
Fourth Quarter 2018 Business Update 28 February 2019 Exhibit 99.2

Forward Looking Statements Safe Harbor Statement Some of the statements made in this presentation are forward-looking statements that involve a number of risks and uncertainties and are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward-looking statements are based upon Intrexon’s current expectations and projections about future events and generally relate to Intrexon’s plans, objectives and expectations for the development of Intrexon’s business, discussion of anticipated clinical trials and future collaborations. Although management believes that the plans and objectives reflected in or suggested by these forward-looking statements are reasonable, all forward-looking statements involve risks and uncertainties and actual future results may be materially different from the plans, objectives and expectations expressed in this presentation. These risks and uncertainties include, but are not limited to, (i) Intrexon’s strategy and overall approach to its business model and its ability to exercise more control and ownership over the development process and commercialization path; (ii) Intrexon’s ability to successfully enter new markets or develop additional products, whether with its collaborators or independently; (iii) Intrexon's ability to successfully enter into optimal strategic relationships with its subsidiaries and operating companies that it may form in the future; (iv) actual or anticipated variations in Intrexon’s operating results; (v) actual or anticipated fluctuations in Intrexon’s competitors’ or its collaborators’ operating results or changes in their respective growth rates; (vi) Intrexon’s cash position; (vii) market conditions in Intrexon’s industry; (viii) the volatility of