 Exhibit 99.1 |
 Page 2 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. This presentation contains forward-looking statements that are subject to a number of risks and uncertainties, many of which are outside our control. All statements regarding our strategy, future operations, financial position, estimated revenues or losses, projected costs, prospects, plans and objectives, other than statements of historical fact included in our filings with the U.S. Securities and Exchange Commission (the “SEC”), are forward-looking statements. When used in this presentation or in answers given to questions asked today, the words “may,” “will,” “could,” “would,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “potential,” “continue,” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. You should not place undue reliance on forward-looking statements. While we believe that we have a reasonable basis for each forward-looking statement that we make, we caution you that these statements are based on a combination of facts and factors currently known by us and projections of future events or conditions, about which we cannot be certain. Forward-looking statements in this presentation should be evaluated together with the many uncertainties that affect our business, and particularly those mentioned in the “Risk Factors” section of our Annual Report on Form 10-K filed with the SEC reporting our financial position and results of operations as of and for the year ended December 31, 2011, in the Registration Statement on Form S-1 that we filed with the SEC on April 6, 2012, as well as subsequent reports filed with the SEC during 2012. In addition, market and industry statistics contained in this presentation are based on information available to us that we believe is accurate. This information is generally based on publications that are not produced for purposes of securities offerings or economic analysis. All forward-looking statements speak only as of the date of this presentation. Except as required by law, we assume no obligation to update these forward-looking statements publicly or to update the factors that could cause actual results to differ materially, even if new information becomes available in the future. Safe Harbor |
