 © 2004-2010 Chelsea Therapeutics, Inc. Northera™ (droxidopa) Preliminary Findings From Study 301 in Symptomatic Neurogenic Orthostatic Hypotension September 2010 |
 NASDAQ: CHTP 2 Forward-Looking Statement Forward-Looking Statement This presentation is being provided for informational and discussion purposes. This presentation is not intended to provide and should not be relied upon as investment advice or an opinion regarding the appropriateness or suitability of any investment. Nothing herein should be construed to be an offer to sell, or a solicitation of an offer to buy, any securities. This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include failure to get regulatory approval for our product candidates, market acceptance for approved products, management of rapid growth, risks of regulatory review and clinical trials, intellectual property risks, and the need to acquire additional products. The reader is referred to the documents that Chelsea Therapeutics International, Ltd. files from time to time with the Securities and Exchange Commission. |
 NASDAQ: CHTP 3 Northera Phase III Program: NOH Northera Phase III Program: NOH Study 302: Pivotal Proof of Efficacy – Withdrawal Design Study 303: Long-Term Safety Extension to Study 302 – includes long-term efficacy assessment after 3 months Study 301: Pivotal Proof of Efficacy – Induction Design Study 304: Long-Term Safety Extension to Study 301 Study 305: 24 HR Blood Pressure Monitoring Study – subgroup of 301 patients Study 306: Pivotal Proof of Efficacy – Induction Design |
