Exhibit 99.2

March 19, 2020 Results FY 2019 and Outlook 2020 Jean-Paul Kress, M.D., CEO Jens Holstein, CFO Malte Peters, M.D., CR&DO

Forward-Looking Statements This communication contains certain forward-looking statements concerning the MorphoSys group of companies, including its financial guidance for 2020, the commencement, timing and results of clinical trials and release of clinical data both in respect of its proprietary product candidates and of product candidates of its collaborators, the development of commercial capabilities, interpretations by regulatory authorities of our clinical data and real-world data analyses, in particular with respect to tafasitamab (MOR208), and the transition of MorphoSys to a fully integrated biopharmaceutical company, the expected time of launch of tafasitamab (MOR208), interaction with regulators, including the potential approval of MorphoSys’s current or future drug candidates, including discussions with the FDA regarding the potential approval to market tafasitamab (MOR208), and expected royalty and milestone payments in connection with MorphoSys’s collaborations. The forward-looking statements contained herein represent the judgment of MorphoSys as of the date of this release and involve known and unknown risks and uncertainties, which might cause the actual results, financial condition and liquidity, performance or achievements of MorphoSys, or industry results, to be materially different from any historic or future results, financial conditions and liquidity, performance or achievements expressed or implied by such forward-looking statements. In addition, even if MorphoSys’s results, performance, financial condition and liquidity, and the development of the industry in which it operates are consistent with such forward-looking statements, they may not