157,755 | — | 2,415,503 | — | 2,573,258 | |||||||||||||||||||||
| Accounts receivable from related parties | 16,542 | 2,418,066 | 667,484 | 441,601 | 2,826,527 | (6,256,244 | ) | 113,976 | ||||||||||||||||||||
| Inventories | — | — | 184,948 | — | 711,053 | (86,904 | ) | 809,097 | ||||||||||||||||||||
| Prepaid expenses and other current assets | 1 | 111,594 | 11,341 | 50 | 662,188 | (1,943 | ) | 783,231 | ||||||||||||||||||||
| Deferred taxes | — | 14,221 | — | — | 317,644 | 18,297 | 350,162 | |||||||||||||||||||||
| Total current assets | 16,666 | 2,691,058 | 1,021,753 | 441,651 | 7,275,316 | (6,293,850 | ) | 5,152,594 | ||||||||||||||||||||
| Property, plant and equipment, net | — | 390 | 168,939 | — | 2,458,364 | (100,401 | ) | 2,527,292 | ||||||||||||||||||||
| Intangible assets | — | 428 | 65,684 | — | 626,432 | — | 692,544 | |||||||||||||||||||||
| Goodwill | — | — | 65,315 | — | 8,075,153 | — | 8,140,468 | |||||||||||||||||||||
| Deferred taxes | — | 9,463 | 4,693 | — | 121,875 | (42,863 | ) | 93,168 | ||||||||||||||||||||
| Other assets | 494,231 | 7,201,295 | 644,523 | 9,320,731 | (6,581,295 | ) | (10,590,890 | ) | 488,595 | |||||||||||||||||||
| Total assets | $ | 510,897 | $ | 9,902,634 | $ | 1,970,907 | $ | 9,762,382 | $ | 11,975,845 | $ | (17,028,004 | ) | $ | 17,094,661 | |||||||||||||
| Current liabilities: | ||||||||||||||||||||||||||||
| Accounts payable | $ | — | $ | 5,738 | $ | 22,387 | $ | — | $ | 392,512 | $ | — | $ | 420,637 | ||||||||||||||
| Accounts payable to related parties | 229 | 952,141 | 670,613 | 1,538,658 | 3,210,393 | (6,250,147 | ) | 121,887 | ||||||||||||||||||||
| Accrued expenses and other current liabilities | 15,866 | 122,000 | 94,978 | 2,054 | 1,292,562 | 9,963 | 1,537,423 | |||||||||||||||||||||
| Short-term borrowings | — | 121 | — | — | 670,550 | — | 670,671 | |||||||||||||||||||||
| Short-term borrowings from related parties | — | — | — | — | 2,004 | 7,679 | 9,683 | |||||||||||||||||||||
| Current portion of long-term debt and capital lease obligations | — | 106,862 | — | 101,145 | 55,975 | — | 263,982 | |||||||||||||||||||||
| Company obligated mandatorily redeemable preferred securities of subsidiary Fresenius Medical Care Capital Trusts holding solely Company-guaranteed debentures of subsidiaries-current portion | — | — | — | — | 625,549 | — | 625,549 | |||||||||||||||||||||
| Income tax payable | 24 | 54,366 | — | — | 62,504 | 648 | 117,542 | |||||||||||||||||||||
| Deferred taxes | — | — | 5,513 | — | 27,143 | (10,307 | ) | 22,349 | ||||||||||||||||||||
| Total current liabilities | 16,119 | 1,241,228 | 793,491 | 1,641,857 | 6,339,192 | (6,242,164 | ) | 3,789,723 | ||||||||||||||||||||
| Long term debt and capital lease obligations, less current portion | 494,231 | 870,348 | — | 1,357,745 | 4,069,605 | (2,482,253 | ) | 4,309,676 | ||||||||||||||||||||
| Long term borrowings from related parties | — | 334,428 | 208,368 | 494,231 | 400,883 | (1,437,910 | ) | — | ||||||||||||||||||||
| Other liabilities | — | 73,382 | 11,241 | — | 184,542 | 24,850 | 294,015 | |||||||||||||||||||||
| Pension liabilities | — | 4,933 | 143,362 | — | 41,855 | — | 190,150 | |||||||||||||||||||||
| Income tax payable | — | 1,057 | — | — | 75,055 | 124,469 | 200,581 | |||||||||||||||||||||
