
Business Update and Full Year and Q4 2021 Financial Results NASDAQ: OCGN 1

2 Forward Looking Statement This presentation contains forward‐looking statements within the meaning of The Private Securities Litigation Reform Act of 1995, which are subject to risks and uncertainties. Ocugen may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words that convey uncertainty of future events or outcomes to identify these forward‐looking statements. Such forward‐looking statements include information about Ocugen’s progress in advancing the review of COVAXIN™ with the FDA, including with respect to its EUA submission for COVAXIN™ for pediatric use, and information about its non‐binding LOI with Liminal to acquire Liminal’s manufacturing site in Belleville, Ontario, and are subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statements, including, among other things, the risk that Ocugen may not be able to successfully negotiate and execute definitive transaction agreements for the acquisition of Liminal’s manufacturing site on acceptable terms, if at all, and the ultimate terms and timing for closing of the transactions contemplated thereby; the risk that Ocugen will not be able to successfully close the acquisition of Liminal’s manufacturing site; risks associated with the planned development and refurbishing of the manufacturing site, including that the expected costs for such development will be greater than currently contemplated or that the planned development will take longer than expected or fail to be completed on a timely basis, if at all; and the risk that Ocugen will not be able to scale production for such site to adequately support manufacturing of its product candidates or the other products that are currently or may in the future be manufactured at such site. In addition, Ocugen’s business is subject to numerous other risks and uncertainties, including, among other things, the uncertainties inherent in research and development, including the ability to meet anticipated clinical endpoints, commencement and/or completion dates for clinical trials, regulatory submission dates, regulatory approval dates and/or launch dates; the risk that Health Canada does not accept its NDS for COVAXIN™ or that Ocugen may not be able to adequately resolve the deficiencies noted by Health Canada with respect to its NDS, for which Ocugen has provided responses that are currently under review by Health Canada; the risk that the FDA could make other decisions that adversely impact its ability to advance the development of COVAXIN™ in the United States even though the FDA’s clinical hold on Ocugen’s IND application for COVAXIN™ has been lifted; risks associated with preliminary and interim data, including the possibility of unfavorable new clinical trial data and further analyses of existing clinical trial data; the risk that the results of in‐vitro studies will not be duplicated in human clinical trials; the risk that clinical trial data is subject to differing interpretations and assessments, including during the peer review/publication process, in the scientific community generally, and by regulatory authorities; whether and when data from Bharat Biotech's clinical trials will be published in scientific journal publications and, if so, when and with what modifications; whether the data and results from the preclinical and clinical studies of COVAXIN™, which have been conducted by Bharat Biotech in India, will be accepted by the FDA or Health Canada or otherwise sufficient to support its EUA submission, planned BLA submission, or its NDS; the size, scope, timing and outcome of any additional clinical trials or studies that Ocugen may be required to conduct to support an EUA or BLA; any additional chemistry, manufacturing, and controls information that Ocugen may be required to submit to the FDA or Health Canada; whether and when a BLA for COVAXIN™ will be submitted to or approved by the FDA; whether developments with respect to the COVID‐19 pandemic will affect the regulatory pathway available for vaccines in the United States, Canada, or other jurisdictions; market demand for COVAXIN™ in the United States or Canada; decisions by the FDA or Health Canada impacting labeling, manufacturing processes, safety and/or other matters that could affect the availability or commercial potential of COVAXIN™ in the United States or Canada, including development of products or therapies by other companies. These and other risks and uncertainties are more fully described in Ocugen’s periodic filings with the Securities and Exchange Commission (SEC), including the risk factors described in the section entitled “Risk Factors” in the quarterly and annual reports that it files with the SEC. Any forward‐looking statements that Ocugen makes in this presentation speak only as of the date of this presentation. Except as required by law, Ocugen assumes no obligation to update forward‐looking statements contained in this presentation whether as a result of new information, future events, or otherwise, after the date of this presentation.

3

Forward Momentum for COVAXIN™ (BBV152) 4 02 • Comprehensive responses submitted to Health Canada against notice of deficiency • Letter‐of‐Intent signed with Liminal BioSciences for acquisition of new Canada‐ based manufacturing facility; • Tech transfer progressing with partner, JHS • U.S. FDA lifts clinical hold on IND submission of COVAXIN™, paving way for clinical trials supporting BLA • Pediatric (2‐18) Emergency Use Authorization submission updated with real‐word safety, and Omicron data 01