be predictive of results or developments in future periods. Among the factors that may result in differences are that MorphoSys’s expectations regarding its 2020 results of operations may be incorrect, MorphoSys’s expectations regarding its development programs may be incorrect, the inherent uncertainties associated with competitive developments, clinical trial and product development activities and regulatory approval requirements (including that MorphoSys may fail to obtain regulatory approval for tafasitamab (MOR208) and that data from MorphoSys’s ongoing clinical research programs may not support registration or further development of its product candidates due to safety, efficacy or other reasons), MorphoSys’s reliance on collaborations with third parties, estimating the commercial potential of its development programs and other risks indicated in the risk factors included in MorphoSys’s Annual Report on Form20-F and other filings with the US Securities and Exchange Commission. Given these uncertainties, the reader is advised not to place any undue reliance on such forward-looking statements. These forward-looking statements speak only as of the date of publication of this document. MorphoSys expressly disclaims any obligation to update any such forward-looking statements in this document to reflect any change in its expectations with regard thereto or any change in events, conditions or circumstances on which any such statement is based or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements, unless specifically required by law or regulation. The compounds discussed in this slide presentation are investigational products being developed by MorphoSys and its partners and are not currently approved by the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA) or any other regulatory authority (except for guselkumab/Tremfya®). There is no guarantee any investigational product will be approved. © MorphoSys—Results FY 2019 and Outlook 2020 2