Intrexon’s stock price; (ix) Intrexon’s ability, and the ability of its collaborators, to protect Intrexon’s intellectual property and other proprietary rights and technologies; (x) Intrexon’s ability, and the ability of its collaborators, to adapt to changes in laws or regulations and policies; (xi) the outcomes of pending and future litigation; (xii) the rate and degree of market acceptance of any products developed by Intrexon, its subsidiaries, collaborations or joint ventures; (xiii) Intrexon’s ability to retain and recruit key personnel; (xiv) Intrexon’s expectations related to the use of proceeds from its public offerings and other financing efforts; and (xv) Intrexon’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing. For a discussion of other risks and uncertainties, and other important factors, any of which could cause Intrexon’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in Intrexon’s Annual Report on Form 10-K and subsequent reports filed with the Securities and Exchange Commission. All information in this presentation is as of the date of the release, and Intrexon undertakes no duty to update this information unless required by law. © 2019 Intrexon Corp. All rights reserved. Intrexon Corporation is sharing the following materials for informational purposes only. Such materials do not constitute an offer to sell or the solicitation of an offer to buy any securities of Intrexon. Any offer and sale of Intrexon’s securities will be made, if at all, only upon the registration and qualification of such securities under all applicable federal and state securities laws or pursuant to an exemption from such requirements. The attached information has been prepared in good faith by Intrexon. However, Intrexon makes no representations or warranties as to the