
ASX: MSB; Nasdaq: MESO Global Leader in Allogeneic Cellular Medicines for Inflammatory Diseases Financial Year Ended June 30, 2020 Exhibit 99.2

CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS This presentation includes forward-looking statements that relate to future events or our future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, levels of activity, performance or achievements to differ materially from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. We make such forward-looking statements pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and other federal securities laws. All statements other than statements of historical facts contained in this presentation are forward-looking statements. Words such as, but not limited to, “believe,” “expect,” “anticipate,” “estimate,” “intend,” “plan,” “targets,” “likely,” “will,” “would,” “could,” and similar expressions or phrases identify forward-looking statements. We have based these forward-looking statements largely on our current expectations and future events , recent changes in regulatory laws, and financial trends that we believe may affect our financial condition, results of operation, business strategy and financial needs. These statements may relate to, but are not limited to: expectations regarding the safety or efficacy of, or potential applications for, Mesoblast's adult stem cell technologies; expectations regarding the strength of Mesoblast's intellectual property, the timeline for Mesoblast's regulatory approval process, and the scalability and efficiency of manufacturing processes; expectations about Mesoblast's ability to grow its business and statements regarding its relationships with current and potential future business partners and future benefits of those relationships; statements concerning Mesoblast's share price or potential market capitalization; and statements concerning Mesoblast's capital requirements and ability to raise future capital, among others. Forward-looking statements should not be read as a guarantee of future performance or results, and actual results may differ from the results anticipated in these forward-looking statements, and the differences may be material and adverse. You should read this presentation together with our financial statements and the notes related thereto, as well as the risk factors, in our most recently filed reports with the SEC or on our website. Uncertainties and risks that may cause Mesoblast's actual results, performance or achievements to be materially different from those which may be expressed or implied by such statements, include, without limitation: risks inherent in the development and commercialization of potential products; uncertainty of clinical trial results or regulatory approvals or clearances; government regulation; the need for future capital; dependence upon collaborators; and protection of our intellectual property rights, among others. Accordingly, you should not place undue reliance on these forward-looking statements. We do not undertake any obligations to publicly update or revise any forward-looking statements, whether as a result of new information, future developments or otherwise. |

