
mesoblast Global Leader in Allogeneic Cellular Medicines for Inflammatory Diseases January 2023 ASX: MSB; Nasdaq: MESO Corporate Presentation BIOTECH SHOWCASE 2023 Exhibit 99.1

CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS This presentation includes forward-looking statements that relate to future events or our future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to differ materially from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. We make such forward- looking statements pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. All statements other than statements of historical facts contained in this presentation are forward-looking statements. Words such as, but not limited to, “believe,” “expect,” “anticipate,” “estimate,” “intend,” “plan,” “targets,” “likely,” “will,” “would,” “could,” and similar expressions or phrases identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and future events , recent changes in regulatory laws, and financial trends that we believe may affect our financial condition, results of operation, business strategy and financial needs. These statements may relate to, but are not limited to: expectations regarding the safety or efficacy of, or potential applications for, Mesoblast's adult stem cell technologies; expectations regarding the strength of Mesoblast's intellectual property, the timeline for Mesoblast's regulatory approval process, and the scalability and efficiency of manufacturing processes; expectations about Mesoblast's ability to grow its business and statements regarding its relationships with current and potential future business partners and future benefits of those relationships; statements concerning Mesoblast's share price or potential market capitalization; and statements concerning Mesoblast's capital requirements and ability to raise future capital, among others. Forward-looking statements should not be read as a guarantee of future performance or results, and actual results may differ from the results anticipated in these forward-looking statements, and the differences may be material and adverse. You should read this presentation together with our financial statements and the notes related thereto, as well as the risk factors, in our most recently filed reports with the SEC or on our website. Uncertainties and risks that may cause Mesoblast's actual results, performance or achievements to be materially different from those which may be expressed or implied by such statements, include, without limitation: risks inherent in the development and commercialization of potential products; uncertainty of clinical trial results or regulatory approvals or clearances; government regulation; the need for future capital; dependence upon collaborators; and protection of our intellectual property rights, among others. Accordingly, you should not place undue reliance on these forward-looking statements. We do not undertake any obligations to publicly update or revise any forward-looking statements, whether as a result of new information, future developments or otherwise.