completeness or accuracy of any such information. Any representations or warranties as to Intrexon shall be limited exclusively to any agreements that may be entered into by Intrexon and to such representations and warranties as may arise under law upon distribution of any prospectus or similar offering document by Intrexon.

Financial Update
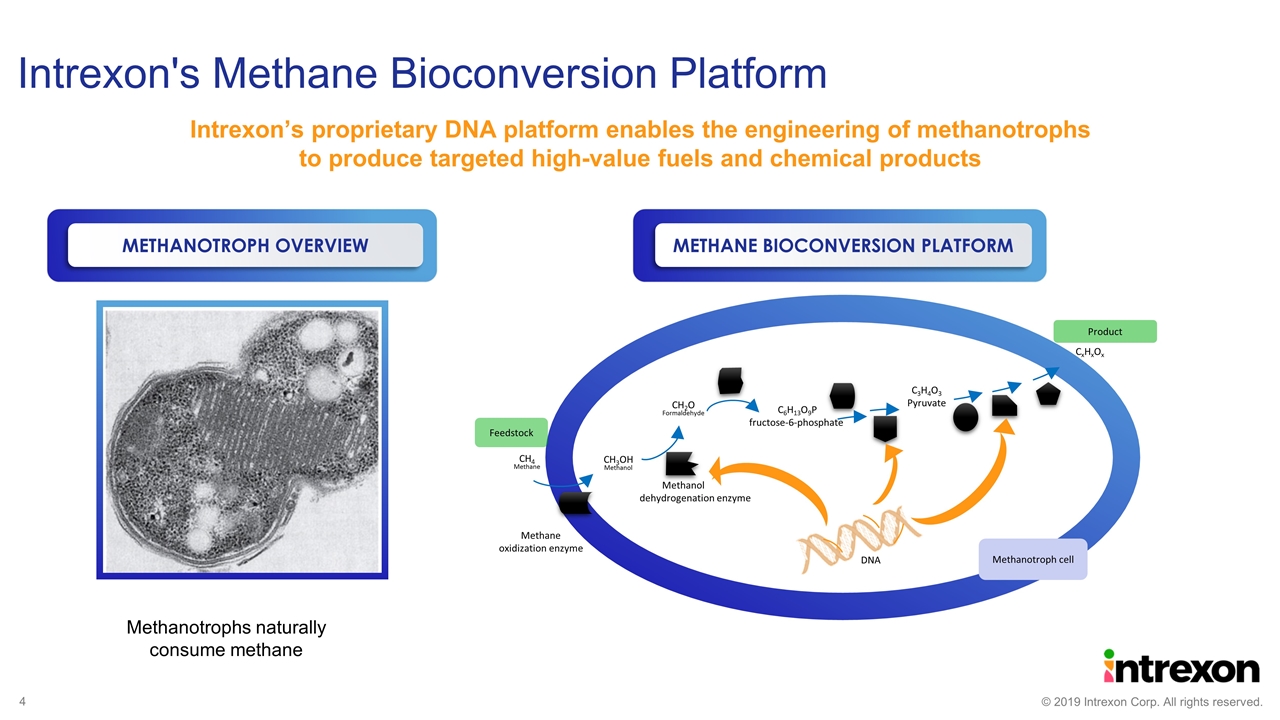
Intrexon's Methane Bioconversion Platform Methanotrophs naturally consume methane CH4 Methane CH3OH Methanol Methane oxidization enzyme CH2O Formaldehyde Methanol dehydrogenation enzyme C6H13O9P fructose-6-phosphate C3H4O3 Pyruvate CxHxOx Feedstock Product Methanotroph cell DNA Intrexon’s proprietary DNA platform enables the engineering of methanotrophs to produce targeted high-value fuels and chemical products METHANE BIOCONVERSION PLATFORM METHANOTROPH OVERVIEW
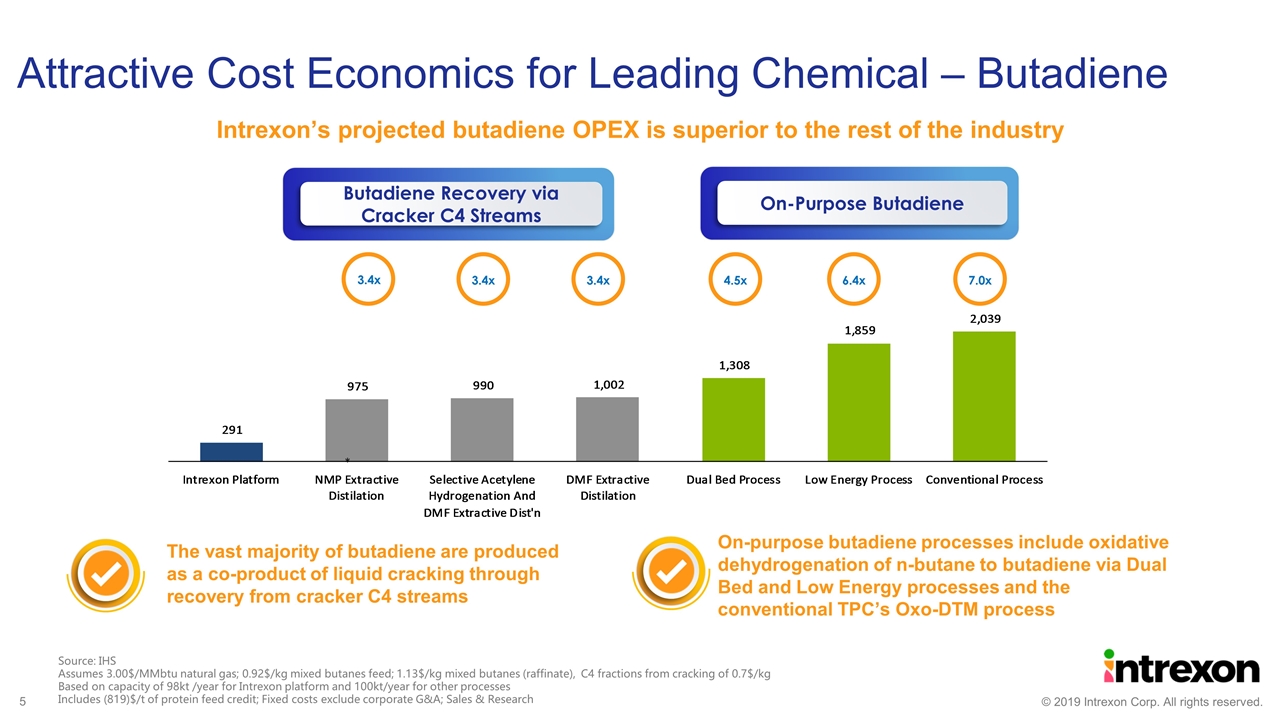
Attractive Cost Economics for Leading Chemical – Butadiene The vast majority of butadiene are produced as a co-product of liquid cracking through recovery from cracker C4 streams On-purpose butadiene processes include oxidative dehydrogenation of n-butane to butadiene via Dual Bed and Low Energy processes and the conventional TPC’s Oxo-DTM process * 3.4x 3.4x 3.4x 4.5x 6.4x 7.0x Source: IHS Assumes 3.00$/MMbtu natural gas; 0.92$/kg mixed butanes feed; 1.13$/kg mixed butanes (raffinate), C4 fractions from cracking of 0.7$/kg Based on capacity of 98kt /year for Intrexon platform and 100kt/year for other processes Includes (819)$/t of protein feed credit; Fixed costs exclude corporate G&A; Sales & Research Intrexon’s projected butadiene OPEX is superior to the rest of the industry On-Purpose Butadiene Butadiene Recovery via Cracker C4 Streams

