 Aerie Pharmaceuticals, Inc. Company Overview February 23, 2015 Building a Major Ophthalmic Pharmaceutical Company Exhibit 99.1 |
 2 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved by regulatory authorities. In addition, any discussion of clinical data results for our Rhopressa™ and Roclatan™ product candidates relate to the results in our Phase 2 clinical trials. The information in this presentation is current only as of its date and may have changed or may change in the future. We undertake no obligation to update this information in light of new information, future events or otherwise. We are not making any representation or warranty that the information in this presentation is accurate or complete. Certain statements in this presentation are “forward-looking statements” within the meaning of the federal securities laws, including beliefs, expectations, estimates, projections and statements relating to our business plans, prospects and objectives, and the assumptions upon which those statements are based. Words such as “may,” “will,” “should,” “would,” “could,” “believe,” “expects,” “anticipates,” “plans,” “intends,” “estimates,” “targets,” “projects,” “potential” or similar expressions are intended to identify these forward- looking statements. These statements are based on the Company’s current plans and expectations. Known and unknown risks, uncertainties and other factors could cause actual results to differ materially from those contemplated by the statements. In evaluating these statements, you should specifically consider various factors that may cause our actual results to differ materially from any forward-looking statements. These risks and uncertainties are described more fully in the quarterly and annual reports that we file with the SEC, particularly in the sections titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” In particular, the preclinical research discussed in this presentation is preliminary and the outcome of such preclinical studies may not be predictive of the outcome of later clinical trials. Any future clinical trial results may not demonstrate safety and efficacy sufficient to obtain regulatory approval related to the preclinical research findings discussed in this presentation. Such forward-looking statements only speak as of the date they are made. We undertake no obligation to publicly update or revise any forward-looking statements, whether because of new information, future events or otherwise, except as otherwise required by law. |