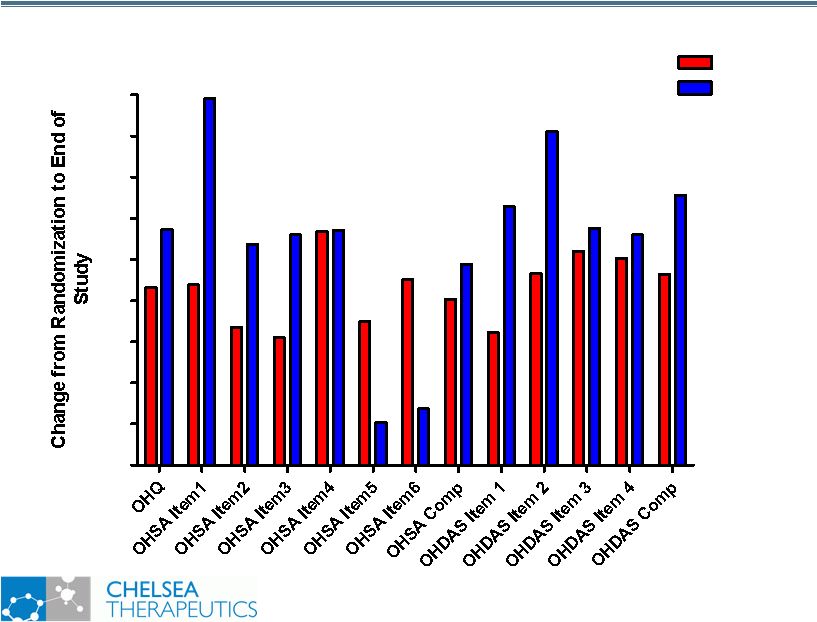
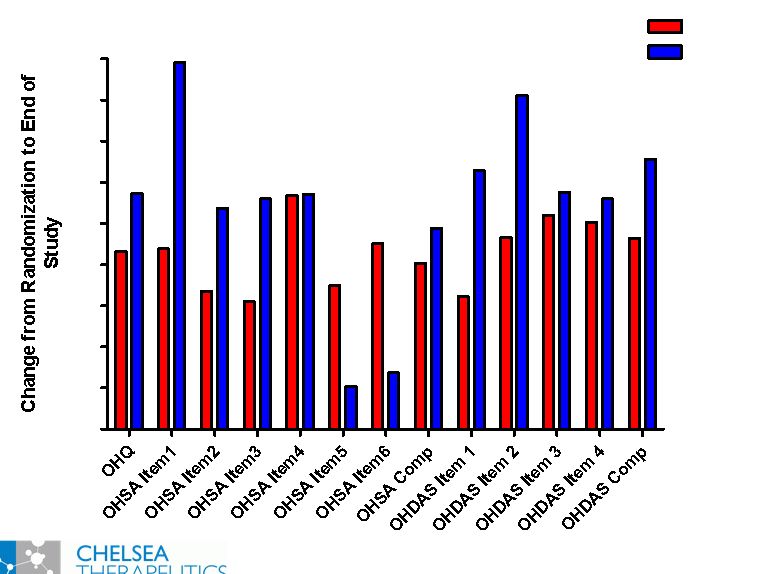
 NASDAQ: CHTP 4 Key Symptomatic Efficacy Outcome Measures Key Symptomatic Efficacy Outcome Measures Orthostatic Hypotension Questionnaire: OHQ • Orthostatic Hypotension Symptom Assessment: OHSA Please circle the number on the scale (0-10) that best rates how severe your symptoms from low blood pressure have been on average over the past week Please respond to every symptom. If you do not experience the symptom, circle zero (0). PLEASE RATE THE SYMPTOMS THAT ARE DUE ONLY TO YOUR LOW BLOOD PRESSURE PROBLEM. 1.Dizziness, lightheadedness, feeling faint, or feeling like you might blackout 2.Problems with vision (blurring, seeing spots, tunnel vision, etc.) 3.Weakness 4.Fatigue 5.Trouble concentrating 6.Head/neck discomfort • Orthostatic Hypotension Daily Activities Scale: OHDAS We are interested in how the low blood pressure symptoms you experience affect your daily life. Please rate each item by circling the number that best represents how much the activity has been interfered with on average over the past week by the low blood pressure symptoms you experienced. 1. Standing short time 2. Standing long time 3. Walking short time 4. Walking long time |
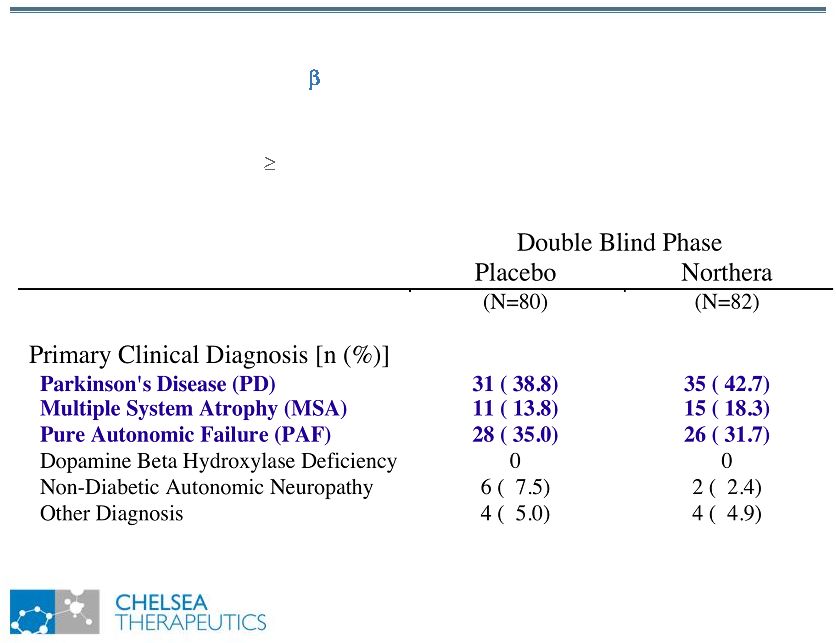 NASDAQ: CHTP 5 Study 301: Baseline Diagnoses Study 301: Baseline Diagnoses Symptomatic, Neurogenic Orthostatic Hypotension Associated with Primary Autonomic Failure, Dopamine -hydroxylase Deficiency & Non-diabetic Autonomic Neuropathies • Diverse patient population with varied & distinct primary diseases • Common feature: 20 mm Hg drop in SBP upon Standing |
