
Q1 2023 Financial Results Update May 9, 2023

PROPRIETARY AND CONFIDENTIAL 2Q1 2023 Investor Update Introduction Bob Burrows Vice President, Investor Relations
This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical fact, including statements regarding the future performance of the Company or our business strategy, future operations, future financial position, future revenues and earnings, projected costs, prospects, plans and objectives of management and the ongoing impact of the COVID-19 pandemic, are forward-looking statements. We generally identify forward-looking statements by using words like “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “forecast,” “goal,” “intend,” “may,” “plan,” should,” “will,” “would,” and similar expressions or variations thereof, or the negative thereof, but these terms are not the exclusive means of identifying such statements. Forward-looking statements are based on our current intentions, beliefs and expectations regarding future events. We cannot guarantee that any forward-looking statement will be accurate. You should realize that if underlying assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results could differ materially from our expectations. You are, therefore, cautioned not to place undue reliance on any forward-looking statement. Any forward-looking statement speaks only as of the date on which such statement is made, and, except as required by law we do not undertake to update any forward-looking statement to reflect new information, events or circumstances. There are a number of important factors that could cause our actual results to differ materially from those indicated by such forward-looking statements, including, among others, the availability of USG funding for contracts related to procurement of our medical countermeasures, including AV7909 (Anthrax Vaccine Adsorbed (AVA), Adjuvanted), BioThrax® (Anthrax Vaccine Adsorbed) and ACAM2000®, (Smallpox (Vaccinia) Vaccine, Live), among others, as well as contracts related to development of medical countermeasures; our ability to meet our commitments to quality and compliance in all of our manufacturing operations; our ability to negotiate additional USG procurement or follow-on contracts for our medical countermeasures products that have expired or will be expiring; the commercial availability, including the timing of availability, of over-the-counter NARCAN® (naloxone HCI) Nasal Spray; the impact of the generic marketplace on NARCAN® (naloxone HCI) Nasal Spray and future NARCAN sales; our ability to perform under our contracts with the USG, including the timing of and specifications relating to deliveries; our ability to provide CDMO services for the development and/or manufacture of product and/or product candidates of our customers at required levels and on required timelines; the ability of our contractors and suppliers to maintain compliance with current good manufacturing practices and other regulatory obligations; our ability to negotiate new CDMO contracts and the negotiation of further commitments related to the collaboration and deployment of capacity toward future commercial manufacturing under our existing CDMO contracts; our ability to collect reimbursement for raw materials and payment of services fees from our CDMO customers; the results of pending shareholder litigation and government investigations and their potential impact on our business; our ability to comply with the operating and financial covenants required by our senior secured credit facilities and the amended and restated credit agreement relating to such facilities, and our 3.875% Senior Unsecured Notes due 2028; our ability to refinance our senior secured credit facilities prior to their maturity in October 2023; the procurement of our product candidates by USG entities under regulatory authorities that permit government procurement of certain medical products prior to U.S. Food and Drug Administration marketing authorization, and corresponding procurement by government entities outside of the United States; the full impact of the COVID-19 pandemic on our markets, operations and employees as well as those of our customers and suppliers; the impact on our revenues from and duration of declines in sales of our vaccine products that target travelers due to the reduction of international travel caused by the COVID-19 pandemic; the ability of the Company and Bavarian Nordic to consummate the transactions contemplated under the agreement pursuant to which we agreed to sell our travel health business, to meet expectations regarding the conditions, timing and completion of the transactions, and to realize the potential benefits of the transactions; the impact of the organizational changes we announced in January 2023 on our business; our ability to identify and acquire companies, businesses, products or product candidates that satisfy our selection criteria; the impact of cyber security incidents, including the risks from the interruption, failure or compromise of our information systems or those of our business partners, collaborators or other third parties; the success of our commercialization, marketing and manufacturing capabilities and strategy; and the accuracy of our estimates regarding future revenues, expenses, capital requirements and needs for additional financing. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from our expectations in any forward-looking statement. When evaluating our forward-looking statements, you should consider this cautionary statement along with the risks identified in our reports filed with the SEC. New factors emerge from time to time and it is not possible for management to predict all such factors, nor can it assess the impact of any such factor on the business or the extent to which any factor, or combination of factors, may cause results to differ materially from those contained in any forward-looking statement. Trademarks Emergent,® BioThrax® (Anthrax Vaccine Adsorbed), RSDL® (Reactive Skin Decontamination Lotion Kit), BAT® (Botulism Antitoxin Heptavalent (A,B,C,D,E,F and G)-(Equine)), Anthrasil® (Anthrax Immune Globulin Intravenous (Human)), VIGIV (Vaccinia Immune Globulin Intravenous (Human)), Trobigard® (atropine sulfate, obidoxime chloride), ACAM2000® (Smallpox (Vaccinia) Vaccine, Live), Vivotif® (Typhoid Vaccine Live Oral Ty21a), Vaxchora® (Cholera Vaccine, Live, Oral), NARCAN® (naloxone HCI) Nasal Spray, TEMBEXA® (brincidofovir) and any and all Emergent BioSolutions Inc. brands, products, services and feature names, logos and slogans are trademarks or registered trademarks of Emergent BioSolutions Inc. or its subsidiaries in the United States or other countries. All other brands, products, services and feature names or trademarks are the property of their respective owners. 3Q1 2023 Investor Update Safe Harbor Statement/Trademarks INTRODUCTION

This presentation contains financial measures (Adjusted Net Income (Loss), Adjusted Net Income (Loss) per Diluted Shares, Adjusted EBITDA (Earnings Before Interest, Taxes, Depreciation and Amortization), Adjusted Gross Margin, Adjusted Gross Margin %, Adjusted Revenues, and Adjusted Cost of Sales) that are considered “non-GAAP” financial measures under applicable Securities and Exchange Commission rules and regulations. These non-GAAP financial measures should be considered supplemental to and not a substitute for financial information prepared in accordance with generally accepted accounting principles. The Company’s definition of these non-GAAP measures may differ from similarly titled measures used by others. For its non-GAAP measures, the Company adjusts for specified items that can be highly variable or difficult to predict, or reflect the noncash impact of charges or accounting changes. As needed, such adjustments are tax effected utilizing the federal statutory tax rate for the U.S., except for changes in the fair value of contingent consideration as the vast majority is non-deductible for tax purposes. The Company views these non-GAAP financial measures as a means to facilitate management’s financial and operational decision-making, including evaluation of the Company’s historical operating results and comparison to competitors’ operating results. These non-GAAP financial measures reflect an additional way of viewing aspects of the Company’s operations that, when viewed with GAAP results and the reconciliations to the corresponding GAAP financial measure, may provide a more complete understanding of factors and trends affecting the Company’s business. For more information on these non-GAAP financial measures, please see the tables captioned "Reconciliation of Net Loss and Net Loss per Diluted Share to Adjusted Net Income (Loss) and Adjusted Net Income (Loss) per Diluted Share," "Reconciliation of Net Income Loss to Adjusted EBITDA," and "Reconciliation of Total Revenues to Adjusted Revenues, Cost of Sales to Adjusted Cost of Sales, and Gross Margin and Gross Margin % to Adjusted Gross Margin and Adjusted Gross Margin %" included at the end of this presentation. The determination of the amounts that are excluded from these non-GAAP financial measures are a matter of management judgment and depend upon, among other factors, the nature of the underlying expense or income amounts. Because non-GAAP financial measures exclude the effect of items that will increase or decrease the Company’s reported results of operations, management strongly encourages investors to review the Company’s consolidated financial statements and publicly filed reports in their entirety. For additional information on the non-GAAP financial measures noted here, please refer to the reconciliation tables provide in the Appendix to this presentation as well as the associated press release which can be found on the Company’s website at www.emergentbiosolutions.com. 4Q1 2023 Investor Update Non-GAAP Financial Measures INTRODUCTION

