Exhibit 99.1
NASDAQ: ETRM Company Presentation 2017 Confidential information. Do not distribute.

2 This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements generally can be identified by the use of words such as expect,” “plan,” “anticipate,” “could,” “may,” “intend,” “will,” “continue,” “future,” other words of similar meaning and the use of future dates. Forward-looking statements in this presentation include statements about the company’s total addressable market and annual procedures; competitive companies, technologies and procedures; the company’s ability to implement its business model and strategic plan, including its commercialization and reimbursement strategies; expansion of the company’s sales team; and implementation and completion of clinical trials. These forward-looking statements are based on the current expectations of our management and involve known and unknown risks and uncertainties that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others: our limited history of operations; our losses since inception and for the foreseeable future; our limited commercial sales experience with our vBloc® System for the treatment of obesity in the United States or in any foreign market other than Australia and the European Community; the competitive industry in which we operate; our ability to maintain compliance with the Nasdaq continued listing requirements; our ability to commercialize our vBloc® System; our dependence on third parties to initiate and perform our clinical trials; the need to obtain regulatory approval for any modifications to our vBloc® System; physician adoption of our vBloc® System and vBloc® Neurometabolic Therapy; our ability to obtain third party coding, coverage or payment levels; ongoing regulatory compliance; our dependence on third party manufacturers and suppliers; the successful development of our sales and marketing capabilities; our ability to raise additional capital when needed; international commercialization and operation; our ability to attract and retain management and other personnel and to manage our growth effectively; potential product liability claims; the cost and management time of operating a public company; potential healthcare fraud and abuse claims; healthcare legislative reform; our ability to obtain and maintain intellectual property protection for our technology and products; our acquisition of BarioSurg, Inc. may involve unexpected costs or liabilities; the ability to recognize benefits of the acquisition; and risks that the acquisition disrupts current plans and operations. These and additional risks and uncertainties are described more fully in the Company's filings with the Securities and Exchange Commission, particularly those factors identified as "risk factors" in the annual report on Form 10-K filed March 8, 2017 and quarterly report on Form 10-Q filed May 15, 2017. We are providing this information as of the date of this presentation and do not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise. Confidential information. Do not distribute.

Addressing $1.5 billion/year surgical device market¹ Safe, minimally invasive, anatomy preserving solutions with durable outcomes Growing pipeline via continued acquisition and development 1. Global Bariatric Surgery Devices Market Analysis and Trends, Industry Forecast to 2025, Accuray Research 3 Confidential information. Do not distribute.

4 EnteroMedics Company Highlights Addressing the Continuum of Care in Obesity Innovative Medical Technologies vBloc® Therapy Gaining Traction Growing Pipeline + Opportunities Data Supporting Coverage Anatomy friendly solutions Minimally invasive (MIS) procedures Reversible/removable devices Strong surgeon relationships and clinical experience Commercialization programs converting leads to implants Relationship with VA validates vBloc as preferred solution More than 700 vBloc patients implanted to date Over 25 published clinical papers Ongoing clinical trials support marketing and reimbursement Large unmet needs in grossly underpenetrated $1.5 billion market¹ 1. Global Bariatric Surgery Devices Market Analysis and Trends, Industry Forecast to 2025, Accuray Research Acquired Gastric Vest System in Q2 2017 Establishing strategic partnerships Future opportunistic add-ons Confidential information. Do not distribute.