4Q & 2019 Earnings Call Agenda Moderation Julia Neugebauer, Ph.D., Director Investor Relations FY19 Highlights & Jean-Paul Kress, M.D., CEO Tafasitamab Launch Preparation Update R&D Update Malte Peters, M.D., CR&DO Financial Review FY2019 & Jens Holstein, CFO Outlook 2020 Closing Remarks Jean-Paul Kress, CEO Q&A Jean-Paul Kress, Jens Holstein, Malte Peters Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 3

FY 2019 Highlights Jean-Paul Kress, CEO © MorphoSys—Results FY 2019 and Outlook 2020 4

2019 Full-Year Highlights Execution on our 2019 Goals Tafasitamab – Strong Clinical Progress Tafasitamab – BLA Filing Accepted, FDA Granted Priority Review, PDUFA Date: Aug 30, 2020 Partnering Deal with IncyteBuild-up U.S. Organization & Launch Preparations Pipeline Progress: MOR202, Otilimab, Gantenerumab, Tremfya®ïƒ¼ Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 5
Global Collaboration for Tafasitamab with Incyte & Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 6

Tafasitamab Launch Preparation Update Jean-Paul Kress, CEO © MorphoSys—Results FY 2019 and Outlook 2020 7

Diffuse Large B Cell Lymphoma Patients with High Medical Need ~30,000 newly diagnosed DLBCL patients in the US each year1) Poor 40% High Unmet Prognosis Medical Need relapse or are refractory to the for relapsed/refractorynon- for new treatment options initial therapy transplant eligible patients 1) DRG Epidemiology data; Kantar Market Research (TPP testing 2018), Friedberg et al., 2011 © MorphoSys—Results FY 2019 and Outlook 2020 8
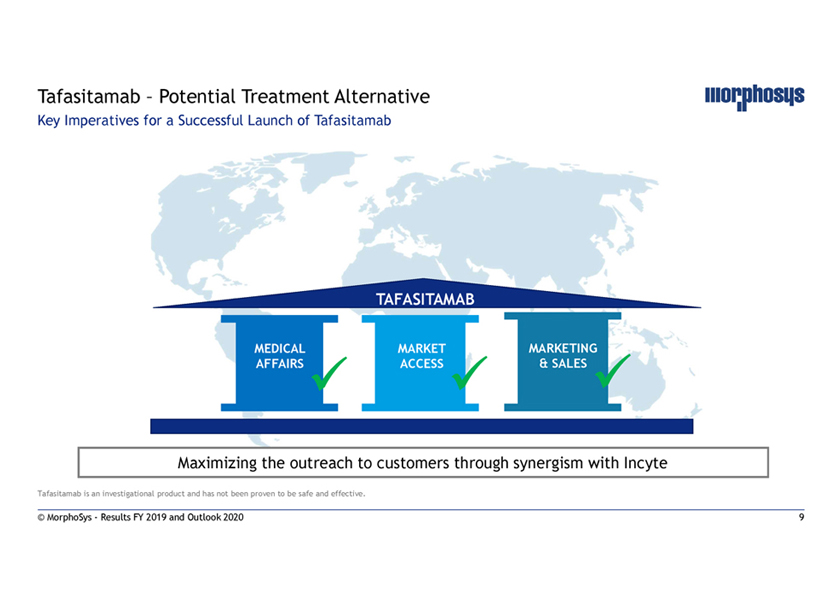
