 Exhibit 99.1 |
 NASDAQ: CHTP 2 Forward-looking Statement This presentation is being provided for informational and discussion purposes. This presentation is not intended to provide and should not be relied upon as investment advice or an opinion regarding the appropriateness or suitability of any investment. Nothing herein should be construed to be an offer to sell, or a solicitation of an offer to buy, any securities. This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include failure to get regulatory approval for our product candidates, market acceptance for approved products, management of rapid growth, risks of regulatory review and clinical trials, intellectual property risks, and the need to acquire additional products. The reader is referred to the documents that Chelsea Therapeutics International, Ltd. files from time to time with the Securities and Exchange Commission. |
 NASDAQ: CHTP 3 Overview Two platform technologies addressing multiple, high-potential global markets • Library of metabolically inert antifolate compounds with potent anti- inflammatory, anti-proliferative and immunological activity for RA, psoriasis, IBD and certain cancers • Co-development of I-3D portfolio of DHODH inhibiting compounds for autoimmune disease and transplant rejection with Active Biotech AB Accelerating revenue opportunities through expedited development of orphan drug candidate • Pursuing US/EU Orphan strategy for Droxidopa - currently approved and marketed in Japan by Dainippon Sumitomo Pharma (DSP) for orthostatic hypotension Experienced management team with proven development expertise and extensive industry network Corporate Overview |
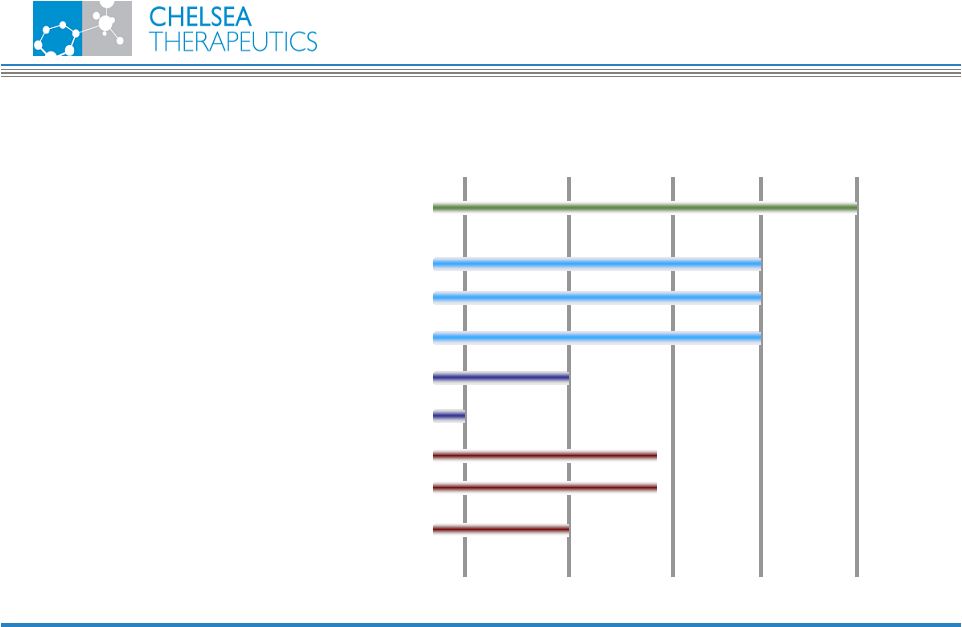 NASDAQ: CHTP 4 INDICATION RESEARCH PRECLINICAL PHASE I PHASE II PHASE III Droxidopa Neurogenic Orthostatic Hypotension CH-1504 Rheumatoid Arthritis Psoriasis Inflammatory Bowel Disease CH-4446 Other Inflammatory Diseases CH-2308 Oncology I-3D Rheumatoid Arthritis Transplant Rejection Other Product Pipeline Corporate Overview |
 NASDAQ: CHTP 5 Droxidopa Droxidopa • Late stage orphan drug candidate for the treatment of neurogenic orthostatic hypotension • Acquired 15+ years of proven safety and efficacy data from Dainippon Sumitomo Pharma to expedite clinical development • Including European Phase II • Approved and marketed in , generating $50 million in annual sales • Significant sales achieved with low price • Accelerates revenue opportunity and establishes commercial infrastructure Japan |