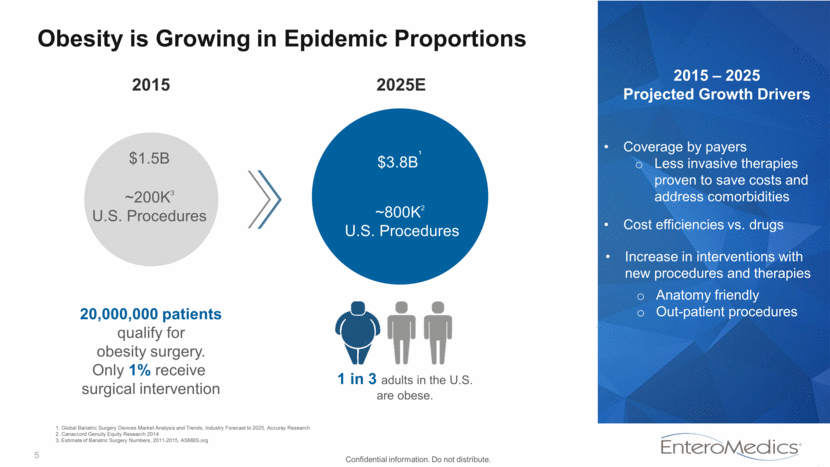
5 Obesity is Growing in Epidemic Proportions $1.5B 2025E ~200K3 U.S. Procedures ~800K2 U.S. Procedures $3.8B¹ 2015 20,000,000 patients qualify for obesity surgery. Only 1% receive surgical intervention 2015 – 2025 Projected Growth Drivers Increase in interventions with new procedures and therapies Anatomy friendly Out-patient procedures Coverage by payers Less invasive therapies proven to save costs and address comorbidities Cost efficiencies vs. drugs Confidential information. Do not distribute. 1 in 3 adults in the U.S. are obese. 1. Global Bariatric Surgery Devices Market Analysis and Trends, Industry Forecast to 2025, Accuray Research 2. Canaccord Genuity Equity Research 2014 3, Estimate of Bariatric Surgery Numbers, 2011-2015, ASMBS,org
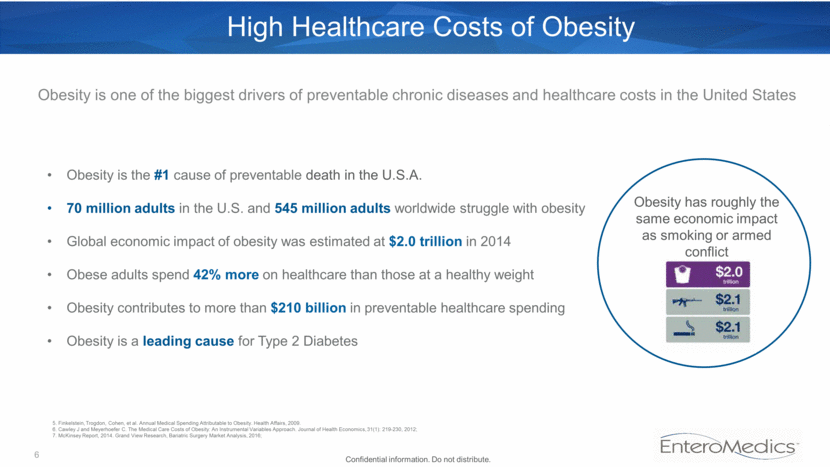
6 High Healthcare Costs of Obesity Obesity is the #1 cause of preventable death in the U.S.A. 70 million adults in the U.S. and 545 million adults worldwide struggle with obesity Global economic impact of obesity was estimated at $2.0 trillion in 2014 Obese adults spend 42% more on healthcare than those at a healthy weight Obesity contributes to more than $210 billion in preventable healthcare spending Obesity is a leading cause for Type 2 Diabetes Obesity has roughly the same economic impact as smoking or armed conflict Obesity is one of the biggest drivers of preventable chronic diseases and healthcare costs in the United States Confidential information. Do not distribute. 5. Finkelstein, Trogdon, Cohen, et al. Annual Medical Spending Attributable to Obesity. Health Affairs, 2009. 