 3 Current Aerie Products: Once-Daily IOP-Lowering Eye Drops for Glaucoma Pre-Clinical Research Findings • Rhopressa™ shows potential to modify diseased tissue • May block fibrotic response in trabecular meshwork cells • May increase perfusion of the trabecular meshwork • AR-13154 shows potential for the treatment of wet AMD • May inhibit ROCK/JAK/PDGFR- • Lesion size reduction in rats exceeds market-leading product • Triple action Rhopressa™ • Inhibits ROCK and NET, lowers EVP, targets diseased tissue • Expect P3 efficacy data mid-Q2 2015, NDA Filing mid-2016 • Quadruple Action Roclatan™ • Fixed combination of Rhopressa™ and latanoprost • P2b achieved all clinical endpoints, P3 to start mid-2015 • Potentially most efficacious IOP-lowering therapy Aerie – Building a Major Ophthalmic Pharmaceutical Company These new preclinical discoveries represent potential breakthroughs Full patent protection through at least 2030; Blockbuster Potential |
 4 FY 2013 U.S. Glaucoma Market = $2.0B; 31.5M TRx Market Share in TRx PGA Market Non-PGA Market Current Product Dashboard PGA: Prostaglandin Analogue; BB: Beta Blocker; AA: Alpha Agonist; CAI: Carbonic Anhydrase Inhibitor Once Daily Bimatoprost Travoprost Latanoprost BB BB Fixed Combo AA CAI 15% 8% 10% 10% 14% 10% 31% 2-3 Times Daily |
 5 New MOAs Rhopressa™ ROCK/NET inhibitor (qd) Phase 3 Roclatan™ ROCK/NET inhibitor + PGA (qd) Phase 3 K-115 (Kowa) ROCK inhibitor (bid) Approved in Japan* Adjunctive Therapy AMA0076 (Amakem) ROCK inhibitor (bid) Phase 2a INO-8875 (Inotek) Adenosine-A1 agonist (bid or qd) Phase 2 OPA-6566 (Acucela) Adenosine-A2a agonist (bid) Phase 1/2 SYL040012 (Sylentis) RNAi beta blocker (qd) Phase 2 New PGAs: not usable as add-on to current PGAs Rhopressa™ and Roclatan™: advanced triple and quadruple MOAs New PGAs BOL-303259 (B+L) NO donating latanoprost (qd) Phase 3 DE-117 (Santen) EP2 agonist (qd) Phase 2 ONO-9054 (Ono) FP/EP3 agonist (qd) Phase 2 Glaucoma Competitors in Pipeline * Approved in Japan 9/29/2014 (Aerie AR-13324) (Aerie PG324) |