Tafasitamab – Potential Treatment Alternative Key Imperatives for a Successful Launch of Tafasitamab TAFASITAMAB MEDICAL MARKET MARKETING AFFAIRS ACCESS & SALES Maximizing the outreach to customers through synergism with Incyte Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 9
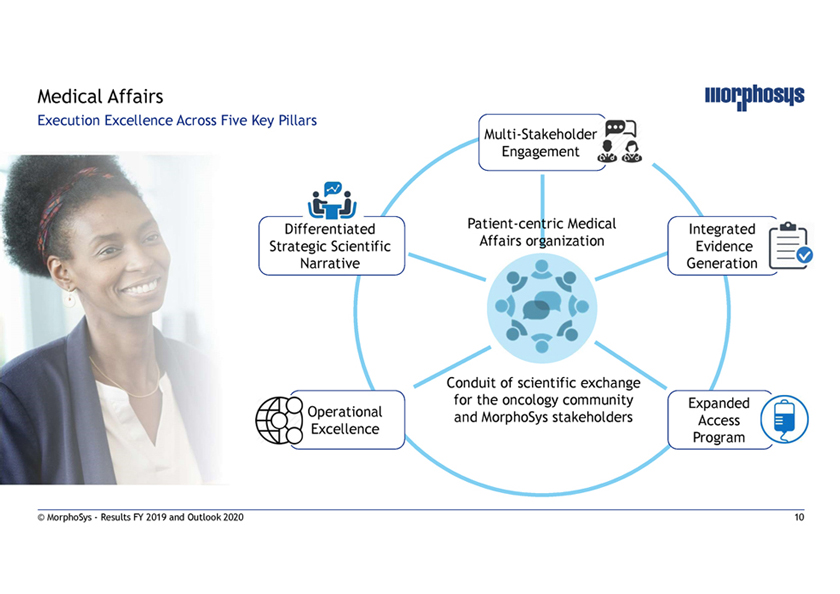
Medical Affairs Execution Excellence Across Five Key Pillars Multi-Stakeholder Engagement Differentiated Patient-centric Medical Integrated Strategic Scientific Affairs organization Evidence Narrative Generation Conduit of scientific exchange for the oncology community Expanded Operational and MorphoSys stakeholders Access Excellence Program © MorphoSys—Results FY 2019 and Outlook 2020 10
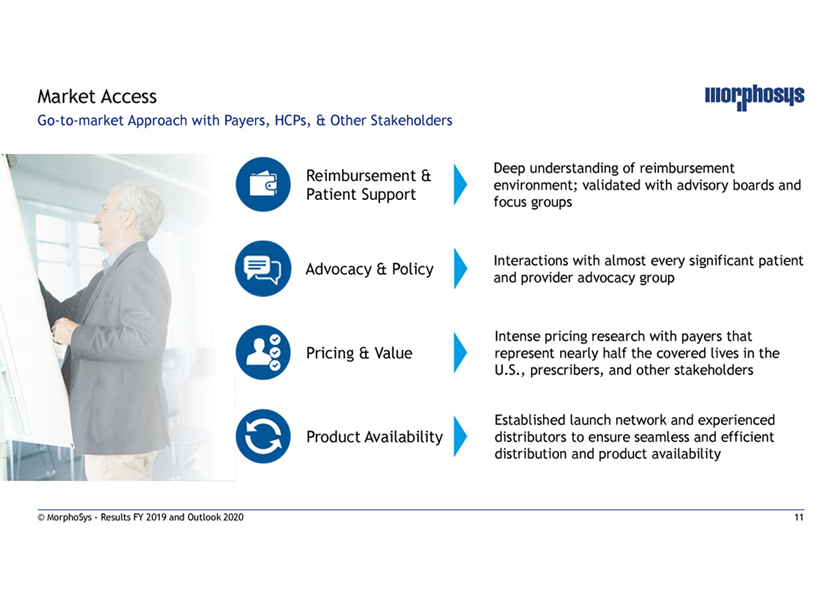
Market AccessGo-to-market Approach with Payers, HCPs, & Other Stakeholders Reimbursement & Deep understanding of reimbursement environment; validated with advisory boards and Patient Support focus groups Interactions with almost every significant patient Advocacy & Policy and provider advocacy group Intense pricing research with payers that Pricing & Value represent nearly half the covered lives in the U.S., prescribers, and other stakeholders Established launch network and experienced Product Availability distributors to ensure seamless and efficient distribution and product availability © MorphoSys—Results FY 2019 and Outlook 2020 11
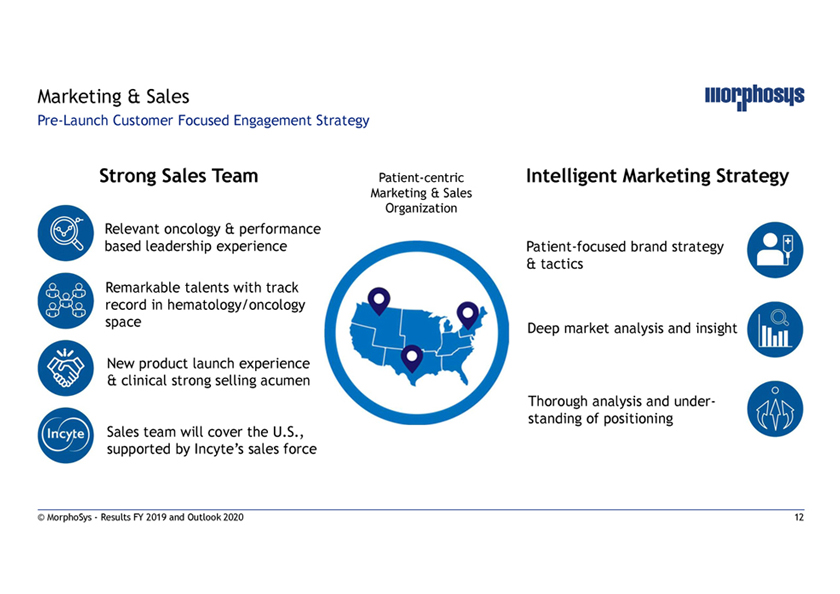
Marketing & SalesPre-Launch Customer Focused Engagement Strategy Strong Sales Team Patient-centric Intelligent Marketing Strategy Marketing & Sales Organization Relevant oncology & performance based leadership experience Patient-focused brand strategy & tactics Remarkable talents with track record in hematology/oncology space Deep market analysis and insight New product launch experience & clinical strong selling acumen Thorough analysis and under- Sales team will cover the U.S., standing of positioning supported by Incyte’s sales force © MorphoSys—Results FY 2019 and Outlook 2020 12