Our Mission Mesoblast is committed to bringing to market innovative cellular medicines to treat serious and life-threatening illnesses |
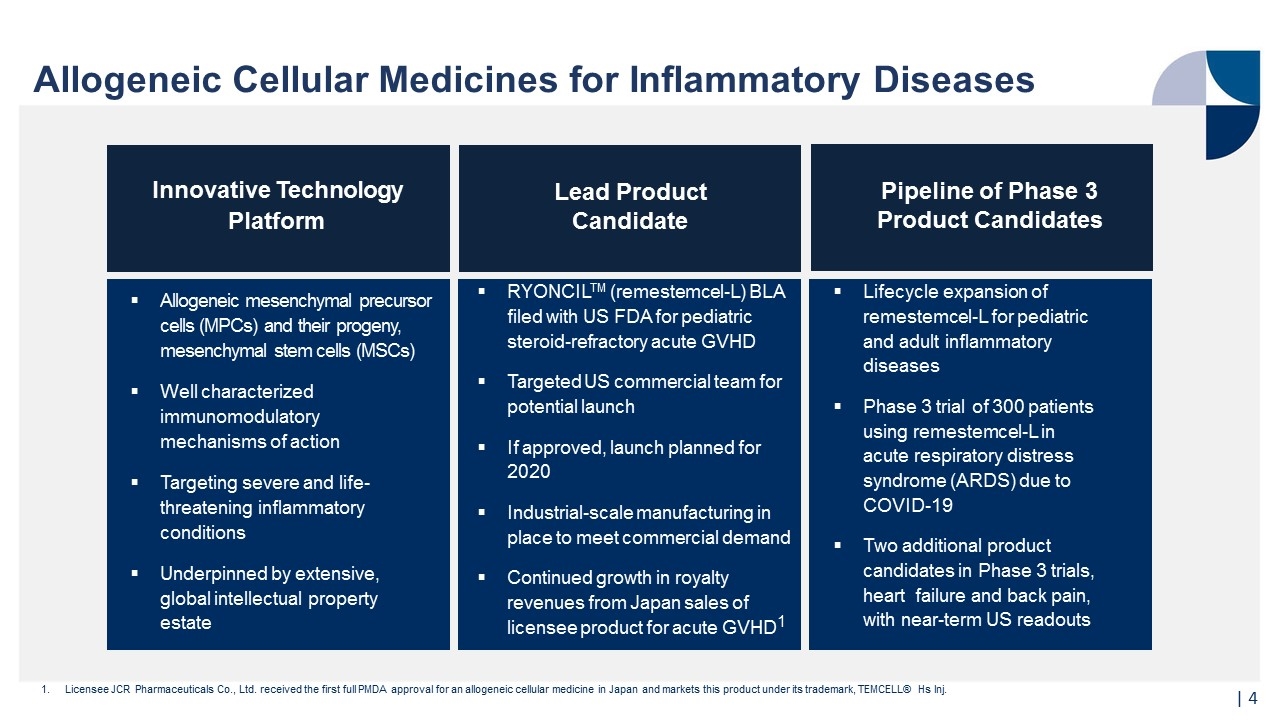
Allogeneic Cellular Medicines for Inflammatory Diseases Pipeline of Phase 3 Product Candidates Innovative Technology Platform Allogeneic mesenchymal precursor cells (MPCs) and their progeny, mesenchymal stem cells (MSCs) Well characterized immunomodulatory mechanisms of action Targeting severe and life- threatening inflammatory conditions Underpinned by extensive, global intellectual property estate Licensee JCR Pharmaceuticals Co., Ltd. received the first full PMDA approval for an allogeneic cellular medicine in Japan and markets this product under its trademark, TEMCELL® Hs Inj. Lead Product Candidate Lifecycle expansion of remestemcel-L for pediatric and adult inflammatory diseases Phase 3 trial of 300 patients using remestemcel-L in acute respiratory distress syndrome (ARDS) due to COVID-19 Two additional product candidates in Phase 3 trials, heart failure and back pain, with near-term US readouts RYONCILTM (remestemcel-L) BLA filed with US FDA for pediatric steroid-refractory acute GVHD Targeted US commercial team for potential launch If approved, launch planned for 2020 Industrial-scale manufacturing in place to meet commercial demand Continued growth in royalty revenues from Japan sales of licensee product for acute GVHD1 |
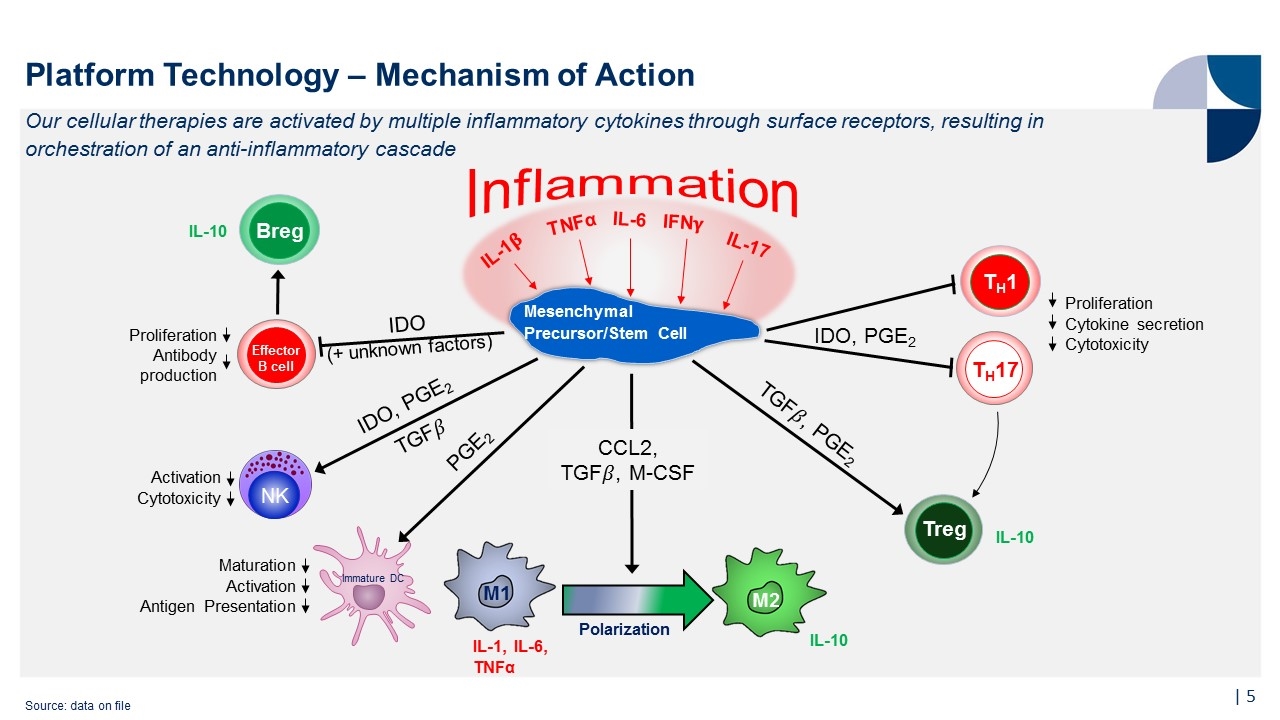
Platform Technology – Mechanism of Action | Our cellular therapies are activated by multiple inflammatory cytokines through surface receptors, resulting in orchestration of an anti-inflammatory cascade Source: data on file Effector B cell IL-1�� TNFα IFN�� IL-17 M2 Polarization Breg Treg Inflammation IDO (+ unknown factors) IDO, PGE2 TGF�� NK Activation Cytotoxicity PGE2 Maturation Activation Antigen Presentation Proliferation Antibody production CCL2, TGF��, M-CSF IL-10 TH17 Proliferation Cytokine secretion Cytotoxicity IDO, PGE2 TGF��, PGE2 IL-10 TH1 M1 Immature DC IL-1, IL-6, TNFα Mesenchymal Precursor/Stem Cell IL-10 IL-6
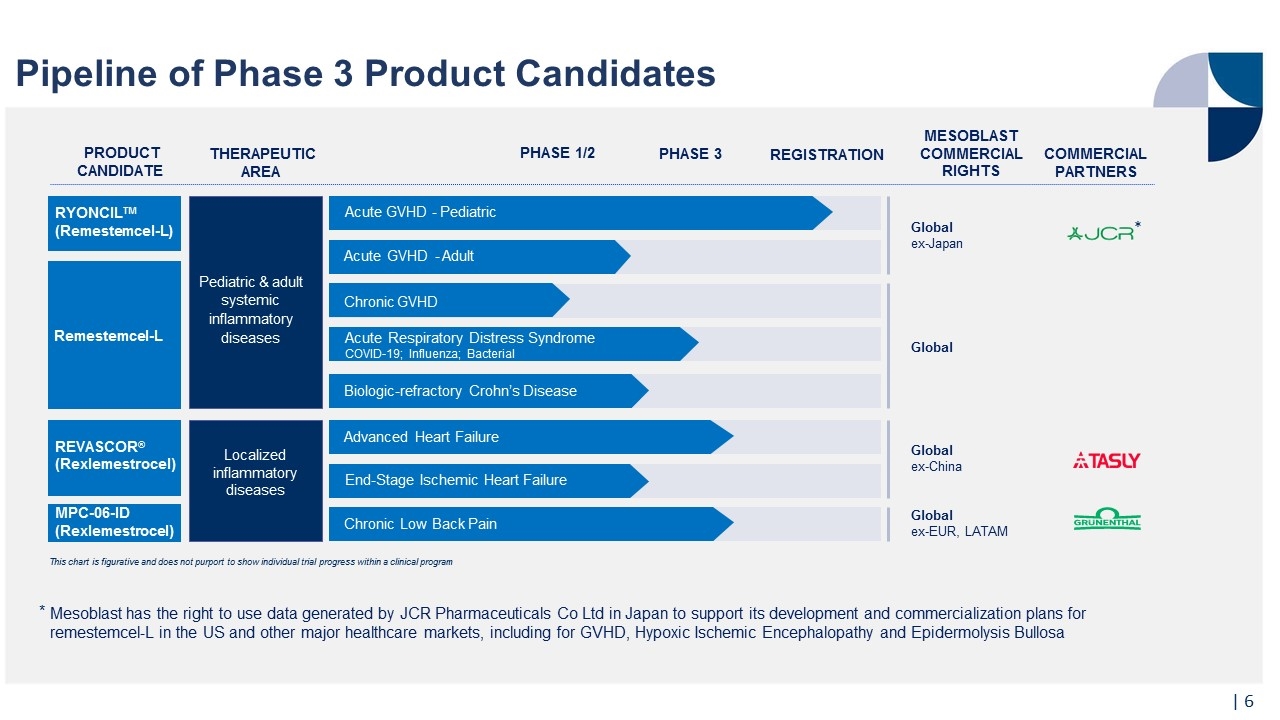
Pipeline of Phase 3 Product Candidates This chart is figurative and does not purport to show individual trial progress within a clinical program Mesoblast has the right to use data generated by JCR Pharmaceuticals Co Ltd in Japan to support its development and commercialization plans for remestemcel-L in the US and other major healthcare markets, including for GVHD, Hypoxic Ischemic Encephalopathy and Epidermolysis Bullosa | Remestemcel-L Chronic GVHD Acute GVHD - Adult Pediatric & adult systemic inflammatory diseases REVASCOR® (Rexlemestrocel) Localized inflammatory diseases Advanced Heart Failure Chronic Low Back Pain End-Stage Ischemic Heart Failure MPC-06-ID (Rexlemestrocel) PRODUCT CANDIDATE THERAPEUTIC AREA PHASE 1/2 PHASE 3 REGISTRATION MESOBLAST COMMERCIAL RIGHTS Global ex-Japan Global ex-China Global ex-EUR, LATAM RYONCILTM (Remestemcel-L) Acute GVHD - Pediatric COMMERCIAL PARTNERS Acute Respiratory Distress Syndrome COVID-19; Influenza; Bacterial Global Biologic-refractory Crohn’s Disease *