Our Mission Mesoblast is committed to bringing to market innovative cellular medicines to treat serious and life-threatening illnesses

4 m e s o b l a s t Investment Highlights First FDA Approval Expected in 2023 Potential 2023 Launch for Remestemcel-L/SR-aGVHD Innovative Late-Stage Pipeline Novel Allogeneic Cell Therapy Platform Cash Resources to Support Operations thru 2023+ Remestemcel-L BLA to be resubmitted for children with steroid-refractory acute graft versus host disease (SR-aGVHD), with potential US approval in mid-CY2023 Rexlemestrocel-L progressing towards initiation of a second pivotal Phase 3 study commencing H1 CY2023 for discogenic chronic low back pain (CLBP), followed by chronic heart failure with reduced ejection fraction (HFrEF) Developing an off-the-shelf, allogeneic cell therapy platform based on mesenchymal stromal cell technology to enable treatment without the need for donor matching or immunosuppression Cash-on-hand of US$85.5 million plus up to an additional US$40 million from existing financing facilities, subject to certain milestones, with potential upside from partnering income

5 m e s o b l a s t Remestemcel-L / SR-aGVHD Biologics License Application (BLA) Resubmission Milestones Updated FDA IND File in Oct-22 New Long-term Survival Data from Phase 3 trial Potency Assay Work Completed Currently in Publishing, ahead of Resubmission Submitted substantial new information on clinical and potency assay items identified in the CRL received from FDA in 2020 to Investigational New Drug (IND) file for remestemcel-L in the treatment of children with SR-aGVHD In Nov-22 released long-term survival results from pivotal trial showing durable survival through 4 years of follow-up. These new long-term survival data are a key component of the BLA resubmission Optimization and validation work completed on the potency assay that was in place at the time of the Phase 3 trial and which demonstrates a relationship between the product’s activity in-vitro and its effects on survival, addressing a key component of CRL Final drafting of the BLA is complete and the document is with the publisher for filing
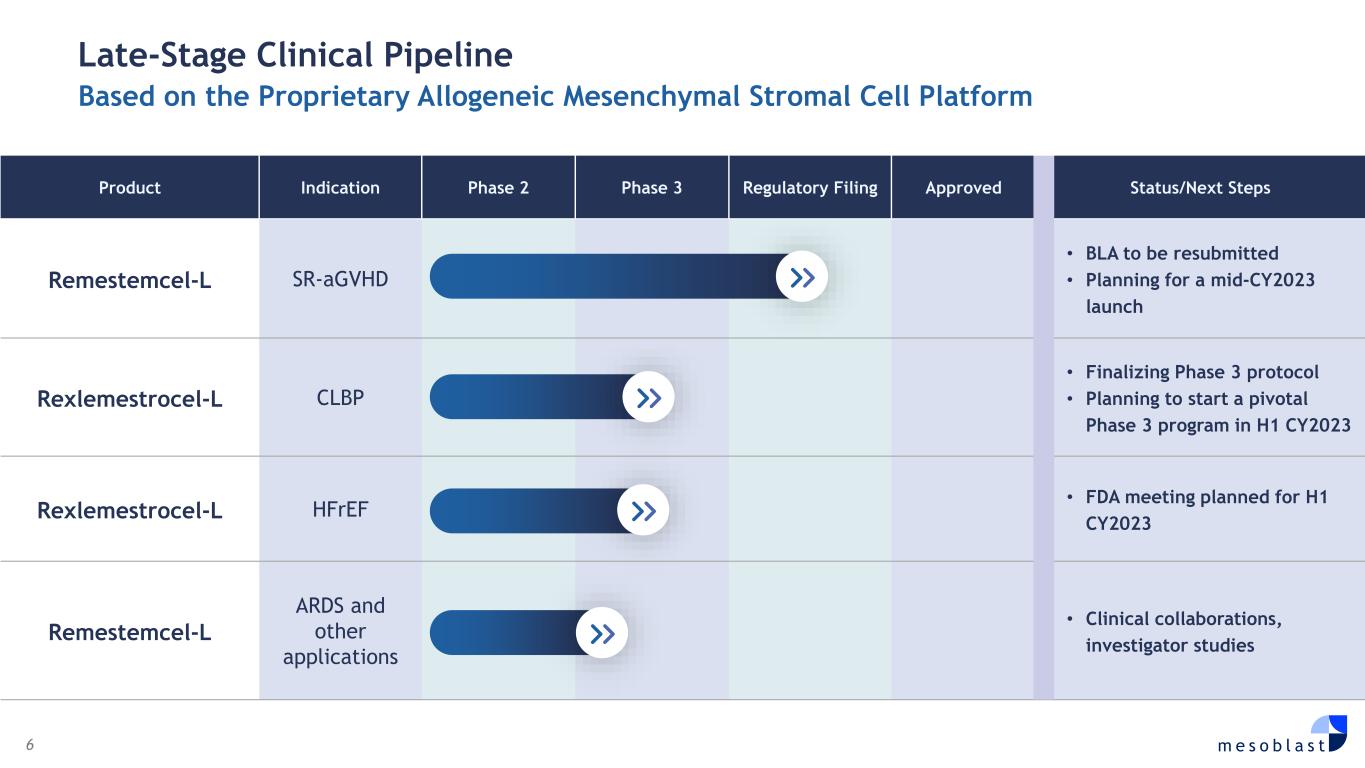
6 m e s o b l a s t Late-Stage Clinical Pipeline Based on the Proprietary Allogeneic Mesenchymal Stromal Cell Platform Product Indication Phase 2 Phase 3 Regulatory Filing Approved Status/Next Steps Remestemcel-L SR-aGVHD • BLA to be resubmitted • Planning for a mid-CY2023 launch Rexlemestrocel-L CLBP • Finalizing Phase 3 protocol • Planning to start a pivotal Phase 3 program in H1 CY2023 Rexlemestrocel-L HFrEF • FDA meeting planned for H1 CY2023 Remestemcel-L ARDS and other applications • Clinical collaborations, investigator studies
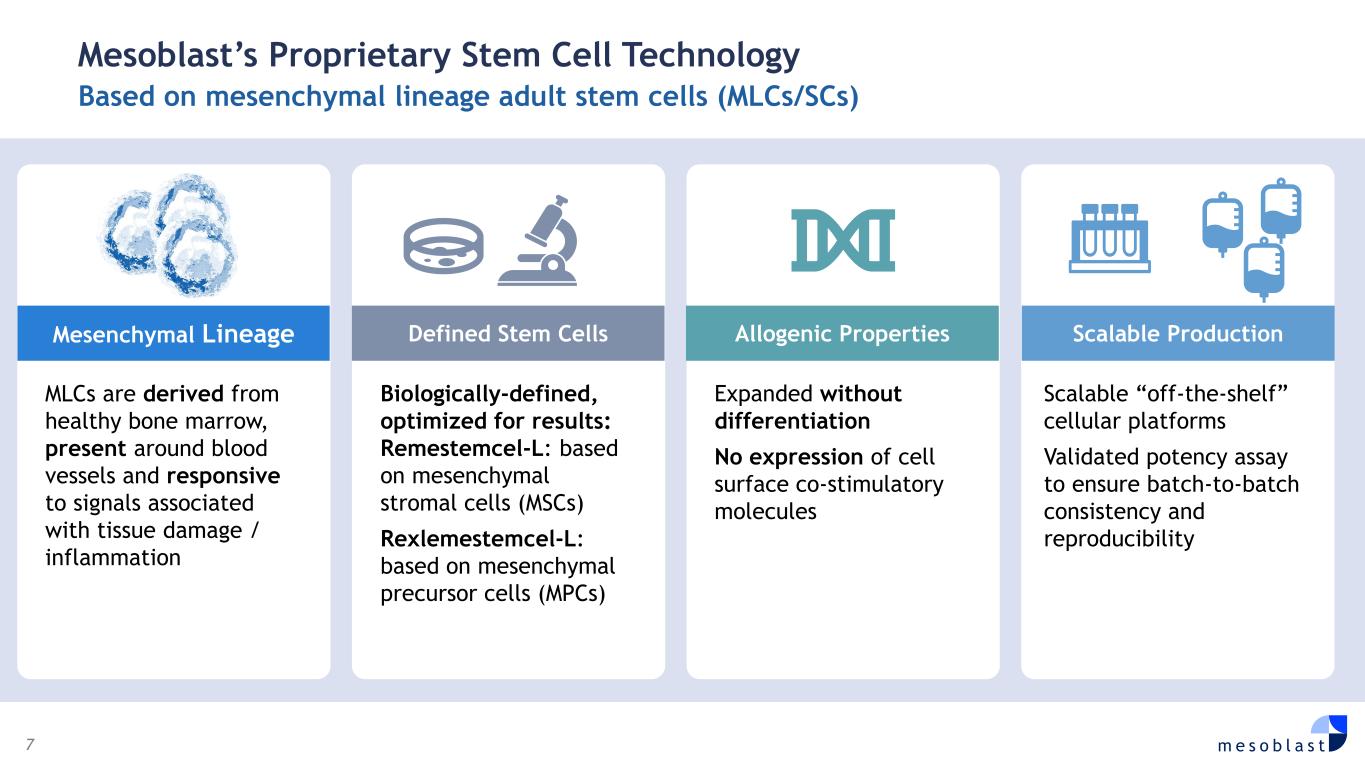