 NASDAQ: CHTP 6 Treating OH Droxidopa Orthostatic Hypotension (OH) • Sudden fall in BP when standing from a sitting/lying position • Multiple pathologies • Cardiovascular, Endocrine and Neurological • Neurogenic OH due to loss of norepinephrine in the peripheral nervous system Present treatment does not address neurogenic OH • Midodrine (ProAmatine ® ) • Increases blood pressure, vasoconstriction, pupil dilation • Side effects include hypertension, piloerection, • US Annual Sales approximately $60 million • Shire, Mylan Pharmaceuticals, Eon Labs, Impax Laboratories • Fludrocortisone (Florinef ® ) • Retains Na + in blood stream • Simply adds volume • Not FDA approved for this indication • Side effects include hypertension, water & sodium retention, K + loss |
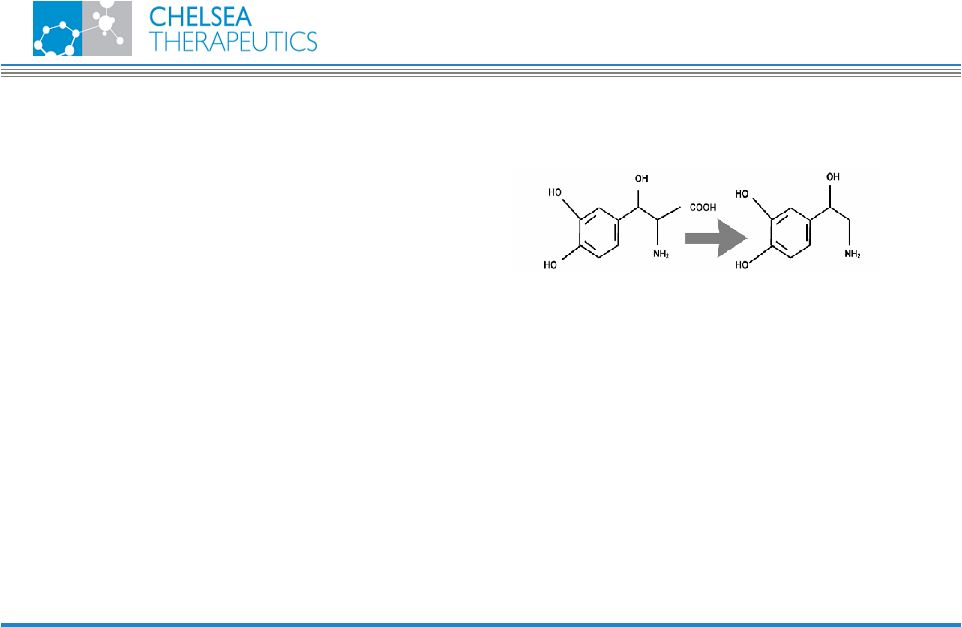 NASDAQ: CHTP 7 Droxidopa Droxidopa • Directly decarboxylated by dopa decarboxylase to form NE • Allows for re-uptake of NE into PNS neurons • Stored NE available for release upon standing • Released NE stimulate receptors for vasoconstriction • Homeostasis mechanisms terminate NE activity • Metabolism • Neuronal uptake • Provides physiological improvement in neurogenic OH patients Droxidopa Norepinephrine • Synthetic amino acid “prodrug” of norepinephrine (NE) • Replenishes depleted NE by endogenous enzymatic pathway |
