Exhibit (a)(2)
– English Translation –
Mandatory Publication pursuant to
Sec. 27 para. 3 in conjunction with Sec. 14 para. 3 sentence 1 of the
German Securities Acquisition and
Takeover Act (Wertpapiererwerbs- und Übernahmegesetz, WpÜG)

Joint Reasoned Statement
of the Management Board and the Supervisory Board
pursuant to Sec. 27 para. 1 WpÜG
of
MorphoSys AG
Semmelweisstrasse 7
82152 Planegg, Germany
on the Voluntary Public Takeover Offer
(Cash Offer)
made by
Novartis BidCo AG
Lichtstrasse 35
4056 Basel, Switzerland
to
the shareholders of MorphoSys AG
for the acquisition of all no-par value bearer shares
including all no-par value bearer shares
represented by American Depositary Shares
in MorphoSys AG
dated 11 April 2024
Shares of MorphoSys AG: ISIN DE0006632003
ADSs of MorphoSys AG: ISIN US6177602025
Tendered Shares of MorphoSys AG: ISIN DE000A4BGGU0
Subsequently Tendered Shares of MorphoSys AG: ISIN DE000A4BGGV8
TABLE OF CONTENTS
| Page | ||||
1 INTRODUCTION | 7 | |||
1.1 Background of the Offer | 7 | |||
1.2 German and U.S. Takeover Rules | 8 | |||
2 GENERAL INFORMATION ABOUT THIS STATEMENT | 9 | |||
2.1 Legal Basis for this Statement | 9 | |||
2.2 Joint Reasoned Statement by the Management Board and the Supervisory Board | 9 | |||
2.3 Certain Defined Terms and Meanings | 10 | |||
2.4 Factual Basis for this Statement | 10 | |||
2.5 Forward Looking Statements | 11 | |||
2.6 No Statement by the Employees of MorphoSys | 11 | |||
2.7 Publication of this Statement and Additional Statements | 12 | |||
2.8 Responsibility of MorphoSys Securityholders | 12 | |||
2.9 MorphoSys Securityholders in the United States | 13 | |||
3 INFORMATION ABOUT MORPHOSYS | 13 | |||
3.1 General Information on MorphoSys | 13 | |||
3.2 Capital and Shareholder Structure of MorphoSys | 14 | |||
3.3 Listings of MorphoSys | 18 | |||
3.4 Management Board and the Supervisory Board of MorphoSys | 18 | |||
3.5 Group Structure and Business Activities of MorphoSys | 18 | |||
3.6 Strategy of MorphoSys | 19 | |||
3.7 Selected Key Financial Figures of MorphoSys | 20 | |||
4 INFORMATION ABOUT THE BIDDER AND NOVARTIS | 24 | |||
4.1 General Information | 25 | |||
4.2 MorphoSys Shares Held by the Bidder and Persons Acting Jointly with it | 27 | |||
4.3 Information about Securities Transactions | 28 | |||
5 INFORMATION ABOUT THE OFFER | 29 | |||
5.1 Implementation of the Offer | 29 | |||
5.2 Publication of the Decision to Launch the Offer | 29 | |||
5.3 BaFin Review and Publication of the Offer Document | 29 | |||
5.4 Publications by the Bidder in Connection with the Offer | 30 | |||
5.5 Material Terms and Conditions of the Offer | 31 | |||
5.6 Acceptance and Settlement of the Offer for MorphoSys Securities | 35 | |||
5.7 Stock-Exchange Trading in Tendered MorphoSys Shares | 38 | |||
5.8 Financing of the Offer | 38 | |||
5.9 Relevance of the Offer Document | 39 | |||
6 EVALUATION OF THE OFFER | 39 | |||
6.1 Type and Amount of the Offer Consideration | 39 | |||
6.2 Opinions of Centerview | 49 | |||
6.3 Certain Unaudited Prospective Financial Information of MorphoSys | 59 | |||
6.4 Adequacy of the Offer Price and other Terms of the Business Combination Agreement | 65 | |||
7 OBJECTIVES AND INTENTIONS OF THE BIDDER, NOVARTIS PHARMA AND NOVARTIS AG AS WELL AS EXPECTED CONSEQUENCES FOR MORPHOSYS | 65 | |||
7.1 Objectives and Intentions set forth in the Offer Document | 65 | |||
2
7.2 Structural Measures | 68 | |||
7.3 Evaluation of the Objectives and Intentions of the Bidder, Novartis Pharma and Novartis AG and Expected Consequences for MorphoSys | 71 | |||
8 CONSEQUENCES FOR MORPHOSYS SECURITYHOLDERS | 72 | |||
8.1 Possible Consequences of Acceptance of the Offer | 72 | |||
8.2 Possible Consequences of Non-Acceptance of the Offer | 73 | |||
9 STATUS OF REGULATORY APPROVALS | 75 | |||
10 TRANSACTION HISTORY AND PAST CONTACTS WITH NOVARTIS AG | 76 | |||
10.1 Transaction History | 76 | |||
10.2 Past Contacts, Transactions, Negotiations and Agreements with Novartis AG | 89 | |||
11 PERSONS OR ASSETS RETAINED, EMPLOYED, COMPENSATED OR USED | 90 | |||
11.1 Centerview | 90 | |||
11.2 Innisfree | 91 | |||
12 PURPOSES OF THE TRANSACTION AND PLANS OR PROPOSALS | 91 | |||
13 INTERESTS OF THE MEMBERS OF THE MANAGEMENT BOARD AND SUPERVISORY BOARD | 91 | |||
13.1 Specific Interests of the Members of the Management Board and Supervisory Board | 91 | |||
13.2 Agreements with Members of the Management Board or Supervisory Board | 94 | |||
14 SHAREHOLDINGS AND INTENTION OF THE MEMBERS OF THE MANAGEMENT BOARD AND OF THE SUPERVISORY BOARD TO ACCEPT THE OFFER | 97 | |||
15 FINAL STATEMENT AND RECOMMENDATION | 97 | |||
ANNEX | 99 | |||
3
DEFINED TERMS
15 January Novartis Offer | 85 | |||
15 January Novartis Offer Conditions | 85 | |||
20 December Company C Proposal | 78 | |||
2024 Incentive Plans | 17 | |||
2024 Performance Share Unit Program | 17 | |||
2024 Restricted Stock Unit Program | 17 | |||
22 December Company C Proposal | 79 | |||
3 January Company C Proposal | 82 | |||
3 January Novartis Proposal | 82 | |||
Acceptance Period | 31 | |||
Additional Acceptance Period | 32 | |||
ADS Deposit Agreement | 70 | |||
ADS Depositary | 36 | |||
ADS Tender Agent | 37 | |||
Affiliates | 28 | |||
AktG | 14 | |||
Appraiser Review Report | 69 | |||
ASH Annual Meeting | 78 | |||
BaFin | 8 | |||
Banking Day | 10 | |||
Beneficiary | 96 | |||
BET | 19 | |||
Bidder | 7 | |||
BörsG | 46 | |||
Business Combination Agreement | 10 | |||
Centerview | 39 | |||
Centerview Engagement Letter | 90 | |||
Centerview Opinions | 49 | |||
Clearstream | 35 | |||
Closing | 16 | |||
Code | 93 | |||
Company A | 76 | |||
Company B | 77 | |||
Company C | 78 | |||
Competing Offer | 32 | |||
Competing Transaction | 47 | |||
Conditional Capital 2016-III | 16 | |||
Conditional Conversion Right | 16 | |||
Confidentiality Agreement | 90 | |||
Converted Cash Award | 92 | |||
Convertible Bonds | 16 | |||
Custodian Bank | 35 | |||
CymaBay Transaction | 56 | |||
Declaration of Acceptance | 35 | |||
Delisting | 46 | |||
DPLTA | 46 | |||
DPLTA Cash Compensation | 69 | |||
DPLTA Report | 69 | |||
DPLTA Valuation | 69 | |||
Dr. Gal | 77 |
Dr. Krebs-Pohl | 77 | |||
Dr. Kress | 78 | |||
Dr. Kreutz | 77 | |||
Dr. Narasimhan | 80 | |||
Dr. White | 76 | |||
DTC | 37 | |||
DVFA | 69 | |||
Equity Consents | 93 | |||
EUR | 10 | |||
EURbn | 10 | |||
EURm | 10 | |||
EV/5-Year Fwd. Revenue Multiple | 55 | |||
EV/CY’28E Revenue | 54 | |||
Exclusivity Agreement | 86 | |||
Executive Committee | 50 | |||
FCA | 44 | |||
FDA | 20 | |||
Final Letter of Intent | 86 | |||
Final Letter of Intent BCA | 86 | |||
Final Letter of Intent Conditions | 86 | |||
Forecasts | 59 | |||
Frankfurt am Main Local Time | 10 | |||
Freshfields | 82 | |||
FSE | 10 | |||
FSE Trading Day | 10 | |||
G&A | 21 | |||
German Takeover Rules | 8 | |||
Germany | 10 | |||
IASB | 20 | |||
IDW | 51 | |||
IDW Methodology | 69 | |||
IDW S 1 Expert | 69 | |||
IDW S 1 Standard | 69 | |||
IFRS | 20 | |||
Incentive Plans | 17 | |||
Incyte | 76 | |||
Incyte Purchase Agreement | 86 | |||
Internal Data | 50 | |||
JAK | 19 | |||
JPM Healthcare Conference | 78 | |||
Law | 34 | |||
Management Board | 9 | |||
MANIFEST-2 Study | 77 | |||
Material Compliance Violation | 34 | |||
Maximum Amount | 97 | |||
Minimum Acceptance Condition | 33 | |||
MorphoSys | 7 | |||
MorphoSys ADR | 7 | |||
MorphoSys ADRs | 7 | |||
MorphoSys ADS | 7 |
4
MorphoSys ADS Holder | 7 | |||
MorphoSys ADS Holders | 7 | |||
MorphoSys ADSs | 7 | |||
MorphoSys Affiliate | 18 | |||
MorphoSys Group | 7 | |||
MorphoSys Incentive Awards | 92 | |||
MorphoSys Securities | 7 | |||
MorphoSys Securityholders | 7 | |||
MorphoSys Share | 7 | |||
MorphoSys Shareholder | 7 | |||
MorphoSys Shareholders | 7 | |||
MorphoSys Shares | 7 | |||
MorphoSys Termination Fee | 47 | |||
Mr. Hoppenot | 85 | |||
Nasdaq | 8 | |||
Nasdaq Trading Day | 10 | |||
New York Local Time | 10 | |||
No Illegality Condition | 33 | |||
No Insolvency Condition | 33 | |||
No Material Compliance Violation Condition | 34 | |||
Novartis | 7 | |||
Novartis AG | 7 | |||
Novartis Pharma | 25 | |||
Novartis Termination Fee | 47 | |||
Offer | 7 | |||
Offer Conditions | 33 | |||
Offer Document | 7 | |||
Offer Opinion | 49 | |||
Offer Price | 7 | |||
Performance Share Unit | 17 | |||
Performance Share Unit Programs | 17 | |||
Projections | 59 | |||
Proposed Transactions | 39 | |||
R&D | 22 | |||
Recurring DPLTA Payment | 69 | |||
Regulatory Clearances | 44 | |||
Remuneration Modifications | 91 | |||
Restricted Stock Unit | 17 | |||
Restricted Stock Unit Programs | 17 | |||
Reuters Article | 88 | |||
Rival Offer | 46 | |||
Rival Offer Termination | 46 | |||
Sandoz Division | 27 | |||
Schedule 14D-9 | 9 | |||
Schedule TO | 7 | |||
SEC | 7 | |||
SEC Relief | 8 | |||
Section 280G | 96 | |||
Selected Company Comparison Companies | 53 | |||
Selected Transactions | 55 | |||
Service Agreements | 94 |
Settlement Deferral | 91 | |||
Signing Opinion | 49 | |||
Skadden | 39 | |||
Specified Governmental Authority | 33 | |||
Squeeze-out | 46 | |||
Statement | 7 | |||
Statutory Minimum Price Requirements | 41 | |||
Stock Option | 17 | |||
Stock Option Programs | 17 | |||
Subsequently Tendered MorphoSys Shares | 36 | |||
Superior Offer | 46 | |||
Supervisory Board | 9 | |||
Supplemental Statements | 12 | |||
Support Arrangement | 93 | |||
Tafasitamab Sale | 85 | |||
Takeover | 7 | |||
Tendered MorphoSys ADSs | 38 | |||
Tendered MorphoSys Shares | 36 | |||
Three-Month Average Price | 42 | |||
U.S. | 10 | |||
U.S. Exchange Act | 8 | |||
U.S. GAAP | 64 | |||
U.S. Offer Rules | 8 | |||
U.S. Working Day | 10 | |||
UmwG | 46 | |||
United States | 10 | |||
Unlevered Free Cash Flow | 61 | |||
USD | 10 | |||
USDbn | 10 | |||
USDm | 10 | |||
WpHG | 14 | |||
WpÜG | 7 | |||
WpÜG Offer Regulation | 8 |
5
NOTE TO THE SHAREHOLDERS OF MORPHOSYS AG
Dear MorphoSys Securityholders,
On 5 February 2024, Novartis AG and MorphoSys jointly announced that they entered into a Business Combination Agreement regarding a voluntary public takeover offer to be made by Novartis BidCo AG (formerly operating as Novartis data42 AG), a wholly-owned subsidiary of Novartis AG, to the MorphoSys Securityholders to acquire all MorphoSys Shares for a cash consideration of EUR 68.00 per MorphoSys Share.
Today, Novartis BidCo AG has launched its voluntary public takeover offer by way of publication of the Offer Document (Angebotsunterlage), as permitted by the German Financial Supervisory Authority (Bundesanstalt für Finanzdienstleistungsaufsicht). This Statement provides the joint reasoned statement (gemeinsame begründete Stellungnahme) of the Management Board and Supervisory Board regarding the Offer launched by Novartis BidCo AG.
Before entering into the Business Combination Agreement, the Management Board and the Supervisory Board made a thorough assessment of the envisaged voluntary public takeover offer and the contemplated acquisition of MorphoSys by Novartis. Consistent with their fiduciary duties, the Management Board and Supervisory Board, following a careful review of alternatives and of the different stakeholders’ interests as well as with the support of their legal and financial advisors, unanimously concluded that the Offer and the contemplated acquisition by Novartis is in the best interest of MorphoSys and its securityholders, the sustainable success of its business as well as clients, employees and other stakeholders.
We find it important to share with you our considerations, views and recommendation with respect to the Offer, which you will find in this Statement. We strongly encourage each MorphoSys Securityholder to read the Offer Document and this Statement carefully. Each MorphoSys Securityholder must make their own decision on whether and to what extent to accept the Offer, taking into account the overall circumstances.
As further set out in this Statement, the Management Board and the Supervisory Board each unanimously resolved to support the Offer and to recommend that MorphoSys Securityholders accept the Offer and tender their MorphoSys Securities in the Offer.
Yours sincerely,
| Jean-Paul Kress, M.D. | Lucinda Crabtree, Ph.D. | Marc Cluzel, M.D., Ph.D. | ||
| Chief Executive Officer and member of the Management Board | Chief Financial Officer and member of the Management Board | Chairman of the Supervisory Board | ||
6
JOINT REASONED STATEMENT
OF THE MANAGEMENT BOARD AND THE SUPERVISORY BOARD
| 1 | INTRODUCTION |
| 1.1 | Background of the Offer |
On 11 April 2024, Novartis BidCo AG (formerly operating as Novartis data42 AG), a stock corporation (Aktiengesellschaft) incorporated under the laws of Switzerland, with registered office at Lichtstrasse 35, 4056 Basel, Switzerland, registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-477.907.492 (the “Bidder”) and a wholly-owned indirect subsidiary of Novartis AG, a stock corporation (Aktiengesellschaft) incorporated under the laws of Switzerland, with registered office at Lichtstrasse 35, 4056 Basel, Switzerland, registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-103.867.266 (“Novartis AG”, and together with the Bidder and its other subsidiaries, “Novartis”) published an offer document (Angebotsunterlage) pursuant to Sec. 11 of the German Securities Acquisition and Takeover Act (Wertpapiererwerbs- und Übernahmegesetz – “WpÜG”) in accordance with Sec. 34 and Sec. 14 paras. 2 and 3 WpÜG for its voluntary public takeover offer (freiwilliges Übernahmeangebot) for the acquisition of all no-par value bearer shares (auf den Inhaber lautende Stückaktien) in MorphoSys AG a stock corporation (Aktiengesellschaft) incorporated under the laws of Germany, with registered office in Semmelweisstrasse 7, 82152 Planegg, Germany, registered with the commercial register of the local court (Amtsgericht) of Munich under HRB 121023 (“MorphoSys” and, together with its subsidiaries, the “MorphoSys Group”). The aforementioned offer document together with its annexes is hereinafter referred to as the “Offer Document”, the voluntary public takeover offer (freiwilliges Übernahmeangebot) for the acquisition of all no-par value bearer shares (auf den Inhaber lautende Stückaktien) in MorphoSys is herein referred to as the “Offer” and the takeover of MorphoSys by the Bidder contemplated by the Offer is herein referred to as the “Takeover”.
As per the Offer Document, an English translation of the Offer Document was filed on 11 April 2024 with the U.S. Securities and Exchange Commission (the “SEC”), the Bidder’s tender offer statement as an exhibit on Schedule TO (the “Schedule TO”). References to the Offer Document in this Statement (this “Statement”) without further specification, refer to both the German version and the English translation of the Offer Document.
The Offer is directed towards all shareholders of MorphoSys (collectively, the “MorphoSys Shareholders” and each individually, a “MorphoSys Shareholder”) and all holders of MorphoSys American Depository Shares representing MorphoSys Shares (collectively, the “MorphoSys ADS Holders” and each individually, a “MorphoSys ADS Holder”). The MorphoSys Shareholders and MorphoSys ADS Holders are hereinafter collectively referred to as the “MorphoSys Securityholders”.
The object of the Offer is the acquisition of (i) all issued no-par value bearer shares (auf den Inhaber lautende Stückaktien) in MorphoSys with a notional interest in the share capital of MorphoSys of EUR 1.00 each, including all ancillary rights, in particular the right to dividends, existing at the time of the settlement of the Offer, not held directly by the Bidder (ISIN DE0006632003) (collectively, the “MorphoSys Shares” and each individually, a “MorphoSys Share”) and (ii) all MorphoSys Shares represented by American Depositary Shares (collectively, the “MorphoSys ADSs” and each individually, a “MorphoSys ADS”) against payment of cash consideration in the amount of EUR 68.00 per MorphoSys Share without interest (the “Offer Price”). The MorphoSys Shares and the MorphoSys ADSs are hereinafter collectively referred to as “MorphoSys Securities”.
MorphoSys ADS may be evidenced by American Depositary Receipts (all MorphoSys ADS represented by American Depository Receipts collectively, the “MorphoSys ADRs” and each, a “MorphoSys ADR”), and each MorphoSys ADS represents one-quarter of one MorphoSys Share. MorphoSys ADS Holders are permitted to tender the underlying MorphoSys Shares in respect of the MorphoSys ADSs in the Offer following their withdrawal from the MorphoSys ADS program (as described in Sec. 13.2 of the
7
Offer Document) or to instruct the ADS Tender Agent (as defined below) to tender the MorphoSys Shares underlying their MorphoSys ADSs in the Offer (as described in Sec. 13.3.1 of the Offer Document). MorphoSys ADS Holders may accept the Offer regardless of whether the total number of MorphoSys ADSs tendered by them is an integral multiple of four (4) (i.e., the Offer may be accepted for one (1), two (2) or three (3) MorphoSys ADSs).
| 1.2 | German and U.S. Takeover Rules |
The Offer will be carried out in accordance with the WpÜG and the German Regulation on the Content of the Offer Document, the Consideration for Takeover Offers and Mandatory Offers and the Exemption from the Obligation to Publish and Launch an Offer (Verordnung über den Inhalt der Angebotsunterlage, die Gegenleistung bei Übernahmeangeboten und Pflichtangeboten und die Befreiung von der Verpflichtung zur Veröffentlichung und zur Abgabe eines Angebots) (the “WpÜG Offer Regulation” and, together with the WpÜG, the “German Takeover Rules”).
Furthermore, the Offer is carried out in accordance with the securities laws of the United States, including the provisions applicable to tender offers of the U.S. Securities Exchange Act of 1934, as amended (the “U.S. Exchange Act”) and the rules and regulations promulgated thereunder (together with the U.S. Exchange Act and the SEC Relief to reconcile certain areas where German law and U.S. law conflict, the “U.S. Offer Rules”).
Given that MorphoSys is a German stock corporation and the MorphoSys Shares are admitted to trading on a regulated market in Germany and within the European Economic Area, the Offer must comply with the terms and conditions of the WpÜG with regard to the consideration offered, the content of the Offer Document and the procedures to be followed in connection with the Offer (Sec. 1 para. 3 WpÜG), the compliance with which will be supervised by the German Federal Financial Supervisory Authority (Bundesanstalt für Finanzdienstleistungsaufsicht) (the “BaFin”). In addition, in view of the base of MorphoSys Securityholders in the United States and as a result of the MorphoSys ADSs being listed on the Nasdaq Global Select Market (the “Nasdaq”), the Offer must also comply with U.S. Offer Rules. In order to reconcile certain areas in which German law and U.S. law conflict, the Bidder applied for exemptive or no-action relief with the SEC prior to the publication of the Offer Document to conduct the Offer in the manner described in the Offer Document, which was granted by the SEC on 9 April 2024 (the “SEC Relief”), see Sec. 21 of the Offer Document.
The BaFin reviewed the German version of the Offer Document as legally required and permitted the publication of the Offer Document on 11 April 2024. On 11 April 2024, the Bidder published the German version of the Offer Document on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition
and made copies thereof available at Deutsche Bank Aktiengesellschaft, TAS, Post-IPO Services, Taunusanlage 12, 60325 Frankfurt am Main, Germany. MorphoSys Securityholders may request to obtain a copy of the Offer Document free of charge via email to dct.tender-offers@db.com or by fax: +49 69 910 38794, indicating a valid mailing address. In addition, the Bidder has made available a non-binding English translation of the Offer Document, which has not been reviewed by the BaFin, on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition.
Notice of the availability of copies of the German version of the Offer Document provided free of charge in Germany and the website at which the German version of the Offer Document is available, has been published by the Bidder on 11 April 2024 in the German Federal Gazette (Bundesanzeiger).
As required under the U.S. Exchange Act, after approval of the publication of the Offer Document by BaFin on the date of publication of the Offer Document the Bidder and Novartis AG filed with the SEC a so-called tender offer statement as exhibit to the Schedule TO, of which the Offer Document forms a part, and may file amendments thereto.
8
The Offer has not been approved or disapproved by the SEC or any state securities commission in the United States, nor has the SEC or any state securities commission in the United States passed upon the fairness or merits of, or upon the accuracy or adequacy of, the information contained herein. Any representation to the contrary is unlawful. The Offer will not be submitted to the review procedures of any other securities regulator outside of Germany and has not been approved or recommended by any such regulator, however, in the United States the Schedule TO has been filed with the SEC and published.
The Bidder does not intend to make the Offer as a public offer pursuant to the applicable law of any jurisdictions other than Germany and the United States.
| 2 | GENERAL INFORMATION ABOUT THIS STATEMENT |
| 2.1 | Legal Basis for this Statement |
Pursuant to Sec. 27 para. 1 sentence 1 WpÜG, the management board (Vorstand) of MorphoSys (the “Management Board”) and the supervisory board (Aufsichtsrat) of MorphoSys (the “Supervisory Board”) are each obliged to issue a reasoned statement (begründete Stellungnahme) on any takeover offer and any amendments thereto. The reasoned statement may be issued jointly by the management board (Vorstand) and the supervisory board (Aufsichtsrat) of a target company (Zielgesellschaft).
Pursuant to Sec. 27 para. 1 sentence 2 WpÜG, this Statement must, in particular, address
| (i) | The type and the amount of the consideration being offered; |
| (ii) | The expected consequences of a successful Offer for MorphoSys and the employees of MorphoSys and their representative bodies, on the terms and conditions of employment, and on the business locations of MorphoSys; |
| (iii) | The objectives pursued by the Bidder with the Offer; and |
| (iv) | The intention of each member of the Management Board and the Supervisory Board to accept the Offer with respect to any MorphoSys Shares that such member holds. |
Pursuant to Sec. 27 para. 2 WpÜG, the employees of the target company, where no works council (Betriebsrat) is established, such as MorphoSys, may directly submit their written comments on the takeover offer to the management board (Vorstand) of the target company.
| 2.2 | Joint Reasoned Statement by the Management Board and the Supervisory Board |
On 11 April 2024, the Offer Document was provided to the Management Board.
Upon receipt of the Offer Document, the Management Board made it available without delay to the Supervisory Board and since no works council has been established at the level of MorphoSys, to the employees of the MorphoSys Group.
The Management Board and the Supervisory Board have each decided to jointly issue and publish this joint reasoned statement (gemeinsame begründete Stellungnahme) pursuant to Sec. 27 para. 1 sentence 1 WpÜG on the Bidder’s Offer. The Management Board and the Supervisory Board, each individually, have discussed the Offer and this Statement and have each unanimously resolved to approve this Statement on 11 April 2024.
Furthermore, the English version of this Statement is being filed by MorphoSys with the SEC as an exhibit to its Solicitation/Recommendation Statement on Schedule 14D-9 filed pursuant to Sec. 14(d)(4) of the U.S. Exchange Act (the “Schedule 14D-9”) and MorphoSys will disseminate the Schedule 14D-9 to MorphoSys Securityholders to the extent required by applicable U.S. Offer Rules and any other applicable law.
9
| 2.3 | Certain Defined Terms and Meanings |
Except as expressly stated otherwise, references in this Statement to a certain time are references to the respective local time in Frankfurt am Main, Germany (“Frankfurt am Main Local Time”) and/or the local time in New York, New York, United States local time (“New York Local Time”).
The specification “EUR” relates to the Euro currency; “EURm” means million Euros; and “EURbn” means billion Euros.
The specification “USD” relates to the United States Dollar currency; “USDm” means million United States Dollars; and “USDbn” means billion United States Dollars.
Except as expressly stated otherwise, expressions such as “currently”, “at the present time”, “now”, “at present” or “today” references in this Statement refer to 11 April 2024, the date of publication of this Statement.
References in this Statement to a “Banking Day” refer to any day on which banks in Frankfurt am Main, Germany and New York, New York, United States, are open for general business with retail customers.
References in this Statement to the “Business Combination Agreement” refer to the Business Combination Agreement entered into on 5 February 2024 by Novartis AG, the Bidder and MorphoSys regarding a voluntary public takeover offer to be made by the Bidder to acquire all MorphoSys Shares (including all MorphoSys Shares represented by MorphoSys ADSs) for a cash consideration of EUR 68.00 per MorphoSys Share.
References in this Statement to an “FSE Trading Day” refer to a day on which the Frankfurt Stock Exchange (Frankfurter Wertpapierbörse) (the “FSE”) is open for trading.
References to “Germany” refer to the Federal Republic of Germany.
References to “Nasdaq Trading Day” refer to a day on which the Nasdaq is open for trading.
References to “United States” or “U.S. “ refer to the United States of America.
All references in this Statement to a “U.S. Working Day” refer to every day except for Saturdays, Sundays and U.S. federal public holidays.
References in this Statement to “best efforts” refer, in accordance with the terms of the Business Combination Agreement, to all such efforts which are from the perspective of a prudent business person reasonable and appropriate (alle wirtschaftlich vernünftigen und angemessenen Bemühungen unternehmen).
To the extent that this Statement contains references to websites on the Internet, these were last accessed by MorphoSys on 11 April 2024.
| 2.4 | Factual Basis for this Statement |
Except as expressly stated otherwise, any information contained in this Statement relating to the Bidder, the persons acting jointly with the Bidder, and the Offer, including the Bidder’s intentions, is based on the information contained in the Offer Document and other publicly available information. The Management Board and the Supervisory Board are not in a position to verify all of the information contained in the Offer Document or to influence the realization of the Bidder’s stated intentions. There can be no assurance that the intentions published in the Offer Document will be realized. Furthermore, Sec. 2.6 of the Offer Document sets forth that the Bidder will update the Offer Document (also with regard to any changed intentions) only to the extent required by either the German Offer Rules or U.S. Offer Rules and permitted under both the German Offer Rules and U.S. Offer Rules.
This Statement may contain hypertext links to information on MorphoSys’ website, Novartis AG’s website or other websites. The information on any such website is not incorporated by reference into this Statement and does not constitute a part of this Statement.
10
The Management Board and the Supervisory Board do not intend to update this Statement and disclaim any obligation to do so, unless required under German or U.S. law.
| 2.5 | Forward Looking Statements |
This Statement contains certain forward-looking statements concerning MorphoSys, the Bidder and the Takeover that involve substantial risks and uncertainties. Forward-looking statements include any statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “goal,” “may,” “might,” “plan,” “predict,” “project,” “seek,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue” and similar expressions. In this Statement, MorphoSys’ forward-looking statements include statements about the parties’ ability to satisfy the Offer Conditions (as defined below) to the consummation of the Takeover; statements about the expected timetable for the consummation of the Takeover; plans, objectives, expectations and intentions of MorphoSys and the Bidder; and the financial condition, results of operations and business of MorphoSys and Novartis AG.
The forward-looking statements contained in this Statement represent the judgment of MorphoSys as of the date of this Statement and involve known and unknown risks and uncertainties, which might cause the actual results, financial condition and liquidity, performance or achievements of MorphoSys, or industry results, to be materially different from any historic or future results, financial conditions and liquidity, performance or achievements expressed or implied by such forward-looking statements. In addition, even if MorphoSys’ results, performance, financial condition and liquidity, and the development of the industry in which it operates are consistent with such forward-looking statements, they may not be predictive of results or developments in future periods. Those risks and uncertainties that could cause the actual results to differ from expectations contemplated by forward-looking statements include, among other things: uncertainties as to the timing of the Takeover; uncertainties as to how many of MorphoSys Securityholders will tender their MorphoSys Securities in the Offer; the possibility that competing offers will be made; the possibility that various conditions for the Offer may not be satisfied or waived; the effects of the Takeover on relationships with employees, other business partners or governmental entities; that the Bidder and Novartis may not realize the potential benefits of the Takeover; transaction costs associated with the Takeover; that the MorphoSys’ expectations may be incorrect; the inherent uncertainties associated with competitive developments, clinical trial and product development activities and regulatory approval requirements; MorphoSys’ reliance on collaborations with third parties; estimating the commercial potential of MorphoSys’ development programs; and other risks indicated in the risk factors included in MorphoSys’ filings with the SEC, including MorphoSys’ Annual Report on Form 20-F, as well as this Statement and the so-called tender offer statement as exhibit to the Schedule TO and related Offer documents to be filed by the Bidder and Novartis. Given these uncertainties, the reader is advised not to place any undue reliance on such forward-looking statements. These forward-looking statements speak only as of the date of publication of this Statement. MorphoSys expressly disclaims any obligation to update any such forward-looking statements in this Statement to reflect any change in its expectations with regard thereto or any change in events, conditions or circumstances on which any such statement is based or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements, unless specifically required by law or regulation.
| 2.6 | No Statement by the Employees of MorphoSys |
Pursuant to Sec. 27 para. 2 WpÜG, the competent works council(s) of the target company (Zielgesellschaft) may provide the management board of a target company (Zielgesellschaft) with a statement on the offer which the management board of the target company (Zielgesellschaft) must, pursuant to Sec. 27 para 2 WpÜG attach to its own statement without prejudice to its obligation to publish its reasoned statement under Sec. 27 para. 3 sentence 1 WpÜG.
At the level of MorphoSys, no works council has been established. The Offer Document has therefore been made available directly to the employees of the MorphoSys Group through MorphoSys’ intranet.
11
As of the date of publication of this Statement, MorphoSys’ employees have not submitted a written statement in connection with the Offer. Hence, no statement of employees or any works council or other employee representative is attached to this Statement in accordance with Sec. 27 para. 2 WpÜG.
This Statement is, in accordance with Sec. 27 para. 3 sentence 2 WpÜG, simultaneously with its publication, being made available to the employees of the MorphoSys Group through MorphoSys’ intranet.
| 2.7 | Publication of this Statement and Additional Statements |
In accordance with Sec. 27 para. 3 and Sec. 14 para. 3 sentence 1 WpÜG, this Statement, as well as any supplements hereto and any additional statements made in connection with any amendments to the Offer (the “Supplemental Statements”) will be published on the internet at the website of MorphoSys in the German language at
https://www.morphosys.com/de/investoren/Novartis-TakeoverOffer
and in the English language at
https://www.morphosys.com/en/investors/Novartis-TakeoverOffer.
Copies of the German version of this Statement and German versions of any Supplemental Statements will be obtainable free of charge at MorphoSys AG, Investor Relations, Semmelweisstrasse 7, 82152 Planegg, Germany, tel.: +49 89 89927 404, email: investors@morphosys.com. The publication of the German version of this Statement and German versions of any Supplemental Statements as well as information about the free distribution of such statements will be announced by notice in the German Federal Gazette (Bundesanzeiger).
The English translation of this Statement is being filed by MorphoSys with the SEC as an exhibit to its Schedule 14D-9 and MorphoSys will disseminate the Schedule 14D-9 to MorphoSys Securityholders to the extent required by applicable U.S. Offer Rules and any other applicable law. A copy of the Schedule 14D-9, including the exhibits and any amendments or supplements thereto and other documents filed with the SEC may also be examined at the SEC after the filing of the Schedule 14D-9 with the SEC on the date of publication of this Statement, free of charge on the internet at the website of the SEC at
https://www.sec.gov/edgar/browse/?CIK=1340243.
Furthermore, the Schedule 14D-9 and any amendments or supplements will be available on the internet at the website of Novartis AG at
https://www.novartis.com/investors/morphosys-acquisition
as well as of MorphoSys at
https://www.morphosys.com/en/investors/Novartis-TakeoverOffer
and copies of the Schedule 14D-9 and any amendments or supplements will be obtainable free of charge by contacting MorphoSys AG, Semmelweisstrasse 7, 82152 Planegg, Germany, tel.: +49 89 89927 404, email: investors@morphosys.com.
| 2.8 | Responsibility of MorphoSys Securityholders |
The description of the Offer contained in this Statement does not purport to be complete. Therefore, the terms and conditions of the Offer Document alone are authoritative and decisive as regards the content and implementation of the Offer. The assessments and recommendations of the Management Board and the Supervisory Board contained in this Statement are not binding upon the MorphoSys Securityholders. Where and to the extent this Statement refers to the Offer or the Offer Document or quotes, summarizes or reproduces portions of them, such statements constitute mere references for information purposes. The
12
Management Board and the Supervisory Board neither adopt the Offer or the Offer Document as their own in any way nor assume any liability for the correctness, accuracy or completeness of the Offer and the Offer Document.
It will be the responsibility of the individual MorphoSys Securityholders to review the Offer Document. MorphoSys Securityholders who accept or do not accept the Offer will be responsible for fulfilling the requirements and conditions that apply to MorphoSys Securityholders as set forth in the Offer Document.
In this Statement, the Management Board and the Supervisory Board have not taken into account the individual circumstances of each MorphoSys Securityholder (including their personal tax situation). Ultimately, each MorphoSys Securityholder must make their own independent decision whether and, if so, to what extent to accept the Offer, taking into account the overall circumstances, their individual situation (including their individual tax situation), and their personal assessment of the possibilities of the future development of the value and share price of MorphoSys Securities. In particular, the specific tax situation of each MorphoSys Securityholder may in individual cases result in valuations different from those presented by the Management Board and the Supervisory Board. The Management Board and the Supervisory Board recommend that each MorphoSys Securityholder seeks individual tax and legal advice from their own tax and legal advisors.
MorphoSys Securityholder who wish to accept the Offer should examine whether accepting the Offer is compatible with any legal obligations that may arise from their personal circumstances (e.g., security interests in the MorphoSys Securities or selling restrictions). It is not practicable for the Management Board and the Supervisory Board to examine each MorphoSys Securityholder’s individual obligations or to take such obligations into account in making their recommendation. The Management Board and the Supervisory Board recommend that all persons who receive the Offer Document outside Germany or the United States or who wish to accept the Offer but are subject to the securities laws of any jurisdiction other than Germany and the United States, inform themselves of the applicable securities laws and act in compliance therewith.
The Management Board and the Supervisory Board do not assume any responsibility for the decisions of MorphoSys Securityholders and their consequences.
| 2.9 | MorphoSys Securityholders in the United States |
This Statement is made in accordance with the statutory requirements of Germany. In addition, MorphoSys is filing the English translation of this Statement with the SEC as an exhibit to the Schedule 14D-9. The Management Board and the Supervisory Board advise MorphoSys Securityholders with place of residence, seat or place of habitual abode in the United States that this Statement has been prepared in accordance with a format and structure generally customary in Germany, which differs from the format and structure customary for a so-called Solicitation/Recommendation Statement in the United States.
Neither the SEC nor any state securities commission in the United States have approved or disapproved this Statement or reviewed this Statement prior to its publication.
| 3 | INFORMATION ABOUT MORPHOSYS |
| 3.1 | General Information on MorphoSys |
MorphoSys was founded in 1992 by Dr. Simon Moroney and Prof. Dr. Andreas Plückthun. Today, MorphoSys is organized as a stock corporation (Aktiengesellschaft) established under the laws of Germany. Its company name (Firma) is MorphoSys AG.
The corporate purpose of MorphoSys is the identification, research, optimization, development, application, commercialization and marketing and distribution of technologies, processes and products in
13
the field of pharmaceuticals, active pharmaceutical ingredients and corresponding intermediates and the provision of related services.
MorphoSys’ financial year is the calendar year.
MorphoSys’ registered business address is at Semmelweisstrasse 7, 82152 Planegg, Germany (tel.: +49 89 899270).
| 3.2 | Capital and Shareholder Structure of MorphoSys |
Share Capital
The share capital (Grundkapital) registered at the time of the publication of this Statement in the commercial register (Handelsregister) of MorphoSys amounts to EUR 37,655,137.00 and is divided into 37,655,137 bearer MorphoSys Shares with no-par value (auf den Inhaber lautende Stückaktien). There are no other classes of shares. The share capital has been fully paid up, each representing a notional interest in the share capital of EUR 1.00. The MorphoSys Shares were created pursuant to the laws of Germany and are denominated in Euro. MorphoSys holds 53,685 MorphoSys Shares as treasury shares.
According to the publication of the total number of voting rights in MorphoSys pursuant to Sec. 41 of the German Securities Trading Act (Wertpapierhandelsgesetz – “WpHG”) on 2 April 2024, the total number of voting rights based on MorphoSys Shares amounted to 37,716,423 on 31 March 2024. The corresponding increase of the share capital from EUR 37,655,137.00 by EUR 61,286.00 to EUR 37,716,423.00 is due to the issue of subscription shares from the Conditional Capital 2016-III (as defined below). Pursuant to Sec. 201 of the German Stock Corporation Act (Aktiengesetz – “AktG”), the Management Board will file an application for the registration of the issuance of the subscription shares with the commercial register (Handelsregister) by no later than at the end of January 2025.
Since 31 March 2024 no new MorphoSys Shares have been issued. Therefore, at the time of publication of this Statement, MorphoSys’ share capital amounts to EUR 37,716,423.00 and is divided into 37,716,423 no-par value bearer shares (auf den Inhaber lautende Stückaktien), each representing a notional interest in the share capital of EUR 1.00.