Agenda 5Q1 2023 Investor Update INTRODUCTION Item Presenter Topic(s) 1 Bob Kramer President and CEO • State of the Company 2 Paul Williams SVP, Products Business • State of the NARCAN Nasal Spray Franchise 3 Rich Lindahl EVP, CFO and Treasurer • Q1 2023 vs. Q1 2022 • FY 2023 Guidance • Q2 2023 Guidance 4 Q&A

PROPRIETARY AND CONFIDENTIALQ1 2023 Investor Update State of the Company Bob Kramer President and Chief Executive Officer 6

Overview of Q1 2023 7Q1 2023 Investor Update STATE OF THE COMPANY • Q1 2023 performance in-line with expectations • Revenues above our guidance range • Operating costs higher than expected • Period profitability negatively impacted • Remain committed to continuing to take actions aimed at strengthening our core business and building a foundation for sustainable, long-term growth

Current Status of 2023 Priorities 8Q1 2023 Investor Update STATE OF THE COMPANY 1. Expect sale of Travel Health business to Bavarian Nordic to close in the second quarter; $270M upfront + up to $110M in sales and development-based milestones 2. Secured NARCAN Nasal Spray approval by FDA as first and only opioid overdose reversal treatment available over the counter; remain focused on expanding access and maintaining affordability; targeting late summer launch 3. Continue to support longstanding partnership with the US government focused on preparedness and response against public health threats – i) three notices of intent to procure from USG for ACAM2000, VIGIV and BAT; ii) AV7909 BLA tracking to July 2023 PDUFA 4. Continue to implement strategy of delivering and strengthening our quality and compliance culture and systems 5. Actively managing our business and capital structure

9Q1 2023 Investor Update State of the NARCAN Franchise Paul Williams Senior Vice President, Products Business

2023 and Future Priorities 10Q1 2023 Investor Update STATE OF THE NARCAN FRANCHISE • FDA approval of NARCAN OTC (3/29/23) – opportunity to expand availability through pharmacies, convenience stores, vending machines, online retailers…anywhere OTC products are found • Meeting with retail partners, finalizing contracts and distribution plans; continuing to support and advocate for potential insurance coverage for Medicare/Medicaid patients • Confident we have sufficient supply chain capacity to fulfill demand; goal of late summer for OTC product available • Committed to working with public interest partners, as they are and will continue to be a critical point of access for NARCAN • Continue to seek markets outside US, namely Canada • Further evaluating making NARCAN as widely accessible as possible +44M 4mg Doses Distributed in US/Canada (since 02/2016) <$50 (<$25/dose) Target retail price for carton of two 4mg. doses

1 1Q1 2023 Investor Update Rich Lindahl Executive Vice President and Chief Financial Officer Financials