Scalable allogeneic “off-the-shelf” cellular platforms Manufacturing meets stringent criteria of international regulatory agencies Robust quality assurance processes ensure final product with batch-to-batch consistency and reproducibility Current capacity sufficient for RYONCIL GVHD launch Commercial Scale Manufacturing Capability | Projected increase in capacity requirements for maturing pipeline, including GVHD label extensions and COVID-19 ARDS Proprietary xeno-free technologies will increase yields and output Moving to 3D bioreactors will reduce labor and improve manufacturing efficiencies These innovations will significantly reduce cost of goods Manufacturing Remestemcel-L © Lonza, reproduced with permission
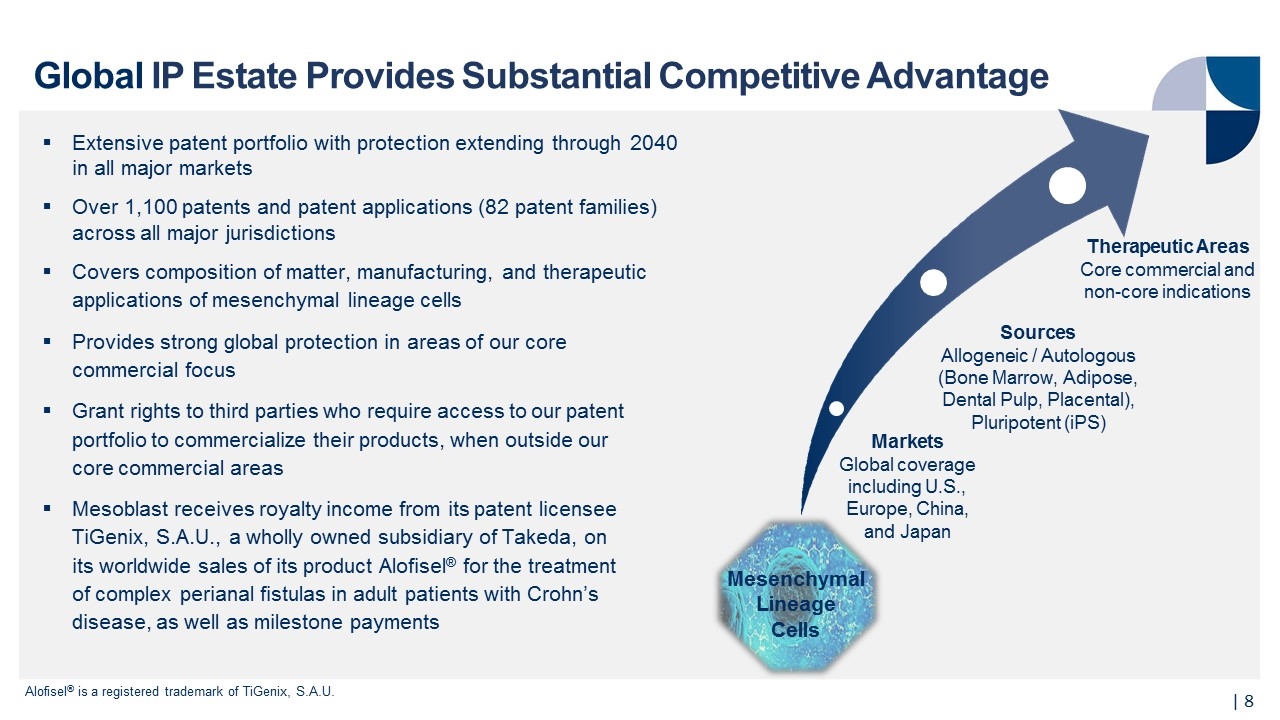
Extensive patent portfolio with protection extending through 2040 in all major markets Over 1,100 patents and patent applications (82 patent families) across all major jurisdictions Covers composition of matter, manufacturing, and therapeutic applications of mesenchymal lineage cells Provides strong global protection in areas of our core commercial focus Grant rights to third parties who require access to our patent portfolio to commercialize their products, when outside our core commercial areas Mesoblast receives royalty income from its patent licensee TiGenix, S.A.U., a wholly owned subsidiary of Takeda, on its worldwide sales of its product Alofisel® for the treatment of complex perianal fistulas in adult patients with Crohn’s disease, as well as milestone payments Mesenchymal Lineage Cells Global IP Estate Provides Substantial Competitive Advantage | Alofisel® is a registered trademark of TiGenix, S.A.U. Sources Allogeneic / Autologous (Bone Marrow, Adipose, Dental Pulp, Placental), Pluripotent (iPS) Therapeutic Areas Core commercial and non-core indications Markets Global coverage including U.S., Europe, China, and Japan

Recent Highlights: Remestemcel-L for Children | Children with Steroid-Refractory Acute Graft Versus Host Disease (SR-aGVHD) FDA Oncologic Drugs Advisory Committee (ODAC) of the FDA voted overwhelmingly (9-1*) in favor that the available data support the efficacy of RYONCIL in pediatric patients with SR-aGVHD The Biologics License Application (BLA) for RYONCIL is under Priority Review by the FDA with an action date of September 30, 2020, under the Prescription Drug User Fee Act (PDUFA) If approved by the PDUFA date, Mesoblast plans RYONCIL launch in US in Q4 2020 in children and adolescents up to 18 years old Preparations advanced for potential launch, with product inventory and commercial team in place Children with COVID-19 Multisystem Inflammatory Syndrome (MIS-C) Established an expanded access protocol (EAP) in the US for compassionate use of remestemcel-L in the treatment of COVID-19 infected children with cardiovascular and other complications of MIS-C The first patient has received treatment under the EAP and has been discharged from the hospital * This vote includes a change to the original vote by one of the ODAC panel members after electronic voting closed

Recent Highlights: Remestemcel-L for Adults | Adults With Acute Respiratory Distress Syndrome (ARDS) due to COVID-19 FDA cleared IND application to treat patients with COVID-19 ARDS, providing a pathway for use under both EAP and in a randomized controlled trial Under emergency compassionate care at Mt Sinai Hospital in New York, 75% of patients (9/12) with moderate to severe ARDS who received two infusions of remestemcel-L were successfully taken off a ventilator and discharged from hospital within a median of 10 days Ongoing Phase 3 randomized, placebo-controlled trial of remestemcel-L in up to 300 ventilator-dependent patients with moderate to severe COVID-19 ARDS, objective is reduction in mortality within 30 days Trial expected to complete recruitment during Q4 CY2020 Independent DSMB has set a review date of early September for first interim analysis of the Phase 3 trial from the first 90 patients after they have all completed 30 days of follow up Adults With Steroid-Refractory Acute Graft Versus Host Disease (SR-aGVHD) Patients with most severe forms of SR-aGVHD continue to have high unmet need and poor survival August 2020 convened advisory meeting with key opinion leaders to develop clinical trial design for post-market study evaluating remestemcel-L in these high-risk patients