 6 2023 U.S. Glaucoma Market and Market Share Projections* Projected Market Share in TRx PGA: Prostaglandin Analogue; BB: Beta Blocker; AA: Alpha Agonist; CAI: Carbonic Anhydrase Inhibitor Data Source: IMS 2013 FY TRX. Internal projection *Projected market shares compiled by an independent market research company U.S. Glaucoma Market = $4.9B; 41.7 M TRx TRx Growth 2013-2023 = 2.3% Roclatan™ + Rhopressa™ May Capture Over 40% Market Share Based on 200 U.S. Physician Responses to Survey PGA’s BB BB Fixed Combo AA CAI Rhopressa™ Roclatan™ 31% 18% 25% 6% 9% 5% 5% |
 7 Managed Care Market Research Summary * Respondents ranked products on a scale of 1-7 ** After pricing was disclosed and discussed Payers’ score on the overall utility of Rhopressa™ was 4.7* (Market research firm states that scores above 4 are high) Payers’ score on the overall utility of Roclatan™ was 5.2* (Market research firm states that scores above 5 are rare) 100% of respondents placed Rhopressa™ and Roclatan™ in Tier 2 or Tier 3 of Commercial and Medicare Part D formularies.** There were no “Not Covered” recommendations. Interviews with 12 decision makers whose companies cover over 42 million Commercial, Medicare and Medicaid lives Payers were impressed by Roclatan™’s unprecedented lowering of IOP Payers were receptive to Rhopressa™’s new MOAs and efficacy at any baseline IOP |
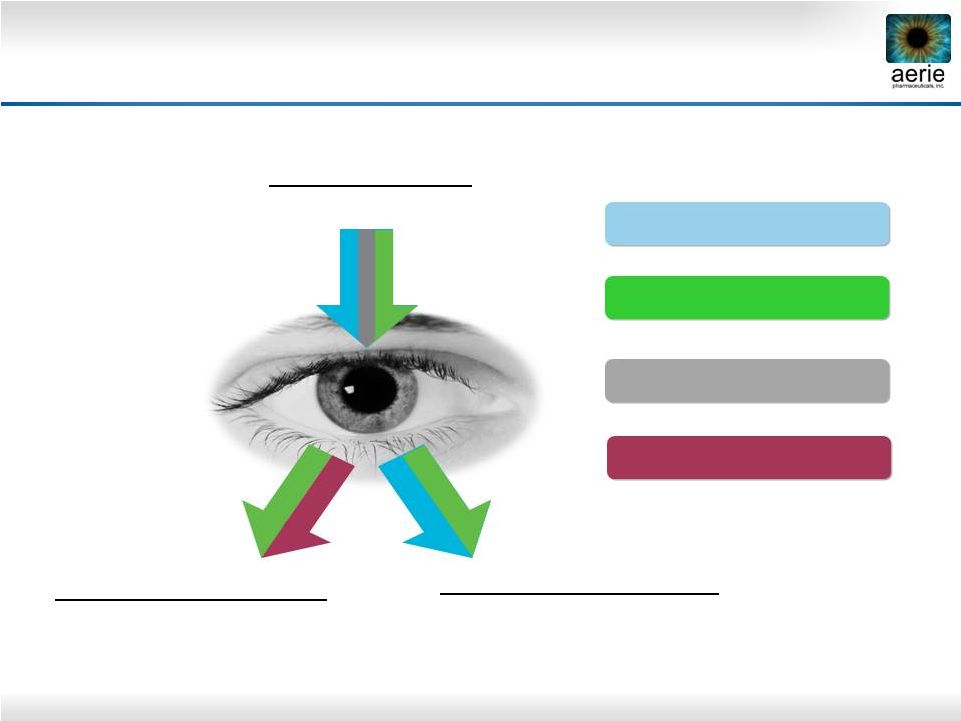 8 increase Decreases Fluid Inflow/Production (Ciliary Processes) Increases Fluid Outflow: Secondary Drain (Uveoscleral Pathway) Increases Fluid Outflow: Primary Drain - Trabecular Meshwork (TM); Lowers EVP - (Episcleral Venous Pressure) Rhopressa™ Roclatan™ AA, BB, CAI PGAs Aerie Products Cover the IOP-lowering Spectrum |
 9 Aerie Product Market Positioning* * Confirmed by Market Research Future drug of choice for the 80% of patients with IOP of 26 mmHg or less Triple-Action Rhopressa™ Future product of choice for patients requiring maximal IOP lowering Quadruple-Action Roclatan™ Also for PGA users as add-on therapy Also for PGA non-responders and those with tolerability concerns Also for patients with low-tension glaucoma Efficacy potentially greater than all currently marketed drugs For patients with IOPs above 26 mmHg Also for patients at any IOP with significant disease progression |
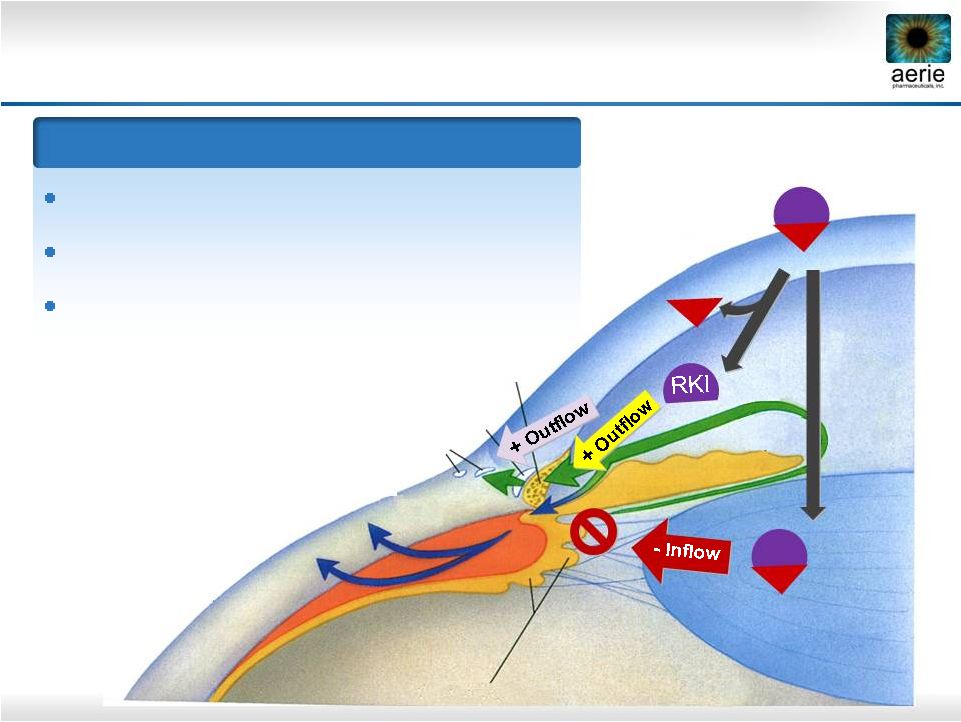 10 Rhopressa ™ NET RKI NET RKI Trabecular Meshwork Triple-Action Rhopressa™ Episcleral Veins Schlemm’s Canal IOP-Lowering Mechanisms ROCK inhibition relaxes TM, increases outflow NET inhibition reduces fluid production ROCK inhibition lowers Episcleral Venous Pressure (EVP) Ciliary Processes Uveoscleral Outflow IOP-Lowering Mechanisms ROCK inhibition relaxes TM, increases outflow NET inhibition reduces fluid production ROCK inhibition lowers Episcleral Venous Pressure (EVP) Cornea |
