UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-K
(Mark One)
☒ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended December 31, 2021
OR
☐ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
For transition period from to
Commission File Number 001-36332
ALDEYRA THERAPEUTICS, INC.
(Exact name of Registrant as specified in its charter)
| |
Delaware | 20-1968197 |
(State or other jurisdiction of incorporation) | (IRS Employer Identification No.) |
131 Hartwell Avenue, Suite 320
Lexington, MA 02421
(Address of principal executive offices)
(781) 761-4904
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| | |
Common Stock, $0.001 par value per share | ALDX | The Nasdaq Stock Market, LLC |
(Title of each class) | (Trading Symbol) | (Name of each exchange on which registered) |
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer a smaller reporting company or an emerging growth company. See the definitions of the “large accelerated filer,” “accelerated filer,” “non-accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| | | |
Large Accelerated Filer | ☐ | Accelerated Filer | ☐ |
Non-Accelerated Filer | ☒ | Smaller reporting company | ☒ |
| Emerging Growth Company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No ☒
As of June 30, 2021, the last business day of the registrant’s last completed second quarter, the aggregate market value of the registrant’s Common Stock held by non-affiliates of the registrant was approximately $648,685,384, based on the closing price of the registrant’s Common Stock, as reported by The Nasdaq Capital Market. Shares of Common Stock held by each executive officer, director and stockholders known by the registrant to be affiliated with such individuals based on public filings and other information known to the registrant have been excluded since such persons may be deemed affiliates. This determination of affiliate status is not necessarily a conclusive determination for other purposes.
As of March 15, 2022 there were 58,098,965 shares of the registrant’s Common Stock issued and outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
Specified portions of the registrant’s proxy statement with respect to the registrant’s 2022 Annual Meeting of Stockholders, which is to be filed pursuant to Regulation 14A within 120 days after the end of the registrant’s fiscal year ended December 31, 2021, are incorporated by reference into Part III of this Annual Report on Form 10-K.
Aldeyra Therapeutics, Inc.
Annual Report on Form 10-K
For the Fiscal Year Ended December 31, 2021
Table of Contents
2
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
Various statements throughout this report are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements involve substantial risks and uncertainties. All statements, other than statements of historical facts, included in this report regarding our strategy, future operations, future financial position, future revenues, projected costs, prospects, plans and objectives of management are forward-looking statements. These statements are subject to risks and uncertainties and are based on information currently available to our management. Words such as, but not limited to, “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,” “contemplates,” “predict,” “project,” “target,” “likely,” “potential,” “continue,” “ongoing,” “design,” “might,” “objective,” “will,” “would,” “should,” “could,” or the negative of these terms and similar expressions or words, identify forward-looking statements. These statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. The events and circumstances reflected in our forward-looking statements may not occur and actual results could differ materially from those projected in our forward-looking statements. Meaningful factors which could cause actual results to differ include, but are not limited to:
•the timing of enrollment, commencement, and completion of our clinical trials;
•the timing and success of preclinical studies and clinical trials conducted by us and our development partners;
•delay in or failure to obtain regulatory approval of our product candidates, including as a result of the U.S. Food and Drug Administration (FDA) not accepting our regulatory filings or requiring additional clinical trials or data prior to review or approval of such filings;
•the ability to maintain regulatory approval of our product candidates, and the labeling for any approved products;
•the risk that prior results, such as signals of safety, activity or durability of effect, observed from preclinical or clinical trials, will not be replicated or will not continue in ongoing or future studies or trials involving our product candidates;
•the scope, progress, expansion, and costs of developing and commercializing our product candidates;
•the current and potential future impact of the COVID-19 pandemic on our business, results of operations and financial position;
•uncertainty as to our ability to commercialize (alone or with others) and obtain reimbursement for our product candidates following regulatory approval, if any;
•the size and growth of the potential markets and pricing for our product candidates and the ability to serve those markets;
•our expectations regarding our expenses and revenue, the sufficiency or use of our cash resources and needs for additional financing;
•the rate and degree of market acceptance of any of our product candidates;
•our expectations regarding competition;
•our anticipated growth strategies;
•our ability to attract or retain key personnel;
•our limited sales and marketing infrastructure;
•our ability to establish and maintain development partnerships;
•our ability to successfully integrate acquisitions into our business;
•our expectations regarding federal, state and foreign regulatory requirements;
3
•political, economic, legal, social and health risks, including the recent COVID-19 pandemic and subsequent public health measures, and war or other military actions, that may affect our business or the global economy
•regulatory developments in the United States and foreign countries;
•our ability to obtain and maintain intellectual property protection for our product candidates; and
•the anticipated trends and challenges in our business and the market in which we operate.
All written and verbal forward-looking statements attributable to us or any person acting on our behalf are expressly qualified in their entirety by the cautionary statements contained or referred to in this section. We caution investors not to rely too heavily on the forward-looking statements we make or that are made on our behalf. We undertake no obligation, and specifically decline any obligation, to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise. You are advised, however, to consult any further disclosures we make on related subjects in any annual, quarterly or current reports that we may file with the Securities and Exchange Commission (SEC).
We encourage you to read the discussion and analysis of our financial condition and our financial statements contained in this annual report on Form 10-K. We also encourage you to read Item 1A of Part 1 of this annual report on Form 10-K, entitled “Risk Factors,” which contains a more complete discussion of the risks and uncertainties associated with our business. In addition to the risks described above and in Item 1A of this report, other unknown or unpredictable factors also could affect our results. Therefore, the information in this report should be read together with other reports and documents that we file with the SEC from time to time, including Forms 10-Q, 8-K and 10-K, which may supplement, modify, supersede or update those risk factors. There can be no assurance that the actual results or developments anticipated by us will be realized or, even if substantially realized, that our results will lead to the expected consequences to, or effects on, us. Therefore, no assurance can be given that the outcomes stated in such forward-looking statements and estimates will be achieved.
As used in this annual report on Form 10-K, the terms “Aldeyra,” “Registrant,” “the Company,” “we,” “us,” and “our” mean Aldeyra Therapeutics, Inc., together with its wholly-owned subsidiaries, unless the context indicates otherwise.
4
INDUSTRY AND MARKET DATA
We obtained the industry, market and certain other data used throughout this annual report on Form 10-K from our own internal estimates and research, as well as from industry and general publications, surveys and studies conducted by third parties. Internal estimates are derived from publicly-available information released by industry analysts and third-party sources, our internal research, and our industry experience, and are based on assumptions made by us based on such data and our knowledge of our industry and market, which we believe to be reasonable. In addition, while we believe the industry, market, and other data included in this annual report on Form 10-K are reliable and based on reasonable assumptions, such data involves risks and uncertainties and are subject to change based on various factors, including those discussed in “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Risk Factors”. These and other factors could cause results to differ materially from those expressed in the estimates made by independent parties and by us.
ITEM 1. BUSINESS
Overview
We are a biotechnology company devoted to discovering and developing innovative therapies designed to treat immune-mediated diseases. Our lead product candidate, reproxalap, is a first-in-class treatment in late-stage development for dry eye disease (DED) and allergic conjunctivitis (AC). We have additional product candidates in development for autoimmune diseases and other immune-mediated disease, as well as for proliferative vitreoretinopathy (PVR) and other retinal diseases. We currently intend to commercialize our products directly or through collaborations. None of our product candidates have been approved for sale in the United States or elsewhere.
Immune-mediated diseases are conditions that result from an imbalance of inhibitory and stimulatory factors that regulate the immune system. Immunological dysregulation can lead to a broad array of conditions that include autoimmune disease, allergy, immunoproliferative disease, and cancer. Many ocular, cardiovascular, metabolic, neurological, and musculoskeletal diseases, affecting tens of millions of patients in the United States and hundreds of millions of patients worldwide, are at least partially immune-mediated. An estimated 7% of western society suffers from some form of immune-mediated disease, and incidence has been increasing. Given the complexity of immune dysregulation, which involves many mediators and signaling pathways, rarely is any single therapeutic approach effective, and today most immune-mediated diseases are generally considered to be inadequately treated. As such, we believe immune-mediated diseases represent considerable unmet medical need, and that demand for novel immune-modulating therapies is high. Consistent with large patient populations and high therapeutic demand, the current market for the treatment of immune-mediated diseases is considerable, representing an excess of $40 billion worldwide.
Our clinical product development pipeline is focused on immune-mediated ocular diseases and select systemic diseases, and encompasses two distinct biological mechanisms of action: reactive aldehyde species (RASP) modulation and dihydrofolate reductase inhibition. The immunological activity of our product candidates generally leads to diminished levels of pathological inflammation via down-regulation of immune cell activation or proliferation.
Our lead product candidate reproxalap is a RASP modulator that has been shown to diminish ocular inflammation, and has demonstrated statistically significant and clinically relevant improvements across a number of Phase 2 and Phase 3 clinical trials in DED and AC when administered topically to the eye as an ophthalmic solution. Additionally, in a Phase 1 clinical trial, ADX-629, a first-in-class orally available RASP modulator, was well-tolerated and no treatment-related adverse events were observed. Relative to subjects treated with placebo, reduction in the commonly described pro-inflammatory RASP malondialdehyde was observed in ADX-629 treated subjects. A growing body of clinical evidence supports the potential and relevance of RASP modulation as a new and differentiated mechanism of action. We have discovered and are developing additional RASP modulators. The RASP modulator platform represents a unique and novel pharmacologic approach that, unlike almost all drugs in use today, are designed to not directly inhibit or activate a particular protein but instead targets a family of small molecules that in turn affect the activity and structure of many proteins at once. RASP modulation, therefore, has the
5
potential to down-regulate pro-inflammatory systems or groups of proteins, and may lead to multiple beneficial clinical effects while avoiding toxicity associated with single-target inhibition or activation.
As we continue to execute on our strategy of expanding our product candidate pipeline, we may license or acquire new immune-modulating approaches with novel therapeutic potential. In January 2019, we acquired Helio Vision, Inc. and thereby obtained rights to ADX-2191, a vitreous-compatible methotrexate formulation for intraocular injection, for the prevention of PVR, a serious sight-threatening retinal disease with no approved treatment, and primary vitreoretinal lymphoma (PVRL), a rare cancer with no approved treatment. In addition, in December 2016, we in-licensed the clinical-stage product candidate ADX-1612 (investigated in oncology under the name ganetespib), which inhibits the protein chaperome, a mechanistically differentiated approach for the potential treatment of inflammatory diseases.
Our active clinical programs include three unique product candidates, representing two distinct mechanisms of action across a number of different potential indications. All of our development plans and timelines are subject to adjustment depending on recruitment rate, regulatory review, preclinical and clinical results, funding, and other factors that could delay the initiation, completion, or reporting of clinical trials. As a result of the COVID-19 pandemic, clinical site availability, staffing, patient recruitment, and patient retention have been negatively affected and the timelines to complete our clinical trials may be delayed. Our pipeline, as of the date of filing this annual report on Form 10-K is illustrated below.
Product Candidate Development Pipeline
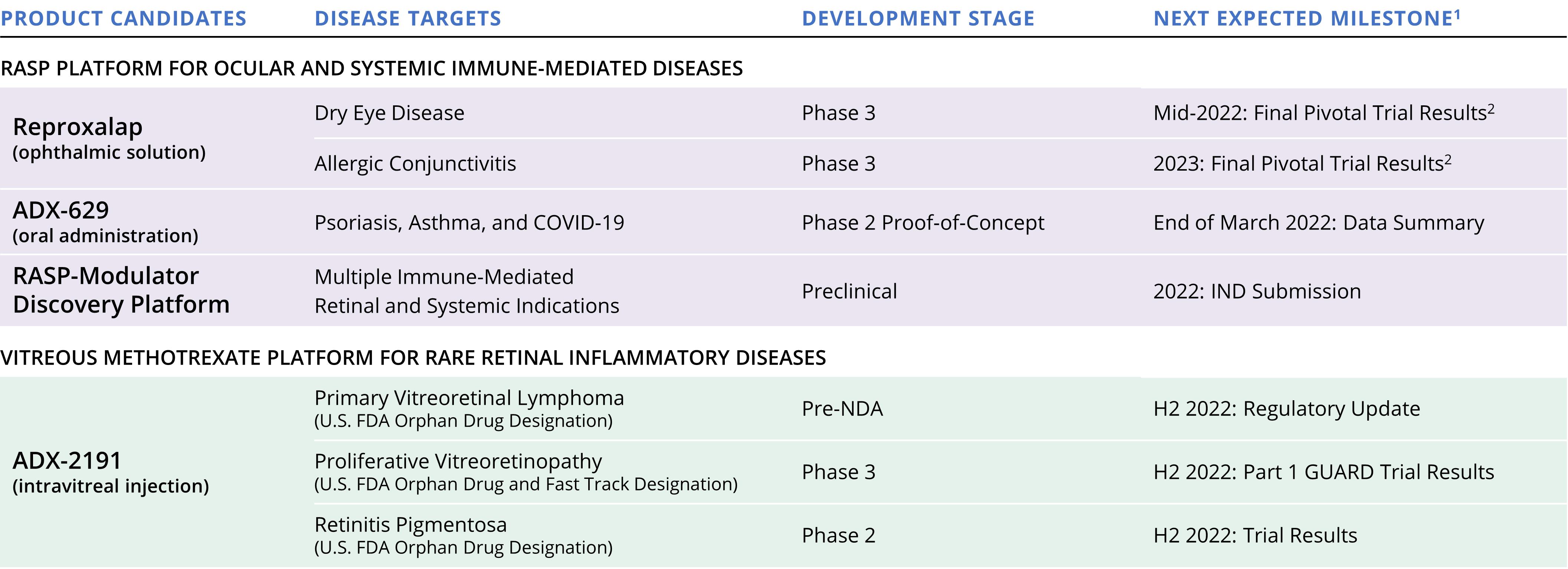
1Timing depends, in part, on restrictions related to the COVID-19 pandemic, the availability of clinical research facilities and staffing, the ability to recruit patients, and regulatory feedback.
2New Drug Application, or NDA submission requirements depend, in part, on clinical results and regulatory feedback.
We have no products approved for sale. We will not receive any revenue from any product candidates that we develop until we obtain regulatory approval and commercialize such products or until we potentially enter into agreements with third parties for the development and commercialization of product candidates. If our development efforts for any of our product candidates result in regulatory approval or we enter into collaboration agreements with third parties, we may generate revenue from product sales or from such third parties. We have primarily funded our operations through the sale of our common stock, convertible preferred stock, convertible promissory notes, warrants, and borrowings under debt facilities.
6
We will need to raise additional capital in the form of debt or equity or through partnerships to fund additional development of our product candidates, and we may in-license, acquire, or invest in complementary businesses or products. In addition, contingent on capital resources, we may augment, diminish, or otherwise modify the clinical development plan described herein.
Since our incorporation, we have devoted substantially all of our resources to the preclinical and clinical development of our product candidates. Our ability to generate revenues, if any, largely depends upon our ability, alone or with others, to complete development of and obtain regulatory approvals for our product candidates, and to successfully manufacture, market, and sell our product candidates. The results of our operations will vary significantly from year-to-year and quarter-to-quarter, and depend on a number of factors, including risks related to our business and industry, risks relating to intellectual property and other legal matters, risks related to our common stock, and other risks that are detailed in the section of this annual report on Form 10-K entitled “Risk Factors".
The Markets for Our Product Candidates
Dry Eye Disease and Allergic Conjunctivitis – Two Prevalent Diseases with Significant Comorbidity
The symptoms of DED - ocular pain, dryness, gritty sensation - and AC - ocular itching and tearing- are chronic and persistently disturbing, impacting quality of life and leading to loss of work and substantial economic burden. DED and AC are two of the most common diseases treated by ophthalmologists and optometrists, and physicians and patients regard therapy as inadequate in a substantial number of cases.
There are approximately 18 million DED patients diagnosed in the United States, but only three topical ocular drugs are approved for DED treatment, cyclosporine (0.05% as Restasis®, 0.05% generic cyclosporine, or 0.09% as CequaTM), lifitegrast (5% as Xiidra®), and loteprednol (0.25% as Eysuvis®). The activity of cyclosporine and lifitegrast has been observed to be minimal or lacking in the majority of patients, and weeks or months of treatment may be required to achieve even modest clinical benefit with over 60% of patients discontinuing treatment within 12 months of initiation. Loteprednol, a generically available corticosteroid, is indicated only for short-term treatment (up to two weeks) due to corticosteroid-associated toxicity, which includes increases in intraocular pressure that may lead to glaucoma, the development of cataracts, ocular infection, and other ocular morbidities.
There are approximately 100 million patients in the United States with AC, and we estimate that up to 30 million of such AC patients do not respond adequately to, or are dissatisfied with, topical antihistamines, the current standard of care. A primary reason for dissatisfaction with antihistamines appears to be lack of durable activity, which may be due to the fact that histamine is only one of the biological mediators of the disease, and the fact that increased histamine levels persist for only 10 to 20 minutes following allergen exposure.
Many patients manifest symptoms of both DED and AC, and differential diagnosis can be challenging for physicians. Approximately half of dry eye patients complain of itching, which is generally considered the result of allergy, and approximately half of AC patients complain of dryness, which is generally considered the result of DED. There are currently no United States Food and Drug Administration (FDA)-approved products that are indicated to treat both DED and AC. Neither cyclosporine nor lifitegrast have been approved for use in patients with AC, and antihistamines are known to exacerbate ocular dryness. Thus, we believe that no currently available drug for DED or AC is likely to be effective for the chronic treatment of the substantial number of patients who experience symptoms of both diseases.
By modulating RASP, which are elevated in a variety of inflammatory diseases, reproxalap represents a novel mechanism for diminishing ocular inflammation in DED and AC. In a number of Phase 2 and Phase 3 clinical trials in DED and AC reproxalap demonstrated consistent statistically significant and clinically relevant activity. We believe that reproxalap may have a commercially differentiated product profile versus currently approved drugs for each indication, having shown the potential for early and broad activity in DED, and durable activity in AC.
Based on the late-stage clinical trial results to date, discussed below, we believe reproxalap could offer differentiated efficacy relative to existing DED medications, particularly with regard to early onset of action and breadth of activity. Thus, our current expectation is that reproxalap could be priced similarly to, or at a premium to, currently marketed drugs for DED, which are generally priced in the range of $480 to $650 per month. The potential
7
size of the DED market is substantial. We estimate there to be 39 million patients with DED in the United States, of which approximately 18 million are diagnosed. Assuming approximately one-half of diagnosed patients are candidates for prescription medication, and assuming approximately four months of therapy per year, the potential total addressable market for reproxalap therapy in DED is greater than $23 billion in the United States.
Contingent on the results of current and planned clinical trials in DED and AC, in addition to regulatory authority approval, we intend to commercialize reproxalap ophthalmic solution directly or through marketing partnerships.
Proliferative Vitreoretinopathy, Primary Vitreoretinal Lymphoma, and Other Retinal Diseases
PVR is a rare inflammatory disorder of the retina that leads to severe retinal scarring and blindness, and is the leading cause of failure of retinal reattachment surgery. Over 50% of PVR cases result in severe uncorrectable vision loss (visual acuity of 20/320 or worse), and 76% of PVR patients suffer from at least moderate uncorrectable vision loss. PVR occurs after up to 10% of surgeries for retinal detachment and 50% or more of surgeries for open globe injury. Based on the prevalence of primary retinal detachment, in addition to retinal detachment that occurs as a result of trauma, we estimate that there are, in aggregate, approximately 20,000 treatable cases of PVR in the United States, Europe, and Japan. By inhibiting cell growth and thereby diminishing scar formation, ADX-2191 has the potential to be the first FDA-approved drug for prevention of PVR. In April 2018, ADX-2191 received orphan drug designation from the FDA for the prevention of PVR and, in June 2020, ADX-2191 was designated an orphan medicinal product by the European Commission for the treatment of retinal detachment. In September 2019, ADX-2191 received fast track designation from the FDA for the prevention of PVR.
Primary vitreoretinal lymphoma (PVRL) is a rare, aggressive, high-grade cancer that arises in the vitreous and retina. An estimated 2,900 people in the United States suffer from PVRL, and approximately 600 new cases of PVRL are diagnosed in the United States per year. The median survival for newly diagnosed patients is less than five years. In January 2021, we received from the FDA preliminary written comments in preparation for a Pre-IND ("Investigational New Drug”) Type B meeting regarding ADX-2191 for the treatment of PVRL. We believe the comments indicated that submission of a New Drug Application (NDA) for ADX-2191 for the treatment of PVRL may be possible without performing clinical trials. In the first quarter of 2021, we held a teleconference with the FDA to discuss the preliminary written comments and clarify the ADX-2191 NDA submission requirements for the treatment of PVRL. In July 2021, ADX-2191 received orphan drug designation from the FDA for the treatment of PVRL. ADX-2191 has the potential to be the first drug approved to treat PVRL.
In addition to PVR and PVRL, the retina is susceptible to a variety of immune-mediated diseases, many of which are mediated by RASP. Inflammatory retinal disorders that involve RASP include both posterior and pan-uveitis, uveitis-associated macular edema, diabetic macular edema, and diabetic retinopathy. Separately, RASP and RASP-adducts accumulate in dry age-related macular degeneration, Stargardt Disease (juvenile dry age-related macular degeneration-like disease), and Sjögren-Larsson Syndrome-associated maculopathy. We believe that the number of patients affected by immune-mediated retinal disorders is considerable. In particular, during 2019 and 2020, the National Eye Institute estimated that diabetic retinopathy and age-related macular degeneration affected approximately 10 million people in the United States, and was projected to double to 20 million individuals by 2050. The global ophthalmic therapeutics market was approximately $21 billion in 2021 with therapies for retinal disorders (age-related, macular degeneration and diabetic macular edema) exceeding $11 billion. Therefore, we believe that the total market potential of RASP modulators for the treatment of retinal disease is substantial.
Immune-Mediated Systemic Diseases
Immune-mediated systemic diseases, such as autoimmune disease, are generally chronic conditions characterized by excessive and misdirected inflammatory responses. In aggregate, autoimmune diseases and related systemic inflammatory disorders represent in excess of 23 million individuals in the United States and collectively, prescription drug sales are expected to exceed $74 billion by 2022. In 2021, the three top selling drugs for these autoimmune conditions accounted for approximately $28 billion in sales.
8
Given the complex pathophysiology of systemic immune-mediated disorders, many of which are caused by a variety of pro-inflammatory mediators, therapy often requires combinations of drugs with distinct mechanisms of action. As such, we believe novel product candidates for immune-mediated diseases are in high demand.
Our RASP modulator platform represents a potential novel therapeutic approach for a variety of common systemic immune-mediated conditions. Because RASP appear to be involved in the generation and potentiation of inflammation in general, we believe the potential therapeutic applicability of RASP modulators is broad. We are not aware of any other company actively developing therapeutics that target RASP. In 2020, we announced the initiation of Phase 2 clinical trials of ADX-629, a first-in-class orally administered RASP modulator, for the treatment of COVID-19, atopic asthma, and psoriasis as part of a systematic strategy to assess activity across different types of immunological diseases. The Phase 2 clinical trials follow a successful Phase 1 clinical trial of ADX-629, in which ADX-629 was well-tolerated, no treatment-related adverse events were observed, and target engagement was evidenced by statistically lower RASP levels in drug-treated subjects relative to controls.
The Competitive Landscape of Our Product Candidates
The pharmaceutical industry is characterized by intense competition and rapid innovation. Our potential competitors include large pharmaceutical and biotechnology companies, specialty pharmaceutical companies, academic institutions, government agencies, and research institutions. We believe that the key competitive factors that will affect the development and potential commercial success of our product candidates are efficacy, safety, tolerability, and the ability to reduce the dependence on, or the dose of, other drug products.
Many of our potential competitors have substantially greater financial, technical, and human resources than we do and significantly greater experience in the discovery and development of product candidates, obtaining FDA and other regulatory approvals of products, and the commercialization of those products. Accordingly, our competitors may be more successful than we may be in obtaining regulatory approval for products and achieving widespread market acceptance. Our competitors’ products may be more effective, or more effectively marketed and sold, than any product that we may commercialize, and may render our product candidates obsolete or non-competitive before we can recover the expenses of developing and commercializing any of our product candidates. Further, competitors with numerous approved products may be able to negotiate pricing and reimbursement that is more favorable than that which we may be able to achieve. We anticipate that we will face intense and increasing competition as new products enter the market and advanced technologies become available. In addition, the development of new treatment methods for the diseases we are targeting could render our products non-competitive or obsolete.
While our product candidates may manifest efficacy, tolerability, or safety advantages, many marketed therapies are generic or may be priced considerably lower than the pricing we anticipate for our product candidates. Pricing, in addition to healthcare plan coverage, prior authorization requirements, step edits, co-pay amounts, and related factors, may discourage the initial or prolonged use of our product candidates. Further, the recent growth of Pharmacy Benefit Managers has diminished the profitability of drug commercialization for smaller companies, and may hamper our ability to support our operations or compete effectively in the marketplace following regulatory approval, if any.
9
RASP Modulator Platform
A number of academic groups have published on the concept of reducing RASP levels, primarily by using compounds with amines (certain nitrogen-containing molecules) that react with RASP through a chemical process known as the Schiff base reaction. Various RASP-binding amines have been described, particularly carnosine (a naturally occurring dipeptide), which has other potential mechanisms of action unrelated to RASP. At least one group has published on the use of certain nitrogen-containing marketed products to temporarily bind retinaldehyde (a RASP) as a potential therapy for retinal disease. Schiff base reactions have also been mentioned as possible explanations for a portion of the activity of aminoguanidine, pyridoxamine, and possibly other non-proprietary amine-containing compounds that have been tested in clinical trials for diabetic nephropathy. However, the Schiff base reaction is reversible, and generally the substrates (precursors) and products of the reaction exist in equilibrium such that, at any point in time, the RASP substrate may be bound or unbound. In this way, Schiff base reactions alone represent temporary RASP binding, and likely lead to the relocation of RASP rather than the elimination or long-term modulation of RASP. We believe that reproxalap and chemically related product candidates that we have discovered are differentiated from the above approaches in that the chemical structures of our product candidates are novel, and the reaction with RASP has been observed to be essentially irreversible in vivo, which, we believe, may result in a more effective means of modulating RASP levels.
Other Immune-Modulating Pharmacotherapies
A myriad of new treatments have been or are being developed to treat inflammatory diseases, and have been used, or in theory could be used, for the treatment of the diseases that our product candidates are intended to target. Immune-modulating products include cytokine inhibitors, immune cell receptor inhibitors, complement inhibitors, phosphodiesterase inhibitors, and Janus kinase inhibitors. Companies that currently market such therapies include Abbvie, Inc., Johnson & Johnson, UCB Inc. and UCB S.A., Amgen, Inc., Bristol-Myers Squibb Co., Eli Lilly and Company, Novartis AG, Regeneron Pharmaceuticals, Inc., Roche, Sanofi, Takeda, AstraZeneca, GlaxoSmithKline, Merck, and Pfizer, Inc. Currently marketed products may manifest efficacy and safety advantages over our product candidates, and may be used to treat the diseases for which we are developing our product candidates.
Methotrexate, the active drug substance of ADX-2191, is generically available and has been used as a chemotherapeutic and immune modulating agent, and other formulations or application methods of methotrexate could be developed for the treatment of retinal diseases. Though not approved by the FDA for the treatment of retinal disease, such as PVRL and PVR, intraocular injection of intravenous methotrexate formulations is the de facto standard of care, and “off-label” methotrexate is now commonly administered for the treatment of PVRL. The off-label intraocular injection of intravenous methotrexate for retinal diseases is an example of a practice known as compounding. The disadvantages of compounding are significant, and include a risk of microbial contamination that can lead to severe ocular infection resulting in vision loss. Unlike compounded intravenous formulations, ADX-2191 is specifically formulated for intraocular injection such that drug concentration, density, pH, and tonicity have been designed to be compatible with the vitreous humor, the fluid in the back of the eye.
10
Competitive Product Candidates by Indication
We believe the primary competitors by indication with respect to our current programs in late stage-clinical testing are as follows:
Competitive Pharmaceuticals by Indication
| |
Indication | Competitive Products |
Dry Eye Disease | Topical immunomodulators, such as cyclosporine (0.05% as Restasis® or 0.09% as CequaTM) and lifitegrast (5% as Xiidra®); topical corticosteroids, such as loteprednol (0.25% as EysuvisTM); an intranasal spray, such as varenicline (0.03mg as TyrvayaTM), and other generic steroids; and artificial tear solutions |
| |
Allergic Conjunctivitis | Topical antihistamines and corticosteroids, which may be generic or sold over-the-counter; nonsteroidal anti-inflammatory drugs (NSAIDs); and mast cell stabilizers |
| |
Proliferative Vitreoretinopathy | Compounded intravenous methotrexate that is injected into the eye |
We believe that there is significant unmet medical need for the diseases that we intend to target. If proven to be safe and effective, we believe that our product candidates could be used in place of, or in addition to, current therapies. Currently available therapies for the treatment of DED are generally considered by physicians and patients to be inadequate, may require weeks or months of treatment to achieve even moderate clinical benefit, and have not demonstrated clinical activity in AC, a common comorbidity. There is no approved therapy for PVR or PVRL.
Many drugs are in development for AC and DED. In addition, generic versions of Restasis® became available in the U.S. in March 2022. We believe that there are currently no drugs in development for both DED and AC; PVR; or PVRL. For the diseases we intend to study, there may be other developmental therapies of which we are not aware.
Our ability to compete successfully will depend in part on our ability to utilize our drug development expertise to identify, develop, secure rights to, and obtain regulatory approvals for promising pharmaceutical products before others are able to develop competitive products. Our ability to compete successfully will also depend on our ability to attract and retain skilled and experienced personnel. Additionally, our ability to compete may be diminished by insurers and other third-party payors, which often encourage the use of cheaper, non-innovative, or generic products.
Clinical Trial Results and Development Plans
Prior to applying for marketing approval, our product candidates must satisfy regulatory authority requirements for safety and efficacy, including pivotal Phase 3 clinical assessment. Our active clinical programs with reproxalap have consistently demonstrated statistically and clinically significant efficacy, and have advanced to late-stage clinical testing. In addition, reproxalap has been observed to be well-tolerated and reported adverse events were generally mild in our clinical trials to date. Our material clinical results have been previously disclosed elsewhere in detail, and we encourage review of all of our clinical trial disclosures. All of our development plans and timelines are subject to adjustment depending on recruitment rate, regulatory review, preclinical and clinical results, funding, and other factors that could delay the initiation, completion, or reporting of clinical trials. As a result of the COVID-19 pandemic, clinical site availability, staffing, patient recruitment, and patient retention have been negatively affected and the timelines to complete our clinical trials may be delayed.
11
Dry Eye Disease
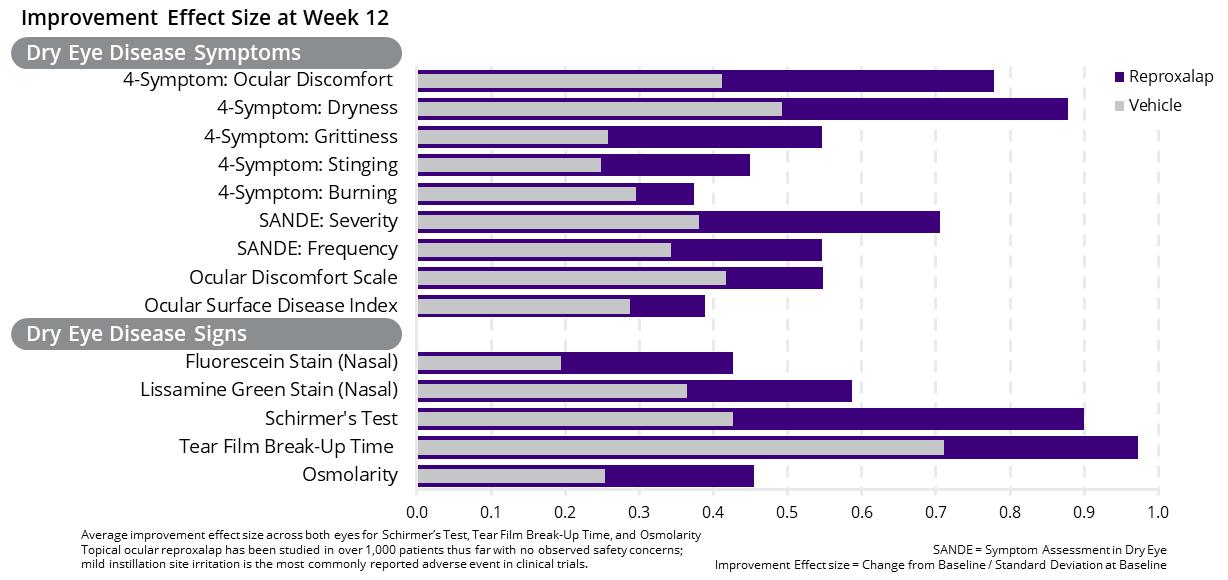
In September 2017, we announced that the results of a randomized, parallel-group, double-masked Phase 2a clinical trial of 0.1% and 0.5% topical ocular reproxalap demonstrated statistically and clinically relevant improvement from baseline in multiple signs and symptoms associated with dry eye disease in as soon as one week. In September 2018, we announced that the results of a randomized, vehicle-controlled, parallel-group, multi-center, double-masked Phase 2b clinical trial of 0.1% and 0.25% topical ocular reproxalap demonstrated statistically significant improvement over vehicle in ocular signs and symptoms associated with DED (Figure 1). Relative to patients treated with vehicle, patients treated with the 0.25% concentration of reproxalap demonstrated statistically significant and clinically relevant reductions in the Four-Symptom Ocular Dryness Score and the Overall Ocular Discomfort Symptom Score. For drug-treated patients, improvement greater than that of vehicle was consistently observed across all symptoms, and activity versus vehicle was evident as early as two weeks, the first assessment following initiation of therapy. The early onset of symptomatic improvement is consistent with the Phase 2a clinical trial of topical ocular reproxalap in DED, and is supportive of a differentiated product profile relative to standard of care. Patients treated with the 0.25% concentration of reproxalap also demonstrated reductions in ocular fluorescein staining score that were statistically superior to those of patients treated with vehicle.
Figure 1: Phase 2b Dry Eye Disease Clinical Trial Results for 0.25% Reproxalap
In April 2019, we initiated the RENEW Trial, an adaptive, two-part, multi-center, randomized, vehicle-controlled, double-masked, parallel-group Phase 3 trial of 0.25% topical ocular reproxalap compared to vehicle in patients with moderate to severe DED. The primary objective of RENEW Part 1 was to confirm dosing regimen, endpoints, and sample size for RENEW Part 2. In Part 1 of RENEW, 422 patients were randomized equally to receive either four-times-daily reproxalap or vehicle for twelve weeks (the constant dosing group) or four-times-daily reproxalap or vehicle for four weeks, followed by twice-daily reproxalap or vehicle for eight weeks (the induction-maintenance dosing group). In December 2019, we announced that, in the induction-maintenance dosing group, the RENEW co-primary endpoint of patient-reported visual analog scale (VAS) ocular dryness from Weeks 2 to 12 was achieved (p=0.0004) (Figure 2), and that activity was observed as early as one week after initiation of therapy (p=0.001) and was maintained until the end of the trial.
12
Figure 2: RENEW Part 1 Results – Co-Primary Symptom Endpoint for the Induction Maintenance Dosing Regimen
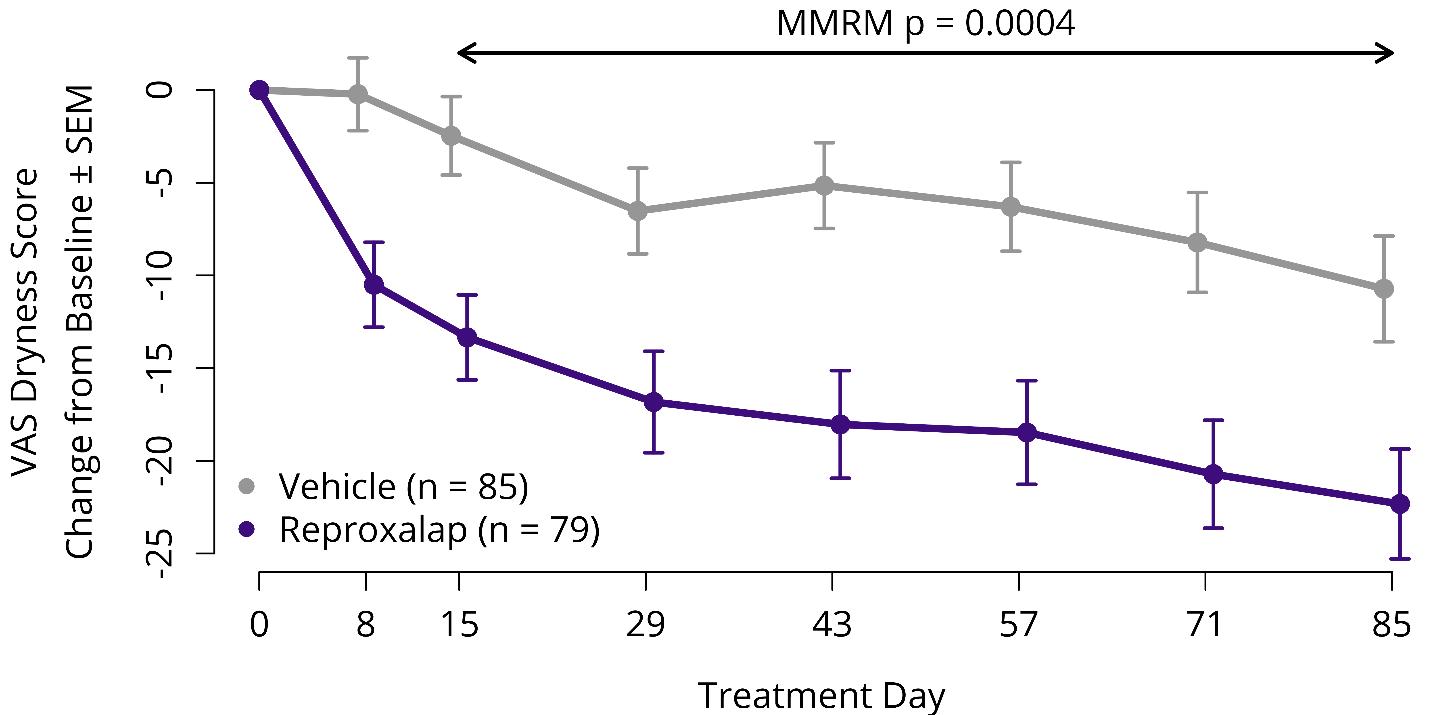
SEM = standard error of the mean; MMRM = mixed model repeated measures
In the induction-maintenance dosing group from Weeks 2 to 12, reproxalap was statistically superior to vehicle in VAS ocular endpoints for itching (p=0.03), foreign body sensation (p=0.004), discomfort (p=0.003), photophobia (p=0.004), and pain (p=0.03) (Figure 3). In the induction-maintenance dosing group from Weeks 2 to 12, reproxalap was also statistically superior to vehicle in Ocular Discomfort & 4-Symptom Questionnaire ocular endpoints for dryness (p=0.01), discomfort (p=0.03), burning (p=0.03), grittiness (p=0.003), and stinging (p=0.02). Although the improvement effect size of the co-primary endpoint of fluorescein nasal region ocular staining did not reach statistical significance, reproxalap was statistically superior to vehicle in reduction from baseline in the induction-maintenance dosing group from Weeks 1 to 4 of treatment (p=0.03), and statistical separation from vehicle was observed at Week 2 (p=0.04). Given that the co-primary symptom endpoint was achieved in RENEW Part 1, the RENEW Trial was concluded and RENEW Part 2 was not performed.
13
Figure 3: RENEW Part 1 Results – Symptom Treatment Differences Over Weeks 2 to 12 for the Induction Maintenance Dosing Regimen
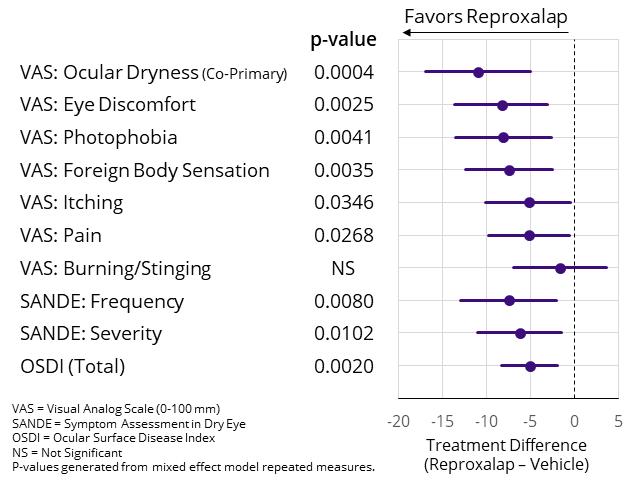
In February of 2020, we announced that the results of a randomized, vehicle-controlled, parallel-group, multi-center, double-masked Phase 2 clinical trial of a novel formulation of 0.25% topical ocular reproxalap demonstrated statistically significant improvement over vehicle for the primary endpoint of VAS ocular dryness score (p=0.03). Consistent with the RENEW Trial, the drug was administered via the induction maintenance regimen. The novel formulation is substantially similar to the formulations used in the Phase 2b clinical trial and the RENEW Trial, differing only in the amount of a single excipient.
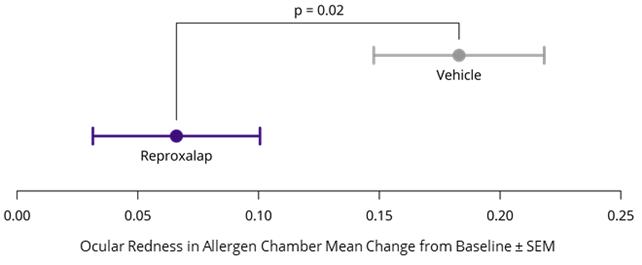
In January 2021, we announced positive top-line symptom, redness, and Schirmer test results from the run-in cohort of the Phase 3 TRANQUILITY clinical trial of 0.25% topical ocular reproxalap in patients with DED. Patients were treated with four doses of reproxalap or vehicle for one day. On the following day, patients were administered a single dose before entry, and a dose 45 minutes after entry, to a dry eye chamber with minimal humidity, high airflow, and forced visual tasking. The run-in cohort, performed at a single center, was double-masked, and randomized 12 patients to receive reproxalap and 11 patients to receive vehicle. Over all time points in aggregate in the dry eye chamber, reproxalap demonstrated statistically significant improvement over vehicle (p=0.03) in ocular redness, an objective sign of dry eye disease.
In November 2021, we announced achievement of the primary endpoint of ocular redness in a randomized, double-masked, vehicle-controlled Phase 2 clinical trial of 0.25% topical ocular reproxalap in DED patients. The protocol of the trial was substantially similar to that of the TRANQUILITY run-in cohort. The primary endpoint of the trial was achieved: ocular redness scores in the reproxalap group during the dry eye chamber were observed to be statistically lower than those of vehicle (p = 0.016, Figure 4). Additionally, Schirmer test scores assessed after the first dose were directionally in favor of reproxalap over vehicle and approached statistical significance (p = 0.068).
14
Figure 4: Phase 2 Dry Eye Chamber Trial Results– Ocular Redness
In December 2021, we announced top-line results from the randomized, double-masked, vehicle-controlled Phase 3 TRANQUILITY Trial of 0.25% topical ocular reproxalap in DED. Although the primary endpoint of ocular redness was not met, statistical significance (p=0.0001, Figure 5) was achieved for the DED sign of Schirmer test, a secondary endpoint, in favor of reproxalap over vehicle. Statistical significance (p<0.0001) in favor of reproxalap over vehicle was also achieved for the post-hoc clinical relevance assessment of proportion of Schirmer test responders that improved from baseline scores (0 to 30 mm) by at least 10 mm.