Financials

Financial Highlights | Full Year FY2020 Compared to Full Year FY2019 92% increase in total revenue to US$32.2m 127% increase in milestone revenues from strategic partnerships to US$25.0m 32% growth in commercialization revenue from sales of TEMCELL to US$6.6m 13% (US$11.9m) reduction in loss after tax, even after US$13.8 million increased investment in commercial readiness for potential US launch of RYONCIL Total reduction in clinical trial expenditure of US$8.6m, inclusive of new Phase 3 COVID-19 ARDS trial Figures are rounded TEMCELL® Hs. Inj. is a registered trademark of JCR Pharmaceuticals Co Ltd.
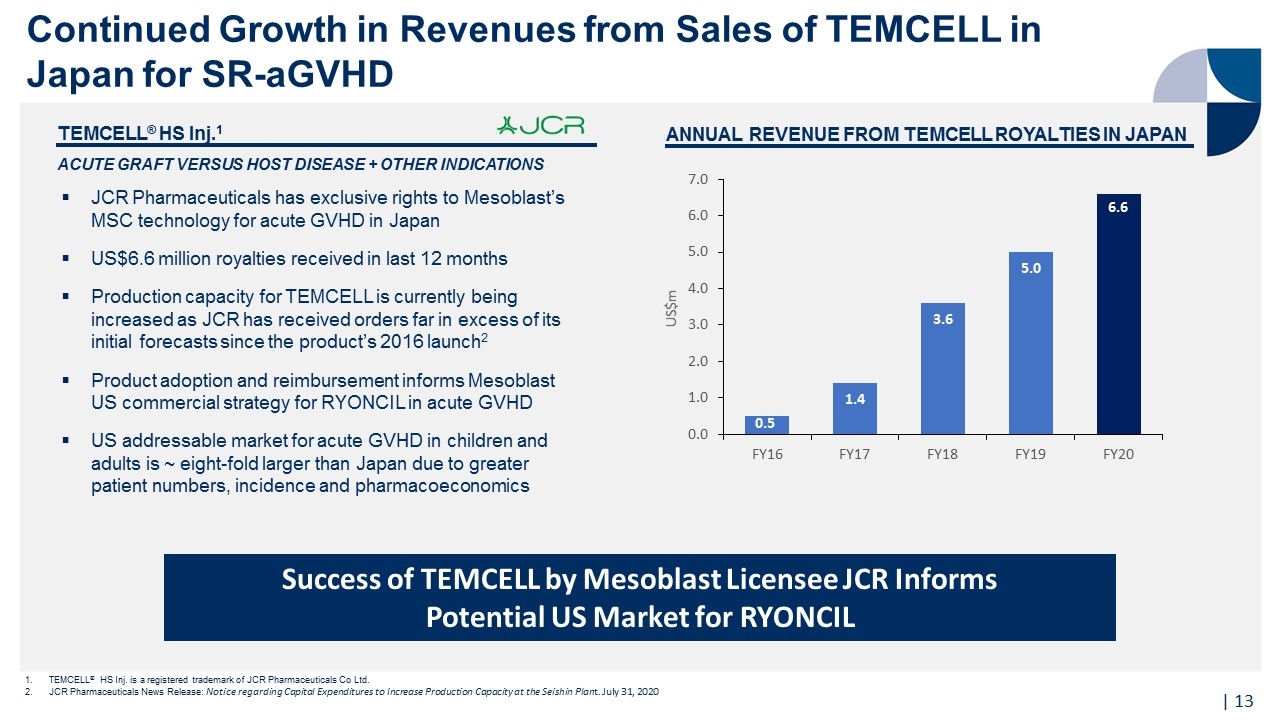
| TEMCELL® HS Inj.1 ACUTE GRAFT VERSUS HOST DISEASE + OTHER INDICATIONS JCR Pharmaceuticals has exclusive rights to Mesoblast’s MSC technology for acute GVHD in Japan US$6.6 million royalties received in last 12 months Production capacity for TEMCELL is currently being increased as JCR has received orders far in excess of its initial forecasts since the product’s 2016 launch2 Product adoption and reimbursement informs Mesoblast US commercial strategy for RYONCIL in acute GVHD US addressable market for acute GVHD in children and adults is ~ eight-fold larger than Japan due to greater patient numbers, incidence and pharmacoeconomics ANNUAL REVENUE FROM TEMCELL ROYALTIES IN JAPAN TEMCELL® HS Inj. is a registered trademark of JCR Pharmaceuticals Co Ltd. JCR Pharmaceuticals News Release: Notice regarding Capital Expenditures to Increase Production Capacity at the Seishin Plant. July 31, 2020 Success of TEMCELL by Mesoblast Licensee JCR Informs Potential US Market for RYONCIL Continued Growth in Revenues from Sales of TEMCELL in Japan for SR-aGVHD
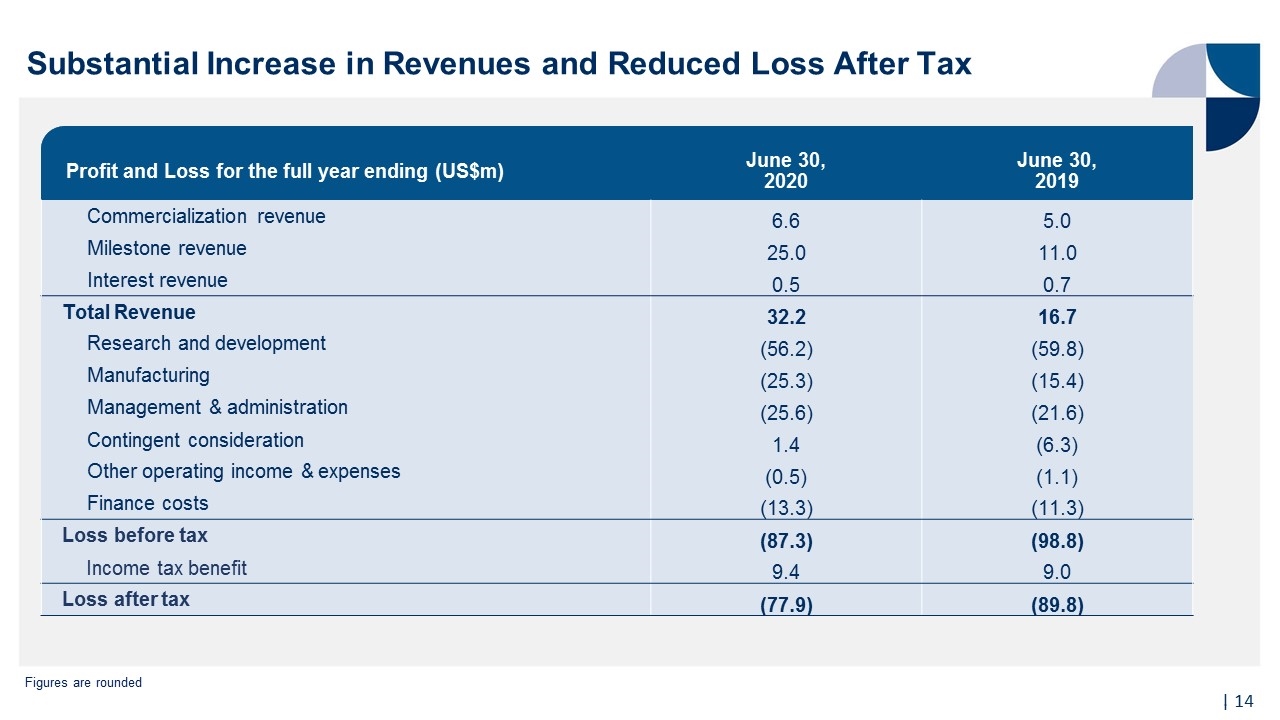
| 11 Substantial Increase in Revenues and Reduced Loss After Tax | Figures are rounded Profit and Loss for the full year ending (US$m) June 30, 2020 June 30, 2019 Commercialization revenue 6.6 5.0 Milestone revenue 25.0 11.0 Interest revenue 0.5 0.7 Total Revenue 32.2 16.7 Research and development (56.2) (59.8) Manufacturing (25.3) (15.4) Management & administration (25.6) (21.6) Contingent consideration 1.4 (6.3) Other operating income & expenses (0.5) (1.1) Finance costs (13.3) (11.3) Loss before tax (87.3) (98.8) Income tax benefit 9.4 9.0 Loss after tax (77.9) (89.8)