 NASDAQ: CHTP 8 Orphan Strategy Droxidopa • Estimated 120,000 patients suffer from chronic, symptomatic neurogenic orthostatic hypotension ~120,000 Total Neurogenic OH 1,000 – 10,000 Multiple System Atrophy (MSA) ~15,000 Pure Autonomic Failure (PAF) ~100,000 Parkinson’s Disease + Lewy Body Dementia Estimated Addressable Patient Population: Indications Associated with Chronic, Symptomatic OH: • Provides 7 years marketing exclusivity in U.S, 10 years in E.U. •Clock starts at commercialization • Chronic, daily oral dosing |
 NASDAQ: CHTP 9 Development Program Droxidopa • Filed Orphan Drug Application with FDA in Q3 2006 • Plan filing with EMEA Authorities Q4 2006 • Prepare clinical material with Dainippon Sumitomo Pharma • Plan Phase III pivotal study with commercial material • Initiate Pivotal Phase III trials in 2007 • Straight forward clinical end-points •1 minute standing BP measures • Plan QOL and Pharmacoeconomics data for price justification • Retain US rights for marketing and sales • Potential out-licensing of European commercial rights |
 NASDAQ: CHTP 10 Partnering with Active Biotech Strategic partnership with Active Biotech to co-develop and commercialize I-3D portfolio • Orally active small molecules that inhibit dihydroorotate dehydrogenase (DHODH) and thereby DNA synthesis in T-lymphocytes • Examples: Leflunomide (Arava®) – RA; Teriflunomide – MS Phase III FK-778 – Transplant rejection Phase II • Two potential lead compounds • Expected