Intrexon’s proprietary yeast strains enable a transformative process for robust production of cannabinoids with consistent yield and purity Platform designed to enable production of multiple target cannabinoids Robust Microbial Production of Cannabinoids for Medical Use
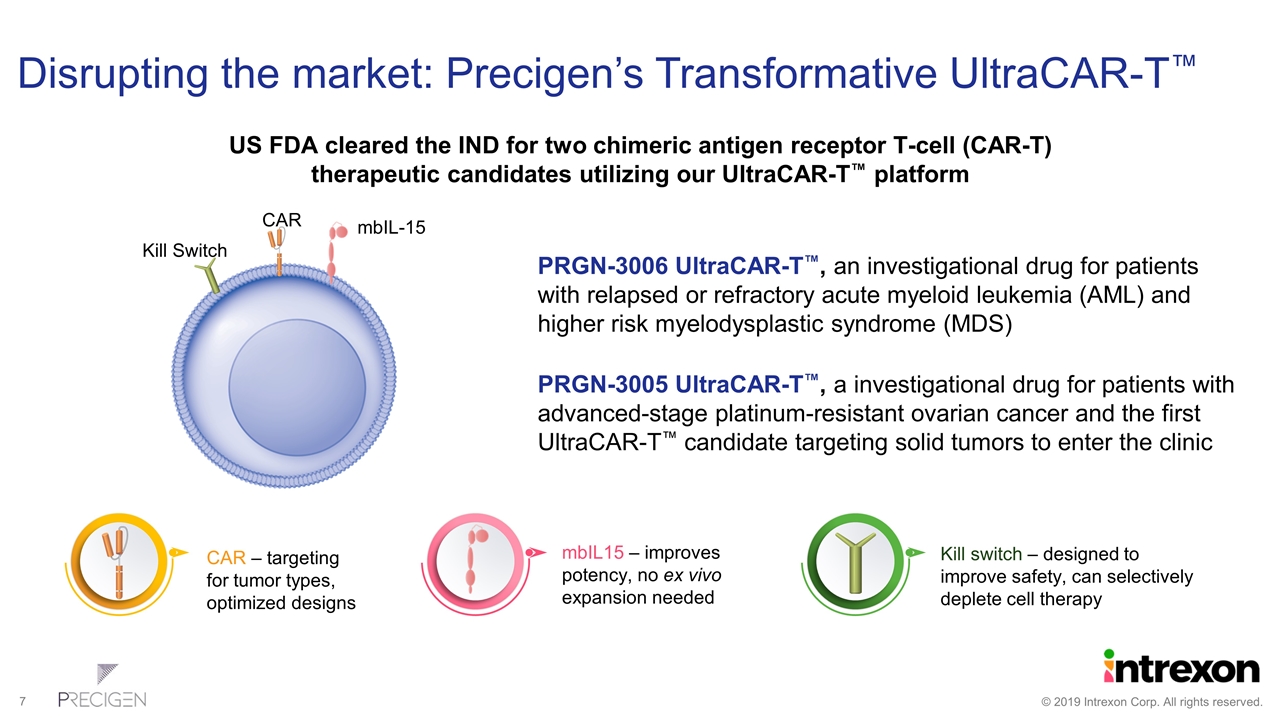
Disrupting the market: Precigen’s Transformative UltraCAR-T™ CAR mbIL-15 Kill Switch US FDA cleared the IND for two chimeric antigen receptor T-cell (CAR-T) therapeutic candidates utilizing our UltraCAR-T™ platform PRGN-3006 UltraCAR-T™, an investigational drug for patients with relapsed or refractory acute myeloid leukemia (AML) and higher risk myelodysplastic syndrome (MDS) PRGN-3005 UltraCAR-T™, a investigational drug for patients with advanced-stage platinum-resistant ovarian cancer and the first UltraCAR-T™ candidate targeting solid tumors to enter the clinic CAR – targeting for tumor types, optimized designs mbIL15 – improves potency, no ex vivo expansion needed Kill switch – designed to improve safety, can selectively deplete cell therapy

UltraCAR-T™ Platform Brings Key Advancements to CAR-T Cell Therapy Non-viral gene transfer using multigenic vectors for expression of multiple effector genes leads to better precision and control of tumor targeting and eliminates the need for virus Sustained persistence and desired phenotype of infused UltraCAR-T helps address T-cell exhaustion, a common issue with current CAR-T therapies T-cell control by incorporation of kill switch technology to potentially improve the safety profile Rapid manufacturing of UltraCAR-T™ cells using our proprietary non-viral gene transfer process, eliminates the need for ex vivo propagation, thus dramatically reducing potential wait times for patients from weeks to less than two days
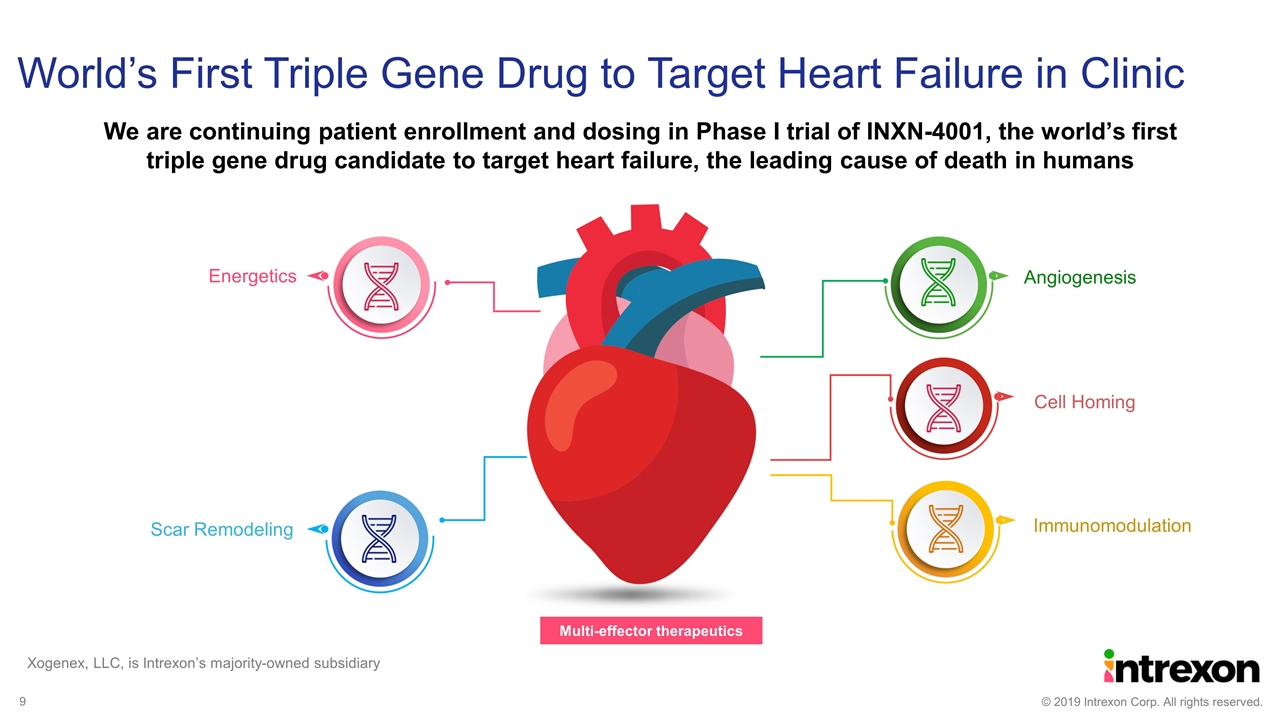