Figure 5: Phase 3 TRANQUILITY Trial Results– Schirmer Test
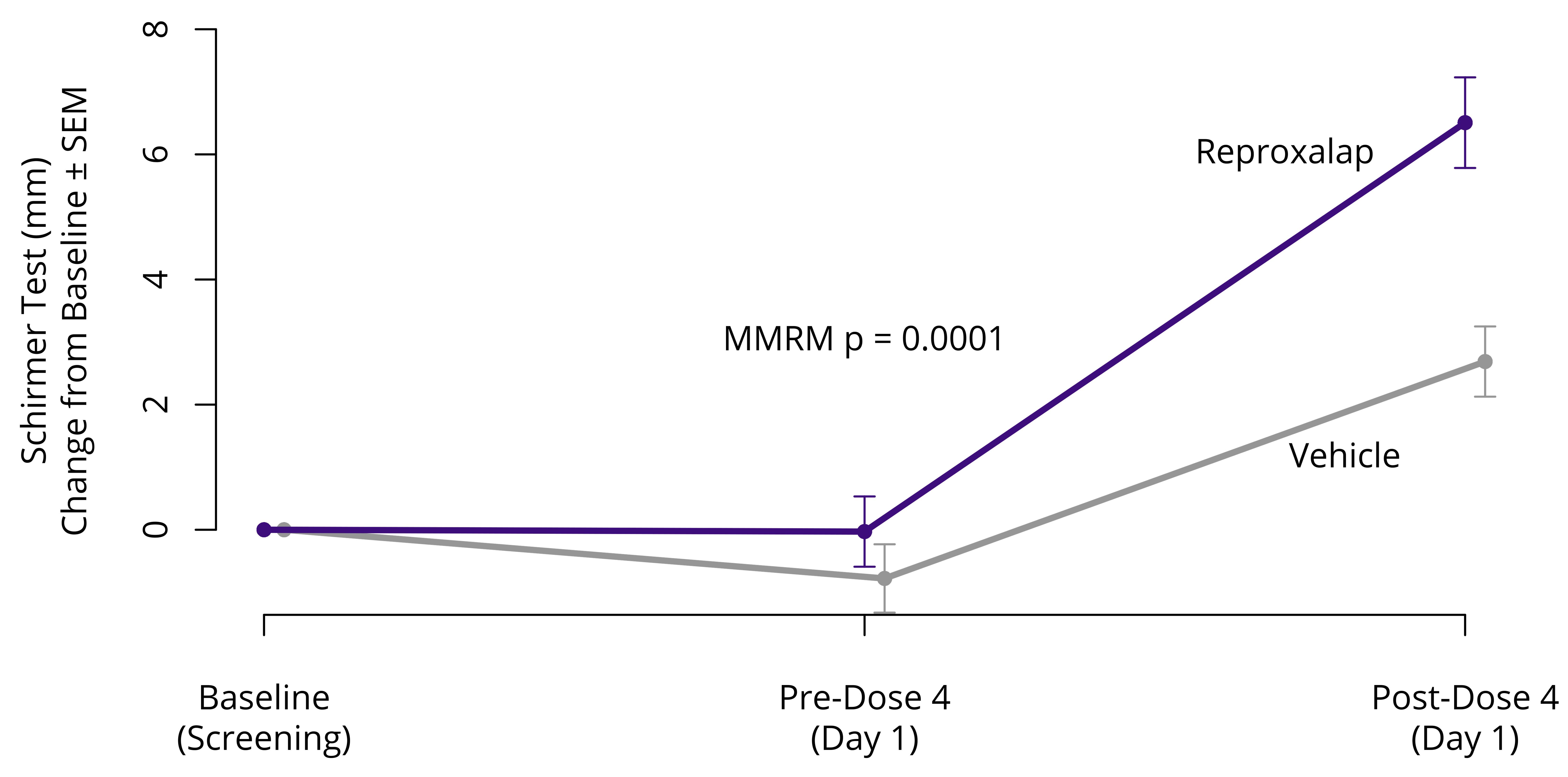
SEM = standard error of the mean; MMRM = mixed model repeated measures
In addition to the TRANQUILITY Trial, we have initiated the TRANQULITY-2 Trial, the protocol of which is substantially similar to that of TRANQUILITY. Using an alpha sharing technique, the primary endpoint of the TRANQUILITY-2 Trial will be met if either Schirmer test or ocular redness is statistically significant in favor of reproxalap over vehicle. Top-line results from TRANQUILITY-2 are expected mid-2022.
Per draft FDA guidance, to be considered for regulatory approval in the United States, a product candidate for the treatment of DED must demonstrate efficacy in a subjective symptom (a patient-reported subjective measure of disease severity) in at least two adequate and well-controlled clinical trials, and efficacy in an objective sign (an independently assessed objective measure of disease severity) in at least two adequate and well-controlled clinical trials. We intend to submit the RENEW Part 1 and Phase 2 novel formulation trials to satisfy the symptom efficacy NDA requirements. Because either ocular redness or Schirmer test qualifies as a sign of DED, subject to a
15
discussion with the FDA, we intend to submit a combination of the Phase 2, TRANQUILITY, and TRANQUILITY-2 chamber trials to satisfy the NDA sign requirements, depending on whether ocular redness or Schirmer test is achieved in TRANQUILITY-2 or in subsequent clinical trials. If ocular redness and Schirmer test is statistically significant in TRANQUILITY-2, then we may elect to submit the NDA with two sign trials in addition to the symptom trials. Pending results from TRANQUILITY-2, a dry eye disease safety trial, and regulatory input, we believe that an NDA filing for reproxalap for the treatment of DED could occur mid-2022.
In all reproxalap DED clinical trials completed to date, no clinically significant safety findings were observed, and reproxalap was well tolerated. Transient and generally mild instillation site discomfort is the most commonly reported adverse event.
Allergic Conjunctivitis
In February 2016, we announced that the results of a randomized, parallel-group, double-masked, vehicle-controlled conjunctival challenge (allergen administered directly to the eye following a single dose of test article) Phase 2a clinical trial of 0.5% topical ocular reproxalap in patients with AC demonstrated statistically and clinically significant activity of reproxalap over vehicle in reducing patient-reported ocular itching score (scale 0 to 4), a clinical endpoint that has served as the basis for prior AC FDA approvals. In June 2017, we announced that the results of a randomized, parallel-group, double-masked, vehicle-controlled, multi-center conjunctival challenge Phase 2b clinical trial of 0.1% and 0.5% topical ocular reproxalap in patients with AC demonstrated statistically and clinically significant activity of reproxalap over vehicle in reducing ocular itching.
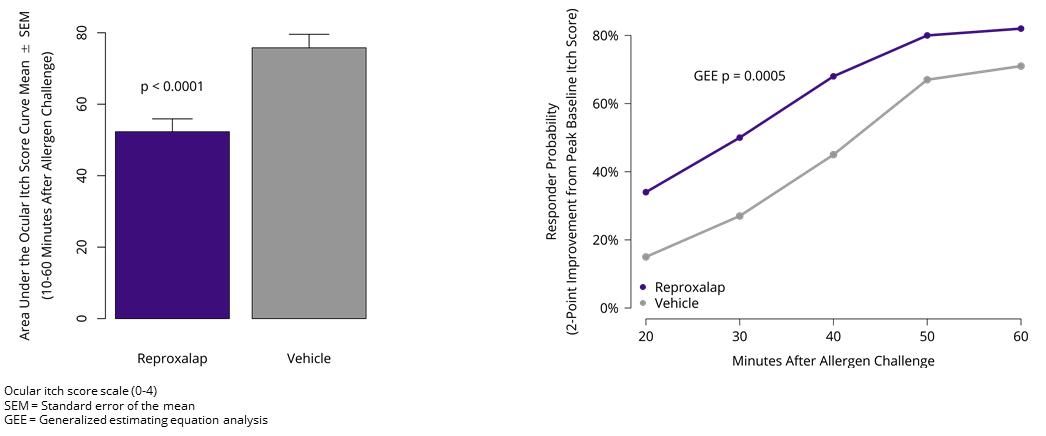
In March 2019, we announced that the randomized, double-masked, vehicle-controlled conjunctival challenge Phase 3 ALLEVIATE clinical trial of 0.25% and 0.5% topical ocular reproxalap in patients with AC met the primary endpoint (patient-reported ocular itching score area under the curve from 10 to 60 minutes after allergen challenge, p<0.0001) and the key secondary endpoint (proportion of patients that improved by two or more points on the four-point itching score, p=0.0005) for 0.25% reproxalap (Figure 6).
Figure 6: Phase 3 ALLEVIATE Trial Results for 0.25% Reproxalap
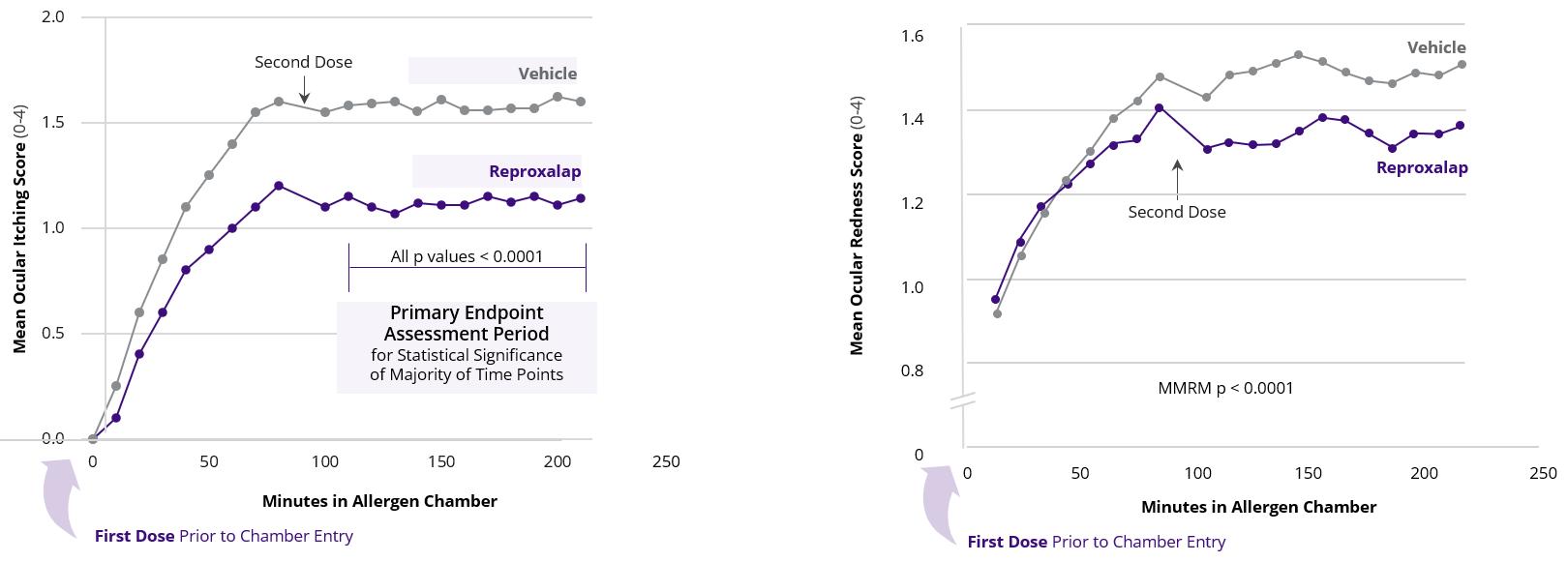
In October 2019, we announced that our randomized, double-masked, vehicle-controlled allergen chamber (aerosolized pollen over 3.5 hours with test article dosing just before and 90 minutes after chamber entry) clinical methods trial of topical ocular reproxalap in patients with AC demonstrated that, relative to patients treated with vehicle, patients treated with 0.25% or 0.5% topical ocular reproxalap demonstrated statistically significant reduction in patient-reported ocular itching (p<0.0001), investigator-assessed ocular redness (p<0.0001), and patient-reported tearing score (p<0.0001). In April 2021, we announced that the randomized, double-masked, vehicle-controlled allergen chamber Phase 3 INVIGORATE clinical trial of 0.25% reproxalap in patients with AC
16
(Figure 7) achieved the primary endpoint (patient-reported ocular itching score after the second dose of test article) and all secondary endpoints (investigator-assessed ocular redness score and patient-reported ocular tearing score).
Figure 7: Phase 3 INVIGORATE Trial Results for 0.25% Reproxalap) - Ocular Itching
In 2022, we initiated the randomized, double-masked, vehicle-controlled allergen chamber Phase 3 INVIGORATE-2 Trial of 0.25% topical ocular reproxalap in patients with AC. Pending positive results from INVIGORATE-2, an allergic conjunctivitis safety trial, and regulatory input, we believe that we will have met NDA requirements for AC, although we expect that the NDA submission for AC would occur after the NDA submission for DED. In all reproxalap allergic conjunctivitis clinical trials completed to date, no clinically significant safety findings were observed, and reproxalap was well tolerated. Transient and generally mild instillation site discomfort is the most commonly reported adverse event.
Proliferative Vitreoretinopathy
Standard of care treatment for PVR results in subsequent retinal detachment surgical rates that approach 40%. In a single-arm, open-label, investigator-sponsored Phase 1b clinical trial performed at the Massachusetts Eye and Ear Infirmary, only approximately 20% of patients with PVR treated with multiple injections of ADX-2191 required subsequent surgery for retinal detachment (Figure 8). Thus, relative to standard of care, ADX-2191 may reduce incidence of retinal detachment following the development of PVR, thereby increasing the probability of preservation of visual function.
17
Figure 8: Phase 1b ADX-2191 Investigator-Sponsored Clinical Trial
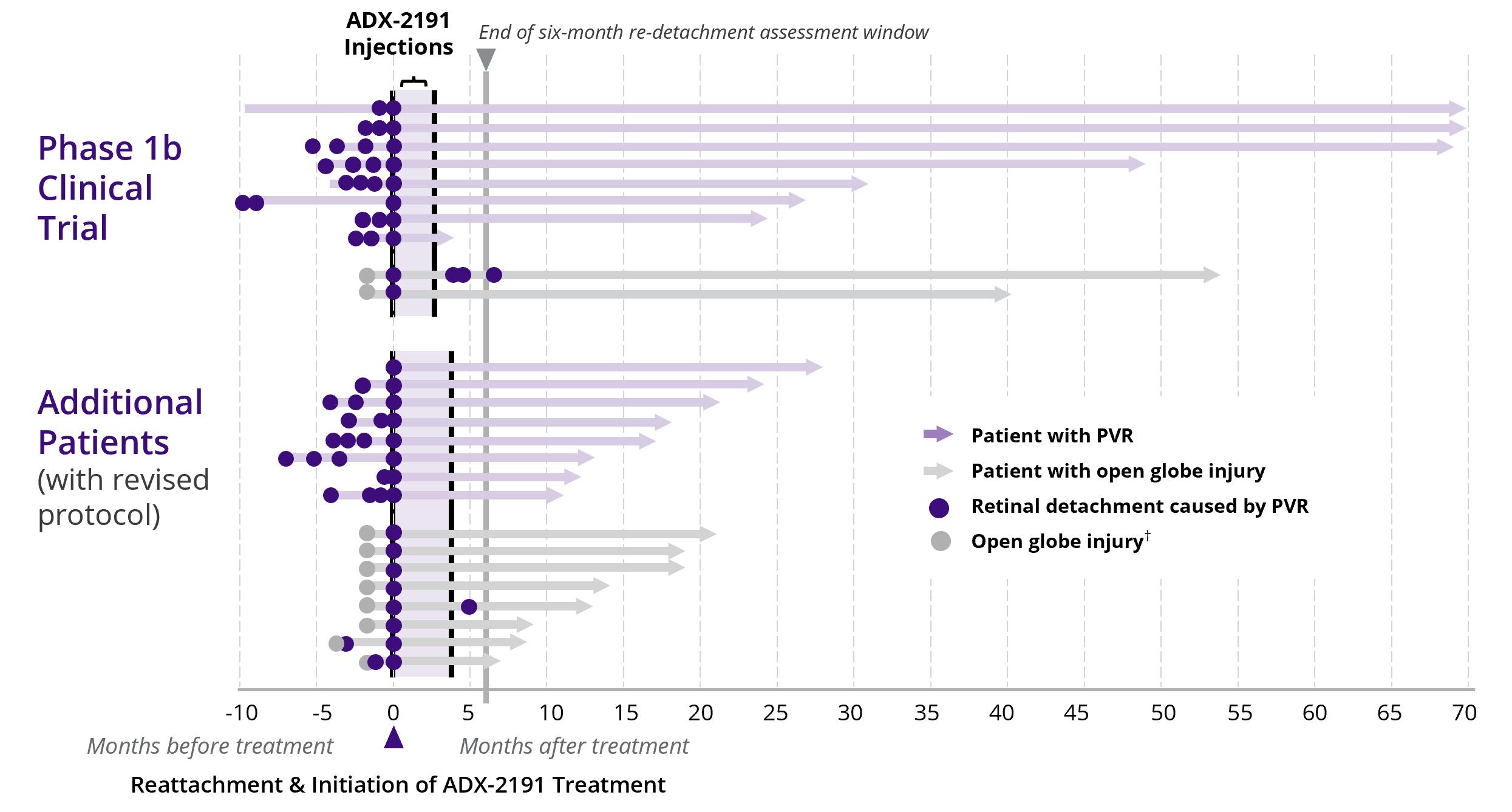
In December 2019, we initiated the Phase 3 GUARD trial, a two-part, multi-center, randomized, controlled, adaptive Phase 3 clinical trial evaluating the efficacy of intravitreal injections of ADX-2191 versus standard-of-care for the prevention of PVR. The primary endpoint of GUARD Part 1 is retinal detachment rate over six months following initiation of therapy relative to historical detachment rates. In January 2022, we announced completion of enrollment in Part 1 of GUARD, and we expect to announce results in the second half of 2022.
Systemic RASP Modulation for the Treatment of Immune-Mediated Diseases
In April 2020, we announced the results of a Phase 1 clinical trial of ADX-629, a first-in-class orally administered RASP modulator. No treatment-related adverse events were observed at any dose tested, and target engagement was evidenced by statistically lower RASP levels in drug-treated subjects relative to controls (Figure 10). Additionally, following ingestion of a controlled high-fat meal, free fatty acids were statistically lower and HDL statistically higher in drug-treated subjects relative to placebo-treated subjects. In December 2020, we announced the initiation of three Phase 2 proof-of-concept clinical trials of ADX-629 in patients with atopic asthma, psoriasis, or COVID-19 infection as part of a systematic strategy to assess activity across different types of immunological diseases. Following completion of the Phase 2 clinical trials, subsequent clinical indications may be selected, depending on safety and tolerability, clinical signals, cytokine profiles, and RASP levels.
18
Figure 9: Phase 1 Clinical Trial of ADX-629
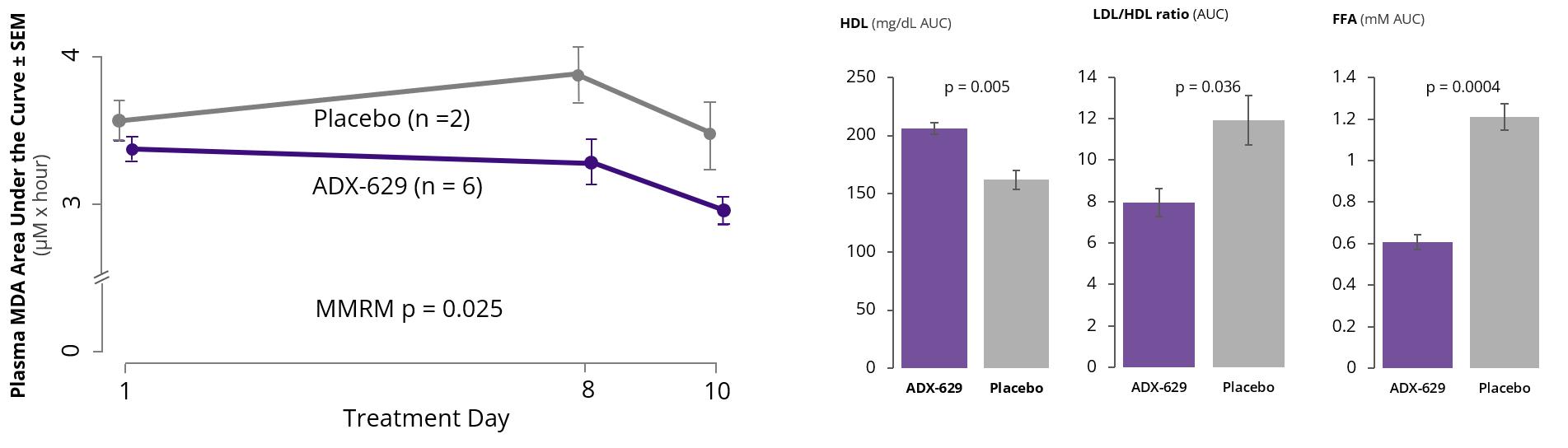
MDA = malondialdehyde; SEM = standard error of the mean; MMRM = mixed model repeated measures; HDL= high-density lipoprotein; LDL = low-density lipoprotein; FFA = free fatty acids
The Science Supporting Our Product Candidates
Reactive Aldehyde Species
In response to infection, injury, endogenous and exogenous chemical triggers, heat, and other stimuli, pro-inflammatory RASP are generated through a variety of metabolic processes, including alcohol oxidation, enzymatic and non-enzymatic lipid oxidation, and polyamine and sphingosine metabolism. RASP appear to effect inflammation signaling via covalent binding to thiol (sulfur-containing) and amine (nitrogen-containing) residues on proteins, including receptors and enzymes. RASP-protein adducts directly influence the function of proteins, leading to activation of intracellular inflammatory factors, including NF-κB, an important mediator in the inflammatory response, and inflammasomes. In addition, RASP adducts bind to Scavenger Receptor A, which also initiates pro-inflammatory signaling and leads to the formation of antibodies against the adducted protein, at least in part explaining the presence of host-directed antibodies in autoimmune diseases such as rheumatoid arthritis. Levels of RASP are generally observed to be elevated in ocular and systemic inflammatory disease, and thus represent therapeutic targets for immune modulation.
Because of the inherent toxicity of RASP, most, if not all, living organisms contain enzymes, such as aldehyde reductases and aldehyde dehydrogenases, that convert RASP into non-toxic molecules. Genetic mutations in the RASP-metabolizing enzymes cause disease. In Sjögren-Larsson Syndrome, for example, mutations in fatty aldehyde dehydrogenase are responsible for skin, neurological, and retinal disease.
Aside from the stimulation of inflammation, there is no generally accepted biological role of high levels of RASP. Some physiologic molecules have RASP forms, including retinaldehyde (a form of Vitamin A) and pyridoxal and pyridoxal phosphate (forms of Vitamin B6), but the activity of physiological RASP is highly restricted by chaperone and other proteins that prevent reaction with other molecules, including our RASP modulators. Thus, pharmacotherapeutic RASP modulation is expected not to adversely affect normal physiologic processes. Consistent with the lack of accessibility of physiologic RASP, our most advanced RASP modulator, reproxalap, which has been administered to approximately 1,500 patients across a number of completed clinical trials, has been observed to be generally well tolerated and has not resulted in any serious adverse events.
The RASP Modulator Platform
The RASP modulator platform represents a unique and novel pharmacologic approach that, unlike almost all drugs in use today, is not designed to directly inhibit or activate a particular protein but instead targets a family of small molecules that in turn affect the activity and structure of many proteins at once. RASP modulation, therefore, has the potential to down-regulate pro-inflammatory systems or groups of proteins, and may lead to multiple beneficial clinical effects while avoiding toxicity associated with single-target inhibition or activation.
19
We are currently developing reproxalap, a new chemical entity, and other novel RASP modulators for the treatment of immune-mediated disease. Reproxalap is a small molecule designed specifically to bind, and thereby allow for the degradation of, RASP. In in vitro and animal studies, reproxalap does not appear to affect most cellular components, including most receptors, enzymes, ion channels, or other proteins. Reproxalap has been shown to outcompete cellular constituents to covalently bind and trap RASP. Reproxalap-RASP adducts appear to be rapidly degraded in cellular environments, after which neither reproxalap nor RASP are detectable. Outside of biological systems, reproxalap-RASP adducts have shown to be remarkably non-reactive and stable, suggesting that reproxalap-RASP binding may be effectively irreversible. By forming covalent drug-RASP adducts that are then degraded, reproxalap and other RASP modulators have the potential to substantially lower RASP levels.
We believe we have been the first to demonstrate the beneficial effects of RASP modulation in a variety of animal models relating to immune-mediated disease, suggesting that reproxalap and analogs may have potent anti-inflammatory effects that persist hours after administration at a variety of different doses relevant to clinical testing.
•In mouse models of ocular inflammation and post-surgical healing, topically applied reproxalap ophthalmic solution reduced ocular redness and inflammatory cytokines comparable to corticosteroid therapy and slowed the development of corneal haze (fibrosis). (Data presented at the Association for Research in Vision and Ophthalmology 2015 Annual Meeting)
•In mice injected with a pro-inflammatory agent known as endotoxin, intraperitoneally administered reproxalap statistically reduced a variety of inflammatory cytokines (protein inflammatory mediators), including IL-5, Il-1ß, IL-17, and TNF-α, while up-regulating the primary anti-inflammatory cytokine, IL-10. Additionally, in models of mouse contact dermatitis (induced by phorbol myristate acetate) and allergic contact dermatitis (induced by sensitivity to oxazolone), reproxalap statistically reduced inflammation as measured by edema (swelling). (Data presented at the American Academy of Asthma Allergy and Immunology 2015 Annual Meeting)
•In a model of radiation mucositis (oral inflammation) in hamsters, chronic subcutaneous administration of reproxalap reduced healing time and decreased fibrosis (scarring). (Data presented at the Multinational Association of Supportive Care in Cancer – International Society of Oral Oncology 2015 Annual Meeting)
•In two different mouse models of inflammatory pain, intraperitoneally administered reproxalap dose-dependently reduced nociceptive behavior, suggesting that reproxalap down-regulates pain signaling in inflammation. (Data presented at the 2016 International Conference on Pain Research and Management)
•In rat cardiomyocyte culture, reproxalap prevented fibrotic transformation, and inhibited NF-κB activation and IL-1ß release. (Data presented at the 2016 American Society for Cell Biology Annual Meeting)
•In a mouse model of lung inflammation, intraperitoneal administration of reproxalap reduced infiltration of inflammatory cells and levels of pro-inflammatory cytokines in the lung. (Data presented at the 2017 World Congress on Inflammation Annual Meeting)
•In a rat model of intraocular inflammation, a single intravitreal injection of ADX-103 reduced the development of retinal pathology. (Data presented at the Association for Research in Vision and Ophthalmology 2018 Annual Meeting)
20
•In a rat model of diabetic macular edema, intravitreal injection of ADX-103 reduced retinal inflammatory cell infiltration. (Data presented at the Association for Research in Vision and Ophthalmology 2018 Annual Meeting)
Thus, we believe that the immune-modulating mechanism of action of RASP modulation is potentially multifactorial – lowering inflammation, reducing healing time, diminishing scarring, and mitigating inflammatory pain – and may ameliorate inflammatory disease and deter disease progression in different ways simultaneously.
In addition to the development of reproxalap, we intend to continue the discovery and development of other novel RASP modulators, and we intend to continue to develop intellectual property around such molecules. We have identified, synthesized, and tested numerous molecules that may be more potent than reproxalap in binding to RASP. We are currently screening novel product candidates to address diseases where topical and systemic administration may reduce RASP-mediated pathology. Clinical development of ADX-629 for the treatment of autoimmune and other inflammatory diseases began in late 2019.
The Potential of ADX- 2191 to Prevent Proliferative Vitreoretinopathy and Treat Primary Vitreoretinal Lymphoma
PVR is characterized by excessive replication and pro-inflammatory activity of retinal cells, at least a portion of which synthesize collagen, the principal component of scar tissue. Retinal scarring can lead to impairment of vision, including blindness, as well as increased probability of further retinal detachments. Methotrexate, the active component of ADX-2191, is a dihydrofolate reductase inhibitor that has been used to treat cancer and autoimmune disease. The anti-proliferative and anti-inflammatory properties of dihydrofolate reductase inhibition are well described. In preclinical studies of primary cell cultures from PVR patients, dihydrofolate reductase inhibition reduced pathological cell proliferation and scar-like collagen deposition. Thus, the observed clinical activity of ADX-2191 in PVR is believed to be the result of down-regulation of aberrant retinal cell proliferation and activity, thereby leading to reduced retinal scarring. Initially as an intravenous medication and subsequently as an intraocular injection, methotrexate has also been used for decades as an anti-proliferative medication to treat PVRL. The goal of methotrextate therapy in PVRL is to prevent metastasis to the central nervous system.
Intellectual Property and Proprietary Rights
Overview
In the United States and abroad, we are building an intellectual property portfolio for reproxalap and other RASP modulators and for therapeutic methods of use of methotrexate for the treatment of retinal disease. We currently seek, and intend to continue to seek, patent protection in the United States and internationally for our product candidates, methods of use, and processes for manufacture, and for other technologies, where appropriate. Our current policy is to actively seek to protect our proprietary position by, among other things, filing patent applications in the United States and abroad relating to proprietary technologies that are important to the development of our business. We also rely on, and will continue to rely on, trade secrets, know-how, continuing technological innovation and in-licensing opportunities to develop and maintain our proprietary position. We cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents that may be granted to us in the future will be commercially useful in protecting our technology.
Our success will depend significantly on our ability to obtain and maintain patent and other proprietary protection for the technologies that we consider important to our business, our ability to defend our patents, and our ability to preserve the confidentiality of our trade secrets and operate our business without infringing the patents and proprietary rights of third parties.
21
Patent Portfolio
Our patent portfolio currently includes patents and patent applications covering the composition, formulation, and uses of reproxalap, ADX-103, ADX-629, ADX-1612, and other novel compounds. As of December 31, 2021, we owned fifteen United States patents and twelve pending United States non-provisional patent applications, as well as numerous foreign counterparts to these patents and patent applications, relating to reproxalap and other RASP modulators. Additionally, we have in-licensed certain patents and patent applications relating to ADX-1612, and retain an exclusive license to certain patents related to the use of ADX-2191 for the prevention or treatment of a number of retinal indications including, proliferative vitreoretinal disease and primary vitreoretinal lymphoma.
We expect the issued reproxalap composition of matter patent in the United States, if the appropriate maintenance, renewal, annuity or other governmental fees are paid, to expire in 2028. It is possible that the term of the composition of matter patent in the United States may be extended up to five additional years under the provisions of the Hatch-Waxman Act. We expect the foreign reproxalap composition of matter patents, if the appropriate maintenance, renewal, annuity or other governmental fees are paid, to expire in 2026. We expect other patent applications in the portfolio, if issued, and if the appropriate maintenance, renewal, annuity or other governmental fees are paid, to expire from 2026 to 2034. Reproxalap composition of matter patents have been issued in Australia, Canada, China, Europe (validated in approximately 14 member countries), Hong Kong, India, Japan, Mexico, Russia and South Korea. Reproxalap composition of matter patent claims are pending in Brazil.
Licenses and Agreements
Madrigal Agreement
We are developing ADX-1612 pursuant to a License Agreement with Madrigal Pharmaceuticals, Inc. (Madrigal), entered into on December 26, 2016 (Madrigal Agreement). Pursuant to the Madrigal Agreement, we obtained an exclusive, worldwide license from Madrigal under certain patents and patent applications, and other licenses to intellectual property, to develop and commercialize CHP inhibitors, including ADX-1612 and other molecules (Madrigal Agreement Products). We have agreed to use our commercially reasonable efforts to develop Madrigal Agreement Products.
In consideration for the rights licensed under the Madrigal Agreement, we paid Madrigal an upfront license fee of $250,000 and are obligated to make future regulatory and development and sales-dependent milestone payments to Madrigal of less than $340 million in the aggregate (over 80% of such amount being tied to our achievement of increasingly greater annual worldwide net sales milestones), as well as royalty payments to Madrigal at a rate which, as a percentage of net sales, is in the high single digits for products containing ADX-1612 and mid-single digits for any other CHP inhibitor product. We are also obligated under the Madrigal Agreement to pay Madrigal a percentage of certain sublicense revenue that we receive in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from the mid-twenties to low-single digits based on the development stage of the product at the time of the sublicense.
The Madrigal Agreement will remain in effect until all payment obligations under the Madrigal Agreement expire. We may terminate the Madrigal Agreement in its entirety or on a Madrigal Agreement Product-by-Madrigal Agreement Product basis with timely notice to Madrigal. Either party may terminate the Madrigal Agreement for uncured material breach by the other party or upon certain insolvency or bankruptcy proceedings involving the other party, both with timely notice to the other party. In addition, Madrigal has the right to terminate the Madrigal Agreement if we, our affiliates, or sublicensees interfere with, challenge the validity or enforceability of, oppose the extension of, or grant of a supplementary protection certificate with respect to any of our licensed patents under the Madrigal Agreement. In the event of an early termination of the Madrigal Agreement, all rights licensed and developed by us under the Madrigal Agreement may revert back to Madrigal. Each party has agreed to indemnify the other party for certain third party claims arising under the Madrigal Agreement.
22
MEEI Agreement
We are developing ADX-2191 pursuant to an Exclusive License Agreement with Massachusetts Eye and Ear Infirmary (MEEI) originally entered into in July 2016 between MEEI and Helio Vision, Inc., as amended, (MEEI Agreement). We assumed the MEEI Agreement in connection with our 2019 acquisition of Helio Vision.
Pursuant and subject to the MEEI Agreement, we obtained an exclusive, worldwide license from MEEI to develop and commercialize ADX-2191 under certain patents and patent applications, and other licenses to intellectual property (MEEI Patent Rights). We have agreed to use our commercially reasonable efforts to develop ADX-2191 and to meet certain specified effort and achievement benchmarks by certain dates.
In consideration for the rights licensed under the MEEI Agreement, Helio Vision issued MEEI a number of shares of its preferred stock and Helio Vision agreed to pay non-creditable non-refundable license maintenance fees to MEEI of $15,000 on each of the second and third anniversary of the MEEI Agreement, $25,000 on each of the fourth and fifth anniversary of the MEEI Agreement and $35,000 on the sixth and each subsequent anniversary of the MEEI Agreement during the term of such agreement. In addition, Helio Vision was obligated to make future sales-dependent milestone payments to MEEI of up to the low seven figures in the aggregate, as well as royalty payments to MEEI at a rate which, as a percentage of net sales, is in the low single digits for products that incorporate or use the MEEI Patent Rights in the United States and as a percentage in the low single digits for products that incorporate or use the MEEI Patent Rights outside the United States. We are also obligated under the MEEI Agreement to pay MEEI a percentage of certain sublicense revenue that we receive in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from low-double digits to mid-single digits based on the date of the sublicense. Following our acquisition of Helio Vision, we became obligated to make any future payments owed under the MEEI Agreement. There is no additional equity consideration issuable under the MEEI Agreement.
The MEEI Agreement will remain in effect until the expiration date of the last to expire patent licensed under the MEEI Agreement. We may terminate the MEEI Agreement with timely written notice to MEEI. MEEI has the right to terminate the MEEI Agreement if we, subject to certain specified cure periods, cease all business operations with respect to licensed products, fail to pay amounts due under the MEEI Agreement, fail to comply with certain due diligence obligations, default in our obligation to maintain insurance, one of our officers is convicted of a felony relating to the manufacture, use, sale or importation of licensed products, we materially breach any provisions of the MEEI Agreement or in the event of our insolvency or bankruptcy.
In the event of an early termination of the MEEI Agreement, all rights licensed and developed by us under the MEEI Agreement may revert back to MEEI. We have agreed to indemnify MEEI for certain claims that may arise under the MEEI Agreement.
Other Intellectual Property Rights
Our marks ALDEYRA THERAPEUTICS and our logo are registered with the United States Patent and Trademark Office.
Confidential Information and Inventions Assignment Agreements
We currently require and will continue to require each of our employees and consultants to execute confidentiality agreements upon the commencement of such individual’s employment, consulting or collaborative relationships with us. These agreements provide that all confidential information developed or made known during the course of the relationship with us be kept confidential and not disclosed to third parties except in specific circumstances. In the case of employees, the agreements provide that all inventions resulting from such individual’s work performed for us, utilizing our property or relating to our business and conceived or completed by the individual during employment shall be our exclusive property to the extent permitted by applicable law. Our consulting agreements also provide for assignment to us of any intellectual property resulting from services performed by a consultant for us.
23
Manufacturing
We do not own or operate manufacturing facilities for the production of our product candidates, nor do we have plans to develop our own manufacturing operations in the foreseeable future. We currently depend on third-party contract manufacturers for all of our required raw materials, drug substance and finished drug product for our preclinical research and clinical trials. We have no immediate plans to purchase, erect, or otherwise create any manufacturing facilities to be owned by us for any of these purposes, and intend to continue to depend on third-party contract manufacturers for the foreseeable future. We do not have any current contractual relationships for the manufacture of commercial supplies of our product candidates. If our product candidates are approved by any regulatory agency, we intend to enter into agreements with third-party contract manufacturers for the commercial production at such time. We may utilize third-party consultants to manage our manufacturing contractors. We believe that the active pharmaceutical ingredient and other materials needed for the formulation of our product candidates are relatively easy to manufacture, and that multiple suppliers and formulators could be employed for this purpose. Further, we believe the raw materials needed for manufacture of our product candidates, as well as other components of our formulations, are generally readily available currently from multiple sources.
Employees
As of December 31, 2021, we had 12 full-time employees and had engaged a number of consultants. We expect that a number of consultants previously engaged in development of our product candidates will participate in ongoing clinical and manufacturing activities. None of our employees is represented by a labor union. We have not experienced any work stoppages, and we consider our relations with our employees to be good.
Human Capital
We recognize that attracting, motivating and retaining talent at all levels is vital to our continued success. Our employees are a significant asset and we aim to create an equitable, inclusive and empowering environment in which our employees can grow and advance their careers, with the overall goal of developing, expanding and retaining our workforce to support our current pipeline and future business goals. By focusing on employee retention and engagement, we also improve our ability to support our clinical trials, our pipeline, our platform technologies, business and operations, and also protect the long-term interests of our stockholders. Our success also depends on our ability to attract, engage and retain a diverse group of employees. Our efforts to recruit and retain a diverse and passionate workforce include providing competitive compensation and benefits packages and ensuring we listen to our employees.
24
We value innovation, passion, data-driven decision making, persistence and honesty, and are building a diverse environment where we believe that our employees thrive and are inspired to contribute to the development of novel therapies.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, motivating, and integrating our existing and future employees. The principal purpose of our incentive plans is to increase shareholder value by attracting, retaining, and motivating employees, consultants, and directors through grants of stock-based compensation awards and payments of cash-based performance bonus awards. We are committed to providing a competitive and comprehensive benefits package to our employees. Our benefits package is designed to meet the individual health and wellness needs of our employees. We plan to continue to refine our efforts related to optimizing our use of human capital as we grow, including improvements in the way we hire, develop, motivate, and retain employees.
Government Regulation
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Food Drug and Cosmetic Act (FDCA) and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Failure to comply with applicable FDA or other requirements may subject a company to a variety of administrative or judicial sanctions, such as the FDA’s refusal to approve pending applications, a clinical hold, warning letters, recall or seizure of products, partial or total suspension of production, withdrawal of the product from the market, injunctions, fines, civil penalties or criminal prosecution.
FDA approval is required before any new drug, new dosage form, new therapeutic use, or new route of administration of a previously approved product, can be marketed in the United States. The process required by the FDA before a new drug product may be marketed in the United States generally involves:
•completion of preclinical laboratory and animal testing and formulation studies in compliance with the FDA’s good laboratory practice (GLP) regulation;
•submission to the FDA of an Investigational New Drug application (IND) for human clinical testing which must become effective before human clinical trials may begin in the United States;
•approval by an independent institutional review board (IRB) at each site where a clinical trial will be performed before the trial may be initiated at that site;
•performance of adequate and well-controlled human clinical trials in accordance with current good clinical practices (cGCP) to establish the safety and efficacy of the proposed product candidate for each intended use;
•submission to the FDA of a new drug application (NDA) which must be accepted for filing by the FDA;
•satisfactory completion of an FDA pre-approval inspection(s) of our office and the facility or facilities at which the product is manufactured to assess compliance with the FDA’s current Good Manufacturing Practices (cGMP) regulations;
•satisfactory completion of an FDA advisory committee review, if applicable;
•payment of user fees, if applicable; and
•FDA review and approval of the NDA.
The preclinical and clinical testing and approval process requires substantial time, effort, and financial resources. Preclinical tests include laboratory evaluation of product chemistry, formulation, manufacturing and control procedures, and stability, as well as animal studies to assess the toxicity and other safety characteristics of the product. The results of preclinical tests, together with manufacturing information, analytical data, and a proposed clinical trial protocol and other information, are submitted as part of an IND to the FDA. Preclinical testing may
25
continue even after the IND is submitted. The IND becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions and places the clinical trial on a partial or complete clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, our submission of an IND may not result in FDA authorization to commence a clinical trial. A separate submission to an existing IND must also be made for each successive clinical trial conducted during product development. Even if the IND becomes effective and the trial proceeds without initial FDA objection, the FDA may stop the trial at a later time if, among other reasons, the potential for unacceptable safety risks arises.
Further, an independent IRB, covering each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial and informed consent information for subjects before the trial commences at that site and it must monitor the study until completed. The FDA, the IRB, or the sponsor may suspend a clinical trial at any time on various grounds, including a finding that the subjects or patients are being exposed to an unacceptable health risk or for failure to comply with the FDA’s or IRB’s requirements. Other conditions may also be imposed.
Clinical trials involve the administration of the investigational new product to human subjects under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide informed consent in writing for participation in the clinical trial. Sponsors of clinical trials generally must register and report, at the NIH-maintained website ClinicalTrials.gov, key parameters and results of certain clinical trials. For purposes of an NDA submission and approval, human clinical trials are typically conducted in the following sequential phases, which may overlap or be combined:
•Phase 1: The investigational drug product is initially introduced into healthy human subjects or patients and tested for safety, dose tolerance, absorption, metabolism, distribution, and excretion.
•Phase 2: The investigational drug product is administered to a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted indications, and to determine dose tolerance and optimal dosage. Multiple Phase 2 clinical trials may be conducted by the sponsor to obtain information prior to beginning larger and more extensive clinical trials.
•Phase 3: When Phase 2 evaluations suggest that certain dosing regimens may be efficacious and may have an acceptable safety profile, Phase 3 trials may be undertaken in larger patient populations to further evaluate dosage and to obtain evidence of potential clinical efficacy and safety. Phase 3 trials may include multiple, geographically-dispersed clinical trial sites. Data generated from these studies may be used to establish the overall risk-benefit profile of the investigational drug product and to provide adequate information for the labeling of the product, if approved.
•Phase 4: In some cases, the FDA may condition approval of an NDA for a product candidate on the sponsor’s commitment to conduct additional clinical trials to further assess the product’s safety and/or effectiveness after NDA approval. Such post-approval trials are typically referred to as Phase 4 trials.
The results of product development, preclinical studies and clinical trials are submitted to the FDA as part of an NDA. NDAs must also contain extensive information relating to the product’s pharmacology, chemistry, manufacturing and controls, and proposed labeling, among other things.
For some products, the FDA may require a risk evaluation and mitigation strategy (REMS) which could include measures imposed by the FDA such as prescribing restrictions, requirements for post-marketing studies, and reporting or certain restrictions on distribution and use. Under federal law, the submission of most NDAs is additionally subject to a substantial application user fee, and the manufacturer and/or sponsor under an approved NDA are also subject to prescription drug program fees. The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an NDA for filing. In this event, the NDA must be resubmitted with the additional information and is subject to payment of additional user fees. The resubmitted application is also subject to review before the FDA accepts it for filing.
26
Once the submission has been accepted for filing, the FDA begins an in-depth substantive review. Under the Prescription Drug User Fee Act (PDUFA), the FDA agrees to specific performance goals for NDA review time through a two-tiered classification system, Standard Review and Priority Review. Standard Review NDAs have a goal of being completed within a ten-month timeframe after acceptance of filing. A Priority Review designation is given to products that offer major advances in treatment or provide a treatment where no adequate therapy exists. The goal for completing a Priority Review is six months after acceptance of filing.
It is likely that our product candidates will be granted a Standard Review. The review process may be extended by the FDA for three additional months to consider certain information or obtain clarification regarding information already provided in the submission. The FDA may refer applications for novel products or products which present difficult questions of safety or efficacy to an advisory committee for review, evaluation, and recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it considers such recommendations carefully when making decisions. In addition, for combination products, the FDA’s review may include the participation of both the FDA’s Center for Drug Evaluation and Research, the Center for Biologics Evaluation and Research, and the FDA’s Center for Devices and Radiological Health. The participation of multiple distinct groups within the FDA has the potential to complicate or prolong review of the application. If the product is deemed a combination product, additional supporting studies may be required, and may delay an NDA submission.
Before approving an NDA, the FDA may inspect our offices and the facility or facilities where the drug substance or drug product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP. FDA may also inspect sponsor facilities to determine if nonclinical and clinical studies were conducted in compliance with applicable regulations and guidelines.
After the FDA evaluates the NDA and, in some cases, the related manufacturing facilities, it may issue an approval letter or a Complete Response Letter (CRL) to indicate that the review cycle for an application is complete and that the application is not ready for approval. CRLs generally outline the deficiencies in the submission and may require substantial additional testing or information in order for the FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when the deficiencies have been addressed to the FDA’s satisfaction, the FDA may issue an approval letter. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications.
Once issued, the FDA may withdraw product approval if ongoing regulatory requirements are not met or if potential adverse safety findings are identified after the product reaches the market. In addition, the FDA may require post-approval testing, including Phase 4 studies, and surveillance programs to monitor the effect of approved products which have been commercialized, and the FDA has the power to prevent or limit further marketing of a product based on the results of these post-marketing programs.
Products may be promoted only for the approved labeled indications and in accordance with the provisions of the approved label, and, even if the FDA approves a product, it may limit the approved indications for use for the product or impose other conditions, including labeling or distribution restrictions or other risk-management mechanisms, such as a Black Box Warning, which highlights a specific warning. Further, if there are any modifications to the product, including changes in indications, labeling, or manufacturing processes or facilities, a company would be required to submit and obtain FDA approval of a new or supplemental NDA, which may require the company to develop additional data or conduct additional preclinical studies and clinical trials.