Q1 2023 Key Financial Highlights 12Q1 2023 Investor Update FINANCIALS 1. Anticipate divestiture of Travel Health business to close in second quarter; implications incorporated into updated 2023 guidance 2. Continued evidence of robust demand for NARCAN from both US PIP and Canadian channels; significant increase in NARCAN FY 2023 guidance 3. Important evidence of continued USG preparedness – recent issuances of ACAM, VIGIV and BAT notices of intent to procure 4. CDMO FY 2023 guidance adjusted down; unwinding of COVID work plus ongoing remediation costs and investments to improve, strengthen quality and compliance network wide 5. Final stages of reaching agreement with bank group on credit facility amend/extend; anticipate closing on or before May 17
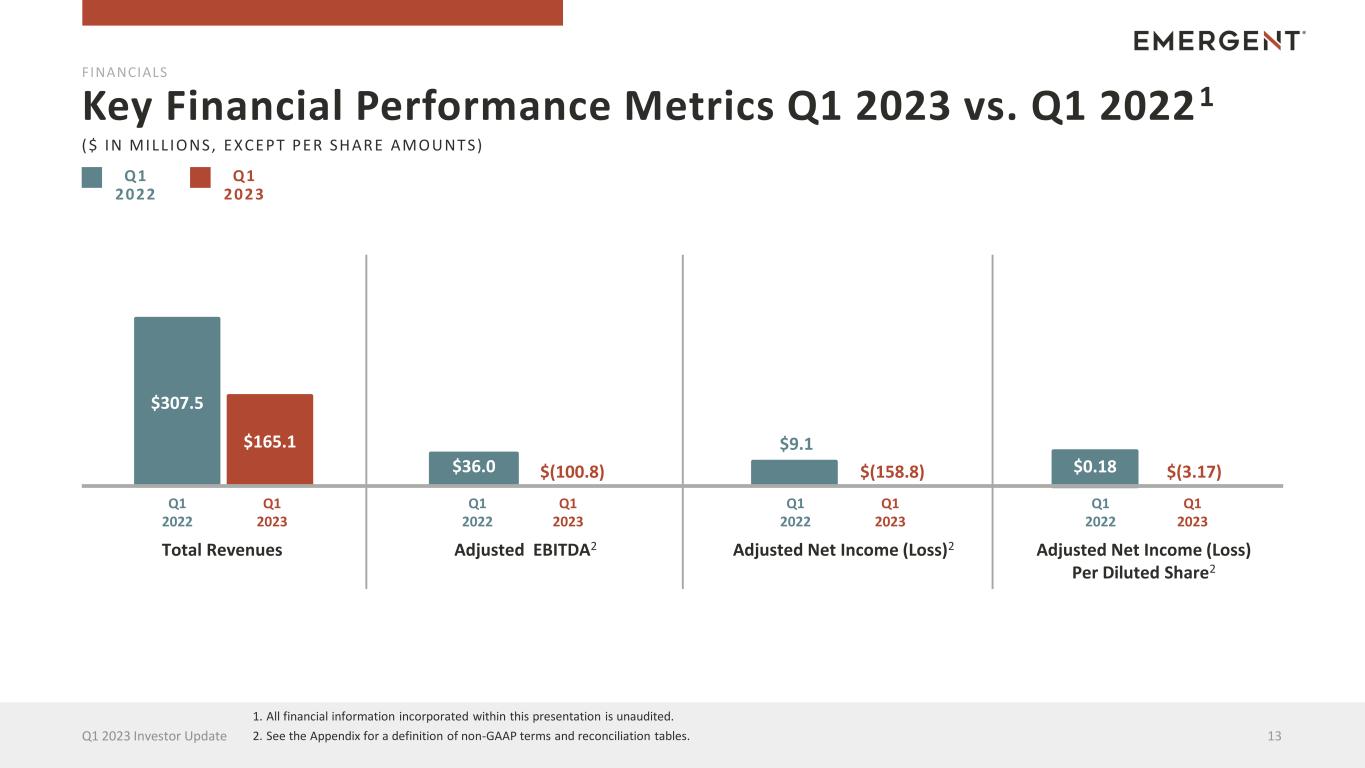
Q1 2022 Key Financial Performance Metrics Q1 2023 vs. Q1 20221 13Q1 2023 Investor Update Total Revenues Adjusted EBITDA2 Adjusted Net Income (Loss) Per Diluted Share2 Adjusted Net Income (Loss)2 $307.5 $165.1 $0.18 FINANCIALS $36.0 $9.1 ($ IN MILL IONS, EXCEPT PER SHARE AMOUNTS) Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 1. All financial information incorporated within this presentation is unaudited. 2. See the Appendix for a definition of non-GAAP terms and reconciliation tables. $(3.17)$(158.8)$(100.8)
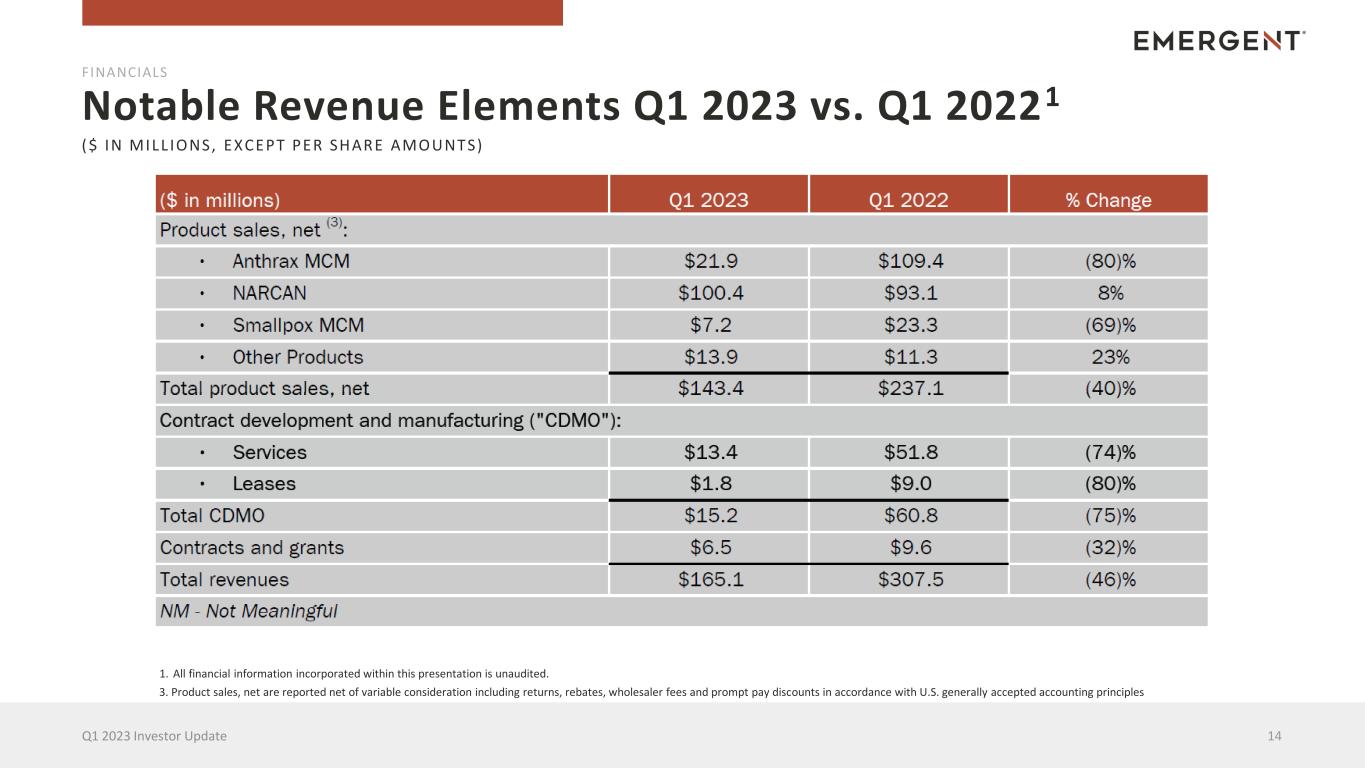
Notable Revenue Elements Q1 2023 vs. Q1 20221 14Q1 2023 Investor Update FINANCIALS ($ IN MILL IONS, EXCEPT PER SHARE AMOUNTS) 1. All financial information incorporated within this presentation is unaudited. 3. Product sales, net are reported net of variable consideration including returns, rebates, wholesaler fees and prompt pay discounts in accordance with U.S. generally accepted accounting principles
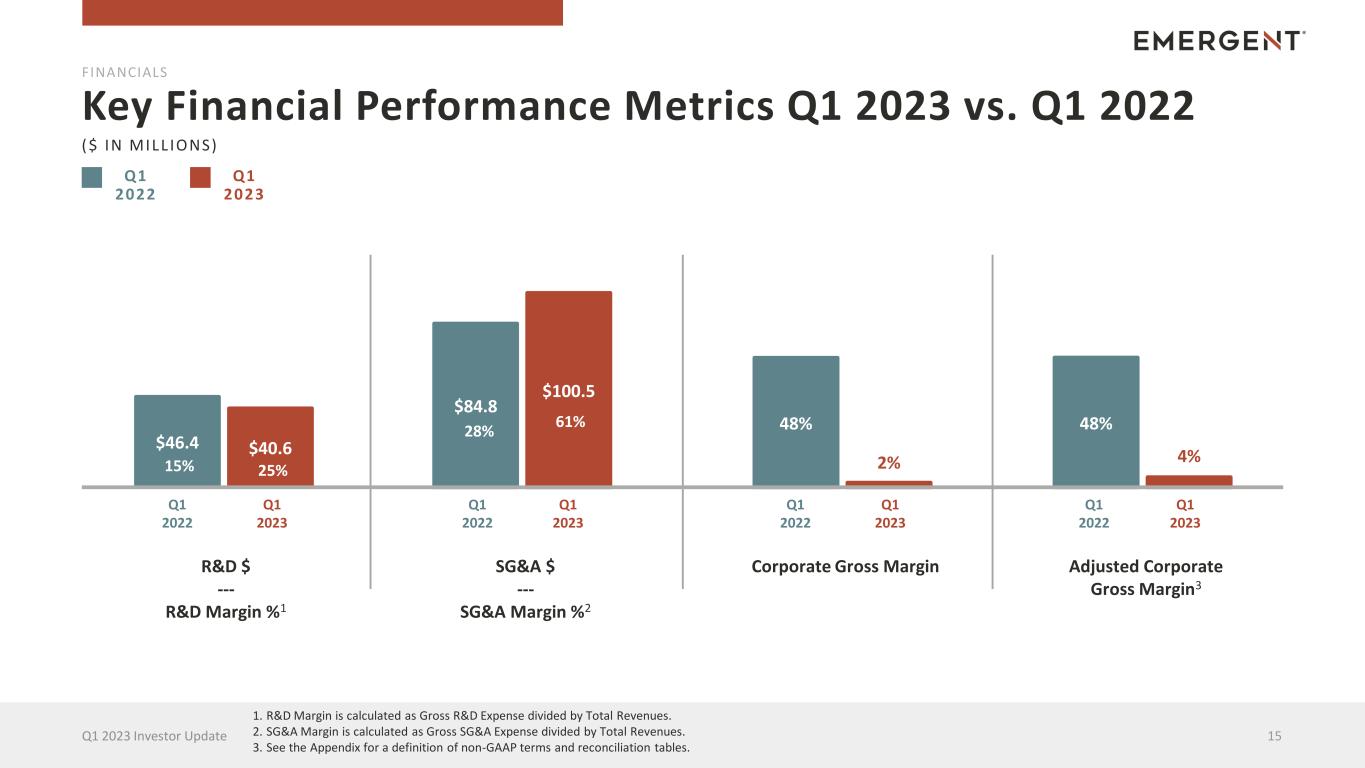
Q1 2022 Key Financial Performance Metrics Q1 2023 vs. Q1 2022 15Q1 2023 Investor Update R&D $ --- R&D Margin %1 SG&A $ --- SG&A Margin %2 Adjusted Corporate Gross Margin3 Corporate Gross Margin $46.4 $40.6 FINANCIALS $84.8 $100.5 48% 2% ($ IN MILL IONS) Q1 2023 48% 4% Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 15% 25% 28% 61% 1. R&D Margin is calculated as Gross R&D Expense divided by Total Revenues. 2. SG&A Margin is calculated as Gross SG&A Expense divided by Total Revenues. 3. See the Appendix for a definition of non-GAAP terms and reconciliation tables.
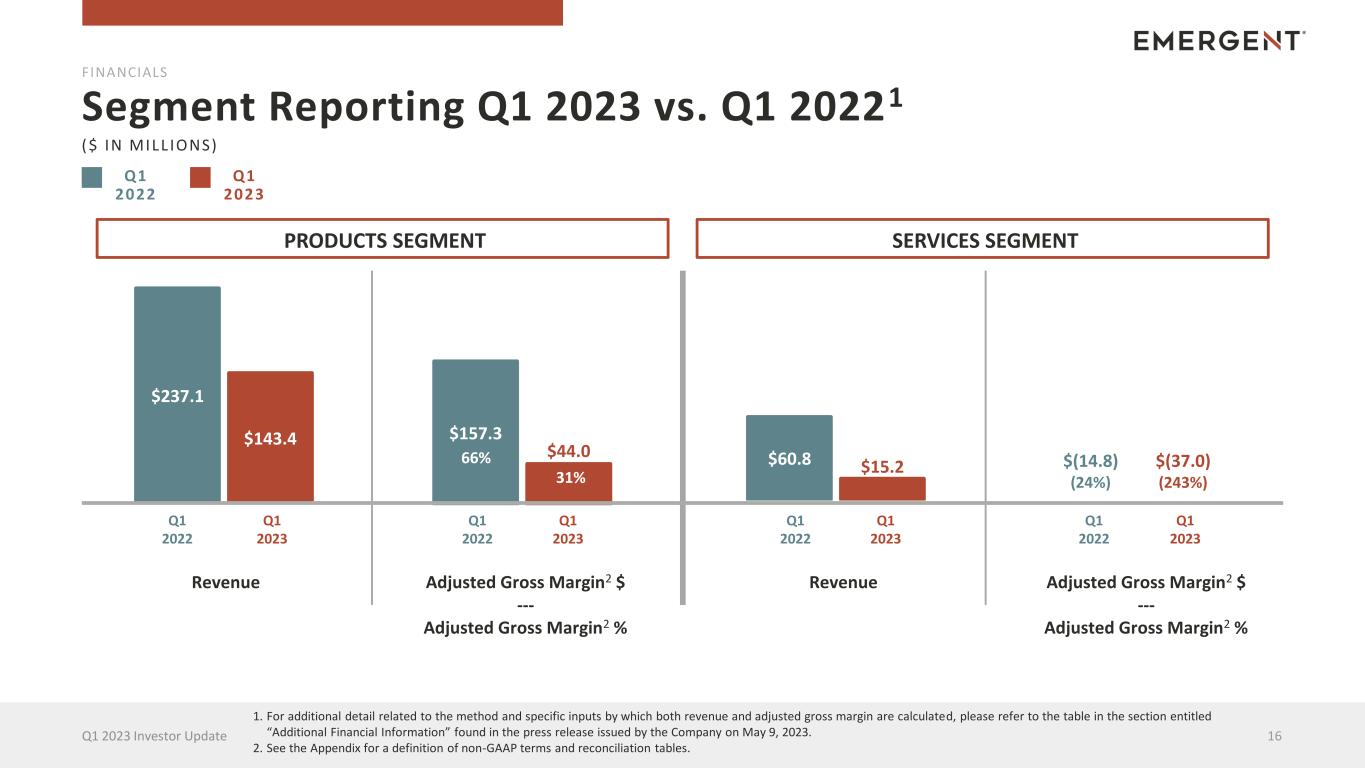
Q1 2022 Segment Reporting Q1 2023 vs. Q1 20221 16Q1 2023 Investor Update Revenue Adjusted Gross Margin2 $ --- Adjusted Gross Margin2 % Adjusted Gross Margin2 $ --- Adjusted Gross Margin2 % Revenue $237.1 $143.4 FINANCIALS $157.3 $44.0 ($ IN MILL IONS) Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 Q1 2022 Q1 2023 PRODUCTS SEGMENT SERVICES SEGMENT 66% 31% $60.8 $15.2 $0.0 $(37.0) (243%) $(14.8) (24%) 1. For additional detail related to the method and specific inputs by which both revenue and adjusted gross margin are calculated, please refer to the table in the section entitled “Additional Financial Information” found in the press release issued by the Company on May 9, 2023. 2. See the Appendix for a definition of non-GAAP terms and reconciliation tables.