7 m e s o b l a s t Mesoblast’s Proprietary Stem Cell Technology Based on mesenchymal lineage adult stem cells (MLCs/SCs) MLCs are derived from healthy bone marrow, present around blood vessels and responsive to signals associated with tissue damage / inflammation Biologically-defined, optimized for results: Remestemcel-L: based on mesenchymal stromal cells (MSCs) Rexlemestemcel-L: based on mesenchymal precursor cells (MPCs) Expanded without differentiation No expression of cell surface co-stimulatory molecules Scalable “off-the-shelf” cellular platforms Validated potency assay to ensure batch-to-batch consistency and reproducibility Mesenchymal Lineage Defined Stem Cells Allogenic Properties Scalable Production

8 m e s o b l a s t Remestemcel-L Steroid-Refractory Acute Graft Versus Host Disease (SR- aGVHD) BLA to be resubmitted. Tracking to a potential mid-CY2023 approval

9 m e s o b l a s t 1. Westin, J., Saliba, RM., Lima, M. (2011) Steroid-refractory acute GVHD: predictors and outcomes. Advances in Hematology. 2. Anthem-HealthCore/Mesoblast claims analysis (2016). Data on file 3. Niederwieser D, Baldomero H, Szer J. (2016) Hematopoietic stem cell transplantation activity worldwide in 2012 and a SWOT analysis of the Worldwide Network for Blood and Marrow Transplantation Group including the global survey. 4. HRSA Transplant Activity Report, CIBMTR, 2019 5. Axt L, Naumann A, Toennies J (2019) Retrospective single center analysis of outcome, risk factors and therapy in steroid refractory graft-versus-host disease after allogeneic hematopoietic cell transplantion. Bone Marrow Transplantation. More than 30,000 allogeneic BMTs performed globally (>20K US/EU) annually, ~20% pediatric3,4 Approx. 1,500 allogeneic BMTs in children and adolescents in US4 Remestemcel-L: Steroid-Refractory Acute Graft Versus Host Disease (SR-aGVHD) SR-aGVHD is associated with mortality rates as high as 90% Acute GVHD is a life- threatening complication that occurs in ~50% of patients receiving allogeneic bone marrow transplants (BMTs)1 Acute GVHD primarily affects skin, GI tract, and liver Steroid-refractory aGVHD is associated with mortality rates as high as 90%1,5 and significant extended hospital stay costs2 Treatment Options Burden of Illness Market Opportunity Corticosteroids are first-line therapy for aGVHD There is only one approved treatment for disease refractory to steroids and no approved treatment in the US for children under 12 years old In Japan, Mesoblast’s licensee has received the only product approval for SR-aGVHD in both children and adults
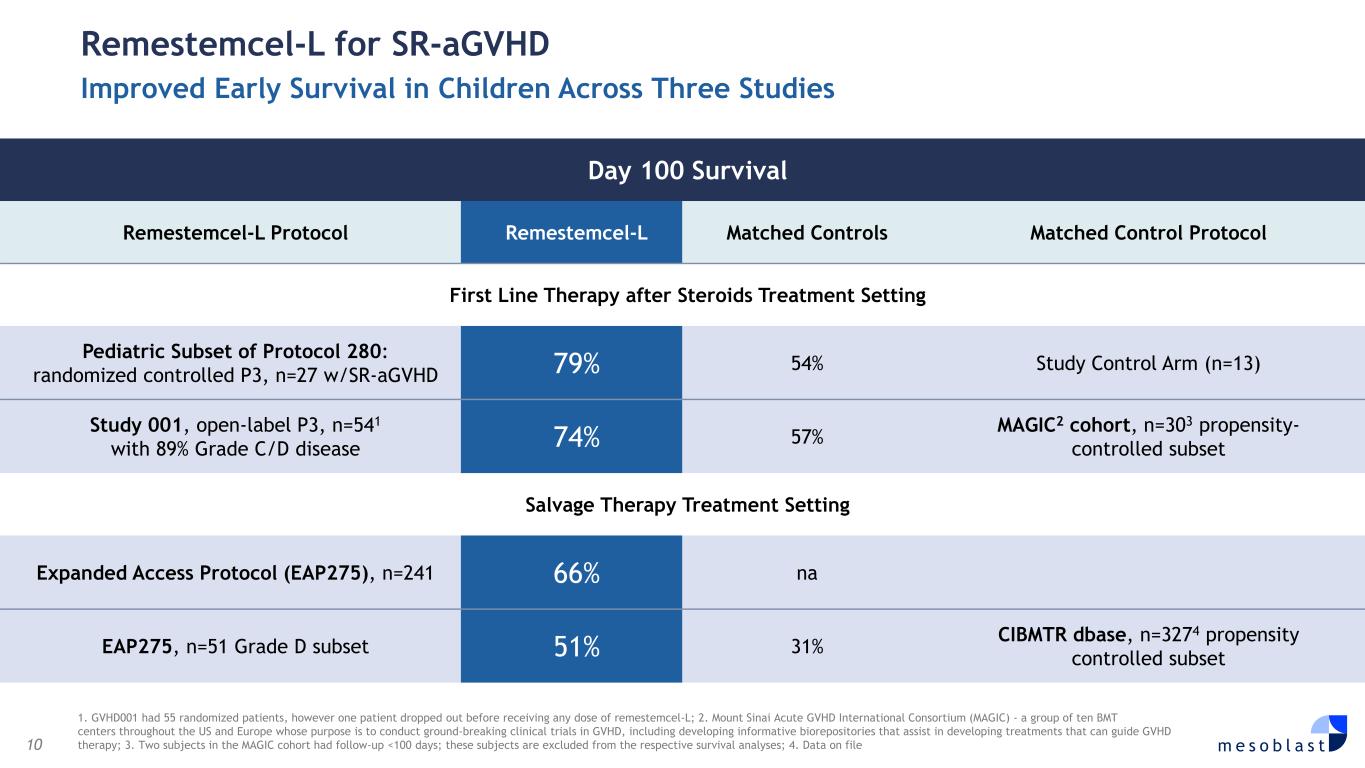
10 m e s o b l a s t 1. GVHD001 had 55 randomized patients, however one patient dropped out before receiving any dose of remestemcel-L; 2. Mount Sinai Acute GVHD International Consortium (MAGIC) - a group of ten BMT centers throughout the US and Europe whose purpose is to conduct ground-breaking clinical trials in GVHD, including developing informative biorepositories that assist in developing treatments that can guide GVHD therapy; 3. Two subjects in the MAGIC cohort had follow-up <100 days; these subjects are excluded from the respective survival analyses; 