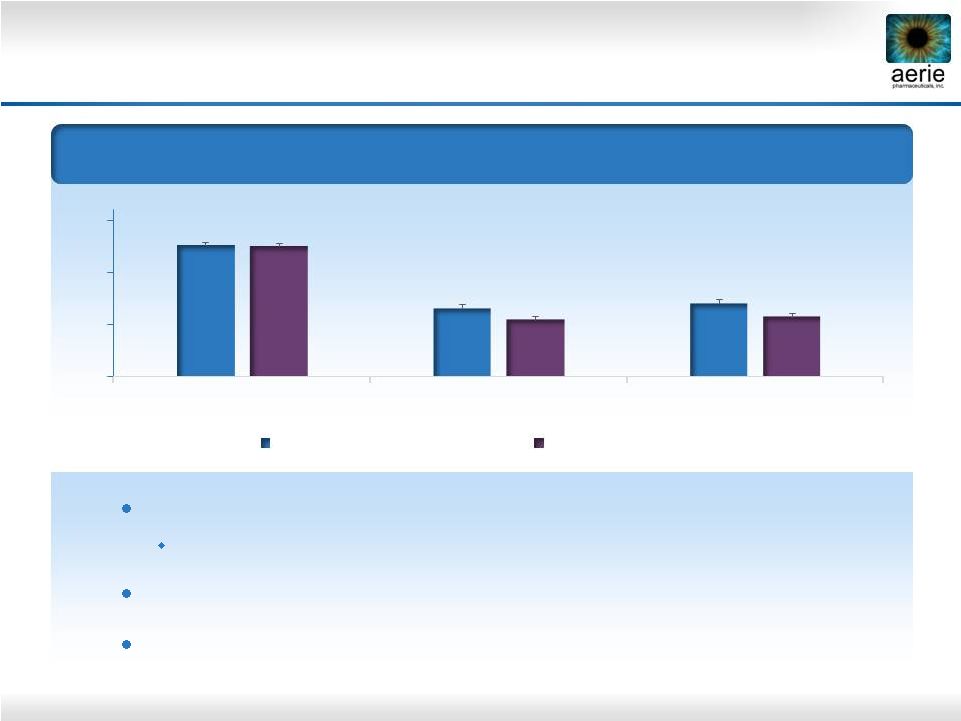 11 Rhopressa™: Powerful Compound for Physiological Lowering of IOP – Phase 2b Results Mean Diurnal IOP – Entry IOP 22-36 mmHg (n=221) 25.5 18.4 18.7 13 18 23 28 Baseline Day 14 Day 28 Rhopressa™ 0.02% (n=71) Latanoprost (n=76) Once-daily PM dosing of 0.02% Rhopressa™ is highly effective IOP -5.7 and -6.2 mmHg on D28 and D14 Rhopressa™ efficacy results within ~1 mmHg of latanoprost Favorable tolerability profile with no systemic side effects 25.6 19.5 20.0 |
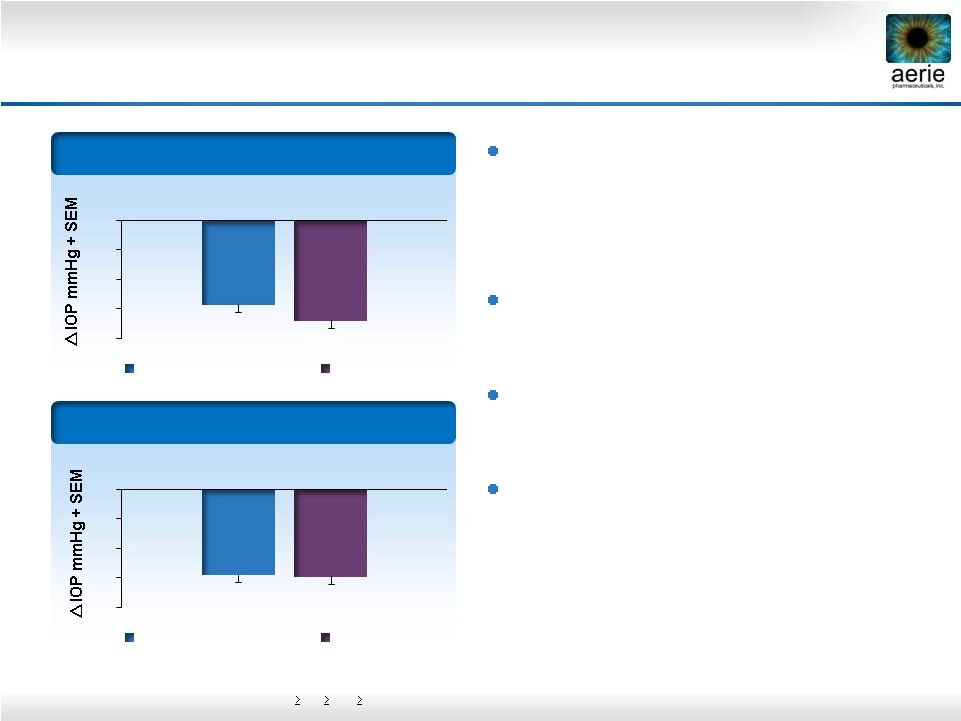 12 Rhopressa™ Differentiated Efficacy Profile Phase 2b baseline IOP entry requirements: 24, 22, 22 mmHg (8 AM, 10 AM, 4 PM) Rhopressa™ and latanoprost clinically and statistically equivalent in patients with moderately elevated IOPs of 22-26 mmHg Latanoprost loses ~1 mmHg efficacy @ baselines of 22-26 mmHg Rhopressa™ maintains consistent efficacy Rhopressa™; 1 st product to treat the diseased tissue (trabecular meshwork) with a once-daily (QD) dose Baseline: 22 – 36 mmHg (n=221) Baseline: 22 – 26 mmHg (n=106) -6.8 -8 -6 -4 -2 0 0.02% Rhopressa™ Latanoprost -5.8 -5.9 -8 -6 -4 -2 0 0.02% Rhopressa™ Latanoprost -5.7 |
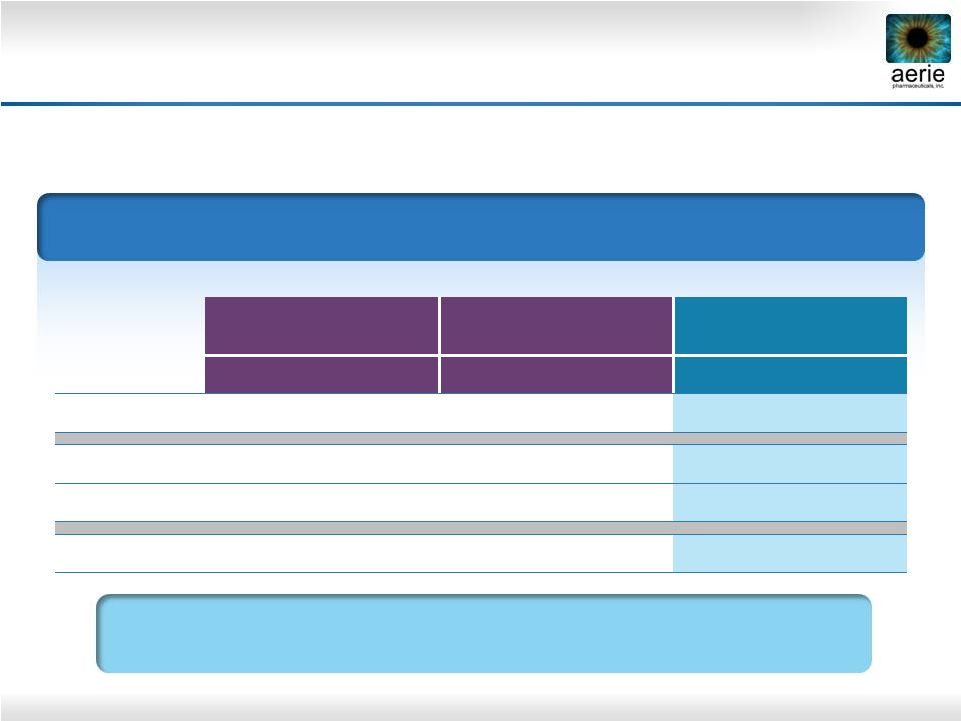 13 Sustained Effect of Rhopressa™ vs. Latanoprost * 36 hours post dose 8 AM Mean IOP (mmHg) by Treatment Group 0.02% Rhopressa ™ Roclatan™ Phase 2b 0.02% Rhopressa ™ Rhopressa™ Phase 2b 0.005% latanoprost Rhopressa™ Phase 2b (n = 78) (n = 71) (n = 76) Baseline 26.6 27.2 26.8 Day 8 20.0 21.1 20.0 Day 29 20.3 21.2 19.2 Day 30* 21.0 22.2 22.4 Rhopressa™ Duration is Superior to Latanoprost 36 Hours After Last Dose |
 14 Rhopressa™ EVP-Lowering Breakthrough Phase 2b data provided first sign of EVP-lowering: Phase 1 study in low baseline IOP subjects: Preclinical in vivo study: Note: Timolol and latanoprost reported to have no effect or to increase EVP in animal models Consistent Efficacy Across Baseline IOPs Lowered Average IOP by Over 30% From 16 Down to 11 mmHg Lowered EVP by 35% |
