 [ 1 ] INVESTOR PRESENTATION January 2011 NASDAQ: ZGNX Exhibit 99.1 |
 [ 2 ] Forward Looking Statements Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking statements. Words such as “believes,” “anticipates,” “plans,” “expects,” “indicates,” “will,” “intends,” “potential,” “suggests,” “assuming,” “designed” and similar expressions are intended to identify forward looking statements. These statements are based on Zogenix’s current beliefs and expectations. These forward-looking statements include statements regarding: the ability to successfully commercialize Sumavel DosePro and its continued sales growth; the ability to obtain additional marketing approvals for Sumavel DosePro in the European Union; the timing of the release of Phase IV data for Sumavel DosePro; the potential for, and timing of, an NDA submission for ZX002 and IND submissions for Zogenix’s additional product candidates; the potential for ZX002 to be the first approved oral, single-entity controlled release formulation of hydrocodone; the completion of enrollment of one of the Phase 3 clinical trials for ZX002; the timing of results for the Phase 3 clinical trials for ZX002; the potential of co-promotion discussions for ZX002; the expansion of Zogenix’s existing sales team; the potential to broaden the application of the DosePro technology; and the potential market penetration of Sumavel DosePro and ZX002. The inclusion of forward-looking statements should not be regarded as a representation by Zogenix that any of its plans will be achieved. Actual results may differ from those set forth in this presentation due to the risk and uncertainties inherent in Zogenix’s business, including, without limitation: the market potential for migraine treatments, and Zogenix’s ability to compete within that market; inadequate therapeutic efficacy or unexpected adverse side effects relating to Sumavel DosePro that could delay or prevent commercialization, or that could result in recalls or product liability claims; Zogenix’s dependence on its collaboration with Astellas Pharma US, Inc. to promote Sumavel DosePro; the ability of Zogenix to ensure adequate and continued supply of Sumavel DosePro to successfully launch commercial sales or meet anticipated market demand; the progress and timing of Zogenix’s clinical trials; the potential that earlier clinical trials may not be predictive of future results; the potential for ZX002 to receive regulatory approval on a timely basis or at all; the potential for adverse safety findings relating to ZX002 to delay or prevent regulatory approval or commercialization; the ability of Zogenix and its licensors to obtain, maintain and successfully enforce adequate patent and other intellectual property protection of its products and product candidates and the ability to operate its business without infringing the intellectual property rights of others; and other risks described in Zogenix’s filings with the Securities and Exchange Commission. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, and Zogenix undertakes no obligation to revise or update this presentation to reflect events or circumstances after the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement. This caution is made under the safe harbor provisions of Section 21E of the Private Securities Litigation Reform Act of 1995. |
 [ 3 ] Commercializing and Developing Products for Treatment of Central Nervous System (CNS) Disorders and Pain RAMPING SALES 1 Single Use, Needle-Free, Commercial Product $3.5B Market Acute Migraines and Cluster Headaches $13B Market Codeine and Morphine Based Pain Products MEDICINE and TECHNOLOGY PHASE 3 DEVELOPMENT ZX002 1 Oral Single-Entity, Controlled Release Hydrocodone st st Sources: Wolters Kluwer Pharma Solutions, Source® PHAST Institution/Retail (12 months ended June 30, 2010) |
 [ 4 ] Established Commercial Infrastructure • Sophisticated Manufacturing and Sales Organization in Place to Support Continued Growth Positioned for Significant Value Creation SUMAVEL ® DosePro ™ : Differentiated, Marketed Product • Full Commercial Launch Underway, Marketing Partnership with Astellas, Quarterly Prescription Growth Phase 3 Ongoing: ZX002 for Chronic Pain • Potential To Be 1st Approved Oral, Single-Entity, Controlled Release Formulation of Hydrocodone DosePro™ Delivery System: Proprietary Technology • Validated, Innovative Needle-Free Delivery System • U.S. FDA approval, initial E.U. marketing authorizations • Broad Range of Potential Applications Experienced Management Team • History of Successful Manufacturing, Development and Commercialization |
 [ 5 ] 1 st Single Use, Needle-Free, Commercial Product Marketed for Treating Acute Migraine and Cluster Headaches RAMPING SALES Launched Mid-January 2010 |
 [ 6 ] 75% of Patients Are Women The 9th Leading Cause of Disability in Women Globally 7th Most Costly Disease to US Employers 30 Million People in the U.S. Suffer Migraines** Symptoms and Nature of Episodes Vary Widely Disabling Headache Pain, 4-72 Hours in Duration $25 B Annual Estimated Cost to Employers 63% Suffer 1 Attack per Month 25% Have 1 Attack per Week 48% Upon Morning Wakening (between 4 -9 am) 29% Reported Vomiting as a Symptom of Migraine Attacks Treatment Depends on Type of Episode Migraine Frequency, Severity, Speed of Onset, and Previous Response to Medication Determine Treatment 25% Have 2 Active Prescriptions Oral Triptans Often Prescribed as 1 st Line Therapy Sources: NHF(2010), World Health Organization (2000), Goetzel et all, JOEM (2004), International Headache Society (www.ihs-headache.org), Thomson Medstat (2006), Lipton et al, Neurology (2002), Fox and Davis, Headache (1998), Lipton et al, Neurology (2001), Boston Healthcare Associates, Inc. (2007) |