Post-Approval Requirements
Once an NDA is approved, a product will be subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to product/facility listing, recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies, and are subject to periodic
27
unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and may require prior FDA approval before being implemented. FDA regulations may also require investigation and correction of any deviations from cGMP and may impose reporting and documentation requirements upon us and any third-party manufacturers. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated seriousness, severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
•restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
•fines, warning letters or holds on post-approval clinical trials;
•refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals;
•product seizure or detention, or refusal to permit the import or export of products; or
•injunctions or the imposition of civil or criminal penalties.
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. The FDA does not regulate the practice of medicine. Physicians may prescribe for off-label uses; manufacturers may only promote for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off label uses, and a company that is found to have improperly promoted off label uses may be subject to significant liability, both at the federal and state levels.
The Food and Drug Administration Amendments Act of 2007 gave the FDA the authority to require a Risk Evaluation and Mitigation Strategy, or REMS, from manufacturers to ensure that the benefits of a drug or biological product outweigh its risks. In determining whether a REMS is necessary, FDA must consider the size of the population likely to use the drug, the seriousness of the disease or condition to be treated, the expected benefit of the drug, the duration of treatment, the seriousness of known or potential adverse events, and whether the drug is a new molecular entity. If the FDA determines a REMS is necessary, the drug sponsor must agree to the REMS plan at the time of approval. A REMS may be required to include various elements, such as a medication guide or patient package insert, a communication plan to educate health care providers of the drug’s risks, limitations on who may prescribe or dispense the drug, or other measures that the FDA deems necessary to assure the safe use of the drug. In addition, the REMS must include a timetable to assess the strategy at 18 months, three years, and seven years after the strategy’s approval. The FDA may also impose a REMS requirement on a drug already on the market if the FDA determines, based on new safety information, that a REMS is necessary to ensure that the drug’s benefits outweigh its risks.
Orphan Drug Designation
The FDA may grant orphan drug designation to a drug intended to treat a rare disease or condition, which is defined as a disease or condition that affects fewer than 200,000 individuals in the United States or more than 200,000 individuals where there is no reasonable expectation that the product development cost will be recovered from product sales in the United States. Orphan drug designation must be requested before submitting an NDA and does not convey any advantage in, or shorten the duration of, the regulatory review or approval process. ADX-2191 has received orphan designation for the prevention of PVR, and for the treatment of PVRL and for the treatment of retinitis pigmentosa.
If an orphan drug-designated product subsequently receives the first FDA approval for the disease specified in the orphan drug designation, the sponsor will be entitled to seven years of product marketing exclusivity, which
28
means that the FDA may not approve any other applications to market the same drug for the same indication, except in very limited and rare circumstances, for seven years. If a competitor obtains approval of the same drug, as defined by the Orphan Drug Act, before we do or if our product candidate is determined to be contained within the competitor’s product for the same indication or disease, the competitor’s exclusivity could block the approval of our product candidate in the designated orphan indication for seven years, unless superior safety or efficacy of our drug is demonstrated.
Patent Term Restoration and Marketing Exclusivity
Depending upon the timing, duration, and specifics of FDA approval of the use of our drug candidates, some of our United States patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Act. The Hatch-Waxman Act permits a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND, and the submission date of an NDA, plus the time between the submission date of an NDA and the approval of that application. Only one patent applicable to an approved drug is eligible for the extension and the application for extension must be made prior to expiration of the patent. The United States Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we intend to apply for restorations of patent term for some of our currently owned or licensed patents to add patent life beyond the current expiration date, depending on the expected length of clinical trials and other factors involved in the submission of the relevant NDA.
Market exclusivity provisions under the FDCA also can delay the submission or the approval of certain applications. The FDCA provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to gain approval of an NDA for a new chemical entity. A drug is a new chemical entity if the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule responsible for the action of the drug substance. During the exclusivity period, the FDA may not accept for review an abbreviated new drug application (ANDA) or a 505(b)(2) NDA submitted by another company for another version of such drug where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement. The FDCA also provides three years of marketing exclusivity for a 505(b)(1) NDA, 505(b)(2) NDA, or supplement to an approved NDA if new clinical investigations other than bioavailability studies (e.g., investigations that support new indications, dosages, or strengths of an existing drug) were conducted or sponsored by the applicant and are deemed by the FDA to be essential to the approval of the application. The three-year exclusivity covers only the conditions associated with the new clinical investigations and does not prohibit the FDA from approving ANDAs for drugs containing the original active agent. Five-year and three-year exclusivity will not delay the submission or approval of a full 505(b)(1) NDA.
Manufacturing Requirements
We and our third-party manufacturers must comply with applicable FDA regulations relating to FDA’s cGMP regulations and, if applicable, quality system regulation requirements for medical devices. The cGMP regulations include requirements relating to, among other things, organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports, and returned or salvaged products. The manufacturing facilities for our products must meet cGMP requirements to the satisfaction of the FDA and may be subject to a pre-approval inspection before we can use them to manufacture our products. We and our third-party manufacturers are also subject to periodic unannounced inspections of facilities by the FDA and other authorities, including procedures and operations used in the testing and manufacture of our products to assess our compliance with applicable regulations. Failure to comply with statutory and regulatory requirements subjects a manufacturer to possible legal or regulatory action, including, among other things, warning letters, voluntary corrective action, the seizure of products, injunctions, consent decrees placing significant restrictions on or suspending manufacturing operations and civil and criminal penalties.
29
Other Regulatory Requirements
We are also subject to various laws and regulations regarding laboratory practices, the experimental use of animals, and the use and disposal of hazardous or potentially hazardous substances in connection with our research. In each of these areas, as above, the FDA has broad regulatory and enforcement powers, including, among other things, the ability to levy fines and civil penalties, suspend or delay issuance of approvals, seize or recall products, and withdraw approvals, any one or more of which could have an adverse effect on our ability to operate our business and generate revenues. Compliance with applicable environmental laws and regulations is expensive, and current or future environmental regulations may impair our research, development and production efforts, which could harm our business, operating results and financial condition. There are evolving legal requirements and other statutory and regulatory regimes that will continue to affect our business.
Research and Development Expenses
Substantially all of our research and development expenses incurred to date have been related to the development of reproxalap and our other product candidates. Our research and development expenses totaled $44.9 million for the year ended December 31, 2021 and $24.7 million for the year ended December 31, 2020.
We anticipate that we will incur additional research and development expenses in the future as we evaluate and possibly pursue the development of our product candidates for additional indications, or develop additional product candidates.
We recognize research and development expenses as they are incurred. Our research and development expenses consist primarily of:
•salaries and related expenses for personnel;
•fees paid to consultants and contract research organizations in conjunction with independently monitoring clinical trials and acquiring and evaluating data in conjunction with clinical trials, including all related fees such as investigator grants, patient screening, lab work and data compilation and statistical analysis;
•costs incurred with third parties related to the establishment of a commercially viable manufacturing process for our product candidates;
•costs related to production of clinical materials, including fees paid to contract manufacturers;
•costs related to upfront, milestone payments under in-licensing agreements as well as costs for unapproved inventory for which there is no future alternative use;
•costs related to compliance with FDA regulatory requirements;
•consulting fees paid to third-parties involved in research and development activities; and
•costs related to stock options or other stock-based compensation granted to personnel in development functions.
We expense both internal and external development costs as they are incurred.
30
We expect that a large percentage of our research and development expenses in the future will be incurred in support of our current and future non-clinical, preclinical and clinical development programs. These expenditures are subject to numerous uncertainties in terms of both their timing and total cost to completion. We expect to continue to develop stable formulations of our product candidates, test such formulations in preclinical studies for toxicology, safety and efficacy and to conduct clinical trials for each product candidate. We anticipate funding clinical trials for our product candidates ourselves, but we may engage collaboration partners at certain stages of clinical development. As we obtain results from clinical trials, we may elect to discontinue or delay clinical trials for certain product candidates or programs in order to focus our resources on more promising product candidates or programs. Completion of clinical trials by us or our future collaborators may take several years or more, the length of time generally varying with the type, complexity, novelty and intended use of a product candidate. The costs of clinical trials may vary significantly over the life of a project owing to but not limited to the following:
•the number of sites included in the trials;
•the length of time required to enroll eligible patients;
•the number of patients that participate in the trials;
•the number of doses that patients receive;
•the drop-out or discontinuation rates of patients;
•the duration of patient follow-up;
•the phase of development the product candidate is in; and
•the efficacy and safety profile of the product candidate.
Our expenses related to clinical trials are based on estimates of the services received and efforts expended pursuant to contracts with multiple research institutions and contract research organizations that conduct and manage clinical trials on our behalf. The financial terms of these agreements are subject to negotiation and vary from contract to contract and may result in uneven payment flows. Generally, these agreements set forth the scope of work to be performed at a fixed fee or unit price. Payments under the contracts depend on factors such as the successful enrollment of patients or the completion of clinical trial milestones. Expenses related to clinical trials generally are accrued based on contracted amounts applied to the level of patient enrollment and activity according to the protocol. If timelines or contracts are modified based upon changes in the clinical trial protocol or scope of work to be performed, we modify our estimates of accrued expenses accordingly on a prospective basis.
None of our product candidates have received FDA or foreign regulatory marketing approval. In order to grant marketing approval, a health authority such as the FDA or foreign regulatory agencies must conclude that clinical and preclinical data establish the safety and efficacy of our product candidates with an appropriate benefit to risk profile relevant to a particular indication, and that the product can be manufactured under cGMP in a reproducible manner to deliver the product’s intended performance in terms of its stability, quality, purity and potency. Until our submission is reviewed by a health authority, there is no way to predict the outcome of their review. Even if the clinical studies meet their predetermined primary endpoints, and a registration dossier is accepted for filing, a health authority could still determine that an appropriate benefit to risk relationship does not exist for the indication that we are seeking.
We cannot forecast with any degree of certainty which of our product candidates will be subject to future collaborations or how such arrangements would affect our development plan or capital requirements.
As a result of the uncertainties discussed above, we are unable to determine the duration and completion costs of our development projects or when and to what extent we will receive cash inflows from the commercialization and sale of an approved product candidate.
Corporate Information
We were incorporated in the state of Delaware on August 13, 2004 as Neuron Systems, Inc. On December 20, 2012, we changed our name to Aldexa Therapeutics, Inc. and on March 17, 2014, we changed our name to Aldeyra
31
Therapeutics, Inc. Our principal executive offices are located at 131 Hartwell Avenue, Suite 320, Lexington, Massachusetts 02421. Our telephone number is (781) 761-4904. Our website address is www.aldeyra.com. Information contained on our website is not incorporated by reference into this annual report on Form 10-K, and you should not consider information contained on our website to be part of this annual report on Form 10-K or in deciding whether to purchase shares of our common stock. Our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to reports filed or furnished pursuant to Sections 13(a) and 15(d) of the Securities Exchange Act of 1934, as amended, are available free of charge on the Investors portion of our website at http://ir.aldeyra.com/ as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC.
32
ITEM 1A. RISK FACTORS
Our business is subject to numerous risks. You should carefully consider the risks described below together with the other information set forth in this annual report on Form 10-K, which could materially affect our business, financial condition, and future results. The risks described below are not the only risks facing our company. Risks and uncertainties not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, prospects, financial condition, and operating results.
Summary of Risks Related to our Business
Below is a summary of the principal factors that make an investment in our common stock speculative or risky. This summary does not address all of the risks that we face. Additional discussion of the risks summarized in this risk factor summary, and other risks that we face, can be found below and should be carefully considered, together with other information in this annual report on Form 10-K and our other filings with the Securities and Exchange Commission before making investment decisions regarding our common stock.
•Our business is dependent in large part on the successful development and commercialization of a single product candidate, reproxalap, for which we are researching multiple indications. We cannot be certain that we will be able to obtain regulatory approval for, or successfully commercialize, reproxalap.
•To generate revenue, we will depend on FDA approval and successful commercialization of our lead product candidate, reproxalap, for the treatment of dry eye disease. If we are unable to successfully conduct the TRANQUILITY-2 trial, prepare and timely submit the planned NDA, and obtain FDA approval, our ability to generate revenue will be significantly delayed.
•We have incurred significant operating losses since inception and we expect to incur significant losses for the foreseeable future. We may never become profitable or, if achieved, be able to sustain profitability.
•We will require substantial additional financing, and a failure to obtain this necessary capital when needed on acceptable terms, or at all, could force us to delay, limit, reduce, or terminate our product development, other operations or commercialization efforts.
•Reproxalap and our other product candidates are subject to extensive regulation, compliance with which is costly and time consuming, and such regulation may cause unanticipated delays, or prevent the receipt of the required approvals to commercialize our product candidates.
•If our competitors develop treatments for the target indications of our product candidates that are approved more quickly than ours, marketed to physicians and patients more effectively or comprehensively, contracted with payors more successfully, or demonstrated to be safer or more effective than our product candidates, our commercial opportunity will be reduced or eliminated.
•If we fail to develop and commercialize our product candidates, we may be unable to grow our business.
•We rely on third parties to conduct our clinical trials. If any third party does not meet our deadlines or otherwise conduct the trials as required and in accordance with regulations, our clinical development programs could be delayed or unsuccessful and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates when expected, or at all.
•Public health emergencies, epidemics or pandemics, such as the ongoing COVID-19 pandemic, may impact our business.
Risks Related to our Business
Our business is dependent in large part on the successful development and commercialization of a single product candidate, reproxalap, for which we are researching multiple indications. We cannot be certain that we will be able to obtain regulatory approval for, or successfully commercialize, reproxalap.
Our product candidates, including reproxalap, will require additional preclinical studies, substantial clinical development and testing, and regulatory approval prior to commercialization. We have not yet completed development of any product candidate. We have only one product candidate that has been the focus of significant
33
clinical development: reproxalap, a novel small molecule chemical entity that is believed to inhibit RASP, a toxic chemical species suspected to cause and exacerbate numerous diseases in humans and animals. We are dependent in large part on successful continued development and ultimate regulatory approval and successful commercialization of reproxalap for our future business success. Any negative results or perceived negative results in clinical trials for one indication may have an adverse effect on our ability to develop and potentially commercialize reproxalap for the treatment of another indication. We have invested, and will continue to invest, a significant portion of our time and financial resources in the development of reproxalap and, if successful, the commercialization of reproxalap. We will need to raise sufficient funds for, and successfully enroll and complete, our current and planned clinical trials of reproxalap and our other product candidates. The future regulatory and commercial success of our product candidates is subject to a number of risks, including the following:
•we may not have sufficient financial and other resources to pursue our business plans, complete necessary clinical trials, and create a commercial infrastructure;
•we may not be able to provide sufficient evidence of safety and efficacy to continue a development program or obtain regulatory approval;
•the results of our clinical trials may not meet the endpoints, or level of statistical or clinical significance required by the FDA, or comparable foreign regulatory bodies, for marketing approval;
•the safety and efficacy results of our later phase or larger clinical trials may not confirm the results of our earlier trials;
•patients in our clinical trials may demonstrate greater response rates or improvements from vehicle or in the non-treatment arm then was expected when designing and powering our clinical trials;
•there may be variability in patients, adjustments to clinical trial procedures, and inclusion of additional clinical trial sites;
•the initial parts of adaptive clinical trials are not designed to be pivotal or definitive, and as such we may not satisfy the designated endpoints and also may need to revise the design or endpoints to achieve success in later parts of the trial or potentially abandon the trial;
•we may not be able to timely or adequately finalize the design or formulation of any product candidate or demonstrate that a formulation of our product candidate will be stable for commercially reasonable time periods;
•the FDA, or comparable foreign regulatory bodies, may implement new standards, or change the interpretation of existing standards or requirements for the regulatory approval, in general or with respect to the indications our product candidates are being developed to treat;
•the FDA, or comparable foreign bodies, may require clinical data in addition to the clinical trial programs we expect or may require changes to the designs and endpoints of the subsequent clinical trials;
•patients in clinical trials for our product candidates may suffer adverse effects or die for reasons that may or may not be related to our product candidates;
•if approved for certain diseases, our product candidates will compete with well-established and other products or therapeutic options already approved for marketing by the FDA, or comparable foreign regulatory bodies;
•competitive products may be more effectively or comprehensively marketed to physicians or patients, or contracted with payors more successfully;
•we may be adversely affected by legislative or regulatory reform of the health care system in the United States or other jurisdictions in which we may do business; and
•we may not be able to obtain, maintain, or enforce our patents and other intellectual property rights.
Of the large number of drugs in development in the pharmaceutical industry, only a small percentage result in the submission of an NDA to the FDA, and even fewer are approved for commercialization. We are currently
34
planning to submit an NDA to the FDA for reproxalap in DED in mid-2022, pending results from TRANQUILITY-2, a dry eye disease safety trial, and regulatory input. Even if our trials are successful, we can provide no assurances that the FDA will accept our NDA for review. Moreover, even if the FDA does accept our NDA for review, there can be no assurance that the FDA will agree with our interpretation of the data, that the FDA will not require us to conduct additional clinical trials or provide additional data, or that the FDA will approve our NDA in a timely fashion, or at all.
Furthermore, even if we do receive regulatory approval to market reproxalap and our other product candidates, any such approval may be subject to limitations on the indicated uses for which we may market the product. Accordingly, even if we are able to obtain the requisite financing to continue to fund our development programs, we cannot assure that reproxalap and our other product candidates will be successfully developed or commercialized. If we or any of our future development partners are unable to develop, or obtain regulatory approval for or, if approved, successfully commercialize, reproxalap and our other product candidates, we may not be able to generate sufficient revenue to continue our business.
To generate revenue, we will depend on FDA approval and successful commercialization of our lead product candidate, reproxalap, for the treatment of dry eye disease. If we are unable to successfully conduct the TRANQUILITY-2 trial, prepare and timely submit the planned NDA, and obtain FDA approval, our ability to generate revenue will be significantly delayed.
Our ability to generate revenue will depend on the successful development, regulatory approval and commercialization of reproxalap. Based on our discussions with and our interpretation of feedback from the FDA, as well as data from our previously completed clinical trials in dry eye disease and the TRANQUILITY-2 trial, we currently plan to submit an NDA to the FDA for reproxalap for the potential treatment of dry eye disease in mid-2022, subject to positive clinical trial results, including results from a dry eye disease safety trial. There can be no assurance that we can prepare and submit an NDA in a timely manner or at all. We have limited experience in preparing, filing, and pursuing applications necessary to gain regulatory approvals. The preparation of an NDA requires a great deal of effort and expertise, and if we do not secure the necessary resources and hire and retain personnel having the requisite expertise to prepare and submit the NDA, the filing of the NDA would be delayed. Further, if an NDA is submitted by the company, there can be no assurance that it will be accepted for filing by the FDA. If the FDA determines after an initial review of the NDA that the data included in the application is insufficient and not ready for formal consideration, we could receive a “refuse to file” notice. The FDA has substantial discretion in the approval process and may disagree with our interpretation of or the sufficiency of the data from our clinical trials. The FDA could also require that we conduct additional studies and submit the results of those studies before the application will be reconsidered, which would require us to expend more resources than we planned or that are available to us, and could substantially delay any approval of our application. It is also possible that additional studies may not suffice to make our application approvable. Even if the NDA is accepted for filing by the FDA, there can be no assurance that it would be approved in a timely manner or at all.
Issues with product quality could have a material adverse effect upon our business, subject us to regulatory actions and cause a loss of customer confidence in us or our products.
Our success depends upon the quality of our products. Quality controls, assurance, and management plays an essential role in meeting customer requirements, preventing defects, improving our product candidates and services, and assuring the safety and efficacy of our product candidates. Our future success depends on our ability to maintain and continuously improve our quality management program. A quality or safety issue may result in adverse inspection reports, warning letters, product recalls or seizures, monetary sanctions, injunctions to halt manufacture and distribution of products, civil or criminal sanctions, costly litigation, refusal of a government to grant approvals and licenses, restrictions on operations, or withdrawal of existing approvals and licenses. An inability to address a quality or safety issue in an effective and timely manner may also cause negative publicity, a loss of customer confidence in us or our future products, which may result in difficulty in successfully launching product candidates and the loss of sales, which could have a material adverse effect on our business, financial condition, and results of operations.
35
In order to commercialize our product candidates, we will need to substantially grow the size of our organization. We may encounter difficulties in managing our growth and expanding our operations successfully.
Because, as of December 31, 2021, we only had 12 full-time employees, we will need to grow our organization to continue development and pursue the potential commercialization of reproxalap and our other product candidates, as well as function as a public company. As we seek to advance reproxalap and other product candidates towards potential commercialization, increase the number of ongoing product development programs, and advance our future product candidates through preclinical studies and clinical trials, we will need to expand our financial, development, regulatory, manufacturing, marketing, and sales capabilities, or contract with third parties to provide these capabilities for us. As our operations expand, we expect that we will need to manage additional relationships with various strategic partners, suppliers, and other third parties. Future growth will impose significant added responsibilities on members of management and require us to retain additional internal capabilities. Our future financial performance and our ability to commercialize our product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To that end, we must be able to manage our development efforts and clinical trials effectively and hire, train, and integrate additional management, clinical and regulatory, financial, administrative and sales, and marketing personnel. We may not be able to accomplish these tasks, and our failure to so accomplish could prevent us from successfully growing our company.
We are highly dependent on the services of our senior management team and certain key consultants.
As a company with a limited number of personnel, we are highly dependent on the development, regulatory, commercial, and financial expertise of our senior management team comprised of three individuals: Todd C. Brady, M.D., Ph.D., our President and Chief Executive Officer; Joshua Reed, M.B.A., our Chief Financial Officer; and Stephen G. Machatha, Ph.D., our Chief Development Officer, as well as certain other employees. In addition, we rely on the services of a number of key consultants, including IP, pharmacokinetic, chemistry, toxicology, drug development, and commercialization consultants. The loss of such individuals or the services of future members of our management team could delay or prevent the further development and commercialization of our product candidates and, if we are not successful in finding suitable replacements, could harm our business.
If we fail to attract and retain senior management and key commercial personnel, we may be unable to successfully develop or commercialize our product candidates.
We will need to expand and effectively manage our managerial, operational, financial, and other resources in order to successfully pursue our clinical development and commercialization efforts. Our success also depends on our continued ability to attract, retain, and motivate highly qualified management and scientific personnel, and we may not be able to do so in the future due to intense competition among biotechnology and pharmaceutical companies, universities, and research organizations for qualified personnel. If we are unable to attract and retain the necessary personnel, we may experience significant impediments to our ability to implement our business strategy.
We expect to expand our management team. Our future performance will depend, in part, on our ability to successfully integrate newly hired executive officers into our management team and our ability to develop an effective working relationship among senior management. Our failure to integrate these individuals and create effective working relationships among them and other members of management could result in inefficiencies in the development and commercialization of our product candidates, adversely affecting future regulatory approvals, sales of our product candidates, and results of our operations.
Our employees, independent contractors, vendors, principal investigators, CROs, and consultants may engage in misconduct or other improper activities, including noncompliance with regulatory standards, regulatory requirements, and insider trading.
We are exposed to the risk that our employees, independent contractors, vendors, principal investigators, CROs and consultants may engage in fraudulent conduct or other illegal activity. Misconduct by these parties could include:
•intentional, reckless, or negligent conduct or disclosure to us of unauthorized activities that violate the regulations of the FDA or similar foreign regulatory authorities;
36
•healthcare fraud and abuse in violation of U.S. and foreign laws and regulations;
•violations of U.S. federal securities laws relating to trading in our common stock; and
•failures to report financial information or data accurately.
In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations govern a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. While we have adopted a code of conduct and implemented other internal controls applicable to all our employees, it is not always possible to identify and deter misconduct by employees and other third parties, and the precautions we take to detect and prevent this activity may not be effective. Additionally, we are subject to the risk that a person could allege fraud or other misconduct, even if none occurred. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business or cause reputational harm, including the imposition of civil, criminal and administrative penalties, and damages; possible exclusion from participation in Medicare, Medicaid, and other federal healthcare programs; and diminished profits and future earnings.
In addition, during the course of our operations, our directors, executives, employees, consultants, and other third parties may have access to material nonpublic information regarding our business, our results of operations, or potential transactions we are considering. We may not be able to prevent trading in our common stock on the basis of, or while having access to, material nonpublic information. If any such person was to be investigated or an action were to be brought against them for insider trading, it could have a negative impact on our reputation and our stock price. Such a claim, with or without merit, could also result in substantial expenditures of time and money, and divert attention of our management team from other tasks important to the success of our business.
Our business is subject to political, economic, legal, and social risks, which could adversely affect our business.
There are significant regulatory, economic and legal barriers in markets in the United States and outside the United States that we must overcome. We may be subject to the burden of complying with a wide variety of national and local laws, including multiple and possibly overlapping and conflicting laws. We also may experience difficulties adapting to new cultures, business customs, and legal systems. Any sales and operations would be subject to political, economic, and social uncertainties including, among others:
•changes and limits in import and export controls;
•increases in custom duties and tariffs;
•changes in currency exchange rates;
•economic and political instability;
•the impact on employees, suppliers, customers, and the global economy related to public health epidemics or pandemics, such as the COVID-19 pandemic, and actions taken in response to such events;
•changes in government regulations and laws;
•absence in some jurisdictions of effective laws to protect our intellectual property rights; and
•currency transfer and other restrictions and regulations that may limit our ability to sell certain products or repatriate profits to the United States.
Changes in United States social, political, regulatory, and economic conditions or in laws and policies governing foreign trade, manufacturing, development, and investment, and any negative sentiments towards the United States as a result of such changes, could adversely affect our business.
37
Any changes related to these and other factors could adversely affect any business operations that we conduct outside the United States.
If we engage in an acquisition, reorganization, or business combination, we will incur a variety of risks that could adversely affect our business operations or our stockholders.
From time to time, we have entered into, and we will continue to consider in the future, strategic business initiatives intended to further the development of our business. These initiatives may include acquiring businesses, technologies, or products, or entering into a business combination with another company. For example, in January 2019 we acquired Helio Vision, Inc. and obtained the rights to ADX-2191, a vitreous-compatible methotrexate formulation for intraocular injection, for the prevention of PVR. Any acquisitions we undertake or have recently completed will likely be accompanied by business risks which may include, among other things:
•the effect of the acquisition on our financial and strategic position and reputation;
•the failure of an acquisition to result in expected benefits, which may include benefits relating to new product candidates, human resources, costs savings, operating efficiencies, goodwill, and other synergies;
•the difficulty, cost, and management effort required to integrate the acquired businesses, including costs and delays in implementing common systems and procedures, and costs and delays caused by communication difficulties;
•the assumption of certain known or unknown liabilities of the acquired business, including litigation-related liabilities;
•the reduction of our cash available for operations and other uses, the increase in amortization expense related to identifiable assets acquired, potentially dilutive issuances of equity securities, or the incurrence of debt;
•the possibility that we will pay more than the value we derive from the acquisition;
•the impairment of relationships with our partners, consultants, or suppliers, or those of the acquired business; and
•the potential loss of key employees of the acquired business.
These factors could harm our business, results of operations, or financial condition.
In addition to the risks commonly encountered in the acquisition of a business or assets as described above, we may also experience risks relating to the challenges and costs of closing a transaction. The risks described above may be exacerbated as a result of managing multiple acquisitions at once.
Security breaches, loss of data, and other disruptions to us or our third-party service providers could compromise sensitive information related to our business or prevent us from accessing critical information and expose us to liability, which could adversely affect our business and our reputation.
In the ordinary course of our business, we and our third-party service providers collect and store sensitive data, including legally protected health information, personally identifiable information about patients, intellectual property, and our proprietary business and financial information. We manage and maintain data utilizing a combination of on-site systems, managed data center systems, and cloud-based data center systems. We face a number of risks related to our protection of, and our service providers’ protection of, this critical information, including loss of access, inappropriate disclosure and inappropriate access, as well as risks associated with our ability to identify and audit such events.
The secure processing, storage, maintenance and transmission of critical information is vital to our operations and business strategy, and we devote significant resources to protecting such information. Although we take measures to protect sensitive information from unauthorized access or disclosure, our information technology and infrastructure may be vulnerable to attacks by hackers or viruses or otherwise breached due to employee error,
38
malfeasance, or other activities. These risks may be heightened during the ongoing COVID-19 pandemic, should any of our employees voluntarily choose to work remotely. While we are not aware of any such attack or breach, if such event would occur and cause interruptions in our operations, our networks would be compromised and the information we store on those networks could be accessed by unauthorized parties, publicly disclosed, lost, or stolen. Any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under federal, state, and international laws that protect the privacy of personal information, such as the Health Insurance Portability and Accountability Act of 1966, or HIPAA, and regulatory penalties. In addition, we may be subject to state laws requiring notification of affected individuals and state regulators in the event of a breach of personal information, which is a broader class of information than the health information protected by HIPAA. Unauthorized access, loss, or dissemination of critical information could also disrupt our ability to conduct research and development activities; collect, process, and prepare company financial information; provide information about our product candidates and other patient and physician education or outreach efforts through our website; manage the administrative aspects of our business; or prevent damage to our reputation, any of which could adversely affect our business.
We generally seek to comply with industry standards and are subject to the terms of our privacy policies and privacy-related obligations to third parties. We strive to comply with all applicable laws, policies, legal obligations, and industry codes of conduct relating to privacy and data protection to the extent possible. However, it is possible that these obligations may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another and may conflict with other rules or our practices. Any failure or perceived failure by us to comply with applicable privacy and data security laws and regulations, our privacy policies, or our privacy-related obligations to third parties, or any compromise of security that results in the unauthorized release or transfer of personal information or other sensitive data, may result in governmental enforcement actions, litigation, or public statements against us by consumer advocacy groups or others and could cause a loss of trust in us, which would have an adverse effect on our reputation and business.
In addition, the interpretation and application of consumer, health-related and data protection laws in the United States, Europe, and elsewhere are often uncertain, contradictory and in flux. It is possible that these laws may be interpreted and applied in a manner that is inconsistent with our practices. If so, this could result in government-imposed fines or orders requiring that we change our practices, which could adversely affect our business. In addition, we are subject to various state laws, including the California Consumer Privacy Act, or CCPA, which, among other things, requires covered companies to provide disclosures to California consumers concerning the collection and sale of personal information, and gives such consumers the right to opt-out of certain sales of personal information. The CCPA provides for civil penalties for violations, as well as a private right of action for security breaches that may increase the likelihood of, and the risks associated with, security breach litigation. Additionally, in November 2020, California passed the California Privacy Rights Act, or CPRA, which expands the CCPA significantly, including by expanding consumers’ rights with respect to certain personal information and creating a new state agency to oversee implementation and enforcement efforts, potentially resulting in further uncertainty and requiring us to incur additional costs and expenses in an effort to comply. Many of the CPRA’s provisions will become effective on January 1, 2023. Further, Virginia enacted the Virginia Consumer Data Protection Act, or CDPA, and, most recently, Colorado passed the Colorado Privacy Act, or CPA, both comprehensive state privacy laws with an effective date of July 1, 2023. The CCPA, CPRA, CDPA, and CPA may increase our compliance costs and potential liability, particularly in the event of a data breach, and could have a material adverse effect on our business, including how we use personal information, our financial condition, and the results of our operations. The CCPA has also prompted a number of proposals for new federal and state privacy legislation that, if passed, could increase our potential liability, increase our compliance costs and adversely affect our business.
Recent developments in Europe have created compliance uncertainty regarding the processing of personal data from Europe. For example, the General Data Protection Regulation, or GDPR, which became effective in the European Union (EU) on May 25, 2018, applies to our activities conducted from an establishment in the EU or related EU data subject. The GDPR creates new compliance obligations applicable to our business, which could cause us to change our business practices, and increases financial penalties for noncompliance (including possible fines of up to 4% of global annual turnover for the preceding financial year or €20 million, whichever is higher, for the most serious infringements). Further, as of January 1, 2021, and the expiry of transitional arrangements agreed to between the United Kingdom (UK) and EU, data processing in the United Kingdom is governed by a United Kingdom version of the GDPR (combining the GDPR and the Data Protection Act 2018), exposing us to two
39
parallel sets of regulations, each of which potentially authorizes similar fines and other potentially divergent enforcement actions for certain violations. The GDPR and UK GDPR require, among other things, that personal information only be transferred outside of the European Economic Area, or the EEA, or the UK, respectively, to jurisdictions that have not been deemed adequate by the European Commission or by the UK data protection regulator, respectively, including the United States, if certain safeguards are taken to legitimize those data transfers. Recent legal developments in the EU have created complexity and uncertainty regarding such transfers. These recent developments require us to review and amend our uses of Standard Contractual Clauses involving the transfer of data covered by the GDPR or UK GDPR outside of the EU or UK, which could increase our compliance costs and adversely affect our business. As a result, we may need to modify the way we treat such information.
In addition to the EU and UK, a growing number of other global jurisdictions are considering or have passed legislation implementing data protection requirements or requiring local storage and processing of data or similar requirements that could increase the cost and complexity of our business. Some of these laws, such as the General Data Protection Law in Brazil, or the Act on the Protection of Personal Information in Japan, impose similar obligations as those under the GDPR. Others, such as those in Russia, India, and China, could potentially impose more stringent obligations, including data localization requirements. If we are unable to meet these evolving legal requirements or if we violate or are perceived to violate any laws, regulations, or other obligations relating to privacy, data protection, or information security, we may harm to our reputation and become subject to investigations, claims, and other remedies, which would expose us to significant fines, penalties, and other damages, all of which would harm our business.
Our internal computer systems, or those of our development partners, third-party clinical research organizations, or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs.
Despite the implementation of security measures, our internal computer systems and those of our current and any future Contract Research Organizations (CROs) and other contractors, consultants, and collaborators are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war or other military conflict, including an escalation of the conflict between Russia and Ukraine, and telecommunication and electrical failures. While to our knowledge we have not experienced any such material system failure, accident, or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties to manufacture our product candidates and conduct clinical trials, and similar events relating to their computer systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our product candidate could be delayed.
We rely on email and other messaging services in connection with our operations. We may be targeted by parties using fraudulent spoofing and phishing emails to misappropriate passwords, payment information, or other personal information, or to introduce viruses through Trojan horse programs or otherwise through our networks, computers, smartphones, tablets, or other devices. Despite our efforts to mitigate the effectiveness of such malicious email campaigns through a variety of control and non-electronic checks, spoofing and phishing may damage our business and increase our costs. These risks may be heightened during the ongoing COVID-19 pandemic, should any of our employees voluntarily choose to work remotely. In addition, due to the political uncertainty involving Russia and Ukraine, there is an increased likelihood that escalation of tensions could result in cyberattacks that could either directly or indirectly impact our operations. Any of these events or circumstances could materially adversely affect our business, financial condition, and operating results.
40
Risks Related to our Financial Position and Capital Requirements
We have incurred significant operating losses since inception and we expect to incur significant losses for the foreseeable future. We may never become profitable or, if achieved, be able to sustain profitability.
We have incurred significant operating losses since we were founded in 2004 and expect to incur significant losses for the next several years as we continue our clinical trial, development programs, and commercial activities for reproxalap and our other product candidates. Net loss for the year ended December 31, 2021 and 2020 was approximately $57.8 million and $37.6 million, respectively. As of December 31, 2021, we had total stockholders’ equity of $205.7 million and an accumulated deficit of $294.7 million. Losses have resulted principally from costs incurred in our clinical trials, research and development programs and from general and administrative expenses. In the future, we intend to continue to conduct research and development, clinical testing, regulatory compliance activities, pre-commercial activities, and, if reproxalap or any of our other product candidates is approved, commercialization efforts, including sales and marketing activities, that, together with anticipated general and administrative expenses, will likely result in our incurring further significant losses for the next several years.
We currently generate no revenue from sales, and we may never be able to commercialize reproxalap or our other product candidates. We do not currently have the required approvals to market any of our product candidates and we may never receive them. We may not be profitable even if we or any of our future development partners succeed in obtaining regulatory approval and commercialize any of our product candidates. Because of the numerous risks and uncertainties associated with developing and commercializing our product candidates, we are unable to predict the extent of any future losses or when we will become profitable, if at all.
We will require substantial additional financing, and a failure to obtain this necessary capital when needed on acceptable terms, or at all, could force us to delay, limit, reduce or terminate our product development, other operations or commercialization efforts.
The development and commercialization of biopharmaceutical products is capital intensive. We are advancing reproxalap and our other product candidates through preclinical and clinical development, including multiple ongoing and planned clinical trials for our product candidates. We expect our expenses to increase in connection with our ongoing activities as we continue the research and development of, and, if successful, seek marketing approval for, our product candidates. In addition, if we obtain marketing approval for any of our product candidates, we expect to incur significant commercialization expenses related to manufacturing, product sales, marketing, and distribution. We may also need to raise additional funds sooner if we choose to pursue additional indications for our product candidates or otherwise expand more rapidly than we presently anticipate. Furthermore, we expect to continue to incur additional costs associated with operating as a public company. Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations. If we are unable to raise capital when needed on attractive terms, if at all, we will be forced to delay, reduce, or eliminate certain of our clinical development plans, research and development programs or future commercialization efforts. In addition, there can be no assurance that we will be able to obtain such financing on commercially reasonable terms or at all. The development process for our product candidates is highly uncertain, and we cannot estimate with certainty the actual amounts necessary to successfully complete the development, regulatory approval, and commercialization of our product candidates. Our operating plans may change as a result of many factors currently unknown to us, and we may need to seek additional funds sooner than expected, through public or private equity, debt financings, or other sources. The amount and timing of any expenditure needed to implement our development and commercialization programs will depend on numerous factors, including:
•the type, number, scope, progress, expansion costs, results, and timing of our clinical trials of reproxalap or any our other product candidates that we are pursuing or may choose to pursue in the future;
•the need for, and the progress, costs, and results of, any additional clinical trials of reproxalap and our other product candidates we may initiate based on the results of our planned clinical trials or discussions
41
with the FDA, including any additional trials the FDA or other regulatory agencies may require evaluating the safety or efficacy of reproxalap and our other product candidates;
•the costs of obtaining, maintaining, and enforcing our patents and other intellectual property rights;
•the costs and timing of obtaining or maintaining manufacturing for reproxalap and our other product candidates, including commercial manufacturing, if any product candidate is approved;
•the costs and timing of establishing sales and marketing capabilities and enhanced internal controls over financial reporting;
•the terms and timing of establishing collaborations, license agreements, and other partnerships on terms favorable to us;
•costs associated with any other product candidates that we may develop, in-license, or acquire, including potential milestone or royalty payments;
•the effect of competing technological and market developments;
•our ability to establish and maintain partnering arrangements for development; and
•the costs associated with being a public company.
Some of these factors are outside of our control. Our existing capital resources are not sufficient to enable us to fund the completion of our clinical trials and remaining development through commercial introduction. We expect that we will need to raise substantial additional funds in the near future.
We have not sold any products, and we do not expect to sell or derive revenue from any product sales for the foreseeable future. We may seek additional funding through collaboration agreements and public or private financings, including debt financings. The state of the global economy and market instability has made the business climate volatile and more costly. Uncertain economic conditions, uncertainty as to the general direction of the macroeconomic environment, and the price of our common stock, are beyond our control and may make any necessary debt or equity financing more difficult, more costly, and more dilutive. Additional funding may not be available to us on acceptable terms, or at all. In addition, the terms of any financing may adversely affect the holdings or the rights of our stockholders or be excessively dilutive. In addition, the issuance of additional shares by us, or the possibility of such issuance, may cause the market price of our shares to decline.
If we are unable to obtain funding on a timely basis, we will be unable to complete the planned clinical trials for reproxalap and our other product candidates, and we may be required to significantly curtail, delay, or discontinue one or more of our preclinical studies, clinical trials or other research or development programs, or the commercialization of any product candidate. We may also be unable to expand our operations or otherwise capitalize on our business opportunities, may need to restructure our organization, or may be required to relinquish rights to our product candidates or other technologies, or otherwise agree to terms unfavorable to us. Any of these occurrences could materially affect our business, financial condition, and results of operations.
Raising additional capital may cause dilution to stockholders, restrict our operations or require us to relinquish rights to its technologies or product candidates.
Until such time, if ever, as we can generate substantial product revenues, we expect to finance our cash needs through a combination of equity offerings, debt financings, collaborations, strategic alliances, licensing arrangements, and marketing and distribution arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, your ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect your rights as a common stockholder. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting its ability to take specific actions, such as incurring additional debt, making capital expenditures, or declaring dividends.
If we raise additional funds through collaborations, strategic alliances, licensing arrangements or marketing and distribution arrangements, we may have to relinquish valuable rights to our technologies, future revenue
42
streams, research programs, or product candidates, or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, we may be required to delay, limit, reduce, or terminate its product development or future commercialization efforts or grant rights to develop and market products or product candidates that we would otherwise prefer to develop and market on our own.
We may allocate our cash and cash equivalents in ways that you and other stockholders may not approve.
Our management has broad discretion in the application of our cash and cash equivalents. Because of the number and variability of factors that will determine our use of our cash and cash equivalents, management’s ultimate use of cash and cash equivalents may vary substantially from the currently intended use. Our management might not apply our cash and cash equivalents in ways that ultimately increase the value of your investment. We expect to use our cash and cash equivalents to: fund our planned clinical trials of reproxalap and our other product candidates; fund potential NDA submissions; fund an initial commercialization of reproxalap, if approved; develop other molecules that relate to immune-mediated disease; pursue regulatory approval for our product candidates; service our debt obligations; and provide working capital and capital for other general corporate purposes. The failure by our management to apply these funds effectively could harm our business. We may invest our cash and cash equivalents in short-term, investment-grade, interest-bearing securities. These investments may not yield a favorable return to our stockholders. If we do not invest or apply our cash and cash equivalents in ways that enhance stockholder value, we may fail to achieve expected financial results, which could cause our stock price to decline.
The terms of our secured debt facility require us to meet certain operating covenants and place restrictions on our operating and financial flexibility. If we raise additional capital through debt financing, the terms of any new debt could further restrict our ability to operate our business.
In March 2019, we entered into a credit facility with Hercules Capital, which was subsequently amended in April 2021, that is secured by a lien covering all of our assets, other than our intellectual property. The loan agreement contains customary affirmative and negative covenants and events of default. Affirmative covenants include, among others, covenants requiring us to maintain our legal existence and governmental approvals, deliver certain financial reports, and maintain insurance coverage. Negative covenants include, among others: restrictions on transferring any part of our business or intellectual property; incurring additional indebtedness; engaging in mergers or acquisitions; paying dividends or making other distributions; making investments; and creating other liens on our assets, in each case subject to customary exceptions. If we raise any additional debt financing, the terms of such additional debt could further restrict our operating and financial flexibility. These restrictions may include, among other things, limitations on borrowing and specific restrictions on the use of our assets, as well as prohibitions on our ability to create liens, pay dividends, redeem capital stock, or make investments. If we default under the terms of the Hercules Credit Facility or any future debt facility, the lender may accelerate all of our repayment obligations and take control of our pledged assets, potentially requiring us to renegotiate our agreement on terms less favorable to us or to immediately cease operations. Further, if we are liquidated, the lender’s right to repayment would be senior to the rights of the holders of our common stock. The lender could declare a default upon the occurrence of any event that they interpret as a material adverse effect as defined under the loan agreement. Any declaration by the lender of an event of default could significantly harm our business and prospects and could cause the price of our common stock to decline. If we raise any additional debt financing, the terms of such additional debt could further restrict our operating and financial flexibility.
Our ability to use net operating loss carryforwards and tax credit carryforwards to offset future taxable income may be limited as a result of transactions involving our common stock.
In general, under Section 382 of the Internal Revenue Code of 1986, as amended, a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change net operating losses (NOLs) and certain other tax assets (tax attributes) to offset future taxable income. In general, an ownership change occurs if the aggregate stock ownership of certain stockholders increases by more than 50 percentage points over such stockholders’ lowest percentage ownership during the testing period (generally three years). Transactions involving our common stock within the testing period, even those outside our control, such as purchases or sales by investors, could result in an ownership change. A limitation on our ability to utilize some or all of our NOLs or credits could have a material adverse effect on our results of operations and cash flows. We believe, prior to December 31, 2020
43
that three ownership changes occurred since inception, and that a fourth ownership change was triggered during the year ended December 31, 2021. Management believes that its aggregate Section 382 limitation (including the additional limitation for recognized "built-in gains") is sufficient so that no current impairment of its pre-ownership change tax attributes is required. Any future ownership changes, including those resulting from our recent or future financing activities, may cause our existing tax attributes to have additional limitations. Any future ownership changes, including those resulting from our recent or future financing activities, may cause our existing tax attributes to have additional limitations. However, subject to annual limitations, Federal NOLs generated in years 2018 and beyond will have an indefinite carryforward period and will not expire. Future changes in federal and state tax laws pertaining to NOL carryforwards may also cause limitations or restrictions from us claiming such NOLs. If the NOL carryforwards become unavailable to us or are fully utilized, our future taxable income will not be shielded from federal and state income taxation absent certain U.S. federal and state tax credits, and the funds otherwise available for general corporate purposes would be reduced.
Governments may impose price controls, which may adversely affect our future profitability.
