Exhibit 99.1

Exhibit 99.1
September 2011
© 2004-2011 Chelsea Therapeutics, Inc.

Forward-Looking Statement
This presentation is being provided for informational and discussion purposes. This presentation is not intended to provide and should not be relied upon as investment advice or an opinion regarding the appropriateness or suitability of any investment. Nothing herein should be construed to be an offer to sell, or a solicitation of an offer to buy, any securities.
This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include failure to get regulatory approval for our product candidates, market acceptance for approved products, management of rapid growth, risks of regulatory review and clinical trials, intellectual property risks, and the need to acquire additional products. The reader is referred to the documents that Chelsea Therapeutics International, Ltd. files from time to time with the Securities and Exchange Commission.
NASDAQ: CHTP

Overview
Two Major Assets Targeting Multiple, High Potential Markets
Droxidopa:
Neurogenic Orthostatic Hypotension*(NortheraTM)
Intradialytic Hypotension* Fibromyalgia Adult ADHD
Freezing of Gait* Chronic Fatigue
Other norepinephrine related indications
Rheumatoid Arthritis
Psoriasis Crohn’s Disease Ankylosing Spondylitis Uveitus Oncology Other Inflammatory Diseases
Non-Metabolized Antifolates:
Five Clinical Trials Expected to Report Data over the next 12-18 months
Phase III Trial: Falls in Parkinson’s Disease Patients with Neurogenic Orthostatic Hypotension Four Phase II Proof of Concept Trials : ADHD, Fibromyalgia, Chronic Fatigue Syndrome & RA
*Approved indication in Japan
NASDAQ: CHTP

Northera™ Overview
Oral “Prodrug”of Norepinephrine: Directly metabolized to form Norepinephrine
• Replenishes Diminished Level of Natural Hormone, Norepinephrine,
Within Autonomic Nervous System
• Unique Mechanism of Action Limits Side Effects Seen with Other Drugs
• Only Chronic Oral Therapy Treating Root Cause of Neurogenic Orthostatic Hypotension
Well Documented Safety and Efficacy
• Marketed Ex US (Japan) Since 1989
• Orthostatic Hypotension associated with Parkinson’s disease
• Freezing of Gait in Parkinson’s Disease
• Hypotension associated with dialysis
NASDAQ: CHTP

Lead Indication: Neurogenic Orthostatic Hypotension
Neurogenic Orthostatic Hypotension (NOH): Sudden Fall in BP When Standing From a Sitting/Lying Position
•Symptoms include dizziness, lightheadedness, syncope
• Some patients cannot stand unaided for more than a few minutes a day
•Caused by Diminished Synthesis and/or Release of the Norepinephrine Used by Autonomic Nerves to Regulate Blood Vessels and Heart
•Three Primary Patient Groups: Parkinson’s disease (PD), multiple system atrophy (MSA) and pure autonomic failure (PAF)
NASDAQ: CHTP

NOH: Current Therapeutic Landscape
Poor Surrogate at Best: Midodrine (ProAmatine®)…alpha agonist: 52,000 patients*
• Not approved for symptomatic benefit
• Provides constant/undifferentiated pressor effect
• Black box warning for supine hypertension…22% at 10 mg. dose
• Alpha agonist side effects
• piloerection (goose bumps, hair standing on end) • paresthesia (tingling, pricking or numbness of skin (scalp)
• Poor patient persistence and compliance
• Poor penetration of PD Market
• $55-60M peak sales (priced at $30/day would generate $315 million annually)
*Verispan, 2007
NASDAQ: CHTP
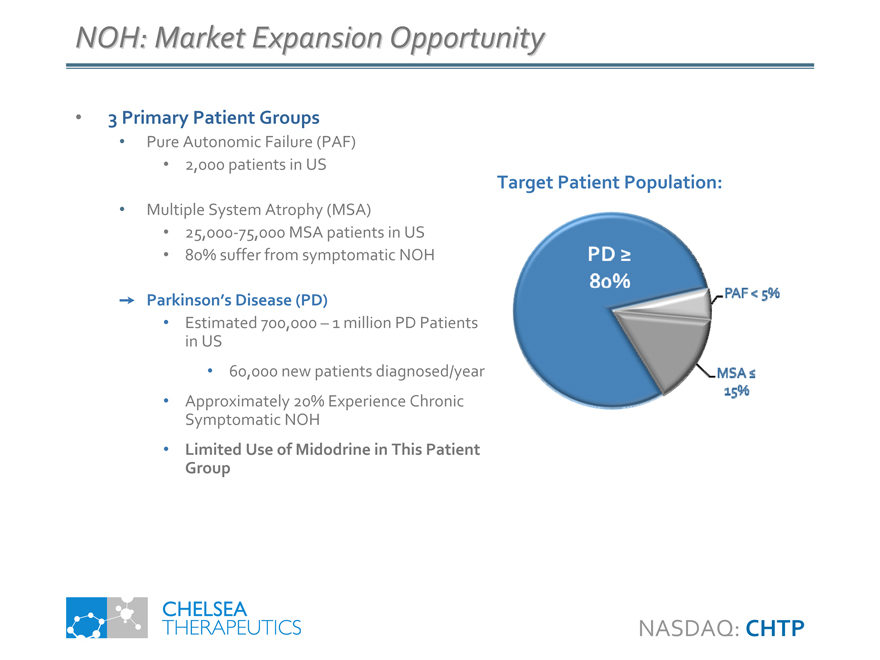
NOH: Market Expansion Opportunity
• 3 Primary Patient Groups
• Pure Autonomic Failure (PAF)
• 2,000 patients in US
• Multiple System Atrophy (MSA)
• 25,000-75,000 MSA patients in US
• 80% suffer from symptomatic NOH
Parkinson’s Disease (PD)
• Estimated 700,000 – 1 million PD Patients in US
• 60,000 new patients diagnosed/year
• Approximately 20% Experience Chronic Symptomatic NOH
• Limited Use of Midodrine in This Patient Group
Target Patient Population:
PD >= 80%
PAF < 5%
MSA <=15%
NASDAQ: CHTP

