 Company Overview January 2017 Exhibit 99.1 |
 2 Important Information Any discussion of the potential use or expected success of our product candidates is subject to our product candidates being approved by regulatory authorities. In addition, any discussion of clinical trial results for Rhopressa™ (netarsudil ophthalmic solution) 0.02% relate to the results in its first Phase 3 registration trials named Rocket 1 and Rocket 2, or Rocket 4 which will be used primarily for European regulatory filing purposes, and for Roclatan™ (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% relate to the results in its Phase 3 registration trial. The information in this presentation is current only as of its date and may have changed or may change in the future. We undertake no obligation to update this information in light of new information, future events or otherwise. We are not making any representation or warranty that the information in this presentation is accurate or complete. Certain statements in this presentation, including the updated guidance presented herein, are “forward-looking statements” within the meaning of the federal securities laws. Words such as “may,” “will,” “should,” “would,” “could,” “believe,” “expects,” “anticipates,” “plans,” “intends,” “estimates,” “targets,” “projects,” “potential” or similar expressions are intended to identify these forward-looking statements. These statements are based on the Company’s current plans and expectations. Known and unknown risks, uncertainties and other factors could cause actual results to differ materially from those contemplated by the statements. In evaluating these statements, you should specifically consider various factors that may cause our actual results to differ materially from any forward-looking statements. Any top line data presented herein is preliminary and based solely on information available to us as of the date of this document and additional information about the results may be disclosed at any time. In addition, the preclinical research discussed in this presentation is preliminary and the outcome of such preclinical studies may not be predictive of the outcome of later trials. Any future clinical trial results may not demonstrate safety and efficacy sufficient to obtain regulatory approval related