Strengthened Balance Sheet | At June 30, 2020, cash on hand was US$129.3 million Mesoblast completed a US$90 million capital raise from global institutional investors in May 2020 Up to an additional US$67.5 million may be available through existing financing facilities and strategic partnerships over next 12 months Capital will be used for Commercial launch of RYONCIL for acute GVHD Scale-up of manufacturing for projected increase in capacity requirements for maturing pipeline, including GVHD label extensions and COVID-19 ARDS Clinical programs supporting label extension strategies and regulatory approvals of Phase 3 assets Figures are rounded
RYONCIL (remestemcel-L): Acute Graft Versus Host Disease
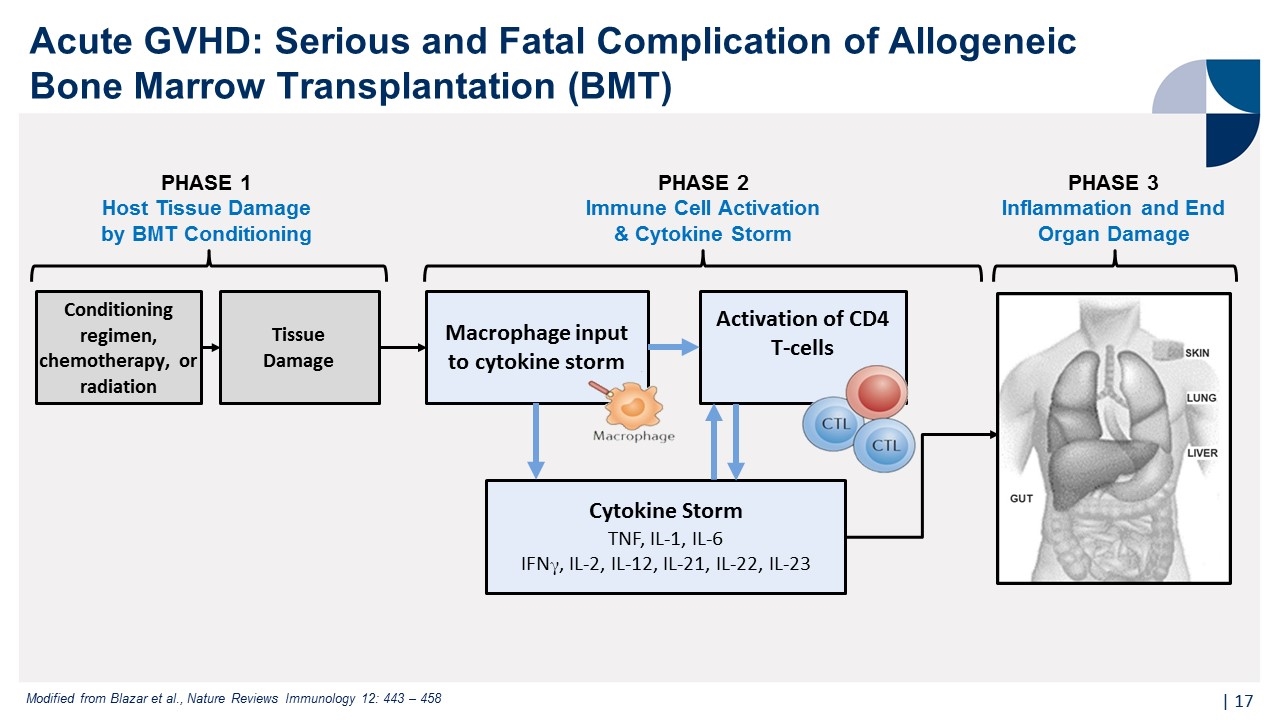
Acute GVHD: Serious and Fatal Complication of Allogeneic Bone Marrow Transplantation (BMT) | PHASE 1 Host Tissue Damage by BMT Conditioning PHASE 2 Immune Cell Activation & Cytokine Storm PHASE 3 Inflammation and End Organ Damage Conditioning regimen, chemotherapy, or radiation Tissue Damage Activation of CD4 T-cells Cytokine Storm TNF, IL-1, IL-6 IFNγ, IL-2, IL-12, IL-21, IL-22, IL-23 Macrophage input to cytokine storm Modified from Blazar et al., Nature Reviews Immunology 12: 443 – 458