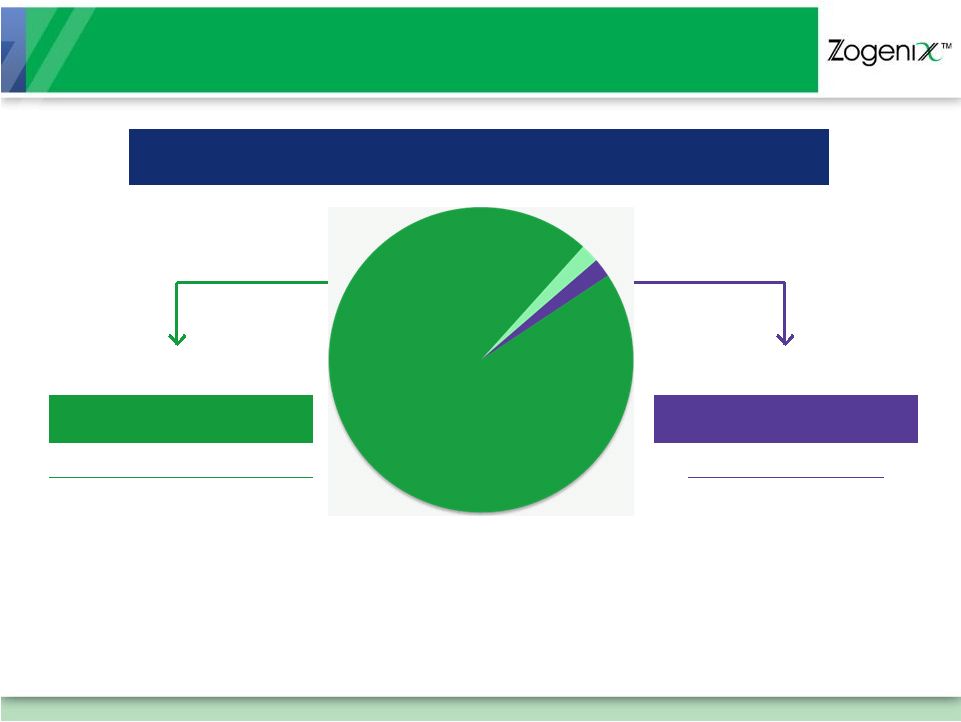 [ 7 ] $3.5 Billion Migraine Market TARGET AUDIENCE: DISSATISFIED ORAL USERS 2% Nasal 2% Injectable* Oral and Nasal 128 Million Doses CONVENIENCE But… Limited Efficacy for Many Attacks Injectable 3.2 Million Doses EFFECTIVE But… 50% of Patients Refuse Needle-based Injection Due to Anxiety, Fear, and Complexity 96% Tablets and Melt Tablets *Sumatriptan Is the Only Injectable Triptan Sources: Palace Healthcare Group, Inc. (2006), Wolters Kluwer Pharma Solutions, Source® PHAST Institution/Retail (12 months ended June 2010) |
 Significant Unmet Needs Patients Want Fast, Complete Pain Relief… … But Most Patients Are Dissatisfied 50% Unsatisfied with Their Current Therapy 75% Said Medication Didn’t Work Fast Enough Majority Said Prescription Oral Medication Not Useful for Every Migraine Attack 30% Fail to Respond to Oral or Nasal Triptans [ 8 ] Sources: Lipton et al, Headache (1999), NHF Survey of 500 Migraine Sufferers (June 2010), Gerson Lehrman Group (2006) |
 SUMAVEL DosePro: A Differentiated Migraine Therapy COMPLETE Pain Free EFFECTIVE Pain Relief EASY TO USE 98% of Patients Correctly Used SUMAVEL DosePro During Migraine Attacks on Their First Try At Home FAST Pain Relief in as Little as 10 Minutes for Some Patients Sources: SUMAVEL DosePro Prescribing Information [ 9 ] |
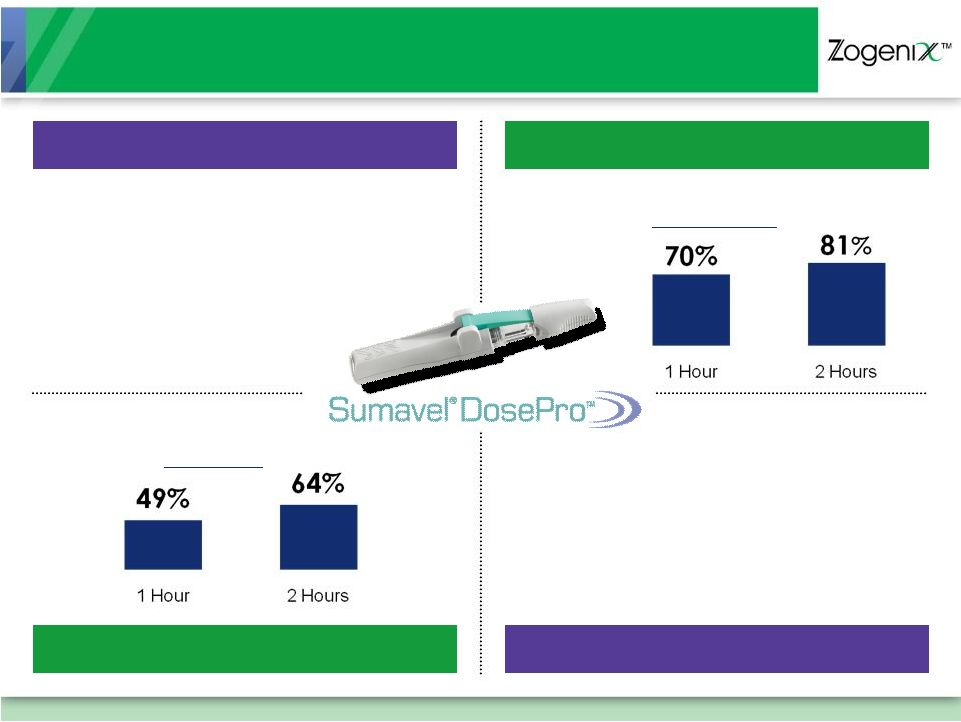
 [ 10 ] Highly Effective Treatment for Migraine Pain Fastest Time to Maximum Drug Concentration in Blood (Hours) Most Effective Patient Pain Relief at 1 Hour 70% 38-43% 28% 20-30% 12 Minutes 1.6 - 2.5 Hours 1 Hour 1.5 Hours ORAL TABLETS ORAL MELT ORAL TABLETS ORAL MELT Sources: Products’ Prescribing Information (sumatriptan injection) (sumatriptan injection) |
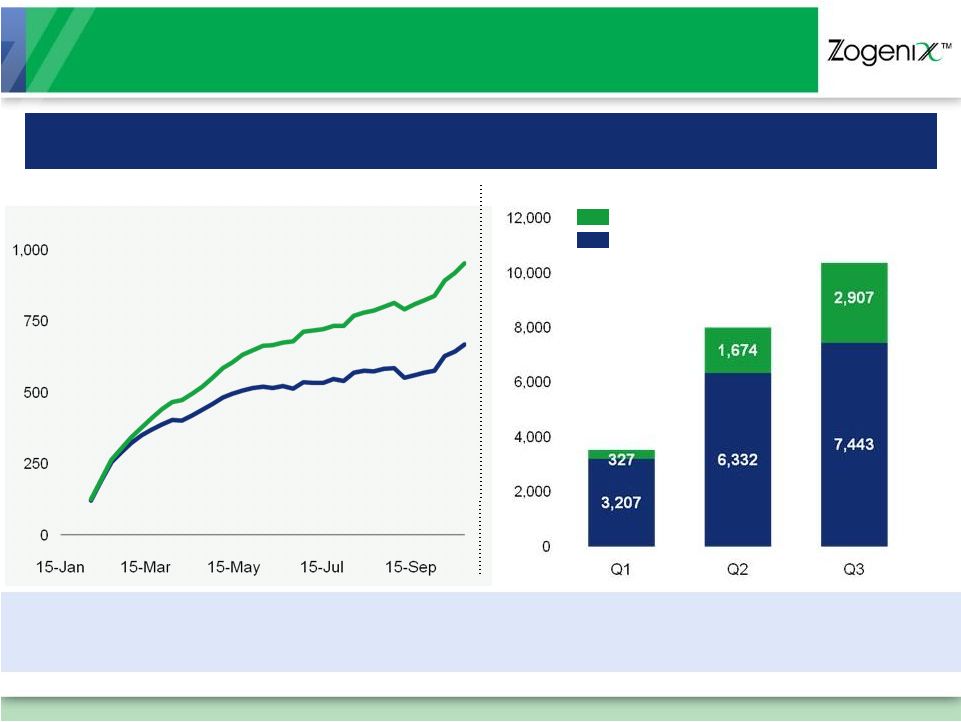 [ 11 ] Rapid, Growing Market Adoption Since January Launch Weekly Prescriptions: Rolling 4-Week Average (Through week ending October 22, 2010) SUMAVEL DosePro Prescriptions: Over 25,000 TRx To Date $1.9M Net Sales $4.2M Net Sales $5.7M Net Sales Refill Prescriptions New Prescriptions Total Prescriptions New Prescriptions Sources: Wolters Kluwer Pharma Solutions (Source® LaunchTrac, (w/e January 15, 2010 – w/e October 22, 2010), Source® PHAST Retail (Jan 2010 – Sep 2010) Refill Prescriptions at 31% of Total (September) Over 6,100 Prescribing Physicians |
 [ 12 ] Market Experience To Date … New Physicians 32% of Prescribing Physicians Had Not Written a Prescription for Needle- Based Sumatriptan Injections in prior 12 months We’re Changing the Way Migraines Are Treated Sources: Wolters Kluwer Pharma Solutions : Source® LaunchTrac (w/e January 15, 2010 – w/e July 30, 2010), Source® Lx PTA (Jan 2010 – Aug 2010); Infomedics, based on 1,071 patient respondents (through September 11, 2010) Patients Switching 77% of Patients Switching Dosage Forms through August were from Oral Triptans Higher Patient Satisfaction Average Patient Satisfaction Score vs. 5.5 (9 point scale) for Prior Migraine Medication; Confirmed by Phase 4 Study Results 7.1 New Patients 34% of Patients Were New to the Triptan Market since launch |