 NASDAQ: CHTP 6 Study 301: Demographic Data Study 301: Demographic Data Double Blind Phase Placebo Northera (N=80) (N=82) Gender [N(%)] Male 42 ( 52.5) 42 ( 51.2) Female 38 ( 47.5) 40 ( 48.8) Ethnicity White 75 ( 93.8) 82 (100.0) Black/African-American 1 ( 1.3) 0 Asian 1 ( 1.3) 0 Hispanic/Latino 3 ( 3.8) 0 Age (years) at Screening n 80 82 Mean (SD) 55.7 (20) 57.4 (17) Median 61.5 60.0 |
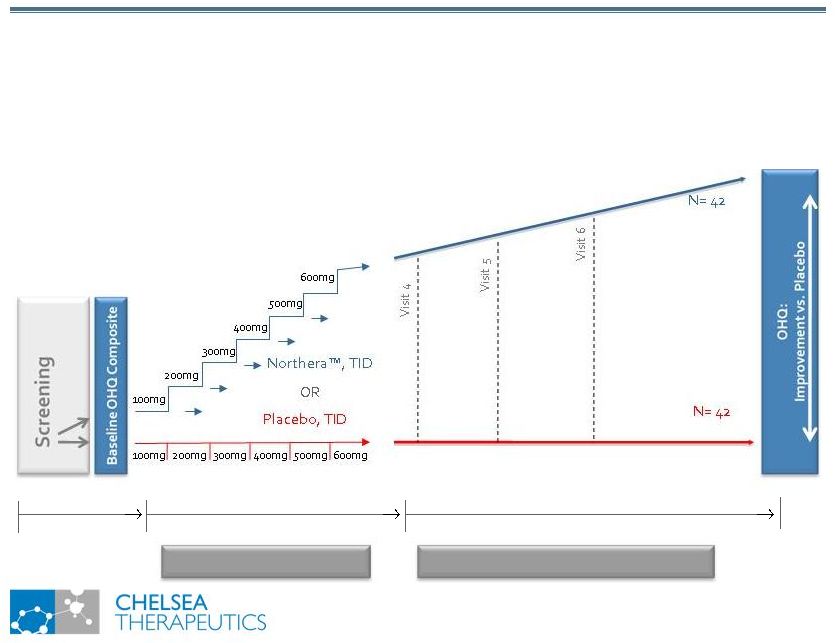
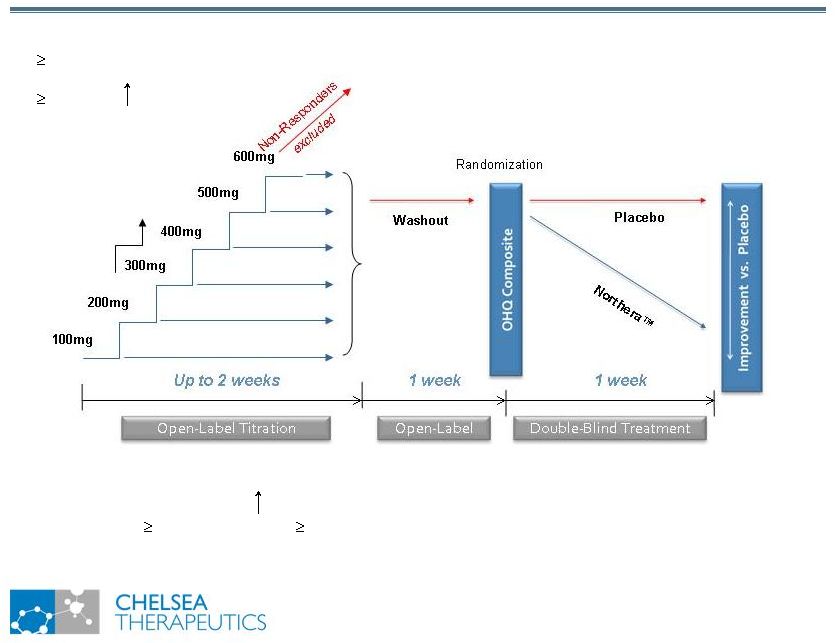 NASDAQ: CHTP 7 Study 301: Trial Design Study 301: Trial Design Titration Stopping Rules: 1.Asymptomatic and >10 mmHG in Standing SBP 2.Sustained SBP 180 mmHg or DBP 110 mmHg 3.Intolerable AEs 4.Maximum Dose of 600mg TID reached Responder Criteria: 1. 1 unit improvement on OHSA item #1 (dizziness); AND 2. 10 mmHg in Standing SBP |
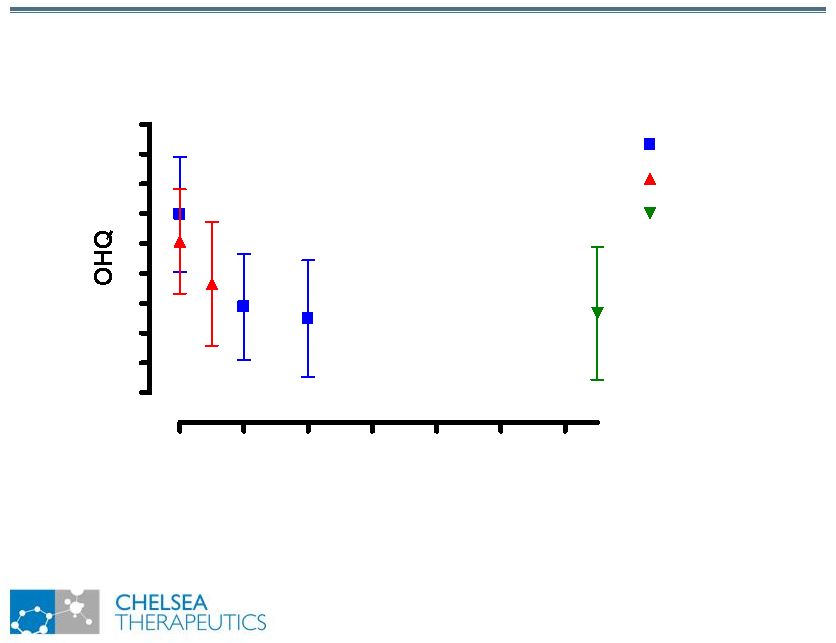
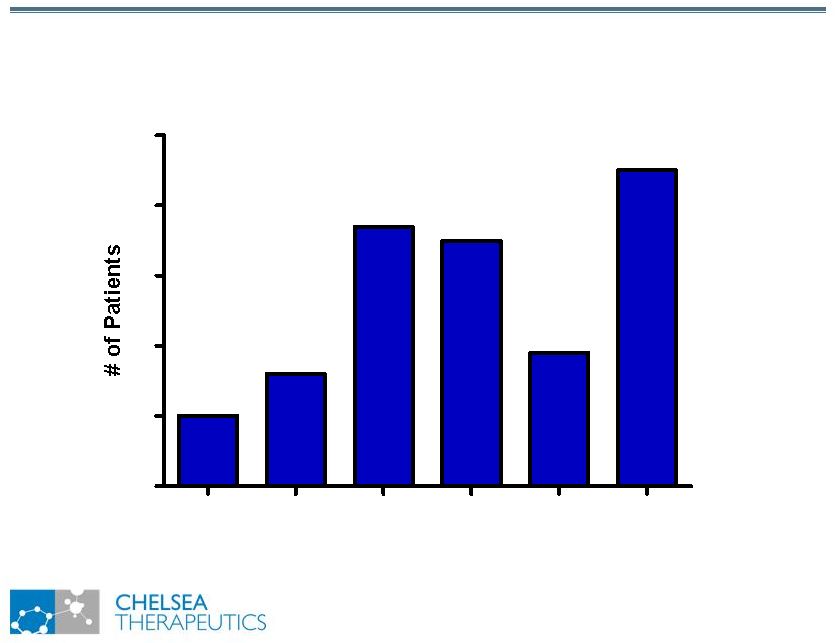 NASDAQ: CHTP 8 Study 301: Optimal Dose Post-Titration Study 301: Optimal Dose Post-Titration 100 200 300 400 500 600 0 10 20 30 40 50 Dose of Northera (mg TID) |
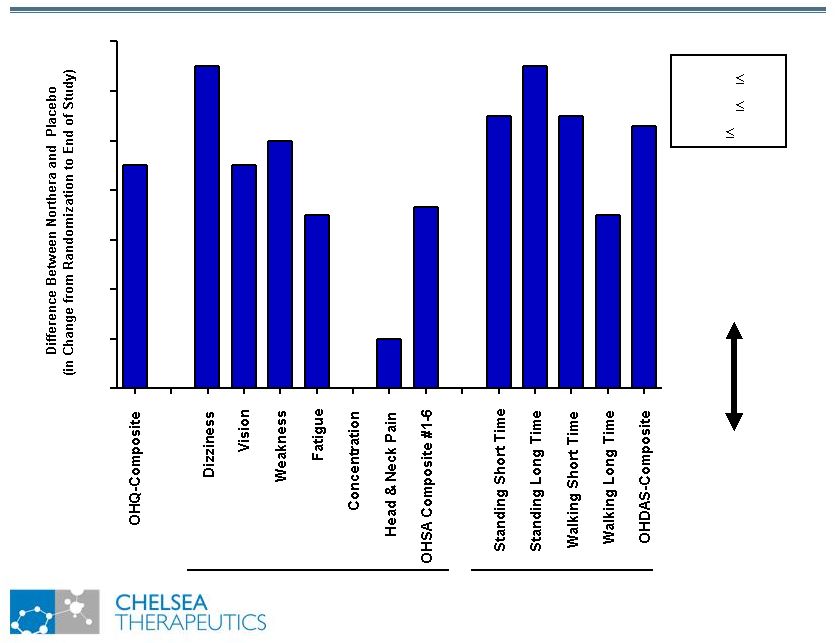 NASDAQ: CHTP Study 301: Broad Symptomatic Benefits Study 301: Broad Symptomatic Benefits Favors Northera Favors Placebo 0.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 OHSA OHDAS *** * p 0.05 ** p 0.01 *** p 0.001 ** * * ** *** ** ** ** ** ** |