to the preclinical research findings discussed in this presentation. These risks and uncertainties are described more fully in the quarterly and annual reports that we file with the SEC, particularly in the sections titled “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” Such forward-looking statements only speak as of the date they are made. We undertake no obligation to publicly update or revise any forward-looking statements, whether because of new information, future events or otherwise, except as otherwise required by law. |
 3 Aerie Late Stage IOP–Lowering Products Pre-Clinical Research • Rhopressa™ • Potential for disease modification and neuroprotection • AR-13154 • Significant lesion size reduction in wet AMD • Drug Delivery - Front and back of the eye • Rhopressa™ (netarsudil ophthalmic solution) 0.02% • Inhibits ROCK, NET, lowers EVP, targets diseased tissue • NDA re-filing expected near end of Q1 2017; entering launch mode • Roclatan™ (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% • Fixed combination of Rhopressa™ and latanoprost • Two P3’s in process; first P3 achieved primary efficacy endpoint Aerie – Building a Major Ophthalmic Pharmaceutical Company Data on file |
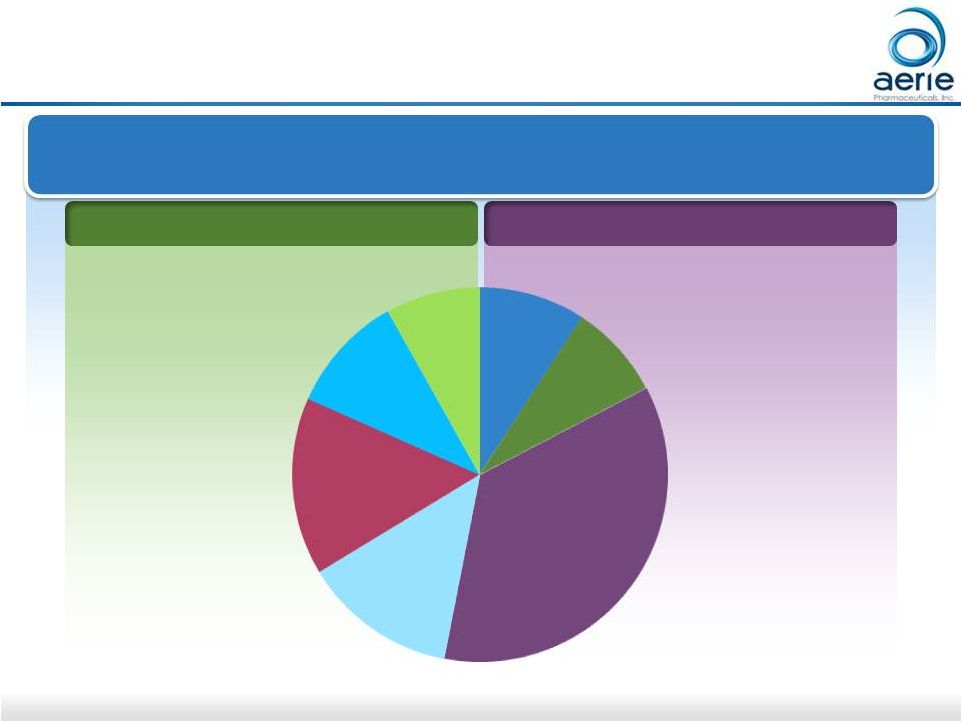 4 FY 2015 U.S. Glaucoma Market = $2.5B; 34M TRx Market Share in TRx PGA Market Non-PGA Market 2015 US Glaucoma Market Once Daily 2-3 Times Daily 9% 8% 35% 13% 15% 10% 8% Bimatoprost Travoprost Latanoprost BB Fixed Combo AA CAI PGA: Prostaglandin Analogue; BB: Beta Blocker; AA: Alpha Agonist; CAI: Carbonic Anhydrase Inhibitor Source: IMS MIDAS. IMS NPA |
 5 ~75% of U.S. Glaucoma Patients Have IOPs that are 25 mmHg at Time of Diagnosis Baseline IOP* The Baltimore Eye Survey 60% 20% 20% 21 mmHg (Normal Tension Glaucoma) 5,308 Individuals Were Screened for the Prevalence of Primary Open-Angle Glaucoma (POAG) and the IOP at Time of Diagnosis 92% of Japanese Patients with POAG, IOPs Were 21 mmHg** * Sommer A, Tielsch JM, Katz J et al. Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans: The Baltimore eye survey. Arch Ophthalmol 1991;109:1090-1095 ** IWASE et al Tajimi study group. Japan Glaucoma Society. Ophthalmology, 2004 Sep, 111 (9): 1641-8. 25 - 34 mmHg 22 - 24 mmHg |