| Deferred taxes | — | — | — | — | 522,521 | (15,625 | ) | 506,896 | ||||||||||||||||||||
| Total liabilities | 510,350 | 2,525,376 | 1,156,462 | 3,493,833 | 11,633,653 | (10,028,633 | ) | 9,291,041 | ||||||||||||||||||||
| Noncontrolling interests subject to put provisions | — | — | — | — | 279,709 | — | 279,709 | |||||||||||||||||||||
| Total FMC-AG & Co. KGaA shareholders’ equity | 547 | 7,377,258 | 814,445 | 6,268,549 | (84,170 | ) | (6,999,371 | ) | 7,377,258 | |||||||||||||||||||
| Noncontrolling interests not subject to put provisions | — | — | — | — | 146,653 | — | 146,653 | |||||||||||||||||||||
| Total equity | 547 | 7,377,258 | 814,445 | 6,268,549 | 62,483 | (6,999,371 | ) | 7,523,911 | ||||||||||||||||||||
| Total liabilities and equity | $ | 510,897 | $ | 9,902,634 | $ | 1,970,907 | $ | 9,762,382 | $ | 11,975,845 | $ | (17,028,004 | ) | $ | 17,094,661 | |||||||||||||
F-58
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| For the year ended December 31, 2011 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| US Finance | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Operating Activities: | ||||||||||||||||||||||||||||
| Net income (loss) | $ | 3,334 | $ | 1,071,154 | $ | 14,915 | $ | 633,793 | $ | 999,109 | $ | (1,545,043 | ) | $ | 1,177,262 | |||||||||||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||||||||||||||||||||||
| Equity affiliate income | — | (872,048 | ) | — | (724,492 | ) | — | 1,596,540 | — | |||||||||||||||||||
| Depreciation and amortization | — | 858 | 49,207 | 5,768 | 514,843 | (13,393 | ) | 557,283 | ||||||||||||||||||||
| Change in deferred taxes, net | — | 12,593 | 2,724 | — | 138,871 | (6,734 | ) | 147,454 | ||||||||||||||||||||
| (Gain) loss on sale of fixed assets and investments | — | (10 | ) | (184 | ) | — | (8,791 | ) | — | (8,985 | ) | |||||||||||||||||
| (Gain) loss on investments | — | 31,502 | 186 | — | — | (31,688 | ) | — | ||||||||||||||||||||
| (Write Up) write-off loans from related parties | — | 44,807 | — | — | — | (44,807 | ) | — | ||||||||||||||||||||
| Compensation expense related to stock options | — | 29,071 | — | — | — | — | 29,071 | |||||||||||||||||||||
| Cash outflow from hedging | — | — | — | — | (58,113 | ) | — | (58,113 | ) | |||||||||||||||||||
| Changes in assets and liabilities, net of amounts from businesses acquired: | ||||||||||||||||||||||||||||
| Trade accounts receivable, net | — | — | (13,401 | ) | — | (239,393 | ) | — | (252,794 | ) | ||||||||||||||||||
| Inventories | — | — | (47,022 | ) | — | (135,071 | ) | 30,203 | (151,890 | ) | ||||||||||||||||||
| Prepaid expenses and other current and non-current assets | — | (133,691 | ) | (3,048 | ) | 86,497 | (99,802 | ) | (46 | ) | (150,090 | ) | ||||||||||||||||
| Accounts receivable from / payable to related parties | (12,372 | ) | (1,183,881 | ) | (51,617 | ) | 54,300 | 1,239,464 | (62,058 | ) | (16,164 | ) | ||||||||||||||||
| Accounts payable, accrued expenses and other current and non-current liabilities | 13,775 | (40,619 | ) | 28,385 | 79 | 131,427 | (641 | ) | 132,406 | |||||||||||||||||||
| Income tax payable | 2,016 | 80,461 | — | (59,093 | ) | (509 | ) | 18,167 | 41,042 | |||||||||||||||||||
| Net cash provided by (used in) operating activities | 6,753 | (959,803 | ) | (19,855 | ) | (3,148 | ) | 2,482,035 | (59,500 | ) | 1,446,482 | |||||||||||||||||
