Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
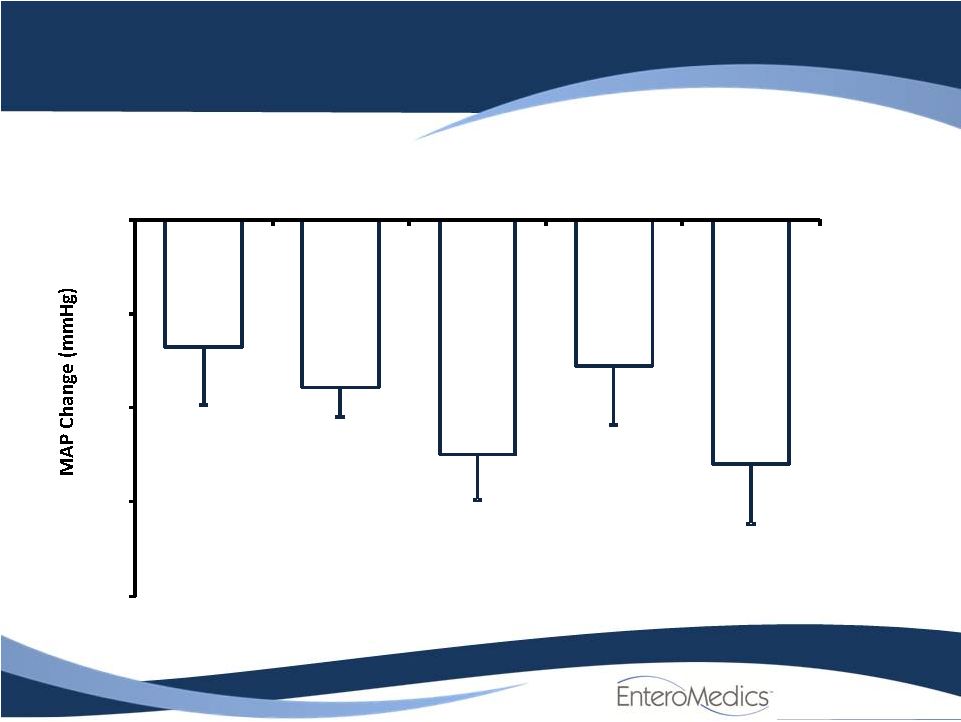
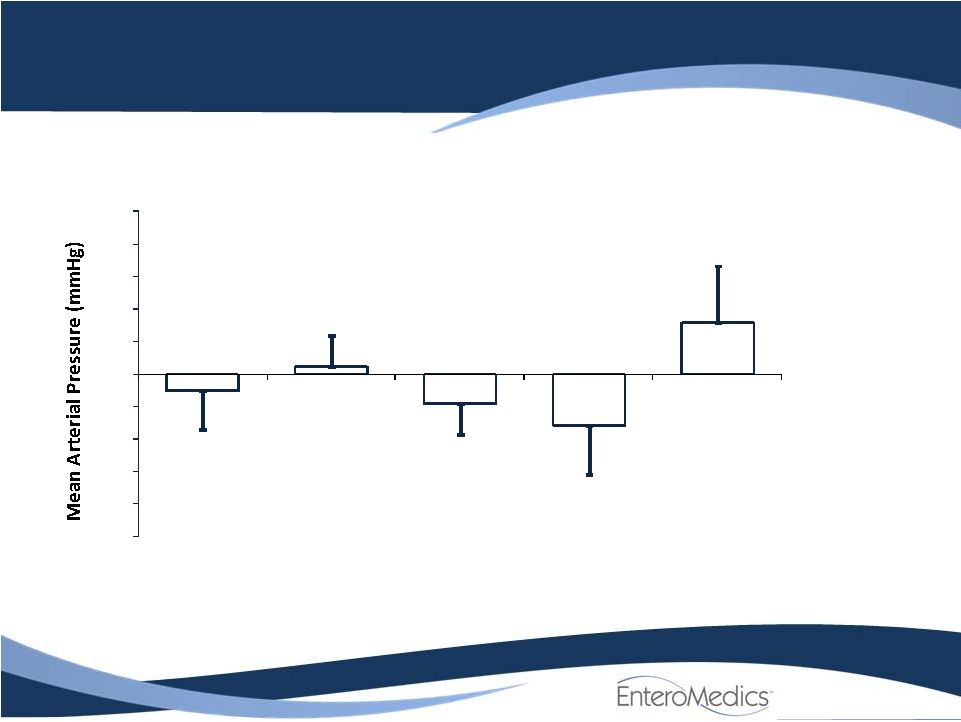
ReShape Lifesciences similar filings
- 23 Sep 11 EnteroMedics Announces Proposed Public Offering of Common Stock and Warrants