6. Cawley J and Meyerhoefer C. The Medical Care Costs of Obesity: An Instrumental Variables Approach. Journal of Health Economics, 31(1): 219-230, 2012; 7. McKinsey Report, 2014. Grand View Research, Bariatric Surgery Market Analysis, 2016;
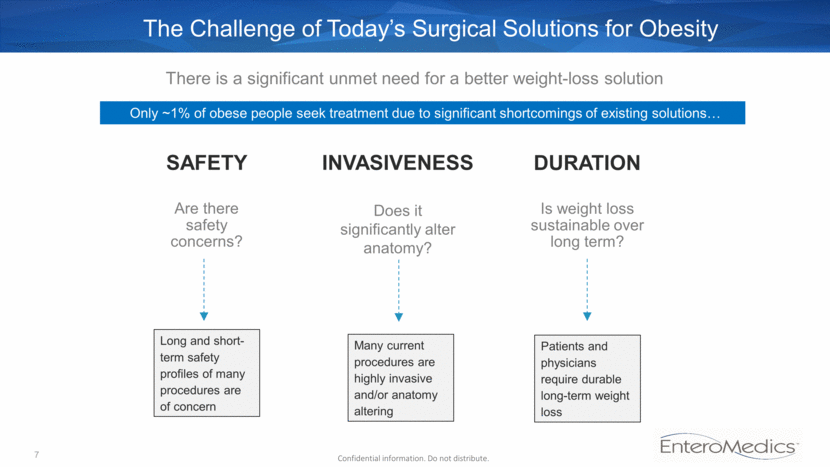
The Challenge of Today’s Surgical Solutions for Obesity SAFETY Are there safety concerns? There is a significant unmet need for a better weight-loss solution Long and short-term safety profiles of many procedures are of concern Many current procedures are highly invasive and/or anatomy altering Only ~1% of obese people seek treatment due to significant shortcomings of existing solutions Patients and physicians require durable long-term weight loss INVASIVENESS Does it significantly alter anatomy? DURATION Is weight loss sustainable over long term? 7 Confidential information. Do not distribute.
Meeting Customer and Patient Needs 8 DESIRABLE SOLUTION INCREASE QUALITY OF LIFE DURABLE WEIGHT LOSS A SAFE SOLUTION An obesity treatment that provides the feeling of fullness with smaller amounts of food, enabling weight loss and reducing the co-morbidities of excess weight A minimally invasive solution that does not cut or remove portions of the stomach and is removable or reversible A safe solution that patients find desirable and can commit to long term A sustainable solution that helps patients work toward accomplishing their weight loss goals Patient needs drive procedural volumes Confidential information. Do not distribute.