Balance Sheet & Cash Flow Metrics 17Q1 2023 Investor Update FINANCIALS As of March 31, 2023 For the Three Months Ended March 31, 2023 C A S H $430.2 A C C O U N T S R E C E I V A B L E $155.9 N E T D E B T P O S I T I O N 1 , 2 $975.0 O P E R A T I N G C A S H F L O W $(184.0) C A P I T A L E X P E N D I T U R E S $15.1 ($ IN MILL IONS) 1. Debt amount indicated on the Company’s balance sheet is net of unamortized debt issuance costs of $7.0M. 2. Net Debt is calculated as Total Debt minus Cash ($1,405M - $430M = $975M).
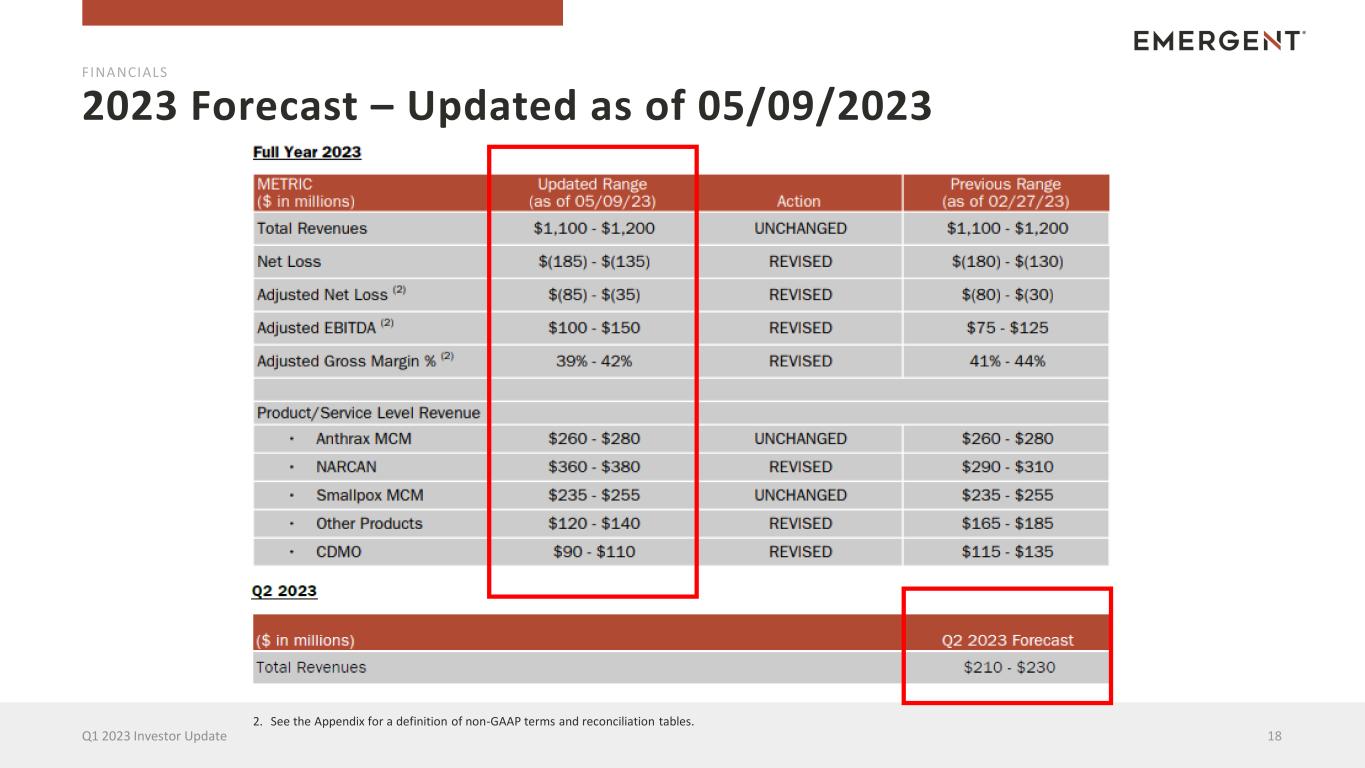
2023 Forecast – Updated as of 05/09/2023 18Q1 2023 Investor Update FINANCIALS 2. See the Appendix for a definition of non-GAAP terms and reconciliation tables.