We are continuing patient enrollment and dosing in Phase I trial of INXN-4001, the world’s first triple gene drug candidate to target heart failure, the leading cause of death in humans World’s First Triple Gene Drug to Target Heart Failure in Clinic Angiogenesis Cell Homing Immunomodulation Scar Remodeling Energetics Multi-effector therapeutics Xogenex, LLC, is Intrexon’s majority-owned subsidiary
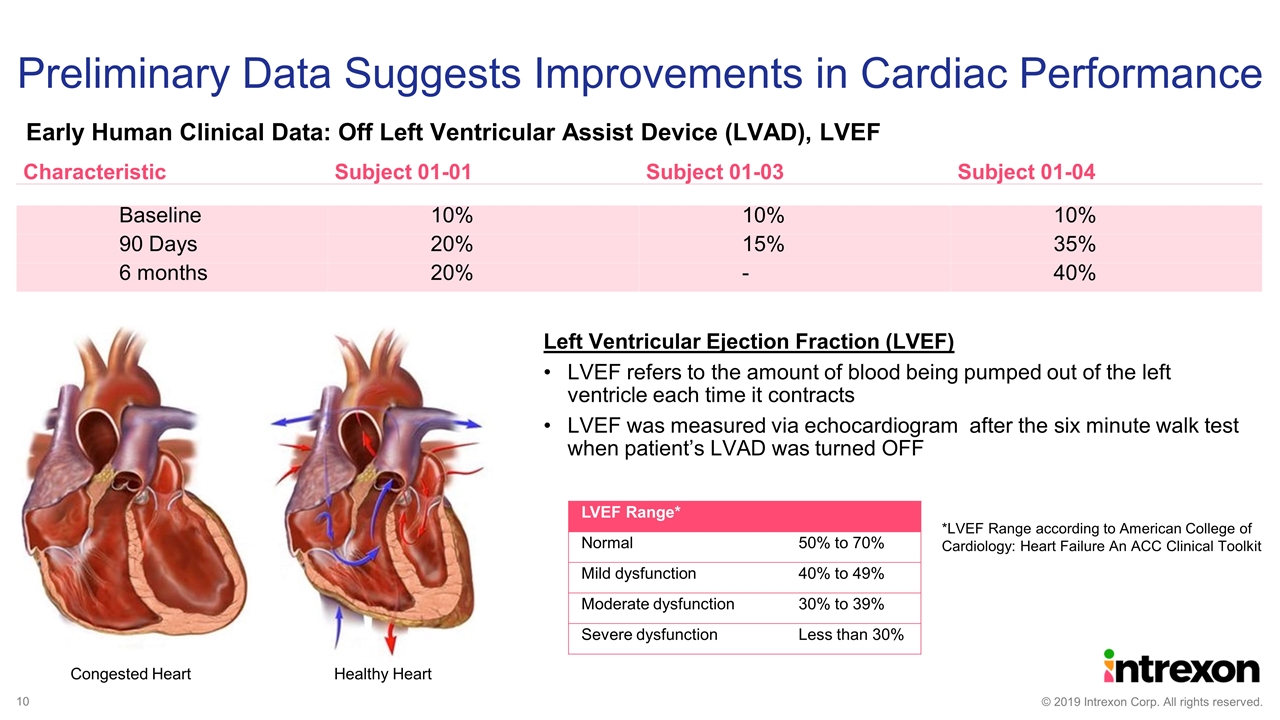
Preliminary Data Suggests Improvements in Cardiac Performance Characteristic Subject 01-01 Subject 01-03 Subject 01-04 Baseline 10% 10% 10% 90 Days 20% 15% 35% 6 months 20% - 40% Left Ventricular Ejection Fraction (LVEF) LVEF refers to the amount of blood being pumped out of the left ventricle each time it contracts LVEF was measured via echocardiogram after the six minute walk test when patient’s LVAD was turned OFF Congested Heart Healthy Heart LVEF Range* Normal 50% to 70% Mild dysfunction 40% to 49% Moderate dysfunction 30% to 39% Severe dysfunction Less than 30% *LVEF Range according to American College of Cardiology: Heart Failure An ACC Clinical Toolkit Early Human Clinical Data: Off Left Ventricular Assist Device (LVAD), LVEF
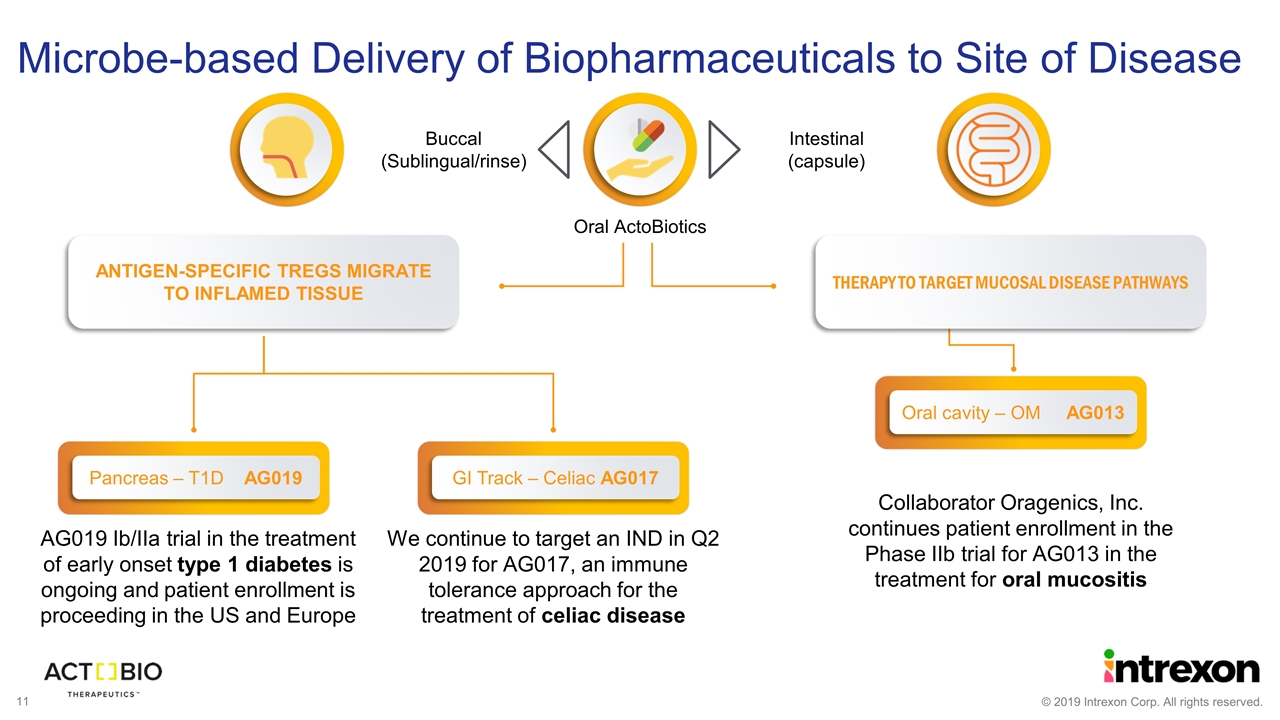
AG019 Ib/IIa trial in the treatment of early onset type 1 diabetes is ongoing and patient enrollment is proceeding in the US and Europe Collaborator Oragenics, Inc. continues patient enrollment in the Phase IIb trial for AG013 in the treatment for oral mucositis ANTIGEN-SPECIFIC TREGS MIGRATE TO INFLAMED TISSUE THERAPY TO TARGET MUCOSAL DISEASE PATHWAYS Oral cavity – OM AG013 Microbe-based Delivery of Biopharmaceuticals to Site of Disease Oral ActoBiotics Intestinal (capsule) Buccal (Sublingual/rinse) GI Track – Celiac AG017 We continue to target an IND in Q2 2019 for AG017, an immune tolerance approach for the treatment of celiac disease Pancreas – T1D AG019