Northera: US Commercial Opportunity in NOH
Attractive US Pricing Model: US sales of $300-$375 million within 3-5 years of launch assuming:
• Chronic Treatment
• Price: $30/day
• Compliance: 70%
Limited Sales Force Requirements ~ 85 Sales Reps
• 10% of Midodrine Sales Generated by Only 232 Physicians
• ~4,000 Physicians Responsible for 50% of Midodrine Sales
• No Competing Sales Force
Personal Promotion: Top 5 Deciles: ~10,000 to 12,000 MDs
• Direct Sales Force Promotion to High Prescribers
• Promotional Support & Spending at Medical Meetings
• Sampling Program for New Patients
• Medical Science Liaison Outreach to KOLs and Patient Support Groups
• Accounts for ~80 % of Effort and Spending
Non-Personal Promotion: ~45,000 MDs
• Journal Advertising
• Direct Mail Educational and Promotional Programs
• Sampling Programs via Mail
NASDAQ: CHTP

Northera NDA: Neurogenic Orthostatic Hypotension
•New Drug Application: Q3 2011
• FDA confirms 301 and supportive 302 data sufficient for NDA in NOH, 306 data not required
• Fast Track Status
• Anticipating Priority Review, Estimated Approval Q1 12
Study 301: Pivotal Proof of Efficacy Study– Induction Design (conducted under SPA)
• Study 304: Long-Term Safety Extension to Study 301
• Study 305: 24 HR Blood Pressure Monitoring Study – Subgroup of 301 Patients
Study 302: Supporting Phase III Efficacy Study – Withdrawal Design
• Study 303: Long-Term Safety Extension to Study 302
NASDAQ: CHTP
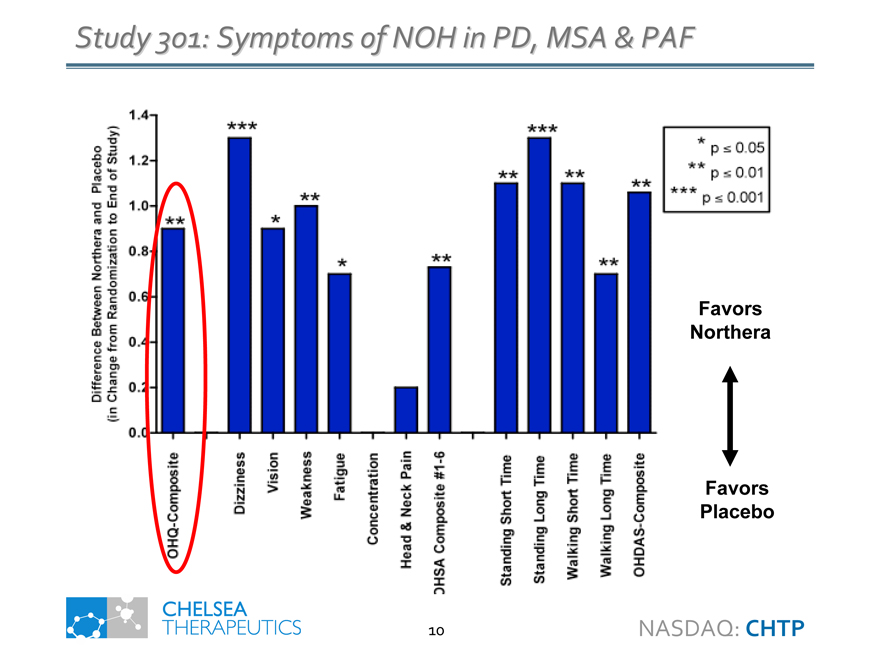
Study 301: Symptoms of NOH in PD, MSA & PAF
Difference Between Northera and Placebo
(in Change from Randomization to End of Study)
1.4
1.2
1.0
0.8
0.6
0.4
0.2
0.0
OHQ-Composite
Dizziness
Vision
Weakness
Fatigue
Concentration
Head & Neck Pain
OHSA Composite #1-6
Standing Short Time
Standing Long Time
Walking Short Time
Walking Long Time
OHDAS-Composite
p <= 0.05
p <= 0.01
p <= 0.001
Favors Northera
Favors Placebo
NASDAQ: CHTP
10
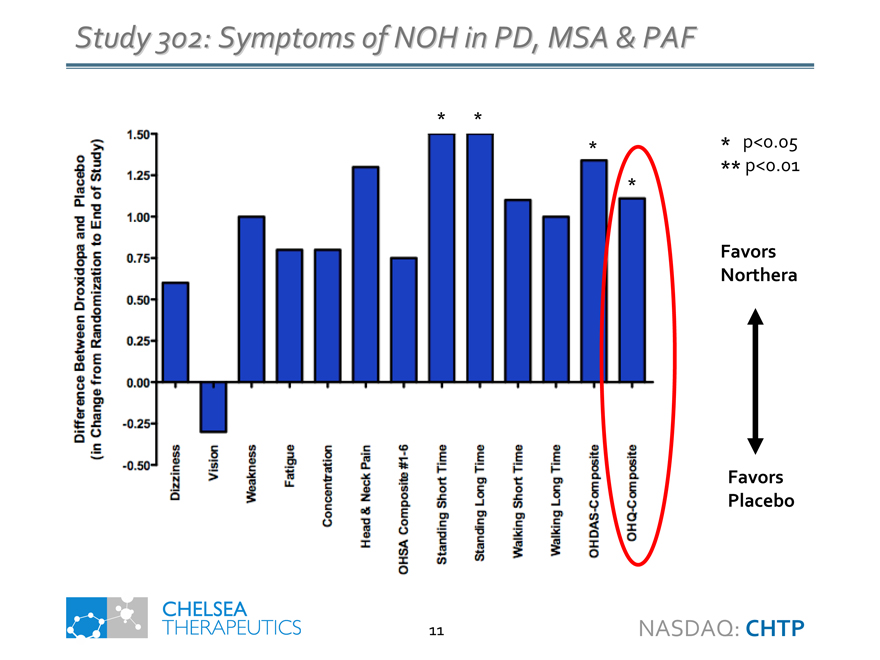
Study 302: Symptoms of NOH in PD, MSA & PAF
Difference Between Droxidopa and Placebo
(in Change from Randomization to End of Study)
1.50
1.25
1.00
0.75
0.50
0.25
0.00
-0.25
-0.50
Dizziness
Vision
Weakness
Fatigue
Concentration
Head & Neck Pain
OHSA Composite #1-6
Standing Short Time
Standing Long Time
Walking Short Time
Walking Long Time
OHDAS-Composite
OHQ-Composite
* p<0.05 ** p<0.01
Favors Northera
Favors Placebo
NASDAQ: CHTP
11
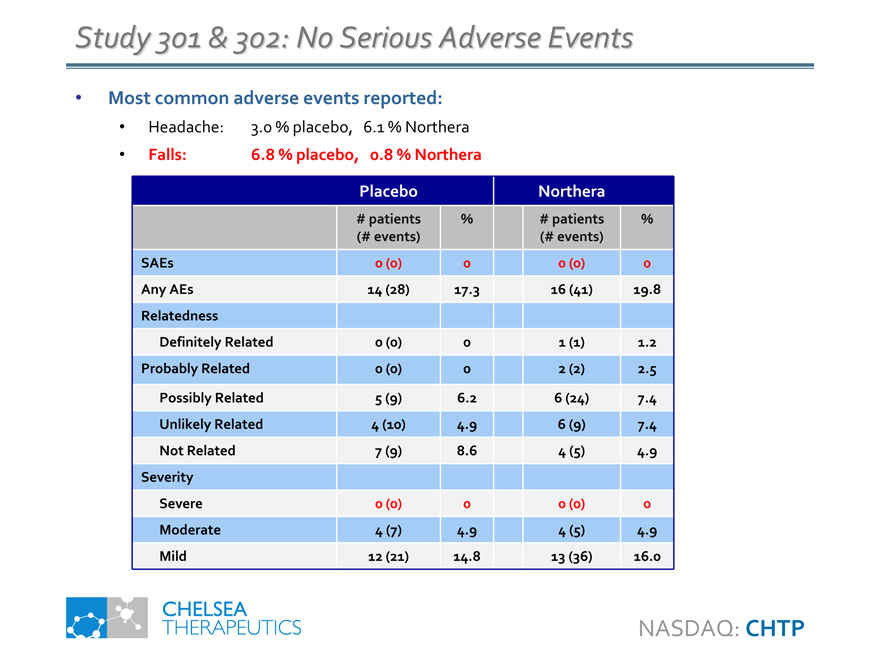
Study 301 & 302: No Serious Adverse Events
Most common adverse events reported:
• Headache: 3.0 % placebo, 6.1 % Northera
• Falls: 6.8 % placebo, 0.8 % Northera
Placebo Northera
# patients % # patients %
(# events) (# events)
SAEs 0 (0) 0 0 (0) 0
Any AEs 14 (28) 17.3 16 (41) 19.8
Relatedness
Definitely Related 0 (0) 0 1 (1) 1.2
Probably Related 0 (0) 0 2 (2) 2.5
Possibly Related 5 (9) 6.2 6 (24) 7.4
Unlikely Related 4 (10) 4.9 6 (9) 7.4
Not Related 7 (9) 8.6 4 (5) 4.9
Severity
Severe 0 (0) 0 0 (0) 0
Moderate 4 (7) 4.9 4 (5) 4.9
Mild 12 (21) 14.8 13 (36) 16.0
NASDAQ: CHTP

Expanding the Label in Parkinson’s Disease: Falls
Injuries sustained in falls are a common source of serious morbidity and mortality
• leading cause of death in the elderly population
#1 reason for patients being admitted to the hospital in PD
2nd most common reason for PD patients to be admitted to institutional care
• 1/3 of PD related nursing home admissions related to falls
Estimated 20% of patients with Parkinson’s disease have symptomatic NOH
• NOH (symptomatic & non-symptomatic) occurs in 43-58% of patients with PD
NASDAQ: CHTP

