
Mobi-C® CERVICAL DISC IDE Clinical Trial Overview Mobi-C Compared to Anterior Cervical Discectomy and Fusion at One-level

key triAl results Mobi-C demonstrated non-inferiority in overall trial success compared to ACDF at 24 months. At 24 months, Mobi-C demonstrated: • Fewer secondary surgeries at the index level compared to ACDF. • lower rates of adverse events determined to be major complications compared to ACDF. • lower rates of adjacent level degeneration compared to ACDF. • A higher NDi success rate compared to ACDF. • A mean rOM in F/e of 10.8° at the index level. • A mean return to work time 7.5 days shorter than ACDF. Mobi-C is a safe and effective surgical option at one-level in the cervical spine from C3-C7 in indicated patients. Note: Please refer to the Mobi-C Summary of Safety and Effectiveness Data (PMA P110002) at www.fda.gov for complete study results. 73.7% 65.3% 0% 10% 20% 30% 40% 50% 60% 70% 80% Mobi-C ACDF % o f R an do m iz ed P ag415 en ts w ith A va ila bl e D at a Overall Trial Success at 24 Months ONE-LEVEL
tAble OF CONteNts Key Trial Results ...................................................................................................................................................................................................................................... 2 Overall Trial Success: The Composite Endpoint ...................................................................................................................................................................... 4 Composite Endpoint Components Neck Disability index ............................................................................................................................................................................................................................... 6 secondary surgeries at the treated level ....................................................................................................................................................................................... 7 Neurologic status ..................................................................................................................................................................................................................................... 7 radiologic Assessment ........................................................................................................................................................................................................................... 8 Adverse events Determined to be Major Complications ........................................................................................................................................................... 9 Secondary Endpoints range of Motion .....................................................................................................................................................................................................................................10 Adjacent segment Degeneration and Adjacent segment surgeries ..................................................................................................................................11 Patient satisfaction ................................................................................................................................................................................................................................11 Clinical Endpoints return to Work ........................................................................................................................................................................................................................................12 Visual Analogue scale ...........................................................................................................................................................................................................................12 Heterotopic Ossification ......................................................................................................................................................................................................................13 Conclusion ...............................................................................................................................................................................................................................................13 Appendix trial Design and Patient Accounting ...............................................................................................................................................................................................14 