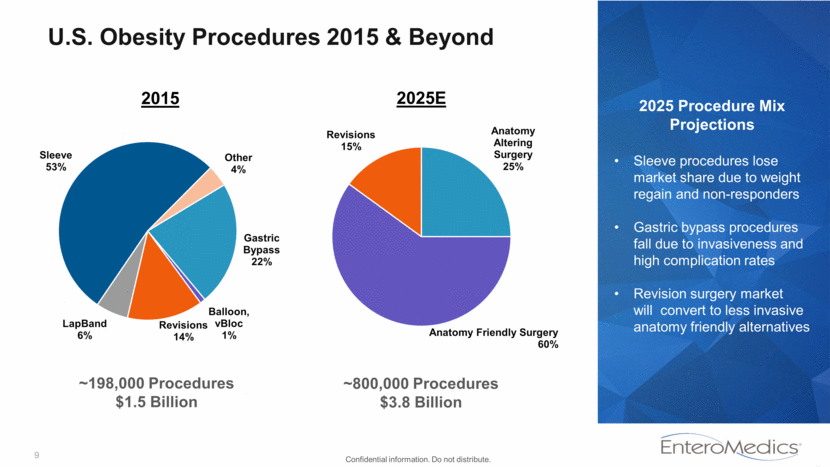
9 U.S. Obesity Procedures 2015 & Beyond 2015 2025E 2025 Procedure Mix Projections Sleeve procedures lose market share due to weight regain and non-responders Gastric bypass procedures fall due to invasiveness and high complication rates Revision surgery market will convert to less invasive anatomy friendly alternatives ~198,000 Procedures $1.5 Billion ~800,000 Procedures $3.8 Billion Confidential information. Do not distribute. Gastric Bypass 22% Balloon, vBloc 1% Revisions 14% LapBand 6% Sleeve 53% Other 4% Anatomy Altering Surgery 25% Anatomy Friendly Surgery 60% Revisions 15%
EnteroMedics’ Current Portfolio 10 Minimally invasive Durable, sustainable, long-term weight loss solution Manages co-morbidities Non anatomy changing; reversible Safe with low complication rates FDA approved In-house 24/7 Customer service component Lifestyle and nutritional advice for our patients Immediate, high volume weight loss solution For morbidly obese, high BMI patients Shorter term, minimally invasive treatment Removable Currently for Investigational Use Only Confidential information. Do not distribute.