Northera sNDA: Reduction in Falls in PD with NOH
Study 306A: Phase III
PreliminaryData Reported January 2011
Preliminary, unblinded data from 51 patients that completed treatment
Data safety monitoring committee recommends trial continue to evaluate the reduction in falls associated with Northera treatment in Parkinson’s disease patients with NOH
Study 306B: Supporting Phase III Efficacy Study
Data: Mid 2012
• FDA Confirmed Suitability of Primary Endpoint: Falls per patient/week
• Powering for approximately 45 % reduction in the number of falls per patient/week
• Target Enrollment: 160 patients
• Anticipate top line data during Q2 2012
Future Studies to Support 306B Findings
• To be initiated post-approval, following 306B results
NASDAQ: CHTP
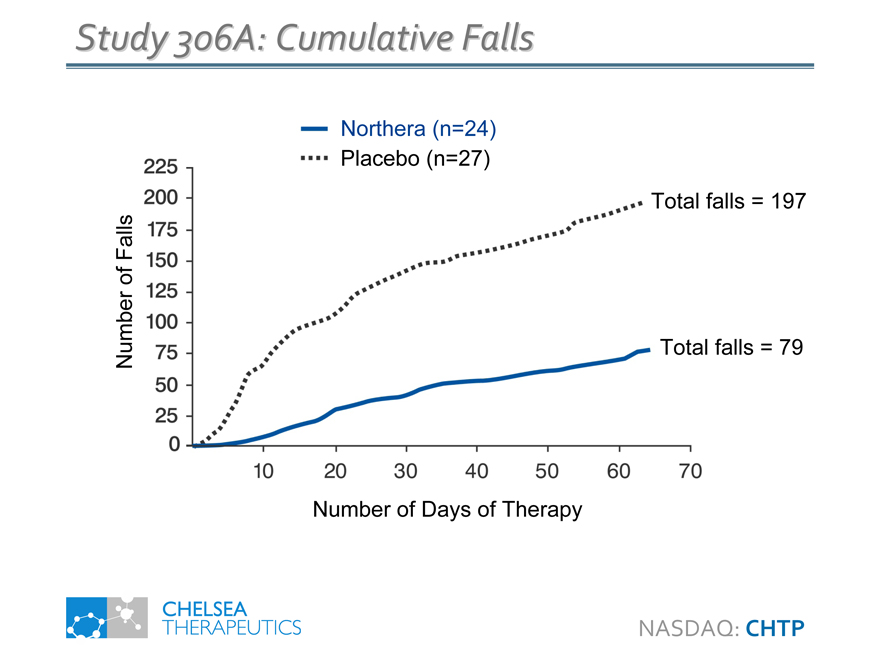
Study 306A: Cumulative Falls
Number of Falls
225
200
175
150
125
100
75
50
25
10
20
30
40
50
60
70
Northera (n=24) Placebo (n=27)
Total falls = 197
Total falls = 79
Number of Days of Therapy
NASDAQ: CHTP

