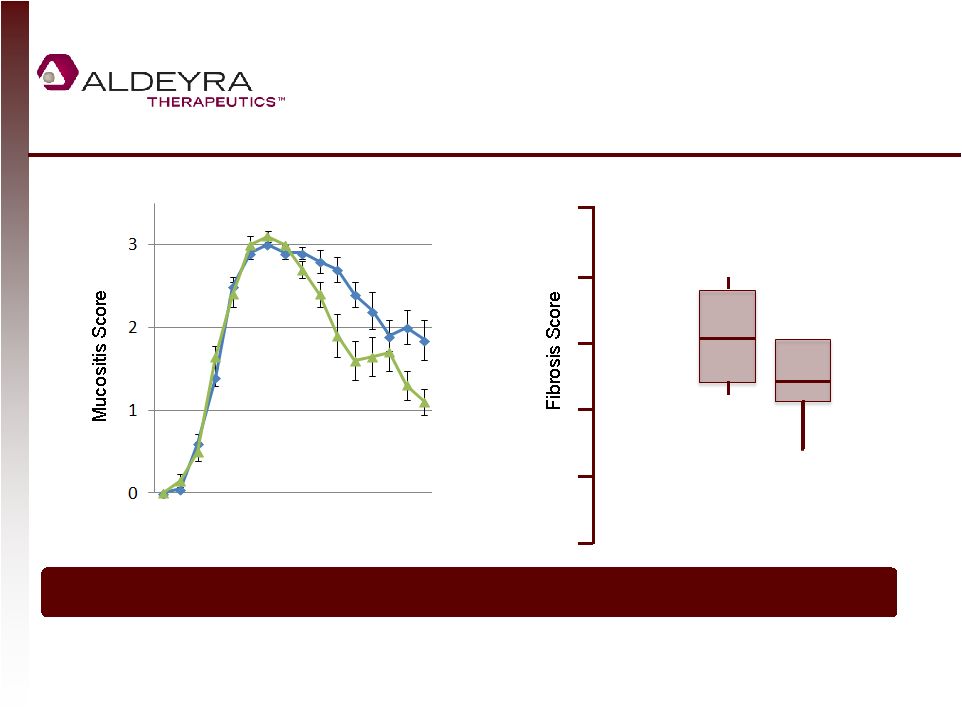
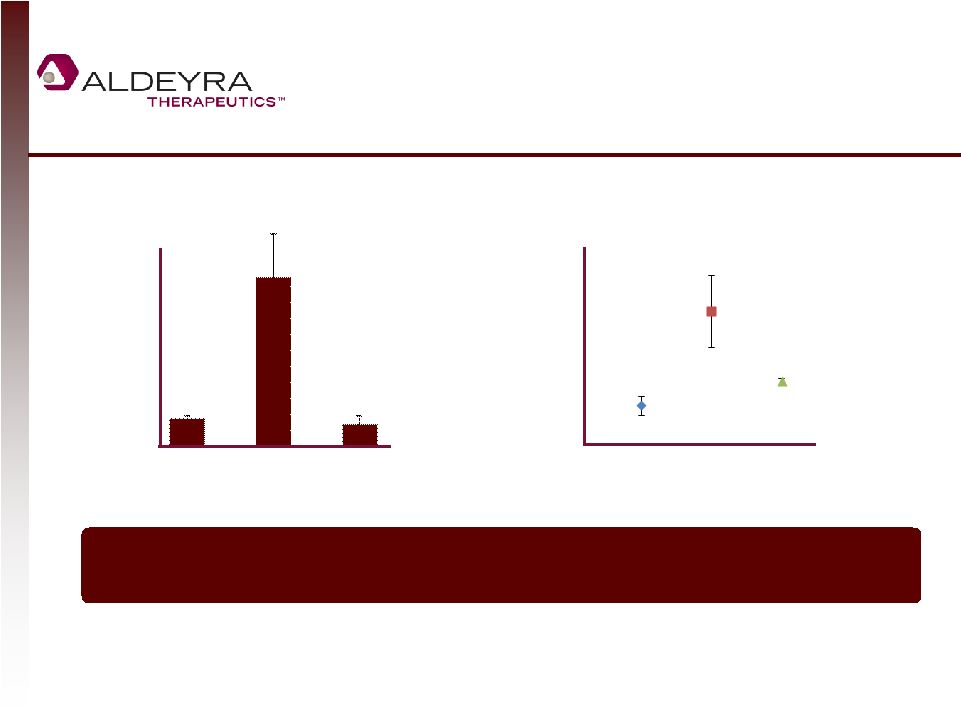
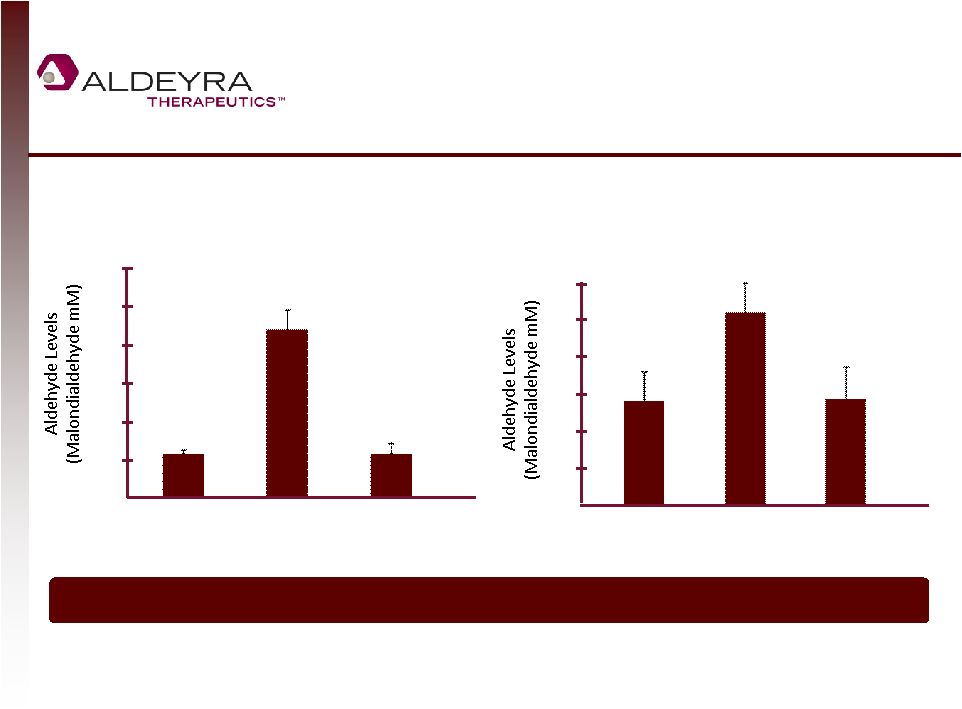
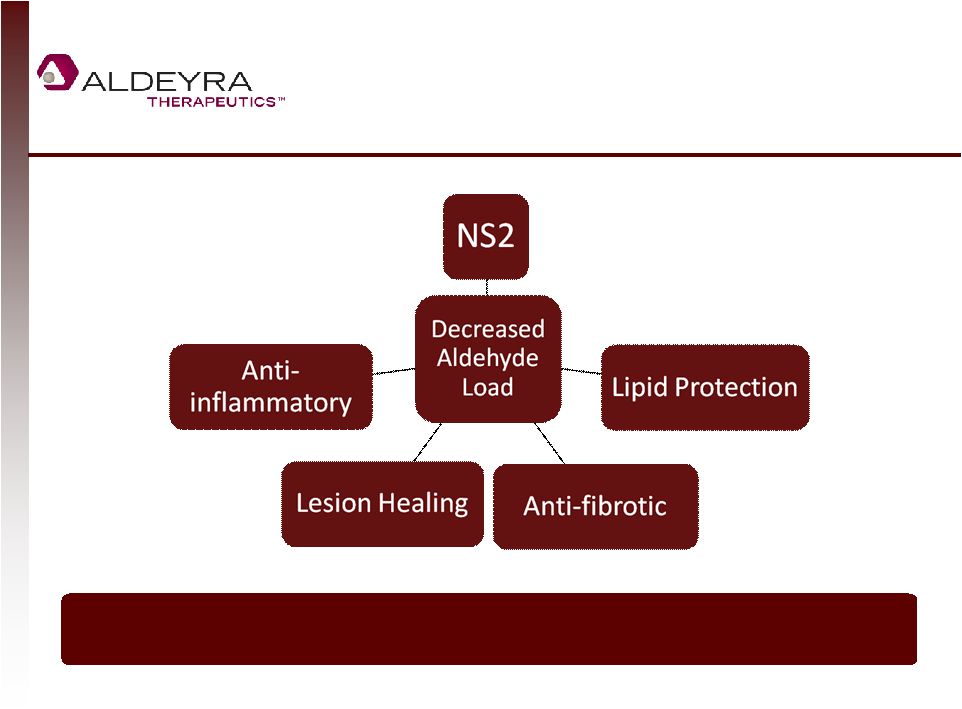
Forward-Looking Statements • This presentation contains forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, including statements regarding Aldeyra's plans for its product candidates. In some cases, you can identify forward-looking statements by terms such as "may," "might," "will," "objective," "intend," "should," "could," "can," "would," "expect," "believe," "anticipate," "project," "target," "design," "estimate," "predict," "potential," "aim," "plan" or the negative of these terms, and similar expressions intended to identify forward-looking statements. • Such forward-looking statements are based upon current expectations that involve risks, changes in circumstances, assumptions and uncertainties. Aldeyra is at an early stage of development and may not ever have any products that generate significant revenue. Important factors that could cause actual results to differ materially from those reflected in Aldeyra's forward-looking statements include, among others, the timing and success of preclinical studies and clinical trials conducted by Aldeyra and its development partners; the ability to obtain and maintain regulatory approval to conduct clinical trials and to commercialize Aldeyra's product candidates, and the labeling for any approved products; the scope, progress, expansion, and costs of developing and commercializing Aldeyra's product candidates; the size and growth of the potential markets for Aldeyra's product candidates and the ability to serve those markets; Aldeyra's expectations regarding Aldeyra's expenses and revenue, the sufficiency of Aldeyra's cash resources and needs for additional financing; Aldeyra's ability to attract or retain key personnel; and other factors that are described in the "Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations" sections of Aldeyra's Quarterly Report on Form 10-Q for the quarter ended September 30, 2014 which is on file with the Securities and Exchange Commission (SEC) and available on the SEC's website at www.sec.gov. • In addition to the risks described above and in Aldeyra's other filings with the SEC, other unknown or unpredictable factors also could affect Aldeyra's results. No forward-looking statements can be guaranteed and actual results may differ materially from such statements. The information in this presentation is provided only as of the date of this release, and Aldeyra undertakes no obligation to update any forward-looking statements contained in this release on account of new information, future events, or otherwise, except as required by law. 2 |