Patient Demographics ..........................................................................................................................................................................................................................15 surgery Data ............................................................................................................................................................................................................................................16 inclusion Criteria .....................................................................................................................................................................................................................................16 exclusion Criteria ....................................................................................................................................................................................................................................17 Commonly reported Adverse events .............................................................................................................................................................................................19 secondary surgical interventions at the index level - Procedure Details .........................................................................................................................19 indications ...............................................................................................................................................................................................................................back Cover 2-3
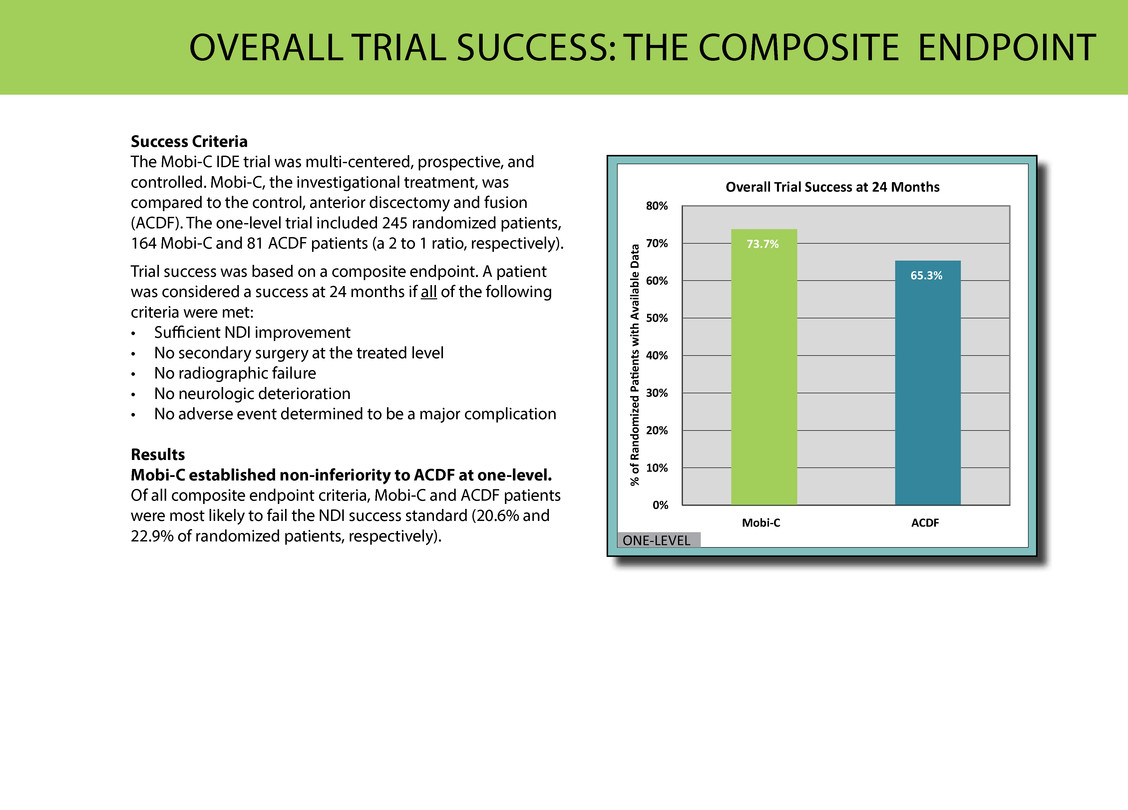
OVerAll triAl suCCess: tHe COMPOsite eNDPOiNt Success Criteria the Mobi-C iDe trial was multi-centered, prospective, and controlled. Mobi-C, the investigational treatment, was compared to the control, anterior discectomy and fusion (ACDF). the one-level trial included 245 randomized patients, 164 Mobi-C and 81 ACDF patients (a 2 to 1 ratio, respectively). trial success was based on a composite endpoint. A patient was considered a success at 24 months if all of the following criteria were met: • sufficient NDi improvement • No secondary surgery at the treated level • No radiographic failure • No neurologic deterioration • No adverse event determined to be a major complication Results Mobi-C established non-inferiority to ACDF at one-level. Of all composite endpoint criteria, Mobi-C and ACDF patients were most likely to fail the NDi success standard (20.6% and 22.9% of randomized patients, respectively). 73.7% 65.3% 0% 10% 20% 30% 40% 50% 60% 70% 80% Mobi-C ACDF % o f R an do m iz ed P ag415 en ts w ith A va ila bl e D at a Overall Trial Success at 24 Months ONE-LEVEL
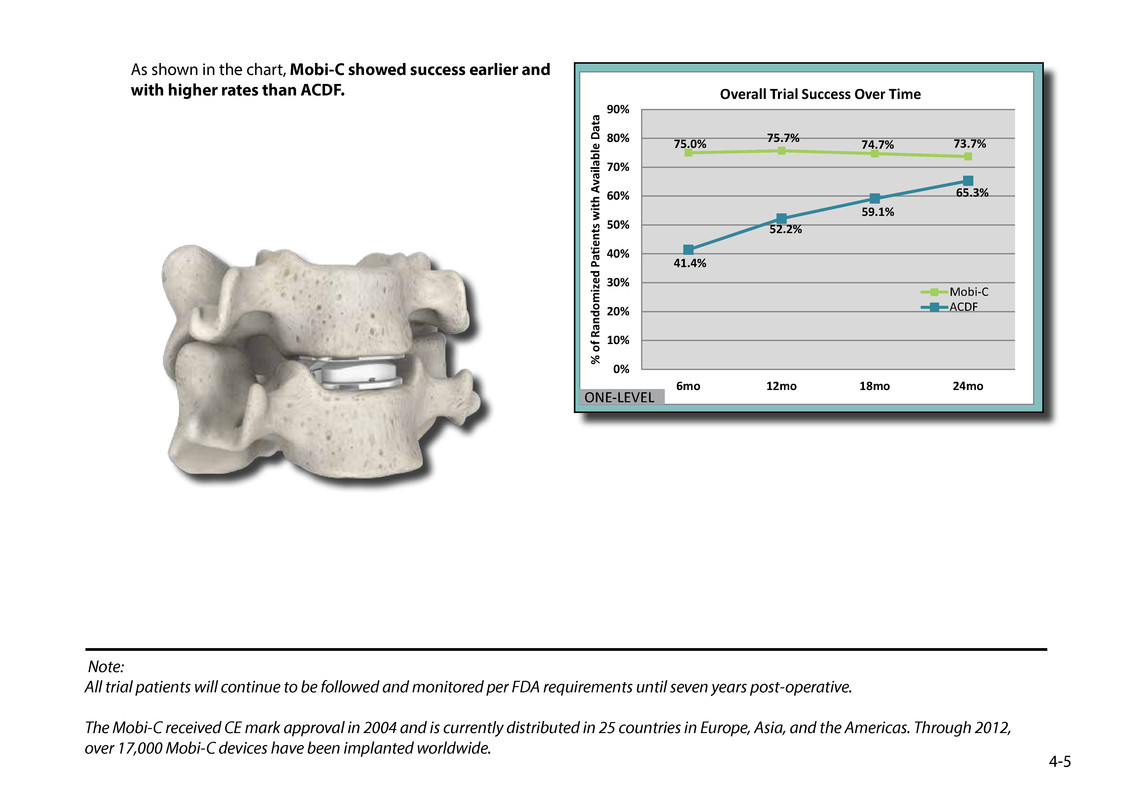
As shown in the chart, Mobi-C showed success earlier and with higher rates than ACDF. Note: All trial patients will continue to be followed and monitored per FDA requirements until seven years post-operative. The Mobi-C received CE mark approval in 2004 and is currently distributed in 25 countries in Europe, Asia, and the Americas. Through 2012, over 17,000 Mobi-C devices have been implanted worldwide. 4-5 75.0% 75.7% 74.7% 73.7% 41.4% 52.2% 59.1% 65.3% 0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 6mo 12mo 18mo 24mo % o f R an do m iz ed Pag415ents with Available D at a Overall Trial Success Over Time Mobi-C ACDF ONE-LEVEL
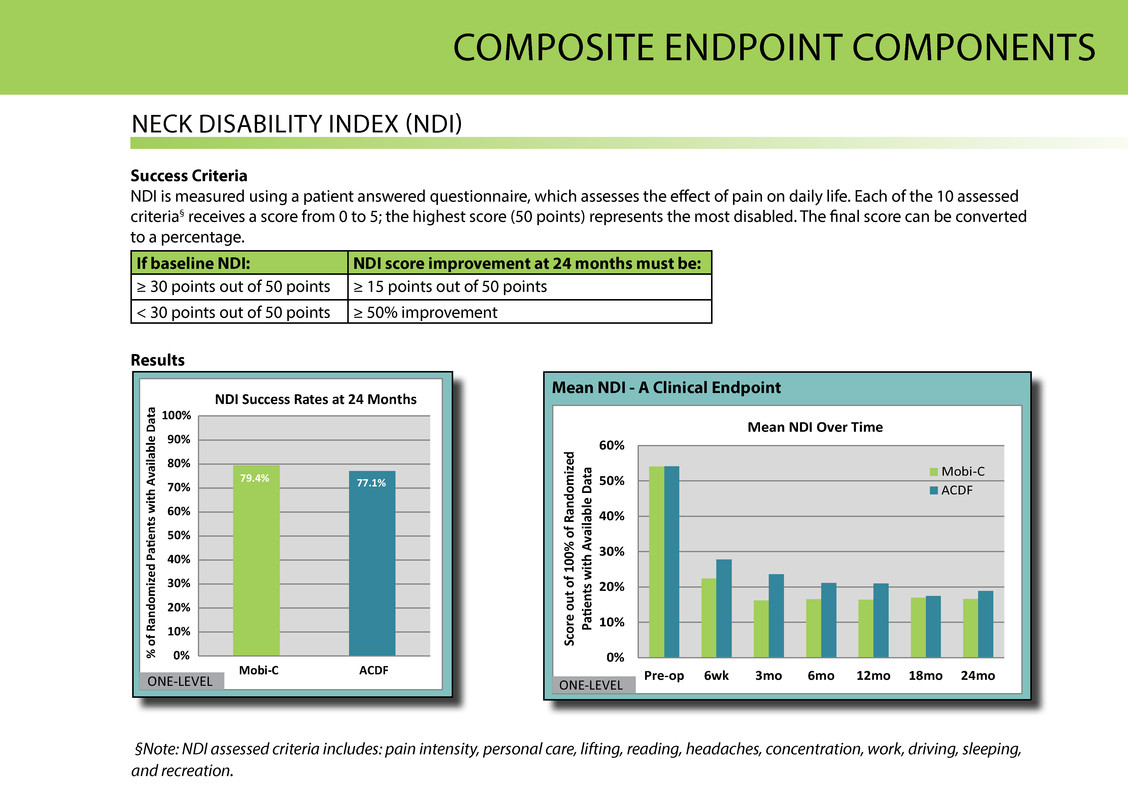
COMPOsite eNDPOiNt COMPONeNts NeCk DisAbility iNDeX (NDi) If baseline NDI: NDI score improvement at 24 months must be: ≥ 30 points out of 50 points ≥ 15 points out of 50 points < 30 points out of 50 points ≥ 50% improvement Results 0% 10% 20% 30% 40% 50% 60% Pre-op 6wk 3mo 6mo 12mo 18mo 24mo Sc or e ou t o f 1 00 % o f R an do m iz ed Pag415ents with Available D at a Mean NDI Over Time Mobi-C ACDF ONE-LEVEL Mean NDI - A Clinical Endpoint 79.4 % 77.1% 0% 10% 20% 30% 40% 50% 60% 70 % 80% 90 % 100% Mobi-C AC DF % o f R an do m iz ed Pag415ents with Available D at a NDI Success Rates at 24 Months ONE-LEVEL Success Criteria NDi is measured using a patient answered questionnaire, which assesses the effect of pain on daily life. each of the 10 assessed criteria§ receives a score from 0 to 5; the highest score (50 points) represents the most disabled. the final score can be converted to a percentage. §Note: NDI assessed criteria includes: pain intensity, personal care, lifting, reading, headaches, concentration, work, driving, sleeping, and recreation.