| Investing Activities: | ||||||||||||||||||||||||||||
| Purchases of property, plant and equipment | — | (221 | ) | (54,545 | ) | — | (569,645 | ) | 26,556 | (597,855 | ) | |||||||||||||||||
| Proceeds from sale of property, plant and equipment | — | — | 775 | — | 26,550 | — | 27,325 | |||||||||||||||||||||
| Disbursement of loans to related parties | — | 1,571,874 | 200 | (1,118,399 | ) | — | (453,675 | ) | — | |||||||||||||||||||
| Acquisitions and investments, net of cash acquired, and net purchases of intangible assets | — | (148,331 | ) | (4,554 | ) | — | (2,529,849 | ) | 897,405 | (1,785,329 | ) | |||||||||||||||||
| Proceeds from divestitures | — | — | 418 | — | 9,990 | (418 | ) | 9,990 | ||||||||||||||||||||
| Net cash provided by (used in) investing activities | — | 1,423,322 | (57,706 | ) | (1,118,399 | ) | (3,062,954 | ) | 469,868 | (2,345,869 | ) | |||||||||||||||||
| Financing Activities: | ||||||||||||||||||||||||||||
| Short-term borrowings, net | — | 26,284 | 77,481 | (298 | ) | (142,444 | ) | — | (38,977 | ) | ||||||||||||||||||
| Long-term debt and capital lease obligations, net | (64,252 | ) | (221,594 | ) | — | 433,455 | 1,147,586 | 453,675 | 1,748,870 | |||||||||||||||||||
| Redemption of trust preferred securities | — | — | — | — | (653,760 | ) | — | (653,760 | ) | |||||||||||||||||||
| Increase (decrease) of accounts receivable securitization program | — | — | — | — | 24,500 | — | 24,500 | |||||||||||||||||||||
| Proceeds from exercise of stock options | — | 81,883 | — | — | 13,010 | — | 94,893 | |||||||||||||||||||||
| Dividends paid | — | (280,649 | ) | — | — | 22 | (22 | ) | (280,649 | ) | ||||||||||||||||||
| Capital increase (decrease) | 57,500 | — | — | 688,390 | 151,097 | (896,987 | ) | — | ||||||||||||||||||||
| Distributions to noncontrolling interest | — | — | — | — | (129,542 | ) | — | (129,542 | ) | |||||||||||||||||||
| Contributions from noncontrolling interest | — | — | — | — | 27,824 | — | 27,824 | |||||||||||||||||||||
| Net cash provided by (used in) financing activities | (6,752 | ) | (394,076 | ) | 77,481 | 1,121,547 | 438,293 | (443,334 | ) | 793,159 | ||||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | — | (216,618 | ) | (1 | ) | — | 257,247 | 22 | 40,650 | |||||||||||||||||||
| Cash and Cash Equivalents: | ||||||||||||||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | 1 | (147,175 | ) | (81 | ) | — | 114,621 | (32,944 | ) | (65,578 | ) | |||||||||||||||||
| Cash and cash equivalents at beginning of period | — | 147,177 | 225 | — | 342,524 | 32,944 | 522,870 | |||||||||||||||||||||
| Cash and cash equivalents at end of period | $ | 1 | $ | 2 | $ | 144 | $ | — | $ | 457,145 | $ | — | $ | 457,292 | ||||||||||||||
F-59
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| For the year ended December 31, 2010 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| Finance III | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Operating Activities: | ||||||||||||||||||||||||||||
| Net income (loss) | $ | 492 | $ | 978,517 | $ | 31,136 | $ | 617,840 | $ | 891,882 | $ | (1,454,471 | ) | $ | 1,065,396 | |||||||||||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||||||||||||||||||||||
| Equity affiliate income | — | (683,735 | ) | — | (664,020 | ) | — | 1,347,755 | — | |||||||||||||||||||