Non-browning Arctic® Apples Available in Select Markets Okanagan Specialty Fruits’ (OSF) picked more than 2,100 bins of Arctic® apples in the 2018 harvest Select retailers are now selling fresh Arctic® sliced apples and ApBitz™ dehydrated apple snacks. ApBitz™ are also available on Amazon.com OSF is planning to plant up to 1,00,000 trees in spring of 2019; additional to the existing ~980,000 trees Consumer reception to the apples has been favorable

Bovine Embryos – Elite Bovine Genetics Technology Trans Ova Genetics is the preeminent leader of bovine genetics in North America: Focus on continuing to expand and improve herd genetics in support of embryo sales More than 575 heifers were added in the last year, with two Jersey heifers ranking #2 and #9 in the world* The herd also includes 15 of the world’s 37 top Holstein bulls based on an industry accepted dairy production index * Based on the most current Council of Dairy Cattle Breeding genomic testing results

Advancing Swine Models for Research and Regenerative Medicine Exemplar Genetics is developing porcine research models, which more accurately replicate human pathology as compared to traditional research models Demand for the MiniSwine Models continues to grow Expansion into regenerative medicine continues with Exemplar and the Mayo Clinic launching a joint venture, Cytotheryx, to develop high-quality source of human liver cells to advance medical research

Maysville commercial facility EnviroFlight’s black soldier fly (BSF) larvae holds considerable promise as an environmentally-friendly toxin-free, sustainable source of high-value nutrients for animal feed The largest black soldier fly larvae facility in the US opened in November 