4. Data on file Remestemcel-L for SR-aGVHD Improved Early Survival in Children Across Three Studies Day 100 Survival Remestemcel-L Protocol Remestemcel-L Matched Controls Matched Control Protocol First Line Therapy after Steroids Treatment Setting Pediatric Subset of Protocol 280: randomized controlled P3, n=27 w/SR-aGVHD 79% 54% Study Control Arm (n=13) Study 001, open-label P3, n=541 with 89% Grade C/D disease 74% 57% MAGIC2 cohort, n=303 propensity- controlled subset Salvage Therapy Treatment Setting Expanded Access Protocol (EAP275), n=241 66% na EAP275, n=51 Grade D subset 51% 31% CIBMTR dbase, n=3274 propensity controlled subset
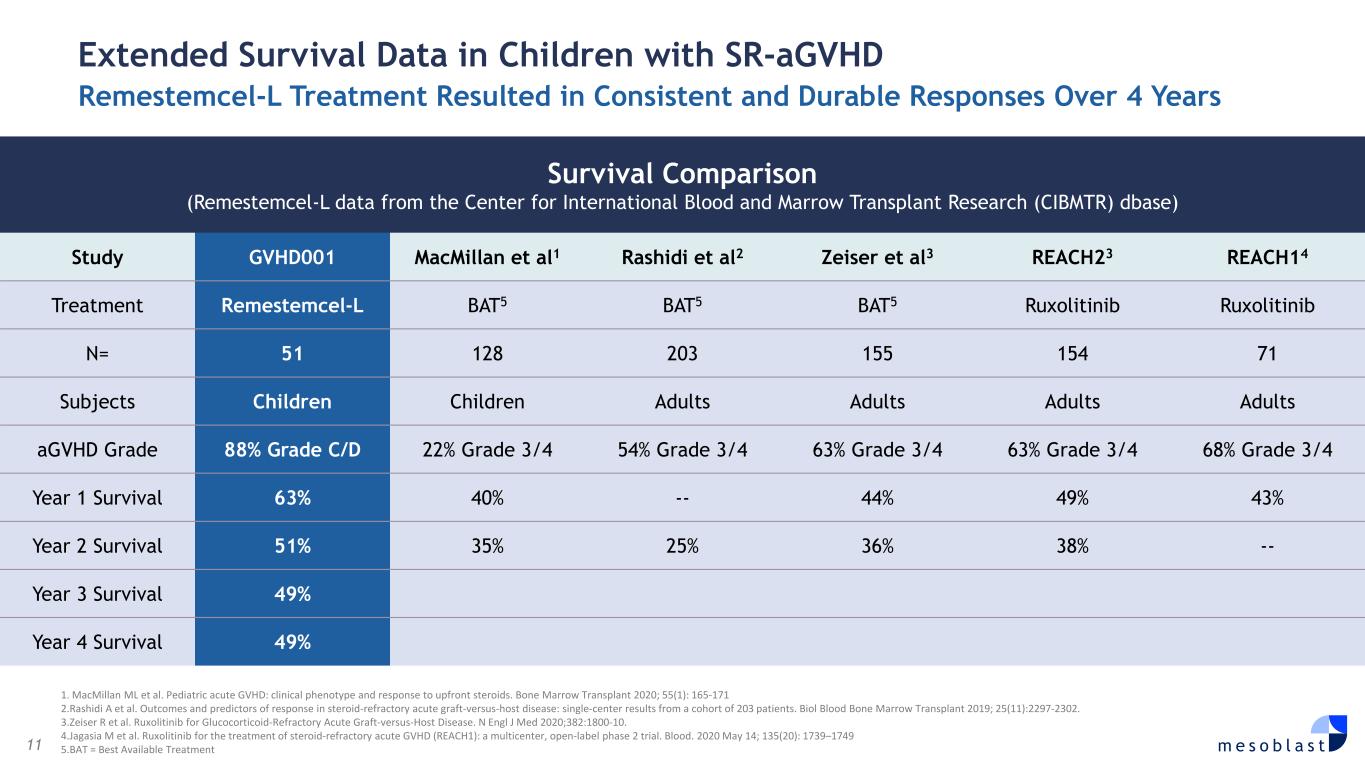
11 m e s o b l a s t 1. MacMillan ML et al. Pediatric acute GVHD: clinical phenotype and response to upfront steroids. Bone Marrow Transplant 2020; 55(1): 165-171 2.Rashidi A et al. Outcomes and predictors of response in steroid-refractory acute graft-versus-host disease: single-center results from a cohort of 203 patients. Biol Blood Bone Marrow Transplant 2019; 25(11):2297-2302. 3.Zeiser R et al. Ruxolitinib for Glucocorticoid-Refractory Acute Graft-versus-Host Disease. N Engl J Med 2020;382:1800-10. 4.Jagasia M et al. Ruxolitinib for the treatment of steroid-refractory acute GVHD (REACH1): a multicenter, open-label phase 2 trial. Blood. 2020 May 14; 135(20): 1739–1749 5.BAT = Best Available Treatment Extended Survival Data in Children with SR-aGVHD Remestemcel-L Treatment Resulted in Consistent and Durable Responses Over 4 Years Survival Comparison (Remestemcel-L data from the Center for International Blood and Marrow Transplant Research (CIBMTR) dbase) Study GVHD001 MacMillan et al1 Rashidi et al2 Zeiser et al3 REACH23 REACH14 Treatment Remestemcel-L BAT5 BAT5 BAT5 Ruxolitinib Ruxolitinib N= 51 128 203 155 154 71 Subjects Children Children Adults Adults Adults Adults aGVHD Grade 88% Grade C/D 22% Grade 3/4 54% Grade 3/4 63% Grade 3/4 63% Grade 3/4 68% Grade 3/4 Year 1 Survival 63% 40% -- 44% 49% 43% Year 2 Survival 51% 35% 25% 36% 38% -- Year 3 Survival 49% Year 4 Survival 49%
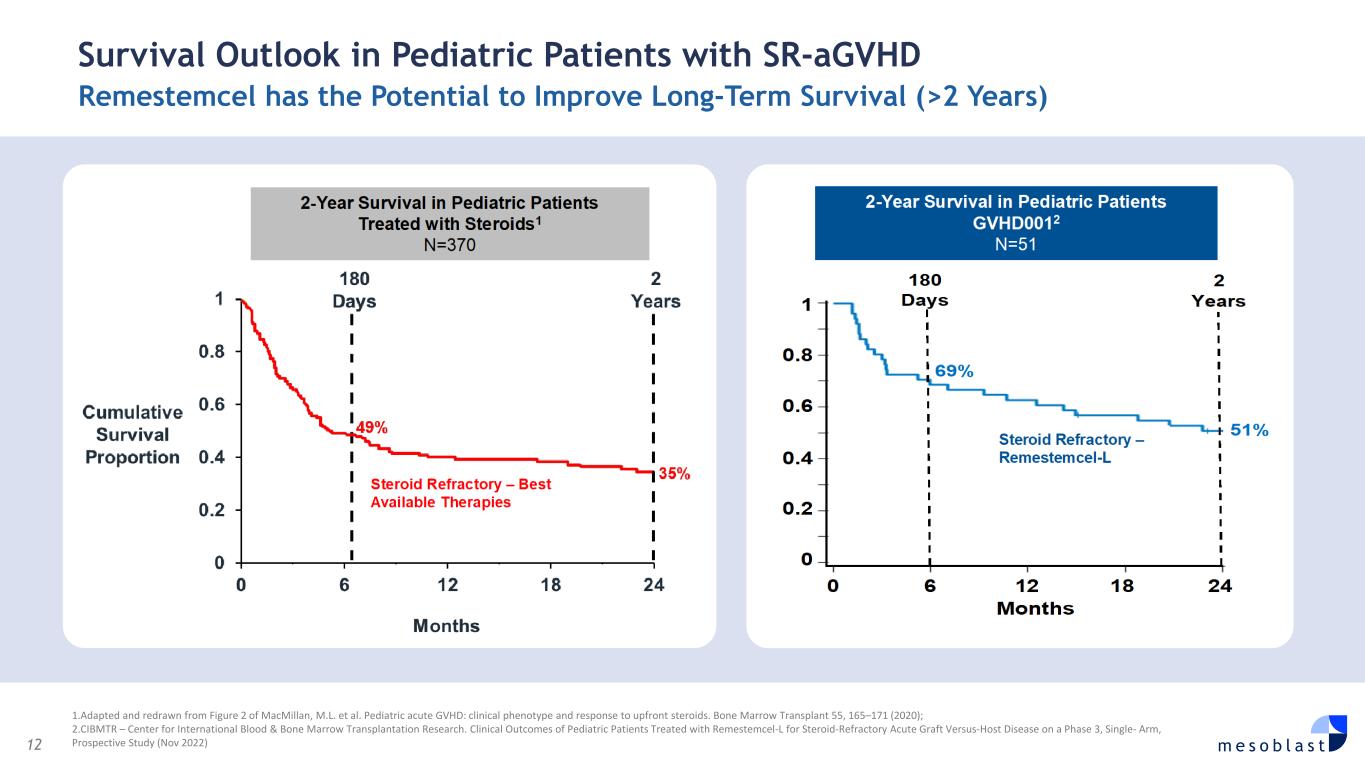
12 m e s o b l a s t Survival Outlook in Pediatric Patients with SR-aGVHD Remestemcel has the Potential to Improve Long-Term Survival (>2 Years) 1.Adapted and redrawn from Figure 2 of MacMillan, M.L. et al. Pediatric acute GVHD: clinical phenotype and response to upfront steroids. Bone Marrow Transplant 55, 165–171 (2020); 2.CIBMTR – Center for International Blood & Bone Marrow Transplantation Research. Clinical Outcomes of Pediatric Patients Treated with Remestemcel-L for Steroid-Refractory Acute Graft Versus-Host Disease on a Phase 3, Single- Arm, Prospective Study (Nov 2022)
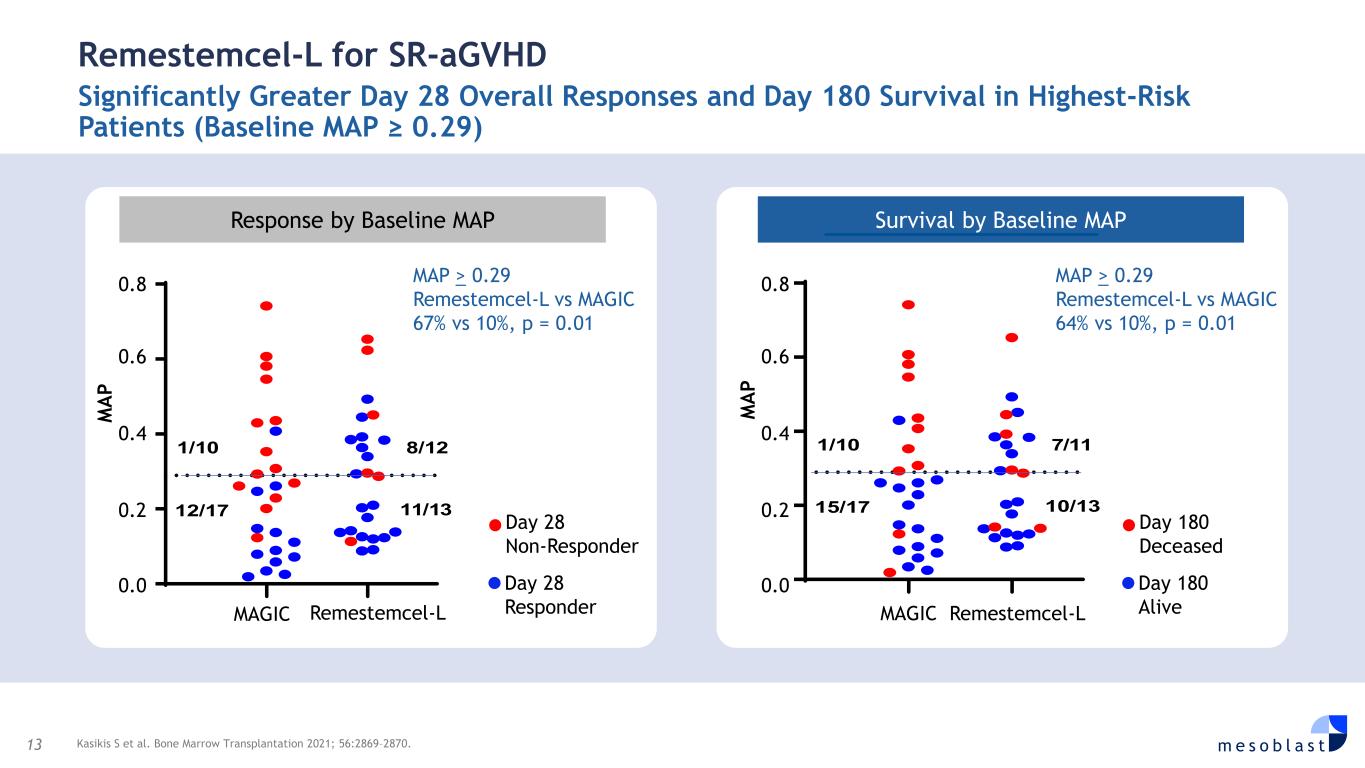