We intend to seek approval to market our product candidates in both the United States and in foreign jurisdictions. If we obtain approval in one or more foreign jurisdictions, we will be subject to rules and regulations in those jurisdictions relating to our product candidates. In some foreign countries, particularly in the EU, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a product candidate. To obtain reimbursement or pricing approval in some countries, we, or our collaborators, may be required to conduct a clinical trial that compares the cost-effectiveness of our drug to other available therapies. Furthermore, in some European countries, the authorities conduct a Health Technology Appraisal to assess the cost-effectiveness of the product, which may significantly impact effective access to the market. If reimbursement of our future products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, we may be unable to achieve or sustain profitability.
Business disruptions could seriously harm our future revenues and financial condition and increase our costs and expenses.
Our operations could be subject to business disruptions such as earthquakes, power shortages, telecommunications failures, water shortages, floods, hurricanes, typhoons, fires, extreme weather conditions, public health epidemics (including the COVID-19 pandemic), regional or larger scale conflicts or geo-political actions, war or other military conflict, including an escalation of the conflict between Russia and Ukraine, trade policies, sanctions, treaties and tariffs and other natural or man-made disasters or other business interruptions, for which we are predominantly self-insured. The occurrence of any of these business disruptions could seriously harm our operations and financial condition, and increase our costs and expenses. We rely on third-party manufacturers to produce reproxalap and our other product candidates. Our ability to obtain clinical and commercial supplies of reproxalap or our other product candidates could be disrupted, if the operations of these suppliers are affected by these business disruptions.
Our quarterly operating results may fluctuate significantly.
We expect our operating results to be subject to quarterly fluctuations. Our net loss and other operating results will be affected by numerous factors, including:
•variations in the level of expenses related to our clinical trial and development programs;
•addition or termination of clinical trials or development programs;
•any intellectual property infringement lawsuit in which we may become involved;
•regulatory developments affecting reproxalap and our other product candidates;
•the impact of the COVID-19 pandemic on our business, results of operations, and financial position;
•our establishment and maintenance of a sales, marketing and distribution infrastructure to commercialize any product candidate for which we may obtain marketing approval;
44
•our execution of any collaborative, licensing, or similar arrangements, and the timing of payments we may make or receive under these arrangements;
•the number of administrative, clinical, regulatory, and scientific personnel we engage;
•nature and terms of stock-based compensation grants; and
•derivative instruments recorded at fair value.
If our quarterly operating results fall below the expectations of investors or securities analysts, the price of our common stock could decline substantially. Furthermore, any quarterly fluctuations in our operating results may, in turn, cause the price of our stock to fluctuate substantially. We believe that quarterly comparisons of our financial results are not necessarily meaningful and should not be relied upon as an indication of our future performance.
Risks Related to the COVID-19 Pandemic
Our business has been and will continue to be adversely affected by the COVID-19 pandemic.
The global spread of COVID-19, and variants thereof, has created significant volatility, uncertainty, and economic disruption. The extent to which the COVID-19 pandemic impacts our business, operations, and financial results will depend on numerous evolving factors that we may not be able to accurately predict, including: the duration and scope of the pandemic; governmental, business and individuals’ actions that have been and continue to be taken in response to the pandemic; the impact of the pandemic on economic activity and actions taken in response; the effect on our ongoing or planned clinical trials; and any closures of our offices or clinical trial facilities. Any of these events could cause or contribute to the risks and uncertainties enumerated in this annual report on Form 10-K and could materially adversely affect our business, financial condition, results of operations, and/or stock price.
We are closely monitoring the impact of the COVID-19 pandemic on all aspects of our business, including how it will impact our employees, clinical trials, development programs, supply chain, and other aspects of our operations. For example, patient enrollment in our GUARD trial, was negatively impacted as a result of limited clinical trial staffing at study sites and some patients electing to delay surgery. While COVID-19 did not have a material adverse effect on our reported results for the year ended December 31, 2021, we are unable to predict the ultimate impact that it may have on our business, future results of operations, financial position, or cash flows. The extent to which our operations may be impacted by the COVID-19 pandemic will depend largely on future developments, which are highly uncertain and cannot be accurately predicted, including new information which may emerge concerning the severity of the outbreak and actions by government authorities to contain the outbreak or diminish its impact.
We have experienced operational interruptions as a result of the COVID-19 pandemic, with certain of our employees electing to work remotely to avoid spread of the disease. Our offices are currently open as allowed under state and local orders. While we have implemented what we believe to be a reasonable protocol to ensure the safety and wellbeing of employees returning to the office, these measures may not be sufficient to mitigate the risks posed by the virus or otherwise be satisfactory to government authorities. A prolonged pandemic, or the threat thereof, could result in lower productivity, voluntary closure of our office, and other disruptions to our business. We may also incur additional costs to remedy damages caused by business disruptions, including clinical trial delays or interruptions. Any of these could have a material adverse effect on our business, financial condition, or results of operations.
Due to the evolving and highly uncertain nature of this event, we cannot predict at this time the full extent to which the COVID-19 pandemic will adversely impact our business, results, or financial condition, which will depend on many factors that are not known at this time. These include, among others, the extent of harm to public health, the continued disruption to clinical trials, and the impact of the global business and economic environment on liquidity, and the availability of capital. We are staying in close communication with our employees, CROs, and suppliers, and acting to mitigate the impact of this dynamic and evolving situation, but there is no guarantee that we will be able to do so.
45
To the extent the COVID-19 pandemic or any worsening of the global business and economic environment as a result thereof adversely affects our business and financial results, it may also have the effect of heightening or exacerbating many of the other risks described in this section of this annual report on Form 10-K.
COVID-19 has and is expected to continue to affect our ability to recruit or retain patients for our clinical trials, disrupt our supply chains, or have other adverse effects on our business and operations.
In addition to those who have been directly affected by COVID-19, millions more have been affected by government efforts around the world to slow the spread of the pandemic through quarantines, travel restrictions, heightened border scrutiny, and other measures. The outbreak and government measures taken in response to the COVID-19 pandemic have also had a significant impact, both direct and indirect, on businesses and commerce, as worker shortages have occurred; supply chains have been disrupted; facilities and production have been suspended; the capital markets and the economy in general has been volatile; and demand for certain goods and services, such as medical services and supplies, has spiked, while demand for other goods and services, such as travel, has fallen. The future progression of the outbreak, including the emergence of COVID-19 variants, and its effects on our business and operations are uncertain. We have and expect to continue to face difficulties recruiting or retaining patients in our ongoing and planned clinical trials due to actions taken by governments and individuals in response to the COVID-19 pandemic. Patient enrollment in our GUARD trial was negatively impacted as a result of limited clinical trial staffing at study sites and some patients electing to delay surgery. We and our third-party contract manufacturers, contract research organizations, and clinical sites may also face disruptions in procuring items that are essential for our research and development activities, including, for example, medical and laboratory supplies used in our clinical trials or preclinical studies, in each case, that are sourced from abroad or for which there are shortages because of ongoing efforts to address the pandemic.
Risks Related to the Development and Commercialization of our Product Candidates
Reproxalap and our other product candidates are subject to extensive regulation, compliance with which is costly and time consuming, and such regulation may cause unanticipated delays, or prevent the receipt of the required approvals to commercialize our product candidates.
The clinical development, manufacturing, labeling, storage, record-keeping, advertising, promotion, import, export, marketing, and distribution of our product candidates are subject to extensive regulation by the FDA in the United States and by comparable authorities in foreign markets. In the United States, we are not permitted to market our product candidates until we receive regulatory approval from the FDA. The process of obtaining regulatory approval is expensive and time-consuming, and can vary substantially based upon the type, complexity, and novelty of the products involved, as well as the target indication, and patient population. Approval policies or regulations may change, and the FDA has substantial discretion in the drug approval process, including the ability to delay, limit, or deny approval of a product candidate for many reasons. Despite the time and expense invested in clinical development of product candidates, regulatory approval, and subsequent commercial success is uncertain and not guaranteed.
Reproxalap and our other product candidates, and the activities associated with development and commercialization, including testing, manufacture, safety, efficacy, recordkeeping, labeling, storage, approval, advertising, promotion, sale, and distribution, are subject to extensive regulation by the FDA and other regulatory agencies in the United States and by comparable authorities in other jurisdictions.
Our ongoing research and development activities and planned clinical development and commercialization for our product candidates may be delayed, modified, or ceased for a variety of reasons, including:
•determining that a product candidate is ineffective or potentially causes harmful side effects during preclinical studies or clinical trials;
•difficulty establishing predictive preclinical models for demonstration of safety and efficacy of a product candidate in one or more potential therapeutic areas for clinical development;
•patients in our clinical trials may demonstrate greater response rates or improvements from vehicle or standard of care than was expected when designing and powering our clinical trials;
46
•delays resulting from or additional protocols being required as result of the COVID-19 pandemic;
•lack of availability of, or difficulty recruiting and retaining, a sufficient number of patients to adequately power our clinical trials;
•difficulties in manufacturing a product candidate, including the inability to manufacture a product candidate in a sufficient quantity, suitable form, or in a cost-effective manner, or under processes acceptable to the FDA for marketing approval or commercial sale;
•the proprietary rights of third parties, which may preclude us from developing or commercializing a product candidate;
•determining that a product candidate may be uneconomical for us to develop or commercialize, or may fail to achieve market acceptance or adequate pricing or reimbursement;
•our expectations regarding our expenses and revenue, the sufficiency or use of our cash resources, and needs for additional financing;
•a safety concern or signal may arise that triggers a clinical hold;
•our inability to secure strategic partners which may be necessary for advancement of a product candidate into clinical development or commercialization; or
•our prioritization of other indications or product candidates for advancement.
The FDA or comparable foreign regulatory authorities can delay, limit, or deny approval of a product candidate for many reasons, including but not limited to:
•such authorities may disagree with the design or implementation of our or any of our future development partners’ clinical trials, including the endpoints of our clinical trials;
•such authorities may require clinical data in addition to clinical trial programs we expect, or may require changes to the designs and endpoints of subsequent clinical trials;
•a competitor product may have patent protection or another type of market exclusivity that delays approval of our product;
•we or any of our future development partners may be unable to demonstrate to the satisfaction of the FDA or other regulatory authorities that a product candidate is safe and effective for any indication;
•such authorities may not accept clinical data from trials if conducted at clinical facilities or in countries where the standard of care is potentially different from the United States;
•the results of clinical trials may not demonstrate the safety or efficacy required by such authorities for approval;
•we or any of our future development partners may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks;
•such authorities may disagree with our interpretation of data from preclinical studies or clinical trials or the design of such trials or require additional trials and data;
•changes in the leadership or operation of such authorities, which may result in, among other things, the implementation of new standards, or changes to the interpretation or enforcement of existing regulatory standards and requirements;
•such authorities may find deficiencies in the manufacturing processes or facilities of third-party manufacturers with which we or any of our future development partners contract for clinical and commercial supplies; or
•the approval policies, standards, or regulations of such authorities may significantly change in a manner rendering our or any of our future development partners’ clinical data insufficient for approval.
47
With respect to foreign markets, approval procedures vary among countries and, in addition to the aforementioned risks, can involve additional product testing, administrative review periods, and agreements with pricing authorities. In addition, events raising questions about the safety of certain marketed pharmaceuticals may result in increased cautiousness by the FDA and comparable foreign regulatory authorities in reviewing new drugs based on safety, efficacy, or other regulatory considerations and may result in significant delays in obtaining regulatory approvals. Any delay in obtaining, or inability to obtain, applicable regulatory approvals would prevent us or any of our future development partners from commercializing our product candidates. Moreover, we cannot predict healthcare reform initiatives, including potential reductions in federal funding or insurance coverage, that may be adopted in the future and whether or not any such reforms would have an adverse effect on our business and our ability to obtain regulatory approval for our current or future product candidates. There are evolving legal requirements that will continue to affect our business.
Because the Company has no experience in commercializing pharmaceutical products, there is a limited amount of information about us upon which to evaluate our product candidates and business prospects.
We have not yet demonstrated an ability to successfully overcome many of the pre-commercial and commercial risks and uncertainties frequently encountered by companies in new and rapidly evolving fields, particularly in the biopharmaceutical area. For example, to execute our business plan we will need to successfully:
•execute our product candidate development activities, including successfully designing and completing our clinical trial programs and product design and formulation of future product candidates, in a cost- effective manner;
•file for and obtain required regulatory approvals for our product candidates;
•manage our spending as costs and expenses increase due to the performance and completion of clinical trials, attempting to obtain regulatory approvals, manufacturing, and commercialization;
•secure substantial additional funding;
•develop and maintain successful strategic relationships;
•build and maintain a strong intellectual property portfolio;
•build and maintain appropriate clinical, regulatory, quality, manufacturing, compliance, sales, distribution, and marketing capabilities on our own or through third parties;
•implement and maintain operational, financial, and management systems;
•price our product candidates, if approved, at expected levels and obtain and maintain sufficient insurance and reimbursement from insurers and other payors; and
•gain broad market acceptance for our product candidates.
If we are unsuccessful in accomplishing these objectives, we may not be able to develop product candidates, raise capital, expand our business, or continue our operations. Further, even if we are successful in clinical trials of product candidates, we may choose to place further development or commercialization on hold given perceived marketing challenges or the relative differences in commercial attractiveness within our portfolio.
The results of preclinical studies and earlier clinical trials are not always predictive of future results. Any product candidate we or any of our future development partners advance into clinical trials, including reproxalap, may not have favorable results in later clinical trials, if any, or receive regulatory approval.
Drug development has inherent risk. We or any of our future development partners will be required to demonstrate through adequate and well-controlled clinical trials that our product candidates are safe and effective, with a favorable benefit-risk profile, for use in their target indications before we can seek regulatory approvals for their commercial sale. Drug development is a long, expensive, and uncertain process, and delay or failure can occur at any stage of development, including after commencement of any of our clinical trials. In addition, as product candidates proceed through development, the trial designs may often be different and may need to evolve and change from phase to phase or within the same phase or same trial, as is the case for adaptive trials; the vehicles or
48
controls may be modified from trial to trial; and the product formulations or manufacturing process may differ due to the need to test product candidate samples that can be manufactured on a commercial scale. Success in run-in cohorts, earlier clinical trials, or clinical trials focused on a different indication does not mean that later clinical trials will be successful because product candidates in later-stage clinical trials may fail to demonstrate sufficient safety or efficacy despite having progressed through other phases of clinical testing. In addition, discussions with regulatory bodies, such as the FDA, may lead to changes in trial designs or programs. Companies frequently suffer significant setbacks in advanced clinical trials, even after run-in cohorts or earlier clinical trials have shown promising results. For example, the results of the TRANQUILITY Trial did not reflect the results of the TRANQUILITY run-in cohort. Moreover, only a small percentage of drugs under development result in the submission of an NDA to the FDA and even fewer are approved for commercialization.
Because we are developing novel product candidates for the treatment of diseases in a manner which there is little clinical drug development experience and, in some cases, are designing adaptive trials or using new endpoints or methodologies, the regulatory pathways for approval are not well defined, and, as a result, there is greater risk that our clinical trials will not result in our desired outcomes or require additional trials.
Our clinical focus is on the development of new products for immune-mediated diseases. We are performing an adaptive trial in proliferative vitreoretinopathy, the GUARD trial, and may do so with other indications in the future. In an adaptive trial, the initial parts of the trial are not designed to be pivotal or definitive. Rather, the initial parts of adaptive trials are expected to provide data to guide subsequent parts of the trial, which could require design changes, including but not limited to, different endpoints. In addition, following the initial parts of adaptive trials, we may, among other things, decide to continue to the subsequent parts of the trial, conclude the trial based on its success or failure in such initial parts, or discuss the trial results and regulatory pathway with regulatory authorities prior to determining next steps with respect to the trial and development program. As such, the likelihood of success in our late-stage clinical programs cannot necessarily be predicted.
We could also face challenges in designing clinical trials and obtaining regulatory approval of our product candidates due to the lack of historical clinical trial experience for novel classes of therapeutics. Thus, it is difficult to determine whether regulatory agencies will be receptive to the approval of our product candidates, and to predict the time and costs associated with obtaining regulatory approvals. The clinical trial requirements of the FDA and other regulatory agencies and the criteria regulators use to determine the safety and efficacy of a product candidate vary substantially according to the type, complexity, novelty, and intended use and market of the potential products. The regulatory approval process for novel product candidates such as ours can be more expensive and require more time and trial data than for other, better known, or more extensively studied classes of product candidates. In addition, it is possible that, as regulatory bodies gain more familiarity with our type of products by reviewing competitor candidates, those agencies could impose new conditions on our products that we did not expect. Any inability to design clinical trials with protocols and endpoints acceptable to applicable regulatory authorities, and to obtain regulatory approvals for our product candidates, would have an adverse impact on our business, prospects, financial condition, and results of operations.
To preserve trial integrity, clinical data from the initial parts of adaptive clinical trials may not be disclosed.
Adaptive clinical trials are often performed such that the initial parts of the trial are used to determine sample size and endpoints for subsequent, possibly pivotal parts of the trial. Results from the initial parts of adaptive trials are therefore not designed to be pivotal or definitive, and, in some cases, detailed trial data may not be disclosed so as not to positively or negatively bias investigators or patients involved in subsequent parts of the trial.
We are performing an adaptive trial in proliferative vitreoretinopathy. For the reasons stated above, detailed results from the initial part of the GUARD trial may not be disclosed until the completion of subsequent parts of the trials, or until the entire adaptive trial has completed. Further, the initial parts of adaptive trials may be performed in part to assess biomarkers or surrogate markers that may require substantial time to generate, analyze, and interpret. Thus, disclosure of clinical results from the initial parts of adaptive trials may also be delayed due to the time required for biomarker or surrogate marker assessment.
49
Because some of our product candidates are, to our knowledge, new chemical entities, it is difficult to predict the time and cost of development and our ability to successfully complete clinical development of these product candidates and obtain the necessary regulatory approvals for commercialization.
Some of our product candidates are, to our knowledge, new chemical entities, and unexpected problems related to new technologies may arise that can cause us to delay, suspend, or terminate our development efforts. As a result, short and long-term safety, as well as prospects for efficacy, are not fully understood and are difficult to predict. Regulatory approvals of new product candidates can be more expensive and take longer than approvals for well-characterized or more extensively studied pharmaceutical product candidates. Following discussions with the FDA and experts in the field, we may determine that it is not cost effective for us to develop one or more of our products in certain indications or we may decide to cease development in that area or seek a strategic partner.
Because RASP is a new objective sign for the treatment of dry eye disease, there is no precedent in measuring and validating the results of studies of RASP in tears, which could impact the timing, cost of development, or interpretation of our trials.
Written meeting minutes with the FDA confirmed the use of RASP as an objective sign for the treatment of dry eye disease. However, to our knowledge, a validated assay to measure RASP in tears has not been developed and the FDA has not established guidelines for the design of trials in which RASP is measured. There is risk that the RASP assays we have developed will not accurately measure RASP or be accepted by the FDA or other regulatory bodies as validated assays.
Any termination or suspension of, or delays in the commencement or completion of, our clinical trials could result in increased costs to us, delay or limit our ability to generate revenue, and adversely affect our commercial prospects.
Delays in the commencement or completion of our planned clinical trials for reproxalap or other product candidates could significantly affect our product development costs and timeline. We do not know whether future trials will begin on time or be completed on schedule, if at all. The commencement and completion of clinical trials can be delayed for a number of reasons, including delays related to:
•the COVID-19 pandemic or responses thereto;
•the FDA, or an institutional review board, or IRB, failing to grant permission to proceed or placing a clinical trial on hold;
•subjects failing to enroll or remain in our clinical trials at the rate we expect;
•subjects choosing an alternative treatment for the indication for which we are developing reproxalap or other product candidates, or participating in competing clinical trials;
•lack of adequate funding to continue the clinical trial;
•subjects experiencing severe, serious, or unexpected drug-related adverse effects, whether drug-related or otherwise;
•a facility manufacturing reproxalap, our other product candidates, or drug product components being ordered by the FDA or other government or regulatory authorities to temporarily or permanently shut down due to violations of cGMP or other applicable requirements, or infections or cross-contaminations of product candidates in the manufacturing process;
•any changes to our manufacturing process that may be necessary or desired;
•inability to timely manufacture sufficient quantities of the applicable product candidate for a clinical trial or expiration of materials intended for use in a clinical trial;
•third-party clinical investigators losing the licenses or permits necessary to perform our clinical trials, not performing our clinical trials on our anticipated schedule or consistent with the clinical trial protocol, cGMP, or regulatory requirements, or other third parties not performing data collection or analysis in a timely or accurate manner;
50
•inspections of clinical trial sites by the FDA or the finding of regulatory violations by the FDA or IRB, that require us or others to undertake corrective action, result in suspension or termination of one or more sites or the imposition of a clinical hold in part or on the entire trial, or that prohibit us from using some or all of the data in support of our marketing applications;
•delays in shipment of clinical trial material reaching clinical sites;
•third-party contractors becoming debarred or suspended or otherwise penalized by the FDA or other government or regulatory authorities for violations of regulatory requirements, in which case we may need to find a substitute contractor, and we may not be able to use some or all of the data produced by such contractors in support of our marketing applications; or
•one or more IRBs refusing to approve, suspending, or terminating the trial at an investigational site; precluding enrollment of additional subjects; or withdrawing its approval of the trial.
Product development costs will increase if we have delays in testing or approval of reproxalap or our other product candidates or if we need to perform more, larger, or longer clinical trials than planned. Additionally, changes in regulatory requirements and policies may occur and we or our partners may need to amend clinical trial protocols to reflect these changes. Amendments may require us to resubmit our clinical trial protocols to IRBs for reexamination, which may impact the costs, timing, or successful completion of a clinical trial. If we experience delays in completion of, or if we, the FDA, or other regulatory authorities, the IRB, other reviewing entities, or any of our clinical trial sites suspend or terminate any of our clinical trials, the commercial prospects for a product candidate may be harmed and our ability to generate product revenues, if any, will be delayed. In addition, many of the factors that cause, or lead to, termination or suspension of, or a delay in the commencement or completion of, clinical trials may also ultimately lead to the denial of regulatory approval of a product candidate. Further, if one or more clinical trials are delayed, our competitors may be able to bring products to market before we do, and the commercial viability of reproxalap or other product candidates could be significantly reduced.
We may find it difficult to enroll patients in our clinical trials or identify patients during commercialization (if our products are approved by regulatory agencies) for product candidates addressing orphan or rare diseases.
As part of our business strategy, we have and continue to evaluate the development and commercialization of product candidates for the treatment of orphan and other rare diseases, including PVR and retinitis pigmentosa. We may not be able to initiate or continue clinical trials if we are unable to locate a sufficient number of eligible patients willing and able to participate in the clinical trials required by the FDA or other non-United States regulatory agencies. In addition, if others develop products for the treatment of similar diseases, we would potentially compete with them for the enrollment in rare patient populations, which may adversely impact the rate of patient enrollment in and the timely completion of our current and planned clinical trials. Any negative results or perceived negative results in clinical trials of our product candidates may make it difficult or impossible to recruit or retain patients in other clinical trials of the same product candidate. Insufficient patient enrollment may be a function of other factors, including the size and nature of the patient population, the nature of the protocol, the proximity of patients to clinical sites, the timing and magnitude of disease symptom presentation, the availability of effective treatments for the relevant disease, and the eligibility criteria for the clinical trial. Our inability to identify and enroll a sufficient number of eligible patients for any of our current or future clinical trials would result in significant delays or may require us to abandon one or more clinical trials or development program. The ongoing COVID-19 pandemic has and is expected to continue to have an impact on our ability to enroll and retain patients in our clinical trials. For instance, patient enrollment in our GUARD trial was negatively impacted as a result of limited clinical trial staffing at study sites and some patients electing to delay surgery. Delays in patient enrollment in the future as a result of these and other factors may result in increased costs or may affect the timing or outcome of our clinical trials, which could prevent us from completing these trials and adversely affect our ability to advance the development of our product candidates. For instance, in rare diseases such as PVR, lack of availability of, or difficulty recruiting or retaining a sufficient number of, patients may make it difficult or cost-prohibitive to sufficiently power our clinical trials, which may not enable us to continue development and seek regulatory approval for the applicable product candidate. Further, if our products are approved by regulatory agencies, we may not be able to identify sufficient number of patients to generate significant revenues.
51
Any product candidate we or any of our future development partners advance into clinical trials may cause unacceptable adverse events or have other properties that may delay or prevent its regulatory approval or commercialization or limit its commercial potential.
Unacceptable adverse events caused by any of our product candidates that we or others advance into clinical trials could cause us or regulatory authorities to interrupt, delay, or halt clinical trials, or impose a clinical hold, potentially resulting in the denial of regulatory approval by the FDA or other regulatory authorities for any or all targeted indications and markets. This in turn could prevent us from completing development or commercializing the affected product candidate and generating revenue from its sale.
We have not yet completed testing of any of our product candidates in humans for the treatment of the indications for which we intend to seek approval, and we currently do not know the full extent of adverse events that will be observed in subjects that receive any of our product candidates. If any of our product candidates cause unacceptable adverse events in clinical trials, which may be larger or longer than those previously conducted, we may not be able to obtain regulatory approval or commercialize such product candidate.
Final marketing approval for reproxalap or our other product candidates by the FDA or other regulatory authorities may be delayed, limited, or denied, any of which would adversely affect our ability to generate operating revenues.
After the completion of our clinical trials, assuming the results of the trials are successful, and the submission of an NDA, we cannot predict whether or when we will obtain regulatory approval to commercialize reproxalap or our other product candidates and we cannot, therefore, predict the timing of any future revenue. We cannot commercialize reproxalap or our other product candidates until the appropriate regulatory authorities have reviewed and approved the applicable applications. We cannot assure you that the regulatory agencies will complete their review processes in a timely manner or that we will obtain regulatory approval for reproxalap or our other product candidates. In addition, we may experience delays or rejections based upon additional government regulation from future legislation or administrative action or changes in FDA policy or leadership during the period of product development, clinical trials, and FDA regulatory review. If marketing approval for reproxalap or our other product candidates is delayed, limited or denied, our ability to market the product candidate, and our ability to generate product sales, would be adversely affected. Such a delay could occur because a competitor product is approved before our product and secures patent protection, market exclusivity, or both, and thereby precludes our product approval for a number of years.
Even if we obtain marketing approval for reproxalap or any other product candidate, it could be subject to restrictions or withdrawal from the market and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with our product candidates, when and if any are approved.
Even if United States regulatory approval is obtained, the FDA may still impose significant restrictions on a product’s indicated uses or marketing or impose ongoing requirements for potentially costly and time-consuming post-approval studies, post-market surveillance, or other potential additional clinical trials. Following approval, if any, of reproxalap or any other product candidate, such candidate will also be subject to ongoing FDA requirements governing the labeling, packaging, storage, distribution, safety surveillance, advertising, promotion, recordkeeping, and reporting of safety and other post-market information. In addition, manufacturers of drug products and their facilities are subject to continual review and periodic inspections by the FDA and other regulatory authorities for compliance with cGMP requirements, including those relating to quality control, quality assurance, and corresponding maintenance of records and documents. If we or a regulatory agency discovers previously unknown problems with a product, such as adverse events of unanticipated seriousness, severity, or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions on that product, the manufacturing facility or us, including requesting recall or withdrawal of the product from the market or suspension of manufacturing.
52
If we or the manufacturing facilities for reproxalap or any other product candidate that may receive regulatory approval, if any, fail to comply with applicable regulatory requirements, a regulatory agency may:
•issue warning letters or untitled letters;
•seek an injunction or impose civil or criminal penalties or monetary fines;
•suspend or withdraw regulatory approval;
•suspend any ongoing clinical trials;
•refuse to approve pending applications or supplements or applications filed by us;
•suspend or impose restrictions on operations, including costly new manufacturing requirements; or
•seize or detain products, refuse to permit the import or export of product, or request us to initiate a product recall.
The occurrence of any event or penalty described above may inhibit our ability to commercialize our product candidates and generate revenue.
The FDA has the authority to require a risk evaluation and mitigation strategy (REMS) plan as part of an NDA or after approval, which may impose further requirements or restrictions on the distribution or use of an approved drug, such as limiting prescribing to certain physicians or medical centers that have undergone specialized training, limiting treatment to patients who meet certain safe-use criteria, and requiring treated patients to enroll in a registry.
In addition, if reproxalap or any of our other product candidates is approved, our product labeling, advertising, and promotion would be subject to regulatory requirements and continuing regulatory review. The FDA strictly regulates the promotional claims that may be made about prescription products. In particular, a product may not be promoted for uses that are not approved by the FDA as reflected in the product’s approved labeling. If we receive marketing approval for a product candidate, physicians may nevertheless prescribe it to patients in a manner that is inconsistent with the approved label. If we are found to have promoted such off-label uses, we may become subject to significant liability. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant sanctions. The federal government has levied large civil and criminal fines against companies for alleged improper promotion and has enjoined several companies from engaging in off-label promotion. The government has also entered into consent decrees and Corporate Integrity Agreements under which specified promotional conduct is changed or curtailed.
Even if we receive regulatory approval for reproxalap or any other product candidate, we still may not be able to successfully commercialize, and the revenue that we generate from its sales, if any, could be limited.
Even if our product candidates receive regulatory approval, they may not gain market acceptance among physicians, patients, healthcare payors, or the medical community. Coverage and reimbursement of our product candidates by third-party payors, including government payors, is also generally necessary for commercial success. In addition, we may not be able to secure advantageous contracts with payors or price our products at the expected level or at levels that make successful commercialization viable. The pricing of our products will be subject to numerous factors, many of which are outside of our control, including the pricing of similar products. The degree of market acceptance of our product candidates will depend on a number of factors, including but not limited to:
•demonstration of clinical efficacy and safety compared to other more-established products;
•the limitation of our targeted patient populations and other limitations or warnings contained in any FDA-approved labeling;
•acceptance of a new formulations by health care providers and their patients;
•the prevalence, seriousness, and severity of any adverse effects;
•new procedures or methods of treatment that may be more effective in treating conditions for which our products are intended to treat;
53
•the safety of product candidates seen in a broader patient group, including their use outside the approved indications;
•pricing and cost-effectiveness, including the cost of treatment in relation to alternative treatments;
•the effectiveness of our or any future collaborators’ sales and marketing strategies;
•our ability to obtain and maintain sufficient, commercially advantageous, and timely third-party coverage or reimbursement from government health care programs, including Medicare and Medicaid, private health insurers and other third-party payors;
•relative convenience and ease of administration;
•the prevalence and severity of adverse events;
•the effectiveness of our sales and marketing efforts;
•unfavorable publicity; and
•the willingness of patients to pay out-of-pocket in the absence of third-party coverage.
In addition, because the active ingredient of ADX-2191 (methotrexate) is a generic drug, a generic manufacturer may be able to develop and market a competitive intravitreal formulation of methotrexate following expiration of commercial exclusivity mandated via certain orphan drug designations. Generic drug competition would have a material and adverse effect on the commercial potential of ADX-2191. Further, our ability to successfully commercialize ADX-2191, if approved, depends on a number of additional factors, including but not limited to, the level of enforcement by the FDA to ensure that compounded copies of commercially available FDA-approved products manufactured by compounding pharmacies, including compounded copies of ADX-2191, that may be in violation of the federal Drug Quality and Security Act (DQSA) and other relevant provisions of the United States Federal Food, Drug, and Cosmetic Act (FDCA), are not produced and dispensed to patients.
Moreover, we cannot predict what healthcare reform initiatives may be adopted in the future. Further federal and state legislative and regulatory developments are likely, and we expect that ongoing initiatives in the United States will increase pressure on drug pricing. Such reforms could have an adverse effect on the pricing of and anticipated revenues from our current or future product candidates for which we may obtain regulatory approval and may affect our overall financial condition and ability to develop drug candidates.
If any product candidate is approved but does not achieve an adequate level of acceptance by physicians, hospitals, healthcare payors, or patients, we may not generate sufficient revenue from that product candidate and may not become or remain profitable. Our efforts to educate the medical community and third-party payors on the benefits of reproxalap or any of our other product candidates may require significant resources and may never be successful. In addition, our ability to successfully commercialize our product candidate will depend on our ability to manufacture our products, differentiate our products from competing products and defend the intellectual property of our products. Competitors with numerous approved products may be able to negotiate pricing and reimbursement that is substantially more advantageous than that which we will be able to negotiate.
Additionally, if any of our competitors’ products are approved and are unable to gain market acceptance for any reason, there could be a market perception that products like reproxalap are not able to adequately meet an unmet medical need. If we are unable to demonstrate to physicians, hospitals, third-party payors, and patients that our products are better alternatives, we may not be able to gain market acceptance for our products at the levels we anticipate and our business may be materially harmed as a result.
If the market opportunities for reproxalap and our other product candidates are smaller than we believe they are and, if we are not able to successfully identify patients and achieve significant market share, our revenues may be adversely affected and our business may suffer.
We focus our research and product development on treatments for immune-mediated diseases. Our estimated addressable markets and market opportunities for our drug candidates are based on a variety of inputs, including data published by third parties, our own market insights and internal market intelligence, and internally generated
54
data and assumptions. We have not independently verified any third-party information and cannot be assured of its accuracy or completeness. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from treatment with our product candidates, are based on estimates. These estimates have been derived from a variety of sources, including scientific literature, surveys of clinics, or market research, and may prove to be incorrect. Further, new studies may change the estimated incidence or prevalence of these diseases. The number of patients may turn out to be lower or more difficult to identify than expected. In addition, our product candidates may not achieve commercial success due to market conditions or regulatory challenges.
Any of these factors may negatively affect our ability to generate revenues from sales of our product and our ability to achieve and maintain profitability, and as a consequence, our business may suffer. In addition, these inaccuracies or errors may cause us to misallocate capital and other critical business resources, which could harm our business.
Reimbursement may be limited or unavailable in certain market segments for our product candidates, which could make it difficult for us to sell our product candidates profitably.
Market acceptance and sales of our product candidates will depend significantly on the availability of adequate insurance coverage and reimbursement from third-party payors for any of our product candidates and may be affected by existing and future health care reform measures. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drugs they will pay for and establish reimbursement levels. The reimbursement levels may be significantly less than the currently anticipated pricing of our product candidates. As a result of negative trends in the general economy in the United States or other jurisdictions in which we may do business, these organizations may be unable to satisfy their reimbursement obligations or may delay payment. Reimbursement by a third-party payor may depend upon a number of factors including the third-party payor’s determination that use of a product candidate is:
•a covered benefit under its health plan;
•safe, effective, and medically necessary;
•appropriate for the specific patient;
•cost-effective, including cost effectiveness relative to existing contracts with other pharmaceutical companies; and
•neither experimental nor investigational.
55
Obtaining coverage and reimbursement approval for a product candidate from a government or other third-party payor is a time-consuming and costly process that could require us to provide supporting scientific, clinical, and cost effectiveness data for the use of the applicable product candidate to the payor. We may not be able to provide data sufficient to gain acceptance with respect to coverage and reimbursement. We cannot be sure that coverage or adequate reimbursement will be available for any of our product candidates. Further, we cannot be sure that reimbursement amounts will not reduce the demand for, or the price of, our product candidates. If reimbursement is not available or is available only in limited levels, we may not be able to commercialize certain of our product candidates profitably, or at all, even if approved. In recent years, through legislative and regulatory actions, the federal government has made substantial changes to the United States healthcare system, including changes to the methods for, and amounts of, Medicare reimbursement. Many members of the United States Congress have attempted to repeal and replace the Patient Protection and Affordable Care Act (PPACA), but they have been unsuccessful in doing so as of the date of the filing of this report. We cannot predict the ultimate form or timing of any repeal or replacement of PPACA or the effect such repeal or replacement would have on our business. Regardless of the impact of repeal or replacement of PPACA on us, the government has shown significant interest in pursuing healthcare reform and reducing healthcare costs. These reforms could significantly reduce payments from Medicare and Medicaid over the next ten years. Reforms or other changes to these payment systems, including modifications to the conditions on qualification for payment, bundling of payments, or the imposition of enrollment limitations on new providers, may change the availability, methods, and rates of reimbursements from Medicare, private insurers, and other third-party payers for our current and future product candidates, if any, for which we are able to obtain regulatory approval. Some of these changes and proposed changes could result in reduced reimbursement rates for such product candidates, if approved, which would adversely affect our business strategy, operations, and financial results.
As a result of legislative proposals and the trend toward managed health care in the United States, third-party payors are increasingly attempting to contain health care costs by limiting both coverage and the level of reimbursement of new drugs. Payors may also refuse to provide coverage of approved product candidates for medical indications other than those for which the FDA has granted market approvals. As a result, significant uncertainty exists as to whether and how much third-party payors will reimburse patients for use of newly approved drugs, which in turn could lower drug pricing. We expect to experience pricing pressures in connection with the sale of our product candidates due to the trend toward managed health care, the increasing influence of health maintenance organizations, larger companies contracting with payors to diminish reimbursement for competitive products, and additional legislative proposals as well as country, regional, or local healthcare budget limitations.
If we fail to develop and commercialize other product candidates, we may be unable to grow our business.
As part of our growth strategy, we plan to evaluate the development and commercialization of other therapies related to immune-mediated diseases. We will evaluate internal opportunities from our compound libraries, and also may choose to continue to in-license or acquire other product candidates, as well as commercial products, to treat patients suffering from immune-mediated disorders with high unmet medical needs and limited treatment options. These other product candidates will require additional, time-consuming development efforts prior to commercial sale, including preclinical studies, clinical trials, and approval by the FDA and/or applicable foreign regulatory authorities. In-licensed product candidates may have been unsuccessfully developed by others in indications similar to those that we may pursue. All product candidates are prone to the risks of failure that are inherent in pharmaceutical product development, including the possibility that the product candidate will not be shown to be sufficiently safe and/or effective for approval by regulatory authorities. In addition, we cannot assure you that any such products that are approved will be manufactured or produced economically, adequately priced, successfully commercialized, or widely accepted in the marketplace or be more effective than other commercially available alternatives.
56
We may not be able to qualify for or obtain various designations from regulators that would have the potential to expedite the review process of one or more of our product candidates, and even if we do receive one or more of such designations there is no guarantee that they will ultimately expedite the process, or aid in our obtaining marketing approval or provide market exclusivity.
There exist several designations that we can apply for from the FDA and other regulators that would provide us with various combinations of the potential for expedited regulatory review, certain financial incentives as well as the potential for post-approval exclusivity for a period of time. These designations include but are not limited to orphan drug designation, breakthrough therapy designation, accelerated approval, fast track status, and priority review for our product candidates. We may seek one or more of these designations for our current and future product candidates. There can be no assurance that any of our other product candidates will qualify for any of these designations. There can also be no assurance that any of our product candidates, that do qualify for these designations, will be granted such designations or that the FDA will not revoke such a designation it grants at a later date. Further, there can be no assurance that any of our product candidates that are granted such designations will ever benefit from such designations or that the FDA would not withdraw such designations once granted. Were we to receive a designation that promised a period of market exclusivity, such as orphan drug exclusivity, such exclusivity may not effectively protect the product from competition because different drugs can be approved for the same condition. Further, with respect to orphan drug status, even after an orphan drug is approved, the FDA can subsequently approve the same drug for the same condition if the FDA concludes that the later drug is clinically superior if it is shown to be safer, more effective, or makes a major contribution to patient care.
We are subject to a multitude of manufacturing risks, any of which could substantially increase our costs and limit supply of our products.
The process of manufacturing our products is complex, highly regulated, and subject to several risks, including:
•The manufacturing of compounds is extremely susceptible to product loss due to contamination, equipment failure, improper installation or operation of equipment, or vendor or operator error. Even minor deviations from normal manufacturing processes could result in reduced production yields, product defects, and other supply disruptions. If microbial, viral, or other contaminations are discovered in our products or in the manufacturing facilities in which our products are made, such manufacturing facilities may need to be closed for an extended period of time to investigate and remedy the contamination.
•The manufacturing facilities in which our products are made could be adversely affected by equipment failures, labor shortages, natural disasters, power failures, and numerous other factors.
•We and our contract manufacturers must comply with the cGMP regulations and guidelines. We and our contract manufacturers may encounter difficulties in achieving quality control and quality assurance, and may experience shortages in qualified personnel. We and our contract manufacturers are subject to inspections by the FDA and comparable agencies in other jurisdictions to confirm compliance with applicable regulatory requirements. Any failure to follow cGMP or other regulatory requirements or any delay, interruption, or other issues that arise in the manufacture, fill-finish, packaging, or storage of our products as a result of a failure of our facilities or the facilities or operations of third parties to comply with regulatory requirements or pass any regulatory authority inspection could significantly impair our ability to develop and commercialize our products, including leading to significant delays in the availability of products for our clinical studies, the termination or hold on a clinical study, or the delay or prevention of a filing or approval of marketing applications for our product candidates. Significant noncompliance could also result in the imposition of sanctions, including fines, injunctions, civil penalties, failure of regulatory authorities to grant marketing approvals for our product candidates, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of products, operating restrictions, and criminal prosecutions, any of which could damage our reputation or impair our ability to develop and commercialize our products. If we are not able to maintain regulatory compliance, we may not be permitted to market our products and/or may be subject to product recalls, seizures, injunctions, or criminal prosecution.
57
In order to conduct clinical trials of our drug candidates, we will need to manufacture them in large quantities. Quality issues may arise during scale-up activities. Our reliance on a limited number of Contract Manufacturing Organizations (CMOs), the complexity of drug manufacturing and the difficulty of scaling up a manufacturing process could cause the delay of clinical trials, regulatory submissions, required approvals or commercialization of our drug candidates, and cause us to incur higher costs and prevent us from commercializing our drug candidates successfully. Furthermore, if our CMOs fail to deliver the required commercial quality and quantities of materials on a timely basis and at commercially reasonable prices, and we are unable to secure one or more replacement CMOs capable of production in a timely manner at a substantially equivalent cost, then testing and clinical trials of that drug candidate may be delayed or infeasible, and regulatory approval or commercial launch of any resulting product may be delayed or not obtained, which could significantly harm our business. In addition, failure of CMOs to comply with regulatory and quality requirements could delay manufacturing or the review of our marketing applications.
Any adverse developments affecting manufacturing operations for our products, including as a result of the COVID-19 pandemic or responses taken thereto, may result in shipment delays; inventory shortages; lot failures; product withdrawals, recalls, approvals; or other interruptions in the supply of our products. We may also have to account for inventory write-offs and incur other charges and expenses for products that fail to meet specifications, undertake costly remediation efforts, or seek more costly manufacturing alternatives.
If our competitors develop treatments for the target indications of our product candidates that are approved more quickly than ours, marketed more successfully, or demonstrated to be safer or more effective than our product candidates, our commercial opportunity will be reduced or eliminated.
We operate in highly competitive segments of the biotechnology market. We face competition from many different sources, including commercial pharmaceutical and biotechnology enterprises, academic institutions, government agencies, and private and public research institutions. Our product candidates, if successfully developed and approved, will compete with established therapies (including generic and over-the-counter drugs) as well as with new treatments that may be introduced by our competitors. With the exception of PVR, PVRL, and retinitis pigmentosa, there are a variety of approved drugs and drug candidates in development for the indications that we intend to test. Current treatments that are used in the United States for dry eye disease include over the counter artificial tears, Restasis®, Xiidra®, CequaTM, Eysuvis®, and TyrvayaTM . In February 2022, the FDA approved the first generic versions of Restasis®, which is expected to become available in the U.S. in the near future. Many of our competitors have significantly greater financial, product candidate development, manufacturing, and marketing resources than we do. Large pharmaceutical and biotechnology companies have extensive experience in clinical testing and obtaining regulatory approval for drugs. In addition, universities and private and public research institutes could be in direct competition with us. We also may compete with these organizations to recruit management, scientists, and clinical development personnel. We will also face competition from these third parties in establishing clinical trial sites, registering subjects for clinical trials, and in identifying and in-licensing new product candidates. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
New developments, including the development of other pharmaceutical technologies and methods of treating disease, occur in the pharmaceutical and life sciences industries at a rapid pace. Developments by competitors may render our product candidates obsolete or noncompetitive. Other parties may discover and patent treatment approaches and compositions that are similar to or different from ours. Competition in drug development is intense. We anticipate that we will face intense and increasing competition as new treatments enter the market and advanced technologies become available.
Our future success depends on our ability to demonstrate and maintain a competitive advantage with respect to the design, development, and commercialization of reproxalap or our other product candidates. Inflammatory diseases may be treated with general immune suppressing therapies, including corticosteroids, some of which are generic. Our potential competitors in inflammatory diseases may be developing novel immune modulating therapies that may be safer or more effective than our product candidates.
58
If we are unable to establish and maintain sales, distribution, and marketing capabilities or enter into agreements with third parties to market, sell, and distribute our product candidates, we may be unable to generate any revenues.
We have no sales or marketing infrastructure and have no experience as a Company in the sale, marketing or distribution of biopharmaceutical products. If reproxalap or any of our other product candidates ultimately receives regulatory approval, we may not be able to effectively market and distribute the product candidate. We will have to invest significant amounts of financial and management resources to develop and maintain internal sales, distribution, and marketing capabilities, some of which will be committed prior to any confirmation that reproxalap or any of our other product candidates will be approved.