Shareholder Structure
To the knowledge of MorphoSys on the basis of the notifications made in accordance with the WpHG and information provided to MorphoSys (as specified in the table below), as of 10 April 2024, 16:00 hours Frankfurt am Main Local Time, the shareholders listed below hold, directly or indirectly, 3% or more of the voting rights from MorphoSys Shares:
| Direct or Indirect Holdings in MorphoSys | ||
Shareholders | Share(1) | |
The Goldman Sachs Group, Inc. | 14.55% | |
UBS Group AG | 12.74% | |
Morgan Stanley | 12.38% | |
Tang, Yue | 11.12% | |
JPMorgan Chase & Co. | 10.07% | |
Barclays Capital Securities Limited | 7.91% | |
Bank of America Corporation | 6.80% | |
Kynam Global Healthcare Offshore Fund, Ltd | 5.33% | |
BlackRock, Inc. | 5.29% | |
DWS Investments GmbH | 4.34% | |
Legoretta, Pablo | 3.91% | |
Royalty Pharma PLC | 3.91% |
| (1) | Including voting rights attributable to instruments within the meaning of Sec. 38 WpHG. |
14
Historic Stock Exchange Prices
The table below sets forth, for the calendar periods indicated, the high and low German closing prices for the MorphoSys Shares (all stock exchanges, including the XETRA electronic trading system of the FSE), and the high and low closing prices per MorphoSys ADS as reported on the Nasdaq.
| Price of a MorphoSys Share in EUR | Price of a MorphoSys ADS in USD | |||||||
| 2022 | High | Low | High | Low | ||||
1st Quarter | 34.26 | 21.55 | 9.73 | 5.87 | ||||
2nd Quarter | 26.91 | 17.27 | 7.39 | 4.52 | ||||
3rd Quarter | 24.03 | 16.54 | 6.14 | 4.14 | ||||
4th Quarter | 23.35 | 12.05 | 5.74 | 3.21 | ||||
| 2023 | ||||||||
1st Quarter | 19.33 | 13.21 | 5.22 | 3.56 | ||||
2nd Quarter | 28.00 | 14.46 | 7.74 | 3.95 | ||||
3rd Quarter | 31.13 | 25.83 | 8.60 | 6.72 | ||||
| 4th Quarter | 35.40 | 16.09 | 9.90 | 4.55 | ||||
| 2024 | ||||||||
1st Quarter | 67.22 | 30.75 | 18.18 | 8.60 | ||||
2nd Quarter (through 4 April 2024) | 67.25 | 67.20 | 18.11 | 18.06 | ||||
Source: Bloomberg as of 19 March 2024
Authorized Capital
The Management Board, with the approval of the Supervisory Board, is authorized to increase the share capital of MorphoSys in one or several tranches up until 16 May 2028 by up to EUR 6,846,388.00 against cash and/or non-cash contributions by issuing up to 6,846,388 new MorphoSys Shares (Authorized Capital 2023-I). The shareholders of MorphoSys are generally entitled to subscription rights; however, the subscription rights may, with the Supervisory Board’s consent, be excluded in certain specified cases, which are in line with market practice.
The Management Board is further authorized to increase MorphoSys’ share capital with the approval of the Supervisory Board, in each case in one or several tranches:
| (i) | by up to EUR 42,715.00 until 30 April 2024 (Authorized Capital 2019-I); |
| (ii) | by up to EUR 41,552.00 until 18 May 2026 (Authorized Capital 2021-III); and |
| (iii) | by up to EUR 1,978,907.00 until 17 May 2027 (Authorized Capital 2022-I), |
in each case by issuing a corresponding number of new MorphoSys Shares against cash or non-cash contribution. In each case, subscription rights are excluded. The authorized capital may in each case be used to grant MorphoSys Shares to directors, officers and employees of MorphoSys US Inc. under a restricted stock unit program (RSUP 2019, 2021 and 2022).
Conditional Capital
According to the articles of association of MorphoSys, its share capital is conditionally increased (i) by up to EUR 2,475,437.00 (Conditional Capital 2016-I) and (ii) by up to EUR 3,289,004.00 (Conditional Capital 2021-I), in each case solely to be used for granting new MorphoSys Shares to holders of conversion or option rights. The conditional capital increase may only be carried out to the extent that the holders of conversion or option rights exercise their conversion or option rights or fulfill conversion obligations arising from such bonds.
15
Furthermore, according to the articles of association of MorphoSys, its share capital is conditionally increased (i) by up to EUR 416,297.00 (“Conditional Capital 2016-III”) and (ii) by up to EUR 507,668.00 (Conditional Capital 2020-I), in each case for the sole purpose of fulfilling certain subscription rights. The conditional capital increase may only be carried out to the extent that holders of specified subscription rights exercise their right to subscribe for MorphoSys Shares. As a result of the aforementioned issuance of subscription shares, the remaining Conditional Capital 2016-III amounts to EUR 355,011.00.
Convertible Bonds
MorphoSys placed non-subordinated, unsecured convertible bonds in 2020 for a nominal amount of EUR 325,000,000, equal to 3,250 bonds with a nominal amount of EUR 100,000 each, and maturing on 16 October 2025 (ISIN DE000A3H2XW6). On 30 March 2023, MorphoSys repurchased outstanding convertible bonds via a modified reverse Dutch auction procedure. At the close of the modified reverse Dutch auction procedure, MorphoSys agreed to repurchase bonds representing EUR 62,900,000 in aggregate principal amount (approximately 19.35% of the outstanding principal amount). The purchase price per EUR 100,000 nominal was EUR 64,000. At the time of the publication of this Statement, convertible bonds with a nominal amount of EUR 262,100,000, with an interest rate of 0.625% per annum, are outstanding (the “Convertible Bonds”).
The Convertible Bonds provide for a right of the holders to conditionally convert their Convertible Bonds into MorphoSys Shares subject to the successful completion of the Offer (the “Conditional Conversion Right”), thereby allowing, but not requiring, the holders of Convertible Bonds to tender the underlying MorphoSys Shares into the Offer during the Additional Acceptance Period (as defined below). Additionally, following the consummation of the Takeover (the “Closing”), the holders of the Convertible Bonds have a right to request an early redemption of their Convertible Bonds at par plus accrued but unpaid interest.
The conversion price under the Convertible Bonds amounts to EUR 131.29. For purposes of the Conditional Conversion Right, this conversion price will be adjusted according to the calculation formula set out in the terms and conditions of the Convertible Bonds. Under the terms and conditions of the Convertible Bonds, the relevant date for the adjustment of the conversion price is the day of the publication of the tender results after the end of the Acceptance Period (as defined below) pursuant to Sec. 23 para. 1 no. 2 WpÜG. Subject to any extension of the Acceptance Period, this publication is expected to be made on 16 May 2024. On this date, the adjusted conversion price will be EUR 117.9105. Due to the calculation formula set out in the terms and conditions of the Convertible Bonds, the adjusted conversion price would be higher if the announcement pursuant to Sec. 23 para. 1 no. 2 WpÜG is made at a later point in time.
In case all holders of the Convertible Bonds were to convert their Convertible Bonds into MorphoSys Shares, a maximum number of 2,222,872 MorphoSys Shares would be issued. However, because the conversion price is, even after the conversion price adjustment, substantially higher than the Offer Price per MorphoSys Share under the Offer, it is expected that holders of Convertible Bonds will refrain from executing their conversion right.
Long Term Incentive Programs
MorphoSys is party to the following long term incentive programs:
| (i) | Several stock option programs for the members of the Management Board, members of management bodies of MorphoSys Affiliates (as defined below) and selected senior managers and employees of MorphoSys Affiliates, under which subscription rights for MorphoSys Shares are granted to the beneficiaries. These stock option programs entitle the relevant beneficiaries, subject to the achievement of certain performance targets and the expiry of a four (4)-year waiting period, |
16
| to acquire newly issued MorphoSys Shares at a certain exercise price whereby one (1) option to purchase MorphoSys Shares entitles the relevant beneficiary to the subscription of one (1) MorphoSys Share (collectively, the “Stock Option Programs”). |
| (ii) | Several performance share unit programs for the members of the Management Board as well as selected senior managers and employees of MorphoSys and MorphoSys Affiliates, under which Performance Share Units are granted to the beneficiaries, which, subject to the achievement of certain performance targets and the expiry of a four (4)-year waiting period, entitle such beneficiaries to a payment claim against MorphoSys corresponding to MorphoSys Share price at the end of the waiting period which is generally settled in cash (collectively, the “Performance Share Unit Programs”). MorphoSys will also be party to a Performance Share Unit Program for 2024 (the “2024 Performance Share Unit Program”). |
| (iii) | Several restricted stock unit programs for senior managers and employees (including directors and officers) of certain MorphoSys Affiliates in the United States, under which Restricted Stock Units are granted to the beneficiaries, which, subject to the achievement of certain performance targets and the expiry of a three (3)-year waiting period, entitle such beneficiaries to a cash payment claim against MorphoSys which is generally settled in MorphoSys Shares, whereby one (1) Restricted Stock Unit Award entitles such beneficiaries to the issuance of one (1) MorphoSys Share(collectively, the “Restricted Stock Unit Programs”). MorphoSys will also be party, subject to the terms set forth in the Disclosure Letter, to a Restricted Stock Unit Program for 2024 (the “2024 Restricted Stock Unit Program”). |
The Stock Option Programs, the Performance Share Unit Programs (including the 2024 Share Unit Program) and the Restricted Stock Unit Programs (including the 2024 Restricted Stock Unit Program) are also collectively referred to as the “Incentive Plans”; and the 2024 Performance Share Unit Program and the 2024 Restricted Stock Unit Program are also collectively referred to as the “2024 Incentive Plans”.
The aggregate economic value of Performance Share Units and Restricted Share Units issued under the 2024 Incentive Plans were, pursuant to the terms of the Business Combination Agreement, required to not exceed the economic value of the awards granted under the Performance Share Unit Program for 2023 and the Restricted Share Unit Program for 2023, in the aggregate, adjusted for a 3.5% salary budget increase. Instruments issued under the 2024 Incentive Plans will not provide for accelerated vesting of such instruments as a result of the consummation of the Takeover, but will provide for accelerated vesting of such instruments as a result of involuntary or constructive termination of the holder’s employment as of or within two (2) years following the Closing.
As of the date of publication of this Statement (i) 271,887 stock options were granted and outstanding under the Incentive Plans (each, a “Stock Option”), (ii) 908,548 restricted stock units (each, a “Restricted Stock Unit”) were granted and outstanding under the Incentive Plans, and (iii) 2,197,439 performance share units (each, a “Performance Share Unit”) were granted and outstanding under the Incentive Plans.
17
| 3.3 | Listings of MorphoSys |
The MorphoSys Shares are admitted to trading on the regulated market (Regulierter Markt) with additional post-admission obligations (Prime Standard) of the FSE under ISIN DE0006632003 under the symbol “MOR”. In addition, the MorphoSys Shares are traded on the regulated unofficial market (Freiverkehr) of the stock exchange in Berlin as well as on the unregulated market on the stock exchanges of Düsseldorf, Hamburg, Hanover, Munich and Stuttgart as well as via Tradegate Exchange. The MorphoSys Shares are currently included in the SDAX and TecDAX, both indices calculated by Deutsche Börse AG and/or its relevant affiliates. In addition, the MorphoSys ADSs are listed on Nasdaq under the symbol “MOR”.
| 3.4 | Management Board and the Supervisory Board of MorphoSys |
The Management Board currently consists of the following persons:
| • | Jean-Paul Kress, M.D., Chief Executive Officer (Vorstandsvorsitzender); and |
| • | Lucinda Crabtree, Ph.D., Chief Financial Officer (Finanzvorstand). |
The Supervisory Board consists of six (6) members and is currently composed of the following persons:
| • | Marc Cluzel, M.D., Ph.D., chairman of the Supervisory Board (Aufsichtsratsvorsitzender); |
| • | George Golumbeski, Ph.D., deputy chairman of the Supervisory Board (stellvertretender Aufsichtsratsvorsitzender); |
| • | Krisja Vermeylen, member of the Supervisory Board (Aufsichtsratsmitglied); |
| • | Michael Brosnan, member of the Supervisory Board (Aufsichtsratsmitglied); |
| • | Sharon Curran, member of the Supervisory Board (Aufsichtsratsmitglied); and |
| • | Andrew Cheng, M.D., Ph.D., member of the Supervisory Board (Aufsichtsratsmitglied). |
| 3.5 | Group Structure and Business Activities of MorphoSys |
Corporate Structure
MorphoSys is the parent company of MorphoSys Group. As of the date of this Statement, MorphoSys Group comprises MorphoSys AG and its three wholly-owned subsidiaries MorphoSys US Inc., Constellation Pharmaceuticals, Inc. and Constellation Securities Corp, which are affiliates (verbundene Unternehmen) of MorphoSys within the meaning of the German stock corporation law (each, a “MorphoSys Affiliate”). The following table provides an overview of the subsidiaries and the participation held by MorphoSys as of the date of this Statement:
Legal name | Registered Office | Business Area | Direct and/or indirect interest | |||||
MorphoSys US Inc. | Dover, Delaware, United States | Commercialization and selling of products in the field of medicines, pharmaceutical compounds and related intermediate products, as well as operation of all businesses necessary and measures related thereto | 100.00 | % | ||||
18
Legal name | Registered Office | Business Area | Direct and/or indirect interest | |||||
| Constellation Pharmaceuticals, Inc. | Dover, Delaware, United States | Clinical-stage biopharmaceutical company developing novel therapeutics for patients with cancers associated with abnormal gene expression or drug resistance, as well as operation of all business necessary and measures related thereto | 100.00 | % | ||||
| Constellation Securities Corporation | Boston, Massachusetts, United States | Engaged exclusively in buying, selling, dealing in, or holding securities on its own behalf for investment purposes | 100.00 | % | ||||
Business Activities
MorphoSys is a global biotechnology company focused on the development and commercialization of innovative cancer medicines. In 2023, MorphoSys advanced its mid-to-late-stage oncology pipeline, focusing its efforts on pelabresib, an investigational bromodomain and extra-terminal domain (“BET”) inhibitor, in first-line myelofibrosis – a debilitating and deadly blood cancer.
MorphoSys’ current clinical programs are:
| (i) | Pelabresib: An investigational selective small-molecule therapy aimed at promoting anti-tumor activity. It is designed to inhibit BET proteins, which may downregulate genes implicated in blood cancers. The Phase 3 MANIFEST-2 study is currently investigating pelabresib in combination with the janus kinase (“JAK”) inhibitor ruxolitinib compared with placebo plus ruxolitinib in JAK inhibitor-naïve patients with myelofibrosis (those who have not been previously treated with a JAK inhibitor). Additionally, it is being explored in the Phase 2 MANIFEST trial in patients with myelofibrosis and high-risk essential thrombocythemia; and |
| (ii) | Tulmimetostat: An investigational next-generation dual inhibitor of Enhancer of Zeste Homolog 2 (EZH2) and EZH1 designed to counter abnormal gene activity that may contribute to cancer development and progression, as well as drug resistance. Tulmimetostat is being investigated in a Phase 1/2 study in patients with advanced solid tumors and lymphomas, including ARID1A-mutated endometrial carcinoma and ovarian clear cell carcinoma, diffuse large B-cell lymphoma, peripheral T-cell lymphoma, BAP1-mutated mesothelioma and metastatic castration-resistant prostate cancer. |
MorphoSys has a world-class team of multi-disciplinary experts based in Munich and Boston, and its senior experts have deep experience and capabilities in biology, chemistry, product discovery, clinical development and commercialization.
On 31 December 2023, the MorphoSys Group had 524 employees (31 December 2022: 629). The MorphoSys Group employed an average of 564 employees in 2023 (2022: 647).
| 3.6 | Strategy of MorphoSys |
MorphoSys is focused on the development and commercialization of innovative cancer medicines, advancing its mid-to-late-stage oncology pipeline in areas where there is a dire need for more effective and well-tolerated therapies.
19
Advancing Clinical Development Programs
MorphoSys believes that a focus on proprietary drug development and commercialization offers the best path to delivering growth and long-term success. MorphoSys is currently furthering the clinical development of its investigational medicines pelabresib and tulmimetostat. To advance these clinical development programs, MorphoSys invests a significant portion of its financial resources.
Pelabresib, MorphoSys’ investigational BET inhibitor, is potentially a foundational first-line treatment for myelofibrosis. Comprehensive Phase 3 MANIFEST-2 results, presented at the American Society of Hematology (ASH) 2023 Annual Meeting and Exposition in December 2023, demonstrated that the combination of pelabresib and the JAK inhibitor ruxolitinib improves all four hallmarks of myelofibrosis – spleen size, anemia, bone marrow fibrosis and disease-associated symptoms – versus placebo plus ruxolitinib, which is the standard of care in myelofibrosis. MorphoSys is confident that the MANIFEST-2 data package – complemented by long-term Phase 2 results – will provide impactful evidence to regulatory agencies as MorphoSys pursues approval for patients with myelofibrosis. As myelofibrosis represents a multi-billion-dollar market opportunity, this endeavor remains MorphoSys’ number one priority. Beyond myelofibrosis, pelabresib has demonstrated potential clinical benefit in early-stage trials in other myeloid diseases – providing additional growth opportunities.
Tulmimetostat was designed to improve on first-generation EZH2 inhibitors through increased potency, longer residence time on target and a longer half-life, offering the potential for enhanced anti-tumor activity. It is currently being evaluated in a Phase 1/2 trial for advanced solid tumors and lymphomas. Updated results from the Phase 2 portion of the study were presented at American Society of Clinical Oncology (ASCO) 2023 Annual Meeting. The data suggest responses or disease stabilization across all solid tumor cohorts, including those with heavily pre-treated patients. Notably, complete and partial responses were also observed in the lymphoma cohort and partial responses were observed in the ARID1A mutated and BAP1 mutated cohorts. In September 2023, the U.S. Food and Drug Administration (“FDA”) granted Fast Track designation for tulmimetostat for the treatment of patients with advanced, recurrent or metastatic ARID1A-mutated endometrial cancer who have progressed on at least one prior line of treatment.
Leveraging Strong Partnerships
MorphoSys’ key partner programs, developed via its legacy antibody technology platform, continue to mature and have the potential to generate significant value. These include ianalumab (Sjögren’s syndrome, lupus nephritis and other autoimmune diseases), abelacimab (venous thromboembolism prevention), setrusumab (osteogenesis imperfecta), bimagrumab (adult obesity) and felzartamab (autoimmune diseases, multiple myeloma). If approved by regulatory authorities, MorphoSys would receive milestone payments and royalties from these programs.
| 3.7 | Selected Key Financial Figures of MorphoSys |
MorphoSys’ audited consolidated financial statements comply with both the International Financial Reporting Standards (“IFRS”) published by the International Accounting Standards Board (“IASB”) and those adopted by the European Union. The audited consolidated financial statements also take into account the supplementary provisions under commercial law, which must be applied in accordance with Sec. 315e para. 1 of the German Commercial Code (Handelsgesetzbuch, HGB).
This section outlines relevant recent and historic financial information in relation to MorphoSys, derived from MorphoSys’ audited consolidated financial statements for the financial year 2023. It shows the recent development of MorphoSys’ business by comparing key financial information for MorphoSys’ financial years 2023 and 2022.
20
| 3.7.1 | Operating Results |
Revenues
Revenues in the financial year 2023 decreased by 14% or EURm 40.0 to EURm 238.3 compared to the previous year (2022: EURm 278.3).
Success-based payments including royalties accounted for 50% or EURm 119.2 (2022: 37% or EURm 103.1) of total revenues. On a regional basis, MorphoSys generated 89% or EURm 211.5 of its commercial revenues from product sales and with biopharmaceutical companies in the U.S. and 11% or EURm 26.8 from customers primarily located in Europe (excluding Germany) and Asia. In the financial year 2022, these percentages were 89% (EURm 248.9) and 11% (EURm 29.3), respectively.
Cost of Sales
Cost of sales increased from EURm 48.6 in the financial year 2022 to EURm 58.4 in the financial year 2023, mainly due to impairments in the amount of EURm 11.9 (2022: EURm 0.0), relating to the recognition of the inventory obsolescence reserve and scrapping of inventories. Acquisition and production costs of inventories slightly increased to EURm 30.7 in the financial year 2023 (2022: EURm 28.8). In addition, amortization and other expenses for intangible assets increased from EURm 9.8 in the financial year 2022 to EURm 10.7 in the financial year 2023. This was offset by a decrease in personnel costs from EURm 9.5 in the financial year 2022 to EURm 8.2 in the financial year 2023.
Gross Profit
Gross Profit amounted to EURm 179.9 in the financial year 2023 (2022: EURm 229.6). This decrease resulted mainly from lower revenues from licenses.
Operating Expenses
In the financial year 2023, operating expenses decreased by 4%, or EURm 18.0, to EURm 432.4 compared to EURm 450.4 in the financial year 2022. The year-over-year decrease resulted mainly from a decrease in expenses for external services and lower expenses for consumables. The decrease was partially offset by higher personnel expenses in the financial year 2023.
Research and development expenses decreased by 5%, or EURm 14.2, to EURm 283.6 in the financial year 2023 compared to the previous year (2022: EURm 297.8). The year-over-year decrease mainly resulted from lower expenses for external laboratory and consulting services and lower expenses for consumables and were partially compensated by higher personnel related expenses.
The combined expenses for selling and general and administration amounted to EURm 147.2 in the financial year 2023 (2022: EURm 152.5). This total mainly includes personnel expenses of EURm 83.0 (2022: EURm 81.0) and expenses for external services of EURm 47.4 (2022: EURm 54.4).
In the financial year 2023, selling expenses amounted to EURm 81.4 compared to EURm 92.4 in the financial year 2022. The decrease by 12%, or EURm 11.0, was due to streamlining and focusing of selling efforts.
General and administrative (“G&A”) expenses increased by 9%, or EURm 5.7, from EURm 60.1 in the financial year 2022 to EURm 65.8 in the financial year 2023. The major driver for this increase were higher personnel expenses that were partly offset by lower expenses for external services.
In the financial year 2023, a goodwill impairment loss of EURm 1.6 was recognized.
21
Research and Development
Research and development (“R&D”) expenses decreased by 5%, or EURm 14.2, to EURm 283.6 in the financial year 2023 compared to the previous year (2022: EURm 297.8). Expenses for external laboratory services and legal and scientific consulting services decreased from EURm 198.1 in financial year 2022 to EURm 170.9 in the financial year 2023. This reflects the current clinical study progress as well as the prioritization activities relating to MorphoSys’ R&D portfolio.
Personnel expenses increased from EURm 65.0 in the financial year 2022 to EURm 80.2 in the financial year 2023, mainly driven by the increase in share-based payment expenses due to the increase in price of MorphoSys Shares, which is the valuation basis for the share-based payment programs. Expenses for intangible assets increased to EURm 16.3 in the financial year 2023 (2022: EURm 14.8). In the financial year 2023 the expense was impacted by an impairment loss amounting to EURm 8.9 relating to the write-off of a license. In the financial year 2022, these were influenced in particular by impairment losses of EURm 7.8 in connection with an impairment of an internally generated intangible asset under development. Depreciation, amortization and other expenses for infrastructure increased from EURm 10.8 in the financial year 2022 to EURm 11.0 in the financial year 2023. In contrast, expenses for consumables decreased from EURm 3.8 in the financial year 2022 to EURm 0.3 in the financial year 2023. In addition, Other expenses decreased from EURm 5.4 in the financial year 2022 to EURm 4.9 in the financial year 2023.
Selling
Selling expenses decreased by 12%, or EURm 11.0, to EURm 81.4 in the financial year 2023 (2022: EURm 92.4). This item mainly includes personnel expenses of EURm 39.8 (2022: EURm 48.6) and expenses for external services of EURm 32.7 (2022: EURm 35.8). The decrease in selling expenses is based on the ongoing measures to streamline and focus sales efforts.
General and Administrative (G&A)
G&A expenses increased by 9%, or EURm 5.7, in the financial year 2023 and amounted to EURm 65.8 (2022: EURm 60.1). The increase was mainly due to higher personnel expenses that amounted to EURm 43.2 in the financial year 2023 (2022: EURm 32.5). In contrast, expenses for external services declined to EURm 14.6 (2022: EURm 18.6) and depreciation, amortization and other expenses for infrastructure decreased from EURm 5.0 in the financial year 2022 to EURm 3.7 in the financial year 2023.
Impairment of Goodwill
In the financial year 2023, an impairment of goodwill in the amount of EURm 1.6 was recorded, which initially resulted from an acquisition in financial year 2010 (2022: EURm 0.0).
Other Income
Other income decreased by 58%, or EURm 7.0, to EURm 5.0 in the financial year 2023 (2022: EURm 12.0) and mainly resulted from lower exchange rate gains of EURm 3.2 (2022: EURm 11.4).
Other Expenses
In the financial year 2023, other expenses decreased by 54%, or EURm 8.5, from EURm 15.6 in the financial year 2022 to EURm 7.1 in the financial year 2023. This decrease was mainly the result of lower exchange rate losses of EURm 6.3 (2022: EURm 15.0).
22
Finance Income
Finance income decreased by 48%, or EURm 198.7, to EURm 213.4 in the financial year 2023 (2022: EURm 412.1) and mainly resulted from items amounting to EURm 115.6 (2022: EURm 361.4) in connection with the changes in plan assumptions of financial assets and financial liabilities from collaborations.
Finance Expenses
Finance expenses decreased by 14%, or EURm 23.9, to EURm 142.0 in the financial year 2023 (2022: EURm 165.9). This decrease was mainly due to the effects from financial liabilities from future payments to Royalty Pharma of EURm 107.2 (2022: EURm 81.2) resulting from differences between planning assumptions and actual figures, foreign currency effects and the application of the effective interest method.
Income Tax Benefits / Expenses
MorphoSys Group recorded total income tax benefit of EURm 1.2 in the financial year 2023 (2022: income tax expense of EURm 168.6), which consisted of current tax income of EURm 1.5 (2022: current tax expense of EURm 0.6) and deferred tax expense of EURm 0.3 (2022: deferred tax expense of EURm 168.0). The effective income tax rate was 0.6% in the financial year 2023 (2022: 962.2%). No deferred tax has been recognized in the current financial year, as the conditions for not recognizing a net asset as at 31 December 2023 are still met.
Consolidated Net Profit/Loss for the Period
In the financial year 2023, the consolidated net loss amounted to EURm 189.7 (2022: consolidated net loss of EURm 151.1).
| 3.7.2 | Liquidity and Capital Resources |
On 31 December 2023, MorphoSys Group had cash and investments of EURm 680.5, compared to EURm 907.2 on 31 December 2022.
Cash Flows
Net Cash Provided by/ Used in Operating Activities
In the financial year 2023, net cash used in operating activities amounted to EURm 295.8 and was mainly attributable to the consolidated net loss of EURm 189.7 (2022: consolidated net loss EURm 151.1) and changes in non-cash items. The consolidated net loss resulted both in the financial years 2023 and 2022 mainly from expenses incurred to finance MorphoSys’ ongoing operations, specifically cost of sales, research and development expenses, selling expenses, and general and administrative expenses.
Net Cash Provided by/ Used in Investing Activities
In the financial year 2023, net cash provided by investing activities amounted to EURm 15.4. This results from EURm 18.2 of interest received, EURm 4.4 cash receipts from sales of investments accounted at fair value through other comprehensive income, EURm 4.6 cash receipts from sales of shares of investment in associates, as well as proceeds from the sale of other financial assets amounting to EURm 3,142.3. These were offset by payments to acquire other financial assets amounting to EURm 3,151.2 and by payments to acquire tangible and intangible assets of EURm 2.9.
23
Net Cash Provided by/ Used in Financing Activities
Net cash provided by financing activities amounted to EURm 43.0 in the financial year 2023 and was primarily driven by the capital increase in December 2023 that resulted in net cash inflows of EURm 96.0. This was partially compensated by cash payments for repurchases of own convertible bonds including transaction costs of EURm 40.8.
Investments
In the financial year 2023, MorphoSys invested EURm 0.4 in property, plant and equipment (2022: EURm 1.9), mainly office and laboratory equipment (i.e., machinery). Depreciation of property, plant and equipment in the financial year 2023 decreased to EURm 2.3 compared to the previous year (2022: EURm 2.9).
MorphoSys invested EURm 2.5 in intangible assets in the financial year 2023 (2022: EURm 13.3). Of this amount, EURm 2.4 was spent on internally generated intangible assets. Amortization of intangible assets amounted to EURm 3.6 in the financial year 2023 (2022: EURm 3.6). In the financial year 2023, impairment losses of EURm 9.6 were recognized on intangible assets, thereof EURm 0.7 on internally generated intangible assets.
Assets
At EURm 2,026.3, total assets as of 31 December 2023, were EURm 370.6 lower compared to 31 December 2022 (EURm 2,396.9).
Current assets decreased in the financial year 2023 by EURm 275.0 to EURm 814.0 compared to the previous year. As of 31 December 2023, cash and cash equivalents amounted to EURm 158.5 compared to EURm 402.4 as of 31 December 2022. This change was mainly due to the consumption of cash for operations in the financial year 2023.
Non-current assets decreased in the financial year 2023 by EURm 95.6 from a balance of EURm 1,307.9 as of 31 December 2022 to EURm 1,212.3 as of 31 December 2023, mainly due to a EURm 42.5 decrease in intangible assets as well as a decrease of “Goodwill” by EURm 13.9, resulting from the change in the EUR/USD exchange rate compared to 31 December 2022.
Liabilities
Current liabilities decreased by EURm 14.0 from EURm 278.3 in the financial year 2022 to EURm 264.3 as of 31 December 2023, mainly as a result of EURm 47.5 decrease in the line item “accounts payable and accruals”. Opposing effect was a EURm 19.4 increase of the current contract liability as well as a EURm 17.6 increase in the current portion of liabilities from future payments to Royalty Pharma in the financial year 2023.
Non-current liabilities (31 December 2023: EURm 1,713.0; 31 December 2022: EURm 1,961.2) decreased mainly due to a reduction in the non-current portion of financial liabilities from collaborations from EURm 217.8 as of 31 December 2022 to EURm 108.9 as of 31 December 2023.
| 4 | INFORMATION ABOUT THE BIDDER AND NOVARTIS |
The following information has been published by the Bidder in the Offer Document. This information has not been verified by the Management Board or the Supervisory Board.
24
| 4.1 | General Information |
Bidder
According to the information provided by the Bidder in Sec. 6.1 of the Offer Document, the Bidder is a stock corporation (Aktiengesellschaft) established under the laws of Switzerland with registered office in Basel, Switzerland and being registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-477.907.492. On the date of the announcement of the Bidder’s decision to launch the Offer pursuant to Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG, the Bidder was registered under the name “Novartis data42 AG”. Since the amendment to the Bidder’s articles of association on 5 February 2024, it is now registered under the name “Novartis BidCo AG”, as published in the Swiss Official Gazette of Commerce (Schweizerisches Handelsamtsblatt) on 9 February 2024. The current business address of the Bidder is Lichtstrasse 35, 4056 Basel, Switzerland (tel.: +41 61 324 1111).
The share capital of the Bidder amounts to CHF 100,000.00 which is divided into 100,000 registered shares. The corporate purpose of the Bidder includes, inter alia, to acquire, hold and manage participations of any kind.
According to Sec. 6.3 of the Offer Document, the Bidder is a wholly-owned direct subsidiary of Novartis Pharma AG (“Novartis Pharma”), and a wholly-owned indirect subsidiary of Novartis AG.
According to Sec. 6.2 of the Offer Document, the members of the Bidder’s board of directors (Verwaltungsrat) are
| • | Daniel Andreas Weiss, chairman of the board of directors (Präsident des Verwaltungsrates); |
| • | Bertrand Richard René Bugnon, member of the board of directors (Mitglied des Verwaltungsrates); and |
| • | Christian Rehm, Ph.D., member of the board of directors (Mitglied des Verwaltungsrates). |
For further details of the positions and current and material mandates during the past five (5) years of current members of the Bidder’s board of directors, see Sec. 6.2 of the Offer Document.
Novartis Pharma
According to the information provided by the Bidder in Sec. 6.3 of the Offer Document, Novartis Pharma, the sole shareholder of the Bidder, is a stock corporation (Aktiengesellschaft) incorporated under the laws of Switzerland, with registered office at Lichtstrasse 35, 4056 Basel, Switzerland, registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-106.052.527.
According to Sec. 6.3 of the Offer Document, Novartis Pharma is a wholly-owned direct subsidiary of Novartis AG.
According to Sec. 6.3 of the Offer Document, the current members of Novartis Pharma’s board of directors (Verwaltungsrat) are
| • | Joerg Reinhardt, Ph.D., chairman of the board of directors (Präsident des Verwaltungsrates); |
| • | Karen L. Hale, member of the board of directors (Mitglied des Verwaltungsrates); and |
| • | Harry Kirsch, member of the board of directors (Mitglied des Verwaltungsrates). |
For further details of the positions and current and material mandates during the past five (5) years of current members of the Bidder’s board of directors, see Sec. 6.3.1 of the Offer Document.
Novartis AG
According to the information provided by the Bidder in Sec. 6.3.2 of the Offer Document, Novartis AG, the sole shareholder of Novartis Pharma and parent company of Novartis, is a stock corporation
25
(Aktiengesellschaft) incorporated under the laws of Switzerland, with registered office at Lichtstrasse 35, 4056 Basel, Switzerland, registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-103.867.266. Novartis AG is a publicly listed company whose stock trades on the SIX Swiss Exchange under the ticker symbol “NOVN” and on the New York Stock Exchange in the form of American Depositary Shares under the ticker symbol NVS. Novartis AG itself is not controlled by any of its shareholders.
The current members of Novartis AG’s board of directors (Verwaltungsrat) are:
| • | Joerg Reinhardt, Ph.D., chairman of the board of directors (Präsident des Verwaltungsrates); |
| • | Simon Moroney, D. Phil, vice-chairman of the board of directors (Vizepräsident des Verwaltungsrates); |
| • | Nancy C. Andrews, M.D., Ph.D., member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Ton Buechner, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Patrice Bula, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Elizabeth (Liz) Doherty, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Bridgette Heller, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Daniel Hochstrasser, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Franz van Houten, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Ana de Pro Gonzalo, member of the board of directors (Mitglied des Verwaltungsrates); |
| • | Charles L. Sawyers, M.D., member of the board of directors (Mitglied des Verwaltungsrates); |
| • | William T. Winters, member of the board of directors (Mitglied des Verwaltungsrates); and |
| • | John D. Young, member of the board of directors (Mitglied des Verwaltungsrates). |
For further details of the positions and current and material mandates during the past five (5) years of current members of the Bidder’s board of directors, see Sec. 6.3.2 of the Offer Document.
Corporate Structure
The following chart illustrates the current shareholder structure of the Bidder as described above:
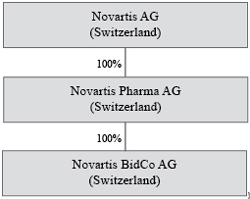
According to the information provided by the Bidder in Sec. 6.4 of the Offer Document, Novartis is specialized in the research, development, manufacturing, distribution, and commercialization and sale of innovative medicines, with a focus on four core therapeutic areas: cardiovascular, renal and metabolic; immunology; neuroscience; oncology; and established brands.
26
No criminal or other proceedings
According to Sec. 6.3.2 of the Offer Document, during the last five (5) years, none of Novartis AG, Novartis Pharma, the Bidder or, after due inquiry of Novartis AG, Novartis Pharma and the Bidder, any of the persons listed in Sec. 6.2 or 6.3 of the Offer Document (i) has been convicted in a criminal proceeding (excluding traffic violations or similar misdemeanors), and/or (ii) was a party to any judicial or administrative proceeding (except for matters that were dismissed without sanction or settlement) that resulted in a judgment, decree, or final order enjoining such person from future violations of, or prohibiting activities subject to, U.S. federal or state securities laws, or a finding of any violation of U.S. federal or state securities laws.
Selected Financial and Other Information about Novartis
The business segments of Novartis are reported in the consolidated financial statements of Novartis AG for the current and prior years reported as:
| (ii) | Continuing operations: Continuing operations include the research, development, manufacturing, distribution, and commercialization and sale of innovative medicines, with a focus on four core therapeutic areas: cardiovascular, renal and metabolic; immunology; neuroscience; oncology, and established brands; and |
| (iii) | Discontinued operations: Discontinued operations include the Sandoz generic pharmaceuticals and biosimilars business (the “Sandoz Division”) and certain corporate activities attributable to Sandoz Division, as well as certain expenses related to the spin-off. |
According to Sec. 6.4 of the Offer Document, during the financial year that ended 31 December 2023, Novartis achieved net sales from continuing operations of USDbn 45.44 (corresponding to approximately EURbn 41.97), and net income from continuing operations amounted to USDbn 8.57 (corresponding to approximately EURbn 7.92) according to the consolidated income statements of Novartis AG as of and for the financial year ended 31 December 2023 and prepared in accordance with IFRS Accounting Standards as issued by IASB.
According to Sec. 6.4 of the Offer Document, as of 31 December 2023, the number of Novartis’ full-time equivalent employees amounted to 76,057.
| 4.2 | MorphoSys Shares Held by the Bidder and Persons Acting Jointly with it |
Persons Acting Jointly with the Bidder
According to the information provided by the Bidder in Sec. 6.5 of the Offer Document, persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 sentence 3 WpÜG are the companies listed in Annex 1 of the Offer Document. Beyond this, the Bidder does not have any subsidiaries; therefore, according to the Offer Document, there are no other persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 WpÜG.
MorphoSys Shares Currently Held by the Bidder and Persons Acting Jointly with the Bidder and by Their Subsidiaries; Attribution of Voting Rights
According to information provided by the Bidder in Sec. 6.6 of the Offer Document, at the time of the publication of the Offer Document, none of the Bidder, persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 WpÜG, or their subsidiaries hold MorphoSys Shares or voting rights attached to MorphoSys Shares and no voting rights attached to MorphoSys Shares are attributable to them pursuant to Sec. 30 WpÜG. None of the Bidder, persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 WpÜG or their subsidiaries hold, directly or indirectly, voting rights in relation to MorphoSys to be disclosed pursuant to Sec. 38 and/or Sec. 39 WpHG.
27
| 4.3 | Information about Securities Transactions |
Previous Securities Transactions
According to the information provided by the Bidder in Sec. 6.7 of the Offer Document, none of the Bidder, the persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 sentence 1 and 3 WpÜG, or their subsidiaries have acquired any MorphoSys Shares or entered into any agreement on the acquisition of MorphoSys Shares in the period starting six (6) months prior to the date of the announcement of the Bidder’s decision to launch the Offer pursuant to Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG until the date of the publication of the Offer Document (i.e., 11 April 2024).
During the past two (2) years prior to the publication of the Offer Document, Novartis AG and MorphoSys have engaged in ordinary course collaboration and global licensing of certain MorphoSys’ assets. The aggregate value of such transactions during the past two (2) years prior to the publication of the Offer Document was approximately USDm 27.7.
Except as set forth in the Offer Document, there have been no transactions or business relationships that would require reporting under the rules and regulations of the SEC applicable to the Offer between Novartis AG, the Bidder, or any of their affiliates within the meaning of U.S. law (“Affiliates”) or—after due inquiry and to the best knowledge and belief of Novartis AG and the Bidder any of the persons listed in Secs. 6.2 or 6.3 of the Offer Document, on the one hand, and MorphoSys or any of its executive officers, members of the Management Board or Supervisory Board and/or a MorphoSys Affiliate, on the other hand. Except as set forth in the Offer Document and Sec. 10.2 of this Statement, there have been no contacts, negotiations, or transactions between Novartis AG or any of its subsidiaries or, after due inquiry and to the best knowledge of Novartis AG and the Bidder, any of the persons listed in Secs. 6.2 or 6.3 of the Offer Document, on the one hand, and MorphoSys or MorphoSys Affiliates, on the other hand, concerning a merger, consolidation or acquisition, tender offer, or other acquisition of any class of MorphoSys’ securities, an election of MorphoSys’ directors, or a sale or other transfer of a material amount of assets of MorphoSys during the past two (2) years prior to the publication of the Offer Document.
Possible Future Securities Transactions
According to the information provided by the Bidder in Sec. 6.8 of the Offer Document, the Bidder and its Affiliates or brokers (acting as agents of the Bidder or its Affiliates) may acquire, or make arrangements to acquire, MorphoSys Shares other than in the course of the Offer, via the stock exchange at market prices or outside the stock exchange on negotiated terms during the period in which the Offer remains open for acceptance or afterwards, provided that (i) such acquisitions or arrangements to acquire (if made during the pendency of the Offer) are made outside of the United States, and (ii) such acquisitions or arrangements to acquire comply with the applicable statutory provisions, in particular German Takeover Rules, and, to the extent applicable, the U.S. Offer Rules. This also applies to other securities convertible into, exchangeable for or exercisable for shares of MorphoSys. The Offer Price must be adjusted to any higher purchase price or an additional payment claim in respect of Tendered MorphoSys Shares (as defined below) and Subsequently Tendered MorphoSys Shares (as defined below) in the amount of the difference between the Offer Price and the higher purchase price paid outside the Offer.
To the extent such acquisitions should occur, information about them, including the number and price of the acquired MorphoSys Shares, will be published according to the applicable statutory provisions, especially Sec. 23 para. 2 WpÜG in conjunction with Sec. 14 para. 3 sentence 1 WpÜG, in the German Federal Gazette (Bundesanzeiger) and on the internet at the website of the Bidder at
https://www.novartis.com/investors/morphosys-acquisition.