 6 Potential drug of choice as adjunctive therapy to PGAs when additional IOP lowering is desired Potential drug of choice for patients requiring maximal IOP lowering Rhopressa ™ Positioning Roclatan ™ Positioning Rhopressa TM and Roclatan TM Potential Product Advantages Rhopressa ™ Advantages • Efficacy vs. other adjunctive therapies • QD PM dose • Lack of serious and systemic AE’s Roclatan ™ Advantages • Efficacy vs. all other glaucoma therapies • QD PM dose • Lack of serious and systemic AE’s |
 7 ++ Data on file from Aerie Phase 3 clinical trials, Rocket 1, Rocket 2, Rocket 4 and Mercury 1. Value of Rhopressa TM ++ • Directed at site of pathology • The trabecular meshwork • Enhanced compliance • Once-a-day dosing • Efficacy • Achieves noninferiority to timolol where AA’s (Brimonidine) and CAI’s failed • Consistent IOP lowering across broad baseline range of Mercury 1 • Additive to prostaglandins • Safety • Lack of serious and systemic adverse events |
 8 **Rhopressa TM achieves non-inferiority to timolol at primary endpoint range of > 20 mmHg to < 25 mmHg *Rhopressa TM achieves non-inferiority to timolol at pre-specified secondary endpoint range of > 20 mmHg to < 24 mmHg; also non-inferior at < 25 mmHg Rhopressa™ Trials for NDA Resubmission Near End of Q1 2017 “Rocket 1” 90-Day Efficacy* Registration Trial U.S. “Rocket 1” 90-Day Efficacy* Registration Trial U.S. Rhopressa™ 0.02% QD 202 patients timolol BID 209 patients (Total enrollment: 411 patients) “Rocket 2” One Year Safety (3 Mo. Interim Efficacy**) Registration Trial U.S. “Rocket 2” One Year Safety (3 Mo. Interim Efficacy**) Registration Trial U.S. Rhopressa™ 0.02% QD 251 patients Rhopressa™ 0.02% BID 254 patients timolol BID 251 patients (Total enrollment: 756 patients) 1 Post-hoc analysis ClinicalTrials.gov Identifier: NCT02207491, NCT02207621 1 |
 9 Rhopressa™ Phase 3 Trial Data to Supplement NDA Filing ClinicalTrials.gov Identifier: NCT02246764, NCT02558374 “Rocket 4” 3 Month Efficacy* 6 Month Safety “Rocket 4” 3 Month Efficacy* 6 Month Safety Rhopressa™ 0.02% QD 351 patients timolol BID 357 patients *Rhopressa™ achieves non-inferiority to timolol at primary endpoint range of > 20 mmHg to < 25 mmHg; also non-inferior at pre-specified range up to < 28 mmHg “Mercury 1” One Year Safety (3 Mo. Interim Efficacy**) Registration Trial “Mercury 1” One Year Safety (3 Mo. Interim Efficacy**) Registration Trial Roclatan™ QD 238 patients Rhopressa™ 0.02% QD 244 patients Latanoprost QD 236 patients **Rhopressa™ achieves non-inferiority to latanoprost at pre-specified secondary endpoint range of > 20 mmHg to < 25 mmHg |