 [ 13 ] SUMAVEL DosePro: CONNECT 2010 Most Innovative Product (MIP) Winner 2010 Winner in Life Sciences – Medical Products category Recognizes emerging technologies and cutting-edge products that drive San Diego’s innovation economy DosePro Technology…as easy as 1-2-3 |
 [ 14 ] Successfully Executing with Well-Established Sales and Marketing Organization ~ 80 Field Sales Reps, Most Have Neurology and/or Migraine Experience Targeting Neurologists, Headache Clinics and Specialists, Select PCPs 12+ Years Average Experience per Sales Rep ~ 400 Field Sales Reps Targeting PCPs, OB/GYNs, EM Physicians and Urologists Multi-Year Co-Marketing and Co-Promotion Agreement $20M Upfront and Milestone Payments Pay-for-Performance Sales Results Positioning SUMAVEL DosePro as a First-Line Alternative Oral Triptan Non-Responders and Dissatisfied Patients Collaboration With Over 475 Sales Representatives |
 [ 15 ] Comprehensive Marketing and Patient Support Programs |
 Broad Reimbursement Coverage Source: Wolters Kluwer Pharma Solutions Source® Dynamic Claims (January 2010 – November 2010) of Reimbursement Claims Submitted Are Approved 79% Tier 3 Reimbursement Strategy Meeting Expectations Broad Coverage with the Largest Commercial Healthcare Plans Range of Physician and Patient Reimbursement and Support Programs ($15 Co-Pay) Non-Retail Initiatives Underway $87 WAC per Unit $522 Sold in 6-Packs $15 Cost to Patient [ 16 ] |
 [ 17 ] High Quality Partnerships Support Domestic and European Commercialization Manufactured and Released Over 1,000,000 SUMAVEL DosePro Units to Date Manufacturing Contract Manufacturers and Component Suppliers Distribution and Marketing Europe |
 [ 18 ] ZX002 1 st Oral, Single-Entity, Controlled Release Hydrocodone for Treating Chronic Pain PHASE 3 DEVELOPMENT |
 ZX002: Filling A Significant Treatment Gap for Chronic Pain Sources: Wolters Kluwer Pharma Solutions, Source® PHAST Retail (12 months ended June 30, 2010), bioStrategies Group (2007) NO Controlled Release Hydrocodone Single Entity Hydrocodone $13 Billion Market (202M TRx) Codeine and Morphine Based Pain Products Hydrocodone Rx Share By Pain Type: ~50% for Chronic Pain Acute Chronic – Other Chronic Back/ OA June 2009 FDA Joint Advisory Committee Meeting Highlighted public health problem of liver injury related to use of acetaminophen in OTC and Rx products $3.1 Billion Annual Sales (126M TRx) Generic, Immediate Release Hydrocodone Combination Products Oral ER Formulations Accepted at Significant Premiums to Generics/IR [ 19 ] |