vBloc® Neurometabolic Therapy 11 More than 700 patients implanted to date 16 centers currently implanting Patient can maintain diet choices Non-anatomy-altering and improved quality of life Safe and effective minimally invasive outpatient procedure Proprietary bioelectronic neuroblocking technology designed to help patients feel full and eat less by intermittently blocking hunger signals on the vagus nerve. Confidential information. Do not distribute.
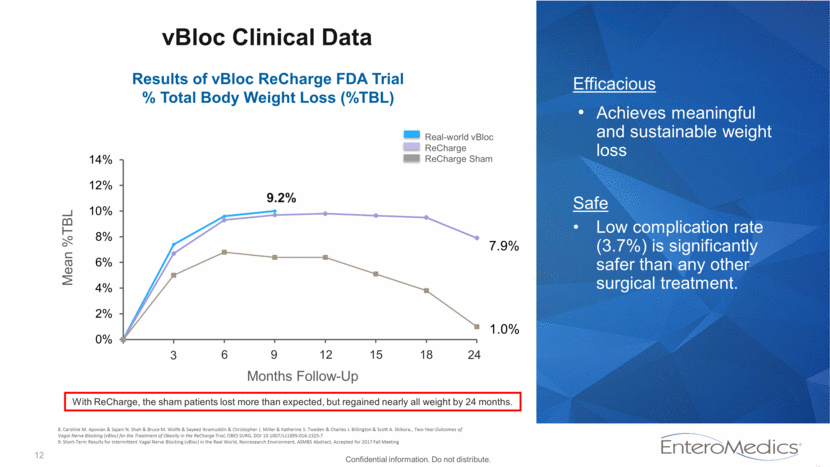
12 vBloc Clinical Data Efficacious Achieves meaningful and sustainable weight loss Safe Low complication rate (3.7%) is significantly safer than any other surgical treatment. Mean %TBL Real-world vBloc ReCharge ReCharge Sham Months Follow-Up Results of vBloc ReCharge FDA Trial % Total Body Weight Loss (%TBL) 3 6 9 24 12 15 18 9.2% With ReCharge, the sham patients lost more than expected, but regained nearly all weight by 24 months. Confidential information. Do not distribute. 8. Caroline M. Apovian & Sajani N. Shah & Bruce M. Wolfe & Sayeed Ikramuddin & Christopher J. Miller & Katherine S. Tweden & Charles J. Billington & Scott A. Shikora., Two-Year Outcomes of Vagal Nerve Blocking (vBloc) for the Treatment of Obesity in the ReCharge Trial, OBES SURG, DOI 10.1007/s11695-016-2325-7 9. Short-Term Results for Intermittent Vagal Nerve Blocking (vBloc) in the Real World, Nonresearch Environment, ASMBS Abstract, Accepted for 2017 Fall Meeting 7.9% 1.0% 0% 2% 4% 6% 8% 10% 12% 14%
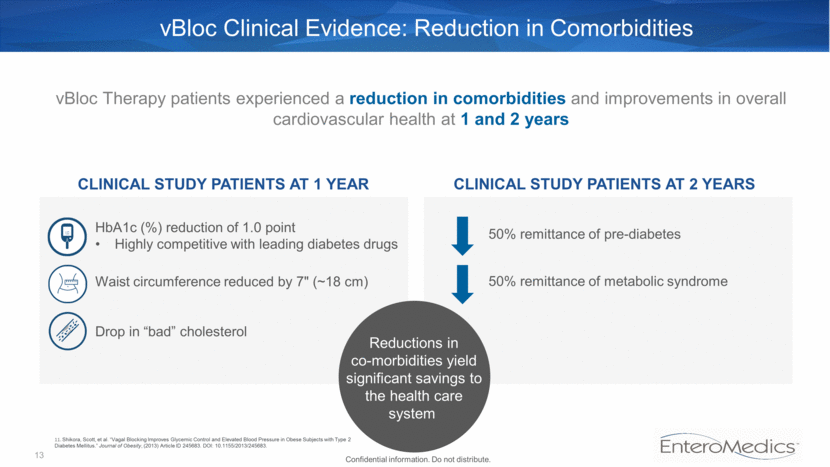
50% remittance of metabolic syndrome Waist circumference reduced by 7" (~18 cm) Drop in “bad” cholesterol HbA1c (%) reduction of 1.0 point Highly competitive with leading diabetes drugs vBloc Clinical Evidence: Reduction in Comorbidities CLINICAL STUDY PATIENTS AT 1 YEAR vBloc Therapy patients experienced a reduction in comorbidities and improvements in overall cardiovascular health at 1 and 2 years CLINICAL STUDY PATIENTS AT 2 YEARS Reductions in co-morbidities yield significant savings to the health care system 13 50% remittance of pre-diabetes Confidential information. Do not distribute. 11. Shikora, Scott, et al. “Vagal Blocking Improves Glycemic Control and Elevated Blood Pressure in Obese Subjects with Type 2 Diabetes Mellitus.” Journal of Obesity, (2013) Article ID 245683. DOI: 10.1155/2013/245683.
14 EnteroMedics acquired the Gastric Vest in May 2017 Minimally invasive No stapling, cutting, or removing the stomach Device is removable / reversible Studies show rapid sustainable weight loss in 12 months Currently for investigational use only The Gastric Vest is a thin, implantable-grade silicon device that wraps around the stomach emulating the effect of conventional weight-loss surgery. Confidential information. Do not distribute.