Phase I trials early 2007 • Potential indications: RA, Transplant rejection, Psoriasis, Lupus Exclusive North and South American commercial rights to all compounds • Split costs of development • Equal partnership model I-3D Portfolio |
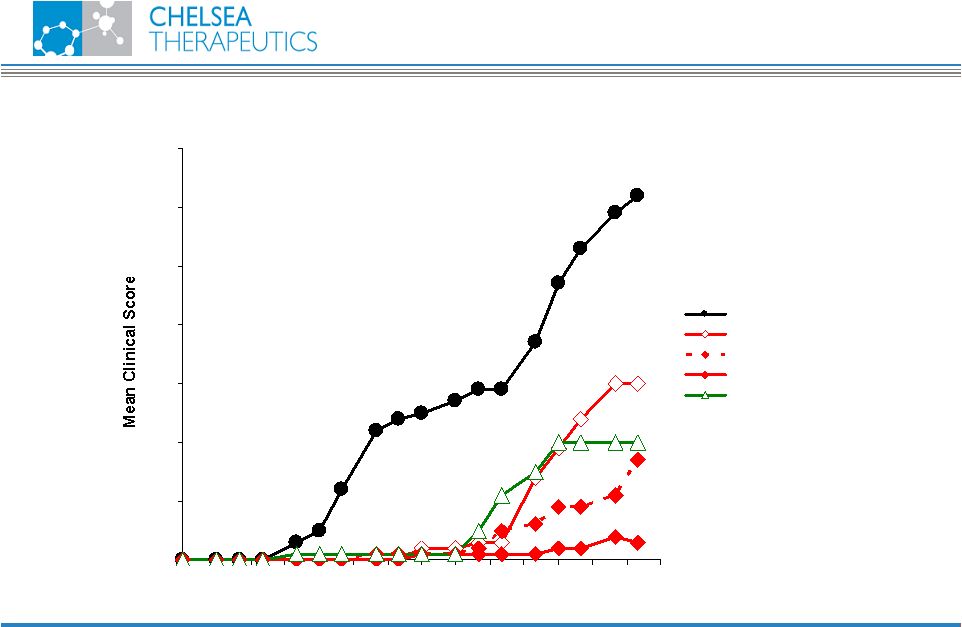 NASDAQ: CHTP 11 Rheumatoid Arthritis I-3D Portfolio 0.0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 14 17 20 23 26 29 32 35 38 41 44 47 50 53 56 Days after Immunization Control (metocel) ABR-224050 3 mg/kg ABR-224050 10 mg/kg ABR-224050 30 mg/kg ABR-214658 10 mg/kg |
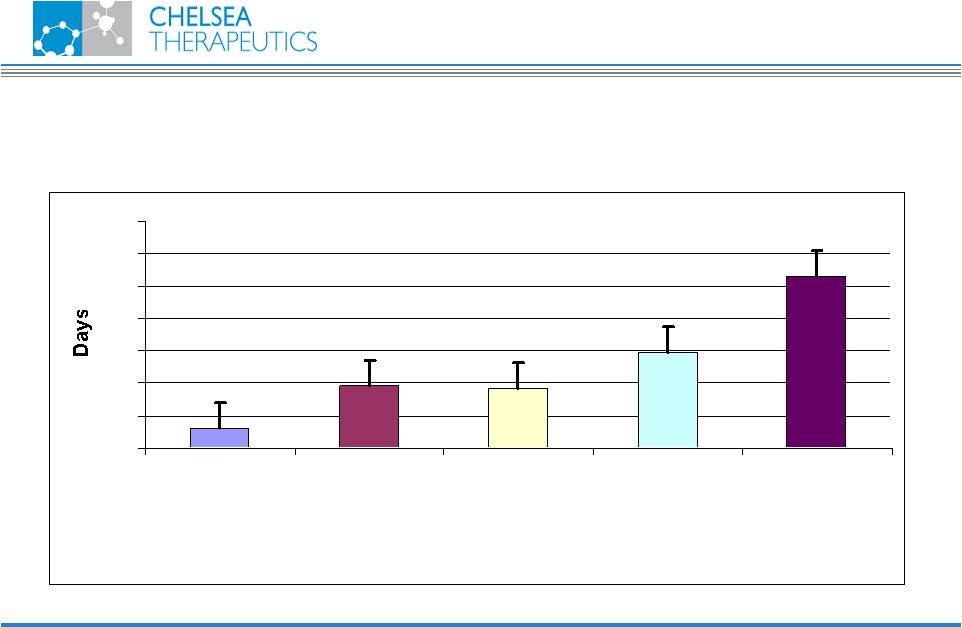 NASDAQ: CHTP 12 Transplant Rejection I-3D Portfolio In vivo efficacy in DA to PVG high responder transplant rejection model 0 10 20 30 40 50 60 70 Control (Methocel) Cyclosporin A (10 mg/kg) ABR-224050 (10 mg/kg) ABR-222417 (30 mg/kg) ABR-222417 (10 mg/kg) + Cyclosporin A (5 mg/kg) |
 NASDAQ: CHTP 13 Lead Antifolate Candidate CH-1504 • Metabolically inert antifolate developed by Dr. Gopal Nair, PhD. in 1999 • Preclinical studies support potential superior efficacy vs. Methotrexate (MTX) • Human data show superior tolerability vs. MTX • MTX remains most widely prescribed RA treatment • Used in combination with most biologic treatments • Global sales approx. $400m CH-1504 |
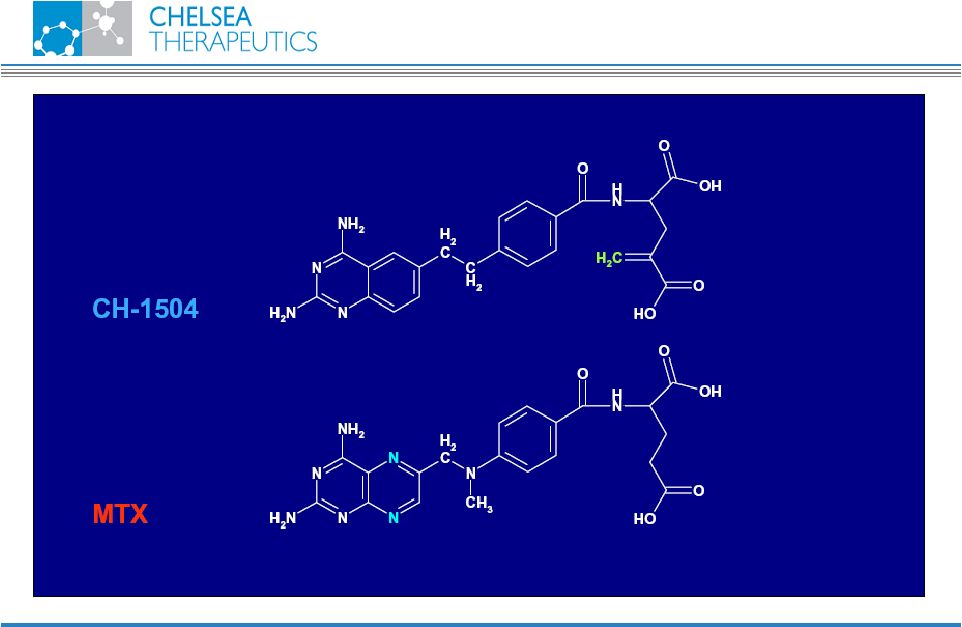 NASDAQ: CHTP 14 Chemistry CH-1504 |