13 m e s o b l a s t Remestemcel-L for SR-aGVHD Significantly Greater Day 28 Overall Responses and Day 180 Survival in Highest-Risk Patients (Baseline MAP ≥ 0.29) Kasikis S et al. Bone Marrow Transplantation 2021; 56:2869–2870. Response by Baseline MAP Survival by Baseline MAP Day 28 Non-Responder Day 28 Responder 1/10 8/12 12/17 11/13 MAP > 0.29 Remestemcel-L vs MAGIC 67% vs 10%, p = 0.01 Remestemcel-LMAGIC M A P 0.8 0.6 0.4 0.2 0.0 1/10 7/11 15/17 10/13 MAP > 0.29 Remestemcel-L vs MAGIC 64% vs 10%, p = 0.01 Remestemcel-LMAGIC M A P 0.8 0.6 0.4 0.2 0.0 Day 180 Deceased Day 180 Alive
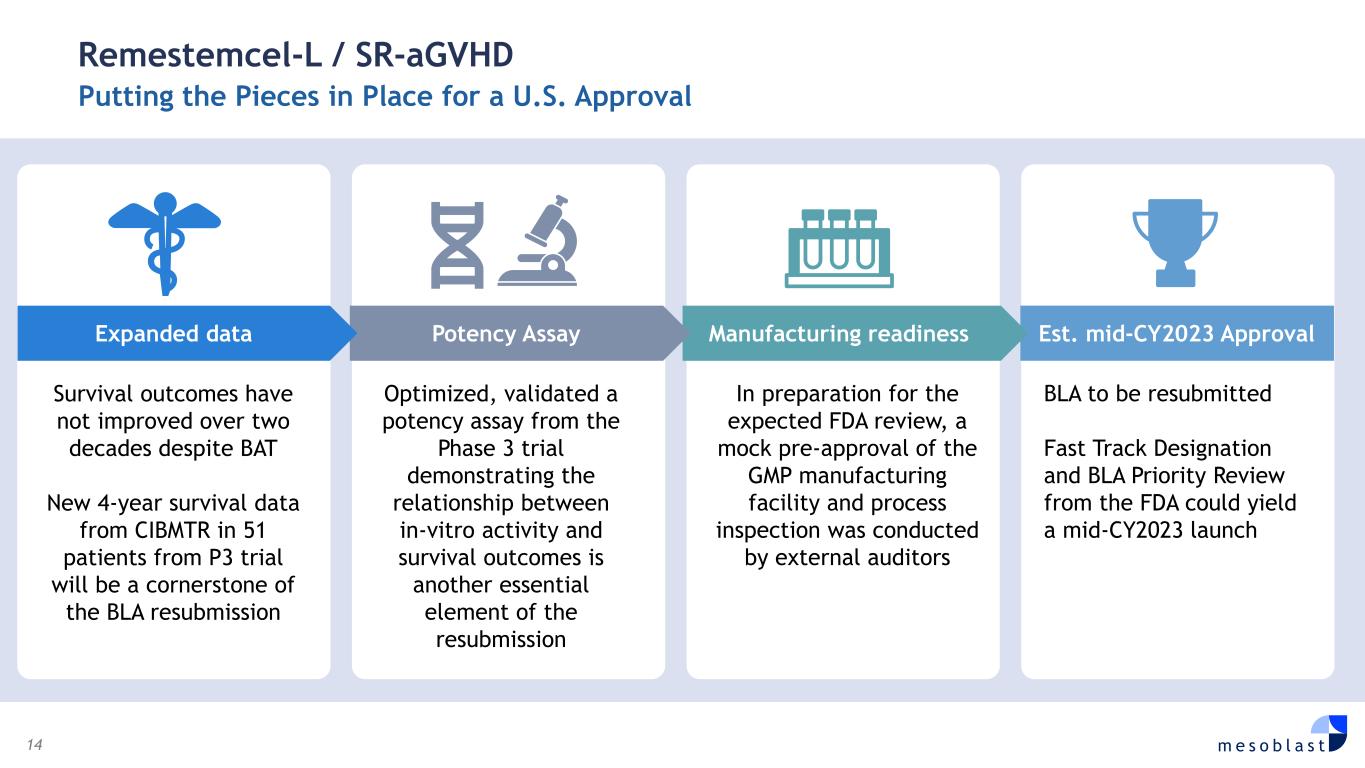
14 m e s o b l a s t Remestemcel-L / SR-aGVHD Putting the Pieces in Place for a U.S. Approval Survival outcomes have not improved over two decades despite BAT New 4-year survival data from CIBMTR in 51 patients from P3 trial will be a cornerstone of the BLA resubmission Optimized, validated a potency assay from the Phase 3 trial demonstrating the relationship between in-vitro activity and survival outcomes is another essential element of the resubmission In preparation for the expected FDA review, a mock pre-approval of the GMP manufacturing facility and process inspection was conducted by external auditors BLA to be resubmitted Fast Track Designation and BLA Priority Review from the FDA could yield a mid-CY2023 launch Est. mid-CY2023 ApprovalManufacturing readinessPotency AssayExpanded data
15 m e s o b l a s t Rexlemestrocel-L Chronic Low Back Pain associated with Degenerative Disc Disease (CLBP) Commence Pivotal Phase 3 Study in CY2023 with Reduction in Pain Primary Endpoint

16 m e s o b l a s t 1. Williams, J., NG, Nawi, Pelzter, K. (2015) Risk factors and disability associated with low back pain in older adults in low-and middle-income countries. Results from the WHO Study on global ageing and adult health (SAGE). PloS One. 2015; 10(6): e0127880., 2.Decision Resources: Chronic Pain December 2015., 3. LEK & NCI opinion leader interviews, and secondary analysis., 4. Navigant: Commercial Assessment for a Proprietary Cell-Based Therapy for DDD in the U.S. and the EU3 – August 2014. Chronic Low Back Pain Due to Degenerative Disc Disease (CLBP) Impacts 7M+ Rexlemestrocel-L represents a potential new paradigm for the treatment of CLBP Over 7m patients are estimated to suffer from CLBP due to degenerative disc disease (DDD) in each of the U.S. and E.U.5 2-4 Minimal treatment options for patients with chronic low back pain (CLBP) who fail conservative therapy include opioids and surgery 50% of opioid prescriptions are for CLBP2 Durable improvement in pain has potential to reduce opioid use and prevent surgical intervention Burden of Illness Treatment Options Market Opportunity Back pain causes more disability than any other condition1 Inflicts substantial direct and indirect costs on the healthcare system,1 including excessive use of opioids in this patient population
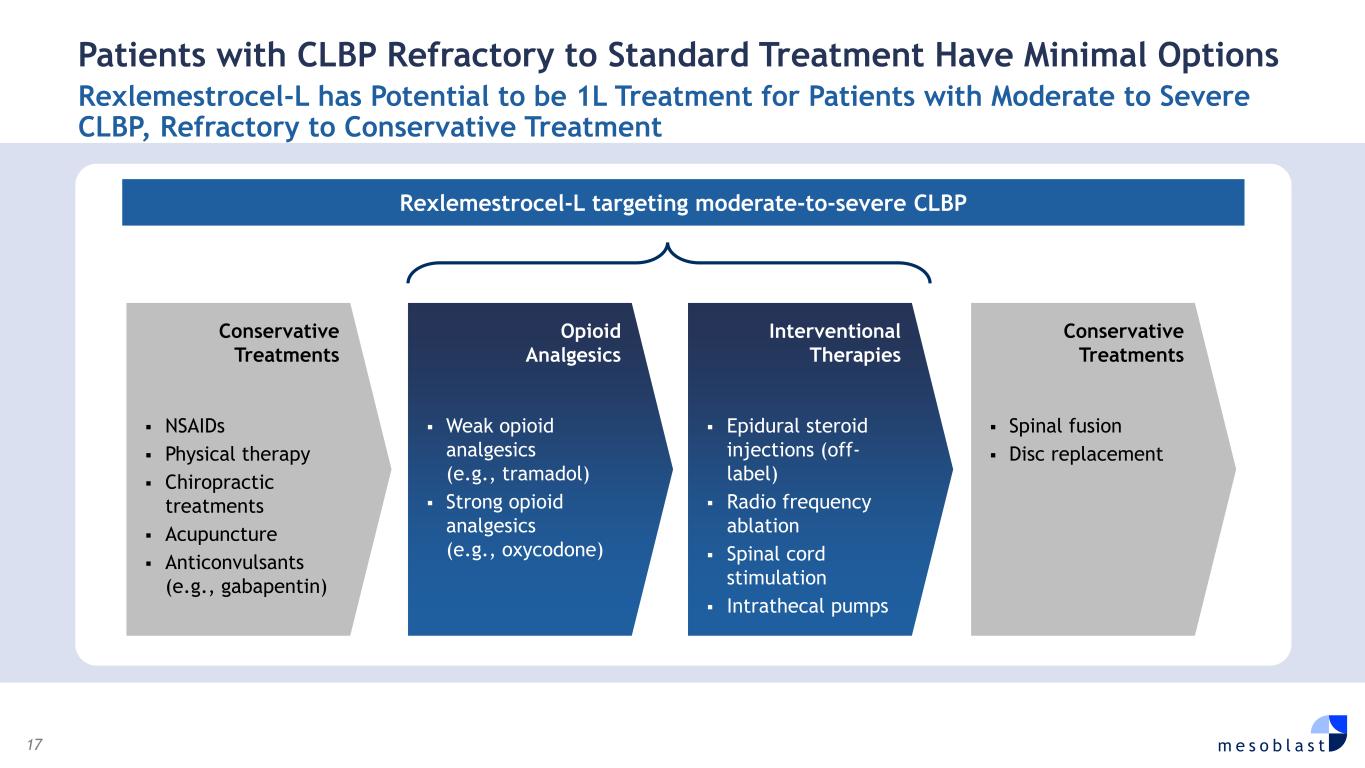