Capturing Falls: A Daily Diary
Each patient prompted to record falls data daily in electronic diary
• A fall is defined as UNEXPECTANTLY coming to rest on the ground, or just a lower level than where patient started e.g. landing on or catching yourself on furniture or a cane
In addition to the number of daily falls, diary prompts for data on most severe fall of the day, including:
• Location and familiarity of location, floor surface, footwear, obstacles involved (chairs, doorways, etc)
• Symptoms prior to fall (lightheadedness, freezing, loss of consciousness, dyskinesia, sleepiness, confusion)
• Activities surrounding fall (changing position, holding something, distracted)
• Injury, amnesia, hospitalization following fall
• ON or OFF (ON - Having a good response to levodopa or OFF - lack of levodopa benefit due to being in between doses)
• Recent changes to PD medications
NASDAQ: CHTP
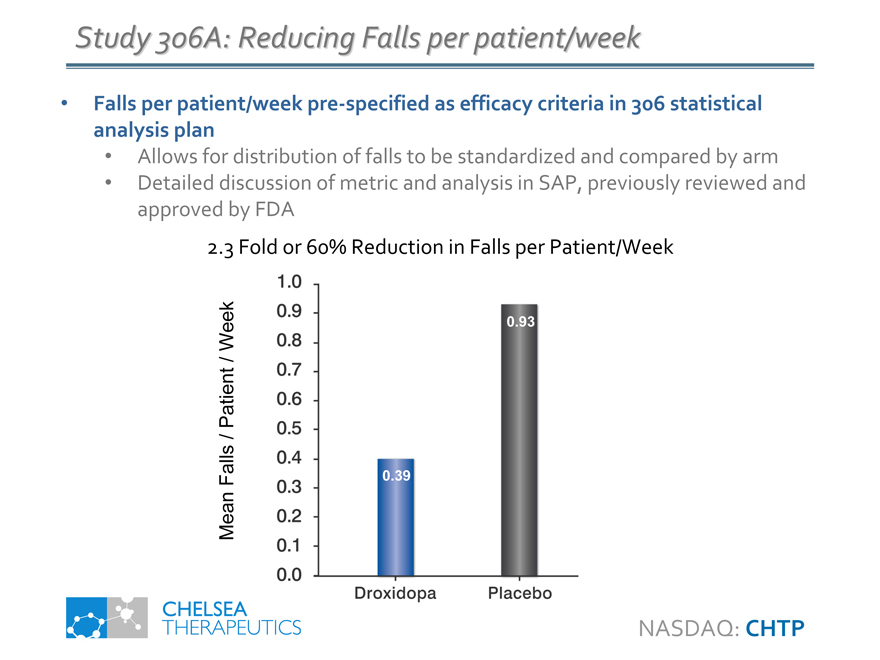
Study 306A: Reducing Falls per patient/week
Falls per patient/week pre-specified as efficacy criteria in 306 statistical analysis plan
• Allows for distribution of falls to be standardized and compared by arm
• Detailed discussion of metric and analysis in SAP, previously reviewed and approved by FDA
2.3 Fold or 60% Reduction in Falls per Patient/Week
Mean Falls / Patient / Week
1.0
0.9
0.8
0.7
0.6
0.5
0.4
0.3
0.2
0.1
0.0
0.39
0.93
Droxidopa