2018 Ability to produce 900 metric tons of product a year and is designed to scale up to 3,200 metric tons Commercial production is underway Orders for products from the new facility already account for ~1/3 of the anticipated annual output Black Soldier Fly Larvae Commercial Facility is Operational

AquAdvantage® Approach for Sustainable Aquaculture AquAdvantage® approach enables salmon to grow to market weight in half the time with less feed in land based systems AquaBounty Technologies, Inc. (NASDAQ: AQB) is Intrexon’s majority-owned subsidiary that pioneered the AquAdvantage® solution AquAdvantage® Salmon (AAS) currently distributed throughout Canada FDA approval to raise AAS at land-based Indiana facility; conventional salmon currently being raised at the facility USDA recently issued labeling rules; AAS awaits official labeling guidelines by the FDA; Prince Edward Island 250 metric ton production facility on track for Q2 2019 completion, allowing for immediate, large-scale production of AAS
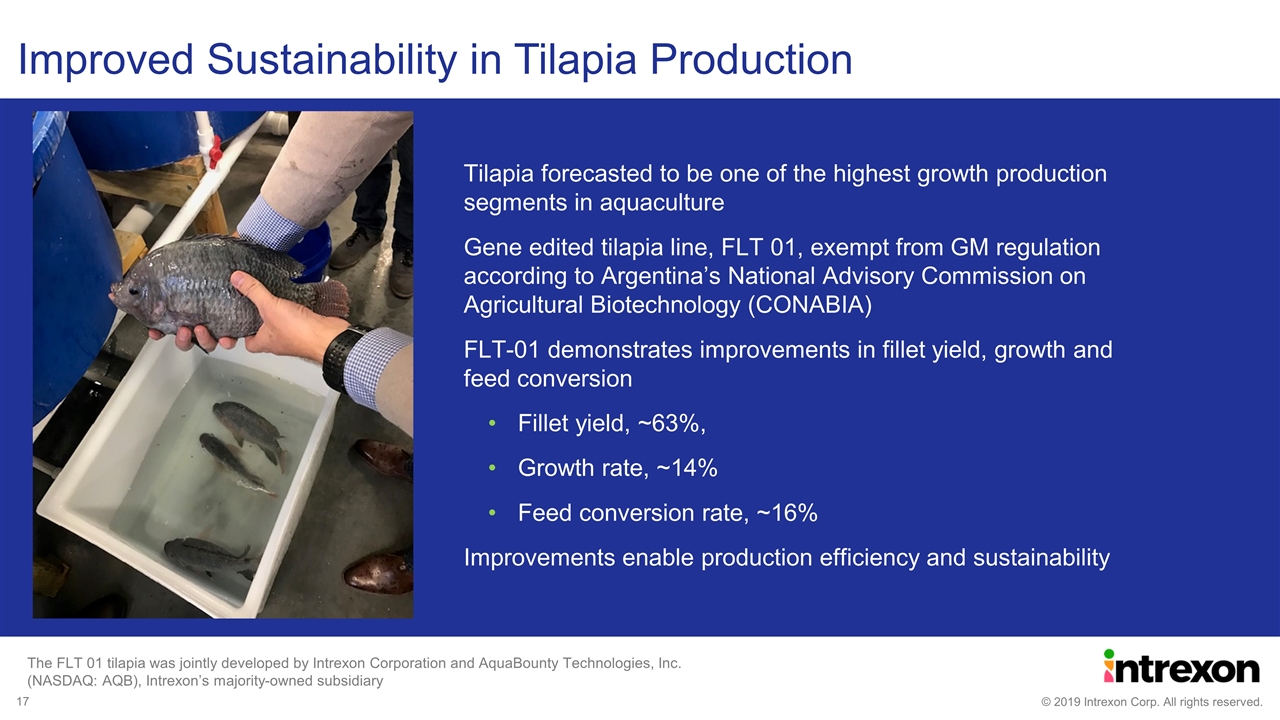
Improved Sustainability in Tilapia Production Tilapia forecasted to be one of the highest growth production segments in aquaculture Gene edited tilapia line, FLT 01, exempt from GM regulation according to Argentina’s National Advisory Commission on Agricultural Biotechnology (CONABIA) FLT-01 demonstrates improvements in fillet yield, growth and feed conversion Fillet yield, ~63%, Growth rate, ~14% Feed conversion rate, ~16% Improvements enable production efficiency and sustainability The FLT 01 tilapia was jointly developed by Intrexon Corporation and AquaBounty Technologies, Inc. (NASDAQ: AQB), Intrexon’s majority-owned subsidiary