In addition, the financial advisors of the Bidder may also act in the ordinary course of trading in securities of MorphoSys, which may include purchases or agreements to purchase such securities.
28
| 5 | INFORMATION ABOUT THE OFFER |
| 5.1 | Implementation of the Offer |
According to the Bidder, the Offer is a voluntary public takeover offer (freiwilliges öffentliches Übernahmeangebot) (cash offer) for the acquisition of the MorphoSys Shares, launched and implemented in accordance with the German Takeover Rules and the U.S. Offer Rules. In order to reconcile certain areas where German Takeover Rules and U.S. Offer Rules conflict, the Bidder has obtained, prior to the publication of the Offer Document, the SEC Relief, as further described in Sec. 21 of the Offer Document.
Moreover, as per Sec. 1.5 of the Offer Document, neither the Bidder nor any persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 WpÜG nor any of its directors, officers or employees assume any responsibility for compliance with any legal requirements other than those applicable in Germany and the United States.
According to the Offer Document, the Bidder does not intend to make the Offer as a public offer pursuant to the applicable law of any jurisdictions other than Germany and the United States. According to the Offer Document, MorphoSys Securityholders may not rely on the legal requirements for the protection of investors of any jurisdiction other than those applicable in Germany and the United States.
| 5.2 | Publication of the Decision to Launch the Offer |
The Bidder published its decision to launch the Offer in accordance with Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG on 5 February 2024. The German version of such publication and an English language version are available on the internet at the website of the Bidder at
https://www.novartis.com/investors/morphosys-acquisition.
In addition, the publication in which the Bidder announced its decision to make the Offer, together with other materials related to the Offer, were filed by the Bidder and Novartis AG with the SEC as preliminary tender offer communications under Schedule TO. Such preliminary tender offer communication and other documents filed with the SEC by the Bidder and Novartis AG as well as MorphoSys in connection with the Offer are available on the internet at the website of the SEC at
https://www.sec.gov/edgar/browse/?CIK=1114448 (for Novartis AG)
and
https:// www.sec.gov/edgar/browse/?CIK=1340243 (for MorphoSys).
| 5.3 | BaFin Review and Publication of the Offer Document |
The BaFin reviewed the German version of the Offer Document in accordance with the German Takeover Rules and permitted the publication of the Offer Document on 11 April 2024. The non-binding English translation of the Offer Document has neither been reviewed nor approved by the BaFin.
The Bidder states in the Offer Document that no announcements, registrations, admissions or approvals of the Offer Document or the Offer by securities exchange authorities or similar authorities outside Germany and the United States have been applied for, have been initiated or are envisaged by the Bidder or Novartis AG. The Offer Document sets forth that neither the SEC nor any state securities commission in the United States has approved or disapproved the Offer or passed upon the fairness or merits of, or upon the accuracy or adequacy of the information contained in the Offer Document.
On 11 April 2024, the Bidder published the German version of the Offer Document on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition
and made copies of the Offer Document available free of charge at Deutsche Bank Aktiengesellschaft, TAS, Post-IPO Services, Taunusanlage 12, 60325 Frankfurt am Main, Germany in accordance with
29
Sec. 14 para. 3 WpÜG. MorphoSys Shareholders may request to obtain a copy of the Offer Document via email to dct.tender-offers@db.com or by fax to +49 69 910 38794, indicating a valid mailing address. In addition, the Bidder has made available the Offer Document including an English language version of the Offer Document, which has not been reviewed by the BaFin, on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition.
Notice of (i) the availability of copies of the Offer Document provided free of charge in Germany and (ii) the Internet address at which the German version of the Offer Document is available, was published by the Bidder on 11 April 2024 in the German Federal Gazette (Bundesanzeiger). In the United States, the corresponding announcement was made in The New York Times (U.S. Edition). In addition, the Bidder intends to mail the English version of the Offer Document to all MorphoSys ADS Holders.
The Schedule TO and any amendments thereto (see Sec. 1.5 of the Offer Document), exhibits to the Schedule TO, and other information that the Bidder and Novartis AG have filed electronically with the SEC may be obtained free of charge on the internet at the website of the SEC at
https://www.sec.gov/edgar/browse/?CIK=1114448.
| 5.4 | Publications by the Bidder in Connection with the Offer |
In Sec. 22 of the Offer Document, the Bidder has stated that all publications and announcements required according to the WpÜG or the applicable capital market laws provisions of the United States in connection with the Offer will be published on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition
and, to the extent necessary pursuant to the WpÜG, in the German Federal Gazette (Bundesanzeiger).
The Bidder has announced that it will publish the notifications pursuant to Sec. 23 para. 1 WpÜG as follows:
| (i) | On a weekly basis after publication of the Offer Document (Sec. 23 para. 1 sentence 1 no. 1 WpÜG); |
| (ii) | On a daily basis during the final week prior to the end of the Acceptance Period (Sec. 23 para. 1 sentence 1 no. 1 WpÜG); |
| (iii) | Without undue delay (unverzüglich) after expiry of the Acceptance Period (Sec. 23 para. 1 sentence 1 no. 2 WpÜG); |
| (iv) | Without undue delay (unverzüglich) after expiry of the Additional Acceptance Period (Sec. 23 para. 1 sentence 1 no. 3 WpÜG); and |
| (v) | Without undue delay after having reached the shareholding required to exclude the other shareholders under Sec. 39a para. 1 and para. 2 WpÜG. |
Furthermore, the Bidder will file publications and announcements in connection with the Offer English with the SEC and will make them available in English on the internet at the website of the SEC at
https:// www.sec.gov/edgar/browse/?CIK=1114448 (for Novartis AG)
and
https:// www.sec.gov/edgar/browse/?CIK=1340243 (for MorphoSys).
30
In addition, in Sec. 12.4 of the Offer Document the Bidder has stated that it will promptly announce on the internet at its website at
https://www.novartis.com/investors/morphosys-acquisition
(in German and in an English translation) and in the German Federal Gazette (Bundesanzeiger) if
| (i) | An Offer Condition has been effectively waived in advance; |
| (ii) | An Offer Condition has been fulfilled; |
| (iii) | All Offer Conditions have either been fulfilled or have been effectively waived in advance; or |
| (iv) | The Offer is not consummated because an Offer Condition has finally not been fulfilled or lapsed. Likewise, the Bidder has stated in the Offer Document that it will promptly announce at the end of the Acceptance Period, as part of the publication according to Sec. 23 para. 1 no. 2 WpÜG, which of the Offer Conditions have been fulfilled by such time. According to the Offer Document, the Bidder will also make public announcements in accordance with the applicable rules, regulations and interpretations of the SEC. |
For further details and other publications of the Bidder, especially pertaining to U.S. law, see Sec. 22 of the Offer Document.
| 5.5 | Material Terms and Conditions of the Offer |
| 5.5.1 | Offer Price |
Subject to the terms and conditions set forth in the Offer Document, the Bidder offers to acquire all MorphoSys Shares (including all MorphoSys Shares represented by MorphoSys ADSs) not held directly by the Bidder (ISIN DE0006632003) at the Offer Price of
EUR 68.00
per MorphoSys Share,
without interest.
The Offer Price per MorphoSys Share applies to all MorphoSys Shares, including all ancillary rights existing at the time of Closing, in particular the right to dividends.
MorphoSys ADS Holders may accept the Offer by (i) surrendering their MorphoSys ADSs to receive MorphoSys Shares, or (ii) by instructing the ADS Tender Agent to tender the MorphoSys Shares underlying their MorphoSys ADSs in the Offer. More details can be found in Sec. 13.2 and Sec. 13.3 of the Offer Document.
| 5.5.2 | Acceptance Period and Additional Acceptance Period |
Acceptance Period
The period for accepting the Offer for the MorphoSys Shares including MorphoSys Shares represented by MorphoSys ADSs began upon publication of the Offer Document on 11 April 2024 and will end on
13 May 2024,
24:00 hours Frankfurt am Main Local Time and
18:00 hours New York Local Time, respectively
(such period, including all extensions resulting from provisions of the WpÜG (but excluding the Additional Acceptance Period described in Sec. 5.3 of the Offer Document) as set out below the “Acceptance Period”).
31
Extension of the Acceptance Period
The period for the acceptance of the Offer will be extended automatically in the following cases:
| (i) | The Bidder may amend the Offer until one (1) business day (Arbeitstag) in Germany before expiry of the Acceptance Period in accordance with Sec. 21 WpÜG. In the event of an amendment of the Offer pursuant to Sec. 21 WpÜG, the Acceptance Period will be automatically extended by two (2) weeks pursuant to Sec. 21 para. 5 sentence 1 WpÜG, if publication of the amended Offer takes place within the last two (2) weeks before expiry of the Acceptance Period. In that event, pursuant to Sec. 5.1 of the Offer Document, the Acceptance Period will be extended until 27 May 2024, 24:00 hours Frankfurt am Main Local Time / 18:00 hours New York Local Time. Pursuant to Sec. 21 para. 5 sentence 2 WpÜG, this applies even if the amended Offer violates statutory provisions. |
| (ii) | If (A) a competing public offer is launched by a third party in accordance with Sec. 22 para. 1 WpÜG (a “Competing Offer”) during the Acceptance Period for the Offer and (B) the Acceptance Period for the Offer expires prior to the end of the acceptance period for the Competing Offer, the Acceptance Period for the Offer will automatically be extended to correspond to the expiry of the acceptance period for the Competing Offer in accordance with Sec. 22 para. 2 WpÜG. Pursuant to Sec. 21 para. 5 sentence 2 WpÜG, this applies even if the Competing Offer is amended or prohibited or violates statutory provisions. |
| (iii) | If a general meeting (Hauptversammlung) of MorphoSys is convened in connection with the Offer following publication of the Offer Document, then the Acceptance Period will be extended to ten (10) weeks beginning with publication of the Offer Document in accordance with Sec. 16 para. 3 WpÜG (i.e., until 20 June 2024, 24:00 hours Frankfurt am Main Local Time / 18:00 hours New York Local Time). |
According to Secs. 5.2 and 21 of the Offer Document, on 9 April 2024, the Bidder obtained the SEC Relief in order to permit the extension of the period for acceptance in compliance with German Takeover Rules by two (2) weeks and not, as stipulated by the U.S. Offer Rules, by only ten (10) U.S. Working Days in case of an amendment to this Offer.
MorphoSys Securityholders that have accepted the Offer have certain withdrawal rights, as further described in Sec. 17 of the Offer Document.
Additional Acceptance Period
MorphoSys Securityholders that have not accepted the Offer within the Acceptance Period can, subject to Sec. 16 para. 2 sentence 1 WpÜG, still accept it within two (2) weeks after publication of the results of the Offer by the Bidder according to Sec. 23 para. 1 sentence 1 no. 2 WpÜG (the “Additional Acceptance Period”), provided that none of the Offer Conditions described in Sec. 5.5.3 of this Statement, and more fully in Sec. 12.1 of the Offer Document, has ultimately lapsed (endgültig ausgefallen) as at the end of the Acceptance Period and such Offer Condition has not been effectively waived by the Bidder in advance. After the end of the Additional Acceptance Period, the Offer cannot be accepted anymore, provided that any put rights pursuant to Sec. 39c WpÜG shall remain unaffected if the Bidder would reach a shareholding of at least ninety-five percent (95%) in MorphoSys after the completion of the Takeover as further described in Sec. 16 para. (i) of the Offer Document.
Subject to an extension of the Acceptance Period in accordance with Sec. 5.2 of the Offer Document, the Additional Acceptance Period will presumably begin on 17 May 2024 and end on 30 May 2024, 24:00 hours Frankfurt am Main Local Time / 18:00 hours New York Local Time.
32
| 5.5.3 | Offer Conditions |
The Offer and the contracts which come into existence with the MorphoSys Securityholders as a result of accepting the Offer are subject to the conditions subsequent, as set forth in more detail in in Sec. 12 of the Offer Document (together the “Offer Conditions”).
Minimum Acceptance Condition
As of the expiration of the Acceptance Period, the total sum of the MorphoSys Shares (including MorphoSys Shares that are represented by MorphoSys ADS),
| (i) | For which the acceptance of the Offer has been effectively declared in accordance with Sec. 13.1.2 of the Offer Document and for which no withdrawal of the agreement entered into as a result of the acceptance of the Offer has been effectively declared; |
| (ii) | For which the acceptance of the Offer has been declared during the Acceptance Period but only becomes effective after the end of the Acceptance Period by re-booking the MorphoSys Shares to ISIN DE000A4BGGU0 in accordance with Secs. 13.3.2 and 13.3.3 of the Offer Document; |
| (iii) | Held directly by the Bidder or a person acting jointly with the Bidder pursuant to Sec. 2 para. 5 WpÜG; |
| (iv) | Attributable to the Bidder or a person acting jointly with the Bidder pursuant to Sec. 2 para. 5 WpÜG pursuant to Sec. 30 WpÜG; and/or |
| (v) | For which the Bidder or persons acting jointly with the Bidder within the meaning of Sec. 2 para. 5 WpÜG have concluded an unconditional or conditional agreement outside the Offer which entitles them to demand the transfer of title to the MorphoSys Shares (including MorphoSys Shares that are represented by MorphoSys ADSs) within the meaning of Sec. 31 para. 6 WpÜG |
equals at 65% of the number of MorphoSys Shares, other than treasury shares, issued as of the expiration of the Acceptance Period (the “Minimum Acceptance Condition”). Based on the number of MorphoSys Shares currently outstanding and the number of MorphoSys treasury shares currently held, this corresponds to 24,480,780 MorphoSys Shares. MorphoSys Shares which are subject to several of the preceding paras. (i) through (v) above will be taken into account only once.
No Insolvency Condition
During the Acceptance Period, MorphoSys did not publish an ad hoc announcement pursuant to Article 17 of the Market Abuse Regulation (Regulation (EU) No. 596/2014 of the European Parliament and of the Council) stating that (i) insolvency proceedings against all or substantially all of the assets of MorphoSys have been filed by MorphoSys or opened by the competent court in Munich, Germany, or (ii) a reason has arisen that would require the filing of an application for the institution of an insolvency proceeding under applicable law (the “No Insolvency Condition”).
No Illegality Condition
During the Acceptance Period, no Law, administrative act, injunction, temporary restraining order, preliminary or permanent injunction, or other order issued by a Specified Governmental Authority prohibits or makes illegal the conduct or consummation of the Offer in accordance with its terms and conditions or the acquisition of ownership of MorphoSys Shares by the Bidder (the “No Illegality Condition”).
“Specified Governmental Authority” means any competent governmental authority in the United States, Germany or any other jurisdiction in which Novartis AG and/or MorphoSys have subsidiaries at the time of the publication of the Offer Document.
33
“Law” means any international, national, federal, state or local law, statute, ordinance, regulation, ruling, order, injunction, decree, award, governmental request, concession, variance, exemption, approval, licence and permit.
No Material Compliance Violation Condition
During the Acceptance Period, no criminal offense or material administrative offense (Ordnungswidrigkeit) relating to applicable corruption, embezzlement, anti-bribery, money-laundering or cartel Laws or Healthcare Laws relating to data integrity by any MorphoSys Group company or a member of a governing body of any MorphoSys Group company, while any such person was operating in their official capacity at or on behalf of such MorphoSys Group company, or officer of any MorphoSys Group company, will have occurred, provided that any such offense constitutes or would constitute inside information for MorphoSys pursuant to Article 7 EU Market Abuse Regulation or has constituted inside information prior to its publication (“Material Compliance Violation” and the Offer Condition described in this paragraph, the “No Material Compliance Violation Condition”).
A Material Compliance Violation will only be deemed to have occurred if, on or before the day before the publication of the results of the Offer pursuant to Sec. 23 para. 1 sentence 1 no. 2 WpÜG, BDO AG Wirtschaftsprüfungsgesellschaft, Hanauer Landstrasse 115, 60314 Frankfurt am Main, Germany, as independent expert, using the due and careful consideration of a diligent professional, has delivered an opinion that a Material Compliance Violation has occurred.
| 5.5.4 | Nonfulfillment and Waiver of Offer Conditions |
Each of the Minimum Acceptance Condition, the No Insolvency Condition, the No Illegality Condition and the No Material Compliance Violation Condition constitute independent and separable conditions.
The Closing will occur if all Offer Conditions have been satisfied or effectively waived in advance by the end of the Acceptance Period.
The Bidder may waive all or individual Offer Conditions in advance – to the extent permissible under statutory law – pursuant to Sec. 21 para. 1 sentence 1 no. 4 WpÜG until one (1) business day (Arbeitstag) prior to the expiry of the Acceptance Period as long as such Offer Conditions have not ultimately lapsed. The previous effective waiver is equivalent to the fulfilment of the relevant Offer Condition.
An effective waiver of an Offer Condition or a reduction of the minimum acceptance threshold of MorphoSys Shares, the acquisition of which the Bidder has made conditional upon the validity of the Offer (as described in Section 8.2.14 of the Offer Document), constitutes an amendment to the Offer. The Bidder is obliged to publish an amendment to the Offer, and therefore also an effective waiver of any Offer Condition, without undue delay in accordance with Sec. 21 para. 2 sentence 1 in connection with Sec. 14 para. 3 sentence 1 WpÜG. In the event an amendment to the Offer is published within the last two (2) weeks prior to the end of the Acceptance Period, the Acceptance Period will be automatically extended by two (2) weeks pursuant to Sec. 21 para. 5 WpÜG. In the event of an effective waiver of any Offer Condition, MorphoSys Securityholders that have accepted the Offer prior to the publication of the amendment of the Offer may withdraw within the Acceptance Period from the agreements that have come into existence as a result of accepting the Offer pursuant to Sec. 21 para. 4 WpÜG.
In the Business Combination Agreement, Novartis AG and MorphoSys agreed that the Bidder may waive the Minimum Acceptance Condition below 50% of the MorphoSys Shares issued as of the expiration of the Acceptance Period only with the prior consent of MorphoSys.
If one or more of the Offer Conditions has not been timely satisfied in accordance with the foregoing and the Bidder did not effectively waive such Offer Condition in advance, the Offer will lapse and the contracts which have been entered as a result of the acceptance of the Offer will automatically cease to exist and not be consummated. For further details of the Offer Conditions please see Sec. 12 of the Offer Document.
34
For further details on the possible previous waiver of Offer Conditions and the legal consequences in the event of a lapse of the Offer, reference is made to Sec. 12.3 of the Offer Document.
| 5.5.5 | Governing Law; Place of Jurisdiction |
According to Sec. 24 of the Offer Document, the Offer and the contracts concluded as a result of the acceptance of the Offer will be governed by German law. The exclusive place of jurisdiction for all legal disputes arising out of, or in connection with, the Offer (and any contract concluded as a result of acceptance of the Offer) will be, to the extent permitted by law, Frankfurt am Main, Germany.
| 5.6 | Acceptance and Settlement of the Offer for MorphoSys Securities |
| 5.6.1 | Acceptance of the Offer Outside Germany, the Member States of the European Union, the European Economic Area and the United States |
The Bidder points out in Sec. 1.7 of the Offer Document that the acceptance of the Offer outside of Germany, the member states of the European Union and the European Economic Area and the United States may be subject to legal restrictions.
MorphoSys Securityholders who come into possession of the Offer Document outside of Germany, the member states of the European Union and the European Economic Area or the United States and who wish to accept the Offer outside of Germany, the member states of the European Union and the European Economic Area or the United States, and/or who are subject to statutory provisions other than those of Germany, the member states of the European Union and the European Economic Area or the United States are advised to inform themselves of and to comply with the relevant applicable statutory provisions. According to Sec. 1.7 of the Offer Document, neither the Bidder or any persons acting jointly with the Bidder nor the ADS Tender Agent assume responsibility for acceptance of the Offer outside of Germany, the member states of the European Union and the European Economic Area and the United States being permissible under the relevant applicable statutory provisions.
| 5.6.2 | Acceptance and Settlement of the Offer for MorphoSys Shares |
Acceptance of the Offer for MorphoSys Shares
MorphoSys Shareholders may accept the Offer as is set out in detail in Sec. 13.1 of the Offer Document.
According to Sec. 13.1.2 of the Offer Document, MorphoSys Shareholders who wish to accept the Offer should contact their custodian bank or other custodian investment service provider (in each case, the “Custodian Bank”) with registered office or a branch in Germany with any questions they may have regarding acceptance of the Offer and the technical aspects of settlement of the Offer. Those banks and service providers have been separately informed about the modalities for acceptance and settlement of the Offer and are required to inform customers who hold MorphoSys Shares in their securities deposit accounts about the Offer and the steps required to accept it.
During the Acceptance Period (please refer to Sec. 13.1.6 of the Offer Document regarding acceptance of the Offer during the Additional Acceptance Period), MorphoSys Shareholders may accept the Offer only by:
| (i) | A declaration of acceptance of the Offer in writing or in text form (the “Declaration of Acceptance”) vis-à-vis their own Custodian Bank, and |
| (ii) | Instructing their Custodian Bank to immediately effect the booking of the MorphoSys Shares which are held in their securities deposit account and for which they wish to accept the Offer to ISIN DE000A4BGGU0 at Clearstream Banking AG, Frankfurt am Main, Germany (“Clearstream”). |
The Declaration of Acceptance will not become effective until the MorphoSys Shares for which the Offer has been accepted during the Acceptance Period have been booked to ISIN DE000A4BGGU0 at
35
Clearstream by no later than 18:00 hours Frankfurt am Main Local Time / 12:00 hours New York Local Time on the second (2nd) FSE Trading Day after expiry of the Acceptance Period (any such MorphoSys Shares, the “Tendered MorphoSys Shares”). Such bookings are to be arranged by the relevant Custodian Bank without undue delay after receipt of the Declaration of Acceptance.
The terms and conditions of the Offer Document apply mutatis mutandis to the acceptance of the Offer during the Additional Acceptance Period (any MorphoSys Shares tendered during the Additional Acceptance Period, the “Subsequently Tendered MorphoSys Shares”) in accordance with the following. Booking at Clearstream of MorphoSys Shares tendered during the Additional Acceptance Period will be regarded timely if it has been effected by no later than 18:00 hours Frankfurt am Main Local Time / 12:00 hours New York Local Time on the second (2nd) FSE Trading Day following expiry of the Additional Acceptance Period to ISIN DE000A4BGGV8.
For further details on the acceptance of the Offer for MorphoSys Shares and the legal consequences of accepting the offer, please refer to Secs. 13.1.2 to 13.1.4 as well as Sec. 13.1.6 of the Offer Document.
Settlement of the Offer for MorphoSys Shares
With respect to the Tendered MorphoSys Shares, the settlement of the Offer will occur as is set out in detail in Sec. 13.1 of the Offer Document. According to Sec. 13.1.5 of the Offer Document, payment of the Offer Price will be effected to the relevant Custodian Bank concurrently and contemporaneously against transfer of the Tendered MorphoSys Shares to the account of the Settlement Agent at Clearstream. The Settlement Agent will cause the Offer Price for the Tendered MorphoSys Shares to be transferred through Clearstream to the relevant Custodian Bank without undue delay, but no later than on the fourth (4th) Banking Day following publication of the tender results after the end of the Acceptance Period pursuant to Sec. 23 para. 1 no. 2 WpÜG (i.e., 23 May 2024 subject to any extension of the Acceptance Period).
According to Sec. 13.1.7 of the Offer Document, payment of the Offer Price will be effected to the relevant Custodian Bank concurrently and contemporaneously against transfer of the Subsequently Tendered MorphoSys Shares to the account of the Settlement Agent at Clearstream. The Settlement Agent will cause the Offer Price for the Subsequently Tendered MorphoSys Shares to be transferred through Clearstream to the relevant Custodian Bank without undue delay, but no later than on the fourth (4th) Banking Day following publication of the tender results after the end of the Additional Acceptance Period pursuant to Sec. 23 para. 1 no. 3 WpÜG (i.e., on 10 June 2024 subject to any extension of the Acceptance Period).
For further details on the settlement of the Offer for MorphoSys Shares, please refer to Secs. 13.1.5 and 13.1.7 of the Offer Document.
| 5.6.3 | Acceptance and Settlement of the Offer for MorphoSys ADSs |
Surrendering MorphoSys ADSs to receive MorphoSys Shares to tender in the Offer
According to the information provided by the Bidder in Sec. 13.2 of the Offer Document, MorphoSys ADS Holders who wish to participate in the acceptance and settlement of the Offer as pertains to MorphoSys Shares (as described in Sec. 13.1 of the Offer Document), may tender the underlying MorphoSys Shares in respect of the MorphoSys ADSs in the Offer following their withdrawal from the MorphoSys ADS program. Each MorphoSys ADS represents one-quarter of one MorphoSys Share deposited with The Bank of New York Mellon (the “ADS Depositary”).
Instead of instructing the ADS Tender Agent to tender the MorphoSys Shares underlying its MorphoSys ADSs in the Offer as described in Sec. 13.3 of the Offer Document, a MorphoSys ADS Holder may tender MorphoSys Shares in the Offer by surrendering its MorphoSys ADSs to the ADS Depositary, paying any governmental charges or cable fees or other charges payable in connection with such surrender
36
and withdrawal (except for any transfer, documentary, sales, use, stamp, registration, value-added and other similar taxes and fees, which shall be paid by the Bidder) and otherwise complying with the terms and conditions of the ADS Deposit Agreement (as defined below). The ADS Depositary’s cancellation fee of USD 0.05 per MorphoSys ADS will be borne by the Bidder.
For further details on surrendering MorphoSys ADSs to receive MorphoSys Shares to tender in the Offer, please refer to Sec. 13.2 of the Offer Document.
Acceptance of the Offer for MorphoSys ADSs
According to Sec. 13.3.1 of the Offer Document, the acceptance of the Offer by MorphoSys ADS Holders for MorphoSys Shares represented by MorphoSys ADSs can only be made through The Bank of New York Mellon as tender agent (the “ADS Tender Agent”) The acceptance notice can be sent to the ADS Tender Agent by mail or courier to the below addresses:
| Registered, Certified or Express Mail | By Overnight Courier | |
| The Bank of New York Mellon Attn: Voluntary Corporate Actions, COY: MPSB P.O. Box 43011 Providence, RI 02940-3011 United States | The Bank of New York Mellon Attn: Voluntary Corporate Actions, COY MPSB 150 Royall Street, Suite V Canton, MA 02021 United States | |
E-Mail (for notices of guaranteed delivery only):
canoticeofguarantee@computershare.com
The acceptance of the Offer for MorphoSys Shares represented by MorphoSys ADSs will be effectuated in a multistage process. MorphoSys ADS Holders may accept the Offer irrespective of whether the total number of ADS tendered by them is an integral multiple of four (i.e., the Offer may be readily accepted for one, two or three ADSs). First, the MorphoSys ADS Holder will tender the MorphoSys ADSs to the ADS Tender Agent (see Sec. 13.3.2 of the Offer Document). Second, the ADS Tender Agent, on behalf of the MorphoSys ADS Holder, will cause the book-entry transfer of such MorphoSys Shares to ISIN DE000A4BGGU0 (see Sec. 13.3.3 of the Offer Document).
According to Sec. 13.3.2 of the Offer Document, the procedure that can be used by MorphoSys ADS Holders, which is described in greater detail in Sec. 13.3.2 of the Offer Document, depends on whether the MorphoSys ADS Holder (i) is a registered holder of MorphoSys ADSs in certificated form evidenced by an MorphoSys ADR, (ii) is a registered holder of MorphoSys ADSs in uncertificated form (i.e., without an MorphoSys ADR) or (iii) holds MorphoSys ADSs in a securities account with a broker or other securities intermediary that is a direct or indirect participant in The Depository Trust Company (“DTC”).
According to Sec. 13.3.5 of the Offer Document, the terms and conditions of the Offer Document shall apply mutatis mutandis (with only slight modifications) to the acceptance of the Offer by MorphoSys ADS Holders within the Additional Acceptance Period.
For further details on the acceptance of the offer for MorphoSys ADSs, please refer to Sec. 13.3 of the Offer Document.
Settlement of the Offer for MorphoSys ADSs
According to Sec. 13.3.6 of the Offer Document, the Bidder will, subject to the terms and conditions of the Offer,
| (i) | With respect to the MorphoSys Shares represented by MorphoSys ADSs properly tendered for sale during the Acceptance Period and for which a withdrawal is no longer possible, pay the Offer Price |
37
| to the ADS Tender Agent’s cash account in Germany via Clearstream without undue delay, however no later than on the fourth (4th) Banking Day, following the publication of the tender results pursuant to Sec. 23 para. 1 no. 2 WpÜG after the expiration of the Acceptance Period; and |
| (ii) | With respect to the MorphoSys Shares represented by MorphoSys ADSs properly tendered for sale during the Additional Acceptance Period and for which a withdrawal is no longer possible, pay the Offer Price to the ADS Tender Agent’s cash account in Germany via Clearstream without undue delay, however no later than on the fourth (4th) Banking Days following the publication of the tender results pursuant to Sec. 23 para. 1 no. 3 WpÜG after the expiration of the Additional Acceptance Period. |
Upon crediting of the Offer Price to the ADS Tender Agent’s cash account in Germany via Clearstream, the Bidder will have fulfilled its obligation to pay the Offer Price. It is the ADS Tender Agent’s responsibility to transfer the Offer Price, after deduction of all applicable costs or withholding taxes, to the MorphoSys ADS Holders without undue delay. The ADS Depositary’s cancellation fee of USD 0.05 per MorphoSys ADS will be borne by the Bidder. The Offer Price will then be paid to the MorphoSys ADS Holders by the ADS Tender Agent in USD. The Offer Price will be converted by the ADS Tender Agent by sale of the EUR amounts received or in any other manner it determines using an exchange rate from EUR to USD determined or assigned by it on the day it effects the conversion.
| 5.7 | Stock-Exchange Trading in Tendered MorphoSys Shares |
Stock-Exchange Trading in Tendered MorphoSys Shares
According to Sec. 13.1.9 of the Offer Document, the Bidder will make the Tendered MorphoSys Shares tradable on the regulated market (Regulierter Markt) of the FSE with additional post-admission obligations (Prime Standard) under ISIN DE000A4BGGU0. Trading is expected to start on the third (3rd) FSE Trading Day after the commencement of the Acceptance Period. Trading with the Tendered MorphoSys Shares on the regulated market (Regulierter Markt) of the FSE is expected to continue until one (1) Banking Day following expiration of the Acceptance Period.
The shares that are tendered as Subsequently Tendered MorphoSys Shares with ISIN DE000A4BGGV8 will not be tradeable.
The Bidder points out that trading volumes and liquidity of the Tendered MorphoSys Shares depend on the specific acceptance rate and therefore may not exist at all or may be low and may be subject to heavy fluctuations. Therefore, it cannot be ruled out that, in the absence of demand, it will be impossible to sell the Tendered MorphoSys Shares on the FSE.
For further details of the stock-exchange trading in (Subsequently) Tendered MorphoSys Shares reference is made to Sec. 13.1.9 of the Offer Document.
Stock-Exchange Trading in Tendered MorphoSys ADSs
According to Sec. 13.3.8 of the Offer Document, there will be no stock market trading in MorphoSys ADSs tendered through the ADS Tender Agent (the “Tendered MorphoSys ADSs”). Tendered MorphoSys ADSs will be blocked on the books of the ADS Depositary or DTC until the MorphoSys Shares represented by MorphoSys ADSs are accepted for purchase, or the MorphoSys ADSs are withdrawn, or returned in the case of termination of the Offer.
| 5.8 | Financing of the Offer |
According to Sec. 14.1 of the Offer Document, the Bidder has indicated that the maximum costs for the implementation of the Offer amount to EUR 2,607,350,356.00 (including transaction costs of approx. EURm 20.5) and will be borne and paid by the Bidder.
38
According to Sec. 14.2 of the Offer Document, the Bidder has taken the necessary measures to ensure that the funds necessary for complete fulfillment of the costs of the Offer will be available to it in due time. The Bidder has secured the necessary financial means to meet its payment obligations at settlement under the Offer by having arranged for cash and committed funds, which will be made available to the Bidder (directly or indirectly) in the form of equity capital and/or shareholder loans or similar instruments for this purpose by Novartis AG. Further information on the financing measures are described in Sec. 14.2 of the Offer Document.
According to Sec. 14.3 of the Offer Document, Deutsche Bank Aktiengesellschaft, registered with the commercial register (Handelsregister) of the local court (Amtsgericht) of Frankfurt am Main, Germany, under the company number HRB 30000, an investment service provider that is independent of the Bidder, has issued the required financing confirmation pursuant to Sec. 13 para. 1 sentence 2 WpÜG dated 5 April 2024. It is attached to the Offer Document as annex 2.
| 5.9 | Relevance of the Offer Document |
The selected information in Sec. 5 of this Statement has been taken solely from the Offer Document of the Bidder and only summarizes certain important information set forth in the Offer Document. Thus, the description of the Offer in this Statement does not purport to be complete and this Statement should be read together with the Offer Document. For further information and details (in particular, details regarding the Offer Conditions, the Acceptance Period, the Additional Acceptance Period, the acceptance and implementation modalities and withdrawal rights), MorphoSys Securityholders should refer to the Offer Document. Only the terms and conditions of the Offer Document are authoritative for the content and execution of the Offer. It is the sole responsibility of each MorphoSys Securityholder to take – to the extent required – all necessary steps and actions under or in connection with the Offer Document.
| 6 | EVALUATION OF THE OFFER |
In evaluating the Offer, the Business Combination Agreement and the transactions contemplated by the Business Combination Agreement (collectively, the “Proposed Transactions”), the Management Board and the Supervisory Board consulted regularly with senior management of MorphoSys, as well as with representatives of MorphoSys’ financial advisor Centerview Partners LLC (“Centerview”) and MorphoSys’ legal advisor Skadden, Arps, Slate, Meagher & Flom LLP (“Skadden”).
In the course of (i) determining that the Offer, the Business Combination Agreement and the Proposed Transactions are in the best interest of MorphoSys, its business, and its shareholders, employees and other stakeholders, (ii) approving the Business Combination Agreement and the Proposed Transactions and (iii) determining to support the Offer and the Proposed Transactions and recommend acceptance of the Offer by the holders of MorphoSys Shares, the Management Board and the Supervisory Board considered numerous factors, including the factors listed below, which are listed in no particular order of importance, each of which, in the view of the Management Board and the Supervisory Board, supported such assessment, determination and recommendation, in addition to the factors mentioned in Sec. 6 of this Statement.
| 6.1 | Type and Amount of the Offer Consideration |
In accordance with and subject to the terms and conditions set forth in the Offer Document, the Bidder offers to acquire all (i) MorphoSys Shares, including all ancillary rights existing at the time of the settlement of the Offer, in particular the right to receive dividends, and (ii) all MorphoSys Shares represented by MorphoSys ADSs, at the Offer Price (consideration within the meaning of Sec. 27 para. 1 sentence 2 no. 1 WpÜG) in an amount of EUR 68.00 in cash for each MorphoSys Share.
As per Sec. 13.1.5 the Offer Document, the payment of the Offer Price in exchange for Tendered MorphoSys Shares validly tendered during the Acceptance Period and not properly withdrawn will be
39
made without undue delay, however no later than on the fourth (4th) Banking Day following the publication of the tender results after the end of the Acceptance Period pursuant to Sec. 23 para. 1 no. 2 WpÜG.
As per Sec. 13.1.7 of the Offer Document, the payment of the Offer Price in exchange for Subsequently Tendered MorphoSys Shares validly tendered during the Additional Acceptance Period will be made without undue delay, however no later than on the fourth (4th) Banking Day following the publication of the tender results after the end of the Additional Acceptance Period pursuant to Sec. 23 para. 1 no. 3 WpÜG.
| 6.1.1 | Adequacy of the Offer Price |
The Management Board and the Supervisory Board considered certain factors concerning the adequacy of the Offer Price, including, among other things, the factors listed below.
Premium to Historical Market Prices
The relationship of the Offer Price to the historical market prices of the MorphoSys Shares, including that the Offer Price represents a premium as set out below.
Based on the stock exchange price of the MorphoSys Shares prior to the publication of the decision of the Bidder to make the Offer on 5 February 2024, the Offer Price of EUR 68.00 includes the following premiums:
| (i) | The stock exchange price (XETRA closing price) on 2 February 2024, the last FSE Trading Day prior to the publication of the Bidder’s decision to launch the Offer pursuant to Sec. 10 para. 1 sentence 1 WpÜG, amounted to EUR 42.16 per MorphoSys Share. Based on this stock exchange price, the Offer Price of EUR 68.00 includes a premium of EUR 25.84 or 61.29%. |
| (ii) | The volume weighted average stock exchange price (XETRA closing price) during the last three (3) months prior to 4 February 2024, the last day prior to the publication of the Bidder’s decision to launch the Offer pursuant to Sec. 10 para. 1 sentence 1 WpÜG, amounted to EUR 28.80 per MorphoSys Share. Based on this average stock exchange price, the Offer Price of EUR 68.00 includes a premium of EUR 39.20 or 136.11%. |
| (iii) | The volume weighted average stock exchange price (XETRA closing price) during the last month prior to the unaffected 25 January 2024 close, the day before first rumors on a potential takeover of MorphoSys surfaced, amounted to EUR 35.00 per MorphoSys Share. Based on this average stock exchange price, the Offer Price of EUR 68.00 includes a premium of EUR 33.00 or 94.29%. The volume weighted average stock exchange price (XETRA closing price) during the last three (3) months prior to the unaffected 25 January 2024 close, the day before first rumors on a potential takeover of MorphoSys surfaced, amounted to EUR 28.11 per MorphoSys Share. Based on this average stock exchange price, the Offer Price of EUR 68.00 includes a premium of EUR 39.89 or 141.91%. |
40
Negotiated Result
The Management Board and the Supervisory Board firmly believe they obtained Novartis AG’s best offer and that, as of the date of the Business Combination Agreement, the Offer Price represented the highest per MorphoSys Share consideration that could reasonably be obtained, based on the fact that MorphoSys achieved an increase in the Offer Price to EUR 68.00 per MorphoSys Share in cash from Novartis AG’s original non-binding proposal of EUR 55.00 per MorphoSys Share in cash on 3 January 2024, as described in Sec. 10.1 of this Statement.
Other Negotiations
The fact that the Offer Price of EUR 68.00 was higher than the last proposal made by Company C (as defined below), which was, as described in Sec. 10.1 of this Statement, the only additional potential bidder, and the fact that Company C did subsequently not express that it would be willing to pay an offer price equal to or higher than EUR 68.00 per MorphoSys Share.
Cash Consideration
The Offer Price will be payable in cash, which provides MorphoSys Securityholders with immediate liquidity and certainty of value of the offered consideration, while eliminating long-term business and execution risk for MorphoSys Securityholders. This provides MorphoSys Securityholders with an opportunity to realize a significant part of the expected long-term value creation immediately and upfront.
Fairness Opinion
The Management Board and the Supervisory Board considered the oral opinion provided by Centerview on 10 April 2024, subsequently confirmed in the written Offer Opinion (as defined below) of Centerview dated such date, that as of such date and based upon and subject to various assumptions made, procedures followed, matters considered, and qualifications and limitations set forth in the Offer Opinion, the Offer Price to be paid to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares and (ii) MorphoSys ADSs) pursuant to the Offer Document by means of the Offer, was fair, from a financial point of view, to such holders and considered the various financial analyses undertaken by Centerview in connection with the Offer Opinion. For further details on the Offer Opinion, please see Sec. 6.2 of this Statement.
MorphoSys’ Financial Condition and Results and Outlook
The Management Board and the Supervisory Board are familiar with the current and historical financial condition and the results of MorphoSys, as well as the prospects and strategic objectives of MorphoSys. The Management Board and the Supervisory Board believe, on the basis of this familiarity, that the Offer Price to be received by MorphoSys Securityholders in the transaction exceeds MorphoSys’ standalone value, including its potential for future growth and acquisition opportunities. In arriving at this conclusion, the Management Board and the Supervisory Board have also taken into account MorphoSys’ current financial performance (as described in Sec. 3.7 of this Statement).