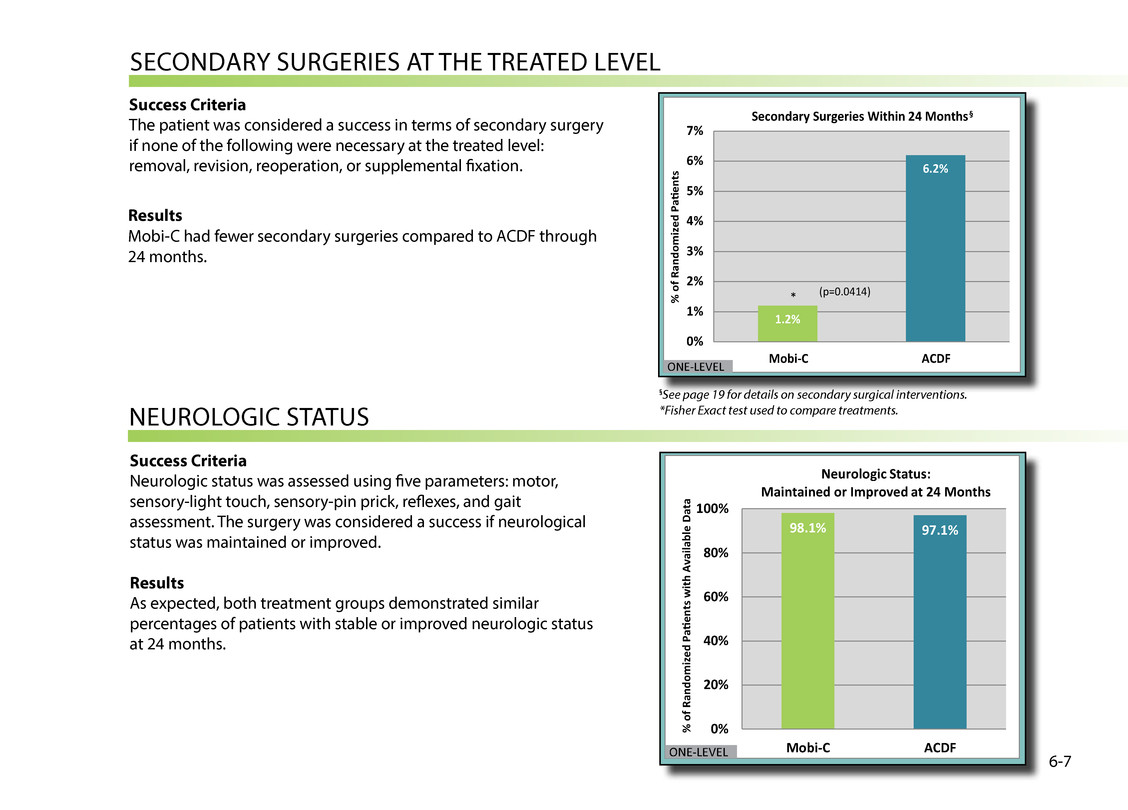
seCONDAry surgeries At tHe treAteD leVel Success Criteria the patient was considered a success in terms of secondary surgery if none of the following were necessary at the treated level: removal, revision, reoperation, or supplemental fixation. Results Success Criteria Neurologic status was assessed using five parameters: motor, sensory-light touch, sensory-pin prick, reflexes, and gait assessment. the surgery was considered a success if neurological status was maintained or improved. Results As expected, both treatment groups demonstrated similar percentages of patients with stable or improved neurologic status at 24 months. NeurOlOgiC stAtus 1.2% 6.2% 0% 1% 2% 3% 4% 5% 6% 7% Mobi-C ACDF % o f R an do m iz ed Pag415en ts Secondary Surgeries Within 24 Months § (p=0.0414)* ONE-LEVEL 9 8.1 % 97.1 % 0% 20% 40% 60% 80 % 100% Mobi-C ACDF % o f R an do m iz ed Pag415ents with Available D at a Neurologic Status: Maintained or Improved at 24 Months ONE-LEVEL 6-7 §See page 19 for details on secondary surgical interventions. *Fisher Exact test used to compare treatments. Mobi-C had fewer secondary surgeries compared to ACDF through 24 months.

rADiOlOgiC AssessMeNt Success Criteria Mobi-C Radiographic Success ACDF Radiographic Success • ≥ 2° segmental movement in flexion/extension, or • Absence of continuous bridging bone • < 2° of segmental movement in flexion/extension, and • Absence of radiolucent lines at ≥ 50% of the graft-vertebral interfaces, and • the presence of bridging trabecular bone For consistency, an independent core lab performed the radiographic analysis for this trial. radiographic outcomes measured include: fusion status, device condition, subsidence, migration, range of motion, radiolucency, and disc height. Results 97.0% 89.3% 0% 20% 40% 60% 80% 100% Mobi-C ACDF % o f R an do m iz ed Pag415ents with Available D at a Radiographic Success Rate at 24 Months ONE-LEVEL Mobi-C radiographic analysis showed no instances of migration, subsidence, or expulsion *Note: To avoid inconsistent treatment and any resulting clinical benefits or disadvantages, non-steroidal anti-inflammatories (NSAIDs) were not allowed prophylactically between one week prior to surgery until three months following surgery in either treatment group, unless intended to treat identified heterotopic ossification (HO). COMPOsite eNDPOiNt COMPONeNts

ADVerse eVeNts DeterMiNeD tO be MAjOr COMPliCAtiONs Success Criteria the Clinical events Committee (CeC) independently determined if an adverse event (Ae)* was a major complication. if so, the study subject was considered a failure. Results 2.8% 3.7% 0% 1% 2% 3% 4% 5% 6% 7% Mobi-C ACDF % of All Pag415en ts AEs Deemed to be Major Complicag415ons Through Month 24 ONE-LEVEL 8-9 *Note: Adverse event data includes the Mobi-C non-randomized training cases. Major Complications Determined Via CEC Review of Adverse Events (One-level) Treatment Group Adverse Event by Patient ACDF (n=81) Worsening arm pain Failure of fusion, C5-C6 Pseudoarthrosis Mobi-C (n=179) Heterotopic bone formation anterior to the device left arm pain temporary paralysis-bilateral le Anterior ossification at C5-C6 Postop hoarseness from removal of the device
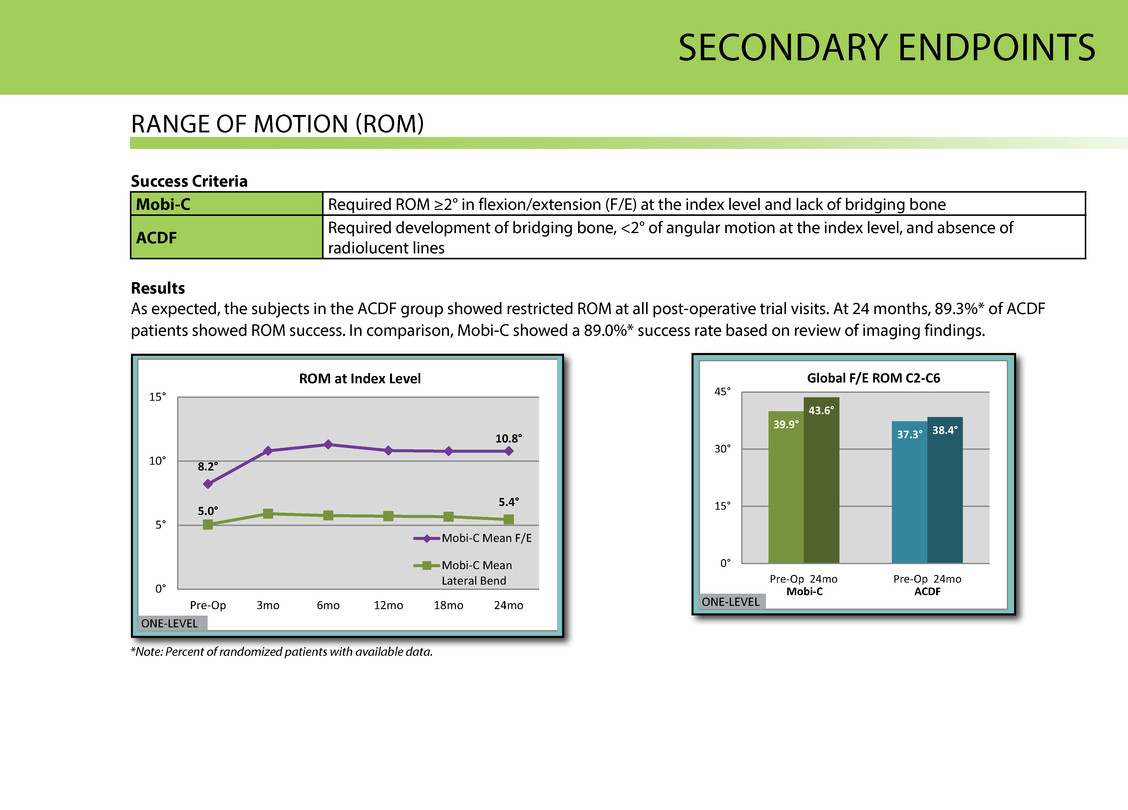
seCONDAry eNDPOiNts rANge OF MOtiON (rOM) Success Criteria Results Mobi-C required rOM ≥2° in flexion/extension (F/e) at the index level and lack of bridging bone ACDF required development of bridging bone, <2° of angular motion at the index level, and absence of radiolucent lines Results As expected, the subjects in the ACDF group showed restricted rOM at all post-operative trial visits. At 24 months, 89.3%* of ACDF patients showed rOM success. in comparison, Mobi-C showed a 89.0%* success rate based on review of imaging findings. 8.2° 10.8° 5.0° 5.4° 0° 5° 10° 15° Pre-Op 3mo 6mo 12mo 18mo 24mo ROM at Index Level Mobi-C Mean F/E Mobi-C Mean Lateral Bend ONE-LEVEL 39.9° 37.3° 43.6° 38.4° 0° 15° 30° 45° Pre-Op 24mo Mobi-C ACDF Pre-Op 24mo Global F/ E ROM C2 -C 6 ONE-LEVEL *Note: Percent of randomized patients with available data.