Placebo
NASDAQ: CHTP
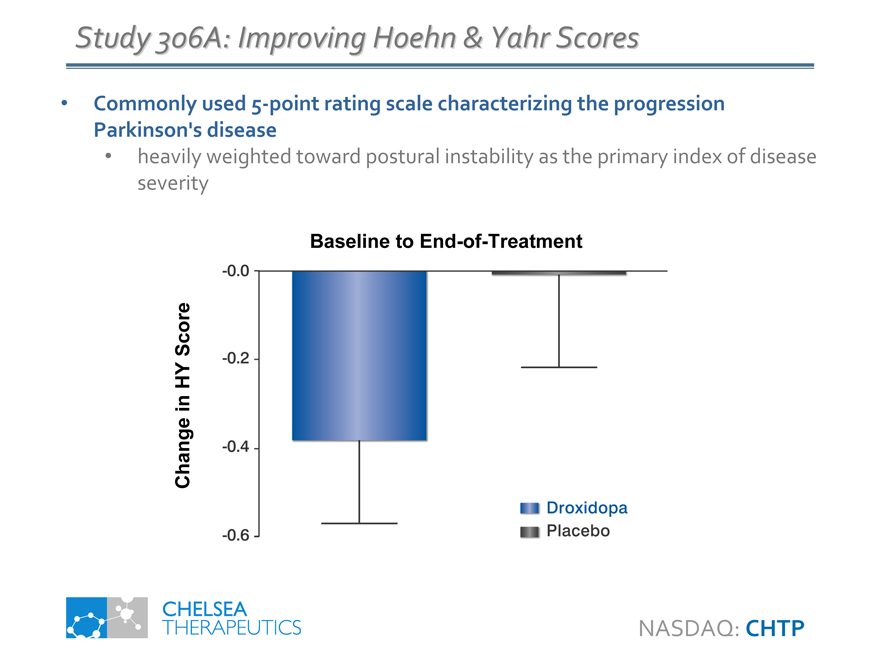
Study 306A: Improving Hoehn & Yahr Scores
Commonly used 5-point rating scale characterizing the progression Parkinson’s disease
• heavily weighted toward postural instability as the primary index of disease severity
Change in HY Score
Baseline to End-of-Treatment
-0.0
-02
-0.4
-0.6
Droxidopa
Placebo
NASDAQ: CHTP
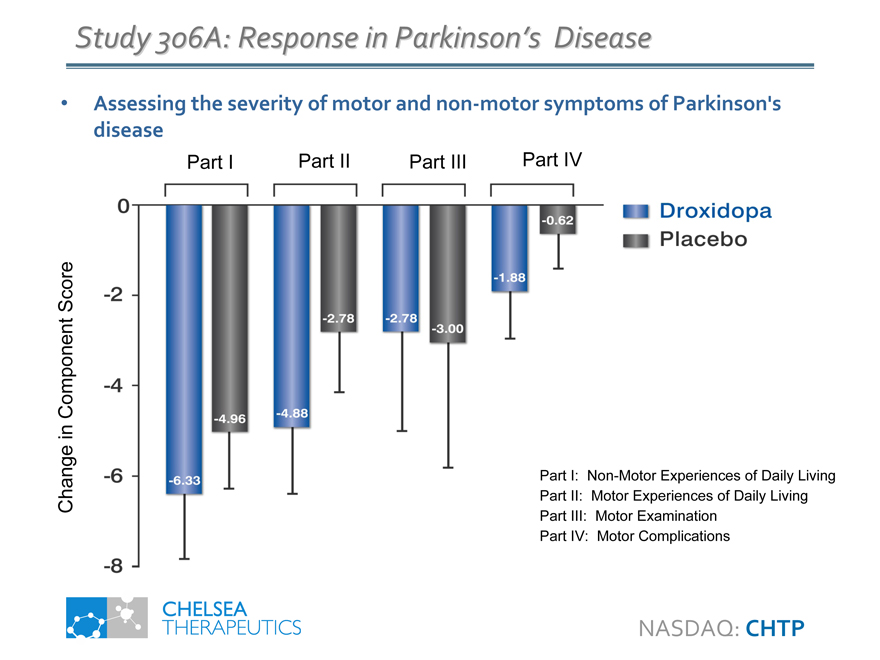
Study 306A: Response in Parkinson’s Disease
Assessing the severity of motor and non-motor symptoms of Parkinson’s disease
Change in Component Score
0
-2
-4
-6
-8
-6.33
-4.96
-4.88
-2.78
-2.78
-3.00
-1.88
-0.62
Droxidopa
Placebo
Part I Part II Part III Part IV
Part I: Non-Motor Experiences of Daily Living
Part II: Motor Experiences of Daily Living
Part III: Motor Examination
Part IV: Motor Complications
NASDAQ: CHTP
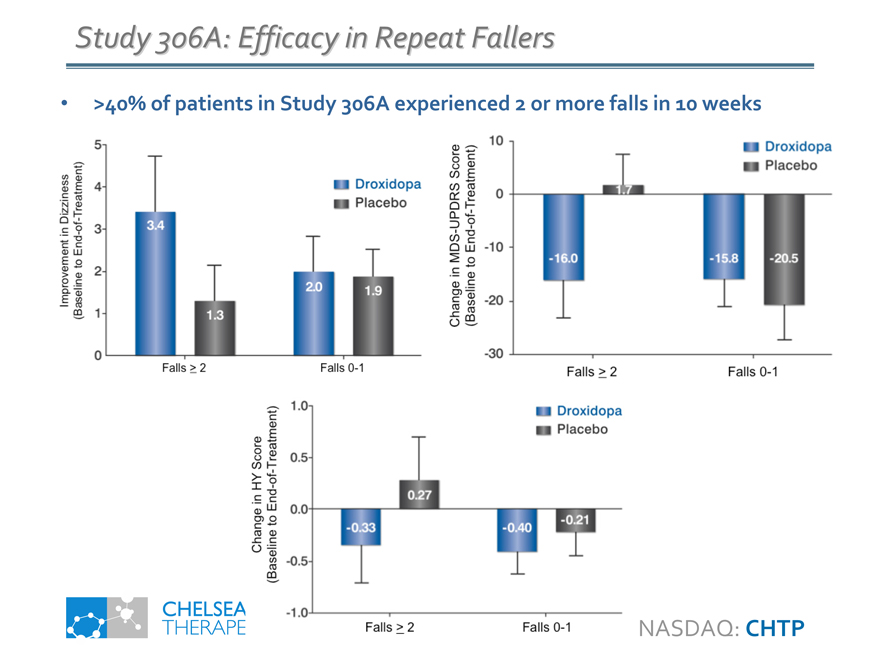
Study 306A: Efficacy in Repeat Fallers
>40% of patients in Study 306A experienced 2 or more falls in 10 weeks
Improvement in Dizziness
(Baseline to End-of-Treatment)
5
4
3
2
1
0
3.4
1.3
2.0
1.9
Falls>=2
Falls 0-1
Droxidopa
Placebo
Change in MDS-UPDRS Score
(Baseline to End-of-Treatment)
10
0
-10
-20
-30
-16.0
1.7
-15.8
-20.5
Falls>=2
Falls 0-1
Droxidopa
Placebo
Change in HY Score
(Baseline to End-of-Treatment)
1.0
0.5
0.0
-0.5
-1.0
-0.33
0.27
-0.40
-0.21
Droxidopa
Placebo
Falls>=2
Falls 0-1
NASDAQ: CHTP