Statutory Minimum Price
Insofar as the Management Board and the Supervisory Board are able to verify on the basis of available information, the value of the Offer Price is in line with the provisions relating to the minimum value of the consideration offered, as set forth in Sec. 31 para. 1 and 7 WpÜG in conjunction with Secs. 4 and 5 WpÜG Offer Regulation (the “Statutory Minimum Price Requirements”):
| (i) | Pursuant to Sec. 5 WpÜG Offer Regulation, the consideration offered in the context of a takeover offer within the meaning of Secs. 29 et seq. WpÜG must be at least equal to the weighted average |
41
| German stock exchange price of the MorphoSys Shares during the three (3)-month period prior to the publication of the Bidder’s decision to launch the Offer pursuant to Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG (the “Three-Month Average Price”). The decision to launch the Offer was published on 5 February 2024. According to Sec. 10.1 of the Offer Document, the Three-Month Average Price of until and including the reference date of 4 February 2024 was communicated by BaFin as being EUR 28.80 per MorphoSys Share. The Offer Price exceeds this minimum offer price required by law; and |
| (ii) | Pursuant to Sec. 4 of the WpÜG Offer Regulation, the consideration offered for the shares of the target company in a takeover offer pursuant to Secs. 29 et seq. WpÜG must be at least equal to the value of the highest consideration paid or agreed by the Bidder, a person acting jointly with the Bidder within the meaning of Sec. 2 para. 5 sentence 1 WpÜG, or any of their subsidiaries for the acquisition of MorphoSys Shares within the last six (6) months prior to the publication of the Offer Document pursuant to Sec. 14 para. 2 sentence 1 WpÜG. According to Sec. 10.1 of the Offer Document, the Bidder, any persons acting jointly with it within the meaning of Sec. 2 para. 5 sentence 1 and 3 WpÜG and its and their subsidiaries have not made any acquisitions of any MorphoSys Shares during the last six (6) months prior to the publication of the Offer Document. |
Therefore, according to Sec. 10.1 of the Offer Document, in accordance with Sec. 31 para. 1 and para. 7 WpÜG in conjunction with Secs. 4 and 5 WpÜG Offer Regulation, the minimum offer price per MorphoSys Share amounts to EUR 28.80. The Offer Price exceeds this minimum offer price by EUR 39.20 or 136.11%.
IDW S 1 Equity Value
Based on the assessment of the IDW S 1 Expert (as defined below), the Bidder considers the equity value of MorphoSys in accordance with the Valuation Methodology (as defined below) to be more than one third lower than the equity value of MorphoSys determined on the basis of the Offer Price, in each case on a fully diluted basis (i.e., taking into account the Stock Options issued under the Incentive Plans as well as the Convertible Bonds). On this basis, the Bidder considers it highly unlikely that the DPLTA Cash Compensation (as defined below) per MorphoSys Share to be offered in connection with the implementation of the DPLTA (as defined below) and to be established in the DPLTA Report (as defined below) will exceed the Offer Price per MorphoSys Share.
MorphoSys’ finance team and the Management Board have received from the Bidder and reviewed the materials underlying the indicative S 1 valuation commissioned by the Bidder. Based on their review of these materials, MorphoSys’ finance team and the Management Board have concluded that the IDW S 1 Expert’s assessment of the equity value of MorphoSys in accordance with the Valuation Methodology is plausible. On this basis, the Management Board and the Supervisory Board believe that the Bidder’s view it highly unlikely that the DPLTA Cash Compensation per MorphoSys Share to be offered in connection with the implementation of the DPLTA and to be established in the DPLTA Report will exceed the Offer Price per MorphoSys Share is also plausible.
42
Recommendations of Research Analysts
The Offer Price of EUR 68.00 exceeds the average target price per MorphoSys Share of EUR 32.74 as set out in the analyst reports by 107.7%. The following table gives an overview of target prices and recommendations of research analysts until 4 February 2024, the last day prior to the publication of the decision to launch an offer pursuant to Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG.
| Recommendations of Research Analysts until 4 February 2024(1) | ||||||
Broker | Date of analysis | MorphoSys Target Price | Recommendation | |||
Morgan Stanley | 02-Feb-24 | EUR 38.00 | Hold | |||
EQUI.TS | 31-Jan-24 | EUR 44.00 | Hold | |||
Goldman Sachs | 30-Jan-24 | EUR 18.00 | Hold | |||
Citi | 30-Jan-24 | EUR 15.00 | Sell | |||
Landesbank Baden-Wuerttemberg | 30-Jan-24 | EUR 40.00 | Hold | |||
Leerink Partners | 30-Jan-24 | EUR 22.00 | Hold | |||
UBS AG | 30-Jan-24 | EUR 47.00 | Buy | |||
Deutsche Bank | 30-Jan-24 | EUR 25.00 | Hold | |||
Van Lanschot Kempen | 21-Dec-23 | EUR 11.00 | Sell | |||
Wells Fargo | 14-Dec-23 | EUR 62.10 | Buy | |||
JP Morgan | 05-Dec-23 | EUR 31.00 | Buy | |||
FMR Frankfurt Main Research AG | 22-Dec-23 | EUR 39.00 | Buy | |||
Oddo BHF | 21-Nov-23 | EUR 33.50 | Buy | |||
| ||||||
Average | EUR 32.74 | |||||
| (1) | Source: Bloomberg, FactSet, broker reports as of 19 March 2024. |
| 6.1.2 | Strategic and Business Rationale |
In order to evaluate the Offer as described in the Offer Document, the Management Board and the Supervisory Board considered certain strategic and business factors, including, among other things, the following factors.
A Stand-Alone Scenario is Less Attractive than a Combination with a Strong Partner
To realize the full potential of MorphoSys’ pipeline, significant additional funding is required and it is uncertain whether MorphoSys will be able to obtain the required funding in the future. A financially strong partner would allow MorphoSys to focus its efforts exclusively on progressing its pipeline assets. Progressing these pipeline assets would also be aided by a partner with additional and complementary expertise in the interaction with industry participants and regulators.
MorphoSys can further expand the pelabresib franchise: MorphoSys has strong Phase 3 data from the MANIFEST-2 trial, pointing to a paradigm shift in myelofibrosis treatment. As such, MorphoSys will file for approval in the U.S. and Europe by mid-2024. Beyond myelofibrosis, MorphoSys also sees great potential in new myeloid disease indications based on robust proof-of-concept results. With additional resources, MorphoSys can leverage pelabresib on a global scale and capture additional growth opportunities much more quickly and effectively.
MorphoSys can progress tulmimetostat with full force: Right now, tulmimetostat is being explored in a Phase 1/2 basket study as a treatment for solid tumors and lymphomas. These show deep and durable responses with tulmimetostat monotherapy heavily pre-treated patients, which physicians have expressed excitement about. As a biotech company, MorphoSys needs to focus its resources, and as such has
43
prioritized its pivotal clinical programs over tulmimetostat. The additional resources resulting from a business combination with Novartis AG are expected to allow MorphoSys to accelerate the development of this promising asset.
Novartis is a Strong Strategic Partner
Novartis is one of the largest global pharmaceutical companies with an excellent reputation and network as well as access to significant financial resources currently unavailable to MorphoSys as a standalone biotech company and commits significant financial support to MorphoSys.
Novartis’s product portfolio lacks an equivalent to pelabresib and pelabresib would be complementary to Novartis’ ruxolitinib. The efficacy of a combined administration of pelabresib and ruxolitinib formed MorphoSys’ recently completed MANIFEST-2 Study (as defined below).
| 6.1.3 | Other Considerations |
The Management Board and the Supervisory Board also considered a number of other factors, including, among other things, the factors listed below.
Likelihood of Completion of the Takeover
The Management Board and the Supervisory Board considered the nature of the Offer Conditions included in the Business Combination Agreement, as well as the likelihood of satisfaction of all those conditions. The Management Board and the Supervisory Board considered and are considering that the Offer and the Takeover pursued therewith would and will likely be consummated, based on, among other things:
| • | The size and scale of Novartis relative to MorphoSys and its history of successfully completing acquisitions; and |
| • | The likelihood that the Offer Conditions (including the minimum acceptance threshold of sixty-five percent (65%) as further specified in Sec. 5.5.3 of this Statement) will be met. |
Further, the Management Board and the Supervisory Board considered the likelihood of the merger control clearances be obtained by national antitrust authorities of the Republic of Austria, Germany and the United States in order to consummate the Takeover (the “Regulatory Clearances”). In this respect, the Management Board and the Supervisory Board considered the following:
| • | The filing in the Republic of Austria has been made on 12 February 2024. On 12 March 2024, the Federal Competition Authority (Bundeswettbewerbsbehörde) (the “FCA”) has confirmed that neither the FCA nor the Federal Cartel Prosecutor (Bundeskartellanwalt ) have filed an application to the Cartel Court for an in-depth review of the Takeover and that, as of 12 March 2024, the stand-still obligation no longer applies. |
| • | The filing in Germany has been made on 13 February 2024. On 12 March 2024, the German Federal Cartel Office (Bundeskartellamt) has confirmed that the Takeover does not fulfill the requirements of a prohibition under Sec. 36 para 1 German Act Against Restraints of Competition (Gesetz gegen Wettbewerbsbeschränkungen) and that it can be completed. |
| • | The filing in the United States has been made with the Federal Trade Commission and the Department of Justice on 16 February 2024, and withdrawn and refiled on 4 March 2024. The second statutory waiting period expired at 23:59 hours (New York Local Time) on 19 March 2024. |
All Regulatory Clearances required for the completion of the Takeover have therefore already been granted and there are no further Offer Conditions existing in this regard. For details see Sec. 11.1 of the Offer Document.
44
Post-Closing Financing
Following the Closing, Novartis AG intends to provide the MorphoSys Group with the financial resources required to continue the business of MorphoSys as contemplated to be conducted by Novartis AG. In addition, following the Closing, Novartis AG will use best efforts to provide MorphoSys with the financial resources required to pay any obligations arising from the implementation of the Offer as and when due, including, but not limited to, any obligations arising under the Convertible Bonds or in connection with the settlement of the Incentive Plans.
Timing
The Management Board and the Supervisory Board considered the anticipated timing of the completion of the Proposed Transactions, and the structure of the transaction as a public tender offer for MorphoSys Shares, which, subject to the satisfaction or previous effective waiver of the applicable Offer Conditions as set forth in the Business Combination Agreement, should provide MorphoSys Securityholders with an opportunity to receive the consideration for their MorphoSys Securities within a timeframe that is customary for transactions of this size and nature. In particular, the Management Board and the Supervisory Board have taken into account that all necessary Regulatory Clearances have already been obtained and that the Closing could take place accordingly without undue delay, but no later than on the fourth (4th) Banking Day following the publication of the tender results after the end of the Acceptance Period pursuant to Sec. 23 para. 1 no. 2 WpÜG (i.e., on 23 May 2024, subject to any extension of the Acceptance Period) and following the publication of the tender results after the end of the Additional Acceptance Period pursuant to Sec. 23 para. 1 no. 3 WpÜG (i.e., on 10 June 2024, subject to any extension of the Additional Acceptance Period), respectively. The Management Board and the Supervisory Board were and are of the view that the potential for Closing within a timeframe that is customary for transactions of this size and nature also reduces the amount of time in which MorphoSys’ business would be subject to potential disruption and uncertainty pending such Closing.
Additional Acceptance Period
The Management Board and the Supervisory Board considered that MorphoSys Securityholders that have not accepted the Offer within the Acceptance Period can still accept it within the Additional Acceptance Period, provided that all of the Offer Conditions as described in Sec. 5.5.3 of this Statement have been satisfied or previously effectively waived by the end of the Acceptance Period. The Management Board and the Supervisory Board consider this important because such Additional Acceptance Period would allow non-tendering MorphoSys Securityholders an additional opportunity to tender their MorphoSys Securities into the Offer.
Acquisition of 100% of the MorphoSys Shares
The Management Board and the Supervisory Board considered that the Offer is predicated on, among other things, the acquisition of 100% of the MorphoSys Shares or the entirety of MorphoSys’ assets and operations. The importance for Novartis to acquire full control over MorphoSys’ operations and assets and ultimately hold 100% of the MorphoSys Shares is based, among other things, on the fact that having a single shareholder and operating without a public listing increases the MorphoSys Group’s and Novartis’s ability to achieve the goals of the Proposed Transactions and implement the actions of its strategy, reduces MorphoSys Group’s costs and enables an efficient capital structure. The Management Board and the Supervisory Board considered that the structural measures, as described in Sec. 7.2 of this Statement, enable Novartis to acquire full control over MorphoSys.
The Management Board and the Supervisory Board further considered the fact that it is typical in friendly public takeover offers for companies incorporated under German law that the bidder and the target agree on certain restructurings following the completion of the offer to enable the bidder to ultimately obtain 100% ownership of the target or to otherwise obtain full control over its business as well as assets.
45
In this context, the Bidder intends to enter into a domination and profit and loss transfer agreement pursuant to Secs. 291 et seqq. AktG with MorphoSys (the “DPLTA”). Upon effectiveness of the DPLTA, the Bidder would be entitled to give binding instructions to the Management Board with regard to the management of MorphoSys and, thus, to exercise control over MorphoSys’ management. In the event that, following Closing, the Bidder holds at least (i) ninety-five percent (95%) (squeeze-out under stock corporation law), or (ii) ninety percent (90%) (merger squeeze-out under transformation law) of the share capital with voting power of MorphoSys, the Bidder intends to pursue a squeeze-out, i.e., the transfer of the MorphoSys Shares of the MorphoSys Shareholders who have not accepted the Offer to the Bidder as the principal shareholder (Hauptaktionär) in return for payment of an appropriate cash settlement pursuant to (A) Secs. 327a et seqq. AktG (squeeze-out under stock corporation law), or (B) Secs. 62 para. 5 of the German Transformation Act (Umwandlungsgesetz – “UmwG”), 327a et seqq. AktG (merger squeeze-out under transformation law) (each, a “Squeeze-out”). The Bidder has no intention to pursue a squeeze-out under takeover law.
Delisting
The Bidder intends, as soon as reasonably possible following the Closing of the Offer, to effect the delisting of the MorphoSys Shares from all regulated markets in the European Economic Area (the “Delisting”). Pursuant to the Business Combination Agreement, MorphoSys will also enable the Delisting of the MorphoSys Shares and the MorphoSys ADS from Nasdaq as well as the deregistration of the MorphoSys Shares under the U.S. Exchange Act. As a requirement of the Delisting, a delisting tender offer pursuant to Sec. 39 para. 2 of the German Stock Exchange Act (Börsengesetz – “BörsG”) would have to be made to all remaining minority shareholders of MorphoSys, whereby any such delisting tender offer would be made in accordance with the U.S. Offer Rules. The Bidder expects the mandatory offer price of the delisting offer to be equal to the Offer Price.
Following the Delisting, MorphoSys Shares would be discontinued from trading on the regulated market (Regulierter Markt) of the FSE, which could make MorphoSys Shares effectively illiquid. The Delisting would also terminate the comprehensive capital-market oriented reporting obligations of MorphoSys.
Considering all of the foregoing, the Management Board and the Supervisory Board considered these proposed Delisting desirable and in the best interest of MorphoSys and its stakeholders. While lately it was difficult for MorphoSys to benefit from the listing of the MorphoSys Shares and its access to the capital markets, MorphoSys would benefit from the termination of the comprehensive reporting obligations associated with the listing.
Fiduciary Out and MorphoSys Termination Fee
The Management Board and the Supervisory Board considered its ability under the Business Combination Agreement to terminate the Business Combination Agreement under certain circumstances after a third party published an offer document relating to an alternative offer which terms are, taking into account all elements of the relevant alternative offer, including the offer consideration per share as well as transaction certainty and timing, more favorable to MorphoSys and its shareholders than the terms of the Offer (a “Superior Offer”), if the failure to do so would be inconsistent with the Management Board’s fiduciary duties to the MorphoSys Securityholders under German law and subject to certain other limitations in the Business Combination Agreement. The Management Board and the Supervisory Board also considered MorphoSys’ obligation to pay to Novartis AG a contractual penalty of EURm 50 in cash in the event that the Business Combination Agreement is terminated
| (i) | By MorphoSys due to (A) a competing public offer for MorphoSys Shares by a third party (a “Rival Offer”) for greater than twenty percent (20%) of the MorphoSys Shares that has been settled and consummated (a “Rival Offer Termination”) or (B) the publication of a Rival Offer, for which the Management Board determines, after giving effect to the proposals made by Novartis AG, that such Rival Offer constitutes a Superior Offer (a “Rival Offer Termination”); |
46
| (ii) | By the Bidder pursuant to a Rival Offer Termination or a termination due to (A) a failure of MorphoSys to include the requirements in this Statement or the Schedule 14D-9 agreed in the Business Combination Agreement for the reasoned statement pursuant to Sec. 27 para. 1 WpÜG, or (B) a change of the content of this Statement or the Schedule 14D-9 in a manner that does not comply with the requirements for the reasoned statement pursuant to Sec. 27 para. 1 WpÜG agreed on in the Business Combination Agreement; or |
| (iii) | By either MorphoSys or the Bidder pursuant to a Superior Proposal Termination and if (A) any person will have publicly disclosed a transaction which is economically or otherwise comparable to a Competing Offer and which, if implemented, would or would be reasonably likely to prevent the Offer from being successfully completed (any such transaction, including a Rival Offer a “Competing Transaction”) after 5 February 2024 and prior to the termination of the Business Combination Agreement and such Competing Transaction has not been publicly and unconditionally withdrawn at least ten (10) Banking Days prior to such termination, and (B) within twelve (12) months of such termination, MorphoSys enters into a definitive agreement with respect to a Competing Transaction or a Competing Transaction is consummated (whereby in both cases it may not necessarily be the same Competing Transaction referred to in (A)), subject to the terms and conditions set forth in the Business Combination Agreement as further explained in Sec. 8.2.16 of the Offer Document |
(a contractual penalty due upon the termination of the Business Combination Agreement in the cases of (i), (ii) or (iii) each, a “MorphoSys Termination Fee”).
The Management Board and the Supervisory Board acknowledged that agreeing to the MorphoSys Termination Fee in the Business Combination Agreement was necessary to induce Novartis AG to enter into the Business Combination Agreement and determined, following the advice of its financial and legal advisors, that, taking into account German fiduciary standards and customary terms and conditions relating to obligation not to actively solicit a Competing Offer, would not prohibit a bona fide third party from making a Superior Offer.
Antitrust Commitments and Novartis Termination Fee
The Management Board and the Supervisory Board considered the fact that the Bidder and Novartis AG have agreed to use best efforts to procure that each and every impediment that may be asserted by any relevant authority in connection with the Regulatory Clearances is avoided or eliminated, so as to enable the Bidder to consummate the Takeover as planned, subject to certain customary regulatory limitations as further explained in Sec. 8.2.12 of the Offer Document.
The Management Board and the Supervisory Board also considered the fact that Novartis AG has agreed to pay to MorphoSys a termination payment of EURm 100 in cash (the “Novartis Termination Fee”) in case the Business Combination Agreement is terminated, amongst others, because the required Regulatory Clearances have not been obtained prior to 5 February 2024, 23:59 hours Frankfurt am Main Local Time / 17:59 hours New York Local Time, subject to the terms and conditions of the Business Combination Agreement as further explained in Sec. 8.2.17 of the Offer Document. Since the Regulatory Approvals set forth in Section 11.1 of the Offer Document have already been obtained, a Novartis Termination Fee can only be considered if the Takeover fails due to the prohibition by another competent authority.
Other Stakeholder Interests
The Management Board and the Supervisory Board considered certain stakeholder interests, including the fact that Novartis AG (i) intends to continue to use the current name “MorphoSys” as the corporate name of MorphoSys and the MorphoSys Affiliates, (ii) intends to maintain the headquarters of MorphoSys in Planegg, Germany, as well as the R&D capability in this location, and (iii) acknowledges that the dedicated workforce of the MorphoSys Group is the foundation for the current and future success of the MorphoSys Group.
47
The Management Board and the Supervisory Board further considered that pursuant to the Business Combination Agreement, Novartis AG acknowledges the outstanding leadership and management skills and capabilities as well as deep industry knowledge of the current members of the Management Board.
Other Terms of Business Combination Agreement
The Management Board and the Supervisory Board considered the other terms and conditions of the Business Combination Agreement, including, among other things, the following agreements concluded therein:
| • | The MorphoSys Group must carry on its business in all material respects in the ordinary course consistent with past practice, including the current strategy, subject to limited exceptions specified in the Business Combination Agreement. |
| • | MorphoSys will not take a number of management measures related to the conduct of its business without the prior written consent of the Bidder, subject to limited exceptions specified in the Business Combination Agreement, and that these covenants may limit the ability of MorphoSys to pursue business opportunities that it would otherwise pursue. |
| • | MorphoSys’ ability, subject to the limitations and conditions in the Business Combination Agreement, to seek specific performance against Novartis AG and the Bidder in relation to their obligations under the Business Combination Agreement, including the Bidder’s obligation to consummate the Offer on the terms of the Business Combination Agreement. |
Uncertainties Associated with the Offer
The Management Board and the Supervisory Board also considered a number of potential risks or negative implications in its deliberations concerning the Business Combination Agreement, the Proposed Transactions and the Offer, including, among other things, the following negative implications that would arise as a result of the transaction:
| • | The Management Board and the Supervisory Board considered the fact that MorphoSys Securityholders accepting the Offer will have no equity interest in MorphoSys following then consummation of the Offer, meaning that they will cease to participate in MorphoSys’ future growth and to benefit from increases in the value of MorphoSys Shares. |
| • | The Management Board and the Supervisory Board considered that the conditions to the Bidder’s obligation to accept MorphoSys Shares tendered and to pay the Offer Price pursuant to the Offer are subject to the Offer Conditions, and the possibility that such conditions may not be satisfied, including as a result of events outside of MorphoSys’ control. |
| • | The Management Board and the Supervisory Board also considered the fact that (i) if the Offer is not completed, MorphoSys’ management and other employees will have expended extensive time and effort and would have experienced significant distractions from their work during the pendency of the Offer, and (ii) MorphoSys has incurred and will incur significant transaction costs to prepare and to consummate the Offer, regardless of whether or not the Offer is consummated, and (iii) MorphoSys would have sold all of its rights to tafasitamab pursuant to the Incyte Purchase Agreement (as defined below) as a method of satisfying the Binding Offer Conditions (as defined below), to the extent not waived by Novartis AG had waived, as described in Sec. 10.1 of this Statement, regardless of whether or not the Offer is consummated. |
| • | The Management Board and the Supervisory Board also considered the fact that, if the Offer is not consummated, the perception of MorphoSys’ continuing business might result in a loss of one or more customers, business partners, collaboration partners or employees and that the trading price of MorphoSys Shares could be adversely affected. |
48
| • | The Management Board and the Supervisory Board considered the effect of the public announcement of the Proposed Transactions on MorphoSys’ operations, employees, customers and suppliers as well as its ability to attract and retain key personnel while the transaction is pending and the possibility of any suit, action or proceeding in respect of the Business Combination Agreement, the Proposed Transactions or the Takeover. |
| • | The Management Board and the Supervisory Board further considered that the financial interests of the Management Board members may be different from or in addition to those of MorphoSys Securityholders, as more fully described in Sec. 13 of this Statement, and considered whether a potential conflict of interest existed in relation to the members of the Management Board or the Supervisory Board, concluding that this was not the case. |
| 6.1.4 | Further Considerations |
For further considerations regarding the Management Board’s and the Supervisory Board’s assessment for the targets and intentions of the Bidder, Novartis Pharma and Novartis AG, please refer to Sec. 7.3 of this Statement.
| 6.2 | Opinions of Centerview |
On 5 February 2024, Centerview rendered to the Management Board and the Supervisory Board its oral fairness opinion, subsequently confirmed in a written opinion dated such date (the “Signing Opinion”), that, as of such date and based upon and subject to various assumptions made, procedures followed, matters considered, and qualifications and limitations upon the review undertaken by Centerview in preparing the Signing Opinion, the Offer Price to be paid to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Business Combination Agreement by means of the Offer, was fair, from a financial point of view, to such holders.
On 10 April 2024, Centerview rendered to the Management Board and the Supervisory Board its oral fairness opinion, subsequently confirmed in a written opinion dated such date (the “Offer Opinion” and together with the Signing Opinion, collectively, the “Centerview Opinions”), that, as of such date and based upon and subject to various assumptions made, procedures followed, matters considered, and qualifications and limitations upon the review undertaken by Centerview in preparing the Offer Opinion, the Offer Price to be paid to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs), pursuant to the Offer Document by means of the Offer, was fair, from a financial point of view, to such holders.
The full text of the Offer Opinion, which describes the assumptions made, procedures followed, matters considered, and qualifications and limitations upon the review undertaken by Centerview in preparing the Offer Opinion, is attached as Annex 1 and is incorporated herein by reference.
The summary of the Offer Opinion set forth below is qualified in its entirety to the full text of the Offer Opinion attached as Annex 1. The full text of the Offer Opinion should be read carefully in its entirety for a description of the assumptions made, procedures followed, matters considered, and qualifications and limitations upon the review undertaken by Centerview in preparing the Offer Opinion.
Centerview’s financial advisory services and the Centerview Opinions were provided solely for the information and assistance of the Management Board and the Supervisory Board (in the capacity of each such board, respectively, and not in any other capacity) in connection with and for purposes of their consideration of, with respect to the Signing Opinion, the Business Combination Agreement, and with respect to the Offer Opinion, the Takeover. The Signing Opinion only addressed the
49
fairness, from a financial point of view, as of the date thereof, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which are, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) of the Offer Price to be paid to such holders pursuant to the Business Combination Agreement. The Signing Opinion did not address any other term or aspect of the Business Combination Agreement and does not constitute a recommendation to any MorphoSys Securityholder as to whether or not such holder should tender MorphoSys Shares in connection with the Offer or how such MorphoSys Securityholder or any other person should act with respect to the Takeover or any other matter. The Offer Opinion only addressed the fairness, from a financial point of view, as of the date thereof, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which are, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) of the Offer Price to be paid to such holders pursuant to the Offer Document by means of the Offer. The Offer Opinion did not address any other term or aspect of the Offer Document or the Takeover and does not constitute a recommendation to any MorphoSys Securityholder as to whether or not such holder should tender MorphoSys Shares in connection with the Offer or how such MorphoSys Securityholder or any other person should act with respect to the Takeover or any other matter.
In connection with rendering the Centerview Opinions described above and performing its related financial analyses, Centerview reviewed, among other things:
| • | Annual Reports on Form 20-F of MorphoSys for the years ended 31 December 2022 and 31 December 2021; |
| • | Certain interim reports to shareholders on Form 6-K of MorphoSys; |
| • | Certain publicly available research analyst reports for MorphoSys; |
| • | Certain other communications from MorphoSys to its shareholders; and |
| • | Certain internal information relating to the business, operations, earnings, cash flow, assets, liabilities and prospects of MorphoSys, including the Projections described in Sec. 6.3 of this Statement prepared by management of MorphoSys and furnished to Centerview by MorphoSys for purposes of Centerview’s analysis, which are collectively referred to in this summary of Centerview’s Opinions as the “Internal Data”. |
Solely in connection with rendering the Signing Opinion described above, Centerview also reviewed:
| • | A draft of the Business Combination Agreement dated 5 February 2024; and |
| • | Annual Report on Form 20-F of MorphoSys for the year ended 31 December 2020. |
Solely in connection with rendering the Offer Opinion described above, Centerview also reviewed:
| • | A draft of the Offer Document dated 10 April 2024, referred to in this summary of Centerview’s Opinions as the “draft offer document”; |
| • | The Business Combination Agreement; and |
| • | Annual Report on Form 20-F of MorphoSys for the year ended 31 December 2023. |
Centerview also participated in discussions with the executive committee of MorphoSys (the “Executive Committee”) and other representatives of MorphoSys regarding their assessment of the Internal Data. In addition, Centerview conducted such other financial studies and analyses and took into account such other information as Centerview deemed appropriate.
Centerview assumed, without independent verification or any responsibility therefor, the accuracy and completeness of the financial, legal, regulatory, tax, accounting and other information supplied to, discussed with, or reviewed by Centerview for purposes of the Centerview Opinions and, with MorphoSys’ consent, Centerview relied upon such information as being complete and accurate. In that
50
regard, Centerview assumed, at MorphoSys’ direction, that the Internal Data (including, without limitation, the Projections) were reasonably prepared on bases reflecting the best currently available estimates and judgments of the management of MorphoSys as to the matters covered thereby and Centerview relied, at MorphoSys’ direction, on the Internal Data for purposes of Centerview’s analysis and the Centerview Opinions. At MorphoSys’ direction, Centerview converted certain figures in the Internal Data from EUR to USD based on an exchange rate as of 2 February 2024 for the Signing Opinion and the Offer Opinion. The EUR to USD exchange rate as of the time Centerview prepared and rendered the Offer Opinion was consistent with the exchange rate as of 2 February 2024. Centerview expressed no view or opinion as to the Internal Data or the assumptions on which it was based. In addition, at MorphoSys’ direction, Centerview did not make any independent evaluation or appraisal of any of the assets or liabilities (contingent, derivative, off-balance-sheet or otherwise) of MorphoSys, nor was Centerview furnished with any such evaluation or appraisal, and was not asked to conduct, and did not conduct, a physical inspection of the properties or assets of MorphoSys. Centerview also assumed, at MorphoSys’ direction, that the Offer Document would not differ in any respect material to Centerview’s analysis or the Offer Opinion from the draft offer document reviewed by Centerview. Centerview also assumed, at MorphoSys’ direction, that the Takeover will be consummated on the terms set forth in the Offer Document and in accordance with all applicable laws and other relevant documents or requirements, without delay or the waiver, modification or amendment of any term, condition or agreement, the effect of which would be material to Centerview’s analysis or the Centerview Opinions and that, in the course of obtaining the necessary governmental, regulatory and other approvals, consents, releases and waivers for the Takeover, no delay, limitation, restriction, condition or other change will be imposed, the effect of which would be material to Centerview’s analysis or the Centerview Opinions. Centerview did not evaluate and did not express any opinion as to the solvency or fair value of MorphoSys, or the ability of MorphoSys to pay its obligations when they come due, or as to the impact of the Takeover on such matters, under any state, federal or other laws relating to bankruptcy, insolvency or similar matters. Centerview is not a legal, regulatory, tax or accounting advisor, and Centerview expressed no opinion as to any legal, regulatory, tax or accounting matters.
Centerview’s assessment differs in a number of important aspects from a valuation performed by qualified auditors and/or from asset-based valuations in general. In particular, Centerview has not performed valuations based upon the guidelines published by the Institute of Chartered Accountants in Germany (Institut der Wirtschaftsprüfer in Deutschland e.V.) (“IDW”) (IDW S 1). The Centerview Opinions do not replace such valuations. Centerview expresses no view on whether, in light of the nature of the Takeover, it may be required or appropriate for MorphoSys to obtain such valuations. In addition, the Centerview Opinions have not been rendered in accordance with the IDW guidelines “Principles for the preparation of Fairness Opinions” (IDW S 8).
Centerview expressed no view as to, and the Centerview Opinions did not address, MorphoSys’ underlying business decision to proceed with or effect the Takeover, or the relative merits of the Takeover as compared to any alternative business strategies or transactions that might be available to MorphoSys or in which MorphoSys might engage. The Signing Opinion was limited to and addressed only the fairness, from a financial point of view, as of the date of the Signing Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) of the Offer Price to be paid to such holders pursuant to the Business Combination Agreement. The Offer Opinion was limited to and addressed only the fairness, from a financial point of view, as of the date of the Offer Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) of the Offer Price to be paid to such holders pursuant to the Offer Document by means of the Offer. For purposes of the Centerview Opinions, Centerview was not asked to, and Centerview did not, express any view on, and the Centerview Opinions did not address, any other term or aspect of, with respect to the Signing Opinion, the Business Combination Agreement, and with respect to the Offer Opinion, the Offer Document or the Takeover, including, without limitation, the structure or form of the Takeover, or any other agreements or arrangements contemplated by the Business
51
Combination Agreement, described in the Offer Document or entered into in connection with or otherwise contemplated by the Takeover, including, without limitation, the fairness of the Takeover or any other term or aspect of the Takeover to, or any consideration to be received in connection therewith by, or the impact of the Takeover on, the holders of any other class of securities, creditors or other constituencies of MorphoSys or any other party. Further, Centerview was not asked to consider, nor did Centerview express any view on, and the Centerview Opinions did not address, any term or aspect of the Incyte Purchase Agreement, pursuant to which certain assets of MorphoSys and MorphoSys US were sold immediately prior to the execution of the Business Combination Agreement, or how the Incyte Purchase Agreement may impact MorphoSys, the Offer Document, the Business Combination Agreement or the Takeover. In addition, Centerview expressed no view or opinion as to the fairness (financial or otherwise) of the amount, nature or any other aspect of any compensation to be paid or payable to any of the officers, directors or employees of MorphoSys or any party, or class of such persons in connection with the Takeover, whether relative to the Offer Price to be paid with respect to the Signing Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Business Combination Agreement, and with respect to the Offer Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Offer Document by means of the Offer. The Centerview Opinions were necessarily based on financial, economic, monetary, currency, market and other conditions and circumstances as in effect on, and the information made available to Centerview as of, the respective dates of the Centerview Opinions. Centerview does not have any obligation or responsibility to update, revise or reaffirm the Centerview Opinions based on circumstances, developments or events occurring after the respective dates of the Centerview Opinions. The Centerview Opinions do not constitute a recommendation to any MorphoSys Securityholder as to whether or not such holder should tender MorphoSys Shares in connection with the Offer, or how such MorphoSys Securityholder or any other person should act with respect to the Takeover or any other matter. Centerview’s financial advisory services and the Centerview Opinions were provided solely for the information and assistance of the Management Board and the Supervisory Board (in the capacity of each such board, respectively, and not in any other capacity) in connection with and for purposes of their consideration of, with respect to the Signing Opinion, the Business Combination Agreement, and with respect to the Offer Opinion, the Takeover. The issuance of each of the Centerview Opinions was approved by the Centerview Partners LLC Fairness Opinion Committee.
| 6.2.1 | Summary of Centerview Financial Analysis |
The following is a summary of the material financial analyses prepared and reviewed with the Management Board and the Supervisory Board in connection with the Centerview Opinions.
The summary set forth below does not purport to be a complete description of the financial analyses performed or factors considered by, and underlying the opinions of, Centerview, nor does the order of the financial analyses described represent the relative importance or weight given to those financial analyses by Centerview. Centerview may have deemed various assumptions more or less probable than other assumptions, so the reference ranges resulting from any particular portion of the analyses summarized below should not be taken to be Centerview’s view of the actual value of MorphoSys. Some of the summaries of the financial analyses set forth below include information presented in tabular format. In order to fully understand the financial analyses, the tables must be read together with the text of each summary, as the tables alone do not constitute a complete description of the financial analyses performed by Centerview. Considering the data in the tables below without considering all financial analyses or factors or the full narrative description of such analyses or factors, including the methodologies and assumptions underlying such analyses or factors, could create a misleading or incomplete view of the processes underlying Centerview’s financial analyses and the Centerview Opinions.
52
In performing its analyses, Centerview made numerous assumptions with respect to industry performance, general business and economic conditions and other matters, many of which are beyond the control of MorphoSys or any other parties to the Takeover. None of MorphoSys, Novartis AG, Bidder or Centerview or any other person assumes responsibility if future results are materially different from those discussed. Any estimates contained in these analyses are not necessarily indicative of actual values or predictive of future results or values, which may be significantly more or less favorable than as set forth below. In addition, analyses relating to the value of MorphoSys do not purport to be appraisals or reflect the prices at which MorphoSys Shares may actually be sold. Accordingly, the assumptions and estimates used in, and the results derived from, the financial analyses are inherently subject to substantial uncertainty. Except as otherwise noted, the following quantitative information, to the extent that it is based on market data, is based on market data as it existed on or before, for purposes of the Signing Opinion, 2 February 2024 (the last full trading day before the Management Board and the Supervisory Board approved the Takeover) and, for purposes of the Offer Opinion, 9 April 2024 (the last full trading day before Centerview rendered the Offer Opinion to the Management Board and the Supervisory Board) and is not necessarily indicative of current market conditions.
| 6.2.2 | Selected Public Company Analysis |
Centerview reviewed and compared certain financial information, ratios and multiples for MorphoSys to corresponding financial information, ratios and multiples for publicly traded companies that Centerview deemed comparable, based on its experience and professional judgment, to MorphoSys (for purposes of this section of this Statement, such selected publicly traded companies are referred to as the “Selected Company Comparison Companies”). Selected Company Comparison Companies consisted of:
| • | Agios Pharmaceuticals, Inc.; |
| • | BioCryst Pharmaceuticals, Inc.; |
| • | Day One Biopharmaceuticals, Inc.; |
| • | Deciphera Pharmaceuticals, Inc.; |
| • | Geron Corporation; |
| • | Harmony Biosciences Holdings, Inc.; |
| • | Mirum Pharmaceuticals, Inc.; |
| • | SpringWorks Therapeutics, Inc.; and |
| • | Syndax Pharmaceuticals, Inc. |
Although none of the Selected Company Comparison Companies used in this analysis is directly comparable to MorphoSys, these companies were selected, among other reasons, because they are publicly traded companies with certain operational and financial characteristics, which, for purposes of its analyses, Centerview considered to be similar to those of MorphoSys. However, because none of the Selected Company Comparison Companies is exactly the same as MorphoSys, Centerview believed that it was inappropriate to, and therefore did not, rely solely on the quantitative results of the selected public company analysis. Accordingly, Centerview also made qualitative judgments, based on its experience and professional judgment, concerning differences between the business, financial and operational characteristics and prospects of MorphoSys and the Selected Company Comparison Companies that could affect the public trading values of each in order to provide a context in which to consider the results of the quantitative analysis.
Using publicly available information obtained from SEC filings, Wall Street research analyst consensus estimates and other data sources as of 2 February 2024 for purposes of the Signing Opinion and 9 April 2024 for purposes of the Offer Opinion, Centerview calculated, for each of the Selected Company
53
Comparison Companies the following figures, ratios and multiples, (i) enterprise value (calculated as the market value of common equity determined using the treasury stock method and taking into account outstanding in-the-money options, other equity awards and other convertible securities, as applicable, plus debt and less cash, after giving effect to certain adjustments for minority interest and contingent consideration) and (ii) enterprise value as a multiple of projected revenue for the calendar year 2028 (such multiple, with respect to the Selected Company Comparison Companies is referred to as the “EV/CY’28E Revenue”). These analyses resulted for such Selected Company Comparison Companies in a median multiple of enterprise value to projected revenue for calendar year 2028 for such Selected Company Comparison Companies.
For purposes of the Offer Opinion, the resulting data were as follows:
| EV/CY’28E Revenue(1) | ||||
Agios Pharmaceuticals, Inc. | 2.1x | |||
BioCryst Pharmaceuticals, Inc. | 1.3x | |||
Day One Biopharmaceuticals, Inc. | 1.7x | |||
Deciphera Pharmaceuticals, Inc. | 2.0x | |||
Geron Corporation | 1.8x | |||
Harmony Biosciences Holdings, Inc. | 1.4x | |||
Mirum Pharmaceuticals, Inc. | 1.8x | |||
SpringWorks Therapeutics, Inc. | 3.9x | |||
Syndax Pharmaceuticals, Inc. | 3.6x | |||
Median | 1.8x | |||
| (1) | For purposes of rendering the Signing Opinion, Centerview utilized data as of 2 February 2024, which resulted in different multiples than as set forth above. |
Based on its analysis and other considerations that Centerview deemed relevant in its professional judgment and experience, Centerview selected a reference range of enterprise value to revenue multiples for the calendar year 2028 of 1.5x to 4.0x. In selecting these references ranges, Centerview made qualitative judgments based on its experience and professional judgment concerning differences between the business, financial and operational characteristics of MorphoSys and the Selected Company Comparison Companies that could affect their public trading values in order to provide a context in which to consider the results of the quantitative analysis. These qualitative judgments related, among other things, to the differing sizes, growth prospects, and commercial profiles and degree of operational risk between MorphoSys and such comparable companies. Centerview applied the enterprise value to revenue multiples for the calendar year 2028 reference range to MorphoSys’ projected adjusted revenue for the calendar year 2028 of EURm 642, as set forth in the Projections, to derive a range of implied equity values for MorphoSys. Centerview then divided such implied equity values by the number of fully-diluted outstanding MorphoSys Shares (determined using the treasury stock method based on 37.7 million MorphoSys Shares (1 MorphoSys ADS = 0.25 MorphoSys Shares) and taking into account 0.3 million outstanding in-the-money Stock Options (1.2 million MorphoSys ADS equivalent) with a weighted average exercise price of EUR 65.56, 1.0 million Restricted Stock Units (4.0 million MorphoSys ADS equivalent) and 2.2 million Performance Share Units (9.0 million MorphoSys ADS equivalent)) as of 31 January 2024, as set forth in the Internal Data, resulting in an implied per MorphoSys Share equity value range of EUR 16.75 to EUR 56.00 (rounded to the nearest EUR 0.05). Centerview then compared this range to the EUR 68.00 per MorphoSys Share value of the Offer Price to be paid, for purposes of the Signing Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Business Combination Agreement, and for purposes of the Offer Opinion, to holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Offer Document by means of the Offer.