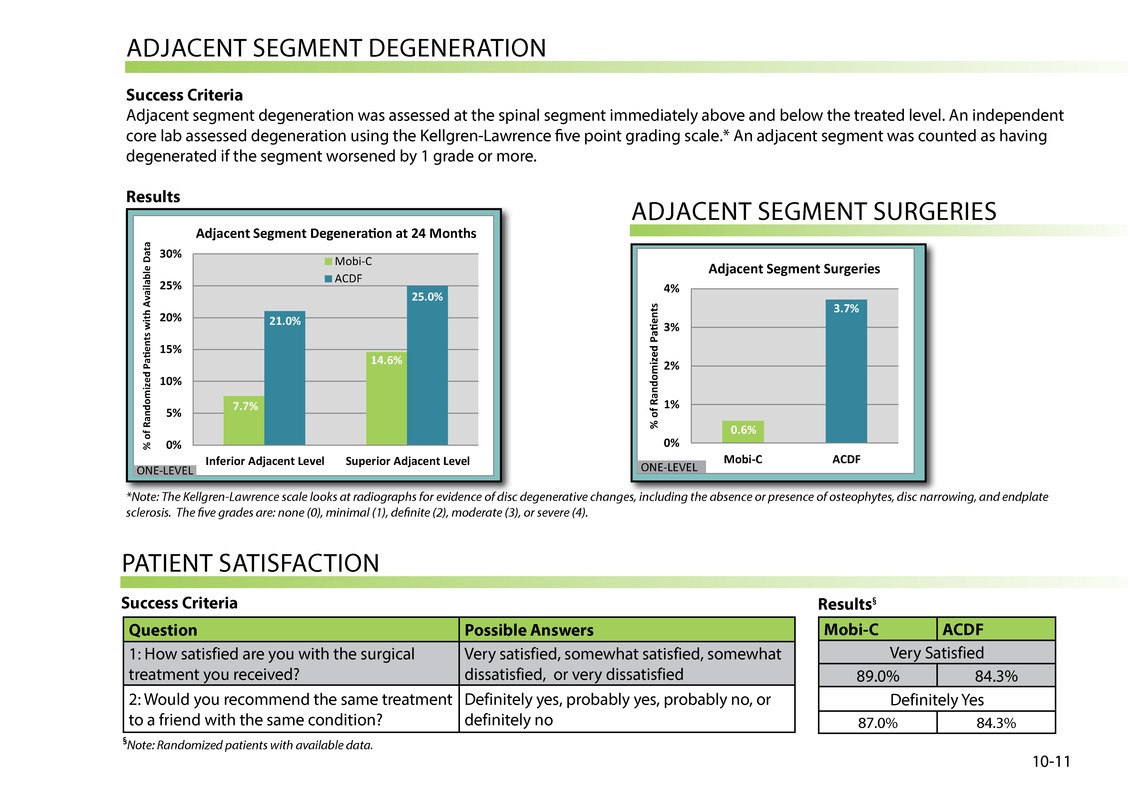
ADjACeNt segMeNt DegeNerAtiON Success Criteria Adjacent segment degeneration was assessed at the spinal segment immediately above and below the treated level. An independent core lab assessed degeneration using the kellgren-lawrence five point grading scale.* An adjacent segment was counted as having degenerated if the segment worsened by 1 grade or more. Results 7.7% 14.6% 21.0% 25.0% 0% 5% 10% 15% 20% 25% 30% Inferior Adjacent Level Superior Adjacent Level % o f R an do m iz ed Pag415ents with Available D at a Adjacent Segment Degenerag415on at 24 Months Mobi-C ACDF ONE-LEVEL ADjACeNt segMeNt surgeries 0.6% 3.7% 0% 1% 2% 3% 4% Mobi-C ACDF % o f R an do m iz ed P ag415 en ts Adjacent Segment Surgeries ONE-LEVEL 10-11 *Note: The Kellgren-Lawrence scale looks at radiographs for evidence of disc degenerative changes, including the absence or presence of osteophytes, disc narrowing, and endplate sclerosis. The five grades are: none (0), minimal (1), definite (2), moderate (3), or severe (4). PAtieNt sAtisFACtiON Question Possible Answers 1: How satisfied are you with the surgical treatment you received? Very satisfied, somewhat satisfied, somewhat dissatisfied, or very dissatisfied 2: Would you recommend the same treatment to a friend with the same condition? Definitely yes, probably yes, probably no, or definitely no Mobi-C ACDF Very satisfied 89.0% 84.3% Definitely yes 87.0% 84.3% Success Criteria Results§ §Note: Randomized patients with available data.