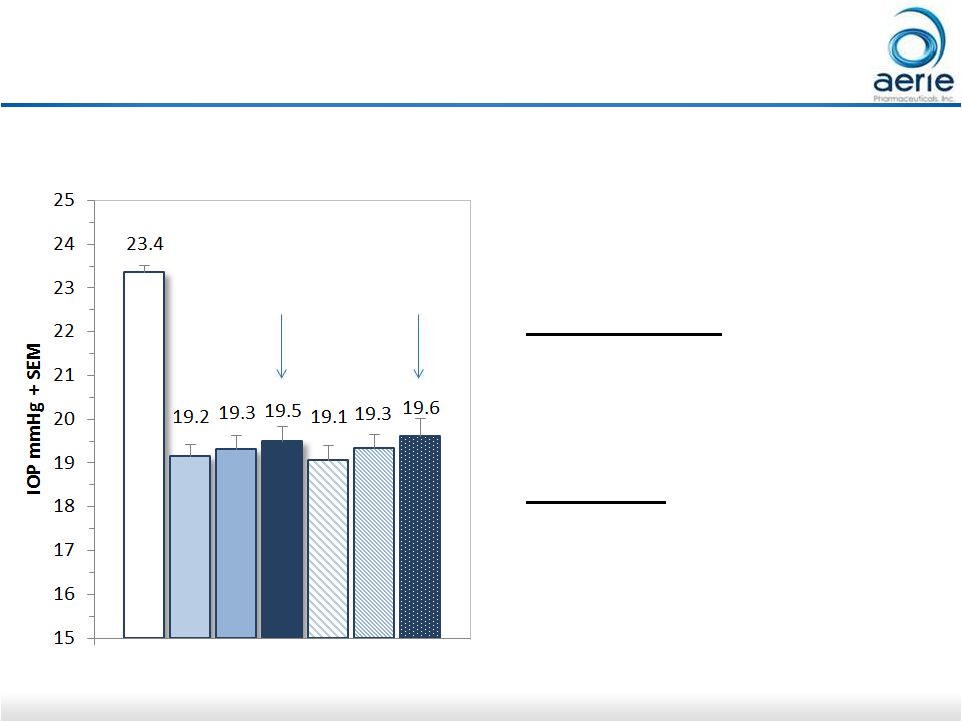 10 Rocket 2: 12 Month 8am IOP Safety Population, Completed Patients (<27 mmHg) • Difference in 8am mean IOP from Day 90 (Month 3) to Month 12: Rhopressa TM QD: • Full population: +0.1 mmHg • Subset analyses: -0.3 to +0.3 mmHg Timolol BID: • Full population: +0.5 mmHg • Subset analyses: +0.1 to +1.4 mmHg Day 90 Month 12 AR-13324 0.02% QD (n=118) BL W2 W6 M3 M6 M9 M12 Data on file: Interim 12-month safety AR-13324 – CS302 |
 11 Rocket 2: Safety/Tolerability Overview of Rhopressa™ QD (Interim 12-Month) * Incidence of conjunctival hyperemia ~50% including baseline at ~20% • There were no drug-related serious adverse events (SAEs) • No new adverse events introduced over the twelve-month period • The most common adverse event was conjunctival hyperemia with ~50% incidence*, the majority mild • Other ocular AEs • AEs occurring in ~5-23% of patients included: conjunctival hemorrhage, corneal verticillata, blurry vision and reduced visual acuity |
 12 Rhopressa™ QD Adverse Events Summary Data on File: Interim 12-month safety AR-13324-CS302 Hyperemia – absent or sporadic for ~90% of patients ~10% of patients had hyperemia AE at all 6 study visits Conjunctival Hemorrhage – sporadic subconjunctival petechiae - none noted at month 12 visit Corneal Verticillata – asymptomatic non-toxic lipid deposits - high resolution rate Visual Acuity Reduced – sporadic, mostly single visit, only one eye - incidence reduced over time Vision Blurred – sporadic and significantly reduced over 12 months |
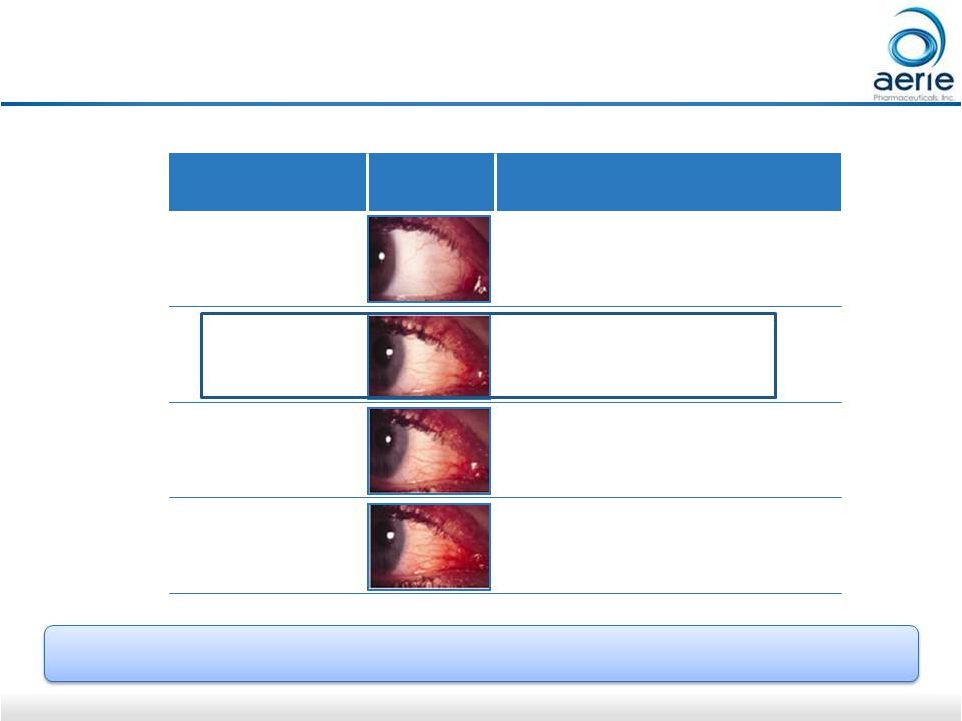 When Present, 80% of Rhopressa QD Hyperemia Graded as Mild* Data on File: AR-13324 – CS302 TM Grade Image Description 0 None/Normal 1 Mild 2 Moderate 3 Severe For illustrative purposes only 13 |