We currently expect that we may build our own sales and marketing organization to support the commercialization in the United States of product candidates for which we receive marketing approval. There are risks involved with establishing our own sales and marketing capabilities. For example, recruiting and training a sales force is expensive and time-consuming and could delay any product launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have incurred these commercialization expenses prematurely or unnecessarily. These efforts may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
If we are unable to establish our own sales and marketing capabilities and enter into arrangements with third parties to perform these services, our revenue from product sales and our profitability, if any, are likely to be lower than if we ourselves were to market and sell any products that we develop. In addition, we may not be successful in entering into arrangements with third parties to market and sell our drug candidates or may be unable to do so on terms that are acceptable to us. Any of these third parties may fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our drug candidates.
Current and future legislation may increase the difficulty and cost for us to obtain regulatory and marketing approval of and commercialize our product candidates, and may affect the prices we may obtain.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding healthcare systems that could prevent or delay marketing approval for our product candidates, restrict or regulate post-approval activities, and affect our ability to profitably sell any product candidates for which we obtain marketing approval. The pharmaceutical industry has been a particular focus of these efforts and has been significantly affected by legislative initiatives. Current laws, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we receive for any FDA approved product.
Healthcare reform measures that may be adopted in the future, may result in reductions in Medicare and other healthcare funding, more rigorous coverage criteria, new payment methodologies, and additional downward pressure on the price that we receive for any approved product and/or the level of reimbursement physicians receive for administering any approved product we might bring to market. Reductions in reimbursement levels may negatively impact the prices we receive or the frequency with which our products are prescribed or administered. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors.
To date, there have been several recent U.S. congressional inquiries and proposed and enacted state and federal legislation and regulation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient support programs, reduce the costs of drugs under Medicare, and reform government program reimbursement methodologies for drug products. For example, included in the Consolidated Appropriations Act, 2021 were several drug price reporting and transparency measures, such as a new requirement for certain Medicare plans to develop tools to display Medicare Part D prescription drug benefit information in real time and for group and health insurance issuers to report information on pharmacy benefit and drug costs to the Secretaries of the Department of Health and Human Services, the Department of Labor, and the Treasury. Additionally, both Congress and the Biden administration have each indicated willingness to continue to
59
seek new legislative and/or administrative measures to address prescription drug costs. For example, on July 9, 2021, President Biden issued an Executive Order to promote competition in the U.S. economy that included several initiatives addressing prescription drugs. Among other provisions, the Executive Order stated that the Biden administration will “support aggressive legislative reforms that would lower prescription drug prices, including by allowing Medicare to negotiate drug prices, by imposing inflation caps, and through other related reforms.” In response to the Executive Order, on September 9, 2021, the Department of Health and Human Services issued a Comprehensive Plan for Addressing High Drug Prices that identified potential legislative policies and administrative tools that Congress and the agency can pursue in order to make drug prices more affordable and equitable, improve and promote competition throughout the prescription drug industry, and foster scientific innovation. Congress has also continued to conduct inquiries into the prescription drug industry’s pricing practices.
At the state level, individual states are increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing. These include legislation and regulations regarding price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, legislative action designed to encourage importation from other countries and bulk purchasing. In addition, regional health care authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other health care programs. These measures could reduce the ultimate demand for our products, if approved, or put pressure on our product pricing.
Legislative and regulatory proposals have also been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. We cannot be sure whether additional legislative changes will be enacted, or whether the FDA regulations, guidance, or interpretations will be changed, or what the impact of such changes on the potential approval and marketing approvals of our drug candidates, if any, may be. Increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post-marketing testing and other requirements.
The continuing efforts of the government, insurance companies, managed care organizations, and other payors of healthcare services to contain or reduce costs of health care may adversely affect:
•the demand for any product candidates for which we may obtain regulatory approval;
•our ability to set a price that we believe is fair for our product candidates;
•our ability to generate revenue and achieve or maintain profitability;
•our ability to identify and establish strategic partnerships;
•the level of taxes that we are required to pay;
•the availability of capital.
If the FDA or comparable foreign regulatory authorities approve generic versions of any of our product candidates that receive marketing approval, or such authorities do not grant our product candidates appropriate periods of data or market exclusivity before approving generic versions of our product candidates, the sales of our product candidates could be adversely affected.
Once an NDA is approved, the drug covered thereby becomes a “reference-listed drug” in the FDA’s publication, “Approved Drug Products with Therapeutic Equivalence Evaluations.” Manufacturers may seek marketing approval of generic versions of reference-listed drugs through submission of abbreviated new drug applications (ANDAs) in the United States. In support of an ANDA, a generic manufacturer need not conduct clinical trials demonstrating safety and efficacy. Rather, the applicant generally must show that its drug is pharmaceutically equivalent to the reference listed drug, in that it has the same active ingredient(s), dosage form, strength, route of administration and conditions of use or labeling as the reference-listed drug, and that the generic version is bioequivalent to the reference-listed drug, meaning it is absorbed in the body at the same rate and to the same extent. Generic drugs may be significantly less costly to bring to market than the reference-listed drug and companies that produce generic drugs are generally able to offer drug products at lower prices. Thus, following the
60
introduction of a generic drug, a significant percentage of the sales of any branded product or reference-listed drug is typically lost to the generic drug.
The FDA may not approve an ANDA for a generic drug until any applicable period of non-patent exclusivity for the reference-listed drug has expired. The FDCA provides a period of five years of non-patent exclusivity for a new drug containing a new chemical entity. During the exclusivity period, the FDA may not accept for review an ANDA or a 505(b)(2) NDA submitted by another company for another version of such product candidate where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity, enforceability or non-infringement. The FDCA also provides three years of marketing exclusivity for a 505(b)(1) NDA, 505(b)(2) NDA or supplement to an approved NDA if new clinical investigations other than bioavailability studies (e.g., investigations that support new indications, dosages, or strengths of an existing drug) were conducted or sponsored by the applicant and are deemed by the FDA to be essential to the approval of the application. This three-year exclusivity covers only the conditions associated with the new clinical investigations and does not prohibit the FDA from approving competitor products for product candidates containing the original active agent for other conditions of use. Five-year and three-year exclusivity will not delay the submission or approval of a full 505(b)(1) NDA. Manufacturers may seek to launch these generic drugs following the expiration of the marketing exclusivity period, even if we still have patent protection for our drug.
In the EU and the UK, innovative medicinal products are authorized based on a full marketing authorization application (as opposed to an application for marketing authorization that relies on data in the marketing authorization dossier for another, previously approved medicinal product). Applications for marketing authorization for innovative medicinal products must contain the results of pharmaceutical tests, preclinical tests, and clinical trials conducted with the medicinal product for which marketing authorization is sought (and where applicable the result of the pediatric studies unless a waiver or a deferral has been obtained - as described further below). In the EU, these applications must be made pursuant to either Directive 2001/83/EC (for the decentralized procedure or the mutual recognition procedure) or Regulation 726/2004 (for the centralized procedure). In the UK, there are various procedures available under the new regulatory legal framework to pharmaceutical products, including the possibility of a recognized assessment conducted by the European authorities under certain circumstance or by applying directly to the UK regulatory authority (MHRA).
Where an applicant for a marketing authorization submits a full dossier containing its own pharmaceutical, pre-clinical tests and clinical trials data, and where the application does not fall within the "global marketing authorization" of an existing medicinal product, the applicant is entitled to eight years of regulatory data protection upon grant of the marketing authorization (the period starts to run from the first marketing authorization in the EU/ European Economic Area [EEA]). During this period, applicants for approval of generics or biosimilars cannot rely on data contained in the marketing authorization dossier submitted for the already authorized, or reference, medicinal product to support their application. After the expiration of the eight-year period of regulatory data protection, the reference medicinal product benefits from a further two-year period of marketing protection. During these two years of marketing protection, no generic or biosimilar medicinal product that relies upon the reference medicinal product’s dossier may be placed on the EU market, but a generic or biosimilar marketing authorization application can be submitted to the competent regulatory authorities in the EU Member States during this time. The two-year period of marketing protection can further be extended by one year if, during the first eight years of the grant of the first marketing authorization, the marketing authorization holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies. However, even if a compound is considered to be a new active substance and the innovator is able to gain the period of regulatory data protection and marketing protection, provided that no other IP or regulatory exclusivities applied, another unrelated company could also apply for a marketing authorization and market another competing medicinal product for the same therapeutic indication if such company obtained its own marketing authorization based on a separate marketing authorization application based on a full self-standing scientific data package supporting the application. The period of regulatory data protection and marketing protection applies in the UK (running from the date of the first authorization in Great Britain).
In the EU, pursuant to Regulation 1901/2006, and in the UK pursuant to the Human Medicines Regulations 2012 (as amended), marketing authorization applications must include pediatric data based on pediatric investigation
61
plans agreed with the EMA if the MAA concerns (i) a new active substance, or (ii) a new indication, pharmacological form, or route of administration (where the product is protected by a supplementary protection certificate or a patent qualifying for a supplementary certificate). Applicants may obtain waivers or deferrals to these requirements in certain circumstances (for example a waiver may be obtained if the condition only occurs in adult populations). Where required, pediatric studies must cover all sub-sets of the pediatric population for both existing and new indications, pharmacological forms and route of administrations. Limited further exclusions apply, including in relation to generic or biosimilar applications. Certain rewards may be available for completion of pediatric studies. For example, where MAAs include the results of all studies conducted in compliance with an agreed pediatric investigation plan, the holder of the patent or supplementary protection certificate may be entitled to a six-month extension to the supplementary protection certificate.
In order to obtain orphan designation in the EEA, the product must fulfill certain challenging criteria. Under Article 3 of Regulation (EC) 141/2000, a medicinal product may be designated as an orphan medicinal product if it meets the following criteria: (1) is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition; and (2) either the prevalence of such condition must not be more than five in 10,000 persons in the EU when the application is made, or without the benefits derived from orphan status, it must be unlikely that the marketing of the medicine would generate sufficient return in the EU to justify the investment needed for its development; and (3) there exists no satisfactory method of diagnosis, prevention or treatment of such condition authorized for marketing in the EU or if such a method exists, the product will be of significant benefit to those affected by the condition, as defined in Regulation (EC) 847/2000.
Products receiving orphan designation in the EU may receive 10 years of orphan market exclusivity, which can be further extended by two years if pediatric studies have been conducted in accordance with an agreed pediatric investigational plan. Applications must first satisfy the orphan designation criteria and apply for orphan designation before making the application for marketing authorization. The applicant must then successfully maintain the orphan designation at the time of the marketing authorization application in order to qualify for 10 years of orphan market exclusivity. During this 10-year period, the competent authorities of the EU Member States and European Commission may not accept applications or grant marketing authorization for other similar medicinal products for the same orphan therapeutic indication. The protection afforded by orphan market exclusivity in the EU may, in some circumstances, be circumvented by competitor products which are demonstrated not to be "similar" or which are authorized for different therapeutic indications. There may be a risk that products may be prescribed "off-label" for the orphan therapeutic indication by healthcare professions in some EU Member States.
There are also three exceptions to the orphan market exclusivity principle. Marketing authorization may be granted to a similar medicinal product for the same orphan therapeutic indication if:
•The second applicant can establish in its application that its medicinal product, although similar to the orphan medicinal product already authorized, is safer, more effective, or otherwise clinically superior;
•The holder of the marketing authorization for the original orphan medicinal product consents to a second orphan medicinal product application; or
•The holder of the marketing authorization for the original orphan medicinal product cannot supply sufficient quantities of orphan medicinal product.
An orphan product can also obtain an additional two years of orphan market exclusivity in the EU if the marketing authorization application contains the results of all pediatric studies conducted in accordance with and agreed pediatric investigation plan. The 10-year market exclusivity may be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation; for example, if the product is sufficiently profitable not to justify maintenance of market exclusivity.
The UK’s regulatory legal framework provides for similar periods of protection, namely regulatory data protection, marketing protection and market exclusivity.
It is important to note that the regulatory protection afforded to medicinal product such as data exclusivity, marketing protection, market exclusivity for orphan indications, and pediatric extension are currently under review at EU level. It is expected that the protection currently afforded in the EU will be reduced in the years to come.
62
Competition that our product candidates may face from generic versions of our product candidates could materially and adversely impact our future revenue, profitability and cash flows and substantially limit our ability to obtain a return on the investments we have made in those product candidates. Our future revenues, profitability, and cash flows could also be materially and adversely affected and our ability to obtain a return on the investments we have made in those product candidates may be substantially limited if our product candidates, if and when approved, are not afforded the appropriate periods of non-patent exclusivity.
Our operations and relationships with actual and potential customers, providers and third-party payors will be subject to applicable anti-kickback, fraud and abuse, and other healthcare laws and regulations, which could expose us to penalties including criminal sanctions, civil penalties, exclusions from government programs, contractual damages, and reputational harm, and could diminish our future profits and earnings.
Our arrangements with third-party payors, physicians, and other potential customers will subject us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we market, sell and distribute any drug candidates for which we obtain marketing approval.
Applicable U.S. federal and state healthcare laws and regulations include the following:
•the federal Anti-Kickback Statute, a criminal law, which prohibits, among other things, persons and entities from knowingly and willfully offering, paying, soliciting or receiving any remuneration, directly or indirectly, in cash or in kind, to induce or reward purchasing, leasing, ordering, or arranging for, referring, or recommending the purchase, lease, or order of any good or service for which payment may be made, in whole or in part, under federal healthcare programs such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. Violations of the federal Anti-Kickback Statute can result in significant civil monetary penalties and criminal fines, as well as imprisonment and exclusion from participation in federal healthcare programs;
•the federal civil False Claims Act, which may be enforced through civil whistleblower or qui tam actions and imposes significant civil penalties, treble damages, and potential exclusion from federal healthcare programs against individuals or entities for, among other things, knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or for making a false record or statement material to an obligation to pay the federal government or for knowingly and improperly avoiding, decreasing, or concealing an obligation to pay money to the federal government. Further, a violation of the federal Anti-Kickback Statute can serve as a basis for liability under the federal civil False Claims Act. There is also the federal Criminal False Claims Act, which is similar to the federal Civil False Claims Act and imposes criminal liability on those that make or present a false, fictitious, or fraudulent claim to the federal government;
•the federal Civil Monetary Penalties Law, which authorizes the imposition of substantial civil monetary penalties against an entity that engages in activities including, among others (1) knowingly presenting, or causing to be presented, a claim for services not provided as claimed or that is otherwise false or fraudulent in any way; (2) arranging for or contracting with an individual or entity that is excluded from participation in federal health care programs to provide items or services reimbursable by a federal health care program; (3) violations of the federal Anti-Kickback Statute; or (4) failing to report and return a known overpayment;
•federal criminal statutes created by the Health Insurance Portability and Accountability Act (HIPAA), which impose criminal liability for, among other things, knowingly and willfully executing or attempting to execute a scheme to defraud any healthcare benefit program, including private insurance plans, or, in any matter involving a healthcare benefit program, for knowingly and willfully making materially false, fictitious, or fraudulent statements in connection with the delivery of or payment for health care benefits;
•HIPAA, as amended by HITECH, and its implementing regulations, which also imposes obligations, including mandatory contractual terms, on certain types of people and entities with respect to safeguarding the privacy, security and transmission of individually identifiable health information;
63
•the FDCA which among other things, strictly regulates drug marketing, prohibits manufacturers from marketing such products for off-label use or misbranding or adulterating their products, and regulates the distribution of samples;
•the federal and state laws that require pharmaceutical manufacturers to report certain calculated product pricing metrics to the government or provide certain discounts or rebates to government authorities or private entities, often as a condition of product coverage and reimbursement under federal healthcare programs
•the federal Physician Payment Sunshine Act, which requires applicable manufacturers of covered drugs, devices, biologics, and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program, among others, to annually track and report payments and other transfers of value provided to U.S.-licensed physicians and teaching hospitals, and for reports submitted on or after January 1, 2022, physician assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, anesthesiologist assistants, and certified nurse-midwives, as well as certain ownership and investment interests held by physicians and their immediate families;
•analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, that may apply to our business practices, including sales or marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers;
•state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and relevant compliance guidance promulgated by the federal government;
•state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures;
•other state laws that prohibit various marketing-related activities, such as the provision of certain kinds of gifts or meals; require the reporting of certain pricing information, including information pertaining to and justifying price increases, or prohibit prescription drug price gouging; and certain state and local laws that require the registration of pharmaceutical sales representatives; and
•state and foreign laws that govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts.
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations, or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties; damages; fines; imprisonment; exclusion of drug candidates from government-funded healthcare programs, such as Medicare and Medicaid; disgorgement; contractual damages; reputational harm; diminished profits and future earnings; and the curtailment or restructuring of our operations. If any physicians or other healthcare providers or entities with whom we expect to do business are found not to be in compliance with applicable laws, they may also be subject to criminal, civil, or administrative sanctions, including exclusions from government-funded healthcare programs. Although effective compliance programs can mitigate the risk of investigation and prosecution for violations of these laws, these risks cannot be entirely eliminated. Any action for an alleged or suspected violation can cause us to incur significant legal expenses and divert management’s attention from the operation of the business, even if such action is successfully defended.
Providing benefits or advantages to induce or reward improper performance generally to induce or encourage the prescription, recommendation, endorsement, purchase, supply, order, or use of medicinal products is prohibited in the EU. The provision of benefits or advantages to induce or reward improper performance is governed by the national anti-bribery laws of EU Member States, and in respect of the U.K., the Bribery Act 2010. Infringement of these laws may result in substantial fines and imprisonment. EU Directive 2001/83/EC, which is the EU Directive governing medicinal products for human use, provides that, where medicinal products are being promoted to
64
healthcare professionals, no gifts, pecuniary advantages, or benefits in kind may be supplied, offered or promised to such individuals unless they are inexpensive and relevant to the practice of medicine or pharmacy. This provision was transposed into the Human Medicines Regulations 2012 and as such remains applicable in the UK.
Payments made to physicians in certain EU Member States must be publicly disclosed. In addition, agreements with healthcare professionals must often be the subject of prior notification and approval by the healthcare professional’s employer, his or her competent professional organization, and/or the regulatory authorities of individual EU Member States. These requirements are set out in national laws, industry codes, or professional codes of conduct, applicable in the EU Member States and in the UK. Failure to comply with these requirements could result in reputational risk, public reprimands, administrative penalties, fines, or imprisonment.
If we market products in a manner that violates healthcare fraud and abuse laws, or if we violate government price reporting laws, we may be subject to civil or criminal penalties.
In addition to FDA restrictions on the marketing of pharmaceutical products, several other types of state and federal healthcare fraud and abuse laws have been applied in recent years to restrict certain marketing practices in the pharmaceutical industry. These laws include false claims statutes and anti-kickback statutes. Because of the breadth of these laws and the narrowness of the safe harbors, it is possible that some of our business activities could be subject to challenge under one or more of these laws.
Federal false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the federal government or knowingly making, or causing to be made, a false statement to get a false claim paid. The federal healthcare program anti-kickback statute prohibits, among other things, knowingly and willfully offering, paying, soliciting, or receiving remuneration to induce, or in return for, purchasing, leasing, ordering, or arranging for the purchase, lease, or order of any healthcare item or service reimbursable under Medicare, Medicaid, or other federally financed healthcare programs. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand, and prescribers, purchasers, and formula managers on the other. Although there are several statutory exemptions and regulatory safe harbors protecting certain common activities from prosecution, the exemptions and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchasing, or recommending may be subject to scrutiny if they do not qualify for an exemption or safe harbor. Our practices may not in all cases meet all of the criteria for safe harbor protection from anti-kickback liability.
Over the past few years, several pharmaceutical and other healthcare companies have been prosecuted under these laws for a variety of alleged promotional and marketing activities, such as: allegedly providing free trips, free goods, sham consulting fees and grants, and other monetary benefits to prescribers; reporting to pricing services inflated average wholesale prices that were then used by federal programs to set reimbursement rates; engaging in off-label promotion that caused claims to be submitted to Medicaid for non-covered, off-label uses; and submitting inflated best price information to the Medicaid Rebate Program to reduce liability for Medicaid rebates. Most states also have statutes or regulations similar to the federal anti-kickback law and false claims laws, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Sanctions under these federal and state laws may include civil monetary penalties, exclusion of a manufacturer’s products from reimbursement under government programs, criminal fines, and imprisonment.
Inadequate funding for the FDA, the SEC, and other government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner, or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels; ability to hire and retain key personnel and accept the payment of user fees; and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of the SEC and other government agencies on which our operations may rely, including those that fund research and development activities, is subject to the political process, which is inherently fluid and unpredictable.
65
Disruptions at the FDA and other agencies, including as a result of or in response to the COVID-19 pandemic, may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA and the SEC, have had to furlough critical FDA, SEC, and other government employees and stop critical activities. If a prolonged government shutdown occurs, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, in our operations as a public company, future government shutdowns could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
The FDA’s ability to review and approve new products may be hindered by a variety of factors, including budget and funding levels; ability to hire and retain key personnel; and statutory, regulatory, and policy changes.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including budget and funding levels; ability to hire and retain key personnel; and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
The ability of the FDA and other government agencies to properly administer their functions is highly dependent on the levels of government funding and the ability to fill key leadership appointments, among various factors. Delays in filling or replacing key positions could significantly impact the ability of the FDA and other agencies to fulfill their functions, and could greatly impact healthcare and the pharmaceutical industry.
In December 2016, the 21st Century Cures Act was signed into law, and was designed to advance medical innovation and empower the FDA with the authority to directly hire positions related to drug and device development and review. In the past, the FDA was often unable to offer key leadership candidates (including scientists) competitive compensation packages as compared to those offered by private industry. The 21st Century Cures Act is designed to streamline the agency’s hiring process and enable the FDA to compete for leadership talent by expanding the narrow ranges that are provided in the existing compensation structures.
66
Disruptions at the FDA and other governmental agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our operating results and business.
Failure to obtain regulatory approval in foreign jurisdictions would prevent us from marketing and commercializing our products abroad and may limit our ability to generate revenue from product sales.
We intend to market and commercialize our product candidates internationally. To market and sell our drug candidates in jurisdictions outside the United States, we must obtain separate marketing approvals and comply with numerous and varying regulatory requirements. The approval procedure varies among countries and can involve additional testing. The time required to obtain approval may differ substantially from that required to obtain FDA approval. The regulatory approval process outside the United States generally includes all of the risks associated with obtaining FDA approval. In addition, in many countries outside the United States, we must secure product reimbursement approvals before regulatory authorities will approve the product for sale in that country. Failure to obtain foreign regulatory approvals on a timely basis or non-compliance with foreign regulatory requirements could result in significant delays, difficulties, and costs for us and could delay or prevent the introduction of our drug candidates in certain countries. Approval by the FDA does not ensure approval by regulatory authorities in other countries or jurisdictions, and approval by one regulatory authority outside the United States does not ensure approval by regulatory authorities in other countries or jurisdictions or by the FDA. We may not be able to file for marketing approvals and may not receive necessary approvals to commercialize our products in any jurisdiction, which would materially impair our ability to generate revenue.
The UK's exit from the EU continues to create political and economic uncertainty, particularly in the UK and the EU. The UK is now being treated as a "third country" by the EU and new UK legislation has taken effect. This means that some regulatory activities, such as batch testing and Qualified Person certification conducted in Great Britain is no longer recognized in the EU. However, the UK and EU have concluded a Trade and Cooperation Agreement (TCA), which has been approved by the UK Parliament, European Council and European Parliament and has limited the disruption to the supply of medicines, particularly by enabling tariff and quota-free trade between the UK and the EU (provided that the rules of origin requirements are met), and has streamlined some issues, for example by enabling mutual recognition of cGMP inspections and certificates. The regulatory framework for medicines that existed before the end of the transition period has also effectively been preserved in UK domestic legislation as "retained EU law." By retaining a snapshot of EU legislation at its core, the UK has prevented substantial divergence to the regulation of medicines (although divergence has appeared in some areas). However, some changes to the UK legislation have been immediately necessary, including the implementation of the Northern Ireland Protocol (NIP), pursuant to which, the EU pharmaceutical legal framework acquis continues to apply in Northern Ireland (subject to periodic consent of the Northern Ireland Legislative Assembly), and only products compliant with EU law can be placed in the Northern Ireland market - adding an extra layer of regulatory complexity. As companies now need to comply with a separate UK regulatory legal framework in order to commercialize medicinal products in Great Britain (namely, England, Wales and Scotland, as EU law continues to apply in Northern Ireland). The UK government is currently trying to renegotiate fundamental aspects of the NIP so this is an unpredictable area for companies in the near future. The TCA allows for future deviation from the current regulatory framework and it is not known if and/or when any deviations may occur, which may have an impact on development, manufacture, marketing authorization, commercial sales and distribution of pharmaceutical products. It is also important to note that obtaining a marketing authorization is not sufficient to gain effective access to the market in the EU and in the UK; companies still need to agree to a reimbursement price for the products and in some jurisdictions, such as the UK and Germany, a further positive recommendation from health technology on cost-effectiveness is required for the products to be actually prescribed and reimbursed by the respective national health systems (see below). If we fail to comply with the regulatory requirements in international markets and thus receive applicable marketing approvals, our target market will be reduced, our ability to realize the full market potential of our drug candidates will be harmed and our business will be adversely affected. We may not obtain foreign regulatory approvals on a timely basis, if at all. Our failure to obtain approval of any of our drug candidates by regulatory authorities in another country may significantly diminish the commercial prospects of that drug candidate and our business prospects could decline.
67
If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to limit commercialization of reproxalap or our other product candidates.
We face an inherent risk of product liability as a result of the clinical testing of reproxalap and our other product candidates and will face an even greater risk if we commercialize our product candidates. For example, we may be sued if reproxalap or our other product candidates allegedly cause injury or are found to be otherwise unsuitable during product testing, manufacturing, marketing, or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product candidate, negligence, strict liability, and a breach of warranties. Claims could also be asserted under state consumer protection acts.
If we cannot successfully defend ourselves against product liability claims, we may incur substantial liabilities or be required to limit commercialization of our product candidates. Even successful defense would require significant financial and management resources. Regardless of the merits or eventual outcome, liability claims may result in:
•decreased demand for reproxalap or our other product candidates;
•injury to our reputation;
•withdrawal of clinical trial participants;
•costs to defend the related litigation;
•a diversion of management’s time and our resources;
•substantial monetary awards to trial participants or patients;
•product recalls, withdrawals or labeling, marketing or promotional restrictions;
•the inability to continue to develop or commercialize reproxalap or our other product candidates; or
•a decline in our stock price.
We maintain product liability insurance with $5.0 million in coverage. Our inability to obtain and retain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of reproxalap or our other product candidates. Although we will maintain such insurance, any claim that may be brought against us could result in a court judgment or settlement in an amount that is not covered, in whole or in part, by our insurance or that is in excess of the limits of our insurance coverage. Our insurance policies will also have various exclusions, and we may be subject to a product liability claim for which we have no coverage. We may have to pay any amounts awarded by a court or negotiated in a settlement that exceed our coverage limitations or that are not covered by our insurance, and we may not have, or be able to obtain, sufficient capital to pay such amounts.
We and our development partners, third-party manufacturers, and suppliers use biological materials and may use hazardous materials, and any claims relating to improper handling, storage, or disposal of these materials could be time consuming or costly.
We and our development partners, third-party manufacturers, and suppliers may use hazardous materials, including chemicals and biological agents and compounds that could be dangerous to human health and safety or the environment. Our operations and the operations of our development partner, third-party manufacturers, and suppliers also produce hazardous waste products. Federal, state, and local laws and regulations govern the use, generation, manufacture, storage, handling, and disposal of these materials and wastes. Compliance with applicable environmental laws and regulations may be expensive, and current or future environmental laws and regulations may impair our product development efforts. In addition, we cannot entirely eliminate the risk of accidental injury or contamination from these materials or wastes. We do not carry specific biological or hazardous waste insurance coverage and our property, casualty, and general liability insurance policies specifically exclude coverage for damages and fines arising from biological or hazardous waste exposure or contamination. Accordingly, in the event
68
of contamination or injury we could be held liable for damages or be penalized with fines in an amount exceeding our resources, and our clinical trials or regulatory approvals could be suspended.
We and any of our future development partners will be required to report to regulatory authorities if any of our approved products cause or contribute to adverse medical events, and any failure to do so would result in sanctions that would materially harm our business.
If we and any of our future development partners are successful in commercializing our products, the FDA and foreign regulatory authorities will require that we and any of our future development partners report certain information about adverse medical events if those products may have caused or contributed to those adverse events. The timing of our obligation to report would be triggered by the date we become aware of the adverse event as well as the nature of the event. We and any of our future development partners may fail to report adverse events we become aware of within the prescribed timeframe or to perform inadequate investigations of their causes. We and any of our future development partners may also fail to appreciate that we have become aware of a reportable adverse event, especially if it is not reported to us as an adverse event or if it is an adverse event that is unexpected or removed in time from the use of our products. If we and any of our future development partners fail to comply with our reporting obligations, the FDA or a foreign regulatory authority could take enforcement action including the issuance of a Warning Letter, the requirement of a labeling change, the initiation of a criminal prosecution, the imposition of civil monetary penalties, the seizure of our products, or delay in approval or clearance of future products.
We are subject to anti-corruption laws, as well as export control laws, customs laws, sanctions laws, and other laws governing our operations. If we fail to comply with these laws, we could be subject to civil or criminal penalties, or other remedial measures and legal expenses, any of which could adversely affect our business, results of operations and financial condition.
Our operations are subject to anti-corruption laws, including the Foreign Corrupt Practices Act (FCPA), the Bribery Act and other anticorruption laws that apply in countries where we do business and may do business in the future. The FCPA, the Bribery Act, and other laws generally prohibit us, our officers, and our employees, and intermediaries from bribing, being bribed, or making other prohibited payments to government officials or other persons to obtain or retain business or gain some other business advantage. We may in the future operate in jurisdictions that pose a high risk of potential FCPA or Bribery Act violations, and we may participate in collaborations and relationships with third parties whose actions could potentially subject us to liability under the FCPA, the Bribery Act, or local anti-corruption laws. In addition, we cannot predict the nature, scope or effect of future regulatory requirements to which our international operations might be subject or the manner in which existing laws might be administered or interpreted.
We also are subject to other laws and regulations governing our international operations, including regulations administered by the governments of the United States, UK, and authorities in the EU, including applicable export control regulations, economic sanctions on countries and persons, customs requirements, and currency exchange regulations, which we collectively refer to as Trade Control Laws.
There is no assurance that we will be completely effective in ensuring our compliance with all applicable anti-corruption laws, including the FCPA, the Bribery Act, or other legal requirements including Trade Control Laws. If we are not in compliance with the FCPA, the Bribery Act, and other anti-corruption laws or Trade Control Laws, we may be subject to criminal and civil penalties, legal expenses, disgorgement, and other sanctions and remedial measures, which could have an adverse impact on our business, financial condition, results of operations, or liquidity. The SEC also may suspend or bar issuers from trading securities on U.S. exchanges for violations of the FCPA’s accounting provisions. Likewise, any investigation of any potential violations of the FCPA; the Bribery Act; or other anti-corruption laws or Trade Control Laws by U.S., U.K., or other authorities also could have an adverse impact on our reputation, our business, results of operations, or financial condition.
69
Risks Related to our Reliance on Third Parties
We rely and will continue to rely on outsourcing arrangements for many of our activities, including clinical development and supply of reproxalap and our other product candidates.
As of December 31, 2021, we had only 12 full-time employees and, as a result, we rely, and expect to continue to rely, on outsourcing arrangements for a significant portion of our activities, including clinical research, data collection and analysis, manufacturing, financial reporting and accounting, and human resources, as well as for certain functions required of publicly traded companies. We may have limited control over third parties and we cannot guarantee that any third party will perform its obligations in an effective and timely manner.
In addition, during challenging and uncertain economic environments, in tight credit markets and during public health epidemics, such as the COVID-19 pandemic, and with the political uncertainty involving Russia and Ukraine, there may be a disruption or delay in the performance of our third party contractors, suppliers, or partners. If such third parties are unable to satisfy their commitments to us, our business and results of operations would be adversely affected.
We rely on third parties to conduct our clinical trials. If any third party does not meet our deadlines or otherwise conduct the trials as required and in accordance with regulations, our clinical development programs could be delayed or unsuccessful and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates when expected, or at all.
We do not have the ability to conduct all aspects of our preclinical testing or clinical trials ourselves. We are dependent on third parties to conduct the clinical trials for reproxalap and for our other product candidates and, therefore, the timing of the initiation and completion of these trials is controlled by such third parties and may occur on substantially different timing from our estimates. Specifically, we use CROs to conduct our clinical trials and we also rely on medical institutions, clinical investigators, and consultants to conduct our trials in accordance with our clinical protocols and regulatory requirements. Our CROs, investigators, and other third parties play a significant role in the conduct of these trials and subsequent collection and analysis of data.
There is no guarantee that CROs, investigators, or other third parties on which we rely for administration and conduct of our clinical trials will devote adequate time and resources to such trials or perform as contractually required. If any of these third parties fails to meet expected deadlines, fails to adhere to our clinical protocols, or otherwise performs in a substandard manner, our clinical trials may be extended, delayed, or terminated. If any of our clinical trial sites terminates for any reason, we may experience the loss of follow-up information on subjects enrolled in our ongoing clinical trials unless we are able to transfer those subjects to another qualified clinical trial site. In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time, and may receive cash or equity compensation in connection with such services. The COVID-19 pandemic or any worsening of the global business and economic environment may have the effect of heightening or exacerbating these risks.
Some of our product candidates may be studied in clinical trials co-sponsored by organizations or agencies other than us, or in investigator-initiated clinical trials, which means we have minimal or no control over the conduct of such trials.
We currently anticipate that part of our strategy for pursuing the wide range of indications potentially addressed by our product candidates will involve investigator-initiated clinical trials. Investigator-initiated clinical trials pose similar risks as those set forth elsewhere in this “Risk Factor” section relating to our internal clinical trials. While investigator-initiated trials may provide us with clinical data that can inform our future development strategy, we generally have less control over the conduct and design of the trials. Because we are not the sponsors of investigator-initiated trials, we do not control the protocols, administration, or conduct of the trials, including follow-up with patients and ongoing collection of data after treatment. As a result, we are subject to risks associated with the way investigator-initiated trials are conducted. In particular, we may be named in lawsuits that would lead to increased costs associated with legal defense. Additional risks include difficulties or delays in communicating with investigators or administrators, procedural delays and other timing issues, and difficulties or differences in interpreting data. Third-party investigators may design clinical trials with clinical endpoints that are more difficult to achieve, or in other ways that increase the risk of negative clinical trial results compared to clinical trials that we
70
may design on our own. Negative results in investigator-initiated clinical trials could have a material adverse effect on our prospects and the perception of our product candidates. As a result, our lack of control over the conduct and timing of, and communications with the FDA regarding, investigator-sponsored trials expose us to additional risks and uncertainties, many of which are outside our control, and the occurrence of which could adversely affect the commercial prospects for our product candidates.
We rely completely on third parties to supply drug substance and manufacture drug product for our clinical trials and preclinical studies. We intend to rely on other third parties to produce commercial supplies of product candidates, and our dependence on third parties could adversely impact our business.
We are completely dependent on third-party suppliers of the drug substance and drug product for our product candidates. If third-party suppliers do not supply sufficient quantities of materials to us on a timely basis and in accordance with applicable specifications and other regulatory requirements, there could be a significant interruption of our supplies, which would adversely affect clinical development and commercialization. Furthermore, if any of our contract manufacturers cannot successfully manufacture material that conforms to our specifications within regulatory requirements, we will not be able to secure and/or maintain regulatory approval, if any, for our product candidates.
We also rely on our contract manufacturers to purchase from third-party suppliers the materials necessary to produce our product candidates for our anticipated clinical trials. We do not have any control over the process or timing of the acquisition of raw materials by our contract manufacturers. Moreover, we currently do not have agreements in place for the commercial production of these raw materials. Any significant delay in the supply of a product candidate or the raw material components thereof for an ongoing clinical trial, including as a result of the COVID-19 pandemic or the conflict between Russia and Ukraine, could considerably delay completion of that clinical trial, product candidate testing, and potential regulatory approval of that product candidate.
We do not expect to have the resources or capacity to commercially manufacture any of our proposed product candidates if approved and will likely continue to be dependent on third-party manufacturers. Our dependence on third parties to manufacture and supply clinical trial materials and any approved product candidates may adversely affect our ability to develop and commercialize our product candidates on a timely basis.
We may not be successful in establishing and maintaining development, commercial, or other strategic partnerships, which could adversely affect our ability to develop and commercialize product candidates.
We have in the past chosen, and may in the future choose, to enter into development or other strategic partnerships, including collaborations with major biotechnology or pharmaceutical companies. We face significant competition in seeking appropriate partners and the negotiation process is time consuming and complex. Moreover, we may not be successful in our efforts to establish other development partnerships or other alternative arrangements for any of our product candidates or programs because our research and development pipeline may be insufficient, our product candidates or programs may be deemed to be at too early a stage of development for collaborative effort, and/or third parties may not view our product candidates or programs as having the requisite commercial or technical potential. Even if we are successful in our efforts to establish development or commercial partnerships, the terms that we agree upon may not be favorable to us and we may not be able to maintain such partnerships if, for example, development or approval of a product candidate is delayed or sales of an approved product candidate are below expectations. Any delay in entering into development partnership agreements related to our product candidates could delay the development and commercialization of our product candidates and reduce competitiveness, if approved.
Moreover, if we fail to maintain partnerships related to our product candidates:
•the development of certain of our current or future product candidates may be terminated or delayed;
•our cash expenditures related to development and commercialization of certain of our current or future product candidates would increase significantly and we may need to seek additional financing;
•we may be required to hire additional employees or otherwise develop expertise, such as sales and marketing expertise, for which we have not budgeted; and
71
•we will bear all of the risk related to the development and commercialization of any such product candidates.
We may not realize the benefits of our current or future strategic alliances.
We have in the past, and may in the future, form strategic alliances, create joint ventures or collaborations, or enter into licensing arrangements with third parties that we believe will complement or augment our existing business, including the continued development or commercialization of reproxalap or our other product candidates. Strategic alliances may require us to incur non-recurring and other charges, increase our near- and long-term expenditures, issue securities that dilute our existing stockholders, or disrupt our management and business. In addition, we face significant competition in seeking appropriate strategic partners, and the negotiation process is time-consuming and complex. Moreover, we may not be successful in our efforts to establish a strategic partnership or other alternative arrangements for reproxalap or our other product candidates because third parties may view the risk of development failure as too significant or the commercial opportunity for our product candidate as too limited. We cannot be certain that, following a strategic transaction or license, we will achieve the revenues or specific net income that justifies such transaction.
Risks Relating to Our Intellectual Property
Our success depends on our and our licensors' ability to protect our intellectual property and our proprietary technologies.
Our commercial success depends in part on our ability to obtain and maintain patent protection and trade secret protection for our product candidates, proprietary technologies, and the use of our product candidates or proprietary technologies as well as our ability to operate without infringing upon the proprietary rights of others. There can be no assurance that our patent applications or those of our licensors will result in additional patents being issued or that issued patents will afford sufficient protection against competitors with similar technology, nor can there be any assurance that the patents issued will not be infringed, designed around, or invalidated by third parties. Even issued patents may later be found unenforceable or may be modified or revoked in proceedings instituted by third parties before various patent offices or in courts. The degree of future protection for our proprietary rights is uncertain. Only limited protection may be available and may not adequately protect our rights or permit us to gain or keep any competitive advantage. This failure to properly protect the intellectual property rights relating to these product candidates could have a material adverse effect on our financial condition and results of operations.
Composition-of-matter patents on the active pharmaceutical ingredient are generally considered to be the strongest form of intellectual property protection for pharmaceutical products, as such patents provide protection without regard to any method of use. While we have issued composition-of-matter patents in the United States and other countries for reproxalap and other product candidates, we cannot be certain that the claims in our patent applications covering composition-of-matter of early stage candidates will be considered patentable by the United States Patent and Trademark Office (USPTO) and courts in the United States or by the patent offices and courts in foreign countries, nor can we be certain that the claims in our issued composition-of-matter patents will not be found invalid or unenforceable if challenged. Method-of-use patents protect the use of a product for the specified method. This type of patent does not prevent a competitor from making and marketing a product that is identical to our product for an indication that is outside the scope of the patented method. Moreover, even if competitors do not actively promote their product for our targeted indications, physicians may prescribe these products off-label. Although off-label prescriptions may infringe or contribute to the infringement of method-of-use patents, the practice is common and such infringement is difficult to prevent or prosecute. In addition, there are possibly treatment compositions and methods that we have not conceived of or attempted to patent, and other parties may discover and patent approaches and compositions that are similar to or different from ours.
The patent application process is subject to numerous risks and uncertainties, and there can be no assurance that we or any of our future development partners will be successful in protecting our product candidates by obtaining and defending patents. These risks and uncertainties include the following:
•the USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment, and other provisions during the patent process. There are
72
situations in which noncompliance can result in abandonment or lapse of a patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. In such an event, competitors might be able to enter the market earlier than would otherwise have been the case;
•patent applications may not result in any patents being issued;
•patents that may be issued or in-licensed may be challenged, invalidated, modified, revoked, circumvented, found to be unenforceable, or otherwise may not provide any competitive advantage;
•our competitors, many of whom have substantially greater resources than we do and many of whom have made significant investments in competing technologies, may seek or may have already obtained patents that will limit, interfere with, or eliminate our ability to make, use, and sell our potential product candidates;
•there may be significant pressure on the United States government and international governmental bodies to limit the scope of patent protection both inside and outside the United States for disease treatments that prove successful, as a matter of public policy regarding worldwide health concerns; and
•countries other than the United States may have patent laws less favorable to patentees than those upheld by United States courts, allowing foreign competitors a better opportunity to create, develop, and market competing product candidates.
In addition, we rely on the protection of our trade secrets and proprietary know-how. Although we have taken steps to protect our trade secrets and unpatented know-how, including entering into confidentiality agreements with third parties, and confidential information and inventions agreements with employees, consultants, and advisors, third parties may still obtain this information or may come upon this or similar information independently. If any of these events occurs or if we otherwise lose protection for our trade secrets or proprietary know-how, the value of our trade secrets or proprietary know-how may be greatly reduced.
Claims by third parties that we infringe their proprietary rights may result in liability for damages or prevent or delay our developmental and commercialization efforts.
The biotechnology industry has been characterized by frequent litigation regarding patent and other intellectual property rights. Because patent applications are maintained in secrecy until the application is published, we may be unaware of third party patents that may be infringed by commercialization of reproxalap or our other product candidates. In addition, identification of third party patent rights that may be relevant to our technology is difficult because patent searching is imperfect due to differences in terminology among patents, incomplete databases, and the difficulty in assessing the meaning of patent claims. Any claims of patent infringement asserted by third parties would be time consuming and could likely:
•result in costly litigation;
•divert the time and attention of our technical personnel and management;
•cause development or commercialization delays;
•prevent us from commercializing reproxalap or our other product candidates until the asserted patent expires or is held finally invalid or not infringed in a court of law;
•require us to develop non-infringing technology; or
•require us to enter into royalty or licensing agreements.
Although no third party has asserted a claim of patent infringement against us, others may hold proprietary rights that could prevent reproxalap or our other product candidates from being marketed. Any patent-related legal action against us claiming damages and seeking to enjoin commercial activities relating to our product candidate or processes could subject us to potential liability for damages and require us to obtain a license to continue to manufacture or market reproxalap or our other product candidates. We cannot predict whether we would prevail in any such actions or that any license required under any of these patents would be made available on commercially acceptable terms, if at all. In addition, we cannot be sure that we could redesign our product candidate or processes
73
to avoid infringement, if necessary. Accordingly, an adverse determination in a judicial or administrative proceeding, or the failure to obtain necessary licenses, could prevent us from developing and commercializing reproxalap or our other product candidates, which could harm our business, financial condition, and operating results.
Any such claims against us could also be deemed to constitute an event of default under the loan and security agreement. In the case of a continuing event of default under the loan, Hercules, could, among other remedies, elect to declare all amounts outstanding to be immediately due and payable and terminate all commitments to extend further credit. In the event we do not or are not able to repay the obligations at the time a default occurred, Hercules may elect to commence and prosecute bankruptcy and/or other insolvency proceedings, or proceed against the collateral granted to Hercules under the loan.
Our issued patents could be found invalid or unenforceable if challenged in court.
If we or any of our future development partners were to initiate legal proceedings against a third party to enforce a patent covering one of our product candidates, or one of our future product candidates, the defendant could counterclaim that our patent is invalid and/or unenforceable. In patent litigation in the United States, defendant counterclaims alleging invalidity and/or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including lack of novelty, obviousness, or non-enablement. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld relevant information from the USPTO, or made a misleading statement during prosecution. Third parties may also raise similar claims before the USPTO, even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. With respect to validity, for example, we cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. If a defendant were to prevail on a legal assertion of invalidity and/or unenforceability, we would lose at least part, and perhaps all, of the patent protection on such product candidate. Such a loss of patent protection would have a material adverse impact on our business.
We may fail to comply with any of our obligations under existing or future agreements pursuant to which we license rights or technology, which could result in the loss of rights or technology that are material to our business.