54
| 6.2.3 | Selected Precedent Transaction Analysis |
Centerview reviewed and analyzed certain information relating to the following selected transactions that Centerview, based on its experience and professional judgment (for purposes of this section of this Statement referred to as “Selected Transactions”), deemed relevant to consider in relation to MorphoSys and the Takeover.
Although none of the Selected Transactions is directly comparable to the Takeover, the Selected Transactions listed below were selected, among other reasons, based on Centerview’s experience and professional judgment, because the Selected Transactions have certain characteristics that, for the purposes of this analysis, may be considered similar to certain characteristics of the Takeover. The reasons for and the circumstances surrounding each of the Selected Transactions analyzed were diverse and there are inherent differences in the business, operations, financial conditions and prospects of MorphoSys and the target companies included in the Selected Transactions analysis. This analysis involved complex considerations and qualitative judgments concerning differences in business, operational, and/or financial characteristics and other factors that could affect the public trading, acquisition or other values of the selected target companies and MorphoSys. However, because none of the Selected Transactions used in this analysis is identical or directly comparable to the Takeover, Centerview believed that it was inappropriate to, and therefore did not, rely solely on the quantitative results of the Selected Transactions analysis. Accordingly, Centerview also made qualitative judgments, based on its experience and professional judgment, concerning differences between the operational, business or financial characteristics of MorphoSys and each target company as well as the Takeover and the Selected Transactions that could affect the transaction values of each in order to provide a context in which to consider the results of the quantitative analysis.
Using publicly available information obtained from SEC filings and other data sources as of the time of the announcement of the Selected Transactions, Centerview calculated, for each Selected Transaction, among other things, the enterprise value implied for the applicable target company based on the consideration payable in the applicable Selected Transaction (calculated as the offer value (determined using the treasury stock method and taking into account outstanding in-the-money options, warrants, restricted stock units, outstanding unvested earnout shares and other convertible securities), plus the book value of debt and certain liabilities less cash and cash equivalents and excluding any contingent consideration), as a multiple of the target company’s five (5)-year forward revenue (such multiple, with respect to the Selected Transactions, the “EV/5-Year Fwd. Revenue Multiple”), for which financial information had been made public at the time of the announcement of such transactions.
55
For purposes of the Offer Opinion, the resulting data were as follows:
Date Announced | Target | Acquiror | EV/5-Year Fwd. Revenue Multiple | |||||
February 2024 | CymaBay Therapeutics, Inc. | Gilead Sciences, Inc. | 5.0x | (1) | ||||
May 2023 | CTI BioPharma Corp. | Swedish Orphan Biovitrum AB | 2.1x | |||||
January 2023 | Albireo Pharma, Inc. | Ipsen, S.A. | 1.0x | |||||
January 2023 | Amryt Pharma PLC | Chiesi Farmaceutici S.p.A. | 1.8x | |||||
August 2022 | ChemoCentryx, Inc. | Amgen Inc. | 3.7x | |||||
April 2022 | Sierra Oncology, Inc. | GlaxoSmithKline plc | 2.8x | |||||
January 2022 | Zogenix, Inc. | UCB S.A. | 2.3x | |||||
September 2021 | Kadmon Holdings, Inc. | Sanofi SA | 3.7x | |||||
February 2021 | Viela Bio, Inc. | Horizon Therapeutics Public Limited Company | 5.6x | |||||
May 2020 | Portola Pharmaceuticals, Inc. | Alexion Pharmaceuticals, Inc. | 3.1x | |||||
Median | 2.9x | (2) | ||||||
| (1) | The transaction between CymaBay Therapeutics, Inc. and Gilead Sciences, Inc. (the “CymaBay Transaction”) had not occurred as of the date of the Signing Opinion and therefore was not considered for purposes of rendering the Signing Opinion. |
| (2) | For purposes of rendering the Signing Opinion, the median EV/5-Year Fwd. Revenue Multiple did not include the CymaBay Transaction and therefore differed from the multiple set forth above. |
Based on its analysis of the relevant metrics for each of the target companies in the Selected Transactions and other considerations that Centerview deemed relevant in its professional judgment and experience, Centerview selected a reference range of enterprise value to five (5)-year forward revenue multiples of 2.0x to 4.0x. In selecting this reference range, Centerview made qualitative judgments based on its experience and professional judgment concerning differences between the business, financial and operational characteristics and prospects of MorphoSys and the target companies included in the Selected Transactions and other factors that could affect the public trading, acquisition or other values of such companies or MorphoSys.
Centerview applied this reference range to MorphoSys’ five (5)-year forward adjusted revenue of EURm 699 (representing three quarters of 2028 adjusted revenue and one quarter of 2029 adjusted revenue), as set forth in the Projections, to derive a range of implied equity values for MorphoSys. Centerview then divided these implied equity values by the number of fully-diluted outstanding MorphoSys Shares as of 31 January 2024, as set forth in the Internal Data, resulting in an implied per MorphoSys Share equity value range of EUR 27.40 to EUR 61.55 (rounded to the nearest EUR 0.05). Centerview then compared this range to the EUR 68.00 per MorphoSys Share value of the Offer Price to be paid for purposes of the Signing Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs), without interest, pursuant to the Business Combination Agreement, and for purposes of the Offer Opinion, to holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs), without interest, pursuant to the Offer Document by means of the Offer.
| 6.2.4 | Discounted Cash Flow Analysis |
Centerview performed a discounted cash flow analysis of MorphoSys based on the Projections as set forth in the Internal Data as further described below in Sec. 6.3 of this Statement, which reflect certain assumptions, including projected future financing needs of MorphoSys. A discounted cash flow analysis is a traditional valuation methodology used to derive a valuation of an asset or set of assets by calculating the “present value” of estimated future cash flows of the asset or set of assets. “Present value” refers to the
56
current value of future cash flows and is obtained by discounting those future cash flows by a discount rate that takes into account macroeconomic assumptions and estimates of risk, the opportunity cost of capital, expected returns and other appropriate factors.
In performing this analysis, Centerview calculated a range of equity values for the MorphoSys Shares by (a) discounting to present value as of 31 March 2024 using discount rates ranging from 11.5% to 13.5% (based on Centerview’s analysis of MorphoSys’ weighted average cost of capital) and using a mid-year convention: (i) the forecasted risk-adjusted, after-tax unlevered free cash flows of MorphoSys over the period beginning on 1 April 2024 and ending on 31 December 2044, as set forth in the Projections, utilized by Centerview at the direction of MorphoSys management and as approved by the Management Board for use by Centerview as described in Sec. 6.3 of this Statement, (ii) an implied terminal value of MorphoSys, calculated by Centerview by assuming that (as directed by MorphoSys management) MorphoSys’ unlevered free cash flows would decline in perpetuity after 31 December 2044 at a rate of free cash flow decline of 40% year over year, and (iii) tax savings from usage of MorphoSys’ estimated U.S. federal and German net operating losses and MorphoSys’ estimated future losses, as provided by MorphoSys management and as described in Sec. 6.3 of this Statement, (b) adding to the foregoing results, MorphoSys’ estimated net cash balance of as of 31 March 2024, as provided by MorphoSys management and as described in Sec. 6.3 of this Statement, and (c) subtracting from the foregoing results the present value of the impact of assumed equity raises, as instructed by MorphoSys management and as described in Sec. 6.3 of this Statement.
Centerview then divided these implied equity values by the number of fully-diluted outstanding MorphoSys Shares as of 31 January 2024, as set forth in the Internal Data, resulting in an implied per MorphoSys Share equity value range of EUR 42.45 to EUR 52.30 (rounded to the nearest EUR 0.05). Centerview then compared the results of the above analysis to the EUR 68.00 per MorphoSys Share value of the Offer Price to be paid for purposes of the Signing Opinion, to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Business Combination Agreement, and for purposes of the Offer Opinion, to holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Offer Document by means of the Offer.
| 6.2.5 | Other Factors |
Centerview noted for the Management Board and the Supervisory Board certain additional factors solely for reference and informational purposes only, including, among other things, the following:
| • | Historical Stock Trading Price Analysis. Centerview reviewed historical closing trading prices of the MorphoSys Shares during the fifty-two (52)-week period ended 2 February 2024 (the last full trading day before the Management Board and the Supervisory Board approved the signing of the Business Combination Agreement), which reflected low and high stock closing prices for MorphoSys during such period of approximately EUR 13.64 to EUR 42.16 per MorphoSys Share; |
| • | Analyst Price Target Analysis. Centerview reviewed stock price targets for the MorphoSys Shares in publicly available Wall Street research analyst reports as of 2 February 2024 (the last full trading day before the Management Board and the Supervisory Board approved the signing of the Business Combination Agreement), which indicated low and high stock price targets for MorphoSys ranging from EUR 11.00 to EUR 63.30 per MorphoSys Share, converted in certain cases from USD to EUR based on an exchange rate as of 2 February 2024 for the Signing Opinion and the Offer Opinion; and |
| • | Precedent Premium Paid Analysis. Centerview performed an analysis of premiums paid in the Selected Transactions involving publicly traded biopharmaceutical companies as set forth in Sec. 6.2.3 of this Statement, for which premium data were available. The premiums in this analysis were calculated by comparing the per share acquisition price in each transaction (excluding contingent |
57
consideration, if any) to the closing price of the target company’s common stock as of the last full unaffected trading day. Based on the foregoing analysis and other considerations that Centerview deemed relevant in its professional judgment and experience, Centerview applied a range of 50% to 115% to the MorphoSys Share price on 25 January 2024, the last full unaffected trading day, of EUR 35.93, which resulted in an implied price range of EUR 53.90 to EUR 77.25 (each rounded to the nearest EUR 0.05), respectively, per MorphoSys Share. |
| 6.2.6 | General |
The preparation of a financial opinion is a complex analytical process involving various determinations as to the most appropriate and relevant methods of financial analysis and the application of those methods to the particular circumstances and, therefore, a financial opinion is not readily susceptible to summary description. In arriving at its opinions, Centerview did not draw, in isolation, conclusions from or with regard to any factor or analysis that it considered. Rather, Centerview made its determination as to fairness on the basis of its experience and professional judgment after considering the results of all of the analyses.
Centerview’s financial analyses and the Centerview Opinions were only one of many factors taken into consideration by the Management Board and the Supervisory Board in its evaluation of the Takeover. Consequently, the analyses described above should not be viewed as determinative of the views of the Management Board and the Supervisory Board or management of MorphoSys with respect to the consideration for the Takeover or as to whether the Management Board and the Supervisory Board would have been willing to determine that a different consideration for the Takeover was fair. The consideration for the Takeover was determined through arm’s-length negotiations between MorphoSys and Novartis AG and was approved by the Management Board and the Supervisory Board. Centerview provided advice to MorphoSys during these negotiations. Centerview did not, however recommend any specific amount of consideration to MorphoSys or the Management Board or the Supervisory Board or that any specific amount of consideration constituted the only appropriate consideration for the Takeover.
Centerview is a securities firm engaged directly and through affiliates and related persons in a number of investment banking, financial advisory and merchant banking activities. From the date that was two (2) years prior to the date of the Signing Opinion until the date of the Offer Opinion, except for Centerview’s current engagement, Centerview had not been engaged to provide financial advisory or other services to MorphoSys, and Centerview did not receive any compensation from MorphoSys during such period. From the date that was two (2) years prior to the date of the Signing Opinion until the date of the Offer Opinion, Centerview was not engaged to provide financial advisory or other services to Novartis AG or the Bidder, and Centerview did not receive any compensation from Novartis AG or the Bidder during such period. Centerview may provide financial advisory and other services to or with respect to MorphoSys or Novartis AG or their respective Affiliates in the future, for which Centerview may receive compensation. Certain (i) of Centerview’s and its affiliates’ directors, officers, members and employees, or family members of such persons, (ii) of Centerview’s affiliates or related investment funds and (iii) investment funds or other persons in which any of the foregoing may have financial interests or with which they may co-invest, may at any time acquire, hold, sell or trade, in debt, equity and other securities or financial instruments (including derivatives, bank loans or other obligations) of, or investments in, MorphoSys, Novartis AG or any of their respective affiliates, or any other party that may be involved in the Takeover.
The Management Board and the Supervisory Board selected Centerview as its financial advisor in connection with the Takeover based on Centerview’s familiarity with MorphoSys, their reputation as an internationally recognized investment banking firm and their substantial experience in similar transactions. Centerview is an internationally recognized investment banking firm that has substantial experience in transactions similar to the Takeover.
In connection with Centerview’s services as the lead financial advisor to the Management Board and the Supervisory Board, MorphoSys has agreed to pay Centerview an aggregate fee of approximately
58
USDm 59.1, USDm 1 of which was payable in connection with the rendering of the Centerview Opinions and approximately USDm 58.1 of which is payable contingent upon consummation of the Takeover. In addition, MorphoSys has agreed to reimburse certain of Centerview’s expenses arising, and to indemnify Centerview against certain liabilities that may arise, out of Centerview’s engagement.
| 6.3 | Certain Unaudited Prospective Financial Information of MorphoSys |
MorphoSys does not, as a matter of course, publicly disclose forecasts or internal projections as to future performance, or results of operations, earnings or other results, due to, among other things, the inherent difficulty of predicting financial performance for future periods and the likelihood that the underlying assumptions and estimates may not be realized.
MorphoSys regularly prepares and maintains long-range financial forecasts for accounting purposes in the ordinary course of business. At the instruction of the Management Board and in connection with the evaluation of the Offer by the Management Board and the Supervisory Board, between December 2023 and February 2024, the Executive Committee, the MorphoSys finance team and the Management Board updated MorphoSys’ year end 2023 long-range financial forecast to reflect certain non-public, unaudited, risk-adjusted, prospective financial information for fiscal years 2024 through 2044 (the “Forecasts”, and collectively with the Unlevered Free Cash Flow (as defined below) described below, the “Projections”). The Forecasts were prepared based on MorphoSys’ continued operation as a stand-alone company and do not take into account the Takeover, including the effect of any business or strategic decision or action that has been or will be taken as a result of the execution of the Business Combination Agreement. The Forecasts have not been revised or amended based on developments that have occurred after the date on which they were prepared, including the Tafasitamab Sale (as defined below) and any business or strategic decision or action that has been or will be taken as a result of the Tafasitamab Sale. The Forecasts do not take into account the Tafasitamab Sale.
Where applicable, the Forecasts assume an exchange rate of EUR 1 = USD 1.080 as of 2 February 2024.
59
The following table presents a summary of the Forecasts:
Projections
Forecasts (Risk Adjusted)
| Fiscal year ended 31 December (all amounts in USDm, unless otherwise specified) | ||||||||||||||||||||||||||||||||||||||||||||
| 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 | ||||||||||||||||||||||||||||||||||
Monjuvi Net Revenue | 95 | 152 | 207 | 243 | 289 | 363 | 377 | 396 | 410 | 306 | 184 | |||||||||||||||||||||||||||||||||
Net Revenue | 141 | 198 | 326 | 559 | 889 | 1,195 | 1,365 | 1,515 | 1,640 | 1,608 | 1,593 | |||||||||||||||||||||||||||||||||
Adjusted Net Revenue(1) | 45 | 55 | 140 | 386 | 693 | 941 | 1,122 | 1,254 | 1,358 | 1,404 | 1,416 | |||||||||||||||||||||||||||||||||
Gross Profit(2) | 116 | 120 | 282 | 471 | 762 | 1,031 | 1,220 | 1,313 | 1,429 | 1,446 | 1,448 | |||||||||||||||||||||||||||||||||
Monjuvi R&D Expense | (232 | ) | (173 | ) | (147 | ) | (107 | ) | (20 | ) | (15 | ) | (12 | ) | (12 | ) | (8 | ) | (8 | ) | — | |||||||||||||||||||||||
Monjuvi R&D Expense (Incyte Share) | 128 | 95 | 81 | 59 | 11 | 8 | 7 | 7 | 4 | 5 | — | |||||||||||||||||||||||||||||||||
Total R&D Expense | (232 | ) | (152 | ) | (142 | ) | (102 | ) | (40 | ) | (29 | ) | (28 | ) | (28 | ) | (28 | ) | (27 | ) | (22 | ) | ||||||||||||||||||||||
Monjuvi S&M Expense | (71 | ) | (72 | ) | (76 | ) | (73 | ) | (74 | ) | (75 | ) | (76 | ) | (77 | ) | (78 | ) | (46 | ) | (26 | ) | ||||||||||||||||||||||
Monjuvi S&M Expense (Incyte Share) | 36 | 36 | 38 | 37 | 37 | 37 | 38 | 38 | 39 | 23 | 13 | |||||||||||||||||||||||||||||||||
Total S&M Expense | (64 | ) | (97 | ) | (119 | ) | (125 | ) | (126 | ) | (128 | ) | (129 | ) | (129 | ) | (130 | ) | (115 | ) | (106 | ) | ||||||||||||||||||||||
Total G&A Expense | (59 | ) | (60 | ) | (38 | ) | (37 | ) | (41 | ) | (43 | ) | (43 | ) | (43 | ) | (41 | ) | (40 | ) | (39 | ) | ||||||||||||||||||||||
Monjuvi U.S. Profit to Partner | (33 | ) | (33 | ) | (41 | ) | (66 | ) | (94 | ) | (115 | ) | (127 | ) | (133 | ) | (140 | ) | (120 | ) | (71 | ) | ||||||||||||||||||||||
EBIT(3) | (272 | ) | (222 | ) | (59 | ) | 141 | 462 | 716 | 892 | 980 | 1,089 | 1,143 | 1,210 | ||||||||||||||||||||||||||||||
| Fiscal year ended 31 December (all amounts in USDm, unless otherwise specified) | ||||||||||||||||||||||||||||||||||||||||||||
| 2035 | 2036 | 2037 | 2038 | 2039 | 2040 | 2041 | 2042 | 2043 | 2044 | |||||||||||||||||||||||||||||||||||
Monjuvi Net Revenue | 110 | 66 | 40 | 24 | 14 | 9 | 5 | 3 | 2 | 1 | ||||||||||||||||||||||||||||||||||
Net Revenue | 1,554 | 1,541 | 715 | 431 | 357 | 300 | 254 | 220 | 130 | 106 | ||||||||||||||||||||||||||||||||||
Adjusted Net Revenue(1) | 1,444 | 1,456 | 619 | 394 | 326 | 296 | 252 | 218 | 129 | 105 | ||||||||||||||||||||||||||||||||||
Gross Profit(2) | 1,418 | 1,410 | 649 | 382 | 315 | 286 | 244 | 211 | 123 | 101 | ||||||||||||||||||||||||||||||||||
Monjuvi R&D Expense | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||
Monjuvi R&D Expense (Incyte Share) | — | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||
Total R&D Expense | (17 | ) | (18 | ) | (18 | ) | (19 | ) | (19 | ) | — | — | — | — | — | |||||||||||||||||||||||||||||
Monjuvi S&M Expense | (16 | ) | (10 | ) | (6 | ) | (4 | ) | (2 | ) | (1 | ) | (1 | ) | (0 | ) | (0 | ) | (0 | ) | ||||||||||||||||||||||||
Monjuvi S&M Expense (Incyte Share) | 8 | 5 | 3 | 2 | 1 | 1 | 0 | 0 | 0 | 0 | ||||||||||||||||||||||||||||||||||
Total S&M Expense | (102 | ) | (99 | ) | (74 | ) | (43 | ) | (31 | ) | (25 | ) | (20 | ) | (15 | ) | (12 | ) | (10 | ) | ||||||||||||||||||||||||
Total G&A Expense | (37 | ) | (36 | ) | (32 | ) | (33 | ) | (32 | ) | (24 | ) | (19 | ) | (14 | ) | (15 | ) | (11 | ) | ||||||||||||||||||||||||
Monjuvi U.S. Profit to Partner | (43 | ) | (25 | ) | (15 | ) | (9 | ) | (5 | ) | (3 | ) | (2 | ) | (1 | ) | (1 | ) | (0 | ) | ||||||||||||||||||||||||
EBIT(3) | 1,219 | 1,232 | 510 | 279 | 228 | 234 | 203 | 181 | 96 | 79 | ||||||||||||||||||||||||||||||||||
| (1) | “Adjusted Net Revenue” means Net Revenue minus half of Monjuvi product revenue minus upfront and milestone payments from collaborators minus outbound royalties to Xencor and Royalty Pharma. |
| (2) | “Gross Profit” means Net Revenue minus cost of goods sold for products minus cost of goods sold for royalties. |
| (3) | “EBIT” means earnings before interest and taxes and refers to Gross Profit minus research and development expenses minus sales and marketing expenses and minus general and administrative expenses minus profit to partner for Monjuvi in the U.S. |
60
The Projections also include unlevered free cash flow for the fiscal years 2024 through 2044 (the “Unlevered Free Cash Flow”), which were reviewed and approved by the Management Board for use by Centerview in connection with its financial analyses and rendering the Centerview Opinions. The Unlevered Free Cash Flow refer to EBIT minus tax expenses, capital expenditures and changes in net working capital plus depreciation and amortization, in each case, as included in the Forecasts.
Various judgments and assumptions were made when preparing the Projections, all of which were provided to Centerview by the Management Board. These include, among others:
| (i) | Estimated cash of EURm 596 as of 31 March 2024; |
| (ii) | An aggregate principal amount of EURm 262 outstanding under the Convertible Bonds at a conversion price of EUR 131.29 per MorphoSys Share, due October of 2025; |
| (iii) | Repayment of the Royalty Pharma Development Funding Bond calculated at 2.2 times the initial amount drawn of EURm 278 (i.e., EURm 611); |
| (iv) | Hypothetical USDm 300 and USDm 200 equity raises conducted in 2025 and 2026, respectively, at an illustrative 10% discount and a 5% spread; |
| (v) | EURm 595 in U.S. federal net operating loss carryforwards as of 31 December 2022, U.S. federal R&D tax credits of EURm 26 as of 31 December 2022, German corporate net operating loss carryforwards of EURm 301 as of 31 December 2022, and German trade tax net operating loss carryforwards of EURm 308 as of 31 December 2022; and |
| (vi) | Taxes based on a U.S. tax rate of 23.7% and German tax rate of 26.7%. |
For purposes of calculating the discounted cash flow, and as described further in Sec. 6.2 of this Statement, at the instruction of the Management Board, Centerview calculated, which calculations were reviewed and approved by the Management Board for use by Centerview, the estimated (i) benefit of taxes saved from tax attributes generated as a result of future estimated losses based on a U.S. tax rate of 23.7% and German tax rate of 26.7% and (ii) the impact of the cost of a future equity raise assuming a hypothetical USDm 300 equity raise in 2025 and assuming a hypothetical USDm 200 equity raise in 2026, which, in each case (i) and (ii) are not reflected in the values in the table below.
Projections
Unlevered Free Cash Flow (Risk Adjusted)
| Financial year ended 31 December (all amounts in USDm, unless otherwise specified) | ||||||||||||||||||||||||||||||||||||||||||||
| 2024 | 2025 | 2026 | 2027 | 2028 | 2029 | 2030 | 2031 | 2032 | 2033 | 2034 | ||||||||||||||||||||||||||||||||||
EBIT | (272 | ) | (222 | ) | (59 | ) | 141 | 462 | 716 | 892 | 980 | 1,089 | 1,143 | 1,210 | ||||||||||||||||||||||||||||||
Tax Expense | — | — | — | (34 | ) | (112 | ) | (175 | ) | (218 | ) | (240 | ) | (267 | ) | (280 | ) | (297 | ) | |||||||||||||||||||||||||
Depreciation & Amortization | 1 | 3 | 3 | 3 | 5 | 7 | 8 | 9 | 10 | 10 | 11 | |||||||||||||||||||||||||||||||||
Capital Expenditures | (1 | ) | (3 | ) | (3 | ) | (3 | ) | (5 | ) | (7 | ) | (8 | ) | (9 | ) | (10 | ) | (10 | ) | (11 | ) | ||||||||||||||||||||||
Change in Net Working Capital | 1 | (3 | ) | (22 | ) | (65 | ) | (80 | ) | (65 | ) | (47 | ) | (34 | ) | (27 | ) | (5 | ) | (4 | ) | |||||||||||||||||||||||
Unlevered Free Cash Flow(1) | (271 | ) | (224 | ) | (81 | ) | 42 | 269 | 476 | 627 | 705 | 796 | 858 | 909 | ||||||||||||||||||||||||||||||
Unlevered Free Cash Flow (EUR) | (251 | ) | (208 | ) | (75 | ) | 39 | 249 | 440 | 581 | 653 | 737 | 794 | 841 | ||||||||||||||||||||||||||||||
| (1) | Unlevered free cash flow is a non-IFRS financial measure. |
61
| Financial year ended 31 December (all amounts in USDm, unless otherwise specified) | ||||||||||||||||||||||||||||||||||||||||||||
| 2035 | 2036 | 2037 | 2038 | 2039 | 2040 | 2041 | 2042 | 2043 | 2044 | |||||||||||||||||||||||||||||||||||
EBIT | 1,219 | 1,232 | 510 | 279 | 228 | 234 | 203 | 181 | 96 | 79 | ||||||||||||||||||||||||||||||||||
Tax Expense | (299 | ) | (303 | ) | (123 | ) | (67 | ) | (55 | ) | (57 | ) | (49 | ) | (44 | ) | (23 | ) | (19 | ) | ||||||||||||||||||||||||
Depreciation & Amortization | 11 | 12 | 8 | 6 | 6 | 5 | 5 | 5 | 2 | 1 | ||||||||||||||||||||||||||||||||||
Capital Expenditures | (11 | ) | (12 | ) | (8 | ) | (6 | ) | (6 | ) | (5 | ) | (5 | ) | (5 | ) | (2 | ) | (1 | ) | ||||||||||||||||||||||||
Change in Net Working Capital | (8 | ) | (4 | ) | 215 | 57 | 17 | 14 | 11 | 8 | 22 | 6 | ||||||||||||||||||||||||||||||||
Unlevered Free Cash Flow(1) | 911 | 926 | 602 | 269 | 190 | 191 | 165 | 145 | 95 | 66 | ||||||||||||||||||||||||||||||||||
Unlevered Free Cash Flow (EUR) | 844 | 857 | 557 | 249 | 176 | 177 | 153 | 134 | 88 | 61 | ||||||||||||||||||||||||||||||||||
| (1) | Unlevered free cash flow is a non-IFRS financial measure. |
The Projections were presented to the Management Board and the Supervisory Board in connection with its consideration of the Offer. The Projections were approved by the Management Board for use by Centerview in connection with the rendering of the Centerview Opinions to the Management Board and the Supervisory Board and in performing the related financial analyses, and, at the instruction of the Management Board, were relied upon by Centerview in connection with the rendering of its Centerview Opinions to the Management Board and the Supervisory Board and in performing its financial analyses as described in Sec. 6.2 of this Statement. The Projections were the only financial projections with respect to MorphoSys used by Centerview in performing its financial analyses. For the avoidance of doubt, MorphoSys did not update the Projections prior to Centerview rendering the Offer Opinion or conducting its updated financial analysis as described in Sec. 6.2 of this Statement.
MorphoSys is summarizing the Projections in this Statement to provide MorphoSys Securityholders with access to certain non-public, unaudited, risk-adjusted prospective financial information that was updated for the Management Board and the Supervisory Board for the purposes described above. MorphoSys makes and has made no representation to Novartis AG or the Bidder, in the Business Combination Agreement or otherwise, concerning any projected financial information, including the Projections. MorphoSys did not provide Novartis AG or the Bidder the Projections prior to entering into the Business Combination Agreement.
The Projections were based upon certain financial, operating and commercial assumptions and estimates developed solely using the information available to the Executive Committee, the MorphoSys finance team and the Management Board at the time the Projections were created.
Cautionary Note About the Projections
The Projections, while necessarily presented with numerical specificity, were based on numerous variables and assumptions and estimates, some as to future events, that were inherently uncertain and many of which were beyond MorphoSys’ control. The Projections reflect numerous estimates and assumptions made by the Executive Committee, the MorphoSys finance team and the Management Board, based on information available at the time the Projections were developed, with respect to industry performance and competition, regulatory conditions, general business, economic, market and financial conditions and matters specific to MorphoSys’ product candidates and Monjuvi, all of which were difficult to predict and many of which were beyond MorphoSys’ control. As a result, there can be no assurance that the Projections accurately reflect future trends or accurately estimate the future market for MorphoSys’ product candidates and Monjuvi. There can be no assurance of the market for, approval of or the timing of such approval of, MorphoSys’ product candidates, and it is possible that other products will be preferable. The Projections also reflect assumptions and estimates as to certain business decisions that are subject to change. Important factors that may affect actual results and cause the Projections not to be
62
achieved include, but are not limited to, (i) MorphoSys’ continued ownership of intellectual property (including rights to Monjuvi and tafasitamab, which MorphoSys subsequently sold after the creation of the Projections and immediately prior to the execution of the Business Combination Agreement pursuant to the Incyte Purchase Agreement), (ii) the timing of regulatory approvals and introductions of new products, (iii) market acceptance of new products, (iv) success of clinical testing, (v) availability of third-party reimbursement, (vi) success of MorphoSys’ ongoing collaboration programs (including with Incyte) and potential future collaborations with third parties through which MorphoSys may develop or commercialize product candidates, (vii) impact of competitive products and pricing, (viii) the availability and use of net operating losses, (ix) the effect of regulatory actions, (x) the effect of global economic conditions, (xi) fluctuations in foreign currency exchange rates, (xii) the cost and effect of changes in tax and other legislations and (xiii) other risk factors described in MorphoSys’ Annual Report on Form 20-F for the fiscal year ended 31 December 2023 and its subsequent Current Reports on Form 6-K. In addition, the Projections may be affected by MorphoSys’ ability to achieve strategic goals, objectives and targets over the applicable periods. As a result, there can be no assurance that the Projections will be realized, and actual results may be materially better or worse than those contained in the Projections. The Projections cover multiple years and, by their nature, become subject to greater uncertainty with each successive year. The Projections were prepared prior to the entry into the Incyte Purchase Agreement, were not subsequently updated to reflect the entry into the Incyte Purchase Agreement and therefore do not take into account the Tafasitamab Sale.
In light of the foregoing factors and the uncertainties inherent in the Projections, MorphoSys Securityholders are cautioned not to place undue, if any, reliance on the Projections. The Projections were not prepared with a view toward public disclosure.
The inclusion of the Projections in this Statement should not be regarded as an indication that MorphoSys, a MorphoSys Affiliate or any of its advisors or representatives (including Centerview) considered or consider the Projections to be predictive of actual future events, and the Projections should not be relied upon as such or construed as financial guidance. Further, the inclusion of the Projections in this Statement does not constitute an admission or representation by MorphoSys, the Management Board and the Supervisory Board, or any of their affiliates, their advisors or representatives (including Centerview) that the information presented is material. Neither MorphoSys nor any MorphoSys Affiliate, their advisors or representatives (including Centerview) assumes any responsibility for the accuracy of this information. Neither MorphoSys nor any MorphoSys Affiliate, advisors, officers, directors or representatives (including Centerview) can give any assurance that actual results will not differ from the Projections, and none of them undertakes any obligation to update or otherwise revise or reconcile the Projections to reflect circumstances existing after the date the Projections were generated or to reflect the occurrence of future events, even in the event that any or all of the assumptions and estimates underlying the Projections are shown to be in error. In this regard, it should be noted that the Projections were prepared prior to the entry into the Incyte Purchase Agreement, were not subsequently updated to reflect the entry into the Incyte Purchase Agreement and therefore do not take into account the Tafasitamab Sale. MorphoSys does not intend to make publicly available any update or other revision to the Projections, except as otherwise required by law.
Neither MorphoSys nor any of its affiliates, advisors, officers, directors or representatives (including Centerview) has made or makes any representation or warranty to any MorphoSys Securityholder or other person, including Novartis AG or the Bidder, regarding the actual performance of MorphoSys compared to the information contained in the Projections, or that the Projections will be realized. The Projections were prepared based on MorphoSys’ continued operation as a stand-alone company and do not take into account the Takeover, including the effect of any business or strategic decision or action that has been or will be taken as a result of the execution of the Business Combination Agreement or the Takeover. The Projections have not been revised or amended based on developments that have occurred after the date on which they were prepared, including the Tafasitamab Sale and any business or strategic decision or action that has been or will be taken as a result of the Tafasitamab Sale. The Projections are subjective in many
63
respects and are forward looking statements that remain subject to interpretation. In this regard, please refer to Sec. 2.5 of this Statement.
As a foreign private issuer under the U.S. securities laws, MorphoSys is not required to prepare consolidated financial statements in accordance with the U.S. generally accepted accounting principles (“U.S. GAAP”) and instead prepares its consolidated financial statements in accordance with IFRS, as issued by the International Accounting Standards Board. The Projections were not prepared with a view to complying with published SEC guidelines, the guidelines established by the American Institute of Certified Public Accountants for preparation and presentation of financial forecasts or U.S. GAAP. Following past practice, the Executive Committee, the MorphoSys finance team and the Management Board prepared the Projections in accordance with IFRS. However, the unlevered free cash flow is a non-IFRS financial measure. Non-IFRS financial measures should not be considered in isolation from, or as a substitute for, financial information presented in compliance with IFRS, and non-IFRS financial measures as used by MorphoSys may not be comparable to similarly titled amounts used by other companies. MorphoSys has not reconciled any non-IFRS financial measures included in the Projections in accordance with IFRS and has not reconciled any non-U.S. GAAP financial measures included in the Projections in accordance with U.S. GAAP because it is not required by law to do so and such conversions would involve significant time and cost. In addition, the Projections were not prepared with the assistance of, or reviewed, compiled or examined by, independent accountants. The Projections are not being included in this Statement to influence any stockholder’s decision on whether to tender their MorphoSys Shares in the Offer, but instead are being included because the Projections were provided to the Management Board and the Supervisory Board to evaluate the Offer and other potential strategic opportunities and were provided to Centerview in connection with the rendering of the Centerview Opinions to the Management Board and the Supervisory Board and in performing its financial analyses as described in Sec. 6.2 of this Statement. The Projections may differ from publicly available analyst estimates, and the Projections do not take into account any events or circumstances after the date they were prepared, including the announcement of the Takeover and the Tafasitamab Sale.
MorphoSys does not intend to update or otherwise revise the Projections to reflect circumstances existing after the date when made or to reflect the occurrence of future events, even in the event that any or all of the assumptions and estimates underlying the Projections are no longer appropriate.
The Projections were prepared, based on updates to MorphoSys’ long-range financial forecasts prepared in the ordinary course of business, between December 2023 and February 2024 by the Executive Committee, the MorphoSys finance team and the Management Board based on their assumptions and estimates about MorphoSys’ continued operation as a stand-alone, publicly traded company, with respect to the development and commercialization of MorphoSys’ product candidates and sales of Monjuvi, including risk and probability adjustments reflecting the Executive Committee, the MorphoSys finance team and the Management Board’s good faith assessment as to the probability of success for MorphoSys’ product candidates and pipeline programs as of the time the Projections were prepared. The Projections include assumptions and estimates regarding sales of Monjuvi, MorphoSys’ collaborations (including with Incyte), out-licensing programs and potential future collaborations for tulmimetostat with third parties, including upfront payments, royalties and milestone payments received in connection with such collaborations and licensing arrangements. The Projections were based on certain internal assumptions and estimates about the probability of success associated with regulatory approvals, launch timing, pricing, sales ramp, market share, competition, market exclusivity, research and development expenses, sales and marketing expenses, general and administrative expenses, effective tax rate and utilization of net operating losses, any future equity raises conducted by MorphoSys, and other relevant factors related to MorphoSys’ long-range operating plan.
The foregoing is a summary of certain key assumptions and estimates and does not purport to be a comprehensive or exhaustive overview of all metrics, assumptions and estimates included or reflected in
64
the Projections. For information on factors that may cause MorphoSys’ future results to vary materially, please refer to Sec. 2.5 of this Statement.
| 6.4 | Adequacy of the Offer Price and other Terms of the Business Combination Agreement |
The Management Board and the Supervisory Board have, each individually, thoroughly evaluated the adequacy (Angemessenheit) of the consideration within the meaning of Sec. 31 para. 1 sentence 1 WpÜG offer in connection with the Offer and the other arrangements included in the Business Combination Agreement. Such evaluations are based on the facts and criteria set out in Secs. 6.1 through 6.3 of this Statement.
While the Management Board and the Supervisory Board have based their evaluations on all facts and criteria set out in Secs. 6.1 through 6.3 of this Statement, the Management Board and Supervisory Board would like to stress the following aspects in respect of the Offer Price, which are set out in more detail in Sec. 6.1.1 of this Statement:
| • | The cash consideration offered is in line with the Statutory Minimum Price Requirements as to the type of consideration and insofar as the Management Board and the Supervisory Board are able to verify on the basis of available information, the amount of the Offer Price also meets the Statutory Minimum Price Requirements. |
| • | The Offer Price represents a substantial premium between 61.29% and 136.11% to the historical market prices of MorphoSys Shares on the FSE. |
| • | In the opinion of the Management Board and Supervisory Board, as of the date of the Business Combination Agreement, the Offer Price of EUR 68.00 represented the highest per MorphoSys Share consideration reasonably obtainable. This is supported by the fact that following discussions and negotiations with various interested parties, the Offer Price was higher than the price per MorphoSys Share in the latest proposals made by any other party. |
After having carefully assessed the content of the Offer Opinion, the Management Board and the Supervisory Board believe that the Offer Opinion support the view of the Management Board and of the Supervisory Board that the Offer Price is fair from a financial point of view towards the MorphoSys Securityholders. The Management Board and the Supervisory Board have each independently assured themselves of the plausibility of the approaches underlying the Offer Opinion, also taking into account events and circumstances since the date of the Offer Opinion. On that basis, the Management Board and the Supervisory Board consider the Offer Opinion relevant for their evaluation of the Offer Price as included in this Statement.
As a result of their careful individual assessment and consideration of qualified external advice, the Management Board and the Supervisory Board each unanimously came to the following conclusions:
| • | The Offer Price is attractive and constitutes adequate (angemessen) consideration; |
| • | The Offer Price is fair to the MorphoSys Shareholders from a financial point of view; and |
| • | The arrangements included in the Business Combination Agreement are in the best interest of MorphoSys and the MorphoSys Securityholders, the sustainable success of its business and of its clients, employees and other stakeholders. |
| 7 | OBJECTIVES AND INTENTIONS OF THE BIDDER, NOVARTIS PHARMA AND NOVARTIS AG AS WELL AS EXPECTED CONSEQUENCES FOR MORPHOSYS |
| 7.1 | Objectives and Intentions set forth in the Offer Document |
The uniform intentions of the Bidder, Novartis Pharma and Novartis AG as set forth in Sec. 9 of the Offer Document are summarized below. As set out in the Offer Document, these intentions are essentially based
65
on the Business Combination Agreement. On the basis of careful consideration and to the best knowledge of MorphoSys, neither the Bidder nor Novartis Pharma nor Novartis AG have any intentions deviating from the intentions and undertakings laid out in Secs. 9.1 to 9.6 of the Offer Document.
| 7.1.1 | Future Business Activity and Strategy, Assets and Future Obligations of MorphoSys |
Future Business Activity and Strategy
According to Sec. 9.1.1 of the Offer Document, the Bidder, Novartis Pharma and Novartis AG intend to further strengthen their position in oncology and hematology, developing and commercializing innovative medicines that transform the lives of cancer patients. The financial resources of the Bidder, Novartis Pharma and Novartis AG, their additional scientific expertise and strong global commercial infrastructure provide a great benefit to MorphoSys Group as it helps accelerate the development and commercialization potential of pelabresib at a greater speed and scale and to further the development of tulmimetostat.
The willingness of Novartis AG and the Bidder to enter into the Business Combination Agreement and agree to the Offer (including the Offer Price) was predicated on, among other things, the acquisition of 100% of the MorphoSys Shares by the Bidder. The Offer is intended to be the first in a series of related transactions to achieve the acquisition of 100% of the MorphoSys Shares. Subject to and until the possible conclusion of a DPLTA between MorphoSys and the Bidder, the conduct of the business operations of MorphoSys based upon the business strategy currently implemented or further developed in the future as well as the final determination of the strategy for MorphoSys’ business and its implementation – subject to required approvals by the Supervisory Board – remain the responsibility of the Management Board. All integration measures taken or proposed by the Bidder will be at arm’s length.