 14 Rhopressa™ 24-hour IOP Pilot Study Demonstrates Effective Nocturnal Efficacy (n=8) (n=8) ** ** ** *** ** ** *** *** ** p<0.01 *** p<0.001 • Netarsudil (active ingredient of Rhopressa™) equally effective during nocturnal and diurnal periods • Current glaucoma medications either have no efficacy at night (beta blockers, alpha agonists) or reduced efficacy at night (PGAs, CAIs) 1 - 6 Data on File: AR-13324-CS204 1. Liu JH, et al. Am J Ophthalmol. 2004; 138:389-395. 2. Gulati V, et al. Arch Ophthalmol. 2012; 130:677-684. 3. Liu JH, et al. Ophthalmology. 2009; 116:449- 454. 4. Liu JH, et al. Ophthalmology. 2010; 117:2075-9. 5. Fan S et al. J Glaucoma. 2014; 23:276-81. 6. Liu JH, et al. Am J Ophthalmol. 2016;169:249-257. |
 Roclatan™ Registration Trial Design Roclatan™ QD ~230 patients Rhopressa™ 0.02% QD ~230 patients latanoprost QD ~230 patients Roclatan™ QD ~230 patients Comparator (TBD) ~230 patients * ClinicalTrials.gov Identifier: NCT02558400 ** ClinicalTrials.gov Identifier: NCT02674854 Roclatan™ QD 238 patients Rhopressa™ 0.02% QD 244 patients latanoprost QD 236 patients “Mercury 1”* One Year Safety (3 Mo. Interim Efficacy) Registration Trial U.S. “Mercury 2”** 90-Day Efficacy Registration Trial U.S. and Canada “Mercury 3” 6 Mo. Efficacy and Safety Registration Trial Europe 15 |
 16 • Roclatan™ met the criteria for demonstrating statistical superiority over both latanoprost and Rhopressa™ for the primary efficacy analysis • IOP-lowering effect of Roclatan™ was greater (1-3 mmHg) than monotherapy with either latanoprost or Rhopressa™ throughout the duration of the study • Roclatan reduced mean diurnal IOPs to 16 mmHg or lower in 61% of patients, a significantly higher percentage than observed in the comparator arms • The Rhopressa arm achieved non-inferiority to latanoprost in the pre-specified range of >20 mmHg to <25 mmHg Mercury 1: Roclatan Achieves Primary Clinical Endpoint Data on file and PG324 - CS301 Top Line Data TM TM TM |
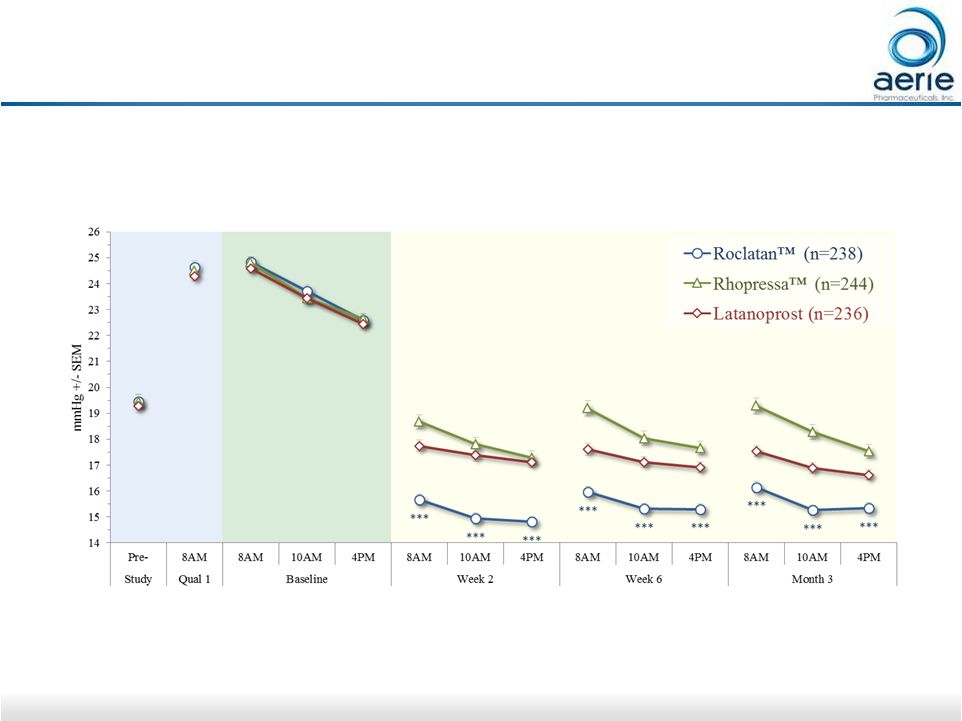 17 Roclatan™ Achieved Statistical Superiority Over Individual Components at All 9 Time Points Mean IOP at Each Time Point (ITT) ***p<0.0001 vs Latanoprost and Rhopressa™ ++ Data on File Based on Mercury 1 Topline Interim 3-month |
 18 Mercury 1: Roclatan Phase 3 Responder Analysis *** *** *** ***p<0.0001 vs Latanoprost and Rhopressa ### p<0.0001 vs Rhopressa , p<0.05 vs Latanoprost Data on file and PG324 - CS301 Top Line Data Day 90: % of Patients with IOP Reduced to 18 mmHg or Lower 14% 23% 32% 42% 54% 15% 25% 39% 54% 69% 33% 44% 61% 71% 82% 0% 20% 40% 60% 80% 100% 14 mmHg 15 mmHg 16 mmHg 17 mmHg 18 mmHg IOP on Treatment Rhopressa™ (n=198) Latanoprost (n=223) Roclatan™ (n=200) ### ### TM TM TM |