17 m e s o b l a s t Patients with CLBP Refractory to Standard Treatment Have Minimal Options Rexlemestrocel-L has Potential to be 1L Treatment for Patients with Moderate to Severe CLBP, Refractory to Conservative Treatment Rexlemestrocel-L targeting moderate-to-severe CLBP NSAIDs Physical therapy Chiropractic treatments Acupuncture Anticonvulsants (e.g., gabapentin) Conservative Treatments Weak opioid analgesics (e.g., tramadol) Strong opioid analgesics (e.g., oxycodone) Opioid Analgesics Epidural steroid injections (off- label) Radio frequency ablation Spinal cord stimulation Intrathecal pumps Interventional Therapies Spinal fusion Disc replacement Conservative Treatments
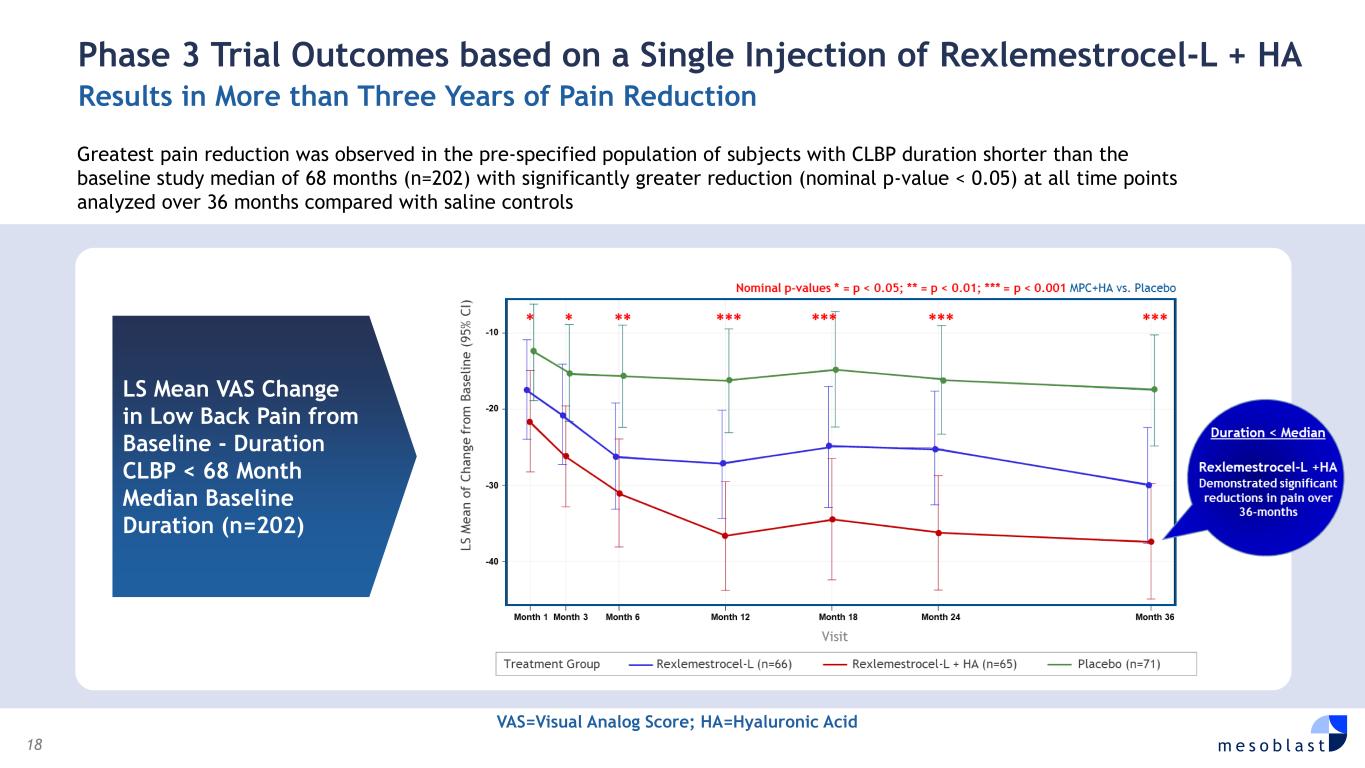
18 m e s o b l a s t LS Mean VAS Change in Low Back Pain from Baseline - Duration CLBP < 68 Month Median Baseline Duration (n=202) Phase 3 Trial Outcomes based on a Single Injection of Rexlemestrocel-L + HA Results in More than Three Years of Pain Reduction Greatest pain reduction was observed in the pre-specified population of subjects with CLBP duration shorter than the baseline study median of 68 months (n=202) with significantly greater reduction (nominal p-value < 0.05) at all time points analyzed over 36 months compared with saline controls VAS=Visual Analog Score; HA=Hyaluronic Acid
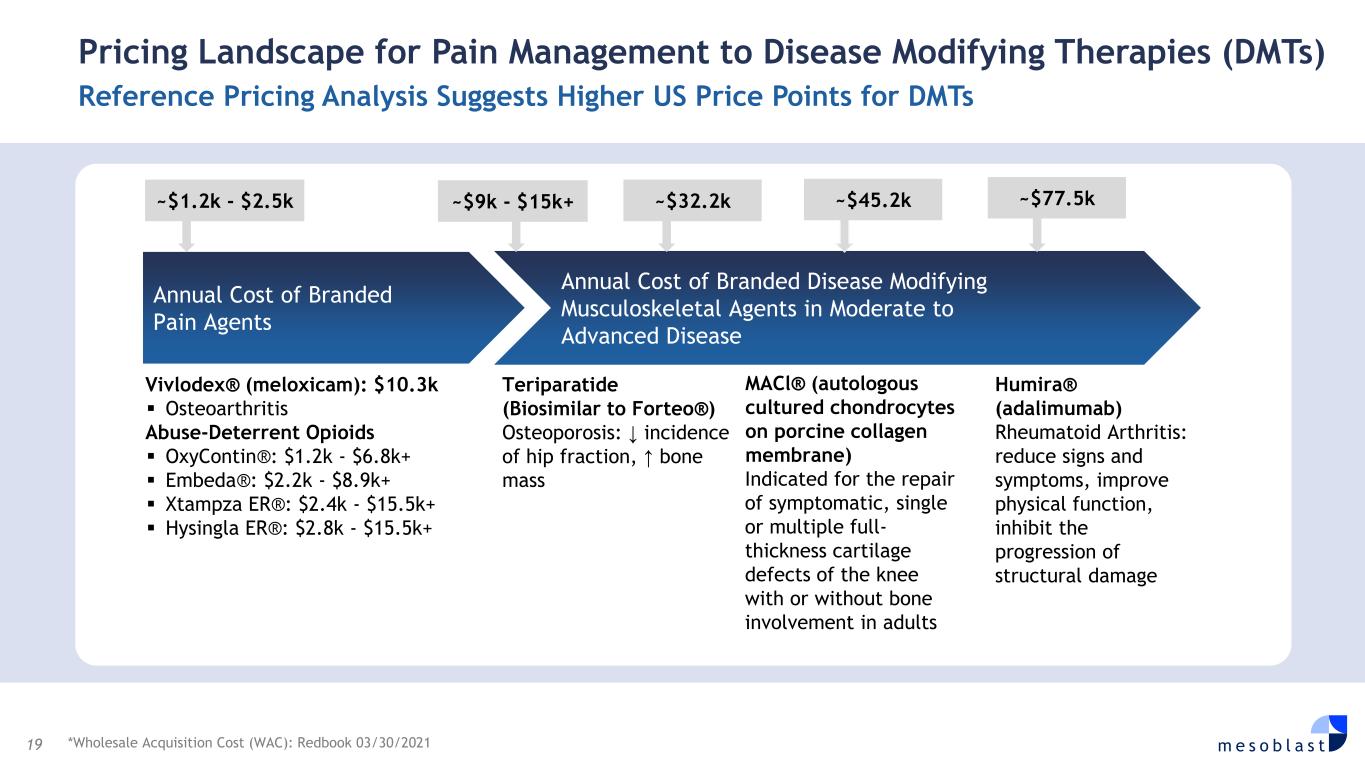
19 m e s o b l a s t Pricing Landscape for Pain Management to Disease Modifying Therapies (DMTs) Reference Pricing Analysis Suggests Higher US Price Points for DMTs *Wholesale Acquisition Cost (WAC): Redbook 03/30/2021 Annual Cost of Branded Pain Agents Annual Cost of Branded Disease Modifying Musculoskeletal Agents in Moderate to Advanced Disease ~$9k - $15k+ ~$32.2k ~$45.2k ~$77.5k~$1.2k - $2.5k Vivlodex® (meloxicam): $10.3k Osteoarthritis Abuse-Deterrent Opioids OxyContin®: $1.2k - $6.8k+ Embeda®: $2.2k - $8.9k+ Xtampza ER®: $2.4k - $15.5k+ Hysingla ER®: $2.8k - $15.5k+ Teriparatide (Biosimilar to Forteo®) Osteoporosis: ↓ incidence of hip fraction, ↑ bone mass MACl® (autologous cultured chondrocytes on porcine collagen membrane) Indicated for the repair of symptomatic, single or multiple full- thickness cartilage defects of the knee with or without bone involvement in adults Humira® (adalimumab) Rheumatoid Arthritis: reduce signs and symptoms, improve physical function, inhibit the progression of structural damage
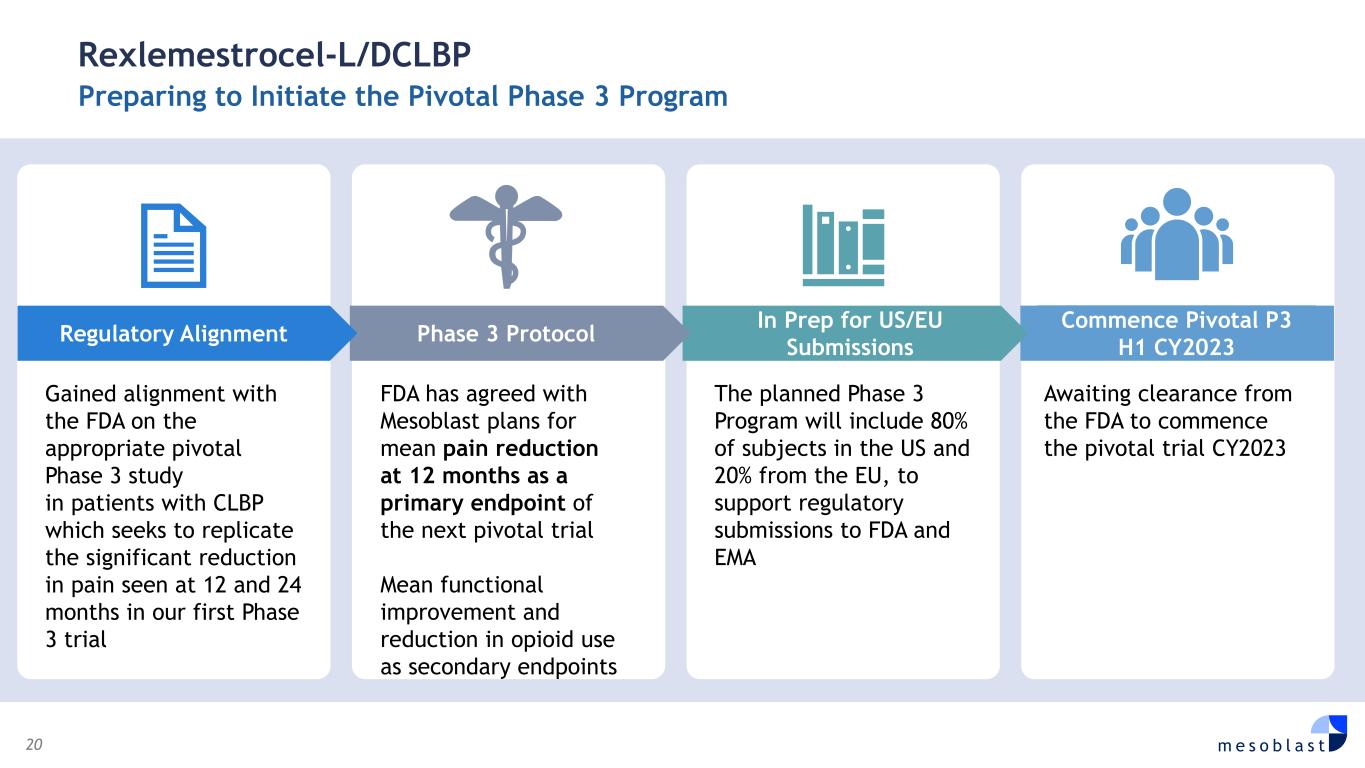
20 m e s o b l a s t Rexlemestrocel-L/DCLBP Preparing to Initiate the Pivotal Phase 3 Program Gained alignment with the FDA on the appropriate pivotal Phase 3 study in patients with CLBP which seeks to replicate the significant reduction in pain seen at 12 and 24 months in our first Phase 3 trial FDA has agreed with Mesoblast plans for mean pain reduction at 12 months as a primary endpoint of the next pivotal trial Mean functional improvement and reduction in opioid use as secondary endpoints The planned Phase 3 Program will include 80% of subjects in the US and 20% from the EU, to support regulatory submissions to FDA and EMA Awaiting clearance from the FDA to commence the pivotal trial CY2023 Commence Pivotal P3 H1 CY2023 In Prep for US/EU SubmissionsPhase 3 Protocol Regulatory Alignment