- 23 Sep 11 EnteroMedics Announces Pricing of $14.5 Million Public Offering
- 2 Sep 11 Regulation FD Disclosure
- 11 Aug 11 Regulation FD Disclosure
- 21 Jul 11 EnteroMedics Reports Second Quarter 2011 Financial Results
- 8 Jul 11 Entry into a Material Definitive Agreement
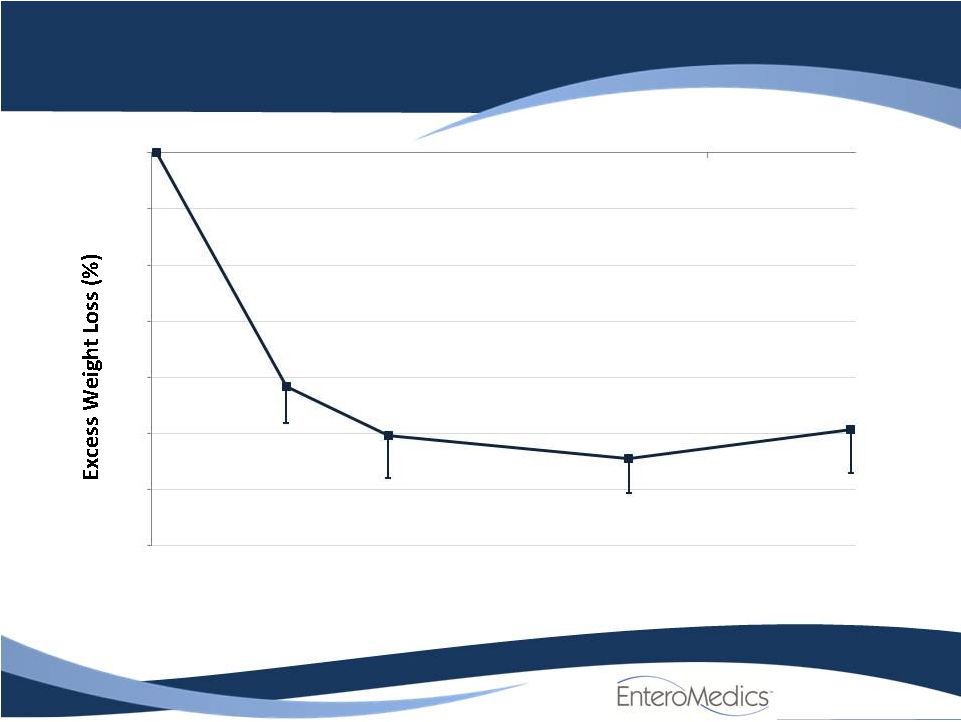
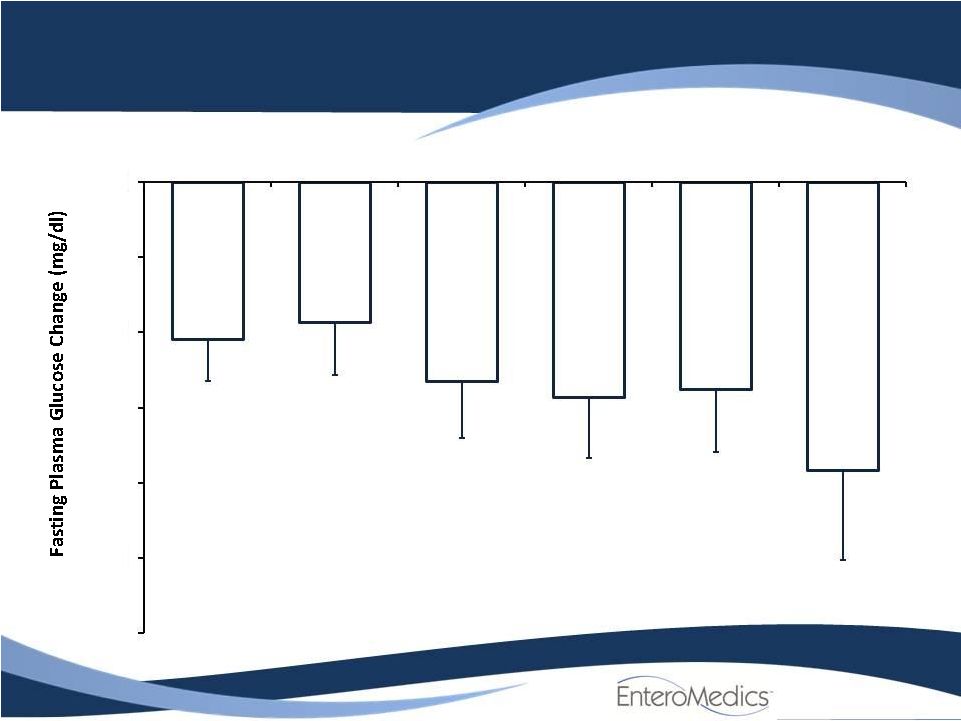
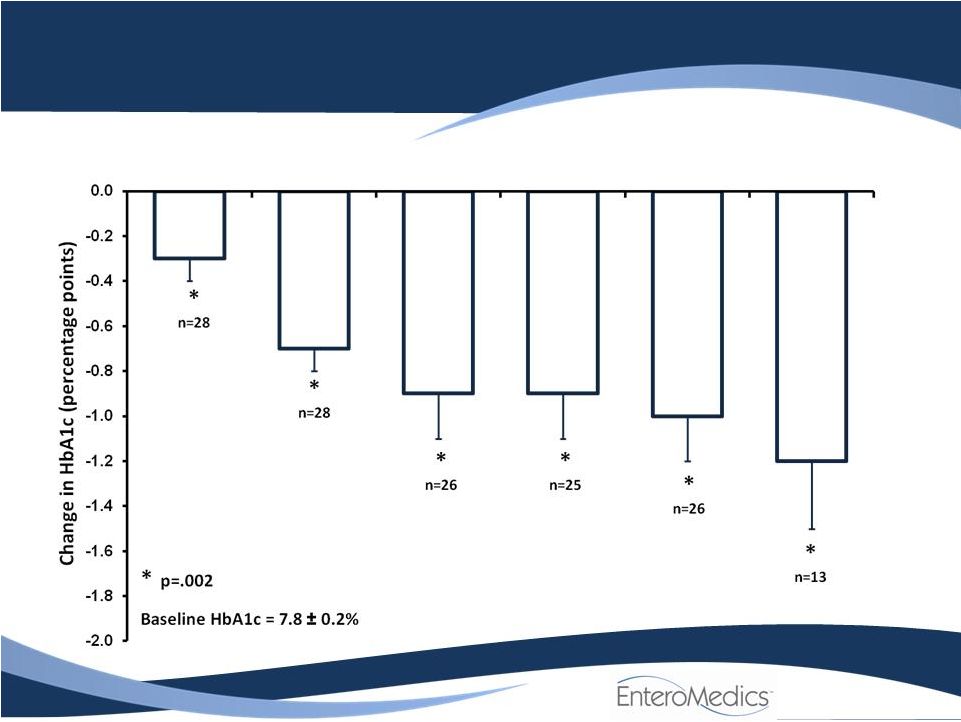
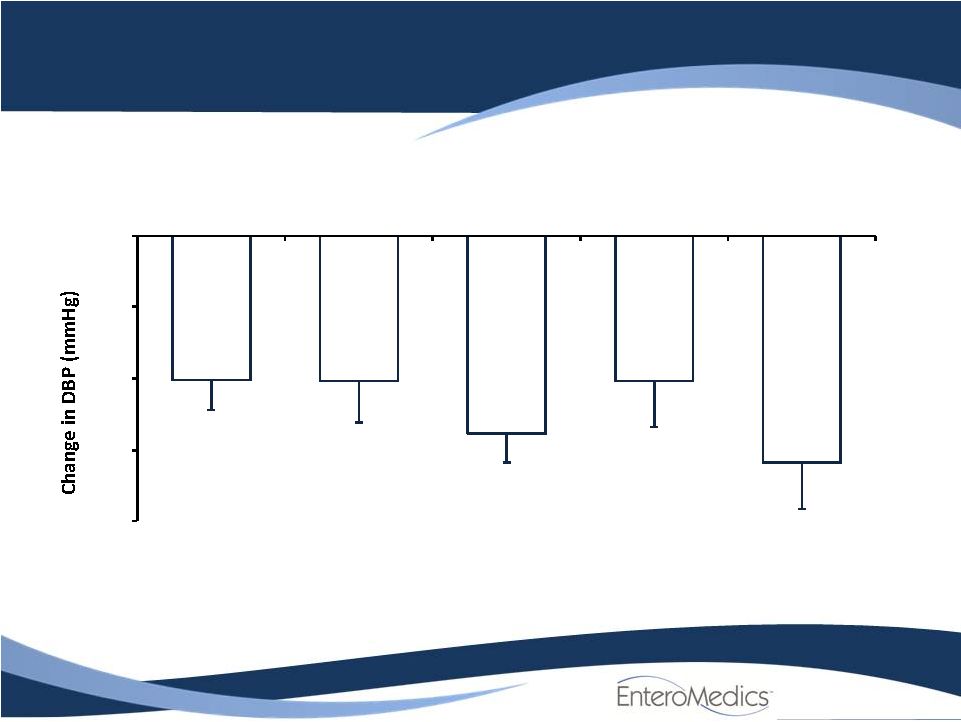
- 14 Jun 11 Company Also Announces Long-term, Updated Data from VBLOC-DM2 and EMPOWER Studies
Filing view
External links