| Depreciation and amortization | — | 1,452 | 47,161 | 888 | 476,647 | (22,924 | ) | 503,224 | ||||||||||||||||||||
| Change in deferred taxes, net | — | (9,645 | ) | (2,636 | ) | — | 30,710 | (3,742 | ) | 14,687 | ||||||||||||||||||
| (Gain) loss on sale of fixed assets and investments | — | (18 | ) | 155 | — | (6,653 | ) | — | (6,516 | ) | ||||||||||||||||||
| (Gain) loss on investments | — | 883 | 28 | — | 225 | (1,136 | ) | — | ||||||||||||||||||||
| Compensation expense related to stock options | — | 27,981 | — | — | — | — | 27,981 | |||||||||||||||||||||
| Changes in assets and liabilities, net of amounts from businesses acquired: | ||||||||||||||||||||||||||||
| Trade accounts receivable, net | — | — | (11,037 | ) | — | (289,237 | ) | — | (300,274 | ) | ||||||||||||||||||
| Inventories | — | — | 6,063 | — | 7,082 | 5,181 | 18,326 | |||||||||||||||||||||
| Prepaid expenses and other current and non-current assets | — | (355 | ) | 804 | 10,725 | (70,862 | ) | (617 | ) | (60,305 | ) | |||||||||||||||||
| Accounts receivable from / payable to related parties | 30 | 76,758 | 105,072 | 34,394 | (314,497 | ) | 89,204 | (9,039 | ) | |||||||||||||||||||
| Accounts payable, accrued expenses and other current and non-current liabilities | (6 | ) | 31,784 | 22,268 | 1,263 | 64,804 | 4,166 | 124,279 | ||||||||||||||||||||
| Income tax payable | (6 | ) | 24,179 | — | (30,025 | ) | (21,201 | ) | 17,419 | (9,634 | ) | |||||||||||||||||
| Net cash provided by (used in) operating activities | 510 | 447,801 | 199,014 | (28,935 | ) | 768,900 | (19,165 | ) | 1,368,125 | |||||||||||||||||||
| Investing Activities: | ||||||||||||||||||||||||||||
| Purchases of property, plant and equipment | — | (340 | ) | (31,749 | ) | — | (522,514 | ) | 30,974 | (523,629 | ) | |||||||||||||||||
| Proceeds from sale of property, plant and equipment | — | 30 | 1,099 | — | 14,979 | — | 16,108 | |||||||||||||||||||||
| Disbursement of loans to related parties | — | 227,151 | 180 | 314,665 | (327,045 | ) | (214,951 | ) | — | |||||||||||||||||||
| Acquisitions and investments, net of cash acquired, and net purchases of intangible assets | — | (273,710 | ) | (19,881 | ) | — | (614,049 | ) | 143,302 | (764,338 | ) | |||||||||||||||||
| Proceeds from divestitures | — | 132,823 | — | — | 14,245 | (233 | ) | 146,835 | ||||||||||||||||||||
| Net cash provided by (used in) investing activities | — | 85,954 | (50,351 | ) | 314,665 | (1,434,384 | ) | (40,908 | ) | (1,125,024 | ) | |||||||||||||||||
| Financing Activities: | ||||||||||||||||||||||||||||
| Short-term borrowings, net | — | — | (148,617 | ) | — | 171,078 | — | 22,461 | ||||||||||||||||||||
| Long-term debt and capital lease obligations, net | — | (146,443 | ) | — | (285,730 | ) | 91,627 | 214,951 | (125,595 | ) | ||||||||||||||||||
| Increase (decrease) of accounts receivable securitization program | — | — | — | — | 296,000 | — | 296,000 | |||||||||||||||||||||
| Proceeds from exercise of stock options | — | 96,204 | — | — | 13,314 | — | 109,518 | |||||||||||||||||||||
| Dividends paid | (495 | ) | (231,967 | ) | — | — | (6,193 | ) | 6,688 | (231,967 | ) | |||||||||||||||||
| Capital increase (decrease) | — | — | — | — | 143,069 | (143,069 | ) | — | ||||||||||||||||||||
| Distributions to noncontrolling interest | — | — | — | — | (111,550 | ) | — | (111,550 | ) | |||||||||||||||||||