21 m e s o b l a s t Rexlemestrocel-L Chronic Heart Failure Reduced Ejection Fraction (HFrEF) Tracking to a potential H1 CY2023 FDA meeting to map out pivotal trial plans and regulatory pathway

22 m e s o b l a s t Chronic Heart Failure (CHF): Rising Incidence and High Mortality New therapies reduce recurrent hospitalization but do not materially improve mortality or major ischemic event rates Cardiovascular disease (CVD) remains the leading cause of death in the United States1 Heart failure affects 6.5 million patients in the US and 26 million patients globally. As populations age, the prevalence is increasing2 Chronic heart failure is a progressive disease with a high mortality that approaches 50% at 5 years2,3 and at least 75% after an initial hospitalization4 Patients with heart failure are also at high risk of recurrent major adverse cardiac events involving large vessels (heart attacks / strokes) 1. Muntner BEJ, et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. Feb 19, 2019. 2. United States Food & Drug Administration. Treatment for Heart Failure: Endpoints for Drug Development. Draft Guidance. June 2019. 3. Taylor CJ, et al. Trends in survival after a diagnosis of heart failure in the United Kingdom 2000-2017: population based cohort study. BMJ. 2019;364:I223. 4. Shah KS, et al. Heart Failure with Preserve, Borderline, and Reduced Ejection Fraction; 5-Year Outcomes. JACC. 2017;Nov12.
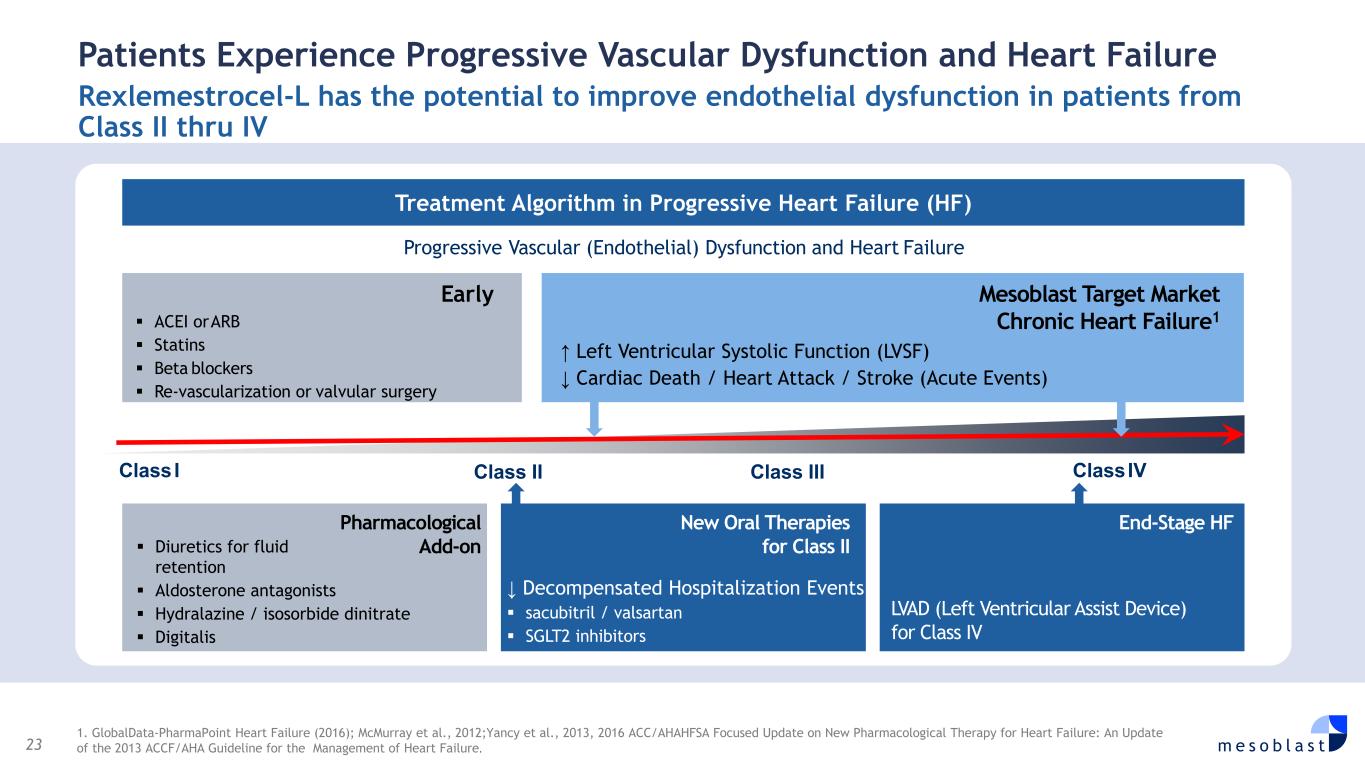
23 m e s o b l a s t Patients Experience Progressive Vascular Dysfunction and Heart Failure Rexlemestrocel-L has the potential to improve endothelial dysfunction in patients from Class II thru IV 1. GlobalData-PharmaPoint Heart Failure (2016); McMurray et al., 2012;Yancy et al., 2013, 2016 ACC/AHAHFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Treatment Algorithm in Progressive Heart Failure (HF) Progressive Vascular (Endothelial) Dysfunction and Heart Failure Early ACEI orARB Statins Beta blockers Re-vascularization or valvular surgery Mesoblast Target Market Chronic Heart Failure1 ↑ Left Ventricular Systolic Function (LVSF) ↓ Cardiac Death / Heart Attack / Stroke (Acute Events) Pharmacological Add-on Diuretics for fluid retention Aldosterone antagonists Hydralazine / isosorbide dinitrate Digitalis New Oral Therapies for Class II ↓ Decompensated Hospitalization Events sacubitril / valsartan SGLT2 inhibitors End-Stage HF LVAD (Left Ventricular Assist Device) for Class IV ClassI ClassIVClass II Class III

24 m e s o b l a s t Rexlemestrocel-L: DREAM Phase 3 Trial in Heart Failure (HFrEF) Improvement in Left Ventricular Systolic Function, as Measured by Left Ventricular Ejection Fraction (LVEF) at 12 Months represents a potential early surrogate endpoint In all treated patients (n=537), rexlemestrocel-L resulted in a 52% greater increase in LVEF from baseline to 12 months compared with controls While both groups had similar LVEF at baseline (28.7% and 28.6%), at 12 months, the least squared mean change from baseline was 5.0 for the rexlemestrocel-L group and 3.3 for controls (p=0.021) In treated patients with