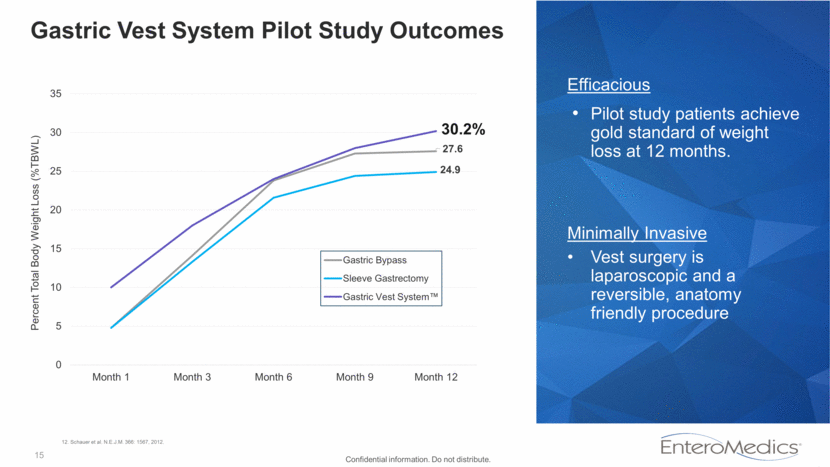
15 Gastric Vest System Pilot Study Outcomes 30.2% Efficacious Pilot study patients achieve gold standard of weight loss at 12 months. Minimally Invasive Vest surgery is laparoscopic and a reversible, anatomy friendly procedure Confidential information. Do not distribute. 12. Schauer et al. N.E.J.M. 366: 1567, 2012. 27.6 24.9 0 5 10 15 20 25 30 35 Month 1 Month 3 Month 6 Month 9 Month 12 Percent Total Body Weight Loss (%TBWL) Gastric Bypass Sleeve Gastrectomy Gastric Vest System™
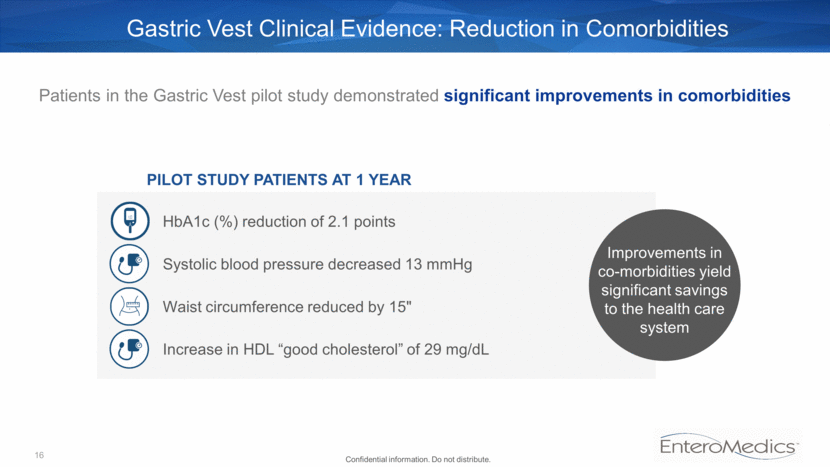
PILOT STUDY PATIENTS AT 1 YEAR Patients in the Gastric Vest pilot study demonstrated significant improvements in comorbidities HbA1c (%) reduction of 2.1 points Systolic blood pressure decreased 13 mmHg Increase in HDL “good cholesterol” of 29 mg/dL Improvements in co-morbidities yield significant savings to the health care system Gastric Vest Clinical Evidence: Reduction in Comorbidities 16 Waist circumference reduced by 15" Confidential information. Do not distribute.
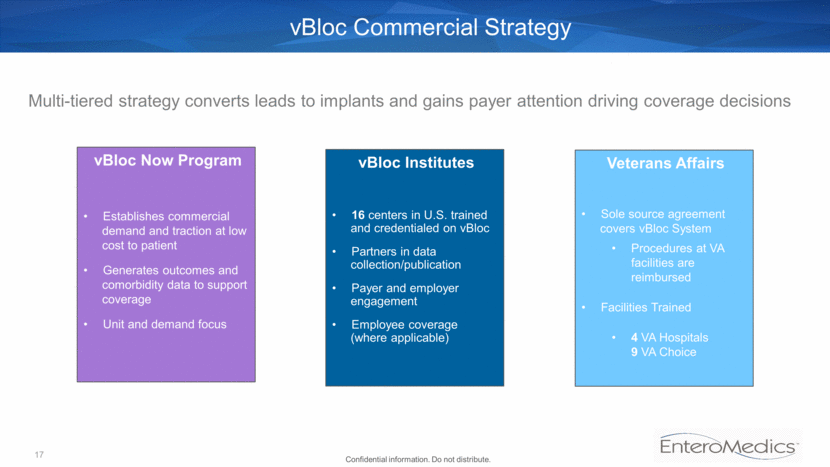
vBloc Commercial Strategy Multi-tiered strategy converts leads to implants and gains payer attention driving coverage decisions vBloc Institutes 16 centers in U.S. trained and credentialed on vBloc Partners in data collection/publication Payer and employer engagement Employee coverage (where applicable) Veterans Affairs Sole source agreement covers vBloc System Procedures at VA facilities are reimbursed Facilities Trained 4 VA Hospitals 9 VA Choice vBloc Now Program Establishes commercial demand and traction at low cost to patient Generates outcomes and comorbidity data to support coverage Unit and demand focus 17 Confidential information. Do not distribute.
Strategy to Collect vBloc Outcomes Data for Reimbursement vBloc Now Program (June 2017) Real-world outcomes on over 125 patients from select vBloc institutes Will include co-morbidity improvement data Data to be utilized to support reimbursement ReNew FDA Post-Market Study (August 2017) Five year, 200 patient trial with vBloc Therapy and vBloc Achieve aftercare program Up to 15 sites Kaiser Diabetic Study (September 2017) Partnered with Kaiser Permanente on a 3 year, 60 patient study Full enrollment expected mid-2018 Goal to demonstrate advantage of vBloc Therapy over non-surgical Rx treatment of diabetes Three initiatives underway supporting coverage and reimbursement decisions with payers 18 Confidential information. Do not distribute.