We are a party to technology licenses, including the in-license agreement for ADX-1612 and an in-license agreement for ADX-2191, and we may enter into additional licenses in the future. Such licenses do, and may in the future, impose commercial, contingent payment, royalty, insurance, indemnification, and other obligations on us. If we fail to comply with these obligations, the licensor may have the right to terminate the license, in which event we could lose valuable rights under our collaboration agreements and our ability to develop product candidates could be impaired. Additionally, should such a license agreement be terminated for any reason, there may be a limited number of replacement licensors, and a significant amount of time may be required to transition to a replacement licensor.
74
Our rights to develop and commercialize ADX-1612 and ADX-2191 are each subject in part to the terms and conditions of a third party license, pursuant to which we have acquired exclusive rights to ADX-1612 and ADX-2191 and other intellectual property. Our rights with respect to the intellectual property to develop and commercialize ADX-1612 and ADX-2191 may terminate, in whole or in part, if we fail to meet certain milestones contained in each of our license agreements relating to the development and commercialization of ADX-1612 and ADX-2191. We may also lose our rights to develop and commercialize either of ADX-1612 or ADX-2191 if we fail to pay required milestones or royalties. In the event of an early termination of our license agreement, all rights licensed and developed by us under this agreement may be extinguished, which may have an adverse effect on our business and results of operations.
We may be subject to claims that we have wrongfully hired an employee from a competitor or that we or our employees, consultants, or agents have wrongfully used or disclosed alleged confidential information or trade secrets of their former employers.
As is common in the biotechnology and pharmaceutical industry, we engage the services of consultants to assist us in the development of our product candidates. Many of these consultants and our employees were previously employed at, or may have previously provided or may be currently providing consulting services to, other biotechnology or pharmaceutical companies including our competitors or potential competitors. We may become subject to claims that our company or an employee, consultant, or agent inadvertently or otherwise used or disclosed trade secrets or other information proprietary to their former employers or their former or current clients. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to our management team.
If we do not obtain protection under the Hatch-Waxman Amendments by extending the patent terms and obtaining data exclusivity for our product candidate, our business may be materially harmed.
Depending upon the timing, duration, and specifics of FDA marketing approval of reproxalap or other product candidates, one or more of our United States patents may be eligible for limited patent term restoration under the Drug Price Competition and Patent Term Restoration Act of 1984, referred to as the Hatch-Waxman Amendments. The Hatch-Waxman Amendments permit a patent restoration term of up to five years as compensation for patent term lost during product development and the FDA regulatory review process. However, we may not be granted an extension because of, for example, failing to apply within applicable deadlines, failing to apply prior to expiration of relevant patents, or otherwise failing to satisfy applicable requirements. Moreover, the applicable time period or the scope of patent protection afforded could be less than we request. If we are unable to obtain patent term extension or restoration or the term of any such extension is less than we request, our competitors may obtain approval of competing products following our patent expiration, and our revenue could be reduced, possibly materially.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest, and our business may be adversely affected. Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition among potential partners or customers in our markets of interest. At times, competitors may adopt trade names or trademarks similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, there could be potential trade name or trademark infringement claims brought by owners of other registered trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively, and our business may be adversely affected. Our efforts to enforce or protect our proprietary rights related to trademarks, trade secrets, domain names, copyrights, or other intellectual property may be ineffective and could result in substantial costs and diversion of resources, and could adversely impact our financial condition or results of operations.
75
Changes in United States patent law could diminish the value of patents in general, thereby impairing our ability to protect our product candidates.
As is the case with other biotechnology companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biopharmaceutical industry involves technological and legal complexity. Therefore, obtaining and enforcing biotechnology patents is costly, time consuming, and inherently uncertain. In addition, Congress may pass patent reform legislation. The Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available or weakening the rights of patent owners. In addition to increasing uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect to the value of patents, once obtained. Depending on decisions by the United States Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways that would weaken our ability to obtain new patents, or to enforce our existing patents and patents we might obtain in the future.
We may not be able to protect our intellectual property rights throughout the world.
While we have issued composition-of-matter patents covering reproxalap and certain of our other product candidates in the United States and other countries, filing, prosecuting, and defending patents on reproxalap and our other product candidates in all countries throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States may be less extensive and of significantly shorter duration than those in the United States. In addition, the laws of some foreign countries do not protect intellectual property rights to the same extent as federal and state laws in the United States. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products, and, further, may export otherwise infringing products to territories where we have patent protection, but where enforcement is not as strong as that in the United States. These products may compete with our product candidates, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to pharmaceuticals, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, could put our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Risks Related to Our Common Stock
In the absence of an active trading market for our common stock, investors may not be able to resell their shares at or above the price at which they purchased them.
In the absence of an active trading market for our common stock, investors may not be able to sell their common stock at or above the price they paid or at the time that they would like to sell. In addition, an inactive market may impair our ability to raise capital by selling shares and may impair our ability to acquire other companies or technologies by using our shares as consideration, which, in turn, could harm our business.
76
The trading price of the shares of our common stock has been and is likely to continue to be highly volatile, and purchasers of our common stock could incur substantial losses.
Our stock price has been and will likely continue to be volatile for the foreseeable future. The stock market in general and the market for biotechnology companies in particular have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. As a result of this volatility, investors may not be able to sell their common stock at or above the price they paid. The market price for our common stock may be influenced by many factors, including:
•results of clinical trials, and the results of trials of our competitors or those of other companies in our market sector;
•the results and status of our research and development and regulatory plans for our product candidates;
•the expectations of investors or securities analysts regarding our business and clinical development program, including interim or final top-line results that we may announce;
•regulatory developments in the United States and foreign countries;
•our ability to enroll and retain patients in our clinical trials;
•variations in our financial results or those of companies that are perceived to be similar to us;
•changes in the structure of healthcare payment systems, especially in light of current reforms to the United States healthcare system;
•announcements by us or our competitors of significant acquisitions, strategic partnerships, joint ventures, or capital commitments;
•market conditions in the pharmaceutical and biotechnology sectors and issuance of securities analysts’ reports or recommendations;
•sales of our stock by insiders and 5% stockholders;
•trading volume of our common stock;
•general economic, industry, regional or larger scale conflicts or geo-political actions, and market conditions other events or factors, many of which are beyond our control, including frequent and dramatic fluctuations in industry indexes that may contain or influence our stock;
•additions or departures of key personnel; and
•intellectual property, product liability, or other litigation against us.
In addition, in the past, stockholders have initiated class action lawsuits against biotechnology and pharmaceutical companies following periods of volatility in the market prices of these companies’ stock. Such litigation, if instituted against us, could cause us to incur substantial costs and divert management’s attention and resources, which could have a material adverse effect on our business, financial condition, and results of operations.
Our failure to meet the continued listing requirements of The Nasdaq Capital Market could result in a delisting of our common stock.
If we fail to satisfy the continued listing requirements of The Nasdaq Capital Market (Nasdaq), such as the corporate governance requirements or the minimum closing bid price requirement, Nasdaq may take steps to de-list our common stock. Such a delisting would likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so. In the event of a delisting, we would expect to take actions to restore our compliance with Nasdaq’s listing requirements, but we can provide no assurance that any such action taken by us would allow our common stock to become listed again, stabilize the market price or improve the liquidity of our common stock, prevent our common stock from dropping below the Nasdaq minimum bid price requirement, or prevent future non-compliance with Nasdaq’s listing requirements.
77
Because a small number of our existing stockholders own a majority of our voting stock, your ability to influence corporate matters will be limited.
As of December 31, 2021, our executive officers, directors, and greater than 5% stockholders, in the aggregate, own approximately 29% of our outstanding common stock. As a result, such persons, acting together, will have the ability to control our management and business affairs and substantially all matters submitted to our stockholders for approval, including the election and removal of directors and approval of any significant transaction. This concentration of ownership may have the effect of delaying, deferring, or preventing a change in control, impeding a merger, consolidation, takeover, or other business combination involving us, or discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of our business, even if such a transaction would benefit other stockholders.
If our shares become subject to the penny stock rules, it would become more difficult to trade our shares.
The SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are generally equity securities with a price of less than $5.00, other than securities registered on certain national securities exchanges or authorized for quotation on certain automated quotation systems, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or system. If we do not retain a listing on The Nasdaq Capital Market and if the price of our common stock is less than $5.00, our common stock will be deemed a penny stock. The penny stock rules require a broker-dealer, before a transaction in a penny stock not otherwise exempt from those rules, to deliver a standardized risk disclosure document containing specified information. In addition, the penny stock rules require that before effecting any transaction in a penny stock not otherwise exempt from those rules, a broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive (i) the purchaser’s written acknowledgment of the receipt of a risk disclosure statement; (ii) a written agreement to transactions involving penny stocks; and (iii) a signed and dated copy of a written suitability statement. These disclosure requirements may have the effect of reducing the trading activity in the secondary market for our common stock, and therefore stockholders may have difficulty selling their shares.
We do not intend to pay dividends on our common stock and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of our common stock.
We have never declared or paid any cash dividend on our common stock, and do not currently intend to do so for the foreseeable future. We currently anticipate that we will retain future earnings for the development, operation, and expansion of our business and do not anticipate declaring or paying any cash dividends for the foreseeable future. In addition, the Hercules Credit Facility currently prohibits, and any future debt financing arrangements may contain terms prohibiting or limiting the amount of, dividends that may be declared or paid on our common stock. Any return to stockholders will therefore be limited to the appreciation of their stock. Therefore, the success of an investment in shares of our common stock will depend upon any future appreciation in the value of our common stock. There is no guarantee that shares of our common stock will appreciate in value or even maintain the price at which our stockholders have purchased shares.
A substantial number of shares of our common stock could be sold into the public market in the near future, which could depress our stock price.
Sales of substantial amounts of our common stock in the public market could reduce the prevailing market prices for our common stock. Substantially all of our outstanding common stock is eligible for sale as is common stock issuable under vested and exercisable stock options. If our existing stockholders sell a large number of shares of our common stock, or the public market perceives that existing stockholders might sell shares of common stock, the market price of our common stock could decline significantly. Existing stockholder sales might also make it more difficult for us to sell additional equity securities at a time and price that we deem appropriate.
78
We are a smaller reporting company, and we cannot be certain if the reduced reporting requirements applicable to smaller reporting companies will make our common stock less attractive to investors.
We are a smaller reporting company under Rule 12b-2 of the Securities Exchange Act of 1934. For as long as we continue to be a smaller reporting company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not smaller reporting companies, including reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements. We cannot predict if investors will find our common stock less attractive because we may rely on smaller reporting company exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock, and our stock price may be more volatile.
We are incurring significant increased costs and demands upon management as a result of operating as a public company.
As a public company, and particularly if and after we cease to be a “smaller reporting company,” we incur significant legal, accounting, and other expenses that we did not incur as a private company. We ceased to be an “emerging growth company,” as defined in the JOBS Act, on December 31, 2019. As a result, we expect to incur additional expenses and to devote increased management time toward ensuring compliance with those requirements applicable to companies that are not emerging growth companies. We are subject to the reporting requirements of the Securities Exchange Act of 1934, as amended, or the Exchange Act, which require, among other things, that we file with the Securities and Exchange Commission, or the SEC, annual, quarterly and current reports with respect to our business and financial condition. In addition, the Sarbanes-Oxley Act, as well as rules subsequently adopted by the SEC and Nasdaq to implement provisions of the Sarbanes-Oxley Act, imposes significant requirements on public companies, including requiring establishment and maintenance of effective disclosure and financial controls and changes in corporate governance practices. Further, in 2010, the Dodd-Frank Wall Street Reform and Consumer Protection Act, or the Dodd-Frank Act, was enacted. There are significant corporate governance and executive compensation related provisions in the Dodd-Frank Act that require the SEC to adopt additional rules and regulations in these areas such as “say on pay” and proxy access. Stockholder activism, the current political environment, and the current high level of government intervention and regulatory reform may result in substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact the manner in which we operate our business in ways we cannot currently anticipate.
We expect the rules and regulations applicable to public companies to continue to substantially increase our legal and financial compliance costs and to make some activities more time-consuming and costly. If public company rules and regulations divert the attention of our management and personnel from other business concerns, our business, financial condition, and results of operations could be adversely affected. Increased costs associated with public company expenses will decrease our net income or increase our net loss, and may require us to reduce costs in other areas of our business or increase the prices of our products or services. For example, public company rules and regulations make it more difficult and more expensive for us to obtain director and officer liability insurance, the cost of which has continued to rise in recent years, and thus we may be required to incur substantial costs to maintain the same or similar coverage. We cannot predict or estimate the amount or timing of additional
79
costs we may incur to respond to these requirements, the impact of which could also make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees, or as executive officers.
If we fail to maintain proper and effective internal control over financial reporting in the future, our ability to produce accurate and timely financial statements could be impaired, which could harm our operating results, investors’ views of us and, as a result, the value of our common stock.
Pursuant to Section 404 of the Sarbanes-Oxley Act, our management is required to report upon the effectiveness of our internal control over financial reporting. The rules governing the standards that must be met for management to assess our internal control over financial reporting are complex and require significant documentation, testing, and possible remediation. To continue to comply with the requirements of being a reporting company under the Exchange Act, we will be required to continue to upgrade and maintain our systems including information technology; implement and maintain additional financial and management controls, reporting systems, and procedures; and hire additional accounting and finance staff. Furthermore, we rely on third-parties, including software and system providers, for ensuring our reporting obligations and effective internal controls, and to the extent these third parties fail to provide adequate service including as a result of any inability to scale to handle our growth and the imposition of these increased reporting and internal controls and procedures, we could incur material costs for upgrading or switching systems and our business could be materially affected.
However, as a smaller reporting company and a non-accelerated filer and in accordance with new SEC rules effective in 2020, our independent registered public accounting firm will not be required to attest to the effectiveness of our internal control over financial reporting pursuant to Section 404 for as long as we are not deemed an “accelerated filer” or “large accelerated filer.”
If we are unable to establish and maintain effective internal controls it could have a material adverse effect on our business, financial condition, results of operations or cash flows.
As we grow, we plan to hire additional personnel and engage in external temporary resources and may implement, document, and modify policies and procedures to maintain effective internal controls. However, we may identify deficiencies and weaknesses or fail to remediate previously identified deficiencies in our internal controls. If material weaknesses or deficiencies in our internal controls exist and go undetected or unremediated, our financial statements could contain material misstatements that, when discovered in the future, could cause us to fail to meet our future reporting obligations and cause the price of our common stock to decline. In addition, we could be subject to sanctions or investigations by the SEC or other regulatory authorities, which would require additional financial and management resources.
If securities or industry analysts do not publish research or reports or publish unfavorable research or reports about our business, our stock price and trading volume could decline.
The trading market for our common stock will depend in part on the research and reports that securities or industry analysts publish about us, our business, our market, or our competitors. If one or more of the analysts who covers us downgrades our stock, our stock price would likely decline. If one or more of these analysts ceases to cover us or fails to regularly publish reports on us, interest in our stock could decrease, which could cause our stock price or trading volume to decline.
80
Anti-takeover provisions in our charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our amended and restated certificate of incorporation and amended and restated bylaws may delay or prevent an acquisition of us or a change in our management. These provisions include:
•authorizing the issuance of “blank check” preferred stock, the terms of which may be established and shares of which may be issued without stockholder approval;
•limiting the removal of directors by the stockholders;
•creating a staggered board of directors;
•prohibiting stockholder action by written consent, thereby requiring all stockholder actions to be taken at a meeting of our stockholders;
•eliminating the ability of stockholders to call a special meeting of stockholders;
•permitting our board of directors to accelerate the vesting of outstanding option grants upon certain transactions that result in a change of control; and
•establishing advance notice requirements for nominations for election to the board of directors or for proposing matters that can be acted upon at stockholder meetings.
In addition, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which limits the ability of stockholders owning in excess of 15% of our outstanding voting stock to merge or combine with us. Although we believe these provisions collectively provide for an opportunity to obtain greater value for stockholders by requiring potential acquirors to negotiate with our board of directors, the provisions would apply even if an offer rejected by our board were considered beneficial by some stockholders. In addition, these provisions may frustrate or prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors, which is responsible for appointing the members of our management.
Our restated certificate of incorporation and amended and restated bylaws provide that the Court of Chancery of the State of Delaware and the federal district courts of the United States will be the exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers, or employees.
Our restated certificate of incorporation provides that the Court of Chancery of the State of Delaware is the exclusive forum for any derivative action or proceeding brought on our behalf, any action asserting a breach of fiduciary duty, any action asserting a claim against us arising pursuant to the Delaware General Corporation Law, our certificate of incorporation or our bylaws or any action asserting a claim against us that is governed by the internal affairs doctrine. This provision would not apply to claims brought to enforce a duty or liability created by the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. Our amended and restated bylaws further provide that the federal district courts of the United States will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act. These choices of forum provisions may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, or other employees and may discourage these types of lawsuits. Furthermore, the enforceability of similar choice of forum provisions in other companies’ certificates of incorporation or bylaws has been challenged in legal proceedings, and it is possible that a court could find these types of provisions to be inapplicable or unenforceable. While the Delaware courts have determined that such choice of forum provisions are facially valid, a stockholder may nevertheless seek to bring a claim in a venue other than those designated in the exclusive-forum provisions, and there can be no assurance that such provisions will be enforced by a court in those other jurisdictions. If a court were to find the exclusive-forum provision contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could harm our business.
81
We are subject to litigation risks.
From time to time, we may become involved in various litigation matters and claims, including regulatory proceedings, administrative proceedings, governmental investigations, and contract disputes. We may face potential claims or liability for, among other things, breach of contract, defamation, libel, fraud, or negligence. We may also face employment-related litigation, including claims of age discrimination, sexual harassment, gender discrimination, immigration violations, or other local, state, and federal labor law violations. Because of the uncertain nature of litigation and insurance coverage decisions, the outcome of such actions and proceedings cannot be predicted with certainty and an unfavorable resolution of one or more of them could have a material adverse effect on our business, financial condition, results of operations, cash flows, and the trading price of our securities. In addition, legal fees and costs associated with prosecuting and defending litigation matters could have a material adverse effect on our business, financial condition, results of operations, and the trading price of our securities.
We could be subject to securities class action litigation.
In the past, securities class action litigation has often been brought against a company following a decline in the market price of its securities. The risk of securities class action litigation is especially relevant for us because biotechnology and pharmaceutical companies have experienced significant stock price volatility in recent years. If we face such litigation, it could result in substantial costs and a diversion of management’s attention and resources, which could harm our business.
Our insurance policies are expensive and protect us only from some business risks, which leaves us exposed to significant uninsured liabilities.
We do not carry insurance for all categories of risk that our business may encounter. Some of the policies we currently maintain include general liability, product and clinical trial liability, workers’ compensation, and directors’ and officers’ insurance. We do not know, however, if we will be able to maintain existing insurance with adequate levels of coverage. Any significant, uninsured liability may require us to pay substantial amounts, which would adversely affect our working capital and results of operations.
U.S. federal income tax reform could adversely affect us.
New legislation or regulation which could affect our tax burden could be enacted by any governmental authority. We cannot predict the timing or extent of such tax-related developments which could have a negative impact on our financial results. Additionally, we use our best judgment in attempting to quantify and reserve for these tax obligations. However, a challenge by a taxing authority, our ability to utilize tax benefits such as carryforwards or tax credits, or a deviation from other tax-related assumptions could have a material adverse effect on our business, results of operations, or financial conditions.
Our business could be negatively affected as a result of the actions of activist stockholders.
Proxy contests have been waged against many companies in the biotechnology industry over the last few years. We may be particularly vulnerable to activist stockholders due to fluctuations in our stock price. If faced with a proxy contest or other type of stockholder activism, we may not be able to respond successfully to the contest or dispute, which would be disruptive to our business. Even if we are successful, our business could be adversely affected by a proxy contest or stockholder dispute involving us or our partners because:
•responding to proxy contests and other actions by activist stockholders can be costly and time-consuming, disrupting operations and diverting the attention of management and employees;
•perceived uncertainties as to future direction may result in the loss of potential acquisitions, collaborations, or in-licensing opportunities, and may make it more difficult to attract and retain qualified personnel and business partners; and
•if individuals are elected to a board of directors with a specific agenda, it may adversely affect our ability to effectively and timely implement our strategic plan and create additional value for our stockholders.
These actions could cause our stock price to experience periods of volatility.
82
None.
ITEM 2. PROPERTIES
Our offices are located in Lexington, Massachusetts. As of December 31, 2021, we had leased approximately 9,351 square feet of office space pursuant to leases that expire in 2023. Management believes that this office space is suitable and adequate to meet our anticipated near-term needs. We anticipate that following the expiration of the lease, additional or alternative space will be available at commercially reasonable terms.
ITEM 3. LEGAL PROCEEDINGS
From time to time, we may become subject to legal proceedings, claims, and litigation arising in the ordinary course of business. We currently are not a party to any threatened or pending material litigation and do not have contingency reserves established for any litigation liabilities. However, third parties might allege that we are infringing their patent rights or that we are otherwise violating their intellectual property rights, including trade names and trademarks. Such third parties may resort to litigation. We accrue contingent liabilities when it is probable that future expenditures will be made and such expenditures can be reasonably estimated.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
83
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Holders of Record
As of December 31, 2021, there were 25 holders of record of our common stock. The actual number of stockholders is greater than this number of record holders and includes stockholders who are beneficial owners but whose shares are held in street name by brokers and other nominees. This number of holders of record also does not include stockholders whose shares may be held in trust by other entities.
Dividends
We have not declared or paid any cash dividends on our common stock since our inception. We do not plan to pay dividends in the foreseeable future. Under our credit facility, we have agreed not to pay any dividends so long as it has any outstanding obligations thereunder. We currently intend to retain all available funds and any future earnings, if any, for use in the operation of our business. Any future determination to declare cash dividends will be made at the discretion of our board of directors, subject to applicable laws, and will depend on our financial condition, results of operations, capital requirements, general business conditions and other factors that our board of directors may deem relevant, and subject to the restrictions contained in future financing instruments. Consequently, stockholders will need to sell shares of our common stock to realize a return on their investment, if any.
Securities Authorized for Issuance under Equity Compensation Plans
The information required by Item 5 of Form 10-K regarding equity compensation plans is incorporated herein by reference to Item 12 of Part III of this annual report on Form 10-K.
Recent Sales of Unregistered Securities
None.
Purchases of Equity Securities by the Issuer and Affiliated Purchases
None.
ITEM 6. [RESERVED]
84
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
You should read the following discussion and analysis of our financial condition and results of operations together with our financial statements and the related notes appearing at the end of this annual report on Form 10-K. Some of the information contained in this discussion and analysis, including information with respect to our plans and strategy for our business, includes forward-looking statements that involve risks, uncertainties and assumptions. You should read the “Risk Factors” and “Special Note Regarding Forward-Looking Statements” sections of this annual report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are a biotechnology company devoted to discovering and developing innovative therapies designed to treat immune-mediated diseases. Our lead product candidate, reproxalap, is a first-in-class treatment in late-stage development for dry eye disease (DED) and allergic conjunctivitis (AC). We have additional product candidates in development for autoimmune diseases and other immune-mediated diseases, as well as for proliferative vitreoretinopathy (PVR) and other retinal diseases. We currently intend to commercialize our products directly or through collaborations. None of our product candidates have been approved for sale in the United States or elsewhere.
Our lead product candidate reproxalap is a RASP (reactive aldehyde species) modulator that has demonstrated statistically significant and clinically relevant improvements across a number of Phase 2 and Phase 3 clinical trials in DED and AC when administered topically to the eye as an ophthalmic solution. Additionally, in a Phase 1 clinical trial, ADX-629, a first-in-class orally available RASP modulator, was well-tolerated and no treatment-related adverse events were observed. Relative to subjects treated with placebo, reduction in the commonly described pro-inflammatory RASP malondialdehyde was observed in ADX-629 treated subjects. A growing body of clinical evidence supports the potential and relevance of RASP modulation as a new and differentiated mechanism of action. We have discovered and are developing additional RASP modulator. The RASP modulator platform represents a unique and novel pharmacologic approach that, unlike almost all drugs in use today, does not directly inhibit or activate a particular protein but instead targets a family of small molecules that in turn affect the activity and structure of many proteins at once. RASP modulation, therefore, has the potential to down-regulate pro-inflammatory systems or groups of proteins, and may lead to multiple beneficial clinical effects while avoiding toxicity associated with single-target inhibition or activation.
As we continue to execute on our strategy of expanding our product candidate pipeline, we may license or acquire new immune-modulating approaches with novel therapeutic potential. In January 2019, we acquired Helio Vision, Inc. (Helio) and thereby obtained rights to ADX-2191, a vitreous-compatible methotrexate formulation for intraocular injection, a serious sight-threatening retinal disease with no approved treatment and primary vitreoretinal lymphoma (PVRL), a rare cancer with no approved treatment. In addition, in December 2016, we in-licensed ADX-1612, which inhibits the protein chaperome, a mechanistically differentiated approach for the potential treatment of inflammatory diseases. In the future, we may enter into additional partnerships that facilitate the development and commercialization of our product candidates.
Since our incorporation, we have devoted substantially all of our resources to the preclinical and clinical development of our product candidates. Our ability to generate revenues largely depends upon our ability, alone or with others, to complete development of our product candidates to obtain regulatory approvals for and to manufacture, market, and sell our product candidates. The results of our operations will vary significantly from year-to-year and quarter-to-quarter, and depend on a number of factors, including risks related to our business and industry, risks relating to intellectual property and other legal matters, risks related to our common stock, and other risks that are detailed in the section of this annual report on Form 10-K entitled “Risk Factors".
In December 2018, we entered into an Open Market Sale Agreement SM (2018 Jefferies Sales Agreement) with Jefferies LLC (Jefferies), as sales agent, pursuant to which we offered and sold, shares of our common stock, for aggregate sales proceeds of $50.0 million from time to time through Jefferies. During the year ended December 31, 2020, we sold, at a volume-weighted average price of $4.62, an aggregate of 9.4 million shares of common stock
85
and received $40.7 million after deducting commissions related to the 2018 Jefferies Sales Agreement and other offering costs. As of December 31, 2020, we had sold the maximum allowable amount and no further sales may be made under the 2018 Jefferies Sales Agreement.
In March 2019, we entered into the Hercules Credit Facility, which provided for a term loan of up to $60.0 million, $15.0 million of which has been drawn-down as of December 31, 2021. In April 2021, the Hercules Credit Facility was amended to, among other things, increase the amount which may become available for draw-down prior to May 2023, subject to the satisfaction of certain conditions contained therein, from $10.0 million to $20.0 million. The Hercules Credit Facility contains customary affirmative and negative covenants and events of default. Affirmative covenants include, among others, covenants requiring us to maintain our legal existence and governmental approvals, deliver certain financial reports, and maintain insurance coverage. Negative covenants include, among others: restrictions on transferring any part of our business or intellectual property; incurring additional indebtedness; engaging in mergers or acquisitions; paying dividends or making other distributions; making investments; and creating other liens on our assets, in each case subject to customary exceptions. The Hercules Credit Facility, as amended, is described in Note 9 to the notes to the consolidated financial statements contained in this annual report on Form 10-K. As of December 31, 2021, $15.0 million was outstanding under the Hercules Credit Facility, and no additional amounts were available for borrowing.
In January 2021, we sold 7.9 million shares of our common stock in an underwritten public offering at $9.50 per share, for an aggregate gross cash purchase price of $74.7 million or proceeds of $70.0 million after underwriters’ discount, commissions, and other offering expenses.
In March 2021, we entered into an Open Market Sales Agreement SM (2021 Jefferies Sales Agreement) with Jefferies, as sales agent, pursuant to which we may offer and sell, from time to time through Jefferies, shares of common stock providing for aggregate sales proceeds of up to $100.0 million. We have no obligation to sell any shares under the 2021 Jefferies Sales Agreement, and could at any time suspend solicitations and offers under the 2021 Jefferies Sales Agreement. No sales had been made pursuant to the 2021 Jefferies Sales Agreement as of December 31, 2021.
In May and June 2021, we sold an aggregate of 10.2 million shares of our common stock at a public offering price of $12.50 per share, in an underwritten public offering, for an aggregate gross cash purchase price of $127.8 million or proceeds of approximately $119.8 million after underwriters’ discount, commissions, and other offering expenses, which included the partial exercise of the underwriters' option to purchase additional shares.
We will need to raise additional capital in the form of debt or equity or through partnerships to fund additional development of our product candidates and, subject to regulatory approval, if any, the commercialization of our product candidates, and we may in-license, acquire, or invest in complementary businesses or products. In addition, as capital resources permit, we may augment or otherwise modify the clinical development plans described herein. However, any disruption in the capital markets caused by the COVID-19 pandemic could make any financing more challenging, and there can be no assurance that we will be able to raise capital on commercially reasonable terms or at all.
Our Agreement with Madrigal
We are developing ADX-1612 pursuant to a License Agreement with Madrigal Pharmaceuticals, Inc. (Madrigal), entered into on December 26, 2016 (Madrigal Agreement). Pursuant to the Madrigal Agreement, we obtained an exclusive, worldwide license from Madrigal under certain patents and patent applications, and other licenses to intellectual property, to develop and commercialize Hsp90 inhibitors, including ADX-1612 (investigated in oncology under the name ganetespib), (collectively, Madrigal Agreement Products). We have agreed to use our commercially reasonable efforts to develop Madrigal Agreement Products.
86
In consideration for the rights licensed under the Madrigal Agreement, we paid Madrigal an upfront license fee of $250,000 and are obligated to make future regulatory and development and sales-dependent milestone payments to Madrigal of less than $340 million in the aggregate (over 80% of such amount being tied to our achievement of increasingly greater annual worldwide net sales milestones), as well as royalty payments to Madrigal at a rate which, as a percentage of net sales, is in the high single digits for products containing ADX-1612 and mid-single digits for any other Hsp90 inhibitor product. We are also obligated under the Madrigal Agreement to pay Madrigal a percentage of certain sublicense revenue that we receive in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from the mid-twenties to low-single digits based on the development stage of the product at the time of the sublicense.
The Madrigal Agreement will remain in effect until all payment obligations under the Madrigal Agreement expire. We may terminate the Madrigal Agreement in its entirety or on a Madrigal Agreement Product-by Madrigal Agreement Product basis with timely notice to Madrigal. Either party may terminate the Madrigal Agreement for uncured material breach by the other party or upon certain insolvency or bankruptcy proceedings involving the other party, both with timely notice to the other party. In addition, Madrigal has the right to terminate the Madrigal Agreement if we, our affiliates, or sublicensees interferes with, challenges the validity or enforceability of, opposes the extension of, or grant of a supplementary protection certificate with respect to any of our licensed patents under the Madrigal Agreement. In the event of an early termination of the Madrigal Agreement, all rights licensed and developed by us under the Madrigal Agreement may revert back to Madrigal. Each party has agreed to indemnify the other party for certain third party claims arising under the Madrigal Agreement.
Our Agreement with MEEI
We are developing ADX-2191 pursuant to an Exclusive License Agreement with Massachusetts Eye and Ear Infirmary (MEEI) originally entered into in July 2016 between MEEI and Helio Vision, Inc., as amended, (MEEI Agreement). We assumed the MEEI Agreement in connection with our 2019 acquisition of Helio Vision.
Pursuant and subject to the MEEI Agreement, we obtained an exclusive, worldwide license from MEEI to develop and commercialize ADX-2191 under certain patents and patent applications, and other licenses to intellectual property (MEEI Patent Rights). We have agreed to use our commercially reasonable efforts to develop ADX-2191 and to meet certain specified effort and achievement benchmarks by certain dates.
In consideration for the rights licensed under the MEEI Agreement, Helio Vision issued MEEI a number of shares of its preferred stock and Helio Vision agreed to pay non-creditable non-refundable license maintenance fees to MEEI of $15,000 on each of the second and third anniversary of the MEEI Agreement, $25,000 on each of the fourth and fifth anniversary of the MEEI Agreement and $35,000 on the sixth and each subsequent anniversary of the MEEI Agreement during the term of such agreement. In addition, Helio Vision was obligated to make future sales-dependent milestone payments to MEEI of up to the low seven figures in the aggregate, as well as royalty payments to MEEI at a rate which, as a percentage of net sales, is in the low single digits for products that incorporate or use the MEEI Patent Rights in the United States and as a percentage in the low single digits for products that incorporate or use the MEEI Patent Rights outside the United States. We are also obligated under the MEEI Agreement to pay MEEI a percentage of certain sublicense revenue that we receive in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from low-double digits to mid-single digits based on the date of the sublicense. Following our acquisition of Helio Vision, we became obligated to make any future payments owed under the MEEI Agreement. There is no additional equity consideration issuable under the MEEI Agreement.
The MEEI Agreement will remain in effect until the expiration date of the last to expire patent licensed under the MEEI Agreement. We may terminate the MEEI Agreement with timely written notice to MEEI. MEEI has the right to terminate the MEEI Agreement if we, subject to certain specified cure periods, cease all business operations with respect to licensed products, fail to pay amounts due under the MEEI Agreement, fail to comply with certain due diligence obligations, default in our obligation to maintain insurance, one of our officers is convicted of a felony
87
relating to the manufacture, use, sale or importation of licensed products, we materially breach any provisions of the MEEI Agreement or in the event of our insolvency or bankruptcy.
In the event of an early termination of the MEEI Agreement, all rights licensed and developed by us under the MEEI Agreement may revert back to MEEI. We have agreed to indemnify MEEI for certain claims that may arise under the MEEI Agreement.
Our Acquisition of Helio Vision, Inc.
On January 28, 2019, we acquired Helio. Upon the closing of the acquisition, we issued an aggregate of 1,160,444 shares of common stock to the former securityholders and an advisor of Helio. In January 2021, pursuant to the terms of the acquisition agreement, we issued an additional 246,562 shares of common stock to the former securityholders of Helio. Subject to the conditions of the acquisition agreement, we are contingently obligated to make additional payments to the former securityholders of Helio as follows: (a) $10.0 million of common stock following approval by the FDA of a new drug approval application for the prevention and/or treatment of PVR or a substantially similar label prior to the 10th anniversary of the closing date; and (b) $2.5 million of common stock following FDA of a new drug approval application for an indication (other than PVR or a substantially similar label) prior to the 12th anniversary of the closing date, provided that in no event shall we be obligated to issue more than 5,248,885 shares of common stock in the aggregate. Additionally, in the event of certain change of control or divestitures by us, certain former convertible noteholders of Helio will be entitled to a tax gross-up payment in an amount not to exceed $1.0 million.
Research and development expenses
We expense all of our research and development expenses as they are incurred. Research and development costs that are paid in advance of performance are capitalized as a prepaid expense until incurred. Research and development expenses primarily include:
•non-clinical development, preclinical research, and clinical trial and regulatory-related costs;
•expenses incurred under agreements with sites and consultants that conduct our clinical trials; and
•employee-related expenses, including salaries, benefits, travel, and stock-based compensation expense.
Substantially all of our research and development expenses to date have been incurred in connection with reproxalap. We expect our research and development expenses to increase for the foreseeable future as we advance reproxalap and other compounds through preclinical and clinical development. The process of conducting clinical trials necessary to obtain regulatory approval is costly and time consuming. We are unable to estimate with any certainty the costs we will incur in the continued development of reproxalap and our other product candidates. Clinical development timelines, the probability of success, and development costs can differ materially from expectations. We may never succeed in achieving marketing approval for our product candidates.
The costs of clinical trials may vary significantly over the life of a project owing to, but not limited to, the following:
•per patient trial costs;
•the number of sites included in the trials;
•the countries in which the trials are conducted;
•delays of, or other effects on, clinical trials resulting from the ongoing COVID-19 pandemic or for other reasons;
•the length of time required to enroll eligible patients;
•the design of the trials;
•the cost of drug manufacturing;
88
•the number of patients that participate in the trials;
•the number of doses that patients receive;
•the costs of assay development, assays, or other assessment of clinical trial endpoints;
•the cost of vehicle or active comparative agents used in trials;
•the drop-out or discontinuation rates of patients;
•potential additional safety monitoring or other studies requested by regulatory agencies;
•the duration of patient follow-up;
•the phase of development the product candidate is in; and
•the efficacy and safety profile of our product candidates.
Included in research and development are expenses associated with asset acquisitions. Assets purchased in an asset acquisition transaction are expensed as in-process research and development unless the assets acquired are deemed to have an alternative future use. Acquired in-process research and development payments are immediately expensed, and include upfront payments, as well as transaction fees and subsequent milestone payments. Development costs incurred after the asset acquisition are expensed as incurred.
We do not expect reproxalap or any of our other product candidates to be commercially available, if at all, before at least 2023.
General and administrative expenses
Our general and administrative expenses consisted primarily of employee-related expenses, including benefits and stock-based compensation for our full-time employees during the years ended December 31, 2021 and 2020. Other general and administrative expenses include professional fees for auditing, tax, and legal services, including patent related costs. We expect that general and administrative expenses will increase in the future as we expand our operating activities, continue to incur additional costs associated with being a publicly-traded company, and maintain compliance with exchange listing and SEC requirements. These increases will likely include higher consulting costs, legal fees, accounting fees, directors’ and officers’ liability insurance premiums, and fees associated with investor relations.
Other income (expense)
Total other income (expense) consists primarily of interest income we earn on interest-bearing accounts, and interest expense incurred on our outstanding debt.
Comprehensive loss
Comprehensive loss is defined as the change in equity during a period from transactions and other events and/or circumstances from non-owner sources. For the year ended December 31, 2021, comprehensive loss is equal to our net loss of $57.8 million and no unrealized loss on marketable securities. For the year ended December 31, 2020, comprehensive loss is equal to our net loss of $37.6 million and a realized loss on marketable securities of $5.9 thousand.
Critical Accounting Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our financial statements, which we have prepared in accordance with generally accepted accounting principles in the United States (US GAAP). The preparation of these financial statements requires us to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements, as well as the expenses during the reported periods. We evaluate these estimates and judgments on an ongoing basis. We base our estimates on historical experience and on various
89
other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Our actual results may differ materially from these estimates under different assumptions or conditions.
While our significant accounting policies are more fully described in Note 2 to our financial statements appearing elsewhere in this annual report on Form 10-K, we believe that the following accounting policies are the most critical in order to fully understand and evaluate our financial condition and results of operations.
Accrued Research and Development Expenses
As part of the process of preparing financial statements, we are required to estimate and accrue research and development expenses. This process involves the following:
•communicating with our applicable personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of actual cost;
•estimating and accruing expenses in our financial statements as of each balance sheet date based on facts and circumstances known to us at the time; and
•periodically confirming the accuracy of our estimates with selected service providers and making adjustments, if necessary.
Examples of estimated research and development expenses that we accrue include:
•fees paid to investigative sites in connection with clinical studies;
•fees paid to contract manufacturing organizations in connection with non-clinical development, preclinical research, and the production of clinical study materials; and
•professional service fees for consulting and related services.
We base our expense accruals related to non-clinical development, preclinical studies, and clinical trials on our estimates of the services received and efforts expended pursuant to contracts with organizations/consultants that conduct and manage clinical studies on our behalf. The financial terms of these agreements vary from contract to contract and may result in uneven payment flows. Payments under some of these contracts may depend on many factors, such as the successful enrollment of patients, site initiation, and the completion of clinical study milestones. Our service providers invoice us monthly in arrears for services performed. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If we do not identify costs that we have begun to incur, or if we underestimate or overestimate the level of services performed or the costs of these services, our actual expenses could differ from our estimates. To date, we have not experienced significant changes in our estimates of accrued research and development expenses after a reporting period. However, due to the nature of estimates, we cannot assure you that we will not make changes to our estimates in the future as we become aware of additional information about the status or conduct of our clinical trials and other research activities.
COVID-19
We are closely monitoring the impact of the COVID-19 pandemic, including the emergence and spread of variants of COVID-19, on all aspects of our business, including how the pandemic continues to impact our employees, clinical trials, development programs, supply chain, and other aspects of our operations. For example, patient enrollment in our GUARD trial was negatively impacted as a result of limited clinical trial staffing at study sites and some patients electing to delay surgery. While the COVID-19 pandemic did not have a material adverse effect on our reported results for the year ended December 31, 2021, we are unable to predict the ultimate impact that the pandemic may have on our business, future results of operations, financial position, or cash flows. The extent to which our operations may be impacted by the COVID-19 pandemic will depend largely on future developments, which are highly uncertain and cannot be accurately predicted, including new information which may emerge concerning the severity of the outbreak, including the emergence and spread of variants of COVID-19, and
90
actions by government authorities to contain the outbreak. Furthermore, the impact of a potential worsening of global economic conditions and the effects of continued disruptions to and volatility in the financial markets remains unknown.
Other Information
Net Operating Loss Carryforwards
As of December 31, 2021, we had federal and state income tax net operating loss (NOL) carryforwards of approximately $226.0 million and $219.0 million, respectively. Federal NOL carryforwards generated through December 31, 2017 and state NOL carryforwards will expire at various dates through 2041. Federal NOLs generated during the years ended December 31, 2018 and thereafter will carry forward indefinitely. As of December 31, 2021, we had federal and state research and development tax credit carryforwards of approximately $6.9 million and $1.6 million, respectively, which will expire at various dates through 2041. Additionally, as of December 31, 2021, we had a federal orphan drug tax credit carryforward of approximately $1.0 million that expires in 2041.
Future changes in federal and state tax laws pertaining to net operating loss carryforwards may also cause limitations or restrictions from us claiming such net operating losses. If the net operating loss carryforwards become unavailable to us or are fully utilized, our future taxable income will not be shielded from federal and state income taxation absent certain U.S. federal and state tax credits, and the funds otherwise available for general corporate purposes would be reduced.
Under Section 382 of the Internal Revenue Code of 1986, as amended, a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change NOLs and certain other tax assets (tax attributes) to offset future taxable income. In general, an ownership change occurs if the aggregate stock ownership of certain stockholders increases by more than 50 percentage points over such stockholders’ lowest percentage ownership during the testing period (generally three years). Transactions involving our common stock within the testing period, even those outside our control such as purchases or sales by investors, could result in an ownership change. A limitation on our ability to utilize some or all our NOLs or credits could have a material adverse effect on our results of operations and cash flows. We believe, prior to December 31, 2020 that three ownership changes occurred since inception, and that a fourth ownership change was triggered during the year ended December 31, 2021. Management believes that its aggregate Section 382 limitation (including the additional limitation for recognized "built-in gains") is sufficient so that no current impairment of its pre-ownership change tax attributes is required. Any future ownership changes, including those resulting from our recent or future financing activities, may cause our existing tax attributes to have additional limitations.
Recent Accounting Pronouncements
Recent accounting pronouncements which may be applicable to us are described in Note 2 to our Consolidated Financial Statements included in the annual report on Form 10-K.
Results of Operations
We anticipate that our results of operations will fluctuate for the foreseeable future due to several factors, including the progress of our research and development efforts, the timing and outcome of clinical trials, and regulatory requirements. Our limited operating history makes predictions of future operations difficult or impossible. Since our inception, we have incurred significant losses.
Comparison of Years Ended December 31, 2021 and 2020
Net loss. Net loss for the years ended December 31, 2021 and 2020 was approximately $57.8 million and $37.6 million, respectively. As of December 31, 2021, we had total stockholders’ equity of $205.7 million. Losses have resulted principally from costs incurred in our clinical trials and other research and development programs, amortization costs of founders’ stock related to the acquisition of Helio in January 2019, and from our general and administrative expenses.
91
Research and development expenses. Research and development expenses were $44.9 million for the year ended December 31, 2021 compared to $24.7 million for the same period in 2020. The increase of $20.2 million is primarily related to an increase in our external research and development expenditures based on planned clinical activities, partially offset by decreases in personnel related costs and $1.2 million in non-cash compensation costs in 2020 related to a portion of a contingent milestone in the Helio agreement that was recognized at December 31, 2020 and was settled in the first quarter of 2021.
General and administrative expenses. General and administrative expenses were $11.3 million for the year ended December 31, 2021, compared to $10.0 million for the year ended December 31, 2020. The increase of approximately $1.3 million is primarily related to increases in legal expenses, insurance costs, and consulting fees.
Acquired in-process research and development expenses. Acquired in-process research and development expenses were nil for the year ended December 31, 2021, compared to $1.8 million for the year ended December 31, 2020. The $1.8 million recorded during the year ended December 31, 2020 of in-process research and development expenses was associated with the January 2019 acquisition of Helio. At December 31, 2020, a contingent milestone related to Helio was determined to be probable and estimable, and we recorded $1.8 million of acquired in-process research and development expense related to a portion of the non-cash compensation costs that occurred as a result of the contingent milestone, which was settled in the first quarter of 2021.
Other income (expense). Total other income (expense) was approximately ($1.6) million for the year ended December 31, 2021, compared to ($1.6) million for the year ended December 31, 2020. Total other income (expense), for both periods, was principally comprised of interest income and interest expense.
Liquidity and Capital Resources
We have funded our operations primarily from the sale of equity securities and convertible equity securities and borrowings under credit facilities. Since inception, we have incurred operating losses and negative cash flows from operating activities and have devoted substantially all our efforts to research and development. At December 31, 2021, we had total stockholders’ equity of approximately $205.7 million and cash and cash equivalents of $229.8 million. During the year ended December 31, 2021, we had net loss of approximately $57.8 million. We expect to generate operating losses for the foreseeable future.