Study 306B: Next Steps
Study 306B
• Primary outcome: falls per patient / week
Powering for 45% reduction in the number of falls per patient / week
• Target total enrollment: 160 patients
• Anticipate top line data during Q2 2012
NASDAQ: CHTP
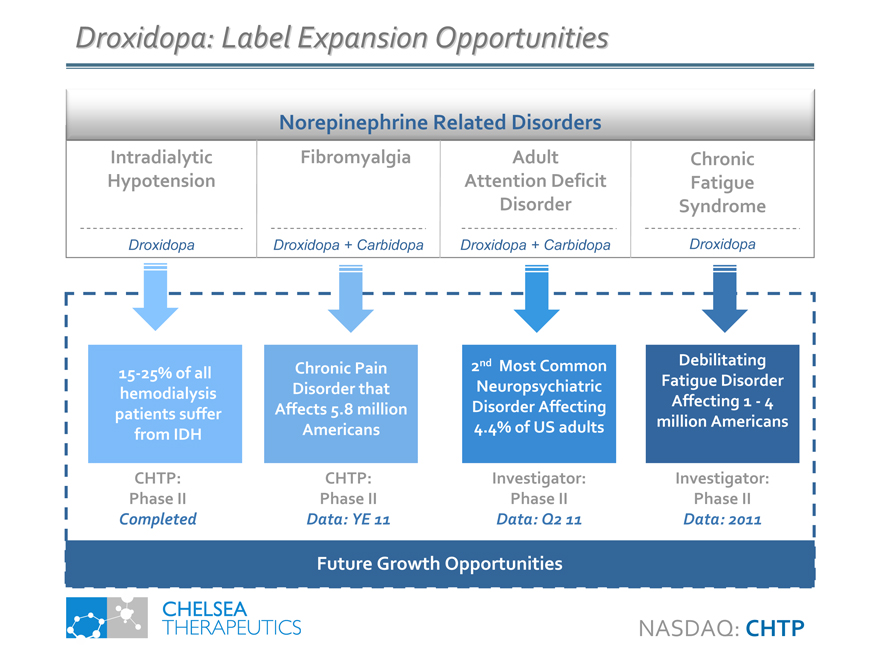
Droxidopa: Label Expansion Opportunities
Norepinephrine Related Disorders
Intradialytic Hypotension
Droxidopa
Fibromyalgia
Droxidopa + Carbidopa
Adult Attention Deficit Disorder
Droxidopa + Carbidopa
Chronic Fatigue Syndrome
Droxidopa
15-25% of all hemodialysis patients suffer from IDH
Chronic Pain Disorder that Affects 5.8 million Americans
2nd Most Common Neuropsychiatric Disorder Affecting 4.4% of US adults
Debilitating Fatigue Disorder Affecting 1—4 million Americans
CHTP: Phase II
Completed
CHTP: Phase II
Data: YE 11
Investigator: Phase II
Data: Q2 11
Investigator: Phase II
Data: 2011
Future Growth Opportunities
NASDAQ: CHTP