Gastric Vest Clinical and Regulatory Path US FDA Study PMA. 200-250 patient pivotal trial with a 12-month weight loss and safety endpoint. Minimum of 2-year follow-up First US PMA implants expected in Q2 2018 with approval estimated Q4 2020, pending FDA review times EU Study: CE Mark trial of 50-100 patients with a 6-month weight loss and safety endpoint. Minimum 12-month follow-up First EU implants will start in Q1 2018 with CE mark estimated Q2 2019 All study designs will include metrics requested by payers (e.g. safety, efficacy, health economics) 19 Confidential information. Do not distribute.

Driving Coverage and Reimbursement MULTI-FACETED REIMBURSEMENT STRATEGY TO DRIVE LONG-TERM COVERAGE Physician Demand Surgeons engage commercial payers and MACs Patient Demand vBloc Access program supports patients during prior authorization and appeals Cost Analysis Study and publish economic savings and complication reduction Data Continue to collect and publish efficacy and safety data Society Support Leverage clinical data through strength of societies, including ASMBS and TOS 20 Confidential information. Do not distribute.

Intellectual Property 21 Bioelectronic Medicine (vBloc) Broad claim coverage for bioelectronic systems and methods related to neuroblocking, neuromodulation, and neurostimulation technology IP covers indications including obesity, bulimia, pancreatitis, heart rate regulation, and glucose regulation 45 granted U.S. patents 45 granted foreign patents in Australia, Europe, China and Japan Additional U.S. and 22 OUS patents pending Gastric Vest Intellectual property for gastric restriction device to treat obesity 4 granted U.S. patents 4 granted foreign patents in China, Israel, Canada and Australia Confidential information. Do not distribute.

Strategic Partnership with Galvani Bioelectronics 22 Confidential information. Do not distribute. Galvani is a joint venture between GlaxoSmithKline and Verily Life Sciences (an Alphabet company) that was established in 2016 to enable the research, development and commercialization of bioelectronic medicines. Collaboration Agreement (for pre-clinical research) Funded co-development of new modified EnteroMedics‘ products Future opportunities (continued IP + technology platform co-development) Intellectual property licensing EnteroMedics will receive payments for its development work and supply under this agreement

EnteroMedics Milestones and Objectives Establish vBloc Reimbursement Grow Pipeline Focus on Commercial Use and Outcomes Commercial coverage Publish real-world outcomes and co-morbidity data Complete CE Mark trial for Gastric Vest Complete US PMA trial for Gastric Vest Strategic partnerships and acquisitions Unit placements Co-morbidity and clinical support 3 23 1 2 Confidential information. Do not distribute.