2023 Forecast – Key Assumptions 19Q1 2023 Investor Update FINANCIALS Revenues and profits in 2023 are expected to be weighted towards the second half of the year • OVERALL — Reflects the impact of the previously announced sale of the Travel Health business to Bavarian Nordic, which is anticipated to close in the second quarter. • Total Revenues — Unchanged, reflecting the neutral impact of the overall updates across all sources of revenues. • Anthrax MCM — Unchanged, reflecting assumptions that have remained constant regarding procurement and delivery of the Company's related products to the U.S. and allied governments. • NARCAN — Revised, primarily reflecting robust demand from the U.S. PIP (public interest) channel and Canadian market. • Smallpox MCM — Unchanged, reflecting assumptions that have remained constant regarding procurement and delivery of the Company's related products to the U.S. and allied governments. • Other Products — Revised, reflecting the removal of the Travel Health products, Vaxchora and Vivotif, following the anticipated completion of the divestiture of this business. • CDMO — Revised, reflecting the impact of recent changes to customer requirements for COVID-related products coupled with continued remediation costs and investments to improve quality and compliance across the Company's manufacturing network. • Adjusted Net Loss — Revised, reflecting the impact of higher NARCAN sales and the Travel Health business divestiture, offset by lower CDMO revenues and an increase in the tax valuation allowance. • Adjusted EBITDA — Revised, reflecting the impact of higher NARCAN sales and the Travel Health business divestiture, offset by lower CDMO revenues. • Adjusted Gross Margin — Revised, reflecting the impact of overall revenue mix.

Summary Comments 20Q1 2023 Investor Update FINANCIALS • First quarter results reflect mix of strong performance in certain core areas offset by ongoing challenges • Remain committed to sustaining revenue growth and improving profitability • Continue to address near term challenges to our credit profile • Remain confident in the impact we are having on patients and customers focused on health security and pandemic preparedness