Non-Metabolized Antifolates
Novel Antifolate with Blockbuster Potential
• Non-metabolized Oral Antifolate Engineered for Improved Efficacy, Safety & Tolerability
• Potential First Line and Combination Treatment for Rheumatoid Arthritis
• Additional High Potential Global Markets: Psoriasis, Crohn’s Disease, Other Immunological Disorders and Cancer
Methotrexate (MTX) remains most widely prescribed RA treatment:
• Limitations are AEs and long-term safety concerns
• Nausea and Diarrhea
• Kidney and Liver Toxicity
• Undergoes significant metabolism
• Est. 1/3 of MTX Patients Only Achieve a Partial Efficacy Response Likely Associated with Variation in Metabolism of MTX
NASDAQ: CHTP
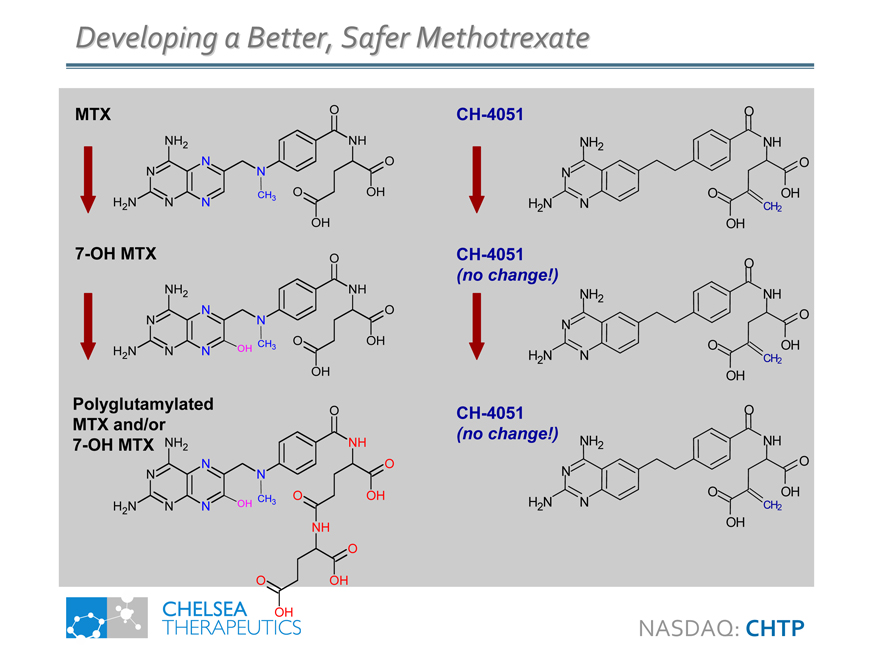
Developing a Better, Safer Methotrexate
MTX
NH2
N
N
H2N
N N
N
CH3
O
NH
O
OH
O
OH
7-OH MTX O
NH2 NH N O
N N
CH O OH H N N N OH 3
2
OH
CH-4051 O
NH2 NH O
N
O OH H2N N CH2 OH
CH-4051
O
(no change!)
NH2 NH O
N
O OH H2N N CH2 OH
O
CH-4051
(no change!)
NH2 NH
O N
O OH H2N N CH2 OH
Polyglutamylated O MTX and/or 7-OH MTX NH2 NH
N O N N
CH O OH H N N N OH 3
2
NH O
O OH
OH
NASDAQ: CHTP