 Page 3 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Catalyst Pharmaceutical Partners is focused on the development and commercialization of prescription drugs targeting rare (orphan) neurological diseases and disorders, including Lambert-Eaton Myasthenic Syndrome (LEMS), infantile spasms, and Tourette’s disorder Headquarters: Coral Gables, FL NASDAQ Capital Market: CPRX Shares outstanding: 41,420,687 Share price (2/7/13): $0.54 Market capitalization: $22.4M Cash and investments: $17M* Lead institutional investors: Federated, Millenium, Sophrosyne Strategic investment: BioMarin (16% and joint development agreement) *As of 9/30/12, pro-forma for BioMarin $5MM investment Catalyst Overview |
 Page 4 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. CPP-109 for Cocaine Addiction – Pivotal Phase II(b) trial showed no statistical difference for primary and secondary end points – Disappointing for all Catalyst stakeholders – Full data set available Q2 – 2013 – Meet with NIDA to discuss complete findings – Expect to present data at conference later this year – Trials for cocaine addicts - difficult patient population Catalyst will not continue development of addiction drugs A Glance In The Rear View Mirror |
 Page 5 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Announced strategic alliance with BioMarin (in October) for the development of Firdapse TM North American license for Firdapse TM to treat neuromuscular diseases including LEMS Strategic fit with our other orphan drug programs – CPP-115 for Infantile Spasms and Tourette’s disorder BioMarin invested $5 million for a 16.6% equity stake Joint development agreement for several remaining studies- sharing costs 50/50 Several milestones to be paid later and a mid-teen royalty payment Firdapse TM /BioMarin Alliance |
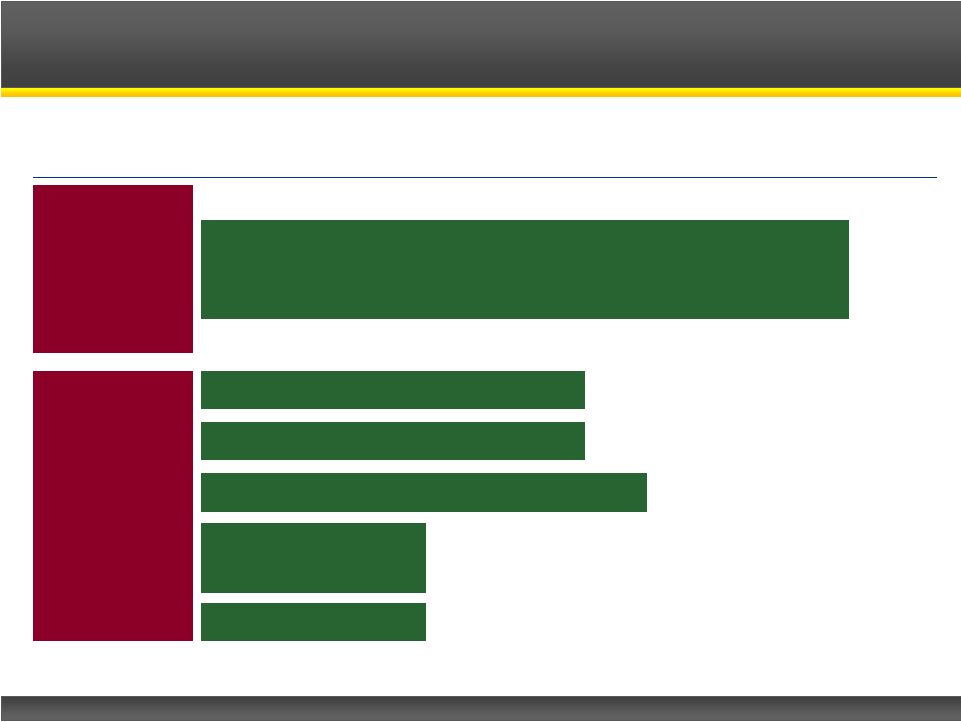 Page 6 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Phase I Preclinical Phase II Phase III Note: *Investigator sponsored study, CPP-109 is a model for CPP-115 to treat this disorder Firdapse TM : Lambert-Eaton Myasthenic Syndrome (LEMS) CPP-115: Complex Partial Seizures CPP-109: Tourette’s Disorder* CPP-115: Infantile Spasms Potassium Channel Blocker GABA-AT Inhibitors CPP-115: Dyskinesia in Parkinson’s CPP-115: MS Product Pipeline |