In December 2018, we entered into an Open Market Sale Agreement SM (2018 Jefferies Sales Agreement) with Jefferies LLC (Jefferies), as sales agent, pursuant to which we offered and sold, shares of our common stock for aggregate sales proceeds of $50.0 million from time to time through Jefferies. From January 1, 2020 through December 31, 2020, we sold, at a volume-weighted average price of $4.62, an aggregate of 9.4 million shares of common stock with net proceeds of $40.7 million after deducting commissions related to the 2018 Jefferies Sales Agreement and other offering costs. As of December 31, 2020, we had sold the maximum allowable amount and no further sales may be made under the 2018 Jefferies Sales Agreement.
In January 2021, we closed an underwritten public offering in which we sold an aggregate of 7.9 million shares of common stock at $9.50 per share. The net proceeds of the offering were approximately $70.0 million, after deducting underwriting discounts, commissions, and other offering expenses.
In March 2021, we entered into an Open Market Sales Agreement SM (2021 Jefferies Sales Agreement) with Jefferies, as sales agent, pursuant to which we may offer and sell, from time to time through Jefferies, shares of common stock providing for aggregate sales proceeds of up to $100.0 million. We have no obligation to sell any shares under the 2021 Jefferies Sales Agreement, and could at any time suspend solicitations and offers under the 2021 Jefferies Sales Agreement. No sales had been made pursuant to the 2021 Jefferies Sales Agreement as of December 31, 2021.
In May and June 2021, we sold an aggregate of 10.2 million shares of our common stock at a public offering price of $12.50 per share, in an underwritten public offering, for an aggregate gross cash purchase price of $127.8 million or proceeds of approximately $119.8 million after underwriters’ discount, commissions, and other offering expenses, which included the partial exercise of the underwriters' option to purchase additional shares.
92
In March 2019, we entered into the Hercules Credit Facility (Loan and Security Agreement), pursuant to which a term loan of up to an aggregate principal amount of $60.0 million may be made available to us. The Loan and Security Agreement provides for (i) an initial term loan advance of up to $5.0 million at our option, which expired unutilized on April 15, 2019; (ii) three additional term loan advances of up to $15.0 million each, at our option, available to us upon the occurrence of certain funding conditions prior to September 30, 2019 (2019 Tranche), March 31, 2020 (2020 Tranche), and March 31, 2021 (2021 Tranche); and (iii) a final additional term loan advance (Fourth Loan Tranche) of up to $10.0 million prior to December 31, 2021, at our option, subject to approval by Lender’s investment committee. We drew down the 2019 Tranche in full in September 2019 and the 2020 Tranche and the 2021 Tranche expired unutilized prior to us satisfying the funding conditions for such tranche. On April 20, 2021, we entered into the First Amendment (First Amendment) to Loan and Security Agreement with Hercules. The First Amendment among other things, (i) increased the Fourth Loan Tranche from $10.0 million to $20.0 million and extended the deadline for drawing down the Fourth Loan Tranche to July 1, 2022; (ii) lowered the variable per annum rate of interest on borrowings under the Loan and Security Agreement to the greater of (a) the Prime Rate plus 3.10% or (b) 8.60%; (iii) extended the expiration of the period in which interest-only payments on borrowings under the Loan and Security Agreement are required from May 1, 2021 to July 1, 2022; and (iv) following the satisfaction of certain conditions in April 2021, further extended the expiration of the interest-only period and the deadline for drawing down the Fourth Loan Tranche to May 1, 2023. As of December 31, 2021, we had not satisfied the funding conditions for the Fourth Loan Tranche.
The Loan and Security Agreement contains customary affirmative and negative covenants and events of default. Affirmative covenants include, among others, covenants requiring us to maintain our legal existence and governmental approvals, deliver certain financial reports, and maintain insurance coverage. Negative covenants include, among others: restrictions on transferring any part of our business or intellectual property; incurring additional indebtedness; engaging in mergers or acquisitions; paying dividends or making other distributions; making investments; and creating other liens on our assets, in each case subject to customary exceptions. As of December 31, 2021, $15.0 million was outstanding under the Hercules Credit Facility, and no additional amounts were available for borrowing.
Following the effective time of the First Amendment, an aggregate of $35.0 million, subject to the terms and conditions of the Loan and Security Agreement, may be made available to us for borrowing, $15.0 million of which was funded prior to the date of the First Amendment.
Based on our current operating plan, we believe that our cash and cash equivalents, as of December 31, 2021, will be sufficient to fund our currently projected operating expenses through the end of 2023, including potential NDA submissions; initial commercialization of reproxalap, if approved; and continued early and late-stage development of our product candidates in ocular and systemic immune-mediated diseases. We will need to secure additional funding in the future, from one or more equity or debt financings, collaborations, or other sources, in order to carry out all of our planned research and development activities, commercialize our product candidates, or conduct any substantial additional development requirements requested by the FDA. At this time, due to the risks inherent in the drug development process, we are unable to estimate with any certainty the costs we will incur in the continued clinical development of reproxalap and our other product candidates. Subsequent trials initiated at a later date will cost considerably more, depending on the results of our prior clinical trials, and feedback from the FDA or other third parties. Accordingly, we will continue to require substantial additional capital to continue our clinical development and potential commercialization activities. The amount and timing of our future funding requirements will depend on many factors, including but not limited to:
•the progress, costs, results, and timing of our clinical development program for reproxalap and our other product candidates, including our current and planned clinical trials;
•the need for, and the progress, costs, and results of, any additional clinical trials of reproxalap or our other product candidates that we may initiate based on the results of our planned clinical trials or discussions with the FDA, including any additional trials the FDA or other regulatory agencies may require evaluating the efficacy and safety of reproxalap and our other product candidates;
•the impact of the COVID-19 pandemic on clinical trial initiation and enrollment and other aspects of our business, results of operations, and financial position;
93
•the outcome, costs, and timing of seeking and obtaining regulatory approvals from the FDA, and any similar regulatory agencies;
•the timing and costs associated with manufacturing reproxalap and our other product candidates for clinical trials and other studies and, if approved, for commercial sale;
•our need and ability to hire additional management, development, and scientific personnel;
•the cost to maintain, expand, and defend the scope of our intellectual property portfolio, including the amount and timing of any payments we may be required to make, or that we may receive, in connection with licensing, filing, prosecuting, defending, and enforcing of any patents or other intellectual property rights;
•the timing and costs associated with establishing sales and marketing infrastructure;
•market acceptance of reproxalap and our other product candidates;
•the costs of acquiring, licensing, or investing in additional businesses, products, product candidates, and technologies; and
•our need to implement additional internal systems and infrastructure, including financial and reporting systems.
We may need or desire to obtain additional capital to finance our operations through debt, equity, or alternative financing arrangements. We may also seek capital through collaborations or partnerships with other companies. The issuance of debt could require us to grant additional liens on certain of our assets that may limit our flexibility. If we raise additional capital by issuing equity securities, the terms and prices for these financings may be much more favorable to the new investors than the terms obtained by our existing stockholders. These financings also may significantly dilute the ownership of our existing stockholders. In addition, the disruption in the capital markets caused by the COVID-19 pandemic could make any financing more challenging, and there can be no assurance that we will be able to obtain such financing on commercially reasonable terms or at all. If we are unable to obtain additional financing, we may be required to reduce the scope of our future activities, which could harm our business, financial condition, and operating results. There can be no assurance that any additional financing required in the future will be available on acceptable terms, if at all.
We will continue to incur costs as a public company including, but not limited to, costs and expenses for directors fees; increased directors and officers insurance; investor relations fees; expenses for compliance with the Sarbanes-Oxley Act of 2002 and related to rules implemented by the SEC and Nasdaq, on which our common stock is listed; and various other costs. The Sarbanes-Oxley Act of 2002 requires that we maintain effective disclosure controls and procedures and internal controls. The following table summarizes our cash flows for the years ended December 31, 2021 and 2020:
| | | | | | | | |
| | Years Ended December 31, | |
| | 2021 | | | 2020 | |
Net cash used in operating activities | | $ | (42,555,907 | ) | | $ | (37,493,455 | ) |
Net cash (used in) provided by investing activities | | | (7,806 | ) | | | 29,023,910 | |
Net cash provided by financing activities | | | 194,496,391 | | | | 41,902,026 | |
Net increase in cash and cash equivalents | | $ | 151,932,678 | | | $ | 33,432,481 | |
Operating Activities. Net cash used in operating activities was $42.6 million in 2021, compared to net cash used in operating activities of $37.5 million in 2020. The primary use of cash was to fund our operations. The increase in the amount of cash used in operating activities for 2021 as compared to 2020 was primarily due to the timing of payments for research and development expenses in both years and higher research and development expenses in 2021.
94
Investing Activities. Net cash (used in) provided by investing activities in 2021 was ($7.8) thousand, related primarily to purchases of fixed assets. Net cash provided by investing activities in 2020 of $29.0 million, related primarily to the sales and maturities of marketable securities partially offset by the purchase of marketable securities.
Financing Activities. Net cash provided by financing activities was $194.5 million for the year ended December 31, 2021, related primarily to the January and May 2021 public offerings, under which we sold an aggregate of 18.1 million shares of our common stock with net proceeds of $189.8 million after deducting commissions and other offering costs. In addition, we received $4.6 million from the proceeds from the exercise of stock options during 2021. Net cash provided by financing activities of $41.9 million for year ended 2020, related primarily the 2018 Jefferies Sales Agreement, under which we sold an aggregate of 9.4 million shares of our common stock resulting in $40.7 million in net proceeds after deducting commissions and other offering costs, and proceeds from the exercise of stock options.
Off-Balance Sheet Arrangements. Through December 31, 2021, we have not entered into and did not have any relationships with unconsolidated entities or financial collaborations, such as entities often referred to as structured finance or special purpose entities, which would have been established for the purpose of facilitating off-balance sheet arrangements or other contractually narrow or limited purpose.
Contractual Obligations and Commitments. We are a smaller reporting company as defined by Rule 12b-2 of the Securities Exchange Act of 1934 and are not required to provide the information under this item.
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Interest rates
Our exposure to market risk is currently confined to our cash, our cash equivalents, our marketable securities (if any) and our Hercules Credit Facility. We have not used derivative financial instruments for speculation or trading purposes. Because of the short-term maturities of our cash, cash equivalents and marketable securities, we do not believe that an increase in market rates would have any significant impact on the realized value of our investments. Our Hercules Credit Facility accrues interest from its date of issue at a variable annual interest rate equal to the greater of (i) 8.60% and (ii) the prime rate (as reported in the Wall Street Journal or any successor publication thereto) plus 3.10%. As of December 31, 2021, $15.0 million was outstanding under the Hercules Credit Facility.
Effects of inflation
Inflation has not had a material impact on our results of operations.
95
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The information required by this Item 8 is contained on pages 105 through 129 of this annual report on Form 10-K and is incorporated herein by reference.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Conclusion Regarding the Effectiveness of Disclosure Controls and Procedures
As of the end of the period covered by this annual report on Form 10-K, we carried out an evaluation under the supervision and with the participation of our Disclosure Committee and our management, including our Chief Executive Officer and our Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures pursuant to Exchange Act Rules 13a-15(e) and 15d-15(e). Disclosure controls are procedures that are designed to ensure that information required to be disclosed in our reports filed under the Securities Exchange Act of 1934, or the Exchange Act, such as this annual report on Form 10-K, is recorded, processed, summarized, and reported within the time periods specified by the United States Securities and Exchange Commission. Disclosure controls are also designed to ensure that such information is accumulated and communicated to our management, including our Chief Executive Officer and our Chief Financial Officer, as appropriate to allow timely decisions regarding required disclosure. Our quarterly evaluation of disclosure controls includes an evaluation of some components of our internal control over financial reporting. We also perform a separate annual evaluation of internal control over financial reporting for the purpose of providing the management report below.
The evaluation of our disclosure controls included a review of their objectives and design, our implementation of the controls and the effect of the controls on the information generated for use in this annual report on Form 10-K. In the course of the control evaluations, we reviewed data errors or control problems identified and sought to confirm that appropriate corrective actions, including process improvements, were being undertaken. This type of evaluation is performed on a quarterly basis so that the conclusions of management, including our Chief Executive Officer and our Chief Financial Officer, concerning the effectiveness of the disclosure controls can be reported in our periodic reports on Form 10-Q and Form 10-K. The overall goals of our evaluation activities are to monitor our disclosure controls and to modify them as necessary. We intend to maintain our disclosure controls as dynamic processes and procedures that we adjust as circumstances merit.
Based on our management’s evaluation (with the participation of our Chief Executive Officer and our Chief Financial Officer), as of the end of the period covered by this report, our Chief Executive Officer and our Chief Financial Officer have concluded that our disclosure controls and procedures were effective.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act. Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
96
Our management utilized the criteria established in the Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) to conduct an assessment of the effectiveness of our internal control over financial reporting as of December 31, 2021. Based on the assessment, our management has concluded that, as of December 31, 2021, our internal control over financial reporting was effective.
Attestation Report on Internal Control over Financial Reporting
This annual report on Form 10-K does not include an attestation report of our independent registered public accounting firm due to the deferral allowed given we are neither an accelerated or large accelerated filer.
Changes in Internal Control over Financial Reporting
There has been no change in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act) during the fourth quarter of 2021 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
ITEM 9B. OTHER INFORMATION
None.
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
97
PART III
ITEM 10. Directors, Executive Officers and Corporate Governance
Except as set forth below, the information required by this item will be contained in our definitive proxy statement to be filed with the SEC in connection with our 2022 Annual Meeting of Stockholders within 120 days after the conclusion of our fiscal year ended December 31, 2021 (Proxy Statement), and is incorporated in this annual report on Form 10-K by reference.
Code of Ethics and Business Conduct
Our board of directors adopted a code of ethics and business conduct that applies to each of our directors, officers and employees. The full text of our code of business conduct is posted on the Corporate Governance portion of our website at http://ir.aldeyra.com/corporate-governance. Any waiver of the code of ethics and business conduct for an executive officer or director may be granted only by our board of directors or a committee thereof and must be timely disclosed as required by applicable law. We have implemented whistleblower procedures that establish format protocols for receiving and handling complaints from employees. Any concerns regarding accounting or auditing matters reported under these procedures will be communicated promptly to the audit committee.
ITEM 11. Executive Compensation
Other than with respect to the Securities Authorized for Issuance under Equity Incentive Plans contained in Item 12 below, the information required by this item will be contained in the Proxy Statement and is incorporated in this annual report on Form 10-K by reference.
ITEM 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
Securities Authorized for Issuance under Equity Incentive Plans
The following table provides information as of December 31, 2021, with respect to shares of our common stock that may be issued, subject to certain vesting requirements, under our existing equity compensation plans, including our 2013 Equity Incentive Plan (2013 Plan), 2010 Employee, Director and Consultant Equity Incentive Plan (2010 Plan), and our 2016 Employee Stock Purchase Plan (2016 ESPP).
| | | | | | | | | | | | | | | |
| | A | | | | B | | | | C | | |
Plan Category | | Number of
Securities to
be Issued
Upon Exercise
of Outstanding
Options, Warrants,
and Rights | | | | Weighted-
Average
Exercise Price of
Outstanding
Options,
Warrants, and
Rights | | | | Number of
Securities
Remaining
Available
for Future
Issuance
Under Equity
Compensation
Plans (Excluding
Securities
Reflected in
Column (A) | | |
Equity compensation plans
approved by security holders | | | 5,124,125 | | (1) | | $ | 6.73 | | (2) | | | 4,277,860 | | (3) |
Equity compensation plans not
approved by security holders | | | — | | | | | — | | | | | — | | |
Total | | | 5,124,125 | | (1) | | $ | 6.73 | | (2) | | | 4,277,860 | | (3) |
(1)Of these shares, 501,255 were underlying then outstanding restricted stock unit awards and 4,224,323 were subject to options then outstanding under the 2013 Plan, 398,547 were subject to options then outstanding under the 2010 Plan.
98
(2)Does not take into account restricted stock units, which have no exercise price.
(3)Represents 3,044,303 shares of common stock available for issuance under our 2013 Plan and 1,233,557 shares of common stock available for issuance under our 2016 ESPP. No shares are available for future issuance under the 2010 Plan. Our 2013 Plan provides for annual increases in the number of shares available for issuance thereunder on the first day of each fiscal year equal to the lower of: (1) 6% of the total number of shares of common stock outstanding at that time; or (2) such other amount as our board of directors may determine. Our 2016 ESPP provides for annual increases in the number of shares available for issuance thereunder on the first day of each fiscal year equal to the lesser of: (1) 1% of the shares of common stock outstanding at that time; and (2) such other amount as our board of directors may determine. On January 1, 2022, an additional 3,485,526 shares became available for future issuance under the 2013 Plan and an additional 580,921 shares became available for future issuance under the 2016 ESPP. The additional shares from the annual increase on January 1, 2022 are not included in the table above.
ITEM 13. Certain Relationships and Related Party Transactions, and Director Independence
The information required by this item will be contained in the Proxy Statement and is incorporated in this annual report on Form 10-K by reference.
ITEM 14. Principal Accounting Fees and Services
The information required by this item will be contained in the Proxy Statement and is incorporated in this annual report on Form 10-K by reference.
99
PART IV
ITEM 15. Exhibits and Financial Statements Schedules
The financial statements filed as part of this annual report on Form 10-K are listed in the Index to Financial Statements. Certain schedules are omitted because they are not applicable, or not required, or because the required information is included in the financial statements or notes thereto. The Exhibits are listed in the Exhibit Index below.
EXHIBIT INDEX
| | |
Exhibit
Number | | Exhibit Title |
| | |
3.1 | | Restated Certificate of Incorporation of Registrant, (filed as Exhibit 3.1 to the Registrant’s Current Report on Form 8-K as filed on May 7, 2014, and incorporated herein by reference) |
| | |
3.2 | | Amended and Restated Bylaws of the Registrant (filed as Exhibit 3.2 to the Registrant’s Current Report on Form 8-K as filed on May 7, 2014, and incorporated herein by reference) |
| | |
4.1 | | Specimen stock certificate evidencing the shares of common stock (filed as Exhibit 4.1 to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
4.2 | | Amended & Restated Investor Rights Agreement dated as of December 20, 2012 (filed as Exhibit 4.2 to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
4.3 | | Description of Securities (filed as Exhibit 4.6 to the Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2019 (as filed on March 12, 2020, and incorporated herein by reference)) |
| | |
10.1 | | Form of Indemnity Agreement for Directors and Officers (filed as Exhibit 10.1 to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
10.2† | | Offer Letter, effective as of August 1, 2013, between the Registrant and Todd C. Brady, M.D., Ph.D. (filed as Exhibit 10.2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on January 6, 2014, and incorporated herein by reference) |
| | |
10.3† | | Offer Letter, effective November 29, 2013 between the Registrant and Todd C. Brady, M.D., Ph.D. (filed as Exhibit 10.4 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on January 6, 2014, and incorporated herein by reference) |
| | |
10.3(a)† | | Offer Letter Amendment, effective February 19, 2014 between the Registrant and Todd C. Brady, M.D., Ph.D. (filed as Exhibit 10.4(a) to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
10.4† | | 2010 Employee, Director and Consultant Equity Incentive Plan, as amended, and form of option agreement thereunder (filed as Exhibit 10.7 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on January 6, 2014, and incorporated herein by reference) |
| | |
10.5† | | 2013 Equity Incentive Plan and form of option agreement thereunder (filed as Exhibit 10.8 to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
10.5(a)† | | Form Notice of Stock Option Grant under the 2013 Equity Incentive Plan (filed as Exhibit 10.8(a) to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
100
| | |
10.5(b)† | | Form Notice of Stock Unit Award under the 2013 Equity Incentive Plan (filed as Exhibit 10.8(b) to Amendment No. 2 to the Registrant’s Registration Statement on Form S-1 (SEC File No. 333-193204), as filed on March 17, 2014, and incorporated herein by reference) |
| | |
10.6 | | Sublease dated September 12, 2014 between the Registrant and MacLean Power L.L.C. (filed as Exhibit 10.15 to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2014 (as filed on November 12, 2014, and incorporated herein by reference)) |
| | |
10.7 | | Sublease dated as of March 7, 2016 between Planck, LLC and the Registrant and Master Lease dated June 3, 2014 between WLC Three VI, L.L.C. and Plank, LLC (filed as Exhibit 10.24 to the Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2015 (as filed on March 30, 2016, and incorporated herein by reference)) |
| | |
10.8† | | Aldeyra Management Cash Incentive Plan (filed as Exhibit 10.25 to the Registrant’s Current Report on Form 8-K as filed on March 18, 2016, and incorporated herein by reference) |
| | |
10.9† | | Aldeyra Therapeutics, Inc. Amended and Restated Change in Control Plan (filed as Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2021 (as filed on August 5, 2021, and incorporated herein by reference)) |
| | |
10.10 | | Lease Agreement by and between WLC Three VI, L.L.C. and the Registrant, dated as of September 11, 2017 (filed as Exhibit 10.27 to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2017 (as filed on November 9, 2017, and incorporated herein by reference)) |
| | |
10.11 | | First Amendment to Lease between WLC Three VI, L.L.C. and the Registrant, dated as of November 27, 2017 (filed as Exhibit 10.28 to the Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2018 (as filed on March 29, 2018, and incorporated herein by reference)). |
| | |
10.12 | | Second Amendment to Lease between WLC Three VI, L.L.C. and the Registrant, dated as of October 7, 2020 (filed as Exhibit 10.33 to the Registrant’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2020 (as filed on November 5, 2020 and incorporated herein by reference)). |
| | |
10.13 | | Third Amendment to Lease between WLC Three VI, L.L.C. and the Registrant, dated as of August 12, 2021 (filed as Exhibit 10.1 to the Registrant's Quarterly Report on Form 10-Q for the quarter ended September 30, 2021 (as filed on October 26, 2021 and incorporated herein by reference)). |
| | |
10.14† | | Amendment No. 1 to the Aldeyra Therapeutics, Inc. 2013 Equity Incentive Plan (filed as Exhibit 10.26 to the Registrant’s Quarterly Report on Form 10-Q (as filed on August 10, 2016, and incorporated herein by reference)) |
| | |
10.15† | | Amendment No. 2 to the Aldeyra Therapeutics, Inc. 2013 Equity Incentive Plan (filed as Exhibit 10.29 to the Registrant’s Quarterly Form 10-Q (as filed on August 9, 2018, and incorporated herein by reference)) |
| | |
10.16† | | Offer Letter, effective as of July 30, 2018, between the Registrant and Joshua Reed (filed as Exhibit 10.30 to the Registrant’s Quarterly Form 10-Q (as filed on November 14, 2018, and incorporated herein by reference)) |
| | |
10.17† | | Aldeyra Therapeutics, Inc. 2016 Employee Stock Purchase Plan (filed as Exhibit 10.27 to the Registrant’s Quarterly Report on Form 10-Q (as filed on August 10, 2016, and incorporated herein by reference)) |
| | |
10.18 | | Agreement and Plan of Merger, dated as of January 24, 2019, by and among Aldeyra Therapeutics, Inc., Helio Vision, Inc., Halo Merger Sub, Inc., Halo Merger Sub, LLC and Josef von Rickenbach, as the Securityholder Representative (filed as Exhibit 2.1 to the Registrant’s Current Report on Form 8-K (as filed on January 29, 2019, and incorporated herein by reference)) |
| | |
10.19†* | | Offer Letter, effective as of October 21, 2015, between the Registrant and Stephen Machatha, Ph.D. |
| | |
10.19(a)†* | | Offer Letter Amendment No. 1, effective as of January 1, 2018, between the Registrant and Stephen Machatha, Ph.D. |
101
| | |
| | |
10.19(b)†* | | Offer Letter Amendment No. 2, effective as of March 23, 2021, between the Registrant and Stephen Machatha, Ph.D. |
| | |
10.20‡ | | License Agreement, dated as of December 26, 2016, by and between Registrant and Madrigal Pharmaceuticals, Inc. (filed as Exhibit 99.2 to the Registrant’s Current Report on Form 8-K (as filed on September 25, 2018, and incorporated herein by reference)) |
| | |
10.21 | | Loan and Security Agreement, dated as of March 25, 2019, by and among the Registrant, certain subsidiaries of the Registrant from time to time party thereto, the several banks and other financial institutions or entities from time to time parties thereto and Hercules Capital, Inc. (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K (as filed on March 26, 2019, and incorporated herein by reference)) |
| | |
10.22 | | First Amendment to Loan and Security Agreement, dated April 20, 2021, by and among the Registrant, Helio Vision, LLC, the several banks and other financial institutions or entities from time to time parties thereto and Hercules Capital, Inc. (filed as Exhibit 10.1 to the Registrant’s Current Report on Form 8-K as filed on April 21, 2021, and incorporated herein by reference). |
| | |
10.23** | | Exclusive License Agreement, effective as of July 7, 2016, between the Massachusetts Eye and Ear Infirmary and Helio Vision, Inc. (filed as Exhibit 10.39 to the Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2019 (as filed on March 12, 2020, and incorporated herein by reference)) |
| | |
10.24** | | Amendment Number 1 and Waiver Agreement dated December 20, 2018 by and between Helio Vision, Inc. and the Massachusetts Eye and Ear Infirmary (filed as Exhibit 10.40 to the Registrant’s Annual Report on Form 10-K for the fiscal year ended December 31, 2019 (as filed on March 12, 2020, and incorporated herein by reference)) |
| | |
23.1* | | Consent of BDO USA, LLP, independent registered public accounting firm |
| | |
31.1* | | Certification of the Chief Executive Officer, as required by Section 302 of the Sarbanes-Oxley Act of 2002 |
| | |
31.2* | | Certification of the Chief Financial Officer as required by Section 302 of the Sarbanes-Oxley Act of 2002 |
| | |
32.1* | | Certifications of the Chief Executive Officer and Chief Financial Officer as required by 18 U.S.C. 1350 |
| | |
101.INS* | | Inline XBRL Instance Document |
| | |
101.SCH* | | Inline XBRL Taxonomy Extension Schema Document |
| | |
101.CAL* | | Inline XBRL Taxonomy Extension Calculation Linkbase Document |
| | |
101.DEF* | | Inline XBRL Taxonomy Extension Definition Linkbase Document |
| | |
101.LAB* | | Inline XBRL Taxonomy Extension Label Linkbase Document |
| | |
101.PRE* | | Inline XBRL Taxonomy Extension Presentation Linkbase Document |
104 | | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
† Compensation Arrangement.
‡ Confidential treatment has been granted with respect to certain portions of this document.
* Filed herewith.
** In accordance with Item 601(b)(2)(ii) of Regulation S-K, certain information (indicated by “*****”) has been excluded from this exhibit because it is both not material and would likely cause competitive harm to the Company if publicly disclosed.
102
The Exhibits listed in the Exhibit Index are filed as part of this annual report on Form 10-K.
ITEM 16. Form 10-K Summary
None.
103
Signatures
Pursuant to the requirements of Section 13 and 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this annual report on Form 10-K to be signed on its behalf by the undersigned, thereunto duly authorized, in the Commonwealth of Massachusetts, on March 17, 2022.
| |
ALDEYRA THERAPEUTICS, INC. |
| |
By: | /s/ Todd C. Brady, M.D., Ph.D. |
| Todd C. Brady, M.D., Ph.D. |
| President and Chief Executive Officer |
Pursuant to the requirements of the Securities Act of 1934, this annual report on Form 10-K has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| | | | |
Signature | | Title | | Date |
| | | | |
/s/ Todd C. Brady, M.D., Ph.D. | | Chief Executive Officer and Director | | March 17, 2022 |
Todd C. Brady, M.D., Ph.D. | | (principal executive officer) | | |
| | | | |
/s/ Joshua Reed | | Chief Financial Officer | | March 17, 2022 |
Joshua Reed | | (principal financial and accounting officer) | | |
| | | | |
/s/ Richard H. Douglas, Ph. D. | | Chairman of the Board of Directors | | March 17, 2022 |
Richard H. Douglas, Ph.D. | | | | |
| | | | |
/s/ Ben Bronstein, M.D. | | Director | | March 17, 2022 |
Ben Bronstein, M.D. | | | | |
| | | | |
/s/ Martin J. Joyce | | Director | | March 17, 2022 |
Martin J. Joyce | | | | |
| | | | |
/s/ Nancy Miller-Rich | | Director | | March 17, 2022 |
Nancy Miller-Rich | | | | |
| | | | |
/s/ Gary Phillips, M.D. | | Director | | March 17, 2022 |
Gary Phillips, M.D. | | | | |
| | | | |
/s/ Neal Walker, D.O. | | Director | | March 17, 2022 |
Neal Walker, D.O. | | | | |
104
ALDEYRA THERAPEUTICS, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
105
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Stockholders and Board of Directors
Aldeyra Therapeutics, Inc.
Lexington, Massachusetts
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Aldeyra Therapeutics, Inc. and subsidiaries (the “Company”) as of December 31, 2021 and 2020, the related consolidated statements of operations, comprehensive loss, stockholders’ equity, and cash flows for each of the two years in the period ended December 31, 2021, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2021 and 2020, and the results of their operations and their cash flows for each of the two years in the period ended December 31, 2021, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that was communicated or required to be communicated to the audit committee and that: (1) relates to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of the critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing separate opinions on the critical audit matter or on the accounts or disclosures to which it relates.
Accrual for Research and Development Costs Related to Clinical Trial Activities
As described in Note 2 and disclosed in Note 8 to the consolidated financial statements, the Company’s accrued research and development costs balance was $8.4 million at December 31, 2021. This accrual includes liabilities for clinical trial activities including clinical studies. Clinical studies are primarily managed internally, with
106
the assistance of third-party service providers, including contract research organizations. The accrual for clinical trial activities is based on an estimate of the percentage of activities completed to date, contractual rates, and amounts invoiced and paid to date.
We identified the assessment of the accrual for research and development costs related to clinical trial activities as a critical audit matter. The determination of the percentage of activities completed to date requires subjective estimates based on discussions with and reports provided by the contract research organizations, oversight of the activities, and the overall project budgets. Testing of the estimates required a higher degree of auditor judgment to evaluate and changes to the estimates could have a significant impact on the amount of accrued clinical trial expenses recorded by the Company.
The primary procedures we performed to address this critical audit matter included:
•Evaluating management’s process for estimating the accrual for clinical trial activities including estimating the activities completed to date and the associated cost incurred for the clinical trial activities.
•For certain contract research organizations, we tested the completeness and accuracy of the underlying billing information received from the contract research organizations used in determining the clinical trial accrual, including corroborating with invoices received and payments made by the Company to the contract research organizations.
•For certain clinical trial studies, we assessed the Company's estimates of the activities completed to date by (i) inspecting original contract terms and change orders including the expected timeline for the related study, (ii) obtaining third party reports detailing site visit information, and (iii) confirming clinical trial progress directly with the contract research organizations and comparing the reported amounts to the Company's estimates.
/s/ BDO USA, LLP
We have served as the Company’s auditor since 2013.
Boston, Massachusetts
March 17, 2022
107
ALDEYRA THERAPEUTICS, INC.
CONSOLIDATED BALANCE SHEETS
| | | | | | | | |
| | December 31, | | | December 31, | |
| | 2021 | | | 2020 | |
ASSETS | | | | | | |
Current assets: | | | | | | |
Cash and cash equivalents | | $ | 104,790,989 | | | $ | 52,858,311 | |
Cash equivalent - reverse repurchase agreements | | | 125,000,000 | | | | 25,000,000 | |
Marketable securities | | | 0 | | | | 0 | |
Prepaid expenses and other current assets | | | 2,961,781 | | | | 5,200,957 | |
Total current assets | | | 232,752,770 | | | | 83,059,268 | |
Fixed assets, net | | | 32,487 | | | | 59,925 | |
Right-of-use assets | | | 351,863 | | | | 233,310 | |
Total assets | | $ | 233,137,120 | | | $ | 83,352,503 | |
LIABILITIES AND STOCKHOLDERS' EQUITY | | | | | | |
Current liabilities: | | | | | | |
Accounts payable | | $ | 1,019,702 | | | $ | 381,638 | |
Accrued expenses | | | 10,523,353 | | | | 8,134,765 | |
Current portion of long-term debt | | | 0 | | | | 3,659,776 | |
Current portion of operating lease liabilities | | | 229,607 | | | | 233,310 | |
Total current liabilities | | | 11,772,662 | | | | 12,409,489 | |
Operating lease liabilities, long-term | | | 125,232 | | | | 0 | |
Long-term debt, net of current portion | | | 15,503,703 | | | | 11,434,456 | |
Total liabilities | | | 27,401,597 | | | | 23,843,945 | |
Commitments and contingencies (Note 13) | | | | | | |
Stockholders' equity: | | | | | | |
Preferred stock, $0.001 par value, 15,000,000 shares authorized, NaN
issued and outstanding | | | — | | | | — | |
Common stock, voting, $0.001 par value; 150,000,000 authorized and
58,081,215 and 38,667,491 shares issued and outstanding, respectively | | | 58,081 | | | | 38,667 | |
Additional paid-in capital | | | 500,369,444 | | | | 296,385,619 | |
Accumulated other comprehensive income | | | 0 | | | | 0 | |
Accumulated deficit | | | (294,692,002 | ) | | | (236,915,728 | ) |
Total stockholders’ equity | | | 205,735,523 | | | | 59,508,558 | |
Total liabilities and stockholders’ equity | | $ | 233,137,120 | | | $ | 83,352,503 | |
The accompanying notes are an integral part of these consolidated financial statements.
108
ALDEYRA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Operating expenses: | | | | | | |
Research and development | | $ | 44,936,532 | | | $ | 24,681,301 | |
Acquired in-process research and development | | | 0 | | | | 1,754,265 | |
General and administrative | | | 11,283,004 | | | | 9,985,454 | |
Loss from operations | | | (56,219,536 | ) | | | (36,421,020 | ) |
Other income (expense): | | | | | | |
Interest income | | | 185,363 | | | | 292,224 | |
Interest expense | | | (1,742,101 | ) | | | (1,904,198 | ) |
Total other income (expense), net | | | (1,556,738 | ) | | | (1,611,974 | ) |
Loss before income taxes | | | (57,776,274 | ) | | | (38,032,994 | ) |
Income tax benefit | | | 0 | | | | 479,265 | |
Net loss | | $ | (57,776,274 | ) | | $ | (37,553,729 | ) |
Net loss per share - basic and diluted | | $ | (1.07 | ) | | $ | (1.11 | ) |
Weighted average common shares outstanding - basic and diluted | | | 54,042,103 | | | | 33,965,955 | |
The accompanying notes are an integral part of these consolidated financial statements.
109
ALDEYRA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Net loss | | $ | (57,776,274 | ) | | $ | (37,553,729 | ) |
Other comprehensive loss: | | | | | | |
Unrealized loss on marketable securities | | | 0 | | | | (5,866 | ) |
Total other comprehensive loss | | $ | 0 | | | $ | (5,866 | ) |
Comprehensive loss | | $ | (57,776,274 | ) | | $ | (37,559,595 | ) |
The accompanying notes are an integral part of these consolidated financial statements.
110
ALDEYRA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
| | | | | | | | | | | | | | | | | | | | | | | | |
| | Stockholders' Equity | |
| | Common Voting Stock | | | | | | Accumulated | | | | | | | |
| | Shares | | | Amount | | | Additional
Paid-in Capital | | | Other
Comprehensive
Income/(Loss),
net of tax | | | Accumulated
Deficit | | | Total
Stockholders'
Equity | |
Balance, December 31, 2019 | | | 28,656,832 | | | $ | 28,657 | | | $ | 247,409,793 | | | $ | 5,866 | | | $ | (199,361,999 | ) | | $ | 48,082,317 | |
Stock-based compensation | | | — | | | | — | | | | 7,083,810 | | | | — | | | | — | | | | 7,083,810 | |
Release of restrictions on Helio founders’ shares | | | 273,616 | | | | 273 | | | | (273 | ) | | | — | | | | — | | | | — | |
Issuance of common stock, net of
issuance costs | | | 9,351,749 | | | | 9,352 | | | | 40,707,192 | | | | — | | | | — | | | | 40,716,544 | |
Issuance of common stock, exercise of stock options | | | 219,244 | | | | 219 | | | | 1,058,297 | | | | — | | | | — | | | | 1,058,516 | |
Issuance of common stock, employee
stock purchase plan | | | 30,254 | | | | 30 | | | | 126,936 | | | | — | | | | — | | | | 126,966 | |
Issuance of common stock, vested
restricted stock awards | | | 135,796 | | | | 136 | | | | (136 | ) | | | — | | | | — | | | | — | |
Other comprehensive loss | | | — | | | | — | | | | — | | | | (5,866 | ) | | | — | | | | (5,866 | ) |
Net loss | | | — | | | | — | | | | — | | | | — | | | | (37,553,729 | ) | | | (37,553,729 | ) |
Balance, December 31, 2020 | | | 38,667,491 | | | | 38,667 | | | | 296,385,619 | | | | — | | | | (236,915,728 | ) | | | 59,508,558 | |
Stock-based compensation | | | — | | | | — | | | | 7,006,857 | | | | — | | | | — | | | | 7,006,857 | |
Release of restrictions on Helio founders’ shares | | | 141,965 | | | | 142 | | | | (142 | ) | | | — | | | | — | | | | — | |
Issuance of common stock, net of
issuance costs | | | 18,091,947 | | | | 18,092 | | | | 189,793,519 | | | | — | | | | — | | | | 189,811,611 | |
Issuance of common stock, exercise of stock options | | | 634,214 | | | | 634 | | | | 4,622,617 | | | | — | | | | — | | | | 4,623,251 | |
Issuance of common stock, employee
stock purchase plan | | | 12,092 | | | | 12 | | | | 61,517 | | | | — | | | | — | | | | 61,529 | |
Issuance of common stock, vested
restricted stock awards | | | 286,944 | | | | 287 | | | | (287 | ) | | | — | | | | — | | | | — | |
Issuance of common stock in connection with Helio Vision, Inc. acquisition milestone | | | 246,562 | | | | 247 | | | | 2,499,744 | | | | — | | | | — | | | | 2,499,991 | |
Other comprehensive loss | | | — | | | | — | | | | — | | | | — | | | | — | | | | — | |
Net loss | | | — | | | | — | | | | — | | | | — | | | | (57,776,274 | ) | | | (57,776,274 | ) |
Balance, December 31, 2021 | | | 58,081,215 | | | $ | 58,081 | | | $ | 500,369,444 | | | $ | — | | | $ | (294,692,002 | ) | | $ | 205,735,523 | |
The accompanying notes are an integral part of these consolidated financial statements.
111
ALDEYRA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
CASH FLOWS FROM OPERATING ACTIVITIES: | | | | | | |
Net loss | | $ | (57,776,274 | ) | | $ | (37,553,729 | ) |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | |
Acquired in-process research and development | | | 0 | | | | 1,754,265 | |
Deferred taxes | | | 0 | | | | (479,265 | ) |
Stock-based compensation | | | 7,108,027 | | | | 7,083,810 | |
Non-cash interest expense | | | 409,471 | | | | 566,020 | |
Net amortization of premium on debt securities available for sale | | | 0 | | | | (91,231 | ) |
Depreciation and amortization | | | 264,180 | | | | 56,221 | |
Change in assets and liabilities: | | | | | | |
Prepaid expenses and other current assets | | | 2,239,176 | | | | (3,396,507 | ) |
Accounts payable | | | 638,064 | | | | (426,664 | ) |
Accrued expenses and other liabilities | | | 4,561,449 | | | | (5,006,375 | ) |
Net cash used in operating activities | | | (42,555,907 | ) | | | (37,493,455 | ) |
CASH FLOWS FROM INVESTING ACTIVITIES: | | | | | | |
Acquisitions of fixed assets | | | (7,806 | ) | | | — | |
Purchases of marketable securities | | | 0 | | | | (5,776,090 | ) |
Sales of marketable securities | | | 0 | | | | 34,800,000 | |
Net cash (used in) provided by investing activities | | | (7,806 | ) | | | 29,023,910 | |
CASH FLOWS FROM FINANCING ACTIVITIES: | | | | | | |
Proceeds from issuance of common stock, net of issuance costs | | | 189,811,611 | | | | 40,716,544 | |
Proceeds from exercise of stock options | | | 4,623,251 | | | | 1,058,516 | |
Proceeds from employee stock purchase plan | | | 61,529 | | | | 126,966 | |
Net cash provided by financing activities | | | 194,496,391 | | | | 41,902,026 | |
NET INCREASE IN CASH AND CASH EQUIVALENTS | | | 151,932,678 | | | | 33,432,481 | |
CASH AND CASH EQUIVALENTS, BEGINNING OF PERIOD | | | 77,858,311 | | | | 44,425,830 | |
CASH AND CASH EQUIVALENTS, END OF PERIOD | | $ | 229,790,989 | | | $ | 77,858,311 | |
SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION: | | | | | | |
Cash paid during the period for interest | | $ | 1,337,083 | | | $ | 1,387,750 | |
SUPPLEMENTAL INFORMATION AND DISCLOSURES OF NONCASH ACTIVITIES: | | | | | | |
Common stock issued in connection with Helio Vision, Inc. acquisition milestone | | $ | 2,499,991 | | | $ | — | |
The accompanying notes are an integral part of these consolidated financial statements.
112
ALDEYRA THERAPEUTICS, INC.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
1. NATURE OF BUSINESS
Aldeyra Therapeutics, Inc. (Aldeyra, Company, we, us and our) was incorporated in the state of Delaware on August 13, 2004 as Neuron Systems, Inc. On December 20, 2012, the Company changed its name to Aldexa Therapeutics, Inc. and, on March 17, 2014, the Company changed its name to Aldeyra Therapeutics, Inc. Aldeyra, together with its wholly-owned subsidiaries, is developing and commercializing next-generation medicines to improve the lives of patients with immune-mediated diseases.
The Company’s principal activities to date include raising capital and research and development activities.
2. BASIS OF PRESENTATION AND SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation – The accompanying consolidated financial statements were prepared in conformity with accounting principles generally accepted in the United States of America (US GAAP).
Risks and Uncertainties –The ongoing research and development activities will be subject to extensive regulation by numerous governmental authorities in the United States. Prior to marketing in the United States, any drug developed by the Company must undergo rigorous preclinical and clinical testing and an extensive regulatory approval process implemented by the United States Food and Drug Administration (FDA) under the Food, Drug and Cosmetic Act. The Company has limited experience in conducting and managing the preclinical and clinical testing necessary to obtain regulatory approval. There can be no assurance that the Company will not encounter problems in the clinical trials that will cause the Company or the FDA to delay or suspend clinical trials.
The Company’s success will depend in part on its ability to obtain patents and product license rights, maintain trade secrets, and operate without infringing on the property rights of others, both in the United States and other countries. There can be no assurance that patents issued to or licensed by the Company will not be challenged, invalidated, circumvented, or that the rights granted thereunder will provide proprietary protection or competitive advantages to the Company.
Based on its current operating plan, the Company believes that its cash and cash equivalents as of December 31, 2021, will be sufficient to fund the Company's currently projected operating expenses through the end of 2023, including potential new drug application (NDA) submissions; initial commercialization of reproxalap, if approved; and continued early and late-stage development of our product candidates in ocular and systemic immune-mediated diseases. As a result of the COVID-19 pandemic, clinical site availability, staffing, and patient recruitment have been negatively affected and the timelines to complete the Company’s clinical trials may be delayed. The Company’s assessment of its liquidity and capital resources includes an estimate of the financial impacts of these changes. The Company will need to secure additional funding in the future, from one or more equity or debt financings, collaborations, or other sources, in order to carry out all of the Company’s planned research and development activities and regulatory activities; commercialize product candidates; or conduct any substantial, additional development requirements requested by the Food and Drug Administration (FDA). Additional funding may not be available to the Company on acceptable terms, or at all. If the Company is unable to secure additional capital, it will be required to significantly decrease the amount of planned expenditures and may be required to cease operations.
Curtailment of operations would cause significant delays in the Company’s efforts to develop and introduce its products to market, which is critical to the realization of its business plan and the future operations of the Company.
Use of Estimates – The preparation of consolidated financial statements in conformity with US GAAP requires management to make estimates and assumptions, including fair value estimates for investments that affect the reported amounts of assets, liabilities, and the disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of expenses during the reporting period. The Company
113
evaluates its estimates and assumptions on an ongoing basis. The most significant estimates in the Company’s financial statements include, but are not limited to, estimates related to clinical trial accruals, estimates related to deferred and accrued research and development costs, and accounting for income taxes and the related valuation allowance. Although these estimates are based on the Company’s knowledge of current events and actions it may undertake in the future, actual results may materially differ from these estimates and assumptions.
Segment Information – Operating segments are defined as components of an enterprise about which separate discrete information is available for evaluation by the chief operating decision maker, or decision-making group, in deciding how to allocate resources and in assessing performance. The Company views its operations and manages its business in 1 segment, which is the identification and development of next-generation medicines to improve the lives of patients with immune-mediated diseases.
Cash and Cash Equivalents – The Company classifies all highly liquid investments with original maturities of three months or less as cash equivalents and all highly liquid investments with original maturities of greater than three months but less than 12 months as current marketable securities. The Company has a policy of making investments only with commercial institutions that have at least an investment grade credit rating. The Company invests its cash primarily in reverse repurchase agreements (RRAs), government securities and obligations, and money market funds.