Following the consummation of the Takeover, the Bidder, Novartis Pharma and Novartis AG intend to fully support MorphoSys in its strategy and to explore opportunities to ensure that the development and commercialization of pelabresib and tulmimetostat is successful. To facilitate this support, they intend to explore effective ways of cooperation and information exchange as is necessary to enable MorphoSys Group to fully benefit from the resources and know-how of the Bidder, Novartis Pharma and Novartis AG. The Bidder, Novartis Pharma and Novartis AG trust in MorphoSys to use, subject to compliance by MorphoSys with all applicable laws, best efforts to take all actions and conduct all activities (including providing access to all data, results and other relevant information of the members of the MorphoSys Group) reasonably requested by the Bidder, Novartis Pharma and Novartis AG in connection with the drafting, preparation, filing and submission of any regulatory permit or other regulatory filing or submission with respect to any MorphoSys product, including pelabresib.
Assets and Future Obligations
According to Sec. 9.1.2 of the Offer Document, the Bidder acknowledges the integrity of MorphoSys Group and its business and major assets. The Bidder, Novartis Pharma and Novartis AG intend to enter into detailed discussions with the Management Board regarding the future utilization of MorphoSys’ operational assets and business operations to ensure the successful commercialization of pelabresib and development of tulmimetostat. Any sale or other disposal of the business or material assets of MorphoSys Group requires prior consent of the Management Board and Supervisory Board even after consummation of the Takeover.
Subject to and until the possible conclusion of a DPLTA, the Bidder, Novartis Pharma and Novartis AG intend not to take any actions relating to, initiate or otherwise support the distribution of an extraordinary dividend (Sonderdividende), the change of the existing dividend policy (Dividendenpraxis) of MorphoSys or other distributions (e.g., through share buy-backs). Instead, the respective dividend policy will duly consider the financial needs of MorphoSys in light of its business strategy currently implemented or further developed in the future by the Management Board.
66
Following the Closing, the Bidder, Novartis Pharma and Novartis AG intend to provide the MorphoSys Group with the financial resources required to continue its business as contemplated to be conducted by Novartis AG. In addition, following the Closing, the Bidder, Novartis Pharma and Novartis AG will use best efforts to provide MorphoSys with the financial resources required to pay any obligations arising from the implementation of the Offer as and when due, including, but not limited to, any obligations arising under the Convertible Bonds or in connection with the settlement of the Incentive Plans.
In case of a termination of bank financing of MorphoSys as a consequence of the change of control following the Closing, the Bidder, Novartis Pharma and Novartis AG intend to provide financing assistance on arm’s length basis vis-à-vis MorphoSys and/or provide assistance to MorphoSys to obtain new credit lines from Bidder’s and/or Novartis’ relationship bank(s) to substitute any bank financing.
The Bidder, Novartis Pharma and Novartis AG have no intentions which would result in an increase of MorphoSys Group’s current indebtedness outside the ordinary course of business.
Company Name
According to Sec. 9.1.3 of the Offer Document, the Bidder, Novartis Pharma and Novartis AG intend to continue to use the current name “MorphoSys” as the corporate name of MorphoSys and its subsidiaries (for the subsidiaries, subject to customary pre- and suffixes to distinguish them from MorphoSys). However, under the Business Combination Agreement, the Bidder, Novartis Pharma and Novartis AG may consider (i) rebranding the MorphoSys products to align with product names and branding customarily used by Novartis, including indicating that such MorphoSys products are sold by Novartis AG, or (ii) requiring all trademarks and other branding of the MorphoSys Group or any MorphoSys product to include a mark or other designation indicating that the MorphoSys Group is a part of Novartis AG’s affiliated group. However, the Bidder, Novartis Pharma and Novartis AG have no such intention.
| 7.1.2 | Headquarters of MorphoSys |
According to Sec. 9.2 of the Offer Document, the Bidder, Novartis Pharma and Novartis AG intend to maintain the registered office and the administrative headquarters of MorphoSys in Planegg, Germany following Closing. Additionally, in consideration of the complementary nature of the products and technologies of MorphoSys, the Bidder, Novartis Pharma and Novartis AG also intend to maintain the R&D capability in this location following Closing.
| 7.1.3 | Employees and Employment Conditions |
According to Sec. 9.3 of the Offer Document, the Bidder acknowledges that the dedicated workforce of the MorphoSys Group is the foundation for the current and future success of the MorphoSys Group and that the current and future success of MorphoSys depends on the creativity and performance of MorphoSys Group’s workforce and their potential for innovation, both of which heavily rely on the competence and the commitment of the employees of MorphoSys.
The Bidder, Novartis Pharma and Novartis AG intend to support the Management Board in maintaining and developing an attractive and competitive framework to retain an highly qualified employee base and in the continued effort to attract talents. The Bidder, Novartis Pharma and Novartis AG intend to use best efforts to retain the MorphoSys Group’s talents amongst its employees. They believe they can properly incentivize such employees consistently with their incentivization of similarly situated employees of Novartis AG and its subsidiaries and further believe they can provide such employees a welcoming and stimulating workplace and an opportunity to align their individual strengths and goals with corresponding opportunities with the MorphoSys Group as well as potentially across Novartis’ global operations.
67
The Bidder, Novartis Pharma and Novartis AG have no further intentions regarding employees and conditions of employment. In particular, the Bidder, Novartis Pharma and Novartis AG have no intentions regarding any employee representation at the level of the MorphoSys Group.
| 7.1.4 | Modification of Remuneration System and Incentive Plans |
Secs. 8.2.8 to 8.2.10 of the Offer Document contain a description of certain undertakings by Novartis AG and MorphoSys with respect to the MorphoSys Group employees’ participation in the remuneration system and the Incentive Plans of MorphoSys.
The Business Combination Agreement provides that, after the Closing, the parties will review MorphoSys’ existing remuneration system for the Management Board as well as the long-term incentive programs for the Management Board and MorphoSys Group employees, and MorphoSys will consider the impact of the Takeover and/or a potential subsequent delisting on the participation of the Management Board and the MorphoSys Group’s employees in the existing Incentive Plans in consultation with Novartis AG and the Bidder.
For a description of the modification of the remuneration system and the Incentive Plans as well as the settlement of the Incentive Plans, see Secs. 13.1.1 and 13.1.2 of this Statement.
| 7.1.5 | Management Board and Supervisory Board of MorphoSys |
According to Sec. 9.4 of the Offer Document, the Bidder acknowledges the outstanding leadership and management skills and capabilities as well as deep industry knowledge of the current members of the Management Board. The Bidder. Novartis Pharma and Novartis AG intend to work with the members of the Management Board to develop a detailed integration plan, to the extent legally permissible, that will help ensure a smooth transition while best positioning MorphoSys’ business for future growth and continued success.
See Secs. 13.1.1 and 13.1.2 of this Statement for a description of certain undertakings by the Bidder, Novartis Pharma and Novartis AG and MorphoSys with respect to the Management Board members’ participation in the existing remuneration system and the Incentive Plans.
Subject to and until the possible conclusion of a DPLTA (see Sec. 9.5.1 of the Offer Document), the Bidder, Novartis Pharma and Novartis AG do not intend to issue instructions to the Management Board.
The Bidder, Novartis Pharma and Novartis AG intend to have, following the Closing, a representation on the Supervisory Board which reflects its shareholding following the Closing. The Bidder, Novartis Pharma and Novartis AG intends its representatives to be appointed in accordance with a respective resolution of the MorphoSys’ shareholders’ meeting and/or by the competent court pursuant to Sec. 104 AktG. The Bidder, Novartis Pharma and Novartis AG and MorphoSys expect such appointment to become effective as of the date the resignation of certain current Supervisory Board members becomes effective.
| 7.2 | Structural Measures |
According to Sec. 9.5 of the Offer Document, the Management Board will continue to manage MorphoSys independently and in its own responsibility. However, the Bidder, Novartis Pharma and Novartis AG intend to implement (or cause to be implemented) certain legal integration measures (as described in Sec. 8.2.6 of the Offer Document) if deemed necessary for the successful development and commercialization of pelabresib and tulmimetostat. These legal integration measures may include the implementation of structuring measures set forth below and in Secs. 9.5.1 to 9.5.4 of the Offer Document. The Bidder and Novartis AG have, under the Business Combination Agreement, explicitly reserved the right to reasonably request MorphoSys to pursue corporate structuring measures such as enterprise
68
agreements (Unternehmensverträge) pursuant to Secs. 291 et seqq. AktG, legal transformations, a squeeze-out under stock corporation law, a merger squeeze-out under transformation law, a squeeze-out under takeover law and any other restructuring or reorganization of the MorphoSys Group.
| 7.2.1 | Domination and Profit and Loss Transfer Agreement |
According to Sec. 9.5.1 of the Offer Document, the Bidder, Novartis Pharma and Novartis AG intend to enter into a DPLTA. Upon effectiveness of the DPLTA, the Bidder would be entitled to give binding instructions to the Management Board with regard to the management of MorphoSys and, thus, to exercise control over MorphoSys’ management. As a result of the effectiveness of the DPLTA, the Bidder would be obliged to compensate any annual net losses (Jahresfehlbetrag) that would be incurred by MorphoSys if no DPLTA was in place and which would not be offset by any withdrawals from other revenue reserves created during the term of the DPLTA. Conversely, MorphoSys would be obliged to transfer to the Bidder all annual net profits (Gewinn) that would accrue without such transfer of profits, less any losses carried forward and any amounts appropriated to the legal reserves. Furthermore, the DPLTA would provide, amongst other things, for an obligation of the Bidder (i) to acquire any MorphoSys Shares held by minority MorphoSys Shareholders, at their request, against payment of an adequate compensation (Abfindung) in cash (the “DPLTA Cash Compensation”), and (ii) to compensate the remaining minority MorphoSys Shareholders whose MorphoSys Shares would not be acquired pursuant to item (i) by paying an annually recurring compensation (Ausgleich) (the “Recurring DPLTA Payment”). The amounts of the DPLTA Cash Compensation and the Recurring DPLTA Payment would be agreed upon by the Bidder and MorphoSys in the DPLTA. The Bidder’s management and the Management Board would issue a joint report explaining and substantiating the terms and conditions of the DPLTA from a legal and financial point of view and especially the nature and the level of the DPLTA Cash Compensation and of the Recurring DPLTA Payment (the “DPLTA Report”). Also, the underlying valuation of MorphoSys to determine those amounts (the “DPLTA Valuation”) would have to be validated by a court-appointed appraiser (Vertragsprüfer) who must review the DPLTA and issue a written report (Prüfungsbericht) on its findings (the “Appraiser Review Report”). The DPLTA valuation must be based on the circumstances prevailing at the time when MorphoSys’ general meeting passes the relevant resolution on the DPLTA.
The DPLTA Valuation to be set forth in the DPLTA Report must be based on the “Principles for the Performance of Business Valuations (IDW S 1)” in its current version (status: 2 April 2008) issued by the IDW (these principles the “IDW S 1 Standard”) and according to the “Best Practice Recommendations Corporate Valuation” in its current version (status: December 2012) published by the German Association for Financial Analysis and Asset Management e.V. (Deutsche Vereinigung für Finanzanalyse und Asset Management e.V.) (“DVFA” and together with the IDW S 1 Standard the “Valuation Methodology”).
An independent financial expert engaged by Novartis AG (the “IDW S 1 Expert”) has determined ranges of a DPLTA Valuation of MorphoSys based MorphoSys’ current business plan provided by MorphoSys for its valuation and publicly available information using the Valuation Methodology. The valuation was based on the application of a risk-equivalent cost of capital and the results were further validated using the multiple-method, applying trading multiples of listed peer group companies and multiples derived from recent comparable transactions.
Based on this valuation, the Bidder considers the equity value of MorphoSys in accordance with the Valuation Methodology to be more than one third lower than the equity value of MorphoSys determined on the basis of the Offer Price, in each case on a fully diluted basis.
Therefore, the Bidder considers it highly unlikely that the DPLTA Cash Compensation per MorphoSys Share to be offered in connection with the implementation of the DPLTA and to be established in the DPLTA Report will exceed the Offer Price per MorphoSys Share.
69
| 7.2.2 | Delisting |
In addition, according to Sec. 9.5.2 of the Offer Document, the Bidder, Novartis Pharma and Novartis AG intend to effect the Delisting as soon as reasonably possible following the Closing. Pursuant to the Business Combination Agreement, MorphoSys will also enable a delisting of the MorphoSys Shares and the MorphoSys ADS from Nasdaq as well as the deregistration of the MorphoSys Shares under the U.S. Exchange Act.
As a requirement of the Delisting, a delisting tender offer pursuant to Sec. 39 para. 2 BörsG would have to be made to all remaining minority shareholders of MorphoSys. The Bidder expects the mandatory offer price of the delisting offer to be equal to the Offer Price.
Any such delisting tender offer will also be made in accordance the U.S. Offer Rules.
Following the Delisting, MorphoSys Shares would be discontinued from trading on the regulated market (Regulierter Markt) of the FSE, which could make MorphoSys Shares effectively illiquid. The Delisting would also terminate the comprehensive capital-market oriented reporting obligations of MorphoSys.
| 7.2.3 | Termination of the ADS Deposit Agreement |
In connection with the Delisting, MorphoSys will terminate the amended and restated deposit agreement among MorphoSys, the ADS Depositary and owners and holders of MorphoSys ADSs, dated as of 18 April 2018 (the “ADS Deposit Agreement”), pursuant to which the ADS Depositary maintains an ADS facility for the MorphoSys Shares. When the ADS Deposit Agreement is terminated, holders of MorphoSys ADSs will only have the right to receive MorphoSys Shares underlying MorphoSys ADSs upon surrender of MorphoSys ADSs and payment of applicable fees of the ADS Depositary. At any time after the date of termination, which shall be at least ninety (90) days following dissemination to MorphoSys ADS Holders of a notice of termination, the ADS Depositary may sell the remaining deposited MorphoSys Shares held under the ADS Deposit Agreement and hold the proceeds of such sale for the benefit of holders of MorphoSys ADSs that have not been surrendered.
| 7.2.4 | Squeeze-out Under Stock Corporation Law or Merger Squeeze-out Under Transformation Law or Squeeze-out Under Takeover Law |
According to Sec. 9.5.4 of the Offer Document, in the event that, following Closing, the Bidder holds at least (i) ninety-five percent (95%), or (ii) ninety percent (90%) of the share capital with voting power of MorphoSys, the Bidder, Novartis Pharma and Novartis AG intend to pursue a Squeeze-out pursuant to (A) Secs. 327a et seqq. AktG (squeeze-out under stock corporation law), or (B) Secs. 62 para. 5 UmwG, 327a et seqq. AktG (merger squeeze-out under transformation law). The Bidder has no intention to pursue a squeeze-out under takeover law.
If the general meeting of MorphoSys resolves the transfer of the MorphoSys Shares of the MorphoSys Shareholders who have not accepted the Offer to the Bidder in return for payment of an appropriate cash settlement pursuant to Secs. 327a et seqq. AktG or Secs. 62 para. 5 UmwG, 327a et seqq. AktG, as the case may be, the amount of the cash settlement to be paid would be determined by reference to the circumstances prevailing at the time of the passing by the general meeting of the resolution on the transfer of the shares. The adequacy of the amount of the cash settlement can be reviewed in a judicial award procedure. The amount of the appropriate cash settlement could be equal to, but could also be higher or lower than, the Offer Price of EUR 68.00 per MorphoSys Share. However, the Bidder considers it highly unlikely that the cash settlement to be paid in connection with the implementation of a squeeze-out will exceed the Offer Price per MorphoSys Share (see Sec. 6.1.1 of this Statement). The implementation of a squeeze-out of the minority shareholders under stock corporation law would result in a delisting of MorphoSys. In case of the implementation of a merger squeeze-out of the minority shareholders under the transformation law, MorphoSys would cease to exist.
70
The Bidder, Novartis Pharma and Novartis AG intend to pursue a squeeze-out under stock corporation law or a merger squeeze-out under transformation law. The Bidder, Novartis Pharma and Novartis AG have no intention to pursue a squeeze-out under takeover law.
| 7.3 | Evaluation of the Objectives and Intentions of the Bidder, Novartis Pharma and Novartis AG and Expected Consequences for MorphoSys |
The Management Board and the Supervisory Board have, each individually, thoroughly assessed the intentions of the Bidder, Novartis Pharma and Novartis AG set forth in Sec. 9 of the Offer Document and described in Secs. 7.1 and 7.2 of this Statement. Such intentions accurately reflect the mutual agreement between Novartis AG and MorphoSys, set forth in the Business Combination Agreement. In the view of the Management Board and the Supervisory Board, the agreed upon objectives and intentions provide the framework for a successful implementation of the acquisition, serve the best interest of MorphoSys and all its stakeholders and offer substantial strategic advantages for MorphoSys with a view to long-term value creation.
The Management Board and the Supervisory Board welcome the intentions of the Bidder, Novartis Pharma and Novartis AG as described in Sec. 7.1 of this Statement, in particular in respect of:
| • | Future business activity, strategy, assets, and future obligations of MorphoSys. In the view of the Management Board and the Supervisory Board, such support and collaboration is likely to be of advantage for MorphoSys’ overall business. The Management Board and the Supervisory Board note that the Bidder’s financial resources, additional scientific expertise and strong global commercial infrastructure provide a considerable benefit to MorphoSys Group as it helps accelerating the development and maximizing the commercialization potential of pelabresib at a greater speed and scale; |
| • | The assets and future obligations of MorphoSys. In particular, the Management Board and the Supervisory Board value the best efforts commitment of the Bidder, Novartis Pharma and Novartis AG to provide MorphoSys with the financial resources required to pay any obligations arising from the implementation of the Offer as and when due, including, but not limited to, any obligations arising under the Convertible Bonds or in connection with the settlement of the Incentive Plans. |
| • | The continued use of the current name “MorphoSys” as the corporate name of MorphoSys and its subsidiaries (for the subsidiaries, subject to customary pre- and suffixes to distinguish them from the parent company). |
| • | The intention to maintain the registered office and the administrative headquarters of MorphoSys in Planegg, Germany, as well as the R&D capability in this location, as described in Sec. 7.1.2 of this Statement. |
| • | MorphoSys’ employees and the respective employment conditions, as described in Sec. 7.1.3 of this Statement. |
| • | The proposed treatment of MorphoSys’ remuneration system and Incentive Plans, as described in Sec. 7.1.4 of this Statement, which is comprehensible, fair to and in the best interest of MorphoSys’ employees. |
| • | The cooperation with the Management Board and the composition of the Supervisory Board following the Closing as described in Sec. 7.1.5 of this Statement. In particular, the Management Board and the Supervisory Board value the intention of the Bidder, Novartis Pharma and Novartis AG to work with the Management Board to develop a detailed integration plan, to the extent legally permissible, that will help ensure a smooth transition while best positioning MorphoSys’ business for future growth and continued success. |
71
| • | The structural measures, such as the DPLTA, the Delisting and any squeeze-out procedures, as described in Sec. 7.2 of this Statement. In particular, the Management Board and the Supervisory Board value the simplification of the shareholder structure and the elimination of extensive publication and reporting obligations as a result of the Delisting. |
| 8 | CONSEQUENCES FOR MORPHOSYS SECURITYHOLDERS |
The following information is to assist MorphoSys Securityholders in assessing the consequences of accepting or not accepting the Offer. It reflects certain considerations the Management Board and Supervisory Board deem relevant in connection with such an evaluation. However, this cannot and does not purport to be a complete list of considerations individual MorphoSys Securityholders may deem relevant, in particular due to differing individual circumstances. MorphoSys Securityholders must therefore form their own opinion as to the consequences of accepting or not accepting the Offer in light of their individual circumstances.
The Management Board and the Supervisory Board reiterate that they do not and cannot offer an assessment of the tax consequences for individual MorphoSys Securityholders, respectively, including whether accepting or not accepting the Offer would result in tax disadvantages (in particular regarding any capital gains tax liabilities). The Management Board and the Supervisory Board note that the Bidder has provided certain information on material German and U.S. federal income tax consequences in Sec. 20 of the Offer Document.
The Management Board and the Supervisory Board recommend that the MorphoSys Securityholders obtain expert advice as appropriate.
| 8.1 | Possible Consequences of Acceptance of the Offer |
All MorphoSys Securityholders intending to accept the Offer should take into account, inter alia, the following:
| • | Those MorphoSys Securityholders that accept or have accepted the Offer will (subject to the Offer ultimately being consummated) no longer profit from any positive performance of the stock exchange price of the MorphoSys Shares or from any favorable development in MorphoSys’ business with the Tendered MorphoSys Shares. |
| • | Furthermore, as set forth in Sec. 17 of the Offer Document, MorphoSys Securityholders that accept or have accepted the Offer may only withdraw their declared acceptance prior to the end of the Acceptance Period. The Management Board and the Supervisory Board note that the Closing of the Offer and the payment of the Offer Price will not take place until all Offer Conditions have either been fulfilled or effectively waived. |
| • | Until one (1) FSE Trading Day following expiration of the Acceptance Period, all Tendered MorphoSys Shares will be traded under the separate ISIN DE000A4BGGU0, and will therefore not be fungible with the MorphoSys Shares not tendered for sale. If the acceptance rate is low, liquidity in such Tendered MorphoSys Shares may also be low. Trading under the respective separate ISIN may take place at a different price than the trading of MorphoSys Shares not tendered in the Offer. MorphoSys Shares represented by MorphoSys ADSs that are tendered in the Offer may not be traded. MorphoSys ADSs that are tendered in the Offer will be blocked on the books of the ADS Depositary or DTC until the MorphoSys Shares represented by MorphoSys ADSs are accepted for purchase, or the MorphoSys ADSs are withdrawn, or returned in the case of termination of the Offer. |
| • | The Bidder is, subject to certain limitations, allowed to acquire additional MorphoSys Shares outside of the Offer, via the stock exchange or off-market at any time. After the Closing, the acquisition by the Bidder of additional MorphoSys Shares on a stock exchange at a higher price than the Offer Price will not require the Bidder to adjust the Offer Price for the benefit of those MorphoSys |
72
Securityholders that accepted the Offer. After the Closing and the expiration of the one (1)-year period pursuant to Sec. 31 para. 5 WpÜG, also acquisitions by the Bidder of additional MorphoSys Shares at a higher price off-market, will not require the Bidder to adjust the Offer Price for the benefit of those MorphoSys Securityholders that have already accepted the Offer. |
| • | After the Closing, the Bidder is expected to have the required majority at a general shareholders’ meeting of MorphoSys to implement certain significant corporate law structural measures. Possible structural measures include, changes to the articles of association, capital increases, exclusion of subscription rights of the MorphoSys Shareholders in capital measures, mergers, dissolution and other structural measures. MorphoSys Shareholders that accept the Offer will not participate in any compensation which is payable by law in the event of certain structural measures realized after the Closing, which compensation may be higher or lower than the Offer Price. As per Sec. 9.5.1 of the Offer Document, the Bidder considers it highly unlikely that the DPLTA Cash Compensation per MorphoSys Share to be offered in connection with the implementation of the DPLTA and to be established in the DPLTA Report will exceed the Offer Price per MorphoSys Share. |
| • | If the general meeting of MorphoSys resolves the transfer of the MorphoSys Shares of the MorphoSys Shareholders who have not accepted the Offer to the Bidder in return for payment of an appropriate cash settlement pursuant to Secs. 327a et seqq. AktG or Secs. 62 para. 5 UmwG, 327a et seqq. AktG, as the case may be, the amount of the cash settlement to be paid would be determined by reference to the circumstances prevailing at the time of the passing by the general meeting of the resolution on the transfer of the shares. The adequacy of the amount of the cash settlement can be reviewed in a judicial award procedure. The amount of the appropriate cash settlement could be equal to, but could also be higher or lower than, the Offer Price of EUR 68.00 per MorphoSys Share. As per Sec. 9.5.1 of the Offer Document, the Bidder considers it highly unlikely that the cash settlement to be paid in connection with the implementation of a squeeze-out will exceed the Offer Price per MorphoSys Share. |
| 8.2 | Possible Consequences of Non-Acceptance of the Offer |
MorphoSys Securityholders who do not intend to accept the Offer should take the following into account:
| • | MorphoSys Shares in respect of which the Offer is not accepted may still be traded on the FSE for as long as they remain listed. However, the present stock market price of the MorphoSys Shares reflects the fact that the Bidder published its decision to launch the Offer on 5 February 2024. It is uncertain whether, following completion of the Takeover, the stock market price of the MorphoSys Shares will remain at its present level, rise above it or fall below it. |
| • | Completion of the Takeover will result in a reduction of the free float of the issued MorphoSys Shares. It is further expected that the supply of and the demand for MorphoSys Shares will be lower than today after completion of the Takeover and therefore that the liquidity of the MorphoSys Shares will decrease. It will therefore be possible, that buy and sell orders with respect to MorphoSys Shares cannot be executed or cannot be executed in a timely fashion. Moreover, the possible limitation of the liquidity of MorphoSys Shares could result in substantially larger price fluctuations of the MorphoSys Shares in the future. |
| • | Depending upon factors such as liquidity, market value of the remaining MorphoSys Shares and the number of MorphoSys Shares purchased pursuant to the Offer, it is possible that, following the Offer, the MorphoSys Shares would no longer constitute “margin securities” for the purposes of the margin regulations of the Federal Reserve Board and, therefore, could no longer be used as collateral for loans made by brokers. |
| • | The Offer is intended to be the first in a series of related transactions to achieve the acquisition of 100% of the MorphoSys Shares. After completion of the Takeover, the Bidder will have the voting majority at the general meeting and could, depending on the acceptance rate, also have the necessary |
73
voting majority to enforce all important structural and other measures under corporate law at the general meeting of MorphoSys. This includes, for example, election and dismissal of shareholder representatives of the Supervisory Board, granting or rejecting discharge of Management Board or Supervisory Board members, amendments to the articles of association, capital increases and, if the majority requirements under statutory law and articles of association have been satisfied, exclusion of subscription rights for shareholders in capital measures as well as reorganizations, mergers and dissolution of MorphoSys. Only in the case of some of the aforementioned measures there would be an obligation for the Bidder under German law to submit to the minority shareholders, on the basis of a company valuation of MorphoSys, an offer to acquire their MorphoSys Shares in exchange for reasonable compensation or to grant other compensation. Because such company valuation would have to be based on circumstances existing at the time of the resolution adopted by the MorphoSys general meeting for the respective measure, such offer for compensation could be equivalent in value to the Offer Price but it could also be lower or higher. Based on the assessment of the IDW S 1 Expert in accordance with Sec. 9.5.1 of the Offer Document, the Bidder considers it highly unlikely that the compensation in connection with certain structural measures will exceed the Offer Price per MorphoSys Share (see Sec. 6.1.1 of this Statement). The implementation of some of these measures could also result in the delisting of the MorphoSys Shares. |
| • | Further, the Bidder intends to enter into a DPLTA with MorphoSys as controlled entity. Upon effectiveness of the DPLTA, the Bidder would be entitled to give binding instructions to the Management Board with regard to the management of MorphoSys and, thus, to exercise control over MorphoSys’ management. As a result of the effectiveness of the DPLTA, the Bidder would be obliged to compensate any annual net losses (Jahresfehlbetrag) that would be incurred by MorphoSys if no DPLTA was in place and which would not be offset by any withdrawals from other revenue reserves created during the term of the DPLTA. Conversely, MorphoSys would be obliged to transfer to the Bidder all annual net profits (Gewinn) that would accrue without such transfer of profits, less any losses carried forward and any amounts appropriated to the legal reserves. Furthermore, the DPLTA would provide, amongst other things, for an obligation of the Bidder to (i) acquire any MorphoSys Shares held by minority MorphoSys Shareholders, at their request, against payment of the DPLTA Cash Compensation, and (ii) compensate the remaining minority MorphoSys Shareholders whose MorphoSys Shares would not be acquired pursuant to item (i) by paying a Recurring DPLTA Payment. The amounts of the DPLTA Cash Compensation and the Recurring DPLTA Payment would be agreed upon by the Bidder and MorphoSys in the DPLTA. The Bidder’s management and the Management Board would issue a DPLTA Report. The underlying DPLTA Valuation would have to be validated by a court-appointed appraiser (Vertragsprüfer) who must issue an Appraiser Review Report. The DPLTA valuation must be based on the circumstances prevailing at the time when MorphoSys’ general meeting passes the relevant resolution on the DPLTA. Based on the assessment of the IDW S 1 Expert, the Bidder considers the equity value of MorphoSys in accordance with the Valuation Methodology to be more than one third lower than the equity value of MorphoSys determined on the basis of the Offer Price, in each case on a fully diluted basis. On this basis, the Bidder considers it highly unlikely that the DPLTA Cash Compensation per MorphoSys Share to be offered in connection with the implementation of the DPLTA and to be established in the DPLTA Report will exceed the Offer Price per MorphoSys Share. |
| • | Additionally, the Bidder intends, as soon as reasonably possible, to effect the discontinuation of the stock listing of the MorphoSys Shares on the FSE and – inter alia – on the regulated unofficial market of the stock exchange in Berlin as well as on the unregulated market of the stock exchanges in Düsseldorf, Hamburg, Hanover, Munich and Stuttgart as well as via Tradegate Exchange. According to the Business Combination Agreement, MorphoSys will also enable a delisting of the MorphoSys Shares and the MorphoSys ADS from Nasdaq as well as the deregistration of the MorphoSys Shares under the U.S. Exchange Act. As a requirement of a Delisting, a delisting tender offer pursuant to Sec. 39 para. 2 BörsG would have to be made to all minority shareholders of MorphoSys. Following the Delisting, MorphoSys Shares would be discontinued from trading on the regulated market |
74
(Regulierter Markt) of the FSE, which could make MorphoSys Shares effectively illiquid. The Delisting would also terminate the comprehensive capital-market oriented reporting obligations of MorphoSys. The termination of the registration under the U.S. Exchange Act would substantially reduce the information required to be furnished by MorphoSys to MorphoSys Securityholders and to the SEC under U.S. disclosure rules and would make certain provisions of the U.S. Exchange Act inapplicable to MorphoSys and the MorphoSys Shares. |
| • | In connection with the Delisting, MorphoSys will terminate the ADS Deposit Agreement, pursuant to which the ADS Depositary maintains an ADS facility for the MorphoSys Shares. If and when the ADS Deposit Agreement is terminated, MorphoSys ADS Holders will only have the right to receive MorphoSys Shares underlying MorphoSys ADSs upon surrender of MorphoSys ADSs and payment of applicable fees of the ADS Depositary. At any time after the date of termination, which shall be at least ninety (90) days following dissemination to MorphoSys ADS Holders of a notice of termination, the ADS Depositary may sell the remaining deposited MorphoSys Shares held under the ADS Deposit Agreement and hold the proceeds of such sale for the benefit of holders of MorphoSys ADSs that have not been surrendered. Whether or not the ADS Deposit Agreement is terminated, if the Offer is consummated, the number of MorphoSys ADSs outstanding is expected to be reduced, which could reduce the liquidity and market value of both the MorphoSys ADSs and their underlying MorphoSys Shares. In addition, the listing of MorphoSys ADSs may be terminated as a result of declined liquidity. |
| • | The Bidder intends to demand a Squeeze-out if it, following completion of the Takeover, holds at least (i) ninety-five percent (95%) (Squeeze-out under stock corporation law), or (ii) ninety percent (90%) (merger Squeeze-out under transformation law) of the share capital with voting power of MorphoSys, and after having implemented all measures necessary for such a Squeeze-out (see Sec. 7.2.4 of this Statement). The Bidder has no intention to pursue a Squeeze-out under takeover law. The implementation of a Squeeze-out of the minority shareholders under stock corporation law would result in a delisting of the MorphoSys Shares on the FSE and – inter alia – on the regulated unofficial market of the stock exchange in Berlin as well as on the unregulated market of the stock exchanges in Düsseldorf, Hamburg, Hanover, Munich and Stuttgart as well as via Tradegate Exchange, the delisting of the MorphoSys Shares and the MorphoSys ADS from Nasdaq, the deregistration of the MorphoSys Shares under the U.S. Exchange Act and the termination of the ADS Deposit Agreement. The Bidder has no intention to pursue a squeeze-out under takeover law. |
| • | If the Bidder were to achieve a shareholding of at least ninety-five percent (95%) in MorphoSys following the completion of the Takeover, securityholders who have not accepted the Offer, are entitled to exercise their put right pursuant to Sec. 39c WpÜG and accept the Offer with their MorphoSys Securities within the put option period pursuant to Sec. 39c WpÜG. This put right is valid for all MorphoSys Securities. The Bidder will publish that it has attained the threshold of ninety-five percent (95%) of the share capital in MorphoSys which is required for a request under Sec. 39a WpÜG, pursuant to Sec. 23 para. 1 sentence 1 no. 4 WpÜG. |
| 9 | STATUS OF REGULATORY APPROVALS |
In Sec. 11.1 of the Offer Document, the Bidder points out that merger control clearances of the Takeover by the relevant authorities in Germany, Austria, and the United States have been obtained. The Bidder does not believe that any additional material pre-merger antitrust filings or clearances are required with respect to the Takeover. The completion of the Offer is therefore no longer contingent on additional merger control clearances and the Offer Conditions therefore do not contain any conditions in this respect. However, to the extent that any additional merger control filings were required pursuant to other applicable foreign antitrust laws, the Bidder would make such filings to the extent possible.
For further details on official approvals and procedures required according to the information provided by the Bidder, please refer to the Bidder’s statements in Sec. 11.1 of the Offer Document.
75
| 10 | TRANSACTION HISTORY AND PAST CONTACTS WITH NOVARTIS AG |
| 10.1 | Transaction History |
The following chronology summarizes the key meetings and events that led to the signing of the Business Combination Agreement. The chronology does not purport to catalogue every interaction of or among
members of the Management Board and the Executive Committee, members of the Supervisory Board, other representatives of MorphoSys, Novartis AG’s representatives and other parties and their representatives.
The Management Board and the Supervisory Board regularly review MorphoSys’ performance, growth prospects and overall strategic direction and evaluate potential opportunities to strengthen MorphoSys’ business and enhance value for its stockholders, in particular with a view on the business, competitive, regulatory, financing and economic environment and developments in the industry in which MorphoSys is active. These reviews have included discussions as to whether MorphoSys should continue to execute on its strategy as a stand-alone company, pursue various partnerships, collaborations, acquisitions or licensing arrangements, seek to improve its capital structure or pursue a sale of the entire company or part of the company. To that end, among other things, MorphoSys has established the Executive Committee which is responsible for, among other things, the development of the strategy and the operational management of MorphoSys. The Executive Committee prepares the decisions for the Management Board`s resolutions and adopts resolutions jointly with the Management Board, provided such resolutions do not fall within the sole responsibility of the Management Board by law or by resolution of the Supervisory Board. The Executive Committee consists of the members of the Management Board and other senior executives from MorphoSys’ core areas of responsibility. Executive Committee members regularly meet with pharmaceutical and other biotechnology companies regarding a variety of potential partnerships, licensing arrangements, joint ventures, collaborations and other strategic transactions, including entering into collaboration and license agreements with Novartis AG for certain MorphoSys programs in 2007, a partnership with Incyte Corporation (“Incyte”) for tafasitamab, a humanized Fc-modified CD19 targeting immunotherapy, which MorphoSys licensed to Incyte in 2020, and the acquisition by MorphoSys of Constellation Pharmaceuticals, Inc., including Constellation Pharmaceuticals, Inc.’s lead asset pelabresib, an investigational selective small molecule BET inhibitor being evaluated for the treatment of myelofibrosis.
In March 2023, in connection with exploratory discussions regarding a potential licensing transaction involving tulmimetostat, an investigational next-generation selective dual inhibitor of EZH2 and EZH1, MorphoSys entered into a confidentiality agreement with a global pharmaceutical company (“Company A”) contemplating the exchange of information related to tulmimetostat and other MorphoSys programs. The confidentiality agreement did not contain a standstill provision, i.e., no obligation of Company A to refrain from acquiring any MorphoSys Shares. Company A had previously engaged in discussions with MorphoSys regarding potential collaborations or licensing opportunities involving Company A and MorphoSys programs, but MorphoSys and Company A had not previously entered into any related transaction.
On 7 June 2023, representatives of Company A met with representatives of MorphoSys, including Dr. Samuel White, Chief of Staff and Vice President of Corporate Strategic Planning (“Dr. White”), at the 2023 BIO International Convention and reiterated Company A’s interest in a potential licensing transaction involving tulmimetostat.
Also on 7 June 2023, representatives of Novartis AG met with representatives of MorphoSys, including Dr. White, at the 2023 BIO International Convention in Boston, United States, in connection with ordinary course business development discussions regarding potential strategic opportunities. During this meeting, representatives of Novartis AG and MorphoSys discussed pelabresib. MorphoSys had previously entered into multiple collaboration and license agreements with Novartis AG for certain MorphoSys programs beginning in 2004, and Novartis AG had engaged in discussions with MorphoSys in connection with
76
evaluating a potential partnership for tafasitamab prior to MorphoSys licensing tafasitamab to Incyte in 2020. Novartis AG did not make a proposal to MorphoSys with respect to any transaction during this meeting.
Beginning on 28 July 2023, MorphoSys shared limited non-public diligence information regarding tulmimetostat with Company A.
On 28 August 2023, Dr. Susanne Kreutz, Novartis AG’s Global Head of Corporate and Business Development (“Dr. Kreutz”), contacted Dr. Barbara Krebs-Pohl, MorphoSys’ Chief Business Officer (“Dr. Krebs-Pohl”), and expressed Novartis AG’s interest in exploring potential strategic opportunities involving pelabresib. Novartis AG did not make a proposal to MorphoSys with respect to any transaction during this interaction.
On 15 September 2023, Dr. Krebs-Pohl spoke with Dr. Aharon Gal, Novartis AG’s Chief Strategy & Growth Officer (“Dr. Gal”), who reiterated Novartis AG’s interest in a potential strategic transaction involving pelabresib. Dr. Gal and Dr. Krebs-Pohl discussed the timing of a potential transaction in relation to the timing of the upcoming announcement of data from MorphoSys’ Phase 3 MANIFEST-2 study investigating pelabresib in combination with the JAK inhibitor ruxolitinib for the treatment of myelofibrosis (the “MANIFEST-2 Study”), which was expected to be announced later that year. Dr. Gal conveyed that Novartis AG would follow up with MorphoSys regarding a potential transaction after the MANIFEST-2 Study data was announced.
On 19 September 2023, in connection with ordinary course business development discussions regarding potential strategic opportunities, a representative of a global pharmaceutical company (“Company B”) contacted Dr. White and expressed an interest in learning more about pelabresib and MorphoSys’ therapeutic programs. Company B had also previously engaged in discussions with MorphoSys in connection with evaluating a potential partnership for tafasitamab prior to MorphoSys licensing tafasitamab to Incyte in 2020.
On 31 October 2023, in connection with further business development discussions regarding potential strategic opportunities, MorphoSys entered into a confidentiality agreement with Company B contemplating the exchange of information regarding MorphoSys’ therapeutic programs. The confidentiality agreement contained a standstill provision which terminated upon entry into the Business Combination Agreement.
On 8 November 2023, representatives of Company A met with representatives of MorphoSys, including Dr. White, at the BIO-Europe 2023 conference in Munich, Germany and expressed continued interest in discussing a potential licensing transaction involving tulmimetostat as well as interest in exploring a potential strategic transaction involving pelabresib, but Company A did not make a proposal to MorphoSys with respect to any transaction at this meeting.
On 13 November 2023, the Supervisory Board held a regularly-scheduled meeting, attended by the Management Board, certain other Executive Committee members, representatives of Centerview and Skadden. From time to time, MorphoSys consulted Centerview regarding strategic advisory matters. Among other matters, the Management Board and the Supervisory Board as well as Executive Committee members discussed strategies for addressing MorphoSys’ long-term capital needs following the anticipated data announcement from the MANIFEST-2 Study, including through an equity financing or other strategic transaction. Representatives of Centerview discussed with the Management Board and the Supervisory Board the current biopharma industry landscape, specifically as it related to oncology, including the possibility that other companies would engage with MorphoSys following announcement of data from the MANIFEST-2 Study.
On 15 November 2023, a representative of Company B met with Dr. White and expressed interest in exploring a potential strategic transaction with MorphoSys. The representative of Company B suggested that Company B’s interest could be broader than just pelabresib, including a transaction involving multiple MorphoSys programs. Following this meeting, Company B submitted a list of diligence questions to MorphoSys regarding MorphoSys’ clinical programs, including tafasitamab and pelabresib.