Forward Momentum for Ocular Portfolio 5 • Phase 1/2 clinical trials studying OCU400 for the treatment of retinitis pigmentosa resulting from genetic mutations of NR2E3 and RHO now enrolling • Currently executing IND‐enabling preclinical studies to support a Phase 1/2a clinical trial OCU200 OCU40001 • IND‐enabling studies ongoing OCU41002 03
6 FINANCIAL UPDATE
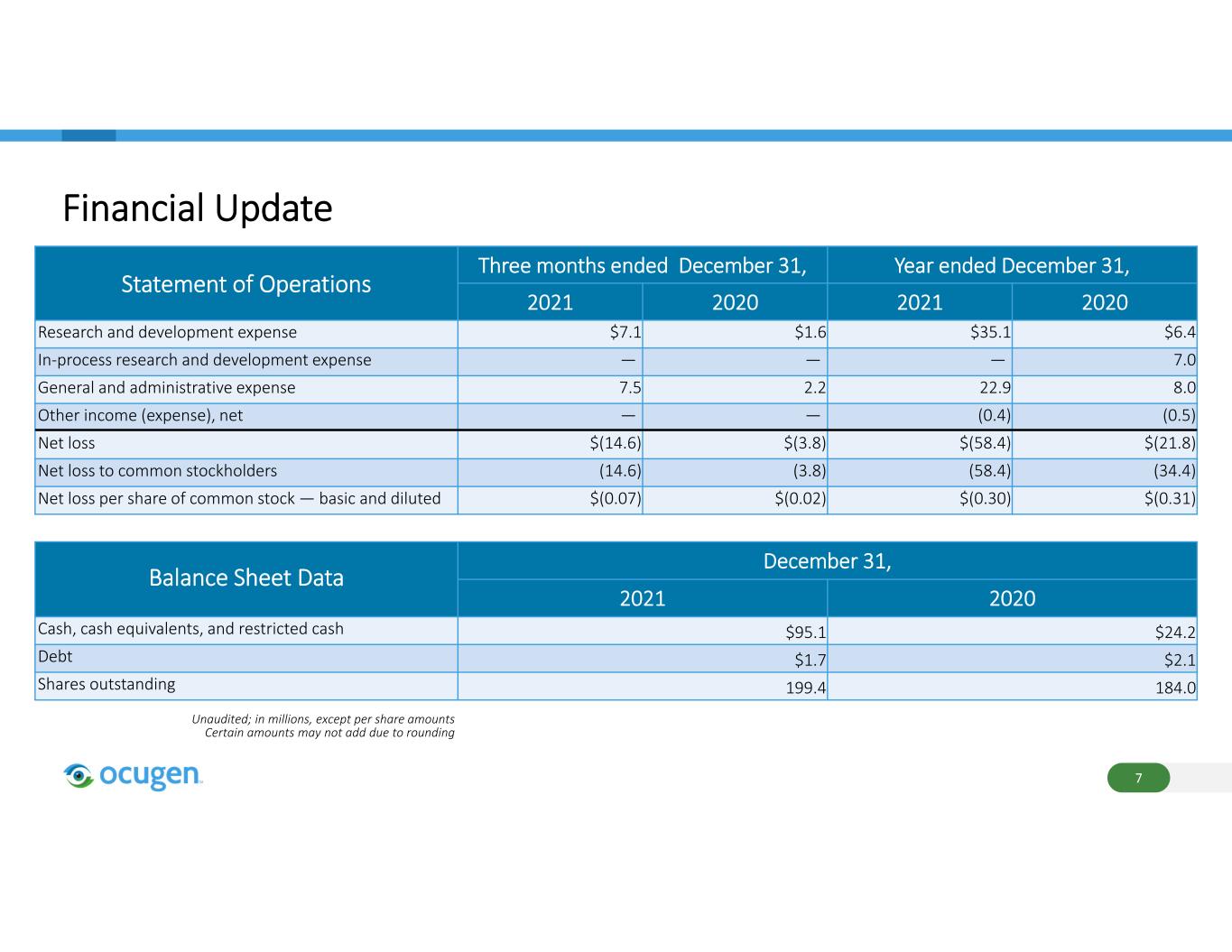
Financial Update Statement of Operations Three months ended December 31, Year ended December 31, 2021 2020 2021 2020 Research and development expense $7.1 $1.6 $35.1 $6.4 In‐process research and development expense — — — 7.0 General and administrative expense 7.5 2.2 22.9 8.0 Other income (expense), net — — (0.4) (0.5) Net loss $(14.6) $(3.8) $(58.4) $(21.8) Net loss to common stockholders (14.6) (3.8) (58.4) (34.4) Net loss per share of common stock — basic and diluted $(0.07) $(0.02) $(0.30) $(0.31) 7 Balance Sheet Data December 31, 2021 2020 Cash, cash equivalents, and restricted cash $95.1 $24.2 Debt $1.7 $2.1 Shares outstanding 199.4 184.0 Unaudited; in millions, except per share amounts Certain amounts may not add due to rounding
8 Q&A

NASDAQ: OCGN 9 Business Update and Full Year and Q4 2021 Financial Results