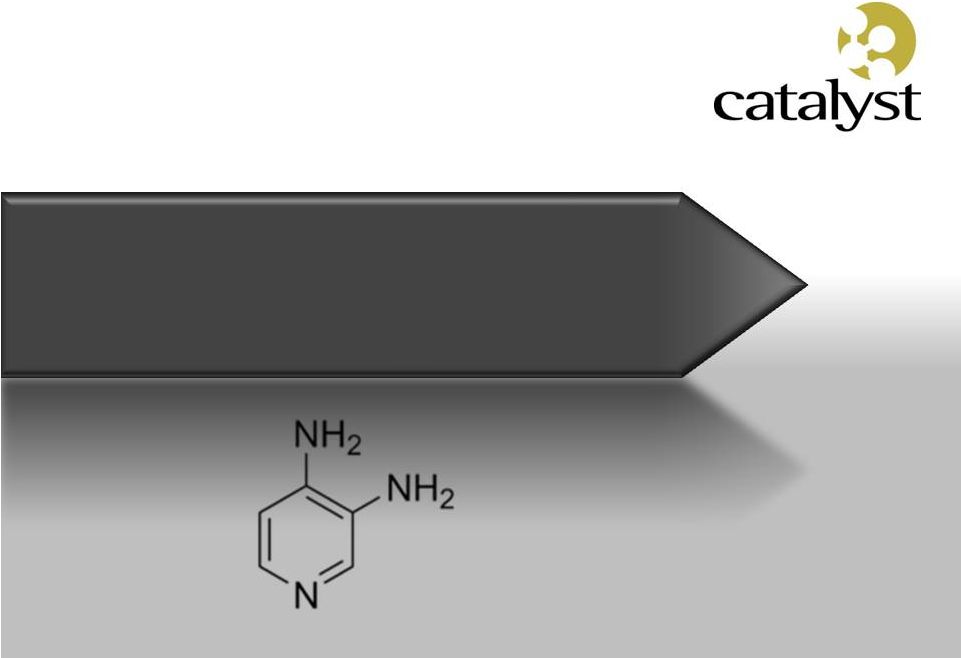 Firdapse TM Potassium Channel Blocker Amifampridine Phosphate (3,4-Diaminopyridine) |
 Page 8 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Lambert-Eaton Myasthenic Syndrome (LEMS) is a rare autoimmune disease caused by auto-antibodies that inhibit acetylcholine release from nerve terminals – Chronic, often severly disabling and progressive – Usually managed by neurologists – ~3,000 patients in the U.S. (10 per 1M prevalence) Continuing pivotal Phase III trial designed and initiated by BioMarin with FDA input Orphan drug designation in the U.S. BioMarin launched Firdapse TM in Europe (2Q10) for the treatment of LEMS – EFNS recommends Firdapse TM as the first-line symptomatic treatment for LEMS – European annual cost of therapy ~$60,000 USD Independent market research indicates annual peak U.S. sales of ~$100 million Opportunities for label expansion Pending composition of matter patent Firdapse TM Opportunity Summary |
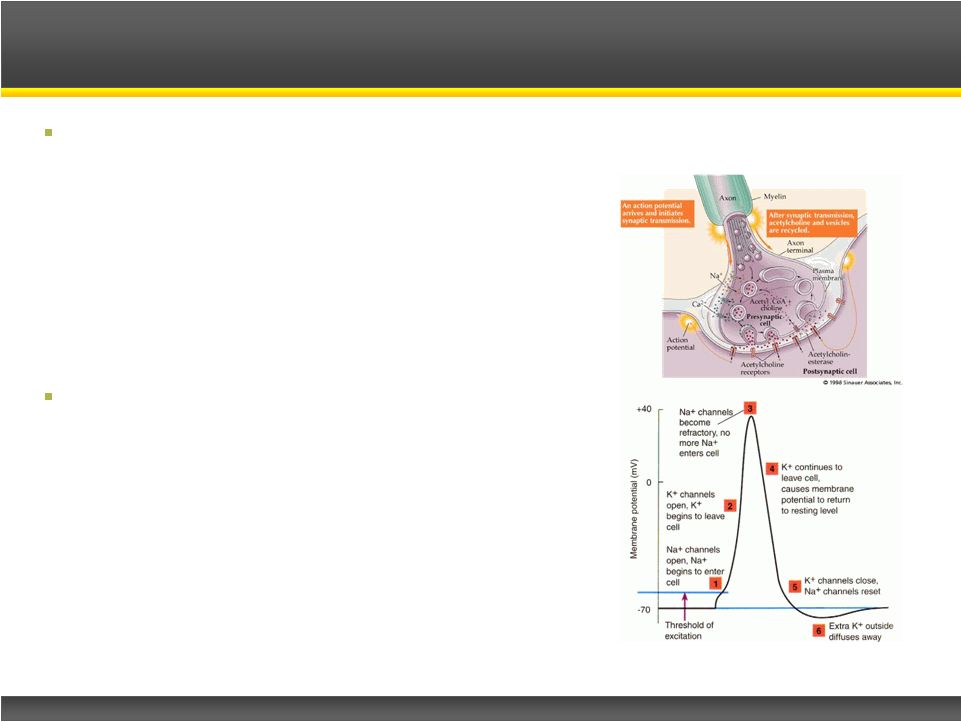 Page 9 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Insufficient ACh release due to antibodies to the pre-synaptic P/Q type voltage gated calcium channel LEMS Disease: – Proximal muscle weakness – Straight forward differential diagnosis – Can be disabling – Often worsens after diagnosis – In some cases, may be life threatening – ~50% of cases associated with SCLC, which has a 12-24 month life expectancy Firdapse TM Treatment: – Potassium channel blocker – Delays neuron repolarization – Voltage gated calcium channels remain open longer – Increased calcium influx causes more acetylcholine to be released to the innervated muscle cells – Restoration of lost muscle strength LEMS and Firdapse TM Treatment |