    [ 20 ] ZX002: Phase 3 Clinical Development Program Study 801 Study 802 Open Label Safety: Enrollment Completed 4Q10 Initiated May 2010, ~55 Sites ~450 patients on therapy Open Label Safety Study to Provide Safety Database for 505(b)(2) NDA: 300 Subjects at 6 Months and 100 Subjects at 1 Year Exposure Broad Spectrum of Chronic Pain Pivotal Phase 3 Efficacy Trial: Enrollment Completion 1Q11 Initiated March 2010, ~60 Sites ~340 patients to be randomized Standard Opioid Trial Design with 12-Weeks Active Treatment Following Dose Titration and Randomization Chronic Lower Back Pain Patients Top Line Data Expected 2H 2011 Intend to Submit NDA by Early 2012 |
 ZX002 Single Entity Hydrocodone, Multiple Strengths (10-50 mg) Uses Elan’s Proprietary SODAS Delivery System to Enhance Hydrocodone Release Profile Planning expansion of existing sales team to 250+ representatives to support ZX002 launch ZX002: A Late Stage Product Candidate with Numerous Benefits Over Existing Opioids Effective12-Hour Pain Relief Enables Dose Optimization and Switching from Other Opioids Improves Adherence and Patient Convenience Provides Consistent, Around-the-Clock Relief Eliminates Safety Risks Associated with Acetaminophen [ 21 ] |
 DosePro: Proprietary Drug Delivery System Validated by FDA Approval of SUMAVEL DosePro 10+ Years Development and Validation US and Foreign IP 61 Issued Patents 31 Patent Applications Pending Proven Ability to Deliver Biologics and Highly Viscous Formulations [ 22 ] |
 DosePro: Broad Range of Potential Applications Attractive Delivery Option for Additional Medications, Including Biologics and Small Molecules DosePro Internal Development Injectable CNS Drug Product Candidates (2 Pre-Clinical) Second Generation Technology (1mL device) Technology Out Licensing Enhance, Differentiate or Extend Injectable Product Lifecycles Prototypes of Second Generation DosePro [ 23 ] |
 [ 24 ] TRx Market 1.26 Million TRx $244 Million 48% of Episodes Upon Morning Wakening (between 4 -9 am) 29% of Sufferers Reported Vomiting as a Symptom of Migraine Attacks 30% of Patients Fail to Respond to Oral or Nasal Triptans Net Revenue/Dose: $70/Unit* 7 Units/TRx Average TRx Value: $490 12.1 Million TRx per Year 1% Triptan Share ZX002 126M Hydrocodone (~50% for Chronic Use) 76M Other ER and IR Opioids Net Revenue/Day: $7.75** Average TRx: 25 Days Average TRx Value: $194 202 Million TRx per Year 1% Hydrocodone Share Large Commercial Opportunities Patient/ Market Segments Prescription Values *Includes Co-Pay Assistance ** Based on current pricing of branded ER opioids Sources: Wolters Kluwer Pharma Solutions , Source® PHAST Retail (12 months ended June 30, 2010) Illustrative Market Penetration 121,000 TRx $59 Million |