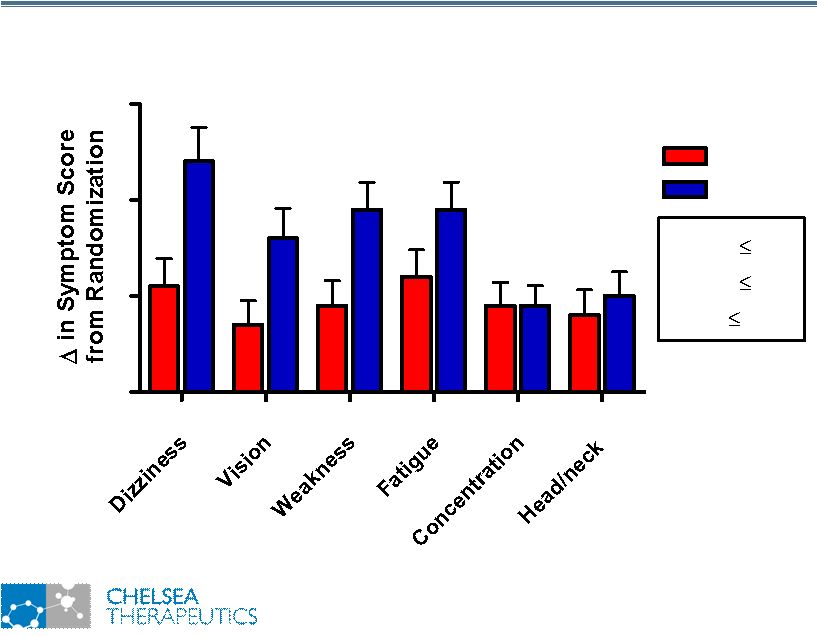 NASDAQ: CHTP 10 Study 301: Change in OHSA Scores Study 301: Change in OHSA Scores 0 1 2 3 Placebo * p 0.05 ** p 0.01 *** p 0.001 *** ** * * Northera |
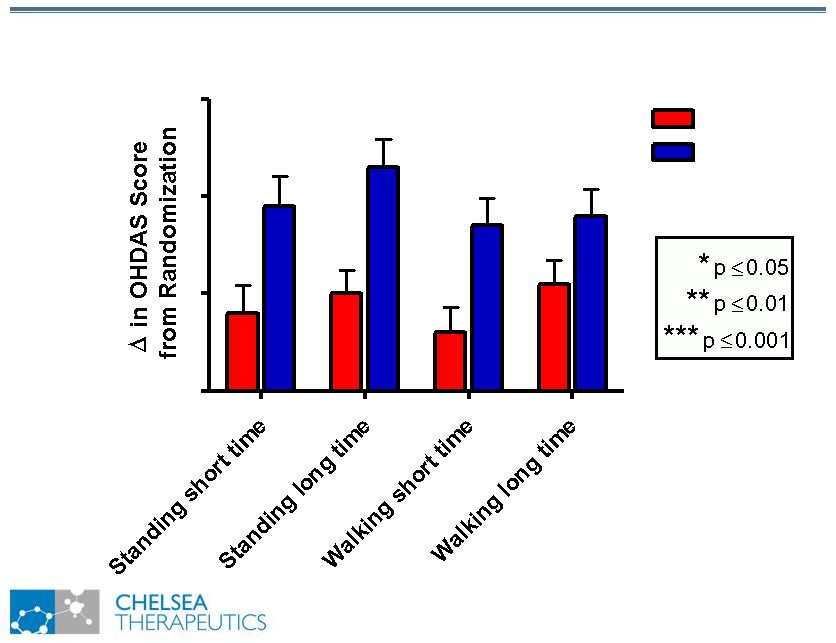 NASDAQ: CHTP 11 Study 301: Change in OHDAS Scores Study 301: Change in OHDAS Scores 0 1 2 3 Placebo *** ** ** ** Northera |
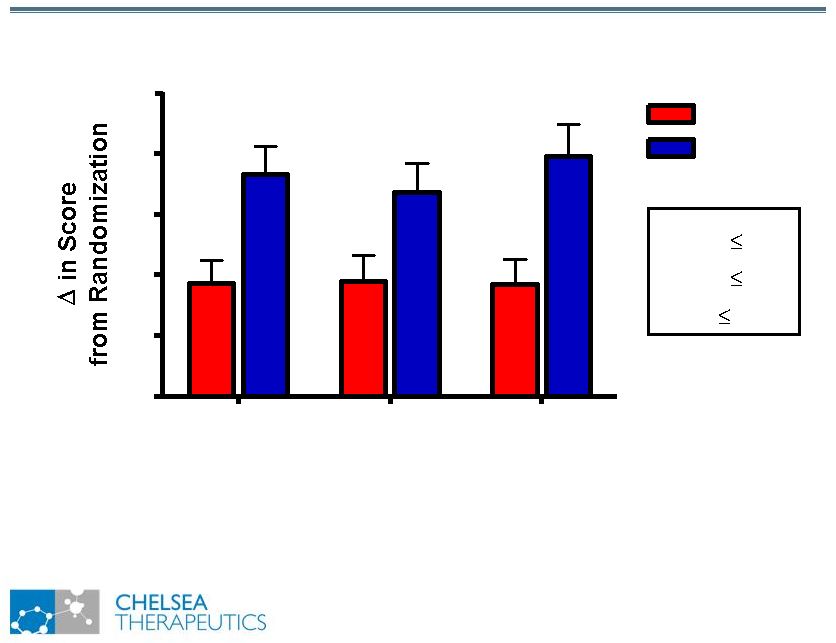 NASDAQ: CHTP 12 Study 301: Change in Composite Scores Study 301: Change in Composite Scores OHQ OHSA OHDAS 0.0 0.5 1.0 1.5 2.0 2.5 Placebo * p 0.05 ** p 0.01 *** p 0.001 ** ** ** Northera |
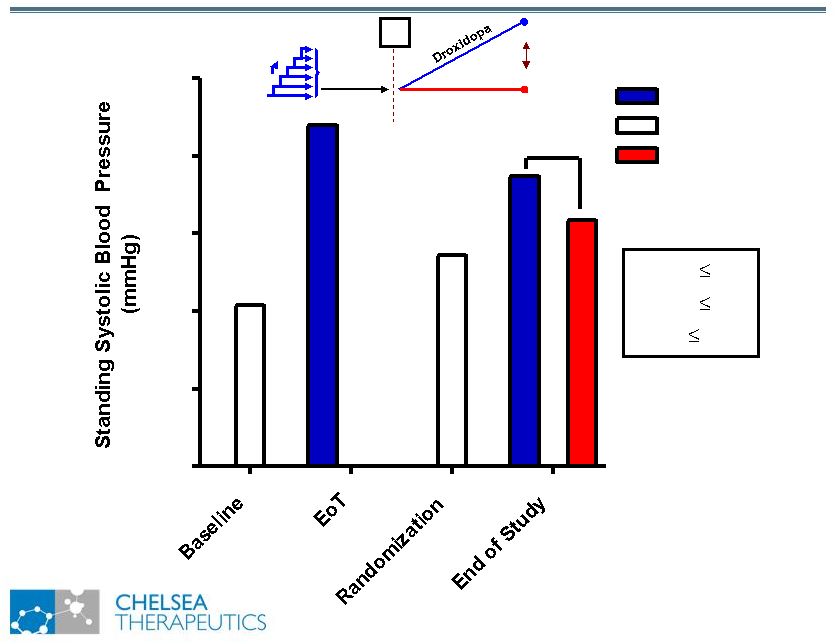 NASDAQ: CHTP 13 Study 301: Effect of Northera on Standing SBP Study 301: Effect of Northera on Standing SBP Washout R Placebo 70 80 90 100 110 120 Northera No Drug Placebo * p 0.05 ** p 0.01 *** p 0.001 *** |
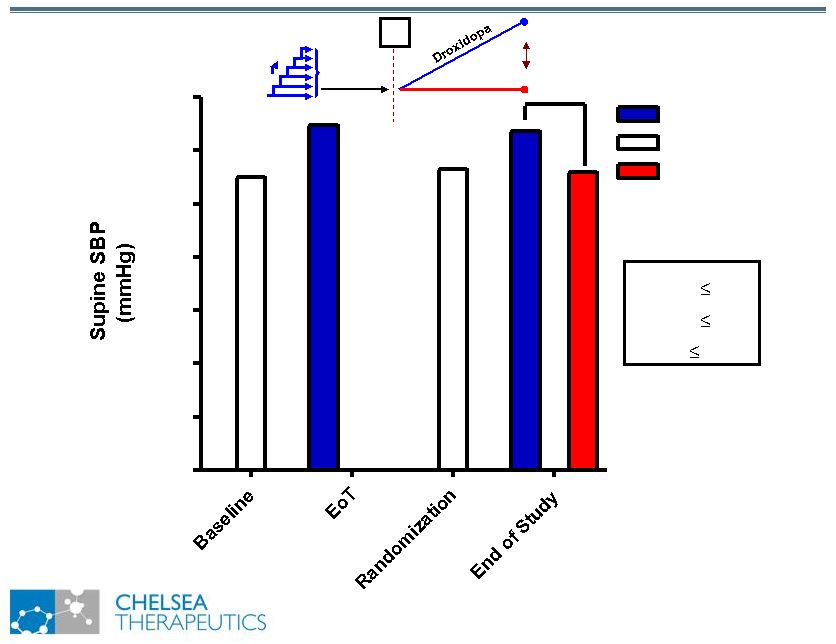 NASDAQ: CHTP 14 Study 301: Effect of Northera on Supine SBP Study 301: Effect of Northera on Supine SBP Washout R Placebo 70 80 90 100 110 120 130 140 Northera No Drug Placebo * p 0.05 ** p 0.01 *** p 0.001 *** |
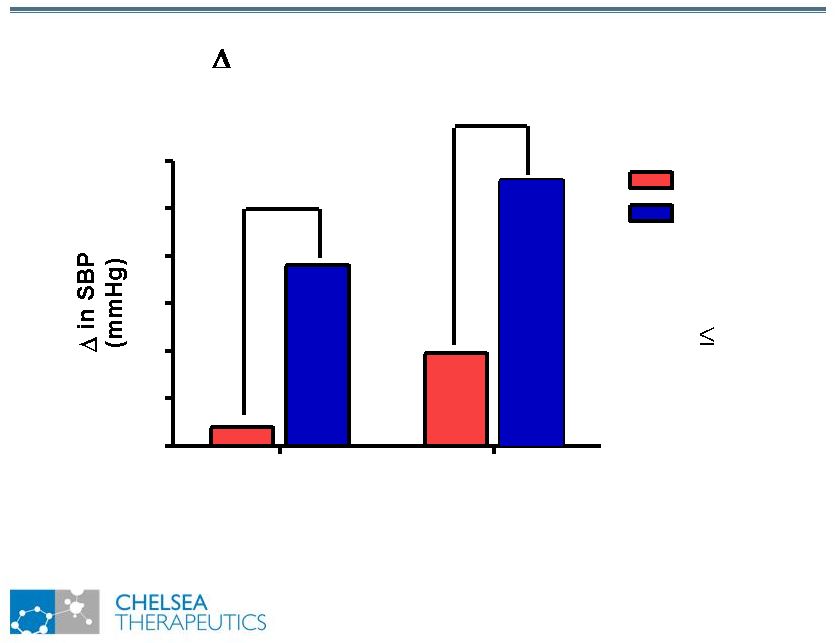 NASDAQ: CHTP 15 Study 301: Effect of Northera Study 301: Effect of Northera on Standing SBP on Standing SBP SBP from randomization Supine Standing 0 2 4 6 8 10 12 Placebo Northera *** p 0.001 *** *** |
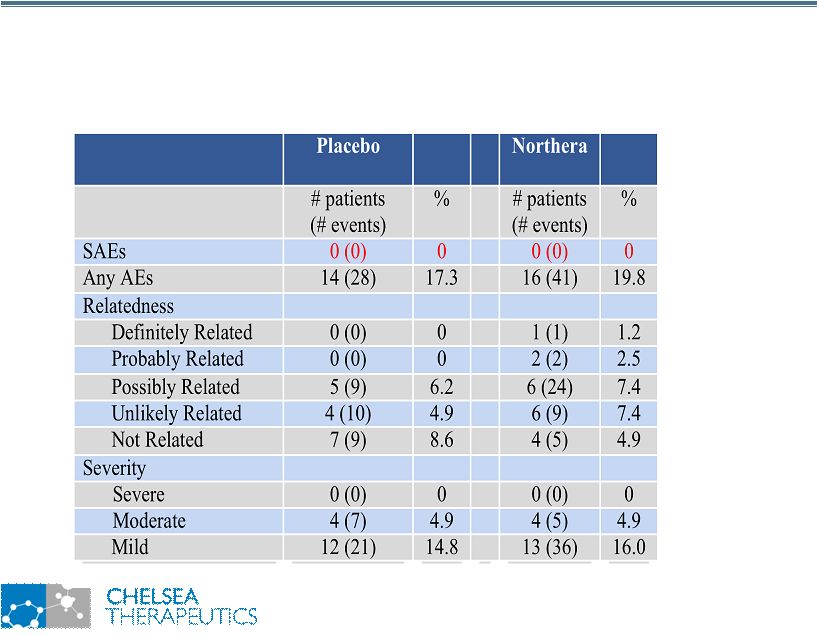 NASDAQ: CHTP 16 Study 301: No Serious Adverse Events Study 301: No Serious Adverse Events • Most common adverse events reported: • Headache: 0.0 % placebo, 7.4 % Northera • Falls: 4.3 % placebo, 0.0 % Northera |