R&D Update Malte Peters, CR&DO © MorphoSys—Results FY 2019 and Outlook 2020 13
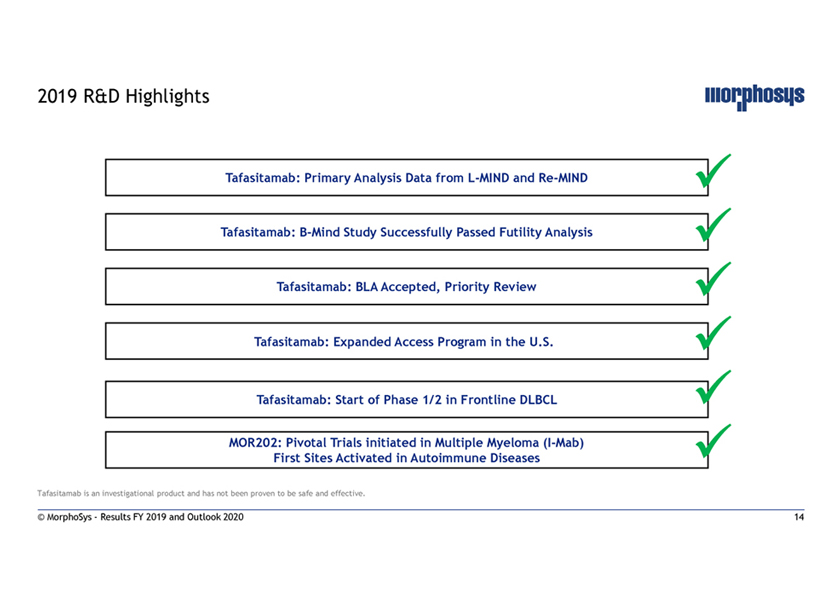
2019 R&D Highlights Tafasitamab: Primary Analysis Data fromL-MIND andRe-MIND Tafasitamab:B-Mind Study Successfully Passed Futility Analysis Tafasitamab: BLA Accepted, Priority Review Tafasitamab: Expanded Access Program in the U.S.. Tafasitamab: Start of Phase 1/2 in Frontline DLBCL  MOR202: Pivotal Trials initiated in Multiple Myeloma(I-Mab) First Sites Activated in Autoimmune Diseases Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 14
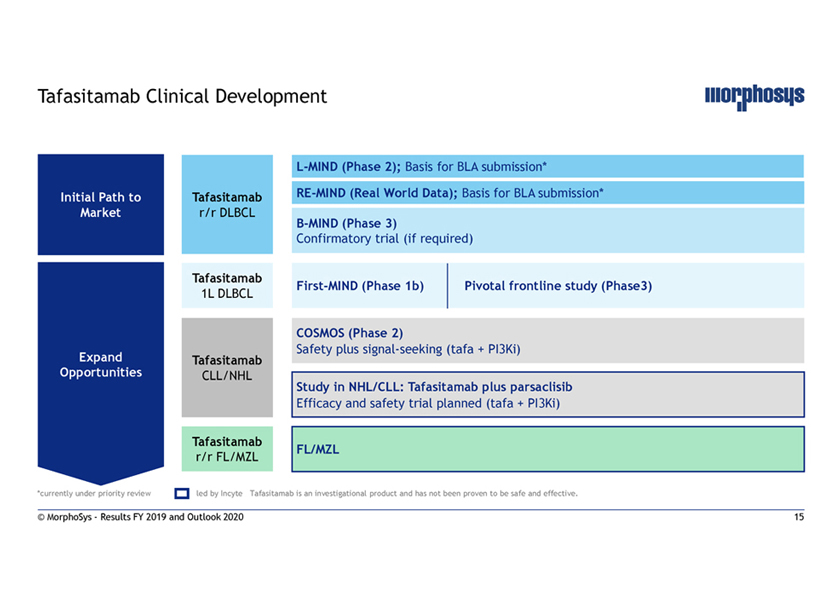
Tafasitamab Clinical DevelopmentL-MIND (Phase 2); Basis for BLA submission* Initial Path to TafasitamabRE-MIND (Real World Data); Basis for BLA submission* Market r/r DLBCLB-MIND (Phase 3) Confirmatory trial (if required) Tafasitamab First-MIND (Phase 1b) Pivotal frontline study (Phase3) 1L DLBCL COSMOS (Phase 2) Safety plus signal-seeking (tafa + PI3Ki) Expand Tafasitamab Opportunities CLL/NHL Study in NHL/CLL: Tafasitamab plus parsaclisib Efficacy and safety trial planned (tafa + PI3Ki) Tafasitamab FL/MZL r/r FL/MZL *currently under priority review led by Incyte Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 15
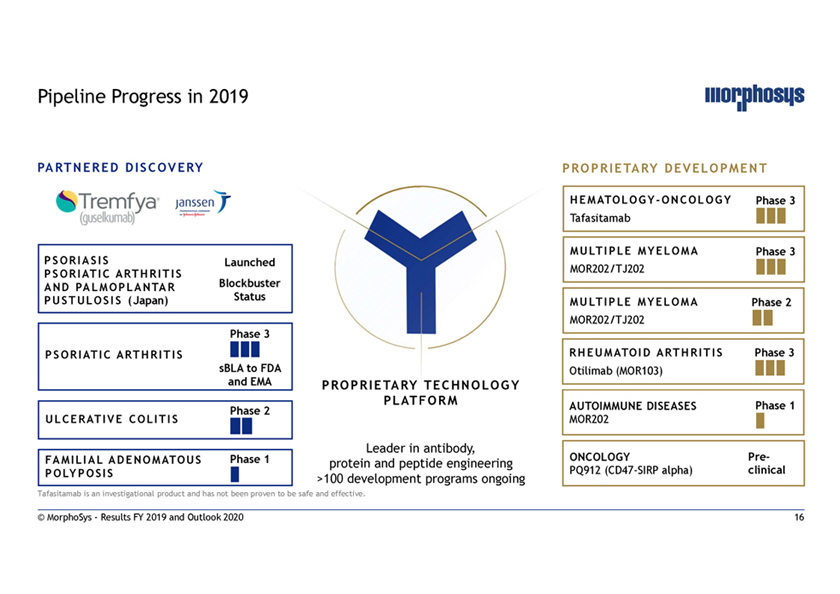
Pipeline Progress in 2019 PARTNERED DISCOVERY PROPRIETARY DEVELOPMENT HEMATOLOGY-ONCOLOGY Phase 3 Tafasitamab MULTIPLE MYELOMA Phase 3 PSORIASIS Launched PSORIATIC ARTHRITIS MOR202/TJ202 AND PALMOPLANTAR Blockbuster PUSTULOSIS (Japan) Status MULTIPLE MYELOMA Phase 2 MOR202/TJ202 Phase 3 PSORIATIC ARTHRITIS RHEUMATOID ARTHRITIS Phase 3 sBLA to FDA Otilimab (MOR103) and EMA PROPRIETARY TECHNOLOGY PLATFORM AUTOIMMUNE DISEASES Phase 1 Phase 2 ULCERATIVE COLITIS MOR202 Leader in antibody, FAMILIAL ADENOMATOUS Phase 1 ONCOLOGY Pre- protein and peptide engineering POLYPOSIS PQ912 (CD47-SIRP alpha) clinical >100 development programs ongoing Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 16