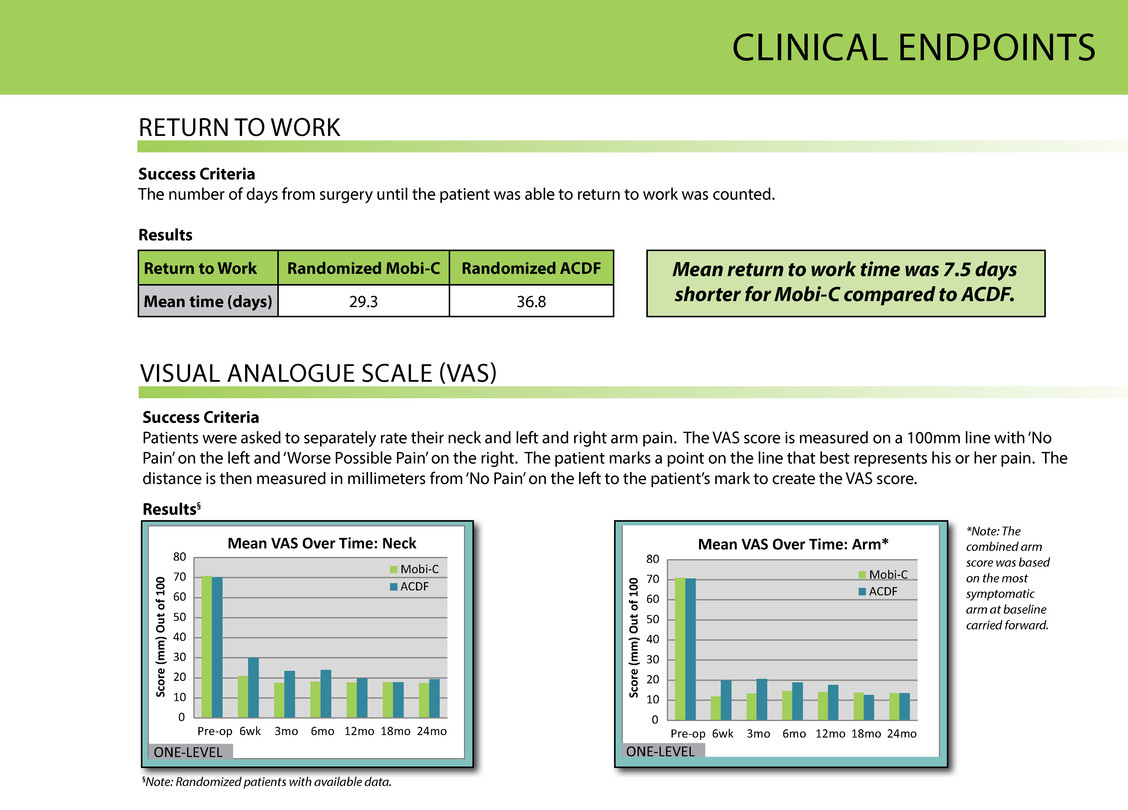
°° CliNiCAl eNDPOiNts VisuAl ANAlOgue sCAle (VAs) Success Criteria Patients were asked to separately rate their neck and left and right arm pain. the VAs score is measured on a 100mm line with ‘No Pain’ on the left and ‘Worse Possible Pain’ on the right. the patient marks a point on the line that best represents his or her pain. the distance is then measured in millimeters from ‘No Pain’ on the left to the patient’s mark to create the VAs score. Results§ returN tO WOrk *Note: The combined arm score was based on the most symptomatic arm at baseline carried forward. 0 10 20 30 40 50 60 70 80 Pre-op 6wk 3mo 6mo 12mo 18mo 24mo Sc or e (m m ) O ut o f 1 00 Mean VAS Over Time: Neck Mobi-C ACDF ONE-LEVEL §Note: Randomized patients with available data. Success Criteria the number of days from surgery until the patient was able to return to work was counted. Results Mean return to work time was 7.5 days shorter for Mobi-C compared to ACDF. Return to Work Randomized Mobi-C Randomized ACDF Mean time (days) 29.3 36.8 0 10 20 30 40 50 60 70 80 Pre-op 6wk 3mo 6mo 12mo 18mo 24mo Sc or e (m m ) O ut o f 1 00 Mean VAS Over Time: Arm * Mobi-C ACDF ONE-LEVEL
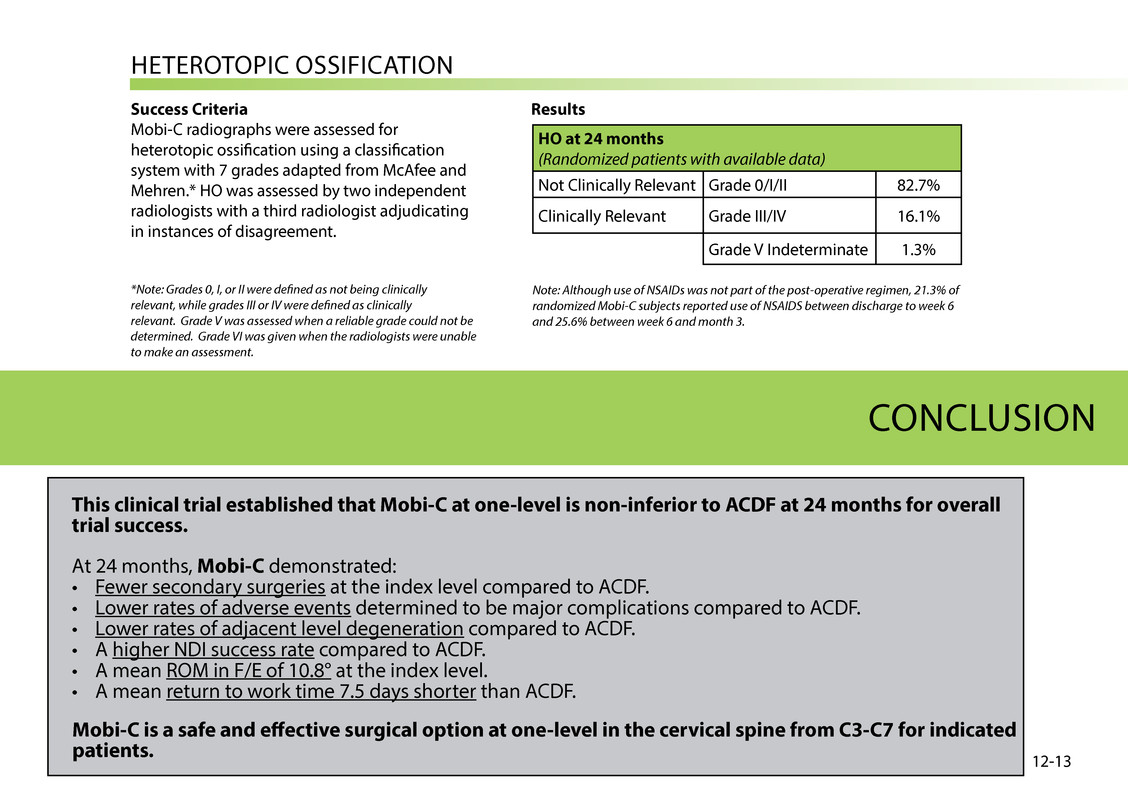
HeterOtOPiC OssiFiCAtiON Success Criteria Mobi-C radiographs were assessed for heterotopic ossification using a classification system with 7 grades adapted from McAfee and Mehren.* HO was assessed by two independent radiologists with a third radiologist adjudicating in instances of disagreement. *Note: Grades 0, I, or II were defined as not being clinically relevant, while grades III or IV were defined as clinically relevant. Grade V was assessed when a reliable grade could not be determined. Grade VI was given when the radiologists were unable to make an assessment. 12-13 This clinical trial established that Mobi-C at one-level is non-inferior to ACDF at 24 months for overall trial success. At 24 months, Mobi-C demonstrated: • Fewer secondary surgeries at the index level compared to ACDF. • lower rates of adverse events determined to be major complications compared to ACDF. • lower rates of adjacent level degeneration compared to ACDF. • A higher NDi success rate compared to ACDF. • A mean rOM in F/e of 10.8° at the index level. • A mean return to work time 7.5 days shorter than ACDF. CONClusiON Results Note: Although use of NSAIDs was not part of the post-operative regimen, 21.3% of randomized Mobi-C subjects reported use of NSAIDS between discharge to week 6 and 25.6% between week 6 and month 3. HO at 24 months (Randomized patients with available data) Not Clinically relevant grade 0/i/ii 82.7% Clinically relevant grade iii/iV 16.1% grade V indeterminate 1.3% Mobi-C is a safe and effective surgical option at one-level in the cervical spine from C3-C7 for indicated patients.