Targeted species-specific method to suppress wild populations of disease-carrying mosquito species Next-gen Friendly™ Mosquitoes for Vector Control Transitioning to 2nd generation self-limiting, male-selecting OX5034 Friendly™ Aedes mosquito: Better economics than prior strains, without need for large-scale production facilities OX5034 in field trial in Indaiatuba, Brazil with government permission to test new release device prototypes in a second city Collaborative Agreements with the Bill & Melinda Gates Foundation to develop Friendly™ Anopheles: First strain targeting mosquito that spreads malaria in the Western hemisphere Second strain targeting mosquito that spreads malaria in India, Middle East and Horn of Africa
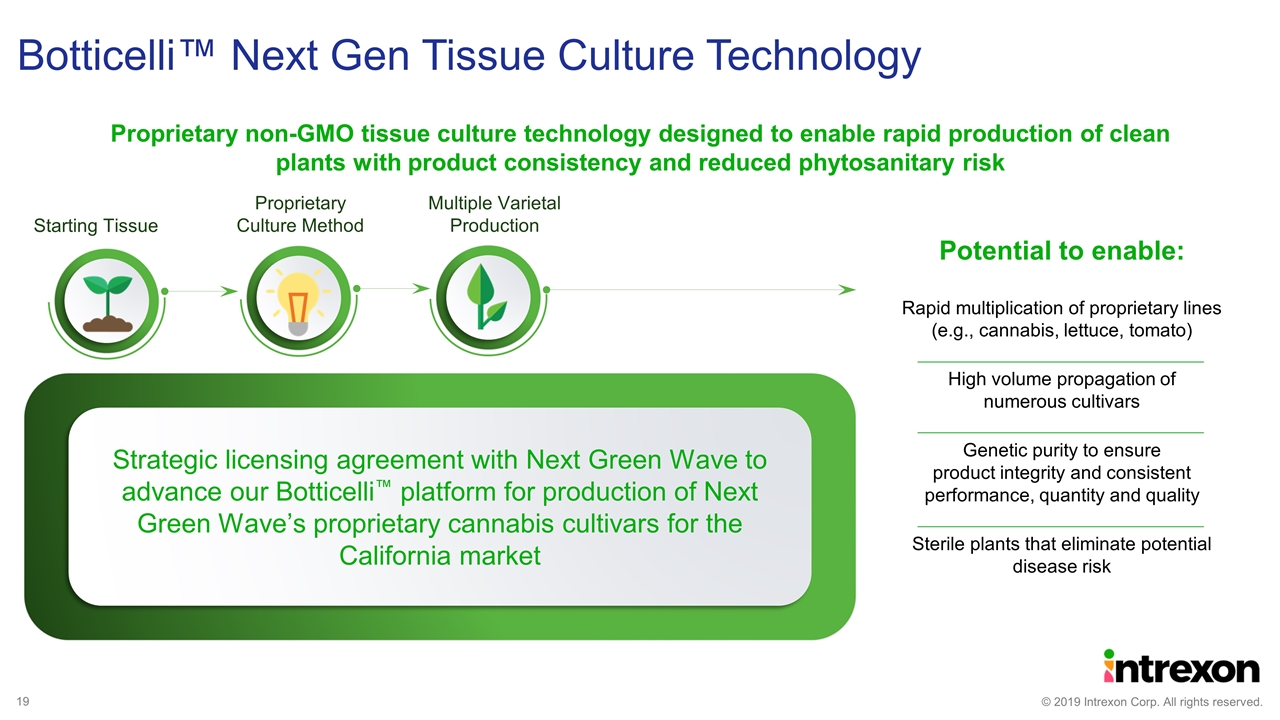
Proprietary non-GMO tissue culture technology designed to enable rapid production of clean plants with product consistency and reduced phytosanitary risk Botticelli™ Next Gen Tissue Culture Technology Potential to enable: Rapid multiplication of proprietary lines (e.g., cannabis, lettuce, tomato) High volume propagation of numerous cultivars Genetic purity to ensure product integrity and consistent performance, quantity and quality Sterile plants that eliminate potential disease risk Starting Tissue Proprietary Culture Method Multiple Varietal Production Strategic licensing agreement with Next Green Wave to advance our Botticelli™ platform for production of Next Green Wave’s proprietary cannabis cultivars for the California market

Diverse areas of expertise (investments across categories) Addressing real world challenges (solving unmet needs) Robust pipeline at various life stages (hedge against timing) World-first milestones (category defining solutions) Best and brightest scientists & engineers (most valued asset) Developing solutions to improve the quality of life & health of the planet Intrexon Overview

Q & A