77
On 20 November 2023, MorphoSys announced positive topline data from the MANIFEST-2 Study and announced that detailed data would be presented at the American Society of Hematology’s 65th Annual Meeting in New Orleans, United States (the “ASH Annual Meeting”).
On 27 November 2023, MorphoSys and Novartis AG entered into a confidentiality agreement contemplating the exchange of information related to MorphoSys’ programs, including pelabresib, in connection with evaluating a potential strategic transaction. The confidentiality agreement did not contain a standstill provision.
On 9 December 2023, representatives of MorphoSys, including Dr. Krebs-Pohl and Dr. White, met with representatives of Novartis AG at the ASH Annual Meeting to discuss pelabresib and the data from the MANIFEST-2 Study to be presented the following day.
On 10 December 2023, MorphoSys presented the positive topline data from the MANIFEST-2 Study at the ASH Annual Meeting.
Later on 10 December 2023, representatives of MorphoSys, including Dr. Krebs-Pohl and Dr. White, met with representatives of Company B at the ASH Annual Meeting to discuss the data from the MANIFEST-2 Study. Representatives of MorphoSys presented non-public materials regarding pelabresib.
On 11 December 2023, representatives of MorphoSys, including Dr. Krebs-Pohl and Dr. White, met with representatives of Company A at the ASH Annual Meeting and discussed potential strategic opportunities. Representatives of Company A again expressed interest in exploring a potential strategic transaction with MorphoSys involving pelabresib but did not make a proposal to MorphoSys with respect to any transaction.
On 12 December 2023, a representative of Company B contacted Dr. White and conveyed that Company B was interested in exploring an acquisition of MorphoSys but that any potential offer would be subject to Company B’s review of diligence information regarding MorphoSys’ clinical programs. Company B did not make a proposal to MorphoSys with respect to any transaction at this meeting.
On 18 December 2023, representatives of MorphoSys, including Dr. White, spoke with representatives of Company B and further discussed a potential acquisition of MorphoSys by Company B. Representatives of Company B reiterated Company B’s interest in reviewing initial diligence information regarding MorphoSys’ clinical programs, and representatives of Company B and MorphoSys discussed the first diligence information that MorphoSys would provide and the timing of Company B’s review. Company B did not make a proposal to MorphoSys with respect to any transaction at this meeting.
On 20 December 2023, the Chief Executive Officer of a global pharmaceutical company (“Company C”) contacted Dr. Jean-Paul Kress, the Chief Executive Officer of MorphoSys (“Dr. Kress”), and informed Dr. Kress that Company C would be making a proposal to acquire MorphoSys with the goal of announcing the takeover prior to 8 January 2024, which was the commencement date for the J.P. Morgan 42nd Annual Healthcare Conference in San Francisco, United States (the “JPM Healthcare Conference”). Following the call, Company C submitted to MorphoSys a written non-binding proposal to acquire all MorphoSys Shares at a price of EUR 50.97 per MorphoSys Share, or USD 14.00 per MorphoSys ADS, in cash (the “20 December Company C Proposal”), subject to Company C’s further diligence. The 20 December Company C Proposal contemplated completing diligence and entering into a transaction agreement within a two (2)-week time period. MorphoSys and Company C had previously collaborated on a certain MorphoSys program, but Company C had not expressed interest in acquiring MorphoSys prior to the 20 December Company C Proposal.
On 21 December 2023, the Supervisory Board held a meeting, attended by the Management Board, certain other Executive Committee members and representatives of Centerview and Skadden, to discuss the 20 December Company C Proposal and other potential strategic opportunities. A representative of Skadden explained legal considerations in connection with the receipt of the 20 December Company C Proposal. A representative of Centerview shared their view of the 20 December Company C Proposal and
78
discussed the possibility that other parties could be interested in a potential transaction based on previous interactions or market position, including Novartis AG, Company B and two other global pharmaceutical companies. The Management Board and the Supervisory Board determined that other parties were unlikely to be in a position to make proposals before 8 January 2024, Company C’s stated timeline for announcing a transaction, and that other parties would be more likely to engage later in January, when the Company expected to be able to share additional internal data from the MANIFEST-2 Study with potentially interested counterparties. The Management Board and the Supervisory Board discussed with Centerview the risk that requesting that other potential counterparties make transaction proposals and execute a definitive agreement on Company C’s suggested timeline could cause otherwise-interested parties to disengage. The Management Board and the Supervisory Board considered options for responding to the 20 December Company C Proposal, including delaying a transaction with Company C until after the upcoming JPM Healthcare Conference to facilitate a larger process with additional participants or using Company C’s desire to announce a transaction prior to the JPM Healthcare Conference to ask Company C for an increase in the offer consideration. The Management Board and the Supervisory Board reasoned that MorphoSys could continue discussions with Company C regarding a near-term acquisition and optimize a proposal while preserving the ability to later engage with additional counterparties if discussions with Company C did result in acceptable transaction terms. The Management Board and the Supervisory Board concluded their discussion by determining that Dr. Kress and representatives of Centerview should message to Company C and Company C’s financial advisor that MorphoSys would be prepared to work with Company C on its desired timeline, with the understanding that Company C would need to increase its offer in order for MorphoSys to consider entering into a transaction prior to the JPM Healthcare Conference.
Following the Supervisory Board meeting on 21 December 2023, Dr. Kress contacted the Chief Executive Officer of Company C and conveyed that MorphoSys was willing to discuss a potential transaction, but that the consideration would have to be higher than the 20 December Company C Proposal for MorphoSys to consider entering into a transaction prior to the JPM Healthcare Conference. Also on 21 December 2023, a representative of Centerview spoke with a representative of Company C’s financial advisor, and, consistent with the decisions and directions of the Management Board and the Supervisory Board, conveyed the same message. The representative of Company C’s financial advisor stated that Company C would provide an updated offer and reiterated that Company C was interested in finalizing and announcing the transaction prior to the JPM Healthcare Conference.
Also on 21 December 2023, MorphoSys began sharing limited non-public diligence information with certain representatives of Company B regarding MorphoSys’ clinical programs and certain corporate matters.
On 22 December 2023, a representative of Company C’s financial advisor communicated to a representative of Centerview a verbal offer for Company C to acquire all MorphoSys Shares with the same upfront consideration payable at closing of EUR 50.97 per MorphoSys Share in cash as contained in the 20 December Company C Proposal, but with an additional earn-out payment equal in value to USD 2 per MorphoSys ADS (corresponding to approximately EUR 7.26 per MorphoSys Share) payable only upon FDA approval of pelabresib (the “22 December Company C Proposal”). The representative of Centerview conveyed to the representative of Company C’s financial advisor that they could bring the 22 December Company C Proposal to MorphoSys, but that, based on prior discussions with MorphoSys, the representative of Centerview believed that Company C would likely have to further increase the consideration offered in order for MorphoSys to consider an acquisition by Company C prior to the JPM Healthcare Conference. In response, the representative of Company C’s financial advisor explained that Company C wished to conduct additional non-public regulatory and clinical diligence prior to making a revised proposal, but that, depending on the outcome of this diligence, Company C had the ability to slightly increase the upfront amount payable at closing offered in the 22 December Company C Proposal and could more significantly increase the amount of the earn-out component offered in the 22 December Company C Proposal. The representative of Centerview offered to discuss with MorphoSys whether
79
MorphoSys would be willing to grant Company C access to one (1) week of regulatory and clinical diligence, with the understanding that following such diligence review, Company C would then make an offer at its best price.
Later on 22 December 2023, Company C’s Chief Executive Officer contacted Dr. Kress and verbally communicated the 22 December Company C Proposal, but emphasized that Company C’s offer would ultimately depend on further diligence and that Company C could potentially increase the amount of the earn-out component of the offer. Company C’s CEO also emphasized that Company C would work quickly towards a final offer to acquire MorphoSys but that timing would ultimately depend on their ability to complete diligence.
Also on 22 December 2023, after receiving the 22 December Company C Proposal, Dr. Krebs-Pohl spoke with Dr. Kreutz about planning a meeting between Dr. Kress and Dr. Vasant Narasimhan, Novartis AG’s Chief Executive Officer (“Dr. Narasimhan”), at the JPM Healthcare Conference to discuss a potential strategic transaction. In response to a question from Dr. Kreutz, Dr. Krebs-Pohl expressed that Novartis AG may miss the opportunity to engage with MorphoSys if Novartis AG waited until the JPM Healthcare Conference to advance discussions regarding a potential strategic transaction. Dr. Kreutz indicated that Novartis AG would be interested in discussing an acquisition of MorphoSys and that Novartis AG could move quickly if MorphoSys was already assessing other opportunities that could be announced at or prior to the JPM Healthcare Conference. In response to a question from Dr. Kreutz, Dr. Krebs-Pohl indicated that MorphoSys expected to receive a draft of a transaction agreement from a third-party in the near future.
Later on 22 December 2023, at the direction of certain Executive Committee members, a representative of Centerview contacted Dr. Kreutz and conveyed that another party had offered to acquire MorphoSys and was prepared to quickly work towards announcing a transaction. Dr. Kreutz reiterated Novartis AG’s interest in pursuing a potential transaction with MorphoSys, as earlier communicated to Dr. Krebs-Pohl, and indicated that Novartis AG could also move quickly to negotiate a transaction and would submit priority diligence questions. Consistent with the determination of the Management Board and the Supervisory Board at the 21 December 2023 Supervisory Board meeting that other parties would be unlikely to be in a position to make proposals before 8 January 2024, and that requesting parties engage on that timeline could cause otherwise-interested parties to disengage, MorphoSys did not, and instructed Centerview to not, inform any other potential counterparty that another party had offered to acquire MorphoSys and was prepared to quickly work towards announcing a transaction.
On 23 December 2023, the Supervisory Board held a meeting, attended by the Management Board, certain other Executive Committee members and representatives of Centerview and Skadden. Executive Committee members and representatives from Centerview updated the Management Board and the Supervisory Board on the discussions with Novartis AG and Company C, including the 22 December Company C Proposal and the discussions the prior day with Novartis AG whereby Novartis AG was informed that MorphoSys was evaluating an acquisition proposal from another third party. After discussion, the Management Board and the Supervisory Board decided to direct Centerview to continue similar competitive messaging and communications with both Novartis AG and Company C. The Management Board and the Supervisory Board reviewed the terms of the 22 December Company C Proposal and determined that, while the 22 December Company C Proposal was likely insufficient, the improved consideration, when compared to the 20 December Company C Proposal, combined with the indicated potential for an additional increase in the consideration based on the conversations from the previous day justified granting Company C access to the requested diligence information subject to entry into a customary confidentiality agreement and an understanding that Company C would submit its best and final proposal by 3 January 2024. Representatives of Centerview then conveyed that, based on the conversations with Novartis AG from the prior day, the representatives of Centerview believed that Novartis AG was preparing to submit initial diligence questions and an offer to acquire MorphoSys on a similar timeline to Company C. The Management Board and the Supervisory Board considered the dynamics of
80
managing interest from both Company C and Novartis AG, including strategies to leverage one party’s interest to increase the consideration offered by the other party, the risk that delaying interactions with Company C in order to further engage with Novartis AG could jeopardize a transaction with Company C and the countervailing risk that requiring Novartis AG to move quickly could jeopardize a transaction with Novartis AG. The Management Board and the Supervisory Board discussed the possibility of soliciting offers from additional parties, including Company B, but decided not to do so at this time because of the perceived low likelihood that any party other than Novartis AG could make a proposal on the accelerated timeline that Company C proposed and in order to manage the risk of discussions around a potential transaction leaking. The Management Board and the Supervisory Board concluded to instruct Executive Committee members and Centerview to proceed with the diligence process, provide Novartis AG and Company C access to a data room containing limited diligence materials and align the timing of the parallel processes with Novartis AG and Company C.
Following the Supervisory Board meeting and at the direction of the Management Board, on 23 December 2023, a representative of Centerview contacted a representative of Company C’s financial advisor to discuss plans for the diligence process and to convey that Company C and MorphoSys would have to enter into a confidentiality agreement appropriate for a whole-company acquisition transaction before Company C could access the diligence materials. That same day, as directed by Executive Committee members, representatives of Centerview provided Company C’s financial advisors with a draft confidentiality agreement, which would be negotiated by representatives of Skadden and Company C’s outside counsel.
On 25 December 2023, Company C provided MorphoSys an initial list of diligence questions.
On 26 December 2023, MorphoSys provided representatives of Novartis AG access to a data room containing limited diligence materials.
On 26 December 2023, representatives of Skadden and Company C’s outside counsel discussed the draft confidentiality agreement between Company C and MorphoSys.
On 27 December 2023, MorphoSys entered into a confidentiality agreement with Company C that contained a standstill provision that terminated upon the entry into the Business Combination Agreement. Following execution of the confidentiality agreement, MorphoSys provided representatives of Company C access to a data room containing limited diligence materials. Throughout the rest of December and until 14 January 2024, representatives of MorphoSys responded to Company C’s initial diligence questions, additional diligence questions and requests for information.
On 28 December 2023, Novartis AG provided MorphoSys an initial list of diligence questions. Throughout the rest of December and until 17 January 2024, representatives of MorphoSys responded to these initial diligence questions, additional diligence questions and requests for information.
On 30 December 2023, at the direction of the Management Board, Centerview communicated to each of Novartis AG and Company C that each party should submit a written non-binding proposal to acquire MorphoSys on 3 January 2024 and outlined expectations for the contents of the proposal, including that the parties include a proposed minimum acceptance threshold condition for a potential takeover offer.
On 3 January 2024, Novartis AG submitted to Centerview a written non-binding offer to acquire all MorphoSys Shares at a price of EUR 55.00 per MorphoSys Share in cash (the “3 January Novartis Proposal”). The 3 January Novartis Proposal indicated that completion of a transaction would be conditional on receiving a minimum acceptance threshold condition of 90%. In the 3 January Novartis Proposal, Novartis AG stated that it expected to be able to enter into a transaction agreement in two (2) weeks.
Also on 3 January 2024, Company C submitted to Centerview a revised written non-binding offer to acquire all MorphoSys Shares at a price of EUR 51.17 per MorphoSys Share, or USD 14.00 per
81
MorphoSys ADS, in cash, plus an earn-out payment of EUR 7.31 per MorphoSys Share, or USD 2.00 per MorphoSys ADS, in cash upon FDA approval of pelabresib (the “3 January Company C Proposal”) The 3 January Company C Proposal did not specify a timeline to signing or propose a minimum acceptance threshold condition.
Also on 3 January 2024, representatives of MorphoSys participated in a diligence call with representatives of Company C and responded to diligence questions relating to regulatory matters. Following this initial diligence call and continuing through 12 January 2024, representatives of MorphoSys participated in additional diligence calls and meetings with representatives of Company C and gave presentations about MorphoSys and its programs to representatives of Company C on topics including tax and scientific matters.
On 4 January 2024, the Supervisory Board held a meeting, attended by the Management Board, certain other Executive Committee members and representatives of Centerview and Skadden. Executive Committee members and representatives of Centerview updated the Management Board and the Supervisory Board on the current status of discussions with Novartis AG and Company C, including the receipt of a proposal from each party. The Management Board and the Supervisory Board reviewed the financial terms of each of the 3 January Novartis Proposal and the 3 January Company C Proposal. Representatives of Skadden next reviewed with the Management Board and the Supervisory Board certain regulatory considerations for a transaction with each of Novartis AG and Company C, including the impact on the certainty and timing of closing of a takeover transaction and strategies for addressing these considerations. Following discussion, the Management Board and the Supervisory Board decided that Executive Committee members and Centerview should continue negotiations with both parties, with an expectation that both parties would need to increase their offers. The Management Board and the Supervisory Board approved that Dr. Kress and Centerview would continue to engage with Novartis AG and Company C and that Centerview should request final offers from Novartis AG and Company C by 15 January 2024.
Later on 4 January 2024, representatives of MorphoSys participated in a diligence call with representatives of Novartis AG and responded to diligence questions relating to intellectual property matters. Following this initial diligence call and continuing through 12 January 2024, representatives of MorphoSys participated in additional diligence calls and meetings with representatives of Novartis AG and gave presentations about MorphoSys and its programs to representatives of Novartis AG on topics including scientific, information technology, labor and employment, corporate legal and compliance matters.
On 5 January 2024, consistent with instruction from the Management Board, Centerview communicated in writing to each of Novartis AG and Company C that each party should submit a “best-and-final” offer by 12:00 hours New York Local Time on 15 January 2024. Centerview informed each party that a draft business combination agreement would be provided by Skadden on 6 January 2024, and outlined a schedule for marking up the agreement with the goal of each party submitting a final draft of the business combination agreement, in the form the party is prepared to sign, on 14 January 2024.
Also on 5 January 2024, a representative of Company B spoke with Dr. White and reiterated Company B’s interest in an acquisition of MorphoSys, but explained that Company B would need to conduct additional diligence before Company B could make an offer. Following this call, MorphoSys granted certain representatives of Company B expanded access to certain additional confidential diligence materials relating to the pelabresib program. MorphoSys offered to expand access to the diligence materials to additional representatives of Company B, but Company B declined.
On 6 January 2024, Skadden sent a draft business combination agreement to each of Freshfields Bruckhaus Deringer LLP, Novartis AG’s outside counsel (“Freshfields”), and Company C’s outside counsel. The draft business combination agreement contemplated, among other things, a potential voluntary public takeover offer pursuant to German law and did not include a specific proposal on a minimum acceptance threshold condition.
82
On 8 January 2024, Dr. Kress, Dr. White and other representatives of MorphoSys met with Dr. Narasimhan, Dr. Gal and other representatives of Novartis AG at the JPM Healthcare Conference to discuss pelabresib and the MANIFEST-2 data and to provide an overview of MorphoSys’ operations.
On 9 January 2024, representatives of MorphoSys, including Dr. White, met with representatives of Company A at the JPM Healthcare Conference to further discuss the pelabresib program, including exploring a potential strategic transaction with MorphoSys involving pelabresib. Dr. White and a representative of Company A agreed to meet again at the JPM Healthcare Conference to discuss further.
Also on 9 January 2024, Dr. Kress, Dr. White and other representatives of MorphoSys met with representatives of Company B to present information about the pelabresib program. Representatives of MorphoSys also responded to questions from representatives of Company B regarding pelabresib and tafasitamab.
Later on 9 January 2024, Dr. Kress met with a representative of Company B at the JPM Healthcare Conference to discuss a potential acquisition of MorphoSys. The representative of Company B conveyed Company B’s continued interest in making an offer to acquire MorphoSys, but explained that Company B was still very early in the diligence process and would need multiple additional weeks to review before Company B would be in a position to make an offer to MorphoSys.
Also on 9 January 2024, each of Company C’s outside counsel and Freshfields returned a revised draft of the business combination agreement to Skadden, following which, discussions and negotiations between representatives of Skadden and representatives of Company C’s outside counsel and Freshfields continued through 15 January 2024.
On 10 January 2024, Skadden sent an initial draft of the confidential disclosure letter in relation to the representations and warranties as well as certain other provisions contained in the business combination agreement that would accompany the business combination agreement to each of Company C’s outside counsel and Freshfields.
On 11 January 2024, Skadden spoke with each of Company C’s outside counsel and Freshfields regarding their 9 January 2024 revised drafts of the business combination agreement. Representatives from Skadden provided each outside counsel with MorphoSys’ and Skadden’s feedback on the respective drafts which would be addressed by Skadden’s next draft. Later that day, Skadden and Freshfields had a call to discuss initial perspectives on SEC exemptive relief which would need to be obtained to address differences between German and U.S. legal requirements governing a potential transaction.
Also on 11 January 2024, as discussed on 9 January, Dr. White met with a representative of Company A at the JPM Healthcare Conference to further engage in exploratory discussions regarding a potential strategic transaction with MorphoSys involving pelabresib. Dr. White expressed MorphoSys’ expectation that a transaction involving both pelabresib and tulmimetostat would need to be structured as an acquisition of all MorphoSys Shares. The representative of Company A conveyed that Company A would discuss further internally and follow up with MorphoSys.
On 12 January 2024, representatives of Novartis AG conducted a virtual site visit to MorphoSys’ Planegg, Germany site to review quality assurance materials relating to clinical operations, technical operations, operational excellence, regulatory compliance, biostatistics, clinical development and patient safety.
Also on 12 January 2024, Dr. Gal contacted a representative of Centerview and suggested that, as an alternative to purchasing all MorphoSys Shares, Novartis AG would be interested in solely purchasing MorphoSys’ pelabresib program, structured as an acquisition of MorphoSys’ wholly-owned subsidiary Constellation Pharmaceuticals, Inc. Dr. Gal indicated that Novartis AG could potentially offer a higher price for the pelabresib program than for MorphoSys as a whole. Following the call with Dr. Gal, a representative of Centerview informed Dr. Kress of the alternative proposal and discussed with Dr. Kress the potential challenges and risks around pursuing such a proposal, including the risk that such a transaction could not be negotiated on the timeline currently contemplated and thus would interfere with
83
the ability to do a transaction with Company C, that selling pelabresib while retaining tafasitamab and MorphoSys’ other earlier stage pipeline programs may not be in MorphoSys’ best interests and that structuring a transaction as an asset sale rather than a whole-company acquisition could be less favorable for MorphoSys by potentially introducing negative tax and employment-related consequences. Following this discussion, at the direction of Dr. Kress, the representative of Centerview informed Dr. Gal that MorphoSys expected Novartis AG to submit an offer to purchase all MorphoSys Shares on 15 January 2024 consistent with the communications and instructions provided on 5 January 2024.
Also on 12 January 2024, Skadden separately returned revised drafts of the business combination agreement to Freshfields and Company C’s outside counsel. Key issues under negotiation were the closing conditions, including conditions related to the number of MorphoSys Shares tendered and closing conditions tied to MorphoSys’ market performance, offer support by MorphoSys, post-closing financing support of MorphoSys, interim conduct of business covenants which would restrict MorphoSys’ business operations between signing and closing and post-closing measures like delisting, implementation of a domination and profit and loss transfer agreement and treatment of employee equity awards, including whether the buyer would be required to pay the full value of equity awards in excess of statutory caps on compensation.
On 13 January 2024, MorphoSys and Centerview executed an engagement letter.
On the evening of 13 January 2024, Freshfields sent a revised draft of the business combination agreement to Skadden.
On the morning of 14 January 2024, Skadden and Freshfields discussed the revised draft business combination agreement.
Also on the morning of 14 January 2024, Company C’s counsel sent a revised draft of the business combination agreement to Skadden. Skadden and Company C’s counsel discussed the draft business combination agreement on the afternoon of 14 January 2024. Compared to the draft business combination agreement submitted by Novartis AG, the Company C draft business combination agreement contained a larger number of open items, including regarding closing certainty, the treatment of employee compensation plans and post-closing financial support.
On 14 January 2024, Dr. Gal informed a representative of Centerview that, following Novartis AG’s review of the confidential diligence materials provided by MorphoSys, Novartis AG was prepared to submit a proposal to acquire MorphoSys for a price of USD 75 per MorphoSys Share (which, applying then-current exchange rates, equated to approximately EUR 68 per MorphoSys Share). Dr. Gal also conveyed that Novartis AG’s willingness to enter into a transaction would be conditioned upon the elimination of certain existing contractual obligations of MorphoSys.
Later on 14 January 2024, Dr. Gal and Dr. Kreutz contacted a representative of Centerview and indicated that Novartis AG was evaluating certain contracts between MorphoSys and third parties which were provided in connection with diligence, and as a result might not be in a position to finalize a transaction on the timeline previously set forth by MorphoSys. Subsequently, the representative of Centerview informed the Management Board of both of the 14 January 2024 conversations with Novartis AG and discussed how to respond.
In light of the comments made by representatives of Novartis AG on 14 January 2024, on the morning of 15 January 2024, at the direction of the Management Board, a representative of Centerview contacted Dr. Gal and explained that, consistent with the expectations previously communicated, MorphoSys expected Novartis AG to make their “best and final” offer later that day.
Later on the morning of 15 January 2024, Company C’s Chief Executive Officer informed a representative of Centerview that Company C would not deliver a final offer and that Company C was withdrawing from the process, citing that it was not in a position to submit a competitive final offer. Company C’s Chief Executive Officer also contacted Dr. Kress and conveyed the same message. Shortly thereafter, MorphoSys terminated Company C’s access to the virtual data room containing confidential diligence materials.
84
Later on 15 January 2024, Novartis AG submitted a binding offer to acquire all MorphoSys Shares at a price of EUR 68.00 per MorphoSys Share in cash (the “15 January Novartis Offer”). The 15 January Novartis Offer contemplated that Freshfields and Skadden would cooperate to finalize the business combination agreement, the most recent draft of which contained a small number of open items that Skadden and Freshfields had been negotiating. MorphoSys’ ability to accept the 15 January Novartis Offer and enter into a business combination agreement with Novartis AG was, however, conditioned on the satisfaction of multiple specific conditions (the “15 January Novartis Offer Conditions”), including conditions which related to the elimination of certain existing contractual obligations of MorphoSys included in its collaboration and license agreement relating to tafasitamab. The 15 January Novartis Offer would remain open for sixty (60) calendar days from 15 January 2024, unless extended by Novartis AG.
Following receipt of the 15 January Novartis Offer, on 15 January 2024, the Supervisory Board held a meeting, attended by the Management Board, certain other Executive Committee members and representatives of Centerview and Skadden. Representatives of Centerview and Executive Committee members informed the Management Board and the Supervisory Board that Company C had declined to submit a final offer and dropped out of the process. The Management Board and representatives of Centerview informed the Supervisory Board that Novartis AG had submitted the 15 January Novartis Offer and that the business combination agreement was almost fully agreed upon with Novartis AG, except for resolution of a small number of remaining open items. A representative of Skadden informed the Management Board and the Supervisory Board that although Novartis AG described the 15 January Novartis Offer as a “binding offer”, certain revisions would be required to make it legally binding and strengthen its enforceability. A representative of Skadden described each of the 15 January Novartis Offer Conditions and discussed how each condition could be satisfied in the allotted time before the offer expired. The Management Board and the Supervisory Board discussed strategies for satisfying the 15 January Novartis Offer Conditions, including the possibility of reaching out to Incyte to explore its interest in acquiring all of MorphoSys’ rights to the tafasitamab program as a method of satisfying the 15 January Novartis Offer Conditions relating to tafasitamab. A representative of Centerview and the Management Board offered their perspectives that, based on the conversations with Novartis AG on 14 January 2024, the sale of MorphoSys’ rights to the tafasitamab program prior to or simultaneous with a transaction with Novartis AG would not affect the price per MorphoSys Share in the 15 January Novartis Offer. Representatives of Skadden discussed the importance of confidentiality for enabling the actions necessary to satisfy the conditions to the 15 January Novartis Offer and noted that while the 15 January Novartis Offer was irrevocable by Novartis AG for its duration, MorphoSys was not required to satisfy the conditions nor was it obligated to accept the offer. Following discussion of the available options, the Management Board and the Supervisory Board decided that MorphoSys should continue to engage with Novartis AG to secure a legally binding offer to which an agreed form of the business combination agreement would be attached and acceptance of which by MorphoSys, and entry into the attached business combination agreement by Novartis AG and MorphoSys, would be at MorphoSys’ discretion and conditioned only on the 15 January Novartis Offer Conditions. The Management Board and the Supervisory Board also decided that Dr. Kress and Centerview should contact Incyte to explore the potential sale of MorphoSys’ rights to the tafasitamab program. Further, the Management Board and the Supervisory Board decided that Centerview should contact Company B to further explore Company B’s interest in an acquisition of MorphoSys as an alternative to a transaction with Novartis AG.
Later on 15 January 2024, as discussed at the Supervisory Board meeting earlier that day, Dr. Kress contacted Hervé Hoppenot, Incyte’s Chief Executive Officer (“Mr. Hoppenot”), about the potential sale to Incyte of MorphoSys’ rights to the tafasitamab program, which is also referred to herein as “Monjuvi”, the brand name under which the products that are produced under the tafasitamab program are being sold (the “Tafasitamab Sale”). Mr. Hoppenot expressed interest in discussing a potential Tafasitamab Sale. As directed by the Management Board and the Supervisory Board, a representative of Centerview later contacted Dr. Pablo Cagnoni, Incyte’s President, to further discuss a potential Tafasitamab Sale. Following these discussions, Incyte’s outside legal counsel and Skadden began negotiating a purchase
85
agreement for the Tafasitamab Sale (the “Incyte Purchase Agreement”), which the parties would negotiate until 5 February 2024.
Also on 15 January 2024, as discussed at the Supervisory Board meeting earlier that day, representatives of Centerview contacted Company B to gauge Company B’s continued interest in a potential acquisition of MorphoSys. Representatives of Company B expressed continued interest in an acquisition of MorphoSys and conveyed that Company B would continue to review diligence materials.
Following the Supervisory Board meeting on 15 January 2024, representatives of Skadden contacted representatives of Freshfields and proposed revisions to the 15 January Novartis Offer, including revisions to the 15 January Novartis Offer Conditions. Subsequently, representatives of Novartis AG requested that MorphoSys enter into an agreement with Novartis AG at the time the binding offer was made by Novartis AG to MorphoSys that would prohibit MorphoSys from soliciting offers from third parties to acquire MorphoSys (the “Exclusivity Agreement”). Members of the Management Board discussed Novartis AG’s request that MorphoSys enter into the Exclusivity Agreement and determined that Skadden should continue engaging with Freshfields to obtain a binding offer from Novartis AG, including by preparing an acceptable Exclusivity Agreement. Later on 15 January 2024, at the direction of members of the Management Board, Skadden provided Freshfields with an initial draft of the Exclusivity Agreement and conveyed that MorphoSys would enter into the Exclusivity Agreement if Novartis AG provided a binding offer letter revised to MorphoSys’ satisfaction.
Later on 16 January 2024, Freshfields returned a revised binding offer letter and a revised draft of the Exclusivity Agreement. Among the proposed changes requested by Novartis AG, the Exclusivity Agreement included more detailed “no shop” provisions which were intended to mirror the agreed-upon and negotiated provisions in the draft business combination agreement, including that MorphoSys could not solicit competing offers but that the Management Board could, consistent with its fiduciary duties, consider superior offers by third parties, and conditioned acceptance of the offer on MorphoSys’ adherence to the interim operating covenants set forth in the draft business combination agreement as if then applicable. Discussions and negotiations among representatives of Skadden and Freshfields regarding the revised binding offer, the form of a business combination agreement to be attached thereto and the Exclusivity Agreement continued until the documents were in agreed form on 18 January 2024. During this time period, representatives of Novartis AG and MorphoSys began discussing how MorphoSys would satisfy the 15 January Novartis Offer Conditions.
On 18 January 2024, Dr. White and other representatives of MorphoSys met with representatives from Company B to discuss regulatory questions related to pelabresib.
Later on 18 January 2024, Novartis AG and MorphoSys agreed on the form of a binding offer, the form of the business combination agreement to be attached thereto and the Exclusivity Agreement. Subsequently, Novartis AG submitted to MorphoSys a binding offer based on the agreed form to acquire all MorphoSys Shares at a price of EUR 68.00 per MorphoSys Share in cash (the “Final Letter of Intent”) that superseded and replaced the 15 January Novartis Offer. Attached to the Final Letter of Intent was an agreed form of the business combination agreement and accompanying disclosure letter (the “Final Letter of Intent BCA”). Acceptance by MorphoSys of the Final Letter of Intent and entry into the Final Letter of Intent BCA by Novartis AG and MorphoSys was conditioned on the satisfaction of multiple specific conditions, including a condition that would be satisfied by consummation of the Tafasitamab Sale (the “Final Letter of Intent Conditions”). The Final Letter of Intent would remain open until 23:59 New York Local Time on 17 March 2024. Novartis AG could revoke and withdraw the Final Letter of Intent only under certain circumstances, including if MorphoSys breached its obligations under the Exclusivity Agreement or if MorphoSys did not comply with the interim operating covenants in the Binding Offer BCA.
Following the receipt of the Final Letter of Intent, later that day on 18 January 2024, MorphoSys and Novartis AG executed the Exclusivity Agreement. Pursuant to the Exclusivity Agreement, from the time of execution until 23:59 Central European time on 17 March 2024, MorphoSys agreed to comply with the “no shop” provisions in the Final Letter of Intent BCA, including that MorphoSys could not solicit
86
competing offers but that the Management Board could, consistent with its fiduciary duties, consider superior offers by third parties. For further details on the “no shop” provisions in the Business Combination Agreement, please see Sec. 8.2.13 of the Offer Document. In connection with entry into the Exclusivity Agreement, MorphoSys terminated Company B’s access to the confidential diligence materials in the virtual data room and terminated Company A’s access to the limited non-public diligence information regarding tulmimetostat.
Between 18 January 2024 and 5 February 2024, MorphoSys, with input from Novartis AG, worked on satisfying the Final Letter of Intent Conditions. During this time, representatives of Novartis AG and MorphoSys discussed the potential waiver of one of the Final Letter of Intent Conditions. In exchange for the waiver of such condition, Novartis AG requested modifications to the Final Letter of Intent BCA, including expanded access covenants and consent rights related to the proposed Tafasitamab Sale. Novartis AG and MorphoSys agreed on the changes in principle, and during this time, Skadden and Freshfields discussed Novartis AG providing an amended offer letter to reflect the proposed changes to Final Letter of Intent BCA and to reflect the waiver of such condition.
On 19 January 2024, Dr. Kress spoke with Dr. Gal and Dr. Kreutz regarding progress on a potential Tafasitamab Sale and the expected timing for entry into the Final Letter of Intent BCA.
On 22 January 2024, the Supervisory Board held a meeting, attended by the Management Board, another Executive Committee member and representatives of Centerview and Skadden, during which Dr. Kress provided the Supervisory Board with an update on the status of a potential transaction with Novartis AG and the Tafasitamab Sale. A representative of Skadden emphasized certain details of the Final Letter of Intent and the Exclusivity Agreement, including that MorphoSys could not solicit competing offers but that the Management Board could, consistent with its fiduciary duties, consider superior offers by third parties. The representative of Skadden explained that Novartis AG could terminate the Final Letter of Intent if MorphoSys did not comply with the interim operating covenants in the Final Letter of Intent BCA. The representative of Skadden provided an update on actions necessary to satisfy the conditions to the Final Letter of Intent. A representative of Skadden summarized the terms of the Final Letter of Intent BCA and the expected transaction timeline if MorphoSys chose to execute the Final Letter of Intent BCA.
On 26 January 2024, a blog post speculated that MorphoSys was engaged in discussions with three large pharmaceutical companies regarding a potential sale without providing any further details. MorphoSys concluded that the blog post referred solely to ongoing general market speculations about MorphoSys being a potential takeover target without any knowledge of MorphoSys’ discussions with Company C and Novartis AG.
Later on 26 January 2024, Dr. Kress and Dr. White contacted Dr. Gal and Dr. Kreutz to emphasize the importance of Novartis AG and MorphoSys working together to quickly to satisfy the remaining Final Letter of Intent Conditions.
On 28 January 2024, representatives of MorphoSys communicated to Novartis AG and other counterparties involved in the satisfaction of the Final Letter of Intent Conditions that MorphoSys was targeting a signing date of 5 February 2024.
During the week of 29 January 2024, representatives of Skadden discussed with representatives of Freshfields the importance of Novartis AG providing an amended offer letter reflecting the proposed changes to the Final Letter of Intent BCA. During this week, representatives of MorphoSys and Skadden continued to keep representatives of Novartis AG and Freshfields apprised of its efforts to satisfy the Final Letter of Intent Conditions and that MorphoSys expected to satisfy all of the Final Letter of Intent Conditions, other than the Final Letter of Intent Condition that MorphoSys expected Novartis AG to waive, by 5 February 2024.
On 31 January 2024, a representative of Company A contacted Dr. White and conveyed that, while Company A was still interested in a potential collaboration involving tulmimetostat, Company A had determined that it was not in a position to pursue a whole-company acquisition at this time and that Company A would not be making a proposal for any strategic transaction.
87
On 5 February 2024, Reuters contacted a representative of MorphoSys and requested comments on a planned article reporting that Novartis AG was in advanced discussions to purchase MorphoSys and was planning to announce a transaction soon (the “Reuters Article”). A representative of MorphoSys informed a representative of Novartis AG of Reuters’ request for comment, and Novartis AG subsequently expressed a desire to immediately finalize the Business Combination Agreement and the Tafasitamab Sale. Given timing constraints, Novartis AG and MorphoSys agreed not to amend the Final Letter of Intent to reflect the removal of the specified Final Letter of Intent Condition (but that Novartis AG would not insist on its satisfaction) and to revise only the Final Letter of Intent BCA. Skadden and Freshfields discussed and agreed on certain modifications to the Final Letter of Intent BCA to account for details of the Tafasitamab Sale and a limited number of additional changes. At the same time, MorphoSys decided to accelerate a joint meeting of the Management Board and the Supervisory Board originally scheduled for later the same day to decide on the Business Combination Agreement, the Incyte Purchase Agreement and the Takeover.
Following these discussions, Novartis AG provided MorphoSys with a proposed final Business Combination Agreement reflecting the changes previously discussed and agreed between the parties, and Novartis AG confirmed that Novartis AG would enter into the Business Combination Agreement immediately upon the execution of the Incyte Purchase Agreement, which would satisfy the Final Letter of Intent Conditions (other than the Final Letter of Intent Condition that Novartis AG had agreed would be waived).
Immediately thereafter, on 5 February 2024, the Management Board and the Supervisory Board held the accelerated joint meeting, attended by representatives of Centerview and Skadden. During the meeting, representatives of Centerview rendered to the Management Board and the Supervisory Board an oral opinion, which was subsequently confirmed by delivery of the Signing Opinion that, as of such date and based upon and subject to various assumptions made, procedures followed, matters considered and qualifications and limitations upon the review undertaken in preparing its opinion, the Offer Price to be paid to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Signing Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Business Combination Agreement by means of the Offer is fair, from a financial point of view, to such MorphoSys Securityholders. Centerview was not asked to consider, nor did Centerview express any view on, and Centerview’s opinion did not address, any term or aspect of the Incyte Purchase Agreement, pursuant to which certain assets of MorphoSys and MorphoSys US Inc. were sold immediately prior to the execution of the Business Combination Agreement, or how the Incyte Purchase Agreement may impact MorphoSys, the Business Combination Agreement or the Takeover. For further details on Centerview’s analysis, please see Sec. 6.2 of this Statement. During the meeting, representatives of Skadden then summarized the changes to the Business Combination Agreement from the Final Letter of Intent BCA and summarized the material terms of the Incyte Purchase Agreement and related documents. The Management Board and the Supervisory Board also discussed the Incyte Purchase Agreement, considered its benefits, including the purchase price and the additional resources that could be reallocated to the development and commercialization of MorphoSys’ other programs, and its risks, including that MorphoSys could sell all of its rights to tafasitamab without the Offer being consummated, and determined that entering into the Incyte Purchase Agreement would be in MorphoSys’ best interests even if the Offer was not consummated. After further discussion, including a discussion regarding various factors described under Sec. 6 of this Statement, the Management Board and the Supervisory Board each unanimously (i) determined that MorphoSys’ entry into the Incyte Purchase Agreement and the Business Combination Agreement and the expected Offer are fair to, and in the best interests of, MorphoSys, including the MorphoSys Securityholders and other stakeholders, (ii) declared it advisable for MorphoSys to enter into the Incyte Purchase Agreement and the Business Combination Agreement, (iii) approved the execution, delivery and performance of the Incyte Purchase Agreement and the Business Combination Agreement and supported the consummation of the Offer, and (iv) subject to review of the offer document published by Novartis AG and provided that the offer document is in accordance with the terms of the Business Combination Agreement, agreed to issue a joint reasoned statement recommending that
88
MorphoSys Securityholders tender their MorphoSys Shares into the Offer and that MorphoSys ADS Holders take the requisite actions to tender the MorphoSys Shares underlying the MorphoSys ADSs into the Offer.
During the joint meeting of the Management Board and the Supervisory Board, Reuters published the Reuters Article.
Following the joint meeting of the Management Board and the Supervisory Board, MorphoSys executed the Incyte Purchase Agreement and ancillary documents with Incyte and MorphoSys US Inc. Immediately after execution of the Incyte Purchase Agreement, a representative of Skadden confirmed to Freshfields that all of the Final Letter of Intent Conditions, other than the Final Letter of Intent Condition that Novartis AG agreed to waive, had been satisfied and MorphoSys executed the revised Business Combination Agreement with Novartis AG and the Bidder.