 19 Mercury 1: Safety/Tolerability Overview of Roclatan • There were no drug-related serious adverse events (SAEs) • There was no evidence of treatment-related systemic effects • The most common adverse event was conjunctival hyperemia with ~50% incidence*, ~80% mild on biomicroscopy • Other ocular AEs – AEs occurring in ~5-11% of subjects receiving Roclatan included: conjunctival hemorrhage, eye pruritus, lacrimation increased and cornea verticillata. * Incidence of conjunctival hyperemia ~50% including baseline at ~20% Data on file and PG324 - CS301 Top Line Data TM TM |
 20 Expanding Aerie Franchise to Europe and Japan • Europe • Current clinical plan expected to satisfy EU regulatory requirements (including Rocket 4 for Rhopressa and Mercury 3 for Roclatan TM ) • Establishing KOL relationships in top 5 countries • Currently developing EU commercialization strategy • Expect to file EU MAA for Rhopressa™ near year-end 2017 • Japan • Discussions regarding potential Rhopressa™ out-licensing are ongoing • Also preparing to advance clinical development on our own / with CRO • Conferring with PMDA to confirm path to approval TM |
 21 Advancing Earlier-Stage Pipeline Data on file • Rhopressa™ • Potential for disease modification • AR-13154 • Significant lesion size reduction in wet AMD • Drug Delivery • Rhopressa™: Front of the eye • AR-13154: Back of the eye |
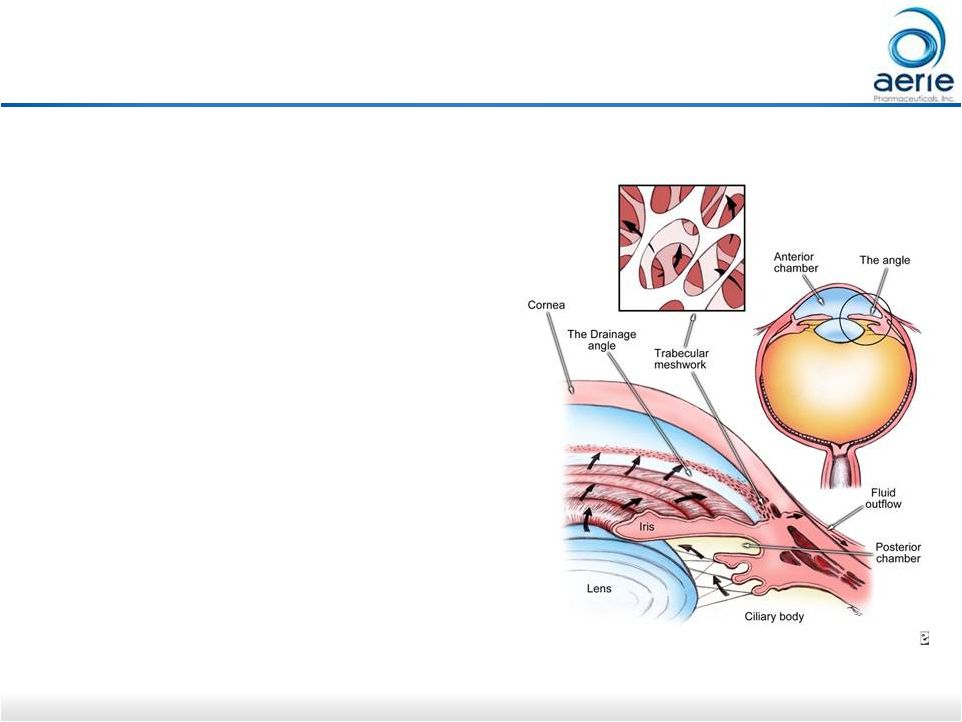
 22 Netarsudil* Causes Expansion of TM Tissue, Opening Spaces for Increased Outflow Dan Stamer (Duke), Haiyan Gong (Boston University) Control + Netarsudil TM: Trabecular Meshwork SC: Schlemm’s Canal Control = buffered saline solution *Active ingredient of Rhopressa™ Source: The Mechanisms of Increasing Outflow Facility by AR-13324M in Human Eyes. Haiyan Gong, Ruiyi Ren, Guorong Li, Casey Kopczynski, Daniel Stamer Increasing Trabecular Outflow, Reducing Fibrosis Could Stop Degeneration of Outflow Tissues in Glaucoma |