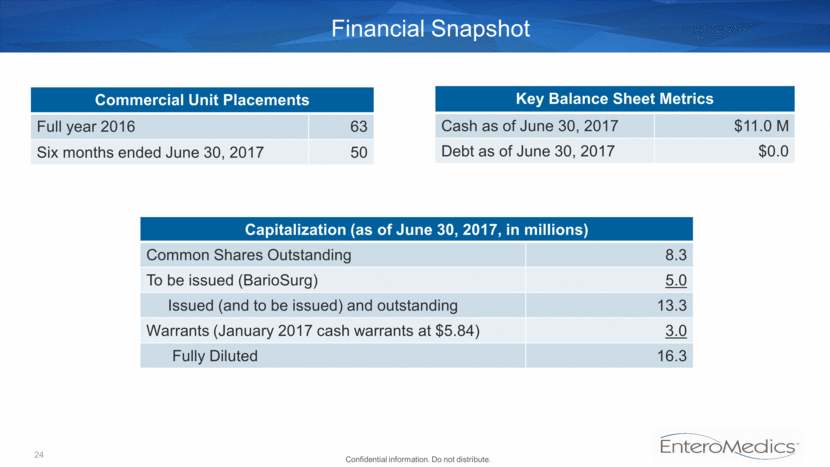
Financial Snapshot Key Balance Sheet Metrics Cash as of June 30, 2017 $11.0 M Debt as of June 30, 2017 $0.0 Commercial Unit Placements Full year 2016 63 Six months ended June 30, 2017 50 Capitalization (as of June 30, 2017, in millions) Common Shares Outstanding 8.3 To be issued (BarioSurg) 5.0 Issued (and to be issued) and outstanding 13.3 Warrants (January 2017 cash warrants at $5.84) 3.0 Fully Diluted 16.3 24 Confidential information. Do not distribute.
Investment Summary 25 Confidential information. Do not distribute. Large, underserved and rapidly expanding market with multiple sub-segments FDA approved vBloc technology gaining commercial traction Growing minimally invasive pipeline for the treatment of obesity via continued acquisition and development

EnteroMedics Inc. (NASDAQ:ETRM) 2800 Patton Road St. Paul, MN 55113 Dan Gladney President, Chief Executive Officer & Chairman of the Board 651-634-3089 dwgladney@enteromedics.com

APPENDIX

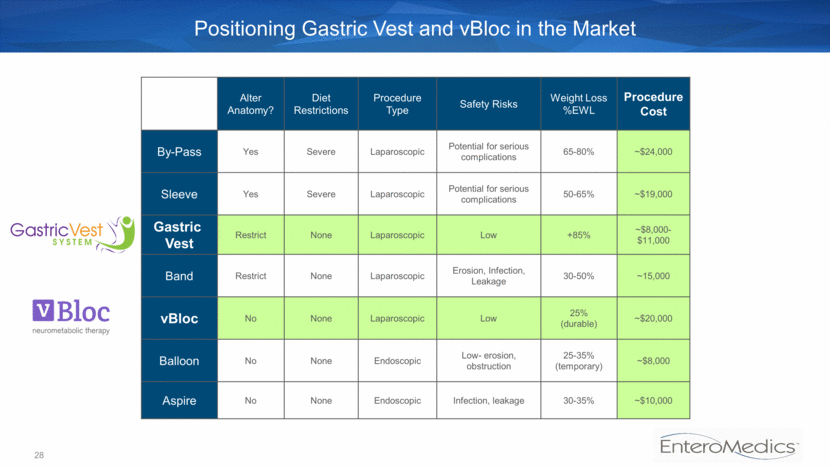
28 Positioning Gastric Vest and vBloc in the Market Alter Anatomy? Diet Restrictions Procedure Type Safety Risks Weight Loss %EWL Procedure Cost By-Pass Yes Severe Laparoscopic Potential for serious complications 65-80% ~$24,000 Sleeve Yes Severe Laparoscopic Potential for serious complications 50-65% ~$19,000 Gastric Vest Restrict None Laparoscopic Low +85% ~$8,000-$11,000 Band Restrict None Laparoscopic Erosion, Infection, Leakage 30-50% ~15,000 vBloc No None Laparoscopic Low 25% (durable) ~$20,000 Balloon No None Endoscopic Low- erosion, obstruction 25-35% (temporary) ~$8,000 Aspire No None Endoscopic Infection, leakage 30-35% ~$10,000