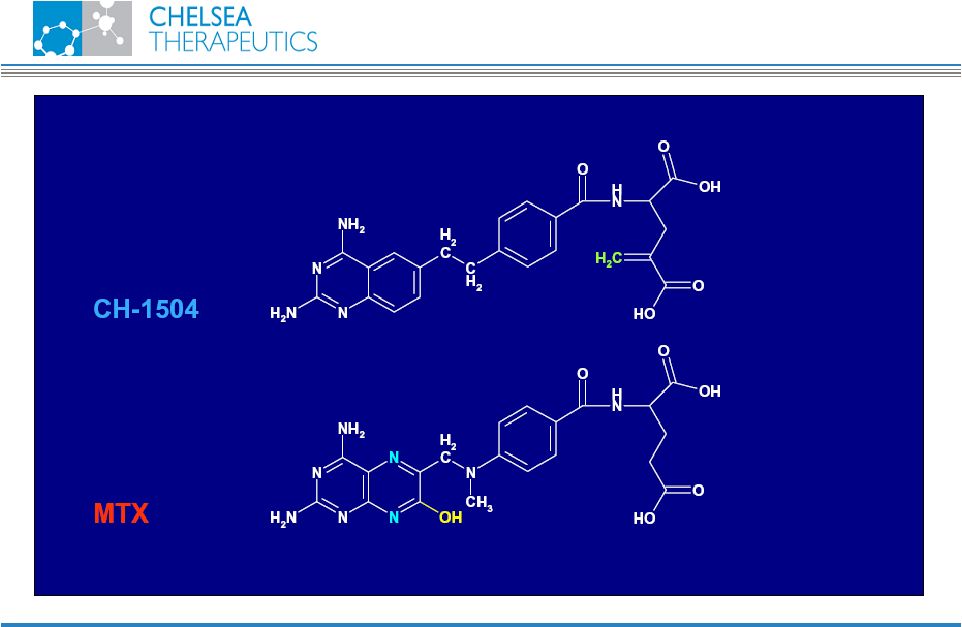 NASDAQ: CHTP 15 Chemistry CH-1504 |
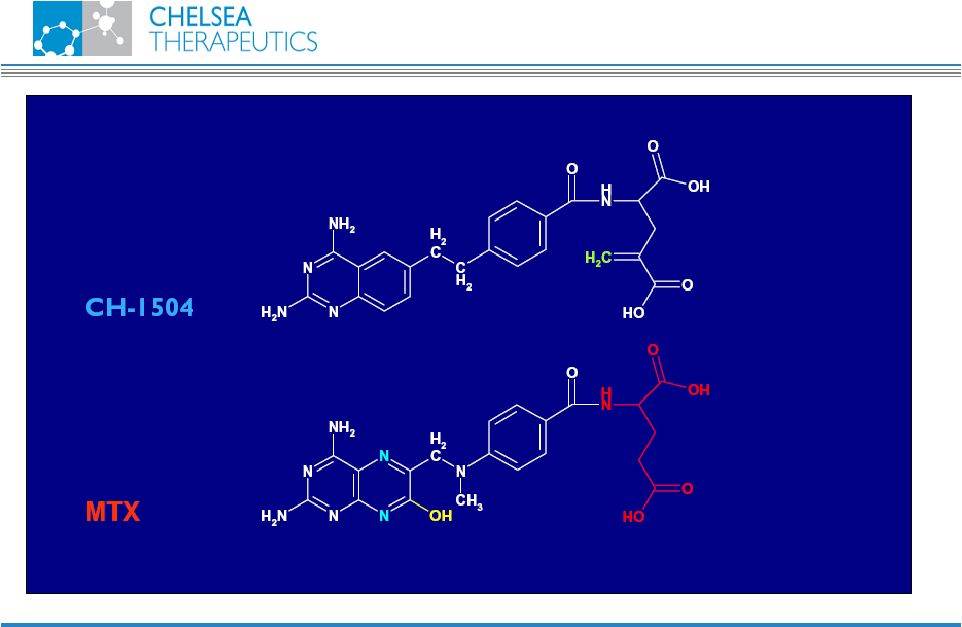 NASDAQ: CHTP 16 Chemistry CH-1504 |
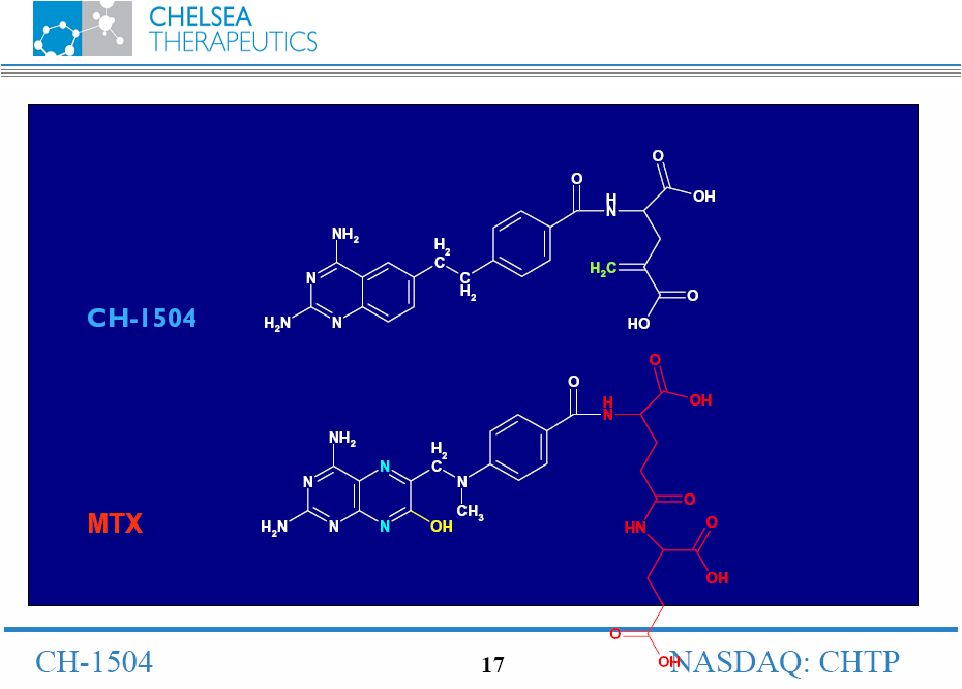 Chemistry |
 NASDAQ: CHTP 18 Preliminary Proof-of-Concept Six-month pilot clinical study - Lima, Peru 2002 • CH-1504 vs. MTX • Open-label, non-randomized, controlled trial design • 20 Chronic refractory RA patients • Patient response evaluated using ACR criteria Swollen joints Tender joints Pain Health Assessment Questionnaire (HAQ) Patient Global Assessment Physician Global Assessment • ACR-accepted PD markers of inflammation Erythrocyte Sedimentation Rate (ESR) C-Reactive Protein (CRP) CH-1504 |
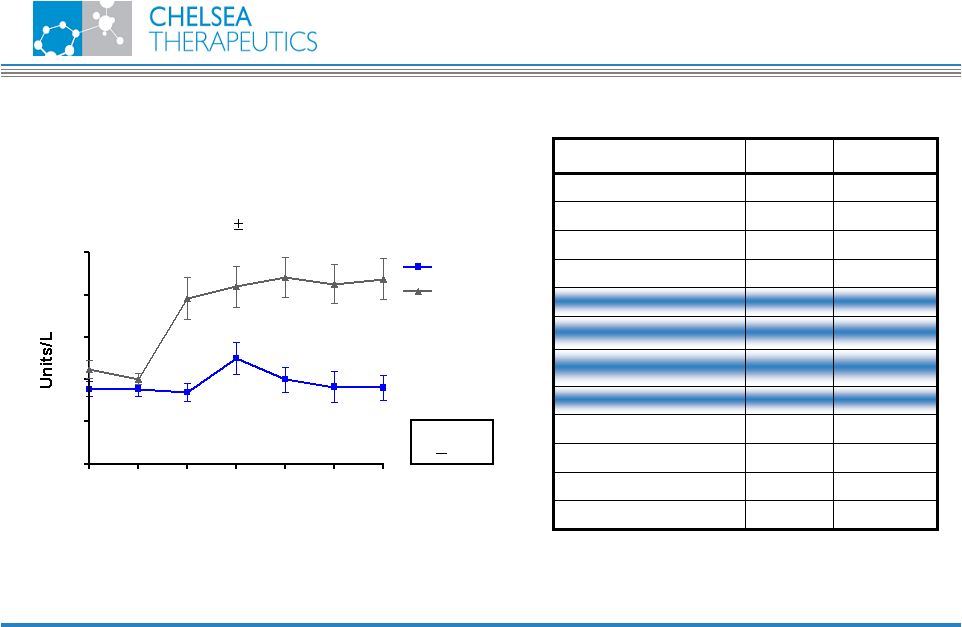 NASDAQ: CHTP 19 SERUM ALT LEVELS (Mean SEM) 0 4 8 12 16 20 24 0 10 20 30 40 50 CH-1504 MTX Time (weeks) Pilot Study Results (N = # abnormal values) No elevation in liver enzymes (3) (2) (2) (3) (3) p < 0.05 CH-1504 10 % 30 % Infections 10 % 10 % Alopecia 10 % 0 % Acne 0 % 0 % Allergic 0 % 20 % Diarrhea 0 % 10 % Abdominal Pain 0 % 20 % Dyspepsia 0 % 20 % Nausea/Vomiting 20 % 0 % Somnolence 10 % 10 % Dizziness 0 % 0 % Withdrawals 0 % 0 % SAE CH-1504 MTX ADVERSE EFFECT |