Financial Review FY2019 & Outlook 2020 Jens Holstein, CFO © MorphoSys—Results FY 2019 and Outlook 2020 17
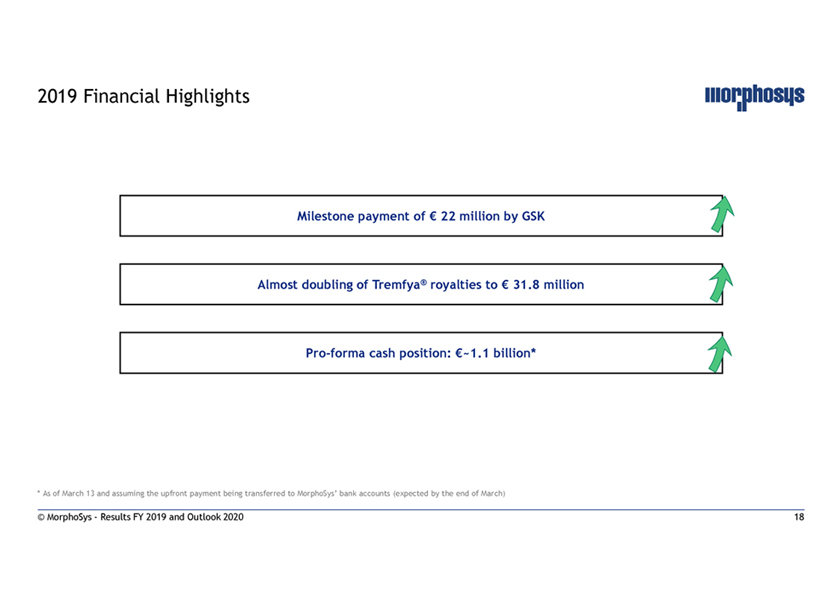
2019 Financial Highlights Milestone payment of € 22 million by GSK Almost doubling of Tremfya® royalties to € 31.8 millionPro-forma cash position: €~1.1 billion* * As of March 13 and assuming the upfront payment being transferred to MorphoSys’ bank accounts (expected by the end of March) © MorphoSys—Results FY 2019 and Outlook 2020 18
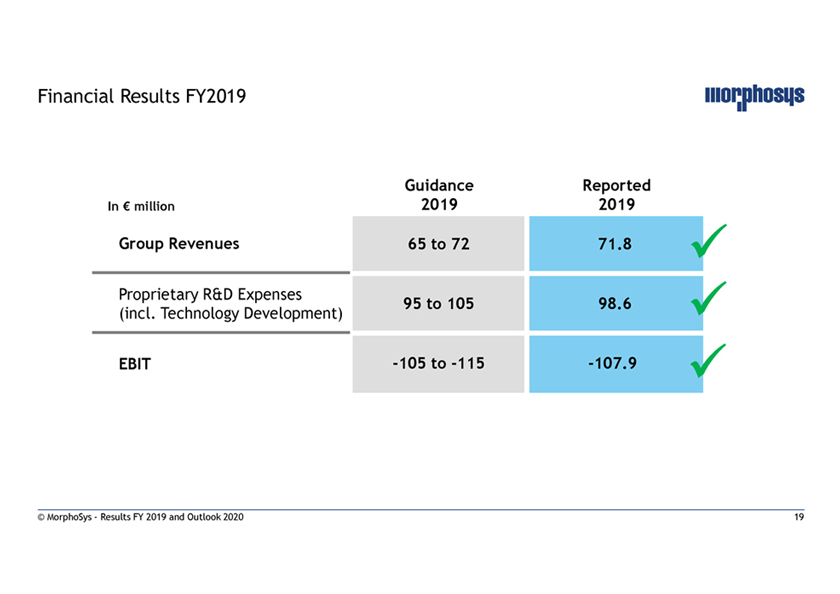
Financial Results FY2019 Guidance Reported In € million 2019 2019 Group Revenues 65 to 72 71.8 Proprietary R&D Expenses 95 to 105 98.6 (incl. Technology Development)  EBIT-105 to-115-107.9 © MorphoSys—Results FY 2019 and Outlook 2020 19
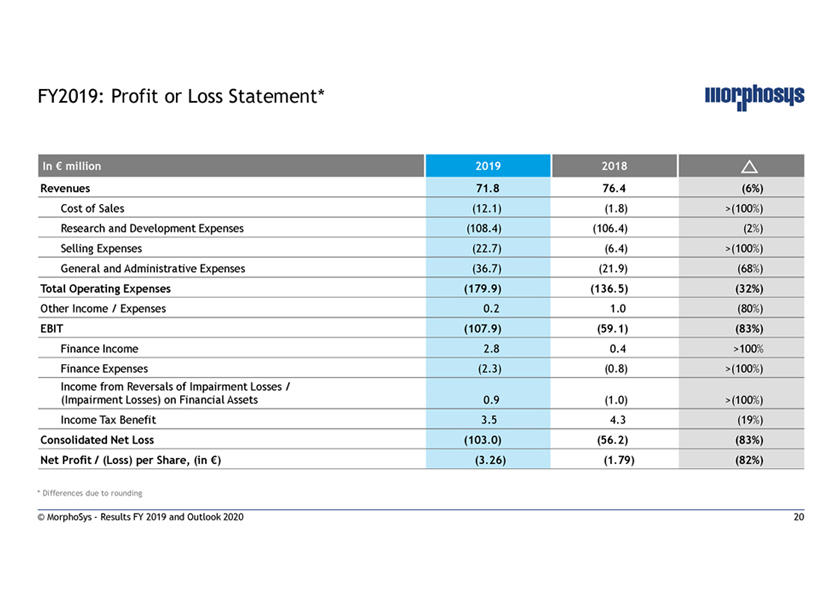
FY2019: Profit or Loss Statement* In € million 2019 2018 Revenues 71.8 76.4 (6%) Cost of Sales (12.1) (1.8) >(100%) Research and Development Expenses (108.4) (106.4) (2%) Selling Expenses (22.7) (6.4) >(100%) General and Administrative Expenses (36.7) (21.9) (68%) Total Operating Expenses (179.9) (136.5) (32%) Other Income / Expenses 0.2 1.0 (80%) EBIT (107.9) (59.1) (83%) Finance Income 2.8 0.4 >100% Finance Expenses (2.3) (0.8) >(100%) Income from Reversals of Impairment Losses / (Impairment Losses) on Financial Assets 0.9 (1.0) >(100%) Income Tax Benefit 3.5 4.3 (19%) Consolidated Net Loss (103.0) (56.2) (83%) Net Profit / (Loss) per Share, (in €) (3.26) (1.79) (82%) * Differences due to rounding © MorphoSys—Results FY 2019 and Outlook 2020 20