PROPRIETARY AND CONFIDENTIAL 21Q1 2023 Investor Update Q&A

22 Appendix Q1 2023 Investor Update
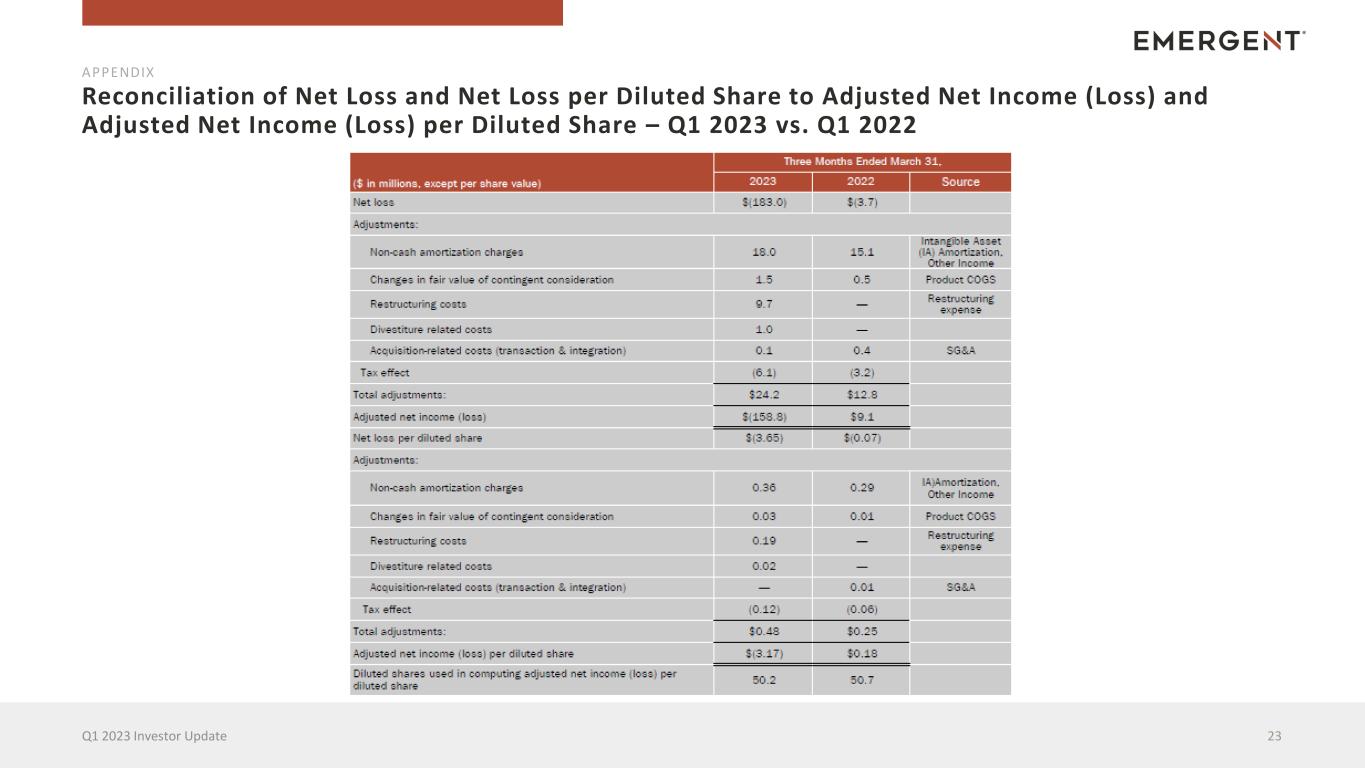
Reconciliation of Net Loss and Net Loss per Diluted Share to Adjusted Net Income (Loss) and Adjusted Net Income (Loss) per Diluted Share – Q1 2023 vs. Q1 2022 23Q1 2023 Investor Update APPENDIX
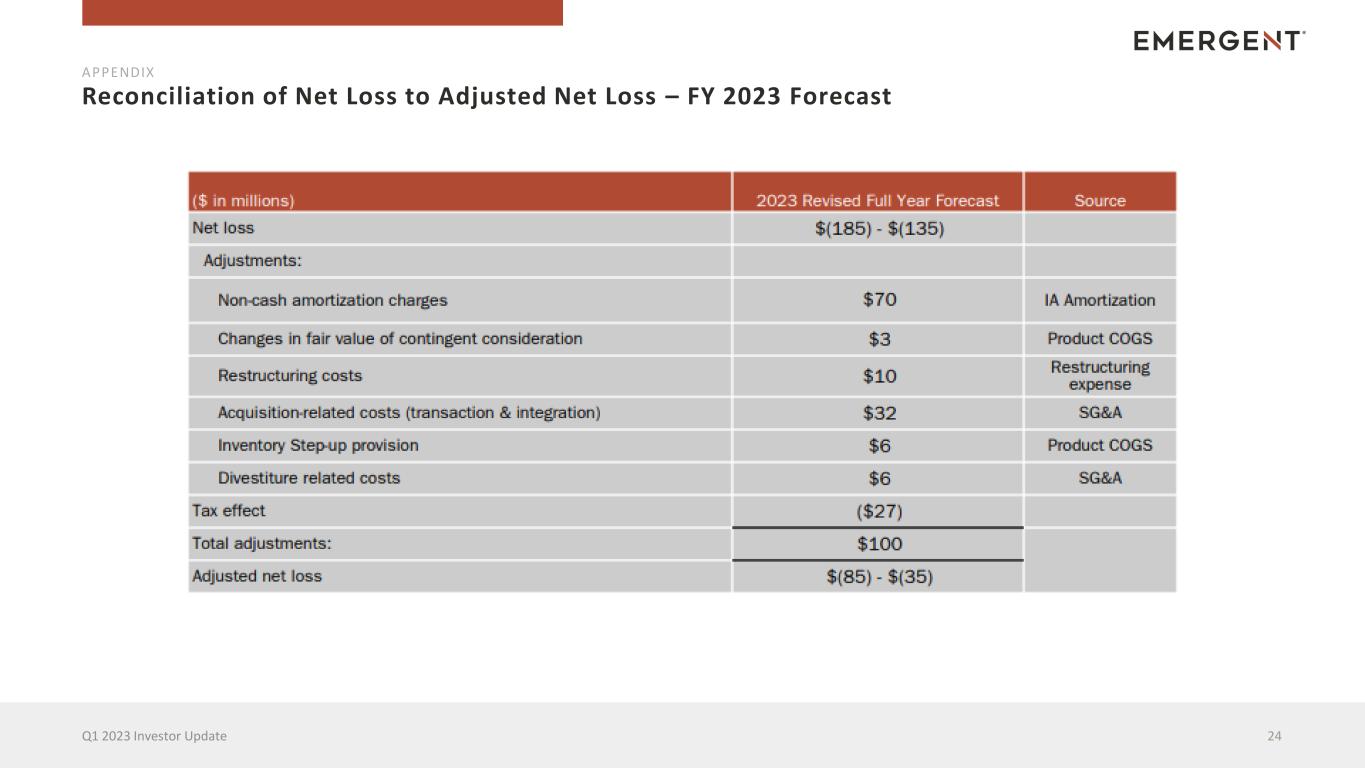
Reconciliation of Net Loss to Adjusted Net Loss – FY 2023 Forecast 24Q1 2023 Investor Update APPENDIX
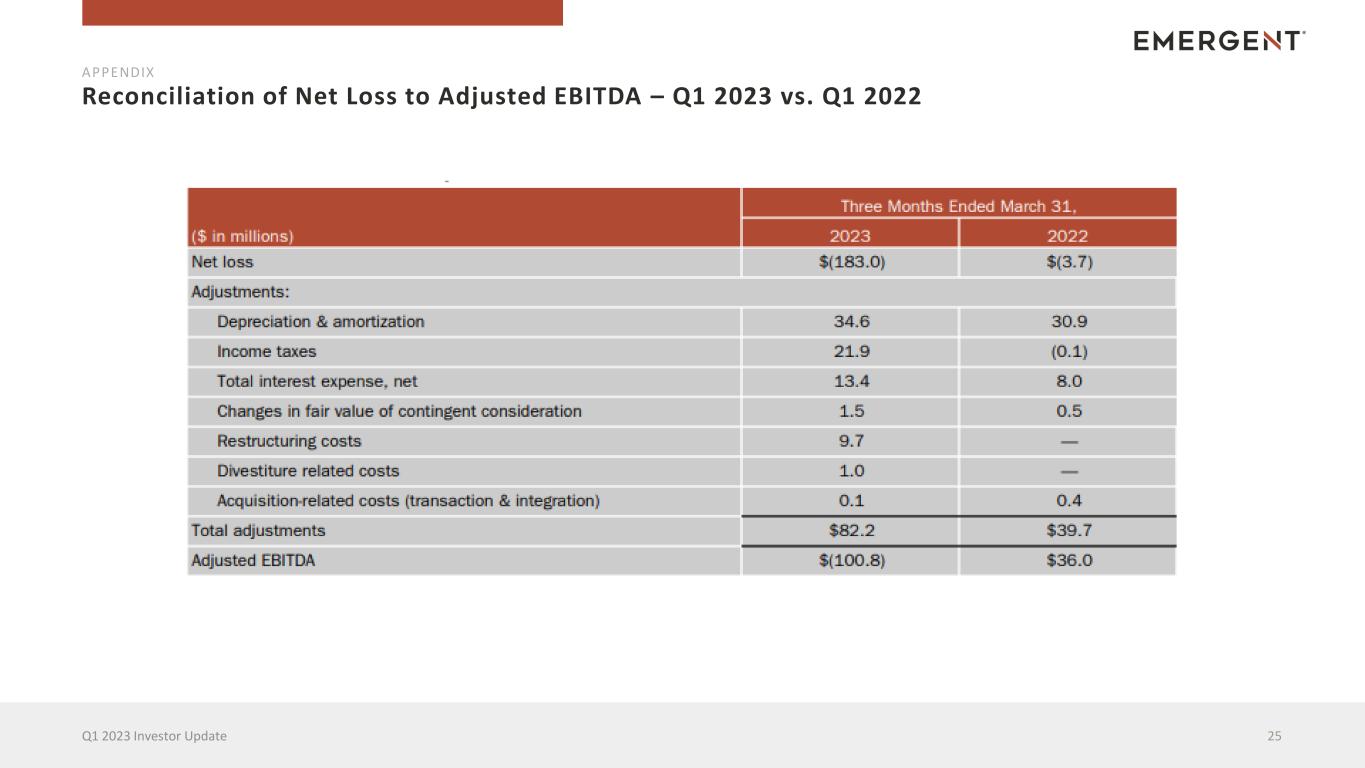