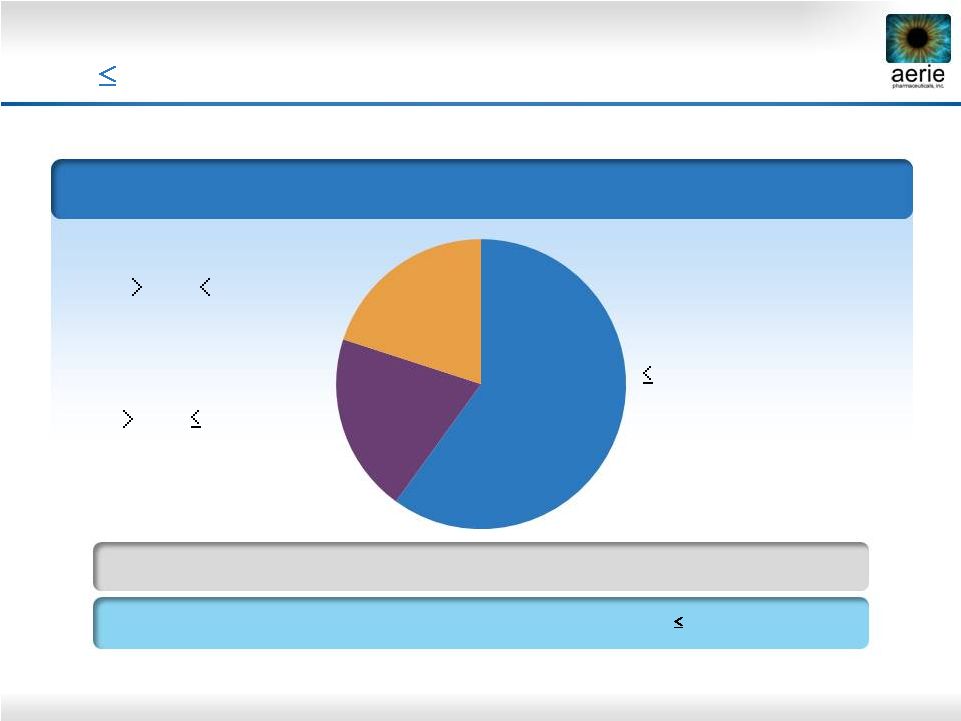 15 Baseline IOP* ~80% of U.S. Glaucoma Patients Have IOPs that are 26 mmHg at Time of Diagnosis The Baltimore Eye Survey * Sommer A, Tielsch JM, Katz J et al. Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans: The Baltimore eye survey. Arch Ophthalmol 1991;109:1090-1095 ** IWASE et al Tajimi study group. Japan Glaucoma Society. Ophthalmology, 2004 Sep, 111 (9): 1641-8. 21 mmHg (Normal Tension Glaucoma) 21 - 26 mmHg 26 - 35 mmHg 10,444 Individuals Were Screened for the Prevalence of Primary Open-Angle Glaucoma (POAG) and the IOP at Time of Diagnosis 92% of Japanese Patients with POAG, IOPs Were 21 mmHg** 20% 20% 60% |
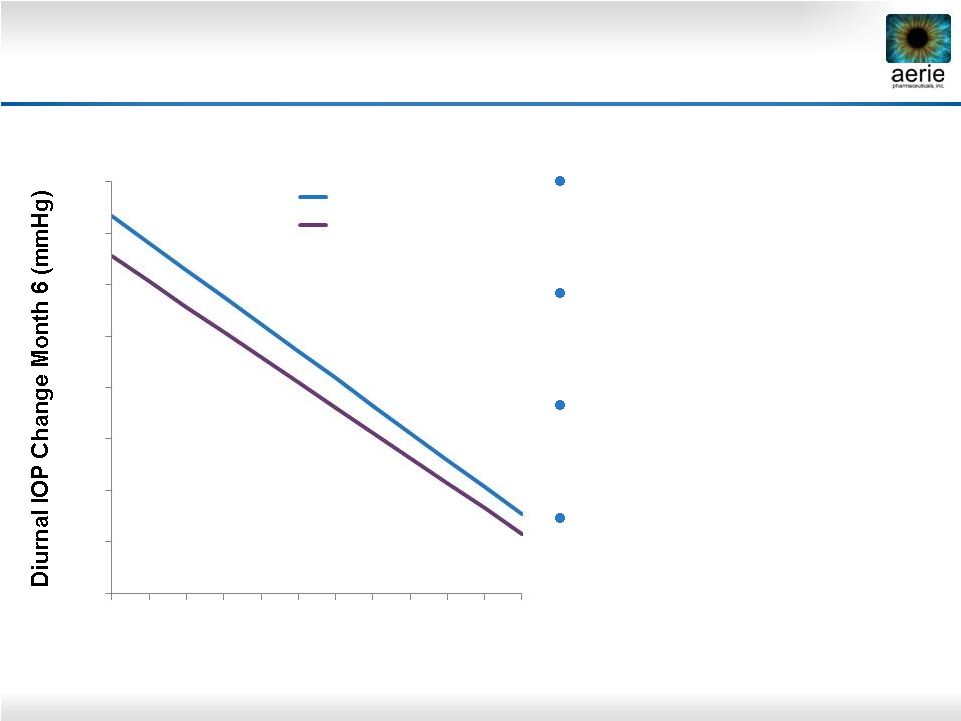 16 Latanoprost and Timolol Show Reduced Efficacy at Lower Baseline IOPs Latanoprost and timolol lose efficacy as baseline IOPs decline Timolol at least 1 mmHg less effective than latanoprost across all published baselines Rhopressa™ equivalent/ non-inferior to latanoprost at baselines 22–26 mmHg Timolol is the standard comparator for glaucoma Phase 3 trials -16 -14 -12 -10 -8 -6 -4 -2 0 16 18 20 22 24 26 28 30 32 34 36 38 Untreated Diurnal IOP (mmHg) Timolol (n=369) Latanoprost (n=460) Pooled data from three latanoprost registration studies. Hedman and Alm; European Journal Ophthalmology; 2000 |
 17 Rhopressa™ Registration Trial Overview Primary efficacy endpoint: IOP at nine time points through Day 90 Phase 3 entry IOP is >20 mmHg and <27 mmHg Non-inferiority design vs. timolol 95% CI within 1.5 mmHg at all time points, within 1.0 mmHg at a majority of time points Combined trials to include approximately 1,300 total patients 100 patients with 12 months of safety data needed for NDA filing Should meet efficacy requirements for EMA filing 300 patients with 6 months safety data needed for EMA filing and 100 with 12 months |
 18 Rhopressa™ Registration Trial Design * * PGAs have been shown to be less effective when dosed BID PGAs have been shown to be less effective when dosed BID “Rocket 1” 90-Day Efficacy Registration Trial Rhopressa™ 0.02% QD ~200 patients Timolol BID ~200 patients “Rocket 2” One Year Safety (3 Mo. Interim Efficacy) Registration Trial “Rocket 3” One Year Safety Registration Trial Canada Rhopressa™ 0.02% QD ~230 patients Rhopressa™ 0.02% BID * ~230 patients Timolol BID ~230 patients Rhopressa™ 0.02% QD ~90 patients Rhopressa™ 0.02% BID ~90 patients Timolol BID ~60 patients |