 Financial Summary 9 months ended September 30, 2010 P&L $ Millions Financial Highlights $ Millions Revenue $ 14.6 Operating Expense $ 66.2 Loss From Operations $ 51.6 Other (Income)/Loss $ 19.9 Net Loss $ 71.5 Gross Invoiced Sales $ 16.4 Invested Capital $ 164.0 Cash Balance $ 11.7 Debt Facility $25M Term Loan $10M Working Capital Line • Balance $2.7M Cash Flow Used In Operations $ 58.5 [ 25 ] |
 2010 Achievements SUMAVEL DosePro Full U.S. Commercial Launch by Zogenix with co-promote partner Astellas Enhanced Formulary Coverage and Patient Support Phase IV Study Completed Consistent Monthly Rx Growth Since Launch 1 Million Commercial Units Produced Initial EU Approvals by Desitin Named a 2010 “Most Innovative Product” Winner by CONNECT ZX002 Initiated Phase 3 Safety Study (Study 802) Initiated Phase 3 Efficacy Study (Study 801) Completed Enrollment in Phase 3 Safety Study Corporate Appointed Ann D. Rhoads to Serve as CFO Established Term Loan and Revolving Line of Credit Completed Initial Public Offering [ 26 ] |
 [ 27 ] 2011 Goals and Milestones SUMAVEL DosePro Continue US Sales Growth Phase IV Data Publication Additional EU Approvals New Direct-to-Patient Initiatives ZX002 Pivotal Study 801 Efficacy Top– Line Results Open Label Study 802 Top-Line Safety Results Initiate Co-Promotion Discussions Pre-NDA Meeting with FDA Prepare to File NDA by early 2012 DosePro Technology File IND for Next Zogenix CNS Product Seek First Licensee for Biologic Building on Track Record of Successful Execution |
 [ 28 ] Experienced Management Team with a History of Successful Development & Commercialization *Co-Founders Aradigm, Invacare 19 John Turanin* VP/GM, Zogenix Technologies GlaxoSmithKline, Elan, Skin Medica, InterMune 30 Mark Thompson VP, Sales & Managed Markets Nektar, Connetics 24 Edward Smith, Ph.D., RAC VP, Regulatory Affairs Avera, Windamere 17 Bret Megargel* VP, Corporate Dev Elan, Allergan, Valient 12 Stephen Jenner VP, Marketing Elan, InterMune 19 Cynthia Robinson, Ph.D. CDO Premier, Sprout, DLJ, Bain & Co, Merrill Lynch 18 Ann Rhoads CFO Aradigm, Cardiff University 20 Stephen Farr, Ph.D.* President & COO GlaxoSmithKline, Elan, InterMune 23 Roger Hawley* CEO Years Experience |
 [ 29 ] Established Commercial Infrastructure • Sophisticated Manufacturing and Sales Organization in Place to Support Continued Growth Positioned for Significant Value Creation SUMAVEL ® DosePro ™ : Differentiated, Marketed Product • Full Commercial Launch Underway, Marketing Partnership with Astellas, Quarterly Prescription Growth Phase 3 Ongoing: ZX002 for Chronic Pain • Potential To Be 1st Approved Oral, Single-Entity, Controlled Release Formulation of Hydrocodone DosePro™ Delivery System: Proprietary Technology • Validated, Innovative Needle-Free Delivery System • U.S. FDA approval, initial E.U. marketing authorizations • Broad Range of Potential Applications Experienced Management Team • History of Successful Manufacturing, Development and Commercialization |