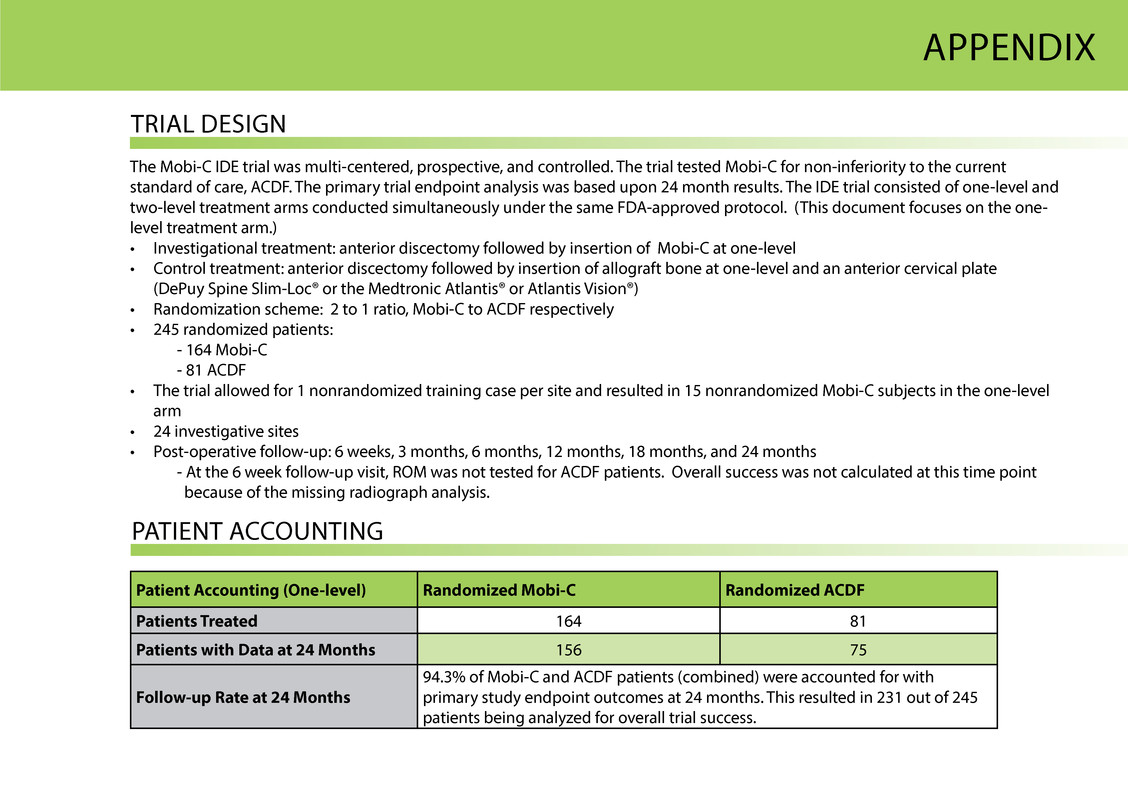
APPeNDiX triAl DesigN the Mobi-C iDe trial was multi-centered, prospective, and controlled. the trial tested Mobi-C for non-inferiority to the current standard of care, ACDF. the primary trial endpoint analysis was based upon 24 month results. the iDe trial consisted of one-level and two-level treatment arms conducted simultaneously under the same FDA-approved protocol. (this document focuses on the one- level treatment arm.) • investigational treatment: anterior discectomy followed by insertion of Mobi-C at one-level • Control treatment: anterior discectomy followed by insertion of allograft bone at one-level and an anterior cervical plate (DePuy spine slim-loc® or the Medtronic Atlantis® or Atlantis Vision®) • randomization scheme: 2 to 1 ratio, Mobi-C to ACDF respectively • 245 randomized patients: - 164 Mobi-C - 81 ACDF • the trial allowed for 1 nonrandomized training case per site and resulted in 15 nonrandomized Mobi-C subjects in the one-level arm • 24 investigative sites • Post-operative follow-up: 6 weeks, 3 months, 6 months, 12 months, 18 months, and 24 months - At the 6 week follow-up visit, rOM was not tested for ACDF patients. Overall success was not calculated at this time point because of the missing radiograph analysis. PAtieNt ACCOuNtiNg Patient Accounting (One-level) Randomized Mobi-C Randomized ACDF Patients Treated 164 81 Patients with Data at 24 Months 156 75 Follow-up Rate at 24 Months 94.3% of Mobi-C and ACDF patients (combined) were accounted for with primary study endpoint outcomes at 24 months. this resulted in 231 out of 245 patients being analyzed for overall trial success.
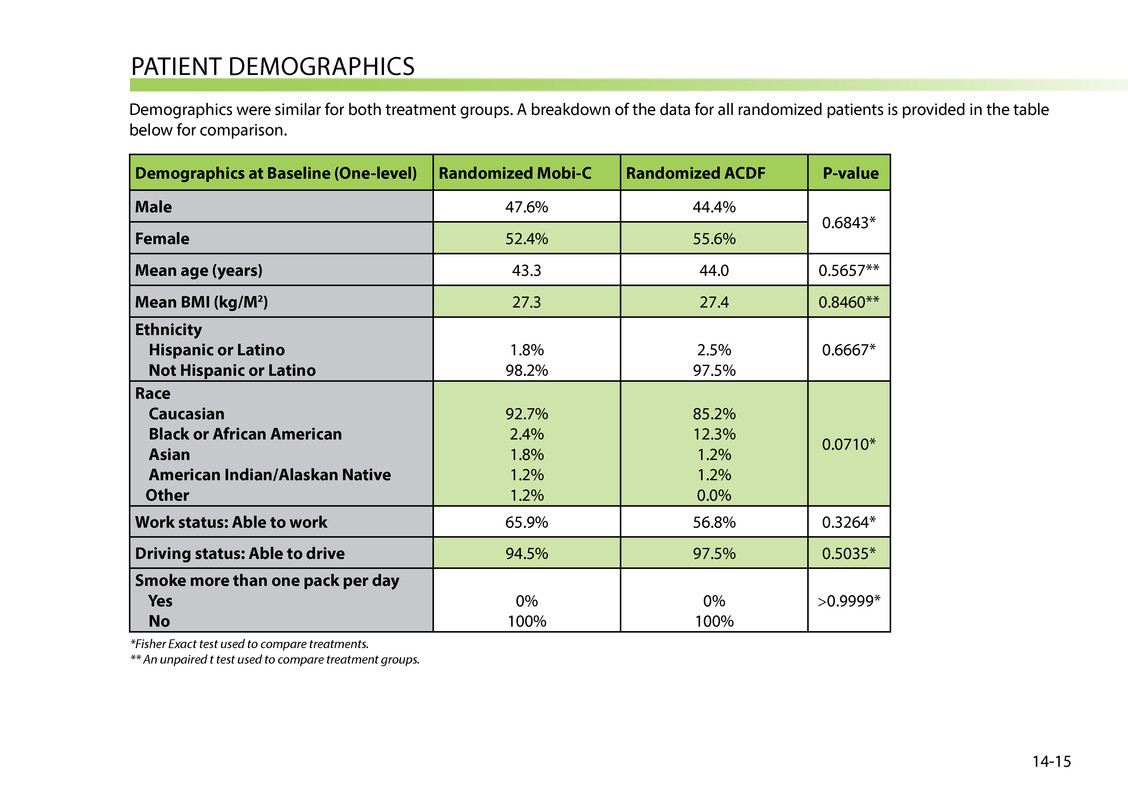
PAtieNt DeMOgrAPHiCs Demographics were similar for both treatment groups. A breakdown of the data for all randomized patients is provided in the table below for comparison. Demographics at Baseline (One-level) Randomized Mobi-C Randomized ACDF P-value Male 47.6% 44.4% 0.6843* Female 52.4% 55.6% Mean age (years) 43.3 44.0 0.5657** Mean BMI (kg/M2) 27.3 27.4 0.8460** Ethnicity Hispanic or Latino Not Hispanic or Latino 1.8% 98.2% 2.5% 97.5% 0.6667* Race Caucasian Black or African American Asian American Indian/Alaskan Native Other 92.7% 2.4% 1.8% 1.2% 1.2% 85.2% 12.3% 1.2% 1.2% 0.0% 0.0710* Work status: Able to work 65.9% 56.8% 0.3264* Driving status: Able to drive 94.5% 97.5% 0.5035* Smoke more than one pack per day Yes No 0% 100% 0% 100% >0.9999* 14-15 *Fisher Exact test used to compare treatments. ** An unpaired t test used to compare treatment groups.