Children with Steroid-Refractory Acute GVHD at High Risk of Treatment Failure and Death | Extremely high unmet medical need More than 2,000 allogeneic BMTs in children and adolescents in US annually1 Despite prophylaxis, ~50% will develop aGVHD2 First-line treatment is corticosteroids Response rate is ~50% Children < 12 years of age have no approved treatment for steroid-refractory acute GVHD Acute GVHD Primarily Affects Skin, GI Tract, and Liver Classic skin rash; Abdominal cramps; Large volumes of diarrhea Rising serum bilirubin (indicative of liver damage or disease) Mortality as high as 70 – 90%2-5 when involving gut and liver 1. HRSA Transplant Activity Report, CIBMTR, 2019; 2. Westin, J., Saliba, RM., Lima, M. (2011) Steroid-refractory acute GVHD: predictors and outcomes. Advances in Hematology; 3. MacMillan, M.L. et al. Pediatric acute GVHD: clinical phenotype and response to upfront steroids. Bone Marrow Transplant 55, 165–171 (2020); 4. Jagasia, M. et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood (2012) 119 (1): 296-307; 5. Axt L, Naumann A, Toennies J (2019) Retrospective single center analysis of outcome, risk factors and therapy in steroid refractory graft-versus-host disease after allogeneic hematopoietic cell transplantation. Bone Marrow Transplantation © J Kurtzberg MD, reproduced with permission
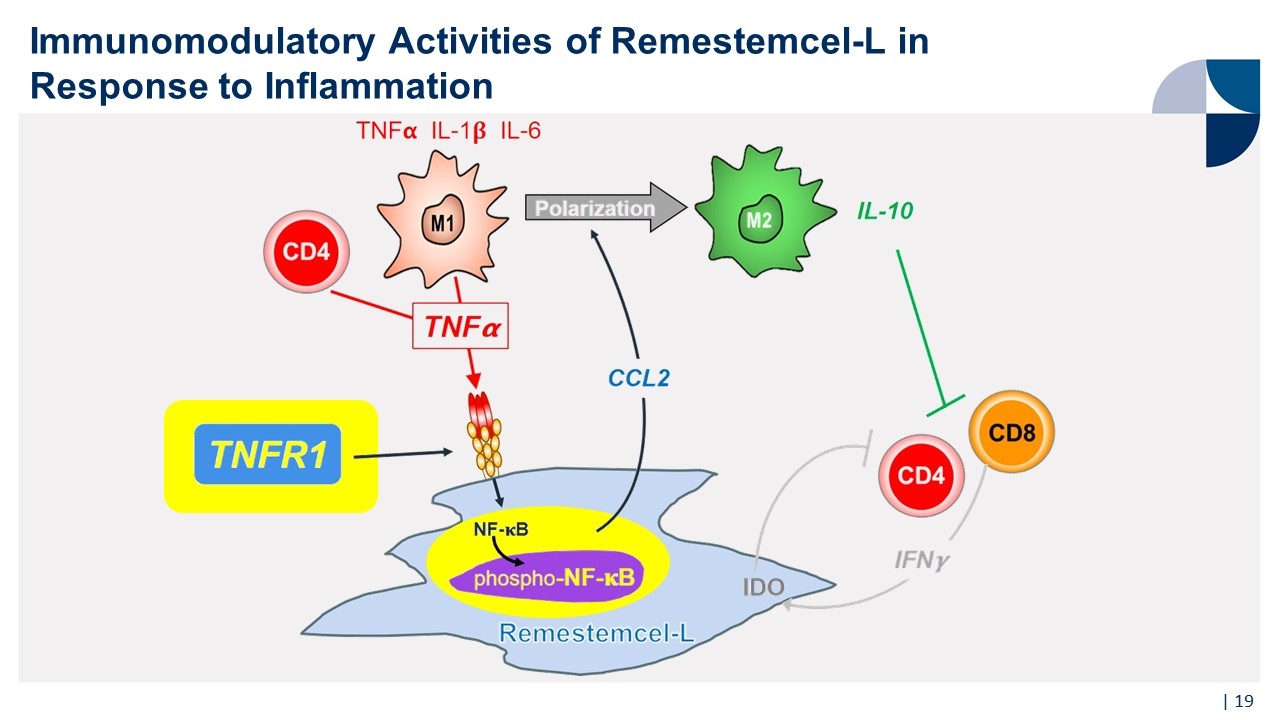
Immunomodulatory Activities of Remestemcel-L in Response to Inflammation |
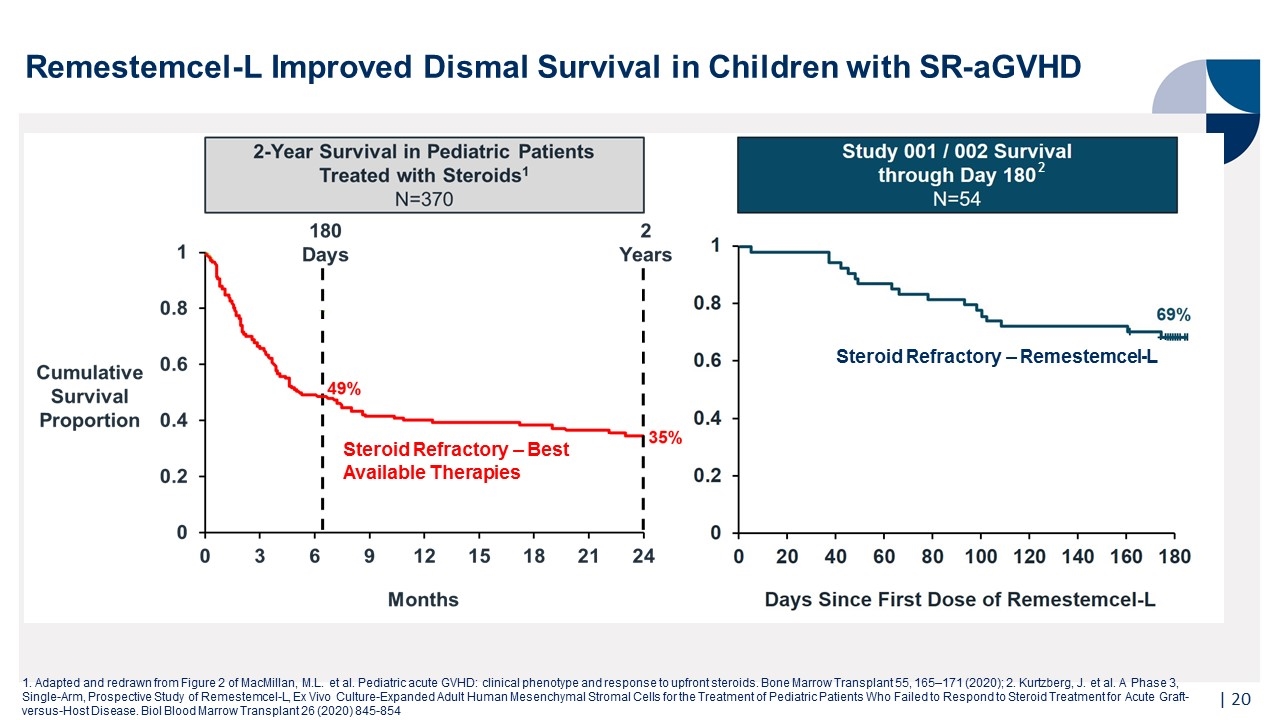
Remestemcel-L Improved Dismal Survival in Children with SR-aGVHD | 1. Adapted and redrawn from Figure 2 of MacMillan, M.L. et al. Pediatric acute GVHD: clinical phenotype and response to upfront steroids. Bone Marrow Transplant 55, 165–171 (2020); 2. Kurtzberg, J. et al. A Phase 3, Single-Arm, Prospective Study of Remestemcel-L, Ex Vivo Culture-Expanded Adult Human Mesenchymal Stromal Cells for the Treatment of Pediatric Patients Who Failed to Respond to Steroid Treatment for Acute Graft-versus-Host Disease. Biol Blood Marrow Transplant 26 (2020) 845-854 Steroid Refractory – Best Available Therapies Steroid Refractory – Remestemcel-L 2

RYONCIL: Anticipated FDA Approval in 2020 Results from three studies using RYONCIL in children and adults with SR-aGVHD support the FDA BLA filing RYONCIL was used as salvage therapy in an expanded access program in 241 children with SR-aGVHD (80% Grade C/D) who failed institutional standard of care RYONCIL was used as first-line therapy in Mesoblast’s open-label Phase 3 trial in 55 children with SR-aGVHD, 89% of whom had Grade C/D disease RYONCIL was used as first-line therapy in a randomized controlled Phase 3 trial of 260 adults and children with SR-aGVHD BLA filing for RYONCIL was accepted by the US FDA for priority review for the treatment of SR-aGVHD in children ODAC of the FDA voted overwhelmingly in favour (9:1)* that the available data support the efficacy of remestemcel-L in pediatric patients with SR-aGVHD The FDA has set a Prescription Drug User Fee Act (PDUFA) action date of September 30, 2020 If approved, RYONCIL launch in the US planned for Q4 CY2020 | * This vote includes a change to the original vote by one of the ODAC panel members after electronic voting closed

RYONCIL: U.S. Regulatory and Commercial Strategy FDA action date set for September 30, 2020 Commercialization strategy in place for product launch Ramp-up for inventory build Building out efficient, targeted sales force - 15 centers account for ~50% of patients Label extension planned for treatment of adult SR-aGVHD Lifecycle strategy |

