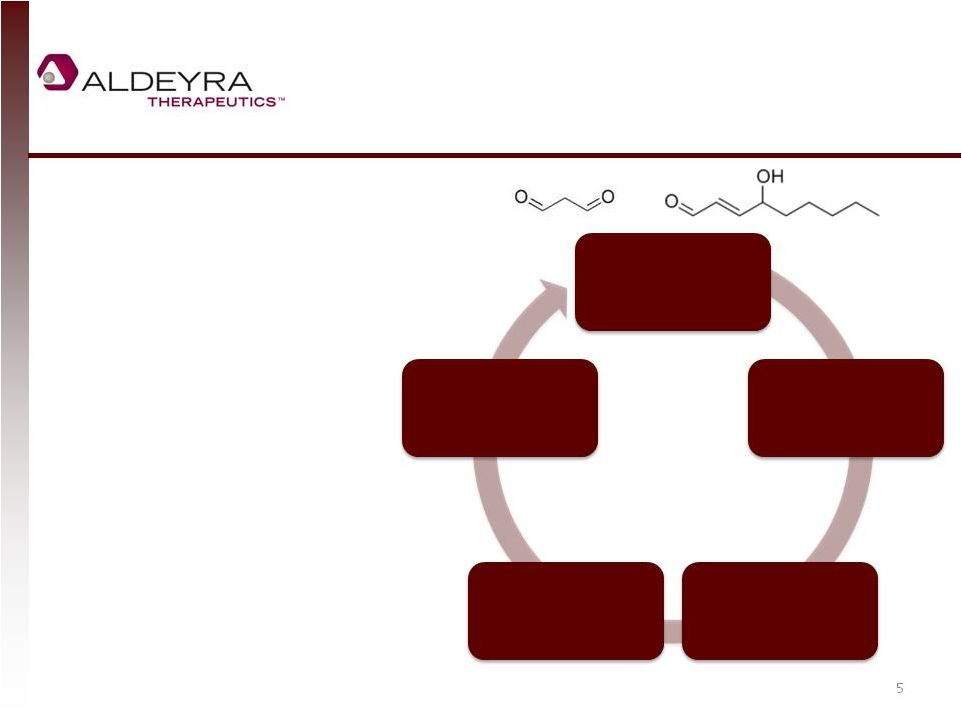
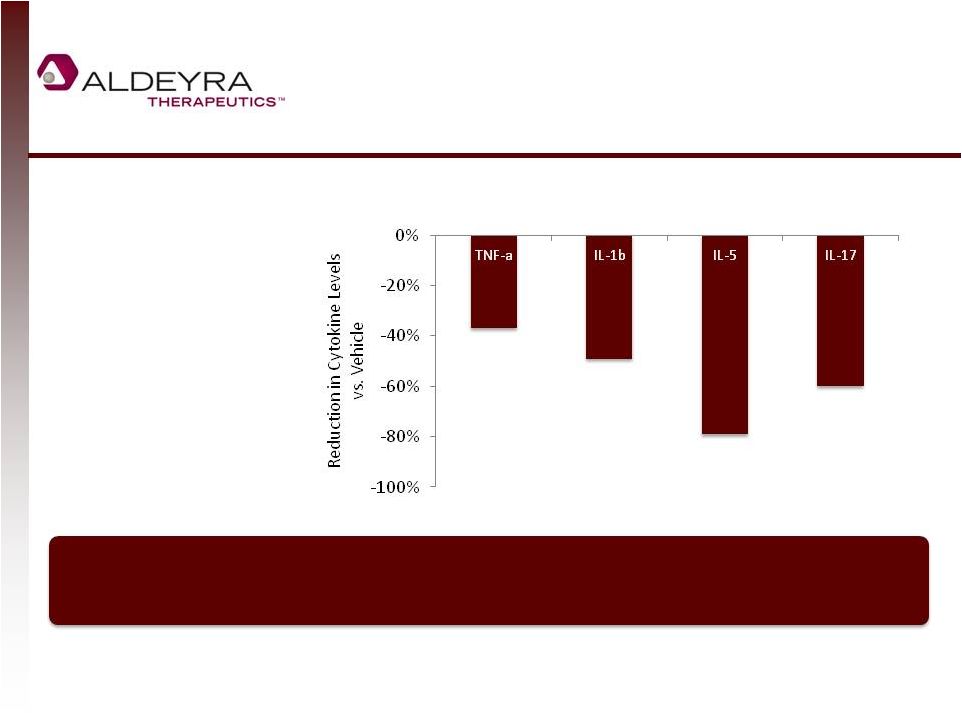
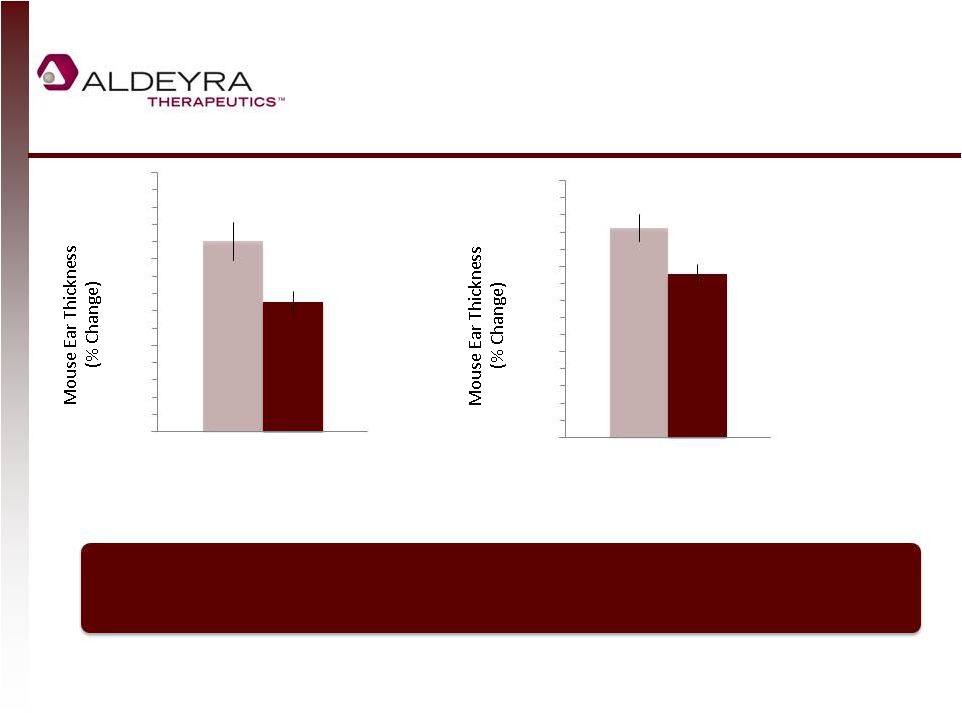
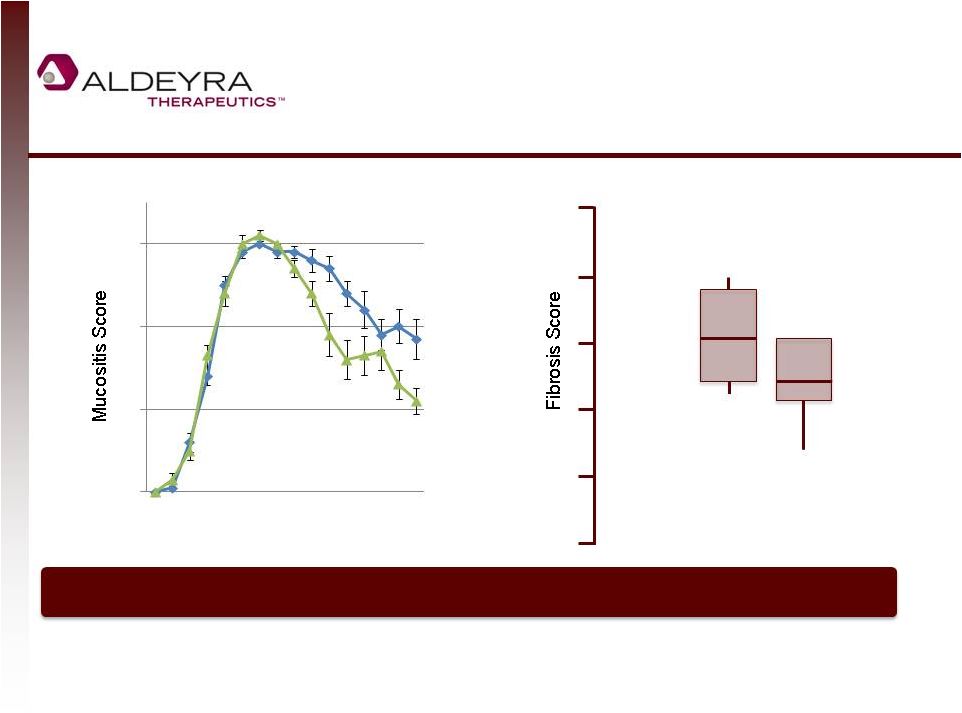
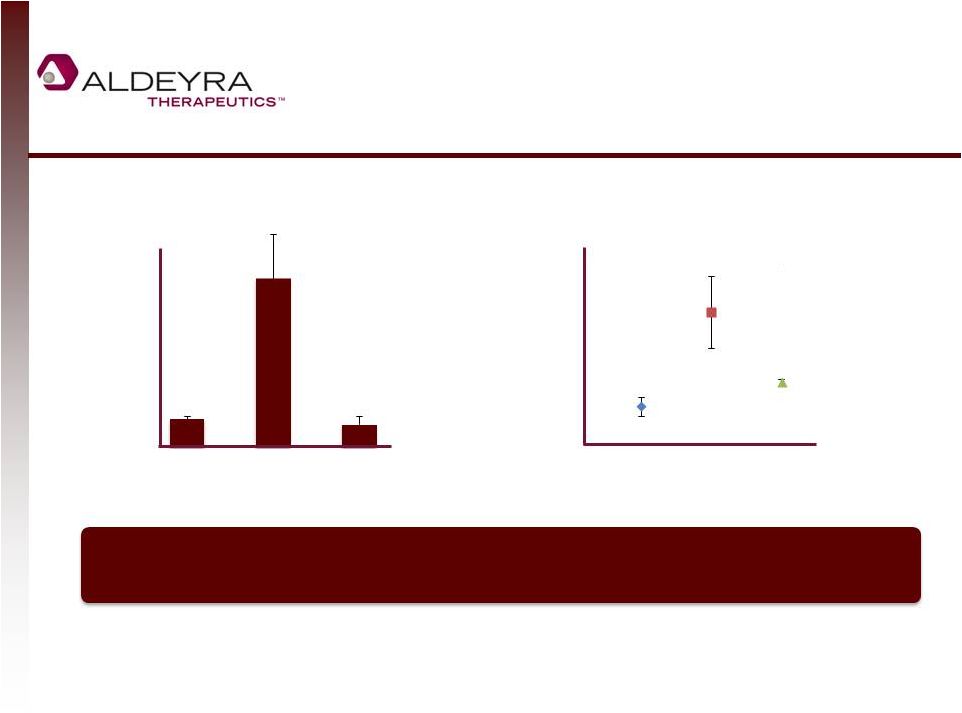
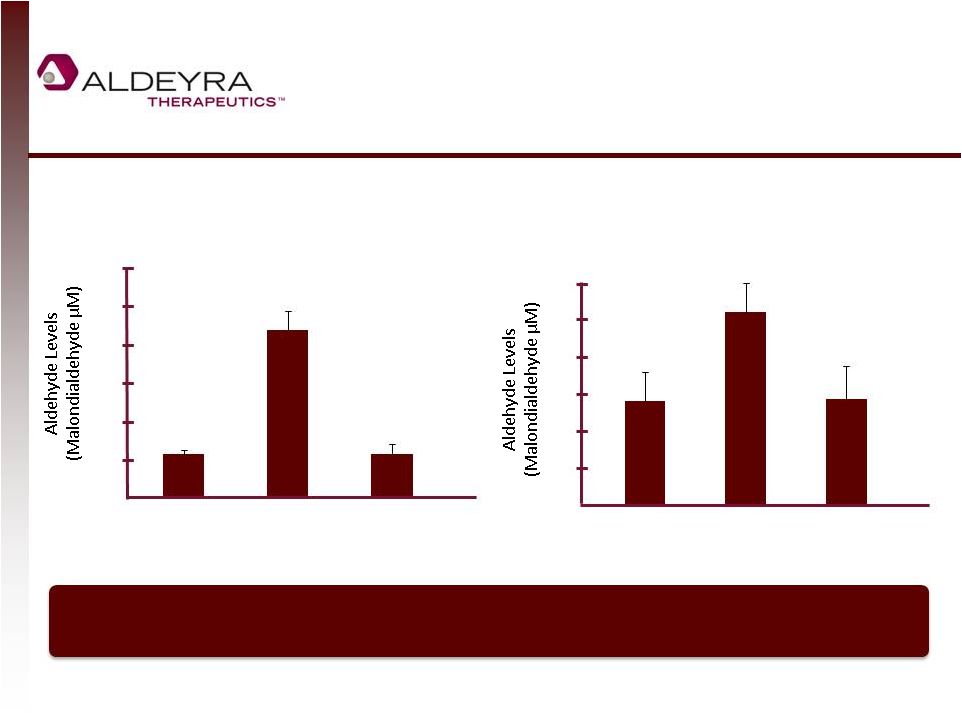
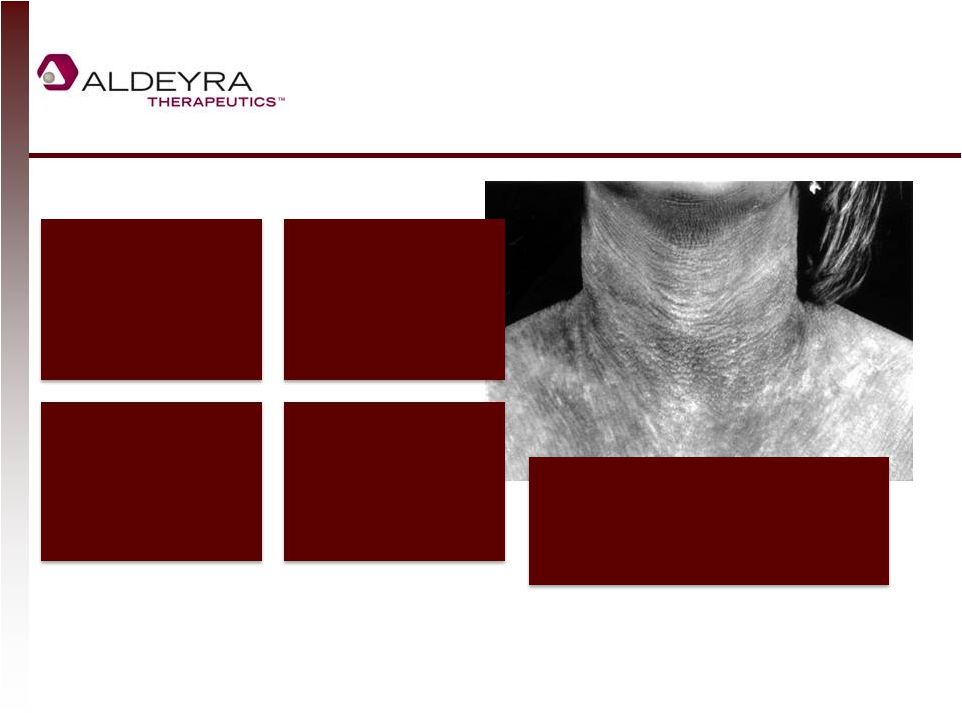
Forward-Looking Statements • This presentation includes statements contains forward-looking statements that involve risks and uncertainties. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “objective,” “intend,” “should,” “could,” “can,” “would,” “expect,” “believe,” “anticipate,” “project,” “target,” “design,” “estimate,” “predict,” “potential,” “plan” or the negative of these terms, and similar expressions intended to identify forward-looking statements. These statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Given these uncertainties, you should not place undue reliance on these forward-looking statements. Forward-looking statements include, but are not limited