  NASDAQ: CHTP Study 301: Response in Parkinson’s Disease Study 301: Response in Parkinson’s Disease 0.00 0.25 0.50 0.75 1.00 1.25 1.50 1.75 2.00 2.25 Placebo Northera |
 NASDAQ: CHTP 18 0 2 4 6 8 10 12 0 1 2 3 4 5 6 7 8 9 302 301 303 Weeks of Treatment Study 302: Parkinson’s Response Over Time Study 302: Parkinson’s Response Over Time |
 NASDAQ: CHTP 19 Study 306: NOH in Parkinson’s Disease Study 306: NOH in Parkinson’s Disease Randomization Visit 2 Up to 2 weeks 8 weeks Visit 1 Visit 3a, 3b, 3c…. Visit 7 2 weeks • Enriched Homogeneous population: 84 PD Patients with NOH • Longer Treatment duration allows active arm to improve and placebo to fail • Primary efficacy measure: OHQ composite • Top-line data Q2 2011 Double-Blind Titration Double-Blind Treatment |
 NASDAQ: CHTP 20 Next Steps: NOH Registration Program Next Steps: NOH Registration Program Complete Study 301 Data Analysis Present full data set: American Autonomic Society Annual Meeting, Nov. 2010 Continue Preparations for Rolling NDA Submission to be initiated first half 2011 Request Pre-NDA Meeting Complete Study 306 Estimated Completion of Enrollment: Year End 2010 Top-line Data: Q2 11 Complete Data Analysis for NDA Complete NDA Submission: Q3 11 Anticipating Robust Filing Package Supportive Data from Study 302 Highly Significant Outcome in Study 301 Confirmatory Data from Study 306 Strong Safety Data from Studies 303, 304 & 305 Meaningful Data from Japanese registration program Fast-Track status should expedite NDA review process |