CRP >2 (n=301), rexlemestrocel-L resulted in 86% greater increase in LVEF from baseline to 12 months compared with controls While both groups had similar LVEF at baseline (29.1% and 28.2%), at 12 months, the least squared mean change from baseline was 5.6 for the rexlemestrocel-L group and 2.9 for controls (p=0.005) LVEF, 2-point MACE, and recurrent hospitalizations due to heart attack or stroke were pre-specified endpoints and the 3-point MACE was a post-hoc analysis of pre-specified endpoint components

25 m e s o b l a s t Days from Day 0 Treatment REX 29/153 (19.0%) vs CTRL 48/148 (32.4%) HR 0.551 95% CI (0.346, 0.876) HR p-value = 0.012 Risk ↓due to REX = 45% Ab se nc e of C ar di ac D ea th o r N on -fa ta l M I o r N on -fa ta l s tr ok e DREAM-HF Phase 3 Trial in HFrEF Rexlemestrocel-L reduced incidence of 3-Point Composite MACE v. controls across all treated patients, with enhanced effect in those with active inflammation as measured by CRP >2 MACE=Major Adverse Cardiovascular Event; 3-Point Composite MACE=Cardiovascular Death, Non-Fatal MI or Non-Fatal Stroke; TTFE=Time To First Event; MI=Myocardial Infarction (Heart Attack); hsCRP=High Sensitivity C-reactive Protein (a measure of systemic inflammation) LVEF, 2-point MACE, and recurrent hospitalizations due to heart attack or stroke were pre-specified endpoints and the 3-point MACE was a post-hoc analysis of pre-specified endpoint components All Treated Patients (n=537) All Treated Patients with hsCRP ≥2 (n=301) TTFE for 3-Point Composite MACE TTFE for 3-Point Composite MACE Kaplan-Meier log rank statistics
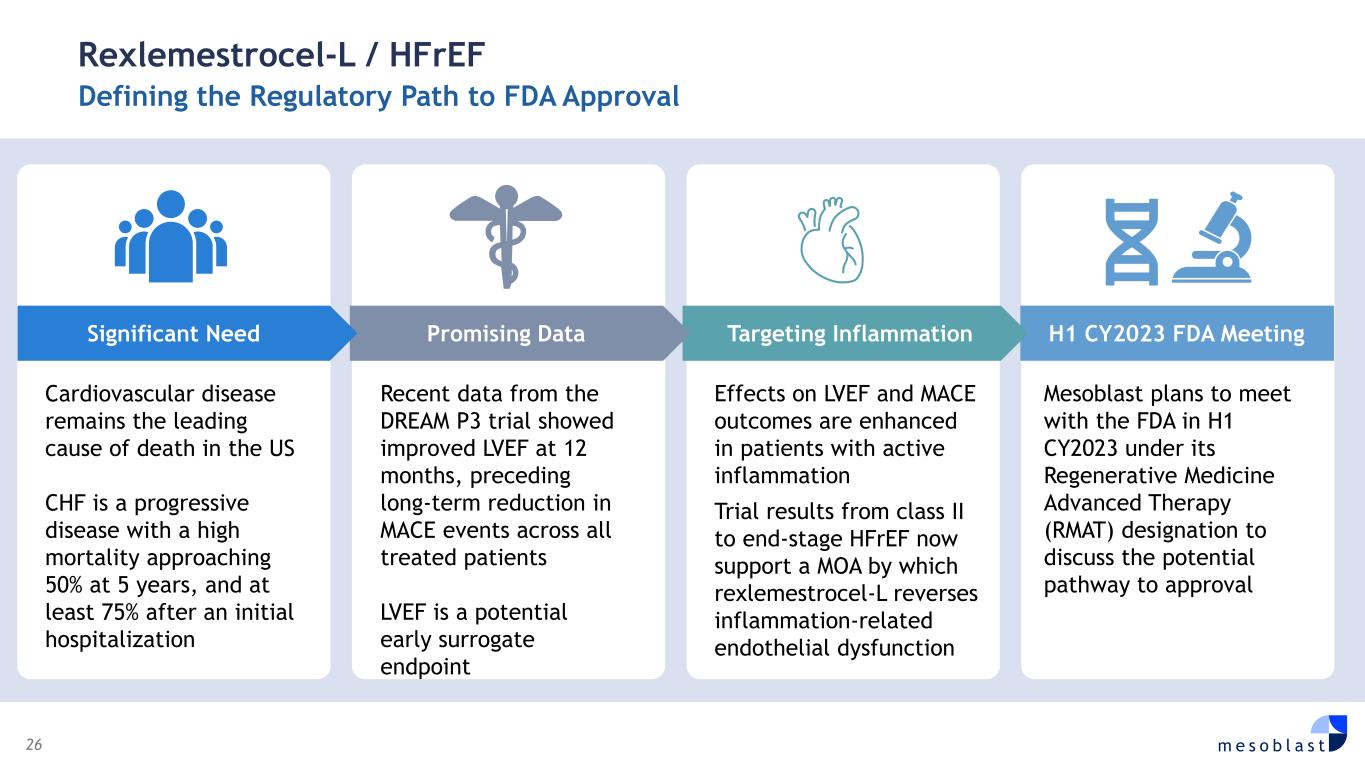
26 m e s o b l a s t Rexlemestrocel-L / HFrEF Defining the Regulatory Path to FDA Approval Cardiovascular disease remains the leading cause of death in the US CHF is a progressive disease with a high mortality approaching 50% at 5 years, and at least 75% after an initial hospitalization Recent data from the DREAM P3 trial showed improved LVEF at 12 months, preceding long-term reduction in MACE events across all treated patients LVEF is a potential early surrogate endpoint Effects on LVEF and MACE outcomes are enhanced in patients with active inflammation Trial results from class II to end-stage HFrEF now support a MOA by which rexlemestrocel-L reverses inflammation-related endothelial dysfunction Mesoblast plans to meet with the FDA in H1 CY2023 under its Regenerative Medicine Advanced Therapy (RMAT) designation to discuss the potential pathway to approval H1 CY2023 FDA Meeting Targeting Inflammation Promising DataSignificant Need
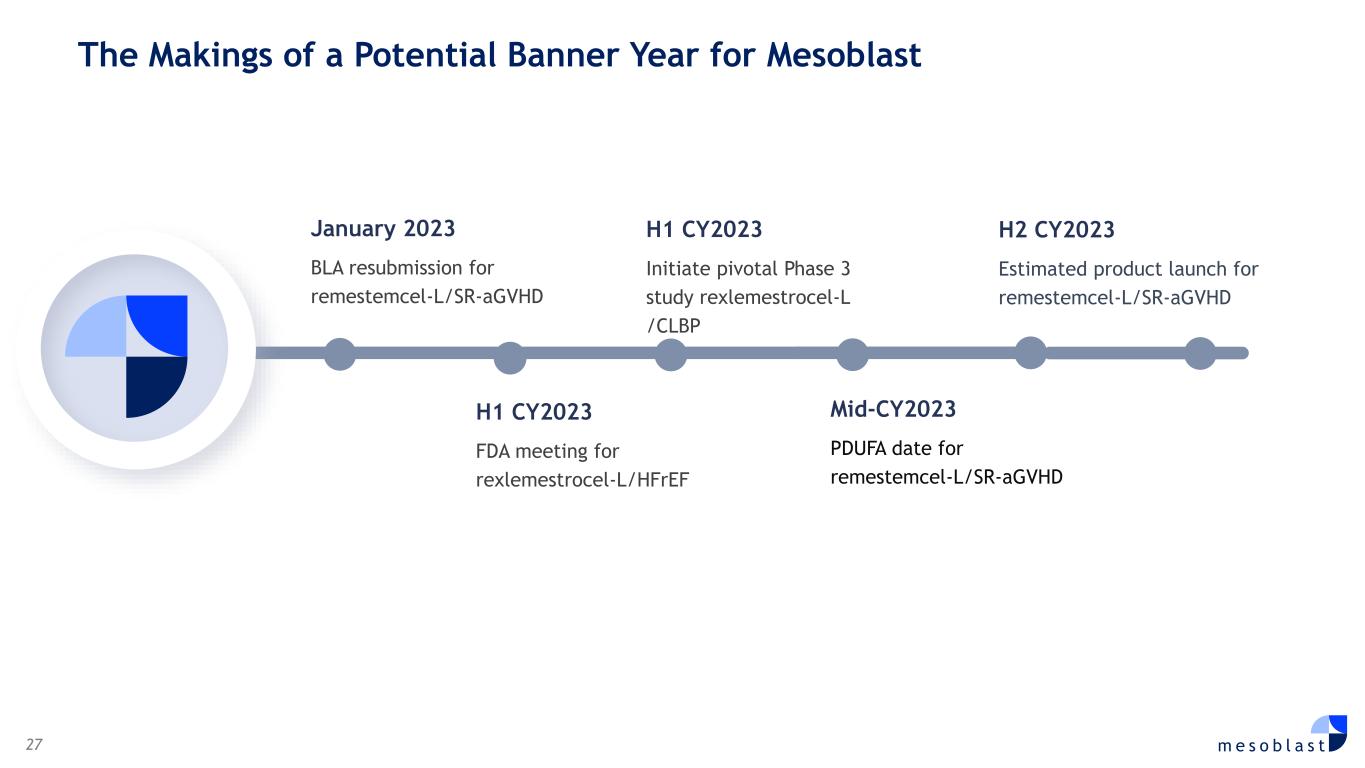
27 m e s o b l a s t H1 CY2023 FDA meeting for rexlemestrocel-L/HFrEF H2 CY2023 Estimated product launch for remestemcel-L/SR-aGVHD Mid-CY2023 PDUFA date for remestemcel-L/SR-aGVHD The Makings of a Potential Banner Year for Mesoblast H1 CY2023 Initiate pivotal Phase 3 study rexlemestrocel-L /CLBP January 2023 BLA resubmission for remestemcel-L/SR-aGVHD

28 m e s o b l a s t mesoblast Thank You