Reconciliation of Net Loss to Adjusted EBITDA – Q1 2023 vs. Q1 2022 25Q1 2023 Investor Update APPENDIX
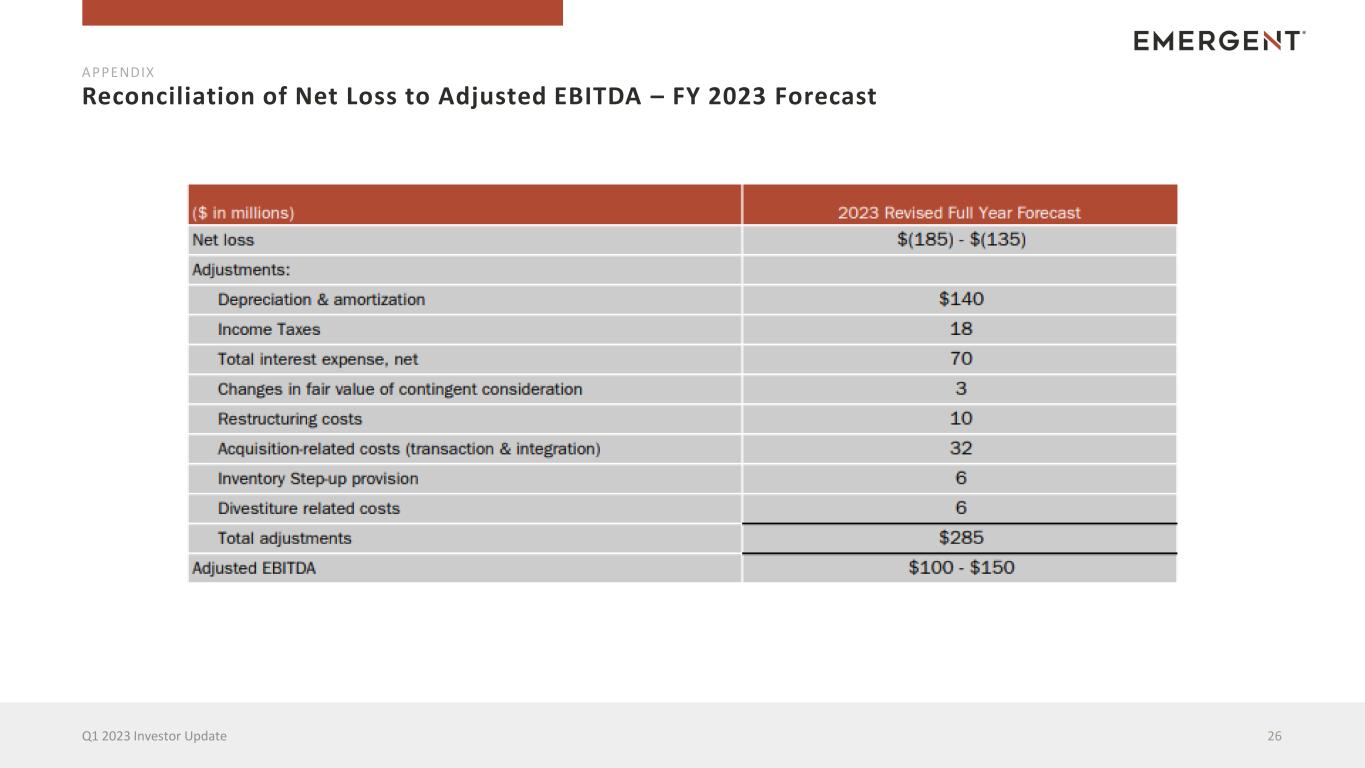
Reconciliation of Net Loss to Adjusted EBITDA – FY 2023 Forecast 26Q1 2023 Investor Update APPENDIX
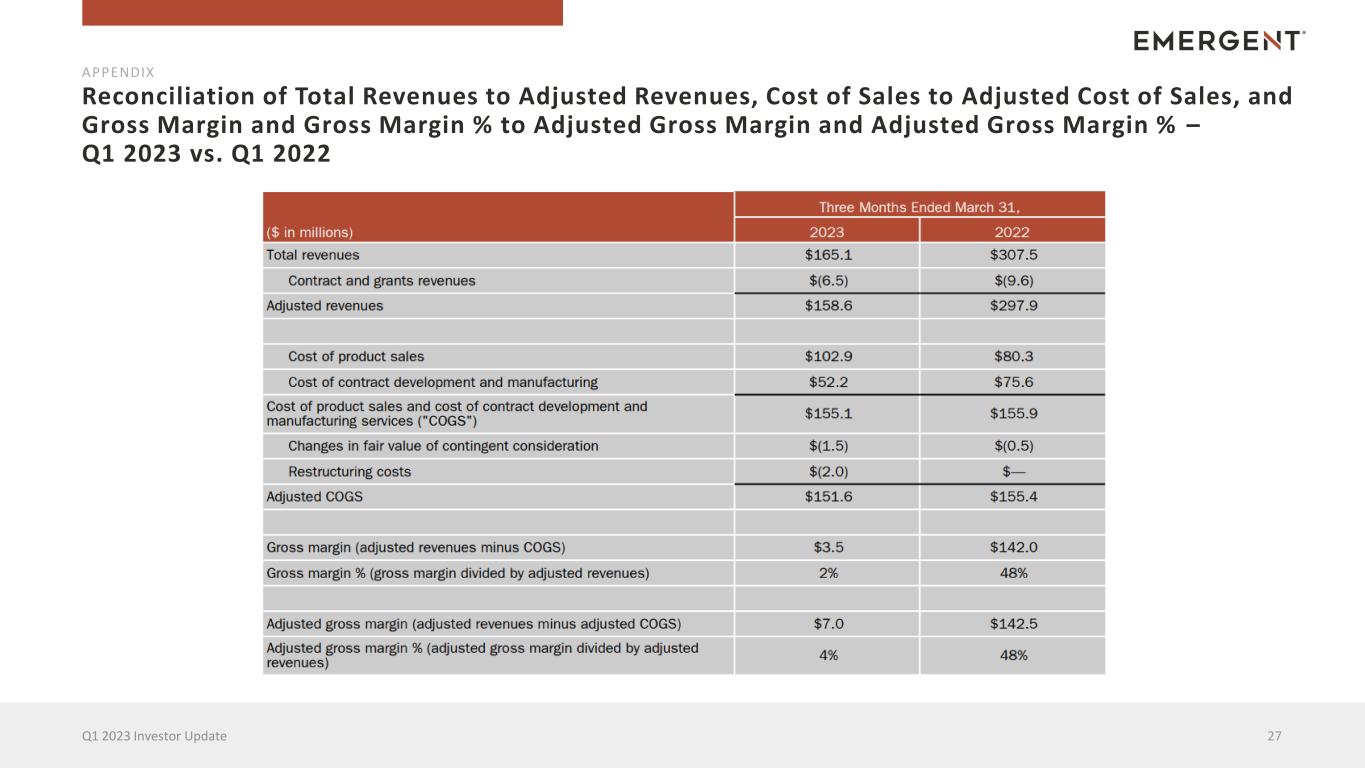
Reconciliation of Total Revenues to Adjusted Revenues, Cost of Sales to Adjusted Cost of Sales, and Gross Margin and Gross Margin % to Adjusted Gross Margin and Adjusted Gross Margin % – Q1 2023 vs. Q1 2022 27Q1 2023 Investor Update APPENDIX
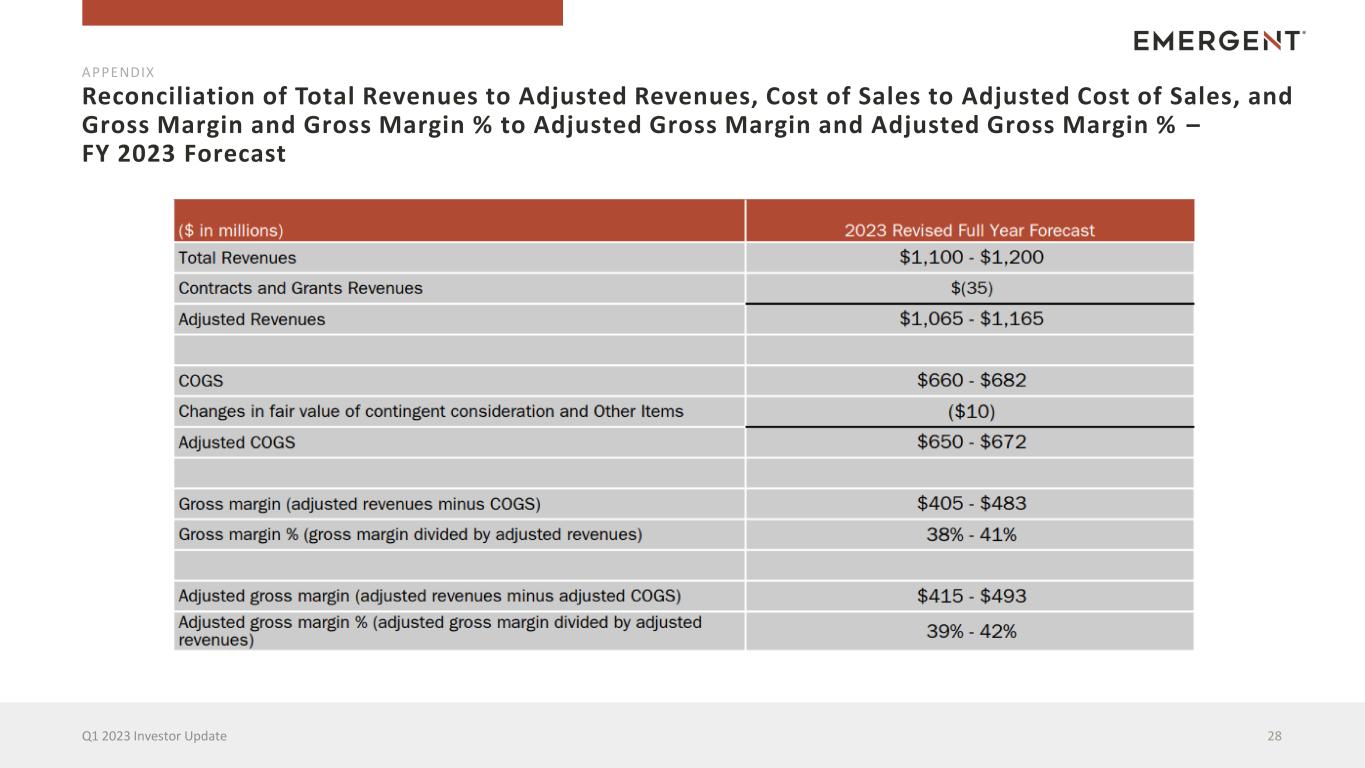