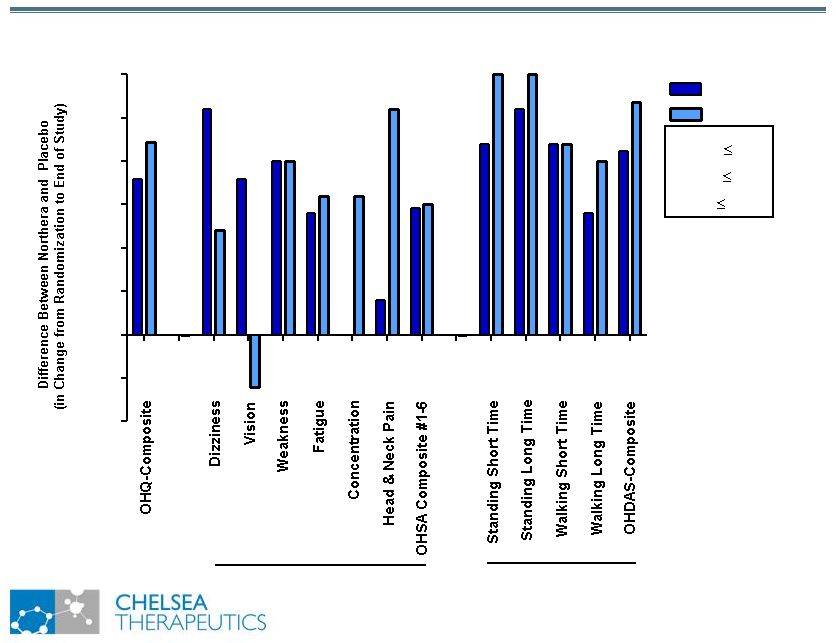 NASDAQ: CHTP Study 301: Broad Symptomatic Benefits Study 301: Broad Symptomatic Benefits -0.50 -0.25 0.00 0.25 0.50 0.75 1.00 1.25 1.50 OHSA OHDAS 302 301 * p 0.05 ** p 0.01 *** p 0.001 * ** *** ** ** * ** ** *** ** ** * * * * |
 NASDAQ: CHTP 22 Positioning Northera Positioning Northera Target Profile: Northera™ is used as first line chronic oral therapy in NOH because it is the only drug proven to reduce the physical symptoms of dizziness, lightheadedness, and falling sensations associated with neurogenic orthostatic hypotension by safely replenishing low levels of the natural neurotransmitter, norepinephrine. Pharmacoeconomic Rationale: Reduction of Falls • Of the estimated 1.7 million nursing home residents in the US, approximately half fall annually, twice the rate for persons dwelling in the community; 11% sustain a serious fall-related injury. • The mean incidence of falls in nursing homes is 1.5 falls per bed per year. The most common precipitating causes include gait and balance disorders, weakness, dizziness, environmental hazards, confusion, visual impairment, and postural hypotension. • $10B for hip fractures (1996)* • Does not include cost for head trauma, soft tissue injury, dislocations, etc. • $20B for all fall related costs (1998)** |