Immediately after the execution of the Incyte Purchase Agreement and the Business Combination Agreement, the Bidder and Novartis AG published the decision to launch the Offer in accordance with Sec. 10 para. 1 sentence 1, para. 3 WpÜG in conjunction with Secs. 29, 34 WpÜG, and MorphoSys issued an ad hoc announcement and a press release announcing the execution of the Business Combination Agreement and the Incyte Purchase Agreement.
On 10 April 2024, the Management Board and the Supervisory Board held a joint meeting, attended by representatives of Centerview and Skadden. During the meeting, representatives of Centerview rendered to the Management Board and the Supervisory Board an oral opinion, which was subsequently confirmed by delivery of the Offer Opinion that, as of such date and based upon and subject to various assumptions made, procedures followed, matters considered and qualifications and limitations upon the review undertaken in preparing its opinion, the Offer Price to be paid to the holders of MorphoSys Shares (other than (i) MorphoSys Shares which were, as of the date of the Offer Opinion, held by MorphoSys as treasury shares, and (ii) MorphoSys ADSs) pursuant to the Offer Document by means of the Offer is fair, from a financial point of view, to such stockholders. Centerview was not asked to consider, nor did Centerview express any view on, and Centerview’s opinion did not address, any term or aspect of the Incyte Purchase Agreement, pursuant to which certain assets of MorphoSys and MorphoSys US Inc. were sold immediately prior to the execution of the Business Combination Agreement, or how the Incyte Purchase Agreement may impact MorphoSys, the Offer Document, the Business Combination Agreement or the Takeover. The published Offer Document does not contain any material changes to the draft version of the Offer Document available on 10 April 2024. For a detailed discussion of the Offer Opinion, please see Sec. 6.2 of this Statement.
On 11 April 2024, the Bidder commenced the Offer. Promptly thereafter, the Management Board and the Supervisory Board resolved to unanimously support the Offer, recommend to all MorphoSys Securityholders to accept the Offer and tender their MorphoSys Securities under the Offer (see Sec. 15 of this Statement).
| 10.2 | Past Contacts, Transactions, Negotiations and Agreements with Novartis AG |
Except as set forth in this Statement, or as otherwise incorporated by reference herein, as of the date of publication of this Statement, there are no material agreements, arrangements or understandings, nor any actual or potential conflicts of interest, between (i) MorphoSys or any MorphoSys Affiliate on the one hand, and (ii) on the other hand (x) any of their respective Management Board members, Supervisory Board members, senior management or MorphoSys Affiliates or (y) Novartis AG or any of its executive officers, directors or one of its Affiliates.
No transactions with respect to MorphoSys Shares have been effected by MorphoSys or, to its knowledge after making reasonable inquiry, by any of the members of the Supervisory Board or Management Board or MorphoSys’ senior management, MorphoSys Affiliates or subsidiaries of MorphoSys during the sixty (60) days prior to the date of publication of this Statement, other than as described in this Statement.
89
| 10.2.1 | Business Combination Agreement |
On 5 February 2024, MorphoSys, the Bidder and Novartis AG entered into the Business Combination Agreement, outlining the key terms and the parties’ mutual understanding of the Takeover.
Further details of the Business Combination Agreement are provided in:
| • | Sec. 5.5 of this Statement, which contains a summary of the material terms and conditions of the Offer, including the Offer Price, the Acceptance Period and the Additional Acceptance Period and the Offer Conditions; |
| • | Sec. 6.1.3 of this Statement, which contains a summary of the fiduciary out for MorphoSys and the MorphoSys Termination Fee as well as commitments relating to obtaining the relevant antitrust clearance (including with respect to the Novartis Termination Fee); |
| • | Sec. 7.1 of this Statement, which contains a description of the objectives and intentions of the Bidder and Novartis AG; and |
| • | Sec. 8.2 of the Offer Document, which includes a description of the non-solicitation undertaking of MorphoSys in Sec. 8.2.13 and a description of the termination rights as well as the MorphoSys Termination Fee and the Novartis Termination Fee in Secs. 8.2.15 to 8.2.17. |
The above-mentioned summaries of material terms of the Business Combination Agreement do not purport to be complete and are subject to the individual agreements made in the Business Combination Agreement, which is incorporated herein by reference, and was filed as Exhibit 99.3 to a Form 6-K filed by MorphoSys with the SEC on 7 February 2024.
| 10.2.2 | Confidentiality Agreement |
On 27 November 2023, MorphoSys entered into a confidentiality and non-disclosure agreement with Novartis AG (the “Confidentiality Agreement”), which is included as Exhibit (e)(3) to the Schedule 14D-9 and is incorporated herein by reference. The Confidentiality Agreement has since then formed the basis among the parties to the Confidentiality Agreement, their affiliates and advisors for the exchange of information relating to MorphoSys and Novartis AG, including their affiliates and business activities.
| 11 | PERSONS OR ASSETS RETAINED, EMPLOYED, COMPENSATED OR USED |
In addition to the below persons who may be deemed to be directly or indirectly employed, retained or compensated to make solicitations or recommendations in connection with the Offer, solicitations or recommendations in connection with the Offer may be made by members of the Management Board, the Supervisory Board, the Executive Committee or employees of MorphoSys, for which services no additional compensation will be paid.
| 11.1 | Centerview |
MorphoSys engaged Centerview as financial advisor in connection with the Takeover and entered with Centerview into an engagement letter dated 13 January 2024 (the “Centerview Engagement Letter”). The Centerview Opinions rendered to the Management Board and Supervisory Board do not constitute a recommendation to any MorphoSys Securityholders as to whether or not such holder should tender MorphoSys Shares in connection with the Offer or how such shareholder or any other person should act with respect to the Takeover or any other matter.
As further described in Sec. 6.2 of this Statement, based on the terms of the Centerview Engagement Letter, Centerview will receive a customary (i) fee for rendering the Centerview Opinions and (ii) transaction fee, in case of clause (ii), subject to and payable upon completion of the Takeover. In
90
addition, MorphoSys has agreed to reimburse Centerview for reasonable expenses incurred in connection with its services and to indemnify Centerview against certain liabilities which may arise out of Centerview’s engagement.
| 11.2 | Innisfree |
MorphoSys engaged Innisfree M&A Incorporated to provide consulting, analytic and further services with regard to the MorphoSys Shareholder base in connection with the Offer, and Innisfree M&A Incorporated may make solicitations or recommendations to MorphoSys Securityholders in connection with the Offer. MorphoSys has agreed to pay customary compensation for such services. In addition, MorphoSys has agreed to reimburse Innisfree for its out-of-pocket expenses and to indemnify it and certain related persons against certain liabilities which may relate to or arise out of the engagement.
| 12 | PURPOSES OF THE TRANSACTION AND PLANS OR PROPOSALS |
Except as described in this Statement, MorphoSys is not undertaking or engaged in any negotiations in response to the Offer that relate to (i) a tender offer or other acquisition of MorphoSys Securities, any of its subsidiaries or any other person, (ii) any extraordinary transaction, such as a merger, reorganization or liquidation, involving MorphoSys or any of its subsidiaries, (iii) any purchase, sale or transfer of a material amount of the assets of MorphoSys or any of its subsidiaries, or (iv) any material change in the present dividend rate or policy, or indebtedness or capitalization of MorphoSys.
Except as otherwise described in this Statement, there are also no transactions, board resolutions, agreements in principle or signed contracts that were entered into in response to the Offer that relate to one or more of the matters referred to in the preceding paragraph.
| 13 | INTERESTS OF THE MEMBERS OF THE MANAGEMENT BOARD AND SUPERVISORY BOARD |
| 13.1 | Specific Interests of the Members of the Management Board and Supervisory Board |
It cannot be ruled out that the Management Board and Supervisory Board members have interests in the Business Combination Agreement and the transactions contemplated thereby, including the Offer, that are different from or in addition to those of MorphoSys, the MorphoSys Securityholders and other stakeholders of MorphoSys. The Management Board and Supervisory Board members have such interests and disclosed and considered them, among other matters, in evaluating the Business Combination Agreement and the Offer and in reaching their respective decision to (i) approve the Business Combination Agreement and the transactions contemplated thereby, and (ii) recommend to the MorphoSys Securityholders the acceptance of the Offer. According to the Business Combination Agreement, the members of the Management Board and the Supervisory Board, to the extent they hold MorphoSys Securities and to the extent permitted by law, intend to tender these MorphoSys Securities in the Offer.
| 13.1.1 | Modification of Remuneration System and Incentive Plans |
The Business Combination Agreement provides that, promptly after the Closing and the Delisting, Novartis AG and the Bidder will procure and take, subject to applicable law, all actions that are required or desirable for the cancellation of (i) the existing remuneration system for the Management Board, (ii) any caps provided for in the terms and conditions of the Incentive Plans for Management Board members which apply to the payouts under such Incentive Plans, and (iii) any caps for payment under these programs provided for in the Incentive Plans for MorphoSys Group employees (together, the “Remuneration Modifications”). MorphoSys will also use its best efforts, subject to applicable law, to defer the settlement of any Incentive Plans (subject to the different treatment for the 2024 Incentive Plans described in Sec. 8.2.10 of the Offer Document) to a point in time shortly after both (i) the Delisting has occurred, and (ii) the Remuneration Modifications have been implemented (the “Settlement Deferral”).
91
Novartis AG and the Bidder will procure and take, subject to applicable law, all actions that are required or desirable to ensure that, consistent with past practice and subject to applicable law, all Management Board members or MorphoSys Group employees who leave the MorphoSys Group following completion of the Offer are compensated for any outstanding remuneration components pursuant to the Incentive Plans with respect to both the vesting and the acceleration of such remuneration components under the Incentive Plans, including such granted under the 2024 Incentive Plans.
| 13.1.2 | Settlement of Incentive Plans |
The Business Combination Agreement provides that, following the earlier of (i) the date on which the Remuneration Modifications and the Settlement Deferral have occurred, and, to the extent legally permissible, and (ii) eleven (11) months following the Closing, Novartis AG and the Bidder will procure and take all actions required or desirable to ensure that, subject to applicable law, MorphoSys settles all of the Incentive Plans under which Stock Options, Restricted Stock Units or Performance Share Units (collectively, the “MorphoSys Incentive Awards”) remain outstanding as of the Closing (other than MorphoSys Incentive Awards granted under the 2024 Incentive Plans) as follows:
| (i) | Each Stock Option that is outstanding as of the Closing, whether or not vested, and that has a per-share exercise price less than the Offer Price shall be settled in exchange for a cash consideration per Stock Option (subject to any applicable withholding) equal to the excess of the Offer Price over the applicable exercise price of such Stock Option (whereby the number of Stock Options of each beneficiary to be settled shall be determined at 100% of performance). Each Stock Option that has an exercise price equal to or greater than the Offer Price forfeits with no consideration. |
| (ii) | Each Performance Share Unit that is outstanding as of the Closing, whether or not vested, shall be settled for cash consideration per Performance Share Unit (subject to any applicable withholding) equal to the Offer Price (whereby the number Performance Share Units of each beneficiary to be settled shall be determined at 100% of performance). |
| (iii) | Each Restricted Stock Unit that is outstanding as of the Closing, whether or not vested, shall be settled for cash consideration per Restricted Stock Unit (subject to any applicable withholding) equal to the Offer Price (whereby the number of Restricted Stock Units of each beneficiary to be settled shall be determined at 100% of performance). |
The Business Combination Agreement provides that each MorphoSys Incentive Award outstanding as of the Closing granted under the 2024 Incentive Plans shall, unlike the other MorphoSys Incentive Awards, not automatically vest as of the Closing but shall be converted into the right of the respective beneficiary to receive an unvested amount in cash (a “Converted Cash Award”) equal to the Offer Price per MorphoSys Incentive Award (determined on the basis of 100% target achievement, if applicable). Converted Cash Awards will generally vest, subject to the continued employment of the holder thereof with Novartis AG and its Affiliates (including the MorphoSys Group), on the same vesting schedule (including with respect to any terms providing for acceleration of vesting upon an involuntary termination of employment) and otherwise on substantially the same terms as the corresponding MorphoSys Incentive Award, subject to the following exceptions:
| (i) | Terms rendered inoperative by reason of the transactions contemplated by the Business Combination Agreement are no longer applicable; |
| (ii) | Other administrative or ministerial changes that are necessary in the reasonable and good faith determination of Novartis AG to facilitate the administration of the Converted Cash Awards; and |
| (iii) | Subject to applicable law, no performance-based vesting metrics or criteria will apply from and after the Delisting. |
MorphoSys, Novartis AG and the Bidder will procure and take, subject to applicable law, all actions that are required or desirable to ensure that, consistent with past practice and subject to applicable law, all
92
holders of Converted Cash Awards that relate to MorphoSys Incentive Awards granted under the 2024 Incentive Plans (including the members of the Management Board) who leave the MorphoSys Group following completion of the Takeover Offer are compensated with respect to the 2024 Incentive Plans and the Converted Cash Awards.
MorphoSys will, to the extent necessary, attempt to obtain the consent to the treatment described above of any holder of a MorphoSys Incentive Award who is not entitled, pursuant to the terms of such MorphoSys Incentive Award, to have payment accelerated in the manner set forth above (the “Equity Consents”). If any holder does not so consent, MorphoSys will settle such awards in cash at their originally scheduled settlement times, with the cash amount equal to the cash amounts described above or based on the cash compensation offered in the Delisting offer, if required by the applicable Incentive Plan.
MorphoSys will, following the Closing and following both the acceleration of MorphoSys Incentive Awards and the provision of the Equity Consents described above, ensure that no holder of MorphoSys Incentive Awards (subject to limited exceptions in the case that the holder of such MorphoSys Incentive Awards does not provide a consent to accelerate their MorphoSys Incentive Awards as described above) will have any right to acquire any securities of MorphoSys, Novartis AG or the Bidder, or to receive any payment or other benefit with respect to any MorphoSys Incentive Award other than as described above.
Any payments with respect to MorphoSys Incentive Awards and Converted Cash Awards held by a United States taxpayer, shall in each case be made in accordance with Sec. 409A of the U.S. Internal Revenue Code of 1986, as amended (the “Code”).
To the extent that any payment made as described above or otherwise relating to the Takeover would trigger any excise tax to any service provider under Sec. 4999 of the Code for any “disqualified individual” who is a United States taxpayer, MorphoSys will, to the extent legally permissible, use its best efforts to enter into agreements to “gross up” the relevant individuals for the further applicable taxes owed, up to a maximum amount of USDm 13, as further described in Sec. 13.2 of this Statement.
Novartis AG, the Bidder and MorphoSys have entered into an arrangement (the “Support Arrangement”) to address a situation in which the actions described above relating to the modification of the remuneration system, the provision of the severance payments or the settlement of the Incentive Plans have not occurred as provided in the Business Combination Agreement. Pursuant to the Support Arrangement, in the event that any payment otherwise required to be made to the Management Board members and employees of the MorphoSys Group as described above and in accordance with their respective agreements is not made either as a result of limitations in the existing remuneration system or as a result of limitations in the Incentive Plans, or in the event that MorphoSys does not enter into the gross-up agreements contemplated, Novartis AG is generally obligated to pay the applicable amounts otherwise owed but so limited, less withholding, to each relevant individual.
| 13.1.3 | Certain Assumptions |
The amounts set forth in the tables below are estimates based on multiple assumptions that may or may not actually occur. Except as otherwise specifically noted, for purposes of quantifying the potential payments and benefits described in this section, the following assumptions, as well as those described in the footnotes to the tables set forth below, were used:
| • | The Delisting occurs on 15 August 2024, which date is an assumed date solely for purposes of the disclosure in this section (which is referred to as the “assumed effective time”), and the date that the Delisting actually occurs may be different than the assumed effective time. |
| • | The Offer Price is EUR 68.00. |
| • | Each Management Board member experiences a termination of employment and becomes eligible to receive severance benefits in connection with the termination of their employment and be compensated with respect to the 2024 Incentive Plans and the Converted Cash Awards immediately following the assumed effective time. |
93
As the amounts indicated below are estimates based on multiple assumptions that may or may not actually occur, the actual amounts, if any, that may be paid or become payable may materially differ from the estimates set forth below.
| 13.1.4 | Settlement of the Performance Share Units of the Management Board |
The following table summarizes, as of 1 April 2024, the outstanding Performance Share Units held by the Management Board members that are expected to be settled after the Delisting. The amounts set forth in the tables below are estimates based on multiple assumptions that may or may not actually occur, including the assumptions described in this Statement. Some of these assumptions are based on information currently available and, as a result, the actual amounts, if any, to be received by any member of the Management Board may differ in material respects from the amounts set forth below.
| Management Board member | No. of MorphoSys PSUs (#)(1) | Resulting Consideration (Capped)(2) | Resulting Consideration (Uncapped)(3) | |||||||||
Jean-Paul Kress, M.D. | 380,209 | EUR 16,517,420 | EUR 25,854,212 | |||||||||
Lucinda Crabtree, Ph.D. | 28,571 | EUR 1,942,828 | EUR 1,942,828 | |||||||||
| (1) | The amounts in this column represent the number of Performance Share Units issued to each Management Board member, except for the 98,484 and 26,262 Performance Share Units that were issued to Jean-Paul Kress, M.D. and Lucinda Crabtree, Ph.D. under the 2024 Performance Share Unit Program, respectively (in this respect, see Sec. 13.2 of this Statement). |
| (2) | The amounts in this column are equal to the product of (i) the number of Performance Share Units held by the Management Board member pursuant to the first column multiplied by (ii) the Offer Price of EUR 68.00, with the result being capped in accordance with the payout caps that are currently applicable to the payouts under these Performance Share Unit Programs. |
| (3) | The amounts in this column are equal to the product of (i) the number of Performance Share Units held by the Management Board member pursuant to the first column multiplied by (ii) the Offer Price of EUR 68.00, without giving effect to any compensation caps. |
As of the date of this Statement, none of the Management Board members holds any Restricted Stock Units. While Dr. Kress holds 81,989 Stock Options, all of these Stock Options have an exercise price equal to or greater than the Offer Price of EUR 68.00 and, as such, forfeit without consideration. Dr. Crabtree does not hold any Stock Options.
Further, no member of the Supervisory Board holds any Performance Share Units, Restricted Stock Units, Stock Options or Converted Cash Awards.
| 13.2 | Agreements with Members of the Management Board or Supervisory Board |
Severance Payments
MorphoSys has entered into service agreements with the Management Board members (together, the “Service Agreements”) that provide for severance benefits upon the termination of such following a change of control. in this regard, the Service Agreements provide that if (i) a change of control occurs and (ii), within one (1) year after the change of control, the Management Board member’s responsibilities are materially reduced, the respective Management Board member has the right to resign from office as a member of the Management Board within three (3) months after the reduction of his/her responsibilities has occurred by giving three (3) months’ notice to the end of a calendar month. The Service Agreement of the relevant Management Board member will terminate at the same time as the resignation from the Management Board becomes effective. Upon such termination, the Management Board member is entitled to a severance payment, in the amount of the annual gross fixed salary and the annual bonus (in
94
an amount based upon the annual bonus paid in the previous year) until the regular expiry of the Service Agreement (31 August 2025 in the case of Dr. Kress and 6 August 2026 in the case of Dr. Crabtree). Under the Service Agreements, severance payments shall not exceed the lesser of twice the annual remuneration and the remuneration for more than the remaining term of the Service Agreement.
Based on the assumptions described above, the table below sets forth the estimated value of the severance payments and benefits that the Management Board members would generally be eligible to receive in case of a termination of services, effective 31 August 2024, which entitles them to receive severance benefits under their respective current Service Agreements (unless otherwise agreed between MorphoSys and the Management Board members).
| Management Board member | Cash Severance(1) | |||
Jean-Paul Kress, M.D. | EUR 2,204,858 | |||
Lucinda Crabtree, Ph.D. | EUR 2,024,123 | |||
| (1) | The Management Board members would each only be entitled to the above severance payment in case prior to the end of 31 May 2024 (i) Closing has occurred, (ii) the Supervisory Board has resolved upon a material reduction in the areas of responsibility of the Management Board member and (iii) as a consequence, the member of the Management Board has provided notice of resignation from their office as Management Board member no later than 31 May 2024. |
The members of the Supervisory Board are not entitled to any severance payments.
Converted Cash Awards
The following table summarizes, as of 1 April 2024, the Performance Share Units granted to the Management Board members under the 2024 Incentive Plans that will become Converted Cash Awards following Delisting.
The amounts set forth in the tables below are estimates based on multiple assumptions, as described above.
| Management Board member | Aggregate No. of MorphoSys PSUs (#)(1) | Resulting Converted Cash Award (2) | ||||||
Jean-Paul Kress, M.D. | 98,484 | EUR 6,696,912 | ||||||
Lucinda Crabtree, Ph.D. | 26,262 | EUR 1,785,816 | ||||||
| (1) | The amounts in this column represent the Performance Share Units that were issued to Jean-Paul Kress, M.D. and Lucinda Crabtree, Ph.D. under the 2024 Performance Share Unit Program, respectively. |
| (2) | The amounts in this column represent the value of the Converted Cash Awards, which are equal to the product of (i) the number of Performance Share Units issued to the Management Board member under the 2024 Performance Share Unit Program (ii) the Offer Price of EUR 68.00. The payout caps under the 2024 Performance Share Unit Program are higher than the uncapped values so will not result in lower values being paid. |
Agreement Not To Compete
The Service Agreements bind the members of the Management Board to post-employment non-compete covenants in exchange for a compensatory payment equal to the Management Board member’s base salary, paid in monthly installments (though, according to the Service Agreement, such payment shall be reduced by the amount of any severance payment to which the Management Board member is entitled, as well as any other income the Management Board member obtains through the provision of services for an
95
employer other than MorphoSys during the same period in case and to the extent such remuneration exceeds the most recent contractual remuneration received from MorphoSys). Dr. Kress’ Service Agreement initially included a (six) 6-month post-employment non-compete covenant. MorphoSys expects to amend Dr. Kress’ Service Agreement at a later date (i.e., after the publication of this Statement) to extend the length of such non-compete covenant to twelve (12) months. Dr. Crabtree’s Service Agreement includes a six (6)-month post-employment non-compete covenant.
280G Gross Up
MorphoSys may be permitted to take certain actions to reduce the amount of any potential “excess parachute payments” for “disqualified individuals” (each as defined in Sec. 280G of the Code (“Section 280G”)) that are United States taxpayers, including Dr. Kress. To the extent that MorphoSys reasonably believes that such disqualified individual(s) may receive or be entitled to receive excess parachute payments in connection with the Takeover, MorphoSys may, after providing Novartis AG a reasonable opportunity to review and provide comments, enter into agreements to pursuant to which each such “disqualified individual” is entitled to receive a compensation for any additional taxes owed under Sec. 4999 of the Code (gross-up) (i.e., a payment which puts the employee in the same after-tax position that the employee would have been in had the excise tax not applied) up to an aggregate limit of USDm 13. MorphoSys has not yet approved any specific actions to mitigate the anticipated impact of Section 280G (other than the extension of the non-compete period described above), but it is anticipated that such gross-up payments will be approved following Delisting.
Agreements with Novartis AG Following the Takeover
As of the date of this Statement, none of the Management Board or Supervisory Board members has entered into any new agreement, arrangement or understanding with Novartis AG or any of its Affiliates regarding the terms and conditions of compensation, incentive pay or employment with MorphoSys after the Takeover. Although no agreements have been entered into at this time with any of the Management Board or Supervisory Board members, prior to or following the completion of the Takeover, they may enter into new agreements and/or amendments to existing employment or severance agreements with Novartis AG or one of its Affiliates regarding their employment with MorphoSys after the Takeover. Such agreements may also include arrangements with regard to the above-described compensation for the 2024 Incentive Plans and the Converted Cash Awards.
Management Board and Supervisory Board Member Indemnification and Insurance
The following summary of the indemnification of Management Board and Supervisory Board members and the D&O insurance does not purport to be complete. A detailed description can be found in the Business Combination Agreement, copy of which is filed as Exhibit 99.3 to a Form 6-K filed by MorphoSys with the SEC on 7 February 2024.
Pursuant to the Business Combination Agreement and subject to the terms thereof and provided that completion of the Takeover has actually occurred, Novartis AG and the Bidder will, for a period of six (6) years following the Closing, indemnify and hold harmless any current or former director or officer of the MorphoSys Group (for purposes of this section, each a “Beneficiary”) from any and all damages. This does not include claims of Novartis AG and the Bidder against MorphoSys expressly provided under the Business Combination Agreement. This is achieved, among other things, by the payment in advance by Novartis AG of the final disposition of any claim, suit or other proceeding of any expenses incurred in defense thereof by Novartis AG or, in their discretion, by the MorphoSys Group indemnifying and holding harmless the respective Beneficiary. Any such indemnification pertains to matters existing or occurring prior to the Closing and arising out of or pertaining to the fact that such person is or was a director or officer of the MorphoSys Group prior to the Closing, or is or was serving at the request of the
96
MorphoSys Group as a director or officer of another legal person prior to the Closing. The indemnification does not depend, to the extend legally permitted, whether any damages asserted or claimed by the Beneficiary, prior to, at or after the Closing, provided that Novartis AG and the Bidder may require a Beneficiary to provide an undertaking by such Beneficiary to repay any amounts paid to such Beneficiary if it is ultimately determined that such Beneficiary is not entitled to be indemnified. Novartis AG and the Bidder will not be so obligated if and to the extent to which (i) a violation leading to such damages was caused by willful misconduct of MorphoSys or any other MorphoSys Group Company or of one of the officers, directors, employees or advisors of the MorphoSys Group, (ii) the relevant Beneficiary is liable to Novartis AG and the Bidder in respect of the respective action or omission, or (iii) the relevant damage is covered under D&O or other insurance at MorphoSys or any other MorphoSys Group Company.
Pursuant to the Business Combination Agreement, for a period of six (6) years following the Closing, the Bidder and Novartis AG will either cause to be maintained in effect the current D&O insurance policies or take out substitute policies for the MorphoSys Group and their current and former directors and officers who are currently covered by the D&O insurance policies. The substitutional policies will be of no less than the existing coverage and will have no other terms less favorable to the insured persons than the D&O insurance policies with respect to claims arising from facts or events that occurred at or before the Closing. However, Novartis AG or the Bidder are not required to pay with respect to such substitutional insurance policies an aggregate annual premium of more than 400% of the aggregate annual premium most recently paid by the MorphoSys Group prior to 5 February 2024 (the “Maximum Amount”). If the Bidder and Novartis AG are unable to obtain the insurance required, they will obtain as much comparable insurance as possible for the years within such six (6)-year period for a premium equal to the Maximum Amount.
In lieu of such insurance, prior to the Closing MorphoSys may, at its option, purchase a “tail” directors’ and officers’, employment practices (if any) and fiduciary (if any) liability insurance policy for the MorphoSys Group and their current and former directors and officers who are currently covered by the D&O insurance policies to provide coverage in an amount not less than the existing coverage and to have other terms not less favorable to the insured persons than the D&O insurance policies with respect to claims arising from factors or events that occurred at or before the Closing; provided that in no event will the cost of any such tail policy exceed the Maximum Amount.
| 14 | SHAREHOLDINGS AND INTENTION OF THE MEMBERS OF THE MANAGEMENT BOARD AND OF THE SUPERVISORY BOARD TO ACCEPT THE OFFER |
As of the date of publication of this Statement, no affiliate or subsidiary of MorphoSys or member of the Management Board holds any MorphoSys Securities. The following members of the Supervisory Board hold MorphoSys Securities:
| Name | Number of MorphoSys Shares | Approximate percentage of total MorphoSys Shares | ||||||
Marc Cluzel, M.D., Ph.D. | 8,525 | 0.02 | % | |||||
Krisja Vermeylen | 3,000 | 0.01 | % | |||||
Michael Brosnan | 5,000 | (1) | 0.01 | % | ||||
| (1) | Represented by 20,000 MorphoSys ADSs. |
All members of the Supervisory Board who hold MorphoSys Securities have individually declared their intention, and still intend, to tender all MorphoSys Securities held by them into the Offer.
| 15 | FINAL STATEMENT AND RECOMMENDATION |
Each MorphoSys Securityholder must make their own decision on whether or not to accept the Offer, taking into account the overall circumstances, their individual situation (including their individual tax
97
situation), and their personal assessment of the possibilities of the future development of the value and share price of MorphoSys Shares.
Subject to applicable law, the Management Board and the Supervisory Board assume no responsibility in the event that the acceptance or non-acceptance of the Offer should subsequently have adverse economic consequences for any MorphoSys Securityholder.
After due and careful consideration of the information in this Statement and the overall circumstances of and in connection with the Offer as well as the goals and intentions of the Bidder, and in light of an overall assessment of the audits, analyses and evaluations performed, the Management Board and the Supervisory Board regard the consideration offered by the Bidder to be attractive, fair and adequate within the terms of Sec. 31 para. 1 WpÜG.
Furthermore, after due consideration, the Management Board and the Supervisory Board, each for itself, (i) unanimously determined that, on the terms of and subject to the conditions of the Business Combination Agreement, the Offer and the related actions as contemplated in the Business Combination Agreement, are in the best interest of MorphoSys and the MorphoSys Securityholders, the sustainable success of its business and of its clients, employees and other stakeholders and (ii) unanimously approved the execution and the delivery of the Business Combination Agreement by MorphoSys and the performance by MorphoSys of its obligations under the Business Combination Agreement and the completion of the transactions contemplated by the Business Combination Agreement, including the Offer.
Therefore, upon due consideration of the Offer and the related actions as contemplated in the Business Combination Agreement and, taking into account the considerations and explanations contained in this Statement, the Management Board and the Supervisory Board unanimously support the Offer and recommend to all MorphoSys Securityholders to accept the Offer and tender their MorphoSys Securities under the Offer.
This Statement and the recommendation given above were both unanimously adopted by the Management Board and the Supervisory Board independently on 11 April 2024.
Planegg, Germany, 11 April 2024
MorphoSys AG
| The Management Board | The Supervisory Board |
98
Annex
Offer Opinion
99

Centerview Partners LLC
31 West 52nd Street
New York, NY 10019
April 10, 2024
The Management Board (Vorstand) and the Supervisory Board (Aufsichtsrat)
MorphoSys AG
Semmelweisstrasse 7, 82152
Planegg, Germany
The Management Board and the Supervisory Board:
You have requested our opinion as to the fairness, from a financial point of view, to the holders of the outstanding no-par value bearer shares (auf den Inhaber lautende Stückaktien), each representing a participation in the registered share capital of MorphoSys AG with a notional amount (rechnerischer Anteil) of EUR 1.00 (the no par-value bearer shares issued by the Company from time to time, the “Shares”) (other than Excluded Shares, as defined below), of MorphoSys AG, a publicly listed stock corporation (Aktiengesellschaft) incorporated under the Laws of Germany (the “Company”), of €68.00 per Share in cash (the “Consideration”) proposed to be paid to such holders pursuant to, and subject to the terms and conditions of, the offer document to be published by Novartis data42 AG, a stock corporation incorporated under the Laws of Switzerland, with registered office at Lichtstraße 35, 4056 Basel, Switzerland and being registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under company number CHE-477.907.492 (the “Bidder”), on April 11, 2024 (the “Offer Document”) in accordance with the terms set forth in the Business Combination Agreement, dated as of February 5, 2024 (the “Agreement”), by and among Novartis AG, a stock corporation incorporated under the Laws of Switzerland, with registered office at Lichtstraße 35, 4056 Basel, Switzerland and being registered with the commercial register office (Handelsregisteramt) of the Canton of Basel-City under CHE-103.867.266 which wholly owns the Bidder (“Parent”), the Bidder, and the Company. The Offer Document provides for the acquisition by the Bidder of all of the Shares (other than any American Depositary Shares of the Company; those American Depository Shares together with the Shares which are for the time being held by the Company as treasury shares (eigene Aktien), the “Excluded Shares”) for the Consideration by means of a voluntary public cash takeover offer (freiwilliges öffentliches Übernahmeangebot) (the “Takeover Offer”) within the meaning of Section 29 para. 1 of the German Takeover Act (the “Transaction”). The terms and conditions of the Transaction are more fully set forth in the Offer Document.
We have acted as lead financial advisor to the Management Board and the Supervisory Board of the Company in connection with the Transaction. We will receive a fee for our services in connection with the Transaction, a portion of which is payable upon the rendering of this opinion and a substantial portion of which is contingent upon the consummation of the Takeover Offer. In addition, the Company has agreed to reimburse certain of our expenses arising, and indemnify us against certain liabilities that may arise, out of our engagement.
We are a securities firm engaged directly and through affiliates and related persons in a number of investment banking, financial advisory and merchant banking activities. In the past two years, except for our
31 WEST 52ND STREET, 22ND FLOOR, NEW YORK, NY 10019
PHONE: (212) 380-2650 FAX: (212) 380-2651 WWW.CENTERVIEWdfsdfPARTNERS.COM
NEW YORK • LONDON • PARIS • SAN FRANCISCO • MENLO PARK
The Management Board and Supervisory Board
MorphoSys AG
04.10.2024
Page 2
current engagement, we have not been engaged to provide financial advisory or other services to the Company, and we have not received any compensation from the Company during such period. In the past two years, we have not been engaged to provide financial advisory or other services to Parent or Bidder, and we have not received any compensation from Parent during such period. We may provide financial advisory and other services to or with respect to the Company or Parent or their respective affiliates in the future, for which we may receive compensation. Certain (i) of our and our affiliates’ directors, officers, members and employees, or family members of such persons, (ii) of our affiliates or related investment funds and (iii) investment funds or other persons in which any of the foregoing may have financial interests or with which they may co-invest, may at any time acquire, hold, sell or trade, in debt, equity and other securities or financial instruments (including derivatives, bank loans or other obligations) of, or investments in, the Company, Parent or any of their respective affiliates, or any other party that may be involved in the Transaction.
In connection with this opinion, we have reviewed, among other things: (i) a draft of the Offer Document dated April 10, 2024 (the “Draft Offer Document”); (ii) the Agreement; (iii) Annual Reports on Form 20-F of the Company for the years ended December 31, 2023, December 31, 2022 and December 31, 2021; (iv) certain interim reports to shareholders on Form 6-K of the Company; (v) certain publicly available research analyst reports for the Company; (vi) certain other communications from the Company to its shareholders; and (vii) certain internal information relating to the business, operations, earnings, cash flow, assets, liabilities and prospects of the Company, including certain financial forecasts, analyses and projections relating to the Company prepared by management of the Company and furnished to us by the Company for purposes of our analysis (the “Forecasts”) (collectively, the “Internal Data”). At your direction, we have converted certain figures in the Forecasts from EUR to USD based on an exchange rate as of February 2, 2024. We have also participated in discussions with members of the senior management and representatives of the Company regarding their assessment of the Internal Data. In addition, we reviewed publicly available financial and stock market data, including valuation multiples, for the Company and compared that data with similar data for certain other companies, the securities of which are publicly traded, in lines of business that we deemed relevant. We also compared certain of the proposed financial terms of the Transaction with the financial terms, to the extent publicly available, of certain other transactions that we deemed relevant and conducted such other financial studies and analyses and took into account such other information as we deemed appropriate.
We have assumed, without independent verification or any responsibility therefor, the accuracy and completeness of the financial, legal, regulatory, tax, accounting and other information supplied to, discussed with, or reviewed by us for purposes of this opinion and have, with your consent, relied upon such information as being complete and accurate. In that regard, we have assumed, at your direction, that the Internal Data (including, without limitation, the Forecasts) has been reasonably prepared on bases reflecting the best currently available estimates and judgments of the management of the Company as to the matters covered thereby and we have relied, at your direction, on the Internal Data for purposes of our analysis and this opinion. We express no view or opinion as to the Internal Data or the assumptions on which it is based. In addition, at your direction, we have not made any independent evaluation or appraisal of any of the assets or liabilities (contingent, derivative, off-balance-sheet or otherwise) of the Company, nor have we been furnished with any such evaluation or appraisal, and we have not been asked to conduct, and did not conduct, a physical inspection of the properties or assets of the Company. We have assumed, at your direction, that the Offer Document will not differ in any respect material to our analysis or this opinion from the Draft Offer Document reviewed by us. We have also assumed, at your direction, that the Transaction will be consummated on the terms set forth in the Offer Document, the Agreement and in accordance with all applicable laws and other relevant documents or requirements, without delay or the waiver, modification or amendment of any term, condition or agreement, the effect of which would be material to our analysis or this opinion and that, in the course of obtaining the necessary governmental, regulatory and other approvals, consents, releases and waivers for the Transaction, no delay, limitation, restriction, condition or other change will be imposed, the effect of which would be material to our
The Management Board and Supervisory Board
MorphoSys AG
04.10.2024
Page 3
analysis or this opinion. We have not evaluated and do not express any opinion as to the solvency or fair value of the Company, or the ability of the Company to pay its obligations when they come due, or as to the impact of the Transaction on such matters, under any state, federal or other laws relating to bankruptcy, insolvency or similar matters. We are not legal, regulatory, tax or accounting advisors, and we express no opinion as to any legal, regulatory, tax or accounting matters.
Our assessment differs in a number of important aspects from a valuation performed by qualified auditors and/or from asset based valuations in general. In particular, we have not performed valuations based upon the guidelines published by the German Institute of Chartered Accountants (IDW) (IDW S 1). This opinion does not replace such valuations. We express no view on whether, in light of the nature of the Transaction, it may be required or appropriate for the Company to obtain such valuations. In addition, this letter has not been rendered in accordance with the IDW guidelines “Principles for the preparation of Fairness Opinions” (IDW S 8).
We express no view as to, and our opinion does not address, the Company’s underlying business decision to proceed with or effect the Transaction, or the relative merits of the Transaction as compared to any alternative business strategies or transactions that might be available to the Company or in which the Company might engage. This opinion is limited to and addresses only the fairness, from a financial point of view, as of the date hereof, to the holders of the Shares (other than Excluded Shares) of the Consideration to be paid to such holders pursuant to the Offer Document. We have not been asked to consider, nor do we express any view on, and our opinion does not address, any other term or aspect of the Offer Document or the Transaction, including, without limitation, the structure or form of the Transaction, or any other agreements or arrangements contemplated by the Agreement, described in the Offer Document or entered into in connection with or otherwise contemplated by the Transaction, including, without limitation, the fairness of the Transaction or any other term or aspect of the Transaction to, or any consideration to be received in connection therewith by, or the impact of the Transaction on, the holders of any other class of securities, creditors or other constituencies of the Company or any other party. Further, we have not been asked to consider, nor do we express any view on, and our opinion does not address, any term or aspect of that certain Purchase Agreement, dated as of February 5, 2024 (the “Asset Purchase Agreement”), by and among the Company, MorphoSys US Inc., a Delaware corporation and a wholly-owned subsidiary of the Company (“MorphoSys US”), and Incyte Corporation, a Delaware corporation, pursuant to which certain assets of the Company and MorphoSys US were sold immediately prior to the execution of the Agreement, or how the Asset Purchase Agreement may impact the Company, the Offer Document, the Agreement or the Transaction. In addition, we express no view or opinion as to the fairness (financial or otherwise) of the amount, nature or any other aspect of any compensation to be paid or payable to any of the officers, directors or employees of the Company or any party, or class of such persons in connection with the Transaction, whether relative to the Consideration to be paid to the holders of the Shares pursuant to the Offer Document or otherwise. Our opinion is necessarily based on financial, economic, monetary, currency, market and other conditions and circumstances as in effect on, and the information made available to us as of, the date hereof, and we do not have any obligation or responsibility to update, revise or reaffirm this opinion based on circumstances, developments or events occurring after the date hereof. Our opinion does not constitute a recommendation to any shareholder of the Company as to whether or not such holder should tender Shares in connection with the Takeover Offer, or how such shareholder or other person should act with respect to the Transaction or any other matter.
Our financial advisory services and the opinion expressed herein are provided for the information and assistance of the Management Board and the Supervisory Board of the Company (in their capacity as members of each such board, respectively, and not in any other capacity) in connection with and for purposes of their consideration of the Transaction. The issuance of this opinion was approved by the Centerview Partners LLC Fairness Opinion Committee.
The Management Board and Supervisory Board
MorphoSys AG
04.10.2024
Page 4
Based upon and subject to the foregoing, including the various assumptions made, procedures followed, matters considered, and qualifications and limitations set forth herein, we are of the opinion, as of the date hereof, that the Consideration to be paid to the holders of Shares (other than Excluded Shares) pursuant to the Offer Document by means of the Takeover Offer is fair, from a financial point of view, to such holders.
| Very truly yours, |
/s/ Centerview Partners |
| CENTERVIEW PARTNERS LLC |