 Page 10 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Unserved U.S. population due to no FDA approved therapy – Off-label therapies include pyridostigmine and immunosuppressants – IVIg and plasmapheresis are also part of the standard of care Amifampridine is currently available to patients via investigator sponsored INDs and expanded access INDs – Difficult for patients to obtain drug • Even with legally allowed options, many physicians are unwilling or unable to utilize them – Unclear if/how safety reporting is being done – Inadequate control of product manufacturing • Uniformity of potency • Stability U.S. LEMS Product Need |
 Page 11 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Study N Dose, duration Efficacy Outcomes McEvoy, 1989 12 Up to 100 mg/day 3 day crossover Significant on CMAP, disability score, arm/leg strength Sanders, 1993 18 Up to 100 mg/day 8 day crossover Significant on QMG Sanders, 2000 26 60 mg/day 6 day parallel arm Significant on QMG, CMAP Wirtz, 2009 9 Single IV dose, 10 mg Significant on isometric force, CMAP Oh, 2009 8 75 – 80 mg/day 3-8 day crossover Significant on symptom severity, QMG, muscle strength and CMAP Amifampridine Proven Efficacy and Safety in LEMS Significant database of exposure in the literature – 1174 patient exposures for all uses – 169 in LEMS patients |
 Page 12 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Trial design – FDA concurred with design in June, 2010 meeting with BioMarin – Ethical design accepted by KOLs and FDA FDA requires one randomized, placebo-controlled, treatment discontinuation trial in LEMS patients – Compares amifampridine efficacy to placebo at the end of a 14-day discontinuation period – Primary endpoint: Muscle strength (Quantitative Myasthenia Gravis score [QMG]) – Secondary endpoint: Walking speed (Timed 25-foot walking test) – Tertiary endpoint: Compound Muscle Action Potential (CMAP) Approximately 1/3 enrolled 7 active sites (4 U.S./3 Europe) with up to 20 to be added to accelerate the study Data Monitoring Committee (DMC) review Q1 2013 Expect to complete double blind stage of trial around end of Q1 2014 Firdapse TM U.S. Phase III Clinical Trial |