RRAs are collateralized by deposits in the form of ‘Government Securities and Obligations’ for an amount not less than 102% of their value. The Company does not record an asset or liability related to the collateral as the Company is not permitted to sell or repledge the associated collateral. The Company has a policy that the collateral has at least an A (or equivalent) credit rating. The Company utilizes a third-party custodian to manage the exchange of funds as well as the requirement that collateral received is maintained at 102% of the value of the RRAs on a daily basis.
Marketable Securities – Marketable securities consist of government securities and obligations with original maturities of more than 90 days. Investments are classified as available-for-sale and are recorded on the balance sheet at fair value with unrealized gains or losses reported as a separate component of other comprehensive income/(loss). Management determines the appropriate classification of its investments at the time of purchase and re-evaluates such determination at each balance sheet date.
Fair Value of Financial Instruments – Financial instruments including cash equivalents and accounts payable are carried in the financial statements at amounts that approximate their fair value based on the short maturities of those instruments. Marketable securities are carried at fair value and are more fully described in Note 6. The carrying amount of the Company’s credit facility with Hercules Capital, Inc. approximates fair value since the effective interest rate approximates market rates currently available to the Company.
Concentration of Credit Risk – Financial instruments that potentially subject the Company to significant concentrations of credit risk principally consist of cash, cash equivalents and marketable securities, if any. The Company places its cash and cash equivalents and marketable securities with financial institutions which management believes have high credit ratings. As part of its cash and investment management processes, the Company performs periodic evaluations of the credit standing of the financial institutions with whom it maintains deposits.
Intellectual Property – The legal and professional costs incurred by the Company to acquire its patent rights are expensed as incurred and included in general and administrative expenses. At December 31, 2021 and 2020, the Company has determined that these expenses have not met the criteria to be capitalized since the future benefits to be derived from the patents is uncertain. Intellectual property related expenses for the years ended December 31, 2021 and 2020 were $1.3 million and $1.0 million, respectively.
Income Taxes – The Company follows the provisions of Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) 740, Income Taxes (ASC 740), in reporting deferred income taxes. ASC 740 requires a company to recognize deferred tax liabilities and assets for expected future income tax consequences of events that have been recognized in the Company’s financial statements. Under this method, deferred tax assets and liabilities are determined based on temporary differences between financial statement carrying amounts and the
114
tax basis of assets and liabilities using enacted tax rates in the years in which the temporary differences are expected to reverse. Valuation allowances are provided if based on the weight of available evidence, it is more likely than not that some or all the deferred tax assets will not be realized.
The Company accounts for uncertain tax positions pursuant to ASC 740 which prescribes a recognition threshold and measurement process for financial statement recognition of uncertain tax positions taken or expected to be taken in a tax return. If the tax position meets this threshold, the benefit to be recognized is measured as the tax benefit having the highest likelihood of being realized upon ultimate settlement with the taxing authority. The Company recognizes interest accrued related to unrecognized tax benefits and penalties in the provision for income taxes. Management is not aware of any uncertain tax positions.
Research and Development Costs – Research and development (R&D) costs are charged to expense as incurred and relate to salaries, employee benefits, stock-based compensation related to employees, consulting services, other operating costs and expenses associated with preclinical and clinical trial activities. Payments made by the Company in advance for research and development services not yet provided and/or for materials not yet received are recorded as prepaid expenses. Accrued liabilities are recorded related to those expenses for which vendors have not yet billed us with respect to services provided and/or materials that we have received.
Preclinical and clinical trial expenses relate to third-party services, subject-related fees at the sites where the Company’s clinical trials are being conducted, laboratory costs, analysis costs, toxicology studies and investigator fees. Costs associated with these expenses are generally payable on the passage of time or when certain milestones are achieved. Expense is recorded during the period incurred or in the period in which a milestone is achieved. In order to ensure that the Company has adequately provided for preclinical and clinical expenses during the proper period, the Company maintains an accrual to cover these expenses. These accruals are assessed on a quarterly basis and are based on such assumptions as expected total cost, the number of subjects and clinical trial sites and length of the study. Actual results may differ from these estimates and could have a material impact on the Company’s reported results. The Company’s historical accrual estimates have not been materially different from actual costs.
In-process research and development – Assets purchased in an asset acquisition transaction are expensed as in-process research and development (IPR&D) unless the assets acquired are deemed to have an alternative future use, provided that the acquired asset did not also include processes or activities that would constitute a “business” as defined under GAAP, the drug has not achieved regulatory approval for marketing and, absent obtaining such approval, has no established alternative future use. Acquired IPR&D payments are immediately expensed in the period in which they are incurred and include upfront payments, as well as transaction fees and subsequent pre-commercial milestone payments. Research and development costs incurred after the acquisition are expensed as incurred.
Stock-Based Compensation – Stock-based payments are accounted for in accordance with the provisions of ASC 718, Compensation – Stock Compensation. For options, the fair value of stock-based payments is estimated, on the date of grant, using the Black-Scholes option pricing model. For restricted stock, fair value is based on the fair value of the stock on the date of grant. The resulting fair value for restricted stock and options expected to vest is recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the applicable restricted stock or option. The Company records the effect of forfeitures and cancellations when they occur.
Comprehensive Loss – Comprehensive loss is defined as the change in equity during a period from transactions and other events and/or circumstances from non-owner sources. For December 31, 2021, comprehensive loss is equal to the Company’s net loss of $57.8 million. For December 31, 2020, comprehensive loss is equal to the Company's net loss of $37.6 million and a realized loss on marketable securities of $5.9 thousand.
Net Loss Per Share – The Company computes net loss per share in accordance with the two-class method. Under the two-class method, net loss is allocated between common stock and other participating securities based on their participation rights. The Company has determined that the nonvested shares issued to the Helio founders represents a participating security and as such the nonvested shares are excluded from basic earnings per share. Net losses are not allocated to the nonvested stockholders for computing net loss per share under the two-class method
115
because nonvested stockholders do not have contractual obligations to share in the losses of the Company. Basic earnings per share is calculated by dividing net loss allocable to common stockholders by the weighted average number of common stock outstanding.
Diluted net loss per share is computed using the more dilutive of (a) the two-class method, or (b) treasury stock method, as applicable, to the potentially dilutive instruments. The weighted-average number of common shares outstanding gives effect to all potentially dilutive common equivalent shares, including outstanding stock options and restricted stock, warrants, and nonvested shares.
Recent Accounting Pronouncements – In June 2016, the FASB issued (ASU) No. 2016-13, Financial Instruments—Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments (ASU 2016-13). ASU 2016-13 requires that credit losses be reported as an allowance using an expected losses model, representing the entity’s current estimate of credit losses expected to be incurred. The accounting guidance currently in effect is based on an incurred loss model. For available-for-sale debt securities with unrealized losses, this standard now requires allowances to be recorded instead of reducing the amortized cost of the investment. The amendments under ASU 2016-13 are effective for interim and annual fiscal periods beginning after December 15, 2022. The Company is continuing to evaluate the impact of ASU 2016-13 but does not expect the adoption of this ASU to have a material impact on its consolidated financial statements.
3. HELIO VISION ACQUISITION
On January 28, 2019 (Closing Date), the Company acquired Helio Vision, Inc. (Helio). As a result of the acquisition, the Company initially issued an aggregate of 1,160,444 shares of common stock to the former securityholders and an advisor of Helio. The founders of Helio were issued 568,627 shares and non-founders were issued 591,817 shares. The Helio founders’ shares are subject to vesting based on continued service to the Company over three years from the Closing Date of which 98% are vested as of December 31, 2021. The Company recognizes the expense associated with the founders’ restricted shares as compensation expense on a straight-line basis as the shares vest over the three-year period. For the year ended December 31, 2021 and 2020, the Company recorded $1.2 million and $1.3 million of research and development compensation expense, respectively, for the founders’ restricted shares.
In January 2021, pursuant to the terms of the acquisition agreement, the Company issued 246,562 shares of its common stock to the former securityholders of Helio (January Shares). In addition, the Company, subject to the conditions of the acquisition agreement, is contingently obligated to make additional payments to the former securityholders of Helio as follows:(a) $10.0 million of common stock following approval by the FDA of a new drug approval application for the prevention and/or treatment of proliferative vitreoretinopathy or a substantially similar label prior to the 10th anniversary of the Closing Date; and (b) $2.5 million of common stock following FDA approval of a new drug application for an indication (other than proliferative vitreoretinopathy) prior to the 12th anniversary of the Closing Date (the shares of common stock issuable pursuant to the preceding clauses (a) and (b) are referred to herein as the Milestone Shares), provided that in no event shall the Company be obligated to issue more than an aggregate of 5,248,885 shares of common stock. Additionally, in the event of certain change of control or divestitures by the Company, certain former convertible noteholders of Helio will be entitled to a tax gross-up payment in an amount not to exceed $1.0 million.
116
The Company determined that liability accounting is not required for the Milestone Shares under FASB ASC Topic 480, Distinguishing Liabilities from Equity (ASC 480). The Company also determined that the Milestone Shares meet the scope exception as a derivative under FASB ASC Topic 815, Derivatives and Hedging (ASC 815), from inception of the Milestone Shares through December 31, 2021. Accordingly, the Milestone Shares are evaluated under FASB ASC Topic 450, Contingencies (ASC 450) and the Company will record a liability related to the Milestone Shares if the milestones are achieved, and the obligation to make additional payment(s) becomes probable. At that time, the Company will record the cost of the Milestone Shares issued to the founders as compensation expense and to the Helio non-founders as IPR&D expense if there is no alternative future use. At December 31, 2020, the issuance of the January Shares was considered probable and $2.5 million was accrued as contingent consideration payable in stock and the Company recorded $1.8 million to IPR&D (Milestone IPR&D), which included a $0.5 million income tax benefit, and $1.2 million of compensation expense related to these January Shares, which amounted to 246,562 shares and were issued during the quarter ended March 31, 2021. No other milestones related to the remaining Milestone Shares are probable of being achieved as of December 31, 2021.
4. NET LOSS PER SHARE
For the years ended December 31, 2021 and 2020, diluted weighted-average common shares outstanding is equal to basic weighted-average common shares due to the Company’s net loss position.
The following potentially dilutive securities outstanding have been excluded from the computation of diluted weighted-average shares outstanding, because such securities had an antidilutive impact:
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Options to purchase common stock | | | 4,622,870 | | | | 4,608,311 | |
Nonvested restricted stock units | | | 501,255 | | | | 927,189 | |
Nonvested founder shares (1) | | | 10,891 | | | | 152,856 | |
Total of common stock equivalents | | | 5,135,016 | | | | 5,688,356 | |
(1) Represents 10,891 and 152,856 shares of common stock that are issued and outstanding but were subject to vesting based on service requirements at December 31, 2021 and 2020, respectively, and are not included in stockholders’ equity.
5. CASH, CASH EQUIVALENTS AND MARKETABLE SECURITIES
At December 31, 2021, cash and cash equivalents were comprised of:
| | | | | | | | | | | | | | | | | | | | |
| | Carrying
Amount | | | Unrecognized
Gain | | | Unrecognized
Loss | | | Estimated Fair
Value | | | Cash and Cash
Equivalents | |
Cash | | $ | 100,364,523 | | | $ | — | | | $ | — | | | $ | 100,364,523 | | | $ | 100,364,523 | |
Money market funds | | | 4,426,466 | | | | — | | | | — | | | | 4,426,466 | | | | 4,426,466 | |
Reverse repurchase agreements | | | 125,000,000 | | | | — | | | | — | | | | 125,000,000 | | | | 125,000,000 | |
Total cash and cash equivalents | | $ | 229,790,989 | | | $ | — | | | $ | — | | | $ | 229,790,989 | | | $ | 229,790,989 | |
At December 31, 2020, cash and cash equivalents were comprised of:
| | | | | | | | | | | | | | | | | | | | |
| | Carrying
Amount | | | Unrecognized
Gain | | | Unrecognized
Loss | | | Estimated Fair
Value | | | Cash and Cash
Equivalents | |
Cash | | $ | 23,494,920 | | | $ | — | | | $ | — | | | $ | 23,494,920 | | | $ | 23,494,920 | |
Money market funds | | | 29,363,391 | | | | — | | | | — | | | | 29,363,391 | | | | 29,363,391 | |
Reverse repurchase agreements | | | 25,000,000 | | | | — | | | | — | | | | 25,000,000 | | | | 25,000,000 | |
Total cash and cash equivalents | | $ | 77,858,311 | | | $ | — | | | $ | — | | | $ | 77,858,311 | | | $ | 77,858,311 | |
There were 0 marketable securities held at December 31, 2021 or December 31, 2020.
117
6. FAIR VALUE MEASUREMENTS
Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value are performed in a manner to maximize the use of observable inputs and minimize the use of unobservable inputs. ASC 820, Fair Value Measurements, establishes a fair value hierarchy based on three levels of inputs, of which the first two are considered observable and the last unobservable, that may be used to measure fair value, which are the following:
Level 1 – Quoted prices in active markets that are accessible at the market date for identical unrestricted assets or liabilities.
Level 2 – Inputs other than Level 1 that are observable, either directly or indirectly, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs for which all significant inputs are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3 – Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
The following table presents information about the Company’s assets measured at fair value at December 31, 2021 and December 31, 2020:
| | | | | | | | | | | | | | | | |
| | December 31, 2021 | |
| | Level 1 | | | Level 2 | | | Level 3 | | | Total | |
Assets: | | | | | | | | | | | | |
Money market funds (a) | | $ | 4,426,466 | | | $ | 0 | | | $ | 0 | | | $ | 4,426,466 | |
Reverse repurchase agreements (b) | | | 0 | | | | 125,000,000 | | | | 0 | | | | 125,000,000 | |
Total assets at fair value | | $ | 4,426,466 | | | $ | 125,000,000 | | | $ | 0 | | | $ | 129,426,466 | |
| | | | | | | | | | | | | | | | |
| | December 31, 2020 | |
| | Level 1 | | | Level 2 | | | Level 3 | | | Total | |
Assets: | | | | | | | | | | | | |
Money market funds (a) | | $ | 29,363,391 | | | $ | 0 | | | $ | 0 | | | $ | 29,363,391 | |
Reverse repurchase agreements (b) | | | 0 | | | | 25,000,000 | | | | 0 | | | | 25,000,000 | |
Total assets at fair value | | $ | 29,363,391 | | | $ | 25,000,000 | | | $ | 0 | | | $ | 54,363,391 | |
(a)Money market funds included in cash and cash equivalents in the consolidated balance sheets, are valued at quoted market prices in active markets.
(b)Reverse repurchase agreements and U.S. government agency securities, if any, are recorded at fair market values, which are determined based on the most recent observable inputs for similar instruments in active markets or quoted prices for identical or similar instruments in markets that are not active or are directly or indirectly observable.
7. PREPAID EXPENSES AND OTHER CURRENT ASSETS
Prepaid expenses and other current assets at December 31, 2021 and 2020 were:
| | | | | | | | |
| | December 31, | | | December 31, | |
| | 2021 | | | 2020 | |
Deferred research and development expenses | | $ | 2,404,145 | | | $ | 4,793,794 | |
Miscellaneous prepaid expenses and other current assets | | | 557,636 | | | | 407,163 | |
Total prepaid expenses and other current assets | | $ | 2,961,781 | | | $ | 5,200,957 | |
118
8. ACCRUED EXPENSES
Accrued expenses at December 31, 2021 and 2020 were:
| | | | | | | | |
| | December 31, | | | December 31, | |
| | 2021 | | | 2020 | |
Accrued compensation | | $ | 1,512,885 | | | $ | 1,323,138 | |
Contingent consideration payable in stock | | | 0 | | | | 2,500,000 | |
Accrued research and development expenses | | | 8,415,560 | | | | 3,944,094 | |
Accrued general and administrative expenses | | | 594,908 | | | | 367,533 | |
Total accrued expenses | | $ | 10,523,353 | | | $ | 8,134,765 | |
9. CREDIT FACILITY
The Company’s long-term debt obligation consists of amounts the Company is obligated to repay under its credit facility with Hercules Capital, Inc. (Hercules). In March 2019, the Company entered into a Loan and Security Agreement with Hercules and several banks and other financial institutions or entities, from time-to-time parties thereto (collectively, referred to as Lender), providing for a term loan of up to $60.0 million, subject to the satisfaction of certain conditions contained therein, that is secured by a lien covering all of the Company’s assets, other than the Company’s intellectual property (Loan and Security Agreement or the Hercules Credit Facility). The Loan and Security Agreement provided for (i) an initial term loan advance of up to $5.0 million at the Company’s option, which expired unutilized on April 15, 2019; (ii) three additional term loan advances of up to $15.0 million each, at the Company’s option, available to the Company upon the occurrence of certain funding conditions prior to September 30, 2019 (2019 Tranche), March 31, 2020 (2020 Tranche), and March 31, 2021 (2021 Tranche); and (iii) a final additional term loan advance (Fourth Loan Tranche) of up to $10.0 million prior to December 31, 2021, at the Company’s option, subject to approval by the Lender’s investment committee. The 2019 Tranche was drawn down in full by the Company in September 2019 and the 2020 Tranche and 2021 Tranche expired unutilized prior to the Company satisfying the funding conditions for such tranche. On April 20, 2021, the Company entered into the First Amendment (First Amendment) to the Loan and Security Agreement with Hercules. The First Amendment, among other things, (i) increased the Fourth Loan Tranche from $10.0 million to $20.0 million and extended the deadline for drawing down the Fourth Loan Tranche to July 1, 2022; (ii) lowered the variable per annum rate of interest on borrowings under the Loan and Security Agreement from the greater of (a) 9.10% and (b) the prime rate (as reported in the Wall Street Journal or any successor publication thereto) plus 3.10% to the greater of (x) the Prime Rate (as defined therein) plus 3.10% or (y) 8.60%; (iii) extended the expiration of the period in which interest-only payments on borrowings under the Loan and Security Agreement are required from May 1, 2021 to July 1, 2022; and (iv) following the satisfaction of certain conditions in April 2021, further extended the expiration of the interest-only period and the deadline for drawing down the Fourth Loan Tranche to May 1, 2023. Repayment of the aggregate outstanding principal balance of the term loan, in monthly installments, commences upon expiration of the interest-only period and continues through October 1, 2023 (Maturity Date). The First Amendment was determined to be a modification in accordance with FASB ASC Topic 470, Debt and did not result in extinguishment.
Associated with this debt facility, the Company incurred a commitment charge of $25,000, transaction costs of $273,186, a fee of $375,000 upon closing, and is required to pay a fee (End of Term Charge) of 6.95% multiplied by the aggregate amount of advances under the Loan and Security Agreement at maturity. The fees, transaction costs, and the End of Term Charge are amortized to interest expense from 2019 through the Maturity Date using the effective interest method. The effective interest rate was 10.8% at December 31, 2021. At the Company’s option, the Company may elect to prepay all, but not less than all, of the outstanding term loan by paying the entire principal balance and all accrued and unpaid interest thereon plus all fees and other amounts due under the Loan and Security Agreement, including a prepayment charge equal to 1.5% of the principal amount being prepaid.
Following the effective time of the First Amendment and as of December 31, 2021, an aggregate of $35 million, subject to the terms and conditions of the Loan and Security Agreement, may be made available to the Company for borrowing, $15 million of which was funded prior to the date of the First Amendment.
119
Long-term debt consisted of the following:
| | | | | | | | |
| | December 31, | | | December 31, | |
| | 2021 | | | 2020 | |
Term loan payable | | $ | 15,000,000 | | | $ | 15,000,000 | |
End of term charge | | | 703,269 | | | | 445,464 | |
Unamortized debt issuance costs | | | (199,566 | ) | | | (351,232 | ) |
Less: current portion | | | 0 | | | | (3,659,776 | ) |
Total long-term debt | | $ | 15,503,703 | | | $ | 11,434,456 | |
Future principal payments, including the End of Term Charge, are as follows for the years ending December 31:
| | | | |
| | | |
2022 | | $ | 0 | |
2023 | | | 16,042,500 | |
Total | | $ | 16,042,500 | |
| | | |
The Loan and Security Agreement also contains certain events of default, representations, warranties and non-financial covenants of the Company. As of December 31, 2021, the Company was in material compliance with all covenants of the Hercules Credit Facility. In addition, subject to the terms of the Loan and Security Agreement, the Company granted the Lender the right to purchase up to an aggregate of $2.0 million of the Company’s equity securities, or instruments exercisable for or convertible into equity securities, sold to investors in financings upon the same terms and conditions afforded to such other investors.
10. STOCKHOLDERS’ EQUITY
Common Stock
Each share of common stock is entitled to one vote. The holders of common stock are also entitled to receive dividends whenever funds are legally available and when declared by the board of directors, subject to the prior rights of holders of all classes of stock outstanding. As of December 31, 2021, a total of 4,622,870, 3,044,303, and 1,233,557, shares of common stock were reserved for issuance upon (i) the exercise of outstanding stock options, (ii) the issuance of stock awards under the Company’s Amended 2013 Plan, and (iii) the issuance of shares under the 2016 ESPP, respectively.
2018 Jefferies Sales Agreement
In December 2018, the Company entered into an Open Market Sales Agreement SM (2018 Jefferies Sales Agreement) with Jefferies LLC (Jefferies), as sales agent, pursuant to which the Company could offer and sell, from time to time through Jefferies, shares of common stock providing for aggregate sales proceeds of up to $50.0 million. During the year ended December 31, 2020, the Company sold, at a volume-weighted average price of $4.62, an aggregate of 9,351,749 shares of common stock and received $40.7 million after deducting commissions related to the 2018 Jefferies Sales Agreement and other offering costs. As of December 31, 2020, the Company had sold the maximum allowable amount and no further sales may be made under the 2018 Jefferies Sales Agreement.
2021 Jefferies Sales Agreement
In March 2021, the Company entered into an Open Market Sales Agreement SM (2021 Jefferies Sales Agreement) with Jefferies, as sales agent, pursuant to which the Company may offer and sell, from time to time through Jefferies, shares of common stock providing for aggregate sales proceeds of up to $100.0 million. The Company has no obligation to sell any shares under the 2021 Jefferies Sales Agreement, and could at any time
120
suspend solicitations and offers under the 2021 Jefferies Sales Agreement. No sales had been made pursuant to the 2021 Jefferies Sales Agreement as of December 31, 2021.
Underwritten Public Offerings
In January 2021, the Company sold 7.9 million shares of its common stock in an underwritten public offering at $9.50 per share, for an aggregate gross cash purchase price of $74.7 million or proceeds of $70.0 million after underwriters’ discount, commissions, and other offering expenses.
In May and June 2021, the Company sold an aggregate of 10.2 million shares of its common stock at a public offering price of $12.50 per share, in an underwritten public offering, for an aggregate gross cash purchase price of $127.8 million or proceeds of $119.8 million after underwriters’ discount, commissions, and other offering expenses, which included the partial exercise of the underwriters' option to purchase additional shares.
11. INCOME TAXES
NaN current or deferred tax provision expenses for federal and state income taxes have been recorded as the Company has incurred losses since inception for tax purposes. Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes.
In assessing the realizability of net deferred taxes in accordance with ASC 740 the Company considers whether it is more likely than not that some portion or all the deferred tax assets will not be realized. Based on the weight of available evidence, primarily the incurrence of net losses since inception, anticipated net losses in the near future, reversals of existing temporary differences and expiration of various federal and state attributes, the Company does not consider it more likely than not that some or all of the net deferred taxes will be realized. Accordingly, a 100% valuation allowance has been applied against net deferred taxes. Effective January 1, 2021, the Company adopted ASU No. 2019-12, Income Taxes (Topic 740), which did not have a material impact to the Company’s financial statements.
As of December 31, 2021, the Company had federal and state income tax net operating loss (NOL) carryforwards of approximately $226.0 million and $219.0 million, respectively. Federal NOL carryforwards generated through December 31, 2017 and state NOL carryforwards will expire at various dates through 2041. The federal NOL carryforwards generated during the year ended December 31, 2018 and thereafter will carryforward indefinitely. As of December 31, 2021, the Company had federal and state research and development tax credit carryforwards of approximately $6.9 million and $1.6 million, respectively, which will expire at various dates through 2041. Additionally, as of December 31, 2021, the Company had a federal orphan drug tax credit carryforward of approximately $1.0 million that expires in 2041.
The Coronavirus Aid, Relief and Economic Security (CARES) Act was enacted in the United States on March 27, 2020. CARES includes several income tax provisions such as NOL carryback and carryforward benefits and other tax deduction benefits. As noted previously, the Company’s U.S. deferred tax asset has a full valuation allowance, accordingly these NOL and other benefit provisions have no impact on the Company’s financial statements for the period ended December 31, 2021.
121
Significant components of the Company’s deferred tax assets and liabilities at December 31, 2021 and 2020 are as follows:
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Deferred Tax Assets | | | | | | |
Federal & state NOL carryforward | | $ | 61,308,787 | | | $ | 48,273,248 | |
Federal & state R&D credit
carryforward | | | 9,200,758 | | | | 6,375,229 | |
Intangibles – net | | | 78,378 | | | | 78,378 | |
Accounts payable and accrued
expenses | | | 2,301,117 | | | | 192,200 | |
Stock options | | | 3,906,501 | | | | 3,762,247 | |
Other items | | | 127,791 | | | | 94,020 | |
Gross deferred tax assets | | | 76,923,332 | | | | 58,775,322 | |
Valuation allowance | | | (76,827,203 | ) | | | (58,711,582 | ) |
Deferred tax assets, net | | $ | 96,129 | | | $ | 63,740 | |
Deferred Tax Liabilities | | | | | | |
Right of use asset | | | (96,129 | ) | | | (63,740 | ) |
TOTAL | | $ | 0 | | | $ | 0 | |
The change in valuation allowance of $18.1 million from December 31, 2020 to December 31, 2021 was primarily the result of an increase in net operating losses and tax credits.
The components of the income tax benefit for the years ended December 31, 2021 and 2020, are as follows:
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Deferred Taxes | | | | | | |
Federal | | $ | 0 | | | $ | (338,924 | ) |
State | | | — | | | | (140,341 | ) |
Total income tax benefit | | $ | 0 | | | $ | (479,265 | ) |
122
Future changes in federal and state tax laws pertaining to net operating loss carryforwards may also cause limitations or restrictions from us claiming such net operating losses. If the net operating loss carryforwards become unavailable to us or are fully utilized, our future taxable income will not be shielded from federal and state income taxation absent certain U.S. federal and state tax credits, and the funds otherwise available for general corporate purposes would be reduced.
Under Section 382 of the Internal Revenue Code of 1986, as amended, a corporation that undergoes an “ownership change” is subject to limitations on its ability to utilize its pre-change NOLs and certain other tax assets to offset future taxable income. In general, an ownership change occurs if the aggregate stock ownership of certain stockholders increases by more than 50 percentage points over such stockholders’ lowest percentage ownership during the testing period (generally three years). Transactions involving the Company’s common stock within the testing period, even those outside the Company’s control such as purchases or sales by investors, could result in an ownership change. A limitation on the Company’s ability to utilize some or all its NOLs or credits could have a material adverse effect on the Company’s results of operations and cash flows. Company believes, prior to December 31, 2020 that the three ownership changes occurred since inception, and that a fourth ownership change was triggered during the year ended December 31, 2021. Management believes that its aggregate Section 382 limitation (including the additional limitation for recognized "built-in gains") is sufficient so that no current impairment of its pre-ownership change tax attributes is required. Any future ownership changes, including those resulting from any recent or future financing activities, may cause our existing tax attributes to have additional limitations.
As of December 31, 2021, the Company is subject to tax in the U.S. (Federal and Massachusetts). The Company is open to examination for the tax years ended December 31, 2021, 2020, 2019 and 2018. In addition, any loss years remain open to the extent that losses are available for carryover to future years.
A reconciliation of the federal statutory tax rate of 21% to the Company’s effective income tax rates are as follows:
| | | | | | | | | | |
| | Years ended December 31, |
| | 2021 | | 2020 |
Statutory tax rate | | | 21.00 | | % | | | 21.00 | | % |
State taxes, net of federal benefits | | | 6.65 | | % | | | 5.06 | | % |
Federal research and development credits | | | 4.29 | | % | | | 3.28 | | % |
Change in valuation allowance | | | (31.35 | ) | % | | | (22.76 | ) | % |
Stock-based compensation | | | (0.73 | ) | % | | | (5.85 | ) | % |
Other | | | 0.14 | | % | | | 0.55 | | % |
Effective tax rate | | | (0.00 | ) | % | | | 1.28 | | % |
123
12. STOCK INCENTIVE PLAN
The Company approved the 2010 Employee, Director and Consultant Equity Incentive Plan (2010 Plan) in September 2010 to replace the 2004 Employee, Director and Consultant Equity Incentive Plan. The 2010 Plan provided for the granting of stock options and restricted stock awards. The 2010 Plan terminated upon the Company’s initial public offering in May 2014. However, grants made under the 2010 Plan are still governed by that plan. As of December 31, 2021, options to purchase 398,547 shares of common stock at a weighted average exercise price of $1.52 per share remained outstanding under the 2010 Plan.
The Company approved the 2013 Equity Incentive Plan in October 2013. The 2013 Equity Incentive Plan became effective immediately on adoption. The 2013 Equity Incentive Plan was amended in June 2016 and June 2018, (Amended 2013 Plan). The Amended 2013 Plan provides for the granting of stock options, restricted stock, stock appreciation rights, stock units, and performance cash awards to certain employees, members of the board of directors and consultants of the Company. On January 1 of each year the aggregate number of common shares that may be issued under the Amended 2013 Plan shall automatically increase by a number of shares equal to the lower of (a) 6% of the total number of shares of common stock outstanding on the last calendar day of the prior fiscal year, or (b) a number of shares of common stock determined by the Company’s board of directors. As of December 31, 2021, options to purchase 4,224,323 shares of common stock at a weighted average exercise price of $7.23 per share and 501,255 shares of common stock underlying restricted stock units remained outstanding under the Amended 2013 Plan. As of December 31, 2021, there were 3,044,303 shares of common stock available for grant under the Amended 2013 Plan. As of January 1, 2022, the number of shares of common stock that may be issued under the Amended 2013 Plan was automatically increased by 3,485,526 shares, increasing the number of shares of common stock available for issuance under the Amended 2013 Plan to 6,529,829.
In 2019 and 2020, the Company granted performance cash awards. The performance cash awards vest in four annual installments from the date of grant and entitle the employees to receive a cash payment, on the earlier of (i) four years from the date of grant or (ii) a change of control, equal in value to the amount by which the then value of the Company’s common stock exceeds the base value. As of December 31, 2021, $0.4 million was accrued as compensation expense for vested performance cash awards.
The Company recognizes stock-based compensation expense over the requisite service period. The Company’s share-based awards are accounted for as equity instruments. The amounts included in the consolidated statements of operations relating to stock-based compensation associated with the two equity incentive plans, performance cash awards, and Helio founders’ shares are as follows:
| | | | | | | | |
| | Years ended December 31, | |
| | 2021 | | | 2020 | |
Research and development expenses | | $ | 3,562,181 | | | $ | 3,798,141 | |
General and administrative expenses | | | 3,545,846 | | | | 3,285,669 | |
Total stock-based compensation expense | | $ | 7,108,027 | | | $ | 7,083,810 | |
Stock Options
Terms of stock option agreements, including vesting requirements, are determined by the board of directors or its compensation committee, subject to the provisions of the respective plan from which they were granted. Options granted by the Company typically vest over a four-year period. The options are subject to acceleration of vesting in the event of certain change of control transactions. The options may be granted for a term of up to ten years from the date of grant. The exercise price for options granted under the Amended 2013 Plan must be at a price no less than 100% of the fair market value of a common share on the date of grant.
124
The following table summarizes option activity under the incentive plans for the year ended December 31, 2021:
| | | | | | | | | | | | | | | | |
| | Number of
Shares | | | Weighted
Average
Exercise
Price | | | Weighted
Average
Contractual
Term | | | Aggregate
Intrinsic
Value(a) | |
Outstanding at December 31, 2020 | | | 4,608,311 | | | $ | 5.73 | | | | | | $ | 7,492,474 | |
Granted | | | 965,169 | | | | 11.40 | | | | | | | 0 | |
Cancelled/Forfeited | | | (316,396 | ) | | | 5.23 | | | | | | | 2,217,529 | |
Exercised | | | (634,214 | ) | | | 7.29 | | | | | | | 2,450,104 | |
Outstanding at December 31, 2021 | | | 4,622,870 | | | $ | 6.73 | | | | 6.71 | | | $ | 1,268,089 | |
Exercisable at December 31, 2021 | | | 3,134,196 | | | $ | 5.90 | | | | 5.79 | | | $ | 1,150,723 | |
(a)The aggregate intrinsic value in this table was calculated on the positive difference, if any, between the closing price per share of the Company’s common stock on December 31, 2021 of $4.00 and the per share exercise price of the underlying options.
The Company records stock-based compensation related to stock options granted at fair value. During the years ended December 31, 2021 and 2020, the Company used the Black-Scholes option-pricing model to estimate the fair value of stock option grants and to determine the related compensation expense. The assumptions used in calculating the fair value of stock-based payment awards represent management’s best estimates. The weighted-average fair value of options granted was $7.95 and $2.57 for the years ended December 31, 2021 and 2020, respectively. The assumptions used in determining fair value of the employee stock options for the years ended December 31, 2021 and 2020, are as follows:
| | | | | | | | |
| | December 31,
2021 | | | December 31,
2020 | |
Expected dividend yield | | | 0 | % | | | 0 | % |
Anticipated volatility | | 81.73% - 83.50% | | | 75.83% - 82.83% | |
Stock price | | $8.33 - $12.97 | | | $2.98 - $6.77 | |
Exercise price | | $8.33 - $12.97 | | | $2.98 - $6.77 | |
Expected life (years) | | 5.50 - 6.08 | | | 5.50 - 6.25 | |
Risk free interest rate | | 0.88% - 1.67% | | | 0.39% - 1.71% | |
The dividend yield of 0 is based on the fact that the Company has never paid cash dividends and have 0 present intention to pay cash dividends. Expected volatility is estimated using the historical volatility of the Company. The Company has estimated the expected life of its employee stock options using the “simplified” method, whereby, the expected life equals the average of the vesting term and the original contractual term of the option for service-based awards since the Company doesn’t have sufficient historical or implied data of its own. The risk-free interest rates for periods within the expected life of the option are based on the yields of zero-coupon United States Treasury securities.
At December 31, 2021, there is approximately $7.5 million of unrecognized compensation cost relating to stock options outstanding, which the Company expects to recognize over a weighted average period of 2.50 years. Total unrecognized compensation cost will be adjusted for future forfeitures, if necessary.
Restricted Stock Units
Terms of restricted stock unit (RSUs) agreements, including vesting requirements, are determined by the board of directors or its compensation committee, subject to the provisions of the Amended 2013 Plan. RSUs granted by the Company typically vest over a four year period. In the event that the employees’ employment with the Company terminates any unvested shares are forfeited and revert to the Company. RSUs are not included in
125
issued and outstanding common stock until the shares are vested and released. The table below summarizes activity relating to RSUs for the year ended December 31, 2021 :
| | | | |
| | Number
of Shares | |
Outstanding at December 31, 2020 | | | 927,189 | |
Granted | | | 46,619 | |
Cancelled/Forfeited | | | (185,610 | ) |
Vested/released | | | (286,943 | ) |
Outstanding at December 31, 2021 | | | 501,255 | |
The weighted-average fair value of RSUs granted was $11.71 and $3.78 per share for the years ended December 31, 2021 and 2020, respectively. As of December 31, 2021, the outstanding RSUs had unamortized stock-based compensation of $1.7 million with a weighted-average remaining recognition period of 1.91 years and an aggregate intrinsic value of $3.0 million.
Employee Stock Purchase Plan
In March 2016, the Company’s board of directors approved the 2016 Employee Stock Purchase Plan (2016 ESPP), which became effective in June 2016 following the approval of the Company’s stockholders. The 2016 ESPP initially authorized the issuance of up to a total of 414,639 shares of the Company’s common stock to participating employees. The number of shares reserved for issuance under the 2016 ESPP automatically increases on the first business day of each fiscal year, commencing in 2017, by a number equal to the lower of (i) 1% of the shares of common stock outstanding on the last business day of the prior fiscal year; or (ii) the number of shares determined by the Company’s Board of Directors. Unless otherwise determined by the administrator of the 2016 ESPP, two offering periods of six months’ duration will begin each year on January 1 and July 1. Participating employees purchase stock under the 2016 ESPP at a price equal to the lower of 85% of the closing price on the applicable offering commencement date or 85% of the closing price on the applicable offering termination date. The fair value of the purchase rights granted under this plan was estimated on the date of grant using the Black-Scholes option-pricing model using assumptions as shown below:
| | | | | | | | |
| | December 31,
2021 | | | December 31,
2020 | |
Expected dividend yield | | | 0 | % | | | 0 | % |
Anticipated volatility | | 81.76% - 82.87% | | | 80.66% - 82.61% | |
Stock price | | $6.53 - $11.31 | | | $4.17 - $5.96 | |
Exercise price | | $6.53 - $11.31 | | | $4.17 - $5.96 | |
Expected life (years) | | | 0.50 | | | | 0.50 | |
Risk free interest rate | | 0.05% - 0.09% | | | 0.17% - 1.57% | |
At December 31, 2021, the Company has 1,233,557 shares available for issuance under the 2016 ESPP. A summary of the weighted-average grant-date fair value, shares issued and total stock-based compensation expense recognized related to the 2016 ESPP for the years ended December 31, 2021 and 2020 are as follows:
| | | | | | | | |
| | December 31,
2021 | | | December 31,
2020 | |
Weighted-average grant-date fair value
per share | | $ | 2.98 | | | $ | 2.13 | |
Total shares issued | | | 12,092 | | | | 30,254 | |
Total stock-based compensation expense | | $ | 45,190 | | | $ | 34,224 | |
126
13. COMMITMENTS AND CONTINGENCIES
Guarantees and Indemnifications
As permitted under Delaware law, the Company indemnifies its officers and directors for certain events or occurrences while the officer or director is, or was, serving at the Company’s request in such capacity. The term of the indemnification is for the officer’s or director’s lifetime. Through December 31, 2021, the Company had not experienced any losses related to these indemnification obligations and 0 material claims were outstanding. The Company does not expect significant claims related to these indemnification obligations, and consequently, concluded that the fair value of these obligations is negligible, and 0 related reserves were established.
In-License Agreements
Madrigal Agreement
The Company is developing ADX-1612 pursuant to a License Agreement with Madrigal Pharmaceuticals, Inc. (Madrigal), entered into on December 26, 2016 (Madrigal Agreement). Pursuant to the Madrigal Agreement, the Company obtained an exclusive, worldwide license from Madrigal under certain patents and patent applications, and other licenses to intellectual property, to develop and commercialize Hsp90 inhibitors, including ADX-1612, (collectively, Madrigal Agreement Products). The Company has agreed to use its commercially reasonable efforts to develop Madrigal Agreement Products.
In consideration for the rights licensed under the Madrigal Agreement, the Company paid Madrigal an upfront license fee of $250,000 and are obligated to make future regulatory and development and sales-dependent milestone payments to Madrigal of less than $340 million in the aggregate (over 80% of such amount being tied to the Company’s achievement of increasingly greater annual worldwide net sales milestones), as well as royalty payments to Madrigal at a rate which, as a percentage of net sales, is in the high single digits for products containing ADX-1612 and mid-single digits for any other Hsp90 inhibitor product. The Company is also obligated under the Madrigal Agreement to pay Madrigal a percentage of certain sublicense revenue that the Company receives in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from the mid-twenties to low-single digits based on the development stage of the product at the time of the sublicense.
The Madrigal Agreement will remain in effect until all payment obligations under the Madrigal Agreement expire. The Company may terminate the Madrigal Agreement in its entirety or on a Madrigal Agreement Product-by Madrigal Agreement Product basis with timely notice to Madrigal. Either party may terminate the Madrigal Agreement for uncured material breach by the other party or upon certain insolvency or bankruptcy proceedings involving the other party, both with timely notice to the other party. In addition, Madrigal has the right to terminate the Madrigal Agreement if the Company, its affiliates, or sublicensees interfere with, challenge the validity or enforceability of, oppose the extension of, or grant of a supplementary protection certificate with respect to any of the Company’s licensed patents under the Madrigal Agreement. In the event of an early termination of the Madrigal Agreement, all rights licensed and developed by the Company under the Madrigal Agreement may revert back to Madrigal. Each party has agreed to indemnify the other party for certain third party claims arising under the Madrigal Agreement.
MEEI Agreement
The Company is developing ADX-2191 pursuant to an Exclusive License Agreement with Massachusetts Eye and Ear Infirmary (MEEI) originally entered into in July 2016 between MEEI and Helio Vision, Inc., as amended, (MEEI Agreement). The Company assumed the MEEI Agreement in connection with its 2019 acquisition of Helio Vision.
Pursuant and subject to the MEEI Agreement, the Company obtained an exclusive, worldwide license from MEEI to develop and commercialize ADX-2191 under certain patents and patent applications, and other licenses to intellectual property (MEEI Patent Rights). The Company has agreed to use commercially reasonable efforts, to develop ADX-2191 and to meet certain specified effort and achievement benchmarks by certain dates.
127
In consideration for the rights licensed under the MEEI Agreement, Helio Vision issued MEEI a number of shares of its preferred stock and Helio Vision agreed to pay non-creditable non-refundable license maintenance fees to MEEI of $15,000 on each of the second and third anniversary of the MEEI Agreement, $25,000 on each of the fourth and fifth anniversary of the MEEI Agreement and $35,000 on the sixth and each subsequent anniversary of the MEEI Agreement during the term of such agreement. In addition, Helio Vision was obligated to make future sales-dependent milestone payments to MEEI of up to the low seven figures in the aggregate, as well as royalty payments to MEEI at a rate which, as a percentage of net sales, is in the low single digits for products that incorporate or use the MEEI Patent Rights in the United States and as a percentage in the low single digits for products that incorporate or use the MEEI Patent Rights outside the United States. The Company is also obligated under the MEEI Agreement to pay MEEI a percentage of certain sublicense revenue that it receives in connection with entering into any sublicensing arrangements with any third parties, at a percentage rate which tiers downward from low-double digits to mid-single digits based on the date of the sublicense. Following the Company’s acquisition of Helio Vision, the Company became obligated to make any future payments owed under the MEEI Agreement. There is 0 additional equity consideration issuable under the MEEI Agreement.
The MEEI Agreement will remain in effect until the expiration date of the last to expire patent licensed under the MEEI Agreement. The Company may terminate the MEEI Agreement with timely written notice to MEEI. MEEI has the right to terminate the MEEI Agreement if it, subject to certain specified cure periods, ceases all business operations with respect to licensed products, fails to pay amounts due under the MEEI Agreement, fail to comply with certain due diligence obligations, defaults in our obligation to maintain insurance, one of our officers is convicted of a felony relating to the manufacture, use, sale or importation of licensed products, we materially breach any provisions of the MEEI Agreement or in the event of its insolvency or bankruptcy.
In the event of an early termination of the MEEI Agreement, all rights licensed and developed by the Company under the MEEI Agreement may revert back to MEEI. The Company has agreed to indemnify MEEI for certain claims that may arise under the MEEI Agreement.
14. LEASES
The Company currently leases an office used to conduct business. The exercise of lease renewal options is at the Company’s discretion and the renewal to extend the lease terms are not included in the Company’s Right-Of-Use assets and lease liabilities as they are not reasonably certain of exercise. The Company regularly evaluates the renewal options and when they are reasonably certain of exercise, the Company includes the renewal period in its lease term. As the Company’s lease does not provide an implicit rate, the Company uses its incremental borrowing rate based on the information available at the lease commencement date in determining the present value of the lease payments. In August 2021, the Company entered into a lease amendment extending the lease by 18 months through June 30, 2023. which had an immaterial impact to the balance sheet as of December 31, 2021. For the years ended December 31, 2021 and 2020, right of use assets obtained in exchange for lease obligations were $0.3 million and NaN, respectively.
As of December 31, 2021, the Company maintained an unamortized Right-Of-Use asset with a corresponding operating lease liability of approximately $0.4 million based on the present value of the minimum rental payments in accordance with ASC Topic 842, Leases. The weighted average discount rate used for leases as of December 31, 2021 is 9.1%. The weighted average lease term as of December 31, 2021 is 1.5 years. The operating lease expense for the year ended December 31, 2021 was $0.2 million. Maturities and balance sheet presentation of our lease liabilities for all operating leases as of December 31, 2021 is as follows:
| | | | | |
Remaining total lease payments | | $ | 381,053 | |
Less: effect of discounting | | | (26,214 | ) |
Present value of lease liabilities | | $ | 354,839 | |
| | | |
Current operating lease liabilities | | $ | 229,607 | |
Non-current operating lease liabilities | | | 125,232 | |
Total | | $ | 354,839 | |
128
The Company’s gross future minimum payments under all non-cancelable operating leases as of December 31, 2021 are:
| | | | | | | | | | | | | | | | | | | | |
| | Total | | | 2022 | | | 2023 | | | 2024 | | | 2025 | |
Operating lease obligations | | $ | 381,053 | | | $ | 252,477 | | | $ | 128,576 | | | $ | 0 | | | $ | 0 | |
None.
129