 • Poor Surrogate at Best: Midodrine (ProAmatine®)…alpha agonist: 52,000 patients* • Not approved for symptomatic benefit • Provides constant/undifferentiated pressor effect: equal effect supine vs. standing • Black box warning for supine hypertension…22% at 10 mg. dose • Alpha agonist side effects • piloerection (goose bumps, hair standing on end) • paresthesia (tingling, pricking or numbness of skin (scalp) • Poor patient persistence and compliance • Poor penetration of PD Market • $55-60M sales (priced at $30/day would generate $315 million annually) NASDAQ: CHTP 23 Current Therapeutic Landscape Current Therapeutic Landscape *Verispan |
 • Attractive US Pricing Model: US sales of $300-$375 million within 3-5 years of launch assuming: • Price: $30/day • Compliance: 70% • Limited Sales Force Requirements ~ 85 Sales Reps • 10% of Midodrine Sales Generated by Only 232 Physicians • ~4,000 physicians responsible for 50% of midodrine sales • No Competing Sales Force • Personal Promotion: Top 5 Deciles: ~20,000 MDs • Direct Sales force promotion to high prescribers • Promotional support & spending at Medical Meetings • Sampling program for new patients • Medical Science Liaison outreach to KOLs and Patient Support Groups • Accounts for ~80 % of effort and spending • Non-Personal Promotion: ~45,000 MDs • Journal advertising • Direct mail educational and promotional programs • Sampling programs via mail NASDAQ: CHTP 24 Northera: US Commercial Opportunity Northera: US Commercial Opportunity |