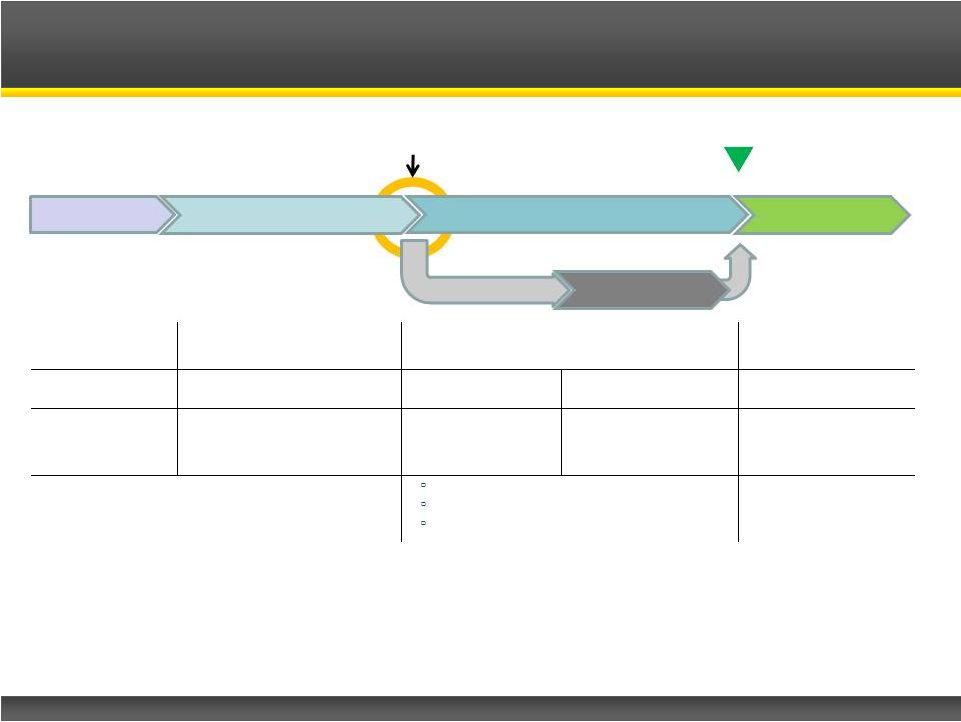 Page 13 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Randomization N=30, 1:1 Dose Taper Last Patient ~March 2014 Firdapse TM U.S. Phase III Clinical Trial Firdapse™ Firdapse™ Firdapse™ Placebo Screening Open Label Run-In Double-Blind Treatment Phase Open-Label Safety Extension Days 1-4 Days 7-91 Day 1-7 Day 8-14 Up to 2 years Efficacy/Baseline Assessments Screening Efficacy/Eligibility Assessments and Dose Adjustments Efficacy Assessments on Day 1 Efficacy Assessments on Days 8 and 14 Safety Assessments and Dose Adjustments 1 : Quantitative Myasthenia Gravis (QMG) 2 : Timed 25 Foot Walk 3 : Compound Muscle Action Potential Not required for NDA filing and approval |
 Page 14 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Congenital Myasthenic Syndrome (CMS) – Prevalence of ~1,500 patients in the U.S. • Eligible for orphan drug designation • Prevalence may be under reported due to complexities of diagnosis – No approved therapy – Differential diagnosis complex – Confirmation of diagnosis from genetic screening for one or more of the 14 known genetic defects Myasthenia Gravis (MG) – Prevalence of ~60,000 patients in the U.S. • Potential to treat a few thousand refractory patients with Firdapse TM – First-line therapy is Mestinon (pyridostigmine), an ACh inhibitor approved before 1982 that is not promoted Firdapse TM Expansion Opportunities |
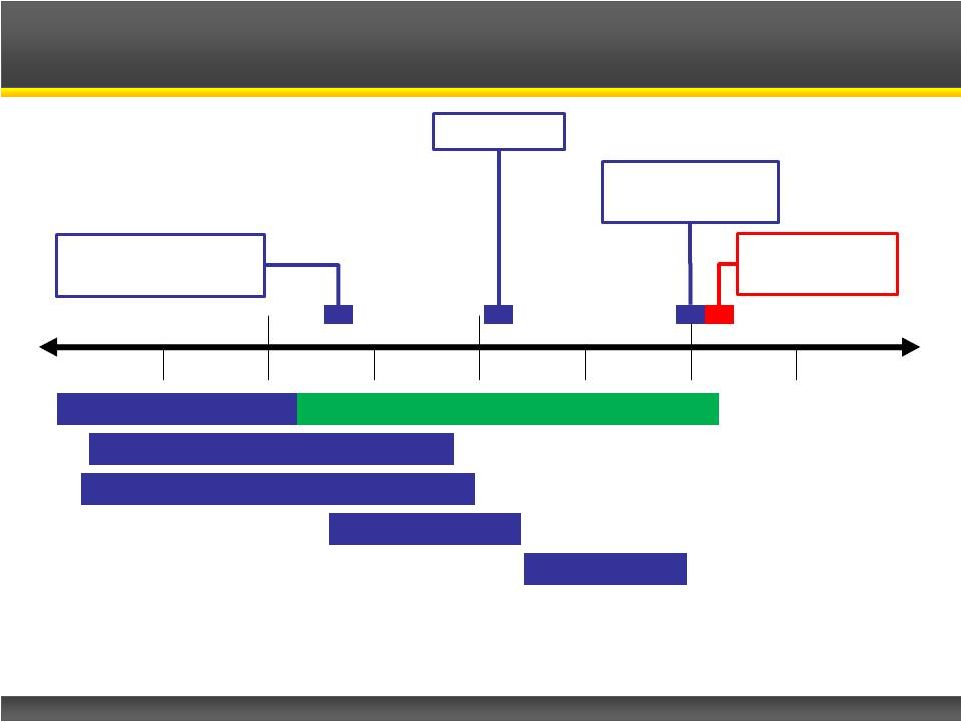 Page 15 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Phase III Clinical Trial Phase III Safety Extension Clinical Safety Studies Pre-Clinical Studies Prepare NDA FDA Review File NDA Estimated FDA Approval Commercial Launch Phase III Top-Line Results Firdapse TM Regulatory Pathway 2013 2014 2015 2016 H1 H2 H1 H2 H1 H2 H1 H2 |
 Page 16 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. First FDA approved drug for LEMS Patient tracking and LEMS support groups to identify patients Market access through private and public payors – Market access research indicates drug will be widely reimbursed Specialty sales force – Initial sales force estimate of 20 sales representatives Orphan drug pricing Patient-assistance program Registry support patients Product education through KOLs Expansion to new indications Firdapse TM Commercialization Strategy |