surgery DAtA Surgery Data (One-level) Randomized Mobi-C Randomized ACDF Mean Est. Blood Loss (mls) 45.0 48.1 Mean Length of Hospital Stay (days) 2.0 2.1 Mean Operative Time (hrs) 1.5 1.3 Levels Treated (One-level) C3-C4 0.6% 4.9% C4-C5 6.7% 2.5% C5-C6 56.1% 56.8% C6-C7 36.6% 35.8% APPeNDiX iNClusiON CriteriA enrollment in the one level Mobi-C® study arm was limited to patients who met the following inclusion criteria. 1. Age 18-69 years. 2. Diagnosis of radiculopathy or myeloradiculopathy of the cervical spine, with pain, paresthesias or paralysis in a specific nerve root distribution C3 through C7, including at least one of the following: - Neck and/or arm pain (at least 30 mm on the 100 mm visual analogue scale [VAs] scale). - Decreased muscle strength of at least one level on the clinical evaluation 0 to 5 scale. - Abnormal sensation including hyperesthesia or hypoesthesia; and/or - Abnormal reflexes. 3. symptomatic at one-level from C3 to C7.

4. radiographically determined pathology at the level to be treated correlating to primary symptoms including at least one of the following: - Decreased disc height on radiography, computed tomography (Ct), or magnetic resonance imaging (Mri) in comparison to a normal adjacent disc. - Degenerative spondylosis on Ct or Mri. - Disc herniation on Ct or Mri. 5. NDi score of ≥ 15/50 or ≥ 30%. 6. unresponsive to nonoperative, conservative treatment (rest, heat, electrotherapy, physical therapy, chiropractic care and/or analgesics) for: - Approximately six weeks from radiculopathy or myeloradiculopathy symptom onset; or - Have the presence of progressive symptoms or signs of nerve root/spinal cord compression despite continued nonoperative conservative treatment. 7. Appropriate for treatment using an anterior surgical approach, including having no prior surgery at the operative level and no prior cervical fusion procedure at any level. 8. reported to be medically cleared for surgery. 9. reported to be physically and mentally able and willing to comply with the Protocol, including the ability to read and complete required forms and willing and able to adhere to the scheduled follow-up visits and requirements of the Protocol. 10. Written informed consent provided by subject or subject’s legally authorized representative. 11. Willingness to discontinue all use of non-steroidal anti-inflammatory drugs (NsAiDs) from one week before surgery until 3 months after surgery. 16-17 eXClusiON CriteriA Patients were not permitted to enroll in the Mobi-C® study if they met any of the following exclusion criteria. 1. reported to have an active systemic infection or infection at the operative site. 2. reported to have a history of or anticipated treatment for active systemic infection, including HiV or Hepatitis C. 3. More than one immobile vertebral level between C1 to C7 from any cause including but not limited to congenital abnormalities and osteoarthritic “spontaneous” fusions. 4. Previous trauma to the C3 to C7 levels resulting in significant bony or disco-ligamentous cervical spine injury. 5. reported to have had any prior spine surgery at the operative level. 6. reported to have had a prior cervical fusion procedure at any level. 7. Axial neck pain in the absence of other symptoms of radiculopathy or myeloradiculopathy justifying the need for surgical intervention. 8. Disc height less than 3 mm as measured from the center of the disc in a neutral position and disc height less than 20% of the anterior-posterior width of the inferior vertebral body. 9. radiographic confirmation of severe facet joint disease or degeneration. 10. reported to have an increased risk of osteoporosis/osteopenia. this was defined as a t-score less than (worse than) -1.5 on a previous or required Hologic sahara or dual energy X-ray absorptiometry (DeXA) scan. All subjects that met one or more of the following were to undergo a Hologic sahara or DeXA scan as part of the trial enrollment procedures: - Females 50 years and older; - Females who were post-menopausal or post-hysterectomy with oophorectomy; - subjects taking bisphosphonate medication for the treatment of osteoporosis; and/or - subjects with history of chronic use of high dose steroids. High dose steroid use is defined as part of exclusion Criterion #22.

APPeNDiX eXClusiON CriteriA CONtiNueD All females less than 50 years of age, and all males, who had not had a Hologic sahara or DeXA scan within six months of surgery, were screened for osteoporosis using the simple Calculated Osteoporosis risk estimation (sCOre) questionnaire. subjects whose screening suggests increased risk (sCOre greater than 6) were to undergo a Hologic sahara or DeXA scan as part of the trial enrollment procedures. 11. reported to have Paget’s disease, osteomalacia or any other metabolic bone disease other than osteoporosis, which is addressed above. 12. reported active malignancy that included a history of any invasive malignancy (except non-melanoma skin cancer), unless the subject had been treated with curative intent and there had been no clinical signs or symptoms of the malignancy for at least five years. 13. symptomatic DDD or significant cervical spondylosis at more than two-levels. 14. spondylolysis. 15. Marked cervical instability on resting lateral or flexion/extension radiographs demonstrated by: - translation ≥ 3.5 mm, and/or - greater than 11° angular difference to that of either adjacent level. 16. known allergy to cobalt, chromium, molybdenum or polyethylene. 17. segmental angulation of greater than 11° at treatment or adjacent levels. 18. reported pregnancy or nursing at time of enrollment, or with plans to become pregnant within the next three years. 19. reported to have rheumatoid arthritis, lupus, or other autoimmune disease that affect the musculoskeletal system. 20. Congenital bony and/or spinal cord abnormalities that affect spinal stability. 21. reported to have diseases or conditions that would preclude accurate clinical evaluation (e.g. neuromuscular disorders). 22. reported concomitant conditions requiring daily, high-dose oral and/or inhaled steroids. High dose steroid use is defined as: - Daily, chronic use of oral steroids of 5 mg/day or greater. - Daily, chronic use of inhaled corticosteroids (at least twice per day). - use of short-term (less than 10 days) oral steroids at a daily dose greater than 40 mg within one month of the trial procedure. 23. reported to have current or recent history of substance abuse (alcoholism and/or narcotic addiction) requiring intervention. 24. Clinically severe Obesity, as defined by National institutes of Health (NiH) Clinical guidelines body Mass index (bMi) > 40). 25. reported use of any other investigational drug or medical device within the last 30 days prior to surgery. 26. evidence of symptomatic moderate to severe facet joint degeneration or disease where the investigator felt this was a major contributor to the subject’s pain as diagnosed by injection and imaging. 27. reported to be taking medications known to potentially interfere with bone/soft tissue healing (e.g., high-dose oral and/or inhaled steroids, immunosuppressant medication, chemotherapeutic agents). High dose steroid use is defined as part of exclusion Criterion #22. 28. reported to have pending personal litigation relating to spinal injury (worker’s compensation was not an exclusion). 29. reported to have a current history of heavy smoking (more than one pack of cigarettes per day). 30. Anticipated or potential relocation greater than 50 miles that may interfere with completion of follow-up examinations. 31. reported to have mental illness or belonged to a vulnerable population, as determined by the investigator (e.g., prisoner or developmentally disabled), that would compromise ability to provide informed consent or compliance with follow-up requirements. 32. reported to have an uncontrolled seizure disorder. 33. reported to have taken epidural steroids within 14 days prior to surgery.