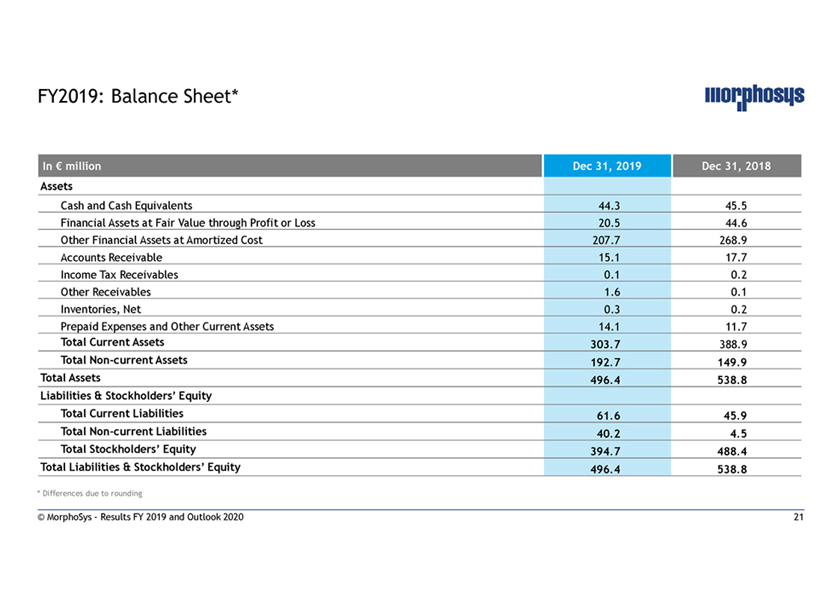
FY2019: Balance Sheet* In € million Dec 31, 2019 Dec 31, 2018 Assets Cash and Cash Equivalents 44.3 45.5 Financial Assets at Fair Value through Profit or Loss 20.5 44.6 Other Financial Assets at Amortized Cost 207.7 268.9 Accounts Receivable 15.1 17.7 Income Tax Receivables 0.1 0.2 Other Receivables 1.6 0.1 Inventories, Net 0.3 0.2 Prepaid Expenses and Other Current Assets 14.1 11.7 Total Current Assets 303.7 388.9 TotalNon-current Assets 192.7 149.9 Total Assets 496.4 538.8 Liabilities & Stockholders’ Equity Total Current Liabilities 61.6 45.9 TotalNon-current Liabilities 40.2 4.5 Total Stockholders’ Equity 394.7 488.4 Total Liabilities & Stockholders’ Equity 496.4 538.8 * Differences due to rounding © MorphoSys—Results FY 2019 and Outlook 2020 21
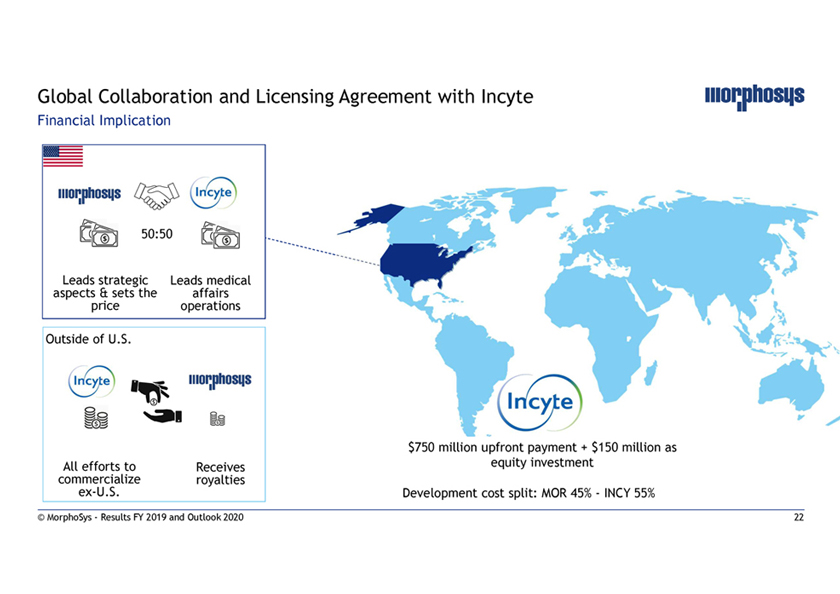
Global Collaboration and Licensing Agreement with Incyte Financial Implication 50:50 Leads strategic Leads medical aspects & sets the affairs price operations Outside of U.S. $750 million upfront payment + $150 million as All efforts to Receives equity investment commercialize royaltiesex-U.S. Development cost split: MOR 45%—INCY 55% © MorphoSys—Results FY 2019 and Outlook 2020 22

Financial Guidance FY2020 Reported Guidance In € million FY2019 FY2020 Group Revenues* 71.8 280 to 290 Total R&D Expenses 108.4 130 to 140 EBIT-107.9-15 to +5 * Revenues do not include potential sales of tafasitamab. Revenues are expected to include royalty income from Tremfya® ranging from EUR 37 to 42 million on constantUS-$ currency. © MorphoSys—Results FY 2019 and Outlook 2020 23

Closing Remarks Jean-Paul Kress, CEO © MorphoSys—Results FY 2019 and Outlook 2020 24
Exceptional, Innovative Biopharmaceuticals To Improve the Lives of Patients Suffering from Serious Diseases Full supply chain and commercial capabilities Leading antibody technologies & innovative science © MorphoSys—Results FY 2019 and Outlook 2020 25