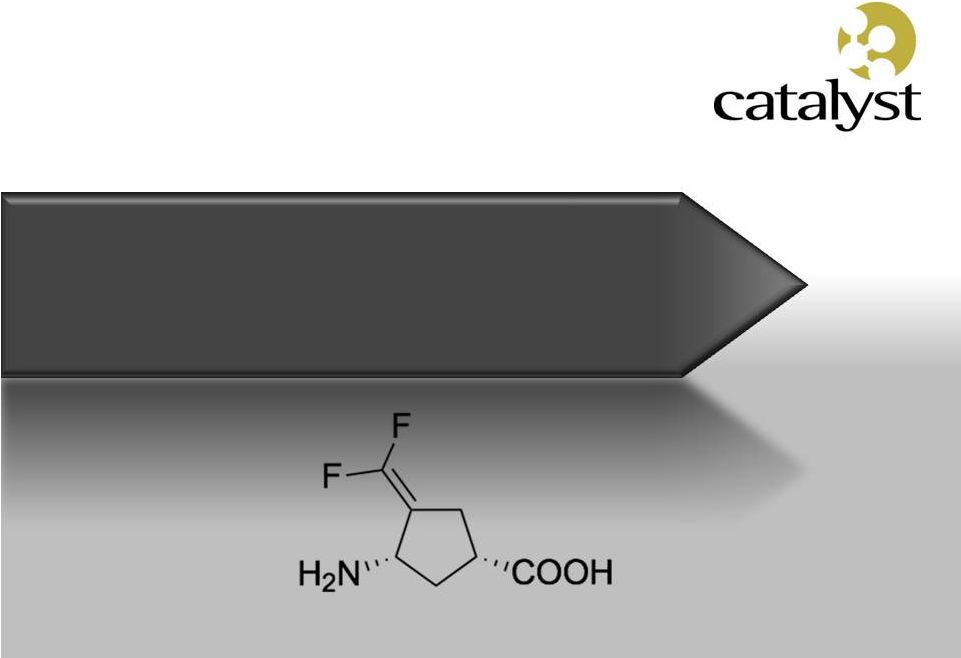 Beyond Firdapse TM : CPP-115 Next Generation GABA-AT Inhibitor |
 Page 18 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Invented by Richard Silverman, Ph.D. – Inventor of Lyrica® (pregabalin); ~$4B in annual sales for Pfizer – Rationally designed drug with enhanced potency, specificity, and safety Exclusive worldwide license to commercialize new GABA-AT inhibitors Includes composition of matter patents to a new class of inhibitors – Protection through 2028 with patent extensions allowed under Patent Term Restoration Act Filed PCT application seeking to protect CPP- 115 in ex-U.S. markets CPP-115 Innovation |
 Page 19 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. CPP-115 Targeted Indications Proven Indications IS CPS Potential New Indications Tourette’s PTSD Movement Disorders Infantile spasms and complex partial seizures are proven indications for inhibition of GABA-AT. CPP-115 could be a safer and more effective alternative. HypoGABAergic signaling pathways are known to play a role in these conditions. |
 Page 20 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Risk of visual field deficits with vigabatrin use – Black box warning on label and REMS program – Occurs in 1/3 to 1/2 of patients – Permanent loss of some peripheral vision Comparative vision safety study in rats 1 For infantile spasms in Multiple Hit Model, and dose shown to inhibit GABA-AT in other studies CPP-115, at 20 times its effective dose, is safer than vigabatrin at its effective dose CPP-115, at its effective dose, will likely be even safer – Potentially no Visual Field Defect (VFD) risk CPP-115 Superior Visual Safety Effective Dose (Rats) Vision Study Dose Vision Safety Margin 45 Day Retinal Function Loss (ERG) 90 Day Retinal Function Loss (ERG) Vigabatrin 300 mg/kg 200 mg/kg ~1 ~30-60% ~45-60% CPP-115 < 1 mg/kg 1 20 mg/kg > 20 ~5-30% ~10-35% |