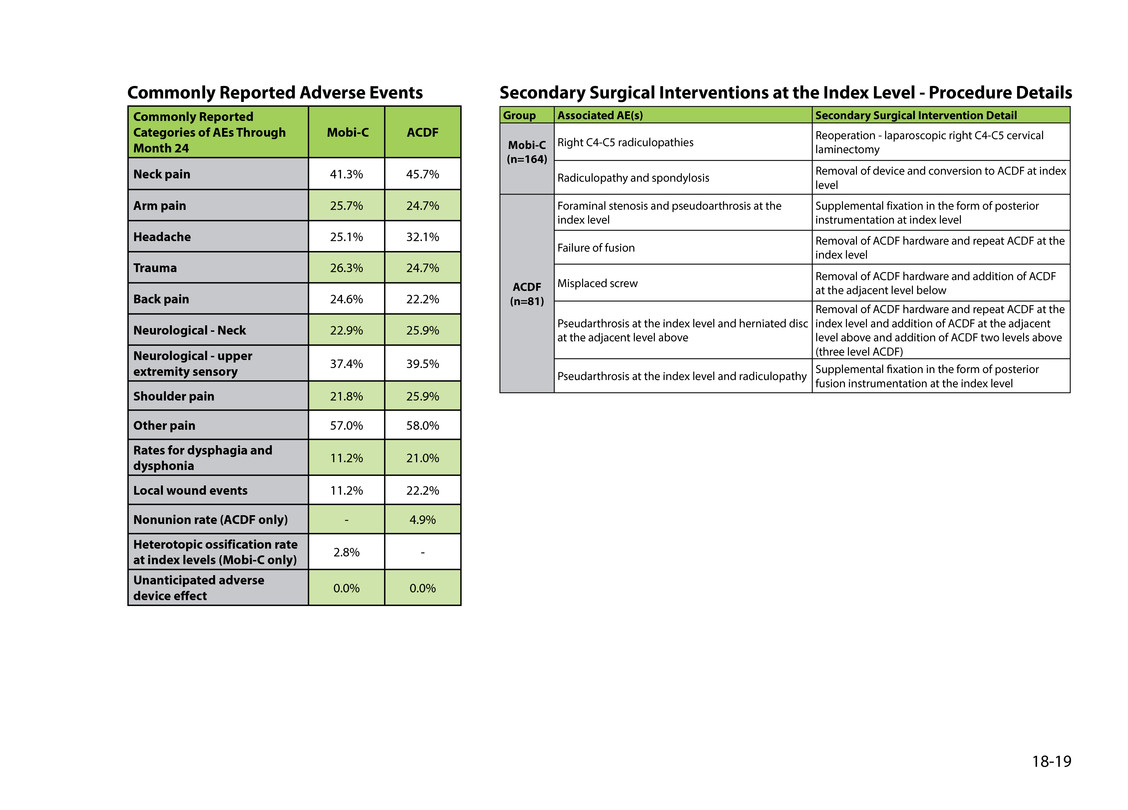
Commonly Reported Categories of AEs Through Month 24 Mobi-C ACDF Neck pain 41.3% 45.7% Arm pain 25.7% 24.7% Headache 25.1% 32.1% Trauma 26.3% 24.7% Back pain 24.6% 22.2% Neurological - Neck 22.9% 25.9% Neurological - upper extremity sensory 37.4% 39.5% Shoulder pain 21.8% 25.9% Other pain 57.0% 58.0% Rates for dysphagia and dysphonia 11.2% 21.0% Local wound events 11.2% 22.2% Nonunion rate (ACDF only) - 4.9% Heterotopic ossification rate at index levels (Mobi-C only) 2.8% - Unanticipated adverse device effect 0.0% 0.0% 18-19 Group Associated AE(s) Secondary Surgical Intervention Detail Mobi-C (n=164) right C4-C5 radiculopathies reoperation - laparoscopic right C4-C5 cervical laminectomy radiculopathy and spondylosis removal of device and conversion to ACDF at index level ACDF (n=81) Foraminal stenosis and pseudoarthrosis at the index level supplemental fixation in the form of posterior instrumentation at index level Failure of fusion removal of ACDF hardware and repeat ACDF at the index level Misplaced screw removal of ACDF hardware and addition of ACDF at the adjacent level below Pseudarthrosis at the index level and herniated disc at the adjacent level above removal of ACDF hardware and repeat ACDF at the index level and addition of ACDF at the adjacent level above and addition of ACDF two levels above (three level ACDF) Pseudarthrosis at the index level and radiculopathy supplemental fixation in the form of posterior fusion instrumentation at the index level Secondary Surgical Interventions at the Index Level - Procedure DetailsCommonly Reported Adverse Events

www.ldrmedical.com www.cervicaldisc.com LDR, LDR Spine, LDR Médical, Avenue, BF+, BF+(Ph), Bi-Pack, C-Plate, Easyspine, Laminotome, L90, MC+, Mobi, Mobi-C, Mobi-L, Mobidisc, ROI, ROI-A, ROI-C, ROI-MC+, ROI-T, SpineTune and VerteBRIDGE are trademarks or registered trademarks of LDR Holding Corporation or its affiliates in France, the United States, and other countries. United States 13785 Research Boulevard Suite 200 Austin, Texas 78750 512.344.3333 France Hôtel de bureau 1 4, rue Gustave Eiffel 10 430 Rosières Près Troyes, FRANCE +33 (0)3 25 82 32 63 China Unit 06, Level 16, Building A, Beijing Global Trade Center #36 North Third Ring Road East, Dongcheng District, Beijing, China, 100013 +86 10 58256655 Brazil Av. Pereira Barreto, 1395-19º Floor Room 192 to 196 Torre Sul-Bairro Paraíso Santo André / São Paulo-Brazil CEP 09190-610 +55 11 4332 7755 re F: M b b 6 r eV A 08.201 3 Indications for Use: the Mobi-C® Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc at one level from C3-C7 following single-level discectomy for intractable radiculopathy (arm pain and/or a neurological deficit) with or without neck pain, or myelopathy due to a single-level abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (Ct, Mri, X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes), and/or visible loss of disc height compared to adjacent levels. the Mobi-C® Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least 6 weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C® Cervical Disc Prosthesis. Note: Please refer to the Mobi-C summary of safety and effectiveness Data (PMA P110002) at www.fda.gov for complete study results.