 23 Rhopressa TM Blocks TGF-beta-Induced Expression of Fibrosis Proteins in Cultured Human TM Cells P. Vasantha Rao, Duke University • TGF 2: Transforming growth factor 2; SMA: Smooth muscle actin; FSP1: Fibroblast-specific protein 1 • Pattabiraman, Padmanabhan P., et. Al., (2015 AOPT Meeting) “Effects of Rho Kinase inhibitor AR-13324 on actin cytoskeleton and TGF 2 and CTGF- induced fibrogenic activity in Human Trabecular Meshwork Cells”. TGF-beta Levels are Elevated in the Aqueous Humor of Patients with Glaucoma Control TGF 2 (8ng/ml) TGF 2 (8 ng/ml) + AR-13324* (500nM) |
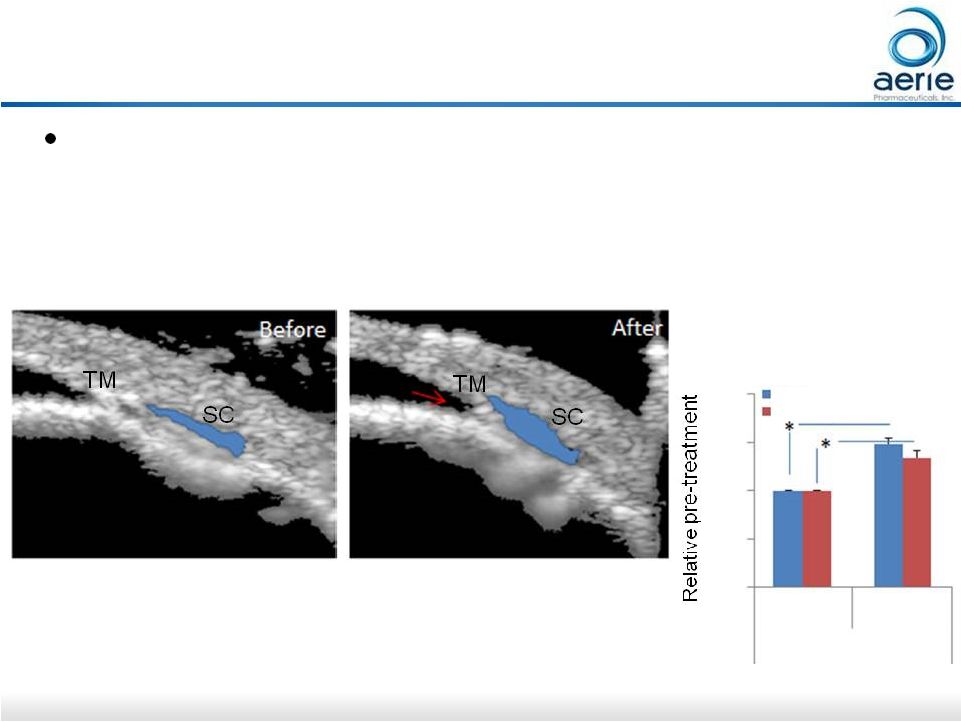 24 Live Imaging of Rhopressa -induced Increase in Trabecular Meshwork Outflow Rhopressa TM -induced increase in trabecular outflow of aqueous humor visualized in real time using live-animal OCT imaging in mice • First study to show glaucoma drug affecting target tissue in real time Rhopressa TM Treatment Blue shading shows Schlemm’s Canal expanding due to enhanced fluid flow through TM Rhopressa TM Li G. et al. Visualization of conventional outflow tissue responses to netarsudil in living mouse eyes. Eur J Pharmacol. 2016 Apr 13. pii: S0014-2999(16)30206-0 before after 0 0.5 1 1.5 2 intensity area TM |
 25 AR-13154 is ROCK Inhibitor with Additional Activity Against PKC, PDGFR and JAK Kinases • 182 Aerie compounds screened against 469 human kinases • AR-13154 identified as lead compound to address: • Vascular dysfunction ROCK, PKC, PDGFR • Fibrosis ROCK, PKC, PDGFR • Inflammation ROCK, JAK Relationship tree of human kinases. TK, TKL, STE, CK1, AGC, CAMK, CMGC, Other: Kinase superfamilies ROCK |
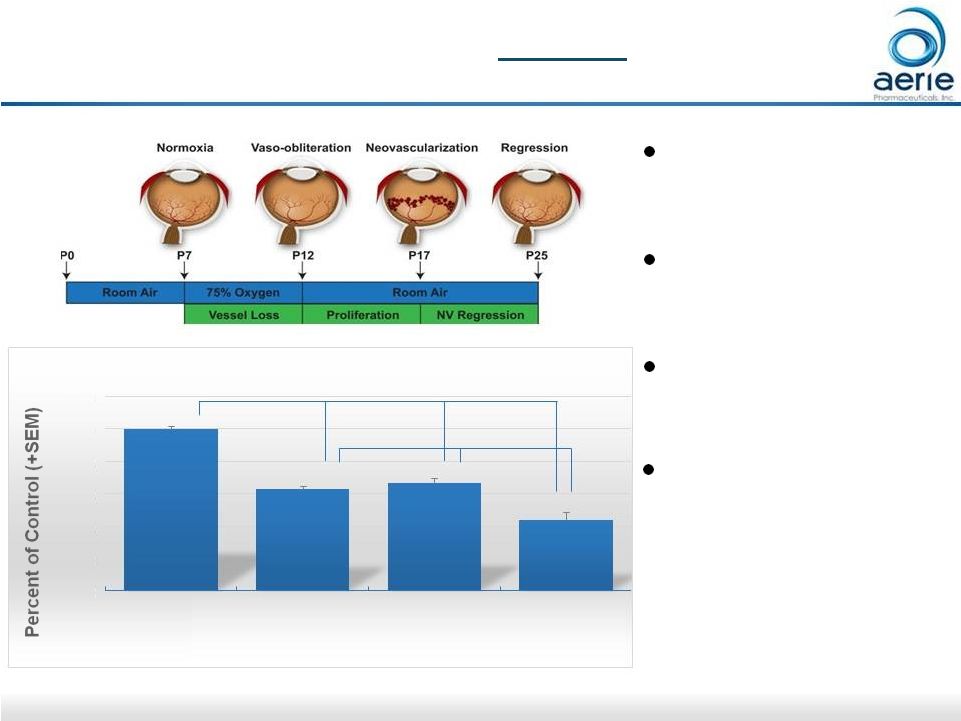 26 Topical AR-13154(S) Provides Additive Efficacy to Eylea® in Proliferative Diabetic Retinopathy Model -37% -34% -57% ** *** *** *** Oxygen-induced retinopathy model of PDR (mouse) 0.06% AR-13154(S) delivered topically from P12 to P17 Eylea ® delivered IP Confirms AR-13154(S) potential as monotherapy and as adjunct to anti-VEGF therapies Data on file; Effectiveness of AR-13154 Monotherapy and Combination Therapy in Animal Models of Wet Age-related Macular Degeneration and Proliferative Diabetic Retinopathy. Cheng-Wen Lin, Jill M. Sturdivant, Mitchell A. deLong and Casey C. Kopczynski 120% 100% 80% 60% 40% 20% 0% Vehicle Control (n=55) AR-13154(S) topical (n=28) Eylea 1mg/kg IP (n=26) AR-13154(S) + Eylea (n=18) Total Neovascular Area |