 NASDAQ: CHTP 25 Study 301: Summary Findings Study 301: Summary Findings • Broad Symptomatic & Functional Benefits • Statistically Significant Improvement in Composite OHQ Score vs. Placebo (p=0.003) • Mean decrease of 1.83 units in composite OHQ score • Statistically Significant Improvement in composite OHSA (p=0.01) and composite OHDAS (p=0.003) • Statistically Significant Improvement in 8 out of 10 Individual OHQ Items • Clear Mechanistic Rational for Symptomatic & Functional Improvements • Statistically Significant Improvement in Standing SBP vs. Placebo (p<0.001) • Mean improvement of 11.2 mmHg in standing SBP • Preferential Effect on Standing SBP vs. Supine SBP • Safe and Well Tolerated • No Serious or Severe Adverse Events • Headache (7.4%): all cases reported mild • Treatment associated with fewer falls • No patients on Northera reported falling vs. 3 patients on Placebo |
 NASDAQ: CHTP 26 Looking Ahead…. Looking Ahead…. Top-line Results Northera: Study 303 Q2 10 Initiate Northera PIII: NOH Study 306 Q2 10 Interim Analysis Droxidopa PII: Fibromyalgia H2 10 Top-line Results Northera PIII: NOH Study 301 Q3 10 Initiate CH-4051 PII: RA Q4 10 Top-line Results Droxidopa Investigator PII: ADHD Q1 11 Top-line Results Northera PIII: NOH Study 306 Q2 11 Un-blinded Interim Data CH-4051: RA Q3 11 Top-line Results Droxidopa PII: Fibromyalgia YE 11 • Operational Focus • Drive Northera Registration Program/NDA • Advance CH-4051 in RA and Droxidopa in additional indications • Execute strategic, cash generating licensing/partnership opportunities • Upcoming Milestones |