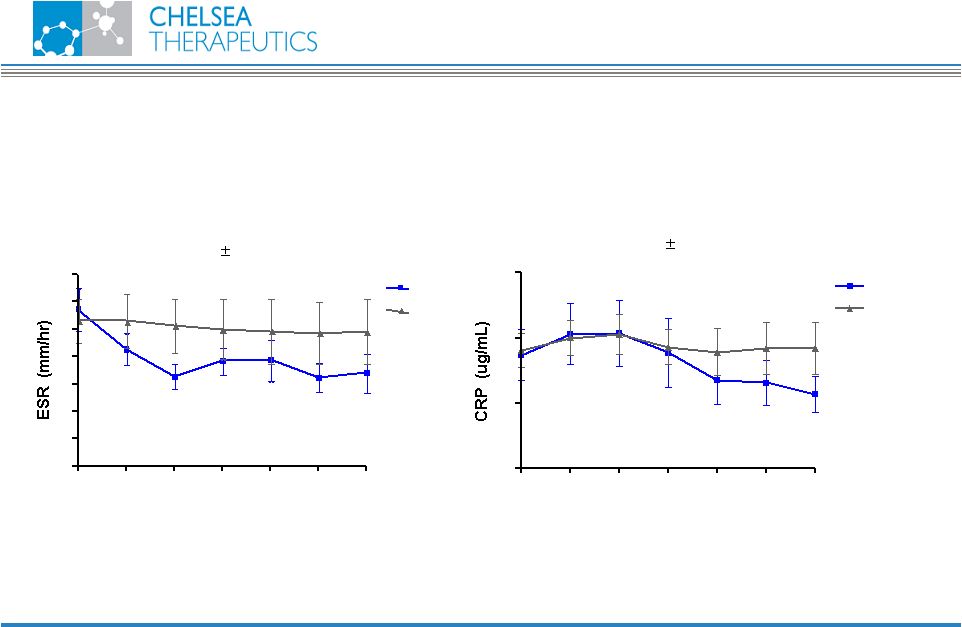 NASDAQ: CHTP 20 Pilot Study Results Normalization of ESR and CRP CH-1504 Erythrocyte Sedimentation Rate (Mean SEM) 0 4 8 12 16 20 24 0 10 20 30 40 50 60 70 CH-1504 MTX Time (weeks) C-Reactive Protein (Mean SEM) 0 4 8 12 16 20 24 0 10 20 30 CH-1504 MTX Time (weeks) |
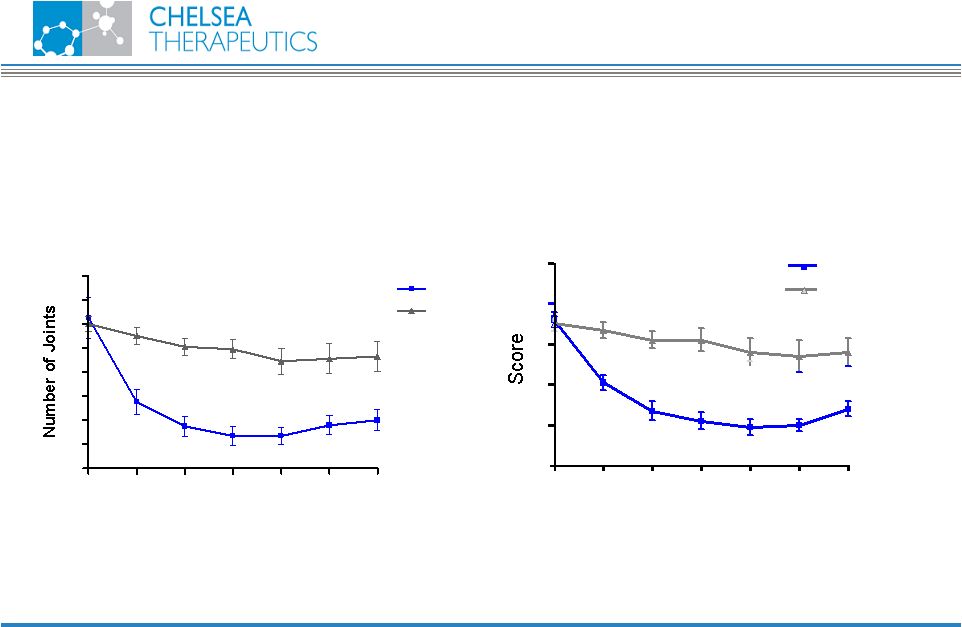 NASDAQ: CHTP 21 Pilot Study Results Better tolerated with improved efficacy Pain (day) (Mean ± SEM) 0 4 8 12 16 20 24 0 2 4 6 8 10 CH-1504 MTX Time (weeks) CH-1504 Swollen Joints (Mean ± SEM) 0 4 8 12 16 20 24 0 2 4 6 8 10 12 14 16 CH-1504 MTX Time (weeks) |
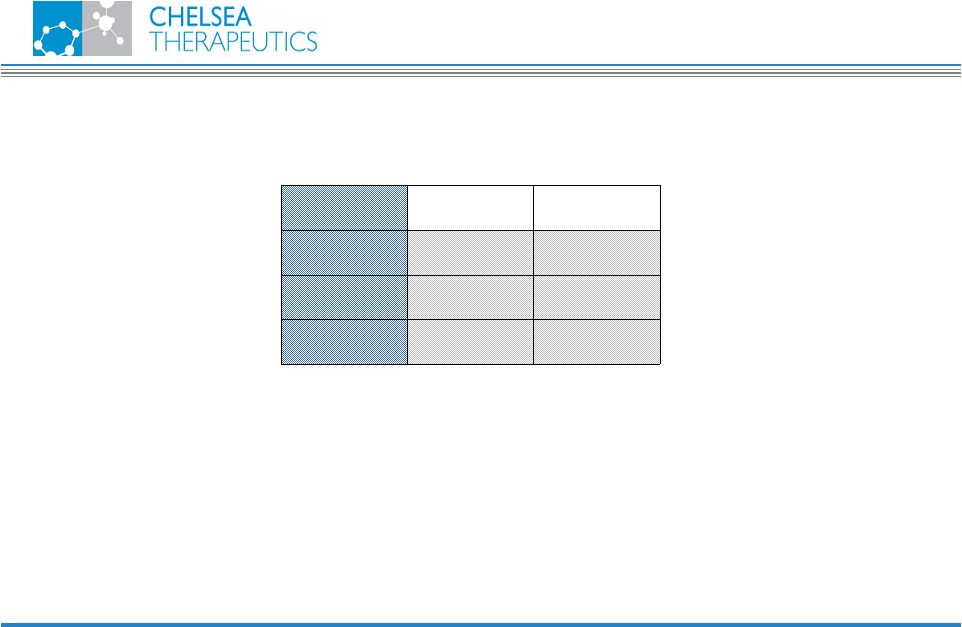 NASDAQ: CHTP 22 ACR Summary Possible earlier onset of action • Week 4: • CH-1504 ACR20: 60% • MTX ACR20: 10% 10% 60% ACR NR 90% 40% ACR 20 40% 10% ACR 50 CH-1504 MTX Week 24 Pilot Study Results CH-1504 |
 NASDAQ: CHTP 23 Phase I Conclusions • Safe and well tolerated at all dose levels • No clinically significant abnormalities • Steady state reached between day 2 and 3 • Maximum plasma concentrations occur 1-2 hours post dose • No accumulation of parent compound found in plasma samples at 24 h post dose • Suitable for a daily dosing regimen • All I-b dosages were above those that are predicted to be therapeutically effective CH-1504 |