to, statements about: our expectations regarding our expenses and revenue, the sufficiency of our cash resources and needs for additional financing; our anticipated growth strategies; our expectations regarding competition; the anticipated trends and challenges in our business and the market in which we operate; the timing and success of preclinical studies and clinical trials conducted by us and our development partners; the ability to obtain and maintain regulatory approval of our product candidates, and the labeling for any approved products; the scope, progress, expansion, and costs of developing and commercializing our product candidates; the size and growth of the potential markets for our product candidates and the ability to serve those markets; the rate and degree of market acceptance of any of our product candidates; our ability to establish and maintain development partnerships; our ability to attract or retain key personnel; our expectations regarding federal, state and foreign regulatory requirements; regulatory developments in the United States and foreign countries; and our ability to obtain and maintain intellectual property protection for our product candidates. • Forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performances or achievements expressed or implied by the forward-looking statements. Any forward-looking statement made by us in this presentation speaks only as of the date on which it is made. Except as required by law, we assume no obligation to update these statements publicly, or to update the reasons actual results could differ materially from those anticipated in these statements, even if new information becomes available in the future. • Although we believe that we have a reasonable basis for each forward-looking statement contained in this presentation, we caution you that forward-looking statements are not guarantees of future performance and that our actual results of operation, financial condition and liquidity, and the development of the industry in which we operate may differ materially from the forward-looking statements contained in this presentation as a result of, among other factors, the factors referenced in our final prospectus filed under Rule 424(b)(4) with the Securities and Exchange Commission on May 2, 2014 and in our subsequent filings with the Securities and Exchange Commission. In addition, even if our results of operation, financial condition and liquidity, and the development of the industry in which we operate are consistent with the forward-looking statements contained in this presentation, they may not be predictive of results or developments in future periods. • You should read carefully our filings with the Securities and Exchange Commission, including risk factors described therein, to better understand the risks and uncertainties inherent in our business and underlying any forward-looking statements. 2 |