| Post-marketing Study in Adults with SR-aGVHD Utilize remestemcel-L manufactured using optimized process Advisory Board of GVHD experts convened August 2020 Planning underway for randomized controlled trial of remestemcel-L vs standard of care in adult SR-aGVHD patients Designed to demonstrate improved overall response and survival Focus on adults with continued high unmet need despite approved therapies or who have not responded to existing therapies Remestemcel-L: Lifecycle Extension Strategy
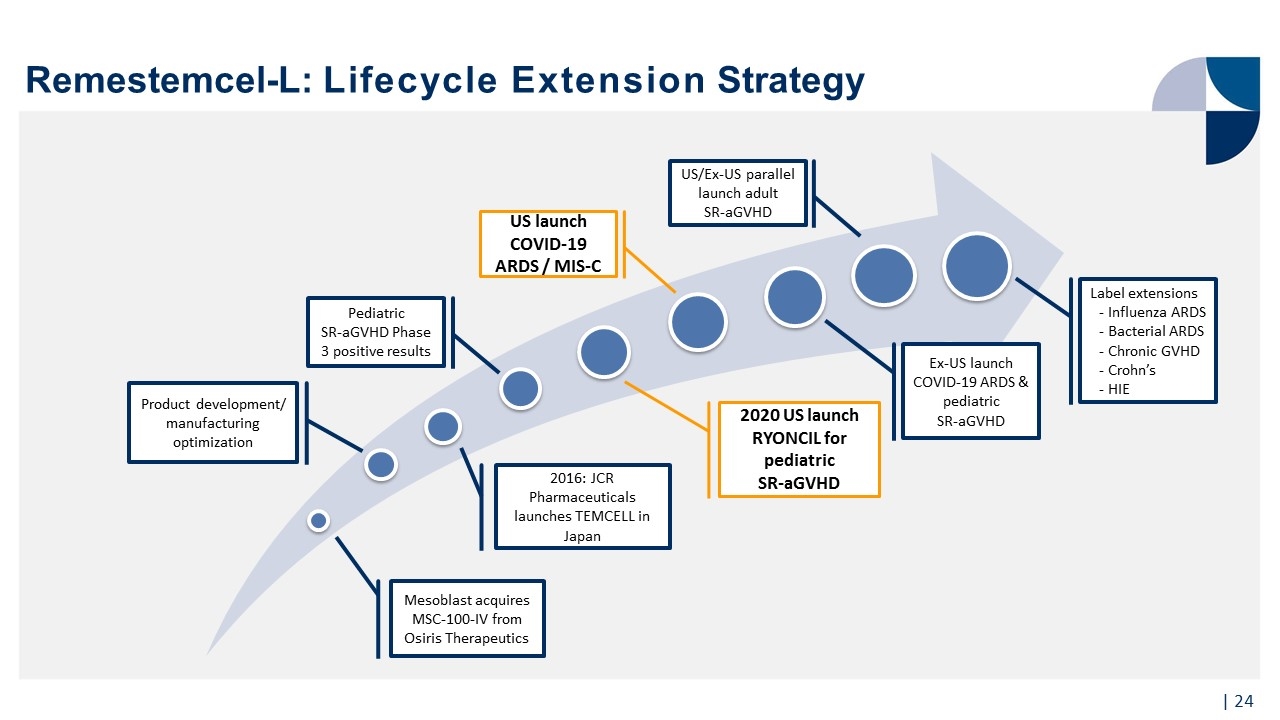
Remestemcel-L: Lifecycle Extension Strategy Mesoblast acquires MSC-100-IV from Osiris Therapeutics Product development/ manufacturing optimization 2016: JCR Pharmaceuticals launches TEMCELL in Japan 2020 US launch RYONCIL for pediatric SR-aGVHD US/Ex-US parallel launch adult SR-aGVHD Pediatric SR-aGVHD Phase 3 positive results Ex-US launch COVID-19 ARDS & pediatric SR-aGVHD US launch COVID-19 ARDS / MIS-C Label extensions Influenza ARDS Bacterial ARDS Chronic GVHD Crohn’s HIE |
Remestemcel-L: Potential New Treatment Paradigm in Adults & Children with COVID-19

Overview – Remestemcel-L for ARDS due to COVID-19 | COVID-19 is a respiratory virus with a high mortality due to a severe inflammatory condition of the lungs called acute respiratory distress syndrome (ARDS) ARDS is caused by cytokine storm in lungs of patients infected with COVID-19 and is the primary cause of death The extensive safety data of remestemcel-L and its anti-inflammatory effects in aGVHD makes a compelling rationale for evaluating remestemcel-L in COVID-19 ARDS Intravenous delivery of remestemcel-L results in selective migration to the lungs making inflammatory lung disease an ideal target for this therapy Remestemcel-L has the potential to tame the cytokine storm in ARDS and may offer a life-saving treatment for those suffering from COVID-19
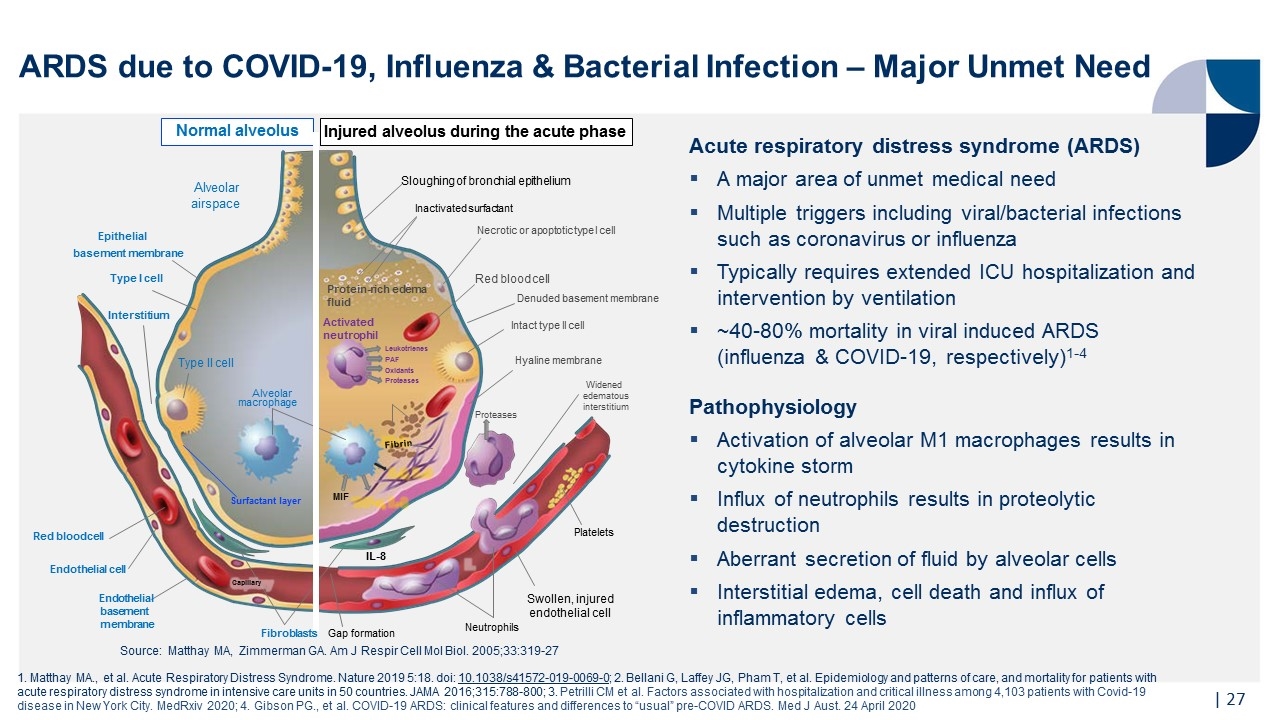
| ARDS due to COVID-19, Influenza & Bacterial Infection – Major Unmet Need Acute respiratory distress syndrome (ARDS) A major area of unmet medical need Multiple triggers including viral/bacterial infections such as coronavirus or influenza Typically requires extended ICU hospitalization and intervention by ventilation ~40-80% mortality in viral induced ARDS (influenza & COVID-19, respectively)1-4 Pathophysiology Activation of alveolar M1 macrophages results in cytokine storm Influx of neutrophils results in proteolytic destruction Aberrant secretion of fluid by alveolar cells Interstitial edema, cell death and influx of inflammatory cells Surfactant layer Interstitium Red blood cell Endothelial basement membrane Fibroblasts Endothelial cell Neutrophils Swollen, injured endothelial cell Platelets Inactivated surfactant Alveolar macrophage Type II cell Gap formation IL-8 Hyaline membrane Red blood cell Alveolar airspace MIF Fibrin Normal alveolus Sloughing of bronchial epithelium Capillary Denuded basement membrane Intact type II cell Necrotic or apoptotic type I cell Protein-rich edema fluid Activated neutrophil Leukotrienes PAF Oxidants Proteases Widened edematous interstitium Proteases Type I cell Epithelial basement membrane Injured alveolus during the acute phase Source: Matthay MA, Zimmerman GA. Am J Respir Cell Mol Biol. 2005;33:319-27 1. Matthay MA., et al. Acute Respiratory Distress Syndrome. Nature 2019 5:18. doi: 10.1038/s41572-019-0069-0; 2. Bellani G, Laffey JG, Pham T, et al. Epidemiology and patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 2016;315:788-800; 3. Petrilli CM et al. Factors associated with hospitalization and critical illness among 4,103 patients with Covid-19 disease in New York City. MedRxiv 2020; 4. Gibson PG., et al. COVID-19 ARDS: clinical features and differences to “usual” pre-COVID ARDS. Med J Aust. 24 April 2020