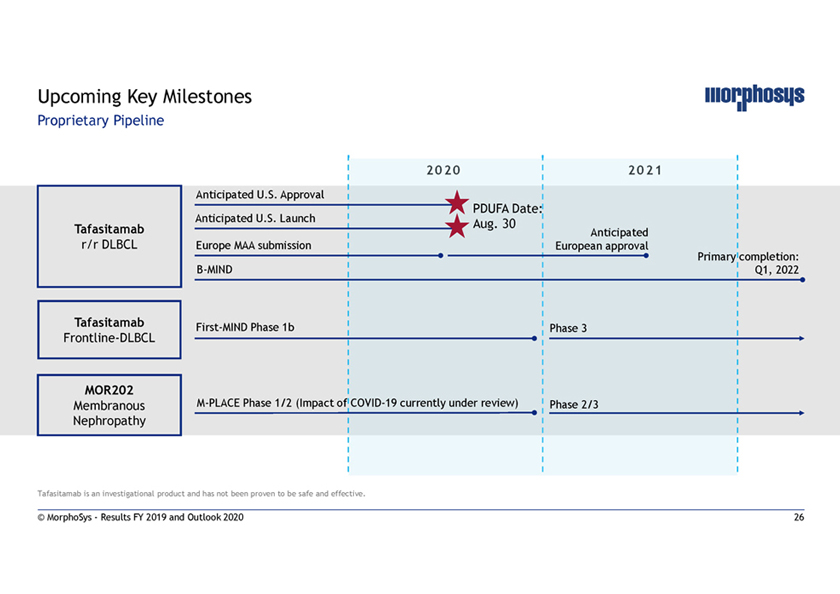
Upcoming Key Milestones Proprietary Pipeline Anticipated U.S. Approval PDUFA Date: Anticipated U.S. Launch Aug. 30 Tafasitamab Anticipated r/r DLBCL Europe MAA submission European approval Primary completion:B-MIND Q1, 2022 Tafasitamab First-MIND Phase 1b Phase 3 Frontline-DLBCL MOR202 MembranousM-PLACE Phase 1/2 (Impact ofCOVID-19 currently under review) Phase 2/3 Nephropathy Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 26

Key Priorities for 2020 Well Positioned for Success and Value Creation Flawless tafasitamab Unlock tafasitamab‘s Progress of our other launch* in the U.S. and full potential with proprietary programs successful Incyte commercialization * given FDA approval; Tafasitamab is an investigational product and has not been proven to be safe and effective. © MorphoSys—Results FY 2019 and Outlook 2020 27

Q & A © MorphoSys—Results FY 2019 and Outlook 2020 28

Thank You www.morphosys.com Tafasitamab (MOR208), MOR202, MOR106, otilimab (MOR103), gantenerumab and all other product candidates discussed in this slide presentation are investigational products being developed by MorphoSys and its partners and are not currently approved by the U.S. Food & Drug Administration (FDA), European Medicine Agency (EMA) or any other regulatory authority (except for guselkumab/ Tremfya®). There is no guarantee any product will be approved in the sought-after indication. HuCAL® , HuCAL GOLD®, HuCAL PLATINUM®, CysDisplay®, RapMAT®, arYla®, Ylanthia®, 100 billion high potentials®, Slonomics®, Lanthio Pharma®, LanthioPep® and ENFORCERTM are trademarks of the MorphoSys Group. Tremfya® is a trademark of Janssen Biotech, Inc. XmAb ® is a trademark of Xencor Inc.