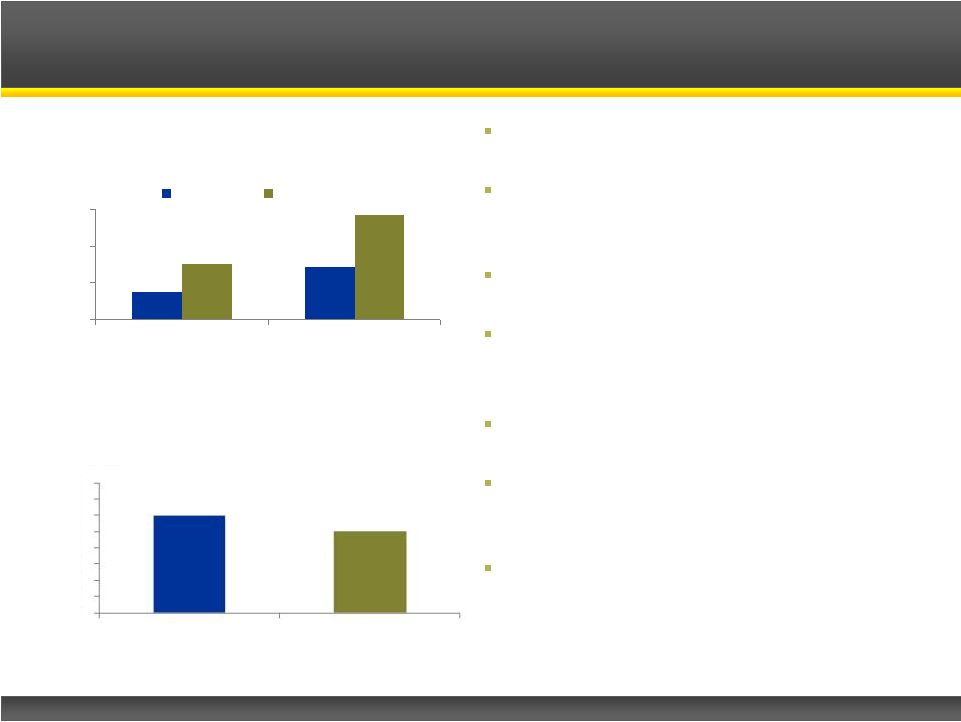 Page 21 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. 0 2,000 4,000 6,000 U.S. Europe Incidence Prevalence Infantile spasms, or West Syndrome, is a catastrophic form of epilepsy for infants Affects 10K – 20K infants worldwide, with nearly one-half of them in the U.S. and Europe 60-70% of patients have underlying disorder Leading therapies are Acthar ® Gel and Sabril ® ; generate ~$100M in U.S. sales – In spite of significant side effects CPP-115 has U.S. and EU orphan drug designations CPP-115 will be a new first-line therapy, as well as for non-responders to existing therapies Opportunity valuation – Lundbeck paid ~$300M for Sabril ® rights – Oppenheimer values Questcor’s Acthar ® Gel infantile spasm franchise at ~$300M Infantile Spasms Opportunity Infantile Spasms Epidemiology (2010) 310M Source: Epilepsia 2010; Population Reference Bureau 2010; Company Reports; Catalyst Estimates U.S. Infantile Spasms Sales (2011E) Sales ($M) 739M Population: 0 10 20 30 40 50 60 $70 $80 Acthar Gel Sabril |
 Page 22 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. “Multiple-Hit Model” for ACTH-refractory infantile spasms (Albert Einstein College of Medicine) – Widely Respected model of infantile spasms – CPP-115 (0.1-1 mg/kg/day i.p.) suppressed spasms at 1/100th the dose of vigabatrin, with better tolerance than vigabatrin 1 – CPP-115 more effective than vigabatrin • Magnitude and duration of seizure reduction greater than vigabatrin • CPP-115 causes no sedation in contrast to vigabatrin which causes severe sedation at therapeutic doses 1 Briggs SW, Ono T, Moshé SL, Galanopoulou AS (2011): presented at the American Epilepsy Society Meeting CPP-115 Infantile Spasms Screening (December 2011) |
 Page 23 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Met with FDA Oct 2011 to discuss IS development plans – Agreed on development strategy through phase II – Relatively standard development pathway to enter phase II • No unusual phase 1 or preclinical requirements, other than preclinical work in juvenile animals for IS indication – Agreed on phase II design • Escalating dose study, N=25-30 infants Utilizing experts Jack Pellock, MD and Don Shields, MD as consultants – Widely respected KOLs for infantile spasms – Accompanied Catalyst to FDA meeting Phase 1 studies (supports any indication) – Phase 1 SAD study completed Q2 2012 – Phase 1 MAD study designed (includes MRI efficacy biomarker) Phase 2 enabling toxicology studies are needed Will seek additional development funding – Potential partners – NIH CPP-115 Development Strategy |