 NASDAQ: CHTP 24 Development Program Optimizing formulation by converting from free acid to salt formulation • Initial formulation led to variation in PK • Early data suggest formulation as a Salt reduces PK variation • Time table for reformulation Selected salt from approximately 25 commercially viable salts Complimentary pharmaceutical enhancements ongoing • Animal toxicology studies, 2H 06 • Bridging toxicology, 2H 06 • Bioequivalence trial, 1H 07 • Maximum tolerated dose trial, 1H 07 • Global Phase II/III, 2H 07 CH-1504 |
 NASDAQ: CHTP 25 Financial Highlights Funding Detail • $14.5M Series A financing priced at $2.62 - December 2004 • $21.5M Private Placement priced at $3.00 - February 2006 Capitalization • Stock • Common Stock Outstanding 19,707,129 • Warrants 3,675,440 • Options 1,591,342 • Fully Diluted Common Stock 24,973,911 • Cash on Hand $18,954,970 Corporate Overview (as of 6/30/06) |
 NASDAQ: CHTP 26 Going Forward Publish Results of CH-1504 Pilot Clinical Study May 06 Journal of Rheumatology 2006, 33:862-4 Present CH-1504 Phase I Data EULAR 2006 June 06 Submit Droxidopa Orphan Drug Application U.S. FDA Sept 06 Select I-3D Lead Compound for IND Enabling Tox. Q3 06 Submit Droxidopa Orphan Drug Application EMEA Q4 06 Initiate I-3D Clinical Trials 1H 07 Conduct CH-1504 Bioequivalence Studies 1H 07 Begin Phase II in RA 2H 07 Publish and Present Droxidopa European Phase II Data 2007 Initiate Droxidopa PIII Clinical trials 2007 Conclusion |