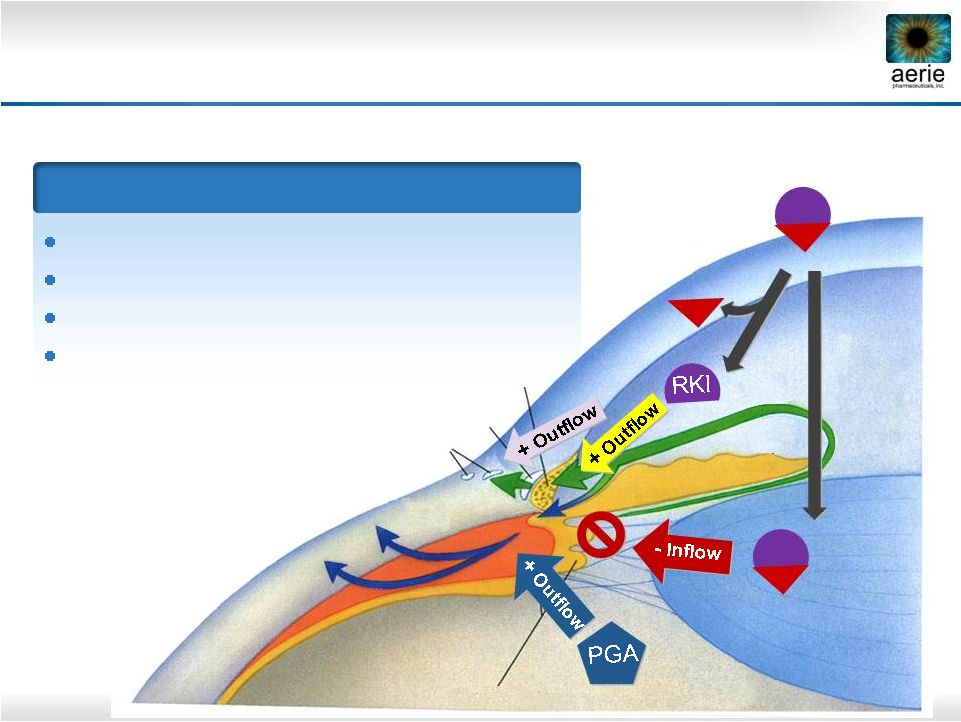 19 Ciliary Processes Cornea Uveoscleral Outflow NET RKI NET RKI Trabecular Meshwork Episcleral Veins Schlemm’s Canal Latanoprost Rhopressa ™ Quadruple-Action Roclatan™ Fixed Combination of Rhopressa™ with Latanoprost IOP-Lowering Mechanisms ROCK inhibition relaxes TM, increases outflow NET inhibition reduces fluid production ROCK inhibition lowers EVP PGA receptor activation increases uveoscleral outflow |
 20 Roclatan™ Phase 2b Clinical Trial Design Phase 2b Protocol Roclatan™ 0.01% vs. Roclatan™ 0.02% vs. Rhopressa™ 0.02% vs. Latanoprost All Dosed QD PM ~300 Patients 28 Days Primary efficacy endpoint: Mean diurnal IOP on Day 29 Two concentrations of Roclatan™ vs. Rhopressa™ 0.02% and latanoprost Trial design follows FDA requirement for fixed-dose combination Statistically significant difference at measured time points Higher combo efficacy vs. components of at least 1–3 mmHg, as previously accepted by FDA for product approval |
 21 Roclatan™ Phase 2b Clinical Trial Performance Achieved primary efficacy measure Superiority over each of the components on day 29 Achieved statistical superiority over the individual components at all time points More efficacious than latanoprost by 1.6 – 3.2 mmHg More efficacious than Rhopressa™ by 1.7 – 3.4 mmHg Main adverse event was hyperemia (eye redness): Reported in 40 percent of patients Mild for the large majority of patients No systemic drug-related adverse events |
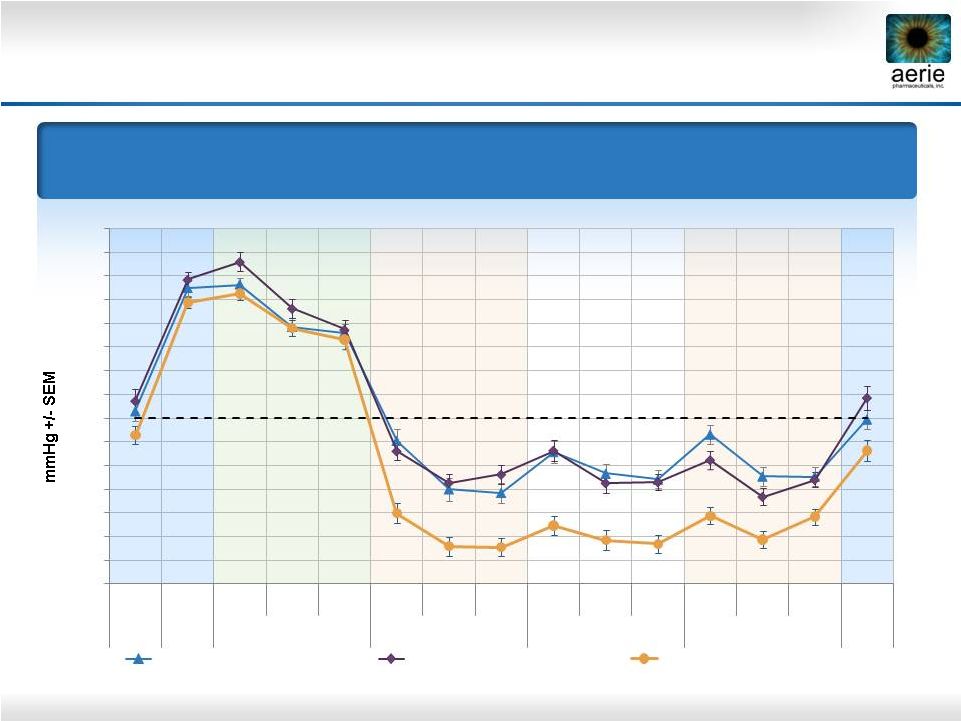 22 Mean IOP at Each Time Point Primary Efficacy Measure 0.02% Roclatan™ Achieved Statistical Superiority Over Individual Components at All Time Points (p<0.001) Roclatan™ Phase 2b, Intent to Treat 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 Pre- 8AM 8AM 10AM 4PM 8AM 10AM 4PM 8AM 10AM 4PM 8AM 10AM 4PM 8AM Study Qual 1 Baseline Day 8 Day 15 Day 29 Day 30 0.02% Rhopressa™ (n=78) 0.005% Latanoprost (n=73) 0.02% Roclatan™ (n=72) |
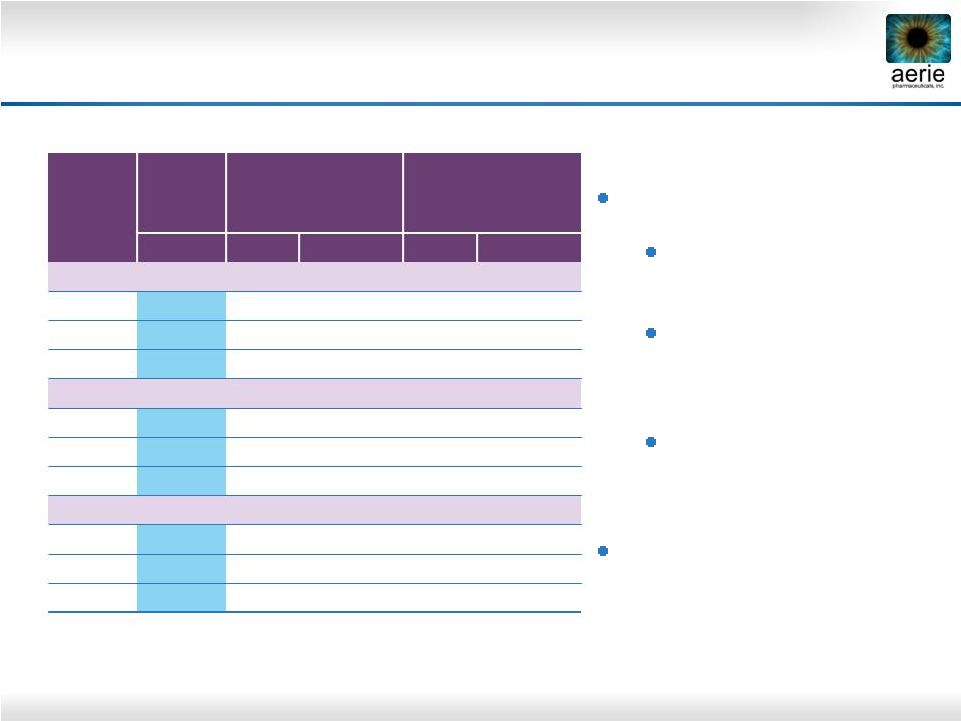 23 Roclatan™ Phase 2b, ITT Mean IOP (mmHg) * Difference between 0.02% Roclatan™ and latanoprost or Rhopressa™ Roclatan™: Produced lowest IOP drop in any trial Was superior to latanoprost by 1.6–3.2 mmHg (p<0.001) Was superior to Rhopressa™ by 1.7–3.4 mmHg (p<0.001) Impressive Rhopressa™ performance 0.02% Roclatan™ (n = 72) 0.005% latanoprost (n = 73) 0.02% Rhopressa™ (n = 78) Mean Mean Difference* Mean Difference* Day 8 8 AM 17.0 19.6 -2.6 20.0 -3.1 10 AM 15.6 18.3 -2.7 18.0 -2.4 4 PM 15.6 18.6 -3.1 17.9 -2.3 Day 15 8 AM 16.5 19.6 -3.2 19.6 -3.1 10 AM 15.8 18.3 -2.4 18.7 -2.8 4 PM 15.7 18.3 -2.6 18.4 -2.7 Day 29 8 AM 16.9 19.2 -2.4 20.3 -3.4 10 AM 15.9 17.7 -1.8 18.6 -2.7 4 PM 16.8 18.4 -1.6 18.5 -1.7 |