 27 AR-13154 Efficacy Driven Primarily by ROCK, PKC Inhibition in Retina Retinal tissue harvested from OIR mouse model AR-13154 effectively converted to active metabolite by esterases Active metabolite keeps ROCK, PKC activity, loses PDGFR, JAK activity Retinal Esterases X AR-13154 Active Inactive Data on file; Effectiveness of AR-13154 Monotherapy and Combination Therapy in Animal Models of Wet Age-related Macular Degeneration and Proliferative Diabetic Retinopathy. Cheng-Wen Lin, Jill M. Sturdivant, Mitchell A. deLong and Casey C. Kopczynski ROCK JAK PKC PDGFR ROCK PKC |
 Products in Development: Preclinical • Netarsudil* sustained-release implant for Glaucoma • Single injection to lower IOP for 3 – 6 months • Potential for increased efficacy, reduced ocular side effects, neuroprotection • Addresses problem of patient adherence to daily drops and gives control to the physician • AR-13154 implant for AMD, DR/DME • ROCK/PKC/PDGFR/JAK multi-kinase inhibitor • Addresses multiple drivers of disease - fibrosis, inflammation, vessel leakage and neovascularization • Efficacy demonstrated as monotherapy and as adjunct to anti-VEGF therapy (preclinical) • Potential to improve long-term outcomes * Active ingredient of Rhopressa™ 28 |
 29 Summary • Key Clinical Priorities • Rhopressa TM : NDA resubmission expected near end of Q1 2017 • Research Initiatives • Rhopressa™ disease modification, neuroprotection, sustained release • AR-13154 potential in wet AMD, etc. • Evaluating Aerie’s 3,000+ proprietary molecules • Business Development and Expansion Opportunities • Drug delivery opportunities for front and back of the eye • EU/JP clinical path and commercialization strategy • Ireland Manufacturing Facility • Fully Financed* Rocket 4 completion Mercury 3 commencement 1H 2017 (EU) Mercury 1 and Mercury 2 completion • Roclatan TM : * $255.6 million in cash and investments at 9/30/16 |
 2016 2017 Rhopressa™ and Roclatan™ Key Milestones 30 Q3-2017: Roclatan™ P3 Mercury 1 Topline safety (12 mos) Near YE 2017: Roclatan™ NDA filing expected 1H-2017: Roclatan™ P3 Mercury 3 (EU) to be initiated Q1-2016: Rhopressa™ Rocket 2 Topline safety (12 mos) Near End of Q1-2017: Rhopressa™ NDA re-filing expected Q4-2016: Rhopressa™ Rocket 4 Topline efficacy (3 mos) Q3-2016: Roclatan™ P3 Mercury 1 Topline efficacy (3 mos) Q2-2017: Roclatan™ P3 Mercury 2 Topline efficacy (3 mos) Q1-2016: Roclatan™ P3 Mercury 2 initiated Q2-2017: Rhopressa™ Rocket 4 Topline safety (6 mos) |