| Contributions from noncontrolling interest | — | — | — | — | 26,416 | — | 26,416 | |||||||||||||||||||||
| Net cash provided by (used in) financing activities | (495 | ) | (282,206 | ) | (148,617 | ) | (285,730 | ) | 623,761 | 78,570 | (14,717 | ) | ||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | — | (104,396 | ) | (15 | ) | — | 97,624 | 48 | (6,739 | ) | ||||||||||||||||||
| Cash and Cash Equivalents: | ||||||||||||||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | 15 | 147,153 | 31 | — | 55,901 | 18,545 | 221,645 | |||||||||||||||||||||
| Cash and cash equivalents at beginning of period | 108 | 24 | 194 | — | 286,500 | 14,399 | 301,225 | |||||||||||||||||||||
| Cash and cash equivalents at end of period | $ | 123 | $ | 147,177 | $ | 225 | $ | — | $ | 342,401 | $ | 32,944 | $ | 522,870 | ||||||||||||||
F-60
Table of Contents
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS — (Continued)
(in thousands, except share data)
| For the year ended December 31, 2009 | ||||||||||||||||||||||||||||
| Issuer | Guarantors | |||||||||||||||||||||||||||
| FMC | FMC - AG & | Non-Guarantor | Combining | Combined | ||||||||||||||||||||||||
| Finance III | Co. KGaA | D-GmbH | FMCH | Subsidiaries | Adjustment | Total | ||||||||||||||||||||||
| Operating Activities: | ||||||||||||||||||||||||||||
| Net income (loss) | $ | 495 | $ | 891,138 | $ | 3,305 | $ | 538,232 | $ | 774,469 | $ | (1,242,419 | ) | $ | 965,220 | |||||||||||||
| Adjustments to reconcile net income to net cash provided by (used in) operating activities: | ||||||||||||||||||||||||||||
| Equity affiliate income | — | (635,395 | ) | — | (560,286 | ) | — | 1,195,681 | — | |||||||||||||||||||
| Depreciation and amortization | — | 1,470 | 38,029 | 888 | 439,196 | (22,498 | ) | 457,085 | ||||||||||||||||||||
| Change in deferred taxes, net | — | 23,191 | 4,707 | — | (15,491 | ) | 9,595 | 22,002 | ||||||||||||||||||||
| Loss (gain) on sale of fixed assets and investments | — | — | 411 | — | (353 | ) | — | 58 | ||||||||||||||||||||
| Loss (gain) on investments | — | 7,063 | — | — | — | (7,063 | ) | — | ||||||||||||||||||||
| (Write Up) write-off loans from related parties | — | 50 | — | — | — | (50 | ) | — | ||||||||||||||||||||
| Compensation expense related to stock options | — | 33,746 | — | — | — | — | 33,746 | |||||||||||||||||||||
| Changes in assets and liabilities, net of amounts from businesses acquired: | ||||||||||||||||||||||||||||
| Trade accounts receivable, net | — | — | (13,874 | ) | — | (28,120 | ) | — | (41,994 | ) | ||||||||||||||||||
| Inventories | — | — | (27,435 | ) | — | (49,213 | ) | (12,285 | ) | (88,933 | ) | |||||||||||||||||
| Prepaid expenses and other current and non-current assets | — | (37,138 | ) | 9,921 | (18,344 | ) | (93,440 | ) | (8,104 | ) | (147,105 | ) | ||||||||||||||||
| Accounts receivable from / payable to related parties | 208 | (388,546 | ) | 7,308 | 39,091 | 256,906 | 79,315 | (5,718 | ) | |||||||||||||||||||
| Accounts payable, accrued expenses and other current and non-current liabilities | (15 | ) | 16,210 | 12,731 | (1,149 | ) | 38,065 | 5,250 | 71,092 | |||||||||||||||||||
| Income tax payable | (160 | ) | (23,961 | ) | — | (14,338 | ) | 71,931 | 39,692 | 73,164 | ||||||||||||||||||