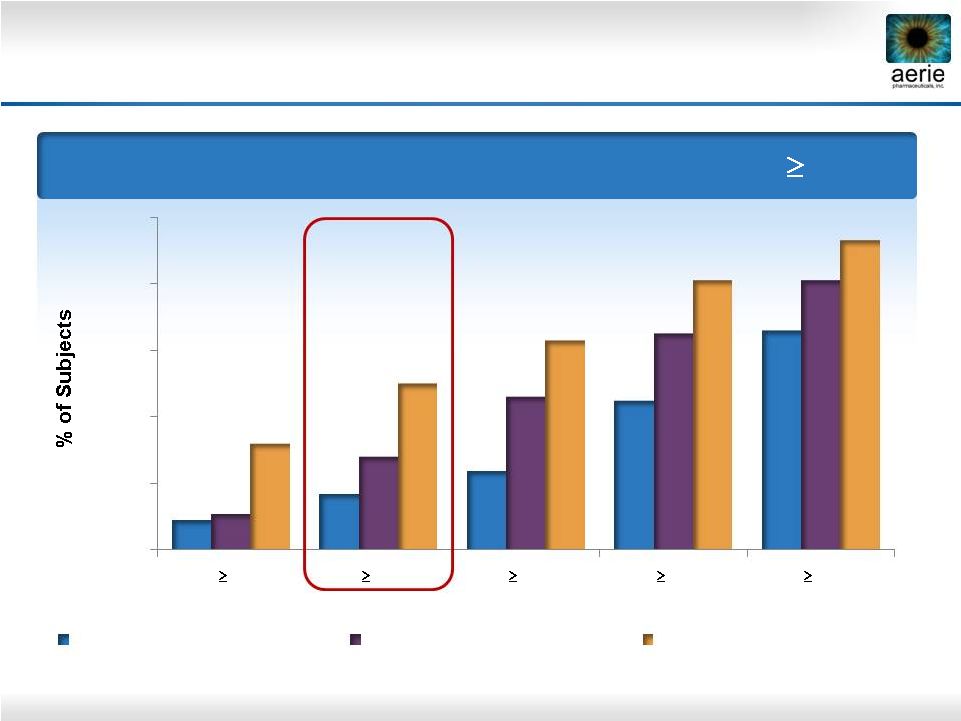 24 Day 29 – % of Patients with IOP Reductions of 20% Roclatan™ Phase 2b Responder Analysis: Goal is to Achieve Lowest IOP Possible 0% 20% 40% 60% 80% 100% 40% 35% 30% 25% 20% 9% 17% 24% 45% 66% 11% 28% 46% 65% 81% 32% 50% 63% 81% 93% Reduction 0.02% Rhopressa™ (n=78) 0.005% Latanoprost (n=73) 0.02% Roclatan™ (n=72) |
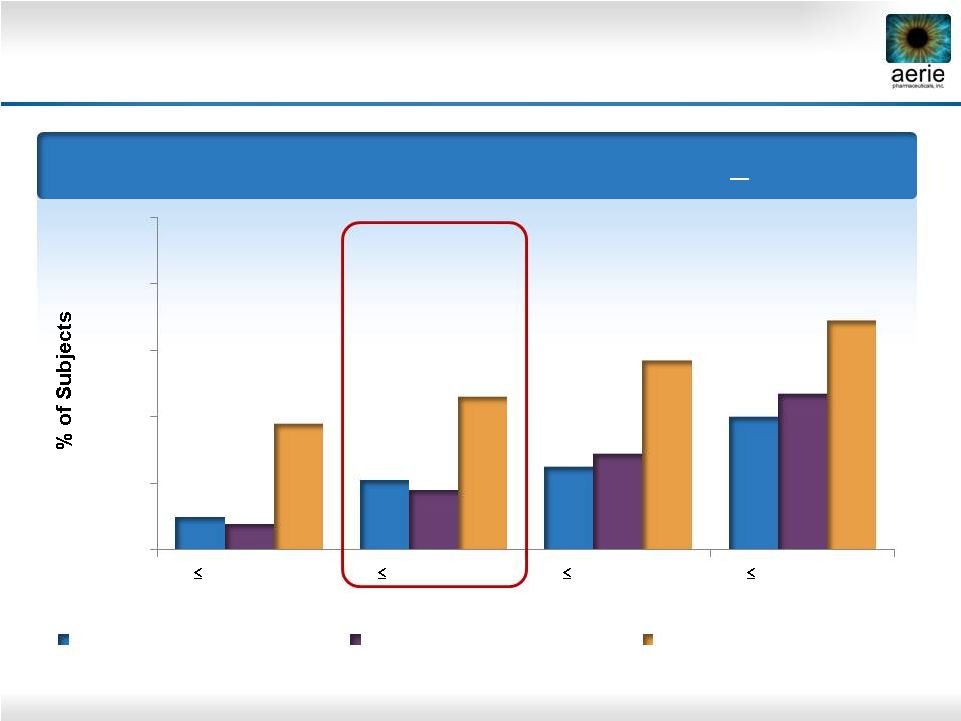 25 Roclatan™ Phase 2b Responder Analysis: Goal is to Achieve Lowest IOP Possible Day 29 – % of Subjects with IOP Reduced to < 18 mmHg 0% 20% 40% 60% 80% 100% 10% 21% 25% 40% 8% 18% 29% 47% 38% 46% 57% 69% Reduction 0.02% Rhopressa™ (n=78) 0.005% Latanoprost (n=73) 0.02% Roclatan™ (n=72) 15 mmHg 16 mmHg 17 mmHg 8 mmHg |
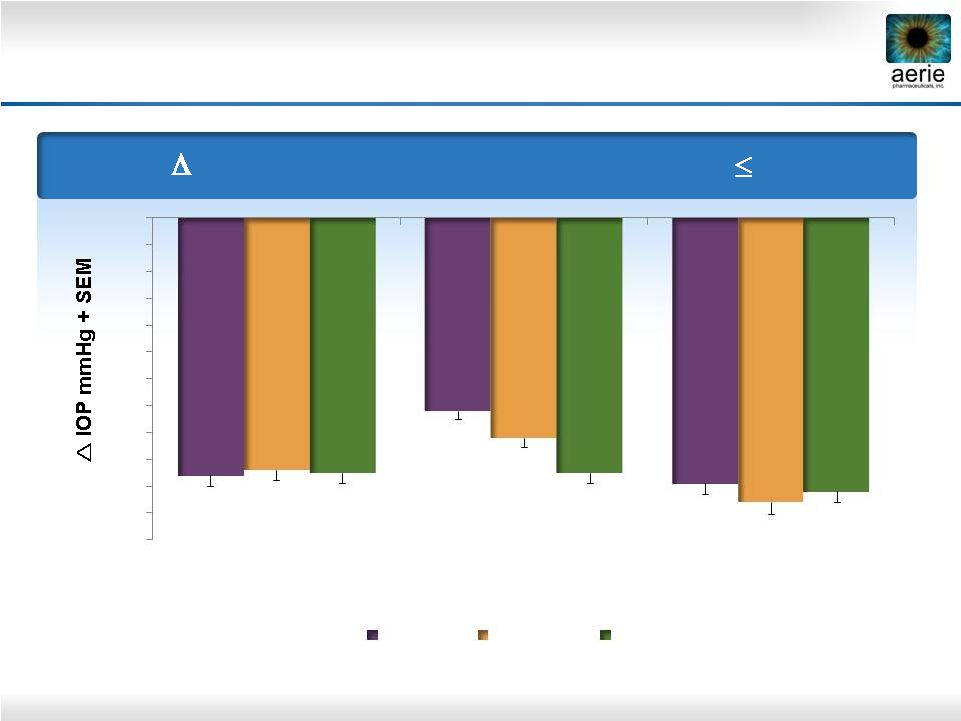 26 Diurnal IOP in Subset Of High Responders 16 mmHg Roclatan™ Phase 2b High Responders: Consistent IOP Drop by Rhopressa™ and Roclatan™ -9.6 -7.2 -9.9 -9.4 -8.2 -10.6 -9.5 -9.5 -10.2 -12 -11 -10 -9 -8 -7 -6 -5 -4 -3 -2 -1 0 0.02% Rhopressa™ (n=16) 0.005% Latanoprost (n=13) 0.02% Roclatan™ (n=31) Day 8 Day 15 Day 29 |
 27 2014 2015 2016 2017 2018 Key Future Milestones June 2014: Roclatan™ Phase 2b clinical trial completed Mid-2015: Roclatan™ Phase 3 to be initiated Mid-2016: Roclatan™ Efficacy results from Phase 3 expected Mid-2017: Roclatan™ NDA filing expected 2H 2018: Roclatan™ Launch expected 3Q 2014: Rhopressa™ Phase 3 initiated Mid-Q2 2015: Rhopressa™ Efficacy results from Phase 3 expected Mid-2016: Rhopressa™ NDA filing expected 2H 2017: Rhopressa™ Launch expected Nearly $200M raised - adequate financing to fund through commercialization |
 28 New Preclinical Research |
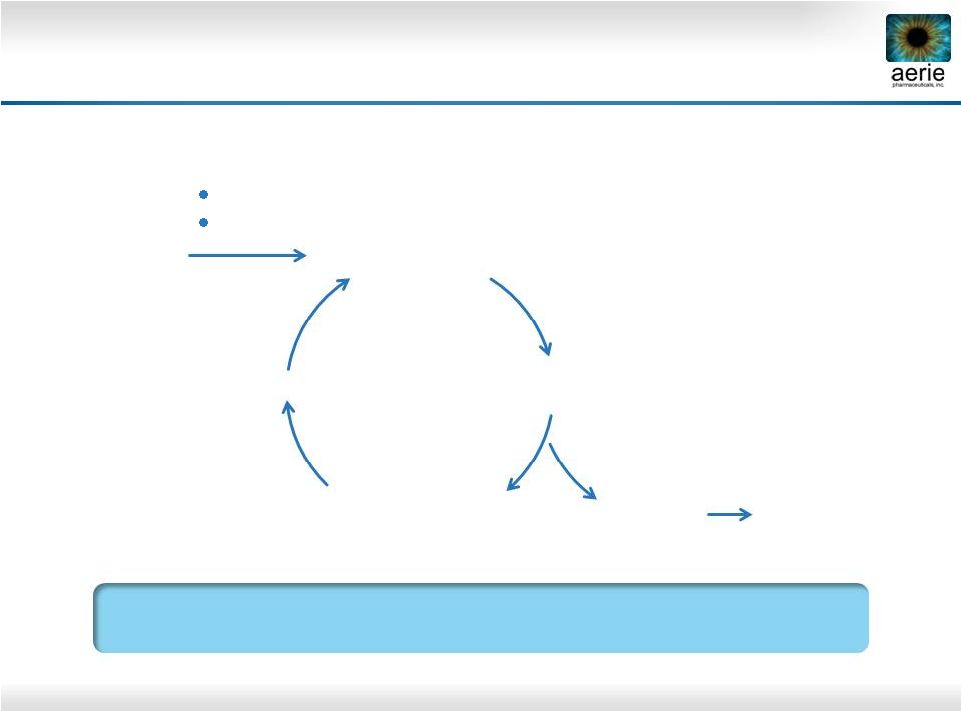 29 Disease Modification: Addressing the Root Cause of Elevated IOP and Vision Loss Healthy TM Less Nutrients, Antioxidants Cellular Stress Fibrosis, Stiffness Contraction Reduced Aqueous Perfusion Area Elevated IOP Cellular Stress Progressive Degeneration of the Trabecular Meshwork Drives Elevated IOP and Vision Loss in Glaucoma Vision Loss Oxidation Aging |