CH-4051: Greater Efficacy, Better Safety
Phase II Proof-of-Concept Study with Racemic Mixture, CH-1504:
Demonstrated Comparable ACR 20 Response Rates To MTX Improved Safety Profile & Tolerability
Key preclinical findings show:
Superior Efficacy: Superior to both CH-1504 and MTX
2-fold Improvement in Signs and Symptoms in Rat Model
2-fold Improvement in Enzymatic Activity (DHFR inhibition)
8-fold Improvement in Cellular Uptake (Reduced Folate Carrier)
Superior Safety/toxicity:
Improved Tolerability in Rat Model
Key findings from phase 1 (SAD/MAD) study show:
Demonstrated safety :
20-fold Higher Doses Than Those Tested with CH-1504 No Serious Adverse Events
Improved pharmacokinetics:
2-fold Improvement in Bioavailability vs. CH-1504
NASDAQ: CHTP
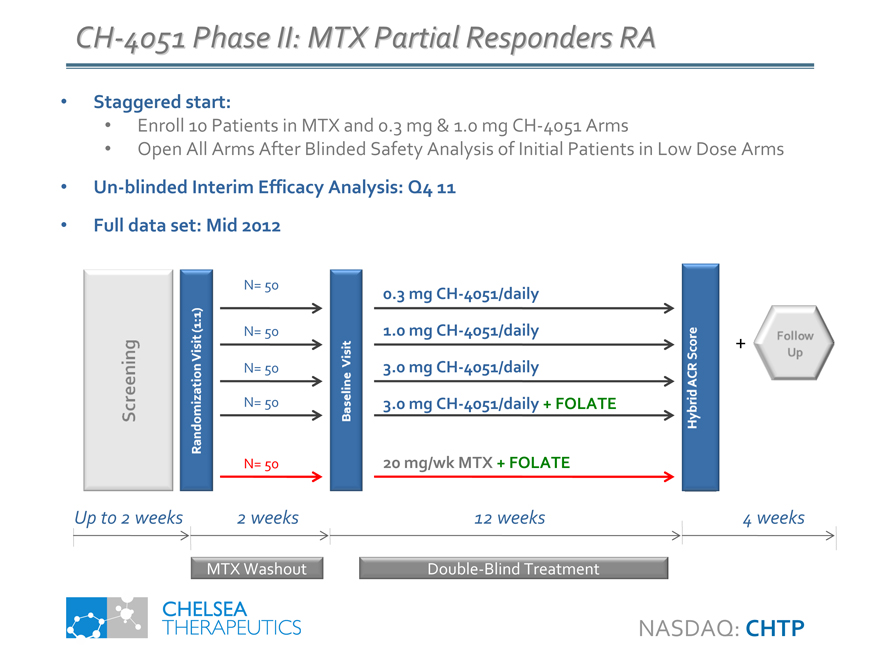
CH-4051 Phase II: MTX Partial Responders RA
• Staggered start:
• Enroll 10 Patients in MTX and 0.3 mg & 1.0 mg CH-4051 Arms
• Open All Arms After Blinded Safety Analysis of Initial Patients in Low Dose Arms
• Un-blinded Interim Efficacy Analysis: Q4 11
• Full data set: Mid 2012
Screening
Randomization Visit (1:1)
N= 50
N= 50 N= 50 N= 50
N= 50
Baseline Visit
0.3 mg CH-4051/daily 1.0 mg CH-4051/daily 3.0 mg CH-4051/daily
3.0 mg CH-4051/daily + FOLATE
20 mg/wk MTX + FOLATE
Hybrid ACR Score
Follow Up
Up to 2 weeks 2 weeks 12 weeks 4 weeks
MTX Washout
Double-Blind Treatment
NASDAQ: CHTP
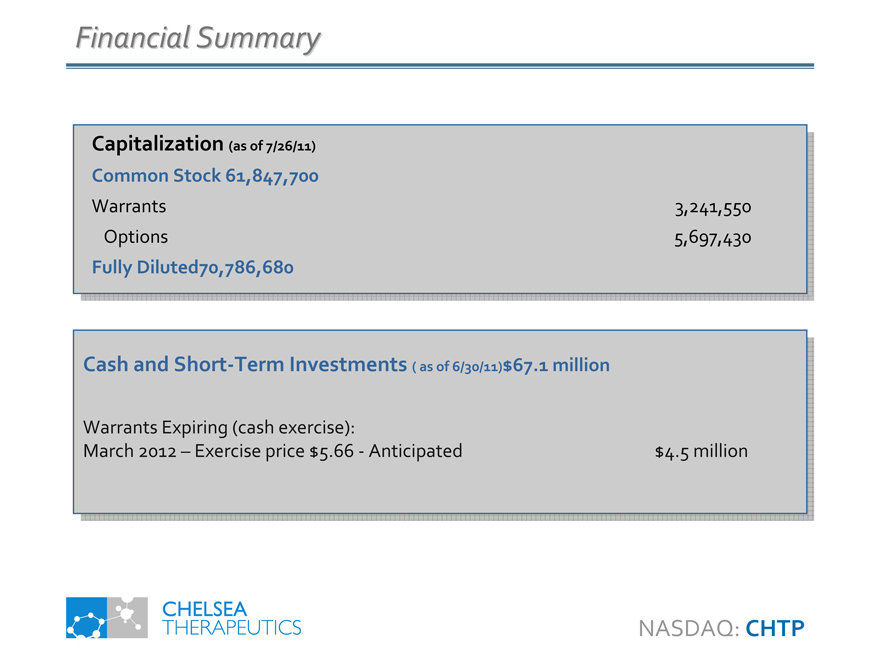
Financial Summary
Capitalization (as of 7/26/11) Common Stock 61,847,700
Warrants Options
Fully Diluted70,786,680
3,241,550 5,697,430
Cash and Short-Term Investments ( as of 6/30/11)$67.1 million
Warrants Expiring (cash exercise):
March 2012 – Exercise price $5.66—Anticipated
$4.5 million
NASDAQ: CHTP

Looking Ahead….
Operational Focus
• Drive Northera Registration Program/NDA
• Advance CH-4051 in RA and Droxidopa in Additional Indications
• Execute Strategic, Cash Generating Licensing/Partnership Opportunities
Upcoming Milestones
Top-line Results Northera PIII: NOH Study 301 Q3 10
Initiate CH-4051 PII: RA Q4 10
Interim Analysis: NOH Study 306 Q1 11
Top-line Results Droxidopa Investigator PII: ADHD July 11
File Northera NDA: NOH Sept. 11
Un-blinded Interim Efficacy Analysis: CH-4051 PII Q4 11
Top-line Results Droxidopa PII: Fibromyalgia Q4 11
Anticipated US Approval of Northera in NOH Q1 12
Top-line Results Northera PIII: NOH Study 306b Q2 12
NASDAQ: CHTP