| Net cash provided by (used in) operating activities | 528 | (112,172 | ) | 35,103 | (15,906 | ) | 1,393,950 | 37,114 | 1,338,617 | |||||||||||||||||||
| Investing Activities: | ||||||||||||||||||||||||||||
| Purchases of property, plant and equipment | — | (152 | ) | (65,684 | ) | — | (537,167 | ) | 29,397 | (573,606 | ) | |||||||||||||||||
| Proceeds from sale of property, plant and equipment | — | — | 731 | — | 10,999 | — | 11,730 | |||||||||||||||||||||
| Disbursement of loans to related parties | — | (7,270 | ) | 178 | 17,240 | — | (10,148 | ) | — | |||||||||||||||||||
| Acquisitions and investments, net of cash acquired, and net purchases of intangible assets | — | (11,841 | ) | (1,900 | ) | — | (185,878 | ) | 11,506 | (188,113 | ) | |||||||||||||||||
| Proceeds from divestitures | — | 13,380 | — | — | 1,965 | 36,620 | 51,965 | |||||||||||||||||||||
| Net cash provided by (used in) investing activities | — | (5,883 | ) | (66,675 | ) | 17,240 | (710,081 | ) | 67,375 | (698,024 | ) | |||||||||||||||||
| Financing Activities: | ||||||||||||||||||||||||||||
| Short-term borrowings, net | — | (95,795 | ) | 31,716 | — | 10,943 | (108,439 | ) | (161,575 | ) | ||||||||||||||||||
| Long-term debt and capital lease obligations, net | — | 396,013 | — | (1,334 | ) | (261,528 | ) | 10,148 | 143,299 | |||||||||||||||||||
| Increase (decrease) of accounts receivable securitization program | — | — | — | — | (325,000 | ) | — | (325,000 | ) | |||||||||||||||||||
| Proceeds from exercise of stock options | — | 64,271 | — | — | 8,123 | — | 72,394 | |||||||||||||||||||||
| Dividends paid | (443 | ) | (231,940 | ) | — | — | (5,321 | ) | 5,764 | (231,940 | ) | |||||||||||||||||
| Capital increase (decrease) | — | — | — | — | (1,874 | ) | 1,874 | — | ||||||||||||||||||||
| Distributions to noncontrolling interest | — | — | — | — | (68,004 | ) | — | (68,004 | ) | |||||||||||||||||||
| Contributions from noncontrolling interest | — | — | — | — | 12,699 | — | 12,699 | |||||||||||||||||||||
| Net cash provided by (used in) financing activities | (443 | ) | 132,549 | 31,716 | (1,334 | ) | (629,962 | ) | (90,653 | ) | (558,127 | ) | ||||||||||||||||
| Effect of exchange rate changes on cash and cash equivalents | — | (14,470 | ) | 6 | — | 11,590 | 49 | (2,825 | ) | |||||||||||||||||||
| Cash and Cash Equivalents: | ||||||||||||||||||||||||||||
| Net increase (decrease) in cash and cash equivalents | 85 | 24 | 150 | — | 65,497 | 13,885 | 79,641 | |||||||||||||||||||||
| Cash and cash equivalents at beginning of period | 23 | — | 44 | — | 221,003 | 514 | 221,584 | |||||||||||||||||||||
| Cash and cash equivalents at end of period | $ | 108 | $ | 24 | $ | 194 | $ | — | $ | 286,500 | $ | 14,399 | $ | 301,225 | ||||||||||||||
F-61
Table of Contents
| 2011 | 2010 | 2009 | ||||||||||
| Allowance for doubtful accounts as of January 1 | $ | 277,139 | $ | 266,449 | $ | 262,836 | ||||||
| Change in valuation allowances as recorded in the consolidated statements of income | 241,598 | 218,496 | 210,124 | |||||||||
| Write-offs and recoveries of amounts previously written-off | (214,612 | ) | (205,666 | ) | (210,166 | ) | ||||||
| Foreign currency translation | (4,374 | ) | (2,140 | ) | 3,656 | |||||||
| Allowance for doubtful accounts as of December 31 | $ | 299,751 | $ | 277,139 | $ | 266,449 | ||||||
S-II