Reconciliation of Total Revenues to Adjusted Revenues, Cost of Sales to Adjusted Cost of Sales, and Gross Margin and Gross Margin % to Adjusted Gross Margin and Adjusted Gross Margin % – FY 2023 Forecast 28Q1 2023 Investor Update APPENDIX
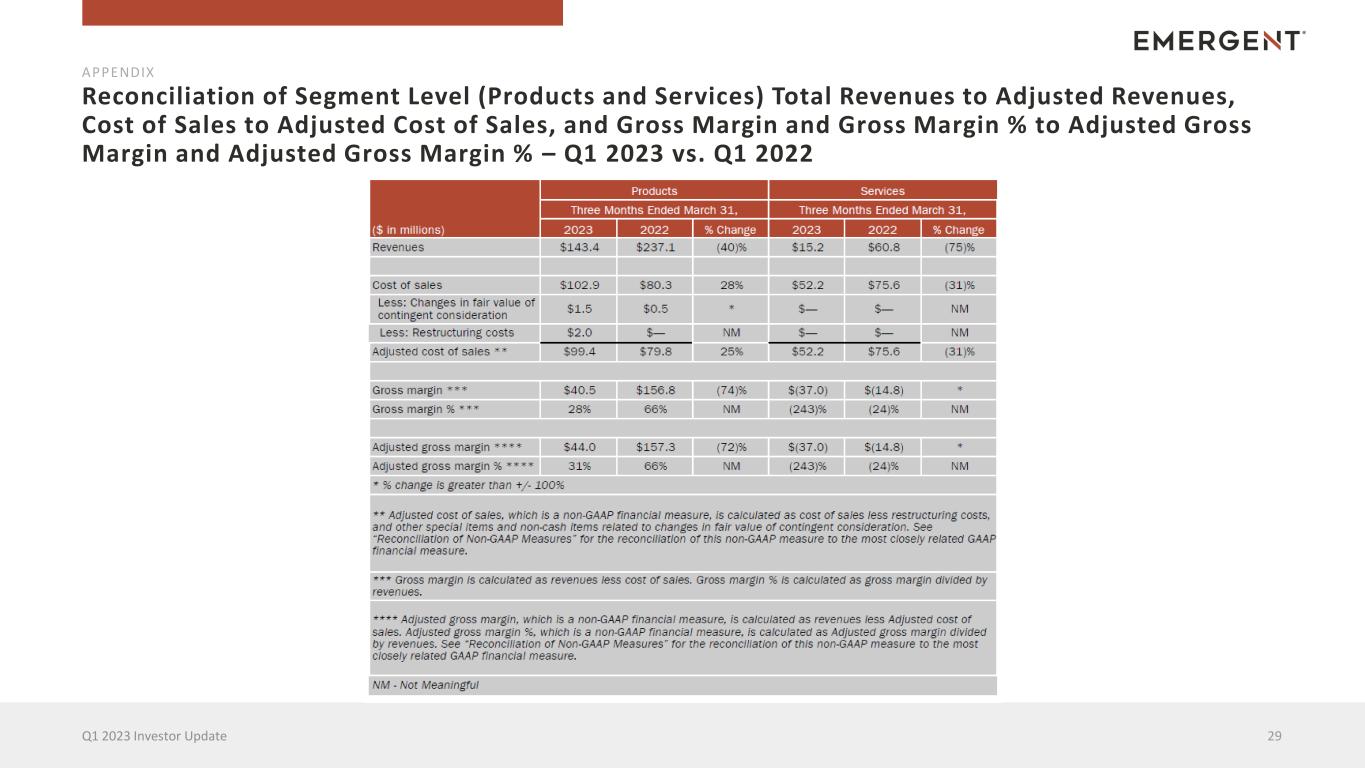
Reconciliation of Segment Level (Products and Services) Total Revenues to Adjusted Revenues, Cost of Sales to Adjusted Cost of Sales, and Gross Margin and Gross Margin % to Adjusted Gross Margin and Adjusted Gross Margin % – Q1 2023 vs. Q1 2022 29Q1 2023 Investor Update APPENDIX

www.emergentbiosolutions.com