 Page 24 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Indications for which no predictive animal models exist – Tourette’s disorder • 6-10 patient, open label, phase I/II study in progress • Top-line results Q4 2013 – Post Traumatic Stress Disorder (PTSD) – CPP-109 (vigabatrin) used as a research “surrogate” for CPP-115 • CPP-115 and CPP-109 have same mechanism of action • Extended duration use in man for CPP-109 allowed with frequent vision testing • Will use CPP-109 until CPP-115 is developed sufficiently to support use in phase 2 studies Indications for which predictive animal models exist – Dyskinesia in Parkinson’s Disease – Multiple Sclerosis CPP-115: Other Potential Indications |
 Page 25 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Acquired Firdapse TM , a phase III asset, for development and commercialization Completed $5,000,000 strategic investment by BioMarin Initiated Phase I/II study for Tourette’s Disorder Met with FDA to define development plan for CPP-115 to treat infantile spasms Granted orphan medicinal product designation in EU for CPP-115 for treatment of West Syndrome (infantile spasms) Reported CPP-115 Phase I(a) study results Filed U.S. provisional patent application for GABA-AT inhibitor use in treatment of Tourette Syndrome Completed common stock public offering Q1 2013 – Firdapse TM DMC meeting results Q4 2013 – Complete enrollment of Firdapse TM phase III clinical trial – Top-line results from Tourette’s Disorder study Q2 2014 – Top-line results from Firdapse TM phase III clinical trial Q1 2015 – File Firdapse TM NDA Catalyst Milestones Recent Milestones Expected Milestones |
 Page 26 Copyright © 2013 Catalyst Pharmaceutical Partners, Inc. Contact Information Catalyst Pharmaceutical Partners, Inc. Investor Relations 355 Alhambra Circle, Suite 1500 Rx Communications Coral Gables, FL 33134 Melody Carey (305) 529-2522 (917) 322-2568 mcarey@rxir.com Patrick J. McEnany Chairman and Chief Executive Officer pmcenany@catalystpharma.com |
 |