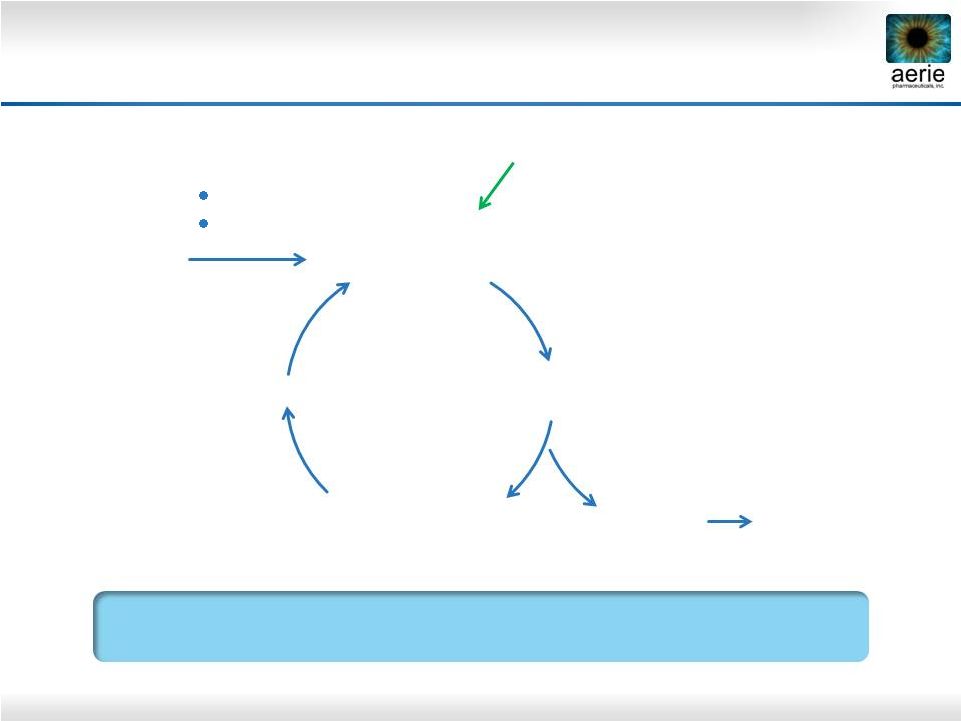 30 Rhopressa™ Has Potential to Improve Health of TM in Patients with Glaucoma Healthy TM More Nutrients, Antioxidants Less Cellular Stress Reduced Fibrosis, Stiffness Contraction Increased Aqueous Perfusion Area Reduced IOP Cellular Stress Reducing Fibrosis, Increasing Trabecular Outflow Could Stop Degeneration of Outflow Tissues in POAG + Rhopressa™ Preserve Vision Oxidation Aging |
 31 AR-13324* May Have Anti-Fibrotic Activity in Human Trabecular Meshwork Cells Control TGF 2 (8 ng/ml) + AR-13324 (500nM) Vasanth Rao, Duke University • Active ingredient of Rhopressa™ • TGF 2: Transforming growth factor 2; SMA: Smooth muscle actin; FSP1: Fibroblast-specific protein 1 AR-13324 May Block TGF-beta-Induced Expression of Fibrosis Proteins in Human TM Cells TGF 2 (8ng/ml) |
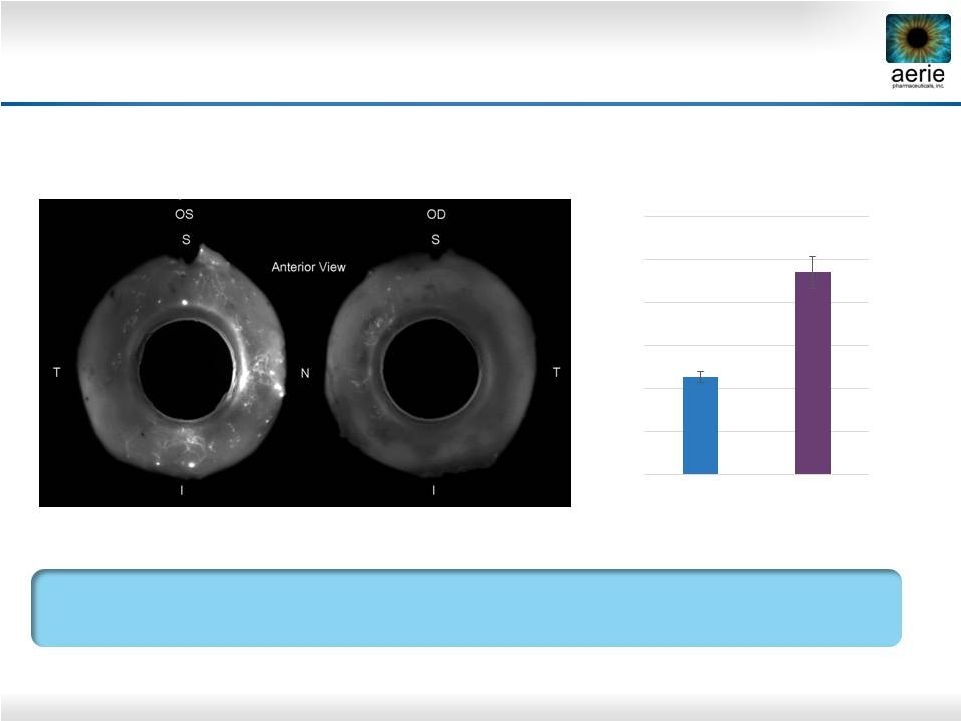 32 + AR-13324 Control AR-13324* May Increase Perfusion of TM Outflow Tissues Dan Stamer (Duke), Haiyan Gong (Boston University) AR-13324’s Ability to Potentially Increase Perfusion of TM Should Provide More Nutrients and Antioxidants to the TM Left (OS) and right (OD) eyes perfused with fluorescent microbeads Control = buffered saline solution *Active ingredient of Rhopressa™ ** Percent Effective Filtration Area 0.00 10.00 20.00 30.00 40.00 50.00 60.00 Anterior PEFA** (%) AR-13324 Control |
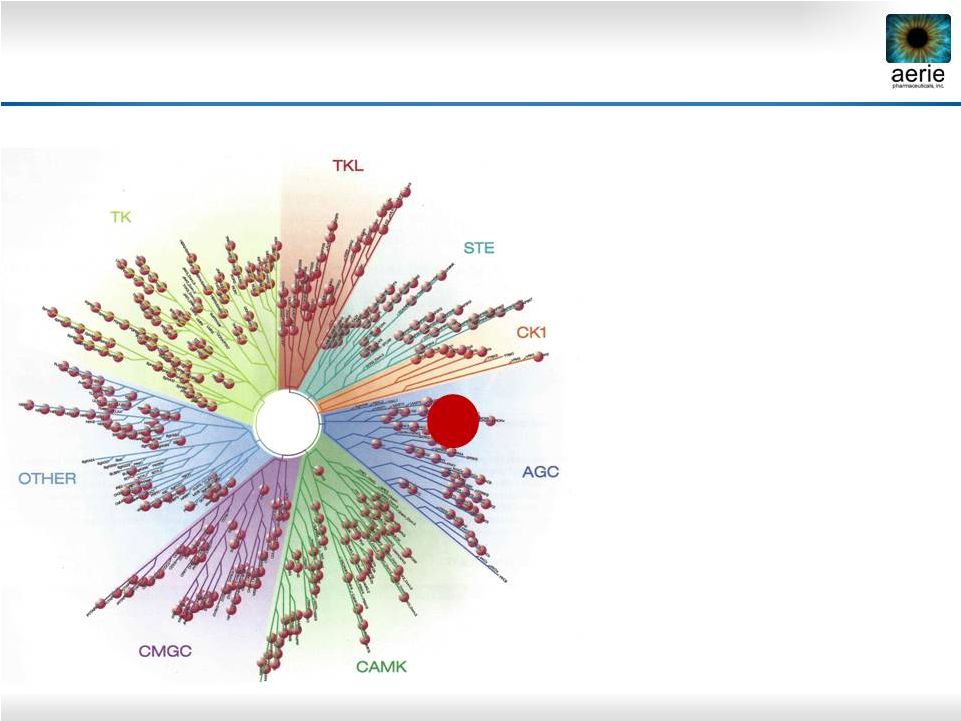 33 Selective Multi-Kinase Inhibitors for AMD/DME • Selective Rho kinase inhibitors previously effective in preclinical models of AMD and DME • Compounds inhibiting multiple disease targets should provide best efficacy • Inflammation • Angiogenesis • Fibrosis • Screened 184 Aerie compounds against 456 human kinases Aerie Kinase Library Screen Relationship tree of human kinases. TK, TKL, STE, CK1, AGC, CAMK, CMGC, Other: Kinase superfamilies ROCK |
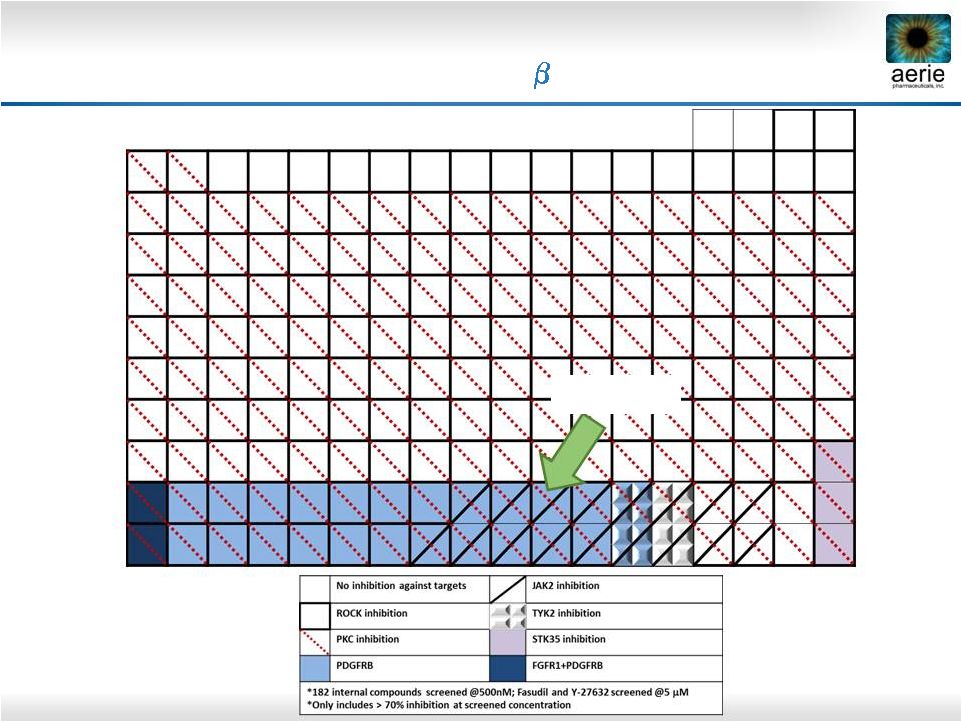 34 KINOMEscan Results: AR-13154 Inhibits AMD Targets ROCK, JAK2, PDGFR- AR-13154 |
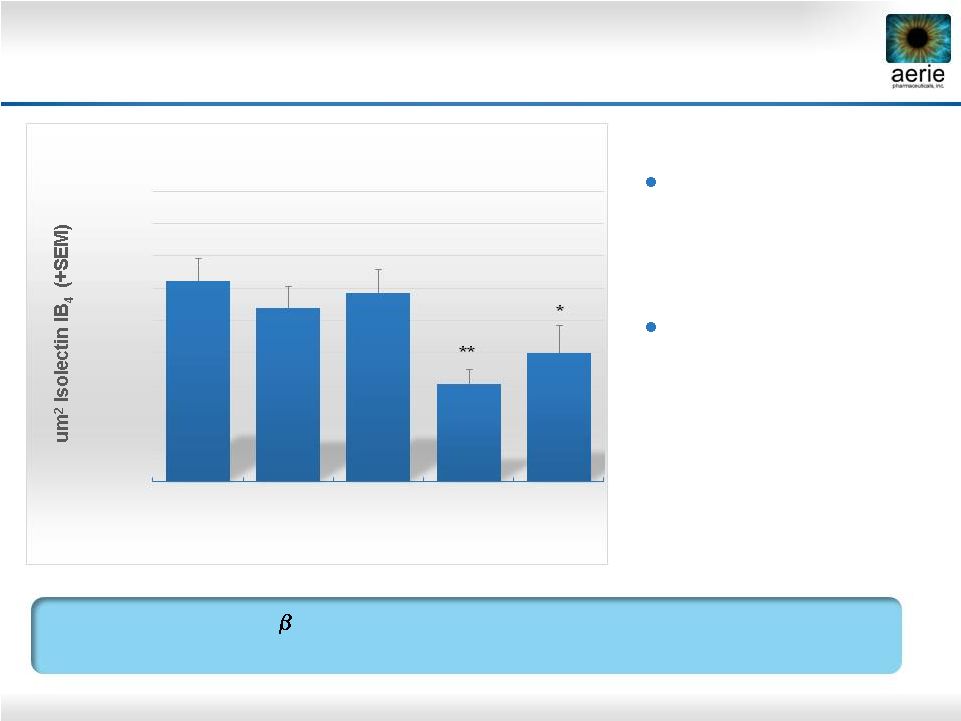 35 Laser-induced choroidal neovascularization (CNV) in rats Compounds delivered by intravitreal injection AR-13154 vs. Eylea in Preclinical AMD Model ROCK/JAK2/PDGFR Inhibitor AR-13154 Numerically More Effective than Eylea ® in Rat Model of AMD 20000 30000 40000 50000 60000 70000 80000 90000 100000 110000 Saline n=49 0.06 ug/mL AR -13154 n=28 0.6 ug/mL AR -13154 n=25 6 ug/mL AR -13154 n=25 800 ug/mL Eylea® n=20 Total CNV Lesion Area (Day 21) ** * * p<0.05 vs. Saline ** p<0.001 |
 Building a Major Ophthalmic Pharmaceutical Company |