Compassionate Use Data from Emergency IND 12 patients with moderate or severe ARDS received two infusions of remestemcel-L at Mt. Sinai Hospital in New York City Nine patients (75%) successfully came off ventilator support at a median of 10 days and were discharged from hospital This contrasts with only 9% of COVID-19 patients able to be extubated and a 12% survival rate in two major NY hospital networks during same time period1,2 This pilot data informed design of the ongoing 300-patient randomized controlled clinical trial Trial is being conducted at up to 30 teaching hospitals in the US Promising Pilot Data in Ventilator-Dependent Patients with COVID-19 ARDS | 1 Petrilli CM et al. Factors associated with hospitalization and critical illness among 4,103 patients with Covid-19 disease in New York City. MedRxiv 2020 doi: https://www.medrxiv.org/content/10.1101/2020.04.08.20057794v1.full.pdf 2. Richardson S et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020. doi:10.1001/jama.2020.6775

| Multi-center, randomized, controlled, blinded study to assess safety and efficacy of remestemcel-L plus standard of care (SOC) versus placebo plus SOC in subjects with moderate/severe ARDS on ventilator due to COVID-19 Up to 300 patients randomized 1:1 to receive SOC with placebo or remestemcel-L Primary endpoint all cause mortality up to 30 days, key secondary days alive off ventilator within 60 days Recruitment expected to complete during Q4 CY2020, with interim analyses planned which could result in early stoppage for efficacy or futility Phase 3 Trial of 300 Patients with ARDS due to COVID-19

Key Milestones for Remestemcel-L in COVID-19 ARDS Interim analyses planned which could result in stopping the trial early for efficacy or futility First interim analysis is due early September after 30% of patients reach the primary endpoint Seek expedited regulatory approval subject to positive data read-out Manufacturing scale-up to meet projected increase in capacity requirements for maturing pipeline, including GVHD label extensions and COVID-19 ARDS Increase manufacturing footprint for capacity expansion Implement proprietary xeno-free technologies to increase yields and output Plan for long-term move to 3D bioreactors to reduce labor and improve manufacturing efficiencies Establish further manufacturing and commercialization partnerships |

Remestemcel-L for Children With COVID-19 | Children hospitalized with COVID-19 develop both ARDS (22%) and a life-threatening inflammation called Multisystem inflammatory Syndrome (MIS-C) which involves multiple critical organs and their vasculature In approximately 50% of cases, MIS-C is associated with significant cardiovascular complications that directly involve heart muscle and may result in decreased cardiac function These children often show no evidence of active COVID-19 infection, but have antibodies against COVID-19, indicating prior infection and suggesting an autoimmune process as the cause of MIS-C Mesoblast has established an EAP filed with the FDA which provides physicians with access to remestemcel-L in COVID-19 infected children aged 2 months-17 years with cardiovascular and other complications of MIS-C The first patient has received treatment under the EAP and has been discharged from the hospital Mesoblast will continue to monitor the outcome in all MIS-C patients treated under the EAP to establish the safety and effectiveness of the protocol in children with this potentially life-threatening complication of COVID-19
Update on Other Phase 3 Product Candidates Heart Failure Chronic Low Back Pain

Partnerships and License Agreements Product candidates MPC-06-ID Strategic partnership to develop and commercialize MPC-06-ID in Europe & Latin America Mesoblast will receive up to US$150 million in upfront and milestone payments prior to product launch Milestone payments could exceed US$1 billion depending on patient adoption Mesoblast will also receive tiered double digit royalties on product sales REVASCORTM Exclusive cardiovascular rights in China Mesoblast received US$40 million in an upfront payment and equity placement Eligible for additional milestones and royalties CHRONIC LOW BACK PAIN - DEGENERATIVE DISC DISEASE CARDIOVASCULAR – CHRONIC HEART FAILURE PREVALENCE CHINA ~4.5 MILLION PREVALENCE EUROPE ~7.0 MILLION Partnerships and License Agreements Phase 3 Product Candidates for Heart Failure and Chronic Low Back Pain |

REVASCOR® for Advanced and End-Stage Heart Failure In December 2019, the Phase 3 trial in advanced heart failure surpassed the number of primary endpoint events required for trial completion Final study visits for all surviving patients have been completed Ongoing quality review of all data is being completed at the study sites Data readout expected during Q4 CY2020 Results may support regulatory approval in the US Results from a sub-study of 70 patients with end-stage ischemic heart failure and a Left Ventricular Assist Device (LVAD), of 159 randomized patients who received either REVASCOR or saline, were presented at the American College of Cardiology (ACC) Virtual Scientific Sessions Conclusions from the study included MPCs had a beneficial effect on LVAD weaning, major mucosal bleeding, serious adverse events, and readmissions in ischemic heart failure patients End-stage ischemic heart failure patients with LVADs are older and have co-morbidities such as diabetes, thereby closely resembling the majority of patients in Mesoblast’s 566-patient Phase 3 trial of REVASCOR for advanced chronic heart failure |

MPC-06-ID for Chronic Low Back Pain Phase 3 trial of MPC-06-ID for chronic low back pain in 404 patients: Final study visits for all patients have been completed Ongoing quality review of all data is being completed at the study sites Data readout expected during Q4 CY2020 Continued operational progress in strategic partnership for chronic lower back pain with Grünenthal in Europe to complete clinical protocol design, obtain regulatory input, and receive clearance from European regulatory authorities to begin European Phase 3 trial Results from the Phase 3 trials will be considered pivotal to support regulatory approval in the US, as well as in Europe |

Major Operational Milestones for the Next 12 Months Remestemcel-L for SR-aGVHD RYONCIL Priority Review for pediatric SR-aGVHD underway with PDUFA date set for September 30, 2020 If approved, US launch of RYONCIL planned for 2020 Commence trial in adults with SR-aGVHD to expand label Remestemcel-L for Acute Respiratory Distress Syndrome (ARDS) in COVID-19 Ongoing recruitment for Phase 3 multicenter, randomized controlled trial in North America Trial completion expected during Q4 CY2020 Establish strategic partnerships for manufacturing and commercialization REVASCOR for Advanced and End-Stage Heart Failure Data readout for advanced chronic heart failure Phase 3 trial during Q4 CY2020 Initiate confirmatory trial in end-stage heart failure MPC-06-ID for Chronic Low Back Pain Data readout for Phase 3 trial during Q4 CY2020 Obtain clearance from European regulatory authorities to begin European Phase 3 trial |

Thank You